User login
Blog: Pretty Hurts
Malcolm Gladwell wrote in "Outliers" that 10,000 hours of practice is the magic number to become an expert at a trade, which is why Dr. Derek Jones gets a little nervous about physician extenders performing dermatologic surgery procedures.
At the annual meeting of the American Society for Dermatologic Surgery in Washington, Dr. Jones reported that RNs receive 500 hours of clinical training, nurse practitioners receive 500-1,500 hours, physician assistants receive 2,800 hours, and dermatologists receive 18,720 hours.
Taking this into consideration, one can understand why Dr. Jones, who is in private practice in Los Angeles, seemed to be disturbed by the syndicated television program "Pretty Hurts," a soap opera-like reality show that documents Rand Rush, RN, (yes, you read that right: RN) as he injects wanna-be celebrity patients in his Beverly Hills office.
"Pretty Hurts is your insider OMFG view into the ridiculous, painful and ugly truth behind maintaining beauty in Hollywood," according to the show's summary. "Once you experience the extreme stop-at-nothing obsession with youth that has become standard in Beverly Hills, you'll never look at 'pretty' the same way again."
According to Rand's website, he is under the supervision (as required by California law) of Dr. Norman Leaf, a plastic surgeon. But, on the show, he appears to be performing with little to no physician supervision.
So who is to blame for non-physicians performing these aesthetic services? "We are," said Dr. Jones. Dermatologists are organizing and teaching at courses geared towards physician extenders.
While he does not believe that it is inappropriate to teach extenders, he said, "What we need here is a rigorous post-training residency program for physician extenders that levels the playing field. We are not going to see physician extenders go away. The genie is out of the bottle." But, he added, more rigorous training would help protect ourselves, our patients, and the specialty.
Dr. Jones disclosed conflicts of interest with Allergan, Galderma, and Merz.
What do you think? Should physician extenders be allowed to perform cosmetic services, such as laser hair removal and cosmetic injections?
-- Amy Pfeiffer
Malcolm Gladwell wrote in "Outliers" that 10,000 hours of practice is the magic number to become an expert at a trade, which is why Dr. Derek Jones gets a little nervous about physician extenders performing dermatologic surgery procedures.
At the annual meeting of the American Society for Dermatologic Surgery in Washington, Dr. Jones reported that RNs receive 500 hours of clinical training, nurse practitioners receive 500-1,500 hours, physician assistants receive 2,800 hours, and dermatologists receive 18,720 hours.
Taking this into consideration, one can understand why Dr. Jones, who is in private practice in Los Angeles, seemed to be disturbed by the syndicated television program "Pretty Hurts," a soap opera-like reality show that documents Rand Rush, RN, (yes, you read that right: RN) as he injects wanna-be celebrity patients in his Beverly Hills office.
"Pretty Hurts is your insider OMFG view into the ridiculous, painful and ugly truth behind maintaining beauty in Hollywood," according to the show's summary. "Once you experience the extreme stop-at-nothing obsession with youth that has become standard in Beverly Hills, you'll never look at 'pretty' the same way again."
According to Rand's website, he is under the supervision (as required by California law) of Dr. Norman Leaf, a plastic surgeon. But, on the show, he appears to be performing with little to no physician supervision.
So who is to blame for non-physicians performing these aesthetic services? "We are," said Dr. Jones. Dermatologists are organizing and teaching at courses geared towards physician extenders.
While he does not believe that it is inappropriate to teach extenders, he said, "What we need here is a rigorous post-training residency program for physician extenders that levels the playing field. We are not going to see physician extenders go away. The genie is out of the bottle." But, he added, more rigorous training would help protect ourselves, our patients, and the specialty.
Dr. Jones disclosed conflicts of interest with Allergan, Galderma, and Merz.
What do you think? Should physician extenders be allowed to perform cosmetic services, such as laser hair removal and cosmetic injections?
-- Amy Pfeiffer
Malcolm Gladwell wrote in "Outliers" that 10,000 hours of practice is the magic number to become an expert at a trade, which is why Dr. Derek Jones gets a little nervous about physician extenders performing dermatologic surgery procedures.
At the annual meeting of the American Society for Dermatologic Surgery in Washington, Dr. Jones reported that RNs receive 500 hours of clinical training, nurse practitioners receive 500-1,500 hours, physician assistants receive 2,800 hours, and dermatologists receive 18,720 hours.
Taking this into consideration, one can understand why Dr. Jones, who is in private practice in Los Angeles, seemed to be disturbed by the syndicated television program "Pretty Hurts," a soap opera-like reality show that documents Rand Rush, RN, (yes, you read that right: RN) as he injects wanna-be celebrity patients in his Beverly Hills office.
"Pretty Hurts is your insider OMFG view into the ridiculous, painful and ugly truth behind maintaining beauty in Hollywood," according to the show's summary. "Once you experience the extreme stop-at-nothing obsession with youth that has become standard in Beverly Hills, you'll never look at 'pretty' the same way again."
According to Rand's website, he is under the supervision (as required by California law) of Dr. Norman Leaf, a plastic surgeon. But, on the show, he appears to be performing with little to no physician supervision.
So who is to blame for non-physicians performing these aesthetic services? "We are," said Dr. Jones. Dermatologists are organizing and teaching at courses geared towards physician extenders.
While he does not believe that it is inappropriate to teach extenders, he said, "What we need here is a rigorous post-training residency program for physician extenders that levels the playing field. We are not going to see physician extenders go away. The genie is out of the bottle." But, he added, more rigorous training would help protect ourselves, our patients, and the specialty.
Dr. Jones disclosed conflicts of interest with Allergan, Galderma, and Merz.
What do you think? Should physician extenders be allowed to perform cosmetic services, such as laser hair removal and cosmetic injections?
-- Amy Pfeiffer
Autologous Cells Improve Acne Scarring
WASHINGTON – The autologous cellular product azficel-T produced significant improvement in acne scarring compared with placebo, according to new study results reported at the annual meeting of the American Society for Dermatologic Surgery.
