User login
Older Men Less Likely to Receive Osteoporosis Screening and Treatment After Bone Fracture
Significantly fewer men received evaluation for osteoporosis following a distal radial fracture, with rates of evaluation unacceptably low according to published guidelines, according to a study published November 5 in Journal of Bone & Joint Surgery.
Treating men for bone fractures, but not the underlying cause, places them at a greater risk for future bone breaks and related complications,” said study investigator Tamara Rozental, MD, Associate Professor of Orthopaedic Surgery, Harvard Medical School in Boston, Massachusetts.
In this study, researchers reviewed the medical records of 95 men and 344 women older than 50, who were treated for a wrist fracture at a single institution between 2007 and 2012. Data collected included age, mechanism of injury, fracture severity, associated comorbidities, and type of treatment. Fractures were classified according to the AO Foundation and Orthopaedic Trauma Association (AO/OTA) classification system. Comorbidities were estimated with use of the Charlson comorbidity index (CCI), providing a weighted score to predict short and long-term outcomes, taking into account the number and severity of predefined comorbid conditions. The WHO online Fracture Risk Assessment Tool (FRAX) was used to estimate the 10-year risk of major osteoporotic fractures in men.
Patient injuries were assessed to determine whether or not they were screened for osteoporosis before their injury and/or if they received a dual-energy x-ray absorptiometry scan and osteoporosis treatment within six months following their wrist fracture.
Fewer men than women underwent bone mass density testing prior to their fracture. Following the wrist fracture, the number of men undergoing osteoporosis assessment continued to be lower, 184 (53%) of women versus 17 (18%) of men.
Study findings also indicate:
• Twenty-one percent of men compared with 55% of women initiated treatment with calcium and vitamin D supplements within six months of injury, and 3% of men versus 22% of women started taking bisphosphonates.
• Using the World Health Organization (WHO) online Fracture Risk Assessment Tool (FRAX), 50% of men who obtained a bone density test were deemed at risk for a second major osteoporotic fracture in the next decade.
• Male sex, less severe fracture patterns and high-energy mechanism of injury were independent predictors of failure to initiate treatment with calcium and vitamin D.
• Overall, the men had less severe fractures than women with 20% of the men and 40% of the women in the study having a “Type-C” fracture.
“The results of this study lead us to suggest that men over the age of 50 with fractures of the distal radius should undergo bone density testing and evaluation with the FRAX algorithm to better identify those at high risk for future fracture and those who would benefit from further treatment,” said Dr. Rozental.
Suggested Reading
Harper CM, Fitzpatrick SK, Zurakowski D, Rozental TD. Distal radial fractures in older men: a missed opportunity? J Bone Joint Surg Am. 2014;96(21):1820-7.
Significantly fewer men received evaluation for osteoporosis following a distal radial fracture, with rates of evaluation unacceptably low according to published guidelines, according to a study published November 5 in Journal of Bone & Joint Surgery.
Treating men for bone fractures, but not the underlying cause, places them at a greater risk for future bone breaks and related complications,” said study investigator Tamara Rozental, MD, Associate Professor of Orthopaedic Surgery, Harvard Medical School in Boston, Massachusetts.
In this study, researchers reviewed the medical records of 95 men and 344 women older than 50, who were treated for a wrist fracture at a single institution between 2007 and 2012. Data collected included age, mechanism of injury, fracture severity, associated comorbidities, and type of treatment. Fractures were classified according to the AO Foundation and Orthopaedic Trauma Association (AO/OTA) classification system. Comorbidities were estimated with use of the Charlson comorbidity index (CCI), providing a weighted score to predict short and long-term outcomes, taking into account the number and severity of predefined comorbid conditions. The WHO online Fracture Risk Assessment Tool (FRAX) was used to estimate the 10-year risk of major osteoporotic fractures in men.
Patient injuries were assessed to determine whether or not they were screened for osteoporosis before their injury and/or if they received a dual-energy x-ray absorptiometry scan and osteoporosis treatment within six months following their wrist fracture.
Fewer men than women underwent bone mass density testing prior to their fracture. Following the wrist fracture, the number of men undergoing osteoporosis assessment continued to be lower, 184 (53%) of women versus 17 (18%) of men.
Study findings also indicate:
• Twenty-one percent of men compared with 55% of women initiated treatment with calcium and vitamin D supplements within six months of injury, and 3% of men versus 22% of women started taking bisphosphonates.
• Using the World Health Organization (WHO) online Fracture Risk Assessment Tool (FRAX), 50% of men who obtained a bone density test were deemed at risk for a second major osteoporotic fracture in the next decade.
• Male sex, less severe fracture patterns and high-energy mechanism of injury were independent predictors of failure to initiate treatment with calcium and vitamin D.
• Overall, the men had less severe fractures than women with 20% of the men and 40% of the women in the study having a “Type-C” fracture.
“The results of this study lead us to suggest that men over the age of 50 with fractures of the distal radius should undergo bone density testing and evaluation with the FRAX algorithm to better identify those at high risk for future fracture and those who would benefit from further treatment,” said Dr. Rozental.
Significantly fewer men received evaluation for osteoporosis following a distal radial fracture, with rates of evaluation unacceptably low according to published guidelines, according to a study published November 5 in Journal of Bone & Joint Surgery.
Treating men for bone fractures, but not the underlying cause, places them at a greater risk for future bone breaks and related complications,” said study investigator Tamara Rozental, MD, Associate Professor of Orthopaedic Surgery, Harvard Medical School in Boston, Massachusetts.
In this study, researchers reviewed the medical records of 95 men and 344 women older than 50, who were treated for a wrist fracture at a single institution between 2007 and 2012. Data collected included age, mechanism of injury, fracture severity, associated comorbidities, and type of treatment. Fractures were classified according to the AO Foundation and Orthopaedic Trauma Association (AO/OTA) classification system. Comorbidities were estimated with use of the Charlson comorbidity index (CCI), providing a weighted score to predict short and long-term outcomes, taking into account the number and severity of predefined comorbid conditions. The WHO online Fracture Risk Assessment Tool (FRAX) was used to estimate the 10-year risk of major osteoporotic fractures in men.
Patient injuries were assessed to determine whether or not they were screened for osteoporosis before their injury and/or if they received a dual-energy x-ray absorptiometry scan and osteoporosis treatment within six months following their wrist fracture.
Fewer men than women underwent bone mass density testing prior to their fracture. Following the wrist fracture, the number of men undergoing osteoporosis assessment continued to be lower, 184 (53%) of women versus 17 (18%) of men.
Study findings also indicate:
• Twenty-one percent of men compared with 55% of women initiated treatment with calcium and vitamin D supplements within six months of injury, and 3% of men versus 22% of women started taking bisphosphonates.
• Using the World Health Organization (WHO) online Fracture Risk Assessment Tool (FRAX), 50% of men who obtained a bone density test were deemed at risk for a second major osteoporotic fracture in the next decade.
• Male sex, less severe fracture patterns and high-energy mechanism of injury were independent predictors of failure to initiate treatment with calcium and vitamin D.
• Overall, the men had less severe fractures than women with 20% of the men and 40% of the women in the study having a “Type-C” fracture.
“The results of this study lead us to suggest that men over the age of 50 with fractures of the distal radius should undergo bone density testing and evaluation with the FRAX algorithm to better identify those at high risk for future fracture and those who would benefit from further treatment,” said Dr. Rozental.
Suggested Reading
Harper CM, Fitzpatrick SK, Zurakowski D, Rozental TD. Distal radial fractures in older men: a missed opportunity? J Bone Joint Surg Am. 2014;96(21):1820-7.
Suggested Reading
Harper CM, Fitzpatrick SK, Zurakowski D, Rozental TD. Distal radial fractures in older men: a missed opportunity? J Bone Joint Surg Am. 2014;96(21):1820-7.
Second Preclinical Autoimmune Disease Proof of Concept Established for INV-17
BOSTON—Innovimmune Biotherapeutics Holding, LLC (Brooklyn, New York) presented data demonstrating successful treatment of rheumatoid arthritis in a murine collagen-induced arthritis (CIA) model with its proprietary oral small molecule Retinoic acid receptor-related Orphan Receptor gamma t (RORγt) modulators from their INV-17 portfolio. The results were presented at the 2014 Annual Meeting of the American College of Rheumatology.
In the CIA study, an INV-17 RORγt modulator lead compound was administered orally for 28 days in a therapeutic regimen following rheumatoid arthritis disease induction. The data demonstrate that mice treated with INV-17 achieved statistically significant reduction in cumulative arthritis score as the primary study end point, in contrast to a vehicle (placebo) group.
Significant improvement in clinical disease scores in the INV-17 group began on day 13, with maximal therapeutic effects observed on day 16 through day 26 and through the end of the study.
“This is a remarkable finding in that a novel therapeutic approach targeting pathogenic T helper 17 (TH17) cells through RORγt modulation provides superior preclinical treatment efficacy in rheumatoid arthritis. These results, which demonstrate successful rheumatoid arthritis disease amelioration in the absence of toxicity, may provide a novel oral disease-modifying antirheumatic drug treatment strategy with an oral INV-17 drug for rheumatoid arthritis and other TH17-mediated autoimmune diseases,” said Ellen M. Ginzler, MD, MPH, Distinguished Teaching Professor of Medicine and Chief, Division of Rheumatology, SUNY Downstate Medical Center in Brooklyn, New York.
BOSTON—Innovimmune Biotherapeutics Holding, LLC (Brooklyn, New York) presented data demonstrating successful treatment of rheumatoid arthritis in a murine collagen-induced arthritis (CIA) model with its proprietary oral small molecule Retinoic acid receptor-related Orphan Receptor gamma t (RORγt) modulators from their INV-17 portfolio. The results were presented at the 2014 Annual Meeting of the American College of Rheumatology.
In the CIA study, an INV-17 RORγt modulator lead compound was administered orally for 28 days in a therapeutic regimen following rheumatoid arthritis disease induction. The data demonstrate that mice treated with INV-17 achieved statistically significant reduction in cumulative arthritis score as the primary study end point, in contrast to a vehicle (placebo) group.
Significant improvement in clinical disease scores in the INV-17 group began on day 13, with maximal therapeutic effects observed on day 16 through day 26 and through the end of the study.
“This is a remarkable finding in that a novel therapeutic approach targeting pathogenic T helper 17 (TH17) cells through RORγt modulation provides superior preclinical treatment efficacy in rheumatoid arthritis. These results, which demonstrate successful rheumatoid arthritis disease amelioration in the absence of toxicity, may provide a novel oral disease-modifying antirheumatic drug treatment strategy with an oral INV-17 drug for rheumatoid arthritis and other TH17-mediated autoimmune diseases,” said Ellen M. Ginzler, MD, MPH, Distinguished Teaching Professor of Medicine and Chief, Division of Rheumatology, SUNY Downstate Medical Center in Brooklyn, New York.
BOSTON—Innovimmune Biotherapeutics Holding, LLC (Brooklyn, New York) presented data demonstrating successful treatment of rheumatoid arthritis in a murine collagen-induced arthritis (CIA) model with its proprietary oral small molecule Retinoic acid receptor-related Orphan Receptor gamma t (RORγt) modulators from their INV-17 portfolio. The results were presented at the 2014 Annual Meeting of the American College of Rheumatology.
In the CIA study, an INV-17 RORγt modulator lead compound was administered orally for 28 days in a therapeutic regimen following rheumatoid arthritis disease induction. The data demonstrate that mice treated with INV-17 achieved statistically significant reduction in cumulative arthritis score as the primary study end point, in contrast to a vehicle (placebo) group.
Significant improvement in clinical disease scores in the INV-17 group began on day 13, with maximal therapeutic effects observed on day 16 through day 26 and through the end of the study.
“This is a remarkable finding in that a novel therapeutic approach targeting pathogenic T helper 17 (TH17) cells through RORγt modulation provides superior preclinical treatment efficacy in rheumatoid arthritis. These results, which demonstrate successful rheumatoid arthritis disease amelioration in the absence of toxicity, may provide a novel oral disease-modifying antirheumatic drug treatment strategy with an oral INV-17 drug for rheumatoid arthritis and other TH17-mediated autoimmune diseases,” said Ellen M. Ginzler, MD, MPH, Distinguished Teaching Professor of Medicine and Chief, Division of Rheumatology, SUNY Downstate Medical Center in Brooklyn, New York.
Data Show Exparels’ Ability to Treat Postsurgical Pain Following Total Knee Arthroplasty
DALLAS—Results of an independent, physician-initiated study designed to evaluate the difference in postsurgical pain and opioid consumption between patients who received Exparel (bupivacaine liposome injectable suspension) versus a multi-drug analgesic cocktail for pain management following total knee arthroplasty (TKA) were presented at the 24th Annual Meeting of the American Association of Hip and Knee Surgeons. Researchers found that patients treated with Exparel reported significantly lower patient-perceived pain scores and morphine sulfate equivalence consumption, and reported higher satisfaction with pain control and overall experience compared with patients who received the multi-drug analgesic cocktail.
“A majority of patients who undergo total knee arthroplasty report dissatisfaction with overall pain control and side effects associated with narcotic medications, so there is clearly a need for a more effective and better-tolerated pain management option,” said Mark A. Snyder, MD, Director of the Orthopaedic Center of Excellence at Good Samaritan Hospital in Cincinnati.
In the double-blind, randomized clinical study, 70 patients who underwent a TKA were randomly assigned to receive either a periarticular injection with Exparel or a multi-drug analgesic cocktail for postsurgical analgesia.
Findings showed that compared with patients who received the multi-drug analgesic cocktail, patients who received EXPAREL reported:
• Significantly lower pain levels on post-op days one and two.
• Higher satisfaction in pain control and overall experience.
• Significantly fewer adverse events.
• Significantly lower total morphine equivalency consumption in the postanesthesia care unit (PACU) and by post-op day two.
“Our study found that Exparel not only provided effective pain control, but also reduced opioid load and improved the patient’s overall experience. In addition, we found that Exparel eliminated the incidence of post-operative falls, a serious patient safety risk resulting from muscle weakness associated with nerve blocks and prolonged indwelling pain catheters, and confusion or disorientation caused by opioids,” stated Dr. Snyder.
Exparel is marketed by Pacira Pharmaceuticals Inc, in Parsippany, New Jersey.
DALLAS—Results of an independent, physician-initiated study designed to evaluate the difference in postsurgical pain and opioid consumption between patients who received Exparel (bupivacaine liposome injectable suspension) versus a multi-drug analgesic cocktail for pain management following total knee arthroplasty (TKA) were presented at the 24th Annual Meeting of the American Association of Hip and Knee Surgeons. Researchers found that patients treated with Exparel reported significantly lower patient-perceived pain scores and morphine sulfate equivalence consumption, and reported higher satisfaction with pain control and overall experience compared with patients who received the multi-drug analgesic cocktail.
“A majority of patients who undergo total knee arthroplasty report dissatisfaction with overall pain control and side effects associated with narcotic medications, so there is clearly a need for a more effective and better-tolerated pain management option,” said Mark A. Snyder, MD, Director of the Orthopaedic Center of Excellence at Good Samaritan Hospital in Cincinnati.
In the double-blind, randomized clinical study, 70 patients who underwent a TKA were randomly assigned to receive either a periarticular injection with Exparel or a multi-drug analgesic cocktail for postsurgical analgesia.
Findings showed that compared with patients who received the multi-drug analgesic cocktail, patients who received EXPAREL reported:
• Significantly lower pain levels on post-op days one and two.
• Higher satisfaction in pain control and overall experience.
• Significantly fewer adverse events.
• Significantly lower total morphine equivalency consumption in the postanesthesia care unit (PACU) and by post-op day two.
“Our study found that Exparel not only provided effective pain control, but also reduced opioid load and improved the patient’s overall experience. In addition, we found that Exparel eliminated the incidence of post-operative falls, a serious patient safety risk resulting from muscle weakness associated with nerve blocks and prolonged indwelling pain catheters, and confusion or disorientation caused by opioids,” stated Dr. Snyder.
Exparel is marketed by Pacira Pharmaceuticals Inc, in Parsippany, New Jersey.
DALLAS—Results of an independent, physician-initiated study designed to evaluate the difference in postsurgical pain and opioid consumption between patients who received Exparel (bupivacaine liposome injectable suspension) versus a multi-drug analgesic cocktail for pain management following total knee arthroplasty (TKA) were presented at the 24th Annual Meeting of the American Association of Hip and Knee Surgeons. Researchers found that patients treated with Exparel reported significantly lower patient-perceived pain scores and morphine sulfate equivalence consumption, and reported higher satisfaction with pain control and overall experience compared with patients who received the multi-drug analgesic cocktail.
“A majority of patients who undergo total knee arthroplasty report dissatisfaction with overall pain control and side effects associated with narcotic medications, so there is clearly a need for a more effective and better-tolerated pain management option,” said Mark A. Snyder, MD, Director of the Orthopaedic Center of Excellence at Good Samaritan Hospital in Cincinnati.
In the double-blind, randomized clinical study, 70 patients who underwent a TKA were randomly assigned to receive either a periarticular injection with Exparel or a multi-drug analgesic cocktail for postsurgical analgesia.
Findings showed that compared with patients who received the multi-drug analgesic cocktail, patients who received EXPAREL reported:
• Significantly lower pain levels on post-op days one and two.
• Higher satisfaction in pain control and overall experience.
• Significantly fewer adverse events.
• Significantly lower total morphine equivalency consumption in the postanesthesia care unit (PACU) and by post-op day two.
“Our study found that Exparel not only provided effective pain control, but also reduced opioid load and improved the patient’s overall experience. In addition, we found that Exparel eliminated the incidence of post-operative falls, a serious patient safety risk resulting from muscle weakness associated with nerve blocks and prolonged indwelling pain catheters, and confusion or disorientation caused by opioids,” stated Dr. Snyder.
Exparel is marketed by Pacira Pharmaceuticals Inc, in Parsippany, New Jersey.
Osteoporosis Drug’s Benefit to Cells Touted in Study
Experiments in mice with a bone disorder similar to osteoporosis after menopause show that an overlooked group of cells are likely crucial to the process of bone loss caused by the disorder, according to a study published online ahead of print October 5 in Nature Medicine. The finding, researchers say, not only raises the research profile of the cells, called preosteoclasts, but also explains the success and activity of an experimental osteoporosis drug currently in phase III clinical trials.
“We didn’t know that the drug affects preosteoclasts, nor did we understand how important preosteoclasts are in maintaining healthy bones,” says Xu Cao, PhD, the Lee H. Riley Jr., MD, Professor of Orthopaedic Surgery at Johns Hopkins University in Baltimore. “Now drug companies hoping to reverse osteoporosis can look for even more drugs that make use of and target these interesting cells.”
Dr. Cao and colleagues grew two cell types separately in the laboratory and collected the liquid around them to test for proteins released by the cells. They found that preosteoclasts, but not mature osteoclasts, secrete platelet-derived growth factor-BB (PDGF-BB). When the preosteoclasts of mice were prevented from making PDGF-BB, the mice had weak bones.
When the mice were given L-235, the animal form of odanacatib, the numbers of their preosteoclasts and osteoclasts increased, and they secreted more PDGF-BB. The increased PDGF-BB brought in more cells for making blood vessels and bone, which led to more of the specialized blood vessels and thicker bones.
To see if the drug could help reverse the increased bone resorption and decreased blood vessel formation of postmenopausal osteoporosis, the investigators simulated menopause in female mice by removing their ovaries.
At first, the mice had thinner bones and fewer blood vessels, but treatment with the drug increased the concentration of PDGF-BB in the blood, the number of specialized blood vessels both inside and outside of the bones, and the overall thickness and density of the bone.
According to Dr. Cao, in addition to slowing bone resorption by blocking cathepsin K, the drug also appears to slow the maturation of preosteoclasts, lengthening the amount of time they secrete PDGF-BB before becoming osteoclasts. With increased PDGF-BB, more specialized blood vessels are made and more bone-building cells arrive, restoring the balance between bone resorption and bone rebuilding.
Odanacatib is produced by Merck & Co. Inc. (Whitehouse Station, New Jersey) and has already gone through phase III clinical trials with good results, according to Dr. Cao.
“It is unusual to see a single drug that decreases bone resorption and increases bone rebuilding at the same time,” Dr. Cao said.
Suggested Reading
Xie H, Cui Z, Wang L, et al. PDGF-BB secreted by preosteoclasts induces angiogenesis during coupling with osteogenesis. Nat Med. 2014 Oct 5. [Epub ahead of print].
Experiments in mice with a bone disorder similar to osteoporosis after menopause show that an overlooked group of cells are likely crucial to the process of bone loss caused by the disorder, according to a study published online ahead of print October 5 in Nature Medicine. The finding, researchers say, not only raises the research profile of the cells, called preosteoclasts, but also explains the success and activity of an experimental osteoporosis drug currently in phase III clinical trials.
“We didn’t know that the drug affects preosteoclasts, nor did we understand how important preosteoclasts are in maintaining healthy bones,” says Xu Cao, PhD, the Lee H. Riley Jr., MD, Professor of Orthopaedic Surgery at Johns Hopkins University in Baltimore. “Now drug companies hoping to reverse osteoporosis can look for even more drugs that make use of and target these interesting cells.”
Dr. Cao and colleagues grew two cell types separately in the laboratory and collected the liquid around them to test for proteins released by the cells. They found that preosteoclasts, but not mature osteoclasts, secrete platelet-derived growth factor-BB (PDGF-BB). When the preosteoclasts of mice were prevented from making PDGF-BB, the mice had weak bones.
When the mice were given L-235, the animal form of odanacatib, the numbers of their preosteoclasts and osteoclasts increased, and they secreted more PDGF-BB. The increased PDGF-BB brought in more cells for making blood vessels and bone, which led to more of the specialized blood vessels and thicker bones.
To see if the drug could help reverse the increased bone resorption and decreased blood vessel formation of postmenopausal osteoporosis, the investigators simulated menopause in female mice by removing their ovaries.
At first, the mice had thinner bones and fewer blood vessels, but treatment with the drug increased the concentration of PDGF-BB in the blood, the number of specialized blood vessels both inside and outside of the bones, and the overall thickness and density of the bone.
According to Dr. Cao, in addition to slowing bone resorption by blocking cathepsin K, the drug also appears to slow the maturation of preosteoclasts, lengthening the amount of time they secrete PDGF-BB before becoming osteoclasts. With increased PDGF-BB, more specialized blood vessels are made and more bone-building cells arrive, restoring the balance between bone resorption and bone rebuilding.
Odanacatib is produced by Merck & Co. Inc. (Whitehouse Station, New Jersey) and has already gone through phase III clinical trials with good results, according to Dr. Cao.
“It is unusual to see a single drug that decreases bone resorption and increases bone rebuilding at the same time,” Dr. Cao said.
Experiments in mice with a bone disorder similar to osteoporosis after menopause show that an overlooked group of cells are likely crucial to the process of bone loss caused by the disorder, according to a study published online ahead of print October 5 in Nature Medicine. The finding, researchers say, not only raises the research profile of the cells, called preosteoclasts, but also explains the success and activity of an experimental osteoporosis drug currently in phase III clinical trials.
“We didn’t know that the drug affects preosteoclasts, nor did we understand how important preosteoclasts are in maintaining healthy bones,” says Xu Cao, PhD, the Lee H. Riley Jr., MD, Professor of Orthopaedic Surgery at Johns Hopkins University in Baltimore. “Now drug companies hoping to reverse osteoporosis can look for even more drugs that make use of and target these interesting cells.”
Dr. Cao and colleagues grew two cell types separately in the laboratory and collected the liquid around them to test for proteins released by the cells. They found that preosteoclasts, but not mature osteoclasts, secrete platelet-derived growth factor-BB (PDGF-BB). When the preosteoclasts of mice were prevented from making PDGF-BB, the mice had weak bones.
When the mice were given L-235, the animal form of odanacatib, the numbers of their preosteoclasts and osteoclasts increased, and they secreted more PDGF-BB. The increased PDGF-BB brought in more cells for making blood vessels and bone, which led to more of the specialized blood vessels and thicker bones.
To see if the drug could help reverse the increased bone resorption and decreased blood vessel formation of postmenopausal osteoporosis, the investigators simulated menopause in female mice by removing their ovaries.
At first, the mice had thinner bones and fewer blood vessels, but treatment with the drug increased the concentration of PDGF-BB in the blood, the number of specialized blood vessels both inside and outside of the bones, and the overall thickness and density of the bone.
According to Dr. Cao, in addition to slowing bone resorption by blocking cathepsin K, the drug also appears to slow the maturation of preosteoclasts, lengthening the amount of time they secrete PDGF-BB before becoming osteoclasts. With increased PDGF-BB, more specialized blood vessels are made and more bone-building cells arrive, restoring the balance between bone resorption and bone rebuilding.
Odanacatib is produced by Merck & Co. Inc. (Whitehouse Station, New Jersey) and has already gone through phase III clinical trials with good results, according to Dr. Cao.
“It is unusual to see a single drug that decreases bone resorption and increases bone rebuilding at the same time,” Dr. Cao said.
Suggested Reading
Xie H, Cui Z, Wang L, et al. PDGF-BB secreted by preosteoclasts induces angiogenesis during coupling with osteogenesis. Nat Med. 2014 Oct 5. [Epub ahead of print].
Suggested Reading
Xie H, Cui Z, Wang L, et al. PDGF-BB secreted by preosteoclasts induces angiogenesis during coupling with osteogenesis. Nat Med. 2014 Oct 5. [Epub ahead of print].
Teenage Baseball Pitchers at Increased Risk of Permanent Shoulder Injury
Young baseball pitchers who throw more than 100 pitches per week are at risk for a newly identified overuse injury that can impede normal shoulder development and lead to additional problems, including rotator cuff tears, according to a study published online ahead of print October 14 in Radiology.
The injury, termed acromial apophysiolysis by the researchers, is characterized by incomplete fusion and tenderness at the acromion. The acromion, which forms the bone at the top of the shoulder, typically develops from four individual bones into one bone during the teenage years.
“We kept seeing this injury over and over again in young athletes who come to the hospital at the end of the baseball season with shoulder pain and edema at the acromion on MRI, but no other imaging findings,” said Johannes B. Roedl, MD, a radiologist in the Musculoskeletal Division at Thomas Jefferson University Hospital in Philadelphia.
Dr. Roedl and a team of researchers conducted a retrospective study of 2,372 consecutive patients between the ages of 15 and 25, who underwent magnetic resonance imaging (MRI) for shoulder pain between 1998 and 2012. The majority of the patients, which included both males and females, were baseball pitchers.
Patients with edema at the acromial apophyses and no other abnormalities on MRI were included in the study group. Association of acromial edema with incomplete fusion , pitching, and clinical findings was determined in the study group and in an age- and sex-matched control group. Association with the development of an os acromial and rotator cuff tears later in life was assessed with follow-up imaging after age 25.
Edema at the acromial apophyses was found in 2.6% (61 of 2,372) and was associated with incomplete fusion of the acromial apophyses and superior shoulder tenderness.
A pitch count of more than 100 pitches per week was a substantial risk factor for developing acromial apophysiolysis (odds ratio 6.5). Among the patients with this overuse injury, 40% threw more than 100 pitches per week, compared to 8% in the control group.
All 61 injured patients took a three-month rest from pitching. One patient underwent surgery while the remaining 60 patients were treated conservatively with non-steroidal pain medication. Follow-up imaging conducted a minimum of two years later after the patients turned 25 were available for 29 of the 61 injured patients and for 23 of the 61 controls. Follow-up imaging revealed that 25 of the 29 patients (86%) with the overuse injury showed incomplete fusion of the acromion, compared to only one of the 23 (4%) controls.
Twenty-one of the 29 patients with the overuse injury continued pitching after the rest period, and all 21 showed incomplete bone fusion at the acromion. Rotator cuff tears were significantly more common among this group than in the control group (68% versus 29%, respectively). The severity of the rotator cuff tears was also higher in the overuse injury group compared with the control group.
“More and more kids are entering sports earlier in life and are overtraining,” said Dr. Roedl. “Baseball players who pitch too much are at risk of developing a stress response and overuse injury to the acromion. It is important to limit stress to the growing bones to allow them to develop normally.”
Suggested Reading
Roedl JB, Morrison WB, Ciccotti MG, Zoga AC. Acromial apophysiolysis: superior shoulder pain and acromial nonfusion in the young throwing athlete. Radiology. 2014 Oct 14:140587 [Epub ahead of print].
Young baseball pitchers who throw more than 100 pitches per week are at risk for a newly identified overuse injury that can impede normal shoulder development and lead to additional problems, including rotator cuff tears, according to a study published online ahead of print October 14 in Radiology.
The injury, termed acromial apophysiolysis by the researchers, is characterized by incomplete fusion and tenderness at the acromion. The acromion, which forms the bone at the top of the shoulder, typically develops from four individual bones into one bone during the teenage years.
“We kept seeing this injury over and over again in young athletes who come to the hospital at the end of the baseball season with shoulder pain and edema at the acromion on MRI, but no other imaging findings,” said Johannes B. Roedl, MD, a radiologist in the Musculoskeletal Division at Thomas Jefferson University Hospital in Philadelphia.
Dr. Roedl and a team of researchers conducted a retrospective study of 2,372 consecutive patients between the ages of 15 and 25, who underwent magnetic resonance imaging (MRI) for shoulder pain between 1998 and 2012. The majority of the patients, which included both males and females, were baseball pitchers.
Patients with edema at the acromial apophyses and no other abnormalities on MRI were included in the study group. Association of acromial edema with incomplete fusion , pitching, and clinical findings was determined in the study group and in an age- and sex-matched control group. Association with the development of an os acromial and rotator cuff tears later in life was assessed with follow-up imaging after age 25.
Edema at the acromial apophyses was found in 2.6% (61 of 2,372) and was associated with incomplete fusion of the acromial apophyses and superior shoulder tenderness.
A pitch count of more than 100 pitches per week was a substantial risk factor for developing acromial apophysiolysis (odds ratio 6.5). Among the patients with this overuse injury, 40% threw more than 100 pitches per week, compared to 8% in the control group.
All 61 injured patients took a three-month rest from pitching. One patient underwent surgery while the remaining 60 patients were treated conservatively with non-steroidal pain medication. Follow-up imaging conducted a minimum of two years later after the patients turned 25 were available for 29 of the 61 injured patients and for 23 of the 61 controls. Follow-up imaging revealed that 25 of the 29 patients (86%) with the overuse injury showed incomplete fusion of the acromion, compared to only one of the 23 (4%) controls.
Twenty-one of the 29 patients with the overuse injury continued pitching after the rest period, and all 21 showed incomplete bone fusion at the acromion. Rotator cuff tears were significantly more common among this group than in the control group (68% versus 29%, respectively). The severity of the rotator cuff tears was also higher in the overuse injury group compared with the control group.
“More and more kids are entering sports earlier in life and are overtraining,” said Dr. Roedl. “Baseball players who pitch too much are at risk of developing a stress response and overuse injury to the acromion. It is important to limit stress to the growing bones to allow them to develop normally.”
Young baseball pitchers who throw more than 100 pitches per week are at risk for a newly identified overuse injury that can impede normal shoulder development and lead to additional problems, including rotator cuff tears, according to a study published online ahead of print October 14 in Radiology.
The injury, termed acromial apophysiolysis by the researchers, is characterized by incomplete fusion and tenderness at the acromion. The acromion, which forms the bone at the top of the shoulder, typically develops from four individual bones into one bone during the teenage years.
“We kept seeing this injury over and over again in young athletes who come to the hospital at the end of the baseball season with shoulder pain and edema at the acromion on MRI, but no other imaging findings,” said Johannes B. Roedl, MD, a radiologist in the Musculoskeletal Division at Thomas Jefferson University Hospital in Philadelphia.
Dr. Roedl and a team of researchers conducted a retrospective study of 2,372 consecutive patients between the ages of 15 and 25, who underwent magnetic resonance imaging (MRI) for shoulder pain between 1998 and 2012. The majority of the patients, which included both males and females, were baseball pitchers.
Patients with edema at the acromial apophyses and no other abnormalities on MRI were included in the study group. Association of acromial edema with incomplete fusion , pitching, and clinical findings was determined in the study group and in an age- and sex-matched control group. Association with the development of an os acromial and rotator cuff tears later in life was assessed with follow-up imaging after age 25.
Edema at the acromial apophyses was found in 2.6% (61 of 2,372) and was associated with incomplete fusion of the acromial apophyses and superior shoulder tenderness.
A pitch count of more than 100 pitches per week was a substantial risk factor for developing acromial apophysiolysis (odds ratio 6.5). Among the patients with this overuse injury, 40% threw more than 100 pitches per week, compared to 8% in the control group.
All 61 injured patients took a three-month rest from pitching. One patient underwent surgery while the remaining 60 patients were treated conservatively with non-steroidal pain medication. Follow-up imaging conducted a minimum of two years later after the patients turned 25 were available for 29 of the 61 injured patients and for 23 of the 61 controls. Follow-up imaging revealed that 25 of the 29 patients (86%) with the overuse injury showed incomplete fusion of the acromion, compared to only one of the 23 (4%) controls.
Twenty-one of the 29 patients with the overuse injury continued pitching after the rest period, and all 21 showed incomplete bone fusion at the acromion. Rotator cuff tears were significantly more common among this group than in the control group (68% versus 29%, respectively). The severity of the rotator cuff tears was also higher in the overuse injury group compared with the control group.
“More and more kids are entering sports earlier in life and are overtraining,” said Dr. Roedl. “Baseball players who pitch too much are at risk of developing a stress response and overuse injury to the acromion. It is important to limit stress to the growing bones to allow them to develop normally.”
Suggested Reading
Roedl JB, Morrison WB, Ciccotti MG, Zoga AC. Acromial apophysiolysis: superior shoulder pain and acromial nonfusion in the young throwing athlete. Radiology. 2014 Oct 14:140587 [Epub ahead of print].
Suggested Reading
Roedl JB, Morrison WB, Ciccotti MG, Zoga AC. Acromial apophysiolysis: superior shoulder pain and acromial nonfusion in the young throwing athlete. Radiology. 2014 Oct 14:140587 [Epub ahead of print].
Increased Incidence of Patella Baja After Total Knee Arthroplasty Revision for Infection
Patellar height may be important in determining function after total knee arthroplasty (TKA). By altering patellofemoral joint mechanics, patella baja may cause several functional issues after TKA.1-8 Patella baja leads to decreased range of motion (ROM) affecting both extension and flexion.5,8,9 Deep flexion can be restricted in TKA patients with patella baja because of tracking limitations associated with an inferiorly displaced patella. As the knee is brought into flexion, the patella can impinge on the anterior aspect of the tibial polyethylene or the tibial tray—presenting a true block to flexion and potentially altering wear.1,10
Another functional issue with patella baja is loss of strength in the extensor mechanism. The patella serves as a fulcrum for the extensor muscles of the knee. When positioned properly and functioning properly, the patella increases the extensor forces generated. When the patella is positioned in baja, the knee generates decreased extensor mechanism force.6,7 This can result in a lag, with the patient being unable to fully extend the knee. Extension-dependent activities are impaired. Patients with weak extensor function can experience poor function with stair climbing, rising from a chair, and exiting an automobile. The improper function and scarring of the patella can result in increased anterior knee pain and worse functional outcome scores after TKAs.3,9
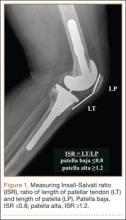
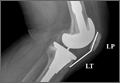
An abnormally positioned patella can either result from or lead to increased scarring in the knee.9,11 Patellar height is often measured with the Insall-Salvati ratio (ISR), which is the patella tendon length (measurement of the tendon from the tibial tubercle to the inferior pole of the patella) divided by the patellar length (longest measured dimension of the patella) (Figure 1).12 Patella baja is defined as an ISR of less than 0.8. Other indices that reference off the tibial plateau (Blackburne-Peel ratio, Canton-Deschamps ratio) reflect an elevation of the joint line, or pseudobaja, and are unreliable for analysis of patella baja after TKA.13
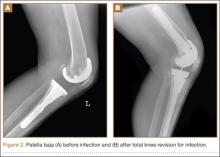
Postoperative patella baja has been reported in 10% to 34% of primary TKAs.4,7 Inferior positioning of the patella and scarring can cause intraoperative difficulty with exposure and may complicate outcomes.9,13 The exposure scar is often larger in TKA revisions for infection compared with primary TKAs.
We conducted a study to compare the incidence of patella baja in noninfected and infected TKA revisions. We hypothesized that, compared with noninfected knees, infected knees treated with nonarticulating spacers would have a higher incidence of patella baja both before and after surgery secondary to more inflammation, immobilization, and related scarring.
Materials and Methods
We conducted a retrospective case–cohort study of 148 consecutive TKA revisions. All TKA revisions were performed between 2003 and 2009 using a mobile-bearing revision system from a single manufacturer. All surgeries were done at a single institution by the 2 senior surgeons. The surgical approach was a standard medial parapatellar approach without patellar eversion. Our institutional review board approved the study and waived the requirement for informed consent, as this was a retrospective study of existing medical records that posed no more than minimal risk to patients.
To properly evaluate patellar height, orthopedic specialty–trained radiologic technicians obtained preoperative and postoperative weight-bearing radiographs using a standardized lateral radiograph in clinic. Two blinded investigators measured ISR radiographically both before surgery (preexplant for septic revisions) and at latest follow-up (postreplant for septic revisions). Patients with inadequate films and/or patellectomies were excluded, along with patients who had less than 6 months of postoperative follow-up.
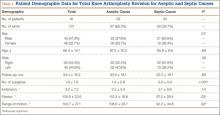
Ninety-one patients (101 TKAs) met the study inclusion criteria. Two groups of cases were compared: aseptic revisions (n = 67) and septic revisions (n = 34). Reasons for aseptic revisions included implant loosening (24/67, 35.8%), instability (12/67, 17.9%), pain (12/67, 17.9%), lysis (5/67, 7.5%), stiffness (3/67, 4.5%), and malrotation (2/67, 3.0%). Infection was determined by Musculoskeletal Infection Society criteria, as documented by positive aspirations and/or intraoperative tissue cultures taken at prosthesis explantation, elevated white blood cell count in the aspirate, elevated percentage of polymorphonuclear (PMN) cells in the aspirate, gross purulence, presence of chronic draining sinus, or histologic analysis revealing acute inflammation with more than 5 PMN cells per high power field.14,15
All infected TKAs were treated with 2-stage revisions. The standard of care at our institution through this series was to use a nonarticulating spacer for the treatment of infection. Weight-bearing status varied by extent of bone damage. Six weeks of culture-specific intravenous antibiotics were administered with assistance from an infectious disease consultant. Reimplantation was performed when clinical and laboratory criteria for resolution of infection were met—specifically, when erythrocyte sedimentation rate was less than 30 mm/h, C-reactive protein level was less than 10 mg/L, and aspirates were culture-negative. Mean (range) follow-up was 33.9 (6.2-75.7) months for aseptic revisions and 32.3 (7.5-94.2) months for septic revisions. Radiographic follow-up was performed at each visit, with weight-bearing anteroposterior and posteroanterior views, along with a lateral knee radiograph. At final follow-up, ROM was recorded by the senior attending evaluating the patient.
