User login
VARC-3 TAVR technical failure definition ‘highly clinically relevant’
A new study offers early validation of the recently released Valve Academic Research Consortium 3 (VARC-3) definition of technical success after transcatheter aortic valve replacement (TAVR) and highlights its role in patient prognosis.
Results show that one in 10 patients (11.6%) undergoing TAVR with contemporary devices and techniques experiences technical failure, according to VARC-3.
At 30 days, patients with technical failure had significantly higher rates of the composite of cardiovascular (CV) death or stroke (11.5% vs. 3.5%), CV death (6.0% vs. 1.0%), and stroke (7.2% vs. 2.9%), compared with those with technical success.
Technical failure after TAVR was also independently associated with a twofold higher risk for CV death or stroke at 1 year (20.0% vs. 10.3%; hazard ratio, 2.01; 95% CI, 1.37-2.95).
Other independent predictors were history of peripheral artery disease (HR, 1.97), New York Heart Association III or IV disease (HR, 1.86), baseline moderate or greater mitral regurgitation (HR, 1.48), atrial fibrillation (HR, 1.40), and Society of Thoracic Surgeons predicted mortality risk (HR, 1.04).
“We were expecting that we were getting better over time with device iterations, with more experience, so we weren’t surprised by the result. But I think what is somewhat surprising is how much of an impact it has on the outcome,” senior study author Thomas Pilgrim, MD, Inselspital, University of Bern, Switzerland, told this news organization.
The VARC-3 document, introduced last year to some controversy, features a heavier focus on patient outcomes, as well as composite safety and efficacy endpoints. The definition of technical success after TAVR includes freedom from death; successful access, delivery of the device, and retrieval of the delivery system; correct positioning of a prosthetic heart valve into the proper anatomical location; and freedom from surgery or intervention related to the device or to an access-related or cardiac structural complication.
The composite endpoint is meant to replace the VARC-2 definition of “device success,” which also included freedom from death and correct valve positioning but required echocardiographic evaluation. With VARC-3, there is an “immediate measure” of success without having to wait for echocardiography, observed Dr. Pilgrim.
As reported in the Journal of the American College of Cardiology Cardiovascular Interventions, TAVR was a technical success in 1,435 of 1,624 (88.4%) patients. Technical failure occurred in 189 patients related to either vascular complications (8.6%) or procedural death or cardiac complications (3.0%).
The VARC-2 endpoint of device success was observed in 66.1% of patients. The high rate of device failure was largely attributed to a 28% incidence of prosthesis-patient mismatch.
“If you use the VARC-2 device success [definition], you include this patient–prosthesis mismatch, the [valve] gradients, [and] regurgitation and then device success is always lower,” Dr. Pilgrim said.
Asked whether the VARC-3 definition may be missing case failures, he replied: “At this stage, we don’t know how important these echocardiographic parameters are for hard clinical endpoints. Maybe the VARC-2 endpoint was too sensitive or the VARC-3 endpoint is not sensitive enough. This is something we just don’t know at this stage.”
Marco Barbanti, MD, an interventional cardiologist at Rodolico Polyclinic University Hospital-San Marco, Catania, Italy, and author of an accompanying editorial, said VARC-3 represents a more accurate indicator of immediate success of the procedure.
“It’s a more pertinent definition according to what really has an impact on prognosis, and, according to the results of this paper, actually, the calibration of this new definition is quite good,” Dr. Barbanti said in an interview.
Patients with VARC-3 technical failure were older, had a higher body mass index, and had more advanced heart failure symptoms than those with technical success. There were no significant differences between the two groups in echocardiographic or CT data, anesthetic strategy, valve type or size, or use of pre- or post-dilation.
All patients underwent TAVR with current balloon-expandable (Sapien 3/Sapien Ultra, Edwards Lifesciences) or self-expanding (Evolut R/PRO [Medtronic], Portico [Abbott], Symetis ACURATE/ACURATE neo [Boston Scientific]) devices between March 2012 and December 2019. A transfemoral approach was used in 92.5% of patients.
In a landmark analysis with the landmark set at 30 days, the effect of technical failure on adverse outcome was limited to the first 30 days (composite endpoint 0-30 days: HR, 3.42; P < .001; 30-360 days: HR, 1.36; P = .266; P for interaction = .002).
At 1 year, the composite of CV death and stroke endpoint occurred in 24.1% of patients with cardiac technical failure, in 18.8% of patients with vascular technical failure, and in 10.3% of patients with technical success.
In multivariate analyses, cardiac and vascular technical failures were independently associated with a 2.6-fold and 1.9-fold increased risk, respectively, for the composite of cardiovascular death and stroke at 1 year.
Female sex, larger device landing zone calcium volume, and earlier procedures (March 2012 to July 2016) were associated with a higher risk for cardiac technical failure, whereas, consistent with previous studies, higher body mass index and use of the Prostar/Manta versus the ProGlide closure device predicted vascular technical failure.
The findings “underscore that technical success is highly clinically relevant and may serve as one of the pivotal endpoints to evaluate the improvement of TAVR or for head-to-head comparisons of new devices in future clinical trials,” the authors conclude.
The findings reflect the experience of a single high-volume center with highly experienced operators in the prospective BERN TAVR registry, however, and may not be generalizable to other heart centers, they note. Although the registry has standardized follow-up, independent analysis of echocardiographic and CT, and independent event adjudication, vascular anatomy was not systematically assessed, and the potential exists for confounding from unmeasured variables.
Dr. Pilgrim reports research grants to the institution from Edwards Lifesciences, Boston Scientific, and Biotronik, personal fees from Biotronik and Boston Scientific, and other from HighLife SAS. Dr. Barbanti is a consultant for Edwards Lifesciences and Boston Scientific.
A version of this article first appeared on Medscape.com.
A new study offers early validation of the recently released Valve Academic Research Consortium 3 (VARC-3) definition of technical success after transcatheter aortic valve replacement (TAVR) and highlights its role in patient prognosis.
Results show that one in 10 patients (11.6%) undergoing TAVR with contemporary devices and techniques experiences technical failure, according to VARC-3.
At 30 days, patients with technical failure had significantly higher rates of the composite of cardiovascular (CV) death or stroke (11.5% vs. 3.5%), CV death (6.0% vs. 1.0%), and stroke (7.2% vs. 2.9%), compared with those with technical success.
Technical failure after TAVR was also independently associated with a twofold higher risk for CV death or stroke at 1 year (20.0% vs. 10.3%; hazard ratio, 2.01; 95% CI, 1.37-2.95).
Other independent predictors were history of peripheral artery disease (HR, 1.97), New York Heart Association III or IV disease (HR, 1.86), baseline moderate or greater mitral regurgitation (HR, 1.48), atrial fibrillation (HR, 1.40), and Society of Thoracic Surgeons predicted mortality risk (HR, 1.04).
“We were expecting that we were getting better over time with device iterations, with more experience, so we weren’t surprised by the result. But I think what is somewhat surprising is how much of an impact it has on the outcome,” senior study author Thomas Pilgrim, MD, Inselspital, University of Bern, Switzerland, told this news organization.
The VARC-3 document, introduced last year to some controversy, features a heavier focus on patient outcomes, as well as composite safety and efficacy endpoints. The definition of technical success after TAVR includes freedom from death; successful access, delivery of the device, and retrieval of the delivery system; correct positioning of a prosthetic heart valve into the proper anatomical location; and freedom from surgery or intervention related to the device or to an access-related or cardiac structural complication.
The composite endpoint is meant to replace the VARC-2 definition of “device success,” which also included freedom from death and correct valve positioning but required echocardiographic evaluation. With VARC-3, there is an “immediate measure” of success without having to wait for echocardiography, observed Dr. Pilgrim.
As reported in the Journal of the American College of Cardiology Cardiovascular Interventions, TAVR was a technical success in 1,435 of 1,624 (88.4%) patients. Technical failure occurred in 189 patients related to either vascular complications (8.6%) or procedural death or cardiac complications (3.0%).
The VARC-2 endpoint of device success was observed in 66.1% of patients. The high rate of device failure was largely attributed to a 28% incidence of prosthesis-patient mismatch.
“If you use the VARC-2 device success [definition], you include this patient–prosthesis mismatch, the [valve] gradients, [and] regurgitation and then device success is always lower,” Dr. Pilgrim said.
Asked whether the VARC-3 definition may be missing case failures, he replied: “At this stage, we don’t know how important these echocardiographic parameters are for hard clinical endpoints. Maybe the VARC-2 endpoint was too sensitive or the VARC-3 endpoint is not sensitive enough. This is something we just don’t know at this stage.”
Marco Barbanti, MD, an interventional cardiologist at Rodolico Polyclinic University Hospital-San Marco, Catania, Italy, and author of an accompanying editorial, said VARC-3 represents a more accurate indicator of immediate success of the procedure.
“It’s a more pertinent definition according to what really has an impact on prognosis, and, according to the results of this paper, actually, the calibration of this new definition is quite good,” Dr. Barbanti said in an interview.
Patients with VARC-3 technical failure were older, had a higher body mass index, and had more advanced heart failure symptoms than those with technical success. There were no significant differences between the two groups in echocardiographic or CT data, anesthetic strategy, valve type or size, or use of pre- or post-dilation.
All patients underwent TAVR with current balloon-expandable (Sapien 3/Sapien Ultra, Edwards Lifesciences) or self-expanding (Evolut R/PRO [Medtronic], Portico [Abbott], Symetis ACURATE/ACURATE neo [Boston Scientific]) devices between March 2012 and December 2019. A transfemoral approach was used in 92.5% of patients.
In a landmark analysis with the landmark set at 30 days, the effect of technical failure on adverse outcome was limited to the first 30 days (composite endpoint 0-30 days: HR, 3.42; P < .001; 30-360 days: HR, 1.36; P = .266; P for interaction = .002).
At 1 year, the composite of CV death and stroke endpoint occurred in 24.1% of patients with cardiac technical failure, in 18.8% of patients with vascular technical failure, and in 10.3% of patients with technical success.
In multivariate analyses, cardiac and vascular technical failures were independently associated with a 2.6-fold and 1.9-fold increased risk, respectively, for the composite of cardiovascular death and stroke at 1 year.
Female sex, larger device landing zone calcium volume, and earlier procedures (March 2012 to July 2016) were associated with a higher risk for cardiac technical failure, whereas, consistent with previous studies, higher body mass index and use of the Prostar/Manta versus the ProGlide closure device predicted vascular technical failure.
The findings “underscore that technical success is highly clinically relevant and may serve as one of the pivotal endpoints to evaluate the improvement of TAVR or for head-to-head comparisons of new devices in future clinical trials,” the authors conclude.
The findings reflect the experience of a single high-volume center with highly experienced operators in the prospective BERN TAVR registry, however, and may not be generalizable to other heart centers, they note. Although the registry has standardized follow-up, independent analysis of echocardiographic and CT, and independent event adjudication, vascular anatomy was not systematically assessed, and the potential exists for confounding from unmeasured variables.
Dr. Pilgrim reports research grants to the institution from Edwards Lifesciences, Boston Scientific, and Biotronik, personal fees from Biotronik and Boston Scientific, and other from HighLife SAS. Dr. Barbanti is a consultant for Edwards Lifesciences and Boston Scientific.
A version of this article first appeared on Medscape.com.
A new study offers early validation of the recently released Valve Academic Research Consortium 3 (VARC-3) definition of technical success after transcatheter aortic valve replacement (TAVR) and highlights its role in patient prognosis.
Results show that one in 10 patients (11.6%) undergoing TAVR with contemporary devices and techniques experiences technical failure, according to VARC-3.
At 30 days, patients with technical failure had significantly higher rates of the composite of cardiovascular (CV) death or stroke (11.5% vs. 3.5%), CV death (6.0% vs. 1.0%), and stroke (7.2% vs. 2.9%), compared with those with technical success.
Technical failure after TAVR was also independently associated with a twofold higher risk for CV death or stroke at 1 year (20.0% vs. 10.3%; hazard ratio, 2.01; 95% CI, 1.37-2.95).
Other independent predictors were history of peripheral artery disease (HR, 1.97), New York Heart Association III or IV disease (HR, 1.86), baseline moderate or greater mitral regurgitation (HR, 1.48), atrial fibrillation (HR, 1.40), and Society of Thoracic Surgeons predicted mortality risk (HR, 1.04).
“We were expecting that we were getting better over time with device iterations, with more experience, so we weren’t surprised by the result. But I think what is somewhat surprising is how much of an impact it has on the outcome,” senior study author Thomas Pilgrim, MD, Inselspital, University of Bern, Switzerland, told this news organization.
The VARC-3 document, introduced last year to some controversy, features a heavier focus on patient outcomes, as well as composite safety and efficacy endpoints. The definition of technical success after TAVR includes freedom from death; successful access, delivery of the device, and retrieval of the delivery system; correct positioning of a prosthetic heart valve into the proper anatomical location; and freedom from surgery or intervention related to the device or to an access-related or cardiac structural complication.
The composite endpoint is meant to replace the VARC-2 definition of “device success,” which also included freedom from death and correct valve positioning but required echocardiographic evaluation. With VARC-3, there is an “immediate measure” of success without having to wait for echocardiography, observed Dr. Pilgrim.
As reported in the Journal of the American College of Cardiology Cardiovascular Interventions, TAVR was a technical success in 1,435 of 1,624 (88.4%) patients. Technical failure occurred in 189 patients related to either vascular complications (8.6%) or procedural death or cardiac complications (3.0%).
The VARC-2 endpoint of device success was observed in 66.1% of patients. The high rate of device failure was largely attributed to a 28% incidence of prosthesis-patient mismatch.
“If you use the VARC-2 device success [definition], you include this patient–prosthesis mismatch, the [valve] gradients, [and] regurgitation and then device success is always lower,” Dr. Pilgrim said.
Asked whether the VARC-3 definition may be missing case failures, he replied: “At this stage, we don’t know how important these echocardiographic parameters are for hard clinical endpoints. Maybe the VARC-2 endpoint was too sensitive or the VARC-3 endpoint is not sensitive enough. This is something we just don’t know at this stage.”
Marco Barbanti, MD, an interventional cardiologist at Rodolico Polyclinic University Hospital-San Marco, Catania, Italy, and author of an accompanying editorial, said VARC-3 represents a more accurate indicator of immediate success of the procedure.
“It’s a more pertinent definition according to what really has an impact on prognosis, and, according to the results of this paper, actually, the calibration of this new definition is quite good,” Dr. Barbanti said in an interview.
Patients with VARC-3 technical failure were older, had a higher body mass index, and had more advanced heart failure symptoms than those with technical success. There were no significant differences between the two groups in echocardiographic or CT data, anesthetic strategy, valve type or size, or use of pre- or post-dilation.
All patients underwent TAVR with current balloon-expandable (Sapien 3/Sapien Ultra, Edwards Lifesciences) or self-expanding (Evolut R/PRO [Medtronic], Portico [Abbott], Symetis ACURATE/ACURATE neo [Boston Scientific]) devices between March 2012 and December 2019. A transfemoral approach was used in 92.5% of patients.
In a landmark analysis with the landmark set at 30 days, the effect of technical failure on adverse outcome was limited to the first 30 days (composite endpoint 0-30 days: HR, 3.42; P < .001; 30-360 days: HR, 1.36; P = .266; P for interaction = .002).
At 1 year, the composite of CV death and stroke endpoint occurred in 24.1% of patients with cardiac technical failure, in 18.8% of patients with vascular technical failure, and in 10.3% of patients with technical success.
In multivariate analyses, cardiac and vascular technical failures were independently associated with a 2.6-fold and 1.9-fold increased risk, respectively, for the composite of cardiovascular death and stroke at 1 year.
Female sex, larger device landing zone calcium volume, and earlier procedures (March 2012 to July 2016) were associated with a higher risk for cardiac technical failure, whereas, consistent with previous studies, higher body mass index and use of the Prostar/Manta versus the ProGlide closure device predicted vascular technical failure.
The findings “underscore that technical success is highly clinically relevant and may serve as one of the pivotal endpoints to evaluate the improvement of TAVR or for head-to-head comparisons of new devices in future clinical trials,” the authors conclude.
The findings reflect the experience of a single high-volume center with highly experienced operators in the prospective BERN TAVR registry, however, and may not be generalizable to other heart centers, they note. Although the registry has standardized follow-up, independent analysis of echocardiographic and CT, and independent event adjudication, vascular anatomy was not systematically assessed, and the potential exists for confounding from unmeasured variables.
Dr. Pilgrim reports research grants to the institution from Edwards Lifesciences, Boston Scientific, and Biotronik, personal fees from Biotronik and Boston Scientific, and other from HighLife SAS. Dr. Barbanti is a consultant for Edwards Lifesciences and Boston Scientific.
A version of this article first appeared on Medscape.com.
FROM JACC: CARDIOVASCULAR INTERVENTIONS
Topline data for aficamten positive in obstructive HCM
The investigational, next-generation cardiac myosin inhibitor aficamten (previously CK-274, Cytokinetics) continues to show promise as a potential treatment for hypertrophic cardiomyopathy (HCM).
Today, the company announced positive topline results from cohort 3 of the REDWOOD-HCM phase 2 clinical trial, which included 13 patients with symptomatic obstructive HCM and a resting or post-Valsalva left ventricular outflow tract pressure gradient (LVOT-G) of 50 mm Hg or greater whose background therapy included disopyramide.
Treatment with aficamten led to substantial reductions in the average resting LVOT-G, as well as the post-Valsalva LVOT-G (defined as resting gradient less than 30 mm Hg and post-Valsalva gradient less than 50 mm Hg), the company reported.
These “clinically relevant” decreases in pressure gradients were achieved with only modest decreases in average left ventricular ejection fraction (LVEF), the company said.
In no patient did LVEF fall below the prespecified safety threshold of 50%.
New York Heart Association (NYHA) functional class was improved in most patients.
The safety and tolerability of aficamten in cohort 3 were consistent with previous experience in the REDWOOD-HCM trial, with no treatment interruptions and no serious treatment-related adverse events.
The pharmacokinetic data from cohort 3 are similar to those observed in REDWOOD-HCM cohorts 1 and 2, which included HCM patients taking background medications exclusive of disopyramide, as reported previously by this news organization.
“We are encouraged by the clinically relevant reductions in the LVOT gradient observed in these medically refractory patients and are pleased with the safety profile of aficamten when administered in combination with disopyramide,” Fady Malik, MD, PhD, Cytokinetics’ executive vice president of research and development, said in a news release.
“These results represent the first report of patients with obstructive HCM treated with a combination of a cardiac myosin inhibitor and disopyramide and support our plan to include this patient population in SEQUOIA-HCM, our phase 3 trial, which is important, given these patients have exhausted other available medical therapies,” Dr. Malik said.
The results from cohort 3 of the REDWOOD-HCM trial will be presented at the upcoming American College of Cardiology Annual Meeting in April.
A version of this article first appeared on Medscape.com.
The investigational, next-generation cardiac myosin inhibitor aficamten (previously CK-274, Cytokinetics) continues to show promise as a potential treatment for hypertrophic cardiomyopathy (HCM).
Today, the company announced positive topline results from cohort 3 of the REDWOOD-HCM phase 2 clinical trial, which included 13 patients with symptomatic obstructive HCM and a resting or post-Valsalva left ventricular outflow tract pressure gradient (LVOT-G) of 50 mm Hg or greater whose background therapy included disopyramide.
Treatment with aficamten led to substantial reductions in the average resting LVOT-G, as well as the post-Valsalva LVOT-G (defined as resting gradient less than 30 mm Hg and post-Valsalva gradient less than 50 mm Hg), the company reported.
These “clinically relevant” decreases in pressure gradients were achieved with only modest decreases in average left ventricular ejection fraction (LVEF), the company said.
In no patient did LVEF fall below the prespecified safety threshold of 50%.
New York Heart Association (NYHA) functional class was improved in most patients.
The safety and tolerability of aficamten in cohort 3 were consistent with previous experience in the REDWOOD-HCM trial, with no treatment interruptions and no serious treatment-related adverse events.
The pharmacokinetic data from cohort 3 are similar to those observed in REDWOOD-HCM cohorts 1 and 2, which included HCM patients taking background medications exclusive of disopyramide, as reported previously by this news organization.
“We are encouraged by the clinically relevant reductions in the LVOT gradient observed in these medically refractory patients and are pleased with the safety profile of aficamten when administered in combination with disopyramide,” Fady Malik, MD, PhD, Cytokinetics’ executive vice president of research and development, said in a news release.
“These results represent the first report of patients with obstructive HCM treated with a combination of a cardiac myosin inhibitor and disopyramide and support our plan to include this patient population in SEQUOIA-HCM, our phase 3 trial, which is important, given these patients have exhausted other available medical therapies,” Dr. Malik said.
The results from cohort 3 of the REDWOOD-HCM trial will be presented at the upcoming American College of Cardiology Annual Meeting in April.
A version of this article first appeared on Medscape.com.
The investigational, next-generation cardiac myosin inhibitor aficamten (previously CK-274, Cytokinetics) continues to show promise as a potential treatment for hypertrophic cardiomyopathy (HCM).
Today, the company announced positive topline results from cohort 3 of the REDWOOD-HCM phase 2 clinical trial, which included 13 patients with symptomatic obstructive HCM and a resting or post-Valsalva left ventricular outflow tract pressure gradient (LVOT-G) of 50 mm Hg or greater whose background therapy included disopyramide.
Treatment with aficamten led to substantial reductions in the average resting LVOT-G, as well as the post-Valsalva LVOT-G (defined as resting gradient less than 30 mm Hg and post-Valsalva gradient less than 50 mm Hg), the company reported.
These “clinically relevant” decreases in pressure gradients were achieved with only modest decreases in average left ventricular ejection fraction (LVEF), the company said.
In no patient did LVEF fall below the prespecified safety threshold of 50%.
New York Heart Association (NYHA) functional class was improved in most patients.
