User login
Atopic dermatitis doubles risk of mental health issues in children
, according to a newly published cohort study of more than 11,000 children between the ages of 3 and 18 years.
Along with previous studies that have also linked AD to depression and other mental health issues in children, these data highlight the need for “clinical awareness of the psychosocial needs of children and adolescents with AD,” reported a multicenter team of investigators from the University of California, San Francisco, the University of Pennsylvania, and the London School of Hygiene and Tropical Medicine.
Unlike some previous studies, in this study, published online in JAMA Dermatology on Sept. 1, children were evaluated longitudinally, rather than at a single point in time, with a mean follow-up of 10 years. For those with active AD, compared with children without AD, the odds ratio for depression overall in any child with AD relative to those without AD was not significant after adjustment for variables such socioeconomic factors.
However, among children with severe AD, the risk was more than twofold greater even after adjustment (adjusted OR, 2.38; 95% confidence interval, 1.21- 4.72), reported the investigators, led by senior author Katrina Abuabara, MD, associate professor of dermatology and epidemiology at UCSF.
Internalizing symptoms seen with mild to severe AD
Internalizing behavior, which is closely linked to depression and describes a spectrum of inward-focusing activities, such as social withdrawal, was significantly more common in children with any degree of AD relative to those without AD: After adjustment, the risk climbed from a 29% increased risk in those with mild AD (aOR, 1.29; 95% CI, 1.06-1.57) to a more than 80% increased risk in children with moderate AD (aOR, 1.84; 95% CI, 1.40-2.41) and in children with severe AD (aOR, 1.90; 95% CI, 1.14-3.16).
In the study, depression was measured with the Short Moods and Feelings Questionnaire (SMFQ). Parental response to the Emotional Symptoms subscale of the Strength and Difficulties Questionnaire (SDQ) was used to measure internalizing behaviors.
The data were drawn from the Avon Longitudinal Study for Parents and Children (ALSPAC), a cohort that enrolled pregnant women in a defined area in southwest England and then followed children born from these pregnancies. Of the 14,062 children enrolled in ALSPAC, data from 11,181 children were available for this study.
In a previous meta-analysis of studies that have documented a link between AD and adverse effects on mood and mental health, an impact was identified in both children and adults. In children, AD was associated with a 27% increase in risk of depression (OR, 1.27; 95% CI, 1.12 -1.45). In adults, the risk was more than doubled (OR, 2.19; 95% CI, 1.87-2.57). The same meta-analysis found that the risk of suicidal ideation among adolescents and adults with AD was increased more than fourfold (OR, 4.32; 95% CI, 1.93-9.66).
In the ALSPAC data, the investigators were unable to find compelling evidence that sleep disturbances or concomitant asthma contributed to the increased risk of depression, which is a mechanism proposed by past investigators.
In an interview, Dr. Abuabara said that these and other data provide the basis for encouraging clinical awareness of the psychological needs of children with AD, but she suggested there is a gap in understanding what this means clinically. “We need more data on how dermatologists can effectively screen and manage these patients before we try to set expectations for clinical practice,” she said.
In addition, these data along with previously published studies suggest that change in mental health outcomes should be included in the evaluation of new therapies, according to Dr. Abuabara. She noted that there are several tools for evaluating mental health in children that might be appropriate, each with their own advantages and disadvantages.
“Ideally, recommendations would be issued through a group consensus process with patients, clinicians, researchers, and industry representatives working together as has been done for other outcomes through the Harmonizing Measures for Eczema (HOME) group,” Dr. Abuabara said.
Mental health assessments recommended
Others who have looked at the relationship between AD and depression have also recommended adding mental health outcomes to an assessment of efficacy for AD therapies.
Jonathan I. Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, is one such investigator. He is already monitoring depression systematically with the Hospital Anxiety and Depression Scale (HADS).
“HADS has been validated in AD and provides very important information about the emotional burden of AD,” explained Dr. Silverberg, whose most recent article on this topic appeared earlier this year. In that study, the relationship between AD and depression was found to be more pronounced in White children from families with lower incomes.
“Just a few hours ago, one of my patients thanked me for asking about their mental health and recognizing the holistic effects of AD,” Dr. Silverberg said.
The recent study based on ALSPAC data add to the evidence that AD, particularly severe AD, produces deleterious effects on mental health in children, and Dr. Silverberg believes clinicians should be acting on this evidence.
“I strongly encourage clinicians to routinely assess mental health. It will elevate the quality of care they provide, and their patients will appreciate them more for it,” he said.
Dr. Abuabara and another author report receiving research funding from Pfizer to their universities for unrelated work; there were no other disclosures. Dr. Silverberg reports financial relationships with more than 15 pharmaceutical companies.
Commentary by Lawrence F. Eichenfield, MD
More severe atopic dermatitis (AD) carries with it significant mental health concerns in children, as well as adults. Multiple studies have shown significantly higher rates of depression, anxiety, and “internalizing behaviors” (discussed as social withdrawal and other inward-focused activities) as well as attention-deficit/hyperactivity disorder. The study by Dr. Abuabara and colleagues is important as it followed children over time (an average of 10 years) and adjusted the data for socioeconomic factors, showing a rate of depression in children with severe AD twice that of those without. It appears that we are in the midst of a mental health crisis in children and teens, with markedly higher rates of pediatric and adolescent depression and anxiety, certainly influenced by COVID-19 societal changes. As the literature has developed on depression and AD, we have appreciated the importance of addressing this as part of our assessment of the disease effect on the individual and family, and it is one factor we consider in selections of systemic vs. topical therapies.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
, according to a newly published cohort study of more than 11,000 children between the ages of 3 and 18 years.
Along with previous studies that have also linked AD to depression and other mental health issues in children, these data highlight the need for “clinical awareness of the psychosocial needs of children and adolescents with AD,” reported a multicenter team of investigators from the University of California, San Francisco, the University of Pennsylvania, and the London School of Hygiene and Tropical Medicine.
Unlike some previous studies, in this study, published online in JAMA Dermatology on Sept. 1, children were evaluated longitudinally, rather than at a single point in time, with a mean follow-up of 10 years. For those with active AD, compared with children without AD, the odds ratio for depression overall in any child with AD relative to those without AD was not significant after adjustment for variables such socioeconomic factors.
However, among children with severe AD, the risk was more than twofold greater even after adjustment (adjusted OR, 2.38; 95% confidence interval, 1.21- 4.72), reported the investigators, led by senior author Katrina Abuabara, MD, associate professor of dermatology and epidemiology at UCSF.
Internalizing symptoms seen with mild to severe AD
Internalizing behavior, which is closely linked to depression and describes a spectrum of inward-focusing activities, such as social withdrawal, was significantly more common in children with any degree of AD relative to those without AD: After adjustment, the risk climbed from a 29% increased risk in those with mild AD (aOR, 1.29; 95% CI, 1.06-1.57) to a more than 80% increased risk in children with moderate AD (aOR, 1.84; 95% CI, 1.40-2.41) and in children with severe AD (aOR, 1.90; 95% CI, 1.14-3.16).
In the study, depression was measured with the Short Moods and Feelings Questionnaire (SMFQ). Parental response to the Emotional Symptoms subscale of the Strength and Difficulties Questionnaire (SDQ) was used to measure internalizing behaviors.
The data were drawn from the Avon Longitudinal Study for Parents and Children (ALSPAC), a cohort that enrolled pregnant women in a defined area in southwest England and then followed children born from these pregnancies. Of the 14,062 children enrolled in ALSPAC, data from 11,181 children were available for this study.
In a previous meta-analysis of studies that have documented a link between AD and adverse effects on mood and mental health, an impact was identified in both children and adults. In children, AD was associated with a 27% increase in risk of depression (OR, 1.27; 95% CI, 1.12 -1.45). In adults, the risk was more than doubled (OR, 2.19; 95% CI, 1.87-2.57). The same meta-analysis found that the risk of suicidal ideation among adolescents and adults with AD was increased more than fourfold (OR, 4.32; 95% CI, 1.93-9.66).
In the ALSPAC data, the investigators were unable to find compelling evidence that sleep disturbances or concomitant asthma contributed to the increased risk of depression, which is a mechanism proposed by past investigators.
In an interview, Dr. Abuabara said that these and other data provide the basis for encouraging clinical awareness of the psychological needs of children with AD, but she suggested there is a gap in understanding what this means clinically. “We need more data on how dermatologists can effectively screen and manage these patients before we try to set expectations for clinical practice,” she said.
In addition, these data along with previously published studies suggest that change in mental health outcomes should be included in the evaluation of new therapies, according to Dr. Abuabara. She noted that there are several tools for evaluating mental health in children that might be appropriate, each with their own advantages and disadvantages.
“Ideally, recommendations would be issued through a group consensus process with patients, clinicians, researchers, and industry representatives working together as has been done for other outcomes through the Harmonizing Measures for Eczema (HOME) group,” Dr. Abuabara said.
Mental health assessments recommended
Others who have looked at the relationship between AD and depression have also recommended adding mental health outcomes to an assessment of efficacy for AD therapies.
Jonathan I. Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, is one such investigator. He is already monitoring depression systematically with the Hospital Anxiety and Depression Scale (HADS).
“HADS has been validated in AD and provides very important information about the emotional burden of AD,” explained Dr. Silverberg, whose most recent article on this topic appeared earlier this year. In that study, the relationship between AD and depression was found to be more pronounced in White children from families with lower incomes.
“Just a few hours ago, one of my patients thanked me for asking about their mental health and recognizing the holistic effects of AD,” Dr. Silverberg said.
The recent study based on ALSPAC data add to the evidence that AD, particularly severe AD, produces deleterious effects on mental health in children, and Dr. Silverberg believes clinicians should be acting on this evidence.
“I strongly encourage clinicians to routinely assess mental health. It will elevate the quality of care they provide, and their patients will appreciate them more for it,” he said.
Dr. Abuabara and another author report receiving research funding from Pfizer to their universities for unrelated work; there were no other disclosures. Dr. Silverberg reports financial relationships with more than 15 pharmaceutical companies.
Commentary by Lawrence F. Eichenfield, MD
More severe atopic dermatitis (AD) carries with it significant mental health concerns in children, as well as adults. Multiple studies have shown significantly higher rates of depression, anxiety, and “internalizing behaviors” (discussed as social withdrawal and other inward-focused activities) as well as attention-deficit/hyperactivity disorder. The study by Dr. Abuabara and colleagues is important as it followed children over time (an average of 10 years) and adjusted the data for socioeconomic factors, showing a rate of depression in children with severe AD twice that of those without. It appears that we are in the midst of a mental health crisis in children and teens, with markedly higher rates of pediatric and adolescent depression and anxiety, certainly influenced by COVID-19 societal changes. As the literature has developed on depression and AD, we have appreciated the importance of addressing this as part of our assessment of the disease effect on the individual and family, and it is one factor we consider in selections of systemic vs. topical therapies.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
, according to a newly published cohort study of more than 11,000 children between the ages of 3 and 18 years.
Along with previous studies that have also linked AD to depression and other mental health issues in children, these data highlight the need for “clinical awareness of the psychosocial needs of children and adolescents with AD,” reported a multicenter team of investigators from the University of California, San Francisco, the University of Pennsylvania, and the London School of Hygiene and Tropical Medicine.
Unlike some previous studies, in this study, published online in JAMA Dermatology on Sept. 1, children were evaluated longitudinally, rather than at a single point in time, with a mean follow-up of 10 years. For those with active AD, compared with children without AD, the odds ratio for depression overall in any child with AD relative to those without AD was not significant after adjustment for variables such socioeconomic factors.
However, among children with severe AD, the risk was more than twofold greater even after adjustment (adjusted OR, 2.38; 95% confidence interval, 1.21- 4.72), reported the investigators, led by senior author Katrina Abuabara, MD, associate professor of dermatology and epidemiology at UCSF.
Internalizing symptoms seen with mild to severe AD
Internalizing behavior, which is closely linked to depression and describes a spectrum of inward-focusing activities, such as social withdrawal, was significantly more common in children with any degree of AD relative to those without AD: After adjustment, the risk climbed from a 29% increased risk in those with mild AD (aOR, 1.29; 95% CI, 1.06-1.57) to a more than 80% increased risk in children with moderate AD (aOR, 1.84; 95% CI, 1.40-2.41) and in children with severe AD (aOR, 1.90; 95% CI, 1.14-3.16).
In the study, depression was measured with the Short Moods and Feelings Questionnaire (SMFQ). Parental response to the Emotional Symptoms subscale of the Strength and Difficulties Questionnaire (SDQ) was used to measure internalizing behaviors.
The data were drawn from the Avon Longitudinal Study for Parents and Children (ALSPAC), a cohort that enrolled pregnant women in a defined area in southwest England and then followed children born from these pregnancies. Of the 14,062 children enrolled in ALSPAC, data from 11,181 children were available for this study.
In a previous meta-analysis of studies that have documented a link between AD and adverse effects on mood and mental health, an impact was identified in both children and adults. In children, AD was associated with a 27% increase in risk of depression (OR, 1.27; 95% CI, 1.12 -1.45). In adults, the risk was more than doubled (OR, 2.19; 95% CI, 1.87-2.57). The same meta-analysis found that the risk of suicidal ideation among adolescents and adults with AD was increased more than fourfold (OR, 4.32; 95% CI, 1.93-9.66).
In the ALSPAC data, the investigators were unable to find compelling evidence that sleep disturbances or concomitant asthma contributed to the increased risk of depression, which is a mechanism proposed by past investigators.
In an interview, Dr. Abuabara said that these and other data provide the basis for encouraging clinical awareness of the psychological needs of children with AD, but she suggested there is a gap in understanding what this means clinically. “We need more data on how dermatologists can effectively screen and manage these patients before we try to set expectations for clinical practice,” she said.
In addition, these data along with previously published studies suggest that change in mental health outcomes should be included in the evaluation of new therapies, according to Dr. Abuabara. She noted that there are several tools for evaluating mental health in children that might be appropriate, each with their own advantages and disadvantages.
“Ideally, recommendations would be issued through a group consensus process with patients, clinicians, researchers, and industry representatives working together as has been done for other outcomes through the Harmonizing Measures for Eczema (HOME) group,” Dr. Abuabara said.
Mental health assessments recommended
Others who have looked at the relationship between AD and depression have also recommended adding mental health outcomes to an assessment of efficacy for AD therapies.
Jonathan I. Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, is one such investigator. He is already monitoring depression systematically with the Hospital Anxiety and Depression Scale (HADS).
“HADS has been validated in AD and provides very important information about the emotional burden of AD,” explained Dr. Silverberg, whose most recent article on this topic appeared earlier this year. In that study, the relationship between AD and depression was found to be more pronounced in White children from families with lower incomes.
“Just a few hours ago, one of my patients thanked me for asking about their mental health and recognizing the holistic effects of AD,” Dr. Silverberg said.
The recent study based on ALSPAC data add to the evidence that AD, particularly severe AD, produces deleterious effects on mental health in children, and Dr. Silverberg believes clinicians should be acting on this evidence.
“I strongly encourage clinicians to routinely assess mental health. It will elevate the quality of care they provide, and their patients will appreciate them more for it,” he said.
Dr. Abuabara and another author report receiving research funding from Pfizer to their universities for unrelated work; there were no other disclosures. Dr. Silverberg reports financial relationships with more than 15 pharmaceutical companies.
Commentary by Lawrence F. Eichenfield, MD
More severe atopic dermatitis (AD) carries with it significant mental health concerns in children, as well as adults. Multiple studies have shown significantly higher rates of depression, anxiety, and “internalizing behaviors” (discussed as social withdrawal and other inward-focused activities) as well as attention-deficit/hyperactivity disorder. The study by Dr. Abuabara and colleagues is important as it followed children over time (an average of 10 years) and adjusted the data for socioeconomic factors, showing a rate of depression in children with severe AD twice that of those without. It appears that we are in the midst of a mental health crisis in children and teens, with markedly higher rates of pediatric and adolescent depression and anxiety, certainly influenced by COVID-19 societal changes. As the literature has developed on depression and AD, we have appreciated the importance of addressing this as part of our assessment of the disease effect on the individual and family, and it is one factor we consider in selections of systemic vs. topical therapies.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
FROM JAMA DERMATOLOGY
Autoeczematization: A Strange Id Reaction of the Skin
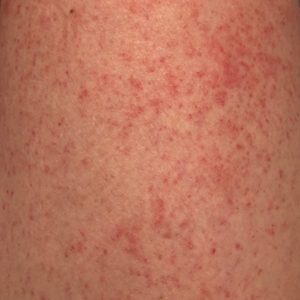
Autoeczematization (AE), or id reaction, is a disseminated eczematous reaction that occurs days or weeks after exposure to a primary stimulus, resulting from a release of antigen(s). Whitfield1 first described AE in 1921, when he postulated that the id reaction was due to sensitization of the skin after a primary stimulus. He called it “a form of auto-intoxication derived from changes in the patient’s own tissues.”1 The exact prevalence of id reactions is unknown; one study showed that 17% of patients with dermatophyte infections developed an id reaction, typically tinea pedis linked with vesicles on the palms.2 Tinea capitis is one of the most common causes of AE in children, which is frequently misdiagnosed as a drug reaction. Approximately 37% of patients diagnosed with stasis dermatitis develop an id reaction (Figure 1). A history of contact dermatitis is common in patients presenting with AE.2-6

Pathophysiology of Id Reactions
An abnormal immune response against autologous skin antigens may be responsible for the development of AE. Shelley5 postulated that hair follicles play an important role in id reactions, as Sharquie et al6 recently emphasized for many skin disorders. The pathogenesis of AE is uncertain, but circulating T lymphocytes play a role in this reaction. Normally, T cells are activated by a release of antigens after a primary exposure to a stimulus. However, overactivation of these T cells induces autoimmune reactions such as AE.7 Activated T lymphocytes express HLA-DR and IL-2 receptor, markers elevated in the peripheral blood of patients undergoing id reactions. After treatment, the levels of activated T lymphocytes decline. An increase in the number of CD25+ T cells and a decrease in the number of suppressor T cells in the blood may occur during an id reaction.7-9 Keratinocytes produce proinflammatory cytokines, such as thymic stromal erythropoietin, IL-25, and IL-33, that activate T cells.10-12 Therefore, the most likely pathogenesis of an id reaction is that T lymphocytes are activated at the primary reaction site due to proinflammatory cytokines released by keratinocytes. These activated T cells then travel systemically via hematogenous dissemination. The spread of activated T lymphocytes produces an eczematous reaction at secondary locations distant to the primary site.9
Clinical and Histopathological Features of Id Reactions
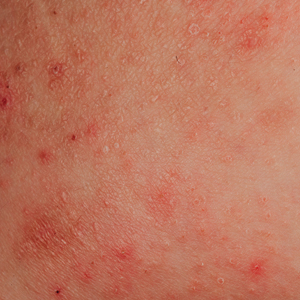
Clinically, AE is first evident as a vesicular dissemination that groups to form papules or nummular patches and usually is present on the legs, feet, arms, and/or trunk (Figure 2). The primary dermatitis is localized to the area that was the site of contact to the offending stimuli. This localized eczematous eruption begins with an acute or subacute onset. It has the appearance of small crusted vesicles with erythema (Figure 1). The first sign of AE is vesicles presenting near the primary site on flexural surfaces or on the hands and feet. A classic example is tinea pedis linked with vesicles on the palms and sides of the fingers, resembling dyshidrotic eczema. Sites of prior cutaneous trauma, such as dermatoses, scars, and burns, are common locations for early AE. In later stages, vesicles disseminate to the legs, arms, and trunk, where they group to form papules and nummular patches in a symmetrical pattern.5,13-15 These lesions may be extremely pruritic. The pruritus may be so intense that it interrupts daily activities and disrupts the ability to fall or stay asleep.16
Histologically, biopsy specimens show psoriasiform spongiotic dermatitis with mononuclear cells contained in the vesicles. Interstitial edema and perivascular lymphohistiocytic infiltrates are evident. Eosinophils also may be present. This pattern is not unique toid reactions.17-19 Although AE is a reaction pattern that may be due to a fungal or bacterial infection, the etiologic agent is not evident microscopically within the eczema itself.
Etiology of Id Reactions
Id reactions most commonly occur from either stasis dermatitis or tinea pedis, although a wide variety of other causes should be considered. Evaluation of the primary site rather than the id reaction may identify an infectious or parasitic agent. Sometimes the AE reaction is specifically named: dermatophytid with dermatophytosis, bacterid with a bacterial infectious process, and tuberculid with tuberculosis. Similarly, there may be reactions to underlying candidiasis, sporotrichosis, histoplasmosis, and other fungal infections that can cause a cutaneous id reaction.18,20-22Mycobacterium species, Pseudomonas, Staphylococcus, and Streptococcus are bacterial causes of AE.15,23-26 Viral infections that can cause an id reaction are herpes simplex virus and molluscum contagiosum.27-29 Scabies, leishmaniasis, and pediculosis capitis are parasitic infections that may be etiologic.14,30,31 In addition, noninfectious stimuli besides stasis dermatitis that can produce id reactions include medications, topical creams, tattoo ink, sutures, radiotherapy, and dyshidrotic eczema. The primary reaction to these agents is a localized dermatitis followed by the immunological response that induces a secondary reaction distant from the primary site.17,18,32-38
Differential Diagnoses
Differential diagnoses include other types of eczema and some vesicular eruptions. Irritant contact dermatitis is another dermatosis that presents as a widespread vesicular eruption due to repetitive exposure to toxic irritants. The rash is erythematous with pustules, blisters, and crusts. It is only found in areas directly exposed to irritants, as opposed to AE, which spreads to areas distant to the primary reaction site. Irritant contact dermatitis presents with more of a burning sensation, whereas AE is more pruritic.39,40 Allergic contact dermatitis presents with erythematous vesicles and papules and sometimes with bullae. There is edema and crust formation, which often can spread past the point of contact in later stages. Similar to AE, there is intense pruritus. However, allergic contact dermatitis most commonly is caused by exposure to metals, cosmetics, and fragrances, whereas infectious agents and stasis dermatitis are the most common causes of AE.40,41 It may be challenging to distinguish AE from other causes of widespread eczematous dissemination. Vesicular eruptions sometimes require distinction from AE, including herpetic infections, insect bite reactions, and drug eruptions.18,42
Treatment
The underlying condition should be treated to mitigate the inflammatory response causing the id reaction. If not skillfully orchestrated, the id reaction can reoccur. For infectious causes of AE, an antifungal, antibacterial, antiviral, or antiparasitic should be given. If stasis dermatitis is responsible for the id reaction, compression stockings and leg elevation are indicated. The id reaction itself is treated with systemic or topical corticosteroids and wet compresses if acute. The goal of these treatments is to reduce patient discomfort caused by the inflammation and pruritus.18,43
Conclusion
Id reactions are an unusual phenomenon that commonly occurs after fungal skin infections and stasis dermatitis. T lymphocytes and keratinocytes may play a key role in this reaction, with newer research further delineating the process and possibly providing enhanced treatment options. Therapy focuses on treating the underlying condition, supplemented with corticosteroids for the autoeczema.
- Whitfield A. Lumleian Lectures on Some Points in the Aetiology of Skin Diseases. Delivered before the Royal College of Physicians of London on March 10th, 15th, and 17th, 1921. Lecture II. Lancet. 1921;2:122-127.
- Cheng N, Rucker Wright D, Cohen BA. Dermatophytid in tinea capitis: rarely reported common phenomenon with clinical implications. Pediatrics. 2011;128:E453-E457.
- Schrom KP, Kobs A, Nedorost S. Clinical psoriasiform dermatitis following dupilumab use for autoeczematization secondary to chronic stasis dermatitis. Cureus. 2020;12:e7831. doi:10.7759/cureus.7831
- Templeton HJ, Lunsford CJ, Allington HV. Autosensitization dermatitis; report of five cases and protocol of an experiment. Arch Derm Syphilol. 1949;59:68-77.
