User login
Late-Onset Acrokeratosis Paraneoplastica of Bazex Associated With Metastatic Adenocarcinoma of the Colon
To the Editor:
Acrokeratosis paraneoplastica (AP) of Bazex is a rare but distinctive acral psoriasiform dermatosis associated with internal malignancy, usually squamous cell carcinoma (SCC), of the upper aerodigestive tract.1,2 Recognizing this paraneoplastic condition is paramount because cutaneous findings often precede the onset of symptoms associated with an occult malignancy.3
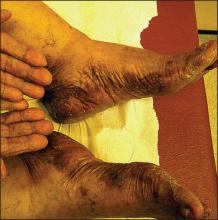
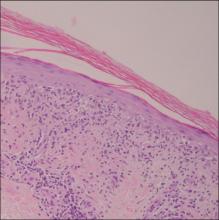
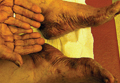
A 76-year-old woman with adenocarcinoma of the transverse colon of 3 years’ duration was referred to the dermatology department. She had a hemicolectomy and was doing well until tumor recurrence with peritoneal metastasis was detected following an exploratory laparotomy 1 year prior to presentation to us. She underwent a total hysterectomy and bilateral salpingo-oophorectomy. Palliative chemotherapy was initiated, and she completed 5 cycles of capecitabine and oxaliplatin. Long-term medications for diabetes mellitus, hypertension, and hyperlipidemia included glibenclamide, nifedipine, hydrochlorothiazide, and losartan. The patient had a progressive pruritic rash of 6 months’ duration that started on the hands and forearms and spread to involve the feet and lower limbs as well as the ears and face. She also experienced progressive thickening of the palms and soles. She had been treated with topical steroids and emollients with no improvement. Clinical examination revealed erythematous scaly patches on all of her limbs, especially on the elbows and knees, and on the ear helices and nose. There also was notable palmoplantar keratoderma with central sparing (Figure 1), onycholysis, and subungual hyperkeratosis. A skin biopsy of the forearm was performed, and histology revealed orthokeratosis, hypergranulosis and basal vacuolar alteration, and superficial perivascular lymphohistiocytic infiltrate with melanophages (Figure 2). Direct immunofluorescence studies and fungal cultures were negative. The cutaneous features of a treatment-resistant acral dermatosis supported the clinical diagnosis of AP, especially in the setting of an internal malignancy. The patient was started on palliative radiotherapy with no notable resolution of the cutaneous lesions. She was lost to follow-up.
First described by Bazex et al1 in 1965, AP is an uncommon but well-recognized paraneoplastic dermatosis associated with an underlying neoplasm. Fewer than 160 cases have been reported, and the majority of cases have been men older than 40 years. The most commonly associated malignancy was SCC (at least 50% of reported cases) involving mainly the oropharynx and larynx, with lung and esophageal SCC also described.2-4 Only a few cases of adenocarcinoma-associated AP have been described, such as adenocarcinoma of the lung, prostate, and stomach.3-5 In a reported case of AP associated with early colon adenocarcinoma, the patient had remarkable cutaneous resolution following successful tumor resection.6 Other reported rare hematologic associations included Hodgkin disease, peripheral T-cell lymphoma, and multiple myeloma.3-5 Karabulut et al4 described a case associated with cholangiocarcinoma and studied the primary sites of malignancies in another 133 patients with AP (118 patients with documented cell type): oropharynx and larynx in 55 (41%); lung in 23 (17%); unknown location in 20 (15%); esophagus in 14 (11%); prostate in 3 (2%); stomach in 3 (2%); and isolated cases involving the liver, thymus, uterus, vulva, breast, urinary bladder, lymph nodes, and bone marrow.
A review of 113 cases of AP showed that only 15% developed cutaneous lesions after the malignancy had been discovered; in the majority of cases (67%), cutaneous lesions were present for an average of 1 year preceding the diagnosis of malignancy.3 In our patient, the late-onset cutaneous involvement corresponded to the progression of the underlying colon adenocarcinoma. In the absence of initial cutaneous involvement or when successful treatment of a tumor results in cutaneous resolution, subsequent emergence of cutaneous lesions of AP signifies tumor progression. The evolution of cutaneous features in AP was well described by Bazex et al.1 Stage 1 of the disease shows initial erythema and psoriasiform scaling of the fingers and toes, spreading to the nose and ear helices (hence acrokeratosis); the tumor frequently remains asymptomatic or undetected. Stage 2 shows palmoplantar keratoderma with central sparing and more extensive facial lesions; progression to stage 3 occurs if the tumor remains undetected or untreated, with further spread of psoriasiform lesions to the elbows, knees, trunk, and scalp.1 Nail involvement occurs in nearly 75% of cases5; typical changes include subungual hyperkeratosis, onycholysis, longitudinal streaks, yellow pigmentation, and rarely onychomadesis. A high index of suspicion of AP is paramount when evaluating any recalcitrant acral dermatosis that fails to respond to appropriate therapy, especially in the presence of constitutional symptoms, typical bulbous enlargement of distal phalanges, or isolated involvement of helices. These findings should prompt physicians to perform an extensive search for an underlying malignancy with complete physical examination, particularly of the head and neck region, with appropriate endoscopic assessment and imaging studies.
A myriad of nonspecific histologic features of AP commonly reported include hyperkeratosis, parakeratosis, acanthosis, and dermal perivascular lymphohistiocytic infiltrate.7 Less common features include dyskeratotic keratinocytes, vacuolar degeneration, bandlike infiltrate, and melanin incontinence.7 The pathogenesis of AP remains elusive. A postulated immunologic mechanism is based on reports of immunoglobulins (IgG, IgA, IgM) and complement (C3) deposition along the basement membrane zone.8 Association with autoimmune disorders such as alopecia areata and vitiligo also has been reported.9 Another possible mechanism is cross-reactivity between antigens found in the tumor and skin, resulting in a T-cell–mediated immune response to tumorlike antigens in the epidermis, or secretion of tumor-originating growth factors responsible for the hyperkeratotic skin changes, such as epidermal growth factor, transforming growth factor a, or insulinlike growth factor 1.3,7,10,11
Spontaneous remission of cutaneous lesions in untreated underlying malignancy is rare. Isolated reports of treatment using topical and systemic steroids, salicylic acid, topical vitamin D analogues, etretinate, and psoralen plus UVA showed minimal improvement.5,7 The mainstay in attaining cutaneous resolution is to detect and eradicate the underlying neoplasm with surgery, chemotherapy, or radiotherapy, or combination therapy.
Our case is noteworthy because of the patient’s gender (female), underlying malignancy (adenocarcinoma of the colon), and late onset of cutaneous involvement, which are all uncommon associations related to paraneoplastic syndrome. The clinical features of AP should be recognized early to facilitate an extensive search for an occult malignancy, and late-onset cutaneous involvement also should be recognized as a marker of tumor relapse or progression.
1. Bazex A, Salvador R, Dupré A, et al. Late symptomatic hepatic porphyria developing to the picture of lipoidoproteinsosis [in French]. Bull Soc Fr Dermatol Syphiligr. 1965;72:182.
2. Witkowski JA, Parish LC. Bazex’s syndrome. paraneoplastic acrokeratosis. JAMA. 1982;248:2883-2884.
3. Bolognia JL. Bazex syndrome: acrokeratosis paraneoplastica. Semin Dermatol. 1995;14:84-89.
4. Karabulut AA, Sahin S, Sahin M, et al. Paraneoplastic acrokeratosis of Bazex (Bazex’s syndrome): report of a female case associated with cholangiocarcinoma and review of the published work. J Dermatol. 2006;33:850-854.
5. Valdivielso M, Longo I, Suárez R. Acrokeratosis paraneoplastica: Bazex syndrome. J Eur Acad Dermatol Venereol. 2005;19:340-344.