The product, Laviv (Fibrocell Science), was recently approved by the Food and Drug Administration for the treatment of moderate to severe nasolabial folds. It is the first FDA-approved personalized cell therapy for aesthetic use, according to Dr. Girish Munavalli of the dermatology department at Wake Forest University, Winston-Salem, N.C.
In the multicenter, randomized, double-blind, placebo-controlled study, skin biopsies were collected from 119 patients with moderate-to-severe depressed acne scarring of at least 9 cm2 for at least 3 years. The biopsies were used to produce individual fibroblasts for each patient. Up to three injections of 2 mL of autologous fibroblasts (10-20 million cells/mL) were given on one cheek and placebo on the other at 14-day intervals in 99 of the patients. Treatment was administered at a dose of 0.1 mL/cm2 into areas of acne scarring. Adverse events were recorded for each cheek.
At 4 months after completion of the treatment, a statistically significant higher percentage of patients had responded to treatment with azficel-T than to treatment with placebo, as rated by both the study investigators (58.7% vs. 42.2%, respectively) and patients (43.1% vs. 18.3%). Patient and evaluator assessments at earlier time points during the study also showed a significantly higher proportion of responses with azficel-T than with placebo at all but one assessment, reported Dr. Munavalli.
The study successfully met its two prospectively defined end points: a 2-point or greater improvement on a 5-point Subject Live Acne Scarring assessment scale, and a 1-point or greater reduction in cheek acne severity on a physician-assessed, validated 5-point Evaluator Live Acne Scar assessment scale.
All reported adverse events were mild or moderate in severity. No subjects experienced serious adverse events, discontinued treatment, or withdrew from the study. The incidence of adverse events occurring in the treatment areas was comparable between azficel-T and placebo. The most commonly reported adverse events were treatment-area erythema (occurring in 11.1%) and swelling (10.1%). In the azficel-T-treated area, erythema was moderate in 5 of the 12 subjects who reported it, and swelling was moderate in 5 of the 11 who reported it. In contrast, all treatment-related adverse events in the placebo-treated areas were mild.
Other adverse events with a possible relationship to study treatment included bruising, rash, irritation, nodules, pain, acne, induration, and headache, Dr. Munavalli reported.
"Our findings show that using a person's own collagen-producing cells in the form of Laviv may provide a promising new alternative to improve acne scarring with minimal downtime and an excellent safety profile," he noted.
Dr. Munavalli received research funding from Fibrocell Science.
WASHINGTON – The autologous cellular product azficel-T produced significant improvement in acne scarring compared with placebo, according to new study results reported at the annual meeting of the American Society for Dermatologic Surgery.
The product, Laviv (Fibrocell Science), was recently approved by the Food and Drug Administration for the treatment of moderate to severe nasolabial folds. It is the first FDA-approved personalized cell therapy for aesthetic use, according to Dr. Girish Munavalli of the dermatology department at Wake Forest University, Winston-Salem, N.C.
In the multicenter, randomized, double-blind, placebo-controlled study, skin biopsies were collected from 119 patients with moderate-to-severe depressed acne scarring of at least 9 cm2 for at least 3 years. The biopsies were used to produce individual fibroblasts for each patient. Up to three injections of 2 mL of autologous fibroblasts (10-20 million cells/mL) were given on one cheek and placebo on the other at 14-day intervals in 99 of the patients. Treatment was administered at a dose of 0.1 mL/cm2 into areas of acne scarring. Adverse events were recorded for each cheek.
At 4 months after completion of the treatment, a statistically significant higher percentage of patients had responded to treatment with azficel-T than to treatment with placebo, as rated by both the study investigators (58.7% vs. 42.2%, respectively) and patients (43.1% vs. 18.3%). Patient and evaluator assessments at earlier time points during the study also showed a significantly higher proportion of responses with azficel-T than with placebo at all but one assessment, reported Dr. Munavalli.
The study successfully met its two prospectively defined end points: a 2-point or greater improvement on a 5-point Subject Live Acne Scarring assessment scale, and a 1-point or greater reduction in cheek acne severity on a physician-assessed, validated 5-point Evaluator Live Acne Scar assessment scale.
All reported adverse events were mild or moderate in severity. No subjects experienced serious adverse events, discontinued treatment, or withdrew from the study. The incidence of adverse events occurring in the treatment areas was comparable between azficel-T and placebo. The most commonly reported adverse events were treatment-area erythema (occurring in 11.1%) and swelling (10.1%). In the azficel-T-treated area, erythema was moderate in 5 of the 12 subjects who reported it, and swelling was moderate in 5 of the 11 who reported it. In contrast, all treatment-related adverse events in the placebo-treated areas were mild.
Other adverse events with a possible relationship to study treatment included bruising, rash, irritation, nodules, pain, acne, induration, and headache, Dr. Munavalli reported.
"Our findings show that using a person's own collagen-producing cells in the form of Laviv may provide a promising new alternative to improve acne scarring with minimal downtime and an excellent safety profile," he noted.
Dr. Munavalli received research funding from Fibrocell Science.
WASHINGTON – The autologous cellular product azficel-T produced significant improvement in acne scarring compared with placebo, according to new study results reported at the annual meeting of the American Society for Dermatologic Surgery.
The product, Laviv (Fibrocell Science), was recently approved by the Food and Drug Administration for the treatment of moderate to severe nasolabial folds. It is the first FDA-approved personalized cell therapy for aesthetic use, according to Dr. Girish Munavalli of the dermatology department at Wake Forest University, Winston-Salem, N.C.
In the multicenter, randomized, double-blind, placebo-controlled study, skin biopsies were collected from 119 patients with moderate-to-severe depressed acne scarring of at least 9 cm2 for at least 3 years. The biopsies were used to produce individual fibroblasts for each patient. Up to three injections of 2 mL of autologous fibroblasts (10-20 million cells/mL) were given on one cheek and placebo on the other at 14-day intervals in 99 of the patients. Treatment was administered at a dose of 0.1 mL/cm2 into areas of acne scarring. Adverse events were recorded for each cheek.