Categorical variables were statistically analyzed with χ2 tests, and continuous variables were analyzed with Student t test, analysis of variance, and univariate analysis of covariance (ANCOVA). Statistical significance was set at P < .05. Intrarater reliability was measured with the intraclass correlation coefficient (ICC). All statistical analysis was performed with Predictive Analytics SoftWare Statistics Version 20.0 (SPSS, Chicago, Illinois).
Results
Ninety-one consecutive patients (43 men, 48 women) were included in this study. Mean (SD) age was 66.4 (10.1) years. Mean (SD) preoperative ISR in septic and aseptic cases was 0.94 (0.25) for men and 1.02 (0.23) for women (P = .10). Mean postoperative ISR in septic and aseptic cases was 0.84 (0.27) for men and 0.99 (0.23) for women (P = .004). There was a sex difference between septic and aseptic revisions. There were 22 men and 36 women in the aseptic group and 21 men and 12 women in the septic group (P = .01). Men were more likely than women to have septic revisions and patella baja. Table 1 compares the patient demographics of the 2 patient populations. Mean (SD) number of surgeries, including irrigation and débridement procedures before reimplantation, was larger for septic revisions, 2.9 (0.9), than for aseptic revisions, 1.4 (0.8) (P < .001).
Infection was the most common reason for revision and accounted for 33.7% (34/101) of all revisions. Noninfectious indications, in declining order of frequency, included loosening (23.8%, 24/101), instability (11.9%, 12/101), pain (11.9%, 12/101), osteolysis (5.0%, 5/101), polyethylene wear (5.0%, 5/101), failed unicompartmental knee (4.0%, 4/101), stiffness (3.0%, 3/101), and patellar problems (2.0%, 2/101) (Table 2). ISR decreased significantly only in infected revisions. It is important to note that there was not a high incidence of stiffness or patellofemoral failure in revision patients before surgery.
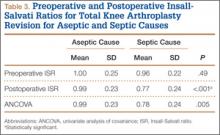
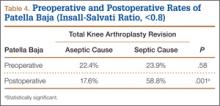
Mean (SD) ISR did not differ between groups before surgery, 1.00 (0.25) for aseptic and 0.96 (0.22) for septic (P = .49), but differed significantly after surgery, 0.99 (0.23) for aseptic and 0.77 (0.24) for septic (P < .001) (Figure 2). The univariate ANCOVA also demonstrated a postoperative difference between groups when taking the preoperative ratio into account: 0.99 (0.23) for aseptic and 0.78 (0.24) for septic (P = .005) (Table 3). Before surgery, 22.4% and 23.9% of the aseptic and septic groups, respectively, had patella baja (P = .58). After surgery, 17.6% and 58.8% of the aseptic and septic groups had patella baja (P = .001) (Table 4). The ICC for preoperative ISR was 0.94, and the ICC for postoperative ISR was 0.96, which indicates excellent agreement of measurements between the 2 blinded investigators.
ROM differed between septic and aseptic groups owing to the difference in postoperative flexion. Mean (SD) postoperative extension was 2.2° (5.4°) for the aseptic group and 5.1° (9.8°) for the septic group—not significantly different (P = .13). Mean (SD) postoperative flexion was 110.2° (18.8°) for the aseptic group and 97.2° (29.4°) for the septic group—significantly different (P = .02). The groups differed significantly (P = .02) in mean (SD) ROM: 108.0° (20.7°) for aseptic and 92.2° (34.6°) for septic (Table 1). ROM was also significantly associated with patella baja (P = .04), as patients with ISR of less than 0.8 had mean (SD) postoperative ROM of 95.1° (31.6°), and patients without patella baja had mean (SD) postoperative ROM of 106.8° (23.6°).
For the septic group, mean (SD) time between first and second stages was 13.0 (8.3) weeks (range, 1-44.3 weeks). Mean (SD) timing of spacer placement was not statistically significantly different (P = .90) between patients who had patella baja, 12.9 (8.8) weeks, and patients who did not have patella baja, 13.2 (7.8) weeks.
Discussion
This study demonstrated that TKAs done for septic reasons resulted in a higher incidence of patella baja and decreased ROM. Incidence of patella baja was higher both before and after revision in septic TKAs than in aseptic TKAs, proving the hypothesis under study. Prerevision incidence was not significantly different, but there was a trend that could not be ignored. This may suggest that there is already an ongoing process in the infected knee that contributes to patella baja; the precise etiology remains unclear and is likely multifactorial. For example, scar formation may be increased in patients with chronic infection, predisposing to patella baja. This assertion is indirectly supported by a recent study from our institution revealing longer average surgical time in septic versus aseptic knee revisions; the difference was thought to reflect increased scar-tissue formation.16 That study also found that patients who underwent septic revisions had significantly more surgical procedures than patients who underwent aseptic revisions. Repetitive surgeries—specifically, repetitive arthrotomies during irrigation and débridement before reimplantation—lead to increased scar formation, which may contribute to preoperative and postoperative patella baja. This may be reflected in the findings that ROM was decreased in patients in the septic group versus patients in the aseptic group and that ROM was decreased in patients with patella baja. In addition, our study found that male patients were more likely to undergo TKA revision for septic reasons and to develop postoperative patella baja. This finding contrasts with that of a study5 that compared preoperative and postoperative ISR in primary TKA and found that women were more likely than men to have patella baja. Although women are more likely to undergo TKA revision,17 men may be more susceptible to infection and subsequent patella baja.
The higher postoperative rate of patella baja in the septic group became statistically significant even when preoperative incidence was considered. This may have been caused by infection-related scarring and by prolonged immobilization of septic knees with use of nonarticulating antibiotic spacers. By keeping these knees immobile with a nonarticulating spacer for a prolonged period in the healing phase of the infection, scar tissue may mature and form over the time between stages. A comparable example may be high tibial osteotomies, in which a high incidence of patella baja has been partly attributed to prolonged casting.11 Future work comparing the results of articulating and nonarticulating spacers will help to determine if immobilization contributes to patella baja in infected TKAs.
There are several limitations to our study. Patient outcome questionnaires were not used, and they would have allowed for the assessment of physical outcomes and emotional satisfaction by comparing outcomes between patients with and without patella baja and comparing septic and aseptic TKAs. In addition, there was no standard method for quantifying difficulty of revision, which would have enabled us to compare difficulty of revision in patients with patella baja.
Conclusion
This study identified a high rate of patella baja and decreased ROM in TKA revisions, particularly infected revisions treated with a nonarticulating spacer. It is important to determine if there are functional consequences. Further investigation is needed regarding the cause, prevention, and management of this potentially debilitating outcome after revision TKA.
1. Aglietti P, Buzzi R, Gaudenzi A. Patello-femoral functional results and complications with the posterior stabilised total condylar knee prosthesis. J Arthroplasty. 1988;3(1):17-25.
2. Fern ED, Winson IG, Getty CJM. Anterior knee pain in rheumatoid patients after total knee replacement: possible selection criteria for patellar resurfacing. J Bone Joint Surg Br. 1992;74(5):745-748.
3. Figgie HE 3rd, Goldberg VM, Heiple KG, Moller HS 3rd, Gordon NH. The influence of tibial-patellofemoral location on function of the knee in patients with the posterior stabilized condylar knee prosthesis. J Bone Surg Surg Am. 1986;68(7):1035-1040.
4. Floren M, Davis J, Peterson MG, Laskin RS. A mini-midvastus capsular approach with patellar displacement decreases the prevalence of patellar baja. J Arthroplasty. 2007;22(6 Suppl 2):51-57.
5. Meneghini RM, Ritter MA, Pierson JL, Meding JB, Berend ME, Faris PM. The effect of the Insall-Salvati ratio on outcome after total knee arthroplasty. J Arthroplasty. 2006;21(6 Suppl 2):116-120.
6. Singerman R, Davy DT, Goldberg VM. Effects of patella alta and patella infera on patellofemoral contact forces. J Biomech. 1994;27(8):1059-1065.
7. Van Eijden TM, Kouwenhoven E, Weijs WA. Mechanics of the patellar articulation: effects of patellar ligament length studied with a mathematical model. Acta Orthop Scand. 1987;58(5):560-566.
8. Weale AE, Murray DW, Newman JH, Ackroyd CE. The length of the patellar tendon after unicompartmental and total knee replacement. J Bone Joint Surg Br. 1999;81(5):790-795.
9. Chonko DJ, Lombardi AV Jr, Berend KR. Patella baja and total knee arthroplasty (TKA): etiology, diagnosis, and management. Surg Technol Int. 2004;12:231-238.
10. Cameron HU, Jung YB. Patella baja complicating total knee arthroplasty. A report of two cases. J Arthroplasty. 1988;3(2):177-180.
11. Scuderi GR, Windsor RE, Insall JN. Observations on patellar height after proximal tibial osteotomy. J Bone Joint Surg Am. 1989;71(2):245-248.
12. Insall JN, Salvati E. Patella position in the normal knee joint. Radiology. 1971;101(1):101-104.
13. Grelsamer RP. Patella baja after total knee arthroplasty: is it really patella baja? J Arthroplasty. 2002;17(1):66-69.
14. Parvizi J, Zmistowski B, Berbari EF, et al. New definition for periprosthetic joint infection: from the Workgroup of the Musculoskeletal Infection Society. Clin Orthop. 2011;469(11):2992-2994.
15. Workgroup Convened by the Musculoskeletal Infection Society. New definition for periprosthetic joint infection. J Arthroplasty. 2011;26(8):1136-1138.
16. Laudermilch DJ, Fedorka CJ, Heyl A, Rao N, McGough RL. Outcomes of revision total knee arthroplasty after methicillin-resistant Staphylococcus aureus infection. Clin Orthop. 2010;468(8):2067-2073.
17. Bozic KJ, Kurtz SM, Lau E, et al. The epidemiology of revision total knee arthroplasty in the United States. Clin Orthop. 2010;468(1):45-51.
Patellar height may be important in determining function after total knee arthroplasty (TKA). By altering patellofemoral joint mechanics, patella baja may cause several functional issues after TKA.1-8 Patella baja leads to decreased range of motion (ROM) affecting both extension and flexion.5,8,9 Deep flexion can be restricted in TKA patients with patella baja because of tracking limitations associated with an inferiorly displaced patella. As the knee is brought into flexion, the patella can impinge on the anterior aspect of the tibial polyethylene or the tibial tray—presenting a true block to flexion and potentially altering wear.1,10
Another functional issue with patella baja is loss of strength in the extensor mechanism. The patella serves as a fulcrum for the extensor muscles of the knee. When positioned properly and functioning properly, the patella increases the extensor forces generated. When the patella is positioned in baja, the knee generates decreased extensor mechanism force.6,7 This can result in a lag, with the patient being unable to fully extend the knee. Extension-dependent activities are impaired. Patients with weak extensor function can experience poor function with stair climbing, rising from a chair, and exiting an automobile. The improper function and scarring of the patella can result in increased anterior knee pain and worse functional outcome scores after TKAs.3,9
An abnormally positioned patella can either result from or lead to increased scarring in the knee.9,11 Patellar height is often measured with the Insall-Salvati ratio (ISR), which is the patella tendon length (measurement of the tendon from the tibial tubercle to the inferior pole of the patella) divided by the patellar length (longest measured dimension of the patella) (Figure 1).12 Patella baja is defined as an ISR of less than 0.8. Other indices that reference off the tibial plateau (Blackburne-Peel ratio, Canton-Deschamps ratio) reflect an elevation of the joint line, or pseudobaja, and are unreliable for analysis of patella baja after TKA.13
Postoperative patella baja has been reported in 10% to 34% of primary TKAs.4,7 Inferior positioning of the patella and scarring can cause intraoperative difficulty with exposure and may complicate outcomes.9,13 The exposure scar is often larger in TKA revisions for infection compared with primary TKAs.
We conducted a study to compare the incidence of patella baja in noninfected and infected TKA revisions. We hypothesized that, compared with noninfected knees, infected knees treated with nonarticulating spacers would have a higher incidence of patella baja both before and after surgery secondary to more inflammation, immobilization, and related scarring.
Materials and Methods
We conducted a retrospective case–cohort study of 148 consecutive TKA revisions. All TKA revisions were performed between 2003 and 2009 using a mobile-bearing revision system from a single manufacturer. All surgeries were done at a single institution by the 2 senior surgeons. The surgical approach was a standard medial parapatellar approach without patellar eversion. Our institutional review board approved the study and waived the requirement for informed consent, as this was a retrospective study of existing medical records that posed no more than minimal risk to patients.
To properly evaluate patellar height, orthopedic specialty–trained radiologic technicians obtained preoperative and postoperative weight-bearing radiographs using a standardized lateral radiograph in clinic. Two blinded investigators measured ISR radiographically both before surgery (preexplant for septic revisions) and at latest follow-up (postreplant for septic revisions). Patients with inadequate films and/or patellectomies were excluded, along with patients who had less than 6 months of postoperative follow-up.
Ninety-one patients (101 TKAs) met the study inclusion criteria. Two groups of cases were compared: aseptic revisions (n = 67) and septic revisions (n = 34). Reasons for aseptic revisions included implant loosening (24/67, 35.8%), instability (12/67, 17.9%), pain (12/67, 17.9%), lysis (5/67, 7.5%), stiffness (3/67, 4.5%), and malrotation (2/67, 3.0%). Infection was determined by Musculoskeletal Infection Society criteria, as documented by positive aspirations and/or intraoperative tissue cultures taken at prosthesis explantation, elevated white blood cell count in the aspirate, elevated percentage of polymorphonuclear (PMN) cells in the aspirate, gross purulence, presence of chronic draining sinus, or histologic analysis revealing acute inflammation with more than 5 PMN cells per high power field.14,15
All infected TKAs were treated with 2-stage revisions. The standard of care at our institution through this series was to use a nonarticulating spacer for the treatment of infection. Weight-bearing status varied by extent of bone damage. Six weeks of culture-specific intravenous antibiotics were administered with assistance from an infectious disease consultant. Reimplantation was performed when clinical and laboratory criteria for resolution of infection were met—specifically, when erythrocyte sedimentation rate was less than 30 mm/h, C-reactive protein level was less than 10 mg/L, and aspirates were culture-negative. Mean (range) follow-up was 33.9 (6.2-75.7) months for aseptic revisions and 32.3 (7.5-94.2) months for septic revisions. Radiographic follow-up was performed at each visit, with weight-bearing anteroposterior and posteroanterior views, along with a lateral knee radiograph. At final follow-up, ROM was recorded by the senior attending evaluating the patient.
Categorical variables were statistically analyzed with χ2 tests, and continuous variables were analyzed with Student t test, analysis of variance, and univariate analysis of covariance (ANCOVA). Statistical significance was set at P < .05. Intrarater reliability was measured with the intraclass correlation coefficient (ICC). All statistical analysis was performed with Predictive Analytics SoftWare Statistics Version 20.0 (SPSS, Chicago, Illinois).
Results
Ninety-one consecutive patients (43 men, 48 women) were included in this study. Mean (SD) age was 66.4 (10.1) years. Mean (SD) preoperative ISR in septic and aseptic cases was 0.94 (0.25) for men and 1.02 (0.23) for women (P = .10). Mean postoperative ISR in septic and aseptic cases was 0.84 (0.27) for men and 0.99 (0.23) for women (P = .004). There was a sex difference between septic and aseptic revisions. There were 22 men and 36 women in the aseptic group and 21 men and 12 women in the septic group (P = .01). Men were more likely than women to have septic revisions and patella baja. Table 1 compares the patient demographics of the 2 patient populations. Mean (SD) number of surgeries, including irrigation and débridement procedures before reimplantation, was larger for septic revisions, 2.9 (0.9), than for aseptic revisions, 1.4 (0.8) (P < .001).
Infection was the most common reason for revision and accounted for 33.7% (34/101) of all revisions. Noninfectious indications, in declining order of frequency, included loosening (23.8%, 24/101), instability (11.9%, 12/101), pain (11.9%, 12/101), osteolysis (5.0%, 5/101), polyethylene wear (5.0%, 5/101), failed unicompartmental knee (4.0%, 4/101), stiffness (3.0%, 3/101), and patellar problems (2.0%, 2/101) (Table 2). ISR decreased significantly only in infected revisions. It is important to note that there was not a high incidence of stiffness or patellofemoral failure in revision patients before surgery.
Mean (SD) ISR did not differ between groups before surgery, 1.00 (0.25) for aseptic and 0.96 (0.22) for septic (P = .49), but differed significantly after surgery, 0.99 (0.23) for aseptic and 0.77 (0.24) for septic (P < .001) (Figure 2). The univariate ANCOVA also demonstrated a postoperative difference between groups when taking the preoperative ratio into account: 0.99 (0.23) for aseptic and 0.78 (0.24) for septic (P = .005) (Table 3). Before surgery, 22.4% and 23.9% of the aseptic and septic groups, respectively, had patella baja (P = .58). After surgery, 17.6% and 58.8% of the aseptic and septic groups had patella baja (P = .001) (Table 4). The ICC for preoperative ISR was 0.94, and the ICC for postoperative ISR was 0.96, which indicates excellent agreement of measurements between the 2 blinded investigators.
ROM differed between septic and aseptic groups owing to the difference in postoperative flexion. Mean (SD) postoperative extension was 2.2° (5.4°) for the aseptic group and 5.1° (9.8°) for the septic group—not significantly different (P = .13). Mean (SD) postoperative flexion was 110.2° (18.8°) for the aseptic group and 97.2° (29.4°) for the septic group—significantly different (P = .02). The groups differed significantly (P = .02) in mean (SD) ROM: 108.0° (20.7°) for aseptic and 92.2° (34.6°) for septic (Table 1). ROM was also significantly associated with patella baja (P = .04), as patients with ISR of less than 0.8 had mean (SD) postoperative ROM of 95.1° (31.6°), and patients without patella baja had mean (SD) postoperative ROM of 106.8° (23.6°).
For the septic group, mean (SD) time between first and second stages was 13.0 (8.3) weeks (range, 1-44.3 weeks). Mean (SD) timing of spacer placement was not statistically significantly different (P = .90) between patients who had patella baja, 12.9 (8.8) weeks, and patients who did not have patella baja, 13.2 (7.8) weeks.
Discussion
This study demonstrated that TKAs done for septic reasons resulted in a higher incidence of patella baja and decreased ROM. Incidence of patella baja was higher both before and after revision in septic TKAs than in aseptic TKAs, proving the hypothesis under study. Prerevision incidence was not significantly different, but there was a trend that could not be ignored. This may suggest that there is already an ongoing process in the infected knee that contributes to patella baja; the precise etiology remains unclear and is likely multifactorial. For example, scar formation may be increased in patients with chronic infection, predisposing to patella baja. This assertion is indirectly supported by a recent study from our institution revealing longer average surgical time in septic versus aseptic knee revisions; the difference was thought to reflect increased scar-tissue formation.16 That study also found that patients who underwent septic revisions had significantly more surgical procedures than patients who underwent aseptic revisions. Repetitive surgeries—specifically, repetitive arthrotomies during irrigation and débridement before reimplantation—lead to increased scar formation, which may contribute to preoperative and postoperative patella baja. This may be reflected in the findings that ROM was decreased in patients in the septic group versus patients in the aseptic group and that ROM was decreased in patients with patella baja. In addition, our study found that male patients were more likely to undergo TKA revision for septic reasons and to develop postoperative patella baja. This finding contrasts with that of a study5 that compared preoperative and postoperative ISR in primary TKA and found that women were more likely than men to have patella baja. Although women are more likely to undergo TKA revision,17 men may be more susceptible to infection and subsequent patella baja.
The higher postoperative rate of patella baja in the septic group became statistically significant even when preoperative incidence was considered. This may have been caused by infection-related scarring and by prolonged immobilization of septic knees with use of nonarticulating antibiotic spacers. By keeping these knees immobile with a nonarticulating spacer for a prolonged period in the healing phase of the infection, scar tissue may mature and form over the time between stages. A comparable example may be high tibial osteotomies, in which a high incidence of patella baja has been partly attributed to prolonged casting.11 Future work comparing the results of articulating and nonarticulating spacers will help to determine if immobilization contributes to patella baja in infected TKAs.
There are several limitations to our study. Patient outcome questionnaires were not used, and they would have allowed for the assessment of physical outcomes and emotional satisfaction by comparing outcomes between patients with and without patella baja and comparing septic and aseptic TKAs. In addition, there was no standard method for quantifying difficulty of revision, which would have enabled us to compare difficulty of revision in patients with patella baja.
Conclusion
This study identified a high rate of patella baja and decreased ROM in TKA revisions, particularly infected revisions treated with a nonarticulating spacer. It is important to determine if there are functional consequences. Further investigation is needed regarding the cause, prevention, and management of this potentially debilitating outcome after revision TKA.
Patellar height may be important in determining function after total knee arthroplasty (TKA). By altering patellofemoral joint mechanics, patella baja may cause several functional issues after TKA.1-8 Patella baja leads to decreased range of motion (ROM) affecting both extension and flexion.5,8,9 Deep flexion can be restricted in TKA patients with patella baja because of tracking limitations associated with an inferiorly displaced patella. As the knee is brought into flexion, the patella can impinge on the anterior aspect of the tibial polyethylene or the tibial tray—presenting a true block to flexion and potentially altering wear.1,10
Another functional issue with patella baja is loss of strength in the extensor mechanism. The patella serves as a fulcrum for the extensor muscles of the knee. When positioned properly and functioning properly, the patella increases the extensor forces generated. When the patella is positioned in baja, the knee generates decreased extensor mechanism force.6,7 This can result in a lag, with the patient being unable to fully extend the knee. Extension-dependent activities are impaired. Patients with weak extensor function can experience poor function with stair climbing, rising from a chair, and exiting an automobile. The improper function and scarring of the patella can result in increased anterior knee pain and worse functional outcome scores after TKAs.3,9
An abnormally positioned patella can either result from or lead to increased scarring in the knee.9,11 Patellar height is often measured with the Insall-Salvati ratio (ISR), which is the patella tendon length (measurement of the tendon from the tibial tubercle to the inferior pole of the patella) divided by the patellar length (longest measured dimension of the patella) (Figure 1).12 Patella baja is defined as an ISR of less than 0.8. Other indices that reference off the tibial plateau (Blackburne-Peel ratio, Canton-Deschamps ratio) reflect an elevation of the joint line, or pseudobaja, and are unreliable for analysis of patella baja after TKA.13
Postoperative patella baja has been reported in 10% to 34% of primary TKAs.4,7 Inferior positioning of the patella and scarring can cause intraoperative difficulty with exposure and may complicate outcomes.9,13 The exposure scar is often larger in TKA revisions for infection compared with primary TKAs.
We conducted a study to compare the incidence of patella baja in noninfected and infected TKA revisions. We hypothesized that, compared with noninfected knees, infected knees treated with nonarticulating spacers would have a higher incidence of patella baja both before and after surgery secondary to more inflammation, immobilization, and related scarring.
Materials and Methods
We conducted a retrospective case–cohort study of 148 consecutive TKA revisions. All TKA revisions were performed between 2003 and 2009 using a mobile-bearing revision system from a single manufacturer. All surgeries were done at a single institution by the 2 senior surgeons. The surgical approach was a standard medial parapatellar approach without patellar eversion. Our institutional review board approved the study and waived the requirement for informed consent, as this was a retrospective study of existing medical records that posed no more than minimal risk to patients.
To properly evaluate patellar height, orthopedic specialty–trained radiologic technicians obtained preoperative and postoperative weight-bearing radiographs using a standardized lateral radiograph in clinic. Two blinded investigators measured ISR radiographically both before surgery (preexplant for septic revisions) and at latest follow-up (postreplant for septic revisions). Patients with inadequate films and/or patellectomies were excluded, along with patients who had less than 6 months of postoperative follow-up.
Ninety-one patients (101 TKAs) met the study inclusion criteria. Two groups of cases were compared: aseptic revisions (n = 67) and septic revisions (n = 34). Reasons for aseptic revisions included implant loosening (24/67, 35.8%), instability (12/67, 17.9%), pain (12/67, 17.9%), lysis (5/67, 7.5%), stiffness (3/67, 4.5%), and malrotation (2/67, 3.0%). Infection was determined by Musculoskeletal Infection Society criteria, as documented by positive aspirations and/or intraoperative tissue cultures taken at prosthesis explantation, elevated white blood cell count in the aspirate, elevated percentage of polymorphonuclear (PMN) cells in the aspirate, gross purulence, presence of chronic draining sinus, or histologic analysis revealing acute inflammation with more than 5 PMN cells per high power field.14,15
All infected TKAs were treated with 2-stage revisions. The standard of care at our institution through this series was to use a nonarticulating spacer for the treatment of infection. Weight-bearing status varied by extent of bone damage. Six weeks of culture-specific intravenous antibiotics were administered with assistance from an infectious disease consultant. Reimplantation was performed when clinical and laboratory criteria for resolution of infection were met—specifically, when erythrocyte sedimentation rate was less than 30 mm/h, C-reactive protein level was less than 10 mg/L, and aspirates were culture-negative. Mean (range) follow-up was 33.9 (6.2-75.7) months for aseptic revisions and 32.3 (7.5-94.2) months for septic revisions. Radiographic follow-up was performed at each visit, with weight-bearing anteroposterior and posteroanterior views, along with a lateral knee radiograph. At final follow-up, ROM was recorded by the senior attending evaluating the patient.
Categorical variables were statistically analyzed with χ2 tests, and continuous variables were analyzed with Student t test, analysis of variance, and univariate analysis of covariance (ANCOVA). Statistical significance was set at P < .05. Intrarater reliability was measured with the intraclass correlation coefficient (ICC). All statistical analysis was performed with Predictive Analytics SoftWare Statistics Version 20.0 (SPSS, Chicago, Illinois).
Results
Ninety-one consecutive patients (43 men, 48 women) were included in this study. Mean (SD) age was 66.4 (10.1) years. Mean (SD) preoperative ISR in septic and aseptic cases was 0.94 (0.25) for men and 1.02 (0.23) for women (P = .10). Mean postoperative ISR in septic and aseptic cases was 0.84 (0.27) for men and 0.99 (0.23) for women (P = .004). There was a sex difference between septic and aseptic revisions. There were 22 men and 36 women in the aseptic group and 21 men and 12 women in the septic group (P = .01). Men were more likely than women to have septic revisions and patella baja. Table 1 compares the patient demographics of the 2 patient populations. Mean (SD) number of surgeries, including irrigation and débridement procedures before reimplantation, was larger for septic revisions, 2.9 (0.9), than for aseptic revisions, 1.4 (0.8) (P < .001).
Infection was the most common reason for revision and accounted for 33.7% (34/101) of all revisions. Noninfectious indications, in declining order of frequency, included loosening (23.8%, 24/101), instability (11.9%, 12/101), pain (11.9%, 12/101), osteolysis (5.0%, 5/101), polyethylene wear (5.0%, 5/101), failed unicompartmental knee (4.0%, 4/101), stiffness (3.0%, 3/101), and patellar problems (2.0%, 2/101) (Table 2). ISR decreased significantly only in infected revisions. It is important to note that there was not a high incidence of stiffness or patellofemoral failure in revision patients before surgery.
Mean (SD) ISR did not differ between groups before surgery, 1.00 (0.25) for aseptic and 0.96 (0.22) for septic (P = .49), but differed significantly after surgery, 0.99 (0.23) for aseptic and 0.77 (0.24) for septic (P < .001) (Figure 2). The univariate ANCOVA also demonstrated a postoperative difference between groups when taking the preoperative ratio into account: 0.99 (0.23) for aseptic and 0.78 (0.24) for septic (P = .005) (Table 3). Before surgery, 22.4% and 23.9% of the aseptic and septic groups, respectively, had patella baja (P = .58). After surgery, 17.6% and 58.8% of the aseptic and septic groups had patella baja (P = .001) (Table 4). The ICC for preoperative ISR was 0.94, and the ICC for postoperative ISR was 0.96, which indicates excellent agreement of measurements between the 2 blinded investigators.
ROM differed between septic and aseptic groups owing to the difference in postoperative flexion. Mean (SD) postoperative extension was 2.2° (5.4°) for the aseptic group and 5.1° (9.8°) for the septic group—not significantly different (P = .13). Mean (SD) postoperative flexion was 110.2° (18.8°) for the aseptic group and 97.2° (29.4°) for the septic group—significantly different (P = .02). The groups differed significantly (P = .02) in mean (SD) ROM: 108.0° (20.7°) for aseptic and 92.2° (34.6°) for septic (Table 1). ROM was also significantly associated with patella baja (P = .04), as patients with ISR of less than 0.8 had mean (SD) postoperative ROM of 95.1° (31.6°), and patients without patella baja had mean (SD) postoperative ROM of 106.8° (23.6°).
For the septic group, mean (SD) time between first and second stages was 13.0 (8.3) weeks (range, 1-44.3 weeks). Mean (SD) timing of spacer placement was not statistically significantly different (P = .90) between patients who had patella baja, 12.9 (8.8) weeks, and patients who did not have patella baja, 13.2 (7.8) weeks.
Discussion
This study demonstrated that TKAs done for septic reasons resulted in a higher incidence of patella baja and decreased ROM. Incidence of patella baja was higher both before and after revision in septic TKAs than in aseptic TKAs, proving the hypothesis under study. Prerevision incidence was not significantly different, but there was a trend that could not be ignored. This may suggest that there is already an ongoing process in the infected knee that contributes to patella baja; the precise etiology remains unclear and is likely multifactorial. For example, scar formation may be increased in patients with chronic infection, predisposing to patella baja. This assertion is indirectly supported by a recent study from our institution revealing longer average surgical time in septic versus aseptic knee revisions; the difference was thought to reflect increased scar-tissue formation.16 That study also found that patients who underwent septic revisions had significantly more surgical procedures than patients who underwent aseptic revisions. Repetitive surgeries—specifically, repetitive arthrotomies during irrigation and débridement before reimplantation—lead to increased scar formation, which may contribute to preoperative and postoperative patella baja. This may be reflected in the findings that ROM was decreased in patients in the septic group versus patients in the aseptic group and that ROM was decreased in patients with patella baja. In addition, our study found that male patients were more likely to undergo TKA revision for septic reasons and to develop postoperative patella baja. This finding contrasts with that of a study5 that compared preoperative and postoperative ISR in primary TKA and found that women were more likely than men to have patella baja. Although women are more likely to undergo TKA revision,17 men may be more susceptible to infection and subsequent patella baja.
The higher postoperative rate of patella baja in the septic group became statistically significant even when preoperative incidence was considered. This may have been caused by infection-related scarring and by prolonged immobilization of septic knees with use of nonarticulating antibiotic spacers. By keeping these knees immobile with a nonarticulating spacer for a prolonged period in the healing phase of the infection, scar tissue may mature and form over the time between stages. A comparable example may be high tibial osteotomies, in which a high incidence of patella baja has been partly attributed to prolonged casting.11 Future work comparing the results of articulating and nonarticulating spacers will help to determine if immobilization contributes to patella baja in infected TKAs.
There are several limitations to our study. Patient outcome questionnaires were not used, and they would have allowed for the assessment of physical outcomes and emotional satisfaction by comparing outcomes between patients with and without patella baja and comparing septic and aseptic TKAs. In addition, there was no standard method for quantifying difficulty of revision, which would have enabled us to compare difficulty of revision in patients with patella baja.
Conclusion
This study identified a high rate of patella baja and decreased ROM in TKA revisions, particularly infected revisions treated with a nonarticulating spacer. It is important to determine if there are functional consequences. Further investigation is needed regarding the cause, prevention, and management of this potentially debilitating outcome after revision TKA.
1. Aglietti P, Buzzi R, Gaudenzi A. Patello-femoral functional results and complications with the posterior stabilised total condylar knee prosthesis. J Arthroplasty. 1988;3(1):17-25.
2. Fern ED, Winson IG, Getty CJM. Anterior knee pain in rheumatoid patients after total knee replacement: possible selection criteria for patellar resurfacing. J Bone Joint Surg Br. 1992;74(5):745-748.
3. Figgie HE 3rd, Goldberg VM, Heiple KG, Moller HS 3rd, Gordon NH. The influence of tibial-patellofemoral location on function of the knee in patients with the posterior stabilized condylar knee prosthesis. J Bone Surg Surg Am. 1986;68(7):1035-1040.
4. Floren M, Davis J, Peterson MG, Laskin RS. A mini-midvastus capsular approach with patellar displacement decreases the prevalence of patellar baja. J Arthroplasty. 2007;22(6 Suppl 2):51-57.
5. Meneghini RM, Ritter MA, Pierson JL, Meding JB, Berend ME, Faris PM. The effect of the Insall-Salvati ratio on outcome after total knee arthroplasty. J Arthroplasty. 2006;21(6 Suppl 2):116-120.
6. Singerman R, Davy DT, Goldberg VM. Effects of patella alta and patella infera on patellofemoral contact forces. J Biomech. 1994;27(8):1059-1065.
7. Van Eijden TM, Kouwenhoven E, Weijs WA. Mechanics of the patellar articulation: effects of patellar ligament length studied with a mathematical model. Acta Orthop Scand. 1987;58(5):560-566.
8. Weale AE, Murray DW, Newman JH, Ackroyd CE. The length of the patellar tendon after unicompartmental and total knee replacement. J Bone Joint Surg Br. 1999;81(5):790-795.
9. Chonko DJ, Lombardi AV Jr, Berend KR. Patella baja and total knee arthroplasty (TKA): etiology, diagnosis, and management. Surg Technol Int. 2004;12:231-238.
10. Cameron HU, Jung YB. Patella baja complicating total knee arthroplasty. A report of two cases. J Arthroplasty. 1988;3(2):177-180.
11. Scuderi GR, Windsor RE, Insall JN. Observations on patellar height after proximal tibial osteotomy. J Bone Joint Surg Am. 1989;71(2):245-248.
12. Insall JN, Salvati E. Patella position in the normal knee joint. Radiology. 1971;101(1):101-104.
13. Grelsamer RP. Patella baja after total knee arthroplasty: is it really patella baja? J Arthroplasty. 2002;17(1):66-69.
14. Parvizi J, Zmistowski B, Berbari EF, et al. New definition for periprosthetic joint infection: from the Workgroup of the Musculoskeletal Infection Society. Clin Orthop. 2011;469(11):2992-2994.
15. Workgroup Convened by the Musculoskeletal Infection Society. New definition for periprosthetic joint infection. J Arthroplasty. 2011;26(8):1136-1138.
16. Laudermilch DJ, Fedorka CJ, Heyl A, Rao N, McGough RL. Outcomes of revision total knee arthroplasty after methicillin-resistant Staphylococcus aureus infection. Clin Orthop. 2010;468(8):2067-2073.
17. Bozic KJ, Kurtz SM, Lau E, et al. The epidemiology of revision total knee arthroplasty in the United States. Clin Orthop. 2010;468(1):45-51.
1. Aglietti P, Buzzi R, Gaudenzi A. Patello-femoral functional results and complications with the posterior stabilised total condylar knee prosthesis. J Arthroplasty. 1988;3(1):17-25.
2. Fern ED, Winson IG, Getty CJM. Anterior knee pain in rheumatoid patients after total knee replacement: possible selection criteria for patellar resurfacing. J Bone Joint Surg Br. 1992;74(5):745-748.
3. Figgie HE 3rd, Goldberg VM, Heiple KG, Moller HS 3rd, Gordon NH. The influence of tibial-patellofemoral location on function of the knee in patients with the posterior stabilized condylar knee prosthesis. J Bone Surg Surg Am. 1986;68(7):1035-1040.
4. Floren M, Davis J, Peterson MG, Laskin RS. A mini-midvastus capsular approach with patellar displacement decreases the prevalence of patellar baja. J Arthroplasty. 2007;22(6 Suppl 2):51-57.
5. Meneghini RM, Ritter MA, Pierson JL, Meding JB, Berend ME, Faris PM. The effect of the Insall-Salvati ratio on outcome after total knee arthroplasty. J Arthroplasty. 2006;21(6 Suppl 2):116-120.
6. Singerman R, Davy DT, Goldberg VM. Effects of patella alta and patella infera on patellofemoral contact forces. J Biomech. 1994;27(8):1059-1065.
7. Van Eijden TM, Kouwenhoven E, Weijs WA. Mechanics of the patellar articulation: effects of patellar ligament length studied with a mathematical model. Acta Orthop Scand. 1987;58(5):560-566.
8. Weale AE, Murray DW, Newman JH, Ackroyd CE. The length of the patellar tendon after unicompartmental and total knee replacement. J Bone Joint Surg Br. 1999;81(5):790-795.
9. Chonko DJ, Lombardi AV Jr, Berend KR. Patella baja and total knee arthroplasty (TKA): etiology, diagnosis, and management. Surg Technol Int. 2004;12:231-238.
10. Cameron HU, Jung YB. Patella baja complicating total knee arthroplasty. A report of two cases. J Arthroplasty. 1988;3(2):177-180.
11. Scuderi GR, Windsor RE, Insall JN. Observations on patellar height after proximal tibial osteotomy. J Bone Joint Surg Am. 1989;71(2):245-248.
12. Insall JN, Salvati E. Patella position in the normal knee joint. Radiology. 1971;101(1):101-104.
13. Grelsamer RP. Patella baja after total knee arthroplasty: is it really patella baja? J Arthroplasty. 2002;17(1):66-69.
14. Parvizi J, Zmistowski B, Berbari EF, et al. New definition for periprosthetic joint infection: from the Workgroup of the Musculoskeletal Infection Society. Clin Orthop. 2011;469(11):2992-2994.
15. Workgroup Convened by the Musculoskeletal Infection Society. New definition for periprosthetic joint infection. J Arthroplasty. 2011;26(8):1136-1138.
16. Laudermilch DJ, Fedorka CJ, Heyl A, Rao N, McGough RL. Outcomes of revision total knee arthroplasty after methicillin-resistant Staphylococcus aureus infection. Clin Orthop. 2010;468(8):2067-2073.
17. Bozic KJ, Kurtz SM, Lau E, et al. The epidemiology of revision total knee arthroplasty in the United States. Clin Orthop. 2010;468(1):45-51.
Concurrent Treatment of a Middle-Third Clavicle Fracture and Type IV Acromioclavicular Dislocation
Acromioclavicular (AC) dislocations and displaced fractures of the middle third of the clavicle rarely occur together. Isolated AC joint separation is often treated nonoperatively with internal coracoclavicular (CC) fixation or reconstruction considered for type IV-VI AC dislocations and some type III injuries.1 Isolated clavicle fractures traditionally have been treated nonoperatively. The current trend is toward internal fixation for displaced and shortened fractures.2 There have been only a handful of reports of concomitant AC dislocation and midshaft clavicle fracture.3-6 Previous treatments have included nonoperative treatment, AC fixation, or internal fixation of the clavicle with ligamentous reconstruction.
