User login
The role of aspirin in primary prevention of cardiovascular disease
Background: Previous studies have shown that aspirin reduces the relative risk of cardiovascular disease (CVD) but also increases the relative risk of bleeding. It is unclear if there are patients without known CVD in whom the absolute risk reduction of CVD outweighs the absolute risk of bleeding. Prognostic CVD and bleeding risk models allow for an assessment of absolute risks and primary preventive interventions.
Study design: Individualized risk-benefit analysis based on sex-specific risk scores and estimates from PREDICT cohort data.
Setting: Primary care practices in New Zealand.
Synopsis: Using the New Zealand–based PREDICT online tool, 245,048 patients had their CVD risk assessed and did not meet exclusion criteria. The online tool predicts CVD events avoided and bleeding events caused by aspirin. When one CVD event was equivalent to one major bleeding event, 2.5% of women and 12.1% of men were classified as benefiting from aspirin (more CVD events avoided than bleeding events caused). When one CVD event was equivalent to two major bleeding events, 21.4% of women and 40.7% of men were classified as benefiting from aspirin. The net-benefit subgroups were older, and had higher baseline 5-year CVD risk, fewer risk factors for bleeding, higher systolic blood pressure, and a higher total cholesterol to HDL cholesterol ratio. Ethnicity and socioeconomic index also influenced benefit or harm.
With use of the upper and lower limits of 95% confidence intervals for models, there were considerable ranges of benefit versus harm. Sex-specific risk scores and meta-analysis have intrinsic uncertainties and results potentially not generalizable outside New Zealand population. Ultimate decision to use aspirin requires shared decision making.
Bottom line: Some patients are likely to derive a net benefit from aspirin for primary prevention of CVD. Risk-benefit models with online tools can help providers and patients estimate these factors to inform shared decision making.
Citation: Selak V et al. Personalized prediction of cardiovascular benefits and bleeding harms for aspirin for primary prevention, a benefit-harm analysis. Ann Intern Med. 2019;71(8):529-39.
Dr. Rupp is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: Previous studies have shown that aspirin reduces the relative risk of cardiovascular disease (CVD) but also increases the relative risk of bleeding. It is unclear if there are patients without known CVD in whom the absolute risk reduction of CVD outweighs the absolute risk of bleeding. Prognostic CVD and bleeding risk models allow for an assessment of absolute risks and primary preventive interventions.
Study design: Individualized risk-benefit analysis based on sex-specific risk scores and estimates from PREDICT cohort data.
Setting: Primary care practices in New Zealand.
Synopsis: Using the New Zealand–based PREDICT online tool, 245,048 patients had their CVD risk assessed and did not meet exclusion criteria. The online tool predicts CVD events avoided and bleeding events caused by aspirin. When one CVD event was equivalent to one major bleeding event, 2.5% of women and 12.1% of men were classified as benefiting from aspirin (more CVD events avoided than bleeding events caused). When one CVD event was equivalent to two major bleeding events, 21.4% of women and 40.7% of men were classified as benefiting from aspirin. The net-benefit subgroups were older, and had higher baseline 5-year CVD risk, fewer risk factors for bleeding, higher systolic blood pressure, and a higher total cholesterol to HDL cholesterol ratio. Ethnicity and socioeconomic index also influenced benefit or harm.
With use of the upper and lower limits of 95% confidence intervals for models, there were considerable ranges of benefit versus harm. Sex-specific risk scores and meta-analysis have intrinsic uncertainties and results potentially not generalizable outside New Zealand population. Ultimate decision to use aspirin requires shared decision making.
Bottom line: Some patients are likely to derive a net benefit from aspirin for primary prevention of CVD. Risk-benefit models with online tools can help providers and patients estimate these factors to inform shared decision making.
Citation: Selak V et al. Personalized prediction of cardiovascular benefits and bleeding harms for aspirin for primary prevention, a benefit-harm analysis. Ann Intern Med. 2019;71(8):529-39.
Dr. Rupp is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: Previous studies have shown that aspirin reduces the relative risk of cardiovascular disease (CVD) but also increases the relative risk of bleeding. It is unclear if there are patients without known CVD in whom the absolute risk reduction of CVD outweighs the absolute risk of bleeding. Prognostic CVD and bleeding risk models allow for an assessment of absolute risks and primary preventive interventions.
Study design: Individualized risk-benefit analysis based on sex-specific risk scores and estimates from PREDICT cohort data.
Setting: Primary care practices in New Zealand.
Synopsis: Using the New Zealand–based PREDICT online tool, 245,048 patients had their CVD risk assessed and did not meet exclusion criteria. The online tool predicts CVD events avoided and bleeding events caused by aspirin. When one CVD event was equivalent to one major bleeding event, 2.5% of women and 12.1% of men were classified as benefiting from aspirin (more CVD events avoided than bleeding events caused). When one CVD event was equivalent to two major bleeding events, 21.4% of women and 40.7% of men were classified as benefiting from aspirin. The net-benefit subgroups were older, and had higher baseline 5-year CVD risk, fewer risk factors for bleeding, higher systolic blood pressure, and a higher total cholesterol to HDL cholesterol ratio. Ethnicity and socioeconomic index also influenced benefit or harm.
With use of the upper and lower limits of 95% confidence intervals for models, there were considerable ranges of benefit versus harm. Sex-specific risk scores and meta-analysis have intrinsic uncertainties and results potentially not generalizable outside New Zealand population. Ultimate decision to use aspirin requires shared decision making.
Bottom line: Some patients are likely to derive a net benefit from aspirin for primary prevention of CVD. Risk-benefit models with online tools can help providers and patients estimate these factors to inform shared decision making.
Citation: Selak V et al. Personalized prediction of cardiovascular benefits and bleeding harms for aspirin for primary prevention, a benefit-harm analysis. Ann Intern Med. 2019;71(8):529-39.
Dr. Rupp is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Detroit cardiologists prevail in retaliation suit against Tenet
After losing at arbitration, as well as in federal court and partially on appeal, Tenet Healthcare is refusing to comment on whether it will continue to battle two Detroit-area cardiologists whom the hospital corporation fired from leadership positions in 2018.
The cardiologists were awarded $10.6 million from an arbitrator, who found that Detroit Medical Center (DMC) and its parent, Tenet, retaliated against Amir Kaki, MD, and Mahir Elder, MD, when the doctors repeatedly reported concerns about patient safety and potential fraud.
The award was made public when it was upheld in federal court in February 2021 and was partially upheld on appeal days later by the Sixth Circuit Court of Appeals.
The Sixth Circuit Court of Appeals denied Tenet’s motion to bar Dr. Kaki and Dr. Elder from returning to work with full privileges but said it would continue to consider the overall appeal. Tenet argued that it needed to keep the cardiologists out of DMC because of “behavioral issues.”
Those allegations are “complete nonsense,” said the cardiologists’ attorney, Deborah Gordon, of Bloomfield Hills, Mich. The alleged problems regarding Dr. Kaki and Dr. Elder were examined by an arbitrator, who “found that all of those complaints were unsubstantiated,” Ms. Gordon said in an interview.
In her final ruling, arbitrator Mary Beth Kelly wrote, “Both Kaki and Elder testified credibly regarding the humiliation, the emotional distress and the reputational damage they have suffered to their national reputations.”
A spokesperson for Tenet and DMC said the organizations had no further comment.
Ms. Gordon said she believes it’s unlikely Tenet will prevail in the Sixth Circuit Court of Appeals, noting that the court already had examined the merits of the case to determine whether Dr. Kaki and Dr. Elder could go back to work. In the court’s opinion, shared in an interview, nothing substantive in Tenet’s appeal prevented the doctors from returning to the hospital, she said.
As of now, both cardiologists have 1 year of privileges at the DMC-affiliated hospitals. Only Dr. Kaki has returned to work, said Ms. Gordon. Neither is speaking to the media, she said.
From respected to reviled
Both Dr. Kaki and Dr. Elder were respected at DMC, according to court filings.
Dr. Kaki was recruited from Weill Cornell Medical College by a Detroit mayor because of his expertise in interventional cardiology. He had staff privileges at DMC beginning in 2012 and was a clinical associate professor and assistant program director of the interventional cardiology fellowship program at Wayne State University in Detroit. He became director of the cardiac catheterization services unit at the new DMC Heart Hospital at Harper-Hutzel Hospital in Detroit in 2014, and 4 years later was appointed director of the facility’s anticoagulation clinic. Dr. Kaki was nominated for and completed Tenet’s Leadership Academy.
Dr. Elder was a clinical professor and assistant fellowship director at Wayne State and was a clinical professor of medicine at Michigan State University. Beginning in 2008, he held directorships at DMC’s cardiac care unit, ambulatory services program, cardiac CT angiogram program, PERT program, and carotid stenting program. Dr. Elder was voted Teacher of the Year for 10 consecutive years by DMC cardiology fellows.
The two doctors aimed high when it came to quality of care and ethics, according to legal filings. Over the years, Dr. Kaki and Dr. Elder repeatedly reported what they considered to be egregious violations of patient safety and of Medicare and Medicaid fraud laws. The clinicians complained about unsterile surgical instruments and the removal of a stat laboratory from the cardiac catheterization unit, noting that the removal would cause delays that would endanger lives.
At peer review meetings, as well as with administrators, they flagged colleagues who they said were performing unnecessary or dangerous procedures solely to generate revenue. At least one doctor falsified records of such a procedure after a patient died, alleged Dr. Kaki and Dr. Elder.
Tenet hired outside attorneys in the fall of 2018, telling Dr. Kaki and Dr. Elder that the legal team would investigate their complaints. However, the investigation was a sham: Filings allege that the investigation was used instead to build a case against Dr. Kaki and Dr. Elder and that Tenet leadership used the inquiry to pressure the cardiologists to resign.
They refused, and in October 2018, they were fired from their leadership positions. DMC and Tenet then held a press conference in which they said that Dr. Kaki and Dr. Elder had been dismissed for “violations” of the “Tenet Standards of Conduct.”
Cardiologists push back
Dr. Kaki and Dr. Elder, however, were not willing to just walk away. They sought reinstatement through an internal DMC appeals panel of their peers. The clinicians who participated on that panel ruled that neither firing was justified.
But DMC’s governing board voted in April 2020 to deny privileges to both cardiologists.
Tenet continued a campaign of retaliation, according to legal filings, by not paying the clinicians for being on call, by removing them from peer review committees, and by prohibiting them from teaching or giving lectures. DMC refused to give Dr. Kaki his personnel record, stating that he was never an employee when he was in the leadership position. Dr. Kaki sued, and a Wayne County Circuit Court judge granted his motion to get his file. DMC and Tenet appealed that ruling but lost.
Eventually, Ms. Gordon sued DMC and Tenet in federal court, alleging the hospital retaliated against the cardiologists, interfered with their ability to earn a living by disparaging them, refused to renew their privileges in 2019, and committed violations under multiple federal and state statutes, including the False Claims Act and the Fair Labor Standards Act.
Tenet successfully argued that the case should go to arbitration.
Arbitrator Mary Beth Kelly, though, ruled in December 2020 that the vast majority of the complaints compiled against the two physicians in the external investigation were not verified or supported and that Tenet and DMC had retaliated against Dr. Kaki and Dr. Elder.
For that harm, Ms. Kelly awarded each clinician $1 million, according to the final ruling shared in an interview.
In addition, she awarded Dr. Kaki $2.3 million in back pay and 2 years of front pay (slightly more than $1 million). She awarded Dr. Elder $2.3 million in back pay and $2.1 million in front pay for 4 years, noting that “his strong association with DMC may make it more difficult for him to successfully transition into the situation he enjoyed prior to termination and nonrenewal.”
The clinicians also were awarded legal fees of $623,816 and court costs of $110,673.
“Wholesale retrial”
To secure the award, Ms. Gordon had to seek a ruling from the U.S. District Court for Eastern Michigan. Tenet asked that court to overturn the arbitrator’s award and to keep it sealed from public view.
In his February ruling, Judge Arthur J. Tarnow wrote that Tenet and DMC “not only attempt to relitigate the legal issues, but also endeavor to introduce a factual counternarrative unmoored from the findings of the Arbitrator and including evidence which the Arbitrator specifically found inadmissible.
“By seeking a wholesale retrial of their case after forcing plaintiffs to arbitrate in the first place,” Tenet and DMC basically ignored the goal of arbitration, which is to relieve judicial congestion and provide a faster and cheaper alternative to litigation, he wrote.
Judge Tarnow also warned Tenet and DMC against taking too long to reinstate privileges for Dr. Kaki and Dr. Elder. If they “continue to delay the restoration of plaintiffs’ privileges in the hopes of a different result on appeal, they will be in violation of this Order,” said the judge.
Tenet, however, tried one more avenue to block the cardiologists from getting privileges, appealing to the Sixth Circuit, which again ordered the company to grant the 1-year privileges.
A version of this article first appeared on Medscape.com.
After losing at arbitration, as well as in federal court and partially on appeal, Tenet Healthcare is refusing to comment on whether it will continue to battle two Detroit-area cardiologists whom the hospital corporation fired from leadership positions in 2018.
The cardiologists were awarded $10.6 million from an arbitrator, who found that Detroit Medical Center (DMC) and its parent, Tenet, retaliated against Amir Kaki, MD, and Mahir Elder, MD, when the doctors repeatedly reported concerns about patient safety and potential fraud.
The award was made public when it was upheld in federal court in February 2021 and was partially upheld on appeal days later by the Sixth Circuit Court of Appeals.
The Sixth Circuit Court of Appeals denied Tenet’s motion to bar Dr. Kaki and Dr. Elder from returning to work with full privileges but said it would continue to consider the overall appeal. Tenet argued that it needed to keep the cardiologists out of DMC because of “behavioral issues.”
Those allegations are “complete nonsense,” said the cardiologists’ attorney, Deborah Gordon, of Bloomfield Hills, Mich. The alleged problems regarding Dr. Kaki and Dr. Elder were examined by an arbitrator, who “found that all of those complaints were unsubstantiated,” Ms. Gordon said in an interview.
In her final ruling, arbitrator Mary Beth Kelly wrote, “Both Kaki and Elder testified credibly regarding the humiliation, the emotional distress and the reputational damage they have suffered to their national reputations.”
A spokesperson for Tenet and DMC said the organizations had no further comment.
Ms. Gordon said she believes it’s unlikely Tenet will prevail in the Sixth Circuit Court of Appeals, noting that the court already had examined the merits of the case to determine whether Dr. Kaki and Dr. Elder could go back to work. In the court’s opinion, shared in an interview, nothing substantive in Tenet’s appeal prevented the doctors from returning to the hospital, she said.
As of now, both cardiologists have 1 year of privileges at the DMC-affiliated hospitals. Only Dr. Kaki has returned to work, said Ms. Gordon. Neither is speaking to the media, she said.
From respected to reviled
Both Dr. Kaki and Dr. Elder were respected at DMC, according to court filings.
Dr. Kaki was recruited from Weill Cornell Medical College by a Detroit mayor because of his expertise in interventional cardiology. He had staff privileges at DMC beginning in 2012 and was a clinical associate professor and assistant program director of the interventional cardiology fellowship program at Wayne State University in Detroit. He became director of the cardiac catheterization services unit at the new DMC Heart Hospital at Harper-Hutzel Hospital in Detroit in 2014, and 4 years later was appointed director of the facility’s anticoagulation clinic. Dr. Kaki was nominated for and completed Tenet’s Leadership Academy.
Dr. Elder was a clinical professor and assistant fellowship director at Wayne State and was a clinical professor of medicine at Michigan State University. Beginning in 2008, he held directorships at DMC’s cardiac care unit, ambulatory services program, cardiac CT angiogram program, PERT program, and carotid stenting program. Dr. Elder was voted Teacher of the Year for 10 consecutive years by DMC cardiology fellows.
The two doctors aimed high when it came to quality of care and ethics, according to legal filings. Over the years, Dr. Kaki and Dr. Elder repeatedly reported what they considered to be egregious violations of patient safety and of Medicare and Medicaid fraud laws. The clinicians complained about unsterile surgical instruments and the removal of a stat laboratory from the cardiac catheterization unit, noting that the removal would cause delays that would endanger lives.
At peer review meetings, as well as with administrators, they flagged colleagues who they said were performing unnecessary or dangerous procedures solely to generate revenue. At least one doctor falsified records of such a procedure after a patient died, alleged Dr. Kaki and Dr. Elder.
Tenet hired outside attorneys in the fall of 2018, telling Dr. Kaki and Dr. Elder that the legal team would investigate their complaints. However, the investigation was a sham: Filings allege that the investigation was used instead to build a case against Dr. Kaki and Dr. Elder and that Tenet leadership used the inquiry to pressure the cardiologists to resign.
They refused, and in October 2018, they were fired from their leadership positions. DMC and Tenet then held a press conference in which they said that Dr. Kaki and Dr. Elder had been dismissed for “violations” of the “Tenet Standards of Conduct.”
Cardiologists push back
Dr. Kaki and Dr. Elder, however, were not willing to just walk away. They sought reinstatement through an internal DMC appeals panel of their peers. The clinicians who participated on that panel ruled that neither firing was justified.
But DMC’s governing board voted in April 2020 to deny privileges to both cardiologists.
Tenet continued a campaign of retaliation, according to legal filings, by not paying the clinicians for being on call, by removing them from peer review committees, and by prohibiting them from teaching or giving lectures. DMC refused to give Dr. Kaki his personnel record, stating that he was never an employee when he was in the leadership position. Dr. Kaki sued, and a Wayne County Circuit Court judge granted his motion to get his file. DMC and Tenet appealed that ruling but lost.
Eventually, Ms. Gordon sued DMC and Tenet in federal court, alleging the hospital retaliated against the cardiologists, interfered with their ability to earn a living by disparaging them, refused to renew their privileges in 2019, and committed violations under multiple federal and state statutes, including the False Claims Act and the Fair Labor Standards Act.
Tenet successfully argued that the case should go to arbitration.
Arbitrator Mary Beth Kelly, though, ruled in December 2020 that the vast majority of the complaints compiled against the two physicians in the external investigation were not verified or supported and that Tenet and DMC had retaliated against Dr. Kaki and Dr. Elder.
For that harm, Ms. Kelly awarded each clinician $1 million, according to the final ruling shared in an interview.
In addition, she awarded Dr. Kaki $2.3 million in back pay and 2 years of front pay (slightly more than $1 million). She awarded Dr. Elder $2.3 million in back pay and $2.1 million in front pay for 4 years, noting that “his strong association with DMC may make it more difficult for him to successfully transition into the situation he enjoyed prior to termination and nonrenewal.”
The clinicians also were awarded legal fees of $623,816 and court costs of $110,673.
“Wholesale retrial”
To secure the award, Ms. Gordon had to seek a ruling from the U.S. District Court for Eastern Michigan. Tenet asked that court to overturn the arbitrator’s award and to keep it sealed from public view.
In his February ruling, Judge Arthur J. Tarnow wrote that Tenet and DMC “not only attempt to relitigate the legal issues, but also endeavor to introduce a factual counternarrative unmoored from the findings of the Arbitrator and including evidence which the Arbitrator specifically found inadmissible.
“By seeking a wholesale retrial of their case after forcing plaintiffs to arbitrate in the first place,” Tenet and DMC basically ignored the goal of arbitration, which is to relieve judicial congestion and provide a faster and cheaper alternative to litigation, he wrote.
Judge Tarnow also warned Tenet and DMC against taking too long to reinstate privileges for Dr. Kaki and Dr. Elder. If they “continue to delay the restoration of plaintiffs’ privileges in the hopes of a different result on appeal, they will be in violation of this Order,” said the judge.
Tenet, however, tried one more avenue to block the cardiologists from getting privileges, appealing to the Sixth Circuit, which again ordered the company to grant the 1-year privileges.
A version of this article first appeared on Medscape.com.
After losing at arbitration, as well as in federal court and partially on appeal, Tenet Healthcare is refusing to comment on whether it will continue to battle two Detroit-area cardiologists whom the hospital corporation fired from leadership positions in 2018.
The cardiologists were awarded $10.6 million from an arbitrator, who found that Detroit Medical Center (DMC) and its parent, Tenet, retaliated against Amir Kaki, MD, and Mahir Elder, MD, when the doctors repeatedly reported concerns about patient safety and potential fraud.
The award was made public when it was upheld in federal court in February 2021 and was partially upheld on appeal days later by the Sixth Circuit Court of Appeals.
The Sixth Circuit Court of Appeals denied Tenet’s motion to bar Dr. Kaki and Dr. Elder from returning to work with full privileges but said it would continue to consider the overall appeal. Tenet argued that it needed to keep the cardiologists out of DMC because of “behavioral issues.”
Those allegations are “complete nonsense,” said the cardiologists’ attorney, Deborah Gordon, of Bloomfield Hills, Mich. The alleged problems regarding Dr. Kaki and Dr. Elder were examined by an arbitrator, who “found that all of those complaints were unsubstantiated,” Ms. Gordon said in an interview.
In her final ruling, arbitrator Mary Beth Kelly wrote, “Both Kaki and Elder testified credibly regarding the humiliation, the emotional distress and the reputational damage they have suffered to their national reputations.”
A spokesperson for Tenet and DMC said the organizations had no further comment.
Ms. Gordon said she believes it’s unlikely Tenet will prevail in the Sixth Circuit Court of Appeals, noting that the court already had examined the merits of the case to determine whether Dr. Kaki and Dr. Elder could go back to work. In the court’s opinion, shared in an interview, nothing substantive in Tenet’s appeal prevented the doctors from returning to the hospital, she said.
As of now, both cardiologists have 1 year of privileges at the DMC-affiliated hospitals. Only Dr. Kaki has returned to work, said Ms. Gordon. Neither is speaking to the media, she said.
From respected to reviled
Both Dr. Kaki and Dr. Elder were respected at DMC, according to court filings.
