User login
HT for women who have had BSO before the age of natural menopause: Discerning the nuances
Women who undergo bilateral salpingo-oophorectomy (BSO) for various indications prior to menopause experience a rapid decline in ovarian hormone levels and consequent vasomotor and other menopausal symptoms. In addition, the resulting estrogen deprivation is associated with such long-term adverse outcomes as osteoporosis and cardiovascular morbidity.
OBG M
Surgical vs natural menopause
Stephanie Faubion, MD, MBA, NCMP: Since the Women’s Health Initiative study was published in 2002,2 many clinicians have been fearful of using systemic HT in menopausal women, and HT use has declined dramatically such that only about 4% to 6% of menopausal women are now receiving systemic HT. Importantly, however, a group of younger menopausal women also are not receiving HT, and that is women who undergo BSO before they reach the average age of menopause, which in the United States is about age 52; this is sometimes referred to as surgical menopause or early surgical menopause. Early surgical menopause has different connotations for long-term health risks than natural menopause at the average age, and we are here to discuss these health effects and their management.
My name is Stephanie Faubion, and I am a women’s health internist and the Chair of the Department of Medicine at Mayo Clinic in Jacksonville, Florida, and Director of Mayo Clinic Women’s Health. I am here with 2 of my esteemed colleagues, Dr. Andrew Kaunitz and Dr. Ekta Kapoor.
Andrew M. Kaunitz, MD, NCMP: Hello, I am an ObGyn with the University of Florida College of Medicine in Jacksonville, with particular interests in contraception, menopause, and gynecologic ultrasonography.
Ekta Kapoor, MBBS, NCMP: And I am an endocrinologist at Mayo Clinic in Rochester with a specific interest in menopause and hormone therapy. I am also the Assistant Director for Mayo Clinic Women’s Health.
Higher-than-standard estrogen doses needed in younger menopausal women
Dr. Faubion: Let’s consider a couple of cases so that we can illustrate some important points regarding hormone management in women who have undergone BSO before the age of natural menopause.
Our first case patient is a woman who is 41 years of age and, because of adenomyosis, she will undergo a hysterectomy. She tells her clinician that she is very concerned about ovarian cancer risk because one of her good friends recently was diagnosed with ovarian cancer, and together they decide to remove her ovaries at the time of hysterectomy. Notably, her ovaries were healthy.
The patient is now menopausal postsurgery, and she is having significant hot flashes and night sweats. She visits her local internist, who is concerned about initiating HT. She is otherwise a healthy woman and does not have any contraindications to HT. Dr. Kaunitz, what would you tell her internist?
Dr. Kaunitz: We are dealing with 2 different issues in terms of decision making about systemic HT for this 41-year-old who has undergone BSO. First, as you mentioned, Dr. Faubion, she has bothersome hot flashes, or vasomotor symptoms. Unless there are contraindications, systemic HT would be appropriate. Although I might start treatment at standard doses, and the accompanying TABLE depicts standard doses for the 2 most common oral estrogen formulations as well as transdermal estradiol, it’s important to recognize that younger menopausal women often will need to use higher-than-standard doses.
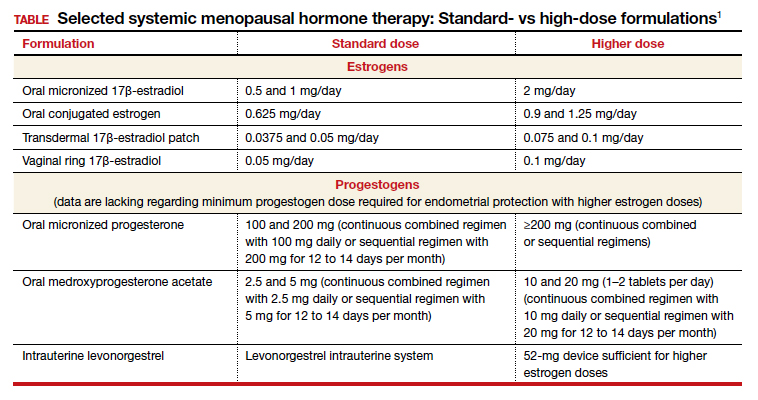
For example, for a 53-year-old woman who has been menopausal for a year or 2 and now has bothersome symptoms, I might start her on estradiol 1 mg tablets with progestin if a uterus is present. However, in this 41-year-old case patient, while I might start treatment at a standard dose, I would anticipate increasing to higher doses, such as 1.5 or 2 mg of daily estradiol until she feels her menopausal symptoms are adequately addressed.
Dr. Faubion: It is important to note that sometimes women with early BSO tend to have more severe vasomotor symptoms. Do you find that sometimes a higher dose is required just to manage symptoms, Dr. Kaunitz?
Dr. Kaunitz: Absolutely, yes. The decision whether or not to use systemic HT might be considered discretionary or elective in the classic 53-year-old woman recently menopausal with hot flashes, a so-called spontaneously or naturally menopausal woman. But my perspective is that unless there are clear contraindications, the decision to start systemic HT in the 41-year-old BSO case patient is actually not discretionary. Unless contraindications are present, it is important not only to treat symptoms but also to prevent an array of chronic major health concerns that are more likely if we don’t prescribe systemic HT.
Continue to: Health effects of not using HT...
Health effects of not using HT
Dr. Faubion: Dr. Kapoor, can you describe the potential long-term adverse health consequences of not using estrogen therapy? Say the same 41-year-old woman does not have many bothersome symptoms. What would you do?
Dr. Kapoor: Thank you for that important question. Building on what Dr. Kaunitz said, in these patients there are really 2 issues that can seem to be independent but are not: The first relates to the immediate consequences of lack of estrogen, ie, the menopause-related symptoms, but the second and perhaps the bigger issue is the long-term risk associated with estrogen deprivation.
The symptoms in these women are often obvious as they can be quite severe and abrupt; one day these women have normal hormone levels and the next day, after BSO, suddenly their hormones are very low. So if symptoms occur, they are usually hard to miss, simply because they are very drastic and very severe.
Historically, patients and their clinicians have targeted these symptoms. Patients experience menopausal symptoms, they seek treatment, and then the clinicians basically titrate the treatment to manage these symptoms. That misses the bigger issue, however, which is that premature estrogen deprivation leads to a host of chronic health conditions, as Dr. Kaunitz mentioned. These mainly include increased risk for cardiovascular disease, diabetes, hypertension, dyslipidemia, increased risk of mortality, dementia, and osteoporosis.
Fairly strong observational evidence suggests that use of estrogen therapy given in replacement doses—doses higher than those typically used in women after natural menopause, therefore considered replacement doses—helps mitigate the risk of some of these adverse health conditions.
In these women, the bigger goal really is to reinstate the hormonal milieu that exists prior to menopause. To your point, Dr. Faubion, if I have a patient who is younger than 46 years, who has her ovaries taken out, and even if she has zero symptoms (and sometimes that does happen), I would still make a case for this patient to utilize hormone therapy unless there is a contraindication such as breast cancer or other estrogen-sensitive cancers.
Dr. Faubion: Again, would you aim for those higher doses rather than treat with the “lowest dose”?
Dr. Kapoor: Absolutely. My punchline to the patients and clinicians in these discussions is that the rules of the game are different for these women. We cannot extrapolate the risks and benefits of HT use in women after natural menopause to younger women who have surgical menopause. Those rules just do not apply with respect to both benefits and risks.
Dr. Faubion: I think it’s important to say that these same “rules” would apply if the women were to go through premature menopause for any other reason, too, such as chemotherapy, radiation therapy, or premature ovarian insufficiency for any number of reasons, including toxic, metabolic, or genetic causes and so on. Would that be true?
Dr. Kapoor: Yes, absolutely so.
Dr. Faubion: Dr. Kaunitz, do you want to add anything?
Dr. Kaunitz: In terms of practical or clinical issues regarding systemic HT management, for the woman in her early 50s who has experienced normal or natural spontaneous menopause, a starting dose of transdermal estradiol would be, for instance, a 0.05-mg patch, which is a patch that over 24 hours releases 0.05 mg of estradiol daily; or standard oral estrogen, including conjugated equine estrogen, a 0.625-mg tablet daily, or estradiol, a 1-mg tablet daily.
But in younger patients, we want to use higher doses. For a patch, for instance, I would aim for a 0.075- or 0.1-mg estradiol patch, which releases a higher daily dose of estradiol than the standard dose. For oral estrogen, the dose would be 0.9- or even 1.25-mg tablets of conjugated equine estrogen or 1.5 mg, which is a 1-mg plus a 0.5-mg estradiol tablet, or a 2-mg estradiol tablet. Estradiol does come in a 2-mg strength.
For oral estrogen, I prefer estradiol because it’s available as a generic medication and often available at a very low cost, sometimes as low as $4 a month from chain pharmacies.
Continue to: Usefulness of monitoring estradiol levels for dosage adjustment...
Usefulness of monitoring estradiol levels for dosage adjustment
Dr. Faubion: That’s a great point, and again it is important to emphasize that we are aiming to recreate the premenopausal hormonal milieu. If you were to check estradiol levels, that would be aiming for a premenopausal range of approximately 80 to 120 pg per mL. Dr. Kapoor, is there utility in monitoring estrogen levels?
Dr. Kapoor: Great question, Dr. Faubion, and as you know it’s a loaded one. We base this on empiric evidence. We know that if the hormonal milieu in a young patient is changed to a postmenopausal one, her risk for many chronic conditions is increased. So if we were to reinstate a premenopausal hormonal milieu, that risk would probably be reduced. It makes good sense to target an empiric goal of 80 to 120 pg per mL of estradiol, which is the average estradiol level in a premenopausal woman. If you were to ask me, however, are there randomized, controlled trial data to support this practice—that is, if you target that level, can you make sure that the risk of diabetes is lower or that the risk of heart disease is lower—that study has yet to be done, and it may not ever be done on a large scale. However, it intuitively makes good sense to target premenopausal estradiol levels.
Dr. Faubion: When might you check an estradiol level in this population? For example, if you are treating a patient with a 0.1-mg estradiol patch and she still has significant hot flashes, would it be useful to check the level?
Dr. Kapoor: It would. In my practice, I check estradiol levels on these patients on an annual basis, regardless of symptoms, but definitely in the patient who has symptoms. It makes good sense, because sometimes these patients don’t absorb the estrogen well, particularly if administered by the transdermal route.
A general rule of thumb is that in the average population, if a patient is on the 0.1-mg patch, for example, you would expect her level to be around 100. If it is much lower than that, which sometimes happens, that speaks for poor absorption. Options at that point would be to treat her with a higher dose patch, depending on what the level is, or switch to a different formulation, such as oral.
In instances in which I have treated patients with a 0.1-mg patch for example, and their estradiol levels are undetectable, that speaks for very poor absorption. For such patients I make a case for switching them to oral therapy. Most definitely that makes sense in a patient who is symptomatic despite treatment. But even for patients who don’t have symptoms, I like to target that level, acknowledging that there is no evidence as such to support this practice.
Dr. Faubion: Dr. Kaunitz, do you want to add anything?
Dr. Kaunitz: Yes, a few practical points. Although patches are available in a wider array of doses than oral estrogen formulations, the highest dose available is 0.1 mg. It’s important for clinicians to recognize that while checking serum levels when indicated can be performed in women using transdermal estradiol or patches, in women who are using oral estrogen, checking blood levels is not going to work well because serum estrogen levels have a daily peak and valley in women who use oral versus transdermal estradiol.
I also wanted to talk about progestins. Although many patients who have had a BSO prior to spontaneous menopause also have had a hysterectomy, others have an intact uterus associated with their BSO, so progestins must be used along with estrogen. And if we are using higher-than-standard doses of estrogen, we also need to use higher-than-standard doses of progestin.
In that classic 53-year-old woman I referred to who had spontaneous normal menopause, if she is taking 1 mg of estradiol daily, or a 0.05-mg patch, or 0.625 mg of conjugated equine estrogen, 2.5 mg of medroxyprogesterone is fine. In fact, that showed excellent progestational protection of the endometrium in the Women’s Health Initiative and in other studies.
However, if we are going to use double the estrogen dose, we should increase the progestin dose too. In some of my patients on higher estrogen doses who have an intact uterus, I’ll use 5 or even 10 mg of daily medroxyprogesterone acetate to ensure adequate progestational suppression.
Dr. Faubion: Another practical tip is that if one is using conjugated equine estrogens, measuring the serum estradiol levels is not useful either.
Dr. Kaunitz: I agree.
Continue to: Oral contraceptives as replacement HT...
Oral contraceptives as replacement HT
Dr. Faubion: Would you comment on use of a birth control pill in this circumstance? Would it be optimal to use a postmenopausal HT regimen as opposed to a birth control pill or combined hormonal contraception?
Dr. Kapoor: In this younger population, sometimes it seems like a more socially acceptable decision to be on a birth control option than on menopausal HT. But there are some issues with being on a contraceptive regimen. One is that we end up using estrogen doses much higher than what is really needed for replacement purposes. It is also a nonphysiologic way of replacement in another sense—as opposed to estradiol, which is the main hormone made by the ovaries, the hormonal contraceptive regimens contain the synthetic estrogen ethinyl estradiol for the most part.
The other issue that is based on some weak evidence is that it appears that the bone health outcomes are probably inferior with combined hormonal contraception. For these reasons, regimens that are based on replacement doses of estradiol are preferred.
Dr. Faubion: Right, although the data are somewhat weak, I agree that thus far it seems optimal to utilize a postmenopausal regimen for various reasons. Dr. Kaunitz, anything to add?
Dr. Kaunitz: Yes, to underscore Dr. Kapoor’s point, a common oral contraceptive that contains 20 µg of ethinyl estradiol is substantially more estrogenic than 1.0 or 2.0 mg of micronized oral estradiol.
Also consider that a 20-µg ethinyl estradiol oral contraceptive may increase the risk of venous thromboembolism more than menopausal doses of oral estradiol, whether it be a micronized estradiol or conjugated equine estrogen.
Dr. Faubion: So the risk may be greater with oral combined hormonal contraception as well?
Dr. Kaunitz: One thing we can do is explain to our patients that their ovaries, prior to surgery or prior to induced menopause, were making substantial quantities of estradiol. Whether we prescribe a patch or oral micronized estradiol, this estrogen is identical to the hormone that their ovaries were making prior to surgery or induced menopause.
Breast cancer concerns
Dr. Faubion: Let’s consider a more complicated case. A 35-year-old woman has an identified BRCA1 mutation; she has not had any cancers but has undergone risk-reducing BSO and her uterus remains. Is this woman a candidate for HT? At what dose, and for how long? Dr. Kaunitz, why don’t you start.
Dr. Kaunitz: That is a challenging case but one that I think our readers will find interesting and maybe even provocative.
We know that women with BRCA1 mutations, the more common of the 2 BRCA mutations, have a very high risk of developing epithelial ovarian cancer at a young age. For this reason, our colleagues in medical oncology who specialize in hereditary ovarian/breast cancer syndromes recommend prophylactic risk-reducing—and I would also say lifesaving—BSO with or without hysterectomy for women with BRCA1 mutations.
However, over the years there has been tremendous reluctance among physicians caring for BRCA patients and the women themselves—I use the term “previvors” to describe BRCA carriers who have not been diagnosed with breast or ovarian cancer—to use HT after BSO because of concerns that HT might increase breast cancer risk in women who are already at high risk for breast cancer.
I assume, Dr. Faubion, that in this case the woman had gynecologic surgery but continues to have intact breasts. Is that correct?
Dr. Faubion: That is correct.
Dr. Kaunitz: Although the assumption has been that it is not safe to prescribe HT in this setting, in fact, the reported cohort studies that have looked at this issue have not found an elevated risk of breast cancer when replacement estrogen, with or without progestin, is prescribed to BRCA1 previvors with intact breasts.
Given what Dr. Kapoor said regarding the morbidity that is associated with BSO without replacement of physiologic estrogen, and also given the severe symptoms that so many of these young menopausal women experience, in my practice I do prescribe estrogen or estrogen-progestin therapy and focus on the higher target doses that we discussed for the earlier case patient who had a hysterectomy for abnormal uterine bleeding with adenomyosis.
Dr. Faubion: Dr. Kapoor, do you agree with this approach? How long would you continue therapy?
Dr. Kapoor: First, in this BRCA1 case we need to appreciate that the indication for the BSO is a legitimate one, in contrast to the first case in which the ovaries were removed in a patient whose average risk of ovarian cancer was low. It is important to recognize that surgery performed in this context is the right thing to do because it does significantly reduce the risk of ovarian cancer.
The second thing to appreciate is that while we reduce the risk of ovarian cancer significantly and make sure that these patients survive longer, it’s striking a fine balance in that you want to make sure that their morbidity is not increased as a result of premature estrogen deprivation.
As Dr. Kaunitz told us, the evidence that we have so far, which granted is not very robust but is fairly strong observational evidence, suggests that the risk of breast cancer is not elevated when these patients are treated with replacement doses of HT.
Having said that, I do have very strong discussions with my patients in this category about having the risk-reducing bilateral mastectomy also, because if they were to get breast cancer because of their increased genetic predisposition, the cancer is likely to grow faster if the patient is on HT. So one of my counseling points to patients is that they strongly consider bilateral mastectomy, which reduces their breast cancer risk by more than 90%. At the same time, I also strongly endorse using HT in replacement doses for the reasons that we have already stated.
Dr. Faubion: Continue HT until age 50 or 52?
Dr. Kapoor: Definitely until that age, and possibly longer, depending on their symptoms. The indications for treating beyond the age of natural menopause are much the same as for women who experience natural menopause.
Dr. Faubion: That is assuming they had a bilateral mastectomy?
Dr. Kapoor: Yes.
Continue to: Continuing HT until the age of natural menopause...
Continuing HT until the age of natural menopause
Dr. Kaunitz: Dr. Kapoor brings up the important point of duration of systemic HT. I agree that similar considerations apply both to the healthy 41-year-old who had a hysterectomy for abnormal uterine bleeding and to the 35-year-old who had risk-reducing surgery because of her BRCA1 mutation.
In the 2 cases, both to treat symptoms and to prevent chronic diseases, it makes sense to continue HT at least until the age of natural menopause. That is consistent with 2017 guidance from The North American Menopause Society (NAMS) position statement on the use of systemic HT, that is, continuing systemic HT at least until the age of natural menopause.3 Then at that point, continuing or discontinuing systemic HT becomes discretionary, and that would be true for both cases. If the patient is slender or has a strong family history of osteoporosis, that tends to push the patient more in terms of continuing systemic HT. Those are just some examples, and Dr. Kapoor may want to detail other relevant considerations.
Dr. Kapoor: I completely agree. The decision is driven by symptoms that are not otherwise well managed, for example, with nonhormone strategies. If we have any concerns utilizing HT beyond the age of natural menopause, then nonhormonal options can be considered; but sometimes those are not as effective. And bone health is very important. You want to avoid using bisphosphonates in younger women and reserve them for older patients in their late 60s and 70s. Hormone therapy use is a very reasonable strategy to prevent bone loss.
Dr. Kaunitz: It is also worth mentioning that sometimes the woman involved in shared decision making with her clinician decides to stop systemic HT. In that setting, should the patient start developing new-onset dyspareunia, vaginal dryness, or other genital or sexuality-related concerns, it takes very little for me to advise that she start low-dose local vaginal estrogen therapy.
Dr. Faubion: In either scenario, if a woman were to develop symptoms consistent with genitourinary syndrome of menopause (GSM), would you use vaginal estrogen in addition to the systemic estrogen or alone after the woman elected to discontinue systemic therapy?
Dr. Kapoor: Yes to both, I would say.
Dr. Kaunitz: As my patients using systemic HT age, often I will lower the dose. For instance, the dose I use in a 53-year-old will be higher than when she is 59 or 62. At the same time, as we lower the dose of systemic estrogen therapy, symptoms of vaginal atrophy or GSM often will appear, and these can be effectively treated by adding low-dose vaginal estrogen therapy. A number of my patients, particularly those who are on lower-than-standard doses of systemic HT, are also using low-dose vaginal estrogen therapy.
There is a “hybrid” product available: the 90-day estradiol vaginal ring. Estring is a low-dose, 2-mg, 90-day estradiol ring that is very useful, but it is effective only for treating GSM or vaginal atrophy. A second menopausal vaginal estradiol ring, Femring, is available in 2 doses: 0.05 mg/day and 0.1 mg/day. These are very effective in treating both systemic issues, such as vasomotor symptoms or prevention of osteoporosis, and very effective in treating GSM or vaginal atrophy. One problem is that Femring, depending on insurance coverage, can be very expensive. It’s not available as a generic, so for insurance or financial reasons I don’t often prescribe it. If I could remove those financial barriers, I would prescribe Femring more often because it is very useful.
Dr. Faubion: You raise an important point, and that is, for women who have been on HT for some time, clinicians often feel the need to slowly reduce the dose. Would you do that same thing, Dr. Kapoor, for a 40-year-old woman? Would you reduce the dose as she approaches age 50? Is there pressure that “she shouldn’t be on that much estrogen”?
Dr. Kapoor: No, I would not feel pressured until the patient turns at least 46. I bring up age 46 because the average age range for menopause is 46 to 55. After that, if there is any concern, we can decrease the dose to half and keep the patient on that until she turns 50 or 51. But most of my patients are on replacement doses until the average age of menopause, which is around 51 years, and that’s when you reduce the dose to that of the typical HT regimens used after natural menopause.
Sometimes patients are told something by a friend or they have read something and they worry about the risk of 2 things. One is breast cancer and the other is venous thromboembolism (VTE), and that may be why they want to be on a lower dose. I counsel patients that while the risk of VTE is real with HT, it is the women after natural menopause who are at risk—because age itself is a risk for VTE—and it also has to do with the kind of HT regimen that a patient is on. High doses of oral estrogens and certain progestogens increase the risk. But again, for estradiol used in replacement doses and the more common progestogens that we now use in practice, such as micronized progesterone, the risk is not the same. The same goes for breast cancer. My biggest message to patients and clinicians who take care of these patients is that the rules that apply to women after natural menopause just do not apply to this very different patient population.
Dr. Faubion: Thank you, Dr. Kaunitz and Dr. Kapoor, for sharing your knowledge and experience. ●
Systemic HT past the age of 65
Dr. Kaunitz: Another practical issue relates to long-term or extended use of systemic HT. It’s not infrequent in my practice to receive mail and faxes from insurance carriers of systemic HT users who are age 65 and older in which the company refers to the American Geriatrics Society’s Beers criteria for potentially inappropriate medication use in older adults,1 suggesting that systemic HT is inappropriate for all women over age 65. In this age group, I use lower doses if I am continuing systemic HT. But the good news is that both NAMS and the American College Obstetricians and Gynecologists indicate that arbitrarily stopping systemic HT at age 65 or for any other arbitrary reason is inappropriate, and that decisions about continuing or discontinuing therapy should be made on an individualized basis using shared decision making. That’s an important message for our readers.
Counseling regarding elective BSO
Dr. Faubion: One final note about elective BSO in the absence of a genetic mutation that predisposes to increased ovarian or breast cancer risk. Fortunately, we have seen rates of oophorectomy before the age of natural menopause decline, but what would your advice be to women or clinicians of these women who say they are “just afraid of ovarian cancer and would like to have their ovaries removed before the age of natural menopause”?
