User login
Highlights in Atopic Dermatitis From AAAAI 2021
Dr Elaine Siegfried, an expert in pediatric dermatology in St. Louis, Missouri, examines some of the key data and research highlights in atopic dermatitis presented at the 2021 American Academy of Allergy, Asthma & Immunology (AAAAI) Virtual Annual Meeting.
Dr Siegfried starts with two studies on the selective JAK1 inhibitor abrocitinib, in which results in both adults and adolescents confirm the efficacy of the 200 mg dose. However, she cautions that the jury is still out on the long-term safety for this class of drugs.
She then reviews two studies looking at whether individuals with atopic dermatitis who are hospitalized with COVID-19 suffer more severe outcomes than the general population.
Dr Siegfried's final selections focus on food allergy, an important aspect of management in patients with atopic dermatitis.
The first two look at the outcomes of food challenges in shrimp-sensitized children and those with a history suggestive of milk allergy.
The last three studies examine the latest data on an oral immunotherapy peanut allergen powder (Palforzia) approved by the FDA and some of the nonclinical factors that could hamper its uptake.
--
Director, Division of Pediatric Dermatology, Cardinal Glennon Children's Hospital, Saint Louis University Health Sciences Center; Owner, Director, Kids Dermatology, St. Louis, Missouri
Elaine C. Siegfried, MD, has disclosed the following relevant financial relationships:
Contracted research from: AI Therapeutics.
Received consulting fees from: Boehringer Ingelheim; Incyte; Regeneron; Sanofi Genzyme; UCB; AbbVie; Verrica; Leo; Novan; Novartis; Pfizer; Pierre Fabre
Received honoraria from: Regeneron; Sanofi Genzyme; Verrica.
Received fees to SSM/SLU related to sponsoring clinical trials from: Regeneron; Verrica; Pierre Fabre; Janssen; Eli Lilly and Company.
Served on the Data Safety Monitoring Board for: UCB; Leo; Pfizer; Novan.
Received grant funding to support 2020-2022 Peds Derm Fellow from: Pfizer.
Dr Elaine Siegfried, an expert in pediatric dermatology in St. Louis, Missouri, examines some of the key data and research highlights in atopic dermatitis presented at the 2021 American Academy of Allergy, Asthma & Immunology (AAAAI) Virtual Annual Meeting.
Dr Siegfried starts with two studies on the selective JAK1 inhibitor abrocitinib, in which results in both adults and adolescents confirm the efficacy of the 200 mg dose. However, she cautions that the jury is still out on the long-term safety for this class of drugs.
She then reviews two studies looking at whether individuals with atopic dermatitis who are hospitalized with COVID-19 suffer more severe outcomes than the general population.
Dr Siegfried's final selections focus on food allergy, an important aspect of management in patients with atopic dermatitis.
The first two look at the outcomes of food challenges in shrimp-sensitized children and those with a history suggestive of milk allergy.
The last three studies examine the latest data on an oral immunotherapy peanut allergen powder (Palforzia) approved by the FDA and some of the nonclinical factors that could hamper its uptake.
--
Director, Division of Pediatric Dermatology, Cardinal Glennon Children's Hospital, Saint Louis University Health Sciences Center; Owner, Director, Kids Dermatology, St. Louis, Missouri
Elaine C. Siegfried, MD, has disclosed the following relevant financial relationships:
Contracted research from: AI Therapeutics.
Received consulting fees from: Boehringer Ingelheim; Incyte; Regeneron; Sanofi Genzyme; UCB; AbbVie; Verrica; Leo; Novan; Novartis; Pfizer; Pierre Fabre
Received honoraria from: Regeneron; Sanofi Genzyme; Verrica.
Received fees to SSM/SLU related to sponsoring clinical trials from: Regeneron; Verrica; Pierre Fabre; Janssen; Eli Lilly and Company.
Served on the Data Safety Monitoring Board for: UCB; Leo; Pfizer; Novan.
Received grant funding to support 2020-2022 Peds Derm Fellow from: Pfizer.
Dr Elaine Siegfried, an expert in pediatric dermatology in St. Louis, Missouri, examines some of the key data and research highlights in atopic dermatitis presented at the 2021 American Academy of Allergy, Asthma & Immunology (AAAAI) Virtual Annual Meeting.
Dr Siegfried starts with two studies on the selective JAK1 inhibitor abrocitinib, in which results in both adults and adolescents confirm the efficacy of the 200 mg dose. However, she cautions that the jury is still out on the long-term safety for this class of drugs.
She then reviews two studies looking at whether individuals with atopic dermatitis who are hospitalized with COVID-19 suffer more severe outcomes than the general population.
Dr Siegfried's final selections focus on food allergy, an important aspect of management in patients with atopic dermatitis.
The first two look at the outcomes of food challenges in shrimp-sensitized children and those with a history suggestive of milk allergy.
The last three studies examine the latest data on an oral immunotherapy peanut allergen powder (Palforzia) approved by the FDA and some of the nonclinical factors that could hamper its uptake.
--
Director, Division of Pediatric Dermatology, Cardinal Glennon Children's Hospital, Saint Louis University Health Sciences Center; Owner, Director, Kids Dermatology, St. Louis, Missouri
Elaine C. Siegfried, MD, has disclosed the following relevant financial relationships:
Contracted research from: AI Therapeutics.
Received consulting fees from: Boehringer Ingelheim; Incyte; Regeneron; Sanofi Genzyme; UCB; AbbVie; Verrica; Leo; Novan; Novartis; Pfizer; Pierre Fabre
Received honoraria from: Regeneron; Sanofi Genzyme; Verrica.
Received fees to SSM/SLU related to sponsoring clinical trials from: Regeneron; Verrica; Pierre Fabre; Janssen; Eli Lilly and Company.
Served on the Data Safety Monitoring Board for: UCB; Leo; Pfizer; Novan.
Received grant funding to support 2020-2022 Peds Derm Fellow from: Pfizer.

Bacteriotherapy passes early test in phase 1 atopic dermatitis study
that also demonstrated “encouraging clinical and mechanistic results,” Richard L. Gallo, MD, PhD, and his coinvestigators have reported in Nature Medicine.
Findings from the 1-week, 54-patient trial of a topical formulation containing Staphylococcus hominis A9 (ShA9) offer evidence that the strain directly kills S. aureus, inhibits the production of S. aureus–generated toxins, and enables expansion of a healthy bacterial community, “allowing the rest of the microbiome to start to recover to normal,” Dr. Gallo, professor and chairman of the department of dermatology at the University of California, San Diego, said in an interview.
“And perhaps most exciting,” Dr. Gallo added, is the finding that the subset of patients with AD who were most responsive to the ShA9 compound – approximately two-thirds of the participants who were randomized to receive it – showed improvement in local EASI (Eczema Area and Severity Index) and SCORAD (Scoring Atopic Dermatitis) scores used to assess inflammation. Plans are underway for a larger and longer trial, he said.
S. aureus commonly colonizes patients with AD and exacerbates disease by causing inflammation. In recent years, Dr. Gallo and other investigators have come to believe that AD is a cyclic disease in which the skin’s microbiome affects the host, and the host affects the microbiome. The goal of bacteriotherapy is to break the cycle of S. aureus colonization and improve the skin immune and barrier dysfunction characteristics of AD, Dr. Gallo said.
ShA9, a bacterium isolated from healthy human skin, was chosen as a potential topical therapy for AD based on its capacity both to selectively kill S. aureus and to inhibit toxin production by S. aureus. Dr. Gallo’s team’s preclinical work involved screening thousands of isolates of coagulase-negative staphylococci for gene products that perform these two functions by expressing both antimicrobial peptides (AMPs) and autoinducing peptides (AIPs), the latter of which inhibit the S. aureus quorum-sending system that leads to toxin production. Most patients with AD lack protective strains of coagulase-negative staphylococci, including S. hominis, prior research has found.
The double-blind phase 1 trial randomized 54 adults with moderate-severe AD affecting the ventral forearms in a 2:1 fashion to receive the proprietary lyophilized preparation of ShA9 or an ShA9-free formulation twice daily for 1 week. All participants were culture positive for S. aureus.
Clinical assessments and skin swabs were obtained before and within an hour after the first application of day 1, and swabs were collected on days 4 and 7 within 4 hours of the first application.
Blinded physician assessments and skin swabs were also obtained at 24, 48, and 96 hours after the final dose on day 7.
Based on structured daily diaries, there were no serious adverse events, and significantly fewer adverse events in those treated with ShA9, compared with the vehicle alone; 55.6% versus 83.3%, respectively, were considered to have adverse events.
The adverse event–reporting system captured the normal fluctuation of eczema and considered any report of fluctuation above baseline to be an adverse event. “Patients treated with the [placebo formulation] had the expected high frequency of itching, burning, and pain that you see with AD but it was encouraging that the frequency of reporting these events was significantly less in those treated with the active [formulation],” Dr. Gallo said in the interview.
Their report describes a decrease in S. aureus in participants treated with ShA9, and increases in ShA9 DNA. Not all S. aureus strains were directly killed by ShA9, but all strains had reduced expression of mRNA for psm-alpha, an important virulence factor. That reduced expression correlated with ShA9 AIPs and improved EASI scores, the latter of which was observed in a post-hoc analysis. “Participants with S. aureus not killed by ShA9 were still sensitive to inhibition of toxin production, a mechanistic outcome that predicted clinical improvement in mice and may require longer therapy to observe clinical improvement in humans,” the investigators wrote.
Local eczema severity was not significantly different between the bacteriotherapy and control groups. But the post-hoc analysis showed that after 7 days of treatment, and up to 4 days after treatment was discontinued, the patients with S. aureus that was sensitive to killing by ShA9 (21 out of 35 total who received the bacteriotherapy) showed improvement in EASI and SCORAD scores, compared with control patients.
Future research will assess the compound in both S. aureus culture-positive and culture-negative patients, and in patients with mild disease, Dr. Gallo said.
The trial was conducted at USCD and the National Jewish Health General Clinical Research Center in Denver, and was sponsored by the National Institute of Allergy and Infectious Diseases. The ShA9 formulation and related technology are licensed to MatriSys Bioscience, of which Dr. Gallo is the cofounder and an advisory board member. Dr. Gallo holds equity interest in the company.
that also demonstrated “encouraging clinical and mechanistic results,” Richard L. Gallo, MD, PhD, and his coinvestigators have reported in Nature Medicine.
Findings from the 1-week, 54-patient trial of a topical formulation containing Staphylococcus hominis A9 (ShA9) offer evidence that the strain directly kills S. aureus, inhibits the production of S. aureus–generated toxins, and enables expansion of a healthy bacterial community, “allowing the rest of the microbiome to start to recover to normal,” Dr. Gallo, professor and chairman of the department of dermatology at the University of California, San Diego, said in an interview.
“And perhaps most exciting,” Dr. Gallo added, is the finding that the subset of patients with AD who were most responsive to the ShA9 compound – approximately two-thirds of the participants who were randomized to receive it – showed improvement in local EASI (Eczema Area and Severity Index) and SCORAD (Scoring Atopic Dermatitis) scores used to assess inflammation. Plans are underway for a larger and longer trial, he said.
S. aureus commonly colonizes patients with AD and exacerbates disease by causing inflammation. In recent years, Dr. Gallo and other investigators have come to believe that AD is a cyclic disease in which the skin’s microbiome affects the host, and the host affects the microbiome. The goal of bacteriotherapy is to break the cycle of S. aureus colonization and improve the skin immune and barrier dysfunction characteristics of AD, Dr. Gallo said.
ShA9, a bacterium isolated from healthy human skin, was chosen as a potential topical therapy for AD based on its capacity both to selectively kill S. aureus and to inhibit toxin production by S. aureus. Dr. Gallo’s team’s preclinical work involved screening thousands of isolates of coagulase-negative staphylococci for gene products that perform these two functions by expressing both antimicrobial peptides (AMPs) and autoinducing peptides (AIPs), the latter of which inhibit the S. aureus quorum-sending system that leads to toxin production. Most patients with AD lack protective strains of coagulase-negative staphylococci, including S. hominis, prior research has found.
The double-blind phase 1 trial randomized 54 adults with moderate-severe AD affecting the ventral forearms in a 2:1 fashion to receive the proprietary lyophilized preparation of ShA9 or an ShA9-free formulation twice daily for 1 week. All participants were culture positive for S. aureus.
Clinical assessments and skin swabs were obtained before and within an hour after the first application of day 1, and swabs were collected on days 4 and 7 within 4 hours of the first application.
Blinded physician assessments and skin swabs were also obtained at 24, 48, and 96 hours after the final dose on day 7.
Based on structured daily diaries, there were no serious adverse events, and significantly fewer adverse events in those treated with ShA9, compared with the vehicle alone; 55.6% versus 83.3%, respectively, were considered to have adverse events.
The adverse event–reporting system captured the normal fluctuation of eczema and considered any report of fluctuation above baseline to be an adverse event. “Patients treated with the [placebo formulation] had the expected high frequency of itching, burning, and pain that you see with AD but it was encouraging that the frequency of reporting these events was significantly less in those treated with the active [formulation],” Dr. Gallo said in the interview.
Their report describes a decrease in S. aureus in participants treated with ShA9, and increases in ShA9 DNA. Not all S. aureus strains were directly killed by ShA9, but all strains had reduced expression of mRNA for psm-alpha, an important virulence factor. That reduced expression correlated with ShA9 AIPs and improved EASI scores, the latter of which was observed in a post-hoc analysis. “Participants with S. aureus not killed by ShA9 were still sensitive to inhibition of toxin production, a mechanistic outcome that predicted clinical improvement in mice and may require longer therapy to observe clinical improvement in humans,” the investigators wrote.
Local eczema severity was not significantly different between the bacteriotherapy and control groups. But the post-hoc analysis showed that after 7 days of treatment, and up to 4 days after treatment was discontinued, the patients with S. aureus that was sensitive to killing by ShA9 (21 out of 35 total who received the bacteriotherapy) showed improvement in EASI and SCORAD scores, compared with control patients.
Future research will assess the compound in both S. aureus culture-positive and culture-negative patients, and in patients with mild disease, Dr. Gallo said.
The trial was conducted at USCD and the National Jewish Health General Clinical Research Center in Denver, and was sponsored by the National Institute of Allergy and Infectious Diseases. The ShA9 formulation and related technology are licensed to MatriSys Bioscience, of which Dr. Gallo is the cofounder and an advisory board member. Dr. Gallo holds equity interest in the company.
that also demonstrated “encouraging clinical and mechanistic results,” Richard L. Gallo, MD, PhD, and his coinvestigators have reported in Nature Medicine.
Findings from the 1-week, 54-patient trial of a topical formulation containing Staphylococcus hominis A9 (ShA9) offer evidence that the strain directly kills S. aureus, inhibits the production of S. aureus–generated toxins, and enables expansion of a healthy bacterial community, “allowing the rest of the microbiome to start to recover to normal,” Dr. Gallo, professor and chairman of the department of dermatology at the University of California, San Diego, said in an interview.
“And perhaps most exciting,” Dr. Gallo added, is the finding that the subset of patients with AD who were most responsive to the ShA9 compound – approximately two-thirds of the participants who were randomized to receive it – showed improvement in local EASI (Eczema Area and Severity Index) and SCORAD (Scoring Atopic Dermatitis) scores used to assess inflammation. Plans are underway for a larger and longer trial, he said.
S. aureus commonly colonizes patients with AD and exacerbates disease by causing inflammation. In recent years, Dr. Gallo and other investigators have come to believe that AD is a cyclic disease in which the skin’s microbiome affects the host, and the host affects the microbiome. The goal of bacteriotherapy is to break the cycle of S. aureus colonization and improve the skin immune and barrier dysfunction characteristics of AD, Dr. Gallo said.
ShA9, a bacterium isolated from healthy human skin, was chosen as a potential topical therapy for AD based on its capacity both to selectively kill S. aureus and to inhibit toxin production by S. aureus. Dr. Gallo’s team’s preclinical work involved screening thousands of isolates of coagulase-negative staphylococci for gene products that perform these two functions by expressing both antimicrobial peptides (AMPs) and autoinducing peptides (AIPs), the latter of which inhibit the S. aureus quorum-sending system that leads to toxin production. Most patients with AD lack protective strains of coagulase-negative staphylococci, including S. hominis, prior research has found.
The double-blind phase 1 trial randomized 54 adults with moderate-severe AD affecting the ventral forearms in a 2:1 fashion to receive the proprietary lyophilized preparation of ShA9 or an ShA9-free formulation twice daily for 1 week. All participants were culture positive for S. aureus.
