User login
A Practical Overview of Pediatric Atopic Dermatitis, Part 3: Differential Diagnosis, Comorbidities, and Measurement of Disease Burden
In parts 1 and 2 of this series on atopic dermatitis (AD),1,2 the current putative pathogenesis, scoring systems for severity grading, and epidemiology were reviewed. Part 3 reviews the differential diagnosis, with an emphasis on the difficulty of differentiation from some rare but notable illnesses, as well as the recently expanding data on comorbidities that identify AD as a multisystem disorder with widespread health implications for the patient.
Differential Diagnosis for Pediatric AD
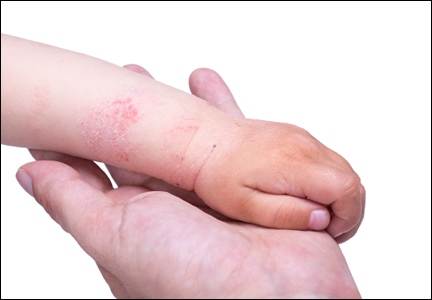
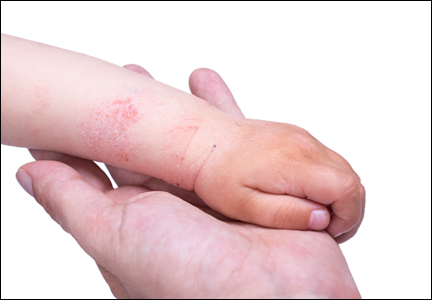
The differential diagnosis for pediatric AD includes chronic dermatoses (eg, seborrheic dermatitis, psoriasis), congenital disorders (eg, Netherton syndrome), malignant diseases (eg, cutaneous T-cell lymphoma [CTCL]), immunodeficiencies, infections, and metabolic disorders.3 Netherton syndrome must be ruled out to prevent extensive drug absorption when treating with topical calcineurin inhibitors (TCIs).4 Due to the presence of bamboo hairs in these patients, a hair mount may aid in the diagnosis of Netherton syndrome. Misdiagnosis of CTCL as AD may complicate the analysis of safety data on TCIs.4,5 Multiple skin biopsies are essential in cases of suspected CTCL to provide an accurate diagnosis. Biopsy can be considered in AD cases with changing and/or unusual morphology, erythrodermic skin changes, and disease that is poorly responsive to multiple therapeutic modalities.
Comorbidities in Pediatric AD
Psychosocial Comorbidities
Pediatric AD often takes a psychological toll on patients as well as household members. Almost half of children with AD are reported to have a severely impaired quality of life (QOL).6 Contributing factors include fatigue, sleep disturbance, activity restriction (eg, inability to participate in sports), and depression.7
Chamlin et al8 developed the Childhood Atopic Dermatitis Impact Scale (CADIS), a 45-item instrument (refined from a 62-item prototype), to measure QOL in young children with AD and their family members. Responses were evaluated with consideration of 5 domains: symptoms and activity limitations/behaviors in children, as well as family/social function, sleep, and emotions in parents. The top 12 factors that parents found most bothersome about AD included itching/scratching, child’s pain/discomfort, sleep issues, embarrassment or worry about appearance, child’s fussiness/irritability/crying/unhappiness, helplessness/can’t control it/predict it, worry about skin infection, dryness of skin/nonsmooth skin, skin bleeding, worry about damage/scars, stares/comments of strangers and other children, and rashes/redness of skin/discoloration. Parents were asked to respond to items about their emotional health and social functioning, such as “My child’s skin condition has strained my relationship with my spouse or partner,” “My child’s skin condition makes me feel sad or depressed,” and “I am bothered by the reaction of strangers to this skin condition.”8
Kiebert et al9 found that AD patients had lower scores on the Short Form-36 Health Survey’s vitality, social functioning, and mental health subscales compared to individuals in the general population. The authors noted that anxiety in AD patients is of particular concern, as stress has been found to trigger the itch-scratch cycle, potentially setting off AD flare-ups.9 Family impact of AD is aggravated by disease severity. Sleeplessness, relationship stress, and time management can all cause family problems in patients with AD.8
In a survey of 3775 older teenagers aged 18 to 19 years (80% response rate out of 4774 prospective participants), 9.7% of participants reported having current AD.10 Suicidal ideation was higher in those with current AD than those without AD (15.5% vs 9.1%). The prevalence of suicidal ideation rose to 23.8% in those with both AD and itch. Diagnosis of AD (as determined through participant responses to the question, ‘‘Do you have, or have you had eczema?’’) was associated with mental health problems in 16.0% of those with AD compared to 10.1% of those without AD, with an especially reduced likelihood of romantic relationships for adolescent boys with AD, as measured using the Strength and Difficulties Questionnaire, which measures 4 problem domains and assesses presence of mental health issues in the past 6 months, and the Hopkins Symptom Checklist 10, which uses 10 questions to measure anxiety and depression symptoms in the past week.10
Dalgard et al11 assessed whether the psychological burden of AD persists in adulthood in an international, multicenter, observational, cross-sectional study conducted in 13 European countries. Each dermatology clinic recruited 250 consecutive adult outpatients to complete a questionnaire along with a control group of 125 hospital employees without skin disease from the same institution but from different departments. The study included a total of 4994 participants (3635 patients and 1359 controls). Clinical depression and anxiety were present in 10.1% and 17.6% of patients, respectively, versus 4.3% and 11.1% of controls, respectively. The prevalence of depression and anxiety was highest in patients with leg ulcers, hand eczema, psoriasis, and AD.11 This study demonstrated that the psychological comorbidities of childhood conditions such as AD may persist into adulthood.
Lymphoma
In a systematic review of the literature and a separate meta-analysis, Legendre et al12 identified a slight increase in lymphoma among AD patients, with an uncertain but potential increase associated with topical corticosteroid application. This finding is similar to trends seen in other systemic inflammatory conditions that involve the skin, such as psoriasis, and is felt to relate to long-term inflammation.
Obesity
Obesity has been associated with a greater risk for moderate to severe AD in children.13,14
Infections
Children with AD are at a higher risk for cutaneous infections and generalization of these infections. The leading infections would be with Staphylococcus aureus, but group A streptococci infections do occur. Herpes simplex virus, vaccinia virus or Kaposi varicelliform eruption (KVE), molluscum with or without dermatitis, and fungal infections occur less commonly but with greater morbidity, largely due to the impaired barrier and some innate reduction in cutaneous immunity.15
Atopic dermatitis in children also is associated with a higher prevalence of extracutaneous infections such as influenza, pneumonia, urinary tract infections, varicella-zoster virus, recurrent ear infections, sinus infections, sore throat, and head or chest colds.16 Children with AD and warts (human papillomavirus infection) have an even greater risk for these comorbidities.17 Warts and molluscum infections may become more extensive in children with AD.18 Generalization of herpetic infections occurs more easily in AD patients due to the impaired skin barrier, which includes generalized skin surface extension of herpes simplex virus type 1, varicella-zoster virus, and historically smallpox. A similar clinical appearance of generalized vesiculopustular lesions with fever can be seen when coxsackievirus A6 infections occur in AD patients; these conditions are called eczema herpeticum due to herpes simplex virus, KVE due to varicella-zoster virus and smallpox, and eczema coxsackium due to coxsackievirus A6,19 though some authors refer to all of these as KVE.20 These generalized viral illnesses overlying AD often result in fever, malaise, pain, and life-threatening skin denudation with risk for dehydration and superinfection with S aureus.7,18 It has been shown that the occurrence of eczema herpeticum in AD is associated with and may be caused by an inability to induce human β-defensin 2 and 3 as well as cathelicidin.21
Staphylococcus aureus colonization has been noted in 90% to 100% of AD cases, which can be associated with a higher eczema area and severity index score.22-24 The role of S aureus in AD includes flare triggering through release of superantigens, leading to IL-31–induced pruritis.25 Recurrent infection with either methicillin-sensitive or methicillin-resistant S aureus has been noted in AD.18,26 Skin infections also occur in AD and appear as erosions and pustules, and coinfection with Streptococcus and Staphylococcus does occur; therefore, cultures often are needed to determine the type of bacteria present on the skin in severe cases and when infection is suspected.27 Perianal bacterial dermatitis is a variant of infected AD occurring in the anal/groin area that is associated with S aureus and/or streptococcal superinfection in which topical corticosteroids and topical anti-infectives can be used. In some severe cases, oral antibiotics may be needed.28
Injury/Hyperactivity
Children aged 0 to 5 years with AD carry an increased risk for injuries requiring medical attention, with association in part due to attention deficit disorder, depression, and anxiety. Antihistamines are believed to aggravate this issue by promoting daytime somnolence29; however, pruritus-induced sleep disturbances in AD also may be responsible for daytime somnolence.30
Contact Allergy and Sensitization
Children with AD may become sensitized to environmental allergens through delayed-type hypersensitivity. The presumed mechanism is that these agents include ingredients added into applied medicaments and application occurs over an impaired skin barrier allowing for absorption and greater risk of antigen presentation. Approximately 50% of children with difficult-to-control AD will react to 1 or more epicutaneous allergens, and patch testing can be performed to identify relevant allergens that can improve skin severity.7 Severe dermatitis and id generalized hypersensitivity reactions in patients with AD and nickel allergic contact dermatitis have been described and may aggravate underlying AD.31
Family Burden of AD
Parents or caregivers of children with moderate and severe AD spend nearly 3 hours a day caring for their child’s skin and experience QOL impairments including lack of sleep and/or privacy, often due to cosleeping; treatment-related financial expenditures; and feelings of hopelessness, guilt, and depression.7
Steroid Phobia
Steroid phobia is the fear of topical application of corticosteroids resulting in systemic side effects including unrealistic fears (eg, fear that the child will develop muscles such as an anabolic steroid user) as well as realistic but statistically low-risk fears (eg, fear of systemic absorption). These fears often result in underutilization of prescribed topical corticosteroid therapies and undertreatment of children with AD.32,33
Financial Burden
The cost of AD can be high in the United States, with adult data demonstrating costs ranging from $371 to $489 per person.34 The last published cost data for pediatric AD was from 2003, with an average cost of $219 per year.35 Costs include time lost from work, household purchases (eg, skin care products), and co-pays for visits and medication, with an estimated average expenditure per person (SE) of $601.06 ($137.26) annually in 2012.36 The cost of ambulatory care and emergency department visits for AD in children in the United States in 1993 was estimated at $364 million.37-39 In 2002, Ellis et al40 estimated the overall cost of AD to be between $900 million and $3.8 billion in the United States (1997-1998) based on projections from claims, prescriptions, and comorbidities reported to a private insurer and Medicaid. Ellis et al41 further determined that topical tacrolimus was similar in cost to high-potency corticosteroids.
Pediatric AD often progresses to adult hand eczema and leads to further morbidity, especially in health care workers.42 Kemp43 reviewed the cost of AD in children and concluded that AD was a condition with major handicap with personal, financial, and social effects. A cost review of studies conducted in 163,700 children with AD showed that costs related to AD totaled $316.7 million per year. The author concluded that there were substantial psychosocial and financial stresses associated with pediatric AD but no clear path to potential reduction in related costs.43
Sleep Disturbances
Sleep disturbances are common in pediatric AD patients. Pruritus usually is exacerbated at bedtime due to reduced humidity and lack of distractions to prevent scratching. Sleep deprivation has a substantial impact on both the patient and his/her household. Parental frustration increases with sleep disturbance.18,44 Sleep deprivation is associated with greater severity, both because it is one of the most difficult aspects of illness and because the associated pruritus makes for greater damage done to the skin through injurious scratching.
Sleep disturbances also may interfere with growth and overnight release of growth hormones.18,44 This latter issue can result in reduced linear growth velocity. Furthermore, sleep deprivation can cause increased risk of accidents and poor school performance.18,44,45
Many children do not outgrow AD. In adults, AD-associated sleep deprivation has been shown to have an association with fatigue, regular daytime sleepiness, and regular insomnia, correlating to number of sick days, doctor visits, and poorer overall health status.45
Inadequate Disease Control
Inadequate disease control has been described by Eichenfeld46 as an important issue in AD at this time. Untreated, undertreated, and improperly treated AD are important issues affecting long-term AD care. He further cited steroid phobia as a contributor to undertreatment.46 Fleischer47 has cited the black box warning present on TCIs as a further deterrent to adequate therapeutic control in our current therapeutic paradigm. Undertreatment may result in uncontrolled disease activity, impaired QOL, infections, and sleep disturbances. The role of undertreatment as a driver of the atopic march is unknown.
Conclusion
Atopic dermatitis is a multisystem disorder that has wide-reaching comorbidities and may mimic a variety of skin conditions. The topic of comorbidities is new and emerging and bears further review to define risk factors, prevention strategies, and long-term monitoring requirements.
- Silverberg NB. A practical overview of pediatric atopic dermatitis, part 1: epidemiology and pathogenesis. Cutis. 2016;97:267-271.
- Silverberg NB. A practical overview of pediatric atopic dermatitis, part 2: triggers and grading. Cutis. 2016;97:326-329.
- Siegfried EC, Hebert AA. Diagnosis of atopic dermatitis: mimics, overlaps, and complications. J Clin Med. 2015;4:884-917.
- Allen A, Siegfried E, Silverman R, et al. Significant absorption of topical tacrolimus in 3 patients with Netherton syndrome. Arch Dermatol. 2001;137:747-750.
- Carr WW. Topical calcineurin inhibitors for atopic dermatitis: review and treatment recommendations. Paediatr Drugs. 2013;15:303-310.
- Chamlin SL, Lai JS, Cella D, et al. Childhood Atopic Dermatitis Impact Scale: reliability, discriminative and concurrent validity, and responsiveness. Arch Dermatol. 2007;143:768-772.
- Tollefson MM, Bruckner AL. Atopic dermatitis: skin-directed management. Pediatrics. 2014;134:E1735-E1744.
- Chamlin SL, Cella D, Frieden IJ, et al. Development of the Childhood Atopic Dermatitis Impact Scale: initial validation of a quality-of-life measure for young children with atopic dermatitis and their families. J Invest Dermatol. 2005;125:1106-1111.
- Kiebert G, Sorensen SV, Revicki D, et al. Atopic dermatitis is associated with a decrement in health-related quality of life. Int J Dermatol. 2002;41:151-158.
- Halvorsen JA, Lien L, Dalgard F, et al. Suicidal ideation, mental health problems, and social function in adolescents with eczema: a population-based study. J Invest Dermatol. 2014;134:1847-1854.
- Dalgard FJ, Gieler U, Tomas-Aragones L, et al. The psychological burden of skin diseases: a cross-sectional multicenter study among dermatological out-patients in 13 European countries. J Invest Dermatol. 2015;135:984-991.
- Legendre L, Barnetche T, Mazereeuw-Hautier J, et al. Risk of lymphoma in patients with atopic dermatitis and the role of topical treatment: a systematic review and meta-analysis. J Am Acad Dermatol. 2015;72:992-1002.
- Koutroulis I, Magnelli L, Gaughan J, et al. Atopic dermatitis is more severe in children over the age of two who have an increased body mass index. Acta Paediatr. 2015;104:713-717.
- Silverberg JI, Becker L, Kwasny M, et al. Central obesity and high blood pressure in pediatric patients with atopic dermatitis. JAMA Dermatol. 2015;151:144-152.
- De D, Kanwar AJ, Handa S. Comparative efficacy of Hanifin and Rajka’s criteria and the UK working party’s diagnostic criteria in diagnosis of atopic dermatitis in a hospital setting in North India. J Eur Acad Dermatol Venereol. 2006;20:853-859.
- Silverberg JI, Silverberg NB. Childhood atopic dermatitis and warts are associated with increased risk of infection: a US population-based study [published online October 4, 2013]. J Allergy Clin Immunol. 2014;133:1041-1047.
- Silverberg J, Garg N, Silverberg NB. New developments in comorbidities of atopic dermatitis. Cutis. 2014;93:222-224.
- Kiken DA, Silverberg NB. Atopic dermatitis in children, part 1: epidemiology, clinical features, and complications. Cutis. 2006;78:241-247.
- Mathes EF, Oza V, Frieden IJ, et al. “Eczema coxsackium” and unusual cutaneous findings in an enterovirus outbreak. Pediatrics. 2013;132:E149-E157.
- Vora RV, Pilani AP, Jivani NB, et al. Kaposi varicelliform eruption. Indian Dermatol Online J. 2015;6:364-366.
- Hata TR, Kotol P, Boguniewicz M, et al. History of eczema herpeticum is associated with the inability to induce human β-defensin (HBD)-2, HBD-3 and cathelicidin in the skin of patients with atopic dermatitis. Br J Dermatol. 2010;163:659-661.
- Abeck D, Mempel M. Staphylococcus aureus colonization in atopic dermatitis and its therapeutic implications. Br J Dermatol. 1998;139:13-16.
- Leyden JJ, Marples RR, Kligman AM. Staphylococcus aureus in the lesions of atopic dermatitis. Br J Dermatol. 1974;90:525-530.
- Lipnharski C, d’Azevedo PA, Quinto VP, et al. Colonization by S. aureus increases the EASI and the number of appointments by patients with atopic dermatitis: cohort with 93 patients. An Bras Dermatol. 2013;88:518-521.
- Paller AS. Latest approaches to treating atopic dermatitis. Chem Immunol Allergy. 2012;96:132-140.
- Groner A, Laing-Grayman D, Silverberg NB. Outpatient pediatric community-acquired methicillin-resistant Staphylococcus aureus: a polymorphous clinical disease. Cutis. 2008;81:115-122.
- Sugarman JL, Hersh AL, Okamura T, et al. A retrospective review of streptococcal infections in pediatric atopic dermatitis. Pediatr Dermatol. 2011;28:230-234.
- Heath C, Desai N, Silverberg NB. Recent microbiological shifts in perianal bacterial dermatitis: Staphylococcus aureus predominance. Pediatr Dermatol. 2009;26:696-700.
- Garg N, Silverberg JI. Association between childhood allergic disease, psychological comorbidity, and injury requiring medical attention. Ann Allergy Asthma Immunol. 2014;112:525-532.
- Lavery MJ, Stull C, Kinney MO, et al. Nocturnal pruritus: the battle for a peaceful night’s sleep. Int J Mol Sci. 2016;17:E425.
- Silverberg NB, Licht J, Friedler S, et al. Nickel contact hypersensitivity in children. Pediatr Dermatol. 2002;19:110-113.
- Aubert-Wastiaux H, Moret L, Le Rhun A, et al. Topical corticosteroid phobia in atopic dermatitis: a study of its nature, origins and frequency. Br J Dermatol. 2011;165:808-814.
- Kojima R, Fujiwara T, Matsuda A, et al. Factors associated with steroid phobia in caregivers of children with atopic dermatitis. Pediatr Dermatol. 2013;30:29-35.
- Silverberg JI. Health care utilization, patient costs, and access to care in US adults with eczema: a population-based study. JAMA Dermatol. 2015;151:743-752.
- Weinmann S, Kamtsiuris P, Henke KD, et al. The costs of atopy and asthma in children: assessment of direct costs and their determinants in a birth cohort. Pediatr Allergy Immunol. 2003;14:18-26.
- Fivenson D, Arnold RJ, Kaniecki DJ, et al. The effect of atopic dermatitis on total burden of illness and quality of life on adults and children in a large managed care organization. J Manag Care Pharm. 2002;8:333-342.
- Verboom P, Hakkaart-Van L, Sturkenboom M, et al. The cost of atopic dermatitis in the Netherlands: an international comparison. Br J Dermatol. 2002;147:716-724.
- Lapidus CS, Schwarz DF, Honig PJ. Atopic dermatitis in children: who cares? who pays? J Am Acad Dermatol. 1993;28:699-703.
- Carroll CL, Balkrishnan R, Feldman SR, et al. The burden of atopic dermatitis: impact on the patient, family, and society. Pediatr Dermatol. 2005;22:192-199.
- Ellis CN, Drake LA, Prendergast MM, et al. Cost of atopic dermatitis and eczema in the United States. J Am Acad Dermatol. 2002;46:361-370.
- Ellis CN, Prendergast MM, Tokar M, et al. Quantifying costs associated with atopic dermatitis. J Manag Care Pharm. 2003;9:278.
- Lee SW, Cheong SH, Byun JY, et al. Occupational hand eczema among nursing staffs in Korea: self-reported hand eczema and contact sensitization of hospital nursing staffs. J Dermatol. 2013;40:182-187.
- Kemp AS. Cost of illness of atopic dermatitis in children: a societal perspective. Pharmacoeconomics. 2003;21:105-113.
- Munro DD. Topical corticosteroid therapy and its effect on the hypothalamic-pituitary-adrenal axis. Dermatologica. 1976;152:173-180.
- Silverberg JI, Garg NK, Paller AS, et al. Sleep disturbances in adults with eczema are associated with impaired overall health: a US population-based study. J Invest Dermatol. 2015;135:56-66.
- Eichenfield LF. Improving outcomes in atopic dermatitis. for advances in dermatology. Dermatology Focus. 2015;34:1-6.
- Fleischer AB Jr. Black box warning for topical calcineurin inhibitors and the death of common sense. Dermatol Online J. 2006;12:2.
In parts 1 and 2 of this series on atopic dermatitis (AD),1,2 the current putative pathogenesis, scoring systems for severity grading, and epidemiology were reviewed. Part 3 reviews the differential diagnosis, with an emphasis on the difficulty of differentiation from some rare but notable illnesses, as well as the recently expanding data on comorbidities that identify AD as a multisystem disorder with widespread health implications for the patient.
Differential Diagnosis for Pediatric AD
The differential diagnosis for pediatric AD includes chronic dermatoses (eg, seborrheic dermatitis, psoriasis), congenital disorders (eg, Netherton syndrome), malignant diseases (eg, cutaneous T-cell lymphoma [CTCL]), immunodeficiencies, infections, and metabolic disorders.3 Netherton syndrome must be ruled out to prevent extensive drug absorption when treating with topical calcineurin inhibitors (TCIs).4 Due to the presence of bamboo hairs in these patients, a hair mount may aid in the diagnosis of Netherton syndrome. Misdiagnosis of CTCL as AD may complicate the analysis of safety data on TCIs.4,5 Multiple skin biopsies are essential in cases of suspected CTCL to provide an accurate diagnosis. Biopsy can be considered in AD cases with changing and/or unusual morphology, erythrodermic skin changes, and disease that is poorly responsive to multiple therapeutic modalities.