We present a previously undescribed technique for internal fixation of this rare shoulder injury. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
While driving an all-terrain vehicle, a healthy 19-year-old right-hand–dominant man hit a bridge and sustained direct impact to his right shoulder. He presented to the emergency department complaining of right shoulder pain and deformity without skin disruption, vascular insufficiency, or neurologic symptoms. Anteroposterior (AP) radiograph showed an oblique, displaced, middle-third clavicle shaft fracture (Figure 1). An associated type IV AC dislocation was confirmed on axillary radiograph (Figure 2) and on an axial cut from a trauma chest computed tomography (CT) scan (Figure 3). The patient was discharged home from the trauma service the next day with a sling for comfort and plans for delayed, elective operative fixation 1 week later.
The patient was placed in a beach-chair position. Through a longitudinal incision extending laterally over the AC joint, the clavicle was exposed for fracture reduction, with care taken to retain soft-tissue attachments. The distal clavicle was buttonholed posteriorly through the trapezius muscle and fascia. The distal fracture fragment was devoid of any remaining CC ligamentous attachment. After satisfactory reduction, a low-profile precontoured clavicle plate (Superior Midshaft Clavicle Plate; Acumed, Hillsboro, Oregon) was placed superiorly; the fracture was compressed through the plate and internally fixed with three 3.5-mm bicortical screws on both sides of the fracture. Approximately 5 mm of the distal clavicle was resected at the AC joint to facilitate adequate AC and CC reduction without disruption of the clavicle fracture. With an adequate CC reduction, a 3.5-mm fully threaded cortical screw was placed through the most distal hole in the clavicle plate, clavicle, and coracoid.
After surgery, the patient was placed into an ARC shoulder immobilizer (Bledsoe, Grand Prairie, Texas) for 6 weeks, removing the immobilizer only for elbow and wrist range of motion (ROM) exercises. Radiographs at 3-month follow-up (Figure 4) showed a healed fracture with no loss of AC or CC reduction. Three months after surgery, another procedure was performed to remove the CC screw. One year after the initial surgery, the patient complained of intermittent soreness over the lateral shoulder but was not limited in his activities and was back to performing manual labor without difficulty. He had full ROM in forward flexion, abduction, internal rotation, and external rotation without weakness, tenderness, or any neurovascular deficit. After CC screw removal, no deformity returned at the shoulder. Radiographs showed a healed fracture with minimal superior subluxation at the AC joint without significant change from the 3-month follow-up (Figure 5).
Discussion
The combined injury pattern of a type IV AC dislocation and a displaced middle-third clavicle shaft fracture is rare. The usual mechanism of injury, as seen in the present case, is a direct blow to the shoulder at the tip of the acromion, though indirect forces from a fall on an outstretched hand are also described.7 Disruption of the CC ligaments with AC separation likely dissipates the stress necessary to create a clavicle fracture in most cases,1 explaining the rarity of this injury. It is imperative to evaluate patients for injury to both the osseous and ligamentous structures.
Previous case reports of concomitant AC separation and midshaft clavicle fracture have described a variety of treatment options, but to date our case represents the only episode in which both the clavicle fracture and the AC joint were treated with open reduction and internal fixation (ORIF). Wurtz and colleagues5 reported on a series of 4 patients with AC disruption and middle-third clavicle fracture. Three of the 4 patients had type IV AC separation; all 3 were treated, 2 acutely and 1 chronically, with open reduction of the AC and CC joints; 2 of these patients had CC screw fixation only after reduction, and the third had 2 Steinmann pins placed across the AC joint without CC screw fixation. All hardware was removed after 12 weeks. The fourth patient had a type II AC dislocation and was treated with closed reduction of the clavicle with no intervention for the AC joint. None of the clavicle fractures in this series were treated with internal fixation. All patients had full and pain-free ROM at 1- to 3-year follow-up.
Juhn and Simonian3 reported on a case of type VI separation with greenstick midshaft clavicle fracture in a hockey player seen 7 days after injury. The patient described some tingling in the upper extremity and had shoulder pain on initial presentation but was noted to have minimal displacement of both the AC joint and the midshaft clavicle fracture. Both injuries were treated nonsurgically with good outcome, and the patient returned to full activity (including hockey) within 14 weeks after injury.
Lancourt4 described the case of a patient with a type V AC dislocation and a displaced midshaft clavicle fracture. The AC joint was treated with Steinmann pin fixation, and the clavicle fracture was treated nonoperatively. The author cited high complication rates of plate fixation for clavicle fractures as the reason for not performing the additional procedure. The pins were removed 8 weeks after surgery. At 3-year follow-up, the patient had good radiographic and clinical outcome.
Yeh and colleagues6 described a patient who sustained a displaced midshaft clavicle fracture and a type IV AC dislocation in a fall from a horse. The patient underwent ORIF of the clavicle fracture with plate fixation. After the procedure, the AC joint was still unstable intraoperatively, and the AC and CC ligaments were reconstructed with semitendinosus allograft. The patient had full and painless ROM at 1-year follow-up.
The present case report serves as a reminder to obtain adequate shoulder radiographs when evaluating “just another clavicle fracture.” The radiographs should include a good axillary view to ensure there is not an associated AC dislocation. Increasingly, some authors have been advocating internal fixation for clavicle fractures, with reports of improved functional outcomes, improved cosmesis, and increased union rates.2 Indications for operative fixation include shortening and 100% displacement,8 and relative indications include open fractures.1 Operative fixation is perhaps more important for younger, athletic, and manual-labor populations. The trend in treatment of clavicle fractures toward operative fixation lends itself well to ORIF of the AC and CC joints; hence, a modern treatment for this rarely described combination injury should include internal plate fixation of the clavicle in addition to CC fixation. This additional procedure requires little extra time and energy in an operative scenario already requiring anesthesia, with easy insertion of the CC screw through the clavicle plate. Use of a CC screw obviates any potential risks associated with use of allograft tissue, and there is no anticipated difficulty with screw removal at 12 weeks.
Alternative options for AC stability include CC reconstruction with ligamentous allograft, ligamentous autograft, or suture/tightrope techniques. A noted advantage of these alternative techniques is less need to return to the operating room for the hardware removal that is recommended with CC screw fixation. However, these procedures potentially increase surgical exposure and operating time. In addition, screw fixation minimizes the possibility of donor-site morbidity from autograft transfers and potential complications from allograft tissue.
Hook plate fixation of the AC joint has also been described. In a recent case report of a similar injury pattern, plate fixation of the clavicle with simultaneous hook plate fixation of the AC joint was described.9 The patient did well but required removal of hardware of the hook plate and the clavicle plate 1 and 3 years after surgery, respectively. Although screw fixation is biomechanically stronger, debate persists about the clinical importance of this increase in strength.1 In the setting of plate fixation for the clavicle, these alternative AC fixations would require technique adjustments, including length of grafts and/or sutures, and raise concerns regarding interaction of the metal with the fixation material.
Critical evaluation of our technique revealed a lucency larger than the screw (Figure 5). However, the screw was not clinically loose at removal. This potential complication, in combination with the bent screw (Figure 4) before removal, highlights the concern for screw breakage with this technique, given the increased construct stiffness caused by the added plate.
Conclusion
As in the other reports mentioned, our patient had an excellent clinical and radiographic outcome. It could be inferred that, if fixation for isolated clavicle fractures demonstrates improved function, better outcomes would be seen for higher-energy fractures associated with AC dislocation. Given the current trend toward surgical fixation for certain clavicle fractures, we recommend that clavicle fractures associated with type IV AC dislocation be treated with ORIF of both injuries.
1. Ring D, Jupiter J. Injuries to the shoulder girdle. In: Browner, BD. Skeletal Trauma. Philadelphia, PA: Elsevier Health Sciences; 2008:1755-1778.
2. Altamimi SA, McKee MD; Canadian Orthopaedic Trauma Society. Nonoperative treatment compared with plate fixation of displaced midshaft clavicular fractures. Surgical technique. J Bone Joint Surg Am. 2008;90(suppl 2 pt 1):1-8.
3. Juhn MS, Simonian PT. Type VI acromioclavicular separation with middle-third clavicle fracture in an ice hockey player. Clin J Sports Med. 2002;12(5):315-317.
4. Lancourt JE. Acromioclavicular dislocation with adjacent clavicular fracture in a horseback rider. A case report. Am J Sports Med. 1990;18(3):321-322.
5. Wurtz LD, Lyons FA, Rockwood CA Jr. Fracture of the middle third of the clavicle and dislocation of the acromioclavicular joint. A report of four cases. J Bone Joint Surg Am. 1992;74(1):133-137.
6. Yeh PC, Miller SR, Cunningham JG, Sethi PM. Midshaft clavicle fracture and acromioclavicular dislocation: a case report of a rare injury. J Shoulder Elbow Surg. 2009;18(5):e1-e4.
7. Stanley D, Trowbridge EA, Norris SH. The mechanism of clavicular fracture. A clinical and biomechanical analysis. J Bone Joint Surg Br. 1988;70(3):461-464.
8. Kim W, McKee MD. Management of acute clavicle fractures. Orthop Clin North Am. 2008;39(4):491-505.
9. Woolf SK, Valentine BJ, Barfield WR, Hartsock LA. Middle-third clavicle fracture with associated type IV acromioclavicular separation: case report and literature review. J Surg Orthop Adv. 2013;22(2):183-186.
Acromioclavicular (AC) dislocations and displaced fractures of the middle third of the clavicle rarely occur together. Isolated AC joint separation is often treated nonoperatively with internal coracoclavicular (CC) fixation or reconstruction considered for type IV-VI AC dislocations and some type III injuries.1 Isolated clavicle fractures traditionally have been treated nonoperatively. The current trend is toward internal fixation for displaced and shortened fractures.2 There have been only a handful of reports of concomitant AC dislocation and midshaft clavicle fracture.3-6 Previous treatments have included nonoperative treatment, AC fixation, or internal fixation of the clavicle with ligamentous reconstruction.
We present a previously undescribed technique for internal fixation of this rare shoulder injury. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
While driving an all-terrain vehicle, a healthy 19-year-old right-hand–dominant man hit a bridge and sustained direct impact to his right shoulder. He presented to the emergency department complaining of right shoulder pain and deformity without skin disruption, vascular insufficiency, or neurologic symptoms. Anteroposterior (AP) radiograph showed an oblique, displaced, middle-third clavicle shaft fracture (Figure 1). An associated type IV AC dislocation was confirmed on axillary radiograph (Figure 2) and on an axial cut from a trauma chest computed tomography (CT) scan (Figure 3). The patient was discharged home from the trauma service the next day with a sling for comfort and plans for delayed, elective operative fixation 1 week later.
The patient was placed in a beach-chair position. Through a longitudinal incision extending laterally over the AC joint, the clavicle was exposed for fracture reduction, with care taken to retain soft-tissue attachments. The distal clavicle was buttonholed posteriorly through the trapezius muscle and fascia. The distal fracture fragment was devoid of any remaining CC ligamentous attachment. After satisfactory reduction, a low-profile precontoured clavicle plate (Superior Midshaft Clavicle Plate; Acumed, Hillsboro, Oregon) was placed superiorly; the fracture was compressed through the plate and internally fixed with three 3.5-mm bicortical screws on both sides of the fracture. Approximately 5 mm of the distal clavicle was resected at the AC joint to facilitate adequate AC and CC reduction without disruption of the clavicle fracture. With an adequate CC reduction, a 3.5-mm fully threaded cortical screw was placed through the most distal hole in the clavicle plate, clavicle, and coracoid.
After surgery, the patient was placed into an ARC shoulder immobilizer (Bledsoe, Grand Prairie, Texas) for 6 weeks, removing the immobilizer only for elbow and wrist range of motion (ROM) exercises. Radiographs at 3-month follow-up (Figure 4) showed a healed fracture with no loss of AC or CC reduction. Three months after surgery, another procedure was performed to remove the CC screw. One year after the initial surgery, the patient complained of intermittent soreness over the lateral shoulder but was not limited in his activities and was back to performing manual labor without difficulty. He had full ROM in forward flexion, abduction, internal rotation, and external rotation without weakness, tenderness, or any neurovascular deficit. After CC screw removal, no deformity returned at the shoulder. Radiographs showed a healed fracture with minimal superior subluxation at the AC joint without significant change from the 3-month follow-up (Figure 5).
Discussion
The combined injury pattern of a type IV AC dislocation and a displaced middle-third clavicle shaft fracture is rare. The usual mechanism of injury, as seen in the present case, is a direct blow to the shoulder at the tip of the acromion, though indirect forces from a fall on an outstretched hand are also described.7 Disruption of the CC ligaments with AC separation likely dissipates the stress necessary to create a clavicle fracture in most cases,1 explaining the rarity of this injury. It is imperative to evaluate patients for injury to both the osseous and ligamentous structures.
Previous case reports of concomitant AC separation and midshaft clavicle fracture have described a variety of treatment options, but to date our case represents the only episode in which both the clavicle fracture and the AC joint were treated with open reduction and internal fixation (ORIF). Wurtz and colleagues5 reported on a series of 4 patients with AC disruption and middle-third clavicle fracture. Three of the 4 patients had type IV AC separation; all 3 were treated, 2 acutely and 1 chronically, with open reduction of the AC and CC joints; 2 of these patients had CC screw fixation only after reduction, and the third had 2 Steinmann pins placed across the AC joint without CC screw fixation. All hardware was removed after 12 weeks. The fourth patient had a type II AC dislocation and was treated with closed reduction of the clavicle with no intervention for the AC joint. None of the clavicle fractures in this series were treated with internal fixation. All patients had full and pain-free ROM at 1- to 3-year follow-up.
Juhn and Simonian3 reported on a case of type VI separation with greenstick midshaft clavicle fracture in a hockey player seen 7 days after injury. The patient described some tingling in the upper extremity and had shoulder pain on initial presentation but was noted to have minimal displacement of both the AC joint and the midshaft clavicle fracture. Both injuries were treated nonsurgically with good outcome, and the patient returned to full activity (including hockey) within 14 weeks after injury.
Lancourt4 described the case of a patient with a type V AC dislocation and a displaced midshaft clavicle fracture. The AC joint was treated with Steinmann pin fixation, and the clavicle fracture was treated nonoperatively. The author cited high complication rates of plate fixation for clavicle fractures as the reason for not performing the additional procedure. The pins were removed 8 weeks after surgery. At 3-year follow-up, the patient had good radiographic and clinical outcome.
Yeh and colleagues6 described a patient who sustained a displaced midshaft clavicle fracture and a type IV AC dislocation in a fall from a horse. The patient underwent ORIF of the clavicle fracture with plate fixation. After the procedure, the AC joint was still unstable intraoperatively, and the AC and CC ligaments were reconstructed with semitendinosus allograft. The patient had full and painless ROM at 1-year follow-up.
The present case report serves as a reminder to obtain adequate shoulder radiographs when evaluating “just another clavicle fracture.” The radiographs should include a good axillary view to ensure there is not an associated AC dislocation. Increasingly, some authors have been advocating internal fixation for clavicle fractures, with reports of improved functional outcomes, improved cosmesis, and increased union rates.2 Indications for operative fixation include shortening and 100% displacement,8 and relative indications include open fractures.1 Operative fixation is perhaps more important for younger, athletic, and manual-labor populations. The trend in treatment of clavicle fractures toward operative fixation lends itself well to ORIF of the AC and CC joints; hence, a modern treatment for this rarely described combination injury should include internal plate fixation of the clavicle in addition to CC fixation. This additional procedure requires little extra time and energy in an operative scenario already requiring anesthesia, with easy insertion of the CC screw through the clavicle plate. Use of a CC screw obviates any potential risks associated with use of allograft tissue, and there is no anticipated difficulty with screw removal at 12 weeks.
Alternative options for AC stability include CC reconstruction with ligamentous allograft, ligamentous autograft, or suture/tightrope techniques. A noted advantage of these alternative techniques is less need to return to the operating room for the hardware removal that is recommended with CC screw fixation. However, these procedures potentially increase surgical exposure and operating time. In addition, screw fixation minimizes the possibility of donor-site morbidity from autograft transfers and potential complications from allograft tissue.
Hook plate fixation of the AC joint has also been described. In a recent case report of a similar injury pattern, plate fixation of the clavicle with simultaneous hook plate fixation of the AC joint was described.9 The patient did well but required removal of hardware of the hook plate and the clavicle plate 1 and 3 years after surgery, respectively. Although screw fixation is biomechanically stronger, debate persists about the clinical importance of this increase in strength.1 In the setting of plate fixation for the clavicle, these alternative AC fixations would require technique adjustments, including length of grafts and/or sutures, and raise concerns regarding interaction of the metal with the fixation material.
Critical evaluation of our technique revealed a lucency larger than the screw (Figure 5). However, the screw was not clinically loose at removal. This potential complication, in combination with the bent screw (Figure 4) before removal, highlights the concern for screw breakage with this technique, given the increased construct stiffness caused by the added plate.
Conclusion
As in the other reports mentioned, our patient had an excellent clinical and radiographic outcome. It could be inferred that, if fixation for isolated clavicle fractures demonstrates improved function, better outcomes would be seen for higher-energy fractures associated with AC dislocation. Given the current trend toward surgical fixation for certain clavicle fractures, we recommend that clavicle fractures associated with type IV AC dislocation be treated with ORIF of both injuries.
Acromioclavicular (AC) dislocations and displaced fractures of the middle third of the clavicle rarely occur together. Isolated AC joint separation is often treated nonoperatively with internal coracoclavicular (CC) fixation or reconstruction considered for type IV-VI AC dislocations and some type III injuries.1 Isolated clavicle fractures traditionally have been treated nonoperatively. The current trend is toward internal fixation for displaced and shortened fractures.2 There have been only a handful of reports of concomitant AC dislocation and midshaft clavicle fracture.3-6 Previous treatments have included nonoperative treatment, AC fixation, or internal fixation of the clavicle with ligamentous reconstruction.
We present a previously undescribed technique for internal fixation of this rare shoulder injury. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
While driving an all-terrain vehicle, a healthy 19-year-old right-hand–dominant man hit a bridge and sustained direct impact to his right shoulder. He presented to the emergency department complaining of right shoulder pain and deformity without skin disruption, vascular insufficiency, or neurologic symptoms. Anteroposterior (AP) radiograph showed an oblique, displaced, middle-third clavicle shaft fracture (Figure 1). An associated type IV AC dislocation was confirmed on axillary radiograph (Figure 2) and on an axial cut from a trauma chest computed tomography (CT) scan (Figure 3). The patient was discharged home from the trauma service the next day with a sling for comfort and plans for delayed, elective operative fixation 1 week later.
The patient was placed in a beach-chair position. Through a longitudinal incision extending laterally over the AC joint, the clavicle was exposed for fracture reduction, with care taken to retain soft-tissue attachments. The distal clavicle was buttonholed posteriorly through the trapezius muscle and fascia. The distal fracture fragment was devoid of any remaining CC ligamentous attachment. After satisfactory reduction, a low-profile precontoured clavicle plate (Superior Midshaft Clavicle Plate; Acumed, Hillsboro, Oregon) was placed superiorly; the fracture was compressed through the plate and internally fixed with three 3.5-mm bicortical screws on both sides of the fracture. Approximately 5 mm of the distal clavicle was resected at the AC joint to facilitate adequate AC and CC reduction without disruption of the clavicle fracture. With an adequate CC reduction, a 3.5-mm fully threaded cortical screw was placed through the most distal hole in the clavicle plate, clavicle, and coracoid.
After surgery, the patient was placed into an ARC shoulder immobilizer (Bledsoe, Grand Prairie, Texas) for 6 weeks, removing the immobilizer only for elbow and wrist range of motion (ROM) exercises. Radiographs at 3-month follow-up (Figure 4) showed a healed fracture with no loss of AC or CC reduction. Three months after surgery, another procedure was performed to remove the CC screw. One year after the initial surgery, the patient complained of intermittent soreness over the lateral shoulder but was not limited in his activities and was back to performing manual labor without difficulty. He had full ROM in forward flexion, abduction, internal rotation, and external rotation without weakness, tenderness, or any neurovascular deficit. After CC screw removal, no deformity returned at the shoulder. Radiographs showed a healed fracture with minimal superior subluxation at the AC joint without significant change from the 3-month follow-up (Figure 5).
Discussion
The combined injury pattern of a type IV AC dislocation and a displaced middle-third clavicle shaft fracture is rare. The usual mechanism of injury, as seen in the present case, is a direct blow to the shoulder at the tip of the acromion, though indirect forces from a fall on an outstretched hand are also described.7 Disruption of the CC ligaments with AC separation likely dissipates the stress necessary to create a clavicle fracture in most cases,1 explaining the rarity of this injury. It is imperative to evaluate patients for injury to both the osseous and ligamentous structures.
Previous case reports of concomitant AC separation and midshaft clavicle fracture have described a variety of treatment options, but to date our case represents the only episode in which both the clavicle fracture and the AC joint were treated with open reduction and internal fixation (ORIF). Wurtz and colleagues5 reported on a series of 4 patients with AC disruption and middle-third clavicle fracture. Three of the 4 patients had type IV AC separation; all 3 were treated, 2 acutely and 1 chronically, with open reduction of the AC and CC joints; 2 of these patients had CC screw fixation only after reduction, and the third had 2 Steinmann pins placed across the AC joint without CC screw fixation. All hardware was removed after 12 weeks. The fourth patient had a type II AC dislocation and was treated with closed reduction of the clavicle with no intervention for the AC joint. None of the clavicle fractures in this series were treated with internal fixation. All patients had full and pain-free ROM at 1- to 3-year follow-up.
Juhn and Simonian3 reported on a case of type VI separation with greenstick midshaft clavicle fracture in a hockey player seen 7 days after injury. The patient described some tingling in the upper extremity and had shoulder pain on initial presentation but was noted to have minimal displacement of both the AC joint and the midshaft clavicle fracture. Both injuries were treated nonsurgically with good outcome, and the patient returned to full activity (including hockey) within 14 weeks after injury.
Lancourt4 described the case of a patient with a type V AC dislocation and a displaced midshaft clavicle fracture. The AC joint was treated with Steinmann pin fixation, and the clavicle fracture was treated nonoperatively. The author cited high complication rates of plate fixation for clavicle fractures as the reason for not performing the additional procedure. The pins were removed 8 weeks after surgery. At 3-year follow-up, the patient had good radiographic and clinical outcome.
Yeh and colleagues6 described a patient who sustained a displaced midshaft clavicle fracture and a type IV AC dislocation in a fall from a horse. The patient underwent ORIF of the clavicle fracture with plate fixation. After the procedure, the AC joint was still unstable intraoperatively, and the AC and CC ligaments were reconstructed with semitendinosus allograft. The patient had full and painless ROM at 1-year follow-up.
The present case report serves as a reminder to obtain adequate shoulder radiographs when evaluating “just another clavicle fracture.” The radiographs should include a good axillary view to ensure there is not an associated AC dislocation. Increasingly, some authors have been advocating internal fixation for clavicle fractures, with reports of improved functional outcomes, improved cosmesis, and increased union rates.2 Indications for operative fixation include shortening and 100% displacement,8 and relative indications include open fractures.1 Operative fixation is perhaps more important for younger, athletic, and manual-labor populations. The trend in treatment of clavicle fractures toward operative fixation lends itself well to ORIF of the AC and CC joints; hence, a modern treatment for this rarely described combination injury should include internal plate fixation of the clavicle in addition to CC fixation. This additional procedure requires little extra time and energy in an operative scenario already requiring anesthesia, with easy insertion of the CC screw through the clavicle plate. Use of a CC screw obviates any potential risks associated with use of allograft tissue, and there is no anticipated difficulty with screw removal at 12 weeks.
Alternative options for AC stability include CC reconstruction with ligamentous allograft, ligamentous autograft, or suture/tightrope techniques. A noted advantage of these alternative techniques is less need to return to the operating room for the hardware removal that is recommended with CC screw fixation. However, these procedures potentially increase surgical exposure and operating time. In addition, screw fixation minimizes the possibility of donor-site morbidity from autograft transfers and potential complications from allograft tissue.
Hook plate fixation of the AC joint has also been described. In a recent case report of a similar injury pattern, plate fixation of the clavicle with simultaneous hook plate fixation of the AC joint was described.9 The patient did well but required removal of hardware of the hook plate and the clavicle plate 1 and 3 years after surgery, respectively. Although screw fixation is biomechanically stronger, debate persists about the clinical importance of this increase in strength.1 In the setting of plate fixation for the clavicle, these alternative AC fixations would require technique adjustments, including length of grafts and/or sutures, and raise concerns regarding interaction of the metal with the fixation material.
Critical evaluation of our technique revealed a lucency larger than the screw (Figure 5). However, the screw was not clinically loose at removal. This potential complication, in combination with the bent screw (Figure 4) before removal, highlights the concern for screw breakage with this technique, given the increased construct stiffness caused by the added plate.
Conclusion
As in the other reports mentioned, our patient had an excellent clinical and radiographic outcome. It could be inferred that, if fixation for isolated clavicle fractures demonstrates improved function, better outcomes would be seen for higher-energy fractures associated with AC dislocation. Given the current trend toward surgical fixation for certain clavicle fractures, we recommend that clavicle fractures associated with type IV AC dislocation be treated with ORIF of both injuries.
1. Ring D, Jupiter J. Injuries to the shoulder girdle. In: Browner, BD. Skeletal Trauma. Philadelphia, PA: Elsevier Health Sciences; 2008:1755-1778.
2. Altamimi SA, McKee MD; Canadian Orthopaedic Trauma Society. Nonoperative treatment compared with plate fixation of displaced midshaft clavicular fractures. Surgical technique. J Bone Joint Surg Am. 2008;90(suppl 2 pt 1):1-8.
3. Juhn MS, Simonian PT. Type VI acromioclavicular separation with middle-third clavicle fracture in an ice hockey player. Clin J Sports Med. 2002;12(5):315-317.
4. Lancourt JE. Acromioclavicular dislocation with adjacent clavicular fracture in a horseback rider. A case report. Am J Sports Med. 1990;18(3):321-322.
5. Wurtz LD, Lyons FA, Rockwood CA Jr. Fracture of the middle third of the clavicle and dislocation of the acromioclavicular joint. A report of four cases. J Bone Joint Surg Am. 1992;74(1):133-137.
6. Yeh PC, Miller SR, Cunningham JG, Sethi PM. Midshaft clavicle fracture and acromioclavicular dislocation: a case report of a rare injury. J Shoulder Elbow Surg. 2009;18(5):e1-e4.
7. Stanley D, Trowbridge EA, Norris SH. The mechanism of clavicular fracture. A clinical and biomechanical analysis. J Bone Joint Surg Br. 1988;70(3):461-464.
8. Kim W, McKee MD. Management of acute clavicle fractures. Orthop Clin North Am. 2008;39(4):491-505.
9. Woolf SK, Valentine BJ, Barfield WR, Hartsock LA. Middle-third clavicle fracture with associated type IV acromioclavicular separation: case report and literature review. J Surg Orthop Adv. 2013;22(2):183-186.
1. Ring D, Jupiter J. Injuries to the shoulder girdle. In: Browner, BD. Skeletal Trauma. Philadelphia, PA: Elsevier Health Sciences; 2008:1755-1778.
2. Altamimi SA, McKee MD; Canadian Orthopaedic Trauma Society. Nonoperative treatment compared with plate fixation of displaced midshaft clavicular fractures. Surgical technique. J Bone Joint Surg Am. 2008;90(suppl 2 pt 1):1-8.
3. Juhn MS, Simonian PT. Type VI acromioclavicular separation with middle-third clavicle fracture in an ice hockey player. Clin J Sports Med. 2002;12(5):315-317.
4. Lancourt JE. Acromioclavicular dislocation with adjacent clavicular fracture in a horseback rider. A case report. Am J Sports Med. 1990;18(3):321-322.
5. Wurtz LD, Lyons FA, Rockwood CA Jr. Fracture of the middle third of the clavicle and dislocation of the acromioclavicular joint. A report of four cases. J Bone Joint Surg Am. 1992;74(1):133-137.
6. Yeh PC, Miller SR, Cunningham JG, Sethi PM. Midshaft clavicle fracture and acromioclavicular dislocation: a case report of a rare injury. J Shoulder Elbow Surg. 2009;18(5):e1-e4.
7. Stanley D, Trowbridge EA, Norris SH. The mechanism of clavicular fracture. A clinical and biomechanical analysis. J Bone Joint Surg Br. 1988;70(3):461-464.
8. Kim W, McKee MD. Management of acute clavicle fractures. Orthop Clin North Am. 2008;39(4):491-505.
9. Woolf SK, Valentine BJ, Barfield WR, Hartsock LA. Middle-third clavicle fracture with associated type IV acromioclavicular separation: case report and literature review. J Surg Orthop Adv. 2013;22(2):183-186.
Does a Prior Hip Arthroscopy Affect Clinical Outcomes in Metal-on-Metal Hip Resurfacing Arthroplasty?
Metal-on-metal hip resurfacing arthroplasty (HRA) remains an alternative to total hip arthroplasty (THA) in appropriately selected, younger, active adults with degenerative hip disease.1-4 While concerns remain regarding the potential for adverse local tissue reactions from wear of the metal-on-metal bearing surface,5-8 10-year data from the Australian Orthopaedic Association National Joint Replacement Registry Annual Report9 showed a revision rate of only 6.3% when the Birmingham Hip Resurfacing (BHR) System was used (Smith & Nephew Inc, Memphis, Tennessee).In addition, in an independent review of 230 consecutive BHRs at a mean follow-up of 10.4 years, Coulter and colleagues10 showed encouraging clinical results, with a mean Oxford Hip Score of 45.0 and a mean University of California at Los Angeles (UCLA) activity score of 7.4.
Similar to the prior increase in popularity of HRA, hip arthroscopy has also become much more commonplace, and its indications continue to evolve.11 Hip arthroscopy has been used in the native hip joint to manage femoroacetabular impingement, labral tears, and iliopsoas tendinopathy, among other conditions.12 In addition, the use of hip arthroscopy has not been limited to the native hip but also has increased as a diagnostic and therapeutic procedure after hip arthroplasties. Bajwa and Villar12 found hip arthroscopy to be diagnostic in 23 of 24 patients who underwent the procedure after a hip arthroplasty, concluding that arthroscopy is a useful adjunct in the diagnosis of symptomatic arthroplasties.
Therefore, hip arthroscopy has been shown to be an effective modality to treat pathology in both the native hip and after hip arthroplasties. However, the effect of a prior hip arthroscopy on the outcome of a subsequent metal-on-metal HRA has not been determined. Piedade and colleagues13 showed a prior knee arthroscopy to increase the risk of postoperative complications and subsequent revision after total knee arthroplasty. Complications included reflex sympathetic dystrophy, undiagnosed pain, infection, stiffness, and component loosening. A prior osteochondroplasty at the femoral head-neck junction could increase the risk of femoral neck fracture after a subsequent HRA. Thus, the purpose of this study was to evaluate the clinical outcomes of a series of patients who received an HRA after a prior hip arthroscopy and to compare these results with a cohort of patients who received an HRA with no prior hip surgeries. Our hypothesis is that a prior hip arthroscopy will lead to inferior outcomes in patients undergoing HRA.
Materials and Methods
This study is a retrospective, case-control study using a 1:2 matching analysis. Dr. Su performed all HRAs, which were enrolled in an institutional review board–approved arthroplasty registry. All HRAs were performed using the BHR System.
The surgical technique for hip resurfacing arthroplasty has been described.1 All procedures were performed via a posterior approach with the patient in the lateral decubitus position. All patients received a hybrid metal-on-metal hip resurfacing, with an uncemented acetabular component and cemented femoral component. Intraoperative anesthesia for all patients was performed via a combined spinal-epidural anesthetic, and an epidural patient-controlled analgesic was used for the first day postoperatively, followed by a transition to oral analgesics. The sizes of the acetabular and femoral components were recorded for each hip resurfacing. Postoperatively, intermittent pneumatic compression devices were placed upon arrival in the recovery room, and active ankle flexion and extension exercises were initiated immediately after the patient’s neurologic function returned.14 Aspirin was used for chemical deep venous thrombosis prophylaxis in all patients postoperatively for a period of 6 weeks. Full weight-bearing, with the use of crutches for assistance with balance, was permitted immediately. Crutches were used for a period of 3 weeks prior to being discontinued.
From a database of 1357 HRAs (all BHR implants) performed between June 2006 and June 2012, 51 patients were identified who received an HRA after a prior hip arthroscopy. Eight patients were excluded because they did not possess adequate clinical documentation or were lost to follow-up. In the remaining 43 patients, there were 32 men and 11 women (21 right hips, 22 left hips), which formed the arthroscopy cohort. Two patients had a history of multiple hip arthroscopies (1 patient with 2 prior procedures, 1 patient with 3 prior procedures). The mean (SD) time from the most recent hip arthroscopy to the HRA was 2.5 (2.5) years. Table 1 presents a summary of the hip arthroscopy procedures (including only the most recent hip arthroscopy procedure in those with multiple arthroscopies).
Patient demographic variables (age, body mass index [BMI]) were recorded preoperatively, along with the Harris Hip Score (HHS),15 UCLA activity score,16 Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) score,17 and preoperative hip range of motion (flexion, extension, abduction, adduction, internal rotation, and external rotation). The same clinical indices were assessed postoperatively along with the Short Form-12 (SF-12) Health Survey Score,18 at the 6-week, 3-month, 6-month, 1-year, and most recent follow-up visits.
Radiographic assessment consisted of a low anteroposterior (AP) pelvic radiograph (with the radiographic beam centered on the pubic symphysis) and a cross-table lateral radiograph obtained at the most recent follow-up visit. Both the acetabular component abduction relative to the inter-teardrop line, and the angle between the femoral stem and the anatomic axis of the femoral shaft (stem-shaft angle) were measured on AP radiographs.19,20 Acetabular component anteversion was measured on the cross-table lateral radiographs as the angle between the projected long axis of the acetabular opening and a line drawn perpendicular to the long axis plane of the body (Figures A, B).21
The same registry database was used to identify patients who received an HRA without a prior history of arthroscopy or hip surgery. A 1:2 matching analysis for those patients with a prior hip arthroscopy to those without a prior hip arthroscopy was performed to formulate a control group (control cohort) of 86 patients. Each patient in the arthroscopy cohort was matched with 2 patients in the control cohort based on the following parameters: age (± 6 years), sex (same), BMI (± 4 kg/m2), femoral head size (± 4 mm), and preoperative HHS and WOMAC scores (± 7 points). In the event an arthroscopy patient matched to 2 or more control patients, the patients who minimized the least squared error among the matching variables were selected.
Statistical Analysis
All data were collected and analyzed using Microsoft Excel software (Microsoft Corporation, Redmond, Washington). Statistical comparisons between the 2 cohorts regarding demographic variables, clinical outcomes, and radiographic alignment were performed using an unpaired, Student 2-tailed t test, with statistical significance set at P ≤ .05.
Results
A comparison of the results of the 1:2 matching analysis between the arthroscopy and control cohorts is presented in Table 2. There was no significant difference in the preoperative age, BMI, femoral head size, HHS, or WOMAC score between the 2 cohorts. However, the control cohort did show a more severe, preoperative flexion contracture (as expressed by a decreased amount of extension) and a decreased amount of preoperative abduction (Table 3). The preoperative UCLA activity score was also decreased in the control cohort, but this was not statistically significant.
The mean (SD) follow-up was 2.0 (1.0) years in the arthroscopy cohort and 2.1 (1.1) years in the control cohort. There was no significant difference in radiographic alignment between the 2 cohorts. The stem-shaft angle was 139.3° (SD, 5.4°) in the arthroscopy cohort (vs 138.3° [SD, 5.5°] in the control cohort; P = .3), the acetabular abduction was 43.9° (SD, 5.8°) in the arthroscopy cohort (vs 42.9° [SD, 6.1°] in the control cohort; P = .4), and the acetabular anteversion was 21.1° (SD, 7.5°) in the arthroscopy cohort (vs 20.8° [SD, 7.1°] in the control cohort; P = .8).
At 6-week follow-up, the arthroscopy cohort showed a significantly decreased WOMAC score compared with the control cohort (72.9 [SD, 15.5] vs 80.5 [SD, 11.8], respectively; P = .05). In addition, there was a trend towards a decreased SF-12 mental component score in the arthroscopy cohort (52.2 [SD, 9.3] vs 56.5 [SD, 7.8] in the control cohort; P = .06). However, none of the remaining clinical indices showed a significant difference between the 2 cohorts, and there was no difference in range of motion between the 2 cohorts at the 6-week follow-up visit (Table 4).
In addition, at 3-month follow-up, no statistically significant differences were seen between the 2 cohorts for any of the clinical indices or range of motion values. Both groups continued to improve rapidly, with HHS of 96.9 (SD, 3.5) in the arthroscopy cohort and 95.5 (SD, 6.6) in the control cohort, and WOMAC scores of 88.7 (SD, 10.2) and 89.5 (SD, 9.8), respectively (Table 5). Similarly, at the 6-month and 1-year follow-up intervals, the 2 cohorts showed continued improvement in their clinical measures, with no statistically significant differences between the 2 cohorts (Tables 6, 7).
At the most recent follow-up visit, more than 1 year after surgery, the HHS was 99.5 (SD, 1.3) in the arthroscopy cohort and 99.2 (SD, 9.7) in the control cohort (P = .9), and the WOMAC score was 93.5 (SD, 11.3) and 92.4 (SD, 12.2), respectively (P = .8). No significant perioperative complications were seen in the arthroscopy cohort. In the arthroscopy cohort, 1 patient was diagnosed with a deep venous thrombosis 2 weeks after the procedure and was placed on low-molecular-weight heparin and coumadin for treatment. A second patient in the arthroscopy cohort had continued serosanguinous drainage for 4 days postoperatively, which resolved with continued compressive dressings. To date, no patients in the arthroscopy or control cohorts have required a second operation or revision of their components.
Discussion
Given the increasing prevalence of hip arthroscopies to treat multiple disorders of the native joint, it is important to assess the potential consequences of these procedures on future arthroplasties. Piedade and colleagues,13 in a retrospective review of 1474 primary total knee arthroplasties, showed a prior bony procedure (high tibial osteotomy, tibial plateau fracture, patellar realignment) to be predictive of decreased range of motion postoperatively. In addition, a prior knee arthroscopy was associated with a higher rate of postoperative complications, with 30% of the complications requiring a reoperation, and 8.3% of the complications requiring a revision total knee arthroplasty. Kaplan-Meier survival curves showed a survival rate of only 86.8% in those patients with a prior knee arthroscopy (vs 98.1% in those without a prior knee surgery).22 Therefore, the purpose of this study was to evaluate the clinical outcomes of a series of patients who received an HRA after a prior hip arthroscopy. After the initial 6-week follow-up visit, no significant difference was seen in the functional outcomes between those patients with or without a history of prior hip arthroscopy who received an HRA.
After analysis of patient outcomes using multiple clinical measurement tools, at 6-week, 3-month, 6-month, 1-year, and most recent follow-up intervals, the only significant difference between the 2 cohorts was the WOMAC score at 6-week follow-up. Interestingly, there was no significant difference seen in the other clinical assessments, including the SF-12 score, HHS, range of motion, or UCLA activity score (although this did trend towards significance). This can be explained by the difference in both the mode of administration and various metrics assessed by these instruments. In comparison to the HHS evaluation, the patient completes the WOMAC (rather than the clinician) and also provides a more detailed assessment of symptoms, pain, stiffness, and activities of daily living.17 Therefore, this study suggests that patients with a prior hip arthroscopy may require more time to return to their activities of daily living after an HRA. However, whether the statistically significant difference between the 2 scores translates into a clinically significant difference can be questioned.