Clinical assessments and skin swabs were obtained before and within an hour after the first application of day 1, and swabs were collected on days 4 and 7 within 4 hours of the first application.
Blinded physician assessments and skin swabs were also obtained at 24, 48, and 96 hours after the final dose on day 7.
Based on structured daily diaries, there were no serious adverse events, and significantly fewer adverse events in those treated with ShA9, compared with the vehicle alone; 55.6% versus 83.3%, respectively, were considered to have adverse events.
The adverse event–reporting system captured the normal fluctuation of eczema and considered any report of fluctuation above baseline to be an adverse event. “Patients treated with the [placebo formulation] had the expected high frequency of itching, burning, and pain that you see with AD but it was encouraging that the frequency of reporting these events was significantly less in those treated with the active [formulation],” Dr. Gallo said in the interview.
Their report describes a decrease in S. aureus in participants treated with ShA9, and increases in ShA9 DNA. Not all S. aureus strains were directly killed by ShA9, but all strains had reduced expression of mRNA for psm-alpha, an important virulence factor. That reduced expression correlated with ShA9 AIPs and improved EASI scores, the latter of which was observed in a post-hoc analysis. “Participants with S. aureus not killed by ShA9 were still sensitive to inhibition of toxin production, a mechanistic outcome that predicted clinical improvement in mice and may require longer therapy to observe clinical improvement in humans,” the investigators wrote.
Local eczema severity was not significantly different between the bacteriotherapy and control groups. But the post-hoc analysis showed that after 7 days of treatment, and up to 4 days after treatment was discontinued, the patients with S. aureus that was sensitive to killing by ShA9 (21 out of 35 total who received the bacteriotherapy) showed improvement in EASI and SCORAD scores, compared with control patients.
Future research will assess the compound in both S. aureus culture-positive and culture-negative patients, and in patients with mild disease, Dr. Gallo said.
The trial was conducted at USCD and the National Jewish Health General Clinical Research Center in Denver, and was sponsored by the National Institute of Allergy and Infectious Diseases. The ShA9 formulation and related technology are licensed to MatriSys Bioscience, of which Dr. Gallo is the cofounder and an advisory board member. Dr. Gallo holds equity interest in the company.
FROM NATURE MEDICINE
Atopic dermatitis in children linked to elevated risk of chronic school absenteeism
.
In addition, parents of children with AD have significantly increased absenteeism from work compared with parents of children without AD.
Those are among key findings from a cross-sectional analysis of data from the Medical Expenditure Panel Surveys (MEPS), reported by Brian T. Cheng and Jonathan I. Silverberg, MD, PhD, MPH. The results were published online March 1 in the Journal of the American Academy of Dermatology.
“Atopic dermatitis is a debilitating disease that profoundly impacts children and their ability to attend school,” the study’s senior author, Dr. Silverberg, director of clinical research in the department of dermatology at George Washington University, Washington, said in an interview. “This is clinically relevant because school absenteeism is a sign of poorly controlled disease and should prompt clinicians to step up their game and aim for tighter control of the child’s atopic dermatitis.”
In an effort to determine the burden and predictors of chronic school absenteeism in children with AD, Mr. Cheng, a medical student at Northwestern University, Chicago, and Dr. Silverberg conducted a cross-sectional retrospective analysis of 124,267 children, adolescents, and young adults between the ages of 3 and 22 years from the 2000-2015 MEPS, which are representative surveys of the U.S. noninstitutionalized population conducted by the Agency for Healthcare Research and Quality. They used ICD-9 codes to determine a diagnosis of AD, psoriasis, and comorbidities; the primary outcome was chronic school absenteeism, defined as missing 15 or more days per year in the United States. MEPS also recorded the number of workdays that parents missed to care for their children or a relative.
The 124,267 individuals evaluated ranged in age between 3 and 22 years. Of these, 3,132 had AD and 200 had psoriasis. In the full cohort, chronic school absenteeism was higher among females, younger children, and those with lower household incomes, and public insurance.
Among children with AD, and those with psoriasis, 68% and 63% missed one or more day of school due to illness, respectively, while 4% in each group missed 15 days or more. Logistic regression analysis revealed that AD was associated with chronic absenteeism overall (adjusted odds ratio, 1.42), and with more severe disease (aOR, 1.33 for mild to moderate disease; aOR, 2.00 for severe disease).
On the other hand, the researchers did not observe any statistical difference in chronic absenteeism among children with versus those without psoriasis (aOR, 1.26).
The researchers also found that parents of children with versus parents of children without AD had a higher prevalence of absenteeism from work (an aOR of 1.28 among fathers, P = .009; and an aOR of 1.24 among mothers, P = .003).
In other findings, chronic absenteeism among children with AD was associated with poor/near poor/low income (aOR, 4.61) and comorbid disease (aOR, 3.35 for depression and aOR, 3.83 for asthma).
The investigators recommend that clinicians screen for and aim to reduce school absenteeism and parental work absenteeism in children with AD.
“I typically ask ‘Has (child’s name) missed any school because of their eczema?’ and follow-up with ‘What about from asthma or allergies?’ ” Dr. Silverberg said. “If the parent’s answer is yes to the first question, then I follow-up with more open-ended probing questions to understand why. Is it from all the doctor visits? Not sleeping well? Severe itch or pain? Poor sleep? Feeling sad or depressed? An answer of yes to each of these would prompt a potentially different treatment decision.”
The study received financial support from the Dermatology Foundation. The authors reported having no financial disclosures.
.
In addition, parents of children with AD have significantly increased absenteeism from work compared with parents of children without AD.
Those are among key findings from a cross-sectional analysis of data from the Medical Expenditure Panel Surveys (MEPS), reported by Brian T. Cheng and Jonathan I. Silverberg, MD, PhD, MPH. The results were published online March 1 in the Journal of the American Academy of Dermatology.
“Atopic dermatitis is a debilitating disease that profoundly impacts children and their ability to attend school,” the study’s senior author, Dr. Silverberg, director of clinical research in the department of dermatology at George Washington University, Washington, said in an interview. “This is clinically relevant because school absenteeism is a sign of poorly controlled disease and should prompt clinicians to step up their game and aim for tighter control of the child’s atopic dermatitis.”
In an effort to determine the burden and predictors of chronic school absenteeism in children with AD, Mr. Cheng, a medical student at Northwestern University, Chicago, and Dr. Silverberg conducted a cross-sectional retrospective analysis of 124,267 children, adolescents, and young adults between the ages of 3 and 22 years from the 2000-2015 MEPS, which are representative surveys of the U.S. noninstitutionalized population conducted by the Agency for Healthcare Research and Quality. They used ICD-9 codes to determine a diagnosis of AD, psoriasis, and comorbidities; the primary outcome was chronic school absenteeism, defined as missing 15 or more days per year in the United States. MEPS also recorded the number of workdays that parents missed to care for their children or a relative.
The 124,267 individuals evaluated ranged in age between 3 and 22 years. Of these, 3,132 had AD and 200 had psoriasis. In the full cohort, chronic school absenteeism was higher among females, younger children, and those with lower household incomes, and public insurance.
Among children with AD, and those with psoriasis, 68% and 63% missed one or more day of school due to illness, respectively, while 4% in each group missed 15 days or more. Logistic regression analysis revealed that AD was associated with chronic absenteeism overall (adjusted odds ratio, 1.42), and with more severe disease (aOR, 1.33 for mild to moderate disease; aOR, 2.00 for severe disease).
On the other hand, the researchers did not observe any statistical difference in chronic absenteeism among children with versus those without psoriasis (aOR, 1.26).
The researchers also found that parents of children with versus parents of children without AD had a higher prevalence of absenteeism from work (an aOR of 1.28 among fathers, P = .009; and an aOR of 1.24 among mothers, P = .003).
In other findings, chronic absenteeism among children with AD was associated with poor/near poor/low income (aOR, 4.61) and comorbid disease (aOR, 3.35 for depression and aOR, 3.83 for asthma).
The investigators recommend that clinicians screen for and aim to reduce school absenteeism and parental work absenteeism in children with AD.
“I typically ask ‘Has (child’s name) missed any school because of their eczema?’ and follow-up with ‘What about from asthma or allergies?’ ” Dr. Silverberg said. “If the parent’s answer is yes to the first question, then I follow-up with more open-ended probing questions to understand why. Is it from all the doctor visits? Not sleeping well? Severe itch or pain? Poor sleep? Feeling sad or depressed? An answer of yes to each of these would prompt a potentially different treatment decision.”
The study received financial support from the Dermatology Foundation. The authors reported having no financial disclosures.
.
In addition, parents of children with AD have significantly increased absenteeism from work compared with parents of children without AD.
Those are among key findings from a cross-sectional analysis of data from the Medical Expenditure Panel Surveys (MEPS), reported by Brian T. Cheng and Jonathan I. Silverberg, MD, PhD, MPH. The results were published online March 1 in the Journal of the American Academy of Dermatology.
“Atopic dermatitis is a debilitating disease that profoundly impacts children and their ability to attend school,” the study’s senior author, Dr. Silverberg, director of clinical research in the department of dermatology at George Washington University, Washington, said in an interview. “This is clinically relevant because school absenteeism is a sign of poorly controlled disease and should prompt clinicians to step up their game and aim for tighter control of the child’s atopic dermatitis.”
In an effort to determine the burden and predictors of chronic school absenteeism in children with AD, Mr. Cheng, a medical student at Northwestern University, Chicago, and Dr. Silverberg conducted a cross-sectional retrospective analysis of 124,267 children, adolescents, and young adults between the ages of 3 and 22 years from the 2000-2015 MEPS, which are representative surveys of the U.S. noninstitutionalized population conducted by the Agency for Healthcare Research and Quality. They used ICD-9 codes to determine a diagnosis of AD, psoriasis, and comorbidities; the primary outcome was chronic school absenteeism, defined as missing 15 or more days per year in the United States. MEPS also recorded the number of workdays that parents missed to care for their children or a relative.
The 124,267 individuals evaluated ranged in age between 3 and 22 years. Of these, 3,132 had AD and 200 had psoriasis. In the full cohort, chronic school absenteeism was higher among females, younger children, and those with lower household incomes, and public insurance.
Among children with AD, and those with psoriasis, 68% and 63% missed one or more day of school due to illness, respectively, while 4% in each group missed 15 days or more. Logistic regression analysis revealed that AD was associated with chronic absenteeism overall (adjusted odds ratio, 1.42), and with more severe disease (aOR, 1.33 for mild to moderate disease; aOR, 2.00 for severe disease).
On the other hand, the researchers did not observe any statistical difference in chronic absenteeism among children with versus those without psoriasis (aOR, 1.26).
The researchers also found that parents of children with versus parents of children without AD had a higher prevalence of absenteeism from work (an aOR of 1.28 among fathers, P = .009; and an aOR of 1.24 among mothers, P = .003).
In other findings, chronic absenteeism among children with AD was associated with poor/near poor/low income (aOR, 4.61) and comorbid disease (aOR, 3.35 for depression and aOR, 3.83 for asthma).
The investigators recommend that clinicians screen for and aim to reduce school absenteeism and parental work absenteeism in children with AD.
“I typically ask ‘Has (child’s name) missed any school because of their eczema?’ and follow-up with ‘What about from asthma or allergies?’ ” Dr. Silverberg said. “If the parent’s answer is yes to the first question, then I follow-up with more open-ended probing questions to understand why. Is it from all the doctor visits? Not sleeping well? Severe itch or pain? Poor sleep? Feeling sad or depressed? An answer of yes to each of these would prompt a potentially different treatment decision.”
The study received financial support from the Dermatology Foundation. The authors reported having no financial disclosures.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Atopic dermatitis, sleep difficulties often intertwined
According to Phyllis C. Zee, MD, PhD, proinflammatory cytokines influence neural processes that affect sleep and circadian rhythm. “It’s almost like when you’re most vulnerable, when you’re sleeping, the immune system is kind of poised for attack,” Dr. Zee, chief of the division of sleep medicine at Northwestern University, Chicago, said at the Revolutionizing Atopic Dermatitis symposium. “This is normal, and perhaps in some of these inflammatory disorders, it’s gone a little haywire.”
Circulation of interleukins and cytokines are high in the morning, become lower in the afternoon, and then get higher again in the evening hours and into the night during sleep, she continued. “Whereas if you look at something like blood flow, it increases on a diurnal basis,” she said. “It’s higher during the day and a little bit lower during the mid-day, and a little bit higher during the evening. That parallels changes in the sebum production of the skin and the transepidermal water loss, which has been implicated in some of the symptoms of AD. What’s curious about this is that the transdermal/epidermal water loss is really highest during the sleep period. Some of this is sleep gated, but some of this is circadian gated as well. There’s a bidirectional relationship between sleep and immunity.”
Disturbance of sleep can have multiple consequences. It can activate the hypothalamic-pituitary-adrenal axis through autonomic activation, increase brain metabolic activity, trigger mood disturbances and cognitive impairment, and cause daytime sleepiness and health consequences that affect cardiometabolic and immunologic health.
One study conducted by Anna B. Fishbein, MD, Dr. Zee, and colleagues at Northwestern examined the effects of sleep duration and sleep disruption and movements in 38 children with and without moderate to severe AD. It found that children with AD get about 1 hour less of sleep per night overall, compared with age-matched healthy controls. “It’s not so much difficulty falling asleep, but more difficulty staying asleep as determined by wake after sleep onset,” said Dr. Zee, who is also a professor of neurology at Northwestern.
A study of 34,613 adults who participated in the 2012 National Health and Nutrition Examination Survey found that eczema increased the odds of fatigue (odds ratio, 2.97), daytime sleepiness (OR, 2.66), and regular insomnia (OR, 2.36).
“Very importantly, it predicted poor health,” said Dr. Zee, who was one of the study’s coauthors. “This gives us an opportunity to think about how we can improve sleep to improve outcomes.”
Dr. Zee advises dermatologists and primary care clinicians to ask patients with AD about their sleep health by using a screening tool such as the self-reported STOP questionnaire, which consists of the following questions: “Do you snore loudly?” “Do you often feel tired, fatigued, or sleepy during daytime?” “Has anyone observed you stop breathing during your sleep?” “Do you have or are you being treated for high blood pressure?”
Other clinical indicators of a sleep disorder, such as obstructive sleep apnea (OSA), include having a neck circumference of 17 inches or greater in men and 16 inches or greater in women. “You want to also do a brief upper-airway examination, the Mallampati classification where you say to the patient, ‘open your mouth, don’t stick your mouth out too much,’ and you look at how crowded the upper airway is,” Dr. Zee said . “Someone with a Mallampati score of 3 has a very high risk of having sleep apnea.”
She also recommends asking patients with AD if they have difficulty falling asleep or staying asleep 3 or more nights per week, and about the frequency and duration of awakenings. “Maybe they have insomnia as a disorder,” she said. “If they have trouble falling asleep, maybe they have a circadian rhythm disorder. You want to ask about snoring, choking, and stop breathing episodes, because those are symptoms of sleep apnea. You want to ask about itch, uncomfortable sensations in the limbs during sleep or while trying to get to sleep, because that may be something like restless legs syndrome. Sleep disorder assessment is important because it impair daytime function, cognition, attention, and disruptive behavior, especially in children.”
For the management of insomnia, try behavioral approaches first. “You don’t want to try medications from the get-go,” Dr. Zee advised. Techniques include sleep hygiene and stimulus control therapy, “to make the bedroom a safe place to sleep. Lower the temperature a little bit and get rid of the allergens as much as possible. Relaxation and cognitive-behavioral therapy can also help. If you get a lot of light during the day, structure your physical activity, and watch what and when you eat.”
An OSA diagnosis requires evaluation of objective information from a sleep study. Common treatments of mild to moderate OSA include nasal continuous positive airway pressure and oral appliances.
Dr. Zee disclosed that she had received research funding from the National Institutes of Health, Jazz Pharmaceuticals, Harmony and Apnimed. She also serves on the scientific advisory board of Eisai, Jazz, CVS-Caremark, Takeda, and Sanofi-Aventis, and holds stock in Teva.
According to Phyllis C. Zee, MD, PhD, proinflammatory cytokines influence neural processes that affect sleep and circadian rhythm. “It’s almost like when you’re most vulnerable, when you’re sleeping, the immune system is kind of poised for attack,” Dr. Zee, chief of the division of sleep medicine at Northwestern University, Chicago, said at the Revolutionizing Atopic Dermatitis symposium. “This is normal, and perhaps in some of these inflammatory disorders, it’s gone a little haywire.”