Dr. Kaunitz: If patients have increased anxiety about ovarian cancer and yet they themselves are not known to be at elevated risk, I emphasize that, fortunately, ovarian cancer is uncommon. It is much less common than other cancers the patient might be familiar with, such as breast or colon or lung cancer. I also emphasize that women who have given birth, particularly multiple times; women who nursed their infants; and women who have used combination hormonal contraceptives, particularly if long term, are at markedly lower risk for ovarian cancer as they get older. We are talking about an uncommon cancer that is even less common if women have given birth, nursed their infants, or used combination contraceptives long term.
Dr. Faubion: Dr. Kapoor, what would you say regarding the increased risk they might incur if they do have their ovaries out?
Dr. Kapoor: As Dr. Kaunitz said, this is an uncommon cancer, and pursuing something to reduce the risk of an uncommon cancer does not benefit the community. That is also my counseling point to patients.
I also talk to them extensively about the risk associated with the ovaries being removed, and I tell them that although we have the option of giving them HT, it is hard to replicate the magic of nature. No matter what concoction or regimen we use, we cannot ensure reinstating health to what it was in the premenopausal state, because estrogen has such myriad effects on the body in so many different organ systems.
Reference
1. American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American Geriatrics Society 2015 updated Beers criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2015;63:2227-2246.
- Kaunitz AM, Kapoor E, Faubion S. Treatment of women after bilateral salpingo-oophorectomy performed prior to natural menopause. JAMA. 2021;326:1429-1430.
- Writing Group for the Women’s Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321-333.
- North American Menopause Society. The 2017 hormone therapy position statement of The North American Menopause Society. J North Am Menopause Soc. 2017;24: 728-753.

Women who undergo bilateral salpingo-oophorectomy (BSO) for various indications prior to menopause experience a rapid decline in ovarian hormone levels and consequent vasomotor and other menopausal symptoms. In addition, the resulting estrogen deprivation is associated with such long-term adverse outcomes as osteoporosis and cardiovascular morbidity.
OBG M
Surgical vs natural menopause
Stephanie Faubion, MD, MBA, NCMP: Since the Women’s Health Initiative study was published in 2002,2 many clinicians have been fearful of using systemic HT in menopausal women, and HT use has declined dramatically such that only about 4% to 6% of menopausal women are now receiving systemic HT. Importantly, however, a group of younger menopausal women also are not receiving HT, and that is women who undergo BSO before they reach the average age of menopause, which in the United States is about age 52; this is sometimes referred to as surgical menopause or early surgical menopause. Early surgical menopause has different connotations for long-term health risks than natural menopause at the average age, and we are here to discuss these health effects and their management.
My name is Stephanie Faubion, and I am a women’s health internist and the Chair of the Department of Medicine at Mayo Clinic in Jacksonville, Florida, and Director of Mayo Clinic Women’s Health. I am here with 2 of my esteemed colleagues, Dr. Andrew Kaunitz and Dr. Ekta Kapoor.
Andrew M. Kaunitz, MD, NCMP: Hello, I am an ObGyn with the University of Florida College of Medicine in Jacksonville, with particular interests in contraception, menopause, and gynecologic ultrasonography.
Ekta Kapoor, MBBS, NCMP: And I am an endocrinologist at Mayo Clinic in Rochester with a specific interest in menopause and hormone therapy. I am also the Assistant Director for Mayo Clinic Women’s Health.
Higher-than-standard estrogen doses needed in younger menopausal women
Dr. Faubion: Let’s consider a couple of cases so that we can illustrate some important points regarding hormone management in women who have undergone BSO before the age of natural menopause.
Our first case patient is a woman who is 41 years of age and, because of adenomyosis, she will undergo a hysterectomy. She tells her clinician that she is very concerned about ovarian cancer risk because one of her good friends recently was diagnosed with ovarian cancer, and together they decide to remove her ovaries at the time of hysterectomy. Notably, her ovaries were healthy.
The patient is now menopausal postsurgery, and she is having significant hot flashes and night sweats. She visits her local internist, who is concerned about initiating HT. She is otherwise a healthy woman and does not have any contraindications to HT. Dr. Kaunitz, what would you tell her internist?
Dr. Kaunitz: We are dealing with 2 different issues in terms of decision making about systemic HT for this 41-year-old who has undergone BSO. First, as you mentioned, Dr. Faubion, she has bothersome hot flashes, or vasomotor symptoms. Unless there are contraindications, systemic HT would be appropriate. Although I might start treatment at standard doses, and the accompanying TABLE depicts standard doses for the 2 most common oral estrogen formulations as well as transdermal estradiol, it’s important to recognize that younger menopausal women often will need to use higher-than-standard doses.

For example, for a 53-year-old woman who has been menopausal for a year or 2 and now has bothersome symptoms, I might start her on estradiol 1 mg tablets with progestin if a uterus is present. However, in this 41-year-old case patient, while I might start treatment at a standard dose, I would anticipate increasing to higher doses, such as 1.5 or 2 mg of daily estradiol until she feels her menopausal symptoms are adequately addressed.
Dr. Faubion: It is important to note that sometimes women with early BSO tend to have more severe vasomotor symptoms. Do you find that sometimes a higher dose is required just to manage symptoms, Dr. Kaunitz?
Dr. Kaunitz: Absolutely, yes. The decision whether or not to use systemic HT might be considered discretionary or elective in the classic 53-year-old woman recently menopausal with hot flashes, a so-called spontaneously or naturally menopausal woman. But my perspective is that unless there are clear contraindications, the decision to start systemic HT in the 41-year-old BSO case patient is actually not discretionary. Unless contraindications are present, it is important not only to treat symptoms but also to prevent an array of chronic major health concerns that are more likely if we don’t prescribe systemic HT.
Continue to: Health effects of not using HT...
Health effects of not using HT
Dr. Faubion: Dr. Kapoor, can you describe the potential long-term adverse health consequences of not using estrogen therapy? Say the same 41-year-old woman does not have many bothersome symptoms. What would you do?
Dr. Kapoor: Thank you for that important question. Building on what Dr. Kaunitz said, in these patients there are really 2 issues that can seem to be independent but are not: The first relates to the immediate consequences of lack of estrogen, ie, the menopause-related symptoms, but the second and perhaps the bigger issue is the long-term risk associated with estrogen deprivation.
The symptoms in these women are often obvious as they can be quite severe and abrupt; one day these women have normal hormone levels and the next day, after BSO, suddenly their hormones are very low. So if symptoms occur, they are usually hard to miss, simply because they are very drastic and very severe.
Historically, patients and their clinicians have targeted these symptoms. Patients experience menopausal symptoms, they seek treatment, and then the clinicians basically titrate the treatment to manage these symptoms. That misses the bigger issue, however, which is that premature estrogen deprivation leads to a host of chronic health conditions, as Dr. Kaunitz mentioned. These mainly include increased risk for cardiovascular disease, diabetes, hypertension, dyslipidemia, increased risk of mortality, dementia, and osteoporosis.
Fairly strong observational evidence suggests that use of estrogen therapy given in replacement doses—doses higher than those typically used in women after natural menopause, therefore considered replacement doses—helps mitigate the risk of some of these adverse health conditions.
In these women, the bigger goal really is to reinstate the hormonal milieu that exists prior to menopause. To your point, Dr. Faubion, if I have a patient who is younger than 46 years, who has her ovaries taken out, and even if she has zero symptoms (and sometimes that does happen), I would still make a case for this patient to utilize hormone therapy unless there is a contraindication such as breast cancer or other estrogen-sensitive cancers.
Dr. Faubion: Again, would you aim for those higher doses rather than treat with the “lowest dose”?
Dr. Kapoor: Absolutely. My punchline to the patients and clinicians in these discussions is that the rules of the game are different for these women. We cannot extrapolate the risks and benefits of HT use in women after natural menopause to younger women who have surgical menopause. Those rules just do not apply with respect to both benefits and risks.
Dr. Faubion: I think it’s important to say that these same “rules” would apply if the women were to go through premature menopause for any other reason, too, such as chemotherapy, radiation therapy, or premature ovarian insufficiency for any number of reasons, including toxic, metabolic, or genetic causes and so on. Would that be true?
Dr. Kapoor: Yes, absolutely so.
Dr. Faubion: Dr. Kaunitz, do you want to add anything?
Dr. Kaunitz: In terms of practical or clinical issues regarding systemic HT management, for the woman in her early 50s who has experienced normal or natural spontaneous menopause, a starting dose of transdermal estradiol would be, for instance, a 0.05-mg patch, which is a patch that over 24 hours releases 0.05 mg of estradiol daily; or standard oral estrogen, including conjugated equine estrogen, a 0.625-mg tablet daily, or estradiol, a 1-mg tablet daily.
But in younger patients, we want to use higher doses. For a patch, for instance, I would aim for a 0.075- or 0.1-mg estradiol patch, which releases a higher daily dose of estradiol than the standard dose. For oral estrogen, the dose would be 0.9- or even 1.25-mg tablets of conjugated equine estrogen or 1.5 mg, which is a 1-mg plus a 0.5-mg estradiol tablet, or a 2-mg estradiol tablet. Estradiol does come in a 2-mg strength.
For oral estrogen, I prefer estradiol because it’s available as a generic medication and often available at a very low cost, sometimes as low as $4 a month from chain pharmacies.
Continue to: Usefulness of monitoring estradiol levels for dosage adjustment...
Usefulness of monitoring estradiol levels for dosage adjustment
Dr. Faubion: That’s a great point, and again it is important to emphasize that we are aiming to recreate the premenopausal hormonal milieu. If you were to check estradiol levels, that would be aiming for a premenopausal range of approximately 80 to 120 pg per mL. Dr. Kapoor, is there utility in monitoring estrogen levels?
Dr. Kapoor: Great question, Dr. Faubion, and as you know it’s a loaded one. We base this on empiric evidence. We know that if the hormonal milieu in a young patient is changed to a postmenopausal one, her risk for many chronic conditions is increased. So if we were to reinstate a premenopausal hormonal milieu, that risk would probably be reduced. It makes good sense to target an empiric goal of 80 to 120 pg per mL of estradiol, which is the average estradiol level in a premenopausal woman. If you were to ask me, however, are there randomized, controlled trial data to support this practice—that is, if you target that level, can you make sure that the risk of diabetes is lower or that the risk of heart disease is lower—that study has yet to be done, and it may not ever be done on a large scale. However, it intuitively makes good sense to target premenopausal estradiol levels.
Dr. Faubion: When might you check an estradiol level in this population? For example, if you are treating a patient with a 0.1-mg estradiol patch and she still has significant hot flashes, would it be useful to check the level?
Dr. Kapoor: It would. In my practice, I check estradiol levels on these patients on an annual basis, regardless of symptoms, but definitely in the patient who has symptoms. It makes good sense, because sometimes these patients don’t absorb the estrogen well, particularly if administered by the transdermal route.
A general rule of thumb is that in the average population, if a patient is on the 0.1-mg patch, for example, you would expect her level to be around 100. If it is much lower than that, which sometimes happens, that speaks for poor absorption. Options at that point would be to treat her with a higher dose patch, depending on what the level is, or switch to a different formulation, such as oral.
In instances in which I have treated patients with a 0.1-mg patch for example, and their estradiol levels are undetectable, that speaks for very poor absorption. For such patients I make a case for switching them to oral therapy. Most definitely that makes sense in a patient who is symptomatic despite treatment. But even for patients who don’t have symptoms, I like to target that level, acknowledging that there is no evidence as such to support this practice.
Dr. Faubion: Dr. Kaunitz, do you want to add anything?
Dr. Kaunitz: Yes, a few practical points. Although patches are available in a wider array of doses than oral estrogen formulations, the highest dose available is 0.1 mg. It’s important for clinicians to recognize that while checking serum levels when indicated can be performed in women using transdermal estradiol or patches, in women who are using oral estrogen, checking blood levels is not going to work well because serum estrogen levels have a daily peak and valley in women who use oral versus transdermal estradiol.
I also wanted to talk about progestins. Although many patients who have had a BSO prior to spontaneous menopause also have had a hysterectomy, others have an intact uterus associated with their BSO, so progestins must be used along with estrogen. And if we are using higher-than-standard doses of estrogen, we also need to use higher-than-standard doses of progestin.
In that classic 53-year-old woman I referred to who had spontaneous normal menopause, if she is taking 1 mg of estradiol daily, or a 0.05-mg patch, or 0.625 mg of conjugated equine estrogen, 2.5 mg of medroxyprogesterone is fine. In fact, that showed excellent progestational protection of the endometrium in the Women’s Health Initiative and in other studies.
However, if we are going to use double the estrogen dose, we should increase the progestin dose too. In some of my patients on higher estrogen doses who have an intact uterus, I’ll use 5 or even 10 mg of daily medroxyprogesterone acetate to ensure adequate progestational suppression.
Dr. Faubion: Another practical tip is that if one is using conjugated equine estrogens, measuring the serum estradiol levels is not useful either.
Dr. Kaunitz: I agree.
Continue to: Oral contraceptives as replacement HT...
Oral contraceptives as replacement HT
Dr. Faubion: Would you comment on use of a birth control pill in this circumstance? Would it be optimal to use a postmenopausal HT regimen as opposed to a birth control pill or combined hormonal contraception?
Dr. Kapoor: In this younger population, sometimes it seems like a more socially acceptable decision to be on a birth control option than on menopausal HT. But there are some issues with being on a contraceptive regimen. One is that we end up using estrogen doses much higher than what is really needed for replacement purposes. It is also a nonphysiologic way of replacement in another sense—as opposed to estradiol, which is the main hormone made by the ovaries, the hormonal contraceptive regimens contain the synthetic estrogen ethinyl estradiol for the most part.
The other issue that is based on some weak evidence is that it appears that the bone health outcomes are probably inferior with combined hormonal contraception. For these reasons, regimens that are based on replacement doses of estradiol are preferred.
Dr. Faubion: Right, although the data are somewhat weak, I agree that thus far it seems optimal to utilize a postmenopausal regimen for various reasons. Dr. Kaunitz, anything to add?
Dr. Kaunitz: Yes, to underscore Dr. Kapoor’s point, a common oral contraceptive that contains 20 µg of ethinyl estradiol is substantially more estrogenic than 1.0 or 2.0 mg of micronized oral estradiol.
Also consider that a 20-µg ethinyl estradiol oral contraceptive may increase the risk of venous thromboembolism more than menopausal doses of oral estradiol, whether it be a micronized estradiol or conjugated equine estrogen.
Dr. Faubion: So the risk may be greater with oral combined hormonal contraception as well?
Dr. Kaunitz: One thing we can do is explain to our patients that their ovaries, prior to surgery or prior to induced menopause, were making substantial quantities of estradiol. Whether we prescribe a patch or oral micronized estradiol, this estrogen is identical to the hormone that their ovaries were making prior to surgery or induced menopause.
Breast cancer concerns
Dr. Faubion: Let’s consider a more complicated case. A 35-year-old woman has an identified BRCA1 mutation; she has not had any cancers but has undergone risk-reducing BSO and her uterus remains. Is this woman a candidate for HT? At what dose, and for how long? Dr. Kaunitz, why don’t you start.
Dr. Kaunitz: That is a challenging case but one that I think our readers will find interesting and maybe even provocative.
We know that women with BRCA1 mutations, the more common of the 2 BRCA mutations, have a very high risk of developing epithelial ovarian cancer at a young age. For this reason, our colleagues in medical oncology who specialize in hereditary ovarian/breast cancer syndromes recommend prophylactic risk-reducing—and I would also say lifesaving—BSO with or without hysterectomy for women with BRCA1 mutations.
However, over the years there has been tremendous reluctance among physicians caring for BRCA patients and the women themselves—I use the term “previvors” to describe BRCA carriers who have not been diagnosed with breast or ovarian cancer—to use HT after BSO because of concerns that HT might increase breast cancer risk in women who are already at high risk for breast cancer.
I assume, Dr. Faubion, that in this case the woman had gynecologic surgery but continues to have intact breasts. Is that correct?
Dr. Faubion: That is correct.
Dr. Kaunitz: Although the assumption has been that it is not safe to prescribe HT in this setting, in fact, the reported cohort studies that have looked at this issue have not found an elevated risk of breast cancer when replacement estrogen, with or without progestin, is prescribed to BRCA1 previvors with intact breasts.
Given what Dr. Kapoor said regarding the morbidity that is associated with BSO without replacement of physiologic estrogen, and also given the severe symptoms that so many of these young menopausal women experience, in my practice I do prescribe estrogen or estrogen-progestin therapy and focus on the higher target doses that we discussed for the earlier case patient who had a hysterectomy for abnormal uterine bleeding with adenomyosis.
Dr. Faubion: Dr. Kapoor, do you agree with this approach? How long would you continue therapy?
Dr. Kapoor: First, in this BRCA1 case we need to appreciate that the indication for the BSO is a legitimate one, in contrast to the first case in which the ovaries were removed in a patient whose average risk of ovarian cancer was low. It is important to recognize that surgery performed in this context is the right thing to do because it does significantly reduce the risk of ovarian cancer.
The second thing to appreciate is that while we reduce the risk of ovarian cancer significantly and make sure that these patients survive longer, it’s striking a fine balance in that you want to make sure that their morbidity is not increased as a result of premature estrogen deprivation.
As Dr. Kaunitz told us, the evidence that we have so far, which granted is not very robust but is fairly strong observational evidence, suggests that the risk of breast cancer is not elevated when these patients are treated with replacement doses of HT.
Having said that, I do have very strong discussions with my patients in this category about having the risk-reducing bilateral mastectomy also, because if they were to get breast cancer because of their increased genetic predisposition, the cancer is likely to grow faster if the patient is on HT. So one of my counseling points to patients is that they strongly consider bilateral mastectomy, which reduces their breast cancer risk by more than 90%. At the same time, I also strongly endorse using HT in replacement doses for the reasons that we have already stated.
Dr. Faubion: Continue HT until age 50 or 52?
Dr. Kapoor: Definitely until that age, and possibly longer, depending on their symptoms. The indications for treating beyond the age of natural menopause are much the same as for women who experience natural menopause.
Dr. Faubion: That is assuming they had a bilateral mastectomy?
Dr. Kapoor: Yes.
Continue to: Continuing HT until the age of natural menopause...
Continuing HT until the age of natural menopause
Dr. Kaunitz: Dr. Kapoor brings up the important point of duration of systemic HT. I agree that similar considerations apply both to the healthy 41-year-old who had a hysterectomy for abnormal uterine bleeding and to the 35-year-old who had risk-reducing surgery because of her BRCA1 mutation.
In the 2 cases, both to treat symptoms and to prevent chronic diseases, it makes sense to continue HT at least until the age of natural menopause. That is consistent with 2017 guidance from The North American Menopause Society (NAMS) position statement on the use of systemic HT, that is, continuing systemic HT at least until the age of natural menopause.3 Then at that point, continuing or discontinuing systemic HT becomes discretionary, and that would be true for both cases. If the patient is slender or has a strong family history of osteoporosis, that tends to push the patient more in terms of continuing systemic HT. Those are just some examples, and Dr. Kapoor may want to detail other relevant considerations.
Dr. Kapoor: I completely agree. The decision is driven by symptoms that are not otherwise well managed, for example, with nonhormone strategies. If we have any concerns utilizing HT beyond the age of natural menopause, then nonhormonal options can be considered; but sometimes those are not as effective. And bone health is very important. You want to avoid using bisphosphonates in younger women and reserve them for older patients in their late 60s and 70s. Hormone therapy use is a very reasonable strategy to prevent bone loss.
Dr. Kaunitz: It is also worth mentioning that sometimes the woman involved in shared decision making with her clinician decides to stop systemic HT. In that setting, should the patient start developing new-onset dyspareunia, vaginal dryness, or other genital or sexuality-related concerns, it takes very little for me to advise that she start low-dose local vaginal estrogen therapy.
Dr. Faubion: In either scenario, if a woman were to develop symptoms consistent with genitourinary syndrome of menopause (GSM), would you use vaginal estrogen in addition to the systemic estrogen or alone after the woman elected to discontinue systemic therapy?
Dr. Kapoor: Yes to both, I would say.
Dr. Kaunitz: As my patients using systemic HT age, often I will lower the dose. For instance, the dose I use in a 53-year-old will be higher than when she is 59 or 62. At the same time, as we lower the dose of systemic estrogen therapy, symptoms of vaginal atrophy or GSM often will appear, and these can be effectively treated by adding low-dose vaginal estrogen therapy. A number of my patients, particularly those who are on lower-than-standard doses of systemic HT, are also using low-dose vaginal estrogen therapy.
There is a “hybrid” product available: the 90-day estradiol vaginal ring. Estring is a low-dose, 2-mg, 90-day estradiol ring that is very useful, but it is effective only for treating GSM or vaginal atrophy. A second menopausal vaginal estradiol ring, Femring, is available in 2 doses: 0.05 mg/day and 0.1 mg/day. These are very effective in treating both systemic issues, such as vasomotor symptoms or prevention of osteoporosis, and very effective in treating GSM or vaginal atrophy. One problem is that Femring, depending on insurance coverage, can be very expensive. It’s not available as a generic, so for insurance or financial reasons I don’t often prescribe it. If I could remove those financial barriers, I would prescribe Femring more often because it is very useful.
Dr. Faubion: You raise an important point, and that is, for women who have been on HT for some time, clinicians often feel the need to slowly reduce the dose. Would you do that same thing, Dr. Kapoor, for a 40-year-old woman? Would you reduce the dose as she approaches age 50? Is there pressure that “she shouldn’t be on that much estrogen”?
Dr. Kapoor: No, I would not feel pressured until the patient turns at least 46. I bring up age 46 because the average age range for menopause is 46 to 55. After that, if there is any concern, we can decrease the dose to half and keep the patient on that until she turns 50 or 51. But most of my patients are on replacement doses until the average age of menopause, which is around 51 years, and that’s when you reduce the dose to that of the typical HT regimens used after natural menopause.
Sometimes patients are told something by a friend or they have read something and they worry about the risk of 2 things. One is breast cancer and the other is venous thromboembolism (VTE), and that may be why they want to be on a lower dose. I counsel patients that while the risk of VTE is real with HT, it is the women after natural menopause who are at risk—because age itself is a risk for VTE—and it also has to do with the kind of HT regimen that a patient is on. High doses of oral estrogens and certain progestogens increase the risk. But again, for estradiol used in replacement doses and the more common progestogens that we now use in practice, such as micronized progesterone, the risk is not the same. The same goes for breast cancer. My biggest message to patients and clinicians who take care of these patients is that the rules that apply to women after natural menopause just do not apply to this very different patient population.
Dr. Faubion: Thank you, Dr. Kaunitz and Dr. Kapoor, for sharing your knowledge and experience. ●
Systemic HT past the age of 65
Dr. Kaunitz: Another practical issue relates to long-term or extended use of systemic HT. It’s not infrequent in my practice to receive mail and faxes from insurance carriers of systemic HT users who are age 65 and older in which the company refers to the American Geriatrics Society’s Beers criteria for potentially inappropriate medication use in older adults,1 suggesting that systemic HT is inappropriate for all women over age 65. In this age group, I use lower doses if I am continuing systemic HT. But the good news is that both NAMS and the American College Obstetricians and Gynecologists indicate that arbitrarily stopping systemic HT at age 65 or for any other arbitrary reason is inappropriate, and that decisions about continuing or discontinuing therapy should be made on an individualized basis using shared decision making. That’s an important message for our readers.
