User login
Osteoporosis Can Affect Men on Large Scale, Too
Significantly fewer men received evaluation for osteoporosis following a distal radial fracture, with rates of evaluation unacceptably low according to published guidelines, according to a study published November 5 in the Journal of Bone and Joint Surgery.
“Given that the prevalence of fragility fractures among men is expected to increase threefold by the year 2050, adequately evaluating and treating men for osteoporosis is of paramount importance,” said lead author Tamara Rozental, MD, an investigator in the Department of Orthopedic Surgery at Beth Israel Deaconess Medical Center and an Associate Professor of Orthopedic Surgery at Harvard Medical School.
Dr. Rozental, who specializes in hand, wrist, and elbow injuries, examined five years of data from 2007 to 2012, from patients who suffered a distal radial fracture.
“We know that a distal radial fracture can often be an early indication of bone loss. We typically see this type of fracture 10 to 15 years before we might see a hip fracture,” said Dr. Rozental. “When we treat fractures of the wrist, it gives us the opportunity to do a bone mass density evaluation and, if necessary, get patients into treatment with the goal of preventing more serious injury, like a hip fracture down the line.”
Even though existing clinical practice guidelines recommend bone mass density evaluation after hip fracture for both men and women, studies continue to show that screening rates are unacceptably low, particularly among men. Dr. Rozental examined the data to see if the same trend would play out when examining clinical follow up to wrist fractures.
Fifty-three percent of women received dual x-ray absorptiometry, compared with only 18% of men. In addition, 21% of men versus 55% of women initiated treatment with calcium and vitamin D supplements within six months of injury, and 3% of men versus 22% of women began taking bisphosphonates.
Studies have shown that men have twice the mortality rate of women both during initial hospitalization and in the year following a hip fracture. Survival rates following a wrist fracture also are lower among men.
“Treating men for bone fractures, but not the underlying cause, places them at a greater risk for future bone breaks and related complications,” said Dr. Rozental. “The results of this study lead us to suggest that men over the age of 50 with fractures of the distal radius should undergo further clinical assessment and bone density testing to better identify those at high risk for future fracture as well as those who would benefit from further treatment.”
Suggested Reading
Harper CM, Fitzpatrick SK, Zurakowski D, Rozental TD. Distal radial fractures in older men: a missed opportunity? J Bone Joint Surg Am. 2014;96(21):1820-1827.
Significantly fewer men received evaluation for osteoporosis following a distal radial fracture, with rates of evaluation unacceptably low according to published guidelines, according to a study published November 5 in the Journal of Bone and Joint Surgery.
“Given that the prevalence of fragility fractures among men is expected to increase threefold by the year 2050, adequately evaluating and treating men for osteoporosis is of paramount importance,” said lead author Tamara Rozental, MD, an investigator in the Department of Orthopedic Surgery at Beth Israel Deaconess Medical Center and an Associate Professor of Orthopedic Surgery at Harvard Medical School.
Dr. Rozental, who specializes in hand, wrist, and elbow injuries, examined five years of data from 2007 to 2012, from patients who suffered a distal radial fracture.
“We know that a distal radial fracture can often be an early indication of bone loss. We typically see this type of fracture 10 to 15 years before we might see a hip fracture,” said Dr. Rozental. “When we treat fractures of the wrist, it gives us the opportunity to do a bone mass density evaluation and, if necessary, get patients into treatment with the goal of preventing more serious injury, like a hip fracture down the line.”
Even though existing clinical practice guidelines recommend bone mass density evaluation after hip fracture for both men and women, studies continue to show that screening rates are unacceptably low, particularly among men. Dr. Rozental examined the data to see if the same trend would play out when examining clinical follow up to wrist fractures.
Fifty-three percent of women received dual x-ray absorptiometry, compared with only 18% of men. In addition, 21% of men versus 55% of women initiated treatment with calcium and vitamin D supplements within six months of injury, and 3% of men versus 22% of women began taking bisphosphonates.
Studies have shown that men have twice the mortality rate of women both during initial hospitalization and in the year following a hip fracture. Survival rates following a wrist fracture also are lower among men.
“Treating men for bone fractures, but not the underlying cause, places them at a greater risk for future bone breaks and related complications,” said Dr. Rozental. “The results of this study lead us to suggest that men over the age of 50 with fractures of the distal radius should undergo further clinical assessment and bone density testing to better identify those at high risk for future fracture as well as those who would benefit from further treatment.”
Significantly fewer men received evaluation for osteoporosis following a distal radial fracture, with rates of evaluation unacceptably low according to published guidelines, according to a study published November 5 in the Journal of Bone and Joint Surgery.
“Given that the prevalence of fragility fractures among men is expected to increase threefold by the year 2050, adequately evaluating and treating men for osteoporosis is of paramount importance,” said lead author Tamara Rozental, MD, an investigator in the Department of Orthopedic Surgery at Beth Israel Deaconess Medical Center and an Associate Professor of Orthopedic Surgery at Harvard Medical School.
Dr. Rozental, who specializes in hand, wrist, and elbow injuries, examined five years of data from 2007 to 2012, from patients who suffered a distal radial fracture.
“We know that a distal radial fracture can often be an early indication of bone loss. We typically see this type of fracture 10 to 15 years before we might see a hip fracture,” said Dr. Rozental. “When we treat fractures of the wrist, it gives us the opportunity to do a bone mass density evaluation and, if necessary, get patients into treatment with the goal of preventing more serious injury, like a hip fracture down the line.”
Even though existing clinical practice guidelines recommend bone mass density evaluation after hip fracture for both men and women, studies continue to show that screening rates are unacceptably low, particularly among men. Dr. Rozental examined the data to see if the same trend would play out when examining clinical follow up to wrist fractures.
Fifty-three percent of women received dual x-ray absorptiometry, compared with only 18% of men. In addition, 21% of men versus 55% of women initiated treatment with calcium and vitamin D supplements within six months of injury, and 3% of men versus 22% of women began taking bisphosphonates.
Studies have shown that men have twice the mortality rate of women both during initial hospitalization and in the year following a hip fracture. Survival rates following a wrist fracture also are lower among men.
“Treating men for bone fractures, but not the underlying cause, places them at a greater risk for future bone breaks and related complications,” said Dr. Rozental. “The results of this study lead us to suggest that men over the age of 50 with fractures of the distal radius should undergo further clinical assessment and bone density testing to better identify those at high risk for future fracture as well as those who would benefit from further treatment.”
Suggested Reading
Harper CM, Fitzpatrick SK, Zurakowski D, Rozental TD. Distal radial fractures in older men: a missed opportunity? J Bone Joint Surg Am. 2014;96(21):1820-1827.
Suggested Reading
Harper CM, Fitzpatrick SK, Zurakowski D, Rozental TD. Distal radial fractures in older men: a missed opportunity? J Bone Joint Surg Am. 2014;96(21):1820-1827.
Inflammation Causes Painful Sensitization in Knee Osteoarthritis
BOSTON—Inflammation related to synovitis or effusion may drive increased sensitization in knee osteoarthritis, according to research presented at the 2014 American College of Rheumatology Annual Meeting.
“It is widely recognized that the level of pain patients experience is not always what one would expect based upon what is seen on their x-rays,” said lead author Tuhina Neogi, MD, PhD, of Boston University School of Medicine.
Using data from the Multicenter Osteoarthritis Study (MOST), researchers looked at test results obtained from 1,111 subjects with or at risk of knee osteoarthritis, including x-rays, magnetic resonance imaging scans (MRI), and standardized somatosensory evaluations of two measures that give insights into the presence of sensitization. These measures were obtained at the knee at baseline and again two years later. The mean age of the subjects in the study was 66.9. The mean body mass index was 29.7, and 62% were female.
The researchers looked at how synovitis, effusion, and bone marrow lesions (BMLs) seen at the baseline assessment might be related to the new development of temporal summation in the same knee two years later among those who did not show signs of it at the baseline visit. They also assessed changes in pressure pain thresholds levels in the same knee between baseline and the visit two years later in all the subjects.
A total of 22.6% developed incident temporal summation by the two-year study visit. Between the baseline and two-year visit, changes in the pressure pain thresholds levels ranged from -7.35 to 7.15 kg/cm2. Synovitis was associated with significant decreases in pressure pain thresholds. Effusion was significantly associated with incident temporal summation. Bone marrow lesions presence or burden was not associated with temporal summation or change in pressure pain thresholds.
The study’s authors concluded that inflammation, such as that associated with synovitis or effusion, may drive sensitization in knee osteoarthritis, while bone marrow lesions do not appear to do so. Furthermore, researchers suggested that early targeting of inflammation in knee osteoarthritis may prevent sensitization and helping to reduce pain severity in people with knee osteoarthritis.
“This is the first such study in knee osteoarthritis to obtain sensitization measures at more than one time-point in such a large number of individuals, providing insights for the first time into how sensitization may develop or change over time in this disease,” said Dr. Neogi.
BOSTON—Inflammation related to synovitis or effusion may drive increased sensitization in knee osteoarthritis, according to research presented at the 2014 American College of Rheumatology Annual Meeting.
“It is widely recognized that the level of pain patients experience is not always what one would expect based upon what is seen on their x-rays,” said lead author Tuhina Neogi, MD, PhD, of Boston University School of Medicine.
Using data from the Multicenter Osteoarthritis Study (MOST), researchers looked at test results obtained from 1,111 subjects with or at risk of knee osteoarthritis, including x-rays, magnetic resonance imaging scans (MRI), and standardized somatosensory evaluations of two measures that give insights into the presence of sensitization. These measures were obtained at the knee at baseline and again two years later. The mean age of the subjects in the study was 66.9. The mean body mass index was 29.7, and 62% were female.
The researchers looked at how synovitis, effusion, and bone marrow lesions (BMLs) seen at the baseline assessment might be related to the new development of temporal summation in the same knee two years later among those who did not show signs of it at the baseline visit. They also assessed changes in pressure pain thresholds levels in the same knee between baseline and the visit two years later in all the subjects.
A total of 22.6% developed incident temporal summation by the two-year study visit. Between the baseline and two-year visit, changes in the pressure pain thresholds levels ranged from -7.35 to 7.15 kg/cm2. Synovitis was associated with significant decreases in pressure pain thresholds. Effusion was significantly associated with incident temporal summation. Bone marrow lesions presence or burden was not associated with temporal summation or change in pressure pain thresholds.
The study’s authors concluded that inflammation, such as that associated with synovitis or effusion, may drive sensitization in knee osteoarthritis, while bone marrow lesions do not appear to do so. Furthermore, researchers suggested that early targeting of inflammation in knee osteoarthritis may prevent sensitization and helping to reduce pain severity in people with knee osteoarthritis.
“This is the first such study in knee osteoarthritis to obtain sensitization measures at more than one time-point in such a large number of individuals, providing insights for the first time into how sensitization may develop or change over time in this disease,” said Dr. Neogi.
BOSTON—Inflammation related to synovitis or effusion may drive increased sensitization in knee osteoarthritis, according to research presented at the 2014 American College of Rheumatology Annual Meeting.
“It is widely recognized that the level of pain patients experience is not always what one would expect based upon what is seen on their x-rays,” said lead author Tuhina Neogi, MD, PhD, of Boston University School of Medicine.
Using data from the Multicenter Osteoarthritis Study (MOST), researchers looked at test results obtained from 1,111 subjects with or at risk of knee osteoarthritis, including x-rays, magnetic resonance imaging scans (MRI), and standardized somatosensory evaluations of two measures that give insights into the presence of sensitization. These measures were obtained at the knee at baseline and again two years later. The mean age of the subjects in the study was 66.9. The mean body mass index was 29.7, and 62% were female.
The researchers looked at how synovitis, effusion, and bone marrow lesions (BMLs) seen at the baseline assessment might be related to the new development of temporal summation in the same knee two years later among those who did not show signs of it at the baseline visit. They also assessed changes in pressure pain thresholds levels in the same knee between baseline and the visit two years later in all the subjects.
A total of 22.6% developed incident temporal summation by the two-year study visit. Between the baseline and two-year visit, changes in the pressure pain thresholds levels ranged from -7.35 to 7.15 kg/cm2. Synovitis was associated with significant decreases in pressure pain thresholds. Effusion was significantly associated with incident temporal summation. Bone marrow lesions presence or burden was not associated with temporal summation or change in pressure pain thresholds.
The study’s authors concluded that inflammation, such as that associated with synovitis or effusion, may drive sensitization in knee osteoarthritis, while bone marrow lesions do not appear to do so. Furthermore, researchers suggested that early targeting of inflammation in knee osteoarthritis may prevent sensitization and helping to reduce pain severity in people with knee osteoarthritis.
“This is the first such study in knee osteoarthritis to obtain sensitization measures at more than one time-point in such a large number of individuals, providing insights for the first time into how sensitization may develop or change over time in this disease,” said Dr. Neogi.
Total Hip Replacement: An Excellent Option to Relieve Pain in Young Juvenile Arthritis Patients
BOSTON—A new study finds that total hip replacement (THR) is an excellent option for patients under age 35, when traditional treatments fail to provide relief. The study, presented at the 2014 American College of Rheumatology Annual Meeting, found that hip replacement lasted at least 10 years in 85% of juvenile idiopathic arthritis (JIA) patients. Twenty years later, 50% of the patients needed a revision surgery.
“Joint replacement can free patients from a life of unrelenting pain. It can enable those in a wheel chair to walk again. Patients can go back to school or work and get their lives back,” said Mark P. Figgie, MD, senior author of the study and Chief of the Surgical Arthritis Service at the Hospital for Special Surgery in New York.
This study evaluated the longevity of implants in juvenile idiopathic arthritis patients ages 35 or younger who underwent hip replacement at Hospital for Special Surgery. “This study followed one of the largest cohorts of patients with JIA to see how they fared 10 years after total hip replacement,” said coinvestigator Ishaan Swarup, MD, an orthopedic resident at the Hospital for Special Surgery. “It is also one of the few studies to look at patient-reported measures, such as pain and the ability to perform activities of daily living.”
Data were collected retrospectively for 56 patients. Forty-one patients had undergone bilateral hip replacement, while 15 individuals had only one side replaced, for a total of 97 hip replacement surgeries. The mean time for follow-up was 12 years. The 10-year and 20-year implant survival was 85% and 50%, respectively.
The researchers found that hip replacement in patients who were 25 or older lasted longer compared to total hip replacement in younger patients. There were no other significant differences in implant longevity based on gender or the use of custom versus standard implants.
Overall, male patients reported better outcomes with respect to activities of daily living. Patients who had received custom hip implants did worse in their reporting of pain and the ability to perform daily activities.
“We were not surprised that the patients who received custom implants had lower scores, since the very fact that they needed a custom implant meant they had more severe joint deformities and more severe disease,” stated Dr. Figgie.
BOSTON—A new study finds that total hip replacement (THR) is an excellent option for patients under age 35, when traditional treatments fail to provide relief. The study, presented at the 2014 American College of Rheumatology Annual Meeting, found that hip replacement lasted at least 10 years in 85% of juvenile idiopathic arthritis (JIA) patients. Twenty years later, 50% of the patients needed a revision surgery.
“Joint replacement can free patients from a life of unrelenting pain. It can enable those in a wheel chair to walk again. Patients can go back to school or work and get their lives back,” said Mark P. Figgie, MD, senior author of the study and Chief of the Surgical Arthritis Service at the Hospital for Special Surgery in New York.
This study evaluated the longevity of implants in juvenile idiopathic arthritis patients ages 35 or younger who underwent hip replacement at Hospital for Special Surgery. “This study followed one of the largest cohorts of patients with JIA to see how they fared 10 years after total hip replacement,” said coinvestigator Ishaan Swarup, MD, an orthopedic resident at the Hospital for Special Surgery. “It is also one of the few studies to look at patient-reported measures, such as pain and the ability to perform activities of daily living.”
Data were collected retrospectively for 56 patients. Forty-one patients had undergone bilateral hip replacement, while 15 individuals had only one side replaced, for a total of 97 hip replacement surgeries. The mean time for follow-up was 12 years. The 10-year and 20-year implant survival was 85% and 50%, respectively.
The researchers found that hip replacement in patients who were 25 or older lasted longer compared to total hip replacement in younger patients. There were no other significant differences in implant longevity based on gender or the use of custom versus standard implants.
Overall, male patients reported better outcomes with respect to activities of daily living. Patients who had received custom hip implants did worse in their reporting of pain and the ability to perform daily activities.
“We were not surprised that the patients who received custom implants had lower scores, since the very fact that they needed a custom implant meant they had more severe joint deformities and more severe disease,” stated Dr. Figgie.
BOSTON—A new study finds that total hip replacement (THR) is an excellent option for patients under age 35, when traditional treatments fail to provide relief. The study, presented at the 2014 American College of Rheumatology Annual Meeting, found that hip replacement lasted at least 10 years in 85% of juvenile idiopathic arthritis (JIA) patients. Twenty years later, 50% of the patients needed a revision surgery.
“Joint replacement can free patients from a life of unrelenting pain. It can enable those in a wheel chair to walk again. Patients can go back to school or work and get their lives back,” said Mark P. Figgie, MD, senior author of the study and Chief of the Surgical Arthritis Service at the Hospital for Special Surgery in New York.
This study evaluated the longevity of implants in juvenile idiopathic arthritis patients ages 35 or younger who underwent hip replacement at Hospital for Special Surgery. “This study followed one of the largest cohorts of patients with JIA to see how they fared 10 years after total hip replacement,” said coinvestigator Ishaan Swarup, MD, an orthopedic resident at the Hospital for Special Surgery. “It is also one of the few studies to look at patient-reported measures, such as pain and the ability to perform activities of daily living.”
Data were collected retrospectively for 56 patients. Forty-one patients had undergone bilateral hip replacement, while 15 individuals had only one side replaced, for a total of 97 hip replacement surgeries. The mean time for follow-up was 12 years. The 10-year and 20-year implant survival was 85% and 50%, respectively.
The researchers found that hip replacement in patients who were 25 or older lasted longer compared to total hip replacement in younger patients. There were no other significant differences in implant longevity based on gender or the use of custom versus standard implants.
Overall, male patients reported better outcomes with respect to activities of daily living. Patients who had received custom hip implants did worse in their reporting of pain and the ability to perform daily activities.
“We were not surprised that the patients who received custom implants had lower scores, since the very fact that they needed a custom implant meant they had more severe joint deformities and more severe disease,” stated Dr. Figgie.
Manual Therapy and Exercise Improve Pain and Function in Osteoarthritis
BOSTON—Patients with hip and knee osteoarthritis (OA) may improve their pain, stiffness, and physical function with sustained physical exercise, manual therapy, or both, according to research presented at the 2014 American College of Rheumatology Annual Meeting.
“The aim of this study was to establish whether providing a comprehensive program of exercise or manual therapy results in significant additional benefits, over and above usual medical care,” said lead author J. Haxby Abbott, DPT, PhD, at the University of Otago in Dunedin, New Zealand.
The participants’ progress was measured using the Western Ontario and McMaster (WOMAC) osteoarthritis index, which calculates scores on a scale of 0 to 240. Lower WOMAC scores indicate improvements in pain, stiffness, and physical function. Participants were also given several physical performance tests, Timed Up and Go, 40-meter fast-paced walk, and a 30-second sit-to-stand. At baseline, the mean age of the osteoarthritis patients in the study was 66, with a mean WOMAC score of 100.8.
After two years, all the participants who engaged in regular exercise, manual therapy, or a combination of both showed improved WOMAC scores that were superior to those who had only the usual medical care for osteoarthritis.
Participants receiving exercise therapy in addition to their usual care showed improvement of 31.7 WOMAC points compared to usual care alone. Participants receiving manual therapy in addition to their usual care showed a relative improvement of 30.1 WOMAC points.
While the difference in WOMAC improvement for participants receiving combined exercise therapy and manual therapy in addition to usual care did not meet the a priori threshold for clinical significance (28 points), there was a trend towards benefit; with this group improving 26.2 WOMAC points more than usual care only. Those participants in the exercise therapy group showed greater mean changes on most physical performance tests than anyone in the other groups.
Adding either exercise therapy or manual therapy to usual medical care is beneficial for people with hip and knee osteoarthritis, the study’s authors concluded. “This study showed that benefits imparted by a comprehensive program of exercise therapy or manual therapy, provided by physical therapists, remain significant to at least two years follow-up,” said Dr. Abbott.
BOSTON—Patients with hip and knee osteoarthritis (OA) may improve their pain, stiffness, and physical function with sustained physical exercise, manual therapy, or both, according to research presented at the 2014 American College of Rheumatology Annual Meeting.
“The aim of this study was to establish whether providing a comprehensive program of exercise or manual therapy results in significant additional benefits, over and above usual medical care,” said lead author J. Haxby Abbott, DPT, PhD, at the University of Otago in Dunedin, New Zealand.
The participants’ progress was measured using the Western Ontario and McMaster (WOMAC) osteoarthritis index, which calculates scores on a scale of 0 to 240. Lower WOMAC scores indicate improvements in pain, stiffness, and physical function. Participants were also given several physical performance tests, Timed Up and Go, 40-meter fast-paced walk, and a 30-second sit-to-stand. At baseline, the mean age of the osteoarthritis patients in the study was 66, with a mean WOMAC score of 100.8.
After two years, all the participants who engaged in regular exercise, manual therapy, or a combination of both showed improved WOMAC scores that were superior to those who had only the usual medical care for osteoarthritis.
Participants receiving exercise therapy in addition to their usual care showed improvement of 31.7 WOMAC points compared to usual care alone. Participants receiving manual therapy in addition to their usual care showed a relative improvement of 30.1 WOMAC points.
While the difference in WOMAC improvement for participants receiving combined exercise therapy and manual therapy in addition to usual care did not meet the a priori threshold for clinical significance (28 points), there was a trend towards benefit; with this group improving 26.2 WOMAC points more than usual care only. Those participants in the exercise therapy group showed greater mean changes on most physical performance tests than anyone in the other groups.
Adding either exercise therapy or manual therapy to usual medical care is beneficial for people with hip and knee osteoarthritis, the study’s authors concluded. “This study showed that benefits imparted by a comprehensive program of exercise therapy or manual therapy, provided by physical therapists, remain significant to at least two years follow-up,” said Dr. Abbott.
BOSTON—Patients with hip and knee osteoarthritis (OA) may improve their pain, stiffness, and physical function with sustained physical exercise, manual therapy, or both, according to research presented at the 2014 American College of Rheumatology Annual Meeting.
“The aim of this study was to establish whether providing a comprehensive program of exercise or manual therapy results in significant additional benefits, over and above usual medical care,” said lead author J. Haxby Abbott, DPT, PhD, at the University of Otago in Dunedin, New Zealand.
The participants’ progress was measured using the Western Ontario and McMaster (WOMAC) osteoarthritis index, which calculates scores on a scale of 0 to 240. Lower WOMAC scores indicate improvements in pain, stiffness, and physical function. Participants were also given several physical performance tests, Timed Up and Go, 40-meter fast-paced walk, and a 30-second sit-to-stand. At baseline, the mean age of the osteoarthritis patients in the study was 66, with a mean WOMAC score of 100.8.
After two years, all the participants who engaged in regular exercise, manual therapy, or a combination of both showed improved WOMAC scores that were superior to those who had only the usual medical care for osteoarthritis.
Participants receiving exercise therapy in addition to their usual care showed improvement of 31.7 WOMAC points compared to usual care alone. Participants receiving manual therapy in addition to their usual care showed a relative improvement of 30.1 WOMAC points.
While the difference in WOMAC improvement for participants receiving combined exercise therapy and manual therapy in addition to usual care did not meet the a priori threshold for clinical significance (28 points), there was a trend towards benefit; with this group improving 26.2 WOMAC points more than usual care only. Those participants in the exercise therapy group showed greater mean changes on most physical performance tests than anyone in the other groups.
Adding either exercise therapy or manual therapy to usual medical care is beneficial for people with hip and knee osteoarthritis, the study’s authors concluded. “This study showed that benefits imparted by a comprehensive program of exercise therapy or manual therapy, provided by physical therapists, remain significant to at least two years follow-up,” said Dr. Abbott.
Biomechanical Comparison of Hamstring Tendon Fixation Devices for Anterior Cruciate Ligament Reconstruction: Part 1. Five Femoral Devices
Anterior cruciate ligament (ACL) reconstruction remains one of the most common orthopedic procedures; almost 100,000 are performed in the United States each year, and they are among the procedures more commonly performed by surgeons specializing in sports medicine and by general orthopedists.1,2 Recent years have seen a trend toward replacing the gold standard of bone–patellar tendon–bone autograft with autograft or allograft hamstring tendon in ACL reconstruction.3 This shift is being made to try to avoid the donor-site morbidity of patellar tendon autografts and decrease the incidence of postoperative anterior knee pain. With increased use of hamstring grafts in ACL reconstruction, graft fixation strength has become a priority in attempts to optimize recovery and rehabilitation.4
Rigid fixation of hamstring grafts is now recognized as a crucial factor in the long-term success of ACL reconstruction. Grafts must withstand both early rehabilitation forces as high as 500 N5 and stresses to the native ACL during healing, which may take up to 12 weeks for soft-tissue incorporation.6
The challenge has been to engineer devices that provide stable, rigid graft fixation that allows expeditious tendon-to-bone healing and increased construct stiffness. Many new fixation devices are being marketed, and there is controversy regarding which provides the best stability and strength.7 Several studies have tested various fixation devices,8-16 but so far several devices have not been compared with one another.
We conducted a study to determine if femoral hamstring fixation devices used in ACL reconstruction differ in fixation strength. We hypothesized we would find no differences.
Materials and Methods
Fifty porcine femurs were harvested after the animals had been euthanized for other studies at our institution. Our study was approved by the institutional animal care and use committee. Specimens were stored at –25°C and, on day of testing, thawed to room temperature. Gracilis and semitendinosus tendon grafts were donated by a tissue bank (LifeNet Health, Virginia Beach, Virginia). The grafts were stored at –25°C; on day of testing, tendons were thawed to room temperature.
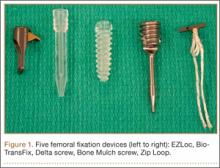
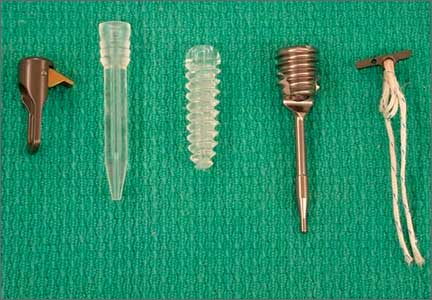
We evaluated 5 different femoral fixation devices (Figure 1): Delta screw and Bio-TransFix (Arthrex, Naples, Florida) and Bone Mulch screw, EZLoc, and Zip Loop (Arthrotek, Warsaw, Indiana). For each device, 10 ACL fixation constructs were tested.
Quadrupled human semitendinosus–gracilis tendon grafts were fixed into the femurs using the 5 femoral fixation devices. All fixations were done to manufacturer specifications.
Cyclic loading was followed by testing with the load-to-failure (LTF) protocol described by Kousa and colleagues.13 Specimens were tested in a custom load fixture (Figure 2). The base fixture used an adjustable angle vise mounted on a free rotary stage and a free x-y translation stage. This system allowed the load axis to be oriented to and aligned with the graft tunnel in the porcine femur, preventing off-axis or torsional loading of the grafts.
Pneumatic grips equipped with a custom pincer attachment allowed the graft to be grasped under a constant grip force during testing, regardless of graft thinning under tensile loads. Graft specimens were initially looped over a 3.8-mm horizontal metal shaft, and the 2 strands were double-looped at the graft insertion site. The 2 free strands were then drawn up around the metal shaft, and the shaft was placed above the serrated jaws. The metal shaft with enveloping tendon strands rested on a flat shelf at the top of the grip serrations. This configuration prevented the metal shaft and tendon strands from being pulled through the serrations when compressive force was applied to the jaws.
Before the study, the grip design was tested. There was no detectable relative motion of the strands at the grip end during graft testing to failure. The pincer attachment allowed close approach of the grips to the specimen at all femoral condyle orientations, so that a 25-mm length of exposed graft could be obtained for each specimen under initial conditions.
In the cyclic loading test, the load was applied parallel to the long axis of the femoral tunnel. A 50-N preload was initially applied to each specimen for 10 seconds, and the length of the exposed graft between grips and graft insertion was recorded. Subsequently, 1500 loading cycles between 50 N and 200 N at a rate of 1 cycle per 2 seconds (0.5 Hz) were performed. Standard force-displacement curves were then generated.
Specimens surviving the cyclic loading then underwent a single-cycle LTF test in which the load was applied parallel to the long axis of the drill hole at a rate of 50 mm per minute.
Residual displacement, stiffness, and ultimate LTF data were recorded from the force-displacement curves. Residual displacement data were generated from the cyclic loading test; residual displacement was determined by subtracting preload displacement from displacement at 1, 10, 50, 100, 250, 500, 1000, and 1500 cycles. Stiffness data were generated from the single-cycle LTF test; stiffness was defined as the linear region slope of the force-displacement curve corresponding to the steepest straight-line tangent to the loading curve. Ultimate LTF data were generated from the single-cycle LTF test; ultimate LTF was defined as the maximum load sustained by the specimen during a constant-displacement-rate tensile test for graft pullout.
Statistical analysis generated standard descriptive statistics: means, standard deviations, and proportions. One-way analysis of variance (ANOVA) was used to determine any statistically significant differences in stiffness, yield load, and residual displacement between the different fixation devices. Differences in force (load) between the single cycle and the cyclic loading test were determined by ANOVA. P < .05 was considered statistically significant for all tests.
Results
The modes of failure for the devices differed slightly (Table). Bone Mulch screw failed with a fracture through the femoral condyle extending to the bone tunnel. Zip Loop and EZLoc failed by pulling through their cortical attachment on the lateral femoral condyle. Bio-TransFix broke in the tunnel during LTF. Delta screw failed with slippage of the fixation device, and the tendons pulled out through the tunnel.
For the cyclic loading tests, only 2 of the 10 Delta screws completed the 1500-cycle loading test before failure. Of the 8 Delta screws that did not complete this testing, the majority failed after about 100 cycles. All 10 tests of Bone Mulch, Zip Loop, EZLoc, and Bio-TransFix completed the 1500-cycle loading test.
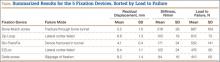
Residual displacement data were calculated from cyclic loading tests (Table). Mean (SD) residual displacement was lowest for Bio-TransFix at 4.1 (0.4) mm, followed by Bone Mulch at 5.2 (1.0) mm, EZLoc at 6.4 (1.1) mm, and Zip Loop at 6.8 (1.3) mm. Delta screws at 8.2 (1.4) mm had the highest residual displacement, though only 2 completed the cyclic tests. Bio-TransFix had significantly (P < .001) less residual displacement compared with EZLoc, Zip Loop, and Delta. Bone Mulch had significantly less residual displacement compared with Zip Loop (P < .05) and Delta (P < .01).
Stiffness data were calculated from LTF tests (Table). Mean (SD) stiffness was highest for Bone Mulch at 218 (25.9) N/mm, followed by Bio-TransFix at 171 (24.2) N/mm, EZLoc at 122 (24.1) N/mm, Zip Loop at 105 (18.9) N/mm, and Delta at 84 (16.4) N/mm. Bone Mulch had significantly (P < .001) higher stiffness compared with Bio-TransFix, EZLoc, Zip Loop, and Delta. Bio-TransFix had significantly (P < .001) higher stiffness compared with EZLoc, Zip Loop, and Delta.
Mean (SD) ultimate LTF was highest for Bone Mulch at 867 (164) N, followed by Zip Loop at 615 (72.3) N, Bio-TransFix at 552 (141) N, EZLoc at 476 (89.7) N, and Delta at 410 (65.3) N (Table). Bone Mulch failed at a statistically significantly (P < .001) higher load compared with Zip Loop, Bio-TransFix, EZLoc, and Delta. There were no significant differences in mean LTF among Zip Loop, Bio-TransFix, EZLoc, and Delta.
Discussion
In this biomechanical comparison of 5 different femoral fixation devices, the Bone Mulch screw had results superior to those of the other implants. Bone Mulch failed at higher LTF and higher stiffness. Bio-TransFix performed well and had residual displacement similar to that of Bone Mulch, but significantly lower LTF. Overall, EZLoc and Zip Loop were similar to each other in performance. The Delta (interference) screw performed poorly with respect to LTF, residual displacement, and stiffness; a large proportion of these screws failed early into cyclic loading.
Bone Mulch and Bio-TransFix overall outperformed the other fixation devices. These 2 devices are cortical-cancellous suspension devices, which provide transcondylar fixation and resist tensile forces perpendicular to the pullout force. Multiple biomechanical studies have found superior performance for these types of devices compared with various implants.10,13,15,16
Our results were similar to those of Kousa and colleagues,13 who found the Bone Mulch screw to provide highest LTF, highest stiffness, and lowest residual displacement. Another study found significantly higher stiffness for the Bone Mulch screw than for the Endobutton, a cortical suspensory fixation device.14 Bone Mulch failure modes differed, however. In the study by Kousa and colleagues,13 3 specimens failed with bending of the screw tip, and 7 failed with rupture of the tendon loop. All specimens in our study failed with fractures through the condyle. It is unclear why the failure modes differed, as we followed similar manufacturer protocols for inserting the device. It is possible the bone mass density of the porcine femurs differed between studies. This was not reported by Kousa and colleagues,13 and we did not perform testing either. However, all the porcine femurs were about the same age for testing of each device in this study.
Bio-TransFix has also been compared with various implants, but not in the same study. Brown and colleagues8 found the TransFix device significantly stiffer than the Endobutton CL. Shen and colleagues16 determined that TransFix had significantly lower residual displacement compared with Endobutton CL. Milano and colleagues15 compared multiple cortical suspensory fixation devices, including Endobutton CL, with TransFix and Bio-TransFix, and concluded the cortical-cancellous devices (TransFix, Bio-TransFix) offered the best and most predictable results in terms of elongation, fixation strength, and stiffness. TransFix has also been shown to be superior to interference screw fixation in biomechanical studies.10,15
Clinical outcomes of studies using TransFix for femoral fixation have been favorable, with improved Lysholm scores and improved laxity according to the KT-1000 test.17 However, multiple prospective studies have found no clinical difference in knee laxity between interference screw and Endobutton at 1- to 2-year follow-up18-20 and no difference in clinical outcome scores, such as the International Knee Documentation Committee score.11,18-20
Although these studies have shown no major clinical differences at short-term follow-up, the early aggressive rehabilitation period is the larger concern. Our study clearly demonstrated the biomechanical strength of transcondylar devices over other devices. The concern with transcondylar devices (vs other devices) is the increased difficulty that inexperienced surgeons have inserting them. In addition, when removed, transcondylar devices leave a large bone void.
In the present study, an important concern with femoral graft fixation is the poor performance of interference screws. Other authors recently expressed concern with using interference screws in soft-tissue ACL grafts—based on biomechanical study results of increased slippage, bone tunnel widening, and less strength.7 In the present study, Delta screws consistently performed poorest with respect to ultimate LTF, residual displacement, and stiffness. Only 20% of these screws completed 1500 cycles. Poor performance of interference screws has also been seen in other studies in tibial graft fixation21,22 and femoral graft fixation.13-15 Given their poor biomechanical properties, as seen in our study and these other studies, we think use of an interference screw alone is a poor choice for fixation.
Combined fixation techniques—interference screw plus other device(s)—may be used in clinical practice, but the present study did not evaluate any. In a biomechanical study, Yoo and colleagues23 compared an interference screw; an interference screw plus a cortical screw and a spiked washer; and a cortical screw and a spiked washer used alone in the tibia. Stiffness nearly doubled, residual displacement was less, and ultimate LTF was significantly higher in the group with the interference screw plus the cortical screw and the spiked washer. In a similar study involving femoral fixation, Oh and colleagues24 demonstrated improved stiffness, residual displacement, and LTF in cyclic testing with the combination of interference screw and Endobutton CL, compared with Endobutton CL alone. Further studies may include direct comparisons of additional femoral fixation techniques using more than 1 device.
The Zip Loop, or Toggle Loc with Zip Loop technology, is a suspensory cortical fixation device. It was initially designed for use in ACL fixation but has also been used in other surgeries, including distal biceps repair25 and ulnar collateral ligament reconstruction.26 The device itself is easy to use; more important, it allows for adjustment of graft length within the bone tunnel after deployment of the cortical fixation. Few biomechanical studies have been conducted with Zip Loop.9,12 The present study is the first to compare Zip Loop with devices other than suspensory cortical fixation devices. Zip Loop performed very well in LTF testing but had lower stiffness and higher residual displacement compared with the transcondylar fixation devices. Despite these findings, we have continued to use this device for femoral fixation in ACL reconstruction because of its ease of insertion, the ability to adjust graft tension within the bone tunnel, and the difficulties encountered inserting and removing transcondylar fixation.
We recognize the limitations in our study design with respect to how axial and cyclical loading compares with the physiologic orientation of the ACL during ambulation and running activities. This biomechanical study was not able to replicate these types of activities. However, it did provide good data supporting early rehabilitation with various fixation devices, though concern with use of interference screws remains.
Conclusion
Superior strength in fixation of hamstring grafts in the femur was demonstrated by Bone Mulch screws, followed closely by Bio-TransFix. Delta screws demonstrated poor displacement, stiffness, and LTF. When used as the sole femoral fixation device, a device with low LTF, decreased stiffness, and high residual displacement should be used cautiously in patients undergoing aggressive rehabilitation.
1. Dooley PJ, Chan DS, Dainty KN, Mohtadi NGH, Whelan DB. Patellar tendon versus hamstring autograft for anterior cruciate ligament rupture in adults. Cochrane Database Syst Rev. 2006;(2):CD005960.
2. Garrett WE Jr, Swiontkowski MF, Weinsten JN, et al. American Board of Orthopaedic Surgery Practice of the Orthopaedic Surgeon: part-II, certification examination case mix. J Bone Joint Surg Am. 2006;88(3):660-667.
3. West RV, Harner CD. Graft selection in anterior cruciate ligament reconstruction. J Am Acad Orthop Surg. 2005;13(3):197-207.
4. Hapa O, Barber FA. ACL fixation devices. Sports Med Arthrosc. 2009;17(4):217-223.
5. Walsh MP, Wijdicks CA, Parker JB, Hapa O, LaPrade RF. A comparison between a retrograde interference screw, suture button, and combined fixation on the tibial side in an all-inside anterior cruciate ligament reconstruction: a biomechanical study in a porcine model. Am J Sports Med. 2009;37(1):160-167.
6. Rodeo SA, Arnoczky SP, Torzilli PA, Hidaka C, Warren RF. Tendon-healing in a bone tunnel. A biomechanical and histological study in the dog. J Bone Joint Surg Am. 1993;75(12):1795-1803.
7. Prodromos CC, Fu FH, Howell SM, Johnson DH, Lawhorn K. Controversies in soft-tissue anterior cruciate ligament reconstruction: grafts, bundles, tunnels, fixation, and harvest. J Am Acad Orthop Surg. 2008;16(7):376-384.
8. Brown CH Jr, Wilson DR, Hecker AT, Ferragamo M. Graft-bone motion and tensile properties of hamstring and patellar tendon anterior cruciate ligament femoral graft fixation under cyclic loading. Arthroscopy. 2004;20(9):922-935.
9. Conner CS, Perez BA, Morris RP, Buckner JW, Buford WL Jr, Ivey FM. Three femoral fixation devices for anterior cruciate ligament reconstruction: comparison of fixation on the lateral cortex versus the anterior cortex. Arthroscopy. 2010;26(6):796-807.
10. Fabbriciani C, Mulas PD, Ziranu F, Deriu L, Zarelli D, Milano G. Mechanical analysis of fixation methods for anterior cruciate ligament reconstruction with hamstring tendon graft. An experimental study in sheep knees. Knee. 2005;12(2):135-138.
11. Harilainen A, Sandelin J, Jansson KA. Cross-pin femoral fixation versus metal interference screw fixation in anterior cruciate ligament reconstruction with hamstring tendons: results of a controlled prospective randomized study with 2-year follow-up. Arthroscopy. 2005;21(1):25-33.
12. Kamelger FS, Onder U, Schmoelz W, Tecklenburg K, Arora R, Fink C. Suspensory fixation of grafts in anterior cruciate ligament reconstruction: a biomechanical comparison of 3 implants. Arthroscopy. 2009;25(7):767-776.
13. Kousa P, Järvinen TL, Vihavainen M, Kannus P, Järvinen M. The fixation strength of six hamstring tendon graft fixation devices in anterior cruciate ligament reconstruction. Part I: femoral site. Am J Sports Med. 2003;31(2):174-181.
14. Kudo T, Tohyama H, Minami A, Yasuda K. The effect of cyclic loading on the biomechanical characteristics of the femur–graft–tibia complex after anterior cruciate ligament reconstruction using Bone Mulch screw/WasherLoc fixation. Clin Biomech. 2005;20(4):414-420.
15. Milano G, Mulas PD, Ziranu F, Piras S, Manunta A, Fabbriciani C. Comparison between different femoral fixation devices for ACL reconstruction with doubled hamstring tendon graft: a biomechanical analysis. Arthroscopy. 2006;22(6):660-668.
16. Shen HC, Chang JH, Lee CH, et al. Biomechanical comparison of cross-pin and Endobutton-CL femoral fixation of a flexor tendon graft for anterior cruciate ligament reconstruction—a porcine femur–graft–tibia complex study. J Surg Res. 2010;161(2):282-287.
17. Asik M, Sen C, Tuncay I, Erdil M, Avci C, Taser OF. The mid- to long-term results of the anterior cruciate ligament reconstruction with hamstring tendons using Transfix technique. Knee Surg Sports Traumatol Arthrosc. 2007;15(8):965-972.
18. Capuano L, Hardy P, Longo UG, Denaro V, Maffulli N. No difference in clinical results between femoral transfixation and bio-interference screw fixation in hamstring tendon ACL reconstruction. A preliminary study. Knee. 2008;15(3):174-179.
19. Price R, Stoney J, Brown G. Prospective randomized comparison of Endobutton versus cross-pin femoral fixation in hamstring anterior cruciate ligament reconstruction with 2-year follow-up. ANZ J Surg. 2010;80(3):162-165.
20. Rose T, Hepp P, Venus J, Stockmar C, Josten C, Lill H. Prospective randomized clinical comparison of femoral transfixation versus bioscrew fixation in hamstring tendon ACL reconstruction—a preliminary report. Knee Surg Sports Traumatol Arthrosc. 2006;14(8):730-738.
21. Kousa P, Järvinen TL, Vihavainen M, Kannus P, Järvinen M. The fixation strength of six hamstring tendon graft fixation devices in anterior cruciate ligament reconstruction. Part II: tibial site. Am J Sports Med. 2003;31(2):182-188.
22. Magen HE, Howell SM, Hull ML. Structural properties of six tibial fixation methods for anterior cruciate ligament soft tissue grafts. Am J Sports Med. 1999;27(1):35-43.
23. Yoo JC, Ahn JH, Kim JH, et al. Biomechanical testing of hybrid hamstring graft tibial fixation in anterior cruciate ligament reconstruction. Knee. 2006;13(6):455-459.
24. Oh YH, Namkoong S, Strauss EJ, et al. Hybrid femoral fixation of soft-tissue grafts in anterior cruciate ligament reconstruction using the Endobutton CL and bioabsorbable interference screws: a biomechanical study. Arthroscopy. 2006;22(11):1218-1224.
25. DiRaimo MJ Jr, Maney MD, Deitch JR. Distal biceps tendon repair using the Toggle Loc with Zip Loop. Orthopedics. 2008;31(12). doi: 10.3928/01477447-20081201-05.