Comorbidities in Pediatric AD
Psychosocial Comorbidities
Pediatric AD often takes a psychological toll on patients as well as household members. Almost half of children with AD are reported to have a severely impaired quality of life (QOL).6 Contributing factors include fatigue, sleep disturbance, activity restriction (eg, inability to participate in sports), and depression.7
Chamlin et al8 developed the Childhood Atopic Dermatitis Impact Scale (CADIS), a 45-item instrument (refined from a 62-item prototype), to measure QOL in young children with AD and their family members. Responses were evaluated with consideration of 5 domains: symptoms and activity limitations/behaviors in children, as well as family/social function, sleep, and emotions in parents. The top 12 factors that parents found most bothersome about AD included itching/scratching, child’s pain/discomfort, sleep issues, embarrassment or worry about appearance, child’s fussiness/irritability/crying/unhappiness, helplessness/can’t control it/predict it, worry about skin infection, dryness of skin/nonsmooth skin, skin bleeding, worry about damage/scars, stares/comments of strangers and other children, and rashes/redness of skin/discoloration. Parents were asked to respond to items about their emotional health and social functioning, such as “My child’s skin condition has strained my relationship with my spouse or partner,” “My child’s skin condition makes me feel sad or depressed,” and “I am bothered by the reaction of strangers to this skin condition.”8
Kiebert et al9 found that AD patients had lower scores on the Short Form-36 Health Survey’s vitality, social functioning, and mental health subscales compared to individuals in the general population. The authors noted that anxiety in AD patients is of particular concern, as stress has been found to trigger the itch-scratch cycle, potentially setting off AD flare-ups.9 Family impact of AD is aggravated by disease severity. Sleeplessness, relationship stress, and time management can all cause family problems in patients with AD.8
In a survey of 3775 older teenagers aged 18 to 19 years (80% response rate out of 4774 prospective participants), 9.7% of participants reported having current AD.10 Suicidal ideation was higher in those with current AD than those without AD (15.5% vs 9.1%). The prevalence of suicidal ideation rose to 23.8% in those with both AD and itch. Diagnosis of AD (as determined through participant responses to the question, ‘‘Do you have, or have you had eczema?’’) was associated with mental health problems in 16.0% of those with AD compared to 10.1% of those without AD, with an especially reduced likelihood of romantic relationships for adolescent boys with AD, as measured using the Strength and Difficulties Questionnaire, which measures 4 problem domains and assesses presence of mental health issues in the past 6 months, and the Hopkins Symptom Checklist 10, which uses 10 questions to measure anxiety and depression symptoms in the past week.10
Dalgard et al11 assessed whether the psychological burden of AD persists in adulthood in an international, multicenter, observational, cross-sectional study conducted in 13 European countries. Each dermatology clinic recruited 250 consecutive adult outpatients to complete a questionnaire along with a control group of 125 hospital employees without skin disease from the same institution but from different departments. The study included a total of 4994 participants (3635 patients and 1359 controls). Clinical depression and anxiety were present in 10.1% and 17.6% of patients, respectively, versus 4.3% and 11.1% of controls, respectively. The prevalence of depression and anxiety was highest in patients with leg ulcers, hand eczema, psoriasis, and AD.11 This study demonstrated that the psychological comorbidities of childhood conditions such as AD may persist into adulthood.
Lymphoma
In a systematic review of the literature and a separate meta-analysis, Legendre et al12 identified a slight increase in lymphoma among AD patients, with an uncertain but potential increase associated with topical corticosteroid application. This finding is similar to trends seen in other systemic inflammatory conditions that involve the skin, such as psoriasis, and is felt to relate to long-term inflammation.
Obesity
Obesity has been associated with a greater risk for moderate to severe AD in children.13,14
Infections
Children with AD are at a higher risk for cutaneous infections and generalization of these infections. The leading infections would be with Staphylococcus aureus, but group A streptococci infections do occur. Herpes simplex virus, vaccinia virus or Kaposi varicelliform eruption (KVE), molluscum with or without dermatitis, and fungal infections occur less commonly but with greater morbidity, largely due to the impaired barrier and some innate reduction in cutaneous immunity.15
Atopic dermatitis in children also is associated with a higher prevalence of extracutaneous infections such as influenza, pneumonia, urinary tract infections, varicella-zoster virus, recurrent ear infections, sinus infections, sore throat, and head or chest colds.16 Children with AD and warts (human papillomavirus infection) have an even greater risk for these comorbidities.17 Warts and molluscum infections may become more extensive in children with AD.18 Generalization of herpetic infections occurs more easily in AD patients due to the impaired skin barrier, which includes generalized skin surface extension of herpes simplex virus type 1, varicella-zoster virus, and historically smallpox. A similar clinical appearance of generalized vesiculopustular lesions with fever can be seen when coxsackievirus A6 infections occur in AD patients; these conditions are called eczema herpeticum due to herpes simplex virus, KVE due to varicella-zoster virus and smallpox, and eczema coxsackium due to coxsackievirus A6,19 though some authors refer to all of these as KVE.20 These generalized viral illnesses overlying AD often result in fever, malaise, pain, and life-threatening skin denudation with risk for dehydration and superinfection with S aureus.7,18 It has been shown that the occurrence of eczema herpeticum in AD is associated with and may be caused by an inability to induce human β-defensin 2 and 3 as well as cathelicidin.21
Staphylococcus aureus colonization has been noted in 90% to 100% of AD cases, which can be associated with a higher eczema area and severity index score.22-24 The role of S aureus in AD includes flare triggering through release of superantigens, leading to IL-31–induced pruritis.25 Recurrent infection with either methicillin-sensitive or methicillin-resistant S aureus has been noted in AD.18,26 Skin infections also occur in AD and appear as erosions and pustules, and coinfection with Streptococcus and Staphylococcus does occur; therefore, cultures often are needed to determine the type of bacteria present on the skin in severe cases and when infection is suspected.27 Perianal bacterial dermatitis is a variant of infected AD occurring in the anal/groin area that is associated with S aureus and/or streptococcal superinfection in which topical corticosteroids and topical anti-infectives can be used. In some severe cases, oral antibiotics may be needed.28
Injury/Hyperactivity
Children aged 0 to 5 years with AD carry an increased risk for injuries requiring medical attention, with association in part due to attention deficit disorder, depression, and anxiety. Antihistamines are believed to aggravate this issue by promoting daytime somnolence29; however, pruritus-induced sleep disturbances in AD also may be responsible for daytime somnolence.30
Contact Allergy and Sensitization
Children with AD may become sensitized to environmental allergens through delayed-type hypersensitivity. The presumed mechanism is that these agents include ingredients added into applied medicaments and application occurs over an impaired skin barrier allowing for absorption and greater risk of antigen presentation. Approximately 50% of children with difficult-to-control AD will react to 1 or more epicutaneous allergens, and patch testing can be performed to identify relevant allergens that can improve skin severity.7 Severe dermatitis and id generalized hypersensitivity reactions in patients with AD and nickel allergic contact dermatitis have been described and may aggravate underlying AD.31
Family Burden of AD
Parents or caregivers of children with moderate and severe AD spend nearly 3 hours a day caring for their child’s skin and experience QOL impairments including lack of sleep and/or privacy, often due to cosleeping; treatment-related financial expenditures; and feelings of hopelessness, guilt, and depression.7
Steroid Phobia
Steroid phobia is the fear of topical application of corticosteroids resulting in systemic side effects including unrealistic fears (eg, fear that the child will develop muscles such as an anabolic steroid user) as well as realistic but statistically low-risk fears (eg, fear of systemic absorption). These fears often result in underutilization of prescribed topical corticosteroid therapies and undertreatment of children with AD.32,33
Financial Burden
The cost of AD can be high in the United States, with adult data demonstrating costs ranging from $371 to $489 per person.34 The last published cost data for pediatric AD was from 2003, with an average cost of $219 per year.35 Costs include time lost from work, household purchases (eg, skin care products), and co-pays for visits and medication, with an estimated average expenditure per person (SE) of $601.06 ($137.26) annually in 2012.36 The cost of ambulatory care and emergency department visits for AD in children in the United States in 1993 was estimated at $364 million.37-39 In 2002, Ellis et al40 estimated the overall cost of AD to be between $900 million and $3.8 billion in the United States (1997-1998) based on projections from claims, prescriptions, and comorbidities reported to a private insurer and Medicaid. Ellis et al41 further determined that topical tacrolimus was similar in cost to high-potency corticosteroids.
Pediatric AD often progresses to adult hand eczema and leads to further morbidity, especially in health care workers.42 Kemp43 reviewed the cost of AD in children and concluded that AD was a condition with major handicap with personal, financial, and social effects. A cost review of studies conducted in 163,700 children with AD showed that costs related to AD totaled $316.7 million per year. The author concluded that there were substantial psychosocial and financial stresses associated with pediatric AD but no clear path to potential reduction in related costs.43
Sleep Disturbances
Sleep disturbances are common in pediatric AD patients. Pruritus usually is exacerbated at bedtime due to reduced humidity and lack of distractions to prevent scratching. Sleep deprivation has a substantial impact on both the patient and his/her household. Parental frustration increases with sleep disturbance.18,44 Sleep deprivation is associated with greater severity, both because it is one of the most difficult aspects of illness and because the associated pruritus makes for greater damage done to the skin through injurious scratching.
Sleep disturbances also may interfere with growth and overnight release of growth hormones.18,44 This latter issue can result in reduced linear growth velocity. Furthermore, sleep deprivation can cause increased risk of accidents and poor school performance.18,44,45
Many children do not outgrow AD. In adults, AD-associated sleep deprivation has been shown to have an association with fatigue, regular daytime sleepiness, and regular insomnia, correlating to number of sick days, doctor visits, and poorer overall health status.45
Inadequate Disease Control
Inadequate disease control has been described by Eichenfeld46 as an important issue in AD at this time. Untreated, undertreated, and improperly treated AD are important issues affecting long-term AD care. He further cited steroid phobia as a contributor to undertreatment.46 Fleischer47 has cited the black box warning present on TCIs as a further deterrent to adequate therapeutic control in our current therapeutic paradigm. Undertreatment may result in uncontrolled disease activity, impaired QOL, infections, and sleep disturbances. The role of undertreatment as a driver of the atopic march is unknown.
Conclusion
Atopic dermatitis is a multisystem disorder that has wide-reaching comorbidities and may mimic a variety of skin conditions. The topic of comorbidities is new and emerging and bears further review to define risk factors, prevention strategies, and long-term monitoring requirements.
In parts 1 and 2 of this series on atopic dermatitis (AD),1,2 the current putative pathogenesis, scoring systems for severity grading, and epidemiology were reviewed. Part 3 reviews the differential diagnosis, with an emphasis on the difficulty of differentiation from some rare but notable illnesses, as well as the recently expanding data on comorbidities that identify AD as a multisystem disorder with widespread health implications for the patient.
Differential Diagnosis for Pediatric AD
The differential diagnosis for pediatric AD includes chronic dermatoses (eg, seborrheic dermatitis, psoriasis), congenital disorders (eg, Netherton syndrome), malignant diseases (eg, cutaneous T-cell lymphoma [CTCL]), immunodeficiencies, infections, and metabolic disorders.3 Netherton syndrome must be ruled out to prevent extensive drug absorption when treating with topical calcineurin inhibitors (TCIs).4 Due to the presence of bamboo hairs in these patients, a hair mount may aid in the diagnosis of Netherton syndrome. Misdiagnosis of CTCL as AD may complicate the analysis of safety data on TCIs.4,5 Multiple skin biopsies are essential in cases of suspected CTCL to provide an accurate diagnosis. Biopsy can be considered in AD cases with changing and/or unusual morphology, erythrodermic skin changes, and disease that is poorly responsive to multiple therapeutic modalities.
Comorbidities in Pediatric AD
Psychosocial Comorbidities
Pediatric AD often takes a psychological toll on patients as well as household members. Almost half of children with AD are reported to have a severely impaired quality of life (QOL).6 Contributing factors include fatigue, sleep disturbance, activity restriction (eg, inability to participate in sports), and depression.7
Chamlin et al8 developed the Childhood Atopic Dermatitis Impact Scale (CADIS), a 45-item instrument (refined from a 62-item prototype), to measure QOL in young children with AD and their family members. Responses were evaluated with consideration of 5 domains: symptoms and activity limitations/behaviors in children, as well as family/social function, sleep, and emotions in parents. The top 12 factors that parents found most bothersome about AD included itching/scratching, child’s pain/discomfort, sleep issues, embarrassment or worry about appearance, child’s fussiness/irritability/crying/unhappiness, helplessness/can’t control it/predict it, worry about skin infection, dryness of skin/nonsmooth skin, skin bleeding, worry about damage/scars, stares/comments of strangers and other children, and rashes/redness of skin/discoloration. Parents were asked to respond to items about their emotional health and social functioning, such as “My child’s skin condition has strained my relationship with my spouse or partner,” “My child’s skin condition makes me feel sad or depressed,” and “I am bothered by the reaction of strangers to this skin condition.”8
Kiebert et al9 found that AD patients had lower scores on the Short Form-36 Health Survey’s vitality, social functioning, and mental health subscales compared to individuals in the general population. The authors noted that anxiety in AD patients is of particular concern, as stress has been found to trigger the itch-scratch cycle, potentially setting off AD flare-ups.9 Family impact of AD is aggravated by disease severity. Sleeplessness, relationship stress, and time management can all cause family problems in patients with AD.8
In a survey of 3775 older teenagers aged 18 to 19 years (80% response rate out of 4774 prospective participants), 9.7% of participants reported having current AD.10 Suicidal ideation was higher in those with current AD than those without AD (15.5% vs 9.1%). The prevalence of suicidal ideation rose to 23.8% in those with both AD and itch. Diagnosis of AD (as determined through participant responses to the question, ‘‘Do you have, or have you had eczema?’’) was associated with mental health problems in 16.0% of those with AD compared to 10.1% of those without AD, with an especially reduced likelihood of romantic relationships for adolescent boys with AD, as measured using the Strength and Difficulties Questionnaire, which measures 4 problem domains and assesses presence of mental health issues in the past 6 months, and the Hopkins Symptom Checklist 10, which uses 10 questions to measure anxiety and depression symptoms in the past week.10
Dalgard et al11 assessed whether the psychological burden of AD persists in adulthood in an international, multicenter, observational, cross-sectional study conducted in 13 European countries. Each dermatology clinic recruited 250 consecutive adult outpatients to complete a questionnaire along with a control group of 125 hospital employees without skin disease from the same institution but from different departments. The study included a total of 4994 participants (3635 patients and 1359 controls). Clinical depression and anxiety were present in 10.1% and 17.6% of patients, respectively, versus 4.3% and 11.1% of controls, respectively. The prevalence of depression and anxiety was highest in patients with leg ulcers, hand eczema, psoriasis, and AD.11 This study demonstrated that the psychological comorbidities of childhood conditions such as AD may persist into adulthood.
Lymphoma
In a systematic review of the literature and a separate meta-analysis, Legendre et al12 identified a slight increase in lymphoma among AD patients, with an uncertain but potential increase associated with topical corticosteroid application. This finding is similar to trends seen in other systemic inflammatory conditions that involve the skin, such as psoriasis, and is felt to relate to long-term inflammation.
Obesity
Obesity has been associated with a greater risk for moderate to severe AD in children.13,14
Infections
Children with AD are at a higher risk for cutaneous infections and generalization of these infections. The leading infections would be with Staphylococcus aureus, but group A streptococci infections do occur. Herpes simplex virus, vaccinia virus or Kaposi varicelliform eruption (KVE), molluscum with or without dermatitis, and fungal infections occur less commonly but with greater morbidity, largely due to the impaired barrier and some innate reduction in cutaneous immunity.15
Atopic dermatitis in children also is associated with a higher prevalence of extracutaneous infections such as influenza, pneumonia, urinary tract infections, varicella-zoster virus, recurrent ear infections, sinus infections, sore throat, and head or chest colds.16 Children with AD and warts (human papillomavirus infection) have an even greater risk for these comorbidities.17 Warts and molluscum infections may become more extensive in children with AD.18 Generalization of herpetic infections occurs more easily in AD patients due to the impaired skin barrier, which includes generalized skin surface extension of herpes simplex virus type 1, varicella-zoster virus, and historically smallpox. A similar clinical appearance of generalized vesiculopustular lesions with fever can be seen when coxsackievirus A6 infections occur in AD patients; these conditions are called eczema herpeticum due to herpes simplex virus, KVE due to varicella-zoster virus and smallpox, and eczema coxsackium due to coxsackievirus A6,19 though some authors refer to all of these as KVE.20 These generalized viral illnesses overlying AD often result in fever, malaise, pain, and life-threatening skin denudation with risk for dehydration and superinfection with S aureus.7,18 It has been shown that the occurrence of eczema herpeticum in AD is associated with and may be caused by an inability to induce human β-defensin 2 and 3 as well as cathelicidin.21
Staphylococcus aureus colonization has been noted in 90% to 100% of AD cases, which can be associated with a higher eczema area and severity index score.22-24 The role of S aureus in AD includes flare triggering through release of superantigens, leading to IL-31–induced pruritis.25 Recurrent infection with either methicillin-sensitive or methicillin-resistant S aureus has been noted in AD.18,26 Skin infections also occur in AD and appear as erosions and pustules, and coinfection with Streptococcus and Staphylococcus does occur; therefore, cultures often are needed to determine the type of bacteria present on the skin in severe cases and when infection is suspected.27 Perianal bacterial dermatitis is a variant of infected AD occurring in the anal/groin area that is associated with S aureus and/or streptococcal superinfection in which topical corticosteroids and topical anti-infectives can be used. In some severe cases, oral antibiotics may be needed.28
Injury/Hyperactivity
Children aged 0 to 5 years with AD carry an increased risk for injuries requiring medical attention, with association in part due to attention deficit disorder, depression, and anxiety. Antihistamines are believed to aggravate this issue by promoting daytime somnolence29; however, pruritus-induced sleep disturbances in AD also may be responsible for daytime somnolence.30
Contact Allergy and Sensitization
Children with AD may become sensitized to environmental allergens through delayed-type hypersensitivity. The presumed mechanism is that these agents include ingredients added into applied medicaments and application occurs over an impaired skin barrier allowing for absorption and greater risk of antigen presentation. Approximately 50% of children with difficult-to-control AD will react to 1 or more epicutaneous allergens, and patch testing can be performed to identify relevant allergens that can improve skin severity.7 Severe dermatitis and id generalized hypersensitivity reactions in patients with AD and nickel allergic contact dermatitis have been described and may aggravate underlying AD.31
Family Burden of AD
Parents or caregivers of children with moderate and severe AD spend nearly 3 hours a day caring for their child’s skin and experience QOL impairments including lack of sleep and/or privacy, often due to cosleeping; treatment-related financial expenditures; and feelings of hopelessness, guilt, and depression.7
Steroid Phobia
Steroid phobia is the fear of topical application of corticosteroids resulting in systemic side effects including unrealistic fears (eg, fear that the child will develop muscles such as an anabolic steroid user) as well as realistic but statistically low-risk fears (eg, fear of systemic absorption). These fears often result in underutilization of prescribed topical corticosteroid therapies and undertreatment of children with AD.32,33
Financial Burden
The cost of AD can be high in the United States, with adult data demonstrating costs ranging from $371 to $489 per person.34 The last published cost data for pediatric AD was from 2003, with an average cost of $219 per year.35 Costs include time lost from work, household purchases (eg, skin care products), and co-pays for visits and medication, with an estimated average expenditure per person (SE) of $601.06 ($137.26) annually in 2012.36 The cost of ambulatory care and emergency department visits for AD in children in the United States in 1993 was estimated at $364 million.37-39 In 2002, Ellis et al40 estimated the overall cost of AD to be between $900 million and $3.8 billion in the United States (1997-1998) based on projections from claims, prescriptions, and comorbidities reported to a private insurer and Medicaid. Ellis et al41 further determined that topical tacrolimus was similar in cost to high-potency corticosteroids.
Pediatric AD often progresses to adult hand eczema and leads to further morbidity, especially in health care workers.42 Kemp43 reviewed the cost of AD in children and concluded that AD was a condition with major handicap with personal, financial, and social effects. A cost review of studies conducted in 163,700 children with AD showed that costs related to AD totaled $316.7 million per year. The author concluded that there were substantial psychosocial and financial stresses associated with pediatric AD but no clear path to potential reduction in related costs.43
Sleep Disturbances
Sleep disturbances are common in pediatric AD patients. Pruritus usually is exacerbated at bedtime due to reduced humidity and lack of distractions to prevent scratching. Sleep deprivation has a substantial impact on both the patient and his/her household. Parental frustration increases with sleep disturbance.18,44 Sleep deprivation is associated with greater severity, both because it is one of the most difficult aspects of illness and because the associated pruritus makes for greater damage done to the skin through injurious scratching.
Sleep disturbances also may interfere with growth and overnight release of growth hormones.18,44 This latter issue can result in reduced linear growth velocity. Furthermore, sleep deprivation can cause increased risk of accidents and poor school performance.18,44,45
Many children do not outgrow AD. In adults, AD-associated sleep deprivation has been shown to have an association with fatigue, regular daytime sleepiness, and regular insomnia, correlating to number of sick days, doctor visits, and poorer overall health status.45
Inadequate Disease Control
Inadequate disease control has been described by Eichenfeld46 as an important issue in AD at this time. Untreated, undertreated, and improperly treated AD are important issues affecting long-term AD care. He further cited steroid phobia as a contributor to undertreatment.46 Fleischer47 has cited the black box warning present on TCIs as a further deterrent to adequate therapeutic control in our current therapeutic paradigm. Undertreatment may result in uncontrolled disease activity, impaired QOL, infections, and sleep disturbances. The role of undertreatment as a driver of the atopic march is unknown.
Conclusion
Atopic dermatitis is a multisystem disorder that has wide-reaching comorbidities and may mimic a variety of skin conditions. The topic of comorbidities is new and emerging and bears further review to define risk factors, prevention strategies, and long-term monitoring requirements.
- Silverberg NB. A practical overview of pediatric atopic dermatitis, part 1: epidemiology and pathogenesis. Cutis. 2016;97:267-271.
- Silverberg NB. A practical overview of pediatric atopic dermatitis, part 2: triggers and grading. Cutis. 2016;97:326-329.
- Siegfried EC, Hebert AA. Diagnosis of atopic dermatitis: mimics, overlaps, and complications. J Clin Med. 2015;4:884-917.
- Allen A, Siegfried E, Silverman R, et al. Significant absorption of topical tacrolimus in 3 patients with Netherton syndrome. Arch Dermatol. 2001;137:747-750.
- Carr WW. Topical calcineurin inhibitors for atopic dermatitis: review and treatment recommendations. Paediatr Drugs. 2013;15:303-310.
- Chamlin SL, Lai JS, Cella D, et al. Childhood Atopic Dermatitis Impact Scale: reliability, discriminative and concurrent validity, and responsiveness. Arch Dermatol. 2007;143:768-772.
- Tollefson MM, Bruckner AL. Atopic dermatitis: skin-directed management. Pediatrics. 2014;134:E1735-E1744.
- Chamlin SL, Cella D, Frieden IJ, et al. Development of the Childhood Atopic Dermatitis Impact Scale: initial validation of a quality-of-life measure for young children with atopic dermatitis and their families. J Invest Dermatol. 2005;125:1106-1111.
- Kiebert G, Sorensen SV, Revicki D, et al. Atopic dermatitis is associated with a decrement in health-related quality of life. Int J Dermatol. 2002;41:151-158.