6. Hsu YS, Lien GS, Lai HH, et al. Acrokeratosis paraneoplastica (Bazex syndrome) with adenocarcinoma of the colon: report of a case and review of the literature. J Gastroenterol. 2000;35:460-464.
7. Bolognia JL, Brewer YP, Cooper DL. Bazex syndrome (acrokeratosis paraneoplastica). an analytic review. Medicine (Baltimore). 1991;70:269-280.
8. Mutasim DF, Meiri G. Bazex syndrome mimicking a primary autoimmune bullous disorder. J Am Acad Dermatol. 1999;40(5, pt 2):822-825.
9. Hara M, Hunayama M, Aiba S, et al. Acrokeratosis paraneoplastica (Bazex syndrome) associated with primary cutaneous squamous cell carcinoma of the lower leg, vitiligo and alopecia areata. Br J Dermatol. 1995;133:121-124.
10. Stone SP, Buescher LS. Life-threatening paraneoplastic cutaneous syndromes. Clin Dermatol. 2005;23:301-306.
11. Politi Y, Ophir J, Brenner S. Cutaneous paraneoplastic syndromes. Acta Derm Venereol. 1993;73:161-170.
To the Editor:
Acrokeratosis paraneoplastica (AP) of Bazex is a rare but distinctive acral psoriasiform dermatosis associated with internal malignancy, usually squamous cell carcinoma (SCC), of the upper aerodigestive tract.1,2 Recognizing this paraneoplastic condition is paramount because cutaneous findings often precede the onset of symptoms associated with an occult malignancy.3
A 76-year-old woman with adenocarcinoma of the transverse colon of 3 years’ duration was referred to the dermatology department. She had a hemicolectomy and was doing well until tumor recurrence with peritoneal metastasis was detected following an exploratory laparotomy 1 year prior to presentation to us. She underwent a total hysterectomy and bilateral salpingo-oophorectomy. Palliative chemotherapy was initiated, and she completed 5 cycles of capecitabine and oxaliplatin. Long-term medications for diabetes mellitus, hypertension, and hyperlipidemia included glibenclamide, nifedipine, hydrochlorothiazide, and losartan. The patient had a progressive pruritic rash of 6 months’ duration that started on the hands and forearms and spread to involve the feet and lower limbs as well as the ears and face. She also experienced progressive thickening of the palms and soles. She had been treated with topical steroids and emollients with no improvement. Clinical examination revealed erythematous scaly patches on all of her limbs, especially on the elbows and knees, and on the ear helices and nose. There also was notable palmoplantar keratoderma with central sparing (Figure 1), onycholysis, and subungual hyperkeratosis. A skin biopsy of the forearm was performed, and histology revealed orthokeratosis, hypergranulosis and basal vacuolar alteration, and superficial perivascular lymphohistiocytic infiltrate with melanophages (Figure 2). Direct immunofluorescence studies and fungal cultures were negative. The cutaneous features of a treatment-resistant acral dermatosis supported the clinical diagnosis of AP, especially in the setting of an internal malignancy. The patient was started on palliative radiotherapy with no notable resolution of the cutaneous lesions. She was lost to follow-up.
First described by Bazex et al1 in 1965, AP is an uncommon but well-recognized paraneoplastic dermatosis associated with an underlying neoplasm. Fewer than 160 cases have been reported, and the majority of cases have been men older than 40 years. The most commonly associated malignancy was SCC (at least 50% of reported cases) involving mainly the oropharynx and larynx, with lung and esophageal SCC also described.2-4 Only a few cases of adenocarcinoma-associated AP have been described, such as adenocarcinoma of the lung, prostate, and stomach.3-5 In a reported case of AP associated with early colon adenocarcinoma, the patient had remarkable cutaneous resolution following successful tumor resection.6 Other reported rare hematologic associations included Hodgkin disease, peripheral T-cell lymphoma, and multiple myeloma.3-5 Karabulut et al4 described a case associated with cholangiocarcinoma and studied the primary sites of malignancies in another 133 patients with AP (118 patients with documented cell type): oropharynx and larynx in 55 (41%); lung in 23 (17%); unknown location in 20 (15%); esophagus in 14 (11%); prostate in 3 (2%); stomach in 3 (2%); and isolated cases involving the liver, thymus, uterus, vulva, breast, urinary bladder, lymph nodes, and bone marrow.
A review of 113 cases of AP showed that only 15% developed cutaneous lesions after the malignancy had been discovered; in the majority of cases (67%), cutaneous lesions were present for an average of 1 year preceding the diagnosis of malignancy.3 In our patient, the late-onset cutaneous involvement corresponded to the progression of the underlying colon adenocarcinoma. In the absence of initial cutaneous involvement or when successful treatment of a tumor results in cutaneous resolution, subsequent emergence of cutaneous lesions of AP signifies tumor progression. The evolution of cutaneous features in AP was well described by Bazex et al.1 Stage 1 of the disease shows initial erythema and psoriasiform scaling of the fingers and toes, spreading to the nose and ear helices (hence acrokeratosis); the tumor frequently remains asymptomatic or undetected. Stage 2 shows palmoplantar keratoderma with central sparing and more extensive facial lesions; progression to stage 3 occurs if the tumor remains undetected or untreated, with further spread of psoriasiform lesions to the elbows, knees, trunk, and scalp.1 Nail involvement occurs in nearly 75% of cases5; typical changes include subungual hyperkeratosis, onycholysis, longitudinal streaks, yellow pigmentation, and rarely onychomadesis. A high index of suspicion of AP is paramount when evaluating any recalcitrant acral dermatosis that fails to respond to appropriate therapy, especially in the presence of constitutional symptoms, typical bulbous enlargement of distal phalanges, or isolated involvement of helices. These findings should prompt physicians to perform an extensive search for an underlying malignancy with complete physical examination, particularly of the head and neck region, with appropriate endoscopic assessment and imaging studies.
A myriad of nonspecific histologic features of AP commonly reported include hyperkeratosis, parakeratosis, acanthosis, and dermal perivascular lymphohistiocytic infiltrate.7 Less common features include dyskeratotic keratinocytes, vacuolar degeneration, bandlike infiltrate, and melanin incontinence.7 The pathogenesis of AP remains elusive. A postulated immunologic mechanism is based on reports of immunoglobulins (IgG, IgA, IgM) and complement (C3) deposition along the basement membrane zone.8 Association with autoimmune disorders such as alopecia areata and vitiligo also has been reported.9 Another possible mechanism is cross-reactivity between antigens found in the tumor and skin, resulting in a T-cell–mediated immune response to tumorlike antigens in the epidermis, or secretion of tumor-originating growth factors responsible for the hyperkeratotic skin changes, such as epidermal growth factor, transforming growth factor a, or insulinlike growth factor 1.3,7,10,11
Spontaneous remission of cutaneous lesions in untreated underlying malignancy is rare. Isolated reports of treatment using topical and systemic steroids, salicylic acid, topical vitamin D analogues, etretinate, and psoralen plus UVA showed minimal improvement.5,7 The mainstay in attaining cutaneous resolution is to detect and eradicate the underlying neoplasm with surgery, chemotherapy, or radiotherapy, or combination therapy.
Our case is noteworthy because of the patient’s gender (female), underlying malignancy (adenocarcinoma of the colon), and late onset of cutaneous involvement, which are all uncommon associations related to paraneoplastic syndrome. The clinical features of AP should be recognized early to facilitate an extensive search for an occult malignancy, and late-onset cutaneous involvement also should be recognized as a marker of tumor relapse or progression.