Counseling regarding elective BSO
Dr. Faubion: One final note about elective BSO in the absence of a genetic mutation that predisposes to increased ovarian or breast cancer risk. Fortunately, we have seen rates of oophorectomy before the age of natural menopause decline, but what would your advice be to women or clinicians of these women who say they are “just afraid of ovarian cancer and would like to have their ovaries removed before the age of natural menopause”?
Dr. Kaunitz: If patients have increased anxiety about ovarian cancer and yet they themselves are not known to be at elevated risk, I emphasize that, fortunately, ovarian cancer is uncommon. It is much less common than other cancers the patient might be familiar with, such as breast or colon or lung cancer. I also emphasize that women who have given birth, particularly multiple times; women who nursed their infants; and women who have used combination hormonal contraceptives, particularly if long term, are at markedly lower risk for ovarian cancer as they get older. We are talking about an uncommon cancer that is even less common if women have given birth, nursed their infants, or used combination contraceptives long term.
Dr. Faubion: Dr. Kapoor, what would you say regarding the increased risk they might incur if they do have their ovaries out?
Dr. Kapoor: As Dr. Kaunitz said, this is an uncommon cancer, and pursuing something to reduce the risk of an uncommon cancer does not benefit the community. That is also my counseling point to patients.
I also talk to them extensively about the risk associated with the ovaries being removed, and I tell them that although we have the option of giving them HT, it is hard to replicate the magic of nature. No matter what concoction or regimen we use, we cannot ensure reinstating health to what it was in the premenopausal state, because estrogen has such myriad effects on the body in so many different organ systems.
Reference
1. American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American Geriatrics Society 2015 updated Beers criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2015;63:2227-2246.

Women who undergo bilateral salpingo-oophorectomy (BSO) for various indications prior to menopause experience a rapid decline in ovarian hormone levels and consequent vasomotor and other menopausal symptoms. In addition, the resulting estrogen deprivation is associated with such long-term adverse outcomes as osteoporosis and cardiovascular morbidity.
OBG M
Surgical vs natural menopause
Stephanie Faubion, MD, MBA, NCMP: Since the Women’s Health Initiative study was published in 2002,2 many clinicians have been fearful of using systemic HT in menopausal women, and HT use has declined dramatically such that only about 4% to 6% of menopausal women are now receiving systemic HT. Importantly, however, a group of younger menopausal women also are not receiving HT, and that is women who undergo BSO before they reach the average age of menopause, which in the United States is about age 52; this is sometimes referred to as surgical menopause or early surgical menopause. Early surgical menopause has different connotations for long-term health risks than natural menopause at the average age, and we are here to discuss these health effects and their management.
My name is Stephanie Faubion, and I am a women’s health internist and the Chair of the Department of Medicine at Mayo Clinic in Jacksonville, Florida, and Director of Mayo Clinic Women’s Health. I am here with 2 of my esteemed colleagues, Dr. Andrew Kaunitz and Dr. Ekta Kapoor.
Andrew M. Kaunitz, MD, NCMP: Hello, I am an ObGyn with the University of Florida College of Medicine in Jacksonville, with particular interests in contraception, menopause, and gynecologic ultrasonography.
Ekta Kapoor, MBBS, NCMP: And I am an endocrinologist at Mayo Clinic in Rochester with a specific interest in menopause and hormone therapy. I am also the Assistant Director for Mayo Clinic Women’s Health.
Higher-than-standard estrogen doses needed in younger menopausal women
Dr. Faubion: Let’s consider a couple of cases so that we can illustrate some important points regarding hormone management in women who have undergone BSO before the age of natural menopause.
Our first case patient is a woman who is 41 years of age and, because of adenomyosis, she will undergo a hysterectomy. She tells her clinician that she is very concerned about ovarian cancer risk because one of her good friends recently was diagnosed with ovarian cancer, and together they decide to remove her ovaries at the time of hysterectomy. Notably, her ovaries were healthy.
The patient is now menopausal postsurgery, and she is having significant hot flashes and night sweats. She visits her local internist, who is concerned about initiating HT. She is otherwise a healthy woman and does not have any contraindications to HT. Dr. Kaunitz, what would you tell her internist?
Dr. Kaunitz: We are dealing with 2 different issues in terms of decision making about systemic HT for this 41-year-old who has undergone BSO. First, as you mentioned, Dr. Faubion, she has bothersome hot flashes, or vasomotor symptoms. Unless there are contraindications, systemic HT would be appropriate. Although I might start treatment at standard doses, and the accompanying TABLE depicts standard doses for the 2 most common oral estrogen formulations as well as transdermal estradiol, it’s important to recognize that younger menopausal women often will need to use higher-than-standard doses.

For example, for a 53-year-old woman who has been menopausal for a year or 2 and now has bothersome symptoms, I might start her on estradiol 1 mg tablets with progestin if a uterus is present. However, in this 41-year-old case patient, while I might start treatment at a standard dose, I would anticipate increasing to higher doses, such as 1.5 or 2 mg of daily estradiol until she feels her menopausal symptoms are adequately addressed.
Dr. Faubion: It is important to note that sometimes women with early BSO tend to have more severe vasomotor symptoms. Do you find that sometimes a higher dose is required just to manage symptoms, Dr. Kaunitz?
Dr. Kaunitz: Absolutely, yes. The decision whether or not to use systemic HT might be considered discretionary or elective in the classic 53-year-old woman recently menopausal with hot flashes, a so-called spontaneously or naturally menopausal woman. But my perspective is that unless there are clear contraindications, the decision to start systemic HT in the 41-year-old BSO case patient is actually not discretionary. Unless contraindications are present, it is important not only to treat symptoms but also to prevent an array of chronic major health concerns that are more likely if we don’t prescribe systemic HT.
Continue to: Health effects of not using HT...
Health effects of not using HT
Dr. Faubion: Dr. Kapoor, can you describe the potential long-term adverse health consequences of not using estrogen therapy? Say the same 41-year-old woman does not have many bothersome symptoms. What would you do?
Dr. Kapoor: Thank you for that important question. Building on what Dr. Kaunitz said, in these patients there are really 2 issues that can seem to be independent but are not: The first relates to the immediate consequences of lack of estrogen, ie, the menopause-related symptoms, but the second and perhaps the bigger issue is the long-term risk associated with estrogen deprivation.
The symptoms in these women are often obvious as they can be quite severe and abrupt; one day these women have normal hormone levels and the next day, after BSO, suddenly their hormones are very low. So if symptoms occur, they are usually hard to miss, simply because they are very drastic and very severe.
Historically, patients and their clinicians have targeted these symptoms. Patients experience menopausal symptoms, they seek treatment, and then the clinicians basically titrate the treatment to manage these symptoms. That misses the bigger issue, however, which is that premature estrogen deprivation leads to a host of chronic health conditions, as Dr. Kaunitz mentioned. These mainly include increased risk for cardiovascular disease, diabetes, hypertension, dyslipidemia, increased risk of mortality, dementia, and osteoporosis.
Fairly strong observational evidence suggests that use of estrogen therapy given in replacement doses—doses higher than those typically used in women after natural menopause, therefore considered replacement doses—helps mitigate the risk of some of these adverse health conditions.
In these women, the bigger goal really is to reinstate the hormonal milieu that exists prior to menopause. To your point, Dr. Faubion, if I have a patient who is younger than 46 years, who has her ovaries taken out, and even if she has zero symptoms (and sometimes that does happen), I would still make a case for this patient to utilize hormone therapy unless there is a contraindication such as breast cancer or other estrogen-sensitive cancers.
Dr. Faubion: Again, would you aim for those higher doses rather than treat with the “lowest dose”?
Dr. Kapoor: Absolutely. My punchline to the patients and clinicians in these discussions is that the rules of the game are different for these women. We cannot extrapolate the risks and benefits of HT use in women after natural menopause to younger women who have surgical menopause. Those rules just do not apply with respect to both benefits and risks.
Dr. Faubion: I think it’s important to say that these same “rules” would apply if the women were to go through premature menopause for any other reason, too, such as chemotherapy, radiation therapy, or premature ovarian insufficiency for any number of reasons, including toxic, metabolic, or genetic causes and so on. Would that be true?
Dr. Kapoor: Yes, absolutely so.
Dr. Faubion: Dr. Kaunitz, do you want to add anything?
Dr. Kaunitz: In terms of practical or clinical issues regarding systemic HT management, for the woman in her early 50s who has experienced normal or natural spontaneous menopause, a starting dose of transdermal estradiol would be, for instance, a 0.05-mg patch, which is a patch that over 24 hours releases 0.05 mg of estradiol daily; or standard oral estrogen, including conjugated equine estrogen, a 0.625-mg tablet daily, or estradiol, a 1-mg tablet daily.
But in younger patients, we want to use higher doses. For a patch, for instance, I would aim for a 0.075- or 0.1-mg estradiol patch, which releases a higher daily dose of estradiol than the standard dose. For oral estrogen, the dose would be 0.9- or even 1.25-mg tablets of conjugated equine estrogen or 1.5 mg, which is a 1-mg plus a 0.5-mg estradiol tablet, or a 2-mg estradiol tablet. Estradiol does come in a 2-mg strength.
For oral estrogen, I prefer estradiol because it’s available as a generic medication and often available at a very low cost, sometimes as low as $4 a month from chain pharmacies.
Continue to: Usefulness of monitoring estradiol levels for dosage adjustment...
Usefulness of monitoring estradiol levels for dosage adjustment
Dr. Faubion: That’s a great point, and again it is important to emphasize that we are aiming to recreate the premenopausal hormonal milieu. If you were to check estradiol levels, that would be aiming for a premenopausal range of approximately 80 to 120 pg per mL. Dr. Kapoor, is there utility in monitoring estrogen levels?
Dr. Kapoor: Great question, Dr. Faubion, and as you know it’s a loaded one. We base this on empiric evidence. We know that if the hormonal milieu in a young patient is changed to a postmenopausal one, her risk for many chronic conditions is increased. So if we were to reinstate a premenopausal hormonal milieu, that risk would probably be reduced. It makes good sense to target an empiric goal of 80 to 120 pg per mL of estradiol, which is the average estradiol level in a premenopausal woman. If you were to ask me, however, are there randomized, controlled trial data to support this practice—that is, if you target that level, can you make sure that the risk of diabetes is lower or that the risk of heart disease is lower—that study has yet to be done, and it may not ever be done on a large scale. However, it intuitively makes good sense to target premenopausal estradiol levels.
Dr. Faubion: When might you check an estradiol level in this population? For example, if you are treating a patient with a 0.1-mg estradiol patch and she still has significant hot flashes, would it be useful to check the level?
Dr. Kapoor: It would. In my practice, I check estradiol levels on these patients on an annual basis, regardless of symptoms, but definitely in the patient who has symptoms. It makes good sense, because sometimes these patients don’t absorb the estrogen well, particularly if administered by the transdermal route.
A general rule of thumb is that in the average population, if a patient is on the 0.1-mg patch, for example, you would expect her level to be around 100. If it is much lower than that, which sometimes happens, that speaks for poor absorption. Options at that point would be to treat her with a higher dose patch, depending on what the level is, or switch to a different formulation, such as oral.
In instances in which I have treated patients with a 0.1-mg patch for example, and their estradiol levels are undetectable, that speaks for very poor absorption. For such patients I make a case for switching them to oral therapy. Most definitely that makes sense in a patient who is symptomatic despite treatment. But even for patients who don’t have symptoms, I like to target that level, acknowledging that there is no evidence as such to support this practice.
Dr. Faubion: Dr. Kaunitz, do you want to add anything?
Dr. Kaunitz: Yes, a few practical points. Although patches are available in a wider array of doses than oral estrogen formulations, the highest dose available is 0.1 mg. It’s important for clinicians to recognize that while checking serum levels when indicated can be performed in women using transdermal estradiol or patches, in women who are using oral estrogen, checking blood levels is not going to work well because serum estrogen levels have a daily peak and valley in women who use oral versus transdermal estradiol.
I also wanted to talk about progestins. Although many patients who have had a BSO prior to spontaneous menopause also have had a hysterectomy, others have an intact uterus associated with their BSO, so progestins must be used along with estrogen. And if we are using higher-than-standard doses of estrogen, we also need to use higher-than-standard doses of progestin.
In that classic 53-year-old woman I referred to who had spontaneous normal menopause, if she is taking 1 mg of estradiol daily, or a 0.05-mg patch, or 0.625 mg of conjugated equine estrogen, 2.5 mg of medroxyprogesterone is fine. In fact, that showed excellent progestational protection of the endometrium in the Women’s Health Initiative and in other studies.
However, if we are going to use double the estrogen dose, we should increase the progestin dose too. In some of my patients on higher estrogen doses who have an intact uterus, I’ll use 5 or even 10 mg of daily medroxyprogesterone acetate to ensure adequate progestational suppression.
Dr. Faubion: Another practical tip is that if one is using conjugated equine estrogens, measuring the serum estradiol levels is not useful either.
Dr. Kaunitz: I agree.
Continue to: Oral contraceptives as replacement HT...
Oral contraceptives as replacement HT
Dr. Faubion: Would you comment on use of a birth control pill in this circumstance? Would it be optimal to use a postmenopausal HT regimen as opposed to a birth control pill or combined hormonal contraception?
Dr. Kapoor: In this younger population, sometimes it seems like a more socially acceptable decision to be on a birth control option than on menopausal HT. But there are some issues with being on a contraceptive regimen. One is that we end up using estrogen doses much higher than what is really needed for replacement purposes. It is also a nonphysiologic way of replacement in another sense—as opposed to estradiol, which is the main hormone made by the ovaries, the hormonal contraceptive regimens contain the synthetic estrogen ethinyl estradiol for the most part.
The other issue that is based on some weak evidence is that it appears that the bone health outcomes are probably inferior with combined hormonal contraception. For these reasons, regimens that are based on replacement doses of estradiol are preferred.
Dr. Faubion: Right, although the data are somewhat weak, I agree that thus far it seems optimal to utilize a postmenopausal regimen for various reasons. Dr. Kaunitz, anything to add?
Dr. Kaunitz: Yes, to underscore Dr. Kapoor’s point, a common oral contraceptive that contains 20 µg of ethinyl estradiol is substantially more estrogenic than 1.0 or 2.0 mg of micronized oral estradiol.
Also consider that a 20-µg ethinyl estradiol oral contraceptive may increase the risk of venous thromboembolism more than menopausal doses of oral estradiol, whether it be a micronized estradiol or conjugated equine estrogen.
Dr. Faubion: So the risk may be greater with oral combined hormonal contraception as well?
Dr. Kaunitz: One thing we can do is explain to our patients that their ovaries, prior to surgery or prior to induced menopause, were making substantial quantities of estradiol. Whether we prescribe a patch or oral micronized estradiol, this estrogen is identical to the hormone that their ovaries were making prior to surgery or induced menopause.
Breast cancer concerns
Dr. Faubion: Let’s consider a more complicated case. A 35-year-old woman has an identified BRCA1 mutation; she has not had any cancers but has undergone risk-reducing BSO and her uterus remains. Is this woman a candidate for HT? At what dose, and for how long? Dr. Kaunitz, why don’t you start.
Dr. Kaunitz: That is a challenging case but one that I think our readers will find interesting and maybe even provocative.
We know that women with BRCA1 mutations, the more common of the 2 BRCA mutations, have a very high risk of developing epithelial ovarian cancer at a young age. For this reason, our colleagues in medical oncology who specialize in hereditary ovarian/breast cancer syndromes recommend prophylactic risk-reducing—and I would also say lifesaving—BSO with or without hysterectomy for women with BRCA1 mutations.
However, over the years there has been tremendous reluctance among physicians caring for BRCA patients and the women themselves—I use the term “previvors” to describe BRCA carriers who have not been diagnosed with breast or ovarian cancer—to use HT after BSO because of concerns that HT might increase breast cancer risk in women who are already at high risk for breast cancer.
I assume, Dr. Faubion, that in this case the woman had gynecologic surgery but continues to have intact breasts. Is that correct?
Dr. Faubion: That is correct.
Dr. Kaunitz: Although the assumption has been that it is not safe to prescribe HT in this setting, in fact, the reported cohort studies that have looked at this issue have not found an elevated risk of breast cancer when replacement estrogen, with or without progestin, is prescribed to BRCA1 previvors with intact breasts.
Given what Dr. Kapoor said regarding the morbidity that is associated with BSO without replacement of physiologic estrogen, and also given the severe symptoms that so many of these young menopausal women experience, in my practice I do prescribe estrogen or estrogen-progestin therapy and focus on the higher target doses that we discussed for the earlier case patient who had a hysterectomy for abnormal uterine bleeding with adenomyosis.
Dr. Faubion: Dr. Kapoor, do you agree with this approach? How long would you continue therapy?
Dr. Kapoor: First, in this BRCA1 case we need to appreciate that the indication for the BSO is a legitimate one, in contrast to the first case in which the ovaries were removed in a patient whose average risk of ovarian cancer was low. It is important to recognize that surgery performed in this context is the right thing to do because it does significantly reduce the risk of ovarian cancer.
The second thing to appreciate is that while we reduce the risk of ovarian cancer significantly and make sure that these patients survive longer, it’s striking a fine balance in that you want to make sure that their morbidity is not increased as a result of premature estrogen deprivation.
As Dr. Kaunitz told us, the evidence that we have so far, which granted is not very robust but is fairly strong observational evidence, suggests that the risk of breast cancer is not elevated when these patients are treated with replacement doses of HT.
Having said that, I do have very strong discussions with my patients in this category about having the risk-reducing bilateral mastectomy also, because if they were to get breast cancer because of their increased genetic predisposition, the cancer is likely to grow faster if the patient is on HT. So one of my counseling points to patients is that they strongly consider bilateral mastectomy, which reduces their breast cancer risk by more than 90%. At the same time, I also strongly endorse using HT in replacement doses for the reasons that we have already stated.
Dr. Faubion: Continue HT until age 50 or 52?
Dr. Kapoor: Definitely until that age, and possibly longer, depending on their symptoms. The indications for treating beyond the age of natural menopause are much the same as for women who experience natural menopause.
Dr. Faubion: That is assuming they had a bilateral mastectomy?
Dr. Kapoor: Yes.
Continue to: Continuing HT until the age of natural menopause...
Continuing HT until the age of natural menopause
Dr. Kaunitz: Dr. Kapoor brings up the important point of duration of systemic HT. I agree that similar considerations apply both to the healthy 41-year-old who had a hysterectomy for abnormal uterine bleeding and to the 35-year-old who had risk-reducing surgery because of her BRCA1 mutation.
In the 2 cases, both to treat symptoms and to prevent chronic diseases, it makes sense to continue HT at least until the age of natural menopause. That is consistent with 2017 guidance from The North American Menopause Society (NAMS) position statement on the use of systemic HT, that is, continuing systemic HT at least until the age of natural menopause.3 Then at that point, continuing or discontinuing systemic HT becomes discretionary, and that would be true for both cases. If the patient is slender or has a strong family history of osteoporosis, that tends to push the patient more in terms of continuing systemic HT. Those are just some examples, and Dr. Kapoor may want to detail other relevant considerations.
Dr. Kapoor: I completely agree. The decision is driven by symptoms that are not otherwise well managed, for example, with nonhormone strategies. If we have any concerns utilizing HT beyond the age of natural menopause, then nonhormonal options can be considered; but sometimes those are not as effective. And bone health is very important. You want to avoid using bisphosphonates in younger women and reserve them for older patients in their late 60s and 70s. Hormone therapy use is a very reasonable strategy to prevent bone loss.
Dr. Kaunitz: It is also worth mentioning that sometimes the woman involved in shared decision making with her clinician decides to stop systemic HT. In that setting, should the patient start developing new-onset dyspareunia, vaginal dryness, or other genital or sexuality-related concerns, it takes very little for me to advise that she start low-dose local vaginal estrogen therapy.
Dr. Faubion: In either scenario, if a woman were to develop symptoms consistent with genitourinary syndrome of menopause (GSM), would you use vaginal estrogen in addition to the systemic estrogen or alone after the woman elected to discontinue systemic therapy?
Dr. Kapoor: Yes to both, I would say.
Dr. Kaunitz: As my patients using systemic HT age, often I will lower the dose. For instance, the dose I use in a 53-year-old will be higher than when she is 59 or 62. At the same time, as we lower the dose of systemic estrogen therapy, symptoms of vaginal atrophy or GSM often will appear, and these can be effectively treated by adding low-dose vaginal estrogen therapy. A number of my patients, particularly those who are on lower-than-standard doses of systemic HT, are also using low-dose vaginal estrogen therapy.
There is a “hybrid” product available: the 90-day estradiol vaginal ring. Estring is a low-dose, 2-mg, 90-day estradiol ring that is very useful, but it is effective only for treating GSM or vaginal atrophy. A second menopausal vaginal estradiol ring, Femring, is available in 2 doses: 0.05 mg/day and 0.1 mg/day. These are very effective in treating both systemic issues, such as vasomotor symptoms or prevention of osteoporosis, and very effective in treating GSM or vaginal atrophy. One problem is that Femring, depending on insurance coverage, can be very expensive. It’s not available as a generic, so for insurance or financial reasons I don’t often prescribe it. If I could remove those financial barriers, I would prescribe Femring more often because it is very useful.
Dr. Faubion: You raise an important point, and that is, for women who have been on HT for some time, clinicians often feel the need to slowly reduce the dose. Would you do that same thing, Dr. Kapoor, for a 40-year-old woman? Would you reduce the dose as she approaches age 50? Is there pressure that “she shouldn’t be on that much estrogen”?
Dr. Kapoor: No, I would not feel pressured until the patient turns at least 46. I bring up age 46 because the average age range for menopause is 46 to 55. After that, if there is any concern, we can decrease the dose to half and keep the patient on that until she turns 50 or 51. But most of my patients are on replacement doses until the average age of menopause, which is around 51 years, and that’s when you reduce the dose to that of the typical HT regimens used after natural menopause.
Sometimes patients are told something by a friend or they have read something and they worry about the risk of 2 things. One is breast cancer and the other is venous thromboembolism (VTE), and that may be why they want to be on a lower dose. I counsel patients that while the risk of VTE is real with HT, it is the women after natural menopause who are at risk—because age itself is a risk for VTE—and it also has to do with the kind of HT regimen that a patient is on. High doses of oral estrogens and certain progestogens increase the risk. But again, for estradiol used in replacement doses and the more common progestogens that we now use in practice, such as micronized progesterone, the risk is not the same. The same goes for breast cancer. My biggest message to patients and clinicians who take care of these patients is that the rules that apply to women after natural menopause just do not apply to this very different patient population.
Dr. Faubion: Thank you, Dr. Kaunitz and Dr. Kapoor, for sharing your knowledge and experience. ●
Systemic HT past the age of 65
Dr. Kaunitz: Another practical issue relates to long-term or extended use of systemic HT. It’s not infrequent in my practice to receive mail and faxes from insurance carriers of systemic HT users who are age 65 and older in which the company refers to the American Geriatrics Society’s Beers criteria for potentially inappropriate medication use in older adults,1 suggesting that systemic HT is inappropriate for all women over age 65. In this age group, I use lower doses if I am continuing systemic HT. But the good news is that both NAMS and the American College Obstetricians and Gynecologists indicate that arbitrarily stopping systemic HT at age 65 or for any other arbitrary reason is inappropriate, and that decisions about continuing or discontinuing therapy should be made on an individualized basis using shared decision making. That’s an important message for our readers.