The safety and tolerability of aficamten in cohort 3 were consistent with previous experience in the REDWOOD-HCM trial, with no treatment interruptions and no serious treatment-related adverse events.
The pharmacokinetic data from cohort 3 are similar to those observed in REDWOOD-HCM cohorts 1 and 2, which included HCM patients taking background medications exclusive of disopyramide, as reported previously by this news organization.
“We are encouraged by the clinically relevant reductions in the LVOT gradient observed in these medically refractory patients and are pleased with the safety profile of aficamten when administered in combination with disopyramide,” Fady Malik, MD, PhD, Cytokinetics’ executive vice president of research and development, said in a news release.
“These results represent the first report of patients with obstructive HCM treated with a combination of a cardiac myosin inhibitor and disopyramide and support our plan to include this patient population in SEQUOIA-HCM, our phase 3 trial, which is important, given these patients have exhausted other available medical therapies,” Dr. Malik said.
The results from cohort 3 of the REDWOOD-HCM trial will be presented at the upcoming American College of Cardiology Annual Meeting in April.
A version of this article first appeared on Medscape.com.
Comparison of Fractional Flow Reserve–Guided PCI and Coronary Bypass Surgery in 3-Vessel Disease
Study Overview
Objective: To determine whether fractional flow reserve (FFR)–guided percutaneous coronary intervention (PCI) is noninferior to coronary-artery bypass grafting (CABG) in patients with 3-vessel coronary artery disease (CAD).
Design: Investigator-initiated, multicenter, international, randomized, controlled trial conducted at 48 sites.
Setting and participants: A total of 1500 patients with angiographically identified 3-vessel CAD not involving the left main coronary artery were randomly assigned to receive FFR-guided PCI with zotarolimus-eluting stents or CABG in a 1:1 ratio. Randomization was stratified according to trial site and diabetes status.
Main outcome measures: The primary end point was major adverse cardiac or cerebrovascular event, defined as death from any cause, myocardial infarction (MI), stroke, or repeat revascularization. The secondary end point was defined as a composite of death, MI, or stroke.
Results: At 1 year, the incidence of the composite primary end point was 10.6% for patients with FFR-guided PCI and 6.9% for patients with CABG (hazard ratio [HR], 1.5; 95% CI, 1.1-2.2; P = .35 for noninferiority), which was not consistent with noninferiority of FFR-guided PCI compared to CABG. The secondary end point occurred in 7.3% of patients in the FFR-guided PCI group compared with 5.2% in the CABG group (HR, 1.4; 95% CI, 0.9-2.1). Individual findings for the outcomes comprising the primary end point for the FFR-guided PCI group vs the CABG group were as follows: death, 1.6% vs 0.9%; MI, 5.2% vs 3.5%; stroke, 0.9% vs 1.1%; and repeat revascularization, 5.9% vs 3.9%. The CABG group had more extended hospital stays and higher incidences of major bleeding, arrhythmia, acute kidney injury, and rehospitalization within 30 days than the FFR-guided PCI group.
Conclusion: FFR-guided PCI was not found to be noninferior to CABG with respect to the incidence of a composite of death, MI, stroke, or repeat revascularization at 1 year.
Commentary
Revascularization for multivessel CAD can be performed by CABG or PCI. Previous studies have shown superior outcomes in patients with multivessel CAD who were treated with CABG compared to PCI.1-3 The Synergy between PCI with Taxus and Cardiac Surgery (SYNTAX) trial, which compared CABG to PCI in patients with multivessel disease or unprotected left main CAD, stratified the anatomic complexity based on SYNTAX score and found that patients with higher anatomic complexity with a high SYNTAX score derive larger benefit from CABG compared to PCI.4 Therefore, the current guidelines favor CABG over PCI in patients with severe 3-vessel disease, except for patients with a lower SYNTAX score (<22) without diabetes.5,6 However, except for a smaller size study,3 the previous trials that led to this recommendation used mostly first-generation drug-eluting stents and have not evaluated second-generation stents that have lower rates of in-stent restenosis and stent thrombosis. In addition, there have been significant improvements in PCI techniques since the study period, including the adoption of a radial approach and superior adjunct pharmacologic therapy. Furthermore, previous studies have not systematically investigated the use of FFR-guided PCI, which has been shown to be superior to angiography-guided PCI or medical treatment alone.7-9
In this context, Fearon and the FAME-3 trial investigators studied the use of FFR-guided PCI with second-generation zotarolimus drug-eluting stents compared to CABG in patients with 3-vessel CAD. They randomized patients with angiographically identified 3-vessel CAD in a 1:1 ratio to receive FFR-guided PCI or CABG at 48 sites internationally. Patients with left main CAD, recent ST-elevation MI, cardiogenic shock, and left-ventricular ejection fraction <30% were excluded. The study results (composite primary end point incidence of 10.6% for patients with FFR-guided PCI vs 6.9% in the CABG group [HR, 1.5; 95% CI, 1.1-2.2; P = 0.35 for noninferiority]) showed that FFR-guided PCI did not meet the noninferiority criterion.
Although the FAME-3 study is an important study, there are a few points to consider. First, 24% of the lesions had a FFR measured at >0.80. The benefit of FFR-guided PCI lies in the number of lesions that are safely deferred compared to angiography-guided PCI. The small number of deferred lesions could have limited the benefit of FFR guidance compared with angiography. Second, this study did not include all comers who had angiographic 3-vessel disease. Patients who had FFR assessment of moderate lesions at the time of diagnostic angiogram and were found to have FFR >0.80 or were deemed single- or 2-vessel disease were likely treated with PCI. Therefore, as the authors point out, the patients included in this study may have been skewed to a higher-risk population compared to previous studies.
Third, the study may not reflect contemporary interventional practice, as the use of intravascular ultrasound was very low (12%). Intravascular ultrasound–guided PCI has been associated with increased luminal gain and improved outcomes compared to angiography-guided PCI.10 Although 20% of the patients in each arm were found to have chronic total occlusions, the completeness of revascularization has not yet been reported. It is possible that the PCI arm had fewer complete revascularizations, which has been shown in previous observational studies to be associated with worse clinical outcomes.11,12
Although the current guidelines favor CABG over PCI in patients with multivessel disease, this recommendation is stratified by anatomic complexity.6 In fact, in the European guidelines, CABG and PCI are both class I recommendations for the treatment of 3-vessel disease with low SYNTAX score in patients without diabetes.5 Although the FAME-3 study failed to show noninferiority in the overall population, when stratified by the SYNTAX score, the major adverse cardiac event rate for the PCI group was numerically lower than that of the CABG group. The results from the FAME-3 study are overall in line with the previous studies and the current guidelines. Future studies are necessary to assess the outcomes of multivessel PCI compared to CABG using the most contemporary interventional practice and achieving complete revascularization in the PCI arm.
Applications for Clinical Practice
In patients with 3-vessel disease, FFR-guided PCI was not found to be noninferior to CABG; this finding is consistent with previous studies.
—Shubham Kanake, BS, Chirag Bavishi, MD, MPH, and Taishi Hirai, MD, University of Missouri, Columbia, MO
Disclosures: None.
1. Farkouh ME, Domanski M, Sleeper LA, et al; FREEDOM Trial Investigators. Strategies for multivessel revascularization in patients with diabetes. N Engl J Med. 2012;367(25):2375-2384. doi:10.1056/NEJMoa1211585
2. Serruys PW, Morice MC, Kappetein AP, et al; SYNTAX Investigators. Percutaneous coronary intervention versus coronary-artery bypass grafting for severe coronary artery disease. N Engl J Med. 2009;360(10):961-972. doi:10.1056/NEJMoa0804626
3. Park SJ, Ahn JM, Kim YH, et al; BEST Trial Investigators. Trial of everolimus-eluting stents or bypass surgery for coronary disease. N Engl J Med. 2015;372(13):1204-1212. doi:10.1056/NEJMoa1415447
4. Stone GW, Kappetein AP, Sabik JF, et al; EXCEL Trial Investigators. Five-year outcomes after PCI or CABG for left main coronary disease. N Engl J Med. 2019; 381(19):1820-1830. doi:10.1056/NEJMoa1909406
5. Neumann FJ, Sousa-Uva M, Ahlsson A, et al; ESC Scientific Document Group. 2018 ESC/EACTS guidelines on myocardial revascularization. Eur Heart J. 2019;40(2):87-165. doi:10.1093/eurheartj/ehy394
6. Writing Committee Members, Lawton JS, Tamis-Holland JE, Bangalore S, et al. 2021 ACC/AHA/SCAI guideline for coronary artery revascularization: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2022;79(2):e21-e129. doi:10.1016/j.jacc.2021.09.006
7. Tonino PAL, De Bruyne B, Pijls NHJ, et al; FAME Study Investigators. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N Engl J Med. 2009;360(3):213-224. doi:10.1056/NEJMoa0807611
8. De Bruyne B, Fearon WF, Pijls NHJ, et al; FAME 2 Trial Investigators. Fractional flow reserve-guided PCI for stable coronary artery disease. N Engl J Med. 2014;371(13):1208-1217. doi:10.1056/NEJMoa1408758
9. Xaplanteris P, Fournier S, Pijls NHJ, et al; FAME 2 Investigators. Five-year outcomes with PCI guided by fractional flow reserve. N Engl J Med. 2018;379(3):250-259. doi:10.1056/NEJMoa1803538
10. Zhang J, Gao X, Kan J, et al. Intravascular ultrasound versus angiography-guided drug-eluting stent implantation: The ULTIMATE trial. J Am Coll Cardiol. 2018;72:3126-3137. doi:10.1016/j.jacc.2018.09.013
11. Garcia S, Sandoval Y, Roukoz H, et al. Outcomes after complete versus incomplete revascularization of patients with multivessel coronary artery disease: a meta-analysis of 89,883 patients enrolled in randomized clinical trials and observational studies. J Am Coll Cardiol. 2013;62:1421-1431. doi:10.1016/j.jacc.2013.05.033
12. Farooq V, Serruys PW, Garcia-Garcia HM et al. The negative impact of incomplete angiographic revascularization on clinical outcomes and its association with total occlusions: the SYNTAX (Synergy Between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery) trial. J Am Coll Cardiol. 2013;61:282-294. doi: 10.1016/j.jacc.2012.10.017
Study Overview
Objective: To determine whether fractional flow reserve (FFR)–guided percutaneous coronary intervention (PCI) is noninferior to coronary-artery bypass grafting (CABG) in patients with 3-vessel coronary artery disease (CAD).
Design: Investigator-initiated, multicenter, international, randomized, controlled trial conducted at 48 sites.
Setting and participants: A total of 1500 patients with angiographically identified 3-vessel CAD not involving the left main coronary artery were randomly assigned to receive FFR-guided PCI with zotarolimus-eluting stents or CABG in a 1:1 ratio. Randomization was stratified according to trial site and diabetes status.
Main outcome measures: The primary end point was major adverse cardiac or cerebrovascular event, defined as death from any cause, myocardial infarction (MI), stroke, or repeat revascularization. The secondary end point was defined as a composite of death, MI, or stroke.
Results: At 1 year, the incidence of the composite primary end point was 10.6% for patients with FFR-guided PCI and 6.9% for patients with CABG (hazard ratio [HR], 1.5; 95% CI, 1.1-2.2; P = .35 for noninferiority), which was not consistent with noninferiority of FFR-guided PCI compared to CABG. The secondary end point occurred in 7.3% of patients in the FFR-guided PCI group compared with 5.2% in the CABG group (HR, 1.4; 95% CI, 0.9-2.1). Individual findings for the outcomes comprising the primary end point for the FFR-guided PCI group vs the CABG group were as follows: death, 1.6% vs 0.9%; MI, 5.2% vs 3.5%; stroke, 0.9% vs 1.1%; and repeat revascularization, 5.9% vs 3.9%. The CABG group had more extended hospital stays and higher incidences of major bleeding, arrhythmia, acute kidney injury, and rehospitalization within 30 days than the FFR-guided PCI group.
Conclusion: FFR-guided PCI was not found to be noninferior to CABG with respect to the incidence of a composite of death, MI, stroke, or repeat revascularization at 1 year.
Commentary
Revascularization for multivessel CAD can be performed by CABG or PCI. Previous studies have shown superior outcomes in patients with multivessel CAD who were treated with CABG compared to PCI.1-3 The Synergy between PCI with Taxus and Cardiac Surgery (SYNTAX) trial, which compared CABG to PCI in patients with multivessel disease or unprotected left main CAD, stratified the anatomic complexity based on SYNTAX score and found that patients with higher anatomic complexity with a high SYNTAX score derive larger benefit from CABG compared to PCI.4 Therefore, the current guidelines favor CABG over PCI in patients with severe 3-vessel disease, except for patients with a lower SYNTAX score (<22) without diabetes.5,6 However, except for a smaller size study,3 the previous trials that led to this recommendation used mostly first-generation drug-eluting stents and have not evaluated second-generation stents that have lower rates of in-stent restenosis and stent thrombosis. In addition, there have been significant improvements in PCI techniques since the study period, including the adoption of a radial approach and superior adjunct pharmacologic therapy. Furthermore, previous studies have not systematically investigated the use of FFR-guided PCI, which has been shown to be superior to angiography-guided PCI or medical treatment alone.7-9
In this context, Fearon and the FAME-3 trial investigators studied the use of FFR-guided PCI with second-generation zotarolimus drug-eluting stents compared to CABG in patients with 3-vessel CAD. They randomized patients with angiographically identified 3-vessel CAD in a 1:1 ratio to receive FFR-guided PCI or CABG at 48 sites internationally. Patients with left main CAD, recent ST-elevation MI, cardiogenic shock, and left-ventricular ejection fraction <30% were excluded. The study results (composite primary end point incidence of 10.6% for patients with FFR-guided PCI vs 6.9% in the CABG group [HR, 1.5; 95% CI, 1.1-2.2; P = 0.35 for noninferiority]) showed that FFR-guided PCI did not meet the noninferiority criterion.
Although the FAME-3 study is an important study, there are a few points to consider. First, 24% of the lesions had a FFR measured at >0.80. The benefit of FFR-guided PCI lies in the number of lesions that are safely deferred compared to angiography-guided PCI. The small number of deferred lesions could have limited the benefit of FFR guidance compared with angiography. Second, this study did not include all comers who had angiographic 3-vessel disease. Patients who had FFR assessment of moderate lesions at the time of diagnostic angiogram and were found to have FFR >0.80 or were deemed single- or 2-vessel disease were likely treated with PCI. Therefore, as the authors point out, the patients included in this study may have been skewed to a higher-risk population compared to previous studies.
Third, the study may not reflect contemporary interventional practice, as the use of intravascular ultrasound was very low (12%). Intravascular ultrasound–guided PCI has been associated with increased luminal gain and improved outcomes compared to angiography-guided PCI.10 Although 20% of the patients in each arm were found to have chronic total occlusions, the completeness of revascularization has not yet been reported. It is possible that the PCI arm had fewer complete revascularizations, which has been shown in previous observational studies to be associated with worse clinical outcomes.11,12
Although the current guidelines favor CABG over PCI in patients with multivessel disease, this recommendation is stratified by anatomic complexity.6 In fact, in the European guidelines, CABG and PCI are both class I recommendations for the treatment of 3-vessel disease with low SYNTAX score in patients without diabetes.5 Although the FAME-3 study failed to show noninferiority in the overall population, when stratified by the SYNTAX score, the major adverse cardiac event rate for the PCI group was numerically lower than that of the CABG group. The results from the FAME-3 study are overall in line with the previous studies and the current guidelines. Future studies are necessary to assess the outcomes of multivessel PCI compared to CABG using the most contemporary interventional practice and achieving complete revascularization in the PCI arm.
Applications for Clinical Practice
In patients with 3-vessel disease, FFR-guided PCI was not found to be noninferior to CABG; this finding is consistent with previous studies.
—Shubham Kanake, BS, Chirag Bavishi, MD, MPH, and Taishi Hirai, MD, University of Missouri, Columbia, MO
Disclosures: None.
Study Overview
Objective: To determine whether fractional flow reserve (FFR)–guided percutaneous coronary intervention (PCI) is noninferior to coronary-artery bypass grafting (CABG) in patients with 3-vessel coronary artery disease (CAD).
Design: Investigator-initiated, multicenter, international, randomized, controlled trial conducted at 48 sites.
Setting and participants: A total of 1500 patients with angiographically identified 3-vessel CAD not involving the left main coronary artery were randomly assigned to receive FFR-guided PCI with zotarolimus-eluting stents or CABG in a 1:1 ratio. Randomization was stratified according to trial site and diabetes status.
Main outcome measures: The primary end point was major adverse cardiac or cerebrovascular event, defined as death from any cause, myocardial infarction (MI), stroke, or repeat revascularization. The secondary end point was defined as a composite of death, MI, or stroke.
Results: At 1 year, the incidence of the composite primary end point was 10.6% for patients with FFR-guided PCI and 6.9% for patients with CABG (hazard ratio [HR], 1.5; 95% CI, 1.1-2.2; P = .35 for noninferiority), which was not consistent with noninferiority of FFR-guided PCI compared to CABG. The secondary end point occurred in 7.3% of patients in the FFR-guided PCI group compared with 5.2% in the CABG group (HR, 1.4; 95% CI, 0.9-2.1). Individual findings for the outcomes comprising the primary end point for the FFR-guided PCI group vs the CABG group were as follows: death, 1.6% vs 0.9%; MI, 5.2% vs 3.5%; stroke, 0.9% vs 1.1%; and repeat revascularization, 5.9% vs 3.9%. The CABG group had more extended hospital stays and higher incidences of major bleeding, arrhythmia, acute kidney injury, and rehospitalization within 30 days than the FFR-guided PCI group.
Conclusion: FFR-guided PCI was not found to be noninferior to CABG with respect to the incidence of a composite of death, MI, stroke, or repeat revascularization at 1 year.
Commentary
Revascularization for multivessel CAD can be performed by CABG or PCI. Previous studies have shown superior outcomes in patients with multivessel CAD who were treated with CABG compared to PCI.1-3 The Synergy between PCI with Taxus and Cardiac Surgery (SYNTAX) trial, which compared CABG to PCI in patients with multivessel disease or unprotected left main CAD, stratified the anatomic complexity based on SYNTAX score and found that patients with higher anatomic complexity with a high SYNTAX score derive larger benefit from CABG compared to PCI.4 Therefore, the current guidelines favor CABG over PCI in patients with severe 3-vessel disease, except for patients with a lower SYNTAX score (<22) without diabetes.5,6 However, except for a smaller size study,3 the previous trials that led to this recommendation used mostly first-generation drug-eluting stents and have not evaluated second-generation stents that have lower rates of in-stent restenosis and stent thrombosis. In addition, there have been significant improvements in PCI techniques since the study period, including the adoption of a radial approach and superior adjunct pharmacologic therapy. Furthermore, previous studies have not systematically investigated the use of FFR-guided PCI, which has been shown to be superior to angiography-guided PCI or medical treatment alone.7-9
In this context, Fearon and the FAME-3 trial investigators studied the use of FFR-guided PCI with second-generation zotarolimus drug-eluting stents compared to CABG in patients with 3-vessel CAD. They randomized patients with angiographically identified 3-vessel CAD in a 1:1 ratio to receive FFR-guided PCI or CABG at 48 sites internationally. Patients with left main CAD, recent ST-elevation MI, cardiogenic shock, and left-ventricular ejection fraction <30% were excluded. The study results (composite primary end point incidence of 10.6% for patients with FFR-guided PCI vs 6.9% in the CABG group [HR, 1.5; 95% CI, 1.1-2.2; P = 0.35 for noninferiority]) showed that FFR-guided PCI did not meet the noninferiority criterion.
Although the FAME-3 study is an important study, there are a few points to consider. First, 24% of the lesions had a FFR measured at >0.80. The benefit of FFR-guided PCI lies in the number of lesions that are safely deferred compared to angiography-guided PCI. The small number of deferred lesions could have limited the benefit of FFR guidance compared with angiography. Second, this study did not include all comers who had angiographic 3-vessel disease. Patients who had FFR assessment of moderate lesions at the time of diagnostic angiogram and were found to have FFR >0.80 or were deemed single- or 2-vessel disease were likely treated with PCI. Therefore, as the authors point out, the patients included in this study may have been skewed to a higher-risk population compared to previous studies.
Third, the study may not reflect contemporary interventional practice, as the use of intravascular ultrasound was very low (12%). Intravascular ultrasound–guided PCI has been associated with increased luminal gain and improved outcomes compared to angiography-guided PCI.10 Although 20% of the patients in each arm were found to have chronic total occlusions, the completeness of revascularization has not yet been reported. It is possible that the PCI arm had fewer complete revascularizations, which has been shown in previous observational studies to be associated with worse clinical outcomes.11,12
Although the current guidelines favor CABG over PCI in patients with multivessel disease, this recommendation is stratified by anatomic complexity.6 In fact, in the European guidelines, CABG and PCI are both class I recommendations for the treatment of 3-vessel disease with low SYNTAX score in patients without diabetes.5 Although the FAME-3 study failed to show noninferiority in the overall population, when stratified by the SYNTAX score, the major adverse cardiac event rate for the PCI group was numerically lower than that of the CABG group. The results from the FAME-3 study are overall in line with the previous studies and the current guidelines. Future studies are necessary to assess the outcomes of multivessel PCI compared to CABG using the most contemporary interventional practice and achieving complete revascularization in the PCI arm.
Applications for Clinical Practice
In patients with 3-vessel disease, FFR-guided PCI was not found to be noninferior to CABG; this finding is consistent with previous studies.
—Shubham Kanake, BS, Chirag Bavishi, MD, MPH, and Taishi Hirai, MD, University of Missouri, Columbia, MO
Disclosures: None.