To the Editor:
Acrokeratosis paraneoplastica (AP) of Bazex is a rare but distinctive acral psoriasiform dermatosis associated with internal malignancy, usually squamous cell carcinoma (SCC), of the upper aerodigestive tract.1,2 Recognizing this paraneoplastic condition is paramount because cutaneous findings often precede the onset of symptoms associated with an occult malignancy.3
A 76-year-old woman with adenocarcinoma of the transverse colon of 3 years’ duration was referred to the dermatology department. She had a hemicolectomy and was doing well until tumor recurrence with peritoneal metastasis was detected following an exploratory laparotomy 1 year prior to presentation to us. She underwent a total hysterectomy and bilateral salpingo-oophorectomy. Palliative chemotherapy was initiated, and she completed 5 cycles of capecitabine and oxaliplatin. Long-term medications for diabetes mellitus, hypertension, and hyperlipidemia included glibenclamide, nifedipine, hydrochlorothiazide, and losartan. The patient had a progressive pruritic rash of 6 months’ duration that started on the hands and forearms and spread to involve the feet and lower limbs as well as the ears and face. She also experienced progressive thickening of the palms and soles. She had been treated with topical steroids and emollients with no improvement. Clinical examination revealed erythematous scaly patches on all of her limbs, especially on the elbows and knees, and on the ear helices and nose. There also was notable palmoplantar keratoderma with central sparing (Figure 1), onycholysis, and subungual hyperkeratosis. A skin biopsy of the forearm was performed, and histology revealed orthokeratosis, hypergranulosis and basal vacuolar alteration, and superficial perivascular lymphohistiocytic infiltrate with melanophages (Figure 2). Direct immunofluorescence studies and fungal cultures were negative. The cutaneous features of a treatment-resistant acral dermatosis supported the clinical diagnosis of AP, especially in the setting of an internal malignancy. The patient was started on palliative radiotherapy with no notable resolution of the cutaneous lesions. She was lost to follow-up.
First described by Bazex et al1 in 1965, AP is an uncommon but well-recognized paraneoplastic dermatosis associated with an underlying neoplasm. Fewer than 160 cases have been reported, and the majority of cases have been men older than 40 years. The most commonly associated malignancy was SCC (at least 50% of reported cases) involving mainly the oropharynx and larynx, with lung and esophageal SCC also described.2-4 Only a few cases of adenocarcinoma-associated AP have been described, such as adenocarcinoma of the lung, prostate, and stomach.3-5 In a reported case of AP associated with early colon adenocarcinoma, the patient had remarkable cutaneous resolution following successful tumor resection.6 Other reported rare hematologic associations included Hodgkin disease, peripheral T-cell lymphoma, and multiple myeloma.3-5 Karabulut et al4 described a case associated with cholangiocarcinoma and studied the primary sites of malignancies in another 133 patients with AP (118 patients with documented cell type): oropharynx and larynx in 55 (41%); lung in 23 (17%); unknown location in 20 (15%); esophagus in 14 (11%); prostate in 3 (2%); stomach in 3 (2%); and isolated cases involving the liver, thymus, uterus, vulva, breast, urinary bladder, lymph nodes, and bone marrow.
A review of 113 cases of AP showed that only 15% developed cutaneous lesions after the malignancy had been discovered; in the majority of cases (67%), cutaneous lesions were present for an average of 1 year preceding the diagnosis of malignancy.3 In our patient, the late-onset cutaneous involvement corresponded to the progression of the underlying colon adenocarcinoma. In the absence of initial cutaneous involvement or when successful treatment of a tumor results in cutaneous resolution, subsequent emergence of cutaneous lesions of AP signifies tumor progression. The evolution of cutaneous features in AP was well described by Bazex et al.1 Stage 1 of the disease shows initial erythema and psoriasiform scaling of the fingers and toes, spreading to the nose and ear helices (hence acrokeratosis); the tumor frequently remains asymptomatic or undetected. Stage 2 shows palmoplantar keratoderma with central sparing and more extensive facial lesions; progression to stage 3 occurs if the tumor remains undetected or untreated, with further spread of psoriasiform lesions to the elbows, knees, trunk, and scalp.1 Nail involvement occurs in nearly 75% of cases5; typical changes include subungual hyperkeratosis, onycholysis, longitudinal streaks, yellow pigmentation, and rarely onychomadesis. A high index of suspicion of AP is paramount when evaluating any recalcitrant acral dermatosis that fails to respond to appropriate therapy, especially in the presence of constitutional symptoms, typical bulbous enlargement of distal phalanges, or isolated involvement of helices. These findings should prompt physicians to perform an extensive search for an underlying malignancy with complete physical examination, particularly of the head and neck region, with appropriate endoscopic assessment and imaging studies.
A myriad of nonspecific histologic features of AP commonly reported include hyperkeratosis, parakeratosis, acanthosis, and dermal perivascular lymphohistiocytic infiltrate.7 Less common features include dyskeratotic keratinocytes, vacuolar degeneration, bandlike infiltrate, and melanin incontinence.7 The pathogenesis of AP remains elusive. A postulated immunologic mechanism is based on reports of immunoglobulins (IgG, IgA, IgM) and complement (C3) deposition along the basement membrane zone.8 Association with autoimmune disorders such as alopecia areata and vitiligo also has been reported.9 Another possible mechanism is cross-reactivity between antigens found in the tumor and skin, resulting in a T-cell–mediated immune response to tumorlike antigens in the epidermis, or secretion of tumor-originating growth factors responsible for the hyperkeratotic skin changes, such as epidermal growth factor, transforming growth factor a, or insulinlike growth factor 1.3,7,10,11
Spontaneous remission of cutaneous lesions in untreated underlying malignancy is rare. Isolated reports of treatment using topical and systemic steroids, salicylic acid, topical vitamin D analogues, etretinate, and psoralen plus UVA showed minimal improvement.5,7 The mainstay in attaining cutaneous resolution is to detect and eradicate the underlying neoplasm with surgery, chemotherapy, or radiotherapy, or combination therapy.
Our case is noteworthy because of the patient’s gender (female), underlying malignancy (adenocarcinoma of the colon), and late onset of cutaneous involvement, which are all uncommon associations related to paraneoplastic syndrome. The clinical features of AP should be recognized early to facilitate an extensive search for an occult malignancy, and late-onset cutaneous involvement also should be recognized as a marker of tumor relapse or progression.
1. Bazex A, Salvador R, Dupré A, et al. Late symptomatic hepatic porphyria developing to the picture of lipoidoproteinsosis [in French]. Bull Soc Fr Dermatol Syphiligr. 1965;72:182.
2. Witkowski JA, Parish LC. Bazex’s syndrome. paraneoplastic acrokeratosis. JAMA. 1982;248:2883-2884.
3. Bolognia JL. Bazex syndrome: acrokeratosis paraneoplastica. Semin Dermatol. 1995;14:84-89.
4. Karabulut AA, Sahin S, Sahin M, et al. Paraneoplastic acrokeratosis of Bazex (Bazex’s syndrome): report of a female case associated with cholangiocarcinoma and review of the published work. J Dermatol. 2006;33:850-854.
5. Valdivielso M, Longo I, Suárez R. Acrokeratosis paraneoplastica: Bazex syndrome. J Eur Acad Dermatol Venereol. 2005;19:340-344.
6. Hsu YS, Lien GS, Lai HH, et al. Acrokeratosis paraneoplastica (Bazex syndrome) with adenocarcinoma of the colon: report of a case and review of the literature. J Gastroenterol. 2000;35:460-464.
7. Bolognia JL, Brewer YP, Cooper DL. Bazex syndrome (acrokeratosis paraneoplastica). an analytic review. Medicine (Baltimore). 1991;70:269-280.
8. Mutasim DF, Meiri G. Bazex syndrome mimicking a primary autoimmune bullous disorder. J Am Acad Dermatol. 1999;40(5, pt 2):822-825.
9. Hara M, Hunayama M, Aiba S, et al. Acrokeratosis paraneoplastica (Bazex syndrome) associated with primary cutaneous squamous cell carcinoma of the lower leg, vitiligo and alopecia areata. Br J Dermatol. 1995;133:121-124.