26. Morgan RJ, Starman JS, Habet NA, et al. A biomechanical evaluation of ulnar collateral ligament reconstruction using a novel technique for ulnar-sided fixation. Am J Sports Med. 2010;38(7):1448-1455.
Anterior cruciate ligament (ACL) reconstruction remains one of the most common orthopedic procedures; almost 100,000 are performed in the United States each year, and they are among the procedures more commonly performed by surgeons specializing in sports medicine and by general orthopedists.1,2 Recent years have seen a trend toward replacing the gold standard of bone–patellar tendon–bone autograft with autograft or allograft hamstring tendon in ACL reconstruction.3 This shift is being made to try to avoid the donor-site morbidity of patellar tendon autografts and decrease the incidence of postoperative anterior knee pain. With increased use of hamstring grafts in ACL reconstruction, graft fixation strength has become a priority in attempts to optimize recovery and rehabilitation.4
Rigid fixation of hamstring grafts is now recognized as a crucial factor in the long-term success of ACL reconstruction. Grafts must withstand both early rehabilitation forces as high as 500 N5 and stresses to the native ACL during healing, which may take up to 12 weeks for soft-tissue incorporation.6
The challenge has been to engineer devices that provide stable, rigid graft fixation that allows expeditious tendon-to-bone healing and increased construct stiffness. Many new fixation devices are being marketed, and there is controversy regarding which provides the best stability and strength.7 Several studies have tested various fixation devices,8-16 but so far several devices have not been compared with one another.
We conducted a study to determine if femoral hamstring fixation devices used in ACL reconstruction differ in fixation strength. We hypothesized we would find no differences.
Materials and Methods
Fifty porcine femurs were harvested after the animals had been euthanized for other studies at our institution. Our study was approved by the institutional animal care and use committee. Specimens were stored at –25°C and, on day of testing, thawed to room temperature. Gracilis and semitendinosus tendon grafts were donated by a tissue bank (LifeNet Health, Virginia Beach, Virginia). The grafts were stored at –25°C; on day of testing, tendons were thawed to room temperature.
We evaluated 5 different femoral fixation devices (Figure 1): Delta screw and Bio-TransFix (Arthrex, Naples, Florida) and Bone Mulch screw, EZLoc, and Zip Loop (Arthrotek, Warsaw, Indiana). For each device, 10 ACL fixation constructs were tested.
Quadrupled human semitendinosus–gracilis tendon grafts were fixed into the femurs using the 5 femoral fixation devices. All fixations were done to manufacturer specifications.
Cyclic loading was followed by testing with the load-to-failure (LTF) protocol described by Kousa and colleagues.13 Specimens were tested in a custom load fixture (Figure 2). The base fixture used an adjustable angle vise mounted on a free rotary stage and a free x-y translation stage. This system allowed the load axis to be oriented to and aligned with the graft tunnel in the porcine femur, preventing off-axis or torsional loading of the grafts.
Pneumatic grips equipped with a custom pincer attachment allowed the graft to be grasped under a constant grip force during testing, regardless of graft thinning under tensile loads. Graft specimens were initially looped over a 3.8-mm horizontal metal shaft, and the 2 strands were double-looped at the graft insertion site. The 2 free strands were then drawn up around the metal shaft, and the shaft was placed above the serrated jaws. The metal shaft with enveloping tendon strands rested on a flat shelf at the top of the grip serrations. This configuration prevented the metal shaft and tendon strands from being pulled through the serrations when compressive force was applied to the jaws.
Before the study, the grip design was tested. There was no detectable relative motion of the strands at the grip end during graft testing to failure. The pincer attachment allowed close approach of the grips to the specimen at all femoral condyle orientations, so that a 25-mm length of exposed graft could be obtained for each specimen under initial conditions.
In the cyclic loading test, the load was applied parallel to the long axis of the femoral tunnel. A 50-N preload was initially applied to each specimen for 10 seconds, and the length of the exposed graft between grips and graft insertion was recorded. Subsequently, 1500 loading cycles between 50 N and 200 N at a rate of 1 cycle per 2 seconds (0.5 Hz) were performed. Standard force-displacement curves were then generated.
Specimens surviving the cyclic loading then underwent a single-cycle LTF test in which the load was applied parallel to the long axis of the drill hole at a rate of 50 mm per minute.
Residual displacement, stiffness, and ultimate LTF data were recorded from the force-displacement curves. Residual displacement data were generated from the cyclic loading test; residual displacement was determined by subtracting preload displacement from displacement at 1, 10, 50, 100, 250, 500, 1000, and 1500 cycles. Stiffness data were generated from the single-cycle LTF test; stiffness was defined as the linear region slope of the force-displacement curve corresponding to the steepest straight-line tangent to the loading curve. Ultimate LTF data were generated from the single-cycle LTF test; ultimate LTF was defined as the maximum load sustained by the specimen during a constant-displacement-rate tensile test for graft pullout.
Statistical analysis generated standard descriptive statistics: means, standard deviations, and proportions. One-way analysis of variance (ANOVA) was used to determine any statistically significant differences in stiffness, yield load, and residual displacement between the different fixation devices. Differences in force (load) between the single cycle and the cyclic loading test were determined by ANOVA. P < .05 was considered statistically significant for all tests.
Results
The modes of failure for the devices differed slightly (Table). Bone Mulch screw failed with a fracture through the femoral condyle extending to the bone tunnel. Zip Loop and EZLoc failed by pulling through their cortical attachment on the lateral femoral condyle. Bio-TransFix broke in the tunnel during LTF. Delta screw failed with slippage of the fixation device, and the tendons pulled out through the tunnel.
For the cyclic loading tests, only 2 of the 10 Delta screws completed the 1500-cycle loading test before failure. Of the 8 Delta screws that did not complete this testing, the majority failed after about 100 cycles. All 10 tests of Bone Mulch, Zip Loop, EZLoc, and Bio-TransFix completed the 1500-cycle loading test.
Residual displacement data were calculated from cyclic loading tests (Table). Mean (SD) residual displacement was lowest for Bio-TransFix at 4.1 (0.4) mm, followed by Bone Mulch at 5.2 (1.0) mm, EZLoc at 6.4 (1.1) mm, and Zip Loop at 6.8 (1.3) mm. Delta screws at 8.2 (1.4) mm had the highest residual displacement, though only 2 completed the cyclic tests. Bio-TransFix had significantly (P < .001) less residual displacement compared with EZLoc, Zip Loop, and Delta. Bone Mulch had significantly less residual displacement compared with Zip Loop (P < .05) and Delta (P < .01).
Stiffness data were calculated from LTF tests (Table). Mean (SD) stiffness was highest for Bone Mulch at 218 (25.9) N/mm, followed by Bio-TransFix at 171 (24.2) N/mm, EZLoc at 122 (24.1) N/mm, Zip Loop at 105 (18.9) N/mm, and Delta at 84 (16.4) N/mm. Bone Mulch had significantly (P < .001) higher stiffness compared with Bio-TransFix, EZLoc, Zip Loop, and Delta. Bio-TransFix had significantly (P < .001) higher stiffness compared with EZLoc, Zip Loop, and Delta.
Mean (SD) ultimate LTF was highest for Bone Mulch at 867 (164) N, followed by Zip Loop at 615 (72.3) N, Bio-TransFix at 552 (141) N, EZLoc at 476 (89.7) N, and Delta at 410 (65.3) N (Table). Bone Mulch failed at a statistically significantly (P < .001) higher load compared with Zip Loop, Bio-TransFix, EZLoc, and Delta. There were no significant differences in mean LTF among Zip Loop, Bio-TransFix, EZLoc, and Delta.
Discussion
In this biomechanical comparison of 5 different femoral fixation devices, the Bone Mulch screw had results superior to those of the other implants. Bone Mulch failed at higher LTF and higher stiffness. Bio-TransFix performed well and had residual displacement similar to that of Bone Mulch, but significantly lower LTF. Overall, EZLoc and Zip Loop were similar to each other in performance. The Delta (interference) screw performed poorly with respect to LTF, residual displacement, and stiffness; a large proportion of these screws failed early into cyclic loading.
Bone Mulch and Bio-TransFix overall outperformed the other fixation devices. These 2 devices are cortical-cancellous suspension devices, which provide transcondylar fixation and resist tensile forces perpendicular to the pullout force. Multiple biomechanical studies have found superior performance for these types of devices compared with various implants.10,13,15,16
Our results were similar to those of Kousa and colleagues,13 who found the Bone Mulch screw to provide highest LTF, highest stiffness, and lowest residual displacement. Another study found significantly higher stiffness for the Bone Mulch screw than for the Endobutton, a cortical suspensory fixation device.14 Bone Mulch failure modes differed, however. In the study by Kousa and colleagues,13 3 specimens failed with bending of the screw tip, and 7 failed with rupture of the tendon loop. All specimens in our study failed with fractures through the condyle. It is unclear why the failure modes differed, as we followed similar manufacturer protocols for inserting the device. It is possible the bone mass density of the porcine femurs differed between studies. This was not reported by Kousa and colleagues,13 and we did not perform testing either. However, all the porcine femurs were about the same age for testing of each device in this study.
Bio-TransFix has also been compared with various implants, but not in the same study. Brown and colleagues8 found the TransFix device significantly stiffer than the Endobutton CL. Shen and colleagues16 determined that TransFix had significantly lower residual displacement compared with Endobutton CL. Milano and colleagues15 compared multiple cortical suspensory fixation devices, including Endobutton CL, with TransFix and Bio-TransFix, and concluded the cortical-cancellous devices (TransFix, Bio-TransFix) offered the best and most predictable results in terms of elongation, fixation strength, and stiffness. TransFix has also been shown to be superior to interference screw fixation in biomechanical studies.10,15
Clinical outcomes of studies using TransFix for femoral fixation have been favorable, with improved Lysholm scores and improved laxity according to the KT-1000 test.17 However, multiple prospective studies have found no clinical difference in knee laxity between interference screw and Endobutton at 1- to 2-year follow-up18-20 and no difference in clinical outcome scores, such as the International Knee Documentation Committee score.11,18-20
Although these studies have shown no major clinical differences at short-term follow-up, the early aggressive rehabilitation period is the larger concern. Our study clearly demonstrated the biomechanical strength of transcondylar devices over other devices. The concern with transcondylar devices (vs other devices) is the increased difficulty that inexperienced surgeons have inserting them. In addition, when removed, transcondylar devices leave a large bone void.
In the present study, an important concern with femoral graft fixation is the poor performance of interference screws. Other authors recently expressed concern with using interference screws in soft-tissue ACL grafts—based on biomechanical study results of increased slippage, bone tunnel widening, and less strength.7 In the present study, Delta screws consistently performed poorest with respect to ultimate LTF, residual displacement, and stiffness. Only 20% of these screws completed 1500 cycles. Poor performance of interference screws has also been seen in other studies in tibial graft fixation21,22 and femoral graft fixation.13-15 Given their poor biomechanical properties, as seen in our study and these other studies, we think use of an interference screw alone is a poor choice for fixation.
Combined fixation techniques—interference screw plus other device(s)—may be used in clinical practice, but the present study did not evaluate any. In a biomechanical study, Yoo and colleagues23 compared an interference screw; an interference screw plus a cortical screw and a spiked washer; and a cortical screw and a spiked washer used alone in the tibia. Stiffness nearly doubled, residual displacement was less, and ultimate LTF was significantly higher in the group with the interference screw plus the cortical screw and the spiked washer. In a similar study involving femoral fixation, Oh and colleagues24 demonstrated improved stiffness, residual displacement, and LTF in cyclic testing with the combination of interference screw and Endobutton CL, compared with Endobutton CL alone. Further studies may include direct comparisons of additional femoral fixation techniques using more than 1 device.
The Zip Loop, or Toggle Loc with Zip Loop technology, is a suspensory cortical fixation device. It was initially designed for use in ACL fixation but has also been used in other surgeries, including distal biceps repair25 and ulnar collateral ligament reconstruction.26 The device itself is easy to use; more important, it allows for adjustment of graft length within the bone tunnel after deployment of the cortical fixation. Few biomechanical studies have been conducted with Zip Loop.9,12 The present study is the first to compare Zip Loop with devices other than suspensory cortical fixation devices. Zip Loop performed very well in LTF testing but had lower stiffness and higher residual displacement compared with the transcondylar fixation devices. Despite these findings, we have continued to use this device for femoral fixation in ACL reconstruction because of its ease of insertion, the ability to adjust graft tension within the bone tunnel, and the difficulties encountered inserting and removing transcondylar fixation.
We recognize the limitations in our study design with respect to how axial and cyclical loading compares with the physiologic orientation of the ACL during ambulation and running activities. This biomechanical study was not able to replicate these types of activities. However, it did provide good data supporting early rehabilitation with various fixation devices, though concern with use of interference screws remains.
Conclusion
Superior strength in fixation of hamstring grafts in the femur was demonstrated by Bone Mulch screws, followed closely by Bio-TransFix. Delta screws demonstrated poor displacement, stiffness, and LTF. When used as the sole femoral fixation device, a device with low LTF, decreased stiffness, and high residual displacement should be used cautiously in patients undergoing aggressive rehabilitation.
Anterior cruciate ligament (ACL) reconstruction remains one of the most common orthopedic procedures; almost 100,000 are performed in the United States each year, and they are among the procedures more commonly performed by surgeons specializing in sports medicine and by general orthopedists.1,2 Recent years have seen a trend toward replacing the gold standard of bone–patellar tendon–bone autograft with autograft or allograft hamstring tendon in ACL reconstruction.3 This shift is being made to try to avoid the donor-site morbidity of patellar tendon autografts and decrease the incidence of postoperative anterior knee pain. With increased use of hamstring grafts in ACL reconstruction, graft fixation strength has become a priority in attempts to optimize recovery and rehabilitation.4
Rigid fixation of hamstring grafts is now recognized as a crucial factor in the long-term success of ACL reconstruction. Grafts must withstand both early rehabilitation forces as high as 500 N5 and stresses to the native ACL during healing, which may take up to 12 weeks for soft-tissue incorporation.6
The challenge has been to engineer devices that provide stable, rigid graft fixation that allows expeditious tendon-to-bone healing and increased construct stiffness. Many new fixation devices are being marketed, and there is controversy regarding which provides the best stability and strength.7 Several studies have tested various fixation devices,8-16 but so far several devices have not been compared with one another.
We conducted a study to determine if femoral hamstring fixation devices used in ACL reconstruction differ in fixation strength. We hypothesized we would find no differences.
Materials and Methods
Fifty porcine femurs were harvested after the animals had been euthanized for other studies at our institution. Our study was approved by the institutional animal care and use committee. Specimens were stored at –25°C and, on day of testing, thawed to room temperature. Gracilis and semitendinosus tendon grafts were donated by a tissue bank (LifeNet Health, Virginia Beach, Virginia). The grafts were stored at –25°C; on day of testing, tendons were thawed to room temperature.
We evaluated 5 different femoral fixation devices (Figure 1): Delta screw and Bio-TransFix (Arthrex, Naples, Florida) and Bone Mulch screw, EZLoc, and Zip Loop (Arthrotek, Warsaw, Indiana). For each device, 10 ACL fixation constructs were tested.
Quadrupled human semitendinosus–gracilis tendon grafts were fixed into the femurs using the 5 femoral fixation devices. All fixations were done to manufacturer specifications.
Cyclic loading was followed by testing with the load-to-failure (LTF) protocol described by Kousa and colleagues.13 Specimens were tested in a custom load fixture (Figure 2). The base fixture used an adjustable angle vise mounted on a free rotary stage and a free x-y translation stage. This system allowed the load axis to be oriented to and aligned with the graft tunnel in the porcine femur, preventing off-axis or torsional loading of the grafts.
Pneumatic grips equipped with a custom pincer attachment allowed the graft to be grasped under a constant grip force during testing, regardless of graft thinning under tensile loads. Graft specimens were initially looped over a 3.8-mm horizontal metal shaft, and the 2 strands were double-looped at the graft insertion site. The 2 free strands were then drawn up around the metal shaft, and the shaft was placed above the serrated jaws. The metal shaft with enveloping tendon strands rested on a flat shelf at the top of the grip serrations. This configuration prevented the metal shaft and tendon strands from being pulled through the serrations when compressive force was applied to the jaws.
Before the study, the grip design was tested. There was no detectable relative motion of the strands at the grip end during graft testing to failure. The pincer attachment allowed close approach of the grips to the specimen at all femoral condyle orientations, so that a 25-mm length of exposed graft could be obtained for each specimen under initial conditions.
In the cyclic loading test, the load was applied parallel to the long axis of the femoral tunnel. A 50-N preload was initially applied to each specimen for 10 seconds, and the length of the exposed graft between grips and graft insertion was recorded. Subsequently, 1500 loading cycles between 50 N and 200 N at a rate of 1 cycle per 2 seconds (0.5 Hz) were performed. Standard force-displacement curves were then generated.
Specimens surviving the cyclic loading then underwent a single-cycle LTF test in which the load was applied parallel to the long axis of the drill hole at a rate of 50 mm per minute.
Residual displacement, stiffness, and ultimate LTF data were recorded from the force-displacement curves. Residual displacement data were generated from the cyclic loading test; residual displacement was determined by subtracting preload displacement from displacement at 1, 10, 50, 100, 250, 500, 1000, and 1500 cycles. Stiffness data were generated from the single-cycle LTF test; stiffness was defined as the linear region slope of the force-displacement curve corresponding to the steepest straight-line tangent to the loading curve. Ultimate LTF data were generated from the single-cycle LTF test; ultimate LTF was defined as the maximum load sustained by the specimen during a constant-displacement-rate tensile test for graft pullout.
Statistical analysis generated standard descriptive statistics: means, standard deviations, and proportions. One-way analysis of variance (ANOVA) was used to determine any statistically significant differences in stiffness, yield load, and residual displacement between the different fixation devices. Differences in force (load) between the single cycle and the cyclic loading test were determined by ANOVA. P < .05 was considered statistically significant for all tests.
Results
The modes of failure for the devices differed slightly (Table). Bone Mulch screw failed with a fracture through the femoral condyle extending to the bone tunnel. Zip Loop and EZLoc failed by pulling through their cortical attachment on the lateral femoral condyle. Bio-TransFix broke in the tunnel during LTF. Delta screw failed with slippage of the fixation device, and the tendons pulled out through the tunnel.
For the cyclic loading tests, only 2 of the 10 Delta screws completed the 1500-cycle loading test before failure. Of the 8 Delta screws that did not complete this testing, the majority failed after about 100 cycles. All 10 tests of Bone Mulch, Zip Loop, EZLoc, and Bio-TransFix completed the 1500-cycle loading test.
Residual displacement data were calculated from cyclic loading tests (Table). Mean (SD) residual displacement was lowest for Bio-TransFix at 4.1 (0.4) mm, followed by Bone Mulch at 5.2 (1.0) mm, EZLoc at 6.4 (1.1) mm, and Zip Loop at 6.8 (1.3) mm. Delta screws at 8.2 (1.4) mm had the highest residual displacement, though only 2 completed the cyclic tests. Bio-TransFix had significantly (P < .001) less residual displacement compared with EZLoc, Zip Loop, and Delta. Bone Mulch had significantly less residual displacement compared with Zip Loop (P < .05) and Delta (P < .01).
Stiffness data were calculated from LTF tests (Table). Mean (SD) stiffness was highest for Bone Mulch at 218 (25.9) N/mm, followed by Bio-TransFix at 171 (24.2) N/mm, EZLoc at 122 (24.1) N/mm, Zip Loop at 105 (18.9) N/mm, and Delta at 84 (16.4) N/mm. Bone Mulch had significantly (P < .001) higher stiffness compared with Bio-TransFix, EZLoc, Zip Loop, and Delta. Bio-TransFix had significantly (P < .001) higher stiffness compared with EZLoc, Zip Loop, and Delta.
Mean (SD) ultimate LTF was highest for Bone Mulch at 867 (164) N, followed by Zip Loop at 615 (72.3) N, Bio-TransFix at 552 (141) N, EZLoc at 476 (89.7) N, and Delta at 410 (65.3) N (Table). Bone Mulch failed at a statistically significantly (P < .001) higher load compared with Zip Loop, Bio-TransFix, EZLoc, and Delta. There were no significant differences in mean LTF among Zip Loop, Bio-TransFix, EZLoc, and Delta.
Discussion
In this biomechanical comparison of 5 different femoral fixation devices, the Bone Mulch screw had results superior to those of the other implants. Bone Mulch failed at higher LTF and higher stiffness. Bio-TransFix performed well and had residual displacement similar to that of Bone Mulch, but significantly lower LTF. Overall, EZLoc and Zip Loop were similar to each other in performance. The Delta (interference) screw performed poorly with respect to LTF, residual displacement, and stiffness; a large proportion of these screws failed early into cyclic loading.
Bone Mulch and Bio-TransFix overall outperformed the other fixation devices. These 2 devices are cortical-cancellous suspension devices, which provide transcondylar fixation and resist tensile forces perpendicular to the pullout force. Multiple biomechanical studies have found superior performance for these types of devices compared with various implants.10,13,15,16
Our results were similar to those of Kousa and colleagues,13 who found the Bone Mulch screw to provide highest LTF, highest stiffness, and lowest residual displacement. Another study found significantly higher stiffness for the Bone Mulch screw than for the Endobutton, a cortical suspensory fixation device.14 Bone Mulch failure modes differed, however. In the study by Kousa and colleagues,13 3 specimens failed with bending of the screw tip, and 7 failed with rupture of the tendon loop. All specimens in our study failed with fractures through the condyle. It is unclear why the failure modes differed, as we followed similar manufacturer protocols for inserting the device. It is possible the bone mass density of the porcine femurs differed between studies. This was not reported by Kousa and colleagues,13 and we did not perform testing either. However, all the porcine femurs were about the same age for testing of each device in this study.
Bio-TransFix has also been compared with various implants, but not in the same study. Brown and colleagues8 found the TransFix device significantly stiffer than the Endobutton CL. Shen and colleagues16 determined that TransFix had significantly lower residual displacement compared with Endobutton CL. Milano and colleagues15 compared multiple cortical suspensory fixation devices, including Endobutton CL, with TransFix and Bio-TransFix, and concluded the cortical-cancellous devices (TransFix, Bio-TransFix) offered the best and most predictable results in terms of elongation, fixation strength, and stiffness. TransFix has also been shown to be superior to interference screw fixation in biomechanical studies.10,15
Clinical outcomes of studies using TransFix for femoral fixation have been favorable, with improved Lysholm scores and improved laxity according to the KT-1000 test.17 However, multiple prospective studies have found no clinical difference in knee laxity between interference screw and Endobutton at 1- to 2-year follow-up18-20 and no difference in clinical outcome scores, such as the International Knee Documentation Committee score.11,18-20
Although these studies have shown no major clinical differences at short-term follow-up, the early aggressive rehabilitation period is the larger concern. Our study clearly demonstrated the biomechanical strength of transcondylar devices over other devices. The concern with transcondylar devices (vs other devices) is the increased difficulty that inexperienced surgeons have inserting them. In addition, when removed, transcondylar devices leave a large bone void.
In the present study, an important concern with femoral graft fixation is the poor performance of interference screws. Other authors recently expressed concern with using interference screws in soft-tissue ACL grafts—based on biomechanical study results of increased slippage, bone tunnel widening, and less strength.7 In the present study, Delta screws consistently performed poorest with respect to ultimate LTF, residual displacement, and stiffness. Only 20% of these screws completed 1500 cycles. Poor performance of interference screws has also been seen in other studies in tibial graft fixation21,22 and femoral graft fixation.13-15 Given their poor biomechanical properties, as seen in our study and these other studies, we think use of an interference screw alone is a poor choice for fixation.
Combined fixation techniques—interference screw plus other device(s)—may be used in clinical practice, but the present study did not evaluate any. In a biomechanical study, Yoo and colleagues23 compared an interference screw; an interference screw plus a cortical screw and a spiked washer; and a cortical screw and a spiked washer used alone in the tibia. Stiffness nearly doubled, residual displacement was less, and ultimate LTF was significantly higher in the group with the interference screw plus the cortical screw and the spiked washer. In a similar study involving femoral fixation, Oh and colleagues24 demonstrated improved stiffness, residual displacement, and LTF in cyclic testing with the combination of interference screw and Endobutton CL, compared with Endobutton CL alone. Further studies may include direct comparisons of additional femoral fixation techniques using more than 1 device.
The Zip Loop, or Toggle Loc with Zip Loop technology, is a suspensory cortical fixation device. It was initially designed for use in ACL fixation but has also been used in other surgeries, including distal biceps repair25 and ulnar collateral ligament reconstruction.26 The device itself is easy to use; more important, it allows for adjustment of graft length within the bone tunnel after deployment of the cortical fixation. Few biomechanical studies have been conducted with Zip Loop.9,12 The present study is the first to compare Zip Loop with devices other than suspensory cortical fixation devices. Zip Loop performed very well in LTF testing but had lower stiffness and higher residual displacement compared with the transcondylar fixation devices. Despite these findings, we have continued to use this device for femoral fixation in ACL reconstruction because of its ease of insertion, the ability to adjust graft tension within the bone tunnel, and the difficulties encountered inserting and removing transcondylar fixation.
We recognize the limitations in our study design with respect to how axial and cyclical loading compares with the physiologic orientation of the ACL during ambulation and running activities. This biomechanical study was not able to replicate these types of activities. However, it did provide good data supporting early rehabilitation with various fixation devices, though concern with use of interference screws remains.
Conclusion
Superior strength in fixation of hamstring grafts in the femur was demonstrated by Bone Mulch screws, followed closely by Bio-TransFix. Delta screws demonstrated poor displacement, stiffness, and LTF. When used as the sole femoral fixation device, a device with low LTF, decreased stiffness, and high residual displacement should be used cautiously in patients undergoing aggressive rehabilitation.
1. Dooley PJ, Chan DS, Dainty KN, Mohtadi NGH, Whelan DB. Patellar tendon versus hamstring autograft for anterior cruciate ligament rupture in adults. Cochrane Database Syst Rev. 2006;(2):CD005960.
2. Garrett WE Jr, Swiontkowski MF, Weinsten JN, et al. American Board of Orthopaedic Surgery Practice of the Orthopaedic Surgeon: part-II, certification examination case mix. J Bone Joint Surg Am. 2006;88(3):660-667.
3. West RV, Harner CD. Graft selection in anterior cruciate ligament reconstruction. J Am Acad Orthop Surg. 2005;13(3):197-207.
4. Hapa O, Barber FA. ACL fixation devices. Sports Med Arthrosc. 2009;17(4):217-223.
5. Walsh MP, Wijdicks CA, Parker JB, Hapa O, LaPrade RF. A comparison between a retrograde interference screw, suture button, and combined fixation on the tibial side in an all-inside anterior cruciate ligament reconstruction: a biomechanical study in a porcine model. Am J Sports Med. 2009;37(1):160-167.
6. Rodeo SA, Arnoczky SP, Torzilli PA, Hidaka C, Warren RF. Tendon-healing in a bone tunnel. A biomechanical and histological study in the dog. J Bone Joint Surg Am. 1993;75(12):1795-1803.
7. Prodromos CC, Fu FH, Howell SM, Johnson DH, Lawhorn K. Controversies in soft-tissue anterior cruciate ligament reconstruction: grafts, bundles, tunnels, fixation, and harvest. J Am Acad Orthop Surg. 2008;16(7):376-384.
8. Brown CH Jr, Wilson DR, Hecker AT, Ferragamo M. Graft-bone motion and tensile properties of hamstring and patellar tendon anterior cruciate ligament femoral graft fixation under cyclic loading. Arthroscopy. 2004;20(9):922-935.
9. Conner CS, Perez BA, Morris RP, Buckner JW, Buford WL Jr, Ivey FM. Three femoral fixation devices for anterior cruciate ligament reconstruction: comparison of fixation on the lateral cortex versus the anterior cortex. Arthroscopy. 2010;26(6):796-807.
10. Fabbriciani C, Mulas PD, Ziranu F, Deriu L, Zarelli D, Milano G. Mechanical analysis of fixation methods for anterior cruciate ligament reconstruction with hamstring tendon graft. An experimental study in sheep knees. Knee. 2005;12(2):135-138.
11. Harilainen A, Sandelin J, Jansson KA. Cross-pin femoral fixation versus metal interference screw fixation in anterior cruciate ligament reconstruction with hamstring tendons: results of a controlled prospective randomized study with 2-year follow-up. Arthroscopy. 2005;21(1):25-33.
12. Kamelger FS, Onder U, Schmoelz W, Tecklenburg K, Arora R, Fink C. Suspensory fixation of grafts in anterior cruciate ligament reconstruction: a biomechanical comparison of 3 implants. Arthroscopy. 2009;25(7):767-776.
13. Kousa P, Järvinen TL, Vihavainen M, Kannus P, Järvinen M. The fixation strength of six hamstring tendon graft fixation devices in anterior cruciate ligament reconstruction. Part I: femoral site. Am J Sports Med. 2003;31(2):174-181.
14. Kudo T, Tohyama H, Minami A, Yasuda K. The effect of cyclic loading on the biomechanical characteristics of the femur–graft–tibia complex after anterior cruciate ligament reconstruction using Bone Mulch screw/WasherLoc fixation. Clin Biomech. 2005;20(4):414-420.
15. Milano G, Mulas PD, Ziranu F, Piras S, Manunta A, Fabbriciani C. Comparison between different femoral fixation devices for ACL reconstruction with doubled hamstring tendon graft: a biomechanical analysis. Arthroscopy. 2006;22(6):660-668.
16. Shen HC, Chang JH, Lee CH, et al. Biomechanical comparison of cross-pin and Endobutton-CL femoral fixation of a flexor tendon graft for anterior cruciate ligament reconstruction—a porcine femur–graft–tibia complex study. J Surg Res. 2010;161(2):282-287.
17. Asik M, Sen C, Tuncay I, Erdil M, Avci C, Taser OF. The mid- to long-term results of the anterior cruciate ligament reconstruction with hamstring tendons using Transfix technique. Knee Surg Sports Traumatol Arthrosc. 2007;15(8):965-972.
18. Capuano L, Hardy P, Longo UG, Denaro V, Maffulli N. No difference in clinical results between femoral transfixation and bio-interference screw fixation in hamstring tendon ACL reconstruction. A preliminary study. Knee. 2008;15(3):174-179.
19. Price R, Stoney J, Brown G. Prospective randomized comparison of Endobutton versus cross-pin femoral fixation in hamstring anterior cruciate ligament reconstruction with 2-year follow-up. ANZ J Surg. 2010;80(3):162-165.
20. Rose T, Hepp P, Venus J, Stockmar C, Josten C, Lill H. Prospective randomized clinical comparison of femoral transfixation versus bioscrew fixation in hamstring tendon ACL reconstruction—a preliminary report. Knee Surg Sports Traumatol Arthrosc. 2006;14(8):730-738.
21. Kousa P, Järvinen TL, Vihavainen M, Kannus P, Järvinen M. The fixation strength of six hamstring tendon graft fixation devices in anterior cruciate ligament reconstruction. Part II: tibial site. Am J Sports Med. 2003;31(2):182-188.
22. Magen HE, Howell SM, Hull ML. Structural properties of six tibial fixation methods for anterior cruciate ligament soft tissue grafts. Am J Sports Med. 1999;27(1):35-43.
23. Yoo JC, Ahn JH, Kim JH, et al. Biomechanical testing of hybrid hamstring graft tibial fixation in anterior cruciate ligament reconstruction. Knee. 2006;13(6):455-459.
24. Oh YH, Namkoong S, Strauss EJ, et al. Hybrid femoral fixation of soft-tissue grafts in anterior cruciate ligament reconstruction using the Endobutton CL and bioabsorbable interference screws: a biomechanical study. Arthroscopy. 2006;22(11):1218-1224.
25. DiRaimo MJ Jr, Maney MD, Deitch JR. Distal biceps tendon repair using the Toggle Loc with Zip Loop. Orthopedics. 2008;31(12). doi: 10.3928/01477447-20081201-05.
26. Morgan RJ, Starman JS, Habet NA, et al. A biomechanical evaluation of ulnar collateral ligament reconstruction using a novel technique for ulnar-sided fixation. Am J Sports Med. 2010;38(7):1448-1455.
1. Dooley PJ, Chan DS, Dainty KN, Mohtadi NGH, Whelan DB. Patellar tendon versus hamstring autograft for anterior cruciate ligament rupture in adults. Cochrane Database Syst Rev. 2006;(2):CD005960.
2. Garrett WE Jr, Swiontkowski MF, Weinsten JN, et al. American Board of Orthopaedic Surgery Practice of the Orthopaedic Surgeon: part-II, certification examination case mix. J Bone Joint Surg Am. 2006;88(3):660-667.
3. West RV, Harner CD. Graft selection in anterior cruciate ligament reconstruction. J Am Acad Orthop Surg. 2005;13(3):197-207.
4. Hapa O, Barber FA. ACL fixation devices. Sports Med Arthrosc. 2009;17(4):217-223.
5. Walsh MP, Wijdicks CA, Parker JB, Hapa O, LaPrade RF. A comparison between a retrograde interference screw, suture button, and combined fixation on the tibial side in an all-inside anterior cruciate ligament reconstruction: a biomechanical study in a porcine model. Am J Sports Med. 2009;37(1):160-167.
6. Rodeo SA, Arnoczky SP, Torzilli PA, Hidaka C, Warren RF. Tendon-healing in a bone tunnel. A biomechanical and histological study in the dog. J Bone Joint Surg Am. 1993;75(12):1795-1803.
7. Prodromos CC, Fu FH, Howell SM, Johnson DH, Lawhorn K. Controversies in soft-tissue anterior cruciate ligament reconstruction: grafts, bundles, tunnels, fixation, and harvest. J Am Acad Orthop Surg. 2008;16(7):376-384.
8. Brown CH Jr, Wilson DR, Hecker AT, Ferragamo M. Graft-bone motion and tensile properties of hamstring and patellar tendon anterior cruciate ligament femoral graft fixation under cyclic loading. Arthroscopy. 2004;20(9):922-935.
9. Conner CS, Perez BA, Morris RP, Buckner JW, Buford WL Jr, Ivey FM. Three femoral fixation devices for anterior cruciate ligament reconstruction: comparison of fixation on the lateral cortex versus the anterior cortex. Arthroscopy. 2010;26(6):796-807.
10. Fabbriciani C, Mulas PD, Ziranu F, Deriu L, Zarelli D, Milano G. Mechanical analysis of fixation methods for anterior cruciate ligament reconstruction with hamstring tendon graft. An experimental study in sheep knees. Knee. 2005;12(2):135-138.
11. Harilainen A, Sandelin J, Jansson KA. Cross-pin femoral fixation versus metal interference screw fixation in anterior cruciate ligament reconstruction with hamstring tendons: results of a controlled prospective randomized study with 2-year follow-up. Arthroscopy. 2005;21(1):25-33.
12. Kamelger FS, Onder U, Schmoelz W, Tecklenburg K, Arora R, Fink C. Suspensory fixation of grafts in anterior cruciate ligament reconstruction: a biomechanical comparison of 3 implants. Arthroscopy. 2009;25(7):767-776.
13. Kousa P, Järvinen TL, Vihavainen M, Kannus P, Järvinen M. The fixation strength of six hamstring tendon graft fixation devices in anterior cruciate ligament reconstruction. Part I: femoral site. Am J Sports Med. 2003;31(2):174-181.
14. Kudo T, Tohyama H, Minami A, Yasuda K. The effect of cyclic loading on the biomechanical characteristics of the femur–graft–tibia complex after anterior cruciate ligament reconstruction using Bone Mulch screw/WasherLoc fixation. Clin Biomech. 2005;20(4):414-420.
15. Milano G, Mulas PD, Ziranu F, Piras S, Manunta A, Fabbriciani C. Comparison between different femoral fixation devices for ACL reconstruction with doubled hamstring tendon graft: a biomechanical analysis. Arthroscopy. 2006;22(6):660-668.
16. Shen HC, Chang JH, Lee CH, et al. Biomechanical comparison of cross-pin and Endobutton-CL femoral fixation of a flexor tendon graft for anterior cruciate ligament reconstruction—a porcine femur–graft–tibia complex study. J Surg Res. 2010;161(2):282-287.
17. Asik M, Sen C, Tuncay I, Erdil M, Avci C, Taser OF. The mid- to long-term results of the anterior cruciate ligament reconstruction with hamstring tendons using Transfix technique. Knee Surg Sports Traumatol Arthrosc. 2007;15(8):965-972.
18. Capuano L, Hardy P, Longo UG, Denaro V, Maffulli N. No difference in clinical results between femoral transfixation and bio-interference screw fixation in hamstring tendon ACL reconstruction. A preliminary study. Knee. 2008;15(3):174-179.
19. Price R, Stoney J, Brown G. Prospective randomized comparison of Endobutton versus cross-pin femoral fixation in hamstring anterior cruciate ligament reconstruction with 2-year follow-up. ANZ J Surg. 2010;80(3):162-165.
20. Rose T, Hepp P, Venus J, Stockmar C, Josten C, Lill H. Prospective randomized clinical comparison of femoral transfixation versus bioscrew fixation in hamstring tendon ACL reconstruction—a preliminary report. Knee Surg Sports Traumatol Arthrosc. 2006;14(8):730-738.
21. Kousa P, Järvinen TL, Vihavainen M, Kannus P, Järvinen M. The fixation strength of six hamstring tendon graft fixation devices in anterior cruciate ligament reconstruction. Part II: tibial site. Am J Sports Med. 2003;31(2):182-188.
22. Magen HE, Howell SM, Hull ML. Structural properties of six tibial fixation methods for anterior cruciate ligament soft tissue grafts. Am J Sports Med. 1999;27(1):35-43.
23. Yoo JC, Ahn JH, Kim JH, et al. Biomechanical testing of hybrid hamstring graft tibial fixation in anterior cruciate ligament reconstruction. Knee. 2006;13(6):455-459.
24. Oh YH, Namkoong S, Strauss EJ, et al. Hybrid femoral fixation of soft-tissue grafts in anterior cruciate ligament reconstruction using the Endobutton CL and bioabsorbable interference screws: a biomechanical study. Arthroscopy. 2006;22(11):1218-1224.
25. DiRaimo MJ Jr, Maney MD, Deitch JR. Distal biceps tendon repair using the Toggle Loc with Zip Loop. Orthopedics. 2008;31(12). doi: 10.3928/01477447-20081201-05.
26. Morgan RJ, Starman JS, Habet NA, et al. A biomechanical evaluation of ulnar collateral ligament reconstruction using a novel technique for ulnar-sided fixation. Am J Sports Med. 2010;38(7):1448-1455.
Anterior Hip Capsuloligamentous Reconstruction for Recurrent Instability After Hip Arthroscopy
Hip arthroscopy has experienced a dramatic increase in popularity, largely resulting from improvements in techniques and technology.1,2 As with any procedure, there are complications associated with arthroscopy of the hip. These include neurapraxia, iatrogenic cartilage and labral injuries, postoperative bleeding, perineal skin necrosis, infection, intra-articular instrument breakage, intra-abdominal fluid extravasation, avascular necrosis, and femoral neck fracture.1-4 Many of these have been attributed to the expected learning curve seen with any new procedure, and are less likely to occur as surgeons become more familiar with the procedure.1 One rare but serious complication is anterior dislocation of the hip.5-7
We present a patient who experienced an anterior hip dislocation and instability after hip arthroscopy, and was successfully treated with an anterior capsuloligamentous reconstruction. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
An otherwise healthy 37-year-old woman presented to our clinic with a 6-month history of right groin pain and an occasional popping sensation during activity, which was unresponsive to hip-specific physical therapy. On physical examination, she was 5 ft 10 in tall, weighed 150 lbs, and appeared in excellent physical condition. She had no signs of systemic ligamentous laxity. She had an otherwise normal musculoskeletal, neurologic, and vascular examination in her bilateral lower extremities. She had a mild antalgic gait on the right leg.
The affected right hip could be flexed painfully to 120º, extended to 0º, adducted 20º, and abducted 45º. At 90º of flexion, her right hip could be externally rotated 30º and internally rotated 20º. Internal rotation during hip flexion beyond 90º caused sharp pain in the groin. Her normal left hip could be flexed to 120º, extended to 0º, adducted 30º, and abducted 60º. At 90º of flexion, her left hip could be externally rotated 50º and internally rotated 30º. She had negative Ober tests bilaterally but had tenderness along the right iliotibial band. She had negative Patrick and Gaenslen tests bilaterally. She had no tenderness in the area of either greater trochanter.
Imaging evaluation included plain radiographs and a magnetic resonance arthrogram (MRA) of the right hip. The plain radiographs showed signs of femoroacetabular impingement, but no joint space narrowing, no dysplasia, and no retroversion of the acetabulum (Figures 1A, 1B). The MRA showed a degenerative peripheral tear of the anterosuperior labrum without significant cartilage wear (Figure 2).
Based upon her findings on physical examination and imaging, we recommended arthroscopic treatment of her right hip pathology. Thirteen months after initial presentation, we performed a right hip arthroscopy with the patient in the supine position. Through modified anterior and anterolateral portals, we used electrocautery to perform a capsulotomy from the 9 o’clock to 12 o’clock positions. A central compartment diagnostic arthroscopy showed mild degenerative fraying of the labrum from the 9 o’clock to 12 o’clock positions without signs of detachment. There was grade III chondral fraying near the articular margin in that same arc. The femoral articular cartilage appeared normal, as did the ligamentum teres. We used a shaver to gently débride the torn labrum down to stable tissue. The frayed cartilage on the acetabulum was also gently débrided.
Traction was released and the hip was flexed. Minimal capsular release and débridement were performed for adequate visualization of the peripheral compartment. A diagnostic examination revealed a significant cam-type impingement lesion from the 12 o’clock to 6 o’clock positions. We performed a femoral neck resection, with a proximal-distal dimension of 15 mm and a depth of 7 mm. A dynamic fluoroscopic examination of the hip joint showed no signs of impingement. In accordance with our standard protocol, the anterior capsulotomy was not repaired.
Postoperatively, the patient was instructed to perform toe-touch weight-bearing with crutches for 2 weeks and to advance to full weight-bearing over the next 2 weeks. She did not use a hip orthosis. She was also advised to avoid combined hip extension/external rotation maneuvers for the first 4 weeks. She took part in a formal hip-specific physical therapy program for a total of 12 weeks. She was seen in clinic at 2, 6, and 12 weeks postoperatively and appeared to have had a typical, uneventful course. We advised her to gradually return to normal activities as tolerated at the 12-week visit.
Four months after the procedure, the patient returned to our clinic for evaluation after a right hip dislocation. Two days prior, she was at a school function with her child and experienced sudden pain and inability to bear weight after she extended and externally rotated her right hip in a low-energy manner. She was taken to an emergency room and found to have an anterior dislocation of the right hip (Figure 3), which was concentrically reduced under anesthesia.
Upon questioning, she reported having had feelings of mild instability of the right hip during demanding activities (jogging, yoga) after sustaining a low-energy fall 1 month prior to her dislocation. On examination, she had significant apprehension about the right hip during gentle external rotation maneuvers. An MRA 2 weeks after the dislocation showed a large defect of the anterosuperior capsuloligamentous complex measuring 4 cm from medial to lateral and 2.5 cm superior to inferior (Figure 4). No loose bodies, chondral injuries, or recurrent tears of the labrum were seen. Typical postoperative changes were observed at the femoral head-neck junction.
Initially, we recommended nonoperative management with 6 weeks of toe-touch weight-bearing and strict avoidance of hip extension–external rotation maneuvers. No hip orthosis was used. After this period, the patient advanced to full weight-bearing and continued in hip-specific physical therapy. Despite continued therapy and avoidance of provocative maneuvers, the patient reported persistent feelings of right hip instability with significant apprehension during extension and external rotation of the right hip. A repeat MRA 4 months after the hip dislocation showed a persistent defect in the anterosuperior capsuloligamentous complex and no signs of avascular necrosis. After 6 months of conservative treatment, we recommended an open capsulorrhaphy of the right hip with autograft iliotibial band reconstruction of the iliofemoral ligament and capsule.