Circulation of interleukins and cytokines are high in the morning, become lower in the afternoon, and then get higher again in the evening hours and into the night during sleep, she continued. “Whereas if you look at something like blood flow, it increases on a diurnal basis,” she said. “It’s higher during the day and a little bit lower during the mid-day, and a little bit higher during the evening. That parallels changes in the sebum production of the skin and the transepidermal water loss, which has been implicated in some of the symptoms of AD. What’s curious about this is that the transdermal/epidermal water loss is really highest during the sleep period. Some of this is sleep gated, but some of this is circadian gated as well. There’s a bidirectional relationship between sleep and immunity.”
Disturbance of sleep can have multiple consequences. It can activate the hypothalamic-pituitary-adrenal axis through autonomic activation, increase brain metabolic activity, trigger mood disturbances and cognitive impairment, and cause daytime sleepiness and health consequences that affect cardiometabolic and immunologic health.
One study conducted by Anna B. Fishbein, MD, Dr. Zee, and colleagues at Northwestern examined the effects of sleep duration and sleep disruption and movements in 38 children with and without moderate to severe AD. It found that children with AD get about 1 hour less of sleep per night overall, compared with age-matched healthy controls. “It’s not so much difficulty falling asleep, but more difficulty staying asleep as determined by wake after sleep onset,” said Dr. Zee, who is also a professor of neurology at Northwestern.
A study of 34,613 adults who participated in the 2012 National Health and Nutrition Examination Survey found that eczema increased the odds of fatigue (odds ratio, 2.97), daytime sleepiness (OR, 2.66), and regular insomnia (OR, 2.36).
“Very importantly, it predicted poor health,” said Dr. Zee, who was one of the study’s coauthors. “This gives us an opportunity to think about how we can improve sleep to improve outcomes.”
Dr. Zee advises dermatologists and primary care clinicians to ask patients with AD about their sleep health by using a screening tool such as the self-reported STOP questionnaire, which consists of the following questions: “Do you snore loudly?” “Do you often feel tired, fatigued, or sleepy during daytime?” “Has anyone observed you stop breathing during your sleep?” “Do you have or are you being treated for high blood pressure?”
Other clinical indicators of a sleep disorder, such as obstructive sleep apnea (OSA), include having a neck circumference of 17 inches or greater in men and 16 inches or greater in women. “You want to also do a brief upper-airway examination, the Mallampati classification where you say to the patient, ‘open your mouth, don’t stick your mouth out too much,’ and you look at how crowded the upper airway is,” Dr. Zee said . “Someone with a Mallampati score of 3 has a very high risk of having sleep apnea.”
She also recommends asking patients with AD if they have difficulty falling asleep or staying asleep 3 or more nights per week, and about the frequency and duration of awakenings. “Maybe they have insomnia as a disorder,” she said. “If they have trouble falling asleep, maybe they have a circadian rhythm disorder. You want to ask about snoring, choking, and stop breathing episodes, because those are symptoms of sleep apnea. You want to ask about itch, uncomfortable sensations in the limbs during sleep or while trying to get to sleep, because that may be something like restless legs syndrome. Sleep disorder assessment is important because it impair daytime function, cognition, attention, and disruptive behavior, especially in children.”
For the management of insomnia, try behavioral approaches first. “You don’t want to try medications from the get-go,” Dr. Zee advised. Techniques include sleep hygiene and stimulus control therapy, “to make the bedroom a safe place to sleep. Lower the temperature a little bit and get rid of the allergens as much as possible. Relaxation and cognitive-behavioral therapy can also help. If you get a lot of light during the day, structure your physical activity, and watch what and when you eat.”
An OSA diagnosis requires evaluation of objective information from a sleep study. Common treatments of mild to moderate OSA include nasal continuous positive airway pressure and oral appliances.
Dr. Zee disclosed that she had received research funding from the National Institutes of Health, Jazz Pharmaceuticals, Harmony and Apnimed. She also serves on the scientific advisory board of Eisai, Jazz, CVS-Caremark, Takeda, and Sanofi-Aventis, and holds stock in Teva.
According to Phyllis C. Zee, MD, PhD, proinflammatory cytokines influence neural processes that affect sleep and circadian rhythm. “It’s almost like when you’re most vulnerable, when you’re sleeping, the immune system is kind of poised for attack,” Dr. Zee, chief of the division of sleep medicine at Northwestern University, Chicago, said at the Revolutionizing Atopic Dermatitis symposium. “This is normal, and perhaps in some of these inflammatory disorders, it’s gone a little haywire.”
Circulation of interleukins and cytokines are high in the morning, become lower in the afternoon, and then get higher again in the evening hours and into the night during sleep, she continued. “Whereas if you look at something like blood flow, it increases on a diurnal basis,” she said. “It’s higher during the day and a little bit lower during the mid-day, and a little bit higher during the evening. That parallels changes in the sebum production of the skin and the transepidermal water loss, which has been implicated in some of the symptoms of AD. What’s curious about this is that the transdermal/epidermal water loss is really highest during the sleep period. Some of this is sleep gated, but some of this is circadian gated as well. There’s a bidirectional relationship between sleep and immunity.”
Disturbance of sleep can have multiple consequences. It can activate the hypothalamic-pituitary-adrenal axis through autonomic activation, increase brain metabolic activity, trigger mood disturbances and cognitive impairment, and cause daytime sleepiness and health consequences that affect cardiometabolic and immunologic health.
One study conducted by Anna B. Fishbein, MD, Dr. Zee, and colleagues at Northwestern examined the effects of sleep duration and sleep disruption and movements in 38 children with and without moderate to severe AD. It found that children with AD get about 1 hour less of sleep per night overall, compared with age-matched healthy controls. “It’s not so much difficulty falling asleep, but more difficulty staying asleep as determined by wake after sleep onset,” said Dr. Zee, who is also a professor of neurology at Northwestern.
A study of 34,613 adults who participated in the 2012 National Health and Nutrition Examination Survey found that eczema increased the odds of fatigue (odds ratio, 2.97), daytime sleepiness (OR, 2.66), and regular insomnia (OR, 2.36).
“Very importantly, it predicted poor health,” said Dr. Zee, who was one of the study’s coauthors. “This gives us an opportunity to think about how we can improve sleep to improve outcomes.”
Dr. Zee advises dermatologists and primary care clinicians to ask patients with AD about their sleep health by using a screening tool such as the self-reported STOP questionnaire, which consists of the following questions: “Do you snore loudly?” “Do you often feel tired, fatigued, or sleepy during daytime?” “Has anyone observed you stop breathing during your sleep?” “Do you have or are you being treated for high blood pressure?”
Other clinical indicators of a sleep disorder, such as obstructive sleep apnea (OSA), include having a neck circumference of 17 inches or greater in men and 16 inches or greater in women. “You want to also do a brief upper-airway examination, the Mallampati classification where you say to the patient, ‘open your mouth, don’t stick your mouth out too much,’ and you look at how crowded the upper airway is,” Dr. Zee said . “Someone with a Mallampati score of 3 has a very high risk of having sleep apnea.”
She also recommends asking patients with AD if they have difficulty falling asleep or staying asleep 3 or more nights per week, and about the frequency and duration of awakenings. “Maybe they have insomnia as a disorder,” she said. “If they have trouble falling asleep, maybe they have a circadian rhythm disorder. You want to ask about snoring, choking, and stop breathing episodes, because those are symptoms of sleep apnea. You want to ask about itch, uncomfortable sensations in the limbs during sleep or while trying to get to sleep, because that may be something like restless legs syndrome. Sleep disorder assessment is important because it impair daytime function, cognition, attention, and disruptive behavior, especially in children.”
For the management of insomnia, try behavioral approaches first. “You don’t want to try medications from the get-go,” Dr. Zee advised. Techniques include sleep hygiene and stimulus control therapy, “to make the bedroom a safe place to sleep. Lower the temperature a little bit and get rid of the allergens as much as possible. Relaxation and cognitive-behavioral therapy can also help. If you get a lot of light during the day, structure your physical activity, and watch what and when you eat.”
An OSA diagnosis requires evaluation of objective information from a sleep study. Common treatments of mild to moderate OSA include nasal continuous positive airway pressure and oral appliances.
Dr. Zee disclosed that she had received research funding from the National Institutes of Health, Jazz Pharmaceuticals, Harmony and Apnimed. She also serves on the scientific advisory board of Eisai, Jazz, CVS-Caremark, Takeda, and Sanofi-Aventis, and holds stock in Teva.
FROM REVOLUTIONIZING AD 2020
What drives treatment satisfaction among adults with atopic dermatitis?
.
Satisfaction scores were higher when specialists prescribed systemic therapy, but were lower when nonspecialists prescribed systemic therapy and when specialists prescribed only topical therapy.
Those are among key findings from an analysis of the Medical Expenditure Panel Surveys reported by Brian T. Cheng during a late-breaking research session at the Revolutionizing Atopic Dermatitis virtual symposium.
“AD management is complex,” said Mr. Cheng, a medical student at Northwestern University, Chicago. “It includes patient education about trigger avoidance, over-the-counter and prescription topical therapies, as well as systemic therapies. Previous studies have shown major decrements to quality of life as well as atopic and non-atopic comorbidities in these patients. The burden of AD and their comorbidities, as well as their management, may impact patient satisfaction.”
Prior studies have demonstrated that patient satisfaction is associated with improvements in clinical outcomes, increased patient retention, and reduced malpractice claims (Br J Dermatol. 2001 Oct;145[4]:617-23, Arch Dermatol 2008 Feb;144[2]:263-5). However, since data on patient satisfaction in AD are limited, Mr. Cheng and the study’s senior author, Jonathan I. Silverberg, MD, PhD, MPH, set out to examine overall patient satisfaction among adults with AD, to determine associations of patient satisfaction with patterns of health care utilization, and to identify predictors of higher satisfaction among these adults.
The researchers conducted a cross-sectional retrospective analysis of 3,810 patients from the 2000-2015 Medical Expenditure Panel Surveys, representative surveys of the U.S. noninstitutionalized population conducted annually by the Agency for Healthcare Research and Quality. They used ICD-9 codes 691 and 692 to determine AD diagnosis and five Consumer Assessment of Health Plans Survey (CAHPS) questions to assess patients’ satisfaction with their clinicians. “These questions have been extensively validated to correlate with global satisfaction,” Mr. Cheng said. “These are not disease-specific and allow for comparison across multiple diseases.”
Next, the researchers created a composite satisfaction score based on the methods of Anthony Jerant, MD, of the University of California, Davis, and colleagues. They adjusted each question in the CAHPS survey to have an equal weight and then summed these into a composite satisfaction score. “We examined patient satisfaction comparing across diseases, and based on the guidelines from the AHRQ to isolate that impact of patient-physician interaction, we adjusted for sociodemographics, mental and physical health status, self-reported health rating, as well as multimorbidity and comorbid diseases.”
Compared with adults who are healthy, adults with AD had lower patient satisfaction overall. “Moreover, people with AD had lower satisfaction compared to those with psoriasis, which may reflect more substantial itch burden as well as the greater comorbid disease challenges in management,” Mr. Cheng said. “It may also reflect the renaissance in psoriasis treatment over the last 10-20 years, giving a wider spectrum of treatment and thus a higher patient satisfaction.”
Among adults with AD, lower satisfaction was consistent across all domains of CAHPS. For the question of “How often health providers listen carefully to you” the adjusted OR (aOR) was 0.87 (P = .008). For the question of “How often health providers explain things in a way that was easy to understand” the aOR was 0.89 (P = .003). For the question of “How often health providers spent enough time with you” the aOR was 0.86 (P = .0001). For “How often providers showed respect for what you had to say” the aOR was 0.91 (P = .02).
Recognizing that treatment regimens are complex and used differently by provider type, the researchers examined interactions between specialists (dermatologists and allergists) and treatment type. “Previous studies found dermatologists treat more severe, chronic AD,” Mr. Cheng said. “We found here that there was lower satisfaction among those treated with topical therapy and by specialists, which may reflect inadequate disease control. We also found lower satisfaction among those treated with systemic therapy by primary care physicians. This may reflect that these patients are not achieving optimal therapy. We found that satisfaction was highest among those treated with systemic therapy and by dermatologists and allergists.”
Socioeconomic, racial/ethnic, and health care disparities were observed in terms of satisfaction among this cohort. The following characteristics were significantly associated with lower patient satisfaction, compared with the general cohort of adults with AD: poor to low income (aOR, –1.82; P less than .0001), multiracial/other race (aOR, –2.34; P = .0001), Hispanic ethnicity (aOR, –1.40; P = .007), and having no insurance coverage (aOR, –4.53; P less than .0001).
“Moreover, those with multimorbidity had even lower satisfaction,” Mr. Cheng said. “In previous studies, AD has been linked with many other comorbidities. This may reflect that these patients are not being adequately managed overall. So, there’s a need here for multidisciplinary care to ensure that all of these comorbidities and the full spectrum of symptoms are being managed adequately.”
He concluded that future research is needed to determine strategies to optimize patient satisfaction in adults with AD.
“I’m not sure how much more provocative you can get in terms of data,” added Dr. Silverberg, director of clinical research and contact dermatitis at George Washington University, Washington. “It’s really eye-opening. I think many clinicians may feel like they’re doing a perfect job in managing this disease. These data suggest that at least at the national level that may not be the case.”
Mr. Cheng reported having no financial disclosures. Dr. Silverberg reported that he is a consultant to and/or an advisory board member for several pharmaceutical companies. He is also a speaker for Regeneron and Sanofi and has received a grant from Galderma.
.
Satisfaction scores were higher when specialists prescribed systemic therapy, but were lower when nonspecialists prescribed systemic therapy and when specialists prescribed only topical therapy.
Those are among key findings from an analysis of the Medical Expenditure Panel Surveys reported by Brian T. Cheng during a late-breaking research session at the Revolutionizing Atopic Dermatitis virtual symposium.
“AD management is complex,” said Mr. Cheng, a medical student at Northwestern University, Chicago. “It includes patient education about trigger avoidance, over-the-counter and prescription topical therapies, as well as systemic therapies. Previous studies have shown major decrements to quality of life as well as atopic and non-atopic comorbidities in these patients. The burden of AD and their comorbidities, as well as their management, may impact patient satisfaction.”
Prior studies have demonstrated that patient satisfaction is associated with improvements in clinical outcomes, increased patient retention, and reduced malpractice claims (Br J Dermatol. 2001 Oct;145[4]:617-23, Arch Dermatol 2008 Feb;144[2]:263-5). However, since data on patient satisfaction in AD are limited, Mr. Cheng and the study’s senior author, Jonathan I. Silverberg, MD, PhD, MPH, set out to examine overall patient satisfaction among adults with AD, to determine associations of patient satisfaction with patterns of health care utilization, and to identify predictors of higher satisfaction among these adults.
The researchers conducted a cross-sectional retrospective analysis of 3,810 patients from the 2000-2015 Medical Expenditure Panel Surveys, representative surveys of the U.S. noninstitutionalized population conducted annually by the Agency for Healthcare Research and Quality. They used ICD-9 codes 691 and 692 to determine AD diagnosis and five Consumer Assessment of Health Plans Survey (CAHPS) questions to assess patients’ satisfaction with their clinicians. “These questions have been extensively validated to correlate with global satisfaction,” Mr. Cheng said. “These are not disease-specific and allow for comparison across multiple diseases.”
Next, the researchers created a composite satisfaction score based on the methods of Anthony Jerant, MD, of the University of California, Davis, and colleagues. They adjusted each question in the CAHPS survey to have an equal weight and then summed these into a composite satisfaction score. “We examined patient satisfaction comparing across diseases, and based on the guidelines from the AHRQ to isolate that impact of patient-physician interaction, we adjusted for sociodemographics, mental and physical health status, self-reported health rating, as well as multimorbidity and comorbid diseases.”
Compared with adults who are healthy, adults with AD had lower patient satisfaction overall. “Moreover, people with AD had lower satisfaction compared to those with psoriasis, which may reflect more substantial itch burden as well as the greater comorbid disease challenges in management,” Mr. Cheng said. “It may also reflect the renaissance in psoriasis treatment over the last 10-20 years, giving a wider spectrum of treatment and thus a higher patient satisfaction.”
