User login
AACE issues ‘cookbook’ algorithm to manage dyslipidemia
A new algorithm on lipid management and prevention of cardiovascular disease from the American Association of Clinical Endocrinologists* (AACE) and the American College of Endocrinology (ACE) is “a nice cookbook” that many clinicians, especially those who are not lipid experts, will find useful, according to writing committee chair Yehuda Handelsman, MD.
The algorithm, published Oct. 10 in Endocrine Practice as 10 slides, or as part of a more detailed consensus statement, is a companion to the 2017 AACE/ACE guidelines for lipid management and includes more recent information about new therapies.
“What we’re trying to do here is to say, ‘focus on LDL-C, triglycerides, high-risk patients, and lifestyle. Understand all the medications available to you to reduce LDL-C and reduce triglycerides,’ ” Dr. Handelsman, of the Metabolic Institute of America, Tarzana, Calif., explained in an interview.
“We touch on lipoprotein(a), which we still don’t have medication for, but it identifies people at high risk, and we need that.”
Clinicians also need to know “that we’ve got some newer drugs in the market that can manage people who have statin intolerance,” Dr. Handelsman added.
“We introduced new therapies like icosapent ethyl” (Vascepa, Amarin) for hypertriglyceridemia, “when to use it, and how to use it. Even though it was not part of the 2017 guideline, we gave recommendations based on current data in the algorithm.”
Although there is no good evidence that lowering triglycerides reduces heart disease, he continued, many experts believe that the target triglyceride level should be less than 150 mg/dL, and the algorithm explains how to treat to this goal.
“Last, and most importantly, I cannot fail to underscore the fact that lifestyle is very important,” he emphasized.
Robert H. Eckel, MD, of the University of Colorado at Denver, Aurora, and president of medicine and science at the American Diabetes Association, who was not involved with this algorithm, said in an interview that the algorithm is important since it offers “the clinician or health care practitioner an approach, a kind of a cookbook or application of the guidelines, for how to manage lipid disorders in patients at risk ... It’s geared for the nonexperts too,” he said.
Dyslipidemia treatment summarized in 10 slides
The AACE/ACE algorithm comprises 10 slides, one each for dyslipidemic states, secondary causes of lipid disorders, screening for and assessing lipid disorders and atherosclerotic CVD (ASCVD) risk, ASCVD risk categories and treatment goals, lifestyle recommendations, treating LDL-C to goal, managing statin intolerance and safety, management of hypertriglyceridemia and the role of icosapent ethyl, assessment and management of elevated lipoprotein(a), and profiles of medications for dyslipidemia.
The algorithm defines five ASCVD risk categories and recommends increasingly lower LDL-C, non–HDL-C, and apo B target levels with increasing risk, but the same triglyceride target for all.
First, “treatment of lipid disorders begins with lifestyle therapy to improve nutrition, physical activity, weight, and other factors that affect lipids,” the consensus statement authors stress.
Next, “LDL-C has been, and remains, the main focus of efforts to improve lipid profiles in individuals at risk for ASCVD” (see table).
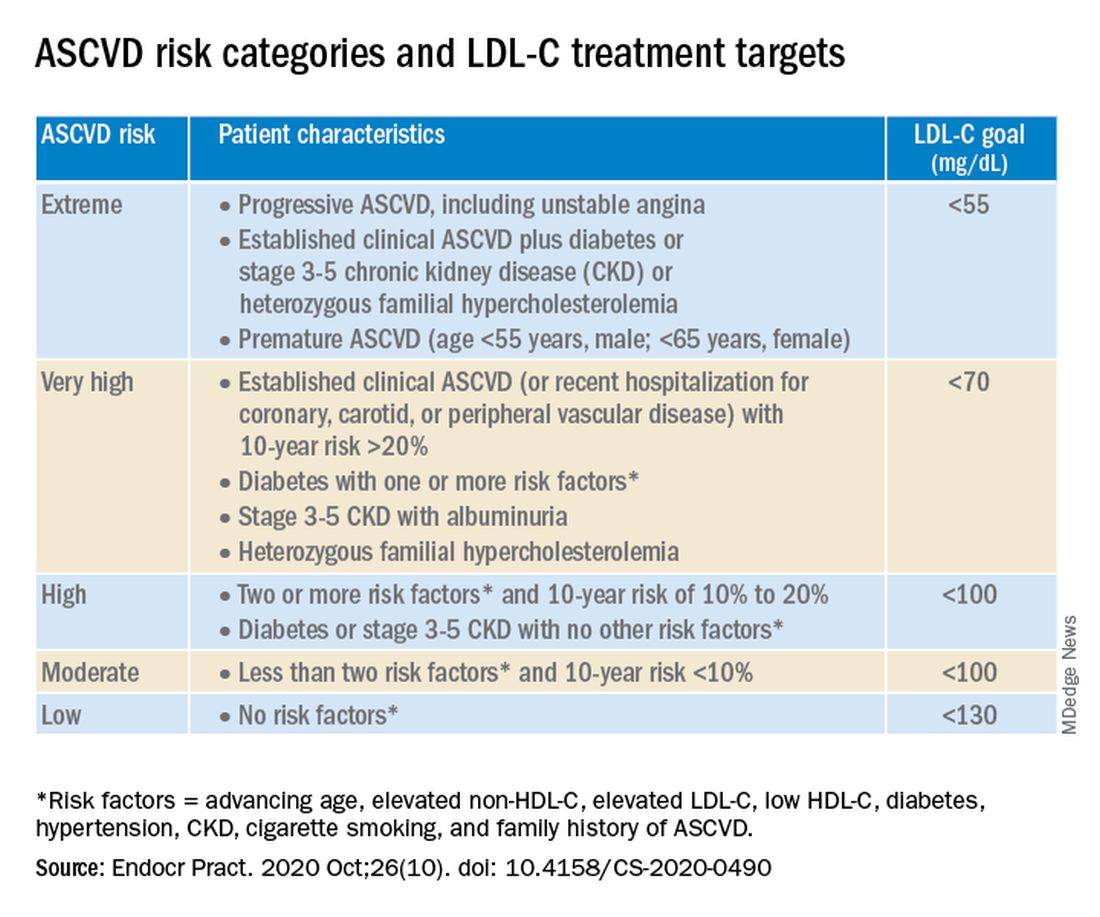
“We stratify [LDL-C] not as a one-treatment-target-for-all,” but rather as extreme, very high, high, moderate, and low ASCVD risk, Dr. Handelsman explained, with different treatment pathways (specified in another slide) to reach different risk-dependent goals.
“Unlike the ACC [American College of Cardiology] guideline, which shows if you want to further reduce LDL after statin give ezetimibe first, we say ‘no’,” he noted. “If somebody has an extreme risk, and you don’t think ezetimibe will get to a goal below 55 mg/dL, you should go first with a PCSK9 [proprotein convertase subtilisin/kexin type 9] inhibitor, and only then add ezetimibe or [colesevelam] or other drugs,” he said.
The consensus statement authors expand on this scenario. “Treatment for patients at extreme risk should begin with lifestyle therapy plus a high-intensity statin (atorvastatin 40 to 80 mg or rosuvastatin 20 to 40 mg, or the highest tolerated statin dose) to achieve an LDL-C goal of less than 55 mg/dL.”
“If LDL-C remains above goal after 3 months,” a PCSK9 inhibitor (evolocumab [Repatha, Amgen] or alirocumab [Praluent, Sanofi/Regeneron]), the cholesterol absorption inhibitor ezetimibe, or the bile acid sequestrant colesevelam (Welchol, Daiichi Sankyo) or the adenosine triphosphate-citrate lyase (ACL) inhibitor bempedoic acid (Nexletol, Esperion) “should be added, depending on required LDL-C lowering, and a third agent should be added if the combination fails to achieve the goal.”
However, “because the cost of ezetimibe is low, it may be preferred over PCSK9 inhibitors as second-line therapy to achieve an LDL-C below 70 mg/dL for patients who require no more than 15%-20% further reduction to reach goals.”
For patients at moderate or high risk, lipid management should begin with a moderate-intensity statin and be increased to a high-intensity statin before adding a second lipid-lowering medication to reach an LDL-C below 100 mg/dL.
According to the consensus statement, the desirable goal for triglycerides is less than 150 mg/dL.
In all patients with triglyceride levels of at least 500 mg/dL, statin therapy should be combined with a fibrate, prescription-grade omega-3 fatty acid, and/or niacin to reduce triglycerides.
In any patient with established ASCVD or diabetes with at least 2 ASCVD risk factors and triglycerides of 135-499 mg/dL, icosapent ethyl should be added to a statin to prevent ASCVD events.
Statement aligns with major guidelines
In general, the 2017 AACE/ACE guidelines and algorithm are “pretty similar” to other guidelines such as the 2018 ACC/American Heart Association (AHA) guidelines for cholesterol management, the 2019 ACC/AHA guidelines for primary prevention of CVD, and the 2019 European Society of Cardiology/European Atherosclerosis Society (ESC/EAS) guidelines for the management of dyslipidemia, according to Dr. Eckel.
They have “all have now taken into consideration the evidence behind PCSK9 inhibitors,” he noted. “That’s important because those drugs have proven to be effective.”
Two differences, he pointed out, are that the 2019 ESC/EAS guidelines suggest that lipoprotein(a) measurement be considered at least once in every adult’s lifetime, and they recommend apo B analysis in people with high triglycerides but normal LDL (or no higher than 100 mg/dL), to identify additional risk.
*AACE changes its name, broadens focus
Shortly after its algorithm was published, AACE announced that it has a new organization name and brand, the American Association of Clinical Endocrinology, which “more clearly defines AACE as a community of individuals who work together to elevate the practice of clinical endocrinology,” according to an Oct. 20 statement.
The change is meant to acknowledge AACE’s “more modern, inclusive approach to endocrinology that supports multidisciplinary care teams – with endocrinologists leading the way.”
Along with the name change is a new global website. The statement notes that “health care professionals and community members can access all of the valuable clinical content such as guidelines, disease state networks and important education by visiting the pro portal in the top right corner of the site, or by going directly to pro.aace.com.”
Dr. Handelsman discloses that he receives research grant support from Amgen, Applied Therapeutics, AstraZeneca, BMS, Gan & Lee, Novo Nordisk, and Sanofi, and he is a consultant and/or speaker for Amarin, BI-Lilly, and Sanofi.
Dr. Eckel has received consultant/advisory board fees from Kowa, Novo Nordisk, and Provention Bio.
A new algorithm on lipid management and prevention of cardiovascular disease from the American Association of Clinical Endocrinologists* (AACE) and the American College of Endocrinology (ACE) is “a nice cookbook” that many clinicians, especially those who are not lipid experts, will find useful, according to writing committee chair Yehuda Handelsman, MD.
The algorithm, published Oct. 10 in Endocrine Practice as 10 slides, or as part of a more detailed consensus statement, is a companion to the 2017 AACE/ACE guidelines for lipid management and includes more recent information about new therapies.
“What we’re trying to do here is to say, ‘focus on LDL-C, triglycerides, high-risk patients, and lifestyle. Understand all the medications available to you to reduce LDL-C and reduce triglycerides,’ ” Dr. Handelsman, of the Metabolic Institute of America, Tarzana, Calif., explained in an interview.
“We touch on lipoprotein(a), which we still don’t have medication for, but it identifies people at high risk, and we need that.”
Clinicians also need to know “that we’ve got some newer drugs in the market that can manage people who have statin intolerance,” Dr. Handelsman added.
“We introduced new therapies like icosapent ethyl” (Vascepa, Amarin) for hypertriglyceridemia, “when to use it, and how to use it. Even though it was not part of the 2017 guideline, we gave recommendations based on current data in the algorithm.”
Although there is no good evidence that lowering triglycerides reduces heart disease, he continued, many experts believe that the target triglyceride level should be less than 150 mg/dL, and the algorithm explains how to treat to this goal.
“Last, and most importantly, I cannot fail to underscore the fact that lifestyle is very important,” he emphasized.
Robert H. Eckel, MD, of the University of Colorado at Denver, Aurora, and president of medicine and science at the American Diabetes Association, who was not involved with this algorithm, said in an interview that the algorithm is important since it offers “the clinician or health care practitioner an approach, a kind of a cookbook or application of the guidelines, for how to manage lipid disorders in patients at risk ... It’s geared for the nonexperts too,” he said.
Dyslipidemia treatment summarized in 10 slides
The AACE/ACE algorithm comprises 10 slides, one each for dyslipidemic states, secondary causes of lipid disorders, screening for and assessing lipid disorders and atherosclerotic CVD (ASCVD) risk, ASCVD risk categories and treatment goals, lifestyle recommendations, treating LDL-C to goal, managing statin intolerance and safety, management of hypertriglyceridemia and the role of icosapent ethyl, assessment and management of elevated lipoprotein(a), and profiles of medications for dyslipidemia.
The algorithm defines five ASCVD risk categories and recommends increasingly lower LDL-C, non–HDL-C, and apo B target levels with increasing risk, but the same triglyceride target for all.
First, “treatment of lipid disorders begins with lifestyle therapy to improve nutrition, physical activity, weight, and other factors that affect lipids,” the consensus statement authors stress.
Next, “LDL-C has been, and remains, the main focus of efforts to improve lipid profiles in individuals at risk for ASCVD” (see table).

“We stratify [LDL-C] not as a one-treatment-target-for-all,” but rather as extreme, very high, high, moderate, and low ASCVD risk, Dr. Handelsman explained, with different treatment pathways (specified in another slide) to reach different risk-dependent goals.
“Unlike the ACC [American College of Cardiology] guideline, which shows if you want to further reduce LDL after statin give ezetimibe first, we say ‘no’,” he noted. “If somebody has an extreme risk, and you don’t think ezetimibe will get to a goal below 55 mg/dL, you should go first with a PCSK9 [proprotein convertase subtilisin/kexin type 9] inhibitor, and only then add ezetimibe or [colesevelam] or other drugs,” he said.
The consensus statement authors expand on this scenario. “Treatment for patients at extreme risk should begin with lifestyle therapy plus a high-intensity statin (atorvastatin 40 to 80 mg or rosuvastatin 20 to 40 mg, or the highest tolerated statin dose) to achieve an LDL-C goal of less than 55 mg/dL.”
“If LDL-C remains above goal after 3 months,” a PCSK9 inhibitor (evolocumab [Repatha, Amgen] or alirocumab [Praluent, Sanofi/Regeneron]), the cholesterol absorption inhibitor ezetimibe, or the bile acid sequestrant colesevelam (Welchol, Daiichi Sankyo) or the adenosine triphosphate-citrate lyase (ACL) inhibitor bempedoic acid (Nexletol, Esperion) “should be added, depending on required LDL-C lowering, and a third agent should be added if the combination fails to achieve the goal.”
However, “because the cost of ezetimibe is low, it may be preferred over PCSK9 inhibitors as second-line therapy to achieve an LDL-C below 70 mg/dL for patients who require no more than 15%-20% further reduction to reach goals.”
For patients at moderate or high risk, lipid management should begin with a moderate-intensity statin and be increased to a high-intensity statin before adding a second lipid-lowering medication to reach an LDL-C below 100 mg/dL.
According to the consensus statement, the desirable goal for triglycerides is less than 150 mg/dL.
In all patients with triglyceride levels of at least 500 mg/dL, statin therapy should be combined with a fibrate, prescription-grade omega-3 fatty acid, and/or niacin to reduce triglycerides.
In any patient with established ASCVD or diabetes with at least 2 ASCVD risk factors and triglycerides of 135-499 mg/dL, icosapent ethyl should be added to a statin to prevent ASCVD events.
Statement aligns with major guidelines
In general, the 2017 AACE/ACE guidelines and algorithm are “pretty similar” to other guidelines such as the 2018 ACC/American Heart Association (AHA) guidelines for cholesterol management, the 2019 ACC/AHA guidelines for primary prevention of CVD, and the 2019 European Society of Cardiology/European Atherosclerosis Society (ESC/EAS) guidelines for the management of dyslipidemia, according to Dr. Eckel.
They have “all have now taken into consideration the evidence behind PCSK9 inhibitors,” he noted. “That’s important because those drugs have proven to be effective.”
Two differences, he pointed out, are that the 2019 ESC/EAS guidelines suggest that lipoprotein(a) measurement be considered at least once in every adult’s lifetime, and they recommend apo B analysis in people with high triglycerides but normal LDL (or no higher than 100 mg/dL), to identify additional risk.
*AACE changes its name, broadens focus
Shortly after its algorithm was published, AACE announced that it has a new organization name and brand, the American Association of Clinical Endocrinology, which “more clearly defines AACE as a community of individuals who work together to elevate the practice of clinical endocrinology,” according to an Oct. 20 statement.
The change is meant to acknowledge AACE’s “more modern, inclusive approach to endocrinology that supports multidisciplinary care teams – with endocrinologists leading the way.”
Along with the name change is a new global website. The statement notes that “health care professionals and community members can access all of the valuable clinical content such as guidelines, disease state networks and important education by visiting the pro portal in the top right corner of the site, or by going directly to pro.aace.com.”
Dr. Handelsman discloses that he receives research grant support from Amgen, Applied Therapeutics, AstraZeneca, BMS, Gan & Lee, Novo Nordisk, and Sanofi, and he is a consultant and/or speaker for Amarin, BI-Lilly, and Sanofi.
Dr. Eckel has received consultant/advisory board fees from Kowa, Novo Nordisk, and Provention Bio.
A new algorithm on lipid management and prevention of cardiovascular disease from the American Association of Clinical Endocrinologists* (AACE) and the American College of Endocrinology (ACE) is “a nice cookbook” that many clinicians, especially those who are not lipid experts, will find useful, according to writing committee chair Yehuda Handelsman, MD.
The algorithm, published Oct. 10 in Endocrine Practice as 10 slides, or as part of a more detailed consensus statement, is a companion to the 2017 AACE/ACE guidelines for lipid management and includes more recent information about new therapies.
“What we’re trying to do here is to say, ‘focus on LDL-C, triglycerides, high-risk patients, and lifestyle. Understand all the medications available to you to reduce LDL-C and reduce triglycerides,’ ” Dr. Handelsman, of the Metabolic Institute of America, Tarzana, Calif., explained in an interview.
“We touch on lipoprotein(a), which we still don’t have medication for, but it identifies people at high risk, and we need that.”
Clinicians also need to know “that we’ve got some newer drugs in the market that can manage people who have statin intolerance,” Dr. Handelsman added.
“We introduced new therapies like icosapent ethyl” (Vascepa, Amarin) for hypertriglyceridemia, “when to use it, and how to use it. Even though it was not part of the 2017 guideline, we gave recommendations based on current data in the algorithm.”
Although there is no good evidence that lowering triglycerides reduces heart disease, he continued, many experts believe that the target triglyceride level should be less than 150 mg/dL, and the algorithm explains how to treat to this goal.