Six months after the dislocation, the patient underwent the recommended procedure. After induction of general anesthesia, she was placed in the supine position on a standard operating table. A Smith-Petersen approach was used to visualize the anterior hip structures. During deep dissection, we observed a large defect, measuring 2.5×4 cm (Figure 5A), in the anterior hip capsule, with only a thin pseudocapsule covering the femoral head. Extensive mobilization of the anterior capsule was unsuccessful.
The decision was made to harvest a graft from the patient’s ipsilateral iliotibial band. A skin incision was made over the iliotibial band in the distal midthigh region, and a 2.5×4-cm graft was harvested from the central portion of the iliotibial band. An arthrotomy was performed on the hip joint (Figure 5B). The labrum appeared healthy without recurrent tearing or fraying, and other than focal thinning on the superior acetabulum, the cartilage appeared healthy. A double-loaded anchor was placed in the supra-acetabular region, and the sutures were passed through the graft. Then, No. 2 nonabsorbable sutures were sequentially placed between the capsular remnant and the graft medially, inferiorly, and laterally. The graft was placed into position (Figure 5C) and the sutures were tied (Figure 5D).
Postoperatively, the patient was allowed toe-touch weight-bearing for 6 weeks, with strict avoidance of extension–external rotation maneuvers. She participated in a 12-week course of physical therapy with gradual advancement of activities. About a year after the capsulorrhaphy, she was able to resume all previous activities with only occasional low-level discomfort. She returned to the clinic 16 months after the capsulorrhaphy complaining of increased pain with long-distance running but denied feelings of instability. We performed an intra-articular hip injection under ultrasound guidance, which provided 100% relief of her symptoms. We obtained an MRA to evaluate for any recurrent capsular or labral injury (Figure 6). The previous anterosuperior capsular defect was not visible, and no signs of recurrent labral or cartilage injury were seen.
Discussion
With the increasing popularity of hip arthroscopy, more complications are being reported as well, including postoperative hip instability. Three separate cases of anterior hip instability have been published in the past several years.5-7
Ranawat and colleagues5 were the first to report a case of postoperative anterior hip dislocation after arthroscopy. Their patient was a 52-year-old woman with right hip pain and generalized ligamentous laxity. Her preoperative radiographs showed no evidence of degenerative changes, dysplasia, or femoroacetabular impingement. An MRA showed a peripheral tear of the anterosuperior labrum. At arthroscopy, her right hip was easily distracted 2 to 3 cm with what they described as “minimal traction.” A small 1- to 2-cm capsulotomy was performed about the anterior portal. A detached labral tear was identified and repaired with an anchor, and no rim resection was performed. To improve visualization of the peripheral compartment, they extended the previous capsulotomy 1 to 2 cm and débrided the edges. A cam-type lesion was identified and resected. Lastly, they performed an anterior capsular plication, specifically including the iliofemoral ligament. Postoperatively, the patient wore a hip orthosis for 6 weeks to prevent extension and external rotation of the hip as well as a foot brace at night for 3 weeks. The patient was allowed to partially bear weight for the first 6 weeks with use of crutches. Approximately 2 months postoperatively, she slipped and fell down a short flight of stairs. She was diagnosed with an anterior hip dislocation. After successful closed reduction, she was treated conservatively with the same regimen used earlier. She remained symptomatic over the next several months with signs of instability and apprehension, and she eventually underwent a repeat hip arthroscopy. A 1- to 2-cm tear of the anterior capsule and iliofemoral ligament was treated with a revision arthroscopic capsular plication. A postoperative regimen similar to that used at the index procedure was instituted and, at most recent follow-up, she was found to have occasional pain without instability.
Matsuda6 reported a case of acute iatrogenic hip dislocation after arthroscopic surgery. His patient was a 39-year-old woman with a mildly retroverted acetabulum leading to impingement about the hip. She had no signs of generalized ligamentous laxity. A hip arthroscopy in the lateral position was performed, with no comment about the extent of the capsulotomy. During the procedure, about 5 mm of anterosuperior acetabulum were removed as part of arthroscopic rim trimming for treatment of the pincer lesion. A femoral osteochondroplasty was also performed (unspecified size) to restore more normal anterolateral offset. One confounding factor was that supranormal hip distraction was needed for 20 minutes to aid in removal of a metallic piece from a radiofrequency ablator, which inadvertently detached. The patient experienced an anterior hip dislocation in the recovery room and was found to be unstable during closed reduction under general anesthesia. A mini-open capsular repair was performed, which showed a 1×1.5-cm defect in the anterolateral capsule. After closure of the defect, the hip was found to be stable under fluoroscopic examination. Postoperatively, the patient was allowed to perform partial weight-bearing in a hip-knee-ankle-foot orthosis for 2 months and then a flexible hip brace for 1 month. At 15-month follow-up, her hip was stable and she was pain-free.
Benali and Katthagen7 highlighted the significant contribution of the labrum to hip stability in a dysplastic hip. Their patient was a 49-year-old woman with mild hip dysplasia and a degenerative bucket-handle tear of the ventrolateral labrum. The patient underwent a near-complete labral resection and rim trimming at an outside institution. The patient began full weight-bearing at 3 weeks postoperatively and noticed considerable groin and back pain (no hip orthosis use was mentioned). After failed treatment for suspected lumbar pathology, she was referred to the authors’ clinic for further evaluation. Plain radiographs showed subluxation of the left hip with degenerative changes. The patient had an uneventful left total hip arthroplasty (THA).
After reviewing the 3 reported cases of hip instability after arthroscopy, we suggest that surgeons fully recognize and appreciate the delicate balance of stability and motion provided by the static and dynamic stabilizers of the hip joint, and be cognizant of potential imbalance created by surgical intervention.8,9 Postarthroscopic hip instability appears to be multifactorial in nature, because all of the reported cases detailed different factors, both patient- and surgeon-related, contributing to instability.
Ranawat and colleagues5 identified several factors that may have contributed to the anterior hip dislocation sustained by their patient, including the patient’s generalized ligamentous laxity, performance of a capsulectomy (with repair of iliofemoral ligament), and a traumatic fall. Benali and Katthagen7 (although they did not perform the index procedure) described the disastrous complication of overzealous labral resection and rim trimming in a patient with hip dysplasia. Matsuda6 performed a labral resection and rim trimming, an extended (unspecified size) capsulotomy, and also used supranormal traction for 20 minutes to remove an iatrogenic foreign body. Surgeons performing hip arthroscopies should be aware of all these factors, because many are directly controlled by the surgeon.
The only factor we feel may have contributed to hip instability in our patient was the performance of a capsulotomy without closure. Our patient was an otherwise healthy woman with no signs of ligamentous laxity, hip dysplasia, or retroversion of the acetabulum. We did not perform a labral resection or rim trimming. We use modified anterior and anterolateral portals, and electrocautery to connect the portals. This typically leads to a release of a thin strip (less than 5 mm wide) of 3 cm of capsule. Based upon findings at rare second-look arthroscopy for recurrent symptoms, Dr. Guanche has observed that the capsulotomy from the initial procedure heals with normal-appearing tissue. Also, during peripheral compartment arthroscopy, we do not routinely release the iliofemoral ligament, and the orbicular ligament is left intact. Instead, we prefer to flex the hip and débride only enough capsular tissue to allow for adequate visualization.
Little has been published on capsulotomy closure after hip arthroscopy, and no consensus exists. Our standard practice is to not close the capsulotomy, which accords with the practice of other surgeons.9 There is concern, however, that extensive capsulotomy leading to injury or disruption of the iliofemoral ligament may cause anterior hip instability, driving other prominent hip arthroscopists to routinely close the capsulotomy.9,10 Myers and colleagues10 published a recent biomechanical study on the role of the labrum and the capsular ligaments in hip stability. They concluded that the iliofemoral ligament plays a significant role in limiting external rotation and anterior translation of the femoral head, and recommended closure of the capsulotomy after arthroscopy. Of note, Dr. Guanche has performed more than 1500 hip arthroscopic procedures in the past 5 years, and we are aware of only 2 patients who have sustained anterior hip dislocations, in spite of our not closing the capsulotomy defect. This highlights an important clinical question in need of further investigation.
Our case also raises questions about the ideal postoperative regimen after standard hip arthroscopy. Although we do not routinely prescribe hip orthoses for our patients, others do.5 We are unaware of any proven benefit to the standard use of hip orthoses, and are concerned over the possible lack of patient compliance and of adequate restraint. We prefer to educate our patients on avoiding the “at-risk” position of hip extension and external rotation and to counsel them on gradual return to activities. Studies are needed to determine the role of these devices in hip arthroscopy, as well as the ideal postoperative activity regimen.
Our patient failed 6 months of conservative treatment after her dislocation and continued to have feelings of hip instability even during light activities. As a result of this failure and given an anatomical defect in the anterior capsuloligamentous complex, we decided our patient would be best treated with reconstruction of the defect. We did not think a revision capsular plication, as done by Ranawat and colleagues,5 was a reasonable option for our patient because of a large defect in the capsular tissue. Even in smaller defects, plication could potentially lead to overtightening of the capsule and subsequent overconstraint of the joint. Also, plication of defects may place excessive strain on the suture, which may fail if the repair is even mildly stressed.
Recurrent anterior hip dislocations, although rare in their own right, are much more common after THA than after hip arthroscopy.11 Fujishiro and colleagues12 described a similar technique to ours developed to treat a patient with recurrent anterior hip instability from anterior capsular insufficiency after multiple revision THA procedures. They used a Leeds-Keio artificial ligament to reconstruct the iliofemoral ligament, and this successfully treated their patient’s instability.
Conclusion
We believe this is the first report of recurrent instability after hip arthroscopy, necessitating reconstruction of the anterior capsuloligamentous complex. This case shows that reconstruction of the iliofemoral ligament with iliotibial band autograft is safe, restores hip stability without compromising function, and should be considered by any hip arthroscopist encountering a similar scenario. It also highlights the importance of the capsuloligamentous complex surrounding the hip joint for its stability and the need for further research to better delineate the indications for capsular repair/closure after capsulotomy.
1. Ilizaliturri VM Jr. Complications of arthroscopic femoroacetabular impingement treatment: a review. Clin Orthop. 2009;467(3):760-768.
2. Clarke MT, Villar RN. Hip arthroscopy: complications in 1054 cases. Clin Orthop. 2003;406:84-88.
3. Smart LR, Oetgen M, Noonan B, Medvecky M. Beginning hip arthroscopy: indications, positioning, portals, basic techniques, and complications. Arthroscopy. 2007;23(12):1348-1353.
4. Sampson TG. Complications of hip arthroscopy. Tech Orthop. 2005;20:63-66.
5. Ranawat AS, McClincy M, Sekiya JK. Anterior dislocation of the hip after arthroscopy in a patient with capsular laxity of the hip. A case report. J Bone Joint Surg Am. 2009;91(1):192-197.
6. Matsuda DK. Acute iatrogenic dislocation following hip impingement arthroscopic surgery. Arthroscopy. 2009;25(4):400-404.
7. Benali Y, Katthagen BD. Hip subluxation as a complication of arthroscopic debridement. Arthroscopy. 2009;25(4):405-407.
8. Shindle MK, Voos JE, Nho SJ, Heyworth BE, Kelly BT. Arthroscopic management of labral tears in the hip. J Bone Joint Surg Am. 2008;90(suppl 4):2-19.
9. Bedi A, Galano G, Walsh C, Kelly BT. Capsular management during hip arthroscopy: from femoroacetabular impingement to instability. Arthroscopy. 2011;27(12):1720-1731.
10. Myers CA, Register BC, Lertwanich P, et al. Role of the acetabular labrum and the iliofemoral ligament in hip stability: an in vitro biplane fluoroscopy study. Am J Sports Med. 2011;39(suppl):85S-91S.
11. Sariali E, Leonard P, Mamoudy P. Dislocation after total hip arthroplasty using Hueter anterior approach. J Arthroplasty. 2008;23(2):266-272.
12. Fujishiro T, Nishikawa T, Takikawa S, Saegusa Y, Yoshiya S, Kurosaka M. Reconstruction of the iliofemoral ligament with an artificial ligament for recurrent anterior dislocation of total hip arthroplasty. J Arthroplasty. 2003;18(4):524-527.
Hip arthroscopy has experienced a dramatic increase in popularity, largely resulting from improvements in techniques and technology.1,2 As with any procedure, there are complications associated with arthroscopy of the hip. These include neurapraxia, iatrogenic cartilage and labral injuries, postoperative bleeding, perineal skin necrosis, infection, intra-articular instrument breakage, intra-abdominal fluid extravasation, avascular necrosis, and femoral neck fracture.1-4 Many of these have been attributed to the expected learning curve seen with any new procedure, and are less likely to occur as surgeons become more familiar with the procedure.1 One rare but serious complication is anterior dislocation of the hip.5-7
We present a patient who experienced an anterior hip dislocation and instability after hip arthroscopy, and was successfully treated with an anterior capsuloligamentous reconstruction. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
An otherwise healthy 37-year-old woman presented to our clinic with a 6-month history of right groin pain and an occasional popping sensation during activity, which was unresponsive to hip-specific physical therapy. On physical examination, she was 5 ft 10 in tall, weighed 150 lbs, and appeared in excellent physical condition. She had no signs of systemic ligamentous laxity. She had an otherwise normal musculoskeletal, neurologic, and vascular examination in her bilateral lower extremities. She had a mild antalgic gait on the right leg.
The affected right hip could be flexed painfully to 120º, extended to 0º, adducted 20º, and abducted 45º. At 90º of flexion, her right hip could be externally rotated 30º and internally rotated 20º. Internal rotation during hip flexion beyond 90º caused sharp pain in the groin. Her normal left hip could be flexed to 120º, extended to 0º, adducted 30º, and abducted 60º. At 90º of flexion, her left hip could be externally rotated 50º and internally rotated 30º. She had negative Ober tests bilaterally but had tenderness along the right iliotibial band. She had negative Patrick and Gaenslen tests bilaterally. She had no tenderness in the area of either greater trochanter.
Imaging evaluation included plain radiographs and a magnetic resonance arthrogram (MRA) of the right hip. The plain radiographs showed signs of femoroacetabular impingement, but no joint space narrowing, no dysplasia, and no retroversion of the acetabulum (Figures 1A, 1B). The MRA showed a degenerative peripheral tear of the anterosuperior labrum without significant cartilage wear (Figure 2).
Based upon her findings on physical examination and imaging, we recommended arthroscopic treatment of her right hip pathology. Thirteen months after initial presentation, we performed a right hip arthroscopy with the patient in the supine position. Through modified anterior and anterolateral portals, we used electrocautery to perform a capsulotomy from the 9 o’clock to 12 o’clock positions. A central compartment diagnostic arthroscopy showed mild degenerative fraying of the labrum from the 9 o’clock to 12 o’clock positions without signs of detachment. There was grade III chondral fraying near the articular margin in that same arc. The femoral articular cartilage appeared normal, as did the ligamentum teres. We used a shaver to gently débride the torn labrum down to stable tissue. The frayed cartilage on the acetabulum was also gently débrided.
Traction was released and the hip was flexed. Minimal capsular release and débridement were performed for adequate visualization of the peripheral compartment. A diagnostic examination revealed a significant cam-type impingement lesion from the 12 o’clock to 6 o’clock positions. We performed a femoral neck resection, with a proximal-distal dimension of 15 mm and a depth of 7 mm. A dynamic fluoroscopic examination of the hip joint showed no signs of impingement. In accordance with our standard protocol, the anterior capsulotomy was not repaired.
Postoperatively, the patient was instructed to perform toe-touch weight-bearing with crutches for 2 weeks and to advance to full weight-bearing over the next 2 weeks. She did not use a hip orthosis. She was also advised to avoid combined hip extension/external rotation maneuvers for the first 4 weeks. She took part in a formal hip-specific physical therapy program for a total of 12 weeks. She was seen in clinic at 2, 6, and 12 weeks postoperatively and appeared to have had a typical, uneventful course. We advised her to gradually return to normal activities as tolerated at the 12-week visit.
Four months after the procedure, the patient returned to our clinic for evaluation after a right hip dislocation. Two days prior, she was at a school function with her child and experienced sudden pain and inability to bear weight after she extended and externally rotated her right hip in a low-energy manner. She was taken to an emergency room and found to have an anterior dislocation of the right hip (Figure 3), which was concentrically reduced under anesthesia.
Upon questioning, she reported having had feelings of mild instability of the right hip during demanding activities (jogging, yoga) after sustaining a low-energy fall 1 month prior to her dislocation. On examination, she had significant apprehension about the right hip during gentle external rotation maneuvers. An MRA 2 weeks after the dislocation showed a large defect of the anterosuperior capsuloligamentous complex measuring 4 cm from medial to lateral and 2.5 cm superior to inferior (Figure 4). No loose bodies, chondral injuries, or recurrent tears of the labrum were seen. Typical postoperative changes were observed at the femoral head-neck junction.
Initially, we recommended nonoperative management with 6 weeks of toe-touch weight-bearing and strict avoidance of hip extension–external rotation maneuvers. No hip orthosis was used. After this period, the patient advanced to full weight-bearing and continued in hip-specific physical therapy. Despite continued therapy and avoidance of provocative maneuvers, the patient reported persistent feelings of right hip instability with significant apprehension during extension and external rotation of the right hip. A repeat MRA 4 months after the hip dislocation showed a persistent defect in the anterosuperior capsuloligamentous complex and no signs of avascular necrosis. After 6 months of conservative treatment, we recommended an open capsulorrhaphy of the right hip with autograft iliotibial band reconstruction of the iliofemoral ligament and capsule.
Six months after the dislocation, the patient underwent the recommended procedure. After induction of general anesthesia, she was placed in the supine position on a standard operating table. A Smith-Petersen approach was used to visualize the anterior hip structures. During deep dissection, we observed a large defect, measuring 2.5×4 cm (Figure 5A), in the anterior hip capsule, with only a thin pseudocapsule covering the femoral head. Extensive mobilization of the anterior capsule was unsuccessful.
The decision was made to harvest a graft from the patient’s ipsilateral iliotibial band. A skin incision was made over the iliotibial band in the distal midthigh region, and a 2.5×4-cm graft was harvested from the central portion of the iliotibial band. An arthrotomy was performed on the hip joint (Figure 5B). The labrum appeared healthy without recurrent tearing or fraying, and other than focal thinning on the superior acetabulum, the cartilage appeared healthy. A double-loaded anchor was placed in the supra-acetabular region, and the sutures were passed through the graft. Then, No. 2 nonabsorbable sutures were sequentially placed between the capsular remnant and the graft medially, inferiorly, and laterally. The graft was placed into position (Figure 5C) and the sutures were tied (Figure 5D).
Postoperatively, the patient was allowed toe-touch weight-bearing for 6 weeks, with strict avoidance of extension–external rotation maneuvers. She participated in a 12-week course of physical therapy with gradual advancement of activities. About a year after the capsulorrhaphy, she was able to resume all previous activities with only occasional low-level discomfort. She returned to the clinic 16 months after the capsulorrhaphy complaining of increased pain with long-distance running but denied feelings of instability. We performed an intra-articular hip injection under ultrasound guidance, which provided 100% relief of her symptoms. We obtained an MRA to evaluate for any recurrent capsular or labral injury (Figure 6). The previous anterosuperior capsular defect was not visible, and no signs of recurrent labral or cartilage injury were seen.
Discussion
With the increasing popularity of hip arthroscopy, more complications are being reported as well, including postoperative hip instability. Three separate cases of anterior hip instability have been published in the past several years.5-7
Ranawat and colleagues5 were the first to report a case of postoperative anterior hip dislocation after arthroscopy. Their patient was a 52-year-old woman with right hip pain and generalized ligamentous laxity. Her preoperative radiographs showed no evidence of degenerative changes, dysplasia, or femoroacetabular impingement. An MRA showed a peripheral tear of the anterosuperior labrum. At arthroscopy, her right hip was easily distracted 2 to 3 cm with what they described as “minimal traction.” A small 1- to 2-cm capsulotomy was performed about the anterior portal. A detached labral tear was identified and repaired with an anchor, and no rim resection was performed. To improve visualization of the peripheral compartment, they extended the previous capsulotomy 1 to 2 cm and débrided the edges. A cam-type lesion was identified and resected. Lastly, they performed an anterior capsular plication, specifically including the iliofemoral ligament. Postoperatively, the patient wore a hip orthosis for 6 weeks to prevent extension and external rotation of the hip as well as a foot brace at night for 3 weeks. The patient was allowed to partially bear weight for the first 6 weeks with use of crutches. Approximately 2 months postoperatively, she slipped and fell down a short flight of stairs. She was diagnosed with an anterior hip dislocation. After successful closed reduction, she was treated conservatively with the same regimen used earlier. She remained symptomatic over the next several months with signs of instability and apprehension, and she eventually underwent a repeat hip arthroscopy. A 1- to 2-cm tear of the anterior capsule and iliofemoral ligament was treated with a revision arthroscopic capsular plication. A postoperative regimen similar to that used at the index procedure was instituted and, at most recent follow-up, she was found to have occasional pain without instability.
Matsuda6 reported a case of acute iatrogenic hip dislocation after arthroscopic surgery. His patient was a 39-year-old woman with a mildly retroverted acetabulum leading to impingement about the hip. She had no signs of generalized ligamentous laxity. A hip arthroscopy in the lateral position was performed, with no comment about the extent of the capsulotomy. During the procedure, about 5 mm of anterosuperior acetabulum were removed as part of arthroscopic rim trimming for treatment of the pincer lesion. A femoral osteochondroplasty was also performed (unspecified size) to restore more normal anterolateral offset. One confounding factor was that supranormal hip distraction was needed for 20 minutes to aid in removal of a metallic piece from a radiofrequency ablator, which inadvertently detached. The patient experienced an anterior hip dislocation in the recovery room and was found to be unstable during closed reduction under general anesthesia. A mini-open capsular repair was performed, which showed a 1×1.5-cm defect in the anterolateral capsule. After closure of the defect, the hip was found to be stable under fluoroscopic examination. Postoperatively, the patient was allowed to perform partial weight-bearing in a hip-knee-ankle-foot orthosis for 2 months and then a flexible hip brace for 1 month. At 15-month follow-up, her hip was stable and she was pain-free.
Benali and Katthagen7 highlighted the significant contribution of the labrum to hip stability in a dysplastic hip. Their patient was a 49-year-old woman with mild hip dysplasia and a degenerative bucket-handle tear of the ventrolateral labrum. The patient underwent a near-complete labral resection and rim trimming at an outside institution. The patient began full weight-bearing at 3 weeks postoperatively and noticed considerable groin and back pain (no hip orthosis use was mentioned). After failed treatment for suspected lumbar pathology, she was referred to the authors’ clinic for further evaluation. Plain radiographs showed subluxation of the left hip with degenerative changes. The patient had an uneventful left total hip arthroplasty (THA).
After reviewing the 3 reported cases of hip instability after arthroscopy, we suggest that surgeons fully recognize and appreciate the delicate balance of stability and motion provided by the static and dynamic stabilizers of the hip joint, and be cognizant of potential imbalance created by surgical intervention.8,9 Postarthroscopic hip instability appears to be multifactorial in nature, because all of the reported cases detailed different factors, both patient- and surgeon-related, contributing to instability.
Ranawat and colleagues5 identified several factors that may have contributed to the anterior hip dislocation sustained by their patient, including the patient’s generalized ligamentous laxity, performance of a capsulectomy (with repair of iliofemoral ligament), and a traumatic fall. Benali and Katthagen7 (although they did not perform the index procedure) described the disastrous complication of overzealous labral resection and rim trimming in a patient with hip dysplasia. Matsuda6 performed a labral resection and rim trimming, an extended (unspecified size) capsulotomy, and also used supranormal traction for 20 minutes to remove an iatrogenic foreign body. Surgeons performing hip arthroscopies should be aware of all these factors, because many are directly controlled by the surgeon.
The only factor we feel may have contributed to hip instability in our patient was the performance of a capsulotomy without closure. Our patient was an otherwise healthy woman with no signs of ligamentous laxity, hip dysplasia, or retroversion of the acetabulum. We did not perform a labral resection or rim trimming. We use modified anterior and anterolateral portals, and electrocautery to connect the portals. This typically leads to a release of a thin strip (less than 5 mm wide) of 3 cm of capsule. Based upon findings at rare second-look arthroscopy for recurrent symptoms, Dr. Guanche has observed that the capsulotomy from the initial procedure heals with normal-appearing tissue. Also, during peripheral compartment arthroscopy, we do not routinely release the iliofemoral ligament, and the orbicular ligament is left intact. Instead, we prefer to flex the hip and débride only enough capsular tissue to allow for adequate visualization.
Little has been published on capsulotomy closure after hip arthroscopy, and no consensus exists. Our standard practice is to not close the capsulotomy, which accords with the practice of other surgeons.9 There is concern, however, that extensive capsulotomy leading to injury or disruption of the iliofemoral ligament may cause anterior hip instability, driving other prominent hip arthroscopists to routinely close the capsulotomy.9,10 Myers and colleagues10 published a recent biomechanical study on the role of the labrum and the capsular ligaments in hip stability. They concluded that the iliofemoral ligament plays a significant role in limiting external rotation and anterior translation of the femoral head, and recommended closure of the capsulotomy after arthroscopy. Of note, Dr. Guanche has performed more than 1500 hip arthroscopic procedures in the past 5 years, and we are aware of only 2 patients who have sustained anterior hip dislocations, in spite of our not closing the capsulotomy defect. This highlights an important clinical question in need of further investigation.
Our case also raises questions about the ideal postoperative regimen after standard hip arthroscopy. Although we do not routinely prescribe hip orthoses for our patients, others do.5 We are unaware of any proven benefit to the standard use of hip orthoses, and are concerned over the possible lack of patient compliance and of adequate restraint. We prefer to educate our patients on avoiding the “at-risk” position of hip extension and external rotation and to counsel them on gradual return to activities. Studies are needed to determine the role of these devices in hip arthroscopy, as well as the ideal postoperative activity regimen.
Our patient failed 6 months of conservative treatment after her dislocation and continued to have feelings of hip instability even during light activities. As a result of this failure and given an anatomical defect in the anterior capsuloligamentous complex, we decided our patient would be best treated with reconstruction of the defect. We did not think a revision capsular plication, as done by Ranawat and colleagues,5 was a reasonable option for our patient because of a large defect in the capsular tissue. Even in smaller defects, plication could potentially lead to overtightening of the capsule and subsequent overconstraint of the joint. Also, plication of defects may place excessive strain on the suture, which may fail if the repair is even mildly stressed.
Recurrent anterior hip dislocations, although rare in their own right, are much more common after THA than after hip arthroscopy.11 Fujishiro and colleagues12 described a similar technique to ours developed to treat a patient with recurrent anterior hip instability from anterior capsular insufficiency after multiple revision THA procedures. They used a Leeds-Keio artificial ligament to reconstruct the iliofemoral ligament, and this successfully treated their patient’s instability.
Conclusion
We believe this is the first report of recurrent instability after hip arthroscopy, necessitating reconstruction of the anterior capsuloligamentous complex. This case shows that reconstruction of the iliofemoral ligament with iliotibial band autograft is safe, restores hip stability without compromising function, and should be considered by any hip arthroscopist encountering a similar scenario. It also highlights the importance of the capsuloligamentous complex surrounding the hip joint for its stability and the need for further research to better delineate the indications for capsular repair/closure after capsulotomy.
Hip arthroscopy has experienced a dramatic increase in popularity, largely resulting from improvements in techniques and technology.1,2 As with any procedure, there are complications associated with arthroscopy of the hip. These include neurapraxia, iatrogenic cartilage and labral injuries, postoperative bleeding, perineal skin necrosis, infection, intra-articular instrument breakage, intra-abdominal fluid extravasation, avascular necrosis, and femoral neck fracture.1-4 Many of these have been attributed to the expected learning curve seen with any new procedure, and are less likely to occur as surgeons become more familiar with the procedure.1 One rare but serious complication is anterior dislocation of the hip.5-7
We present a patient who experienced an anterior hip dislocation and instability after hip arthroscopy, and was successfully treated with an anterior capsuloligamentous reconstruction. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
An otherwise healthy 37-year-old woman presented to our clinic with a 6-month history of right groin pain and an occasional popping sensation during activity, which was unresponsive to hip-specific physical therapy. On physical examination, she was 5 ft 10 in tall, weighed 150 lbs, and appeared in excellent physical condition. She had no signs of systemic ligamentous laxity. She had an otherwise normal musculoskeletal, neurologic, and vascular examination in her bilateral lower extremities. She had a mild antalgic gait on the right leg.
The affected right hip could be flexed painfully to 120º, extended to 0º, adducted 20º, and abducted 45º. At 90º of flexion, her right hip could be externally rotated 30º and internally rotated 20º. Internal rotation during hip flexion beyond 90º caused sharp pain in the groin. Her normal left hip could be flexed to 120º, extended to 0º, adducted 30º, and abducted 60º. At 90º of flexion, her left hip could be externally rotated 50º and internally rotated 30º. She had negative Ober tests bilaterally but had tenderness along the right iliotibial band. She had negative Patrick and Gaenslen tests bilaterally. She had no tenderness in the area of either greater trochanter.
Imaging evaluation included plain radiographs and a magnetic resonance arthrogram (MRA) of the right hip. The plain radiographs showed signs of femoroacetabular impingement, but no joint space narrowing, no dysplasia, and no retroversion of the acetabulum (Figures 1A, 1B). The MRA showed a degenerative peripheral tear of the anterosuperior labrum without significant cartilage wear (Figure 2).
Based upon her findings on physical examination and imaging, we recommended arthroscopic treatment of her right hip pathology. Thirteen months after initial presentation, we performed a right hip arthroscopy with the patient in the supine position. Through modified anterior and anterolateral portals, we used electrocautery to perform a capsulotomy from the 9 o’clock to 12 o’clock positions. A central compartment diagnostic arthroscopy showed mild degenerative fraying of the labrum from the 9 o’clock to 12 o’clock positions without signs of detachment. There was grade III chondral fraying near the articular margin in that same arc. The femoral articular cartilage appeared normal, as did the ligamentum teres. We used a shaver to gently débride the torn labrum down to stable tissue. The frayed cartilage on the acetabulum was also gently débrided.
Traction was released and the hip was flexed. Minimal capsular release and débridement were performed for adequate visualization of the peripheral compartment. A diagnostic examination revealed a significant cam-type impingement lesion from the 12 o’clock to 6 o’clock positions. We performed a femoral neck resection, with a proximal-distal dimension of 15 mm and a depth of 7 mm. A dynamic fluoroscopic examination of the hip joint showed no signs of impingement. In accordance with our standard protocol, the anterior capsulotomy was not repaired.
Postoperatively, the patient was instructed to perform toe-touch weight-bearing with crutches for 2 weeks and to advance to full weight-bearing over the next 2 weeks. She did not use a hip orthosis. She was also advised to avoid combined hip extension/external rotation maneuvers for the first 4 weeks. She took part in a formal hip-specific physical therapy program for a total of 12 weeks. She was seen in clinic at 2, 6, and 12 weeks postoperatively and appeared to have had a typical, uneventful course. We advised her to gradually return to normal activities as tolerated at the 12-week visit.
Four months after the procedure, the patient returned to our clinic for evaluation after a right hip dislocation. Two days prior, she was at a school function with her child and experienced sudden pain and inability to bear weight after she extended and externally rotated her right hip in a low-energy manner. She was taken to an emergency room and found to have an anterior dislocation of the right hip (Figure 3), which was concentrically reduced under anesthesia.
Upon questioning, she reported having had feelings of mild instability of the right hip during demanding activities (jogging, yoga) after sustaining a low-energy fall 1 month prior to her dislocation. On examination, she had significant apprehension about the right hip during gentle external rotation maneuvers. An MRA 2 weeks after the dislocation showed a large defect of the anterosuperior capsuloligamentous complex measuring 4 cm from medial to lateral and 2.5 cm superior to inferior (Figure 4). No loose bodies, chondral injuries, or recurrent tears of the labrum were seen. Typical postoperative changes were observed at the femoral head-neck junction.
Initially, we recommended nonoperative management with 6 weeks of toe-touch weight-bearing and strict avoidance of hip extension–external rotation maneuvers. No hip orthosis was used. After this period, the patient advanced to full weight-bearing and continued in hip-specific physical therapy. Despite continued therapy and avoidance of provocative maneuvers, the patient reported persistent feelings of right hip instability with significant apprehension during extension and external rotation of the right hip. A repeat MRA 4 months after the hip dislocation showed a persistent defect in the anterosuperior capsuloligamentous complex and no signs of avascular necrosis. After 6 months of conservative treatment, we recommended an open capsulorrhaphy of the right hip with autograft iliotibial band reconstruction of the iliofemoral ligament and capsule.
Six months after the dislocation, the patient underwent the recommended procedure. After induction of general anesthesia, she was placed in the supine position on a standard operating table. A Smith-Petersen approach was used to visualize the anterior hip structures. During deep dissection, we observed a large defect, measuring 2.5×4 cm (Figure 5A), in the anterior hip capsule, with only a thin pseudocapsule covering the femoral head. Extensive mobilization of the anterior capsule was unsuccessful.
The decision was made to harvest a graft from the patient’s ipsilateral iliotibial band. A skin incision was made over the iliotibial band in the distal midthigh region, and a 2.5×4-cm graft was harvested from the central portion of the iliotibial band. An arthrotomy was performed on the hip joint (Figure 5B). The labrum appeared healthy without recurrent tearing or fraying, and other than focal thinning on the superior acetabulum, the cartilage appeared healthy. A double-loaded anchor was placed in the supra-acetabular region, and the sutures were passed through the graft. Then, No. 2 nonabsorbable sutures were sequentially placed between the capsular remnant and the graft medially, inferiorly, and laterally. The graft was placed into position (Figure 5C) and the sutures were tied (Figure 5D).
Postoperatively, the patient was allowed toe-touch weight-bearing for 6 weeks, with strict avoidance of extension–external rotation maneuvers. She participated in a 12-week course of physical therapy with gradual advancement of activities. About a year after the capsulorrhaphy, she was able to resume all previous activities with only occasional low-level discomfort. She returned to the clinic 16 months after the capsulorrhaphy complaining of increased pain with long-distance running but denied feelings of instability. We performed an intra-articular hip injection under ultrasound guidance, which provided 100% relief of her symptoms. We obtained an MRA to evaluate for any recurrent capsular or labral injury (Figure 6). The previous anterosuperior capsular defect was not visible, and no signs of recurrent labral or cartilage injury were seen.
Discussion
With the increasing popularity of hip arthroscopy, more complications are being reported as well, including postoperative hip instability. Three separate cases of anterior hip instability have been published in the past several years.5-7
Ranawat and colleagues5 were the first to report a case of postoperative anterior hip dislocation after arthroscopy. Their patient was a 52-year-old woman with right hip pain and generalized ligamentous laxity. Her preoperative radiographs showed no evidence of degenerative changes, dysplasia, or femoroacetabular impingement. An MRA showed a peripheral tear of the anterosuperior labrum. At arthroscopy, her right hip was easily distracted 2 to 3 cm with what they described as “minimal traction.” A small 1- to 2-cm capsulotomy was performed about the anterior portal. A detached labral tear was identified and repaired with an anchor, and no rim resection was performed. To improve visualization of the peripheral compartment, they extended the previous capsulotomy 1 to 2 cm and débrided the edges. A cam-type lesion was identified and resected. Lastly, they performed an anterior capsular plication, specifically including the iliofemoral ligament. Postoperatively, the patient wore a hip orthosis for 6 weeks to prevent extension and external rotation of the hip as well as a foot brace at night for 3 weeks. The patient was allowed to partially bear weight for the first 6 weeks with use of crutches. Approximately 2 months postoperatively, she slipped and fell down a short flight of stairs. She was diagnosed with an anterior hip dislocation. After successful closed reduction, she was treated conservatively with the same regimen used earlier. She remained symptomatic over the next several months with signs of instability and apprehension, and she eventually underwent a repeat hip arthroscopy. A 1- to 2-cm tear of the anterior capsule and iliofemoral ligament was treated with a revision arthroscopic capsular plication. A postoperative regimen similar to that used at the index procedure was instituted and, at most recent follow-up, she was found to have occasional pain without instability.
Matsuda6 reported a case of acute iatrogenic hip dislocation after arthroscopic surgery. His patient was a 39-year-old woman with a mildly retroverted acetabulum leading to impingement about the hip. She had no signs of generalized ligamentous laxity. A hip arthroscopy in the lateral position was performed, with no comment about the extent of the capsulotomy. During the procedure, about 5 mm of anterosuperior acetabulum were removed as part of arthroscopic rim trimming for treatment of the pincer lesion. A femoral osteochondroplasty was also performed (unspecified size) to restore more normal anterolateral offset. One confounding factor was that supranormal hip distraction was needed for 20 minutes to aid in removal of a metallic piece from a radiofrequency ablator, which inadvertently detached. The patient experienced an anterior hip dislocation in the recovery room and was found to be unstable during closed reduction under general anesthesia. A mini-open capsular repair was performed, which showed a 1×1.5-cm defect in the anterolateral capsule. After closure of the defect, the hip was found to be stable under fluoroscopic examination. Postoperatively, the patient was allowed to perform partial weight-bearing in a hip-knee-ankle-foot orthosis for 2 months and then a flexible hip brace for 1 month. At 15-month follow-up, her hip was stable and she was pain-free.
Benali and Katthagen7 highlighted the significant contribution of the labrum to hip stability in a dysplastic hip. Their patient was a 49-year-old woman with mild hip dysplasia and a degenerative bucket-handle tear of the ventrolateral labrum. The patient underwent a near-complete labral resection and rim trimming at an outside institution. The patient began full weight-bearing at 3 weeks postoperatively and noticed considerable groin and back pain (no hip orthosis use was mentioned). After failed treatment for suspected lumbar pathology, she was referred to the authors’ clinic for further evaluation. Plain radiographs showed subluxation of the left hip with degenerative changes. The patient had an uneventful left total hip arthroplasty (THA).
After reviewing the 3 reported cases of hip instability after arthroscopy, we suggest that surgeons fully recognize and appreciate the delicate balance of stability and motion provided by the static and dynamic stabilizers of the hip joint, and be cognizant of potential imbalance created by surgical intervention.8,9 Postarthroscopic hip instability appears to be multifactorial in nature, because all of the reported cases detailed different factors, both patient- and surgeon-related, contributing to instability.
Ranawat and colleagues5 identified several factors that may have contributed to the anterior hip dislocation sustained by their patient, including the patient’s generalized ligamentous laxity, performance of a capsulectomy (with repair of iliofemoral ligament), and a traumatic fall. Benali and Katthagen7 (although they did not perform the index procedure) described the disastrous complication of overzealous labral resection and rim trimming in a patient with hip dysplasia. Matsuda6 performed a labral resection and rim trimming, an extended (unspecified size) capsulotomy, and also used supranormal traction for 20 minutes to remove an iatrogenic foreign body. Surgeons performing hip arthroscopies should be aware of all these factors, because many are directly controlled by the surgeon.
The only factor we feel may have contributed to hip instability in our patient was the performance of a capsulotomy without closure. Our patient was an otherwise healthy woman with no signs of ligamentous laxity, hip dysplasia, or retroversion of the acetabulum. We did not perform a labral resection or rim trimming. We use modified anterior and anterolateral portals, and electrocautery to connect the portals. This typically leads to a release of a thin strip (less than 5 mm wide) of 3 cm of capsule. Based upon findings at rare second-look arthroscopy for recurrent symptoms, Dr. Guanche has observed that the capsulotomy from the initial procedure heals with normal-appearing tissue. Also, during peripheral compartment arthroscopy, we do not routinely release the iliofemoral ligament, and the orbicular ligament is left intact. Instead, we prefer to flex the hip and débride only enough capsular tissue to allow for adequate visualization.
Little has been published on capsulotomy closure after hip arthroscopy, and no consensus exists. Our standard practice is to not close the capsulotomy, which accords with the practice of other surgeons.9 There is concern, however, that extensive capsulotomy leading to injury or disruption of the iliofemoral ligament may cause anterior hip instability, driving other prominent hip arthroscopists to routinely close the capsulotomy.9,10 Myers and colleagues10 published a recent biomechanical study on the role of the labrum and the capsular ligaments in hip stability. They concluded that the iliofemoral ligament plays a significant role in limiting external rotation and anterior translation of the femoral head, and recommended closure of the capsulotomy after arthroscopy. Of note, Dr. Guanche has performed more than 1500 hip arthroscopic procedures in the past 5 years, and we are aware of only 2 patients who have sustained anterior hip dislocations, in spite of our not closing the capsulotomy defect. This highlights an important clinical question in need of further investigation.
Our case also raises questions about the ideal postoperative regimen after standard hip arthroscopy. Although we do not routinely prescribe hip orthoses for our patients, others do.5 We are unaware of any proven benefit to the standard use of hip orthoses, and are concerned over the possible lack of patient compliance and of adequate restraint. We prefer to educate our patients on avoiding the “at-risk” position of hip extension and external rotation and to counsel them on gradual return to activities. Studies are needed to determine the role of these devices in hip arthroscopy, as well as the ideal postoperative activity regimen.
Our patient failed 6 months of conservative treatment after her dislocation and continued to have feelings of hip instability even during light activities. As a result of this failure and given an anatomical defect in the anterior capsuloligamentous complex, we decided our patient would be best treated with reconstruction of the defect. We did not think a revision capsular plication, as done by Ranawat and colleagues,5 was a reasonable option for our patient because of a large defect in the capsular tissue. Even in smaller defects, plication could potentially lead to overtightening of the capsule and subsequent overconstraint of the joint. Also, plication of defects may place excessive strain on the suture, which may fail if the repair is even mildly stressed.
Recurrent anterior hip dislocations, although rare in their own right, are much more common after THA than after hip arthroscopy.11 Fujishiro and colleagues12 described a similar technique to ours developed to treat a patient with recurrent anterior hip instability from anterior capsular insufficiency after multiple revision THA procedures. They used a Leeds-Keio artificial ligament to reconstruct the iliofemoral ligament, and this successfully treated their patient’s instability.
Conclusion
We believe this is the first report of recurrent instability after hip arthroscopy, necessitating reconstruction of the anterior capsuloligamentous complex. This case shows that reconstruction of the iliofemoral ligament with iliotibial band autograft is safe, restores hip stability without compromising function, and should be considered by any hip arthroscopist encountering a similar scenario. It also highlights the importance of the capsuloligamentous complex surrounding the hip joint for its stability and the need for further research to better delineate the indications for capsular repair/closure after capsulotomy.
1. Ilizaliturri VM Jr. Complications of arthroscopic femoroacetabular impingement treatment: a review. Clin Orthop. 2009;467(3):760-768.
2. Clarke MT, Villar RN. Hip arthroscopy: complications in 1054 cases. Clin Orthop. 2003;406:84-88.
3. Smart LR, Oetgen M, Noonan B, Medvecky M. Beginning hip arthroscopy: indications, positioning, portals, basic techniques, and complications. Arthroscopy. 2007;23(12):1348-1353.
4. Sampson TG. Complications of hip arthroscopy. Tech Orthop. 2005;20:63-66.
5. Ranawat AS, McClincy M, Sekiya JK. Anterior dislocation of the hip after arthroscopy in a patient with capsular laxity of the hip. A case report. J Bone Joint Surg Am. 2009;91(1):192-197.
6. Matsuda DK. Acute iatrogenic dislocation following hip impingement arthroscopic surgery. Arthroscopy. 2009;25(4):400-404.
7. Benali Y, Katthagen BD. Hip subluxation as a complication of arthroscopic debridement. Arthroscopy. 2009;25(4):405-407.