- Halvorsen JA, Lien L, Dalgard F, et al. Suicidal ideation, mental health problems, and social function in adolescents with eczema: a population-based study. J Invest Dermatol. 2014;134:1847-1854.
- Dalgard FJ, Gieler U, Tomas-Aragones L, et al. The psychological burden of skin diseases: a cross-sectional multicenter study among dermatological out-patients in 13 European countries. J Invest Dermatol. 2015;135:984-991.
- Legendre L, Barnetche T, Mazereeuw-Hautier J, et al. Risk of lymphoma in patients with atopic dermatitis and the role of topical treatment: a systematic review and meta-analysis. J Am Acad Dermatol. 2015;72:992-1002.
- Koutroulis I, Magnelli L, Gaughan J, et al. Atopic dermatitis is more severe in children over the age of two who have an increased body mass index. Acta Paediatr. 2015;104:713-717.
- Silverberg JI, Becker L, Kwasny M, et al. Central obesity and high blood pressure in pediatric patients with atopic dermatitis. JAMA Dermatol. 2015;151:144-152.
- De D, Kanwar AJ, Handa S. Comparative efficacy of Hanifin and Rajka’s criteria and the UK working party’s diagnostic criteria in diagnosis of atopic dermatitis in a hospital setting in North India. J Eur Acad Dermatol Venereol. 2006;20:853-859.
- Silverberg JI, Silverberg NB. Childhood atopic dermatitis and warts are associated with increased risk of infection: a US population-based study [published online October 4, 2013]. J Allergy Clin Immunol. 2014;133:1041-1047.
- Silverberg J, Garg N, Silverberg NB. New developments in comorbidities of atopic dermatitis. Cutis. 2014;93:222-224.
- Kiken DA, Silverberg NB. Atopic dermatitis in children, part 1: epidemiology, clinical features, and complications. Cutis. 2006;78:241-247.
- Mathes EF, Oza V, Frieden IJ, et al. “Eczema coxsackium” and unusual cutaneous findings in an enterovirus outbreak. Pediatrics. 2013;132:E149-E157.
- Vora RV, Pilani AP, Jivani NB, et al. Kaposi varicelliform eruption. Indian Dermatol Online J. 2015;6:364-366.
- Hata TR, Kotol P, Boguniewicz M, et al. History of eczema herpeticum is associated with the inability to induce human β-defensin (HBD)-2, HBD-3 and cathelicidin in the skin of patients with atopic dermatitis. Br J Dermatol. 2010;163:659-661.
- Abeck D, Mempel M. Staphylococcus aureus colonization in atopic dermatitis and its therapeutic implications. Br J Dermatol. 1998;139:13-16.
- Leyden JJ, Marples RR, Kligman AM. Staphylococcus aureus in the lesions of atopic dermatitis. Br J Dermatol. 1974;90:525-530.
- Lipnharski C, d’Azevedo PA, Quinto VP, et al. Colonization by S. aureus increases the EASI and the number of appointments by patients with atopic dermatitis: cohort with 93 patients. An Bras Dermatol. 2013;88:518-521.
- Paller AS. Latest approaches to treating atopic dermatitis. Chem Immunol Allergy. 2012;96:132-140.
- Groner A, Laing-Grayman D, Silverberg NB. Outpatient pediatric community-acquired methicillin-resistant Staphylococcus aureus: a polymorphous clinical disease. Cutis. 2008;81:115-122.
- Sugarman JL, Hersh AL, Okamura T, et al. A retrospective review of streptococcal infections in pediatric atopic dermatitis. Pediatr Dermatol. 2011;28:230-234.
- Heath C, Desai N, Silverberg NB. Recent microbiological shifts in perianal bacterial dermatitis: Staphylococcus aureus predominance. Pediatr Dermatol. 2009;26:696-700.
- Garg N, Silverberg JI. Association between childhood allergic disease, psychological comorbidity, and injury requiring medical attention. Ann Allergy Asthma Immunol. 2014;112:525-532.
- Lavery MJ, Stull C, Kinney MO, et al. Nocturnal pruritus: the battle for a peaceful night’s sleep. Int J Mol Sci. 2016;17:E425.
- Silverberg NB, Licht J, Friedler S, et al. Nickel contact hypersensitivity in children. Pediatr Dermatol. 2002;19:110-113.
- Aubert-Wastiaux H, Moret L, Le Rhun A, et al. Topical corticosteroid phobia in atopic dermatitis: a study of its nature, origins and frequency. Br J Dermatol. 2011;165:808-814.
- Kojima R, Fujiwara T, Matsuda A, et al. Factors associated with steroid phobia in caregivers of children with atopic dermatitis. Pediatr Dermatol. 2013;30:29-35.
- Silverberg JI. Health care utilization, patient costs, and access to care in US adults with eczema: a population-based study. JAMA Dermatol. 2015;151:743-752.
- Weinmann S, Kamtsiuris P, Henke KD, et al. The costs of atopy and asthma in children: assessment of direct costs and their determinants in a birth cohort. Pediatr Allergy Immunol. 2003;14:18-26.
- Fivenson D, Arnold RJ, Kaniecki DJ, et al. The effect of atopic dermatitis on total burden of illness and quality of life on adults and children in a large managed care organization. J Manag Care Pharm. 2002;8:333-342.
- Verboom P, Hakkaart-Van L, Sturkenboom M, et al. The cost of atopic dermatitis in the Netherlands: an international comparison. Br J Dermatol. 2002;147:716-724.
- Lapidus CS, Schwarz DF, Honig PJ. Atopic dermatitis in children: who cares? who pays? J Am Acad Dermatol. 1993;28:699-703.
- Carroll CL, Balkrishnan R, Feldman SR, et al. The burden of atopic dermatitis: impact on the patient, family, and society. Pediatr Dermatol. 2005;22:192-199.
- Ellis CN, Drake LA, Prendergast MM, et al. Cost of atopic dermatitis and eczema in the United States. J Am Acad Dermatol. 2002;46:361-370.
- Ellis CN, Prendergast MM, Tokar M, et al. Quantifying costs associated with atopic dermatitis. J Manag Care Pharm. 2003;9:278.
- Lee SW, Cheong SH, Byun JY, et al. Occupational hand eczema among nursing staffs in Korea: self-reported hand eczema and contact sensitization of hospital nursing staffs. J Dermatol. 2013;40:182-187.
- Kemp AS. Cost of illness of atopic dermatitis in children: a societal perspective. Pharmacoeconomics. 2003;21:105-113.
- Munro DD. Topical corticosteroid therapy and its effect on the hypothalamic-pituitary-adrenal axis. Dermatologica. 1976;152:173-180.
- Silverberg JI, Garg NK, Paller AS, et al. Sleep disturbances in adults with eczema are associated with impaired overall health: a US population-based study. J Invest Dermatol. 2015;135:56-66.
- Eichenfield LF. Improving outcomes in atopic dermatitis. for advances in dermatology. Dermatology Focus. 2015;34:1-6.
- Fleischer AB Jr. Black box warning for topical calcineurin inhibitors and the death of common sense. Dermatol Online J. 2006;12:2.
- Silverberg NB. A practical overview of pediatric atopic dermatitis, part 1: epidemiology and pathogenesis. Cutis. 2016;97:267-271.
- Silverberg NB. A practical overview of pediatric atopic dermatitis, part 2: triggers and grading. Cutis. 2016;97:326-329.
- Siegfried EC, Hebert AA. Diagnosis of atopic dermatitis: mimics, overlaps, and complications. J Clin Med. 2015;4:884-917.
- Allen A, Siegfried E, Silverman R, et al. Significant absorption of topical tacrolimus in 3 patients with Netherton syndrome. Arch Dermatol. 2001;137:747-750.
- Carr WW. Topical calcineurin inhibitors for atopic dermatitis: review and treatment recommendations. Paediatr Drugs. 2013;15:303-310.
- Chamlin SL, Lai JS, Cella D, et al. Childhood Atopic Dermatitis Impact Scale: reliability, discriminative and concurrent validity, and responsiveness. Arch Dermatol. 2007;143:768-772.
- Tollefson MM, Bruckner AL. Atopic dermatitis: skin-directed management. Pediatrics. 2014;134:E1735-E1744.
- Chamlin SL, Cella D, Frieden IJ, et al. Development of the Childhood Atopic Dermatitis Impact Scale: initial validation of a quality-of-life measure for young children with atopic dermatitis and their families. J Invest Dermatol. 2005;125:1106-1111.
- Kiebert G, Sorensen SV, Revicki D, et al. Atopic dermatitis is associated with a decrement in health-related quality of life. Int J Dermatol. 2002;41:151-158.
- Halvorsen JA, Lien L, Dalgard F, et al. Suicidal ideation, mental health problems, and social function in adolescents with eczema: a population-based study. J Invest Dermatol. 2014;134:1847-1854.
- Dalgard FJ, Gieler U, Tomas-Aragones L, et al. The psychological burden of skin diseases: a cross-sectional multicenter study among dermatological out-patients in 13 European countries. J Invest Dermatol. 2015;135:984-991.
- Legendre L, Barnetche T, Mazereeuw-Hautier J, et al. Risk of lymphoma in patients with atopic dermatitis and the role of topical treatment: a systematic review and meta-analysis. J Am Acad Dermatol. 2015;72:992-1002.
- Koutroulis I, Magnelli L, Gaughan J, et al. Atopic dermatitis is more severe in children over the age of two who have an increased body mass index. Acta Paediatr. 2015;104:713-717.
- Silverberg JI, Becker L, Kwasny M, et al. Central obesity and high blood pressure in pediatric patients with atopic dermatitis. JAMA Dermatol. 2015;151:144-152.
- De D, Kanwar AJ, Handa S. Comparative efficacy of Hanifin and Rajka’s criteria and the UK working party’s diagnostic criteria in diagnosis of atopic dermatitis in a hospital setting in North India. J Eur Acad Dermatol Venereol. 2006;20:853-859.
- Silverberg JI, Silverberg NB. Childhood atopic dermatitis and warts are associated with increased risk of infection: a US population-based study [published online October 4, 2013]. J Allergy Clin Immunol. 2014;133:1041-1047.
- Silverberg J, Garg N, Silverberg NB. New developments in comorbidities of atopic dermatitis. Cutis. 2014;93:222-224.
- Kiken DA, Silverberg NB. Atopic dermatitis in children, part 1: epidemiology, clinical features, and complications. Cutis. 2006;78:241-247.
- Mathes EF, Oza V, Frieden IJ, et al. “Eczema coxsackium” and unusual cutaneous findings in an enterovirus outbreak. Pediatrics. 2013;132:E149-E157.
- Vora RV, Pilani AP, Jivani NB, et al. Kaposi varicelliform eruption. Indian Dermatol Online J. 2015;6:364-366.
- Hata TR, Kotol P, Boguniewicz M, et al. History of eczema herpeticum is associated with the inability to induce human β-defensin (HBD)-2, HBD-3 and cathelicidin in the skin of patients with atopic dermatitis. Br J Dermatol. 2010;163:659-661.
- Abeck D, Mempel M. Staphylococcus aureus colonization in atopic dermatitis and its therapeutic implications. Br J Dermatol. 1998;139:13-16.
- Leyden JJ, Marples RR, Kligman AM. Staphylococcus aureus in the lesions of atopic dermatitis. Br J Dermatol. 1974;90:525-530.
- Lipnharski C, d’Azevedo PA, Quinto VP, et al. Colonization by S. aureus increases the EASI and the number of appointments by patients with atopic dermatitis: cohort with 93 patients. An Bras Dermatol. 2013;88:518-521.
- Paller AS. Latest approaches to treating atopic dermatitis. Chem Immunol Allergy. 2012;96:132-140.
- Groner A, Laing-Grayman D, Silverberg NB. Outpatient pediatric community-acquired methicillin-resistant Staphylococcus aureus: a polymorphous clinical disease. Cutis. 2008;81:115-122.
- Sugarman JL, Hersh AL, Okamura T, et al. A retrospective review of streptococcal infections in pediatric atopic dermatitis. Pediatr Dermatol. 2011;28:230-234.
- Heath C, Desai N, Silverberg NB. Recent microbiological shifts in perianal bacterial dermatitis: Staphylococcus aureus predominance. Pediatr Dermatol. 2009;26:696-700.
- Garg N, Silverberg JI. Association between childhood allergic disease, psychological comorbidity, and injury requiring medical attention. Ann Allergy Asthma Immunol. 2014;112:525-532.
- Lavery MJ, Stull C, Kinney MO, et al. Nocturnal pruritus: the battle for a peaceful night’s sleep. Int J Mol Sci. 2016;17:E425.
- Silverberg NB, Licht J, Friedler S, et al. Nickel contact hypersensitivity in children. Pediatr Dermatol. 2002;19:110-113.
- Aubert-Wastiaux H, Moret L, Le Rhun A, et al. Topical corticosteroid phobia in atopic dermatitis: a study of its nature, origins and frequency. Br J Dermatol. 2011;165:808-814.
- Kojima R, Fujiwara T, Matsuda A, et al. Factors associated with steroid phobia in caregivers of children with atopic dermatitis. Pediatr Dermatol. 2013;30:29-35.
- Silverberg JI. Health care utilization, patient costs, and access to care in US adults with eczema: a population-based study. JAMA Dermatol. 2015;151:743-752.
- Weinmann S, Kamtsiuris P, Henke KD, et al. The costs of atopy and asthma in children: assessment of direct costs and their determinants in a birth cohort. Pediatr Allergy Immunol. 2003;14:18-26.
- Fivenson D, Arnold RJ, Kaniecki DJ, et al. The effect of atopic dermatitis on total burden of illness and quality of life on adults and children in a large managed care organization. J Manag Care Pharm. 2002;8:333-342.
- Verboom P, Hakkaart-Van L, Sturkenboom M, et al. The cost of atopic dermatitis in the Netherlands: an international comparison. Br J Dermatol. 2002;147:716-724.
- Lapidus CS, Schwarz DF, Honig PJ. Atopic dermatitis in children: who cares? who pays? J Am Acad Dermatol. 1993;28:699-703.
- Carroll CL, Balkrishnan R, Feldman SR, et al. The burden of atopic dermatitis: impact on the patient, family, and society. Pediatr Dermatol. 2005;22:192-199.
- Ellis CN, Drake LA, Prendergast MM, et al. Cost of atopic dermatitis and eczema in the United States. J Am Acad Dermatol. 2002;46:361-370.
- Ellis CN, Prendergast MM, Tokar M, et al. Quantifying costs associated with atopic dermatitis. J Manag Care Pharm. 2003;9:278.
- Lee SW, Cheong SH, Byun JY, et al. Occupational hand eczema among nursing staffs in Korea: self-reported hand eczema and contact sensitization of hospital nursing staffs. J Dermatol. 2013;40:182-187.
- Kemp AS. Cost of illness of atopic dermatitis in children: a societal perspective. Pharmacoeconomics. 2003;21:105-113.
- Munro DD. Topical corticosteroid therapy and its effect on the hypothalamic-pituitary-adrenal axis. Dermatologica. 1976;152:173-180.
- Silverberg JI, Garg NK, Paller AS, et al. Sleep disturbances in adults with eczema are associated with impaired overall health: a US population-based study. J Invest Dermatol. 2015;135:56-66.
- Eichenfield LF. Improving outcomes in atopic dermatitis. for advances in dermatology. Dermatology Focus. 2015;34:1-6.
- Fleischer AB Jr. Black box warning for topical calcineurin inhibitors and the death of common sense. Dermatol Online J. 2006;12:2.
Practice Points
- Atopic dermatitis (AD) has a variety of comorbidities including psychosocial disorders, obesity, and infection.
- A variety of skin conditions can mimic AD.
- Atopic dermatitis can be complicated by coinfections.
Crisaborole’s safety holds up in long-term atopic dermatitis trial
SCOTTSDALE, ARIZ. – The phosphodiesterase-4 inhibitor crisaborole was well tolerated over 6 to 12 months, yielding no major safety signals during a multicenter, open-label extension study of patients with mild-to-moderate atopic dermatitis.
These safety results held up across age groups and over time, said Dr. Lawrence Eichenfield, a dermatologist at Children’s Hospital, San Diego, and at the University of San Diego School of Medicine. “The majority of treatment-emergent adverse events were considered mild to moderate and not related to treatment. There were no reports of long-term cutaneous reactions, such as atrophy or telangiectasia,” he and his associates added.
Atopic dermatitis has lacked widely accepted treatment options. Despite attempts to educate patients and parents about topical steroids, many are afraid to use them, and topical calcineurin inhibitors have a black box warning for cancer risk. Not surprisingly, therefore, a 2% ointment of crisaborole made headlines in 2015 after meeting its efficacy and safety endpoints in two pivotal phase III trials of patients with mild-to-moderate atopic dermatitis. Based on those results, Anacor Pharmaceuticals filed a new drug application for the novel boron-based small molecule in January 2016.
The pivotal trials lasted just 28 days, so to assess long-term safety, Dr. Eichenfield and his associates enrolled a subgroup of 517 patients aged 2 to 72 years into a single-arm, open-label, 48-week extension study of crisaborole. About 31% of participants had received the control vehicle during the pivotal trials, while the rest had received crisaborole and tolerated it well enough to continue using it. Patients applied crisaborole twice daily during treatment cycles of 28 days, and were evaluated on days 1, 8, and 29 for up to 12 treatment cycles. Patients whose skin became clear or almost clear went off treatment, but they were still assessed for adverse effects at the same frequency.
In all, 396 patients used crisaborole for at least 6 months, and 271 completed 12 months of treatment, the researchers reported at the annual meeting of the Society for Investigative Dermatology. Only nine (1.7%) patients stopped treatment during the extension study because of treatment-emergent adverse effects. A total of 65% of patients had at least one treatment-emergent adverse event during the initial phase III trials, the extension study, or both. These were usually mildly or moderately severe and included nasopharyngitis, upper respiratory infections, cough, and/or fever, all of which were considered unrelated to treatment.
Treatment-related adverse events included flares of atopic dermatitis, burning or stinging at the application site, and application site infection, which affected 3.1%, 2.3%, and 1.2%, respectively, of patients in the extension study. None of these events were considered serious. Notably, 11% of patients experienced atopic dermatitis flares in the original phase III trials, the researchers reported. Patients who could not tolerate crisaborole were excluded from the extension study, which might help explain the lower flare rate (3%) with long-term treatment.
“Crisaborole topical ointment, 2%, demonstrated a favorable long-term safety profile for the treatment of patients aged 2 years and older with mild-to-moderate atopic dermatitis,” the researchers concluded. The Food and Drug Administration accepted the new drug application in March.
Anacor Pharmaceuticals makes crisaborole and funded the study. Dr. Eichenfield has served as an investigator and consultant to Anacor. Three coinvestigators also reported affiliations with Anacor.
SCOTTSDALE, ARIZ. – The phosphodiesterase-4 inhibitor crisaborole was well tolerated over 6 to 12 months, yielding no major safety signals during a multicenter, open-label extension study of patients with mild-to-moderate atopic dermatitis.
These safety results held up across age groups and over time, said Dr. Lawrence Eichenfield, a dermatologist at Children’s Hospital, San Diego, and at the University of San Diego School of Medicine. “The majority of treatment-emergent adverse events were considered mild to moderate and not related to treatment. There were no reports of long-term cutaneous reactions, such as atrophy or telangiectasia,” he and his associates added.
Atopic dermatitis has lacked widely accepted treatment options. Despite attempts to educate patients and parents about topical steroids, many are afraid to use them, and topical calcineurin inhibitors have a black box warning for cancer risk. Not surprisingly, therefore, a 2% ointment of crisaborole made headlines in 2015 after meeting its efficacy and safety endpoints in two pivotal phase III trials of patients with mild-to-moderate atopic dermatitis. Based on those results, Anacor Pharmaceuticals filed a new drug application for the novel boron-based small molecule in January 2016.
The pivotal trials lasted just 28 days, so to assess long-term safety, Dr. Eichenfield and his associates enrolled a subgroup of 517 patients aged 2 to 72 years into a single-arm, open-label, 48-week extension study of crisaborole. About 31% of participants had received the control vehicle during the pivotal trials, while the rest had received crisaborole and tolerated it well enough to continue using it. Patients applied crisaborole twice daily during treatment cycles of 28 days, and were evaluated on days 1, 8, and 29 for up to 12 treatment cycles. Patients whose skin became clear or almost clear went off treatment, but they were still assessed for adverse effects at the same frequency.
In all, 396 patients used crisaborole for at least 6 months, and 271 completed 12 months of treatment, the researchers reported at the annual meeting of the Society for Investigative Dermatology. Only nine (1.7%) patients stopped treatment during the extension study because of treatment-emergent adverse effects. A total of 65% of patients had at least one treatment-emergent adverse event during the initial phase III trials, the extension study, or both. These were usually mildly or moderately severe and included nasopharyngitis, upper respiratory infections, cough, and/or fever, all of which were considered unrelated to treatment.
Treatment-related adverse events included flares of atopic dermatitis, burning or stinging at the application site, and application site infection, which affected 3.1%, 2.3%, and 1.2%, respectively, of patients in the extension study. None of these events were considered serious. Notably, 11% of patients experienced atopic dermatitis flares in the original phase III trials, the researchers reported. Patients who could not tolerate crisaborole were excluded from the extension study, which might help explain the lower flare rate (3%) with long-term treatment.
“Crisaborole topical ointment, 2%, demonstrated a favorable long-term safety profile for the treatment of patients aged 2 years and older with mild-to-moderate atopic dermatitis,” the researchers concluded. The Food and Drug Administration accepted the new drug application in March.
Anacor Pharmaceuticals makes crisaborole and funded the study. Dr. Eichenfield has served as an investigator and consultant to Anacor. Three coinvestigators also reported affiliations with Anacor.
SCOTTSDALE, ARIZ. – The phosphodiesterase-4 inhibitor crisaborole was well tolerated over 6 to 12 months, yielding no major safety signals during a multicenter, open-label extension study of patients with mild-to-moderate atopic dermatitis.
These safety results held up across age groups and over time, said Dr. Lawrence Eichenfield, a dermatologist at Children’s Hospital, San Diego, and at the University of San Diego School of Medicine. “The majority of treatment-emergent adverse events were considered mild to moderate and not related to treatment. There were no reports of long-term cutaneous reactions, such as atrophy or telangiectasia,” he and his associates added.
Atopic dermatitis has lacked widely accepted treatment options. Despite attempts to educate patients and parents about topical steroids, many are afraid to use them, and topical calcineurin inhibitors have a black box warning for cancer risk. Not surprisingly, therefore, a 2% ointment of crisaborole made headlines in 2015 after meeting its efficacy and safety endpoints in two pivotal phase III trials of patients with mild-to-moderate atopic dermatitis. Based on those results, Anacor Pharmaceuticals filed a new drug application for the novel boron-based small molecule in January 2016.