“Last, and most importantly, I cannot fail to underscore the fact that lifestyle is very important,” he emphasized.
Robert H. Eckel, MD, of the University of Colorado at Denver, Aurora, and president of medicine and science at the American Diabetes Association, who was not involved with this algorithm, said in an interview that the algorithm is important since it offers “the clinician or health care practitioner an approach, a kind of a cookbook or application of the guidelines, for how to manage lipid disorders in patients at risk ... It’s geared for the nonexperts too,” he said.
Dyslipidemia treatment summarized in 10 slides
The AACE/ACE algorithm comprises 10 slides, one each for dyslipidemic states, secondary causes of lipid disorders, screening for and assessing lipid disorders and atherosclerotic CVD (ASCVD) risk, ASCVD risk categories and treatment goals, lifestyle recommendations, treating LDL-C to goal, managing statin intolerance and safety, management of hypertriglyceridemia and the role of icosapent ethyl, assessment and management of elevated lipoprotein(a), and profiles of medications for dyslipidemia.
The algorithm defines five ASCVD risk categories and recommends increasingly lower LDL-C, non–HDL-C, and apo B target levels with increasing risk, but the same triglyceride target for all.
First, “treatment of lipid disorders begins with lifestyle therapy to improve nutrition, physical activity, weight, and other factors that affect lipids,” the consensus statement authors stress.
Next, “LDL-C has been, and remains, the main focus of efforts to improve lipid profiles in individuals at risk for ASCVD” (see table).

“We stratify [LDL-C] not as a one-treatment-target-for-all,” but rather as extreme, very high, high, moderate, and low ASCVD risk, Dr. Handelsman explained, with different treatment pathways (specified in another slide) to reach different risk-dependent goals.
“Unlike the ACC [American College of Cardiology] guideline, which shows if you want to further reduce LDL after statin give ezetimibe first, we say ‘no’,” he noted. “If somebody has an extreme risk, and you don’t think ezetimibe will get to a goal below 55 mg/dL, you should go first with a PCSK9 [proprotein convertase subtilisin/kexin type 9] inhibitor, and only then add ezetimibe or [colesevelam] or other drugs,” he said.
The consensus statement authors expand on this scenario. “Treatment for patients at extreme risk should begin with lifestyle therapy plus a high-intensity statin (atorvastatin 40 to 80 mg or rosuvastatin 20 to 40 mg, or the highest tolerated statin dose) to achieve an LDL-C goal of less than 55 mg/dL.”
“If LDL-C remains above goal after 3 months,” a PCSK9 inhibitor (evolocumab [Repatha, Amgen] or alirocumab [Praluent, Sanofi/Regeneron]), the cholesterol absorption inhibitor ezetimibe, or the bile acid sequestrant colesevelam (Welchol, Daiichi Sankyo) or the adenosine triphosphate-citrate lyase (ACL) inhibitor bempedoic acid (Nexletol, Esperion) “should be added, depending on required LDL-C lowering, and a third agent should be added if the combination fails to achieve the goal.”
However, “because the cost of ezetimibe is low, it may be preferred over PCSK9 inhibitors as second-line therapy to achieve an LDL-C below 70 mg/dL for patients who require no more than 15%-20% further reduction to reach goals.”
For patients at moderate or high risk, lipid management should begin with a moderate-intensity statin and be increased to a high-intensity statin before adding a second lipid-lowering medication to reach an LDL-C below 100 mg/dL.
According to the consensus statement, the desirable goal for triglycerides is less than 150 mg/dL.
In all patients with triglyceride levels of at least 500 mg/dL, statin therapy should be combined with a fibrate, prescription-grade omega-3 fatty acid, and/or niacin to reduce triglycerides.
In any patient with established ASCVD or diabetes with at least 2 ASCVD risk factors and triglycerides of 135-499 mg/dL, icosapent ethyl should be added to a statin to prevent ASCVD events.
Statement aligns with major guidelines
In general, the 2017 AACE/ACE guidelines and algorithm are “pretty similar” to other guidelines such as the 2018 ACC/American Heart Association (AHA) guidelines for cholesterol management, the 2019 ACC/AHA guidelines for primary prevention of CVD, and the 2019 European Society of Cardiology/European Atherosclerosis Society (ESC/EAS) guidelines for the management of dyslipidemia, according to Dr. Eckel.
They have “all have now taken into consideration the evidence behind PCSK9 inhibitors,” he noted. “That’s important because those drugs have proven to be effective.”
Two differences, he pointed out, are that the 2019 ESC/EAS guidelines suggest that lipoprotein(a) measurement be considered at least once in every adult’s lifetime, and they recommend apo B analysis in people with high triglycerides but normal LDL (or no higher than 100 mg/dL), to identify additional risk.
*AACE changes its name, broadens focus
Shortly after its algorithm was published, AACE announced that it has a new organization name and brand, the American Association of Clinical Endocrinology, which “more clearly defines AACE as a community of individuals who work together to elevate the practice of clinical endocrinology,” according to an Oct. 20 statement.
The change is meant to acknowledge AACE’s “more modern, inclusive approach to endocrinology that supports multidisciplinary care teams – with endocrinologists leading the way.”
Along with the name change is a new global website. The statement notes that “health care professionals and community members can access all of the valuable clinical content such as guidelines, disease state networks and important education by visiting the pro portal in the top right corner of the site, or by going directly to pro.aace.com.”
Dr. Handelsman discloses that he receives research grant support from Amgen, Applied Therapeutics, AstraZeneca, BMS, Gan & Lee, Novo Nordisk, and Sanofi, and he is a consultant and/or speaker for Amarin, BI-Lilly, and Sanofi.
Dr. Eckel has received consultant/advisory board fees from Kowa, Novo Nordisk, and Provention Bio.
Artificially sweetened drinks add to CVD risk
Sugary and artificially sweetened drinks are each associated with an increased risk of developing cardiovascular disease, according to results from a large prospective cohort study.
However, the design of that study fails to take into account other sources of dietary sugar, according to one expert.
In a research letter published online Oct. 26 in the Journal of the American College of Cardiology, Eloi Chazelas, a PhD candidate at Sorbonne Paris Nord University in Paris, and colleagues, shared results from nearly 105,000 subjects (79% women, mean age 43 at baseline, median follow up 6.6 years) enrolled in the NutriNet-Santé cohort study.
In this observational study, which began recruiting in 2009, dietary patterns are self-reported by subjects, while health outcomes are validated by investigators.
Mr. Chazelas and his colleagues identified 1,379 first incident cases of stroke, transient ischemic attack, myocardial infarction, acute coronary syndrome, and angioplasty in the cohort during 2009-2019. Cases that occurred during the first 3 years’ follow up were excluded from the analysis, to avoid potential reverse causality bias.
After adjustment for a wide range of dietary, demographic and health confounders, the investigators found that high consumers of sugary drinks or artificially sweetened drinks saw 20% and 32% higher risk of such events, respectively, compared with people who reported drinking neither beverage type (hazard ratio: 1.20; 95% confidence interval 1.04-1.40, P for trend < .0009 and HR: 1.32; 95% CI, 1.00-1.73, P for trend < .03).
Sugary drinks were defined as containing 5% or more of sugars, including natural fruit juices. The high consumers in the study had a median intake of 185 mL per day of sugary drinks, or 176 mL per day for artificially sweetened drinks. Natural noncaloric sweeteners such as Stevia were included in the artificially sweetened group.
The findings, Mr. Chazelas and colleagues wrote in their analysis, add to evidence that artificially sweetened beverages “might not be a healthy substitute for sugary drinks.” While research has suggested that artificial sweeteners induce glucose intolerance by disturbing gut microbiota, they noted, more and bigger studies are needed to understand the mechanisms by which they might bear on cardiovascular disease risk.
Robert A. Vogel, MD, of the University of Colorado Denver, urged caution in interpreting the researchers’ results. In an interview, Dr. Vogel, a preventive cardiologist, said that it is “notoriously difficult” to evaluate what a food or food group does to the body outside of a carefully controlled trial. What little randomized trial evidence exists comparing the health effects of artificially sweetened and sugary drinks includes a 2012 trial in children that found diet drinks associated with reductions in body fat – if anything a positive indication for heart health.
With adults enrolled in an observational study, things are much more easily confounded, Dr. Vogel said. “So subjects self-report that they’re not consuming one thing – sugary or sweetened beverages. What else are they putting into their diet? Maybe they’re eating dessert and consuming sugar that way. Try as you will to unconfound, to do a multivariate correction for all these factors is just very difficult.”
In addition, Dr. Vogel noted, the investigators made no attempt to discern among the different sweeteners consumed. “Stevia, saccharine, Sucralose – it’s highly unlikely that each of these agents has the same effect on gut microbiota.”
In 2019, researchers led by Mr. Chazelas looked at cancer risk in high consumers of the sugary and artificially sweetened drinks in some 107,000 patients from the cohort, and reported that sugary drinks were significantly associated with the risk of overall cancer. They saw no similar association for artificially sweetened drinks.
The NutriNet-Santé study is funded by the French government, and the investigators disclosed no financial support from commercial entities. Dr. Vogel has received research support from Sanofi and speaking fees from Regeneron.
Sugary and artificially sweetened drinks are each associated with an increased risk of developing cardiovascular disease, according to results from a large prospective cohort study.
However, the design of that study fails to take into account other sources of dietary sugar, according to one expert.
In a research letter published online Oct. 26 in the Journal of the American College of Cardiology, Eloi Chazelas, a PhD candidate at Sorbonne Paris Nord University in Paris, and colleagues, shared results from nearly 105,000 subjects (79% women, mean age 43 at baseline, median follow up 6.6 years) enrolled in the NutriNet-Santé cohort study.
In this observational study, which began recruiting in 2009, dietary patterns are self-reported by subjects, while health outcomes are validated by investigators.
Mr. Chazelas and his colleagues identified 1,379 first incident cases of stroke, transient ischemic attack, myocardial infarction, acute coronary syndrome, and angioplasty in the cohort during 2009-2019. Cases that occurred during the first 3 years’ follow up were excluded from the analysis, to avoid potential reverse causality bias.
After adjustment for a wide range of dietary, demographic and health confounders, the investigators found that high consumers of sugary drinks or artificially sweetened drinks saw 20% and 32% higher risk of such events, respectively, compared with people who reported drinking neither beverage type (hazard ratio: 1.20; 95% confidence interval 1.04-1.40, P for trend < .0009 and HR: 1.32; 95% CI, 1.00-1.73, P for trend < .03).
Sugary drinks were defined as containing 5% or more of sugars, including natural fruit juices. The high consumers in the study had a median intake of 185 mL per day of sugary drinks, or 176 mL per day for artificially sweetened drinks. Natural noncaloric sweeteners such as Stevia were included in the artificially sweetened group.
The findings, Mr. Chazelas and colleagues wrote in their analysis, add to evidence that artificially sweetened beverages “might not be a healthy substitute for sugary drinks.” While research has suggested that artificial sweeteners induce glucose intolerance by disturbing gut microbiota, they noted, more and bigger studies are needed to understand the mechanisms by which they might bear on cardiovascular disease risk.
Robert A. Vogel, MD, of the University of Colorado Denver, urged caution in interpreting the researchers’ results. In an interview, Dr. Vogel, a preventive cardiologist, said that it is “notoriously difficult” to evaluate what a food or food group does to the body outside of a carefully controlled trial. What little randomized trial evidence exists comparing the health effects of artificially sweetened and sugary drinks includes a 2012 trial in children that found diet drinks associated with reductions in body fat – if anything a positive indication for heart health.
With adults enrolled in an observational study, things are much more easily confounded, Dr. Vogel said. “So subjects self-report that they’re not consuming one thing – sugary or sweetened beverages. What else are they putting into their diet? Maybe they’re eating dessert and consuming sugar that way. Try as you will to unconfound, to do a multivariate correction for all these factors is just very difficult.”
In addition, Dr. Vogel noted, the investigators made no attempt to discern among the different sweeteners consumed. “Stevia, saccharine, Sucralose – it’s highly unlikely that each of these agents has the same effect on gut microbiota.”
In 2019, researchers led by Mr. Chazelas looked at cancer risk in high consumers of the sugary and artificially sweetened drinks in some 107,000 patients from the cohort, and reported that sugary drinks were significantly associated with the risk of overall cancer. They saw no similar association for artificially sweetened drinks.
The NutriNet-Santé study is funded by the French government, and the investigators disclosed no financial support from commercial entities. Dr. Vogel has received research support from Sanofi and speaking fees from Regeneron.
Sugary and artificially sweetened drinks are each associated with an increased risk of developing cardiovascular disease, according to results from a large prospective cohort study.
However, the design of that study fails to take into account other sources of dietary sugar, according to one expert.
In a research letter published online Oct. 26 in the Journal of the American College of Cardiology, Eloi Chazelas, a PhD candidate at Sorbonne Paris Nord University in Paris, and colleagues, shared results from nearly 105,000 subjects (79% women, mean age 43 at baseline, median follow up 6.6 years) enrolled in the NutriNet-Santé cohort study.
In this observational study, which began recruiting in 2009, dietary patterns are self-reported by subjects, while health outcomes are validated by investigators.
Mr. Chazelas and his colleagues identified 1,379 first incident cases of stroke, transient ischemic attack, myocardial infarction, acute coronary syndrome, and angioplasty in the cohort during 2009-2019. Cases that occurred during the first 3 years’ follow up were excluded from the analysis, to avoid potential reverse causality bias.
After adjustment for a wide range of dietary, demographic and health confounders, the investigators found that high consumers of sugary drinks or artificially sweetened drinks saw 20% and 32% higher risk of such events, respectively, compared with people who reported drinking neither beverage type (hazard ratio: 1.20; 95% confidence interval 1.04-1.40, P for trend < .0009 and HR: 1.32; 95% CI, 1.00-1.73, P for trend < .03).
Sugary drinks were defined as containing 5% or more of sugars, including natural fruit juices. The high consumers in the study had a median intake of 185 mL per day of sugary drinks, or 176 mL per day for artificially sweetened drinks. Natural noncaloric sweeteners such as Stevia were included in the artificially sweetened group.
The findings, Mr. Chazelas and colleagues wrote in their analysis, add to evidence that artificially sweetened beverages “might not be a healthy substitute for sugary drinks.” While research has suggested that artificial sweeteners induce glucose intolerance by disturbing gut microbiota, they noted, more and bigger studies are needed to understand the mechanisms by which they might bear on cardiovascular disease risk.
Robert A. Vogel, MD, of the University of Colorado Denver, urged caution in interpreting the researchers’ results. In an interview, Dr. Vogel, a preventive cardiologist, said that it is “notoriously difficult” to evaluate what a food or food group does to the body outside of a carefully controlled trial. What little randomized trial evidence exists comparing the health effects of artificially sweetened and sugary drinks includes a 2012 trial in children that found diet drinks associated with reductions in body fat – if anything a positive indication for heart health.
With adults enrolled in an observational study, things are much more easily confounded, Dr. Vogel said. “So subjects self-report that they’re not consuming one thing – sugary or sweetened beverages. What else are they putting into their diet? Maybe they’re eating dessert and consuming sugar that way. Try as you will to unconfound, to do a multivariate correction for all these factors is just very difficult.”
In addition, Dr. Vogel noted, the investigators made no attempt to discern among the different sweeteners consumed. “Stevia, saccharine, Sucralose – it’s highly unlikely that each of these agents has the same effect on gut microbiota.”
In 2019, researchers led by Mr. Chazelas looked at cancer risk in high consumers of the sugary and artificially sweetened drinks in some 107,000 patients from the cohort, and reported that sugary drinks were significantly associated with the risk of overall cancer. They saw no similar association for artificially sweetened drinks.
The NutriNet-Santé study is funded by the French government, and the investigators disclosed no financial support from commercial entities. Dr. Vogel has received research support from Sanofi and speaking fees from Regeneron.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Bariatric surgery linked to longer life
A new analysis of the Swedish Obese Subjects (SOS) study shows that bariatric surgery is associated with about a 3-year increase in lifespan, compared with obese patients who do not undergo surgery. Still, surgery did not restore normal lifespan: Surgical patients’ lifespan remained less than that of a sample from the general Swedish population. The study follows other reports suggesting reduced mortality after bariatric surgery, but with a longer follow-up.
“These data add even more evidence to the growing literature showing that patients who undergo bariatric surgery experience a reduction in all-cause long-term mortality. In making decisions around bariatric surgical procedures and care, patients and their health care providers need to understand the trade-offs between improved weight, health, and longer-term survival versus the surgical risks and problems over time,” said Anita P. Courcoulas, MD, MPH, chief of minimally invasive bariatric and general surgery at the University of Pittsburgh Medical Center, said in an interview. Dr. Courcoulas was not involved in the study.
The results appeared in the New England Journal of Medicine.
The SOS study drew from 25 surgical departments and 480 primary health care centers in Sweden. The researchers examined data from 2,007 patients who underwent bariatric surgery between 1987 and 2001, and compared their outcomes to 2,040 matched controls. All were between age 37 and 60 years, with a body mass index (BMI) of at least 34 kg/m2 for men and 38 for women. They also compared outcomes with 1,135 randomly sampled from the Swedish population registry.
Procedures included banding (18%), vertical banded gastroplasty (69%), and gastric bypass (13%). After an initial BMI reduction of about 11, the surgery group stabilized by year 8 at a BMI about 7 lower than baseline, and there was little change in BMI among controls.
After a mean follow-up of 24 years (interquartile range, 22-27 years), there were 10.7 deaths per 1,000 person-years in the surgery group, 13.2 among obese controls, and 5.2 in the general population (hazard ratio, 0.77 for surgery versus no surgery; P < .001). The general population had a lower mortality than nonsurgical controls (HR, 0.44; P < .001).
The surgery group had a higher median life expectancy than controls (median, 2.4 years; adjusted difference, 3.0 years; P < .001). The general population group had a median life expectancy that was 7.4 years higher than the control group (adjusted difference, 8.5 years; P < .001). The surgery group’s median life expectancy was still shorter than the general population reference (adjusted difference, 5.5 years; P < .001).
Cardiovascular disease risk was lower in the surgery group (HR, 0.70; 95% confidence interval, 0.57-0.85), as was risk of MI (HR, 0.51; 95% CI, 0.33-0.79), heart failure (HR, 0.52; 95% CI, 0.31-0.88), and stroke (HR, 0.45; 95% CI, 0.24-0.84). Cancer mortality was also lower (HR, 0.77; 95% CI, 0.61-0.96).
In the surgery group, causes of death that were elevated over the general population included cardiovascular causes (HR, 2.64; 95% CI, 1.78-3.91) and noncardiovascular causes, mainly infections; postsurgical complications; and factors such as alcoholism, suicide, or trauma (HR, 1.50; 95% CI, 1.18-1.91).
