User login
Be wary of ‘for eczema’ claims on labels of popular moisturizers
Be wary of “for eczema” advertising claims contained on the labels of popular skin moisturizers.
Results from a study presented during the virtual annual meeting of the Society for Pediatric Dermatology found that .
“Prescription medications are important for managing eczema flares, but a lot of the work in treating eczema is preventative, done by consistently moisturizing the skin at home with drug store products,” co-first study author Catherine L. Ludwig, said in an interview. “Allergic contact dermatitis occurs more commonly in people with eczema. A previous study was done in characterizing the allergenic potential of drug store moisturizers and found that 88% of moisturizers contain at least one common allergen. Many moisturizers are marketed specifically to eczema, but the allergen content of these products are unknown.”
For the current study, Ms. Ludwig, a medical student at the University of Illinois at Chicago and co-first author Alyssa M. Thompson, a medical student at the University of Arizona, Tucson, and their colleagues compiled a list of the top 30 moisturizers “for eczema” sold by Amazon, Target, and Walmart. For each moisturizer they recorded common ingredients and marketing claims related to benefits for atopic dermatitis, including eczema relief, sensitive/gentle skin, hypoallergenic, anti-itch, anti-inflammatory, clinically proven, oatmeal, dermatologist recommended/approved, organic, fragrance-free, for baby, or National Eczema Association approved. To establish allergenic potential, the researchers used MATLAB to compare ingredient lists to compounds listed as common allergens in the American Contact Dermatitis Society’s Contact Allergen Management Program database (ACDS CAMP). Next, they used the Mann-Whitney U test to evaluate differences in allergen count between products with and without specific marketing claims.
Ms. Ludwig and her associates found that 28 of 30 products analyzed (93%) contained at least one allergen, with an overall average allergen count of 3.60. The three most prevalent allergens were cetyl alcohol (70%), phenoxyethanol (50%), and aloe (33%). “Anti-inflammatory” moisturizers had the greatest average number of allergens (4.00), followed by “anti-itch” (3.71) and “oatmeal” (3.71). Only products claiming to be “hypoallergenic” had significantly lower allergenic ingredient count (an average of 2.45) than those without the claim (P = .011).
“It was validating to see that eczema moisturizer products marketed as ‘hypoallergenic’ truly do have fewer allergenic ingredients than moisturizers without the claim,” Ms. Ludwig said. “However, it was surprising to see that even products marketed to eczema patients, who have a higher prevalence of allergic contact dermatitis, contain an average of 3.6 common allergens. As dermatology providers, we can relay to patients and parents that relying solely on ‘for eczema’ claims is not advisable. Clinicians should acquaint themselves with the top allergens (cetyl alcohol, phenoxyethanol, and aloe) and keep these ingredients, as well as affordability and patient preferences, in mind when making product recommendations.”
The study’s senior author, Vivian Y. Shi, MD, is a stock shareholder of Learn Health and has served as an advisory board member and/or investigator, and/or received research funding from AbbVie, Burt’s Bees, GpSkin, LEO Pharma, Eli Lilly, Menlo Therapeutics, Novartis, Pfizer, Regeneron, Sanofi Genzyme, Skin Actives Scientific, and SUN Pharma, and the Foundation for Atopic Dermatitis, Global Parents for Eczema Research, and the National Eczema Association. The other study authors reported having no financial disclosures.
Be wary of “for eczema” advertising claims contained on the labels of popular skin moisturizers.
Results from a study presented during the virtual annual meeting of the Society for Pediatric Dermatology found that .
“Prescription medications are important for managing eczema flares, but a lot of the work in treating eczema is preventative, done by consistently moisturizing the skin at home with drug store products,” co-first study author Catherine L. Ludwig, said in an interview. “Allergic contact dermatitis occurs more commonly in people with eczema. A previous study was done in characterizing the allergenic potential of drug store moisturizers and found that 88% of moisturizers contain at least one common allergen. Many moisturizers are marketed specifically to eczema, but the allergen content of these products are unknown.”
For the current study, Ms. Ludwig, a medical student at the University of Illinois at Chicago and co-first author Alyssa M. Thompson, a medical student at the University of Arizona, Tucson, and their colleagues compiled a list of the top 30 moisturizers “for eczema” sold by Amazon, Target, and Walmart. For each moisturizer they recorded common ingredients and marketing claims related to benefits for atopic dermatitis, including eczema relief, sensitive/gentle skin, hypoallergenic, anti-itch, anti-inflammatory, clinically proven, oatmeal, dermatologist recommended/approved, organic, fragrance-free, for baby, or National Eczema Association approved. To establish allergenic potential, the researchers used MATLAB to compare ingredient lists to compounds listed as common allergens in the American Contact Dermatitis Society’s Contact Allergen Management Program database (ACDS CAMP). Next, they used the Mann-Whitney U test to evaluate differences in allergen count between products with and without specific marketing claims.
Ms. Ludwig and her associates found that 28 of 30 products analyzed (93%) contained at least one allergen, with an overall average allergen count of 3.60. The three most prevalent allergens were cetyl alcohol (70%), phenoxyethanol (50%), and aloe (33%). “Anti-inflammatory” moisturizers had the greatest average number of allergens (4.00), followed by “anti-itch” (3.71) and “oatmeal” (3.71). Only products claiming to be “hypoallergenic” had significantly lower allergenic ingredient count (an average of 2.45) than those without the claim (P = .011).
“It was validating to see that eczema moisturizer products marketed as ‘hypoallergenic’ truly do have fewer allergenic ingredients than moisturizers without the claim,” Ms. Ludwig said. “However, it was surprising to see that even products marketed to eczema patients, who have a higher prevalence of allergic contact dermatitis, contain an average of 3.6 common allergens. As dermatology providers, we can relay to patients and parents that relying solely on ‘for eczema’ claims is not advisable. Clinicians should acquaint themselves with the top allergens (cetyl alcohol, phenoxyethanol, and aloe) and keep these ingredients, as well as affordability and patient preferences, in mind when making product recommendations.”
The study’s senior author, Vivian Y. Shi, MD, is a stock shareholder of Learn Health and has served as an advisory board member and/or investigator, and/or received research funding from AbbVie, Burt’s Bees, GpSkin, LEO Pharma, Eli Lilly, Menlo Therapeutics, Novartis, Pfizer, Regeneron, Sanofi Genzyme, Skin Actives Scientific, and SUN Pharma, and the Foundation for Atopic Dermatitis, Global Parents for Eczema Research, and the National Eczema Association. The other study authors reported having no financial disclosures.
Be wary of “for eczema” advertising claims contained on the labels of popular skin moisturizers.
Results from a study presented during the virtual annual meeting of the Society for Pediatric Dermatology found that .
“Prescription medications are important for managing eczema flares, but a lot of the work in treating eczema is preventative, done by consistently moisturizing the skin at home with drug store products,” co-first study author Catherine L. Ludwig, said in an interview. “Allergic contact dermatitis occurs more commonly in people with eczema. A previous study was done in characterizing the allergenic potential of drug store moisturizers and found that 88% of moisturizers contain at least one common allergen. Many moisturizers are marketed specifically to eczema, but the allergen content of these products are unknown.”
For the current study, Ms. Ludwig, a medical student at the University of Illinois at Chicago and co-first author Alyssa M. Thompson, a medical student at the University of Arizona, Tucson, and their colleagues compiled a list of the top 30 moisturizers “for eczema” sold by Amazon, Target, and Walmart. For each moisturizer they recorded common ingredients and marketing claims related to benefits for atopic dermatitis, including eczema relief, sensitive/gentle skin, hypoallergenic, anti-itch, anti-inflammatory, clinically proven, oatmeal, dermatologist recommended/approved, organic, fragrance-free, for baby, or National Eczema Association approved. To establish allergenic potential, the researchers used MATLAB to compare ingredient lists to compounds listed as common allergens in the American Contact Dermatitis Society’s Contact Allergen Management Program database (ACDS CAMP). Next, they used the Mann-Whitney U test to evaluate differences in allergen count between products with and without specific marketing claims.
Ms. Ludwig and her associates found that 28 of 30 products analyzed (93%) contained at least one allergen, with an overall average allergen count of 3.60. The three most prevalent allergens were cetyl alcohol (70%), phenoxyethanol (50%), and aloe (33%). “Anti-inflammatory” moisturizers had the greatest average number of allergens (4.00), followed by “anti-itch” (3.71) and “oatmeal” (3.71). Only products claiming to be “hypoallergenic” had significantly lower allergenic ingredient count (an average of 2.45) than those without the claim (P = .011).
“It was validating to see that eczema moisturizer products marketed as ‘hypoallergenic’ truly do have fewer allergenic ingredients than moisturizers without the claim,” Ms. Ludwig said. “However, it was surprising to see that even products marketed to eczema patients, who have a higher prevalence of allergic contact dermatitis, contain an average of 3.6 common allergens. As dermatology providers, we can relay to patients and parents that relying solely on ‘for eczema’ claims is not advisable. Clinicians should acquaint themselves with the top allergens (cetyl alcohol, phenoxyethanol, and aloe) and keep these ingredients, as well as affordability and patient preferences, in mind when making product recommendations.”
The study’s senior author, Vivian Y. Shi, MD, is a stock shareholder of Learn Health and has served as an advisory board member and/or investigator, and/or received research funding from AbbVie, Burt’s Bees, GpSkin, LEO Pharma, Eli Lilly, Menlo Therapeutics, Novartis, Pfizer, Regeneron, Sanofi Genzyme, Skin Actives Scientific, and SUN Pharma, and the Foundation for Atopic Dermatitis, Global Parents for Eczema Research, and the National Eczema Association. The other study authors reported having no financial disclosures.
FROM SPD 2020
Subcutaneous nemolizumab eases itching for atopic dermatitis
of 215 patients in Japan.
Controlling the pruritus associated with atopic dermatitis (AD) can have a significant impact on patients’ quality of life, wrote Kenji Kabashima, MD, PhD, of the department of dermatology at Kyoto University, and coauthors. Frequent scratching can cause not only mechanical skin damage, but also may enhance inflammatory reactions and contribute to sleep problems.
In earlier phase studies, nemolizumab, a humanized monoclonal antibody against interleukin-31 receptor A, showed efficacy in reducing pruritus in patients with AD, but has not been well studied in patients who are also using topical agents, they wrote.
In the study published in the New England Journal of Medicine, the researchers randomized 143 patients with AD and moderate to severe pruritus to 60 mg of subcutaneous nemolizumab and 72 patients to a placebo every 4 weeks for 16 weeks. All patients were aged 13 years and older with a confirmed AD diagnosis and a history of inadequate response to or inability to use treatments, including topical glucocorticoids and oral antihistamines. Their average age was 40 years, approximately two-thirds were male, and the average disease duration was approximately 30 years. Topical treatments included a medium potency glucocorticoid in 97% of patients in both groups, and a topical calcineurin inhibitor in 41% of those on nemolizumab, and 40% of those on placebo; almost 90% of the patients in both groups were on oral antihistamines.
At 16 weeks, scores on the visual analog scale for pruritus (the primary outcome) significantly improved from baseline in the nemolizumab group, compared with the placebo group (a mean change of –42.8% and –21.4%, respectively, P < .001).
In addition, more patients in the nemolizumab group, compared with the placebo group (40% vs. 22%) achieved a score of 4 or less on the Dermatology Life Quality Index, with lower scores reflecting less impact of disease on daily life. In addition, more patients in the nemolizumab group, compared with the placebo group (55% vs. 21%) achieved a score of 7 or less on the Insomnia Severity Index.
During the study, 71% of the patients in each group reported adverse events, most were mild or moderate. The most common adverse event was worsening AD, reported by 24% of the nemolizumab patients and 21% of the placebo patients. Reactions related to the injection occurred in 8% of nemolizumab patients and 3% of placebo patients. Cytokine abnormalities, which included an increased level of thymus and activation regulated chemokine, were reported in 10 (7%) of the patients on nemolizumab, none of which occurred in those on placebo. “Most were not accompanied by a worsening of signs of or the extent of atopic dermatitis,” the authors wrote.
Severe adverse events were reported in three patients (2%) in the nemolizumab group, which were Meniere’s disease, acute pancreatitis, and AD in one patient each. No severe adverse events were reported in the placebo group. In addition, three patients in the nemolizumab group experienced four treatment-related adverse events that led them to discontinue treatment: AD, Meniere’s disease, alopecia, and peripheral edema.
The study findings were limited by several factors including the relatively short treatment period, inclusion only of Japanese patients, inclusion of patients aged as young as 13 years, and the inability to draw conclusions from the secondary endpoints such as quality of life and sleep issues, the researchers noted.
However, the results suggest that “nemolizumab plus topical agents may ameliorate both pruritus and signs of eczema and may lessen the severity of atopic dermatitis by disrupting the itch-scratch cycle,” they added.
“Novel therapies [for AD] are needed, as there are still patients who need better disease control despite current therapies, and AD is a heterogeneous disease that may need different treatment approaches,” Eric Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland, said in an interview.
Dr. Simpson, who was not an investigator in this study, said that he was somewhat surprised that the itch reduction was lower in the current study, compared with previous studies by the same group. Also surprising was the increase in cytokine abnormalities in the nemolizumab group, which “needs further study.”
Overall, the data “provide support that blockade of the IL-31 receptor improves itch in AD and appears to have some effect on inflammation,” Dr. Simpson said.
One challenge to the clinical use of nemolizumab will be identifying “where this type of drug fits into the treatment paradigm,” and determining whether specific patients whose disease is driven more by this neuroimmune pathway could benefit more than with the traditional IL-4 or IL-13 blockade, he said.
The study was supported by Maruho. Dr. Kabashima disclosed consulting fees from Maruho and two coauthors were Maruho employees. Dr. Simpson had no financial conflicts relevant to this study, but he reported receiving research grants and other financial relationships with manufacturers of AD therapies.
SOURCE: Kabashima K et al. N Engl J Med. 2020 Jul 9. doi: 10.1056/NEJMoa1917006.
of 215 patients in Japan.
Controlling the pruritus associated with atopic dermatitis (AD) can have a significant impact on patients’ quality of life, wrote Kenji Kabashima, MD, PhD, of the department of dermatology at Kyoto University, and coauthors. Frequent scratching can cause not only mechanical skin damage, but also may enhance inflammatory reactions and contribute to sleep problems.