Dr. Kaki was recruited from Weill Cornell Medical College by a Detroit mayor because of his expertise in interventional cardiology. He had staff privileges at DMC beginning in 2012 and was a clinical associate professor and assistant program director of the interventional cardiology fellowship program at Wayne State University in Detroit. He became director of the cardiac catheterization services unit at the new DMC Heart Hospital at Harper-Hutzel Hospital in Detroit in 2014, and 4 years later was appointed director of the facility’s anticoagulation clinic. Dr. Kaki was nominated for and completed Tenet’s Leadership Academy.
Dr. Elder was a clinical professor and assistant fellowship director at Wayne State and was a clinical professor of medicine at Michigan State University. Beginning in 2008, he held directorships at DMC’s cardiac care unit, ambulatory services program, cardiac CT angiogram program, PERT program, and carotid stenting program. Dr. Elder was voted Teacher of the Year for 10 consecutive years by DMC cardiology fellows.
The two doctors aimed high when it came to quality of care and ethics, according to legal filings. Over the years, Dr. Kaki and Dr. Elder repeatedly reported what they considered to be egregious violations of patient safety and of Medicare and Medicaid fraud laws. The clinicians complained about unsterile surgical instruments and the removal of a stat laboratory from the cardiac catheterization unit, noting that the removal would cause delays that would endanger lives.
At peer review meetings, as well as with administrators, they flagged colleagues who they said were performing unnecessary or dangerous procedures solely to generate revenue. At least one doctor falsified records of such a procedure after a patient died, alleged Dr. Kaki and Dr. Elder.
Tenet hired outside attorneys in the fall of 2018, telling Dr. Kaki and Dr. Elder that the legal team would investigate their complaints. However, the investigation was a sham: Filings allege that the investigation was used instead to build a case against Dr. Kaki and Dr. Elder and that Tenet leadership used the inquiry to pressure the cardiologists to resign.
They refused, and in October 2018, they were fired from their leadership positions. DMC and Tenet then held a press conference in which they said that Dr. Kaki and Dr. Elder had been dismissed for “violations” of the “Tenet Standards of Conduct.”
Cardiologists push back
Dr. Kaki and Dr. Elder, however, were not willing to just walk away. They sought reinstatement through an internal DMC appeals panel of their peers. The clinicians who participated on that panel ruled that neither firing was justified.
But DMC’s governing board voted in April 2020 to deny privileges to both cardiologists.
Tenet continued a campaign of retaliation, according to legal filings, by not paying the clinicians for being on call, by removing them from peer review committees, and by prohibiting them from teaching or giving lectures. DMC refused to give Dr. Kaki his personnel record, stating that he was never an employee when he was in the leadership position. Dr. Kaki sued, and a Wayne County Circuit Court judge granted his motion to get his file. DMC and Tenet appealed that ruling but lost.
Eventually, Ms. Gordon sued DMC and Tenet in federal court, alleging the hospital retaliated against the cardiologists, interfered with their ability to earn a living by disparaging them, refused to renew their privileges in 2019, and committed violations under multiple federal and state statutes, including the False Claims Act and the Fair Labor Standards Act.
Tenet successfully argued that the case should go to arbitration.
Arbitrator Mary Beth Kelly, though, ruled in December 2020 that the vast majority of the complaints compiled against the two physicians in the external investigation were not verified or supported and that Tenet and DMC had retaliated against Dr. Kaki and Dr. Elder.
For that harm, Ms. Kelly awarded each clinician $1 million, according to the final ruling shared in an interview.
In addition, she awarded Dr. Kaki $2.3 million in back pay and 2 years of front pay (slightly more than $1 million). She awarded Dr. Elder $2.3 million in back pay and $2.1 million in front pay for 4 years, noting that “his strong association with DMC may make it more difficult for him to successfully transition into the situation he enjoyed prior to termination and nonrenewal.”
The clinicians also were awarded legal fees of $623,816 and court costs of $110,673.
“Wholesale retrial”
To secure the award, Ms. Gordon had to seek a ruling from the U.S. District Court for Eastern Michigan. Tenet asked that court to overturn the arbitrator’s award and to keep it sealed from public view.
In his February ruling, Judge Arthur J. Tarnow wrote that Tenet and DMC “not only attempt to relitigate the legal issues, but also endeavor to introduce a factual counternarrative unmoored from the findings of the Arbitrator and including evidence which the Arbitrator specifically found inadmissible.
“By seeking a wholesale retrial of their case after forcing plaintiffs to arbitrate in the first place,” Tenet and DMC basically ignored the goal of arbitration, which is to relieve judicial congestion and provide a faster and cheaper alternative to litigation, he wrote.
Judge Tarnow also warned Tenet and DMC against taking too long to reinstate privileges for Dr. Kaki and Dr. Elder. If they “continue to delay the restoration of plaintiffs’ privileges in the hopes of a different result on appeal, they will be in violation of this Order,” said the judge.
Tenet, however, tried one more avenue to block the cardiologists from getting privileges, appealing to the Sixth Circuit, which again ordered the company to grant the 1-year privileges.
A version of this article first appeared on Medscape.com.
Meta-analysis supports late thrombectomy in selected stroke patients
of data from six clinical trials.
Results of the AURORA analysis showed that for every 100 patients treated with thrombectomy, 33 patients will have less disability, and 27 patients will achieve an independent level of functioning compared with patients who receive only standard medical care.
The benefit of mechanical removal of the clot for selected patients who may have salvageable brain tissue, as identified through the use of various imaging modalities, was maintained whether the patient had a “wake-up stroke” or the onset of symptoms was witnessed, regardless of the point in time within the late window. In fact, the benefit of intervention was greater for patients who presented in the latter part of the late time window.
Never too late for urgent medical care
“While the findings of this analysis do not contradict the mantra that the earlier treatment is instituted, the higher the chance of a good outcome, they highlight the fact that it is never too late to seek urgent medical care,” said lead investigator Tudor Jovin, MD, chair of neurology at Cooper University Hospital, Cherry Hill, New Jersey.
“The implications of the findings from AURORA are that they could lead to a change in guidelines from endorsement of thrombectomy as level 1a recommendation in eligible patients presenting in the 6- to 16-hour time window to a 6- to 24-hour time window,” said Dr. Jovin.
“Furthermore, there are strong signals of benefit of thrombectomy in patients who are not selected based on volumetric analysis of baseline infarct (core) or extent of tissue at risk (penumbra), such that when those imaging modalities are not available or contraindicated, selection based on noncontrast CT and clinical information only may be acceptable,” he added. “Finally, the possibility of benefit from thrombectomy performed beyond 24 hours from last seen well is real and should be explored in future studies.”
The AURORA findings were presented at the virtual International Stroke Conference (ISC) 2021.
The objective of the study was to provide a more precise estimate of the benefit of thrombectomy for patients with stroke when performed within 6-24 hours after the patient was last seen well, Dr. Jovin explained.
He said the 6-hour cutoff was chosen somewhat arbitrarily, but added, “It is highly consequential, as it marks the point of demarcation between the early and late time window, and virtually all guidelines recommend different approaches, dependent on whether patients present before or after 6 hours from symptom onset.”
The 6+ hour window
Dr. Jovin pointed out that for patients who present beyond 6 hours, treatment options are more restricted, because the data on thrombectomy in this later period come mainly from two North American trials (DEFUSE 3 and DAWN) that had very stringent imaging criteria for enrollment.
“We wanted to create a heterogeneous dataset with regard to geography and selection criteria by forming the AURORA collaboration,” he commented. Their study involved an individual-level pooled analysis of all patients who underwent randomization after 6 hours from the time that they were last seen well. Patients were randomly assigned to receive either best medical therapy alone or best medical therapy plus thrombectomy (either with stent retrievers or aspiration) for anterior circulation proximal large-vessel occlusion stroke.
The data came from six trials: DAWN (which enrolled patients 6-24 hours from stroke onset), DEFUSE 3 (6-16 hours), ESCAPE (0-12 hours), REVASCAT (0-8 hours), RESILIENT (0-8 hours), and POSITIVE (0-12 hours). In total, 505 patients were included in the meta-analysis, 266 in the intervention group, and 239 in the control group.
“By pooling data on patients presenting after 6 hours from all these trails, we achieve greater precision for treatment effect estimation and increased the power for subgroup analysis,” Dr. Jovin noted.
Although the majority of the patients were in the DAWN and DIFFUSE 3 trials (n = 388), “there are still a good number from the other four trials (n = 117),” Dr. Jovin reported.
Most of the trials used Modified Rankin Scale (mRS) ordinal or shift analysis as their primary endpoint, which was the also the endpoint chosen for this meta-analysis.
Imaging selection criteria ranged from fully automated software-generated quantitative volumetric analysis of baseline infarct (core) or tissue at risk to CT perfusion and plain CT/CTA. The minimum ASPECTS score was 5 or 6.
There were no imbalances in baseline characteristics. The median NIH Stroke Scale score was 16, and the median ASPECTS score was 8. The median time to randomization was 10.5 hours.
With regard to safety, there was no significant difference in rates of symptomatic intracerebral hemorrhage (5.3% in the intervention group vs. 3.3% in the control group; P = .23) or in mortality at 90 days (16.5% vs. 19.3%; P = .87). Jovin noted that these results are very similar to those from the HERMES meta-analysis of patients treated in the early time window.
The primary outcome – ordinal analysis of the mRS distribution – showed an adjusted odds ratio of 2.54 (P < .0001) for benefit in the intervention group. The number needed to treat to reduce disability was 3. “This is again very similar to the HERMES meta-analysis of patients in the early window,” Dr. Jovin said.
The P value for heterogeneity of treatment effect across the six studies was nonsignificant.
Secondary outcome analysis showed an “almost 27%” difference in good functional outcome (MRS, 0-2) between the two groups (45.9% in the intervention group vs. 19.3% in the control group), which translates into a number needed to treat of 3.8, Dr. Jovin reported.
Subgroup analysis showed a treatment effect favoring intervention across all prespecified subgroup factors, including age, sex, occlusion location, mode of presentation (wake-up vs. witnessed), and ASPECTS score, with the caveat that most of the patients were enrolled with ASPECTS scores of 7 or greater.
Early versus late
Surprisingly, although thrombectomy was found to be beneficial in both the 6- to 12-hour and 12- to 24-hour time window, the magnitude of benefit was significantly higher in the later rather than the earlier time window. The odds ratio of a better outcome with thrombectomy on the mRS shift analysis in those presenting in the 6- to 12-hour period was 1.78, compared with 5.86 in the 12- to 24-hour time window.
“This should not be interpreted as a higher chance of a good outcome if treated late. In fact, the rate of good outcomes were numerically higher in the earlier treated patients, but the difference comes from the control group, which did much worse in patients randomized in the later time period,” Dr. Jovin said.
“Aurora was the goddess of dawn [in ancient Roman mythology], so this is a very fitting name, as it reminds us that we are in the dawn of a new era where patients are selected based on physiological data rather than on time, and we certainly hope that this work has brought us closer to this reality,” Dr. Jovin concluded.
Commenting on the study, Michael Hill, MD, University of Calgary (Alta.), said: “The work provides pooled empiric data to support the concept that time to treatment is no longer the sole threshold variable to be used in treatment decision-making. Instead, time is now simply another variable to consider in the context of clinical and imaging factors.”
Dr. Hill, who headed up the ESCAPE trial and was also involved in the current meta-analysis, added: “This meta-analysis supports the concept of patient selection using the ‘good scan’ model, rather than using a time-based concept of patient eligibility for endovascular therapy. It will further push changes in care, because the implication is that all patients with more severe acute stroke presentations need emergency neurovascular imaging to decide if they are eligible for treatment.”
The AURORA meta-analysis was funded by Stryker Neurovascular. Dr. Jovin reports stock holdings in Anaconda, Route 92, VizAi, FreeOx, Blockade Medical, Methinks, and Corindus; personal fees from Cerenovus and Contego Medical; travel support from Fundacio Ictus; and grant support from Medtronic and Stryker Neurovascular.
A version of this article first appeared on Medscape.com.
of data from six clinical trials.
Results of the AURORA analysis showed that for every 100 patients treated with thrombectomy, 33 patients will have less disability, and 27 patients will achieve an independent level of functioning compared with patients who receive only standard medical care.
The benefit of mechanical removal of the clot for selected patients who may have salvageable brain tissue, as identified through the use of various imaging modalities, was maintained whether the patient had a “wake-up stroke” or the onset of symptoms was witnessed, regardless of the point in time within the late window. In fact, the benefit of intervention was greater for patients who presented in the latter part of the late time window.
Never too late for urgent medical care
“While the findings of this analysis do not contradict the mantra that the earlier treatment is instituted, the higher the chance of a good outcome, they highlight the fact that it is never too late to seek urgent medical care,” said lead investigator Tudor Jovin, MD, chair of neurology at Cooper University Hospital, Cherry Hill, New Jersey.
“The implications of the findings from AURORA are that they could lead to a change in guidelines from endorsement of thrombectomy as level 1a recommendation in eligible patients presenting in the 6- to 16-hour time window to a 6- to 24-hour time window,” said Dr. Jovin.
“Furthermore, there are strong signals of benefit of thrombectomy in patients who are not selected based on volumetric analysis of baseline infarct (core) or extent of tissue at risk (penumbra), such that when those imaging modalities are not available or contraindicated, selection based on noncontrast CT and clinical information only may be acceptable,” he added. “Finally, the possibility of benefit from thrombectomy performed beyond 24 hours from last seen well is real and should be explored in future studies.”
The AURORA findings were presented at the virtual International Stroke Conference (ISC) 2021.
The objective of the study was to provide a more precise estimate of the benefit of thrombectomy for patients with stroke when performed within 6-24 hours after the patient was last seen well, Dr. Jovin explained.
He said the 6-hour cutoff was chosen somewhat arbitrarily, but added, “It is highly consequential, as it marks the point of demarcation between the early and late time window, and virtually all guidelines recommend different approaches, dependent on whether patients present before or after 6 hours from symptom onset.”
The 6+ hour window
Dr. Jovin pointed out that for patients who present beyond 6 hours, treatment options are more restricted, because the data on thrombectomy in this later period come mainly from two North American trials (DEFUSE 3 and DAWN) that had very stringent imaging criteria for enrollment.
“We wanted to create a heterogeneous dataset with regard to geography and selection criteria by forming the AURORA collaboration,” he commented. Their study involved an individual-level pooled analysis of all patients who underwent randomization after 6 hours from the time that they were last seen well. Patients were randomly assigned to receive either best medical therapy alone or best medical therapy plus thrombectomy (either with stent retrievers or aspiration) for anterior circulation proximal large-vessel occlusion stroke.
The data came from six trials: DAWN (which enrolled patients 6-24 hours from stroke onset), DEFUSE 3 (6-16 hours), ESCAPE (0-12 hours), REVASCAT (0-8 hours), RESILIENT (0-8 hours), and POSITIVE (0-12 hours). In total, 505 patients were included in the meta-analysis, 266 in the intervention group, and 239 in the control group.
“By pooling data on patients presenting after 6 hours from all these trails, we achieve greater precision for treatment effect estimation and increased the power for subgroup analysis,” Dr. Jovin noted.
Although the majority of the patients were in the DAWN and DIFFUSE 3 trials (n = 388), “there are still a good number from the other four trials (n = 117),” Dr. Jovin reported.
Most of the trials used Modified Rankin Scale (mRS) ordinal or shift analysis as their primary endpoint, which was the also the endpoint chosen for this meta-analysis.
Imaging selection criteria ranged from fully automated software-generated quantitative volumetric analysis of baseline infarct (core) or tissue at risk to CT perfusion and plain CT/CTA. The minimum ASPECTS score was 5 or 6.
There were no imbalances in baseline characteristics. The median NIH Stroke Scale score was 16, and the median ASPECTS score was 8. The median time to randomization was 10.5 hours.
With regard to safety, there was no significant difference in rates of symptomatic intracerebral hemorrhage (5.3% in the intervention group vs. 3.3% in the control group; P = .23) or in mortality at 90 days (16.5% vs. 19.3%; P = .87). Jovin noted that these results are very similar to those from the HERMES meta-analysis of patients treated in the early time window.
The primary outcome – ordinal analysis of the mRS distribution – showed an adjusted odds ratio of 2.54 (P < .0001) for benefit in the intervention group. The number needed to treat to reduce disability was 3. “This is again very similar to the HERMES meta-analysis of patients in the early window,” Dr. Jovin said.
The P value for heterogeneity of treatment effect across the six studies was nonsignificant.
Secondary outcome analysis showed an “almost 27%” difference in good functional outcome (MRS, 0-2) between the two groups (45.9% in the intervention group vs. 19.3% in the control group), which translates into a number needed to treat of 3.8, Dr. Jovin reported.
Subgroup analysis showed a treatment effect favoring intervention across all prespecified subgroup factors, including age, sex, occlusion location, mode of presentation (wake-up vs. witnessed), and ASPECTS score, with the caveat that most of the patients were enrolled with ASPECTS scores of 7 or greater.
Early versus late
Surprisingly, although thrombectomy was found to be beneficial in both the 6- to 12-hour and 12- to 24-hour time window, the magnitude of benefit was significantly higher in the later rather than the earlier time window. The odds ratio of a better outcome with thrombectomy on the mRS shift analysis in those presenting in the 6- to 12-hour period was 1.78, compared with 5.86 in the 12- to 24-hour time window.
“This should not be interpreted as a higher chance of a good outcome if treated late. In fact, the rate of good outcomes were numerically higher in the earlier treated patients, but the difference comes from the control group, which did much worse in patients randomized in the later time period,” Dr. Jovin said.
“Aurora was the goddess of dawn [in ancient Roman mythology], so this is a very fitting name, as it reminds us that we are in the dawn of a new era where patients are selected based on physiological data rather than on time, and we certainly hope that this work has brought us closer to this reality,” Dr. Jovin concluded.
Commenting on the study, Michael Hill, MD, University of Calgary (Alta.), said: “The work provides pooled empiric data to support the concept that time to treatment is no longer the sole threshold variable to be used in treatment decision-making. Instead, time is now simply another variable to consider in the context of clinical and imaging factors.”
Dr. Hill, who headed up the ESCAPE trial and was also involved in the current meta-analysis, added: “This meta-analysis supports the concept of patient selection using the ‘good scan’ model, rather than using a time-based concept of patient eligibility for endovascular therapy. It will further push changes in care, because the implication is that all patients with more severe acute stroke presentations need emergency neurovascular imaging to decide if they are eligible for treatment.”
The AURORA meta-analysis was funded by Stryker Neurovascular. Dr. Jovin reports stock holdings in Anaconda, Route 92, VizAi, FreeOx, Blockade Medical, Methinks, and Corindus; personal fees from Cerenovus and Contego Medical; travel support from Fundacio Ictus; and grant support from Medtronic and Stryker Neurovascular.
A version of this article first appeared on Medscape.com.
of data from six clinical trials.
Results of the AURORA analysis showed that for every 100 patients treated with thrombectomy, 33 patients will have less disability, and 27 patients will achieve an independent level of functioning compared with patients who receive only standard medical care.
The benefit of mechanical removal of the clot for selected patients who may have salvageable brain tissue, as identified through the use of various imaging modalities, was maintained whether the patient had a “wake-up stroke” or the onset of symptoms was witnessed, regardless of the point in time within the late window. In fact, the benefit of intervention was greater for patients who presented in the latter part of the late time window.
Never too late for urgent medical care
“While the findings of this analysis do not contradict the mantra that the earlier treatment is instituted, the higher the chance of a good outcome, they highlight the fact that it is never too late to seek urgent medical care,” said lead investigator Tudor Jovin, MD, chair of neurology at Cooper University Hospital, Cherry Hill, New Jersey.
“The implications of the findings from AURORA are that they could lead to a change in guidelines from endorsement of thrombectomy as level 1a recommendation in eligible patients presenting in the 6- to 16-hour time window to a 6- to 24-hour time window,” said Dr. Jovin.
“Furthermore, there are strong signals of benefit of thrombectomy in patients who are not selected based on volumetric analysis of baseline infarct (core) or extent of tissue at risk (penumbra), such that when those imaging modalities are not available or contraindicated, selection based on noncontrast CT and clinical information only may be acceptable,” he added. “Finally, the possibility of benefit from thrombectomy performed beyond 24 hours from last seen well is real and should be explored in future studies.”
The AURORA findings were presented at the virtual International Stroke Conference (ISC) 2021.
The objective of the study was to provide a more precise estimate of the benefit of thrombectomy for patients with stroke when performed within 6-24 hours after the patient was last seen well, Dr. Jovin explained.
He said the 6-hour cutoff was chosen somewhat arbitrarily, but added, “It is highly consequential, as it marks the point of demarcation between the early and late time window, and virtually all guidelines recommend different approaches, dependent on whether patients present before or after 6 hours from symptom onset.”
The 6+ hour window
Dr. Jovin pointed out that for patients who present beyond 6 hours, treatment options are more restricted, because the data on thrombectomy in this later period come mainly from two North American trials (DEFUSE 3 and DAWN) that had very stringent imaging criteria for enrollment.
“We wanted to create a heterogeneous dataset with regard to geography and selection criteria by forming the AURORA collaboration,” he commented. Their study involved an individual-level pooled analysis of all patients who underwent randomization after 6 hours from the time that they were last seen well. Patients were randomly assigned to receive either best medical therapy alone or best medical therapy plus thrombectomy (either with stent retrievers or aspiration) for anterior circulation proximal large-vessel occlusion stroke.
The data came from six trials: DAWN (which enrolled patients 6-24 hours from stroke onset), DEFUSE 3 (6-16 hours), ESCAPE (0-12 hours), REVASCAT (0-8 hours), RESILIENT (0-8 hours), and POSITIVE (0-12 hours). In total, 505 patients were included in the meta-analysis, 266 in the intervention group, and 239 in the control group.