At 4 months after completion of the treatment, a statistically significant higher percentage of patients had responded to treatment with azficel-T than to treatment with placebo, as rated by both the study investigators (58.7% vs. 42.2%, respectively) and patients (43.1% vs. 18.3%). Patient and evaluator assessments at earlier time points during the study also showed a significantly higher proportion of responses with azficel-T than with placebo at all but one assessment, reported Dr. Munavalli.
The study successfully met its two prospectively defined end points: a 2-point or greater improvement on a 5-point Subject Live Acne Scarring assessment scale, and a 1-point or greater reduction in cheek acne severity on a physician-assessed, validated 5-point Evaluator Live Acne Scar assessment scale.
All reported adverse events were mild or moderate in severity. No subjects experienced serious adverse events, discontinued treatment, or withdrew from the study. The incidence of adverse events occurring in the treatment areas was comparable between azficel-T and placebo. The most commonly reported adverse events were treatment-area erythema (occurring in 11.1%) and swelling (10.1%). In the azficel-T-treated area, erythema was moderate in 5 of the 12 subjects who reported it, and swelling was moderate in 5 of the 11 who reported it. In contrast, all treatment-related adverse events in the placebo-treated areas were mild.
Other adverse events with a possible relationship to study treatment included bruising, rash, irritation, nodules, pain, acne, induration, and headache, Dr. Munavalli reported.
"Our findings show that using a person's own collagen-producing cells in the form of Laviv may provide a promising new alternative to improve acne scarring with minimal downtime and an excellent safety profile," he noted.
Dr. Munavalli received research funding from Fibrocell Science.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY FOR DERMATOLOGIC SURGERY
Major Finding: At 4 months after completion of the treatment, a statistically significant higher percentage of patients had responded to treatment with Laviv than to treatment with placebo, as rated by both the study investigators (58.7% vs. 42.2%, respectively) and patients (43.1% vs. 18.3%).
Data Source: A multicenter, randomized, double-blind, placebo-controlled study of 119 patients who had moderate to severe depressed acne scarring of at least 9 cm2 for at least 3 years.
Disclosures: Dr. Munavalli received research funding from Fibrocell Science.
Light-Based Treatment of Pigmented Lesions
Fractional Lasers for Acne Scarring in Patients With Skin of Color
A Survey Examination of Patients With Hydroa Vacciniforme
Survival Similar With 2- vs. 4-cm Margins for Thick Melanomas
A 2-cm resection margin resulted in the same 5-year overall survival and recurrence-free survival as did a 4-cm margin in a randomized controlled trial of more than 900 patients with cutaneous melanoma thicker than 2 mm.
The findings suggest that in patients with thicker melanomas, a 2-cm margin is safe and sufficient, according to Dr. Peter Gillgren of Karolinska Institute, Stockholm, and Stockholm Söder Hospital and his colleagues.
At a median follow-up of 6.7 years, the 5-year overall survival was 65% in 465 patients randomized to treatment with a 2-cm surgical resection margin, as well as in 471 patients randomized to treatment with a 4-cm resection margin. Recurrence-free survival at 5 years was 56% in both groups, and 10-year survival was 50% in both groups, the investigators reported (Lancet 2011 Oct. 24 [doi:10.1016/S0140-6736(11)61546-8]).
"The failure to clear genetically abnormal melanocytes with an adequately wide excision might be the precursor to locoregional recurrence."
Surgical resection margin size in patients with cutaneous melanoma thicker than 2 mm has been a point of controversy, largely due to a paucity of data comparing outcomes based on margin size, but most international guidelines suggest an excision margin of 2-3 cm for thick melanomas, the investigators noted. "A trade-off exists between a wide excision, with consequent surgical difficulties, and the relapse risk with a narrow excision, which could compromise disease-free survival, or worse, overall survival," they wrote.
However, the findings of this study indicate that not only is survival similar with 2-cm and 4-cm margins, but that the smaller margin size also improves the likelihood of skin closure without skin grafting or skin flaps. Primary closure was possible in 69% vs. 37% of patients in the 2-cm and 4-cm groups, respectively, while split skin grafts were used in 12% and 47% of the patients in the two groups. And surgical flaps were used in 4% and 6% of patients in the two groups, noted the investigators.
Patients in this study, which was launched by the Swedish Melanoma Study Group in cooperation with the Danish Melanoma Group, were adults aged 75 years or younger with a primary cutaneous melanoma thicker than 2 mm and with clinically localized disease on the trunk or upper or lower extremities. Patients were enrolled from Jan. 22, 1992, to May 19, 2004. One patient in each group was lost to follow-up but was included in the analysis.
Despite some limitations, such as protocol violations in 15% of cases and the fact that the study was planned as an equivalency trial that was to include 2,000 patients, the investigators noted that the study is the largest randomized controlled trial to date of resection margins for thick melanomas, and that they believe the results provide "the best evidence yet about the size of surgical excision margins."
"We show that with a surgical margin of 2 cm, the skin can be closed without skin grafting or skin flaps in most cases," they wrote, noting that previous data have already shown that hospital stay is longer in patients treated with a 4-cm margin, and that complication rates are higher in patients treated with split skin grafts, compared with primary sutures.
A meta-analysis of all randomized trials of cutaneous melanoma thicker than 2 mm should be conducted, they concluded.
The study was funded by the Swedish Cancer Society and the Stockholm Cancer Society. The authors reported having no relevant financial disclosures.
In an editorial that accompanied the article by Dr. Gillgren and
colleagues, Dr. John F. Thompson and Dr. David W. Ollila reiterated that
the optimal excisional margin size for cutaneous melanoma has not been
clear. These findings, therefore, provide welcome news, but the next
question to be addressed is whether a 2-cm margin is preferable to a
1-cm margin, they wrote, noting that a large-scale multicenter trial to
address this question is in development (Lancet 2011 Oct. 24 [doi:10.1016/S0140-6736(11)61615-2]).