The clinical outcomes of this series of patients were excellent at the short-term follow-up, and both groups achieved clinical results comparable to prior reported results of HRA.1,10,23,24 However, despite these results, there are several limitations to this study. First, longer-term follow-up is required to determine if any significant differences (such as aseptic loosening, infection, and prosthesis survival) are associated with a prior hip arthroscopy. In addition, this study included a relatively small cohort of patients who had a prior hip arthroscopy. However, a relatively large, single-surgeon database of 1357 HRAs was reviewed, with only 51 cases being reported (3.7%). With the increasing popularity of hip arthroscopy, the number of patients presenting for HRA will likely continue to increase. However, despite these limitations, this study shows that a prior hip arthroscopy does not appear to affect the short-term, clinical outcomes of a metal-on-metal HRA.
1. Amstutz HC, Beaulé PE, Dorey FJ, Le Duff MJ, Campbell PA, Gruen TA. Metal-on-metal hybrid surface arthroplasty. Surgical Technique. J Bone Joint Surg Am. 2006;88(suppl 1 Pt 2):234-249.
2. Daniel J, Pynsent PB, McMinn DJ. Metal-on-metal resurfacing of the hip in patients under the age of 55 years with osteoarthritis. J Bone Joint Surg Br. 2004;86(2):177-184.
3. Pollard TC, Baker RP, Eastaugh-Waring SJ, Bannister GC. Treatment of the young active patient with osteoarthritis of the hip. A five- to seven-year comparison of hybrid total hip arthroplasty and metal-on-metal resurfacing. J Bone Joint Surg Br. 2006;88(5):592-600.
4. Treacy RB, McBryde CW, Pynsent PB. Birmingham hip resurfacing arthroplasty. A minimum follow-up of five years. J Bone Joint Surg Br. 2005;87(2):167-170.
5. Amstutz HC, Le Duff MJ, Campbell PA, Gruen TA, Wisk LE. Clinical and radiographic results of metal-on-metal hip resurfacing with a minimum ten-year follow-up. J Bone Joint Surg Am. 2010;92(16):2663-2671.
6. Daniel J, Ziaee H, Pradhan C, Pynsent PB, McMinn DJ. Blood and urine metal ion levels in young and active patients after Birmingham hip resurfacing arthroplasty: four-year results of a prospective longitudinal study.
J Bone Joint Surg Br. 2007;89(2):169-173.
7. deSouza RM, Parsons NR, Oni T, Dalton P, Costa M, Krikler S. Metal ion levels following resurfacing arthroplasty of the hip: serial results over a ten-year period. J Bone Joint Surg Br. 2010;92(12):1642-1647.
8. Kwon YM, Thomas P, Summer B, et al. Lymphocyte proliferation responses in patients with pseudotumors following metal-on-metal hip resurfacing arthroplasty. J Orthop Res. 2010;28(4):444-450.
9. Australian Orthopaedic Association National Joint Replacement Registry. Annual Report 2011. Adelaide: Australian Orthopaedic Association; 2011. https://aoanjrr.dmac.adelaide.edu.au/annual-reports-2011. Accessed September 16, 2014.
10. Coulter G, Young DA, Dalziel RE, Shimmin AJ. Birmingham hip resurfacing at a mean of ten years: results from an independent centre. J Bone Joint Surg Br. 2012;94(3):315-321.
11. McCarthy JC, Jarrett BT, Ojeifo O, Lee JA, Bragdon CR. What factors influence long-term survivorship after hip arthroscopy? Clin Orthop. 2011;469(2):362-371.
12. Bajwa AS, Villar RN. Arthroscopy of the hip in patients following joint replacement. J Bone Joint Surg Br. 2011;93(7):890-896.
13. Piedade SR, Pinaroli A, Servien E, Neyret P. Is previous knee arthroscopy related to worse results in primary total knee arthroplasty? Knee Surg Sports Traumatol Arthrosc. 2009;17(4):328-333.
14. Gonzalez Della Valle A, Serota A, Go G, et al. Venous thromboembolism is rare with a multimodal prophylaxis protocol after total hip arthroplasty. Clin Orthop. 2006;(444):146-153.
15. Harris WH. Traumatic arthritis of the hip after dislocation and acetabular fractures: treatment by mold arthroplasty. An end-result study using a new method of result evaluation. J Bone Joint Surg Am. 1969;51(4):737-755.
16. Kershaw CJ, Atkins RM, Dodd CA, Bulstrode CJ. Revision total hip arthroplasty for aseptic failure. A review of 276 cases. J Bone Joint Surg Br. 1991;73(4):564-568.
17. Bellamy N. WOMAC: a 20-year experiential review of a patient-centered self-reported health status questionnaire. J Rheumatol. 2002;29(12):2473-2476.
18. Ware J Jr, Kosinski M, Keller SD. A 12-Item Short-Form Health Survey: construction of scales and preliminary tests of reliability and validity. Med Care. 1996;34(3):220-233.
19. Clark JM, Freeman MA, Witham D. The relationship of neck orientation to the shape of the proximal femur. J Arthroplasty. 1987;2(2):99-109.
20. Lewinnek GE, Lewis JL, Tarr R, Compere CL, Zimmerman JR. Dislocations after total hip-replacement arthroplasties. J Bone Joint Surg Am. 1978;60(2):217-220.
21. Yao L, Yao J, Gold RH. Measurement of acetabular version on the axiolateral radiograph. Clin Orthop. 1995;(316):106-111.
22. Piedade SR, Pinaroli A, Servien E, Neyret P. TKA outcomes after prior bone and soft tissue knee surgery. Knee Surg Sports Traumatol Arthrosc. 2013;21(12):2737-2743.
23. Amstutz HC, Beaulé PE, Dorey FJ, Le Duff MJ, Campbell PA, Gruen TA. Metal-on-metal hybrid surface arthroplasty: two to six-year follow-up study. J Bone Joint Surg Am. 2004;86(1):28-39.
24. Steffen RT, Pandit HP, Palan J, et al. The five-year results of the Birmingham Hip Resurfacing arthroplasty: an independent series. J Bone Joint Surg Br. 2008;90(4):436-441.
Metal-on-metal hip resurfacing arthroplasty (HRA) remains an alternative to total hip arthroplasty (THA) in appropriately selected, younger, active adults with degenerative hip disease.1-4 While concerns remain regarding the potential for adverse local tissue reactions from wear of the metal-on-metal bearing surface,5-8 10-year data from the Australian Orthopaedic Association National Joint Replacement Registry Annual Report9 showed a revision rate of only 6.3% when the Birmingham Hip Resurfacing (BHR) System was used (Smith & Nephew Inc, Memphis, Tennessee).In addition, in an independent review of 230 consecutive BHRs at a mean follow-up of 10.4 years, Coulter and colleagues10 showed encouraging clinical results, with a mean Oxford Hip Score of 45.0 and a mean University of California at Los Angeles (UCLA) activity score of 7.4.
Similar to the prior increase in popularity of HRA, hip arthroscopy has also become much more commonplace, and its indications continue to evolve.11 Hip arthroscopy has been used in the native hip joint to manage femoroacetabular impingement, labral tears, and iliopsoas tendinopathy, among other conditions.12 In addition, the use of hip arthroscopy has not been limited to the native hip but also has increased as a diagnostic and therapeutic procedure after hip arthroplasties. Bajwa and Villar12 found hip arthroscopy to be diagnostic in 23 of 24 patients who underwent the procedure after a hip arthroplasty, concluding that arthroscopy is a useful adjunct in the diagnosis of symptomatic arthroplasties.
Therefore, hip arthroscopy has been shown to be an effective modality to treat pathology in both the native hip and after hip arthroplasties. However, the effect of a prior hip arthroscopy on the outcome of a subsequent metal-on-metal HRA has not been determined. Piedade and colleagues13 showed a prior knee arthroscopy to increase the risk of postoperative complications and subsequent revision after total knee arthroplasty. Complications included reflex sympathetic dystrophy, undiagnosed pain, infection, stiffness, and component loosening. A prior osteochondroplasty at the femoral head-neck junction could increase the risk of femoral neck fracture after a subsequent HRA. Thus, the purpose of this study was to evaluate the clinical outcomes of a series of patients who received an HRA after a prior hip arthroscopy and to compare these results with a cohort of patients who received an HRA with no prior hip surgeries. Our hypothesis is that a prior hip arthroscopy will lead to inferior outcomes in patients undergoing HRA.
Materials and Methods
This study is a retrospective, case-control study using a 1:2 matching analysis. Dr. Su performed all HRAs, which were enrolled in an institutional review board–approved arthroplasty registry. All HRAs were performed using the BHR System.
The surgical technique for hip resurfacing arthroplasty has been described.1 All procedures were performed via a posterior approach with the patient in the lateral decubitus position. All patients received a hybrid metal-on-metal hip resurfacing, with an uncemented acetabular component and cemented femoral component. Intraoperative anesthesia for all patients was performed via a combined spinal-epidural anesthetic, and an epidural patient-controlled analgesic was used for the first day postoperatively, followed by a transition to oral analgesics. The sizes of the acetabular and femoral components were recorded for each hip resurfacing. Postoperatively, intermittent pneumatic compression devices were placed upon arrival in the recovery room, and active ankle flexion and extension exercises were initiated immediately after the patient’s neurologic function returned.14 Aspirin was used for chemical deep venous thrombosis prophylaxis in all patients postoperatively for a period of 6 weeks. Full weight-bearing, with the use of crutches for assistance with balance, was permitted immediately. Crutches were used for a period of 3 weeks prior to being discontinued.
From a database of 1357 HRAs (all BHR implants) performed between June 2006 and June 2012, 51 patients were identified who received an HRA after a prior hip arthroscopy. Eight patients were excluded because they did not possess adequate clinical documentation or were lost to follow-up. In the remaining 43 patients, there were 32 men and 11 women (21 right hips, 22 left hips), which formed the arthroscopy cohort. Two patients had a history of multiple hip arthroscopies (1 patient with 2 prior procedures, 1 patient with 3 prior procedures). The mean (SD) time from the most recent hip arthroscopy to the HRA was 2.5 (2.5) years. Table 1 presents a summary of the hip arthroscopy procedures (including only the most recent hip arthroscopy procedure in those with multiple arthroscopies).
Patient demographic variables (age, body mass index [BMI]) were recorded preoperatively, along with the Harris Hip Score (HHS),15 UCLA activity score,16 Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) score,17 and preoperative hip range of motion (flexion, extension, abduction, adduction, internal rotation, and external rotation). The same clinical indices were assessed postoperatively along with the Short Form-12 (SF-12) Health Survey Score,18 at the 6-week, 3-month, 6-month, 1-year, and most recent follow-up visits.
Radiographic assessment consisted of a low anteroposterior (AP) pelvic radiograph (with the radiographic beam centered on the pubic symphysis) and a cross-table lateral radiograph obtained at the most recent follow-up visit. Both the acetabular component abduction relative to the inter-teardrop line, and the angle between the femoral stem and the anatomic axis of the femoral shaft (stem-shaft angle) were measured on AP radiographs.19,20 Acetabular component anteversion was measured on the cross-table lateral radiographs as the angle between the projected long axis of the acetabular opening and a line drawn perpendicular to the long axis plane of the body (Figures A, B).21
The same registry database was used to identify patients who received an HRA without a prior history of arthroscopy or hip surgery. A 1:2 matching analysis for those patients with a prior hip arthroscopy to those without a prior hip arthroscopy was performed to formulate a control group (control cohort) of 86 patients. Each patient in the arthroscopy cohort was matched with 2 patients in the control cohort based on the following parameters: age (± 6 years), sex (same), BMI (± 4 kg/m2), femoral head size (± 4 mm), and preoperative HHS and WOMAC scores (± 7 points). In the event an arthroscopy patient matched to 2 or more control patients, the patients who minimized the least squared error among the matching variables were selected.
Statistical Analysis
All data were collected and analyzed using Microsoft Excel software (Microsoft Corporation, Redmond, Washington). Statistical comparisons between the 2 cohorts regarding demographic variables, clinical outcomes, and radiographic alignment were performed using an unpaired, Student 2-tailed t test, with statistical significance set at P ≤ .05.
Results
A comparison of the results of the 1:2 matching analysis between the arthroscopy and control cohorts is presented in Table 2. There was no significant difference in the preoperative age, BMI, femoral head size, HHS, or WOMAC score between the 2 cohorts. However, the control cohort did show a more severe, preoperative flexion contracture (as expressed by a decreased amount of extension) and a decreased amount of preoperative abduction (Table 3). The preoperative UCLA activity score was also decreased in the control cohort, but this was not statistically significant.
The mean (SD) follow-up was 2.0 (1.0) years in the arthroscopy cohort and 2.1 (1.1) years in the control cohort. There was no significant difference in radiographic alignment between the 2 cohorts. The stem-shaft angle was 139.3° (SD, 5.4°) in the arthroscopy cohort (vs 138.3° [SD, 5.5°] in the control cohort; P = .3), the acetabular abduction was 43.9° (SD, 5.8°) in the arthroscopy cohort (vs 42.9° [SD, 6.1°] in the control cohort; P = .4), and the acetabular anteversion was 21.1° (SD, 7.5°) in the arthroscopy cohort (vs 20.8° [SD, 7.1°] in the control cohort; P = .8).
At 6-week follow-up, the arthroscopy cohort showed a significantly decreased WOMAC score compared with the control cohort (72.9 [SD, 15.5] vs 80.5 [SD, 11.8], respectively; P = .05). In addition, there was a trend towards a decreased SF-12 mental component score in the arthroscopy cohort (52.2 [SD, 9.3] vs 56.5 [SD, 7.8] in the control cohort; P = .06). However, none of the remaining clinical indices showed a significant difference between the 2 cohorts, and there was no difference in range of motion between the 2 cohorts at the 6-week follow-up visit (Table 4).
In addition, at 3-month follow-up, no statistically significant differences were seen between the 2 cohorts for any of the clinical indices or range of motion values. Both groups continued to improve rapidly, with HHS of 96.9 (SD, 3.5) in the arthroscopy cohort and 95.5 (SD, 6.6) in the control cohort, and WOMAC scores of 88.7 (SD, 10.2) and 89.5 (SD, 9.8), respectively (Table 5). Similarly, at the 6-month and 1-year follow-up intervals, the 2 cohorts showed continued improvement in their clinical measures, with no statistically significant differences between the 2 cohorts (Tables 6, 7).
At the most recent follow-up visit, more than 1 year after surgery, the HHS was 99.5 (SD, 1.3) in the arthroscopy cohort and 99.2 (SD, 9.7) in the control cohort (P = .9), and the WOMAC score was 93.5 (SD, 11.3) and 92.4 (SD, 12.2), respectively (P = .8). No significant perioperative complications were seen in the arthroscopy cohort. In the arthroscopy cohort, 1 patient was diagnosed with a deep venous thrombosis 2 weeks after the procedure and was placed on low-molecular-weight heparin and coumadin for treatment. A second patient in the arthroscopy cohort had continued serosanguinous drainage for 4 days postoperatively, which resolved with continued compressive dressings. To date, no patients in the arthroscopy or control cohorts have required a second operation or revision of their components.
Discussion
Given the increasing prevalence of hip arthroscopies to treat multiple disorders of the native joint, it is important to assess the potential consequences of these procedures on future arthroplasties. Piedade and colleagues,13 in a retrospective review of 1474 primary total knee arthroplasties, showed a prior bony procedure (high tibial osteotomy, tibial plateau fracture, patellar realignment) to be predictive of decreased range of motion postoperatively. In addition, a prior knee arthroscopy was associated with a higher rate of postoperative complications, with 30% of the complications requiring a reoperation, and 8.3% of the complications requiring a revision total knee arthroplasty. Kaplan-Meier survival curves showed a survival rate of only 86.8% in those patients with a prior knee arthroscopy (vs 98.1% in those without a prior knee surgery).22 Therefore, the purpose of this study was to evaluate the clinical outcomes of a series of patients who received an HRA after a prior hip arthroscopy. After the initial 6-week follow-up visit, no significant difference was seen in the functional outcomes between those patients with or without a history of prior hip arthroscopy who received an HRA.
After analysis of patient outcomes using multiple clinical measurement tools, at 6-week, 3-month, 6-month, 1-year, and most recent follow-up intervals, the only significant difference between the 2 cohorts was the WOMAC score at 6-week follow-up. Interestingly, there was no significant difference seen in the other clinical assessments, including the SF-12 score, HHS, range of motion, or UCLA activity score (although this did trend towards significance). This can be explained by the difference in both the mode of administration and various metrics assessed by these instruments. In comparison to the HHS evaluation, the patient completes the WOMAC (rather than the clinician) and also provides a more detailed assessment of symptoms, pain, stiffness, and activities of daily living.17 Therefore, this study suggests that patients with a prior hip arthroscopy may require more time to return to their activities of daily living after an HRA. However, whether the statistically significant difference between the 2 scores translates into a clinically significant difference can be questioned.
The clinical outcomes of this series of patients were excellent at the short-term follow-up, and both groups achieved clinical results comparable to prior reported results of HRA.1,10,23,24 However, despite these results, there are several limitations to this study. First, longer-term follow-up is required to determine if any significant differences (such as aseptic loosening, infection, and prosthesis survival) are associated with a prior hip arthroscopy. In addition, this study included a relatively small cohort of patients who had a prior hip arthroscopy. However, a relatively large, single-surgeon database of 1357 HRAs was reviewed, with only 51 cases being reported (3.7%). With the increasing popularity of hip arthroscopy, the number of patients presenting for HRA will likely continue to increase. However, despite these limitations, this study shows that a prior hip arthroscopy does not appear to affect the short-term, clinical outcomes of a metal-on-metal HRA.
Metal-on-metal hip resurfacing arthroplasty (HRA) remains an alternative to total hip arthroplasty (THA) in appropriately selected, younger, active adults with degenerative hip disease.1-4 While concerns remain regarding the potential for adverse local tissue reactions from wear of the metal-on-metal bearing surface,5-8 10-year data from the Australian Orthopaedic Association National Joint Replacement Registry Annual Report9 showed a revision rate of only 6.3% when the Birmingham Hip Resurfacing (BHR) System was used (Smith & Nephew Inc, Memphis, Tennessee).In addition, in an independent review of 230 consecutive BHRs at a mean follow-up of 10.4 years, Coulter and colleagues10 showed encouraging clinical results, with a mean Oxford Hip Score of 45.0 and a mean University of California at Los Angeles (UCLA) activity score of 7.4.
Similar to the prior increase in popularity of HRA, hip arthroscopy has also become much more commonplace, and its indications continue to evolve.11 Hip arthroscopy has been used in the native hip joint to manage femoroacetabular impingement, labral tears, and iliopsoas tendinopathy, among other conditions.12 In addition, the use of hip arthroscopy has not been limited to the native hip but also has increased as a diagnostic and therapeutic procedure after hip arthroplasties. Bajwa and Villar12 found hip arthroscopy to be diagnostic in 23 of 24 patients who underwent the procedure after a hip arthroplasty, concluding that arthroscopy is a useful adjunct in the diagnosis of symptomatic arthroplasties.
Therefore, hip arthroscopy has been shown to be an effective modality to treat pathology in both the native hip and after hip arthroplasties. However, the effect of a prior hip arthroscopy on the outcome of a subsequent metal-on-metal HRA has not been determined. Piedade and colleagues13 showed a prior knee arthroscopy to increase the risk of postoperative complications and subsequent revision after total knee arthroplasty. Complications included reflex sympathetic dystrophy, undiagnosed pain, infection, stiffness, and component loosening. A prior osteochondroplasty at the femoral head-neck junction could increase the risk of femoral neck fracture after a subsequent HRA. Thus, the purpose of this study was to evaluate the clinical outcomes of a series of patients who received an HRA after a prior hip arthroscopy and to compare these results with a cohort of patients who received an HRA with no prior hip surgeries. Our hypothesis is that a prior hip arthroscopy will lead to inferior outcomes in patients undergoing HRA.
Materials and Methods
This study is a retrospective, case-control study using a 1:2 matching analysis. Dr. Su performed all HRAs, which were enrolled in an institutional review board–approved arthroplasty registry. All HRAs were performed using the BHR System.
The surgical technique for hip resurfacing arthroplasty has been described.1 All procedures were performed via a posterior approach with the patient in the lateral decubitus position. All patients received a hybrid metal-on-metal hip resurfacing, with an uncemented acetabular component and cemented femoral component. Intraoperative anesthesia for all patients was performed via a combined spinal-epidural anesthetic, and an epidural patient-controlled analgesic was used for the first day postoperatively, followed by a transition to oral analgesics. The sizes of the acetabular and femoral components were recorded for each hip resurfacing. Postoperatively, intermittent pneumatic compression devices were placed upon arrival in the recovery room, and active ankle flexion and extension exercises were initiated immediately after the patient’s neurologic function returned.14 Aspirin was used for chemical deep venous thrombosis prophylaxis in all patients postoperatively for a period of 6 weeks. Full weight-bearing, with the use of crutches for assistance with balance, was permitted immediately. Crutches were used for a period of 3 weeks prior to being discontinued.
From a database of 1357 HRAs (all BHR implants) performed between June 2006 and June 2012, 51 patients were identified who received an HRA after a prior hip arthroscopy. Eight patients were excluded because they did not possess adequate clinical documentation or were lost to follow-up. In the remaining 43 patients, there were 32 men and 11 women (21 right hips, 22 left hips), which formed the arthroscopy cohort. Two patients had a history of multiple hip arthroscopies (1 patient with 2 prior procedures, 1 patient with 3 prior procedures). The mean (SD) time from the most recent hip arthroscopy to the HRA was 2.5 (2.5) years. Table 1 presents a summary of the hip arthroscopy procedures (including only the most recent hip arthroscopy procedure in those with multiple arthroscopies).
Patient demographic variables (age, body mass index [BMI]) were recorded preoperatively, along with the Harris Hip Score (HHS),15 UCLA activity score,16 Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) score,17 and preoperative hip range of motion (flexion, extension, abduction, adduction, internal rotation, and external rotation). The same clinical indices were assessed postoperatively along with the Short Form-12 (SF-12) Health Survey Score,18 at the 6-week, 3-month, 6-month, 1-year, and most recent follow-up visits.
Radiographic assessment consisted of a low anteroposterior (AP) pelvic radiograph (with the radiographic beam centered on the pubic symphysis) and a cross-table lateral radiograph obtained at the most recent follow-up visit. Both the acetabular component abduction relative to the inter-teardrop line, and the angle between the femoral stem and the anatomic axis of the femoral shaft (stem-shaft angle) were measured on AP radiographs.19,20 Acetabular component anteversion was measured on the cross-table lateral radiographs as the angle between the projected long axis of the acetabular opening and a line drawn perpendicular to the long axis plane of the body (Figures A, B).21
The same registry database was used to identify patients who received an HRA without a prior history of arthroscopy or hip surgery. A 1:2 matching analysis for those patients with a prior hip arthroscopy to those without a prior hip arthroscopy was performed to formulate a control group (control cohort) of 86 patients. Each patient in the arthroscopy cohort was matched with 2 patients in the control cohort based on the following parameters: age (± 6 years), sex (same), BMI (± 4 kg/m2), femoral head size (± 4 mm), and preoperative HHS and WOMAC scores (± 7 points). In the event an arthroscopy patient matched to 2 or more control patients, the patients who minimized the least squared error among the matching variables were selected.
Statistical Analysis
All data were collected and analyzed using Microsoft Excel software (Microsoft Corporation, Redmond, Washington). Statistical comparisons between the 2 cohorts regarding demographic variables, clinical outcomes, and radiographic alignment were performed using an unpaired, Student 2-tailed t test, with statistical significance set at P ≤ .05.
Results
A comparison of the results of the 1:2 matching analysis between the arthroscopy and control cohorts is presented in Table 2. There was no significant difference in the preoperative age, BMI, femoral head size, HHS, or WOMAC score between the 2 cohorts. However, the control cohort did show a more severe, preoperative flexion contracture (as expressed by a decreased amount of extension) and a decreased amount of preoperative abduction (Table 3). The preoperative UCLA activity score was also decreased in the control cohort, but this was not statistically significant.
The mean (SD) follow-up was 2.0 (1.0) years in the arthroscopy cohort and 2.1 (1.1) years in the control cohort. There was no significant difference in radiographic alignment between the 2 cohorts. The stem-shaft angle was 139.3° (SD, 5.4°) in the arthroscopy cohort (vs 138.3° [SD, 5.5°] in the control cohort; P = .3), the acetabular abduction was 43.9° (SD, 5.8°) in the arthroscopy cohort (vs 42.9° [SD, 6.1°] in the control cohort; P = .4), and the acetabular anteversion was 21.1° (SD, 7.5°) in the arthroscopy cohort (vs 20.8° [SD, 7.1°] in the control cohort; P = .8).
At 6-week follow-up, the arthroscopy cohort showed a significantly decreased WOMAC score compared with the control cohort (72.9 [SD, 15.5] vs 80.5 [SD, 11.8], respectively; P = .05). In addition, there was a trend towards a decreased SF-12 mental component score in the arthroscopy cohort (52.2 [SD, 9.3] vs 56.5 [SD, 7.8] in the control cohort; P = .06). However, none of the remaining clinical indices showed a significant difference between the 2 cohorts, and there was no difference in range of motion between the 2 cohorts at the 6-week follow-up visit (Table 4).
In addition, at 3-month follow-up, no statistically significant differences were seen between the 2 cohorts for any of the clinical indices or range of motion values. Both groups continued to improve rapidly, with HHS of 96.9 (SD, 3.5) in the arthroscopy cohort and 95.5 (SD, 6.6) in the control cohort, and WOMAC scores of 88.7 (SD, 10.2) and 89.5 (SD, 9.8), respectively (Table 5). Similarly, at the 6-month and 1-year follow-up intervals, the 2 cohorts showed continued improvement in their clinical measures, with no statistically significant differences between the 2 cohorts (Tables 6, 7).
At the most recent follow-up visit, more than 1 year after surgery, the HHS was 99.5 (SD, 1.3) in the arthroscopy cohort and 99.2 (SD, 9.7) in the control cohort (P = .9), and the WOMAC score was 93.5 (SD, 11.3) and 92.4 (SD, 12.2), respectively (P = .8). No significant perioperative complications were seen in the arthroscopy cohort. In the arthroscopy cohort, 1 patient was diagnosed with a deep venous thrombosis 2 weeks after the procedure and was placed on low-molecular-weight heparin and coumadin for treatment. A second patient in the arthroscopy cohort had continued serosanguinous drainage for 4 days postoperatively, which resolved with continued compressive dressings. To date, no patients in the arthroscopy or control cohorts have required a second operation or revision of their components.
Discussion
Given the increasing prevalence of hip arthroscopies to treat multiple disorders of the native joint, it is important to assess the potential consequences of these procedures on future arthroplasties. Piedade and colleagues,13 in a retrospective review of 1474 primary total knee arthroplasties, showed a prior bony procedure (high tibial osteotomy, tibial plateau fracture, patellar realignment) to be predictive of decreased range of motion postoperatively. In addition, a prior knee arthroscopy was associated with a higher rate of postoperative complications, with 30% of the complications requiring a reoperation, and 8.3% of the complications requiring a revision total knee arthroplasty. Kaplan-Meier survival curves showed a survival rate of only 86.8% in those patients with a prior knee arthroscopy (vs 98.1% in those without a prior knee surgery).22 Therefore, the purpose of this study was to evaluate the clinical outcomes of a series of patients who received an HRA after a prior hip arthroscopy. After the initial 6-week follow-up visit, no significant difference was seen in the functional outcomes between those patients with or without a history of prior hip arthroscopy who received an HRA.
After analysis of patient outcomes using multiple clinical measurement tools, at 6-week, 3-month, 6-month, 1-year, and most recent follow-up intervals, the only significant difference between the 2 cohorts was the WOMAC score at 6-week follow-up. Interestingly, there was no significant difference seen in the other clinical assessments, including the SF-12 score, HHS, range of motion, or UCLA activity score (although this did trend towards significance). This can be explained by the difference in both the mode of administration and various metrics assessed by these instruments. In comparison to the HHS evaluation, the patient completes the WOMAC (rather than the clinician) and also provides a more detailed assessment of symptoms, pain, stiffness, and activities of daily living.17 Therefore, this study suggests that patients with a prior hip arthroscopy may require more time to return to their activities of daily living after an HRA. However, whether the statistically significant difference between the 2 scores translates into a clinically significant difference can be questioned.
The clinical outcomes of this series of patients were excellent at the short-term follow-up, and both groups achieved clinical results comparable to prior reported results of HRA.1,10,23,24 However, despite these results, there are several limitations to this study. First, longer-term follow-up is required to determine if any significant differences (such as aseptic loosening, infection, and prosthesis survival) are associated with a prior hip arthroscopy. In addition, this study included a relatively small cohort of patients who had a prior hip arthroscopy. However, a relatively large, single-surgeon database of 1357 HRAs was reviewed, with only 51 cases being reported (3.7%). With the increasing popularity of hip arthroscopy, the number of patients presenting for HRA will likely continue to increase. However, despite these limitations, this study shows that a prior hip arthroscopy does not appear to affect the short-term, clinical outcomes of a metal-on-metal HRA.
1. Amstutz HC, Beaulé PE, Dorey FJ, Le Duff MJ, Campbell PA, Gruen TA. Metal-on-metal hybrid surface arthroplasty. Surgical Technique. J Bone Joint Surg Am. 2006;88(suppl 1 Pt 2):234-249.
2. Daniel J, Pynsent PB, McMinn DJ. Metal-on-metal resurfacing of the hip in patients under the age of 55 years with osteoarthritis. J Bone Joint Surg Br. 2004;86(2):177-184.
3. Pollard TC, Baker RP, Eastaugh-Waring SJ, Bannister GC. Treatment of the young active patient with osteoarthritis of the hip. A five- to seven-year comparison of hybrid total hip arthroplasty and metal-on-metal resurfacing. J Bone Joint Surg Br. 2006;88(5):592-600.
4. Treacy RB, McBryde CW, Pynsent PB. Birmingham hip resurfacing arthroplasty. A minimum follow-up of five years. J Bone Joint Surg Br. 2005;87(2):167-170.
5. Amstutz HC, Le Duff MJ, Campbell PA, Gruen TA, Wisk LE. Clinical and radiographic results of metal-on-metal hip resurfacing with a minimum ten-year follow-up. J Bone Joint Surg Am. 2010;92(16):2663-2671.
6. Daniel J, Ziaee H, Pradhan C, Pynsent PB, McMinn DJ. Blood and urine metal ion levels in young and active patients after Birmingham hip resurfacing arthroplasty: four-year results of a prospective longitudinal study.
J Bone Joint Surg Br. 2007;89(2):169-173.
7. deSouza RM, Parsons NR, Oni T, Dalton P, Costa M, Krikler S. Metal ion levels following resurfacing arthroplasty of the hip: serial results over a ten-year period. J Bone Joint Surg Br. 2010;92(12):1642-1647.
8. Kwon YM, Thomas P, Summer B, et al. Lymphocyte proliferation responses in patients with pseudotumors following metal-on-metal hip resurfacing arthroplasty. J Orthop Res. 2010;28(4):444-450.
9. Australian Orthopaedic Association National Joint Replacement Registry. Annual Report 2011. Adelaide: Australian Orthopaedic Association; 2011. https://aoanjrr.dmac.adelaide.edu.au/annual-reports-2011. Accessed September 16, 2014.
10. Coulter G, Young DA, Dalziel RE, Shimmin AJ. Birmingham hip resurfacing at a mean of ten years: results from an independent centre. J Bone Joint Surg Br. 2012;94(3):315-321.
11. McCarthy JC, Jarrett BT, Ojeifo O, Lee JA, Bragdon CR. What factors influence long-term survivorship after hip arthroscopy? Clin Orthop. 2011;469(2):362-371.
12. Bajwa AS, Villar RN. Arthroscopy of the hip in patients following joint replacement. J Bone Joint Surg Br. 2011;93(7):890-896.
13. Piedade SR, Pinaroli A, Servien E, Neyret P. Is previous knee arthroscopy related to worse results in primary total knee arthroplasty? Knee Surg Sports Traumatol Arthrosc. 2009;17(4):328-333.
14. Gonzalez Della Valle A, Serota A, Go G, et al. Venous thromboembolism is rare with a multimodal prophylaxis protocol after total hip arthroplasty. Clin Orthop. 2006;(444):146-153.
15. Harris WH. Traumatic arthritis of the hip after dislocation and acetabular fractures: treatment by mold arthroplasty. An end-result study using a new method of result evaluation. J Bone Joint Surg Am. 1969;51(4):737-755.
16. Kershaw CJ, Atkins RM, Dodd CA, Bulstrode CJ. Revision total hip arthroplasty for aseptic failure. A review of 276 cases. J Bone Joint Surg Br. 1991;73(4):564-568.
17. Bellamy N. WOMAC: a 20-year experiential review of a patient-centered self-reported health status questionnaire. J Rheumatol. 2002;29(12):2473-2476.
18. Ware J Jr, Kosinski M, Keller SD. A 12-Item Short-Form Health Survey: construction of scales and preliminary tests of reliability and validity. Med Care. 1996;34(3):220-233.
19. Clark JM, Freeman MA, Witham D. The relationship of neck orientation to the shape of the proximal femur. J Arthroplasty. 1987;2(2):99-109.
20. Lewinnek GE, Lewis JL, Tarr R, Compere CL, Zimmerman JR. Dislocations after total hip-replacement arthroplasties. J Bone Joint Surg Am. 1978;60(2):217-220.
21. Yao L, Yao J, Gold RH. Measurement of acetabular version on the axiolateral radiograph. Clin Orthop. 1995;(316):106-111.
22. Piedade SR, Pinaroli A, Servien E, Neyret P. TKA outcomes after prior bone and soft tissue knee surgery. Knee Surg Sports Traumatol Arthrosc. 2013;21(12):2737-2743.
23. Amstutz HC, Beaulé PE, Dorey FJ, Le Duff MJ, Campbell PA, Gruen TA. Metal-on-metal hybrid surface arthroplasty: two to six-year follow-up study. J Bone Joint Surg Am. 2004;86(1):28-39.
24. Steffen RT, Pandit HP, Palan J, et al. The five-year results of the Birmingham Hip Resurfacing arthroplasty: an independent series. J Bone Joint Surg Br. 2008;90(4):436-441.
1. Amstutz HC, Beaulé PE, Dorey FJ, Le Duff MJ, Campbell PA, Gruen TA. Metal-on-metal hybrid surface arthroplasty. Surgical Technique. J Bone Joint Surg Am. 2006;88(suppl 1 Pt 2):234-249.
2. Daniel J, Pynsent PB, McMinn DJ. Metal-on-metal resurfacing of the hip in patients under the age of 55 years with osteoarthritis. J Bone Joint Surg Br. 2004;86(2):177-184.
3. Pollard TC, Baker RP, Eastaugh-Waring SJ, Bannister GC. Treatment of the young active patient with osteoarthritis of the hip. A five- to seven-year comparison of hybrid total hip arthroplasty and metal-on-metal resurfacing. J Bone Joint Surg Br. 2006;88(5):592-600.
4. Treacy RB, McBryde CW, Pynsent PB. Birmingham hip resurfacing arthroplasty. A minimum follow-up of five years. J Bone Joint Surg Br. 2005;87(2):167-170.
5. Amstutz HC, Le Duff MJ, Campbell PA, Gruen TA, Wisk LE. Clinical and radiographic results of metal-on-metal hip resurfacing with a minimum ten-year follow-up. J Bone Joint Surg Am. 2010;92(16):2663-2671.
6. Daniel J, Ziaee H, Pradhan C, Pynsent PB, McMinn DJ. Blood and urine metal ion levels in young and active patients after Birmingham hip resurfacing arthroplasty: four-year results of a prospective longitudinal study.
J Bone Joint Surg Br. 2007;89(2):169-173.
7. deSouza RM, Parsons NR, Oni T, Dalton P, Costa M, Krikler S. Metal ion levels following resurfacing arthroplasty of the hip: serial results over a ten-year period. J Bone Joint Surg Br. 2010;92(12):1642-1647.
8. Kwon YM, Thomas P, Summer B, et al. Lymphocyte proliferation responses in patients with pseudotumors following metal-on-metal hip resurfacing arthroplasty. J Orthop Res. 2010;28(4):444-450.
9. Australian Orthopaedic Association National Joint Replacement Registry. Annual Report 2011. Adelaide: Australian Orthopaedic Association; 2011. https://aoanjrr.dmac.adelaide.edu.au/annual-reports-2011. Accessed September 16, 2014.
10. Coulter G, Young DA, Dalziel RE, Shimmin AJ. Birmingham hip resurfacing at a mean of ten years: results from an independent centre. J Bone Joint Surg Br. 2012;94(3):315-321.
11. McCarthy JC, Jarrett BT, Ojeifo O, Lee JA, Bragdon CR. What factors influence long-term survivorship after hip arthroscopy? Clin Orthop. 2011;469(2):362-371.
12. Bajwa AS, Villar RN. Arthroscopy of the hip in patients following joint replacement. J Bone Joint Surg Br. 2011;93(7):890-896.
13. Piedade SR, Pinaroli A, Servien E, Neyret P. Is previous knee arthroscopy related to worse results in primary total knee arthroplasty? Knee Surg Sports Traumatol Arthrosc. 2009;17(4):328-333.
14. Gonzalez Della Valle A, Serota A, Go G, et al. Venous thromboembolism is rare with a multimodal prophylaxis protocol after total hip arthroplasty. Clin Orthop. 2006;(444):146-153.
15. Harris WH. Traumatic arthritis of the hip after dislocation and acetabular fractures: treatment by mold arthroplasty. An end-result study using a new method of result evaluation. J Bone Joint Surg Am. 1969;51(4):737-755.
16. Kershaw CJ, Atkins RM, Dodd CA, Bulstrode CJ. Revision total hip arthroplasty for aseptic failure. A review of 276 cases. J Bone Joint Surg Br. 1991;73(4):564-568.
17. Bellamy N. WOMAC: a 20-year experiential review of a patient-centered self-reported health status questionnaire. J Rheumatol. 2002;29(12):2473-2476.
18. Ware J Jr, Kosinski M, Keller SD. A 12-Item Short-Form Health Survey: construction of scales and preliminary tests of reliability and validity. Med Care. 1996;34(3):220-233.
19. Clark JM, Freeman MA, Witham D. The relationship of neck orientation to the shape of the proximal femur. J Arthroplasty. 1987;2(2):99-109.
20. Lewinnek GE, Lewis JL, Tarr R, Compere CL, Zimmerman JR. Dislocations after total hip-replacement arthroplasties. J Bone Joint Surg Am. 1978;60(2):217-220.
21. Yao L, Yao J, Gold RH. Measurement of acetabular version on the axiolateral radiograph. Clin Orthop. 1995;(316):106-111.