“By pooling data on patients presenting after 6 hours from all these trails, we achieve greater precision for treatment effect estimation and increased the power for subgroup analysis,” Dr. Jovin noted.
Although the majority of the patients were in the DAWN and DIFFUSE 3 trials (n = 388), “there are still a good number from the other four trials (n = 117),” Dr. Jovin reported.
Most of the trials used Modified Rankin Scale (mRS) ordinal or shift analysis as their primary endpoint, which was the also the endpoint chosen for this meta-analysis.
Imaging selection criteria ranged from fully automated software-generated quantitative volumetric analysis of baseline infarct (core) or tissue at risk to CT perfusion and plain CT/CTA. The minimum ASPECTS score was 5 or 6.
There were no imbalances in baseline characteristics. The median NIH Stroke Scale score was 16, and the median ASPECTS score was 8. The median time to randomization was 10.5 hours.
With regard to safety, there was no significant difference in rates of symptomatic intracerebral hemorrhage (5.3% in the intervention group vs. 3.3% in the control group; P = .23) or in mortality at 90 days (16.5% vs. 19.3%; P = .87). Jovin noted that these results are very similar to those from the HERMES meta-analysis of patients treated in the early time window.
The primary outcome – ordinal analysis of the mRS distribution – showed an adjusted odds ratio of 2.54 (P < .0001) for benefit in the intervention group. The number needed to treat to reduce disability was 3. “This is again very similar to the HERMES meta-analysis of patients in the early window,” Dr. Jovin said.
The P value for heterogeneity of treatment effect across the six studies was nonsignificant.
Secondary outcome analysis showed an “almost 27%” difference in good functional outcome (MRS, 0-2) between the two groups (45.9% in the intervention group vs. 19.3% in the control group), which translates into a number needed to treat of 3.8, Dr. Jovin reported.
Subgroup analysis showed a treatment effect favoring intervention across all prespecified subgroup factors, including age, sex, occlusion location, mode of presentation (wake-up vs. witnessed), and ASPECTS score, with the caveat that most of the patients were enrolled with ASPECTS scores of 7 or greater.
Early versus late
Surprisingly, although thrombectomy was found to be beneficial in both the 6- to 12-hour and 12- to 24-hour time window, the magnitude of benefit was significantly higher in the later rather than the earlier time window. The odds ratio of a better outcome with thrombectomy on the mRS shift analysis in those presenting in the 6- to 12-hour period was 1.78, compared with 5.86 in the 12- to 24-hour time window.
“This should not be interpreted as a higher chance of a good outcome if treated late. In fact, the rate of good outcomes were numerically higher in the earlier treated patients, but the difference comes from the control group, which did much worse in patients randomized in the later time period,” Dr. Jovin said.
“Aurora was the goddess of dawn [in ancient Roman mythology], so this is a very fitting name, as it reminds us that we are in the dawn of a new era where patients are selected based on physiological data rather than on time, and we certainly hope that this work has brought us closer to this reality,” Dr. Jovin concluded.
Commenting on the study, Michael Hill, MD, University of Calgary (Alta.), said: “The work provides pooled empiric data to support the concept that time to treatment is no longer the sole threshold variable to be used in treatment decision-making. Instead, time is now simply another variable to consider in the context of clinical and imaging factors.”
Dr. Hill, who headed up the ESCAPE trial and was also involved in the current meta-analysis, added: “This meta-analysis supports the concept of patient selection using the ‘good scan’ model, rather than using a time-based concept of patient eligibility for endovascular therapy. It will further push changes in care, because the implication is that all patients with more severe acute stroke presentations need emergency neurovascular imaging to decide if they are eligible for treatment.”
The AURORA meta-analysis was funded by Stryker Neurovascular. Dr. Jovin reports stock holdings in Anaconda, Route 92, VizAi, FreeOx, Blockade Medical, Methinks, and Corindus; personal fees from Cerenovus and Contego Medical; travel support from Fundacio Ictus; and grant support from Medtronic and Stryker Neurovascular.
A version of this article first appeared on Medscape.com.
FROM ISC 2021
PCI and CABG for left main disease have equal outcomes at 5 years
Background: While PCI with drug-eluting stents has become more accepted as treatment for some patients with left main disease, long-term outcomes from randomized control trials comparing PCI with CABG have yet to be clearly established.
Study design: International, open-label, multicenter, randomized trial.
Setting: A total of 126 sites in 17 countries.
Synopsis: Patients with low or intermediate anatomical complexity with 70% visual stenosis of the left main coronary artery or 50%-70% stenosis by noninvasive testing were randomized to either PCI (948) or CABG (957). Dual-antiplatelet therapy was given to PCI patients and aspirin to CABG patients. At 5 years there was no significant difference in the rate of the composite of death, stroke, or myocardial infarction (22.0% with PCI vs. 19.2% with CABG; difference, 2.8 percentage points; 95% CI, –0.9 to 6.5; P = .13). This was consistent across subgroups.
There were numerical differences in nonpowered secondary outcomes that may represent effects but should be interpreted cautiously: ischemia-driven revascularization (16.9% with PCI vs. 10% with CABG), transient ischemic attack plus stroke (3.3% with PCI vs. 5.2% with CABG), and death from any cause (3% with PCI vs. 9.9% with CABG). There was no significant difference in cardiovascular events, MI, or stroke.
One interesting limitation was that patients who had PCI were more commonly on dual-antiplatelet therapy and angiotensin converting–enzyme inhibitors, whereas CABG patients were more often on beta-blockers, diuretics, anticoagulants, and antiarrhythmics.
Bottom line: PCI and CABG treatments for left main disease have no significant difference in the composite outcome of death, stroke, or MI at 5 years.
Citation: Stone GW et al. Five-year outcomes after PCI or CABG for left main coronary disease. N Engl J Med. 2019;381:1820-30.
Dr. Horton is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: While PCI with drug-eluting stents has become more accepted as treatment for some patients with left main disease, long-term outcomes from randomized control trials comparing PCI with CABG have yet to be clearly established.
Study design: International, open-label, multicenter, randomized trial.
Setting: A total of 126 sites in 17 countries.
Synopsis: Patients with low or intermediate anatomical complexity with 70% visual stenosis of the left main coronary artery or 50%-70% stenosis by noninvasive testing were randomized to either PCI (948) or CABG (957). Dual-antiplatelet therapy was given to PCI patients and aspirin to CABG patients. At 5 years there was no significant difference in the rate of the composite of death, stroke, or myocardial infarction (22.0% with PCI vs. 19.2% with CABG; difference, 2.8 percentage points; 95% CI, –0.9 to 6.5; P = .13). This was consistent across subgroups.
There were numerical differences in nonpowered secondary outcomes that may represent effects but should be interpreted cautiously: ischemia-driven revascularization (16.9% with PCI vs. 10% with CABG), transient ischemic attack plus stroke (3.3% with PCI vs. 5.2% with CABG), and death from any cause (3% with PCI vs. 9.9% with CABG). There was no significant difference in cardiovascular events, MI, or stroke.
One interesting limitation was that patients who had PCI were more commonly on dual-antiplatelet therapy and angiotensin converting–enzyme inhibitors, whereas CABG patients were more often on beta-blockers, diuretics, anticoagulants, and antiarrhythmics.
Bottom line: PCI and CABG treatments for left main disease have no significant difference in the composite outcome of death, stroke, or MI at 5 years.
Citation: Stone GW et al. Five-year outcomes after PCI or CABG for left main coronary disease. N Engl J Med. 2019;381:1820-30.
Dr. Horton is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: While PCI with drug-eluting stents has become more accepted as treatment for some patients with left main disease, long-term outcomes from randomized control trials comparing PCI with CABG have yet to be clearly established.
Study design: International, open-label, multicenter, randomized trial.
Setting: A total of 126 sites in 17 countries.
Synopsis: Patients with low or intermediate anatomical complexity with 70% visual stenosis of the left main coronary artery or 50%-70% stenosis by noninvasive testing were randomized to either PCI (948) or CABG (957). Dual-antiplatelet therapy was given to PCI patients and aspirin to CABG patients. At 5 years there was no significant difference in the rate of the composite of death, stroke, or myocardial infarction (22.0% with PCI vs. 19.2% with CABG; difference, 2.8 percentage points; 95% CI, –0.9 to 6.5; P = .13). This was consistent across subgroups.
There were numerical differences in nonpowered secondary outcomes that may represent effects but should be interpreted cautiously: ischemia-driven revascularization (16.9% with PCI vs. 10% with CABG), transient ischemic attack plus stroke (3.3% with PCI vs. 5.2% with CABG), and death from any cause (3% with PCI vs. 9.9% with CABG). There was no significant difference in cardiovascular events, MI, or stroke.
One interesting limitation was that patients who had PCI were more commonly on dual-antiplatelet therapy and angiotensin converting–enzyme inhibitors, whereas CABG patients were more often on beta-blockers, diuretics, anticoagulants, and antiarrhythmics.
Bottom line: PCI and CABG treatments for left main disease have no significant difference in the composite outcome of death, stroke, or MI at 5 years.
Citation: Stone GW et al. Five-year outcomes after PCI or CABG for left main coronary disease. N Engl J Med. 2019;381:1820-30.
Dr. Horton is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
The best exercises for BP control? European statement sorts it out
Recommendations for prescribing exercise to control high blood pressure have been put forward by various medical organizations and expert panels, but finding the bandwidth to craft personalized exercise training for their patients poses a challenge for clinicians.
Now, European cardiology societies have issued a consensus statement that offers an algorithm of sorts for developing personalized exercise programs as part of overall management approach for patients with or at risk of high BP.
The statement, published in the European Journal of Preventive Cardiology and issued by the European Association of Preventive Cardiology and the European Society of Cardiology Council on Hypertension, claims to be the first document to focus on personalized exercise for BP.
The statement draws on a systematic review, including meta-analyses, to produce guidance on how to lower BP in three specific types of patients: Those with hypertension (>140/90 mm Hg), high-normal blood pressure (130-139/85-89 mm Hg), and normal blood pressure (<130/84 mm Hg).
By making recommendations for these three specific groups, along with providing guidance for combined exercise – that is, blending aerobic exercise with resistance training (RT) – the consensus statement goes one step further than recommendations other organizations have issued, Matthew W. Martinez, MD, said in an interview.
“What it adds is an algorithmic approach, if you will,” said Dr. Martinez, a sports medicine cardiologist at Morristown (N.J.) Medical Center. “There are some recommendations to help the clinicians to decide what they’re going to offer individuals, but what’s a challenge for us when seeing patients is finding the time to deliver the message and explain how valuable nutrition and exercise are.”
Guidelines, updates, and statements that include the role of exercise in BP control have been issued by the European Society of Cardiology, American Heart Association, and American College of Sports Medicine (Med Sci Sports Exercise. 2019;51:1314-23).
The European consensus statement includes the expected range of BP lowering for each activity. For example, aerobic exercise for patients with hypertension should lead to a reduction from –4.9 to –12 mm Hg systolic and –3.4 to –5.8 mm Hg diastolic.
The consensus statement recommends the following exercise priorities based on a patient’s blood pressure:
- Hypertension: Aerobic training (AT) as a first-line exercise therapy; and low- to moderate-intensity RT – equally using dynamic and isometric RT – as second-line therapy. In non-White patients, dynamic RT should be considered as a first-line therapy. RT can be combined with aerobic exercise on an individual basis if the clinician determines either form of RT would provide a metabolic benefit.
- High-to-normal BP: Dynamic RT as a first-line exercise, which the systematic review determined led to greater BP reduction than that of aerobic training. “Isometric RT is likely to elicit similar if not superior BP-lowering effects as [dynamic RT], but the level of evidence is low and the available data are scarce,” wrote first author Henner Hanssen, MD, of the University of Basel, Switzerland, and coauthors. Combining dynamic resistance training with aerobic training “may be preferable” to dynamic RT alone in patients with a combination of cardiovascular risk factors.
- Normal BP: Isometric RT may be indicated as a first-line intervention in individuals with a family or gestational history or obese or overweight people currently with normal BP. This advice includes a caveat: “The number of studies is limited and the 95% confidence intervals are large,” Dr. Hanssen and coauthors noted. AT is also an option in these patients, with more high-quality meta-analyses than the recommendation for isometric RT. “Hence, the BP-lowering effects of [isometric RT] as compared to AT may be overestimated and both exercise modalities may have similar BP-lowering effects in individuals with normotension,” wrote the consensus statement authors.
They note that more research is needed to validate the BP-lowering effects of combined exercise.
The statement acknowledges the difficulty clinicians face in managing patients with high blood pressure. “From a socioeconomic health perspective, it is a major challenge to develop, promote, and implement individually tailored exercise programs for patients with hypertension under consideration of sustainable costs,” wrote Dr. Hanssen and coauthors.
Dr. Martinez noted that one strength of the consensus statement is that it addresses the impact exercise can have on vascular health and metabolic function. And, it points out existing knowledge gaps.
“Are we going to see greater applicability of this as we use IT health technology?” he asked. “Are wearables and telehealth going to help deliver this message more easily, more frequently? Is there work to be done in terms of differences in gender? Do men and women respond differently, and is there a different exercise prescription based on that as well as ethnicity? We well know there’s a different treatment for African Americans compared to other ethnic groups.”
The statement also raises the stakes for using exercise as part of a multifaceted, integrated approach to hypertension management, he said.
“It’s not enough to talk just about exercise or nutrition, or to just give an antihypertension medicine,” Dr. Martinez said. “Perhaps the sweet spot is in integrating an approach that includes all three.”
Consensus statement coauthor Antonio Coca, MD, reported financial relationships with Abbott, Berlin-Chemie, Biolab, Boehringer-Ingelheim, Ferrer, Menarini, Merck, Novartis and Sanofi-Aventis. Coauthor Maria Simonenko, MD, reported financial relationships with Novartis and Sanofi-Aventis. Linda Pescatello, PhD, is lead author of the American College of Sports Medicine 2019 statement. Dr. Hanssen and all other authors have no disclosures. Dr. Martinez has no relevant relationships to disclose.
Recommendations for prescribing exercise to control high blood pressure have been put forward by various medical organizations and expert panels, but finding the bandwidth to craft personalized exercise training for their patients poses a challenge for clinicians.
Now, European cardiology societies have issued a consensus statement that offers an algorithm of sorts for developing personalized exercise programs as part of overall management approach for patients with or at risk of high BP.
The statement, published in the European Journal of Preventive Cardiology and issued by the European Association of Preventive Cardiology and the European Society of Cardiology Council on Hypertension, claims to be the first document to focus on personalized exercise for BP.
The statement draws on a systematic review, including meta-analyses, to produce guidance on how to lower BP in three specific types of patients: Those with hypertension (>140/90 mm Hg), high-normal blood pressure (130-139/85-89 mm Hg), and normal blood pressure (<130/84 mm Hg).
By making recommendations for these three specific groups, along with providing guidance for combined exercise – that is, blending aerobic exercise with resistance training (RT) – the consensus statement goes one step further than recommendations other organizations have issued, Matthew W. Martinez, MD, said in an interview.
“What it adds is an algorithmic approach, if you will,” said Dr. Martinez, a sports medicine cardiologist at Morristown (N.J.) Medical Center. “There are some recommendations to help the clinicians to decide what they’re going to offer individuals, but what’s a challenge for us when seeing patients is finding the time to deliver the message and explain how valuable nutrition and exercise are.”
Guidelines, updates, and statements that include the role of exercise in BP control have been issued by the European Society of Cardiology, American Heart Association, and American College of Sports Medicine (Med Sci Sports Exercise. 2019;51:1314-23).
The European consensus statement includes the expected range of BP lowering for each activity. For example, aerobic exercise for patients with hypertension should lead to a reduction from –4.9 to –12 mm Hg systolic and –3.4 to –5.8 mm Hg diastolic.
The consensus statement recommends the following exercise priorities based on a patient’s blood pressure:
- Hypertension: Aerobic training (AT) as a first-line exercise therapy; and low- to moderate-intensity RT – equally using dynamic and isometric RT – as second-line therapy. In non-White patients, dynamic RT should be considered as a first-line therapy. RT can be combined with aerobic exercise on an individual basis if the clinician determines either form of RT would provide a metabolic benefit.
- High-to-normal BP: Dynamic RT as a first-line exercise, which the systematic review determined led to greater BP reduction than that of aerobic training. “Isometric RT is likely to elicit similar if not superior BP-lowering effects as [dynamic RT], but the level of evidence is low and the available data are scarce,” wrote first author Henner Hanssen, MD, of the University of Basel, Switzerland, and coauthors. Combining dynamic resistance training with aerobic training “may be preferable” to dynamic RT alone in patients with a combination of cardiovascular risk factors.
- Normal BP: Isometric RT may be indicated as a first-line intervention in individuals with a family or gestational history or obese or overweight people currently with normal BP. This advice includes a caveat: “The number of studies is limited and the 95% confidence intervals are large,” Dr. Hanssen and coauthors noted. AT is also an option in these patients, with more high-quality meta-analyses than the recommendation for isometric RT. “Hence, the BP-lowering effects of [isometric RT] as compared to AT may be overestimated and both exercise modalities may have similar BP-lowering effects in individuals with normotension,” wrote the consensus statement authors.
They note that more research is needed to validate the BP-lowering effects of combined exercise.
The statement acknowledges the difficulty clinicians face in managing patients with high blood pressure. “From a socioeconomic health perspective, it is a major challenge to develop, promote, and implement individually tailored exercise programs for patients with hypertension under consideration of sustainable costs,” wrote Dr. Hanssen and coauthors.
Dr. Martinez noted that one strength of the consensus statement is that it addresses the impact exercise can have on vascular health and metabolic function. And, it points out existing knowledge gaps.
“Are we going to see greater applicability of this as we use IT health technology?” he asked. “Are wearables and telehealth going to help deliver this message more easily, more frequently? Is there work to be done in terms of differences in gender? Do men and women respond differently, and is there a different exercise prescription based on that as well as ethnicity? We well know there’s a different treatment for African Americans compared to other ethnic groups.”
The statement also raises the stakes for using exercise as part of a multifaceted, integrated approach to hypertension management, he said.
“It’s not enough to talk just about exercise or nutrition, or to just give an antihypertension medicine,” Dr. Martinez said. “Perhaps the sweet spot is in integrating an approach that includes all three.”
Consensus statement coauthor Antonio Coca, MD, reported financial relationships with Abbott, Berlin-Chemie, Biolab, Boehringer-Ingelheim, Ferrer, Menarini, Merck, Novartis and Sanofi-Aventis. Coauthor Maria Simonenko, MD, reported financial relationships with Novartis and Sanofi-Aventis. Linda Pescatello, PhD, is lead author of the American College of Sports Medicine 2019 statement. Dr. Hanssen and all other authors have no disclosures. Dr. Martinez has no relevant relationships to disclose.
Recommendations for prescribing exercise to control high blood pressure have been put forward by various medical organizations and expert panels, but finding the bandwidth to craft personalized exercise training for their patients poses a challenge for clinicians.
Now, European cardiology societies have issued a consensus statement that offers an algorithm of sorts for developing personalized exercise programs as part of overall management approach for patients with or at risk of high BP.
The statement, published in the European Journal of Preventive Cardiology and issued by the European Association of Preventive Cardiology and the European Society of Cardiology Council on Hypertension, claims to be the first document to focus on personalized exercise for BP.
The statement draws on a systematic review, including meta-analyses, to produce guidance on how to lower BP in three specific types of patients: Those with hypertension (>140/90 mm Hg), high-normal blood pressure (130-139/85-89 mm Hg), and normal blood pressure (<130/84 mm Hg).
By making recommendations for these three specific groups, along with providing guidance for combined exercise – that is, blending aerobic exercise with resistance training (RT) – the consensus statement goes one step further than recommendations other organizations have issued, Matthew W. Martinez, MD, said in an interview.
“What it adds is an algorithmic approach, if you will,” said Dr. Martinez, a sports medicine cardiologist at Morristown (N.J.) Medical Center. “There are some recommendations to help the clinicians to decide what they’re going to offer individuals, but what’s a challenge for us when seeing patients is finding the time to deliver the message and explain how valuable nutrition and exercise are.”
Guidelines, updates, and statements that include the role of exercise in BP control have been issued by the European Society of Cardiology, American Heart Association, and American College of Sports Medicine (Med Sci Sports Exercise. 2019;51:1314-23).
The European consensus statement includes the expected range of BP lowering for each activity. For example, aerobic exercise for patients with hypertension should lead to a reduction from –4.9 to –12 mm Hg systolic and –3.4 to –5.8 mm Hg diastolic.
The consensus statement recommends the following exercise priorities based on a patient’s blood pressure:
- Hypertension: Aerobic training (AT) as a first-line exercise therapy; and low- to moderate-intensity RT – equally using dynamic and isometric RT – as second-line therapy. In non-White patients, dynamic RT should be considered as a first-line therapy. RT can be combined with aerobic exercise on an individual basis if the clinician determines either form of RT would provide a metabolic benefit.
- High-to-normal BP: Dynamic RT as a first-line exercise, which the systematic review determined led to greater BP reduction than that of aerobic training. “Isometric RT is likely to elicit similar if not superior BP-lowering effects as [dynamic RT], but the level of evidence is low and the available data are scarce,” wrote first author Henner Hanssen, MD, of the University of Basel, Switzerland, and coauthors. Combining dynamic resistance training with aerobic training “may be preferable” to dynamic RT alone in patients with a combination of cardiovascular risk factors.
- Normal BP: Isometric RT may be indicated as a first-line intervention in individuals with a family or gestational history or obese or overweight people currently with normal BP. This advice includes a caveat: “The number of studies is limited and the 95% confidence intervals are large,” Dr. Hanssen and coauthors noted. AT is also an option in these patients, with more high-quality meta-analyses than the recommendation for isometric RT. “Hence, the BP-lowering effects of [isometric RT] as compared to AT may be overestimated and both exercise modalities may have similar BP-lowering effects in individuals with normotension,” wrote the consensus statement authors.
