User login
MDedge latest news is breaking news from medical conferences, journals, guidelines, the FDA and CDC.
Total Intravenous Anesthesia Enables Earlier Facial Nerve Monitoring Than Sevoflurane in Ear Surgery
TOPLINE:
Total intravenous anesthesia (TIVA) enables earlier intraoperative monitoring of facial nerve activity than sevoflurane anesthesia during ear surgery, with reduced patient-ventilator dyssynchrony and fewer requirements for postoperative antiemetics.
METHODOLOGY:
- Researchers evaluated the difference in the timeliness of intraoperative monitoring of facial nerve activity during ear surgery with TIVA vs sevoflurane anesthesia.
- They included 98 patients aged 18-74 years undergoing ear surgery between November 2021 and November 2022; patients were randomly assigned to receive either TIVA or sevoflurane during the procedure. Of these, 92 were included in the final analysis.
- Neuromuscular function was monitored quantitatively throughout anesthesia with train-of-four counts and train-of-four ratios.
- The time from the administration of rocuronium to the start of facial nerve monitoring was recorded.
- The primary outcome measure focused on the recovery index, defined as the time interval between a train-of-four ratio of 0.25 and 0.75; the key secondary outcome was the time to reach a train-of-four ratio of 0.25 from rocuronium administration.
TAKEAWAY:
- The time to reach a train-of-four ratio of 0.25 was achieved earlier with TIVA than with sevoflurane (34 minutes vs 51 minutes; P < .001).
- Patient-ventilator dyssynchrony occurred less frequently in the TIVA group than in the sevoflurane group (15% vs 39%; P = .01).
- Postoperative requests for antiemetics were less frequent in the TIVA group than in the sevoflurane group (2% vs 17%; P = .03).
IN PRACTICE:
“We suggest that TIVA may be a better choice than sevoflurane anesthesia to meet an earlier request” for intraoperative facial nerve monitoring by surgeons, the study authors wrote.
SOURCE:
The study was led by Yu Jeong Bang, MD, of the Department of Anesthesiology and Pain Medicine at Sungkyunkwan University School of Medicine, in Seoul, Republic of Korea. It was published online on November 27, 2024, in The Canadian Journal of Anesthesia.
LIMITATIONS:
A careful interpretation of results may be necessary when clinicians use balanced anesthesia, such as sevoflurane with adjuvants like opioids or nonopioids. The feasibility of intraoperative facial nerve monitoring was decided by the surgeon during surgery, and the lowest stimulation intensity threshold for electromyography amplitude was not detected, as it was not the focus of this study. Although patients requiring intraoperative facial nerve monitoring during ear surgery were enrolled, some did not undergo the procedure based on the surgeon’s judgment.
DISCLOSURES:
This study did not receive any funding. The authors disclosed no relevant conflicts of interest.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Total intravenous anesthesia (TIVA) enables earlier intraoperative monitoring of facial nerve activity than sevoflurane anesthesia during ear surgery, with reduced patient-ventilator dyssynchrony and fewer requirements for postoperative antiemetics.
METHODOLOGY:
- Researchers evaluated the difference in the timeliness of intraoperative monitoring of facial nerve activity during ear surgery with TIVA vs sevoflurane anesthesia.
- They included 98 patients aged 18-74 years undergoing ear surgery between November 2021 and November 2022; patients were randomly assigned to receive either TIVA or sevoflurane during the procedure. Of these, 92 were included in the final analysis.
- Neuromuscular function was monitored quantitatively throughout anesthesia with train-of-four counts and train-of-four ratios.
- The time from the administration of rocuronium to the start of facial nerve monitoring was recorded.
- The primary outcome measure focused on the recovery index, defined as the time interval between a train-of-four ratio of 0.25 and 0.75; the key secondary outcome was the time to reach a train-of-four ratio of 0.25 from rocuronium administration.
TAKEAWAY:
- The time to reach a train-of-four ratio of 0.25 was achieved earlier with TIVA than with sevoflurane (34 minutes vs 51 minutes; P < .001).
- Patient-ventilator dyssynchrony occurred less frequently in the TIVA group than in the sevoflurane group (15% vs 39%; P = .01).
- Postoperative requests for antiemetics were less frequent in the TIVA group than in the sevoflurane group (2% vs 17%; P = .03).
IN PRACTICE:
“We suggest that TIVA may be a better choice than sevoflurane anesthesia to meet an earlier request” for intraoperative facial nerve monitoring by surgeons, the study authors wrote.
SOURCE:
The study was led by Yu Jeong Bang, MD, of the Department of Anesthesiology and Pain Medicine at Sungkyunkwan University School of Medicine, in Seoul, Republic of Korea. It was published online on November 27, 2024, in The Canadian Journal of Anesthesia.
LIMITATIONS:
A careful interpretation of results may be necessary when clinicians use balanced anesthesia, such as sevoflurane with adjuvants like opioids or nonopioids. The feasibility of intraoperative facial nerve monitoring was decided by the surgeon during surgery, and the lowest stimulation intensity threshold for electromyography amplitude was not detected, as it was not the focus of this study. Although patients requiring intraoperative facial nerve monitoring during ear surgery were enrolled, some did not undergo the procedure based on the surgeon’s judgment.
DISCLOSURES:
This study did not receive any funding. The authors disclosed no relevant conflicts of interest.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Total intravenous anesthesia (TIVA) enables earlier intraoperative monitoring of facial nerve activity than sevoflurane anesthesia during ear surgery, with reduced patient-ventilator dyssynchrony and fewer requirements for postoperative antiemetics.
METHODOLOGY:
- Researchers evaluated the difference in the timeliness of intraoperative monitoring of facial nerve activity during ear surgery with TIVA vs sevoflurane anesthesia.
- They included 98 patients aged 18-74 years undergoing ear surgery between November 2021 and November 2022; patients were randomly assigned to receive either TIVA or sevoflurane during the procedure. Of these, 92 were included in the final analysis.
- Neuromuscular function was monitored quantitatively throughout anesthesia with train-of-four counts and train-of-four ratios.
- The time from the administration of rocuronium to the start of facial nerve monitoring was recorded.
- The primary outcome measure focused on the recovery index, defined as the time interval between a train-of-four ratio of 0.25 and 0.75; the key secondary outcome was the time to reach a train-of-four ratio of 0.25 from rocuronium administration.
TAKEAWAY:
- The time to reach a train-of-four ratio of 0.25 was achieved earlier with TIVA than with sevoflurane (34 minutes vs 51 minutes; P < .001).
- Patient-ventilator dyssynchrony occurred less frequently in the TIVA group than in the sevoflurane group (15% vs 39%; P = .01).
- Postoperative requests for antiemetics were less frequent in the TIVA group than in the sevoflurane group (2% vs 17%; P = .03).
IN PRACTICE:
“We suggest that TIVA may be a better choice than sevoflurane anesthesia to meet an earlier request” for intraoperative facial nerve monitoring by surgeons, the study authors wrote.
SOURCE:
The study was led by Yu Jeong Bang, MD, of the Department of Anesthesiology and Pain Medicine at Sungkyunkwan University School of Medicine, in Seoul, Republic of Korea. It was published online on November 27, 2024, in The Canadian Journal of Anesthesia.
LIMITATIONS:
A careful interpretation of results may be necessary when clinicians use balanced anesthesia, such as sevoflurane with adjuvants like opioids or nonopioids. The feasibility of intraoperative facial nerve monitoring was decided by the surgeon during surgery, and the lowest stimulation intensity threshold for electromyography amplitude was not detected, as it was not the focus of this study. Although patients requiring intraoperative facial nerve monitoring during ear surgery were enrolled, some did not undergo the procedure based on the surgeon’s judgment.
DISCLOSURES:
This study did not receive any funding. The authors disclosed no relevant conflicts of interest.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Should Patients With Thyroid Issues Take GLP-1s? It Depends
Clinicians frequently reach out to Catherine Varney, DO, asking if it’s safe to prescribe glucagon-like peptide 1 receptor agonists (GLP-1 RAs) to patients with thyroid disorders, such as hypothyroidism, goiter, hyperthyroidism, and nodules.
The concerns often stem from prior animal studies that revealed a link between the medications and abnormal alterations in thyroid C cells.
“What physicians need to understand is that the increased risk of thyroid cancers is specifically in those with a personal or family history of multiple endocrine neoplasia 2 (MEN2) syndrome or medullary thyroid carcinoma [MTC],” said Varney, an assistant professor of family medicine at the University of Virginia (UVA) and obesity medicine director for UVA Health, both in Charlottesville.
MTC is a common manifestation of the MEN2 syndrome, a genetic disorder that affects the endocrine glands and can cause tumors in the thyroid gland. However, MTC is very rare, making up only 3%-5% of all thyroid cancers, Varney said.
More common types of thyroid cancers, such as papillary thyroid cancer, are not contraindications to GLP-1 RAs, said Laura Davisson, MD, MPH, a professor of medicine and director of Medical Weight Management at West Virginia University, Morgantown. Papillary thyroid cancer makes up about 80% of all thyroid cancers.
If physicians are thinking about prescribing GLP-1 RAs, it’s important to learn the specific types of thyroid disorders in a patient or in their family history, Davisson said. A history of thyroid conditions, such as Graves disease, Hashimoto thyroiditis, or disorders requiring a patient to take thyroid medicine, such as levothyroxine, does not preclude a person from taking GLP-1 RAs.
If a patient does not know their family history, then a discussion about whether GLP-1 RAs are right for them should take place, Davisson said.
“They should take into consideration the risks and benefits of the medicine, and the fact that medullary thyroid cancer and MEN2 syndrome are rare, making the chances of them running in their family low,” she said. “If the patient understands the risks, they may decide the potential benefits may outweigh the risks.”
Keep in mind that the elevated risk for medullary thyroid carcinoma has not been clearly demonstrated in humans during clinical trials, only in rodents, Varney added. Rodents are more likely to develop thyroid cancer after exposure to a GLP-1 RA because rodents have higher numbers of GLP-1 receptors in their thyroid cells.
The risk for thyroid cancer is also dose dependent, and the amount of GLP-1 RA that induced thyroid tumors in the rodents was 8-60 times higher than that given to humans, Varney said.
“Nonetheless, there have been case reports of MTC in humans after taking GLP-1 RA,” she said.
What About GLP-1 RAs and Pancreatitis?
While some studies show that GLP-1 RA analogs can increase the risk for pancreatitis, it’s important to know the reason behind pancreatitis before making a prescription decision, said Anila Chadha, MD, a family physician and obesity medicine physician at Dignity Health in Bakersfield, California.
If the pancreatitis is caused by alcohol or is medication induced, GLP-1 RAs are not recommended, Chadha said. If gallbladder stones are behind the pancreatitis, that’s different.
“If a patient has a well-documented history of gallstone-induced pancreatitis and the patient has had a cholecystectomy, it would be OK to prescribe GLP-1 [medications] after shared decision-making with the patient,” she said.
Some patients worry about taking GLP-1 RAs if they have other types of pancreatic disease beyond pancreatitis, said Davisson, but guidelines do not identify conditions such as pancreatic cysts or a family history of pancreatic cancer as contraindications to GLP-1 RAs.
However, be mindful that GLP-1 RA medications can increase gallbladder disease, Chadha said. A 2022 analysis in JAMA Internal Medicine, for example, found that the use of GLP-1 RAs was associated with an increased risk for gallbladder or biliary diseases, especially when used at higher doses, for longer durations, and for weight loss.
If a history of gallstones is present, physicians should tell patients that GLP-1 RA analogs can increase biliary colic, chances of cholecystitis, or even cholecystitis, Chadha said.
Gallbladder disease itself is not a contraindication, she said, but these patients can experience more disease complications when taking GLP-1 RAs.
Varney recently treated a middle-aged female patient who experienced biliary colic after taking a GLP-1 RA medication. The patient had a starting body mass index (BMI) of about 45 and lost 24% of her starting weight over 12 months using a combination of strengthening and cardiovascular training; a reduced-calorie, high-protein diet; and semaglutide for weight loss, Varney said.
The patient began developing upper gastrointestinal symptoms, including mild biliary colic, without concerning lab findings. An ultrasound revealed that she had new gallstones, compared with an ultrasound a few years prior. Varney started the patient on ursodeoxycholic acid, and over the course of 6 weeks, the patient’s symptoms gradually improved and eventually resolved.
“In a situation like this, many physicians might have discontinued the obesity medication, potentially deciding not to restart it due to the symptoms she was experiencing,” Varney said. “However, I believe it’s important to recognize that these medications offer far more than just weight loss benefits. The positive effects extend to cardiovascular, metabolic, and mental health, which are often overlooked when the focus is solely on the number on the scale or BMI.”
Other medical conditions that GLP-1 RAs may exacerbate include gastroparesis and diabetic retinopathy, Chadha noted. GLP-1 RA drugs can worsen these conditions and are not recommended for patients with such a history.
Nor are GLP-1 RA medications a good idea for patients struggling with severe constipation.
“It is very difficult to tolerate these medications because it can make constipation worse,” Chadha said. “In addition, if a patient struggles with severe heartburn, it can make the tolerability of these medications difficult.”
What Patients Should Know Before Using GLP-1 RAs
Not every overweight patient is a good candidate for GLP-1 RAs, and it’s important that doctors explain why the medications may not be right for them, Davisson said.
“If a patient only has a few pounds to lose (15-20 lb) and does not accept that these medications are intended to be a long-term treatment for the chronic diseases of diabetes or obesity, then they might not be a good candidate for the medication,” Davisson said.
Also essential is making sure patients are willing to follow appropriate healthy eating guidelines when on the medications, including getting enough protein and produce to maintain lean body mass and the necessary fiber to keep bowel habits regular.
Varney often hears concerns from physicians about the long-term effects of GLP-1 RAs. She said this class of medications has been used for nearly 20 years to treat type 2 diabetes, and the data over the past two decades have shown that the benefits of GLP-1 RAs far outweigh the risks for most patients.
Recent studies have also highlighted significant long-term benefits, including a marked reduction in cardiovascular events, heart failure, and kidney disease, she said.
“These outcomes underscore the effectiveness and safety of GLP-1 RAs not just for obesity treatment but for improving overall health, particularly in high-risk populations,” she said. “As physicians, it’s crucial to consider this extensive body of evidence when evaluating the use of these medications for patients, especially when the potential health benefits extend well beyond weight loss alone.”
A version of this article first appeared on Medscape.com.
Clinicians frequently reach out to Catherine Varney, DO, asking if it’s safe to prescribe glucagon-like peptide 1 receptor agonists (GLP-1 RAs) to patients with thyroid disorders, such as hypothyroidism, goiter, hyperthyroidism, and nodules.
The concerns often stem from prior animal studies that revealed a link between the medications and abnormal alterations in thyroid C cells.
“What physicians need to understand is that the increased risk of thyroid cancers is specifically in those with a personal or family history of multiple endocrine neoplasia 2 (MEN2) syndrome or medullary thyroid carcinoma [MTC],” said Varney, an assistant professor of family medicine at the University of Virginia (UVA) and obesity medicine director for UVA Health, both in Charlottesville.
MTC is a common manifestation of the MEN2 syndrome, a genetic disorder that affects the endocrine glands and can cause tumors in the thyroid gland. However, MTC is very rare, making up only 3%-5% of all thyroid cancers, Varney said.
More common types of thyroid cancers, such as papillary thyroid cancer, are not contraindications to GLP-1 RAs, said Laura Davisson, MD, MPH, a professor of medicine and director of Medical Weight Management at West Virginia University, Morgantown. Papillary thyroid cancer makes up about 80% of all thyroid cancers.
If physicians are thinking about prescribing GLP-1 RAs, it’s important to learn the specific types of thyroid disorders in a patient or in their family history, Davisson said. A history of thyroid conditions, such as Graves disease, Hashimoto thyroiditis, or disorders requiring a patient to take thyroid medicine, such as levothyroxine, does not preclude a person from taking GLP-1 RAs.
If a patient does not know their family history, then a discussion about whether GLP-1 RAs are right for them should take place, Davisson said.
“They should take into consideration the risks and benefits of the medicine, and the fact that medullary thyroid cancer and MEN2 syndrome are rare, making the chances of them running in their family low,” she said. “If the patient understands the risks, they may decide the potential benefits may outweigh the risks.”
Keep in mind that the elevated risk for medullary thyroid carcinoma has not been clearly demonstrated in humans during clinical trials, only in rodents, Varney added. Rodents are more likely to develop thyroid cancer after exposure to a GLP-1 RA because rodents have higher numbers of GLP-1 receptors in their thyroid cells.
The risk for thyroid cancer is also dose dependent, and the amount of GLP-1 RA that induced thyroid tumors in the rodents was 8-60 times higher than that given to humans, Varney said.
“Nonetheless, there have been case reports of MTC in humans after taking GLP-1 RA,” she said.
What About GLP-1 RAs and Pancreatitis?
While some studies show that GLP-1 RA analogs can increase the risk for pancreatitis, it’s important to know the reason behind pancreatitis before making a prescription decision, said Anila Chadha, MD, a family physician and obesity medicine physician at Dignity Health in Bakersfield, California.
If the pancreatitis is caused by alcohol or is medication induced, GLP-1 RAs are not recommended, Chadha said. If gallbladder stones are behind the pancreatitis, that’s different.
“If a patient has a well-documented history of gallstone-induced pancreatitis and the patient has had a cholecystectomy, it would be OK to prescribe GLP-1 [medications] after shared decision-making with the patient,” she said.
Some patients worry about taking GLP-1 RAs if they have other types of pancreatic disease beyond pancreatitis, said Davisson, but guidelines do not identify conditions such as pancreatic cysts or a family history of pancreatic cancer as contraindications to GLP-1 RAs.
However, be mindful that GLP-1 RA medications can increase gallbladder disease, Chadha said. A 2022 analysis in JAMA Internal Medicine, for example, found that the use of GLP-1 RAs was associated with an increased risk for gallbladder or biliary diseases, especially when used at higher doses, for longer durations, and for weight loss.
If a history of gallstones is present, physicians should tell patients that GLP-1 RA analogs can increase biliary colic, chances of cholecystitis, or even cholecystitis, Chadha said.
Gallbladder disease itself is not a contraindication, she said, but these patients can experience more disease complications when taking GLP-1 RAs.
Varney recently treated a middle-aged female patient who experienced biliary colic after taking a GLP-1 RA medication. The patient had a starting body mass index (BMI) of about 45 and lost 24% of her starting weight over 12 months using a combination of strengthening and cardiovascular training; a reduced-calorie, high-protein diet; and semaglutide for weight loss, Varney said.
The patient began developing upper gastrointestinal symptoms, including mild biliary colic, without concerning lab findings. An ultrasound revealed that she had new gallstones, compared with an ultrasound a few years prior. Varney started the patient on ursodeoxycholic acid, and over the course of 6 weeks, the patient’s symptoms gradually improved and eventually resolved.
“In a situation like this, many physicians might have discontinued the obesity medication, potentially deciding not to restart it due to the symptoms she was experiencing,” Varney said. “However, I believe it’s important to recognize that these medications offer far more than just weight loss benefits. The positive effects extend to cardiovascular, metabolic, and mental health, which are often overlooked when the focus is solely on the number on the scale or BMI.”
Other medical conditions that GLP-1 RAs may exacerbate include gastroparesis and diabetic retinopathy, Chadha noted. GLP-1 RA drugs can worsen these conditions and are not recommended for patients with such a history.
Nor are GLP-1 RA medications a good idea for patients struggling with severe constipation.
“It is very difficult to tolerate these medications because it can make constipation worse,” Chadha said. “In addition, if a patient struggles with severe heartburn, it can make the tolerability of these medications difficult.”
What Patients Should Know Before Using GLP-1 RAs
Not every overweight patient is a good candidate for GLP-1 RAs, and it’s important that doctors explain why the medications may not be right for them, Davisson said.
“If a patient only has a few pounds to lose (15-20 lb) and does not accept that these medications are intended to be a long-term treatment for the chronic diseases of diabetes or obesity, then they might not be a good candidate for the medication,” Davisson said.
Also essential is making sure patients are willing to follow appropriate healthy eating guidelines when on the medications, including getting enough protein and produce to maintain lean body mass and the necessary fiber to keep bowel habits regular.
Varney often hears concerns from physicians about the long-term effects of GLP-1 RAs. She said this class of medications has been used for nearly 20 years to treat type 2 diabetes, and the data over the past two decades have shown that the benefits of GLP-1 RAs far outweigh the risks for most patients.
Recent studies have also highlighted significant long-term benefits, including a marked reduction in cardiovascular events, heart failure, and kidney disease, she said.
“These outcomes underscore the effectiveness and safety of GLP-1 RAs not just for obesity treatment but for improving overall health, particularly in high-risk populations,” she said. “As physicians, it’s crucial to consider this extensive body of evidence when evaluating the use of these medications for patients, especially when the potential health benefits extend well beyond weight loss alone.”
A version of this article first appeared on Medscape.com.
Clinicians frequently reach out to Catherine Varney, DO, asking if it’s safe to prescribe glucagon-like peptide 1 receptor agonists (GLP-1 RAs) to patients with thyroid disorders, such as hypothyroidism, goiter, hyperthyroidism, and nodules.
The concerns often stem from prior animal studies that revealed a link between the medications and abnormal alterations in thyroid C cells.
“What physicians need to understand is that the increased risk of thyroid cancers is specifically in those with a personal or family history of multiple endocrine neoplasia 2 (MEN2) syndrome or medullary thyroid carcinoma [MTC],” said Varney, an assistant professor of family medicine at the University of Virginia (UVA) and obesity medicine director for UVA Health, both in Charlottesville.
MTC is a common manifestation of the MEN2 syndrome, a genetic disorder that affects the endocrine glands and can cause tumors in the thyroid gland. However, MTC is very rare, making up only 3%-5% of all thyroid cancers, Varney said.
More common types of thyroid cancers, such as papillary thyroid cancer, are not contraindications to GLP-1 RAs, said Laura Davisson, MD, MPH, a professor of medicine and director of Medical Weight Management at West Virginia University, Morgantown. Papillary thyroid cancer makes up about 80% of all thyroid cancers.
If physicians are thinking about prescribing GLP-1 RAs, it’s important to learn the specific types of thyroid disorders in a patient or in their family history, Davisson said. A history of thyroid conditions, such as Graves disease, Hashimoto thyroiditis, or disorders requiring a patient to take thyroid medicine, such as levothyroxine, does not preclude a person from taking GLP-1 RAs.
If a patient does not know their family history, then a discussion about whether GLP-1 RAs are right for them should take place, Davisson said.
“They should take into consideration the risks and benefits of the medicine, and the fact that medullary thyroid cancer and MEN2 syndrome are rare, making the chances of them running in their family low,” she said. “If the patient understands the risks, they may decide the potential benefits may outweigh the risks.”
Keep in mind that the elevated risk for medullary thyroid carcinoma has not been clearly demonstrated in humans during clinical trials, only in rodents, Varney added. Rodents are more likely to develop thyroid cancer after exposure to a GLP-1 RA because rodents have higher numbers of GLP-1 receptors in their thyroid cells.
The risk for thyroid cancer is also dose dependent, and the amount of GLP-1 RA that induced thyroid tumors in the rodents was 8-60 times higher than that given to humans, Varney said.
“Nonetheless, there have been case reports of MTC in humans after taking GLP-1 RA,” she said.
What About GLP-1 RAs and Pancreatitis?
While some studies show that GLP-1 RA analogs can increase the risk for pancreatitis, it’s important to know the reason behind pancreatitis before making a prescription decision, said Anila Chadha, MD, a family physician and obesity medicine physician at Dignity Health in Bakersfield, California.
If the pancreatitis is caused by alcohol or is medication induced, GLP-1 RAs are not recommended, Chadha said. If gallbladder stones are behind the pancreatitis, that’s different.
“If a patient has a well-documented history of gallstone-induced pancreatitis and the patient has had a cholecystectomy, it would be OK to prescribe GLP-1 [medications] after shared decision-making with the patient,” she said.
Some patients worry about taking GLP-1 RAs if they have other types of pancreatic disease beyond pancreatitis, said Davisson, but guidelines do not identify conditions such as pancreatic cysts or a family history of pancreatic cancer as contraindications to GLP-1 RAs.
However, be mindful that GLP-1 RA medications can increase gallbladder disease, Chadha said. A 2022 analysis in JAMA Internal Medicine, for example, found that the use of GLP-1 RAs was associated with an increased risk for gallbladder or biliary diseases, especially when used at higher doses, for longer durations, and for weight loss.
If a history of gallstones is present, physicians should tell patients that GLP-1 RA analogs can increase biliary colic, chances of cholecystitis, or even cholecystitis, Chadha said.
Gallbladder disease itself is not a contraindication, she said, but these patients can experience more disease complications when taking GLP-1 RAs.
Varney recently treated a middle-aged female patient who experienced biliary colic after taking a GLP-1 RA medication. The patient had a starting body mass index (BMI) of about 45 and lost 24% of her starting weight over 12 months using a combination of strengthening and cardiovascular training; a reduced-calorie, high-protein diet; and semaglutide for weight loss, Varney said.
The patient began developing upper gastrointestinal symptoms, including mild biliary colic, without concerning lab findings. An ultrasound revealed that she had new gallstones, compared with an ultrasound a few years prior. Varney started the patient on ursodeoxycholic acid, and over the course of 6 weeks, the patient’s symptoms gradually improved and eventually resolved.
“In a situation like this, many physicians might have discontinued the obesity medication, potentially deciding not to restart it due to the symptoms she was experiencing,” Varney said. “However, I believe it’s important to recognize that these medications offer far more than just weight loss benefits. The positive effects extend to cardiovascular, metabolic, and mental health, which are often overlooked when the focus is solely on the number on the scale or BMI.”
Other medical conditions that GLP-1 RAs may exacerbate include gastroparesis and diabetic retinopathy, Chadha noted. GLP-1 RA drugs can worsen these conditions and are not recommended for patients with such a history.
Nor are GLP-1 RA medications a good idea for patients struggling with severe constipation.