10. Stone SP, Buescher LS. Life-threatening paraneoplastic cutaneous syndromes. Clin Dermatol. 2005;23:301-306.
11. Politi Y, Ophir J, Brenner S. Cutaneous paraneoplastic syndromes. Acta Derm Venereol. 1993;73:161-170.
1. Bazex A, Salvador R, Dupré A, et al. Late symptomatic hepatic porphyria developing to the picture of lipoidoproteinsosis [in French]. Bull Soc Fr Dermatol Syphiligr. 1965;72:182.
2. Witkowski JA, Parish LC. Bazex’s syndrome. paraneoplastic acrokeratosis. JAMA. 1982;248:2883-2884.
3. Bolognia JL. Bazex syndrome: acrokeratosis paraneoplastica. Semin Dermatol. 1995;14:84-89.
4. Karabulut AA, Sahin S, Sahin M, et al. Paraneoplastic acrokeratosis of Bazex (Bazex’s syndrome): report of a female case associated with cholangiocarcinoma and review of the published work. J Dermatol. 2006;33:850-854.
5. Valdivielso M, Longo I, Suárez R. Acrokeratosis paraneoplastica: Bazex syndrome. J Eur Acad Dermatol Venereol. 2005;19:340-344.
6. Hsu YS, Lien GS, Lai HH, et al. Acrokeratosis paraneoplastica (Bazex syndrome) with adenocarcinoma of the colon: report of a case and review of the literature. J Gastroenterol. 2000;35:460-464.
7. Bolognia JL, Brewer YP, Cooper DL. Bazex syndrome (acrokeratosis paraneoplastica). an analytic review. Medicine (Baltimore). 1991;70:269-280.
8. Mutasim DF, Meiri G. Bazex syndrome mimicking a primary autoimmune bullous disorder. J Am Acad Dermatol. 1999;40(5, pt 2):822-825.
9. Hara M, Hunayama M, Aiba S, et al. Acrokeratosis paraneoplastica (Bazex syndrome) associated with primary cutaneous squamous cell carcinoma of the lower leg, vitiligo and alopecia areata. Br J Dermatol. 1995;133:121-124.
10. Stone SP, Buescher LS. Life-threatening paraneoplastic cutaneous syndromes. Clin Dermatol. 2005;23:301-306.
11. Politi Y, Ophir J, Brenner S. Cutaneous paraneoplastic syndromes. Acta Derm Venereol. 1993;73:161-170.
Practice Question Answers: Allergic Contact Dermatitis, Part 1
1. Patients with a documented contact allergy to caine mix should avoid all of the following except:
a. benzocaine
b. butacaine
c. lidocaine
d. procaine
e. tetracaine
2. A patient with atopic dermatitis whose condition is worsening with the use of topical steroids is referred for patch testing and found to have positivity to budesonide. Which of the following would be an appropriate topical steroid to prescribe to this patient?
a. desonide
b. desoximetasone
c. fluocinolone
d. fluocinonide
e. triamcinolone
3. A patient with a documented contact allergy to carba mix should avoid which of the following systemic medications?
a. ciprofloxacin
b. disulfiram
c. gold sodium thiomalate
d. hydroxyzine
e. piroxicam
4. Bronopol (2-bromo-2-nitropropane-1,3-diol) can cross-react with all of the following except:
a. diazolidinyl urea
b. DMDM hydantoin
c. imidazolidinyl urea
d. p-aminobenzoic acid
e. quaternium-15
5. Allergy to cocamidopropyl betaine is thought to be secondary to which of the following?
a. amidoamine
b. benzoic acid
c. bronopol
d. Myroxylon pereirae
e. N-isopropyl-N'-phenyl parapheylenediamine
1. Patients with a documented contact allergy to caine mix should avoid all of the following except:
a. benzocaine
b. butacaine
c. lidocaine
d. procaine
e. tetracaine
2. A patient with atopic dermatitis whose condition is worsening with the use of topical steroids is referred for patch testing and found to have positivity to budesonide. Which of the following would be an appropriate topical steroid to prescribe to this patient?
a. desonide
b. desoximetasone
c. fluocinolone
d. fluocinonide
e. triamcinolone
3. A patient with a documented contact allergy to carba mix should avoid which of the following systemic medications?
a. ciprofloxacin
b. disulfiram
c. gold sodium thiomalate
d. hydroxyzine
e. piroxicam
4. Bronopol (2-bromo-2-nitropropane-1,3-diol) can cross-react with all of the following except:
a. diazolidinyl urea
b. DMDM hydantoin
c. imidazolidinyl urea
d. p-aminobenzoic acid
e. quaternium-15
5. Allergy to cocamidopropyl betaine is thought to be secondary to which of the following?
a. amidoamine
b. benzoic acid
c. bronopol
d. Myroxylon pereirae
e. N-isopropyl-N'-phenyl parapheylenediamine
1. Patients with a documented contact allergy to caine mix should avoid all of the following except:
a. benzocaine
b. butacaine
c. lidocaine
d. procaine
e. tetracaine
2. A patient with atopic dermatitis whose condition is worsening with the use of topical steroids is referred for patch testing and found to have positivity to budesonide. Which of the following would be an appropriate topical steroid to prescribe to this patient?
a. desonide
b. desoximetasone
c. fluocinolone
d. fluocinonide
e. triamcinolone
3. A patient with a documented contact allergy to carba mix should avoid which of the following systemic medications?
a. ciprofloxacin
b. disulfiram
c. gold sodium thiomalate
d. hydroxyzine
e. piroxicam
4. Bronopol (2-bromo-2-nitropropane-1,3-diol) can cross-react with all of the following except:
a. diazolidinyl urea
b. DMDM hydantoin
c. imidazolidinyl urea
d. p-aminobenzoic acid
e. quaternium-15
5. Allergy to cocamidopropyl betaine is thought to be secondary to which of the following?
a. amidoamine
b. benzoic acid
c. bronopol
d. Myroxylon pereirae
e. N-isopropyl-N'-phenyl parapheylenediamine
Allergic Contact Dermatitis, Part 1
Midodrine-Induced Acute Generalized Exanthematous Pustulosis
Lichenoid Photosensitivity: An Unusual Reaction to Doxycycline and an Unusual Response
New Developments in Comorbidities of Atopic Dermatitis
Practice Question Answers: Medications in Dermatology, Part 2
1. A 40-year-old woman is diagnosed with systemic lupus erythematosus. You discuss treatment options and decide to start hydroxychloroquine. What laboratory tests and monitoring are required prior to starting this medication?
a. complete blood cell count with differential and glucose-6-phosphate dehydrogenase
b. complete blood cell count with differential and complete metabolic profile
c. ophthalmology evaluation and glucose-6-phosphate dehydrogenase
d. b and c
2. Two months ago you saw a 30-year-old woman with a history of severe atopic dermatitis. She had been using topical steroids with not much improvement. You decided to start a systemic medication. Within 1 month of drug initiation, she called your office to tell you that she is much better but has noticed unwanted hair on her face lately. Which medication is most likely implicated?
a. cyclosporine
b. dapsone
c. hydroxychloroquine
d. methotrexate
3. A 70-year-old man with type 2 diabetes mellitus who drinks 10 cans of beer per week presents to the emergency department with a 3-day history of diffuse tense bullae and pruritus on the legs and trunk. Direct immunofluorescence displayed linear deposition of IgG and C3 at the dermoepidermal junction, confirming your clinical diagnosis. What is the best long-term treatment option for this patient?
a. combination of oral steroids plus methotrexate
b. oral steroids and mycophenolate mofetil
c. oral steroids only
d. topical steroids only
4. A 45-year-old Venezuelan man presents with painful nodules on his bilateral lower legs. A biopsy demonstrates acid-fast bacilli, and a multidrug regimen is initiated for erythema nodosum leprosum. Which of the following is the mechanism of action of the treatment that is US Food and Drug Administration approved for this condition?