1. Farkouh ME, Domanski M, Sleeper LA, et al; FREEDOM Trial Investigators. Strategies for multivessel revascularization in patients with diabetes. N Engl J Med. 2012;367(25):2375-2384. doi:10.1056/NEJMoa1211585
2. Serruys PW, Morice MC, Kappetein AP, et al; SYNTAX Investigators. Percutaneous coronary intervention versus coronary-artery bypass grafting for severe coronary artery disease. N Engl J Med. 2009;360(10):961-972. doi:10.1056/NEJMoa0804626
3. Park SJ, Ahn JM, Kim YH, et al; BEST Trial Investigators. Trial of everolimus-eluting stents or bypass surgery for coronary disease. N Engl J Med. 2015;372(13):1204-1212. doi:10.1056/NEJMoa1415447
4. Stone GW, Kappetein AP, Sabik JF, et al; EXCEL Trial Investigators. Five-year outcomes after PCI or CABG for left main coronary disease. N Engl J Med. 2019; 381(19):1820-1830. doi:10.1056/NEJMoa1909406
5. Neumann FJ, Sousa-Uva M, Ahlsson A, et al; ESC Scientific Document Group. 2018 ESC/EACTS guidelines on myocardial revascularization. Eur Heart J. 2019;40(2):87-165. doi:10.1093/eurheartj/ehy394
6. Writing Committee Members, Lawton JS, Tamis-Holland JE, Bangalore S, et al. 2021 ACC/AHA/SCAI guideline for coronary artery revascularization: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2022;79(2):e21-e129. doi:10.1016/j.jacc.2021.09.006
7. Tonino PAL, De Bruyne B, Pijls NHJ, et al; FAME Study Investigators. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N Engl J Med. 2009;360(3):213-224. doi:10.1056/NEJMoa0807611
8. De Bruyne B, Fearon WF, Pijls NHJ, et al; FAME 2 Trial Investigators. Fractional flow reserve-guided PCI for stable coronary artery disease. N Engl J Med. 2014;371(13):1208-1217. doi:10.1056/NEJMoa1408758
9. Xaplanteris P, Fournier S, Pijls NHJ, et al; FAME 2 Investigators. Five-year outcomes with PCI guided by fractional flow reserve. N Engl J Med. 2018;379(3):250-259. doi:10.1056/NEJMoa1803538
10. Zhang J, Gao X, Kan J, et al. Intravascular ultrasound versus angiography-guided drug-eluting stent implantation: The ULTIMATE trial. J Am Coll Cardiol. 2018;72:3126-3137. doi:10.1016/j.jacc.2018.09.013
11. Garcia S, Sandoval Y, Roukoz H, et al. Outcomes after complete versus incomplete revascularization of patients with multivessel coronary artery disease: a meta-analysis of 89,883 patients enrolled in randomized clinical trials and observational studies. J Am Coll Cardiol. 2013;62:1421-1431. doi:10.1016/j.jacc.2013.05.033
12. Farooq V, Serruys PW, Garcia-Garcia HM et al. The negative impact of incomplete angiographic revascularization on clinical outcomes and its association with total occlusions: the SYNTAX (Synergy Between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery) trial. J Am Coll Cardiol. 2013;61:282-294. doi: 10.1016/j.jacc.2012.10.017
1. Farkouh ME, Domanski M, Sleeper LA, et al; FREEDOM Trial Investigators. Strategies for multivessel revascularization in patients with diabetes. N Engl J Med. 2012;367(25):2375-2384. doi:10.1056/NEJMoa1211585
2. Serruys PW, Morice MC, Kappetein AP, et al; SYNTAX Investigators. Percutaneous coronary intervention versus coronary-artery bypass grafting for severe coronary artery disease. N Engl J Med. 2009;360(10):961-972. doi:10.1056/NEJMoa0804626
3. Park SJ, Ahn JM, Kim YH, et al; BEST Trial Investigators. Trial of everolimus-eluting stents or bypass surgery for coronary disease. N Engl J Med. 2015;372(13):1204-1212. doi:10.1056/NEJMoa1415447
4. Stone GW, Kappetein AP, Sabik JF, et al; EXCEL Trial Investigators. Five-year outcomes after PCI or CABG for left main coronary disease. N Engl J Med. 2019; 381(19):1820-1830. doi:10.1056/NEJMoa1909406
5. Neumann FJ, Sousa-Uva M, Ahlsson A, et al; ESC Scientific Document Group. 2018 ESC/EACTS guidelines on myocardial revascularization. Eur Heart J. 2019;40(2):87-165. doi:10.1093/eurheartj/ehy394
6. Writing Committee Members, Lawton JS, Tamis-Holland JE, Bangalore S, et al. 2021 ACC/AHA/SCAI guideline for coronary artery revascularization: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2022;79(2):e21-e129. doi:10.1016/j.jacc.2021.09.006
7. Tonino PAL, De Bruyne B, Pijls NHJ, et al; FAME Study Investigators. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N Engl J Med. 2009;360(3):213-224. doi:10.1056/NEJMoa0807611
8. De Bruyne B, Fearon WF, Pijls NHJ, et al; FAME 2 Trial Investigators. Fractional flow reserve-guided PCI for stable coronary artery disease. N Engl J Med. 2014;371(13):1208-1217. doi:10.1056/NEJMoa1408758
9. Xaplanteris P, Fournier S, Pijls NHJ, et al; FAME 2 Investigators. Five-year outcomes with PCI guided by fractional flow reserve. N Engl J Med. 2018;379(3):250-259. doi:10.1056/NEJMoa1803538
10. Zhang J, Gao X, Kan J, et al. Intravascular ultrasound versus angiography-guided drug-eluting stent implantation: The ULTIMATE trial. J Am Coll Cardiol. 2018;72:3126-3137. doi:10.1016/j.jacc.2018.09.013
11. Garcia S, Sandoval Y, Roukoz H, et al. Outcomes after complete versus incomplete revascularization of patients with multivessel coronary artery disease: a meta-analysis of 89,883 patients enrolled in randomized clinical trials and observational studies. J Am Coll Cardiol. 2013;62:1421-1431. doi:10.1016/j.jacc.2013.05.033
12. Farooq V, Serruys PW, Garcia-Garcia HM et al. The negative impact of incomplete angiographic revascularization on clinical outcomes and its association with total occlusions: the SYNTAX (Synergy Between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery) trial. J Am Coll Cardiol. 2013;61:282-294. doi: 10.1016/j.jacc.2012.10.017
Dr. Jeremiah Stamler, pioneer of preventive cardiology, dies at 102
On the occasion of his 100th birthday, The Washington Post wrote of the trailblazing cardiologist and scientist Jeremiah Dr. Stamler, MD: “You may not know him, but he may have saved your life.”
Hyperbole, it was not.
Over a career spanning more than 70 years, Dr. Stamler transformed medicine and the public’s understanding of diet and lifestyle in cardiovascular health and helped introduce the concept of readily measured ‘risk factors’ such as cholesterol, hypertension, smoking, and diabetes.
Dr. Stamler, the founding chair and a professor emeritus of preventive medicine at Northwestern University’s Feinberg School of Medicine, Chicago, died Wednesday at his home in Sag Harbor, New York, at age 102.
“It is no exaggeration to say that few people in history have had as great an impact on human health,” Donald Lloyd-Jones, MD, chair of the department of preventive medicine at Feinberg and president of the American Heart Association, said in a statement.
“Jerry was a giant intellect who founded the fields of cardiovascular epidemiology and preventive cardiology and led [the way] in defining new prevention concepts right up until his last days,” Dr. Lloyd-Jones added in a statement issued by the university.
Tom Frieden, MD, former director of the Centers for Disease Control and Prevention, tweeted, “Jerry and my father did research on sodium together in the early 1950s. He was a giant in the field of public health, and we’re still benefiting from his brilliance and dedication.”
Roger Blumenthal, MD, director of the Johns Hopkins Ciccarone Center for Prevention of Cardiovascular Disease, tweeted, “R.I.P., Dr. Jeremiah Stamler, ‘the father of preventive cardiology,’ dies at 102 – a true legendary force for health.”
The son of Russian immigrants, Dr. Stamler was born in Brooklyn in 1919 and received a bachelor’s degree from Columbia University and a medical degree from State University of New York.
Discharged from the U.S. Army with the rank of captain, Dr. Stamler and his first wife, Rose, herself a distinguished cardiology researcher, moved to Chicago in 1947 and began researching nutrition and atherosclerosis under pioneering cardiology researcher Louis N. Katz, MD, ultimately showing that atherosclerosis could be introduced by changing the diet of chickens. She died in 1998.
Dr. Stamler also worked for Chicago’s Public Health Department in the 1950s, starting a rheumatic fever prevention program for children and the Chicago Coronary Prevention Evaluation Program, working with higher-risk middle-aged men.
Dr. Stamler’s international INTERSALT study established an independent relationship between blood pressure and increased sodium intake, as well as body mass index and heavy alcohol intake. First published in 1988, the research faced opposition from fellow scientists and the food industry alike.
In a 2006 interview, Dr. Stamler said he and fellow researchers began pressing the American Heart Association in the late 1950s to adopt a public policy of support to improve lifestyles, including smoking cessation and better nutrition. “It took some doing. The AHA was initially reluctant and was under pressure from industry.”
Their efforts were rewarded with the AHA’s first statement on smoking in 1959 and first statement on diet in 1960, whereas, Dr. Stamler noted, “the first World Health Organization statement did not come out until the 1980s.”
Philip Greenland, MD, professor of cardiology and former chair of preventive medicine at Northwestern, described Dr. Stamler as a “force for truth that never backed down when confronted by others who did not share his passion for truth and the best science.”
“I loved working with him since I always knew he would make our research better, clearer, more relevant, and more impactful,” he said in the AHA statement.
A lifelong activist and opponent of the Vietnam War, Dr. Stamler was subpoenaed in May 1965 by the House Un-American Activities Committee (HUAC) along with his nutritionist-assistant Yolanda Hall. Rather than pleading the Fifth Amendment against self-incrimination, Dr. Stamler and Ms. Hall refused to testify before the committee and were charged with contempt of Congress.
With the help of local attorneys, Dr. Stamler filed a civil suit against the HUAC, charging that its mandate was unconstitutional. After 8½ years of litigation that went all the way to the Supreme Court, the government agreed to drop its indictment against Dr. Stamler and he dropped his civil suit against the committee.
A year after the Stamler v. Willis case ended, the House voted to terminate the HUAC. In an essay detailing the high-profile case, Henry Blackburn quipped, “They simply did not know who they were taking on when they tagged ol’ Jerry Stamler.”
“Dr. Stamler’s exceptional science was paralleled by his remarkable humanity. He was a champion of our best American ideals, he was fearless when facing the status quo, and he was tireless in the pursuit of what was right and just. He remains a beacon for all that is noble in medicine,” said Clyde Yancy, MD, MSc, Northwestern’s chair of cardiology.
Over the course of his career, Dr. Stamler published more than 670 peer-reviewed papers, 22 books and monographs, and his work has been cited more than 56,000 times. A committed mentor, Dr. Stamler was the 2014 recipient of the AHA’s Eugene Braunwald Academic Mentorship Award.
A lifelong proponent of the Mediterranean diet, Dr. Stamler divided his time between New York, a home in Italy, and Chicago, with his wife Gloria Beckerman Stamler, whom he married in 2004 and who preceded him in death.
A version of this article first appeared on Medscape.com.
On the occasion of his 100th birthday, The Washington Post wrote of the trailblazing cardiologist and scientist Jeremiah Dr. Stamler, MD: “You may not know him, but he may have saved your life.”
Hyperbole, it was not.
Over a career spanning more than 70 years, Dr. Stamler transformed medicine and the public’s understanding of diet and lifestyle in cardiovascular health and helped introduce the concept of readily measured ‘risk factors’ such as cholesterol, hypertension, smoking, and diabetes.
Dr. Stamler, the founding chair and a professor emeritus of preventive medicine at Northwestern University’s Feinberg School of Medicine, Chicago, died Wednesday at his home in Sag Harbor, New York, at age 102.
“It is no exaggeration to say that few people in history have had as great an impact on human health,” Donald Lloyd-Jones, MD, chair of the department of preventive medicine at Feinberg and president of the American Heart Association, said in a statement.
“Jerry was a giant intellect who founded the fields of cardiovascular epidemiology and preventive cardiology and led [the way] in defining new prevention concepts right up until his last days,” Dr. Lloyd-Jones added in a statement issued by the university.
Tom Frieden, MD, former director of the Centers for Disease Control and Prevention, tweeted, “Jerry and my father did research on sodium together in the early 1950s. He was a giant in the field of public health, and we’re still benefiting from his brilliance and dedication.”
Roger Blumenthal, MD, director of the Johns Hopkins Ciccarone Center for Prevention of Cardiovascular Disease, tweeted, “R.I.P., Dr. Jeremiah Stamler, ‘the father of preventive cardiology,’ dies at 102 – a true legendary force for health.”
The son of Russian immigrants, Dr. Stamler was born in Brooklyn in 1919 and received a bachelor’s degree from Columbia University and a medical degree from State University of New York.
Discharged from the U.S. Army with the rank of captain, Dr. Stamler and his first wife, Rose, herself a distinguished cardiology researcher, moved to Chicago in 1947 and began researching nutrition and atherosclerosis under pioneering cardiology researcher Louis N. Katz, MD, ultimately showing that atherosclerosis could be introduced by changing the diet of chickens. She died in 1998.
Dr. Stamler also worked for Chicago’s Public Health Department in the 1950s, starting a rheumatic fever prevention program for children and the Chicago Coronary Prevention Evaluation Program, working with higher-risk middle-aged men.
Dr. Stamler’s international INTERSALT study established an independent relationship between blood pressure and increased sodium intake, as well as body mass index and heavy alcohol intake. First published in 1988, the research faced opposition from fellow scientists and the food industry alike.
In a 2006 interview, Dr. Stamler said he and fellow researchers began pressing the American Heart Association in the late 1950s to adopt a public policy of support to improve lifestyles, including smoking cessation and better nutrition. “It took some doing. The AHA was initially reluctant and was under pressure from industry.”
Their efforts were rewarded with the AHA’s first statement on smoking in 1959 and first statement on diet in 1960, whereas, Dr. Stamler noted, “the first World Health Organization statement did not come out until the 1980s.”
Philip Greenland, MD, professor of cardiology and former chair of preventive medicine at Northwestern, described Dr. Stamler as a “force for truth that never backed down when confronted by others who did not share his passion for truth and the best science.”
“I loved working with him since I always knew he would make our research better, clearer, more relevant, and more impactful,” he said in the AHA statement.
A lifelong activist and opponent of the Vietnam War, Dr. Stamler was subpoenaed in May 1965 by the House Un-American Activities Committee (HUAC) along with his nutritionist-assistant Yolanda Hall. Rather than pleading the Fifth Amendment against self-incrimination, Dr. Stamler and Ms. Hall refused to testify before the committee and were charged with contempt of Congress.
With the help of local attorneys, Dr. Stamler filed a civil suit against the HUAC, charging that its mandate was unconstitutional. After 8½ years of litigation that went all the way to the Supreme Court, the government agreed to drop its indictment against Dr. Stamler and he dropped his civil suit against the committee.
A year after the Stamler v. Willis case ended, the House voted to terminate the HUAC. In an essay detailing the high-profile case, Henry Blackburn quipped, “They simply did not know who they were taking on when they tagged ol’ Jerry Stamler.”
“Dr. Stamler’s exceptional science was paralleled by his remarkable humanity. He was a champion of our best American ideals, he was fearless when facing the status quo, and he was tireless in the pursuit of what was right and just. He remains a beacon for all that is noble in medicine,” said Clyde Yancy, MD, MSc, Northwestern’s chair of cardiology.
Over the course of his career, Dr. Stamler published more than 670 peer-reviewed papers, 22 books and monographs, and his work has been cited more than 56,000 times. A committed mentor, Dr. Stamler was the 2014 recipient of the AHA’s Eugene Braunwald Academic Mentorship Award.
A lifelong proponent of the Mediterranean diet, Dr. Stamler divided his time between New York, a home in Italy, and Chicago, with his wife Gloria Beckerman Stamler, whom he married in 2004 and who preceded him in death.
A version of this article first appeared on Medscape.com.
On the occasion of his 100th birthday, The Washington Post wrote of the trailblazing cardiologist and scientist Jeremiah Dr. Stamler, MD: “You may not know him, but he may have saved your life.”
Hyperbole, it was not.
Over a career spanning more than 70 years, Dr. Stamler transformed medicine and the public’s understanding of diet and lifestyle in cardiovascular health and helped introduce the concept of readily measured ‘risk factors’ such as cholesterol, hypertension, smoking, and diabetes.
Dr. Stamler, the founding chair and a professor emeritus of preventive medicine at Northwestern University’s Feinberg School of Medicine, Chicago, died Wednesday at his home in Sag Harbor, New York, at age 102.
“It is no exaggeration to say that few people in history have had as great an impact on human health,” Donald Lloyd-Jones, MD, chair of the department of preventive medicine at Feinberg and president of the American Heart Association, said in a statement.
“Jerry was a giant intellect who founded the fields of cardiovascular epidemiology and preventive cardiology and led [the way] in defining new prevention concepts right up until his last days,” Dr. Lloyd-Jones added in a statement issued by the university.
Tom Frieden, MD, former director of the Centers for Disease Control and Prevention, tweeted, “Jerry and my father did research on sodium together in the early 1950s. He was a giant in the field of public health, and we’re still benefiting from his brilliance and dedication.”
Roger Blumenthal, MD, director of the Johns Hopkins Ciccarone Center for Prevention of Cardiovascular Disease, tweeted, “R.I.P., Dr. Jeremiah Stamler, ‘the father of preventive cardiology,’ dies at 102 – a true legendary force for health.”
The son of Russian immigrants, Dr. Stamler was born in Brooklyn in 1919 and received a bachelor’s degree from Columbia University and a medical degree from State University of New York.
Discharged from the U.S. Army with the rank of captain, Dr. Stamler and his first wife, Rose, herself a distinguished cardiology researcher, moved to Chicago in 1947 and began researching nutrition and atherosclerosis under pioneering cardiology researcher Louis N. Katz, MD, ultimately showing that atherosclerosis could be introduced by changing the diet of chickens. She died in 1998.
Dr. Stamler also worked for Chicago’s Public Health Department in the 1950s, starting a rheumatic fever prevention program for children and the Chicago Coronary Prevention Evaluation Program, working with higher-risk middle-aged men.
Dr. Stamler’s international INTERSALT study established an independent relationship between blood pressure and increased sodium intake, as well as body mass index and heavy alcohol intake. First published in 1988, the research faced opposition from fellow scientists and the food industry alike.
In a 2006 interview, Dr. Stamler said he and fellow researchers began pressing the American Heart Association in the late 1950s to adopt a public policy of support to improve lifestyles, including smoking cessation and better nutrition. “It took some doing. The AHA was initially reluctant and was under pressure from industry.”
Their efforts were rewarded with the AHA’s first statement on smoking in 1959 and first statement on diet in 1960, whereas, Dr. Stamler noted, “the first World Health Organization statement did not come out until the 1980s.”
Philip Greenland, MD, professor of cardiology and former chair of preventive medicine at Northwestern, described Dr. Stamler as a “force for truth that never backed down when confronted by others who did not share his passion for truth and the best science.”
“I loved working with him since I always knew he would make our research better, clearer, more relevant, and more impactful,” he said in the AHA statement.
A lifelong activist and opponent of the Vietnam War, Dr. Stamler was subpoenaed in May 1965 by the House Un-American Activities Committee (HUAC) along with his nutritionist-assistant Yolanda Hall. Rather than pleading the Fifth Amendment against self-incrimination, Dr. Stamler and Ms. Hall refused to testify before the committee and were charged with contempt of Congress.
With the help of local attorneys, Dr. Stamler filed a civil suit against the HUAC, charging that its mandate was unconstitutional. After 8½ years of litigation that went all the way to the Supreme Court, the government agreed to drop its indictment against Dr. Stamler and he dropped his civil suit against the committee.
A year after the Stamler v. Willis case ended, the House voted to terminate the HUAC. In an essay detailing the high-profile case, Henry Blackburn quipped, “They simply did not know who they were taking on when they tagged ol’ Jerry Stamler.”
“Dr. Stamler’s exceptional science was paralleled by his remarkable humanity. He was a champion of our best American ideals, he was fearless when facing the status quo, and he was tireless in the pursuit of what was right and just. He remains a beacon for all that is noble in medicine,” said Clyde Yancy, MD, MSc, Northwestern’s chair of cardiology.
Over the course of his career, Dr. Stamler published more than 670 peer-reviewed papers, 22 books and monographs, and his work has been cited more than 56,000 times. A committed mentor, Dr. Stamler was the 2014 recipient of the AHA’s Eugene Braunwald Academic Mentorship Award.
A lifelong proponent of the Mediterranean diet, Dr. Stamler divided his time between New York, a home in Italy, and Chicago, with his wife Gloria Beckerman Stamler, whom he married in 2004 and who preceded him in death.
A version of this article first appeared on Medscape.com.
A dermatologist-led model for CVD prevention in psoriasis may be feasible
A – may be feasible, given the positive perspectives expressed by both clinicians and patients in a set of electronic surveys, researchers say.
In an analysis of survey responses from 183 dermatologists and 322 patients, John S. Barbieri, MD, MBA, and coinvestigators found that more than two-thirds of dermatologists (69.3%) agreed it “seems doable” to check lipids and calculate a 10-year cardiovascular risk score, and over one-third (36.1%) agreed they could prescribe statins when indicated.
The patient survey was distributed through the National Psoriasis Foundation to individuals who were seeing a dermatologist or rheumatologist for psoriatic disease; the clinician survey was distributed through the American Academy of Dermatology to dermatologists who reported caring for patients with psoriasis. (A survey of rheumatologists was similarly conducted, but the number of participants fell short of the needed sample size.)