|
|
Another area of importance is "proper understanding of the inherent
tumor biology necessary for a safe excision margin," they noted,
explaining that assessment of margins using hematoxylin and eosin
staining is a "relatively crude pathological technique."
Investigators in another recent study used "comparative genomic
hybridization and fluorescent in situ hybridization to identify and map
genetically abnormal melanocytes in histopathologically normal epidermis
in acral melanoma wide excision specimens," Dr. Thompson of the
Melanoma Institute Australia at the Poche Centre in North Sydney and Dr.
Ollila of the University of North Carolina at Chapel Hill wrote.
They found abnormal melanocytes – extending a mean of 6.1 mm and 4.5
mm from the histologically assessed margin of in situ melanomas and
invasive melanomas, respectively – in 84% of 19 cases (J. Invest. Dermatol. 2008;128:2024-30).
"The failure to clear genetically abnormal melanocytes with an
adequately wide excision might be the precursor to locoregional
recurrence, which in turn could reduce survival," the investigators
wrote, noting that sophisticated multidisciplinary science may provide
the most rational approach to future recommendations for excisional
margin size in melanoma patients.
Dr. Thompson is with the Melanoma Institute Australia at the Poche Centre in North Sydney. Dr. Ollila is with the division of surgical oncology and endocrine surgery at the University of North Carolina at Chapel Hill. They reported having no relevant financial disclosures.
In an editorial that accompanied the article by Dr. Gillgren and
colleagues, Dr. John F. Thompson and Dr. David W. Ollila reiterated that
the optimal excisional margin size for cutaneous melanoma has not been
clear. These findings, therefore, provide welcome news, but the next
question to be addressed is whether a 2-cm margin is preferable to a
1-cm margin, they wrote, noting that a large-scale multicenter trial to
address this question is in development (Lancet 2011 Oct. 24 [doi:10.1016/S0140-6736(11)61615-2]).

|
|
Another area of importance is "proper understanding of the inherent
tumor biology necessary for a safe excision margin," they noted,
explaining that assessment of margins using hematoxylin and eosin
staining is a "relatively crude pathological technique."
Investigators in another recent study used "comparative genomic
hybridization and fluorescent in situ hybridization to identify and map
genetically abnormal melanocytes in histopathologically normal epidermis
in acral melanoma wide excision specimens," Dr. Thompson of the
Melanoma Institute Australia at the Poche Centre in North Sydney and Dr.
Ollila of the University of North Carolina at Chapel Hill wrote.
They found abnormal melanocytes – extending a mean of 6.1 mm and 4.5
mm from the histologically assessed margin of in situ melanomas and
invasive melanomas, respectively – in 84% of 19 cases (J. Invest. Dermatol. 2008;128:2024-30).
"The failure to clear genetically abnormal melanocytes with an
adequately wide excision might be the precursor to locoregional
recurrence, which in turn could reduce survival," the investigators
wrote, noting that sophisticated multidisciplinary science may provide
the most rational approach to future recommendations for excisional
margin size in melanoma patients.
Dr. Thompson is with the Melanoma Institute Australia at the Poche Centre in North Sydney. Dr. Ollila is with the division of surgical oncology and endocrine surgery at the University of North Carolina at Chapel Hill. They reported having no relevant financial disclosures.
In an editorial that accompanied the article by Dr. Gillgren and
colleagues, Dr. John F. Thompson and Dr. David W. Ollila reiterated that
the optimal excisional margin size for cutaneous melanoma has not been
clear. These findings, therefore, provide welcome news, but the next
question to be addressed is whether a 2-cm margin is preferable to a
1-cm margin, they wrote, noting that a large-scale multicenter trial to
address this question is in development (Lancet 2011 Oct. 24 [doi:10.1016/S0140-6736(11)61615-2]).

|
|
Another area of importance is "proper understanding of the inherent
tumor biology necessary for a safe excision margin," they noted,
explaining that assessment of margins using hematoxylin and eosin
staining is a "relatively crude pathological technique."
Investigators in another recent study used "comparative genomic
hybridization and fluorescent in situ hybridization to identify and map
genetically abnormal melanocytes in histopathologically normal epidermis
in acral melanoma wide excision specimens," Dr. Thompson of the
Melanoma Institute Australia at the Poche Centre in North Sydney and Dr.
Ollila of the University of North Carolina at Chapel Hill wrote.
They found abnormal melanocytes – extending a mean of 6.1 mm and 4.5
mm from the histologically assessed margin of in situ melanomas and
invasive melanomas, respectively – in 84% of 19 cases (J. Invest. Dermatol. 2008;128:2024-30).
"The failure to clear genetically abnormal melanocytes with an
adequately wide excision might be the precursor to locoregional
recurrence, which in turn could reduce survival," the investigators
wrote, noting that sophisticated multidisciplinary science may provide
the most rational approach to future recommendations for excisional
margin size in melanoma patients.
Dr. Thompson is with the Melanoma Institute Australia at the Poche Centre in North Sydney. Dr. Ollila is with the division of surgical oncology and endocrine surgery at the University of North Carolina at Chapel Hill. They reported having no relevant financial disclosures.
A 2-cm resection margin resulted in the same 5-year overall survival and recurrence-free survival as did a 4-cm margin in a randomized controlled trial of more than 900 patients with cutaneous melanoma thicker than 2 mm.
The findings suggest that in patients with thicker melanomas, a 2-cm margin is safe and sufficient, according to Dr. Peter Gillgren of Karolinska Institute, Stockholm, and Stockholm Söder Hospital and his colleagues.
At a median follow-up of 6.7 years, the 5-year overall survival was 65% in 465 patients randomized to treatment with a 2-cm surgical resection margin, as well as in 471 patients randomized to treatment with a 4-cm resection margin. Recurrence-free survival at 5 years was 56% in both groups, and 10-year survival was 50% in both groups, the investigators reported (Lancet 2011 Oct. 24 [doi:10.1016/S0140-6736(11)61546-8]).