- Shelley WB. Id reaction. In: Consultations in Dermatology. Saunders; 1972:262-267.
- Sharquie KE, Noaimi AA, Flayih RA. Clinical and histopathological findings in patients with follicular dermatoses: all skin diseases starts in the hair follicles as new hypothesis. Am J Clin Res Rev. 2020;4:17.
- Kasteler JS, Petersen MJ, Vance JE, et al. Circulating activated T lymphocytes in autoeczematization. Arch Dermatol. 1992;128:795-798.
- González-Amaro R, Baranda L, Abud-Mendoza C, et al. Autoeczematization is associated with abnormal immune recognition of autologous skin antigens. J Am Acad Dermatol. 1993;28:56-60.
- Cunningham MJ, Zone JJ, Petersen MJ, et al. Circulating activated (DR-positive) T lymphocytes in a patient with autoeczematization. J Am Acad Dermatol. 1986;14:1039-1041.
- Furue M, Ulzii D, Vu YH, et al. Pathogenesis of atopic dermatitis: current paradigm. Iran J Immunol. 2019;16:97-107.
- Uchi H, Terao H, Koga T, et al. Cytokines and chemokines in the epidermis. J Dermatol Sci. 2000;24(suppl 1):S29-S38.
- Bos JD, Kapsenberg ML. The skin immune system: progress in cutaneous biology. Immunol Today. 1993;14:75-78.
- Young AW Jr. Dynamics of autosensitization dermatitis; a clinical and microscopic concept of autoeczematization. AMA Arch Derm. 1958;77:495-502.
- Brenner S, Wolf R, Landau M. Scabid: an unusual id reaction to scabies. Int J Dermatol. 1993;32:128-129.
- Yamany T, Schwartz RA. Infectious eczematoid dermatitis: a comprehensive review. J Eur Acad Dermatol Venereol. 2015;29:203-208.
- Wang X, Li L, Shi X, et al. Itching and its related factors in subtypes of eczema: a cross-sectional multicenter study in tertiary hospitals of China. Sci Rep. 2018;8:10754.
- Price A, Tavazoie M, Meehan SA, et al. Id reaction associated with red tattoo ink. Cutis. 2018;102:E32-E34.
- Ilkit M, Durdu M, Karaks¸ M. Cutaneous id reactions: a comprehensive review of clinical manifestations, epidemiology, etiology, and management. Crit Rev Microbiol. 2012;38:191-202.
- Kaner SR. Dermatitis venenata of the feet with a generalized “id” reaction. J Am Podiatry Assoc. 1970;60:199-204.
- Jordan L, Jackson NA, Carter-Snell B, et al. Pustular tinea id reaction. Cutis. 2019;103:E3-E4.
- Crum N, Hardaway C, Graham B. Development of an idlike reaction during treatment for acute pulmonary histoplasmosis: a new cutaneous manifestation in histoplasmosis. J Am Acad Dermatol. 2003;48(2 suppl):S5-S6.
- Chirac A, Brzezinski P, Chiriac AE, et al. Autosensitisation (autoeczematisation) reactions in a case of diaper dermatitis candidiasis. Niger Med J. 2014;55:274-275.
- Singh PY, Sinha P, Baveja S, et al. Immune-mediated tuberculous uveitis—a rare association with papulonecrotic tuberculid. Indian J Ophthalmol. 2019;67:1207-1209.
- Urso B, Georgesen C, Harp J. Papulonecrotic tuberculid secondary to Mycobacterium avium complex. Cutis. 2019;104:E11-E13.
- Choudhri SH, Magro CM, Crowson AN, et al. An id reaction to Mycobacterium leprae: first documented case. Cutis. 1994;54:282-286.
- Park JW, Jeong GJ, Seo SJ, et al. Pseudomonas toe web infection and autosensitisation dermatitis: diagnostic and therapeutic challenge. Int Wound J. 2020;17:1543-1544. doi:10.1111/iwj.13386
- Netchiporouk E, Cohen BA. Recognizing and managing eczematous id reactions to molluscum contagiosum virus in children. Pediatrics. 2012;129:E1072-E1075.
- Aurelian L, Ono F, Burnett J. Herpes simplex virus (HSV)-associated erythema multiforme (HAEM): a viral disease with an autoimmune component. Dermatol Online J. 2003;9:1.
- Rocamora V, Romaní J, Puig L, et al. Id reaction to molluscum contagiosum. Pediatr Dermatol. 1996;13:349-350.
- Yes¸ilova Y, Özbilgin A, Turan E, et al. Clinical exacerbation developing during treatment of cutaneous leishmaniasis: an id reaction? Turkiye Parazitol Derg. 2014;38:281-282.
- Connor CJ, Selby JC, Wanat KA. Severe pediculosis capitus: a case of “crusted lice” with autoeczematization. Dermatol Online J. 2016;22:13030/qt7c91z913.
- Shelley WB. The autoimmune mechanism in clinical dermatology. Arch Dermatol. 1962;86:27-34.
- Bosworth A, Hull PR. Disseminated eczema following radiotherapy: a case report. J Cutan Med Surg. 2018;22:353-355.
- Lowther C, Miedler JD, Cockerell CJ. Id-like reaction to BCG therapy for bladder cancer. Cutis. 2013;91:145-151.
- Huerth KA, Glick PL, Glick ZR. Cutaneous id reaction after using cyanoacrylate for wound closure. Cutis. 2020;105:E11-E13.
- Amini S, Burdick AE, Janniger CK. Dyshidrotic eczema (pompholyx). Updated April 22, 2020. Accessed August 23, 2021. https://emedicine.medscape.com/article/1122527-overview
- Sundaresan S, Migden MR, Silapunt S. Stasis dermatitis: pathophysiology, evaluation, and management. Am J Clin Dermatol. 2017;18:383-390.
- Hughes JDM, Pratt MD. Allergic contact dermatitis and autoeczematization to proctosedyl® cream and proctomyxin® cream. Case Rep Dermatol. 2018;10:238-246.
- Bains SN, Nash P, Fonacier L. Irritant contact dermatitis. Clin Rev Allergy Immunol. 2019;56:99-109.
- Novak-Bilic´ G, Vucˇic´ M, Japundžic´ I, et al. Irritant and allergic contact dermatitis—skin lesion characteristics. Acta Clin Croat. 2018;57:713-720.
- Nassau S, Fonacier L. Allergic contact dermatitis. Med Clin North Am. 2020;104:61-76.
- Lewis DJ, Schlichte MJ, Dao H Jr. Atypical disseminated herpes zoster: management guidelines in immunocompromised patients. Cutis. 2017;100:321-330.
- Nedorost S, White S, Rowland DY, et al. Development and implementation of an order set to improve value of care for patients with severe stasis dermatitis. J Am Acad Dermatol. 2019;80:815-817.
Autoeczematization (AE), or id reaction, is a disseminated eczematous reaction that occurs days or weeks after exposure to a primary stimulus, resulting from a release of antigen(s). Whitfield1 first described AE in 1921, when he postulated that the id reaction was due to sensitization of the skin after a primary stimulus. He called it “a form of auto-intoxication derived from changes in the patient’s own tissues.”1 The exact prevalence of id reactions is unknown; one study showed that 17% of patients with dermatophyte infections developed an id reaction, typically tinea pedis linked with vesicles on the palms.2 Tinea capitis is one of the most common causes of AE in children, which is frequently misdiagnosed as a drug reaction. Approximately 37% of patients diagnosed with stasis dermatitis develop an id reaction (Figure 1). A history of contact dermatitis is common in patients presenting with AE.2-6

Pathophysiology of Id Reactions
An abnormal immune response against autologous skin antigens may be responsible for the development of AE. Shelley5 postulated that hair follicles play an important role in id reactions, as Sharquie et al6 recently emphasized for many skin disorders. The pathogenesis of AE is uncertain, but circulating T lymphocytes play a role in this reaction. Normally, T cells are activated by a release of antigens after a primary exposure to a stimulus. However, overactivation of these T cells induces autoimmune reactions such as AE.7 Activated T lymphocytes express HLA-DR and IL-2 receptor, markers elevated in the peripheral blood of patients undergoing id reactions. After treatment, the levels of activated T lymphocytes decline. An increase in the number of CD25+ T cells and a decrease in the number of suppressor T cells in the blood may occur during an id reaction.7-9 Keratinocytes produce proinflammatory cytokines, such as thymic stromal erythropoietin, IL-25, and IL-33, that activate T cells.10-12 Therefore, the most likely pathogenesis of an id reaction is that T lymphocytes are activated at the primary reaction site due to proinflammatory cytokines released by keratinocytes. These activated T cells then travel systemically via hematogenous dissemination. The spread of activated T lymphocytes produces an eczematous reaction at secondary locations distant to the primary site.9
Clinical and Histopathological Features of Id Reactions
Clinically, AE is first evident as a vesicular dissemination that groups to form papules or nummular patches and usually is present on the legs, feet, arms, and/or trunk (Figure 2). The primary dermatitis is localized to the area that was the site of contact to the offending stimuli. This localized eczematous eruption begins with an acute or subacute onset. It has the appearance of small crusted vesicles with erythema (Figure 1). The first sign of AE is vesicles presenting near the primary site on flexural surfaces or on the hands and feet. A classic example is tinea pedis linked with vesicles on the palms and sides of the fingers, resembling dyshidrotic eczema. Sites of prior cutaneous trauma, such as dermatoses, scars, and burns, are common locations for early AE. In later stages, vesicles disseminate to the legs, arms, and trunk, where they group to form papules and nummular patches in a symmetrical pattern.5,13-15 These lesions may be extremely pruritic. The pruritus may be so intense that it interrupts daily activities and disrupts the ability to fall or stay asleep.16

Histologically, biopsy specimens show psoriasiform spongiotic dermatitis with mononuclear cells contained in the vesicles. Interstitial edema and perivascular lymphohistiocytic infiltrates are evident. Eosinophils also may be present. This pattern is not unique toid reactions.17-19 Although AE is a reaction pattern that may be due to a fungal or bacterial infection, the etiologic agent is not evident microscopically within the eczema itself.
Etiology of Id Reactions
Id reactions most commonly occur from either stasis dermatitis or tinea pedis, although a wide variety of other causes should be considered. Evaluation of the primary site rather than the id reaction may identify an infectious or parasitic agent. Sometimes the AE reaction is specifically named: dermatophytid with dermatophytosis, bacterid with a bacterial infectious process, and tuberculid with tuberculosis. Similarly, there may be reactions to underlying candidiasis, sporotrichosis, histoplasmosis, and other fungal infections that can cause a cutaneous id reaction.18,20-22Mycobacterium species, Pseudomonas, Staphylococcus, and Streptococcus are bacterial causes of AE.15,23-26 Viral infections that can cause an id reaction are herpes simplex virus and molluscum contagiosum.27-29 Scabies, leishmaniasis, and pediculosis capitis are parasitic infections that may be etiologic.14,30,31 In addition, noninfectious stimuli besides stasis dermatitis that can produce id reactions include medications, topical creams, tattoo ink, sutures, radiotherapy, and dyshidrotic eczema. The primary reaction to these agents is a localized dermatitis followed by the immunological response that induces a secondary reaction distant from the primary site.17,18,32-38
Differential Diagnoses
Differential diagnoses include other types of eczema and some vesicular eruptions. Irritant contact dermatitis is another dermatosis that presents as a widespread vesicular eruption due to repetitive exposure to toxic irritants. The rash is erythematous with pustules, blisters, and crusts. It is only found in areas directly exposed to irritants, as opposed to AE, which spreads to areas distant to the primary reaction site. Irritant contact dermatitis presents with more of a burning sensation, whereas AE is more pruritic.39,40 Allergic contact dermatitis presents with erythematous vesicles and papules and sometimes with bullae. There is edema and crust formation, which often can spread past the point of contact in later stages. Similar to AE, there is intense pruritus. However, allergic contact dermatitis most commonly is caused by exposure to metals, cosmetics, and fragrances, whereas infectious agents and stasis dermatitis are the most common causes of AE.40,41 It may be challenging to distinguish AE from other causes of widespread eczematous dissemination. Vesicular eruptions sometimes require distinction from AE, including herpetic infections, insect bite reactions, and drug eruptions.18,42
Treatment
The underlying condition should be treated to mitigate the inflammatory response causing the id reaction. If not skillfully orchestrated, the id reaction can reoccur. For infectious causes of AE, an antifungal, antibacterial, antiviral, or antiparasitic should be given. If stasis dermatitis is responsible for the id reaction, compression stockings and leg elevation are indicated. The id reaction itself is treated with systemic or topical corticosteroids and wet compresses if acute. The goal of these treatments is to reduce patient discomfort caused by the inflammation and pruritus.18,43
Conclusion
Id reactions are an unusual phenomenon that commonly occurs after fungal skin infections and stasis dermatitis. T lymphocytes and keratinocytes may play a key role in this reaction, with newer research further delineating the process and possibly providing enhanced treatment options. Therapy focuses on treating the underlying condition, supplemented with corticosteroids for the autoeczema.
Autoeczematization (AE), or id reaction, is a disseminated eczematous reaction that occurs days or weeks after exposure to a primary stimulus, resulting from a release of antigen(s). Whitfield1 first described AE in 1921, when he postulated that the id reaction was due to sensitization of the skin after a primary stimulus. He called it “a form of auto-intoxication derived from changes in the patient’s own tissues.”1 The exact prevalence of id reactions is unknown; one study showed that 17% of patients with dermatophyte infections developed an id reaction, typically tinea pedis linked with vesicles on the palms.2 Tinea capitis is one of the most common causes of AE in children, which is frequently misdiagnosed as a drug reaction. Approximately 37% of patients diagnosed with stasis dermatitis develop an id reaction (Figure 1). A history of contact dermatitis is common in patients presenting with AE.2-6

Pathophysiology of Id Reactions
An abnormal immune response against autologous skin antigens may be responsible for the development of AE. Shelley5 postulated that hair follicles play an important role in id reactions, as Sharquie et al6 recently emphasized for many skin disorders. The pathogenesis of AE is uncertain, but circulating T lymphocytes play a role in this reaction. Normally, T cells are activated by a release of antigens after a primary exposure to a stimulus. However, overactivation of these T cells induces autoimmune reactions such as AE.7 Activated T lymphocytes express HLA-DR and IL-2 receptor, markers elevated in the peripheral blood of patients undergoing id reactions. After treatment, the levels of activated T lymphocytes decline. An increase in the number of CD25+ T cells and a decrease in the number of suppressor T cells in the blood may occur during an id reaction.7-9 Keratinocytes produce proinflammatory cytokines, such as thymic stromal erythropoietin, IL-25, and IL-33, that activate T cells.10-12 Therefore, the most likely pathogenesis of an id reaction is that T lymphocytes are activated at the primary reaction site due to proinflammatory cytokines released by keratinocytes. These activated T cells then travel systemically via hematogenous dissemination. The spread of activated T lymphocytes produces an eczematous reaction at secondary locations distant to the primary site.9
Clinical and Histopathological Features of Id Reactions
Clinically, AE is first evident as a vesicular dissemination that groups to form papules or nummular patches and usually is present on the legs, feet, arms, and/or trunk (Figure 2). The primary dermatitis is localized to the area that was the site of contact to the offending stimuli. This localized eczematous eruption begins with an acute or subacute onset. It has the appearance of small crusted vesicles with erythema (Figure 1). The first sign of AE is vesicles presenting near the primary site on flexural surfaces or on the hands and feet. A classic example is tinea pedis linked with vesicles on the palms and sides of the fingers, resembling dyshidrotic eczema. Sites of prior cutaneous trauma, such as dermatoses, scars, and burns, are common locations for early AE. In later stages, vesicles disseminate to the legs, arms, and trunk, where they group to form papules and nummular patches in a symmetrical pattern.5,13-15 These lesions may be extremely pruritic. The pruritus may be so intense that it interrupts daily activities and disrupts the ability to fall or stay asleep.16

Histologically, biopsy specimens show psoriasiform spongiotic dermatitis with mononuclear cells contained in the vesicles. Interstitial edema and perivascular lymphohistiocytic infiltrates are evident. Eosinophils also may be present. This pattern is not unique toid reactions.17-19 Although AE is a reaction pattern that may be due to a fungal or bacterial infection, the etiologic agent is not evident microscopically within the eczema itself.
Etiology of Id Reactions
Id reactions most commonly occur from either stasis dermatitis or tinea pedis, although a wide variety of other causes should be considered. Evaluation of the primary site rather than the id reaction may identify an infectious or parasitic agent. Sometimes the AE reaction is specifically named: dermatophytid with dermatophytosis, bacterid with a bacterial infectious process, and tuberculid with tuberculosis. Similarly, there may be reactions to underlying candidiasis, sporotrichosis, histoplasmosis, and other fungal infections that can cause a cutaneous id reaction.18,20-22Mycobacterium species, Pseudomonas, Staphylococcus, and Streptococcus are bacterial causes of AE.15,23-26 Viral infections that can cause an id reaction are herpes simplex virus and molluscum contagiosum.27-29 Scabies, leishmaniasis, and pediculosis capitis are parasitic infections that may be etiologic.14,30,31 In addition, noninfectious stimuli besides stasis dermatitis that can produce id reactions include medications, topical creams, tattoo ink, sutures, radiotherapy, and dyshidrotic eczema. The primary reaction to these agents is a localized dermatitis followed by the immunological response that induces a secondary reaction distant from the primary site.17,18,32-38
Differential Diagnoses
Differential diagnoses include other types of eczema and some vesicular eruptions. Irritant contact dermatitis is another dermatosis that presents as a widespread vesicular eruption due to repetitive exposure to toxic irritants. The rash is erythematous with pustules, blisters, and crusts. It is only found in areas directly exposed to irritants, as opposed to AE, which spreads to areas distant to the primary reaction site. Irritant contact dermatitis presents with more of a burning sensation, whereas AE is more pruritic.39,40 Allergic contact dermatitis presents with erythematous vesicles and papules and sometimes with bullae. There is edema and crust formation, which often can spread past the point of contact in later stages. Similar to AE, there is intense pruritus. However, allergic contact dermatitis most commonly is caused by exposure to metals, cosmetics, and fragrances, whereas infectious agents and stasis dermatitis are the most common causes of AE.40,41 It may be challenging to distinguish AE from other causes of widespread eczematous dissemination. Vesicular eruptions sometimes require distinction from AE, including herpetic infections, insect bite reactions, and drug eruptions.18,42
Treatment
The underlying condition should be treated to mitigate the inflammatory response causing the id reaction. If not skillfully orchestrated, the id reaction can reoccur. For infectious causes of AE, an antifungal, antibacterial, antiviral, or antiparasitic should be given. If stasis dermatitis is responsible for the id reaction, compression stockings and leg elevation are indicated. The id reaction itself is treated with systemic or topical corticosteroids and wet compresses if acute. The goal of these treatments is to reduce patient discomfort caused by the inflammation and pruritus.18,43
Conclusion
Id reactions are an unusual phenomenon that commonly occurs after fungal skin infections and stasis dermatitis. T lymphocytes and keratinocytes may play a key role in this reaction, with newer research further delineating the process and possibly providing enhanced treatment options. Therapy focuses on treating the underlying condition, supplemented with corticosteroids for the autoeczema.
- Whitfield A. Lumleian Lectures on Some Points in the Aetiology of Skin Diseases. Delivered before the Royal College of Physicians of London on March 10th, 15th, and 17th, 1921. Lecture II. Lancet. 1921;2:122-127.
- Cheng N, Rucker Wright D, Cohen BA. Dermatophytid in tinea capitis: rarely reported common phenomenon with clinical implications. Pediatrics. 2011;128:E453-E457.
- Schrom KP, Kobs A, Nedorost S. Clinical psoriasiform dermatitis following dupilumab use for autoeczematization secondary to chronic stasis dermatitis. Cureus. 2020;12:e7831. doi:10.7759/cureus.7831
- Templeton HJ, Lunsford CJ, Allington HV. Autosensitization dermatitis; report of five cases and protocol of an experiment. Arch Derm Syphilol. 1949;59:68-77.
- Shelley WB. Id reaction. In: Consultations in Dermatology. Saunders; 1972:262-267.
- Sharquie KE, Noaimi AA, Flayih RA. Clinical and histopathological findings in patients with follicular dermatoses: all skin diseases starts in the hair follicles as new hypothesis. Am J Clin Res Rev. 2020;4:17.
- Kasteler JS, Petersen MJ, Vance JE, et al. Circulating activated T lymphocytes in autoeczematization. Arch Dermatol. 1992;128:795-798.
- González-Amaro R, Baranda L, Abud-Mendoza C, et al. Autoeczematization is associated with abnormal immune recognition of autologous skin antigens. J Am Acad Dermatol. 1993;28:56-60.
- Cunningham MJ, Zone JJ, Petersen MJ, et al. Circulating activated (DR-positive) T lymphocytes in a patient with autoeczematization. J Am Acad Dermatol. 1986;14:1039-1041.
- Furue M, Ulzii D, Vu YH, et al. Pathogenesis of atopic dermatitis: current paradigm. Iran J Immunol. 2019;16:97-107.
- Uchi H, Terao H, Koga T, et al. Cytokines and chemokines in the epidermis. J Dermatol Sci. 2000;24(suppl 1):S29-S38.
- Bos JD, Kapsenberg ML. The skin immune system: progress in cutaneous biology. Immunol Today. 1993;14:75-78.
- Young AW Jr. Dynamics of autosensitization dermatitis; a clinical and microscopic concept of autoeczematization. AMA Arch Derm. 1958;77:495-502.
- Brenner S, Wolf R, Landau M. Scabid: an unusual id reaction to scabies. Int J Dermatol. 1993;32:128-129.
- Yamany T, Schwartz RA. Infectious eczematoid dermatitis: a comprehensive review. J Eur Acad Dermatol Venereol. 2015;29:203-208.
- Wang X, Li L, Shi X, et al. Itching and its related factors in subtypes of eczema: a cross-sectional multicenter study in tertiary hospitals of China. Sci Rep. 2018;8:10754.
- Price A, Tavazoie M, Meehan SA, et al. Id reaction associated with red tattoo ink. Cutis. 2018;102:E32-E34.
- Ilkit M, Durdu M, Karaks¸ M. Cutaneous id reactions: a comprehensive review of clinical manifestations, epidemiology, etiology, and management. Crit Rev Microbiol. 2012;38:191-202.
- Kaner SR. Dermatitis venenata of the feet with a generalized “id” reaction. J Am Podiatry Assoc. 1970;60:199-204.
- Jordan L, Jackson NA, Carter-Snell B, et al. Pustular tinea id reaction. Cutis. 2019;103:E3-E4.
- Crum N, Hardaway C, Graham B. Development of an idlike reaction during treatment for acute pulmonary histoplasmosis: a new cutaneous manifestation in histoplasmosis. J Am Acad Dermatol. 2003;48(2 suppl):S5-S6.
- Chirac A, Brzezinski P, Chiriac AE, et al. Autosensitisation (autoeczematisation) reactions in a case of diaper dermatitis candidiasis. Niger Med J. 2014;55:274-275.
- Singh PY, Sinha P, Baveja S, et al. Immune-mediated tuberculous uveitis—a rare association with papulonecrotic tuberculid. Indian J Ophthalmol. 2019;67:1207-1209.
- Urso B, Georgesen C, Harp J. Papulonecrotic tuberculid secondary to Mycobacterium avium complex. Cutis. 2019;104:E11-E13.
- Choudhri SH, Magro CM, Crowson AN, et al. An id reaction to Mycobacterium leprae: first documented case. Cutis. 1994;54:282-286.
- Park JW, Jeong GJ, Seo SJ, et al. Pseudomonas toe web infection and autosensitisation dermatitis: diagnostic and therapeutic challenge. Int Wound J. 2020;17:1543-1544. doi:10.1111/iwj.13386
- Netchiporouk E, Cohen BA. Recognizing and managing eczematous id reactions to molluscum contagiosum virus in children. Pediatrics. 2012;129:E1072-E1075.