They note that more research is needed to validate the BP-lowering effects of combined exercise.
The statement acknowledges the difficulty clinicians face in managing patients with high blood pressure. “From a socioeconomic health perspective, it is a major challenge to develop, promote, and implement individually tailored exercise programs for patients with hypertension under consideration of sustainable costs,” wrote Dr. Hanssen and coauthors.
Dr. Martinez noted that one strength of the consensus statement is that it addresses the impact exercise can have on vascular health and metabolic function. And, it points out existing knowledge gaps.
“Are we going to see greater applicability of this as we use IT health technology?” he asked. “Are wearables and telehealth going to help deliver this message more easily, more frequently? Is there work to be done in terms of differences in gender? Do men and women respond differently, and is there a different exercise prescription based on that as well as ethnicity? We well know there’s a different treatment for African Americans compared to other ethnic groups.”
The statement also raises the stakes for using exercise as part of a multifaceted, integrated approach to hypertension management, he said.
“It’s not enough to talk just about exercise or nutrition, or to just give an antihypertension medicine,” Dr. Martinez said. “Perhaps the sweet spot is in integrating an approach that includes all three.”
Consensus statement coauthor Antonio Coca, MD, reported financial relationships with Abbott, Berlin-Chemie, Biolab, Boehringer-Ingelheim, Ferrer, Menarini, Merck, Novartis and Sanofi-Aventis. Coauthor Maria Simonenko, MD, reported financial relationships with Novartis and Sanofi-Aventis. Linda Pescatello, PhD, is lead author of the American College of Sports Medicine 2019 statement. Dr. Hanssen and all other authors have no disclosures. Dr. Martinez has no relevant relationships to disclose.
FROM THE EUROPEAN JOURNAL OF PREVENTIVE CARDIOLOGY
Long-haul COVID-19 brings welcome attention to POTS
Before COVID-19, postural orthostatic tachycardia syndrome (POTS) was one of those diseases that many people, including physicians, dismissed.
“They thought it was just anxious, crazy young women,” said Pam R. Taub, MD, who runs the cardiac rehabilitation program at the University of California, San Diego.
The cryptic autonomic condition was estimated to affect 1-3 million Americans before the pandemic hit. Now case reports confirm that it is a manifestation of postacute sequelae of SARS-CoV-2 infection (PASC), or so-called long-haul COVID-19.
“I’m excited that this condition that has been so often the ugly stepchild of both cardiology and neurology is getting some attention,” said Dr. Taub. She said she is hopeful that the National Institutes of Health’s commitment to PASC research will benefit patients affected by the cardiovascular dysautonomia characterized by orthostatic intolerance in the absence of orthostatic hypotension.
Postinfection POTS is not exclusive to SARS-CoV-2. It has been reported after Lyme disease and Epstein-Barr virus infections, for example. One theory is that some of the antibodies generated against the virus cross react and damage the autonomic nervous system, which regulates heart rate and blood pressure, Dr. Taub explained.
It is not known whether COVID-19 is more likely to trigger POTS than are other infections or whether the rise in cases merely reflects the fact that more than 115 million people worldwide have been infected with the novel coronavirus.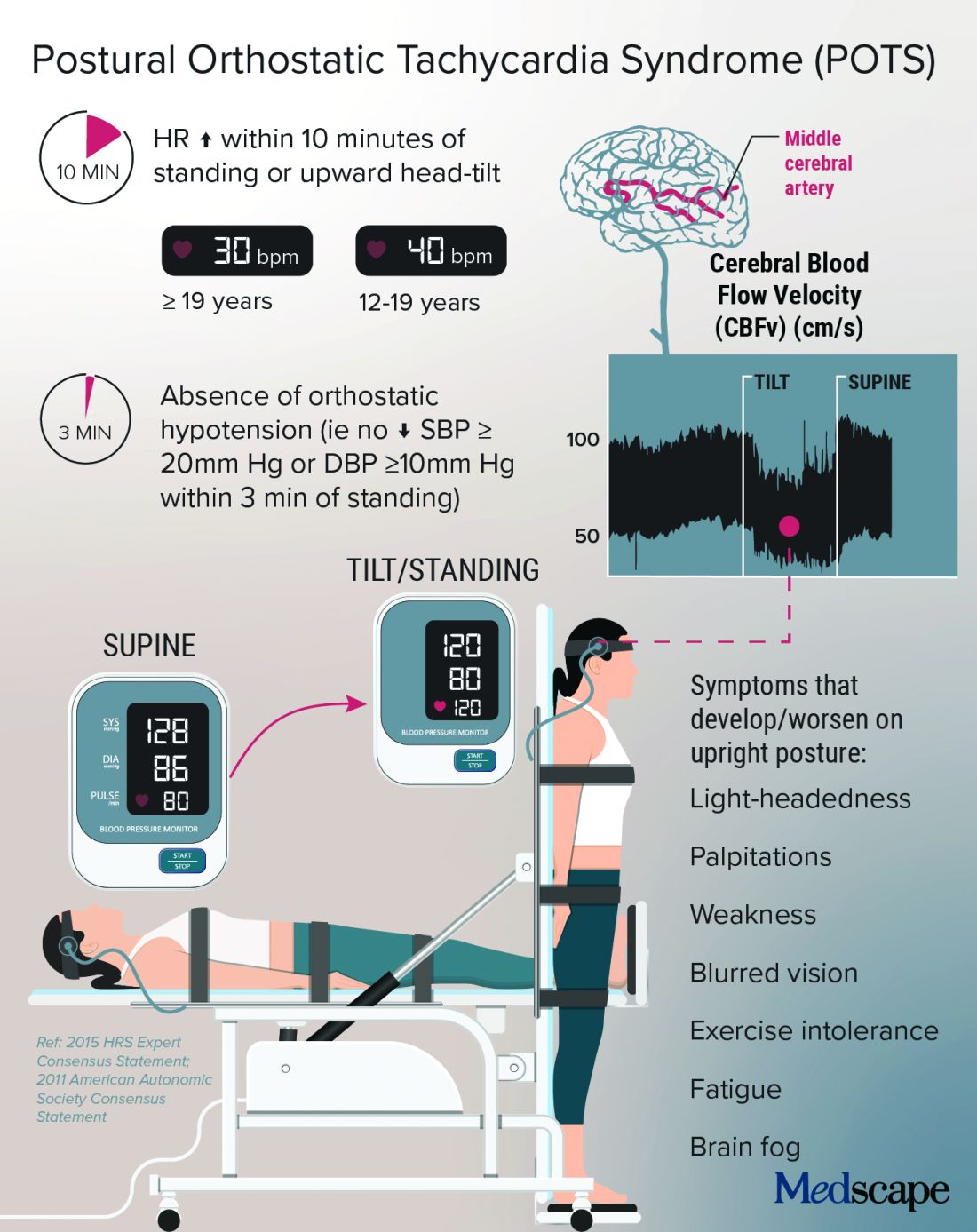
Low blood volume, dysregulation of the autonomic nervous system, and autoimmunity may all play a role in POTS, perhaps leading to distinct subtypes, according to a State of the Science document from the NIH; the National Heart, Lung, and Blood Institute; and the National Institute of Neurological Disorders and Stroke.
In Dr. Taub’s experience, “The truth is that patients actually have a mix of the subtypes.”
Kamal Shouman, MD, an autonomic neurologist at Mayo Clinic, Rochester, Minn., said in an interview that he has seen patients present with post–COVID-19 POTS in “all flavors,” including “neuropathic POTS, which is thought of as the classic postinfectious phenomenon.”
Why does it mostly affect athletic women?
The condition, which can be the result of dehydration or prolonged bed rest, leading to deconditioning, affects women disproportionately.
According to Manesh Patel, MD, if a patient with POTS who is not a young woman is presented on medical rounds, the response is, “Tell me again why you think this patient has POTS.”
Dr. Patel, chief of the division of cardiology at Duke University, Durham, N.C., has a theory for why many of the women who have POTS are athletes or are highly active: They likely have an underlying predisposition, compounded by a smaller body volume, leaving less margin for error. “If they decondition and lose 500 cc’s, it makes a bigger difference to them than, say, a 300-pound offensive lineman,” Dr. Patel explained.
That hypothesis makes sense to Dr. Taub, who added, “There are just some people metabolically that are more hyperadrenergic,” and it may be that “all their activity really helps tone down that sympathetic output,” but the infection affects these regulatory processes, and deconditioning disrupts things further.
Women also have more autoimmune disorders than do men. The driving force of the dysregulation of the autonomic nervous system is thought to be “immune mediated; we think it’s triggered by a response to a virus,” she said.
Dr. Shouman said the underlying susceptibility may predispose toward orthostatic intolerance. For example, patients will tell him, “Well, many years ago, I was prone to fainting.” He emphasized that POTS is not exclusive to women – he sees men with POTS, and one of the three recent case reports of post–COVID-19 POTS involved a 37-year-old man. So far, the male POTS patients that Dr. Patel has encountered have been deconditioned athletes.
Poor (wo)man’s tilt test and treatment options
POTS is typically diagnosed with a tilt test and transcranial Doppler. Dr. Taub described her “poor man’s tilt test” of asking the patient to lie down for 5-10 minutes and then having the patient stand up.
She likes the fact that transcranial Doppler helps validate the brain fog that patients report, which can be dismissed as “just your excuse for not wanting to work.” If blood perfusion to the brain is cut by 40%-50%, “how are you going to think clearly?” she said.
Dr. Shouman noted that overall volume expansion with salt water, compression garments, and a graduated exercise program play a major role in the rehabilitation of all POTS patients.
He likes to tailor treatments to the most likely underlying cause. But patients should first undergo a medical assessment by their internists to make sure there isn’t a primary lung or heart problem.
“Once the decision is made for them to be evaluated in the autonomic practice and [a] POTS diagnosis is made, I think it is very useful to determine what type of POTS,” he said.
With hyperadrenergic POTS, “you are looking at a standing norepinephrine level of over 600 pg/mL or so.” For these patients, drugs such as ivabradine or beta-blockers can help, he noted.
Dr. Taub recently conducted a small study that showed a benefit with the selective If channel blocker ivabradine for patients with hyperadrenergic POTS unrelated to COVID-19. She tends to favor ivabradine over beta-blockers because it lowers heart rate but not blood pressure. In addition, beta-blockers can exacerbate fatigue and brain fog.
A small crossover study will compare propranolol and ivabradine in POTS. For someone who is very hypovolemic, “you might try a salt tablet or a prescription drug like fludrocortisone,” Dr. Taub explained.
Another problem that patients with POTS experience is an inability to exercise because of orthostatic intolerance. Recumbent exercise targets deconditioning and can tamp down the hyperadrenergic effect. Dr. Shouman’s approach is to start gradually with swimming or the use of a recumbent bike or a rowing machine.
Dr. Taub recommends wearables to patients because POTS is “a very dynamic condition” that is easy to overmedicate or undermedicate. If it’s a good day, the patients are well hydrated, and the standing heart rate is only 80 bpm, she tells them they could titrate down their second dose of ivabradine, for example. The feedback from wearables also helps patients manage their exercise response.
For Dr. Shouman, wearables are not always as accurate as he would like. He tells his patients that it’s okay to use one as long as it doesn’t become a source of anxiety such that they’re constantly checking it.
POTS hope: A COVID-19 silver lining?
With increasing attention being paid to long-haul COVID-19, are there any concerns that POTS will get lost among the myriad symptoms connected to PASC?
Dr. Shouman cautioned, “Not all long COVID is POTS,” and said that clinicians at long-haul clinics should be able to recognize the different conditions “when POTS is suspected. I think it is useful for those providers to make the appropriate referral for POTS clinic autonomic assessment.”
He and his colleagues at Mayo have seen quite a few patients who have post–COVID-19 autonomic dysfunction, such as vasodepressor syncope, not just POTS. They plan to write about this soon.
“Of all the things I treat in cardiology, this is the most complex, because there’s so many different systems involved,” said Dr. Taub, who has seen patients recover fully from POTS. “There’s a spectrum, and there’s people that are definitely on one end of the spectrum where they have very severe diseases.”
For her, the important message is, “No matter where you are on the spectrum, there are things we can do to make your symptoms better.” And with grant funding for PASC research, “hopefully we will address the mechanisms of disease, and we’ll be able to cure this,” she said.
Dr. Patel has served as a consultant for Bayer, Janssen, AstraZeneca, and Heartflow and has received research grants from Bayer, Janssen, AstraZeneca, and the National Heart, Lung, and Blood Institute. Dr. Shouman reports no relevant financial relationships. Dr. Taub has served as a consultant for Amgen, Bayer, Esperion, Boehringer Ingelheim, Novo Nordisk, and Sanofi; is a shareholder in Epirium Bio; and has received research grants from the National Institutes of Health, the American Heart Association, and the Department of Homeland Security/FEMA.
A version of this article first appeared on Medscape.com.
Before COVID-19, postural orthostatic tachycardia syndrome (POTS) was one of those diseases that many people, including physicians, dismissed.
“They thought it was just anxious, crazy young women,” said Pam R. Taub, MD, who runs the cardiac rehabilitation program at the University of California, San Diego.
The cryptic autonomic condition was estimated to affect 1-3 million Americans before the pandemic hit. Now case reports confirm that it is a manifestation of postacute sequelae of SARS-CoV-2 infection (PASC), or so-called long-haul COVID-19.
“I’m excited that this condition that has been so often the ugly stepchild of both cardiology and neurology is getting some attention,” said Dr. Taub. She said she is hopeful that the National Institutes of Health’s commitment to PASC research will benefit patients affected by the cardiovascular dysautonomia characterized by orthostatic intolerance in the absence of orthostatic hypotension.
Postinfection POTS is not exclusive to SARS-CoV-2. It has been reported after Lyme disease and Epstein-Barr virus infections, for example. One theory is that some of the antibodies generated against the virus cross react and damage the autonomic nervous system, which regulates heart rate and blood pressure, Dr. Taub explained.
It is not known whether COVID-19 is more likely to trigger POTS than are other infections or whether the rise in cases merely reflects the fact that more than 115 million people worldwide have been infected with the novel coronavirus.
Low blood volume, dysregulation of the autonomic nervous system, and autoimmunity may all play a role in POTS, perhaps leading to distinct subtypes, according to a State of the Science document from the NIH; the National Heart, Lung, and Blood Institute; and the National Institute of Neurological Disorders and Stroke.
In Dr. Taub’s experience, “The truth is that patients actually have a mix of the subtypes.”
Kamal Shouman, MD, an autonomic neurologist at Mayo Clinic, Rochester, Minn., said in an interview that he has seen patients present with post–COVID-19 POTS in “all flavors,” including “neuropathic POTS, which is thought of as the classic postinfectious phenomenon.”
Why does it mostly affect athletic women?
The condition, which can be the result of dehydration or prolonged bed rest, leading to deconditioning, affects women disproportionately.
According to Manesh Patel, MD, if a patient with POTS who is not a young woman is presented on medical rounds, the response is, “Tell me again why you think this patient has POTS.”
Dr. Patel, chief of the division of cardiology at Duke University, Durham, N.C., has a theory for why many of the women who have POTS are athletes or are highly active: They likely have an underlying predisposition, compounded by a smaller body volume, leaving less margin for error. “If they decondition and lose 500 cc’s, it makes a bigger difference to them than, say, a 300-pound offensive lineman,” Dr. Patel explained.
That hypothesis makes sense to Dr. Taub, who added, “There are just some people metabolically that are more hyperadrenergic,” and it may be that “all their activity really helps tone down that sympathetic output,” but the infection affects these regulatory processes, and deconditioning disrupts things further.
Women also have more autoimmune disorders than do men. The driving force of the dysregulation of the autonomic nervous system is thought to be “immune mediated; we think it’s triggered by a response to a virus,” she said.
Dr. Shouman said the underlying susceptibility may predispose toward orthostatic intolerance. For example, patients will tell him, “Well, many years ago, I was prone to fainting.” He emphasized that POTS is not exclusive to women – he sees men with POTS, and one of the three recent case reports of post–COVID-19 POTS involved a 37-year-old man. So far, the male POTS patients that Dr. Patel has encountered have been deconditioned athletes.
Poor (wo)man’s tilt test and treatment options
POTS is typically diagnosed with a tilt test and transcranial Doppler. Dr. Taub described her “poor man’s tilt test” of asking the patient to lie down for 5-10 minutes and then having the patient stand up.
She likes the fact that transcranial Doppler helps validate the brain fog that patients report, which can be dismissed as “just your excuse for not wanting to work.” If blood perfusion to the brain is cut by 40%-50%, “how are you going to think clearly?” she said.
Dr. Shouman noted that overall volume expansion with salt water, compression garments, and a graduated exercise program play a major role in the rehabilitation of all POTS patients.
He likes to tailor treatments to the most likely underlying cause. But patients should first undergo a medical assessment by their internists to make sure there isn’t a primary lung or heart problem.
“Once the decision is made for them to be evaluated in the autonomic practice and [a] POTS diagnosis is made, I think it is very useful to determine what type of POTS,” he said.
With hyperadrenergic POTS, “you are looking at a standing norepinephrine level of over 600 pg/mL or so.” For these patients, drugs such as ivabradine or beta-blockers can help, he noted.
Dr. Taub recently conducted a small study that showed a benefit with the selective If channel blocker ivabradine for patients with hyperadrenergic POTS unrelated to COVID-19. She tends to favor ivabradine over beta-blockers because it lowers heart rate but not blood pressure. In addition, beta-blockers can exacerbate fatigue and brain fog.
A small crossover study will compare propranolol and ivabradine in POTS. For someone who is very hypovolemic, “you might try a salt tablet or a prescription drug like fludrocortisone,” Dr. Taub explained.
Another problem that patients with POTS experience is an inability to exercise because of orthostatic intolerance. Recumbent exercise targets deconditioning and can tamp down the hyperadrenergic effect. Dr. Shouman’s approach is to start gradually with swimming or the use of a recumbent bike or a rowing machine.
Dr. Taub recommends wearables to patients because POTS is “a very dynamic condition” that is easy to overmedicate or undermedicate. If it’s a good day, the patients are well hydrated, and the standing heart rate is only 80 bpm, she tells them they could titrate down their second dose of ivabradine, for example. The feedback from wearables also helps patients manage their exercise response.
For Dr. Shouman, wearables are not always as accurate as he would like. He tells his patients that it’s okay to use one as long as it doesn’t become a source of anxiety such that they’re constantly checking it.
POTS hope: A COVID-19 silver lining?
With increasing attention being paid to long-haul COVID-19, are there any concerns that POTS will get lost among the myriad symptoms connected to PASC?
Dr. Shouman cautioned, “Not all long COVID is POTS,” and said that clinicians at long-haul clinics should be able to recognize the different conditions “when POTS is suspected. I think it is useful for those providers to make the appropriate referral for POTS clinic autonomic assessment.”
He and his colleagues at Mayo have seen quite a few patients who have post–COVID-19 autonomic dysfunction, such as vasodepressor syncope, not just POTS. They plan to write about this soon.
“Of all the things I treat in cardiology, this is the most complex, because there’s so many different systems involved,” said Dr. Taub, who has seen patients recover fully from POTS. “There’s a spectrum, and there’s people that are definitely on one end of the spectrum where they have very severe diseases.”
For her, the important message is, “No matter where you are on the spectrum, there are things we can do to make your symptoms better.” And with grant funding for PASC research, “hopefully we will address the mechanisms of disease, and we’ll be able to cure this,” she said.
Dr. Patel has served as a consultant for Bayer, Janssen, AstraZeneca, and Heartflow and has received research grants from Bayer, Janssen, AstraZeneca, and the National Heart, Lung, and Blood Institute. Dr. Shouman reports no relevant financial relationships. Dr. Taub has served as a consultant for Amgen, Bayer, Esperion, Boehringer Ingelheim, Novo Nordisk, and Sanofi; is a shareholder in Epirium Bio; and has received research grants from the National Institutes of Health, the American Heart Association, and the Department of Homeland Security/FEMA.
A version of this article first appeared on Medscape.com.
Before COVID-19, postural orthostatic tachycardia syndrome (POTS) was one of those diseases that many people, including physicians, dismissed.
“They thought it was just anxious, crazy young women,” said Pam R. Taub, MD, who runs the cardiac rehabilitation program at the University of California, San Diego.
The cryptic autonomic condition was estimated to affect 1-3 million Americans before the pandemic hit. Now case reports confirm that it is a manifestation of postacute sequelae of SARS-CoV-2 infection (PASC), or so-called long-haul COVID-19.
“I’m excited that this condition that has been so often the ugly stepchild of both cardiology and neurology is getting some attention,” said Dr. Taub. She said she is hopeful that the National Institutes of Health’s commitment to PASC research will benefit patients affected by the cardiovascular dysautonomia characterized by orthostatic intolerance in the absence of orthostatic hypotension.
Postinfection POTS is not exclusive to SARS-CoV-2. It has been reported after Lyme disease and Epstein-Barr virus infections, for example. One theory is that some of the antibodies generated against the virus cross react and damage the autonomic nervous system, which regulates heart rate and blood pressure, Dr. Taub explained.
It is not known whether COVID-19 is more likely to trigger POTS than are other infections or whether the rise in cases merely reflects the fact that more than 115 million people worldwide have been infected with the novel coronavirus.
Low blood volume, dysregulation of the autonomic nervous system, and autoimmunity may all play a role in POTS, perhaps leading to distinct subtypes, according to a State of the Science document from the NIH; the National Heart, Lung, and Blood Institute; and the National Institute of Neurological Disorders and Stroke.