Most patients surveyed indicated they would be receptive to their dermatologist (or rheumatologist) playing a larger role in screening and managing CVD risk, and that they would be similarly likely to follow recommendations regarding risk screening and management whether the advice came their dermatologist/rheumatologist or from their PCP.
The clinician survey focused on lipids and statin use, and did not address other elements of risk management. Still, the researchers see their findings as an early but promising step in finding better models to improve cardiovascular outcomes for patients with psoriatic disease, who too often do not engage with their PCPs despite their increased risk of CVD and a higher risk of premature mortality from CVD.
Fewer than half of commercially insured adults aged under 65 years visit a PCP each year, the researchers noted. And among the patients in their survey, approximately 20% did not have a PCP or had not seen their PCP in the past year.
Other research has shown that only a small minority of patients with psoriasis have an encounter with their PCP within a year of establishing care with their dermatologist, and that “over half of patients with psoriasis have undetected risk factors like dyslipidemia or hypertension,” Dr. Barbieri, of the department of dermatology at Brigham and Women’s Hospital, Boston, said in an interview.
“There’s a gap here, a missing link in the chain of cardiovascular disease prevention,” he said. “What if the dermatologist or rheumatologist could be more engaged in [CV] risk protection? ... It’s the idea of meeting the patients where they are.”
The surveys
The clinician survey focused on statins because of their ease of use, efficacy and safety, and the need for minimal monitoring, Dr. Barbieri said in the interview. “On the spectrum of things you can do for cardiovascular disease prevention, it’s one of the easiest ones.”
In an accompanying editorial, cardiologists Michael S. Garshick, MD, MS, and Jeffrey S. Berger, MD, MS, both of the department of medicine, New York University, wrote that, “despite the well-described association between psoriasis and CVD, only 35% of patients with psoriasis diagnosed with hyperlipidemia are adequately treated with statin therapy.”
“For many of these patients, their dermatologist or rheumatologist may be their only source of contact with the health care system,” they added.
Most studies targeting CVD risk in psoriasis have focused on targeting psoriatic inflammation, and few studies have explored strategies to improve modifiable CVD risk factor control with pharmacological therapy, they said.
In addition to the questions about receptiveness to identifying and potentially treating CVD risk with statins, the dermatologist survey included a best-worst scaling choice experiment to assess preferences for implementation approaches. Dermatologists were asked to rank their preferences for eight implementation strategies that have been shown in published studies to help increase statin prescribing rates.
The three highest-ranked strategies among dermatologists were clinical decision support, physician educational outreach, and patient education materials. The lowest-ranked strategies were comparisons with peers, a pay-for-performance option, and a mobile app/texting service to remind patients to undergo CVD risk screening.
Of the 183 dermatologists in the survey, 28.4% were from academic settings, 11.5% were from multispecialty groups, and 45.4% were from dermatology groups. (A low response rate of 5.2% for dermatologists raises some questions about the generalizability of the findings, Dr. Garshick and Dr. Berger noted in their editorial.)
Where to go from here?
Asked to comment on the results, Jashin J. Wu, MD, founder and CEO of the Dermatology Research and Education Foundation, Irvine, Calif., who was not involved with the study, said that a larger role in CVD risk management is “not likely to find traction with everyday dermatologists.”
“It’s already a big ask for community dermatologists to go through the approval process to get biologics for patients, so I don’t think many would be willing to add more to their plate by taking a bigger role in CVD management,” he said in an interview. He generally has not prescribed statins, “as I don’t feel that is in my scope of work.”
In the interview, Dr. Barbieri said that a parallel qualitative study, not yet published, has looked at the facilitators and barriers – including time constraints and concern about scope of practice – to statin prescribing and other elements of cardiovascular risk reduction.
All told, he said, a centralized care coordinator model may be the best approach to engage the dermatologist more in CVD prevention, including lipid management, but to also “offload some of the management responsibility.”
In this model, which is partially described by Dr. Barbieri and colleagues, the dermatologist (or rheumatologist) would educate the patient, measure blood pressure and check a lipid panel, and refer the patient to a coordinator who would, in turn, collect more information and calculate a 10-year CVD risk score.
Using a protocol-driven clinical decision support approach, the care coordinator would provide counseling about diet, exercise, and smoking cessation, and about whether statin therapy or blood pressure management is indicated.
“That coordinator would be in a good position to help the patient work with their PCP, if they have one, to find a PCP if they don’t, or to use telemedicine or work with their dermatologist or rheumatologist,” Dr. Barbieri said.
The centralized care coordinator service could be funded through grants, charitable funds, and patient assistance funds so that it is free to patients, he said, and could possibly be “housed in the National Psoriasis Foundation.”
Dr. Barbieri said he and his colleagues plan to design a clinical trial to test whether such a model can be adopted in practice and whether it can improve outcomes associated with CVD risk management.
In their editorial, Dr. Garshick and Dr. Berger, who is director of NYU Langone’s Center for the Prevention of Cardiovascular Disease, wrote that many patients with psoriatic disease have or are at risk for cardiometabolic conditions, and that CVD risk reduction should extend beyond lipid management to include blood pressure, glucose lowering, obesity management, and antiplatelet therapy.
The joint AAD-NPF guidelines for the management and treatment of psoriasis with awareness and attention to comorbidities, published in 2019, were among the first to formally recognize the enhanced CVD risk of patients with psoriasis, they noted.
The guidelines call upon dermatologists to inform patients of the psoriasis-CVD association and ensure their patients are engaged with their PCP or cardiologist for appropriate screening. Now, the editorialists say, “moving the needle forward includes refining and developing modifiable CVD risk reduction strategies for patients with psoriasis, and collaboration between the fields of dermatology, rheumatology, and cardiology is key.”
Incorporating a preventive cardiologist into combined dermatology-rheumatology clinics, or partnering as a freestanding cardioinflammatory clinic, also have potential to improve CVD risk, they wrote.
The survey study was supported by a grant from the NPF Psoriasis Prevention Initiative. Dr. Barbieri reported no conflicts of interest. Several authors disclosed consulting fees and grants from numerous pharmaceutical companies. Dr. Berger reported receiving personal fees from Janssen and grants from AstraZeneca outside of the submitted work. Dr. Garshick reported receiving personal fees from AbbVie outside of the submitted work.
A – may be feasible, given the positive perspectives expressed by both clinicians and patients in a set of electronic surveys, researchers say.
In an analysis of survey responses from 183 dermatologists and 322 patients, John S. Barbieri, MD, MBA, and coinvestigators found that more than two-thirds of dermatologists (69.3%) agreed it “seems doable” to check lipids and calculate a 10-year cardiovascular risk score, and over one-third (36.1%) agreed they could prescribe statins when indicated.
The patient survey was distributed through the National Psoriasis Foundation to individuals who were seeing a dermatologist or rheumatologist for psoriatic disease; the clinician survey was distributed through the American Academy of Dermatology to dermatologists who reported caring for patients with psoriasis. (A survey of rheumatologists was similarly conducted, but the number of participants fell short of the needed sample size.)
Most patients surveyed indicated they would be receptive to their dermatologist (or rheumatologist) playing a larger role in screening and managing CVD risk, and that they would be similarly likely to follow recommendations regarding risk screening and management whether the advice came their dermatologist/rheumatologist or from their PCP.
The clinician survey focused on lipids and statin use, and did not address other elements of risk management. Still, the researchers see their findings as an early but promising step in finding better models to improve cardiovascular outcomes for patients with psoriatic disease, who too often do not engage with their PCPs despite their increased risk of CVD and a higher risk of premature mortality from CVD.
Fewer than half of commercially insured adults aged under 65 years visit a PCP each year, the researchers noted. And among the patients in their survey, approximately 20% did not have a PCP or had not seen their PCP in the past year.
Other research has shown that only a small minority of patients with psoriasis have an encounter with their PCP within a year of establishing care with their dermatologist, and that “over half of patients with psoriasis have undetected risk factors like dyslipidemia or hypertension,” Dr. Barbieri, of the department of dermatology at Brigham and Women’s Hospital, Boston, said in an interview.
“There’s a gap here, a missing link in the chain of cardiovascular disease prevention,” he said. “What if the dermatologist or rheumatologist could be more engaged in [CV] risk protection? ... It’s the idea of meeting the patients where they are.”
The surveys
The clinician survey focused on statins because of their ease of use, efficacy and safety, and the need for minimal monitoring, Dr. Barbieri said in the interview. “On the spectrum of things you can do for cardiovascular disease prevention, it’s one of the easiest ones.”
In an accompanying editorial, cardiologists Michael S. Garshick, MD, MS, and Jeffrey S. Berger, MD, MS, both of the department of medicine, New York University, wrote that, “despite the well-described association between psoriasis and CVD, only 35% of patients with psoriasis diagnosed with hyperlipidemia are adequately treated with statin therapy.”
“For many of these patients, their dermatologist or rheumatologist may be their only source of contact with the health care system,” they added.
Most studies targeting CVD risk in psoriasis have focused on targeting psoriatic inflammation, and few studies have explored strategies to improve modifiable CVD risk factor control with pharmacological therapy, they said.
In addition to the questions about receptiveness to identifying and potentially treating CVD risk with statins, the dermatologist survey included a best-worst scaling choice experiment to assess preferences for implementation approaches. Dermatologists were asked to rank their preferences for eight implementation strategies that have been shown in published studies to help increase statin prescribing rates.
The three highest-ranked strategies among dermatologists were clinical decision support, physician educational outreach, and patient education materials. The lowest-ranked strategies were comparisons with peers, a pay-for-performance option, and a mobile app/texting service to remind patients to undergo CVD risk screening.
Of the 183 dermatologists in the survey, 28.4% were from academic settings, 11.5% were from multispecialty groups, and 45.4% were from dermatology groups. (A low response rate of 5.2% for dermatologists raises some questions about the generalizability of the findings, Dr. Garshick and Dr. Berger noted in their editorial.)
Where to go from here?
Asked to comment on the results, Jashin J. Wu, MD, founder and CEO of the Dermatology Research and Education Foundation, Irvine, Calif., who was not involved with the study, said that a larger role in CVD risk management is “not likely to find traction with everyday dermatologists.”
“It’s already a big ask for community dermatologists to go through the approval process to get biologics for patients, so I don’t think many would be willing to add more to their plate by taking a bigger role in CVD management,” he said in an interview. He generally has not prescribed statins, “as I don’t feel that is in my scope of work.”
In the interview, Dr. Barbieri said that a parallel qualitative study, not yet published, has looked at the facilitators and barriers – including time constraints and concern about scope of practice – to statin prescribing and other elements of cardiovascular risk reduction.
All told, he said, a centralized care coordinator model may be the best approach to engage the dermatologist more in CVD prevention, including lipid management, but to also “offload some of the management responsibility.”
In this model, which is partially described by Dr. Barbieri and colleagues, the dermatologist (or rheumatologist) would educate the patient, measure blood pressure and check a lipid panel, and refer the patient to a coordinator who would, in turn, collect more information and calculate a 10-year CVD risk score.
Using a protocol-driven clinical decision support approach, the care coordinator would provide counseling about diet, exercise, and smoking cessation, and about whether statin therapy or blood pressure management is indicated.
“That coordinator would be in a good position to help the patient work with their PCP, if they have one, to find a PCP if they don’t, or to use telemedicine or work with their dermatologist or rheumatologist,” Dr. Barbieri said.
The centralized care coordinator service could be funded through grants, charitable funds, and patient assistance funds so that it is free to patients, he said, and could possibly be “housed in the National Psoriasis Foundation.”
Dr. Barbieri said he and his colleagues plan to design a clinical trial to test whether such a model can be adopted in practice and whether it can improve outcomes associated with CVD risk management.
In their editorial, Dr. Garshick and Dr. Berger, who is director of NYU Langone’s Center for the Prevention of Cardiovascular Disease, wrote that many patients with psoriatic disease have or are at risk for cardiometabolic conditions, and that CVD risk reduction should extend beyond lipid management to include blood pressure, glucose lowering, obesity management, and antiplatelet therapy.
The joint AAD-NPF guidelines for the management and treatment of psoriasis with awareness and attention to comorbidities, published in 2019, were among the first to formally recognize the enhanced CVD risk of patients with psoriasis, they noted.
The guidelines call upon dermatologists to inform patients of the psoriasis-CVD association and ensure their patients are engaged with their PCP or cardiologist for appropriate screening. Now, the editorialists say, “moving the needle forward includes refining and developing modifiable CVD risk reduction strategies for patients with psoriasis, and collaboration between the fields of dermatology, rheumatology, and cardiology is key.”
Incorporating a preventive cardiologist into combined dermatology-rheumatology clinics, or partnering as a freestanding cardioinflammatory clinic, also have potential to improve CVD risk, they wrote.
The survey study was supported by a grant from the NPF Psoriasis Prevention Initiative. Dr. Barbieri reported no conflicts of interest. Several authors disclosed consulting fees and grants from numerous pharmaceutical companies. Dr. Berger reported receiving personal fees from Janssen and grants from AstraZeneca outside of the submitted work. Dr. Garshick reported receiving personal fees from AbbVie outside of the submitted work.
A – may be feasible, given the positive perspectives expressed by both clinicians and patients in a set of electronic surveys, researchers say.
In an analysis of survey responses from 183 dermatologists and 322 patients, John S. Barbieri, MD, MBA, and coinvestigators found that more than two-thirds of dermatologists (69.3%) agreed it “seems doable” to check lipids and calculate a 10-year cardiovascular risk score, and over one-third (36.1%) agreed they could prescribe statins when indicated.
The patient survey was distributed through the National Psoriasis Foundation to individuals who were seeing a dermatologist or rheumatologist for psoriatic disease; the clinician survey was distributed through the American Academy of Dermatology to dermatologists who reported caring for patients with psoriasis. (A survey of rheumatologists was similarly conducted, but the number of participants fell short of the needed sample size.)
Most patients surveyed indicated they would be receptive to their dermatologist (or rheumatologist) playing a larger role in screening and managing CVD risk, and that they would be similarly likely to follow recommendations regarding risk screening and management whether the advice came their dermatologist/rheumatologist or from their PCP.
The clinician survey focused on lipids and statin use, and did not address other elements of risk management. Still, the researchers see their findings as an early but promising step in finding better models to improve cardiovascular outcomes for patients with psoriatic disease, who too often do not engage with their PCPs despite their increased risk of CVD and a higher risk of premature mortality from CVD.
Fewer than half of commercially insured adults aged under 65 years visit a PCP each year, the researchers noted. And among the patients in their survey, approximately 20% did not have a PCP or had not seen their PCP in the past year.
Other research has shown that only a small minority of patients with psoriasis have an encounter with their PCP within a year of establishing care with their dermatologist, and that “over half of patients with psoriasis have undetected risk factors like dyslipidemia or hypertension,” Dr. Barbieri, of the department of dermatology at Brigham and Women’s Hospital, Boston, said in an interview.
“There’s a gap here, a missing link in the chain of cardiovascular disease prevention,” he said. “What if the dermatologist or rheumatologist could be more engaged in [CV] risk protection? ... It’s the idea of meeting the patients where they are.”
The surveys
The clinician survey focused on statins because of their ease of use, efficacy and safety, and the need for minimal monitoring, Dr. Barbieri said in the interview. “On the spectrum of things you can do for cardiovascular disease prevention, it’s one of the easiest ones.”
In an accompanying editorial, cardiologists Michael S. Garshick, MD, MS, and Jeffrey S. Berger, MD, MS, both of the department of medicine, New York University, wrote that, “despite the well-described association between psoriasis and CVD, only 35% of patients with psoriasis diagnosed with hyperlipidemia are adequately treated with statin therapy.”
“For many of these patients, their dermatologist or rheumatologist may be their only source of contact with the health care system,” they added.
Most studies targeting CVD risk in psoriasis have focused on targeting psoriatic inflammation, and few studies have explored strategies to improve modifiable CVD risk factor control with pharmacological therapy, they said.
In addition to the questions about receptiveness to identifying and potentially treating CVD risk with statins, the dermatologist survey included a best-worst scaling choice experiment to assess preferences for implementation approaches. Dermatologists were asked to rank their preferences for eight implementation strategies that have been shown in published studies to help increase statin prescribing rates.
The three highest-ranked strategies among dermatologists were clinical decision support, physician educational outreach, and patient education materials. The lowest-ranked strategies were comparisons with peers, a pay-for-performance option, and a mobile app/texting service to remind patients to undergo CVD risk screening.
Of the 183 dermatologists in the survey, 28.4% were from academic settings, 11.5% were from multispecialty groups, and 45.4% were from dermatology groups. (A low response rate of 5.2% for dermatologists raises some questions about the generalizability of the findings, Dr. Garshick and Dr. Berger noted in their editorial.)
Where to go from here?
Asked to comment on the results, Jashin J. Wu, MD, founder and CEO of the Dermatology Research and Education Foundation, Irvine, Calif., who was not involved with the study, said that a larger role in CVD risk management is “not likely to find traction with everyday dermatologists.”
“It’s already a big ask for community dermatologists to go through the approval process to get biologics for patients, so I don’t think many would be willing to add more to their plate by taking a bigger role in CVD management,” he said in an interview. He generally has not prescribed statins, “as I don’t feel that is in my scope of work.”
In the interview, Dr. Barbieri said that a parallel qualitative study, not yet published, has looked at the facilitators and barriers – including time constraints and concern about scope of practice – to statin prescribing and other elements of cardiovascular risk reduction.
All told, he said, a centralized care coordinator model may be the best approach to engage the dermatologist more in CVD prevention, including lipid management, but to also “offload some of the management responsibility.”
In this model, which is partially described by Dr. Barbieri and colleagues, the dermatologist (or rheumatologist) would educate the patient, measure blood pressure and check a lipid panel, and refer the patient to a coordinator who would, in turn, collect more information and calculate a 10-year CVD risk score.
Using a protocol-driven clinical decision support approach, the care coordinator would provide counseling about diet, exercise, and smoking cessation, and about whether statin therapy or blood pressure management is indicated.
“That coordinator would be in a good position to help the patient work with their PCP, if they have one, to find a PCP if they don’t, or to use telemedicine or work with their dermatologist or rheumatologist,” Dr. Barbieri said.
The centralized care coordinator service could be funded through grants, charitable funds, and patient assistance funds so that it is free to patients, he said, and could possibly be “housed in the National Psoriasis Foundation.”
Dr. Barbieri said he and his colleagues plan to design a clinical trial to test whether such a model can be adopted in practice and whether it can improve outcomes associated with CVD risk management.
In their editorial, Dr. Garshick and Dr. Berger, who is director of NYU Langone’s Center for the Prevention of Cardiovascular Disease, wrote that many patients with psoriatic disease have or are at risk for cardiometabolic conditions, and that CVD risk reduction should extend beyond lipid management to include blood pressure, glucose lowering, obesity management, and antiplatelet therapy.
The joint AAD-NPF guidelines for the management and treatment of psoriasis with awareness and attention to comorbidities, published in 2019, were among the first to formally recognize the enhanced CVD risk of patients with psoriasis, they noted.
The guidelines call upon dermatologists to inform patients of the psoriasis-CVD association and ensure their patients are engaged with their PCP or cardiologist for appropriate screening. Now, the editorialists say, “moving the needle forward includes refining and developing modifiable CVD risk reduction strategies for patients with psoriasis, and collaboration between the fields of dermatology, rheumatology, and cardiology is key.”
Incorporating a preventive cardiologist into combined dermatology-rheumatology clinics, or partnering as a freestanding cardioinflammatory clinic, also have potential to improve CVD risk, they wrote.
The survey study was supported by a grant from the NPF Psoriasis Prevention Initiative. Dr. Barbieri reported no conflicts of interest. Several authors disclosed consulting fees and grants from numerous pharmaceutical companies. Dr. Berger reported receiving personal fees from Janssen and grants from AstraZeneca outside of the submitted work. Dr. Garshick reported receiving personal fees from AbbVie outside of the submitted work.
FROM JAMA DERMATOLOGY
Ischemic stroke rates higher in young women than young men
Young women appear to be at a higher risk of ischemic stroke than young men, according to a new systematic review of studies on this topic.
The review included 19 studies that reported on sex-specific stroke incidence among young adults and found that overall, in young adults aged 18-35 years, there were 44% more women with ischemic strokes than men.
This gap narrowed in the age group 35-45 years, for which there was conflicting evidence whether more men or women have ischemic strokes.
“An assertion that young women may be disproportionately at risk of ischemic stroke represents a significant departure from our current scientific understanding and may have important implications about the etiology of ischemic strokes in young adults,” the authors note.
“One of the take-home messages from this study is that stroke happens across the entire age spectrum, including young adults, even if they do not have traditional risk factors,” study coauthor Sharon N. Poisson, MD, associate professor of neurology at the University of Colorado Anschutz Medical Campus, Denver, told this news organization.
“If a young person presents with focal neurological symptoms, the possibility of a stroke should not be discounted just because they may not fit the typical profile of a stroke patient. We need more education of the population that young people – including young women – can have a stroke and that fast action to call emergency services is critical,” she said.
The study was published online Jan. 24 in the journal Stroke as part of a special “Go Red for Women” spotlight issue.
The researchers note that historically it has been believed that men have a higher incidence of stroke in every age group until very old age. However, recent evidence focused on the young adult age group has reported that there are more young women (ages 18-45) with ischemic strokes compared with young men, suggesting that young women may be disproportionately at risk compared with their male counterparts.
Pointing out that a better understanding of these sex differences is important in implementing strategies that can more effectively prevent and treat strokes in this age group, the researchers conducted the current review to synthesize the updated evidence.
They searched PubMed from January 2008 to July 2021 for relevant studies that were population-based and reported stroke incidence by sex or sex-specific incidence rate ratios of young adults age 45 and younger. Statistical synthesis was performed to estimate sex difference by age group (less than or equal to 35, 35-45 and less than or equal to 45 years) and stroke type.