8. Shindle MK, Voos JE, Nho SJ, Heyworth BE, Kelly BT. Arthroscopic management of labral tears in the hip. J Bone Joint Surg Am. 2008;90(suppl 4):2-19.
9. Bedi A, Galano G, Walsh C, Kelly BT. Capsular management during hip arthroscopy: from femoroacetabular impingement to instability. Arthroscopy. 2011;27(12):1720-1731.
10. Myers CA, Register BC, Lertwanich P, et al. Role of the acetabular labrum and the iliofemoral ligament in hip stability: an in vitro biplane fluoroscopy study. Am J Sports Med. 2011;39(suppl):85S-91S.
11. Sariali E, Leonard P, Mamoudy P. Dislocation after total hip arthroplasty using Hueter anterior approach. J Arthroplasty. 2008;23(2):266-272.
12. Fujishiro T, Nishikawa T, Takikawa S, Saegusa Y, Yoshiya S, Kurosaka M. Reconstruction of the iliofemoral ligament with an artificial ligament for recurrent anterior dislocation of total hip arthroplasty. J Arthroplasty. 2003;18(4):524-527.
1. Ilizaliturri VM Jr. Complications of arthroscopic femoroacetabular impingement treatment: a review. Clin Orthop. 2009;467(3):760-768.
2. Clarke MT, Villar RN. Hip arthroscopy: complications in 1054 cases. Clin Orthop. 2003;406:84-88.
3. Smart LR, Oetgen M, Noonan B, Medvecky M. Beginning hip arthroscopy: indications, positioning, portals, basic techniques, and complications. Arthroscopy. 2007;23(12):1348-1353.
4. Sampson TG. Complications of hip arthroscopy. Tech Orthop. 2005;20:63-66.
5. Ranawat AS, McClincy M, Sekiya JK. Anterior dislocation of the hip after arthroscopy in a patient with capsular laxity of the hip. A case report. J Bone Joint Surg Am. 2009;91(1):192-197.
6. Matsuda DK. Acute iatrogenic dislocation following hip impingement arthroscopic surgery. Arthroscopy. 2009;25(4):400-404.
7. Benali Y, Katthagen BD. Hip subluxation as a complication of arthroscopic debridement. Arthroscopy. 2009;25(4):405-407.
8. Shindle MK, Voos JE, Nho SJ, Heyworth BE, Kelly BT. Arthroscopic management of labral tears in the hip. J Bone Joint Surg Am. 2008;90(suppl 4):2-19.
9. Bedi A, Galano G, Walsh C, Kelly BT. Capsular management during hip arthroscopy: from femoroacetabular impingement to instability. Arthroscopy. 2011;27(12):1720-1731.
10. Myers CA, Register BC, Lertwanich P, et al. Role of the acetabular labrum and the iliofemoral ligament in hip stability: an in vitro biplane fluoroscopy study. Am J Sports Med. 2011;39(suppl):85S-91S.
11. Sariali E, Leonard P, Mamoudy P. Dislocation after total hip arthroplasty using Hueter anterior approach. J Arthroplasty. 2008;23(2):266-272.
12. Fujishiro T, Nishikawa T, Takikawa S, Saegusa Y, Yoshiya S, Kurosaka M. Reconstruction of the iliofemoral ligament with an artificial ligament for recurrent anterior dislocation of total hip arthroplasty. J Arthroplasty. 2003;18(4):524-527.
Improved Function and Joint Kinematics After Correction of Tibial Malalignment
The tibia is the most commonly fractured long bone in adults, and tibial malunions occur in up to 60% of these patients.1,2 Persistent tibial malalignment, particularly varus alignment, negatively alters gait and joint kinematics, leading to altered weight-bearing forces across the knee and ankle joints. These altered forces may lead to osteoarthritis.3-8
Several studies have identified a relationship between extent of tibial malalignment and changes in joint reaction forces.3,5-7,9-13 Puno and colleagues14 developed a mathematical model to better define the changes in neighboring joints relative to the pattern of the tibia malalignment. Not surprisingly, their work showed that, with distal tibial malunions, altered stress concentrations were realized at the ankle joint, and more proximal tibial deformities led to larger alterations in the joint stresses at the knee. More recently, van der Schoot and colleagues8 found a high prevalence of ipsilateral ankle osteoarthritis with tibial malalignment of 5° or more, and Greenwood and colleagues15 showed a higher incidence of knee pain, lower limb osteoarthritis, and disability in patients with previous tibia fractures. Given these findings, it would seem that correction of tibial malalignment would lead to normative lower extremity joint kinematic values, joint reaction forces, and overall quality of life (QOL).
The ability to ambulate has been recognized as an important milestone in functional recovery after lower extremity injury.2,16,17 Gait analysis, assessment of joint kinematics, and QOL and health status questionnaires can provide information to evaluate rehabilitation protocols, treatment algorithms, and surgical outcomes. Recently, these measures have been used to assess patients recovering from acetabular fractures, femoral shaft fractures, and calcaneal fractures.4,11,17-24 However, no study has used these measures to assess the benefits of surgical correction of malaligned tibias.
We conducted a study to determine improvement in gait, joint kinematics, and patients’ perceptions of overall well-being after surgical correction of tibial malunions. The null hypothesis was that correction of tibial malunion would have no effect on gait, joint kinematics, or patients’ perceptions of function and QOL.
Materials and Methods
This prospective double-time-point study, which was approved by the Institutional Review Board of Washington University/Barnes-Jewish Hospital, evaluated 11 consecutive patients with a varus tibial malunion treated by a single surgeon between September 2003 and January 2006. All patients were treated using a technique that included oblique osteotomy and open reduction and internal fixation (ORIF) or osteotomy and intramedullary nailing. Study inclusion criteria were age 18 years or older; symptomatic varus malunion of the tibia of 10º or more; absence of a developmental or pathologic process leading to the fracture and subsequent deformity; no neurologic deficit of either lower extremity or contralateral lower extremity deformity; and ability to ambulate 9 meters with or without use of an assistive device.
The 11 patients (6 men, 5 women) who met these criteria enrolled in the study. Mean age was 53 years (range, 43-68 years). Eight malunions involved the left tibia. The mechanisms of injury were motor vehicle crash (6 patients), fall from a great height (3), being struck by a motor vehicle (1), and gunshot (1). Mean time from injury to corrective surgery was 16.9 years (range, 1-34 years). Before surgery, each patient had a thorough physical examination, with plain radiographs, including anteroposterior (AP), lateral, and oblique views, obtained to assess degree of limb malalignment. Patients completed the Short Form-36 (SF-36) and the Musculoskeletal Function Assessment (MFA) and underwent joint kinematics and gait analysis. Five malunions were located in the mid-diaphysis of the tibia, 3 in the proximal third, and 2 in the distal third of the tibial shaft. One patient had posttraumatic deformity involving the proximal and the mid-diaphysis (Table 1). After surgery, each patient was followed at regular intervals in the surgeon’s private office. Minimum follow-up was 7 months (mean, 11 months; range 7-17 months). At follow-up, radiographs were obtained, and each patient completed the SF-36 and the MFA and underwent joint kinematics and gait analysis.
We obtained preoperative AP and lateral radiographs of the malaligned and contralateral normal tibias for each patient. Angular deformity was determined in the sagittal and coronal planes to determine location and magnitude of the deformity. Specifically, on each AP and lateral radiograph, a line was drawn the length of the tibia proximal and distal to the area of the deformity. The angle generated by the intersection of these lines on the AP and lateral radiographs was then plotted on a grid to determine the precise plane and magnitude of the deformity (Table 2).1,12 Clinically, relevant rotational deformity of the involved limb was assessed by physical examination, and the results were compared with those of the contralateral limb. Owing to the lack of considerable rotational deformity in any of these 11 patients, we did not obtain computed tomography scans for further assessment of rotation.
Perioperative intravenous antibiotics were administered: 2 g cefazolin 30 minutes before incision and 1 g every 8 hours for 24 hours after surgery. A pneumatic tourniquet was placed on the proximal thigh, and the entire leg was prepared and draped in a sterile fashion. The limb was elevated and exsanguinated with an Esmark bandage and the tourniquet raised to 250 mm Hg. With fluoroscopy, the site of the tibial deformity was identified. Generally, an incision was made centered over the apex of the deformity and one fingerbreadth lateral to the palpable tibial crest. In most cases, the anterolateral aspect of the tibia was exposed while minimizing soft-tissue and periosteal stripping. The plane of the maximum deformity was identified with both direct visualization and fluoroscopy. The osteotomy was performed with an oscillating saw, and in each case a fibular osteotomy was also performed. Malalignment was corrected using a combination of manual manipulation and femoral distractor.25,26 Intraoperative biplanar radiographs were compared with our preoperative plan and with reversed images of the contralateral tibia to assess correction of the deformity. If lengthening was required, in addition to the tibial osteotomy, a corticotomy was created, and a circular external fixator applied and distraction osteogenesis performed.
We maintained the limbs in a short-leg splint for about 10 days after surgery and then initiated active-assisted range of motion of neighboring joints. Patients were maintained on toe-touch weight-bearing for the initial 6 weeks and were then advanced to partial weight-bearing (23 kg). Physical therapy for lower extremity strengthening and gait training was started 6 weeks after surgery. Three months after surgery, patients were advanced to weight-bearing as tolerated and were allowed to return to their activities of daily living without restrictions if radiographs and clinical examination were consistent with healing of the osteotomy.
Each patient was examined and radiographs obtained at regular intervals (2, 6, and 12 weeks and then about every 3 months) after surgery until healing. Bone union was determined by history and physical examination with pain-free weight-bearing without use of assistive devices and by return of functional use of the extremity. Radiographic union was considered to have occurred when bridging trabeculae were present across the osteotomy and there was no loosening or failure of the implants. Occasionally, if there were questions regarding healing, a musculoskeletal radiologist was consulted. Acceptable tibia alignment was defined as alignment of less than 5° varus or less than 10° valgus in the coronal plane and less than 15° procurvatum or recurvatum in the sagittal plane. Immediate postoperative radiographs and most recent radiographs were used to determine the final amount of angular correction.27
Two patients subsequently required secondary operative procedures. One had varus collapse through the regenerate, and the other developed a nonunion of the osteotomy site and required exchange intramedullary nailing. In each case, the final assessment was done after the patient had healed after the second surgery and had fully recovered.
Perceived Functional Assessment
The MFA is a 100-item self-administered QOL questionnaire designed to assess self-perception of physical, psychological, and social well-being in patients with a musculoskeletal injury or condition. The MFA provides a summary score and separate score for each of 10 functional domains. The lower the score, the better the patient’s perception of function. Validated and published norms are available.20,28-30
Perceived Health Status
The Short Form-36 is a 36-item multipurpose self-administered health survey questionnaire. The SF-36, which assesses overall health status, provides a Physical Component Score (PCS) and a Mental Component Score (MCS). The higher the score, the better the patient’s perception of function. Validated and published norms are available.31
Gait Analysis
Video data from a 6-camera high-resolution system (Motion Analysis, Santa Rosa, California) were used to assess gait. A set of 3 reflective surface markers was placed on each of 4 areas: trunk, thighs, legs, and feet.18,19 The patient walked barefoot along a 9-meter walkway, and video data were collected during the middle 2 meters. For each patient, data from 4 to 7 trials were collected. Computerized software produced data describing the averaged joint angle as a function of the gait cycle for each of the 3 principal planes of the body. Specific points in the gait cycle were analyzed. Variables included maximum knee varus in stance phase; maximum knee valgus in swing; maximum knee flexion in stance and swing; minimum knee flexion in stance; maximum ankle inversion in terminal stance; maximum ankle eversion in stance; maximum ankle dorsiflexion in stance and swing; and maximum ankle plantarflexion at takeoff. In addition to the lower extremity joint kinematics, angular measurements, basic gait measurements of step length, stride length, cadence, and speed were also recorded.
Statistical Analysis
Paired t tests were used to determine if significant changes occurred as a consequence of the surgery for the outcome variables (P < .05). Normative gait data were used to assess the quality of any changes that occurred in the variables, but no statistical analysis was performed to determine significant differences.18
Results
All 11 patients had clinical and radiographic evidence of healing and deformity correction at most recent follow-up. Nine patients (82%) healed after the index procedure. Mean total angular correction in the coronal plane was 21° (range, 14° varus to 7° valgus), and mean total angular correction in the sagittal plane was 9° (range, 21° recurvatum to 15° procurvatum) (Table 2).
For the group, mean preoperative MFA score was 39 (SD, 18; range, 10-69), and mean postoperative MFA score was 28 (SD, 14; range, 8-53). Patients reported the most improvement in 2 domains: In Leisure, mean (SD) preoperative score was 8 (2), and mean postoperative score was 5 (2); in Emotional, mean preoperative score was 5 (2), and mean postoperative score was 4 (1). The other domains were not significantly different between the 2 assessments.
On the SF-36, mean (SD) PCS significantly (P < .05) improved from 32 (8) to 43 (9). Mean (SD) MCS showed little change: preoperative, 46 (16); postoperative, 48 (13). The PCS subcategories that showed the most improvement were Physical Function, mean (SD) preoperative, 26 (20), to postoperative, 52 (26); Role of Physical Health, preoperative, 18 (24), to postoperative, 60 (41); and Bodily Pain, preoperative, 39 (27), to 58 (18).
The results from the preoperative and postoperative gait analysis showed no significant differences for the ankle, knee, and hip variables during swing phase (Table 3). In an analysis of the changes in joint kinematics during stance, maximum hip adduction (increased) and maximum knee varus (decreased) on the operative side were significantly improved toward normative values as a consequence of the surgery (Table 3). The other kinematic stance variables were not significantly different. No significant changes were observed in stance time, step length, stride length, cadence, or speed as a consequence of the surgery (Table 4).
Discussion
Correction of malaligned tibias leads to improved limb alignment and patients’ perceptions of functional abilities and health but had a limited effect on joint kinematics and gait. In a group of like patients, we used common techniques to realign malunited tibias and validated instruments to measure functional outcome, health status, joint kinematics, and gait. The goals of this study were to evaluate changes in perceived function and health status and changes in joint kinematics and gait as a result of correction of a posttraumatic limb deformity.
Other investigators have reported outcomes of treating symptomatic malunions,32 nonunions,24 and leg-length discrepancies.33 In these reports, correction of deformity improved patient satisfaction and function, though objective means of assessment were infrequently used. Good results were reported with use of a dome-shaped supramalleolar osteotomy for the correction of tibial malunion.32 In this study, supramalleolar osteotomy was performed on 8 patients for correction of a malunited tibia. Postoperative assessment included subjective assessment of pain, limp, appearance, instability, and activity. Of these 8 patients, 7 reported overall symptomatic improvement after healing, and the 1 who lost the deformity correction remained symptomatic. Significant improvement in overall health has been reported after successful treatment of tibia nonunions.24 The investigators used the SF-36 to assess patients who underwent treatment for a tibial nonunion. Analysis of these patients’ results showed a significant improvement in physical and mental functioning after healing. In addition, improved gait symmetry was reported in patients successfully treated for leg-length discrepancies.33 Unfortunately, how improvement in gait related to overall patient function was not assessed. In the present study, we used stringent objective and subjective validated instruments to assess changes in joint gait kinematics and functional outcome before and after treatment of a tibial malunion. In general, our results are consistent with published results and indicate that realignment of tibial malunions improves patients’ perceptions of function. Our results also indicate improvements toward normative values in maximal hip adduction and knee varus, thus confirming the efficacy of the surgery from a functional perspective. Unfortunately, we did not show significant improvements in the remaining joint kinematics measurements or temporal gait parameters.
It is not entirely clear whether tibial malalignment leads to degenerative changes of the ipsilateral knee and/or ankle and what role this might play in functioning. In a retrospective analysis of 92 patients, angular deformity within 15° of normal alignment did not lead to ankle arthrosis.9 Milner and colleagues4 found that, though varus malunion of the tibia may lead to arthrosis of the medial compartment of the knee, other factors were more important in causing arthrosis of the ankle.
Wu and colleagues34 used tibial osteotomies in New Zealand white rabbits to investigate cartilage and bone changes of the knee after creation of varus or valgus tibial deformities. Thirty-four weeks after osteotomy, rabbits with up to 30° of deformity had severe cartilage changes with osteophytes, fibrillation, derangement of cell columns, and associated increased subchondral bone density of the knees. Cadaveric studies have also shown increased contact pressures within the knees and ankles with ever increasing amounts of tibial deformity.6,10 In each cadaveric study, malalignment in the distal third of the tibia caused the largest changes in the ankle, and changes in the alignment in the proximal third caused the largest changes in the knee.
Consistent with these animal and cadaveric studies are several retrospective clinical studies that have correlated tibial malalignment (particularly varus) with development of knee and ankle arthrosis.3,5,8 Kettelkamp and colleagues3 found a direct correlation between magnitude of deformity and length of time with development of knee arthrosis. These findings have led many to recommend that surgeons try to restore tibial alignment to as near normal as possible to reduce the likelihood of arthrosis after tibia fracture. We found significant improvement toward normative values for maximum hip adduction (increased) and tibial varus (decreased) after surgery. These improvements would shift the weight-bearing forces back to the central part of the knee and therefore more uniformly distribute weight-bearing forces.
Posttraumatic arthrosis that develops after fracture is thought to result from increased joint pressures and possibly factors related to the injury. Although surgical correction of tibial alignment is unlikely to reverse these cartilage changes, it may restore joint pressure symmetry and “offload” compromised compartments. Offloading of already degenerative compartments may explain our patients’ improved perceptions of function and overall health status.
There were several limitations to our study. First, plain radiographs of malaligned and uninjured tibia and fibula were used, and these do not allow complete assessment of the weight-bearing access of the limb. Our patients, however, had isolated tibia fractures, which involved a normal limb before injury, so any alterations in joint kinematics, gait, or function would likely be the result of the fracture. Another limitation of our study is its nonrandomized design. However, the patients reflect the typical heterogeneous trauma patient population, who typically develop tibial malunions and seek correction. Another limitation was the lack of a treatment protocol regarding exact surgical technique and implants used to stabilize the osteotomies. In general, the patients were treated similarly, and their preoperative and postoperative assessments were exactly the same, as was their state-of-the-art joint kinematics and gait analysis, combined with the use of previously validated outcome measures. In addition, the lack of improvement in gait could have resulted from postoperative physical therapy that focused on joint mobilization and muscle strengthening and not on correction of abnormal gait parameters noted on preoperative gait analysis. Despite the potential limitations of the study, surgical correction of these symptomatic tibial malunions resulted in significant improvement in functional outcome and improved joint kinematics on the operative side.
Conclusion
Significant effort should be made to restore and maintain near-anatomical tibial alignment until a tibia fracture is healed. In patients who develop a symptomatic tibial malunion, surgical correction should be undertaken with the intent to restore normal limb alignment and improve joint kinematics, function, and overall health status.
1. Probe RA. Lower extremity angular malunion: evaluation and surgical correction. J Am Acad Orthop Surg. 2003;11(5):302-311.
2. van der Linden W, Larsson K. Plate fixation versus conservative treatment of tibial shaft fractures. A randomized trial. J Bone Joint Surg Am. 1979;61(6):873-878.
3. Kettelkamp DB, Hillberry BM, Murrish DE, Heck DA. Degenerative arthritis of the knee secondary to fracture malunion. Clin Orthop. 1988;(234):159-169.
4. Milner SA, Davis TR, Muir KR, Greenwood DC, Doherty M. Long-term outcome after tibial shaft fracture: is malunion important? J Bone Joint Surg Am. 2002;84(6):971-980.
5. Puno RM, Vaughan JJ, Stetten ML, Johnson JR. Long-term effects of tibial angular malunion on the knee and ankle joints. J Orthop Trauma. 1991;5(3):247-254.
6. Tarr RR, Resnick CT, Wagner KS, Sarmiento A. Changes in tibiotalar joint contact areas following experimentally induced tibial angular deformities. Clin Orthop. 1985;(199):72-80.
7. Ting AJ, Tarr RR, Sarmiento A, Wagner K, Resnick C. The role of subtalar motion and ankle contact pressure changes from angular deformities of the tibia. Foot Ankle. 1987;7(5):290-299.
8. van der Schoot DK, Den Outer AJ, Bode PJ, Obermann WR, van Vugt AB. Degenerative changes at the knee and ankle related to malunion of tibial fractures. 15-year follow-up of 88 patients. J Bone Joint Surg Br. 1996;78(5):722-725.
9. Kristensen KD, Kiaer T, Blicher J. No arthrosis of the ankle 20 years after malaligned tibial-shaft fracture. Acta Orthop Scand. 1989;60(2):208-209.
10. McKellop HA, Sigholm G, Redfern FC, Doyle B, Sarmiento A, Luck JV Sr. The effect of simulated fracture-angulations of the tibia on cartilage pressures in the knee joint. J Bone Joint Surg Am. 1991;73(9):1382-1391.
11. Merchant TC, Dietz FR. Long-term follow-up after fractures of the tibial and fibular shafts. J Bone Joint Surg Am. 1989;71(4):599-606.
12. Paley D, Herzenberg JE, Tetsworth K, McKie J, Bhave A. Deformity planning for frontal and sagittal plane corrective osteotomies. Orthop Clin North Am. 1994;25(3):425-465.
13. Perry J. Gait Analysis: Normal and Pathological Function. Thorofare, NJ: Slack; 1992.
14. Puno RM, Vaughan JJ, von Fraunhofer JA, Stetten ML, Johnson JR. A method of determining the angular malalignments of the knee and ankle joints resulting from a tibial malunion. Clin Orthop. 1987;(223):213-219.
15. Greenwood DC, Muir KR, Doherty M, Milner SA, Stevens M, Davis TR. Conservatively managed tibial shaft fractures in Nottingham, UK: are pain, osteoarthritis, and disability long-term complications? J Epidemiol Community Health. 1997;51(6):701-704.
16. Dehne E, Deffer PA, Hall RM, Brown PW, Johnson EV. The natural history of the fractured tibia. Surg Clin North Am. 1961;41(6):1495-1513.
17. Kitaoka HB, Schaap EJ, Chao EY, An KN. Displaced intra-articular fractures of the calcaneus treated non-operatively. Clinical results and analysis of motion and ground-reaction and temporal forces. J Bone Joint Surg Am. 1994;76(10):1531-1540.
18. Borrelli J Jr, Goldfarb C, Ricci W, Wagner JM, Engsberg JR. Functional outcome after isolated acetabular fractures. J Orthop Trauma. 2002;16(2):73-81.
19. Borrelli J Jr, Ricci WM, Anglen JO, Gregush R, Engsberg J. Muscle strength recovery and its effects on outcome after open reduction and internal fixation of acetabular fractures. J Orthop Trauma. 2006;20(6):388-395.
20. Jaglal S, Lakhani Z, Schatzker J. Reliability, validity, and responsiveness of the lower extremity measure for patients with a hip fracture. J Bone Joint Surg Am. 2000;82(7):955-962.
21. Madsen MS, Ritter MA, Morris HH, et al. The effect of total hip arthroplasty surgical approach on gait. J Orthop Res. 2004;22(1):44-50.
22. Mittlmeier T, Morlock MM, Hertlein H, et al. Analysis of morphology and gait function after intraarticular calcaneal fracture. J Orthop Trauma. 1993;7(4):303-310.
23. Song KM, Halliday SE, Little DG. The effect of limb-length discrepancy on gait. J Bone Joint Surg Am. 1997;79(11):1690-1698.
24. Zlowodzki M, Obremskey WT, Thomison JB, Kregor PJ. Functional outcome after treatment of lower-extremity nonunions. J Trauma. 2005;58(2):312-317.
25. Sanders R, Anglen JO, Mark JB. Oblique osteotomy for the correction of tibial malunion. J Bone Joint Surg Am. 1995;77(2):240-246.
26. Sangeorzan BJ, Sangeorzan BP, Hansen ST Jr, Judd RP. Mathematically directed single-cut osteotomy for correction of tibial malunion. J Orthop Trauma. 1989;3(4):267-275.
27. Borrelli J Jr, Leduc S, Gregush R, Ricci WM. Tricortical bone grafts for treatment of malaligned tibias and fibulas. Clin Orthop. 2009;467(4):1056-1063.
28. Engelberg R, Martin DP, Agel J, Obremsky W, Coronado G, Swiontkowski MF. Musculoskeletal Function Assessment instrument: criterion and construct validity. J Orthop Res. 1996;14(2):182-192.
29. Engelberg R, Martin DP, Agel J, Swiontkowski MF. Musculoskeletal Function Assessment: reference values for patient and non-patient samples. J Orthop Res. 1999;17(1):101-109.
30. Swiontkowski MF, Engelberg R, Martin DP, Agel J. Short Musculoskeletal Function Assessment questionnaire: validity, reliability, and responsiveness. J Bone Joint Surg Am. 1999;81(9):1245-1260.
31. Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30(6):473-483.
32. Graehl PM, Hersh MR, Heckman JD. Supramalleolar osteotomy for the treatment of symptomatic tibial malunion. J Orthop Trauma. 1987;1(4):281-292.
33. Bhave A, Paley D, Herzenberg JE. Improvement in gait parameters after lengthening for the treatment of limb-length discrepancy. J Bone Joint Surg Am. 1999;81(4):529-534.
34. Wu DD, Burr DB, Boyd RD, Radin EL. Bone and cartilage changes following experimental varus or valgus tibial angulation. J Orthop Res. 1990;8(4):572-585.
The tibia is the most commonly fractured long bone in adults, and tibial malunions occur in up to 60% of these patients.1,2 Persistent tibial malalignment, particularly varus alignment, negatively alters gait and joint kinematics, leading to altered weight-bearing forces across the knee and ankle joints. These altered forces may lead to osteoarthritis.3-8
Several studies have identified a relationship between extent of tibial malalignment and changes in joint reaction forces.3,5-7,9-13 Puno and colleagues14 developed a mathematical model to better define the changes in neighboring joints relative to the pattern of the tibia malalignment. Not surprisingly, their work showed that, with distal tibial malunions, altered stress concentrations were realized at the ankle joint, and more proximal tibial deformities led to larger alterations in the joint stresses at the knee. More recently, van der Schoot and colleagues8 found a high prevalence of ipsilateral ankle osteoarthritis with tibial malalignment of 5° or more, and Greenwood and colleagues15 showed a higher incidence of knee pain, lower limb osteoarthritis, and disability in patients with previous tibia fractures. Given these findings, it would seem that correction of tibial malalignment would lead to normative lower extremity joint kinematic values, joint reaction forces, and overall quality of life (QOL).
The ability to ambulate has been recognized as an important milestone in functional recovery after lower extremity injury.2,16,17 Gait analysis, assessment of joint kinematics, and QOL and health status questionnaires can provide information to evaluate rehabilitation protocols, treatment algorithms, and surgical outcomes. Recently, these measures have been used to assess patients recovering from acetabular fractures, femoral shaft fractures, and calcaneal fractures.4,11,17-24 However, no study has used these measures to assess the benefits of surgical correction of malaligned tibias.
We conducted a study to determine improvement in gait, joint kinematics, and patients’ perceptions of overall well-being after surgical correction of tibial malunions. The null hypothesis was that correction of tibial malunion would have no effect on gait, joint kinematics, or patients’ perceptions of function and QOL.
Materials and Methods
This prospective double-time-point study, which was approved by the Institutional Review Board of Washington University/Barnes-Jewish Hospital, evaluated 11 consecutive patients with a varus tibial malunion treated by a single surgeon between September 2003 and January 2006. All patients were treated using a technique that included oblique osteotomy and open reduction and internal fixation (ORIF) or osteotomy and intramedullary nailing. Study inclusion criteria were age 18 years or older; symptomatic varus malunion of the tibia of 10º or more; absence of a developmental or pathologic process leading to the fracture and subsequent deformity; no neurologic deficit of either lower extremity or contralateral lower extremity deformity; and ability to ambulate 9 meters with or without use of an assistive device.
The 11 patients (6 men, 5 women) who met these criteria enrolled in the study. Mean age was 53 years (range, 43-68 years). Eight malunions involved the left tibia. The mechanisms of injury were motor vehicle crash (6 patients), fall from a great height (3), being struck by a motor vehicle (1), and gunshot (1). Mean time from injury to corrective surgery was 16.9 years (range, 1-34 years). Before surgery, each patient had a thorough physical examination, with plain radiographs, including anteroposterior (AP), lateral, and oblique views, obtained to assess degree of limb malalignment. Patients completed the Short Form-36 (SF-36) and the Musculoskeletal Function Assessment (MFA) and underwent joint kinematics and gait analysis. Five malunions were located in the mid-diaphysis of the tibia, 3 in the proximal third, and 2 in the distal third of the tibial shaft. One patient had posttraumatic deformity involving the proximal and the mid-diaphysis (Table 1). After surgery, each patient was followed at regular intervals in the surgeon’s private office. Minimum follow-up was 7 months (mean, 11 months; range 7-17 months). At follow-up, radiographs were obtained, and each patient completed the SF-36 and the MFA and underwent joint kinematics and gait analysis.
We obtained preoperative AP and lateral radiographs of the malaligned and contralateral normal tibias for each patient. Angular deformity was determined in the sagittal and coronal planes to determine location and magnitude of the deformity. Specifically, on each AP and lateral radiograph, a line was drawn the length of the tibia proximal and distal to the area of the deformity. The angle generated by the intersection of these lines on the AP and lateral radiographs was then plotted on a grid to determine the precise plane and magnitude of the deformity (Table 2).1,12 Clinically, relevant rotational deformity of the involved limb was assessed by physical examination, and the results were compared with those of the contralateral limb. Owing to the lack of considerable rotational deformity in any of these 11 patients, we did not obtain computed tomography scans for further assessment of rotation.
Perioperative intravenous antibiotics were administered: 2 g cefazolin 30 minutes before incision and 1 g every 8 hours for 24 hours after surgery. A pneumatic tourniquet was placed on the proximal thigh, and the entire leg was prepared and draped in a sterile fashion. The limb was elevated and exsanguinated with an Esmark bandage and the tourniquet raised to 250 mm Hg. With fluoroscopy, the site of the tibial deformity was identified. Generally, an incision was made centered over the apex of the deformity and one fingerbreadth lateral to the palpable tibial crest. In most cases, the anterolateral aspect of the tibia was exposed while minimizing soft-tissue and periosteal stripping. The plane of the maximum deformity was identified with both direct visualization and fluoroscopy. The osteotomy was performed with an oscillating saw, and in each case a fibular osteotomy was also performed. Malalignment was corrected using a combination of manual manipulation and femoral distractor.25,26 Intraoperative biplanar radiographs were compared with our preoperative plan and with reversed images of the contralateral tibia to assess correction of the deformity. If lengthening was required, in addition to the tibial osteotomy, a corticotomy was created, and a circular external fixator applied and distraction osteogenesis performed.
We maintained the limbs in a short-leg splint for about 10 days after surgery and then initiated active-assisted range of motion of neighboring joints. Patients were maintained on toe-touch weight-bearing for the initial 6 weeks and were then advanced to partial weight-bearing (23 kg). Physical therapy for lower extremity strengthening and gait training was started 6 weeks after surgery. Three months after surgery, patients were advanced to weight-bearing as tolerated and were allowed to return to their activities of daily living without restrictions if radiographs and clinical examination were consistent with healing of the osteotomy.
Each patient was examined and radiographs obtained at regular intervals (2, 6, and 12 weeks and then about every 3 months) after surgery until healing. Bone union was determined by history and physical examination with pain-free weight-bearing without use of assistive devices and by return of functional use of the extremity. Radiographic union was considered to have occurred when bridging trabeculae were present across the osteotomy and there was no loosening or failure of the implants. Occasionally, if there were questions regarding healing, a musculoskeletal radiologist was consulted. Acceptable tibia alignment was defined as alignment of less than 5° varus or less than 10° valgus in the coronal plane and less than 15° procurvatum or recurvatum in the sagittal plane. Immediate postoperative radiographs and most recent radiographs were used to determine the final amount of angular correction.27
Two patients subsequently required secondary operative procedures. One had varus collapse through the regenerate, and the other developed a nonunion of the osteotomy site and required exchange intramedullary nailing. In each case, the final assessment was done after the patient had healed after the second surgery and had fully recovered.
Perceived Functional Assessment
The MFA is a 100-item self-administered QOL questionnaire designed to assess self-perception of physical, psychological, and social well-being in patients with a musculoskeletal injury or condition. The MFA provides a summary score and separate score for each of 10 functional domains. The lower the score, the better the patient’s perception of function. Validated and published norms are available.20,28-30
Perceived Health Status
The Short Form-36 is a 36-item multipurpose self-administered health survey questionnaire. The SF-36, which assesses overall health status, provides a Physical Component Score (PCS) and a Mental Component Score (MCS). The higher the score, the better the patient’s perception of function. Validated and published norms are available.31
Gait Analysis
Video data from a 6-camera high-resolution system (Motion Analysis, Santa Rosa, California) were used to assess gait. A set of 3 reflective surface markers was placed on each of 4 areas: trunk, thighs, legs, and feet.18,19 The patient walked barefoot along a 9-meter walkway, and video data were collected during the middle 2 meters. For each patient, data from 4 to 7 trials were collected. Computerized software produced data describing the averaged joint angle as a function of the gait cycle for each of the 3 principal planes of the body. Specific points in the gait cycle were analyzed. Variables included maximum knee varus in stance phase; maximum knee valgus in swing; maximum knee flexion in stance and swing; minimum knee flexion in stance; maximum ankle inversion in terminal stance; maximum ankle eversion in stance; maximum ankle dorsiflexion in stance and swing; and maximum ankle plantarflexion at takeoff. In addition to the lower extremity joint kinematics, angular measurements, basic gait measurements of step length, stride length, cadence, and speed were also recorded.
Statistical Analysis
Paired t tests were used to determine if significant changes occurred as a consequence of the surgery for the outcome variables (P < .05). Normative gait data were used to assess the quality of any changes that occurred in the variables, but no statistical analysis was performed to determine significant differences.18
Results
All 11 patients had clinical and radiographic evidence of healing and deformity correction at most recent follow-up. Nine patients (82%) healed after the index procedure. Mean total angular correction in the coronal plane was 21° (range, 14° varus to 7° valgus), and mean total angular correction in the sagittal plane was 9° (range, 21° recurvatum to 15° procurvatum) (Table 2).
For the group, mean preoperative MFA score was 39 (SD, 18; range, 10-69), and mean postoperative MFA score was 28 (SD, 14; range, 8-53). Patients reported the most improvement in 2 domains: In Leisure, mean (SD) preoperative score was 8 (2), and mean postoperative score was 5 (2); in Emotional, mean preoperative score was 5 (2), and mean postoperative score was 4 (1). The other domains were not significantly different between the 2 assessments.
On the SF-36, mean (SD) PCS significantly (P < .05) improved from 32 (8) to 43 (9). Mean (SD) MCS showed little change: preoperative, 46 (16); postoperative, 48 (13). The PCS subcategories that showed the most improvement were Physical Function, mean (SD) preoperative, 26 (20), to postoperative, 52 (26); Role of Physical Health, preoperative, 18 (24), to postoperative, 60 (41); and Bodily Pain, preoperative, 39 (27), to 58 (18).
The results from the preoperative and postoperative gait analysis showed no significant differences for the ankle, knee, and hip variables during swing phase (Table 3). In an analysis of the changes in joint kinematics during stance, maximum hip adduction (increased) and maximum knee varus (decreased) on the operative side were significantly improved toward normative values as a consequence of the surgery (Table 3). The other kinematic stance variables were not significantly different. No significant changes were observed in stance time, step length, stride length, cadence, or speed as a consequence of the surgery (Table 4).
Discussion
Correction of malaligned tibias leads to improved limb alignment and patients’ perceptions of functional abilities and health but had a limited effect on joint kinematics and gait. In a group of like patients, we used common techniques to realign malunited tibias and validated instruments to measure functional outcome, health status, joint kinematics, and gait. The goals of this study were to evaluate changes in perceived function and health status and changes in joint kinematics and gait as a result of correction of a posttraumatic limb deformity.
Other investigators have reported outcomes of treating symptomatic malunions,32 nonunions,24 and leg-length discrepancies.33 In these reports, correction of deformity improved patient satisfaction and function, though objective means of assessment were infrequently used. Good results were reported with use of a dome-shaped supramalleolar osteotomy for the correction of tibial malunion.32 In this study, supramalleolar osteotomy was performed on 8 patients for correction of a malunited tibia. Postoperative assessment included subjective assessment of pain, limp, appearance, instability, and activity. Of these 8 patients, 7 reported overall symptomatic improvement after healing, and the 1 who lost the deformity correction remained symptomatic. Significant improvement in overall health has been reported after successful treatment of tibia nonunions.24 The investigators used the SF-36 to assess patients who underwent treatment for a tibial nonunion. Analysis of these patients’ results showed a significant improvement in physical and mental functioning after healing. In addition, improved gait symmetry was reported in patients successfully treated for leg-length discrepancies.33 Unfortunately, how improvement in gait related to overall patient function was not assessed. In the present study, we used stringent objective and subjective validated instruments to assess changes in joint gait kinematics and functional outcome before and after treatment of a tibial malunion. In general, our results are consistent with published results and indicate that realignment of tibial malunions improves patients’ perceptions of function. Our results also indicate improvements toward normative values in maximal hip adduction and knee varus, thus confirming the efficacy of the surgery from a functional perspective. Unfortunately, we did not show significant improvements in the remaining joint kinematics measurements or temporal gait parameters.
It is not entirely clear whether tibial malalignment leads to degenerative changes of the ipsilateral knee and/or ankle and what role this might play in functioning. In a retrospective analysis of 92 patients, angular deformity within 15° of normal alignment did not lead to ankle arthrosis.9 Milner and colleagues4 found that, though varus malunion of the tibia may lead to arthrosis of the medial compartment of the knee, other factors were more important in causing arthrosis of the ankle.
Wu and colleagues34 used tibial osteotomies in New Zealand white rabbits to investigate cartilage and bone changes of the knee after creation of varus or valgus tibial deformities. Thirty-four weeks after osteotomy, rabbits with up to 30° of deformity had severe cartilage changes with osteophytes, fibrillation, derangement of cell columns, and associated increased subchondral bone density of the knees. Cadaveric studies have also shown increased contact pressures within the knees and ankles with ever increasing amounts of tibial deformity.6,10 In each cadaveric study, malalignment in the distal third of the tibia caused the largest changes in the ankle, and changes in the alignment in the proximal third caused the largest changes in the knee.
Consistent with these animal and cadaveric studies are several retrospective clinical studies that have correlated tibial malalignment (particularly varus) with development of knee and ankle arthrosis.3,5,8 Kettelkamp and colleagues3 found a direct correlation between magnitude of deformity and length of time with development of knee arthrosis. These findings have led many to recommend that surgeons try to restore tibial alignment to as near normal as possible to reduce the likelihood of arthrosis after tibia fracture. We found significant improvement toward normative values for maximum hip adduction (increased) and tibial varus (decreased) after surgery. These improvements would shift the weight-bearing forces back to the central part of the knee and therefore more uniformly distribute weight-bearing forces.
Posttraumatic arthrosis that develops after fracture is thought to result from increased joint pressures and possibly factors related to the injury. Although surgical correction of tibial alignment is unlikely to reverse these cartilage changes, it may restore joint pressure symmetry and “offload” compromised compartments. Offloading of already degenerative compartments may explain our patients’ improved perceptions of function and overall health status.
There were several limitations to our study. First, plain radiographs of malaligned and uninjured tibia and fibula were used, and these do not allow complete assessment of the weight-bearing access of the limb. Our patients, however, had isolated tibia fractures, which involved a normal limb before injury, so any alterations in joint kinematics, gait, or function would likely be the result of the fracture. Another limitation of our study is its nonrandomized design. However, the patients reflect the typical heterogeneous trauma patient population, who typically develop tibial malunions and seek correction. Another limitation was the lack of a treatment protocol regarding exact surgical technique and implants used to stabilize the osteotomies. In general, the patients were treated similarly, and their preoperative and postoperative assessments were exactly the same, as was their state-of-the-art joint kinematics and gait analysis, combined with the use of previously validated outcome measures. In addition, the lack of improvement in gait could have resulted from postoperative physical therapy that focused on joint mobilization and muscle strengthening and not on correction of abnormal gait parameters noted on preoperative gait analysis. Despite the potential limitations of the study, surgical correction of these symptomatic tibial malunions resulted in significant improvement in functional outcome and improved joint kinematics on the operative side.
Conclusion
Significant effort should be made to restore and maintain near-anatomical tibial alignment until a tibia fracture is healed. In patients who develop a symptomatic tibial malunion, surgical correction should be undertaken with the intent to restore normal limb alignment and improve joint kinematics, function, and overall health status.
The tibia is the most commonly fractured long bone in adults, and tibial malunions occur in up to 60% of these patients.1,2 Persistent tibial malalignment, particularly varus alignment, negatively alters gait and joint kinematics, leading to altered weight-bearing forces across the knee and ankle joints. These altered forces may lead to osteoarthritis.3-8
Several studies have identified a relationship between extent of tibial malalignment and changes in joint reaction forces.3,5-7,9-13 Puno and colleagues14 developed a mathematical model to better define the changes in neighboring joints relative to the pattern of the tibia malalignment. Not surprisingly, their work showed that, with distal tibial malunions, altered stress concentrations were realized at the ankle joint, and more proximal tibial deformities led to larger alterations in the joint stresses at the knee. More recently, van der Schoot and colleagues8 found a high prevalence of ipsilateral ankle osteoarthritis with tibial malalignment of 5° or more, and Greenwood and colleagues15 showed a higher incidence of knee pain, lower limb osteoarthritis, and disability in patients with previous tibia fractures. Given these findings, it would seem that correction of tibial malalignment would lead to normative lower extremity joint kinematic values, joint reaction forces, and overall quality of life (QOL).
The ability to ambulate has been recognized as an important milestone in functional recovery after lower extremity injury.2,16,17 Gait analysis, assessment of joint kinematics, and QOL and health status questionnaires can provide information to evaluate rehabilitation protocols, treatment algorithms, and surgical outcomes. Recently, these measures have been used to assess patients recovering from acetabular fractures, femoral shaft fractures, and calcaneal fractures.4,11,17-24 However, no study has used these measures to assess the benefits of surgical correction of malaligned tibias.
We conducted a study to determine improvement in gait, joint kinematics, and patients’ perceptions of overall well-being after surgical correction of tibial malunions. The null hypothesis was that correction of tibial malunion would have no effect on gait, joint kinematics, or patients’ perceptions of function and QOL.
Materials and Methods
This prospective double-time-point study, which was approved by the Institutional Review Board of Washington University/Barnes-Jewish Hospital, evaluated 11 consecutive patients with a varus tibial malunion treated by a single surgeon between September 2003 and January 2006. All patients were treated using a technique that included oblique osteotomy and open reduction and internal fixation (ORIF) or osteotomy and intramedullary nailing. Study inclusion criteria were age 18 years or older; symptomatic varus malunion of the tibia of 10º or more; absence of a developmental or pathologic process leading to the fracture and subsequent deformity; no neurologic deficit of either lower extremity or contralateral lower extremity deformity; and ability to ambulate 9 meters with or without use of an assistive device.
The 11 patients (6 men, 5 women) who met these criteria enrolled in the study. Mean age was 53 years (range, 43-68 years). Eight malunions involved the left tibia. The mechanisms of injury were motor vehicle crash (6 patients), fall from a great height (3), being struck by a motor vehicle (1), and gunshot (1). Mean time from injury to corrective surgery was 16.9 years (range, 1-34 years). Before surgery, each patient had a thorough physical examination, with plain radiographs, including anteroposterior (AP), lateral, and oblique views, obtained to assess degree of limb malalignment. Patients completed the Short Form-36 (SF-36) and the Musculoskeletal Function Assessment (MFA) and underwent joint kinematics and gait analysis. Five malunions were located in the mid-diaphysis of the tibia, 3 in the proximal third, and 2 in the distal third of the tibial shaft. One patient had posttraumatic deformity involving the proximal and the mid-diaphysis (Table 1). After surgery, each patient was followed at regular intervals in the surgeon’s private office. Minimum follow-up was 7 months (mean, 11 months; range 7-17 months). At follow-up, radiographs were obtained, and each patient completed the SF-36 and the MFA and underwent joint kinematics and gait analysis.