“It is very difficult to tolerate these medications because it can make constipation worse,” Chadha said. “In addition, if a patient struggles with severe heartburn, it can make the tolerability of these medications difficult.”
What Patients Should Know Before Using GLP-1 RAs
Not every overweight patient is a good candidate for GLP-1 RAs, and it’s important that doctors explain why the medications may not be right for them, Davisson said.
“If a patient only has a few pounds to lose (15-20 lb) and does not accept that these medications are intended to be a long-term treatment for the chronic diseases of diabetes or obesity, then they might not be a good candidate for the medication,” Davisson said.
Also essential is making sure patients are willing to follow appropriate healthy eating guidelines when on the medications, including getting enough protein and produce to maintain lean body mass and the necessary fiber to keep bowel habits regular.
Varney often hears concerns from physicians about the long-term effects of GLP-1 RAs. She said this class of medications has been used for nearly 20 years to treat type 2 diabetes, and the data over the past two decades have shown that the benefits of GLP-1 RAs far outweigh the risks for most patients.
Recent studies have also highlighted significant long-term benefits, including a marked reduction in cardiovascular events, heart failure, and kidney disease, she said.
“These outcomes underscore the effectiveness and safety of GLP-1 RAs not just for obesity treatment but for improving overall health, particularly in high-risk populations,” she said. “As physicians, it’s crucial to consider this extensive body of evidence when evaluating the use of these medications for patients, especially when the potential health benefits extend well beyond weight loss alone.”
A version of this article first appeared on Medscape.com.
Popular Diabetes Drug May Raise Vascular Surgery Risk
TOPLINE:
Among older veterans with type 2 diabetes (T2D), the use of sodium-glucose cotransporter 2 (SGLT2) inhibitors as an add-on therapy is associated with a higher risk for peripheral artery disease (PAD)-related surgical events than the use of dipeptidyl peptidase 4 (DPP-4) inhibitors.
METHODOLOGY:
- Some placebo-controlled randomized trials have reported an increased risk for amputation with the use of SGLT2 inhibitors in patients with underlying cardiovascular diseases; however, the evidence remains unconfirmed by other subsequent trials.
- Researchers conducted a retrospective study of US veterans with T2D initiating SGLT2 inhibitors or DPP-4 inhibitors (a reference drug) as an add-on to metformin, sulfonylurea, or insulin treatment alone or in combination.
- The primary outcome was the time to the first surgical event for PAD (amputation, peripheral revascularization and bypass, or peripheral vascular stent).
- A Cox proportional hazards model was used to compare PAD event risk between the SGLT2 inhibitor and DPP-4 inhibitor groups, allowing events up to 90 days or 360 days after stopping SGLT2 inhibitors.
TAKEAWAY:
- After propensity score weighting, 76,072 episodes of SGLT2 inhibitor use (94% empagliflozin, 4% canagliflozin, and 2% dapagliflozin) and 75,833 episodes of DPP-4 inhibitor use (45% saxagliptin, 34% alogliptin, 15% sitagliptin, and 6% linagliptin) were included.
- Participants had a median age of 69 years and a median duration of diabetes of 10.1 years.
- SGLT2 inhibitor users had higher PAD-related surgical events than DPP-4 inhibitor users (874 vs 780), with event rates of 11.2 vs 10.0 per 1000 person-years (adjusted hazard ratio [aHR], 1.18).
- The results remained consistent after 90 and 360 days of stopping SGLT2 inhibitors.
- SGLT2 inhibitor use was also associated with a higher risk for amputation (aHR, 1.15) and revascularization (aHR, 1.25) events than DPP-4 inhibitor use.
IN PRACTICE:
“These results underscore the need to determine the safety of [SGLT2 inhibitor] use among patients with diabetes who remain at very high risk for PAD,” the authors wrote.
SOURCE:
This study was led by Katherine E. Griffin, Geriatric Research Education and Clinical Center, Tennessee Valley Healthcare System, Nashville, and published online in Diabetes Care.
LIMITATIONS:
The study excluded patients whose initial diabetes treatment was not metformin, insulin, or sulfonylurea, which might have influenced the interpretation of the results. The median follow-up period of approximately 0.7 years for both groups may have affected the number of amputations and revascularization events observed. The study population primarily comprised White men, limiting generalizability to women and other demographic groups.
DISCLOSURES:
The study received funding through an investigator-initiated grant from the Veterans Affairs Clinical Science Research and Development. Two authors received partial research support through a grant from the Center for Diabetes Translation Research. All authors received partial support from the VETWISE-LHS Center of Innovation. No potential conflicts of interest relevant to the article were reported.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Among older veterans with type 2 diabetes (T2D), the use of sodium-glucose cotransporter 2 (SGLT2) inhibitors as an add-on therapy is associated with a higher risk for peripheral artery disease (PAD)-related surgical events than the use of dipeptidyl peptidase 4 (DPP-4) inhibitors.
METHODOLOGY:
- Some placebo-controlled randomized trials have reported an increased risk for amputation with the use of SGLT2 inhibitors in patients with underlying cardiovascular diseases; however, the evidence remains unconfirmed by other subsequent trials.
- Researchers conducted a retrospective study of US veterans with T2D initiating SGLT2 inhibitors or DPP-4 inhibitors (a reference drug) as an add-on to metformin, sulfonylurea, or insulin treatment alone or in combination.
- The primary outcome was the time to the first surgical event for PAD (amputation, peripheral revascularization and bypass, or peripheral vascular stent).
- A Cox proportional hazards model was used to compare PAD event risk between the SGLT2 inhibitor and DPP-4 inhibitor groups, allowing events up to 90 days or 360 days after stopping SGLT2 inhibitors.
TAKEAWAY:
- After propensity score weighting, 76,072 episodes of SGLT2 inhibitor use (94% empagliflozin, 4% canagliflozin, and 2% dapagliflozin) and 75,833 episodes of DPP-4 inhibitor use (45% saxagliptin, 34% alogliptin, 15% sitagliptin, and 6% linagliptin) were included.
- Participants had a median age of 69 years and a median duration of diabetes of 10.1 years.
- SGLT2 inhibitor users had higher PAD-related surgical events than DPP-4 inhibitor users (874 vs 780), with event rates of 11.2 vs 10.0 per 1000 person-years (adjusted hazard ratio [aHR], 1.18).
- The results remained consistent after 90 and 360 days of stopping SGLT2 inhibitors.
- SGLT2 inhibitor use was also associated with a higher risk for amputation (aHR, 1.15) and revascularization (aHR, 1.25) events than DPP-4 inhibitor use.
IN PRACTICE:
“These results underscore the need to determine the safety of [SGLT2 inhibitor] use among patients with diabetes who remain at very high risk for PAD,” the authors wrote.
SOURCE:
This study was led by Katherine E. Griffin, Geriatric Research Education and Clinical Center, Tennessee Valley Healthcare System, Nashville, and published online in Diabetes Care.
LIMITATIONS:
The study excluded patients whose initial diabetes treatment was not metformin, insulin, or sulfonylurea, which might have influenced the interpretation of the results. The median follow-up period of approximately 0.7 years for both groups may have affected the number of amputations and revascularization events observed. The study population primarily comprised White men, limiting generalizability to women and other demographic groups.
DISCLOSURES:
The study received funding through an investigator-initiated grant from the Veterans Affairs Clinical Science Research and Development. Two authors received partial research support through a grant from the Center for Diabetes Translation Research. All authors received partial support from the VETWISE-LHS Center of Innovation. No potential conflicts of interest relevant to the article were reported.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Among older veterans with type 2 diabetes (T2D), the use of sodium-glucose cotransporter 2 (SGLT2) inhibitors as an add-on therapy is associated with a higher risk for peripheral artery disease (PAD)-related surgical events than the use of dipeptidyl peptidase 4 (DPP-4) inhibitors.
METHODOLOGY:
- Some placebo-controlled randomized trials have reported an increased risk for amputation with the use of SGLT2 inhibitors in patients with underlying cardiovascular diseases; however, the evidence remains unconfirmed by other subsequent trials.
- Researchers conducted a retrospective study of US veterans with T2D initiating SGLT2 inhibitors or DPP-4 inhibitors (a reference drug) as an add-on to metformin, sulfonylurea, or insulin treatment alone or in combination.
- The primary outcome was the time to the first surgical event for PAD (amputation, peripheral revascularization and bypass, or peripheral vascular stent).
- A Cox proportional hazards model was used to compare PAD event risk between the SGLT2 inhibitor and DPP-4 inhibitor groups, allowing events up to 90 days or 360 days after stopping SGLT2 inhibitors.
TAKEAWAY:
- After propensity score weighting, 76,072 episodes of SGLT2 inhibitor use (94% empagliflozin, 4% canagliflozin, and 2% dapagliflozin) and 75,833 episodes of DPP-4 inhibitor use (45% saxagliptin, 34% alogliptin, 15% sitagliptin, and 6% linagliptin) were included.
- Participants had a median age of 69 years and a median duration of diabetes of 10.1 years.
- SGLT2 inhibitor users had higher PAD-related surgical events than DPP-4 inhibitor users (874 vs 780), with event rates of 11.2 vs 10.0 per 1000 person-years (adjusted hazard ratio [aHR], 1.18).
- The results remained consistent after 90 and 360 days of stopping SGLT2 inhibitors.
- SGLT2 inhibitor use was also associated with a higher risk for amputation (aHR, 1.15) and revascularization (aHR, 1.25) events than DPP-4 inhibitor use.
IN PRACTICE:
“These results underscore the need to determine the safety of [SGLT2 inhibitor] use among patients with diabetes who remain at very high risk for PAD,” the authors wrote.
SOURCE:
This study was led by Katherine E. Griffin, Geriatric Research Education and Clinical Center, Tennessee Valley Healthcare System, Nashville, and published online in Diabetes Care.
LIMITATIONS:
The study excluded patients whose initial diabetes treatment was not metformin, insulin, or sulfonylurea, which might have influenced the interpretation of the results. The median follow-up period of approximately 0.7 years for both groups may have affected the number of amputations and revascularization events observed. The study population primarily comprised White men, limiting generalizability to women and other demographic groups.
DISCLOSURES:
The study received funding through an investigator-initiated grant from the Veterans Affairs Clinical Science Research and Development. Two authors received partial research support through a grant from the Center for Diabetes Translation Research. All authors received partial support from the VETWISE-LHS Center of Innovation. No potential conflicts of interest relevant to the article were reported.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Impact of NSAID Use on Bleeding Rates for Patients Taking Rivaroxaban or Apixaban
Impact of NSAID Use on Bleeding Rates for Patients Taking Rivaroxaban or Apixaban
Clinical practice has shifted from vitamin K antagonists to direct oral anticoagulants (DOACs) for atrial fibrillation treatment due to their more favorable risk-benefit profile and less lifestyle modification required.1,2 However, the advantage of a lower bleeding risk with DOACs could be compromised by potentially problematic pharmacokinetic interactions like those conferred by antiplatelets or nonsteroidal anti-inflammatory drugs (NSAIDs).3,4 Treating a patient needing anticoagulation with a DOAC who has comorbidities may introduce unavoidable drug-drug interactions. This particularly happens with over-the-counter and prescription NSAIDs used for the management of pain and inflammatory conditions.5
NSAIDs primarily affect 2 cyclooxygenase (COX) enzyme isomers, COX-1 and COX-2.6 COX-1 helps maintain gastrointestinal (GI) mucosa integrity and platelet aggregation processes, whereas COX-2 is engaged in pain signaling and inflammation mediation. COX-1 inhibition is associated with more bleeding-related adverse events (AEs), especially in the GI tract. COX-2 inhibition is thought to provide analgesia and anti-inflammatory properties without elevating bleeding risk. This premise is responsible for the preferential use of celecoxib, a COX-2 selective NSAID, which should confer a lower bleeding risk compared to nonselective NSAIDs such as ibuprofen and naproxen.7 NSAIDs have been documented as independent risk factors for bleeding. NSAID users are about 3 times as likely to develop GI AEs compared to nonNSAID users.8
Many clinicians aim to further mitigate NSAID-associated bleeding risk by coprescribing a proton pump inhibitor (PPI). PPIs provide gastroprotection against NSAID-induced mucosal injury and sequential complication of GI bleeding. In a multicenter randomized control trial, patients who received concomitant PPI therapy while undergoing chronic NSAID therapy—including nonselective and COX-2 selective NSAIDs—had a significantly lower risk of GI ulcer development (placebo, 17.0%; 20 mg esomeprazole, 5.2%; 40 mg esomeprazole, 4.6%).9 Current clinical guidelines for preventing NSAIDassociated bleeding complications recommend using a COX-2 selective NSAID in combination with PPI therapy for patients at high risk for GI-related bleeding, including the concomitant use of anticoagulants.10
There is evidence suggesting an increased bleeding risk with NSAIDs when used in combination with vitamin K antagonists such as warfarin.11,12 A systematic review of warfarin and concomitant NSAID use found an increased risk of overall bleeding with NSAID use in combination with warfarin (odds ratio 1.58; 95% CI, 1.18-2.12), compared to warfarin alone.12
Posthoc analyses of randomized clinical trials have also demonstrated an increased bleeding risk with oral anticoagulation and concomitant NSAID use.13,14 In the RE-LY trial, NSAID users on warfarin or dabigatran had a statistically significant increased risk of major bleeding compared to non-NSAID users (hazard ratio [HR] 1.68; 95% CI, 1.40- 2.02; P < .001).13 In the ARISTOTLE trial, patients on warfarin or apixaban who were incident NSAID users were found to have an increased risk of major bleeding (HR 1.61; 95% CI, 1.11-2.33) and clinically relevant nonmajor bleeding (HR 1.70; 95% CI, 1.16- 2.48).14 These trials found a statistically significant increased bleeding risk associated with NSAID use, though the populations evaluated included patients taking warfarin and patients taking DOACs. These trials did not evaluate the bleeding risk of concomitant NSAID use among DOACs alone.
Evidence on NSAID-associated bleeding risk with DOACs is lacking in settings where the patient population, prescribing practices, and monitoring levels are variable. Within the Veterans Health Administration, clinical pharmacist practitioners (CPPs) in anticoagulation clinics oversee DOAC therapy management. CPPs monitor safety and efficacy of DOAC therapies through a population health management tool, the DOAC Dashboard.15 The DOAC Dashboard creates alerts for patients who may require an intervention based on certain clinical parameters, such as drug-drug interactions.16 Whenever a patient on a DOAC is prescribed an NSAID, an alert is generated on the DOAC Dashboard to flag the CPPs for the potential need for an intervention. If NSAID therapy remains clinically indicated, CPPs may recommend risk reduction strategies such as a COX-2 selective NSAID or coprescribing a PPI.10
The DOAC Dashboard provides an ideal setting for investigating the effects of NSAID use, NSAID selectivity, and PPI coprescribing on DOAC bleeding rates. With an increasing population of patients receiving anticoagulation therapy with a DOAC, more guidance regarding the bleeding risk of concomitant NSAID use with DOACs is needed. Studies evaluating the bleeding risk with concomitant NSAID use in patients on a DOAC alone are limited. This is the first study to date to compare bleeding risk with concomitant NSAID use between DOACs. This study provides information on bleeding risk with NSAID use among commonly prescribed DOACs, rivaroxaban and apixaban, and the potential impacts of current risk reduction strategies.
METHODS
This single-center retrospective cohort review was performed using the electronic health records (EHRs) of patients enrolled in the US Department of Veterans Affairs (VA) Mountain Home Healthcare System who received rivaroxaban or apixaban from December 2020 to December 2022. This study received approval from the East Tennessee State University/VA Institutional Review Board committee.
Patients were identified through the DOAC Dashboard, aged 21 to 100 years, and received rivaroxaban or apixaban at a therapeutic dose: rivaroxaban 10 to 20 mg daily or apixaban 2.5 to 5 mg twice daily. Patients were excluded if they were prescribed dual antiplatelet therapy, received rivaroxaban at dosing indicated for peripheral vascular disease, were undergoing dialysis, had evidence of moderate to severe hepatic impairment or any hepatic disease with coagulopathy, were undergoing chemotherapy or radiation, or had hematological conditions with predisposed bleeding risk. These patients were excluded to mitigate the potential confounding impact from nontherapeutic DOAC dosing strategies and conditions associated with an increased bleeding risk.
Eligible patients were stratified based on NSAID use. NSAID users were defined as patients prescribed an oral NSAID, including both acute and chronic courses, at any point during the study time frame while actively on a DOAC. Bleeding events were reviewed to evaluate rates between rivaroxaban and apixaban among NSAID and nonNSAID users. Identified NSAID users were further assessed for NSAID selectivity and PPI coprescribing as a subgroup analysis for the secondary assessment.
Data Collection
Baseline data were collected, including age, body mass index, anticoagulation indication, DOAC agent, DOAC dose, and DOAC total daily dose. Baseline serum creatinine levels, liver function tests, hemoglobin levels, and platelet counts were collected from the most recent data available immediately prior to the bleeding event, if applicable.
The DOAC Dashboard was reviewed for active and dismissed drug interaction alerts to identify patients taking rivaroxaban or apixaban who were prescribed an NSAID. Patients were categorized in the NSAID group if an interacting drug alert with an NSAID was reported during the study time frame. Data available through the interacting drug alerts on NSAID use were limited to the interacting drug name and date of the reported flag. Manual EHR review was required to confirm dates of NSAID therapy initiation and NSAID discontinuation, if applicable.
Data regarding concomitant antiplatelet use were obtained through review of the active and dismissed drug interaction alerts on the DOAC Dashboard. Concomitant antiplatelet use was defined as the prescribing of a single antiplatelet agent at any point while receiving DOAC therapy. Data on concomitant antiplatelets were collected regardless of NSAID status.
Data on coprescribed PPI therapy were obtained through manual EHR review of identified NSAID users. Coprescribed PPI therapy was defined as the prescribing of a PPI at any point during NSAID therapy. Data regarding PPI use among non-NSAID users were not collected because the secondary endpoint was designed to assess PPI use only among patients coprescribed a DOAC and NSAID.
Outcomes
Bleeding events were identified through an outcomes report generated by the DOAC Dashboard based on International Classification of Diseases, Tenth Revision diagnosis codes associated with a bleeding event. The outcomes report captures diagnoses from the outpatient and inpatient care settings. Reported bleeding events were limited to patients who received a DOAC at any point in the 6 months prior to the event and excluded patients with recent DOAC initiation within 7 days of the event, as these patients are not captured on the DOAC Dashboard.
All reported bleeding events were manually reviewed in the EHR and categorized as a major or clinically relevant nonmajor bleed, according to International Society of Thrombosis and Haemostasis criteria. Validated bleeding events were then crossreferenced with the interacting drug alerts report to identify events with potentially overlapping NSAID therapy at the time of the event. Overlapping NSAID therapy was defined as the prescribing of an NSAID at any point in the 6 months prior to the event. All events with potential overlapping NSAID therapies were manually reviewed for confirmation of NSAID status at the time of the event.
The primary endpoint was a composite of any bleeding event per International Society of Thrombosis and Haemostasis criteria. The secondary endpoint evaluated the potential impact of NSAID selectivity or PPI coprescribing on the bleeding rate among the NSAID user groups.
Statistical Analysis
Analyses were performed consistent with the methods used in the ARISTOTLE and RE-LY trials. It was determined that a sample size of 504 patients, with ≥ 168 patients in each group, would provide 80% power using a 2-sided a of 0.05. HRs with 95% CIs and respective P values were calculated using a SPSS-adapted online calculator.
RESULTS
The DOAC Dashboard identified 681 patients on rivaroxaban and 3225 patients on apixaban; 72 patients on rivaroxaban (10.6%) and 300 patients on apixaban (9.3%) were NSAID users. The mean age of NSAID users was 66.9 years in the rivaroxaban group and 72.4 years in the apixaban group. The mean age of non-NSAID users was 71.5 years in the rivaroxaban group and 75.6 years in the apixaban group. No appreciable differences were observed among subgroups in body mass index, renal function, hepatic function, hemoglobin, or platelet counts, and no statistically significant differences were identified (Table 1). Antiplatelet agents identified included aspirin, clopidogrel, prasugrel, and ticagrelor. Fifteen patients (20.3%) in the rivaroxaban group and 87 patients (28.7%) in the apixaban group had concomitant antiplatelet and NSAID use. Forty-five patients on rivaroxaban (60.8%) and 170 (55.9%) on apixaban were prescribed concomitant PPI and NSAID at baseline. Among non-NSAID users, there was concomitant antiplatelet use for 265 patients (43.6%) in the rivaroxaban group and 1401 patients (47.9%) in the apixaban group. Concomitant PPI use was identified among 63 patients (60.0%) taking selective NSAIDs and 182 (57.2%) taking nonselective NSAIDs.
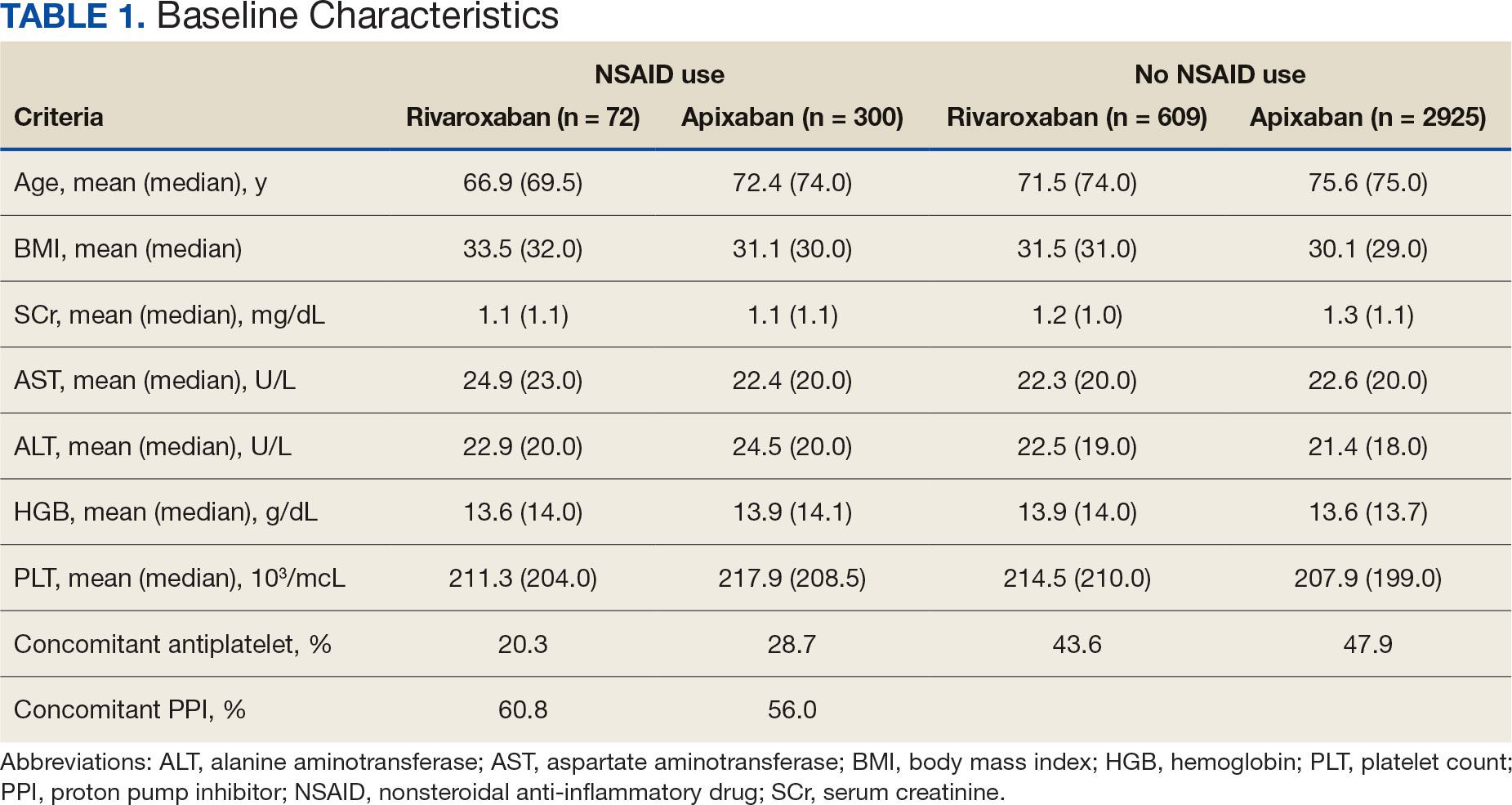
A total of 423 courses of NSAIDs were identified: 85 NSAID courses in the rivaroxaban group and 338 NSAID courses in the apixaban group. Most NSAID courses involved a nonselective NSAID in the rivaroxaban and apixaban NSAID user groups: 75.2% (n = 318) aggregately compared to 71.8% (n = 61) and 76.0% (n = 257) in the rivaroxaban and apixaban groups, respectively. The most frequent NSAID courses identified were meloxicam (26.7%; n = 113), celecoxib (24.8%; n = 105), ibuprofen (19.1%; n = 81), and naproxen (13.5%; n = 57). Data regarding NSAID therapy initiation and discontinuation dates were not readily available. As a result, the duration of NSAID courses was not captured.
There was no statistically significant difference in bleeding rates between rivaroxaban and apixaban among NSAID users (HR 1.04; 95% CI, 0.98-1.12) or non-NSAID users (HR 1.15; 95% CI, 0.80-1.66) (Table 2). Apixaban non-NSAID users had a higher rate of major bleeds (HR 0.32; 95% CI, 0.17-0.61) while rivaroxaban non-NSAID users had a higher rate of clinically relevant nonmajor bleeds (HR 1.63; 95% CI, 1.10-2.54).