- Aurelian L, Ono F, Burnett J. Herpes simplex virus (HSV)-associated erythema multiforme (HAEM): a viral disease with an autoimmune component. Dermatol Online J. 2003;9:1.
- Rocamora V, Romaní J, Puig L, et al. Id reaction to molluscum contagiosum. Pediatr Dermatol. 1996;13:349-350.
- Yes¸ilova Y, Özbilgin A, Turan E, et al. Clinical exacerbation developing during treatment of cutaneous leishmaniasis: an id reaction? Turkiye Parazitol Derg. 2014;38:281-282.
- Connor CJ, Selby JC, Wanat KA. Severe pediculosis capitus: a case of “crusted lice” with autoeczematization. Dermatol Online J. 2016;22:13030/qt7c91z913.
- Shelley WB. The autoimmune mechanism in clinical dermatology. Arch Dermatol. 1962;86:27-34.
- Bosworth A, Hull PR. Disseminated eczema following radiotherapy: a case report. J Cutan Med Surg. 2018;22:353-355.
- Lowther C, Miedler JD, Cockerell CJ. Id-like reaction to BCG therapy for bladder cancer. Cutis. 2013;91:145-151.
- Huerth KA, Glick PL, Glick ZR. Cutaneous id reaction after using cyanoacrylate for wound closure. Cutis. 2020;105:E11-E13.
- Amini S, Burdick AE, Janniger CK. Dyshidrotic eczema (pompholyx). Updated April 22, 2020. Accessed August 23, 2021. https://emedicine.medscape.com/article/1122527-overview
- Sundaresan S, Migden MR, Silapunt S. Stasis dermatitis: pathophysiology, evaluation, and management. Am J Clin Dermatol. 2017;18:383-390.
- Hughes JDM, Pratt MD. Allergic contact dermatitis and autoeczematization to proctosedyl® cream and proctomyxin® cream. Case Rep Dermatol. 2018;10:238-246.
- Bains SN, Nash P, Fonacier L. Irritant contact dermatitis. Clin Rev Allergy Immunol. 2019;56:99-109.
- Novak-Bilic´ G, Vucˇic´ M, Japundžic´ I, et al. Irritant and allergic contact dermatitis—skin lesion characteristics. Acta Clin Croat. 2018;57:713-720.
- Nassau S, Fonacier L. Allergic contact dermatitis. Med Clin North Am. 2020;104:61-76.
- Lewis DJ, Schlichte MJ, Dao H Jr. Atypical disseminated herpes zoster: management guidelines in immunocompromised patients. Cutis. 2017;100:321-330.
- Nedorost S, White S, Rowland DY, et al. Development and implementation of an order set to improve value of care for patients with severe stasis dermatitis. J Am Acad Dermatol. 2019;80:815-817.
- Whitfield A. Lumleian Lectures on Some Points in the Aetiology of Skin Diseases. Delivered before the Royal College of Physicians of London on March 10th, 15th, and 17th, 1921. Lecture II. Lancet. 1921;2:122-127.
- Cheng N, Rucker Wright D, Cohen BA. Dermatophytid in tinea capitis: rarely reported common phenomenon with clinical implications. Pediatrics. 2011;128:E453-E457.
- Schrom KP, Kobs A, Nedorost S. Clinical psoriasiform dermatitis following dupilumab use for autoeczematization secondary to chronic stasis dermatitis. Cureus. 2020;12:e7831. doi:10.7759/cureus.7831
- Templeton HJ, Lunsford CJ, Allington HV. Autosensitization dermatitis; report of five cases and protocol of an experiment. Arch Derm Syphilol. 1949;59:68-77.
- Shelley WB. Id reaction. In: Consultations in Dermatology. Saunders; 1972:262-267.
- Sharquie KE, Noaimi AA, Flayih RA. Clinical and histopathological findings in patients with follicular dermatoses: all skin diseases starts in the hair follicles as new hypothesis. Am J Clin Res Rev. 2020;4:17.
- Kasteler JS, Petersen MJ, Vance JE, et al. Circulating activated T lymphocytes in autoeczematization. Arch Dermatol. 1992;128:795-798.
- González-Amaro R, Baranda L, Abud-Mendoza C, et al. Autoeczematization is associated with abnormal immune recognition of autologous skin antigens. J Am Acad Dermatol. 1993;28:56-60.
- Cunningham MJ, Zone JJ, Petersen MJ, et al. Circulating activated (DR-positive) T lymphocytes in a patient with autoeczematization. J Am Acad Dermatol. 1986;14:1039-1041.
- Furue M, Ulzii D, Vu YH, et al. Pathogenesis of atopic dermatitis: current paradigm. Iran J Immunol. 2019;16:97-107.
- Uchi H, Terao H, Koga T, et al. Cytokines and chemokines in the epidermis. J Dermatol Sci. 2000;24(suppl 1):S29-S38.
- Bos JD, Kapsenberg ML. The skin immune system: progress in cutaneous biology. Immunol Today. 1993;14:75-78.
- Young AW Jr. Dynamics of autosensitization dermatitis; a clinical and microscopic concept of autoeczematization. AMA Arch Derm. 1958;77:495-502.
- Brenner S, Wolf R, Landau M. Scabid: an unusual id reaction to scabies. Int J Dermatol. 1993;32:128-129.
- Yamany T, Schwartz RA. Infectious eczematoid dermatitis: a comprehensive review. J Eur Acad Dermatol Venereol. 2015;29:203-208.
- Wang X, Li L, Shi X, et al. Itching and its related factors in subtypes of eczema: a cross-sectional multicenter study in tertiary hospitals of China. Sci Rep. 2018;8:10754.
- Price A, Tavazoie M, Meehan SA, et al. Id reaction associated with red tattoo ink. Cutis. 2018;102:E32-E34.
- Ilkit M, Durdu M, Karaks¸ M. Cutaneous id reactions: a comprehensive review of clinical manifestations, epidemiology, etiology, and management. Crit Rev Microbiol. 2012;38:191-202.
- Kaner SR. Dermatitis venenata of the feet with a generalized “id” reaction. J Am Podiatry Assoc. 1970;60:199-204.
- Jordan L, Jackson NA, Carter-Snell B, et al. Pustular tinea id reaction. Cutis. 2019;103:E3-E4.
- Crum N, Hardaway C, Graham B. Development of an idlike reaction during treatment for acute pulmonary histoplasmosis: a new cutaneous manifestation in histoplasmosis. J Am Acad Dermatol. 2003;48(2 suppl):S5-S6.
- Chirac A, Brzezinski P, Chiriac AE, et al. Autosensitisation (autoeczematisation) reactions in a case of diaper dermatitis candidiasis. Niger Med J. 2014;55:274-275.
- Singh PY, Sinha P, Baveja S, et al. Immune-mediated tuberculous uveitis—a rare association with papulonecrotic tuberculid. Indian J Ophthalmol. 2019;67:1207-1209.
- Urso B, Georgesen C, Harp J. Papulonecrotic tuberculid secondary to Mycobacterium avium complex. Cutis. 2019;104:E11-E13.
- Choudhri SH, Magro CM, Crowson AN, et al. An id reaction to Mycobacterium leprae: first documented case. Cutis. 1994;54:282-286.
- Park JW, Jeong GJ, Seo SJ, et al. Pseudomonas toe web infection and autosensitisation dermatitis: diagnostic and therapeutic challenge. Int Wound J. 2020;17:1543-1544. doi:10.1111/iwj.13386
- Netchiporouk E, Cohen BA. Recognizing and managing eczematous id reactions to molluscum contagiosum virus in children. Pediatrics. 2012;129:E1072-E1075.
- Aurelian L, Ono F, Burnett J. Herpes simplex virus (HSV)-associated erythema multiforme (HAEM): a viral disease with an autoimmune component. Dermatol Online J. 2003;9:1.
- Rocamora V, Romaní J, Puig L, et al. Id reaction to molluscum contagiosum. Pediatr Dermatol. 1996;13:349-350.
- Yes¸ilova Y, Özbilgin A, Turan E, et al. Clinical exacerbation developing during treatment of cutaneous leishmaniasis: an id reaction? Turkiye Parazitol Derg. 2014;38:281-282.
- Connor CJ, Selby JC, Wanat KA. Severe pediculosis capitus: a case of “crusted lice” with autoeczematization. Dermatol Online J. 2016;22:13030/qt7c91z913.
- Shelley WB. The autoimmune mechanism in clinical dermatology. Arch Dermatol. 1962;86:27-34.
- Bosworth A, Hull PR. Disseminated eczema following radiotherapy: a case report. J Cutan Med Surg. 2018;22:353-355.
- Lowther C, Miedler JD, Cockerell CJ. Id-like reaction to BCG therapy for bladder cancer. Cutis. 2013;91:145-151.
- Huerth KA, Glick PL, Glick ZR. Cutaneous id reaction after using cyanoacrylate for wound closure. Cutis. 2020;105:E11-E13.
- Amini S, Burdick AE, Janniger CK. Dyshidrotic eczema (pompholyx). Updated April 22, 2020. Accessed August 23, 2021. https://emedicine.medscape.com/article/1122527-overview
- Sundaresan S, Migden MR, Silapunt S. Stasis dermatitis: pathophysiology, evaluation, and management. Am J Clin Dermatol. 2017;18:383-390.
- Hughes JDM, Pratt MD. Allergic contact dermatitis and autoeczematization to proctosedyl® cream and proctomyxin® cream. Case Rep Dermatol. 2018;10:238-246.
- Bains SN, Nash P, Fonacier L. Irritant contact dermatitis. Clin Rev Allergy Immunol. 2019;56:99-109.
- Novak-Bilic´ G, Vucˇic´ M, Japundžic´ I, et al. Irritant and allergic contact dermatitis—skin lesion characteristics. Acta Clin Croat. 2018;57:713-720.
- Nassau S, Fonacier L. Allergic contact dermatitis. Med Clin North Am. 2020;104:61-76.
- Lewis DJ, Schlichte MJ, Dao H Jr. Atypical disseminated herpes zoster: management guidelines in immunocompromised patients. Cutis. 2017;100:321-330.
- Nedorost S, White S, Rowland DY, et al. Development and implementation of an order set to improve value of care for patients with severe stasis dermatitis. J Am Acad Dermatol. 2019;80:815-817.
Practice Points
- Autoeczematization, or id reaction, is a disseminated reaction of the skin occurring at a site distant to a primary cutaneous infection or stimulus.
- T lymphocytes and keratinocytes are postulated to be involved in the pathogenesis of id reactions.
- Therapy includes treating the underlying pathology while providing topical corticosteroids for the autoeczematous lesions.
Atopic Dermatitis Oral Therapies: What Are Patients Learning on YouTube?
To the Editor:
Oral immunosuppressive therapies are prescribed for moderate to severe atopic dermatitis. Patients often consult YouTube to make informed decisions about these therapies. In the United States, most health-related online searches are initiated through a search engine, which frequently leads to social media sites such as YouTube. Recent studies have examined the reasons why users turn to the Internet for health-related information, indicating that users typically seek specific information regarding health concerns.1,2 Furthermore, social media platforms such as YouTube are a popular means of sharing health information with the public.3-5 Currently, YouTube has more than 1 billion registered users, and 30 million health-related videos are watched each day.6 Almost one-third of US consumers use YouTube, Facebook, and Twitter to obtain medical information.7 YouTube is a versatile tool because of its video-discovery mechanisms such as a keyword-based search engine, video-recommendation system, highlight feature for videos on home pages, and the capacity to embed YouTube videos on various web pages.8 Searchers use videos that are short, fast paced, emotion evoking, from credible sources, recently uploaded, and relevant to the searcher for aiding in health decisions.9 Furthermore, studies have demonstrated YouTube’s capacity to support a change in attitude and increase users’ knowledge. In fact, YouTube had higher impact on recall, attitudes, and behaviors when compared with written materials on other social media platforms, such as Facebook and Twitter.9 We conducted a cross-sectional study to examine the quality of YouTube videos on oral therapies for atopic dermatitis, such as cyclosporine, methotrexate, azathioprine, and mycophenolate mofetil.
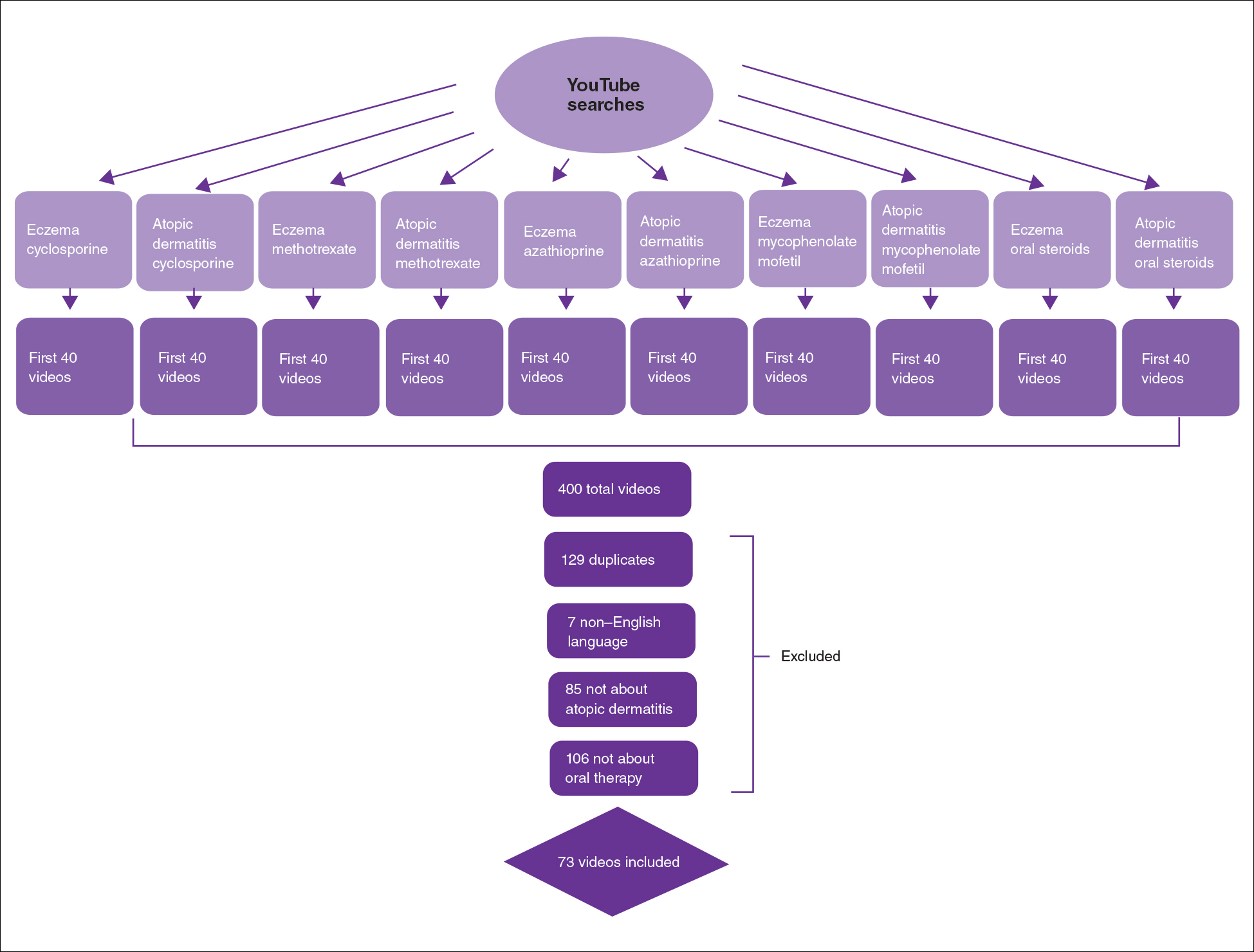
On April 23, 2020, we performed 8 searches using a private browser with default filters on YouTube (Figure). Injectables were not included in the analysis, as the YouTube experience on dupilumab previously has been investigated.10 The top 40 videos from each search were screened by 3 researchers. Duplicates, non–English-language videos, and videos that did not discuss atopic dermatitis or oral therapies were excluded, resulting in 73 videos included in this analysis. Testimonials generated by patients made up 39 of 73 (53.4%) videos. Health care professionals created 23 of 73 (31.5%) videos, and educators with financial interest created 11 of 73 (15.1%) videos. The dates of production for the videos spanned from 2008 to 2020.
The major topics addressed in the videos were symptomatic changes (63 [68.8% of all topics discussed]), adverse effects (52 [67.5%]), and quality-of-life changes (37 [48.1%]). Of the videos included, the majority (42/73 [57.5%]) contained a neutral tone about the medication, citing advantages and disadvantages with therapy, while 22 of 73 (30.1%) had an encouraging tone, and 9 of 73 (12.3%) had a discouraging tone. Regarding videos with positive tones, there were 17 videos on cyclosporine, 9 on azathioprine, 7 on methotrexate, 4 on oral steroids, and 2 on mycophenolate mofetil. Regarding videos with negative tones, there were 4 on cyclosporine, 3 on azathioprine, 2 on methotrexate, and 2 on mycophenolate mofetil.
Of the videos made with financial interest, the majority (28/34 [77.8%]) were more suitable for informing health care providers rather than patients, containing jargon as well as complex information on clinical trials, dosing, and mechanisms of action. From the videos discussing clinical recommendations, there were 9 of 73 (12.3%) Grade A recommendations (eg, citing evidence-based information and clinical trials) and 64 of 73 (87.7%) Grade B recommendations (eg, anecdotal information on patient experience). Thirty-seven of 73 (50.7%) videos were evidence based, and 36 of 73 (49.3%) were non–evidence based. Six videos were patient-oriented news broadcasts.
Patient-generated testimonials had the most views (mean, 9238.4) and highest interaction ratio (the sum of likes, dislikes, and comments divided by the number of views)(mean, 0.027), while health care provider–generated videos had fewer views (mean, 9218.7) and a lower interaction ratio (mean, 0.011). Financial-based videos had 4233.4 views on average, with an average interaction ratio of 0.014. Based on these results, biased, patient-generated content comprised greater than 50% of YouTube videos about oral therapies for atopic dermatitis and was quite likely to be engaged with by users. Thus, these patient testimonials have great potential to affect decision-making.
The high number of patient-generated videos about oral therapies was consistent with prior studies of YouTube videos about therapies for numerous conditions.11-13 Dermatologists should consider utilizing YouTube for providing evidence-based, patient-oriented information about novel therapeutics. They may consider collaborating with patients to assist with their creation of YouTube videos and directing patients to credible resources by the American Academy of Dermatology and Canadian Dermatology Association for decision-making.
Importantly, this analysis is limited by its lack of quality-assessment tools for video-based resources such as JAMA score and DISCERN score.14,15 However, these metrics have limited ability to evaluate audiovisual elements, indicating the need for novel tools to score their validity.
- Fox S, Duggan M. Health online 2013. January 15, 2013. Accessed August 15, 2021. https://www.pewresearch.org/internet/2013/01/15/health-online-2013/
- Ní Ríordáin R, McCreary C. Dental patients’ use of the Internet. Br Dent J. 2009;207:583-586, 575.
- Fergie G, Hilton S, Hunt K. Young adults’ experiences of seeking online information about diabetes and mental health in the age of social media. Health Expect. 2016;19:1324-1335.
- Antheunis ML, Tates K, Nieboer TE. Patients’ and health professionals’ use of social media in health care: motives, barriers and expectations. Patient Educ Couns. 2013;92:426-431.
- McGregor F, Somner JE, Bourne RR, et al. Social media use by patients with glaucoma: what can we learn? Ophthalmic Physiol Opt. 2014;34:46-52.
- YouTube Statistics—15 Amazing Stats for 2015. Published April 30, 2015. Accessed August 27, 2021. YouTube.com/watch?v=9ZLBSPzY7GQ
- Health Research Institute. Social media “likes” healthcare: from marketing to social business. April 2012. Accessed August 15, 2021. https://www.pwc.com/us/en/health-industries/health-research-institute/publications/pdf/health-care-social-media-report.pdf
- Zhou R, Khemmarat S, Gao L, et al. How YouTube videos are discovered and its impact on videos views. Multimed Tools Appl. 2016;75:6035-6058.
- Haslam K, Doucette H, Hachey S, et al. YouTube videos as health decision aids for the public: an integrative review. Can J Dent Hyg. 2019;53:53-66.
- Pithadia D, Reynolds K, Lee E, et al. Dupilumab for atopic dermatitis: what are patients learning on YouTube [published online ahead of print April 16,2020]? J Dermatolog Treat. doi: 10.1080/09546634.2020.1755418
- Tolu S, Yurdakul OV, Basaran B, et al. English-language videos on YouTube as a source of information on self-administer subcutaneous anti-tumour necrosis factor agent injections. Rheumatol Int. 2018;38:1285-1292.
- Reynolds KA, Pithadia DJ, Lee EB, et al. A cross-sectional study of YouTube videos about psoriasis biologics. Int J Dermatol. 2019;58:E61-E62.
- Kocyigit BF, Akaltun MS. Does YouTube provide high quality information? assessment of secukinumab videos. Rheumatol Int. 2019;39:1263-1268.
- Qi J, Trang T, Doong J, et al. Misinformation is prevalent in psoriasis-related YouTube videos. Dermatol Online J. 2016;22:13030/qt7qc9z2m5
- Gokcen HB, Gumussuyu G. A quality analysis of disc herniation videos on YouTube. World Neurosurg. 2019;124:E799-E804.
To the Editor:
Oral immunosuppressive therapies are prescribed for moderate to severe atopic dermatitis. Patients often consult YouTube to make informed decisions about these therapies. In the United States, most health-related online searches are initiated through a search engine, which frequently leads to social media sites such as YouTube. Recent studies have examined the reasons why users turn to the Internet for health-related information, indicating that users typically seek specific information regarding health concerns.1,2 Furthermore, social media platforms such as YouTube are a popular means of sharing health information with the public.3-5 Currently, YouTube has more than 1 billion registered users, and 30 million health-related videos are watched each day.6 Almost one-third of US consumers use YouTube, Facebook, and Twitter to obtain medical information.7 YouTube is a versatile tool because of its video-discovery mechanisms such as a keyword-based search engine, video-recommendation system, highlight feature for videos on home pages, and the capacity to embed YouTube videos on various web pages.8 Searchers use videos that are short, fast paced, emotion evoking, from credible sources, recently uploaded, and relevant to the searcher for aiding in health decisions.9 Furthermore, studies have demonstrated YouTube’s capacity to support a change in attitude and increase users’ knowledge. In fact, YouTube had higher impact on recall, attitudes, and behaviors when compared with written materials on other social media platforms, such as Facebook and Twitter.9 We conducted a cross-sectional study to examine the quality of YouTube videos on oral therapies for atopic dermatitis, such as cyclosporine, methotrexate, azathioprine, and mycophenolate mofetil.
On April 23, 2020, we performed 8 searches using a private browser with default filters on YouTube (Figure). Injectables were not included in the analysis, as the YouTube experience on dupilumab previously has been investigated.10 The top 40 videos from each search were screened by 3 researchers. Duplicates, non–English-language videos, and videos that did not discuss atopic dermatitis or oral therapies were excluded, resulting in 73 videos included in this analysis. Testimonials generated by patients made up 39 of 73 (53.4%) videos. Health care professionals created 23 of 73 (31.5%) videos, and educators with financial interest created 11 of 73 (15.1%) videos. The dates of production for the videos spanned from 2008 to 2020.

The major topics addressed in the videos were symptomatic changes (63 [68.8% of all topics discussed]), adverse effects (52 [67.5%]), and quality-of-life changes (37 [48.1%]). Of the videos included, the majority (42/73 [57.5%]) contained a neutral tone about the medication, citing advantages and disadvantages with therapy, while 22 of 73 (30.1%) had an encouraging tone, and 9 of 73 (12.3%) had a discouraging tone. Regarding videos with positive tones, there were 17 videos on cyclosporine, 9 on azathioprine, 7 on methotrexate, 4 on oral steroids, and 2 on mycophenolate mofetil. Regarding videos with negative tones, there were 4 on cyclosporine, 3 on azathioprine, 2 on methotrexate, and 2 on mycophenolate mofetil.