The pivotal trials lasted just 28 days, so to assess long-term safety, Dr. Eichenfield and his associates enrolled a subgroup of 517 patients aged 2 to 72 years into a single-arm, open-label, 48-week extension study of crisaborole. About 31% of participants had received the control vehicle during the pivotal trials, while the rest had received crisaborole and tolerated it well enough to continue using it. Patients applied crisaborole twice daily during treatment cycles of 28 days, and were evaluated on days 1, 8, and 29 for up to 12 treatment cycles. Patients whose skin became clear or almost clear went off treatment, but they were still assessed for adverse effects at the same frequency.
In all, 396 patients used crisaborole for at least 6 months, and 271 completed 12 months of treatment, the researchers reported at the annual meeting of the Society for Investigative Dermatology. Only nine (1.7%) patients stopped treatment during the extension study because of treatment-emergent adverse effects. A total of 65% of patients had at least one treatment-emergent adverse event during the initial phase III trials, the extension study, or both. These were usually mildly or moderately severe and included nasopharyngitis, upper respiratory infections, cough, and/or fever, all of which were considered unrelated to treatment.
Treatment-related adverse events included flares of atopic dermatitis, burning or stinging at the application site, and application site infection, which affected 3.1%, 2.3%, and 1.2%, respectively, of patients in the extension study. None of these events were considered serious. Notably, 11% of patients experienced atopic dermatitis flares in the original phase III trials, the researchers reported. Patients who could not tolerate crisaborole were excluded from the extension study, which might help explain the lower flare rate (3%) with long-term treatment.
“Crisaborole topical ointment, 2%, demonstrated a favorable long-term safety profile for the treatment of patients aged 2 years and older with mild-to-moderate atopic dermatitis,” the researchers concluded. The Food and Drug Administration accepted the new drug application in March.
Anacor Pharmaceuticals makes crisaborole and funded the study. Dr. Eichenfield has served as an investigator and consultant to Anacor. Three coinvestigators also reported affiliations with Anacor.
AT THE 2016 SID ANNUAL MEETING
Key clinical point: The topical phosphodiesterase-4 inhibitor crisaborole was safe and well tolerated for up to 48 weeks in patients with mild-to-moderate atopic dermatitis.
Major finding: The most common treatment–related adverse events were atopic dermatitis flare (3%), stinging and burning at the application site (2%), and application site infection (1%). None were serious.
Data source: A single-arm, multicenter, open-label, 48-week extension study of 517 patients with mild-to-moderate atopic dermatitis.
Disclosures: Anacor Pharmaceuticals makes crisaborole and funded the study. Dr. Eichenfield has served as an investigator and consultant to Anacor. Three coinvestigators also reported affiliations with Anacor.
Atopic dermatitis increases risk of ADHD, comorbidities add to it
Atopic dermatitis (AD) is associated with an increased risk for attention-deficit/hyperactivity disorder (ADHD) in children and adults, and a host of factors and comorbid conditions increase this risk, according to a study published in the British Journal of Dermatology.
Mark A. Strom and his associates at Northwestern University in Chicago examined data from 19 U.S. population-based surveys on 354,416 children ages 2-17 years and 34,613 adults over the age of 18 to determine if childhood and adult AD and the severity of AD were associated with ADHD. Additionally, the investigators sought to identify factors contributing to this association (Br J Dermatol. 2016 Apr 23. doi: 10.1111/bjd.14697).
The prevalence of ADHD was 9.4% in children with AD and 7.1% in children without AD. For children, the study results obtained from multivariate models adjusting for age, sex, sociodemographic variables, allergic disease, and health care utilization confirmed the previously established increase in risk for ADHD in those with AD for an adjusted odds ratio of 1.14. Severe AD and sleep disturbance were found to independently and synergistically contribute to the increased risk of ADHD. Also, the presence of AD in the absence of other allergic disease was associated with an increased risk for ADHD. Obesity, headaches, and anemia were found to further increase the risk for ADHD in those with AD.
Results from the adjusted multivariate models also established an increased risk for ADHD among adults with AD, for an adjusted odds ratio of 1.61. The presence of asthma, headaches, and insomnia increased this risk. The analysis additionally revealed that a body mass index indicative of underweight status in adults with AD was protective against the risk for developing ADHD.
Mr. Strom and his associates found a significant decrease in their initial odds ratios when they added outpatient health care visitation to their multivariate models, which resulted in an overall pooled odds ratio for the relationship between AD and ADHD just above statistical significance. They speculated that children with AD or ADHD would be more likely to visit health care providers, which could increase the likelihood for diagnosis of comorbid conditions.
Funding was provided by the Agency for Healthcare Research and Quality and the Dermatology Foundation. The authors reported no conflicts of interest.
Atopic dermatitis (AD) is associated with an increased risk for attention-deficit/hyperactivity disorder (ADHD) in children and adults, and a host of factors and comorbid conditions increase this risk, according to a study published in the British Journal of Dermatology.
Mark A. Strom and his associates at Northwestern University in Chicago examined data from 19 U.S. population-based surveys on 354,416 children ages 2-17 years and 34,613 adults over the age of 18 to determine if childhood and adult AD and the severity of AD were associated with ADHD. Additionally, the investigators sought to identify factors contributing to this association (Br J Dermatol. 2016 Apr 23. doi: 10.1111/bjd.14697).
The prevalence of ADHD was 9.4% in children with AD and 7.1% in children without AD. For children, the study results obtained from multivariate models adjusting for age, sex, sociodemographic variables, allergic disease, and health care utilization confirmed the previously established increase in risk for ADHD in those with AD for an adjusted odds ratio of 1.14. Severe AD and sleep disturbance were found to independently and synergistically contribute to the increased risk of ADHD. Also, the presence of AD in the absence of other allergic disease was associated with an increased risk for ADHD. Obesity, headaches, and anemia were found to further increase the risk for ADHD in those with AD.
Results from the adjusted multivariate models also established an increased risk for ADHD among adults with AD, for an adjusted odds ratio of 1.61. The presence of asthma, headaches, and insomnia increased this risk. The analysis additionally revealed that a body mass index indicative of underweight status in adults with AD was protective against the risk for developing ADHD.
Mr. Strom and his associates found a significant decrease in their initial odds ratios when they added outpatient health care visitation to their multivariate models, which resulted in an overall pooled odds ratio for the relationship between AD and ADHD just above statistical significance. They speculated that children with AD or ADHD would be more likely to visit health care providers, which could increase the likelihood for diagnosis of comorbid conditions.
Funding was provided by the Agency for Healthcare Research and Quality and the Dermatology Foundation. The authors reported no conflicts of interest.
Atopic dermatitis (AD) is associated with an increased risk for attention-deficit/hyperactivity disorder (ADHD) in children and adults, and a host of factors and comorbid conditions increase this risk, according to a study published in the British Journal of Dermatology.
Mark A. Strom and his associates at Northwestern University in Chicago examined data from 19 U.S. population-based surveys on 354,416 children ages 2-17 years and 34,613 adults over the age of 18 to determine if childhood and adult AD and the severity of AD were associated with ADHD. Additionally, the investigators sought to identify factors contributing to this association (Br J Dermatol. 2016 Apr 23. doi: 10.1111/bjd.14697).
The prevalence of ADHD was 9.4% in children with AD and 7.1% in children without AD. For children, the study results obtained from multivariate models adjusting for age, sex, sociodemographic variables, allergic disease, and health care utilization confirmed the previously established increase in risk for ADHD in those with AD for an adjusted odds ratio of 1.14. Severe AD and sleep disturbance were found to independently and synergistically contribute to the increased risk of ADHD. Also, the presence of AD in the absence of other allergic disease was associated with an increased risk for ADHD. Obesity, headaches, and anemia were found to further increase the risk for ADHD in those with AD.
Results from the adjusted multivariate models also established an increased risk for ADHD among adults with AD, for an adjusted odds ratio of 1.61. The presence of asthma, headaches, and insomnia increased this risk. The analysis additionally revealed that a body mass index indicative of underweight status in adults with AD was protective against the risk for developing ADHD.
Mr. Strom and his associates found a significant decrease in their initial odds ratios when they added outpatient health care visitation to their multivariate models, which resulted in an overall pooled odds ratio for the relationship between AD and ADHD just above statistical significance. They speculated that children with AD or ADHD would be more likely to visit health care providers, which could increase the likelihood for diagnosis of comorbid conditions.
Funding was provided by the Agency for Healthcare Research and Quality and the Dermatology Foundation. The authors reported no conflicts of interest.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Key clinical point: Several factors contribute to the increased risk for ADHD in children and adults with atopic dermatitis.
Major finding: Children and adults with atopic dermatitis had increased odds of having ADHD of 1.14, which were further increased by the presence of certain comorbidities.
Data sources: Nineteen U.S. population-based surveys on 354,416 children aged 2-17 and 34,613 adults over the age of 18.
Disclosures: Funding was provided by the Agency for Healthcare Research and Quality and the Dermatology Foundation. The authors reported no conflicts of interest.
Food allergy development linked to S. aureus colonization in children with AD
Staphylococcus aureus colonization is associated with development of food allergy in children with atopic dermatitis (AD), according to a letter to the editor from Dr. Andrea L. Jones and her associates.
In a study of 718 patients with AD, median food allergen–specific IgE levels to peanut were highest in patients with methicillin-resistant Staphylococcus aureus (MRSA) at 77.7 kilounits of allergen per liter. Patients with methicillin-sensitive S. aureus (MSSA) had median food allergen–specific IgE (sIgE) levels to peanut of 38.9 kUA/L, and patients without S. aureus had median sIgE levels to peanut of 4.3 kUA/L, below the 95% positive predictive value of oral food challenge reaction in patients of 14 kUA/L.
Total IgE levels were highest in AD patients with MRSA at 4,498 kU/L, but were also elevated in patients with MSSA at 2,709 kU/L, compared with 217 kU/L for patients without S. aureus colonization.
“Studies are needed to assess the association between S. aureus skin colonization and food allergy in patients with AD. Confirmation of our current observations opens up the possibility that therapy directed at eradicating S. aureus colonization will be important in the prevention of food allergen sensitization and possibly food allergy in patients with AD,” the investigators concluded.
Find the full letter in the Journal of Allergy and Clinical Immunology (2016 Apr. doi: 10.1016/j.jaci.2016.01.010).
Staphylococcus aureus colonization is associated with development of food allergy in children with atopic dermatitis (AD), according to a letter to the editor from Dr. Andrea L. Jones and her associates.
In a study of 718 patients with AD, median food allergen–specific IgE levels to peanut were highest in patients with methicillin-resistant Staphylococcus aureus (MRSA) at 77.7 kilounits of allergen per liter. Patients with methicillin-sensitive S. aureus (MSSA) had median food allergen–specific IgE (sIgE) levels to peanut of 38.9 kUA/L, and patients without S. aureus had median sIgE levels to peanut of 4.3 kUA/L, below the 95% positive predictive value of oral food challenge reaction in patients of 14 kUA/L.
Total IgE levels were highest in AD patients with MRSA at 4,498 kU/L, but were also elevated in patients with MSSA at 2,709 kU/L, compared with 217 kU/L for patients without S. aureus colonization.
“Studies are needed to assess the association between S. aureus skin colonization and food allergy in patients with AD. Confirmation of our current observations opens up the possibility that therapy directed at eradicating S. aureus colonization will be important in the prevention of food allergen sensitization and possibly food allergy in patients with AD,” the investigators concluded.
Find the full letter in the Journal of Allergy and Clinical Immunology (2016 Apr. doi: 10.1016/j.jaci.2016.01.010).
Staphylococcus aureus colonization is associated with development of food allergy in children with atopic dermatitis (AD), according to a letter to the editor from Dr. Andrea L. Jones and her associates.
In a study of 718 patients with AD, median food allergen–specific IgE levels to peanut were highest in patients with methicillin-resistant Staphylococcus aureus (MRSA) at 77.7 kilounits of allergen per liter. Patients with methicillin-sensitive S. aureus (MSSA) had median food allergen–specific IgE (sIgE) levels to peanut of 38.9 kUA/L, and patients without S. aureus had median sIgE levels to peanut of 4.3 kUA/L, below the 95% positive predictive value of oral food challenge reaction in patients of 14 kUA/L.
Total IgE levels were highest in AD patients with MRSA at 4,498 kU/L, but were also elevated in patients with MSSA at 2,709 kU/L, compared with 217 kU/L for patients without S. aureus colonization.
“Studies are needed to assess the association between S. aureus skin colonization and food allergy in patients with AD. Confirmation of our current observations opens up the possibility that therapy directed at eradicating S. aureus colonization will be important in the prevention of food allergen sensitization and possibly food allergy in patients with AD,” the investigators concluded.
Find the full letter in the Journal of Allergy and Clinical Immunology (2016 Apr. doi: 10.1016/j.jaci.2016.01.010).
FROM THE JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY
Vitamin D supplementation cuts dust mite atopy
BALTIMORE – Three months of daily, oral treatment with a relatively high but safe dosage of a vitamin D supplement to pregnant mothers during late gestation followed by continued oral supplementation to their neonates during the first 6 months of life led to a significant reduction in the prevalence of dust-mite skin reactivity in those children once they reached 18 months old in a randomized, controlled trial with 259 mothers and infants.
And in a preliminary assessment that tallied the number of children who required primary care office visits for asthma through age 18 months, children who had received the highest vitamin D supplementation also showed a statistically significant reduction of these visits, compared with the placebo control children, Dr. Cameron C. Grant reported at the annual meeting of the Pediatric Academic Societies.
This suggestion that the vitamin D intervention could cut asthma development is not completely certain because in 18-month-old children, diagnosis of asthma is “very insecure,” noted Dr. Grant, a pediatrician at the University of Auckland, New Zealand and at Starship Children’s Hospital, also in Auckland. In addition, a limitation of the observed effect on dust mite atopy on skin-test challenge was that this follow-up occurred in only 186 (72%) of the 259 infants who participated in the study.
The study’s premise was that vitamin D is an immune system modulator, and that New Zealand provides an excellent setting to test the hypothesis that normalized vitamin D levels can help prevent development of atopy and asthma because many of the country’s residents are vitamin D deficient due to their diet and sun avoidance to prevent skin cancers. Results from prior studies had shown that 57% of New Zealand neonates have inadequate levels of vitamin D at birth, defined as a serum level of 25-hydroxyvitamin D of less than 20 ng/ml (less than 50 nmol/L), Dr. Grant noted.
“I think this intervention will only work in populations that are vitamin D deficient,” Dr. Grant said in an interview. In his study, the average serum level of 25-hydroxyvitamin D among control neonates was 38 nmol/L (about 15 ng/mL). In contrast, neonates born to mothers who had received a daily, higher-dose vitamin D supplement during the third trimester had serum measures that were roughly twice that level.
The study enrolled 260 pregnant women from the Auckland area with a single pregnancy at 26-30 weeks’ gestation; average gestational age at baseline was 27 weeks. Dr. Grant and his associates randomized the mothers to receive 1,000 IU oral vitamin D daily, 2,000 oral vitamin D daily, or placebo. The women delivered 259 infants. Infants born to women on the lower dosage supplement then received 400 IU vitamin daily for 6 months, those born to mothers on the higher level supplement received 800 IU vitamin D daily for 6 months, and those born to mothers in the placebo group received placebo supplements daily for 6 months.
Both supplement regimens led to statistically significant increases in serum levels of 25-hydroxyvitamin D in maternal serum at 36 weeks’ gestation, in cord blood at delivery, in the neonates’ serum at ages 2 months and 4 months, and in infant serum in the higher dosage group at 6 months of age, compared with similar measures taken at all these time points in the placebo group.
In addition, the neonates in the higher dosage group had significantly higher serum levels at 2, 4, and 6 months, compared with the lower dosage group. When measured a final time at 18-month follow-up, a year after the end of vitamin D supplementation, average serum levels of 25-hydroxyvitamin D in an three subgroups of children were virtually identical and similar to maternal serum levels at baseline. Dr. Grant and his associates had previously reported these findings and also had documented the safety of both the low and high levels of vitamin D supplements for both mothers and their children (Pediatrics. 2014 Jan;133[1]:e143-53).
The new findings reported by Dr. Grant focused on clinical outcomes at 18 months. He and his colleagues ran skin-prick testing on 186 of the 259 (72%) children in the study (the remaining children weren’t available for this follow-up assessment). They tested three aeroallergens: cat, pollen, and house dust mite. They saw no significant differences in the prevalence of positive skin-prick reactions among the three study groups to cat and pollen, but prevalence levels of positive reactions to dust mite were 9% in the controls, 3% of children in the low-dosage group, and none in the high dosage group. The difference between the controls and high dosage groups was statistically significant; the difference between the controls and the low dosage group was not significant, Dr. Grant said. Additional testing of specific IgE responses to four different dust mite antigens showed statistically significant reductions in responses to each of the four antigens among the high dosage children, compared with the controls and with the low dosage children.
The researchers also tallied the number of acute, primary care office visits during the first 18 months of life among the children in each of the three subgroups for a variety of respiratory diagnoses. The three groups showed no significant differences in total number of office visits for most of these diagnoses, including colds, otitis media, croup, and bronchitis. However, about 12% of children in the control group had been seen in a primary care office for a diagnosis of asthma, compared with none of the children in the low dosage group and about 4% in the high-dosage group. The differences between the two intervention groups and the control group were statistically significant. Dr. Grant cautioned that this finding is very preliminary and that any conclusions about the impact of vitamin D supplements on asthma incidence must await studies with larger numbers of children who are followed to an older age.
Dr. Grant had no disclosures.
On Twitter @mitchelzoler
BALTIMORE – Three months of daily, oral treatment with a relatively high but safe dosage of a vitamin D supplement to pregnant mothers during late gestation followed by continued oral supplementation to their neonates during the first 6 months of life led to a significant reduction in the prevalence of dust-mite skin reactivity in those children once they reached 18 months old in a randomized, controlled trial with 259 mothers and infants.
And in a preliminary assessment that tallied the number of children who required primary care office visits for asthma through age 18 months, children who had received the highest vitamin D supplementation also showed a statistically significant reduction of these visits, compared with the placebo control children, Dr. Cameron C. Grant reported at the annual meeting of the Pediatric Academic Societies.
This suggestion that the vitamin D intervention could cut asthma development is not completely certain because in 18-month-old children, diagnosis of asthma is “very insecure,” noted Dr. Grant, a pediatrician at the University of Auckland, New Zealand and at Starship Children’s Hospital, also in Auckland. In addition, a limitation of the observed effect on dust mite atopy on skin-test challenge was that this follow-up occurred in only 186 (72%) of the 259 infants who participated in the study.
The study’s premise was that vitamin D is an immune system modulator, and that New Zealand provides an excellent setting to test the hypothesis that normalized vitamin D levels can help prevent development of atopy and asthma because many of the country’s residents are vitamin D deficient due to their diet and sun avoidance to prevent skin cancers. Results from prior studies had shown that 57% of New Zealand neonates have inadequate levels of vitamin D at birth, defined as a serum level of 25-hydroxyvitamin D of less than 20 ng/ml (less than 50 nmol/L), Dr. Grant noted.
“I think this intervention will only work in populations that are vitamin D deficient,” Dr. Grant said in an interview. In his study, the average serum level of 25-hydroxyvitamin D among control neonates was 38 nmol/L (about 15 ng/mL). In contrast, neonates born to mothers who had received a daily, higher-dose vitamin D supplement during the third trimester had serum measures that were roughly twice that level.
The study enrolled 260 pregnant women from the Auckland area with a single pregnancy at 26-30 weeks’ gestation; average gestational age at baseline was 27 weeks. Dr. Grant and his associates randomized the mothers to receive 1,000 IU oral vitamin D daily, 2,000 oral vitamin D daily, or placebo. The women delivered 259 infants. Infants born to women on the lower dosage supplement then received 400 IU vitamin daily for 6 months, those born to mothers on the higher level supplement received 800 IU vitamin D daily for 6 months, and those born to mothers in the placebo group received placebo supplements daily for 6 months.
Both supplement regimens led to statistically significant increases in serum levels of 25-hydroxyvitamin D in maternal serum at 36 weeks’ gestation, in cord blood at delivery, in the neonates’ serum at ages 2 months and 4 months, and in infant serum in the higher dosage group at 6 months of age, compared with similar measures taken at all these time points in the placebo group.
In addition, the neonates in the higher dosage group had significantly higher serum levels at 2, 4, and 6 months, compared with the lower dosage group. When measured a final time at 18-month follow-up, a year after the end of vitamin D supplementation, average serum levels of 25-hydroxyvitamin D in an three subgroups of children were virtually identical and similar to maternal serum levels at baseline. Dr. Grant and his associates had previously reported these findings and also had documented the safety of both the low and high levels of vitamin D supplements for both mothers and their children (Pediatrics. 2014 Jan;133[1]:e143-53).
The new findings reported by Dr. Grant focused on clinical outcomes at 18 months. He and his colleagues ran skin-prick testing on 186 of the 259 (72%) children in the study (the remaining children weren’t available for this follow-up assessment). They tested three aeroallergens: cat, pollen, and house dust mite. They saw no significant differences in the prevalence of positive skin-prick reactions among the three study groups to cat and pollen, but prevalence levels of positive reactions to dust mite were 9% in the controls, 3% of children in the low-dosage group, and none in the high dosage group. The difference between the controls and high dosage groups was statistically significant; the difference between the controls and the low dosage group was not significant, Dr. Grant said. Additional testing of specific IgE responses to four different dust mite antigens showed statistically significant reductions in responses to each of the four antigens among the high dosage children, compared with the controls and with the low dosage children.
The researchers also tallied the number of acute, primary care office visits during the first 18 months of life among the children in each of the three subgroups for a variety of respiratory diagnoses. The three groups showed no significant differences in total number of office visits for most of these diagnoses, including colds, otitis media, croup, and bronchitis. However, about 12% of children in the control group had been seen in a primary care office for a diagnosis of asthma, compared with none of the children in the low dosage group and about 4% in the high-dosage group. The differences between the two intervention groups and the control group were statistically significant. Dr. Grant cautioned that this finding is very preliminary and that any conclusions about the impact of vitamin D supplements on asthma incidence must await studies with larger numbers of children who are followed to an older age.
Dr. Grant had no disclosures.
On Twitter @mitchelzoler
BALTIMORE – Three months of daily, oral treatment with a relatively high but safe dosage of a vitamin D supplement to pregnant mothers during late gestation followed by continued oral supplementation to their neonates during the first 6 months of life led to a significant reduction in the prevalence of dust-mite skin reactivity in those children once they reached 18 months old in a randomized, controlled trial with 259 mothers and infants.
And in a preliminary assessment that tallied the number of children who required primary care office visits for asthma through age 18 months, children who had received the highest vitamin D supplementation also showed a statistically significant reduction of these visits, compared with the placebo control children, Dr. Cameron C. Grant reported at the annual meeting of the Pediatric Academic Societies.
This suggestion that the vitamin D intervention could cut asthma development is not completely certain because in 18-month-old children, diagnosis of asthma is “very insecure,” noted Dr. Grant, a pediatrician at the University of Auckland, New Zealand and at Starship Children’s Hospital, also in Auckland. In addition, a limitation of the observed effect on dust mite atopy on skin-test challenge was that this follow-up occurred in only 186 (72%) of the 259 infants who participated in the study.