In Dr. Taub’s experience, “The truth is that patients actually have a mix of the subtypes.”
Kamal Shouman, MD, an autonomic neurologist at Mayo Clinic, Rochester, Minn., said in an interview that he has seen patients present with post–COVID-19 POTS in “all flavors,” including “neuropathic POTS, which is thought of as the classic postinfectious phenomenon.”
Why does it mostly affect athletic women?
The condition, which can be the result of dehydration or prolonged bed rest, leading to deconditioning, affects women disproportionately.
According to Manesh Patel, MD, if a patient with POTS who is not a young woman is presented on medical rounds, the response is, “Tell me again why you think this patient has POTS.”
Dr. Patel, chief of the division of cardiology at Duke University, Durham, N.C., has a theory for why many of the women who have POTS are athletes or are highly active: They likely have an underlying predisposition, compounded by a smaller body volume, leaving less margin for error. “If they decondition and lose 500 cc’s, it makes a bigger difference to them than, say, a 300-pound offensive lineman,” Dr. Patel explained.
That hypothesis makes sense to Dr. Taub, who added, “There are just some people metabolically that are more hyperadrenergic,” and it may be that “all their activity really helps tone down that sympathetic output,” but the infection affects these regulatory processes, and deconditioning disrupts things further.
Women also have more autoimmune disorders than do men. The driving force of the dysregulation of the autonomic nervous system is thought to be “immune mediated; we think it’s triggered by a response to a virus,” she said.
Dr. Shouman said the underlying susceptibility may predispose toward orthostatic intolerance. For example, patients will tell him, “Well, many years ago, I was prone to fainting.” He emphasized that POTS is not exclusive to women – he sees men with POTS, and one of the three recent case reports of post–COVID-19 POTS involved a 37-year-old man. So far, the male POTS patients that Dr. Patel has encountered have been deconditioned athletes.
Poor (wo)man’s tilt test and treatment options
POTS is typically diagnosed with a tilt test and transcranial Doppler. Dr. Taub described her “poor man’s tilt test” of asking the patient to lie down for 5-10 minutes and then having the patient stand up.
She likes the fact that transcranial Doppler helps validate the brain fog that patients report, which can be dismissed as “just your excuse for not wanting to work.” If blood perfusion to the brain is cut by 40%-50%, “how are you going to think clearly?” she said.
Dr. Shouman noted that overall volume expansion with salt water, compression garments, and a graduated exercise program play a major role in the rehabilitation of all POTS patients.
He likes to tailor treatments to the most likely underlying cause. But patients should first undergo a medical assessment by their internists to make sure there isn’t a primary lung or heart problem.
“Once the decision is made for them to be evaluated in the autonomic practice and [a] POTS diagnosis is made, I think it is very useful to determine what type of POTS,” he said.
With hyperadrenergic POTS, “you are looking at a standing norepinephrine level of over 600 pg/mL or so.” For these patients, drugs such as ivabradine or beta-blockers can help, he noted.
Dr. Taub recently conducted a small study that showed a benefit with the selective If channel blocker ivabradine for patients with hyperadrenergic POTS unrelated to COVID-19. She tends to favor ivabradine over beta-blockers because it lowers heart rate but not blood pressure. In addition, beta-blockers can exacerbate fatigue and brain fog.
A small crossover study will compare propranolol and ivabradine in POTS. For someone who is very hypovolemic, “you might try a salt tablet or a prescription drug like fludrocortisone,” Dr. Taub explained.
Another problem that patients with POTS experience is an inability to exercise because of orthostatic intolerance. Recumbent exercise targets deconditioning and can tamp down the hyperadrenergic effect. Dr. Shouman’s approach is to start gradually with swimming or the use of a recumbent bike or a rowing machine.
Dr. Taub recommends wearables to patients because POTS is “a very dynamic condition” that is easy to overmedicate or undermedicate. If it’s a good day, the patients are well hydrated, and the standing heart rate is only 80 bpm, she tells them they could titrate down their second dose of ivabradine, for example. The feedback from wearables also helps patients manage their exercise response.
For Dr. Shouman, wearables are not always as accurate as he would like. He tells his patients that it’s okay to use one as long as it doesn’t become a source of anxiety such that they’re constantly checking it.
POTS hope: A COVID-19 silver lining?
With increasing attention being paid to long-haul COVID-19, are there any concerns that POTS will get lost among the myriad symptoms connected to PASC?
Dr. Shouman cautioned, “Not all long COVID is POTS,” and said that clinicians at long-haul clinics should be able to recognize the different conditions “when POTS is suspected. I think it is useful for those providers to make the appropriate referral for POTS clinic autonomic assessment.”
He and his colleagues at Mayo have seen quite a few patients who have post–COVID-19 autonomic dysfunction, such as vasodepressor syncope, not just POTS. They plan to write about this soon.
“Of all the things I treat in cardiology, this is the most complex, because there’s so many different systems involved,” said Dr. Taub, who has seen patients recover fully from POTS. “There’s a spectrum, and there’s people that are definitely on one end of the spectrum where they have very severe diseases.”
For her, the important message is, “No matter where you are on the spectrum, there are things we can do to make your symptoms better.” And with grant funding for PASC research, “hopefully we will address the mechanisms of disease, and we’ll be able to cure this,” she said.
Dr. Patel has served as a consultant for Bayer, Janssen, AstraZeneca, and Heartflow and has received research grants from Bayer, Janssen, AstraZeneca, and the National Heart, Lung, and Blood Institute. Dr. Shouman reports no relevant financial relationships. Dr. Taub has served as a consultant for Amgen, Bayer, Esperion, Boehringer Ingelheim, Novo Nordisk, and Sanofi; is a shareholder in Epirium Bio; and has received research grants from the National Institutes of Health, the American Heart Association, and the Department of Homeland Security/FEMA.
A version of this article first appeared on Medscape.com.
Febuxostat, allopurinol real-world cardiovascular risk appears equal
Febuxostat (Uloric) was not associated with increased cardiovascular risk in patients with gout when compared to those who used allopurinol, in an analysis of new users of the drugs in Medicare fee-for-service claims data from the period of 2008-2016.
The findings, published March 25 in the Journal of the American Heart Association, update and echo the results from a similar previous study by the same Brigham and Women’s Hospital research group that covered 2008-2013 Medicare claims data. That original claims data study from 2018 sought to confirm the findings of the postmarketing surveillance CARES (Cardiovascular Safety of Febuxostat and Allopurinol in Patients With Gout and Cardiovascular Morbidities) trial that led to a boxed warning for increased risk of cardiovascular and all-cause mortality vs. allopurinol. The trial, however, did not show a higher rate of major adverse cardiovascular events (MACE) overall with febuxostat.
The recency of the new data with more febuxostat-exposed patients overall provides greater reassurance on the safety of the drug, corresponding author Seoyoung C. Kim, MD, of Brigham and Women’s Hospital and Harvard Medical School, Boston, said in an interview. “We also were able to get data on cause of death, which we did not have before when we conducted our first paper.”
Dr. Kim said she was not surprised by any of the findings, which were consistent with the results of her earlier work. “Our result on CV death also was consistent and reassuring,” she noted.
The newest Medicare claims study also corroborates results from FAST (Febuxostat Versus Allopurinol Streamlined Trial), a separate postmarketing surveillance study that was ordered by the European Medicines Agency after febuxostat’s approval in 2009. It showed that the two drugs were noninferior to each other for the risk of all-cause mortality or a composite cardiovascular outcome (hospitalization for nonfatal myocardial infarction, biomarker-positive acute coronary syndrome, nonfatal stroke, or cardiovascular death).
“While CARES showed higher CV death and all-cause death rates in febuxostat compared to allopurinol, FAST did not,” Dr. Kim noted. “Our study of more than 111,000 older gout patients treated with either febuxostat or allopurinol in real-world settings also did not find a difference in the risk of MACE, CV mortality, or all-cause mortality,” she added. “Taking these data all together, I think we can be more certain about the CV safety of febuxostat when its use is clinically indicated or needed,” she said.
Study details
Dr. Kim, first author Ajinkya Pawar, PhD, of Brigham and Women’s, and colleagues identified 467,461 people with gout aged 65 years and older who had been enrolled in Medicare for at least a year. They then used propensity-score matching to compare 27,881 first-time users of febuxostat with 83,643 first-time users of allopurinol on the primary outcome of the incidence of major adverse cardiovascular events (MACE), defined as the first occurrence of myocardial infarction, stroke, or cardiovascular mortality.
In the updated study, the mean follow‐up periods for febuxostat and allopurinol were 284 days and 339 days, respectively. Overall, febuxostat was noninferior to allopurinol with regard to MACE (hazard ratio, 0.99; 95% confidence interval, 0.93-1.05), and the results were consistent among patients with baseline CVD (HR, 0.94). In addition, rates of secondary outcomes of MI, stroke, and cardiovascular mortality were not significantly different between febuxostat and allopurinol patients, except for all-cause mortality (HR, 0.92; 95% CI, 0.87-0.98).
The study findings were limited mainly by the potential bias caused by nonadherence to medications, and potential for residual confounding and misclassification bias, the researchers noted.
However, the study was strengthened by its incident new-user design that allowed only patients with no use of either medication for a year before the first dispensing and its active comparator design, and the data are generalizable to the greater population of older gout patients, they said.
Consequently, the data from this large, real-world study support the safety of febuxostat with regard to cardiovascular risk in gout patients, including those with baseline cardiovascular disease, they concluded.
The study was supported by the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital. Dr. Kim disclosed research grants to Brigham and Women’s Hospital from Roche, Pfizer, AbbVie, and Bristol‐Myers Squibb for unrelated studies. Another author reported serving as the principal investigator with research grants from Vertex, Bayer, and Novartis to Brigham and Women’s Hospital for unrelated projects.
Febuxostat (Uloric) was not associated with increased cardiovascular risk in patients with gout when compared to those who used allopurinol, in an analysis of new users of the drugs in Medicare fee-for-service claims data from the period of 2008-2016.
The findings, published March 25 in the Journal of the American Heart Association, update and echo the results from a similar previous study by the same Brigham and Women’s Hospital research group that covered 2008-2013 Medicare claims data. That original claims data study from 2018 sought to confirm the findings of the postmarketing surveillance CARES (Cardiovascular Safety of Febuxostat and Allopurinol in Patients With Gout and Cardiovascular Morbidities) trial that led to a boxed warning for increased risk of cardiovascular and all-cause mortality vs. allopurinol. The trial, however, did not show a higher rate of major adverse cardiovascular events (MACE) overall with febuxostat.
The recency of the new data with more febuxostat-exposed patients overall provides greater reassurance on the safety of the drug, corresponding author Seoyoung C. Kim, MD, of Brigham and Women’s Hospital and Harvard Medical School, Boston, said in an interview. “We also were able to get data on cause of death, which we did not have before when we conducted our first paper.”
Dr. Kim said she was not surprised by any of the findings, which were consistent with the results of her earlier work. “Our result on CV death also was consistent and reassuring,” she noted.
The newest Medicare claims study also corroborates results from FAST (Febuxostat Versus Allopurinol Streamlined Trial), a separate postmarketing surveillance study that was ordered by the European Medicines Agency after febuxostat’s approval in 2009. It showed that the two drugs were noninferior to each other for the risk of all-cause mortality or a composite cardiovascular outcome (hospitalization for nonfatal myocardial infarction, biomarker-positive acute coronary syndrome, nonfatal stroke, or cardiovascular death).
“While CARES showed higher CV death and all-cause death rates in febuxostat compared to allopurinol, FAST did not,” Dr. Kim noted. “Our study of more than 111,000 older gout patients treated with either febuxostat or allopurinol in real-world settings also did not find a difference in the risk of MACE, CV mortality, or all-cause mortality,” she added. “Taking these data all together, I think we can be more certain about the CV safety of febuxostat when its use is clinically indicated or needed,” she said.
Study details
Dr. Kim, first author Ajinkya Pawar, PhD, of Brigham and Women’s, and colleagues identified 467,461 people with gout aged 65 years and older who had been enrolled in Medicare for at least a year. They then used propensity-score matching to compare 27,881 first-time users of febuxostat with 83,643 first-time users of allopurinol on the primary outcome of the incidence of major adverse cardiovascular events (MACE), defined as the first occurrence of myocardial infarction, stroke, or cardiovascular mortality.
In the updated study, the mean follow‐up periods for febuxostat and allopurinol were 284 days and 339 days, respectively. Overall, febuxostat was noninferior to allopurinol with regard to MACE (hazard ratio, 0.99; 95% confidence interval, 0.93-1.05), and the results were consistent among patients with baseline CVD (HR, 0.94). In addition, rates of secondary outcomes of MI, stroke, and cardiovascular mortality were not significantly different between febuxostat and allopurinol patients, except for all-cause mortality (HR, 0.92; 95% CI, 0.87-0.98).
The study findings were limited mainly by the potential bias caused by nonadherence to medications, and potential for residual confounding and misclassification bias, the researchers noted.
However, the study was strengthened by its incident new-user design that allowed only patients with no use of either medication for a year before the first dispensing and its active comparator design, and the data are generalizable to the greater population of older gout patients, they said.
Consequently, the data from this large, real-world study support the safety of febuxostat with regard to cardiovascular risk in gout patients, including those with baseline cardiovascular disease, they concluded.
The study was supported by the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital. Dr. Kim disclosed research grants to Brigham and Women’s Hospital from Roche, Pfizer, AbbVie, and Bristol‐Myers Squibb for unrelated studies. Another author reported serving as the principal investigator with research grants from Vertex, Bayer, and Novartis to Brigham and Women’s Hospital for unrelated projects.
Febuxostat (Uloric) was not associated with increased cardiovascular risk in patients with gout when compared to those who used allopurinol, in an analysis of new users of the drugs in Medicare fee-for-service claims data from the period of 2008-2016.
The findings, published March 25 in the Journal of the American Heart Association, update and echo the results from a similar previous study by the same Brigham and Women’s Hospital research group that covered 2008-2013 Medicare claims data. That original claims data study from 2018 sought to confirm the findings of the postmarketing surveillance CARES (Cardiovascular Safety of Febuxostat and Allopurinol in Patients With Gout and Cardiovascular Morbidities) trial that led to a boxed warning for increased risk of cardiovascular and all-cause mortality vs. allopurinol. The trial, however, did not show a higher rate of major adverse cardiovascular events (MACE) overall with febuxostat.
The recency of the new data with more febuxostat-exposed patients overall provides greater reassurance on the safety of the drug, corresponding author Seoyoung C. Kim, MD, of Brigham and Women’s Hospital and Harvard Medical School, Boston, said in an interview. “We also were able to get data on cause of death, which we did not have before when we conducted our first paper.”
Dr. Kim said she was not surprised by any of the findings, which were consistent with the results of her earlier work. “Our result on CV death also was consistent and reassuring,” she noted.
The newest Medicare claims study also corroborates results from FAST (Febuxostat Versus Allopurinol Streamlined Trial), a separate postmarketing surveillance study that was ordered by the European Medicines Agency after febuxostat’s approval in 2009. It showed that the two drugs were noninferior to each other for the risk of all-cause mortality or a composite cardiovascular outcome (hospitalization for nonfatal myocardial infarction, biomarker-positive acute coronary syndrome, nonfatal stroke, or cardiovascular death).
“While CARES showed higher CV death and all-cause death rates in febuxostat compared to allopurinol, FAST did not,” Dr. Kim noted. “Our study of more than 111,000 older gout patients treated with either febuxostat or allopurinol in real-world settings also did not find a difference in the risk of MACE, CV mortality, or all-cause mortality,” she added. “Taking these data all together, I think we can be more certain about the CV safety of febuxostat when its use is clinically indicated or needed,” she said.
Study details
Dr. Kim, first author Ajinkya Pawar, PhD, of Brigham and Women’s, and colleagues identified 467,461 people with gout aged 65 years and older who had been enrolled in Medicare for at least a year. They then used propensity-score matching to compare 27,881 first-time users of febuxostat with 83,643 first-time users of allopurinol on the primary outcome of the incidence of major adverse cardiovascular events (MACE), defined as the first occurrence of myocardial infarction, stroke, or cardiovascular mortality.
In the updated study, the mean follow‐up periods for febuxostat and allopurinol were 284 days and 339 days, respectively. Overall, febuxostat was noninferior to allopurinol with regard to MACE (hazard ratio, 0.99; 95% confidence interval, 0.93-1.05), and the results were consistent among patients with baseline CVD (HR, 0.94). In addition, rates of secondary outcomes of MI, stroke, and cardiovascular mortality were not significantly different between febuxostat and allopurinol patients, except for all-cause mortality (HR, 0.92; 95% CI, 0.87-0.98).
The study findings were limited mainly by the potential bias caused by nonadherence to medications, and potential for residual confounding and misclassification bias, the researchers noted.
However, the study was strengthened by its incident new-user design that allowed only patients with no use of either medication for a year before the first dispensing and its active comparator design, and the data are generalizable to the greater population of older gout patients, they said.
Consequently, the data from this large, real-world study support the safety of febuxostat with regard to cardiovascular risk in gout patients, including those with baseline cardiovascular disease, they concluded.
The study was supported by the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital. Dr. Kim disclosed research grants to Brigham and Women’s Hospital from Roche, Pfizer, AbbVie, and Bristol‐Myers Squibb for unrelated studies. Another author reported serving as the principal investigator with research grants from Vertex, Bayer, and Novartis to Brigham and Women’s Hospital for unrelated projects.
FROM THE JOURNAL OF THE AMERICAN HEART ASSOCIATION
In-hospital mobility impairment in older MI patients predicts postdischarge functional decline
Background: The ability to independently perform daily activities is highly valued by patients, yet it is commonly impaired in older adults after hospitalization for MI. Risk of functional decline in this population is not well understood, but may relate to reduced mobility while hospitalized.
Study design: Prospective cohort.
Setting: A total of 94 academic and community hospitals in the United States.
Synopsis: More than 3,000 adults aged 75 years and older who were hospitalized for acute myocardial infarction were enrolled in the prospective cohort SILVER-AMI; 2,587 patients within this cohort were evaluated for in-hospital mobility with the Timed “Up and Go” test. At 6-month follow-up, loss of independent performance of activities of daily living (ADL) and of the ability to walk 0.25 miles were both associated in a dose-dependent manner with in-hospital mobility. Severe in-hospital mobility impairment was associated with ADL decline with an adjusted odds ratio of 5.45 (95% confidence interval, 3.29-9.01).
While in-hospital mobility is predictive of future functional decline in this population, this observational study cannot establish whether attempts to improve mobility in hospitalized patients will prevent future functional decline.
Bottom line: Lower performance on the Timed “Up and Go” test of mobility among older patients hospitalized for MI is associated with functional decline 6 months after hospitalization.
Citation: Hajduk AM et al. Association between mobility measured during hospitalization and functional outcomes in older adults with acute myocardial infarction in the SILVER-AMI study. JAMA Intern Med. 2019 Oct 7. doi: 10.1001/jamainternmed.2019.4114.
Dr. Gerstenberger is a hospitalist and clinical assistant professor of medicine at the University of Utah, Salt Lake City.
Background: The ability to independently perform daily activities is highly valued by patients, yet it is commonly impaired in older adults after hospitalization for MI. Risk of functional decline in this population is not well understood, but may relate to reduced mobility while hospitalized.
Study design: Prospective cohort.
Setting: A total of 94 academic and community hospitals in the United States.
Synopsis: More than 3,000 adults aged 75 years and older who were hospitalized for acute myocardial infarction were enrolled in the prospective cohort SILVER-AMI; 2,587 patients within this cohort were evaluated for in-hospital mobility with the Timed “Up and Go” test. At 6-month follow-up, loss of independent performance of activities of daily living (ADL) and of the ability to walk 0.25 miles were both associated in a dose-dependent manner with in-hospital mobility. Severe in-hospital mobility impairment was associated with ADL decline with an adjusted odds ratio of 5.45 (95% confidence interval, 3.29-9.01).
While in-hospital mobility is predictive of future functional decline in this population, this observational study cannot establish whether attempts to improve mobility in hospitalized patients will prevent future functional decline.
Bottom line: Lower performance on the Timed “Up and Go” test of mobility among older patients hospitalized for MI is associated with functional decline 6 months after hospitalization.
Citation: Hajduk AM et al. Association between mobility measured during hospitalization and functional outcomes in older adults with acute myocardial infarction in the SILVER-AMI study. JAMA Intern Med. 2019 Oct 7. doi: 10.1001/jamainternmed.2019.4114.
Dr. Gerstenberger is a hospitalist and clinical assistant professor of medicine at the University of Utah, Salt Lake City.
Background: The ability to independently perform daily activities is highly valued by patients, yet it is commonly impaired in older adults after hospitalization for MI. Risk of functional decline in this population is not well understood, but may relate to reduced mobility while hospitalized.
Study design: Prospective cohort.
Setting: A total of 94 academic and community hospitals in the United States.
Synopsis: More than 3,000 adults aged 75 years and older who were hospitalized for acute myocardial infarction were enrolled in the prospective cohort SILVER-AMI; 2,587 patients within this cohort were evaluated for in-hospital mobility with the Timed “Up and Go” test. At 6-month follow-up, loss of independent performance of activities of daily living (ADL) and of the ability to walk 0.25 miles were both associated in a dose-dependent manner with in-hospital mobility. Severe in-hospital mobility impairment was associated with ADL decline with an adjusted odds ratio of 5.45 (95% confidence interval, 3.29-9.01).
While in-hospital mobility is predictive of future functional decline in this population, this observational study cannot establish whether attempts to improve mobility in hospitalized patients will prevent future functional decline.
Bottom line: Lower performance on the Timed “Up and Go” test of mobility among older patients hospitalized for MI is associated with functional decline 6 months after hospitalization.