They found 19 relevant studies, including three that reported on overlapping data, with a total of 69,793 young adults (33,775 women and 36,018 men).
Nine studies did not show a statistically significant sex difference among young adults less than or equal to 45 years. Three studies found higher rates of ischemic stroke among men among young adults less than or equal to 30 to 35 years. Four studies showed more women with ischemic strokes among young adults less than or equal to 35 years.
Overall, there was an effect of a significantly higher incidence of ischemic stroke in women younger than age 35 years, with an incidence rate ratio (IRR) of 1.44. In the 35- to 45-year age group, there was a nonsignificant sex difference in the rate of ischemic stroke, with a slight trend toward a higher incidence in women (IRR, 1.08).
“In this study the sex difference was not clear in the 35-45 age group. But in the age group of over 45 years we know that men have a higher risk of stroke than women, which is probably related to a higher level of atherosclerotic risk factors,” Dr. Poisson commented.
“Interpreting data on stroke in young people is challenging, as stroke is not so common in this population,” she said. “Combining multiple studies helps, but this also introduces a lot of variability, so we need to interpret these results with some caution. However, this is certainly intriguing data and suggests that something interesting may be going on in young adults,” she added. “These observations give us an initial clue that we need to look further into this issue.”
The study did not look at the possible mechanisms behind the results, as the current data came from administrative datasets that are limited in terms of the information collected.
But Dr. Poisson noted that the traditional risk factors for stroke are high blood pressure and the usual atherosclerotic factors such as high cholesterol.
“These are normally more common in men than in women, and myocardial infarction is more common in younger men than in younger women. But the observation that young women may have a higher risk of stroke than young men suggests that something different may be going on in the mechanism for stroke.”
She pointed out that women have some unique risk factors for stroke, including oral contraceptive use, pregnancy, and the postpartum period, particularly pre-eclampsia during pregnancy. In addition, migraine, especially migraine with aura, is associated with an increased stroke risk, and migraine is more common in young women than in young men.
“We don’t completely understand the role of these risk factors, but they may contribute to the results that we found,” Dr. Poisson commented. “The role of estrogen in stroke is complicated. While estrogen is generally thought to be protective against atherosclerotic risk factors, it also increases risk of clotting, so high estrogen states like pregnancy increase risk of stroke,” she added.
To better understand what is happening, prospectively collected clinical data on younger patients who have had a stroke are needed. Some such studies are underway, but a concerted effort to do this in a large, multicenter registry would be desirable, Dr. Poisson said.
She noted that the presentation of a stroke in young people would be similar to that in the older population, with the most recent acronym to help recognize stroke symptoms being “BE FAST” – balance, eyes (vision), face (drooping), arm, speech (slurred), time (call emergency services quickly).
Call for more women in clinical trials
In an accompanying commentary, Cheryl Bushnell, MD, professor of neurology at Wake Forest School of Medicine, Winston-Salem, N.C., and Moira Kapral, MD, professor in medicine and health policy at the University of Toronto, say these findings support the need for further study to understand and address the causes and risk factors of stroke in young women.
However, they point out that representation and reporting of women in clinical trials of acute stroke continues to be suboptimal, and they call for improved incorporation of sex and gender into study design, analysis, and interpretation, which they say is critical for producing research that is broadly generalizable and applicable to different populations.
Coauthor Stacey L. Daugherty, MD, is funded by the National Institutes of Health. Dr. Poisson and Dr. Kapral have disclosed no relevant financial relationships. Dr. Bushnell reports ownership interest in Care Directions.
A version of this article first appeared on Medscape.com.
Young women appear to be at a higher risk of ischemic stroke than young men, according to a new systematic review of studies on this topic.
The review included 19 studies that reported on sex-specific stroke incidence among young adults and found that overall, in young adults aged 18-35 years, there were 44% more women with ischemic strokes than men.
This gap narrowed in the age group 35-45 years, for which there was conflicting evidence whether more men or women have ischemic strokes.
“An assertion that young women may be disproportionately at risk of ischemic stroke represents a significant departure from our current scientific understanding and may have important implications about the etiology of ischemic strokes in young adults,” the authors note.
“One of the take-home messages from this study is that stroke happens across the entire age spectrum, including young adults, even if they do not have traditional risk factors,” study coauthor Sharon N. Poisson, MD, associate professor of neurology at the University of Colorado Anschutz Medical Campus, Denver, told this news organization.
“If a young person presents with focal neurological symptoms, the possibility of a stroke should not be discounted just because they may not fit the typical profile of a stroke patient. We need more education of the population that young people – including young women – can have a stroke and that fast action to call emergency services is critical,” she said.
The study was published online Jan. 24 in the journal Stroke as part of a special “Go Red for Women” spotlight issue.
The researchers note that historically it has been believed that men have a higher incidence of stroke in every age group until very old age. However, recent evidence focused on the young adult age group has reported that there are more young women (ages 18-45) with ischemic strokes compared with young men, suggesting that young women may be disproportionately at risk compared with their male counterparts.
Pointing out that a better understanding of these sex differences is important in implementing strategies that can more effectively prevent and treat strokes in this age group, the researchers conducted the current review to synthesize the updated evidence.
They searched PubMed from January 2008 to July 2021 for relevant studies that were population-based and reported stroke incidence by sex or sex-specific incidence rate ratios of young adults age 45 and younger. Statistical synthesis was performed to estimate sex difference by age group (less than or equal to 35, 35-45 and less than or equal to 45 years) and stroke type.
They found 19 relevant studies, including three that reported on overlapping data, with a total of 69,793 young adults (33,775 women and 36,018 men).
Nine studies did not show a statistically significant sex difference among young adults less than or equal to 45 years. Three studies found higher rates of ischemic stroke among men among young adults less than or equal to 30 to 35 years. Four studies showed more women with ischemic strokes among young adults less than or equal to 35 years.
Overall, there was an effect of a significantly higher incidence of ischemic stroke in women younger than age 35 years, with an incidence rate ratio (IRR) of 1.44. In the 35- to 45-year age group, there was a nonsignificant sex difference in the rate of ischemic stroke, with a slight trend toward a higher incidence in women (IRR, 1.08).
“In this study the sex difference was not clear in the 35-45 age group. But in the age group of over 45 years we know that men have a higher risk of stroke than women, which is probably related to a higher level of atherosclerotic risk factors,” Dr. Poisson commented.
“Interpreting data on stroke in young people is challenging, as stroke is not so common in this population,” she said. “Combining multiple studies helps, but this also introduces a lot of variability, so we need to interpret these results with some caution. However, this is certainly intriguing data and suggests that something interesting may be going on in young adults,” she added. “These observations give us an initial clue that we need to look further into this issue.”
The study did not look at the possible mechanisms behind the results, as the current data came from administrative datasets that are limited in terms of the information collected.
But Dr. Poisson noted that the traditional risk factors for stroke are high blood pressure and the usual atherosclerotic factors such as high cholesterol.
“These are normally more common in men than in women, and myocardial infarction is more common in younger men than in younger women. But the observation that young women may have a higher risk of stroke than young men suggests that something different may be going on in the mechanism for stroke.”
She pointed out that women have some unique risk factors for stroke, including oral contraceptive use, pregnancy, and the postpartum period, particularly pre-eclampsia during pregnancy. In addition, migraine, especially migraine with aura, is associated with an increased stroke risk, and migraine is more common in young women than in young men.
“We don’t completely understand the role of these risk factors, but they may contribute to the results that we found,” Dr. Poisson commented. “The role of estrogen in stroke is complicated. While estrogen is generally thought to be protective against atherosclerotic risk factors, it also increases risk of clotting, so high estrogen states like pregnancy increase risk of stroke,” she added.
To better understand what is happening, prospectively collected clinical data on younger patients who have had a stroke are needed. Some such studies are underway, but a concerted effort to do this in a large, multicenter registry would be desirable, Dr. Poisson said.
She noted that the presentation of a stroke in young people would be similar to that in the older population, with the most recent acronym to help recognize stroke symptoms being “BE FAST” – balance, eyes (vision), face (drooping), arm, speech (slurred), time (call emergency services quickly).
Call for more women in clinical trials
In an accompanying commentary, Cheryl Bushnell, MD, professor of neurology at Wake Forest School of Medicine, Winston-Salem, N.C., and Moira Kapral, MD, professor in medicine and health policy at the University of Toronto, say these findings support the need for further study to understand and address the causes and risk factors of stroke in young women.
However, they point out that representation and reporting of women in clinical trials of acute stroke continues to be suboptimal, and they call for improved incorporation of sex and gender into study design, analysis, and interpretation, which they say is critical for producing research that is broadly generalizable and applicable to different populations.
Coauthor Stacey L. Daugherty, MD, is funded by the National Institutes of Health. Dr. Poisson and Dr. Kapral have disclosed no relevant financial relationships. Dr. Bushnell reports ownership interest in Care Directions.
A version of this article first appeared on Medscape.com.
Young women appear to be at a higher risk of ischemic stroke than young men, according to a new systematic review of studies on this topic.
The review included 19 studies that reported on sex-specific stroke incidence among young adults and found that overall, in young adults aged 18-35 years, there were 44% more women with ischemic strokes than men.
This gap narrowed in the age group 35-45 years, for which there was conflicting evidence whether more men or women have ischemic strokes.
“An assertion that young women may be disproportionately at risk of ischemic stroke represents a significant departure from our current scientific understanding and may have important implications about the etiology of ischemic strokes in young adults,” the authors note.
“One of the take-home messages from this study is that stroke happens across the entire age spectrum, including young adults, even if they do not have traditional risk factors,” study coauthor Sharon N. Poisson, MD, associate professor of neurology at the University of Colorado Anschutz Medical Campus, Denver, told this news organization.
“If a young person presents with focal neurological symptoms, the possibility of a stroke should not be discounted just because they may not fit the typical profile of a stroke patient. We need more education of the population that young people – including young women – can have a stroke and that fast action to call emergency services is critical,” she said.
The study was published online Jan. 24 in the journal Stroke as part of a special “Go Red for Women” spotlight issue.
The researchers note that historically it has been believed that men have a higher incidence of stroke in every age group until very old age. However, recent evidence focused on the young adult age group has reported that there are more young women (ages 18-45) with ischemic strokes compared with young men, suggesting that young women may be disproportionately at risk compared with their male counterparts.
Pointing out that a better understanding of these sex differences is important in implementing strategies that can more effectively prevent and treat strokes in this age group, the researchers conducted the current review to synthesize the updated evidence.
They searched PubMed from January 2008 to July 2021 for relevant studies that were population-based and reported stroke incidence by sex or sex-specific incidence rate ratios of young adults age 45 and younger. Statistical synthesis was performed to estimate sex difference by age group (less than or equal to 35, 35-45 and less than or equal to 45 years) and stroke type.
They found 19 relevant studies, including three that reported on overlapping data, with a total of 69,793 young adults (33,775 women and 36,018 men).
Nine studies did not show a statistically significant sex difference among young adults less than or equal to 45 years. Three studies found higher rates of ischemic stroke among men among young adults less than or equal to 30 to 35 years. Four studies showed more women with ischemic strokes among young adults less than or equal to 35 years.
Overall, there was an effect of a significantly higher incidence of ischemic stroke in women younger than age 35 years, with an incidence rate ratio (IRR) of 1.44. In the 35- to 45-year age group, there was a nonsignificant sex difference in the rate of ischemic stroke, with a slight trend toward a higher incidence in women (IRR, 1.08).
“In this study the sex difference was not clear in the 35-45 age group. But in the age group of over 45 years we know that men have a higher risk of stroke than women, which is probably related to a higher level of atherosclerotic risk factors,” Dr. Poisson commented.
“Interpreting data on stroke in young people is challenging, as stroke is not so common in this population,” she said. “Combining multiple studies helps, but this also introduces a lot of variability, so we need to interpret these results with some caution. However, this is certainly intriguing data and suggests that something interesting may be going on in young adults,” she added. “These observations give us an initial clue that we need to look further into this issue.”
The study did not look at the possible mechanisms behind the results, as the current data came from administrative datasets that are limited in terms of the information collected.
But Dr. Poisson noted that the traditional risk factors for stroke are high blood pressure and the usual atherosclerotic factors such as high cholesterol.
“These are normally more common in men than in women, and myocardial infarction is more common in younger men than in younger women. But the observation that young women may have a higher risk of stroke than young men suggests that something different may be going on in the mechanism for stroke.”
She pointed out that women have some unique risk factors for stroke, including oral contraceptive use, pregnancy, and the postpartum period, particularly pre-eclampsia during pregnancy. In addition, migraine, especially migraine with aura, is associated with an increased stroke risk, and migraine is more common in young women than in young men.
“We don’t completely understand the role of these risk factors, but they may contribute to the results that we found,” Dr. Poisson commented. “The role of estrogen in stroke is complicated. While estrogen is generally thought to be protective against atherosclerotic risk factors, it also increases risk of clotting, so high estrogen states like pregnancy increase risk of stroke,” she added.
To better understand what is happening, prospectively collected clinical data on younger patients who have had a stroke are needed. Some such studies are underway, but a concerted effort to do this in a large, multicenter registry would be desirable, Dr. Poisson said.
She noted that the presentation of a stroke in young people would be similar to that in the older population, with the most recent acronym to help recognize stroke symptoms being “BE FAST” – balance, eyes (vision), face (drooping), arm, speech (slurred), time (call emergency services quickly).
Call for more women in clinical trials
In an accompanying commentary, Cheryl Bushnell, MD, professor of neurology at Wake Forest School of Medicine, Winston-Salem, N.C., and Moira Kapral, MD, professor in medicine and health policy at the University of Toronto, say these findings support the need for further study to understand and address the causes and risk factors of stroke in young women.
However, they point out that representation and reporting of women in clinical trials of acute stroke continues to be suboptimal, and they call for improved incorporation of sex and gender into study design, analysis, and interpretation, which they say is critical for producing research that is broadly generalizable and applicable to different populations.
Coauthor Stacey L. Daugherty, MD, is funded by the National Institutes of Health. Dr. Poisson and Dr. Kapral have disclosed no relevant financial relationships. Dr. Bushnell reports ownership interest in Care Directions.
A version of this article first appeared on Medscape.com.
Yoga maneuver may prevent vasovagal syncope
The tadasana exercise – a movement-based contemplative practice that gradually corrects orthostatic imbalance by strengthening protective neuromuscular reflexes – practiced for just 15 minutes twice a day, was associated with the complete elimination of episodes of vasovagal syncope for many patients.
“These exercises are very easy to perform, inexpensive, and very effective. This is a very easy fix for a scary and potentially dangerous condition,” lead author Hygriv Rao, MD, said in an interview. “We are excited about these results. We thought it would work, but we did not expect it to be so effective. It seems to work for almost all patients.
“We found that, with the tadasana maneuver, episodes of full syncope, where the patient actually loses consciousness, ceased completely, and episodes of near-syncope, where the patient feels faint but does not completely pass out, were greatly reduced,” Dr. Rao added. “The actual loss of consciousness, which is the most dangerous part, is practically gone. This gives a lot of confidence to patients and their families.”
The researchers reported their initial results from a pilot study of the technique in a letter to JACC: Clinical Electrophysiology that was published online Jan. 26, 2022.
Dr. Rao, a cardiologist at the KIMS Hospitals, Hyderabad, India, explained that vasovagal syncope is a brief loss of consciousness caused by a neurologically induced drop in blood pressure caused by faulty neuromuscular reflexes.
It is typically triggered by emotional stress, prolonged standing, or getting up from a sitting position too quickly.
Very few treatments have been shown effective, with current management approaches involving avoiding triggers, increasing fluids, and if the individual feels an episode coming on, they can take steps to stop it by lying down, raising their legs, or lowering their head to increase blood flow to the brain.
“Recently, there has been a lot of interest in yoga as a preventative therapy for vasovagal syncope,” Dr. Rao noted. “We considered various yoga positions and we chose the tadasana maneuver to study in this context as it resembles exercises sometimes given to patients with vasovagal syncope but with some differences including the addition of synchronized breathing, which may help stabilize autonomic tone.”
For the tadasana maneuver, the individual stands straight with their feet together, arms by their side (against a wall if they need support), and alternatively lift the front and back part of their feet.
They first lift their toes with their weight resting on the ball of their feet, then after a few seconds they raise their heels with their weight on the front of the foot. Then after a few more seconds they lift their arms over their shoulders, stretching upward while standing on their toes.
These movements are synchronized with breathing exercises, with the individual taking a deep breath in as they lift their arms and breathing out again on lowering the arms.
“Each movement takes a few seconds, and each cycle of movements takes about 2 minutes. If this is performed 8 times, then this would take about 15 minutes. We recommend this 15-minute routine twice a day,” Dr. Rao said.
For the current study, 113 patients diagnosed with recurrent vasovagal disorder were counseled to practice standard physical maneuvers and maintain adequate hydration. Medications were prescribed at the discretion of the treating physician.
Of these, 61 patients were additionally trained to practice the tadasana maneuver and asked to practice the movement for 15 minutes twice a day. The mean durations of symptoms and follow-up in the two groups were similar. The average follow-up was about 20 months.
Results showed that episodes of both near-syncope and syncope decreased in both groups but there was a much larger reduction in the patients practicing the tadasana maneuver.
Before treatment, the 52 patients in the conventional group experienced 163 syncope or near-syncope events. At follow-up, 22 symptom recurrences occurred in 12 patients (23%). Total mean events per patient declined from 3 to 0.4.
Full syncope events in this group declined from 65 in 32 patients to 2 in 2 patients (mean per patient, 1.3 to 1), and near-syncope events fell from 98 in 34 patients to 20 in 10 patients (mean per patient, 2.0 to 0.4).
In the tadasana group, 61 patients had 378 syncope/near-syncope events before treatment; at follow-up, only 6 events occurred in 5 patients (8%). Per patient, total events declined from a mean of 6 to 0.1.
Full syncope events fell from 108 in 48 patients to 0 (mean per patient, 1.8 to 0), and near-syncope events declined from 269 in 33 patients to 6 in 5 patients (mean per patient, 4.4 to 0.1).
“This combination of exercise and breathing influences the neuromuscular reflex malfunction that occurs in vasovagal syncope,” Dr. Rao noted. “The movements focus on strengthening neuromuscular reflexes in the quadriceps and the calf muscles, which can increase the blood circulation and venous return, thus preventing blood pooling in the lower body.”
The researchers said this pilot study offers three main findings. First, both conventional therapy and conventional plus tadasana therapy appeared to benefit patients, compared with their respective baseline symptom burden. Second, application of tadasana as an adjunctive treatment was associated with fewer total event recurrences (that is, syncope and near-syncope combined), and third, tadasana was well tolerated, with no adverse events reported.
“The reduction in total events (i.e., syncope and near-syncope events), compared with pretreatment numbers, was substantial and most tadasana patients were managed without any pharmacotherapy,” the authors reported.
Dr. Rao noted that at baseline almost all patients in both groups were taking medications for the condition, but during the study these medications were reduced as fewer episodes occurred. At the end of the follow-up, 80% of the conventional group were still taking medication, compared with just 14% of those in the tadasana group.
Patients had an initial training session in person with a yoga instructor and then received follow-on training by video online. Dr. Rao said there was a very high rate of compliance, “almost 100%.”
He reports that a total of 200 patients have now been treated with this approach at his hospital with very similar results to those seen in the initial study.
This work was supported in part by a grant from the Dr Earl E. Bakken Family in support of heart-brain research. Dr. Rao disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The tadasana exercise – a movement-based contemplative practice that gradually corrects orthostatic imbalance by strengthening protective neuromuscular reflexes – practiced for just 15 minutes twice a day, was associated with the complete elimination of episodes of vasovagal syncope for many patients.
“These exercises are very easy to perform, inexpensive, and very effective. This is a very easy fix for a scary and potentially dangerous condition,” lead author Hygriv Rao, MD, said in an interview. “We are excited about these results. We thought it would work, but we did not expect it to be so effective. It seems to work for almost all patients.
“We found that, with the tadasana maneuver, episodes of full syncope, where the patient actually loses consciousness, ceased completely, and episodes of near-syncope, where the patient feels faint but does not completely pass out, were greatly reduced,” Dr. Rao added. “The actual loss of consciousness, which is the most dangerous part, is practically gone. This gives a lot of confidence to patients and their families.”
The researchers reported their initial results from a pilot study of the technique in a letter to JACC: Clinical Electrophysiology that was published online Jan. 26, 2022.
Dr. Rao, a cardiologist at the KIMS Hospitals, Hyderabad, India, explained that vasovagal syncope is a brief loss of consciousness caused by a neurologically induced drop in blood pressure caused by faulty neuromuscular reflexes.
It is typically triggered by emotional stress, prolonged standing, or getting up from a sitting position too quickly.
Very few treatments have been shown effective, with current management approaches involving avoiding triggers, increasing fluids, and if the individual feels an episode coming on, they can take steps to stop it by lying down, raising their legs, or lowering their head to increase blood flow to the brain.
“Recently, there has been a lot of interest in yoga as a preventative therapy for vasovagal syncope,” Dr. Rao noted. “We considered various yoga positions and we chose the tadasana maneuver to study in this context as it resembles exercises sometimes given to patients with vasovagal syncope but with some differences including the addition of synchronized breathing, which may help stabilize autonomic tone.”
For the tadasana maneuver, the individual stands straight with their feet together, arms by their side (against a wall if they need support), and alternatively lift the front and back part of their feet.
They first lift their toes with their weight resting on the ball of their feet, then after a few seconds they raise their heels with their weight on the front of the foot. Then after a few more seconds they lift their arms over their shoulders, stretching upward while standing on their toes.
These movements are synchronized with breathing exercises, with the individual taking a deep breath in as they lift their arms and breathing out again on lowering the arms.