The study’s premise was that vitamin D is an immune system modulator, and that New Zealand provides an excellent setting to test the hypothesis that normalized vitamin D levels can help prevent development of atopy and asthma because many of the country’s residents are vitamin D deficient due to their diet and sun avoidance to prevent skin cancers. Results from prior studies had shown that 57% of New Zealand neonates have inadequate levels of vitamin D at birth, defined as a serum level of 25-hydroxyvitamin D of less than 20 ng/ml (less than 50 nmol/L), Dr. Grant noted.
“I think this intervention will only work in populations that are vitamin D deficient,” Dr. Grant said in an interview. In his study, the average serum level of 25-hydroxyvitamin D among control neonates was 38 nmol/L (about 15 ng/mL). In contrast, neonates born to mothers who had received a daily, higher-dose vitamin D supplement during the third trimester had serum measures that were roughly twice that level.
The study enrolled 260 pregnant women from the Auckland area with a single pregnancy at 26-30 weeks’ gestation; average gestational age at baseline was 27 weeks. Dr. Grant and his associates randomized the mothers to receive 1,000 IU oral vitamin D daily, 2,000 oral vitamin D daily, or placebo. The women delivered 259 infants. Infants born to women on the lower dosage supplement then received 400 IU vitamin daily for 6 months, those born to mothers on the higher level supplement received 800 IU vitamin D daily for 6 months, and those born to mothers in the placebo group received placebo supplements daily for 6 months.
Both supplement regimens led to statistically significant increases in serum levels of 25-hydroxyvitamin D in maternal serum at 36 weeks’ gestation, in cord blood at delivery, in the neonates’ serum at ages 2 months and 4 months, and in infant serum in the higher dosage group at 6 months of age, compared with similar measures taken at all these time points in the placebo group.
In addition, the neonates in the higher dosage group had significantly higher serum levels at 2, 4, and 6 months, compared with the lower dosage group. When measured a final time at 18-month follow-up, a year after the end of vitamin D supplementation, average serum levels of 25-hydroxyvitamin D in an three subgroups of children were virtually identical and similar to maternal serum levels at baseline. Dr. Grant and his associates had previously reported these findings and also had documented the safety of both the low and high levels of vitamin D supplements for both mothers and their children (Pediatrics. 2014 Jan;133[1]:e143-53).
The new findings reported by Dr. Grant focused on clinical outcomes at 18 months. He and his colleagues ran skin-prick testing on 186 of the 259 (72%) children in the study (the remaining children weren’t available for this follow-up assessment). They tested three aeroallergens: cat, pollen, and house dust mite. They saw no significant differences in the prevalence of positive skin-prick reactions among the three study groups to cat and pollen, but prevalence levels of positive reactions to dust mite were 9% in the controls, 3% of children in the low-dosage group, and none in the high dosage group. The difference between the controls and high dosage groups was statistically significant; the difference between the controls and the low dosage group was not significant, Dr. Grant said. Additional testing of specific IgE responses to four different dust mite antigens showed statistically significant reductions in responses to each of the four antigens among the high dosage children, compared with the controls and with the low dosage children.
The researchers also tallied the number of acute, primary care office visits during the first 18 months of life among the children in each of the three subgroups for a variety of respiratory diagnoses. The three groups showed no significant differences in total number of office visits for most of these diagnoses, including colds, otitis media, croup, and bronchitis. However, about 12% of children in the control group had been seen in a primary care office for a diagnosis of asthma, compared with none of the children in the low dosage group and about 4% in the high-dosage group. The differences between the two intervention groups and the control group were statistically significant. Dr. Grant cautioned that this finding is very preliminary and that any conclusions about the impact of vitamin D supplements on asthma incidence must await studies with larger numbers of children who are followed to an older age.
Dr. Grant had no disclosures.
On Twitter @mitchelzoler
AT THE PAS ANNUAL MEETING
Key clinical point: Maternal treatment to achieve adequate vitamin D levels during late gestation followed by neonatal vitamin D supplementation significantly cut dust mite atopy at 18 months of age, along with a suggestion of reduced asthma incidence.
Major finding: Dust mite reactivity at 18 months occurred in no children treated with higher vitamin D supplementation and in 9% of controls.
Data source: A randomized, controlled, single-center study with 260 pregnant women who delivered 259 infants.
Disclosures: Dr. Grant had no disclosures.
A Practical Overview of Pediatric Atopic Dermatitis, Part 2: Triggers and Grading
Atopic dermatitis (AD) may be triggered by viral infections, food allergens, weather, and other causes, and it may trigger an inflammatory progression known as the atopic march. This article reviews research on triggers of pediatric AD so that dermatologists may discuss trigger avoidance with patients and guardians. Other factors affecting AD development include genetics and hygiene. Grading of AD also is discussed.
The Atopic March
The persistence of AD in untreated skin can trigger an inflammatory progression called the atopic march in which food and environmental allergies as well as asthma may occur progressively due to ongoing inflammatory triggering.1 In a study of asthma and food allergy reporting and management in public schools in Chicago, Illinois, food allergies were seen in 9.3% of asthmatic students (n=18,000), and 40.1% of food allergic students (n=4000) had asthma.2 An observational study by Flohr et al3 in London, England, included 619 exclusively breastfed infants who were recruited at 3 months of age. The investigators determined that food sensitization was unrelated to the presence of filaggrin mutations, type of eczema (flexural vs nonflexural), and transepidermal water loss but was associated with AD severity as determined by SCORAD (SCORing Atopic Dermatitis), a composite score of AD that includes pruritus as a factor in severity. Other AD associations included 3 leading food allergens: eggs, milk, and peanuts. No association with cod, wheat, or sesame allergy was noted. The investigators concluded that AD and AD severity were the leading skin-related risk factors for food allergies and therefore food allergy development in breastfed infants was probably mediated by cutaneous antigen-presenting cells.3
The skin has been documented to react to contact with known food allergens4 and is known to be a route of allergic sensitization to allergens such as fragrance in patients with AD.5,6 Two phenotypes of eczema that have been associated with asthma development are severe AD disease and multiple environmental allergies, supporting the theory of the atopic march.7 There also is evidence that release of danger-associated proteins from an impaired barrier also may trigger asthma.8 An analysis of the 2007 National Survey of Children’s Health, a population-based study of91,642 children aged 0 to 17 years, showed that children with AD had a higher prevalence of comorbid asthma (25.1% vs 12.3%), hay fever (34.4% vs 14.3%), and food allergies (15.1% vs 3.6%) compared to children without AD.9 A recent article provided detailed information on how food and diet interplay with AD.10
Triggers of Disease Flares
Triggers are the leading source of AD flare initiation, and avoidance of triggers is an important mechanism by which patients can control disease activity. Despite the best skin care and trigger avoidance, disease flares occur, sometimes due to ongoing inflammation and other times due to inability to prevent flares such as heat and humidity. A survey of patients with AD in Spain identified the following triggers: cosmetic products, clothing, mites, detergents/soaps, and temperature changes.11 In childhood, wool also is a known trigger of AD.12 Viral infections including respiratory syncytial virus may trigger the first onset of AD.13 Patients with AD may become allergic to fragrance and metals causing disease exacerbation on exposure.14,15 Food allergens contribute to approximately 40% of cases of AD in infancy but are not the cause of AD. The best evidence for improvement of AD with food allergen avoidance exists for egg white allergy.16 Food avoidance programs should be developed in conjunction with an allergist, as it is no longer advised in many cases to completely withdraw foods; therefore, an allergist has to assess the level of allergic severity and the risk-benefit ratio of food avoidance or introduction.17 Emotional stressors, heat, and humidity, as well as indoor heating in the winter months, can cause AD flares.18
A study by Silverberg et al19 provided evidence of climate influences on the US prevalence of childhood eczema using a merged analysis of the 2007 National Survey of Children’s Health and the 2006-2007 National Climate Data Center and Weather Service. Results showed that eczema prevalence was significantly lower when associated with higher annual relative humidity (P=.01), UV index (P<.0001), and highest-quartile air temperature (P=.002).19 The Pediatric Eczema Elective Registry also showed that warm, humid, and high-sun-exposure climates are associated with poorly controlled eczema in affected patients.20 The association of eczema with latitude as well as its negative association with mean annual outdoor temperature has been described by Weiland et al21 in the ISAAC (International Study of Asthma and Allergies in Childhood) study. Long airplane flights in low humidity can trigger eczema in adults. Climate has been postulated to affect eczema through alterations in filaggrin and skin barrier function.22 Indoor temperature and humidity regulation may be used adjunctively for daily flare prevention.
Genetics and AD
Of 762 infants in a birth cohort with a parent with atopy in Cincinnati, Ohio, 39% developed eczema by the age of 3 years. Single nucleotide polymorphisms of IL-4Rα 175 V and CD14-159 C/T were linked to greater eczema risk at 2 to 3 years of age.23 Monozygotic twins have a concordance rate of 0.72 to 0.86 versus 0.21 to 0.23 in dizygotic twins, demonstrating a strong genetic component in the development of AD.24 Linkage to AD has been positively made to the epidermal differentiation complex on human chromosome 1q21, which contains the genes for filaggrin and other proteins such as loricrin. Other genes linked to AD include the serine protease inhibitor SPINK5 (serine peptidase inhibitor, Kazal type 5) implicated in Netherton syndrome (triad of ichthyosis linearis circumflexa, bamboo hair, and atopic disorders); RANTES (regulated on activation, normal T-expressed, and secreted), which has been associated with severity of AD; IL-4; and IL-13.5,25,26
The Hygiene Hypothesis
Atopic dermatitis is more common in wealthy developed countries, leading some to believe that hygiene and relative reduction in illness via vaccination have contributed to the rise of AD prevalence in developed nations.13,27 There currently is evidence demonstrating that wild-type varicella infection confers long-standing protection against AD and mediates reduced total IgE and peripheral blood lymphocytes.27
Grading of AD
Grading of AD is a subject of controversy, as there currently are no uniform grading scales.28 A recent outcomes group attempted to determine the best scale for disease monitoring. Schmitt et al29 presented the Harmonizing Outcome Measures for Eczema (HOME) roadmap, which was intended to determine a core outcome set for eczema; however, because these outcome measurements have not yet been standardized, only the eczema assessment and severity index (EASI) scoring system meets criteria for standardization. In clinical practice, physicians often assign mild, moderate, or severe labeling based on their general sense of the disease extent using an investigator global assessment score.28
The EASI score is a well-validated composite score of AD severity based on 4 body regions: (1) head and neck, (2) trunk (including genital area), (3) upper limbs, and (4) lower limbs (including buttocks). The total area of involvement in each region is graded on a scale of 0 to 6, and AD severity is graded as a composite of 4 parameters (ranked on a scale of 0–3), including redness (erythema, inflammation), thickness (induration, papulation, swelling [acute eczema]), scratching (excoriation), and lichenification (prurigo nodules [chronic eczema]). The surface area of each region relative to body size is used as a multiplying factor, resulting in the following severity strata: 0=clear; 0.1–1.0=almost clear; 1.1–7.0=mild; 7.1–21.0=moderate; 21.1–50.0=severe; 50.1–72.0=very severe (κ=0.75).30-32 The six area, six sign AD (SASSAD) score32,33 is a similar score without adjustment for body surface area by region.34
An older, now less frequently used eczema score is the SCORAD, which addressed surface area by rule of nines and severity of 6 features—redness, swelling, oozing/crusting, scratch marks, skin thickening (lichenification), dryness (assessed in an area with no inflammation)—by region on a scale of 0 to 3. A subjective symptom parameter for itching and sleeplessness helped highlight that these comorbidities are important in gauging disease activity and impact on a child’s life.35
Natural History of AD
The clinical dogma has been that AD would improve with age, with reduction at grade school entry and perhaps full disappearance in adulthood; however, 3 recent surveys have suggested otherwise. The ISAAC group has found prevalence of AD in wealthy developed countries among children aged 6 to 7 years to be at a consistent increase.36 A US-based survey from the National Health Interview Survey showed a 1-year prevalence of 10.2% of active AD in adults and 9.8% when occupational dermatitis was excluded.37 Halvorsen et al38 demonstrated that eczema prevalence is 9.7% in individuals aged 18 to 19 years.
A prospective trial of eighth graders followed from 1995 to 2010 demonstrated that AD persisted in 50% at school age. Persistent eczema into adulthood was associated with early-onset childhood allergic rhinitis and hand eczema.39 In a cohort of hand eczema patients (N=368), 28% had AD and 39% had an atopic illness.40 An association with allergic contact dermatitis and increased IgE to Malassezia furfur was further associated.41
Conclusion
The role of triggers and allergens in disease activity in AD is an important consideration in children with AD and requires ongoing consideration with age and varied exposures. Understanding the grading of AD is important in evaluating clinical trial data. The natural history of AD has changed, which is important for the practitioner to note when counseling patients and guardians.
- Li M. Current evidence of epidermal barrier dysfunction and thymic stromal lymphopoietin in the atopic march. Eur Respir Rev. 2014;23:292-298.
- Gupta RS, Rivkina V, DeSantiago-Cardenas L, et al. Asthma and food allergy management in Chicago public schools. Pediatrics. 2014;134:729-736.
- Flohr C, Perkin M, Logan K, et al. Atopic dermatitis and disease severity are the main risk factors for food sensitization in exclusively breastfed infants. J Invest Dermatol. 2014;134:345-350.
- Silverberg NB. Food, glorious food. Cutis. 2011;87:267-268.
- De Benedetto A, Kubo A, Beck LA. Skin barrier disruption: a requirement for allergen sensitization? J Invest Dermatol. 2012;132:949-963.
- Thyssen JP, McFadden JP, Kimber I. The multiple factors affecting the association between atopic dermatitis and contact sensitization. Allergy. 2014;69:28-36.
- Amat F, Saint-Pierre P, Bourrat E, et al. Early-onset atopic dermatitis in children: which are the phenotypes at risk of asthma? results from the ORCA Cohort. PLoS One. 2015;10:e0131369.
- Demehri S, Morimoto M, Holtzman MJ, et al. Skin-derived TSLP triggers progression from epidermal-barrier defects to asthma. PLoS Biol. 2009;7:e1000067.
- Silverberg JI, Simpson EL. Association between severe eczema in children and multiple comorbid conditions and increased healthcare utilization. Pediatr Allergy Immunol. 2013;24:476-486.
- Silverberg NB, Lee-Wong M, Yosipovitch G. Diet and atopic dermatitis. Cutis. 2016;97:227-232.
- Ortiz de Frutos FJ, Torrelo A, de Lucas R, et al. Patient perspectives on triggers, adherence to medical recommendations, and disease control in atopic dermatitis: the DATOP study. Actas Dermosifiliogr. 2014;105:487-496.
- Ricci G, Patrizi A, Bellini F, et al. Use of textiles in atopic dermatitis: care of atopic dermatitis. Curr Probl Dermatol. 2006;33:127-143.
- Welliver RC, Wong DT, Sun M, et al. The development of respiratory syncytial virus-specific IgE and the release of histamine in nasopharyngeal secretions after infection. N Engl J Med. 1981;305:841-846.
- Aquino M, Fonacier L. The role of contact dermatitis in patients with atopic dermatitis. J Allergy Clin Immunol Pract. 2014;2:382-387.
- Brod BA, Treat JR, Rothe MJ, et al. Allergic contact dermatitis: kids are not just little people. Clin Dermatol. 2015;33:605-612.
- Martorell A, Alonso E, Boné J, et al. Position document: IgE-mediated allergy to egg protein. Allergol Immunopathol (Madr). 2013;41:320-336.
- Sicherer SH. Early introduction of peanut to infants at high allergic risk can reduce peanut allergy at age 5 years [published online September 17, 2015]. Evid Based Med. 2015;20:204.
- Kiken DA, Silverberg NB. Atopic dermatitis in children, part 1: epidemiology, clinical features, and complications. Cutis. 2006;78:241-247.
- Silverberg JI, Hanifin J, Simpson EL. Climatic factors are associated with childhood eczema prevalence in the United States. J Invest Dermatol. 2013;133:1752-1759.
- Sargen MR, Hoffstad O, Margolis DJ. Warm, humid, and high sun exposure climates are associated with poorly controlled eczema: PEER (Pediatric Eczema Elective Registry) cohort, 2004-2012. J Invest Dermatol. 2014;134:51-57.
- Weiland SK, Hüsing A, Strachan DP, et al. Climate and the prevalence of symptoms of asthma, allergic rhinitis, and atopic eczema in children. Occup Environ Med. 2004;61:609-615.
- Langan SM, Irvine AD. Childhood eczema and the importance of the physical environment. J Invest Dermatol. 2013;133:1706-1709.
- Biagini Myers JM, Wang N, LeMasters GK, et al. Genetic and environmental risk factors for childhood eczema development and allergic sensitization in the CCAAPS cohort. J Invest Dermatol. 2010;130:430-437.
- Brown SJ, McLean WH. Eczema genetics: current state of knowledge and future goals. J Invest Dermatol. 2009;129:543-552.
- Hanifin JM. Evolving concepts of pathogenesis in atopic dermatitis and other eczemas. J Invest Dermatol. 2009;129:320-322.
- Paller AS. Latest approaches to treating atopic dermatitis. Chem Immunol Allergy. 2012;96:132-140.
- Silverberg JI, Norowitz KB, Kleiman E, et al. Association between varicella zoster virus infection and atopic dermatitis in early and late childhood: a case-control study. J Allergy Clin Immunol. 2010;126:300-305.
- Futamura M, Leshem YA, Thomas KS, et al. A systematic review of Investigator Global Assessment (IGA) in atopic dermatitis (AD) trials: many options, no standards. J Am Acad Dermatol. 2016;74:288-294.
- Schmitt J, Apfelbacher C, Spuls PI, et al. The Harmonizing Outcome Measures for Eczema (HOME) roadmap: a methodological framework to develop core sets of outcome measurements in dermatology. J Invest Dermatol. 2015;135:24-30.
- Hanifin JM, Thurston M, Omoto M, et al. The eczema area and severity index (EASI): assessment of reliability in atopic dermatitis. EASI Evaluator Group. Exp Dermatol. 2001;10:11-18.
- Leshem YA, Hajar T, Hanifin JM, et al. What the Eczema Area and Severity Index score tells us about the severity of atopic dermatitis: an interpretability study. Br J Dermatol. 2015;172:1353-1357.
- Barbier N, Paul C, Luger T, et al. Validation of the Eczema Area and Severity Index for atopic dermatitis in a cohort of 1550 patients from the pimecrolimus cream 1% randomized controlled clinical trials programme. Br J Dermatol. 2004;150:96-102.
- Berth-Jones J. Six area, six sign atopic dermatitis (SASSAD) severity score: a simple system for monitoring disease activity in atopic dermatitis. Br J Dermatol. 1996;135(suppl 48):25-30.
- Zhao CY, Tran AQ, Lazo-Dizon JP, et al. A pilot comparison study of four clinician-rated atopic dermatitis severity scales. Br J Dermatol. 2015;173:488-497.
- Kunz B, Oranje AP, Labrèze L, et al. Clinical validation and guidelines for the SCORAD index: consensus report of the European Task Force on Atopic Dermatitis. Dermatology. 1997;195:10-19.
- Williams H, Stewart A, von Mutius E, et al. Is eczema really on the increase worldwide? J Allergy Clin Immunol. 2008;121:947-954.
- Silverberg JI, Hanifin JM. Adult eczema prevalence and associations with asthma and other health and demographic factors: a US population-based study. J Allergy Clin Immunol. 2013;132:1132-1138.
- Halvorsen JA, Lien L, Dalgard F, et al. Suicidal ideation, mental health problems, and social function in adolescents with eczema: a population-based study. J Invest Dermatol. 2014;134:1847-1854.
- Mortz CG, Andersen KE, Dellgren C, et al. Atopic dermatitis from adolescence to adulthood in the TOACS cohort: prevalence, persistence, and comorbidities. Allergy. 2015;70:836-845.
- Rystedt I. Atopic background in patients with occupational hand eczema. Contact Dermatitis. 1985;12:247-254.
- Mortz CG, Andersen KE, Dellgren C, et al. Atopic dermatitis from adolescence to adulthood in the TOACS cohort: prevalence, persistence and comorbidities. Allergy. 2015;70:836-845.
Atopic dermatitis (AD) may be triggered by viral infections, food allergens, weather, and other causes, and it may trigger an inflammatory progression known as the atopic march. This article reviews research on triggers of pediatric AD so that dermatologists may discuss trigger avoidance with patients and guardians. Other factors affecting AD development include genetics and hygiene. Grading of AD also is discussed.
The Atopic March
The persistence of AD in untreated skin can trigger an inflammatory progression called the atopic march in which food and environmental allergies as well as asthma may occur progressively due to ongoing inflammatory triggering.1 In a study of asthma and food allergy reporting and management in public schools in Chicago, Illinois, food allergies were seen in 9.3% of asthmatic students (n=18,000), and 40.1% of food allergic students (n=4000) had asthma.2 An observational study by Flohr et al3 in London, England, included 619 exclusively breastfed infants who were recruited at 3 months of age. The investigators determined that food sensitization was unrelated to the presence of filaggrin mutations, type of eczema (flexural vs nonflexural), and transepidermal water loss but was associated with AD severity as determined by SCORAD (SCORing Atopic Dermatitis), a composite score of AD that includes pruritus as a factor in severity. Other AD associations included 3 leading food allergens: eggs, milk, and peanuts. No association with cod, wheat, or sesame allergy was noted. The investigators concluded that AD and AD severity were the leading skin-related risk factors for food allergies and therefore food allergy development in breastfed infants was probably mediated by cutaneous antigen-presenting cells.3
The skin has been documented to react to contact with known food allergens4 and is known to be a route of allergic sensitization to allergens such as fragrance in patients with AD.5,6 Two phenotypes of eczema that have been associated with asthma development are severe AD disease and multiple environmental allergies, supporting the theory of the atopic march.7 There also is evidence that release of danger-associated proteins from an impaired barrier also may trigger asthma.8 An analysis of the 2007 National Survey of Children’s Health, a population-based study of91,642 children aged 0 to 17 years, showed that children with AD had a higher prevalence of comorbid asthma (25.1% vs 12.3%), hay fever (34.4% vs 14.3%), and food allergies (15.1% vs 3.6%) compared to children without AD.9 A recent article provided detailed information on how food and diet interplay with AD.10
Triggers of Disease Flares
Triggers are the leading source of AD flare initiation, and avoidance of triggers is an important mechanism by which patients can control disease activity. Despite the best skin care and trigger avoidance, disease flares occur, sometimes due to ongoing inflammation and other times due to inability to prevent flares such as heat and humidity. A survey of patients with AD in Spain identified the following triggers: cosmetic products, clothing, mites, detergents/soaps, and temperature changes.11 In childhood, wool also is a known trigger of AD.12 Viral infections including respiratory syncytial virus may trigger the first onset of AD.13 Patients with AD may become allergic to fragrance and metals causing disease exacerbation on exposure.14,15 Food allergens contribute to approximately 40% of cases of AD in infancy but are not the cause of AD. The best evidence for improvement of AD with food allergen avoidance exists for egg white allergy.16 Food avoidance programs should be developed in conjunction with an allergist, as it is no longer advised in many cases to completely withdraw foods; therefore, an allergist has to assess the level of allergic severity and the risk-benefit ratio of food avoidance or introduction.17 Emotional stressors, heat, and humidity, as well as indoor heating in the winter months, can cause AD flares.18
A study by Silverberg et al19 provided evidence of climate influences on the US prevalence of childhood eczema using a merged analysis of the 2007 National Survey of Children’s Health and the 2006-2007 National Climate Data Center and Weather Service. Results showed that eczema prevalence was significantly lower when associated with higher annual relative humidity (P=.01), UV index (P<.0001), and highest-quartile air temperature (P=.002).19 The Pediatric Eczema Elective Registry also showed that warm, humid, and high-sun-exposure climates are associated with poorly controlled eczema in affected patients.20 The association of eczema with latitude as well as its negative association with mean annual outdoor temperature has been described by Weiland et al21 in the ISAAC (International Study of Asthma and Allergies in Childhood) study. Long airplane flights in low humidity can trigger eczema in adults. Climate has been postulated to affect eczema through alterations in filaggrin and skin barrier function.22 Indoor temperature and humidity regulation may be used adjunctively for daily flare prevention.