Among adults with AD, lower satisfaction was consistent across all domains of CAHPS. For the question of “How often health providers listen carefully to you” the adjusted OR (aOR) was 0.87 (P = .008). For the question of “How often health providers explain things in a way that was easy to understand” the aOR was 0.89 (P = .003). For the question of “How often health providers spent enough time with you” the aOR was 0.86 (P = .0001). For “How often providers showed respect for what you had to say” the aOR was 0.91 (P = .02).
Recognizing that treatment regimens are complex and used differently by provider type, the researchers examined interactions between specialists (dermatologists and allergists) and treatment type. “Previous studies found dermatologists treat more severe, chronic AD,” Mr. Cheng said. “We found here that there was lower satisfaction among those treated with topical therapy and by specialists, which may reflect inadequate disease control. We also found lower satisfaction among those treated with systemic therapy by primary care physicians. This may reflect that these patients are not achieving optimal therapy. We found that satisfaction was highest among those treated with systemic therapy and by dermatologists and allergists.”
Socioeconomic, racial/ethnic, and health care disparities were observed in terms of satisfaction among this cohort. The following characteristics were significantly associated with lower patient satisfaction, compared with the general cohort of adults with AD: poor to low income (aOR, –1.82; P less than .0001), multiracial/other race (aOR, –2.34; P = .0001), Hispanic ethnicity (aOR, –1.40; P = .007), and having no insurance coverage (aOR, –4.53; P less than .0001).
“Moreover, those with multimorbidity had even lower satisfaction,” Mr. Cheng said. “In previous studies, AD has been linked with many other comorbidities. This may reflect that these patients are not being adequately managed overall. So, there’s a need here for multidisciplinary care to ensure that all of these comorbidities and the full spectrum of symptoms are being managed adequately.”
He concluded that future research is needed to determine strategies to optimize patient satisfaction in adults with AD.
“I’m not sure how much more provocative you can get in terms of data,” added Dr. Silverberg, director of clinical research and contact dermatitis at George Washington University, Washington. “It’s really eye-opening. I think many clinicians may feel like they’re doing a perfect job in managing this disease. These data suggest that at least at the national level that may not be the case.”
Mr. Cheng reported having no financial disclosures. Dr. Silverberg reported that he is a consultant to and/or an advisory board member for several pharmaceutical companies. He is also a speaker for Regeneron and Sanofi and has received a grant from Galderma.
.
Satisfaction scores were higher when specialists prescribed systemic therapy, but were lower when nonspecialists prescribed systemic therapy and when specialists prescribed only topical therapy.
Those are among key findings from an analysis of the Medical Expenditure Panel Surveys reported by Brian T. Cheng during a late-breaking research session at the Revolutionizing Atopic Dermatitis virtual symposium.
“AD management is complex,” said Mr. Cheng, a medical student at Northwestern University, Chicago. “It includes patient education about trigger avoidance, over-the-counter and prescription topical therapies, as well as systemic therapies. Previous studies have shown major decrements to quality of life as well as atopic and non-atopic comorbidities in these patients. The burden of AD and their comorbidities, as well as their management, may impact patient satisfaction.”
Prior studies have demonstrated that patient satisfaction is associated with improvements in clinical outcomes, increased patient retention, and reduced malpractice claims (Br J Dermatol. 2001 Oct;145[4]:617-23, Arch Dermatol 2008 Feb;144[2]:263-5). However, since data on patient satisfaction in AD are limited, Mr. Cheng and the study’s senior author, Jonathan I. Silverberg, MD, PhD, MPH, set out to examine overall patient satisfaction among adults with AD, to determine associations of patient satisfaction with patterns of health care utilization, and to identify predictors of higher satisfaction among these adults.
The researchers conducted a cross-sectional retrospective analysis of 3,810 patients from the 2000-2015 Medical Expenditure Panel Surveys, representative surveys of the U.S. noninstitutionalized population conducted annually by the Agency for Healthcare Research and Quality. They used ICD-9 codes 691 and 692 to determine AD diagnosis and five Consumer Assessment of Health Plans Survey (CAHPS) questions to assess patients’ satisfaction with their clinicians. “These questions have been extensively validated to correlate with global satisfaction,” Mr. Cheng said. “These are not disease-specific and allow for comparison across multiple diseases.”
Next, the researchers created a composite satisfaction score based on the methods of Anthony Jerant, MD, of the University of California, Davis, and colleagues. They adjusted each question in the CAHPS survey to have an equal weight and then summed these into a composite satisfaction score. “We examined patient satisfaction comparing across diseases, and based on the guidelines from the AHRQ to isolate that impact of patient-physician interaction, we adjusted for sociodemographics, mental and physical health status, self-reported health rating, as well as multimorbidity and comorbid diseases.”
Compared with adults who are healthy, adults with AD had lower patient satisfaction overall. “Moreover, people with AD had lower satisfaction compared to those with psoriasis, which may reflect more substantial itch burden as well as the greater comorbid disease challenges in management,” Mr. Cheng said. “It may also reflect the renaissance in psoriasis treatment over the last 10-20 years, giving a wider spectrum of treatment and thus a higher patient satisfaction.”
Among adults with AD, lower satisfaction was consistent across all domains of CAHPS. For the question of “How often health providers listen carefully to you” the adjusted OR (aOR) was 0.87 (P = .008). For the question of “How often health providers explain things in a way that was easy to understand” the aOR was 0.89 (P = .003). For the question of “How often health providers spent enough time with you” the aOR was 0.86 (P = .0001). For “How often providers showed respect for what you had to say” the aOR was 0.91 (P = .02).
Recognizing that treatment regimens are complex and used differently by provider type, the researchers examined interactions between specialists (dermatologists and allergists) and treatment type. “Previous studies found dermatologists treat more severe, chronic AD,” Mr. Cheng said. “We found here that there was lower satisfaction among those treated with topical therapy and by specialists, which may reflect inadequate disease control. We also found lower satisfaction among those treated with systemic therapy by primary care physicians. This may reflect that these patients are not achieving optimal therapy. We found that satisfaction was highest among those treated with systemic therapy and by dermatologists and allergists.”
Socioeconomic, racial/ethnic, and health care disparities were observed in terms of satisfaction among this cohort. The following characteristics were significantly associated with lower patient satisfaction, compared with the general cohort of adults with AD: poor to low income (aOR, –1.82; P less than .0001), multiracial/other race (aOR, –2.34; P = .0001), Hispanic ethnicity (aOR, –1.40; P = .007), and having no insurance coverage (aOR, –4.53; P less than .0001).
“Moreover, those with multimorbidity had even lower satisfaction,” Mr. Cheng said. “In previous studies, AD has been linked with many other comorbidities. This may reflect that these patients are not being adequately managed overall. So, there’s a need here for multidisciplinary care to ensure that all of these comorbidities and the full spectrum of symptoms are being managed adequately.”
He concluded that future research is needed to determine strategies to optimize patient satisfaction in adults with AD.
“I’m not sure how much more provocative you can get in terms of data,” added Dr. Silverberg, director of clinical research and contact dermatitis at George Washington University, Washington. “It’s really eye-opening. I think many clinicians may feel like they’re doing a perfect job in managing this disease. These data suggest that at least at the national level that may not be the case.”
Mr. Cheng reported having no financial disclosures. Dr. Silverberg reported that he is a consultant to and/or an advisory board member for several pharmaceutical companies. He is also a speaker for Regeneron and Sanofi and has received a grant from Galderma.
FROM REVOLUTIONIZING AD 2020
Novel oral agent effective in teens with atopic dermatitis
Abrocitinib, an investigational drug proven to be a safe and effective treatment for moderate to severe atopic dermatitis (AD) in adults 18 years and older, is also safe and effective in patients aged 12-17 years, according to a randomized trial of the oral, once-daily Janus kinase (JAK) 1 selective inhibitor, used in combination with medicated topical therapy.
The results, from the phase 3 JADE TEEN study, were presented during an oral abstract session at the annual meeting of the American Academy of Allergy, Asthma, and Immunology, held virtually this year.
“We’re very excited about the introduction of oral JAKs into our armamentarium for atopic dermatitis,” lead author Lawrence Eichenfield, MD, professor of dermatology and pediatrics, University of California, San Diego, and chief of pediatric and adolescent dermatology, Rady Children’s Hospital, also in San Diego, said in an interview.
AD ranges in severity, and there is a great deal of moderate to severe AD that has a tremendous negative impact on the individual, Dr. Eichenfield said. “Traditionally we have treated it with intermittent topical corticosteroids, but this has left a significant percentage of patients without long-term disease control.”
JAK inhibitors are effective mediators of the inflammation response that occurs in moderate to severe AD. They inhibit the stimulation of the JAK pathway and allow anti-inflammatory effects and therefore have potential, especially in more severe disease, Dr. Eichenfield said.
In the current study, which is a spin-off of the original study that looked at abrocitinib in adults, he and his team randomly assigned 285 teens (mean age, 14.9 years; 50.9% male; 56.1% White) with moderate to severe AD to receive one of the following treatments for 12 weeks: abrocitinib 200 mg plus topical therapy (95); abrocitinib 100 mg plus topical therapy (95); or placebo, which consisted of topical therapy alone (95).
The primary endpoints were an Investigator’s Global Assessment response of clear or almost clear (scores of 0 and 1, respectively), with an improvement of at least 2 points, and an improvement in Eczema Area and Severity Index score of at least 75% at week 12.
Secondary endpoints included an improvement in Peak Pruritus Numerical Rating Scale (PP-NRS) response of at least 4 points at week 12.
The teens who received abrocitinib along with medicated topical therapy showed significant improvement in the severity of their AD at the end of the 12-week period, compared with those in the placebo group.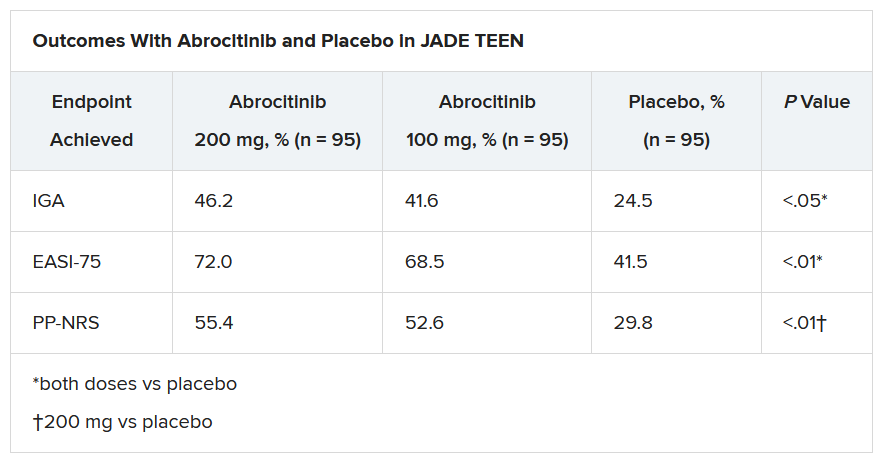
“The percentage of patients achieving essentially no itch, as captured in the fact that more than half of those on the higher dose of abrocitinib made it to no itch, is a new data point and is important to note,” Dr. Eichenfield said. “A lot of the other medicines don’t really get a significant percentage of the population to an itch score of 0 to 1. This drug brought about a rapid and profound itch relief.”
He added: “The results from JADE TEEN extend the drug’s utility in this younger population and show that abrocitinib performs the same with regard to efficacy and safety in the teenagers. Having atopic dermatitis that does not respond to treatment is especially hard for adolescents, but now we know that abrocitinib will be safe and effective and so we now have something to offer these kids.”
“Abrocitinib achieved a good response in this study that was statistically significant, compared to standard treatment,” Jonathan A. Bernstein, MD, professor of medicine at the University of Cincinnati, commented in an interview.
“JAK inhibitors are very promising, and this study adds to that promise. They play an important role in atopic dermatitis, so obviously, teenagers with AD represent an important population,” said Dr. Bernstein, who was not part of the study. “These results are very encouraging, and I think that we will probably see some of these JAK inhibitors approved by the FDA, if not this year, probably next.”
The study was sponsored by Pfizer. Dr. Eichenfield serves as an investigator, speaker, and consultant for Pfizer; and as an investigator, speaker, consultant, and/or is on a data safety monitoring board for AbbVie, Almirall, Amgen, Arcutis, Asana, Dermavant, Dermira, Forte, Galderma, Ichnos/Glenmark, Incyte, LEO, Lilly, L’Oreal, Novartis, Regeneron, Sanofi-Genzyme, and Verrica. Dr. Bernstein disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Abrocitinib, an investigational drug proven to be a safe and effective treatment for moderate to severe atopic dermatitis (AD) in adults 18 years and older, is also safe and effective in patients aged 12-17 years, according to a randomized trial of the oral, once-daily Janus kinase (JAK) 1 selective inhibitor, used in combination with medicated topical therapy.
The results, from the phase 3 JADE TEEN study, were presented during an oral abstract session at the annual meeting of the American Academy of Allergy, Asthma, and Immunology, held virtually this year.
“We’re very excited about the introduction of oral JAKs into our armamentarium for atopic dermatitis,” lead author Lawrence Eichenfield, MD, professor of dermatology and pediatrics, University of California, San Diego, and chief of pediatric and adolescent dermatology, Rady Children’s Hospital, also in San Diego, said in an interview.
AD ranges in severity, and there is a great deal of moderate to severe AD that has a tremendous negative impact on the individual, Dr. Eichenfield said. “Traditionally we have treated it with intermittent topical corticosteroids, but this has left a significant percentage of patients without long-term disease control.”
JAK inhibitors are effective mediators of the inflammation response that occurs in moderate to severe AD. They inhibit the stimulation of the JAK pathway and allow anti-inflammatory effects and therefore have potential, especially in more severe disease, Dr. Eichenfield said.
In the current study, which is a spin-off of the original study that looked at abrocitinib in adults, he and his team randomly assigned 285 teens (mean age, 14.9 years; 50.9% male; 56.1% White) with moderate to severe AD to receive one of the following treatments for 12 weeks: abrocitinib 200 mg plus topical therapy (95); abrocitinib 100 mg plus topical therapy (95); or placebo, which consisted of topical therapy alone (95).
The primary endpoints were an Investigator’s Global Assessment response of clear or almost clear (scores of 0 and 1, respectively), with an improvement of at least 2 points, and an improvement in Eczema Area and Severity Index score of at least 75% at week 12.
Secondary endpoints included an improvement in Peak Pruritus Numerical Rating Scale (PP-NRS) response of at least 4 points at week 12.
The teens who received abrocitinib along with medicated topical therapy showed significant improvement in the severity of their AD at the end of the 12-week period, compared with those in the placebo group.
“The percentage of patients achieving essentially no itch, as captured in the fact that more than half of those on the higher dose of abrocitinib made it to no itch, is a new data point and is important to note,” Dr. Eichenfield said. “A lot of the other medicines don’t really get a significant percentage of the population to an itch score of 0 to 1. This drug brought about a rapid and profound itch relief.”
He added: “The results from JADE TEEN extend the drug’s utility in this younger population and show that abrocitinib performs the same with regard to efficacy and safety in the teenagers. Having atopic dermatitis that does not respond to treatment is especially hard for adolescents, but now we know that abrocitinib will be safe and effective and so we now have something to offer these kids.”
“Abrocitinib achieved a good response in this study that was statistically significant, compared to standard treatment,” Jonathan A. Bernstein, MD, professor of medicine at the University of Cincinnati, commented in an interview.
“JAK inhibitors are very promising, and this study adds to that promise. They play an important role in atopic dermatitis, so obviously, teenagers with AD represent an important population,” said Dr. Bernstein, who was not part of the study. “These results are very encouraging, and I think that we will probably see some of these JAK inhibitors approved by the FDA, if not this year, probably next.”
The study was sponsored by Pfizer. Dr. Eichenfield serves as an investigator, speaker, and consultant for Pfizer; and as an investigator, speaker, consultant, and/or is on a data safety monitoring board for AbbVie, Almirall, Amgen, Arcutis, Asana, Dermavant, Dermira, Forte, Galderma, Ichnos/Glenmark, Incyte, LEO, Lilly, L’Oreal, Novartis, Regeneron, Sanofi-Genzyme, and Verrica. Dr. Bernstein disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Abrocitinib, an investigational drug proven to be a safe and effective treatment for moderate to severe atopic dermatitis (AD) in adults 18 years and older, is also safe and effective in patients aged 12-17 years, according to a randomized trial of the oral, once-daily Janus kinase (JAK) 1 selective inhibitor, used in combination with medicated topical therapy.