“Each movement takes a few seconds, and each cycle of movements takes about 2 minutes. If this is performed 8 times, then this would take about 15 minutes. We recommend this 15-minute routine twice a day,” Dr. Rao said.
For the current study, 113 patients diagnosed with recurrent vasovagal disorder were counseled to practice standard physical maneuvers and maintain adequate hydration. Medications were prescribed at the discretion of the treating physician.
Of these, 61 patients were additionally trained to practice the tadasana maneuver and asked to practice the movement for 15 minutes twice a day. The mean durations of symptoms and follow-up in the two groups were similar. The average follow-up was about 20 months.
Results showed that episodes of both near-syncope and syncope decreased in both groups but there was a much larger reduction in the patients practicing the tadasana maneuver.
Before treatment, the 52 patients in the conventional group experienced 163 syncope or near-syncope events. At follow-up, 22 symptom recurrences occurred in 12 patients (23%). Total mean events per patient declined from 3 to 0.4.
Full syncope events in this group declined from 65 in 32 patients to 2 in 2 patients (mean per patient, 1.3 to 1), and near-syncope events fell from 98 in 34 patients to 20 in 10 patients (mean per patient, 2.0 to 0.4).
In the tadasana group, 61 patients had 378 syncope/near-syncope events before treatment; at follow-up, only 6 events occurred in 5 patients (8%). Per patient, total events declined from a mean of 6 to 0.1.
Full syncope events fell from 108 in 48 patients to 0 (mean per patient, 1.8 to 0), and near-syncope events declined from 269 in 33 patients to 6 in 5 patients (mean per patient, 4.4 to 0.1).
“This combination of exercise and breathing influences the neuromuscular reflex malfunction that occurs in vasovagal syncope,” Dr. Rao noted. “The movements focus on strengthening neuromuscular reflexes in the quadriceps and the calf muscles, which can increase the blood circulation and venous return, thus preventing blood pooling in the lower body.”
The researchers said this pilot study offers three main findings. First, both conventional therapy and conventional plus tadasana therapy appeared to benefit patients, compared with their respective baseline symptom burden. Second, application of tadasana as an adjunctive treatment was associated with fewer total event recurrences (that is, syncope and near-syncope combined), and third, tadasana was well tolerated, with no adverse events reported.
“The reduction in total events (i.e., syncope and near-syncope events), compared with pretreatment numbers, was substantial and most tadasana patients were managed without any pharmacotherapy,” the authors reported.
Dr. Rao noted that at baseline almost all patients in both groups were taking medications for the condition, but during the study these medications were reduced as fewer episodes occurred. At the end of the follow-up, 80% of the conventional group were still taking medication, compared with just 14% of those in the tadasana group.
Patients had an initial training session in person with a yoga instructor and then received follow-on training by video online. Dr. Rao said there was a very high rate of compliance, “almost 100%.”
He reports that a total of 200 patients have now been treated with this approach at his hospital with very similar results to those seen in the initial study.
This work was supported in part by a grant from the Dr Earl E. Bakken Family in support of heart-brain research. Dr. Rao disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The tadasana exercise – a movement-based contemplative practice that gradually corrects orthostatic imbalance by strengthening protective neuromuscular reflexes – practiced for just 15 minutes twice a day, was associated with the complete elimination of episodes of vasovagal syncope for many patients.
“These exercises are very easy to perform, inexpensive, and very effective. This is a very easy fix for a scary and potentially dangerous condition,” lead author Hygriv Rao, MD, said in an interview. “We are excited about these results. We thought it would work, but we did not expect it to be so effective. It seems to work for almost all patients.
“We found that, with the tadasana maneuver, episodes of full syncope, where the patient actually loses consciousness, ceased completely, and episodes of near-syncope, where the patient feels faint but does not completely pass out, were greatly reduced,” Dr. Rao added. “The actual loss of consciousness, which is the most dangerous part, is practically gone. This gives a lot of confidence to patients and their families.”
The researchers reported their initial results from a pilot study of the technique in a letter to JACC: Clinical Electrophysiology that was published online Jan. 26, 2022.
Dr. Rao, a cardiologist at the KIMS Hospitals, Hyderabad, India, explained that vasovagal syncope is a brief loss of consciousness caused by a neurologically induced drop in blood pressure caused by faulty neuromuscular reflexes.
It is typically triggered by emotional stress, prolonged standing, or getting up from a sitting position too quickly.
Very few treatments have been shown effective, with current management approaches involving avoiding triggers, increasing fluids, and if the individual feels an episode coming on, they can take steps to stop it by lying down, raising their legs, or lowering their head to increase blood flow to the brain.
“Recently, there has been a lot of interest in yoga as a preventative therapy for vasovagal syncope,” Dr. Rao noted. “We considered various yoga positions and we chose the tadasana maneuver to study in this context as it resembles exercises sometimes given to patients with vasovagal syncope but with some differences including the addition of synchronized breathing, which may help stabilize autonomic tone.”
For the tadasana maneuver, the individual stands straight with their feet together, arms by their side (against a wall if they need support), and alternatively lift the front and back part of their feet.
They first lift their toes with their weight resting on the ball of their feet, then after a few seconds they raise their heels with their weight on the front of the foot. Then after a few more seconds they lift their arms over their shoulders, stretching upward while standing on their toes.
These movements are synchronized with breathing exercises, with the individual taking a deep breath in as they lift their arms and breathing out again on lowering the arms.
“Each movement takes a few seconds, and each cycle of movements takes about 2 minutes. If this is performed 8 times, then this would take about 15 minutes. We recommend this 15-minute routine twice a day,” Dr. Rao said.
For the current study, 113 patients diagnosed with recurrent vasovagal disorder were counseled to practice standard physical maneuvers and maintain adequate hydration. Medications were prescribed at the discretion of the treating physician.
Of these, 61 patients were additionally trained to practice the tadasana maneuver and asked to practice the movement for 15 minutes twice a day. The mean durations of symptoms and follow-up in the two groups were similar. The average follow-up was about 20 months.
Results showed that episodes of both near-syncope and syncope decreased in both groups but there was a much larger reduction in the patients practicing the tadasana maneuver.
Before treatment, the 52 patients in the conventional group experienced 163 syncope or near-syncope events. At follow-up, 22 symptom recurrences occurred in 12 patients (23%). Total mean events per patient declined from 3 to 0.4.
Full syncope events in this group declined from 65 in 32 patients to 2 in 2 patients (mean per patient, 1.3 to 1), and near-syncope events fell from 98 in 34 patients to 20 in 10 patients (mean per patient, 2.0 to 0.4).
In the tadasana group, 61 patients had 378 syncope/near-syncope events before treatment; at follow-up, only 6 events occurred in 5 patients (8%). Per patient, total events declined from a mean of 6 to 0.1.
Full syncope events fell from 108 in 48 patients to 0 (mean per patient, 1.8 to 0), and near-syncope events declined from 269 in 33 patients to 6 in 5 patients (mean per patient, 4.4 to 0.1).
“This combination of exercise and breathing influences the neuromuscular reflex malfunction that occurs in vasovagal syncope,” Dr. Rao noted. “The movements focus on strengthening neuromuscular reflexes in the quadriceps and the calf muscles, which can increase the blood circulation and venous return, thus preventing blood pooling in the lower body.”
The researchers said this pilot study offers three main findings. First, both conventional therapy and conventional plus tadasana therapy appeared to benefit patients, compared with their respective baseline symptom burden. Second, application of tadasana as an adjunctive treatment was associated with fewer total event recurrences (that is, syncope and near-syncope combined), and third, tadasana was well tolerated, with no adverse events reported.
“The reduction in total events (i.e., syncope and near-syncope events), compared with pretreatment numbers, was substantial and most tadasana patients were managed without any pharmacotherapy,” the authors reported.
Dr. Rao noted that at baseline almost all patients in both groups were taking medications for the condition, but during the study these medications were reduced as fewer episodes occurred. At the end of the follow-up, 80% of the conventional group were still taking medication, compared with just 14% of those in the tadasana group.
Patients had an initial training session in person with a yoga instructor and then received follow-on training by video online. Dr. Rao said there was a very high rate of compliance, “almost 100%.”
He reports that a total of 200 patients have now been treated with this approach at his hospital with very similar results to those seen in the initial study.
This work was supported in part by a grant from the Dr Earl E. Bakken Family in support of heart-brain research. Dr. Rao disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JACC: CLINICAL ELECTROPHYSIOLOGY
‘Physiology fights back’ when people are trying to lose weight
Metabolic adaptation – slowing of metabolism in response to weight loss – increases the length of time needed to achieve a target lower weight, a new study of premenopausal women with overweight reports.
All of the 65 sedentary young and middle-aged women with overweight who were on a low-calorie diet (800 calories/day) attained their target lower weight – corresponding to a body mass index (BMI) of 25 kg/m2 or less – after 66-252 days.
But a woman with the largest metabolic adaptation needed to stay on the diet for an extra 70 days, compared with a woman with no metabolic adaptation, to reach the target BMI, after adjusting for dietary adherence.
The study by Catia Martins, PhD, and colleagues was published Jan. 27, 2022, in Obesity.
“Even though adherence to the diet is clearly the most important determinant of time to reach weight loss goals,” wrote Dr. Martins and colleagues, “the present findings are of great clinical relevance as they mean that individuals who are struggling to achieve weight-loss goals, despite assuring compliance with the diet, may indeed be ‘suffering’ from metabolic adaptation during active weight loss.”
Therefore, “clinicians need to consider metabolic adaptation when assessing resistance to weight loss,” they concluded.
Good news: Metabolic adaption ceases when weight stabilizes
“This study shows that a longer than expected duration of intervention to achieve weight loss targets might be due to metabolic adaptation, even after controlling for adherence to the diet,” Dr. Martins said in an interview.
Metabolic adaptation while on a diet makes it harder to lose the last pound than to lose the first pound because as weight loss progresses metabolic adaptation increases, she noted.
However, “the good news is that this mechanism disappears once weight is stabilized (a new energy balance is established), and it is not a predictor of weight regain in the long term,” noted Dr. Martins, associate professor, nutrition sciences research, University of Alabama at Birmingham.
The group published a study in 2020 showing that metabolic adaptation does not predict weight regain at 1 year, and another study, published a few months earlier, showed it is not a barrier to weight-loss maintenance.
The current study findings “provide further evidence of the ways that physiology fights back when people are trying to lose weight,” David B. Sarwer, PhD, who was not involved with this research, said in a press release from the Obesity Society.
“A countless number of environmental variables and other social determinants of health also make weight loss and maintenance challenging for many individuals,” added Dr. Sarwer, director of the Center for Obesity Research and Education at Temple University, Philadelphia.
“Nevertheless, it is import to remember that even a modest weight loss of 5% of initial body weight – much smaller than seen in this study – is associated with clinically significant improvements in weight-related health issues for many individuals,” he stressed.
16% weight loss at 5 weeks with 800-calorie/day diet
It is unclear whether metabolic adaptation contributes to resistance to weight loss by increasing the time necessary to achieve weight-loss goals.
To investigate this, Dr. Martins and associates analyzed data from 36 White women and 29 Black women, aged 20-41 years (mean age, 36), who had a mean BMI of 28.6 and had participated in the diet arms of two studies (ROMEO and JULIET) conducted at Martins’ institution.
Participants received food containing 20% to 22% fat, 20% to 22% protein, and 56% to 58% carbohydrate provided by the center’s research kitchen.
On average, the women were 64% compliant with the diet and lost 12.5 kg (27.6 pounds), a 16% weight loss, over 155 days.
Metabolic adaption was measured 4 weeks after weight stabilization after reaching the weight-loss target.
On average, participants’ resting metabolic rate after weight loss was 46 kcal lower than what would be expected for their lower body weight.
Metabolic adaptation after weight loss was a significant predictor of time to reach the weight-loss goal, after adjusting for target weight loss, energy deficit, and adherence to the diet (R2 adjusted, 0.63; P < .001).
The study findings may not be generalizable to men, older patients, or people with a higher BMI, so further research is needed in a broader population, the researchers concluded.
The research was supported by National Institutes of Health grants. Dr. Martins was supported by a sabbatical grant from the Liaison Committee for Education, Research, and Innovation in Central Norway and the Norwegian University of Science and Technology. The researchers have no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
Metabolic adaptation – slowing of metabolism in response to weight loss – increases the length of time needed to achieve a target lower weight, a new study of premenopausal women with overweight reports.
All of the 65 sedentary young and middle-aged women with overweight who were on a low-calorie diet (800 calories/day) attained their target lower weight – corresponding to a body mass index (BMI) of 25 kg/m2 or less – after 66-252 days.
But a woman with the largest metabolic adaptation needed to stay on the diet for an extra 70 days, compared with a woman with no metabolic adaptation, to reach the target BMI, after adjusting for dietary adherence.
The study by Catia Martins, PhD, and colleagues was published Jan. 27, 2022, in Obesity.
“Even though adherence to the diet is clearly the most important determinant of time to reach weight loss goals,” wrote Dr. Martins and colleagues, “the present findings are of great clinical relevance as they mean that individuals who are struggling to achieve weight-loss goals, despite assuring compliance with the diet, may indeed be ‘suffering’ from metabolic adaptation during active weight loss.”
Therefore, “clinicians need to consider metabolic adaptation when assessing resistance to weight loss,” they concluded.
Good news: Metabolic adaption ceases when weight stabilizes
“This study shows that a longer than expected duration of intervention to achieve weight loss targets might be due to metabolic adaptation, even after controlling for adherence to the diet,” Dr. Martins said in an interview.
Metabolic adaptation while on a diet makes it harder to lose the last pound than to lose the first pound because as weight loss progresses metabolic adaptation increases, she noted.
However, “the good news is that this mechanism disappears once weight is stabilized (a new energy balance is established), and it is not a predictor of weight regain in the long term,” noted Dr. Martins, associate professor, nutrition sciences research, University of Alabama at Birmingham.
The group published a study in 2020 showing that metabolic adaptation does not predict weight regain at 1 year, and another study, published a few months earlier, showed it is not a barrier to weight-loss maintenance.
The current study findings “provide further evidence of the ways that physiology fights back when people are trying to lose weight,” David B. Sarwer, PhD, who was not involved with this research, said in a press release from the Obesity Society.
“A countless number of environmental variables and other social determinants of health also make weight loss and maintenance challenging for many individuals,” added Dr. Sarwer, director of the Center for Obesity Research and Education at Temple University, Philadelphia.
“Nevertheless, it is import to remember that even a modest weight loss of 5% of initial body weight – much smaller than seen in this study – is associated with clinically significant improvements in weight-related health issues for many individuals,” he stressed.
16% weight loss at 5 weeks with 800-calorie/day diet
It is unclear whether metabolic adaptation contributes to resistance to weight loss by increasing the time necessary to achieve weight-loss goals.
To investigate this, Dr. Martins and associates analyzed data from 36 White women and 29 Black women, aged 20-41 years (mean age, 36), who had a mean BMI of 28.6 and had participated in the diet arms of two studies (ROMEO and JULIET) conducted at Martins’ institution.
Participants received food containing 20% to 22% fat, 20% to 22% protein, and 56% to 58% carbohydrate provided by the center’s research kitchen.
On average, the women were 64% compliant with the diet and lost 12.5 kg (27.6 pounds), a 16% weight loss, over 155 days.
Metabolic adaption was measured 4 weeks after weight stabilization after reaching the weight-loss target.
On average, participants’ resting metabolic rate after weight loss was 46 kcal lower than what would be expected for their lower body weight.
Metabolic adaptation after weight loss was a significant predictor of time to reach the weight-loss goal, after adjusting for target weight loss, energy deficit, and adherence to the diet (R2 adjusted, 0.63; P < .001).
The study findings may not be generalizable to men, older patients, or people with a higher BMI, so further research is needed in a broader population, the researchers concluded.
The research was supported by National Institutes of Health grants. Dr. Martins was supported by a sabbatical grant from the Liaison Committee for Education, Research, and Innovation in Central Norway and the Norwegian University of Science and Technology. The researchers have no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
Metabolic adaptation – slowing of metabolism in response to weight loss – increases the length of time needed to achieve a target lower weight, a new study of premenopausal women with overweight reports.
All of the 65 sedentary young and middle-aged women with overweight who were on a low-calorie diet (800 calories/day) attained their target lower weight – corresponding to a body mass index (BMI) of 25 kg/m2 or less – after 66-252 days.
But a woman with the largest metabolic adaptation needed to stay on the diet for an extra 70 days, compared with a woman with no metabolic adaptation, to reach the target BMI, after adjusting for dietary adherence.
The study by Catia Martins, PhD, and colleagues was published Jan. 27, 2022, in Obesity.
“Even though adherence to the diet is clearly the most important determinant of time to reach weight loss goals,” wrote Dr. Martins and colleagues, “the present findings are of great clinical relevance as they mean that individuals who are struggling to achieve weight-loss goals, despite assuring compliance with the diet, may indeed be ‘suffering’ from metabolic adaptation during active weight loss.”
Therefore, “clinicians need to consider metabolic adaptation when assessing resistance to weight loss,” they concluded.
Good news: Metabolic adaption ceases when weight stabilizes
“This study shows that a longer than expected duration of intervention to achieve weight loss targets might be due to metabolic adaptation, even after controlling for adherence to the diet,” Dr. Martins said in an interview.
Metabolic adaptation while on a diet makes it harder to lose the last pound than to lose the first pound because as weight loss progresses metabolic adaptation increases, she noted.
However, “the good news is that this mechanism disappears once weight is stabilized (a new energy balance is established), and it is not a predictor of weight regain in the long term,” noted Dr. Martins, associate professor, nutrition sciences research, University of Alabama at Birmingham.
The group published a study in 2020 showing that metabolic adaptation does not predict weight regain at 1 year, and another study, published a few months earlier, showed it is not a barrier to weight-loss maintenance.
The current study findings “provide further evidence of the ways that physiology fights back when people are trying to lose weight,” David B. Sarwer, PhD, who was not involved with this research, said in a press release from the Obesity Society.
“A countless number of environmental variables and other social determinants of health also make weight loss and maintenance challenging for many individuals,” added Dr. Sarwer, director of the Center for Obesity Research and Education at Temple University, Philadelphia.
“Nevertheless, it is import to remember that even a modest weight loss of 5% of initial body weight – much smaller than seen in this study – is associated with clinically significant improvements in weight-related health issues for many individuals,” he stressed.
16% weight loss at 5 weeks with 800-calorie/day diet
It is unclear whether metabolic adaptation contributes to resistance to weight loss by increasing the time necessary to achieve weight-loss goals.
To investigate this, Dr. Martins and associates analyzed data from 36 White women and 29 Black women, aged 20-41 years (mean age, 36), who had a mean BMI of 28.6 and had participated in the diet arms of two studies (ROMEO and JULIET) conducted at Martins’ institution.
Participants received food containing 20% to 22% fat, 20% to 22% protein, and 56% to 58% carbohydrate provided by the center’s research kitchen.
On average, the women were 64% compliant with the diet and lost 12.5 kg (27.6 pounds), a 16% weight loss, over 155 days.
Metabolic adaption was measured 4 weeks after weight stabilization after reaching the weight-loss target.
On average, participants’ resting metabolic rate after weight loss was 46 kcal lower than what would be expected for their lower body weight.
Metabolic adaptation after weight loss was a significant predictor of time to reach the weight-loss goal, after adjusting for target weight loss, energy deficit, and adherence to the diet (R2 adjusted, 0.63; P < .001).
The study findings may not be generalizable to men, older patients, or people with a higher BMI, so further research is needed in a broader population, the researchers concluded.
The research was supported by National Institutes of Health grants. Dr. Martins was supported by a sabbatical grant from the Liaison Committee for Education, Research, and Innovation in Central Norway and the Norwegian University of Science and Technology. The researchers have no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
FROM OBESITY
Early, subtle, cardiac changes tied to midlife cognitive decline
new research suggests.
Cardiovascular disease risk factors such as high blood pressure, high cholesterol, and diabetes have been associated with an increased risk for cognitive impairment, but much less is known about heart structure and function and the risks for cognition.
“We showed for the first time that, even before the occurrence of cardiovascular disease, people with abnormalities in cardiac structure and function as early as in young adulthood have lower midlife cognition,” investigators Laure Rouch, PharmD, PhD, and Kristine Yaffe, MD, both with the department of psychiatry, University of California, San Francisco, said in a joint email.
“This study reminds us that heart health is key to brain health and that the overlap and interplay between the two is not limited to patients with end-stage heart disease,” Dr. Rouch and Dr. Yaffe said.
The findings were published online Jan. 26, 2022, in Neurology.
Heart/brain connection
The analysis included 2,653 participants in the Coronary Artery Risk Development in Young Adults (CARDIA) study.
Echocardiograms were obtained at year 5, 25, and 30 study visits – at mean ages of 30, 50, and 55 years, respectively. At year 30, participants underwent a standard battery of tests measuring global cognition, processing speed, executive function, delayed verbal memory, and verbal fluency.
Over 25 years, there was an average increase in left ventricular mass of 0.27 g/m2 per year – from a mean of 80.5 g/m2 in year 5 to 86.0 g/m2 in year 30.
Left atrial volume increased by an average of 0.42 mL/m2 per year, from 16 mL/m2 in year 5 to 26 mL/m2 in year 30.
Left ventricular ejection fraction (LVEF) decreased by 0.11% per year, from 63.3% in year 5 to 59.7% in year 30.
After adjustment for demographics and education, an increase in left ventricular mass of at least 1 standard deviation over 25 years was associated with lower cognition on most tests (P ≤ .02).
An increase in left atrial volume over the study period was associated with lower global cognition (P = .04), whereas a decrease in LVEF was not associated with cognition. Further adjustment for cardiovascular risk factors yielded similar results.
“A more effective collaboration is needed between cardiologists and neurologists to promote healthy brain aging,” Dr. Rouch and Dr. Yaffe said.