"The failure to clear genetically abnormal melanocytes with an adequately wide excision might be the precursor to locoregional recurrence."
Surgical resection margin size in patients with cutaneous melanoma thicker than 2 mm has been a point of controversy, largely due to a paucity of data comparing outcomes based on margin size, but most international guidelines suggest an excision margin of 2-3 cm for thick melanomas, the investigators noted. "A trade-off exists between a wide excision, with consequent surgical difficulties, and the relapse risk with a narrow excision, which could compromise disease-free survival, or worse, overall survival," they wrote.
However, the findings of this study indicate that not only is survival similar with 2-cm and 4-cm margins, but that the smaller margin size also improves the likelihood of skin closure without skin grafting or skin flaps. Primary closure was possible in 69% vs. 37% of patients in the 2-cm and 4-cm groups, respectively, while split skin grafts were used in 12% and 47% of the patients in the two groups. And surgical flaps were used in 4% and 6% of patients in the two groups, noted the investigators.
Patients in this study, which was launched by the Swedish Melanoma Study Group in cooperation with the Danish Melanoma Group, were adults aged 75 years or younger with a primary cutaneous melanoma thicker than 2 mm and with clinically localized disease on the trunk or upper or lower extremities. Patients were enrolled from Jan. 22, 1992, to May 19, 2004. One patient in each group was lost to follow-up but was included in the analysis.
Despite some limitations, such as protocol violations in 15% of cases and the fact that the study was planned as an equivalency trial that was to include 2,000 patients, the investigators noted that the study is the largest randomized controlled trial to date of resection margins for thick melanomas, and that they believe the results provide "the best evidence yet about the size of surgical excision margins."
"We show that with a surgical margin of 2 cm, the skin can be closed without skin grafting or skin flaps in most cases," they wrote, noting that previous data have already shown that hospital stay is longer in patients treated with a 4-cm margin, and that complication rates are higher in patients treated with split skin grafts, compared with primary sutures.
A meta-analysis of all randomized trials of cutaneous melanoma thicker than 2 mm should be conducted, they concluded.
The study was funded by the Swedish Cancer Society and the Stockholm Cancer Society. The authors reported having no relevant financial disclosures.
A 2-cm resection margin resulted in the same 5-year overall survival and recurrence-free survival as did a 4-cm margin in a randomized controlled trial of more than 900 patients with cutaneous melanoma thicker than 2 mm.
The findings suggest that in patients with thicker melanomas, a 2-cm margin is safe and sufficient, according to Dr. Peter Gillgren of Karolinska Institute, Stockholm, and Stockholm Söder Hospital and his colleagues.
At a median follow-up of 6.7 years, the 5-year overall survival was 65% in 465 patients randomized to treatment with a 2-cm surgical resection margin, as well as in 471 patients randomized to treatment with a 4-cm resection margin. Recurrence-free survival at 5 years was 56% in both groups, and 10-year survival was 50% in both groups, the investigators reported (Lancet 2011 Oct. 24 [doi:10.1016/S0140-6736(11)61546-8]).
"The failure to clear genetically abnormal melanocytes with an adequately wide excision might be the precursor to locoregional recurrence."
Surgical resection margin size in patients with cutaneous melanoma thicker than 2 mm has been a point of controversy, largely due to a paucity of data comparing outcomes based on margin size, but most international guidelines suggest an excision margin of 2-3 cm for thick melanomas, the investigators noted. "A trade-off exists between a wide excision, with consequent surgical difficulties, and the relapse risk with a narrow excision, which could compromise disease-free survival, or worse, overall survival," they wrote.
However, the findings of this study indicate that not only is survival similar with 2-cm and 4-cm margins, but that the smaller margin size also improves the likelihood of skin closure without skin grafting or skin flaps. Primary closure was possible in 69% vs. 37% of patients in the 2-cm and 4-cm groups, respectively, while split skin grafts were used in 12% and 47% of the patients in the two groups. And surgical flaps were used in 4% and 6% of patients in the two groups, noted the investigators.
Patients in this study, which was launched by the Swedish Melanoma Study Group in cooperation with the Danish Melanoma Group, were adults aged 75 years or younger with a primary cutaneous melanoma thicker than 2 mm and with clinically localized disease on the trunk or upper or lower extremities. Patients were enrolled from Jan. 22, 1992, to May 19, 2004. One patient in each group was lost to follow-up but was included in the analysis.
Despite some limitations, such as protocol violations in 15% of cases and the fact that the study was planned as an equivalency trial that was to include 2,000 patients, the investigators noted that the study is the largest randomized controlled trial to date of resection margins for thick melanomas, and that they believe the results provide "the best evidence yet about the size of surgical excision margins."
"We show that with a surgical margin of 2 cm, the skin can be closed without skin grafting or skin flaps in most cases," they wrote, noting that previous data have already shown that hospital stay is longer in patients treated with a 4-cm margin, and that complication rates are higher in patients treated with split skin grafts, compared with primary sutures.
A meta-analysis of all randomized trials of cutaneous melanoma thicker than 2 mm should be conducted, they concluded.
The study was funded by the Swedish Cancer Society and the Stockholm Cancer Society. The authors reported having no relevant financial disclosures.
FROM THE LANCET
Major Finding: At a median follow-up of 6.7 years, the 5-year overall survival was 65% in 465 patients randomized to treatment with a 2-cm surgical resection margin, as well as in 471 patients randomized to treatment with a 4-cm resection margin.
Data Source: A multicenter, randomized controlled trial of 936 patients aged 75 years or younger with a primary cutaneous melanoma thicker than 2 mm.
Disclosures: This study was funded by the Swedish Cancer Society and the Stockholm Cancer Society. The authors reported having no relevant financial disclosures.