The study is limited by its retrospective nature, and because the surgical techniques used at the time are less effective than those used today, and could lead to weight gain over time. As a result, many patients who underwent surgery remained heavier than the general population. It’s also possible that negative health effects accumulated before surgery and persisted afterwards, according to Dr. Courcoulas.
The findings are likely generalizable to people with obesity, many of whom choose not to undergo bariatric surgery despite the potential benefits. “The population studied in SOS had a similar profile of underlying medical diseases to those groups who undergo bariatric surgery today and in the U.S. and around the world,” said Dr. Courcoulas.
The study was funded by the Swedish Research Council and others. Dr. Courcoulas has no relevant financial disclosures
SOURCE: Carlsson L et al. N Engl J Med. 2020 Oct 15. doi: 10.1056/NEJMoa2002449.
A new analysis of the Swedish Obese Subjects (SOS) study shows that bariatric surgery is associated with about a 3-year increase in lifespan, compared with obese patients who do not undergo surgery. Still, surgery did not restore normal lifespan: Surgical patients’ lifespan remained less than that of a sample from the general Swedish population. The study follows other reports suggesting reduced mortality after bariatric surgery, but with a longer follow-up.
“These data add even more evidence to the growing literature showing that patients who undergo bariatric surgery experience a reduction in all-cause long-term mortality. In making decisions around bariatric surgical procedures and care, patients and their health care providers need to understand the trade-offs between improved weight, health, and longer-term survival versus the surgical risks and problems over time,” said Anita P. Courcoulas, MD, MPH, chief of minimally invasive bariatric and general surgery at the University of Pittsburgh Medical Center, said in an interview. Dr. Courcoulas was not involved in the study.
The results appeared in the New England Journal of Medicine.
The SOS study drew from 25 surgical departments and 480 primary health care centers in Sweden. The researchers examined data from 2,007 patients who underwent bariatric surgery between 1987 and 2001, and compared their outcomes to 2,040 matched controls. All were between age 37 and 60 years, with a body mass index (BMI) of at least 34 kg/m2 for men and 38 for women. They also compared outcomes with 1,135 randomly sampled from the Swedish population registry.
Procedures included banding (18%), vertical banded gastroplasty (69%), and gastric bypass (13%). After an initial BMI reduction of about 11, the surgery group stabilized by year 8 at a BMI about 7 lower than baseline, and there was little change in BMI among controls.
After a mean follow-up of 24 years (interquartile range, 22-27 years), there were 10.7 deaths per 1,000 person-years in the surgery group, 13.2 among obese controls, and 5.2 in the general population (hazard ratio, 0.77 for surgery versus no surgery; P < .001). The general population had a lower mortality than nonsurgical controls (HR, 0.44; P < .001).
The surgery group had a higher median life expectancy than controls (median, 2.4 years; adjusted difference, 3.0 years; P < .001). The general population group had a median life expectancy that was 7.4 years higher than the control group (adjusted difference, 8.5 years; P < .001). The surgery group’s median life expectancy was still shorter than the general population reference (adjusted difference, 5.5 years; P < .001).
Cardiovascular disease risk was lower in the surgery group (HR, 0.70; 95% confidence interval, 0.57-0.85), as was risk of MI (HR, 0.51; 95% CI, 0.33-0.79), heart failure (HR, 0.52; 95% CI, 0.31-0.88), and stroke (HR, 0.45; 95% CI, 0.24-0.84). Cancer mortality was also lower (HR, 0.77; 95% CI, 0.61-0.96).
In the surgery group, causes of death that were elevated over the general population included cardiovascular causes (HR, 2.64; 95% CI, 1.78-3.91) and noncardiovascular causes, mainly infections; postsurgical complications; and factors such as alcoholism, suicide, or trauma (HR, 1.50; 95% CI, 1.18-1.91).
The study is limited by its retrospective nature, and because the surgical techniques used at the time are less effective than those used today, and could lead to weight gain over time. As a result, many patients who underwent surgery remained heavier than the general population. It’s also possible that negative health effects accumulated before surgery and persisted afterwards, according to Dr. Courcoulas.
The findings are likely generalizable to people with obesity, many of whom choose not to undergo bariatric surgery despite the potential benefits. “The population studied in SOS had a similar profile of underlying medical diseases to those groups who undergo bariatric surgery today and in the U.S. and around the world,” said Dr. Courcoulas.
The study was funded by the Swedish Research Council and others. Dr. Courcoulas has no relevant financial disclosures
SOURCE: Carlsson L et al. N Engl J Med. 2020 Oct 15. doi: 10.1056/NEJMoa2002449.
A new analysis of the Swedish Obese Subjects (SOS) study shows that bariatric surgery is associated with about a 3-year increase in lifespan, compared with obese patients who do not undergo surgery. Still, surgery did not restore normal lifespan: Surgical patients’ lifespan remained less than that of a sample from the general Swedish population. The study follows other reports suggesting reduced mortality after bariatric surgery, but with a longer follow-up.
“These data add even more evidence to the growing literature showing that patients who undergo bariatric surgery experience a reduction in all-cause long-term mortality. In making decisions around bariatric surgical procedures and care, patients and their health care providers need to understand the trade-offs between improved weight, health, and longer-term survival versus the surgical risks and problems over time,” said Anita P. Courcoulas, MD, MPH, chief of minimally invasive bariatric and general surgery at the University of Pittsburgh Medical Center, said in an interview. Dr. Courcoulas was not involved in the study.
The results appeared in the New England Journal of Medicine.
The SOS study drew from 25 surgical departments and 480 primary health care centers in Sweden. The researchers examined data from 2,007 patients who underwent bariatric surgery between 1987 and 2001, and compared their outcomes to 2,040 matched controls. All were between age 37 and 60 years, with a body mass index (BMI) of at least 34 kg/m2 for men and 38 for women. They also compared outcomes with 1,135 randomly sampled from the Swedish population registry.
Procedures included banding (18%), vertical banded gastroplasty (69%), and gastric bypass (13%). After an initial BMI reduction of about 11, the surgery group stabilized by year 8 at a BMI about 7 lower than baseline, and there was little change in BMI among controls.
After a mean follow-up of 24 years (interquartile range, 22-27 years), there were 10.7 deaths per 1,000 person-years in the surgery group, 13.2 among obese controls, and 5.2 in the general population (hazard ratio, 0.77 for surgery versus no surgery; P < .001). The general population had a lower mortality than nonsurgical controls (HR, 0.44; P < .001).
The surgery group had a higher median life expectancy than controls (median, 2.4 years; adjusted difference, 3.0 years; P < .001). The general population group had a median life expectancy that was 7.4 years higher than the control group (adjusted difference, 8.5 years; P < .001). The surgery group’s median life expectancy was still shorter than the general population reference (adjusted difference, 5.5 years; P < .001).
Cardiovascular disease risk was lower in the surgery group (HR, 0.70; 95% confidence interval, 0.57-0.85), as was risk of MI (HR, 0.51; 95% CI, 0.33-0.79), heart failure (HR, 0.52; 95% CI, 0.31-0.88), and stroke (HR, 0.45; 95% CI, 0.24-0.84). Cancer mortality was also lower (HR, 0.77; 95% CI, 0.61-0.96).
In the surgery group, causes of death that were elevated over the general population included cardiovascular causes (HR, 2.64; 95% CI, 1.78-3.91) and noncardiovascular causes, mainly infections; postsurgical complications; and factors such as alcoholism, suicide, or trauma (HR, 1.50; 95% CI, 1.18-1.91).
The study is limited by its retrospective nature, and because the surgical techniques used at the time are less effective than those used today, and could lead to weight gain over time. As a result, many patients who underwent surgery remained heavier than the general population. It’s also possible that negative health effects accumulated before surgery and persisted afterwards, according to Dr. Courcoulas.
The findings are likely generalizable to people with obesity, many of whom choose not to undergo bariatric surgery despite the potential benefits. “The population studied in SOS had a similar profile of underlying medical diseases to those groups who undergo bariatric surgery today and in the U.S. and around the world,” said Dr. Courcoulas.
The study was funded by the Swedish Research Council and others. Dr. Courcoulas has no relevant financial disclosures
SOURCE: Carlsson L et al. N Engl J Med. 2020 Oct 15. doi: 10.1056/NEJMoa2002449.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Intravascular lithotripsy hailed as ‘game changer’ for coronary calcification
aimed at gaining U.S. regulatory approval.
The technology is basically the same as in extracorporeal lithotripsy, used for the treatment of kidney stones for more than 30 years: namely, transmission of pulsed acoustic pressure waves in order to fracture calcium. For interventional cardiology purposes, however, the transmitter is located within a balloon angioplasty catheter, Dean J. Kereiakes, MD, explained in presenting the study results at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
In Disrupt CAD III, intravascular lithotripsy far exceeded the procedural success and 30-day freedom from major adverse cardiovascular event (MACE) performance targets set in conjunction with the Food and Drug Administration. In so doing, the intravascular lithotripsy device developed by Shockwave Medical successfully addressed one of the banes of contemporary interventional cardiology: heavily calcified coronary lesions.
Currently available technologies targeting such lesions, including noncompliant high-pressure balloons, intravascular lasers, cutting balloons, and orbital and rotational atherectomy, often yield suboptimal results, noted Dr. Kereiakes, medical director of the Christ Hospital Heart and Cardiovascular Center in Cincinnati.
Severe vascular calcifications are becoming more common, due in part to an aging population and the growing prevalence of hypertension, diabetes, and renal insufficiency. Severely calcified coronary lesions complicate percutaneous coronary intervention. They’re associated with increased risks of dissection, perforation, and periprocedural MI. Moreover, heavily calcified lesions impede stent delivery and expansion – and stent underexpansion is the leading predictor of restenosis and stent thrombosis, he observed at the meeting, sponsored by the Cardiovascular Research Foundation. Disrupt CAD III was a prospective single-arm study of 384 patients at 47 sites in the United States and several European countries. All participants had de novo coronary calcifications graded as severe by core laboratory assessment, with a mean calcified length of 47.9 mm by quantitative coronary angiography and a mean calcium angle and thickness of 292.5 degrees and 0.96 mm by optical coherence tomography.
“It’s staggering, the level of calcification these patients had. It’s jaw dropping,” Dr. Kereiakes observed.
Intravascular lithotripsy was used to prepare these severely calcified lesions for stenting. The intervention entailed transmission of acoustic waves circumferentially and transmurally at 1 pulse per second through tissue at an effective pressure of about 50 atm. Patients received an average of 69 pulses.
This was not a randomized trial; there was no sham-treated control arm. Instead, the comparator group selected under regulatory guidance was comprised of patients who had received orbital atherectomy for severe coronary calcifications in the earlier, similarly designed ORBIT II trial, which led to FDA marketing approval of that technology.
Key outcomes
The procedural success rate, defined as successful stent delivery with less than a 50% residual stenosis and no in-hospital MACE, was 92.4% in Disrupt CAD III, compared to 83.4% for orbital atherectomy in ORBIT II. The primary safety endpoint of freedom from cardiac death, MI, or target vessel revascularization at 30 days was achieved in 92.2% of patients in the intravascular lithotripsy trial, versus 84.4% in ORBIT II.
The 30-day MACE rate of 7.8% in Disrupt CAD III was primarily driven by periprocedural MIs, which occurred in 6.8% of participants. Only one-third of the MIs were clinically relevant by the Society for Coronary Angiography and Intervention definition. There were two cardiac deaths and three cases of stent thrombosis, all of which were associated with known predictors of the complication. There was 1 case each of dissection, abrupt closure, and perforation, but no instances of slow flow or no reflow at the procedure’s end. Transient lithotripsy-induced left ventricular capture occurred in 41% of patients, but they were benign events with no lasting consequences.
The device was able to cross and deliver acoustic pressure wave therapy to 98.2% of lesions. The mean diameter stenosis preprocedure was 65.1%, dropping to 37.2% post lithotripsy, with a final in-stent residual stenosis diameter of 11.9%, with a 1.7-mm acute gain. The average stent expansion at the site of maximum calcification was 102%, with a minimum stent area of 6.5 mm2.
Optical coherence imaging revealed that 67% of treated lesions had circumferential and transmural fractures of both deep and superficial calcium post lithotripsy. Yet outcomes were the same regardless of whether fractures were evident on imaging.
At 30-day follow-up, 72.9% of patients had no angina, up from just 12.6% of participants pre-PCI. Follow-up will continue for 2 years.
Outcomes were similar for the first case done at each participating center and all cases thereafter.
“The ease of use was remarkable,” Dr. Kereiakes recalled. “The learning curve is virtually nonexistent.”
The reaction
At a press conference where Dr. Kereiakes presented the Disrupt CAD III results, discussant Allen Jeremias, MD, said he found the results compelling.
“The success rate is high, I think it’s relatively easy to use, as demonstrated, and I think the results are spectacular,” said Dr. Jeremias, director of interventional cardiology research and associate director of the cardiac catheterization laboratory at St. Francis Hospital in Roslyn, N.Y.
Cardiologists “really don’t do a good job most of the time” with severely calcified coronary lesions, added Dr. Jeremias, who wasn’t involved in the trial.
“A lot of times these patients have inadequate stent outcomes when we do intravascular imaging. So to do something to try to basically crack the calcium and expand the stent is, I think, critically important in these patients, and this is an amazing technology that accomplishes that,” the cardiologist said.
Juan F. Granada, MD, of Columbia University, New York, who moderated the press conference, said, “Some of the debulking techniques used for calcified stenoses actually require a lot of training, knowledge, experience, and hospital infrastructure.
I really think having a technology that is easy to use and familiar to all interventional cardiologists, such as a balloon, could potentially be a disruptive change in our field.”
“It’s an absolute game changer,” agreed Dr. Jeremias.
Dr. Kereiakes reported serving as a consultant to a handful of medical device companies, including Shockwave Medical, which sponsored Disrupt CAD III.
SOURCE: Kereiakes DJ. TCT 2020. Late Breaking Clinical Science session 2.
aimed at gaining U.S. regulatory approval.
The technology is basically the same as in extracorporeal lithotripsy, used for the treatment of kidney stones for more than 30 years: namely, transmission of pulsed acoustic pressure waves in order to fracture calcium. For interventional cardiology purposes, however, the transmitter is located within a balloon angioplasty catheter, Dean J. Kereiakes, MD, explained in presenting the study results at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
In Disrupt CAD III, intravascular lithotripsy far exceeded the procedural success and 30-day freedom from major adverse cardiovascular event (MACE) performance targets set in conjunction with the Food and Drug Administration. In so doing, the intravascular lithotripsy device developed by Shockwave Medical successfully addressed one of the banes of contemporary interventional cardiology: heavily calcified coronary lesions.
Currently available technologies targeting such lesions, including noncompliant high-pressure balloons, intravascular lasers, cutting balloons, and orbital and rotational atherectomy, often yield suboptimal results, noted Dr. Kereiakes, medical director of the Christ Hospital Heart and Cardiovascular Center in Cincinnati.
Severe vascular calcifications are becoming more common, due in part to an aging population and the growing prevalence of hypertension, diabetes, and renal insufficiency. Severely calcified coronary lesions complicate percutaneous coronary intervention. They’re associated with increased risks of dissection, perforation, and periprocedural MI. Moreover, heavily calcified lesions impede stent delivery and expansion – and stent underexpansion is the leading predictor of restenosis and stent thrombosis, he observed at the meeting, sponsored by the Cardiovascular Research Foundation. Disrupt CAD III was a prospective single-arm study of 384 patients at 47 sites in the United States and several European countries. All participants had de novo coronary calcifications graded as severe by core laboratory assessment, with a mean calcified length of 47.9 mm by quantitative coronary angiography and a mean calcium angle and thickness of 292.5 degrees and 0.96 mm by optical coherence tomography.
“It’s staggering, the level of calcification these patients had. It’s jaw dropping,” Dr. Kereiakes observed.
Intravascular lithotripsy was used to prepare these severely calcified lesions for stenting. The intervention entailed transmission of acoustic waves circumferentially and transmurally at 1 pulse per second through tissue at an effective pressure of about 50 atm. Patients received an average of 69 pulses.
This was not a randomized trial; there was no sham-treated control arm. Instead, the comparator group selected under regulatory guidance was comprised of patients who had received orbital atherectomy for severe coronary calcifications in the earlier, similarly designed ORBIT II trial, which led to FDA marketing approval of that technology.
Key outcomes
The procedural success rate, defined as successful stent delivery with less than a 50% residual stenosis and no in-hospital MACE, was 92.4% in Disrupt CAD III, compared to 83.4% for orbital atherectomy in ORBIT II. The primary safety endpoint of freedom from cardiac death, MI, or target vessel revascularization at 30 days was achieved in 92.2% of patients in the intravascular lithotripsy trial, versus 84.4% in ORBIT II.
The 30-day MACE rate of 7.8% in Disrupt CAD III was primarily driven by periprocedural MIs, which occurred in 6.8% of participants. Only one-third of the MIs were clinically relevant by the Society for Coronary Angiography and Intervention definition. There were two cardiac deaths and three cases of stent thrombosis, all of which were associated with known predictors of the complication. There was 1 case each of dissection, abrupt closure, and perforation, but no instances of slow flow or no reflow at the procedure’s end. Transient lithotripsy-induced left ventricular capture occurred in 41% of patients, but they were benign events with no lasting consequences.
The device was able to cross and deliver acoustic pressure wave therapy to 98.2% of lesions. The mean diameter stenosis preprocedure was 65.1%, dropping to 37.2% post lithotripsy, with a final in-stent residual stenosis diameter of 11.9%, with a 1.7-mm acute gain. The average stent expansion at the site of maximum calcification was 102%, with a minimum stent area of 6.5 mm2.
Optical coherence imaging revealed that 67% of treated lesions had circumferential and transmural fractures of both deep and superficial calcium post lithotripsy. Yet outcomes were the same regardless of whether fractures were evident on imaging.
At 30-day follow-up, 72.9% of patients had no angina, up from just 12.6% of participants pre-PCI. Follow-up will continue for 2 years.
Outcomes were similar for the first case done at each participating center and all cases thereafter.
“The ease of use was remarkable,” Dr. Kereiakes recalled. “The learning curve is virtually nonexistent.”
The reaction
At a press conference where Dr. Kereiakes presented the Disrupt CAD III results, discussant Allen Jeremias, MD, said he found the results compelling.
“The success rate is high, I think it’s relatively easy to use, as demonstrated, and I think the results are spectacular,” said Dr. Jeremias, director of interventional cardiology research and associate director of the cardiac catheterization laboratory at St. Francis Hospital in Roslyn, N.Y.
Cardiologists “really don’t do a good job most of the time” with severely calcified coronary lesions, added Dr. Jeremias, who wasn’t involved in the trial.