Counseling regarding elective BSO
Dr. Faubion: One final note about elective BSO in the absence of a genetic mutation that predisposes to increased ovarian or breast cancer risk. Fortunately, we have seen rates of oophorectomy before the age of natural menopause decline, but what would your advice be to women or clinicians of these women who say they are “just afraid of ovarian cancer and would like to have their ovaries removed before the age of natural menopause”?
Dr. Kaunitz: If patients have increased anxiety about ovarian cancer and yet they themselves are not known to be at elevated risk, I emphasize that, fortunately, ovarian cancer is uncommon. It is much less common than other cancers the patient might be familiar with, such as breast or colon or lung cancer. I also emphasize that women who have given birth, particularly multiple times; women who nursed their infants; and women who have used combination hormonal contraceptives, particularly if long term, are at markedly lower risk for ovarian cancer as they get older. We are talking about an uncommon cancer that is even less common if women have given birth, nursed their infants, or used combination contraceptives long term.
Dr. Faubion: Dr. Kapoor, what would you say regarding the increased risk they might incur if they do have their ovaries out?
Dr. Kapoor: As Dr. Kaunitz said, this is an uncommon cancer, and pursuing something to reduce the risk of an uncommon cancer does not benefit the community. That is also my counseling point to patients.
I also talk to them extensively about the risk associated with the ovaries being removed, and I tell them that although we have the option of giving them HT, it is hard to replicate the magic of nature. No matter what concoction or regimen we use, we cannot ensure reinstating health to what it was in the premenopausal state, because estrogen has such myriad effects on the body in so many different organ systems.
Reference
1. American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American Geriatrics Society 2015 updated Beers criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2015;63:2227-2246.
- Kaunitz AM, Kapoor E, Faubion S. Treatment of women after bilateral salpingo-oophorectomy performed prior to natural menopause. JAMA. 2021;326:1429-1430.
- Writing Group for the Women’s Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321-333.
- North American Menopause Society. The 2017 hormone therapy position statement of The North American Menopause Society. J North Am Menopause Soc. 2017;24: 728-753.
- Kaunitz AM, Kapoor E, Faubion S. Treatment of women after bilateral salpingo-oophorectomy performed prior to natural menopause. JAMA. 2021;326:1429-1430.
- Writing Group for the Women’s Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321-333.
- North American Menopause Society. The 2017 hormone therapy position statement of The North American Menopause Society. J North Am Menopause Soc. 2017;24: 728-753.
Fibroids: Growing management options for a prevalent problem
OBG Manag. 33(12). | doi 10.12788/obgm.0169
2022 Update on obstetrics
Obstetrical practice saw updates in 2021 to 3 major areas of pregnancy management: preterm birth prevention, antepartum fetal surveillance, and the use of antenatal corticosteroids.
Updated guidance on predicting and preventing spontaneous PTB
American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins–Obstetrics. Prediction and prevention of spontaneous preterm birth: ACOG practice bulletin, number 234. Obstet Gynecol. 2021;138:e65-e90.
Preterm birth (PTB) continues to pose a challenge in clinical obstetrics, with the most recently reported rate of 10.2% in the United States.1 This accounts for almost 75% of perinatal mortality and more than half of neonatal morbidity, in which effects last well past the neonatal period. PTB is classified as spontaneous (following preterm labor, preterm prelabor rupture of membranes, or cervical insufficiency) or iatrogenic (indicated due to maternal and/or fetal complications).
Assessing risk for PTB
The single strongest predictor of subsequent PTB is a history of spontaneous PTB. Recurrence risk is further increased by the number of prior PTBs and the gestational age at prior PTB. Identification of and intervention for a short cervix has been shown to prolong gestation. Transvaginal ultrasonography of the cervix is the most accurate method for evaluating cervical length (CL). Specific examination criteria exist to ensure that CL measurements are reproducible and reliable.2 A short CL is generally defined as a measurement of less than 25 mm between 16 and 24 weeks’ gestation.
Screening strategies
The American College of Obstetricians and Gynecologists (ACOG), with an endorsement from the Society for Maternal-Fetal Medicine (SMFM), recommends cervical evaluation during the anatomy ultrasound exam between 18 0/7 and 22 6/7 weeks’ gestation in all pregnant patients regardless of prior PTB.3 If transabdominal imaging is concerning for a shortened cervix, transvaginal ultrasonography should be performed to assess the CL.
Serial transvaginal CL measurements are recommended between 16 0/7 and 24 0/7 weeks’ gestation for patients with a current singleton pregnancy and history of a spontaneous PTB, but not for patients with a history of iatrogenic or indicated PTB.
Interventions: Mind your p’s and c’s
Interventions to reduce the risk of spontaneous PTB depend on whether the current pregnancy is a singleton, twins, or higher-order multiples; CL measurement; and history of spontaneous PTB. Preconception optimization of underlying medical conditions also is important to reduce the risk of recurrent indicated PTB.
Continue to: Progesterone...
Progesterone
Vaginal administration. Several trials have shown that vaginal progesterone can be used to reduce the risk of spontaneous PTB in asymptomatic patients with a singleton pregnancy, incidental finding of a short cervix (<25 mm), and no history of spontaneous PTB. This is a change from the prior recommendation of CL of less than 20 mm. In the setting of a twin pregnancy, regardless of CL, data do not definitively support the use of vaginal progesterone.
Intramuscular administration.4,5 The popularity of intramuscular progesterone has waxed and waned. At present, ACOG recommends that all patients with a singleton pregnancy and history of spontaneous PTB be offered progesterone beginning at 16 0/7 weeks’ gestation following a shared decision-making process that includes the limited data of efficacy noted in existing studies.
In a twin pregnancy with no history of spontaneous PTB, the use of intramuscular progesterone has been shown to potentially increase the risk of PTB and admission to the neonatal intensive care unit. As such, intramuscular progesterone in the setting of a twin gestation without a history of spontaneous PTB is not recommended. When a prior spontaneous PTB has occurred, there may be some benefit to intramuscular progesterone in twin gestations.
Cerclage
Ultrasound indicated. In a singleton pregnancy with an incidental finding of short cervix (<25 mm) and no history of PTB, the use of cerclage is of uncertain benefit. Effectiveness may be seen if the cervix is less than 10 mm. Ultrasound-indicated cerclage should be considered in a singleton pregnancy with a CL less than 25 mm and a history of spontaneous PTB.
Possibly one of the most controversial topics is ultrasound-indicated cerclage placement in twin gestation. As with many situations in obstetrics, data regarding ultrasound-indicated cerclage in twin gestation is based on small retrospective studies fraught with bias. Results from these studies range from no benefit, to potential benefit, to even possible increased risk of PTB. Since data are limited, as we await more evidence, it is recommended that the clinician and patient use shared decision making to decide on cerclage placement in a twin gestation.
Exam indicated. In a singleton pregnancy with a dilated cervix on digital or speculum exam between 16 0/7 to 23 6/7 weeks’ gestation, a physical exam–indicated cerclage should be offered. Exam-indicated cerclage also may reduce the incidence of PTB in twin gestations with cervical dilation between 16 0/7 and 23 6/7 weeks’ gestation. Indomethacin tocolysis and perioperative antibiotics should be considered when an exam-indicated cerclage is placed.
As the limits of viability are continually pushed earlier, more in-depth conversation is needed with patients who are considering an exam-indicated cerclage. The nuances of periviability and the likelihood that an exam-indicated cerclage will commit a pregnancy to a periviable or extremely preterm birth should be discussed in detail using a shared decision making model.
Regardless of whether the cerclage is ultrasound or exam indicated, once it is placed there is no utility in additional CL ultrasound monitoring.
Pessary
Vaginal pessaries for prevention of PTB have not gained popularity in the United States as they have in other countries. Trials are being conducted to determine the utility of vaginal pessary, but current data have not proven its effectiveness in preventing PTB in the setting of singleton pregnancy, short cervix, and no history of spontaneous PTB. So for now, pessary is not recommended. The same can be said for use in the twin gestation.
- All patients should have cervical evaluation during pregnancy. Serial imaging is reserved for those with a history of spontaneous PTB.
- Progesterone supplementation should be offered to patients with a singleton pregnancy and a history of spontaneous PTB or to patients with a singleton pregnancy and no history of spontaneous PTB who have cervical shortening at less than 24 weeks.
- Cerclage may be offered between 16 and 24 weeks for a cervical length less than 25 mm in a patient with a singleton gestation who has a history of spontaneous PTB (<10 mm if no history of spontaneous PTB) or for a dilated cervix on exam regardless of history.
- Women who have a twin gestation with cervical dilation may be offered physical exam–indicated cerclage.
Which patients may benefit from antepartum fetal surveillance and when to initiate it
American College of Obstetricians and Gynecologists’ Committee on Obstetrics Practice, Society for Maternal-Fetal Medicine. Indications for outpatient antenatal surveillance: ACOG committee opinion, number 828. Obstet Gynecol. 2021;137:e177-e197.
American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins—Obstetrics. Antepartum fetal surveillance: ACOG practice bulletin, number 229. Obstet Gynecol. 2021;137:e116-e127.
The ultimate purpose of antenatal fetal surveillance is to prevent stillbirth. However, stillbirth has multiple etiologies, not all of which are preventable with testing. In June 2021, ACOG released a new Committee Opinion containing guidelines for fetal surveillance, including suggested gestational age at initiation and frequency of testing, for the most common high-risk conditions. ACOG also released an update to the Practice Bulletin on antepartum fetal surveillance; additions include randomized controlled trial level data on the utility of fetal kick counts (FKCs) and recommendations that align with the new Committee Opinion.
Data for the efficacy of antepartum fetal surveillance are lacking, mainly due to the difficulty of performing prospective studies in stillbirth. The existing evidence is subject to intervention bias, as deliveries increase in tested patients, and recommendations rely heavily on expert consensus and nonrandomized studies. Antenatal testing is also time, cost, and labor intensive, with the risk of intervention for a false-positive result. Despite these limitations, obstetrical practices routinely perform antenatal fetal surveillance.
The new guidelines: The why, when, and how often
Why. Antepartum fetal surveillance is suggested for conditions that have a risk of stillbirth greater than 0.8 per 1,000 (that is, the false-negative rate of a biophysical profile or a modified biophysical profile) and the relative risk or odds ratio is greater than 2.0 for stillbirth compared with unaffected pregnancies.
When. For most conditions, ACOG recommends initiation of testing at 32 weeks or later, with notable earlier exceptions for some of the highest-risk patients. For certain conditions, such as fetal growth restriction and hypertensive disorders of pregnancy, the recommendation is to start “at diagnosis,” with the corollary “or at a gestational age when delivery would be considered because of abnormal results.” Shared decision making with the patient about pregnancy goals therefore is required, particularly in cases of fetal anomalies, genetic conditions, or at very early gestational ages.
How often. The recommended frequency of testing is at least weekly. Testing frequency should be increased to twice-weekly outpatient or daily inpatient for the most complicated pregnancies (for example, fetal growth restriction with abnormal umbilical artery Doppler studies, preeclampsia with severe features).
Once or twice weekly is an option for many conditions, which gives the clinician the opportunity to assess clinical stability as well as the patient’s input in terms of logistics and anxiety.
Patients with multiple conditions may fall into the “individualized” category, as do patients with suboptimal control of conditions (for example, diabetes, hypertension) that may affect the fetus as the pregnancy progresses.
New diagnoses included for surveillance
Several diagnoses not previously included now qualify for antepartum fetal surveillance under the new guidelines, most notably:
- history of obstetrical complications in the immediate preceding pregnancy
—history of prior fetal growth restriction requiring preterm delivery
—history of prior preeclampsia requiring delivery
- alcohol use of 5 or more drinks per day
- in vitro fertilization
- abnormal serum markers
—pregnancy-associated plasma protein A (PAPP-A) in the fifth or lower percentile or 0.4 multiples of the median (MoM)
—second trimester inhibin A of 2 or greater MoM
- prepregnancy body mass index (BMI)
—this is divided into 2 categories for timing of initiation of testing:
- 37 weeks for BMI of 35 to 39.9 kg/m2
- 34 weeks for BMI of or greater than 40 kg/m2.
Fetal kick counts
The major change to the updated Practice Bulletin on antenatal surveillance is the inclusion of data on FKCs, a simple modality of fetal surveillance that does not require a clinical visit. For FKCs, a meta-analysis of more than 450,000 patients did not demonstrate a difference in perinatal death between the FKC intervention group (0.54%) and the control group (0.59%). Of note, there were small but statistically significant increases in the rates of induction of labor, cesarean delivery, and preterm delivery in the FKC intervention group. Therefore, this update does not recommend a formal program of FKCs for all patients.
- The antenatal fetal surveillance guidelines are just that—guidelines, not mandates. Their use will need to be adapted for specific patient populations and practice management patterns.
- Many conditions qualify for “individualized” surveillance, which offers the opportunity for detailed discussions on the patient’s care. This includes shared decision making with patients to meet their goals for the pregnancy.
- Although patient-perceived decreased fetal movement always warrants clinical evaluation, a regular program of fetal kick count monitoring is not recommended for all patients due to lack of data supporting its benefit in reducing perinatal death.
- As with any change, new guidelines potentially are a source of frustration, so a concerted effort by obstetrical clinicians to agree on adoption of the guidelines is needed. Additional clinical resources and both clinician and patient education may be required depending on current practice style, as the new strategy may increase the number of appointments and ultrasound exams required.
Continue to: Use of antenatal corticosteroids now may be considered at 22 weeks’ gestation...
Use of antenatal corticosteroids now may be considered at 22 weeks’ gestation
American College of Obstetricians and Gynecologists. Use of antenatal corticosteroids at 22 weeks of gestation: ACOG practice advisory. September 2021. https://www .acog.org/clinical/clinical-guidance/practice-advisory /articles/2021/09/use-of-antenatal-corticosteroids-at -22-weeks-of-gestation. Accessed December 11, 2021.
In September 2021, ACOG and SMFM released a Practice Advisory updating the current recommendations for the administration of antenatal corticosteroids in the periviable period (22 to 25 6/7 weeks’ gestation). Whereas the prior lower limit of gestational age for consideration of steroids was 23 weeks, the new recommendation now extends this consideration down to 22 weeks.
The cited data include a meta-analysis of more than 2,200 patients in which the survival rate of infants born between 22 and 22 6/7 weeks who were exposed to antenatal steroids was 39% compared with 19.5% in the unexposed group. Another study of more than 1,000 patients demonstrated a statistically significant increase in overall survival in patients treated with antenatal steroids plus life support compared with life support only (38.5% vs 17.7%). Survival without major morbidity in this study, although increased from 1% to 4.4%, was still low.
Recommendation carries caveats
Given this information, the Practice Advisory offers a 2C level recommendation (weak recommendation, low quality of evidence) for antenatal steroids at 22 to 22 6/7 weeks’ gestation if neonatal resuscitation is planned, acknowledging the limitations and potential bias of the available data.
The Practice Advisory emphasizes the importance of counseling and patient involvement in the decision making. This requires a multidisciplinary collaboration among the neonatology and obstetrical teams, flexibility in the plan after birth depending on the infant’s condition, and redirection of care if appropriate. Estimated fetal weight, the presence of multiple gestations, fetal biologic sex, and any anomalies are also important in helping families make an informed decision for their particular pregnancy. As described in the Obstetric Care Consensus on periviable birth,6 it is important to remember that considerations and recommendations are not the same as requirements, and redirection of care to comfort and family memory making is not the same as withholding care.
The rest of the recommendations for the administration of antenatal steroids remain the same: Antenatal steroids are not recommended at less than 22 weeks due to lack of evidence of benefit, and they continue to be recommended at 24 weeks and beyond. ●
- Antenatal corticosteroids may be considered at 22 to 22 6/7 weeks’ gestation if, after thorough patient counseling, neonatal resuscitation is desired and planned by the family.
- The overall likelihood of survival and survival without major morbidities continues to be very low in the periviable period, especially at 22 weeks. Gestational age is only one of the many factors that must be considered in the shared decision making for this very difficult decision.
- Palliative care is a valid and appropriate option for patients facing a periviable delivery after appropriate counseling or after evaluation of the infant has occurred after birth.
- Martin JA, Hamilton BE, Osterman MJK. Births in the United States, 2019. NCHS Data Brief, no 387. Hyattsville, MD: National Center for Health Statistics. October 2020. www.cdc.gov/nchs /data/databriefs/db387-H.pdf. Accessed December 20, 2021.
- To MS, Skentou C, Chan C, et al. Cervical assessment at the routine 23-week scan: standardizing techniques. Ultrasound Obstet Gynecol. 2001;17:217-219.
- American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins–Obstetrics. Prediction and prevention of spontaneous preterm birth: ACOG practice bulletin, number 234. Obstet Gynecol. 2021;138:e65-e90.
- Society for Maternal-Fetal Medicine (SMFM) Publications Committee. SMFM statement: use of 17-alpha hydroxyprogesterone caproate for prevention of recurrent preterm birth. Am J Obstet Gynecol. 2020;223:B16-B18.
- Blackwell SC, Gyamfi-Bannerman C, Biggio JR Jr, et al. 17-OHPC to prevent recurrent preterm birth in singleton gestations (PROLONG study): a multicenter, international, randomized double-blind trial. Am J Perinatol. 2020;37:127-136.
- American College of Obstetricians and Gynecologists; Society for Maternal-Fetal Medicine. Obstetric care consensus no. 6: Periviable birth. Obstet Gynecol. 2017;130:e187-e199.
Obstetrical practice saw updates in 2021 to 3 major areas of pregnancy management: preterm birth prevention, antepartum fetal surveillance, and the use of antenatal corticosteroids.
Updated guidance on predicting and preventing spontaneous PTB
American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins–Obstetrics. Prediction and prevention of spontaneous preterm birth: ACOG practice bulletin, number 234. Obstet Gynecol. 2021;138:e65-e90.
Preterm birth (PTB) continues to pose a challenge in clinical obstetrics, with the most recently reported rate of 10.2% in the United States.1 This accounts for almost 75% of perinatal mortality and more than half of neonatal morbidity, in which effects last well past the neonatal period. PTB is classified as spontaneous (following preterm labor, preterm prelabor rupture of membranes, or cervical insufficiency) or iatrogenic (indicated due to maternal and/or fetal complications).
Assessing risk for PTB
The single strongest predictor of subsequent PTB is a history of spontaneous PTB. Recurrence risk is further increased by the number of prior PTBs and the gestational age at prior PTB. Identification of and intervention for a short cervix has been shown to prolong gestation. Transvaginal ultrasonography of the cervix is the most accurate method for evaluating cervical length (CL). Specific examination criteria exist to ensure that CL measurements are reproducible and reliable.2 A short CL is generally defined as a measurement of less than 25 mm between 16 and 24 weeks’ gestation.
Screening strategies
The American College of Obstetricians and Gynecologists (ACOG), with an endorsement from the Society for Maternal-Fetal Medicine (SMFM), recommends cervical evaluation during the anatomy ultrasound exam between 18 0/7 and 22 6/7 weeks’ gestation in all pregnant patients regardless of prior PTB.3 If transabdominal imaging is concerning for a shortened cervix, transvaginal ultrasonography should be performed to assess the CL.
Serial transvaginal CL measurements are recommended between 16 0/7 and 24 0/7 weeks’ gestation for patients with a current singleton pregnancy and history of a spontaneous PTB, but not for patients with a history of iatrogenic or indicated PTB.
Interventions: Mind your p’s and c’s
Interventions to reduce the risk of spontaneous PTB depend on whether the current pregnancy is a singleton, twins, or higher-order multiples; CL measurement; and history of spontaneous PTB. Preconception optimization of underlying medical conditions also is important to reduce the risk of recurrent indicated PTB.
Continue to: Progesterone...
Progesterone
Vaginal administration. Several trials have shown that vaginal progesterone can be used to reduce the risk of spontaneous PTB in asymptomatic patients with a singleton pregnancy, incidental finding of a short cervix (<25 mm), and no history of spontaneous PTB. This is a change from the prior recommendation of CL of less than 20 mm. In the setting of a twin pregnancy, regardless of CL, data do not definitively support the use of vaginal progesterone.
Intramuscular administration.4,5 The popularity of intramuscular progesterone has waxed and waned. At present, ACOG recommends that all patients with a singleton pregnancy and history of spontaneous PTB be offered progesterone beginning at 16 0/7 weeks’ gestation following a shared decision-making process that includes the limited data of efficacy noted in existing studies.
In a twin pregnancy with no history of spontaneous PTB, the use of intramuscular progesterone has been shown to potentially increase the risk of PTB and admission to the neonatal intensive care unit. As such, intramuscular progesterone in the setting of a twin gestation without a history of spontaneous PTB is not recommended. When a prior spontaneous PTB has occurred, there may be some benefit to intramuscular progesterone in twin gestations.
Cerclage
Ultrasound indicated. In a singleton pregnancy with an incidental finding of short cervix (<25 mm) and no history of PTB, the use of cerclage is of uncertain benefit. Effectiveness may be seen if the cervix is less than 10 mm. Ultrasound-indicated cerclage should be considered in a singleton pregnancy with a CL less than 25 mm and a history of spontaneous PTB.
Possibly one of the most controversial topics is ultrasound-indicated cerclage placement in twin gestation. As with many situations in obstetrics, data regarding ultrasound-indicated cerclage in twin gestation is based on small retrospective studies fraught with bias. Results from these studies range from no benefit, to potential benefit, to even possible increased risk of PTB. Since data are limited, as we await more evidence, it is recommended that the clinician and patient use shared decision making to decide on cerclage placement in a twin gestation.
Exam indicated. In a singleton pregnancy with a dilated cervix on digital or speculum exam between 16 0/7 to 23 6/7 weeks’ gestation, a physical exam–indicated cerclage should be offered. Exam-indicated cerclage also may reduce the incidence of PTB in twin gestations with cervical dilation between 16 0/7 and 23 6/7 weeks’ gestation. Indomethacin tocolysis and perioperative antibiotics should be considered when an exam-indicated cerclage is placed.
As the limits of viability are continually pushed earlier, more in-depth conversation is needed with patients who are considering an exam-indicated cerclage. The nuances of periviability and the likelihood that an exam-indicated cerclage will commit a pregnancy to a periviable or extremely preterm birth should be discussed in detail using a shared decision making model.
Regardless of whether the cerclage is ultrasound or exam indicated, once it is placed there is no utility in additional CL ultrasound monitoring.
Pessary
Vaginal pessaries for prevention of PTB have not gained popularity in the United States as they have in other countries. Trials are being conducted to determine the utility of vaginal pessary, but current data have not proven its effectiveness in preventing PTB in the setting of singleton pregnancy, short cervix, and no history of spontaneous PTB. So for now, pessary is not recommended. The same can be said for use in the twin gestation.
- All patients should have cervical evaluation during pregnancy. Serial imaging is reserved for those with a history of spontaneous PTB.
- Progesterone supplementation should be offered to patients with a singleton pregnancy and a history of spontaneous PTB or to patients with a singleton pregnancy and no history of spontaneous PTB who have cervical shortening at less than 24 weeks.