22. Piedade SR, Pinaroli A, Servien E, Neyret P. TKA outcomes after prior bone and soft tissue knee surgery. Knee Surg Sports Traumatol Arthrosc. 2013;21(12):2737-2743.
23. Amstutz HC, Beaulé PE, Dorey FJ, Le Duff MJ, Campbell PA, Gruen TA. Metal-on-metal hybrid surface arthroplasty: two to six-year follow-up study. J Bone Joint Surg Am. 2004;86(1):28-39.
24. Steffen RT, Pandit HP, Palan J, et al. The five-year results of the Birmingham Hip Resurfacing arthroplasty: an independent series. J Bone Joint Surg Br. 2008;90(4):436-441.
The Role of Computed Tomography for Postoperative Evaluation of Percutaneous Sacroiliac Screw Fixation and Description of a “Safe Zone”
Pelvic injuries account for 3% of all skeletal fractures.1 Injury to the sacroiliac (SI) joint is frequently associated with unstable pelvic ring fractures, which are potentially life-threatening injuries. Surgical fixation of these injuries is preferred to nonoperative treatment given the potential for improved reduction and early mobilization and weight-bearing, thereby decreasing perioperative morbidity and improving functional outcome.2
The classic method of surgical fixation of the SI joint consisted of open reduction and internal fixation. This method carried a substantial risk for large dissection, iatrogenic nerve injury, and increased blood loss to the already traumatized patient.3 Percutaneous fixation allows for a shorter operating time, decreased soft-tissue stripping, and decreased blood loss compared with a traditional open procedure.4 However, posterior pelvic anatomy is complex and variable, and reports have found screw misplacements as high as 24%5 and neurologic complication rates up to 18%.6-9
Various imaging modalities, including fluoroscopy,5 computed tomography (CT),6-7 fluoroscopic CT, and computer-assisted techniques5,9 have been used to achieve proper screw placement. Conventional fluoroscopy is the standard for intraoperative screw placement. However, acceptable reduction of the SI joint and proper implantation of the screws without perforation of the neural foramina is challenging, especially when coupled with difficulties of fluoroscopic imaging and variations in pelvic anatomy.
Sacral dysplasia has been reported to occur in up to 20% to 40% of the population and has significant implications in patients indicated for iliosacral screw placement.10 Incorrect placement of iliosacral screws may result in iatrogenic neurovascular complications.11-13 Malpositioned screws using fluoroscopic guidance have been reported in 2% to 15% of patients with an incidence of neurologic compromise between 0.5% and 7.7%. As little as 4° of misdirection can result in damage to neurovascular structures.14
At our institution, we routinely obtained postoperative CT to evaluate the placement of SI screws. The objective of this retrospective study is to evaluate the rate of revision surgery of percutaneous SI screw fixation, to determine whether CT is an accurate tool for evaluation of the reduction and the need for revision surgery, and to decide if any violation of the neural foramina is safe.
Materials and Methods
After institutional review board approval, we retrospectively reviewed and evaluated medical records and radiographs of all patients who sustained unstable pelvic ring fractures between July 1, 2005, and June 30, 2010. We identified all patients who were treated with closed reductions and percutaneous iliosacral screw fixation, according to the method described by Routt in 1995.4 We excluded all pelvic fractures in patients who underwent open reduction for the posterior injury or did not have percutaneous SI screws placed, those with spinal injury, and those without follow-up. Of the 46 patients who met the inclusion criteria were 26 men and 20 women with a mean age of 42 years (range, 16 to 73 years). Motor vehicle accidents accounted for 13 cases; 19 were crush injuries and 14 were falls from height. Seventeen patients (37%) met the radiographic criteria for sacral dysmorphism. Forty-two of the 46 patients were polytrauma patients with associated musculoskeletal injuries and/or abdominal, chest, or head injuries.
Six patients presented with some neurologic deficit at the time of injury; all fractures were closed. The initial imaging study included plain anteroposterior (AP), inlet, and outlet radiographs of the pelvis and a pelvic CT scan. Using the classification of Young and Burgess,15 there were 3 vertical shear injuries, 13 lateral compression–type injuries, 17 anterior-posterior–type injuries, 7 sacral fractures, and 6 combination- or unclassifiable-type pelvic injuries. Of the sacral fractures, there were 3 Denis zone 1, 3 Denis zone 2, and 1 Denis zone 3.
The pelvic CT scan included the entire pelvis from the ilium to the ischial tuberosities. Each scan consisted of either a 5.0-mm or a 2.5-mm sequential axial image. A picture archiving and communication system (PACS) workstation using Centricity version 2.1 (GE Medical Systems, Waukesha, Wisconsin) was used to analyze each scan with a bone algorithm. On PACS, each initial displacement was characterized by the amount of SI joint widening at the level of the S1 and was measured using digital calipers.
Surgery
Mean time to surgery was 4 days (range, 2 to 15 days) after the injury. A total of 51 SI screws were implanted in 46 patients. We achieved closed reduction of the posterior pelvic ring by various techniques, including compression with percutaneous partially threaded screw fixation. In the cases in which the posterior ring lesion was associated with a pure pubic symphysis disruption, the anterior pelvis was initially reduced and stabilized with small-fragment plate fixation (Synthes, Inc, Paoli, Pennsylvania). The posterior complex was stabilized with 1 screw in 41 patients, 2 cases required a transiliac screw, and 2 screws (S1 and S2) were placed in each of the remaining 3 cases. Definitive stabilization of the posterior pelvis was achieved with percutaneous, partially threaded 7.3- or 7.5-mm–diameter cannulated screws (Synthes, Inc, and Zimmer Inc, Warsaw, Indiana, respectively) in 42 fractures and 6.5-mm screws (Synthes, Inc) in 4 fractures. In 11 cases where the fracture was through the sacrum, fully threaded cannulated screws were used to avoid compression. Screw insertion was performed under fluoroscopic guidance with inlet, outlet, and lateral sacral views. One of 2 fellowship-trained trauma surgeons performed the surgeries. Rehabilitation plans were customized to each patient based on concomitant injuries.
Postoperative Assessment
AP, lateral sacral, and inlet and outlet postoperative radiographs were taken in all cases within 24 hours after surgery. Pelvic CT was also obtained within 24 hours of surgery to review reduction and screw placement.
Using the measurement tool on the PACS system, we measured the penetration of the screw into the foramen. Screws were graded as intraosseous (completely contained within the sacral bone), skived (less than 2 mm of partial penetration into the S1 foramen), or extruded (the screw not contained by the bone). Screw penetration of the S1 was evaluated on the radiographic images as well as the axial images of the CT scans.
After surgery, the senior orthopedic resident and attending surgeon performed and documented detailed neurologic evaluations. They reviewed the medical record for neurologic deficit following surgical fixation.
Results
The mean follow-up time was 12 months (range, 8 months to 2 years). Two patients expired secondary to associated injuries. There were no early deaths related to the pelvic surgery. Stable fixation, including bone or ligamentous healing, as well as full weight-bearing status, was noted in every case. No case exhibited loss of reduction or implant failure or infection.
According to Matta’s criteria of anatomic reduction within 1 cm, all patients were found to have satisfactory reductions.7 Six of 46 patients had documented preoperative neurologic deficits. After percutaneous screw fixation, 10 of 46 patients had postoperative neurologic deficit, 2 of which were unchanged from preoperative evaluation. Of the 8 patients with new/altered postoperative neurologic deficit, CT showed neural foramen penetration greater than 2.1 mm in only 2 patients. Both patients underwent screw revision, resulting in improved neurologic deficit. The remaining 4 patients did not have foramen penetration and improved their neurologic function over the course of 2 weeks with return to presurgical status by 6 weeks without necessitating screw removal.
Twenty-three of the 51 screws (45%) had some violation of the S1 foramen on the CT. There were 17 patients with dysmorphic sacrums in which 21 S1 screws were placed. Eleven of 21 (52%) screws showed some penetration of the S1 foramen on CT. There were 29 patients with normal sacral morphology in which 30 S1 screws were placed. Twelve of 30 (40%) screws penetrated the S1 foramen. All violations were in the superior one-third position of the foramen. Two of 46 (4%; 1 with dysmorphism, 1 without) had a new neurologic deficit associated with the surgery (Table). CT showed sacral foramen penetration, and both screws were revised with a better neurologic examination.
High-resolution CTs were obtained in 32 patients, while 14 patients underwent the standard 5.0-mm–cut CTs. Of the 32 patients in which a 2.5-mm high-resolution CT was obtained, 20 (62.5%) had evidence of screw penetration (Figures 1, 2). All violations of the S1 neural foramen were in the superior portion of the foramen.
When compared with patients who had a 5.0-mm CT, the patients who underwent a high-resolution CT were more likely to show neural foramen penetration (P = .3). The average screw penetration into the S1 neural foramen measured 3.3 mm (range, 1.6-5.7 mm) in dysmorphic sacrum and 2.7 mm (range, 1.4-7 mm) in normal sacrum. However, in our study, any foramen penetration of less than 2.1 mm on CT did not result in neurologic deficit.
Discussion
Pelvic fractures are fairly common and represent approximately 5% of all trauma admissions and 3% of all skeletal fractures nationwide.1 The current treatment for SI disruption is either nonoperative or operative. Surgical fixation is technically demanding and surgeons often need a long learning curve to acquire the demanding technique because of the limitations of radiographic visualization of the relevant landmarks.16
Letournel17 developed the technique for iliosacral screw fixation for the treatment of posterior pelvic ring injuries, where 1 or 2 large screws (6.5-7.3 mm in diameter) are inserted under fluoroscopic guidance through the ilium, across the SI articulation, and into the superior sacral vertebral bodies using percutaneous techniques. Currently, the standard procedure to accomplish the percutaneous placement of iliosacral screws derives mainly from the technique described by Matta with the C-arm fluoroscopy visualizing the pelvis in 3 views: strict AP, inlet, and outlet views.7
Routt and colleagues4 recommend a strict lateral view of the sacrum, particularly when crossing the narrow zone of the sacral alar. They reported high union rates and accurate placement of the screws.4 There are limitations to the use of biplanar fluoroscopy because the intraoperative images are not orthogonal, with the average arc (67º) between the ideal inlet and outlet. However, because of the variability in sacral anatomy, CT guidance was recommended by others.2,6,8,18 Operating in a CT suite had other complications. Misinterpretation of CT led to “in-out-in” screws, which resulted in neurapraxia.
In our study, we used the technique described by Matta and colleagues for placement of the screws and performed a postoperative CT to evaluate screw placement and to assess pelvic reduction.7 We had a high penetration rate using CT, which increased with better resolution, even though none of the radiographs showed any obvious evidence of misplacement of the screws. Ebraheim and colleagues6 described the relationship of the S1 nerve root in its neural foramen and found it to be approximately 8.7 mm inferior and 7.8 mm medial to the starting point for a pedicle screw. Given these numbers, it is possible that a large amount of skiving can be tolerated contingent on an adequate reduction of the SI joint.
Because of our high rates of skiving and low rates of neurologic deficit, a new “safe zone” for screw insertion can be expanded to include skiving of the S1 neural foramen up to 3 mm without fear of nerve root injury. However, drilling and screw insertion at higher speeds can also cause neurologic injury secondary to thermal injury or soft tissue being caught up in a rotating drill/screw.
Evaluation of placement of percutaneous SI screw placement in our study resulted in neural foramen penetration in 43% of SI screws, which is higher than other studies.14,19,20 Our study showed that screw penetration up to 2 mm does not correlate with neurologic deficit. Iatrogenic neurologic deficit secondary to perforation of the foramina occurred in only 1 patient. Penetration of the foramina in all cases was in the superior portion of the foramen. We propose that there is a safe zone within the S1 neural foramen, and small amounts of penetration in the superior one-third of the foramen on axial CT images do not correlate with neurologic deficit. This potential safe zone is predicated on adequate reduction of the SI joint.
Neural foramen penetration shown on postoperative CT does not necessarily correlate with neurologic deficit. A postoperative CT is not indicated unless there are findings of a postoperative nerve injury. Our ideal screw placement skives the superior S1 foramen allowing for a larger screw diameter in a safe zone.
CT-guided placement has been proposed; however, concerns about radiation exposure, cost, and feasibility with similar outcomes compared with fluoroscopic-guided screw placement has resulted in its falling out of favor.
Iatrogenic nerve injuries are reported to occur in 0% to 6% of all percutaneous SI screw placement.14,21 Risk factors for iatrogenic nerve injury while using fluoroscopic guidance include sacral morphologic abnormalities, presence of intestinal gas, or contrast.22 Although these may be minimized with proper use of fluoroscopy, obtaining anatomic reduction as well as a thorough understanding of the pelvic morphology, the surgeon must be prepared to obtain further studies, such as a CT scan, if there is postoperative neurologic deficit.
Based on our findings, we do not routinely obtain a postoperative CT for SI screw placement, unless there is concern for malreduction or there is neurologic deficit. We also believe that up to 2 mm of foramen penetration is safe and does not result in neurologic deficit.
1. Failinger MS, McGanity PL. Unstable fractures of the pelvic ring. J Bone and Joint Surg Am. 1992;74(5):781-791.
2. Smith HE, Yuan PS, Sasso R, Papadopolous S, Vaccaro AR. An evaluation of image-guided technologies in the placement of percutaneous iliosacral screws. Spine (Phila Pa 1976). 2006;31(2):234-238.
3. Judet R, Judet J, Letournel E. Fractures of the acetabulum: classification and surgical approaches for open reduction. Preliminary report. J Bone Joint Surg Am. 1964;46(16):1615-1646.
4. Routt ML Jr, Kregor PJ, Simonian PT, Mayo KA. Early results of percutaneous iliosacral screws placed with the patient in the supine position. J Orthop Trauma. 1995;9(3):207-214.
5. Tonetti J, Carrat L, Blendea S, et al. Clinical results of percutaneous pelvic surgery. Computer assisted surgery using ultrasound compared to standard fluoroscopy. Comput Aided Surg. 2001;6(4):204-211.
6. Ebraheim NA, Coombs R, Jackson WT, Rusin JJ. Percutaneous computed tomography-guided stabilization of posterior pelvic fractures. Clin Orthop. 1994;(307):222-228.
7. Keating JF, Werier J, Blachut P, et al. Early fixation of the vertically unstable pelvis: the role of iliosacral screw fixation of the posterior lesion. J Orthop Trauma. 1999;13(2):107-113.
8. Webb LX, de Araujo W, Donofrio P, et al. Electromyography monitoring for percutaneous placement of iliosacral screws. J Orthop Trauma. 2000;14(4):245-254.
9. Barrick EF, O’Mara JW, Lane HE 3rd. Iliosacral screw insertion using computer-assisted CT image guidance: a laboratory study. Comput Aided Surg. 1998;3(6):289-296.
10. Routt ML Jr, Simonian PT, Agnew SG, Mann FA. Radiographic recognition of the sacral alar slope for optimal placement of iliosacral screws: a cadaveric and clinical study. J Orthop Trauma. 1996;10(3):171-177.
11. Altman DT, Jones CB, Routt ML Jr. Superior gluteal artery injury during iliosacral screw placement. J Orthop Trauma. 1999;13(3):220-227.
12. Stephen DJ. Pseudoaneurysm of the superior gluteal arterial system: an unusual cause of pain after a pelvic fracture. J Trauma. 1997;43(1):146-149.
13. Stöckle U, König B, Hofstetter R, Nolte LP, Haas NP. [Navigation assisted by image conversion. An experimental study on pelvic screw fixation]
[in German]. Unfallchirurg. 2001;104(3):215-220.
14. Templeman D, Schmidt A, Freese J, Weisman I, et al. Proximity of iliosacral screws to neurovascular structures after internal fixation. Clin Orthop. 1996;(329):194-198.
15. Young JW, Burgess AR, Brumback RJ, Poka A. Pelvic fractures: value of plain radiography in early assessment and management. Radiology. 1986;160(2):445-451.
16. Graves ML, Routt ML Jr. Iliosacral screw placement: are uniplanar changes realistic based on standard fluoroscopic imaging? J Trauma. 2011;7(1):204-208.
17. Letournel E. Pelvic fractures. Injury. 1978;10(2):145-148.
18. Blake-Toker AM, Hawkins L, Nadalo L, et al. CT guided percutaneous fixation of sacroiliac fractures in trauma patients. J Trauma. 2001;51(6):1117-1121.
19. Hinsche AF, Giannoudis PV, Smith RM. Fluoroscopy-based multiplanar image guidance for insertion of sacroiliac screws. Clin Orthop. 2002;(395):135-144.
20. van den Bosch EW, van Zwienen CM, van Vugt AB. Fluoroscopic positioning of sacroiliac screws in 88 patients. J Trauma. 2002;53(1):44-48.
21. Cole JD, Blum DA, Ansel LJ. Outcome after fixation of unstable posterior pelvic ring injuries. Clin Orthop. 1996;(329):160-179.
22. Routt ML Jr, Simonian PT. Closed reduction and percutaneous skeletal fixation of sacral fractures. Clin Orthop. 1996;(329):121-128.
Pelvic injuries account for 3% of all skeletal fractures.1 Injury to the sacroiliac (SI) joint is frequently associated with unstable pelvic ring fractures, which are potentially life-threatening injuries. Surgical fixation of these injuries is preferred to nonoperative treatment given the potential for improved reduction and early mobilization and weight-bearing, thereby decreasing perioperative morbidity and improving functional outcome.2
The classic method of surgical fixation of the SI joint consisted of open reduction and internal fixation. This method carried a substantial risk for large dissection, iatrogenic nerve injury, and increased blood loss to the already traumatized patient.3 Percutaneous fixation allows for a shorter operating time, decreased soft-tissue stripping, and decreased blood loss compared with a traditional open procedure.4 However, posterior pelvic anatomy is complex and variable, and reports have found screw misplacements as high as 24%5 and neurologic complication rates up to 18%.6-9
Various imaging modalities, including fluoroscopy,5 computed tomography (CT),6-7 fluoroscopic CT, and computer-assisted techniques5,9 have been used to achieve proper screw placement. Conventional fluoroscopy is the standard for intraoperative screw placement. However, acceptable reduction of the SI joint and proper implantation of the screws without perforation of the neural foramina is challenging, especially when coupled with difficulties of fluoroscopic imaging and variations in pelvic anatomy.
Sacral dysplasia has been reported to occur in up to 20% to 40% of the population and has significant implications in patients indicated for iliosacral screw placement.10 Incorrect placement of iliosacral screws may result in iatrogenic neurovascular complications.11-13 Malpositioned screws using fluoroscopic guidance have been reported in 2% to 15% of patients with an incidence of neurologic compromise between 0.5% and 7.7%. As little as 4° of misdirection can result in damage to neurovascular structures.14
At our institution, we routinely obtained postoperative CT to evaluate the placement of SI screws. The objective of this retrospective study is to evaluate the rate of revision surgery of percutaneous SI screw fixation, to determine whether CT is an accurate tool for evaluation of the reduction and the need for revision surgery, and to decide if any violation of the neural foramina is safe.
Materials and Methods
After institutional review board approval, we retrospectively reviewed and evaluated medical records and radiographs of all patients who sustained unstable pelvic ring fractures between July 1, 2005, and June 30, 2010. We identified all patients who were treated with closed reductions and percutaneous iliosacral screw fixation, according to the method described by Routt in 1995.4 We excluded all pelvic fractures in patients who underwent open reduction for the posterior injury or did not have percutaneous SI screws placed, those with spinal injury, and those without follow-up. Of the 46 patients who met the inclusion criteria were 26 men and 20 women with a mean age of 42 years (range, 16 to 73 years). Motor vehicle accidents accounted for 13 cases; 19 were crush injuries and 14 were falls from height. Seventeen patients (37%) met the radiographic criteria for sacral dysmorphism. Forty-two of the 46 patients were polytrauma patients with associated musculoskeletal injuries and/or abdominal, chest, or head injuries.
Six patients presented with some neurologic deficit at the time of injury; all fractures were closed. The initial imaging study included plain anteroposterior (AP), inlet, and outlet radiographs of the pelvis and a pelvic CT scan. Using the classification of Young and Burgess,15 there were 3 vertical shear injuries, 13 lateral compression–type injuries, 17 anterior-posterior–type injuries, 7 sacral fractures, and 6 combination- or unclassifiable-type pelvic injuries. Of the sacral fractures, there were 3 Denis zone 1, 3 Denis zone 2, and 1 Denis zone 3.
The pelvic CT scan included the entire pelvis from the ilium to the ischial tuberosities. Each scan consisted of either a 5.0-mm or a 2.5-mm sequential axial image. A picture archiving and communication system (PACS) workstation using Centricity version 2.1 (GE Medical Systems, Waukesha, Wisconsin) was used to analyze each scan with a bone algorithm. On PACS, each initial displacement was characterized by the amount of SI joint widening at the level of the S1 and was measured using digital calipers.
Surgery
Mean time to surgery was 4 days (range, 2 to 15 days) after the injury. A total of 51 SI screws were implanted in 46 patients. We achieved closed reduction of the posterior pelvic ring by various techniques, including compression with percutaneous partially threaded screw fixation. In the cases in which the posterior ring lesion was associated with a pure pubic symphysis disruption, the anterior pelvis was initially reduced and stabilized with small-fragment plate fixation (Synthes, Inc, Paoli, Pennsylvania). The posterior complex was stabilized with 1 screw in 41 patients, 2 cases required a transiliac screw, and 2 screws (S1 and S2) were placed in each of the remaining 3 cases. Definitive stabilization of the posterior pelvis was achieved with percutaneous, partially threaded 7.3- or 7.5-mm–diameter cannulated screws (Synthes, Inc, and Zimmer Inc, Warsaw, Indiana, respectively) in 42 fractures and 6.5-mm screws (Synthes, Inc) in 4 fractures. In 11 cases where the fracture was through the sacrum, fully threaded cannulated screws were used to avoid compression. Screw insertion was performed under fluoroscopic guidance with inlet, outlet, and lateral sacral views. One of 2 fellowship-trained trauma surgeons performed the surgeries. Rehabilitation plans were customized to each patient based on concomitant injuries.
Postoperative Assessment
AP, lateral sacral, and inlet and outlet postoperative radiographs were taken in all cases within 24 hours after surgery. Pelvic CT was also obtained within 24 hours of surgery to review reduction and screw placement.
Using the measurement tool on the PACS system, we measured the penetration of the screw into the foramen. Screws were graded as intraosseous (completely contained within the sacral bone), skived (less than 2 mm of partial penetration into the S1 foramen), or extruded (the screw not contained by the bone). Screw penetration of the S1 was evaluated on the radiographic images as well as the axial images of the CT scans.
After surgery, the senior orthopedic resident and attending surgeon performed and documented detailed neurologic evaluations. They reviewed the medical record for neurologic deficit following surgical fixation.
Results
The mean follow-up time was 12 months (range, 8 months to 2 years). Two patients expired secondary to associated injuries. There were no early deaths related to the pelvic surgery. Stable fixation, including bone or ligamentous healing, as well as full weight-bearing status, was noted in every case. No case exhibited loss of reduction or implant failure or infection.
According to Matta’s criteria of anatomic reduction within 1 cm, all patients were found to have satisfactory reductions.7 Six of 46 patients had documented preoperative neurologic deficits. After percutaneous screw fixation, 10 of 46 patients had postoperative neurologic deficit, 2 of which were unchanged from preoperative evaluation. Of the 8 patients with new/altered postoperative neurologic deficit, CT showed neural foramen penetration greater than 2.1 mm in only 2 patients. Both patients underwent screw revision, resulting in improved neurologic deficit. The remaining 4 patients did not have foramen penetration and improved their neurologic function over the course of 2 weeks with return to presurgical status by 6 weeks without necessitating screw removal.
Twenty-three of the 51 screws (45%) had some violation of the S1 foramen on the CT. There were 17 patients with dysmorphic sacrums in which 21 S1 screws were placed. Eleven of 21 (52%) screws showed some penetration of the S1 foramen on CT. There were 29 patients with normal sacral morphology in which 30 S1 screws were placed. Twelve of 30 (40%) screws penetrated the S1 foramen. All violations were in the superior one-third position of the foramen. Two of 46 (4%; 1 with dysmorphism, 1 without) had a new neurologic deficit associated with the surgery (Table). CT showed sacral foramen penetration, and both screws were revised with a better neurologic examination.
High-resolution CTs were obtained in 32 patients, while 14 patients underwent the standard 5.0-mm–cut CTs. Of the 32 patients in which a 2.5-mm high-resolution CT was obtained, 20 (62.5%) had evidence of screw penetration (Figures 1, 2). All violations of the S1 neural foramen were in the superior portion of the foramen.
When compared with patients who had a 5.0-mm CT, the patients who underwent a high-resolution CT were more likely to show neural foramen penetration (P = .3). The average screw penetration into the S1 neural foramen measured 3.3 mm (range, 1.6-5.7 mm) in dysmorphic sacrum and 2.7 mm (range, 1.4-7 mm) in normal sacrum. However, in our study, any foramen penetration of less than 2.1 mm on CT did not result in neurologic deficit.
Discussion
Pelvic fractures are fairly common and represent approximately 5% of all trauma admissions and 3% of all skeletal fractures nationwide.1 The current treatment for SI disruption is either nonoperative or operative. Surgical fixation is technically demanding and surgeons often need a long learning curve to acquire the demanding technique because of the limitations of radiographic visualization of the relevant landmarks.16
Letournel17 developed the technique for iliosacral screw fixation for the treatment of posterior pelvic ring injuries, where 1 or 2 large screws (6.5-7.3 mm in diameter) are inserted under fluoroscopic guidance through the ilium, across the SI articulation, and into the superior sacral vertebral bodies using percutaneous techniques. Currently, the standard procedure to accomplish the percutaneous placement of iliosacral screws derives mainly from the technique described by Matta with the C-arm fluoroscopy visualizing the pelvis in 3 views: strict AP, inlet, and outlet views.7
Routt and colleagues4 recommend a strict lateral view of the sacrum, particularly when crossing the narrow zone of the sacral alar. They reported high union rates and accurate placement of the screws.4 There are limitations to the use of biplanar fluoroscopy because the intraoperative images are not orthogonal, with the average arc (67º) between the ideal inlet and outlet. However, because of the variability in sacral anatomy, CT guidance was recommended by others.2,6,8,18 Operating in a CT suite had other complications. Misinterpretation of CT led to “in-out-in” screws, which resulted in neurapraxia.
In our study, we used the technique described by Matta and colleagues for placement of the screws and performed a postoperative CT to evaluate screw placement and to assess pelvic reduction.7 We had a high penetration rate using CT, which increased with better resolution, even though none of the radiographs showed any obvious evidence of misplacement of the screws. Ebraheim and colleagues6 described the relationship of the S1 nerve root in its neural foramen and found it to be approximately 8.7 mm inferior and 7.8 mm medial to the starting point for a pedicle screw. Given these numbers, it is possible that a large amount of skiving can be tolerated contingent on an adequate reduction of the SI joint.
Because of our high rates of skiving and low rates of neurologic deficit, a new “safe zone” for screw insertion can be expanded to include skiving of the S1 neural foramen up to 3 mm without fear of nerve root injury. However, drilling and screw insertion at higher speeds can also cause neurologic injury secondary to thermal injury or soft tissue being caught up in a rotating drill/screw.
Evaluation of placement of percutaneous SI screw placement in our study resulted in neural foramen penetration in 43% of SI screws, which is higher than other studies.14,19,20 Our study showed that screw penetration up to 2 mm does not correlate with neurologic deficit. Iatrogenic neurologic deficit secondary to perforation of the foramina occurred in only 1 patient. Penetration of the foramina in all cases was in the superior portion of the foramen. We propose that there is a safe zone within the S1 neural foramen, and small amounts of penetration in the superior one-third of the foramen on axial CT images do not correlate with neurologic deficit. This potential safe zone is predicated on adequate reduction of the SI joint.
Neural foramen penetration shown on postoperative CT does not necessarily correlate with neurologic deficit. A postoperative CT is not indicated unless there are findings of a postoperative nerve injury. Our ideal screw placement skives the superior S1 foramen allowing for a larger screw diameter in a safe zone.
CT-guided placement has been proposed; however, concerns about radiation exposure, cost, and feasibility with similar outcomes compared with fluoroscopic-guided screw placement has resulted in its falling out of favor.
Iatrogenic nerve injuries are reported to occur in 0% to 6% of all percutaneous SI screw placement.14,21 Risk factors for iatrogenic nerve injury while using fluoroscopic guidance include sacral morphologic abnormalities, presence of intestinal gas, or contrast.22 Although these may be minimized with proper use of fluoroscopy, obtaining anatomic reduction as well as a thorough understanding of the pelvic morphology, the surgeon must be prepared to obtain further studies, such as a CT scan, if there is postoperative neurologic deficit.
Based on our findings, we do not routinely obtain a postoperative CT for SI screw placement, unless there is concern for malreduction or there is neurologic deficit. We also believe that up to 2 mm of foramen penetration is safe and does not result in neurologic deficit.
Pelvic injuries account for 3% of all skeletal fractures.1 Injury to the sacroiliac (SI) joint is frequently associated with unstable pelvic ring fractures, which are potentially life-threatening injuries. Surgical fixation of these injuries is preferred to nonoperative treatment given the potential for improved reduction and early mobilization and weight-bearing, thereby decreasing perioperative morbidity and improving functional outcome.2
The classic method of surgical fixation of the SI joint consisted of open reduction and internal fixation. This method carried a substantial risk for large dissection, iatrogenic nerve injury, and increased blood loss to the already traumatized patient.3 Percutaneous fixation allows for a shorter operating time, decreased soft-tissue stripping, and decreased blood loss compared with a traditional open procedure.4 However, posterior pelvic anatomy is complex and variable, and reports have found screw misplacements as high as 24%5 and neurologic complication rates up to 18%.6-9
Various imaging modalities, including fluoroscopy,5 computed tomography (CT),6-7 fluoroscopic CT, and computer-assisted techniques5,9 have been used to achieve proper screw placement. Conventional fluoroscopy is the standard for intraoperative screw placement. However, acceptable reduction of the SI joint and proper implantation of the screws without perforation of the neural foramina is challenging, especially when coupled with difficulties of fluoroscopic imaging and variations in pelvic anatomy.
Sacral dysplasia has been reported to occur in up to 20% to 40% of the population and has significant implications in patients indicated for iliosacral screw placement.10 Incorrect placement of iliosacral screws may result in iatrogenic neurovascular complications.11-13 Malpositioned screws using fluoroscopic guidance have been reported in 2% to 15% of patients with an incidence of neurologic compromise between 0.5% and 7.7%. As little as 4° of misdirection can result in damage to neurovascular structures.14
At our institution, we routinely obtained postoperative CT to evaluate the placement of SI screws. The objective of this retrospective study is to evaluate the rate of revision surgery of percutaneous SI screw fixation, to determine whether CT is an accurate tool for evaluation of the reduction and the need for revision surgery, and to decide if any violation of the neural foramina is safe.
Materials and Methods
After institutional review board approval, we retrospectively reviewed and evaluated medical records and radiographs of all patients who sustained unstable pelvic ring fractures between July 1, 2005, and June 30, 2010. We identified all patients who were treated with closed reductions and percutaneous iliosacral screw fixation, according to the method described by Routt in 1995.4 We excluded all pelvic fractures in patients who underwent open reduction for the posterior injury or did not have percutaneous SI screws placed, those with spinal injury, and those without follow-up. Of the 46 patients who met the inclusion criteria were 26 men and 20 women with a mean age of 42 years (range, 16 to 73 years). Motor vehicle accidents accounted for 13 cases; 19 were crush injuries and 14 were falls from height. Seventeen patients (37%) met the radiographic criteria for sacral dysmorphism. Forty-two of the 46 patients were polytrauma patients with associated musculoskeletal injuries and/or abdominal, chest, or head injuries.
Six patients presented with some neurologic deficit at the time of injury; all fractures were closed. The initial imaging study included plain anteroposterior (AP), inlet, and outlet radiographs of the pelvis and a pelvic CT scan. Using the classification of Young and Burgess,15 there were 3 vertical shear injuries, 13 lateral compression–type injuries, 17 anterior-posterior–type injuries, 7 sacral fractures, and 6 combination- or unclassifiable-type pelvic injuries. Of the sacral fractures, there were 3 Denis zone 1, 3 Denis zone 2, and 1 Denis zone 3.
The pelvic CT scan included the entire pelvis from the ilium to the ischial tuberosities. Each scan consisted of either a 5.0-mm or a 2.5-mm sequential axial image. A picture archiving and communication system (PACS) workstation using Centricity version 2.1 (GE Medical Systems, Waukesha, Wisconsin) was used to analyze each scan with a bone algorithm. On PACS, each initial displacement was characterized by the amount of SI joint widening at the level of the S1 and was measured using digital calipers.
Surgery
Mean time to surgery was 4 days (range, 2 to 15 days) after the injury. A total of 51 SI screws were implanted in 46 patients. We achieved closed reduction of the posterior pelvic ring by various techniques, including compression with percutaneous partially threaded screw fixation. In the cases in which the posterior ring lesion was associated with a pure pubic symphysis disruption, the anterior pelvis was initially reduced and stabilized with small-fragment plate fixation (Synthes, Inc, Paoli, Pennsylvania). The posterior complex was stabilized with 1 screw in 41 patients, 2 cases required a transiliac screw, and 2 screws (S1 and S2) were placed in each of the remaining 3 cases. Definitive stabilization of the posterior pelvis was achieved with percutaneous, partially threaded 7.3- or 7.5-mm–diameter cannulated screws (Synthes, Inc, and Zimmer Inc, Warsaw, Indiana, respectively) in 42 fractures and 6.5-mm screws (Synthes, Inc) in 4 fractures. In 11 cases where the fracture was through the sacrum, fully threaded cannulated screws were used to avoid compression. Screw insertion was performed under fluoroscopic guidance with inlet, outlet, and lateral sacral views. One of 2 fellowship-trained trauma surgeons performed the surgeries. Rehabilitation plans were customized to each patient based on concomitant injuries.
Postoperative Assessment
AP, lateral sacral, and inlet and outlet postoperative radiographs were taken in all cases within 24 hours after surgery. Pelvic CT was also obtained within 24 hours of surgery to review reduction and screw placement.
Using the measurement tool on the PACS system, we measured the penetration of the screw into the foramen. Screws were graded as intraosseous (completely contained within the sacral bone), skived (less than 2 mm of partial penetration into the S1 foramen), or extruded (the screw not contained by the bone). Screw penetration of the S1 was evaluated on the radiographic images as well as the axial images of the CT scans.
After surgery, the senior orthopedic resident and attending surgeon performed and documented detailed neurologic evaluations. They reviewed the medical record for neurologic deficit following surgical fixation.
Results
The mean follow-up time was 12 months (range, 8 months to 2 years). Two patients expired secondary to associated injuries. There were no early deaths related to the pelvic surgery. Stable fixation, including bone or ligamentous healing, as well as full weight-bearing status, was noted in every case. No case exhibited loss of reduction or implant failure or infection.
According to Matta’s criteria of anatomic reduction within 1 cm, all patients were found to have satisfactory reductions.7 Six of 46 patients had documented preoperative neurologic deficits. After percutaneous screw fixation, 10 of 46 patients had postoperative neurologic deficit, 2 of which were unchanged from preoperative evaluation. Of the 8 patients with new/altered postoperative neurologic deficit, CT showed neural foramen penetration greater than 2.1 mm in only 2 patients. Both patients underwent screw revision, resulting in improved neurologic deficit. The remaining 4 patients did not have foramen penetration and improved their neurologic function over the course of 2 weeks with return to presurgical status by 6 weeks without necessitating screw removal.
Twenty-three of the 51 screws (45%) had some violation of the S1 foramen on the CT. There were 17 patients with dysmorphic sacrums in which 21 S1 screws were placed. Eleven of 21 (52%) screws showed some penetration of the S1 foramen on CT. There were 29 patients with normal sacral morphology in which 30 S1 screws were placed. Twelve of 30 (40%) screws penetrated the S1 foramen. All violations were in the superior one-third position of the foramen. Two of 46 (4%; 1 with dysmorphism, 1 without) had a new neurologic deficit associated with the surgery (Table). CT showed sacral foramen penetration, and both screws were revised with a better neurologic examination.
High-resolution CTs were obtained in 32 patients, while 14 patients underwent the standard 5.0-mm–cut CTs. Of the 32 patients in which a 2.5-mm high-resolution CT was obtained, 20 (62.5%) had evidence of screw penetration (Figures 1, 2). All violations of the S1 neural foramen were in the superior portion of the foramen.
When compared with patients who had a 5.0-mm CT, the patients who underwent a high-resolution CT were more likely to show neural foramen penetration (P = .3). The average screw penetration into the S1 neural foramen measured 3.3 mm (range, 1.6-5.7 mm) in dysmorphic sacrum and 2.7 mm (range, 1.4-7 mm) in normal sacrum. However, in our study, any foramen penetration of less than 2.1 mm on CT did not result in neurologic deficit.
Discussion
Pelvic fractures are fairly common and represent approximately 5% of all trauma admissions and 3% of all skeletal fractures nationwide.1 The current treatment for SI disruption is either nonoperative or operative. Surgical fixation is technically demanding and surgeons often need a long learning curve to acquire the demanding technique because of the limitations of radiographic visualization of the relevant landmarks.16
Letournel17 developed the technique for iliosacral screw fixation for the treatment of posterior pelvic ring injuries, where 1 or 2 large screws (6.5-7.3 mm in diameter) are inserted under fluoroscopic guidance through the ilium, across the SI articulation, and into the superior sacral vertebral bodies using percutaneous techniques. Currently, the standard procedure to accomplish the percutaneous placement of iliosacral screws derives mainly from the technique described by Matta with the C-arm fluoroscopy visualizing the pelvis in 3 views: strict AP, inlet, and outlet views.7
Routt and colleagues4 recommend a strict lateral view of the sacrum, particularly when crossing the narrow zone of the sacral alar. They reported high union rates and accurate placement of the screws.4 There are limitations to the use of biplanar fluoroscopy because the intraoperative images are not orthogonal, with the average arc (67º) between the ideal inlet and outlet. However, because of the variability in sacral anatomy, CT guidance was recommended by others.2,6,8,18 Operating in a CT suite had other complications. Misinterpretation of CT led to “in-out-in” screws, which resulted in neurapraxia.
In our study, we used the technique described by Matta and colleagues for placement of the screws and performed a postoperative CT to evaluate screw placement and to assess pelvic reduction.7 We had a high penetration rate using CT, which increased with better resolution, even though none of the radiographs showed any obvious evidence of misplacement of the screws. Ebraheim and colleagues6 described the relationship of the S1 nerve root in its neural foramen and found it to be approximately 8.7 mm inferior and 7.8 mm medial to the starting point for a pedicle screw. Given these numbers, it is possible that a large amount of skiving can be tolerated contingent on an adequate reduction of the SI joint.
Because of our high rates of skiving and low rates of neurologic deficit, a new “safe zone” for screw insertion can be expanded to include skiving of the S1 neural foramen up to 3 mm without fear of nerve root injury. However, drilling and screw insertion at higher speeds can also cause neurologic injury secondary to thermal injury or soft tissue being caught up in a rotating drill/screw.