“A lot of times these patients have inadequate stent outcomes when we do intravascular imaging. So to do something to try to basically crack the calcium and expand the stent is, I think, critically important in these patients, and this is an amazing technology that accomplishes that,” the cardiologist said.
Juan F. Granada, MD, of Columbia University, New York, who moderated the press conference, said, “Some of the debulking techniques used for calcified stenoses actually require a lot of training, knowledge, experience, and hospital infrastructure.
I really think having a technology that is easy to use and familiar to all interventional cardiologists, such as a balloon, could potentially be a disruptive change in our field.”
“It’s an absolute game changer,” agreed Dr. Jeremias.
Dr. Kereiakes reported serving as a consultant to a handful of medical device companies, including Shockwave Medical, which sponsored Disrupt CAD III.
SOURCE: Kereiakes DJ. TCT 2020. Late Breaking Clinical Science session 2.
aimed at gaining U.S. regulatory approval.
The technology is basically the same as in extracorporeal lithotripsy, used for the treatment of kidney stones for more than 30 years: namely, transmission of pulsed acoustic pressure waves in order to fracture calcium. For interventional cardiology purposes, however, the transmitter is located within a balloon angioplasty catheter, Dean J. Kereiakes, MD, explained in presenting the study results at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
In Disrupt CAD III, intravascular lithotripsy far exceeded the procedural success and 30-day freedom from major adverse cardiovascular event (MACE) performance targets set in conjunction with the Food and Drug Administration. In so doing, the intravascular lithotripsy device developed by Shockwave Medical successfully addressed one of the banes of contemporary interventional cardiology: heavily calcified coronary lesions.
Currently available technologies targeting such lesions, including noncompliant high-pressure balloons, intravascular lasers, cutting balloons, and orbital and rotational atherectomy, often yield suboptimal results, noted Dr. Kereiakes, medical director of the Christ Hospital Heart and Cardiovascular Center in Cincinnati.
Severe vascular calcifications are becoming more common, due in part to an aging population and the growing prevalence of hypertension, diabetes, and renal insufficiency. Severely calcified coronary lesions complicate percutaneous coronary intervention. They’re associated with increased risks of dissection, perforation, and periprocedural MI. Moreover, heavily calcified lesions impede stent delivery and expansion – and stent underexpansion is the leading predictor of restenosis and stent thrombosis, he observed at the meeting, sponsored by the Cardiovascular Research Foundation. Disrupt CAD III was a prospective single-arm study of 384 patients at 47 sites in the United States and several European countries. All participants had de novo coronary calcifications graded as severe by core laboratory assessment, with a mean calcified length of 47.9 mm by quantitative coronary angiography and a mean calcium angle and thickness of 292.5 degrees and 0.96 mm by optical coherence tomography.
“It’s staggering, the level of calcification these patients had. It’s jaw dropping,” Dr. Kereiakes observed.
Intravascular lithotripsy was used to prepare these severely calcified lesions for stenting. The intervention entailed transmission of acoustic waves circumferentially and transmurally at 1 pulse per second through tissue at an effective pressure of about 50 atm. Patients received an average of 69 pulses.
This was not a randomized trial; there was no sham-treated control arm. Instead, the comparator group selected under regulatory guidance was comprised of patients who had received orbital atherectomy for severe coronary calcifications in the earlier, similarly designed ORBIT II trial, which led to FDA marketing approval of that technology.
Key outcomes
The procedural success rate, defined as successful stent delivery with less than a 50% residual stenosis and no in-hospital MACE, was 92.4% in Disrupt CAD III, compared to 83.4% for orbital atherectomy in ORBIT II. The primary safety endpoint of freedom from cardiac death, MI, or target vessel revascularization at 30 days was achieved in 92.2% of patients in the intravascular lithotripsy trial, versus 84.4% in ORBIT II.
The 30-day MACE rate of 7.8% in Disrupt CAD III was primarily driven by periprocedural MIs, which occurred in 6.8% of participants. Only one-third of the MIs were clinically relevant by the Society for Coronary Angiography and Intervention definition. There were two cardiac deaths and three cases of stent thrombosis, all of which were associated with known predictors of the complication. There was 1 case each of dissection, abrupt closure, and perforation, but no instances of slow flow or no reflow at the procedure’s end. Transient lithotripsy-induced left ventricular capture occurred in 41% of patients, but they were benign events with no lasting consequences.
The device was able to cross and deliver acoustic pressure wave therapy to 98.2% of lesions. The mean diameter stenosis preprocedure was 65.1%, dropping to 37.2% post lithotripsy, with a final in-stent residual stenosis diameter of 11.9%, with a 1.7-mm acute gain. The average stent expansion at the site of maximum calcification was 102%, with a minimum stent area of 6.5 mm2.
Optical coherence imaging revealed that 67% of treated lesions had circumferential and transmural fractures of both deep and superficial calcium post lithotripsy. Yet outcomes were the same regardless of whether fractures were evident on imaging.
At 30-day follow-up, 72.9% of patients had no angina, up from just 12.6% of participants pre-PCI. Follow-up will continue for 2 years.
Outcomes were similar for the first case done at each participating center and all cases thereafter.
“The ease of use was remarkable,” Dr. Kereiakes recalled. “The learning curve is virtually nonexistent.”
The reaction
At a press conference where Dr. Kereiakes presented the Disrupt CAD III results, discussant Allen Jeremias, MD, said he found the results compelling.
“The success rate is high, I think it’s relatively easy to use, as demonstrated, and I think the results are spectacular,” said Dr. Jeremias, director of interventional cardiology research and associate director of the cardiac catheterization laboratory at St. Francis Hospital in Roslyn, N.Y.
Cardiologists “really don’t do a good job most of the time” with severely calcified coronary lesions, added Dr. Jeremias, who wasn’t involved in the trial.
“A lot of times these patients have inadequate stent outcomes when we do intravascular imaging. So to do something to try to basically crack the calcium and expand the stent is, I think, critically important in these patients, and this is an amazing technology that accomplishes that,” the cardiologist said.
Juan F. Granada, MD, of Columbia University, New York, who moderated the press conference, said, “Some of the debulking techniques used for calcified stenoses actually require a lot of training, knowledge, experience, and hospital infrastructure.
I really think having a technology that is easy to use and familiar to all interventional cardiologists, such as a balloon, could potentially be a disruptive change in our field.”
“It’s an absolute game changer,” agreed Dr. Jeremias.
Dr. Kereiakes reported serving as a consultant to a handful of medical device companies, including Shockwave Medical, which sponsored Disrupt CAD III.
SOURCE: Kereiakes DJ. TCT 2020. Late Breaking Clinical Science session 2.
FROM TCT 2020
Key clinical point: Intravascular lithotripsy was safe and effective for treatment of severely calcified coronary stenoses in a pivotal trial.
Major finding: The 30-day rate of freedom from major adverse cardiovascular events was 92.2%, well above the prespecified performance goal of 84.4%.
Study details: Disrupt CAD III study is a multicenter, single-arm, prospective study of intravascular lithotripsy in 384 patients with severe coronary calcification.
Disclosures: The presenter reported serving as a consultant to Shockwave Medical Inc., the study sponsor, as well as several other medical device companies.
Source: Kereiakes DJ. TCT 2020. Late Breaking Clinical Science session 2.
Benefit of rivaroxaban after limb revascularization greatest in those with comorbid CAD
The absolute benefit of adding low-dose rivaroxaban to low-dose aspirin following revascularization for symptomatic lower-extremity peripheral artery disease (PAD) is significantly greater in patients with comorbid coronary artery disease (CAD), according to a new secondary analysis of the VOYAGER PAD trial.
“These findings suggest heterogeneity of prognostic risk for ischemic events in lower-extremity PAD patients, and may support shared decision-making with these patients,” William R. Hiatt, MD, observed in presenting the study results at the virtual annual congress of the European Society of Cardiology.
VOYAGER PAD was a 3-year, 34-country clinical trial in which 6,564 patients with symptomatic PAD who had recently undergone lower-limb revascularization were randomized in double-blind fashion to rivaroxaban (Xarelto) at 2.5 mg twice daily or placebo on top of background standard therapy with low-dose aspirin.
Among the 2,067 participants with baseline comorbid CAD, the primary outcome – a composite comprised of cardiovascular death, acute MI, ischemic stroke, acute limb ischemia, and major amputation – occurred in 18.9% of the rivaroxaban group at 3 years and 24.3% on placebo, for a highly significant 22% relative risk reduction.
In contrast, in the 4,497 patients with PAD only, the primary outcome occurred in 16.1% of those on rivaroxaban and 17.9% of controls, an 11% relative risk reduction which failed to reach statistical significance. The absolute risk reduction achieved with rivaroxaban was 5.4% in patients with PAD plus CAD versus 1.8% in those with PAD alone. Thus, the significant clinical benefit with rivaroxaban plus aspirin previously reported in the overall study population, with a number needed to treat for 3 years of 39 in order to prevent one primary outcome event, was largely driven by the superior outcomes in the dual-diagnosis subgroup, reported Dr. Hiatt, professor of medicine at the University of Colorado at Denver, Aurora.
“A strategy of rivaroxaban at 2.5 mg twice daily plus low-dose aspirin versus low-dose aspirin alone reduces ischemic events of the limb, brain, and heart, but also increases bleeding, with an overall net benefit,” the cardiologist said. “In particular, the benefits of this strategy for MI and ischemic stroke are robust, especially in patients with PAD and CAD.”
Indeed, the MI rate at 3 years in the dual diagnosis subgroup was 7.3% with rivaroxaban and 8.8% with placebo, for a 23% relative risk reduction, compared with rates of 3.3% and 3.7%, respectively, in patients with PAD only. Similarly, ischemic stroke occurred in 2.9% of patients with PAD and CAD in the rivaroxaban group, compared with 3.9% with placebo, whereas the rate in the PAD only group was identical at 2.6% regardless of whether patients were on rivaroxaban or placebo.
In patients without CAD, the clinical benefit of rivaroxaban was driven by reductions in severe limb events. Their rate of acute limb ischemia was 5.2% with rivaroxaban, compared with 8.3% with placebo, for a 37% relative risk reduction. In contrast, the reduction in acute limb ischemia with rivaroxaban in patients with PAD and CAD wasn’t significantly different from placebo.
Thrombolysis in Myocardial Infarction major bleeding occurred in 2.4% of patients with PAD and CAD on rivaroxaban, compared with 1.1% on placebo, and in 1.7% and 1.5% of patients with PAD alone. Of note, rates of ischemic stroke or fatal hemorrhage were low and similar at less than 1% in all four groups, Dr. Hiatt noted.
VOYAGER PAD was sponsored by Bayer and Janssen. Dr. Hiatt reported receiving research grant support from those two companies as well as Amgen.
The absolute benefit of adding low-dose rivaroxaban to low-dose aspirin following revascularization for symptomatic lower-extremity peripheral artery disease (PAD) is significantly greater in patients with comorbid coronary artery disease (CAD), according to a new secondary analysis of the VOYAGER PAD trial.
“These findings suggest heterogeneity of prognostic risk for ischemic events in lower-extremity PAD patients, and may support shared decision-making with these patients,” William R. Hiatt, MD, observed in presenting the study results at the virtual annual congress of the European Society of Cardiology.
VOYAGER PAD was a 3-year, 34-country clinical trial in which 6,564 patients with symptomatic PAD who had recently undergone lower-limb revascularization were randomized in double-blind fashion to rivaroxaban (Xarelto) at 2.5 mg twice daily or placebo on top of background standard therapy with low-dose aspirin.
Among the 2,067 participants with baseline comorbid CAD, the primary outcome – a composite comprised of cardiovascular death, acute MI, ischemic stroke, acute limb ischemia, and major amputation – occurred in 18.9% of the rivaroxaban group at 3 years and 24.3% on placebo, for a highly significant 22% relative risk reduction.
In contrast, in the 4,497 patients with PAD only, the primary outcome occurred in 16.1% of those on rivaroxaban and 17.9% of controls, an 11% relative risk reduction which failed to reach statistical significance. The absolute risk reduction achieved with rivaroxaban was 5.4% in patients with PAD plus CAD versus 1.8% in those with PAD alone. Thus, the significant clinical benefit with rivaroxaban plus aspirin previously reported in the overall study population, with a number needed to treat for 3 years of 39 in order to prevent one primary outcome event, was largely driven by the superior outcomes in the dual-diagnosis subgroup, reported Dr. Hiatt, professor of medicine at the University of Colorado at Denver, Aurora.
“A strategy of rivaroxaban at 2.5 mg twice daily plus low-dose aspirin versus low-dose aspirin alone reduces ischemic events of the limb, brain, and heart, but also increases bleeding, with an overall net benefit,” the cardiologist said. “In particular, the benefits of this strategy for MI and ischemic stroke are robust, especially in patients with PAD and CAD.”
Indeed, the MI rate at 3 years in the dual diagnosis subgroup was 7.3% with rivaroxaban and 8.8% with placebo, for a 23% relative risk reduction, compared with rates of 3.3% and 3.7%, respectively, in patients with PAD only. Similarly, ischemic stroke occurred in 2.9% of patients with PAD and CAD in the rivaroxaban group, compared with 3.9% with placebo, whereas the rate in the PAD only group was identical at 2.6% regardless of whether patients were on rivaroxaban or placebo.
In patients without CAD, the clinical benefit of rivaroxaban was driven by reductions in severe limb events. Their rate of acute limb ischemia was 5.2% with rivaroxaban, compared with 8.3% with placebo, for a 37% relative risk reduction. In contrast, the reduction in acute limb ischemia with rivaroxaban in patients with PAD and CAD wasn’t significantly different from placebo.
Thrombolysis in Myocardial Infarction major bleeding occurred in 2.4% of patients with PAD and CAD on rivaroxaban, compared with 1.1% on placebo, and in 1.7% and 1.5% of patients with PAD alone. Of note, rates of ischemic stroke or fatal hemorrhage were low and similar at less than 1% in all four groups, Dr. Hiatt noted.
VOYAGER PAD was sponsored by Bayer and Janssen. Dr. Hiatt reported receiving research grant support from those two companies as well as Amgen.
The absolute benefit of adding low-dose rivaroxaban to low-dose aspirin following revascularization for symptomatic lower-extremity peripheral artery disease (PAD) is significantly greater in patients with comorbid coronary artery disease (CAD), according to a new secondary analysis of the VOYAGER PAD trial.
“These findings suggest heterogeneity of prognostic risk for ischemic events in lower-extremity PAD patients, and may support shared decision-making with these patients,” William R. Hiatt, MD, observed in presenting the study results at the virtual annual congress of the European Society of Cardiology.
VOYAGER PAD was a 3-year, 34-country clinical trial in which 6,564 patients with symptomatic PAD who had recently undergone lower-limb revascularization were randomized in double-blind fashion to rivaroxaban (Xarelto) at 2.5 mg twice daily or placebo on top of background standard therapy with low-dose aspirin.
Among the 2,067 participants with baseline comorbid CAD, the primary outcome – a composite comprised of cardiovascular death, acute MI, ischemic stroke, acute limb ischemia, and major amputation – occurred in 18.9% of the rivaroxaban group at 3 years and 24.3% on placebo, for a highly significant 22% relative risk reduction.
In contrast, in the 4,497 patients with PAD only, the primary outcome occurred in 16.1% of those on rivaroxaban and 17.9% of controls, an 11% relative risk reduction which failed to reach statistical significance. The absolute risk reduction achieved with rivaroxaban was 5.4% in patients with PAD plus CAD versus 1.8% in those with PAD alone. Thus, the significant clinical benefit with rivaroxaban plus aspirin previously reported in the overall study population, with a number needed to treat for 3 years of 39 in order to prevent one primary outcome event, was largely driven by the superior outcomes in the dual-diagnosis subgroup, reported Dr. Hiatt, professor of medicine at the University of Colorado at Denver, Aurora.
“A strategy of rivaroxaban at 2.5 mg twice daily plus low-dose aspirin versus low-dose aspirin alone reduces ischemic events of the limb, brain, and heart, but also increases bleeding, with an overall net benefit,” the cardiologist said. “In particular, the benefits of this strategy for MI and ischemic stroke are robust, especially in patients with PAD and CAD.”
Indeed, the MI rate at 3 years in the dual diagnosis subgroup was 7.3% with rivaroxaban and 8.8% with placebo, for a 23% relative risk reduction, compared with rates of 3.3% and 3.7%, respectively, in patients with PAD only. Similarly, ischemic stroke occurred in 2.9% of patients with PAD and CAD in the rivaroxaban group, compared with 3.9% with placebo, whereas the rate in the PAD only group was identical at 2.6% regardless of whether patients were on rivaroxaban or placebo.
In patients without CAD, the clinical benefit of rivaroxaban was driven by reductions in severe limb events. Their rate of acute limb ischemia was 5.2% with rivaroxaban, compared with 8.3% with placebo, for a 37% relative risk reduction. In contrast, the reduction in acute limb ischemia with rivaroxaban in patients with PAD and CAD wasn’t significantly different from placebo.
Thrombolysis in Myocardial Infarction major bleeding occurred in 2.4% of patients with PAD and CAD on rivaroxaban, compared with 1.1% on placebo, and in 1.7% and 1.5% of patients with PAD alone. Of note, rates of ischemic stroke or fatal hemorrhage were low and similar at less than 1% in all four groups, Dr. Hiatt noted.
VOYAGER PAD was sponsored by Bayer and Janssen. Dr. Hiatt reported receiving research grant support from those two companies as well as Amgen.
FROM ESC CONGRESS 2020
Psychosocial resilience associated with better cardiovascular health in Blacks
Resilience might deserve targeting
Increased psychosocial resilience, which captures a sense of purpose, optimism, and life-coping strategies, correlates with improved cardiovascular (CV) health in Black Americans, according to a study that might hold a key for identifying new strategies for CV disease prevention.
“Our findings highlight the importance of individual psychosocial factors that promote cardiovascular health among Black adults, traditionally considered to be a high-risk population,” according to a team of authors collaborating on a study produced by the Morehouse-Emory Cardiovascular Center for Health Equity in Atlanta.
Studies associating psychosocial resilience with improved health outcomes have been published before. In a 12-study review of this concept, it was emphasized that resilience is a dynamic process, not a personality trait, and has shown promise as a target of efforts to relieve the burden of disease (Johnston MC et al. Psychosomatics 2015;56:168-80).
In this study, which received partial support from the American Heart Association, psychosocial resilience was evaluated at both the individual level and at the community level among 389 Black adults living in Atlanta. The senior author was Tené T. Lewis, PhD, of the department of epidemiology at Emory’s Rollins School of Public Health (Circ Cardiovasc Qual Outcomes 2020 Oct 7;13:3006638).