a. inhibits chemotaxis
b. inhibits dihydrofolate reductase
c. inhibits tumor necrosis factor α
d. suppresses T-cell function and B-cell antibody production
5. A patient consults her physician because of several side effects from a medication she started 2 weeks ago due to erythematous to violaceous papules on the legs from palpable purpura. She reports diarrhea, abdominal pain, and fatigue. Which medication is she taking?
a. azathioprine
b. colchicine
c. dapsone
d. methotrexate
1. A 40-year-old woman is diagnosed with systemic lupus erythematosus. You discuss treatment options and decide to start hydroxychloroquine. What laboratory tests and monitoring are required prior to starting this medication?
a. complete blood cell count with differential and glucose-6-phosphate dehydrogenase
b. complete blood cell count with differential and complete metabolic profile
c. ophthalmology evaluation and glucose-6-phosphate dehydrogenase
d. b and c
2. Two months ago you saw a 30-year-old woman with a history of severe atopic dermatitis. She had been using topical steroids with not much improvement. You decided to start a systemic medication. Within 1 month of drug initiation, she called your office to tell you that she is much better but has noticed unwanted hair on her face lately. Which medication is most likely implicated?
a. cyclosporine
b. dapsone
c. hydroxychloroquine
d. methotrexate
3. A 70-year-old man with type 2 diabetes mellitus who drinks 10 cans of beer per week presents to the emergency department with a 3-day history of diffuse tense bullae and pruritus on the legs and trunk. Direct immunofluorescence displayed linear deposition of IgG and C3 at the dermoepidermal junction, confirming your clinical diagnosis. What is the best long-term treatment option for this patient?
a. combination of oral steroids plus methotrexate
b. oral steroids and mycophenolate mofetil
c. oral steroids only
d. topical steroids only
4. A 45-year-old Venezuelan man presents with painful nodules on his bilateral lower legs. A biopsy demonstrates acid-fast bacilli, and a multidrug regimen is initiated for erythema nodosum leprosum. Which of the following is the mechanism of action of the treatment that is US Food and Drug Administration approved for this condition?
a. inhibits chemotaxis
b. inhibits dihydrofolate reductase
c. inhibits tumor necrosis factor α
d. suppresses T-cell function and B-cell antibody production
5. A patient consults her physician because of several side effects from a medication she started 2 weeks ago due to erythematous to violaceous papules on the legs from palpable purpura. She reports diarrhea, abdominal pain, and fatigue. Which medication is she taking?
a. azathioprine
b. colchicine
c. dapsone
d. methotrexate
1. A 40-year-old woman is diagnosed with systemic lupus erythematosus. You discuss treatment options and decide to start hydroxychloroquine. What laboratory tests and monitoring are required prior to starting this medication?
a. complete blood cell count with differential and glucose-6-phosphate dehydrogenase
b. complete blood cell count with differential and complete metabolic profile
c. ophthalmology evaluation and glucose-6-phosphate dehydrogenase
d. b and c
2. Two months ago you saw a 30-year-old woman with a history of severe atopic dermatitis. She had been using topical steroids with not much improvement. You decided to start a systemic medication. Within 1 month of drug initiation, she called your office to tell you that she is much better but has noticed unwanted hair on her face lately. Which medication is most likely implicated?
a. cyclosporine
b. dapsone
c. hydroxychloroquine
d. methotrexate
3. A 70-year-old man with type 2 diabetes mellitus who drinks 10 cans of beer per week presents to the emergency department with a 3-day history of diffuse tense bullae and pruritus on the legs and trunk. Direct immunofluorescence displayed linear deposition of IgG and C3 at the dermoepidermal junction, confirming your clinical diagnosis. What is the best long-term treatment option for this patient?
a. combination of oral steroids plus methotrexate
b. oral steroids and mycophenolate mofetil
c. oral steroids only
d. topical steroids only
4. A 45-year-old Venezuelan man presents with painful nodules on his bilateral lower legs. A biopsy demonstrates acid-fast bacilli, and a multidrug regimen is initiated for erythema nodosum leprosum. Which of the following is the mechanism of action of the treatment that is US Food and Drug Administration approved for this condition?
a. inhibits chemotaxis
b. inhibits dihydrofolate reductase
c. inhibits tumor necrosis factor α
d. suppresses T-cell function and B-cell antibody production
5. A patient consults her physician because of several side effects from a medication she started 2 weeks ago due to erythematous to violaceous papules on the legs from palpable purpura. She reports diarrhea, abdominal pain, and fatigue. Which medication is she taking?
a. azathioprine
b. colchicine
c. dapsone
d. methotrexate
Medications in Dermatology, Part 2: Immunosuppressives
Counterphobia and Poor Sun Protection Practices in First-Degree Relatives of Melanoma Patients
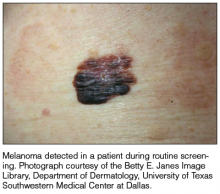
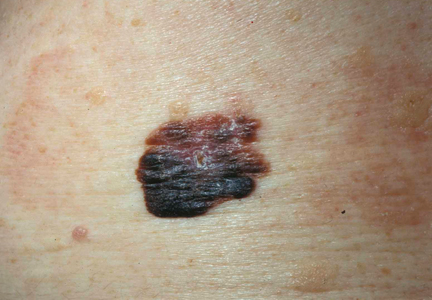
It is widely accepted that there are several factors that may independently elevate an individual’s risk for melanoma, such as a history of childhood sunburns, family history of melanoma, and poor sun protection practices. Several studies have examined risk behaviors in melanoma patients following their diagnosis and have reported findings such as increased UV exposure patterns, persistent tanning bed use, and sun-protective behaviors similar to those of the general population (Figure).1-4
Although first-degree relatives (FDRs) of melanoma patients are at an increased risk for melanoma, they also have been found to exhibit surprisingly poor sun protection practices. In one retrospective analysis, Geller et al5 found that frequent sunburns, high rates of tanning bed use, and low rates of sunscreen use were common among children of health care workers who reported a personal or family history of skin cancer. An independent study reported that merely 37% (37/100) of FDRs of melanoma patients use sunscreen more than half of the time, and considerably fewer wear protective clothing or seek shade while outdoors.6 Given their increased risk for developing melanoma, it is likely to be assumed that FDRs of melanoma patients practice diligent sun protection. The underlying reasons for the failure of this at-risk population to adhere strongly to sun protection practices warrants special attention.
Manne et al7 conducted a survey in a group of FDRs of melanoma patients with self-reported poor sun protection practices to evaluate the demographic, medical, psychological, educational (knowledge of sun protection guidelines), and social influences that correlate with sun protection and sunbathing practices. More effective sun protective behaviors were identified in FDRs with higher education, fewer perceived benefits of sunbathing, more prominent photoaging concerns, and greater sunscreen self-efficacy. The authors concluded that sun-protective behavior in FDRs was not associated with prior knowledge about sunscreen or UV exposure, their relative’s melanoma stage, or physician recommendations for sun protection.7
Factors that have been documented as influencing sun-protective behavior in the general population include knowledge of the benefits of sun protection; attitudes toward tanning and sun protection; subjective norms regarding the beauty and perceived health of a tan; and optimistic bias, which is a cognitive mechanism that causes a person to believe that he/she is at lesser risk for experiencing a negative outcome compared to others. Additionally, sun protection behaviors are influenced by the immediacy of getting the reward (the perceived benefits of tanning) versus the delayed punishment (development of skin cancer).6 Although all of these elements may be important for some individuals, we believe that a subset of FDRs of melanoma patients may be susceptible to the phenomenon known as counterphobia.