Of the videos made with financial interest, the majority (28/34 [77.8%]) were more suitable for informing health care providers rather than patients, containing jargon as well as complex information on clinical trials, dosing, and mechanisms of action. From the videos discussing clinical recommendations, there were 9 of 73 (12.3%) Grade A recommendations (eg, citing evidence-based information and clinical trials) and 64 of 73 (87.7%) Grade B recommendations (eg, anecdotal information on patient experience). Thirty-seven of 73 (50.7%) videos were evidence based, and 36 of 73 (49.3%) were non–evidence based. Six videos were patient-oriented news broadcasts.
Patient-generated testimonials had the most views (mean, 9238.4) and highest interaction ratio (the sum of likes, dislikes, and comments divided by the number of views)(mean, 0.027), while health care provider–generated videos had fewer views (mean, 9218.7) and a lower interaction ratio (mean, 0.011). Financial-based videos had 4233.4 views on average, with an average interaction ratio of 0.014. Based on these results, biased, patient-generated content comprised greater than 50% of YouTube videos about oral therapies for atopic dermatitis and was quite likely to be engaged with by users. Thus, these patient testimonials have great potential to affect decision-making.
The high number of patient-generated videos about oral therapies was consistent with prior studies of YouTube videos about therapies for numerous conditions.11-13 Dermatologists should consider utilizing YouTube for providing evidence-based, patient-oriented information about novel therapeutics. They may consider collaborating with patients to assist with their creation of YouTube videos and directing patients to credible resources by the American Academy of Dermatology and Canadian Dermatology Association for decision-making.
Importantly, this analysis is limited by its lack of quality-assessment tools for video-based resources such as JAMA score and DISCERN score.14,15 However, these metrics have limited ability to evaluate audiovisual elements, indicating the need for novel tools to score their validity.
To the Editor:
Oral immunosuppressive therapies are prescribed for moderate to severe atopic dermatitis. Patients often consult YouTube to make informed decisions about these therapies. In the United States, most health-related online searches are initiated through a search engine, which frequently leads to social media sites such as YouTube. Recent studies have examined the reasons why users turn to the Internet for health-related information, indicating that users typically seek specific information regarding health concerns.1,2 Furthermore, social media platforms such as YouTube are a popular means of sharing health information with the public.3-5 Currently, YouTube has more than 1 billion registered users, and 30 million health-related videos are watched each day.6 Almost one-third of US consumers use YouTube, Facebook, and Twitter to obtain medical information.7 YouTube is a versatile tool because of its video-discovery mechanisms such as a keyword-based search engine, video-recommendation system, highlight feature for videos on home pages, and the capacity to embed YouTube videos on various web pages.8 Searchers use videos that are short, fast paced, emotion evoking, from credible sources, recently uploaded, and relevant to the searcher for aiding in health decisions.9 Furthermore, studies have demonstrated YouTube’s capacity to support a change in attitude and increase users’ knowledge. In fact, YouTube had higher impact on recall, attitudes, and behaviors when compared with written materials on other social media platforms, such as Facebook and Twitter.9 We conducted a cross-sectional study to examine the quality of YouTube videos on oral therapies for atopic dermatitis, such as cyclosporine, methotrexate, azathioprine, and mycophenolate mofetil.
On April 23, 2020, we performed 8 searches using a private browser with default filters on YouTube (Figure). Injectables were not included in the analysis, as the YouTube experience on dupilumab previously has been investigated.10 The top 40 videos from each search were screened by 3 researchers. Duplicates, non–English-language videos, and videos that did not discuss atopic dermatitis or oral therapies were excluded, resulting in 73 videos included in this analysis. Testimonials generated by patients made up 39 of 73 (53.4%) videos. Health care professionals created 23 of 73 (31.5%) videos, and educators with financial interest created 11 of 73 (15.1%) videos. The dates of production for the videos spanned from 2008 to 2020.

The major topics addressed in the videos were symptomatic changes (63 [68.8% of all topics discussed]), adverse effects (52 [67.5%]), and quality-of-life changes (37 [48.1%]). Of the videos included, the majority (42/73 [57.5%]) contained a neutral tone about the medication, citing advantages and disadvantages with therapy, while 22 of 73 (30.1%) had an encouraging tone, and 9 of 73 (12.3%) had a discouraging tone. Regarding videos with positive tones, there were 17 videos on cyclosporine, 9 on azathioprine, 7 on methotrexate, 4 on oral steroids, and 2 on mycophenolate mofetil. Regarding videos with negative tones, there were 4 on cyclosporine, 3 on azathioprine, 2 on methotrexate, and 2 on mycophenolate mofetil.
Of the videos made with financial interest, the majority (28/34 [77.8%]) were more suitable for informing health care providers rather than patients, containing jargon as well as complex information on clinical trials, dosing, and mechanisms of action. From the videos discussing clinical recommendations, there were 9 of 73 (12.3%) Grade A recommendations (eg, citing evidence-based information and clinical trials) and 64 of 73 (87.7%) Grade B recommendations (eg, anecdotal information on patient experience). Thirty-seven of 73 (50.7%) videos were evidence based, and 36 of 73 (49.3%) were non–evidence based. Six videos were patient-oriented news broadcasts.
Patient-generated testimonials had the most views (mean, 9238.4) and highest interaction ratio (the sum of likes, dislikes, and comments divided by the number of views)(mean, 0.027), while health care provider–generated videos had fewer views (mean, 9218.7) and a lower interaction ratio (mean, 0.011). Financial-based videos had 4233.4 views on average, with an average interaction ratio of 0.014. Based on these results, biased, patient-generated content comprised greater than 50% of YouTube videos about oral therapies for atopic dermatitis and was quite likely to be engaged with by users. Thus, these patient testimonials have great potential to affect decision-making.
The high number of patient-generated videos about oral therapies was consistent with prior studies of YouTube videos about therapies for numerous conditions.11-13 Dermatologists should consider utilizing YouTube for providing evidence-based, patient-oriented information about novel therapeutics. They may consider collaborating with patients to assist with their creation of YouTube videos and directing patients to credible resources by the American Academy of Dermatology and Canadian Dermatology Association for decision-making.
Importantly, this analysis is limited by its lack of quality-assessment tools for video-based resources such as JAMA score and DISCERN score.14,15 However, these metrics have limited ability to evaluate audiovisual elements, indicating the need for novel tools to score their validity.
- Fox S, Duggan M. Health online 2013. January 15, 2013. Accessed August 15, 2021. https://www.pewresearch.org/internet/2013/01/15/health-online-2013/
- Ní Ríordáin R, McCreary C. Dental patients’ use of the Internet. Br Dent J. 2009;207:583-586, 575.
- Fergie G, Hilton S, Hunt K. Young adults’ experiences of seeking online information about diabetes and mental health in the age of social media. Health Expect. 2016;19:1324-1335.
- Antheunis ML, Tates K, Nieboer TE. Patients’ and health professionals’ use of social media in health care: motives, barriers and expectations. Patient Educ Couns. 2013;92:426-431.
- McGregor F, Somner JE, Bourne RR, et al. Social media use by patients with glaucoma: what can we learn? Ophthalmic Physiol Opt. 2014;34:46-52.
- YouTube Statistics—15 Amazing Stats for 2015. Published April 30, 2015. Accessed August 27, 2021. YouTube.com/watch?v=9ZLBSPzY7GQ
- Health Research Institute. Social media “likes” healthcare: from marketing to social business. April 2012. Accessed August 15, 2021. https://www.pwc.com/us/en/health-industries/health-research-institute/publications/pdf/health-care-social-media-report.pdf
- Zhou R, Khemmarat S, Gao L, et al. How YouTube videos are discovered and its impact on videos views. Multimed Tools Appl. 2016;75:6035-6058.
- Haslam K, Doucette H, Hachey S, et al. YouTube videos as health decision aids for the public: an integrative review. Can J Dent Hyg. 2019;53:53-66.
- Pithadia D, Reynolds K, Lee E, et al. Dupilumab for atopic dermatitis: what are patients learning on YouTube [published online ahead of print April 16,2020]? J Dermatolog Treat. doi: 10.1080/09546634.2020.1755418
- Tolu S, Yurdakul OV, Basaran B, et al. English-language videos on YouTube as a source of information on self-administer subcutaneous anti-tumour necrosis factor agent injections. Rheumatol Int. 2018;38:1285-1292.
- Reynolds KA, Pithadia DJ, Lee EB, et al. A cross-sectional study of YouTube videos about psoriasis biologics. Int J Dermatol. 2019;58:E61-E62.
- Kocyigit BF, Akaltun MS. Does YouTube provide high quality information? assessment of secukinumab videos. Rheumatol Int. 2019;39:1263-1268.
- Qi J, Trang T, Doong J, et al. Misinformation is prevalent in psoriasis-related YouTube videos. Dermatol Online J. 2016;22:13030/qt7qc9z2m5
- Gokcen HB, Gumussuyu G. A quality analysis of disc herniation videos on YouTube. World Neurosurg. 2019;124:E799-E804.
- Fox S, Duggan M. Health online 2013. January 15, 2013. Accessed August 15, 2021. https://www.pewresearch.org/internet/2013/01/15/health-online-2013/
- Ní Ríordáin R, McCreary C. Dental patients’ use of the Internet. Br Dent J. 2009;207:583-586, 575.
- Fergie G, Hilton S, Hunt K. Young adults’ experiences of seeking online information about diabetes and mental health in the age of social media. Health Expect. 2016;19:1324-1335.
- Antheunis ML, Tates K, Nieboer TE. Patients’ and health professionals’ use of social media in health care: motives, barriers and expectations. Patient Educ Couns. 2013;92:426-431.
- McGregor F, Somner JE, Bourne RR, et al. Social media use by patients with glaucoma: what can we learn? Ophthalmic Physiol Opt. 2014;34:46-52.
- YouTube Statistics—15 Amazing Stats for 2015. Published April 30, 2015. Accessed August 27, 2021. YouTube.com/watch?v=9ZLBSPzY7GQ
- Health Research Institute. Social media “likes” healthcare: from marketing to social business. April 2012. Accessed August 15, 2021. https://www.pwc.com/us/en/health-industries/health-research-institute/publications/pdf/health-care-social-media-report.pdf
- Zhou R, Khemmarat S, Gao L, et al. How YouTube videos are discovered and its impact on videos views. Multimed Tools Appl. 2016;75:6035-6058.
- Haslam K, Doucette H, Hachey S, et al. YouTube videos as health decision aids for the public: an integrative review. Can J Dent Hyg. 2019;53:53-66.
- Pithadia D, Reynolds K, Lee E, et al. Dupilumab for atopic dermatitis: what are patients learning on YouTube [published online ahead of print April 16,2020]? J Dermatolog Treat. doi: 10.1080/09546634.2020.1755418
- Tolu S, Yurdakul OV, Basaran B, et al. English-language videos on YouTube as a source of information on self-administer subcutaneous anti-tumour necrosis factor agent injections. Rheumatol Int. 2018;38:1285-1292.
- Reynolds KA, Pithadia DJ, Lee EB, et al. A cross-sectional study of YouTube videos about psoriasis biologics. Int J Dermatol. 2019;58:E61-E62.
- Kocyigit BF, Akaltun MS. Does YouTube provide high quality information? assessment of secukinumab videos. Rheumatol Int. 2019;39:1263-1268.
- Qi J, Trang T, Doong J, et al. Misinformation is prevalent in psoriasis-related YouTube videos. Dermatol Online J. 2016;22:13030/qt7qc9z2m5
- Gokcen HB, Gumussuyu G. A quality analysis of disc herniation videos on YouTube. World Neurosurg. 2019;124:E799-E804.
Practice Points
- Patient-based YouTube videos comprised the majority of videos on oral therapies for atopic dermatitis, with the greatest views and interaction ratio.
- Most YouTube videos on this topic contained a neutral tone and Grade B recommendations, thus meriting production of more evidence-based videos in collaboration with patients on the YouTube platform.
Atopic Dermatitis Topical Therapies: Study of YouTube Videos as a Source of Patient Information
To the Editor:
Atopic dermatitis (eczema) affects approximately 20% of children worldwide.1 In atopic dermatitis management, patient education is crucial for optimal outcomes.2 The COVID-19 pandemic has impacted patient-physician interactions. To ensure safety of patients and physicians, visits may have been canceled, postponed, or conducted virtually, leaving less time for discussion and questions.3 As a consequence, patients may seek information about atopic dermatitis from alternative sources, including YouTube videos. We performed a cross-sectional study to analyze YouTube videos about topical treatments for atopic dermatitis.
During the week of July 16, 2020, we performed 4 private browser YouTube searches with default filters using the following terms: eczema topicals, eczema topical treatments, atopic dermatitis topicals, and atopic dermatitis topical treatments. For video selection, we defined topical treatments as topical corticosteroids, topical calcineurin inhibitors, crisaborole, emollients, wet wraps, and any prospective treatment topically administered. For each of the 4 searches, 2 researchers (A.M. and A.T.) independently examined the top 75 videos, yielding a total of 300 videos. Of them, 98 videos were duplicates, 19 videos were not about atopic dermatitis, and 91 videos were not about topical treatments, leaving a total of 92 videos for analysis (Figure 1).
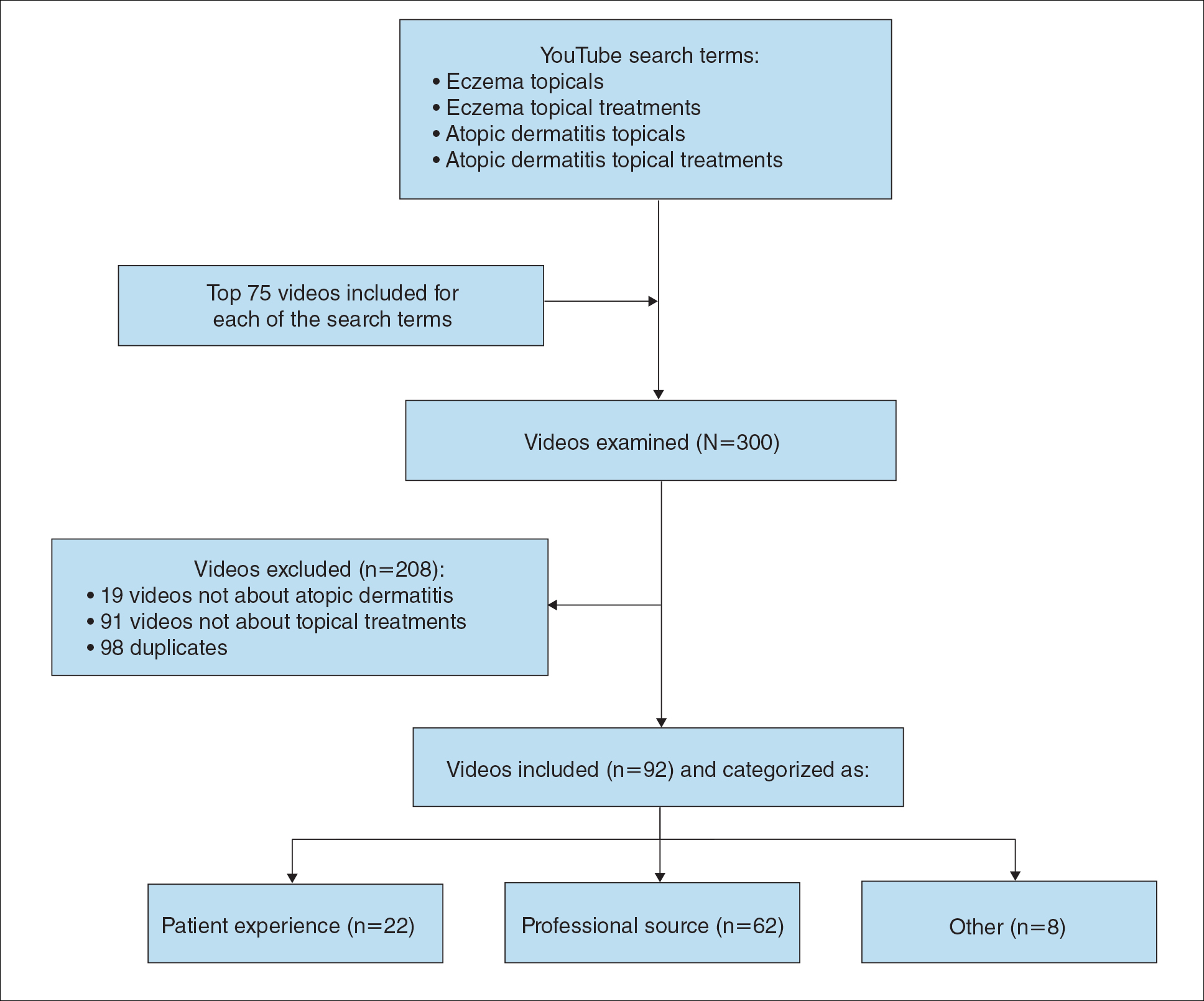
For the 92 included videos, the length; upload year; number of views, likes, dislikes, and comments; interaction ratio (IR)(the sum of likes, dislikes, and comments divided by the number of views); and video content were determined. The videos were placed into mutually exclusive categories as follows: (1) patient experience, defined as a video about patient perspective; (2) professional source, defined as a video featuring a physician, physician extender, pharmacist, or scientist, or produced by a formal organization; or (3) other. The DISCERN Instrument was used for grading the reliability and quality of the 92 included videos. This instrument consists of 16 questions with the responses rated on a scale of 1 to 5.4 For analysis of DISCERN scores, patient experience and other videos were grouped together as nonprofessional source videos. A 2-sample t-test was used to compare DISCERN scores between professional source and nonprofessional source videos.
Most videos were uploaded in 2017 (n=19), 2018 (n=23), and 2019 (n=25), but 20 were uploaded in 2012-2016 and 5 were uploaded in 2020. The 92 videos had a mean length of 8 minutes and 35 seconds (range, 30 seconds to 62 minutes and 23 seconds).
Patient experience videos accounted for 23.9% (n=22) of videos. These videos discussed topical steroid withdrawal (TSW)(n=16), instructions for making emollients (n=2), and treatment successes (n=4). Professional source videos represented 67.4% (n=62) of videos. Of them, 40.3% (n=25) were physician oriented, defined as having extensive medical terminology or qualifying for continuing medical education credit. Three (4.8%) of the professional source videos were sponsored by a drug company. Other constituted the remaining 8.7% (n=8) of videos. Patient experience videos had more views (median views [interquartile range], 6865 [10,307]) and higher engagement (median IR [interquartile range], 0.038 [0.022]) than professional source videos (views: median views [interquartile range], 1052.5 [10,610.5]; engagement: median IR [interquartile range], 0.006 [0.008]).
Although less popular, professional source videos had a significantly higher DISCERN overall quality rating score (question 16) compared to those categorized as nonprofessional source (3.92 vs 1.53; P<.001). In contrast, nonprofessional source videos scored significantly higher on the quality-of-life question (question 13) compared to professional source videos (3.90 vs 2.56; P<.001)(eTable). (Three professional source videos were removed from YouTube before DISCERN scores could be assigned.)
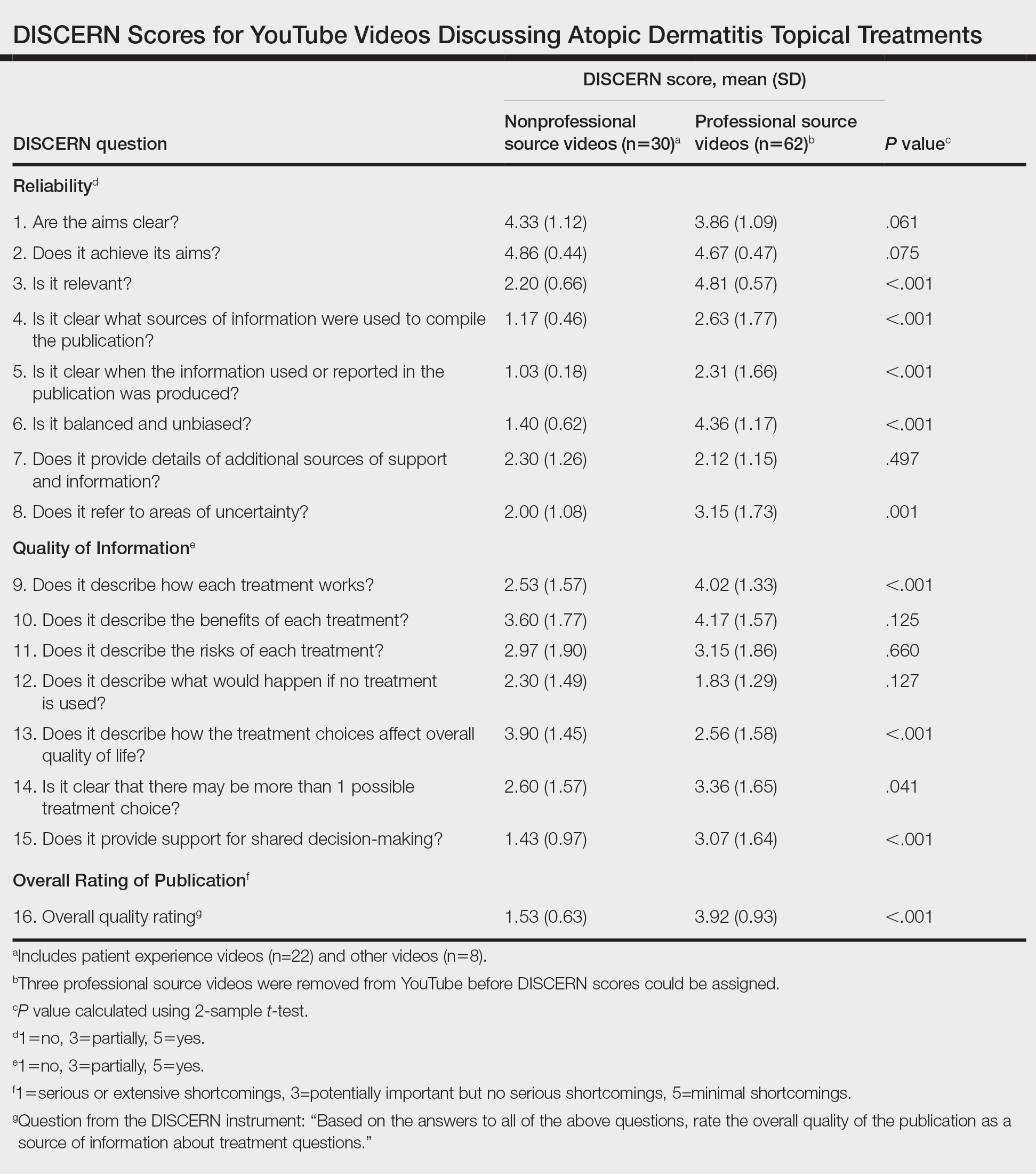
Notably, 20.7% (n=19) of the 92 videos discussed TSW, and most of them were patient experiences (n=16). Other categories included topical steroids excluding TSW (n=11), steroid phobia (n=2), topical calcineurin inhibitors (n=2), crisaborole (n=6), news broadcast (n=7), wet wraps (n=5), product advertisement (n=7), and research (n=11)(Figure 2). Interestingly, there were no videos focusing on the calcineurin inhibitor black box warning.

Similar to prior studies, our results indicate preference for patient-generated videos over videos produced by or including a professional source.5 Additionally, only 3 of 19 videos about TSW were from a professional source, increasing the potential for patient misconceptions about topical corticosteroids. Future studies should examine the educational impact of patient-generated videos as well as features that make the patient experience videos more desirable for viewing.