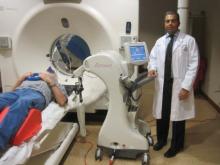
Mini X-Ray Device Treats Nonmelanoma Skin Cancers
MIAMI BEACH – An x-ray tube about the size of a sewing needle can safely deliver radiation to nonmelanoma skin tumors on the nose, scalp, or other highly visible areas of skin with good efficacy and good to excellent cosmetic results, an investigator reported at the annual meeting of the American Society for Radiation Oncology.
About 93% of patients treated with an electronic brachytherapy system from Xoft had cosmetic results judged to meet Radiation Therapy Oncology Group (RTOG) criteria for "excellent" at 1 year, said Dr. Ajay Bhatnagar of Cancer Treatment Services Arizona, Casa Grande.
"To date, the treatment of nonmelanoma skin cancer with electronic brachytherapy is comparable to traditional brachytherapy, with excellent cosmesis, acceptable acute toxicities, and no recurrences to date. Brachytherapy is an excellent modality for elderly patients with nonmelanoma skin cancers in cosmetically sensitive locations," he said.
The electronic brachytherapy system delivers a radiation dose to tumors equivalent to that of an iridium-192, high dose rate surface (Leipzig) applicator, but without the need for a radioisotope. The electronic device is a miniature, high dose rate, low-energy x-ray tube that produces x-rays with a maximum of 50 keV of energy.
Dr. Bhatnagar looked at data on 102 patients treated with the device for 120 nonmelanoma skin cancer lesions. All patients received 5 Gy in eight dose fractions for a total of 40 Gy delivered in two fractions per week for 4 weeks. The prescription depth, based on CT simulation, varied from 3 to 7 mm. The treatment goal was to achieve a minimum 5-mm treatment margin wherever possible.
The device was placed with the patient immobilized and wearing a flex shield that prevents excessive radiation exposure to the clinician. Skin care included application of petrolatum ointment during treatment and aloe vera gel for 1 month afterward.
Of the 120 lesions treated, 59% had basal cell histology, 36% were squamous cell, 2% were Merkel cell carcinomas, 3% were cutaneous T-cell lymphoma lesions, and 1% had a mixed basal-squamous histology.
About one-third of the lesions were on the nose, another third on the face, with the remainder on the ears, extremities, scalp, and torso.
Nearly all patients (91%) experienced an acute rash with treatment, and 25% had itching of the treatment site. The most frequent late adverse effect was hypopigmentation of the treated skin, which occurred in 11% of lesions from 3 to 6 months.
At 1 month post treatment, cosmesis was rated as excellent (no change to slight atrophy or pigment change; slight hair loss; or no changes to slight induration or loss of subcutaneous fat) in about 77% of 81 patients evaluated; the remainder had "good" cosmesis (patch atrophy, moderate telangiectasia, total hair loss; moderate fibrosis but asymptomatic, slight field contracture with less than 10% linear reduction).
A physician who was not involved in the study commented that radiation therapy offers certain advantages over surgery when tumors are located in prominent locations, such as the face.
"We’ve been treating these lesions for close to 100 years now, and radiation does very well," said Dr. Keith A. Cengel, of the University of Pennsylvania School of Medicine in Philadelphia. "The one drawback – and I tell this to my patients when I treat with radiation – is that [subsequent] surgery can be more difficult and have a higher complication rate if you have a recurrence."
Mohs surgery, the most common form of treatment for basal or squamous cell skin cancers, may itself leave a comparatively deep and wide wound that may not heal as smoothly as a radiation-treated lesion, he added.
Dr. Cengel comoderated the session in which the electronic brachytherapy data were presented.
The study was sponsored by Xoft, manufacturer of the brachytherapy device. Dr. Bhatnagar receives funding for serving as principal investigator of a prospective multicenter study of the device.
MIAMI BEACH – An x-ray tube about the size of a sewing needle can safely deliver radiation to nonmelanoma skin tumors on the nose, scalp, or other highly visible areas of skin with good efficacy and good to excellent cosmetic results, an investigator reported at the annual meeting of the American Society for Radiation Oncology.
About 93% of patients treated with an electronic brachytherapy system from Xoft had cosmetic results judged to meet Radiation Therapy Oncology Group (RTOG) criteria for "excellent" at 1 year, said Dr. Ajay Bhatnagar of Cancer Treatment Services Arizona, Casa Grande.
"To date, the treatment of nonmelanoma skin cancer with electronic brachytherapy is comparable to traditional brachytherapy, with excellent cosmesis, acceptable acute toxicities, and no recurrences to date. Brachytherapy is an excellent modality for elderly patients with nonmelanoma skin cancers in cosmetically sensitive locations," he said.
The electronic brachytherapy system delivers a radiation dose to tumors equivalent to that of an iridium-192, high dose rate surface (Leipzig) applicator, but without the need for a radioisotope. The electronic device is a miniature, high dose rate, low-energy x-ray tube that produces x-rays with a maximum of 50 keV of energy.
Dr. Bhatnagar looked at data on 102 patients treated with the device for 120 nonmelanoma skin cancer lesions. All patients received 5 Gy in eight dose fractions for a total of 40 Gy delivered in two fractions per week for 4 weeks. The prescription depth, based on CT simulation, varied from 3 to 7 mm. The treatment goal was to achieve a minimum 5-mm treatment margin wherever possible.
The device was placed with the patient immobilized and wearing a flex shield that prevents excessive radiation exposure to the clinician. Skin care included application of petrolatum ointment during treatment and aloe vera gel for 1 month afterward.
Of the 120 lesions treated, 59% had basal cell histology, 36% were squamous cell, 2% were Merkel cell carcinomas, 3% were cutaneous T-cell lymphoma lesions, and 1% had a mixed basal-squamous histology.
About one-third of the lesions were on the nose, another third on the face, with the remainder on the ears, extremities, scalp, and torso.
Nearly all patients (91%) experienced an acute rash with treatment, and 25% had itching of the treatment site. The most frequent late adverse effect was hypopigmentation of the treated skin, which occurred in 11% of lesions from 3 to 6 months.