The results, from the phase 3 JADE TEEN study, were presented during an oral abstract session at the annual meeting of the American Academy of Allergy, Asthma, and Immunology, held virtually this year.
“We’re very excited about the introduction of oral JAKs into our armamentarium for atopic dermatitis,” lead author Lawrence Eichenfield, MD, professor of dermatology and pediatrics, University of California, San Diego, and chief of pediatric and adolescent dermatology, Rady Children’s Hospital, also in San Diego, said in an interview.
AD ranges in severity, and there is a great deal of moderate to severe AD that has a tremendous negative impact on the individual, Dr. Eichenfield said. “Traditionally we have treated it with intermittent topical corticosteroids, but this has left a significant percentage of patients without long-term disease control.”
JAK inhibitors are effective mediators of the inflammation response that occurs in moderate to severe AD. They inhibit the stimulation of the JAK pathway and allow anti-inflammatory effects and therefore have potential, especially in more severe disease, Dr. Eichenfield said.
In the current study, which is a spin-off of the original study that looked at abrocitinib in adults, he and his team randomly assigned 285 teens (mean age, 14.9 years; 50.9% male; 56.1% White) with moderate to severe AD to receive one of the following treatments for 12 weeks: abrocitinib 200 mg plus topical therapy (95); abrocitinib 100 mg plus topical therapy (95); or placebo, which consisted of topical therapy alone (95).
The primary endpoints were an Investigator’s Global Assessment response of clear or almost clear (scores of 0 and 1, respectively), with an improvement of at least 2 points, and an improvement in Eczema Area and Severity Index score of at least 75% at week 12.
Secondary endpoints included an improvement in Peak Pruritus Numerical Rating Scale (PP-NRS) response of at least 4 points at week 12.
The teens who received abrocitinib along with medicated topical therapy showed significant improvement in the severity of their AD at the end of the 12-week period, compared with those in the placebo group.
“The percentage of patients achieving essentially no itch, as captured in the fact that more than half of those on the higher dose of abrocitinib made it to no itch, is a new data point and is important to note,” Dr. Eichenfield said. “A lot of the other medicines don’t really get a significant percentage of the population to an itch score of 0 to 1. This drug brought about a rapid and profound itch relief.”
He added: “The results from JADE TEEN extend the drug’s utility in this younger population and show that abrocitinib performs the same with regard to efficacy and safety in the teenagers. Having atopic dermatitis that does not respond to treatment is especially hard for adolescents, but now we know that abrocitinib will be safe and effective and so we now have something to offer these kids.”
“Abrocitinib achieved a good response in this study that was statistically significant, compared to standard treatment,” Jonathan A. Bernstein, MD, professor of medicine at the University of Cincinnati, commented in an interview.
“JAK inhibitors are very promising, and this study adds to that promise. They play an important role in atopic dermatitis, so obviously, teenagers with AD represent an important population,” said Dr. Bernstein, who was not part of the study. “These results are very encouraging, and I think that we will probably see some of these JAK inhibitors approved by the FDA, if not this year, probably next.”
The study was sponsored by Pfizer. Dr. Eichenfield serves as an investigator, speaker, and consultant for Pfizer; and as an investigator, speaker, consultant, and/or is on a data safety monitoring board for AbbVie, Almirall, Amgen, Arcutis, Asana, Dermavant, Dermira, Forte, Galderma, Ichnos/Glenmark, Incyte, LEO, Lilly, L’Oreal, Novartis, Regeneron, Sanofi-Genzyme, and Verrica. Dr. Bernstein disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Severe atopic dermatitis often puts a dent in quality of life
In his role as head of the division of pediatric behavioral health at National Jewish Health, Denver, Bruce G. Bender, PhD, helps children and adults navigate the adverse effects of severe atopic dermatitis (AD) on their quality of life.
“There have been many surveys of adults with AD who report impairment of their sleep, reduced activity level, increased work absence, financial burden, emotional distress, and social avoidance,” he said at the Revolutionizing Atopic Dermatitis virtual symposium. “Similarly, children with AD or their parents report emotional distress, reduced activity, and increased school absence, social avoidance, and sleep disturbance. Families report financial burdens, conflict, particularly among the adults, social avoidance, sleep disturbance in the parents, and reduction of well-being in the siblings.”
In an effort to objectively measure sleep change in this population, Dr. Bender and colleagues recruited 14 adults with AD and 14 healthy controls who wore an ActiGraph for 1 week and completed questionnaires about sleep, itch, and quality of life. Patients with AD were awake almost twice as many minutes each night as the healthy controls (a mean of 57.3 vs. 32.3 minutes, respectively; P = .0480). Consequently, their sleep efficiency was significantly reduced based on the Pittsburgh sleep quality index (a mean of 90.6 vs. 95; P = .0305).
In another study, Dr. Bender and colleagues enrolled 20 adults with AD who underwent 2 nights of polysomnography and actigraphy. The lab was set up to measure a scratching event, which was recorded when a burst of electromyographic activity of at least 3 seconds was accompanied by a visible scratching motion. “We learned that sleep efficiency as measured by both PSG and actigraphy correlated with total body surface area and scratching index,” he said. “As we might assume, the more skin involved, the more patients scratch, the less well they sleep.”
Behavioral, neurocognitive effects
In a separate study of AD, sleep, and behavior, the researchers studied 1,041 children with asthma who were enrolled in the Childhood Asthma Management Program at eight North American sites. They used baseline parent ratings on standardized sleep and behavior rating scales and found that increased awakenings were associated with increased school absence and daytime behavior problems. “So, not only do children with AD sleep less well, but this shows up to impair their functioning during the day,” said Dr. Bender, professor of psychiatry at the University of Colorado, Denver.
In a report from Australia, researchers set out to explore the association between sleep and neurocognitive function in 21 children with eczema and 20 healthy controls. Participants underwent cognitive testing and polysomnography. The authors found that the children with eczema demonstrated lower test scores. Reduced scores were correlated with parental reports of sleep problems but not polysomnography.
In a much larger study funded by the Agency for Healthcare Research and Quality, investigators analyzed data on 354,416 children and 34,613 adults from 19 U.S. population surveys including the National Health Interview Survey 1997-2013 and the National Survey of Children’s Health 2003/4 and 2007/8. They found that AD was associated with ADHD in children (adjusted odds ratio, 1.14) and adults (aOR, 1.61). Higher odds of ADHD were found in children who had significant sleep disturbance (aOR, 16.83) and other allergic disease and asthma (aOR, 1.61).
“All of these findings show that AD can impact quality of life, especially sleep, with the result of poorer daytime functioning,” Dr. Bender said. “But those studies don’t answer this question: Are patients with AD at increased risk for psychological disorders such as depression and anxiety?”
Impact on depression, anxiety
Two systematic reviews on the topic suggest that patients with AD are twice as likely to experience depression. One was published in 2018 and the other in 2019. The 2018 review reported a little more than a twofold increase (OR, 2.19), the 2019 review a little bit less (OR, 1.71).
“At the more severe end of the depression continuum, we sometimes see suicidal ideation and suicide attempts,” Dr. Bender said. “A number of studies have asked whether these are increased in patients with AD. Quite a few studies collectively show an increased incidence of suicidal ideation. The question of suicide attempts is reflected in fewer studies. And while the result is small, it is significant. There is a significant increase reported of suicide attempts in AD patients.”
The 2018 review also found an increased incidence of anxiety in AD patients: a little more than twofold in adults (OR, 2.19) and a little less than twofold in children (OR, 1.81).
“It’s a two-way relationship between AD and psychological factors,” Dr. Bender said. “We generally think about AD – the stress that it brings, the burden that it puts on children, adults, and families. But it can work the other way around,” he said, referring to patients who have psychological problems, experience a great deal of stress, have trouble being adherent to their treatment regimen, and find it difficult to resist scratching. “The behavioral/psychological characteristics of the patient also drive the AD. It is well established that acute and chronic stress can result in a worsening of skin conditions in AD patients.”
Behavioral health interventions that have been described in the literature include cognitive therapy, stress management, biofeedback, hypnotherapy, relaxation training, mindfulness, habit reversal, and patient education – some of which have been tested in randomized trials. “All of them report a decrease in scratching as a consequence of the behavioral intervention,” Dr. Bender said.
“Other studies have been reported that look at the impact of behavioral interventions on the severity of the skin condition. Most report an improvement in the skin condition from these behavioral interventions but it’s not a perfect literature.” Critiques of these studies include the fact that there is often not enough detail about the intervention or the framework for the intervention that would allow a clinician to test an intervention in another study or actually pull that intervention into clinical practice (Cochrane Database Syst Rev. 2014 Jan 7;2014[1]:CD004054), (Int Arch Allergy Immunol.2007;144[1]:1-9).
“Some of the studies lack rigorous designs, some have sampling bias, and some have inadequate outcome measurements,” he said. “We really need additional, high-quality studies to look at what is helpful for patients with AD.”
Dr. Bender reported having no financial disclosures.
In his role as head of the division of pediatric behavioral health at National Jewish Health, Denver, Bruce G. Bender, PhD, helps children and adults navigate the adverse effects of severe atopic dermatitis (AD) on their quality of life.
“There have been many surveys of adults with AD who report impairment of their sleep, reduced activity level, increased work absence, financial burden, emotional distress, and social avoidance,” he said at the Revolutionizing Atopic Dermatitis virtual symposium. “Similarly, children with AD or their parents report emotional distress, reduced activity, and increased school absence, social avoidance, and sleep disturbance. Families report financial burdens, conflict, particularly among the adults, social avoidance, sleep disturbance in the parents, and reduction of well-being in the siblings.”
In an effort to objectively measure sleep change in this population, Dr. Bender and colleagues recruited 14 adults with AD and 14 healthy controls who wore an ActiGraph for 1 week and completed questionnaires about sleep, itch, and quality of life. Patients with AD were awake almost twice as many minutes each night as the healthy controls (a mean of 57.3 vs. 32.3 minutes, respectively; P = .0480). Consequently, their sleep efficiency was significantly reduced based on the Pittsburgh sleep quality index (a mean of 90.6 vs. 95; P = .0305).
In another study, Dr. Bender and colleagues enrolled 20 adults with AD who underwent 2 nights of polysomnography and actigraphy. The lab was set up to measure a scratching event, which was recorded when a burst of electromyographic activity of at least 3 seconds was accompanied by a visible scratching motion. “We learned that sleep efficiency as measured by both PSG and actigraphy correlated with total body surface area and scratching index,” he said. “As we might assume, the more skin involved, the more patients scratch, the less well they sleep.”
Behavioral, neurocognitive effects
In a separate study of AD, sleep, and behavior, the researchers studied 1,041 children with asthma who were enrolled in the Childhood Asthma Management Program at eight North American sites. They used baseline parent ratings on standardized sleep and behavior rating scales and found that increased awakenings were associated with increased school absence and daytime behavior problems. “So, not only do children with AD sleep less well, but this shows up to impair their functioning during the day,” said Dr. Bender, professor of psychiatry at the University of Colorado, Denver.
In a report from Australia, researchers set out to explore the association between sleep and neurocognitive function in 21 children with eczema and 20 healthy controls. Participants underwent cognitive testing and polysomnography. The authors found that the children with eczema demonstrated lower test scores. Reduced scores were correlated with parental reports of sleep problems but not polysomnography.
In a much larger study funded by the Agency for Healthcare Research and Quality, investigators analyzed data on 354,416 children and 34,613 adults from 19 U.S. population surveys including the National Health Interview Survey 1997-2013 and the National Survey of Children’s Health 2003/4 and 2007/8. They found that AD was associated with ADHD in children (adjusted odds ratio, 1.14) and adults (aOR, 1.61). Higher odds of ADHD were found in children who had significant sleep disturbance (aOR, 16.83) and other allergic disease and asthma (aOR, 1.61).
“All of these findings show that AD can impact quality of life, especially sleep, with the result of poorer daytime functioning,” Dr. Bender said. “But those studies don’t answer this question: Are patients with AD at increased risk for psychological disorders such as depression and anxiety?”
Impact on depression, anxiety
Two systematic reviews on the topic suggest that patients with AD are twice as likely to experience depression. One was published in 2018 and the other in 2019. The 2018 review reported a little more than a twofold increase (OR, 2.19), the 2019 review a little bit less (OR, 1.71).
“At the more severe end of the depression continuum, we sometimes see suicidal ideation and suicide attempts,” Dr. Bender said. “A number of studies have asked whether these are increased in patients with AD. Quite a few studies collectively show an increased incidence of suicidal ideation. The question of suicide attempts is reflected in fewer studies. And while the result is small, it is significant. There is a significant increase reported of suicide attempts in AD patients.”
The 2018 review also found an increased incidence of anxiety in AD patients: a little more than twofold in adults (OR, 2.19) and a little less than twofold in children (OR, 1.81).
“It’s a two-way relationship between AD and psychological factors,” Dr. Bender said. “We generally think about AD – the stress that it brings, the burden that it puts on children, adults, and families. But it can work the other way around,” he said, referring to patients who have psychological problems, experience a great deal of stress, have trouble being adherent to their treatment regimen, and find it difficult to resist scratching. “The behavioral/psychological characteristics of the patient also drive the AD. It is well established that acute and chronic stress can result in a worsening of skin conditions in AD patients.”
Behavioral health interventions that have been described in the literature include cognitive therapy, stress management, biofeedback, hypnotherapy, relaxation training, mindfulness, habit reversal, and patient education – some of which have been tested in randomized trials. “All of them report a decrease in scratching as a consequence of the behavioral intervention,” Dr. Bender said.
“Other studies have been reported that look at the impact of behavioral interventions on the severity of the skin condition. Most report an improvement in the skin condition from these behavioral interventions but it’s not a perfect literature.” Critiques of these studies include the fact that there is often not enough detail about the intervention or the framework for the intervention that would allow a clinician to test an intervention in another study or actually pull that intervention into clinical practice (Cochrane Database Syst Rev. 2014 Jan 7;2014[1]:CD004054), (Int Arch Allergy Immunol.2007;144[1]:1-9).
“Some of the studies lack rigorous designs, some have sampling bias, and some have inadequate outcome measurements,” he said. “We really need additional, high-quality studies to look at what is helpful for patients with AD.”
Dr. Bender reported having no financial disclosures.
In his role as head of the division of pediatric behavioral health at National Jewish Health, Denver, Bruce G. Bender, PhD, helps children and adults navigate the adverse effects of severe atopic dermatitis (AD) on their quality of life.
“There have been many surveys of adults with AD who report impairment of their sleep, reduced activity level, increased work absence, financial burden, emotional distress, and social avoidance,” he said at the Revolutionizing Atopic Dermatitis virtual symposium. “Similarly, children with AD or their parents report emotional distress, reduced activity, and increased school absence, social avoidance, and sleep disturbance. Families report financial burdens, conflict, particularly among the adults, social avoidance, sleep disturbance in the parents, and reduction of well-being in the siblings.”
In an effort to objectively measure sleep change in this population, Dr. Bender and colleagues recruited 14 adults with AD and 14 healthy controls who wore an ActiGraph for 1 week and completed questionnaires about sleep, itch, and quality of life. Patients with AD were awake almost twice as many minutes each night as the healthy controls (a mean of 57.3 vs. 32.3 minutes, respectively; P = .0480). Consequently, their sleep efficiency was significantly reduced based on the Pittsburgh sleep quality index (a mean of 90.6 vs. 95; P = .0305).
In another study, Dr. Bender and colleagues enrolled 20 adults with AD who underwent 2 nights of polysomnography and actigraphy. The lab was set up to measure a scratching event, which was recorded when a burst of electromyographic activity of at least 3 seconds was accompanied by a visible scratching motion. “We learned that sleep efficiency as measured by both PSG and actigraphy correlated with total body surface area and scratching index,” he said. “As we might assume, the more skin involved, the more patients scratch, the less well they sleep.”
Behavioral, neurocognitive effects
In a separate study of AD, sleep, and behavior, the researchers studied 1,041 children with asthma who were enrolled in the Childhood Asthma Management Program at eight North American sites. They used baseline parent ratings on standardized sleep and behavior rating scales and found that increased awakenings were associated with increased school absence and daytime behavior problems. “So, not only do children with AD sleep less well, but this shows up to impair their functioning during the day,” said Dr. Bender, professor of psychiatry at the University of Colorado, Denver.