In earlier phase studies, nemolizumab, a humanized monoclonal antibody against interleukin-31 receptor A, showed efficacy in reducing pruritus in patients with AD, but has not been well studied in patients who are also using topical agents, they wrote.
In the study published in the New England Journal of Medicine, the researchers randomized 143 patients with AD and moderate to severe pruritus to 60 mg of subcutaneous nemolizumab and 72 patients to a placebo every 4 weeks for 16 weeks. All patients were aged 13 years and older with a confirmed AD diagnosis and a history of inadequate response to or inability to use treatments, including topical glucocorticoids and oral antihistamines. Their average age was 40 years, approximately two-thirds were male, and the average disease duration was approximately 30 years. Topical treatments included a medium potency glucocorticoid in 97% of patients in both groups, and a topical calcineurin inhibitor in 41% of those on nemolizumab, and 40% of those on placebo; almost 90% of the patients in both groups were on oral antihistamines.
At 16 weeks, scores on the visual analog scale for pruritus (the primary outcome) significantly improved from baseline in the nemolizumab group, compared with the placebo group (a mean change of –42.8% and –21.4%, respectively, P < .001).
In addition, more patients in the nemolizumab group, compared with the placebo group (40% vs. 22%) achieved a score of 4 or less on the Dermatology Life Quality Index, with lower scores reflecting less impact of disease on daily life. In addition, more patients in the nemolizumab group, compared with the placebo group (55% vs. 21%) achieved a score of 7 or less on the Insomnia Severity Index.
During the study, 71% of the patients in each group reported adverse events, most were mild or moderate. The most common adverse event was worsening AD, reported by 24% of the nemolizumab patients and 21% of the placebo patients. Reactions related to the injection occurred in 8% of nemolizumab patients and 3% of placebo patients. Cytokine abnormalities, which included an increased level of thymus and activation regulated chemokine, were reported in 10 (7%) of the patients on nemolizumab, none of which occurred in those on placebo. “Most were not accompanied by a worsening of signs of or the extent of atopic dermatitis,” the authors wrote.
Severe adverse events were reported in three patients (2%) in the nemolizumab group, which were Meniere’s disease, acute pancreatitis, and AD in one patient each. No severe adverse events were reported in the placebo group. In addition, three patients in the nemolizumab group experienced four treatment-related adverse events that led them to discontinue treatment: AD, Meniere’s disease, alopecia, and peripheral edema.
The study findings were limited by several factors including the relatively short treatment period, inclusion only of Japanese patients, inclusion of patients aged as young as 13 years, and the inability to draw conclusions from the secondary endpoints such as quality of life and sleep issues, the researchers noted.
However, the results suggest that “nemolizumab plus topical agents may ameliorate both pruritus and signs of eczema and may lessen the severity of atopic dermatitis by disrupting the itch-scratch cycle,” they added.
“Novel therapies [for AD] are needed, as there are still patients who need better disease control despite current therapies, and AD is a heterogeneous disease that may need different treatment approaches,” Eric Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland, said in an interview.
Dr. Simpson, who was not an investigator in this study, said that he was somewhat surprised that the itch reduction was lower in the current study, compared with previous studies by the same group. Also surprising was the increase in cytokine abnormalities in the nemolizumab group, which “needs further study.”
Overall, the data “provide support that blockade of the IL-31 receptor improves itch in AD and appears to have some effect on inflammation,” Dr. Simpson said.
One challenge to the clinical use of nemolizumab will be identifying “where this type of drug fits into the treatment paradigm,” and determining whether specific patients whose disease is driven more by this neuroimmune pathway could benefit more than with the traditional IL-4 or IL-13 blockade, he said.
The study was supported by Maruho. Dr. Kabashima disclosed consulting fees from Maruho and two coauthors were Maruho employees. Dr. Simpson had no financial conflicts relevant to this study, but he reported receiving research grants and other financial relationships with manufacturers of AD therapies.
SOURCE: Kabashima K et al. N Engl J Med. 2020 Jul 9. doi: 10.1056/NEJMoa1917006.
of 215 patients in Japan.
Controlling the pruritus associated with atopic dermatitis (AD) can have a significant impact on patients’ quality of life, wrote Kenji Kabashima, MD, PhD, of the department of dermatology at Kyoto University, and coauthors. Frequent scratching can cause not only mechanical skin damage, but also may enhance inflammatory reactions and contribute to sleep problems.
In earlier phase studies, nemolizumab, a humanized monoclonal antibody against interleukin-31 receptor A, showed efficacy in reducing pruritus in patients with AD, but has not been well studied in patients who are also using topical agents, they wrote.
In the study published in the New England Journal of Medicine, the researchers randomized 143 patients with AD and moderate to severe pruritus to 60 mg of subcutaneous nemolizumab and 72 patients to a placebo every 4 weeks for 16 weeks. All patients were aged 13 years and older with a confirmed AD diagnosis and a history of inadequate response to or inability to use treatments, including topical glucocorticoids and oral antihistamines. Their average age was 40 years, approximately two-thirds were male, and the average disease duration was approximately 30 years. Topical treatments included a medium potency glucocorticoid in 97% of patients in both groups, and a topical calcineurin inhibitor in 41% of those on nemolizumab, and 40% of those on placebo; almost 90% of the patients in both groups were on oral antihistamines.
At 16 weeks, scores on the visual analog scale for pruritus (the primary outcome) significantly improved from baseline in the nemolizumab group, compared with the placebo group (a mean change of –42.8% and –21.4%, respectively, P < .001).
In addition, more patients in the nemolizumab group, compared with the placebo group (40% vs. 22%) achieved a score of 4 or less on the Dermatology Life Quality Index, with lower scores reflecting less impact of disease on daily life. In addition, more patients in the nemolizumab group, compared with the placebo group (55% vs. 21%) achieved a score of 7 or less on the Insomnia Severity Index.
During the study, 71% of the patients in each group reported adverse events, most were mild or moderate. The most common adverse event was worsening AD, reported by 24% of the nemolizumab patients and 21% of the placebo patients. Reactions related to the injection occurred in 8% of nemolizumab patients and 3% of placebo patients. Cytokine abnormalities, which included an increased level of thymus and activation regulated chemokine, were reported in 10 (7%) of the patients on nemolizumab, none of which occurred in those on placebo. “Most were not accompanied by a worsening of signs of or the extent of atopic dermatitis,” the authors wrote.
Severe adverse events were reported in three patients (2%) in the nemolizumab group, which were Meniere’s disease, acute pancreatitis, and AD in one patient each. No severe adverse events were reported in the placebo group. In addition, three patients in the nemolizumab group experienced four treatment-related adverse events that led them to discontinue treatment: AD, Meniere’s disease, alopecia, and peripheral edema.
The study findings were limited by several factors including the relatively short treatment period, inclusion only of Japanese patients, inclusion of patients aged as young as 13 years, and the inability to draw conclusions from the secondary endpoints such as quality of life and sleep issues, the researchers noted.
However, the results suggest that “nemolizumab plus topical agents may ameliorate both pruritus and signs of eczema and may lessen the severity of atopic dermatitis by disrupting the itch-scratch cycle,” they added.
“Novel therapies [for AD] are needed, as there are still patients who need better disease control despite current therapies, and AD is a heterogeneous disease that may need different treatment approaches,” Eric Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland, said in an interview.
Dr. Simpson, who was not an investigator in this study, said that he was somewhat surprised that the itch reduction was lower in the current study, compared with previous studies by the same group. Also surprising was the increase in cytokine abnormalities in the nemolizumab group, which “needs further study.”
Overall, the data “provide support that blockade of the IL-31 receptor improves itch in AD and appears to have some effect on inflammation,” Dr. Simpson said.
One challenge to the clinical use of nemolizumab will be identifying “where this type of drug fits into the treatment paradigm,” and determining whether specific patients whose disease is driven more by this neuroimmune pathway could benefit more than with the traditional IL-4 or IL-13 blockade, he said.
The study was supported by Maruho. Dr. Kabashima disclosed consulting fees from Maruho and two coauthors were Maruho employees. Dr. Simpson had no financial conflicts relevant to this study, but he reported receiving research grants and other financial relationships with manufacturers of AD therapies.
SOURCE: Kabashima K et al. N Engl J Med. 2020 Jul 9. doi: 10.1056/NEJMoa1917006.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Variations in Preference for Topical Vehicles Among Demographic Groups
Topical medication is a mainstay in the treatment of dermatologic conditions. Adherence to medication regimens can be challenging in patients requiring long-term topical treatment, and nonadherence is multifactorial. A major modifiable contributing factor is patient dissatisfaction with the vehicle used. Medications often have options for different topical preparations. Therefore, it is important to consider patient preference when prescribing topical treatments to maximize adherence, ensure patient satisfaction, and optimize outcomes.
We hypothesized that notable differences exist among demographic groups regarding preference for topical vehicles. Little research has been conducted to delineate trends. This study aimed to identify variations in preference for creams, lotions, and ointments by age, gender, and ethnicity.
Methods
Data were collected through surveys distributed to all patients seen at the Truman Medical Center University Health Dermatology Clinic in Kansas City, Missouri, between September 2018 and June 2019. The study was approved by the University of Missouri Kansas City institutional review board. An estimated response rate of 95% was achieved. Each patient was informed that the survey was voluntary and anonymous, and declining to complete the survey had no effect on the care provided. Each patient completed only 1 survey and returned it to a collection box before departing from clinic.
In the survey, patients provided demographic information, including age, gender, and ethnicity. Age groups included patients younger than 40 years, 40 to 60 years, and older than 60 years. Gender groups included male and female. Ethnicity included white, black, Hispanic/Latino, and Asian/Pacific Islander or other. Patients then chose 1 of 3 options for topical vehicle preference: cream, lotion, or ointment. Each of these options was accompanied by a brief description of the vehicle, a photograph, and examples of common commercial products to aid in decision-making. The expected values were calculated based on a probability distribution under the assumption that variables have no association. Therefore, the discrepancy between the expected value and the observed value was used to describe the significance of the association between variables.
Data were analyzed using χ2 tests with the aid of a statistician. P<.05 was considered statistically significant.
Results
A total of 404 surveys were collected and recorded. Data showed statistically significant trends in each demographic parameter.
Age
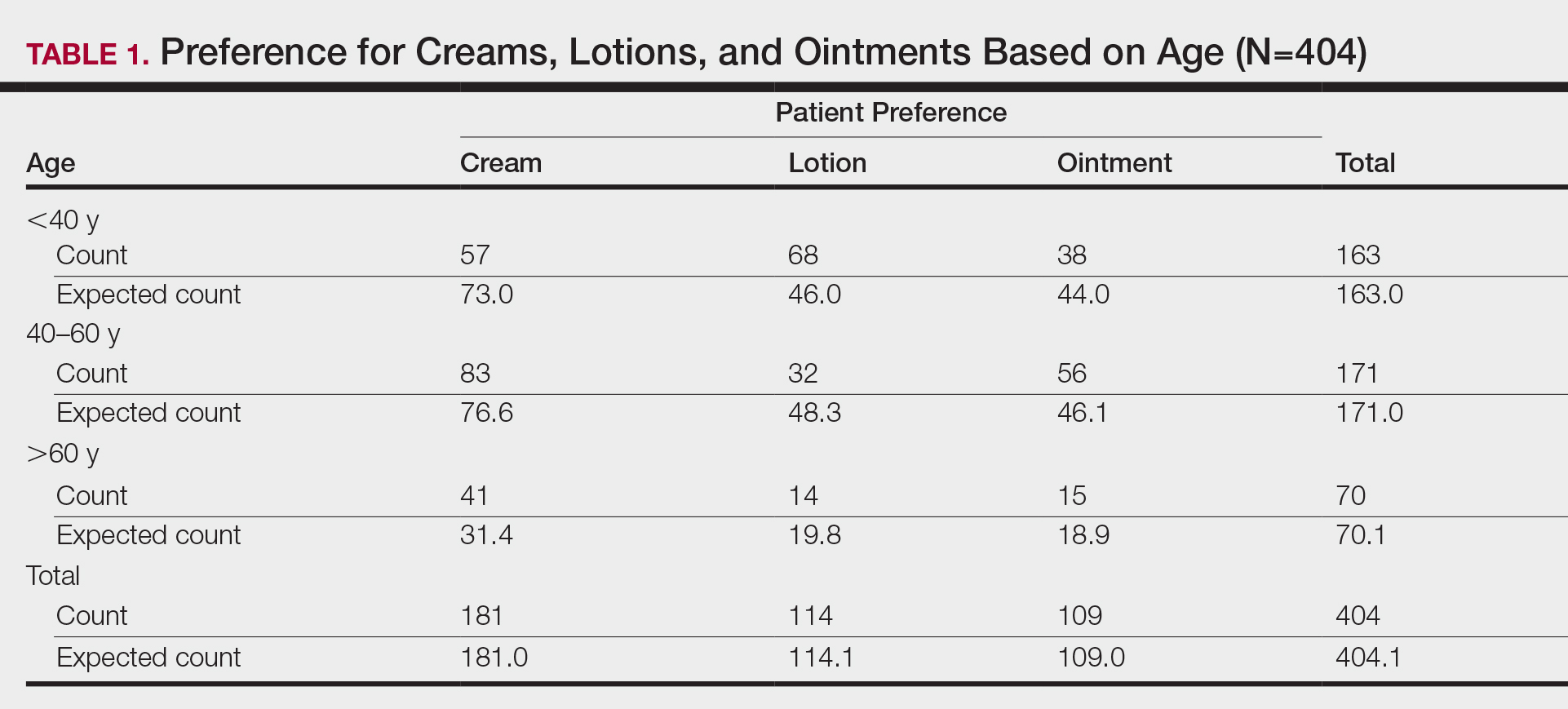
First, we analyzed differences in preference based on age (Table 1). Of 404 patients, 163 were younger than 40 years, 171 were aged 40 to 60 years, and 70 were older than 60 years. Patients younger than 40 years preferred lotion (68 vs 46.0 expected). Patients aged 40 to 60 years showed preference for cream (83 vs 76.6 expected) and ointment (56 vs 46.1 expected). Patients older than 60 years preferred cream (41 vs 31.4 expected). These findings were statistically significant (P<.0001).