We obtained preoperative AP and lateral radiographs of the malaligned and contralateral normal tibias for each patient. Angular deformity was determined in the sagittal and coronal planes to determine location and magnitude of the deformity. Specifically, on each AP and lateral radiograph, a line was drawn the length of the tibia proximal and distal to the area of the deformity. The angle generated by the intersection of these lines on the AP and lateral radiographs was then plotted on a grid to determine the precise plane and magnitude of the deformity (Table 2).1,12 Clinically, relevant rotational deformity of the involved limb was assessed by physical examination, and the results were compared with those of the contralateral limb. Owing to the lack of considerable rotational deformity in any of these 11 patients, we did not obtain computed tomography scans for further assessment of rotation.
Perioperative intravenous antibiotics were administered: 2 g cefazolin 30 minutes before incision and 1 g every 8 hours for 24 hours after surgery. A pneumatic tourniquet was placed on the proximal thigh, and the entire leg was prepared and draped in a sterile fashion. The limb was elevated and exsanguinated with an Esmark bandage and the tourniquet raised to 250 mm Hg. With fluoroscopy, the site of the tibial deformity was identified. Generally, an incision was made centered over the apex of the deformity and one fingerbreadth lateral to the palpable tibial crest. In most cases, the anterolateral aspect of the tibia was exposed while minimizing soft-tissue and periosteal stripping. The plane of the maximum deformity was identified with both direct visualization and fluoroscopy. The osteotomy was performed with an oscillating saw, and in each case a fibular osteotomy was also performed. Malalignment was corrected using a combination of manual manipulation and femoral distractor.25,26 Intraoperative biplanar radiographs were compared with our preoperative plan and with reversed images of the contralateral tibia to assess correction of the deformity. If lengthening was required, in addition to the tibial osteotomy, a corticotomy was created, and a circular external fixator applied and distraction osteogenesis performed.
We maintained the limbs in a short-leg splint for about 10 days after surgery and then initiated active-assisted range of motion of neighboring joints. Patients were maintained on toe-touch weight-bearing for the initial 6 weeks and were then advanced to partial weight-bearing (23 kg). Physical therapy for lower extremity strengthening and gait training was started 6 weeks after surgery. Three months after surgery, patients were advanced to weight-bearing as tolerated and were allowed to return to their activities of daily living without restrictions if radiographs and clinical examination were consistent with healing of the osteotomy.
Each patient was examined and radiographs obtained at regular intervals (2, 6, and 12 weeks and then about every 3 months) after surgery until healing. Bone union was determined by history and physical examination with pain-free weight-bearing without use of assistive devices and by return of functional use of the extremity. Radiographic union was considered to have occurred when bridging trabeculae were present across the osteotomy and there was no loosening or failure of the implants. Occasionally, if there were questions regarding healing, a musculoskeletal radiologist was consulted. Acceptable tibia alignment was defined as alignment of less than 5° varus or less than 10° valgus in the coronal plane and less than 15° procurvatum or recurvatum in the sagittal plane. Immediate postoperative radiographs and most recent radiographs were used to determine the final amount of angular correction.27
Two patients subsequently required secondary operative procedures. One had varus collapse through the regenerate, and the other developed a nonunion of the osteotomy site and required exchange intramedullary nailing. In each case, the final assessment was done after the patient had healed after the second surgery and had fully recovered.
Perceived Functional Assessment
The MFA is a 100-item self-administered QOL questionnaire designed to assess self-perception of physical, psychological, and social well-being in patients with a musculoskeletal injury or condition. The MFA provides a summary score and separate score for each of 10 functional domains. The lower the score, the better the patient’s perception of function. Validated and published norms are available.20,28-30
Perceived Health Status
The Short Form-36 is a 36-item multipurpose self-administered health survey questionnaire. The SF-36, which assesses overall health status, provides a Physical Component Score (PCS) and a Mental Component Score (MCS). The higher the score, the better the patient’s perception of function. Validated and published norms are available.31
Gait Analysis
Video data from a 6-camera high-resolution system (Motion Analysis, Santa Rosa, California) were used to assess gait. A set of 3 reflective surface markers was placed on each of 4 areas: trunk, thighs, legs, and feet.18,19 The patient walked barefoot along a 9-meter walkway, and video data were collected during the middle 2 meters. For each patient, data from 4 to 7 trials were collected. Computerized software produced data describing the averaged joint angle as a function of the gait cycle for each of the 3 principal planes of the body. Specific points in the gait cycle were analyzed. Variables included maximum knee varus in stance phase; maximum knee valgus in swing; maximum knee flexion in stance and swing; minimum knee flexion in stance; maximum ankle inversion in terminal stance; maximum ankle eversion in stance; maximum ankle dorsiflexion in stance and swing; and maximum ankle plantarflexion at takeoff. In addition to the lower extremity joint kinematics, angular measurements, basic gait measurements of step length, stride length, cadence, and speed were also recorded.
Statistical Analysis
Paired t tests were used to determine if significant changes occurred as a consequence of the surgery for the outcome variables (P < .05). Normative gait data were used to assess the quality of any changes that occurred in the variables, but no statistical analysis was performed to determine significant differences.18
Results
All 11 patients had clinical and radiographic evidence of healing and deformity correction at most recent follow-up. Nine patients (82%) healed after the index procedure. Mean total angular correction in the coronal plane was 21° (range, 14° varus to 7° valgus), and mean total angular correction in the sagittal plane was 9° (range, 21° recurvatum to 15° procurvatum) (Table 2).
For the group, mean preoperative MFA score was 39 (SD, 18; range, 10-69), and mean postoperative MFA score was 28 (SD, 14; range, 8-53). Patients reported the most improvement in 2 domains: In Leisure, mean (SD) preoperative score was 8 (2), and mean postoperative score was 5 (2); in Emotional, mean preoperative score was 5 (2), and mean postoperative score was 4 (1). The other domains were not significantly different between the 2 assessments.
On the SF-36, mean (SD) PCS significantly (P < .05) improved from 32 (8) to 43 (9). Mean (SD) MCS showed little change: preoperative, 46 (16); postoperative, 48 (13). The PCS subcategories that showed the most improvement were Physical Function, mean (SD) preoperative, 26 (20), to postoperative, 52 (26); Role of Physical Health, preoperative, 18 (24), to postoperative, 60 (41); and Bodily Pain, preoperative, 39 (27), to 58 (18).
The results from the preoperative and postoperative gait analysis showed no significant differences for the ankle, knee, and hip variables during swing phase (Table 3). In an analysis of the changes in joint kinematics during stance, maximum hip adduction (increased) and maximum knee varus (decreased) on the operative side were significantly improved toward normative values as a consequence of the surgery (Table 3). The other kinematic stance variables were not significantly different. No significant changes were observed in stance time, step length, stride length, cadence, or speed as a consequence of the surgery (Table 4).
Discussion
Correction of malaligned tibias leads to improved limb alignment and patients’ perceptions of functional abilities and health but had a limited effect on joint kinematics and gait. In a group of like patients, we used common techniques to realign malunited tibias and validated instruments to measure functional outcome, health status, joint kinematics, and gait. The goals of this study were to evaluate changes in perceived function and health status and changes in joint kinematics and gait as a result of correction of a posttraumatic limb deformity.
Other investigators have reported outcomes of treating symptomatic malunions,32 nonunions,24 and leg-length discrepancies.33 In these reports, correction of deformity improved patient satisfaction and function, though objective means of assessment were infrequently used. Good results were reported with use of a dome-shaped supramalleolar osteotomy for the correction of tibial malunion.32 In this study, supramalleolar osteotomy was performed on 8 patients for correction of a malunited tibia. Postoperative assessment included subjective assessment of pain, limp, appearance, instability, and activity. Of these 8 patients, 7 reported overall symptomatic improvement after healing, and the 1 who lost the deformity correction remained symptomatic. Significant improvement in overall health has been reported after successful treatment of tibia nonunions.24 The investigators used the SF-36 to assess patients who underwent treatment for a tibial nonunion. Analysis of these patients’ results showed a significant improvement in physical and mental functioning after healing. In addition, improved gait symmetry was reported in patients successfully treated for leg-length discrepancies.33 Unfortunately, how improvement in gait related to overall patient function was not assessed. In the present study, we used stringent objective and subjective validated instruments to assess changes in joint gait kinematics and functional outcome before and after treatment of a tibial malunion. In general, our results are consistent with published results and indicate that realignment of tibial malunions improves patients’ perceptions of function. Our results also indicate improvements toward normative values in maximal hip adduction and knee varus, thus confirming the efficacy of the surgery from a functional perspective. Unfortunately, we did not show significant improvements in the remaining joint kinematics measurements or temporal gait parameters.
It is not entirely clear whether tibial malalignment leads to degenerative changes of the ipsilateral knee and/or ankle and what role this might play in functioning. In a retrospective analysis of 92 patients, angular deformity within 15° of normal alignment did not lead to ankle arthrosis.9 Milner and colleagues4 found that, though varus malunion of the tibia may lead to arthrosis of the medial compartment of the knee, other factors were more important in causing arthrosis of the ankle.
Wu and colleagues34 used tibial osteotomies in New Zealand white rabbits to investigate cartilage and bone changes of the knee after creation of varus or valgus tibial deformities. Thirty-four weeks after osteotomy, rabbits with up to 30° of deformity had severe cartilage changes with osteophytes, fibrillation, derangement of cell columns, and associated increased subchondral bone density of the knees. Cadaveric studies have also shown increased contact pressures within the knees and ankles with ever increasing amounts of tibial deformity.6,10 In each cadaveric study, malalignment in the distal third of the tibia caused the largest changes in the ankle, and changes in the alignment in the proximal third caused the largest changes in the knee.
Consistent with these animal and cadaveric studies are several retrospective clinical studies that have correlated tibial malalignment (particularly varus) with development of knee and ankle arthrosis.3,5,8 Kettelkamp and colleagues3 found a direct correlation between magnitude of deformity and length of time with development of knee arthrosis. These findings have led many to recommend that surgeons try to restore tibial alignment to as near normal as possible to reduce the likelihood of arthrosis after tibia fracture. We found significant improvement toward normative values for maximum hip adduction (increased) and tibial varus (decreased) after surgery. These improvements would shift the weight-bearing forces back to the central part of the knee and therefore more uniformly distribute weight-bearing forces.
Posttraumatic arthrosis that develops after fracture is thought to result from increased joint pressures and possibly factors related to the injury. Although surgical correction of tibial alignment is unlikely to reverse these cartilage changes, it may restore joint pressure symmetry and “offload” compromised compartments. Offloading of already degenerative compartments may explain our patients’ improved perceptions of function and overall health status.
There were several limitations to our study. First, plain radiographs of malaligned and uninjured tibia and fibula were used, and these do not allow complete assessment of the weight-bearing access of the limb. Our patients, however, had isolated tibia fractures, which involved a normal limb before injury, so any alterations in joint kinematics, gait, or function would likely be the result of the fracture. Another limitation of our study is its nonrandomized design. However, the patients reflect the typical heterogeneous trauma patient population, who typically develop tibial malunions and seek correction. Another limitation was the lack of a treatment protocol regarding exact surgical technique and implants used to stabilize the osteotomies. In general, the patients were treated similarly, and their preoperative and postoperative assessments were exactly the same, as was their state-of-the-art joint kinematics and gait analysis, combined with the use of previously validated outcome measures. In addition, the lack of improvement in gait could have resulted from postoperative physical therapy that focused on joint mobilization and muscle strengthening and not on correction of abnormal gait parameters noted on preoperative gait analysis. Despite the potential limitations of the study, surgical correction of these symptomatic tibial malunions resulted in significant improvement in functional outcome and improved joint kinematics on the operative side.
Conclusion
Significant effort should be made to restore and maintain near-anatomical tibial alignment until a tibia fracture is healed. In patients who develop a symptomatic tibial malunion, surgical correction should be undertaken with the intent to restore normal limb alignment and improve joint kinematics, function, and overall health status.
1. Probe RA. Lower extremity angular malunion: evaluation and surgical correction. J Am Acad Orthop Surg. 2003;11(5):302-311.
2. van der Linden W, Larsson K. Plate fixation versus conservative treatment of tibial shaft fractures. A randomized trial. J Bone Joint Surg Am. 1979;61(6):873-878.
3. Kettelkamp DB, Hillberry BM, Murrish DE, Heck DA. Degenerative arthritis of the knee secondary to fracture malunion. Clin Orthop. 1988;(234):159-169.
4. Milner SA, Davis TR, Muir KR, Greenwood DC, Doherty M. Long-term outcome after tibial shaft fracture: is malunion important? J Bone Joint Surg Am. 2002;84(6):971-980.
5. Puno RM, Vaughan JJ, Stetten ML, Johnson JR. Long-term effects of tibial angular malunion on the knee and ankle joints. J Orthop Trauma. 1991;5(3):247-254.
6. Tarr RR, Resnick CT, Wagner KS, Sarmiento A. Changes in tibiotalar joint contact areas following experimentally induced tibial angular deformities. Clin Orthop. 1985;(199):72-80.
7. Ting AJ, Tarr RR, Sarmiento A, Wagner K, Resnick C. The role of subtalar motion and ankle contact pressure changes from angular deformities of the tibia. Foot Ankle. 1987;7(5):290-299.
8. van der Schoot DK, Den Outer AJ, Bode PJ, Obermann WR, van Vugt AB. Degenerative changes at the knee and ankle related to malunion of tibial fractures. 15-year follow-up of 88 patients. J Bone Joint Surg Br. 1996;78(5):722-725.
9. Kristensen KD, Kiaer T, Blicher J. No arthrosis of the ankle 20 years after malaligned tibial-shaft fracture. Acta Orthop Scand. 1989;60(2):208-209.
10. McKellop HA, Sigholm G, Redfern FC, Doyle B, Sarmiento A, Luck JV Sr. The effect of simulated fracture-angulations of the tibia on cartilage pressures in the knee joint. J Bone Joint Surg Am. 1991;73(9):1382-1391.
11. Merchant TC, Dietz FR. Long-term follow-up after fractures of the tibial and fibular shafts. J Bone Joint Surg Am. 1989;71(4):599-606.
12. Paley D, Herzenberg JE, Tetsworth K, McKie J, Bhave A. Deformity planning for frontal and sagittal plane corrective osteotomies. Orthop Clin North Am. 1994;25(3):425-465.
13. Perry J. Gait Analysis: Normal and Pathological Function. Thorofare, NJ: Slack; 1992.
14. Puno RM, Vaughan JJ, von Fraunhofer JA, Stetten ML, Johnson JR. A method of determining the angular malalignments of the knee and ankle joints resulting from a tibial malunion. Clin Orthop. 1987;(223):213-219.
15. Greenwood DC, Muir KR, Doherty M, Milner SA, Stevens M, Davis TR. Conservatively managed tibial shaft fractures in Nottingham, UK: are pain, osteoarthritis, and disability long-term complications? J Epidemiol Community Health. 1997;51(6):701-704.
16. Dehne E, Deffer PA, Hall RM, Brown PW, Johnson EV. The natural history of the fractured tibia. Surg Clin North Am. 1961;41(6):1495-1513.
17. Kitaoka HB, Schaap EJ, Chao EY, An KN. Displaced intra-articular fractures of the calcaneus treated non-operatively. Clinical results and analysis of motion and ground-reaction and temporal forces. J Bone Joint Surg Am. 1994;76(10):1531-1540.
18. Borrelli J Jr, Goldfarb C, Ricci W, Wagner JM, Engsberg JR. Functional outcome after isolated acetabular fractures. J Orthop Trauma. 2002;16(2):73-81.
19. Borrelli J Jr, Ricci WM, Anglen JO, Gregush R, Engsberg J. Muscle strength recovery and its effects on outcome after open reduction and internal fixation of acetabular fractures. J Orthop Trauma. 2006;20(6):388-395.
20. Jaglal S, Lakhani Z, Schatzker J. Reliability, validity, and responsiveness of the lower extremity measure for patients with a hip fracture. J Bone Joint Surg Am. 2000;82(7):955-962.
21. Madsen MS, Ritter MA, Morris HH, et al. The effect of total hip arthroplasty surgical approach on gait. J Orthop Res. 2004;22(1):44-50.
22. Mittlmeier T, Morlock MM, Hertlein H, et al. Analysis of morphology and gait function after intraarticular calcaneal fracture. J Orthop Trauma. 1993;7(4):303-310.
23. Song KM, Halliday SE, Little DG. The effect of limb-length discrepancy on gait. J Bone Joint Surg Am. 1997;79(11):1690-1698.
24. Zlowodzki M, Obremskey WT, Thomison JB, Kregor PJ. Functional outcome after treatment of lower-extremity nonunions. J Trauma. 2005;58(2):312-317.
25. Sanders R, Anglen JO, Mark JB. Oblique osteotomy for the correction of tibial malunion. J Bone Joint Surg Am. 1995;77(2):240-246.
26. Sangeorzan BJ, Sangeorzan BP, Hansen ST Jr, Judd RP. Mathematically directed single-cut osteotomy for correction of tibial malunion. J Orthop Trauma. 1989;3(4):267-275.
27. Borrelli J Jr, Leduc S, Gregush R, Ricci WM. Tricortical bone grafts for treatment of malaligned tibias and fibulas. Clin Orthop. 2009;467(4):1056-1063.
28. Engelberg R, Martin DP, Agel J, Obremsky W, Coronado G, Swiontkowski MF. Musculoskeletal Function Assessment instrument: criterion and construct validity. J Orthop Res. 1996;14(2):182-192.
29. Engelberg R, Martin DP, Agel J, Swiontkowski MF. Musculoskeletal Function Assessment: reference values for patient and non-patient samples. J Orthop Res. 1999;17(1):101-109.
30. Swiontkowski MF, Engelberg R, Martin DP, Agel J. Short Musculoskeletal Function Assessment questionnaire: validity, reliability, and responsiveness. J Bone Joint Surg Am. 1999;81(9):1245-1260.
31. Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30(6):473-483.
32. Graehl PM, Hersh MR, Heckman JD. Supramalleolar osteotomy for the treatment of symptomatic tibial malunion. J Orthop Trauma. 1987;1(4):281-292.
33. Bhave A, Paley D, Herzenberg JE. Improvement in gait parameters after lengthening for the treatment of limb-length discrepancy. J Bone Joint Surg Am. 1999;81(4):529-534.
34. Wu DD, Burr DB, Boyd RD, Radin EL. Bone and cartilage changes following experimental varus or valgus tibial angulation. J Orthop Res. 1990;8(4):572-585.
1. Probe RA. Lower extremity angular malunion: evaluation and surgical correction. J Am Acad Orthop Surg. 2003;11(5):302-311.
2. van der Linden W, Larsson K. Plate fixation versus conservative treatment of tibial shaft fractures. A randomized trial. J Bone Joint Surg Am. 1979;61(6):873-878.
3. Kettelkamp DB, Hillberry BM, Murrish DE, Heck DA. Degenerative arthritis of the knee secondary to fracture malunion. Clin Orthop. 1988;(234):159-169.
4. Milner SA, Davis TR, Muir KR, Greenwood DC, Doherty M. Long-term outcome after tibial shaft fracture: is malunion important? J Bone Joint Surg Am. 2002;84(6):971-980.
5. Puno RM, Vaughan JJ, Stetten ML, Johnson JR. Long-term effects of tibial angular malunion on the knee and ankle joints. J Orthop Trauma. 1991;5(3):247-254.
6. Tarr RR, Resnick CT, Wagner KS, Sarmiento A. Changes in tibiotalar joint contact areas following experimentally induced tibial angular deformities. Clin Orthop. 1985;(199):72-80.
7. Ting AJ, Tarr RR, Sarmiento A, Wagner K, Resnick C. The role of subtalar motion and ankle contact pressure changes from angular deformities of the tibia. Foot Ankle. 1987;7(5):290-299.
8. van der Schoot DK, Den Outer AJ, Bode PJ, Obermann WR, van Vugt AB. Degenerative changes at the knee and ankle related to malunion of tibial fractures. 15-year follow-up of 88 patients. J Bone Joint Surg Br. 1996;78(5):722-725.
9. Kristensen KD, Kiaer T, Blicher J. No arthrosis of the ankle 20 years after malaligned tibial-shaft fracture. Acta Orthop Scand. 1989;60(2):208-209.
10. McKellop HA, Sigholm G, Redfern FC, Doyle B, Sarmiento A, Luck JV Sr. The effect of simulated fracture-angulations of the tibia on cartilage pressures in the knee joint. J Bone Joint Surg Am. 1991;73(9):1382-1391.
11. Merchant TC, Dietz FR. Long-term follow-up after fractures of the tibial and fibular shafts. J Bone Joint Surg Am. 1989;71(4):599-606.
12. Paley D, Herzenberg JE, Tetsworth K, McKie J, Bhave A. Deformity planning for frontal and sagittal plane corrective osteotomies. Orthop Clin North Am. 1994;25(3):425-465.
13. Perry J. Gait Analysis: Normal and Pathological Function. Thorofare, NJ: Slack; 1992.
14. Puno RM, Vaughan JJ, von Fraunhofer JA, Stetten ML, Johnson JR. A method of determining the angular malalignments of the knee and ankle joints resulting from a tibial malunion. Clin Orthop. 1987;(223):213-219.
15. Greenwood DC, Muir KR, Doherty M, Milner SA, Stevens M, Davis TR. Conservatively managed tibial shaft fractures in Nottingham, UK: are pain, osteoarthritis, and disability long-term complications? J Epidemiol Community Health. 1997;51(6):701-704.
16. Dehne E, Deffer PA, Hall RM, Brown PW, Johnson EV. The natural history of the fractured tibia. Surg Clin North Am. 1961;41(6):1495-1513.
17. Kitaoka HB, Schaap EJ, Chao EY, An KN. Displaced intra-articular fractures of the calcaneus treated non-operatively. Clinical results and analysis of motion and ground-reaction and temporal forces. J Bone Joint Surg Am. 1994;76(10):1531-1540.
18. Borrelli J Jr, Goldfarb C, Ricci W, Wagner JM, Engsberg JR. Functional outcome after isolated acetabular fractures. J Orthop Trauma. 2002;16(2):73-81.
19. Borrelli J Jr, Ricci WM, Anglen JO, Gregush R, Engsberg J. Muscle strength recovery and its effects on outcome after open reduction and internal fixation of acetabular fractures. J Orthop Trauma. 2006;20(6):388-395.
20. Jaglal S, Lakhani Z, Schatzker J. Reliability, validity, and responsiveness of the lower extremity measure for patients with a hip fracture. J Bone Joint Surg Am. 2000;82(7):955-962.
21. Madsen MS, Ritter MA, Morris HH, et al. The effect of total hip arthroplasty surgical approach on gait. J Orthop Res. 2004;22(1):44-50.
22. Mittlmeier T, Morlock MM, Hertlein H, et al. Analysis of morphology and gait function after intraarticular calcaneal fracture. J Orthop Trauma. 1993;7(4):303-310.
23. Song KM, Halliday SE, Little DG. The effect of limb-length discrepancy on gait. J Bone Joint Surg Am. 1997;79(11):1690-1698.
24. Zlowodzki M, Obremskey WT, Thomison JB, Kregor PJ. Functional outcome after treatment of lower-extremity nonunions. J Trauma. 2005;58(2):312-317.
25. Sanders R, Anglen JO, Mark JB. Oblique osteotomy for the correction of tibial malunion. J Bone Joint Surg Am. 1995;77(2):240-246.
26. Sangeorzan BJ, Sangeorzan BP, Hansen ST Jr, Judd RP. Mathematically directed single-cut osteotomy for correction of tibial malunion. J Orthop Trauma. 1989;3(4):267-275.
27. Borrelli J Jr, Leduc S, Gregush R, Ricci WM. Tricortical bone grafts for treatment of malaligned tibias and fibulas. Clin Orthop. 2009;467(4):1056-1063.
28. Engelberg R, Martin DP, Agel J, Obremsky W, Coronado G, Swiontkowski MF. Musculoskeletal Function Assessment instrument: criterion and construct validity. J Orthop Res. 1996;14(2):182-192.
29. Engelberg R, Martin DP, Agel J, Swiontkowski MF. Musculoskeletal Function Assessment: reference values for patient and non-patient samples. J Orthop Res. 1999;17(1):101-109.
30. Swiontkowski MF, Engelberg R, Martin DP, Agel J. Short Musculoskeletal Function Assessment questionnaire: validity, reliability, and responsiveness. J Bone Joint Surg Am. 1999;81(9):1245-1260.
31. Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30(6):473-483.
32. Graehl PM, Hersh MR, Heckman JD. Supramalleolar osteotomy for the treatment of symptomatic tibial malunion. J Orthop Trauma. 1987;1(4):281-292.
33. Bhave A, Paley D, Herzenberg JE. Improvement in gait parameters after lengthening for the treatment of limb-length discrepancy. J Bone Joint Surg Am. 1999;81(4):529-534.
34. Wu DD, Burr DB, Boyd RD, Radin EL. Bone and cartilage changes following experimental varus or valgus tibial angulation. J Orthop Res. 1990;8(4):572-585.
Efficacy of Skin Preparation in Eradicating Organisms Before Total Knee Arthroplasty
Knee arthroplasty continues to be one of the most common and successful methods for treating severe arthritis and other painful arthropathies. Increasing steadily from 1998 to 2008, and with more than 676,000 procedures performed in 2008, knee arthroplasty remains the most common surgical joint replacement procedure.1
Although perioperative and long-term complications are uncommon, infection remains one of the most serious complications of total knee arthroplasty (TKA). Some studies have found a post-TKA infection rate of less than 1%.2 The solution of 2% chlorhexidine gluconate and 70% isopropyl alcohol (Chloraprep; Medi-Flex, Overland Park, Kansas) is commonly used for antiseptic skin preparation before surgery. Studies have shown significant decreases in post-TKA infection rates with preoperative use.3,4 Another study evaluated the efficacy of 3 different skin solutions and found Chloraprep to be the most efficient in eradicating bacteria from the foot and ankle before surgery. The investigators noted that, even with preoperative use of Chloraprep, 23% of patients had residual bacteria on the surface of the skin between the toes.5 Like the foot, the popliteal fossa is an intertriginous area that may harbor normal flora, including gram-positive cocci, in large numbers, mainly because of the contact between 2 skin surfaces. Although postoperative infection rates decrease with use of Chloraprep, its presurgical efficacy in killing bacteria on another intertriginous area, the popliteal fossa, is largely unknown. Also unknown are susceptible organism species and organism population numbers.
Concerned that our skin preparation might be ineffective, we conducted a study to evaluate the efficacy of Chloraprep skin preparation in eradicating organisms before TKA, to isolate the type and number of organisms, and to evaluate several other contributing factors that could lead to infection.
Materials and Methods
This prospective study included 99 patients who were undergoing primary TKA at John Peter Smith Hospital between July 1, 2011 and August 31, 2012. An attempt was made to enroll consecutive TKA patients, and all patients agreed to participate, but a few were not enrolled because the study team had not asked for their consent before they were taken to the operating room. Patients did not receive monetary compensation for participation. Exclusion criteria were pregnancy, imprisonment, and age under 18 years. The study was approved by the institutional review boards at John Peter Smith Hospital and the University of North Texas Health Science Center.
Each lower extremity was prepared with Chloraprep according to the manufacturer’s instructions. Preparation was done by well-trained operating room staff members who were supervised by the surgeon (Dr. Sanchez or Dr. Wagner) but were not involved in the study. With use of the Chloraprep applicator, the solution was applied in a back-and-forth manner to the entire operative leg for at least 30 seconds, and then discarded. This scrub procedure was repeated with a second applicator before standard drapes were placed. The leg was left to air-dry for at least 30 seconds, and the drapes were placed before postsolution swabbing and before the iodine-impregnated adhesive drape was placed around the knee. During drying, the solution was not blotted, wiped away, or touched with instrumentation. Patients were swabbed with an epidermal sterile swab in the popliteal fossa of the knee undergoing surgery, both before solution application (presolution swab) and after (postsolution swab). Only the operating surgeon participated in swabbing the patients. Aerobic and anaerobic swabs were vigorously rubbed over a 2- to 3-in wide area across the entire posterior flexion crease surface.
The collected pre- and postsolution swabs were sent to John Peter Smith Laboratory for identification of organisms. Anaerobic swabs were cultured in thioglycolate broth and on 4 plates: MacConkey agar, Columbia colistin–nalidixic acid agar, chocolate agar, and sheep blood agar. Aerobic swabs were cultured in thioglycolate broth with hemin and vitamin K and on 4 plates: anaerobic blood agar, bile esculin agar, kanamycin and vancomycin agar, and Columbia colistin–nalidixic acid agar. Anaerobic plates were incubated in an anoxic environment. The plates were then read daily, and final reports were issued after 48 hours (for aerobic bacterial isolates) and 72 hours (for anaerobic bacterial isolates), as was the standard at the time.
Additional patient data were collected for possible correlations: American Society of Anesthesiologists (ASA) classification (physical status),6 body mass index (BMI), age, sex, arthroplasty type (unilateral, bilateral), and diabetic status. In addition, patients were asked if they had used Hibiclens antiseptic/antimicrobial skin cleanser daily during the week before surgery—as they had been instructed to do—and the number of times they had used the cleanser.
Study data were analyzed and were used to stratify patients into several groups. Each group had multiple factors evaluated.
Descriptive statistics were used to characterize the patient demographic information. Chi-square analyses were performed to evaluate the difference between presence of organisms before and after solution application, and the data were also layered with reported Hibiclens cleanser use. In addition, binary logistic regression was used to determine if demographic variables could predict presence of organism isolates before and after solution application. Data analyses were conducted using IBM SPSS Statistics Version 20.
Results
No patient had a postoperative infection. Culture isolates grew in 20 (20%) of the 99 patients before solution application and in 5 (5%) of the 99 after application. Of the 20 patients with presolution culture isolates, 16 (80%) had 1 bacterial isolate, and 4 (20%) had 2 or more species. Presolution isolates included normal flora (10, 50%), coagulase-negative Staphylococcus aureus (6, 30%), rare Bacillus (3, 15%), Micrococcus luteus (1, 5%), rare gram-negative (1, 5%), rare gram-positive (1, 5%), and Staphylococcus hominis (1, 5%) (Figure 1). Postsolution isolates included coagulase-negative S aureus (3, 60%), rare Bacillus (1, 20%), and rare Serratia odorifera (1, 20%) (Figure 2). Two postsolution isolates did not have an associated presolution isolate. Presolution organism isolation was an important predictor of postsolution organism isolation (P < .046).
BMI was recorded for all patients. Mean BMI was 35 (range, 20-63). Distribution was as follows: BMI under 20 (3 patients), under 30 (30 patients), under 40 (47 patients), under 50 (14 patients), under 60 (4 patients), and over 60 (1 patient). Mean presolution BMI was significantly (P < .03) higher for patients with bacterial isolates than for patients without isolates (38 and 34, respectively). Mean postsolution BMI was 40 for patients with bacterial isolates and 35 for patients without isolates (Figure 3). Of the 33 patients with BMI under 30, 3 (9%) had presolution isolates and 1 (3%) had postsolution isolates. Of the 66 patients with BMI over 30, 17 (26%) had presolution isolates and 4 (6%) had postsolution isolates (Table).
Of the 99 patients, 30 (30%) had diabetes. Of these 30 patients, 9 (30%) had presolution isolates (45% of all presolution isolates) and 3 (10%) had postsolution isolates (60% of all postsolution isolates.) Although neither pre- nor postsolution results were statistically significant (P = .172) for increasing organism isolation in patients with diabetes, the odds ratio for these patients was 3.6 when the focus was on the likelihood of postsolution organism isolation.
Mean age was 57 years (range, 29-87 years). Results were not statistically significant for age being a likely factor for organism isolate prediction.
There were 81 women and 18 men in the study. Of the 81 women, 16 (20%) had positive presolution cultures and 5 (6%) had positive postsolution cultures. Of the 18 men, 4 (22%) had positive presolution cultures and none had a positive postsolution culture.
Race was recorded. Forty-nine patients were white, 27 black, 18 Hispanic, and 5 unknown. Presolution, 12 whites (24%), 5 blacks (19%), and 3 Hispanics (17%) had positive cultures. Postsolution, 1 white (2%), 1 black (4%), 3 Hispanics (17%), and 1 patient of unknown race (20%) had positive cultures.
ASA classifications were recorded and analyzed. Of the 99 patients, 38 were classified ASA-2, 60 were ASA-3, and 1 was ASA-4. Presolution, 9 (24%) of the 38 ASA-2 patients and 11 (18%) of the 60 ASA-3 patients had positive cultures; postsolution, 2 (5%) of the 38 ASA-2 patients and 3 (5%) of the 60 ASA-3 patients had positive cultures. The 1 ASA-4 patient had neither presolution nor postsolution positive cultures.
Types of TKA (bilateral, unilateral) were recorded. Of the 99 patients, 89 had unilateral TKAs and 10 had bilateral TKAs. Presolution, 19 (21%) of the 89 unilaterals and 1 (10%) of the 10 bilaterals had positive cultures. Postsolution, 5 (6%) of the 89 unilaterals and none of the 10 bilaterals had positive cultures.
Patients were also verbally asked how many cleanser baths they had taken before surgery. Of the 99 patients, 88 reported having taken 1 or more cleanser baths, and 1 reported no baths; 10 patients’ responses were not available. The 88 patients who had taken at least 1 cleanser bath were divided into 3 groups: 1 bath (35 patients), 2 baths (49 patients), and 3 or more baths (4 patients). Presolution, positive cultures were found for 18 (20%) of the 88 patients; for 7 (20%) of the 35 patients with 1 bath; for 10 (20%) of the 49 patients with 2 baths; and for 1 (25%) of the 4 patients with 3 or more baths. Postsolution, positive cultures were found for 5 (6%) of the 88 patients; for 2 (6%) of the 35 patients with 1 bath; for 3 (6%) of the 49 patients with 2 baths; and for 0 (0%) of the 4 patients with 3 or more baths. The 1 patient with no baths did not have a positive culture. Of the 10 patients whose responses were unavailable, 2 patients had positive presolution cultures and no patients had a positive postsolution culture.
Discussion
The efficacy of using Chloraprep before TKA has not been well assessed in orthopedic practice. However, compared with other preoperative solutions, chlorhexidine has been shown to be significantly better in preventing post-TKA infections.4 Other studies have found it far more effective than other commonly used surgical preparations in eliminating microorganisms in hip arthroplasty and foot surgery.5,7 Our study, focused on the efficacy of Chloraprep in killing bacteria, found the solution effective in removing 85% (17/20) of cultured presolution organisms.
Of the bacterial isolates cultured, normal flora were effectively removed from all associated postsolution cultures. Although most of the bacterial isolates were eliminated after solution application, both coagulase-negative S aureus and rare Bacillus species were found both pre- and postsolution, suggesting either inadequate skin preparation or resistant bacteria.
With respect to the secondary variables, our study data showed that BMI was an important predictor for bacterial isolates, significantly so presolution (P < .03). Mean BMI for the overall study was 35, firmly in the obese category. Only when BMI increased to 38 did it become significant as a predictor for postsolution organisms. Mean postsolution BMI was even higher, 40, which is in the morbidly obese category. Interestingly, the percentage of nonobese patients (BMI, <30) with positive presolution cultures was only 9%, versus the 20% with positive presolution cultures overall. In addition, 1 nonobese patient had positive postsolution cultures.
Other studies have linked higher BMI to higher rates of surgical site infection and other complications, but it is unknown if the infections are due to higher bacterial counts in the patients with high BMI or to other factors, such as reduced wound healing or decreased immune response. More research is needed to determine if the number of organisms in patients with high BMI correlates to a higher risk for surgical site infection.8 As expected, along with BMI (>38), presolution organism isolation was an important predictor for postsolution organism isolation. Patients with presolution organism isolation were 24 times more likely to have postsolution isolates.
Even though diabetic status was not significant for predicting bacterial isolation, patients with diabetes were 3.6 times more likely than patients without diabetes to have a positive culture. Other studies have shown that, compared with patients without diabetes, patients with diabetes had a higher chance of postoperative infection.9,10
In this study, 18 of 20 patients with presolution organism isolates reported they had been compliant in taking the recommended preoperative cleanser baths. This finding may indicate that preoperative cleanser baths are ineffective. However, only 20% of our patients had positive presolution cultures, whereas Ostrander and colleagues5 reported 30% positive pre-preparation cultures from the anterior knee. A recent Cochrane Database System Review did not provide clear evidence of benefit for preoperative showering or bathing with chlorhexidine over other wash products.11 Although their benefit may be questionable, we will continue to recommend preoperative cleanser baths.
One limitation of this study is sample size. Although size was sufficient for determining the efficacy of Chloraprep in the intertriginous area of the back of the knee, the lack of statistical significance (eg, effect of diabetes) may not be accurate. In addition, because the nurse who prepared patients’ skin was aware of the study and was supervised in every case, it is possible that the preparation was done more carefully than usual, resulting in more negative cultures than average. Also, compliance in taking preoperative cleanser baths was subjectively determined. Patients may have reported more baths than were actually taken. Still another study limitation is that 2 postsolution isolates did not have an associated presolution isolate. Although we think this may have resulted from laboratory contamination, it is possible the presolution swabs did not accurately determine true bacterial counts in these cases.
Conclusion
A study that showed significant residual bacteria between patients’ toes after chlorhexidine skin preparation5 left us concerned that Chloraprep skin preparation for TKA might not be adequate. The present study showed that this solution was effective in eliminating bacteria from the intertriginous area of the back of the knee in 95% of patients. Skin preparation appears to be less effective in patients with higher BMI.
1. Losina E, Thornhill TS, Rome BN, Wright J, Katz JN. The dramatic increase in total knee replacement utilization rates in the United States cannot be fully explained by growth in population size and the obesity epidemic. J Bone Joint Surg Am. 2012;94(3):201-207.
2. Poultsides LA, Ma Y, Della Valle AG, Chiu YL, Sculco TP, Memtsoudis SG. In-hospital surgical site infections after primary hip and knee arthroplasty—incidence and risk factors. J Arthroplasty. 2013;28(3):385-389.
3. Johnson AJ, Kapadia BH, Daley JA, Molina CB, Mont MA. Chlorhexidine reduces infections in knee arthroplasty. J Knee Surg. 2013;26(3):213-218.
4. Zywiel MG, Daley JA, Delanois RE, Naziri Q, Johnson AJ, Mont MA. Advance pre-operative chlorhexidine reduces the incidence of surgical site infections in knee arthroplasty. Int Orthop. 2011;35(7):1001-1006.
5. Ostrander RV, Botte MJ, Brage ME. Efficacy of surgical preparation solutions in foot and ankle surgery. J Bone Joint Surg Am. 2005;87(5):980-985.
6. Wolters U, Wolf T, Stützer H, Schröder T. ASA classification and perioperative variables as predictors of postoperative outcome. Br J Anaesth. 1996;77(2):217-222.
7. Johnson AJ, Daley JA, Zywiel MG, Delanois RE, Mont MA. Preoperative chlorhexidine preparation and the incidence of surgical site infections after hip arthroplasty. J Arthroplasty. 2010;25(6 suppl):98-102.
8. Samson AJ, Mercer GE, Campbell DG. Total knee replacement in the morbidly obese: a literature review. ANZ J Surg. 2010;80(9):595-599.
9. Iorio R, Williams KM, Marcantonio AJ, Specht LM, Tilzey JF, Healy WL. Diabetes mellitus, hemoglobin A1C, and the incidence of total joint arthroplasty infection. J Arthroplasty. 2012;27(5):726-729.
10. Viens NA, Hug KT, Marchant MH, Cook C, Vail TP, Bolognesi MP. Role of diabetes type in perioperative outcomes after hip and knee arthroplasty in the United States. J Surg Orthop Adv. 2012;21(4):253-260.
11. Webster J, Osborne S. Preoperative bathing or showering with skin antiseptics to prevent surgical site infection. Cochrane Database Syst Rev. 2012;9:CD004985.
Knee arthroplasty continues to be one of the most common and successful methods for treating severe arthritis and other painful arthropathies. Increasing steadily from 1998 to 2008, and with more than 676,000 procedures performed in 2008, knee arthroplasty remains the most common surgical joint replacement procedure.1
Although perioperative and long-term complications are uncommon, infection remains one of the most serious complications of total knee arthroplasty (TKA). Some studies have found a post-TKA infection rate of less than 1%.2 The solution of 2% chlorhexidine gluconate and 70% isopropyl alcohol (Chloraprep; Medi-Flex, Overland Park, Kansas) is commonly used for antiseptic skin preparation before surgery. Studies have shown significant decreases in post-TKA infection rates with preoperative use.3,4 Another study evaluated the efficacy of 3 different skin solutions and found Chloraprep to be the most efficient in eradicating bacteria from the foot and ankle before surgery. The investigators noted that, even with preoperative use of Chloraprep, 23% of patients had residual bacteria on the surface of the skin between the toes.5 Like the foot, the popliteal fossa is an intertriginous area that may harbor normal flora, including gram-positive cocci, in large numbers, mainly because of the contact between 2 skin surfaces. Although postoperative infection rates decrease with use of Chloraprep, its presurgical efficacy in killing bacteria on another intertriginous area, the popliteal fossa, is largely unknown. Also unknown are susceptible organism species and organism population numbers.
Concerned that our skin preparation might be ineffective, we conducted a study to evaluate the efficacy of Chloraprep skin preparation in eradicating organisms before TKA, to isolate the type and number of organisms, and to evaluate several other contributing factors that could lead to infection.
Materials and Methods
This prospective study included 99 patients who were undergoing primary TKA at John Peter Smith Hospital between July 1, 2011 and August 31, 2012. An attempt was made to enroll consecutive TKA patients, and all patients agreed to participate, but a few were not enrolled because the study team had not asked for their consent before they were taken to the operating room. Patients did not receive monetary compensation for participation. Exclusion criteria were pregnancy, imprisonment, and age under 18 years. The study was approved by the institutional review boards at John Peter Smith Hospital and the University of North Texas Health Science Center.
Each lower extremity was prepared with Chloraprep according to the manufacturer’s instructions. Preparation was done by well-trained operating room staff members who were supervised by the surgeon (Dr. Sanchez or Dr. Wagner) but were not involved in the study. With use of the Chloraprep applicator, the solution was applied in a back-and-forth manner to the entire operative leg for at least 30 seconds, and then discarded. This scrub procedure was repeated with a second applicator before standard drapes were placed. The leg was left to air-dry for at least 30 seconds, and the drapes were placed before postsolution swabbing and before the iodine-impregnated adhesive drape was placed around the knee. During drying, the solution was not blotted, wiped away, or touched with instrumentation. Patients were swabbed with an epidermal sterile swab in the popliteal fossa of the knee undergoing surgery, both before solution application (presolution swab) and after (postsolution swab). Only the operating surgeon participated in swabbing the patients. Aerobic and anaerobic swabs were vigorously rubbed over a 2- to 3-in wide area across the entire posterior flexion crease surface.