- Mueller SM, Hongler VNS, Jungo P, et al. Fiction, falsehoods, and few facts: cross-sectional study on the content-related quality of atopic eczema-related videos on YouTube. J Med Internet Res. 2020;22:e15599. doi:10.2196/15599
- Torres T, Ferreira EO, Gonçalo M, et al. Update on atopic dermatitis. Acta Med Port. 2019;32:606-613. doi:10.20344/amp.11963
- Vogler SA, Lightner AL. Rethinking how we care for our patients in a time of social distancing during the COVID-19 pandemic. Br J Surg. 2020;107:937-939. doi:10.1002/bjs.11636
- The DISCERN Instrument. discern online. Accessed January 22, 2021. http://www.discern.org.uk/discern_instrument.php
- Pithadia DJ, Reynolds KA, Lee EB, et al. Dupilumab for atopic dermatitis: what are patients learning on YouTube? [published online April 16, 2020]. J Dermatolog Treat. doi:10.1080/09546634.2020.1755418
To the Editor:
Atopic dermatitis (eczema) affects approximately 20% of children worldwide.1 In atopic dermatitis management, patient education is crucial for optimal outcomes.2 The COVID-19 pandemic has impacted patient-physician interactions. To ensure safety of patients and physicians, visits may have been canceled, postponed, or conducted virtually, leaving less time for discussion and questions.3 As a consequence, patients may seek information about atopic dermatitis from alternative sources, including YouTube videos. We performed a cross-sectional study to analyze YouTube videos about topical treatments for atopic dermatitis.
During the week of July 16, 2020, we performed 4 private browser YouTube searches with default filters using the following terms: eczema topicals, eczema topical treatments, atopic dermatitis topicals, and atopic dermatitis topical treatments. For video selection, we defined topical treatments as topical corticosteroids, topical calcineurin inhibitors, crisaborole, emollients, wet wraps, and any prospective treatment topically administered. For each of the 4 searches, 2 researchers (A.M. and A.T.) independently examined the top 75 videos, yielding a total of 300 videos. Of them, 98 videos were duplicates, 19 videos were not about atopic dermatitis, and 91 videos were not about topical treatments, leaving a total of 92 videos for analysis (Figure 1).

For the 92 included videos, the length; upload year; number of views, likes, dislikes, and comments; interaction ratio (IR)(the sum of likes, dislikes, and comments divided by the number of views); and video content were determined. The videos were placed into mutually exclusive categories as follows: (1) patient experience, defined as a video about patient perspective; (2) professional source, defined as a video featuring a physician, physician extender, pharmacist, or scientist, or produced by a formal organization; or (3) other. The DISCERN Instrument was used for grading the reliability and quality of the 92 included videos. This instrument consists of 16 questions with the responses rated on a scale of 1 to 5.4 For analysis of DISCERN scores, patient experience and other videos were grouped together as nonprofessional source videos. A 2-sample t-test was used to compare DISCERN scores between professional source and nonprofessional source videos.
Most videos were uploaded in 2017 (n=19), 2018 (n=23), and 2019 (n=25), but 20 were uploaded in 2012-2016 and 5 were uploaded in 2020. The 92 videos had a mean length of 8 minutes and 35 seconds (range, 30 seconds to 62 minutes and 23 seconds).
Patient experience videos accounted for 23.9% (n=22) of videos. These videos discussed topical steroid withdrawal (TSW)(n=16), instructions for making emollients (n=2), and treatment successes (n=4). Professional source videos represented 67.4% (n=62) of videos. Of them, 40.3% (n=25) were physician oriented, defined as having extensive medical terminology or qualifying for continuing medical education credit. Three (4.8%) of the professional source videos were sponsored by a drug company. Other constituted the remaining 8.7% (n=8) of videos. Patient experience videos had more views (median views [interquartile range], 6865 [10,307]) and higher engagement (median IR [interquartile range], 0.038 [0.022]) than professional source videos (views: median views [interquartile range], 1052.5 [10,610.5]; engagement: median IR [interquartile range], 0.006 [0.008]).
Although less popular, professional source videos had a significantly higher DISCERN overall quality rating score (question 16) compared to those categorized as nonprofessional source (3.92 vs 1.53; P<.001). In contrast, nonprofessional source videos scored significantly higher on the quality-of-life question (question 13) compared to professional source videos (3.90 vs 2.56; P<.001)(eTable). (Three professional source videos were removed from YouTube before DISCERN scores could be assigned.)

Notably, 20.7% (n=19) of the 92 videos discussed TSW, and most of them were patient experiences (n=16). Other categories included topical steroids excluding TSW (n=11), steroid phobia (n=2), topical calcineurin inhibitors (n=2), crisaborole (n=6), news broadcast (n=7), wet wraps (n=5), product advertisement (n=7), and research (n=11)(Figure 2). Interestingly, there were no videos focusing on the calcineurin inhibitor black box warning.

Similar to prior studies, our results indicate preference for patient-generated videos over videos produced by or including a professional source.5 Additionally, only 3 of 19 videos about TSW were from a professional source, increasing the potential for patient misconceptions about topical corticosteroids. Future studies should examine the educational impact of patient-generated videos as well as features that make the patient experience videos more desirable for viewing.
To the Editor:
Atopic dermatitis (eczema) affects approximately 20% of children worldwide.1 In atopic dermatitis management, patient education is crucial for optimal outcomes.2 The COVID-19 pandemic has impacted patient-physician interactions. To ensure safety of patients and physicians, visits may have been canceled, postponed, or conducted virtually, leaving less time for discussion and questions.3 As a consequence, patients may seek information about atopic dermatitis from alternative sources, including YouTube videos. We performed a cross-sectional study to analyze YouTube videos about topical treatments for atopic dermatitis.
During the week of July 16, 2020, we performed 4 private browser YouTube searches with default filters using the following terms: eczema topicals, eczema topical treatments, atopic dermatitis topicals, and atopic dermatitis topical treatments. For video selection, we defined topical treatments as topical corticosteroids, topical calcineurin inhibitors, crisaborole, emollients, wet wraps, and any prospective treatment topically administered. For each of the 4 searches, 2 researchers (A.M. and A.T.) independently examined the top 75 videos, yielding a total of 300 videos. Of them, 98 videos were duplicates, 19 videos were not about atopic dermatitis, and 91 videos were not about topical treatments, leaving a total of 92 videos for analysis (Figure 1).

For the 92 included videos, the length; upload year; number of views, likes, dislikes, and comments; interaction ratio (IR)(the sum of likes, dislikes, and comments divided by the number of views); and video content were determined. The videos were placed into mutually exclusive categories as follows: (1) patient experience, defined as a video about patient perspective; (2) professional source, defined as a video featuring a physician, physician extender, pharmacist, or scientist, or produced by a formal organization; or (3) other. The DISCERN Instrument was used for grading the reliability and quality of the 92 included videos. This instrument consists of 16 questions with the responses rated on a scale of 1 to 5.4 For analysis of DISCERN scores, patient experience and other videos were grouped together as nonprofessional source videos. A 2-sample t-test was used to compare DISCERN scores between professional source and nonprofessional source videos.
Most videos were uploaded in 2017 (n=19), 2018 (n=23), and 2019 (n=25), but 20 were uploaded in 2012-2016 and 5 were uploaded in 2020. The 92 videos had a mean length of 8 minutes and 35 seconds (range, 30 seconds to 62 minutes and 23 seconds).
Patient experience videos accounted for 23.9% (n=22) of videos. These videos discussed topical steroid withdrawal (TSW)(n=16), instructions for making emollients (n=2), and treatment successes (n=4). Professional source videos represented 67.4% (n=62) of videos. Of them, 40.3% (n=25) were physician oriented, defined as having extensive medical terminology or qualifying for continuing medical education credit. Three (4.8%) of the professional source videos were sponsored by a drug company. Other constituted the remaining 8.7% (n=8) of videos. Patient experience videos had more views (median views [interquartile range], 6865 [10,307]) and higher engagement (median IR [interquartile range], 0.038 [0.022]) than professional source videos (views: median views [interquartile range], 1052.5 [10,610.5]; engagement: median IR [interquartile range], 0.006 [0.008]).
Although less popular, professional source videos had a significantly higher DISCERN overall quality rating score (question 16) compared to those categorized as nonprofessional source (3.92 vs 1.53; P<.001). In contrast, nonprofessional source videos scored significantly higher on the quality-of-life question (question 13) compared to professional source videos (3.90 vs 2.56; P<.001)(eTable). (Three professional source videos were removed from YouTube before DISCERN scores could be assigned.)

Notably, 20.7% (n=19) of the 92 videos discussed TSW, and most of them were patient experiences (n=16). Other categories included topical steroids excluding TSW (n=11), steroid phobia (n=2), topical calcineurin inhibitors (n=2), crisaborole (n=6), news broadcast (n=7), wet wraps (n=5), product advertisement (n=7), and research (n=11)(Figure 2). Interestingly, there were no videos focusing on the calcineurin inhibitor black box warning.

Similar to prior studies, our results indicate preference for patient-generated videos over videos produced by or including a professional source.5 Additionally, only 3 of 19 videos about TSW were from a professional source, increasing the potential for patient misconceptions about topical corticosteroids. Future studies should examine the educational impact of patient-generated videos as well as features that make the patient experience videos more desirable for viewing.
- Mueller SM, Hongler VNS, Jungo P, et al. Fiction, falsehoods, and few facts: cross-sectional study on the content-related quality of atopic eczema-related videos on YouTube. J Med Internet Res. 2020;22:e15599. doi:10.2196/15599
- Torres T, Ferreira EO, Gonçalo M, et al. Update on atopic dermatitis. Acta Med Port. 2019;32:606-613. doi:10.20344/amp.11963
- Vogler SA, Lightner AL. Rethinking how we care for our patients in a time of social distancing during the COVID-19 pandemic. Br J Surg. 2020;107:937-939. doi:10.1002/bjs.11636
- The DISCERN Instrument. discern online. Accessed January 22, 2021. http://www.discern.org.uk/discern_instrument.php
- Pithadia DJ, Reynolds KA, Lee EB, et al. Dupilumab for atopic dermatitis: what are patients learning on YouTube? [published online April 16, 2020]. J Dermatolog Treat. doi:10.1080/09546634.2020.1755418
- Mueller SM, Hongler VNS, Jungo P, et al. Fiction, falsehoods, and few facts: cross-sectional study on the content-related quality of atopic eczema-related videos on YouTube. J Med Internet Res. 2020;22:e15599. doi:10.2196/15599
- Torres T, Ferreira EO, Gonçalo M, et al. Update on atopic dermatitis. Acta Med Port. 2019;32:606-613. doi:10.20344/amp.11963
- Vogler SA, Lightner AL. Rethinking how we care for our patients in a time of social distancing during the COVID-19 pandemic. Br J Surg. 2020;107:937-939. doi:10.1002/bjs.11636
- The DISCERN Instrument. discern online. Accessed January 22, 2021. http://www.discern.org.uk/discern_instrument.php
- Pithadia DJ, Reynolds KA, Lee EB, et al. Dupilumab for atopic dermatitis: what are patients learning on YouTube? [published online April 16, 2020]. J Dermatolog Treat. doi:10.1080/09546634.2020.1755418
Practice Points
- YouTube is a readily accessible resource for educating patients about topical treatments for atopic dermatitis.
- Although professional source videos comprised a larger percentage of the videos included within our study, patient experience videos had a higher number of views and engagement.
- Twenty-one percent (19/92) of the videos examined in our study discussed topical steroid withdrawal, and the majority of them were patient experience videos.
Atopic Dermatitis: Evolution and Revolution in Therapy
Atopic dermatitis (AD) is an incredibly common chronic skin disease, affecting up to 25% of children and 7% of adults in the United States.1,2 Despite the prevalence of this disease and its impact on patient quality of life, research and scholarly work in AD has been limited until recent years. A PubMed search of articles indexed for MEDLINE using the term atopic dermatitis showed that there were fewer than 500 articles published in 2000 and 965 in 2010; with our more recent acceleration in research, there were 2168 articles published in 2020 and more than 1300 published in just the first half of 2021 (through June). This new research includes insights into the pathogenesis of AD and study of the disease impact and comorbidities as well as an extensive amount of drug development and clinical trial work for new topical and systemic therapies.
New Agents to Treat AD
The 2016 approval of crisaborole,3 a phosphodiesterase 4 inhibitor, followed by the approval of dupilumab, an IL-4 and IL-13 pathway inhibitor and the first biologic agent approved for AD,4 ushered in a new age of therapy. We currently are awaiting the incorporation of a new set of topical nonsteroidal agents, oral Janus kinase (JAK) inhibitors, and new biologic agents for AD, several of which have completed phase 3 trials and extended safety evaluations. How these new drugs will impact our standard treatment across the spectrum of care for AD is not yet known.
The emergence of new systemic therapies is timely, as the most used systemic medications previously were oral corticosteroids, despite their use being advised against in standard practice guidelines. Other agents such as methotrexate, cyclosporine, azathioprine, and mycophenolate are discussed in the literature and AD treatment guidelines as being potentially useful, though absence of US Food and Drug Administration (FDA) approval and the need for frequent laboratory monitoring, as well as drug-specific side effects and an increased risk of infection, limit their use in the United States, especially in pediatric and adolescent populations.5
The approval of dupilumab as a systemic therapy—initially for adults and subsequently for teenagers (12–17 years of age) and then children (6–11 years of age)—has markedly influenced the standard of care for moderate to severe AD. This agent has been shown to have a considerable impact on disease severity and quality of life, with a good safety profile and the added benefit of not requiring continuous (or any) laboratory monitoring.6-8 Ongoing studies of dupilumab in children (ClinicalTrials.gov identifiers NCT02612454, NCT03346434), including those younger than 1 year,9 raise the question of how commonly this medication might be incorporated into care across the entire age spectrum of patients with AD. What standards will there be for assessment of severity, disease impact, and persistence to warrant use in younger ages? Will early treatment with novel systemic agents change the overall course of the disease and minimize the development of comorbidities? The answers to these questions remain to be seen.
JAK Inhibitors for AD
Additional novel therapeutics currently are undergoing studies for treatment of AD, most notably the oral JAK inhibitors upadacitinib,10 baricitinib,11 and abrocitinib.12 Each of these agents has completed phase 3 trials for AD. Two of these agents—upadacitinib and baricitinib—have prior FDA approval for use in other disease states. Of note, baricitinib is already approved for treatment of moderate to severe AD in adults in more than 40 countries13; however, the use of these agents in other diseases brings about concerns of malignancy, severe infection, and thrombosis. In the clinical trials for AD, many of these events have not been seen, but the number of patients treated is limited, and longer-term safety assessment is important.10,11
How will the oral JAK inhibitors be incorporated into care compared to biologic agents such as dupilumab? Tolerance and more serious potential adverse events are concerns, with nausea, headaches, and acneform eruptions being associated with some of the medications, in addition to potential issues with herpes simplex and zoster infections. However, oral JAK inhibitors have the benefit of not requiring injections, something that many patients may prefer, and data show that these drugs generally are associated with a rapid reduction in pruritus and, depending on the drug, very quick and profound effects on objective signs of AD.10-12 Two head-to-head studies have been completed comparing dupilumab to oral JAK inhibitors in adults: the JADE COMPARE trial examining dupilumab vs abrocitinib12 and the Heads UP trial comparing dupilumab vs upadacitinib.14 Compared to dupilumab, higher-dose abrocitinib showed more rapid responses, superiority in itch response, and similarity or superiority in other outcomes depending on the time point of the evaluation. Adverse event profiles differed; for example, abrocitinib was associated with more nausea, acneform eruptions, and herpes zoster, while dupilumab had higher rates of conjunctivitis.12 Upadacitinib, which was only studied at higher dosing (30 mg daily), showed superiority to dupilumab in itch response and in improvement in AD severity in multiple outcome measures; however, there were increases in serious infections, eczema herpeticum, herpes zoster, and laboratory-related adverse events.14 Dupilumab has the advantage of studies of extended use along with real-world experience, generally with excellent safety and tolerance other than injection-site reactions and conjunctivitis.8 Biologics targeting IL-13—tralokinumab and lebrikizumab—also are to be added to our armamentarium.15,16 The addition of these agents and JAK inhibitors as new systemic treatment options points to the quickly evolving future of AD treatment for patients with extensive disease.
New topical therapies in development provide even more treatment options. New nonsteroidal topicals include topical JAK inhibitors such as ruxolitinib17; tapinarof,18 an aryl hydrocarbon receptor modulator; and phosphodiesterase 4 inhibitors. These agents may be useful either as monotherapy, as studied, potentially without the regional limitations associated with stronger topical corticosteroids, but also should be useful in clinical practice as part of therapeutic regimens with other topical steroid and nonsteroidal agents.
The Microbiome and AD
In addition, research looking at topical microbes as specific interventions that may mediate the microbiome and inflammation of AD are intriguing. A recent phase 1 trial from the University of California San Diego19 indicated that topical bacteriotherapy directed at decreasing Staphylococcus aureus may provide an impact in AD. Observations by Kong et al20 showed that gram-negative microbiome differences are seen in AD patients compared to unaffected individuals, which has fueled studies showing that Roseomonas mucosa, a gram-negative skin commensal, when applied as a topical live biotherapeutic agent has improved disease severity in children and adults with AD.21 Although further studies are underway, these initial data suggest a role for microbiome-modifying therapies as AD treatment.
Chronic Hand Eczema
Chronic hand eczema (CHE), which has considerable overlap with AD in many patients, especially children and adolescents,22-24 is another area of interesting research. This high-prevalence condition is associated with allergic and irritant contact dermatitis24-26—conditions that are both considered alternative diagnoses for and exacerbators of AD27—and is a disease process currently being targeted for new therapies. Delgocitinib (NCT04872101, NCT04871711), the novel JAK inhibitor ARQ-252 (NCT04378569), among other topical agents, as well as systemic therapeutics such as gusacitinib (NCT03728504), are in active trials for CHE. Given CHE’s impact on quality of life28 and its overlap with AD, investigation into this disorder can help drive future AD research as well as lead to better knowledge and treatment of CHE.
Final Thoughts
Despite the promising results of these myriad new therapies in AD, there are many factors that influence how and when we use these drugs, including their approval status, FDA labeling, and the ability of patients to access and afford treatment. Additionally, continued study is needed to evaluate the long-term safety and extended efficacy of newer drugs, such as the oral JAK inhibitors. Despite these hurdles, the current landscape of research and development is rapidly evolving. Compared to the many years when only one main group of therapies was a reasonable option for patients, the future of AD treatment looks bright.
- Eichenfield LF, Tom WL, Chamlin SL, et al. Guidelines of care for the management of atopic dermatitis: section 1. diagnosis and assessment of atopic dermatitis. J Am Acad Dermatol. 2014;70:338-351. doi:10.1016/j.jaad.2013.10.010
- Chiesa Fuxench ZC, Block JK, Boguniewicz M, et al. Atopic dermatitis in America study: a cross-sectional study examining the prevalence and disease burden of atopic dermatitis in the US adult population. J Invest Dermatol. 2019;139:583-590. doi:10.1016/j.jid.2018.08.028
- FDA approves Eucrisa for eczema. News release. US Food and Drug Administration; December 14, 2016. Accessed August 16, 2021. https://www.fda.gov/news-events/press-announcements/fda-approves-eucrisa-eczema
- Gooderham MJ, Hong HC, Eshtiaghi P, et al. Dupilumab: a review of its use in the treatment of atopic dermatitis. J Am Acad Dermatol. 2018;78(3 suppl 1):S28-S36. doi:10.1016/j.jaad.2017.12.022
- Sidbury R, Davis DM, Cohen DE, et al. Guidelines of care for the management of atopic dermatitis: section 3. management and treatment with phototherapy and systemic agents. J Am Acad Dermatol. 2014;71:327-349. doi:10.1016/j.jaad.2014.03.030
- Paller AS, Siegfried EC, Thaçi D, et al. Efficacy and safety of dupilumab with concomitant topical corticosteroids in children 6 to 11 years old with severe atopic dermatitis: a randomized, double-blinded, placebo-controlled phase 3 trial. J Am Acad Dermatol. 2020;83:1282-1293. doi:10.1016/j.jaad.2020.06.054
- Simpson EL, Paller AS, Siegfried EC, et al. Efficacy and safety of dupilumab in adolescents with uncontrolled moderate to severe atopic dermatitis: a phase 3 randomized clinical trial. JAMA Dermatol. 2020;156:44-56. doi:10.1001/jamadermatol.2019.3336
- Deleuran M, Thaçi D, Beck LA, et al. Dupilumab shows long-term safety and efficacy in patients with moderate to severe atopic dermatitis enrolled in a phase 3 open-label extension study. J Am Acad Dermatol. 2020;82:377-388. doi:10.1016/j.jaad.2019.07.074
- Paller AS, Siegfried EC, Simpson EL, et al. A phase 2, open-label study of single-dose dupilumab in children aged 6 months to <6 years with severe uncontrolled atopic dermatitis: pharmacokinetics, safety and efficacy. J Eur Acad Dermatol Venereol. 2021;35:464-475. doi: 10.1111/jdv.16928
- Reich K, Teixeira HD, de Bruin-Weller M, et al. Safety and efficacy of upadacitinib in combination with topical corticosteroids in adolescents and adults with moderate-to-severe atopic dermatitis (AD Up): results from a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2021;397:2169-2181. doi:10.1016/S0140-6736(21)00589-4
- Simpson EL, Forman S, Silverberg JI, et al. Baricitinib in patients with moderate-to-severe atopic dermatitis: results from a randomized monotherapy phase 3 trial in the United States and Canada (BREEZE-AD5). J Am Acad Dermatol. 2021;85:62-70. doi:10.1016/j.jaad.2021.02.028
- Bieber T, Simpson EL, Silverberg JI, et al. Abrocitinib versus placebo or dupilumab for atopic dermatitis. N Engl J Med. 2021;384:1101-1112. doi:10.1056/NEJMoa2019380
- Lilly and Incyte provide update on supplemental New Drug Application for baricitinib for the treatment of moderate to severe atopic dermatitis. News release. Eli Lilly and Company; July 16, 2021. Accessed August 16, 2021. https://investor.lilly.com/news-releases/news-release-details/lilly-and-incyte-provide-update-supplemental new-drug
- Blauvelt A, Teixeira HD, Simpson EL, et al. Efficacy and safety of upadacitinib vs dupilumab in adults with moderate-to-severe atopic dermatitis: a randomized clinical trial [published online August 4, 2021]. JAMA Dermatol. doi:10.1001/jamadermatol.2021.3023
- Guttman-Yassky E, Blauvelt A, Eichenfield LF, et al. Efficacy and safety of lebrikizumab, a high-affinity interleukin 13 inhibitor, in adults with moderate to severe atopic dermatitis: a phase 2b randomized clinical trial. JAMA Dermatol. 2020;156:411-420. doi:10.1001/jamadermatol.2020.0079
- Silverberg JI, Toth D, Bieber T, et al. Tralokinumab plus topical corticosteroids for the treatment of moderate-to-severe atopic dermatitis: results from the double-blind, randomized, multicentre,placebo-controlled phase III ECZTRA 3 trial. Br J Dermatol. 2021;184:450-463. doi:10.1111/bjd.19573
- Papp K, Szepietowski JC, Kircik L, et al. Efficacy and safety of ruxolitinib cream for the treatment of atopic dermatitis: results from 2 phase 3, randomized, double-blind studies [published online May 4, 2021]. J Am Acad Dermatol. doi:10.1016/j.jaad.2021.04.085
- Paller AS, Stein Gold L, Soung J, et al. Efficacy and patient-reported outcomes from a phase 2b, randomized clinical trial of tapinarof cream for the treatment of adolescents and adults with atopic dermatitis. J Am Acad Dermatol. 2021;84:632-638. doi:10.1016/j.jaad.2020.05.135
- Nakatsuji, T, Hata TR, Tong Y, et al. Development of a human skin commensal microbe for bacteriotherapy of atopic dermatitis and use in a phase 1 randomized clinical trial [published online February 22, 2021]. Nat Med. 2021;27:700-709. doi:10.1038/s41591-021-01256-2
- Kong HH, Oh J, Deming C, et al. Temporal shifts in the skin microbiome associated with disease flares and treatment in children with atopic dermatitis. Genome Res. 2012;22:850-859. doi:10.1101/gr.131029.111
- Myles IA, Castillo CR, Barbian KD, et al. Therapeutic responses to Roseomonas mucosa in atopic dermatitis may involve lipid-mediated TNF-related epithelial repair. Sci Transl Med. 2020;12:eaaz8631. doi:10.1126/scitranslmed.aaz8631
- Mortz CG, Lauritsen JM, Bindslev-Jensen C, et al. Prevalence of atopic dermatitis, asthma, allergic rhinitis, and hand and contact dermatitis in adolescents. The Odense Adolescence Cohort Study on Atopic Diseases and Dermatitis. Br J Dermatol. 2001;144:523-532. doi:10.1046/j.1365-2133.2001.04078.x
- Grönhagen C, Lidén C, Wahlgren CF, et al. Hand eczema and atopic dermatitis in adolescents: a prospective cohort study from the BAMSE project. Br J Dermatol. 2015;173:1175-1182. doi:10.1111/bjd.14019
- Mortz CG, Lauritsen JM, Bindslev-Jensen C, et al. Contact allergy and allergic contact dermatitis in adolescents: prevalence measures and associations. The Odense Adolescence Cohort Study on Atopic Diseases and Dermatitis (TOACS). Acta Derm Venereol. 2002;82:352-358. doi:10.1080/000155502320624087
- Isaksson M, Olhardt S, Rådehed J, et al. Children with atopic dermatitis should always be patch-tested if they have hand or foot dermatitis. Acta Derm Venereol. 2015;95:583-586. doi:10.2340/00015555-1995
- Silverberg JI, Warshaw EM, Maibach HI, et al. Hand eczema in children referred for patch testing: North American Contact Dermatitis Group Data, 2000-2016. Br J Dermatol. 2021;185:185-194. doi:10.1111/bjd.19818
- Agner T, Elsner P. Hand eczema: epidemiology, prognosis and prevention. J Eur Acad Dermatol Venereol. 2020;34(suppl 1):4-12. doi:10.1111/jdv.16061
- Cazzaniga S, Ballmer-Weber BK, Gräni N, et al. Medical, psychological and socio-economic implications of chronic hand eczema: a cross-sectional study. J Eur Acad Dermatol Venereol. 2016;30:628-637. doi:10.1111/jdv.13479
Atopic dermatitis (AD) is an incredibly common chronic skin disease, affecting up to 25% of children and 7% of adults in the United States.1,2 Despite the prevalence of this disease and its impact on patient quality of life, research and scholarly work in AD has been limited until recent years. A PubMed search of articles indexed for MEDLINE using the term atopic dermatitis showed that there were fewer than 500 articles published in 2000 and 965 in 2010; with our more recent acceleration in research, there were 2168 articles published in 2020 and more than 1300 published in just the first half of 2021 (through June). This new research includes insights into the pathogenesis of AD and study of the disease impact and comorbidities as well as an extensive amount of drug development and clinical trial work for new topical and systemic therapies.