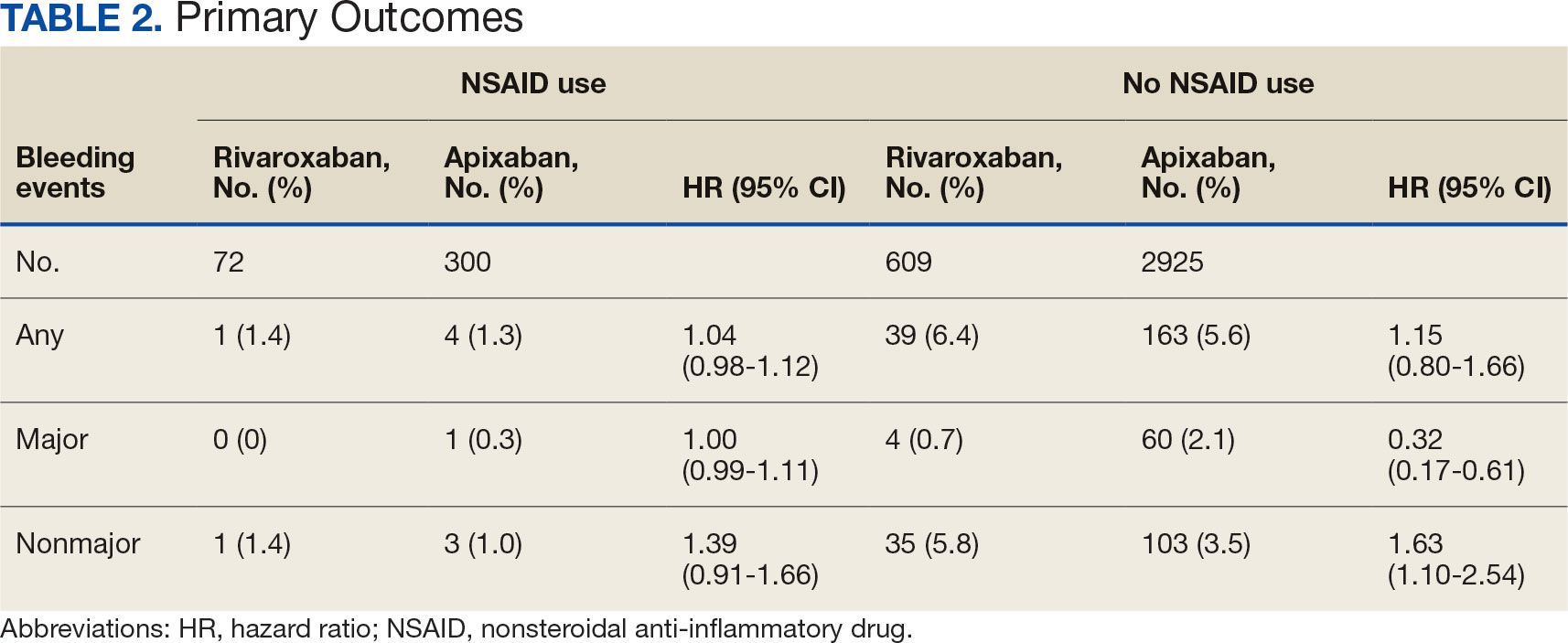
The sample size for the secondary endpoint consisted of bleeding events that were confirmed to have had an overlapping NSAID prescribed at the time of the event. For this secondary assessment, there was 1 rivaroxaban NSAID user bleeding event and 4 apixaban NSAID user bleeding events. For the rivaroxaban NSAID user bleeding event, the NSAID was nonselective and a PPI was not coprescribed. For the apixaban NSAID user bleeding events, 2 NSAIDs were nonselective and 2 were selective. All patients with apixaban and NSAID bleeding events had a coprescribed PPI. There was no clinically significant difference in the bleeding rates observed for NSAID selectivity or PPI coprescribing among the NSAID user subgroups.
DISCUSSION
This study found that there was no statistically significant difference for bleeding rates of major and nonmajor bleeding events between rivaroxaban and apixaban among NSAID users and non-NSAID users. This study did not identify a clinically significant impact on bleeding rates from NSAID selectivity or PPI coprescribing among the NSAID users.
There were notable but not statistically significant differences in baseline characteristics observed between the NSAID and non-NSAID user groups. On average, the rivaroxaban and apixaban NSAID users were younger compared with those not taking NSAIDs. NSAIDs, specifically nonselective NSAIDs, are recognized as potentially inappropriate medications for older adults given that this population is at an increased risk for GI ulcer development and/or GI bleeding.17 The non-NSAID user group likely consisted of older patients compared to the NSAID user group as clinicians may avoid prescribing NSAIDs to older adults regardless of concomitant DOAC therapy.
In addition to having an older patient population, non-NSAID users were more frequently prescribed a concomitant antiplatelet when compared with NSAID users. This prescribing pattern may be due to clinicians avoiding the use of NSAIDs in patients receiving DOAC therapy in combination with antiplatelet therapy, as these patients have been found to have an increased bleeding rate compared to DOAC therapy alone.18
Non-NSAID users had an overall higher bleeding rate for both major and nonmajor bleeding events. Based on this observation, it could be hypothesized that antiplatelet agents have a higher risk of bleeding in comparison to NSAIDs. In a subanalysis of the EXPAND study evaluating risk factors of major bleeding in patients receiving rivaroxaban, concomitant use of antiplatelet agents demonstrated a statistically significant increased risk of bleeding (HR 1.6; 95% CI, 1.2-2.3; P = .003) while concomitant use of NSAIDs did not (HR 0.8; 95% CI, 0.3-2.2; P = .67).19
In assessing PPI status at baseline, a majority of both rivaroxaban and apixaban NSAID users were coprescribed a PPI. This trend aligns with current clinical guideline recommendations for the prescribing of PPI therapy for GI protection in high-risk patients, such as those on DOAC therapy and concomitant NSAID therapy.10 Given the high proportion of NSAID users coprescribed a PPI at baseline, it may be possible that the true incidence of NSAID-associated bleeding events was higher than what this study found. This observation may reflect the impact from timely implementation of risk mitigation strategies by CPPs in the anticoagulation clinic. However, this study was not constructed to assess the efficacy of PPI use in this manner.
It is important to note the patients included in this study were followed by a pharmacist in an anticoagulation clinic using the DOAC Dashboard.15 This population management tool allows CPPs to make proactive interventions when a patient taking a DOAC receives an NSAID prescription, such as recommending the coprescribing of a PPI or use of a selective NSAID.10,16 These standards of care may have contributed to an overall reduced bleeding rate among the NSAID user group and may not be reflective of private practice.
The planned analysis of this study was modeled after the posthoc analysis of the RE-LY and ARISTOTLE trials. Both trials demonstrated an increased risk of bleeding with oral anticoagulation, including DOAC and warfarin, in combination with NSAID use. However, both trials found that NSAID use in patients treated with a DOAC was not independently associated with increased bleeding events compared with warfarin.13,14 The results of this study are comparable to the RE-LY and ARISTOTLE findings that NSAID use among patients treated with rivaroxaban or apixaban did not demonstrate a statistically significant increased bleeding risk.
Studies of NSAID use in combination with DOAC therapy have been limited to patient populations consisting of both DOAC and warfarin. Evidence from these trials outlines the increased bleeding risk associated with NSAID use in combination with oral anticoagulation; however, these patient populations include those on a DOAC and warfarin.13,14,19,20 Given the limited evidence on NSAID use among DOACs alone, it is assumed NSAID use in combination with DOACs has a similar risk of bleeding as warfarin use. This may cause clinicians to automatically exclude NSAID therapy as a treatment option for patients on a DOAC who are otherwise clinically appropriate candidates, such as those with underlying inflammatory conditions. Avoiding NSAID therapy in this patient population may lead to suboptimal pain management and increase the risk of patient harm from methods such as inappropriate opioid therapy prescribing.
DOAC therapy should not be a universal limitation to the use of NSAIDs. Although the risk of bleeding with NSAID therapy is always present, deliberate NSAID prescribing in addition to the timely implementation of risk mitigation strategies may provide an avenue for safe NSAID prescribing in patients receiving a DOAC. A population health-based approach to DOAC management, such as the DOAC Dashboard, appears to be effective at preventing patient harm when NSAIDs are prescribed in conjunction with DOACs.
Limitations
The DOAC Dashboard has been shown to be effective and efficient at monitoring DOAC therapy from a population-based approach.16 Reports generated through the DOAC Dashboard provide convenient access to patient data which allows for timely interventions; however, there are limits to its use for data collection. All the data elements necessary to properly assess bleeding risk with validated tools, such as HAS-BLED (hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, labile international normalized ratio, elderly, drugs/ alcohol concomitantly), are not available on DOAC Dashboard reports. Due to this constraint, bleeding risk assessments were not conducted at baseline and this study was unable to include risk modeling. Additionally, data elements like initiation and discontinuation dates and duration of therapies were not readily available. As a result, this study was unable to incorporate time as a data point.
This was a retrospective study that relied on manual review of chart documentation to verify bleeding events, but data obtained through the DOAC Dashboard were transferred directly from the EHR.15 Bleeding events available for evaluation were restricted to those that occurred at a VA facility. Additionally, the sample size within the rivaroxaban NSAID user group did not reach the predefined sample size required to reach power and may have been too small to detect a difference if one did exist. The secondary assessment had a low sample size of NSAID user bleeding events, making it difficult to fully assess its impact on NSAID selectivity and PPI coprescribing on bleeding rates. All courses of NSAIDs were equally valued regardless of the dose or therapy duration; however, this is consistent with how NSAID use was defined in the RE-LY and ARISTOTLE trials.
CONCLUSIONS
This retrospective cohort review found no statistically significant difference in the composite bleeding rates between rivaroxaban and apixaban among NSAID users and non-NSAID users. Moreover, there was no clinically significant impact observed for bleeding rates in regard to NSAID selectivity and PPI coprescribing among NSAID users. However, coprescribing of PPI therapy to patients on a DOAC who are clinically indicated for an NSAID may reduce the risk of bleeding. Population health management tools, such as the DOAC Dashboard, may also allow clinicians to safely prescribe NSAIDs to patients on a DOAC. Further large-scale observational studies are needed to quantify the real-world risk of bleeding with concomitant NSAID use among DOACs alone and to evaluate the impact from NSAID selectivity or PPI coprescribing.
- Ruff CT, Giugliano RP, Braunwald E, et al. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a meta-analysis of randomised trials. Lancet. 2014;383(9921):955-962. doi:10.1016/S0140-6736(13)62343-0
- Ageno W, Gallus AS, Wittkowsky A, Crowther M, Hylek EM, Palareti G. Oral anticoagulant therapy: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e44S-e88S. doi:10.1378/chest.11-2292
- Eikelboom J, Merli G. Bleeding with direct oral anticoagulants vs warfarin: clinical experience. Am J Med. 2016;129(11S):S33-S40. doi:10.1016/j.amjmed.2016.06.003
- Vranckx P, Valgimigli M, Heidbuchel H. The significance of drug-drug and drug-food interactions of oral anticoagulation. Arrhythm Electrophysiol Rev. 2018;7(1):55-61. doi:10.15420/aer.2017.50.1
- Davis JS, Lee HY, Kim J, et al. Use of non-steroidal antiinflammatory drugs in US adults: changes over time and by demographic. Open Heart. 2017;4(1):e000550. doi:10.1136/openhrt-2016-000550
- Schafer AI. Effects of nonsteroidal antiinflammatory drugs on platelet function and systemic hemostasis. J Clin Pharmacol. 1995;35(3):209-219. doi:10.1002/j.1552-4604.1995.tb04050.x
- Al-Saeed A. Gastrointestinal and cardiovascular risk of nonsteroidal anti-inflammatory drugs. Oman Med J. 2011;26(6):385-391. doi:10.5001/omj.2011.101
- Gabriel SE, Jaakkimainen L, Bombardier C. Risk for serious gastrointestinal complications related to use of nonsteroidal anti-inflammatory drugs. Ann Intern Med. 1991;115(10):787-796. doi:10.7326/0003-4819-115-10-787
- Scheiman JM, Yeomans ND, Talley NJ, et al. Prevention of ulcers by esomeprazole in at-risk patients using non-selective NSAIDs and COX-2 inhibitors. Am J Gastroenterol. 2006;101(4):701-710. doi:10.1111/j.1572-0241.2006.00499.x
- Freedberg DE, Kim LS, Yang YX. The risks and benefits of long-term use of proton pump inhibitors: expert review and best practice advice from the American Gastroenterological Association. Gastroenterology. 2017;152(4):706-715. doi:10.1053/j.gastro.2017.01.031
- Lamberts M, Lip GYH, Hansen ML, et al. Relation of nonsteroidal anti-inflammatory drugs to serious bleeding and thromboembolism risk in patients with atrial fibrillation receiving antithrombotic therapy: a nationwide cohort study. Ann Intern Med. 2014;161(10):690-698. doi:10.7326/M13-1581
- Villa Zapata L, Hansten PD, Panic J, et al. Risk of bleeding with exposure to warfarin and nonsteroidal anti-inflammatory drugs: a systematic review and metaanalysis. Thromb Haemost. 2020;120(7):1066-1074. doi:10.1055/s-0040-1710592
- Kent AP, Brueckmann M, Fraessdorf M, et al. Concomitant oral anticoagulant and nonsteroidal anti-inflammatory drug therapy in patients with atrial fibrillation. J Am Coll Cardiol. 2018;72(3):255-267. doi:10.1016/j.jacc.2018.04.063
- Dalgaard F, Mulder H, Wojdyla DM, et al. Patients with atrial fibrillation taking nonsteroidal antiinflammatory drugs and oral anticoagulants in the ARISTOTLE Trial. Circulation. 2020;141(1):10-20. doi:10.1161/CIRCULATIONAHA.119.041296
- Allen AL, Lucas J, Parra D, et al. Shifting the paradigm: a population health approach to the management of direct oral anticoagulants. J Am Heart Asssoc. 2021;10(24):e022758. doi:10.1161/JAHA.121.022758
- . Valencia D, Spoutz P, Stoppi J, et al. Impact of a direct oral anticoagulant population management tool on anticoagulation therapy monitoring in clinical practice. Ann Pharmacother. 2019;53(8):806-811. doi:10.1177/1060028019835843
- By the 2023 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2023 Updated AGS Beers Criteria® for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2023;71(7):2052-2081. doi:10.1111/jgs.18372
- Kumar S, Danik SB, Altman RK, et al. Non-vitamin K antagonist oral anticoagulants and antiplatelet therapy for stroke prevention in patients with atrial fibrillation. Cardiol Rev. 2016;24(5):218-223. doi:10.1097/CRD.0000000000000088
- Sakuma I, Uchiyama S, Atarashi H, et al. Clinical risk factors of stroke and major bleeding in patients with nonvalvular atrial fibrillation under rivaroxaban: the EXPAND study sub-analysis. Heart Vessels. 2019;34(11):1839-1851. doi:10.1007/s00380-019-01425-x
- Davidson BL, Verheijen S, Lensing AWA, et al. Bleeding risk of patients with acute venous thromboembolism taking nonsteroidal anti-inflammatory drugs or aspirin. JAMA Intern Med. 2014;174(6):947-953. doi:10.1001/jamainternmed.2014.946
Clinical practice has shifted from vitamin K antagonists to direct oral anticoagulants (DOACs) for atrial fibrillation treatment due to their more favorable risk-benefit profile and less lifestyle modification required.1,2 However, the advantage of a lower bleeding risk with DOACs could be compromised by potentially problematic pharmacokinetic interactions like those conferred by antiplatelets or nonsteroidal anti-inflammatory drugs (NSAIDs).3,4 Treating a patient needing anticoagulation with a DOAC who has comorbidities may introduce unavoidable drug-drug interactions. This particularly happens with over-the-counter and prescription NSAIDs used for the management of pain and inflammatory conditions.5
NSAIDs primarily affect 2 cyclooxygenase (COX) enzyme isomers, COX-1 and COX-2.6 COX-1 helps maintain gastrointestinal (GI) mucosa integrity and platelet aggregation processes, whereas COX-2 is engaged in pain signaling and inflammation mediation. COX-1 inhibition is associated with more bleeding-related adverse events (AEs), especially in the GI tract. COX-2 inhibition is thought to provide analgesia and anti-inflammatory properties without elevating bleeding risk. This premise is responsible for the preferential use of celecoxib, a COX-2 selective NSAID, which should confer a lower bleeding risk compared to nonselective NSAIDs such as ibuprofen and naproxen.7 NSAIDs have been documented as independent risk factors for bleeding. NSAID users are about 3 times as likely to develop GI AEs compared to nonNSAID users.8
Many clinicians aim to further mitigate NSAID-associated bleeding risk by coprescribing a proton pump inhibitor (PPI). PPIs provide gastroprotection against NSAID-induced mucosal injury and sequential complication of GI bleeding. In a multicenter randomized control trial, patients who received concomitant PPI therapy while undergoing chronic NSAID therapy—including nonselective and COX-2 selective NSAIDs—had a significantly lower risk of GI ulcer development (placebo, 17.0%; 20 mg esomeprazole, 5.2%; 40 mg esomeprazole, 4.6%).9 Current clinical guidelines for preventing NSAIDassociated bleeding complications recommend using a COX-2 selective NSAID in combination with PPI therapy for patients at high risk for GI-related bleeding, including the concomitant use of anticoagulants.10
There is evidence suggesting an increased bleeding risk with NSAIDs when used in combination with vitamin K antagonists such as warfarin.11,12 A systematic review of warfarin and concomitant NSAID use found an increased risk of overall bleeding with NSAID use in combination with warfarin (odds ratio 1.58; 95% CI, 1.18-2.12), compared to warfarin alone.12
Posthoc analyses of randomized clinical trials have also demonstrated an increased bleeding risk with oral anticoagulation and concomitant NSAID use.13,14 In the RE-LY trial, NSAID users on warfarin or dabigatran had a statistically significant increased risk of major bleeding compared to non-NSAID users (hazard ratio [HR] 1.68; 95% CI, 1.40- 2.02; P < .001).13 In the ARISTOTLE trial, patients on warfarin or apixaban who were incident NSAID users were found to have an increased risk of major bleeding (HR 1.61; 95% CI, 1.11-2.33) and clinically relevant nonmajor bleeding (HR 1.70; 95% CI, 1.16- 2.48).14 These trials found a statistically significant increased bleeding risk associated with NSAID use, though the populations evaluated included patients taking warfarin and patients taking DOACs. These trials did not evaluate the bleeding risk of concomitant NSAID use among DOACs alone.
Evidence on NSAID-associated bleeding risk with DOACs is lacking in settings where the patient population, prescribing practices, and monitoring levels are variable. Within the Veterans Health Administration, clinical pharmacist practitioners (CPPs) in anticoagulation clinics oversee DOAC therapy management. CPPs monitor safety and efficacy of DOAC therapies through a population health management tool, the DOAC Dashboard.15 The DOAC Dashboard creates alerts for patients who may require an intervention based on certain clinical parameters, such as drug-drug interactions.16 Whenever a patient on a DOAC is prescribed an NSAID, an alert is generated on the DOAC Dashboard to flag the CPPs for the potential need for an intervention. If NSAID therapy remains clinically indicated, CPPs may recommend risk reduction strategies such as a COX-2 selective NSAID or coprescribing a PPI.10
The DOAC Dashboard provides an ideal setting for investigating the effects of NSAID use, NSAID selectivity, and PPI coprescribing on DOAC bleeding rates. With an increasing population of patients receiving anticoagulation therapy with a DOAC, more guidance regarding the bleeding risk of concomitant NSAID use with DOACs is needed. Studies evaluating the bleeding risk with concomitant NSAID use in patients on a DOAC alone are limited. This is the first study to date to compare bleeding risk with concomitant NSAID use between DOACs. This study provides information on bleeding risk with NSAID use among commonly prescribed DOACs, rivaroxaban and apixaban, and the potential impacts of current risk reduction strategies.
METHODS
This single-center retrospective cohort review was performed using the electronic health records (EHRs) of patients enrolled in the US Department of Veterans Affairs (VA) Mountain Home Healthcare System who received rivaroxaban or apixaban from December 2020 to December 2022. This study received approval from the East Tennessee State University/VA Institutional Review Board committee.
Patients were identified through the DOAC Dashboard, aged 21 to 100 years, and received rivaroxaban or apixaban at a therapeutic dose: rivaroxaban 10 to 20 mg daily or apixaban 2.5 to 5 mg twice daily. Patients were excluded if they were prescribed dual antiplatelet therapy, received rivaroxaban at dosing indicated for peripheral vascular disease, were undergoing dialysis, had evidence of moderate to severe hepatic impairment or any hepatic disease with coagulopathy, were undergoing chemotherapy or radiation, or had hematological conditions with predisposed bleeding risk. These patients were excluded to mitigate the potential confounding impact from nontherapeutic DOAC dosing strategies and conditions associated with an increased bleeding risk.
Eligible patients were stratified based on NSAID use. NSAID users were defined as patients prescribed an oral NSAID, including both acute and chronic courses, at any point during the study time frame while actively on a DOAC. Bleeding events were reviewed to evaluate rates between rivaroxaban and apixaban among NSAID and nonNSAID users. Identified NSAID users were further assessed for NSAID selectivity and PPI coprescribing as a subgroup analysis for the secondary assessment.
Data Collection
Baseline data were collected, including age, body mass index, anticoagulation indication, DOAC agent, DOAC dose, and DOAC total daily dose. Baseline serum creatinine levels, liver function tests, hemoglobin levels, and platelet counts were collected from the most recent data available immediately prior to the bleeding event, if applicable.
The DOAC Dashboard was reviewed for active and dismissed drug interaction alerts to identify patients taking rivaroxaban or apixaban who were prescribed an NSAID. Patients were categorized in the NSAID group if an interacting drug alert with an NSAID was reported during the study time frame. Data available through the interacting drug alerts on NSAID use were limited to the interacting drug name and date of the reported flag. Manual EHR review was required to confirm dates of NSAID therapy initiation and NSAID discontinuation, if applicable.
Data regarding concomitant antiplatelet use were obtained through review of the active and dismissed drug interaction alerts on the DOAC Dashboard. Concomitant antiplatelet use was defined as the prescribing of a single antiplatelet agent at any point while receiving DOAC therapy. Data on concomitant antiplatelets were collected regardless of NSAID status.
Data on coprescribed PPI therapy were obtained through manual EHR review of identified NSAID users. Coprescribed PPI therapy was defined as the prescribing of a PPI at any point during NSAID therapy. Data regarding PPI use among non-NSAID users were not collected because the secondary endpoint was designed to assess PPI use only among patients coprescribed a DOAC and NSAID.
Outcomes
Bleeding events were identified through an outcomes report generated by the DOAC Dashboard based on International Classification of Diseases, Tenth Revision diagnosis codes associated with a bleeding event. The outcomes report captures diagnoses from the outpatient and inpatient care settings. Reported bleeding events were limited to patients who received a DOAC at any point in the 6 months prior to the event and excluded patients with recent DOAC initiation within 7 days of the event, as these patients are not captured on the DOAC Dashboard.
All reported bleeding events were manually reviewed in the EHR and categorized as a major or clinically relevant nonmajor bleed, according to International Society of Thrombosis and Haemostasis criteria. Validated bleeding events were then crossreferenced with the interacting drug alerts report to identify events with potentially overlapping NSAID therapy at the time of the event. Overlapping NSAID therapy was defined as the prescribing of an NSAID at any point in the 6 months prior to the event. All events with potential overlapping NSAID therapies were manually reviewed for confirmation of NSAID status at the time of the event.
The primary endpoint was a composite of any bleeding event per International Society of Thrombosis and Haemostasis criteria. The secondary endpoint evaluated the potential impact of NSAID selectivity or PPI coprescribing on the bleeding rate among the NSAID user groups.
Statistical Analysis
Analyses were performed consistent with the methods used in the ARISTOTLE and RE-LY trials. It was determined that a sample size of 504 patients, with ≥ 168 patients in each group, would provide 80% power using a 2-sided a of 0.05. HRs with 95% CIs and respective P values were calculated using a SPSS-adapted online calculator.
RESULTS
The DOAC Dashboard identified 681 patients on rivaroxaban and 3225 patients on apixaban; 72 patients on rivaroxaban (10.6%) and 300 patients on apixaban (9.3%) were NSAID users. The mean age of NSAID users was 66.9 years in the rivaroxaban group and 72.4 years in the apixaban group. The mean age of non-NSAID users was 71.5 years in the rivaroxaban group and 75.6 years in the apixaban group. No appreciable differences were observed among subgroups in body mass index, renal function, hepatic function, hemoglobin, or platelet counts, and no statistically significant differences were identified (Table 1). Antiplatelet agents identified included aspirin, clopidogrel, prasugrel, and ticagrelor. Fifteen patients (20.3%) in the rivaroxaban group and 87 patients (28.7%) in the apixaban group had concomitant antiplatelet and NSAID use. Forty-five patients on rivaroxaban (60.8%) and 170 (55.9%) on apixaban were prescribed concomitant PPI and NSAID at baseline. Among non-NSAID users, there was concomitant antiplatelet use for 265 patients (43.6%) in the rivaroxaban group and 1401 patients (47.9%) in the apixaban group. Concomitant PPI use was identified among 63 patients (60.0%) taking selective NSAIDs and 182 (57.2%) taking nonselective NSAIDs.

A total of 423 courses of NSAIDs were identified: 85 NSAID courses in the rivaroxaban group and 338 NSAID courses in the apixaban group. Most NSAID courses involved a nonselective NSAID in the rivaroxaban and apixaban NSAID user groups: 75.2% (n = 318) aggregately compared to 71.8% (n = 61) and 76.0% (n = 257) in the rivaroxaban and apixaban groups, respectively. The most frequent NSAID courses identified were meloxicam (26.7%; n = 113), celecoxib (24.8%; n = 105), ibuprofen (19.1%; n = 81), and naproxen (13.5%; n = 57). Data regarding NSAID therapy initiation and discontinuation dates were not readily available. As a result, the duration of NSAID courses was not captured.
There was no statistically significant difference in bleeding rates between rivaroxaban and apixaban among NSAID users (HR 1.04; 95% CI, 0.98-1.12) or non-NSAID users (HR 1.15; 95% CI, 0.80-1.66) (Table 2). Apixaban non-NSAID users had a higher rate of major bleeds (HR 0.32; 95% CI, 0.17-0.61) while rivaroxaban non-NSAID users had a higher rate of clinically relevant nonmajor bleeds (HR 1.63; 95% CI, 1.10-2.54).