In a report from Australia, researchers set out to explore the association between sleep and neurocognitive function in 21 children with eczema and 20 healthy controls. Participants underwent cognitive testing and polysomnography. The authors found that the children with eczema demonstrated lower test scores. Reduced scores were correlated with parental reports of sleep problems but not polysomnography.
In a much larger study funded by the Agency for Healthcare Research and Quality, investigators analyzed data on 354,416 children and 34,613 adults from 19 U.S. population surveys including the National Health Interview Survey 1997-2013 and the National Survey of Children’s Health 2003/4 and 2007/8. They found that AD was associated with ADHD in children (adjusted odds ratio, 1.14) and adults (aOR, 1.61). Higher odds of ADHD were found in children who had significant sleep disturbance (aOR, 16.83) and other allergic disease and asthma (aOR, 1.61).
“All of these findings show that AD can impact quality of life, especially sleep, with the result of poorer daytime functioning,” Dr. Bender said. “But those studies don’t answer this question: Are patients with AD at increased risk for psychological disorders such as depression and anxiety?”
Impact on depression, anxiety
Two systematic reviews on the topic suggest that patients with AD are twice as likely to experience depression. One was published in 2018 and the other in 2019. The 2018 review reported a little more than a twofold increase (OR, 2.19), the 2019 review a little bit less (OR, 1.71).
“At the more severe end of the depression continuum, we sometimes see suicidal ideation and suicide attempts,” Dr. Bender said. “A number of studies have asked whether these are increased in patients with AD. Quite a few studies collectively show an increased incidence of suicidal ideation. The question of suicide attempts is reflected in fewer studies. And while the result is small, it is significant. There is a significant increase reported of suicide attempts in AD patients.”
The 2018 review also found an increased incidence of anxiety in AD patients: a little more than twofold in adults (OR, 2.19) and a little less than twofold in children (OR, 1.81).
“It’s a two-way relationship between AD and psychological factors,” Dr. Bender said. “We generally think about AD – the stress that it brings, the burden that it puts on children, adults, and families. But it can work the other way around,” he said, referring to patients who have psychological problems, experience a great deal of stress, have trouble being adherent to their treatment regimen, and find it difficult to resist scratching. “The behavioral/psychological characteristics of the patient also drive the AD. It is well established that acute and chronic stress can result in a worsening of skin conditions in AD patients.”
Behavioral health interventions that have been described in the literature include cognitive therapy, stress management, biofeedback, hypnotherapy, relaxation training, mindfulness, habit reversal, and patient education – some of which have been tested in randomized trials. “All of them report a decrease in scratching as a consequence of the behavioral intervention,” Dr. Bender said.
“Other studies have been reported that look at the impact of behavioral interventions on the severity of the skin condition. Most report an improvement in the skin condition from these behavioral interventions but it’s not a perfect literature.” Critiques of these studies include the fact that there is often not enough detail about the intervention or the framework for the intervention that would allow a clinician to test an intervention in another study or actually pull that intervention into clinical practice (Cochrane Database Syst Rev. 2014 Jan 7;2014[1]:CD004054), (Int Arch Allergy Immunol.2007;144[1]:1-9).
“Some of the studies lack rigorous designs, some have sampling bias, and some have inadequate outcome measurements,” he said. “We really need additional, high-quality studies to look at what is helpful for patients with AD.”
Dr. Bender reported having no financial disclosures.
FROM REVOLUTIONIZING AD 2020
Novel agent shows promise against cat allergy
One dose of the novel agent, REGN1908-1909 (Regeneron Pharmaceuticals) resulted in a rapid and durable reduction in cat-allergen-induced bronchoconstriction in cat-allergic subjects with mild asthma.
The finding, from a phase 2 randomized placebo-controlled study, is good news for the millions of people who are plagued by cat allergies, the investigators say.
The study, which was sponsored by Regeneron, was presented in a late breaking oral abstract session at the annual meeting of the American Academy of Allergy, Asthma, and Immunology, held virtually this year.
“REGN1908-1909 contains antibodies against Fel d 1, the major cat allergen, and here we show that it quickly and lastingly reduces acute bronchoconstriction in people with cat allergy,” lead author Frederic J. de Blay, MD, Strasbourg University Hospital, France, said in an interview.
Dr. de Blay admitted he is “quite excited” about the results.
“This study was performed in an environmental exposure chamber, and we clearly demonstrate that these antibodies decrease the asthmatic response to cat allergen within 8 days, and that these effects last 3 months. I never saw that in my life. I was a little bit skeptical at first, so to obtain such robust results after just 8 days, after just one injection, I was very surprised,” he said.
Dr. de Blay and his team screened potential participants to make sure they were cat allergic by exposing them to cat allergen for up to 2 hours while they were in the environmental exposure chamber. To be eligible for the study, participants had to show an early asthmatic response (EAR), defined as a reduction in forced expiratory volume in 1 second (FEV1) of at least 20% from baseline.
The participants were then randomized to receive either single-dose REGN1908-1909, 600 mg, subcutaneously (n = 29 patients) or placebo (n = 27 patients) prior to cat-allergen exposure in the controlled environmental chamber.
Dr. de Blay developed the chamber used in the study: the ALYATEC environmental exposure chamber.
“The chamber is 60 meters square, or 150 cubic meters, and can accommodate 20 patients. We are able to nebulize cat allergen, mice allergen, or whatever we wish to study so we can standardize the exposure. We can control the particle size and the amount so we know the exact amount of allergen that the patient has been exposed to,” he explained.
To test the efficacy of REGN1908-1909 in reducing acute bronchoconstriction, or EAR, the researchers measured FEV1 at baseline, and on days 8, 29, 57, and 85 in both groups. During each exposure, measurements were taken every 10 minutes for periods that lasted up to 4 hours.
They found that the probability of remaining in the chamber with no asthmatic response was substantially elevated in the group treated with REGN1908-1909.
Compared with placebo, REGN1908-1909 significantly increased the median time to EAR, from 51 minutes at baseline to more than 4 hours on day 8, (hazard ratio [HR], 0.36; P < .0083), day 29 (HR, 0.24; P < .0001), day 57 (HR, 0.45; P = .0222), and day 85 (HR, 0.27; P = .0003).
The FEV1 area under the curve (AUC) was also better with REGN1908-1909 than with placebo at day 8 (15.2% vs. 1.6%; P < .001). And a single dose reduced skin-test reactivity to cat allergen at 1 week, which persisted for up to 4 months.
In addition, participants who received REGN1908-1909 were able to tolerate a threefold higher amount of the cat allergen than those who received placebo (P = .003).
“We initially gave 40 nanograms of cat allergen, and then 8 days later they were able to stay longer in the chamber and inhale more of the allergen, to almost triple the amount they had originally been given. That 40 nanograms is very close to real world exposure,” Dr. de Blay noted.
Regeneron plans to start a phase 3 trial soon, he reported.
Promising results
“The study is well designed and shows a reduction in drop of FEV1 in response to cat allergen provocation and a decreased AUC in cat SPT response over 4 months,” Jonathan A. Bernstein, MD, professor of medicine at the University of Cincinnati, said in an interview.
“These are very promising results, which show that REGN1908-1909 can be a novel treatment for cat-induced asthma, which is often the only sensitization patients have. And they love their cats – one-third of the U.S. population has a cat and one-third has a dog, and 50% have both,” noted Dr. Bernstein, who was not involved with the study.
“This novel study used our scientific knowledge of the cat allergen itself to design a targeted antibody-based treatment that demonstrates significant benefit even after the first shot,” added Edwin H. Kim, MD, director of the UNC Food Allergy Initiative at the University of North Carolina at Chapel Hill.
“This strategy has the potential to revolutionize not only our treatment of common environmental allergies but also other allergic diseases with well-described triggers, such as food and drug allergy,” Dr. Kim, who was not part of the study, said in an interview.
Dr. de Blay reported a financial relationship with Regeneron Pharmaceuticals, which sponsored the study. Dr. Bernstein and Dr. Kim have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
One dose of the novel agent, REGN1908-1909 (Regeneron Pharmaceuticals) resulted in a rapid and durable reduction in cat-allergen-induced bronchoconstriction in cat-allergic subjects with mild asthma.
The finding, from a phase 2 randomized placebo-controlled study, is good news for the millions of people who are plagued by cat allergies, the investigators say.
The study, which was sponsored by Regeneron, was presented in a late breaking oral abstract session at the annual meeting of the American Academy of Allergy, Asthma, and Immunology, held virtually this year.
“REGN1908-1909 contains antibodies against Fel d 1, the major cat allergen, and here we show that it quickly and lastingly reduces acute bronchoconstriction in people with cat allergy,” lead author Frederic J. de Blay, MD, Strasbourg University Hospital, France, said in an interview.
Dr. de Blay admitted he is “quite excited” about the results.
“This study was performed in an environmental exposure chamber, and we clearly demonstrate that these antibodies decrease the asthmatic response to cat allergen within 8 days, and that these effects last 3 months. I never saw that in my life. I was a little bit skeptical at first, so to obtain such robust results after just 8 days, after just one injection, I was very surprised,” he said.
Dr. de Blay and his team screened potential participants to make sure they were cat allergic by exposing them to cat allergen for up to 2 hours while they were in the environmental exposure chamber. To be eligible for the study, participants had to show an early asthmatic response (EAR), defined as a reduction in forced expiratory volume in 1 second (FEV1) of at least 20% from baseline.
The participants were then randomized to receive either single-dose REGN1908-1909, 600 mg, subcutaneously (n = 29 patients) or placebo (n = 27 patients) prior to cat-allergen exposure in the controlled environmental chamber.
Dr. de Blay developed the chamber used in the study: the ALYATEC environmental exposure chamber.
“The chamber is 60 meters square, or 150 cubic meters, and can accommodate 20 patients. We are able to nebulize cat allergen, mice allergen, or whatever we wish to study so we can standardize the exposure. We can control the particle size and the amount so we know the exact amount of allergen that the patient has been exposed to,” he explained.
To test the efficacy of REGN1908-1909 in reducing acute bronchoconstriction, or EAR, the researchers measured FEV1 at baseline, and on days 8, 29, 57, and 85 in both groups. During each exposure, measurements were taken every 10 minutes for periods that lasted up to 4 hours.
They found that the probability of remaining in the chamber with no asthmatic response was substantially elevated in the group treated with REGN1908-1909.
Compared with placebo, REGN1908-1909 significantly increased the median time to EAR, from 51 minutes at baseline to more than 4 hours on day 8, (hazard ratio [HR], 0.36; P < .0083), day 29 (HR, 0.24; P < .0001), day 57 (HR, 0.45; P = .0222), and day 85 (HR, 0.27; P = .0003).
The FEV1 area under the curve (AUC) was also better with REGN1908-1909 than with placebo at day 8 (15.2% vs. 1.6%; P < .001). And a single dose reduced skin-test reactivity to cat allergen at 1 week, which persisted for up to 4 months.
In addition, participants who received REGN1908-1909 were able to tolerate a threefold higher amount of the cat allergen than those who received placebo (P = .003).
“We initially gave 40 nanograms of cat allergen, and then 8 days later they were able to stay longer in the chamber and inhale more of the allergen, to almost triple the amount they had originally been given. That 40 nanograms is very close to real world exposure,” Dr. de Blay noted.
Regeneron plans to start a phase 3 trial soon, he reported.
Promising results
“The study is well designed and shows a reduction in drop of FEV1 in response to cat allergen provocation and a decreased AUC in cat SPT response over 4 months,” Jonathan A. Bernstein, MD, professor of medicine at the University of Cincinnati, said in an interview.
“These are very promising results, which show that REGN1908-1909 can be a novel treatment for cat-induced asthma, which is often the only sensitization patients have. And they love their cats – one-third of the U.S. population has a cat and one-third has a dog, and 50% have both,” noted Dr. Bernstein, who was not involved with the study.
“This novel study used our scientific knowledge of the cat allergen itself to design a targeted antibody-based treatment that demonstrates significant benefit even after the first shot,” added Edwin H. Kim, MD, director of the UNC Food Allergy Initiative at the University of North Carolina at Chapel Hill.
“This strategy has the potential to revolutionize not only our treatment of common environmental allergies but also other allergic diseases with well-described triggers, such as food and drug allergy,” Dr. Kim, who was not part of the study, said in an interview.
Dr. de Blay reported a financial relationship with Regeneron Pharmaceuticals, which sponsored the study. Dr. Bernstein and Dr. Kim have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
One dose of the novel agent, REGN1908-1909 (Regeneron Pharmaceuticals) resulted in a rapid and durable reduction in cat-allergen-induced bronchoconstriction in cat-allergic subjects with mild asthma.
The finding, from a phase 2 randomized placebo-controlled study, is good news for the millions of people who are plagued by cat allergies, the investigators say.
The study, which was sponsored by Regeneron, was presented in a late breaking oral abstract session at the annual meeting of the American Academy of Allergy, Asthma, and Immunology, held virtually this year.
“REGN1908-1909 contains antibodies against Fel d 1, the major cat allergen, and here we show that it quickly and lastingly reduces acute bronchoconstriction in people with cat allergy,” lead author Frederic J. de Blay, MD, Strasbourg University Hospital, France, said in an interview.
Dr. de Blay admitted he is “quite excited” about the results.
“This study was performed in an environmental exposure chamber, and we clearly demonstrate that these antibodies decrease the asthmatic response to cat allergen within 8 days, and that these effects last 3 months. I never saw that in my life. I was a little bit skeptical at first, so to obtain such robust results after just 8 days, after just one injection, I was very surprised,” he said.
Dr. de Blay and his team screened potential participants to make sure they were cat allergic by exposing them to cat allergen for up to 2 hours while they were in the environmental exposure chamber. To be eligible for the study, participants had to show an early asthmatic response (EAR), defined as a reduction in forced expiratory volume in 1 second (FEV1) of at least 20% from baseline.
The participants were then randomized to receive either single-dose REGN1908-1909, 600 mg, subcutaneously (n = 29 patients) or placebo (n = 27 patients) prior to cat-allergen exposure in the controlled environmental chamber.
Dr. de Blay developed the chamber used in the study: the ALYATEC environmental exposure chamber.
“The chamber is 60 meters square, or 150 cubic meters, and can accommodate 20 patients. We are able to nebulize cat allergen, mice allergen, or whatever we wish to study so we can standardize the exposure. We can control the particle size and the amount so we know the exact amount of allergen that the patient has been exposed to,” he explained.
To test the efficacy of REGN1908-1909 in reducing acute bronchoconstriction, or EAR, the researchers measured FEV1 at baseline, and on days 8, 29, 57, and 85 in both groups. During each exposure, measurements were taken every 10 minutes for periods that lasted up to 4 hours.
They found that the probability of remaining in the chamber with no asthmatic response was substantially elevated in the group treated with REGN1908-1909.
Compared with placebo, REGN1908-1909 significantly increased the median time to EAR, from 51 minutes at baseline to more than 4 hours on day 8, (hazard ratio [HR], 0.36; P < .0083), day 29 (HR, 0.24; P < .0001), day 57 (HR, 0.45; P = .0222), and day 85 (HR, 0.27; P = .0003).
The FEV1 area under the curve (AUC) was also better with REGN1908-1909 than with placebo at day 8 (15.2% vs. 1.6%; P < .001). And a single dose reduced skin-test reactivity to cat allergen at 1 week, which persisted for up to 4 months.
In addition, participants who received REGN1908-1909 were able to tolerate a threefold higher amount of the cat allergen than those who received placebo (P = .003).
“We initially gave 40 nanograms of cat allergen, and then 8 days later they were able to stay longer in the chamber and inhale more of the allergen, to almost triple the amount they had originally been given. That 40 nanograms is very close to real world exposure,” Dr. de Blay noted.
Regeneron plans to start a phase 3 trial soon, he reported.
Promising results
“The study is well designed and shows a reduction in drop of FEV1 in response to cat allergen provocation and a decreased AUC in cat SPT response over 4 months,” Jonathan A. Bernstein, MD, professor of medicine at the University of Cincinnati, said in an interview.
“These are very promising results, which show that REGN1908-1909 can be a novel treatment for cat-induced asthma, which is often the only sensitization patients have. And they love their cats – one-third of the U.S. population has a cat and one-third has a dog, and 50% have both,” noted Dr. Bernstein, who was not involved with the study.
“This novel study used our scientific knowledge of the cat allergen itself to design a targeted antibody-based treatment that demonstrates significant benefit even after the first shot,” added Edwin H. Kim, MD, director of the UNC Food Allergy Initiative at the University of North Carolina at Chapel Hill.