Genetics and AD
Of 762 infants in a birth cohort with a parent with atopy in Cincinnati, Ohio, 39% developed eczema by the age of 3 years. Single nucleotide polymorphisms of IL-4Rα 175 V and CD14-159 C/T were linked to greater eczema risk at 2 to 3 years of age.23 Monozygotic twins have a concordance rate of 0.72 to 0.86 versus 0.21 to 0.23 in dizygotic twins, demonstrating a strong genetic component in the development of AD.24 Linkage to AD has been positively made to the epidermal differentiation complex on human chromosome 1q21, which contains the genes for filaggrin and other proteins such as loricrin. Other genes linked to AD include the serine protease inhibitor SPINK5 (serine peptidase inhibitor, Kazal type 5) implicated in Netherton syndrome (triad of ichthyosis linearis circumflexa, bamboo hair, and atopic disorders); RANTES (regulated on activation, normal T-expressed, and secreted), which has been associated with severity of AD; IL-4; and IL-13.5,25,26
The Hygiene Hypothesis
Atopic dermatitis is more common in wealthy developed countries, leading some to believe that hygiene and relative reduction in illness via vaccination have contributed to the rise of AD prevalence in developed nations.13,27 There currently is evidence demonstrating that wild-type varicella infection confers long-standing protection against AD and mediates reduced total IgE and peripheral blood lymphocytes.27
Grading of AD
Grading of AD is a subject of controversy, as there currently are no uniform grading scales.28 A recent outcomes group attempted to determine the best scale for disease monitoring. Schmitt et al29 presented the Harmonizing Outcome Measures for Eczema (HOME) roadmap, which was intended to determine a core outcome set for eczema; however, because these outcome measurements have not yet been standardized, only the eczema assessment and severity index (EASI) scoring system meets criteria for standardization. In clinical practice, physicians often assign mild, moderate, or severe labeling based on their general sense of the disease extent using an investigator global assessment score.28
The EASI score is a well-validated composite score of AD severity based on 4 body regions: (1) head and neck, (2) trunk (including genital area), (3) upper limbs, and (4) lower limbs (including buttocks). The total area of involvement in each region is graded on a scale of 0 to 6, and AD severity is graded as a composite of 4 parameters (ranked on a scale of 0–3), including redness (erythema, inflammation), thickness (induration, papulation, swelling [acute eczema]), scratching (excoriation), and lichenification (prurigo nodules [chronic eczema]). The surface area of each region relative to body size is used as a multiplying factor, resulting in the following severity strata: 0=clear; 0.1–1.0=almost clear; 1.1–7.0=mild; 7.1–21.0=moderate; 21.1–50.0=severe; 50.1–72.0=very severe (κ=0.75).30-32 The six area, six sign AD (SASSAD) score32,33 is a similar score without adjustment for body surface area by region.34
An older, now less frequently used eczema score is the SCORAD, which addressed surface area by rule of nines and severity of 6 features—redness, swelling, oozing/crusting, scratch marks, skin thickening (lichenification), dryness (assessed in an area with no inflammation)—by region on a scale of 0 to 3. A subjective symptom parameter for itching and sleeplessness helped highlight that these comorbidities are important in gauging disease activity and impact on a child’s life.35
Natural History of AD
The clinical dogma has been that AD would improve with age, with reduction at grade school entry and perhaps full disappearance in adulthood; however, 3 recent surveys have suggested otherwise. The ISAAC group has found prevalence of AD in wealthy developed countries among children aged 6 to 7 years to be at a consistent increase.36 A US-based survey from the National Health Interview Survey showed a 1-year prevalence of 10.2% of active AD in adults and 9.8% when occupational dermatitis was excluded.37 Halvorsen et al38 demonstrated that eczema prevalence is 9.7% in individuals aged 18 to 19 years.
A prospective trial of eighth graders followed from 1995 to 2010 demonstrated that AD persisted in 50% at school age. Persistent eczema into adulthood was associated with early-onset childhood allergic rhinitis and hand eczema.39 In a cohort of hand eczema patients (N=368), 28% had AD and 39% had an atopic illness.40 An association with allergic contact dermatitis and increased IgE to Malassezia furfur was further associated.41
Conclusion
The role of triggers and allergens in disease activity in AD is an important consideration in children with AD and requires ongoing consideration with age and varied exposures. Understanding the grading of AD is important in evaluating clinical trial data. The natural history of AD has changed, which is important for the practitioner to note when counseling patients and guardians.
Atopic dermatitis (AD) may be triggered by viral infections, food allergens, weather, and other causes, and it may trigger an inflammatory progression known as the atopic march. This article reviews research on triggers of pediatric AD so that dermatologists may discuss trigger avoidance with patients and guardians. Other factors affecting AD development include genetics and hygiene. Grading of AD also is discussed.
The Atopic March
The persistence of AD in untreated skin can trigger an inflammatory progression called the atopic march in which food and environmental allergies as well as asthma may occur progressively due to ongoing inflammatory triggering.1 In a study of asthma and food allergy reporting and management in public schools in Chicago, Illinois, food allergies were seen in 9.3% of asthmatic students (n=18,000), and 40.1% of food allergic students (n=4000) had asthma.2 An observational study by Flohr et al3 in London, England, included 619 exclusively breastfed infants who were recruited at 3 months of age. The investigators determined that food sensitization was unrelated to the presence of filaggrin mutations, type of eczema (flexural vs nonflexural), and transepidermal water loss but was associated with AD severity as determined by SCORAD (SCORing Atopic Dermatitis), a composite score of AD that includes pruritus as a factor in severity. Other AD associations included 3 leading food allergens: eggs, milk, and peanuts. No association with cod, wheat, or sesame allergy was noted. The investigators concluded that AD and AD severity were the leading skin-related risk factors for food allergies and therefore food allergy development in breastfed infants was probably mediated by cutaneous antigen-presenting cells.3
The skin has been documented to react to contact with known food allergens4 and is known to be a route of allergic sensitization to allergens such as fragrance in patients with AD.5,6 Two phenotypes of eczema that have been associated with asthma development are severe AD disease and multiple environmental allergies, supporting the theory of the atopic march.7 There also is evidence that release of danger-associated proteins from an impaired barrier also may trigger asthma.8 An analysis of the 2007 National Survey of Children’s Health, a population-based study of91,642 children aged 0 to 17 years, showed that children with AD had a higher prevalence of comorbid asthma (25.1% vs 12.3%), hay fever (34.4% vs 14.3%), and food allergies (15.1% vs 3.6%) compared to children without AD.9 A recent article provided detailed information on how food and diet interplay with AD.10
Triggers of Disease Flares
Triggers are the leading source of AD flare initiation, and avoidance of triggers is an important mechanism by which patients can control disease activity. Despite the best skin care and trigger avoidance, disease flares occur, sometimes due to ongoing inflammation and other times due to inability to prevent flares such as heat and humidity. A survey of patients with AD in Spain identified the following triggers: cosmetic products, clothing, mites, detergents/soaps, and temperature changes.11 In childhood, wool also is a known trigger of AD.12 Viral infections including respiratory syncytial virus may trigger the first onset of AD.13 Patients with AD may become allergic to fragrance and metals causing disease exacerbation on exposure.14,15 Food allergens contribute to approximately 40% of cases of AD in infancy but are not the cause of AD. The best evidence for improvement of AD with food allergen avoidance exists for egg white allergy.16 Food avoidance programs should be developed in conjunction with an allergist, as it is no longer advised in many cases to completely withdraw foods; therefore, an allergist has to assess the level of allergic severity and the risk-benefit ratio of food avoidance or introduction.17 Emotional stressors, heat, and humidity, as well as indoor heating in the winter months, can cause AD flares.18
A study by Silverberg et al19 provided evidence of climate influences on the US prevalence of childhood eczema using a merged analysis of the 2007 National Survey of Children’s Health and the 2006-2007 National Climate Data Center and Weather Service. Results showed that eczema prevalence was significantly lower when associated with higher annual relative humidity (P=.01), UV index (P<.0001), and highest-quartile air temperature (P=.002).19 The Pediatric Eczema Elective Registry also showed that warm, humid, and high-sun-exposure climates are associated with poorly controlled eczema in affected patients.20 The association of eczema with latitude as well as its negative association with mean annual outdoor temperature has been described by Weiland et al21 in the ISAAC (International Study of Asthma and Allergies in Childhood) study. Long airplane flights in low humidity can trigger eczema in adults. Climate has been postulated to affect eczema through alterations in filaggrin and skin barrier function.22 Indoor temperature and humidity regulation may be used adjunctively for daily flare prevention.
Genetics and AD
Of 762 infants in a birth cohort with a parent with atopy in Cincinnati, Ohio, 39% developed eczema by the age of 3 years. Single nucleotide polymorphisms of IL-4Rα 175 V and CD14-159 C/T were linked to greater eczema risk at 2 to 3 years of age.23 Monozygotic twins have a concordance rate of 0.72 to 0.86 versus 0.21 to 0.23 in dizygotic twins, demonstrating a strong genetic component in the development of AD.24 Linkage to AD has been positively made to the epidermal differentiation complex on human chromosome 1q21, which contains the genes for filaggrin and other proteins such as loricrin. Other genes linked to AD include the serine protease inhibitor SPINK5 (serine peptidase inhibitor, Kazal type 5) implicated in Netherton syndrome (triad of ichthyosis linearis circumflexa, bamboo hair, and atopic disorders); RANTES (regulated on activation, normal T-expressed, and secreted), which has been associated with severity of AD; IL-4; and IL-13.5,25,26
The Hygiene Hypothesis
Atopic dermatitis is more common in wealthy developed countries, leading some to believe that hygiene and relative reduction in illness via vaccination have contributed to the rise of AD prevalence in developed nations.13,27 There currently is evidence demonstrating that wild-type varicella infection confers long-standing protection against AD and mediates reduced total IgE and peripheral blood lymphocytes.27
Grading of AD
Grading of AD is a subject of controversy, as there currently are no uniform grading scales.28 A recent outcomes group attempted to determine the best scale for disease monitoring. Schmitt et al29 presented the Harmonizing Outcome Measures for Eczema (HOME) roadmap, which was intended to determine a core outcome set for eczema; however, because these outcome measurements have not yet been standardized, only the eczema assessment and severity index (EASI) scoring system meets criteria for standardization. In clinical practice, physicians often assign mild, moderate, or severe labeling based on their general sense of the disease extent using an investigator global assessment score.28
The EASI score is a well-validated composite score of AD severity based on 4 body regions: (1) head and neck, (2) trunk (including genital area), (3) upper limbs, and (4) lower limbs (including buttocks). The total area of involvement in each region is graded on a scale of 0 to 6, and AD severity is graded as a composite of 4 parameters (ranked on a scale of 0–3), including redness (erythema, inflammation), thickness (induration, papulation, swelling [acute eczema]), scratching (excoriation), and lichenification (prurigo nodules [chronic eczema]). The surface area of each region relative to body size is used as a multiplying factor, resulting in the following severity strata: 0=clear; 0.1–1.0=almost clear; 1.1–7.0=mild; 7.1–21.0=moderate; 21.1–50.0=severe; 50.1–72.0=very severe (κ=0.75).30-32 The six area, six sign AD (SASSAD) score32,33 is a similar score without adjustment for body surface area by region.34
An older, now less frequently used eczema score is the SCORAD, which addressed surface area by rule of nines and severity of 6 features—redness, swelling, oozing/crusting, scratch marks, skin thickening (lichenification), dryness (assessed in an area with no inflammation)—by region on a scale of 0 to 3. A subjective symptom parameter for itching and sleeplessness helped highlight that these comorbidities are important in gauging disease activity and impact on a child’s life.35
Natural History of AD
The clinical dogma has been that AD would improve with age, with reduction at grade school entry and perhaps full disappearance in adulthood; however, 3 recent surveys have suggested otherwise. The ISAAC group has found prevalence of AD in wealthy developed countries among children aged 6 to 7 years to be at a consistent increase.36 A US-based survey from the National Health Interview Survey showed a 1-year prevalence of 10.2% of active AD in adults and 9.8% when occupational dermatitis was excluded.37 Halvorsen et al38 demonstrated that eczema prevalence is 9.7% in individuals aged 18 to 19 years.
A prospective trial of eighth graders followed from 1995 to 2010 demonstrated that AD persisted in 50% at school age. Persistent eczema into adulthood was associated with early-onset childhood allergic rhinitis and hand eczema.39 In a cohort of hand eczema patients (N=368), 28% had AD and 39% had an atopic illness.40 An association with allergic contact dermatitis and increased IgE to Malassezia furfur was further associated.41
Conclusion
The role of triggers and allergens in disease activity in AD is an important consideration in children with AD and requires ongoing consideration with age and varied exposures. Understanding the grading of AD is important in evaluating clinical trial data. The natural history of AD has changed, which is important for the practitioner to note when counseling patients and guardians.
- Li M. Current evidence of epidermal barrier dysfunction and thymic stromal lymphopoietin in the atopic march. Eur Respir Rev. 2014;23:292-298.
- Gupta RS, Rivkina V, DeSantiago-Cardenas L, et al. Asthma and food allergy management in Chicago public schools. Pediatrics. 2014;134:729-736.
- Flohr C, Perkin M, Logan K, et al. Atopic dermatitis and disease severity are the main risk factors for food sensitization in exclusively breastfed infants. J Invest Dermatol. 2014;134:345-350.
- Silverberg NB. Food, glorious food. Cutis. 2011;87:267-268.
- De Benedetto A, Kubo A, Beck LA. Skin barrier disruption: a requirement for allergen sensitization? J Invest Dermatol. 2012;132:949-963.
- Thyssen JP, McFadden JP, Kimber I. The multiple factors affecting the association between atopic dermatitis and contact sensitization. Allergy. 2014;69:28-36.
- Amat F, Saint-Pierre P, Bourrat E, et al. Early-onset atopic dermatitis in children: which are the phenotypes at risk of asthma? results from the ORCA Cohort. PLoS One. 2015;10:e0131369.
- Demehri S, Morimoto M, Holtzman MJ, et al. Skin-derived TSLP triggers progression from epidermal-barrier defects to asthma. PLoS Biol. 2009;7:e1000067.
- Silverberg JI, Simpson EL. Association between severe eczema in children and multiple comorbid conditions and increased healthcare utilization. Pediatr Allergy Immunol. 2013;24:476-486.
- Silverberg NB, Lee-Wong M, Yosipovitch G. Diet and atopic dermatitis. Cutis. 2016;97:227-232.
- Ortiz de Frutos FJ, Torrelo A, de Lucas R, et al. Patient perspectives on triggers, adherence to medical recommendations, and disease control in atopic dermatitis: the DATOP study. Actas Dermosifiliogr. 2014;105:487-496.
- Ricci G, Patrizi A, Bellini F, et al. Use of textiles in atopic dermatitis: care of atopic dermatitis. Curr Probl Dermatol. 2006;33:127-143.
- Welliver RC, Wong DT, Sun M, et al. The development of respiratory syncytial virus-specific IgE and the release of histamine in nasopharyngeal secretions after infection. N Engl J Med. 1981;305:841-846.
- Aquino M, Fonacier L. The role of contact dermatitis in patients with atopic dermatitis. J Allergy Clin Immunol Pract. 2014;2:382-387.
- Brod BA, Treat JR, Rothe MJ, et al. Allergic contact dermatitis: kids are not just little people. Clin Dermatol. 2015;33:605-612.
- Martorell A, Alonso E, Boné J, et al. Position document: IgE-mediated allergy to egg protein. Allergol Immunopathol (Madr). 2013;41:320-336.
- Sicherer SH. Early introduction of peanut to infants at high allergic risk can reduce peanut allergy at age 5 years [published online September 17, 2015]. Evid Based Med. 2015;20:204.
- Kiken DA, Silverberg NB. Atopic dermatitis in children, part 1: epidemiology, clinical features, and complications. Cutis. 2006;78:241-247.
- Silverberg JI, Hanifin J, Simpson EL. Climatic factors are associated with childhood eczema prevalence in the United States. J Invest Dermatol. 2013;133:1752-1759.
- Sargen MR, Hoffstad O, Margolis DJ. Warm, humid, and high sun exposure climates are associated with poorly controlled eczema: PEER (Pediatric Eczema Elective Registry) cohort, 2004-2012. J Invest Dermatol. 2014;134:51-57.
- Weiland SK, Hüsing A, Strachan DP, et al. Climate and the prevalence of symptoms of asthma, allergic rhinitis, and atopic eczema in children. Occup Environ Med. 2004;61:609-615.
- Langan SM, Irvine AD. Childhood eczema and the importance of the physical environment. J Invest Dermatol. 2013;133:1706-1709.
- Biagini Myers JM, Wang N, LeMasters GK, et al. Genetic and environmental risk factors for childhood eczema development and allergic sensitization in the CCAAPS cohort. J Invest Dermatol. 2010;130:430-437.
- Brown SJ, McLean WH. Eczema genetics: current state of knowledge and future goals. J Invest Dermatol. 2009;129:543-552.
- Hanifin JM. Evolving concepts of pathogenesis in atopic dermatitis and other eczemas. J Invest Dermatol. 2009;129:320-322.
- Paller AS. Latest approaches to treating atopic dermatitis. Chem Immunol Allergy. 2012;96:132-140.
- Silverberg JI, Norowitz KB, Kleiman E, et al. Association between varicella zoster virus infection and atopic dermatitis in early and late childhood: a case-control study. J Allergy Clin Immunol. 2010;126:300-305.
- Futamura M, Leshem YA, Thomas KS, et al. A systematic review of Investigator Global Assessment (IGA) in atopic dermatitis (AD) trials: many options, no standards. J Am Acad Dermatol. 2016;74:288-294.
- Schmitt J, Apfelbacher C, Spuls PI, et al. The Harmonizing Outcome Measures for Eczema (HOME) roadmap: a methodological framework to develop core sets of outcome measurements in dermatology. J Invest Dermatol. 2015;135:24-30.
- Hanifin JM, Thurston M, Omoto M, et al. The eczema area and severity index (EASI): assessment of reliability in atopic dermatitis. EASI Evaluator Group. Exp Dermatol. 2001;10:11-18.
- Leshem YA, Hajar T, Hanifin JM, et al. What the Eczema Area and Severity Index score tells us about the severity of atopic dermatitis: an interpretability study. Br J Dermatol. 2015;172:1353-1357.
- Barbier N, Paul C, Luger T, et al. Validation of the Eczema Area and Severity Index for atopic dermatitis in a cohort of 1550 patients from the pimecrolimus cream 1% randomized controlled clinical trials programme. Br J Dermatol. 2004;150:96-102.
- Berth-Jones J. Six area, six sign atopic dermatitis (SASSAD) severity score: a simple system for monitoring disease activity in atopic dermatitis. Br J Dermatol. 1996;135(suppl 48):25-30.
- Zhao CY, Tran AQ, Lazo-Dizon JP, et al. A pilot comparison study of four clinician-rated atopic dermatitis severity scales. Br J Dermatol. 2015;173:488-497.
- Kunz B, Oranje AP, Labrèze L, et al. Clinical validation and guidelines for the SCORAD index: consensus report of the European Task Force on Atopic Dermatitis. Dermatology. 1997;195:10-19.
- Williams H, Stewart A, von Mutius E, et al. Is eczema really on the increase worldwide? J Allergy Clin Immunol. 2008;121:947-954.
- Silverberg JI, Hanifin JM. Adult eczema prevalence and associations with asthma and other health and demographic factors: a US population-based study. J Allergy Clin Immunol. 2013;132:1132-1138.
- Halvorsen JA, Lien L, Dalgard F, et al. Suicidal ideation, mental health problems, and social function in adolescents with eczema: a population-based study. J Invest Dermatol. 2014;134:1847-1854.
- Mortz CG, Andersen KE, Dellgren C, et al. Atopic dermatitis from adolescence to adulthood in the TOACS cohort: prevalence, persistence, and comorbidities. Allergy. 2015;70:836-845.
- Rystedt I. Atopic background in patients with occupational hand eczema. Contact Dermatitis. 1985;12:247-254.
- Mortz CG, Andersen KE, Dellgren C, et al. Atopic dermatitis from adolescence to adulthood in the TOACS cohort: prevalence, persistence and comorbidities. Allergy. 2015;70:836-845.
- Li M. Current evidence of epidermal barrier dysfunction and thymic stromal lymphopoietin in the atopic march. Eur Respir Rev. 2014;23:292-298.
- Gupta RS, Rivkina V, DeSantiago-Cardenas L, et al. Asthma and food allergy management in Chicago public schools. Pediatrics. 2014;134:729-736.
- Flohr C, Perkin M, Logan K, et al. Atopic dermatitis and disease severity are the main risk factors for food sensitization in exclusively breastfed infants. J Invest Dermatol. 2014;134:345-350.
- Silverberg NB. Food, glorious food. Cutis. 2011;87:267-268.
- De Benedetto A, Kubo A, Beck LA. Skin barrier disruption: a requirement for allergen sensitization? J Invest Dermatol. 2012;132:949-963.
- Thyssen JP, McFadden JP, Kimber I. The multiple factors affecting the association between atopic dermatitis and contact sensitization. Allergy. 2014;69:28-36.
- Amat F, Saint-Pierre P, Bourrat E, et al. Early-onset atopic dermatitis in children: which are the phenotypes at risk of asthma? results from the ORCA Cohort. PLoS One. 2015;10:e0131369.
- Demehri S, Morimoto M, Holtzman MJ, et al. Skin-derived TSLP triggers progression from epidermal-barrier defects to asthma. PLoS Biol. 2009;7:e1000067.