Gender
Next, we evaluated variations based on gender (Table 2). Of 404 patients, 254 were female and 150 were male. Females preferred cream (127 vs 113.8 expected). Males exhibited preference for lotion (50 vs 42.3 expected) and ointment (46 vs 40.5 expected). Differences between genders were statistically significant (P=.023).

Ethnicity
We then analyzed preferences based on ethnicity (Table 3). Of 404 patients, 30 were Hispanic/Latino, 26 were Asian/Pacific Islander or other, 227 were white, and 121 were black. Hispanic/Latino patients showed equivocal findings, aligning with expected counts. Asian/Pacific Islander or other patients exhibited slight preferences for cream (14 vs 11.6 expected) and lotion (10 vs 7.3 expected). White patients preferred cream (119 vs 101.7 expected) and lotion (82 vs 64.1 expected). Black patients showed strong preference for ointment (72 vs 32.6 expected). Differences in preferences based on ethnicity were statistically significant (P<.0001).

Comment
Topical medication is a mainstay of dermatologic therapy. Many topical preparations (or vehicles) exist, including ointments, creams, lotions, gels, solutions, and foams. Vehicle type not only influences bioavailability of the prepared medication but also has a notable impact on adherence and subsequent efficacy of the topical therapy.
Medication adherence is especially challenging in dermatology, as topical medications play a central role in treatment. Compliance with the medication regimen is paramount in treatment efficacy.1 In dermatology, adherence with oral medications is higher than it is for topical medications2; various factors contribute to this difference. Compliance may decline with topical treatment due to time-consuming application, misunderstanding about the disease or the treatment regimen, frequency of administration, dissatisfaction with efficacy or appearance, and other variables.3
Other factors have been found to be important to topical medication adherence; younger age, female gender, marriage, employment, nonsmoking, nondrinking, and higher cognitive ability were associated with higher topical medication adherence.4 Our study focused on one factor: identification of demographic-specific preferences that might have implications on adherence within the studied demographic groups.
It is known that individual preferences exist when patients are choosing a topical preparation. However, a PubMed search of articles indexed for MEDLINE using the terms topical, vehicle, preparation, adherence, and preference revealed few studies that examined the preference for topical vehicle by age, gender, or ethnicity.
Existing studies have examined preferences for topical preparations based on specific disease states; this literature, albeit limited, demonstrates that preferences for topical product formulations vary among acne, atopic dermatitis, and plaque psoriasis patients.5 Other studies focus on specific patient populations or medications. For example, one study found that preference for corticosteroid vehicles among psoriasis patients was highly variable and choice of vehicle was critical to adherence.6 Another study highlighted differences in vehicle choice between younger and older age groups with psoriasis.7
Given the limited data overall, it was our goal to determine if any patterns of preference existed by age, gender, or ethnicity, regardless of disease state or indication for topical product. Importantly, over-the-counter products—cosmetic or otherwise—were not differentiated from prescribed topical medications. Our survey elucidated significant differences in preference by age, gender, and ethnicity.
Notable Findings
Regarding age, patients younger than 40 years preferred lotion, patients aged 40 to 60 years preferred cream, and patients older than 60 years preferred cream. Analysis based on gender showed that females preferred cream, and males preferred lotion and ointment. Analysis based on ethnicity most notably demonstrated a strong preference for ointment in black patients while showing preference for cream in white patients.
Potential Biases and Pitfalls
Limitations of this study included the small Hispanic/Latino and Asian/Pacific Islander populations surveyed, possible misunderstanding of the survey by respondents, and the potential for surveys being filled out twice by the same patient. Future surveys could be conducted over a longer period to increase the total sample size and to better characterize less-represented populations, such as Hispanic and Asian patients. To avoid repeat participation, the first question of the survey asked patients to indicate if they had previously completed the survey and instructed patients who had to return the repeat survey to the front desk.
To limit other errors, our survey included concise accessible descriptions of each preparation along with clear representative photographs and examples of common brands. Still, it is possible that some mistakes could have been made while patients filled out the survey based on comprehension deficits, oversight, or other reasons. It also is possible that preference might vary individually depending on the indication of the topical product—cosmetic or therapeutic—or even by anatomic site of application. Neither of these considerations was assessed specifically in our survey.
Conclusion
Our hope is that this study helps practitioners better anticipate topical preferences among patients with the ultimate goal of increasing medication adherence and patient outcomes. Nevertheless, although these general trends can provide helpful guidance, we acknowledge that individual preferences vary, and care should always be patient centered.
Acknowledgment
We thank An-Lin Cheng, PhD (Kansas City, Missouri), for assistance with the statistical analysis.
- Kircik LH. Vehicles always matter. J Drugs Dermatol. 2019;18:s99.
- Furue M, Onozuka D, Takeuchi S, et al. Poor adherence to oral andtopical medication in 3096 dermatological patients as assessed by the Morisky Medication Adherence Scale-8. Br J Dermatol. 2015;172:272-275.
- Tan X, Feldman SR, Chang, J, et al. Topical drug delivery systems in dermatology: a review of patient adherence issues. Expert Opin Drug Deliv. 2012;9:1263-1271.
- Ahn CS, Culp L, Huang WW, et al. Adherence in dermatology. J Dermatolog Treat. 2017;28:94-103.
- Eastman WJ, Malahias S, Delconte J, et al. Assessing attributes of topical vehicles for the treatment of acne, atopic dermatitis, and plaque psoriasis. Cutis. 2014;94:46-53.
- Felix K, Unrue E, Inyang M, et al. Patients preferences for different corticosteroid vehicles are highly variable. J Dermatolog Treat. 2019;31:147-151.
- Hong C-H, Papp KA, Lophaven KW, et al. Patients with psoriasis have different preferences for topical therapy, highlighting the importance of individualized treatment approaches: randomized phase IIIb PSO-INSIGHTFUL study. J Eur Acad Dermatol Venereol. 2017;31:1876-1883.
Topical medication is a mainstay in the treatment of dermatologic conditions. Adherence to medication regimens can be challenging in patients requiring long-term topical treatment, and nonadherence is multifactorial. A major modifiable contributing factor is patient dissatisfaction with the vehicle used. Medications often have options for different topical preparations. Therefore, it is important to consider patient preference when prescribing topical treatments to maximize adherence, ensure patient satisfaction, and optimize outcomes.
We hypothesized that notable differences exist among demographic groups regarding preference for topical vehicles. Little research has been conducted to delineate trends. This study aimed to identify variations in preference for creams, lotions, and ointments by age, gender, and ethnicity.
Methods
Data were collected through surveys distributed to all patients seen at the Truman Medical Center University Health Dermatology Clinic in Kansas City, Missouri, between September 2018 and June 2019. The study was approved by the University of Missouri Kansas City institutional review board. An estimated response rate of 95% was achieved. Each patient was informed that the survey was voluntary and anonymous, and declining to complete the survey had no effect on the care provided. Each patient completed only 1 survey and returned it to a collection box before departing from clinic.
In the survey, patients provided demographic information, including age, gender, and ethnicity. Age groups included patients younger than 40 years, 40 to 60 years, and older than 60 years. Gender groups included male and female. Ethnicity included white, black, Hispanic/Latino, and Asian/Pacific Islander or other. Patients then chose 1 of 3 options for topical vehicle preference: cream, lotion, or ointment. Each of these options was accompanied by a brief description of the vehicle, a photograph, and examples of common commercial products to aid in decision-making. The expected values were calculated based on a probability distribution under the assumption that variables have no association. Therefore, the discrepancy between the expected value and the observed value was used to describe the significance of the association between variables.
Data were analyzed using χ2 tests with the aid of a statistician. P<.05 was considered statistically significant.
Results
A total of 404 surveys were collected and recorded. Data showed statistically significant trends in each demographic parameter.
Age
First, we analyzed differences in preference based on age (Table 1). Of 404 patients, 163 were younger than 40 years, 171 were aged 40 to 60 years, and 70 were older than 60 years. Patients younger than 40 years preferred lotion (68 vs 46.0 expected). Patients aged 40 to 60 years showed preference for cream (83 vs 76.6 expected) and ointment (56 vs 46.1 expected). Patients older than 60 years preferred cream (41 vs 31.4 expected). These findings were statistically significant (P<.0001).

Gender
Next, we evaluated variations based on gender (Table 2). Of 404 patients, 254 were female and 150 were male. Females preferred cream (127 vs 113.8 expected). Males exhibited preference for lotion (50 vs 42.3 expected) and ointment (46 vs 40.5 expected). Differences between genders were statistically significant (P=.023).

Ethnicity
We then analyzed preferences based on ethnicity (Table 3). Of 404 patients, 30 were Hispanic/Latino, 26 were Asian/Pacific Islander or other, 227 were white, and 121 were black. Hispanic/Latino patients showed equivocal findings, aligning with expected counts. Asian/Pacific Islander or other patients exhibited slight preferences for cream (14 vs 11.6 expected) and lotion (10 vs 7.3 expected). White patients preferred cream (119 vs 101.7 expected) and lotion (82 vs 64.1 expected). Black patients showed strong preference for ointment (72 vs 32.6 expected). Differences in preferences based on ethnicity were statistically significant (P<.0001).

Comment
Topical medication is a mainstay of dermatologic therapy. Many topical preparations (or vehicles) exist, including ointments, creams, lotions, gels, solutions, and foams. Vehicle type not only influences bioavailability of the prepared medication but also has a notable impact on adherence and subsequent efficacy of the topical therapy.
Medication adherence is especially challenging in dermatology, as topical medications play a central role in treatment. Compliance with the medication regimen is paramount in treatment efficacy.1 In dermatology, adherence with oral medications is higher than it is for topical medications2; various factors contribute to this difference. Compliance may decline with topical treatment due to time-consuming application, misunderstanding about the disease or the treatment regimen, frequency of administration, dissatisfaction with efficacy or appearance, and other variables.3
Other factors have been found to be important to topical medication adherence; younger age, female gender, marriage, employment, nonsmoking, nondrinking, and higher cognitive ability were associated with higher topical medication adherence.4 Our study focused on one factor: identification of demographic-specific preferences that might have implications on adherence within the studied demographic groups.
It is known that individual preferences exist when patients are choosing a topical preparation. However, a PubMed search of articles indexed for MEDLINE using the terms topical, vehicle, preparation, adherence, and preference revealed few studies that examined the preference for topical vehicle by age, gender, or ethnicity.
Existing studies have examined preferences for topical preparations based on specific disease states; this literature, albeit limited, demonstrates that preferences for topical product formulations vary among acne, atopic dermatitis, and plaque psoriasis patients.5 Other studies focus on specific patient populations or medications. For example, one study found that preference for corticosteroid vehicles among psoriasis patients was highly variable and choice of vehicle was critical to adherence.6 Another study highlighted differences in vehicle choice between younger and older age groups with psoriasis.7
Given the limited data overall, it was our goal to determine if any patterns of preference existed by age, gender, or ethnicity, regardless of disease state or indication for topical product. Importantly, over-the-counter products—cosmetic or otherwise—were not differentiated from prescribed topical medications. Our survey elucidated significant differences in preference by age, gender, and ethnicity.
Notable Findings
Regarding age, patients younger than 40 years preferred lotion, patients aged 40 to 60 years preferred cream, and patients older than 60 years preferred cream. Analysis based on gender showed that females preferred cream, and males preferred lotion and ointment. Analysis based on ethnicity most notably demonstrated a strong preference for ointment in black patients while showing preference for cream in white patients.
Potential Biases and Pitfalls
Limitations of this study included the small Hispanic/Latino and Asian/Pacific Islander populations surveyed, possible misunderstanding of the survey by respondents, and the potential for surveys being filled out twice by the same patient. Future surveys could be conducted over a longer period to increase the total sample size and to better characterize less-represented populations, such as Hispanic and Asian patients. To avoid repeat participation, the first question of the survey asked patients to indicate if they had previously completed the survey and instructed patients who had to return the repeat survey to the front desk.
To limit other errors, our survey included concise accessible descriptions of each preparation along with clear representative photographs and examples of common brands. Still, it is possible that some mistakes could have been made while patients filled out the survey based on comprehension deficits, oversight, or other reasons. It also is possible that preference might vary individually depending on the indication of the topical product—cosmetic or therapeutic—or even by anatomic site of application. Neither of these considerations was assessed specifically in our survey.
Conclusion
Our hope is that this study helps practitioners better anticipate topical preferences among patients with the ultimate goal of increasing medication adherence and patient outcomes. Nevertheless, although these general trends can provide helpful guidance, we acknowledge that individual preferences vary, and care should always be patient centered.
Acknowledgment
We thank An-Lin Cheng, PhD (Kansas City, Missouri), for assistance with the statistical analysis.
Topical medication is a mainstay in the treatment of dermatologic conditions. Adherence to medication regimens can be challenging in patients requiring long-term topical treatment, and nonadherence is multifactorial. A major modifiable contributing factor is patient dissatisfaction with the vehicle used. Medications often have options for different topical preparations. Therefore, it is important to consider patient preference when prescribing topical treatments to maximize adherence, ensure patient satisfaction, and optimize outcomes.
We hypothesized that notable differences exist among demographic groups regarding preference for topical vehicles. Little research has been conducted to delineate trends. This study aimed to identify variations in preference for creams, lotions, and ointments by age, gender, and ethnicity.
Methods
Data were collected through surveys distributed to all patients seen at the Truman Medical Center University Health Dermatology Clinic in Kansas City, Missouri, between September 2018 and June 2019. The study was approved by the University of Missouri Kansas City institutional review board. An estimated response rate of 95% was achieved. Each patient was informed that the survey was voluntary and anonymous, and declining to complete the survey had no effect on the care provided. Each patient completed only 1 survey and returned it to a collection box before departing from clinic.