Psychosocial resilience was calculated across the domains of environmental mastery, purpose of life, optimism, coping, and lack of depression with standardized tests, such as the Life Orientation Test-Revised questionnaire for optimism and the Ryff Scales of Psychological Well-Being for the domains of environmental mastery and purpose of life. A composite score for psychosocial resilience was reached by calculating the median score across the measured domains.
Patients with high psychosocial resilience, defined as a composite score above the median, or low resilience, defined as a lower score, were then compared for CV health based on the AHA’s Life’s Simple 7 (LS7) score.
LS7 scores incorporate measures for exercise, diet, smoking history, blood pressure, glucose, cholesterol, and body mass index. Composite LS7 scores range from 0 to 14. Prior work cited by the authors have associated each 1-unit increase in LS7 score with a 13% lower risk of CVD.
As a continuous variable for CV risk at the individual level, each higher standard-deviation increment in the composite psychosocial resilience score was associated with a highly significant 0.42-point increase in LS7 score (P < .001) for study participants. In other words, increasing resilience predicted lower CV risk scores.
Resilience was also calculated at the community level by looking at census tract-level rates of CV mortality and morbidity relative to socioeconomic status. Again, high CV resilience, defined as scores above the median, were compared with lower scores across neighborhoods with similar median household income. As a continuous variable in this analysis, each higher standard-deviation increment in the resilience score was associated with a 0.27-point increase in LS7 score (P = .01).
After adjustment for sociodemographic factors, the association between psychosocial resilience and CV health remained significant for both the individual and community calculations, according to the authors. When examined jointly, high individual psychosocial resilience remained independently associated with improved CV health, but living in a high-resilience neighborhood was not an independent predictor.
When evaluated individually, each of the domains in the psychosocial resistance score were positively correlated with higher LS7 scores, meaning lower CV risk. The strongest associations on a statistical level were low depressive symptoms (P = .001), environmental mastery (P = .006), and purpose in life (P = .009).
The impact of high psychosocial resistance scores was greatest in Black adults living in low-resilience neighborhoods. Among these subjects, high resilience was associated with a nearly 1-point increase in LS7 score relative to low resilience (8.38 vs. 7.42). This was unexpected, but it “is consistent with some broader conceptual literature that posits that individual psychosocial resilience matters more under conditions of adversity,” the authors reported.
Understanding disparities is key
Black race has repeatedly been associated with an increased risk of CV events, but this study is valuable for providing a fresh perspective on the potential reasons, according to the authors of an accompanying editorial, Amber E. Johnson, MD, and Jared Magnani, MD, who are both affiliated with the division of cardiology at the University of Pittsburgh (Circ Cardiovasc Qual Outcomes 2020 Oct 7. doi: 10.1161/CIRCOUTCOMES.120.007357.
“Clinicians increasingly recognize that race-based disparities do not stem inherently from race; instead, the disparities stem from the underlying social determinations of health,” they wrote, citing such variables as unequal access to pay and acceptable living conditions “and the structural racism that perpetuates them.”
They agreed with the authors that promotion of psychosocial resilience among Black people living in communities with poor CV health has the potential to improve CV outcomes, but they warned that this is complex. Although they contend that resilience techniques can be taught, they cautioned there might be limitations if the underlying factors associated with poor psychosocial resilience remain unchanged.
“Thus, the superficial application of positive psychology strategies is likely insufficient to bring parity to CV health outcomes,” they wrote, concluding that strategies to promote health equity would negate the need for interventions to bolster resilience.
Studies that focus on Black adults and cardiovascular health, including this investigation into the role of psychosocial factors “are much needed and very welcome,” said Harlan M. Krumholz, MD, a cardiologist and professor in the Institute for Social and Policy Studies at Yale University, New Haven, Conn.
He sees a broad array of potential directions of research.
“The study opens many questions about whether the resilience can be strengthened by interventions; whether addressing structural racism could reduce the need for such resilience, and whether this association is specific to Black adults in an urban center or is generally present in other settings and in other populations,” Dr. Krumholz said.
An effort is now needed to determine “whether this is a marker or a mediator of cardiovascular health,” he added.
In either case, resilience is a potentially important factor for understanding racial disparities in CV-disease prevalence and outcomes, according to the authors of the accompanying editorial and Dr. Krumholz.
SOURCE: Kim JH et al. Circ Cardiovasc Qual Outcomes. 2020 Oct 7;13:e006638.
Resilience might deserve targeting
Resilience might deserve targeting
Increased psychosocial resilience, which captures a sense of purpose, optimism, and life-coping strategies, correlates with improved cardiovascular (CV) health in Black Americans, according to a study that might hold a key for identifying new strategies for CV disease prevention.
“Our findings highlight the importance of individual psychosocial factors that promote cardiovascular health among Black adults, traditionally considered to be a high-risk population,” according to a team of authors collaborating on a study produced by the Morehouse-Emory Cardiovascular Center for Health Equity in Atlanta.
Studies associating psychosocial resilience with improved health outcomes have been published before. In a 12-study review of this concept, it was emphasized that resilience is a dynamic process, not a personality trait, and has shown promise as a target of efforts to relieve the burden of disease (Johnston MC et al. Psychosomatics 2015;56:168-80).
In this study, which received partial support from the American Heart Association, psychosocial resilience was evaluated at both the individual level and at the community level among 389 Black adults living in Atlanta. The senior author was Tené T. Lewis, PhD, of the department of epidemiology at Emory’s Rollins School of Public Health (Circ Cardiovasc Qual Outcomes 2020 Oct 7;13:3006638).
Psychosocial resilience was calculated across the domains of environmental mastery, purpose of life, optimism, coping, and lack of depression with standardized tests, such as the Life Orientation Test-Revised questionnaire for optimism and the Ryff Scales of Psychological Well-Being for the domains of environmental mastery and purpose of life. A composite score for psychosocial resilience was reached by calculating the median score across the measured domains.
Patients with high psychosocial resilience, defined as a composite score above the median, or low resilience, defined as a lower score, were then compared for CV health based on the AHA’s Life’s Simple 7 (LS7) score.
LS7 scores incorporate measures for exercise, diet, smoking history, blood pressure, glucose, cholesterol, and body mass index. Composite LS7 scores range from 0 to 14. Prior work cited by the authors have associated each 1-unit increase in LS7 score with a 13% lower risk of CVD.
As a continuous variable for CV risk at the individual level, each higher standard-deviation increment in the composite psychosocial resilience score was associated with a highly significant 0.42-point increase in LS7 score (P < .001) for study participants. In other words, increasing resilience predicted lower CV risk scores.
Resilience was also calculated at the community level by looking at census tract-level rates of CV mortality and morbidity relative to socioeconomic status. Again, high CV resilience, defined as scores above the median, were compared with lower scores across neighborhoods with similar median household income. As a continuous variable in this analysis, each higher standard-deviation increment in the resilience score was associated with a 0.27-point increase in LS7 score (P = .01).
After adjustment for sociodemographic factors, the association between psychosocial resilience and CV health remained significant for both the individual and community calculations, according to the authors. When examined jointly, high individual psychosocial resilience remained independently associated with improved CV health, but living in a high-resilience neighborhood was not an independent predictor.
When evaluated individually, each of the domains in the psychosocial resistance score were positively correlated with higher LS7 scores, meaning lower CV risk. The strongest associations on a statistical level were low depressive symptoms (P = .001), environmental mastery (P = .006), and purpose in life (P = .009).
The impact of high psychosocial resistance scores was greatest in Black adults living in low-resilience neighborhoods. Among these subjects, high resilience was associated with a nearly 1-point increase in LS7 score relative to low resilience (8.38 vs. 7.42). This was unexpected, but it “is consistent with some broader conceptual literature that posits that individual psychosocial resilience matters more under conditions of adversity,” the authors reported.
Understanding disparities is key
Black race has repeatedly been associated with an increased risk of CV events, but this study is valuable for providing a fresh perspective on the potential reasons, according to the authors of an accompanying editorial, Amber E. Johnson, MD, and Jared Magnani, MD, who are both affiliated with the division of cardiology at the University of Pittsburgh (Circ Cardiovasc Qual Outcomes 2020 Oct 7. doi: 10.1161/CIRCOUTCOMES.120.007357.
“Clinicians increasingly recognize that race-based disparities do not stem inherently from race; instead, the disparities stem from the underlying social determinations of health,” they wrote, citing such variables as unequal access to pay and acceptable living conditions “and the structural racism that perpetuates them.”
They agreed with the authors that promotion of psychosocial resilience among Black people living in communities with poor CV health has the potential to improve CV outcomes, but they warned that this is complex. Although they contend that resilience techniques can be taught, they cautioned there might be limitations if the underlying factors associated with poor psychosocial resilience remain unchanged.
“Thus, the superficial application of positive psychology strategies is likely insufficient to bring parity to CV health outcomes,” they wrote, concluding that strategies to promote health equity would negate the need for interventions to bolster resilience.
Studies that focus on Black adults and cardiovascular health, including this investigation into the role of psychosocial factors “are much needed and very welcome,” said Harlan M. Krumholz, MD, a cardiologist and professor in the Institute for Social and Policy Studies at Yale University, New Haven, Conn.
He sees a broad array of potential directions of research.
“The study opens many questions about whether the resilience can be strengthened by interventions; whether addressing structural racism could reduce the need for such resilience, and whether this association is specific to Black adults in an urban center or is generally present in other settings and in other populations,” Dr. Krumholz said.
An effort is now needed to determine “whether this is a marker or a mediator of cardiovascular health,” he added.
In either case, resilience is a potentially important factor for understanding racial disparities in CV-disease prevalence and outcomes, according to the authors of the accompanying editorial and Dr. Krumholz.
SOURCE: Kim JH et al. Circ Cardiovasc Qual Outcomes. 2020 Oct 7;13:e006638.
Increased psychosocial resilience, which captures a sense of purpose, optimism, and life-coping strategies, correlates with improved cardiovascular (CV) health in Black Americans, according to a study that might hold a key for identifying new strategies for CV disease prevention.
“Our findings highlight the importance of individual psychosocial factors that promote cardiovascular health among Black adults, traditionally considered to be a high-risk population,” according to a team of authors collaborating on a study produced by the Morehouse-Emory Cardiovascular Center for Health Equity in Atlanta.
Studies associating psychosocial resilience with improved health outcomes have been published before. In a 12-study review of this concept, it was emphasized that resilience is a dynamic process, not a personality trait, and has shown promise as a target of efforts to relieve the burden of disease (Johnston MC et al. Psychosomatics 2015;56:168-80).
In this study, which received partial support from the American Heart Association, psychosocial resilience was evaluated at both the individual level and at the community level among 389 Black adults living in Atlanta. The senior author was Tené T. Lewis, PhD, of the department of epidemiology at Emory’s Rollins School of Public Health (Circ Cardiovasc Qual Outcomes 2020 Oct 7;13:3006638).
Psychosocial resilience was calculated across the domains of environmental mastery, purpose of life, optimism, coping, and lack of depression with standardized tests, such as the Life Orientation Test-Revised questionnaire for optimism and the Ryff Scales of Psychological Well-Being for the domains of environmental mastery and purpose of life. A composite score for psychosocial resilience was reached by calculating the median score across the measured domains.
Patients with high psychosocial resilience, defined as a composite score above the median, or low resilience, defined as a lower score, were then compared for CV health based on the AHA’s Life’s Simple 7 (LS7) score.
LS7 scores incorporate measures for exercise, diet, smoking history, blood pressure, glucose, cholesterol, and body mass index. Composite LS7 scores range from 0 to 14. Prior work cited by the authors have associated each 1-unit increase in LS7 score with a 13% lower risk of CVD.
As a continuous variable for CV risk at the individual level, each higher standard-deviation increment in the composite psychosocial resilience score was associated with a highly significant 0.42-point increase in LS7 score (P < .001) for study participants. In other words, increasing resilience predicted lower CV risk scores.
Resilience was also calculated at the community level by looking at census tract-level rates of CV mortality and morbidity relative to socioeconomic status. Again, high CV resilience, defined as scores above the median, were compared with lower scores across neighborhoods with similar median household income. As a continuous variable in this analysis, each higher standard-deviation increment in the resilience score was associated with a 0.27-point increase in LS7 score (P = .01).
After adjustment for sociodemographic factors, the association between psychosocial resilience and CV health remained significant for both the individual and community calculations, according to the authors. When examined jointly, high individual psychosocial resilience remained independently associated with improved CV health, but living in a high-resilience neighborhood was not an independent predictor.
When evaluated individually, each of the domains in the psychosocial resistance score were positively correlated with higher LS7 scores, meaning lower CV risk. The strongest associations on a statistical level were low depressive symptoms (P = .001), environmental mastery (P = .006), and purpose in life (P = .009).
The impact of high psychosocial resistance scores was greatest in Black adults living in low-resilience neighborhoods. Among these subjects, high resilience was associated with a nearly 1-point increase in LS7 score relative to low resilience (8.38 vs. 7.42). This was unexpected, but it “is consistent with some broader conceptual literature that posits that individual psychosocial resilience matters more under conditions of adversity,” the authors reported.
Understanding disparities is key
Black race has repeatedly been associated with an increased risk of CV events, but this study is valuable for providing a fresh perspective on the potential reasons, according to the authors of an accompanying editorial, Amber E. Johnson, MD, and Jared Magnani, MD, who are both affiliated with the division of cardiology at the University of Pittsburgh (Circ Cardiovasc Qual Outcomes 2020 Oct 7. doi: 10.1161/CIRCOUTCOMES.120.007357.
“Clinicians increasingly recognize that race-based disparities do not stem inherently from race; instead, the disparities stem from the underlying social determinations of health,” they wrote, citing such variables as unequal access to pay and acceptable living conditions “and the structural racism that perpetuates them.”
They agreed with the authors that promotion of psychosocial resilience among Black people living in communities with poor CV health has the potential to improve CV outcomes, but they warned that this is complex. Although they contend that resilience techniques can be taught, they cautioned there might be limitations if the underlying factors associated with poor psychosocial resilience remain unchanged.
“Thus, the superficial application of positive psychology strategies is likely insufficient to bring parity to CV health outcomes,” they wrote, concluding that strategies to promote health equity would negate the need for interventions to bolster resilience.
Studies that focus on Black adults and cardiovascular health, including this investigation into the role of psychosocial factors “are much needed and very welcome,” said Harlan M. Krumholz, MD, a cardiologist and professor in the Institute for Social and Policy Studies at Yale University, New Haven, Conn.
He sees a broad array of potential directions of research.
“The study opens many questions about whether the resilience can be strengthened by interventions; whether addressing structural racism could reduce the need for such resilience, and whether this association is specific to Black adults in an urban center or is generally present in other settings and in other populations,” Dr. Krumholz said.
An effort is now needed to determine “whether this is a marker or a mediator of cardiovascular health,” he added.
In either case, resilience is a potentially important factor for understanding racial disparities in CV-disease prevalence and outcomes, according to the authors of the accompanying editorial and Dr. Krumholz.
SOURCE: Kim JH et al. Circ Cardiovasc Qual Outcomes. 2020 Oct 7;13:e006638.
FROM CIRCULATION: CARDIOVASCULAR QUALITY AND OUTCOMES
Stress tests before knee, hip replacement surgeries down, with no ill effects
Cardiac stress testing before hip and knee replacements has dropped steadily since 2006, according to results from a new study that also showed major cardiac complications to be low in the absence of stress testing – even among people with established risk factors.
Routine stress testing before noncardiac surgeries has come under fire in recent decades as an overuse of resources and a burden on patients. Practice guidelines issued in 2007 and 2014 by the American College of Cardiology and the American Heart Association sought to limit the use of preoperative testing to patients with specific cardiovascular risk factors who might have their management changed by the test results.
For their study, published online in JAMA Cardiology, Daniel S. Rubin, MD, of the University of Chicago and colleagues looked at employee-based insurance data, which included Medicare Advantage claims, for more than 800,000 total hip or knee arthroplasties (28% hip and 72% knee replacements) conducted between 2004 and 2017.
While some 10% of the cohort (mean age 62, 58% women) received a stress test in the 2 months before surgery, the investigators found that the frequency of preoperative stress testing dropped annually starting in late 2006, when it peaked at about 14%, to about 7% in 2017. Older age, male sex and a Revised Cardiac Risk Index score of 1 or greater were all associated with a higher likelihood of being tested.
The overall frequency of myocardial infarction or cardiac arrest was 0.24%, occurring in 1,677 of 686,067 patients. While the rate was higher in patients with at least one RCRI condition, this did not differ significantly between those who received a preoperative stress test and those who did not (0.60%; 221 of 36,554 vs. 0.57%; 694 of 122,466 patients.
The 2007 and 2014 ACC/AHA guidelines make clear that patients with zero RCRI conditions – which comprise a history of ischemic heart disease, heart failure, insulin therapy for diabetes, cerebrovascular disease, or chronic kidney disease – should not receive a stress test before an intermediate-risk surgery such as a hip or knee replacement. But in this study, Dr. Rubin and his colleagues found that almost half of patients who had no RCRI risk factors were stress tested anyway. This means, Dr. Rubin said in an interview, that “there’s still room for improvement” in reducing testing.
“I never want to question how a physician chooses to practice, but I have to applaud physicians for reining in the use of this test. We’re using less of this test and yet the incidence of myocardial infarction and cardiac arrest is also going down, which also calls into question whether we’re getting better at choosing the right patients for the test; or the test doesn’t impact outcomes; or overall health of these patients is improving,” he said.
One surprise finding in the study, Dr. Rubin noted, was a higher rate of complications among people without RCRI conditions who were stress tested, compared with those who were not, with a mean complication rate of 0.27%, compared with 0.14% among those who did not receive a test (P < .001). “The RCRI doesn’t capture certain things,” Dr. Rubin said. “And we know that no risk stratification tool is going to capture everything.”
The RCRI, he noted, is based on a clinical history. “If you haven’t been diagnosed yet, it won’t appear as a risk factor, even if you’re clearly at risk. The question then becomes for a physician, do you do the test or not? On a day-to-day basis it’s hard to make that decision because you want what’s best for the individual patient – and it’s hard to generalize from a study of 800,000 people what’s right for that one patient. That said, it doesn’t appear that stress testing improves outcomes and a decrease in testing appears appropriate.”
Dr. Rubin and his colleagues described as a weakness of their study that it did not capture the full scope of preoperative stress testing among Medicare patients, who are older and therefore more likely to be tested.
That the 2007 and 2014 practice guidelines bore on the drop in testing was not demonstrated by Dr. Rubin and colleagues’ study, which saw declines begin even before the guidelines were published. Nonetheless, the results appear to validate the approach advocated in the guidelines, said guideline coauthor Joshua Beckman, MD, of Vanderbilt University, whose recent research has focused on identifying risk factors for MI after noncardiac surgery.