Counterphobia is a neurotic response to anxiety in which an individual actively pursues situations that heighten his/her fear rather than fleeing from a feared object or behavior.8 Most insight into counterphobia has come from the experiences of children who have parent(s) with a debilitating or fatal diagnosis. Due to their immature coping mechanisms, some children are at risk for maladaptive behavioral responses. The loss of a parent typically produces severe psychological trauma in all children, but in those who develop counterphobia, it manifests as a heightened fear of death and vulnerability to their parent’s illness. This maladaptive response is dependent on self-identification with the parent, especially among daughters of lost mothers and sons of lost fathers, and this fear remains with the child through adulthood. A survey of 154 motherless daughters found that women aged 19 to 35 years have the highest level of obsessive thoughts of mortality and more than 75% believe they will succumb to their mother’s illness (92% in the case of cancer).9 Despite this fear, children may exhibit health-compromising behaviors related to the diagnoses that led to their parents’ deaths; for example, counterphobia has been identified as a pathologic factor behind sexually promiscuous practices in the children of patients with AIDS, and it also may explain high-risk drinking behavior in a child whose parent died from hepatocellular carcinoma due to a history of alcoholic cirrhosis. Similarly, counterphobia can manifest as the deliberate refusal to undergo a mammogram in a woman whose mother died of breast cancer.9 Psychologists have hypothesized that counterphobic pursuits may result from attempts to master the anxiety associated with fear of injury or death as well as from the notion that attempts at risk-factor reduction are futile, as their death is certain.10
The strong influence of counterphobia on perspectives of health and mortality among individuals affected by early loss of a parent is well documented. An assessment of the subjective life expectancy, death anxiety, and health-related behaviors of college students who lost a parent revealed that these individuals estimated their own life spans to be shorter than college students with 2 living parents.11 Moreover, when students were explicitly instructed to predict their life expectancy based on a purely objective mentality rather than one influenced by personal feelings, the exclusion of emotion yielded a longer projected life span. This finding highlights the magnitude of the psychological forces influencing the ethos of individuals affected by premature parental loss. In the same study, individuals who had experienced early loss of a parent believed they would die of the same condition that caused their parent’s death, a finding accompanied by notably poorer diet and smoking behaviors, which might be expected among those with counterphobic defenses.11
Although Manne et al7 did not find an association between melanoma disease severity and sun-protective behavior in FDRs, the study design did not allow for assessment of potential counterphobic responses, which are most likely to develop in younger individuals who strongly identify with the family member whose disease was disabling or fatal. For example, the study included adult relatives (mean age, 46 years) of melanoma patients diagnosed in the preceding 4 years. Furthermore, fewer than 20% (108/545) of the patients had stage III or IV melanoma, and it was not known if melanoma patients communicated the diagnosis to their family members.7
A practice gap exists in FDRs of melanoma patients who are largely assumed to be practicing adequate, if not heightened, sun protection practices. Given that this group demonstrates poor sun protection practices, it is important to identify reasons for such behavior that may extend beyond what is currently known and may include counterphobia. Based on research performed in other medical conditions, the individuals most at risk for counterphobic responses are young children of patients diagnosed with a disabling or fatal condition, but whether in cases of melanoma counterphobia exists as a maladaptive response and whether such a response may occur in all close relatives, not just offspring, is unknown. Currently, the type of measure(s) that may mitigate poor risk factor modification due to counterphobia, including sun protection practices, is unknown. However, physician knowledge of counterphobic responses as a possibility in close relatives of melanoma patients may improve physician efforts to modify behavior in this unique, high-risk population.
The multimodal pathway of melanoma development suggests that individuals with an underlying genetic predisposition for melanoma who also neglect sun-protective measures are an especially high-risk group.12 As such, targeted education and screening of this patient population may be warranted (Table 1). Although it is incumbent on physicians to incorporate concerted screening, counseling, and focused interventions for newly diagnosed melanoma patients, taking similar measures to counsel and educate immediate relatives who may be at high risk for poor sun protection practices also is encouraged (Table 2).
We believe that recognition of counterphobic behavior is critical in the evaluation of FDRs of melanoma patients. Heightened awareness may bolster primary prevention efforts, especially in our patients with genetic diatheses toward melanoma development.
1. Idorn L, Datta P, Heydenreich J, et al. A 3-year follow-up of sun behavior in patients with cutaneous malignant melanoma [published online ahead of print October 2, 2013]. JAMA Dermatol. doi:10.1001/jamadermatol.2013.5098.
2. Idorn LW, Datta P, Heydenreich J, et al. Sun behaviour after cutaneous malignant melanoma: a study based on ultraviolet radiation measurements and sun diary data [published online ahead of print]. Br J Dermatol. 2013;168:367-373.
3. Mayer D, Layman A, Carlson J. Sun-protection behaviors of melanoma survivors. J Am Acad Dermatol. 2012;66:e9-e10.
4. Lee TK, Brazier AS, Shoveller JA, et al. Sun-related behavior after a diagnosis of cutaneous malignant melanoma. Melanoma Res. 2007;17:51-55.
5. Geller AC, Brooks DR, Colditz GA, et al. Sun protection practices among offspring of women with personal or family history of skin cancer. Pediatrics. 2006;117:e688-e694.
6. Azzarello LM, Dessureault S, Jacobsen PB. Sun-protective behavior among individuals with a family history of melanoma. Cancer Epidemiol Boomarkers Prev. 2006;15:142-145.
7. Manne SL, Coups EJ, Jacobsen PB, et al. Sun protection and sunbathing practices among at-risk family members of patients with melanoma. BMC Public Health. 2011;11:122.
8. Fenichel O. The Psychoanalytic Theory of Neurosis. Oxford, United Kingdom: Taylor & Francis; 1999.
9. Edelman H. Motherless Daughters: The Legacy of Loss. 2nd ed. Cambridge, MA: Da Capo Press; 2006.
10. Poznanski E, Arthur B. The counterphobic defense in children. Child Psychiatry Hum Dev. 1971;1:178-191.
11. Denes-Raj V, Ehrlichman H. Effects of premature parental death on subjective life expectancy, death anxiety, and health behavior. Omega: Journal of Death and Dying. 1991;23:309-321.
12. Hayward NK. Genetics of melanoma predisposition. Oncogene. 2003;22:3053-3062.
13. Arthey S, Clarke VA. Suntanning and sun protection: a review of the psychologial literature. Soc Sci Med. 1995;40:265-274.
It is widely accepted that there are several factors that may independently elevate an individual’s risk for melanoma, such as a history of childhood sunburns, family history of melanoma, and poor sun protection practices. Several studies have examined risk behaviors in melanoma patients following their diagnosis and have reported findings such as increased UV exposure patterns, persistent tanning bed use, and sun-protective behaviors similar to those of the general population (Figure).1-4
Although first-degree relatives (FDRs) of melanoma patients are at an increased risk for melanoma, they also have been found to exhibit surprisingly poor sun protection practices. In one retrospective analysis, Geller et al5 found that frequent sunburns, high rates of tanning bed use, and low rates of sunscreen use were common among children of health care workers who reported a personal or family history of skin cancer. An independent study reported that merely 37% (37/100) of FDRs of melanoma patients use sunscreen more than half of the time, and considerably fewer wear protective clothing or seek shade while outdoors.6 Given their increased risk for developing melanoma, it is likely to be assumed that FDRs of melanoma patients practice diligent sun protection. The underlying reasons for the failure of this at-risk population to adhere strongly to sun protection practices warrants special attention.
Manne et al7 conducted a survey in a group of FDRs of melanoma patients with self-reported poor sun protection practices to evaluate the demographic, medical, psychological, educational (knowledge of sun protection guidelines), and social influences that correlate with sun protection and sunbathing practices. More effective sun protective behaviors were identified in FDRs with higher education, fewer perceived benefits of sunbathing, more prominent photoaging concerns, and greater sunscreen self-efficacy. The authors concluded that sun-protective behavior in FDRs was not associated with prior knowledge about sunscreen or UV exposure, their relative’s melanoma stage, or physician recommendations for sun protection.7
Factors that have been documented as influencing sun-protective behavior in the general population include knowledge of the benefits of sun protection; attitudes toward tanning and sun protection; subjective norms regarding the beauty and perceived health of a tan; and optimistic bias, which is a cognitive mechanism that causes a person to believe that he/she is at lesser risk for experiencing a negative outcome compared to others. Additionally, sun protection behaviors are influenced by the immediacy of getting the reward (the perceived benefits of tanning) versus the delayed punishment (development of skin cancer).6 Although all of these elements may be important for some individuals, we believe that a subset of FDRs of melanoma patients may be susceptible to the phenomenon known as counterphobia.