In the survey, patients provided demographic information, including age, gender, and ethnicity. Age groups included patients younger than 40 years, 40 to 60 years, and older than 60 years. Gender groups included male and female. Ethnicity included white, black, Hispanic/Latino, and Asian/Pacific Islander or other. Patients then chose 1 of 3 options for topical vehicle preference: cream, lotion, or ointment. Each of these options was accompanied by a brief description of the vehicle, a photograph, and examples of common commercial products to aid in decision-making. The expected values were calculated based on a probability distribution under the assumption that variables have no association. Therefore, the discrepancy between the expected value and the observed value was used to describe the significance of the association between variables.
Data were analyzed using χ2 tests with the aid of a statistician. P<.05 was considered statistically significant.
Results
A total of 404 surveys were collected and recorded. Data showed statistically significant trends in each demographic parameter.
Age
First, we analyzed differences in preference based on age (Table 1). Of 404 patients, 163 were younger than 40 years, 171 were aged 40 to 60 years, and 70 were older than 60 years. Patients younger than 40 years preferred lotion (68 vs 46.0 expected). Patients aged 40 to 60 years showed preference for cream (83 vs 76.6 expected) and ointment (56 vs 46.1 expected). Patients older than 60 years preferred cream (41 vs 31.4 expected). These findings were statistically significant (P<.0001).

Gender
Next, we evaluated variations based on gender (Table 2). Of 404 patients, 254 were female and 150 were male. Females preferred cream (127 vs 113.8 expected). Males exhibited preference for lotion (50 vs 42.3 expected) and ointment (46 vs 40.5 expected). Differences between genders were statistically significant (P=.023).

Ethnicity
We then analyzed preferences based on ethnicity (Table 3). Of 404 patients, 30 were Hispanic/Latino, 26 were Asian/Pacific Islander or other, 227 were white, and 121 were black. Hispanic/Latino patients showed equivocal findings, aligning with expected counts. Asian/Pacific Islander or other patients exhibited slight preferences for cream (14 vs 11.6 expected) and lotion (10 vs 7.3 expected). White patients preferred cream (119 vs 101.7 expected) and lotion (82 vs 64.1 expected). Black patients showed strong preference for ointment (72 vs 32.6 expected). Differences in preferences based on ethnicity were statistically significant (P<.0001).

Comment
Topical medication is a mainstay of dermatologic therapy. Many topical preparations (or vehicles) exist, including ointments, creams, lotions, gels, solutions, and foams. Vehicle type not only influences bioavailability of the prepared medication but also has a notable impact on adherence and subsequent efficacy of the topical therapy.
Medication adherence is especially challenging in dermatology, as topical medications play a central role in treatment. Compliance with the medication regimen is paramount in treatment efficacy.1 In dermatology, adherence with oral medications is higher than it is for topical medications2; various factors contribute to this difference. Compliance may decline with topical treatment due to time-consuming application, misunderstanding about the disease or the treatment regimen, frequency of administration, dissatisfaction with efficacy or appearance, and other variables.3
Other factors have been found to be important to topical medication adherence; younger age, female gender, marriage, employment, nonsmoking, nondrinking, and higher cognitive ability were associated with higher topical medication adherence.4 Our study focused on one factor: identification of demographic-specific preferences that might have implications on adherence within the studied demographic groups.
It is known that individual preferences exist when patients are choosing a topical preparation. However, a PubMed search of articles indexed for MEDLINE using the terms topical, vehicle, preparation, adherence, and preference revealed few studies that examined the preference for topical vehicle by age, gender, or ethnicity.
Existing studies have examined preferences for topical preparations based on specific disease states; this literature, albeit limited, demonstrates that preferences for topical product formulations vary among acne, atopic dermatitis, and plaque psoriasis patients.5 Other studies focus on specific patient populations or medications. For example, one study found that preference for corticosteroid vehicles among psoriasis patients was highly variable and choice of vehicle was critical to adherence.6 Another study highlighted differences in vehicle choice between younger and older age groups with psoriasis.7
Given the limited data overall, it was our goal to determine if any patterns of preference existed by age, gender, or ethnicity, regardless of disease state or indication for topical product. Importantly, over-the-counter products—cosmetic or otherwise—were not differentiated from prescribed topical medications. Our survey elucidated significant differences in preference by age, gender, and ethnicity.
Notable Findings
Regarding age, patients younger than 40 years preferred lotion, patients aged 40 to 60 years preferred cream, and patients older than 60 years preferred cream. Analysis based on gender showed that females preferred cream, and males preferred lotion and ointment. Analysis based on ethnicity most notably demonstrated a strong preference for ointment in black patients while showing preference for cream in white patients.
Potential Biases and Pitfalls
Limitations of this study included the small Hispanic/Latino and Asian/Pacific Islander populations surveyed, possible misunderstanding of the survey by respondents, and the potential for surveys being filled out twice by the same patient. Future surveys could be conducted over a longer period to increase the total sample size and to better characterize less-represented populations, such as Hispanic and Asian patients. To avoid repeat participation, the first question of the survey asked patients to indicate if they had previously completed the survey and instructed patients who had to return the repeat survey to the front desk.
To limit other errors, our survey included concise accessible descriptions of each preparation along with clear representative photographs and examples of common brands. Still, it is possible that some mistakes could have been made while patients filled out the survey based on comprehension deficits, oversight, or other reasons. It also is possible that preference might vary individually depending on the indication of the topical product—cosmetic or therapeutic—or even by anatomic site of application. Neither of these considerations was assessed specifically in our survey.
Conclusion
Our hope is that this study helps practitioners better anticipate topical preferences among patients with the ultimate goal of increasing medication adherence and patient outcomes. Nevertheless, although these general trends can provide helpful guidance, we acknowledge that individual preferences vary, and care should always be patient centered.
Acknowledgment
We thank An-Lin Cheng, PhD (Kansas City, Missouri), for assistance with the statistical analysis.
- Kircik LH. Vehicles always matter. J Drugs Dermatol. 2019;18:s99.
- Furue M, Onozuka D, Takeuchi S, et al. Poor adherence to oral andtopical medication in 3096 dermatological patients as assessed by the Morisky Medication Adherence Scale-8. Br J Dermatol. 2015;172:272-275.
- Tan X, Feldman SR, Chang, J, et al. Topical drug delivery systems in dermatology: a review of patient adherence issues. Expert Opin Drug Deliv. 2012;9:1263-1271.
- Ahn CS, Culp L, Huang WW, et al. Adherence in dermatology. J Dermatolog Treat. 2017;28:94-103.
- Eastman WJ, Malahias S, Delconte J, et al. Assessing attributes of topical vehicles for the treatment of acne, atopic dermatitis, and plaque psoriasis. Cutis. 2014;94:46-53.
- Felix K, Unrue E, Inyang M, et al. Patients preferences for different corticosteroid vehicles are highly variable. J Dermatolog Treat. 2019;31:147-151.
- Hong C-H, Papp KA, Lophaven KW, et al. Patients with psoriasis have different preferences for topical therapy, highlighting the importance of individualized treatment approaches: randomized phase IIIb PSO-INSIGHTFUL study. J Eur Acad Dermatol Venereol. 2017;31:1876-1883.
- Kircik LH. Vehicles always matter. J Drugs Dermatol. 2019;18:s99.
- Furue M, Onozuka D, Takeuchi S, et al. Poor adherence to oral andtopical medication in 3096 dermatological patients as assessed by the Morisky Medication Adherence Scale-8. Br J Dermatol. 2015;172:272-275.
- Tan X, Feldman SR, Chang, J, et al. Topical drug delivery systems in dermatology: a review of patient adherence issues. Expert Opin Drug Deliv. 2012;9:1263-1271.
- Ahn CS, Culp L, Huang WW, et al. Adherence in dermatology. J Dermatolog Treat. 2017;28:94-103.
- Eastman WJ, Malahias S, Delconte J, et al. Assessing attributes of topical vehicles for the treatment of acne, atopic dermatitis, and plaque psoriasis. Cutis. 2014;94:46-53.
- Felix K, Unrue E, Inyang M, et al. Patients preferences for different corticosteroid vehicles are highly variable. J Dermatolog Treat. 2019;31:147-151.
- Hong C-H, Papp KA, Lophaven KW, et al. Patients with psoriasis have different preferences for topical therapy, highlighting the importance of individualized treatment approaches: randomized phase IIIb PSO-INSIGHTFUL study. J Eur Acad Dermatol Venereol. 2017;31:1876-1883.
Practice Points
- Variations exist in preference for topical vehicles by age group, gender, and ethnicity.
- Identifying and utilizing preferred treatment options can help maximize patient outcomes.
Eczema may increase lymphoma risk, cohort studies suggest
according to two matched longitudinal cohort studies from England and Denmark.
“In this study, no evidence was found that people with atopic eczema are at increased risk of most cancers. An exception is the observed association between atopic eczema and lymphoma, particularly NHL, [which] increased with eczema severity,” Kathryn E. Mansfield, PhD, wrote in JAMA Dermatology. Adjusted hazard ratios for NHL in the English cohort were 1.06 (99% CI, 0.90-1.25) for mild atopic eczema, 1.24 (99% CI, 1.04-1.48) for moderate eczema, and 2.08 (99% CI, 1.42-3.04) for severe eczema, reported Dr. Mansfield of the London School of Hygiene and Tropical Medicine and associates.
Past studies of a possible link between atopic eczema and cancer have produced conflicting evidence, which might reflect “two competing theories” – that cancer risk falls with greater immune surveillance, and that cancer risk rises with immune stimulation, the researchers wrote. Immunosuppressive treatment and an impaired skin barrier might also increase the risk of cancer, but the evidence is conflicting.
For the study, they analyzed electronic health records linked with hospital admissions and death records in England and national health registry data from Denmark. The English cohort included 471,970 adults with atopic eczema and 2,239,775 adults without atopic eczema. The Danish cohort was composed of individuals of any age, including 44,945 who had eczema and with 445,673 who did not. Participants were matched based on factors such as age, sex, and primary care practice. The researchers excluded individuals with a history of cancer, apart from nonmelanoma skin cancer or keratinocyte cancer. (For analyses of skin cancer risk, they also excluded individuals with a history of nonmelanoma skin cancer.)
Overall, there was “little evidence” for a link between atopic eczema and cancer (adjusted hazard ratio in England, 1.04; 99% CI, 1.02-1.06; aHR in Denmark, 1.05; 99% CI, 0.95-1.16) or for most specific types of cancer, the investigators wrote.
In England, however, eczema was associated with a significantly increased risk for noncutaneous lymphoma, with an adjusted HRs of 1.19 (99% CI, 1.07-1.34) for NHL, and 1.48 (99% CI, 1.07-2.04) for Hodgkin lymphoma. Lymphoma risk was highest among adults with severe eczema, defined as those who had been prescribed a systemic treatment for their disease, who had received phototherapy, or who had been referred to a specialist or admitted to a hospital for atopic eczema. Point estimates in the Danish cohort also revealed a higher risk for lymphoma among individuals with moderate to severe atopic eczema, compared with those with eczema, but the 99% CIs crossed 1.0.
The findings highlight the need to be aware of, screen for, and study the pathogenesis of heightened lymphoma risk among patients with atopic eczema, said Shawn Demehri, MD, PhD, of the department of dermatology and cancer center, at Massachusetts General Hospital, Boston, who was not involved in the study.
“Prospectively collected data from large cohorts of eczema patients is a strength of this study,” he said in an interview. “However, the age range included in the study is suboptimal for assessing cancer as an outcome. The lower incidence of cancer in younger individuals hinders the ability to detect differences in cancer risk between the two groups.” (Approximately 57% of individuals in the English cohort were aged 18-44 years, while approximately 70% of those in the Danish cohort were less than 18 years.)
Understanding how eczema affects the risk of non-Hodgkin lymphoma is an important future direction of research, Dr. Demehri emphasized. “The landscape of atopic eczema therapeutics has dramatically changed in the recent years. It will be very interesting to determine how new biologics impact cancer risk in eczema patients.”
Partial support for the work was provided by the Wellcome Trust, the Royal Society, the Dagmar Marshalls Fund, and the Aase and Ejnar Danielsens Fund. Dr. Mansfield disclosed support from a Wellcome Trust grant. Her coinvestigators disclosed ties to TARGET-DERM, Pfizer, and GlaxoSmithKline, and from the Wellcome Trust, Medical Research Council, the National Institute for Health Research, the British Heart Foundation, Diabetes UK, and IMI Horizon 2020 funding BIOMAP. Dr. Demehri reported having no relevant conflicts of interest.
SOURCE: Mansfield KE et al. JAMA Dermatol. 2020 Jun 24. doi: 10.1001/jamadermatol.2020.1948.
according to two matched longitudinal cohort studies from England and Denmark.
“In this study, no evidence was found that people with atopic eczema are at increased risk of most cancers. An exception is the observed association between atopic eczema and lymphoma, particularly NHL, [which] increased with eczema severity,” Kathryn E. Mansfield, PhD, wrote in JAMA Dermatology. Adjusted hazard ratios for NHL in the English cohort were 1.06 (99% CI, 0.90-1.25) for mild atopic eczema, 1.24 (99% CI, 1.04-1.48) for moderate eczema, and 2.08 (99% CI, 1.42-3.04) for severe eczema, reported Dr. Mansfield of the London School of Hygiene and Tropical Medicine and associates.
Past studies of a possible link between atopic eczema and cancer have produced conflicting evidence, which might reflect “two competing theories” – that cancer risk falls with greater immune surveillance, and that cancer risk rises with immune stimulation, the researchers wrote. Immunosuppressive treatment and an impaired skin barrier might also increase the risk of cancer, but the evidence is conflicting.
For the study, they analyzed electronic health records linked with hospital admissions and death records in England and national health registry data from Denmark. The English cohort included 471,970 adults with atopic eczema and 2,239,775 adults without atopic eczema. The Danish cohort was composed of individuals of any age, including 44,945 who had eczema and with 445,673 who did not. Participants were matched based on factors such as age, sex, and primary care practice. The researchers excluded individuals with a history of cancer, apart from nonmelanoma skin cancer or keratinocyte cancer. (For analyses of skin cancer risk, they also excluded individuals with a history of nonmelanoma skin cancer.)
Overall, there was “little evidence” for a link between atopic eczema and cancer (adjusted hazard ratio in England, 1.04; 99% CI, 1.02-1.06; aHR in Denmark, 1.05; 99% CI, 0.95-1.16) or for most specific types of cancer, the investigators wrote.