Citation: Hajduk AM et al. Association between mobility measured during hospitalization and functional outcomes in older adults with acute myocardial infarction in the SILVER-AMI study. JAMA Intern Med. 2019 Oct 7. doi: 10.1001/jamainternmed.2019.4114.
Dr. Gerstenberger is a hospitalist and clinical assistant professor of medicine at the University of Utah, Salt Lake City.
Arcalyst gets FDA nod as first therapy for recurrent pericarditis
The Food and Drug Administration has approved rilonacept (Arcalyst) to treat recurrent pericarditis and reduce the risk for recurrence in adults and children 12 years and older.
Approval of the weekly subcutaneous injection offers patients the first and only FDA-approved therapy for recurrent pericarditis, the agency said in a release.
Recurrent pericarditis is characterized by a remitting relapsing inflammation of the pericardium, and therapeutic options have been limited to NSAIDs, colchicine, and corticosteroids.
Rilonacept is a recombinant fusion protein that blocks interleukin-1 alpha and interleukin-1 beta signaling. It is already approved by the FDA to treat a group of rare inherited inflammatory diseases called cryopyrin-associated periodic syndromes.
The new indication is based on the pivotal phase 3 RHAPSODY trial in 86 patients with acute symptoms of recurrent pericarditis and systemic inflammation. After randomization, pericarditis recurred in 2 of 30 patients (7%) treated with rilonacept and in 23 of 31 patients (74%) treated with placebo, representing a 96% reduction in the relative risk for recurrence with rilonacept.
Patients who received rilonacept were also pain free or had minimal pain on 98% of trial days, whereas those who received placebo had minimal or no pain on 46% of trial days.
The most common adverse effects of rilonacept are injection-site reactions and upper-respiratory tract infections.
Serious, life-threatening infections have been reported in patients taking rilonacept, according to the FDA. Patients with active or chronic infections should not take the drug.
The FDA label also advises that patients should avoid live vaccines while taking rilonacept and that it should be discontinued if a hypersensitivity reaction occurs.
The commercial launch is expected in April, according to the company.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved rilonacept (Arcalyst) to treat recurrent pericarditis and reduce the risk for recurrence in adults and children 12 years and older.
Approval of the weekly subcutaneous injection offers patients the first and only FDA-approved therapy for recurrent pericarditis, the agency said in a release.
Recurrent pericarditis is characterized by a remitting relapsing inflammation of the pericardium, and therapeutic options have been limited to NSAIDs, colchicine, and corticosteroids.
Rilonacept is a recombinant fusion protein that blocks interleukin-1 alpha and interleukin-1 beta signaling. It is already approved by the FDA to treat a group of rare inherited inflammatory diseases called cryopyrin-associated periodic syndromes.
The new indication is based on the pivotal phase 3 RHAPSODY trial in 86 patients with acute symptoms of recurrent pericarditis and systemic inflammation. After randomization, pericarditis recurred in 2 of 30 patients (7%) treated with rilonacept and in 23 of 31 patients (74%) treated with placebo, representing a 96% reduction in the relative risk for recurrence with rilonacept.
Patients who received rilonacept were also pain free or had minimal pain on 98% of trial days, whereas those who received placebo had minimal or no pain on 46% of trial days.
The most common adverse effects of rilonacept are injection-site reactions and upper-respiratory tract infections.
Serious, life-threatening infections have been reported in patients taking rilonacept, according to the FDA. Patients with active or chronic infections should not take the drug.
The FDA label also advises that patients should avoid live vaccines while taking rilonacept and that it should be discontinued if a hypersensitivity reaction occurs.
The commercial launch is expected in April, according to the company.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved rilonacept (Arcalyst) to treat recurrent pericarditis and reduce the risk for recurrence in adults and children 12 years and older.
Approval of the weekly subcutaneous injection offers patients the first and only FDA-approved therapy for recurrent pericarditis, the agency said in a release.
Recurrent pericarditis is characterized by a remitting relapsing inflammation of the pericardium, and therapeutic options have been limited to NSAIDs, colchicine, and corticosteroids.
Rilonacept is a recombinant fusion protein that blocks interleukin-1 alpha and interleukin-1 beta signaling. It is already approved by the FDA to treat a group of rare inherited inflammatory diseases called cryopyrin-associated periodic syndromes.
The new indication is based on the pivotal phase 3 RHAPSODY trial in 86 patients with acute symptoms of recurrent pericarditis and systemic inflammation. After randomization, pericarditis recurred in 2 of 30 patients (7%) treated with rilonacept and in 23 of 31 patients (74%) treated with placebo, representing a 96% reduction in the relative risk for recurrence with rilonacept.
Patients who received rilonacept were also pain free or had minimal pain on 98% of trial days, whereas those who received placebo had minimal or no pain on 46% of trial days.
The most common adverse effects of rilonacept are injection-site reactions and upper-respiratory tract infections.
Serious, life-threatening infections have been reported in patients taking rilonacept, according to the FDA. Patients with active or chronic infections should not take the drug.
The FDA label also advises that patients should avoid live vaccines while taking rilonacept and that it should be discontinued if a hypersensitivity reaction occurs.
The commercial launch is expected in April, according to the company.
A version of this article first appeared on Medscape.com.
Implementing the AMI READMITS Risk Assessment Score to Increase Referrals Among Patients With Type I Myocardial Infarction
From The Johns Hopkins Hospital, Baltimore, MD (Dr. Muganlinskaya and Dr. Skojec, retired); The George Washington University, Washington, DC (Dr. Posey); and Johns Hopkins University, Baltimore, MD (Dr. Resar).
Abstract
Objective: Assessing the risk characteristics of patients with acute myocardial infarction (MI) can help providers make appropriate referral decisions. This quality improvement project sought to improve timely, appropriate referrals among patients with type I MI by adding a risk assessment, the AMI READMITS score, to the existing referral protocol.
Methods: Patients’ chart data were analyzed to assess changes in referrals and timely follow-up appointments from pre-intervention to intervention. A survey assessed providers’ satisfaction with the new referral protocol.
Results: Among 57 patients (n = 29 preintervention; n = 28 intervention), documented referrals increased significantly from 66% to 89% (χ2 = 4.571, df = 1, P = 0.033); and timely appointments increased by 10%, which was not significant (χ2 = 3.550, df = 2, P = 0.169). Most providers agreed that the new protocol was easy to use, useful in making referral decisions, and improved the referral process. All agreed the risk score should be incorporated into electronic clinical notes. Provider opinions related to implementing the risk score in clinical practice were mixed. Qualitative feedback suggests this was due to limited validation of the AMI READMITS score in reducing readmissions.
Conclusions: Our risk-based referral protocol helped to increase appropriate referrals among patients with type I MI. Provider adoption may be enhanced by incorporating the protocol into electronic clinical notes. Research to further validate the accuracy of the AMI READMITS score in predicting readmissions may support adoption of the protocol in clinical practice.
Keywords: quality improvement; type I myocardial infarction; referral process; readmission risk; risk assessment; chart review.
Early follow-up after discharge is an important strategy to reduce the risk of unplanned hospital readmissions among patients with various conditions.1-3 While patient confounding factors, such as chronic health problems, environment, socioeconomic status, and literacy, make it difficult to avoid all unplanned readmissions, early follow-up may help providers identify and appropriately manage some health-related issues, and as such is a pivotal element of a readmission prevention strategy.4 There is evidence that patients with non-ST elevation myocardial infarction (NSTEMI) who have an outpatient appointment with a physician within 7 days after discharge have a lower risk of 30-day readmission.5
Our hospital’s postmyocardial infarction clinic was created to prevent unplanned readmissions within 30 days after discharge among patients with type I myocardial infarction (MI). Since inception, the number of referrals has been much lower than expected. In 2018, the total number of patients discharged from the hospital with type I MI and any troponin I level above 0.40 ng/mL was 313. Most of these patients were discharged from the hospital’s cardiac units; however, only 91 referrals were made. To increase referrals, the cardiology nurse practitioners (NPs) developed a post-MI referral protocol (Figure 1). However, this protocol was not consistently used and referrals to the clinic remained low.
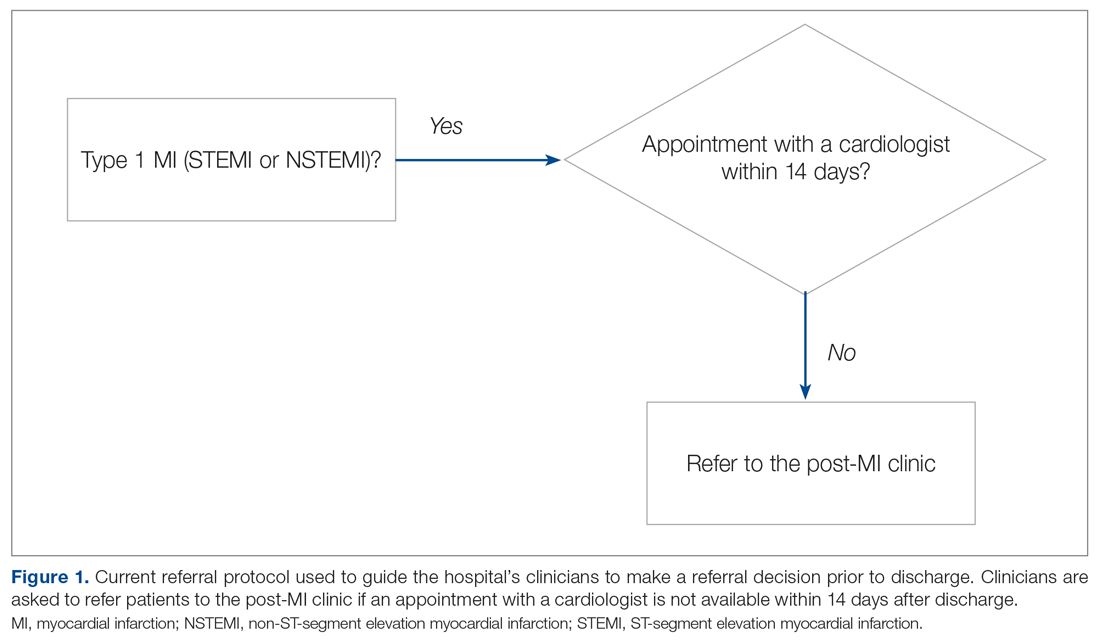
Evidence-based risk assessment tools have the potential to increase effective patient management. For example, cardiology providers at the hospital utilize various scores, such as CHA2DS2-VASc6 and the Society of Thoracic Surgery risk score,7 to plan patient management. Among the scores used to predict unplanned readmissions for MI patients, the most promising is the AMI READMITS score.8 Unlike other nonspecific prediction models, the AMI READMITS score was developed based on variables extracted from the electronic health records (EHRs) of patients who were hospitalized for MI and readmitted within 30 days after discharge. Recognizing the potential to increase referrals by integrating an MI-specific risk assessment, this quality improvement study modified the existing referral protocol to include the patients’ AMI READMITS score and recommendations for follow-up.
Currently, there are no clear recommendations on how soon after discharge patients with MI should undergo follow-up. As research data vary, we selected 7 days follow-up for patients from high risk groups based on the “See you in 7” initiative for patients with heart failure (HF) and MI,9,10 as well as evidence that patients with NSTEMI have a lower risk of 30-day readmission if they have follow-up within 7 days after discharge5; and we selected 14 days follow-up for patients from low-risk groups based on evidence that postdischarge follow-up within 14 days reduces risk of 30-day readmission in patients with acute myocardial infarction (AMI) and/or acutely decompensated HF.11
Methods
This project was designed to answer the following question: For adult patients with type I MI, does implementation of a readmission risk assessment referral protocol increase the percentage of referrals and appointments scheduled within a recommended time? Anticipated outcomes included: (1) increased referrals to a cardiologist or the post-MI clinic; (2) increased scheduled follow-up appointments within 7 to 14 days; (3) provider satisfaction with the usability and usefulness of the new protocol; and (4) consistent provider adoption of the new risk assessment referral protocol.
To evaluate the degree to which these outcomes were achieved, we reviewed patient charts for 2 months prior and 2 months during implementation of the new referral protocol. As shown in Figure 2, the new protocol added the following process steps to the existing protocol: calculation of the AMI READMITS score, recommendations for follow-up based on patients’ risk score, and guidance to refer patients to the post-MI clinic if patients did not have an appointment with a cardiologist within 7 to 14 days after discharge. Patients’ risk assessment scores were obtained from forms completed by clinicians during the intervention. Clinician’s perceptions related to the usability and usefulness of the new protocol and feedback related to its long-term adoption were assessed using a descriptive survey.
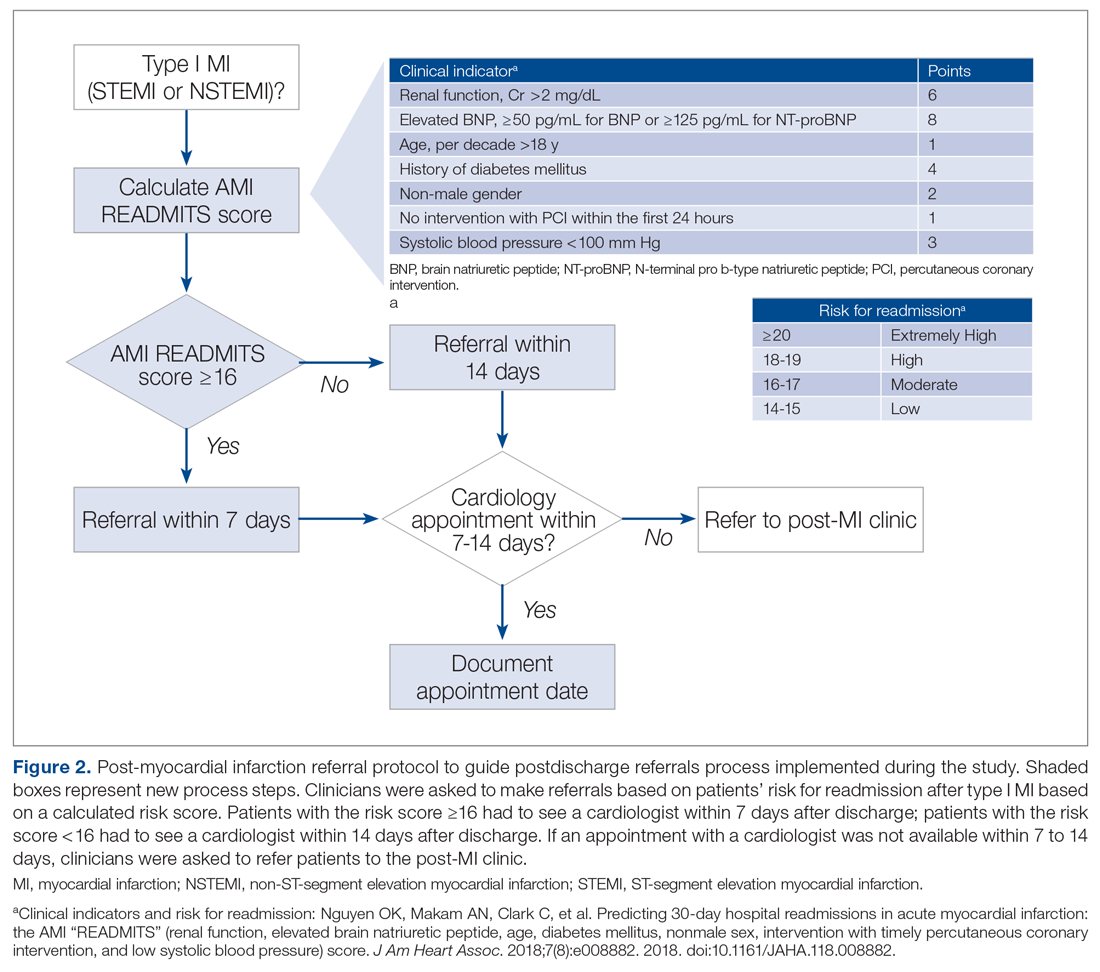
The institutional review board classified this project as a quality improvement project. To avoid potential loss of patient privacy, no identifiable data were collected, a unique identifier unrelated to patients’ records was generated for each patient, and data were saved on a password-protected cardiology office computer.
Population
The project population included all adult patients (≥ 18 years old) with type I MI who were admitted or transferred to the hospital, had a percutaneous coronary intervention (PCI), or were managed without PCI and discharged from the hospital’s cardiac care unit (CCU) and progressive cardiac care unit (PCCU). The criteria for type I MI included the “detection of a rise and/or fall of cardiac troponin with at least 1 value above the 99th percentile and with at least 1 of the following: symptoms of acute myocardial ischemia; new ischemic electrocardiographic (ECG) changes; development of new pathological Q waves; imaging evidence of new loss of viable myocardium or new regional wall motion abnormality in a pattern consistent with an ischemic etiology; identification of a coronary thrombus by angiography including intracoronary imaging or by autopsy.”12 The study excluded patients with type I MI who were referred for coronary bypass surgery.
Intervention
The revised risk assessment protocol was implemented within the CCU and PCCU. The lead investigator met with each provider to discuss the role of the post-MI clinic, current referral rates, the purpose of the project, and the new referral process to be completed during the project for each patient discharged with type I MI. Cardiology NPs, fellows, and residents were asked to use the risk-assessment form to calculate patients’ risk for readmission, and refer patients to the post-MI clinic if an appointment with a cardiologist was not available within 7 to 14 days after discharge. Every week during the intervention phase, the investigator sent reminder emails to ensure form completion. Providers were asked to calculate and write the score, the discharge and referral dates, where referrals were made (a cardiologist or the post-MI clinic), date of appointment, and reason for not scheduling an appointment or not referring on the risk assessment form, and to drop the completed forms in specific labeled boxes located at the CCU and PCCU work stations. The investigator collected the completed forms weekly. When the number of discharged patients did not match the number of completed forms, the investigator followed up with discharging providers to understand why.
Data and Data Collection
Data to determine whether the use of the new protocol increased discharge referrals among patients with type I MI within the recommended timeframes were collected by electronic chart review. Data included discharging unit, patients’ age, gender, admission and discharge date, diagnosis, referral to a cardiologist and the post-MI clinic, and appointment date. Clinical data needed to calculate the AMI READMITS score was also collected: PCI within 24 hours, serum creatinine, systolic blood pressure (SBP), brain natriuretic peptide (BNP), and diabetes status.
Data to assess provider satisfaction with the usability and usefulness of the new protocol were gathered through an online survey. The survey included 1 question related to the providers’ role, 1 question asking whether they used the risk assessment for each patient, and 5 Likert-items assessing the ease of usage. An additional open-ended question asked providers to share feedback related to integrating the AMI READMITS risk assessment score to the post-MI referral protocol long term.
To evaluate how consistently providers utilized the new referral protocol when discharging patients with type I MI, the number of completed forms was compared with the number of those patients who were discharged.
Statistical Analysis
Descriptive statistics were used to summarize patient demographics and to calculate the frequency of referrals before and during the intervention. Chi-square statistics were calculated to determine whether the change in percentage of referrals and timely referrals was significant. Descriptive statistics were used to determine the level of provider satisfaction related to each survey item. A content analysis method was used to synthesize themes from the open-ended question asking clinicians to share their feedback related to the new protocol.
Results
Fifty-seven patients met the study inclusion criteria: 29 patients during the preintervention phase and 28 patients during the intervention phase. There were 35 male (61.4%) and 22 female (38.6%) patients. Twenty-five patients (43.9%) were from age groups 41 through 60 years and 61 through 80 years, respectively, representing the majority of included patients. Seven patients (12.3%) were from the 81 years and older age group. There were no patients in the age group 18 through 40 years. Based on the AMI READMITS score calculation, 57.9% (n = 33) patients were from a low-risk group (includes extremely low and low risk for readmission) and 42.1% (n = 24) were from a high-risk group (includes moderate, high, and extremely high risk for readmission).
Provider adoption of the new protocol during the intervention was high. Referral forms were completed for 82% (n = 23) of the 28 patients during the intervention. Analysis findings showed a statistically significant increase in documented referrals after implementing the new referral protocol. During the preintervention phase, 66% (n = 19) of patients with type I MI were referred to see a cardiologist or an NP at a post-MI clinic and there was no documented referral for 34% (n = 10) of patients. During the intervention phase, 89% (n = 25) of patients were referred and there was no documented referral for 11% (n = 3) of patients. Chi-square results indicated that the increase in referrals was significant (χ2 = 4.571, df = 1, P = 0.033).
Data analysis examined whether patient referrals fell within the recommended timeframe of 7 days for the high-risk group (included moderate-to-extremely high risk) and 14 days for the low-risk group (included low-to-extremely low risk). During the preintervention phase, 31% (n = 9) of patient referrals were scheduled as recommended; 28% (n = 8) of patient referrals were scheduled but delayed; and there was no referral date documented for 41% (n = 12) of patients. During the intervention phase, referrals scheduled as recommended increased to 53% (n = 15); 25% (n = 7) of referrals were scheduled but delayed; and there was no referral date documented for 21.4% (n = 6) of patients. The change in appointments scheduled as recommended was not significant (χ2 = 3.550, df = 2, P = 0.169).
Surveys were emailed to 25 cardiology fellows and 3 cardiology NPs who participated in this study. Eighteen of the 28 clinicians (15 cardiology fellows and 3 cardiology NPs) responded for a response rate of 64%. One of several residents who rotated through the CCU and PCCU during the intervention also completed the survey, for a total of 19 participants. When asked if the protocol was easy to use, 79% agreed or strongly agreed. Eighteen of the 19 participants (95%) agreed or strongly agreed that the protocol was useful in making referral decisions. Sixty-eight percent agreed or strongly agreed that the AMI READMITS risk assessment score improves referral process. All participants agreed or strongly agreed that there should be an option to incorporate the AMI READMITS risk assessment score into electronic clinical notes. When asked whether the AMI READMITS risk score should be implemented in clinical practice, responses were mixed (Figure 3). A common theme among the 4 participants who responded with comments was the need for additional data to validate the usefulness of the AMI READMITS to reduce readmissions. In addition, 1 participant commented that “manual calculation [of the risk score] is not ideal.”