“This strategy has the potential to revolutionize not only our treatment of common environmental allergies but also other allergic diseases with well-described triggers, such as food and drug allergy,” Dr. Kim, who was not part of the study, said in an interview.
Dr. de Blay reported a financial relationship with Regeneron Pharmaceuticals, which sponsored the study. Dr. Bernstein and Dr. Kim have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AAAAI
Mepolizumab reduced exacerbations in patients with asthma and atopy, depression comorbidities
, according to research from the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“Mepolizumab has clearly been shown to improve severe asthma control in many clinical trials, but atopy, obesity, and depression/anxiety affect patients with asthma at an increased rate,” Thomas B. Casale, MD, former AAAAI president and professor of medicine and pediatrics at the University of South Florida in Tampa, said in a presentation at the meeting. “Yet, few studies have examined whether asthma therapy with these comorbidities works.”
Dr. Casale and colleagues performed a retrospective analysis of patients in the United States from the MarketScan Commercial and Medicare Supplemental Database between November 2014 and December 2018 who had atopy, obesity, or depression/anxiety in addition to asthma and were receiving mepolizumab. Atopy in the study was defined as allergic rhinitis, anaphylaxis, atopic dermatitis, conjunctivitis, eosinophilic esophagitis, and food allergies. Patients were at least age 12 years, had at least one diagnosis for asthma, at least one diagnosis code for atopic disease, obesity, or depression/anxiety at baseline, and at least two administrations of mepolizumab within 180 days.
The researchers examined the number of exacerbations, oral corticosteroid (OCS) claims, and OCS bursts per year at 12-month follow-up, compared with baseline. They identified exacerbations by examining patients who had an emergency department or outpatient claim related to their asthma, and a claim for systemic corticosteroids made in the 4 days prior to or 5 days after a visit, or if their inpatient hospital admission contained a primary asthma diagnosis. Dr. Casale and colleagues measured OCS bursts as a pharmacy claim of at least 20 mg of prednisone per day for between 3 and 28 days plus a claim for an emergency department visit related to asthma in the 7 days prior or 6 days after the claim.
At baseline, patients across all groups were mean age 50.5-52.4 years with a Charleson Comorbidity Index score between 1.1 and 1.4, a majority were women (59.0%-72.0%) and nearly all were commercially insured (88.0%-90.0%). Patients who used biologics at baseline and/or used a biologic that wasn’t mepolizumab during the follow-up period were excluded.
Medication claims in the groups included inhaled corticosteroids (ICS) (36.8%-48.6%), ICS/long-acting beta-agonist (LABA) (60.2%-63.0%), LABA/ long-acting muscarinic antagonist (LAMA) (1.2%-3.5%), ICS/LABA/LAMA (21.2%-25.1%), short-acting beta-agonist (SABA) (83.2%-87.7%), LAMA alone (33.5%-42.1%), or leukotriene receptor antagonist (LTRA).
In the non–mutually exclusive group of patients with atopy (468 patients), 28.0% had comorbid obesity and 26.0% had comorbid depression/anxiety. For patients with obesity categorized in a non–mutually exclusive subgroup (171 patients), 79.0% had comorbid atopy and 32.0% had comorbid depression/anxiety. Among patients with non–mutually exclusive depression/anxiety (173 patients), 70.0% had comorbid atopy, while 32.0% had comorbid obesity.
The results showed the mean number of overall exacerbations decreased by 48% at 12 months in the atopic group (2.3 vs. 1.2; P < .001), 52% in the group with obesity (2.5 vs. 1.2; P < .001), and 38% in the depression/anxiety group (2.4 vs. 1.5; P < .001). The mean number of exacerbations leading to hospitalizations decreased by 64% in the atopic group (0.11 vs. 0.04; P < .001), 65% in the group with obesity (0.20 vs. 0.07; P < .001), and 68% in the group with depression/anxiety (0.22 vs. 0.07; P < .001).
The researchers also found the mean number of OCS claims and OCS bursts also significantly decreased over the 12-month follow-up period. Mean OCS claims decreased by 33% for patients in the atopic group (5.5 vs. 3.7; P < .001), by 38% in the group with obesity (6.1 vs. 3.8; P < .001), and by 31% in the group with depression/anxiety (6.2 vs. 4.3; P < .001).
The mean number of OCS bursts also significantly decreased by 40% in the atopic group (2.0 vs. 2.1; P < .001), 48% in the group with obesity (2.3 vs. 1.2; P < .001), and by 37% in the group with depression/anxiety (1.9 vs. 1.2; P < .001). In total, 69% of patients with comorbid atopy, 70.8% of patients with comorbid obesity, and 68.2% of patients with comorbid depression/anxiety experienced a mean decrease in their OCS dose over 12 months.
“These data demonstrate that patients with asthma and atopy, obesity, or depression and anxiety have significantly fewer exacerbations and reduced OCS use in a real-world setting with treatment of mepolizumab,” Dr. Casale said. “Thus, holistic patient care for severe asthma is critical, and mepolizumab provides tangible clinical benefit despite the complexities of medical comorbidities.”
This study was funded by GlaxoSmithKline, and the company also funded graphic design support of the poster. Dr. Casale reports he has received research funds from GlaxoSmithKline. Four authors report being current or former GlaxoSmithKline employees; three authors report holding stock and/or shares of GlaxoSmithKline. Three authors are IBM Watson Health employees, a company GlaxoSmithKline has provided research funding.
, according to research from the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“Mepolizumab has clearly been shown to improve severe asthma control in many clinical trials, but atopy, obesity, and depression/anxiety affect patients with asthma at an increased rate,” Thomas B. Casale, MD, former AAAAI president and professor of medicine and pediatrics at the University of South Florida in Tampa, said in a presentation at the meeting. “Yet, few studies have examined whether asthma therapy with these comorbidities works.”
Dr. Casale and colleagues performed a retrospective analysis of patients in the United States from the MarketScan Commercial and Medicare Supplemental Database between November 2014 and December 2018 who had atopy, obesity, or depression/anxiety in addition to asthma and were receiving mepolizumab. Atopy in the study was defined as allergic rhinitis, anaphylaxis, atopic dermatitis, conjunctivitis, eosinophilic esophagitis, and food allergies. Patients were at least age 12 years, had at least one diagnosis for asthma, at least one diagnosis code for atopic disease, obesity, or depression/anxiety at baseline, and at least two administrations of mepolizumab within 180 days.
The researchers examined the number of exacerbations, oral corticosteroid (OCS) claims, and OCS bursts per year at 12-month follow-up, compared with baseline. They identified exacerbations by examining patients who had an emergency department or outpatient claim related to their asthma, and a claim for systemic corticosteroids made in the 4 days prior to or 5 days after a visit, or if their inpatient hospital admission contained a primary asthma diagnosis. Dr. Casale and colleagues measured OCS bursts as a pharmacy claim of at least 20 mg of prednisone per day for between 3 and 28 days plus a claim for an emergency department visit related to asthma in the 7 days prior or 6 days after the claim.
At baseline, patients across all groups were mean age 50.5-52.4 years with a Charleson Comorbidity Index score between 1.1 and 1.4, a majority were women (59.0%-72.0%) and nearly all were commercially insured (88.0%-90.0%). Patients who used biologics at baseline and/or used a biologic that wasn’t mepolizumab during the follow-up period were excluded.
Medication claims in the groups included inhaled corticosteroids (ICS) (36.8%-48.6%), ICS/long-acting beta-agonist (LABA) (60.2%-63.0%), LABA/ long-acting muscarinic antagonist (LAMA) (1.2%-3.5%), ICS/LABA/LAMA (21.2%-25.1%), short-acting beta-agonist (SABA) (83.2%-87.7%), LAMA alone (33.5%-42.1%), or leukotriene receptor antagonist (LTRA).
In the non–mutually exclusive group of patients with atopy (468 patients), 28.0% had comorbid obesity and 26.0% had comorbid depression/anxiety. For patients with obesity categorized in a non–mutually exclusive subgroup (171 patients), 79.0% had comorbid atopy and 32.0% had comorbid depression/anxiety. Among patients with non–mutually exclusive depression/anxiety (173 patients), 70.0% had comorbid atopy, while 32.0% had comorbid obesity.
The results showed the mean number of overall exacerbations decreased by 48% at 12 months in the atopic group (2.3 vs. 1.2; P < .001), 52% in the group with obesity (2.5 vs. 1.2; P < .001), and 38% in the depression/anxiety group (2.4 vs. 1.5; P < .001). The mean number of exacerbations leading to hospitalizations decreased by 64% in the atopic group (0.11 vs. 0.04; P < .001), 65% in the group with obesity (0.20 vs. 0.07; P < .001), and 68% in the group with depression/anxiety (0.22 vs. 0.07; P < .001).
The researchers also found the mean number of OCS claims and OCS bursts also significantly decreased over the 12-month follow-up period. Mean OCS claims decreased by 33% for patients in the atopic group (5.5 vs. 3.7; P < .001), by 38% in the group with obesity (6.1 vs. 3.8; P < .001), and by 31% in the group with depression/anxiety (6.2 vs. 4.3; P < .001).
The mean number of OCS bursts also significantly decreased by 40% in the atopic group (2.0 vs. 2.1; P < .001), 48% in the group with obesity (2.3 vs. 1.2; P < .001), and by 37% in the group with depression/anxiety (1.9 vs. 1.2; P < .001). In total, 69% of patients with comorbid atopy, 70.8% of patients with comorbid obesity, and 68.2% of patients with comorbid depression/anxiety experienced a mean decrease in their OCS dose over 12 months.
“These data demonstrate that patients with asthma and atopy, obesity, or depression and anxiety have significantly fewer exacerbations and reduced OCS use in a real-world setting with treatment of mepolizumab,” Dr. Casale said. “Thus, holistic patient care for severe asthma is critical, and mepolizumab provides tangible clinical benefit despite the complexities of medical comorbidities.”
This study was funded by GlaxoSmithKline, and the company also funded graphic design support of the poster. Dr. Casale reports he has received research funds from GlaxoSmithKline. Four authors report being current or former GlaxoSmithKline employees; three authors report holding stock and/or shares of GlaxoSmithKline. Three authors are IBM Watson Health employees, a company GlaxoSmithKline has provided research funding.
, according to research from the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“Mepolizumab has clearly been shown to improve severe asthma control in many clinical trials, but atopy, obesity, and depression/anxiety affect patients with asthma at an increased rate,” Thomas B. Casale, MD, former AAAAI president and professor of medicine and pediatrics at the University of South Florida in Tampa, said in a presentation at the meeting. “Yet, few studies have examined whether asthma therapy with these comorbidities works.”
Dr. Casale and colleagues performed a retrospective analysis of patients in the United States from the MarketScan Commercial and Medicare Supplemental Database between November 2014 and December 2018 who had atopy, obesity, or depression/anxiety in addition to asthma and were receiving mepolizumab. Atopy in the study was defined as allergic rhinitis, anaphylaxis, atopic dermatitis, conjunctivitis, eosinophilic esophagitis, and food allergies. Patients were at least age 12 years, had at least one diagnosis for asthma, at least one diagnosis code for atopic disease, obesity, or depression/anxiety at baseline, and at least two administrations of mepolizumab within 180 days.
The researchers examined the number of exacerbations, oral corticosteroid (OCS) claims, and OCS bursts per year at 12-month follow-up, compared with baseline. They identified exacerbations by examining patients who had an emergency department or outpatient claim related to their asthma, and a claim for systemic corticosteroids made in the 4 days prior to or 5 days after a visit, or if their inpatient hospital admission contained a primary asthma diagnosis. Dr. Casale and colleagues measured OCS bursts as a pharmacy claim of at least 20 mg of prednisone per day for between 3 and 28 days plus a claim for an emergency department visit related to asthma in the 7 days prior or 6 days after the claim.
At baseline, patients across all groups were mean age 50.5-52.4 years with a Charleson Comorbidity Index score between 1.1 and 1.4, a majority were women (59.0%-72.0%) and nearly all were commercially insured (88.0%-90.0%). Patients who used biologics at baseline and/or used a biologic that wasn’t mepolizumab during the follow-up period were excluded.
Medication claims in the groups included inhaled corticosteroids (ICS) (36.8%-48.6%), ICS/long-acting beta-agonist (LABA) (60.2%-63.0%), LABA/ long-acting muscarinic antagonist (LAMA) (1.2%-3.5%), ICS/LABA/LAMA (21.2%-25.1%), short-acting beta-agonist (SABA) (83.2%-87.7%), LAMA alone (33.5%-42.1%), or leukotriene receptor antagonist (LTRA).
In the non–mutually exclusive group of patients with atopy (468 patients), 28.0% had comorbid obesity and 26.0% had comorbid depression/anxiety. For patients with obesity categorized in a non–mutually exclusive subgroup (171 patients), 79.0% had comorbid atopy and 32.0% had comorbid depression/anxiety. Among patients with non–mutually exclusive depression/anxiety (173 patients), 70.0% had comorbid atopy, while 32.0% had comorbid obesity.
The results showed the mean number of overall exacerbations decreased by 48% at 12 months in the atopic group (2.3 vs. 1.2; P < .001), 52% in the group with obesity (2.5 vs. 1.2; P < .001), and 38% in the depression/anxiety group (2.4 vs. 1.5; P < .001). The mean number of exacerbations leading to hospitalizations decreased by 64% in the atopic group (0.11 vs. 0.04; P < .001), 65% in the group with obesity (0.20 vs. 0.07; P < .001), and 68% in the group with depression/anxiety (0.22 vs. 0.07; P < .001).
The researchers also found the mean number of OCS claims and OCS bursts also significantly decreased over the 12-month follow-up period. Mean OCS claims decreased by 33% for patients in the atopic group (5.5 vs. 3.7; P < .001), by 38% in the group with obesity (6.1 vs. 3.8; P < .001), and by 31% in the group with depression/anxiety (6.2 vs. 4.3; P < .001).
The mean number of OCS bursts also significantly decreased by 40% in the atopic group (2.0 vs. 2.1; P < .001), 48% in the group with obesity (2.3 vs. 1.2; P < .001), and by 37% in the group with depression/anxiety (1.9 vs. 1.2; P < .001). In total, 69% of patients with comorbid atopy, 70.8% of patients with comorbid obesity, and 68.2% of patients with comorbid depression/anxiety experienced a mean decrease in their OCS dose over 12 months.
“These data demonstrate that patients with asthma and atopy, obesity, or depression and anxiety have significantly fewer exacerbations and reduced OCS use in a real-world setting with treatment of mepolizumab,” Dr. Casale said. “Thus, holistic patient care for severe asthma is critical, and mepolizumab provides tangible clinical benefit despite the complexities of medical comorbidities.”
This study was funded by GlaxoSmithKline, and the company also funded graphic design support of the poster. Dr. Casale reports he has received research funds from GlaxoSmithKline. Four authors report being current or former GlaxoSmithKline employees; three authors report holding stock and/or shares of GlaxoSmithKline. Three authors are IBM Watson Health employees, a company GlaxoSmithKline has provided research funding.
FROM AAAAI 2021
Peanut sublingual immunotherapy feasible and effective in toddlers
Sublingual immunotherapy for the treatment of peanut allergy is safe and effective, even in children as young as age 1 year.
In a double-blind, placebo-controlled, food challenge (DBPCFC) of some 36 peanut-allergic children (mean age 2.2 years, range 1-4 years), those who were randomly assigned to receive peanut sublingual immunotherapy (PNSLIT) showed significant desensitization compared with those who received placebo.
In addition, there was a “strong potential” for sustained unresponsiveness at 3 months for the toddlers who received the active treatment.
The findings were presented in a late breaking oral abstract session at the 2021 American Academy of Allergy, Asthma & Immunology virtual annual meeting (Abstract L2).
“A year ago, the Food and Drug Administration approved the oral agent Palforzia (peanut allergen powder) for the treatment of peanut allergy in children 4 and older, and it is a great option, but I think what we have learned over time is that this approach is not for everybody,” Edwin H. Kim, MD, director of the UNC Food Allergy Initiative, University of North Carolina at Chapel Hill, said in an interview.
Palforzia is a powder that is mixed in food like yogurt or pudding which the child then eats daily, according to a rigorous schedule. But Palforzia treatment presents some difficulties.