In England, however, eczema was associated with a significantly increased risk for noncutaneous lymphoma, with an adjusted HRs of 1.19 (99% CI, 1.07-1.34) for NHL, and 1.48 (99% CI, 1.07-2.04) for Hodgkin lymphoma. Lymphoma risk was highest among adults with severe eczema, defined as those who had been prescribed a systemic treatment for their disease, who had received phototherapy, or who had been referred to a specialist or admitted to a hospital for atopic eczema. Point estimates in the Danish cohort also revealed a higher risk for lymphoma among individuals with moderate to severe atopic eczema, compared with those with eczema, but the 99% CIs crossed 1.0.
The findings highlight the need to be aware of, screen for, and study the pathogenesis of heightened lymphoma risk among patients with atopic eczema, said Shawn Demehri, MD, PhD, of the department of dermatology and cancer center, at Massachusetts General Hospital, Boston, who was not involved in the study.
“Prospectively collected data from large cohorts of eczema patients is a strength of this study,” he said in an interview. “However, the age range included in the study is suboptimal for assessing cancer as an outcome. The lower incidence of cancer in younger individuals hinders the ability to detect differences in cancer risk between the two groups.” (Approximately 57% of individuals in the English cohort were aged 18-44 years, while approximately 70% of those in the Danish cohort were less than 18 years.)
Understanding how eczema affects the risk of non-Hodgkin lymphoma is an important future direction of research, Dr. Demehri emphasized. “The landscape of atopic eczema therapeutics has dramatically changed in the recent years. It will be very interesting to determine how new biologics impact cancer risk in eczema patients.”
Partial support for the work was provided by the Wellcome Trust, the Royal Society, the Dagmar Marshalls Fund, and the Aase and Ejnar Danielsens Fund. Dr. Mansfield disclosed support from a Wellcome Trust grant. Her coinvestigators disclosed ties to TARGET-DERM, Pfizer, and GlaxoSmithKline, and from the Wellcome Trust, Medical Research Council, the National Institute for Health Research, the British Heart Foundation, Diabetes UK, and IMI Horizon 2020 funding BIOMAP. Dr. Demehri reported having no relevant conflicts of interest.
SOURCE: Mansfield KE et al. JAMA Dermatol. 2020 Jun 24. doi: 10.1001/jamadermatol.2020.1948.
according to two matched longitudinal cohort studies from England and Denmark.
“In this study, no evidence was found that people with atopic eczema are at increased risk of most cancers. An exception is the observed association between atopic eczema and lymphoma, particularly NHL, [which] increased with eczema severity,” Kathryn E. Mansfield, PhD, wrote in JAMA Dermatology. Adjusted hazard ratios for NHL in the English cohort were 1.06 (99% CI, 0.90-1.25) for mild atopic eczema, 1.24 (99% CI, 1.04-1.48) for moderate eczema, and 2.08 (99% CI, 1.42-3.04) for severe eczema, reported Dr. Mansfield of the London School of Hygiene and Tropical Medicine and associates.
Past studies of a possible link between atopic eczema and cancer have produced conflicting evidence, which might reflect “two competing theories” – that cancer risk falls with greater immune surveillance, and that cancer risk rises with immune stimulation, the researchers wrote. Immunosuppressive treatment and an impaired skin barrier might also increase the risk of cancer, but the evidence is conflicting.
For the study, they analyzed electronic health records linked with hospital admissions and death records in England and national health registry data from Denmark. The English cohort included 471,970 adults with atopic eczema and 2,239,775 adults without atopic eczema. The Danish cohort was composed of individuals of any age, including 44,945 who had eczema and with 445,673 who did not. Participants were matched based on factors such as age, sex, and primary care practice. The researchers excluded individuals with a history of cancer, apart from nonmelanoma skin cancer or keratinocyte cancer. (For analyses of skin cancer risk, they also excluded individuals with a history of nonmelanoma skin cancer.)
Overall, there was “little evidence” for a link between atopic eczema and cancer (adjusted hazard ratio in England, 1.04; 99% CI, 1.02-1.06; aHR in Denmark, 1.05; 99% CI, 0.95-1.16) or for most specific types of cancer, the investigators wrote.
In England, however, eczema was associated with a significantly increased risk for noncutaneous lymphoma, with an adjusted HRs of 1.19 (99% CI, 1.07-1.34) for NHL, and 1.48 (99% CI, 1.07-2.04) for Hodgkin lymphoma. Lymphoma risk was highest among adults with severe eczema, defined as those who had been prescribed a systemic treatment for their disease, who had received phototherapy, or who had been referred to a specialist or admitted to a hospital for atopic eczema. Point estimates in the Danish cohort also revealed a higher risk for lymphoma among individuals with moderate to severe atopic eczema, compared with those with eczema, but the 99% CIs crossed 1.0.
The findings highlight the need to be aware of, screen for, and study the pathogenesis of heightened lymphoma risk among patients with atopic eczema, said Shawn Demehri, MD, PhD, of the department of dermatology and cancer center, at Massachusetts General Hospital, Boston, who was not involved in the study.
“Prospectively collected data from large cohorts of eczema patients is a strength of this study,” he said in an interview. “However, the age range included in the study is suboptimal for assessing cancer as an outcome. The lower incidence of cancer in younger individuals hinders the ability to detect differences in cancer risk between the two groups.” (Approximately 57% of individuals in the English cohort were aged 18-44 years, while approximately 70% of those in the Danish cohort were less than 18 years.)
Understanding how eczema affects the risk of non-Hodgkin lymphoma is an important future direction of research, Dr. Demehri emphasized. “The landscape of atopic eczema therapeutics has dramatically changed in the recent years. It will be very interesting to determine how new biologics impact cancer risk in eczema patients.”
Partial support for the work was provided by the Wellcome Trust, the Royal Society, the Dagmar Marshalls Fund, and the Aase and Ejnar Danielsens Fund. Dr. Mansfield disclosed support from a Wellcome Trust grant. Her coinvestigators disclosed ties to TARGET-DERM, Pfizer, and GlaxoSmithKline, and from the Wellcome Trust, Medical Research Council, the National Institute for Health Research, the British Heart Foundation, Diabetes UK, and IMI Horizon 2020 funding BIOMAP. Dr. Demehri reported having no relevant conflicts of interest.
SOURCE: Mansfield KE et al. JAMA Dermatol. 2020 Jun 24. doi: 10.1001/jamadermatol.2020.1948.
FROM JAMA DERMATOLOGY
More phase 3 data reported for abrocitinib for atopic dermatitis
Melinda Gooderham, MD, reported at the virtual annual meeting of the American Academy of Dermatology.
The positive results of this 391-patient, international, randomized, double-blind, placebo-controlled clinical trial mirror those previously reported in the identically designed JADE-MONO-1 pivotal phase 3 trial, noted Dr. Gooderham, medical director of the SKiN Centre for Dermatology and a dermatologist at Queen’s University in Kingston, Ont.
Participants in JADE-MONO-2 were randomized 2:2:1 to abrocitinib at 200 mg once daily, 100 mg once daily, or placebo for 12 weeks. The coprimary endpoint of skin clearance as reflected in an Investigator’s Global Assessment (IGA) score of 0 or 1 (clear or almost clear) with an improvement of at least two grades at week 12 was achieved in 38.1% and 28.4% of patients on 200 and 100 mg of the JAK-1 inhibitor, respectively, compared with 9.1% of placebo-treated controls. The other coprimary endpoint – significant improvement in disease extent as defined by at least a 75% reduction from baseline in Eczema Area and Severity Index (EASI-75 response) at 12 weeks – was reached in 61% of patients on abrocitinib at 200 mg/day, 44.5% on 100 mg/day, and 10.4% of controls.
A key secondary endpoint was improvement in itch based on at least a 4-point improvement at week 12 on the Peak Pruritus Numerical Rating Scale from a mean baseline score of 7. This outcome was reached by 55.3% of patients on abrocitinib at 200 mg, 45.2% on 100 mg, and 11.5% on placebo. Of note, the reduction in itch was impressively fast, with significant separation from placebo occurring within the first 24 hours of the study, after just a single dose of abrocitinib. By week 2, roughly one-third of patients on high-dose and one-quarter of those on low-dose abrocitinib had already reached the itch endpoint, the dermatologist continued.
The improvement in pruritus scores in abrocitinib-treated patients was accompanied by significantly greater gains on validated measures of quality of life, another secondary endpoint. The EASI-90 response rate, yet another key secondary outcome, was 37.7% with abrocitinib at 200 mg, 23.9% with 100 mg, and 3.9% with placebo.
The safety profile of abrocitinib was essentially the same as for placebo with the exception of a 3.2% incidence of thrombocytopenia in patients on abrocitinib at 200 mg/day; no cases occurred in controls or patients on abrocitinib at 100 mg/day. Although venous thromboembolism has arisen as a potential concern in clinical trials of oral JAK inhibitors for rheumatoid arthritis, there were no cases in JADE-MONO-2. A long-term safety extension study in JADE-MONO participants is underway.
In an interview, Dr. Gooderham said that, based on the phase 2 study data that’s available for upadacitinib, another JAK-1-selective oral agent, abrocitinib and upadacitinib appear to be in the same ballpark with respect to efficacy as defined by IGA response, EASI improvement, and itch relief.
“The JAK-1 selectivity does seem to offer some advantage in levels of response over more broad JAK inhibition, such as with baritinib,” she added.
Asked how she foresees abrocitinib fitting into clinical practice, should it win regulatory approval for treatment of atopic dermatitis, Dr. Gooderham said it might be considered on a par with the injectable interleukin-4 and -13 inhibitor dupilumab (Dupixent) as next-line therapy after failure on topical therapy or as an option in patients who haven’t responded to or could not tolerate dupilumab. Abrocitinib will be an attractive option for patients who prefer oral therapy and will be an especially appealing medication in patients with a strong itch component to their atopic dermatitis, she added.
The results of JADE COMPARE, a phase 3, head-to-head randomized comparison of abrocitinib and dupilumab, are expected to be presented later this year at the virtual annual congress of the European Academy of Dermatology and Venereology. Pfizer has announced the key results, reporting that the JAK inhibitor at 200 mg/day achieved significantly greater improvements than dupilumab in the coprimary IGA and EASI-75 endpoints at 12 weeks.
JADE-MONO-2 was sponsored by Pfizer. Dr. Gooderham reported receiving research grants from that company and close to two dozen others.
The JADE-MONO-2 results have been published online (JAMA Dermatol. 2020 Jun 3;e201406. doi: 10.1001/jamadermatol.2020.1406).
Melinda Gooderham, MD, reported at the virtual annual meeting of the American Academy of Dermatology.
The positive results of this 391-patient, international, randomized, double-blind, placebo-controlled clinical trial mirror those previously reported in the identically designed JADE-MONO-1 pivotal phase 3 trial, noted Dr. Gooderham, medical director of the SKiN Centre for Dermatology and a dermatologist at Queen’s University in Kingston, Ont.
Participants in JADE-MONO-2 were randomized 2:2:1 to abrocitinib at 200 mg once daily, 100 mg once daily, or placebo for 12 weeks. The coprimary endpoint of skin clearance as reflected in an Investigator’s Global Assessment (IGA) score of 0 or 1 (clear or almost clear) with an improvement of at least two grades at week 12 was achieved in 38.1% and 28.4% of patients on 200 and 100 mg of the JAK-1 inhibitor, respectively, compared with 9.1% of placebo-treated controls. The other coprimary endpoint – significant improvement in disease extent as defined by at least a 75% reduction from baseline in Eczema Area and Severity Index (EASI-75 response) at 12 weeks – was reached in 61% of patients on abrocitinib at 200 mg/day, 44.5% on 100 mg/day, and 10.4% of controls.
A key secondary endpoint was improvement in itch based on at least a 4-point improvement at week 12 on the Peak Pruritus Numerical Rating Scale from a mean baseline score of 7. This outcome was reached by 55.3% of patients on abrocitinib at 200 mg, 45.2% on 100 mg, and 11.5% on placebo. Of note, the reduction in itch was impressively fast, with significant separation from placebo occurring within the first 24 hours of the study, after just a single dose of abrocitinib. By week 2, roughly one-third of patients on high-dose and one-quarter of those on low-dose abrocitinib had already reached the itch endpoint, the dermatologist continued.
The improvement in pruritus scores in abrocitinib-treated patients was accompanied by significantly greater gains on validated measures of quality of life, another secondary endpoint. The EASI-90 response rate, yet another key secondary outcome, was 37.7% with abrocitinib at 200 mg, 23.9% with 100 mg, and 3.9% with placebo.
The safety profile of abrocitinib was essentially the same as for placebo with the exception of a 3.2% incidence of thrombocytopenia in patients on abrocitinib at 200 mg/day; no cases occurred in controls or patients on abrocitinib at 100 mg/day. Although venous thromboembolism has arisen as a potential concern in clinical trials of oral JAK inhibitors for rheumatoid arthritis, there were no cases in JADE-MONO-2. A long-term safety extension study in JADE-MONO participants is underway.
In an interview, Dr. Gooderham said that, based on the phase 2 study data that’s available for upadacitinib, another JAK-1-selective oral agent, abrocitinib and upadacitinib appear to be in the same ballpark with respect to efficacy as defined by IGA response, EASI improvement, and itch relief.
“The JAK-1 selectivity does seem to offer some advantage in levels of response over more broad JAK inhibition, such as with baritinib,” she added.
Asked how she foresees abrocitinib fitting into clinical practice, should it win regulatory approval for treatment of atopic dermatitis, Dr. Gooderham said it might be considered on a par with the injectable interleukin-4 and -13 inhibitor dupilumab (Dupixent) as next-line therapy after failure on topical therapy or as an option in patients who haven’t responded to or could not tolerate dupilumab. Abrocitinib will be an attractive option for patients who prefer oral therapy and will be an especially appealing medication in patients with a strong itch component to their atopic dermatitis, she added.
The results of JADE COMPARE, a phase 3, head-to-head randomized comparison of abrocitinib and dupilumab, are expected to be presented later this year at the virtual annual congress of the European Academy of Dermatology and Venereology. Pfizer has announced the key results, reporting that the JAK inhibitor at 200 mg/day achieved significantly greater improvements than dupilumab in the coprimary IGA and EASI-75 endpoints at 12 weeks.