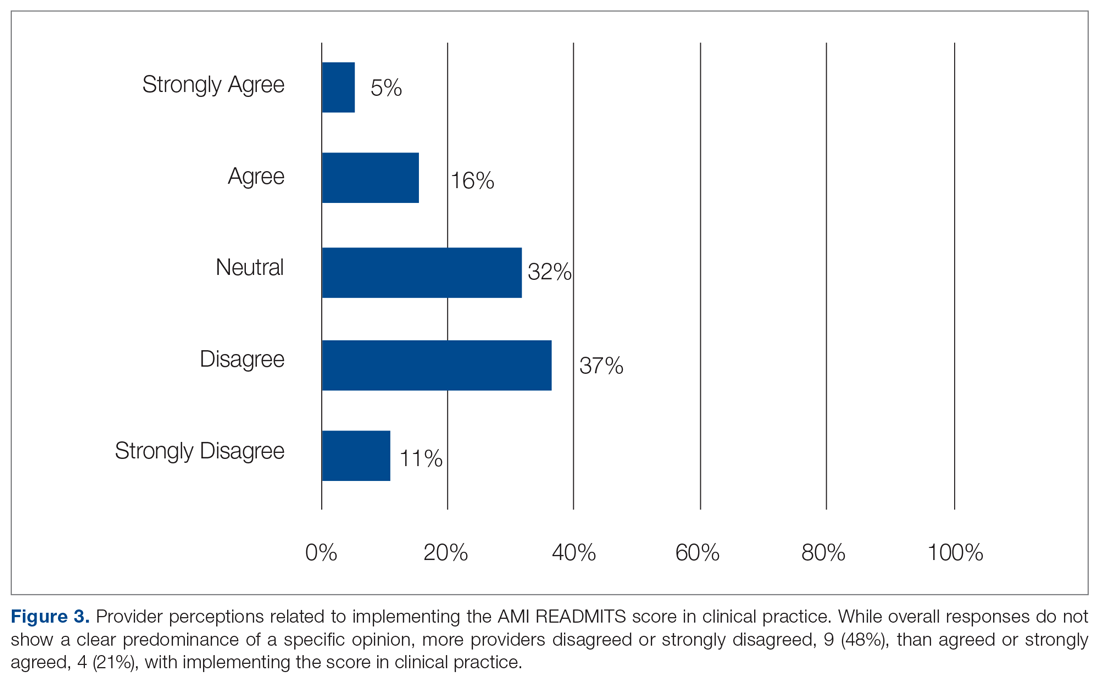
Discussion
This project demonstrated that implementing an evidence-based referral protocol integrating the AMI-READMITS score can increase timely postdischarge referrals among patients with type I MI. The percentage of appropriately scheduled appointments increased during the intervention phase; however, a relatively high number of appointments were scheduled outside of the recommended timeframe, similar to preintervention. Thus, while the new protocol increased referrals and provider documentation of these referrals, it appears that challenges in scheduling timely referral appointments remained. This project did not examine the reasons for delayed appointments.
The survey findings indicated that providers were generally satisfied with the usability and usefulness of the new risk assessment protocol. A large majority agreed or strongly agreed that it was easy to use and useful in making referral decisions, and most agreed or strongly agreed that it improves the referral process. Mixed opinions regarding implementing the AMI READMITS score in clinical practice, combined with qualitative findings, suggest that a lack of external validation of the AMI READMITS presents a barrier to its long-term adoption. All providers who participated in the survey agreed or strongly agreed that the risk assessment should be incorporated into electronic clinical notes. We have begun the process of working with the EHR vendor to automate the AMI risk-assessment within the referral work-flow, which will provide an opportunity for a follow-up quality improvement study.
This quality improvement project has several limitations. First, it implemented a small change in 2 inpatient units at 1 hospital using a simple pre- posttest design. Therefore, the findings are not generalizable to other settings. Prior to the intervention, some referrals may have been made without documentation. While the authors were able to trace undocumented referrals for patients who were referred to the post-MI clinic or to a cardiologist affiliated with the hospital, some patients may have been referred to cardiologists who were not affiliated with the hospital. Another limitation was that the self-created provider survey used was not tested in other clinical settings; thus, it cannot be determined whether the sensitivity and specificity of the survey questions are high. In addition, the clinical providers who participated in the study knew the study team, which may have influenced their behavior during the study period. Furthermore, the identified improvement in clinicians’ referral practices may not be sustainable due to the complexity and effort required to manually calculate the risk score. This limitation could be eliminated by integrating the risk score calculation into the EHR.
Conclusion
Early follow-up after discharge plays an important role in supporting patients’ self-management of some risk factors (ie, diet, weight, and smoking) and identifying gaps in postdischarge care which may lead to readmission. This project provides evidence that integrating the AMI READMITS risk assessment score into the referral process can help to guide discharge decision-making and increase timely, appropriate referrals for patients with MI. Integration of a specific risk assessment, such as the AMI READMITS, within the post-MI referral protocol may help clinicians make more efficient, educated referral decisions. Future studies should explore more specifically how and why the new protocol impacts clinicians’ decision-making and behavior related to post-MI referrals. In addition, future studies should investigate challenges associated with scheduling postdischarge appointments. It will be important to investigate how integration of the new protocol within the EHR may increase efficiency, consistency, and provider satisfaction with the new referral process. Additional research investigating the effects of the AMI READMITS score on readmissions reduction will be important to promote long-term adoption of the improved referral protocol in clinical practice.
Acknowledgments: The authors thank Shelly Conaway, ANP-BC, MSN, Angela Street, ANP-BC, MSN, Andrew Geis, ACNP-BC, MSN, Richard P. Jones II, MD, Eunice Young, MD, Joy Rothwell, MSN, RN-BC, Allison Olazo, MBA, MSN, RN-BC, Elizabeth Heck, RN-BC, and Matthew Trojanowski, MHA, MS, RRT, CSSBB for their support of this study.
Corresponding author: Nailya Muganlinskaya, DNP, MPH, ACNP-BC, MSN, The Johns Hopkins Hospital, 1800 Orleans St, Baltimore, MD 21287; nmuganl1@jhmi.edu.
Financial disclosures: None.
1. Why it is important to improve care transitions? Society of Hospital Medicine. Accessed June 15, 2020. https://www.hospitalmedicine.org/clinical-topics/care-transitions/
2. Tong L, Arnold T, Yang J, et al. The association between outpatient follow-up visits and all-cause non-elective 30-day readmissions: a retrospective observational cohort study. PloS One. 2018;13(7):e0200691.
3. Jackson C, Shahsahebi M, Wedlake T, DuBard CA. Timeliness of outpatient follow-up: an evidence-based approach for planning after hospital discharge. Ann Fam Med. 2015;13(2):115-22.
4. Health Research & Educational Trust. Preventable Readmissions Change Package. American Hospital Association. Updated December 2015. Accessed June 10, 2020. https://www.aha.org/sites/default/files/hiin/HRETHEN_ChangePackage_Readmissions.pd
5. Tung Y-C, Chang G-M, Chang H-Y, Yu T-H. Relationship between early physician follow-up and 30-day readmission after acute myocardial infarction and heart failure. Plos One. 2017;12(1):e0170061.
6. Kaplan RM, Koehler J, Zieger PD, et al. Stroke risk as a function of atrial fibrillation duration and CHA2DS2-VASc score. Circulation. 2019;140(20):1639-46.
7. Balan P, Zhao Y, Johnson S, et al. The Society of Thoracic Surgery Risk Score as a predictor of 30-day mortality in transcatheter vs surgical aortic valve replacement: a single-center experience and its implications for the development of a TAVR risk-prediction model. J Invasive Cardiol. 2017;29(3):109-14.
8. Smith LN, Makam AN, Darden D, et al. Acute myocardial infarction readmission risk prediction models: A systematic review of model performance. Circ Cardiovasc Qual Outcomes9.9. 2018;11(1):e003885.
9. Baker H, Oliver-McNeil S, Deng L, Hummel SL. See you in 7: regional hospital collaboration and outcomes in Medicare heart failure patients. JACC Heart Fail. 2015;3(10):765-73.
10. Batten A, Jaeger C, Griffen D, et al. See you in 7: improving acute myocardial infarction follow-up care. BMJ Open Qual. 2018;7(2):e000296.
11. Lee DW, Armistead L, Coleman H, et al. Abstract 15387: Post-discharge follow-up within 14 days reduces 30-day hospital readmission rates in patients with acute myocardial infarction and/or acutely decompensated heart failure. Circulation. 2018;134 (1):A 15387.
12. Thygesen K, Alpert JS, Jaffe AS, et al. Fourth universal definition of myocardial infarction. Circulation. 2018;138 (20):e:618-51.
From The Johns Hopkins Hospital, Baltimore, MD (Dr. Muganlinskaya and Dr. Skojec, retired); The George Washington University, Washington, DC (Dr. Posey); and Johns Hopkins University, Baltimore, MD (Dr. Resar).
Abstract
Objective: Assessing the risk characteristics of patients with acute myocardial infarction (MI) can help providers make appropriate referral decisions. This quality improvement project sought to improve timely, appropriate referrals among patients with type I MI by adding a risk assessment, the AMI READMITS score, to the existing referral protocol.
Methods: Patients’ chart data were analyzed to assess changes in referrals and timely follow-up appointments from pre-intervention to intervention. A survey assessed providers’ satisfaction with the new referral protocol.
Results: Among 57 patients (n = 29 preintervention; n = 28 intervention), documented referrals increased significantly from 66% to 89% (χ2 = 4.571, df = 1, P = 0.033); and timely appointments increased by 10%, which was not significant (χ2 = 3.550, df = 2, P = 0.169). Most providers agreed that the new protocol was easy to use, useful in making referral decisions, and improved the referral process. All agreed the risk score should be incorporated into electronic clinical notes. Provider opinions related to implementing the risk score in clinical practice were mixed. Qualitative feedback suggests this was due to limited validation of the AMI READMITS score in reducing readmissions.
Conclusions: Our risk-based referral protocol helped to increase appropriate referrals among patients with type I MI. Provider adoption may be enhanced by incorporating the protocol into electronic clinical notes. Research to further validate the accuracy of the AMI READMITS score in predicting readmissions may support adoption of the protocol in clinical practice.
Keywords: quality improvement; type I myocardial infarction; referral process; readmission risk; risk assessment; chart review.
Early follow-up after discharge is an important strategy to reduce the risk of unplanned hospital readmissions among patients with various conditions.1-3 While patient confounding factors, such as chronic health problems, environment, socioeconomic status, and literacy, make it difficult to avoid all unplanned readmissions, early follow-up may help providers identify and appropriately manage some health-related issues, and as such is a pivotal element of a readmission prevention strategy.4 There is evidence that patients with non-ST elevation myocardial infarction (NSTEMI) who have an outpatient appointment with a physician within 7 days after discharge have a lower risk of 30-day readmission.5
Our hospital’s postmyocardial infarction clinic was created to prevent unplanned readmissions within 30 days after discharge among patients with type I myocardial infarction (MI). Since inception, the number of referrals has been much lower than expected. In 2018, the total number of patients discharged from the hospital with type I MI and any troponin I level above 0.40 ng/mL was 313. Most of these patients were discharged from the hospital’s cardiac units; however, only 91 referrals were made. To increase referrals, the cardiology nurse practitioners (NPs) developed a post-MI referral protocol (Figure 1). However, this protocol was not consistently used and referrals to the clinic remained low.

Evidence-based risk assessment tools have the potential to increase effective patient management. For example, cardiology providers at the hospital utilize various scores, such as CHA2DS2-VASc6 and the Society of Thoracic Surgery risk score,7 to plan patient management. Among the scores used to predict unplanned readmissions for MI patients, the most promising is the AMI READMITS score.8 Unlike other nonspecific prediction models, the AMI READMITS score was developed based on variables extracted from the electronic health records (EHRs) of patients who were hospitalized for MI and readmitted within 30 days after discharge. Recognizing the potential to increase referrals by integrating an MI-specific risk assessment, this quality improvement study modified the existing referral protocol to include the patients’ AMI READMITS score and recommendations for follow-up.
Currently, there are no clear recommendations on how soon after discharge patients with MI should undergo follow-up. As research data vary, we selected 7 days follow-up for patients from high risk groups based on the “See you in 7” initiative for patients with heart failure (HF) and MI,9,10 as well as evidence that patients with NSTEMI have a lower risk of 30-day readmission if they have follow-up within 7 days after discharge5; and we selected 14 days follow-up for patients from low-risk groups based on evidence that postdischarge follow-up within 14 days reduces risk of 30-day readmission in patients with acute myocardial infarction (AMI) and/or acutely decompensated HF.11
Methods
This project was designed to answer the following question: For adult patients with type I MI, does implementation of a readmission risk assessment referral protocol increase the percentage of referrals and appointments scheduled within a recommended time? Anticipated outcomes included: (1) increased referrals to a cardiologist or the post-MI clinic; (2) increased scheduled follow-up appointments within 7 to 14 days; (3) provider satisfaction with the usability and usefulness of the new protocol; and (4) consistent provider adoption of the new risk assessment referral protocol.
To evaluate the degree to which these outcomes were achieved, we reviewed patient charts for 2 months prior and 2 months during implementation of the new referral protocol. As shown in Figure 2, the new protocol added the following process steps to the existing protocol: calculation of the AMI READMITS score, recommendations for follow-up based on patients’ risk score, and guidance to refer patients to the post-MI clinic if patients did not have an appointment with a cardiologist within 7 to 14 days after discharge. Patients’ risk assessment scores were obtained from forms completed by clinicians during the intervention. Clinician’s perceptions related to the usability and usefulness of the new protocol and feedback related to its long-term adoption were assessed using a descriptive survey.

The institutional review board classified this project as a quality improvement project. To avoid potential loss of patient privacy, no identifiable data were collected, a unique identifier unrelated to patients’ records was generated for each patient, and data were saved on a password-protected cardiology office computer.
Population
The project population included all adult patients (≥ 18 years old) with type I MI who were admitted or transferred to the hospital, had a percutaneous coronary intervention (PCI), or were managed without PCI and discharged from the hospital’s cardiac care unit (CCU) and progressive cardiac care unit (PCCU). The criteria for type I MI included the “detection of a rise and/or fall of cardiac troponin with at least 1 value above the 99th percentile and with at least 1 of the following: symptoms of acute myocardial ischemia; new ischemic electrocardiographic (ECG) changes; development of new pathological Q waves; imaging evidence of new loss of viable myocardium or new regional wall motion abnormality in a pattern consistent with an ischemic etiology; identification of a coronary thrombus by angiography including intracoronary imaging or by autopsy.”12 The study excluded patients with type I MI who were referred for coronary bypass surgery.
Intervention
The revised risk assessment protocol was implemented within the CCU and PCCU. The lead investigator met with each provider to discuss the role of the post-MI clinic, current referral rates, the purpose of the project, and the new referral process to be completed during the project for each patient discharged with type I MI. Cardiology NPs, fellows, and residents were asked to use the risk-assessment form to calculate patients’ risk for readmission, and refer patients to the post-MI clinic if an appointment with a cardiologist was not available within 7 to 14 days after discharge. Every week during the intervention phase, the investigator sent reminder emails to ensure form completion. Providers were asked to calculate and write the score, the discharge and referral dates, where referrals were made (a cardiologist or the post-MI clinic), date of appointment, and reason for not scheduling an appointment or not referring on the risk assessment form, and to drop the completed forms in specific labeled boxes located at the CCU and PCCU work stations. The investigator collected the completed forms weekly. When the number of discharged patients did not match the number of completed forms, the investigator followed up with discharging providers to understand why.
Data and Data Collection
Data to determine whether the use of the new protocol increased discharge referrals among patients with type I MI within the recommended timeframes were collected by electronic chart review. Data included discharging unit, patients’ age, gender, admission and discharge date, diagnosis, referral to a cardiologist and the post-MI clinic, and appointment date. Clinical data needed to calculate the AMI READMITS score was also collected: PCI within 24 hours, serum creatinine, systolic blood pressure (SBP), brain natriuretic peptide (BNP), and diabetes status.
Data to assess provider satisfaction with the usability and usefulness of the new protocol were gathered through an online survey. The survey included 1 question related to the providers’ role, 1 question asking whether they used the risk assessment for each patient, and 5 Likert-items assessing the ease of usage. An additional open-ended question asked providers to share feedback related to integrating the AMI READMITS risk assessment score to the post-MI referral protocol long term.
To evaluate how consistently providers utilized the new referral protocol when discharging patients with type I MI, the number of completed forms was compared with the number of those patients who were discharged.
Statistical Analysis
Descriptive statistics were used to summarize patient demographics and to calculate the frequency of referrals before and during the intervention. Chi-square statistics were calculated to determine whether the change in percentage of referrals and timely referrals was significant. Descriptive statistics were used to determine the level of provider satisfaction related to each survey item. A content analysis method was used to synthesize themes from the open-ended question asking clinicians to share their feedback related to the new protocol.
Results
Fifty-seven patients met the study inclusion criteria: 29 patients during the preintervention phase and 28 patients during the intervention phase. There were 35 male (61.4%) and 22 female (38.6%) patients. Twenty-five patients (43.9%) were from age groups 41 through 60 years and 61 through 80 years, respectively, representing the majority of included patients. Seven patients (12.3%) were from the 81 years and older age group. There were no patients in the age group 18 through 40 years. Based on the AMI READMITS score calculation, 57.9% (n = 33) patients were from a low-risk group (includes extremely low and low risk for readmission) and 42.1% (n = 24) were from a high-risk group (includes moderate, high, and extremely high risk for readmission).
Provider adoption of the new protocol during the intervention was high. Referral forms were completed for 82% (n = 23) of the 28 patients during the intervention. Analysis findings showed a statistically significant increase in documented referrals after implementing the new referral protocol. During the preintervention phase, 66% (n = 19) of patients with type I MI were referred to see a cardiologist or an NP at a post-MI clinic and there was no documented referral for 34% (n = 10) of patients. During the intervention phase, 89% (n = 25) of patients were referred and there was no documented referral for 11% (n = 3) of patients. Chi-square results indicated that the increase in referrals was significant (χ2 = 4.571, df = 1, P = 0.033).
Data analysis examined whether patient referrals fell within the recommended timeframe of 7 days for the high-risk group (included moderate-to-extremely high risk) and 14 days for the low-risk group (included low-to-extremely low risk). During the preintervention phase, 31% (n = 9) of patient referrals were scheduled as recommended; 28% (n = 8) of patient referrals were scheduled but delayed; and there was no referral date documented for 41% (n = 12) of patients. During the intervention phase, referrals scheduled as recommended increased to 53% (n = 15); 25% (n = 7) of referrals were scheduled but delayed; and there was no referral date documented for 21.4% (n = 6) of patients. The change in appointments scheduled as recommended was not significant (χ2 = 3.550, df = 2, P = 0.169).
Surveys were emailed to 25 cardiology fellows and 3 cardiology NPs who participated in this study. Eighteen of the 28 clinicians (15 cardiology fellows and 3 cardiology NPs) responded for a response rate of 64%. One of several residents who rotated through the CCU and PCCU during the intervention also completed the survey, for a total of 19 participants. When asked if the protocol was easy to use, 79% agreed or strongly agreed. Eighteen of the 19 participants (95%) agreed or strongly agreed that the protocol was useful in making referral decisions. Sixty-eight percent agreed or strongly agreed that the AMI READMITS risk assessment score improves referral process. All participants agreed or strongly agreed that there should be an option to incorporate the AMI READMITS risk assessment score into electronic clinical notes. When asked whether the AMI READMITS risk score should be implemented in clinical practice, responses were mixed (Figure 3). A common theme among the 4 participants who responded with comments was the need for additional data to validate the usefulness of the AMI READMITS to reduce readmissions. In addition, 1 participant commented that “manual calculation [of the risk score] is not ideal.”

Discussion
This project demonstrated that implementing an evidence-based referral protocol integrating the AMI-READMITS score can increase timely postdischarge referrals among patients with type I MI. The percentage of appropriately scheduled appointments increased during the intervention phase; however, a relatively high number of appointments were scheduled outside of the recommended timeframe, similar to preintervention. Thus, while the new protocol increased referrals and provider documentation of these referrals, it appears that challenges in scheduling timely referral appointments remained. This project did not examine the reasons for delayed appointments.
The survey findings indicated that providers were generally satisfied with the usability and usefulness of the new risk assessment protocol. A large majority agreed or strongly agreed that it was easy to use and useful in making referral decisions, and most agreed or strongly agreed that it improves the referral process. Mixed opinions regarding implementing the AMI READMITS score in clinical practice, combined with qualitative findings, suggest that a lack of external validation of the AMI READMITS presents a barrier to its long-term adoption. All providers who participated in the survey agreed or strongly agreed that the risk assessment should be incorporated into electronic clinical notes. We have begun the process of working with the EHR vendor to automate the AMI risk-assessment within the referral work-flow, which will provide an opportunity for a follow-up quality improvement study.
This quality improvement project has several limitations. First, it implemented a small change in 2 inpatient units at 1 hospital using a simple pre- posttest design. Therefore, the findings are not generalizable to other settings. Prior to the intervention, some referrals may have been made without documentation. While the authors were able to trace undocumented referrals for patients who were referred to the post-MI clinic or to a cardiologist affiliated with the hospital, some patients may have been referred to cardiologists who were not affiliated with the hospital. Another limitation was that the self-created provider survey used was not tested in other clinical settings; thus, it cannot be determined whether the sensitivity and specificity of the survey questions are high. In addition, the clinical providers who participated in the study knew the study team, which may have influenced their behavior during the study period. Furthermore, the identified improvement in clinicians’ referral practices may not be sustainable due to the complexity and effort required to manually calculate the risk score. This limitation could be eliminated by integrating the risk score calculation into the EHR.