The sample size for the secondary endpoint consisted of bleeding events that were confirmed to have had an overlapping NSAID prescribed at the time of the event. For this secondary assessment, there was 1 rivaroxaban NSAID user bleeding event and 4 apixaban NSAID user bleeding events. For the rivaroxaban NSAID user bleeding event, the NSAID was nonselective and a PPI was not coprescribed. For the apixaban NSAID user bleeding events, 2 NSAIDs were nonselective and 2 were selective. All patients with apixaban and NSAID bleeding events had a coprescribed PPI. There was no clinically significant difference in the bleeding rates observed for NSAID selectivity or PPI coprescribing among the NSAID user subgroups.
DISCUSSION
This study found that there was no statistically significant difference for bleeding rates of major and nonmajor bleeding events between rivaroxaban and apixaban among NSAID users and non-NSAID users. This study did not identify a clinically significant impact on bleeding rates from NSAID selectivity or PPI coprescribing among the NSAID users.
There were notable but not statistically significant differences in baseline characteristics observed between the NSAID and non-NSAID user groups. On average, the rivaroxaban and apixaban NSAID users were younger compared with those not taking NSAIDs. NSAIDs, specifically nonselective NSAIDs, are recognized as potentially inappropriate medications for older adults given that this population is at an increased risk for GI ulcer development and/or GI bleeding.17 The non-NSAID user group likely consisted of older patients compared to the NSAID user group as clinicians may avoid prescribing NSAIDs to older adults regardless of concomitant DOAC therapy.
In addition to having an older patient population, non-NSAID users were more frequently prescribed a concomitant antiplatelet when compared with NSAID users. This prescribing pattern may be due to clinicians avoiding the use of NSAIDs in patients receiving DOAC therapy in combination with antiplatelet therapy, as these patients have been found to have an increased bleeding rate compared to DOAC therapy alone.18
Non-NSAID users had an overall higher bleeding rate for both major and nonmajor bleeding events. Based on this observation, it could be hypothesized that antiplatelet agents have a higher risk of bleeding in comparison to NSAIDs. In a subanalysis of the EXPAND study evaluating risk factors of major bleeding in patients receiving rivaroxaban, concomitant use of antiplatelet agents demonstrated a statistically significant increased risk of bleeding (HR 1.6; 95% CI, 1.2-2.3; P = .003) while concomitant use of NSAIDs did not (HR 0.8; 95% CI, 0.3-2.2; P = .67).19
In assessing PPI status at baseline, a majority of both rivaroxaban and apixaban NSAID users were coprescribed a PPI. This trend aligns with current clinical guideline recommendations for the prescribing of PPI therapy for GI protection in high-risk patients, such as those on DOAC therapy and concomitant NSAID therapy.10 Given the high proportion of NSAID users coprescribed a PPI at baseline, it may be possible that the true incidence of NSAID-associated bleeding events was higher than what this study found. This observation may reflect the impact from timely implementation of risk mitigation strategies by CPPs in the anticoagulation clinic. However, this study was not constructed to assess the efficacy of PPI use in this manner.
It is important to note the patients included in this study were followed by a pharmacist in an anticoagulation clinic using the DOAC Dashboard.15 This population management tool allows CPPs to make proactive interventions when a patient taking a DOAC receives an NSAID prescription, such as recommending the coprescribing of a PPI or use of a selective NSAID.10,16 These standards of care may have contributed to an overall reduced bleeding rate among the NSAID user group and may not be reflective of private practice.
The planned analysis of this study was modeled after the posthoc analysis of the RE-LY and ARISTOTLE trials. Both trials demonstrated an increased risk of bleeding with oral anticoagulation, including DOAC and warfarin, in combination with NSAID use. However, both trials found that NSAID use in patients treated with a DOAC was not independently associated with increased bleeding events compared with warfarin.13,14 The results of this study are comparable to the RE-LY and ARISTOTLE findings that NSAID use among patients treated with rivaroxaban or apixaban did not demonstrate a statistically significant increased bleeding risk.
Studies of NSAID use in combination with DOAC therapy have been limited to patient populations consisting of both DOAC and warfarin. Evidence from these trials outlines the increased bleeding risk associated with NSAID use in combination with oral anticoagulation; however, these patient populations include those on a DOAC and warfarin.13,14,19,20 Given the limited evidence on NSAID use among DOACs alone, it is assumed NSAID use in combination with DOACs has a similar risk of bleeding as warfarin use. This may cause clinicians to automatically exclude NSAID therapy as a treatment option for patients on a DOAC who are otherwise clinically appropriate candidates, such as those with underlying inflammatory conditions. Avoiding NSAID therapy in this patient population may lead to suboptimal pain management and increase the risk of patient harm from methods such as inappropriate opioid therapy prescribing.
DOAC therapy should not be a universal limitation to the use of NSAIDs. Although the risk of bleeding with NSAID therapy is always present, deliberate NSAID prescribing in addition to the timely implementation of risk mitigation strategies may provide an avenue for safe NSAID prescribing in patients receiving a DOAC. A population health-based approach to DOAC management, such as the DOAC Dashboard, appears to be effective at preventing patient harm when NSAIDs are prescribed in conjunction with DOACs.
Limitations
The DOAC Dashboard has been shown to be effective and efficient at monitoring DOAC therapy from a population-based approach.16 Reports generated through the DOAC Dashboard provide convenient access to patient data which allows for timely interventions; however, there are limits to its use for data collection. All the data elements necessary to properly assess bleeding risk with validated tools, such as HAS-BLED (hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, labile international normalized ratio, elderly, drugs/ alcohol concomitantly), are not available on DOAC Dashboard reports. Due to this constraint, bleeding risk assessments were not conducted at baseline and this study was unable to include risk modeling. Additionally, data elements like initiation and discontinuation dates and duration of therapies were not readily available. As a result, this study was unable to incorporate time as a data point.
This was a retrospective study that relied on manual review of chart documentation to verify bleeding events, but data obtained through the DOAC Dashboard were transferred directly from the EHR.15 Bleeding events available for evaluation were restricted to those that occurred at a VA facility. Additionally, the sample size within the rivaroxaban NSAID user group did not reach the predefined sample size required to reach power and may have been too small to detect a difference if one did exist. The secondary assessment had a low sample size of NSAID user bleeding events, making it difficult to fully assess its impact on NSAID selectivity and PPI coprescribing on bleeding rates. All courses of NSAIDs were equally valued regardless of the dose or therapy duration; however, this is consistent with how NSAID use was defined in the RE-LY and ARISTOTLE trials.
CONCLUSIONS
This retrospective cohort review found no statistically significant difference in the composite bleeding rates between rivaroxaban and apixaban among NSAID users and non-NSAID users. Moreover, there was no clinically significant impact observed for bleeding rates in regard to NSAID selectivity and PPI coprescribing among NSAID users. However, coprescribing of PPI therapy to patients on a DOAC who are clinically indicated for an NSAID may reduce the risk of bleeding. Population health management tools, such as the DOAC Dashboard, may also allow clinicians to safely prescribe NSAIDs to patients on a DOAC. Further large-scale observational studies are needed to quantify the real-world risk of bleeding with concomitant NSAID use among DOACs alone and to evaluate the impact from NSAID selectivity or PPI coprescribing.
Clinical practice has shifted from vitamin K antagonists to direct oral anticoagulants (DOACs) for atrial fibrillation treatment due to their more favorable risk-benefit profile and less lifestyle modification required.1,2 However, the advantage of a lower bleeding risk with DOACs could be compromised by potentially problematic pharmacokinetic interactions like those conferred by antiplatelets or nonsteroidal anti-inflammatory drugs (NSAIDs).3,4 Treating a patient needing anticoagulation with a DOAC who has comorbidities may introduce unavoidable drug-drug interactions. This particularly happens with over-the-counter and prescription NSAIDs used for the management of pain and inflammatory conditions.5
NSAIDs primarily affect 2 cyclooxygenase (COX) enzyme isomers, COX-1 and COX-2.6 COX-1 helps maintain gastrointestinal (GI) mucosa integrity and platelet aggregation processes, whereas COX-2 is engaged in pain signaling and inflammation mediation. COX-1 inhibition is associated with more bleeding-related adverse events (AEs), especially in the GI tract. COX-2 inhibition is thought to provide analgesia and anti-inflammatory properties without elevating bleeding risk. This premise is responsible for the preferential use of celecoxib, a COX-2 selective NSAID, which should confer a lower bleeding risk compared to nonselective NSAIDs such as ibuprofen and naproxen.7 NSAIDs have been documented as independent risk factors for bleeding. NSAID users are about 3 times as likely to develop GI AEs compared to nonNSAID users.8
Many clinicians aim to further mitigate NSAID-associated bleeding risk by coprescribing a proton pump inhibitor (PPI). PPIs provide gastroprotection against NSAID-induced mucosal injury and sequential complication of GI bleeding. In a multicenter randomized control trial, patients who received concomitant PPI therapy while undergoing chronic NSAID therapy—including nonselective and COX-2 selective NSAIDs—had a significantly lower risk of GI ulcer development (placebo, 17.0%; 20 mg esomeprazole, 5.2%; 40 mg esomeprazole, 4.6%).9 Current clinical guidelines for preventing NSAIDassociated bleeding complications recommend using a COX-2 selective NSAID in combination with PPI therapy for patients at high risk for GI-related bleeding, including the concomitant use of anticoagulants.10
There is evidence suggesting an increased bleeding risk with NSAIDs when used in combination with vitamin K antagonists such as warfarin.11,12 A systematic review of warfarin and concomitant NSAID use found an increased risk of overall bleeding with NSAID use in combination with warfarin (odds ratio 1.58; 95% CI, 1.18-2.12), compared to warfarin alone.12
Posthoc analyses of randomized clinical trials have also demonstrated an increased bleeding risk with oral anticoagulation and concomitant NSAID use.13,14 In the RE-LY trial, NSAID users on warfarin or dabigatran had a statistically significant increased risk of major bleeding compared to non-NSAID users (hazard ratio [HR] 1.68; 95% CI, 1.40- 2.02; P < .001).13 In the ARISTOTLE trial, patients on warfarin or apixaban who were incident NSAID users were found to have an increased risk of major bleeding (HR 1.61; 95% CI, 1.11-2.33) and clinically relevant nonmajor bleeding (HR 1.70; 95% CI, 1.16- 2.48).14 These trials found a statistically significant increased bleeding risk associated with NSAID use, though the populations evaluated included patients taking warfarin and patients taking DOACs. These trials did not evaluate the bleeding risk of concomitant NSAID use among DOACs alone.
Evidence on NSAID-associated bleeding risk with DOACs is lacking in settings where the patient population, prescribing practices, and monitoring levels are variable. Within the Veterans Health Administration, clinical pharmacist practitioners (CPPs) in anticoagulation clinics oversee DOAC therapy management. CPPs monitor safety and efficacy of DOAC therapies through a population health management tool, the DOAC Dashboard.15 The DOAC Dashboard creates alerts for patients who may require an intervention based on certain clinical parameters, such as drug-drug interactions.16 Whenever a patient on a DOAC is prescribed an NSAID, an alert is generated on the DOAC Dashboard to flag the CPPs for the potential need for an intervention. If NSAID therapy remains clinically indicated, CPPs may recommend risk reduction strategies such as a COX-2 selective NSAID or coprescribing a PPI.10
The DOAC Dashboard provides an ideal setting for investigating the effects of NSAID use, NSAID selectivity, and PPI coprescribing on DOAC bleeding rates. With an increasing population of patients receiving anticoagulation therapy with a DOAC, more guidance regarding the bleeding risk of concomitant NSAID use with DOACs is needed. Studies evaluating the bleeding risk with concomitant NSAID use in patients on a DOAC alone are limited. This is the first study to date to compare bleeding risk with concomitant NSAID use between DOACs. This study provides information on bleeding risk with NSAID use among commonly prescribed DOACs, rivaroxaban and apixaban, and the potential impacts of current risk reduction strategies.
METHODS
This single-center retrospective cohort review was performed using the electronic health records (EHRs) of patients enrolled in the US Department of Veterans Affairs (VA) Mountain Home Healthcare System who received rivaroxaban or apixaban from December 2020 to December 2022. This study received approval from the East Tennessee State University/VA Institutional Review Board committee.
Patients were identified through the DOAC Dashboard, aged 21 to 100 years, and received rivaroxaban or apixaban at a therapeutic dose: rivaroxaban 10 to 20 mg daily or apixaban 2.5 to 5 mg twice daily. Patients were excluded if they were prescribed dual antiplatelet therapy, received rivaroxaban at dosing indicated for peripheral vascular disease, were undergoing dialysis, had evidence of moderate to severe hepatic impairment or any hepatic disease with coagulopathy, were undergoing chemotherapy or radiation, or had hematological conditions with predisposed bleeding risk. These patients were excluded to mitigate the potential confounding impact from nontherapeutic DOAC dosing strategies and conditions associated with an increased bleeding risk.
Eligible patients were stratified based on NSAID use. NSAID users were defined as patients prescribed an oral NSAID, including both acute and chronic courses, at any point during the study time frame while actively on a DOAC. Bleeding events were reviewed to evaluate rates between rivaroxaban and apixaban among NSAID and nonNSAID users. Identified NSAID users were further assessed for NSAID selectivity and PPI coprescribing as a subgroup analysis for the secondary assessment.
Data Collection
Baseline data were collected, including age, body mass index, anticoagulation indication, DOAC agent, DOAC dose, and DOAC total daily dose. Baseline serum creatinine levels, liver function tests, hemoglobin levels, and platelet counts were collected from the most recent data available immediately prior to the bleeding event, if applicable.
The DOAC Dashboard was reviewed for active and dismissed drug interaction alerts to identify patients taking rivaroxaban or apixaban who were prescribed an NSAID. Patients were categorized in the NSAID group if an interacting drug alert with an NSAID was reported during the study time frame. Data available through the interacting drug alerts on NSAID use were limited to the interacting drug name and date of the reported flag. Manual EHR review was required to confirm dates of NSAID therapy initiation and NSAID discontinuation, if applicable.
Data regarding concomitant antiplatelet use were obtained through review of the active and dismissed drug interaction alerts on the DOAC Dashboard. Concomitant antiplatelet use was defined as the prescribing of a single antiplatelet agent at any point while receiving DOAC therapy. Data on concomitant antiplatelets were collected regardless of NSAID status.
Data on coprescribed PPI therapy were obtained through manual EHR review of identified NSAID users. Coprescribed PPI therapy was defined as the prescribing of a PPI at any point during NSAID therapy. Data regarding PPI use among non-NSAID users were not collected because the secondary endpoint was designed to assess PPI use only among patients coprescribed a DOAC and NSAID.
Outcomes
Bleeding events were identified through an outcomes report generated by the DOAC Dashboard based on International Classification of Diseases, Tenth Revision diagnosis codes associated with a bleeding event. The outcomes report captures diagnoses from the outpatient and inpatient care settings. Reported bleeding events were limited to patients who received a DOAC at any point in the 6 months prior to the event and excluded patients with recent DOAC initiation within 7 days of the event, as these patients are not captured on the DOAC Dashboard.
All reported bleeding events were manually reviewed in the EHR and categorized as a major or clinically relevant nonmajor bleed, according to International Society of Thrombosis and Haemostasis criteria. Validated bleeding events were then crossreferenced with the interacting drug alerts report to identify events with potentially overlapping NSAID therapy at the time of the event. Overlapping NSAID therapy was defined as the prescribing of an NSAID at any point in the 6 months prior to the event. All events with potential overlapping NSAID therapies were manually reviewed for confirmation of NSAID status at the time of the event.
The primary endpoint was a composite of any bleeding event per International Society of Thrombosis and Haemostasis criteria. The secondary endpoint evaluated the potential impact of NSAID selectivity or PPI coprescribing on the bleeding rate among the NSAID user groups.
Statistical Analysis
Analyses were performed consistent with the methods used in the ARISTOTLE and RE-LY trials. It was determined that a sample size of 504 patients, with ≥ 168 patients in each group, would provide 80% power using a 2-sided a of 0.05. HRs with 95% CIs and respective P values were calculated using a SPSS-adapted online calculator.
RESULTS
The DOAC Dashboard identified 681 patients on rivaroxaban and 3225 patients on apixaban; 72 patients on rivaroxaban (10.6%) and 300 patients on apixaban (9.3%) were NSAID users. The mean age of NSAID users was 66.9 years in the rivaroxaban group and 72.4 years in the apixaban group. The mean age of non-NSAID users was 71.5 years in the rivaroxaban group and 75.6 years in the apixaban group. No appreciable differences were observed among subgroups in body mass index, renal function, hepatic function, hemoglobin, or platelet counts, and no statistically significant differences were identified (Table 1). Antiplatelet agents identified included aspirin, clopidogrel, prasugrel, and ticagrelor. Fifteen patients (20.3%) in the rivaroxaban group and 87 patients (28.7%) in the apixaban group had concomitant antiplatelet and NSAID use. Forty-five patients on rivaroxaban (60.8%) and 170 (55.9%) on apixaban were prescribed concomitant PPI and NSAID at baseline. Among non-NSAID users, there was concomitant antiplatelet use for 265 patients (43.6%) in the rivaroxaban group and 1401 patients (47.9%) in the apixaban group. Concomitant PPI use was identified among 63 patients (60.0%) taking selective NSAIDs and 182 (57.2%) taking nonselective NSAIDs.

A total of 423 courses of NSAIDs were identified: 85 NSAID courses in the rivaroxaban group and 338 NSAID courses in the apixaban group. Most NSAID courses involved a nonselective NSAID in the rivaroxaban and apixaban NSAID user groups: 75.2% (n = 318) aggregately compared to 71.8% (n = 61) and 76.0% (n = 257) in the rivaroxaban and apixaban groups, respectively. The most frequent NSAID courses identified were meloxicam (26.7%; n = 113), celecoxib (24.8%; n = 105), ibuprofen (19.1%; n = 81), and naproxen (13.5%; n = 57). Data regarding NSAID therapy initiation and discontinuation dates were not readily available. As a result, the duration of NSAID courses was not captured.
There was no statistically significant difference in bleeding rates between rivaroxaban and apixaban among NSAID users (HR 1.04; 95% CI, 0.98-1.12) or non-NSAID users (HR 1.15; 95% CI, 0.80-1.66) (Table 2). Apixaban non-NSAID users had a higher rate of major bleeds (HR 0.32; 95% CI, 0.17-0.61) while rivaroxaban non-NSAID users had a higher rate of clinically relevant nonmajor bleeds (HR 1.63; 95% CI, 1.10-2.54).

The sample size for the secondary endpoint consisted of bleeding events that were confirmed to have had an overlapping NSAID prescribed at the time of the event. For this secondary assessment, there was 1 rivaroxaban NSAID user bleeding event and 4 apixaban NSAID user bleeding events. For the rivaroxaban NSAID user bleeding event, the NSAID was nonselective and a PPI was not coprescribed. For the apixaban NSAID user bleeding events, 2 NSAIDs were nonselective and 2 were selective. All patients with apixaban and NSAID bleeding events had a coprescribed PPI. There was no clinically significant difference in the bleeding rates observed for NSAID selectivity or PPI coprescribing among the NSAID user subgroups.
DISCUSSION
This study found that there was no statistically significant difference for bleeding rates of major and nonmajor bleeding events between rivaroxaban and apixaban among NSAID users and non-NSAID users. This study did not identify a clinically significant impact on bleeding rates from NSAID selectivity or PPI coprescribing among the NSAID users.
There were notable but not statistically significant differences in baseline characteristics observed between the NSAID and non-NSAID user groups. On average, the rivaroxaban and apixaban NSAID users were younger compared with those not taking NSAIDs. NSAIDs, specifically nonselective NSAIDs, are recognized as potentially inappropriate medications for older adults given that this population is at an increased risk for GI ulcer development and/or GI bleeding.17 The non-NSAID user group likely consisted of older patients compared to the NSAID user group as clinicians may avoid prescribing NSAIDs to older adults regardless of concomitant DOAC therapy.
In addition to having an older patient population, non-NSAID users were more frequently prescribed a concomitant antiplatelet when compared with NSAID users. This prescribing pattern may be due to clinicians avoiding the use of NSAIDs in patients receiving DOAC therapy in combination with antiplatelet therapy, as these patients have been found to have an increased bleeding rate compared to DOAC therapy alone.18
Non-NSAID users had an overall higher bleeding rate for both major and nonmajor bleeding events. Based on this observation, it could be hypothesized that antiplatelet agents have a higher risk of bleeding in comparison to NSAIDs. In a subanalysis of the EXPAND study evaluating risk factors of major bleeding in patients receiving rivaroxaban, concomitant use of antiplatelet agents demonstrated a statistically significant increased risk of bleeding (HR 1.6; 95% CI, 1.2-2.3; P = .003) while concomitant use of NSAIDs did not (HR 0.8; 95% CI, 0.3-2.2; P = .67).19
In assessing PPI status at baseline, a majority of both rivaroxaban and apixaban NSAID users were coprescribed a PPI. This trend aligns with current clinical guideline recommendations for the prescribing of PPI therapy for GI protection in high-risk patients, such as those on DOAC therapy and concomitant NSAID therapy.10 Given the high proportion of NSAID users coprescribed a PPI at baseline, it may be possible that the true incidence of NSAID-associated bleeding events was higher than what this study found. This observation may reflect the impact from timely implementation of risk mitigation strategies by CPPs in the anticoagulation clinic. However, this study was not constructed to assess the efficacy of PPI use in this manner.
It is important to note the patients included in this study were followed by a pharmacist in an anticoagulation clinic using the DOAC Dashboard.15 This population management tool allows CPPs to make proactive interventions when a patient taking a DOAC receives an NSAID prescription, such as recommending the coprescribing of a PPI or use of a selective NSAID.10,16 These standards of care may have contributed to an overall reduced bleeding rate among the NSAID user group and may not be reflective of private practice.
The planned analysis of this study was modeled after the posthoc analysis of the RE-LY and ARISTOTLE trials. Both trials demonstrated an increased risk of bleeding with oral anticoagulation, including DOAC and warfarin, in combination with NSAID use. However, both trials found that NSAID use in patients treated with a DOAC was not independently associated with increased bleeding events compared with warfarin.13,14 The results of this study are comparable to the RE-LY and ARISTOTLE findings that NSAID use among patients treated with rivaroxaban or apixaban did not demonstrate a statistically significant increased bleeding risk.
Studies of NSAID use in combination with DOAC therapy have been limited to patient populations consisting of both DOAC and warfarin. Evidence from these trials outlines the increased bleeding risk associated with NSAID use in combination with oral anticoagulation; however, these patient populations include those on a DOAC and warfarin.13,14,19,20 Given the limited evidence on NSAID use among DOACs alone, it is assumed NSAID use in combination with DOACs has a similar risk of bleeding as warfarin use. This may cause clinicians to automatically exclude NSAID therapy as a treatment option for patients on a DOAC who are otherwise clinically appropriate candidates, such as those with underlying inflammatory conditions. Avoiding NSAID therapy in this patient population may lead to suboptimal pain management and increase the risk of patient harm from methods such as inappropriate opioid therapy prescribing.
DOAC therapy should not be a universal limitation to the use of NSAIDs. Although the risk of bleeding with NSAID therapy is always present, deliberate NSAID prescribing in addition to the timely implementation of risk mitigation strategies may provide an avenue for safe NSAID prescribing in patients receiving a DOAC. A population health-based approach to DOAC management, such as the DOAC Dashboard, appears to be effective at preventing patient harm when NSAIDs are prescribed in conjunction with DOACs.
Limitations
The DOAC Dashboard has been shown to be effective and efficient at monitoring DOAC therapy from a population-based approach.16 Reports generated through the DOAC Dashboard provide convenient access to patient data which allows for timely interventions; however, there are limits to its use for data collection. All the data elements necessary to properly assess bleeding risk with validated tools, such as HAS-BLED (hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, labile international normalized ratio, elderly, drugs/ alcohol concomitantly), are not available on DOAC Dashboard reports. Due to this constraint, bleeding risk assessments were not conducted at baseline and this study was unable to include risk modeling. Additionally, data elements like initiation and discontinuation dates and duration of therapies were not readily available. As a result, this study was unable to incorporate time as a data point.
This was a retrospective study that relied on manual review of chart documentation to verify bleeding events, but data obtained through the DOAC Dashboard were transferred directly from the EHR.15 Bleeding events available for evaluation were restricted to those that occurred at a VA facility. Additionally, the sample size within the rivaroxaban NSAID user group did not reach the predefined sample size required to reach power and may have been too small to detect a difference if one did exist. The secondary assessment had a low sample size of NSAID user bleeding events, making it difficult to fully assess its impact on NSAID selectivity and PPI coprescribing on bleeding rates. All courses of NSAIDs were equally valued regardless of the dose or therapy duration; however, this is consistent with how NSAID use was defined in the RE-LY and ARISTOTLE trials.
CONCLUSIONS
This retrospective cohort review found no statistically significant difference in the composite bleeding rates between rivaroxaban and apixaban among NSAID users and non-NSAID users. Moreover, there was no clinically significant impact observed for bleeding rates in regard to NSAID selectivity and PPI coprescribing among NSAID users. However, coprescribing of PPI therapy to patients on a DOAC who are clinically indicated for an NSAID may reduce the risk of bleeding. Population health management tools, such as the DOAC Dashboard, may also allow clinicians to safely prescribe NSAIDs to patients on a DOAC. Further large-scale observational studies are needed to quantify the real-world risk of bleeding with concomitant NSAID use among DOACs alone and to evaluate the impact from NSAID selectivity or PPI coprescribing.