- Cerclage may be offered between 16 and 24 weeks for a cervical length less than 25 mm in a patient with a singleton gestation who has a history of spontaneous PTB (<10 mm if no history of spontaneous PTB) or for a dilated cervix on exam regardless of history.
- Women who have a twin gestation with cervical dilation may be offered physical exam–indicated cerclage.
Which patients may benefit from antepartum fetal surveillance and when to initiate it
American College of Obstetricians and Gynecologists’ Committee on Obstetrics Practice, Society for Maternal-Fetal Medicine. Indications for outpatient antenatal surveillance: ACOG committee opinion, number 828. Obstet Gynecol. 2021;137:e177-e197.
American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins—Obstetrics. Antepartum fetal surveillance: ACOG practice bulletin, number 229. Obstet Gynecol. 2021;137:e116-e127.
The ultimate purpose of antenatal fetal surveillance is to prevent stillbirth. However, stillbirth has multiple etiologies, not all of which are preventable with testing. In June 2021, ACOG released a new Committee Opinion containing guidelines for fetal surveillance, including suggested gestational age at initiation and frequency of testing, for the most common high-risk conditions. ACOG also released an update to the Practice Bulletin on antepartum fetal surveillance; additions include randomized controlled trial level data on the utility of fetal kick counts (FKCs) and recommendations that align with the new Committee Opinion.
Data for the efficacy of antepartum fetal surveillance are lacking, mainly due to the difficulty of performing prospective studies in stillbirth. The existing evidence is subject to intervention bias, as deliveries increase in tested patients, and recommendations rely heavily on expert consensus and nonrandomized studies. Antenatal testing is also time, cost, and labor intensive, with the risk of intervention for a false-positive result. Despite these limitations, obstetrical practices routinely perform antenatal fetal surveillance.
The new guidelines: The why, when, and how often
Why. Antepartum fetal surveillance is suggested for conditions that have a risk of stillbirth greater than 0.8 per 1,000 (that is, the false-negative rate of a biophysical profile or a modified biophysical profile) and the relative risk or odds ratio is greater than 2.0 for stillbirth compared with unaffected pregnancies.
When. For most conditions, ACOG recommends initiation of testing at 32 weeks or later, with notable earlier exceptions for some of the highest-risk patients. For certain conditions, such as fetal growth restriction and hypertensive disorders of pregnancy, the recommendation is to start “at diagnosis,” with the corollary “or at a gestational age when delivery would be considered because of abnormal results.” Shared decision making with the patient about pregnancy goals therefore is required, particularly in cases of fetal anomalies, genetic conditions, or at very early gestational ages.
How often. The recommended frequency of testing is at least weekly. Testing frequency should be increased to twice-weekly outpatient or daily inpatient for the most complicated pregnancies (for example, fetal growth restriction with abnormal umbilical artery Doppler studies, preeclampsia with severe features).
Once or twice weekly is an option for many conditions, which gives the clinician the opportunity to assess clinical stability as well as the patient’s input in terms of logistics and anxiety.
Patients with multiple conditions may fall into the “individualized” category, as do patients with suboptimal control of conditions (for example, diabetes, hypertension) that may affect the fetus as the pregnancy progresses.
New diagnoses included for surveillance
Several diagnoses not previously included now qualify for antepartum fetal surveillance under the new guidelines, most notably:
- history of obstetrical complications in the immediate preceding pregnancy
—history of prior fetal growth restriction requiring preterm delivery
—history of prior preeclampsia requiring delivery
- alcohol use of 5 or more drinks per day
- in vitro fertilization
- abnormal serum markers
—pregnancy-associated plasma protein A (PAPP-A) in the fifth or lower percentile or 0.4 multiples of the median (MoM)
—second trimester inhibin A of 2 or greater MoM
- prepregnancy body mass index (BMI)
—this is divided into 2 categories for timing of initiation of testing:
- 37 weeks for BMI of 35 to 39.9 kg/m2
- 34 weeks for BMI of or greater than 40 kg/m2.
Fetal kick counts
The major change to the updated Practice Bulletin on antenatal surveillance is the inclusion of data on FKCs, a simple modality of fetal surveillance that does not require a clinical visit. For FKCs, a meta-analysis of more than 450,000 patients did not demonstrate a difference in perinatal death between the FKC intervention group (0.54%) and the control group (0.59%). Of note, there were small but statistically significant increases in the rates of induction of labor, cesarean delivery, and preterm delivery in the FKC intervention group. Therefore, this update does not recommend a formal program of FKCs for all patients.
- The antenatal fetal surveillance guidelines are just that—guidelines, not mandates. Their use will need to be adapted for specific patient populations and practice management patterns.
- Many conditions qualify for “individualized” surveillance, which offers the opportunity for detailed discussions on the patient’s care. This includes shared decision making with patients to meet their goals for the pregnancy.
- Although patient-perceived decreased fetal movement always warrants clinical evaluation, a regular program of fetal kick count monitoring is not recommended for all patients due to lack of data supporting its benefit in reducing perinatal death.
- As with any change, new guidelines potentially are a source of frustration, so a concerted effort by obstetrical clinicians to agree on adoption of the guidelines is needed. Additional clinical resources and both clinician and patient education may be required depending on current practice style, as the new strategy may increase the number of appointments and ultrasound exams required.
Continue to: Use of antenatal corticosteroids now may be considered at 22 weeks’ gestation...
Use of antenatal corticosteroids now may be considered at 22 weeks’ gestation
American College of Obstetricians and Gynecologists. Use of antenatal corticosteroids at 22 weeks of gestation: ACOG practice advisory. September 2021. https://www .acog.org/clinical/clinical-guidance/practice-advisory /articles/2021/09/use-of-antenatal-corticosteroids-at -22-weeks-of-gestation. Accessed December 11, 2021.
In September 2021, ACOG and SMFM released a Practice Advisory updating the current recommendations for the administration of antenatal corticosteroids in the periviable period (22 to 25 6/7 weeks’ gestation). Whereas the prior lower limit of gestational age for consideration of steroids was 23 weeks, the new recommendation now extends this consideration down to 22 weeks.
The cited data include a meta-analysis of more than 2,200 patients in which the survival rate of infants born between 22 and 22 6/7 weeks who were exposed to antenatal steroids was 39% compared with 19.5% in the unexposed group. Another study of more than 1,000 patients demonstrated a statistically significant increase in overall survival in patients treated with antenatal steroids plus life support compared with life support only (38.5% vs 17.7%). Survival without major morbidity in this study, although increased from 1% to 4.4%, was still low.
Recommendation carries caveats
Given this information, the Practice Advisory offers a 2C level recommendation (weak recommendation, low quality of evidence) for antenatal steroids at 22 to 22 6/7 weeks’ gestation if neonatal resuscitation is planned, acknowledging the limitations and potential bias of the available data.
The Practice Advisory emphasizes the importance of counseling and patient involvement in the decision making. This requires a multidisciplinary collaboration among the neonatology and obstetrical teams, flexibility in the plan after birth depending on the infant’s condition, and redirection of care if appropriate. Estimated fetal weight, the presence of multiple gestations, fetal biologic sex, and any anomalies are also important in helping families make an informed decision for their particular pregnancy. As described in the Obstetric Care Consensus on periviable birth,6 it is important to remember that considerations and recommendations are not the same as requirements, and redirection of care to comfort and family memory making is not the same as withholding care.
The rest of the recommendations for the administration of antenatal steroids remain the same: Antenatal steroids are not recommended at less than 22 weeks due to lack of evidence of benefit, and they continue to be recommended at 24 weeks and beyond. ●
- Antenatal corticosteroids may be considered at 22 to 22 6/7 weeks’ gestation if, after thorough patient counseling, neonatal resuscitation is desired and planned by the family.
- The overall likelihood of survival and survival without major morbidities continues to be very low in the periviable period, especially at 22 weeks. Gestational age is only one of the many factors that must be considered in the shared decision making for this very difficult decision.
- Palliative care is a valid and appropriate option for patients facing a periviable delivery after appropriate counseling or after evaluation of the infant has occurred after birth.
Obstetrical practice saw updates in 2021 to 3 major areas of pregnancy management: preterm birth prevention, antepartum fetal surveillance, and the use of antenatal corticosteroids.
Updated guidance on predicting and preventing spontaneous PTB
American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins–Obstetrics. Prediction and prevention of spontaneous preterm birth: ACOG practice bulletin, number 234. Obstet Gynecol. 2021;138:e65-e90.
Preterm birth (PTB) continues to pose a challenge in clinical obstetrics, with the most recently reported rate of 10.2% in the United States.1 This accounts for almost 75% of perinatal mortality and more than half of neonatal morbidity, in which effects last well past the neonatal period. PTB is classified as spontaneous (following preterm labor, preterm prelabor rupture of membranes, or cervical insufficiency) or iatrogenic (indicated due to maternal and/or fetal complications).
Assessing risk for PTB
The single strongest predictor of subsequent PTB is a history of spontaneous PTB. Recurrence risk is further increased by the number of prior PTBs and the gestational age at prior PTB. Identification of and intervention for a short cervix has been shown to prolong gestation. Transvaginal ultrasonography of the cervix is the most accurate method for evaluating cervical length (CL). Specific examination criteria exist to ensure that CL measurements are reproducible and reliable.2 A short CL is generally defined as a measurement of less than 25 mm between 16 and 24 weeks’ gestation.
Screening strategies
The American College of Obstetricians and Gynecologists (ACOG), with an endorsement from the Society for Maternal-Fetal Medicine (SMFM), recommends cervical evaluation during the anatomy ultrasound exam between 18 0/7 and 22 6/7 weeks’ gestation in all pregnant patients regardless of prior PTB.3 If transabdominal imaging is concerning for a shortened cervix, transvaginal ultrasonography should be performed to assess the CL.
Serial transvaginal CL measurements are recommended between 16 0/7 and 24 0/7 weeks’ gestation for patients with a current singleton pregnancy and history of a spontaneous PTB, but not for patients with a history of iatrogenic or indicated PTB.
Interventions: Mind your p’s and c’s
Interventions to reduce the risk of spontaneous PTB depend on whether the current pregnancy is a singleton, twins, or higher-order multiples; CL measurement; and history of spontaneous PTB. Preconception optimization of underlying medical conditions also is important to reduce the risk of recurrent indicated PTB.
Continue to: Progesterone...
Progesterone
Vaginal administration. Several trials have shown that vaginal progesterone can be used to reduce the risk of spontaneous PTB in asymptomatic patients with a singleton pregnancy, incidental finding of a short cervix (<25 mm), and no history of spontaneous PTB. This is a change from the prior recommendation of CL of less than 20 mm. In the setting of a twin pregnancy, regardless of CL, data do not definitively support the use of vaginal progesterone.
Intramuscular administration.4,5 The popularity of intramuscular progesterone has waxed and waned. At present, ACOG recommends that all patients with a singleton pregnancy and history of spontaneous PTB be offered progesterone beginning at 16 0/7 weeks’ gestation following a shared decision-making process that includes the limited data of efficacy noted in existing studies.
In a twin pregnancy with no history of spontaneous PTB, the use of intramuscular progesterone has been shown to potentially increase the risk of PTB and admission to the neonatal intensive care unit. As such, intramuscular progesterone in the setting of a twin gestation without a history of spontaneous PTB is not recommended. When a prior spontaneous PTB has occurred, there may be some benefit to intramuscular progesterone in twin gestations.
Cerclage
Ultrasound indicated. In a singleton pregnancy with an incidental finding of short cervix (<25 mm) and no history of PTB, the use of cerclage is of uncertain benefit. Effectiveness may be seen if the cervix is less than 10 mm. Ultrasound-indicated cerclage should be considered in a singleton pregnancy with a CL less than 25 mm and a history of spontaneous PTB.
Possibly one of the most controversial topics is ultrasound-indicated cerclage placement in twin gestation. As with many situations in obstetrics, data regarding ultrasound-indicated cerclage in twin gestation is based on small retrospective studies fraught with bias. Results from these studies range from no benefit, to potential benefit, to even possible increased risk of PTB. Since data are limited, as we await more evidence, it is recommended that the clinician and patient use shared decision making to decide on cerclage placement in a twin gestation.
Exam indicated. In a singleton pregnancy with a dilated cervix on digital or speculum exam between 16 0/7 to 23 6/7 weeks’ gestation, a physical exam–indicated cerclage should be offered. Exam-indicated cerclage also may reduce the incidence of PTB in twin gestations with cervical dilation between 16 0/7 and 23 6/7 weeks’ gestation. Indomethacin tocolysis and perioperative antibiotics should be considered when an exam-indicated cerclage is placed.
As the limits of viability are continually pushed earlier, more in-depth conversation is needed with patients who are considering an exam-indicated cerclage. The nuances of periviability and the likelihood that an exam-indicated cerclage will commit a pregnancy to a periviable or extremely preterm birth should be discussed in detail using a shared decision making model.
Regardless of whether the cerclage is ultrasound or exam indicated, once it is placed there is no utility in additional CL ultrasound monitoring.
Pessary
Vaginal pessaries for prevention of PTB have not gained popularity in the United States as they have in other countries. Trials are being conducted to determine the utility of vaginal pessary, but current data have not proven its effectiveness in preventing PTB in the setting of singleton pregnancy, short cervix, and no history of spontaneous PTB. So for now, pessary is not recommended. The same can be said for use in the twin gestation.
- All patients should have cervical evaluation during pregnancy. Serial imaging is reserved for those with a history of spontaneous PTB.
- Progesterone supplementation should be offered to patients with a singleton pregnancy and a history of spontaneous PTB or to patients with a singleton pregnancy and no history of spontaneous PTB who have cervical shortening at less than 24 weeks.
- Cerclage may be offered between 16 and 24 weeks for a cervical length less than 25 mm in a patient with a singleton gestation who has a history of spontaneous PTB (<10 mm if no history of spontaneous PTB) or for a dilated cervix on exam regardless of history.
- Women who have a twin gestation with cervical dilation may be offered physical exam–indicated cerclage.
Which patients may benefit from antepartum fetal surveillance and when to initiate it
American College of Obstetricians and Gynecologists’ Committee on Obstetrics Practice, Society for Maternal-Fetal Medicine. Indications for outpatient antenatal surveillance: ACOG committee opinion, number 828. Obstet Gynecol. 2021;137:e177-e197.
American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins—Obstetrics. Antepartum fetal surveillance: ACOG practice bulletin, number 229. Obstet Gynecol. 2021;137:e116-e127.
The ultimate purpose of antenatal fetal surveillance is to prevent stillbirth. However, stillbirth has multiple etiologies, not all of which are preventable with testing. In June 2021, ACOG released a new Committee Opinion containing guidelines for fetal surveillance, including suggested gestational age at initiation and frequency of testing, for the most common high-risk conditions. ACOG also released an update to the Practice Bulletin on antepartum fetal surveillance; additions include randomized controlled trial level data on the utility of fetal kick counts (FKCs) and recommendations that align with the new Committee Opinion.
Data for the efficacy of antepartum fetal surveillance are lacking, mainly due to the difficulty of performing prospective studies in stillbirth. The existing evidence is subject to intervention bias, as deliveries increase in tested patients, and recommendations rely heavily on expert consensus and nonrandomized studies. Antenatal testing is also time, cost, and labor intensive, with the risk of intervention for a false-positive result. Despite these limitations, obstetrical practices routinely perform antenatal fetal surveillance.
The new guidelines: The why, when, and how often
Why. Antepartum fetal surveillance is suggested for conditions that have a risk of stillbirth greater than 0.8 per 1,000 (that is, the false-negative rate of a biophysical profile or a modified biophysical profile) and the relative risk or odds ratio is greater than 2.0 for stillbirth compared with unaffected pregnancies.
When. For most conditions, ACOG recommends initiation of testing at 32 weeks or later, with notable earlier exceptions for some of the highest-risk patients. For certain conditions, such as fetal growth restriction and hypertensive disorders of pregnancy, the recommendation is to start “at diagnosis,” with the corollary “or at a gestational age when delivery would be considered because of abnormal results.” Shared decision making with the patient about pregnancy goals therefore is required, particularly in cases of fetal anomalies, genetic conditions, or at very early gestational ages.
How often. The recommended frequency of testing is at least weekly. Testing frequency should be increased to twice-weekly outpatient or daily inpatient for the most complicated pregnancies (for example, fetal growth restriction with abnormal umbilical artery Doppler studies, preeclampsia with severe features).
Once or twice weekly is an option for many conditions, which gives the clinician the opportunity to assess clinical stability as well as the patient’s input in terms of logistics and anxiety.
Patients with multiple conditions may fall into the “individualized” category, as do patients with suboptimal control of conditions (for example, diabetes, hypertension) that may affect the fetus as the pregnancy progresses.
New diagnoses included for surveillance
Several diagnoses not previously included now qualify for antepartum fetal surveillance under the new guidelines, most notably:
- history of obstetrical complications in the immediate preceding pregnancy
—history of prior fetal growth restriction requiring preterm delivery
—history of prior preeclampsia requiring delivery
- alcohol use of 5 or more drinks per day
- in vitro fertilization
- abnormal serum markers
—pregnancy-associated plasma protein A (PAPP-A) in the fifth or lower percentile or 0.4 multiples of the median (MoM)
—second trimester inhibin A of 2 or greater MoM
- prepregnancy body mass index (BMI)
—this is divided into 2 categories for timing of initiation of testing:
- 37 weeks for BMI of 35 to 39.9 kg/m2
- 34 weeks for BMI of or greater than 40 kg/m2.
Fetal kick counts
The major change to the updated Practice Bulletin on antenatal surveillance is the inclusion of data on FKCs, a simple modality of fetal surveillance that does not require a clinical visit. For FKCs, a meta-analysis of more than 450,000 patients did not demonstrate a difference in perinatal death between the FKC intervention group (0.54%) and the control group (0.59%). Of note, there were small but statistically significant increases in the rates of induction of labor, cesarean delivery, and preterm delivery in the FKC intervention group. Therefore, this update does not recommend a formal program of FKCs for all patients.
- The antenatal fetal surveillance guidelines are just that—guidelines, not mandates. Their use will need to be adapted for specific patient populations and practice management patterns.
- Many conditions qualify for “individualized” surveillance, which offers the opportunity for detailed discussions on the patient’s care. This includes shared decision making with patients to meet their goals for the pregnancy.
- Although patient-perceived decreased fetal movement always warrants clinical evaluation, a regular program of fetal kick count monitoring is not recommended for all patients due to lack of data supporting its benefit in reducing perinatal death.
- As with any change, new guidelines potentially are a source of frustration, so a concerted effort by obstetrical clinicians to agree on adoption of the guidelines is needed. Additional clinical resources and both clinician and patient education may be required depending on current practice style, as the new strategy may increase the number of appointments and ultrasound exams required.
Continue to: Use of antenatal corticosteroids now may be considered at 22 weeks’ gestation...
Use of antenatal corticosteroids now may be considered at 22 weeks’ gestation
American College of Obstetricians and Gynecologists. Use of antenatal corticosteroids at 22 weeks of gestation: ACOG practice advisory. September 2021. https://www .acog.org/clinical/clinical-guidance/practice-advisory /articles/2021/09/use-of-antenatal-corticosteroids-at -22-weeks-of-gestation. Accessed December 11, 2021.
In September 2021, ACOG and SMFM released a Practice Advisory updating the current recommendations for the administration of antenatal corticosteroids in the periviable period (22 to 25 6/7 weeks’ gestation). Whereas the prior lower limit of gestational age for consideration of steroids was 23 weeks, the new recommendation now extends this consideration down to 22 weeks.
The cited data include a meta-analysis of more than 2,200 patients in which the survival rate of infants born between 22 and 22 6/7 weeks who were exposed to antenatal steroids was 39% compared with 19.5% in the unexposed group. Another study of more than 1,000 patients demonstrated a statistically significant increase in overall survival in patients treated with antenatal steroids plus life support compared with life support only (38.5% vs 17.7%). Survival without major morbidity in this study, although increased from 1% to 4.4%, was still low.
Recommendation carries caveats
Given this information, the Practice Advisory offers a 2C level recommendation (weak recommendation, low quality of evidence) for antenatal steroids at 22 to 22 6/7 weeks’ gestation if neonatal resuscitation is planned, acknowledging the limitations and potential bias of the available data.
The Practice Advisory emphasizes the importance of counseling and patient involvement in the decision making. This requires a multidisciplinary collaboration among the neonatology and obstetrical teams, flexibility in the plan after birth depending on the infant’s condition, and redirection of care if appropriate. Estimated fetal weight, the presence of multiple gestations, fetal biologic sex, and any anomalies are also important in helping families make an informed decision for their particular pregnancy. As described in the Obstetric Care Consensus on periviable birth,6 it is important to remember that considerations and recommendations are not the same as requirements, and redirection of care to comfort and family memory making is not the same as withholding care.
The rest of the recommendations for the administration of antenatal steroids remain the same: Antenatal steroids are not recommended at less than 22 weeks due to lack of evidence of benefit, and they continue to be recommended at 24 weeks and beyond. ●
- Antenatal corticosteroids may be considered at 22 to 22 6/7 weeks’ gestation if, after thorough patient counseling, neonatal resuscitation is desired and planned by the family.
- The overall likelihood of survival and survival without major morbidities continues to be very low in the periviable period, especially at 22 weeks. Gestational age is only one of the many factors that must be considered in the shared decision making for this very difficult decision.
- Palliative care is a valid and appropriate option for patients facing a periviable delivery after appropriate counseling or after evaluation of the infant has occurred after birth.
- Martin JA, Hamilton BE, Osterman MJK. Births in the United States, 2019. NCHS Data Brief, no 387. Hyattsville, MD: National Center for Health Statistics. October 2020. www.cdc.gov/nchs /data/databriefs/db387-H.pdf. Accessed December 20, 2021.
- To MS, Skentou C, Chan C, et al. Cervical assessment at the routine 23-week scan: standardizing techniques. Ultrasound Obstet Gynecol. 2001;17:217-219.
- American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins–Obstetrics. Prediction and prevention of spontaneous preterm birth: ACOG practice bulletin, number 234. Obstet Gynecol. 2021;138:e65-e90.
- Society for Maternal-Fetal Medicine (SMFM) Publications Committee. SMFM statement: use of 17-alpha hydroxyprogesterone caproate for prevention of recurrent preterm birth. Am J Obstet Gynecol. 2020;223:B16-B18.
- Blackwell SC, Gyamfi-Bannerman C, Biggio JR Jr, et al. 17-OHPC to prevent recurrent preterm birth in singleton gestations (PROLONG study): a multicenter, international, randomized double-blind trial. Am J Perinatol. 2020;37:127-136.
- American College of Obstetricians and Gynecologists; Society for Maternal-Fetal Medicine. Obstetric care consensus no. 6: Periviable birth. Obstet Gynecol. 2017;130:e187-e199.
- Martin JA, Hamilton BE, Osterman MJK. Births in the United States, 2019. NCHS Data Brief, no 387. Hyattsville, MD: National Center for Health Statistics. October 2020. www.cdc.gov/nchs /data/databriefs/db387-H.pdf. Accessed December 20, 2021.
- To MS, Skentou C, Chan C, et al. Cervical assessment at the routine 23-week scan: standardizing techniques. Ultrasound Obstet Gynecol. 2001;17:217-219.