New Agents to Treat AD
The 2016 approval of crisaborole,3 a phosphodiesterase 4 inhibitor, followed by the approval of dupilumab, an IL-4 and IL-13 pathway inhibitor and the first biologic agent approved for AD,4 ushered in a new age of therapy. We currently are awaiting the incorporation of a new set of topical nonsteroidal agents, oral Janus kinase (JAK) inhibitors, and new biologic agents for AD, several of which have completed phase 3 trials and extended safety evaluations. How these new drugs will impact our standard treatment across the spectrum of care for AD is not yet known.
The emergence of new systemic therapies is timely, as the most used systemic medications previously were oral corticosteroids, despite their use being advised against in standard practice guidelines. Other agents such as methotrexate, cyclosporine, azathioprine, and mycophenolate are discussed in the literature and AD treatment guidelines as being potentially useful, though absence of US Food and Drug Administration (FDA) approval and the need for frequent laboratory monitoring, as well as drug-specific side effects and an increased risk of infection, limit their use in the United States, especially in pediatric and adolescent populations.5
The approval of dupilumab as a systemic therapy—initially for adults and subsequently for teenagers (12–17 years of age) and then children (6–11 years of age)—has markedly influenced the standard of care for moderate to severe AD. This agent has been shown to have a considerable impact on disease severity and quality of life, with a good safety profile and the added benefit of not requiring continuous (or any) laboratory monitoring.6-8 Ongoing studies of dupilumab in children (ClinicalTrials.gov identifiers NCT02612454, NCT03346434), including those younger than 1 year,9 raise the question of how commonly this medication might be incorporated into care across the entire age spectrum of patients with AD. What standards will there be for assessment of severity, disease impact, and persistence to warrant use in younger ages? Will early treatment with novel systemic agents change the overall course of the disease and minimize the development of comorbidities? The answers to these questions remain to be seen.
JAK Inhibitors for AD
Additional novel therapeutics currently are undergoing studies for treatment of AD, most notably the oral JAK inhibitors upadacitinib,10 baricitinib,11 and abrocitinib.12 Each of these agents has completed phase 3 trials for AD. Two of these agents—upadacitinib and baricitinib—have prior FDA approval for use in other disease states. Of note, baricitinib is already approved for treatment of moderate to severe AD in adults in more than 40 countries13; however, the use of these agents in other diseases brings about concerns of malignancy, severe infection, and thrombosis. In the clinical trials for AD, many of these events have not been seen, but the number of patients treated is limited, and longer-term safety assessment is important.10,11
How will the oral JAK inhibitors be incorporated into care compared to biologic agents such as dupilumab? Tolerance and more serious potential adverse events are concerns, with nausea, headaches, and acneform eruptions being associated with some of the medications, in addition to potential issues with herpes simplex and zoster infections. However, oral JAK inhibitors have the benefit of not requiring injections, something that many patients may prefer, and data show that these drugs generally are associated with a rapid reduction in pruritus and, depending on the drug, very quick and profound effects on objective signs of AD.10-12 Two head-to-head studies have been completed comparing dupilumab to oral JAK inhibitors in adults: the JADE COMPARE trial examining dupilumab vs abrocitinib12 and the Heads UP trial comparing dupilumab vs upadacitinib.14 Compared to dupilumab, higher-dose abrocitinib showed more rapid responses, superiority in itch response, and similarity or superiority in other outcomes depending on the time point of the evaluation. Adverse event profiles differed; for example, abrocitinib was associated with more nausea, acneform eruptions, and herpes zoster, while dupilumab had higher rates of conjunctivitis.12 Upadacitinib, which was only studied at higher dosing (30 mg daily), showed superiority to dupilumab in itch response and in improvement in AD severity in multiple outcome measures; however, there were increases in serious infections, eczema herpeticum, herpes zoster, and laboratory-related adverse events.14 Dupilumab has the advantage of studies of extended use along with real-world experience, generally with excellent safety and tolerance other than injection-site reactions and conjunctivitis.8 Biologics targeting IL-13—tralokinumab and lebrikizumab—also are to be added to our armamentarium.15,16 The addition of these agents and JAK inhibitors as new systemic treatment options points to the quickly evolving future of AD treatment for patients with extensive disease.
New topical therapies in development provide even more treatment options. New nonsteroidal topicals include topical JAK inhibitors such as ruxolitinib17; tapinarof,18 an aryl hydrocarbon receptor modulator; and phosphodiesterase 4 inhibitors. These agents may be useful either as monotherapy, as studied, potentially without the regional limitations associated with stronger topical corticosteroids, but also should be useful in clinical practice as part of therapeutic regimens with other topical steroid and nonsteroidal agents.
The Microbiome and AD
In addition, research looking at topical microbes as specific interventions that may mediate the microbiome and inflammation of AD are intriguing. A recent phase 1 trial from the University of California San Diego19 indicated that topical bacteriotherapy directed at decreasing Staphylococcus aureus may provide an impact in AD. Observations by Kong et al20 showed that gram-negative microbiome differences are seen in AD patients compared to unaffected individuals, which has fueled studies showing that Roseomonas mucosa, a gram-negative skin commensal, when applied as a topical live biotherapeutic agent has improved disease severity in children and adults with AD.21 Although further studies are underway, these initial data suggest a role for microbiome-modifying therapies as AD treatment.
Chronic Hand Eczema
Chronic hand eczema (CHE), which has considerable overlap with AD in many patients, especially children and adolescents,22-24 is another area of interesting research. This high-prevalence condition is associated with allergic and irritant contact dermatitis24-26—conditions that are both considered alternative diagnoses for and exacerbators of AD27—and is a disease process currently being targeted for new therapies. Delgocitinib (NCT04872101, NCT04871711), the novel JAK inhibitor ARQ-252 (NCT04378569), among other topical agents, as well as systemic therapeutics such as gusacitinib (NCT03728504), are in active trials for CHE. Given CHE’s impact on quality of life28 and its overlap with AD, investigation into this disorder can help drive future AD research as well as lead to better knowledge and treatment of CHE.
Final Thoughts
Despite the promising results of these myriad new therapies in AD, there are many factors that influence how and when we use these drugs, including their approval status, FDA labeling, and the ability of patients to access and afford treatment. Additionally, continued study is needed to evaluate the long-term safety and extended efficacy of newer drugs, such as the oral JAK inhibitors. Despite these hurdles, the current landscape of research and development is rapidly evolving. Compared to the many years when only one main group of therapies was a reasonable option for patients, the future of AD treatment looks bright.
Atopic dermatitis (AD) is an incredibly common chronic skin disease, affecting up to 25% of children and 7% of adults in the United States.1,2 Despite the prevalence of this disease and its impact on patient quality of life, research and scholarly work in AD has been limited until recent years. A PubMed search of articles indexed for MEDLINE using the term atopic dermatitis showed that there were fewer than 500 articles published in 2000 and 965 in 2010; with our more recent acceleration in research, there were 2168 articles published in 2020 and more than 1300 published in just the first half of 2021 (through June). This new research includes insights into the pathogenesis of AD and study of the disease impact and comorbidities as well as an extensive amount of drug development and clinical trial work for new topical and systemic therapies.
New Agents to Treat AD
The 2016 approval of crisaborole,3 a phosphodiesterase 4 inhibitor, followed by the approval of dupilumab, an IL-4 and IL-13 pathway inhibitor and the first biologic agent approved for AD,4 ushered in a new age of therapy. We currently are awaiting the incorporation of a new set of topical nonsteroidal agents, oral Janus kinase (JAK) inhibitors, and new biologic agents for AD, several of which have completed phase 3 trials and extended safety evaluations. How these new drugs will impact our standard treatment across the spectrum of care for AD is not yet known.
The emergence of new systemic therapies is timely, as the most used systemic medications previously were oral corticosteroids, despite their use being advised against in standard practice guidelines. Other agents such as methotrexate, cyclosporine, azathioprine, and mycophenolate are discussed in the literature and AD treatment guidelines as being potentially useful, though absence of US Food and Drug Administration (FDA) approval and the need for frequent laboratory monitoring, as well as drug-specific side effects and an increased risk of infection, limit their use in the United States, especially in pediatric and adolescent populations.5
The approval of dupilumab as a systemic therapy—initially for adults and subsequently for teenagers (12–17 years of age) and then children (6–11 years of age)—has markedly influenced the standard of care for moderate to severe AD. This agent has been shown to have a considerable impact on disease severity and quality of life, with a good safety profile and the added benefit of not requiring continuous (or any) laboratory monitoring.6-8 Ongoing studies of dupilumab in children (ClinicalTrials.gov identifiers NCT02612454, NCT03346434), including those younger than 1 year,9 raise the question of how commonly this medication might be incorporated into care across the entire age spectrum of patients with AD. What standards will there be for assessment of severity, disease impact, and persistence to warrant use in younger ages? Will early treatment with novel systemic agents change the overall course of the disease and minimize the development of comorbidities? The answers to these questions remain to be seen.
JAK Inhibitors for AD
Additional novel therapeutics currently are undergoing studies for treatment of AD, most notably the oral JAK inhibitors upadacitinib,10 baricitinib,11 and abrocitinib.12 Each of these agents has completed phase 3 trials for AD. Two of these agents—upadacitinib and baricitinib—have prior FDA approval for use in other disease states. Of note, baricitinib is already approved for treatment of moderate to severe AD in adults in more than 40 countries13; however, the use of these agents in other diseases brings about concerns of malignancy, severe infection, and thrombosis. In the clinical trials for AD, many of these events have not been seen, but the number of patients treated is limited, and longer-term safety assessment is important.10,11
How will the oral JAK inhibitors be incorporated into care compared to biologic agents such as dupilumab? Tolerance and more serious potential adverse events are concerns, with nausea, headaches, and acneform eruptions being associated with some of the medications, in addition to potential issues with herpes simplex and zoster infections. However, oral JAK inhibitors have the benefit of not requiring injections, something that many patients may prefer, and data show that these drugs generally are associated with a rapid reduction in pruritus and, depending on the drug, very quick and profound effects on objective signs of AD.10-12 Two head-to-head studies have been completed comparing dupilumab to oral JAK inhibitors in adults: the JADE COMPARE trial examining dupilumab vs abrocitinib12 and the Heads UP trial comparing dupilumab vs upadacitinib.14 Compared to dupilumab, higher-dose abrocitinib showed more rapid responses, superiority in itch response, and similarity or superiority in other outcomes depending on the time point of the evaluation. Adverse event profiles differed; for example, abrocitinib was associated with more nausea, acneform eruptions, and herpes zoster, while dupilumab had higher rates of conjunctivitis.12 Upadacitinib, which was only studied at higher dosing (30 mg daily), showed superiority to dupilumab in itch response and in improvement in AD severity in multiple outcome measures; however, there were increases in serious infections, eczema herpeticum, herpes zoster, and laboratory-related adverse events.14 Dupilumab has the advantage of studies of extended use along with real-world experience, generally with excellent safety and tolerance other than injection-site reactions and conjunctivitis.8 Biologics targeting IL-13—tralokinumab and lebrikizumab—also are to be added to our armamentarium.15,16 The addition of these agents and JAK inhibitors as new systemic treatment options points to the quickly evolving future of AD treatment for patients with extensive disease.
New topical therapies in development provide even more treatment options. New nonsteroidal topicals include topical JAK inhibitors such as ruxolitinib17; tapinarof,18 an aryl hydrocarbon receptor modulator; and phosphodiesterase 4 inhibitors. These agents may be useful either as monotherapy, as studied, potentially without the regional limitations associated with stronger topical corticosteroids, but also should be useful in clinical practice as part of therapeutic regimens with other topical steroid and nonsteroidal agents.
The Microbiome and AD
In addition, research looking at topical microbes as specific interventions that may mediate the microbiome and inflammation of AD are intriguing. A recent phase 1 trial from the University of California San Diego19 indicated that topical bacteriotherapy directed at decreasing Staphylococcus aureus may provide an impact in AD. Observations by Kong et al20 showed that gram-negative microbiome differences are seen in AD patients compared to unaffected individuals, which has fueled studies showing that Roseomonas mucosa, a gram-negative skin commensal, when applied as a topical live biotherapeutic agent has improved disease severity in children and adults with AD.21 Although further studies are underway, these initial data suggest a role for microbiome-modifying therapies as AD treatment.
Chronic Hand Eczema
Chronic hand eczema (CHE), which has considerable overlap with AD in many patients, especially children and adolescents,22-24 is another area of interesting research. This high-prevalence condition is associated with allergic and irritant contact dermatitis24-26—conditions that are both considered alternative diagnoses for and exacerbators of AD27—and is a disease process currently being targeted for new therapies. Delgocitinib (NCT04872101, NCT04871711), the novel JAK inhibitor ARQ-252 (NCT04378569), among other topical agents, as well as systemic therapeutics such as gusacitinib (NCT03728504), are in active trials for CHE. Given CHE’s impact on quality of life28 and its overlap with AD, investigation into this disorder can help drive future AD research as well as lead to better knowledge and treatment of CHE.
Final Thoughts
Despite the promising results of these myriad new therapies in AD, there are many factors that influence how and when we use these drugs, including their approval status, FDA labeling, and the ability of patients to access and afford treatment. Additionally, continued study is needed to evaluate the long-term safety and extended efficacy of newer drugs, such as the oral JAK inhibitors. Despite these hurdles, the current landscape of research and development is rapidly evolving. Compared to the many years when only one main group of therapies was a reasonable option for patients, the future of AD treatment looks bright.
- Eichenfield LF, Tom WL, Chamlin SL, et al. Guidelines of care for the management of atopic dermatitis: section 1. diagnosis and assessment of atopic dermatitis. J Am Acad Dermatol. 2014;70:338-351. doi:10.1016/j.jaad.2013.10.010
- Chiesa Fuxench ZC, Block JK, Boguniewicz M, et al. Atopic dermatitis in America study: a cross-sectional study examining the prevalence and disease burden of atopic dermatitis in the US adult population. J Invest Dermatol. 2019;139:583-590. doi:10.1016/j.jid.2018.08.028
- FDA approves Eucrisa for eczema. News release. US Food and Drug Administration; December 14, 2016. Accessed August 16, 2021. https://www.fda.gov/news-events/press-announcements/fda-approves-eucrisa-eczema
- Gooderham MJ, Hong HC, Eshtiaghi P, et al. Dupilumab: a review of its use in the treatment of atopic dermatitis. J Am Acad Dermatol. 2018;78(3 suppl 1):S28-S36. doi:10.1016/j.jaad.2017.12.022
- Sidbury R, Davis DM, Cohen DE, et al. Guidelines of care for the management of atopic dermatitis: section 3. management and treatment with phototherapy and systemic agents. J Am Acad Dermatol. 2014;71:327-349. doi:10.1016/j.jaad.2014.03.030
- Paller AS, Siegfried EC, Thaçi D, et al. Efficacy and safety of dupilumab with concomitant topical corticosteroids in children 6 to 11 years old with severe atopic dermatitis: a randomized, double-blinded, placebo-controlled phase 3 trial. J Am Acad Dermatol. 2020;83:1282-1293. doi:10.1016/j.jaad.2020.06.054
- Simpson EL, Paller AS, Siegfried EC, et al. Efficacy and safety of dupilumab in adolescents with uncontrolled moderate to severe atopic dermatitis: a phase 3 randomized clinical trial. JAMA Dermatol. 2020;156:44-56. doi:10.1001/jamadermatol.2019.3336
- Deleuran M, Thaçi D, Beck LA, et al. Dupilumab shows long-term safety and efficacy in patients with moderate to severe atopic dermatitis enrolled in a phase 3 open-label extension study. J Am Acad Dermatol. 2020;82:377-388. doi:10.1016/j.jaad.2019.07.074
- Paller AS, Siegfried EC, Simpson EL, et al. A phase 2, open-label study of single-dose dupilumab in children aged 6 months to <6 years with severe uncontrolled atopic dermatitis: pharmacokinetics, safety and efficacy. J Eur Acad Dermatol Venereol. 2021;35:464-475. doi: 10.1111/jdv.16928
- Reich K, Teixeira HD, de Bruin-Weller M, et al. Safety and efficacy of upadacitinib in combination with topical corticosteroids in adolescents and adults with moderate-to-severe atopic dermatitis (AD Up): results from a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2021;397:2169-2181. doi:10.1016/S0140-6736(21)00589-4
- Simpson EL, Forman S, Silverberg JI, et al. Baricitinib in patients with moderate-to-severe atopic dermatitis: results from a randomized monotherapy phase 3 trial in the United States and Canada (BREEZE-AD5). J Am Acad Dermatol. 2021;85:62-70. doi:10.1016/j.jaad.2021.02.028
- Bieber T, Simpson EL, Silverberg JI, et al. Abrocitinib versus placebo or dupilumab for atopic dermatitis. N Engl J Med. 2021;384:1101-1112. doi:10.1056/NEJMoa2019380
- Lilly and Incyte provide update on supplemental New Drug Application for baricitinib for the treatment of moderate to severe atopic dermatitis. News release. Eli Lilly and Company; July 16, 2021. Accessed August 16, 2021. https://investor.lilly.com/news-releases/news-release-details/lilly-and-incyte-provide-update-supplemental new-drug
- Blauvelt A, Teixeira HD, Simpson EL, et al. Efficacy and safety of upadacitinib vs dupilumab in adults with moderate-to-severe atopic dermatitis: a randomized clinical trial [published online August 4, 2021]. JAMA Dermatol. doi:10.1001/jamadermatol.2021.3023
- Guttman-Yassky E, Blauvelt A, Eichenfield LF, et al. Efficacy and safety of lebrikizumab, a high-affinity interleukin 13 inhibitor, in adults with moderate to severe atopic dermatitis: a phase 2b randomized clinical trial. JAMA Dermatol. 2020;156:411-420. doi:10.1001/jamadermatol.2020.0079
- Silverberg JI, Toth D, Bieber T, et al. Tralokinumab plus topical corticosteroids for the treatment of moderate-to-severe atopic dermatitis: results from the double-blind, randomized, multicentre,placebo-controlled phase III ECZTRA 3 trial. Br J Dermatol. 2021;184:450-463. doi:10.1111/bjd.19573
- Papp K, Szepietowski JC, Kircik L, et al. Efficacy and safety of ruxolitinib cream for the treatment of atopic dermatitis: results from 2 phase 3, randomized, double-blind studies [published online May 4, 2021]. J Am Acad Dermatol. doi:10.1016/j.jaad.2021.04.085
- Paller AS, Stein Gold L, Soung J, et al. Efficacy and patient-reported outcomes from a phase 2b, randomized clinical trial of tapinarof cream for the treatment of adolescents and adults with atopic dermatitis. J Am Acad Dermatol. 2021;84:632-638. doi:10.1016/j.jaad.2020.05.135
- Nakatsuji, T, Hata TR, Tong Y, et al. Development of a human skin commensal microbe for bacteriotherapy of atopic dermatitis and use in a phase 1 randomized clinical trial [published online February 22, 2021]. Nat Med. 2021;27:700-709. doi:10.1038/s41591-021-01256-2
- Kong HH, Oh J, Deming C, et al. Temporal shifts in the skin microbiome associated with disease flares and treatment in children with atopic dermatitis. Genome Res. 2012;22:850-859. doi:10.1101/gr.131029.111
- Myles IA, Castillo CR, Barbian KD, et al. Therapeutic responses to Roseomonas mucosa in atopic dermatitis may involve lipid-mediated TNF-related epithelial repair. Sci Transl Med. 2020;12:eaaz8631. doi:10.1126/scitranslmed.aaz8631
- Mortz CG, Lauritsen JM, Bindslev-Jensen C, et al. Prevalence of atopic dermatitis, asthma, allergic rhinitis, and hand and contact dermatitis in adolescents. The Odense Adolescence Cohort Study on Atopic Diseases and Dermatitis. Br J Dermatol. 2001;144:523-532. doi:10.1046/j.1365-2133.2001.04078.x
- Grönhagen C, Lidén C, Wahlgren CF, et al. Hand eczema and atopic dermatitis in adolescents: a prospective cohort study from the BAMSE project. Br J Dermatol. 2015;173:1175-1182. doi:10.1111/bjd.14019
- Mortz CG, Lauritsen JM, Bindslev-Jensen C, et al. Contact allergy and allergic contact dermatitis in adolescents: prevalence measures and associations. The Odense Adolescence Cohort Study on Atopic Diseases and Dermatitis (TOACS). Acta Derm Venereol. 2002;82:352-358. doi:10.1080/000155502320624087
- Isaksson M, Olhardt S, Rådehed J, et al. Children with atopic dermatitis should always be patch-tested if they have hand or foot dermatitis. Acta Derm Venereol. 2015;95:583-586. doi:10.2340/00015555-1995
- Silverberg JI, Warshaw EM, Maibach HI, et al. Hand eczema in children referred for patch testing: North American Contact Dermatitis Group Data, 2000-2016. Br J Dermatol. 2021;185:185-194. doi:10.1111/bjd.19818
- Agner T, Elsner P. Hand eczema: epidemiology, prognosis and prevention. J Eur Acad Dermatol Venereol. 2020;34(suppl 1):4-12. doi:10.1111/jdv.16061
- Cazzaniga S, Ballmer-Weber BK, Gräni N, et al. Medical, psychological and socio-economic implications of chronic hand eczema: a cross-sectional study. J Eur Acad Dermatol Venereol. 2016;30:628-637. doi:10.1111/jdv.13479
- Eichenfield LF, Tom WL, Chamlin SL, et al. Guidelines of care for the management of atopic dermatitis: section 1. diagnosis and assessment of atopic dermatitis. J Am Acad Dermatol. 2014;70:338-351. doi:10.1016/j.jaad.2013.10.010
- Chiesa Fuxench ZC, Block JK, Boguniewicz M, et al. Atopic dermatitis in America study: a cross-sectional study examining the prevalence and disease burden of atopic dermatitis in the US adult population. J Invest Dermatol. 2019;139:583-590. doi:10.1016/j.jid.2018.08.028
- FDA approves Eucrisa for eczema. News release. US Food and Drug Administration; December 14, 2016. Accessed August 16, 2021. https://www.fda.gov/news-events/press-announcements/fda-approves-eucrisa-eczema