JADE-MONO-2 was sponsored by Pfizer. Dr. Gooderham reported receiving research grants from that company and close to two dozen others.
The JADE-MONO-2 results have been published online (JAMA Dermatol. 2020 Jun 3;e201406. doi: 10.1001/jamadermatol.2020.1406).
Melinda Gooderham, MD, reported at the virtual annual meeting of the American Academy of Dermatology.
The positive results of this 391-patient, international, randomized, double-blind, placebo-controlled clinical trial mirror those previously reported in the identically designed JADE-MONO-1 pivotal phase 3 trial, noted Dr. Gooderham, medical director of the SKiN Centre for Dermatology and a dermatologist at Queen’s University in Kingston, Ont.
Participants in JADE-MONO-2 were randomized 2:2:1 to abrocitinib at 200 mg once daily, 100 mg once daily, or placebo for 12 weeks. The coprimary endpoint of skin clearance as reflected in an Investigator’s Global Assessment (IGA) score of 0 or 1 (clear or almost clear) with an improvement of at least two grades at week 12 was achieved in 38.1% and 28.4% of patients on 200 and 100 mg of the JAK-1 inhibitor, respectively, compared with 9.1% of placebo-treated controls. The other coprimary endpoint – significant improvement in disease extent as defined by at least a 75% reduction from baseline in Eczema Area and Severity Index (EASI-75 response) at 12 weeks – was reached in 61% of patients on abrocitinib at 200 mg/day, 44.5% on 100 mg/day, and 10.4% of controls.
A key secondary endpoint was improvement in itch based on at least a 4-point improvement at week 12 on the Peak Pruritus Numerical Rating Scale from a mean baseline score of 7. This outcome was reached by 55.3% of patients on abrocitinib at 200 mg, 45.2% on 100 mg, and 11.5% on placebo. Of note, the reduction in itch was impressively fast, with significant separation from placebo occurring within the first 24 hours of the study, after just a single dose of abrocitinib. By week 2, roughly one-third of patients on high-dose and one-quarter of those on low-dose abrocitinib had already reached the itch endpoint, the dermatologist continued.
The improvement in pruritus scores in abrocitinib-treated patients was accompanied by significantly greater gains on validated measures of quality of life, another secondary endpoint. The EASI-90 response rate, yet another key secondary outcome, was 37.7% with abrocitinib at 200 mg, 23.9% with 100 mg, and 3.9% with placebo.
The safety profile of abrocitinib was essentially the same as for placebo with the exception of a 3.2% incidence of thrombocytopenia in patients on abrocitinib at 200 mg/day; no cases occurred in controls or patients on abrocitinib at 100 mg/day. Although venous thromboembolism has arisen as a potential concern in clinical trials of oral JAK inhibitors for rheumatoid arthritis, there were no cases in JADE-MONO-2. A long-term safety extension study in JADE-MONO participants is underway.
In an interview, Dr. Gooderham said that, based on the phase 2 study data that’s available for upadacitinib, another JAK-1-selective oral agent, abrocitinib and upadacitinib appear to be in the same ballpark with respect to efficacy as defined by IGA response, EASI improvement, and itch relief.
“The JAK-1 selectivity does seem to offer some advantage in levels of response over more broad JAK inhibition, such as with baritinib,” she added.
Asked how she foresees abrocitinib fitting into clinical practice, should it win regulatory approval for treatment of atopic dermatitis, Dr. Gooderham said it might be considered on a par with the injectable interleukin-4 and -13 inhibitor dupilumab (Dupixent) as next-line therapy after failure on topical therapy or as an option in patients who haven’t responded to or could not tolerate dupilumab. Abrocitinib will be an attractive option for patients who prefer oral therapy and will be an especially appealing medication in patients with a strong itch component to their atopic dermatitis, she added.
The results of JADE COMPARE, a phase 3, head-to-head randomized comparison of abrocitinib and dupilumab, are expected to be presented later this year at the virtual annual congress of the European Academy of Dermatology and Venereology. Pfizer has announced the key results, reporting that the JAK inhibitor at 200 mg/day achieved significantly greater improvements than dupilumab in the coprimary IGA and EASI-75 endpoints at 12 weeks.
JADE-MONO-2 was sponsored by Pfizer. Dr. Gooderham reported receiving research grants from that company and close to two dozen others.
The JADE-MONO-2 results have been published online (JAMA Dermatol. 2020 Jun 3;e201406. doi: 10.1001/jamadermatol.2020.1406).
FROM AAD 20
Tralokinumab found effective in phase 3 atopic dermatitis studies
presented at the virtual annual meeting of the American Academy of Dermatology.
Tralokinumab is a fully human monoclonal antibody which binds specifically to interleukin-13 and thereby prevents downstream IL-13 signaling. In contrast, dupilumab (Dupixent), at present the only approved biologic agent for AD, blocks both the IL-13 and IL-4 pathways.
Two of the pivotal phase 3 trials presented at AAD 2020 – ECZTRA 1 and ECZTRA 2 – were identically designed, randomized, double-blind, placebo-controlled, 52-week, multinational monotherapy studies including a collective 1,596 adults with moderate to severe AD. In contrast, ECZTRA 3 was a 380-patient, double-blind, randomized, 32-week study of tralokinumab in combination with a topical corticosteroid versus placebo injections plus a topical corticosteroid.
“I would say the take-home point of these trials is they are proof of principle that blocking just IL-13 can be an effective approach. The studies help us understand that IL-13 is an important driver cytokine for the disease,” Eric Simpson, MD, lead clinical investigator for ECZTRA 2, said in an interview.
In all three phase 3 trials, the primary endpoint was achievement of a clinical response as defined by an Investigator Global Assessment score of clear or almost clear skin (IGA 0/1) plus at least a 75% improvement in the Eczema Area and Severity Index score (EASI-75) at week 16. In ECZTRA 1 and 2, this was accomplished in 16% and 22% of patients on 300 mg of tralokinumab administered subcutaneously every 2 weeks, compared with 7% and 11% of placebo-treated controls.
Patients with a clinical response at week 16 were then rerandomized to tralokinumab either every other week or every 4 weeks or to placebo for an additional 36 weeks. At 52 weeks, 51% and 59% of patients in ECZTRA 1 and 2, respectively, who had a clinical response at week 16 maintained an IGA 0/1 response while on tralokinumab every 2 weeks, as did 39% and 45% of those switched to treatment every 4 weeks. Similarly, 60% and 56% of clinical responders at week 16 maintained an EASI-75 response at week 52 with tralokinumab every 2 weeks, as did 49% and 51% of those rerandomized to treatment every 4 weeks.
The safety profile of tralokinumab in the two monotherapy trials was comparable with placebo.
In the ECZTRA studies, tralokinumab achieved significant improvement at week 16 in secondary endpoints including itch, health-related quality of life, and severity and extent of skin lesions.
How does tralokinumab, with its narrower focus targeting a single cytokine, stack up against dupilumab, the dual IL-13/IL-4 inhibitor that’s transformed the treatment of patients with moderate or severe AD?
Dr. Simpson, who was also principal investigator in a pivotal phase 3 trial for dupilumab, emphasized that no firm conclusions can be drawn because there have been no head-to-head comparative trials and the tralokinumab and dupilumab trials had different patient populations, geographic locations, and washout periods. With those caveats, however, he commented that, “just on the surface, numerically, for the monotherapy studies, dupilumab hit some higher targets than tralokinumab in terms of the percentage of patients clear or almost clear.”
In terms of safety, it appears that the risk of conjunctivitis may be lower with tralokinumab than dupilumab, with rates of 7% and 3% through 52 weeks in ECZTRA 1 and 2, respectively, versus 2% with placebo, although again this is “a caveated conclusion,” said Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland.
Tralokinumab combination therapy in ECZTRA 3
At 16 weeks, 39% of patients treated with tralokinumab plus topical corticosteroids had an IGA of 0/1 and 56% had an EASI-75 response, compared with 26% and 36% of patients on topical corticosteroids plus biweekly placebo injections. More than 90% of patients with a good clinical response at week 16 maintained that response at week 32 while on tralokinumab biweekly plus topical steroids. Among good responders at week 16 who were rerandomized to 300 mg of tralokinumab every 4 weeks plus topical steroids, 78% still had an IGA of 0/1 at week 32, and 91% had an EASI-75, reported Jonathan I. Silverberg, MD, PhD, director of clinical research and contact dermatitis at George Washington University, Washington.
A randomized, placebo-controlled combination therapy study such as this provides information that’s especially useful in clinical practice, Dr. Simpson observed.
“When I’m talking to patients about any biologics or oral therapies, I usually quote the figures from the combination therapy studies because the vast majority of our patients are using topical therapy in addition to systemics,” he said in the interview.
Asked how he envisions tralokinumab’s role in clinical practice, should the drug receive regulatory approval, Dr. Simpson said that he welcomes the prospect of having an additional treatment option to discuss with patients. Tralokinumab could be considered either as first-line therapy in patients who are failing on topical therapy or for patients who don’t respond adequately to or experience limiting side effects on dupilumab.
“There isn’t any established, published treatment algorithm in atopic dermatitis, probably for good reason, since we don’t have data to tell us you should start here and then move there. Those are long, difficult studies to perform,” Dr. Simpson said.
LEO Pharma has announced that it has applied for marketing approval for tralokinumab to the European Medicines Agency and plans to do so with the Food and Drug Administration by year’s end.
Dr. Simpson reported receiving research grants from and serving as a consultant to LEO Pharma, sponsor of the ECZTRA trials. He has similar financial relationships with close to a dozen other pharmaceutical companies.
presented at the virtual annual meeting of the American Academy of Dermatology.
Tralokinumab is a fully human monoclonal antibody which binds specifically to interleukin-13 and thereby prevents downstream IL-13 signaling. In contrast, dupilumab (Dupixent), at present the only approved biologic agent for AD, blocks both the IL-13 and IL-4 pathways.
Two of the pivotal phase 3 trials presented at AAD 2020 – ECZTRA 1 and ECZTRA 2 – were identically designed, randomized, double-blind, placebo-controlled, 52-week, multinational monotherapy studies including a collective 1,596 adults with moderate to severe AD. In contrast, ECZTRA 3 was a 380-patient, double-blind, randomized, 32-week study of tralokinumab in combination with a topical corticosteroid versus placebo injections plus a topical corticosteroid.
“I would say the take-home point of these trials is they are proof of principle that blocking just IL-13 can be an effective approach. The studies help us understand that IL-13 is an important driver cytokine for the disease,” Eric Simpson, MD, lead clinical investigator for ECZTRA 2, said in an interview.
In all three phase 3 trials, the primary endpoint was achievement of a clinical response as defined by an Investigator Global Assessment score of clear or almost clear skin (IGA 0/1) plus at least a 75% improvement in the Eczema Area and Severity Index score (EASI-75) at week 16. In ECZTRA 1 and 2, this was accomplished in 16% and 22% of patients on 300 mg of tralokinumab administered subcutaneously every 2 weeks, compared with 7% and 11% of placebo-treated controls.
Patients with a clinical response at week 16 were then rerandomized to tralokinumab either every other week or every 4 weeks or to placebo for an additional 36 weeks. At 52 weeks, 51% and 59% of patients in ECZTRA 1 and 2, respectively, who had a clinical response at week 16 maintained an IGA 0/1 response while on tralokinumab every 2 weeks, as did 39% and 45% of those switched to treatment every 4 weeks. Similarly, 60% and 56% of clinical responders at week 16 maintained an EASI-75 response at week 52 with tralokinumab every 2 weeks, as did 49% and 51% of those rerandomized to treatment every 4 weeks.
The safety profile of tralokinumab in the two monotherapy trials was comparable with placebo.
In the ECZTRA studies, tralokinumab achieved significant improvement at week 16 in secondary endpoints including itch, health-related quality of life, and severity and extent of skin lesions.
How does tralokinumab, with its narrower focus targeting a single cytokine, stack up against dupilumab, the dual IL-13/IL-4 inhibitor that’s transformed the treatment of patients with moderate or severe AD?
Dr. Simpson, who was also principal investigator in a pivotal phase 3 trial for dupilumab, emphasized that no firm conclusions can be drawn because there have been no head-to-head comparative trials and the tralokinumab and dupilumab trials had different patient populations, geographic locations, and washout periods. With those caveats, however, he commented that, “just on the surface, numerically, for the monotherapy studies, dupilumab hit some higher targets than tralokinumab in terms of the percentage of patients clear or almost clear.”
In terms of safety, it appears that the risk of conjunctivitis may be lower with tralokinumab than dupilumab, with rates of 7% and 3% through 52 weeks in ECZTRA 1 and 2, respectively, versus 2% with placebo, although again this is “a caveated conclusion,” said Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland.
Tralokinumab combination therapy in ECZTRA 3
At 16 weeks, 39% of patients treated with tralokinumab plus topical corticosteroids had an IGA of 0/1 and 56% had an EASI-75 response, compared with 26% and 36% of patients on topical corticosteroids plus biweekly placebo injections. More than 90% of patients with a good clinical response at week 16 maintained that response at week 32 while on tralokinumab biweekly plus topical steroids. Among good responders at week 16 who were rerandomized to 300 mg of tralokinumab every 4 weeks plus topical steroids, 78% still had an IGA of 0/1 at week 32, and 91% had an EASI-75, reported Jonathan I. Silverberg, MD, PhD, director of clinical research and contact dermatitis at George Washington University, Washington.
A randomized, placebo-controlled combination therapy study such as this provides information that’s especially useful in clinical practice, Dr. Simpson observed.
“When I’m talking to patients about any biologics or oral therapies, I usually quote the figures from the combination therapy studies because the vast majority of our patients are using topical therapy in addition to systemics,” he said in the interview.
Asked how he envisions tralokinumab’s role in clinical practice, should the drug receive regulatory approval, Dr. Simpson said that he welcomes the prospect of having an additional treatment option to discuss with patients. Tralokinumab could be considered either as first-line therapy in patients who are failing on topical therapy or for patients who don’t respond adequately to or experience limiting side effects on dupilumab.