“I hope that the guidelines have helped in changing the culture for the use of preoperative stress testing as a regular thing,” Dr. Beckman said in an interview. “In fact, the guidelines say you shouldn’t do anything before an operation that you wouldn’t do anyway. So these findings are certainly in agreement with what we’re suggesting and support the idea that unless you have something that is unstable or active, stress testing isn’t likely to help.”
Annemarie Thompson, MD, of Duke University in Durham, N.C., another coauthor on the 2014 guidelines, commented in an interview that Dr. Rubin and colleagues’ findings of a doubled rate of complications among people without RCRI conditions who were stress tested, compared with those who were not might mean something “other than just sheer overuse or overordering of tests inappropriately.”
Rather, she said, physicians might be seeing something in the clinic that cannot be captured by a screening tool reliant on existing diagnoses. “Maybe when they’re sitting in front of you in a clinic, they’re so immobile that you’re left wondering. Or maybe they haven’t been seen by a doctor in a long time,” Dr. Thompson said. “So they don’t have diagnoses if they haven’t been followed. I think what [this finding] shows is that clinicians are detecting something. They may not know what it is. But we have to give a little wiggle room to the clinician who is sitting there looking at a patient who looks like they may not make it through surgery.”
Dr. Thompson said it would be helpful, after a big-data study like this one, to go through the clinical histories of those patients – in this study fewer than 100 – who had no RCRI risk factors and yet were stress tested and ended up having complications. “Until then we’re not going to solve the mystery,” she said. “But it’s a very, very interesting study.”
Dr. Rubin is the president of DRDR Mobile Health, a company that creates mobile applications for health care and from which he has not received compensation. One of his coauthors on the study, Dr. Peter Nagele, reported fee income from Roche Diagnostics. Dr. Beckman disclosed personal fees from AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, and other pharmaceutical manufacturers. Dr. Thompson has no disclosures.
SOURCE: Rubin et al. JAMA Cardiol. 2020 Sep 30. doi: 10.1001/jamacardio.2020.4311.
Cardiac stress testing before hip and knee replacements has dropped steadily since 2006, according to results from a new study that also showed major cardiac complications to be low in the absence of stress testing – even among people with established risk factors.
Routine stress testing before noncardiac surgeries has come under fire in recent decades as an overuse of resources and a burden on patients. Practice guidelines issued in 2007 and 2014 by the American College of Cardiology and the American Heart Association sought to limit the use of preoperative testing to patients with specific cardiovascular risk factors who might have their management changed by the test results.
For their study, published online in JAMA Cardiology, Daniel S. Rubin, MD, of the University of Chicago and colleagues looked at employee-based insurance data, which included Medicare Advantage claims, for more than 800,000 total hip or knee arthroplasties (28% hip and 72% knee replacements) conducted between 2004 and 2017.
While some 10% of the cohort (mean age 62, 58% women) received a stress test in the 2 months before surgery, the investigators found that the frequency of preoperative stress testing dropped annually starting in late 2006, when it peaked at about 14%, to about 7% in 2017. Older age, male sex and a Revised Cardiac Risk Index score of 1 or greater were all associated with a higher likelihood of being tested.
The overall frequency of myocardial infarction or cardiac arrest was 0.24%, occurring in 1,677 of 686,067 patients. While the rate was higher in patients with at least one RCRI condition, this did not differ significantly between those who received a preoperative stress test and those who did not (0.60%; 221 of 36,554 vs. 0.57%; 694 of 122,466 patients.
The 2007 and 2014 ACC/AHA guidelines make clear that patients with zero RCRI conditions – which comprise a history of ischemic heart disease, heart failure, insulin therapy for diabetes, cerebrovascular disease, or chronic kidney disease – should not receive a stress test before an intermediate-risk surgery such as a hip or knee replacement. But in this study, Dr. Rubin and his colleagues found that almost half of patients who had no RCRI risk factors were stress tested anyway. This means, Dr. Rubin said in an interview, that “there’s still room for improvement” in reducing testing.
“I never want to question how a physician chooses to practice, but I have to applaud physicians for reining in the use of this test. We’re using less of this test and yet the incidence of myocardial infarction and cardiac arrest is also going down, which also calls into question whether we’re getting better at choosing the right patients for the test; or the test doesn’t impact outcomes; or overall health of these patients is improving,” he said.
One surprise finding in the study, Dr. Rubin noted, was a higher rate of complications among people without RCRI conditions who were stress tested, compared with those who were not, with a mean complication rate of 0.27%, compared with 0.14% among those who did not receive a test (P < .001). “The RCRI doesn’t capture certain things,” Dr. Rubin said. “And we know that no risk stratification tool is going to capture everything.”
The RCRI, he noted, is based on a clinical history. “If you haven’t been diagnosed yet, it won’t appear as a risk factor, even if you’re clearly at risk. The question then becomes for a physician, do you do the test or not? On a day-to-day basis it’s hard to make that decision because you want what’s best for the individual patient – and it’s hard to generalize from a study of 800,000 people what’s right for that one patient. That said, it doesn’t appear that stress testing improves outcomes and a decrease in testing appears appropriate.”
Dr. Rubin and his colleagues described as a weakness of their study that it did not capture the full scope of preoperative stress testing among Medicare patients, who are older and therefore more likely to be tested.
That the 2007 and 2014 practice guidelines bore on the drop in testing was not demonstrated by Dr. Rubin and colleagues’ study, which saw declines begin even before the guidelines were published. Nonetheless, the results appear to validate the approach advocated in the guidelines, said guideline coauthor Joshua Beckman, MD, of Vanderbilt University, whose recent research has focused on identifying risk factors for MI after noncardiac surgery.
“I hope that the guidelines have helped in changing the culture for the use of preoperative stress testing as a regular thing,” Dr. Beckman said in an interview. “In fact, the guidelines say you shouldn’t do anything before an operation that you wouldn’t do anyway. So these findings are certainly in agreement with what we’re suggesting and support the idea that unless you have something that is unstable or active, stress testing isn’t likely to help.”
Annemarie Thompson, MD, of Duke University in Durham, N.C., another coauthor on the 2014 guidelines, commented in an interview that Dr. Rubin and colleagues’ findings of a doubled rate of complications among people without RCRI conditions who were stress tested, compared with those who were not might mean something “other than just sheer overuse or overordering of tests inappropriately.”
Rather, she said, physicians might be seeing something in the clinic that cannot be captured by a screening tool reliant on existing diagnoses. “Maybe when they’re sitting in front of you in a clinic, they’re so immobile that you’re left wondering. Or maybe they haven’t been seen by a doctor in a long time,” Dr. Thompson said. “So they don’t have diagnoses if they haven’t been followed. I think what [this finding] shows is that clinicians are detecting something. They may not know what it is. But we have to give a little wiggle room to the clinician who is sitting there looking at a patient who looks like they may not make it through surgery.”
Dr. Thompson said it would be helpful, after a big-data study like this one, to go through the clinical histories of those patients – in this study fewer than 100 – who had no RCRI risk factors and yet were stress tested and ended up having complications. “Until then we’re not going to solve the mystery,” she said. “But it’s a very, very interesting study.”
Dr. Rubin is the president of DRDR Mobile Health, a company that creates mobile applications for health care and from which he has not received compensation. One of his coauthors on the study, Dr. Peter Nagele, reported fee income from Roche Diagnostics. Dr. Beckman disclosed personal fees from AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, and other pharmaceutical manufacturers. Dr. Thompson has no disclosures.
SOURCE: Rubin et al. JAMA Cardiol. 2020 Sep 30. doi: 10.1001/jamacardio.2020.4311.
Cardiac stress testing before hip and knee replacements has dropped steadily since 2006, according to results from a new study that also showed major cardiac complications to be low in the absence of stress testing – even among people with established risk factors.
Routine stress testing before noncardiac surgeries has come under fire in recent decades as an overuse of resources and a burden on patients. Practice guidelines issued in 2007 and 2014 by the American College of Cardiology and the American Heart Association sought to limit the use of preoperative testing to patients with specific cardiovascular risk factors who might have their management changed by the test results.
For their study, published online in JAMA Cardiology, Daniel S. Rubin, MD, of the University of Chicago and colleagues looked at employee-based insurance data, which included Medicare Advantage claims, for more than 800,000 total hip or knee arthroplasties (28% hip and 72% knee replacements) conducted between 2004 and 2017.
While some 10% of the cohort (mean age 62, 58% women) received a stress test in the 2 months before surgery, the investigators found that the frequency of preoperative stress testing dropped annually starting in late 2006, when it peaked at about 14%, to about 7% in 2017. Older age, male sex and a Revised Cardiac Risk Index score of 1 or greater were all associated with a higher likelihood of being tested.
The overall frequency of myocardial infarction or cardiac arrest was 0.24%, occurring in 1,677 of 686,067 patients. While the rate was higher in patients with at least one RCRI condition, this did not differ significantly between those who received a preoperative stress test and those who did not (0.60%; 221 of 36,554 vs. 0.57%; 694 of 122,466 patients.
The 2007 and 2014 ACC/AHA guidelines make clear that patients with zero RCRI conditions – which comprise a history of ischemic heart disease, heart failure, insulin therapy for diabetes, cerebrovascular disease, or chronic kidney disease – should not receive a stress test before an intermediate-risk surgery such as a hip or knee replacement. But in this study, Dr. Rubin and his colleagues found that almost half of patients who had no RCRI risk factors were stress tested anyway. This means, Dr. Rubin said in an interview, that “there’s still room for improvement” in reducing testing.
“I never want to question how a physician chooses to practice, but I have to applaud physicians for reining in the use of this test. We’re using less of this test and yet the incidence of myocardial infarction and cardiac arrest is also going down, which also calls into question whether we’re getting better at choosing the right patients for the test; or the test doesn’t impact outcomes; or overall health of these patients is improving,” he said.
One surprise finding in the study, Dr. Rubin noted, was a higher rate of complications among people without RCRI conditions who were stress tested, compared with those who were not, with a mean complication rate of 0.27%, compared with 0.14% among those who did not receive a test (P < .001). “The RCRI doesn’t capture certain things,” Dr. Rubin said. “And we know that no risk stratification tool is going to capture everything.”
The RCRI, he noted, is based on a clinical history. “If you haven’t been diagnosed yet, it won’t appear as a risk factor, even if you’re clearly at risk. The question then becomes for a physician, do you do the test or not? On a day-to-day basis it’s hard to make that decision because you want what’s best for the individual patient – and it’s hard to generalize from a study of 800,000 people what’s right for that one patient. That said, it doesn’t appear that stress testing improves outcomes and a decrease in testing appears appropriate.”
Dr. Rubin and his colleagues described as a weakness of their study that it did not capture the full scope of preoperative stress testing among Medicare patients, who are older and therefore more likely to be tested.
That the 2007 and 2014 practice guidelines bore on the drop in testing was not demonstrated by Dr. Rubin and colleagues’ study, which saw declines begin even before the guidelines were published. Nonetheless, the results appear to validate the approach advocated in the guidelines, said guideline coauthor Joshua Beckman, MD, of Vanderbilt University, whose recent research has focused on identifying risk factors for MI after noncardiac surgery.
“I hope that the guidelines have helped in changing the culture for the use of preoperative stress testing as a regular thing,” Dr. Beckman said in an interview. “In fact, the guidelines say you shouldn’t do anything before an operation that you wouldn’t do anyway. So these findings are certainly in agreement with what we’re suggesting and support the idea that unless you have something that is unstable or active, stress testing isn’t likely to help.”
Annemarie Thompson, MD, of Duke University in Durham, N.C., another coauthor on the 2014 guidelines, commented in an interview that Dr. Rubin and colleagues’ findings of a doubled rate of complications among people without RCRI conditions who were stress tested, compared with those who were not might mean something “other than just sheer overuse or overordering of tests inappropriately.”
Rather, she said, physicians might be seeing something in the clinic that cannot be captured by a screening tool reliant on existing diagnoses. “Maybe when they’re sitting in front of you in a clinic, they’re so immobile that you’re left wondering. Or maybe they haven’t been seen by a doctor in a long time,” Dr. Thompson said. “So they don’t have diagnoses if they haven’t been followed. I think what [this finding] shows is that clinicians are detecting something. They may not know what it is. But we have to give a little wiggle room to the clinician who is sitting there looking at a patient who looks like they may not make it through surgery.”
Dr. Thompson said it would be helpful, after a big-data study like this one, to go through the clinical histories of those patients – in this study fewer than 100 – who had no RCRI risk factors and yet were stress tested and ended up having complications. “Until then we’re not going to solve the mystery,” she said. “But it’s a very, very interesting study.”
Dr. Rubin is the president of DRDR Mobile Health, a company that creates mobile applications for health care and from which he has not received compensation. One of his coauthors on the study, Dr. Peter Nagele, reported fee income from Roche Diagnostics. Dr. Beckman disclosed personal fees from AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, and other pharmaceutical manufacturers. Dr. Thompson has no disclosures.
SOURCE: Rubin et al. JAMA Cardiol. 2020 Sep 30. doi: 10.1001/jamacardio.2020.4311.
FROM JAMA CARDIOLOGY
AHA scientific statement highlights cardiorenal benefit of new diabetes drugs
To protect the heart and kidneys, sodium-glucose transporter 2 (SGLT2) inhibitors and glucagonlike peptide–1 (GLP-1) receptor agonists should be considered for people with type 2 diabetes and chronic kidney disease (CKD), the American Heart Association advised in a new scientific statement.
Taken together, the results of relevant clinical trials indicate that SGLT2 inhibitors and GLP-1 receptor agonists safely and significantly reduce the risk for cardiovascular (CV) events, death, and the slow progression of CKD to end-stage kidney disease, including the risks for dialysis, transplantation, and death, the writing group says.
The scientific statement was published online Sept. 28 in Circulation.
“There has been rapid reporting of high-quality data in the cardio-renal-metabolic space with significant heart and kidney benefits, particularly with these two newer classes of antihyperglycemic agents,” Janani Rangaswami, MD, who chaired the writing group, said in an interview.
“More recent data show benefits in chronic kidney disease and heart failure even in patients without diabetes,” said Dr. Rangaswami, Einstein Medical Center and Sidney Kimmel Medical College, both in Philadelphia.
“These data are practice-changing in both cardiology and nephrology, and usher in a new era of disease-modifying therapies in heart and kidney disease,” Dr. Rangaswami added.
Recommendations at a glance
- Provide early and ongoing assessment of risks for CVD and CKD to patients who may benefit from SGLT2 inhibitors of GLP-1 receptor agonists.
- Tailor medication choices that meet the needs of individual patients. Realize that, given “consistent class-wide effects,” the choice of a specific SGLT2 inhibitor or GLP-1 receptor agonist may be dictated by affordability, coverage, and formulary considerations.
- Adjust all medications in tandem with these medicines and consider the burden of polypharmacy, which is common among people with type 2 diabetes. Adjust concomitant therapies and deprescribe where possible.
- Identify risks for hypoglycemia and educate patients on the signs so they can seek treatment quickly.
- Monitor and control high blood pressure.
- Counsel patients about the risks for and symptoms of euglycemic diabetic ketoacidosis when taking SGLT2 inhibitors, as well as classic DKA, which can be fatal.
- Regularly screen and counsel patients about foot care to prevent foot ulcers or blisters that can quickly become infected and lead to amputation.
The writing group identified two additional patient subgroups that may benefit from SGLT2 inhibitors and GLP-1 receptor agonists: those with heart failure with reduced ejection fraction with or without diabetes; and those with CKD who do not have diabetes. They say more data are anticipated to validate the use of SGLT2 inhibitors and GLP-1 receptor agonists in these “at-risk” patients.
Collaborative care model
The writing group proposed a collaborative care model, bridging cardiologists, nephrologists, endocrinologists, and primary care physicians, to help facilitate the “prompt and appropriate” integration of these new classes of medications in the management of patients with type 2 diabetes and CKD.
There is “an unmet need for a cardio-renal-metabolic care model that incorporates best practices in the real world to help align these therapies, especially with vulnerable high-risk patients with cardiorenal disease, and to overcome barriers toward uptake of these agents. Hopefully this statement provides some guidance to the cardiology and nephrology communities in that area,” Dr. Rangaswami said in an interview.
But old habits die hard, as research continues to show the slow adoption of these newer medications in the real world.
For example, a large observational study published last year showed a “striking” discordance between evidence-based, guideline-recommended use of SGLT2 inhibitors for the treatment of type 2 diabetes and their actual uptake in clinical practice.
Paradoxically, patients with CVD, heart failure, hypertension, CKD, and those at risk for hypoglycemia were less apt to receive an SGLT2 inhibitor than other patients.
“The relatively slow uptake of these agents is multifactorial,” Dr. Rangaswami said. “Cardiologists and nephrologists may suffer from some level of ‘therapeutic inertia’ when using new agents they are unfamiliar with and originally branded as ‘antidiabetic’ agents, with the perception of these agents being outside the scope of their practice.”
Two other factors are also at play. “The current health care system is based on ‘specialty silos,’ where specialists tend to stick to the traditional scope of their specialty and are reluctant to view these agents as part of their therapeutic armamentarium. Finally, insurance coverage barriers and affordability also limit the use on a widespread basis,” Dr. Rangaswami said.
A version of this article originally appeared on Medscape.com .
To protect the heart and kidneys, sodium-glucose transporter 2 (SGLT2) inhibitors and glucagonlike peptide–1 (GLP-1) receptor agonists should be considered for people with type 2 diabetes and chronic kidney disease (CKD), the American Heart Association advised in a new scientific statement.
Taken together, the results of relevant clinical trials indicate that SGLT2 inhibitors and GLP-1 receptor agonists safely and significantly reduce the risk for cardiovascular (CV) events, death, and the slow progression of CKD to end-stage kidney disease, including the risks for dialysis, transplantation, and death, the writing group says.
The scientific statement was published online Sept. 28 in Circulation.
“There has been rapid reporting of high-quality data in the cardio-renal-metabolic space with significant heart and kidney benefits, particularly with these two newer classes of antihyperglycemic agents,” Janani Rangaswami, MD, who chaired the writing group, said in an interview.
“More recent data show benefits in chronic kidney disease and heart failure even in patients without diabetes,” said Dr. Rangaswami, Einstein Medical Center and Sidney Kimmel Medical College, both in Philadelphia.
“These data are practice-changing in both cardiology and nephrology, and usher in a new era of disease-modifying therapies in heart and kidney disease,” Dr. Rangaswami added.
Recommendations at a glance
- Provide early and ongoing assessment of risks for CVD and CKD to patients who may benefit from SGLT2 inhibitors of GLP-1 receptor agonists.
- Tailor medication choices that meet the needs of individual patients. Realize that, given “consistent class-wide effects,” the choice of a specific SGLT2 inhibitor or GLP-1 receptor agonist may be dictated by affordability, coverage, and formulary considerations.