- Gooderham MJ, Hong HC, Eshtiaghi P, et al. Dupilumab: a review of its use in the treatment of atopic dermatitis. J Am Acad Dermatol. 2018;78(3 suppl 1):S28-S36. doi:10.1016/j.jaad.2017.12.022
- Sidbury R, Davis DM, Cohen DE, et al. Guidelines of care for the management of atopic dermatitis: section 3. management and treatment with phototherapy and systemic agents. J Am Acad Dermatol. 2014;71:327-349. doi:10.1016/j.jaad.2014.03.030
- Paller AS, Siegfried EC, Thaçi D, et al. Efficacy and safety of dupilumab with concomitant topical corticosteroids in children 6 to 11 years old with severe atopic dermatitis: a randomized, double-blinded, placebo-controlled phase 3 trial. J Am Acad Dermatol. 2020;83:1282-1293. doi:10.1016/j.jaad.2020.06.054
- Simpson EL, Paller AS, Siegfried EC, et al. Efficacy and safety of dupilumab in adolescents with uncontrolled moderate to severe atopic dermatitis: a phase 3 randomized clinical trial. JAMA Dermatol. 2020;156:44-56. doi:10.1001/jamadermatol.2019.3336
- Deleuran M, Thaçi D, Beck LA, et al. Dupilumab shows long-term safety and efficacy in patients with moderate to severe atopic dermatitis enrolled in a phase 3 open-label extension study. J Am Acad Dermatol. 2020;82:377-388. doi:10.1016/j.jaad.2019.07.074
- Paller AS, Siegfried EC, Simpson EL, et al. A phase 2, open-label study of single-dose dupilumab in children aged 6 months to <6 years with severe uncontrolled atopic dermatitis: pharmacokinetics, safety and efficacy. J Eur Acad Dermatol Venereol. 2021;35:464-475. doi: 10.1111/jdv.16928
- Reich K, Teixeira HD, de Bruin-Weller M, et al. Safety and efficacy of upadacitinib in combination with topical corticosteroids in adolescents and adults with moderate-to-severe atopic dermatitis (AD Up): results from a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2021;397:2169-2181. doi:10.1016/S0140-6736(21)00589-4
- Simpson EL, Forman S, Silverberg JI, et al. Baricitinib in patients with moderate-to-severe atopic dermatitis: results from a randomized monotherapy phase 3 trial in the United States and Canada (BREEZE-AD5). J Am Acad Dermatol. 2021;85:62-70. doi:10.1016/j.jaad.2021.02.028
- Bieber T, Simpson EL, Silverberg JI, et al. Abrocitinib versus placebo or dupilumab for atopic dermatitis. N Engl J Med. 2021;384:1101-1112. doi:10.1056/NEJMoa2019380
- Lilly and Incyte provide update on supplemental New Drug Application for baricitinib for the treatment of moderate to severe atopic dermatitis. News release. Eli Lilly and Company; July 16, 2021. Accessed August 16, 2021. https://investor.lilly.com/news-releases/news-release-details/lilly-and-incyte-provide-update-supplemental new-drug
- Blauvelt A, Teixeira HD, Simpson EL, et al. Efficacy and safety of upadacitinib vs dupilumab in adults with moderate-to-severe atopic dermatitis: a randomized clinical trial [published online August 4, 2021]. JAMA Dermatol. doi:10.1001/jamadermatol.2021.3023
- Guttman-Yassky E, Blauvelt A, Eichenfield LF, et al. Efficacy and safety of lebrikizumab, a high-affinity interleukin 13 inhibitor, in adults with moderate to severe atopic dermatitis: a phase 2b randomized clinical trial. JAMA Dermatol. 2020;156:411-420. doi:10.1001/jamadermatol.2020.0079
- Silverberg JI, Toth D, Bieber T, et al. Tralokinumab plus topical corticosteroids for the treatment of moderate-to-severe atopic dermatitis: results from the double-blind, randomized, multicentre,placebo-controlled phase III ECZTRA 3 trial. Br J Dermatol. 2021;184:450-463. doi:10.1111/bjd.19573
- Papp K, Szepietowski JC, Kircik L, et al. Efficacy and safety of ruxolitinib cream for the treatment of atopic dermatitis: results from 2 phase 3, randomized, double-blind studies [published online May 4, 2021]. J Am Acad Dermatol. doi:10.1016/j.jaad.2021.04.085
- Paller AS, Stein Gold L, Soung J, et al. Efficacy and patient-reported outcomes from a phase 2b, randomized clinical trial of tapinarof cream for the treatment of adolescents and adults with atopic dermatitis. J Am Acad Dermatol. 2021;84:632-638. doi:10.1016/j.jaad.2020.05.135
- Nakatsuji, T, Hata TR, Tong Y, et al. Development of a human skin commensal microbe for bacteriotherapy of atopic dermatitis and use in a phase 1 randomized clinical trial [published online February 22, 2021]. Nat Med. 2021;27:700-709. doi:10.1038/s41591-021-01256-2
- Kong HH, Oh J, Deming C, et al. Temporal shifts in the skin microbiome associated with disease flares and treatment in children with atopic dermatitis. Genome Res. 2012;22:850-859. doi:10.1101/gr.131029.111
- Myles IA, Castillo CR, Barbian KD, et al. Therapeutic responses to Roseomonas mucosa in atopic dermatitis may involve lipid-mediated TNF-related epithelial repair. Sci Transl Med. 2020;12:eaaz8631. doi:10.1126/scitranslmed.aaz8631
- Mortz CG, Lauritsen JM, Bindslev-Jensen C, et al. Prevalence of atopic dermatitis, asthma, allergic rhinitis, and hand and contact dermatitis in adolescents. The Odense Adolescence Cohort Study on Atopic Diseases and Dermatitis. Br J Dermatol. 2001;144:523-532. doi:10.1046/j.1365-2133.2001.04078.x
- Grönhagen C, Lidén C, Wahlgren CF, et al. Hand eczema and atopic dermatitis in adolescents: a prospective cohort study from the BAMSE project. Br J Dermatol. 2015;173:1175-1182. doi:10.1111/bjd.14019
- Mortz CG, Lauritsen JM, Bindslev-Jensen C, et al. Contact allergy and allergic contact dermatitis in adolescents: prevalence measures and associations. The Odense Adolescence Cohort Study on Atopic Diseases and Dermatitis (TOACS). Acta Derm Venereol. 2002;82:352-358. doi:10.1080/000155502320624087
- Isaksson M, Olhardt S, Rådehed J, et al. Children with atopic dermatitis should always be patch-tested if they have hand or foot dermatitis. Acta Derm Venereol. 2015;95:583-586. doi:10.2340/00015555-1995
- Silverberg JI, Warshaw EM, Maibach HI, et al. Hand eczema in children referred for patch testing: North American Contact Dermatitis Group Data, 2000-2016. Br J Dermatol. 2021;185:185-194. doi:10.1111/bjd.19818
- Agner T, Elsner P. Hand eczema: epidemiology, prognosis and prevention. J Eur Acad Dermatol Venereol. 2020;34(suppl 1):4-12. doi:10.1111/jdv.16061
- Cazzaniga S, Ballmer-Weber BK, Gräni N, et al. Medical, psychological and socio-economic implications of chronic hand eczema: a cross-sectional study. J Eur Acad Dermatol Venereol. 2016;30:628-637. doi:10.1111/jdv.13479
Three JAK inhibitors get boxed warnings, modified indications
The arthritis and ulcerative colitis medicine tofacitinib (Xeljanz, Xeljanz XR) poses an increased risk of serious cardiac events such as heart attack or stroke, cancer, blood clots, and death, the Food and Drug Administration announced Sept 1.
Manufacturers of this drug along with other Janus kinase (JAK) inhibitors baricitinib (Olumiant) and upadacitinib (Rinvoq) must update their boxed warnings to include information about these health risks. The FDA made the determination after new study data from Pfizer, which manufacturers Xeljanz, found an association between a lower dose of Xeljanz and increased risk of blood clots and death.
“Recommendations for healthcare professionals will include consideration of the benefits and risks for the individual patient prior to initiating or continuing therapy,” the agency stated.
The FDA is limiting all approved uses of these three medications to patients who have not responded well to tumor necrosis factor (TNF) blockers to ensure their benefits outweigh their risks. Tofacitinib is indicated for rheumatoid arthritis, psoriatic arthritis, ulcerative colitis, and polyarticular course juvenile idiopathic arthritis. Baricitinib and upadacitinib are approved only for RA. The FDA included baricitinib and upadacitinib in the warning because of the similar properties they share with tofacitinib, even though they haven’t been studied as extensively.
“We believe this update will bring important clarity for healthcare plans on the risk/benefit profile of Xeljanz, which is a medicine informed by more clinical data than any other JAK inhibitor,” Pfizer said in a statement.
Investigators for the ORAL Surveillance trial compared two doses of tofacitinib (5 mg twice daily and 10 mg twice daily) with TNF blockers in patients with rheumatoid arthritis who were aged 50 years or older with at least one additional cardiovascular risk factor.
For both dose regimens of tofacitinib, they found an increased risk of major adverse cardiovascular events, malignancies, thrombosis, and death compared with the TNF blocker regimen. In addition, rates of lung cancers and lymphomas were higher with tofacitinib. In trial data released earlier this year, Pfizer revealed that the tofacitinib group had a much higher incidence of adjudicated malignancies compared with the TNF blocker group (1.13 vs. 0.77 per 100 person-years; hazard ratio, 1.48; 95% confidence interval, 1.04-2.09).
Impact on clinical practice
Physicians treating patients who have rheumatoid arthritis with tofacitinib may initially decrease prescriptions following the FDA’s drug safety communication, said Daniel E. Furst, MD, professor of medicine (emeritus) at the University of California, Los Angeles, adjunct professor at the University of Washington, Seattle, and a research professor at the University of Florence (Italy) – particularly those with a principal mechanism of action slightly different from that of tofacitinib, he added.
“Tofacitinib is principally a JAK 1,3 inhibitor at usual concentrations, whereas upadacitinib and baricitinib are JAK 1,2 inhibitors. Thus, I speculate that the tofacitinib prescriptions will go down more than the upadacitinib and baricitinib prescriptions,” he said in an interview.
Some patients may also be worried about taking tofacitinib, particularly those with previous events or predisposing conditions, Dr. Furst noted.
“First and foremost, I think we need to actually look at the data in a publication rather than just an FDA statement before making huge changes in our practice,” he advised.
“I am looking forward to the data finally being published ... It’s interesting that the full data still isn’t really out there beyond the press releases and an abstract. I think there’s a lot more to learn about how these drugs work and who is really at risk for harmful events,” said Alexis R. Ogdie, MD, MSCE, associate professor of medicine and epidemiology at the University of Pennsylvania, Philadelphia.
Pfizer’s data also may be affecting FDA approvals of other JAK inhibitors. This past summer, AbbVie and Eli Lilly stated that the FDA’s ongoing assessment of the safety trial was delaying the agency’s decisions about expanding use of their respective drugs upadacitinib and baricitinib.
“I think many rheumatologists have already taken this information in, and begun to incorporate it into their discussions with their patients” since it has been over a year since the first public release of information about the ORAL Surveillance trial, said Arthur Kavanaugh, MD, professor of medicine at the University of California, San Diego. “I don’t know that it will affect the approvals, but it will impact their labels.”
Wariness to prescribing tofacitinib may be lower for patients younger than those in the ORAL Surveillance trial without additional cardiovascular risk factors who are taking tofacitinib for non-RA indications, said gastroenterologist Miguel Regueiro, MD.
“The JAK inhibitor warning by the FDA is an important consideration for any prescriber or patient. The risk of cardiovascular disease and venous thromboembolism with this class of medicine appears higher in older rheumatoid arthritis patients with underlying cardiovascular disease. While the warning applies to all JAK inhibitors and likely the newer selective JAK inhibitors to come, we need to weigh the risk and benefit based on the indication for prescribing,” said Dr. Regueiro, chair of the Digestive Disease and Surgery Institute and of the department of gastroenterology, hepatology and nutrition at the Cleveland Clinic in Ohio.
“I do think that there will be a heightened awareness and wariness for older RA patients and for the prescribers. However, for inflammatory bowel disease (and other non-RA indications), it does not appear that the risk for cardiovascular disease and VTE are significantly increased. To that end, in my own practice, I still use tofacitinib for ulcerative colitis and will do the same for the selective JAK inhibitors to come for IBD. Of course, as with any medication, we need to have discussions with our patients, alert them to potential side effects and have an open line of communication for any questions or concerns.”
Gastroenterologist Stephen Hanauer, MD, professor of medicine at Northwestern University, Chicago, thought that while patients with RA have many other treatment options besides JAK inhibitors, fewer options available to patients with IBD “may motivate the use of oral [sphingosine-1-phosphate receptor modulator] agents such as ozanimod, although IBD patients are younger and [have fewer] MACE risk factors than RA patients, so absolute risk is very small in the ulcerative colitis population.”
Pfizer’s data may be affecting FDA approvals of other JAK inhibitors. This past summer, AbbVie and Eli Lilly stated that the FDA’s ongoing assessment of the safety trial was delaying the agency’s decisions about expanding use of their respective drugs upadacitinib and baricitinib.
The agency’s decision corroborates an earlier 2019 warning about the increased risk of blood clots and of death in patients with ulcerative colitis taking 10 mg tofacitinib twice daily.
The FDA said that two other JAK inhibitors, ruxolitinib (Jakafi) and fedratinib (Inrebic), are not indicated for the treatment of arthritis and other inflammatory conditions, and so are not a part of the updates being required.
Baricitinib, abrocitinib, and upadacitinib are currently under FDA review for treating atopic dermatitis (AD); a topical formulation of the JAK1/2 inhibitor ruxolitinib is under review for treating AD. Reviews for all 4 have been extended. In September 2020, baricitinib was approved for treating moderate to severe AD in Europe, at a dose of 4 mg once a day, with recommendations that the dose can be reduced to 2 mg once a day when the disease is under control, and that the dose may need to be reduced in patients with impaired kidney function, those with an increased risk of infections, and those older than aged 75 years.
In an interview, Jacob Thyssen, MD, PhD, professor of dermatology at the University of Copenhagen, said that in the EU, there has been “extensive education” about cardiovascular risks with baricitinib “and it is my impression that payers and dermatologists in Europe are confident that it is safe to use in AD.” In addition, there has been an emphasis on the differences in cardiovascular risk factors between RA and AD patients, “given that the latter group is generally young and lean.” In the United States, he added, it will be interesting to see which doses of the JAK inhibitors will be approved for AD.
Dr. Thyssen disclosed that he is a speaker, advisory board member and/or investigator for Regeneron, Sanofi-Genzyme, Eli Lilly, Pfizer, LEO Pharma, AbbVie, and Almirall.
*This story was updated 9/3/21 and 9/6/2021.
A version of this article first appeared on Medscape.com.
The arthritis and ulcerative colitis medicine tofacitinib (Xeljanz, Xeljanz XR) poses an increased risk of serious cardiac events such as heart attack or stroke, cancer, blood clots, and death, the Food and Drug Administration announced Sept 1.
Manufacturers of this drug along with other Janus kinase (JAK) inhibitors baricitinib (Olumiant) and upadacitinib (Rinvoq) must update their boxed warnings to include information about these health risks. The FDA made the determination after new study data from Pfizer, which manufacturers Xeljanz, found an association between a lower dose of Xeljanz and increased risk of blood clots and death.
“Recommendations for healthcare professionals will include consideration of the benefits and risks for the individual patient prior to initiating or continuing therapy,” the agency stated.
The FDA is limiting all approved uses of these three medications to patients who have not responded well to tumor necrosis factor (TNF) blockers to ensure their benefits outweigh their risks. Tofacitinib is indicated for rheumatoid arthritis, psoriatic arthritis, ulcerative colitis, and polyarticular course juvenile idiopathic arthritis. Baricitinib and upadacitinib are approved only for RA. The FDA included baricitinib and upadacitinib in the warning because of the similar properties they share with tofacitinib, even though they haven’t been studied as extensively.
“We believe this update will bring important clarity for healthcare plans on the risk/benefit profile of Xeljanz, which is a medicine informed by more clinical data than any other JAK inhibitor,” Pfizer said in a statement.
Investigators for the ORAL Surveillance trial compared two doses of tofacitinib (5 mg twice daily and 10 mg twice daily) with TNF blockers in patients with rheumatoid arthritis who were aged 50 years or older with at least one additional cardiovascular risk factor.
For both dose regimens of tofacitinib, they found an increased risk of major adverse cardiovascular events, malignancies, thrombosis, and death compared with the TNF blocker regimen. In addition, rates of lung cancers and lymphomas were higher with tofacitinib. In trial data released earlier this year, Pfizer revealed that the tofacitinib group had a much higher incidence of adjudicated malignancies compared with the TNF blocker group (1.13 vs. 0.77 per 100 person-years; hazard ratio, 1.48; 95% confidence interval, 1.04-2.09).
Impact on clinical practice
Physicians treating patients who have rheumatoid arthritis with tofacitinib may initially decrease prescriptions following the FDA’s drug safety communication, said Daniel E. Furst, MD, professor of medicine (emeritus) at the University of California, Los Angeles, adjunct professor at the University of Washington, Seattle, and a research professor at the University of Florence (Italy) – particularly those with a principal mechanism of action slightly different from that of tofacitinib, he added.
“Tofacitinib is principally a JAK 1,3 inhibitor at usual concentrations, whereas upadacitinib and baricitinib are JAK 1,2 inhibitors. Thus, I speculate that the tofacitinib prescriptions will go down more than the upadacitinib and baricitinib prescriptions,” he said in an interview.
Some patients may also be worried about taking tofacitinib, particularly those with previous events or predisposing conditions, Dr. Furst noted.
“First and foremost, I think we need to actually look at the data in a publication rather than just an FDA statement before making huge changes in our practice,” he advised.
“I am looking forward to the data finally being published ... It’s interesting that the full data still isn’t really out there beyond the press releases and an abstract. I think there’s a lot more to learn about how these drugs work and who is really at risk for harmful events,” said Alexis R. Ogdie, MD, MSCE, associate professor of medicine and epidemiology at the University of Pennsylvania, Philadelphia.
Pfizer’s data also may be affecting FDA approvals of other JAK inhibitors. This past summer, AbbVie and Eli Lilly stated that the FDA’s ongoing assessment of the safety trial was delaying the agency’s decisions about expanding use of their respective drugs upadacitinib and baricitinib.
“I think many rheumatologists have already taken this information in, and begun to incorporate it into their discussions with their patients” since it has been over a year since the first public release of information about the ORAL Surveillance trial, said Arthur Kavanaugh, MD, professor of medicine at the University of California, San Diego. “I don’t know that it will affect the approvals, but it will impact their labels.”
Wariness to prescribing tofacitinib may be lower for patients younger than those in the ORAL Surveillance trial without additional cardiovascular risk factors who are taking tofacitinib for non-RA indications, said gastroenterologist Miguel Regueiro, MD.
“The JAK inhibitor warning by the FDA is an important consideration for any prescriber or patient. The risk of cardiovascular disease and venous thromboembolism with this class of medicine appears higher in older rheumatoid arthritis patients with underlying cardiovascular disease. While the warning applies to all JAK inhibitors and likely the newer selective JAK inhibitors to come, we need to weigh the risk and benefit based on the indication for prescribing,” said Dr. Regueiro, chair of the Digestive Disease and Surgery Institute and of the department of gastroenterology, hepatology and nutrition at the Cleveland Clinic in Ohio.
“I do think that there will be a heightened awareness and wariness for older RA patients and for the prescribers. However, for inflammatory bowel disease (and other non-RA indications), it does not appear that the risk for cardiovascular disease and VTE are significantly increased. To that end, in my own practice, I still use tofacitinib for ulcerative colitis and will do the same for the selective JAK inhibitors to come for IBD. Of course, as with any medication, we need to have discussions with our patients, alert them to potential side effects and have an open line of communication for any questions or concerns.”
Gastroenterologist Stephen Hanauer, MD, professor of medicine at Northwestern University, Chicago, thought that while patients with RA have many other treatment options besides JAK inhibitors, fewer options available to patients with IBD “may motivate the use of oral [sphingosine-1-phosphate receptor modulator] agents such as ozanimod, although IBD patients are younger and [have fewer] MACE risk factors than RA patients, so absolute risk is very small in the ulcerative colitis population.”
Pfizer’s data may be affecting FDA approvals of other JAK inhibitors. This past summer, AbbVie and Eli Lilly stated that the FDA’s ongoing assessment of the safety trial was delaying the agency’s decisions about expanding use of their respective drugs upadacitinib and baricitinib.
The agency’s decision corroborates an earlier 2019 warning about the increased risk of blood clots and of death in patients with ulcerative colitis taking 10 mg tofacitinib twice daily.
The FDA said that two other JAK inhibitors, ruxolitinib (Jakafi) and fedratinib (Inrebic), are not indicated for the treatment of arthritis and other inflammatory conditions, and so are not a part of the updates being required.
Baricitinib, abrocitinib, and upadacitinib are currently under FDA review for treating atopic dermatitis (AD); a topical formulation of the JAK1/2 inhibitor ruxolitinib is under review for treating AD. Reviews for all 4 have been extended. In September 2020, baricitinib was approved for treating moderate to severe AD in Europe, at a dose of 4 mg once a day, with recommendations that the dose can be reduced to 2 mg once a day when the disease is under control, and that the dose may need to be reduced in patients with impaired kidney function, those with an increased risk of infections, and those older than aged 75 years.
In an interview, Jacob Thyssen, MD, PhD, professor of dermatology at the University of Copenhagen, said that in the EU, there has been “extensive education” about cardiovascular risks with baricitinib “and it is my impression that payers and dermatologists in Europe are confident that it is safe to use in AD.” In addition, there has been an emphasis on the differences in cardiovascular risk factors between RA and AD patients, “given that the latter group is generally young and lean.” In the United States, he added, it will be interesting to see which doses of the JAK inhibitors will be approved for AD.
Dr. Thyssen disclosed that he is a speaker, advisory board member and/or investigator for Regeneron, Sanofi-Genzyme, Eli Lilly, Pfizer, LEO Pharma, AbbVie, and Almirall.
*This story was updated 9/3/21 and 9/6/2021.
A version of this article first appeared on Medscape.com.
The arthritis and ulcerative colitis medicine tofacitinib (Xeljanz, Xeljanz XR) poses an increased risk of serious cardiac events such as heart attack or stroke, cancer, blood clots, and death, the Food and Drug Administration announced Sept 1.