- Ruff CT, Giugliano RP, Braunwald E, et al. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a meta-analysis of randomised trials. Lancet. 2014;383(9921):955-962. doi:10.1016/S0140-6736(13)62343-0
- Ageno W, Gallus AS, Wittkowsky A, Crowther M, Hylek EM, Palareti G. Oral anticoagulant therapy: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e44S-e88S. doi:10.1378/chest.11-2292
- Eikelboom J, Merli G. Bleeding with direct oral anticoagulants vs warfarin: clinical experience. Am J Med. 2016;129(11S):S33-S40. doi:10.1016/j.amjmed.2016.06.003
- Vranckx P, Valgimigli M, Heidbuchel H. The significance of drug-drug and drug-food interactions of oral anticoagulation. Arrhythm Electrophysiol Rev. 2018;7(1):55-61. doi:10.15420/aer.2017.50.1
- Davis JS, Lee HY, Kim J, et al. Use of non-steroidal antiinflammatory drugs in US adults: changes over time and by demographic. Open Heart. 2017;4(1):e000550. doi:10.1136/openhrt-2016-000550
- Schafer AI. Effects of nonsteroidal antiinflammatory drugs on platelet function and systemic hemostasis. J Clin Pharmacol. 1995;35(3):209-219. doi:10.1002/j.1552-4604.1995.tb04050.x
- Al-Saeed A. Gastrointestinal and cardiovascular risk of nonsteroidal anti-inflammatory drugs. Oman Med J. 2011;26(6):385-391. doi:10.5001/omj.2011.101
- Gabriel SE, Jaakkimainen L, Bombardier C. Risk for serious gastrointestinal complications related to use of nonsteroidal anti-inflammatory drugs. Ann Intern Med. 1991;115(10):787-796. doi:10.7326/0003-4819-115-10-787
- Scheiman JM, Yeomans ND, Talley NJ, et al. Prevention of ulcers by esomeprazole in at-risk patients using non-selective NSAIDs and COX-2 inhibitors. Am J Gastroenterol. 2006;101(4):701-710. doi:10.1111/j.1572-0241.2006.00499.x
- Freedberg DE, Kim LS, Yang YX. The risks and benefits of long-term use of proton pump inhibitors: expert review and best practice advice from the American Gastroenterological Association. Gastroenterology. 2017;152(4):706-715. doi:10.1053/j.gastro.2017.01.031
- Lamberts M, Lip GYH, Hansen ML, et al. Relation of nonsteroidal anti-inflammatory drugs to serious bleeding and thromboembolism risk in patients with atrial fibrillation receiving antithrombotic therapy: a nationwide cohort study. Ann Intern Med. 2014;161(10):690-698. doi:10.7326/M13-1581
- Villa Zapata L, Hansten PD, Panic J, et al. Risk of bleeding with exposure to warfarin and nonsteroidal anti-inflammatory drugs: a systematic review and metaanalysis. Thromb Haemost. 2020;120(7):1066-1074. doi:10.1055/s-0040-1710592
- Kent AP, Brueckmann M, Fraessdorf M, et al. Concomitant oral anticoagulant and nonsteroidal anti-inflammatory drug therapy in patients with atrial fibrillation. J Am Coll Cardiol. 2018;72(3):255-267. doi:10.1016/j.jacc.2018.04.063
- Dalgaard F, Mulder H, Wojdyla DM, et al. Patients with atrial fibrillation taking nonsteroidal antiinflammatory drugs and oral anticoagulants in the ARISTOTLE Trial. Circulation. 2020;141(1):10-20. doi:10.1161/CIRCULATIONAHA.119.041296
- Allen AL, Lucas J, Parra D, et al. Shifting the paradigm: a population health approach to the management of direct oral anticoagulants. J Am Heart Asssoc. 2021;10(24):e022758. doi:10.1161/JAHA.121.022758
- . Valencia D, Spoutz P, Stoppi J, et al. Impact of a direct oral anticoagulant population management tool on anticoagulation therapy monitoring in clinical practice. Ann Pharmacother. 2019;53(8):806-811. doi:10.1177/1060028019835843
- By the 2023 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2023 Updated AGS Beers Criteria® for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2023;71(7):2052-2081. doi:10.1111/jgs.18372
- Kumar S, Danik SB, Altman RK, et al. Non-vitamin K antagonist oral anticoagulants and antiplatelet therapy for stroke prevention in patients with atrial fibrillation. Cardiol Rev. 2016;24(5):218-223. doi:10.1097/CRD.0000000000000088
- Sakuma I, Uchiyama S, Atarashi H, et al. Clinical risk factors of stroke and major bleeding in patients with nonvalvular atrial fibrillation under rivaroxaban: the EXPAND study sub-analysis. Heart Vessels. 2019;34(11):1839-1851. doi:10.1007/s00380-019-01425-x
- Davidson BL, Verheijen S, Lensing AWA, et al. Bleeding risk of patients with acute venous thromboembolism taking nonsteroidal anti-inflammatory drugs or aspirin. JAMA Intern Med. 2014;174(6):947-953. doi:10.1001/jamainternmed.2014.946
- Ruff CT, Giugliano RP, Braunwald E, et al. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a meta-analysis of randomised trials. Lancet. 2014;383(9921):955-962. doi:10.1016/S0140-6736(13)62343-0
- Ageno W, Gallus AS, Wittkowsky A, Crowther M, Hylek EM, Palareti G. Oral anticoagulant therapy: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e44S-e88S. doi:10.1378/chest.11-2292
- Eikelboom J, Merli G. Bleeding with direct oral anticoagulants vs warfarin: clinical experience. Am J Med. 2016;129(11S):S33-S40. doi:10.1016/j.amjmed.2016.06.003
- Vranckx P, Valgimigli M, Heidbuchel H. The significance of drug-drug and drug-food interactions of oral anticoagulation. Arrhythm Electrophysiol Rev. 2018;7(1):55-61. doi:10.15420/aer.2017.50.1
- Davis JS, Lee HY, Kim J, et al. Use of non-steroidal antiinflammatory drugs in US adults: changes over time and by demographic. Open Heart. 2017;4(1):e000550. doi:10.1136/openhrt-2016-000550
- Schafer AI. Effects of nonsteroidal antiinflammatory drugs on platelet function and systemic hemostasis. J Clin Pharmacol. 1995;35(3):209-219. doi:10.1002/j.1552-4604.1995.tb04050.x
- Al-Saeed A. Gastrointestinal and cardiovascular risk of nonsteroidal anti-inflammatory drugs. Oman Med J. 2011;26(6):385-391. doi:10.5001/omj.2011.101
- Gabriel SE, Jaakkimainen L, Bombardier C. Risk for serious gastrointestinal complications related to use of nonsteroidal anti-inflammatory drugs. Ann Intern Med. 1991;115(10):787-796. doi:10.7326/0003-4819-115-10-787
- Scheiman JM, Yeomans ND, Talley NJ, et al. Prevention of ulcers by esomeprazole in at-risk patients using non-selective NSAIDs and COX-2 inhibitors. Am J Gastroenterol. 2006;101(4):701-710. doi:10.1111/j.1572-0241.2006.00499.x
- Freedberg DE, Kim LS, Yang YX. The risks and benefits of long-term use of proton pump inhibitors: expert review and best practice advice from the American Gastroenterological Association. Gastroenterology. 2017;152(4):706-715. doi:10.1053/j.gastro.2017.01.031
- Lamberts M, Lip GYH, Hansen ML, et al. Relation of nonsteroidal anti-inflammatory drugs to serious bleeding and thromboembolism risk in patients with atrial fibrillation receiving antithrombotic therapy: a nationwide cohort study. Ann Intern Med. 2014;161(10):690-698. doi:10.7326/M13-1581
- Villa Zapata L, Hansten PD, Panic J, et al. Risk of bleeding with exposure to warfarin and nonsteroidal anti-inflammatory drugs: a systematic review and metaanalysis. Thromb Haemost. 2020;120(7):1066-1074. doi:10.1055/s-0040-1710592
- Kent AP, Brueckmann M, Fraessdorf M, et al. Concomitant oral anticoagulant and nonsteroidal anti-inflammatory drug therapy in patients with atrial fibrillation. J Am Coll Cardiol. 2018;72(3):255-267. doi:10.1016/j.jacc.2018.04.063
- Dalgaard F, Mulder H, Wojdyla DM, et al. Patients with atrial fibrillation taking nonsteroidal antiinflammatory drugs and oral anticoagulants in the ARISTOTLE Trial. Circulation. 2020;141(1):10-20. doi:10.1161/CIRCULATIONAHA.119.041296
- Allen AL, Lucas J, Parra D, et al. Shifting the paradigm: a population health approach to the management of direct oral anticoagulants. J Am Heart Asssoc. 2021;10(24):e022758. doi:10.1161/JAHA.121.022758
- . Valencia D, Spoutz P, Stoppi J, et al. Impact of a direct oral anticoagulant population management tool on anticoagulation therapy monitoring in clinical practice. Ann Pharmacother. 2019;53(8):806-811. doi:10.1177/1060028019835843
- By the 2023 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2023 Updated AGS Beers Criteria® for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2023;71(7):2052-2081. doi:10.1111/jgs.18372
- Kumar S, Danik SB, Altman RK, et al. Non-vitamin K antagonist oral anticoagulants and antiplatelet therapy for stroke prevention in patients with atrial fibrillation. Cardiol Rev. 2016;24(5):218-223. doi:10.1097/CRD.0000000000000088
- Sakuma I, Uchiyama S, Atarashi H, et al. Clinical risk factors of stroke and major bleeding in patients with nonvalvular atrial fibrillation under rivaroxaban: the EXPAND study sub-analysis. Heart Vessels. 2019;34(11):1839-1851. doi:10.1007/s00380-019-01425-x
- Davidson BL, Verheijen S, Lensing AWA, et al. Bleeding risk of patients with acute venous thromboembolism taking nonsteroidal anti-inflammatory drugs or aspirin. JAMA Intern Med. 2014;174(6):947-953. doi:10.1001/jamainternmed.2014.946
Impact of NSAID Use on Bleeding Rates for Patients Taking Rivaroxaban or Apixaban
Impact of NSAID Use on Bleeding Rates for Patients Taking Rivaroxaban or Apixaban
Cultural Respect vs Individual Patient Autonomy: A Delicate Balancing Act
Cultural competency is one of the most important values in the practice of medicine. Defined as the “ability to collaborate effectively with individuals from different cultures,” this type of competence “improves healthcare experiences and outcomes.” But within the context of cultural familiarity, it’s equally important to “understand that each person is an individual and may or may not adhere to certain cultural beliefs or practices common in his or her culture,” according to the Agency for Healthcare Research and Quality’s (AHRQ’s) Health Literacy Universal Precautions Toolkit.
Sarah Candler, MD, MPH, an internal medicine physician specializing in primary care for older adults in Washington, DC, said that the medical code of ethics consists of several pillars, with patient autonomy as the “first and most primary of those pillars.” She calls the balance of patient autonomy and cultural respect a “complicated tightrope to walk,” but says that these ethical principles can inform medical decisions and the patient-physician relationship.
Cultural Familiarity
It’s important to be as familiar as possible with the patient’s culture, Santina Wheat, MD, program director, Northwestern McGaw Family Medicine Residency at Delnor Hospital, Geneva, told this news organization. “For example, we serve many Orthodox Jewish patients. We had a meeting with rabbis from the community to present to us what religious laws might affect our patients. Until recently, I was delivering babies, and there was always a 24-hour emergency rabbi on call if an Orthodox patient wanted the input of a rabbi into her decisions.”
Jay W. Lee, MD, MPH, a member of the board of directors of the American Academy of Family Physicians, also sets out to educate himself about the cultural norms of his patients if they come from populations he’s not familiar with. “For example, this comes up when a new refugee population comes to the United States — most recently, there was a population of Afghan refugees,” Lee told this news organization.
Lee spent “a lot of time trying to learn about their cultural norms,” which prepared him to “ask more targeted questions about the patient’s understanding of the tests we were ordering or treatment options we were bringing forward.”
Lee, also the medical director at Integrated Health Partners of Southern California and associate clinical professor of family medicine at the University of California, Irvine, said it might be best if the physician is “language congruent or culturally similar.” Lee is of Asian descent and also speaks Spanish fluently. “I enjoy cultural exchanges with my patients, and I encourage patients to find a physician who’s the best fit.” But being from the same culture isn’t absolutely necessary for building relationships with the patient. “The key is offering the patient autonomy” while understanding the cultural context.
Don’t Assume ... Always Ask
Cultural familiarity doesn’t equate with stereotyping, Wheat emphasized. “Proceeding without assumptions opens the opportunity to ask questions for clarification and understanding and to improve patient care,” said Lee.
Sara Glass, PhD, LCSW, agrees. She’s the clinical director of Soul Wellness NYC, New York City, a psychotherapy practice that specializes in treating trauma. Based on her own experiences, she knows that some physicians and other healthcare professionals confuse cultural sensitivity with cultural stereotyping.
Glass, formerly Hasidic and ultra-Orthodox, shared an example from her own life. During the delivery of her second child, she sustained a vaginal tear. At her 6-week postpartum visit, her ob/gyn said, “Just remind me when you’re in your ninth month next time, and I can sew it up right after you deliver.”
Much of this physician’s practice “consisted of Hasidic women who looked just like me, wearing the same garb — head coverings such as wigs and scarves and long skirts. Most women in that community have multiple pregnancies,” Glass told this news organization. “My sister has 10 children, and that’s not unusual. The doctor simply assumed I’d be going on to have more babies without asking if that’s what I wanted.”
Glass says she was also never given information by her physician about the range of available contraceptive options. The rabbis of the Hasidic sect to which Glass belonged allowed women to practice contraception for 6 months following childbirth, or for longer, in the setting of certain medical conditions, but only certain types of birth control were religiously permissible. Other options were not mentioned to her by her physician, and she didn’t know that they existed.
Making no assumptions applies not only to patients from other cultures but also to all patients — including members of “mainstream American culture.”
Candler recalls a young patient with a new baby, who shared “how exhausted she was and how much time, energy, and work it took to care for children,” Candler recounted. “To me, it sounded as though she didn’t want another child, and I was about to offer contraception when it occurred to me to first ask if she wanted to have more children.” Candler was surprised when the patient said that, although she wasn’t actively looking to become pregnant again, she didn’t want to take preventive measures. “I’m so glad I asked, rather than simply assuming.”
Culture Is Mutable
Important questions to ask patients include whether there are aspects of their culture or religion that might affect their care — which can include medications they may feel uncomfortable using — and what family members they want to have involved in clinical discussions and decisions, said Wheat.
Lee described treating a refugee from Afghanistan who was in her sixth month of pregnancy. “I quickly needed to learn about what her expectations were for her care and my presence as a male on her care team,” he recounted. Lee arranged for the patient to receive prenatal care from a different clinician and arranged for follow-up for her husband and children. “Everyone had good results.”
Candler noted that some patients choose their physician specifically because that practitioner is conversant with their culture and respectful of its mores — especially when physicians share the same culture as the patient. But that level of familiarity can make it easy to forget to ask questions about the experience of the individual patient within that culture.
Moreover, Glass suggested, some physicians who treat patients from a particular culture or religious group may be concerned about offending them or antagonizing religious leaders if they discuss medical options that aren’t accepted or practiced in that community or culture, such as vasectomy for male contraception. “But that deprives patients of knowing what choices are available and making truly informed decisions.”
This is especially important because “culture is mutable,” said Candler, and religious or cultural practices can “look one way on paper but be implemented, adopted, or executed in a completely different way by every human being who lives in that culture.” The best cultural competency “comes from continuing to build relationships with our patients. But even in a single visit, a single hospitalization, we should get to know patients as human beings, not just members of a given culture.”
There are cultures in which families want to be the liaison between the patient and the physician and to make decisions on the patient’s behalf. “I always ask patients what role they want their family members to play even if the cultural expectation is that the family will be heavily involved,” Candler said.
Sometimes, this can be awkward, and families might become upset. Candler described an elderly, frail patient who was diagnosed with end-stage cancer. She had always relied heavily on family to care for her. Concerned about overburdening them, she didn’t want them to know her diagnosis. The patient was mentally competent to make that decision.
“Usually, I would have had the family at the bedside so I could be sure everyone was appropriately informed and prepared for what lay ahead, but in this case, I couldn’t do so,” Candler said. “I had to inform her entire care team not to discuss the cancer diagnosis with any family members because this was the patient’s express wish. And when the family asked me if the diagnosis was cancer, I had to respond, ‘I’m so sorry, but your loved one doesn’t want us to discuss details of her diagnosis.’”
Other patients don’t want to know their own diagnosis and specifically ask Candler to inform a family member. “I’ve had patients request that I tell their children. They want their children to make decisions on their behalf.”
The main thing, Candler emphasized, is to “ask the patient, make sure the patient is competent to make that decision, thoroughly document the patient’s decision in the chart, and respect whatever that decision is.”
You Can Revisit the Questions
Having a longitudinal relationship means that the physician can revisit the same questions at different junctures because people’s perspectives sometimes change over time. “Discussing what a patient wants isn’t necessarily a one-time occurrence,” Wheat said. For example, “I’ve had situations where a patient has been a member of Jehovah’s Witnesses and won’t accept blood products — like transfusions — in treatment. I tell these patients that if an emergent situation arises, I would like to have the conversation again.”
Of course, sometimes patients are seen in the emergency department or in other situations where the physician has no prior relationship with them. “I always go into a room, especially with new patients, aiming to build rapport, communicate with a high level of respect, introduce myself, explain my approach, and understand the patient’s wishes,” Lee said. “As scenarios play out, I ask in multiple ways for the patient to confirm those wishes.”
He acknowledges that this can be time-consuming, “but it helps ensure the care that patient receives is complete, thorough, comprehensive, and respectful of the patient’s values and wishes.”
Candler disclosed paid part-time clinical work at CuraCapitol Primary Care Services, volunteer advocacy (reimbursed for travel) for the American College of Physicians, volunteer advocacy (reimbursed for travel) for the American Medical Association while serving on their Task Force to Preserve the Patient-Physician Relationship, and serving as a partner representative (reimbursed for time) for the AHRQ’s Person-Centered Care Planning Partnership, representing the American College of Physicians. Lee, Wheat, and Glass disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cultural competency is one of the most important values in the practice of medicine. Defined as the “ability to collaborate effectively with individuals from different cultures,” this type of competence “improves healthcare experiences and outcomes.” But within the context of cultural familiarity, it’s equally important to “understand that each person is an individual and may or may not adhere to certain cultural beliefs or practices common in his or her culture,” according to the Agency for Healthcare Research and Quality’s (AHRQ’s) Health Literacy Universal Precautions Toolkit.
Sarah Candler, MD, MPH, an internal medicine physician specializing in primary care for older adults in Washington, DC, said that the medical code of ethics consists of several pillars, with patient autonomy as the “first and most primary of those pillars.” She calls the balance of patient autonomy and cultural respect a “complicated tightrope to walk,” but says that these ethical principles can inform medical decisions and the patient-physician relationship.
Cultural Familiarity
It’s important to be as familiar as possible with the patient’s culture, Santina Wheat, MD, program director, Northwestern McGaw Family Medicine Residency at Delnor Hospital, Geneva, told this news organization. “For example, we serve many Orthodox Jewish patients. We had a meeting with rabbis from the community to present to us what religious laws might affect our patients. Until recently, I was delivering babies, and there was always a 24-hour emergency rabbi on call if an Orthodox patient wanted the input of a rabbi into her decisions.”
Jay W. Lee, MD, MPH, a member of the board of directors of the American Academy of Family Physicians, also sets out to educate himself about the cultural norms of his patients if they come from populations he’s not familiar with. “For example, this comes up when a new refugee population comes to the United States — most recently, there was a population of Afghan refugees,” Lee told this news organization.
Lee spent “a lot of time trying to learn about their cultural norms,” which prepared him to “ask more targeted questions about the patient’s understanding of the tests we were ordering or treatment options we were bringing forward.”
Lee, also the medical director at Integrated Health Partners of Southern California and associate clinical professor of family medicine at the University of California, Irvine, said it might be best if the physician is “language congruent or culturally similar.” Lee is of Asian descent and also speaks Spanish fluently. “I enjoy cultural exchanges with my patients, and I encourage patients to find a physician who’s the best fit.” But being from the same culture isn’t absolutely necessary for building relationships with the patient. “The key is offering the patient autonomy” while understanding the cultural context.
Don’t Assume ... Always Ask
Cultural familiarity doesn’t equate with stereotyping, Wheat emphasized. “Proceeding without assumptions opens the opportunity to ask questions for clarification and understanding and to improve patient care,” said Lee.
Sara Glass, PhD, LCSW, agrees. She’s the clinical director of Soul Wellness NYC, New York City, a psychotherapy practice that specializes in treating trauma. Based on her own experiences, she knows that some physicians and other healthcare professionals confuse cultural sensitivity with cultural stereotyping.
Glass, formerly Hasidic and ultra-Orthodox, shared an example from her own life. During the delivery of her second child, she sustained a vaginal tear. At her 6-week postpartum visit, her ob/gyn said, “Just remind me when you’re in your ninth month next time, and I can sew it up right after you deliver.”
Much of this physician’s practice “consisted of Hasidic women who looked just like me, wearing the same garb — head coverings such as wigs and scarves and long skirts. Most women in that community have multiple pregnancies,” Glass told this news organization. “My sister has 10 children, and that’s not unusual. The doctor simply assumed I’d be going on to have more babies without asking if that’s what I wanted.”
Glass says she was also never given information by her physician about the range of available contraceptive options. The rabbis of the Hasidic sect to which Glass belonged allowed women to practice contraception for 6 months following childbirth, or for longer, in the setting of certain medical conditions, but only certain types of birth control were religiously permissible. Other options were not mentioned to her by her physician, and she didn’t know that they existed.
Making no assumptions applies not only to patients from other cultures but also to all patients — including members of “mainstream American culture.”
Candler recalls a young patient with a new baby, who shared “how exhausted she was and how much time, energy, and work it took to care for children,” Candler recounted. “To me, it sounded as though she didn’t want another child, and I was about to offer contraception when it occurred to me to first ask if she wanted to have more children.” Candler was surprised when the patient said that, although she wasn’t actively looking to become pregnant again, she didn’t want to take preventive measures. “I’m so glad I asked, rather than simply assuming.”
Culture Is Mutable
Important questions to ask patients include whether there are aspects of their culture or religion that might affect their care — which can include medications they may feel uncomfortable using — and what family members they want to have involved in clinical discussions and decisions, said Wheat.
Lee described treating a refugee from Afghanistan who was in her sixth month of pregnancy. “I quickly needed to learn about what her expectations were for her care and my presence as a male on her care team,” he recounted. Lee arranged for the patient to receive prenatal care from a different clinician and arranged for follow-up for her husband and children. “Everyone had good results.”
Candler noted that some patients choose their physician specifically because that practitioner is conversant with their culture and respectful of its mores — especially when physicians share the same culture as the patient. But that level of familiarity can make it easy to forget to ask questions about the experience of the individual patient within that culture.
Moreover, Glass suggested, some physicians who treat patients from a particular culture or religious group may be concerned about offending them or antagonizing religious leaders if they discuss medical options that aren’t accepted or practiced in that community or culture, such as vasectomy for male contraception. “But that deprives patients of knowing what choices are available and making truly informed decisions.”
This is especially important because “culture is mutable,” said Candler, and religious or cultural practices can “look one way on paper but be implemented, adopted, or executed in a completely different way by every human being who lives in that culture.” The best cultural competency “comes from continuing to build relationships with our patients. But even in a single visit, a single hospitalization, we should get to know patients as human beings, not just members of a given culture.”
There are cultures in which families want to be the liaison between the patient and the physician and to make decisions on the patient’s behalf. “I always ask patients what role they want their family members to play even if the cultural expectation is that the family will be heavily involved,” Candler said.
Sometimes, this can be awkward, and families might become upset. Candler described an elderly, frail patient who was diagnosed with end-stage cancer. She had always relied heavily on family to care for her. Concerned about overburdening them, she didn’t want them to know her diagnosis. The patient was mentally competent to make that decision.
“Usually, I would have had the family at the bedside so I could be sure everyone was appropriately informed and prepared for what lay ahead, but in this case, I couldn’t do so,” Candler said. “I had to inform her entire care team not to discuss the cancer diagnosis with any family members because this was the patient’s express wish. And when the family asked me if the diagnosis was cancer, I had to respond, ‘I’m so sorry, but your loved one doesn’t want us to discuss details of her diagnosis.’”
Other patients don’t want to know their own diagnosis and specifically ask Candler to inform a family member. “I’ve had patients request that I tell their children. They want their children to make decisions on their behalf.”
The main thing, Candler emphasized, is to “ask the patient, make sure the patient is competent to make that decision, thoroughly document the patient’s decision in the chart, and respect whatever that decision is.”
You Can Revisit the Questions
Having a longitudinal relationship means that the physician can revisit the same questions at different junctures because people’s perspectives sometimes change over time. “Discussing what a patient wants isn’t necessarily a one-time occurrence,” Wheat said. For example, “I’ve had situations where a patient has been a member of Jehovah’s Witnesses and won’t accept blood products — like transfusions — in treatment. I tell these patients that if an emergent situation arises, I would like to have the conversation again.”
Of course, sometimes patients are seen in the emergency department or in other situations where the physician has no prior relationship with them. “I always go into a room, especially with new patients, aiming to build rapport, communicate with a high level of respect, introduce myself, explain my approach, and understand the patient’s wishes,” Lee said. “As scenarios play out, I ask in multiple ways for the patient to confirm those wishes.”
He acknowledges that this can be time-consuming, “but it helps ensure the care that patient receives is complete, thorough, comprehensive, and respectful of the patient’s values and wishes.”
Candler disclosed paid part-time clinical work at CuraCapitol Primary Care Services, volunteer advocacy (reimbursed for travel) for the American College of Physicians, volunteer advocacy (reimbursed for travel) for the American Medical Association while serving on their Task Force to Preserve the Patient-Physician Relationship, and serving as a partner representative (reimbursed for time) for the AHRQ’s Person-Centered Care Planning Partnership, representing the American College of Physicians. Lee, Wheat, and Glass disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cultural competency is one of the most important values in the practice of medicine. Defined as the “ability to collaborate effectively with individuals from different cultures,” this type of competence “improves healthcare experiences and outcomes.” But within the context of cultural familiarity, it’s equally important to “understand that each person is an individual and may or may not adhere to certain cultural beliefs or practices common in his or her culture,” according to the Agency for Healthcare Research and Quality’s (AHRQ’s) Health Literacy Universal Precautions Toolkit.