- American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins–Obstetrics. Prediction and prevention of spontaneous preterm birth: ACOG practice bulletin, number 234. Obstet Gynecol. 2021;138:e65-e90.
- Society for Maternal-Fetal Medicine (SMFM) Publications Committee. SMFM statement: use of 17-alpha hydroxyprogesterone caproate for prevention of recurrent preterm birth. Am J Obstet Gynecol. 2020;223:B16-B18.
- Blackwell SC, Gyamfi-Bannerman C, Biggio JR Jr, et al. 17-OHPC to prevent recurrent preterm birth in singleton gestations (PROLONG study): a multicenter, international, randomized double-blind trial. Am J Perinatol. 2020;37:127-136.
- American College of Obstetricians and Gynecologists; Society for Maternal-Fetal Medicine. Obstetric care consensus no. 6: Periviable birth. Obstet Gynecol. 2017;130:e187-e199.
The troubling trend of repackaging feminine hygiene products for the next generation
Feminine hygiene products have been commercially available for decades. They are commonly marketed to reduce odor or clean vaginal discharge and menses. Multiple formulas are available as topical washes, wipes, creams, sprays, powders, deodorants, and douches.1 Products on the market range from those used externally on the vulva, such as wipes and sprays, to liquid solutions used intravaginally, such as washes and douches.
Who uses feminine hygiene products?
According to a 2006 study, the majority of women who use douches started using them between age 15 and 19 years, but some women initiate this practice habit as early as age 10 to 14.1 Predictably, women who douche are more likely to perceive douche products as safe.1
Demographic data on douche utilization are mixed: Some studies show that there are no significant racial differences in douching practices,2 while others have found that Black and African American women are more likely to practice douching than White and Hispanic women.1,3 Studies have shown a significant difference in attitudes toward douching and knowledge of normal vaginal symptoms among US racial demographics, although this must be examined through the historical context of racism and the lens of medical anthropology.4
Women cite that common reasons they use feminine hygiene products are to feel clean, to control odor, and to use after menses and intercourse.1,2
Modern marketing approaches
From wipes to soaps to douches, feminine hygiene products often are advertised to promote “funk-free periods”5 and “freshness,” fostering an environment in which women and men develop unrealistic standards for what is considered normal genital odor and resulting in poor body image.6
Recently, Vagisil (Combe Incorporated) marketing efforts faced backlash from the ObGyn community for targeting younger populations with a specific product line for adolescents called OMV! In addition, attention has been drawn to VCF vaginal odor eliminating film (Apothecus Pharmaceutical Corp), small stamp-sized dissolving films that are placed in the vaginal canal in contact with the epithelium. This product has entered the market of feminine hygiene products accompanied by slogans of eliminating “feminine odor” and providing “confidence for women to be intimate.”
Continue to: Effects of feminine hygiene products on the vaginal microbiome...
Effects of feminine hygiene products on the vaginal microbiome
Frequent use of feminine hygiene products has been associated with recurrent vaginitis, bacterial vaginosis, and general irritation/itch,7,8 which can cause more discharge and odor. Ironically, this may result in women using the product more frequently since they often seek out these products to eliminate odor and discharge.1,2
The pH of the vagina changes during a woman’s lifetime, but in the reproductive years, the normal pH range is typically 3.8 to 4.4.9 This range allows for a normal vaginal flora to form with bacteria such as Lactobacillus species and Gardnerella vaginalis, while feminine hygiene products have a wide range of pH.9,10
Regardless of the formulation, most feminine hygiene products contain ingredients and compositions that potentially are detrimental to the health of the vulva and vagina. Many products contain acidic ingredients, such as citric acid, lactic acid, and dehydroacetic acid, that can alter the vaginal pH and weaken the vaginal barrier by wiping out normal vaginal flora10 despite being advertised for use on “sensitive areas” (TABLE). Lactic acid also has been found to increase diverse anaerobic bacteria in the vaginal microbiome.11 Some feminine hygiene products have been shown to suppress Lactobacillus growth at 2 hours after use and to kill all lactobacilli at 24 hours.10 Shifts in microbiota numbers often occur when the vaginal pH has been altered, as is frequently the case with feminine hygiene products. In the absence of microbiome bacteria, the presence of vaginal hygiene products has been shown to increase interleukin-8 (IL-8), suggesting a proinflammatory reaction.10

A study in the United Kingdom found that women who used bubble bath, antiseptics, or douche products had a higher incidence of bacterial vaginosis compared with women who did not use such products.7 Women in Canada who used feminine hygiene products were more likely to report adverse conditions, including yeast infections, bacterial vaginosis, urinary tract infections, and sexually transmitted diseases.8 Furthermore, a significant association exists between vaginal douching and endometrial infection by bacterial vaginosis–associated organisms.12
Additionally, a study that analyzed volatile organic compound levels in the blood with the use of feminine hygiene products revealed a significant positive dose-exposure relationship between the frequency of vaginal douching in the last 6 months and concentrations of 1,4-dichloromethane, one of the volatile organic compounds.3 This points to the issue of not only disruption of pH and vaginal flora but also to the introduction of harmful substances that can further disrupt the vaginal barrier.
Understand the products to help educate patients
Use of feminine hygiene products is common among women. While women depend on the market to filter out products that are considered unsafe or may have harmful side effects,1 unfortunately that is not necessarily the case. With increasingly more feminine products on the market and the target demographic becoming younger, women of all ages are susceptible to misinformation that could affect their vaginal health long term.
It is vital that clinicians understand the topical effects of these products in order to properly educate and counsel patients. Ultimately, research on feminine hygiene products is limited and, as more products come to market, we must continue to reassess the effects of topical products on the vaginal epithelium and vulvar tissues. ●
- Grimley DM, Annang L, Foushee HR, et al. Vaginal douches and other feminine hygiene products: women’s practices and perceptions of product safety. Matern Child Health J. 2006;10:303-310. doi: 10.1007/s10995-005-0054-y.
- Foch BJ, McDaniel ND, Chacko MR. Racial differences in vaginal douching knowledge, attitude, and practices among sexually active adolescents. J Pediatr Adolesc Gynecol. 2001;14:29-33. doi: 10.1016/S1083-3188(00)00080-2.
- Lin N, Ding N, Meza-Wilson E, et al. Volatile organic compounds in feminine hygiene products sold in the US market: a survey of products and health risks. Environ Int. 2020;144:105740. doi: 10.1016/j.envint.2020.105740.
- Wayne State University Digital Commons. Guy-Lee AK. Rituals reproducing race: African American women’s feminine hygiene practices, shared experiences, and power. 2017. http://digitalcommons.wayne.edu/oa_dissertations/1806. Accessed December 13, 2021.
- YouTube. OMV! by Vagisil—Intimate care products designed by teens. July 10, 2020. www.youtube.com/ watch?v=VkVsCagrAw0. Accessed December 13, 2021.
- Jenkins A, O’Doherty KC. The clean vagina, the healthy vagina, and the dirty vagina: exploring women’s portrayals of the vagina in relation to vaginal cleansing product use. Fem Psychol. 2021;31:192-211. doi: 10.1177/0959353520944144.
- Rajamanoharan S, Low N, Jones SB, et al. Bacterial vaginosis, ethnicity, and the use of genital cleansing agents: a case control study. Sex Transm Dis. 1999;26:404-409.
- Crann SE, Cunningham S, Albert A, et al. Vaginal health and hygiene practices and product use in Canada: a national cross-sectional survey. BMC Womens Health. 2018;18:52. doi: 10.1186/s12905-018-0543-y.
- Chen Y, Bruning E, Rubino J, et al. Role of female intimate hygiene in vulvovaginal health: global hygiene practices and product usage. Womens Health (London). 2017;13:58-67. doi: 10.1177/1745505717731011.
- Fashemi B, Delaney ML, Onderdonk AB, et al. Effects of feminine hygiene products on the vaginal mucosal biome. Microb Ecol Health Dis. 2013;24. doi: 10.3402/mehd. v24i0.19703.
- Van der Veer C, Bruisten SM, Van Houdt R, et al. Effects of an over-the-counter lactic-acid containing intra-vaginal douching product on the vaginal microbiota. BMC Microbiol. 2019;19:168. doi: 10.1186/s12866-019-1545-0.
- Gondwe T, Ness R, Totten PA, et al. Novel bacterial vaginosis-associated organisms mediate the relationship between vaginal douching and pelvic inflammatory disease. Sex Transm Infect. 2020;96:439-444. doi: 10.1136/ sextrans-2019-054191.
Feminine hygiene products have been commercially available for decades. They are commonly marketed to reduce odor or clean vaginal discharge and menses. Multiple formulas are available as topical washes, wipes, creams, sprays, powders, deodorants, and douches.1 Products on the market range from those used externally on the vulva, such as wipes and sprays, to liquid solutions used intravaginally, such as washes and douches.
Who uses feminine hygiene products?
According to a 2006 study, the majority of women who use douches started using them between age 15 and 19 years, but some women initiate this practice habit as early as age 10 to 14.1 Predictably, women who douche are more likely to perceive douche products as safe.1
Demographic data on douche utilization are mixed: Some studies show that there are no significant racial differences in douching practices,2 while others have found that Black and African American women are more likely to practice douching than White and Hispanic women.1,3 Studies have shown a significant difference in attitudes toward douching and knowledge of normal vaginal symptoms among US racial demographics, although this must be examined through the historical context of racism and the lens of medical anthropology.4
Women cite that common reasons they use feminine hygiene products are to feel clean, to control odor, and to use after menses and intercourse.1,2
Modern marketing approaches
From wipes to soaps to douches, feminine hygiene products often are advertised to promote “funk-free periods”5 and “freshness,” fostering an environment in which women and men develop unrealistic standards for what is considered normal genital odor and resulting in poor body image.6
Recently, Vagisil (Combe Incorporated) marketing efforts faced backlash from the ObGyn community for targeting younger populations with a specific product line for adolescents called OMV! In addition, attention has been drawn to VCF vaginal odor eliminating film (Apothecus Pharmaceutical Corp), small stamp-sized dissolving films that are placed in the vaginal canal in contact with the epithelium. This product has entered the market of feminine hygiene products accompanied by slogans of eliminating “feminine odor” and providing “confidence for women to be intimate.”
Continue to: Effects of feminine hygiene products on the vaginal microbiome...
Effects of feminine hygiene products on the vaginal microbiome
Frequent use of feminine hygiene products has been associated with recurrent vaginitis, bacterial vaginosis, and general irritation/itch,7,8 which can cause more discharge and odor. Ironically, this may result in women using the product more frequently since they often seek out these products to eliminate odor and discharge.1,2
The pH of the vagina changes during a woman’s lifetime, but in the reproductive years, the normal pH range is typically 3.8 to 4.4.9 This range allows for a normal vaginal flora to form with bacteria such as Lactobacillus species and Gardnerella vaginalis, while feminine hygiene products have a wide range of pH.9,10
Regardless of the formulation, most feminine hygiene products contain ingredients and compositions that potentially are detrimental to the health of the vulva and vagina. Many products contain acidic ingredients, such as citric acid, lactic acid, and dehydroacetic acid, that can alter the vaginal pH and weaken the vaginal barrier by wiping out normal vaginal flora10 despite being advertised for use on “sensitive areas” (TABLE). Lactic acid also has been found to increase diverse anaerobic bacteria in the vaginal microbiome.11 Some feminine hygiene products have been shown to suppress Lactobacillus growth at 2 hours after use and to kill all lactobacilli at 24 hours.10 Shifts in microbiota numbers often occur when the vaginal pH has been altered, as is frequently the case with feminine hygiene products. In the absence of microbiome bacteria, the presence of vaginal hygiene products has been shown to increase interleukin-8 (IL-8), suggesting a proinflammatory reaction.10



A study in the United Kingdom found that women who used bubble bath, antiseptics, or douche products had a higher incidence of bacterial vaginosis compared with women who did not use such products.7 Women in Canada who used feminine hygiene products were more likely to report adverse conditions, including yeast infections, bacterial vaginosis, urinary tract infections, and sexually transmitted diseases.8 Furthermore, a significant association exists between vaginal douching and endometrial infection by bacterial vaginosis–associated organisms.12
Additionally, a study that analyzed volatile organic compound levels in the blood with the use of feminine hygiene products revealed a significant positive dose-exposure relationship between the frequency of vaginal douching in the last 6 months and concentrations of 1,4-dichloromethane, one of the volatile organic compounds.3 This points to the issue of not only disruption of pH and vaginal flora but also to the introduction of harmful substances that can further disrupt the vaginal barrier.
Understand the products to help educate patients
Use of feminine hygiene products is common among women. While women depend on the market to filter out products that are considered unsafe or may have harmful side effects,1 unfortunately that is not necessarily the case. With increasingly more feminine products on the market and the target demographic becoming younger, women of all ages are susceptible to misinformation that could affect their vaginal health long term.
It is vital that clinicians understand the topical effects of these products in order to properly educate and counsel patients. Ultimately, research on feminine hygiene products is limited and, as more products come to market, we must continue to reassess the effects of topical products on the vaginal epithelium and vulvar tissues. ●
Feminine hygiene products have been commercially available for decades. They are commonly marketed to reduce odor or clean vaginal discharge and menses. Multiple formulas are available as topical washes, wipes, creams, sprays, powders, deodorants, and douches.1 Products on the market range from those used externally on the vulva, such as wipes and sprays, to liquid solutions used intravaginally, such as washes and douches.
Who uses feminine hygiene products?
According to a 2006 study, the majority of women who use douches started using them between age 15 and 19 years, but some women initiate this practice habit as early as age 10 to 14.1 Predictably, women who douche are more likely to perceive douche products as safe.1
Demographic data on douche utilization are mixed: Some studies show that there are no significant racial differences in douching practices,2 while others have found that Black and African American women are more likely to practice douching than White and Hispanic women.1,3 Studies have shown a significant difference in attitudes toward douching and knowledge of normal vaginal symptoms among US racial demographics, although this must be examined through the historical context of racism and the lens of medical anthropology.4
Women cite that common reasons they use feminine hygiene products are to feel clean, to control odor, and to use after menses and intercourse.1,2
Modern marketing approaches
From wipes to soaps to douches, feminine hygiene products often are advertised to promote “funk-free periods”5 and “freshness,” fostering an environment in which women and men develop unrealistic standards for what is considered normal genital odor and resulting in poor body image.6
Recently, Vagisil (Combe Incorporated) marketing efforts faced backlash from the ObGyn community for targeting younger populations with a specific product line for adolescents called OMV! In addition, attention has been drawn to VCF vaginal odor eliminating film (Apothecus Pharmaceutical Corp), small stamp-sized dissolving films that are placed in the vaginal canal in contact with the epithelium. This product has entered the market of feminine hygiene products accompanied by slogans of eliminating “feminine odor” and providing “confidence for women to be intimate.”
Continue to: Effects of feminine hygiene products on the vaginal microbiome...
Effects of feminine hygiene products on the vaginal microbiome
Frequent use of feminine hygiene products has been associated with recurrent vaginitis, bacterial vaginosis, and general irritation/itch,7,8 which can cause more discharge and odor. Ironically, this may result in women using the product more frequently since they often seek out these products to eliminate odor and discharge.1,2
The pH of the vagina changes during a woman’s lifetime, but in the reproductive years, the normal pH range is typically 3.8 to 4.4.9 This range allows for a normal vaginal flora to form with bacteria such as Lactobacillus species and Gardnerella vaginalis, while feminine hygiene products have a wide range of pH.9,10
Regardless of the formulation, most feminine hygiene products contain ingredients and compositions that potentially are detrimental to the health of the vulva and vagina. Many products contain acidic ingredients, such as citric acid, lactic acid, and dehydroacetic acid, that can alter the vaginal pH and weaken the vaginal barrier by wiping out normal vaginal flora10 despite being advertised for use on “sensitive areas” (TABLE). Lactic acid also has been found to increase diverse anaerobic bacteria in the vaginal microbiome.11 Some feminine hygiene products have been shown to suppress Lactobacillus growth at 2 hours after use and to kill all lactobacilli at 24 hours.10 Shifts in microbiota numbers often occur when the vaginal pH has been altered, as is frequently the case with feminine hygiene products. In the absence of microbiome bacteria, the presence of vaginal hygiene products has been shown to increase interleukin-8 (IL-8), suggesting a proinflammatory reaction.10



A study in the United Kingdom found that women who used bubble bath, antiseptics, or douche products had a higher incidence of bacterial vaginosis compared with women who did not use such products.7 Women in Canada who used feminine hygiene products were more likely to report adverse conditions, including yeast infections, bacterial vaginosis, urinary tract infections, and sexually transmitted diseases.8 Furthermore, a significant association exists between vaginal douching and endometrial infection by bacterial vaginosis–associated organisms.12
Additionally, a study that analyzed volatile organic compound levels in the blood with the use of feminine hygiene products revealed a significant positive dose-exposure relationship between the frequency of vaginal douching in the last 6 months and concentrations of 1,4-dichloromethane, one of the volatile organic compounds.3 This points to the issue of not only disruption of pH and vaginal flora but also to the introduction of harmful substances that can further disrupt the vaginal barrier.
Understand the products to help educate patients
Use of feminine hygiene products is common among women. While women depend on the market to filter out products that are considered unsafe or may have harmful side effects,1 unfortunately that is not necessarily the case. With increasingly more feminine products on the market and the target demographic becoming younger, women of all ages are susceptible to misinformation that could affect their vaginal health long term.
It is vital that clinicians understand the topical effects of these products in order to properly educate and counsel patients. Ultimately, research on feminine hygiene products is limited and, as more products come to market, we must continue to reassess the effects of topical products on the vaginal epithelium and vulvar tissues. ●
- Grimley DM, Annang L, Foushee HR, et al. Vaginal douches and other feminine hygiene products: women’s practices and perceptions of product safety. Matern Child Health J. 2006;10:303-310. doi: 10.1007/s10995-005-0054-y.
- Foch BJ, McDaniel ND, Chacko MR. Racial differences in vaginal douching knowledge, attitude, and practices among sexually active adolescents. J Pediatr Adolesc Gynecol. 2001;14:29-33. doi: 10.1016/S1083-3188(00)00080-2.
- Lin N, Ding N, Meza-Wilson E, et al. Volatile organic compounds in feminine hygiene products sold in the US market: a survey of products and health risks. Environ Int. 2020;144:105740. doi: 10.1016/j.envint.2020.105740.
- Wayne State University Digital Commons. Guy-Lee AK. Rituals reproducing race: African American women’s feminine hygiene practices, shared experiences, and power. 2017. http://digitalcommons.wayne.edu/oa_dissertations/1806. Accessed December 13, 2021.
- YouTube. OMV! by Vagisil—Intimate care products designed by teens. July 10, 2020. www.youtube.com/ watch?v=VkVsCagrAw0. Accessed December 13, 2021.
- Jenkins A, O’Doherty KC. The clean vagina, the healthy vagina, and the dirty vagina: exploring women’s portrayals of the vagina in relation to vaginal cleansing product use. Fem Psychol. 2021;31:192-211. doi: 10.1177/0959353520944144.
- Rajamanoharan S, Low N, Jones SB, et al. Bacterial vaginosis, ethnicity, and the use of genital cleansing agents: a case control study. Sex Transm Dis. 1999;26:404-409.
- Crann SE, Cunningham S, Albert A, et al. Vaginal health and hygiene practices and product use in Canada: a national cross-sectional survey. BMC Womens Health. 2018;18:52. doi: 10.1186/s12905-018-0543-y.
- Chen Y, Bruning E, Rubino J, et al. Role of female intimate hygiene in vulvovaginal health: global hygiene practices and product usage. Womens Health (London). 2017;13:58-67. doi: 10.1177/1745505717731011.
- Fashemi B, Delaney ML, Onderdonk AB, et al. Effects of feminine hygiene products on the vaginal mucosal biome. Microb Ecol Health Dis. 2013;24. doi: 10.3402/mehd. v24i0.19703.
- Van der Veer C, Bruisten SM, Van Houdt R, et al. Effects of an over-the-counter lactic-acid containing intra-vaginal douching product on the vaginal microbiota. BMC Microbiol. 2019;19:168. doi: 10.1186/s12866-019-1545-0.
- Gondwe T, Ness R, Totten PA, et al. Novel bacterial vaginosis-associated organisms mediate the relationship between vaginal douching and pelvic inflammatory disease. Sex Transm Infect. 2020;96:439-444. doi: 10.1136/ sextrans-2019-054191.
- Grimley DM, Annang L, Foushee HR, et al. Vaginal douches and other feminine hygiene products: women’s practices and perceptions of product safety. Matern Child Health J. 2006;10:303-310. doi: 10.1007/s10995-005-0054-y.
- Foch BJ, McDaniel ND, Chacko MR. Racial differences in vaginal douching knowledge, attitude, and practices among sexually active adolescents. J Pediatr Adolesc Gynecol. 2001;14:29-33. doi: 10.1016/S1083-3188(00)00080-2.
- Lin N, Ding N, Meza-Wilson E, et al. Volatile organic compounds in feminine hygiene products sold in the US market: a survey of products and health risks. Environ Int. 2020;144:105740. doi: 10.1016/j.envint.2020.105740.
- Wayne State University Digital Commons. Guy-Lee AK. Rituals reproducing race: African American women’s feminine hygiene practices, shared experiences, and power. 2017. http://digitalcommons.wayne.edu/oa_dissertations/1806. Accessed December 13, 2021.
- YouTube. OMV! by Vagisil—Intimate care products designed by teens. July 10, 2020. www.youtube.com/ watch?v=VkVsCagrAw0. Accessed December 13, 2021.
- Jenkins A, O’Doherty KC. The clean vagina, the healthy vagina, and the dirty vagina: exploring women’s portrayals of the vagina in relation to vaginal cleansing product use. Fem Psychol. 2021;31:192-211. doi: 10.1177/0959353520944144.
- Rajamanoharan S, Low N, Jones SB, et al. Bacterial vaginosis, ethnicity, and the use of genital cleansing agents: a case control study. Sex Transm Dis. 1999;26:404-409.
- Crann SE, Cunningham S, Albert A, et al. Vaginal health and hygiene practices and product use in Canada: a national cross-sectional survey. BMC Womens Health. 2018;18:52. doi: 10.1186/s12905-018-0543-y.
- Chen Y, Bruning E, Rubino J, et al. Role of female intimate hygiene in vulvovaginal health: global hygiene practices and product usage. Womens Health (London). 2017;13:58-67. doi: 10.1177/1745505717731011.
- Fashemi B, Delaney ML, Onderdonk AB, et al. Effects of feminine hygiene products on the vaginal mucosal biome. Microb Ecol Health Dis. 2013;24. doi: 10.3402/mehd. v24i0.19703.
- Van der Veer C, Bruisten SM, Van Houdt R, et al. Effects of an over-the-counter lactic-acid containing intra-vaginal douching product on the vaginal microbiota. BMC Microbiol. 2019;19:168. doi: 10.1186/s12866-019-1545-0.
- Gondwe T, Ness R, Totten PA, et al. Novel bacterial vaginosis-associated organisms mediate the relationship between vaginal douching and pelvic inflammatory disease. Sex Transm Infect. 2020;96:439-444. doi: 10.1136/ sextrans-2019-054191.