At 1 month post treatment, cosmesis was rated as excellent (no change to slight atrophy or pigment change; slight hair loss; or no changes to slight induration or loss of subcutaneous fat) in about 77% of 81 patients evaluated; the remainder had "good" cosmesis (patch atrophy, moderate telangiectasia, total hair loss; moderate fibrosis but asymptomatic, slight field contracture with less than 10% linear reduction).
A physician who was not involved in the study commented that radiation therapy offers certain advantages over surgery when tumors are located in prominent locations, such as the face.
"We’ve been treating these lesions for close to 100 years now, and radiation does very well," said Dr. Keith A. Cengel, of the University of Pennsylvania School of Medicine in Philadelphia. "The one drawback – and I tell this to my patients when I treat with radiation – is that [subsequent] surgery can be more difficult and have a higher complication rate if you have a recurrence."
Mohs surgery, the most common form of treatment for basal or squamous cell skin cancers, may itself leave a comparatively deep and wide wound that may not heal as smoothly as a radiation-treated lesion, he added.
Dr. Cengel comoderated the session in which the electronic brachytherapy data were presented.
The study was sponsored by Xoft, manufacturer of the brachytherapy device. Dr. Bhatnagar receives funding for serving as principal investigator of a prospective multicenter study of the device.
MIAMI BEACH – An x-ray tube about the size of a sewing needle can safely deliver radiation to nonmelanoma skin tumors on the nose, scalp, or other highly visible areas of skin with good efficacy and good to excellent cosmetic results, an investigator reported at the annual meeting of the American Society for Radiation Oncology.
About 93% of patients treated with an electronic brachytherapy system from Xoft had cosmetic results judged to meet Radiation Therapy Oncology Group (RTOG) criteria for "excellent" at 1 year, said Dr. Ajay Bhatnagar of Cancer Treatment Services Arizona, Casa Grande.
"To date, the treatment of nonmelanoma skin cancer with electronic brachytherapy is comparable to traditional brachytherapy, with excellent cosmesis, acceptable acute toxicities, and no recurrences to date. Brachytherapy is an excellent modality for elderly patients with nonmelanoma skin cancers in cosmetically sensitive locations," he said.
The electronic brachytherapy system delivers a radiation dose to tumors equivalent to that of an iridium-192, high dose rate surface (Leipzig) applicator, but without the need for a radioisotope. The electronic device is a miniature, high dose rate, low-energy x-ray tube that produces x-rays with a maximum of 50 keV of energy.
Dr. Bhatnagar looked at data on 102 patients treated with the device for 120 nonmelanoma skin cancer lesions. All patients received 5 Gy in eight dose fractions for a total of 40 Gy delivered in two fractions per week for 4 weeks. The prescription depth, based on CT simulation, varied from 3 to 7 mm. The treatment goal was to achieve a minimum 5-mm treatment margin wherever possible.
The device was placed with the patient immobilized and wearing a flex shield that prevents excessive radiation exposure to the clinician. Skin care included application of petrolatum ointment during treatment and aloe vera gel for 1 month afterward.
Of the 120 lesions treated, 59% had basal cell histology, 36% were squamous cell, 2% were Merkel cell carcinomas, 3% were cutaneous T-cell lymphoma lesions, and 1% had a mixed basal-squamous histology.
About one-third of the lesions were on the nose, another third on the face, with the remainder on the ears, extremities, scalp, and torso.
Nearly all patients (91%) experienced an acute rash with treatment, and 25% had itching of the treatment site. The most frequent late adverse effect was hypopigmentation of the treated skin, which occurred in 11% of lesions from 3 to 6 months.
At 1 month post treatment, cosmesis was rated as excellent (no change to slight atrophy or pigment change; slight hair loss; or no changes to slight induration or loss of subcutaneous fat) in about 77% of 81 patients evaluated; the remainder had "good" cosmesis (patch atrophy, moderate telangiectasia, total hair loss; moderate fibrosis but asymptomatic, slight field contracture with less than 10% linear reduction).
A physician who was not involved in the study commented that radiation therapy offers certain advantages over surgery when tumors are located in prominent locations, such as the face.
"We’ve been treating these lesions for close to 100 years now, and radiation does very well," said Dr. Keith A. Cengel, of the University of Pennsylvania School of Medicine in Philadelphia. "The one drawback – and I tell this to my patients when I treat with radiation – is that [subsequent] surgery can be more difficult and have a higher complication rate if you have a recurrence."
Mohs surgery, the most common form of treatment for basal or squamous cell skin cancers, may itself leave a comparatively deep and wide wound that may not heal as smoothly as a radiation-treated lesion, he added.
Dr. Cengel comoderated the session in which the electronic brachytherapy data were presented.
The study was sponsored by Xoft, manufacturer of the brachytherapy device. Dr. Bhatnagar receives funding for serving as principal investigator of a prospective multicenter study of the device.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY FOR RADIATION ONCOLOGY
Major Finding: Approximately 93% of patients treated for nonmelanoma skin cancers with an electronic brachytherapy device had cosmetic results rated as excellent at 1 year, according to Radiation Therapy Oncology Group criteria.
Data Source: Retrospective case study of 102 patients with 120 basal cell, squamous cell, or other nonmelanoma cancers
Disclosures: The study was sponsored by Xoft, manufacturer of the brachytherapy device. Dr. Bhatnagar receives funding for serving as principal investigator of a prospective multicenter study of the device.
Restylane Receives Indication for Lip Augmentation
The Food and Drug Administration has approved Restylane for lip augmentation, the manufacturer announced Oct. 11.
The hyaluronic acid dermal filler was first approved in 2005 for mid to deep dermal implantation for the correction of moderate to severe facial wrinkles and folds, and has been used off-label for lip augmentation. Restylane (Medicis Aesthetics) is a hyaluronic acid gel generated by Streptococcus bacteria, chemically crosslinked with 1,4 butanediol diglycidyl ether.