Evaluation of placement of percutaneous SI screw placement in our study resulted in neural foramen penetration in 43% of SI screws, which is higher than other studies.14,19,20 Our study showed that screw penetration up to 2 mm does not correlate with neurologic deficit. Iatrogenic neurologic deficit secondary to perforation of the foramina occurred in only 1 patient. Penetration of the foramina in all cases was in the superior portion of the foramen. We propose that there is a safe zone within the S1 neural foramen, and small amounts of penetration in the superior one-third of the foramen on axial CT images do not correlate with neurologic deficit. This potential safe zone is predicated on adequate reduction of the SI joint.
Neural foramen penetration shown on postoperative CT does not necessarily correlate with neurologic deficit. A postoperative CT is not indicated unless there are findings of a postoperative nerve injury. Our ideal screw placement skives the superior S1 foramen allowing for a larger screw diameter in a safe zone.
CT-guided placement has been proposed; however, concerns about radiation exposure, cost, and feasibility with similar outcomes compared with fluoroscopic-guided screw placement has resulted in its falling out of favor.
Iatrogenic nerve injuries are reported to occur in 0% to 6% of all percutaneous SI screw placement.14,21 Risk factors for iatrogenic nerve injury while using fluoroscopic guidance include sacral morphologic abnormalities, presence of intestinal gas, or contrast.22 Although these may be minimized with proper use of fluoroscopy, obtaining anatomic reduction as well as a thorough understanding of the pelvic morphology, the surgeon must be prepared to obtain further studies, such as a CT scan, if there is postoperative neurologic deficit.
Based on our findings, we do not routinely obtain a postoperative CT for SI screw placement, unless there is concern for malreduction or there is neurologic deficit. We also believe that up to 2 mm of foramen penetration is safe and does not result in neurologic deficit.
1. Failinger MS, McGanity PL. Unstable fractures of the pelvic ring. J Bone and Joint Surg Am. 1992;74(5):781-791.
2. Smith HE, Yuan PS, Sasso R, Papadopolous S, Vaccaro AR. An evaluation of image-guided technologies in the placement of percutaneous iliosacral screws. Spine (Phila Pa 1976). 2006;31(2):234-238.
3. Judet R, Judet J, Letournel E. Fractures of the acetabulum: classification and surgical approaches for open reduction. Preliminary report. J Bone Joint Surg Am. 1964;46(16):1615-1646.
4. Routt ML Jr, Kregor PJ, Simonian PT, Mayo KA. Early results of percutaneous iliosacral screws placed with the patient in the supine position. J Orthop Trauma. 1995;9(3):207-214.
5. Tonetti J, Carrat L, Blendea S, et al. Clinical results of percutaneous pelvic surgery. Computer assisted surgery using ultrasound compared to standard fluoroscopy. Comput Aided Surg. 2001;6(4):204-211.
6. Ebraheim NA, Coombs R, Jackson WT, Rusin JJ. Percutaneous computed tomography-guided stabilization of posterior pelvic fractures. Clin Orthop. 1994;(307):222-228.
7. Keating JF, Werier J, Blachut P, et al. Early fixation of the vertically unstable pelvis: the role of iliosacral screw fixation of the posterior lesion. J Orthop Trauma. 1999;13(2):107-113.
8. Webb LX, de Araujo W, Donofrio P, et al. Electromyography monitoring for percutaneous placement of iliosacral screws. J Orthop Trauma. 2000;14(4):245-254.
9. Barrick EF, O’Mara JW, Lane HE 3rd. Iliosacral screw insertion using computer-assisted CT image guidance: a laboratory study. Comput Aided Surg. 1998;3(6):289-296.
10. Routt ML Jr, Simonian PT, Agnew SG, Mann FA. Radiographic recognition of the sacral alar slope for optimal placement of iliosacral screws: a cadaveric and clinical study. J Orthop Trauma. 1996;10(3):171-177.
11. Altman DT, Jones CB, Routt ML Jr. Superior gluteal artery injury during iliosacral screw placement. J Orthop Trauma. 1999;13(3):220-227.
12. Stephen DJ. Pseudoaneurysm of the superior gluteal arterial system: an unusual cause of pain after a pelvic fracture. J Trauma. 1997;43(1):146-149.
13. Stöckle U, König B, Hofstetter R, Nolte LP, Haas NP. [Navigation assisted by image conversion. An experimental study on pelvic screw fixation]
[in German]. Unfallchirurg. 2001;104(3):215-220.
14. Templeman D, Schmidt A, Freese J, Weisman I, et al. Proximity of iliosacral screws to neurovascular structures after internal fixation. Clin Orthop. 1996;(329):194-198.
15. Young JW, Burgess AR, Brumback RJ, Poka A. Pelvic fractures: value of plain radiography in early assessment and management. Radiology. 1986;160(2):445-451.
16. Graves ML, Routt ML Jr. Iliosacral screw placement: are uniplanar changes realistic based on standard fluoroscopic imaging? J Trauma. 2011;7(1):204-208.
17. Letournel E. Pelvic fractures. Injury. 1978;10(2):145-148.
18. Blake-Toker AM, Hawkins L, Nadalo L, et al. CT guided percutaneous fixation of sacroiliac fractures in trauma patients. J Trauma. 2001;51(6):1117-1121.
19. Hinsche AF, Giannoudis PV, Smith RM. Fluoroscopy-based multiplanar image guidance for insertion of sacroiliac screws. Clin Orthop. 2002;(395):135-144.
20. van den Bosch EW, van Zwienen CM, van Vugt AB. Fluoroscopic positioning of sacroiliac screws in 88 patients. J Trauma. 2002;53(1):44-48.
21. Cole JD, Blum DA, Ansel LJ. Outcome after fixation of unstable posterior pelvic ring injuries. Clin Orthop. 1996;(329):160-179.
22. Routt ML Jr, Simonian PT. Closed reduction and percutaneous skeletal fixation of sacral fractures. Clin Orthop. 1996;(329):121-128.
1. Failinger MS, McGanity PL. Unstable fractures of the pelvic ring. J Bone and Joint Surg Am. 1992;74(5):781-791.
2. Smith HE, Yuan PS, Sasso R, Papadopolous S, Vaccaro AR. An evaluation of image-guided technologies in the placement of percutaneous iliosacral screws. Spine (Phila Pa 1976). 2006;31(2):234-238.
3. Judet R, Judet J, Letournel E. Fractures of the acetabulum: classification and surgical approaches for open reduction. Preliminary report. J Bone Joint Surg Am. 1964;46(16):1615-1646.
4. Routt ML Jr, Kregor PJ, Simonian PT, Mayo KA. Early results of percutaneous iliosacral screws placed with the patient in the supine position. J Orthop Trauma. 1995;9(3):207-214.
5. Tonetti J, Carrat L, Blendea S, et al. Clinical results of percutaneous pelvic surgery. Computer assisted surgery using ultrasound compared to standard fluoroscopy. Comput Aided Surg. 2001;6(4):204-211.
6. Ebraheim NA, Coombs R, Jackson WT, Rusin JJ. Percutaneous computed tomography-guided stabilization of posterior pelvic fractures. Clin Orthop. 1994;(307):222-228.
7. Keating JF, Werier J, Blachut P, et al. Early fixation of the vertically unstable pelvis: the role of iliosacral screw fixation of the posterior lesion. J Orthop Trauma. 1999;13(2):107-113.
8. Webb LX, de Araujo W, Donofrio P, et al. Electromyography monitoring for percutaneous placement of iliosacral screws. J Orthop Trauma. 2000;14(4):245-254.
9. Barrick EF, O’Mara JW, Lane HE 3rd. Iliosacral screw insertion using computer-assisted CT image guidance: a laboratory study. Comput Aided Surg. 1998;3(6):289-296.
10. Routt ML Jr, Simonian PT, Agnew SG, Mann FA. Radiographic recognition of the sacral alar slope for optimal placement of iliosacral screws: a cadaveric and clinical study. J Orthop Trauma. 1996;10(3):171-177.
11. Altman DT, Jones CB, Routt ML Jr. Superior gluteal artery injury during iliosacral screw placement. J Orthop Trauma. 1999;13(3):220-227.
12. Stephen DJ. Pseudoaneurysm of the superior gluteal arterial system: an unusual cause of pain after a pelvic fracture. J Trauma. 1997;43(1):146-149.
13. Stöckle U, König B, Hofstetter R, Nolte LP, Haas NP. [Navigation assisted by image conversion. An experimental study on pelvic screw fixation]
[in German]. Unfallchirurg. 2001;104(3):215-220.
14. Templeman D, Schmidt A, Freese J, Weisman I, et al. Proximity of iliosacral screws to neurovascular structures after internal fixation. Clin Orthop. 1996;(329):194-198.
15. Young JW, Burgess AR, Brumback RJ, Poka A. Pelvic fractures: value of plain radiography in early assessment and management. Radiology. 1986;160(2):445-451.
16. Graves ML, Routt ML Jr. Iliosacral screw placement: are uniplanar changes realistic based on standard fluoroscopic imaging? J Trauma. 2011;7(1):204-208.
17. Letournel E. Pelvic fractures. Injury. 1978;10(2):145-148.
18. Blake-Toker AM, Hawkins L, Nadalo L, et al. CT guided percutaneous fixation of sacroiliac fractures in trauma patients. J Trauma. 2001;51(6):1117-1121.
19. Hinsche AF, Giannoudis PV, Smith RM. Fluoroscopy-based multiplanar image guidance for insertion of sacroiliac screws. Clin Orthop. 2002;(395):135-144.
20. van den Bosch EW, van Zwienen CM, van Vugt AB. Fluoroscopic positioning of sacroiliac screws in 88 patients. J Trauma. 2002;53(1):44-48.
21. Cole JD, Blum DA, Ansel LJ. Outcome after fixation of unstable posterior pelvic ring injuries. Clin Orthop. 1996;(329):160-179.
22. Routt ML Jr, Simonian PT. Closed reduction and percutaneous skeletal fixation of sacral fractures. Clin Orthop. 1996;(329):121-128.
Large-Diameter Femoral Heads in Total Hip Arthroplasty: An Evidence-Based Review
A common cause for total hip arthroplasty (THA) revision is joint instability.1,2 The reported incidence of dislocation in primary THA ranges from 0.4% to 5.8%,3-5 but this rate increases after revision surgery.1,3-8 Use of large-diameter femoral heads has been proposed to decrease the risks for instability and to improve impingement-free range of motion (ROM).
The biomechanical rationale for using large-diameter femoral heads is that they must travel farther before subluxation or dislocation occurs (jump distance). Despite these benefits, there were initial concerns that catastrophic failure and high levels of volumetric wear would occur if these heads were used with conventional polyethylene liners. These concerns led to the development of alternative bearing surfaces, particularly metal-on-metal bearings, which offered theoretical benefits of large-diameter articulations that improved stability while purportedly being highly wear-resistant.9-11 However, concerns about adverse local soft-tissue reactions and high blood concentrations of metal ions tempered the initial enthusiasm for metal bearings.12-16 Fortunately, highly cross-linked polyethylene and fourth-generation ceramic bearing surfaces, with improved toughness and better wear properties, may allow use of large-diameter heads without the need for metal-on-metal bearings.17,18
In this article, we review the concepts and principles behind use of large-diameter ceramic or cobalt-chromium femoral heads on polyethylene-bearing surfaces in THA with particular attention to biomechanics, early concerns about polyethylene wear and rim fractures, recent improvements in material properties of polyethylene and ceramic bearings, dislocation rates, and clinical and functional outcomes.
Definitions
For this review, we define large-diameter femoral heads as having diameters of 36 mm or more and conventional or small-diameter femoral heads as having diameters between 22 and 32 mm.
Biomechanics
Head–Neck Ratio, Impingement-Free ROM, and Jump Distance
Several implant design principles have been proposed to reduce the risks for impingement and dislocation. Of these, large femoral head diameters have been extensively studied.19,20 It is well known that impingement of the femoral neck on the cup edge promotes edge loading and higher wear rates. In addition, impingement increases the tendency of the head to sublux from the acetabulum. One strategy for avoiding this component-to-component impingement is to increase the head–neck ratio (HNR), the ratio of the femoral head to the neck diameter. Biomechanically, increased HNRs lead to delayed contact between the femoral neck and the acetabular liner.21,22 Therefore, with large femoral heads, which have large HNRs, impingement occurs later and at larger ROMs—compared with small-diameter femoral heads, which have lower HNRs and are more prone to early impingement and subluxation (Figure 1).23-26
In a cadaveric study of 6 hips, Bartz and colleagues23 reported a significantly higher preimpingement ROM when the prosthetic head size increased from 22 mm to 28 mm (P < .05). They found a change from prosthetic to osseous impingement when the head size increased from 22 mm to 32 mm. Similar results were observed in a computer simulation model by Cinotti and colleagues,27 who demonstrated that increasing the femoral head size from 28 mm to 38 mm resulted in a 5° improvement in ROM. However, the largest gains were observed when the heads with the smallest diameters were upsized; ROM improved only marginally when femoral head size was further increased from 32 mm to 38 mm. The primary reason for the lack of expected improvement in ROM with head sizes of more than 32 mm is often bone-on-bone impingement. Burroughs and colleagues28 demonstrated that the 38-mm and 44-mm heads virtually eliminated component-to-component impingement except in extremes of external rotation. However, there were no differences in ROM between 38-mm and 44-mm heads because of osseous impingement. In addition, large heads are less likely to sublux or dislocate, as they need to travel farther before reaching the edge of the acetabular cup before dislocation. This is known as the jump distance, and it corresponds to the depth of the acetabular shell, which in turn equates with the radius of the femoral head (Figures 2A, 2B). For this reason, the larger the femoral head diameter, the farther the jump distance and, correspondingly, the lower the risk for dislocation.29
Elevated liners historically were used to increase the jump distance for dislocation.30 These liners, however, can increase impingement at the extremes of motion.31 Some of these problems can be avoided with use of larger heads, which have increased jump distances without additional risks for impingement. Moreover, large heads create a suction effect that provides passive resistance to dislocation.32 With head diameters beyond 38 mm, impingement-free ROM often plateaus. However, the jump distance required for dislocations to occur continues to increase as femoral head diameters increase in size. Thus, patients may experience fewer motion benefits but continue to benefit from overall stability with femoral head sizes increasing beyond 38 mm.
Current evidence suggests there may be substantial benefits toward improved stability from increasing head diameters from 22 mm to 38 mm because of the increase in jump distances and improvements in prosthetic impingement-free ROM. However, there may be little gain in ROM from increasing the head diameters beyond these dimensions because of the potential risks of bony impingement. Nevertheless, there may be some additional benefits toward stability from improvement in jump distances with incremental head sizes
beyond 38 mm.29,33,34
Finite Element Analysis Studies
Finite element analysis of large-diameter heads in THA has shown that, at optimal cup inclination (45°), most stresses occur on the articular surface of the liner. However, these stresses remain well below the yield strength of the polyethylene liners.29 With increasing abduction angles, the stress concentration increases substantially because of the decreased contact surface area. At these angles, the point of maximum contact moves toward the rim of the polyethylene liner, which can lead to rim fractures or failure of locking mechanisms.29,35,36
Early Concerns With Large-Diameter Femoral Heads: Wear, Liner Failure, and Fracture of Ceramic Components
Use of small-diameter femoral heads started with the first report by Charnley37 of “low frictional torque arthroplasty.” Charnley initially considered a 41.5-mm femoral head, but he thought it would increase risks for acetabular loosening from high frictional torque generated by the large head, and he switched to a small-diameter (22.5 mm) design. One of the tradeoffs with smaller diameter heads was decreased jump height in addition to increased linear wear.
Large femoral heads used with cemented polyethylene acetabular components historically have been associated with increased rates of volumetric wear but low rates of linear wear, which potentially may increase the risk for osteolysis.38-40 However, newer highly cross-linked polyethylene liners have shown improved in vitro and in vivo volumetric wear characteristics and potentially lower linear wear rates compared with earlier designs (Table 1).28,41-43
Another concern about earlier generations of large femoral heads was the risk for catastrophic liner failure on conventional polyethylene. This was originally reported by Berry and colleagues,47 who described wear-through and failure in patients with thin (< 5 mm) acetabular cups. However, these concerns have been largely addressed by the development of highly cross-linked polyethylene, which has improved wear characteristics and fatigue resistance.48
Recent Improvements in Material Properties of Polyethylene and Ceramic Bearings
The development of highly cross-linked polyethylene and fourth-generation ceramics has renewed interest in large-diameter bearings in THA. These bearing surfaces improve wear, enhance material properties, and have superior oxidation resistance.42,48-53
We now briefly describe the methods used to improve the material properties of polyethylene and ceramics. Studies have shown that increasing the radiation dose (up to 200 kGy) increases cross-linking and causes an inverse exponential decrease in polyethylene wear.28,41,48-51 However, increasing radiation doses also increases production of free radicals, which diminish the material strength of these polyethylenes. The current generation of highly cross-linked polyethylene liners is produced through a variety of manufacturing strategies to improve cross-linking and reduce wear. These strategies include differential radiation doses (50-100 kGy), techniques (electron beam, radiation), and thermal treatments (melting, annealing). Moreover, to enhance the material properties and reduce the incidence of rim cracking and delamination, authors have proposed using vitamin E supplementation to minimize the amount of subsurface oxidation that occurs as an inevitable consequence of free radical formation during fabrication.54,55 A terminal sterilization process (eg, gas plasma, ethylene oxide, or gamma sterilization in nitrogen) is needed to make commercial, highly cross-linked polyethylene.52,53
Fourth-generation ceramics manufactured with nano-sized yttria-stabilized tetragonal zirconia particles in a stable alumina matrix have more fracture toughness and improved wear characteristics.54,55 In addition, oxide additives (eg, chromium oxide, strontium oxide) improve hardness and dissipate energy by deflecting cracks to prevent their propagation.56 Moreover, the smaller grain sizes of fourth-generation ceramic bearings compared with third-generation designs (0.8 µm vs 1-5 µm) cause less disruption of the fluid film layer, which ultimately results in improved wear performance.57
Multiple studies have found reduced wear rates with metal and ceramic large heads coupled with highly cross-linked polyethylene-bearings (Table 2).17,41,50,58 Bragdon and colleagues,58 using radiostereometric analysis in 25 patients, found no significant differences in mean head penetration rates between 36-mm and 28-mm cobalt-chromium (Co-Cr) heads articulating with highly cross-linked polyethylene cups at a mean follow-up of 3 years (0.035 mm/y vs 0.046 mm/y; P = .11). Geller and colleagues,64 in their study of 42 patients with large-diameter (> 32 mm) Co-Cr femoral heads, found low mean (SD) linear wear rates of 0.06 (0.41) mm/y at a mean follow-up of 3 years. D’Antonio and colleagues,65 in a multicenter study, reported low average linear wear (0.015 mm/y) and volumetric wear (12.1 mm3/y) over 5 years using sequentially annealed cross-linked polyethylene. In vitro reports suggest that large-diameter ceramic heads may have lower wear properties than Co-Cr heads do. Galvin and colleagues,66 in an in vitro hip simulator study, found that large-diameter ceramic heads on highly cross-linked ultrahigh-molecular-weight polyethylene had 40% reductions in steady-state wear rates compared with Co-Cr heads on highly cross-linked bearings (4.7 vs 8.1 mm3/million cycles; P < 0.01).
Dislocation Rates
Several patient, surgeon, and implant factors affect the rate of dislocations after THA. Multiple implant options utilize the biomechanical advantage that large-diameter heads have in improving stability. Various alternatives include use of constrained tripolar heads, dual-mobility bearings, and conventional large-diameter heads with standard liners.67-69
Large-Diameter Heads
Despite the biomechanical advantages of large-diameter metal-on-polyethylene bearings, prior studies have questioned use of these bearings because of risks for increased wear and rim failures. However, the improved wear properties of highly cross-linked polyethylene, elaborated earlier, have led to a reappraisal of this option (Table 2).4,70 Howie and colleagues,71 in a randomized control trial of 644 patients, also found significantly lower rates of dislocation after primary THA with 36-mm heads compared with 28-mm heads (1.3% vs 5.4%; P = .012); in addition, fewer dislocations occurred with 36-mm heads than with 28-mm heads (4.9% vs 12.2%; P = .27) in a series of 44 patients in revision settings. Similarly, in a study conducted with 39,271 Medicare patients between 1998 and 2007, Malkani and colleagues72 found a decrease in the dislocation rate, from 4.21% to 2.14%, with use of large-diameter femoral heads. These results have been confirmed by several other authors.34,66,73,74 Similar results were observed in 65,992 patients in the Australian National Joint Replacement Registry by Conroy and colleagues,75 who reported a significant decrease in the risk for dislocation with large heads (≥ 30 mm) compared with 22-mm heads (relative risk, 1.0 vs 3.1; P ≤ .001).
Few studies have analyzed the role of large-diameter femoral heads in the presence of compromised soft tissues around the hip. Kung and Ries,76 evaluating the influence of large-diameter heads in the presence and absence of a deficient abductor mechanism, demonstrated statistically significant reductions in rates of dislocation after 230 revision THAs when the abductor mechanism was intact with use of 36-mm heads compared with 28-mm heads (12.7% vs 0%; P = .015). With abductor deficiency, though, the positive effect of large heads in reducing dislocation rates was substantially reduced and was similar to that of small heads (P = .74).76
Large heads considerably improve overall stability and lower dislocation rates in THA. With the development of newer ceramics and highly cross-linked polyethylenes, the wear rates reported in multiple studies appear to be less concerning.
Constrained Tripolar Heads
Tripolar heads have been proposed as treatment options for improving stability in patients with chronic and recurrent instability after THA. The tripolar implant consists of a metal head that snap-fits into a polyethylene liner with a polished Co-Cr backing. This bipolar head articulates with a polyethylene bearing that is press-fitted onto a metal acetabular shell and constrained by a metal ring snapped to the outer polyethylene bearing. The bipolar component behaves as a large-diameter femoral head, and the metal ring provides additional restraint, further improving stability.
Williams and colleagues77 performed a systematic review and reported on the outcomes of constrained tripolar liners in 1199 hips at a mean follow-up of 4 years (range, 2-10 years). The mean dislocation rate was 10%, and the mean rate of revision surgery unrelated to instability was 4%. In a study of 43 hips at a mean follow-up of 4 years (range, 2-9 years), Zywiel and colleagues78 reported on the clinical and radiographic outcomes of tripolar constrained liners. Their study group had a mean Harris Hip Score (HHS) of 82 points (range, 38-100 points) and overall survival of 91%, with no evidence of radiographic loosening during follow-up. Despite the improvements in stability with constrained tripolar liners, some authors have reported multiple mechanisms of failure with these devices.79-81 In a study of 43 failed constrained tripolar liners with a mean time to failure of about 2 years, Guyen and colleagues79 identified 5 different failure modes (types 1-5) involving all 4 interfaces in these components.
Encouraging outcomes have been reported at midterm follow-up with tripolar constrained liners. However, concerns about failure at the interfaces suggest that use of these components should be restricted to patients with deficient abductor mechanisms or neuromuscular compromise, low-demand elderly patients, and salvage cases of recurrent dislocations.79
Dual-Mobility Bearings
For more than 20 years, different dual-mobility bearings have been used for difficult acetabular reconstructive scenarios and prevention of instability.82,83 Dual-mobility cups provide constructs that snap-fit a small-diameter femoral head within a large polyethylene insert that articulates with a fixed metal shell. This effectively increases the functional head diameter.
Various authors have reported excellent survivorship rates (92%-99%) and low dislocation rates for these bearings at 5- to 10-year follow-up.82,84-90 Philippot and colleagues,86 in a recent study of 438 hips with dual-mobility cups, reported excellent survivorship (96%) and no early or late instability within a 15-year follow-up. Bouchet and colleagues69 compared dual-mobility bearings (105 hips) with conventional metal-on-polythene bearings (108 hips) and found significantly (P < .05) lower dislocation rates for the dual-mobility implants at a minimum 1-year follow-up. The French Society of Orthopaedics and Traumatology performed a multicenter analysis of 3473 hips with dual-mobility cups implanted in France between January 1998 and December 2003.87 During a mean follow-up of 7 years (range, 5-11 years), there were 15 dislocations (0.43%), 14 of which occurred early, within 3 months of implantation (0.4%). Aseptic implant survivorship was 95% at 10-year follow-up.
Use of these bearings has recently increased in the United States. Short-term and midterm follow-up data show low rates of dislocation and wear. Long-term data are to come.
Clinical and Functional Outcomes of Large-Diameter Femoral Heads
There is a paucity of long-term outcomes data on use of large-diameter heads with highly cross-linked polyethylene bearings. Short-term and midterm clinical results appear to be excellent, with low rates of wear, osteolysis, and aseptic loosening.28,41,73,89-92
Plate and colleagues91 compared the effects of large-diameter (≥ 36 mm) and small-diameter (26 mm, 28 mm) metal heads on highly cross-linked polyethylene bearings. At a mean follow-up of 5 years (range, 4-8.4 years), the large-head cohort had a mean HHS of 90 points (range, 50-100 points) and no dislocations or radiographic evidence of stem or cup loosening. Similarly, Meftah and colleagues93 reported 100% stem survivorship and excellent clinical outcomes—a mean Western Ontario and McMaster Universities Arthritis Index (WOMAC) score of 30 points—for 72 hips with use of large ceramic heads (≥ 32 mm) on highly cross-linked polyethylene at a mean follow-up of 3 years. Gagala and colleagues94 reported excellent clinical and radiographic outcomes in 50 hips (18 ceramic on ceramic, 32 ceramic on polyethylene; 36-mm heads) at a mean follow-up of 3.5 years. Mean HHS was 94 points, and there was no evidence of liner fractures, aseptic loosening, or osteolysis.
In summary, large-diameter femoral heads in THA have become increasingly popular because of improvements in the material properties and wear characteristics of highly cross-linked polyethylene and fourth-generation ceramics. Despite the potential advantages of large heads in preventing dislocations, the basic surgical tenets of placing the acetabular component in appropriate alignment remain firmly established. Implants with functionally large heads (eg, dual-mobility bearings, constrained tripolar liners) may play an important role in patients at high risk for dislocation—particularly elderly patients with poor neuromuscular muscle coordination or deficient abductors, trauma patients, and patients with prior dislocations. Short-term and midterm results are excellent; rates of wear, aseptic loosening, and osteolysis are low. However, long-term outcomes data are needed to support widespread use of large heads in younger and more active patients.
1. Dorr LD, Wolf AW, Chandler R, Conaty JP. Classification and treatment of dislocations of total hip arthroplasty. Clin Orthop. 1983;(173):151-158.
2. Dorr LD, Wan Z. Causes of and treatment protocol for instability of total hip replacement. Clin Orthop. 1998;(355):144-151.
3. Turner RS. Postoperative total hip prosthetic femoral head dislocations. Incidence, etiologic factors, and management. Clin Orthop. 1994;(301):196-204.
4. Woo RY, Morrey BF. Dislocations after total hip arthroplasty. J Bone Joint Surg Am. 1982;64(9):1295-1306.
5. Etienne A, Cupic Z, Charnley J. Postoperative dislocation after Charnley low-friction arthroplasty. Clin Orthop. 1978;(132):19-23.
6. Fackler CD, Poss R. Dislocation in total hip arthroplasties. Clin Orthop. 1980;(151):169-178.
7. Joshi A, Lee CM, Markovic L, Vlatis G, Murphy JC. Prognosis of dislocation after total hip arthroplasty. J Arthroplasty. 1998;13(1):17-21.
8. Lindberg HO, Carlsson AS, Gentz CF, Pettersson H. Recurrent and non-recurrent dislocation following total hip arthroplasty. Acta Orthop Scand. 1982;53(6):947-952.
9. Eswaramoorthy V, Moonot P, Kalairajah Y, Biant LC, Field RE. The Metasul metal-on-metal articulation in primary total hip replacement: clinical and radiological results at ten years. J Bone Joint Surg Br. 2008;90(10):1278-1283.
10. Grubl A, Marker M, Brodner W, et al. Long-term follow-up of metal-on-metal total hip replacement. J Orthop Res. 2007;25(7):841-848.
11. Leslie I, Williams S, Brown C, et al. Effect of bearing size on the long-term wear, wear debris, and ion levels of large diameter metal-on-metal hip replacements—an in vitro study. J Biomed Mater Res B Appl Biomater. 2008;87(1):163-172.
12. Verhaar JA. The hard lesson of metal-on-metal hip implants [in Dutch]. Ned Tijdschr Geneeskd. 2012;156(42):A5564.
13. Fabi D, Levine B, Paprosky W, et al. Metal-on-metal total hip arthroplasty: causes and high incidence of early failure. Orthopedics. 2012;35(7):e1009-e1016.
14. Heneghan C, Langton D, Thompson M. Ongoing problems with metal-on-metal hip implants. BMJ. 2012;344:e1349.
15. Lee RK, Nevelos J, Vigdorchik J, Markel DC. Bearing surfaces for hip arthroplasty—is metal-on-metal a passing fancy? Surg Technol Int. 2012;22:243-249.
16. Voleti PB, Baldwin KD, Lee GC. Metal-on-metal vs conventional total hip arthroplasty: a systematic review and meta-analysis of randomized controlled trials. J Arthroplasty. 2012;27(10):1844-1849.
17. Urban JA, Garvin KL, Boese CK, et al. Ceramic-on-polyethylene bearing surfaces in total hip arthroplasty. Seventeen to twenty-one-year results.
J Bone Joint Surg Am. 2001;83(11):1688-1694.
18. Callaghan JJ, Liu SS. Ceramic on crosslinked polyethylene in total hip replacement: any better than metal on crosslinked polyethylene? Iowa Orthop J. 2009;29:1-4.
19. Barrack RL. Dislocation after total hip arthroplasty: implant design and orientation. J Am Acad Orthop Surg. 2003;11(2):89-99.
20. Krushell RJ, Burke DW, Harris WH. Elevated-rim acetabular components. Effect on range of motion and stability in total hip arthroplasty. J Arthroplasty. 1991;6(suppl):S53-S58.
21. Morrey BF. Instability after total hip arthroplasty. Orthop Clin North Am. 1992;23(2):237-248.
22. Morrey BF. Dislocation. In: Morrey BF, ed. Joint Replacement Arthroplasty. New York, NY: Churchill Livingstone; 1991:851-865.
23. Bartz RL, Nobel PC, Kadakia NR, Tullos HS. The effect of femoral component head size on posterior dislocation of the artificial hip joint. J Bone Joint Surg Am. 2000;82(9):1300-1307.
24. Nicholas RM, Orr JF, Mollan RA, Calderwood JW, Nixon JR, Watson P. Dislocation of total hip replacements. A comparative study of standard, long posterior wall and augmented acetabular components. J Bone Joint Surg Br. 1990;72(3):418-422.
25. McCollum DE, Gray WJ. Dislocation after total hip arthroplasty. Causes and prevention. Clin Orthop. 1990;(261):159-170.
26. Herrlin K, Selvik G, Pettersson H, Kesek P, Onnerfalt R, Ohlin A. Position, orientation and component interaction in dislocation of the total hip prosthesis. Acta Radiol. 1988;29(4):441-444.
27. Cinotti G, Lucioli N, Malagoli A, Calderoli C, Cassese F. Do large femoral heads reduce the risks of impingement in total hip arthroplasty with optimal and non-optimal cup positioning? Int Orthop. 2011;35(3):317-323.
28. Burroughs BR, Rubash HE, Harris WH. Femoral head sizes larger than 32 mm against highly cross-linked polyethylene. Clin Orthop. 2002;(405):150-157.
29. Crowninshield RD, Maloney WJ, Wentz DH, Humphrey SM, Blanchard CR. Biomechanics of large femoral heads: what they do and don‘t do. Clin Orthop. 2004;(429):102-107.
30. Charnley J. Low Friction Arthroplasty of the Hip: Theory and Practice. New York, NY: Springer; 1979.
31. Yamaguchi M, Akisue T, Bauer TW, Hashimoto Y. The spatial location of impingement in total hip arthroplasty. J Arthroplasty. 2000;15(3):305-313.
32. Peters CL, McPherson E, Jackson JD, Erickson JA. Reduction in early dislocation rate with large-diameter femoral heads in primary total hip arthroplasty. J Arthroplasty. 2007;22(6 suppl 2):140-144.
33. Masonis JL, Bourne RB. Surgical approach, abductor function, and total hip arthroplasty dislocation. Clin Orthop. 2002;(405):46-53.
34. Beaule PE, Schmalzried TP, Udomkiat P, Amstutz HC. Jumbo femoral head for the treatment of recurrent dislocation following total hip replacement. J Bone Joint Surg Am. 2002;84(2):256-263.
35. Oral E, Malhi AS, Muratoglu OK. Mechanisms of decrease in fatigue crack propagation resistance in irradiated and melted UHMWPE. Biomaterials. 2006;27(6):917-925.
36. Baker DA, Bellare A, Pruitt L. The effects of degree of crosslinking on the fatigue crack initiation and propagation resistance of orthopedic-grade polyethylene. J Biomed Mater Res A. 2003;66(1):146-154.
37. Charnley J. Total hip replacement by low-friction arthroplasty. Clin Orthop. 1970;(72):7-21.
38. Kabo JM, Gebhard JS, Loren G, Amstutz HC. In vivo wear of polyethylene acetabular components. J Bone Joint Surg Br. 1993;75(2):254-258.
39. Livermore J, Ilstrup D, Morrey B. Effect of femoral head size on wear of the polyethylene acetabular component. J Bone Joint Surg Am. 1990;72(4):518-528.
40. Ma SM, Kabo JM, Amstutz HC. Frictional torque in surface and conventional hip replacement. J Bone Joint Surg Am. 1983;65(3):366-370.
41. Muratoglu OK, Bragdon CR, O‘Connor D, et al. Larger diameter femoral heads used in conjunction with a highly cross-linked ultra-high molecular weight polyethylene: a new concept. J Arthroplasty. 2001;16(8 suppl 1):24-30.
42. Thomas GER, Simpson DJ, Mehmood S, et al. The seven-year wear of highly cross-linked polyethylene in total hip arthroplasty: a double-blind, randomized controlled trial using radiostereometric analysis. J Bone Joint Surg Am. 2011;93(8):716-722.
43. Mutimer J, Devane PA, Adams K, Horne JG. Highly crosslinked polyethylene reduces wear in total hip arthroplasty at 5 years. Clin Orthop. 2010;468(12):3228-3233.
44. Bragdon CR, Doerner M, Martell J, Jarrett B, Palm H, Malchau H. The 2012 John Charnley Award: clinical multicenter studies of the wear performance of highly crosslinked remelted polyethylene in THA. Clin Orthop. 2013;471(2):393-402.
45. Lachiewicz PF, Heckman DS, Soileau ES, Mangla J, Martell JM. Femoral head size and wear of highly cross-linked polyethylene at 5 to 8 years. Clin Orthop. 2009;467(12):3290-3296.
46. Sychterz CJ, Engh CA Jr, Young AM, Hopper RH Jr, Engh CA. Comparison of in vivo wear between polyethylene liners articulating with ceramic and cobalt-chrome femoral heads. J Bone Joint Surg Br. 2000;82(7):948-951.
47. Berry DJ, Barnes CL, Scott RD, Cabanela ME, Poss R. Catastrophic failure of the polyethylene liner of uncemented acetabular components.
J Bone Joint Surg Br. 1994;76(4):575-578.
48. McKellop H, Shen FW, Lu B, Campbell P, Salovey R. Development of an extremely wear-resistant ultra high molecular weight polyethylene for total hip replacements. J Orthop Res. 1999;17(2):157-167.
49. Wang A, Essner A, Polineni VK, Stark C, Dumbleton JH. Lubrication and wear of ultra-high molecular weight polyethylene in total joint replacements. Tribol Int. 1998;31(1-3):17-33.
50. Estok DM 2nd, Burroughs BR, Muratoglu OK, Harris WH. Comparison of hip simulator wear of 2 different highly cross-linked ultra high molecular weight polyethylene acetabular components using both 32- and 38-mm femoral heads. J Arthroplasty. 2007;22(4):581-589.
51. Muratoglu OK, Bragdon CR, O‘Connor DO, et al. Unified wear model for highly crosslinked ultra-high molecular weight polyethylenes (UHMWPE). Biomaterials. 1999;20(16):1463-1470.
52. Harris WH, Muratoglu OK. A review of current cross-linked polyethylenes used in total joint arthroplasty. Clin Orthop. 2005;(430):46-52.
53. Muratoglu OK, Bragdon CR, O‘Connor DO, Jasty M, Harris WH. A novel method of cross-linking ultra-high-molecular-weight polyethylene to improve wear, reduce oxidation, and retain mechanical properties. Recipient of the 1999 HAP Paul Award. J Arthroplasty. 2001;16(2):149-160.
54. Bal BS, Garino J, Ries M, Rahaman MN. A review of ceramic bearing materials in total joint arthroplasty. Hip Int. 2007;17(1):21-30.
55. Traina F, De Fine M, Di Martino A, Faldini C. Fracture of ceramic bearing surfaces following total hip replacement: a systematic review. Biomed Res Int. 2013;2013:157247.
56. Cai YZ, Yan SG. Development of ceramic-on-ceramic implants for total hip arthroplasty. Orthop Surg. 2010;2(3):175-181.
57. Stewart TD, Tipper JL, Insley G, Streicher RM, Ingham E, Fisher J. Long-term wear of ceramic matrix composite materials for hip prostheses under severe swing phase microseparation. J Biomed Mater Res B Appl Biomater. 2003;66(2):567-573.
58. Bragdon CR, Greene ME, Freiberg AA, Harris WH, Malchau H. Radiostereometric analysis comparison of wear of highly cross-linked polyethylene against 36- vs 28-mm femoral heads. J Arthroplasty. 2007;22(6 suppl 2):125-129.
59. Lombardi AV Jr, Skeels MD, Berend KR, Adams JB, Franchi OJ. Do large heads enhance stability and restore native anatomy in primary total hip arthroplasty? Clin Orthop. 2011;469(6):1547-1553.
60. Lachiewicz PF, Soileau ES. Low early and late dislocation rates with 36- and 40-mm heads in patients at high risk for dislocation. Clin Orthop. 2013;471(2):439-443.
61. Cai P, Hu Y, Xie J. Large-diameter Delta ceramic-on-ceramic versus common-sized ceramic-on-polyethylene bearings in THA. Orthopedics. 2012;35(9):e1307-e1313.
62. Park KS, Yoon TR, Hwang SY, Lee KB. Modified minimally invasive two-incision total hip arthroplasty using large diameter femoral head. Indian
J Orthop. 2012;46(1):29-35.
63. Garbuz DS, Masri BA, Duncan CP, et al. The Frank Stinchfield Award: dislocation in revision THA: do large heads (36 and 40 mm) result in reduced dislocation rates in a randomized clinical trial? Clin Orthop. 2012;470(2):351-356.
64. Geller JA, Malchau H, Bragdon C, Greene M, Harris WH, Freiberg AA. Large diameter femoral heads on highly cross-linked polyethylene: minimum 3-year results. Clin Orthop. 2006;(447):53-59.