“Palforzia requires getting the powder dose, mixing it with food, like pudding or apple sauce, then eating it, which can take up to 30 minutes depending on age and kids’ cooperation. It tastes and smells like peanut which can cause aversion. Kids have to refrain from exercise or strenuous activity for at least 30 minutes before and after dosing and have to be observed for up to 2 hours post dose for symptoms,” Dr. Kim said.
“It’s a great drug, but the treatment could be overly difficult for certain families to be able to do, and in some cases the side effects may be more than certain patients are able or willing to handle, so there is a real urgent need for alternative approaches,” Dr. Kim said. “SLIT is several drops under the tongue, held for 2 minutes, swallowed and done.”
In the current placebo-controlled study, he and his group tested the feasibility, efficacy, and safety of the sublingual approach to peanut allergy in children age 4 years and younger.
Both groups were similar with regard to gender, race, ethnicity, atopic history, peanut skin prick test, and qualifying DBPCFC, and all children were previously allergic with positive blood and skin tests, with a positive reaction during baseline food challenge, thus proving the allergy and establishing the baseline threshold.
“We have learned from some studies, for instance the DEVIL and LEAP studies, that strongly suggest that the immune systems in younger patients may be more amenable to change, and there may be some justification for early intervention,” he said.
“Based on both of those ideas, we wanted to take our sublingual approach, which we have shown to have a pretty good efficacy in older children, and bring it down to this younger group and see if it still could have the same efficacy and also maintain what seems to be a very good safety signal.”
The researchers randomly assigned the children to receive PNSLIT at a daily maintenance dose of 4 mg peanut protein (n = 19) or to receive placebo (n = 17) for 36 months.
“There was a 5- to 6-month buildup period where the SLIT dose was increased every 1-2 weeks up to the target dose of 4 mg, and then the final dose of 4 mg was continued through to the end of the study,” Dr. Kim noted.
Over a total of 20,593 potential dosing days, the children took 91.2% of SLIT doses and 93.5% of placebo doses.
At the end of the 3-year study period, the children were challenged by DBPCFC with up to 4,333 mg of peanut protein.
Sustained unresponsiveness was assessed by an identical DBPCFC after discontinuation of the immunotherapy for 3 months.
Cumulative tolerated dose increased from a median of 143 mg to 4,443 mg in the PNSLIT group, compared with a median of 43 mg to 143 mg in the placebo group (P < .0001).
Fourteen of the children receiving PNSLIT, and none of the children receiving placebo, passed the desensitization food challenge. Twelve of the children receiving PNSLIT and two of the children receiving placebo passed the sustained unresponsiveness challenge.
Children who underwent the immunotherapy saw a decrease in their peanut skin prick test from 10 mm to 3.25 mm, compared to an increase from 11.5 mm to 12 mm with placebo (P < .0001).
The most common side effect reported was itching or irritation in the mouth. Most side effects resolved on their own, although some patients used an antihistamine. Getting children as young as 1 to hold the dose under their tongue was a challenge in some instances, but it eventually worked out, Dr. Kim said.
“It took a lot of work from the parents as well as from our research coordinators in trying to train these young kids to, first of all, allow us to put the peanut medication in the mouth and then to try as best as possible to keep it in their mouth for up to 2 minutes, but the families involved in our study were very dedicated and so we were able to get through that,” he said.
Study merits larger numbers
“Among the 36 who completed the 3 years of therapy, the authors report significant rates of desensitization among treated children compared with those receiving placebo. Furthermore, this effect was persistent for at least 3 months after stopping therapy in a subgroup of the children,” said Leonard B. Bacharier, MD, director of the Center for Pediatric Asthma, Monroe Carell Jr. Children’s Hospital at Vanderbilt, Nashville, Tenn.
“Overall, these findings suggest the promise of peanut SLIT, which should be studied in larger numbers of preschool children,” Dr. Bacharier, who was not part of the study, said in an interview.
Jonathan A. Bernstein, MD, professor of medicine, University of Cincinnati, agreed.
“It’s a well-designed study, it’s small, but it’s promising,” Dr. Bernstein, who was not involved with the study, said in an interview.
“They did show that most of the patients who got the sublingual therapy were able to get to the target dose and develop tolerance, so I think it’s promising. We know that this stuff works. This is just more data from a well-controlled study in a younger population,” he said.
“We do OIT [oral immunotherapy] and sublingual but we don’t do it in such young children in our practice. The youngest is 3 years old, because they have to understand what is going on and cooperate. If they don’t cooperate it’s not possible.”
Dr. Kim reported financial relationships with DBV Technologies, Kenota Health, Ukko, Aimmune Therapeutics, ALK, AllerGenis, Belhaven Pharma, Duke Clinical Research Institute, Nutricia, NIH/NIAID, NIH/NCCIH, NIH/Immune Tolerance Network, FARE, and the Wallace Foundation. Dr. Bacharier and Dr. Bernstein have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Sublingual immunotherapy for the treatment of peanut allergy is safe and effective, even in children as young as age 1 year.
In a double-blind, placebo-controlled, food challenge (DBPCFC) of some 36 peanut-allergic children (mean age 2.2 years, range 1-4 years), those who were randomly assigned to receive peanut sublingual immunotherapy (PNSLIT) showed significant desensitization compared with those who received placebo.
In addition, there was a “strong potential” for sustained unresponsiveness at 3 months for the toddlers who received the active treatment.
The findings were presented in a late breaking oral abstract session at the 2021 American Academy of Allergy, Asthma & Immunology virtual annual meeting (Abstract L2).
“A year ago, the Food and Drug Administration approved the oral agent Palforzia (peanut allergen powder) for the treatment of peanut allergy in children 4 and older, and it is a great option, but I think what we have learned over time is that this approach is not for everybody,” Edwin H. Kim, MD, director of the UNC Food Allergy Initiative, University of North Carolina at Chapel Hill, said in an interview.
Palforzia is a powder that is mixed in food like yogurt or pudding which the child then eats daily, according to a rigorous schedule. But Palforzia treatment presents some difficulties.
“Palforzia requires getting the powder dose, mixing it with food, like pudding or apple sauce, then eating it, which can take up to 30 minutes depending on age and kids’ cooperation. It tastes and smells like peanut which can cause aversion. Kids have to refrain from exercise or strenuous activity for at least 30 minutes before and after dosing and have to be observed for up to 2 hours post dose for symptoms,” Dr. Kim said.
“It’s a great drug, but the treatment could be overly difficult for certain families to be able to do, and in some cases the side effects may be more than certain patients are able or willing to handle, so there is a real urgent need for alternative approaches,” Dr. Kim said. “SLIT is several drops under the tongue, held for 2 minutes, swallowed and done.”
In the current placebo-controlled study, he and his group tested the feasibility, efficacy, and safety of the sublingual approach to peanut allergy in children age 4 years and younger.
Both groups were similar with regard to gender, race, ethnicity, atopic history, peanut skin prick test, and qualifying DBPCFC, and all children were previously allergic with positive blood and skin tests, with a positive reaction during baseline food challenge, thus proving the allergy and establishing the baseline threshold.
“We have learned from some studies, for instance the DEVIL and LEAP studies, that strongly suggest that the immune systems in younger patients may be more amenable to change, and there may be some justification for early intervention,” he said.
“Based on both of those ideas, we wanted to take our sublingual approach, which we have shown to have a pretty good efficacy in older children, and bring it down to this younger group and see if it still could have the same efficacy and also maintain what seems to be a very good safety signal.”
The researchers randomly assigned the children to receive PNSLIT at a daily maintenance dose of 4 mg peanut protein (n = 19) or to receive placebo (n = 17) for 36 months.
“There was a 5- to 6-month buildup period where the SLIT dose was increased every 1-2 weeks up to the target dose of 4 mg, and then the final dose of 4 mg was continued through to the end of the study,” Dr. Kim noted.
Over a total of 20,593 potential dosing days, the children took 91.2% of SLIT doses and 93.5% of placebo doses.
At the end of the 3-year study period, the children were challenged by DBPCFC with up to 4,333 mg of peanut protein.
Sustained unresponsiveness was assessed by an identical DBPCFC after discontinuation of the immunotherapy for 3 months.
Cumulative tolerated dose increased from a median of 143 mg to 4,443 mg in the PNSLIT group, compared with a median of 43 mg to 143 mg in the placebo group (P < .0001).
Fourteen of the children receiving PNSLIT, and none of the children receiving placebo, passed the desensitization food challenge. Twelve of the children receiving PNSLIT and two of the children receiving placebo passed the sustained unresponsiveness challenge.
Children who underwent the immunotherapy saw a decrease in their peanut skin prick test from 10 mm to 3.25 mm, compared to an increase from 11.5 mm to 12 mm with placebo (P < .0001).
The most common side effect reported was itching or irritation in the mouth. Most side effects resolved on their own, although some patients used an antihistamine. Getting children as young as 1 to hold the dose under their tongue was a challenge in some instances, but it eventually worked out, Dr. Kim said.
“It took a lot of work from the parents as well as from our research coordinators in trying to train these young kids to, first of all, allow us to put the peanut medication in the mouth and then to try as best as possible to keep it in their mouth for up to 2 minutes, but the families involved in our study were very dedicated and so we were able to get through that,” he said.
Study merits larger numbers
“Among the 36 who completed the 3 years of therapy, the authors report significant rates of desensitization among treated children compared with those receiving placebo. Furthermore, this effect was persistent for at least 3 months after stopping therapy in a subgroup of the children,” said Leonard B. Bacharier, MD, director of the Center for Pediatric Asthma, Monroe Carell Jr. Children’s Hospital at Vanderbilt, Nashville, Tenn.
“Overall, these findings suggest the promise of peanut SLIT, which should be studied in larger numbers of preschool children,” Dr. Bacharier, who was not part of the study, said in an interview.
Jonathan A. Bernstein, MD, professor of medicine, University of Cincinnati, agreed.
“It’s a well-designed study, it’s small, but it’s promising,” Dr. Bernstein, who was not involved with the study, said in an interview.
“They did show that most of the patients who got the sublingual therapy were able to get to the target dose and develop tolerance, so I think it’s promising. We know that this stuff works. This is just more data from a well-controlled study in a younger population,” he said.
“We do OIT [oral immunotherapy] and sublingual but we don’t do it in such young children in our practice. The youngest is 3 years old, because they have to understand what is going on and cooperate. If they don’t cooperate it’s not possible.”
Dr. Kim reported financial relationships with DBV Technologies, Kenota Health, Ukko, Aimmune Therapeutics, ALK, AllerGenis, Belhaven Pharma, Duke Clinical Research Institute, Nutricia, NIH/NIAID, NIH/NCCIH, NIH/Immune Tolerance Network, FARE, and the Wallace Foundation. Dr. Bacharier and Dr. Bernstein have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Sublingual immunotherapy for the treatment of peanut allergy is safe and effective, even in children as young as age 1 year.
In a double-blind, placebo-controlled, food challenge (DBPCFC) of some 36 peanut-allergic children (mean age 2.2 years, range 1-4 years), those who were randomly assigned to receive peanut sublingual immunotherapy (PNSLIT) showed significant desensitization compared with those who received placebo.
In addition, there was a “strong potential” for sustained unresponsiveness at 3 months for the toddlers who received the active treatment.
The findings were presented in a late breaking oral abstract session at the 2021 American Academy of Allergy, Asthma & Immunology virtual annual meeting (Abstract L2).
“A year ago, the Food and Drug Administration approved the oral agent Palforzia (peanut allergen powder) for the treatment of peanut allergy in children 4 and older, and it is a great option, but I think what we have learned over time is that this approach is not for everybody,” Edwin H. Kim, MD, director of the UNC Food Allergy Initiative, University of North Carolina at Chapel Hill, said in an interview.
Palforzia is a powder that is mixed in food like yogurt or pudding which the child then eats daily, according to a rigorous schedule. But Palforzia treatment presents some difficulties.
“Palforzia requires getting the powder dose, mixing it with food, like pudding or apple sauce, then eating it, which can take up to 30 minutes depending on age and kids’ cooperation. It tastes and smells like peanut which can cause aversion. Kids have to refrain from exercise or strenuous activity for at least 30 minutes before and after dosing and have to be observed for up to 2 hours post dose for symptoms,” Dr. Kim said.
“It’s a great drug, but the treatment could be overly difficult for certain families to be able to do, and in some cases the side effects may be more than certain patients are able or willing to handle, so there is a real urgent need for alternative approaches,” Dr. Kim said. “SLIT is several drops under the tongue, held for 2 minutes, swallowed and done.”
In the current placebo-controlled study, he and his group tested the feasibility, efficacy, and safety of the sublingual approach to peanut allergy in children age 4 years and younger.
Both groups were similar with regard to gender, race, ethnicity, atopic history, peanut skin prick test, and qualifying DBPCFC, and all children were previously allergic with positive blood and skin tests, with a positive reaction during baseline food challenge, thus proving the allergy and establishing the baseline threshold.
“We have learned from some studies, for instance the DEVIL and LEAP studies, that strongly suggest that the immune systems in younger patients may be more amenable to change, and there may be some justification for early intervention,” he said.
“Based on both of those ideas, we wanted to take our sublingual approach, which we have shown to have a pretty good efficacy in older children, and bring it down to this younger group and see if it still could have the same efficacy and also maintain what seems to be a very good safety signal.”
The researchers randomly assigned the children to receive PNSLIT at a daily maintenance dose of 4 mg peanut protein (n = 19) or to receive placebo (n = 17) for 36 months.
“There was a 5- to 6-month buildup period where the SLIT dose was increased every 1-2 weeks up to the target dose of 4 mg, and then the final dose of 4 mg was continued through to the end of the study,” Dr. Kim noted.
Over a total of 20,593 potential dosing days, the children took 91.2% of SLIT doses and 93.5% of placebo doses.
At the end of the 3-year study period, the children were challenged by DBPCFC with up to 4,333 mg of peanut protein.
Sustained unresponsiveness was assessed by an identical DBPCFC after discontinuation of the immunotherapy for 3 months.
Cumulative tolerated dose increased from a median of 143 mg to 4,443 mg in the PNSLIT group, compared with a median of 43 mg to 143 mg in the placebo group (P < .0001).
Fourteen of the children receiving PNSLIT, and none of the children receiving placebo, passed the desensitization food challenge. Twelve of the children receiving PNSLIT and two of the children receiving placebo passed the sustained unresponsiveness challenge.
Children who underwent the immunotherapy saw a decrease in their peanut skin prick test from 10 mm to 3.25 mm, compared to an increase from 11.5 mm to 12 mm with placebo (P < .0001).
The most common side effect reported was itching or irritation in the mouth. Most side effects resolved on their own, although some patients used an antihistamine. Getting children as young as 1 to hold the dose under their tongue was a challenge in some instances, but it eventually worked out, Dr. Kim said.
“It took a lot of work from the parents as well as from our research coordinators in trying to train these young kids to, first of all, allow us to put the peanut medication in the mouth and then to try as best as possible to keep it in their mouth for up to 2 minutes, but the families involved in our study were very dedicated and so we were able to get through that,” he said.
Study merits larger numbers
“Among the 36 who completed the 3 years of therapy, the authors report significant rates of desensitization among treated children compared with those receiving placebo. Furthermore, this effect was persistent for at least 3 months after stopping therapy in a subgroup of the children,” said Leonard B. Bacharier, MD, director of the Center for Pediatric Asthma, Monroe Carell Jr. Children’s Hospital at Vanderbilt, Nashville, Tenn.
“Overall, these findings suggest the promise of peanut SLIT, which should be studied in larger numbers of preschool children,” Dr. Bacharier, who was not part of the study, said in an interview.
Jonathan A. Bernstein, MD, professor of medicine, University of Cincinnati, agreed.
“It’s a well-designed study, it’s small, but it’s promising,” Dr. Bernstein, who was not involved with the study, said in an interview.
“They did show that most of the patients who got the sublingual therapy were able to get to the target dose and develop tolerance, so I think it’s promising. We know that this stuff works. This is just more data from a well-controlled study in a younger population,” he said.
“We do OIT [oral immunotherapy] and sublingual but we don’t do it in such young children in our practice. The youngest is 3 years old, because they have to understand what is going on and cooperate. If they don’t cooperate it’s not possible.”
Dr. Kim reported financial relationships with DBV Technologies, Kenota Health, Ukko, Aimmune Therapeutics, ALK, AllerGenis, Belhaven Pharma, Duke Clinical Research Institute, Nutricia, NIH/NIAID, NIH/NCCIH, NIH/Immune Tolerance Network, FARE, and the Wallace Foundation. Dr. Bacharier and Dr. Bernstein have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AAAAI