“There isn’t any established, published treatment algorithm in atopic dermatitis, probably for good reason, since we don’t have data to tell us you should start here and then move there. Those are long, difficult studies to perform,” Dr. Simpson said.
LEO Pharma has announced that it has applied for marketing approval for tralokinumab to the European Medicines Agency and plans to do so with the Food and Drug Administration by year’s end.
Dr. Simpson reported receiving research grants from and serving as a consultant to LEO Pharma, sponsor of the ECZTRA trials. He has similar financial relationships with close to a dozen other pharmaceutical companies.
presented at the virtual annual meeting of the American Academy of Dermatology.
Tralokinumab is a fully human monoclonal antibody which binds specifically to interleukin-13 and thereby prevents downstream IL-13 signaling. In contrast, dupilumab (Dupixent), at present the only approved biologic agent for AD, blocks both the IL-13 and IL-4 pathways.
Two of the pivotal phase 3 trials presented at AAD 2020 – ECZTRA 1 and ECZTRA 2 – were identically designed, randomized, double-blind, placebo-controlled, 52-week, multinational monotherapy studies including a collective 1,596 adults with moderate to severe AD. In contrast, ECZTRA 3 was a 380-patient, double-blind, randomized, 32-week study of tralokinumab in combination with a topical corticosteroid versus placebo injections plus a topical corticosteroid.
“I would say the take-home point of these trials is they are proof of principle that blocking just IL-13 can be an effective approach. The studies help us understand that IL-13 is an important driver cytokine for the disease,” Eric Simpson, MD, lead clinical investigator for ECZTRA 2, said in an interview.
In all three phase 3 trials, the primary endpoint was achievement of a clinical response as defined by an Investigator Global Assessment score of clear or almost clear skin (IGA 0/1) plus at least a 75% improvement in the Eczema Area and Severity Index score (EASI-75) at week 16. In ECZTRA 1 and 2, this was accomplished in 16% and 22% of patients on 300 mg of tralokinumab administered subcutaneously every 2 weeks, compared with 7% and 11% of placebo-treated controls.
Patients with a clinical response at week 16 were then rerandomized to tralokinumab either every other week or every 4 weeks or to placebo for an additional 36 weeks. At 52 weeks, 51% and 59% of patients in ECZTRA 1 and 2, respectively, who had a clinical response at week 16 maintained an IGA 0/1 response while on tralokinumab every 2 weeks, as did 39% and 45% of those switched to treatment every 4 weeks. Similarly, 60% and 56% of clinical responders at week 16 maintained an EASI-75 response at week 52 with tralokinumab every 2 weeks, as did 49% and 51% of those rerandomized to treatment every 4 weeks.
The safety profile of tralokinumab in the two monotherapy trials was comparable with placebo.
In the ECZTRA studies, tralokinumab achieved significant improvement at week 16 in secondary endpoints including itch, health-related quality of life, and severity and extent of skin lesions.
How does tralokinumab, with its narrower focus targeting a single cytokine, stack up against dupilumab, the dual IL-13/IL-4 inhibitor that’s transformed the treatment of patients with moderate or severe AD?
Dr. Simpson, who was also principal investigator in a pivotal phase 3 trial for dupilumab, emphasized that no firm conclusions can be drawn because there have been no head-to-head comparative trials and the tralokinumab and dupilumab trials had different patient populations, geographic locations, and washout periods. With those caveats, however, he commented that, “just on the surface, numerically, for the monotherapy studies, dupilumab hit some higher targets than tralokinumab in terms of the percentage of patients clear or almost clear.”
In terms of safety, it appears that the risk of conjunctivitis may be lower with tralokinumab than dupilumab, with rates of 7% and 3% through 52 weeks in ECZTRA 1 and 2, respectively, versus 2% with placebo, although again this is “a caveated conclusion,” said Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland.
Tralokinumab combination therapy in ECZTRA 3
At 16 weeks, 39% of patients treated with tralokinumab plus topical corticosteroids had an IGA of 0/1 and 56% had an EASI-75 response, compared with 26% and 36% of patients on topical corticosteroids plus biweekly placebo injections. More than 90% of patients with a good clinical response at week 16 maintained that response at week 32 while on tralokinumab biweekly plus topical steroids. Among good responders at week 16 who were rerandomized to 300 mg of tralokinumab every 4 weeks plus topical steroids, 78% still had an IGA of 0/1 at week 32, and 91% had an EASI-75, reported Jonathan I. Silverberg, MD, PhD, director of clinical research and contact dermatitis at George Washington University, Washington.
A randomized, placebo-controlled combination therapy study such as this provides information that’s especially useful in clinical practice, Dr. Simpson observed.
“When I’m talking to patients about any biologics or oral therapies, I usually quote the figures from the combination therapy studies because the vast majority of our patients are using topical therapy in addition to systemics,” he said in the interview.
Asked how he envisions tralokinumab’s role in clinical practice, should the drug receive regulatory approval, Dr. Simpson said that he welcomes the prospect of having an additional treatment option to discuss with patients. Tralokinumab could be considered either as first-line therapy in patients who are failing on topical therapy or for patients who don’t respond adequately to or experience limiting side effects on dupilumab.
“There isn’t any established, published treatment algorithm in atopic dermatitis, probably for good reason, since we don’t have data to tell us you should start here and then move there. Those are long, difficult studies to perform,” Dr. Simpson said.
LEO Pharma has announced that it has applied for marketing approval for tralokinumab to the European Medicines Agency and plans to do so with the Food and Drug Administration by year’s end.
Dr. Simpson reported receiving research grants from and serving as a consultant to LEO Pharma, sponsor of the ECZTRA trials. He has similar financial relationships with close to a dozen other pharmaceutical companies.
FROM AAD 2020
Pediatric Dermatology: A Supplement to Pediatric News & Dermatology News
Content includes:
- Early onset of atopic dermatitis linked to poorer control, could signify more persistent disease
- Patients with actopic dermatitis should be routinely asked about conjunctivitis
- Hope on the horizon: New cantharidin formulation alleviates molluscum contagiosum in pivotal trials
- Patch testing in atopic dermatitis: When and how
- Topical calcineurin inhibitors are an effective treatment option for periorificial dermatitis
- Psychology consults for children’s skin issues can boost adherence, wellness
Content includes:
- Early onset of atopic dermatitis linked to poorer control, could signify more persistent disease
- Patients with actopic dermatitis should be routinely asked about conjunctivitis
- Hope on the horizon: New cantharidin formulation alleviates molluscum contagiosum in pivotal trials
- Patch testing in atopic dermatitis: When and how
- Topical calcineurin inhibitors are an effective treatment option for periorificial dermatitis
- Psychology consults for children’s skin issues can boost adherence, wellness
Content includes:
- Early onset of atopic dermatitis linked to poorer control, could signify more persistent disease
- Patients with actopic dermatitis should be routinely asked about conjunctivitis
- Hope on the horizon: New cantharidin formulation alleviates molluscum contagiosum in pivotal trials
- Patch testing in atopic dermatitis: When and how
- Topical calcineurin inhibitors are an effective treatment option for periorificial dermatitis
- Psychology consults for children’s skin issues can boost adherence, wellness
Pediatric Dermatology: A Supplement to Pediatric News & Dermatology News
Content includes:
- Early onset of atopic dermatitis linked to poorer control, could signify more persistent disease
- Patients with actopic dermatitis should be routinely asked about conjunctivitis
- Hope on the horizon: New cantharidin formulation alleviates molluscum contagiosum in pivotal trials
- Patch testing in atopic dermatitis: When and how
- Topical calcineurin inhibitors are an effective treatment option for periorificial dermatitis
- Psychology consults for children’s skin issues can boost adherence, wellness
Content includes:
- Early onset of atopic dermatitis linked to poorer control, could signify more persistent disease
- Patients with actopic dermatitis should be routinely asked about conjunctivitis
- Hope on the horizon: New cantharidin formulation alleviates molluscum contagiosum in pivotal trials
- Patch testing in atopic dermatitis: When and how
- Topical calcineurin inhibitors are an effective treatment option for periorificial dermatitis
- Psychology consults for children’s skin issues can boost adherence, wellness
Content includes:
- Early onset of atopic dermatitis linked to poorer control, could signify more persistent disease
- Patients with actopic dermatitis should be routinely asked about conjunctivitis
- Hope on the horizon: New cantharidin formulation alleviates molluscum contagiosum in pivotal trials
- Patch testing in atopic dermatitis: When and how
- Topical calcineurin inhibitors are an effective treatment option for periorificial dermatitis
- Psychology consults for children’s skin issues can boost adherence, wellness
Atopic dermatitis in adults, children linked to neuropsychiatric disorders
according to a study presented at the annual meeting of the Society for Investigative Dermatology, held virtually.
“The risk increase ranges from as low as 5% up to 59%, depending on the outcome, with generally greater effects observed among the adults,” Joy Wan, MD, a postdoctoral dermatology fellow at the University of Pennsylvania, Philadelphia, said in her presentation. The risk was independent of other atopic disease, gender, age, and socioeconomic status.
Dr. Wan and colleagues conducted a cohort study of patients with AD in the United Kingdom using data from the Health Improvement Network (THIN) electronic records database, matching AD patients in THIN with up to five patients without AD, similar in age and also registered to general practices. The researchers validated AD disease status using an algorithm that identified patients with a diagnostic code and two therapy codes related to AD. Outcomes of interest included anxiety, depression, bipolar disorder, obsessive-compulsive disorder, ADHD, schizophrenia, and autism. Patients entered into the cohort when they were diagnosed with AD, registered by a practice, or when data from a practice was reported to THIN. The researchers stopped following patients when they developed a neuropsychiatric outcome of interest, left a practice, died, or when the study ended.
“Previous studies have found associations between atopic dermatitis and anxiety, depression, and attention-deficit/hyperactivity disorder. However, many previous studies had been cross-sectional and they were unable to evaluate the directionality of association between atopic dermatitis and neuropsychiatric outcomes, while other previous studies have relied on the self-report of atopic dermatitis and outcomes as well,” Dr. Wan said. “Thus, longitudinal studies, using validated measures of atopic dermatitis, and those that include the entire age span, are really needed.”
Overall, 434,859 children and adolescents under aged 18 with AD in the THIN database were matched to 1,983,589 controls, and 644,802 adults with AD were matched to almost 2,900,000 adults without AD. In the pediatric group, demographics were mostly balanced between children with and without AD: the average age ranged between about 5 and almost 6 years. In pediatric patients with AD, there was a higher rate of allergic rhinitis (6.2% vs. 4%) and asthma (13.5% vs. 9.3%) than in the control group.
For adults, the average age was about 48 years in both groups. Compared with patients who did not have AD, adults with AD also had higher rates of allergic rhinitis (15.2% vs. 9.6%) and asthma (19.9% vs. 12.6%).
After adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis, Dr. Wan and colleagues found greater rates of bipolar disorder (hazard ratio, 1.34; 95% confidence interval, 1.09-1.65), obsessive-compulsive disorder (HR, 1.30; 95% CI, 1.21-1.41), anxiety (HR, 1.09; 95% CI, 1.07-1.11), and depression (HR, 1.06; 95% CI, 1.04-1.08) among children and adolescents with AD, compared with controls.
In the adult cohort, a diagnosis of AD was associated with an increased risk of autism (HR, 1.53; 95% CI, 1.30-1.80), obsessive-compulsive disorder (HR, 1.49; 95% CI, 1.40-1.59), ADHD (HR, 1.31; 95% CI, 1.13-1.53), anxiety (HR, 1.17; 95% CI, 1.15-1.18), depression (HR, 1.15; 95% CI, 1.14-1.16), and bipolar disorder (HR, 1.12; 95% CI, 1.04-1.21), after adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis.
One reason for the increased associations among the adults, even for ADHD and autism, which are more characteristically diagnosed in childhood, Dr. Wan said, is that, since they looked at incident outcomes, “many children may already have had these prevalent comorbidities at the time of the entry in the cohort.”
She noted that the study may have observation bias or unknown confounders, but she hopes these results raise awareness of the association between AD and neuropsychiatric disorders, although more research is needed to determine how AD severity affects neuropsychiatric outcomes. “Additional work is needed to really understand the mechanisms that drive these associations, whether it’s mediated through symptoms of atopic dermatitis such as itch and poor sleep, or potentially the stigma of having a chronic skin disease, or perhaps shared pathophysiology between atopic dermatitis and these neuropsychiatric diseases,” she said.
The study was funded by a grant from Pfizer. Dr. Wan reports receiving research funding from Pfizer paid to the University of Pennsylvania.
according to a study presented at the annual meeting of the Society for Investigative Dermatology, held virtually.
“The risk increase ranges from as low as 5% up to 59%, depending on the outcome, with generally greater effects observed among the adults,” Joy Wan, MD, a postdoctoral dermatology fellow at the University of Pennsylvania, Philadelphia, said in her presentation. The risk was independent of other atopic disease, gender, age, and socioeconomic status.
Dr. Wan and colleagues conducted a cohort study of patients with AD in the United Kingdom using data from the Health Improvement Network (THIN) electronic records database, matching AD patients in THIN with up to five patients without AD, similar in age and also registered to general practices. The researchers validated AD disease status using an algorithm that identified patients with a diagnostic code and two therapy codes related to AD. Outcomes of interest included anxiety, depression, bipolar disorder, obsessive-compulsive disorder, ADHD, schizophrenia, and autism. Patients entered into the cohort when they were diagnosed with AD, registered by a practice, or when data from a practice was reported to THIN. The researchers stopped following patients when they developed a neuropsychiatric outcome of interest, left a practice, died, or when the study ended.
“Previous studies have found associations between atopic dermatitis and anxiety, depression, and attention-deficit/hyperactivity disorder. However, many previous studies had been cross-sectional and they were unable to evaluate the directionality of association between atopic dermatitis and neuropsychiatric outcomes, while other previous studies have relied on the self-report of atopic dermatitis and outcomes as well,” Dr. Wan said. “Thus, longitudinal studies, using validated measures of atopic dermatitis, and those that include the entire age span, are really needed.”