65. D’Antonio JA, Capello WN, Ramakrishnan R. Second-generation annealed highly cross-linked polyethylene exhibits low wear. Clin Orthop. 2012;470(6):1696-1704.
66. Galvin AL, Jennings LM, Tipper JL, Ingham E, Fisher J. Wear and creep of highly crosslinked polyethylene against cobalt chrome and ceramic femoral heads. Proc Inst Mech Eng H. 2010;224(10):1175-1183.
67. Skeels MD, Berend KR, Lombardi AV Jr. The dislocator, early and late: the role of large heads. Orthopedics. 2009;32(9).
68. Plate JF, Seyler TM, Stroh DA, Issa K, Akbar M, Mont MA. Risk of dislocation using large- vs. small-diameter femoral heads in total hip arthroplasty. BMC Res Notes. 2012;5(1):553.
69. Bouchet R, Mercier N, Saragaglia D. Posterior approach and dislocation rate: a 213 total hip replacements case–control study comparing the dual mobility cup with a conventional 28-mm metal head/polyethylene prosthesis. Orthop Traumatol Surg Res. 2011;97(1):2-7.
70. Ali Khan MA, Brakenbury PH, Reynolds IS. Dislocation following total hip replacement. J Bone Joint Surg Br. 1981;63(2):214-218.
71. Howie DW, Holubowycz OT, Middleton R. Large femoral heads decrease the incidence of dislocation after total hip arthroplasty: a randomized controlled trial. J Bone Joint Surg Am. 2012;94(12):1095-1102.
72. Malkani AL, Ong KL, Lau E, Kurtz SM, Justice BJ, Manley MT. Early- and late-term dislocation risk after primary hip arthroplasty in the Medicare population. J Arthroplasty. 2010;25(6 suppl):21-25.
73. Berry DJ, von Knoch M, Schleck CD, Harmsen WS. Effect of femoral head diameter and operative approach on risk of dislocation after primary total hip arthroplasty. J Bone Joint Surg Am. 2005;87(11):2456-2463.
74. Cho MR, Lee HS, Lee SW, Choi CH, Kim SK, Ko SB. Results after total hip arthroplasty with a large head and bipolar arthroplasty in patients with displaced femoral neck fractures. J Arthroplasty. 2011;26(6):893-896.
75. Conroy JL, Whitehouse SL, Graves SE, Pratt NL, Ryan P, Crawford RW. Risk factors for revision for early dislocation in total hip arthroplasty.
J Arthroplasty. 2008;23(6):867-872.
76. Kung PL, Ries MD. Effect of femoral head size and abductors on dislocation after revision THA. Clin Orthop. 2007;(465):170-174.
77. Williams JT Jr, Ragland PS, Clarke S. Constrained components for the unstable hip following total hip arthroplasty: a literature review. Int Orthop. 2007;31(3):273-277.
78. Zywiel MG, Mustafa LH, Bonutti PM, Mont MA. Are abductor muscle quality and previous revision surgery predictors of constrained liner failure in hip arthroplasty? Int Orthop. 2011;35(6):797-802.
79. Guyen O, Lewallen DG, Cabanela ME. Modes of failure of Osteonics constrained tripolar implants: a retrospective analysis of forty-three failed implants. J Bone Joint Surg Am. 2008;90(7):1553-1560.
80. Banks LN, McElwain JP. An unusual mode of failure of a tripolar constrained acetabular liner: a case report. Arch Orthop Trauma Surg. 2010;130(4):503-505.
81. Robertson WJ, Mattern CJ, Hur J, Su EP, Pellicci PM. Failure mechanisms and closed reduction of a constrained tripolar acetabular liner.
J Arthroplasty. 2009;24(2):322.e5-e11.
82. Aubriot JH, Lesimple P, Leclercq S. Study of Bousquet‘s non-cemented acetabular implant in 100 hybrid total hip prostheses (Charnley type cemented femoral component). Average 5-year follow-up [in French]. Acta Orthop Belg. 1993;59(suppl 1):267-271.
83. Farizon F, de Lavison R, Azoulai JJ, Bousquet G. Results with a cementless alumina-coated cup with dual mobility. A twelve-year follow-up study. Int Orthop. 1998;22(4):219-224.
84. Mertl P, Combes A, Leiber-Wackenheim F, Fessy MH, Girard J, Migaud H. Recurrence of dislocation following total hip arthroplasty revision using dual mobility cups was rare in 180 hips followed over 7 years. HSS J. 2012;8(3):251-256.
85. Langlais FL, Ropars M, Gaucher F, Musset T, Chaix O. Dual mobility cemented cups have low dislocation rates in THA revisions. Clin Orthop. 2008;466(2):389-395.
86. Philippot R, Farizon F, Camilleri JP, et al. Survival of dual mobility socket with a mean 17 years follow-up [in French]. Rev Chir Orthop Reparatrice Appar Mot. 2008;94(1):43-48.
87. Adam P, Philippe R, Ehlinger M, et al. Dual mobility cups hip arthroplasty as a treatment for displaced fracture of the femoral neck in the elderly.
A prospective, systematic, multicenter study with specific focus on postoperative dislocation. Orthop Traumatol Surg Res. 2012;98(3):296-300.
88. Fessy MH. La double mobilité [Dual mobility]. Revue de Chirurgie Orthopédique et Traumatologique. 2010;96(7):891-898.
89. Mont MA, Issa K, Naziri Q, Harwin SF, Delanois RE, Johnson AJ. The use of dual-mobility bearings in difficult hip arthroplasty reconstructive cases. Surg Technol Int. 2011;21:234-240.
90. Sayeed SA, Mont MA, Costa CR, et al. Early outcomes of sequentially cross-linked thin polyethylene liners with large diameter femoral heads in total hip arthroplasty. Bull NYU Hosp Jt Dis. 2011;69(suppl 1):S90-S94.
91. Plate JF, Seyler TM, Stroh DA, Issa K, Akbar M, Mont MA. Risk of dislocation using large- vs. small-diameter femoral heads in total hip arthroplasty. BMC Res Notes. 2012;5(1):553.
92. Sato T, Nakashima Y, Akiyama M, et al. Wear resistant performance of highly cross-linked and annealed ultra-high molecular weight polyethylene against ceramic heads in total hip arthroplasty. J Orthop Res. 2012;30(12):2031-2037.
93. Meftah M, Ebrahimpour PB, He C, Ranawat AS, Ranawat CS. Preliminary clinical and radiographic results of large ceramic heads on highly cross-linked polyethylene. Orthopedics. 2011;34(6):133.
94. Gagala J, Mazurkiewicz T, Dajewski Z. Large diameter femoral heads in primary alumina/alumina and XSPE/alumina total hip arthroplasty.
A follow-up study of 50 hips after average 40 months and review of literature [in Polish]. Chir Narzadow Ruchu Ortop Pol. 2011;76(1):14-20.
A common cause for total hip arthroplasty (THA) revision is joint instability.1,2 The reported incidence of dislocation in primary THA ranges from 0.4% to 5.8%,3-5 but this rate increases after revision surgery.1,3-8 Use of large-diameter femoral heads has been proposed to decrease the risks for instability and to improve impingement-free range of motion (ROM).
The biomechanical rationale for using large-diameter femoral heads is that they must travel farther before subluxation or dislocation occurs (jump distance). Despite these benefits, there were initial concerns that catastrophic failure and high levels of volumetric wear would occur if these heads were used with conventional polyethylene liners. These concerns led to the development of alternative bearing surfaces, particularly metal-on-metal bearings, which offered theoretical benefits of large-diameter articulations that improved stability while purportedly being highly wear-resistant.9-11 However, concerns about adverse local soft-tissue reactions and high blood concentrations of metal ions tempered the initial enthusiasm for metal bearings.12-16 Fortunately, highly cross-linked polyethylene and fourth-generation ceramic bearing surfaces, with improved toughness and better wear properties, may allow use of large-diameter heads without the need for metal-on-metal bearings.17,18
In this article, we review the concepts and principles behind use of large-diameter ceramic or cobalt-chromium femoral heads on polyethylene-bearing surfaces in THA with particular attention to biomechanics, early concerns about polyethylene wear and rim fractures, recent improvements in material properties of polyethylene and ceramic bearings, dislocation rates, and clinical and functional outcomes.
Definitions
For this review, we define large-diameter femoral heads as having diameters of 36 mm or more and conventional or small-diameter femoral heads as having diameters between 22 and 32 mm.
Biomechanics
Head–Neck Ratio, Impingement-Free ROM, and Jump Distance
Several implant design principles have been proposed to reduce the risks for impingement and dislocation. Of these, large femoral head diameters have been extensively studied.19,20 It is well known that impingement of the femoral neck on the cup edge promotes edge loading and higher wear rates. In addition, impingement increases the tendency of the head to sublux from the acetabulum. One strategy for avoiding this component-to-component impingement is to increase the head–neck ratio (HNR), the ratio of the femoral head to the neck diameter. Biomechanically, increased HNRs lead to delayed contact between the femoral neck and the acetabular liner.21,22 Therefore, with large femoral heads, which have large HNRs, impingement occurs later and at larger ROMs—compared with small-diameter femoral heads, which have lower HNRs and are more prone to early impingement and subluxation (Figure 1).23-26
In a cadaveric study of 6 hips, Bartz and colleagues23 reported a significantly higher preimpingement ROM when the prosthetic head size increased from 22 mm to 28 mm (P < .05). They found a change from prosthetic to osseous impingement when the head size increased from 22 mm to 32 mm. Similar results were observed in a computer simulation model by Cinotti and colleagues,27 who demonstrated that increasing the femoral head size from 28 mm to 38 mm resulted in a 5° improvement in ROM. However, the largest gains were observed when the heads with the smallest diameters were upsized; ROM improved only marginally when femoral head size was further increased from 32 mm to 38 mm. The primary reason for the lack of expected improvement in ROM with head sizes of more than 32 mm is often bone-on-bone impingement. Burroughs and colleagues28 demonstrated that the 38-mm and 44-mm heads virtually eliminated component-to-component impingement except in extremes of external rotation. However, there were no differences in ROM between 38-mm and 44-mm heads because of osseous impingement. In addition, large heads are less likely to sublux or dislocate, as they need to travel farther before reaching the edge of the acetabular cup before dislocation. This is known as the jump distance, and it corresponds to the depth of the acetabular shell, which in turn equates with the radius of the femoral head (Figures 2A, 2B). For this reason, the larger the femoral head diameter, the farther the jump distance and, correspondingly, the lower the risk for dislocation.29
Elevated liners historically were used to increase the jump distance for dislocation.30 These liners, however, can increase impingement at the extremes of motion.31 Some of these problems can be avoided with use of larger heads, which have increased jump distances without additional risks for impingement. Moreover, large heads create a suction effect that provides passive resistance to dislocation.32 With head diameters beyond 38 mm, impingement-free ROM often plateaus. However, the jump distance required for dislocations to occur continues to increase as femoral head diameters increase in size. Thus, patients may experience fewer motion benefits but continue to benefit from overall stability with femoral head sizes increasing beyond 38 mm.
Current evidence suggests there may be substantial benefits toward improved stability from increasing head diameters from 22 mm to 38 mm because of the increase in jump distances and improvements in prosthetic impingement-free ROM. However, there may be little gain in ROM from increasing the head diameters beyond these dimensions because of the potential risks of bony impingement. Nevertheless, there may be some additional benefits toward stability from improvement in jump distances with incremental head sizes
beyond 38 mm.29,33,34
Finite Element Analysis Studies
Finite element analysis of large-diameter heads in THA has shown that, at optimal cup inclination (45°), most stresses occur on the articular surface of the liner. However, these stresses remain well below the yield strength of the polyethylene liners.29 With increasing abduction angles, the stress concentration increases substantially because of the decreased contact surface area. At these angles, the point of maximum contact moves toward the rim of the polyethylene liner, which can lead to rim fractures or failure of locking mechanisms.29,35,36
Early Concerns With Large-Diameter Femoral Heads: Wear, Liner Failure, and Fracture of Ceramic Components
Use of small-diameter femoral heads started with the first report by Charnley37 of “low frictional torque arthroplasty.” Charnley initially considered a 41.5-mm femoral head, but he thought it would increase risks for acetabular loosening from high frictional torque generated by the large head, and he switched to a small-diameter (22.5 mm) design. One of the tradeoffs with smaller diameter heads was decreased jump height in addition to increased linear wear.
Large femoral heads used with cemented polyethylene acetabular components historically have been associated with increased rates of volumetric wear but low rates of linear wear, which potentially may increase the risk for osteolysis.38-40 However, newer highly cross-linked polyethylene liners have shown improved in vitro and in vivo volumetric wear characteristics and potentially lower linear wear rates compared with earlier designs (Table 1).28,41-43
Another concern about earlier generations of large femoral heads was the risk for catastrophic liner failure on conventional polyethylene. This was originally reported by Berry and colleagues,47 who described wear-through and failure in patients with thin (< 5 mm) acetabular cups. However, these concerns have been largely addressed by the development of highly cross-linked polyethylene, which has improved wear characteristics and fatigue resistance.48
Recent Improvements in Material Properties of Polyethylene and Ceramic Bearings
The development of highly cross-linked polyethylene and fourth-generation ceramics has renewed interest in large-diameter bearings in THA. These bearing surfaces improve wear, enhance material properties, and have superior oxidation resistance.42,48-53
We now briefly describe the methods used to improve the material properties of polyethylene and ceramics. Studies have shown that increasing the radiation dose (up to 200 kGy) increases cross-linking and causes an inverse exponential decrease in polyethylene wear.28,41,48-51 However, increasing radiation doses also increases production of free radicals, which diminish the material strength of these polyethylenes. The current generation of highly cross-linked polyethylene liners is produced through a variety of manufacturing strategies to improve cross-linking and reduce wear. These strategies include differential radiation doses (50-100 kGy), techniques (electron beam, radiation), and thermal treatments (melting, annealing). Moreover, to enhance the material properties and reduce the incidence of rim cracking and delamination, authors have proposed using vitamin E supplementation to minimize the amount of subsurface oxidation that occurs as an inevitable consequence of free radical formation during fabrication.54,55 A terminal sterilization process (eg, gas plasma, ethylene oxide, or gamma sterilization in nitrogen) is needed to make commercial, highly cross-linked polyethylene.52,53
Fourth-generation ceramics manufactured with nano-sized yttria-stabilized tetragonal zirconia particles in a stable alumina matrix have more fracture toughness and improved wear characteristics.54,55 In addition, oxide additives (eg, chromium oxide, strontium oxide) improve hardness and dissipate energy by deflecting cracks to prevent their propagation.56 Moreover, the smaller grain sizes of fourth-generation ceramic bearings compared with third-generation designs (0.8 µm vs 1-5 µm) cause less disruption of the fluid film layer, which ultimately results in improved wear performance.57
Multiple studies have found reduced wear rates with metal and ceramic large heads coupled with highly cross-linked polyethylene-bearings (Table 2).17,41,50,58 Bragdon and colleagues,58 using radiostereometric analysis in 25 patients, found no significant differences in mean head penetration rates between 36-mm and 28-mm cobalt-chromium (Co-Cr) heads articulating with highly cross-linked polyethylene cups at a mean follow-up of 3 years (0.035 mm/y vs 0.046 mm/y; P = .11). Geller and colleagues,64 in their study of 42 patients with large-diameter (> 32 mm) Co-Cr femoral heads, found low mean (SD) linear wear rates of 0.06 (0.41) mm/y at a mean follow-up of 3 years. D’Antonio and colleagues,65 in a multicenter study, reported low average linear wear (0.015 mm/y) and volumetric wear (12.1 mm3/y) over 5 years using sequentially annealed cross-linked polyethylene. In vitro reports suggest that large-diameter ceramic heads may have lower wear properties than Co-Cr heads do. Galvin and colleagues,66 in an in vitro hip simulator study, found that large-diameter ceramic heads on highly cross-linked ultrahigh-molecular-weight polyethylene had 40% reductions in steady-state wear rates compared with Co-Cr heads on highly cross-linked bearings (4.7 vs 8.1 mm3/million cycles; P < 0.01).
Dislocation Rates
Several patient, surgeon, and implant factors affect the rate of dislocations after THA. Multiple implant options utilize the biomechanical advantage that large-diameter heads have in improving stability. Various alternatives include use of constrained tripolar heads, dual-mobility bearings, and conventional large-diameter heads with standard liners.67-69
Large-Diameter Heads
Despite the biomechanical advantages of large-diameter metal-on-polyethylene bearings, prior studies have questioned use of these bearings because of risks for increased wear and rim failures. However, the improved wear properties of highly cross-linked polyethylene, elaborated earlier, have led to a reappraisal of this option (Table 2).4,70 Howie and colleagues,71 in a randomized control trial of 644 patients, also found significantly lower rates of dislocation after primary THA with 36-mm heads compared with 28-mm heads (1.3% vs 5.4%; P = .012); in addition, fewer dislocations occurred with 36-mm heads than with 28-mm heads (4.9% vs 12.2%; P = .27) in a series of 44 patients in revision settings. Similarly, in a study conducted with 39,271 Medicare patients between 1998 and 2007, Malkani and colleagues72 found a decrease in the dislocation rate, from 4.21% to 2.14%, with use of large-diameter femoral heads. These results have been confirmed by several other authors.34,66,73,74 Similar results were observed in 65,992 patients in the Australian National Joint Replacement Registry by Conroy and colleagues,75 who reported a significant decrease in the risk for dislocation with large heads (≥ 30 mm) compared with 22-mm heads (relative risk, 1.0 vs 3.1; P ≤ .001).
Few studies have analyzed the role of large-diameter femoral heads in the presence of compromised soft tissues around the hip. Kung and Ries,76 evaluating the influence of large-diameter heads in the presence and absence of a deficient abductor mechanism, demonstrated statistically significant reductions in rates of dislocation after 230 revision THAs when the abductor mechanism was intact with use of 36-mm heads compared with 28-mm heads (12.7% vs 0%; P = .015). With abductor deficiency, though, the positive effect of large heads in reducing dislocation rates was substantially reduced and was similar to that of small heads (P = .74).76
Large heads considerably improve overall stability and lower dislocation rates in THA. With the development of newer ceramics and highly cross-linked polyethylenes, the wear rates reported in multiple studies appear to be less concerning.
Constrained Tripolar Heads
Tripolar heads have been proposed as treatment options for improving stability in patients with chronic and recurrent instability after THA. The tripolar implant consists of a metal head that snap-fits into a polyethylene liner with a polished Co-Cr backing. This bipolar head articulates with a polyethylene bearing that is press-fitted onto a metal acetabular shell and constrained by a metal ring snapped to the outer polyethylene bearing. The bipolar component behaves as a large-diameter femoral head, and the metal ring provides additional restraint, further improving stability.
Williams and colleagues77 performed a systematic review and reported on the outcomes of constrained tripolar liners in 1199 hips at a mean follow-up of 4 years (range, 2-10 years). The mean dislocation rate was 10%, and the mean rate of revision surgery unrelated to instability was 4%. In a study of 43 hips at a mean follow-up of 4 years (range, 2-9 years), Zywiel and colleagues78 reported on the clinical and radiographic outcomes of tripolar constrained liners. Their study group had a mean Harris Hip Score (HHS) of 82 points (range, 38-100 points) and overall survival of 91%, with no evidence of radiographic loosening during follow-up. Despite the improvements in stability with constrained tripolar liners, some authors have reported multiple mechanisms of failure with these devices.79-81 In a study of 43 failed constrained tripolar liners with a mean time to failure of about 2 years, Guyen and colleagues79 identified 5 different failure modes (types 1-5) involving all 4 interfaces in these components.
Encouraging outcomes have been reported at midterm follow-up with tripolar constrained liners. However, concerns about failure at the interfaces suggest that use of these components should be restricted to patients with deficient abductor mechanisms or neuromuscular compromise, low-demand elderly patients, and salvage cases of recurrent dislocations.79
Dual-Mobility Bearings
For more than 20 years, different dual-mobility bearings have been used for difficult acetabular reconstructive scenarios and prevention of instability.82,83 Dual-mobility cups provide constructs that snap-fit a small-diameter femoral head within a large polyethylene insert that articulates with a fixed metal shell. This effectively increases the functional head diameter.
Various authors have reported excellent survivorship rates (92%-99%) and low dislocation rates for these bearings at 5- to 10-year follow-up.82,84-90 Philippot and colleagues,86 in a recent study of 438 hips with dual-mobility cups, reported excellent survivorship (96%) and no early or late instability within a 15-year follow-up. Bouchet and colleagues69 compared dual-mobility bearings (105 hips) with conventional metal-on-polythene bearings (108 hips) and found significantly (P < .05) lower dislocation rates for the dual-mobility implants at a minimum 1-year follow-up. The French Society of Orthopaedics and Traumatology performed a multicenter analysis of 3473 hips with dual-mobility cups implanted in France between January 1998 and December 2003.87 During a mean follow-up of 7 years (range, 5-11 years), there were 15 dislocations (0.43%), 14 of which occurred early, within 3 months of implantation (0.4%). Aseptic implant survivorship was 95% at 10-year follow-up.
Use of these bearings has recently increased in the United States. Short-term and midterm follow-up data show low rates of dislocation and wear. Long-term data are to come.
Clinical and Functional Outcomes of Large-Diameter Femoral Heads
There is a paucity of long-term outcomes data on use of large-diameter heads with highly cross-linked polyethylene bearings. Short-term and midterm clinical results appear to be excellent, with low rates of wear, osteolysis, and aseptic loosening.28,41,73,89-92
Plate and colleagues91 compared the effects of large-diameter (≥ 36 mm) and small-diameter (26 mm, 28 mm) metal heads on highly cross-linked polyethylene bearings. At a mean follow-up of 5 years (range, 4-8.4 years), the large-head cohort had a mean HHS of 90 points (range, 50-100 points) and no dislocations or radiographic evidence of stem or cup loosening. Similarly, Meftah and colleagues93 reported 100% stem survivorship and excellent clinical outcomes—a mean Western Ontario and McMaster Universities Arthritis Index (WOMAC) score of 30 points—for 72 hips with use of large ceramic heads (≥ 32 mm) on highly cross-linked polyethylene at a mean follow-up of 3 years. Gagala and colleagues94 reported excellent clinical and radiographic outcomes in 50 hips (18 ceramic on ceramic, 32 ceramic on polyethylene; 36-mm heads) at a mean follow-up of 3.5 years. Mean HHS was 94 points, and there was no evidence of liner fractures, aseptic loosening, or osteolysis.
In summary, large-diameter femoral heads in THA have become increasingly popular because of improvements in the material properties and wear characteristics of highly cross-linked polyethylene and fourth-generation ceramics. Despite the potential advantages of large heads in preventing dislocations, the basic surgical tenets of placing the acetabular component in appropriate alignment remain firmly established. Implants with functionally large heads (eg, dual-mobility bearings, constrained tripolar liners) may play an important role in patients at high risk for dislocation—particularly elderly patients with poor neuromuscular muscle coordination or deficient abductors, trauma patients, and patients with prior dislocations. Short-term and midterm results are excellent; rates of wear, aseptic loosening, and osteolysis are low. However, long-term outcomes data are needed to support widespread use of large heads in younger and more active patients.
A common cause for total hip arthroplasty (THA) revision is joint instability.1,2 The reported incidence of dislocation in primary THA ranges from 0.4% to 5.8%,3-5 but this rate increases after revision surgery.1,3-8 Use of large-diameter femoral heads has been proposed to decrease the risks for instability and to improve impingement-free range of motion (ROM).
The biomechanical rationale for using large-diameter femoral heads is that they must travel farther before subluxation or dislocation occurs (jump distance). Despite these benefits, there were initial concerns that catastrophic failure and high levels of volumetric wear would occur if these heads were used with conventional polyethylene liners. These concerns led to the development of alternative bearing surfaces, particularly metal-on-metal bearings, which offered theoretical benefits of large-diameter articulations that improved stability while purportedly being highly wear-resistant.9-11 However, concerns about adverse local soft-tissue reactions and high blood concentrations of metal ions tempered the initial enthusiasm for metal bearings.12-16 Fortunately, highly cross-linked polyethylene and fourth-generation ceramic bearing surfaces, with improved toughness and better wear properties, may allow use of large-diameter heads without the need for metal-on-metal bearings.17,18
In this article, we review the concepts and principles behind use of large-diameter ceramic or cobalt-chromium femoral heads on polyethylene-bearing surfaces in THA with particular attention to biomechanics, early concerns about polyethylene wear and rim fractures, recent improvements in material properties of polyethylene and ceramic bearings, dislocation rates, and clinical and functional outcomes.
Definitions
For this review, we define large-diameter femoral heads as having diameters of 36 mm or more and conventional or small-diameter femoral heads as having diameters between 22 and 32 mm.
Biomechanics
Head–Neck Ratio, Impingement-Free ROM, and Jump Distance
Several implant design principles have been proposed to reduce the risks for impingement and dislocation. Of these, large femoral head diameters have been extensively studied.19,20 It is well known that impingement of the femoral neck on the cup edge promotes edge loading and higher wear rates. In addition, impingement increases the tendency of the head to sublux from the acetabulum. One strategy for avoiding this component-to-component impingement is to increase the head–neck ratio (HNR), the ratio of the femoral head to the neck diameter. Biomechanically, increased HNRs lead to delayed contact between the femoral neck and the acetabular liner.21,22 Therefore, with large femoral heads, which have large HNRs, impingement occurs later and at larger ROMs—compared with small-diameter femoral heads, which have lower HNRs and are more prone to early impingement and subluxation (Figure 1).23-26
In a cadaveric study of 6 hips, Bartz and colleagues23 reported a significantly higher preimpingement ROM when the prosthetic head size increased from 22 mm to 28 mm (P < .05). They found a change from prosthetic to osseous impingement when the head size increased from 22 mm to 32 mm. Similar results were observed in a computer simulation model by Cinotti and colleagues,27 who demonstrated that increasing the femoral head size from 28 mm to 38 mm resulted in a 5° improvement in ROM. However, the largest gains were observed when the heads with the smallest diameters were upsized; ROM improved only marginally when femoral head size was further increased from 32 mm to 38 mm. The primary reason for the lack of expected improvement in ROM with head sizes of more than 32 mm is often bone-on-bone impingement. Burroughs and colleagues28 demonstrated that the 38-mm and 44-mm heads virtually eliminated component-to-component impingement except in extremes of external rotation. However, there were no differences in ROM between 38-mm and 44-mm heads because of osseous impingement. In addition, large heads are less likely to sublux or dislocate, as they need to travel farther before reaching the edge of the acetabular cup before dislocation. This is known as the jump distance, and it corresponds to the depth of the acetabular shell, which in turn equates with the radius of the femoral head (Figures 2A, 2B). For this reason, the larger the femoral head diameter, the farther the jump distance and, correspondingly, the lower the risk for dislocation.29
Elevated liners historically were used to increase the jump distance for dislocation.30 These liners, however, can increase impingement at the extremes of motion.31 Some of these problems can be avoided with use of larger heads, which have increased jump distances without additional risks for impingement. Moreover, large heads create a suction effect that provides passive resistance to dislocation.32 With head diameters beyond 38 mm, impingement-free ROM often plateaus. However, the jump distance required for dislocations to occur continues to increase as femoral head diameters increase in size. Thus, patients may experience fewer motion benefits but continue to benefit from overall stability with femoral head sizes increasing beyond 38 mm.
Current evidence suggests there may be substantial benefits toward improved stability from increasing head diameters from 22 mm to 38 mm because of the increase in jump distances and improvements in prosthetic impingement-free ROM. However, there may be little gain in ROM from increasing the head diameters beyond these dimensions because of the potential risks of bony impingement. Nevertheless, there may be some additional benefits toward stability from improvement in jump distances with incremental head sizes
beyond 38 mm.29,33,34
Finite Element Analysis Studies
Finite element analysis of large-diameter heads in THA has shown that, at optimal cup inclination (45°), most stresses occur on the articular surface of the liner. However, these stresses remain well below the yield strength of the polyethylene liners.29 With increasing abduction angles, the stress concentration increases substantially because of the decreased contact surface area. At these angles, the point of maximum contact moves toward the rim of the polyethylene liner, which can lead to rim fractures or failure of locking mechanisms.29,35,36
Early Concerns With Large-Diameter Femoral Heads: Wear, Liner Failure, and Fracture of Ceramic Components
Use of small-diameter femoral heads started with the first report by Charnley37 of “low frictional torque arthroplasty.” Charnley initially considered a 41.5-mm femoral head, but he thought it would increase risks for acetabular loosening from high frictional torque generated by the large head, and he switched to a small-diameter (22.5 mm) design. One of the tradeoffs with smaller diameter heads was decreased jump height in addition to increased linear wear.
Large femoral heads used with cemented polyethylene acetabular components historically have been associated with increased rates of volumetric wear but low rates of linear wear, which potentially may increase the risk for osteolysis.38-40 However, newer highly cross-linked polyethylene liners have shown improved in vitro and in vivo volumetric wear characteristics and potentially lower linear wear rates compared with earlier designs (Table 1).28,41-43
Another concern about earlier generations of large femoral heads was the risk for catastrophic liner failure on conventional polyethylene. This was originally reported by Berry and colleagues,47 who described wear-through and failure in patients with thin (< 5 mm) acetabular cups. However, these concerns have been largely addressed by the development of highly cross-linked polyethylene, which has improved wear characteristics and fatigue resistance.48
Recent Improvements in Material Properties of Polyethylene and Ceramic Bearings
The development of highly cross-linked polyethylene and fourth-generation ceramics has renewed interest in large-diameter bearings in THA. These bearing surfaces improve wear, enhance material properties, and have superior oxidation resistance.42,48-53
We now briefly describe the methods used to improve the material properties of polyethylene and ceramics. Studies have shown that increasing the radiation dose (up to 200 kGy) increases cross-linking and causes an inverse exponential decrease in polyethylene wear.28,41,48-51 However, increasing radiation doses also increases production of free radicals, which diminish the material strength of these polyethylenes. The current generation of highly cross-linked polyethylene liners is produced through a variety of manufacturing strategies to improve cross-linking and reduce wear. These strategies include differential radiation doses (50-100 kGy), techniques (electron beam, radiation), and thermal treatments (melting, annealing). Moreover, to enhance the material properties and reduce the incidence of rim cracking and delamination, authors have proposed using vitamin E supplementation to minimize the amount of subsurface oxidation that occurs as an inevitable consequence of free radical formation during fabrication.54,55 A terminal sterilization process (eg, gas plasma, ethylene oxide, or gamma sterilization in nitrogen) is needed to make commercial, highly cross-linked polyethylene.52,53
Fourth-generation ceramics manufactured with nano-sized yttria-stabilized tetragonal zirconia particles in a stable alumina matrix have more fracture toughness and improved wear characteristics.54,55 In addition, oxide additives (eg, chromium oxide, strontium oxide) improve hardness and dissipate energy by deflecting cracks to prevent their propagation.56 Moreover, the smaller grain sizes of fourth-generation ceramic bearings compared with third-generation designs (0.8 µm vs 1-5 µm) cause less disruption of the fluid film layer, which ultimately results in improved wear performance.57
Multiple studies have found reduced wear rates with metal and ceramic large heads coupled with highly cross-linked polyethylene-bearings (Table 2).17,41,50,58 Bragdon and colleagues,58 using radiostereometric analysis in 25 patients, found no significant differences in mean head penetration rates between 36-mm and 28-mm cobalt-chromium (Co-Cr) heads articulating with highly cross-linked polyethylene cups at a mean follow-up of 3 years (0.035 mm/y vs 0.046 mm/y; P = .11). Geller and colleagues,64 in their study of 42 patients with large-diameter (> 32 mm) Co-Cr femoral heads, found low mean (SD) linear wear rates of 0.06 (0.41) mm/y at a mean follow-up of 3 years. D’Antonio and colleagues,65 in a multicenter study, reported low average linear wear (0.015 mm/y) and volumetric wear (12.1 mm3/y) over 5 years using sequentially annealed cross-linked polyethylene. In vitro reports suggest that large-diameter ceramic heads may have lower wear properties than Co-Cr heads do. Galvin and colleagues,66 in an in vitro hip simulator study, found that large-diameter ceramic heads on highly cross-linked ultrahigh-molecular-weight polyethylene had 40% reductions in steady-state wear rates compared with Co-Cr heads on highly cross-linked bearings (4.7 vs 8.1 mm3/million cycles; P < 0.01).
Dislocation Rates
Several patient, surgeon, and implant factors affect the rate of dislocations after THA. Multiple implant options utilize the biomechanical advantage that large-diameter heads have in improving stability. Various alternatives include use of constrained tripolar heads, dual-mobility bearings, and conventional large-diameter heads with standard liners.67-69
Large-Diameter Heads
Despite the biomechanical advantages of large-diameter metal-on-polyethylene bearings, prior studies have questioned use of these bearings because of risks for increased wear and rim failures. However, the improved wear properties of highly cross-linked polyethylene, elaborated earlier, have led to a reappraisal of this option (Table 2).4,70 Howie and colleagues,71 in a randomized control trial of 644 patients, also found significantly lower rates of dislocation after primary THA with 36-mm heads compared with 28-mm heads (1.3% vs 5.4%; P = .012); in addition, fewer dislocations occurred with 36-mm heads than with 28-mm heads (4.9% vs 12.2%; P = .27) in a series of 44 patients in revision settings. Similarly, in a study conducted with 39,271 Medicare patients between 1998 and 2007, Malkani and colleagues72 found a decrease in the dislocation rate, from 4.21% to 2.14%, with use of large-diameter femoral heads. These results have been confirmed by several other authors.34,66,73,74 Similar results were observed in 65,992 patients in the Australian National Joint Replacement Registry by Conroy and colleagues,75 who reported a significant decrease in the risk for dislocation with large heads (≥ 30 mm) compared with 22-mm heads (relative risk, 1.0 vs 3.1; P ≤ .001).
Few studies have analyzed the role of large-diameter femoral heads in the presence of compromised soft tissues around the hip. Kung and Ries,76 evaluating the influence of large-diameter heads in the presence and absence of a deficient abductor mechanism, demonstrated statistically significant reductions in rates of dislocation after 230 revision THAs when the abductor mechanism was intact with use of 36-mm heads compared with 28-mm heads (12.7% vs 0%; P = .015). With abductor deficiency, though, the positive effect of large heads in reducing dislocation rates was substantially reduced and was similar to that of small heads (P = .74).76
Large heads considerably improve overall stability and lower dislocation rates in THA. With the development of newer ceramics and highly cross-linked polyethylenes, the wear rates reported in multiple studies appear to be less concerning.
Constrained Tripolar Heads
Tripolar heads have been proposed as treatment options for improving stability in patients with chronic and recurrent instability after THA. The tripolar implant consists of a metal head that snap-fits into a polyethylene liner with a polished Co-Cr backing. This bipolar head articulates with a polyethylene bearing that is press-fitted onto a metal acetabular shell and constrained by a metal ring snapped to the outer polyethylene bearing. The bipolar component behaves as a large-diameter femoral head, and the metal ring provides additional restraint, further improving stability.
Williams and colleagues77 performed a systematic review and reported on the outcomes of constrained tripolar liners in 1199 hips at a mean follow-up of 4 years (range, 2-10 years). The mean dislocation rate was 10%, and the mean rate of revision surgery unrelated to instability was 4%. In a study of 43 hips at a mean follow-up of 4 years (range, 2-9 years), Zywiel and colleagues78 reported on the clinical and radiographic outcomes of tripolar constrained liners. Their study group had a mean Harris Hip Score (HHS) of 82 points (range, 38-100 points) and overall survival of 91%, with no evidence of radiographic loosening during follow-up. Despite the improvements in stability with constrained tripolar liners, some authors have reported multiple mechanisms of failure with these devices.79-81 In a study of 43 failed constrained tripolar liners with a mean time to failure of about 2 years, Guyen and colleagues79 identified 5 different failure modes (types 1-5) involving all 4 interfaces in these components.
Encouraging outcomes have been reported at midterm follow-up with tripolar constrained liners. However, concerns about failure at the interfaces suggest that use of these components should be restricted to patients with deficient abductor mechanisms or neuromuscular compromise, low-demand elderly patients, and salvage cases of recurrent dislocations.79
Dual-Mobility Bearings
For more than 20 years, different dual-mobility bearings have been used for difficult acetabular reconstructive scenarios and prevention of instability.82,83 Dual-mobility cups provide constructs that snap-fit a small-diameter femoral head within a large polyethylene insert that articulates with a fixed metal shell. This effectively increases the functional head diameter.
Various authors have reported excellent survivorship rates (92%-99%) and low dislocation rates for these bearings at 5- to 10-year follow-up.82,84-90 Philippot and colleagues,86 in a recent study of 438 hips with dual-mobility cups, reported excellent survivorship (96%) and no early or late instability within a 15-year follow-up. Bouchet and colleagues69 compared dual-mobility bearings (105 hips) with conventional metal-on-polythene bearings (108 hips) and found significantly (P < .05) lower dislocation rates for the dual-mobility implants at a minimum 1-year follow-up. The French Society of Orthopaedics and Traumatology performed a multicenter analysis of 3473 hips with dual-mobility cups implanted in France between January 1998 and December 2003.87 During a mean follow-up of 7 years (range, 5-11 years), there were 15 dislocations (0.43%), 14 of which occurred early, within 3 months of implantation (0.4%). Aseptic implant survivorship was 95% at 10-year follow-up.
Use of these bearings has recently increased in the United States. Short-term and midterm follow-up data show low rates of dislocation and wear. Long-term data are to come.
Clinical and Functional Outcomes of Large-Diameter Femoral Heads
There is a paucity of long-term outcomes data on use of large-diameter heads with highly cross-linked polyethylene bearings. Short-term and midterm clinical results appear to be excellent, with low rates of wear, osteolysis, and aseptic loosening.28,41,73,89-92
Plate and colleagues91 compared the effects of large-diameter (≥ 36 mm) and small-diameter (26 mm, 28 mm) metal heads on highly cross-linked polyethylene bearings. At a mean follow-up of 5 years (range, 4-8.4 years), the large-head cohort had a mean HHS of 90 points (range, 50-100 points) and no dislocations or radiographic evidence of stem or cup loosening. Similarly, Meftah and colleagues93 reported 100% stem survivorship and excellent clinical outcomes—a mean Western Ontario and McMaster Universities Arthritis Index (WOMAC) score of 30 points—for 72 hips with use of large ceramic heads (≥ 32 mm) on highly cross-linked polyethylene at a mean follow-up of 3 years. Gagala and colleagues94 reported excellent clinical and radiographic outcomes in 50 hips (18 ceramic on ceramic, 32 ceramic on polyethylene; 36-mm heads) at a mean follow-up of 3.5 years. Mean HHS was 94 points, and there was no evidence of liner fractures, aseptic loosening, or osteolysis.
In summary, large-diameter femoral heads in THA have become increasingly popular because of improvements in the material properties and wear characteristics of highly cross-linked polyethylene and fourth-generation ceramics. Despite the potential advantages of large heads in preventing dislocations, the basic surgical tenets of placing the acetabular component in appropriate alignment remain firmly established. Implants with functionally large heads (eg, dual-mobility bearings, constrained tripolar liners) may play an important role in patients at high risk for dislocation—particularly elderly patients with poor neuromuscular muscle coordination or deficient abductors, trauma patients, and patients with prior dislocations. Short-term and midterm results are excellent; rates of wear, aseptic loosening, and osteolysis are low. However, long-term outcomes data are needed to support widespread use of large heads in younger and more active patients.