At a meeting in April, an FDA advisory panel voted 6-0 with 1 abstention that the benefits of using the filler as a submucosal injection for lip augmentation outweighed its risks, and that the filler was safe and effective for the expanded indication.
At that meeting, the panel reviewed the results of a study of 135 patients who received lip augmentation with Restylane (with a mean filler volume of 2.9 cc per patient, with a range of 0.6-5.6 cc per patient) and 45 patients with no treatment. At 8 weeks, 92% of patients who received Restylane were considered responders; almost all patients (99%) experienced adverse events, which included expected treatment-emergent adverse events such as bruising, redness, swelling, pain, tenderness, itching, and skin exfoliation. Of those who were treated, 40% experienced adverse outcomes that they felt affected their daily activity or were disabling, and 15% experienced adverse events (typically swelling and tenderness) that lasted more than 15 days.
Among the concerns expressed by the panel was the lack of men and people with dark skin in the study.
The Food and Drug Administration has approved Restylane for lip augmentation, the manufacturer announced Oct. 11.
The hyaluronic acid dermal filler was first approved in 2005 for mid to deep dermal implantation for the correction of moderate to severe facial wrinkles and folds, and has been used off-label for lip augmentation. Restylane (Medicis Aesthetics) is a hyaluronic acid gel generated by Streptococcus bacteria, chemically crosslinked with 1,4 butanediol diglycidyl ether.
At a meeting in April, an FDA advisory panel voted 6-0 with 1 abstention that the benefits of using the filler as a submucosal injection for lip augmentation outweighed its risks, and that the filler was safe and effective for the expanded indication.
At that meeting, the panel reviewed the results of a study of 135 patients who received lip augmentation with Restylane (with a mean filler volume of 2.9 cc per patient, with a range of 0.6-5.6 cc per patient) and 45 patients with no treatment. At 8 weeks, 92% of patients who received Restylane were considered responders; almost all patients (99%) experienced adverse events, which included expected treatment-emergent adverse events such as bruising, redness, swelling, pain, tenderness, itching, and skin exfoliation. Of those who were treated, 40% experienced adverse outcomes that they felt affected their daily activity or were disabling, and 15% experienced adverse events (typically swelling and tenderness) that lasted more than 15 days.
Among the concerns expressed by the panel was the lack of men and people with dark skin in the study.
The Food and Drug Administration has approved Restylane for lip augmentation, the manufacturer announced Oct. 11.
The hyaluronic acid dermal filler was first approved in 2005 for mid to deep dermal implantation for the correction of moderate to severe facial wrinkles and folds, and has been used off-label for lip augmentation. Restylane (Medicis Aesthetics) is a hyaluronic acid gel generated by Streptococcus bacteria, chemically crosslinked with 1,4 butanediol diglycidyl ether.
At a meeting in April, an FDA advisory panel voted 6-0 with 1 abstention that the benefits of using the filler as a submucosal injection for lip augmentation outweighed its risks, and that the filler was safe and effective for the expanded indication.
At that meeting, the panel reviewed the results of a study of 135 patients who received lip augmentation with Restylane (with a mean filler volume of 2.9 cc per patient, with a range of 0.6-5.6 cc per patient) and 45 patients with no treatment. At 8 weeks, 92% of patients who received Restylane were considered responders; almost all patients (99%) experienced adverse events, which included expected treatment-emergent adverse events such as bruising, redness, swelling, pain, tenderness, itching, and skin exfoliation. Of those who were treated, 40% experienced adverse outcomes that they felt affected their daily activity or were disabling, and 15% experienced adverse events (typically swelling and tenderness) that lasted more than 15 days.
Among the concerns expressed by the panel was the lack of men and people with dark skin in the study.
The Skinny Vodcast: Episode 2
Skin & Allergy News editor Amy Pfeiffer and reporter Naseem Miller review hot news in dermatology with the experts. And, guest dermatologist Lily Talakoub tackles the sea of cleansers.
Skin & Allergy News editor Amy Pfeiffer and reporter Naseem Miller review hot news in dermatology with the experts. And, guest dermatologist Lily Talakoub tackles the sea of cleansers.
Skin & Allergy News editor Amy Pfeiffer and reporter Naseem Miller review hot news in dermatology with the experts. And, guest dermatologist Lily Talakoub tackles the sea of cleansers.
Fillers Help Rejuvenate Aging Hands
When patients ask about fillers for their face, it presents the opportunity to also talk to them about their aging hands, according to Dr. Dee Anna Glaser, a professor and vice-chairman of the department of dermatology at St. Louis University School of Medicine.
Prepared fillers such as hyaluronic acid can be used to rejuvenate aging hands, she said. The amount used is about two to three syringes for both hands, recovery is quick, and the results can last up a year.
The procedure is also relatively safe, and aside from the initial bruising and lumpiness, the odds of infection are low, said Dr. Glaser. However, she did not advise using permanent fillers.
When patients ask about fillers for their face, it presents the opportunity to also talk to them about their aging hands, according to Dr. Dee Anna Glaser, a professor and vice-chairman of the department of dermatology at St. Louis University School of Medicine.
Prepared fillers such as hyaluronic acid can be used to rejuvenate aging hands, she said. The amount used is about two to three syringes for both hands, recovery is quick, and the results can last up a year.
The procedure is also relatively safe, and aside from the initial bruising and lumpiness, the odds of infection are low, said Dr. Glaser. However, she did not advise using permanent fillers.
When patients ask about fillers for their face, it presents the opportunity to also talk to them about their aging hands, according to Dr. Dee Anna Glaser, a professor and vice-chairman of the department of dermatology at St. Louis University School of Medicine.
Prepared fillers such as hyaluronic acid can be used to rejuvenate aging hands, she said. The amount used is about two to three syringes for both hands, recovery is quick, and the results can last up a year.
The procedure is also relatively safe, and aside from the initial bruising and lumpiness, the odds of infection are low, said Dr. Glaser. However, she did not advise using permanent fillers.