Overall, 434,859 children and adolescents under aged 18 with AD in the THIN database were matched to 1,983,589 controls, and 644,802 adults with AD were matched to almost 2,900,000 adults without AD. In the pediatric group, demographics were mostly balanced between children with and without AD: the average age ranged between about 5 and almost 6 years. In pediatric patients with AD, there was a higher rate of allergic rhinitis (6.2% vs. 4%) and asthma (13.5% vs. 9.3%) than in the control group.
For adults, the average age was about 48 years in both groups. Compared with patients who did not have AD, adults with AD also had higher rates of allergic rhinitis (15.2% vs. 9.6%) and asthma (19.9% vs. 12.6%).
After adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis, Dr. Wan and colleagues found greater rates of bipolar disorder (hazard ratio, 1.34; 95% confidence interval, 1.09-1.65), obsessive-compulsive disorder (HR, 1.30; 95% CI, 1.21-1.41), anxiety (HR, 1.09; 95% CI, 1.07-1.11), and depression (HR, 1.06; 95% CI, 1.04-1.08) among children and adolescents with AD, compared with controls.
In the adult cohort, a diagnosis of AD was associated with an increased risk of autism (HR, 1.53; 95% CI, 1.30-1.80), obsessive-compulsive disorder (HR, 1.49; 95% CI, 1.40-1.59), ADHD (HR, 1.31; 95% CI, 1.13-1.53), anxiety (HR, 1.17; 95% CI, 1.15-1.18), depression (HR, 1.15; 95% CI, 1.14-1.16), and bipolar disorder (HR, 1.12; 95% CI, 1.04-1.21), after adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis.
One reason for the increased associations among the adults, even for ADHD and autism, which are more characteristically diagnosed in childhood, Dr. Wan said, is that, since they looked at incident outcomes, “many children may already have had these prevalent comorbidities at the time of the entry in the cohort.”
She noted that the study may have observation bias or unknown confounders, but she hopes these results raise awareness of the association between AD and neuropsychiatric disorders, although more research is needed to determine how AD severity affects neuropsychiatric outcomes. “Additional work is needed to really understand the mechanisms that drive these associations, whether it’s mediated through symptoms of atopic dermatitis such as itch and poor sleep, or potentially the stigma of having a chronic skin disease, or perhaps shared pathophysiology between atopic dermatitis and these neuropsychiatric diseases,” she said.
The study was funded by a grant from Pfizer. Dr. Wan reports receiving research funding from Pfizer paid to the University of Pennsylvania.
according to a study presented at the annual meeting of the Society for Investigative Dermatology, held virtually.
“The risk increase ranges from as low as 5% up to 59%, depending on the outcome, with generally greater effects observed among the adults,” Joy Wan, MD, a postdoctoral dermatology fellow at the University of Pennsylvania, Philadelphia, said in her presentation. The risk was independent of other atopic disease, gender, age, and socioeconomic status.
Dr. Wan and colleagues conducted a cohort study of patients with AD in the United Kingdom using data from the Health Improvement Network (THIN) electronic records database, matching AD patients in THIN with up to five patients without AD, similar in age and also registered to general practices. The researchers validated AD disease status using an algorithm that identified patients with a diagnostic code and two therapy codes related to AD. Outcomes of interest included anxiety, depression, bipolar disorder, obsessive-compulsive disorder, ADHD, schizophrenia, and autism. Patients entered into the cohort when they were diagnosed with AD, registered by a practice, or when data from a practice was reported to THIN. The researchers stopped following patients when they developed a neuropsychiatric outcome of interest, left a practice, died, or when the study ended.
“Previous studies have found associations between atopic dermatitis and anxiety, depression, and attention-deficit/hyperactivity disorder. However, many previous studies had been cross-sectional and they were unable to evaluate the directionality of association between atopic dermatitis and neuropsychiatric outcomes, while other previous studies have relied on the self-report of atopic dermatitis and outcomes as well,” Dr. Wan said. “Thus, longitudinal studies, using validated measures of atopic dermatitis, and those that include the entire age span, are really needed.”
Overall, 434,859 children and adolescents under aged 18 with AD in the THIN database were matched to 1,983,589 controls, and 644,802 adults with AD were matched to almost 2,900,000 adults without AD. In the pediatric group, demographics were mostly balanced between children with and without AD: the average age ranged between about 5 and almost 6 years. In pediatric patients with AD, there was a higher rate of allergic rhinitis (6.2% vs. 4%) and asthma (13.5% vs. 9.3%) than in the control group.
For adults, the average age was about 48 years in both groups. Compared with patients who did not have AD, adults with AD also had higher rates of allergic rhinitis (15.2% vs. 9.6%) and asthma (19.9% vs. 12.6%).
After adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis, Dr. Wan and colleagues found greater rates of bipolar disorder (hazard ratio, 1.34; 95% confidence interval, 1.09-1.65), obsessive-compulsive disorder (HR, 1.30; 95% CI, 1.21-1.41), anxiety (HR, 1.09; 95% CI, 1.07-1.11), and depression (HR, 1.06; 95% CI, 1.04-1.08) among children and adolescents with AD, compared with controls.
In the adult cohort, a diagnosis of AD was associated with an increased risk of autism (HR, 1.53; 95% CI, 1.30-1.80), obsessive-compulsive disorder (HR, 1.49; 95% CI, 1.40-1.59), ADHD (HR, 1.31; 95% CI, 1.13-1.53), anxiety (HR, 1.17; 95% CI, 1.15-1.18), depression (HR, 1.15; 95% CI, 1.14-1.16), and bipolar disorder (HR, 1.12; 95% CI, 1.04-1.21), after adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis.
One reason for the increased associations among the adults, even for ADHD and autism, which are more characteristically diagnosed in childhood, Dr. Wan said, is that, since they looked at incident outcomes, “many children may already have had these prevalent comorbidities at the time of the entry in the cohort.”
She noted that the study may have observation bias or unknown confounders, but she hopes these results raise awareness of the association between AD and neuropsychiatric disorders, although more research is needed to determine how AD severity affects neuropsychiatric outcomes. “Additional work is needed to really understand the mechanisms that drive these associations, whether it’s mediated through symptoms of atopic dermatitis such as itch and poor sleep, or potentially the stigma of having a chronic skin disease, or perhaps shared pathophysiology between atopic dermatitis and these neuropsychiatric diseases,” she said.
The study was funded by a grant from Pfizer. Dr. Wan reports receiving research funding from Pfizer paid to the University of Pennsylvania.
FROM SID 2020
Gaps in Treatment Guidelines for Atopic Dermatitis
Treatment options for atopic dermatitis have evolved significantly in the past several years, but the current guidelines have yet to catch up.
Drs Steven Feldman and Lindsay Strowd, from Wake Forest School of Medicine, discuss gaps in the American Academy of Dermatology treatment guidelines for atopic dermatitis.
The current guidelines have not been updated to include medications approved for atopic dermatitis, including crisaborole, a steroid-sparing ointment used to treat mild to moderate disease in patients 3 months of age and older.
Another drug that has been approved since the 2014 guidelines is the biologic dupilumab, which is a monoclonal antibody that acts on the IL-4 receptor. The agent inhibits the binding of IL-4 receptors to the principal cytokines responsible for mediating the disease. Dupilumab is administered by injection and is approved for patients 6 years and older with moderate to severe disease.
The doctors also discuss therapies for atopic dermatitis currently in development, including topical and oral JAK inhibitors. They agree that the long-term benefit of topical JAK inhibitors may be limited, but that oral JAK inhibitors have the potential to be more effective than dupilumab and more acceptable to patients who do not like injections.
--
Steven R. Feldman, MD, PhD, Professor, Department of Dermatology, Wake Forest School of Medicine, Winston-Salem, North Carolina
Steven R. Feldman, MD, PhD, has disclosed the following relevant financial relationships:
Serve(d) as a consultant for: AbbVie; Alvotech; Advance Medical; Almirall; Arena; Bristol-Myers Squibb; Caremark; Amgen; Celgene; Galderma Laboratories; Gerson Lehrman Group; Guidepoint Global; Helsinn; Janssen; Kikaku; Leo Pharma; Eli Lilly and Company; Merck; Mylan; Novartis; Ortho Dermatology; Pfizer; Regeneron; Sanofi; Sienna; Sun Pharma; Suncare Research; Xenoport
Serve(d) as a speaker for: AbbVie; Amgen; Celgene; Janssen; Leo Pharma; Eli Lilly and Company; Novartis; Ortho Dermatology; Pfizer; Regeneron; Sanofi; Sun Pharma
Receive(d) grant support from: AbbVie; Amgen; Celgene; Galderma Laboratories; Janssen; Eli Lilly and Company; Novartis; Pfizer; Regeneron; Sanofi
Receive(d) royalties from: Informa; UpToDate; Xlibris
Holds stock in: Causa Technologies; Medical Quality Enhancement Corporation
Serves as founder and chief technology officer for: Causa Technologies
Lindsay C. Strowd, MD, Associate Professor, Vice Chair, Department of Dermatology, Wake Forest School of Medicine, Winston-Salem, North Carolina
Lindsay C. Strowd, MD, has disclosed no relevant financial relationships.
Treatment options for atopic dermatitis have evolved significantly in the past several years, but the current guidelines have yet to catch up.
Drs Steven Feldman and Lindsay Strowd, from Wake Forest School of Medicine, discuss gaps in the American Academy of Dermatology treatment guidelines for atopic dermatitis.
The current guidelines have not been updated to include medications approved for atopic dermatitis, including crisaborole, a steroid-sparing ointment used to treat mild to moderate disease in patients 3 months of age and older.
Another drug that has been approved since the 2014 guidelines is the biologic dupilumab, which is a monoclonal antibody that acts on the IL-4 receptor. The agent inhibits the binding of IL-4 receptors to the principal cytokines responsible for mediating the disease. Dupilumab is administered by injection and is approved for patients 6 years and older with moderate to severe disease.
The doctors also discuss therapies for atopic dermatitis currently in development, including topical and oral JAK inhibitors. They agree that the long-term benefit of topical JAK inhibitors may be limited, but that oral JAK inhibitors have the potential to be more effective than dupilumab and more acceptable to patients who do not like injections.
--
Steven R. Feldman, MD, PhD, Professor, Department of Dermatology, Wake Forest School of Medicine, Winston-Salem, North Carolina
Steven R. Feldman, MD, PhD, has disclosed the following relevant financial relationships:
Serve(d) as a consultant for: AbbVie; Alvotech; Advance Medical; Almirall; Arena; Bristol-Myers Squibb; Caremark; Amgen; Celgene; Galderma Laboratories; Gerson Lehrman Group; Guidepoint Global; Helsinn; Janssen; Kikaku; Leo Pharma; Eli Lilly and Company; Merck; Mylan; Novartis; Ortho Dermatology; Pfizer; Regeneron; Sanofi; Sienna; Sun Pharma; Suncare Research; Xenoport
Serve(d) as a speaker for: AbbVie; Amgen; Celgene; Janssen; Leo Pharma; Eli Lilly and Company; Novartis; Ortho Dermatology; Pfizer; Regeneron; Sanofi; Sun Pharma
Receive(d) grant support from: AbbVie; Amgen; Celgene; Galderma Laboratories; Janssen; Eli Lilly and Company; Novartis; Pfizer; Regeneron; Sanofi
Receive(d) royalties from: Informa; UpToDate; Xlibris
Holds stock in: Causa Technologies; Medical Quality Enhancement Corporation
Serves as founder and chief technology officer for: Causa Technologies
Lindsay C. Strowd, MD, Associate Professor, Vice Chair, Department of Dermatology, Wake Forest School of Medicine, Winston-Salem, North Carolina
Lindsay C. Strowd, MD, has disclosed no relevant financial relationships.
Treatment options for atopic dermatitis have evolved significantly in the past several years, but the current guidelines have yet to catch up.
Drs Steven Feldman and Lindsay Strowd, from Wake Forest School of Medicine, discuss gaps in the American Academy of Dermatology treatment guidelines for atopic dermatitis.
The current guidelines have not been updated to include medications approved for atopic dermatitis, including crisaborole, a steroid-sparing ointment used to treat mild to moderate disease in patients 3 months of age and older.
Another drug that has been approved since the 2014 guidelines is the biologic dupilumab, which is a monoclonal antibody that acts on the IL-4 receptor. The agent inhibits the binding of IL-4 receptors to the principal cytokines responsible for mediating the disease. Dupilumab is administered by injection and is approved for patients 6 years and older with moderate to severe disease.
The doctors also discuss therapies for atopic dermatitis currently in development, including topical and oral JAK inhibitors. They agree that the long-term benefit of topical JAK inhibitors may be limited, but that oral JAK inhibitors have the potential to be more effective than dupilumab and more acceptable to patients who do not like injections.
--
Steven R. Feldman, MD, PhD, Professor, Department of Dermatology, Wake Forest School of Medicine, Winston-Salem, North Carolina
Steven R. Feldman, MD, PhD, has disclosed the following relevant financial relationships:
Serve(d) as a consultant for: AbbVie; Alvotech; Advance Medical; Almirall; Arena; Bristol-Myers Squibb; Caremark; Amgen; Celgene; Galderma Laboratories; Gerson Lehrman Group; Guidepoint Global; Helsinn; Janssen; Kikaku; Leo Pharma; Eli Lilly and Company; Merck; Mylan; Novartis; Ortho Dermatology; Pfizer; Regeneron; Sanofi; Sienna; Sun Pharma; Suncare Research; Xenoport
Serve(d) as a speaker for: AbbVie; Amgen; Celgene; Janssen; Leo Pharma; Eli Lilly and Company; Novartis; Ortho Dermatology; Pfizer; Regeneron; Sanofi; Sun Pharma
Receive(d) grant support from: AbbVie; Amgen; Celgene; Galderma Laboratories; Janssen; Eli Lilly and Company; Novartis; Pfizer; Regeneron; Sanofi
Receive(d) royalties from: Informa; UpToDate; Xlibris
Holds stock in: Causa Technologies; Medical Quality Enhancement Corporation
Serves as founder and chief technology officer for: Causa Technologies
Lindsay C. Strowd, MD, Associate Professor, Vice Chair, Department of Dermatology, Wake Forest School of Medicine, Winston-Salem, North Carolina
Lindsay C. Strowd, MD, has disclosed no relevant financial relationships.