“Echocardiography is a widely available, relatively inexpensive, and noninvasive imaging method that could be integrated into a risk assessment for cognitive impairment,” they added.
Looking ahead, the investigators noted there is a need for further research to determine whether interventions to improve cardiac structure and diastolic function could also benefit brain health.
They should also investigate the role of arterial stiffness and cerebral small vessel disease in the relationship between cardiac structure, function, and cognition, the researchers added.
First structural biomarker
Commenting on the study, Shaheen E. Lakhan, MD, PhD, a neurologist in Newton, Mass., said the study is important because, “thus far, the connections have really been physiological parameters,” such as blood pressure and cognitive health.
“This is really strong evidence of a structural cardiac biomarker that can be measured before and independent of changes in physiology or diseased state,” said Dr. Lakhan, who was not involved with the research.
As more and more interventions are being introduced for addressing disorders of cognition, “this potential structural finding may serve as a solid biomarker to determine” what lifestyle or drug therapy should be taken, he added.
Also weighing in on the findings, Pierre Fayad, MD, professor in the department of neurological sciences and director of the Nebraska Stroke Center, University of Nebraska Medical Center, Omaha, said CARDIA is “an important study” providing “precious data.”
The reported changes in cardiac structure and function “precede the clinical symptomatology, as the follow-up stops before they enter into later adulthood, where the risk of clinical events dramatically rises. Meaning these patients still have not had stroke, congestive heart failure, heart attack or dementia, but some of them could be on that trajectory later in their life,” Dr. Fayad told this news organization.
Documenting such changes over time is “valuable to give an insight into what leads us to such progression,” he noted.
How reliably predictive the findings are for eventual clinical cognitive impairment “will need to be confirmed and verified” in future studies, he added.
“If verified, it could be helpful to provide interventions to those with the left atrial volume enlargement marker and see their effectiveness at preventing eventual clinical cognitive impairment,” said Dr. Fayad.
The CARDIA study is supported by the National Heart, Lung, and Blood Institute in collaboration with the University of Alabama at Birmingham, Northwestern University, the University of Minnesota, and the Kaiser Foundation Research Institute. Rouch, Lakhan, and Dr. Fayad have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
Cardiovascular disease risk factors such as high blood pressure, high cholesterol, and diabetes have been associated with an increased risk for cognitive impairment, but much less is known about heart structure and function and the risks for cognition.
“We showed for the first time that, even before the occurrence of cardiovascular disease, people with abnormalities in cardiac structure and function as early as in young adulthood have lower midlife cognition,” investigators Laure Rouch, PharmD, PhD, and Kristine Yaffe, MD, both with the department of psychiatry, University of California, San Francisco, said in a joint email.
“This study reminds us that heart health is key to brain health and that the overlap and interplay between the two is not limited to patients with end-stage heart disease,” Dr. Rouch and Dr. Yaffe said.
The findings were published online Jan. 26, 2022, in Neurology.
Heart/brain connection
The analysis included 2,653 participants in the Coronary Artery Risk Development in Young Adults (CARDIA) study.
Echocardiograms were obtained at year 5, 25, and 30 study visits – at mean ages of 30, 50, and 55 years, respectively. At year 30, participants underwent a standard battery of tests measuring global cognition, processing speed, executive function, delayed verbal memory, and verbal fluency.
Over 25 years, there was an average increase in left ventricular mass of 0.27 g/m2 per year – from a mean of 80.5 g/m2 in year 5 to 86.0 g/m2 in year 30.
Left atrial volume increased by an average of 0.42 mL/m2 per year, from 16 mL/m2 in year 5 to 26 mL/m2 in year 30.
Left ventricular ejection fraction (LVEF) decreased by 0.11% per year, from 63.3% in year 5 to 59.7% in year 30.
After adjustment for demographics and education, an increase in left ventricular mass of at least 1 standard deviation over 25 years was associated with lower cognition on most tests (P ≤ .02).
An increase in left atrial volume over the study period was associated with lower global cognition (P = .04), whereas a decrease in LVEF was not associated with cognition. Further adjustment for cardiovascular risk factors yielded similar results.
“A more effective collaboration is needed between cardiologists and neurologists to promote healthy brain aging,” Dr. Rouch and Dr. Yaffe said.
“Echocardiography is a widely available, relatively inexpensive, and noninvasive imaging method that could be integrated into a risk assessment for cognitive impairment,” they added.
Looking ahead, the investigators noted there is a need for further research to determine whether interventions to improve cardiac structure and diastolic function could also benefit brain health.
They should also investigate the role of arterial stiffness and cerebral small vessel disease in the relationship between cardiac structure, function, and cognition, the researchers added.
First structural biomarker
Commenting on the study, Shaheen E. Lakhan, MD, PhD, a neurologist in Newton, Mass., said the study is important because, “thus far, the connections have really been physiological parameters,” such as blood pressure and cognitive health.
“This is really strong evidence of a structural cardiac biomarker that can be measured before and independent of changes in physiology or diseased state,” said Dr. Lakhan, who was not involved with the research.
As more and more interventions are being introduced for addressing disorders of cognition, “this potential structural finding may serve as a solid biomarker to determine” what lifestyle or drug therapy should be taken, he added.
Also weighing in on the findings, Pierre Fayad, MD, professor in the department of neurological sciences and director of the Nebraska Stroke Center, University of Nebraska Medical Center, Omaha, said CARDIA is “an important study” providing “precious data.”
The reported changes in cardiac structure and function “precede the clinical symptomatology, as the follow-up stops before they enter into later adulthood, where the risk of clinical events dramatically rises. Meaning these patients still have not had stroke, congestive heart failure, heart attack or dementia, but some of them could be on that trajectory later in their life,” Dr. Fayad told this news organization.
Documenting such changes over time is “valuable to give an insight into what leads us to such progression,” he noted.
How reliably predictive the findings are for eventual clinical cognitive impairment “will need to be confirmed and verified” in future studies, he added.
“If verified, it could be helpful to provide interventions to those with the left atrial volume enlargement marker and see their effectiveness at preventing eventual clinical cognitive impairment,” said Dr. Fayad.
The CARDIA study is supported by the National Heart, Lung, and Blood Institute in collaboration with the University of Alabama at Birmingham, Northwestern University, the University of Minnesota, and the Kaiser Foundation Research Institute. Rouch, Lakhan, and Dr. Fayad have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
Cardiovascular disease risk factors such as high blood pressure, high cholesterol, and diabetes have been associated with an increased risk for cognitive impairment, but much less is known about heart structure and function and the risks for cognition.
“We showed for the first time that, even before the occurrence of cardiovascular disease, people with abnormalities in cardiac structure and function as early as in young adulthood have lower midlife cognition,” investigators Laure Rouch, PharmD, PhD, and Kristine Yaffe, MD, both with the department of psychiatry, University of California, San Francisco, said in a joint email.
“This study reminds us that heart health is key to brain health and that the overlap and interplay between the two is not limited to patients with end-stage heart disease,” Dr. Rouch and Dr. Yaffe said.
The findings were published online Jan. 26, 2022, in Neurology.
Heart/brain connection
The analysis included 2,653 participants in the Coronary Artery Risk Development in Young Adults (CARDIA) study.
Echocardiograms were obtained at year 5, 25, and 30 study visits – at mean ages of 30, 50, and 55 years, respectively. At year 30, participants underwent a standard battery of tests measuring global cognition, processing speed, executive function, delayed verbal memory, and verbal fluency.
Over 25 years, there was an average increase in left ventricular mass of 0.27 g/m2 per year – from a mean of 80.5 g/m2 in year 5 to 86.0 g/m2 in year 30.
Left atrial volume increased by an average of 0.42 mL/m2 per year, from 16 mL/m2 in year 5 to 26 mL/m2 in year 30.
Left ventricular ejection fraction (LVEF) decreased by 0.11% per year, from 63.3% in year 5 to 59.7% in year 30.
After adjustment for demographics and education, an increase in left ventricular mass of at least 1 standard deviation over 25 years was associated with lower cognition on most tests (P ≤ .02).
An increase in left atrial volume over the study period was associated with lower global cognition (P = .04), whereas a decrease in LVEF was not associated with cognition. Further adjustment for cardiovascular risk factors yielded similar results.
“A more effective collaboration is needed between cardiologists and neurologists to promote healthy brain aging,” Dr. Rouch and Dr. Yaffe said.
“Echocardiography is a widely available, relatively inexpensive, and noninvasive imaging method that could be integrated into a risk assessment for cognitive impairment,” they added.
Looking ahead, the investigators noted there is a need for further research to determine whether interventions to improve cardiac structure and diastolic function could also benefit brain health.
They should also investigate the role of arterial stiffness and cerebral small vessel disease in the relationship between cardiac structure, function, and cognition, the researchers added.
First structural biomarker
Commenting on the study, Shaheen E. Lakhan, MD, PhD, a neurologist in Newton, Mass., said the study is important because, “thus far, the connections have really been physiological parameters,” such as blood pressure and cognitive health.
“This is really strong evidence of a structural cardiac biomarker that can be measured before and independent of changes in physiology or diseased state,” said Dr. Lakhan, who was not involved with the research.
As more and more interventions are being introduced for addressing disorders of cognition, “this potential structural finding may serve as a solid biomarker to determine” what lifestyle or drug therapy should be taken, he added.
Also weighing in on the findings, Pierre Fayad, MD, professor in the department of neurological sciences and director of the Nebraska Stroke Center, University of Nebraska Medical Center, Omaha, said CARDIA is “an important study” providing “precious data.”
The reported changes in cardiac structure and function “precede the clinical symptomatology, as the follow-up stops before they enter into later adulthood, where the risk of clinical events dramatically rises. Meaning these patients still have not had stroke, congestive heart failure, heart attack or dementia, but some of them could be on that trajectory later in their life,” Dr. Fayad told this news organization.
Documenting such changes over time is “valuable to give an insight into what leads us to such progression,” he noted.
How reliably predictive the findings are for eventual clinical cognitive impairment “will need to be confirmed and verified” in future studies, he added.
“If verified, it could be helpful to provide interventions to those with the left atrial volume enlargement marker and see their effectiveness at preventing eventual clinical cognitive impairment,” said Dr. Fayad.
The CARDIA study is supported by the National Heart, Lung, and Blood Institute in collaboration with the University of Alabama at Birmingham, Northwestern University, the University of Minnesota, and the Kaiser Foundation Research Institute. Rouch, Lakhan, and Dr. Fayad have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY
Hong Kong, U.S., Israeli data illuminate COVID vaccine myocarditis
Why some COVID-19 vaccines seem occasionally to cause a distinctive form of myocarditis, and why adolescent boys and young men appear most vulnerable, remain a mystery. But the entity’s prevalence, nuances of presentation, and likely clinical course have come into sharper view after recent additions to the literature.
Two new publications all but confirm that the rare cases of myocarditis closely following vaccination against SARS-CoV-2, primarily with one of the mRNA-based vaccines from Pfizer-BioNTech and Moderna, is a clinically different creature from myocarditis physicians were likely to see before the pandemic.
A third report unveils rates of hospitalization for myocarditis linked to Pfizer-BioNTech vaccination in the 12- to 15-year age group, based on active surveillance across Israel. Of note, the rates were lower than corresponding numbers among the country’s 16- to 19-year-olds published in late 2021 by the same authors.
No link with CoronaVac
A case-control study covering almost the entire population of Hong Kong from February to August 2021 confirms a slight but significant excess risk for myocarditis and, to a lesser degree, pericarditis, after injections of the Pfizer-BioNTech vaccine. As consistently reported from other studies, the risks were highest in adolescent and young adult males and after a second dose.
The study estimated an overall carditis incidence of 5.7 cases per million doses of Pfizer-BioNTech, for a risk 3.5 times that in the unvaccinated Hong Kong population. Carditis rates after a first dose were about 2.5 per million and 10 per million after a second dose.
Hong Kong launched its public SARS-CoV-2 immunization program in late February 2021 with the Chinese-made CoronaVac (Sinovac) inactivated-virus vaccine, and introduced the mRNA-based alternative several weeks later. By August 2021, the vaccines had reached about 3.3 million people in the region – 49% of the Hong Kong population at least 12 years of age.
In a novel finding, there were no excesses in carditis cases after CoronaVac vaccination. The difference between vaccines likely isn’t caused by chance, because three-fourths of the carditis-associated Pfizer-BioNTech injections arose within a week, whereas “71% of cases following the use of CoronaVac occurred more than 30 days after vaccination,” senior author Ian Chi Kei Wong, PhD, University of Hong Kong, said in an interview.
“This onset distribution for cases having received CoronaVac demonstrates that it is highly unlikely the carditis cases are related to the vaccine,” he said. And that “plausibly implies a specific underlying mechanism between vaccination and carditis that may only be applicable to mRNA vaccines.”
That inference is in line with case reports and other research, including large population-based studies from Israel and Denmark, although a recent study from the United Kingdom hinted at a potential excess myocarditis risk associated with the adenovirus-based AstraZeneca-Oxford vaccine.
The Hong Kong study identified 160 patients age 12 or older with a first diagnosis of carditis during February to August 2021, in electronic health records covering nearly the entire region.
“We used laboratory test results of troponin levels to further eliminate unlikely cases of carditis,” Dr. Wong said. The health records were linked to a “population-based vaccination record” maintained by the government’s department of health.
About 10 control patients from among all hospitalized patients without carditis were matched by age, sex, and admission date to each of the 160 carditis cases. About 83% of cases and 92% of the controls were unvaccinated.
Among those who received the Pfizer-BioNTech vaccine, representing 12.5% of cases and 4.2% of controls, the estimated carditis incidence was 0.57 per 100,000 doses. For those who received CoronaVac, representing 4.4% of cases and 3.9% of controls, it was 0.31 per 100,000 doses.
In adjusted analysis, the odds ratios for carditis among Pfizer-BioNTech vaccine recipients, compared with unvaccinated controls, were 3.57 (95% confidence interval, 1.93-6.60) overall, 4.68 (95% CI, 2.25-9.71) for males, 2.22 (95% CI, 0.57-8.69) for females, 2.41 (95% CI, 1.18-4.90) for ages 18 and older, and 13.8 (95% CI, 2.86-110.4) for ages 12-17
Myocarditis accounted for most of the excess cases, with an overall OR of 9.29 (95% CI, 3.94-21.9). The OR reached only 1.06 (95% CI, 0.35-3.22) for pericarditis alone.
The case-control study is noteworthy for its design, which contrasts with the many recent case series and passive or active surveillance studies, and even the more robust population-based studies of vaccine-related myocarditis, observed Dongngan Truong, MD, University of Utah and Primary Children’s Hospital, both in Salt Lake City, who wasn’t part of the study.
Among its strengths, she said in an interview, are its linkage of comprehensive hospital and vaccination data sets for two different vaccines; and that it corroborates other research suggesting there is “something in particular about mRNA vaccination that seems to be associated with the development of myocarditis.”
Active surveillance in Israel
In an October 2021 report based on an Israeli Ministry of Health database covering up to May 2021, rates of myocarditis arising within 21 days of a second Pfizer-BioNTech dose in 16- to 19-year-olds reached about 1 per 6,637 males and 1 per 99,853 females. Those numbers compared with 1 per 26,000 males and 1 per 218,000 females across all age groups.
Now authors led by Dror Mevorach, MD, Hadassah Medical Center, Jerusalem, have published corresponding numbers from the same data base for myocarditis associated with the same vaccine in males and females aged 12-15.
Their research covers 404,407 people in that age group who received a first dose of the mRNA-based vaccine and 326,463 who received the second dose from June to October, 2021. Only 18 cases of myocarditis were observed within 21 days of either dose.
The estimated rates for males were 0.56 cases per 100,000 after a first dose and 8.09 cases per 100,000 after a second dose.
For females, the estimates were 0 cases per 100,000 after a first dose and 0.69 cases per 100,000 after a second dose.
“The pattern observed, mainly following the second vaccination in males, suggests causality,” the group wrote.
Leveraging passive surveillance reports
Another new report adds a twist to updated numbers from the U.S. Vaccine Adverse Event Reporting System (VAERS).
Prevalences derived from the passive-surveillance data base, known for including case records of inconsistent quality or completeness, are considered especially prone to reporting bias, the authors acknowledged.
The current analysis, however, plunges deep into VAERS-reported cases of presumed SARS-CoV-2 vaccine-associated myocarditis to help clarify “more of the characteristics of the patients and some of the treatments and short-term outcomes,” Matthew E. Oster, MD, MPH, said in an interview.
Dr. Oster, from the Centers for Disease Control and Prevention and Emory University, Atlanta, is lead author on the study’s Jan. 25, 2022, publication in JAMA.
The group reviewed charts and interviewed involved clinicians to adjudicate and document presentations, therapies, and the clinical course of cases reported as SARS-CoV-2 vaccine–associated myocarditis from December 2020 to August 2021. Out of the nearly 2000 reports, which were limited to patients younger than 30, the group identified 1,626 likely cases of such myocarditis arising within 7 days of a second mRNA vaccine dose.
The confirmed cases consistently represented higher prevalences than expected compared with prepandemic myocarditis claims data for both sexes and across age groups spanning 12-29 years.
For example, rates were highest for adolescent males – about 106 and 71 cases per million second doses of the Pfizer-BioNTech vaccine in those aged 16-17 and 12-16, respectively, for example. They were lowest for women aged 25-29, at 2.23 cases per million second Pfizer-BioNTech doses; the highest rate among females was about 11 per million for the 16-17 age group.
The observed rates, Dr. Oster said, represent an update to VAERS numbers published June 2021 in Morbidity and Mortality Weekly Report covering cases through June 2021.
“Overall, the general risk of having myocarditis from the vaccines is still extremely low. Even in the highest risk groups, it is still extremely low, and still lower than the risk of having cardiac complications from COVID,” he noted.
How do patients fare clinically?
From their chart reviews and interviews with case clinicians, Dr. Oster said, “we started to learn quickly that this is really a different type of myocarditis.”
For example, its onset, typically within a few days of the potential immunologic cause, was more rapid than in viral myocarditis, and its symptoms resolved faster, the report notes. Clinical presentations tended to be less severe, treatments not as intensive, and outcomes not as serious, compared with “the kind of typical viral myocarditis that most of the providers were used to taking care of in the past,” he said. “The pattern for these cases was very consistent.”
The study covered VAERS reports of suspected myocarditis arising within a week of first dose of a mRNA-based vaccine from the United States launch of public vaccination in December 2020 to August 2021, the CDC-based group reported. By then, more than 192 million people in the country had received either the Pfizer-BioNTech (age 12 or older) or Moderna (age 18 or older) vaccines.
Of the 1,991 reports of myocarditis, including 391 also involving pericarditis, 1,626 met the study’s definition for myocarditis on adjudication; about 82% of the latter cases were in males.
Based on the investigators’ review of charts and clinician interviews connected with 826 cases that met their definition of myocarditis in patients younger than 30, 89% reported “chest pain, pressure, or discomfort” and 30% reported dyspnea or shortness of breath. Troponin levels were elevated in 98%, 72% of patients who underwent electrocardiography showed abnormalities, and 12% of those with echocardiography had left ventricular ejection fractions less than 50%.
About 96% were hospitalized, and presenting symptoms resolved by discharge in 87% of those with available data, the group noted. Among patients with data on in-hospital therapy, they wrote, NSAIDs were the most common therapy, in 87%.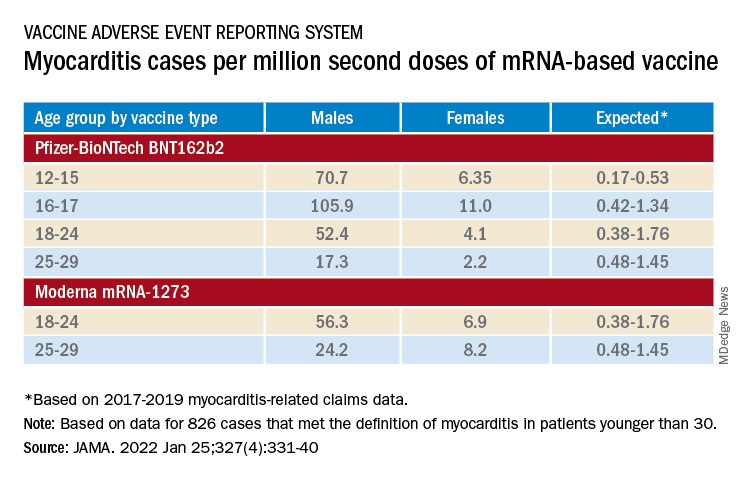
‘Mild and self-limiting’
The case-control study from Hong Kong didn’t specifically examine patients’ treatment and clinical course, but it does portray their vaccine-associated myocarditis as contrasting with more familiar viral myocarditis.
Patients with “typical” myocarditis tend to be “overall much sicker than what we’re seeing with myocarditis following vaccination,” Dr. Truong agreed. None of the 20 patients with myocarditis after Pfizer-BioNTech vaccination in Hong Kong were admitted to the intensive care unit. That, she added, suggests none required extracorporeal membrane oxygenation or vasoactive support, often necessary in viral myocarditis. “And they had shorter hospital stays.”
In contrast, Dr. Wong noted, 14 of the study’s unvaccinated patients required ICU admission; 12 of them died during the follow-up period. None with vaccine-related carditis died during the study’s follow-up. “We also showed that cases following [Pfizer-BioNTech] vaccination were all mild and self-limiting.”
Dr. Truong largely agreed that SARS-CoV-2 vaccine myocarditis and most myocarditis seen before the pandemic can be viewed as distinct clinical entities, “at least in the short term. I think we do need to follow these patients to look at more long-term outcomes, because at this point I don’t think we know the long-term implications. But at least in the short term, it seems like these patients are different, are much less sick, and recover pretty quickly overall.”