The collected pre- and postsolution swabs were sent to John Peter Smith Laboratory for identification of organisms. Anaerobic swabs were cultured in thioglycolate broth and on 4 plates: MacConkey agar, Columbia colistin–nalidixic acid agar, chocolate agar, and sheep blood agar. Aerobic swabs were cultured in thioglycolate broth with hemin and vitamin K and on 4 plates: anaerobic blood agar, bile esculin agar, kanamycin and vancomycin agar, and Columbia colistin–nalidixic acid agar. Anaerobic plates were incubated in an anoxic environment. The plates were then read daily, and final reports were issued after 48 hours (for aerobic bacterial isolates) and 72 hours (for anaerobic bacterial isolates), as was the standard at the time.
Additional patient data were collected for possible correlations: American Society of Anesthesiologists (ASA) classification (physical status),6 body mass index (BMI), age, sex, arthroplasty type (unilateral, bilateral), and diabetic status. In addition, patients were asked if they had used Hibiclens antiseptic/antimicrobial skin cleanser daily during the week before surgery—as they had been instructed to do—and the number of times they had used the cleanser.
Study data were analyzed and were used to stratify patients into several groups. Each group had multiple factors evaluated.
Descriptive statistics were used to characterize the patient demographic information. Chi-square analyses were performed to evaluate the difference between presence of organisms before and after solution application, and the data were also layered with reported Hibiclens cleanser use. In addition, binary logistic regression was used to determine if demographic variables could predict presence of organism isolates before and after solution application. Data analyses were conducted using IBM SPSS Statistics Version 20.
Results
No patient had a postoperative infection. Culture isolates grew in 20 (20%) of the 99 patients before solution application and in 5 (5%) of the 99 after application. Of the 20 patients with presolution culture isolates, 16 (80%) had 1 bacterial isolate, and 4 (20%) had 2 or more species. Presolution isolates included normal flora (10, 50%), coagulase-negative Staphylococcus aureus (6, 30%), rare Bacillus (3, 15%), Micrococcus luteus (1, 5%), rare gram-negative (1, 5%), rare gram-positive (1, 5%), and Staphylococcus hominis (1, 5%) (Figure 1). Postsolution isolates included coagulase-negative S aureus (3, 60%), rare Bacillus (1, 20%), and rare Serratia odorifera (1, 20%) (Figure 2). Two postsolution isolates did not have an associated presolution isolate. Presolution organism isolation was an important predictor of postsolution organism isolation (P < .046).
BMI was recorded for all patients. Mean BMI was 35 (range, 20-63). Distribution was as follows: BMI under 20 (3 patients), under 30 (30 patients), under 40 (47 patients), under 50 (14 patients), under 60 (4 patients), and over 60 (1 patient). Mean presolution BMI was significantly (P < .03) higher for patients with bacterial isolates than for patients without isolates (38 and 34, respectively). Mean postsolution BMI was 40 for patients with bacterial isolates and 35 for patients without isolates (Figure 3). Of the 33 patients with BMI under 30, 3 (9%) had presolution isolates and 1 (3%) had postsolution isolates. Of the 66 patients with BMI over 30, 17 (26%) had presolution isolates and 4 (6%) had postsolution isolates (Table).
Of the 99 patients, 30 (30%) had diabetes. Of these 30 patients, 9 (30%) had presolution isolates (45% of all presolution isolates) and 3 (10%) had postsolution isolates (60% of all postsolution isolates.) Although neither pre- nor postsolution results were statistically significant (P = .172) for increasing organism isolation in patients with diabetes, the odds ratio for these patients was 3.6 when the focus was on the likelihood of postsolution organism isolation.
Mean age was 57 years (range, 29-87 years). Results were not statistically significant for age being a likely factor for organism isolate prediction.
There were 81 women and 18 men in the study. Of the 81 women, 16 (20%) had positive presolution cultures and 5 (6%) had positive postsolution cultures. Of the 18 men, 4 (22%) had positive presolution cultures and none had a positive postsolution culture.
Race was recorded. Forty-nine patients were white, 27 black, 18 Hispanic, and 5 unknown. Presolution, 12 whites (24%), 5 blacks (19%), and 3 Hispanics (17%) had positive cultures. Postsolution, 1 white (2%), 1 black (4%), 3 Hispanics (17%), and 1 patient of unknown race (20%) had positive cultures.
ASA classifications were recorded and analyzed. Of the 99 patients, 38 were classified ASA-2, 60 were ASA-3, and 1 was ASA-4. Presolution, 9 (24%) of the 38 ASA-2 patients and 11 (18%) of the 60 ASA-3 patients had positive cultures; postsolution, 2 (5%) of the 38 ASA-2 patients and 3 (5%) of the 60 ASA-3 patients had positive cultures. The 1 ASA-4 patient had neither presolution nor postsolution positive cultures.
Types of TKA (bilateral, unilateral) were recorded. Of the 99 patients, 89 had unilateral TKAs and 10 had bilateral TKAs. Presolution, 19 (21%) of the 89 unilaterals and 1 (10%) of the 10 bilaterals had positive cultures. Postsolution, 5 (6%) of the 89 unilaterals and none of the 10 bilaterals had positive cultures.
Patients were also verbally asked how many cleanser baths they had taken before surgery. Of the 99 patients, 88 reported having taken 1 or more cleanser baths, and 1 reported no baths; 10 patients’ responses were not available. The 88 patients who had taken at least 1 cleanser bath were divided into 3 groups: 1 bath (35 patients), 2 baths (49 patients), and 3 or more baths (4 patients). Presolution, positive cultures were found for 18 (20%) of the 88 patients; for 7 (20%) of the 35 patients with 1 bath; for 10 (20%) of the 49 patients with 2 baths; and for 1 (25%) of the 4 patients with 3 or more baths. Postsolution, positive cultures were found for 5 (6%) of the 88 patients; for 2 (6%) of the 35 patients with 1 bath; for 3 (6%) of the 49 patients with 2 baths; and for 0 (0%) of the 4 patients with 3 or more baths. The 1 patient with no baths did not have a positive culture. Of the 10 patients whose responses were unavailable, 2 patients had positive presolution cultures and no patients had a positive postsolution culture.
Discussion
The efficacy of using Chloraprep before TKA has not been well assessed in orthopedic practice. However, compared with other preoperative solutions, chlorhexidine has been shown to be significantly better in preventing post-TKA infections.4 Other studies have found it far more effective than other commonly used surgical preparations in eliminating microorganisms in hip arthroplasty and foot surgery.5,7 Our study, focused on the efficacy of Chloraprep in killing bacteria, found the solution effective in removing 85% (17/20) of cultured presolution organisms.
Of the bacterial isolates cultured, normal flora were effectively removed from all associated postsolution cultures. Although most of the bacterial isolates were eliminated after solution application, both coagulase-negative S aureus and rare Bacillus species were found both pre- and postsolution, suggesting either inadequate skin preparation or resistant bacteria.
With respect to the secondary variables, our study data showed that BMI was an important predictor for bacterial isolates, significantly so presolution (P < .03). Mean BMI for the overall study was 35, firmly in the obese category. Only when BMI increased to 38 did it become significant as a predictor for postsolution organisms. Mean postsolution BMI was even higher, 40, which is in the morbidly obese category. Interestingly, the percentage of nonobese patients (BMI, <30) with positive presolution cultures was only 9%, versus the 20% with positive presolution cultures overall. In addition, 1 nonobese patient had positive postsolution cultures.
Other studies have linked higher BMI to higher rates of surgical site infection and other complications, but it is unknown if the infections are due to higher bacterial counts in the patients with high BMI or to other factors, such as reduced wound healing or decreased immune response. More research is needed to determine if the number of organisms in patients with high BMI correlates to a higher risk for surgical site infection.8 As expected, along with BMI (>38), presolution organism isolation was an important predictor for postsolution organism isolation. Patients with presolution organism isolation were 24 times more likely to have postsolution isolates.
Even though diabetic status was not significant for predicting bacterial isolation, patients with diabetes were 3.6 times more likely than patients without diabetes to have a positive culture. Other studies have shown that, compared with patients without diabetes, patients with diabetes had a higher chance of postoperative infection.9,10
In this study, 18 of 20 patients with presolution organism isolates reported they had been compliant in taking the recommended preoperative cleanser baths. This finding may indicate that preoperative cleanser baths are ineffective. However, only 20% of our patients had positive presolution cultures, whereas Ostrander and colleagues5 reported 30% positive pre-preparation cultures from the anterior knee. A recent Cochrane Database System Review did not provide clear evidence of benefit for preoperative showering or bathing with chlorhexidine over other wash products.11 Although their benefit may be questionable, we will continue to recommend preoperative cleanser baths.
One limitation of this study is sample size. Although size was sufficient for determining the efficacy of Chloraprep in the intertriginous area of the back of the knee, the lack of statistical significance (eg, effect of diabetes) may not be accurate. In addition, because the nurse who prepared patients’ skin was aware of the study and was supervised in every case, it is possible that the preparation was done more carefully than usual, resulting in more negative cultures than average. Also, compliance in taking preoperative cleanser baths was subjectively determined. Patients may have reported more baths than were actually taken. Still another study limitation is that 2 postsolution isolates did not have an associated presolution isolate. Although we think this may have resulted from laboratory contamination, it is possible the presolution swabs did not accurately determine true bacterial counts in these cases.
Conclusion
A study that showed significant residual bacteria between patients’ toes after chlorhexidine skin preparation5 left us concerned that Chloraprep skin preparation for TKA might not be adequate. The present study showed that this solution was effective in eliminating bacteria from the intertriginous area of the back of the knee in 95% of patients. Skin preparation appears to be less effective in patients with higher BMI.
Knee arthroplasty continues to be one of the most common and successful methods for treating severe arthritis and other painful arthropathies. Increasing steadily from 1998 to 2008, and with more than 676,000 procedures performed in 2008, knee arthroplasty remains the most common surgical joint replacement procedure.1
Although perioperative and long-term complications are uncommon, infection remains one of the most serious complications of total knee arthroplasty (TKA). Some studies have found a post-TKA infection rate of less than 1%.2 The solution of 2% chlorhexidine gluconate and 70% isopropyl alcohol (Chloraprep; Medi-Flex, Overland Park, Kansas) is commonly used for antiseptic skin preparation before surgery. Studies have shown significant decreases in post-TKA infection rates with preoperative use.3,4 Another study evaluated the efficacy of 3 different skin solutions and found Chloraprep to be the most efficient in eradicating bacteria from the foot and ankle before surgery. The investigators noted that, even with preoperative use of Chloraprep, 23% of patients had residual bacteria on the surface of the skin between the toes.5 Like the foot, the popliteal fossa is an intertriginous area that may harbor normal flora, including gram-positive cocci, in large numbers, mainly because of the contact between 2 skin surfaces. Although postoperative infection rates decrease with use of Chloraprep, its presurgical efficacy in killing bacteria on another intertriginous area, the popliteal fossa, is largely unknown. Also unknown are susceptible organism species and organism population numbers.
Concerned that our skin preparation might be ineffective, we conducted a study to evaluate the efficacy of Chloraprep skin preparation in eradicating organisms before TKA, to isolate the type and number of organisms, and to evaluate several other contributing factors that could lead to infection.
Materials and Methods
This prospective study included 99 patients who were undergoing primary TKA at John Peter Smith Hospital between July 1, 2011 and August 31, 2012. An attempt was made to enroll consecutive TKA patients, and all patients agreed to participate, but a few were not enrolled because the study team had not asked for their consent before they were taken to the operating room. Patients did not receive monetary compensation for participation. Exclusion criteria were pregnancy, imprisonment, and age under 18 years. The study was approved by the institutional review boards at John Peter Smith Hospital and the University of North Texas Health Science Center.
Each lower extremity was prepared with Chloraprep according to the manufacturer’s instructions. Preparation was done by well-trained operating room staff members who were supervised by the surgeon (Dr. Sanchez or Dr. Wagner) but were not involved in the study. With use of the Chloraprep applicator, the solution was applied in a back-and-forth manner to the entire operative leg for at least 30 seconds, and then discarded. This scrub procedure was repeated with a second applicator before standard drapes were placed. The leg was left to air-dry for at least 30 seconds, and the drapes were placed before postsolution swabbing and before the iodine-impregnated adhesive drape was placed around the knee. During drying, the solution was not blotted, wiped away, or touched with instrumentation. Patients were swabbed with an epidermal sterile swab in the popliteal fossa of the knee undergoing surgery, both before solution application (presolution swab) and after (postsolution swab). Only the operating surgeon participated in swabbing the patients. Aerobic and anaerobic swabs were vigorously rubbed over a 2- to 3-in wide area across the entire posterior flexion crease surface.
The collected pre- and postsolution swabs were sent to John Peter Smith Laboratory for identification of organisms. Anaerobic swabs were cultured in thioglycolate broth and on 4 plates: MacConkey agar, Columbia colistin–nalidixic acid agar, chocolate agar, and sheep blood agar. Aerobic swabs were cultured in thioglycolate broth with hemin and vitamin K and on 4 plates: anaerobic blood agar, bile esculin agar, kanamycin and vancomycin agar, and Columbia colistin–nalidixic acid agar. Anaerobic plates were incubated in an anoxic environment. The plates were then read daily, and final reports were issued after 48 hours (for aerobic bacterial isolates) and 72 hours (for anaerobic bacterial isolates), as was the standard at the time.
Additional patient data were collected for possible correlations: American Society of Anesthesiologists (ASA) classification (physical status),6 body mass index (BMI), age, sex, arthroplasty type (unilateral, bilateral), and diabetic status. In addition, patients were asked if they had used Hibiclens antiseptic/antimicrobial skin cleanser daily during the week before surgery—as they had been instructed to do—and the number of times they had used the cleanser.
Study data were analyzed and were used to stratify patients into several groups. Each group had multiple factors evaluated.
Descriptive statistics were used to characterize the patient demographic information. Chi-square analyses were performed to evaluate the difference between presence of organisms before and after solution application, and the data were also layered with reported Hibiclens cleanser use. In addition, binary logistic regression was used to determine if demographic variables could predict presence of organism isolates before and after solution application. Data analyses were conducted using IBM SPSS Statistics Version 20.
Results
No patient had a postoperative infection. Culture isolates grew in 20 (20%) of the 99 patients before solution application and in 5 (5%) of the 99 after application. Of the 20 patients with presolution culture isolates, 16 (80%) had 1 bacterial isolate, and 4 (20%) had 2 or more species. Presolution isolates included normal flora (10, 50%), coagulase-negative Staphylococcus aureus (6, 30%), rare Bacillus (3, 15%), Micrococcus luteus (1, 5%), rare gram-negative (1, 5%), rare gram-positive (1, 5%), and Staphylococcus hominis (1, 5%) (Figure 1). Postsolution isolates included coagulase-negative S aureus (3, 60%), rare Bacillus (1, 20%), and rare Serratia odorifera (1, 20%) (Figure 2). Two postsolution isolates did not have an associated presolution isolate. Presolution organism isolation was an important predictor of postsolution organism isolation (P < .046).
BMI was recorded for all patients. Mean BMI was 35 (range, 20-63). Distribution was as follows: BMI under 20 (3 patients), under 30 (30 patients), under 40 (47 patients), under 50 (14 patients), under 60 (4 patients), and over 60 (1 patient). Mean presolution BMI was significantly (P < .03) higher for patients with bacterial isolates than for patients without isolates (38 and 34, respectively). Mean postsolution BMI was 40 for patients with bacterial isolates and 35 for patients without isolates (Figure 3). Of the 33 patients with BMI under 30, 3 (9%) had presolution isolates and 1 (3%) had postsolution isolates. Of the 66 patients with BMI over 30, 17 (26%) had presolution isolates and 4 (6%) had postsolution isolates (Table).
Of the 99 patients, 30 (30%) had diabetes. Of these 30 patients, 9 (30%) had presolution isolates (45% of all presolution isolates) and 3 (10%) had postsolution isolates (60% of all postsolution isolates.) Although neither pre- nor postsolution results were statistically significant (P = .172) for increasing organism isolation in patients with diabetes, the odds ratio for these patients was 3.6 when the focus was on the likelihood of postsolution organism isolation.
Mean age was 57 years (range, 29-87 years). Results were not statistically significant for age being a likely factor for organism isolate prediction.
There were 81 women and 18 men in the study. Of the 81 women, 16 (20%) had positive presolution cultures and 5 (6%) had positive postsolution cultures. Of the 18 men, 4 (22%) had positive presolution cultures and none had a positive postsolution culture.
Race was recorded. Forty-nine patients were white, 27 black, 18 Hispanic, and 5 unknown. Presolution, 12 whites (24%), 5 blacks (19%), and 3 Hispanics (17%) had positive cultures. Postsolution, 1 white (2%), 1 black (4%), 3 Hispanics (17%), and 1 patient of unknown race (20%) had positive cultures.
ASA classifications were recorded and analyzed. Of the 99 patients, 38 were classified ASA-2, 60 were ASA-3, and 1 was ASA-4. Presolution, 9 (24%) of the 38 ASA-2 patients and 11 (18%) of the 60 ASA-3 patients had positive cultures; postsolution, 2 (5%) of the 38 ASA-2 patients and 3 (5%) of the 60 ASA-3 patients had positive cultures. The 1 ASA-4 patient had neither presolution nor postsolution positive cultures.
Types of TKA (bilateral, unilateral) were recorded. Of the 99 patients, 89 had unilateral TKAs and 10 had bilateral TKAs. Presolution, 19 (21%) of the 89 unilaterals and 1 (10%) of the 10 bilaterals had positive cultures. Postsolution, 5 (6%) of the 89 unilaterals and none of the 10 bilaterals had positive cultures.
Patients were also verbally asked how many cleanser baths they had taken before surgery. Of the 99 patients, 88 reported having taken 1 or more cleanser baths, and 1 reported no baths; 10 patients’ responses were not available. The 88 patients who had taken at least 1 cleanser bath were divided into 3 groups: 1 bath (35 patients), 2 baths (49 patients), and 3 or more baths (4 patients). Presolution, positive cultures were found for 18 (20%) of the 88 patients; for 7 (20%) of the 35 patients with 1 bath; for 10 (20%) of the 49 patients with 2 baths; and for 1 (25%) of the 4 patients with 3 or more baths. Postsolution, positive cultures were found for 5 (6%) of the 88 patients; for 2 (6%) of the 35 patients with 1 bath; for 3 (6%) of the 49 patients with 2 baths; and for 0 (0%) of the 4 patients with 3 or more baths. The 1 patient with no baths did not have a positive culture. Of the 10 patients whose responses were unavailable, 2 patients had positive presolution cultures and no patients had a positive postsolution culture.
Discussion
The efficacy of using Chloraprep before TKA has not been well assessed in orthopedic practice. However, compared with other preoperative solutions, chlorhexidine has been shown to be significantly better in preventing post-TKA infections.4 Other studies have found it far more effective than other commonly used surgical preparations in eliminating microorganisms in hip arthroplasty and foot surgery.5,7 Our study, focused on the efficacy of Chloraprep in killing bacteria, found the solution effective in removing 85% (17/20) of cultured presolution organisms.
Of the bacterial isolates cultured, normal flora were effectively removed from all associated postsolution cultures. Although most of the bacterial isolates were eliminated after solution application, both coagulase-negative S aureus and rare Bacillus species were found both pre- and postsolution, suggesting either inadequate skin preparation or resistant bacteria.
With respect to the secondary variables, our study data showed that BMI was an important predictor for bacterial isolates, significantly so presolution (P < .03). Mean BMI for the overall study was 35, firmly in the obese category. Only when BMI increased to 38 did it become significant as a predictor for postsolution organisms. Mean postsolution BMI was even higher, 40, which is in the morbidly obese category. Interestingly, the percentage of nonobese patients (BMI, <30) with positive presolution cultures was only 9%, versus the 20% with positive presolution cultures overall. In addition, 1 nonobese patient had positive postsolution cultures.
Other studies have linked higher BMI to higher rates of surgical site infection and other complications, but it is unknown if the infections are due to higher bacterial counts in the patients with high BMI or to other factors, such as reduced wound healing or decreased immune response. More research is needed to determine if the number of organisms in patients with high BMI correlates to a higher risk for surgical site infection.8 As expected, along with BMI (>38), presolution organism isolation was an important predictor for postsolution organism isolation. Patients with presolution organism isolation were 24 times more likely to have postsolution isolates.
Even though diabetic status was not significant for predicting bacterial isolation, patients with diabetes were 3.6 times more likely than patients without diabetes to have a positive culture. Other studies have shown that, compared with patients without diabetes, patients with diabetes had a higher chance of postoperative infection.9,10
In this study, 18 of 20 patients with presolution organism isolates reported they had been compliant in taking the recommended preoperative cleanser baths. This finding may indicate that preoperative cleanser baths are ineffective. However, only 20% of our patients had positive presolution cultures, whereas Ostrander and colleagues5 reported 30% positive pre-preparation cultures from the anterior knee. A recent Cochrane Database System Review did not provide clear evidence of benefit for preoperative showering or bathing with chlorhexidine over other wash products.11 Although their benefit may be questionable, we will continue to recommend preoperative cleanser baths.
One limitation of this study is sample size. Although size was sufficient for determining the efficacy of Chloraprep in the intertriginous area of the back of the knee, the lack of statistical significance (eg, effect of diabetes) may not be accurate. In addition, because the nurse who prepared patients’ skin was aware of the study and was supervised in every case, it is possible that the preparation was done more carefully than usual, resulting in more negative cultures than average. Also, compliance in taking preoperative cleanser baths was subjectively determined. Patients may have reported more baths than were actually taken. Still another study limitation is that 2 postsolution isolates did not have an associated presolution isolate. Although we think this may have resulted from laboratory contamination, it is possible the presolution swabs did not accurately determine true bacterial counts in these cases.
Conclusion
A study that showed significant residual bacteria between patients’ toes after chlorhexidine skin preparation5 left us concerned that Chloraprep skin preparation for TKA might not be adequate. The present study showed that this solution was effective in eliminating bacteria from the intertriginous area of the back of the knee in 95% of patients. Skin preparation appears to be less effective in patients with higher BMI.
1. Losina E, Thornhill TS, Rome BN, Wright J, Katz JN. The dramatic increase in total knee replacement utilization rates in the United States cannot be fully explained by growth in population size and the obesity epidemic. J Bone Joint Surg Am. 2012;94(3):201-207.
2. Poultsides LA, Ma Y, Della Valle AG, Chiu YL, Sculco TP, Memtsoudis SG. In-hospital surgical site infections after primary hip and knee arthroplasty—incidence and risk factors. J Arthroplasty. 2013;28(3):385-389.
3. Johnson AJ, Kapadia BH, Daley JA, Molina CB, Mont MA. Chlorhexidine reduces infections in knee arthroplasty. J Knee Surg. 2013;26(3):213-218.
4. Zywiel MG, Daley JA, Delanois RE, Naziri Q, Johnson AJ, Mont MA. Advance pre-operative chlorhexidine reduces the incidence of surgical site infections in knee arthroplasty. Int Orthop. 2011;35(7):1001-1006.
5. Ostrander RV, Botte MJ, Brage ME. Efficacy of surgical preparation solutions in foot and ankle surgery. J Bone Joint Surg Am. 2005;87(5):980-985.
6. Wolters U, Wolf T, Stützer H, Schröder T. ASA classification and perioperative variables as predictors of postoperative outcome. Br J Anaesth. 1996;77(2):217-222.
7. Johnson AJ, Daley JA, Zywiel MG, Delanois RE, Mont MA. Preoperative chlorhexidine preparation and the incidence of surgical site infections after hip arthroplasty. J Arthroplasty. 2010;25(6 suppl):98-102.
8. Samson AJ, Mercer GE, Campbell DG. Total knee replacement in the morbidly obese: a literature review. ANZ J Surg. 2010;80(9):595-599.
9. Iorio R, Williams KM, Marcantonio AJ, Specht LM, Tilzey JF, Healy WL. Diabetes mellitus, hemoglobin A1C, and the incidence of total joint arthroplasty infection. J Arthroplasty. 2012;27(5):726-729.
10. Viens NA, Hug KT, Marchant MH, Cook C, Vail TP, Bolognesi MP. Role of diabetes type in perioperative outcomes after hip and knee arthroplasty in the United States. J Surg Orthop Adv. 2012;21(4):253-260.
11. Webster J, Osborne S. Preoperative bathing or showering with skin antiseptics to prevent surgical site infection. Cochrane Database Syst Rev. 2012;9:CD004985.
1. Losina E, Thornhill TS, Rome BN, Wright J, Katz JN. The dramatic increase in total knee replacement utilization rates in the United States cannot be fully explained by growth in population size and the obesity epidemic. J Bone Joint Surg Am. 2012;94(3):201-207.
2. Poultsides LA, Ma Y, Della Valle AG, Chiu YL, Sculco TP, Memtsoudis SG. In-hospital surgical site infections after primary hip and knee arthroplasty—incidence and risk factors. J Arthroplasty. 2013;28(3):385-389.
3. Johnson AJ, Kapadia BH, Daley JA, Molina CB, Mont MA. Chlorhexidine reduces infections in knee arthroplasty. J Knee Surg. 2013;26(3):213-218.
4. Zywiel MG, Daley JA, Delanois RE, Naziri Q, Johnson AJ, Mont MA. Advance pre-operative chlorhexidine reduces the incidence of surgical site infections in knee arthroplasty. Int Orthop. 2011;35(7):1001-1006.
5. Ostrander RV, Botte MJ, Brage ME. Efficacy of surgical preparation solutions in foot and ankle surgery. J Bone Joint Surg Am. 2005;87(5):980-985.
6. Wolters U, Wolf T, Stützer H, Schröder T. ASA classification and perioperative variables as predictors of postoperative outcome. Br J Anaesth. 1996;77(2):217-222.
7. Johnson AJ, Daley JA, Zywiel MG, Delanois RE, Mont MA. Preoperative chlorhexidine preparation and the incidence of surgical site infections after hip arthroplasty. J Arthroplasty. 2010;25(6 suppl):98-102.
8. Samson AJ, Mercer GE, Campbell DG. Total knee replacement in the morbidly obese: a literature review. ANZ J Surg. 2010;80(9):595-599.
9. Iorio R, Williams KM, Marcantonio AJ, Specht LM, Tilzey JF, Healy WL. Diabetes mellitus, hemoglobin A1C, and the incidence of total joint arthroplasty infection. J Arthroplasty. 2012;27(5):726-729.
10. Viens NA, Hug KT, Marchant MH, Cook C, Vail TP, Bolognesi MP. Role of diabetes type in perioperative outcomes after hip and knee arthroplasty in the United States. J Surg Orthop Adv. 2012;21(4):253-260.
11. Webster J, Osborne S. Preoperative bathing or showering with skin antiseptics to prevent surgical site infection. Cochrane Database Syst Rev. 2012;9:CD004985.
Effect of Day of the Week of Primary Total Hip Arthroplasty on Length of Stay at a University-Based Teaching Medical Center
With health care costs increasing and economic resources diminishing, substantial efforts have been directed toward improving the quality of care delivered in a cost-effective manner. For a total hip arthroplasty (THA) performed in the United States between 1997 and 2001, total hospital cost, including direct and indirect costs, was estimated as averaging $13,339.1 In 2012, this cost was estimated to be between $43,000 and $100,000.2 This overall cost estimate, along with the rate at which the procedure is performed, may present an opportunity for cost savings.
Length of hospital stay (LHS) is an important outcome measure that has been assessed for optimal health care delivery. Prolonged LHS implies increased resource expenditure. Therefore, it is crucial to identify factors associated with prolonged LHS in order to reduce costs. Investigations have identified factors shown to affect LHS after THA. These factors include advanced age, medical comorbidities, obesity, intraoperative time, anesthesia technique, surgical site infection, and incision length.3-7
We conducted a study to identify the patient and clinical factors that affect LHS and to determine whether the specific day of the week when primary THA is performed affects LHS at a large tertiary-care university-based medical center. This information may prove valuable to hospital planning committees allotting operating room time and floor staffing for elective surgical cases with the goal of delivering cost-efficient care.
Materials and Methods
After obtaining institutional review board approval for this study, we retrospectively analyzed all primary unilateral THAs (273 patients) performed at our institution, a tertiary-care teaching hospital, between January 2010 and May 2011. The majority of the surgeries were performed through a posterior approach, and a majority of the implants were uncemented. All patients followed the same postoperative clinical pathway; no fast-track pathway was used.
The combined effects of day of surgery, American Society of Anesthesiologists (ASA) grade, anesthesia type, intraoperative time, estimated blood loss (EBL), incision length, presence of complications, age, sex, body mass index (BMI), disposition (skilled nursing facility vs home), transfusion, hematocrit, and hemoglobin on LHS were analyzed using a multiple quasi-Poisson regression model that included a random effect for surgeon. A Poisson regression model (typically used for count data) was deemed appropriate, as LHS was reported in whole days; a quasi-Poisson model relaxes the Poisson model assumption that the variance in the data equals the mean. The random effect for surgeon adjusts for any correlation among data from surgeries conducted by the same surgeon.
All complications were recorded. Complications included excess wound drainage,8 wound hematoma (a case of excess wound drainage necessitated surgical irrigation and débridement), new-onset atrial fibrillation, non-ST-elevation myocardial infarction, atrial flutter, urinary tract infection, pulmonary embolism, disseminated intravascular coagulation, hepatic decompensation as manifested by elevated liver enzymes, pneumonia, gastroesophageal reflux disease, gastric ulcer, sepsis, delirium, hypotension, and dysphagia.
The parameter estimates reported from the quasi-Poisson regression model are incident rate ratios (IRRs). IRR represents the change in expected LHS for a 1-unit change in a continuous variable (eg, age) or between categories of a categorical variable (eg, sex). IRR higher than 1 indicates higher risk as the continuous variable increases or a higher risk relative to the comparator group for a categorical variable. IRR lower than 1 indicates lower risk.
Results
Table 1 summarizes patient characteristics by surgical day. Mean LHS ranged from a minimum of 3.7 days for patients who had surgery on a Monday to a maximum of 4.2 days for patients who had surgery on a Thursday.
Table 2 summarizes results of the multivariate quasi-Poisson regression analysis of LHS by surgical day, ASA grade, anesthesia type, intraoperative time, EBL, incision length, presence of complications, age, sex, and BMI. With all other variables included in the model adjusted for, each additional point in ASA grade was associated with a 12% increase in LHS (P = .019). In addition, with all other variables included in the model adjusted for, LHS was 33% longer for patients with complications than for patients without complications (P < .001) and 12% longer for patients who received transfusions than for patients who did not (P = .046). LHS did not differ significantly by the day of the week when the surgery was performed (P = .496). Disposition status (skilled nursing facility vs home) as a variable to determine LHS did approach statistical significance (P = .061). As the effect size we were interested in detecting was an approximate 1-day increase in LHS for patients who had surgery later in the week relative to patients who had surgery earlier in the week, our sample size was adequate (range of required sample size, 200-300 patients). This study had 99% power to detect a 27% increase in LHS (equivalent to 1 day or more).
Discussion
This retrospective analysis explored how day of the week of primary THA affected LHS. Various confounders, such as surgery and patient factors, were also examined so that the multivariate analysis would be able to isolate the effects of surgical day of the week on LHS.
Effect of day of the week of primary THA on LHS was not investigated in the United States before. In Denmark, in a study similar to ours, Husted and colleagues4 found a 400% increase in the probability of LHS of more than 3 days when patients operated on a Thursday were compared with patients operated on a Friday. The authors reasoned that the Thursday patients most likely had a compromised physical therapy protocol owing to the inclusion of weekend days in the crucial postoperative period. LHS was consequently increased so that these patients would achieve their therapy goals before being discharged. Our investigation showed that LHS did not differ significantly by surgical day of the week. Although patients who had THA on a Thursday had 15% longer LHS than patients who had THA on a Monday, this difference was not statistically significant (P = .496), even though the study was adequately powered to detect a change in LHS of a whole day.
Table 3 summarizes the difference in quantum of workforce on weekdays and weekends at our center. The physiotherapy sessions were reduced to 1 per day. Nurse practitioners and discharge planners were not available on weekends, and some skilled nursing facilities and rehabilitation centers refused to accept patients on weekends. At our center, a teaching institute, the clinical duties of discharge planners and nurse practitioners were assumed by licensed physicians (orthopedic residents covering the arthroplasty team on weekends). This could be one of several possible reasons our study failed to detect statistically significant difference between the 2 groups. This kind of alternative arrangement may not be possible at many other centers. However, our study results provide a reasonably accurate logistical aim with regard to workforce availability on weekends to keep LHS in check.
The importance of giving patients an inpatient physical therapy regimen in timely fashion has been demonstrated in other studies. Munin and colleagues,9 in a randomized controlled trial, evaluated 71 patients who underwent elective hip and knee arthroplasty and received 2 different physical therapy regimens. Patients started their in-treatment physical therapy on postoperative day 3 or 7. Mean total LHS was shorter in the 3-day group (11.7 days) than in the 7-day group (14.5 days) (P < .001). Brusco and colleagues10 also showed that introducing weekend physical therapy services significantly reduced LHS in patients who underwent THA (10.6 vs 12.5 days; P < .05). Rapoport and Judd-Van Eerd11 retrospectively analyzed orthopedic surgery LHS, comparing patients treated in a community hospital during a period of 5-days-a-week physical therapy coverage and patients treated during a period of 7-days-a-week physical therapy coverage. The 7-days-a-week group had significantly statistically shorter mean LHS.
Another rationale for analyzing the impact of surgical day of the week stems from the expectation that patients who undergo THA on Wednesday or Thursday and are scheduled to have physical therapy or be discharged on the weekend may be affected not only by reduced inpatient weekend physical therapy coverage but also by difficulties in being transferred to a skilled nursing facility or rehabilitation center if not discharged home. In our study, the patients who were to be discharged to a rehabilitation center were delayed by 12.5%, and this statistic trended toward significance (P = .061). Our literature search did not turn up any studies, US or European, specifically linking LHS to discharge disposition (whether patient is discharged home or to a skilled nursing facility or rehabilitation center).
Reduced medical staffing on weekends may not only affect the quality of in-hospital patient care but may also result in unnecessary delays in discharge. Chow and Szeto12 retrospectively analyzed the medical records of all acute medical wards in a university hospital and compared weekend discharge rates before and after implementation of a work ordinance, which decreased the physician workforce by half on Saturday and Sunday. Results showed a 2.7% decrease in the weekend discharge rate after the work ordinance was established. The number of weekday discharges between the 2 time periods did not differ. Increasing the workforce availability presents a challenge in academic medical centers where graduate medical education enforces a strict cap on resident duty hours. Under these circumstances, a more feasible approach to decreasing LHS for THA patients is for surgical planning committees to provide the joint replacement services with operative block times early in the workweek.
Even though the organizational structure at our center is strong enough to provide for an adequate weekend workforce to discharge these patients, this study had a few limitations. We could not study readmission rates and whether the transition to home health and home physical therapy for the patients who went home was seamless.
We found that only 3 patient characteristics had a significant effect on LHS: higher ASA grade (a surrogate for medical comorbidities), requirement for blood transfusion, and presence of complications. In Denmark, blood transfusion increased the likelihood of longer LHS by 400%.4 In that study, patients who were ASA grades 1 and 2 had 60% and 20% decreased likelihood of LHS of more than 3 days compared with patients who were ASA grade 3. Similarly, in 2009, Mears and colleagues5 found 4 factors related to increased LHS: female sex (P < .001), older age (P < .001), higher ASA grade (3, P < .01; 4, P < .001), and increased blood loss (P < .001).5
Conclusion
Over the past decade, there has been a significant reduction in LHS after THA, from a mean of 3 weeks to 4 days. Advances in implant technology, delivery of in-home physical therapy, and improved prevention and management of postoperative complications have contributed to this decline. Early identification of patients with transfusion requirements may be helpful in expediting their care. Although guidelines are in place for transfusion, further study in this regard may be needed. It is important to continue to identify surgery and patient factors that affect LHS, but the importance of organizational and planning issues in optimizing hospital health care expenditures cannot be ignored. Further study of providing a specific discharge planning service to identify patients’ discharge needs (home vs extended care facility) may help reduce LHS.
1. Antoniou J, Martineau PA, Filion KB, et al. In-hospital cost of total hip arthroplasty in Canada and the United States. J Bone Joint Surg Am. 2004;86(11):2435-2439.
2. Kumar S, Breuing R, Chahal R. Globalization of health care delivery in the United States through medical tourism. J Health Commun. 2012;17(2):177-198.
3. Foote J, Panchoo K, Blair P, Bannister G. Length of stay following primary total hip replacement. Ann R Coll Surg Engl. 2009;91(6):500-504.
4. Husted H, Holm G, Jacobsen S. Predictors of length of stay and patient satisfaction after hip and knee replacement surgery: fast-track experience in 712 patients. Acta Orthop. 2008;79(2):168-173.
5. Mears DC, Mears SC, Chelly JE, Dai F, Vulakovich KL. THA with a minimally invasive technique, multi-modal anesthesia, and home rehabilitation: factors associated with early discharge? Clin Orthop. 2009;467(6):1412-1417.
6. Peck CN, Foster A, McLauchlan GJ. Reducing incision length or intensifying rehabilitation: what makes the difference to length of stay in total hip replacement in a UK setting? Int Orthop. 2006;30(5):395-398.
7. Weaver F, Hynes D, Hopkinson W, et al. Preoperative risks and outcomes of hip and knee arthroplasty in the Veterans Health Administration. J Arthroplasty. 2003;18(6):693-708.
8. Patel VP, Walsh M, Sehgal B, Preston C, DeWal H, Di Cesare PE. Factors associated with prolonged wound drainage after primary total hip and knee arthroplasty. J Bone Joint Surg Am. 2007;89(1):33-38.
9. Munin MC, Rudy TE, Glynn NW, Crossett LS, Rubash HE. Early inpatient rehabilitation after elective hip and knee arthroplasty. JAMA. 1998;279(11):847-852.
10. Brusco NK, Shields N, Taylor NF, Paratz J. A Saturday physiotherapy service may decrease length of stay in patients undergoing rehabilitation in hospital: a randomised controlled trial. Aust J Physiother. 2007;53(2):75-81.
11. Rapoport J, Judd-Van Eerd M. Impact of physical therapy weekend coverage on length of stay in an acute care community hospital. Phys Ther. 1989;69(1):32-37.
12. Chow KM, Szeto CC. Impact of enforcing the Labour Ordinance, with 1-in-7-day off for hospital doctors, on weekend hospital discharge rate. J Public Health (Oxf). 2005;27(2):189-191.
With health care costs increasing and economic resources diminishing, substantial efforts have been directed toward improving the quality of care delivered in a cost-effective manner. For a total hip arthroplasty (THA) performed in the United States between 1997 and 2001, total hospital cost, including direct and indirect costs, was estimated as averaging $13,339.1 In 2012, this cost was estimated to be between $43,000 and $100,000.2 This overall cost estimate, along with the rate at which the procedure is performed, may present an opportunity for cost savings.
Length of hospital stay (LHS) is an important outcome measure that has been assessed for optimal health care delivery. Prolonged LHS implies increased resource expenditure. Therefore, it is crucial to identify factors associated with prolonged LHS in order to reduce costs. Investigations have identified factors shown to affect LHS after THA. These factors include advanced age, medical comorbidities, obesity, intraoperative time, anesthesia technique, surgical site infection, and incision length.3-7
We conducted a study to identify the patient and clinical factors that affect LHS and to determine whether the specific day of the week when primary THA is performed affects LHS at a large tertiary-care university-based medical center. This information may prove valuable to hospital planning committees allotting operating room time and floor staffing for elective surgical cases with the goal of delivering cost-efficient care.
Materials and Methods
After obtaining institutional review board approval for this study, we retrospectively analyzed all primary unilateral THAs (273 patients) performed at our institution, a tertiary-care teaching hospital, between January 2010 and May 2011. The majority of the surgeries were performed through a posterior approach, and a majority of the implants were uncemented. All patients followed the same postoperative clinical pathway; no fast-track pathway was used.
The combined effects of day of surgery, American Society of Anesthesiologists (ASA) grade, anesthesia type, intraoperative time, estimated blood loss (EBL), incision length, presence of complications, age, sex, body mass index (BMI), disposition (skilled nursing facility vs home), transfusion, hematocrit, and hemoglobin on LHS were analyzed using a multiple quasi-Poisson regression model that included a random effect for surgeon. A Poisson regression model (typically used for count data) was deemed appropriate, as LHS was reported in whole days; a quasi-Poisson model relaxes the Poisson model assumption that the variance in the data equals the mean. The random effect for surgeon adjusts for any correlation among data from surgeries conducted by the same surgeon.
All complications were recorded. Complications included excess wound drainage,8 wound hematoma (a case of excess wound drainage necessitated surgical irrigation and débridement), new-onset atrial fibrillation, non-ST-elevation myocardial infarction, atrial flutter, urinary tract infection, pulmonary embolism, disseminated intravascular coagulation, hepatic decompensation as manifested by elevated liver enzymes, pneumonia, gastroesophageal reflux disease, gastric ulcer, sepsis, delirium, hypotension, and dysphagia.
The parameter estimates reported from the quasi-Poisson regression model are incident rate ratios (IRRs). IRR represents the change in expected LHS for a 1-unit change in a continuous variable (eg, age) or between categories of a categorical variable (eg, sex). IRR higher than 1 indicates higher risk as the continuous variable increases or a higher risk relative to the comparator group for a categorical variable. IRR lower than 1 indicates lower risk.
Results
Table 1 summarizes patient characteristics by surgical day. Mean LHS ranged from a minimum of 3.7 days for patients who had surgery on a Monday to a maximum of 4.2 days for patients who had surgery on a Thursday.
Table 2 summarizes results of the multivariate quasi-Poisson regression analysis of LHS by surgical day, ASA grade, anesthesia type, intraoperative time, EBL, incision length, presence of complications, age, sex, and BMI. With all other variables included in the model adjusted for, each additional point in ASA grade was associated with a 12% increase in LHS (P = .019). In addition, with all other variables included in the model adjusted for, LHS was 33% longer for patients with complications than for patients without complications (P < .001) and 12% longer for patients who received transfusions than for patients who did not (P = .046). LHS did not differ significantly by the day of the week when the surgery was performed (P = .496). Disposition status (skilled nursing facility vs home) as a variable to determine LHS did approach statistical significance (P = .061). As the effect size we were interested in detecting was an approximate 1-day increase in LHS for patients who had surgery later in the week relative to patients who had surgery earlier in the week, our sample size was adequate (range of required sample size, 200-300 patients). This study had 99% power to detect a 27% increase in LHS (equivalent to 1 day or more).
Discussion
This retrospective analysis explored how day of the week of primary THA affected LHS. Various confounders, such as surgery and patient factors, were also examined so that the multivariate analysis would be able to isolate the effects of surgical day of the week on LHS.
Effect of day of the week of primary THA on LHS was not investigated in the United States before. In Denmark, in a study similar to ours, Husted and colleagues4 found a 400% increase in the probability of LHS of more than 3 days when patients operated on a Thursday were compared with patients operated on a Friday. The authors reasoned that the Thursday patients most likely had a compromised physical therapy protocol owing to the inclusion of weekend days in the crucial postoperative period. LHS was consequently increased so that these patients would achieve their therapy goals before being discharged. Our investigation showed that LHS did not differ significantly by surgical day of the week. Although patients who had THA on a Thursday had 15% longer LHS than patients who had THA on a Monday, this difference was not statistically significant (P = .496), even though the study was adequately powered to detect a change in LHS of a whole day.
Table 3 summarizes the difference in quantum of workforce on weekdays and weekends at our center. The physiotherapy sessions were reduced to 1 per day. Nurse practitioners and discharge planners were not available on weekends, and some skilled nursing facilities and rehabilitation centers refused to accept patients on weekends. At our center, a teaching institute, the clinical duties of discharge planners and nurse practitioners were assumed by licensed physicians (orthopedic residents covering the arthroplasty team on weekends). This could be one of several possible reasons our study failed to detect statistically significant difference between the 2 groups. This kind of alternative arrangement may not be possible at many other centers. However, our study results provide a reasonably accurate logistical aim with regard to workforce availability on weekends to keep LHS in check.