Sarah Candler, MD, MPH, an internal medicine physician specializing in primary care for older adults in Washington, DC, said that the medical code of ethics consists of several pillars, with patient autonomy as the “first and most primary of those pillars.” She calls the balance of patient autonomy and cultural respect a “complicated tightrope to walk,” but says that these ethical principles can inform medical decisions and the patient-physician relationship.
Cultural Familiarity
It’s important to be as familiar as possible with the patient’s culture, Santina Wheat, MD, program director, Northwestern McGaw Family Medicine Residency at Delnor Hospital, Geneva, told this news organization. “For example, we serve many Orthodox Jewish patients. We had a meeting with rabbis from the community to present to us what religious laws might affect our patients. Until recently, I was delivering babies, and there was always a 24-hour emergency rabbi on call if an Orthodox patient wanted the input of a rabbi into her decisions.”
Jay W. Lee, MD, MPH, a member of the board of directors of the American Academy of Family Physicians, also sets out to educate himself about the cultural norms of his patients if they come from populations he’s not familiar with. “For example, this comes up when a new refugee population comes to the United States — most recently, there was a population of Afghan refugees,” Lee told this news organization.
Lee spent “a lot of time trying to learn about their cultural norms,” which prepared him to “ask more targeted questions about the patient’s understanding of the tests we were ordering or treatment options we were bringing forward.”
Lee, also the medical director at Integrated Health Partners of Southern California and associate clinical professor of family medicine at the University of California, Irvine, said it might be best if the physician is “language congruent or culturally similar.” Lee is of Asian descent and also speaks Spanish fluently. “I enjoy cultural exchanges with my patients, and I encourage patients to find a physician who’s the best fit.” But being from the same culture isn’t absolutely necessary for building relationships with the patient. “The key is offering the patient autonomy” while understanding the cultural context.
Don’t Assume ... Always Ask
Cultural familiarity doesn’t equate with stereotyping, Wheat emphasized. “Proceeding without assumptions opens the opportunity to ask questions for clarification and understanding and to improve patient care,” said Lee.
Sara Glass, PhD, LCSW, agrees. She’s the clinical director of Soul Wellness NYC, New York City, a psychotherapy practice that specializes in treating trauma. Based on her own experiences, she knows that some physicians and other healthcare professionals confuse cultural sensitivity with cultural stereotyping.
Glass, formerly Hasidic and ultra-Orthodox, shared an example from her own life. During the delivery of her second child, she sustained a vaginal tear. At her 6-week postpartum visit, her ob/gyn said, “Just remind me when you’re in your ninth month next time, and I can sew it up right after you deliver.”
Much of this physician’s practice “consisted of Hasidic women who looked just like me, wearing the same garb — head coverings such as wigs and scarves and long skirts. Most women in that community have multiple pregnancies,” Glass told this news organization. “My sister has 10 children, and that’s not unusual. The doctor simply assumed I’d be going on to have more babies without asking if that’s what I wanted.”
Glass says she was also never given information by her physician about the range of available contraceptive options. The rabbis of the Hasidic sect to which Glass belonged allowed women to practice contraception for 6 months following childbirth, or for longer, in the setting of certain medical conditions, but only certain types of birth control were religiously permissible. Other options were not mentioned to her by her physician, and she didn’t know that they existed.
Making no assumptions applies not only to patients from other cultures but also to all patients — including members of “mainstream American culture.”
Candler recalls a young patient with a new baby, who shared “how exhausted she was and how much time, energy, and work it took to care for children,” Candler recounted. “To me, it sounded as though she didn’t want another child, and I was about to offer contraception when it occurred to me to first ask if she wanted to have more children.” Candler was surprised when the patient said that, although she wasn’t actively looking to become pregnant again, she didn’t want to take preventive measures. “I’m so glad I asked, rather than simply assuming.”
Culture Is Mutable
Important questions to ask patients include whether there are aspects of their culture or religion that might affect their care — which can include medications they may feel uncomfortable using — and what family members they want to have involved in clinical discussions and decisions, said Wheat.
Lee described treating a refugee from Afghanistan who was in her sixth month of pregnancy. “I quickly needed to learn about what her expectations were for her care and my presence as a male on her care team,” he recounted. Lee arranged for the patient to receive prenatal care from a different clinician and arranged for follow-up for her husband and children. “Everyone had good results.”
Candler noted that some patients choose their physician specifically because that practitioner is conversant with their culture and respectful of its mores — especially when physicians share the same culture as the patient. But that level of familiarity can make it easy to forget to ask questions about the experience of the individual patient within that culture.
Moreover, Glass suggested, some physicians who treat patients from a particular culture or religious group may be concerned about offending them or antagonizing religious leaders if they discuss medical options that aren’t accepted or practiced in that community or culture, such as vasectomy for male contraception. “But that deprives patients of knowing what choices are available and making truly informed decisions.”
This is especially important because “culture is mutable,” said Candler, and religious or cultural practices can “look one way on paper but be implemented, adopted, or executed in a completely different way by every human being who lives in that culture.” The best cultural competency “comes from continuing to build relationships with our patients. But even in a single visit, a single hospitalization, we should get to know patients as human beings, not just members of a given culture.”
There are cultures in which families want to be the liaison between the patient and the physician and to make decisions on the patient’s behalf. “I always ask patients what role they want their family members to play even if the cultural expectation is that the family will be heavily involved,” Candler said.
Sometimes, this can be awkward, and families might become upset. Candler described an elderly, frail patient who was diagnosed with end-stage cancer. She had always relied heavily on family to care for her. Concerned about overburdening them, she didn’t want them to know her diagnosis. The patient was mentally competent to make that decision.
“Usually, I would have had the family at the bedside so I could be sure everyone was appropriately informed and prepared for what lay ahead, but in this case, I couldn’t do so,” Candler said. “I had to inform her entire care team not to discuss the cancer diagnosis with any family members because this was the patient’s express wish. And when the family asked me if the diagnosis was cancer, I had to respond, ‘I’m so sorry, but your loved one doesn’t want us to discuss details of her diagnosis.’”
Other patients don’t want to know their own diagnosis and specifically ask Candler to inform a family member. “I’ve had patients request that I tell their children. They want their children to make decisions on their behalf.”
The main thing, Candler emphasized, is to “ask the patient, make sure the patient is competent to make that decision, thoroughly document the patient’s decision in the chart, and respect whatever that decision is.”
You Can Revisit the Questions
Having a longitudinal relationship means that the physician can revisit the same questions at different junctures because people’s perspectives sometimes change over time. “Discussing what a patient wants isn’t necessarily a one-time occurrence,” Wheat said. For example, “I’ve had situations where a patient has been a member of Jehovah’s Witnesses and won’t accept blood products — like transfusions — in treatment. I tell these patients that if an emergent situation arises, I would like to have the conversation again.”
Of course, sometimes patients are seen in the emergency department or in other situations where the physician has no prior relationship with them. “I always go into a room, especially with new patients, aiming to build rapport, communicate with a high level of respect, introduce myself, explain my approach, and understand the patient’s wishes,” Lee said. “As scenarios play out, I ask in multiple ways for the patient to confirm those wishes.”
He acknowledges that this can be time-consuming, “but it helps ensure the care that patient receives is complete, thorough, comprehensive, and respectful of the patient’s values and wishes.”
Candler disclosed paid part-time clinical work at CuraCapitol Primary Care Services, volunteer advocacy (reimbursed for travel) for the American College of Physicians, volunteer advocacy (reimbursed for travel) for the American Medical Association while serving on their Task Force to Preserve the Patient-Physician Relationship, and serving as a partner representative (reimbursed for time) for the AHRQ’s Person-Centered Care Planning Partnership, representing the American College of Physicians. Lee, Wheat, and Glass disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Strategies to Manage Metabolic Health During the Holidays
Interventions during holidays and school vacations can help prevent children and adults gaining weight, according to a recent systematic review and meta-analysis published in Obesity Reviews.
Evidence suggests that certain times of the year, such as the Christmas holidays and summer vacations, are associated with weight gain. In adults, up to 50% of the total annual weight gain occurs during December.
In 2023, the United Nations Children’s Fund reported that more than four million children younger than 5 years and nearly 50 million children and adolescents aged 5-19 years in Latin America and the Caribbean were affected by overweight. Among adults, more than 50% of individuals in every country in the region live with obesity.
These alarming figures call for urgent action from governments, healthcare professionals, and multidisciplinary teams to implement prevention strategies and promote further research.
Study Significance
Michelle Maree Haby de Sosa, PhD, an epidemiologist and researcher at the Department of Chemical-Biological and Nutritional Sciences at the University of Sonora, Hermosillo, Mexico, led the study. She explained that the research team first conducted a narrative review on weight gain during the festive season. “We found that the 6 weeks between December and mid-January represent a critical period when people gain half the weight they put on all year. This highlights the importance of addressing obesity and overweight by promoting lifestyle changes and prevention strategies to tackle this public health issue.”
The researchers then conducted a systematic review of global interventions not only to publish findings but also to educate healthcare professionals and stakeholders. They searched databases such as Medline, EMBASE, PsycINFO, SciELO, LILACS, and Cochrane, focusing on randomized controlled trials. These were supplemented with gray literature and references from relevant articles, as well as additional data requested from study authors.
Key Findings
The review included studies from the United States (10), the United Kingdom (one), and Chile (one). Of these, two had a low risk for bias, two moderate, seven high, and one critical.
Most interventions targeted school-aged children or adults. According to Haby de Sosa, achieving consistent results in adolescents was challenging due to the difficulty of changing behaviors in this age group. In contrast, interventions for school-aged children were implemented primarily during day camp visits, where participants were divided into control and intervention groups.
The interventions included nutrition classes, physical activity, and the provision of healthy meals, which resulted in less weight gain compared with control groups.
In children, the meta-analysis of four of seven studies conducted during summer vacations (six interventions) found a small but significant reduction in body mass index z-scores in the intervention group (−0.06; 95% CI, −0.10 to −0.01; P = .01; I² = 0%; very low-certainty evidence).
Among adults, interventions also generally proved effective, despite variations in implementation. A meta-analysis of five studies involving 462 participants (234 intervention, 228 control) showed a slight reduction in body weight (−0.99 kg; 95% CI, −2.15 to 0.18; P = 0.10; I² = 89%).
Three key intervention areas were identified: Nutrition, physical activity, and psychological support including behavioral and cognitive elements. Strict diets were generally not a priority; instead, participants were advised to reduce consumption of high-calorie food and sugary beverages while increasing their intake of vegetables.
Promising Interventions
The study highlighted specific interventions for children and adults:
- Children: 6- to 8-week summer camps with daily physical activities such as sports and crafts, complemented by free, nutritious meals.
- Adults: Daily weight monitoring paired with nutrition counseling based on social cognitive theory. Interventions lasted 4 to 8 weeks, spanning mid-November to early January.
Expert Recommendations
Carlos Cristi-Montero, PhD, a researcher at Pontificia Universidad Católica de Valparaíso, Chile, and an author of a Chilean intervention study, shared insights with this news organization.
He emphasized the importance of portion control for children. “During the holidays, families prepare calorie-rich dishes but often fail to consider portion sizes,” he noted. “Children are treated like adults, which contributes to excessive caloric intake. Our interventions focused on teaching people about portion control, the caloric content of their meals, and the risks of overweight and obesity, as well as the benefits of healthy eating.”
He also stressed the importance of evaluating not just weight but body composition, using tools like dual-energy x-ray absorptiometry to measure fat and muscle mass.
Cristi-Montero also highlighted the importance of physical activity: “We emphasize the value of exercise and staying active as key strategies to prevent weight gain.”
Steps for Successful Interventions
Educating teachers and parents to reinforce healthy behaviors is also vital, according to Cristi-Montero, as obesity impacts not only metabolic health but also academic performance and mental health.
Both Haby de Sosa and Cristi-Montero agreed that primary care professionals have an important role in driving effective interventions, alongside participation in research to refine prevention strategies. Multidisciplinary teams — including nutritionists, psychologists, exercise specialists, teachers, and parents — can play a part in preventing weight gain during holidays.
Future Directions
The University of Sonora research team is currently conducting a controlled trial in Hermosillo, Mexico, involving adult participants divided into intervention and control groups. Preliminary results, already published online, highlight the effectiveness of strategies such as nutrition education, physical activity, regular weight goals, and psychological support in promoting habit changes.
“Interventions to prevent weight gain during the holidays and summer vacations are necessary,” the authors concluded, emphasizing the need for further research to evaluate their effectiveness in the region.
Haby de Sosa or Cristi-Montero declared no relevant financial conflicts of interest.
Natalia Martínez Medina, disclosed the following: Consultant or advisor for: AstraZeneca (former); Sanofi (former). Speaker or a member of a speaker’s bureau for: AstraZeneca (former); Sanofi (former). Research funding from: AstraZeneca (former); Sanofi (former). Contracted researcher for: AstraZeneca (former); Sanofi (former). Employee of: AstraZeneca (former); Sanofi (former).
This story was translated from Medscape’s Spanish edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Interventions during holidays and school vacations can help prevent children and adults gaining weight, according to a recent systematic review and meta-analysis published in Obesity Reviews.
Evidence suggests that certain times of the year, such as the Christmas holidays and summer vacations, are associated with weight gain. In adults, up to 50% of the total annual weight gain occurs during December.
In 2023, the United Nations Children’s Fund reported that more than four million children younger than 5 years and nearly 50 million children and adolescents aged 5-19 years in Latin America and the Caribbean were affected by overweight. Among adults, more than 50% of individuals in every country in the region live with obesity.
These alarming figures call for urgent action from governments, healthcare professionals, and multidisciplinary teams to implement prevention strategies and promote further research.
Study Significance
Michelle Maree Haby de Sosa, PhD, an epidemiologist and researcher at the Department of Chemical-Biological and Nutritional Sciences at the University of Sonora, Hermosillo, Mexico, led the study. She explained that the research team first conducted a narrative review on weight gain during the festive season. “We found that the 6 weeks between December and mid-January represent a critical period when people gain half the weight they put on all year. This highlights the importance of addressing obesity and overweight by promoting lifestyle changes and prevention strategies to tackle this public health issue.”
The researchers then conducted a systematic review of global interventions not only to publish findings but also to educate healthcare professionals and stakeholders. They searched databases such as Medline, EMBASE, PsycINFO, SciELO, LILACS, and Cochrane, focusing on randomized controlled trials. These were supplemented with gray literature and references from relevant articles, as well as additional data requested from study authors.
Key Findings
The review included studies from the United States (10), the United Kingdom (one), and Chile (one). Of these, two had a low risk for bias, two moderate, seven high, and one critical.
Most interventions targeted school-aged children or adults. According to Haby de Sosa, achieving consistent results in adolescents was challenging due to the difficulty of changing behaviors in this age group. In contrast, interventions for school-aged children were implemented primarily during day camp visits, where participants were divided into control and intervention groups.
The interventions included nutrition classes, physical activity, and the provision of healthy meals, which resulted in less weight gain compared with control groups.
In children, the meta-analysis of four of seven studies conducted during summer vacations (six interventions) found a small but significant reduction in body mass index z-scores in the intervention group (−0.06; 95% CI, −0.10 to −0.01; P = .01; I² = 0%; very low-certainty evidence).
Among adults, interventions also generally proved effective, despite variations in implementation. A meta-analysis of five studies involving 462 participants (234 intervention, 228 control) showed a slight reduction in body weight (−0.99 kg; 95% CI, −2.15 to 0.18; P = 0.10; I² = 89%).
Three key intervention areas were identified: Nutrition, physical activity, and psychological support including behavioral and cognitive elements. Strict diets were generally not a priority; instead, participants were advised to reduce consumption of high-calorie food and sugary beverages while increasing their intake of vegetables.
Promising Interventions
The study highlighted specific interventions for children and adults:
- Children: 6- to 8-week summer camps with daily physical activities such as sports and crafts, complemented by free, nutritious meals.
- Adults: Daily weight monitoring paired with nutrition counseling based on social cognitive theory. Interventions lasted 4 to 8 weeks, spanning mid-November to early January.
Expert Recommendations
Carlos Cristi-Montero, PhD, a researcher at Pontificia Universidad Católica de Valparaíso, Chile, and an author of a Chilean intervention study, shared insights with this news organization.
He emphasized the importance of portion control for children. “During the holidays, families prepare calorie-rich dishes but often fail to consider portion sizes,” he noted. “Children are treated like adults, which contributes to excessive caloric intake. Our interventions focused on teaching people about portion control, the caloric content of their meals, and the risks of overweight and obesity, as well as the benefits of healthy eating.”
He also stressed the importance of evaluating not just weight but body composition, using tools like dual-energy x-ray absorptiometry to measure fat and muscle mass.
Cristi-Montero also highlighted the importance of physical activity: “We emphasize the value of exercise and staying active as key strategies to prevent weight gain.”
Steps for Successful Interventions
Educating teachers and parents to reinforce healthy behaviors is also vital, according to Cristi-Montero, as obesity impacts not only metabolic health but also academic performance and mental health.
Both Haby de Sosa and Cristi-Montero agreed that primary care professionals have an important role in driving effective interventions, alongside participation in research to refine prevention strategies. Multidisciplinary teams — including nutritionists, psychologists, exercise specialists, teachers, and parents — can play a part in preventing weight gain during holidays.
Future Directions
The University of Sonora research team is currently conducting a controlled trial in Hermosillo, Mexico, involving adult participants divided into intervention and control groups. Preliminary results, already published online, highlight the effectiveness of strategies such as nutrition education, physical activity, regular weight goals, and psychological support in promoting habit changes.
“Interventions to prevent weight gain during the holidays and summer vacations are necessary,” the authors concluded, emphasizing the need for further research to evaluate their effectiveness in the region.
Haby de Sosa or Cristi-Montero declared no relevant financial conflicts of interest.
Natalia Martínez Medina, disclosed the following: Consultant or advisor for: AstraZeneca (former); Sanofi (former). Speaker or a member of a speaker’s bureau for: AstraZeneca (former); Sanofi (former). Research funding from: AstraZeneca (former); Sanofi (former). Contracted researcher for: AstraZeneca (former); Sanofi (former). Employee of: AstraZeneca (former); Sanofi (former).
This story was translated from Medscape’s Spanish edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Interventions during holidays and school vacations can help prevent children and adults gaining weight, according to a recent systematic review and meta-analysis published in Obesity Reviews.
Evidence suggests that certain times of the year, such as the Christmas holidays and summer vacations, are associated with weight gain. In adults, up to 50% of the total annual weight gain occurs during December.
In 2023, the United Nations Children’s Fund reported that more than four million children younger than 5 years and nearly 50 million children and adolescents aged 5-19 years in Latin America and the Caribbean were affected by overweight. Among adults, more than 50% of individuals in every country in the region live with obesity.
These alarming figures call for urgent action from governments, healthcare professionals, and multidisciplinary teams to implement prevention strategies and promote further research.
Study Significance
Michelle Maree Haby de Sosa, PhD, an epidemiologist and researcher at the Department of Chemical-Biological and Nutritional Sciences at the University of Sonora, Hermosillo, Mexico, led the study. She explained that the research team first conducted a narrative review on weight gain during the festive season. “We found that the 6 weeks between December and mid-January represent a critical period when people gain half the weight they put on all year. This highlights the importance of addressing obesity and overweight by promoting lifestyle changes and prevention strategies to tackle this public health issue.”
The researchers then conducted a systematic review of global interventions not only to publish findings but also to educate healthcare professionals and stakeholders. They searched databases such as Medline, EMBASE, PsycINFO, SciELO, LILACS, and Cochrane, focusing on randomized controlled trials. These were supplemented with gray literature and references from relevant articles, as well as additional data requested from study authors.
Key Findings
The review included studies from the United States (10), the United Kingdom (one), and Chile (one). Of these, two had a low risk for bias, two moderate, seven high, and one critical.
Most interventions targeted school-aged children or adults. According to Haby de Sosa, achieving consistent results in adolescents was challenging due to the difficulty of changing behaviors in this age group. In contrast, interventions for school-aged children were implemented primarily during day camp visits, where participants were divided into control and intervention groups.
The interventions included nutrition classes, physical activity, and the provision of healthy meals, which resulted in less weight gain compared with control groups.
In children, the meta-analysis of four of seven studies conducted during summer vacations (six interventions) found a small but significant reduction in body mass index z-scores in the intervention group (−0.06; 95% CI, −0.10 to −0.01; P = .01; I² = 0%; very low-certainty evidence).
Among adults, interventions also generally proved effective, despite variations in implementation. A meta-analysis of five studies involving 462 participants (234 intervention, 228 control) showed a slight reduction in body weight (−0.99 kg; 95% CI, −2.15 to 0.18; P = 0.10; I² = 89%).
Three key intervention areas were identified: Nutrition, physical activity, and psychological support including behavioral and cognitive elements. Strict diets were generally not a priority; instead, participants were advised to reduce consumption of high-calorie food and sugary beverages while increasing their intake of vegetables.
Promising Interventions
The study highlighted specific interventions for children and adults:
- Children: 6- to 8-week summer camps with daily physical activities such as sports and crafts, complemented by free, nutritious meals.
- Adults: Daily weight monitoring paired with nutrition counseling based on social cognitive theory. Interventions lasted 4 to 8 weeks, spanning mid-November to early January.
Expert Recommendations
Carlos Cristi-Montero, PhD, a researcher at Pontificia Universidad Católica de Valparaíso, Chile, and an author of a Chilean intervention study, shared insights with this news organization.
He emphasized the importance of portion control for children. “During the holidays, families prepare calorie-rich dishes but often fail to consider portion sizes,” he noted. “Children are treated like adults, which contributes to excessive caloric intake. Our interventions focused on teaching people about portion control, the caloric content of their meals, and the risks of overweight and obesity, as well as the benefits of healthy eating.”
He also stressed the importance of evaluating not just weight but body composition, using tools like dual-energy x-ray absorptiometry to measure fat and muscle mass.
Cristi-Montero also highlighted the importance of physical activity: “We emphasize the value of exercise and staying active as key strategies to prevent weight gain.”
Steps for Successful Interventions
Educating teachers and parents to reinforce healthy behaviors is also vital, according to Cristi-Montero, as obesity impacts not only metabolic health but also academic performance and mental health.
Both Haby de Sosa and Cristi-Montero agreed that primary care professionals have an important role in driving effective interventions, alongside participation in research to refine prevention strategies. Multidisciplinary teams — including nutritionists, psychologists, exercise specialists, teachers, and parents — can play a part in preventing weight gain during holidays.
Future Directions
The University of Sonora research team is currently conducting a controlled trial in Hermosillo, Mexico, involving adult participants divided into intervention and control groups. Preliminary results, already published online, highlight the effectiveness of strategies such as nutrition education, physical activity, regular weight goals, and psychological support in promoting habit changes.
“Interventions to prevent weight gain during the holidays and summer vacations are necessary,” the authors concluded, emphasizing the need for further research to evaluate their effectiveness in the region.
Haby de Sosa or Cristi-Montero declared no relevant financial conflicts of interest.
Natalia Martínez Medina, disclosed the following: Consultant or advisor for: AstraZeneca (former); Sanofi (former). Speaker or a member of a speaker’s bureau for: AstraZeneca (former); Sanofi (former). Research funding from: AstraZeneca (former); Sanofi (former). Contracted researcher for: AstraZeneca (former); Sanofi (former). Employee of: AstraZeneca (former); Sanofi (former).
This story was translated from Medscape’s Spanish edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Allergic Contact Dermatitis: New Culprits
New allergens responsible for contact dermatitis emerge regularly. During the Dermatology Days of Paris 2024 conference, Angèle Soria, MD, PhD, a dermatologist at Tenon Hospital in Paris, France, outlined four major categories driving this trend. Among them are (meth)acrylates found in nail cosmetics used in salons or do-it-yourself false nail kits that can be bought online.
Isothiazolinones
a preservative used in many cosmetics; (meth)acrylates; essential oils; and epoxy resins used in industry and leisure activities.
Around 15 years ago, parabens, commonly used as preservatives in cosmetics, were identified as endocrine disruptors. In response, they were largely replaced by newer preservatives, notably MI. However, this led to a proliferation of allergic contact dermatitis in Europe between 2010 and 2013.
“About 10% of the population that we tested showed allergies to these preservatives, primarily found in cosmetics,” explained Soria. Since 2015, the use of MI in leave-on cosmetics has been prohibited in Europe and its concentration restricted in rinse-off products. However, cosmetics sold online from outside Europe may not comply with these regulations.
MI is also present in water-based paints to prevent mold. “A few years ago, we started seeing patients with facial angioedema, sometimes combined with asthma, caused by these isothiazolinone preservatives, including in patients who are not professional painters,” said Soria. More recently, attention has shifted to MI’s presence in household cleaning products. A 2020 Spanish study found MI in 76% of 34 analyzed cleaning products.
MI-based fungicides are also used to treat leather during transport, which can lead to contact allergies among professionals and consumers alike. Additionally, MI has been identified in children’s toys, including slime gels, and in florists’ gel cubes used to preserve flowers.
“We are therefore surrounded by these preservatives, which are no longer only in cosmetics,” warned the dermatologist.
(Meth)acrylates
Another major allergen category is (meth)acrylates, responsible for many cases of allergic contact dermatitis. Acrylates and their derivatives are widely used in everyday items. They are low–molecular weight monomers, sensitizing on contact with the skin. Their polymerized forms include materials like Plexiglas.
“We are currently witnessing an epidemic of contact dermatitis in the general population, mainly due to nail cosmetics, such as semipermanent nail polishes and at-home false nail kits,” reported Soria. Nail cosmetics account for 97% of new sensitization cases involving (meth)acrylates. These allergens often cause severe dermatitis, prompting the European Union to mandate labeling in 2020, warning that these products are “for professional use only” and can “cause allergic reactions.”