Buccal Fat Pad Reduction With Intraoperative Fat Transfer to the Temple
The buccal fat pad (Bichat fat pad) is a tubular-shaped collection of adipose tissue that occupies a prominent position in the midface. The buccal fat pad has been described as having 3 lobes: an anterior lobe, which is anterior to the masseter muscle; an intermediate lobe between the masseter and buccinator muscles; and a posterior lobe between the temporal masticatory space.1 There are 4 extensions from the body of the buccal fat pad: the buccal, the sublevator, the melolabial, and the pterygoid. It is the buccal extension and main body that are removed intraorally to achieve midfacial and lower facial contouring, as these support the contours of the cheeks. The deep fat pad within the temporal fossa is a true extension of the buccal fat pad (Figure).2 It has a complex relationship to the facial structures, with known variability in the positions of the buccal branch of the facial nerve and the parotid duct.3 The parotid duct travels over, superior to, or through the buccal extension 42%, 32%, and 26% of the time, respectively. The duct travels along the surface of the masseter, then pierces the buccinator to drain into the vestibule of the mouth at the second superior molar tooth. The buccal branch of the facial nerve travels on the surface of the buccal fat pad 73% of the time, whereas 27% of the time it travels deeper through the buccal extension.4 A study that used ultrasonography to map the surface anatomy path of the parotid duct in 50 healthy patients showed that the duct was within 1.5 cm of the middle half of a line between the lower border of the tragus and the oral commissure in 93% of individuals.5 We describe a technique in which part of the buccal fat pad is removed and the fat is transferred to the temple to achieve aesthetically pleasing facial contouring. We used a vertical line from the lateral canthus as a surface anatomy landmark to determine when the duct emerges from the gland and is most susceptible to injury.
Operative Technique
Correct instrumentation is important to obtain appropriate anatomic exposure for this procedure. The surgical tray should include 4-0 poliglecaprone 25 suture, bite guards, a needle driver, a hemostat, surgical scissors, toothed forceps, a Beaver surgical handle with #15 blade, a protected diathermy needle, cotton tip applicators, and gauze.
Fat Harvest—With the patient supine, bite blocks are placed, and the buccal fat pad incision line is marked with a surgical marker. A 1-cm line is drawn approximately 4 cm posterior to the oral commissure by the buccal bite marks. The location is verified by balloting externally on the buccal fat pad on the cheek. The incision line is then anesthetized transorally with lidocaine and epinephrine-containing solution. The cheek is retracted laterally with Caldwell-Luc retractors, and a 1-cm incision is made and carried through the mucosa and superficial muscle using the Colorado needle. Scissors are then used to spread the deeper muscle fibers to expose the deeper fascia and fat pads. Metzenbaum scissors are used to gently spread the fat while the surgeon places pressure on the external cheek, manipulating the fat into the wound. Without excess traction, the walnut-sized portion of the fat pad that protrudes is grasped with Debakey forceps, gently teased into the field, clamped at its base with a curved hemostat, and excised. The stump is electrocoagulated with an extendable protected Colorado needle, with care to prevent inadvertent cauterization of the lips. The wound is closed with a single 4-0 poliglecaprone-25 suture.
A 5-cc Luer lock syringe is preloaded with 2 cc of normal saline and attached to another 5-cc Luer lock syringe via a female-female attachment. The excised fat is then placed in a 5-cc Luer lock syringe by removing the plunger. The plunger is then reinstalled, and the fat is injected back and forth approximately 30 times. The fat is centrifuged at 3500 rpm for 3 minutes. The purified fat is then transferred to a 1-cc Luer lock syringe attached to an 18-gauge needle.
Fat Injection—The authors use an 18-gauge needle to perform depot injections into the temporal fossae above the periosteum. This is a relatively safe area of the face to inject, but care must be taken to avoid injury to the superficial temporal artery. Between 1.5 and 3 cc of high-quality fat usually are administered to each temple.
Aftercare Instructions—The patient is instructed to have a soft diet for 24 to 48 hours and can return to work the next day. The patient also is given prophylactic antibiotics with Gram-negative coverage for 7 days (amoxicillin-clavulanate 875 mg/125 mg orally twice daily for 7 days).
Candidates for Buccal Fat Pad Reduction
Buccal fat pad reduction has become an increasingly popular technique for midface and lower face shaping to decrease the appearance of a round face. To achieve an aesthetically pleasing midface, surgeons should consider enhancing zygomatic eminences while emphasizing the border between the zygomatic prominence and cheek hollow.6 Selection criteria for buccal fat pad reduction are not well established. One study recommended avoiding the procedure in pregnant or lactating patients, patients with chronic illnesses, patients on blood-thinning agents, and patients younger than 18 years. In addition, this study suggested ensuring the malar fullness is in the anteromedial portion of the face, as posterolateral fullness may be due to masseter hypertrophy.6
Complications From Buccal Fat Pad Reduction
Complications associated with buccal fat pad reduction include inadvertent damage to surrounding structures, including the buccal branch of the facial nerve and parotid duct. Because the location of the facial nerve in relation to the parotid duct is highly variable, surgeons must be aware of its anatomy to avoid unintentional damage. Hwang et al7 reported that the parotid duct and buccal branches of the facial nerves passed through the buccal extension in 26.3% of cadavers. The transbuccal approach is preferred over the sub–superficial muscular aponeurotic system approach largely because it avoids these structures. In addition, blunt dissection may further decrease chances of injury. Although the long-term effects are unknown, there is a potential risk for facial hollowing.3 The use of preprocedure ultrasonography to quantify the buccal fat pad may avoid overresection and enhanced potential for facial hollowing.6
Avoidance of Temporal Hollowing
Because the buccal fat pad extends into the temporal space, buccal fat pad reduction may lead to further temporal hollowing, contributing to an aged appearance. The authors’ technique addresses both midface and upper face contouring in one minimally invasive procedure. Temporal hollowing commonly has been corrected with autologous fat grafting from the thigh or abdomen, which leads to an additional scar at the donor site. Our technique relies on autologous adjacent fat transfer from previously removed buccal fat. In addition, compared with the use of hyaluronic acid fillers for temple reflation, fat transfer largely is safe and biocompatible. Major complications of autologous fat transfer to the temples include nodularity or fat clumping, fat necrosis, sensory or motor nerve damage, and edema or ecchymosis.4 Also, with time there will be ongoing hollowing of the temples as part of the aging process with soft tissue and bone resorption. Therefore, further volume restoration procedures may be required in the future to address these dynamic changes.
Conclusion
The buccal fat pad has been extensively used to reconstruct oral defects, including oroantral and cranial base defects, owing to its high vascularity.6 However, there also is great potential to utilize buccal fat for autologous fat transfer to improve temporal wasting. Further studies are needed to determine optimal technique as well as longer-term safety and efficacy of this procedure.
- Zhang HM, Yan YP, Qi KM, et al. Anatomical structure of the buccal fat pad and its clinical adaptations. Plast Reconstr Surg. 2002;109:2509-2518.
- Yousuf S, Tubbs RS, Wartmann CT, et al. A review of the gross anatomy, functions, pathology, and clinical uses of the buccal fat pad. Surg Radiol Anat. 2010;32:427-436.
- Benjamin M, Reish RG. Buccal fat pad excision: proceed with caution. Plast Reconstr Surg Glob Open. 2018;6:E1970.
- Tzikas TL. Fat grafting volume restoration to the brow and temporal regions. Facial Plast Surg. 2018;34:164-172.
- Stringer MD, Mirjalili SA, Meredith SJ, et al. Redefining the surface anatomy of the parotid duct: an in vivo ultrasound study. Plast Reconstr Surg. 2012;130:1032-1037.
- Sezgin B, Tatar S, Boge M, et al. The excision of the buccal fat pad for cheek refinement: volumetric considerations. Aesthet Surg J. 2019;39:585-592.
- Hwang K, Cho HJ, Battuvshin D, et al. Interrelated buccal fat pad with facial buccal branches and parotid duct. J Craniofac Surg. 2005;16:658-660.
The buccal fat pad (Bichat fat pad) is a tubular-shaped collection of adipose tissue that occupies a prominent position in the midface. The buccal fat pad has been described as having 3 lobes: an anterior lobe, which is anterior to the masseter muscle; an intermediate lobe between the masseter and buccinator muscles; and a posterior lobe between the temporal masticatory space.1 There are 4 extensions from the body of the buccal fat pad: the buccal, the sublevator, the melolabial, and the pterygoid. It is the buccal extension and main body that are removed intraorally to achieve midfacial and lower facial contouring, as these support the contours of the cheeks. The deep fat pad within the temporal fossa is a true extension of the buccal fat pad (Figure).2 It has a complex relationship to the facial structures, with known variability in the positions of the buccal branch of the facial nerve and the parotid duct.3 The parotid duct travels over, superior to, or through the buccal extension 42%, 32%, and 26% of the time, respectively. The duct travels along the surface of the masseter, then pierces the buccinator to drain into the vestibule of the mouth at the second superior molar tooth. The buccal branch of the facial nerve travels on the surface of the buccal fat pad 73% of the time, whereas 27% of the time it travels deeper through the buccal extension.4 A study that used ultrasonography to map the surface anatomy path of the parotid duct in 50 healthy patients showed that the duct was within 1.5 cm of the middle half of a line between the lower border of the tragus and the oral commissure in 93% of individuals.5 We describe a technique in which part of the buccal fat pad is removed and the fat is transferred to the temple to achieve aesthetically pleasing facial contouring. We used a vertical line from the lateral canthus as a surface anatomy landmark to determine when the duct emerges from the gland and is most susceptible to injury.

Operative Technique
Correct instrumentation is important to obtain appropriate anatomic exposure for this procedure. The surgical tray should include 4-0 poliglecaprone 25 suture, bite guards, a needle driver, a hemostat, surgical scissors, toothed forceps, a Beaver surgical handle with #15 blade, a protected diathermy needle, cotton tip applicators, and gauze.
Fat Harvest—With the patient supine, bite blocks are placed, and the buccal fat pad incision line is marked with a surgical marker. A 1-cm line is drawn approximately 4 cm posterior to the oral commissure by the buccal bite marks. The location is verified by balloting externally on the buccal fat pad on the cheek. The incision line is then anesthetized transorally with lidocaine and epinephrine-containing solution. The cheek is retracted laterally with Caldwell-Luc retractors, and a 1-cm incision is made and carried through the mucosa and superficial muscle using the Colorado needle. Scissors are then used to spread the deeper muscle fibers to expose the deeper fascia and fat pads. Metzenbaum scissors are used to gently spread the fat while the surgeon places pressure on the external cheek, manipulating the fat into the wound. Without excess traction, the walnut-sized portion of the fat pad that protrudes is grasped with Debakey forceps, gently teased into the field, clamped at its base with a curved hemostat, and excised. The stump is electrocoagulated with an extendable protected Colorado needle, with care to prevent inadvertent cauterization of the lips. The wound is closed with a single 4-0 poliglecaprone-25 suture.
A 5-cc Luer lock syringe is preloaded with 2 cc of normal saline and attached to another 5-cc Luer lock syringe via a female-female attachment. The excised fat is then placed in a 5-cc Luer lock syringe by removing the plunger. The plunger is then reinstalled, and the fat is injected back and forth approximately 30 times. The fat is centrifuged at 3500 rpm for 3 minutes. The purified fat is then transferred to a 1-cc Luer lock syringe attached to an 18-gauge needle.
Fat Injection—The authors use an 18-gauge needle to perform depot injections into the temporal fossae above the periosteum. This is a relatively safe area of the face to inject, but care must be taken to avoid injury to the superficial temporal artery. Between 1.5 and 3 cc of high-quality fat usually are administered to each temple.
Aftercare Instructions—The patient is instructed to have a soft diet for 24 to 48 hours and can return to work the next day. The patient also is given prophylactic antibiotics with Gram-negative coverage for 7 days (amoxicillin-clavulanate 875 mg/125 mg orally twice daily for 7 days).
Candidates for Buccal Fat Pad Reduction
Buccal fat pad reduction has become an increasingly popular technique for midface and lower face shaping to decrease the appearance of a round face. To achieve an aesthetically pleasing midface, surgeons should consider enhancing zygomatic eminences while emphasizing the border between the zygomatic prominence and cheek hollow.6 Selection criteria for buccal fat pad reduction are not well established. One study recommended avoiding the procedure in pregnant or lactating patients, patients with chronic illnesses, patients on blood-thinning agents, and patients younger than 18 years. In addition, this study suggested ensuring the malar fullness is in the anteromedial portion of the face, as posterolateral fullness may be due to masseter hypertrophy.6
Complications From Buccal Fat Pad Reduction
Complications associated with buccal fat pad reduction include inadvertent damage to surrounding structures, including the buccal branch of the facial nerve and parotid duct. Because the location of the facial nerve in relation to the parotid duct is highly variable, surgeons must be aware of its anatomy to avoid unintentional damage. Hwang et al7 reported that the parotid duct and buccal branches of the facial nerves passed through the buccal extension in 26.3% of cadavers. The transbuccal approach is preferred over the sub–superficial muscular aponeurotic system approach largely because it avoids these structures. In addition, blunt dissection may further decrease chances of injury. Although the long-term effects are unknown, there is a potential risk for facial hollowing.3 The use of preprocedure ultrasonography to quantify the buccal fat pad may avoid overresection and enhanced potential for facial hollowing.6
Avoidance of Temporal Hollowing
Because the buccal fat pad extends into the temporal space, buccal fat pad reduction may lead to further temporal hollowing, contributing to an aged appearance. The authors’ technique addresses both midface and upper face contouring in one minimally invasive procedure. Temporal hollowing commonly has been corrected with autologous fat grafting from the thigh or abdomen, which leads to an additional scar at the donor site. Our technique relies on autologous adjacent fat transfer from previously removed buccal fat. In addition, compared with the use of hyaluronic acid fillers for temple reflation, fat transfer largely is safe and biocompatible. Major complications of autologous fat transfer to the temples include nodularity or fat clumping, fat necrosis, sensory or motor nerve damage, and edema or ecchymosis.4 Also, with time there will be ongoing hollowing of the temples as part of the aging process with soft tissue and bone resorption. Therefore, further volume restoration procedures may be required in the future to address these dynamic changes.
Conclusion
The buccal fat pad has been extensively used to reconstruct oral defects, including oroantral and cranial base defects, owing to its high vascularity.6 However, there also is great potential to utilize buccal fat for autologous fat transfer to improve temporal wasting. Further studies are needed to determine optimal technique as well as longer-term safety and efficacy of this procedure.
The buccal fat pad (Bichat fat pad) is a tubular-shaped collection of adipose tissue that occupies a prominent position in the midface. The buccal fat pad has been described as having 3 lobes: an anterior lobe, which is anterior to the masseter muscle; an intermediate lobe between the masseter and buccinator muscles; and a posterior lobe between the temporal masticatory space.1 There are 4 extensions from the body of the buccal fat pad: the buccal, the sublevator, the melolabial, and the pterygoid. It is the buccal extension and main body that are removed intraorally to achieve midfacial and lower facial contouring, as these support the contours of the cheeks. The deep fat pad within the temporal fossa is a true extension of the buccal fat pad (Figure).2 It has a complex relationship to the facial structures, with known variability in the positions of the buccal branch of the facial nerve and the parotid duct.3 The parotid duct travels over, superior to, or through the buccal extension 42%, 32%, and 26% of the time, respectively. The duct travels along the surface of the masseter, then pierces the buccinator to drain into the vestibule of the mouth at the second superior molar tooth. The buccal branch of the facial nerve travels on the surface of the buccal fat pad 73% of the time, whereas 27% of the time it travels deeper through the buccal extension.4 A study that used ultrasonography to map the surface anatomy path of the parotid duct in 50 healthy patients showed that the duct was within 1.5 cm of the middle half of a line between the lower border of the tragus and the oral commissure in 93% of individuals.5 We describe a technique in which part of the buccal fat pad is removed and the fat is transferred to the temple to achieve aesthetically pleasing facial contouring. We used a vertical line from the lateral canthus as a surface anatomy landmark to determine when the duct emerges from the gland and is most susceptible to injury.

Operative Technique
Correct instrumentation is important to obtain appropriate anatomic exposure for this procedure. The surgical tray should include 4-0 poliglecaprone 25 suture, bite guards, a needle driver, a hemostat, surgical scissors, toothed forceps, a Beaver surgical handle with #15 blade, a protected diathermy needle, cotton tip applicators, and gauze.
Fat Harvest—With the patient supine, bite blocks are placed, and the buccal fat pad incision line is marked with a surgical marker. A 1-cm line is drawn approximately 4 cm posterior to the oral commissure by the buccal bite marks. The location is verified by balloting externally on the buccal fat pad on the cheek. The incision line is then anesthetized transorally with lidocaine and epinephrine-containing solution. The cheek is retracted laterally with Caldwell-Luc retractors, and a 1-cm incision is made and carried through the mucosa and superficial muscle using the Colorado needle. Scissors are then used to spread the deeper muscle fibers to expose the deeper fascia and fat pads. Metzenbaum scissors are used to gently spread the fat while the surgeon places pressure on the external cheek, manipulating the fat into the wound. Without excess traction, the walnut-sized portion of the fat pad that protrudes is grasped with Debakey forceps, gently teased into the field, clamped at its base with a curved hemostat, and excised. The stump is electrocoagulated with an extendable protected Colorado needle, with care to prevent inadvertent cauterization of the lips. The wound is closed with a single 4-0 poliglecaprone-25 suture.
A 5-cc Luer lock syringe is preloaded with 2 cc of normal saline and attached to another 5-cc Luer lock syringe via a female-female attachment. The excised fat is then placed in a 5-cc Luer lock syringe by removing the plunger. The plunger is then reinstalled, and the fat is injected back and forth approximately 30 times. The fat is centrifuged at 3500 rpm for 3 minutes. The purified fat is then transferred to a 1-cc Luer lock syringe attached to an 18-gauge needle.
Fat Injection—The authors use an 18-gauge needle to perform depot injections into the temporal fossae above the periosteum. This is a relatively safe area of the face to inject, but care must be taken to avoid injury to the superficial temporal artery. Between 1.5 and 3 cc of high-quality fat usually are administered to each temple.
Aftercare Instructions—The patient is instructed to have a soft diet for 24 to 48 hours and can return to work the next day. The patient also is given prophylactic antibiotics with Gram-negative coverage for 7 days (amoxicillin-clavulanate 875 mg/125 mg orally twice daily for 7 days).
Candidates for Buccal Fat Pad Reduction
Buccal fat pad reduction has become an increasingly popular technique for midface and lower face shaping to decrease the appearance of a round face. To achieve an aesthetically pleasing midface, surgeons should consider enhancing zygomatic eminences while emphasizing the border between the zygomatic prominence and cheek hollow.6 Selection criteria for buccal fat pad reduction are not well established. One study recommended avoiding the procedure in pregnant or lactating patients, patients with chronic illnesses, patients on blood-thinning agents, and patients younger than 18 years. In addition, this study suggested ensuring the malar fullness is in the anteromedial portion of the face, as posterolateral fullness may be due to masseter hypertrophy.6
Complications From Buccal Fat Pad Reduction
Complications associated with buccal fat pad reduction include inadvertent damage to surrounding structures, including the buccal branch of the facial nerve and parotid duct. Because the location of the facial nerve in relation to the parotid duct is highly variable, surgeons must be aware of its anatomy to avoid unintentional damage. Hwang et al7 reported that the parotid duct and buccal branches of the facial nerves passed through the buccal extension in 26.3% of cadavers. The transbuccal approach is preferred over the sub–superficial muscular aponeurotic system approach largely because it avoids these structures. In addition, blunt dissection may further decrease chances of injury. Although the long-term effects are unknown, there is a potential risk for facial hollowing.3 The use of preprocedure ultrasonography to quantify the buccal fat pad may avoid overresection and enhanced potential for facial hollowing.6
Avoidance of Temporal Hollowing
Because the buccal fat pad extends into the temporal space, buccal fat pad reduction may lead to further temporal hollowing, contributing to an aged appearance. The authors’ technique addresses both midface and upper face contouring in one minimally invasive procedure. Temporal hollowing commonly has been corrected with autologous fat grafting from the thigh or abdomen, which leads to an additional scar at the donor site. Our technique relies on autologous adjacent fat transfer from previously removed buccal fat. In addition, compared with the use of hyaluronic acid fillers for temple reflation, fat transfer largely is safe and biocompatible. Major complications of autologous fat transfer to the temples include nodularity or fat clumping, fat necrosis, sensory or motor nerve damage, and edema or ecchymosis.4 Also, with time there will be ongoing hollowing of the temples as part of the aging process with soft tissue and bone resorption. Therefore, further volume restoration procedures may be required in the future to address these dynamic changes.
Conclusion
The buccal fat pad has been extensively used to reconstruct oral defects, including oroantral and cranial base defects, owing to its high vascularity.6 However, there also is great potential to utilize buccal fat for autologous fat transfer to improve temporal wasting. Further studies are needed to determine optimal technique as well as longer-term safety and efficacy of this procedure.
- Zhang HM, Yan YP, Qi KM, et al. Anatomical structure of the buccal fat pad and its clinical adaptations. Plast Reconstr Surg. 2002;109:2509-2518.
- Yousuf S, Tubbs RS, Wartmann CT, et al. A review of the gross anatomy, functions, pathology, and clinical uses of the buccal fat pad. Surg Radiol Anat. 2010;32:427-436.
- Benjamin M, Reish RG. Buccal fat pad excision: proceed with caution. Plast Reconstr Surg Glob Open. 2018;6:E1970.
- Tzikas TL. Fat grafting volume restoration to the brow and temporal regions. Facial Plast Surg. 2018;34:164-172.
- Stringer MD, Mirjalili SA, Meredith SJ, et al. Redefining the surface anatomy of the parotid duct: an in vivo ultrasound study. Plast Reconstr Surg. 2012;130:1032-1037.
- Sezgin B, Tatar S, Boge M, et al. The excision of the buccal fat pad for cheek refinement: volumetric considerations. Aesthet Surg J. 2019;39:585-592.
- Hwang K, Cho HJ, Battuvshin D, et al. Interrelated buccal fat pad with facial buccal branches and parotid duct. J Craniofac Surg. 2005;16:658-660.
- Zhang HM, Yan YP, Qi KM, et al. Anatomical structure of the buccal fat pad and its clinical adaptations. Plast Reconstr Surg. 2002;109:2509-2518.
- Yousuf S, Tubbs RS, Wartmann CT, et al. A review of the gross anatomy, functions, pathology, and clinical uses of the buccal fat pad. Surg Radiol Anat. 2010;32:427-436.
- Benjamin M, Reish RG. Buccal fat pad excision: proceed with caution. Plast Reconstr Surg Glob Open. 2018;6:E1970.
- Tzikas TL. Fat grafting volume restoration to the brow and temporal regions. Facial Plast Surg. 2018;34:164-172.
- Stringer MD, Mirjalili SA, Meredith SJ, et al. Redefining the surface anatomy of the parotid duct: an in vivo ultrasound study. Plast Reconstr Surg. 2012;130:1032-1037.
- Sezgin B, Tatar S, Boge M, et al. The excision of the buccal fat pad for cheek refinement: volumetric considerations. Aesthet Surg J. 2019;39:585-592.