Counterphobia is a neurotic response to anxiety in which an individual actively pursues situations that heighten his/her fear rather than fleeing from a feared object or behavior.8 Most insight into counterphobia has come from the experiences of children who have parent(s) with a debilitating or fatal diagnosis. Due to their immature coping mechanisms, some children are at risk for maladaptive behavioral responses. The loss of a parent typically produces severe psychological trauma in all children, but in those who develop counterphobia, it manifests as a heightened fear of death and vulnerability to their parent’s illness. This maladaptive response is dependent on self-identification with the parent, especially among daughters of lost mothers and sons of lost fathers, and this fear remains with the child through adulthood. A survey of 154 motherless daughters found that women aged 19 to 35 years have the highest level of obsessive thoughts of mortality and more than 75% believe they will succumb to their mother’s illness (92% in the case of cancer).9 Despite this fear, children may exhibit health-compromising behaviors related to the diagnoses that led to their parents’ deaths; for example, counterphobia has been identified as a pathologic factor behind sexually promiscuous practices in the children of patients with AIDS, and it also may explain high-risk drinking behavior in a child whose parent died from hepatocellular carcinoma due to a history of alcoholic cirrhosis. Similarly, counterphobia can manifest as the deliberate refusal to undergo a mammogram in a woman whose mother died of breast cancer.9 Psychologists have hypothesized that counterphobic pursuits may result from attempts to master the anxiety associated with fear of injury or death as well as from the notion that attempts at risk-factor reduction are futile, as their death is certain.10
The strong influence of counterphobia on perspectives of health and mortality among individuals affected by early loss of a parent is well documented. An assessment of the subjective life expectancy, death anxiety, and health-related behaviors of college students who lost a parent revealed that these individuals estimated their own life spans to be shorter than college students with 2 living parents.11 Moreover, when students were explicitly instructed to predict their life expectancy based on a purely objective mentality rather than one influenced by personal feelings, the exclusion of emotion yielded a longer projected life span. This finding highlights the magnitude of the psychological forces influencing the ethos of individuals affected by premature parental loss. In the same study, individuals who had experienced early loss of a parent believed they would die of the same condition that caused their parent’s death, a finding accompanied by notably poorer diet and smoking behaviors, which might be expected among those with counterphobic defenses.11
Although Manne et al7 did not find an association between melanoma disease severity and sun-protective behavior in FDRs, the study design did not allow for assessment of potential counterphobic responses, which are most likely to develop in younger individuals who strongly identify with the family member whose disease was disabling or fatal. For example, the study included adult relatives (mean age, 46 years) of melanoma patients diagnosed in the preceding 4 years. Furthermore, fewer than 20% (108/545) of the patients had stage III or IV melanoma, and it was not known if melanoma patients communicated the diagnosis to their family members.7
A practice gap exists in FDRs of melanoma patients who are largely assumed to be practicing adequate, if not heightened, sun protection practices. Given that this group demonstrates poor sun protection practices, it is important to identify reasons for such behavior that may extend beyond what is currently known and may include counterphobia. Based on research performed in other medical conditions, the individuals most at risk for counterphobic responses are young children of patients diagnosed with a disabling or fatal condition, but whether in cases of melanoma counterphobia exists as a maladaptive response and whether such a response may occur in all close relatives, not just offspring, is unknown. Currently, the type of measure(s) that may mitigate poor risk factor modification due to counterphobia, including sun protection practices, is unknown. However, physician knowledge of counterphobic responses as a possibility in close relatives of melanoma patients may improve physician efforts to modify behavior in this unique, high-risk population.
The multimodal pathway of melanoma development suggests that individuals with an underlying genetic predisposition for melanoma who also neglect sun-protective measures are an especially high-risk group.12 As such, targeted education and screening of this patient population may be warranted (Table 1). Although it is incumbent on physicians to incorporate concerted screening, counseling, and focused interventions for newly diagnosed melanoma patients, taking similar measures to counsel and educate immediate relatives who may be at high risk for poor sun protection practices also is encouraged (Table 2).
We believe that recognition of counterphobic behavior is critical in the evaluation of FDRs of melanoma patients. Heightened awareness may bolster primary prevention efforts, especially in our patients with genetic diatheses toward melanoma development.
It is widely accepted that there are several factors that may independently elevate an individual’s risk for melanoma, such as a history of childhood sunburns, family history of melanoma, and poor sun protection practices. Several studies have examined risk behaviors in melanoma patients following their diagnosis and have reported findings such as increased UV exposure patterns, persistent tanning bed use, and sun-protective behaviors similar to those of the general population (Figure).1-4
Although first-degree relatives (FDRs) of melanoma patients are at an increased risk for melanoma, they also have been found to exhibit surprisingly poor sun protection practices. In one retrospective analysis, Geller et al5 found that frequent sunburns, high rates of tanning bed use, and low rates of sunscreen use were common among children of health care workers who reported a personal or family history of skin cancer. An independent study reported that merely 37% (37/100) of FDRs of melanoma patients use sunscreen more than half of the time, and considerably fewer wear protective clothing or seek shade while outdoors.6 Given their increased risk for developing melanoma, it is likely to be assumed that FDRs of melanoma patients practice diligent sun protection. The underlying reasons for the failure of this at-risk population to adhere strongly to sun protection practices warrants special attention.
Manne et al7 conducted a survey in a group of FDRs of melanoma patients with self-reported poor sun protection practices to evaluate the demographic, medical, psychological, educational (knowledge of sun protection guidelines), and social influences that correlate with sun protection and sunbathing practices. More effective sun protective behaviors were identified in FDRs with higher education, fewer perceived benefits of sunbathing, more prominent photoaging concerns, and greater sunscreen self-efficacy. The authors concluded that sun-protective behavior in FDRs was not associated with prior knowledge about sunscreen or UV exposure, their relative’s melanoma stage, or physician recommendations for sun protection.7
Factors that have been documented as influencing sun-protective behavior in the general population include knowledge of the benefits of sun protection; attitudes toward tanning and sun protection; subjective norms regarding the beauty and perceived health of a tan; and optimistic bias, which is a cognitive mechanism that causes a person to believe that he/she is at lesser risk for experiencing a negative outcome compared to others. Additionally, sun protection behaviors are influenced by the immediacy of getting the reward (the perceived benefits of tanning) versus the delayed punishment (development of skin cancer).6 Although all of these elements may be important for some individuals, we believe that a subset of FDRs of melanoma patients may be susceptible to the phenomenon known as counterphobia.