Beyond nail cosmetics, these allergens are also found in dental products (such as trays), ECG electrodes, prosthetics, glucose sensors, surgical adhesives, and some electronic devices like earbuds and phone screens. Notably, patients sensitized to acrylates via nail kits may experience reactions during dental treatments involving acrylates.
Investigating Essential Oil Use
Essential oils, distinct from vegetable oils like almond or argan, are another known allergen. Often considered risk-free due to their “natural” label, these products are widely used topically, orally, or via inhalation for various purposes, such as treating respiratory infections or creating relaxing atmospheres. However, essential oils contain fragrant molecules like terpenes, which can become highly allergenic over time, especially after repeated exposure.
Soria emphasized the importance of asking patients about their use of essential oils, especially tea tree and lavender oils, which are commonly used but rarely mentioned by patients unless prompted.
Epoxy Resins in Recreational Use
Epoxy resins are a growing cause of contact allergies, not just in professional settings such as aeronautics and construction work but also increasingly in recreational activities. Soria highlighted the case of a 12-year-old girl hospitalized for severe facial edema after engaging in resin crafts inspired by TikTok. For 6 months, she had been creating resin objects, such as bowls and cutting boards, using vinyl gloves and a Filtering FacePiece 2 mask under adult supervision.
“The growing popularity and online availability of epoxy resins mean that allergic reactions should now be considered even in nonprofessional contexts,” warned Soria.
Clinical Approach
When dermatologists suspect allergic contact dermatitis, the first step is to treat the condition with corticosteroid creams. This is followed by a detailed patient interview to identify suspected allergens in products they’ve used.
Patch testing is then conducted to confirm the allergen. Small chambers containing potential allergens are applied to the upper back for 48 hours without removal. Results are read 2-5 days later, with some cases requiring a 7-day follow-up.
The patient’s occupation is an important factor, as certain professions, such as hairdressing, healthcare, or beauty therapy, are known to trigger allergic contact dermatitis. Similarly, certain hobbies may also play a role.
A thorough approach ensures accurate diagnosis and targeted prevention strategies.
This story was translated from Medscape’s French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
New allergens responsible for contact dermatitis emerge regularly. During the Dermatology Days of Paris 2024 conference, Angèle Soria, MD, PhD, a dermatologist at Tenon Hospital in Paris, France, outlined four major categories driving this trend. Among them are (meth)acrylates found in nail cosmetics used in salons or do-it-yourself false nail kits that can be bought online.
Isothiazolinones
a preservative used in many cosmetics; (meth)acrylates; essential oils; and epoxy resins used in industry and leisure activities.
Around 15 years ago, parabens, commonly used as preservatives in cosmetics, were identified as endocrine disruptors. In response, they were largely replaced by newer preservatives, notably MI. However, this led to a proliferation of allergic contact dermatitis in Europe between 2010 and 2013.
“About 10% of the population that we tested showed allergies to these preservatives, primarily found in cosmetics,” explained Soria. Since 2015, the use of MI in leave-on cosmetics has been prohibited in Europe and its concentration restricted in rinse-off products. However, cosmetics sold online from outside Europe may not comply with these regulations.
MI is also present in water-based paints to prevent mold. “A few years ago, we started seeing patients with facial angioedema, sometimes combined with asthma, caused by these isothiazolinone preservatives, including in patients who are not professional painters,” said Soria. More recently, attention has shifted to MI’s presence in household cleaning products. A 2020 Spanish study found MI in 76% of 34 analyzed cleaning products.
MI-based fungicides are also used to treat leather during transport, which can lead to contact allergies among professionals and consumers alike. Additionally, MI has been identified in children’s toys, including slime gels, and in florists’ gel cubes used to preserve flowers.
“We are therefore surrounded by these preservatives, which are no longer only in cosmetics,” warned the dermatologist.
(Meth)acrylates
Another major allergen category is (meth)acrylates, responsible for many cases of allergic contact dermatitis. Acrylates and their derivatives are widely used in everyday items. They are low–molecular weight monomers, sensitizing on contact with the skin. Their polymerized forms include materials like Plexiglas.
“We are currently witnessing an epidemic of contact dermatitis in the general population, mainly due to nail cosmetics, such as semipermanent nail polishes and at-home false nail kits,” reported Soria. Nail cosmetics account for 97% of new sensitization cases involving (meth)acrylates. These allergens often cause severe dermatitis, prompting the European Union to mandate labeling in 2020, warning that these products are “for professional use only” and can “cause allergic reactions.”
Beyond nail cosmetics, these allergens are also found in dental products (such as trays), ECG electrodes, prosthetics, glucose sensors, surgical adhesives, and some electronic devices like earbuds and phone screens. Notably, patients sensitized to acrylates via nail kits may experience reactions during dental treatments involving acrylates.
Investigating Essential Oil Use
Essential oils, distinct from vegetable oils like almond or argan, are another known allergen. Often considered risk-free due to their “natural” label, these products are widely used topically, orally, or via inhalation for various purposes, such as treating respiratory infections or creating relaxing atmospheres. However, essential oils contain fragrant molecules like terpenes, which can become highly allergenic over time, especially after repeated exposure.
Soria emphasized the importance of asking patients about their use of essential oils, especially tea tree and lavender oils, which are commonly used but rarely mentioned by patients unless prompted.
Epoxy Resins in Recreational Use
Epoxy resins are a growing cause of contact allergies, not just in professional settings such as aeronautics and construction work but also increasingly in recreational activities. Soria highlighted the case of a 12-year-old girl hospitalized for severe facial edema after engaging in resin crafts inspired by TikTok. For 6 months, she had been creating resin objects, such as bowls and cutting boards, using vinyl gloves and a Filtering FacePiece 2 mask under adult supervision.
“The growing popularity and online availability of epoxy resins mean that allergic reactions should now be considered even in nonprofessional contexts,” warned Soria.
Clinical Approach
When dermatologists suspect allergic contact dermatitis, the first step is to treat the condition with corticosteroid creams. This is followed by a detailed patient interview to identify suspected allergens in products they’ve used.
Patch testing is then conducted to confirm the allergen. Small chambers containing potential allergens are applied to the upper back for 48 hours without removal. Results are read 2-5 days later, with some cases requiring a 7-day follow-up.
The patient’s occupation is an important factor, as certain professions, such as hairdressing, healthcare, or beauty therapy, are known to trigger allergic contact dermatitis. Similarly, certain hobbies may also play a role.
A thorough approach ensures accurate diagnosis and targeted prevention strategies.
This story was translated from Medscape’s French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
New allergens responsible for contact dermatitis emerge regularly. During the Dermatology Days of Paris 2024 conference, Angèle Soria, MD, PhD, a dermatologist at Tenon Hospital in Paris, France, outlined four major categories driving this trend. Among them are (meth)acrylates found in nail cosmetics used in salons or do-it-yourself false nail kits that can be bought online.
Isothiazolinones
a preservative used in many cosmetics; (meth)acrylates; essential oils; and epoxy resins used in industry and leisure activities.
Around 15 years ago, parabens, commonly used as preservatives in cosmetics, were identified as endocrine disruptors. In response, they were largely replaced by newer preservatives, notably MI. However, this led to a proliferation of allergic contact dermatitis in Europe between 2010 and 2013.
“About 10% of the population that we tested showed allergies to these preservatives, primarily found in cosmetics,” explained Soria. Since 2015, the use of MI in leave-on cosmetics has been prohibited in Europe and its concentration restricted in rinse-off products. However, cosmetics sold online from outside Europe may not comply with these regulations.
MI is also present in water-based paints to prevent mold. “A few years ago, we started seeing patients with facial angioedema, sometimes combined with asthma, caused by these isothiazolinone preservatives, including in patients who are not professional painters,” said Soria. More recently, attention has shifted to MI’s presence in household cleaning products. A 2020 Spanish study found MI in 76% of 34 analyzed cleaning products.
MI-based fungicides are also used to treat leather during transport, which can lead to contact allergies among professionals and consumers alike. Additionally, MI has been identified in children’s toys, including slime gels, and in florists’ gel cubes used to preserve flowers.
“We are therefore surrounded by these preservatives, which are no longer only in cosmetics,” warned the dermatologist.
(Meth)acrylates
Another major allergen category is (meth)acrylates, responsible for many cases of allergic contact dermatitis. Acrylates and their derivatives are widely used in everyday items. They are low–molecular weight monomers, sensitizing on contact with the skin. Their polymerized forms include materials like Plexiglas.
“We are currently witnessing an epidemic of contact dermatitis in the general population, mainly due to nail cosmetics, such as semipermanent nail polishes and at-home false nail kits,” reported Soria. Nail cosmetics account for 97% of new sensitization cases involving (meth)acrylates. These allergens often cause severe dermatitis, prompting the European Union to mandate labeling in 2020, warning that these products are “for professional use only” and can “cause allergic reactions.”
Beyond nail cosmetics, these allergens are also found in dental products (such as trays), ECG electrodes, prosthetics, glucose sensors, surgical adhesives, and some electronic devices like earbuds and phone screens. Notably, patients sensitized to acrylates via nail kits may experience reactions during dental treatments involving acrylates.
Investigating Essential Oil Use
Essential oils, distinct from vegetable oils like almond or argan, are another known allergen. Often considered risk-free due to their “natural” label, these products are widely used topically, orally, or via inhalation for various purposes, such as treating respiratory infections or creating relaxing atmospheres. However, essential oils contain fragrant molecules like terpenes, which can become highly allergenic over time, especially after repeated exposure.
Soria emphasized the importance of asking patients about their use of essential oils, especially tea tree and lavender oils, which are commonly used but rarely mentioned by patients unless prompted.
Epoxy Resins in Recreational Use
Epoxy resins are a growing cause of contact allergies, not just in professional settings such as aeronautics and construction work but also increasingly in recreational activities. Soria highlighted the case of a 12-year-old girl hospitalized for severe facial edema after engaging in resin crafts inspired by TikTok. For 6 months, she had been creating resin objects, such as bowls and cutting boards, using vinyl gloves and a Filtering FacePiece 2 mask under adult supervision.
“The growing popularity and online availability of epoxy resins mean that allergic reactions should now be considered even in nonprofessional contexts,” warned Soria.
Clinical Approach
When dermatologists suspect allergic contact dermatitis, the first step is to treat the condition with corticosteroid creams. This is followed by a detailed patient interview to identify suspected allergens in products they’ve used.
Patch testing is then conducted to confirm the allergen. Small chambers containing potential allergens are applied to the upper back for 48 hours without removal. Results are read 2-5 days later, with some cases requiring a 7-day follow-up.
The patient’s occupation is an important factor, as certain professions, such as hairdressing, healthcare, or beauty therapy, are known to trigger allergic contact dermatitis. Similarly, certain hobbies may also play a role.
A thorough approach ensures accurate diagnosis and targeted prevention strategies.
This story was translated from Medscape’s French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Skin Cancer Risk Elevated Among Blood, Marrow Transplant Survivors
TOPLINE:
with a cumulative incidence of 27.4% over 30 years, according to the results of a cohort study.
METHODOLOGY:
- The retrospective cohort study included 3880 BMT survivors (median age, 44 years; 55.8% men; 4.9% Black, 12.1 Hispanic, and 74.7% non-Hispanic White individuals) who underwent transplant between 1974 to 2014.
- Participants completed the BMT Survivor Study survey and were followed up for a median of 9.5 years.
- The primary outcomes were the development of subsequent cutaneous malignant neoplasms (BCC, SCC, or melanoma).
TAKEAWAY:
- The 30-year cumulative incidence of any cutaneous malignant neoplasm was 27.4% — 18% for BCC, 9.8% for SCC, and 3.7% for melanoma.
- A higher risk for skin cancer was reported for patients aged 50 years or more (subdistribution hazard ratio [SHR], 2.23; 95% CI, 1.83-2.71), and men (SHR, 1.40; 95% CI, 1.18-1.65).
- Allogeneic BMT with chronic graft-vs-host disease (cGVHD) increased the risk for skin cancer (SHR, 1.84; 95% CI, 1.37-2.47), compared with autologous BMT, while post-BMT immunosuppression increased risk for all types (overall SHR, 1.53; 95% CI, 1.26-1.86).
- The risk for any skin cancer was significantly lower in Black individuals (SHR, 0.14; 95% CI, 0.05-0.37), Hispanic individuals (SHR, 0.29; 95%CI, 0.20-0.62), and patients of other races or who were multiracial (SHR, 0.22; 95% CI, 0.13-0.37) than in non-Hispanic White patients.
IN PRACTICE:
In the study, “risk factors for post-BMT cutaneous malignant neoplasms included pretransplant treatment with a monoclonal antibody, cGVHD, and posttransplant immunosuppression,” the authors wrote, adding that the findings “could inform targeted surveillance of BMT survivors.” Most BMT survivors, “do not undergo routine dermatologic surveillance, highlighting the need to understand risk factors and incorporate risk-informed dermatologic surveillance into survivorship care plans.”
SOURCE:
The study was led by Kristy K. Broman, MD, MPH, University of Alabama at Birmingham, and was published online on December 18 in JAMA Dermatology.
LIMITATIONS:
Limitations included self-reported data and possible underreporting of melanoma cases in the SEER database. Additionally, the study did not capture other risk factors for cutaneous malignant neoplasms such as skin phototype, ultraviolet light exposure, or family history. The duration of posttransplant immunosuppression was not collected, and surveys were administered at variable intervals, though all were completed more than 2 years post BMT.
DISCLOSURES:
The study was supported by the National Cancer Institute (NCI) and the Leukemia and Lymphoma Society. Broman received grants from NCI, the National Center for Advancing Translational Sciences, the American Society of Clinical Oncology, and the American College of Surgeons. Another author reported receiving grants outside this work.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
with a cumulative incidence of 27.4% over 30 years, according to the results of a cohort study.
METHODOLOGY:
- The retrospective cohort study included 3880 BMT survivors (median age, 44 years; 55.8% men; 4.9% Black, 12.1 Hispanic, and 74.7% non-Hispanic White individuals) who underwent transplant between 1974 to 2014.
- Participants completed the BMT Survivor Study survey and were followed up for a median of 9.5 years.
- The primary outcomes were the development of subsequent cutaneous malignant neoplasms (BCC, SCC, or melanoma).
TAKEAWAY:
- The 30-year cumulative incidence of any cutaneous malignant neoplasm was 27.4% — 18% for BCC, 9.8% for SCC, and 3.7% for melanoma.
- A higher risk for skin cancer was reported for patients aged 50 years or more (subdistribution hazard ratio [SHR], 2.23; 95% CI, 1.83-2.71), and men (SHR, 1.40; 95% CI, 1.18-1.65).
- Allogeneic BMT with chronic graft-vs-host disease (cGVHD) increased the risk for skin cancer (SHR, 1.84; 95% CI, 1.37-2.47), compared with autologous BMT, while post-BMT immunosuppression increased risk for all types (overall SHR, 1.53; 95% CI, 1.26-1.86).
- The risk for any skin cancer was significantly lower in Black individuals (SHR, 0.14; 95% CI, 0.05-0.37), Hispanic individuals (SHR, 0.29; 95%CI, 0.20-0.62), and patients of other races or who were multiracial (SHR, 0.22; 95% CI, 0.13-0.37) than in non-Hispanic White patients.
IN PRACTICE:
In the study, “risk factors for post-BMT cutaneous malignant neoplasms included pretransplant treatment with a monoclonal antibody, cGVHD, and posttransplant immunosuppression,” the authors wrote, adding that the findings “could inform targeted surveillance of BMT survivors.” Most BMT survivors, “do not undergo routine dermatologic surveillance, highlighting the need to understand risk factors and incorporate risk-informed dermatologic surveillance into survivorship care plans.”
SOURCE:
The study was led by Kristy K. Broman, MD, MPH, University of Alabama at Birmingham, and was published online on December 18 in JAMA Dermatology.
LIMITATIONS:
Limitations included self-reported data and possible underreporting of melanoma cases in the SEER database. Additionally, the study did not capture other risk factors for cutaneous malignant neoplasms such as skin phototype, ultraviolet light exposure, or family history. The duration of posttransplant immunosuppression was not collected, and surveys were administered at variable intervals, though all were completed more than 2 years post BMT.
DISCLOSURES:
The study was supported by the National Cancer Institute (NCI) and the Leukemia and Lymphoma Society. Broman received grants from NCI, the National Center for Advancing Translational Sciences, the American Society of Clinical Oncology, and the American College of Surgeons. Another author reported receiving grants outside this work.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
with a cumulative incidence of 27.4% over 30 years, according to the results of a cohort study.
METHODOLOGY:
- The retrospective cohort study included 3880 BMT survivors (median age, 44 years; 55.8% men; 4.9% Black, 12.1 Hispanic, and 74.7% non-Hispanic White individuals) who underwent transplant between 1974 to 2014.
- Participants completed the BMT Survivor Study survey and were followed up for a median of 9.5 years.
- The primary outcomes were the development of subsequent cutaneous malignant neoplasms (BCC, SCC, or melanoma).
TAKEAWAY:
- The 30-year cumulative incidence of any cutaneous malignant neoplasm was 27.4% — 18% for BCC, 9.8% for SCC, and 3.7% for melanoma.
- A higher risk for skin cancer was reported for patients aged 50 years or more (subdistribution hazard ratio [SHR], 2.23; 95% CI, 1.83-2.71), and men (SHR, 1.40; 95% CI, 1.18-1.65).
- Allogeneic BMT with chronic graft-vs-host disease (cGVHD) increased the risk for skin cancer (SHR, 1.84; 95% CI, 1.37-2.47), compared with autologous BMT, while post-BMT immunosuppression increased risk for all types (overall SHR, 1.53; 95% CI, 1.26-1.86).
- The risk for any skin cancer was significantly lower in Black individuals (SHR, 0.14; 95% CI, 0.05-0.37), Hispanic individuals (SHR, 0.29; 95%CI, 0.20-0.62), and patients of other races or who were multiracial (SHR, 0.22; 95% CI, 0.13-0.37) than in non-Hispanic White patients.
IN PRACTICE:
In the study, “risk factors for post-BMT cutaneous malignant neoplasms included pretransplant treatment with a monoclonal antibody, cGVHD, and posttransplant immunosuppression,” the authors wrote, adding that the findings “could inform targeted surveillance of BMT survivors.” Most BMT survivors, “do not undergo routine dermatologic surveillance, highlighting the need to understand risk factors and incorporate risk-informed dermatologic surveillance into survivorship care plans.”
SOURCE:
The study was led by Kristy K. Broman, MD, MPH, University of Alabama at Birmingham, and was published online on December 18 in JAMA Dermatology.
LIMITATIONS:
Limitations included self-reported data and possible underreporting of melanoma cases in the SEER database. Additionally, the study did not capture other risk factors for cutaneous malignant neoplasms such as skin phototype, ultraviolet light exposure, or family history. The duration of posttransplant immunosuppression was not collected, and surveys were administered at variable intervals, though all were completed more than 2 years post BMT.
DISCLOSURES:
The study was supported by the National Cancer Institute (NCI) and the Leukemia and Lymphoma Society. Broman received grants from NCI, the National Center for Advancing Translational Sciences, the American Society of Clinical Oncology, and the American College of Surgeons. Another author reported receiving grants outside this work.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
The Protein Problem: The Unsolved Mystery of AI Drug Dev
The question has been lingering for years in medical science circles. Since 2020, when the artificial intelligence (AI) model AlphaFold made it possible to predict protein structures, would the technology open the drug discovery floodgates?
Short answer: No. At least not yet.
The longer answer goes something like this:
A drug target (such as a mutation) is like a lock. The right drug (a protein designed to bind to the mutation, stopping its activity) is the key. But proteins are fidgety and flexible.
“They’re basically molecular springs,” said Gabriel Monteiro da Silva, PhD, a computational chemistry research scientist at Genesis Therapeutics. “Your key can bend and alter the shape of the lock, and if you don’t account for that, your key might fail.”
This is the protein problem in drug development. Another issue making this challenge so vexing is that proteins don’t act in isolation. Their interactions with other proteins, ribonucleic acid, and DNA can affect how they bind to molecules and the shapes they adopt.
Newer versions of AlphaFold, such as AlphaFold Multimer and AlphaFold 3 (the code for which was recently revealed for academic use), can predict many interactions among proteins and between proteins and other molecules. But these tools still have weak points scientists are trying to overcome or work around.
“Those kinds of dynamics and multiple conformations are still quite challenging for the AI models to predict,” said James Zou, PhD, associate professor of biomedical data science at Stanford University in California.
“We’re finding more and more that the only way we can make these structures useful for drug discovery is if we incorporate dynamics, if we incorporate more physics into the model,” said Monteiro da Silva.
Monteiro da Silva spent 3 years during his PhD at Brown University, Providence, Rhode Island, running physics-based simulations in the lab, trying to understand why proteins carrying certain mutations are drug resistant. His results showed how “the changing landscape of shapes that a protein can take” prevented the drug from binding.
It took him 3 years to model just four mutations.
AI can do better — and the struggle is fascinating. By developing models that build on the predictive power of AlphaFold, scientists are uncovering new details about protein activity — insights that can lead to new therapeutics and reveal why existing ones stop working — much faster than they could with traditional methods or AlphaFold alone.
New Windows into Protein Dynamics
A notable step, “but that’s just the starting point,” said Pedro Beltrao, PhD, an associate professor at Institute of Molecular Systems Biology, ETH Zurich in Switzerland. “It’s still very difficult, given a pocket, to actually design the drug or figure out what the pocket binds.”
Going back to the lock-and-key analogy: While he was at Brown, with a team of researchers in the Rubenstein Group, Monteiro da Silva helped create a model to better understand how mutations affect “the shape and dynamics of the lock.” They manipulated the amino acid sequences of proteins, guiding their evolution. This enabled them to use AlphaFold to predict “protein ensembles” and how frequently those ensembles appear. Each ensemble represents the many different shapes a protein can take under given conditions.
“Essentially, it tries to find the most common shapes that a protein will take over an arbitrary amount of time,” Monteiro da Silva said. “If we can predict these ensembles at scale and fast, then we can screen many mutations that cause resistance and develop drugs that will not be affected by that resistance.”
To evaluate their method, the researchers focused on ABL1, a well-studied kinase that causes leukemia. ABL1 can be drugged – unless it carries or develops a mutation that causes drug resistance. Currently there are no drugs that work against proteins carrying those mutations, according to Monteiro da Silva. The researchers used their hybrid AI-meets-physics method to investigate how drugs bind to different ABL1 mutations, screening 100 mutations in just 1 month.
“It’s not going to be perfect for every one of them. But if we have 100 and we get 20 with good accuracy, that’s better than doing four over 3 years,” Monteiro da Silva said.
A forthcoming paper will make their model publicly available in “an easy-to-use graphical interface” that they hope clinicians and medicinal chemists will try out. It can also complement other AI-based tools that dig into protein dynamics, according to Monteiro da Silva.
Complementary Tools to Speed Up Discovery
Another aspect of the protein problem is scale. One protein can interact with hundreds of other proteins, which in turn may interact with hundreds more, all of which comprise the human interactome.
Feixiong Cheng, PhD, helped build PIONEER, a deep learning model that predicts the three-dimensional (3D) structure of interactions between proteins across the interactome.
Most disease mutations disrupt specific interactions between proteins, making their affinity stronger or weaker, explained Cheng. To treat a disease without causing major side effects, scientists need a precise understanding of those interactions.
“From the drug discovery perspective, we cannot just focus on single proteins. We have to understand the protein environment, in particular how the protein interacts with other proteins,” said Cheng, director of Cleveland Clinic Genome Center, Cleveland.
PIONEER helps by blending AlphaFold’s protein structure predictions with next-generation sequencing, a type of genomic research that identifies mutations in the human genome. The model predicts the 3D structure of the places where proteins interact — the binding sites, or interfaces — across the interactome.
“We tell you not only that a binds b, but where on a and where on b the two proteins interact,” said Haiyuan Yu, PhD, director of the Center for Innovative Proteomics, Cornell University, and co-creator of PIONEER.
This can help scientists understand “why a mutation, protein, or even network is a good target for therapeutic discovery,” Cheng said.
The researchers validated PIONEER’s predictions in the lab, testing the impacts of roughly 3000 mutations on 7000 pairs of interacting proteins. Based on their findings, they plan to develop and test treatments for lung and endometrial cancer.
PIONEER can also help scientists home in on how a mutation causes a disease, such as by showing recurrent mutations.
“If you find cancer mutations hitting an interface again and again and again, it means that this is likely to be driving cancer progression,” said Beltrao.
Beltrao’s lab and others have looked for recurrent mutations by using AlphaFold Multimer and AlphaFold 3 to directly model protein interactions. It’s a much slower approach (Pioneer is more than 5000 faster than AlphaFold Multimer, according to Cheng). But it could allow scientists to model interfaces that are not shown by PIONEER.
“You will need many different things to try to come up with a structural modeling of the interactome, and all these will have limitations,” said Beltrao. “Their method is a very good step forward, and there’ll be other approaches that are complementary, to continue to add details.”
And It Wouldn’t be an AI Mission Without ChatGPT
Large language models, such as ChatGPT, are another way that scientists are adding details to protein structure predictions. Zou used GPT-4 to “fine tune” a protein language model, called evolutionary scale modeling (ESM-2), which predicts protein structures directly from a protein sequence.
First, they trained ChatGPT on thousands of papers and studies containing information about the functions, biophysical properties, and disease relevance of different mutations. Next, they used the trained model to “teach” ESM-2, boosting its ability “to predict which mutations are likely to have larger effects or smaller effects,” Zou said. The same could be done for a model like AlphaFold, according to Zou.
“They are quite complementary in that the large language model contains a lot more information about the functions and the biophysics of different mutations and proteins as captured in text,” he said, whereas “you can’t give AlphaFold a piece of paper.”
Exactly how AlphaFold makes its predictions is another mystery. “It will somehow learn protein dynamics phenomenologically,” said Monteiro da Silva. He and others are trying to understand how that happens, in hopes of creating even more accurate predictive models. But for the time being, AI-based methods still need assistance from physics.