- Silverberg JI, Simpson EL. Association between severe eczema in children and multiple comorbid conditions and increased healthcare utilization. Pediatr Allergy Immunol. 2013;24:476-486.
- Silverberg NB, Lee-Wong M, Yosipovitch G. Diet and atopic dermatitis. Cutis. 2016;97:227-232.
- Ortiz de Frutos FJ, Torrelo A, de Lucas R, et al. Patient perspectives on triggers, adherence to medical recommendations, and disease control in atopic dermatitis: the DATOP study. Actas Dermosifiliogr. 2014;105:487-496.
- Ricci G, Patrizi A, Bellini F, et al. Use of textiles in atopic dermatitis: care of atopic dermatitis. Curr Probl Dermatol. 2006;33:127-143.
- Welliver RC, Wong DT, Sun M, et al. The development of respiratory syncytial virus-specific IgE and the release of histamine in nasopharyngeal secretions after infection. N Engl J Med. 1981;305:841-846.
- Aquino M, Fonacier L. The role of contact dermatitis in patients with atopic dermatitis. J Allergy Clin Immunol Pract. 2014;2:382-387.
- Brod BA, Treat JR, Rothe MJ, et al. Allergic contact dermatitis: kids are not just little people. Clin Dermatol. 2015;33:605-612.
- Martorell A, Alonso E, Boné J, et al. Position document: IgE-mediated allergy to egg protein. Allergol Immunopathol (Madr). 2013;41:320-336.
- Sicherer SH. Early introduction of peanut to infants at high allergic risk can reduce peanut allergy at age 5 years [published online September 17, 2015]. Evid Based Med. 2015;20:204.
- Kiken DA, Silverberg NB. Atopic dermatitis in children, part 1: epidemiology, clinical features, and complications. Cutis. 2006;78:241-247.
- Silverberg JI, Hanifin J, Simpson EL. Climatic factors are associated with childhood eczema prevalence in the United States. J Invest Dermatol. 2013;133:1752-1759.
- Sargen MR, Hoffstad O, Margolis DJ. Warm, humid, and high sun exposure climates are associated with poorly controlled eczema: PEER (Pediatric Eczema Elective Registry) cohort, 2004-2012. J Invest Dermatol. 2014;134:51-57.
- Weiland SK, Hüsing A, Strachan DP, et al. Climate and the prevalence of symptoms of asthma, allergic rhinitis, and atopic eczema in children. Occup Environ Med. 2004;61:609-615.
- Langan SM, Irvine AD. Childhood eczema and the importance of the physical environment. J Invest Dermatol. 2013;133:1706-1709.
- Biagini Myers JM, Wang N, LeMasters GK, et al. Genetic and environmental risk factors for childhood eczema development and allergic sensitization in the CCAAPS cohort. J Invest Dermatol. 2010;130:430-437.
- Brown SJ, McLean WH. Eczema genetics: current state of knowledge and future goals. J Invest Dermatol. 2009;129:543-552.
- Hanifin JM. Evolving concepts of pathogenesis in atopic dermatitis and other eczemas. J Invest Dermatol. 2009;129:320-322.
- Paller AS. Latest approaches to treating atopic dermatitis. Chem Immunol Allergy. 2012;96:132-140.
- Silverberg JI, Norowitz KB, Kleiman E, et al. Association between varicella zoster virus infection and atopic dermatitis in early and late childhood: a case-control study. J Allergy Clin Immunol. 2010;126:300-305.
- Futamura M, Leshem YA, Thomas KS, et al. A systematic review of Investigator Global Assessment (IGA) in atopic dermatitis (AD) trials: many options, no standards. J Am Acad Dermatol. 2016;74:288-294.
- Schmitt J, Apfelbacher C, Spuls PI, et al. The Harmonizing Outcome Measures for Eczema (HOME) roadmap: a methodological framework to develop core sets of outcome measurements in dermatology. J Invest Dermatol. 2015;135:24-30.
- Hanifin JM, Thurston M, Omoto M, et al. The eczema area and severity index (EASI): assessment of reliability in atopic dermatitis. EASI Evaluator Group. Exp Dermatol. 2001;10:11-18.
- Leshem YA, Hajar T, Hanifin JM, et al. What the Eczema Area and Severity Index score tells us about the severity of atopic dermatitis: an interpretability study. Br J Dermatol. 2015;172:1353-1357.
- Barbier N, Paul C, Luger T, et al. Validation of the Eczema Area and Severity Index for atopic dermatitis in a cohort of 1550 patients from the pimecrolimus cream 1% randomized controlled clinical trials programme. Br J Dermatol. 2004;150:96-102.
- Berth-Jones J. Six area, six sign atopic dermatitis (SASSAD) severity score: a simple system for monitoring disease activity in atopic dermatitis. Br J Dermatol. 1996;135(suppl 48):25-30.
- Zhao CY, Tran AQ, Lazo-Dizon JP, et al. A pilot comparison study of four clinician-rated atopic dermatitis severity scales. Br J Dermatol. 2015;173:488-497.
- Kunz B, Oranje AP, Labrèze L, et al. Clinical validation and guidelines for the SCORAD index: consensus report of the European Task Force on Atopic Dermatitis. Dermatology. 1997;195:10-19.
- Williams H, Stewart A, von Mutius E, et al. Is eczema really on the increase worldwide? J Allergy Clin Immunol. 2008;121:947-954.
- Silverberg JI, Hanifin JM. Adult eczema prevalence and associations with asthma and other health and demographic factors: a US population-based study. J Allergy Clin Immunol. 2013;132:1132-1138.
- Halvorsen JA, Lien L, Dalgard F, et al. Suicidal ideation, mental health problems, and social function in adolescents with eczema: a population-based study. J Invest Dermatol. 2014;134:1847-1854.
- Mortz CG, Andersen KE, Dellgren C, et al. Atopic dermatitis from adolescence to adulthood in the TOACS cohort: prevalence, persistence, and comorbidities. Allergy. 2015;70:836-845.
- Rystedt I. Atopic background in patients with occupational hand eczema. Contact Dermatitis. 1985;12:247-254.
- Mortz CG, Andersen KE, Dellgren C, et al. Atopic dermatitis from adolescence to adulthood in the TOACS cohort: prevalence, persistence and comorbidities. Allergy. 2015;70:836-845.
Practice Points
- Atopic dermatitis (AD) can be triggered by viral infections, weather, and food allergens.
- The scoring of AD is largely used experimentally and includes the eczema assessment and severity index; the SCORAD (SCORing Atopic Dermatitis); and the six area, six sign AD (SASSAD) scores.
- There is a strong genetic contribution to the development of AD.
- Children with AD may have persistent disease into adulthood in half of cases.
Sublingual immunotherapy for allergy-related asthma
Immunotherapy using sublingual tablets containing house dust mite allergen extended the interval until patients developed a moderate asthma exacerbation in a manufacturer-sponsored clinical trial reported online April 26 in JAMA.
However, patients’ scores on both the Asthma Control Questionnaire and the Asthma Quality of Life Questionnaire showed no difference between active treatment and placebo. And 25%-27% of the study participants dropped out of the study, usually citing asthma exacerbations, adverse events, or “withdrawal of consent.” Further studies are needed to assess long-term efficacy and safety, said Dr. J. Christian Virchow of the department of pulmonology/intensive care medicine, University of Rostock (Germany), and his associates.
The trial, involving 834 adults with asthma related to house dust mite allergy that was not well controlled by inhaled corticosteroids and short-acting beta-agonists, was performed at 109 sites in 13 European countries during a 2-year period. These participants were randomly assigned to receive add-on daily sublingual tablets containing low-dose dust-mite extract (275 patients), high-dose extract (282 patients), or placebo (277 patients) for 7-12 months. During the final 6 months of the intervention, corticosteroids were reduced by half for 3 months and then withdrawn for 3 months.
The primary efficacy outcome (time to the first asthma exacerbation) was extended by both doses of active drug, compared with placebo, with hazard ratios of 0.69 for the lower dose and 0.66 for the higher dose, the investigators said (JAMA. 2016 Apr 26;315[16]:1715-25).
Adverse events were significantly more frequent with active treatment, affecting 39% of patients receiving the lower dose and 46% of those receiving the higher dose of active immunotherapy, compared with only 17% of patients receiving placebo. However, this study was not adequately powered to compare adverse events across groups, Dr. Virchow and his associates noted.
The most frequently reported adverse events were oral pruritus, mouth edema, and throat irritation, which developed within a median of 1-2 minutes of taking the first dose on day 1 and persisted for a median of 4-23 days. There were 32 serious adverse events, including erosive esophagitis, hepatocellular injury, arthralgia, laryngeal edema, and asthma.
This trial was limited in that treatment duration was much shorter than that for a standard course of immunotherapy, which is often 3 years. This prevents drawing conclusions regarding the sustained effect of the treatment. “Furthermore, because the ultimate aim of allergen immunotherapy is disease modification beyond the duration of treatment, a follow-up after the end of treatment would have been relevant,” the investigators said.
This study was sponsored by the Danish pharmaceutical company ALK. Dr. Virchow reported ties to 31 industry sources; his associates also reported ties to numerous industry sources.
Sublingual immunotherapy appears to be somewhat less effective than subcutaneous immunotherapy, but it offers several advantages. It doesn’t require injections, can be self-administered, doesn’t require dose escalations, and carries a much lower risk of anaphylaxis. However, in this study there were no significant differences in patients’ responses to questionnaires regarding either asthma control or quality of life.
The main disadvantage is that sublingual immunotherapy requires adherence to daily dosing, and research has consistently shown low rates of long-term adherence. In one study, 55%-82% of patients failed to complete the recommended course of sublingual immunotherapy. In another, only 44% of patients renewed their prescriptions after 1 year of treatment, only 28% did so after 2 years, and only 13% did so after 3 years.
Dr. Robert A. Wood is in the division of allergy and immunology, department of pediatrics, at Johns Hopkins University, Baltimore. He reported ties to DBV Technologies, the Immune Tolerance Network, Stallergenes, Sanofi, and UpToDate. Dr. Wood made these remarks in an editorial accompanying Dr. Virchow’s report (JAMA. 2016 Apr 26;315:1711-2).
Sublingual immunotherapy appears to be somewhat less effective than subcutaneous immunotherapy, but it offers several advantages. It doesn’t require injections, can be self-administered, doesn’t require dose escalations, and carries a much lower risk of anaphylaxis. However, in this study there were no significant differences in patients’ responses to questionnaires regarding either asthma control or quality of life.
The main disadvantage is that sublingual immunotherapy requires adherence to daily dosing, and research has consistently shown low rates of long-term adherence. In one study, 55%-82% of patients failed to complete the recommended course of sublingual immunotherapy. In another, only 44% of patients renewed their prescriptions after 1 year of treatment, only 28% did so after 2 years, and only 13% did so after 3 years.
Dr. Robert A. Wood is in the division of allergy and immunology, department of pediatrics, at Johns Hopkins University, Baltimore. He reported ties to DBV Technologies, the Immune Tolerance Network, Stallergenes, Sanofi, and UpToDate. Dr. Wood made these remarks in an editorial accompanying Dr. Virchow’s report (JAMA. 2016 Apr 26;315:1711-2).
Sublingual immunotherapy appears to be somewhat less effective than subcutaneous immunotherapy, but it offers several advantages. It doesn’t require injections, can be self-administered, doesn’t require dose escalations, and carries a much lower risk of anaphylaxis. However, in this study there were no significant differences in patients’ responses to questionnaires regarding either asthma control or quality of life.
The main disadvantage is that sublingual immunotherapy requires adherence to daily dosing, and research has consistently shown low rates of long-term adherence. In one study, 55%-82% of patients failed to complete the recommended course of sublingual immunotherapy. In another, only 44% of patients renewed their prescriptions after 1 year of treatment, only 28% did so after 2 years, and only 13% did so after 3 years.
Dr. Robert A. Wood is in the division of allergy and immunology, department of pediatrics, at Johns Hopkins University, Baltimore. He reported ties to DBV Technologies, the Immune Tolerance Network, Stallergenes, Sanofi, and UpToDate. Dr. Wood made these remarks in an editorial accompanying Dr. Virchow’s report (JAMA. 2016 Apr 26;315:1711-2).
Immunotherapy using sublingual tablets containing house dust mite allergen extended the interval until patients developed a moderate asthma exacerbation in a manufacturer-sponsored clinical trial reported online April 26 in JAMA.
However, patients’ scores on both the Asthma Control Questionnaire and the Asthma Quality of Life Questionnaire showed no difference between active treatment and placebo. And 25%-27% of the study participants dropped out of the study, usually citing asthma exacerbations, adverse events, or “withdrawal of consent.” Further studies are needed to assess long-term efficacy and safety, said Dr. J. Christian Virchow of the department of pulmonology/intensive care medicine, University of Rostock (Germany), and his associates.
The trial, involving 834 adults with asthma related to house dust mite allergy that was not well controlled by inhaled corticosteroids and short-acting beta-agonists, was performed at 109 sites in 13 European countries during a 2-year period. These participants were randomly assigned to receive add-on daily sublingual tablets containing low-dose dust-mite extract (275 patients), high-dose extract (282 patients), or placebo (277 patients) for 7-12 months. During the final 6 months of the intervention, corticosteroids were reduced by half for 3 months and then withdrawn for 3 months.
The primary efficacy outcome (time to the first asthma exacerbation) was extended by both doses of active drug, compared with placebo, with hazard ratios of 0.69 for the lower dose and 0.66 for the higher dose, the investigators said (JAMA. 2016 Apr 26;315[16]:1715-25).
Adverse events were significantly more frequent with active treatment, affecting 39% of patients receiving the lower dose and 46% of those receiving the higher dose of active immunotherapy, compared with only 17% of patients receiving placebo. However, this study was not adequately powered to compare adverse events across groups, Dr. Virchow and his associates noted.
The most frequently reported adverse events were oral pruritus, mouth edema, and throat irritation, which developed within a median of 1-2 minutes of taking the first dose on day 1 and persisted for a median of 4-23 days. There were 32 serious adverse events, including erosive esophagitis, hepatocellular injury, arthralgia, laryngeal edema, and asthma.
This trial was limited in that treatment duration was much shorter than that for a standard course of immunotherapy, which is often 3 years. This prevents drawing conclusions regarding the sustained effect of the treatment. “Furthermore, because the ultimate aim of allergen immunotherapy is disease modification beyond the duration of treatment, a follow-up after the end of treatment would have been relevant,” the investigators said.
This study was sponsored by the Danish pharmaceutical company ALK. Dr. Virchow reported ties to 31 industry sources; his associates also reported ties to numerous industry sources.
Immunotherapy using sublingual tablets containing house dust mite allergen extended the interval until patients developed a moderate asthma exacerbation in a manufacturer-sponsored clinical trial reported online April 26 in JAMA.
However, patients’ scores on both the Asthma Control Questionnaire and the Asthma Quality of Life Questionnaire showed no difference between active treatment and placebo. And 25%-27% of the study participants dropped out of the study, usually citing asthma exacerbations, adverse events, or “withdrawal of consent.” Further studies are needed to assess long-term efficacy and safety, said Dr. J. Christian Virchow of the department of pulmonology/intensive care medicine, University of Rostock (Germany), and his associates.
The trial, involving 834 adults with asthma related to house dust mite allergy that was not well controlled by inhaled corticosteroids and short-acting beta-agonists, was performed at 109 sites in 13 European countries during a 2-year period. These participants were randomly assigned to receive add-on daily sublingual tablets containing low-dose dust-mite extract (275 patients), high-dose extract (282 patients), or placebo (277 patients) for 7-12 months. During the final 6 months of the intervention, corticosteroids were reduced by half for 3 months and then withdrawn for 3 months.
The primary efficacy outcome (time to the first asthma exacerbation) was extended by both doses of active drug, compared with placebo, with hazard ratios of 0.69 for the lower dose and 0.66 for the higher dose, the investigators said (JAMA. 2016 Apr 26;315[16]:1715-25).
Adverse events were significantly more frequent with active treatment, affecting 39% of patients receiving the lower dose and 46% of those receiving the higher dose of active immunotherapy, compared with only 17% of patients receiving placebo. However, this study was not adequately powered to compare adverse events across groups, Dr. Virchow and his associates noted.
The most frequently reported adverse events were oral pruritus, mouth edema, and throat irritation, which developed within a median of 1-2 minutes of taking the first dose on day 1 and persisted for a median of 4-23 days. There were 32 serious adverse events, including erosive esophagitis, hepatocellular injury, arthralgia, laryngeal edema, and asthma.
This trial was limited in that treatment duration was much shorter than that for a standard course of immunotherapy, which is often 3 years. This prevents drawing conclusions regarding the sustained effect of the treatment. “Furthermore, because the ultimate aim of allergen immunotherapy is disease modification beyond the duration of treatment, a follow-up after the end of treatment would have been relevant,” the investigators said.
This study was sponsored by the Danish pharmaceutical company ALK. Dr. Virchow reported ties to 31 industry sources; his associates also reported ties to numerous industry sources.
FROM JAMA
Key clinical point: Sublingual tablets containing house dust mite allergen immunotherapy extended the interval until a moderate or severe asthma exacerbation.
Major finding: The primary efficacy outcome (time to the first asthma exacerbation) was extended by both doses of active drug, compared with placebo, with hazard ratios of 0.69 for the lower dose and 0.66 for the higher dose.
Data source: An industry-sponsored international randomized placebo-controlled trial involving 834 patients.
Disclosures: This study was sponsored by the Danish pharmaceutical company ALK. Dr. Virchow reported ties to 31 industry sources; his associates also reported ties to numerous industry sources.
Atopic dermatitis early in childhood tied to increased risk of autism, ADHD
Children who are diagnosed with atopic dermatitis before the age of 2 are more likely to be diagnosed with autism spectrum disorder or attention-deficit/hyperactivity disorder, according to Tzu-Chu Liao and associates.
Of the 387,262 children diagnosed with atopic dermatitis (AD) before the age of 2 included in the study, 0.5% were diagnosed with autism spectrum disorder (ASD), and 3.7% were diagnosed with attention-deficit/hyperactivity disorder (ADHD). In the control group, 0.4% were diagnosed with ASD, and 2.9% were diagnosed with ADHD. The hazard ratios for children exposed to atopic disorders before the age of 2 were 1.1 for ASD and 1.16 for ADHD.
Among children diagnosed early with AD, being male was the most significant risk factor for developing ASD (HR, 4.92) or ADHD (HR, 3.28). An urban/suburban residence was also a significant risk factor, as was persistent AD and emerging atopic respiratory disease in childhood.
“These findings suggest a possible etiologic communality between the diagnosis of allergic disorders along with comorbid ASD or ADHD. The atopic diathesis approach might influence the attention of child psychiatrists and pediatricians toward the diagnosis of ASD and ADHD. Further attention should be given to the management of allergic manifestations when treating symptoms of ASD and ADHD,” the investigators concluded.
Find the study in the Journal of Pediatrics (doi: 10.1016/j.jpeds.2015.12.063).
Children who are diagnosed with atopic dermatitis before the age of 2 are more likely to be diagnosed with autism spectrum disorder or attention-deficit/hyperactivity disorder, according to Tzu-Chu Liao and associates.
Of the 387,262 children diagnosed with atopic dermatitis (AD) before the age of 2 included in the study, 0.5% were diagnosed with autism spectrum disorder (ASD), and 3.7% were diagnosed with attention-deficit/hyperactivity disorder (ADHD). In the control group, 0.4% were diagnosed with ASD, and 2.9% were diagnosed with ADHD. The hazard ratios for children exposed to atopic disorders before the age of 2 were 1.1 for ASD and 1.16 for ADHD.
Among children diagnosed early with AD, being male was the most significant risk factor for developing ASD (HR, 4.92) or ADHD (HR, 3.28). An urban/suburban residence was also a significant risk factor, as was persistent AD and emerging atopic respiratory disease in childhood.
“These findings suggest a possible etiologic communality between the diagnosis of allergic disorders along with comorbid ASD or ADHD. The atopic diathesis approach might influence the attention of child psychiatrists and pediatricians toward the diagnosis of ASD and ADHD. Further attention should be given to the management of allergic manifestations when treating symptoms of ASD and ADHD,” the investigators concluded.
Find the study in the Journal of Pediatrics (doi: 10.1016/j.jpeds.2015.12.063).
Children who are diagnosed with atopic dermatitis before the age of 2 are more likely to be diagnosed with autism spectrum disorder or attention-deficit/hyperactivity disorder, according to Tzu-Chu Liao and associates.
Of the 387,262 children diagnosed with atopic dermatitis (AD) before the age of 2 included in the study, 0.5% were diagnosed with autism spectrum disorder (ASD), and 3.7% were diagnosed with attention-deficit/hyperactivity disorder (ADHD). In the control group, 0.4% were diagnosed with ASD, and 2.9% were diagnosed with ADHD. The hazard ratios for children exposed to atopic disorders before the age of 2 were 1.1 for ASD and 1.16 for ADHD.
Among children diagnosed early with AD, being male was the most significant risk factor for developing ASD (HR, 4.92) or ADHD (HR, 3.28). An urban/suburban residence was also a significant risk factor, as was persistent AD and emerging atopic respiratory disease in childhood.
“These findings suggest a possible etiologic communality between the diagnosis of allergic disorders along with comorbid ASD or ADHD. The atopic diathesis approach might influence the attention of child psychiatrists and pediatricians toward the diagnosis of ASD and ADHD. Further attention should be given to the management of allergic manifestations when treating symptoms of ASD and ADHD,” the investigators concluded.
Find the study in the Journal of Pediatrics (doi: 10.1016/j.jpeds.2015.12.063).
FROM THE JOURNAL OF PEDIATRICS
New treatments bring hope for severe atopic dermatitis
LOS ANGELES – For patients with severe atopic dermatitis and their families and treating physicians, there is big news: finally, there is light at the end of the tunnel, Dr. Lisa A. Beck declared in a plenary lecture at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Better drugs are on the way. The new agents in the developmental pipeline target specific immunologic pathways that appear to be central to atopic dermatitis. Moreover, exciting recent evidence indicates it’s possible to noninvasively identify children at high risk for atopic dermatitis and intervene preventively to reduce the likelihood of actually developing the disease, according to Dr. Beck, professor of dermatology and medicine at the University of Rochester (N.Y.).
“Many pharmaceutical companies have now turned their attention to atopic dermatitis and aren’t just focusing on asthma anymore. The biggest pipeline appears to involve drugs that might target the Th2 [T helper 2 cells] pathway, either by trying to eliminate alarmins such as TSLP [thymic stromal lymphopoietin], or reverse the effects of the Th2 cytokines interleukin-4 and -13, either alone or together, or prevent the recruitment of activated T cells,” she said.
Dr. Beck presented an update on three such promising investigational approaches on the horizon: the IL-4 and IL-13 inhibitor dupilumab; oral and topical Janus associated kinase (JAK) inhibitors; and anti-IgE therapies.