Manufacturers of this drug along with other Janus kinase (JAK) inhibitors baricitinib (Olumiant) and upadacitinib (Rinvoq) must update their boxed warnings to include information about these health risks. The FDA made the determination after new study data from Pfizer, which manufacturers Xeljanz, found an association between a lower dose of Xeljanz and increased risk of blood clots and death.
“Recommendations for healthcare professionals will include consideration of the benefits and risks for the individual patient prior to initiating or continuing therapy,” the agency stated.
The FDA is limiting all approved uses of these three medications to patients who have not responded well to tumor necrosis factor (TNF) blockers to ensure their benefits outweigh their risks. Tofacitinib is indicated for rheumatoid arthritis, psoriatic arthritis, ulcerative colitis, and polyarticular course juvenile idiopathic arthritis. Baricitinib and upadacitinib are approved only for RA. The FDA included baricitinib and upadacitinib in the warning because of the similar properties they share with tofacitinib, even though they haven’t been studied as extensively.
“We believe this update will bring important clarity for healthcare plans on the risk/benefit profile of Xeljanz, which is a medicine informed by more clinical data than any other JAK inhibitor,” Pfizer said in a statement.
Investigators for the ORAL Surveillance trial compared two doses of tofacitinib (5 mg twice daily and 10 mg twice daily) with TNF blockers in patients with rheumatoid arthritis who were aged 50 years or older with at least one additional cardiovascular risk factor.
For both dose regimens of tofacitinib, they found an increased risk of major adverse cardiovascular events, malignancies, thrombosis, and death compared with the TNF blocker regimen. In addition, rates of lung cancers and lymphomas were higher with tofacitinib. In trial data released earlier this year, Pfizer revealed that the tofacitinib group had a much higher incidence of adjudicated malignancies compared with the TNF blocker group (1.13 vs. 0.77 per 100 person-years; hazard ratio, 1.48; 95% confidence interval, 1.04-2.09).
Impact on clinical practice
Physicians treating patients who have rheumatoid arthritis with tofacitinib may initially decrease prescriptions following the FDA’s drug safety communication, said Daniel E. Furst, MD, professor of medicine (emeritus) at the University of California, Los Angeles, adjunct professor at the University of Washington, Seattle, and a research professor at the University of Florence (Italy) – particularly those with a principal mechanism of action slightly different from that of tofacitinib, he added.
“Tofacitinib is principally a JAK 1,3 inhibitor at usual concentrations, whereas upadacitinib and baricitinib are JAK 1,2 inhibitors. Thus, I speculate that the tofacitinib prescriptions will go down more than the upadacitinib and baricitinib prescriptions,” he said in an interview.
Some patients may also be worried about taking tofacitinib, particularly those with previous events or predisposing conditions, Dr. Furst noted.
“First and foremost, I think we need to actually look at the data in a publication rather than just an FDA statement before making huge changes in our practice,” he advised.
“I am looking forward to the data finally being published ... It’s interesting that the full data still isn’t really out there beyond the press releases and an abstract. I think there’s a lot more to learn about how these drugs work and who is really at risk for harmful events,” said Alexis R. Ogdie, MD, MSCE, associate professor of medicine and epidemiology at the University of Pennsylvania, Philadelphia.
Pfizer’s data also may be affecting FDA approvals of other JAK inhibitors. This past summer, AbbVie and Eli Lilly stated that the FDA’s ongoing assessment of the safety trial was delaying the agency’s decisions about expanding use of their respective drugs upadacitinib and baricitinib.
“I think many rheumatologists have already taken this information in, and begun to incorporate it into their discussions with their patients” since it has been over a year since the first public release of information about the ORAL Surveillance trial, said Arthur Kavanaugh, MD, professor of medicine at the University of California, San Diego. “I don’t know that it will affect the approvals, but it will impact their labels.”
Wariness to prescribing tofacitinib may be lower for patients younger than those in the ORAL Surveillance trial without additional cardiovascular risk factors who are taking tofacitinib for non-RA indications, said gastroenterologist Miguel Regueiro, MD.
“The JAK inhibitor warning by the FDA is an important consideration for any prescriber or patient. The risk of cardiovascular disease and venous thromboembolism with this class of medicine appears higher in older rheumatoid arthritis patients with underlying cardiovascular disease. While the warning applies to all JAK inhibitors and likely the newer selective JAK inhibitors to come, we need to weigh the risk and benefit based on the indication for prescribing,” said Dr. Regueiro, chair of the Digestive Disease and Surgery Institute and of the department of gastroenterology, hepatology and nutrition at the Cleveland Clinic in Ohio.
“I do think that there will be a heightened awareness and wariness for older RA patients and for the prescribers. However, for inflammatory bowel disease (and other non-RA indications), it does not appear that the risk for cardiovascular disease and VTE are significantly increased. To that end, in my own practice, I still use tofacitinib for ulcerative colitis and will do the same for the selective JAK inhibitors to come for IBD. Of course, as with any medication, we need to have discussions with our patients, alert them to potential side effects and have an open line of communication for any questions or concerns.”
Gastroenterologist Stephen Hanauer, MD, professor of medicine at Northwestern University, Chicago, thought that while patients with RA have many other treatment options besides JAK inhibitors, fewer options available to patients with IBD “may motivate the use of oral [sphingosine-1-phosphate receptor modulator] agents such as ozanimod, although IBD patients are younger and [have fewer] MACE risk factors than RA patients, so absolute risk is very small in the ulcerative colitis population.”
Pfizer’s data may be affecting FDA approvals of other JAK inhibitors. This past summer, AbbVie and Eli Lilly stated that the FDA’s ongoing assessment of the safety trial was delaying the agency’s decisions about expanding use of their respective drugs upadacitinib and baricitinib.
The agency’s decision corroborates an earlier 2019 warning about the increased risk of blood clots and of death in patients with ulcerative colitis taking 10 mg tofacitinib twice daily.
The FDA said that two other JAK inhibitors, ruxolitinib (Jakafi) and fedratinib (Inrebic), are not indicated for the treatment of arthritis and other inflammatory conditions, and so are not a part of the updates being required.
Baricitinib, abrocitinib, and upadacitinib are currently under FDA review for treating atopic dermatitis (AD); a topical formulation of the JAK1/2 inhibitor ruxolitinib is under review for treating AD. Reviews for all 4 have been extended. In September 2020, baricitinib was approved for treating moderate to severe AD in Europe, at a dose of 4 mg once a day, with recommendations that the dose can be reduced to 2 mg once a day when the disease is under control, and that the dose may need to be reduced in patients with impaired kidney function, those with an increased risk of infections, and those older than aged 75 years.
In an interview, Jacob Thyssen, MD, PhD, professor of dermatology at the University of Copenhagen, said that in the EU, there has been “extensive education” about cardiovascular risks with baricitinib “and it is my impression that payers and dermatologists in Europe are confident that it is safe to use in AD.” In addition, there has been an emphasis on the differences in cardiovascular risk factors between RA and AD patients, “given that the latter group is generally young and lean.” In the United States, he added, it will be interesting to see which doses of the JAK inhibitors will be approved for AD.
Dr. Thyssen disclosed that he is a speaker, advisory board member and/or investigator for Regeneron, Sanofi-Genzyme, Eli Lilly, Pfizer, LEO Pharma, AbbVie, and Almirall.
*This story was updated 9/3/21 and 9/6/2021.
A version of this article first appeared on Medscape.com.
Clinical Edge Journal Scan Commentary: Atopic Dermatitis September 2021
George Washington University School of Medicine and Health Sciences
Washington, DC
Atopic dermatitis (AD) is a complex disease with varying degrees of itch, pain, eczematous skin lesions and quality of life impact. Research over the past decade uncovered myriad associations of AD with comorbid health disorders. There are well-established associations of AD with atopic comorbidities in children and adults, including asthma, hay fever, food allergies and less commonly eosinophilic esophagitis. AD is also associated with higher rates of mental health disorders, including depression, anxiety and attention deficit (hyperactivity) disorder.
AD patients also have multiple risk factors for hypertension, including chronic sleep deprivation and limitations on physical activity from itch. Yousaf et al conducted a systematic literature review and meta-analysis of 19 studies and found significantly increased likelihood of hypertension in patients with AD compared to healthy controls, particularly moderate-to-severe AD. Though, the odds of hypertension were lower in patients with AD compared to psoriasis.
Sleep disturbances (SD) are also common in AD patients. Manjunath et al conducted a cross-sectional, dermatology practice-based study to examine clinical differences in geriatric vs younger adult AD patients. Geriatric age was not associated with any significant differences of AD severity. However, geriatric AD patients had significantly more nights of SD, particularly trouble staying asleep, and increased fatigue than younger adults. In general, having good sleep hygiene and getting adequate sleep are important for overall health and longevity. SD therefore warrant particular attention in clinical management of AD as they are often modifiable with improved AD control.
Likewise, the myriad comorbidities associated with AD may lead to poorer health outcomes, such as hospitalization. Edigin et al conducted a longitudinal study of 23,410 adults hospitalized in the United States with AD. Hospitalizations rates increased between 1998 and 2018 owing to comorbid health disorders, but not AD itself.
Together, these results highlight the importance of holistic management of AD patients, including atopic and non-atopic comorbidities. However, many questions remain about how and when to best screen for various comorbidities. Generally, more severe AD is one of the strongest predictors of atopic and mental health comorbidities, as well as sleep disturbances and hypertension as shown in the abovementioned studies. Additionally, geriatric AD patients warrant closer monitoring of SD. Of course, screening patients for these comorbidities can take up precious time in a busy clinical practice. Though, it is a worthwhile investment of time and will improve patients’ health outcomes and the quality of care you provide for patients.
George Washington University School of Medicine and Health Sciences
Washington, DC
Atopic dermatitis (AD) is a complex disease with varying degrees of itch, pain, eczematous skin lesions and quality of life impact. Research over the past decade uncovered myriad associations of AD with comorbid health disorders. There are well-established associations of AD with atopic comorbidities in children and adults, including asthma, hay fever, food allergies and less commonly eosinophilic esophagitis. AD is also associated with higher rates of mental health disorders, including depression, anxiety and attention deficit (hyperactivity) disorder.
AD patients also have multiple risk factors for hypertension, including chronic sleep deprivation and limitations on physical activity from itch. Yousaf et al conducted a systematic literature review and meta-analysis of 19 studies and found significantly increased likelihood of hypertension in patients with AD compared to healthy controls, particularly moderate-to-severe AD. Though, the odds of hypertension were lower in patients with AD compared to psoriasis.
Sleep disturbances (SD) are also common in AD patients. Manjunath et al conducted a cross-sectional, dermatology practice-based study to examine clinical differences in geriatric vs younger adult AD patients. Geriatric age was not associated with any significant differences of AD severity. However, geriatric AD patients had significantly more nights of SD, particularly trouble staying asleep, and increased fatigue than younger adults. In general, having good sleep hygiene and getting adequate sleep are important for overall health and longevity. SD therefore warrant particular attention in clinical management of AD as they are often modifiable with improved AD control.
Likewise, the myriad comorbidities associated with AD may lead to poorer health outcomes, such as hospitalization. Edigin et al conducted a longitudinal study of 23,410 adults hospitalized in the United States with AD. Hospitalizations rates increased between 1998 and 2018 owing to comorbid health disorders, but not AD itself.
Together, these results highlight the importance of holistic management of AD patients, including atopic and non-atopic comorbidities. However, many questions remain about how and when to best screen for various comorbidities. Generally, more severe AD is one of the strongest predictors of atopic and mental health comorbidities, as well as sleep disturbances and hypertension as shown in the abovementioned studies. Additionally, geriatric AD patients warrant closer monitoring of SD. Of course, screening patients for these comorbidities can take up precious time in a busy clinical practice. Though, it is a worthwhile investment of time and will improve patients’ health outcomes and the quality of care you provide for patients.
George Washington University School of Medicine and Health Sciences
Washington, DC
Atopic dermatitis (AD) is a complex disease with varying degrees of itch, pain, eczematous skin lesions and quality of life impact. Research over the past decade uncovered myriad associations of AD with comorbid health disorders. There are well-established associations of AD with atopic comorbidities in children and adults, including asthma, hay fever, food allergies and less commonly eosinophilic esophagitis. AD is also associated with higher rates of mental health disorders, including depression, anxiety and attention deficit (hyperactivity) disorder.
AD patients also have multiple risk factors for hypertension, including chronic sleep deprivation and limitations on physical activity from itch. Yousaf et al conducted a systematic literature review and meta-analysis of 19 studies and found significantly increased likelihood of hypertension in patients with AD compared to healthy controls, particularly moderate-to-severe AD. Though, the odds of hypertension were lower in patients with AD compared to psoriasis.
Sleep disturbances (SD) are also common in AD patients. Manjunath et al conducted a cross-sectional, dermatology practice-based study to examine clinical differences in geriatric vs younger adult AD patients. Geriatric age was not associated with any significant differences of AD severity. However, geriatric AD patients had significantly more nights of SD, particularly trouble staying asleep, and increased fatigue than younger adults. In general, having good sleep hygiene and getting adequate sleep are important for overall health and longevity. SD therefore warrant particular attention in clinical management of AD as they are often modifiable with improved AD control.
Likewise, the myriad comorbidities associated with AD may lead to poorer health outcomes, such as hospitalization. Edigin et al conducted a longitudinal study of 23,410 adults hospitalized in the United States with AD. Hospitalizations rates increased between 1998 and 2018 owing to comorbid health disorders, but not AD itself.
Together, these results highlight the importance of holistic management of AD patients, including atopic and non-atopic comorbidities. However, many questions remain about how and when to best screen for various comorbidities. Generally, more severe AD is one of the strongest predictors of atopic and mental health comorbidities, as well as sleep disturbances and hypertension as shown in the abovementioned studies. Additionally, geriatric AD patients warrant closer monitoring of SD. Of course, screening patients for these comorbidities can take up precious time in a busy clinical practice. Though, it is a worthwhile investment of time and will improve patients’ health outcomes and the quality of care you provide for patients.
Atopic dermatitis: Inadequate disease control and QoL impairments among patients receiving topical therapy
Key clinical point: Almost a fifth of patients with atopic dermatitis (AD) receiving topical therapy had an uncontrolled disease and reported impairment in work productivity along with a lower quality of life (QoL).
Major finding: Overall, physicians identified 24.5% of patients as having uncontrolled disease. Patients with uncontrolled vs controlled disease had higher impairment in QoL (Dermatology Life Quality Index, 8.8 vs 6.0; P = .0003) and work productivity (Work Productivity and Activity Impairment, 23.5 vs 16.2; P = .0488).
Study details: Findings are from a retrospective, point-in-time study including 394 adults and 144 adolescent patients with moderate-to-severe AD who received topical therapy for at least 1 month.
Disclosures: This study was funded by Incyte Corporation. JH Lofland and VN Joish declared being employees and shareholders of Incyte Corporation. Three of the other authors declared being employees of Adelphi Real World.
Source: Anderson P et al. Dermatol Ther (Heidelb). 2021 Jul 15. doi: 10.1007/s13555-021-00580-2.
Key clinical point: Almost a fifth of patients with atopic dermatitis (AD) receiving topical therapy had an uncontrolled disease and reported impairment in work productivity along with a lower quality of life (QoL).
Major finding: Overall, physicians identified 24.5% of patients as having uncontrolled disease. Patients with uncontrolled vs controlled disease had higher impairment in QoL (Dermatology Life Quality Index, 8.8 vs 6.0; P = .0003) and work productivity (Work Productivity and Activity Impairment, 23.5 vs 16.2; P = .0488).
Study details: Findings are from a retrospective, point-in-time study including 394 adults and 144 adolescent patients with moderate-to-severe AD who received topical therapy for at least 1 month.
Disclosures: This study was funded by Incyte Corporation. JH Lofland and VN Joish declared being employees and shareholders of Incyte Corporation. Three of the other authors declared being employees of Adelphi Real World.
Source: Anderson P et al. Dermatol Ther (Heidelb). 2021 Jul 15. doi: 10.1007/s13555-021-00580-2.
Key clinical point: Almost a fifth of patients with atopic dermatitis (AD) receiving topical therapy had an uncontrolled disease and reported impairment in work productivity along with a lower quality of life (QoL).
Major finding: Overall, physicians identified 24.5% of patients as having uncontrolled disease. Patients with uncontrolled vs controlled disease had higher impairment in QoL (Dermatology Life Quality Index, 8.8 vs 6.0; P = .0003) and work productivity (Work Productivity and Activity Impairment, 23.5 vs 16.2; P = .0488).
Study details: Findings are from a retrospective, point-in-time study including 394 adults and 144 adolescent patients with moderate-to-severe AD who received topical therapy for at least 1 month.
Disclosures: This study was funded by Incyte Corporation. JH Lofland and VN Joish declared being employees and shareholders of Incyte Corporation. Three of the other authors declared being employees of Adelphi Real World.
Source: Anderson P et al. Dermatol Ther (Heidelb). 2021 Jul 15. doi: 10.1007/s13555-021-00580-2.
Atopic dermatitis: Rapid improvement in skin pain with baricitinib
Key clinical point: Baricitinib monotherapy improved clinically burdensome symptom of skin pain on the first day itself after the first dose in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: By day 2, skin pain numerical rating scale scores changed significantly from baseline with baricitinib vs placebo in BREEZE-AD1 (baricitinib 4 mg, −11.9%; 2 mg, −6.4%; 1 mg, −6.2%; all P less than .05), BREEZE-AD2 (baricitinib 4 mg, −12.6%; 2 mg, −5.6%; 1 mg, −6.9%; all P less than .05), and BREEZE-AD7 (baricitinib 4 mg, −6.9%; 2 mg, −7.9%; both P less than .05).
Study details: Findings are from an analysis of 3 phase 3 trials (BREEZE-AD1, BREEZE-AD2, and BREEZE-AD7) including 1,568 patients with moderate-to-severe AD with inadequate response to existing topical therapies who were randomly assigned to baricitinib or placebo with or without topical corticosteroids.
Disclosures: This work was funded by Eli Lilly and Company. Some of the authors declared receiving grants, honoraria, consulting, and/or lecturing fees from and/or serving as advisory board member, speaker, and/or investigator for various sources including Eli Lilly. Two authors declared being employees and shareholders of Eli Lilly.
Source: Thyssen JP et al. Dermatol Ther (Heidelb). 2021 Jul 18. doi: 10.1007/s13555-021-00577-x.
Key clinical point: Baricitinib monotherapy improved clinically burdensome symptom of skin pain on the first day itself after the first dose in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: By day 2, skin pain numerical rating scale scores changed significantly from baseline with baricitinib vs placebo in BREEZE-AD1 (baricitinib 4 mg, −11.9%; 2 mg, −6.4%; 1 mg, −6.2%; all P less than .05), BREEZE-AD2 (baricitinib 4 mg, −12.6%; 2 mg, −5.6%; 1 mg, −6.9%; all P less than .05), and BREEZE-AD7 (baricitinib 4 mg, −6.9%; 2 mg, −7.9%; both P less than .05).
Study details: Findings are from an analysis of 3 phase 3 trials (BREEZE-AD1, BREEZE-AD2, and BREEZE-AD7) including 1,568 patients with moderate-to-severe AD with inadequate response to existing topical therapies who were randomly assigned to baricitinib or placebo with or without topical corticosteroids.
Disclosures: This work was funded by Eli Lilly and Company. Some of the authors declared receiving grants, honoraria, consulting, and/or lecturing fees from and/or serving as advisory board member, speaker, and/or investigator for various sources including Eli Lilly. Two authors declared being employees and shareholders of Eli Lilly.
Source: Thyssen JP et al. Dermatol Ther (Heidelb). 2021 Jul 18. doi: 10.1007/s13555-021-00577-x.
Key clinical point: Baricitinib monotherapy improved clinically burdensome symptom of skin pain on the first day itself after the first dose in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: By day 2, skin pain numerical rating scale scores changed significantly from baseline with baricitinib vs placebo in BREEZE-AD1 (baricitinib 4 mg, −11.9%; 2 mg, −6.4%; 1 mg, −6.2%; all P less than .05), BREEZE-AD2 (baricitinib 4 mg, −12.6%; 2 mg, −5.6%; 1 mg, −6.9%; all P less than .05), and BREEZE-AD7 (baricitinib 4 mg, −6.9%; 2 mg, −7.9%; both P less than .05).
Study details: Findings are from an analysis of 3 phase 3 trials (BREEZE-AD1, BREEZE-AD2, and BREEZE-AD7) including 1,568 patients with moderate-to-severe AD with inadequate response to existing topical therapies who were randomly assigned to baricitinib or placebo with or without topical corticosteroids.
Disclosures: This work was funded by Eli Lilly and Company. Some of the authors declared receiving grants, honoraria, consulting, and/or lecturing fees from and/or serving as advisory board member, speaker, and/or investigator for various sources including Eli Lilly. Two authors declared being employees and shareholders of Eli Lilly.
Source: Thyssen JP et al. Dermatol Ther (Heidelb). 2021 Jul 18. doi: 10.1007/s13555-021-00577-x.
Abrocitinib emerges as a potential treatment option for moderate-to-severe atopic dermatitis
Key clinical point: Preliminary evidence from a meta-analysis suggests that abrocitinib was significantly beneficial with a tolerable adverse event profile in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: At 12 weeks, patients treated with abrocitinib achieved a significantly higher Investigator’s Global Assessment response vs those treated with placebo (risk ratio [RR], 3.52; P less than .00001). Treatment-emergent adverse effects were mostly mild and manageable, with a higher risk in the abrocitinib vs placebo group (RR, 1.17; P = .002).
Study details: Findings are from a meta-analysis of 4 randomized controlled trials, which assessed clinical outcomes in 1,175 patients with moderate-to-severe AD with inadequate response to topical medications who received abrocitinib 100 or 200 mg and 334 control participants who received placebo.
Disclosures: The study did not receive any funding. No conflict of interests was reported.
Source: Meher BR et al. J Dermatol Treat. 2021 Jul 27. doi: 10.1080/09546634.2021.1961997.
Key clinical point: Preliminary evidence from a meta-analysis suggests that abrocitinib was significantly beneficial with a tolerable adverse event profile in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: At 12 weeks, patients treated with abrocitinib achieved a significantly higher Investigator’s Global Assessment response vs those treated with placebo (risk ratio [RR], 3.52; P less than .00001). Treatment-emergent adverse effects were mostly mild and manageable, with a higher risk in the abrocitinib vs placebo group (RR, 1.17; P = .002).
Study details: Findings are from a meta-analysis of 4 randomized controlled trials, which assessed clinical outcomes in 1,175 patients with moderate-to-severe AD with inadequate response to topical medications who received abrocitinib 100 or 200 mg and 334 control participants who received placebo.
Disclosures: The study did not receive any funding. No conflict of interests was reported.
Source: Meher BR et al. J Dermatol Treat. 2021 Jul 27. doi: 10.1080/09546634.2021.1961997.
Key clinical point: Preliminary evidence from a meta-analysis suggests that abrocitinib was significantly beneficial with a tolerable adverse event profile in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: At 12 weeks, patients treated with abrocitinib achieved a significantly higher Investigator’s Global Assessment response vs those treated with placebo (risk ratio [RR], 3.52; P less than .00001). Treatment-emergent adverse effects were mostly mild and manageable, with a higher risk in the abrocitinib vs placebo group (RR, 1.17; P = .002).
Study details: Findings are from a meta-analysis of 4 randomized controlled trials, which assessed clinical outcomes in 1,175 patients with moderate-to-severe AD with inadequate response to topical medications who received abrocitinib 100 or 200 mg and 334 control participants who received placebo.
Disclosures: The study did not receive any funding. No conflict of interests was reported.
Source: Meher BR et al. J Dermatol Treat. 2021 Jul 27. doi: 10.1080/09546634.2021.1961997.