Conclusion
Early follow-up after discharge plays an important role in supporting patients’ self-management of some risk factors (ie, diet, weight, and smoking) and identifying gaps in postdischarge care which may lead to readmission. This project provides evidence that integrating the AMI READMITS risk assessment score into the referral process can help to guide discharge decision-making and increase timely, appropriate referrals for patients with MI. Integration of a specific risk assessment, such as the AMI READMITS, within the post-MI referral protocol may help clinicians make more efficient, educated referral decisions. Future studies should explore more specifically how and why the new protocol impacts clinicians’ decision-making and behavior related to post-MI referrals. In addition, future studies should investigate challenges associated with scheduling postdischarge appointments. It will be important to investigate how integration of the new protocol within the EHR may increase efficiency, consistency, and provider satisfaction with the new referral process. Additional research investigating the effects of the AMI READMITS score on readmissions reduction will be important to promote long-term adoption of the improved referral protocol in clinical practice.
Acknowledgments: The authors thank Shelly Conaway, ANP-BC, MSN, Angela Street, ANP-BC, MSN, Andrew Geis, ACNP-BC, MSN, Richard P. Jones II, MD, Eunice Young, MD, Joy Rothwell, MSN, RN-BC, Allison Olazo, MBA, MSN, RN-BC, Elizabeth Heck, RN-BC, and Matthew Trojanowski, MHA, MS, RRT, CSSBB for their support of this study.
Corresponding author: Nailya Muganlinskaya, DNP, MPH, ACNP-BC, MSN, The Johns Hopkins Hospital, 1800 Orleans St, Baltimore, MD 21287; nmuganl1@jhmi.edu.
Financial disclosures: None.
From The Johns Hopkins Hospital, Baltimore, MD (Dr. Muganlinskaya and Dr. Skojec, retired); The George Washington University, Washington, DC (Dr. Posey); and Johns Hopkins University, Baltimore, MD (Dr. Resar).
Abstract
Objective: Assessing the risk characteristics of patients with acute myocardial infarction (MI) can help providers make appropriate referral decisions. This quality improvement project sought to improve timely, appropriate referrals among patients with type I MI by adding a risk assessment, the AMI READMITS score, to the existing referral protocol.
Methods: Patients’ chart data were analyzed to assess changes in referrals and timely follow-up appointments from pre-intervention to intervention. A survey assessed providers’ satisfaction with the new referral protocol.
Results: Among 57 patients (n = 29 preintervention; n = 28 intervention), documented referrals increased significantly from 66% to 89% (χ2 = 4.571, df = 1, P = 0.033); and timely appointments increased by 10%, which was not significant (χ2 = 3.550, df = 2, P = 0.169). Most providers agreed that the new protocol was easy to use, useful in making referral decisions, and improved the referral process. All agreed the risk score should be incorporated into electronic clinical notes. Provider opinions related to implementing the risk score in clinical practice were mixed. Qualitative feedback suggests this was due to limited validation of the AMI READMITS score in reducing readmissions.
Conclusions: Our risk-based referral protocol helped to increase appropriate referrals among patients with type I MI. Provider adoption may be enhanced by incorporating the protocol into electronic clinical notes. Research to further validate the accuracy of the AMI READMITS score in predicting readmissions may support adoption of the protocol in clinical practice.
Keywords: quality improvement; type I myocardial infarction; referral process; readmission risk; risk assessment; chart review.
Early follow-up after discharge is an important strategy to reduce the risk of unplanned hospital readmissions among patients with various conditions.1-3 While patient confounding factors, such as chronic health problems, environment, socioeconomic status, and literacy, make it difficult to avoid all unplanned readmissions, early follow-up may help providers identify and appropriately manage some health-related issues, and as such is a pivotal element of a readmission prevention strategy.4 There is evidence that patients with non-ST elevation myocardial infarction (NSTEMI) who have an outpatient appointment with a physician within 7 days after discharge have a lower risk of 30-day readmission.5
Our hospital’s postmyocardial infarction clinic was created to prevent unplanned readmissions within 30 days after discharge among patients with type I myocardial infarction (MI). Since inception, the number of referrals has been much lower than expected. In 2018, the total number of patients discharged from the hospital with type I MI and any troponin I level above 0.40 ng/mL was 313. Most of these patients were discharged from the hospital’s cardiac units; however, only 91 referrals were made. To increase referrals, the cardiology nurse practitioners (NPs) developed a post-MI referral protocol (Figure 1). However, this protocol was not consistently used and referrals to the clinic remained low.

Evidence-based risk assessment tools have the potential to increase effective patient management. For example, cardiology providers at the hospital utilize various scores, such as CHA2DS2-VASc6 and the Society of Thoracic Surgery risk score,7 to plan patient management. Among the scores used to predict unplanned readmissions for MI patients, the most promising is the AMI READMITS score.8 Unlike other nonspecific prediction models, the AMI READMITS score was developed based on variables extracted from the electronic health records (EHRs) of patients who were hospitalized for MI and readmitted within 30 days after discharge. Recognizing the potential to increase referrals by integrating an MI-specific risk assessment, this quality improvement study modified the existing referral protocol to include the patients’ AMI READMITS score and recommendations for follow-up.
Currently, there are no clear recommendations on how soon after discharge patients with MI should undergo follow-up. As research data vary, we selected 7 days follow-up for patients from high risk groups based on the “See you in 7” initiative for patients with heart failure (HF) and MI,9,10 as well as evidence that patients with NSTEMI have a lower risk of 30-day readmission if they have follow-up within 7 days after discharge5; and we selected 14 days follow-up for patients from low-risk groups based on evidence that postdischarge follow-up within 14 days reduces risk of 30-day readmission in patients with acute myocardial infarction (AMI) and/or acutely decompensated HF.11
Methods
This project was designed to answer the following question: For adult patients with type I MI, does implementation of a readmission risk assessment referral protocol increase the percentage of referrals and appointments scheduled within a recommended time? Anticipated outcomes included: (1) increased referrals to a cardiologist or the post-MI clinic; (2) increased scheduled follow-up appointments within 7 to 14 days; (3) provider satisfaction with the usability and usefulness of the new protocol; and (4) consistent provider adoption of the new risk assessment referral protocol.
To evaluate the degree to which these outcomes were achieved, we reviewed patient charts for 2 months prior and 2 months during implementation of the new referral protocol. As shown in Figure 2, the new protocol added the following process steps to the existing protocol: calculation of the AMI READMITS score, recommendations for follow-up based on patients’ risk score, and guidance to refer patients to the post-MI clinic if patients did not have an appointment with a cardiologist within 7 to 14 days after discharge. Patients’ risk assessment scores were obtained from forms completed by clinicians during the intervention. Clinician’s perceptions related to the usability and usefulness of the new protocol and feedback related to its long-term adoption were assessed using a descriptive survey.

The institutional review board classified this project as a quality improvement project. To avoid potential loss of patient privacy, no identifiable data were collected, a unique identifier unrelated to patients’ records was generated for each patient, and data were saved on a password-protected cardiology office computer.
Population
The project population included all adult patients (≥ 18 years old) with type I MI who were admitted or transferred to the hospital, had a percutaneous coronary intervention (PCI), or were managed without PCI and discharged from the hospital’s cardiac care unit (CCU) and progressive cardiac care unit (PCCU). The criteria for type I MI included the “detection of a rise and/or fall of cardiac troponin with at least 1 value above the 99th percentile and with at least 1 of the following: symptoms of acute myocardial ischemia; new ischemic electrocardiographic (ECG) changes; development of new pathological Q waves; imaging evidence of new loss of viable myocardium or new regional wall motion abnormality in a pattern consistent with an ischemic etiology; identification of a coronary thrombus by angiography including intracoronary imaging or by autopsy.”12 The study excluded patients with type I MI who were referred for coronary bypass surgery.
Intervention
The revised risk assessment protocol was implemented within the CCU and PCCU. The lead investigator met with each provider to discuss the role of the post-MI clinic, current referral rates, the purpose of the project, and the new referral process to be completed during the project for each patient discharged with type I MI. Cardiology NPs, fellows, and residents were asked to use the risk-assessment form to calculate patients’ risk for readmission, and refer patients to the post-MI clinic if an appointment with a cardiologist was not available within 7 to 14 days after discharge. Every week during the intervention phase, the investigator sent reminder emails to ensure form completion. Providers were asked to calculate and write the score, the discharge and referral dates, where referrals were made (a cardiologist or the post-MI clinic), date of appointment, and reason for not scheduling an appointment or not referring on the risk assessment form, and to drop the completed forms in specific labeled boxes located at the CCU and PCCU work stations. The investigator collected the completed forms weekly. When the number of discharged patients did not match the number of completed forms, the investigator followed up with discharging providers to understand why.
Data and Data Collection
Data to determine whether the use of the new protocol increased discharge referrals among patients with type I MI within the recommended timeframes were collected by electronic chart review. Data included discharging unit, patients’ age, gender, admission and discharge date, diagnosis, referral to a cardiologist and the post-MI clinic, and appointment date. Clinical data needed to calculate the AMI READMITS score was also collected: PCI within 24 hours, serum creatinine, systolic blood pressure (SBP), brain natriuretic peptide (BNP), and diabetes status.
Data to assess provider satisfaction with the usability and usefulness of the new protocol were gathered through an online survey. The survey included 1 question related to the providers’ role, 1 question asking whether they used the risk assessment for each patient, and 5 Likert-items assessing the ease of usage. An additional open-ended question asked providers to share feedback related to integrating the AMI READMITS risk assessment score to the post-MI referral protocol long term.
To evaluate how consistently providers utilized the new referral protocol when discharging patients with type I MI, the number of completed forms was compared with the number of those patients who were discharged.
Statistical Analysis
Descriptive statistics were used to summarize patient demographics and to calculate the frequency of referrals before and during the intervention. Chi-square statistics were calculated to determine whether the change in percentage of referrals and timely referrals was significant. Descriptive statistics were used to determine the level of provider satisfaction related to each survey item. A content analysis method was used to synthesize themes from the open-ended question asking clinicians to share their feedback related to the new protocol.
Results
Fifty-seven patients met the study inclusion criteria: 29 patients during the preintervention phase and 28 patients during the intervention phase. There were 35 male (61.4%) and 22 female (38.6%) patients. Twenty-five patients (43.9%) were from age groups 41 through 60 years and 61 through 80 years, respectively, representing the majority of included patients. Seven patients (12.3%) were from the 81 years and older age group. There were no patients in the age group 18 through 40 years. Based on the AMI READMITS score calculation, 57.9% (n = 33) patients were from a low-risk group (includes extremely low and low risk for readmission) and 42.1% (n = 24) were from a high-risk group (includes moderate, high, and extremely high risk for readmission).
Provider adoption of the new protocol during the intervention was high. Referral forms were completed for 82% (n = 23) of the 28 patients during the intervention. Analysis findings showed a statistically significant increase in documented referrals after implementing the new referral protocol. During the preintervention phase, 66% (n = 19) of patients with type I MI were referred to see a cardiologist or an NP at a post-MI clinic and there was no documented referral for 34% (n = 10) of patients. During the intervention phase, 89% (n = 25) of patients were referred and there was no documented referral for 11% (n = 3) of patients. Chi-square results indicated that the increase in referrals was significant (χ2 = 4.571, df = 1, P = 0.033).
Data analysis examined whether patient referrals fell within the recommended timeframe of 7 days for the high-risk group (included moderate-to-extremely high risk) and 14 days for the low-risk group (included low-to-extremely low risk). During the preintervention phase, 31% (n = 9) of patient referrals were scheduled as recommended; 28% (n = 8) of patient referrals were scheduled but delayed; and there was no referral date documented for 41% (n = 12) of patients. During the intervention phase, referrals scheduled as recommended increased to 53% (n = 15); 25% (n = 7) of referrals were scheduled but delayed; and there was no referral date documented for 21.4% (n = 6) of patients. The change in appointments scheduled as recommended was not significant (χ2 = 3.550, df = 2, P = 0.169).
Surveys were emailed to 25 cardiology fellows and 3 cardiology NPs who participated in this study. Eighteen of the 28 clinicians (15 cardiology fellows and 3 cardiology NPs) responded for a response rate of 64%. One of several residents who rotated through the CCU and PCCU during the intervention also completed the survey, for a total of 19 participants. When asked if the protocol was easy to use, 79% agreed or strongly agreed. Eighteen of the 19 participants (95%) agreed or strongly agreed that the protocol was useful in making referral decisions. Sixty-eight percent agreed or strongly agreed that the AMI READMITS risk assessment score improves referral process. All participants agreed or strongly agreed that there should be an option to incorporate the AMI READMITS risk assessment score into electronic clinical notes. When asked whether the AMI READMITS risk score should be implemented in clinical practice, responses were mixed (Figure 3). A common theme among the 4 participants who responded with comments was the need for additional data to validate the usefulness of the AMI READMITS to reduce readmissions. In addition, 1 participant commented that “manual calculation [of the risk score] is not ideal.”

Discussion
This project demonstrated that implementing an evidence-based referral protocol integrating the AMI-READMITS score can increase timely postdischarge referrals among patients with type I MI. The percentage of appropriately scheduled appointments increased during the intervention phase; however, a relatively high number of appointments were scheduled outside of the recommended timeframe, similar to preintervention. Thus, while the new protocol increased referrals and provider documentation of these referrals, it appears that challenges in scheduling timely referral appointments remained. This project did not examine the reasons for delayed appointments.
The survey findings indicated that providers were generally satisfied with the usability and usefulness of the new risk assessment protocol. A large majority agreed or strongly agreed that it was easy to use and useful in making referral decisions, and most agreed or strongly agreed that it improves the referral process. Mixed opinions regarding implementing the AMI READMITS score in clinical practice, combined with qualitative findings, suggest that a lack of external validation of the AMI READMITS presents a barrier to its long-term adoption. All providers who participated in the survey agreed or strongly agreed that the risk assessment should be incorporated into electronic clinical notes. We have begun the process of working with the EHR vendor to automate the AMI risk-assessment within the referral work-flow, which will provide an opportunity for a follow-up quality improvement study.
This quality improvement project has several limitations. First, it implemented a small change in 2 inpatient units at 1 hospital using a simple pre- posttest design. Therefore, the findings are not generalizable to other settings. Prior to the intervention, some referrals may have been made without documentation. While the authors were able to trace undocumented referrals for patients who were referred to the post-MI clinic or to a cardiologist affiliated with the hospital, some patients may have been referred to cardiologists who were not affiliated with the hospital. Another limitation was that the self-created provider survey used was not tested in other clinical settings; thus, it cannot be determined whether the sensitivity and specificity of the survey questions are high. In addition, the clinical providers who participated in the study knew the study team, which may have influenced their behavior during the study period. Furthermore, the identified improvement in clinicians’ referral practices may not be sustainable due to the complexity and effort required to manually calculate the risk score. This limitation could be eliminated by integrating the risk score calculation into the EHR.
Conclusion
Early follow-up after discharge plays an important role in supporting patients’ self-management of some risk factors (ie, diet, weight, and smoking) and identifying gaps in postdischarge care which may lead to readmission. This project provides evidence that integrating the AMI READMITS risk assessment score into the referral process can help to guide discharge decision-making and increase timely, appropriate referrals for patients with MI. Integration of a specific risk assessment, such as the AMI READMITS, within the post-MI referral protocol may help clinicians make more efficient, educated referral decisions. Future studies should explore more specifically how and why the new protocol impacts clinicians’ decision-making and behavior related to post-MI referrals. In addition, future studies should investigate challenges associated with scheduling postdischarge appointments. It will be important to investigate how integration of the new protocol within the EHR may increase efficiency, consistency, and provider satisfaction with the new referral process. Additional research investigating the effects of the AMI READMITS score on readmissions reduction will be important to promote long-term adoption of the improved referral protocol in clinical practice.
Acknowledgments: The authors thank Shelly Conaway, ANP-BC, MSN, Angela Street, ANP-BC, MSN, Andrew Geis, ACNP-BC, MSN, Richard P. Jones II, MD, Eunice Young, MD, Joy Rothwell, MSN, RN-BC, Allison Olazo, MBA, MSN, RN-BC, Elizabeth Heck, RN-BC, and Matthew Trojanowski, MHA, MS, RRT, CSSBB for their support of this study.
Corresponding author: Nailya Muganlinskaya, DNP, MPH, ACNP-BC, MSN, The Johns Hopkins Hospital, 1800 Orleans St, Baltimore, MD 21287; nmuganl1@jhmi.edu.
Financial disclosures: None.
1. Why it is important to improve care transitions? Society of Hospital Medicine. Accessed June 15, 2020. https://www.hospitalmedicine.org/clinical-topics/care-transitions/
2. Tong L, Arnold T, Yang J, et al. The association between outpatient follow-up visits and all-cause non-elective 30-day readmissions: a retrospective observational cohort study. PloS One. 2018;13(7):e0200691.
3. Jackson C, Shahsahebi M, Wedlake T, DuBard CA. Timeliness of outpatient follow-up: an evidence-based approach for planning after hospital discharge. Ann Fam Med. 2015;13(2):115-22.
4. Health Research & Educational Trust. Preventable Readmissions Change Package. American Hospital Association. Updated December 2015. Accessed June 10, 2020. https://www.aha.org/sites/default/files/hiin/HRETHEN_ChangePackage_Readmissions.pd
5. Tung Y-C, Chang G-M, Chang H-Y, Yu T-H. Relationship between early physician follow-up and 30-day readmission after acute myocardial infarction and heart failure. Plos One. 2017;12(1):e0170061.
6. Kaplan RM, Koehler J, Zieger PD, et al. Stroke risk as a function of atrial fibrillation duration and CHA2DS2-VASc score. Circulation. 2019;140(20):1639-46.
7. Balan P, Zhao Y, Johnson S, et al. The Society of Thoracic Surgery Risk Score as a predictor of 30-day mortality in transcatheter vs surgical aortic valve replacement: a single-center experience and its implications for the development of a TAVR risk-prediction model. J Invasive Cardiol. 2017;29(3):109-14.
8. Smith LN, Makam AN, Darden D, et al. Acute myocardial infarction readmission risk prediction models: A systematic review of model performance. Circ Cardiovasc Qual Outcomes9.9. 2018;11(1):e003885.
9. Baker H, Oliver-McNeil S, Deng L, Hummel SL. See you in 7: regional hospital collaboration and outcomes in Medicare heart failure patients. JACC Heart Fail. 2015;3(10):765-73.
10. Batten A, Jaeger C, Griffen D, et al. See you in 7: improving acute myocardial infarction follow-up care. BMJ Open Qual. 2018;7(2):e000296.
11. Lee DW, Armistead L, Coleman H, et al. Abstract 15387: Post-discharge follow-up within 14 days reduces 30-day hospital readmission rates in patients with acute myocardial infarction and/or acutely decompensated heart failure. Circulation. 2018;134 (1):A 15387.
12. Thygesen K, Alpert JS, Jaffe AS, et al. Fourth universal definition of myocardial infarction. Circulation. 2018;138 (20):e:618-51.
1. Why it is important to improve care transitions? Society of Hospital Medicine. Accessed June 15, 2020. https://www.hospitalmedicine.org/clinical-topics/care-transitions/
2. Tong L, Arnold T, Yang J, et al. The association between outpatient follow-up visits and all-cause non-elective 30-day readmissions: a retrospective observational cohort study. PloS One. 2018;13(7):e0200691.
3. Jackson C, Shahsahebi M, Wedlake T, DuBard CA. Timeliness of outpatient follow-up: an evidence-based approach for planning after hospital discharge. Ann Fam Med. 2015;13(2):115-22.
4. Health Research & Educational Trust. Preventable Readmissions Change Package. American Hospital Association. Updated December 2015. Accessed June 10, 2020. https://www.aha.org/sites/default/files/hiin/HRETHEN_ChangePackage_Readmissions.pd
5. Tung Y-C, Chang G-M, Chang H-Y, Yu T-H. Relationship between early physician follow-up and 30-day readmission after acute myocardial infarction and heart failure. Plos One. 2017;12(1):e0170061.
6. Kaplan RM, Koehler J, Zieger PD, et al. Stroke risk as a function of atrial fibrillation duration and CHA2DS2-VASc score. Circulation. 2019;140(20):1639-46.
7. Balan P, Zhao Y, Johnson S, et al. The Society of Thoracic Surgery Risk Score as a predictor of 30-day mortality in transcatheter vs surgical aortic valve replacement: a single-center experience and its implications for the development of a TAVR risk-prediction model. J Invasive Cardiol. 2017;29(3):109-14.
8. Smith LN, Makam AN, Darden D, et al. Acute myocardial infarction readmission risk prediction models: A systematic review of model performance. Circ Cardiovasc Qual Outcomes9.9. 2018;11(1):e003885.
9. Baker H, Oliver-McNeil S, Deng L, Hummel SL. See you in 7: regional hospital collaboration and outcomes in Medicare heart failure patients. JACC Heart Fail. 2015;3(10):765-73.
10. Batten A, Jaeger C, Griffen D, et al. See you in 7: improving acute myocardial infarction follow-up care. BMJ Open Qual. 2018;7(2):e000296.
11. Lee DW, Armistead L, Coleman H, et al. Abstract 15387: Post-discharge follow-up within 14 days reduces 30-day hospital readmission rates in patients with acute myocardial infarction and/or acutely decompensated heart failure. Circulation. 2018;134 (1):A 15387.
12. Thygesen K, Alpert JS, Jaffe AS, et al. Fourth universal definition of myocardial infarction. Circulation. 2018;138 (20):e:618-51.