Counterphobia is a neurotic response to anxiety in which an individual actively pursues situations that heighten his/her fear rather than fleeing from a feared object or behavior.8 Most insight into counterphobia has come from the experiences of children who have parent(s) with a debilitating or fatal diagnosis. Due to their immature coping mechanisms, some children are at risk for maladaptive behavioral responses. The loss of a parent typically produces severe psychological trauma in all children, but in those who develop counterphobia, it manifests as a heightened fear of death and vulnerability to their parent’s illness. This maladaptive response is dependent on self-identification with the parent, especially among daughters of lost mothers and sons of lost fathers, and this fear remains with the child through adulthood. A survey of 154 motherless daughters found that women aged 19 to 35 years have the highest level of obsessive thoughts of mortality and more than 75% believe they will succumb to their mother’s illness (92% in the case of cancer).9 Despite this fear, children may exhibit health-compromising behaviors related to the diagnoses that led to their parents’ deaths; for example, counterphobia has been identified as a pathologic factor behind sexually promiscuous practices in the children of patients with AIDS, and it also may explain high-risk drinking behavior in a child whose parent died from hepatocellular carcinoma due to a history of alcoholic cirrhosis. Similarly, counterphobia can manifest as the deliberate refusal to undergo a mammogram in a woman whose mother died of breast cancer.9 Psychologists have hypothesized that counterphobic pursuits may result from attempts to master the anxiety associated with fear of injury or death as well as from the notion that attempts at risk-factor reduction are futile, as their death is certain.10
The strong influence of counterphobia on perspectives of health and mortality among individuals affected by early loss of a parent is well documented. An assessment of the subjective life expectancy, death anxiety, and health-related behaviors of college students who lost a parent revealed that these individuals estimated their own life spans to be shorter than college students with 2 living parents.11 Moreover, when students were explicitly instructed to predict their life expectancy based on a purely objective mentality rather than one influenced by personal feelings, the exclusion of emotion yielded a longer projected life span. This finding highlights the magnitude of the psychological forces influencing the ethos of individuals affected by premature parental loss. In the same study, individuals who had experienced early loss of a parent believed they would die of the same condition that caused their parent’s death, a finding accompanied by notably poorer diet and smoking behaviors, which might be expected among those with counterphobic defenses.11
Although Manne et al7 did not find an association between melanoma disease severity and sun-protective behavior in FDRs, the study design did not allow for assessment of potential counterphobic responses, which are most likely to develop in younger individuals who strongly identify with the family member whose disease was disabling or fatal. For example, the study included adult relatives (mean age, 46 years) of melanoma patients diagnosed in the preceding 4 years. Furthermore, fewer than 20% (108/545) of the patients had stage III or IV melanoma, and it was not known if melanoma patients communicated the diagnosis to their family members.7
A practice gap exists in FDRs of melanoma patients who are largely assumed to be practicing adequate, if not heightened, sun protection practices. Given that this group demonstrates poor sun protection practices, it is important to identify reasons for such behavior that may extend beyond what is currently known and may include counterphobia. Based on research performed in other medical conditions, the individuals most at risk for counterphobic responses are young children of patients diagnosed with a disabling or fatal condition, but whether in cases of melanoma counterphobia exists as a maladaptive response and whether such a response may occur in all close relatives, not just offspring, is unknown. Currently, the type of measure(s) that may mitigate poor risk factor modification due to counterphobia, including sun protection practices, is unknown. However, physician knowledge of counterphobic responses as a possibility in close relatives of melanoma patients may improve physician efforts to modify behavior in this unique, high-risk population.
The multimodal pathway of melanoma development suggests that individuals with an underlying genetic predisposition for melanoma who also neglect sun-protective measures are an especially high-risk group.12 As such, targeted education and screening of this patient population may be warranted (Table 1). Although it is incumbent on physicians to incorporate concerted screening, counseling, and focused interventions for newly diagnosed melanoma patients, taking similar measures to counsel and educate immediate relatives who may be at high risk for poor sun protection practices also is encouraged (Table 2).
We believe that recognition of counterphobic behavior is critical in the evaluation of FDRs of melanoma patients. Heightened awareness may bolster primary prevention efforts, especially in our patients with genetic diatheses toward melanoma development.
1. Idorn L, Datta P, Heydenreich J, et al. A 3-year follow-up of sun behavior in patients with cutaneous malignant melanoma [published online ahead of print October 2, 2013]. JAMA Dermatol. doi:10.1001/jamadermatol.2013.5098.
2. Idorn LW, Datta P, Heydenreich J, et al. Sun behaviour after cutaneous malignant melanoma: a study based on ultraviolet radiation measurements and sun diary data [published online ahead of print]. Br J Dermatol. 2013;168:367-373.
3. Mayer D, Layman A, Carlson J. Sun-protection behaviors of melanoma survivors. J Am Acad Dermatol. 2012;66:e9-e10.
4. Lee TK, Brazier AS, Shoveller JA, et al. Sun-related behavior after a diagnosis of cutaneous malignant melanoma. Melanoma Res. 2007;17:51-55.
5. Geller AC, Brooks DR, Colditz GA, et al. Sun protection practices among offspring of women with personal or family history of skin cancer. Pediatrics. 2006;117:e688-e694.
6. Azzarello LM, Dessureault S, Jacobsen PB. Sun-protective behavior among individuals with a family history of melanoma. Cancer Epidemiol Boomarkers Prev. 2006;15:142-145.
7. Manne SL, Coups EJ, Jacobsen PB, et al. Sun protection and sunbathing practices among at-risk family members of patients with melanoma. BMC Public Health. 2011;11:122.
8. Fenichel O. The Psychoanalytic Theory of Neurosis. Oxford, United Kingdom: Taylor & Francis; 1999.
9. Edelman H. Motherless Daughters: The Legacy of Loss. 2nd ed. Cambridge, MA: Da Capo Press; 2006.
10. Poznanski E, Arthur B. The counterphobic defense in children. Child Psychiatry Hum Dev. 1971;1:178-191.
11. Denes-Raj V, Ehrlichman H. Effects of premature parental death on subjective life expectancy, death anxiety, and health behavior. Omega: Journal of Death and Dying. 1991;23:309-321.
12. Hayward NK. Genetics of melanoma predisposition. Oncogene. 2003;22:3053-3062.
13. Arthey S, Clarke VA. Suntanning and sun protection: a review of the psychologial literature. Soc Sci Med. 1995;40:265-274.
1. Idorn L, Datta P, Heydenreich J, et al. A 3-year follow-up of sun behavior in patients with cutaneous malignant melanoma [published online ahead of print October 2, 2013]. JAMA Dermatol. doi:10.1001/jamadermatol.2013.5098.
2. Idorn LW, Datta P, Heydenreich J, et al. Sun behaviour after cutaneous malignant melanoma: a study based on ultraviolet radiation measurements and sun diary data [published online ahead of print]. Br J Dermatol. 2013;168:367-373.
3. Mayer D, Layman A, Carlson J. Sun-protection behaviors of melanoma survivors. J Am Acad Dermatol. 2012;66:e9-e10.
4. Lee TK, Brazier AS, Shoveller JA, et al. Sun-related behavior after a diagnosis of cutaneous malignant melanoma. Melanoma Res. 2007;17:51-55.
5. Geller AC, Brooks DR, Colditz GA, et al. Sun protection practices among offspring of women with personal or family history of skin cancer. Pediatrics. 2006;117:e688-e694.
6. Azzarello LM, Dessureault S, Jacobsen PB. Sun-protective behavior among individuals with a family history of melanoma. Cancer Epidemiol Boomarkers Prev. 2006;15:142-145.
7. Manne SL, Coups EJ, Jacobsen PB, et al. Sun protection and sunbathing practices among at-risk family members of patients with melanoma. BMC Public Health. 2011;11:122.
8. Fenichel O. The Psychoanalytic Theory of Neurosis. Oxford, United Kingdom: Taylor & Francis; 1999.
9. Edelman H. Motherless Daughters: The Legacy of Loss. 2nd ed. Cambridge, MA: Da Capo Press; 2006.
10. Poznanski E, Arthur B. The counterphobic defense in children. Child Psychiatry Hum Dev. 1971;1:178-191.
11. Denes-Raj V, Ehrlichman H. Effects of premature parental death on subjective life expectancy, death anxiety, and health behavior. Omega: Journal of Death and Dying. 1991;23:309-321.
12. Hayward NK. Genetics of melanoma predisposition. Oncogene. 2003;22:3053-3062.
13. Arthey S, Clarke VA. Suntanning and sun protection: a review of the psychologial literature. Soc Sci Med. 1995;40:265-274.