Dr. Oster emphasized that the many and varied acute and long-term hazards from contracting COVID-19 far outweigh any risk for myocarditis from vaccination. But for individuals who were hit with myocarditis soon after their first mRNA vaccine dose, who have already established their susceptibility, he and his colleagues would recommend that they “consider alternatives and not get the vaccine again.”
Dr. Oster reported no relevant financial relationships. Dr. Wong and colleagues did not report any relevant disclosures. Dr. Truong has previously disclosed serving as a consultant to Pfizer.
A version of this article first appeared on Medscape.com.
Why some COVID-19 vaccines seem occasionally to cause a distinctive form of myocarditis, and why adolescent boys and young men appear most vulnerable, remain a mystery. But the entity’s prevalence, nuances of presentation, and likely clinical course have come into sharper view after recent additions to the literature.
Two new publications all but confirm that the rare cases of myocarditis closely following vaccination against SARS-CoV-2, primarily with one of the mRNA-based vaccines from Pfizer-BioNTech and Moderna, is a clinically different creature from myocarditis physicians were likely to see before the pandemic.
A third report unveils rates of hospitalization for myocarditis linked to Pfizer-BioNTech vaccination in the 12- to 15-year age group, based on active surveillance across Israel. Of note, the rates were lower than corresponding numbers among the country’s 16- to 19-year-olds published in late 2021 by the same authors.
No link with CoronaVac
A case-control study covering almost the entire population of Hong Kong from February to August 2021 confirms a slight but significant excess risk for myocarditis and, to a lesser degree, pericarditis, after injections of the Pfizer-BioNTech vaccine. As consistently reported from other studies, the risks were highest in adolescent and young adult males and after a second dose.
The study estimated an overall carditis incidence of 5.7 cases per million doses of Pfizer-BioNTech, for a risk 3.5 times that in the unvaccinated Hong Kong population. Carditis rates after a first dose were about 2.5 per million and 10 per million after a second dose.
Hong Kong launched its public SARS-CoV-2 immunization program in late February 2021 with the Chinese-made CoronaVac (Sinovac) inactivated-virus vaccine, and introduced the mRNA-based alternative several weeks later. By August 2021, the vaccines had reached about 3.3 million people in the region – 49% of the Hong Kong population at least 12 years of age.
In a novel finding, there were no excesses in carditis cases after CoronaVac vaccination. The difference between vaccines likely isn’t caused by chance, because three-fourths of the carditis-associated Pfizer-BioNTech injections arose within a week, whereas “71% of cases following the use of CoronaVac occurred more than 30 days after vaccination,” senior author Ian Chi Kei Wong, PhD, University of Hong Kong, said in an interview.
“This onset distribution for cases having received CoronaVac demonstrates that it is highly unlikely the carditis cases are related to the vaccine,” he said. And that “plausibly implies a specific underlying mechanism between vaccination and carditis that may only be applicable to mRNA vaccines.”
That inference is in line with case reports and other research, including large population-based studies from Israel and Denmark, although a recent study from the United Kingdom hinted at a potential excess myocarditis risk associated with the adenovirus-based AstraZeneca-Oxford vaccine.
The Hong Kong study identified 160 patients age 12 or older with a first diagnosis of carditis during February to August 2021, in electronic health records covering nearly the entire region.
“We used laboratory test results of troponin levels to further eliminate unlikely cases of carditis,” Dr. Wong said. The health records were linked to a “population-based vaccination record” maintained by the government’s department of health.
About 10 control patients from among all hospitalized patients without carditis were matched by age, sex, and admission date to each of the 160 carditis cases. About 83% of cases and 92% of the controls were unvaccinated.
Among those who received the Pfizer-BioNTech vaccine, representing 12.5% of cases and 4.2% of controls, the estimated carditis incidence was 0.57 per 100,000 doses. For those who received CoronaVac, representing 4.4% of cases and 3.9% of controls, it was 0.31 per 100,000 doses.
In adjusted analysis, the odds ratios for carditis among Pfizer-BioNTech vaccine recipients, compared with unvaccinated controls, were 3.57 (95% confidence interval, 1.93-6.60) overall, 4.68 (95% CI, 2.25-9.71) for males, 2.22 (95% CI, 0.57-8.69) for females, 2.41 (95% CI, 1.18-4.90) for ages 18 and older, and 13.8 (95% CI, 2.86-110.4) for ages 12-17
Myocarditis accounted for most of the excess cases, with an overall OR of 9.29 (95% CI, 3.94-21.9). The OR reached only 1.06 (95% CI, 0.35-3.22) for pericarditis alone.
The case-control study is noteworthy for its design, which contrasts with the many recent case series and passive or active surveillance studies, and even the more robust population-based studies of vaccine-related myocarditis, observed Dongngan Truong, MD, University of Utah and Primary Children’s Hospital, both in Salt Lake City, who wasn’t part of the study.
Among its strengths, she said in an interview, are its linkage of comprehensive hospital and vaccination data sets for two different vaccines; and that it corroborates other research suggesting there is “something in particular about mRNA vaccination that seems to be associated with the development of myocarditis.”
Active surveillance in Israel
In an October 2021 report based on an Israeli Ministry of Health database covering up to May 2021, rates of myocarditis arising within 21 days of a second Pfizer-BioNTech dose in 16- to 19-year-olds reached about 1 per 6,637 males and 1 per 99,853 females. Those numbers compared with 1 per 26,000 males and 1 per 218,000 females across all age groups.
Now authors led by Dror Mevorach, MD, Hadassah Medical Center, Jerusalem, have published corresponding numbers from the same data base for myocarditis associated with the same vaccine in males and females aged 12-15.
Their research covers 404,407 people in that age group who received a first dose of the mRNA-based vaccine and 326,463 who received the second dose from June to October, 2021. Only 18 cases of myocarditis were observed within 21 days of either dose.
The estimated rates for males were 0.56 cases per 100,000 after a first dose and 8.09 cases per 100,000 after a second dose.
For females, the estimates were 0 cases per 100,000 after a first dose and 0.69 cases per 100,000 after a second dose.
“The pattern observed, mainly following the second vaccination in males, suggests causality,” the group wrote.
Leveraging passive surveillance reports
Another new report adds a twist to updated numbers from the U.S. Vaccine Adverse Event Reporting System (VAERS).
Prevalences derived from the passive-surveillance data base, known for including case records of inconsistent quality or completeness, are considered especially prone to reporting bias, the authors acknowledged.
The current analysis, however, plunges deep into VAERS-reported cases of presumed SARS-CoV-2 vaccine-associated myocarditis to help clarify “more of the characteristics of the patients and some of the treatments and short-term outcomes,” Matthew E. Oster, MD, MPH, said in an interview.
Dr. Oster, from the Centers for Disease Control and Prevention and Emory University, Atlanta, is lead author on the study’s Jan. 25, 2022, publication in JAMA.
The group reviewed charts and interviewed involved clinicians to adjudicate and document presentations, therapies, and the clinical course of cases reported as SARS-CoV-2 vaccine–associated myocarditis from December 2020 to August 2021. Out of the nearly 2000 reports, which were limited to patients younger than 30, the group identified 1,626 likely cases of such myocarditis arising within 7 days of a second mRNA vaccine dose.
The confirmed cases consistently represented higher prevalences than expected compared with prepandemic myocarditis claims data for both sexes and across age groups spanning 12-29 years.
For example, rates were highest for adolescent males – about 106 and 71 cases per million second doses of the Pfizer-BioNTech vaccine in those aged 16-17 and 12-16, respectively, for example. They were lowest for women aged 25-29, at 2.23 cases per million second Pfizer-BioNTech doses; the highest rate among females was about 11 per million for the 16-17 age group.
The observed rates, Dr. Oster said, represent an update to VAERS numbers published June 2021 in Morbidity and Mortality Weekly Report covering cases through June 2021.
“Overall, the general risk of having myocarditis from the vaccines is still extremely low. Even in the highest risk groups, it is still extremely low, and still lower than the risk of having cardiac complications from COVID,” he noted.
How do patients fare clinically?
From their chart reviews and interviews with case clinicians, Dr. Oster said, “we started to learn quickly that this is really a different type of myocarditis.”
For example, its onset, typically within a few days of the potential immunologic cause, was more rapid than in viral myocarditis, and its symptoms resolved faster, the report notes. Clinical presentations tended to be less severe, treatments not as intensive, and outcomes not as serious, compared with “the kind of typical viral myocarditis that most of the providers were used to taking care of in the past,” he said. “The pattern for these cases was very consistent.”
The study covered VAERS reports of suspected myocarditis arising within a week of first dose of a mRNA-based vaccine from the United States launch of public vaccination in December 2020 to August 2021, the CDC-based group reported. By then, more than 192 million people in the country had received either the Pfizer-BioNTech (age 12 or older) or Moderna (age 18 or older) vaccines.
Of the 1,991 reports of myocarditis, including 391 also involving pericarditis, 1,626 met the study’s definition for myocarditis on adjudication; about 82% of the latter cases were in males.
Based on the investigators’ review of charts and clinician interviews connected with 826 cases that met their definition of myocarditis in patients younger than 30, 89% reported “chest pain, pressure, or discomfort” and 30% reported dyspnea or shortness of breath. Troponin levels were elevated in 98%, 72% of patients who underwent electrocardiography showed abnormalities, and 12% of those with echocardiography had left ventricular ejection fractions less than 50%.
About 96% were hospitalized, and presenting symptoms resolved by discharge in 87% of those with available data, the group noted. Among patients with data on in-hospital therapy, they wrote, NSAIDs were the most common therapy, in 87%.
‘Mild and self-limiting’
The case-control study from Hong Kong didn’t specifically examine patients’ treatment and clinical course, but it does portray their vaccine-associated myocarditis as contrasting with more familiar viral myocarditis.
Patients with “typical” myocarditis tend to be “overall much sicker than what we’re seeing with myocarditis following vaccination,” Dr. Truong agreed. None of the 20 patients with myocarditis after Pfizer-BioNTech vaccination in Hong Kong were admitted to the intensive care unit. That, she added, suggests none required extracorporeal membrane oxygenation or vasoactive support, often necessary in viral myocarditis. “And they had shorter hospital stays.”
In contrast, Dr. Wong noted, 14 of the study’s unvaccinated patients required ICU admission; 12 of them died during the follow-up period. None with vaccine-related carditis died during the study’s follow-up. “We also showed that cases following [Pfizer-BioNTech] vaccination were all mild and self-limiting.”
Dr. Truong largely agreed that SARS-CoV-2 vaccine myocarditis and most myocarditis seen before the pandemic can be viewed as distinct clinical entities, “at least in the short term. I think we do need to follow these patients to look at more long-term outcomes, because at this point I don’t think we know the long-term implications. But at least in the short term, it seems like these patients are different, are much less sick, and recover pretty quickly overall.”
Dr. Oster emphasized that the many and varied acute and long-term hazards from contracting COVID-19 far outweigh any risk for myocarditis from vaccination. But for individuals who were hit with myocarditis soon after their first mRNA vaccine dose, who have already established their susceptibility, he and his colleagues would recommend that they “consider alternatives and not get the vaccine again.”
Dr. Oster reported no relevant financial relationships. Dr. Wong and colleagues did not report any relevant disclosures. Dr. Truong has previously disclosed serving as a consultant to Pfizer.
A version of this article first appeared on Medscape.com.
Why some COVID-19 vaccines seem occasionally to cause a distinctive form of myocarditis, and why adolescent boys and young men appear most vulnerable, remain a mystery. But the entity’s prevalence, nuances of presentation, and likely clinical course have come into sharper view after recent additions to the literature.
Two new publications all but confirm that the rare cases of myocarditis closely following vaccination against SARS-CoV-2, primarily with one of the mRNA-based vaccines from Pfizer-BioNTech and Moderna, is a clinically different creature from myocarditis physicians were likely to see before the pandemic.
A third report unveils rates of hospitalization for myocarditis linked to Pfizer-BioNTech vaccination in the 12- to 15-year age group, based on active surveillance across Israel. Of note, the rates were lower than corresponding numbers among the country’s 16- to 19-year-olds published in late 2021 by the same authors.
No link with CoronaVac
A case-control study covering almost the entire population of Hong Kong from February to August 2021 confirms a slight but significant excess risk for myocarditis and, to a lesser degree, pericarditis, after injections of the Pfizer-BioNTech vaccine. As consistently reported from other studies, the risks were highest in adolescent and young adult males and after a second dose.
The study estimated an overall carditis incidence of 5.7 cases per million doses of Pfizer-BioNTech, for a risk 3.5 times that in the unvaccinated Hong Kong population. Carditis rates after a first dose were about 2.5 per million and 10 per million after a second dose.
Hong Kong launched its public SARS-CoV-2 immunization program in late February 2021 with the Chinese-made CoronaVac (Sinovac) inactivated-virus vaccine, and introduced the mRNA-based alternative several weeks later. By August 2021, the vaccines had reached about 3.3 million people in the region – 49% of the Hong Kong population at least 12 years of age.
In a novel finding, there were no excesses in carditis cases after CoronaVac vaccination. The difference between vaccines likely isn’t caused by chance, because three-fourths of the carditis-associated Pfizer-BioNTech injections arose within a week, whereas “71% of cases following the use of CoronaVac occurred more than 30 days after vaccination,” senior author Ian Chi Kei Wong, PhD, University of Hong Kong, said in an interview.
“This onset distribution for cases having received CoronaVac demonstrates that it is highly unlikely the carditis cases are related to the vaccine,” he said. And that “plausibly implies a specific underlying mechanism between vaccination and carditis that may only be applicable to mRNA vaccines.”
That inference is in line with case reports and other research, including large population-based studies from Israel and Denmark, although a recent study from the United Kingdom hinted at a potential excess myocarditis risk associated with the adenovirus-based AstraZeneca-Oxford vaccine.
The Hong Kong study identified 160 patients age 12 or older with a first diagnosis of carditis during February to August 2021, in electronic health records covering nearly the entire region.
“We used laboratory test results of troponin levels to further eliminate unlikely cases of carditis,” Dr. Wong said. The health records were linked to a “population-based vaccination record” maintained by the government’s department of health.
About 10 control patients from among all hospitalized patients without carditis were matched by age, sex, and admission date to each of the 160 carditis cases. About 83% of cases and 92% of the controls were unvaccinated.
Among those who received the Pfizer-BioNTech vaccine, representing 12.5% of cases and 4.2% of controls, the estimated carditis incidence was 0.57 per 100,000 doses. For those who received CoronaVac, representing 4.4% of cases and 3.9% of controls, it was 0.31 per 100,000 doses.
In adjusted analysis, the odds ratios for carditis among Pfizer-BioNTech vaccine recipients, compared with unvaccinated controls, were 3.57 (95% confidence interval, 1.93-6.60) overall, 4.68 (95% CI, 2.25-9.71) for males, 2.22 (95% CI, 0.57-8.69) for females, 2.41 (95% CI, 1.18-4.90) for ages 18 and older, and 13.8 (95% CI, 2.86-110.4) for ages 12-17
Myocarditis accounted for most of the excess cases, with an overall OR of 9.29 (95% CI, 3.94-21.9). The OR reached only 1.06 (95% CI, 0.35-3.22) for pericarditis alone.
The case-control study is noteworthy for its design, which contrasts with the many recent case series and passive or active surveillance studies, and even the more robust population-based studies of vaccine-related myocarditis, observed Dongngan Truong, MD, University of Utah and Primary Children’s Hospital, both in Salt Lake City, who wasn’t part of the study.
Among its strengths, she said in an interview, are its linkage of comprehensive hospital and vaccination data sets for two different vaccines; and that it corroborates other research suggesting there is “something in particular about mRNA vaccination that seems to be associated with the development of myocarditis.”
Active surveillance in Israel
In an October 2021 report based on an Israeli Ministry of Health database covering up to May 2021, rates of myocarditis arising within 21 days of a second Pfizer-BioNTech dose in 16- to 19-year-olds reached about 1 per 6,637 males and 1 per 99,853 females. Those numbers compared with 1 per 26,000 males and 1 per 218,000 females across all age groups.
Now authors led by Dror Mevorach, MD, Hadassah Medical Center, Jerusalem, have published corresponding numbers from the same data base for myocarditis associated with the same vaccine in males and females aged 12-15.
Their research covers 404,407 people in that age group who received a first dose of the mRNA-based vaccine and 326,463 who received the second dose from June to October, 2021. Only 18 cases of myocarditis were observed within 21 days of either dose.
The estimated rates for males were 0.56 cases per 100,000 after a first dose and 8.09 cases per 100,000 after a second dose.
For females, the estimates were 0 cases per 100,000 after a first dose and 0.69 cases per 100,000 after a second dose.
“The pattern observed, mainly following the second vaccination in males, suggests causality,” the group wrote.
Leveraging passive surveillance reports
Another new report adds a twist to updated numbers from the U.S. Vaccine Adverse Event Reporting System (VAERS).
Prevalences derived from the passive-surveillance data base, known for including case records of inconsistent quality or completeness, are considered especially prone to reporting bias, the authors acknowledged.
The current analysis, however, plunges deep into VAERS-reported cases of presumed SARS-CoV-2 vaccine-associated myocarditis to help clarify “more of the characteristics of the patients and some of the treatments and short-term outcomes,” Matthew E. Oster, MD, MPH, said in an interview.
Dr. Oster, from the Centers for Disease Control and Prevention and Emory University, Atlanta, is lead author on the study’s Jan. 25, 2022, publication in JAMA.
The group reviewed charts and interviewed involved clinicians to adjudicate and document presentations, therapies, and the clinical course of cases reported as SARS-CoV-2 vaccine–associated myocarditis from December 2020 to August 2021. Out of the nearly 2000 reports, which were limited to patients younger than 30, the group identified 1,626 likely cases of such myocarditis arising within 7 days of a second mRNA vaccine dose.
The confirmed cases consistently represented higher prevalences than expected compared with prepandemic myocarditis claims data for both sexes and across age groups spanning 12-29 years.
For example, rates were highest for adolescent males – about 106 and 71 cases per million second doses of the Pfizer-BioNTech vaccine in those aged 16-17 and 12-16, respectively, for example. They were lowest for women aged 25-29, at 2.23 cases per million second Pfizer-BioNTech doses; the highest rate among females was about 11 per million for the 16-17 age group.
The observed rates, Dr. Oster said, represent an update to VAERS numbers published June 2021 in Morbidity and Mortality Weekly Report covering cases through June 2021.
“Overall, the general risk of having myocarditis from the vaccines is still extremely low. Even in the highest risk groups, it is still extremely low, and still lower than the risk of having cardiac complications from COVID,” he noted.
How do patients fare clinically?
From their chart reviews and interviews with case clinicians, Dr. Oster said, “we started to learn quickly that this is really a different type of myocarditis.”
For example, its onset, typically within a few days of the potential immunologic cause, was more rapid than in viral myocarditis, and its symptoms resolved faster, the report notes. Clinical presentations tended to be less severe, treatments not as intensive, and outcomes not as serious, compared with “the kind of typical viral myocarditis that most of the providers were used to taking care of in the past,” he said. “The pattern for these cases was very consistent.”
The study covered VAERS reports of suspected myocarditis arising within a week of first dose of a mRNA-based vaccine from the United States launch of public vaccination in December 2020 to August 2021, the CDC-based group reported. By then, more than 192 million people in the country had received either the Pfizer-BioNTech (age 12 or older) or Moderna (age 18 or older) vaccines.
Of the 1,991 reports of myocarditis, including 391 also involving pericarditis, 1,626 met the study’s definition for myocarditis on adjudication; about 82% of the latter cases were in males.
Based on the investigators’ review of charts and clinician interviews connected with 826 cases that met their definition of myocarditis in patients younger than 30, 89% reported “chest pain, pressure, or discomfort” and 30% reported dyspnea or shortness of breath. Troponin levels were elevated in 98%, 72% of patients who underwent electrocardiography showed abnormalities, and 12% of those with echocardiography had left ventricular ejection fractions less than 50%.
About 96% were hospitalized, and presenting symptoms resolved by discharge in 87% of those with available data, the group noted. Among patients with data on in-hospital therapy, they wrote, NSAIDs were the most common therapy, in 87%.
‘Mild and self-limiting’
The case-control study from Hong Kong didn’t specifically examine patients’ treatment and clinical course, but it does portray their vaccine-associated myocarditis as contrasting with more familiar viral myocarditis.
Patients with “typical” myocarditis tend to be “overall much sicker than what we’re seeing with myocarditis following vaccination,” Dr. Truong agreed. None of the 20 patients with myocarditis after Pfizer-BioNTech vaccination in Hong Kong were admitted to the intensive care unit. That, she added, suggests none required extracorporeal membrane oxygenation or vasoactive support, often necessary in viral myocarditis. “And they had shorter hospital stays.”
In contrast, Dr. Wong noted, 14 of the study’s unvaccinated patients required ICU admission; 12 of them died during the follow-up period. None with vaccine-related carditis died during the study’s follow-up. “We also showed that cases following [Pfizer-BioNTech] vaccination were all mild and self-limiting.”
Dr. Truong largely agreed that SARS-CoV-2 vaccine myocarditis and most myocarditis seen before the pandemic can be viewed as distinct clinical entities, “at least in the short term. I think we do need to follow these patients to look at more long-term outcomes, because at this point I don’t think we know the long-term implications. But at least in the short term, it seems like these patients are different, are much less sick, and recover pretty quickly overall.”
Dr. Oster emphasized that the many and varied acute and long-term hazards from contracting COVID-19 far outweigh any risk for myocarditis from vaccination. But for individuals who were hit with myocarditis soon after their first mRNA vaccine dose, who have already established their susceptibility, he and his colleagues would recommend that they “consider alternatives and not get the vaccine again.”
Dr. Oster reported no relevant financial relationships. Dr. Wong and colleagues did not report any relevant disclosures. Dr. Truong has previously disclosed serving as a consultant to Pfizer.
A version of this article first appeared on Medscape.com.