1. Dorr LD, Wolf AW, Chandler R, Conaty JP. Classification and treatment of dislocations of total hip arthroplasty. Clin Orthop. 1983;(173):151-158.
2. Dorr LD, Wan Z. Causes of and treatment protocol for instability of total hip replacement. Clin Orthop. 1998;(355):144-151.
3. Turner RS. Postoperative total hip prosthetic femoral head dislocations. Incidence, etiologic factors, and management. Clin Orthop. 1994;(301):196-204.
4. Woo RY, Morrey BF. Dislocations after total hip arthroplasty. J Bone Joint Surg Am. 1982;64(9):1295-1306.
5. Etienne A, Cupic Z, Charnley J. Postoperative dislocation after Charnley low-friction arthroplasty. Clin Orthop. 1978;(132):19-23.
6. Fackler CD, Poss R. Dislocation in total hip arthroplasties. Clin Orthop. 1980;(151):169-178.
7. Joshi A, Lee CM, Markovic L, Vlatis G, Murphy JC. Prognosis of dislocation after total hip arthroplasty. J Arthroplasty. 1998;13(1):17-21.
8. Lindberg HO, Carlsson AS, Gentz CF, Pettersson H. Recurrent and non-recurrent dislocation following total hip arthroplasty. Acta Orthop Scand. 1982;53(6):947-952.
9. Eswaramoorthy V, Moonot P, Kalairajah Y, Biant LC, Field RE. The Metasul metal-on-metal articulation in primary total hip replacement: clinical and radiological results at ten years. J Bone Joint Surg Br. 2008;90(10):1278-1283.
10. Grubl A, Marker M, Brodner W, et al. Long-term follow-up of metal-on-metal total hip replacement. J Orthop Res. 2007;25(7):841-848.
11. Leslie I, Williams S, Brown C, et al. Effect of bearing size on the long-term wear, wear debris, and ion levels of large diameter metal-on-metal hip replacements—an in vitro study. J Biomed Mater Res B Appl Biomater. 2008;87(1):163-172.
12. Verhaar JA. The hard lesson of metal-on-metal hip implants [in Dutch]. Ned Tijdschr Geneeskd. 2012;156(42):A5564.
13. Fabi D, Levine B, Paprosky W, et al. Metal-on-metal total hip arthroplasty: causes and high incidence of early failure. Orthopedics. 2012;35(7):e1009-e1016.
14. Heneghan C, Langton D, Thompson M. Ongoing problems with metal-on-metal hip implants. BMJ. 2012;344:e1349.
15. Lee RK, Nevelos J, Vigdorchik J, Markel DC. Bearing surfaces for hip arthroplasty—is metal-on-metal a passing fancy? Surg Technol Int. 2012;22:243-249.
16. Voleti PB, Baldwin KD, Lee GC. Metal-on-metal vs conventional total hip arthroplasty: a systematic review and meta-analysis of randomized controlled trials. J Arthroplasty. 2012;27(10):1844-1849.
17. Urban JA, Garvin KL, Boese CK, et al. Ceramic-on-polyethylene bearing surfaces in total hip arthroplasty. Seventeen to twenty-one-year results.
J Bone Joint Surg Am. 2001;83(11):1688-1694.
18. Callaghan JJ, Liu SS. Ceramic on crosslinked polyethylene in total hip replacement: any better than metal on crosslinked polyethylene? Iowa Orthop J. 2009;29:1-4.
19. Barrack RL. Dislocation after total hip arthroplasty: implant design and orientation. J Am Acad Orthop Surg. 2003;11(2):89-99.
20. Krushell RJ, Burke DW, Harris WH. Elevated-rim acetabular components. Effect on range of motion and stability in total hip arthroplasty. J Arthroplasty. 1991;6(suppl):S53-S58.
21. Morrey BF. Instability after total hip arthroplasty. Orthop Clin North Am. 1992;23(2):237-248.
22. Morrey BF. Dislocation. In: Morrey BF, ed. Joint Replacement Arthroplasty. New York, NY: Churchill Livingstone; 1991:851-865.
23. Bartz RL, Nobel PC, Kadakia NR, Tullos HS. The effect of femoral component head size on posterior dislocation of the artificial hip joint. J Bone Joint Surg Am. 2000;82(9):1300-1307.
24. Nicholas RM, Orr JF, Mollan RA, Calderwood JW, Nixon JR, Watson P. Dislocation of total hip replacements. A comparative study of standard, long posterior wall and augmented acetabular components. J Bone Joint Surg Br. 1990;72(3):418-422.
25. McCollum DE, Gray WJ. Dislocation after total hip arthroplasty. Causes and prevention. Clin Orthop. 1990;(261):159-170.
26. Herrlin K, Selvik G, Pettersson H, Kesek P, Onnerfalt R, Ohlin A. Position, orientation and component interaction in dislocation of the total hip prosthesis. Acta Radiol. 1988;29(4):441-444.
27. Cinotti G, Lucioli N, Malagoli A, Calderoli C, Cassese F. Do large femoral heads reduce the risks of impingement in total hip arthroplasty with optimal and non-optimal cup positioning? Int Orthop. 2011;35(3):317-323.
28. Burroughs BR, Rubash HE, Harris WH. Femoral head sizes larger than 32 mm against highly cross-linked polyethylene. Clin Orthop. 2002;(405):150-157.
29. Crowninshield RD, Maloney WJ, Wentz DH, Humphrey SM, Blanchard CR. Biomechanics of large femoral heads: what they do and don‘t do. Clin Orthop. 2004;(429):102-107.
30. Charnley J. Low Friction Arthroplasty of the Hip: Theory and Practice. New York, NY: Springer; 1979.
31. Yamaguchi M, Akisue T, Bauer TW, Hashimoto Y. The spatial location of impingement in total hip arthroplasty. J Arthroplasty. 2000;15(3):305-313.
32. Peters CL, McPherson E, Jackson JD, Erickson JA. Reduction in early dislocation rate with large-diameter femoral heads in primary total hip arthroplasty. J Arthroplasty. 2007;22(6 suppl 2):140-144.
33. Masonis JL, Bourne RB. Surgical approach, abductor function, and total hip arthroplasty dislocation. Clin Orthop. 2002;(405):46-53.
34. Beaule PE, Schmalzried TP, Udomkiat P, Amstutz HC. Jumbo femoral head for the treatment of recurrent dislocation following total hip replacement. J Bone Joint Surg Am. 2002;84(2):256-263.
35. Oral E, Malhi AS, Muratoglu OK. Mechanisms of decrease in fatigue crack propagation resistance in irradiated and melted UHMWPE. Biomaterials. 2006;27(6):917-925.
36. Baker DA, Bellare A, Pruitt L. The effects of degree of crosslinking on the fatigue crack initiation and propagation resistance of orthopedic-grade polyethylene. J Biomed Mater Res A. 2003;66(1):146-154.
37. Charnley J. Total hip replacement by low-friction arthroplasty. Clin Orthop. 1970;(72):7-21.
38. Kabo JM, Gebhard JS, Loren G, Amstutz HC. In vivo wear of polyethylene acetabular components. J Bone Joint Surg Br. 1993;75(2):254-258.
39. Livermore J, Ilstrup D, Morrey B. Effect of femoral head size on wear of the polyethylene acetabular component. J Bone Joint Surg Am. 1990;72(4):518-528.
40. Ma SM, Kabo JM, Amstutz HC. Frictional torque in surface and conventional hip replacement. J Bone Joint Surg Am. 1983;65(3):366-370.
41. Muratoglu OK, Bragdon CR, O‘Connor D, et al. Larger diameter femoral heads used in conjunction with a highly cross-linked ultra-high molecular weight polyethylene: a new concept. J Arthroplasty. 2001;16(8 suppl 1):24-30.
42. Thomas GER, Simpson DJ, Mehmood S, et al. The seven-year wear of highly cross-linked polyethylene in total hip arthroplasty: a double-blind, randomized controlled trial using radiostereometric analysis. J Bone Joint Surg Am. 2011;93(8):716-722.
43. Mutimer J, Devane PA, Adams K, Horne JG. Highly crosslinked polyethylene reduces wear in total hip arthroplasty at 5 years. Clin Orthop. 2010;468(12):3228-3233.
44. Bragdon CR, Doerner M, Martell J, Jarrett B, Palm H, Malchau H. The 2012 John Charnley Award: clinical multicenter studies of the wear performance of highly crosslinked remelted polyethylene in THA. Clin Orthop. 2013;471(2):393-402.
45. Lachiewicz PF, Heckman DS, Soileau ES, Mangla J, Martell JM. Femoral head size and wear of highly cross-linked polyethylene at 5 to 8 years. Clin Orthop. 2009;467(12):3290-3296.
46. Sychterz CJ, Engh CA Jr, Young AM, Hopper RH Jr, Engh CA. Comparison of in vivo wear between polyethylene liners articulating with ceramic and cobalt-chrome femoral heads. J Bone Joint Surg Br. 2000;82(7):948-951.
47. Berry DJ, Barnes CL, Scott RD, Cabanela ME, Poss R. Catastrophic failure of the polyethylene liner of uncemented acetabular components.
J Bone Joint Surg Br. 1994;76(4):575-578.
48. McKellop H, Shen FW, Lu B, Campbell P, Salovey R. Development of an extremely wear-resistant ultra high molecular weight polyethylene for total hip replacements. J Orthop Res. 1999;17(2):157-167.
49. Wang A, Essner A, Polineni VK, Stark C, Dumbleton JH. Lubrication and wear of ultra-high molecular weight polyethylene in total joint replacements. Tribol Int. 1998;31(1-3):17-33.
50. Estok DM 2nd, Burroughs BR, Muratoglu OK, Harris WH. Comparison of hip simulator wear of 2 different highly cross-linked ultra high molecular weight polyethylene acetabular components using both 32- and 38-mm femoral heads. J Arthroplasty. 2007;22(4):581-589.
51. Muratoglu OK, Bragdon CR, O‘Connor DO, et al. Unified wear model for highly crosslinked ultra-high molecular weight polyethylenes (UHMWPE). Biomaterials. 1999;20(16):1463-1470.
52. Harris WH, Muratoglu OK. A review of current cross-linked polyethylenes used in total joint arthroplasty. Clin Orthop. 2005;(430):46-52.
53. Muratoglu OK, Bragdon CR, O‘Connor DO, Jasty M, Harris WH. A novel method of cross-linking ultra-high-molecular-weight polyethylene to improve wear, reduce oxidation, and retain mechanical properties. Recipient of the 1999 HAP Paul Award. J Arthroplasty. 2001;16(2):149-160.
54. Bal BS, Garino J, Ries M, Rahaman MN. A review of ceramic bearing materials in total joint arthroplasty. Hip Int. 2007;17(1):21-30.
55. Traina F, De Fine M, Di Martino A, Faldini C. Fracture of ceramic bearing surfaces following total hip replacement: a systematic review. Biomed Res Int. 2013;2013:157247.
56. Cai YZ, Yan SG. Development of ceramic-on-ceramic implants for total hip arthroplasty. Orthop Surg. 2010;2(3):175-181.
57. Stewart TD, Tipper JL, Insley G, Streicher RM, Ingham E, Fisher J. Long-term wear of ceramic matrix composite materials for hip prostheses under severe swing phase microseparation. J Biomed Mater Res B Appl Biomater. 2003;66(2):567-573.
58. Bragdon CR, Greene ME, Freiberg AA, Harris WH, Malchau H. Radiostereometric analysis comparison of wear of highly cross-linked polyethylene against 36- vs 28-mm femoral heads. J Arthroplasty. 2007;22(6 suppl 2):125-129.
59. Lombardi AV Jr, Skeels MD, Berend KR, Adams JB, Franchi OJ. Do large heads enhance stability and restore native anatomy in primary total hip arthroplasty? Clin Orthop. 2011;469(6):1547-1553.
60. Lachiewicz PF, Soileau ES. Low early and late dislocation rates with 36- and 40-mm heads in patients at high risk for dislocation. Clin Orthop. 2013;471(2):439-443.
61. Cai P, Hu Y, Xie J. Large-diameter Delta ceramic-on-ceramic versus common-sized ceramic-on-polyethylene bearings in THA. Orthopedics. 2012;35(9):e1307-e1313.
62. Park KS, Yoon TR, Hwang SY, Lee KB. Modified minimally invasive two-incision total hip arthroplasty using large diameter femoral head. Indian
J Orthop. 2012;46(1):29-35.
63. Garbuz DS, Masri BA, Duncan CP, et al. The Frank Stinchfield Award: dislocation in revision THA: do large heads (36 and 40 mm) result in reduced dislocation rates in a randomized clinical trial? Clin Orthop. 2012;470(2):351-356.
64. Geller JA, Malchau H, Bragdon C, Greene M, Harris WH, Freiberg AA. Large diameter femoral heads on highly cross-linked polyethylene: minimum 3-year results. Clin Orthop. 2006;(447):53-59.
65. D’Antonio JA, Capello WN, Ramakrishnan R. Second-generation annealed highly cross-linked polyethylene exhibits low wear. Clin Orthop. 2012;470(6):1696-1704.
66. Galvin AL, Jennings LM, Tipper JL, Ingham E, Fisher J. Wear and creep of highly crosslinked polyethylene against cobalt chrome and ceramic femoral heads. Proc Inst Mech Eng H. 2010;224(10):1175-1183.
67. Skeels MD, Berend KR, Lombardi AV Jr. The dislocator, early and late: the role of large heads. Orthopedics. 2009;32(9).
68. Plate JF, Seyler TM, Stroh DA, Issa K, Akbar M, Mont MA. Risk of dislocation using large- vs. small-diameter femoral heads in total hip arthroplasty. BMC Res Notes. 2012;5(1):553.
69. Bouchet R, Mercier N, Saragaglia D. Posterior approach and dislocation rate: a 213 total hip replacements case–control study comparing the dual mobility cup with a conventional 28-mm metal head/polyethylene prosthesis. Orthop Traumatol Surg Res. 2011;97(1):2-7.
70. Ali Khan MA, Brakenbury PH, Reynolds IS. Dislocation following total hip replacement. J Bone Joint Surg Br. 1981;63(2):214-218.
71. Howie DW, Holubowycz OT, Middleton R. Large femoral heads decrease the incidence of dislocation after total hip arthroplasty: a randomized controlled trial. J Bone Joint Surg Am. 2012;94(12):1095-1102.
72. Malkani AL, Ong KL, Lau E, Kurtz SM, Justice BJ, Manley MT. Early- and late-term dislocation risk after primary hip arthroplasty in the Medicare population. J Arthroplasty. 2010;25(6 suppl):21-25.
73. Berry DJ, von Knoch M, Schleck CD, Harmsen WS. Effect of femoral head diameter and operative approach on risk of dislocation after primary total hip arthroplasty. J Bone Joint Surg Am. 2005;87(11):2456-2463.
74. Cho MR, Lee HS, Lee SW, Choi CH, Kim SK, Ko SB. Results after total hip arthroplasty with a large head and bipolar arthroplasty in patients with displaced femoral neck fractures. J Arthroplasty. 2011;26(6):893-896.
75. Conroy JL, Whitehouse SL, Graves SE, Pratt NL, Ryan P, Crawford RW. Risk factors for revision for early dislocation in total hip arthroplasty.
J Arthroplasty. 2008;23(6):867-872.
76. Kung PL, Ries MD. Effect of femoral head size and abductors on dislocation after revision THA. Clin Orthop. 2007;(465):170-174.
77. Williams JT Jr, Ragland PS, Clarke S. Constrained components for the unstable hip following total hip arthroplasty: a literature review. Int Orthop. 2007;31(3):273-277.
78. Zywiel MG, Mustafa LH, Bonutti PM, Mont MA. Are abductor muscle quality and previous revision surgery predictors of constrained liner failure in hip arthroplasty? Int Orthop. 2011;35(6):797-802.
79. Guyen O, Lewallen DG, Cabanela ME. Modes of failure of Osteonics constrained tripolar implants: a retrospective analysis of forty-three failed implants. J Bone Joint Surg Am. 2008;90(7):1553-1560.
80. Banks LN, McElwain JP. An unusual mode of failure of a tripolar constrained acetabular liner: a case report. Arch Orthop Trauma Surg. 2010;130(4):503-505.
81. Robertson WJ, Mattern CJ, Hur J, Su EP, Pellicci PM. Failure mechanisms and closed reduction of a constrained tripolar acetabular liner.
J Arthroplasty. 2009;24(2):322.e5-e11.
82. Aubriot JH, Lesimple P, Leclercq S. Study of Bousquet‘s non-cemented acetabular implant in 100 hybrid total hip prostheses (Charnley type cemented femoral component). Average 5-year follow-up [in French]. Acta Orthop Belg. 1993;59(suppl 1):267-271.
83. Farizon F, de Lavison R, Azoulai JJ, Bousquet G. Results with a cementless alumina-coated cup with dual mobility. A twelve-year follow-up study. Int Orthop. 1998;22(4):219-224.
84. Mertl P, Combes A, Leiber-Wackenheim F, Fessy MH, Girard J, Migaud H. Recurrence of dislocation following total hip arthroplasty revision using dual mobility cups was rare in 180 hips followed over 7 years. HSS J. 2012;8(3):251-256.
85. Langlais FL, Ropars M, Gaucher F, Musset T, Chaix O. Dual mobility cemented cups have low dislocation rates in THA revisions. Clin Orthop. 2008;466(2):389-395.
86. Philippot R, Farizon F, Camilleri JP, et al. Survival of dual mobility socket with a mean 17 years follow-up [in French]. Rev Chir Orthop Reparatrice Appar Mot. 2008;94(1):43-48.
87. Adam P, Philippe R, Ehlinger M, et al. Dual mobility cups hip arthroplasty as a treatment for displaced fracture of the femoral neck in the elderly.
A prospective, systematic, multicenter study with specific focus on postoperative dislocation. Orthop Traumatol Surg Res. 2012;98(3):296-300.
88. Fessy MH. La double mobilité [Dual mobility]. Revue de Chirurgie Orthopédique et Traumatologique. 2010;96(7):891-898.
89. Mont MA, Issa K, Naziri Q, Harwin SF, Delanois RE, Johnson AJ. The use of dual-mobility bearings in difficult hip arthroplasty reconstructive cases. Surg Technol Int. 2011;21:234-240.
90. Sayeed SA, Mont MA, Costa CR, et al. Early outcomes of sequentially cross-linked thin polyethylene liners with large diameter femoral heads in total hip arthroplasty. Bull NYU Hosp Jt Dis. 2011;69(suppl 1):S90-S94.
91. Plate JF, Seyler TM, Stroh DA, Issa K, Akbar M, Mont MA. Risk of dislocation using large- vs. small-diameter femoral heads in total hip arthroplasty. BMC Res Notes. 2012;5(1):553.
92. Sato T, Nakashima Y, Akiyama M, et al. Wear resistant performance of highly cross-linked and annealed ultra-high molecular weight polyethylene against ceramic heads in total hip arthroplasty. J Orthop Res. 2012;30(12):2031-2037.
93. Meftah M, Ebrahimpour PB, He C, Ranawat AS, Ranawat CS. Preliminary clinical and radiographic results of large ceramic heads on highly cross-linked polyethylene. Orthopedics. 2011;34(6):133.
94. Gagala J, Mazurkiewicz T, Dajewski Z. Large diameter femoral heads in primary alumina/alumina and XSPE/alumina total hip arthroplasty.
A follow-up study of 50 hips after average 40 months and review of literature [in Polish]. Chir Narzadow Ruchu Ortop Pol. 2011;76(1):14-20.
1. Dorr LD, Wolf AW, Chandler R, Conaty JP. Classification and treatment of dislocations of total hip arthroplasty. Clin Orthop. 1983;(173):151-158.
2. Dorr LD, Wan Z. Causes of and treatment protocol for instability of total hip replacement. Clin Orthop. 1998;(355):144-151.
3. Turner RS. Postoperative total hip prosthetic femoral head dislocations. Incidence, etiologic factors, and management. Clin Orthop. 1994;(301):196-204.
4. Woo RY, Morrey BF. Dislocations after total hip arthroplasty. J Bone Joint Surg Am. 1982;64(9):1295-1306.
5. Etienne A, Cupic Z, Charnley J. Postoperative dislocation after Charnley low-friction arthroplasty. Clin Orthop. 1978;(132):19-23.
6. Fackler CD, Poss R. Dislocation in total hip arthroplasties. Clin Orthop. 1980;(151):169-178.
7. Joshi A, Lee CM, Markovic L, Vlatis G, Murphy JC. Prognosis of dislocation after total hip arthroplasty. J Arthroplasty. 1998;13(1):17-21.
8. Lindberg HO, Carlsson AS, Gentz CF, Pettersson H. Recurrent and non-recurrent dislocation following total hip arthroplasty. Acta Orthop Scand. 1982;53(6):947-952.
9. Eswaramoorthy V, Moonot P, Kalairajah Y, Biant LC, Field RE. The Metasul metal-on-metal articulation in primary total hip replacement: clinical and radiological results at ten years. J Bone Joint Surg Br. 2008;90(10):1278-1283.
10. Grubl A, Marker M, Brodner W, et al. Long-term follow-up of metal-on-metal total hip replacement. J Orthop Res. 2007;25(7):841-848.
11. Leslie I, Williams S, Brown C, et al. Effect of bearing size on the long-term wear, wear debris, and ion levels of large diameter metal-on-metal hip replacements—an in vitro study. J Biomed Mater Res B Appl Biomater. 2008;87(1):163-172.
12. Verhaar JA. The hard lesson of metal-on-metal hip implants [in Dutch]. Ned Tijdschr Geneeskd. 2012;156(42):A5564.
13. Fabi D, Levine B, Paprosky W, et al. Metal-on-metal total hip arthroplasty: causes and high incidence of early failure. Orthopedics. 2012;35(7):e1009-e1016.
14. Heneghan C, Langton D, Thompson M. Ongoing problems with metal-on-metal hip implants. BMJ. 2012;344:e1349.
15. Lee RK, Nevelos J, Vigdorchik J, Markel DC. Bearing surfaces for hip arthroplasty—is metal-on-metal a passing fancy? Surg Technol Int. 2012;22:243-249.
16. Voleti PB, Baldwin KD, Lee GC. Metal-on-metal vs conventional total hip arthroplasty: a systematic review and meta-analysis of randomized controlled trials. J Arthroplasty. 2012;27(10):1844-1849.
17. Urban JA, Garvin KL, Boese CK, et al. Ceramic-on-polyethylene bearing surfaces in total hip arthroplasty. Seventeen to twenty-one-year results.
J Bone Joint Surg Am. 2001;83(11):1688-1694.
18. Callaghan JJ, Liu SS. Ceramic on crosslinked polyethylene in total hip replacement: any better than metal on crosslinked polyethylene? Iowa Orthop J. 2009;29:1-4.
19. Barrack RL. Dislocation after total hip arthroplasty: implant design and orientation. J Am Acad Orthop Surg. 2003;11(2):89-99.
20. Krushell RJ, Burke DW, Harris WH. Elevated-rim acetabular components. Effect on range of motion and stability in total hip arthroplasty. J Arthroplasty. 1991;6(suppl):S53-S58.
21. Morrey BF. Instability after total hip arthroplasty. Orthop Clin North Am. 1992;23(2):237-248.
22. Morrey BF. Dislocation. In: Morrey BF, ed. Joint Replacement Arthroplasty. New York, NY: Churchill Livingstone; 1991:851-865.
23. Bartz RL, Nobel PC, Kadakia NR, Tullos HS. The effect of femoral component head size on posterior dislocation of the artificial hip joint. J Bone Joint Surg Am. 2000;82(9):1300-1307.
24. Nicholas RM, Orr JF, Mollan RA, Calderwood JW, Nixon JR, Watson P. Dislocation of total hip replacements. A comparative study of standard, long posterior wall and augmented acetabular components. J Bone Joint Surg Br. 1990;72(3):418-422.
25. McCollum DE, Gray WJ. Dislocation after total hip arthroplasty. Causes and prevention. Clin Orthop. 1990;(261):159-170.
26. Herrlin K, Selvik G, Pettersson H, Kesek P, Onnerfalt R, Ohlin A. Position, orientation and component interaction in dislocation of the total hip prosthesis. Acta Radiol. 1988;29(4):441-444.
27. Cinotti G, Lucioli N, Malagoli A, Calderoli C, Cassese F. Do large femoral heads reduce the risks of impingement in total hip arthroplasty with optimal and non-optimal cup positioning? Int Orthop. 2011;35(3):317-323.
28. Burroughs BR, Rubash HE, Harris WH. Femoral head sizes larger than 32 mm against highly cross-linked polyethylene. Clin Orthop. 2002;(405):150-157.
29. Crowninshield RD, Maloney WJ, Wentz DH, Humphrey SM, Blanchard CR. Biomechanics of large femoral heads: what they do and don‘t do. Clin Orthop. 2004;(429):102-107.
30. Charnley J. Low Friction Arthroplasty of the Hip: Theory and Practice. New York, NY: Springer; 1979.
31. Yamaguchi M, Akisue T, Bauer TW, Hashimoto Y. The spatial location of impingement in total hip arthroplasty. J Arthroplasty. 2000;15(3):305-313.
32. Peters CL, McPherson E, Jackson JD, Erickson JA. Reduction in early dislocation rate with large-diameter femoral heads in primary total hip arthroplasty. J Arthroplasty. 2007;22(6 suppl 2):140-144.
33. Masonis JL, Bourne RB. Surgical approach, abductor function, and total hip arthroplasty dislocation. Clin Orthop. 2002;(405):46-53.
34. Beaule PE, Schmalzried TP, Udomkiat P, Amstutz HC. Jumbo femoral head for the treatment of recurrent dislocation following total hip replacement. J Bone Joint Surg Am. 2002;84(2):256-263.
35. Oral E, Malhi AS, Muratoglu OK. Mechanisms of decrease in fatigue crack propagation resistance in irradiated and melted UHMWPE. Biomaterials. 2006;27(6):917-925.
36. Baker DA, Bellare A, Pruitt L. The effects of degree of crosslinking on the fatigue crack initiation and propagation resistance of orthopedic-grade polyethylene. J Biomed Mater Res A. 2003;66(1):146-154.
37. Charnley J. Total hip replacement by low-friction arthroplasty. Clin Orthop. 1970;(72):7-21.
38. Kabo JM, Gebhard JS, Loren G, Amstutz HC. In vivo wear of polyethylene acetabular components. J Bone Joint Surg Br. 1993;75(2):254-258.
39. Livermore J, Ilstrup D, Morrey B. Effect of femoral head size on wear of the polyethylene acetabular component. J Bone Joint Surg Am. 1990;72(4):518-528.
40. Ma SM, Kabo JM, Amstutz HC. Frictional torque in surface and conventional hip replacement. J Bone Joint Surg Am. 1983;65(3):366-370.
41. Muratoglu OK, Bragdon CR, O‘Connor D, et al. Larger diameter femoral heads used in conjunction with a highly cross-linked ultra-high molecular weight polyethylene: a new concept. J Arthroplasty. 2001;16(8 suppl 1):24-30.
42. Thomas GER, Simpson DJ, Mehmood S, et al. The seven-year wear of highly cross-linked polyethylene in total hip arthroplasty: a double-blind, randomized controlled trial using radiostereometric analysis. J Bone Joint Surg Am. 2011;93(8):716-722.
43. Mutimer J, Devane PA, Adams K, Horne JG. Highly crosslinked polyethylene reduces wear in total hip arthroplasty at 5 years. Clin Orthop. 2010;468(12):3228-3233.
44. Bragdon CR, Doerner M, Martell J, Jarrett B, Palm H, Malchau H. The 2012 John Charnley Award: clinical multicenter studies of the wear performance of highly crosslinked remelted polyethylene in THA. Clin Orthop. 2013;471(2):393-402.
45. Lachiewicz PF, Heckman DS, Soileau ES, Mangla J, Martell JM. Femoral head size and wear of highly cross-linked polyethylene at 5 to 8 years. Clin Orthop. 2009;467(12):3290-3296.
46. Sychterz CJ, Engh CA Jr, Young AM, Hopper RH Jr, Engh CA. Comparison of in vivo wear between polyethylene liners articulating with ceramic and cobalt-chrome femoral heads. J Bone Joint Surg Br. 2000;82(7):948-951.
47. Berry DJ, Barnes CL, Scott RD, Cabanela ME, Poss R. Catastrophic failure of the polyethylene liner of uncemented acetabular components.
J Bone Joint Surg Br. 1994;76(4):575-578.
48. McKellop H, Shen FW, Lu B, Campbell P, Salovey R. Development of an extremely wear-resistant ultra high molecular weight polyethylene for total hip replacements. J Orthop Res. 1999;17(2):157-167.
49. Wang A, Essner A, Polineni VK, Stark C, Dumbleton JH. Lubrication and wear of ultra-high molecular weight polyethylene in total joint replacements. Tribol Int. 1998;31(1-3):17-33.
50. Estok DM 2nd, Burroughs BR, Muratoglu OK, Harris WH. Comparison of hip simulator wear of 2 different highly cross-linked ultra high molecular weight polyethylene acetabular components using both 32- and 38-mm femoral heads. J Arthroplasty. 2007;22(4):581-589.
51. Muratoglu OK, Bragdon CR, O‘Connor DO, et al. Unified wear model for highly crosslinked ultra-high molecular weight polyethylenes (UHMWPE). Biomaterials. 1999;20(16):1463-1470.
52. Harris WH, Muratoglu OK. A review of current cross-linked polyethylenes used in total joint arthroplasty. Clin Orthop. 2005;(430):46-52.
53. Muratoglu OK, Bragdon CR, O‘Connor DO, Jasty M, Harris WH. A novel method of cross-linking ultra-high-molecular-weight polyethylene to improve wear, reduce oxidation, and retain mechanical properties. Recipient of the 1999 HAP Paul Award. J Arthroplasty. 2001;16(2):149-160.
54. Bal BS, Garino J, Ries M, Rahaman MN. A review of ceramic bearing materials in total joint arthroplasty. Hip Int. 2007;17(1):21-30.
55. Traina F, De Fine M, Di Martino A, Faldini C. Fracture of ceramic bearing surfaces following total hip replacement: a systematic review. Biomed Res Int. 2013;2013:157247.
56. Cai YZ, Yan SG. Development of ceramic-on-ceramic implants for total hip arthroplasty. Orthop Surg. 2010;2(3):175-181.
57. Stewart TD, Tipper JL, Insley G, Streicher RM, Ingham E, Fisher J. Long-term wear of ceramic matrix composite materials for hip prostheses under severe swing phase microseparation. J Biomed Mater Res B Appl Biomater. 2003;66(2):567-573.
58. Bragdon CR, Greene ME, Freiberg AA, Harris WH, Malchau H. Radiostereometric analysis comparison of wear of highly cross-linked polyethylene against 36- vs 28-mm femoral heads. J Arthroplasty. 2007;22(6 suppl 2):125-129.
59. Lombardi AV Jr, Skeels MD, Berend KR, Adams JB, Franchi OJ. Do large heads enhance stability and restore native anatomy in primary total hip arthroplasty? Clin Orthop. 2011;469(6):1547-1553.
60. Lachiewicz PF, Soileau ES. Low early and late dislocation rates with 36- and 40-mm heads in patients at high risk for dislocation. Clin Orthop. 2013;471(2):439-443.
61. Cai P, Hu Y, Xie J. Large-diameter Delta ceramic-on-ceramic versus common-sized ceramic-on-polyethylene bearings in THA. Orthopedics. 2012;35(9):e1307-e1313.
62. Park KS, Yoon TR, Hwang SY, Lee KB. Modified minimally invasive two-incision total hip arthroplasty using large diameter femoral head. Indian
J Orthop. 2012;46(1):29-35.
63. Garbuz DS, Masri BA, Duncan CP, et al. The Frank Stinchfield Award: dislocation in revision THA: do large heads (36 and 40 mm) result in reduced dislocation rates in a randomized clinical trial? Clin Orthop. 2012;470(2):351-356.
64. Geller JA, Malchau H, Bragdon C, Greene M, Harris WH, Freiberg AA. Large diameter femoral heads on highly cross-linked polyethylene: minimum 3-year results. Clin Orthop. 2006;(447):53-59.
65. D’Antonio JA, Capello WN, Ramakrishnan R. Second-generation annealed highly cross-linked polyethylene exhibits low wear. Clin Orthop. 2012;470(6):1696-1704.
66. Galvin AL, Jennings LM, Tipper JL, Ingham E, Fisher J. Wear and creep of highly crosslinked polyethylene against cobalt chrome and ceramic femoral heads. Proc Inst Mech Eng H. 2010;224(10):1175-1183.
67. Skeels MD, Berend KR, Lombardi AV Jr. The dislocator, early and late: the role of large heads. Orthopedics. 2009;32(9).
68. Plate JF, Seyler TM, Stroh DA, Issa K, Akbar M, Mont MA. Risk of dislocation using large- vs. small-diameter femoral heads in total hip arthroplasty. BMC Res Notes. 2012;5(1):553.
69. Bouchet R, Mercier N, Saragaglia D. Posterior approach and dislocation rate: a 213 total hip replacements case–control study comparing the dual mobility cup with a conventional 28-mm metal head/polyethylene prosthesis. Orthop Traumatol Surg Res. 2011;97(1):2-7.
70. Ali Khan MA, Brakenbury PH, Reynolds IS. Dislocation following total hip replacement. J Bone Joint Surg Br. 1981;63(2):214-218.
71. Howie DW, Holubowycz OT, Middleton R. Large femoral heads decrease the incidence of dislocation after total hip arthroplasty: a randomized controlled trial. J Bone Joint Surg Am. 2012;94(12):1095-1102.
72. Malkani AL, Ong KL, Lau E, Kurtz SM, Justice BJ, Manley MT. Early- and late-term dislocation risk after primary hip arthroplasty in the Medicare population. J Arthroplasty. 2010;25(6 suppl):21-25.
73. Berry DJ, von Knoch M, Schleck CD, Harmsen WS. Effect of femoral head diameter and operative approach on risk of dislocation after primary total hip arthroplasty. J Bone Joint Surg Am. 2005;87(11):2456-2463.
74. Cho MR, Lee HS, Lee SW, Choi CH, Kim SK, Ko SB. Results after total hip arthroplasty with a large head and bipolar arthroplasty in patients with displaced femoral neck fractures. J Arthroplasty. 2011;26(6):893-896.
75. Conroy JL, Whitehouse SL, Graves SE, Pratt NL, Ryan P, Crawford RW. Risk factors for revision for early dislocation in total hip arthroplasty.
J Arthroplasty. 2008;23(6):867-872.
76. Kung PL, Ries MD. Effect of femoral head size and abductors on dislocation after revision THA. Clin Orthop. 2007;(465):170-174.
77. Williams JT Jr, Ragland PS, Clarke S. Constrained components for the unstable hip following total hip arthroplasty: a literature review. Int Orthop. 2007;31(3):273-277.
78. Zywiel MG, Mustafa LH, Bonutti PM, Mont MA. Are abductor muscle quality and previous revision surgery predictors of constrained liner failure in hip arthroplasty? Int Orthop. 2011;35(6):797-802.
79. Guyen O, Lewallen DG, Cabanela ME. Modes of failure of Osteonics constrained tripolar implants: a retrospective analysis of forty-three failed implants. J Bone Joint Surg Am. 2008;90(7):1553-1560.
80. Banks LN, McElwain JP. An unusual mode of failure of a tripolar constrained acetabular liner: a case report. Arch Orthop Trauma Surg. 2010;130(4):503-505.
81. Robertson WJ, Mattern CJ, Hur J, Su EP, Pellicci PM. Failure mechanisms and closed reduction of a constrained tripolar acetabular liner.
J Arthroplasty. 2009;24(2):322.e5-e11.
82. Aubriot JH, Lesimple P, Leclercq S. Study of Bousquet‘s non-cemented acetabular implant in 100 hybrid total hip prostheses (Charnley type cemented femoral component). Average 5-year follow-up [in French]. Acta Orthop Belg. 1993;59(suppl 1):267-271.
83. Farizon F, de Lavison R, Azoulai JJ, Bousquet G. Results with a cementless alumina-coated cup with dual mobility. A twelve-year follow-up study. Int Orthop. 1998;22(4):219-224.
84. Mertl P, Combes A, Leiber-Wackenheim F, Fessy MH, Girard J, Migaud H. Recurrence of dislocation following total hip arthroplasty revision using dual mobility cups was rare in 180 hips followed over 7 years. HSS J. 2012;8(3):251-256.
85. Langlais FL, Ropars M, Gaucher F, Musset T, Chaix O. Dual mobility cemented cups have low dislocation rates in THA revisions. Clin Orthop. 2008;466(2):389-395.
86. Philippot R, Farizon F, Camilleri JP, et al. Survival of dual mobility socket with a mean 17 years follow-up [in French]. Rev Chir Orthop Reparatrice Appar Mot. 2008;94(1):43-48.
87. Adam P, Philippe R, Ehlinger M, et al. Dual mobility cups hip arthroplasty as a treatment for displaced fracture of the femoral neck in the elderly.
A prospective, systematic, multicenter study with specific focus on postoperative dislocation. Orthop Traumatol Surg Res. 2012;98(3):296-300.
88. Fessy MH. La double mobilité [Dual mobility]. Revue de Chirurgie Orthopédique et Traumatologique. 2010;96(7):891-898.
89. Mont MA, Issa K, Naziri Q, Harwin SF, Delanois RE, Johnson AJ. The use of dual-mobility bearings in difficult hip arthroplasty reconstructive cases. Surg Technol Int. 2011;21:234-240.
90. Sayeed SA, Mont MA, Costa CR, et al. Early outcomes of sequentially cross-linked thin polyethylene liners with large diameter femoral heads in total hip arthroplasty. Bull NYU Hosp Jt Dis. 2011;69(suppl 1):S90-S94.
91. Plate JF, Seyler TM, Stroh DA, Issa K, Akbar M, Mont MA. Risk of dislocation using large- vs. small-diameter femoral heads in total hip arthroplasty. BMC Res Notes. 2012;5(1):553.
92. Sato T, Nakashima Y, Akiyama M, et al. Wear resistant performance of highly cross-linked and annealed ultra-high molecular weight polyethylene against ceramic heads in total hip arthroplasty. J Orthop Res. 2012;30(12):2031-2037.
93. Meftah M, Ebrahimpour PB, He C, Ranawat AS, Ranawat CS. Preliminary clinical and radiographic results of large ceramic heads on highly cross-linked polyethylene. Orthopedics. 2011;34(6):133.
94. Gagala J, Mazurkiewicz T, Dajewski Z. Large diameter femoral heads in primary alumina/alumina and XSPE/alumina total hip arthroplasty.
A follow-up study of 50 hips after average 40 months and review of literature [in Polish]. Chir Narzadow Ruchu Ortop Pol. 2011;76(1):14-20.