“The dream is that we achieve a state where we rely on just the fast methods, and they’re accurate enough,” he said. “But we’re so far from that.”
A version of this article first appeared on Medscape.com.
The question has been lingering for years in medical science circles. Since 2020, when the artificial intelligence (AI) model AlphaFold made it possible to predict protein structures, would the technology open the drug discovery floodgates?
Short answer: No. At least not yet.
The longer answer goes something like this:
A drug target (such as a mutation) is like a lock. The right drug (a protein designed to bind to the mutation, stopping its activity) is the key. But proteins are fidgety and flexible.
“They’re basically molecular springs,” said Gabriel Monteiro da Silva, PhD, a computational chemistry research scientist at Genesis Therapeutics. “Your key can bend and alter the shape of the lock, and if you don’t account for that, your key might fail.”
This is the protein problem in drug development. Another issue making this challenge so vexing is that proteins don’t act in isolation. Their interactions with other proteins, ribonucleic acid, and DNA can affect how they bind to molecules and the shapes they adopt.
Newer versions of AlphaFold, such as AlphaFold Multimer and AlphaFold 3 (the code for which was recently revealed for academic use), can predict many interactions among proteins and between proteins and other molecules. But these tools still have weak points scientists are trying to overcome or work around.
“Those kinds of dynamics and multiple conformations are still quite challenging for the AI models to predict,” said James Zou, PhD, associate professor of biomedical data science at Stanford University in California.
“We’re finding more and more that the only way we can make these structures useful for drug discovery is if we incorporate dynamics, if we incorporate more physics into the model,” said Monteiro da Silva.
Monteiro da Silva spent 3 years during his PhD at Brown University, Providence, Rhode Island, running physics-based simulations in the lab, trying to understand why proteins carrying certain mutations are drug resistant. His results showed how “the changing landscape of shapes that a protein can take” prevented the drug from binding.
It took him 3 years to model just four mutations.
AI can do better — and the struggle is fascinating. By developing models that build on the predictive power of AlphaFold, scientists are uncovering new details about protein activity — insights that can lead to new therapeutics and reveal why existing ones stop working — much faster than they could with traditional methods or AlphaFold alone.
New Windows into Protein Dynamics
A notable step, “but that’s just the starting point,” said Pedro Beltrao, PhD, an associate professor at Institute of Molecular Systems Biology, ETH Zurich in Switzerland. “It’s still very difficult, given a pocket, to actually design the drug or figure out what the pocket binds.”
Going back to the lock-and-key analogy: While he was at Brown, with a team of researchers in the Rubenstein Group, Monteiro da Silva helped create a model to better understand how mutations affect “the shape and dynamics of the lock.” They manipulated the amino acid sequences of proteins, guiding their evolution. This enabled them to use AlphaFold to predict “protein ensembles” and how frequently those ensembles appear. Each ensemble represents the many different shapes a protein can take under given conditions.
“Essentially, it tries to find the most common shapes that a protein will take over an arbitrary amount of time,” Monteiro da Silva said. “If we can predict these ensembles at scale and fast, then we can screen many mutations that cause resistance and develop drugs that will not be affected by that resistance.”
To evaluate their method, the researchers focused on ABL1, a well-studied kinase that causes leukemia. ABL1 can be drugged – unless it carries or develops a mutation that causes drug resistance. Currently there are no drugs that work against proteins carrying those mutations, according to Monteiro da Silva. The researchers used their hybrid AI-meets-physics method to investigate how drugs bind to different ABL1 mutations, screening 100 mutations in just 1 month.
“It’s not going to be perfect for every one of them. But if we have 100 and we get 20 with good accuracy, that’s better than doing four over 3 years,” Monteiro da Silva said.
A forthcoming paper will make their model publicly available in “an easy-to-use graphical interface” that they hope clinicians and medicinal chemists will try out. It can also complement other AI-based tools that dig into protein dynamics, according to Monteiro da Silva.
Complementary Tools to Speed Up Discovery
Another aspect of the protein problem is scale. One protein can interact with hundreds of other proteins, which in turn may interact with hundreds more, all of which comprise the human interactome.
Feixiong Cheng, PhD, helped build PIONEER, a deep learning model that predicts the three-dimensional (3D) structure of interactions between proteins across the interactome.
Most disease mutations disrupt specific interactions between proteins, making their affinity stronger or weaker, explained Cheng. To treat a disease without causing major side effects, scientists need a precise understanding of those interactions.
“From the drug discovery perspective, we cannot just focus on single proteins. We have to understand the protein environment, in particular how the protein interacts with other proteins,” said Cheng, director of Cleveland Clinic Genome Center, Cleveland.
PIONEER helps by blending AlphaFold’s protein structure predictions with next-generation sequencing, a type of genomic research that identifies mutations in the human genome. The model predicts the 3D structure of the places where proteins interact — the binding sites, or interfaces — across the interactome.
“We tell you not only that a binds b, but where on a and where on b the two proteins interact,” said Haiyuan Yu, PhD, director of the Center for Innovative Proteomics, Cornell University, and co-creator of PIONEER.
This can help scientists understand “why a mutation, protein, or even network is a good target for therapeutic discovery,” Cheng said.
The researchers validated PIONEER’s predictions in the lab, testing the impacts of roughly 3000 mutations on 7000 pairs of interacting proteins. Based on their findings, they plan to develop and test treatments for lung and endometrial cancer.
PIONEER can also help scientists home in on how a mutation causes a disease, such as by showing recurrent mutations.
“If you find cancer mutations hitting an interface again and again and again, it means that this is likely to be driving cancer progression,” said Beltrao.
Beltrao’s lab and others have looked for recurrent mutations by using AlphaFold Multimer and AlphaFold 3 to directly model protein interactions. It’s a much slower approach (Pioneer is more than 5000 faster than AlphaFold Multimer, according to Cheng). But it could allow scientists to model interfaces that are not shown by PIONEER.
“You will need many different things to try to come up with a structural modeling of the interactome, and all these will have limitations,” said Beltrao. “Their method is a very good step forward, and there’ll be other approaches that are complementary, to continue to add details.”
And It Wouldn’t be an AI Mission Without ChatGPT
Large language models, such as ChatGPT, are another way that scientists are adding details to protein structure predictions. Zou used GPT-4 to “fine tune” a protein language model, called evolutionary scale modeling (ESM-2), which predicts protein structures directly from a protein sequence.
First, they trained ChatGPT on thousands of papers and studies containing information about the functions, biophysical properties, and disease relevance of different mutations. Next, they used the trained model to “teach” ESM-2, boosting its ability “to predict which mutations are likely to have larger effects or smaller effects,” Zou said. The same could be done for a model like AlphaFold, according to Zou.
“They are quite complementary in that the large language model contains a lot more information about the functions and the biophysics of different mutations and proteins as captured in text,” he said, whereas “you can’t give AlphaFold a piece of paper.”
Exactly how AlphaFold makes its predictions is another mystery. “It will somehow learn protein dynamics phenomenologically,” said Monteiro da Silva. He and others are trying to understand how that happens, in hopes of creating even more accurate predictive models. But for the time being, AI-based methods still need assistance from physics.
“The dream is that we achieve a state where we rely on just the fast methods, and they’re accurate enough,” he said. “But we’re so far from that.”
A version of this article first appeared on Medscape.com.
The question has been lingering for years in medical science circles. Since 2020, when the artificial intelligence (AI) model AlphaFold made it possible to predict protein structures, would the technology open the drug discovery floodgates?
Short answer: No. At least not yet.
The longer answer goes something like this:
A drug target (such as a mutation) is like a lock. The right drug (a protein designed to bind to the mutation, stopping its activity) is the key. But proteins are fidgety and flexible.
“They’re basically molecular springs,” said Gabriel Monteiro da Silva, PhD, a computational chemistry research scientist at Genesis Therapeutics. “Your key can bend and alter the shape of the lock, and if you don’t account for that, your key might fail.”
This is the protein problem in drug development. Another issue making this challenge so vexing is that proteins don’t act in isolation. Their interactions with other proteins, ribonucleic acid, and DNA can affect how they bind to molecules and the shapes they adopt.
Newer versions of AlphaFold, such as AlphaFold Multimer and AlphaFold 3 (the code for which was recently revealed for academic use), can predict many interactions among proteins and between proteins and other molecules. But these tools still have weak points scientists are trying to overcome or work around.
“Those kinds of dynamics and multiple conformations are still quite challenging for the AI models to predict,” said James Zou, PhD, associate professor of biomedical data science at Stanford University in California.
“We’re finding more and more that the only way we can make these structures useful for drug discovery is if we incorporate dynamics, if we incorporate more physics into the model,” said Monteiro da Silva.
Monteiro da Silva spent 3 years during his PhD at Brown University, Providence, Rhode Island, running physics-based simulations in the lab, trying to understand why proteins carrying certain mutations are drug resistant. His results showed how “the changing landscape of shapes that a protein can take” prevented the drug from binding.
It took him 3 years to model just four mutations.
AI can do better — and the struggle is fascinating. By developing models that build on the predictive power of AlphaFold, scientists are uncovering new details about protein activity — insights that can lead to new therapeutics and reveal why existing ones stop working — much faster than they could with traditional methods or AlphaFold alone.
New Windows into Protein Dynamics
A notable step, “but that’s just the starting point,” said Pedro Beltrao, PhD, an associate professor at Institute of Molecular Systems Biology, ETH Zurich in Switzerland. “It’s still very difficult, given a pocket, to actually design the drug or figure out what the pocket binds.”
Going back to the lock-and-key analogy: While he was at Brown, with a team of researchers in the Rubenstein Group, Monteiro da Silva helped create a model to better understand how mutations affect “the shape and dynamics of the lock.” They manipulated the amino acid sequences of proteins, guiding their evolution. This enabled them to use AlphaFold to predict “protein ensembles” and how frequently those ensembles appear. Each ensemble represents the many different shapes a protein can take under given conditions.
“Essentially, it tries to find the most common shapes that a protein will take over an arbitrary amount of time,” Monteiro da Silva said. “If we can predict these ensembles at scale and fast, then we can screen many mutations that cause resistance and develop drugs that will not be affected by that resistance.”
To evaluate their method, the researchers focused on ABL1, a well-studied kinase that causes leukemia. ABL1 can be drugged – unless it carries or develops a mutation that causes drug resistance. Currently there are no drugs that work against proteins carrying those mutations, according to Monteiro da Silva. The researchers used their hybrid AI-meets-physics method to investigate how drugs bind to different ABL1 mutations, screening 100 mutations in just 1 month.
“It’s not going to be perfect for every one of them. But if we have 100 and we get 20 with good accuracy, that’s better than doing four over 3 years,” Monteiro da Silva said.
A forthcoming paper will make their model publicly available in “an easy-to-use graphical interface” that they hope clinicians and medicinal chemists will try out. It can also complement other AI-based tools that dig into protein dynamics, according to Monteiro da Silva.
Complementary Tools to Speed Up Discovery
Another aspect of the protein problem is scale. One protein can interact with hundreds of other proteins, which in turn may interact with hundreds more, all of which comprise the human interactome.
Feixiong Cheng, PhD, helped build PIONEER, a deep learning model that predicts the three-dimensional (3D) structure of interactions between proteins across the interactome.
Most disease mutations disrupt specific interactions between proteins, making their affinity stronger or weaker, explained Cheng. To treat a disease without causing major side effects, scientists need a precise understanding of those interactions.
“From the drug discovery perspective, we cannot just focus on single proteins. We have to understand the protein environment, in particular how the protein interacts with other proteins,” said Cheng, director of Cleveland Clinic Genome Center, Cleveland.
PIONEER helps by blending AlphaFold’s protein structure predictions with next-generation sequencing, a type of genomic research that identifies mutations in the human genome. The model predicts the 3D structure of the places where proteins interact — the binding sites, or interfaces — across the interactome.
“We tell you not only that a binds b, but where on a and where on b the two proteins interact,” said Haiyuan Yu, PhD, director of the Center for Innovative Proteomics, Cornell University, and co-creator of PIONEER.
This can help scientists understand “why a mutation, protein, or even network is a good target for therapeutic discovery,” Cheng said.
The researchers validated PIONEER’s predictions in the lab, testing the impacts of roughly 3000 mutations on 7000 pairs of interacting proteins. Based on their findings, they plan to develop and test treatments for lung and endometrial cancer.
PIONEER can also help scientists home in on how a mutation causes a disease, such as by showing recurrent mutations.
“If you find cancer mutations hitting an interface again and again and again, it means that this is likely to be driving cancer progression,” said Beltrao.
Beltrao’s lab and others have looked for recurrent mutations by using AlphaFold Multimer and AlphaFold 3 to directly model protein interactions. It’s a much slower approach (Pioneer is more than 5000 faster than AlphaFold Multimer, according to Cheng). But it could allow scientists to model interfaces that are not shown by PIONEER.
“You will need many different things to try to come up with a structural modeling of the interactome, and all these will have limitations,” said Beltrao. “Their method is a very good step forward, and there’ll be other approaches that are complementary, to continue to add details.”
And It Wouldn’t be an AI Mission Without ChatGPT
Large language models, such as ChatGPT, are another way that scientists are adding details to protein structure predictions. Zou used GPT-4 to “fine tune” a protein language model, called evolutionary scale modeling (ESM-2), which predicts protein structures directly from a protein sequence.
First, they trained ChatGPT on thousands of papers and studies containing information about the functions, biophysical properties, and disease relevance of different mutations. Next, they used the trained model to “teach” ESM-2, boosting its ability “to predict which mutations are likely to have larger effects or smaller effects,” Zou said. The same could be done for a model like AlphaFold, according to Zou.
“They are quite complementary in that the large language model contains a lot more information about the functions and the biophysics of different mutations and proteins as captured in text,” he said, whereas “you can’t give AlphaFold a piece of paper.”
Exactly how AlphaFold makes its predictions is another mystery. “It will somehow learn protein dynamics phenomenologically,” said Monteiro da Silva. He and others are trying to understand how that happens, in hopes of creating even more accurate predictive models. But for the time being, AI-based methods still need assistance from physics.
“The dream is that we achieve a state where we rely on just the fast methods, and they’re accurate enough,” he said. “But we’re so far from that.”
A version of this article first appeared on Medscape.com.
Does Marijuana Harm Your Lungs? The Unclear Truth
During a recent walk with my 6-year-old, he told me he smelled marijuana. His comment speaks to its increased (and more open) use since legalization in our state. The macho, misguided part of my dad psyche was proud of his “street cred” but the thinking part of my brain was concerned. He seemed a little young for a talk about drugs.
I was able to provide a simple, watered-down list of reasons why he shouldn’t smoke marijuana or anything else. The “drugs are bad” aphorism sufficed for my 6-year-old but wasn’t worthy of an academic pulmonologist.
I retired from the military 2 years ago, so marijuana (I’m using the terms “marijuana” and “cannabis” interchangeably here) knowledge wasn’t required for regular practice. I recall one 60-year-old patient who reported smoking four joints a day for years. He had marked emphysema on CT, severe obstruction on spirometry, and he was functionally limited. Buttressed by scattered reports of acute lung injury caused by dabbing or marijuana vaping, this anecdotal “n of 1” led to a predictably pedantic conclusion: Smoking marijuana is bad for the lungs and preaching cessation is worth my time and effort.
I now work in an inner-city hospital. My 6-year-old could identify the smell permeating the hallways and clinic rooms. I’ve reverted to counseling cessation using little more than my “drugs are bad” speech. When I came across a recent review in Seminars in Respiratory and Critical Care Medicine, I recognized the opportunity to read and do better. This summary is based heavily on that review.
Spoiler alert: The data aren’t great. By federal law, marijuana has been illegal in the United States since 1970, so neither funding nor recruitment has come easy. There’s lots of observational data that depend on self-report and are confounded by cigarette use. A lack of regulation results in variations in composition and concentration. In summary, though, smoking marijuana is associated with changes to the bronchial tree and respiratory symptoms, similar to those seen with chronic bronchitis. These symptoms improve with cessation.
The relationship between marijuana and airflow obstruction and lung function is complicated. A mix of contradictory data shows a reduction in the ratio of the forced expiratory volume in the first 1 second to the forced vital capacity (FEV1/FVC), an increase in FVC, and changes in conductance.
Biologic plausibility, essential to bolster causality but easy to manufacture, seems intuitive for the airway changes (decreased FEV1/FVC and conductance). The increase in FVC, explained by either the anti-inflammatory properties of delta-9-tetrahydrocannabinol (THC) or the impact from deep inhalations typical of marijuana use, is more difficult to understand. Regardless, I came away from the review less confident about marijuana’s impact on lung structure and function.
The Seminars review also explores marijuana’s association with lung cancer, emphysema, and other structural changes seen on CT of the chest. There’s certainly noise here but the data at present are underwhelming.
This all speaks to the general misconception I’ve had, perhaps shared by others, that the well-defined effects on the lung from tobacco abuse can be extrapolated to marijuana. In the past, I’d even gone so far as to equate a pack-year (smoking one pack of cigarettes per day for a year) to a joint-year (smoking one joint per day for a year), a rather dramatic oversimplification. While both are attempts to quantify exposure, the latter connotes far less information. The content of a joint can vary considerably in ways that the content of cigarettes does not, and there have been no formal studies of the comparative impact on the lung.
Final Thoughts
The nuance here matters for several reasons. Legalization means an increase in use and presumably more open reporting by patients. In a vacuum, it seems reasonable to council cessation to reduce symptoms and because additional harms can be assumed, given what we know about smoke inhalation in general. Will cessation drive patients to an increase in tobacco use where harm is better established?
Given its mixed effects on lung function, is it worth spending behavior change capital, the most precious of patient commodities, on marijuana counseling? Marijuana has numerous effects outside the lung that haven’t been touched on here. How should those be incorporated into our guidance? Legalization and regulation provide the opportunity to obtain the better data that are sorely needed.
Aaron B. Holley, MD, is a professor of medicine at Uniformed Services University in Bethesda, Maryland, and a pulmonary/sleep and critical care medicine physician at MedStar Washington Hospital Center in Washington, DC. He has disclosed the relevant financial relationships with Metapharm, CHEST College, and WebMD.
A version of this article first appeared on Medscape.com.
During a recent walk with my 6-year-old, he told me he smelled marijuana. His comment speaks to its increased (and more open) use since legalization in our state. The macho, misguided part of my dad psyche was proud of his “street cred” but the thinking part of my brain was concerned. He seemed a little young for a talk about drugs.
I was able to provide a simple, watered-down list of reasons why he shouldn’t smoke marijuana or anything else. The “drugs are bad” aphorism sufficed for my 6-year-old but wasn’t worthy of an academic pulmonologist.
I retired from the military 2 years ago, so marijuana (I’m using the terms “marijuana” and “cannabis” interchangeably here) knowledge wasn’t required for regular practice. I recall one 60-year-old patient who reported smoking four joints a day for years. He had marked emphysema on CT, severe obstruction on spirometry, and he was functionally limited. Buttressed by scattered reports of acute lung injury caused by dabbing or marijuana vaping, this anecdotal “n of 1” led to a predictably pedantic conclusion: Smoking marijuana is bad for the lungs and preaching cessation is worth my time and effort.
I now work in an inner-city hospital. My 6-year-old could identify the smell permeating the hallways and clinic rooms. I’ve reverted to counseling cessation using little more than my “drugs are bad” speech. When I came across a recent review in Seminars in Respiratory and Critical Care Medicine, I recognized the opportunity to read and do better. This summary is based heavily on that review.
Spoiler alert: The data aren’t great. By federal law, marijuana has been illegal in the United States since 1970, so neither funding nor recruitment has come easy. There’s lots of observational data that depend on self-report and are confounded by cigarette use. A lack of regulation results in variations in composition and concentration. In summary, though, smoking marijuana is associated with changes to the bronchial tree and respiratory symptoms, similar to those seen with chronic bronchitis. These symptoms improve with cessation.
The relationship between marijuana and airflow obstruction and lung function is complicated. A mix of contradictory data shows a reduction in the ratio of the forced expiratory volume in the first 1 second to the forced vital capacity (FEV1/FVC), an increase in FVC, and changes in conductance.
Biologic plausibility, essential to bolster causality but easy to manufacture, seems intuitive for the airway changes (decreased FEV1/FVC and conductance). The increase in FVC, explained by either the anti-inflammatory properties of delta-9-tetrahydrocannabinol (THC) or the impact from deep inhalations typical of marijuana use, is more difficult to understand. Regardless, I came away from the review less confident about marijuana’s impact on lung structure and function.
The Seminars review also explores marijuana’s association with lung cancer, emphysema, and other structural changes seen on CT of the chest. There’s certainly noise here but the data at present are underwhelming.
This all speaks to the general misconception I’ve had, perhaps shared by others, that the well-defined effects on the lung from tobacco abuse can be extrapolated to marijuana. In the past, I’d even gone so far as to equate a pack-year (smoking one pack of cigarettes per day for a year) to a joint-year (smoking one joint per day for a year), a rather dramatic oversimplification. While both are attempts to quantify exposure, the latter connotes far less information. The content of a joint can vary considerably in ways that the content of cigarettes does not, and there have been no formal studies of the comparative impact on the lung.
Final Thoughts
The nuance here matters for several reasons. Legalization means an increase in use and presumably more open reporting by patients. In a vacuum, it seems reasonable to council cessation to reduce symptoms and because additional harms can be assumed, given what we know about smoke inhalation in general. Will cessation drive patients to an increase in tobacco use where harm is better established?
Given its mixed effects on lung function, is it worth spending behavior change capital, the most precious of patient commodities, on marijuana counseling? Marijuana has numerous effects outside the lung that haven’t been touched on here. How should those be incorporated into our guidance? Legalization and regulation provide the opportunity to obtain the better data that are sorely needed.
Aaron B. Holley, MD, is a professor of medicine at Uniformed Services University in Bethesda, Maryland, and a pulmonary/sleep and critical care medicine physician at MedStar Washington Hospital Center in Washington, DC. He has disclosed the relevant financial relationships with Metapharm, CHEST College, and WebMD.
A version of this article first appeared on Medscape.com.
During a recent walk with my 6-year-old, he told me he smelled marijuana. His comment speaks to its increased (and more open) use since legalization in our state. The macho, misguided part of my dad psyche was proud of his “street cred” but the thinking part of my brain was concerned. He seemed a little young for a talk about drugs.
I was able to provide a simple, watered-down list of reasons why he shouldn’t smoke marijuana or anything else. The “drugs are bad” aphorism sufficed for my 6-year-old but wasn’t worthy of an academic pulmonologist.
I retired from the military 2 years ago, so marijuana (I’m using the terms “marijuana” and “cannabis” interchangeably here) knowledge wasn’t required for regular practice. I recall one 60-year-old patient who reported smoking four joints a day for years. He had marked emphysema on CT, severe obstruction on spirometry, and he was functionally limited. Buttressed by scattered reports of acute lung injury caused by dabbing or marijuana vaping, this anecdotal “n of 1” led to a predictably pedantic conclusion: Smoking marijuana is bad for the lungs and preaching cessation is worth my time and effort.
I now work in an inner-city hospital. My 6-year-old could identify the smell permeating the hallways and clinic rooms. I’ve reverted to counseling cessation using little more than my “drugs are bad” speech. When I came across a recent review in Seminars in Respiratory and Critical Care Medicine, I recognized the opportunity to read and do better. This summary is based heavily on that review.
Spoiler alert: The data aren’t great. By federal law, marijuana has been illegal in the United States since 1970, so neither funding nor recruitment has come easy. There’s lots of observational data that depend on self-report and are confounded by cigarette use. A lack of regulation results in variations in composition and concentration. In summary, though, smoking marijuana is associated with changes to the bronchial tree and respiratory symptoms, similar to those seen with chronic bronchitis. These symptoms improve with cessation.
The relationship between marijuana and airflow obstruction and lung function is complicated. A mix of contradictory data shows a reduction in the ratio of the forced expiratory volume in the first 1 second to the forced vital capacity (FEV1/FVC), an increase in FVC, and changes in conductance.
Biologic plausibility, essential to bolster causality but easy to manufacture, seems intuitive for the airway changes (decreased FEV1/FVC and conductance). The increase in FVC, explained by either the anti-inflammatory properties of delta-9-tetrahydrocannabinol (THC) or the impact from deep inhalations typical of marijuana use, is more difficult to understand. Regardless, I came away from the review less confident about marijuana’s impact on lung structure and function.
The Seminars review also explores marijuana’s association with lung cancer, emphysema, and other structural changes seen on CT of the chest. There’s certainly noise here but the data at present are underwhelming.
This all speaks to the general misconception I’ve had, perhaps shared by others, that the well-defined effects on the lung from tobacco abuse can be extrapolated to marijuana. In the past, I’d even gone so far as to equate a pack-year (smoking one pack of cigarettes per day for a year) to a joint-year (smoking one joint per day for a year), a rather dramatic oversimplification. While both are attempts to quantify exposure, the latter connotes far less information. The content of a joint can vary considerably in ways that the content of cigarettes does not, and there have been no formal studies of the comparative impact on the lung.
Final Thoughts
The nuance here matters for several reasons. Legalization means an increase in use and presumably more open reporting by patients. In a vacuum, it seems reasonable to council cessation to reduce symptoms and because additional harms can be assumed, given what we know about smoke inhalation in general. Will cessation drive patients to an increase in tobacco use where harm is better established?
Given its mixed effects on lung function, is it worth spending behavior change capital, the most precious of patient commodities, on marijuana counseling? Marijuana has numerous effects outside the lung that haven’t been touched on here. How should those be incorporated into our guidance? Legalization and regulation provide the opportunity to obtain the better data that are sorely needed.
Aaron B. Holley, MD, is a professor of medicine at Uniformed Services University in Bethesda, Maryland, and a pulmonary/sleep and critical care medicine physician at MedStar Washington Hospital Center in Washington, DC. He has disclosed the relevant financial relationships with Metapharm, CHEST College, and WebMD.
A version of this article first appeared on Medscape.com.