- Adjust all medications in tandem with these medicines and consider the burden of polypharmacy, which is common among people with type 2 diabetes. Adjust concomitant therapies and deprescribe where possible.
- Identify risks for hypoglycemia and educate patients on the signs so they can seek treatment quickly.
- Monitor and control high blood pressure.
- Counsel patients about the risks for and symptoms of euglycemic diabetic ketoacidosis when taking SGLT2 inhibitors, as well as classic DKA, which can be fatal.
- Regularly screen and counsel patients about foot care to prevent foot ulcers or blisters that can quickly become infected and lead to amputation.
The writing group identified two additional patient subgroups that may benefit from SGLT2 inhibitors and GLP-1 receptor agonists: those with heart failure with reduced ejection fraction with or without diabetes; and those with CKD who do not have diabetes. They say more data are anticipated to validate the use of SGLT2 inhibitors and GLP-1 receptor agonists in these “at-risk” patients.
Collaborative care model
The writing group proposed a collaborative care model, bridging cardiologists, nephrologists, endocrinologists, and primary care physicians, to help facilitate the “prompt and appropriate” integration of these new classes of medications in the management of patients with type 2 diabetes and CKD.
There is “an unmet need for a cardio-renal-metabolic care model that incorporates best practices in the real world to help align these therapies, especially with vulnerable high-risk patients with cardiorenal disease, and to overcome barriers toward uptake of these agents. Hopefully this statement provides some guidance to the cardiology and nephrology communities in that area,” Dr. Rangaswami said in an interview.
But old habits die hard, as research continues to show the slow adoption of these newer medications in the real world.
For example, a large observational study published last year showed a “striking” discordance between evidence-based, guideline-recommended use of SGLT2 inhibitors for the treatment of type 2 diabetes and their actual uptake in clinical practice.
Paradoxically, patients with CVD, heart failure, hypertension, CKD, and those at risk for hypoglycemia were less apt to receive an SGLT2 inhibitor than other patients.
“The relatively slow uptake of these agents is multifactorial,” Dr. Rangaswami said. “Cardiologists and nephrologists may suffer from some level of ‘therapeutic inertia’ when using new agents they are unfamiliar with and originally branded as ‘antidiabetic’ agents, with the perception of these agents being outside the scope of their practice.”
Two other factors are also at play. “The current health care system is based on ‘specialty silos,’ where specialists tend to stick to the traditional scope of their specialty and are reluctant to view these agents as part of their therapeutic armamentarium. Finally, insurance coverage barriers and affordability also limit the use on a widespread basis,” Dr. Rangaswami said.
A version of this article originally appeared on Medscape.com .
To protect the heart and kidneys, sodium-glucose transporter 2 (SGLT2) inhibitors and glucagonlike peptide–1 (GLP-1) receptor agonists should be considered for people with type 2 diabetes and chronic kidney disease (CKD), the American Heart Association advised in a new scientific statement.
Taken together, the results of relevant clinical trials indicate that SGLT2 inhibitors and GLP-1 receptor agonists safely and significantly reduce the risk for cardiovascular (CV) events, death, and the slow progression of CKD to end-stage kidney disease, including the risks for dialysis, transplantation, and death, the writing group says.
The scientific statement was published online Sept. 28 in Circulation.
“There has been rapid reporting of high-quality data in the cardio-renal-metabolic space with significant heart and kidney benefits, particularly with these two newer classes of antihyperglycemic agents,” Janani Rangaswami, MD, who chaired the writing group, said in an interview.
“More recent data show benefits in chronic kidney disease and heart failure even in patients without diabetes,” said Dr. Rangaswami, Einstein Medical Center and Sidney Kimmel Medical College, both in Philadelphia.
“These data are practice-changing in both cardiology and nephrology, and usher in a new era of disease-modifying therapies in heart and kidney disease,” Dr. Rangaswami added.
Recommendations at a glance
- Provide early and ongoing assessment of risks for CVD and CKD to patients who may benefit from SGLT2 inhibitors of GLP-1 receptor agonists.
- Tailor medication choices that meet the needs of individual patients. Realize that, given “consistent class-wide effects,” the choice of a specific SGLT2 inhibitor or GLP-1 receptor agonist may be dictated by affordability, coverage, and formulary considerations.
- Adjust all medications in tandem with these medicines and consider the burden of polypharmacy, which is common among people with type 2 diabetes. Adjust concomitant therapies and deprescribe where possible.
- Identify risks for hypoglycemia and educate patients on the signs so they can seek treatment quickly.
- Monitor and control high blood pressure.
- Counsel patients about the risks for and symptoms of euglycemic diabetic ketoacidosis when taking SGLT2 inhibitors, as well as classic DKA, which can be fatal.
- Regularly screen and counsel patients about foot care to prevent foot ulcers or blisters that can quickly become infected and lead to amputation.
The writing group identified two additional patient subgroups that may benefit from SGLT2 inhibitors and GLP-1 receptor agonists: those with heart failure with reduced ejection fraction with or without diabetes; and those with CKD who do not have diabetes. They say more data are anticipated to validate the use of SGLT2 inhibitors and GLP-1 receptor agonists in these “at-risk” patients.
Collaborative care model
The writing group proposed a collaborative care model, bridging cardiologists, nephrologists, endocrinologists, and primary care physicians, to help facilitate the “prompt and appropriate” integration of these new classes of medications in the management of patients with type 2 diabetes and CKD.
There is “an unmet need for a cardio-renal-metabolic care model that incorporates best practices in the real world to help align these therapies, especially with vulnerable high-risk patients with cardiorenal disease, and to overcome barriers toward uptake of these agents. Hopefully this statement provides some guidance to the cardiology and nephrology communities in that area,” Dr. Rangaswami said in an interview.
But old habits die hard, as research continues to show the slow adoption of these newer medications in the real world.
For example, a large observational study published last year showed a “striking” discordance between evidence-based, guideline-recommended use of SGLT2 inhibitors for the treatment of type 2 diabetes and their actual uptake in clinical practice.
Paradoxically, patients with CVD, heart failure, hypertension, CKD, and those at risk for hypoglycemia were less apt to receive an SGLT2 inhibitor than other patients.
“The relatively slow uptake of these agents is multifactorial,” Dr. Rangaswami said. “Cardiologists and nephrologists may suffer from some level of ‘therapeutic inertia’ when using new agents they are unfamiliar with and originally branded as ‘antidiabetic’ agents, with the perception of these agents being outside the scope of their practice.”
Two other factors are also at play. “The current health care system is based on ‘specialty silos,’ where specialists tend to stick to the traditional scope of their specialty and are reluctant to view these agents as part of their therapeutic armamentarium. Finally, insurance coverage barriers and affordability also limit the use on a widespread basis,” Dr. Rangaswami said.
A version of this article originally appeared on Medscape.com .
Higher glycemic time in range may benefit T2D patients
Patients with type 2 or type 1 diabetes who stay in a blood glucose range of 70-180 mg/dL at least 70% of the time have the lowest rates of major adverse coronary events, severe hypoglycemic episodes, and microvascular events, according to a post hoc analysis of data collected from 5,774 patients with type 2 diabetes.
Data collected by the DEVOTE trial showed that every additional 10% of the time that a patient with type 2 diabetes (T2D) spent in their target range for blood glucose linked with a significant 6% reduced rate for developing a major adverse cardiovascular event (MACE), Richard M. Bergenstal, MD, said at the virtual annual meeting of the European Association for the Study of Diabetes.
For every 10% increase in time in range (TIR), patients showed an average 10% drop in their incidence of severe hypoglycemic episodes.
Increasing evidence from post hoc analyses
These findings confirmed a prior post hoc analysis of data collected in the DCCT trial (NCT00360815), which were published in the New England Journal of Medicine, although those results showed significant relationships between increased TIR and decreased rates of retinopathy and microalbuminuria. For every 10% drop in TIR, retinopathy rose by 64% and microalbuminuria increased by 40%, according to a post hoc analysis of the DCCT data that Dr. Bergenstal helped run and was published in Diabetes Care.
“It’s becoming clear that time in range is an important metric for diabetes management, and our new findings and those previously reported with the DCCT data make it look like time in range is becoming a good marker for clinical outcomes as well,” said Dr. Bergenstal, an endocrinologist at the Park Nicollet Clinic in Minneapolis.
“It’s a new concept, getting time-in-range data,” said Dr. Bergenstal, who was a coauthor of recommendations from Diabetes Care that were made in 2019 by an expert panel organized by the Advanced Technologies & Treatments for Diabetes Congress. “We think this will be a good marker to keep glycemia in a safe range, and the results look positive.” Patients who stay in the blood glucose range of 70-180 mg/dL (3.9-10.0 mmol/L) at least 70% of the time generally have an hemoglobin A1c of about 7%, which is what makes it a good target for patients and clinicians to focus on. Patients with a 50% TIR rate generally have an HbA1c of about 8%.
But a TIR assessment can be more informative than HbA1c, said the 2019 recommendations document. It called TIR assessments “appropriate and useful as clinical targets and outcome measurements that complement A1c for a wide range of people with diabetes.”
Data mining from DEVOTE
The analysis run by Dr. Bergenstal and his associates used data from 5,774 of the 7,637 patients enrolled in the DEVOTE trial, for whom adequate longitudinal blood glucose data were available to derive and track TIR. DEVOTE had the primary aim of comparing two different types of insulin in patients with T2D, according to its explanation in the New England Journal of Medicine. The DEVOTE patients did not undergo routine continuous blood glucose monitoring, so derivation of TIR was the only option with the dataset, Dr. Bergenstal said. “We’re trying to get continuous blood monitoring into T2D trials,” he said.
The post hoc analysis showed that, during the study’s follow-up of just under 2 years, patients who maintained a derived TIR of 70%-100% had about a 6% MACE rate, which peaked at nearly twice that in patients whose TIR was 30% or less. The analysis showed a roughly positive linear relationship between TIR and MACE rates across the range of TIR values. In an adjusted analysis, patients with at least a 70% TIR had a significant 31% lower rate of MACE events, compared with patients whose TIR was 50% or less.
A second analysis that looked for the association between TIR and incidence of hypoglycemic episodes showed a somewhat similar positive relationship, with incidence rates of severe hypoglycemia episodes of about 4%-5% among patients with a TIR of 70% or greater, and a rate of about 7% in patients with a TIR of 30% or less, spiking to 14% among patients with a TIR of 10% or less. In an adjusted analysis, patients with a TIR of at least 70% had a significant 46% lower rate of severe hypoglycemic events, compared with patients whose TIR was 50% or less. This finding belies a common misconception that the tighter glycemic control that produces a higher TIR will lead to increased episodes of severe hypoglycemia, Dr. Bergenstal noted.
He also reported less extensive data on microvascular events. In an adjusted analysis, patients with a TIR of at least 70% had a significant 40% cut in these events compared with patients with 50% or less TIR.
DEVOTE was funded by Novo Nordisk. Dr. Bergenstal has had financial relationships with Novo Nordisk and several other companies.
SOURCE: Bergenstal R et al. EASD 2020, abstract 159.
Patients with type 2 or type 1 diabetes who stay in a blood glucose range of 70-180 mg/dL at least 70% of the time have the lowest rates of major adverse coronary events, severe hypoglycemic episodes, and microvascular events, according to a post hoc analysis of data collected from 5,774 patients with type 2 diabetes.
Data collected by the DEVOTE trial showed that every additional 10% of the time that a patient with type 2 diabetes (T2D) spent in their target range for blood glucose linked with a significant 6% reduced rate for developing a major adverse cardiovascular event (MACE), Richard M. Bergenstal, MD, said at the virtual annual meeting of the European Association for the Study of Diabetes.
For every 10% increase in time in range (TIR), patients showed an average 10% drop in their incidence of severe hypoglycemic episodes.
Increasing evidence from post hoc analyses
These findings confirmed a prior post hoc analysis of data collected in the DCCT trial (NCT00360815), which were published in the New England Journal of Medicine, although those results showed significant relationships between increased TIR and decreased rates of retinopathy and microalbuminuria. For every 10% drop in TIR, retinopathy rose by 64% and microalbuminuria increased by 40%, according to a post hoc analysis of the DCCT data that Dr. Bergenstal helped run and was published in Diabetes Care.
“It’s becoming clear that time in range is an important metric for diabetes management, and our new findings and those previously reported with the DCCT data make it look like time in range is becoming a good marker for clinical outcomes as well,” said Dr. Bergenstal, an endocrinologist at the Park Nicollet Clinic in Minneapolis.
“It’s a new concept, getting time-in-range data,” said Dr. Bergenstal, who was a coauthor of recommendations from Diabetes Care that were made in 2019 by an expert panel organized by the Advanced Technologies & Treatments for Diabetes Congress. “We think this will be a good marker to keep glycemia in a safe range, and the results look positive.” Patients who stay in the blood glucose range of 70-180 mg/dL (3.9-10.0 mmol/L) at least 70% of the time generally have an hemoglobin A1c of about 7%, which is what makes it a good target for patients and clinicians to focus on. Patients with a 50% TIR rate generally have an HbA1c of about 8%.
But a TIR assessment can be more informative than HbA1c, said the 2019 recommendations document. It called TIR assessments “appropriate and useful as clinical targets and outcome measurements that complement A1c for a wide range of people with diabetes.”
Data mining from DEVOTE
The analysis run by Dr. Bergenstal and his associates used data from 5,774 of the 7,637 patients enrolled in the DEVOTE trial, for whom adequate longitudinal blood glucose data were available to derive and track TIR. DEVOTE had the primary aim of comparing two different types of insulin in patients with T2D, according to its explanation in the New England Journal of Medicine. The DEVOTE patients did not undergo routine continuous blood glucose monitoring, so derivation of TIR was the only option with the dataset, Dr. Bergenstal said. “We’re trying to get continuous blood monitoring into T2D trials,” he said.
The post hoc analysis showed that, during the study’s follow-up of just under 2 years, patients who maintained a derived TIR of 70%-100% had about a 6% MACE rate, which peaked at nearly twice that in patients whose TIR was 30% or less. The analysis showed a roughly positive linear relationship between TIR and MACE rates across the range of TIR values. In an adjusted analysis, patients with at least a 70% TIR had a significant 31% lower rate of MACE events, compared with patients whose TIR was 50% or less.
A second analysis that looked for the association between TIR and incidence of hypoglycemic episodes showed a somewhat similar positive relationship, with incidence rates of severe hypoglycemia episodes of about 4%-5% among patients with a TIR of 70% or greater, and a rate of about 7% in patients with a TIR of 30% or less, spiking to 14% among patients with a TIR of 10% or less. In an adjusted analysis, patients with a TIR of at least 70% had a significant 46% lower rate of severe hypoglycemic events, compared with patients whose TIR was 50% or less. This finding belies a common misconception that the tighter glycemic control that produces a higher TIR will lead to increased episodes of severe hypoglycemia, Dr. Bergenstal noted.
He also reported less extensive data on microvascular events. In an adjusted analysis, patients with a TIR of at least 70% had a significant 40% cut in these events compared with patients with 50% or less TIR.
DEVOTE was funded by Novo Nordisk. Dr. Bergenstal has had financial relationships with Novo Nordisk and several other companies.
SOURCE: Bergenstal R et al. EASD 2020, abstract 159.
Patients with type 2 or type 1 diabetes who stay in a blood glucose range of 70-180 mg/dL at least 70% of the time have the lowest rates of major adverse coronary events, severe hypoglycemic episodes, and microvascular events, according to a post hoc analysis of data collected from 5,774 patients with type 2 diabetes.
Data collected by the DEVOTE trial showed that every additional 10% of the time that a patient with type 2 diabetes (T2D) spent in their target range for blood glucose linked with a significant 6% reduced rate for developing a major adverse cardiovascular event (MACE), Richard M. Bergenstal, MD, said at the virtual annual meeting of the European Association for the Study of Diabetes.
For every 10% increase in time in range (TIR), patients showed an average 10% drop in their incidence of severe hypoglycemic episodes.
Increasing evidence from post hoc analyses
These findings confirmed a prior post hoc analysis of data collected in the DCCT trial (NCT00360815), which were published in the New England Journal of Medicine, although those results showed significant relationships between increased TIR and decreased rates of retinopathy and microalbuminuria. For every 10% drop in TIR, retinopathy rose by 64% and microalbuminuria increased by 40%, according to a post hoc analysis of the DCCT data that Dr. Bergenstal helped run and was published in Diabetes Care.
“It’s becoming clear that time in range is an important metric for diabetes management, and our new findings and those previously reported with the DCCT data make it look like time in range is becoming a good marker for clinical outcomes as well,” said Dr. Bergenstal, an endocrinologist at the Park Nicollet Clinic in Minneapolis.
“It’s a new concept, getting time-in-range data,” said Dr. Bergenstal, who was a coauthor of recommendations from Diabetes Care that were made in 2019 by an expert panel organized by the Advanced Technologies & Treatments for Diabetes Congress. “We think this will be a good marker to keep glycemia in a safe range, and the results look positive.” Patients who stay in the blood glucose range of 70-180 mg/dL (3.9-10.0 mmol/L) at least 70% of the time generally have an hemoglobin A1c of about 7%, which is what makes it a good target for patients and clinicians to focus on. Patients with a 50% TIR rate generally have an HbA1c of about 8%.
But a TIR assessment can be more informative than HbA1c, said the 2019 recommendations document. It called TIR assessments “appropriate and useful as clinical targets and outcome measurements that complement A1c for a wide range of people with diabetes.”
Data mining from DEVOTE
The analysis run by Dr. Bergenstal and his associates used data from 5,774 of the 7,637 patients enrolled in the DEVOTE trial, for whom adequate longitudinal blood glucose data were available to derive and track TIR. DEVOTE had the primary aim of comparing two different types of insulin in patients with T2D, according to its explanation in the New England Journal of Medicine. The DEVOTE patients did not undergo routine continuous blood glucose monitoring, so derivation of TIR was the only option with the dataset, Dr. Bergenstal said. “We’re trying to get continuous blood monitoring into T2D trials,” he said.
The post hoc analysis showed that, during the study’s follow-up of just under 2 years, patients who maintained a derived TIR of 70%-100% had about a 6% MACE rate, which peaked at nearly twice that in patients whose TIR was 30% or less. The analysis showed a roughly positive linear relationship between TIR and MACE rates across the range of TIR values. In an adjusted analysis, patients with at least a 70% TIR had a significant 31% lower rate of MACE events, compared with patients whose TIR was 50% or less.
A second analysis that looked for the association between TIR and incidence of hypoglycemic episodes showed a somewhat similar positive relationship, with incidence rates of severe hypoglycemia episodes of about 4%-5% among patients with a TIR of 70% or greater, and a rate of about 7% in patients with a TIR of 30% or less, spiking to 14% among patients with a TIR of 10% or less. In an adjusted analysis, patients with a TIR of at least 70% had a significant 46% lower rate of severe hypoglycemic events, compared with patients whose TIR was 50% or less. This finding belies a common misconception that the tighter glycemic control that produces a higher TIR will lead to increased episodes of severe hypoglycemia, Dr. Bergenstal noted.