Dupilumab: This fully human monoclonal antibody that blocks IL-4 and IL-13 is also being developed as a treatment for eosinophilic asthma. Dr. Beck was first author of a report on a series of four phase II randomized trials of dupilumab for moderate to severe atopic dermatitis in adults. The publication caused a stir, with dupilumab-treated patients showing marked and rapid improvement to a degree previously unseen in the treatment of this disease (N Engl J Med. 2014;371[2]:130-9).
In the 12-week study, for example, 85% of dupilumab-treated patients achieved at least a 50% improvement in the Eczema Area and Severity Index (EASI) score, compared with 35% of placebo-treated controls, with a significant between-group difference seen in the first week. Maximum improvement – a 75%-80% reduction in EASI scores – was noted at 6-8 weeks. Forty percent of dupilumab-treated patients achieved clear or near-clear skin by investigator’s global assessment, compared with just 7% of controls.
Itching decreased markedly beginning in the first week, too. The investigational agent’s side effect profile was similar to placebo. Phase III clinical trials in atopic dermatitis are ongoing.
A study by other investigators found that dupilumab resulted in rapid improvement in the molecular signature of atopic dermatitis in skin biopsy specimens (J Allergy Clin Immunol. 2014 Dec;134[6]:1293-300). The observed changes in gene expression suggest that dupilumab might have a beneficial effect on the dysfunctional skin barrier that is a hallmark of atopic dermatitis. Further studies are now being planned to take a closer look at that possibility.
JAK inhibitors: “We’re all really excited about this approach because dogs, too, get allergic dermatitis, and in 2013 a JAK 1 and 3 inhibitor [oclacitinib, Apoquel] was approved as a veterinary medicine therapy. It has resulted in dramatic improvement in itch within 1 week of administration, as well as significant improvement in the dermatitis,” Dr. Beck said.
Three JAK inhibitors are now in phase II clinical trials for atopic dermatitis in humans: the JAK 1 and 3 inhibitor tofacitinib (Xeljanz), both as a topical ointment and the familiar oral formulation; baricitinib, an oral JAK 1 and 2 inhibitor; and an agent known for now as PF-04965842, which is an oral inhibitor specifically of JAK 1.
“JAK inhibitors have been quite effective in treating a number of other inflammatory conditions, as well as cancers. I think they will have a role in the treatment of atopic dermatitis. The biggest concerns will be the off-target effects,” she predicted.
Anti-IgE agents: Omalizumab (Xolair), a humanized monoclonal antibody that binds to IgE, has gotten mixed reviews as an investigational treatment for atopic dermatitis. The best study to date, a randomized, single-center, placebo-controlled, double-blind, 16-week clinical trial, found that omalizumab depleted IgE but didn’t improve the clinical course of atopic dermatitis (J Dtsch Dermatol Ges. 2010 Dec;8[12]:990-8). Nonetheless, a phase II trial of omalizumab is ongoing. Plus, ligelizumab, an anti-IgE monoclonal antibody with a higher affinity for IgE than omalizumab, is also in a phase II trial for adult atopic dermatitis.
“Anti-IgE therapy, I think, is still not dead in atopic dermatitis. I look forward to seeing whether omalizumab will work in unique subsets of patients, or whether a more potent anti-IgE molecule will be more beneficial,” Dr. Beck commented.
As exciting as the prospects are for these investigational agents, there also have been several recent important advances in the prevention of atopic dermatitis, she continued. Investigators led by Dr. Alan D. Irvine of Trinity College, Dublin, noninvasively measured transepidermal water loss in early infancy in more than 1,900 Irish 2-day-old infants and found that those in the 75th percentile for this early marker of skin barrier dysfunction were at 3.1-fold increased risk for diagnosis of atopic dermatitis by age 2 years (J Allergy Clin Immunol. 2016 Apr;137[4]:1111-6). This new-found ability to identify at-risk infants will be extremely helpful in designing atopic dermatitis prevention studies, according to Dr. Beck.
The other advance in prevention was provided via a randomized trial by Dr. Eric L. Simpson of Oregon Health and Science University, Portland, and his coinvestigators. They randomized a group of infants at high risk for atopic dermatitis to daily application of any of five OTC skin moisturizers or a no-moisturizer control group from 2 weeks through 6 months of age. The study hypothesis was that the moisturizers would help reverse the skin barrier abnormalities that play a key role in atopic dermatitis. The hypothesis was borne out by the finding that there was at least a 50% reduction in physician-diagnosed atopic dermatitis by age 6 months in the daily moisturizer group (J Allergy Clin Immunol. 2014 Oct;134[4]:818-23).
Dr. Beck concluded by describing a likely near-term atopic dermatitis prevention and management scenario: High-risk infants will be identified on the basis of noninvasive assessment of epithelial features, such as transepidermal water loss or the presence of high levels of thymic stromal lymphopoietin on the skin surface. Encouragement of daily moisturizing for these high-risk infants will prevent some of them from going on to develop eczema.
For those who do get eczema, dilute bleach baths will help in restoring normal skin barrier function, as was confirmed in an in-press study by Dr. Beck and her coinvestigators, who found that 46% of a group of adults with atopic dermatitis experienced at least a 50% improvement in EASI scores, a big improvement in itch, and reduced transepidermal water loss after 12 weeks of the bleach baths. As other investigators have reported, the bleach baths were very well tolerated and safe.
Dr. Beck reported serving as a consultant to eight pharmaceutical companies with an interest in developing new treatments for atopic dermatitis.
LOS ANGELES – For patients with severe atopic dermatitis and their families and treating physicians, there is big news: finally, there is light at the end of the tunnel, Dr. Lisa A. Beck declared in a plenary lecture at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Better drugs are on the way. The new agents in the developmental pipeline target specific immunologic pathways that appear to be central to atopic dermatitis. Moreover, exciting recent evidence indicates it’s possible to noninvasively identify children at high risk for atopic dermatitis and intervene preventively to reduce the likelihood of actually developing the disease, according to Dr. Beck, professor of dermatology and medicine at the University of Rochester (N.Y.).
“Many pharmaceutical companies have now turned their attention to atopic dermatitis and aren’t just focusing on asthma anymore. The biggest pipeline appears to involve drugs that might target the Th2 [T helper 2 cells] pathway, either by trying to eliminate alarmins such as TSLP [thymic stromal lymphopoietin], or reverse the effects of the Th2 cytokines interleukin-4 and -13, either alone or together, or prevent the recruitment of activated T cells,” she said.
Dr. Beck presented an update on three such promising investigational approaches on the horizon: the IL-4 and IL-13 inhibitor dupilumab; oral and topical Janus associated kinase (JAK) inhibitors; and anti-IgE therapies.
Dupilumab: This fully human monoclonal antibody that blocks IL-4 and IL-13 is also being developed as a treatment for eosinophilic asthma. Dr. Beck was first author of a report on a series of four phase II randomized trials of dupilumab for moderate to severe atopic dermatitis in adults. The publication caused a stir, with dupilumab-treated patients showing marked and rapid improvement to a degree previously unseen in the treatment of this disease (N Engl J Med. 2014;371[2]:130-9).
In the 12-week study, for example, 85% of dupilumab-treated patients achieved at least a 50% improvement in the Eczema Area and Severity Index (EASI) score, compared with 35% of placebo-treated controls, with a significant between-group difference seen in the first week. Maximum improvement – a 75%-80% reduction in EASI scores – was noted at 6-8 weeks. Forty percent of dupilumab-treated patients achieved clear or near-clear skin by investigator’s global assessment, compared with just 7% of controls.
Itching decreased markedly beginning in the first week, too. The investigational agent’s side effect profile was similar to placebo. Phase III clinical trials in atopic dermatitis are ongoing.
A study by other investigators found that dupilumab resulted in rapid improvement in the molecular signature of atopic dermatitis in skin biopsy specimens (J Allergy Clin Immunol. 2014 Dec;134[6]:1293-300). The observed changes in gene expression suggest that dupilumab might have a beneficial effect on the dysfunctional skin barrier that is a hallmark of atopic dermatitis. Further studies are now being planned to take a closer look at that possibility.
JAK inhibitors: “We’re all really excited about this approach because dogs, too, get allergic dermatitis, and in 2013 a JAK 1 and 3 inhibitor [oclacitinib, Apoquel] was approved as a veterinary medicine therapy. It has resulted in dramatic improvement in itch within 1 week of administration, as well as significant improvement in the dermatitis,” Dr. Beck said.
Three JAK inhibitors are now in phase II clinical trials for atopic dermatitis in humans: the JAK 1 and 3 inhibitor tofacitinib (Xeljanz), both as a topical ointment and the familiar oral formulation; baricitinib, an oral JAK 1 and 2 inhibitor; and an agent known for now as PF-04965842, which is an oral inhibitor specifically of JAK 1.
“JAK inhibitors have been quite effective in treating a number of other inflammatory conditions, as well as cancers. I think they will have a role in the treatment of atopic dermatitis. The biggest concerns will be the off-target effects,” she predicted.
Anti-IgE agents: Omalizumab (Xolair), a humanized monoclonal antibody that binds to IgE, has gotten mixed reviews as an investigational treatment for atopic dermatitis. The best study to date, a randomized, single-center, placebo-controlled, double-blind, 16-week clinical trial, found that omalizumab depleted IgE but didn’t improve the clinical course of atopic dermatitis (J Dtsch Dermatol Ges. 2010 Dec;8[12]:990-8). Nonetheless, a phase II trial of omalizumab is ongoing. Plus, ligelizumab, an anti-IgE monoclonal antibody with a higher affinity for IgE than omalizumab, is also in a phase II trial for adult atopic dermatitis.
“Anti-IgE therapy, I think, is still not dead in atopic dermatitis. I look forward to seeing whether omalizumab will work in unique subsets of patients, or whether a more potent anti-IgE molecule will be more beneficial,” Dr. Beck commented.
As exciting as the prospects are for these investigational agents, there also have been several recent important advances in the prevention of atopic dermatitis, she continued. Investigators led by Dr. Alan D. Irvine of Trinity College, Dublin, noninvasively measured transepidermal water loss in early infancy in more than 1,900 Irish 2-day-old infants and found that those in the 75th percentile for this early marker of skin barrier dysfunction were at 3.1-fold increased risk for diagnosis of atopic dermatitis by age 2 years (J Allergy Clin Immunol. 2016 Apr;137[4]:1111-6). This new-found ability to identify at-risk infants will be extremely helpful in designing atopic dermatitis prevention studies, according to Dr. Beck.
The other advance in prevention was provided via a randomized trial by Dr. Eric L. Simpson of Oregon Health and Science University, Portland, and his coinvestigators. They randomized a group of infants at high risk for atopic dermatitis to daily application of any of five OTC skin moisturizers or a no-moisturizer control group from 2 weeks through 6 months of age. The study hypothesis was that the moisturizers would help reverse the skin barrier abnormalities that play a key role in atopic dermatitis. The hypothesis was borne out by the finding that there was at least a 50% reduction in physician-diagnosed atopic dermatitis by age 6 months in the daily moisturizer group (J Allergy Clin Immunol. 2014 Oct;134[4]:818-23).
Dr. Beck concluded by describing a likely near-term atopic dermatitis prevention and management scenario: High-risk infants will be identified on the basis of noninvasive assessment of epithelial features, such as transepidermal water loss or the presence of high levels of thymic stromal lymphopoietin on the skin surface. Encouragement of daily moisturizing for these high-risk infants will prevent some of them from going on to develop eczema.
For those who do get eczema, dilute bleach baths will help in restoring normal skin barrier function, as was confirmed in an in-press study by Dr. Beck and her coinvestigators, who found that 46% of a group of adults with atopic dermatitis experienced at least a 50% improvement in EASI scores, a big improvement in itch, and reduced transepidermal water loss after 12 weeks of the bleach baths. As other investigators have reported, the bleach baths were very well tolerated and safe.
Dr. Beck reported serving as a consultant to eight pharmaceutical companies with an interest in developing new treatments for atopic dermatitis.
LOS ANGELES – For patients with severe atopic dermatitis and their families and treating physicians, there is big news: finally, there is light at the end of the tunnel, Dr. Lisa A. Beck declared in a plenary lecture at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Better drugs are on the way. The new agents in the developmental pipeline target specific immunologic pathways that appear to be central to atopic dermatitis. Moreover, exciting recent evidence indicates it’s possible to noninvasively identify children at high risk for atopic dermatitis and intervene preventively to reduce the likelihood of actually developing the disease, according to Dr. Beck, professor of dermatology and medicine at the University of Rochester (N.Y.).
“Many pharmaceutical companies have now turned their attention to atopic dermatitis and aren’t just focusing on asthma anymore. The biggest pipeline appears to involve drugs that might target the Th2 [T helper 2 cells] pathway, either by trying to eliminate alarmins such as TSLP [thymic stromal lymphopoietin], or reverse the effects of the Th2 cytokines interleukin-4 and -13, either alone or together, or prevent the recruitment of activated T cells,” she said.
Dr. Beck presented an update on three such promising investigational approaches on the horizon: the IL-4 and IL-13 inhibitor dupilumab; oral and topical Janus associated kinase (JAK) inhibitors; and anti-IgE therapies.
Dupilumab: This fully human monoclonal antibody that blocks IL-4 and IL-13 is also being developed as a treatment for eosinophilic asthma. Dr. Beck was first author of a report on a series of four phase II randomized trials of dupilumab for moderate to severe atopic dermatitis in adults. The publication caused a stir, with dupilumab-treated patients showing marked and rapid improvement to a degree previously unseen in the treatment of this disease (N Engl J Med. 2014;371[2]:130-9).
In the 12-week study, for example, 85% of dupilumab-treated patients achieved at least a 50% improvement in the Eczema Area and Severity Index (EASI) score, compared with 35% of placebo-treated controls, with a significant between-group difference seen in the first week. Maximum improvement – a 75%-80% reduction in EASI scores – was noted at 6-8 weeks. Forty percent of dupilumab-treated patients achieved clear or near-clear skin by investigator’s global assessment, compared with just 7% of controls.
Itching decreased markedly beginning in the first week, too. The investigational agent’s side effect profile was similar to placebo. Phase III clinical trials in atopic dermatitis are ongoing.
A study by other investigators found that dupilumab resulted in rapid improvement in the molecular signature of atopic dermatitis in skin biopsy specimens (J Allergy Clin Immunol. 2014 Dec;134[6]:1293-300). The observed changes in gene expression suggest that dupilumab might have a beneficial effect on the dysfunctional skin barrier that is a hallmark of atopic dermatitis. Further studies are now being planned to take a closer look at that possibility.
JAK inhibitors: “We’re all really excited about this approach because dogs, too, get allergic dermatitis, and in 2013 a JAK 1 and 3 inhibitor [oclacitinib, Apoquel] was approved as a veterinary medicine therapy. It has resulted in dramatic improvement in itch within 1 week of administration, as well as significant improvement in the dermatitis,” Dr. Beck said.
Three JAK inhibitors are now in phase II clinical trials for atopic dermatitis in humans: the JAK 1 and 3 inhibitor tofacitinib (Xeljanz), both as a topical ointment and the familiar oral formulation; baricitinib, an oral JAK 1 and 2 inhibitor; and an agent known for now as PF-04965842, which is an oral inhibitor specifically of JAK 1.
“JAK inhibitors have been quite effective in treating a number of other inflammatory conditions, as well as cancers. I think they will have a role in the treatment of atopic dermatitis. The biggest concerns will be the off-target effects,” she predicted.
Anti-IgE agents: Omalizumab (Xolair), a humanized monoclonal antibody that binds to IgE, has gotten mixed reviews as an investigational treatment for atopic dermatitis. The best study to date, a randomized, single-center, placebo-controlled, double-blind, 16-week clinical trial, found that omalizumab depleted IgE but didn’t improve the clinical course of atopic dermatitis (J Dtsch Dermatol Ges. 2010 Dec;8[12]:990-8). Nonetheless, a phase II trial of omalizumab is ongoing. Plus, ligelizumab, an anti-IgE monoclonal antibody with a higher affinity for IgE than omalizumab, is also in a phase II trial for adult atopic dermatitis.
“Anti-IgE therapy, I think, is still not dead in atopic dermatitis. I look forward to seeing whether omalizumab will work in unique subsets of patients, or whether a more potent anti-IgE molecule will be more beneficial,” Dr. Beck commented.
As exciting as the prospects are for these investigational agents, there also have been several recent important advances in the prevention of atopic dermatitis, she continued. Investigators led by Dr. Alan D. Irvine of Trinity College, Dublin, noninvasively measured transepidermal water loss in early infancy in more than 1,900 Irish 2-day-old infants and found that those in the 75th percentile for this early marker of skin barrier dysfunction were at 3.1-fold increased risk for diagnosis of atopic dermatitis by age 2 years (J Allergy Clin Immunol. 2016 Apr;137[4]:1111-6). This new-found ability to identify at-risk infants will be extremely helpful in designing atopic dermatitis prevention studies, according to Dr. Beck.
The other advance in prevention was provided via a randomized trial by Dr. Eric L. Simpson of Oregon Health and Science University, Portland, and his coinvestigators. They randomized a group of infants at high risk for atopic dermatitis to daily application of any of five OTC skin moisturizers or a no-moisturizer control group from 2 weeks through 6 months of age. The study hypothesis was that the moisturizers would help reverse the skin barrier abnormalities that play a key role in atopic dermatitis. The hypothesis was borne out by the finding that there was at least a 50% reduction in physician-diagnosed atopic dermatitis by age 6 months in the daily moisturizer group (J Allergy Clin Immunol. 2014 Oct;134[4]:818-23).
Dr. Beck concluded by describing a likely near-term atopic dermatitis prevention and management scenario: High-risk infants will be identified on the basis of noninvasive assessment of epithelial features, such as transepidermal water loss or the presence of high levels of thymic stromal lymphopoietin on the skin surface. Encouragement of daily moisturizing for these high-risk infants will prevent some of them from going on to develop eczema.
For those who do get eczema, dilute bleach baths will help in restoring normal skin barrier function, as was confirmed in an in-press study by Dr. Beck and her coinvestigators, who found that 46% of a group of adults with atopic dermatitis experienced at least a 50% improvement in EASI scores, a big improvement in itch, and reduced transepidermal water loss after 12 weeks of the bleach baths. As other investigators have reported, the bleach baths were very well tolerated and safe.
Dr. Beck reported serving as a consultant to eight pharmaceutical companies with an interest in developing new treatments for atopic dermatitis.
EXPERT ANALYSIS FROM THE 2016 AAAAI ANNUAL MEETING
Cochrane Review nixes specific allergen immunotherapy for atopic dermatitis
LOS ANGELES – A new Cochrane systematic review and meta-analysis has concluded there is no consistent evidence that specific allergen immunotherapy is beneficial in patients with atopic dermatitis, Dr. Herman H. Tam reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“We know that for specific allergen immunotherapy, there have been really good results in allergic rhinitis and venom allergy. For atopic dermatitis, however, dating back almost 40 years, we found there have only been 12 randomized trials using standardized allergen extracts. The quality of evidence was low, and the study findings have been inconsistent,” Dr. Tam, first author of the Cochrane review, said in an interview.
The dozen trials included a total of 733 children and adults in nine countries. Because of insufficient follow-up in most of the studies, coupled with the use of a variety of endpoints, the analysis concluded that “specific allergen immunotherapy cannot be recommended for atopic eczema at present” (Cochrane Database Syst Rev. 2016 Feb 12;2:CD008774).
“We found no consistent evidence that specific allergen immunotherapy provides a treatment benefit for people with allergic eczema, compared with placebo or no treatment. The message of this review is that we need more large randomized trials with better controls, modern high-quality allergen extracts that have proven themselves in other allergic diseases, and patient-centered outcome measures,” according to Dr. Tam, who participated in the Cochrane review while at Imperial College London and is now a pediatric resident at the University of Manitoba, Winnipeg.
He reported having no relevant financial conflicts.
LOS ANGELES – A new Cochrane systematic review and meta-analysis has concluded there is no consistent evidence that specific allergen immunotherapy is beneficial in patients with atopic dermatitis, Dr. Herman H. Tam reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“We know that for specific allergen immunotherapy, there have been really good results in allergic rhinitis and venom allergy. For atopic dermatitis, however, dating back almost 40 years, we found there have only been 12 randomized trials using standardized allergen extracts. The quality of evidence was low, and the study findings have been inconsistent,” Dr. Tam, first author of the Cochrane review, said in an interview.
The dozen trials included a total of 733 children and adults in nine countries. Because of insufficient follow-up in most of the studies, coupled with the use of a variety of endpoints, the analysis concluded that “specific allergen immunotherapy cannot be recommended for atopic eczema at present” (Cochrane Database Syst Rev. 2016 Feb 12;2:CD008774).
“We found no consistent evidence that specific allergen immunotherapy provides a treatment benefit for people with allergic eczema, compared with placebo or no treatment. The message of this review is that we need more large randomized trials with better controls, modern high-quality allergen extracts that have proven themselves in other allergic diseases, and patient-centered outcome measures,” according to Dr. Tam, who participated in the Cochrane review while at Imperial College London and is now a pediatric resident at the University of Manitoba, Winnipeg.
He reported having no relevant financial conflicts.
LOS ANGELES – A new Cochrane systematic review and meta-analysis has concluded there is no consistent evidence that specific allergen immunotherapy is beneficial in patients with atopic dermatitis, Dr. Herman H. Tam reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“We know that for specific allergen immunotherapy, there have been really good results in allergic rhinitis and venom allergy. For atopic dermatitis, however, dating back almost 40 years, we found there have only been 12 randomized trials using standardized allergen extracts. The quality of evidence was low, and the study findings have been inconsistent,” Dr. Tam, first author of the Cochrane review, said in an interview.
The dozen trials included a total of 733 children and adults in nine countries. Because of insufficient follow-up in most of the studies, coupled with the use of a variety of endpoints, the analysis concluded that “specific allergen immunotherapy cannot be recommended for atopic eczema at present” (Cochrane Database Syst Rev. 2016 Feb 12;2:CD008774).
“We found no consistent evidence that specific allergen immunotherapy provides a treatment benefit for people with allergic eczema, compared with placebo or no treatment. The message of this review is that we need more large randomized trials with better controls, modern high-quality allergen extracts that have proven themselves in other allergic diseases, and patient-centered outcome measures,” according to Dr. Tam, who participated in the Cochrane review while at Imperial College London and is now a pediatric resident at the University of Manitoba, Winnipeg.
He reported having no relevant financial conflicts.
AT 2016 AAAAI ANNUAL MEETING
Key clinical point: The available evidence doesn’t support using specific allergen immunotherapy for atopic dermatitis.
Major finding: A new Cochrane systematic review and meta-analysis has found “no consistent evidence” that specific allergen immunotherapy is more effective than placebo in treating atopic dermatitis.
Data source: This was a Cochrane systematic review and meta-analysis of 12 randomized controlled trials of specific allergen immunotherapy in 733 pediatric and adult atopic dermatitis patients.
Disclosures: The presenter reported having no relevant financial conflicts of interest.