The importance of giving patients an inpatient physical therapy regimen in timely fashion has been demonstrated in other studies. Munin and colleagues,9 in a randomized controlled trial, evaluated 71 patients who underwent elective hip and knee arthroplasty and received 2 different physical therapy regimens. Patients started their in-treatment physical therapy on postoperative day 3 or 7. Mean total LHS was shorter in the 3-day group (11.7 days) than in the 7-day group (14.5 days) (P < .001). Brusco and colleagues10 also showed that introducing weekend physical therapy services significantly reduced LHS in patients who underwent THA (10.6 vs 12.5 days; P < .05). Rapoport and Judd-Van Eerd11 retrospectively analyzed orthopedic surgery LHS, comparing patients treated in a community hospital during a period of 5-days-a-week physical therapy coverage and patients treated during a period of 7-days-a-week physical therapy coverage. The 7-days-a-week group had significantly statistically shorter mean LHS.
Another rationale for analyzing the impact of surgical day of the week stems from the expectation that patients who undergo THA on Wednesday or Thursday and are scheduled to have physical therapy or be discharged on the weekend may be affected not only by reduced inpatient weekend physical therapy coverage but also by difficulties in being transferred to a skilled nursing facility or rehabilitation center if not discharged home. In our study, the patients who were to be discharged to a rehabilitation center were delayed by 12.5%, and this statistic trended toward significance (P = .061). Our literature search did not turn up any studies, US or European, specifically linking LHS to discharge disposition (whether patient is discharged home or to a skilled nursing facility or rehabilitation center).
Reduced medical staffing on weekends may not only affect the quality of in-hospital patient care but may also result in unnecessary delays in discharge. Chow and Szeto12 retrospectively analyzed the medical records of all acute medical wards in a university hospital and compared weekend discharge rates before and after implementation of a work ordinance, which decreased the physician workforce by half on Saturday and Sunday. Results showed a 2.7% decrease in the weekend discharge rate after the work ordinance was established. The number of weekday discharges between the 2 time periods did not differ. Increasing the workforce availability presents a challenge in academic medical centers where graduate medical education enforces a strict cap on resident duty hours. Under these circumstances, a more feasible approach to decreasing LHS for THA patients is for surgical planning committees to provide the joint replacement services with operative block times early in the workweek.
Even though the organizational structure at our center is strong enough to provide for an adequate weekend workforce to discharge these patients, this study had a few limitations. We could not study readmission rates and whether the transition to home health and home physical therapy for the patients who went home was seamless.
We found that only 3 patient characteristics had a significant effect on LHS: higher ASA grade (a surrogate for medical comorbidities), requirement for blood transfusion, and presence of complications. In Denmark, blood transfusion increased the likelihood of longer LHS by 400%.4 In that study, patients who were ASA grades 1 and 2 had 60% and 20% decreased likelihood of LHS of more than 3 days compared with patients who were ASA grade 3. Similarly, in 2009, Mears and colleagues5 found 4 factors related to increased LHS: female sex (P < .001), older age (P < .001), higher ASA grade (3, P < .01; 4, P < .001), and increased blood loss (P < .001).5
Conclusion
Over the past decade, there has been a significant reduction in LHS after THA, from a mean of 3 weeks to 4 days. Advances in implant technology, delivery of in-home physical therapy, and improved prevention and management of postoperative complications have contributed to this decline. Early identification of patients with transfusion requirements may be helpful in expediting their care. Although guidelines are in place for transfusion, further study in this regard may be needed. It is important to continue to identify surgery and patient factors that affect LHS, but the importance of organizational and planning issues in optimizing hospital health care expenditures cannot be ignored. Further study of providing a specific discharge planning service to identify patients’ discharge needs (home vs extended care facility) may help reduce LHS.
With health care costs increasing and economic resources diminishing, substantial efforts have been directed toward improving the quality of care delivered in a cost-effective manner. For a total hip arthroplasty (THA) performed in the United States between 1997 and 2001, total hospital cost, including direct and indirect costs, was estimated as averaging $13,339.1 In 2012, this cost was estimated to be between $43,000 and $100,000.2 This overall cost estimate, along with the rate at which the procedure is performed, may present an opportunity for cost savings.
Length of hospital stay (LHS) is an important outcome measure that has been assessed for optimal health care delivery. Prolonged LHS implies increased resource expenditure. Therefore, it is crucial to identify factors associated with prolonged LHS in order to reduce costs. Investigations have identified factors shown to affect LHS after THA. These factors include advanced age, medical comorbidities, obesity, intraoperative time, anesthesia technique, surgical site infection, and incision length.3-7
We conducted a study to identify the patient and clinical factors that affect LHS and to determine whether the specific day of the week when primary THA is performed affects LHS at a large tertiary-care university-based medical center. This information may prove valuable to hospital planning committees allotting operating room time and floor staffing for elective surgical cases with the goal of delivering cost-efficient care.
Materials and Methods
After obtaining institutional review board approval for this study, we retrospectively analyzed all primary unilateral THAs (273 patients) performed at our institution, a tertiary-care teaching hospital, between January 2010 and May 2011. The majority of the surgeries were performed through a posterior approach, and a majority of the implants were uncemented. All patients followed the same postoperative clinical pathway; no fast-track pathway was used.
The combined effects of day of surgery, American Society of Anesthesiologists (ASA) grade, anesthesia type, intraoperative time, estimated blood loss (EBL), incision length, presence of complications, age, sex, body mass index (BMI), disposition (skilled nursing facility vs home), transfusion, hematocrit, and hemoglobin on LHS were analyzed using a multiple quasi-Poisson regression model that included a random effect for surgeon. A Poisson regression model (typically used for count data) was deemed appropriate, as LHS was reported in whole days; a quasi-Poisson model relaxes the Poisson model assumption that the variance in the data equals the mean. The random effect for surgeon adjusts for any correlation among data from surgeries conducted by the same surgeon.
All complications were recorded. Complications included excess wound drainage,8 wound hematoma (a case of excess wound drainage necessitated surgical irrigation and débridement), new-onset atrial fibrillation, non-ST-elevation myocardial infarction, atrial flutter, urinary tract infection, pulmonary embolism, disseminated intravascular coagulation, hepatic decompensation as manifested by elevated liver enzymes, pneumonia, gastroesophageal reflux disease, gastric ulcer, sepsis, delirium, hypotension, and dysphagia.
The parameter estimates reported from the quasi-Poisson regression model are incident rate ratios (IRRs). IRR represents the change in expected LHS for a 1-unit change in a continuous variable (eg, age) or between categories of a categorical variable (eg, sex). IRR higher than 1 indicates higher risk as the continuous variable increases or a higher risk relative to the comparator group for a categorical variable. IRR lower than 1 indicates lower risk.
Results
Table 1 summarizes patient characteristics by surgical day. Mean LHS ranged from a minimum of 3.7 days for patients who had surgery on a Monday to a maximum of 4.2 days for patients who had surgery on a Thursday.
Table 2 summarizes results of the multivariate quasi-Poisson regression analysis of LHS by surgical day, ASA grade, anesthesia type, intraoperative time, EBL, incision length, presence of complications, age, sex, and BMI. With all other variables included in the model adjusted for, each additional point in ASA grade was associated with a 12% increase in LHS (P = .019). In addition, with all other variables included in the model adjusted for, LHS was 33% longer for patients with complications than for patients without complications (P < .001) and 12% longer for patients who received transfusions than for patients who did not (P = .046). LHS did not differ significantly by the day of the week when the surgery was performed (P = .496). Disposition status (skilled nursing facility vs home) as a variable to determine LHS did approach statistical significance (P = .061). As the effect size we were interested in detecting was an approximate 1-day increase in LHS for patients who had surgery later in the week relative to patients who had surgery earlier in the week, our sample size was adequate (range of required sample size, 200-300 patients). This study had 99% power to detect a 27% increase in LHS (equivalent to 1 day or more).
Discussion
This retrospective analysis explored how day of the week of primary THA affected LHS. Various confounders, such as surgery and patient factors, were also examined so that the multivariate analysis would be able to isolate the effects of surgical day of the week on LHS.
Effect of day of the week of primary THA on LHS was not investigated in the United States before. In Denmark, in a study similar to ours, Husted and colleagues4 found a 400% increase in the probability of LHS of more than 3 days when patients operated on a Thursday were compared with patients operated on a Friday. The authors reasoned that the Thursday patients most likely had a compromised physical therapy protocol owing to the inclusion of weekend days in the crucial postoperative period. LHS was consequently increased so that these patients would achieve their therapy goals before being discharged. Our investigation showed that LHS did not differ significantly by surgical day of the week. Although patients who had THA on a Thursday had 15% longer LHS than patients who had THA on a Monday, this difference was not statistically significant (P = .496), even though the study was adequately powered to detect a change in LHS of a whole day.
Table 3 summarizes the difference in quantum of workforce on weekdays and weekends at our center. The physiotherapy sessions were reduced to 1 per day. Nurse practitioners and discharge planners were not available on weekends, and some skilled nursing facilities and rehabilitation centers refused to accept patients on weekends. At our center, a teaching institute, the clinical duties of discharge planners and nurse practitioners were assumed by licensed physicians (orthopedic residents covering the arthroplasty team on weekends). This could be one of several possible reasons our study failed to detect statistically significant difference between the 2 groups. This kind of alternative arrangement may not be possible at many other centers. However, our study results provide a reasonably accurate logistical aim with regard to workforce availability on weekends to keep LHS in check.
The importance of giving patients an inpatient physical therapy regimen in timely fashion has been demonstrated in other studies. Munin and colleagues,9 in a randomized controlled trial, evaluated 71 patients who underwent elective hip and knee arthroplasty and received 2 different physical therapy regimens. Patients started their in-treatment physical therapy on postoperative day 3 or 7. Mean total LHS was shorter in the 3-day group (11.7 days) than in the 7-day group (14.5 days) (P < .001). Brusco and colleagues10 also showed that introducing weekend physical therapy services significantly reduced LHS in patients who underwent THA (10.6 vs 12.5 days; P < .05). Rapoport and Judd-Van Eerd11 retrospectively analyzed orthopedic surgery LHS, comparing patients treated in a community hospital during a period of 5-days-a-week physical therapy coverage and patients treated during a period of 7-days-a-week physical therapy coverage. The 7-days-a-week group had significantly statistically shorter mean LHS.
Another rationale for analyzing the impact of surgical day of the week stems from the expectation that patients who undergo THA on Wednesday or Thursday and are scheduled to have physical therapy or be discharged on the weekend may be affected not only by reduced inpatient weekend physical therapy coverage but also by difficulties in being transferred to a skilled nursing facility or rehabilitation center if not discharged home. In our study, the patients who were to be discharged to a rehabilitation center were delayed by 12.5%, and this statistic trended toward significance (P = .061). Our literature search did not turn up any studies, US or European, specifically linking LHS to discharge disposition (whether patient is discharged home or to a skilled nursing facility or rehabilitation center).
Reduced medical staffing on weekends may not only affect the quality of in-hospital patient care but may also result in unnecessary delays in discharge. Chow and Szeto12 retrospectively analyzed the medical records of all acute medical wards in a university hospital and compared weekend discharge rates before and after implementation of a work ordinance, which decreased the physician workforce by half on Saturday and Sunday. Results showed a 2.7% decrease in the weekend discharge rate after the work ordinance was established. The number of weekday discharges between the 2 time periods did not differ. Increasing the workforce availability presents a challenge in academic medical centers where graduate medical education enforces a strict cap on resident duty hours. Under these circumstances, a more feasible approach to decreasing LHS for THA patients is for surgical planning committees to provide the joint replacement services with operative block times early in the workweek.
Even though the organizational structure at our center is strong enough to provide for an adequate weekend workforce to discharge these patients, this study had a few limitations. We could not study readmission rates and whether the transition to home health and home physical therapy for the patients who went home was seamless.
We found that only 3 patient characteristics had a significant effect on LHS: higher ASA grade (a surrogate for medical comorbidities), requirement for blood transfusion, and presence of complications. In Denmark, blood transfusion increased the likelihood of longer LHS by 400%.4 In that study, patients who were ASA grades 1 and 2 had 60% and 20% decreased likelihood of LHS of more than 3 days compared with patients who were ASA grade 3. Similarly, in 2009, Mears and colleagues5 found 4 factors related to increased LHS: female sex (P < .001), older age (P < .001), higher ASA grade (3, P < .01; 4, P < .001), and increased blood loss (P < .001).5
Conclusion
Over the past decade, there has been a significant reduction in LHS after THA, from a mean of 3 weeks to 4 days. Advances in implant technology, delivery of in-home physical therapy, and improved prevention and management of postoperative complications have contributed to this decline. Early identification of patients with transfusion requirements may be helpful in expediting their care. Although guidelines are in place for transfusion, further study in this regard may be needed. It is important to continue to identify surgery and patient factors that affect LHS, but the importance of organizational and planning issues in optimizing hospital health care expenditures cannot be ignored. Further study of providing a specific discharge planning service to identify patients’ discharge needs (home vs extended care facility) may help reduce LHS.
1. Antoniou J, Martineau PA, Filion KB, et al. In-hospital cost of total hip arthroplasty in Canada and the United States. J Bone Joint Surg Am. 2004;86(11):2435-2439.
2. Kumar S, Breuing R, Chahal R. Globalization of health care delivery in the United States through medical tourism. J Health Commun. 2012;17(2):177-198.
3. Foote J, Panchoo K, Blair P, Bannister G. Length of stay following primary total hip replacement. Ann R Coll Surg Engl. 2009;91(6):500-504.
4. Husted H, Holm G, Jacobsen S. Predictors of length of stay and patient satisfaction after hip and knee replacement surgery: fast-track experience in 712 patients. Acta Orthop. 2008;79(2):168-173.
5. Mears DC, Mears SC, Chelly JE, Dai F, Vulakovich KL. THA with a minimally invasive technique, multi-modal anesthesia, and home rehabilitation: factors associated with early discharge? Clin Orthop. 2009;467(6):1412-1417.
6. Peck CN, Foster A, McLauchlan GJ. Reducing incision length or intensifying rehabilitation: what makes the difference to length of stay in total hip replacement in a UK setting? Int Orthop. 2006;30(5):395-398.
7. Weaver F, Hynes D, Hopkinson W, et al. Preoperative risks and outcomes of hip and knee arthroplasty in the Veterans Health Administration. J Arthroplasty. 2003;18(6):693-708.
8. Patel VP, Walsh M, Sehgal B, Preston C, DeWal H, Di Cesare PE. Factors associated with prolonged wound drainage after primary total hip and knee arthroplasty. J Bone Joint Surg Am. 2007;89(1):33-38.
9. Munin MC, Rudy TE, Glynn NW, Crossett LS, Rubash HE. Early inpatient rehabilitation after elective hip and knee arthroplasty. JAMA. 1998;279(11):847-852.
10. Brusco NK, Shields N, Taylor NF, Paratz J. A Saturday physiotherapy service may decrease length of stay in patients undergoing rehabilitation in hospital: a randomised controlled trial. Aust J Physiother. 2007;53(2):75-81.
11. Rapoport J, Judd-Van Eerd M. Impact of physical therapy weekend coverage on length of stay in an acute care community hospital. Phys Ther. 1989;69(1):32-37.
12. Chow KM, Szeto CC. Impact of enforcing the Labour Ordinance, with 1-in-7-day off for hospital doctors, on weekend hospital discharge rate. J Public Health (Oxf). 2005;27(2):189-191.
1. Antoniou J, Martineau PA, Filion KB, et al. In-hospital cost of total hip arthroplasty in Canada and the United States. J Bone Joint Surg Am. 2004;86(11):2435-2439.
2. Kumar S, Breuing R, Chahal R. Globalization of health care delivery in the United States through medical tourism. J Health Commun. 2012;17(2):177-198.
3. Foote J, Panchoo K, Blair P, Bannister G. Length of stay following primary total hip replacement. Ann R Coll Surg Engl. 2009;91(6):500-504.
4. Husted H, Holm G, Jacobsen S. Predictors of length of stay and patient satisfaction after hip and knee replacement surgery: fast-track experience in 712 patients. Acta Orthop. 2008;79(2):168-173.
5. Mears DC, Mears SC, Chelly JE, Dai F, Vulakovich KL. THA with a minimally invasive technique, multi-modal anesthesia, and home rehabilitation: factors associated with early discharge? Clin Orthop. 2009;467(6):1412-1417.
6. Peck CN, Foster A, McLauchlan GJ. Reducing incision length or intensifying rehabilitation: what makes the difference to length of stay in total hip replacement in a UK setting? Int Orthop. 2006;30(5):395-398.
7. Weaver F, Hynes D, Hopkinson W, et al. Preoperative risks and outcomes of hip and knee arthroplasty in the Veterans Health Administration. J Arthroplasty. 2003;18(6):693-708.
8. Patel VP, Walsh M, Sehgal B, Preston C, DeWal H, Di Cesare PE. Factors associated with prolonged wound drainage after primary total hip and knee arthroplasty. J Bone Joint Surg Am. 2007;89(1):33-38.
9. Munin MC, Rudy TE, Glynn NW, Crossett LS, Rubash HE. Early inpatient rehabilitation after elective hip and knee arthroplasty. JAMA. 1998;279(11):847-852.
10. Brusco NK, Shields N, Taylor NF, Paratz J. A Saturday physiotherapy service may decrease length of stay in patients undergoing rehabilitation in hospital: a randomised controlled trial. Aust J Physiother. 2007;53(2):75-81.
11. Rapoport J, Judd-Van Eerd M. Impact of physical therapy weekend coverage on length of stay in an acute care community hospital. Phys Ther. 1989;69(1):32-37.
12. Chow KM, Szeto CC. Impact of enforcing the Labour Ordinance, with 1-in-7-day off for hospital doctors, on weekend hospital discharge rate. J Public Health (Oxf). 2005;27(2):189-191.
Metal Ion Levels in Maternal and Placental Blood After Metal-on-Metal Total Hip Arthroplasty
Serum metal ion levels are increased after primary total hip arthroplasty (THA) with all types of head-liner bearing surfaces.1-4 In recent years, there has been increasing concern regarding elevated metal ion levels and adverse local and systemic effects, in particular, from metal-on-metal (MOM) implants.5 There have been reports of adverse local tissue reactions (ALTR) and systemic effects associated with elevated metal ion levels from MOM THA.6-10 MOM hip resurfacings have been advocated in the literature for use in select patient populations, such as young, active individuals.11,12 Purported benefits of MOM-bearing surfaces in this patient population include reduced wear and the prevention of osteolysis.13 While the incidence of ALTR has been reported to be approximately 1% within 5 years after MOM hip resurfacing, the prevalence of ALTR at long-term follow-up is unknown.14 Adverse local tissue reactions in hip resurfacing are related in part to femoral head coverage by the acetabular cup, as well as component design and orientation. The risk for ALTR with MOM THA has been reported to correlate with the level of serum metal ion levels because of the bearing surface, along with taper corrosion and corrosion secondary to the large surface area of the femoral head.13-15 The overall clinical and prognostic value of metal ion levels in cases of MOM bearings remains controversial and without clear cut-off values.15
The long-term physiologic response to elevated serum metal ion levels from THA implants remains unknown and is of particular concern in pregnant women because of potential transplacental transfer. Several studies have shown that DNA (deoxyribonucleic acid) and chromosomal changes can occur in patients with both MOM and non-MOM hip implants.16-18 Techniques to accurately measure the levels of metal ions, including cobalt, chromium, and titanium in the serum, have improved substantially in recent years and have been used successfully in clinical applications with low detection limits.2,4,19,20 Evidence shows that pregnancy-related complications in women with well-functioning cemented, hybrid, or uncemented metal-on-polyethylene THA are not different from those in healthy women.21 However, it is unclear if metal ions can cross the placenta and negatively affect the development of a fetus in mothers with MOM-bearing THAs.22 We conducted this study to determine if metal ions can traverse the placenta by measuring serum metal ion concentrations in mothers with and without MOM THA and the corresponding levels in umbilical cord blood samples.
Materials and Methods
Ten patients were prospectively enrolled in this study, which was approved by the institutional review boards at all 3 participating hospitals prior to initiation. All patients provided informed consent and agreed to maternal and umbilical cord blood sampling. Seven of the 10 patients did not have any metallic implants and served as controls. The remaining 3 patients had either a MOM unilateral primary THA (1 patient) or unilateral MOM hip-resurfacing arthroplasty (2 patients) with no other metal implants in the body. For all subjects, maternal and umbilical cord blood was obtained at the time of delivery.
Group Demographics
The 3 women in the implant group had a mean age of 32.3 years (range, 23-39 years) at time of delivery. The first patient had posttraumatic osteoarthritis and underwent right THA using a modular MOM 60-mm acetabular component, a 36-mm cobalt chrome head, and a cementless, titanium proximally porous-coated femoral component (Pinnacle Total Hip System and Summit Total Hip System; DePuy Synthes, Warsaw, Indiana). Her infant was born 2 years after index THA. The second patient had a diagnosis of degenerative osteoarthritis secondary to developmental dysplasia of the hip and underwent a hybrid MOM hip resurfacing with a monoblock 50-mm cup and 40-mm cobalt chrome head (Conserve Plus; Wright Medical Technology, Arlington, Tennessee). She gave birth to her infant 6 years after her hip resurfacing. The third patient also had a diagnosis of degenerative osteoarthritis secondary to developmental dysplasia of the hip and underwent hip resurfacing with a 54-mm monoblock cup and 44-mm cobalt chrome head (Conserve Plus). Her infant was born 4 years after her resurfacing arthroplasty. All of the infants were born healthy, and the deliveries were uneventful and without complications. Seven women with a mean age of 32.1 years (range, 24-37 years) and their infants served as controls at the time of delivery. None of the women in the control group had a history of renal impairment, inherited genetic disorders, or metal implants.
Serum Metal Ion Analysis
Blood samples were collected using S-Monovette polypropylene syringes (Sarstedt, Princeton, New Jersey), a multi-adapter, and infusion set following an established technique.1 All vessels and utensils used for specimen collection were verified to be free of metal contamination. Three 10-mL syringes were drawn, and each syringe was labeled to indicate the sequence of collection. The first 10 mL were drawn to rinse the needle and adapter. Blood was then allowed to clot naturally and centrifuged at 1850 rpm for 30 minutes, separating samples into cell and serum fractions that were stored in labeled vials at -80ºC. All specimen manipulations after collection were carried out in a class-100 environment using a SterilGARD Biological Safety Cabinet (Baker, Sanford, Maine) and class-100 gloves (Oak Technical, Ravenna, Ohio) to minimize atmospheric and manual contamination. Maternal and umbilical cord blood metal ion levels were tested using high-resolution sector-field inductively-coupled plasma-mass spectrometry (HR-SF-ICPMS) (Element 2; Thermo Fisher Scientific, Bremen, Germany) following the method of additions as described previously.23 The HR-SF-ICPMS machine was equipped with an SC-E2 autosampler, Teflon nebulizer and spray chamber, sapphire injector (Elemental Scientific, Omaha, Nebraska) and platinum cones. All calibration and internal standard solutions were prepared by gradual dilutions of single-element standard solutions (1000 μg/mL from High Purity Standards, Charleston, South Carolina). The certified reference material, Seronorm Trace Elements Serum (SERO, Billingstad, Norway), were routinely analyzed with samples. The serum concentrations of cobalt, chromium, titanium, and nickel were measured with detection limits in ng/mL (parts per billion) of 0.04 for cobalt, 0.015 for chromium, 0.2 for titanium, and 0.17 for nickel.4 Concentrations below the detection limit were approximated as one-half of the detection limit by convention to calculate means.
Statistical Analysis
The data reported are the means for each group for each of the metal ion levels analyzed. Intergroup comparisons were made with the Mann-Whitney-Wilcoxon test using SPSS statistics software (SPSS Science Inc, Chicago, Illinois) to compare implant and control groups in regards to serum metal ion levels. Intragroup comparisons were made using the Friedman test with significance set at P < .05. Spearman rank-order correlation tests were used to investigate relationships between maternal and infant serum metal ion levels.
Results
The Table shows the mean serum metal ion levels of chromium, cobalt, titanium, and nickel for both groups. The implant-group mothers had significantly higher chromium and cobalt levels than the control-group mothers, with mean chromium levels of 1.87 ng/mL vs 0.16 ng/mL (P = .01) and mean cobalt levels of 0.97 ng/mL vs 0.20 ng/mL (P = .01), respectively. All control-group maternal chromium and cobalt levels were lower than the implant group. There were no significant differences detected between the implant-group and control-group mothers with respect to serum titanium or nickel levels.
The implant-group cord blood samples also had significantly higher chromium and cobalt levels than did the control-group infants with mean chromium levels of 0.29 ng/mL vs 0.10 ng/mL (P = .03) and mean cobalt levels of 0.49 ng/mL vs 0.16 ng/mL (P = .01), respectively. All but 1 of the control-group infants had chromium levels that were lower than the implant-group infant chromium levels. All of the control-group infant cobalt levels were lower than the implant-group infant cobalt levels. In the mother (I-1) who had a titanium-containing implant, her titanium level was 1.77 ng/mL and her cord blood level was 0.78 ng/mL. In contrast, the other 2 patients did not have titanium-containing implants and had corresponding metal levels of 0.10 ng/mL and cord blood levels either below the detection limit or just slightly above it. No statistically significant differences were found between the implant- and control-group infants with respect to serum titanium or nickel levels.
Considering the implant and control groups separately, we found no statistically significant differences between the maternal and infant titanium levels or the maternal and infant nickel levels. In the implant group, the mother’s chromium level was always higher than her infant’s chromium level, and the mother’s cobalt level was always higher than her infant’s cobalt level (P= .08). In the control group, there was no correlation between the maternal and infant chromium levels, or between the maternal and infant cobalt levels. In the implant group, the maternal and infant chromium levels were highly correlated (r = 1), as were the maternal and infant cobalt levels (r = 1).
When infants’ chromium levels were expressed as a percentage of their mothers’ chromium levels, the mean was 15.4% (range, 12.3%-18%) for the implant group and 58.9% (range, 13.5%-165.7%) for the control group (P = .05). The mean infant cobalt level, expressed as percentage of maternal cobalt level, was 50% (range, 45.8%-53.1%) for the implant group and 76.6% (range, 59.1%-150.8%) for the control group (P = .01).
Discussion
Cobalt and chromium serum metal ion levels obtained from mothers with MOM implants and umbilical cord samples were significantly elevated in comparison with controls. There was also a strong correlation between implant-group maternal cobalt and chromium levels and implant-group infant cobalt and chromium levels; however, no such correlation existed in the control group. Our results suggest that cobalt and chromium cross the placental barrier. Implant mothers had higher chromium and cobalt levels compared with implant babies, and these babies had approximately 15% of the level of chromium and 50% of the level of cobalt when compared to implant maternal levels. This finding suggests that the placenta modulates the transfer of chromium and cobalt to the fetus.
Two studies have reported chromium and cobalt levels in maternal and umbilical cord sera after MOM THA.5,24 Brodner and colleagues5 determined the maternal serum levels of cobalt and chromium in 3 women 3.8 years after MOM THA and compared those to cobalt and chromium levels obtained from umbilical cord blood. At the time of delivery, the maternal chromium concentrations in the 3 patients were 1.6 ng/mL, 0.5 ng/mL, and 0.9 ng/mL, and the cobalt concentrations were 1 ng/mL in 1 patient and below the detection limit in the other 2 patients. Cobalt and chromium concentrations of the 3 umbilical cord sera were below the detection limit. The authors concluded that cobalt and chromium did not cross the placenta based on their laboratory detection limits. Metal ion levels were measured using atomic absorption spectrometry with relatively high detection limits of 0.3 ng/mL, which is not as sensitive as the HR-SF-ICPMS technique used in the present study that has detection limits of 0.04 ng/mL for cobalt and 0.015 ng/mL for chromium. The relatively high detection limits of atomic absorption spectrometry were likely responsible for the authors’ inability to detect elevated chromium and cobalt levels in umbilical cord sera.
Ziaee and colleagues24 used HR-SF-ICPMS, as we did in this study, to measure the mean concentrations of cobalt and chromium ion levels in 10 maternal and umbilical cord blood samples in women with Birmingham MOM hip-resurfacing prostheses (Smith & Nephew, Warwick, United Kingdom). Nine of those patients had a unilateral resurfacing and 1 patient had bilateral-resurfacing prostheses. The mean maternal age was 31 years, and mean duration between hip resurfacing and delivery was 53 months. Ten normal controls were also tested with a mean maternal age of 30.9 years. The authors found that the mean cord blood level of cobalt in the study patients was 0.83 ng/mL, significantly higher (P < .01) than cobalt levels in the control group, which measured 0.33 ng/mL. The mean cord blood levels of chromium in the study and control groups were 0.37 ng/mL and 0.19 ng/mL, respectively. No children were reported to have evidence of congenital anomalies. Similar to our findings, they noted a modulatory effect on the transfer of metal ions across the placenta in patients with MOM prostheses.24 They reported the relative levels of cobalt and chromium in offspring to be 60.4% and 29.4% of the maternal ion levels, respectively. Control-group infants had mean cobalt and chromium levels that were 98.5% and 97.2% of mean maternal levels, respectively.
The transfer of metal ions across the placenta in control subjects is an expected finding because cobalt and chromium are essential trace elements required by the developing fetus. Rudge and colleagues25 estimated a transplacental transfer rate of 45% for cobalt in a series of 62 paired samples of maternal and cord blood. DeSouza and coauthors26 reported a series of 3 patients with MOM hip resurfacings who had the prosthesis in situ during pregnancy and found no teratogenic effects of metal ion transfer across the placenta. Umbilical cord blood chromium levels were less than 25% of the maternal serum levels and cord blood cobalt levels were approximately 50% that of maternal blood.26 In an animal experiment, Wallach and Verch18 also reported that maternal chromium levels can be decreased because of placental uptake.
Ziaee and colleagues24 tested metal ion levels using whole blood in contrast to serum, as we did in the present study. Daniel and coauthors16, who reported on the validity of serum levels as a surrogate measure of systemic exposure to metal ions in hip replacement, suggested that serum and whole blood metal ion levels cannot be interconverted because metal within cells are not in dynamic equilibrium with extracellular levels. They concluded that serum metal ion concentrations are not a useful surrogate measure of systemic metal ion exposure based on the wide variability seen in normalized and Bland-Altman scatterplots.16 However, it is important to note that Bland-Altman plots are user-dependent in determining significance, and results can vary based on the parameters tested. A high correlation does not automatically imply that there is a good agreement between 2 methods because a widespread sample could influence results. Whole blood analysis requires more processing steps, thus providing an increased chance of contamination and variability compared with serum metal level analysis. In our experiences, serum metal ion analysis has been shown to have accurate and reproducible results in clinical situations.2,4
While there is insufficient literature that specifically studies the effects of elevated metal ion levels on maternal and fetal subjects, there have been no reported negative effects in human babies even when maternal ion levels are elevated enough to be associated with ALTR. A case report by Fritzsche and colleagues27 reported a mother with bilateral MOM THA, a recurrent pseudotumor, and high blood levels of chromium (39 ng/mL) and cobalt (138 ng/mL) at 12 weeks gestation. The child was born at 38 weeks gestation with cord blood chromium and cobalt levels of 2.1 ng/mL and 75 ng/mL, respectively. The infant’s metal ion levels remained elevated at age 8 weeks with a chromium level of 2.5 ng/mL and cobalt level of 13 ng/mL and no signs of toxicity by age 14 weeks. In an animal model, Saxena and colleagues28 found that chromium in the hexavalent form passed through the placenta in mice and rats that were fed high doses of potassium dichromate. Trivalent chromium was not found to cross the placenta. In a follow-up study, Junaid and coauthors29 investigated the effects of elevated chromium levels in female mice given potassium dichromate in drinking water on days 14 to 19 of pregnancy. Animals receiving high-dose chromium had significantly higher incidences of postimplantation loss along with subdermal hemorrhagic patches and reduced ossification.29 Cobalt has not been shown to be teratogenic or cause fetotoxicity in a rat animal model given daily doses of as much as 100 mg/kg cobalt (II) chloride on days 6 to 15 of gestation.30
It is important to recognize that rodent data are limited and may not provide accurate translational insight into the effects of metal ions in human maternal and fetal subjects. Mammalian species have significant heterogeneity in the structure and function of their placentas. Rurak31 has shown that rodents have an additional persisting yolk sac placenta that allows the transfer of maternal immunoglobulins to the fetus. Humans, on the other hand, have a yolk sac placenta that regresses early in pregnancy. Differing placental biologic function makes it difficult to extrapolate the effects of metal ions in rodents to human subjects.
It is also important to note that serum levels of cobalt, chromium, and titanium can remain persistently elevated in well-functioning metal-on-polyethylene THA for several years and that elevated metal ion levels are not confined to MOM bearings.2 Levine and colleagues4 reported that serum levels of cobalt, chromium, and titanium remain persistently elevated after 10 years in a cohort of 27 well-functioning primary metal-on-polyethylene THA (hybrid, cobalt-chrome, titanium). Cobalt concentrations were elevated in all implants compared with controls at all follow-up periods through 10 years with absolute values less than 1 ng/mL. The authors noted that metal release at the modular femoral head-neck junctions was likely the dominant source of serum cobalt and chromium rather than passive dissolution. Hsu and colleagues32 have also shown that patients undergoing a second metal-on-polyethylene THA after primary THA have elevated serum metal ion levels (cobalt, chromium, titanium) up to 6 years after second surgery. Reported cobalt concentrations in patients with unilateral THA reached a maximum of 0.5 ng/mL during the follow-up course compared with 1.5 ng/mL for patients with bilateral THA. It is unknown what the potential metal ion transfer load would be in mothers with metal-on-polyethylene THA and associated taper corrosion to their infants.
Conclusion
Mothers with MOM-bearing implants and their children have higher cobalt and chromium levels than control subjects, demonstrating that the placenta is not a complete barrier to metal ion transport, although it seems to have a modulating effect. Physicians and women of child-bearing age should be aware of these findings when considering the use of MOM-bearing couples for THA. The effects of metal ions on long-term maternal and fetal health require research through serial clinical exams and metal ion level testing in prospective studies of different THA-bearing surfaces.
1. Jacobs JJ, Skipor AK, Black J, Urban R, Galante JO. Release and excretion of metal in patients who have a total hip-replacement component made of titanium-base alloy. J Bone Joint Surg Am. 1991;73(10):1475-1486.
2. Jacobs JJ, Skipor AK, Patterson LM, et al. Metal release in patients who have had a primary total hip arthroplasty. A prospective, controlled, longitudinal study. J Bone Joint Surg Am. 1998;80(10):1447-1458.
3. Jacobs JJ, Skipor AK, Urban RM, Black J, Manion LM, Galante JO. Transport of metal degradation products of titanium alloy total hip replacements to reticuloendothelial organs. An autopsy study. Trans Soc Biomater. 1994;18:318-325.
4. Levine BR, Hsu AR, Skipor AK, et al. Ten-year outcome of serum metal ion levels after primary total hip arthroplasty: a concise follow-up of a previous report. J Bone Joint Surg Am. 2013;95(6):512-518.
5. Brodner W, Grohs JG, Bancher-Todesca D, et al. Does the placenta inhibit the passage of chromium and cobalt after metal-on-metal total hip arthroplasty? J Arthroplasty. 2004;19(8 suppl 3):102-106.
6. Hsu AR, Gross CE, Levine BR. Pseudotumor from modular neck corrosion after ceramic-on-polyethylene total hip arthroplasty. Am J Orthop. 2012;41(9):422-426.
7. Tower SS. Arthroprosthetic cobaltism: neurological and cardiac manifestations in two patients with metal-on-metal arthroplasty: a case report. J Bone Joint Surg Am. 2010;92(17):2847-2851.
8. Urban RM, Jacobs JJ, Tomlinson MJ, Gavrilovic J, Black J, Peoc’h M. Dissemination of wear particles to the liver, spleen, and abdominal lymph nodes of patients with hip or knee replacement. J Bone Joint Surg Am. 2000;82(4):457-476.
9. Watters TS, Eward WC, Hallows RK, Dodd LG, Wellman SS, Bolognesi MP. Pseudotumor with superimposed periprosthetic infection following metal-on-metal total hip arthroplasty: a case report. J Bone Joint Surg Am. 2010;92(7):1666-1669.
10. Willert HG, Buchhorn GH, Fayyazi A, et al. Metal-on-metal bearings and hypersensitivity in patients with artificial hip joints. A clinical and histomorphological study. J Bone Joint Surg Am. 2005;87(1):28-36.
11. Nunley RM, Della Valle CJ, Barrack RL. Is patient selection important for hip resurfacing? Clin Orthop. 2009;467(1):56-65.
12. Treacy RB, McBryde CW, Pynsent PB. Birmingham hip resurfacing arthroplasty. A minimum follow-up of five years. J Bone Joint Surg Br. 2005;87(2):167-170.
13. Dorr LD, Wan Z, Longjohn DB, Dubois B, Murken R. Total hip arthroplasty with use of the Metasul metal-on-metal articulation. Four to seven-year results. J Bone Joint Surg Am. 2000;82(6):789-798.
14. Pandit H, Glyn-Jones S, McLardy-Smith P, et al. Pseudotumours associated with metal-on-metal hip resurfacings. J Bone Joint Surg Br. 2008;90(7):847-851.
15. Hartmann A, Hannemann F, Lutzner J, et al. Metal ion concentrations in body fluids after implantation of hip replacements with metal-on-metal bearing--systematic review of clinical and epidemiological studies. PLoS One. 2013;8(8):e70359.
16. Daniel J, Ziaee H, Pynsent PB, McMinn DJ. The validity of serum levels as a surrogate measure of systemic exposure to metal ions in hip replacement. J Bone Joint Surg Br. 2007;89(6):736-741.
17. Merritt K, Brown SA. Release of hexavalent chromium from corrosion of stainless steel and cobalt-chromium alloys. J Biomed Mater Res. 1995;29(5):627-633.
18. Wallach S, Verch RL. Placental transport of chromium. J Am Coll Nutr. 1984;3(1):69-74.
19. Jacobs JJ, Gilbert JL, Urban RM. Corrosion of metal orthopaedic implants. J Bone Joint Surg Am. 1998;80(2):268-282.
20. Van Der Straeten C, Grammatopoulos G, Gill HS, Calistri A, Campbell P, De Smet KA. The 2012 Otto Aufranc Award: The interpretation of metal ion levels in unilateral and bilateral hip resurfacing. Clin Orthop. 2013;471(2):377-385.
21. Sierra RJ, Trousdale RT, Cabanela ME. Pregnancy and childbirth after total hip arthroplasty. J Bone Joint Surg Br. 2005;87(1):21-24.
22. Antoniou J, Zukor DJ, Mwale F, Minarik W, Petit A, Huk OL. Metal ion levels in the blood of patients after hip resurfacing: a comparison between twenty-eight and thirty-six-millimeter-head metal-on-metal prostheses. J Bone Joint Surg Am. 2008;90(Suppl 3):142-148.
23. Iavicoli I, Falcone G, Alessandrelli M, et al. The release of metals from metal-on-metal surface arthroplasty of the hip. J Trace Elem Med Biol. 2006;20(1):25-31.
24. Ziaee H, Daniel J, Datta AK, Blunt S, McMinn DJ. Transplacental transfer of cobalt and chromium in patients with metal-on-metal hip arthroplasty: a controlled study. J Bone Joint Surg Br. 2007;89(3):301-305.
25. Rudge CV, Rollin HB, Nogueira CM, Thomassen Y, Rudge MC, Odland JO. The placenta as a barrier for toxic and essential elements in paired maternal and cord blood samples of South African delivering women. J Environ Monit. 2009;11(7):1322-1330.
26. deSouza RM, Wallace D, Costa ML, Krikler SJ. Transplacental passage of metal ions in women with hip resurfacing: no teratogenic effects observed. Hip Int. 2012;22(1):96-99.
27. Fritzsche J, Borisch C, Schaefer C. Case report: High chromium and cobalt levels in a pregnant patient with bilateral metal-on-metal hip arthroplasties. Clin Orthop. 2012;470(8):2325-2331.
28. Saxena DK, Murthy RC, Jain VK, Chandra SV. Fetoplacental-maternal uptake of hexavalent chromium administered orally in rats and mice. Bull Environ Contam Toxicol. 1990;45(3):430-435.
29. Junaid M, Murthy RC, Saxena DK. Chromium fetotoxicity in mice during late pregnancy. Vet Hum Toxicol. 1995;37(4):320-323.
30. Paternain JL, Domingo JL, Corbella J. Developmental toxicity of cobalt in the rat. J Toxicol Environ Health. 1988;24(2):193-200.
31. Rurak DW. Development and function of the placenta. In: Harding R, Bocking AD, eds. Fetal Growth and Development. Cambridge, UK: Cambridge University Press; 2001.
32. Hsu AR, Levine BR, Skipor AK, Hallab NJ, Paprosky WG, Jacobs JJ. Effect of a second joint arthroplasty on metal ion levels after primary total hip arthroplasty. Am J Orthop. 2013;42(10):E84-E87.
Serum metal ion levels are increased after primary total hip arthroplasty (THA) with all types of head-liner bearing surfaces.1-4 In recent years, there has been increasing concern regarding elevated metal ion levels and adverse local and systemic effects, in particular, from metal-on-metal (MOM) implants.5 There have been reports of adverse local tissue reactions (ALTR) and systemic effects associated with elevated metal ion levels from MOM THA.6-10 MOM hip resurfacings have been advocated in the literature for use in select patient populations, such as young, active individuals.11,12 Purported benefits of MOM-bearing surfaces in this patient population include reduced wear and the prevention of osteolysis.13 While the incidence of ALTR has been reported to be approximately 1% within 5 years after MOM hip resurfacing, the prevalence of ALTR at long-term follow-up is unknown.14 Adverse local tissue reactions in hip resurfacing are related in part to femoral head coverage by the acetabular cup, as well as component design and orientation. The risk for ALTR with MOM THA has been reported to correlate with the level of serum metal ion levels because of the bearing surface, along with taper corrosion and corrosion secondary to the large surface area of the femoral head.13-15 The overall clinical and prognostic value of metal ion levels in cases of MOM bearings remains controversial and without clear cut-off values.15
The long-term physiologic response to elevated serum metal ion levels from THA implants remains unknown and is of particular concern in pregnant women because of potential transplacental transfer. Several studies have shown that DNA (deoxyribonucleic acid) and chromosomal changes can occur in patients with both MOM and non-MOM hip implants.16-18 Techniques to accurately measure the levels of metal ions, including cobalt, chromium, and titanium in the serum, have improved substantially in recent years and have been used successfully in clinical applications with low detection limits.2,4,19,20 Evidence shows that pregnancy-related complications in women with well-functioning cemented, hybrid, or uncemented metal-on-polyethylene THA are not different from those in healthy women.21 However, it is unclear if metal ions can cross the placenta and negatively affect the development of a fetus in mothers with MOM-bearing THAs.22 We conducted this study to determine if metal ions can traverse the placenta by measuring serum metal ion concentrations in mothers with and without MOM THA and the corresponding levels in umbilical cord blood samples.
Materials and Methods
Ten patients were prospectively enrolled in this study, which was approved by the institutional review boards at all 3 participating hospitals prior to initiation. All patients provided informed consent and agreed to maternal and umbilical cord blood sampling. Seven of the 10 patients did not have any metallic implants and served as controls. The remaining 3 patients had either a MOM unilateral primary THA (1 patient) or unilateral MOM hip-resurfacing arthroplasty (2 patients) with no other metal implants in the body. For all subjects, maternal and umbilical cord blood was obtained at the time of delivery.