He also reported less extensive data on microvascular events. In an adjusted analysis, patients with a TIR of at least 70% had a significant 40% cut in these events compared with patients with 50% or less TIR.
DEVOTE was funded by Novo Nordisk. Dr. Bergenstal has had financial relationships with Novo Nordisk and several other companies.
SOURCE: Bergenstal R et al. EASD 2020, abstract 159.
FROM EASD 2020
Exercise cuts diabetes death risk by a third in two studies
Type 2 diabetes patients could lower their risk for death from any cause by up to a third by exercising at a moderate to high level or by cycling, according to data from two studies reported at the virtual annual meeting of the European Association for the Study of Diabetes.
Yun-Ju Lai, MD, and colleagues from the Puli branch of Taichung Veterans General Hospital in Nantou, Taiwan, found that persons with type 2 diabetes who exercised at moderate to high intensity had a 25%-32% decreased risk for death, compared with those who did not exercise.
In a separate study, Mathias Ried-Larsen, MSc, PhD, group leader at the Centre for Physical Activity Research, Rigshospitalet, Copenhagen, and associates found that cycling was associated with a 25%-31% decreased risk for all-cause death compared to no cycling, and that cycling also reduced cardiovascular mortality.
Results fit with ADA recommendations
“There is really nothing surprising about these results as others have shown that regular participation in physical activity lowers both overall mortality rates and morbidity,” commented Sheri Colberg-Ochs, PhD, professor emerita in exercise science at Old Dominion University in Norfolk, Va., in an interview.
“Regular exercise participation lowers the risk of mortality in almost all populations with many different health conditions. It is not specific to people with type 2 diabetes,” Dr. Colberg-Ochs said. “These data add further support to the ADA [American Diabetes Association] recommendations by again suggesting that being more active leads to many health benefits for people with type 2 diabetes.”
Dr. Colberg-Ochs, who was not involved in either study, is recognized by the ADA as an Outstanding Educator in Diabetes. She was also involved in writing the ADA’s position statement on physical activity/exercise in diabetes, which advocate that adults with type 2 diabetes should reduce sedentary time and undertake both aerobic and resistance exercise training to help optimize their glycemic and general health outcomes.
Asian population understudied
In an interview Dr. Lai acknowledged that epidemiologic studies had shown that exercise reduced the risk of cardiovascular events and mortality in subjects with type 2 diabetes. “However, the dose of exercise capacity for reducing mortality risk in people with type 2 diabetes was not yet well investigated, especially in the Asian population.”
Dr. Lai and colleagues analyzed data on 4,859 subjects drawn from two Taiwanese databases – the National Health Interview Survey and the National Health Insurance research database – to study what effect exercise “capacity” had on the risk for death in those with type 2 diabetes.
“Information about physical activity during leisure time was collected by asking the questions: ‘How often do you exercise every week? What kind of exercise do you do? How long do you do the exercise?’, Dr. Lai said. “We included nearly all kinds of exercise in the analysis, such as jogging, swimming, walking, dancing, riding, and so on.”
Each exercise had an activity intensity code expressed as kilocalories per minute. This was used to determine the exercise “capacity” by multiplying it by how frequently the exercise was performed per week and for how long each time.
“I don’t think ‘capacity’ is the right word to use here. The equation they used describes their exercise ‘volume,’ not their capacity. Self-reported exercise is notoriously inaccurate,” Dr. Colberg-Ochs observed. Furthermore, “just asking people how much they exercise and at what intensity [without using a validated exercise questionnaire] gives questionable results.”
The study’s findings, however, were clear: Those who exercised at a higher level had a significantly decreased risk for all-cause mortality than did those with no exercise habits. The hazard ratio for death by any cause was 0.75 for those who undertook a moderate level of exercise, burning 0-800 kcal per week. Exercising at a higher level burned more than 800 kcal had a HR of 0.68. A significant (P < .01) trend in favor of more exercise was noted.
Cycling reduces all-cause and cardiovascular mortality
In their prospective cohort study, Dr. Ried-Larsen and associates took a more specific look at the effects of exercise on mortality in diabetes by studying a single exercise: cycling. They sampled data on more than 5,000 people collected as part of the European Prospective Investigation into Cancer and Nutrition study. First, they identified participants with diabetes – although they couldn’t distinguish type 1 from type 2 forms because this was self-reported or obtained from registries. They then identified those who reported cycling at their baseline assessment and those who reported a change in cycling habits at their second examination around 5 years later.
At baseline, 38% of participants reported that they cycled every week. The mean age was 56 years, diabetes duration was 8 years, one-fifth were smokers, and the average body mass index was 29 kg/m2.
Participants who reported cycling up to 1 hour every week at baseline had a 25% reduction in all-cause mortality, compared with those who did not cycle. The biggest reduction (31%) in all-cause mortality was seen for cycling 2.5-5 hours a week; cycling for 1-2.5 hours, and for more than 5 hours, yielded 23% and 24% risk reductions, respectively.
A reverse J–shaped relationship between cycling duration and reduction in all-cause mortality was seen, Dr. Ried-Larsen noted during a live oral session at the virtual meeting. “The maximum benefit [was at] around 5 hours per week, and the benefits persisted until around 9 hours per week.” Adjustment for the prevalence of stroke, MI, cancer, hyperlipidemia, hypertension, and central obesity did not alter the findings.
“The direction of the association was the same for cardiovascular mortality as all-cause mortality, although a bit weaker, with the maximum benefit being around 4 hours per week, and that persisted up until around 8 hours per week,” Dr. Ried-Larsen said.
The benefits of cycling on all-cause and cardiovascular mortality were lost, however, if those who cycled at baseline stopped by the second examination. Those who did not cycle at the first but did at the second examination got a benefit on both, as did those who continued cycling.
“Cycling is among one of the preferred activities for diabetes patients, so it actually may help them to achieve the recommend level of physical activity,” Dr. Ried-Larsen said.
Tailored exercise program important
Advice for exercise “should be tailored to the individual and based on starting fitness levels and activity levels,” Dr. Colberg-Ochs recommended.
“Those who are the most sedentary and the least fit have the most to gain from doing any activity. They should be advised to start out slowly and progress slowly with both aerobic activities and some resistance training,” Dr. Colberg-Ochs said.
She added: “In addition, individuals over 40 should engage in regular balance training, and all individuals should do some flexibility exercises.”
The studies received no commercial funding and all those mentioned in this article had no conflicts of interest to disclose.
SOURCE: Lai Y-J et al. EASD 2020, Poster presentation 267; Ried-Larsen M et al. EASD 2020, Oral presentation 194.
Type 2 diabetes patients could lower their risk for death from any cause by up to a third by exercising at a moderate to high level or by cycling, according to data from two studies reported at the virtual annual meeting of the European Association for the Study of Diabetes.
Yun-Ju Lai, MD, and colleagues from the Puli branch of Taichung Veterans General Hospital in Nantou, Taiwan, found that persons with type 2 diabetes who exercised at moderate to high intensity had a 25%-32% decreased risk for death, compared with those who did not exercise.
In a separate study, Mathias Ried-Larsen, MSc, PhD, group leader at the Centre for Physical Activity Research, Rigshospitalet, Copenhagen, and associates found that cycling was associated with a 25%-31% decreased risk for all-cause death compared to no cycling, and that cycling also reduced cardiovascular mortality.
Results fit with ADA recommendations
“There is really nothing surprising about these results as others have shown that regular participation in physical activity lowers both overall mortality rates and morbidity,” commented Sheri Colberg-Ochs, PhD, professor emerita in exercise science at Old Dominion University in Norfolk, Va., in an interview.
“Regular exercise participation lowers the risk of mortality in almost all populations with many different health conditions. It is not specific to people with type 2 diabetes,” Dr. Colberg-Ochs said. “These data add further support to the ADA [American Diabetes Association] recommendations by again suggesting that being more active leads to many health benefits for people with type 2 diabetes.”
Dr. Colberg-Ochs, who was not involved in either study, is recognized by the ADA as an Outstanding Educator in Diabetes. She was also involved in writing the ADA’s position statement on physical activity/exercise in diabetes, which advocate that adults with type 2 diabetes should reduce sedentary time and undertake both aerobic and resistance exercise training to help optimize their glycemic and general health outcomes.
Asian population understudied
In an interview Dr. Lai acknowledged that epidemiologic studies had shown that exercise reduced the risk of cardiovascular events and mortality in subjects with type 2 diabetes. “However, the dose of exercise capacity for reducing mortality risk in people with type 2 diabetes was not yet well investigated, especially in the Asian population.”
Dr. Lai and colleagues analyzed data on 4,859 subjects drawn from two Taiwanese databases – the National Health Interview Survey and the National Health Insurance research database – to study what effect exercise “capacity” had on the risk for death in those with type 2 diabetes.
“Information about physical activity during leisure time was collected by asking the questions: ‘How often do you exercise every week? What kind of exercise do you do? How long do you do the exercise?’, Dr. Lai said. “We included nearly all kinds of exercise in the analysis, such as jogging, swimming, walking, dancing, riding, and so on.”
Each exercise had an activity intensity code expressed as kilocalories per minute. This was used to determine the exercise “capacity” by multiplying it by how frequently the exercise was performed per week and for how long each time.
“I don’t think ‘capacity’ is the right word to use here. The equation they used describes their exercise ‘volume,’ not their capacity. Self-reported exercise is notoriously inaccurate,” Dr. Colberg-Ochs observed. Furthermore, “just asking people how much they exercise and at what intensity [without using a validated exercise questionnaire] gives questionable results.”
The study’s findings, however, were clear: Those who exercised at a higher level had a significantly decreased risk for all-cause mortality than did those with no exercise habits. The hazard ratio for death by any cause was 0.75 for those who undertook a moderate level of exercise, burning 0-800 kcal per week. Exercising at a higher level burned more than 800 kcal had a HR of 0.68. A significant (P < .01) trend in favor of more exercise was noted.
Cycling reduces all-cause and cardiovascular mortality
In their prospective cohort study, Dr. Ried-Larsen and associates took a more specific look at the effects of exercise on mortality in diabetes by studying a single exercise: cycling. They sampled data on more than 5,000 people collected as part of the European Prospective Investigation into Cancer and Nutrition study. First, they identified participants with diabetes – although they couldn’t distinguish type 1 from type 2 forms because this was self-reported or obtained from registries. They then identified those who reported cycling at their baseline assessment and those who reported a change in cycling habits at their second examination around 5 years later.
At baseline, 38% of participants reported that they cycled every week. The mean age was 56 years, diabetes duration was 8 years, one-fifth were smokers, and the average body mass index was 29 kg/m2.
Participants who reported cycling up to 1 hour every week at baseline had a 25% reduction in all-cause mortality, compared with those who did not cycle. The biggest reduction (31%) in all-cause mortality was seen for cycling 2.5-5 hours a week; cycling for 1-2.5 hours, and for more than 5 hours, yielded 23% and 24% risk reductions, respectively.
A reverse J–shaped relationship between cycling duration and reduction in all-cause mortality was seen, Dr. Ried-Larsen noted during a live oral session at the virtual meeting. “The maximum benefit [was at] around 5 hours per week, and the benefits persisted until around 9 hours per week.” Adjustment for the prevalence of stroke, MI, cancer, hyperlipidemia, hypertension, and central obesity did not alter the findings.
“The direction of the association was the same for cardiovascular mortality as all-cause mortality, although a bit weaker, with the maximum benefit being around 4 hours per week, and that persisted up until around 8 hours per week,” Dr. Ried-Larsen said.
The benefits of cycling on all-cause and cardiovascular mortality were lost, however, if those who cycled at baseline stopped by the second examination. Those who did not cycle at the first but did at the second examination got a benefit on both, as did those who continued cycling.
“Cycling is among one of the preferred activities for diabetes patients, so it actually may help them to achieve the recommend level of physical activity,” Dr. Ried-Larsen said.
Tailored exercise program important
Advice for exercise “should be tailored to the individual and based on starting fitness levels and activity levels,” Dr. Colberg-Ochs recommended.
“Those who are the most sedentary and the least fit have the most to gain from doing any activity. They should be advised to start out slowly and progress slowly with both aerobic activities and some resistance training,” Dr. Colberg-Ochs said.
She added: “In addition, individuals over 40 should engage in regular balance training, and all individuals should do some flexibility exercises.”
The studies received no commercial funding and all those mentioned in this article had no conflicts of interest to disclose.
SOURCE: Lai Y-J et al. EASD 2020, Poster presentation 267; Ried-Larsen M et al. EASD 2020, Oral presentation 194.
Type 2 diabetes patients could lower their risk for death from any cause by up to a third by exercising at a moderate to high level or by cycling, according to data from two studies reported at the virtual annual meeting of the European Association for the Study of Diabetes.
Yun-Ju Lai, MD, and colleagues from the Puli branch of Taichung Veterans General Hospital in Nantou, Taiwan, found that persons with type 2 diabetes who exercised at moderate to high intensity had a 25%-32% decreased risk for death, compared with those who did not exercise.
In a separate study, Mathias Ried-Larsen, MSc, PhD, group leader at the Centre for Physical Activity Research, Rigshospitalet, Copenhagen, and associates found that cycling was associated with a 25%-31% decreased risk for all-cause death compared to no cycling, and that cycling also reduced cardiovascular mortality.
Results fit with ADA recommendations
“There is really nothing surprising about these results as others have shown that regular participation in physical activity lowers both overall mortality rates and morbidity,” commented Sheri Colberg-Ochs, PhD, professor emerita in exercise science at Old Dominion University in Norfolk, Va., in an interview.
“Regular exercise participation lowers the risk of mortality in almost all populations with many different health conditions. It is not specific to people with type 2 diabetes,” Dr. Colberg-Ochs said. “These data add further support to the ADA [American Diabetes Association] recommendations by again suggesting that being more active leads to many health benefits for people with type 2 diabetes.”
Dr. Colberg-Ochs, who was not involved in either study, is recognized by the ADA as an Outstanding Educator in Diabetes. She was also involved in writing the ADA’s position statement on physical activity/exercise in diabetes, which advocate that adults with type 2 diabetes should reduce sedentary time and undertake both aerobic and resistance exercise training to help optimize their glycemic and general health outcomes.
Asian population understudied
In an interview Dr. Lai acknowledged that epidemiologic studies had shown that exercise reduced the risk of cardiovascular events and mortality in subjects with type 2 diabetes. “However, the dose of exercise capacity for reducing mortality risk in people with type 2 diabetes was not yet well investigated, especially in the Asian population.”
Dr. Lai and colleagues analyzed data on 4,859 subjects drawn from two Taiwanese databases – the National Health Interview Survey and the National Health Insurance research database – to study what effect exercise “capacity” had on the risk for death in those with type 2 diabetes.
“Information about physical activity during leisure time was collected by asking the questions: ‘How often do you exercise every week? What kind of exercise do you do? How long do you do the exercise?’, Dr. Lai said. “We included nearly all kinds of exercise in the analysis, such as jogging, swimming, walking, dancing, riding, and so on.”
Each exercise had an activity intensity code expressed as kilocalories per minute. This was used to determine the exercise “capacity” by multiplying it by how frequently the exercise was performed per week and for how long each time.
“I don’t think ‘capacity’ is the right word to use here. The equation they used describes their exercise ‘volume,’ not their capacity. Self-reported exercise is notoriously inaccurate,” Dr. Colberg-Ochs observed. Furthermore, “just asking people how much they exercise and at what intensity [without using a validated exercise questionnaire] gives questionable results.”
The study’s findings, however, were clear: Those who exercised at a higher level had a significantly decreased risk for all-cause mortality than did those with no exercise habits. The hazard ratio for death by any cause was 0.75 for those who undertook a moderate level of exercise, burning 0-800 kcal per week. Exercising at a higher level burned more than 800 kcal had a HR of 0.68. A significant (P < .01) trend in favor of more exercise was noted.
Cycling reduces all-cause and cardiovascular mortality
In their prospective cohort study, Dr. Ried-Larsen and associates took a more specific look at the effects of exercise on mortality in diabetes by studying a single exercise: cycling. They sampled data on more than 5,000 people collected as part of the European Prospective Investigation into Cancer and Nutrition study. First, they identified participants with diabetes – although they couldn’t distinguish type 1 from type 2 forms because this was self-reported or obtained from registries. They then identified those who reported cycling at their baseline assessment and those who reported a change in cycling habits at their second examination around 5 years later.
At baseline, 38% of participants reported that they cycled every week. The mean age was 56 years, diabetes duration was 8 years, one-fifth were smokers, and the average body mass index was 29 kg/m2.
Participants who reported cycling up to 1 hour every week at baseline had a 25% reduction in all-cause mortality, compared with those who did not cycle. The biggest reduction (31%) in all-cause mortality was seen for cycling 2.5-5 hours a week; cycling for 1-2.5 hours, and for more than 5 hours, yielded 23% and 24% risk reductions, respectively.
A reverse J–shaped relationship between cycling duration and reduction in all-cause mortality was seen, Dr. Ried-Larsen noted during a live oral session at the virtual meeting. “The maximum benefit [was at] around 5 hours per week, and the benefits persisted until around 9 hours per week.” Adjustment for the prevalence of stroke, MI, cancer, hyperlipidemia, hypertension, and central obesity did not alter the findings.
“The direction of the association was the same for cardiovascular mortality as all-cause mortality, although a bit weaker, with the maximum benefit being around 4 hours per week, and that persisted up until around 8 hours per week,” Dr. Ried-Larsen said.
The benefits of cycling on all-cause and cardiovascular mortality were lost, however, if those who cycled at baseline stopped by the second examination. Those who did not cycle at the first but did at the second examination got a benefit on both, as did those who continued cycling.
“Cycling is among one of the preferred activities for diabetes patients, so it actually may help them to achieve the recommend level of physical activity,” Dr. Ried-Larsen said.
Tailored exercise program important
Advice for exercise “should be tailored to the individual and based on starting fitness levels and activity levels,” Dr. Colberg-Ochs recommended.
“Those who are the most sedentary and the least fit have the most to gain from doing any activity. They should be advised to start out slowly and progress slowly with both aerobic activities and some resistance training,” Dr. Colberg-Ochs said.
She added: “In addition, individuals over 40 should engage in regular balance training, and all individuals should do some flexibility exercises.”
The studies received no commercial funding and all those mentioned in this article had no conflicts of interest to disclose.
SOURCE: Lai Y-J et al. EASD 2020, Poster presentation 267; Ried-Larsen M et al. EASD 2020, Oral presentation 194.
FROM EASD 2020