Group Demographics
The 3 women in the implant group had a mean age of 32.3 years (range, 23-39 years) at time of delivery. The first patient had posttraumatic osteoarthritis and underwent right THA using a modular MOM 60-mm acetabular component, a 36-mm cobalt chrome head, and a cementless, titanium proximally porous-coated femoral component (Pinnacle Total Hip System and Summit Total Hip System; DePuy Synthes, Warsaw, Indiana). Her infant was born 2 years after index THA. The second patient had a diagnosis of degenerative osteoarthritis secondary to developmental dysplasia of the hip and underwent a hybrid MOM hip resurfacing with a monoblock 50-mm cup and 40-mm cobalt chrome head (Conserve Plus; Wright Medical Technology, Arlington, Tennessee). She gave birth to her infant 6 years after her hip resurfacing. The third patient also had a diagnosis of degenerative osteoarthritis secondary to developmental dysplasia of the hip and underwent hip resurfacing with a 54-mm monoblock cup and 44-mm cobalt chrome head (Conserve Plus). Her infant was born 4 years after her resurfacing arthroplasty. All of the infants were born healthy, and the deliveries were uneventful and without complications. Seven women with a mean age of 32.1 years (range, 24-37 years) and their infants served as controls at the time of delivery. None of the women in the control group had a history of renal impairment, inherited genetic disorders, or metal implants.
Serum Metal Ion Analysis
Blood samples were collected using S-Monovette polypropylene syringes (Sarstedt, Princeton, New Jersey), a multi-adapter, and infusion set following an established technique.1 All vessels and utensils used for specimen collection were verified to be free of metal contamination. Three 10-mL syringes were drawn, and each syringe was labeled to indicate the sequence of collection. The first 10 mL were drawn to rinse the needle and adapter. Blood was then allowed to clot naturally and centrifuged at 1850 rpm for 30 minutes, separating samples into cell and serum fractions that were stored in labeled vials at -80ºC. All specimen manipulations after collection were carried out in a class-100 environment using a SterilGARD Biological Safety Cabinet (Baker, Sanford, Maine) and class-100 gloves (Oak Technical, Ravenna, Ohio) to minimize atmospheric and manual contamination. Maternal and umbilical cord blood metal ion levels were tested using high-resolution sector-field inductively-coupled plasma-mass spectrometry (HR-SF-ICPMS) (Element 2; Thermo Fisher Scientific, Bremen, Germany) following the method of additions as described previously.23 The HR-SF-ICPMS machine was equipped with an SC-E2 autosampler, Teflon nebulizer and spray chamber, sapphire injector (Elemental Scientific, Omaha, Nebraska) and platinum cones. All calibration and internal standard solutions were prepared by gradual dilutions of single-element standard solutions (1000 μg/mL from High Purity Standards, Charleston, South Carolina). The certified reference material, Seronorm Trace Elements Serum (SERO, Billingstad, Norway), were routinely analyzed with samples. The serum concentrations of cobalt, chromium, titanium, and nickel were measured with detection limits in ng/mL (parts per billion) of 0.04 for cobalt, 0.015 for chromium, 0.2 for titanium, and 0.17 for nickel.4 Concentrations below the detection limit were approximated as one-half of the detection limit by convention to calculate means.
Statistical Analysis
The data reported are the means for each group for each of the metal ion levels analyzed. Intergroup comparisons were made with the Mann-Whitney-Wilcoxon test using SPSS statistics software (SPSS Science Inc, Chicago, Illinois) to compare implant and control groups in regards to serum metal ion levels. Intragroup comparisons were made using the Friedman test with significance set at P < .05. Spearman rank-order correlation tests were used to investigate relationships between maternal and infant serum metal ion levels.
Results
The Table shows the mean serum metal ion levels of chromium, cobalt, titanium, and nickel for both groups. The implant-group mothers had significantly higher chromium and cobalt levels than the control-group mothers, with mean chromium levels of 1.87 ng/mL vs 0.16 ng/mL (P = .01) and mean cobalt levels of 0.97 ng/mL vs 0.20 ng/mL (P = .01), respectively. All control-group maternal chromium and cobalt levels were lower than the implant group. There were no significant differences detected between the implant-group and control-group mothers with respect to serum titanium or nickel levels.
The implant-group cord blood samples also had significantly higher chromium and cobalt levels than did the control-group infants with mean chromium levels of 0.29 ng/mL vs 0.10 ng/mL (P = .03) and mean cobalt levels of 0.49 ng/mL vs 0.16 ng/mL (P = .01), respectively. All but 1 of the control-group infants had chromium levels that were lower than the implant-group infant chromium levels. All of the control-group infant cobalt levels were lower than the implant-group infant cobalt levels. In the mother (I-1) who had a titanium-containing implant, her titanium level was 1.77 ng/mL and her cord blood level was 0.78 ng/mL. In contrast, the other 2 patients did not have titanium-containing implants and had corresponding metal levels of 0.10 ng/mL and cord blood levels either below the detection limit or just slightly above it. No statistically significant differences were found between the implant- and control-group infants with respect to serum titanium or nickel levels.
Considering the implant and control groups separately, we found no statistically significant differences between the maternal and infant titanium levels or the maternal and infant nickel levels. In the implant group, the mother’s chromium level was always higher than her infant’s chromium level, and the mother’s cobalt level was always higher than her infant’s cobalt level (P= .08). In the control group, there was no correlation between the maternal and infant chromium levels, or between the maternal and infant cobalt levels. In the implant group, the maternal and infant chromium levels were highly correlated (r = 1), as were the maternal and infant cobalt levels (r = 1).
When infants’ chromium levels were expressed as a percentage of their mothers’ chromium levels, the mean was 15.4% (range, 12.3%-18%) for the implant group and 58.9% (range, 13.5%-165.7%) for the control group (P = .05). The mean infant cobalt level, expressed as percentage of maternal cobalt level, was 50% (range, 45.8%-53.1%) for the implant group and 76.6% (range, 59.1%-150.8%) for the control group (P = .01).
Discussion
Cobalt and chromium serum metal ion levels obtained from mothers with MOM implants and umbilical cord samples were significantly elevated in comparison with controls. There was also a strong correlation between implant-group maternal cobalt and chromium levels and implant-group infant cobalt and chromium levels; however, no such correlation existed in the control group. Our results suggest that cobalt and chromium cross the placental barrier. Implant mothers had higher chromium and cobalt levels compared with implant babies, and these babies had approximately 15% of the level of chromium and 50% of the level of cobalt when compared to implant maternal levels. This finding suggests that the placenta modulates the transfer of chromium and cobalt to the fetus.
Two studies have reported chromium and cobalt levels in maternal and umbilical cord sera after MOM THA.5,24 Brodner and colleagues5 determined the maternal serum levels of cobalt and chromium in 3 women 3.8 years after MOM THA and compared those to cobalt and chromium levels obtained from umbilical cord blood. At the time of delivery, the maternal chromium concentrations in the 3 patients were 1.6 ng/mL, 0.5 ng/mL, and 0.9 ng/mL, and the cobalt concentrations were 1 ng/mL in 1 patient and below the detection limit in the other 2 patients. Cobalt and chromium concentrations of the 3 umbilical cord sera were below the detection limit. The authors concluded that cobalt and chromium did not cross the placenta based on their laboratory detection limits. Metal ion levels were measured using atomic absorption spectrometry with relatively high detection limits of 0.3 ng/mL, which is not as sensitive as the HR-SF-ICPMS technique used in the present study that has detection limits of 0.04 ng/mL for cobalt and 0.015 ng/mL for chromium. The relatively high detection limits of atomic absorption spectrometry were likely responsible for the authors’ inability to detect elevated chromium and cobalt levels in umbilical cord sera.
Ziaee and colleagues24 used HR-SF-ICPMS, as we did in this study, to measure the mean concentrations of cobalt and chromium ion levels in 10 maternal and umbilical cord blood samples in women with Birmingham MOM hip-resurfacing prostheses (Smith & Nephew, Warwick, United Kingdom). Nine of those patients had a unilateral resurfacing and 1 patient had bilateral-resurfacing prostheses. The mean maternal age was 31 years, and mean duration between hip resurfacing and delivery was 53 months. Ten normal controls were also tested with a mean maternal age of 30.9 years. The authors found that the mean cord blood level of cobalt in the study patients was 0.83 ng/mL, significantly higher (P < .01) than cobalt levels in the control group, which measured 0.33 ng/mL. The mean cord blood levels of chromium in the study and control groups were 0.37 ng/mL and 0.19 ng/mL, respectively. No children were reported to have evidence of congenital anomalies. Similar to our findings, they noted a modulatory effect on the transfer of metal ions across the placenta in patients with MOM prostheses.24 They reported the relative levels of cobalt and chromium in offspring to be 60.4% and 29.4% of the maternal ion levels, respectively. Control-group infants had mean cobalt and chromium levels that were 98.5% and 97.2% of mean maternal levels, respectively.
The transfer of metal ions across the placenta in control subjects is an expected finding because cobalt and chromium are essential trace elements required by the developing fetus. Rudge and colleagues25 estimated a transplacental transfer rate of 45% for cobalt in a series of 62 paired samples of maternal and cord blood. DeSouza and coauthors26 reported a series of 3 patients with MOM hip resurfacings who had the prosthesis in situ during pregnancy and found no teratogenic effects of metal ion transfer across the placenta. Umbilical cord blood chromium levels were less than 25% of the maternal serum levels and cord blood cobalt levels were approximately 50% that of maternal blood.26 In an animal experiment, Wallach and Verch18 also reported that maternal chromium levels can be decreased because of placental uptake.
Ziaee and colleagues24 tested metal ion levels using whole blood in contrast to serum, as we did in the present study. Daniel and coauthors16, who reported on the validity of serum levels as a surrogate measure of systemic exposure to metal ions in hip replacement, suggested that serum and whole blood metal ion levels cannot be interconverted because metal within cells are not in dynamic equilibrium with extracellular levels. They concluded that serum metal ion concentrations are not a useful surrogate measure of systemic metal ion exposure based on the wide variability seen in normalized and Bland-Altman scatterplots.16 However, it is important to note that Bland-Altman plots are user-dependent in determining significance, and results can vary based on the parameters tested. A high correlation does not automatically imply that there is a good agreement between 2 methods because a widespread sample could influence results. Whole blood analysis requires more processing steps, thus providing an increased chance of contamination and variability compared with serum metal level analysis. In our experiences, serum metal ion analysis has been shown to have accurate and reproducible results in clinical situations.2,4
While there is insufficient literature that specifically studies the effects of elevated metal ion levels on maternal and fetal subjects, there have been no reported negative effects in human babies even when maternal ion levels are elevated enough to be associated with ALTR. A case report by Fritzsche and colleagues27 reported a mother with bilateral MOM THA, a recurrent pseudotumor, and high blood levels of chromium (39 ng/mL) and cobalt (138 ng/mL) at 12 weeks gestation. The child was born at 38 weeks gestation with cord blood chromium and cobalt levels of 2.1 ng/mL and 75 ng/mL, respectively. The infant’s metal ion levels remained elevated at age 8 weeks with a chromium level of 2.5 ng/mL and cobalt level of 13 ng/mL and no signs of toxicity by age 14 weeks. In an animal model, Saxena and colleagues28 found that chromium in the hexavalent form passed through the placenta in mice and rats that were fed high doses of potassium dichromate. Trivalent chromium was not found to cross the placenta. In a follow-up study, Junaid and coauthors29 investigated the effects of elevated chromium levels in female mice given potassium dichromate in drinking water on days 14 to 19 of pregnancy. Animals receiving high-dose chromium had significantly higher incidences of postimplantation loss along with subdermal hemorrhagic patches and reduced ossification.29 Cobalt has not been shown to be teratogenic or cause fetotoxicity in a rat animal model given daily doses of as much as 100 mg/kg cobalt (II) chloride on days 6 to 15 of gestation.30
It is important to recognize that rodent data are limited and may not provide accurate translational insight into the effects of metal ions in human maternal and fetal subjects. Mammalian species have significant heterogeneity in the structure and function of their placentas. Rurak31 has shown that rodents have an additional persisting yolk sac placenta that allows the transfer of maternal immunoglobulins to the fetus. Humans, on the other hand, have a yolk sac placenta that regresses early in pregnancy. Differing placental biologic function makes it difficult to extrapolate the effects of metal ions in rodents to human subjects.
It is also important to note that serum levels of cobalt, chromium, and titanium can remain persistently elevated in well-functioning metal-on-polyethylene THA for several years and that elevated metal ion levels are not confined to MOM bearings.2 Levine and colleagues4 reported that serum levels of cobalt, chromium, and titanium remain persistently elevated after 10 years in a cohort of 27 well-functioning primary metal-on-polyethylene THA (hybrid, cobalt-chrome, titanium). Cobalt concentrations were elevated in all implants compared with controls at all follow-up periods through 10 years with absolute values less than 1 ng/mL. The authors noted that metal release at the modular femoral head-neck junctions was likely the dominant source of serum cobalt and chromium rather than passive dissolution. Hsu and colleagues32 have also shown that patients undergoing a second metal-on-polyethylene THA after primary THA have elevated serum metal ion levels (cobalt, chromium, titanium) up to 6 years after second surgery. Reported cobalt concentrations in patients with unilateral THA reached a maximum of 0.5 ng/mL during the follow-up course compared with 1.5 ng/mL for patients with bilateral THA. It is unknown what the potential metal ion transfer load would be in mothers with metal-on-polyethylene THA and associated taper corrosion to their infants.
Conclusion
Mothers with MOM-bearing implants and their children have higher cobalt and chromium levels than control subjects, demonstrating that the placenta is not a complete barrier to metal ion transport, although it seems to have a modulating effect. Physicians and women of child-bearing age should be aware of these findings when considering the use of MOM-bearing couples for THA. The effects of metal ions on long-term maternal and fetal health require research through serial clinical exams and metal ion level testing in prospective studies of different THA-bearing surfaces.
Serum metal ion levels are increased after primary total hip arthroplasty (THA) with all types of head-liner bearing surfaces.1-4 In recent years, there has been increasing concern regarding elevated metal ion levels and adverse local and systemic effects, in particular, from metal-on-metal (MOM) implants.5 There have been reports of adverse local tissue reactions (ALTR) and systemic effects associated with elevated metal ion levels from MOM THA.6-10 MOM hip resurfacings have been advocated in the literature for use in select patient populations, such as young, active individuals.11,12 Purported benefits of MOM-bearing surfaces in this patient population include reduced wear and the prevention of osteolysis.13 While the incidence of ALTR has been reported to be approximately 1% within 5 years after MOM hip resurfacing, the prevalence of ALTR at long-term follow-up is unknown.14 Adverse local tissue reactions in hip resurfacing are related in part to femoral head coverage by the acetabular cup, as well as component design and orientation. The risk for ALTR with MOM THA has been reported to correlate with the level of serum metal ion levels because of the bearing surface, along with taper corrosion and corrosion secondary to the large surface area of the femoral head.13-15 The overall clinical and prognostic value of metal ion levels in cases of MOM bearings remains controversial and without clear cut-off values.15
The long-term physiologic response to elevated serum metal ion levels from THA implants remains unknown and is of particular concern in pregnant women because of potential transplacental transfer. Several studies have shown that DNA (deoxyribonucleic acid) and chromosomal changes can occur in patients with both MOM and non-MOM hip implants.16-18 Techniques to accurately measure the levels of metal ions, including cobalt, chromium, and titanium in the serum, have improved substantially in recent years and have been used successfully in clinical applications with low detection limits.2,4,19,20 Evidence shows that pregnancy-related complications in women with well-functioning cemented, hybrid, or uncemented metal-on-polyethylene THA are not different from those in healthy women.21 However, it is unclear if metal ions can cross the placenta and negatively affect the development of a fetus in mothers with MOM-bearing THAs.22 We conducted this study to determine if metal ions can traverse the placenta by measuring serum metal ion concentrations in mothers with and without MOM THA and the corresponding levels in umbilical cord blood samples.
Materials and Methods
Ten patients were prospectively enrolled in this study, which was approved by the institutional review boards at all 3 participating hospitals prior to initiation. All patients provided informed consent and agreed to maternal and umbilical cord blood sampling. Seven of the 10 patients did not have any metallic implants and served as controls. The remaining 3 patients had either a MOM unilateral primary THA (1 patient) or unilateral MOM hip-resurfacing arthroplasty (2 patients) with no other metal implants in the body. For all subjects, maternal and umbilical cord blood was obtained at the time of delivery.
Group Demographics
The 3 women in the implant group had a mean age of 32.3 years (range, 23-39 years) at time of delivery. The first patient had posttraumatic osteoarthritis and underwent right THA using a modular MOM 60-mm acetabular component, a 36-mm cobalt chrome head, and a cementless, titanium proximally porous-coated femoral component (Pinnacle Total Hip System and Summit Total Hip System; DePuy Synthes, Warsaw, Indiana). Her infant was born 2 years after index THA. The second patient had a diagnosis of degenerative osteoarthritis secondary to developmental dysplasia of the hip and underwent a hybrid MOM hip resurfacing with a monoblock 50-mm cup and 40-mm cobalt chrome head (Conserve Plus; Wright Medical Technology, Arlington, Tennessee). She gave birth to her infant 6 years after her hip resurfacing. The third patient also had a diagnosis of degenerative osteoarthritis secondary to developmental dysplasia of the hip and underwent hip resurfacing with a 54-mm monoblock cup and 44-mm cobalt chrome head (Conserve Plus). Her infant was born 4 years after her resurfacing arthroplasty. All of the infants were born healthy, and the deliveries were uneventful and without complications. Seven women with a mean age of 32.1 years (range, 24-37 years) and their infants served as controls at the time of delivery. None of the women in the control group had a history of renal impairment, inherited genetic disorders, or metal implants.
Serum Metal Ion Analysis
Blood samples were collected using S-Monovette polypropylene syringes (Sarstedt, Princeton, New Jersey), a multi-adapter, and infusion set following an established technique.1 All vessels and utensils used for specimen collection were verified to be free of metal contamination. Three 10-mL syringes were drawn, and each syringe was labeled to indicate the sequence of collection. The first 10 mL were drawn to rinse the needle and adapter. Blood was then allowed to clot naturally and centrifuged at 1850 rpm for 30 minutes, separating samples into cell and serum fractions that were stored in labeled vials at -80ºC. All specimen manipulations after collection were carried out in a class-100 environment using a SterilGARD Biological Safety Cabinet (Baker, Sanford, Maine) and class-100 gloves (Oak Technical, Ravenna, Ohio) to minimize atmospheric and manual contamination. Maternal and umbilical cord blood metal ion levels were tested using high-resolution sector-field inductively-coupled plasma-mass spectrometry (HR-SF-ICPMS) (Element 2; Thermo Fisher Scientific, Bremen, Germany) following the method of additions as described previously.23 The HR-SF-ICPMS machine was equipped with an SC-E2 autosampler, Teflon nebulizer and spray chamber, sapphire injector (Elemental Scientific, Omaha, Nebraska) and platinum cones. All calibration and internal standard solutions were prepared by gradual dilutions of single-element standard solutions (1000 μg/mL from High Purity Standards, Charleston, South Carolina). The certified reference material, Seronorm Trace Elements Serum (SERO, Billingstad, Norway), were routinely analyzed with samples. The serum concentrations of cobalt, chromium, titanium, and nickel were measured with detection limits in ng/mL (parts per billion) of 0.04 for cobalt, 0.015 for chromium, 0.2 for titanium, and 0.17 for nickel.4 Concentrations below the detection limit were approximated as one-half of the detection limit by convention to calculate means.
Statistical Analysis
The data reported are the means for each group for each of the metal ion levels analyzed. Intergroup comparisons were made with the Mann-Whitney-Wilcoxon test using SPSS statistics software (SPSS Science Inc, Chicago, Illinois) to compare implant and control groups in regards to serum metal ion levels. Intragroup comparisons were made using the Friedman test with significance set at P < .05. Spearman rank-order correlation tests were used to investigate relationships between maternal and infant serum metal ion levels.
Results
The Table shows the mean serum metal ion levels of chromium, cobalt, titanium, and nickel for both groups. The implant-group mothers had significantly higher chromium and cobalt levels than the control-group mothers, with mean chromium levels of 1.87 ng/mL vs 0.16 ng/mL (P = .01) and mean cobalt levels of 0.97 ng/mL vs 0.20 ng/mL (P = .01), respectively. All control-group maternal chromium and cobalt levels were lower than the implant group. There were no significant differences detected between the implant-group and control-group mothers with respect to serum titanium or nickel levels.
The implant-group cord blood samples also had significantly higher chromium and cobalt levels than did the control-group infants with mean chromium levels of 0.29 ng/mL vs 0.10 ng/mL (P = .03) and mean cobalt levels of 0.49 ng/mL vs 0.16 ng/mL (P = .01), respectively. All but 1 of the control-group infants had chromium levels that were lower than the implant-group infant chromium levels. All of the control-group infant cobalt levels were lower than the implant-group infant cobalt levels. In the mother (I-1) who had a titanium-containing implant, her titanium level was 1.77 ng/mL and her cord blood level was 0.78 ng/mL. In contrast, the other 2 patients did not have titanium-containing implants and had corresponding metal levels of 0.10 ng/mL and cord blood levels either below the detection limit or just slightly above it. No statistically significant differences were found between the implant- and control-group infants with respect to serum titanium or nickel levels.
Considering the implant and control groups separately, we found no statistically significant differences between the maternal and infant titanium levels or the maternal and infant nickel levels. In the implant group, the mother’s chromium level was always higher than her infant’s chromium level, and the mother’s cobalt level was always higher than her infant’s cobalt level (P= .08). In the control group, there was no correlation between the maternal and infant chromium levels, or between the maternal and infant cobalt levels. In the implant group, the maternal and infant chromium levels were highly correlated (r = 1), as were the maternal and infant cobalt levels (r = 1).
When infants’ chromium levels were expressed as a percentage of their mothers’ chromium levels, the mean was 15.4% (range, 12.3%-18%) for the implant group and 58.9% (range, 13.5%-165.7%) for the control group (P = .05). The mean infant cobalt level, expressed as percentage of maternal cobalt level, was 50% (range, 45.8%-53.1%) for the implant group and 76.6% (range, 59.1%-150.8%) for the control group (P = .01).
Discussion
Cobalt and chromium serum metal ion levels obtained from mothers with MOM implants and umbilical cord samples were significantly elevated in comparison with controls. There was also a strong correlation between implant-group maternal cobalt and chromium levels and implant-group infant cobalt and chromium levels; however, no such correlation existed in the control group. Our results suggest that cobalt and chromium cross the placental barrier. Implant mothers had higher chromium and cobalt levels compared with implant babies, and these babies had approximately 15% of the level of chromium and 50% of the level of cobalt when compared to implant maternal levels. This finding suggests that the placenta modulates the transfer of chromium and cobalt to the fetus.
Two studies have reported chromium and cobalt levels in maternal and umbilical cord sera after MOM THA.5,24 Brodner and colleagues5 determined the maternal serum levels of cobalt and chromium in 3 women 3.8 years after MOM THA and compared those to cobalt and chromium levels obtained from umbilical cord blood. At the time of delivery, the maternal chromium concentrations in the 3 patients were 1.6 ng/mL, 0.5 ng/mL, and 0.9 ng/mL, and the cobalt concentrations were 1 ng/mL in 1 patient and below the detection limit in the other 2 patients. Cobalt and chromium concentrations of the 3 umbilical cord sera were below the detection limit. The authors concluded that cobalt and chromium did not cross the placenta based on their laboratory detection limits. Metal ion levels were measured using atomic absorption spectrometry with relatively high detection limits of 0.3 ng/mL, which is not as sensitive as the HR-SF-ICPMS technique used in the present study that has detection limits of 0.04 ng/mL for cobalt and 0.015 ng/mL for chromium. The relatively high detection limits of atomic absorption spectrometry were likely responsible for the authors’ inability to detect elevated chromium and cobalt levels in umbilical cord sera.
Ziaee and colleagues24 used HR-SF-ICPMS, as we did in this study, to measure the mean concentrations of cobalt and chromium ion levels in 10 maternal and umbilical cord blood samples in women with Birmingham MOM hip-resurfacing prostheses (Smith & Nephew, Warwick, United Kingdom). Nine of those patients had a unilateral resurfacing and 1 patient had bilateral-resurfacing prostheses. The mean maternal age was 31 years, and mean duration between hip resurfacing and delivery was 53 months. Ten normal controls were also tested with a mean maternal age of 30.9 years. The authors found that the mean cord blood level of cobalt in the study patients was 0.83 ng/mL, significantly higher (P < .01) than cobalt levels in the control group, which measured 0.33 ng/mL. The mean cord blood levels of chromium in the study and control groups were 0.37 ng/mL and 0.19 ng/mL, respectively. No children were reported to have evidence of congenital anomalies. Similar to our findings, they noted a modulatory effect on the transfer of metal ions across the placenta in patients with MOM prostheses.24 They reported the relative levels of cobalt and chromium in offspring to be 60.4% and 29.4% of the maternal ion levels, respectively. Control-group infants had mean cobalt and chromium levels that were 98.5% and 97.2% of mean maternal levels, respectively.
The transfer of metal ions across the placenta in control subjects is an expected finding because cobalt and chromium are essential trace elements required by the developing fetus. Rudge and colleagues25 estimated a transplacental transfer rate of 45% for cobalt in a series of 62 paired samples of maternal and cord blood. DeSouza and coauthors26 reported a series of 3 patients with MOM hip resurfacings who had the prosthesis in situ during pregnancy and found no teratogenic effects of metal ion transfer across the placenta. Umbilical cord blood chromium levels were less than 25% of the maternal serum levels and cord blood cobalt levels were approximately 50% that of maternal blood.26 In an animal experiment, Wallach and Verch18 also reported that maternal chromium levels can be decreased because of placental uptake.
Ziaee and colleagues24 tested metal ion levels using whole blood in contrast to serum, as we did in the present study. Daniel and coauthors16, who reported on the validity of serum levels as a surrogate measure of systemic exposure to metal ions in hip replacement, suggested that serum and whole blood metal ion levels cannot be interconverted because metal within cells are not in dynamic equilibrium with extracellular levels. They concluded that serum metal ion concentrations are not a useful surrogate measure of systemic metal ion exposure based on the wide variability seen in normalized and Bland-Altman scatterplots.16 However, it is important to note that Bland-Altman plots are user-dependent in determining significance, and results can vary based on the parameters tested. A high correlation does not automatically imply that there is a good agreement between 2 methods because a widespread sample could influence results. Whole blood analysis requires more processing steps, thus providing an increased chance of contamination and variability compared with serum metal level analysis. In our experiences, serum metal ion analysis has been shown to have accurate and reproducible results in clinical situations.2,4
While there is insufficient literature that specifically studies the effects of elevated metal ion levels on maternal and fetal subjects, there have been no reported negative effects in human babies even when maternal ion levels are elevated enough to be associated with ALTR. A case report by Fritzsche and colleagues27 reported a mother with bilateral MOM THA, a recurrent pseudotumor, and high blood levels of chromium (39 ng/mL) and cobalt (138 ng/mL) at 12 weeks gestation. The child was born at 38 weeks gestation with cord blood chromium and cobalt levels of 2.1 ng/mL and 75 ng/mL, respectively. The infant’s metal ion levels remained elevated at age 8 weeks with a chromium level of 2.5 ng/mL and cobalt level of 13 ng/mL and no signs of toxicity by age 14 weeks. In an animal model, Saxena and colleagues28 found that chromium in the hexavalent form passed through the placenta in mice and rats that were fed high doses of potassium dichromate. Trivalent chromium was not found to cross the placenta. In a follow-up study, Junaid and coauthors29 investigated the effects of elevated chromium levels in female mice given potassium dichromate in drinking water on days 14 to 19 of pregnancy. Animals receiving high-dose chromium had significantly higher incidences of postimplantation loss along with subdermal hemorrhagic patches and reduced ossification.29 Cobalt has not been shown to be teratogenic or cause fetotoxicity in a rat animal model given daily doses of as much as 100 mg/kg cobalt (II) chloride on days 6 to 15 of gestation.30
It is important to recognize that rodent data are limited and may not provide accurate translational insight into the effects of metal ions in human maternal and fetal subjects. Mammalian species have significant heterogeneity in the structure and function of their placentas. Rurak31 has shown that rodents have an additional persisting yolk sac placenta that allows the transfer of maternal immunoglobulins to the fetus. Humans, on the other hand, have a yolk sac placenta that regresses early in pregnancy. Differing placental biologic function makes it difficult to extrapolate the effects of metal ions in rodents to human subjects.
It is also important to note that serum levels of cobalt, chromium, and titanium can remain persistently elevated in well-functioning metal-on-polyethylene THA for several years and that elevated metal ion levels are not confined to MOM bearings.2 Levine and colleagues4 reported that serum levels of cobalt, chromium, and titanium remain persistently elevated after 10 years in a cohort of 27 well-functioning primary metal-on-polyethylene THA (hybrid, cobalt-chrome, titanium). Cobalt concentrations were elevated in all implants compared with controls at all follow-up periods through 10 years with absolute values less than 1 ng/mL. The authors noted that metal release at the modular femoral head-neck junctions was likely the dominant source of serum cobalt and chromium rather than passive dissolution. Hsu and colleagues32 have also shown that patients undergoing a second metal-on-polyethylene THA after primary THA have elevated serum metal ion levels (cobalt, chromium, titanium) up to 6 years after second surgery. Reported cobalt concentrations in patients with unilateral THA reached a maximum of 0.5 ng/mL during the follow-up course compared with 1.5 ng/mL for patients with bilateral THA. It is unknown what the potential metal ion transfer load would be in mothers with metal-on-polyethylene THA and associated taper corrosion to their infants.
Conclusion
Mothers with MOM-bearing implants and their children have higher cobalt and chromium levels than control subjects, demonstrating that the placenta is not a complete barrier to metal ion transport, although it seems to have a modulating effect. Physicians and women of child-bearing age should be aware of these findings when considering the use of MOM-bearing couples for THA. The effects of metal ions on long-term maternal and fetal health require research through serial clinical exams and metal ion level testing in prospective studies of different THA-bearing surfaces.
1. Jacobs JJ, Skipor AK, Black J, Urban R, Galante JO. Release and excretion of metal in patients who have a total hip-replacement component made of titanium-base alloy. J Bone Joint Surg Am. 1991;73(10):1475-1486.
2. Jacobs JJ, Skipor AK, Patterson LM, et al. Metal release in patients who have had a primary total hip arthroplasty. A prospective, controlled, longitudinal study. J Bone Joint Surg Am. 1998;80(10):1447-1458.
3. Jacobs JJ, Skipor AK, Urban RM, Black J, Manion LM, Galante JO. Transport of metal degradation products of titanium alloy total hip replacements to reticuloendothelial organs. An autopsy study. Trans Soc Biomater. 1994;18:318-325.
4. Levine BR, Hsu AR, Skipor AK, et al. Ten-year outcome of serum metal ion levels after primary total hip arthroplasty: a concise follow-up of a previous report. J Bone Joint Surg Am. 2013;95(6):512-518.
5. Brodner W, Grohs JG, Bancher-Todesca D, et al. Does the placenta inhibit the passage of chromium and cobalt after metal-on-metal total hip arthroplasty? J Arthroplasty. 2004;19(8 suppl 3):102-106.
6. Hsu AR, Gross CE, Levine BR. Pseudotumor from modular neck corrosion after ceramic-on-polyethylene total hip arthroplasty. Am J Orthop. 2012;41(9):422-426.
7. Tower SS. Arthroprosthetic cobaltism: neurological and cardiac manifestations in two patients with metal-on-metal arthroplasty: a case report. J Bone Joint Surg Am. 2010;92(17):2847-2851.
8. Urban RM, Jacobs JJ, Tomlinson MJ, Gavrilovic J, Black J, Peoc’h M. Dissemination of wear particles to the liver, spleen, and abdominal lymph nodes of patients with hip or knee replacement. J Bone Joint Surg Am. 2000;82(4):457-476.
9. Watters TS, Eward WC, Hallows RK, Dodd LG, Wellman SS, Bolognesi MP. Pseudotumor with superimposed periprosthetic infection following metal-on-metal total hip arthroplasty: a case report. J Bone Joint Surg Am. 2010;92(7):1666-1669.
10. Willert HG, Buchhorn GH, Fayyazi A, et al. Metal-on-metal bearings and hypersensitivity in patients with artificial hip joints. A clinical and histomorphological study. J Bone Joint Surg Am. 2005;87(1):28-36.
11. Nunley RM, Della Valle CJ, Barrack RL. Is patient selection important for hip resurfacing? Clin Orthop. 2009;467(1):56-65.
12. Treacy RB, McBryde CW, Pynsent PB. Birmingham hip resurfacing arthroplasty. A minimum follow-up of five years. J Bone Joint Surg Br. 2005;87(2):167-170.
13. Dorr LD, Wan Z, Longjohn DB, Dubois B, Murken R. Total hip arthroplasty with use of the Metasul metal-on-metal articulation. Four to seven-year results. J Bone Joint Surg Am. 2000;82(6):789-798.
14. Pandit H, Glyn-Jones S, McLardy-Smith P, et al. Pseudotumours associated with metal-on-metal hip resurfacings. J Bone Joint Surg Br. 2008;90(7):847-851.
15. Hartmann A, Hannemann F, Lutzner J, et al. Metal ion concentrations in body fluids after implantation of hip replacements with metal-on-metal bearing--systematic review of clinical and epidemiological studies. PLoS One. 2013;8(8):e70359.
16. Daniel J, Ziaee H, Pynsent PB, McMinn DJ. The validity of serum levels as a surrogate measure of systemic exposure to metal ions in hip replacement. J Bone Joint Surg Br. 2007;89(6):736-741.
17. Merritt K, Brown SA. Release of hexavalent chromium from corrosion of stainless steel and cobalt-chromium alloys. J Biomed Mater Res. 1995;29(5):627-633.
18. Wallach S, Verch RL. Placental transport of chromium. J Am Coll Nutr. 1984;3(1):69-74.
19. Jacobs JJ, Gilbert JL, Urban RM. Corrosion of metal orthopaedic implants. J Bone Joint Surg Am. 1998;80(2):268-282.
20. Van Der Straeten C, Grammatopoulos G, Gill HS, Calistri A, Campbell P, De Smet KA. The 2012 Otto Aufranc Award: The interpretation of metal ion levels in unilateral and bilateral hip resurfacing. Clin Orthop. 2013;471(2):377-385.
21. Sierra RJ, Trousdale RT, Cabanela ME. Pregnancy and childbirth after total hip arthroplasty. J Bone Joint Surg Br. 2005;87(1):21-24.
22. Antoniou J, Zukor DJ, Mwale F, Minarik W, Petit A, Huk OL. Metal ion levels in the blood of patients after hip resurfacing: a comparison between twenty-eight and thirty-six-millimeter-head metal-on-metal prostheses. J Bone Joint Surg Am. 2008;90(Suppl 3):142-148.
23. Iavicoli I, Falcone G, Alessandrelli M, et al. The release of metals from metal-on-metal surface arthroplasty of the hip. J Trace Elem Med Biol. 2006;20(1):25-31.
24. Ziaee H, Daniel J, Datta AK, Blunt S, McMinn DJ. Transplacental transfer of cobalt and chromium in patients with metal-on-metal hip arthroplasty: a controlled study. J Bone Joint Surg Br. 2007;89(3):301-305.
25. Rudge CV, Rollin HB, Nogueira CM, Thomassen Y, Rudge MC, Odland JO. The placenta as a barrier for toxic and essential elements in paired maternal and cord blood samples of South African delivering women. J Environ Monit. 2009;11(7):1322-1330.
26. deSouza RM, Wallace D, Costa ML, Krikler SJ. Transplacental passage of metal ions in women with hip resurfacing: no teratogenic effects observed. Hip Int. 2012;22(1):96-99.
27. Fritzsche J, Borisch C, Schaefer C. Case report: High chromium and cobalt levels in a pregnant patient with bilateral metal-on-metal hip arthroplasties. Clin Orthop. 2012;470(8):2325-2331.
28. Saxena DK, Murthy RC, Jain VK, Chandra SV. Fetoplacental-maternal uptake of hexavalent chromium administered orally in rats and mice. Bull Environ Contam Toxicol. 1990;45(3):430-435.
29. Junaid M, Murthy RC, Saxena DK. Chromium fetotoxicity in mice during late pregnancy. Vet Hum Toxicol. 1995;37(4):320-323.
30. Paternain JL, Domingo JL, Corbella J. Developmental toxicity of cobalt in the rat. J Toxicol Environ Health. 1988;24(2):193-200.
31. Rurak DW. Development and function of the placenta. In: Harding R, Bocking AD, eds. Fetal Growth and Development. Cambridge, UK: Cambridge University Press; 2001.
32. Hsu AR, Levine BR, Skipor AK, Hallab NJ, Paprosky WG, Jacobs JJ. Effect of a second joint arthroplasty on metal ion levels after primary total hip arthroplasty. Am J Orthop. 2013;42(10):E84-E87.
1. Jacobs JJ, Skipor AK, Black J, Urban R, Galante JO. Release and excretion of metal in patients who have a total hip-replacement component made of titanium-base alloy. J Bone Joint Surg Am. 1991;73(10):1475-1486.
2. Jacobs JJ, Skipor AK, Patterson LM, et al. Metal release in patients who have had a primary total hip arthroplasty. A prospective, controlled, longitudinal study. J Bone Joint Surg Am. 1998;80(10):1447-1458.
3. Jacobs JJ, Skipor AK, Urban RM, Black J, Manion LM, Galante JO. Transport of metal degradation products of titanium alloy total hip replacements to reticuloendothelial organs. An autopsy study. Trans Soc Biomater. 1994;18:318-325.
4. Levine BR, Hsu AR, Skipor AK, et al. Ten-year outcome of serum metal ion levels after primary total hip arthroplasty: a concise follow-up of a previous report. J Bone Joint Surg Am. 2013;95(6):512-518.
5. Brodner W, Grohs JG, Bancher-Todesca D, et al. Does the placenta inhibit the passage of chromium and cobalt after metal-on-metal total hip arthroplasty? J Arthroplasty. 2004;19(8 suppl 3):102-106.
6. Hsu AR, Gross CE, Levine BR. Pseudotumor from modular neck corrosion after ceramic-on-polyethylene total hip arthroplasty. Am J Orthop. 2012;41(9):422-426.
7. Tower SS. Arthroprosthetic cobaltism: neurological and cardiac manifestations in two patients with metal-on-metal arthroplasty: a case report. J Bone Joint Surg Am. 2010;92(17):2847-2851.
8. Urban RM, Jacobs JJ, Tomlinson MJ, Gavrilovic J, Black J, Peoc’h M. Dissemination of wear particles to the liver, spleen, and abdominal lymph nodes of patients with hip or knee replacement. J Bone Joint Surg Am. 2000;82(4):457-476.
9. Watters TS, Eward WC, Hallows RK, Dodd LG, Wellman SS, Bolognesi MP. Pseudotumor with superimposed periprosthetic infection following metal-on-metal total hip arthroplasty: a case report. J Bone Joint Surg Am. 2010;92(7):1666-1669.
10. Willert HG, Buchhorn GH, Fayyazi A, et al. Metal-on-metal bearings and hypersensitivity in patients with artificial hip joints. A clinical and histomorphological study. J Bone Joint Surg Am. 2005;87(1):28-36.
11. Nunley RM, Della Valle CJ, Barrack RL. Is patient selection important for hip resurfacing? Clin Orthop. 2009;467(1):56-65.
12. Treacy RB, McBryde CW, Pynsent PB. Birmingham hip resurfacing arthroplasty. A minimum follow-up of five years. J Bone Joint Surg Br. 2005;87(2):167-170.
13. Dorr LD, Wan Z, Longjohn DB, Dubois B, Murken R. Total hip arthroplasty with use of the Metasul metal-on-metal articulation. Four to seven-year results. J Bone Joint Surg Am. 2000;82(6):789-798.
14. Pandit H, Glyn-Jones S, McLardy-Smith P, et al. Pseudotumours associated with metal-on-metal hip resurfacings. J Bone Joint Surg Br. 2008;90(7):847-851.
15. Hartmann A, Hannemann F, Lutzner J, et al. Metal ion concentrations in body fluids after implantation of hip replacements with metal-on-metal bearing--systematic review of clinical and epidemiological studies. PLoS One. 2013;8(8):e70359.
16. Daniel J, Ziaee H, Pynsent PB, McMinn DJ. The validity of serum levels as a surrogate measure of systemic exposure to metal ions in hip replacement. J Bone Joint Surg Br. 2007;89(6):736-741.
17. Merritt K, Brown SA. Release of hexavalent chromium from corrosion of stainless steel and cobalt-chromium alloys. J Biomed Mater Res. 1995;29(5):627-633.
18. Wallach S, Verch RL. Placental transport of chromium. J Am Coll Nutr. 1984;3(1):69-74.
19. Jacobs JJ, Gilbert JL, Urban RM. Corrosion of metal orthopaedic implants. J Bone Joint Surg Am. 1998;80(2):268-282.
20. Van Der Straeten C, Grammatopoulos G, Gill HS, Calistri A, Campbell P, De Smet KA. The 2012 Otto Aufranc Award: The interpretation of metal ion levels in unilateral and bilateral hip resurfacing. Clin Orthop. 2013;471(2):377-385.
21. Sierra RJ, Trousdale RT, Cabanela ME. Pregnancy and childbirth after total hip arthroplasty. J Bone Joint Surg Br. 2005;87(1):21-24.
22. Antoniou J, Zukor DJ, Mwale F, Minarik W, Petit A, Huk OL. Metal ion levels in the blood of patients after hip resurfacing: a comparison between twenty-eight and thirty-six-millimeter-head metal-on-metal prostheses. J Bone Joint Surg Am. 2008;90(Suppl 3):142-148.
23. Iavicoli I, Falcone G, Alessandrelli M, et al. The release of metals from metal-on-metal surface arthroplasty of the hip. J Trace Elem Med Biol. 2006;20(1):25-31.
24. Ziaee H, Daniel J, Datta AK, Blunt S, McMinn DJ. Transplacental transfer of cobalt and chromium in patients with metal-on-metal hip arthroplasty: a controlled study. J Bone Joint Surg Br. 2007;89(3):301-305.
25. Rudge CV, Rollin HB, Nogueira CM, Thomassen Y, Rudge MC, Odland JO. The placenta as a barrier for toxic and essential elements in paired maternal and cord blood samples of South African delivering women. J Environ Monit. 2009;11(7):1322-1330.
26. deSouza RM, Wallace D, Costa ML, Krikler SJ. Transplacental passage of metal ions in women with hip resurfacing: no teratogenic effects observed. Hip Int. 2012;22(1):96-99.
27. Fritzsche J, Borisch C, Schaefer C. Case report: High chromium and cobalt levels in a pregnant patient with bilateral metal-on-metal hip arthroplasties. Clin Orthop. 2012;470(8):2325-2331.
28. Saxena DK, Murthy RC, Jain VK, Chandra SV. Fetoplacental-maternal uptake of hexavalent chromium administered orally in rats and mice. Bull Environ Contam Toxicol. 1990;45(3):430-435.
29. Junaid M, Murthy RC, Saxena DK. Chromium fetotoxicity in mice during late pregnancy. Vet Hum Toxicol. 1995;37(4):320-323.
30. Paternain JL, Domingo JL, Corbella J. Developmental toxicity of cobalt in the rat. J Toxicol Environ Health. 1988;24(2):193-200.
31. Rurak DW. Development and function of the placenta. In: Harding R, Bocking AD, eds. Fetal Growth and Development. Cambridge, UK: Cambridge University Press; 2001.
32. Hsu AR, Levine BR, Skipor AK, Hallab NJ, Paprosky WG, Jacobs JJ. Effect of a second joint arthroplasty on metal ion levels after primary total hip arthroplasty. Am J Orthop. 2013;42(10):E84-E87.