- Hwang K, Cho HJ, Battuvshin D, et al. Interrelated buccal fat pad with facial buccal branches and parotid duct. J Craniofac Surg. 2005;16:658-660.
Practice Points
- Buccal fat pad reduction is an increasingly popular procedure for facial shaping.
- Buccal fat pad reduction in addition to natural aging can result in volume depletion of the temporal fossae.
- Removed buccal fat can be transferred to the temples for increased volume.
Nicotine and Nicotine Replacement Therapy Use During Myocardial Perfusion Imaging
Chest pain is one of the most common concerns in patients presenting to the emergency department in the United States, accounting for approximately 7.6 million visits annually.1 Given the high mortality rate associated with acute coronary syndromes, prompt evaluation of chest pain is essential.2 Even in mild cases, recognition of newly onset or worsening coronary artery disease (CAD) is crucial to ensure that patients receive optimal medication therapy.
In symptomatic patients with risk factors for CAD, such as advanced age, hypertension, hyperlipidemia, obesity, and diabetes mellitus, myocardial perfusion imaging (MPI) is frequently used as a modality to assess the presence, location, and severity of ischemic or infarcted myocardium.2 MPI requires administration of a radiopharmaceutical before and after the patient undergoes a form of stress.2 This radiopharmaceutical is then detected in the myocardium with a nuclear camera, and images are obtained of the heart to assess myocardial blood flow.2
MPI can be performed using exercise-induced stress via a treadmill, or medication-induced stress (Table 1). In both strategies, healthy coronary arteries dilate to provide the myocardium with more blood flow to meet the increasing myocardial oxygen demand during this period of stress. While healthy vessels are able to dilate appropriately, coronary arteries with flow-limiting stenoses are unable to dilate to the same extent in response to stress.2 Because radioactive isotope uptake by the myocardium is directly related to arterial blood flow, MPI is able to demonstrate a mismatch in coronary blood flow between healthy and diseased coronary arteries indicated by differences in radioisotope uptake.2 The presence of such a mismatch, in conjunction with clinical history, potentially suggests the presence of CAD.
Prior to conducting MPI with a medication, certain substances should be avoided. For instance, methylxanthines, such as caffeine, aminophylline, and theophylline, antagonize adenosine receptors and can have major drug interactions with regadenoson, adenosine, and dipyridamole. Therefore, it is advised that these substances be stopped for at least 12 hours before testing.3 In some cases, other medications that can affect coronary blood flow, such as long-acting nitrates, β-blockers, and calcium channel blockers, are recommended to be avoided for 12 to 48 hours in order to obtain the most accurate depiction of underlying coronary disease.4
Because nicotine and nicotine replacement therapy (NRT) may have substantial effects on coronary circulation, a current area of controversy is whether these should be stopped prior to the use of a stress-inducing medication during MPI. To date, no formal drug interaction studies have been conducted between nicotine and regadenoson.5 Similarly, the ADVANCE MPI 2 Trial, which led to the US Food and Drug Administration approval of regadenoson, did not specify restrictions on the use of nicotine prior to stress testing in the protocol.6 However, as this trial was multicenter, investigators admit that individual study sites could have had their own restrictions on the use of nicotine prior to stress testing with regadenoson, but this information was not collected.6 The current review focuses on how the simultaneous use of nicotine or NRT during MPI with pharmacologic agents, such as regadenoson, may affect the accuracy of imaging results and the clinical impact of this interaction.
Nicotine Coronary Artery Effect
It is well documented that long-term cigarette smoking is a major risk factor for CAD.7 Compared with nonsmokers, cigarette smokers experience 2 times greater risk of morbidity and mortality from ischemic heart disease.7 There are several mechanisms by which nicotine induces damage to the myocardium (Figure). Nicotine has direct effects on both the sympathetic nervous system (SNS) and myocardial endothelium.8 Together, these factors result in reduced coronary blood flow, leading to less oxygen supply to meet an increased oxygen demand, resulting in myocardial ischemia.
Nicotine’s effect on coronary vasomotor tone occurs primarily through noradrenergic stimulation of α and β receptors associated with coronary vasoconstriction or vasodilation, respectively.9,10 These competing influences on coronary blood flow appear to manifest differently based on whether patients are at rest or in a stressed state. A study by Czerin and colleagues demonstrated that in healthy patients with relatively short smoking histories and in a healthy nonsmoker control group, coronary blood flow increased by 25% and 40%, respectively, with nicotine use at rest.9 However, when these patients were stressed with dipyramidole and while smoking during the examination, myocardial blood flow was reduced by 11% in the study group and 14% in the control group.9 This is likely because the patients studied had relatively healthy coronary arteries that were able to maximally dilate when stressed. In this scenario, nicotine’s dilatory effects are offset by nicotine’s α-receptor–mediated vasoconstriction effects.9 Of note, patients in the study group experienced a somewhat diminished increase in coronary blood flow at rest with nicotine use, suggesting that even a short smoking history may damage the myocardial endothelium, rendering it less responsive to nicotine’s vasodilatory effects.9
These principles similarly apply to patients with underlying moderate-to-severe cardiovascular disease (CVD). With nicotine use at rest, patients with significant CAD do not experience as dramatic of an increase in coronary blood flow, which typically decreases or remains the same despite increased myocardial work.10 This may be because patients with moderate-to-severe CAD often have flow-limiting stenoses and damaged endothelium that do not allow vessels to respond as efficiently to increased myocardial demand or to nicotine’s β-receptor–mediated vasodilatory effects.10,11 Moreover, when stressed, diseased coronary arteries are not able to further dilate and nicotine’s α-receptor–mediated vasoconstriction effects dominate.10,11
In a study by Quillen and colleagues of patients with moderate-to-severe CAD, the mean diameter of proximal coronary artery segments decreased by 5%, the distal coronary diameter decreased 8%, and the coronary vascular resistance increased by 21% while smoking at rest.12 The investigators did not analyze how parameters changed when these diseased coronary arteries were stressed using a medication during MPI. However, it can be predicted that coronary arteries would have constricted to a similar or greater degree than observed in Czerin and colleagues’ study, given that the underlying myocardium was diseased and more susceptible to nicotine’s vasoconstriction effects.9 Importantly, these studies have several limitations, most notably that they are older and have small sample sizes. Additionally, while statistically significant differences were found in the degree of changes in coronary circulation with nicotine use at rest and during stress, it is unclear whether this translates to a clinically significant and impactful finding.9-12
Nicotine Replacement Therapy and Stress Testing
Given the association between cigarette smoking and CAD, medical practitioners strongly encourage patients to quit smoking to reduce their risk of adverse cardiovascular outcomes. Various smoking cessation treatments are available for patients. Common, readily accessible forms of therapy include nicotine replacement products (Table 2).
Early studies of NRT in patients with underlying CVD found an increased risk of cardiovascular events, such as myocardial infarction, presumably due to the nicotine content of these products.13,14 However, the concentration of nicotine in NRT is substantially lower than that found in cigarettes and in some formulations, such as transdermal patches, nicotine is delivered over a prolonged period of time.15 For this reason, NRT is thought to be safe in patients with underlying CVD and stable ischemic heart disease. A recent systematic review and meta-analysis found that while NRT may be associated with tachycardia, it did not increase the risk of more serious cardiovascular adverse effects (AEs).16,17
Given the lower nicotine concentration in NRT products, the associated hemodynamic effect of nicotine also is thought to be less pronounced. In a study conducted by Tzivoni and colleagues in patients with CAD using transdermal nicotine patches, no differences in blood pressure, heart rate, ischemia, or arrhythmias were found from baseline to 2 weeks.18 These findings were further confirmed in a small study by Lucini and colleagues, which found that nicotine patches produced slight hemodynamic effects, but to a lesser extent than cigarette smoking.19 For the NRT gum formulation, while a small study found that 4 mg produced coronary vasoconstriction in patients with underlying CAD, a study by Nitenberg and Antony demonstrated that healthy and diseased coronary arteries did not significantly constrict while patients were using nicotine gum both before and after a cold pressor test, suggesting a lesser degree of coronary vasoconstriction than nicotine from cigarette smoking.20,21 Similar findings have been described with the nicotine intranasal spray in a study by Keeley and colleagues, which showed no additional AEs on myocardial demand or vasoconstriction when an intranasal nicotine spray was added to cigarette smoking.22 Importantly, a review of the transdermal and gum formulations found that these less pronounced hemodynamic effects were observed across different doses of NRT; however, further studies are needed to clarify the relationship between NRT dose and cardiovascular effects.23
Overall, NRT does not seem to activate the SNS to the same degree as nicotine obtained via cigarette smoking and likely does not increase the myocardial oxygen demand as much. Additionally, by containing a lower concentration of nicotine, NRT may not impair the myocardium’s ability to supply oxygen to coronary arteries to the same extent as nicotine from cigarette smoking. Therefore, the effects of NRT on MPI using a stress-inducing medication may not be as pronounced. However, due to study limitations, results should be interpreted cautiously.18-23
Conclusions
Because of the close relationship between cigarette smoking and CAD, many patients with underlying CVD are either current smokers or may be using NRT for smoking cessation. Therefore, the question of whether to refrain from nicotine use prior to MPI is clinically relevant. Currently, there is a lack of high-quality studies demonstrating the effects of nicotine and NRT on coronary perfusion. Because of this, the impact of nicotine and NRT use on the accuracy of MPI using stress-inducing medications remains uncertain. Nevertheless, given that nicotine and NRT may largely affect the accuracy of imaging results, several institutions have adopted protocols that prohibit patients from using these drugs on the day of nuclear stress testing.
There are currently no data specifying the number of hours to hold nicotine products prior to cardiac stress testing. It is generally recommended that other medications that affect coronary blood flow be held for 5 half-lives before conducting MPI.4 Following the same guidance for nicotine and NRT may present a reasonable approach to ensure accurate imaging results. Based on the discussed literature, patients should be instructed to refrain from cigarette smoking for at least 5 to 10 hours prior to MPI, given nicotine’s half-life of about 1 to 2 hours.24
The data for NRT are less clear. While use of NRT may not be an absolute contraindication to conducting MPI, it is important to consider that this may affect the accuracy of results. Given this uncertainty, it is likely ideal to hold NRT prior to MPI, based on the specific formulation of NRT and that product's half-life. Further robust studies are needed to analyze the impact of nicotine and NRT on the accuracy of nuclear stress testing using a medication.
1. Rui P, Kang K, Ashman JJ. National Hospital Ambulatory Medical Care Survey: 2016 emergency department summary tables. Published 2016. Accessed March 30, 2020. https://www.cdc.gov/nchs/data/nhamcs/web_tables/2016_ed_web_tables.pdf
2. Lange RA. Cardiovascular testing. In: DiPiro JT, Talbert RL, Yee GC, Matzke GR, Wells BG, Posey L. eds. Pharmacotherapy: A Pathophysiologic Approach, 10th ed. McGraw Hill; 2017.
3. Mace S. Observation Medicine: Principles and Protocols. Cambridge University Press; 2017.
4. Currie GM. Pharmacology, part 4: nuclear cardiology. J Nucl Med Technol. 2019;47(2):97-110. doi:10.2967/jnmt.118.219675
5. Regadenoson; Package insert. Astellas Pharma US Inc; 2008.
6. Iskandrian AE, Bateman TM, Belardinelli L, et al. Adenosine versus regadenoson comparative evaluation in myocardial perfusion imaging: results of the ADVANCE phase 3 multicenter international trial. J Nucl Cardiol. 2007;14(5):645-658. doi:10.1016/j.nuclcard.2007.06.114
7. Hajar R. Risk factors for coronary artery disease: historical perspectives. Heart Views. 2017;18(3):109-114. doi:10.4103/HEARTVIEWS.HEARTVIEWS_106_17
8. Benowitz NL, Burbank AD. Cardiovascular toxicity of nicotine: implications for electronic cigarette use. Trends Cardiovasc Med. 2016;26(6):515-523. doi:10.1016/j.tcm.2016.03.001
9. Czernin J, Sun K, Brunken R, Böttcher M, Phelps M, Schelbert H. Effect of acute and long-term smoking on myocardial blood flow and flow reserve. Circulation. 1995;91:2891-2897. doi:10.1161/01.CIR.91.12.2891
10. Winniford MD, Wheelan KR, Kremers MS, et al. Smoking-induced coronary vasoconstriction in patients with atherosclerotic coronary artery disease: evidence for adrenergically mediated alterations in coronary artery tone. Circulation. 1986;73(4):662-667. doi:10.1161/01.cir.73.4.662
11. Klein LW, Ambrose J, Pichard A, Holt J, Gorlin R, Teichholz LE. Acute coronary hemodynamic response to cigarette smoking in patients with coronary artery disease. J Am Coll Cardiol. 1984;3(4):879-886. doi:10.1016/s0735-1097(84)80344-7
12. Quillen JE, Rossen JD, Oskarsson HJ, Minor RL Jr, Lopez AG, Winniford MD. Acute effect of cigarette smoking on the coronary circulation: constriction of epicardial and resistance vessels. J Am Coll Cardiol. 1993;22(3):642-647. doi:10.1016/0735-1097(93)90170-6
13. Dacosta A, Guy JM, Tardy B, et al. Myocardial infarction and nicotine patch: a contributing or causative factor?. Eur Heart J. 1993;14(12):1709-1711. doi:10.1093/eurheartj/14.12.1709
14. Ottervanger JP, Festen JM, de Vries AG, Stricker BH. Acute myocardial infarction while using the nicotine patch. Chest. 1995;107(6):1765-1766. doi:10.1378/chest.107.6.1765
15. Dollerup J, Vestbo J, Murray-Thomas T, et al. Cardiovascular risks in smokers treated with nicotine replacement therapy: a historical cohort study. Clin Epidemiol. 2017;9:231-243. Published 2017 Apr 26. doi:10.2147/CLEP.S127775
16. Mills EJ, Wu P, Lockhart I, Wilson K, Ebbert JO. Adverse events associated with nicotine replacement therapy (NRT) for smoking cessation. A systematic review and meta-analysis of one hundred and twenty studies involving 177,390 individuals. Tob Induc Dis. 2010;8(1):8. Published 2010 Jul 13. doi:10.1186/1617-9625-8-8
17. Mills EJ, Thorlund K, Eapen S, Wu P, Prochaska JJ. Cardiovascular events associated with smoking cessation pharmacotherapies: a network meta-analysis. Circulation. 2014;129(1):28-41. doi:10.1161/CIRCULATIONAHA.113.003961
18. Tzivoni D, Keren A, Meyler S, Khoury Z, Lerer T, Brunel P. Cardiovascular safety of transdermal nicotine patches in patients with coronary artery disease who try to quit smoking. Cardiovasc Drugs Ther. 1998;12(3):239-244. doi:10.1023/a:1007757530765
19. Lucini D, Bertocchi F, Malliani A, Pagani M. Autonomic effects of nicotine patch administration in habitual cigarette smokers: a double-blind, placebo-controlled study using spectral analysis of RR interval and systolic arterial pressure variabilities. J Cardiovasc Pharmacol. 1998;31(5):714-720. doi:10.1097/00005344-199805000-00010
20. Kaijser L, Berglund B. Effect of nicotine on coronary blood-flow in man. Clin Physiol. 1985;5(6):541-552. doi:10.1111/j.1475-097x.1985.tb00767.x
21. Nitenberg A, Antony I. Effects of nicotine gum on coronary vasomotor responses during sympathetic stimulation in patients with coronary artery stenosis. J Cardiovasc Pharmacol. 1999;34(5):694-699. doi:10.1097/00005344-199911000-00011
22. Keeley EC, Pirwitz MJ, Landau C, et al. Intranasal nicotine spray does not augment the adverse effects of cigarette smoking on myocardial oxygen demand or coronary arterial dimensions. Am J Med. 1996;101(4):357-363. doi:10.1016/s0002-9343(96)00237-9
23. Benowitz NL, Gourlay SG. Cardiovascular toxicity of nicotine: implications for nicotine replacement therapy. J Am Coll Cardiol. 1997;29(7):1422-1431. doi:10.1016/s0735-1097(97)00079-x
24. Flowers L. Nicotine replacement therapy. Amer J Psych. 2017;11(6):4-7.
25. Adenosine; Package insert. Astellas Pharma US Inc; 1989.
26. Dipyridamole; Package insert. Boehringer Ingelheim Pharmaceuticals Inc; 2019.
27. Dobutamine; Package insert. Baxter Healthcare Corporation; 2012.
Chest pain is one of the most common concerns in patients presenting to the emergency department in the United States, accounting for approximately 7.6 million visits annually.1 Given the high mortality rate associated with acute coronary syndromes, prompt evaluation of chest pain is essential.2 Even in mild cases, recognition of newly onset or worsening coronary artery disease (CAD) is crucial to ensure that patients receive optimal medication therapy.
In symptomatic patients with risk factors for CAD, such as advanced age, hypertension, hyperlipidemia, obesity, and diabetes mellitus, myocardial perfusion imaging (MPI) is frequently used as a modality to assess the presence, location, and severity of ischemic or infarcted myocardium.2 MPI requires administration of a radiopharmaceutical before and after the patient undergoes a form of stress.2 This radiopharmaceutical is then detected in the myocardium with a nuclear camera, and images are obtained of the heart to assess myocardial blood flow.2
MPI can be performed using exercise-induced stress via a treadmill, or medication-induced stress (Table 1). In both strategies, healthy coronary arteries dilate to provide the myocardium with more blood flow to meet the increasing myocardial oxygen demand during this period of stress. While healthy vessels are able to dilate appropriately, coronary arteries with flow-limiting stenoses are unable to dilate to the same extent in response to stress.2 Because radioactive isotope uptake by the myocardium is directly related to arterial blood flow, MPI is able to demonstrate a mismatch in coronary blood flow between healthy and diseased coronary arteries indicated by differences in radioisotope uptake.2 The presence of such a mismatch, in conjunction with clinical history, potentially suggests the presence of CAD.
Prior to conducting MPI with a medication, certain substances should be avoided. For instance, methylxanthines, such as caffeine, aminophylline, and theophylline, antagonize adenosine receptors and can have major drug interactions with regadenoson, adenosine, and dipyridamole. Therefore, it is advised that these substances be stopped for at least 12 hours before testing.3 In some cases, other medications that can affect coronary blood flow, such as long-acting nitrates, β-blockers, and calcium channel blockers, are recommended to be avoided for 12 to 48 hours in order to obtain the most accurate depiction of underlying coronary disease.4
Because nicotine and nicotine replacement therapy (NRT) may have substantial effects on coronary circulation, a current area of controversy is whether these should be stopped prior to the use of a stress-inducing medication during MPI. To date, no formal drug interaction studies have been conducted between nicotine and regadenoson.5 Similarly, the ADVANCE MPI 2 Trial, which led to the US Food and Drug Administration approval of regadenoson, did not specify restrictions on the use of nicotine prior to stress testing in the protocol.6 However, as this trial was multicenter, investigators admit that individual study sites could have had their own restrictions on the use of nicotine prior to stress testing with regadenoson, but this information was not collected.6 The current review focuses on how the simultaneous use of nicotine or NRT during MPI with pharmacologic agents, such as regadenoson, may affect the accuracy of imaging results and the clinical impact of this interaction.
Nicotine Coronary Artery Effect
It is well documented that long-term cigarette smoking is a major risk factor for CAD.7 Compared with nonsmokers, cigarette smokers experience 2 times greater risk of morbidity and mortality from ischemic heart disease.7 There are several mechanisms by which nicotine induces damage to the myocardium (Figure). Nicotine has direct effects on both the sympathetic nervous system (SNS) and myocardial endothelium.8 Together, these factors result in reduced coronary blood flow, leading to less oxygen supply to meet an increased oxygen demand, resulting in myocardial ischemia.
Nicotine’s effect on coronary vasomotor tone occurs primarily through noradrenergic stimulation of α and β receptors associated with coronary vasoconstriction or vasodilation, respectively.9,10 These competing influences on coronary blood flow appear to manifest differently based on whether patients are at rest or in a stressed state. A study by Czerin and colleagues demonstrated that in healthy patients with relatively short smoking histories and in a healthy nonsmoker control group, coronary blood flow increased by 25% and 40%, respectively, with nicotine use at rest.9 However, when these patients were stressed with dipyramidole and while smoking during the examination, myocardial blood flow was reduced by 11% in the study group and 14% in the control group.9 This is likely because the patients studied had relatively healthy coronary arteries that were able to maximally dilate when stressed. In this scenario, nicotine’s dilatory effects are offset by nicotine’s α-receptor–mediated vasoconstriction effects.9 Of note, patients in the study group experienced a somewhat diminished increase in coronary blood flow at rest with nicotine use, suggesting that even a short smoking history may damage the myocardial endothelium, rendering it less responsive to nicotine’s vasodilatory effects.9
These principles similarly apply to patients with underlying moderate-to-severe cardiovascular disease (CVD). With nicotine use at rest, patients with significant CAD do not experience as dramatic of an increase in coronary blood flow, which typically decreases or remains the same despite increased myocardial work.10 This may be because patients with moderate-to-severe CAD often have flow-limiting stenoses and damaged endothelium that do not allow vessels to respond as efficiently to increased myocardial demand or to nicotine’s β-receptor–mediated vasodilatory effects.10,11 Moreover, when stressed, diseased coronary arteries are not able to further dilate and nicotine’s α-receptor–mediated vasoconstriction effects dominate.10,11
In a study by Quillen and colleagues of patients with moderate-to-severe CAD, the mean diameter of proximal coronary artery segments decreased by 5%, the distal coronary diameter decreased 8%, and the coronary vascular resistance increased by 21% while smoking at rest.12 The investigators did not analyze how parameters changed when these diseased coronary arteries were stressed using a medication during MPI. However, it can be predicted that coronary arteries would have constricted to a similar or greater degree than observed in Czerin and colleagues’ study, given that the underlying myocardium was diseased and more susceptible to nicotine’s vasoconstriction effects.9 Importantly, these studies have several limitations, most notably that they are older and have small sample sizes. Additionally, while statistically significant differences were found in the degree of changes in coronary circulation with nicotine use at rest and during stress, it is unclear whether this translates to a clinically significant and impactful finding.9-12
Nicotine Replacement Therapy and Stress Testing
Given the association between cigarette smoking and CAD, medical practitioners strongly encourage patients to quit smoking to reduce their risk of adverse cardiovascular outcomes. Various smoking cessation treatments are available for patients. Common, readily accessible forms of therapy include nicotine replacement products (Table 2).
Early studies of NRT in patients with underlying CVD found an increased risk of cardiovascular events, such as myocardial infarction, presumably due to the nicotine content of these products.13,14 However, the concentration of nicotine in NRT is substantially lower than that found in cigarettes and in some formulations, such as transdermal patches, nicotine is delivered over a prolonged period of time.15 For this reason, NRT is thought to be safe in patients with underlying CVD and stable ischemic heart disease. A recent systematic review and meta-analysis found that while NRT may be associated with tachycardia, it did not increase the risk of more serious cardiovascular adverse effects (AEs).16,17
Given the lower nicotine concentration in NRT products, the associated hemodynamic effect of nicotine also is thought to be less pronounced. In a study conducted by Tzivoni and colleagues in patients with CAD using transdermal nicotine patches, no differences in blood pressure, heart rate, ischemia, or arrhythmias were found from baseline to 2 weeks.18 These findings were further confirmed in a small study by Lucini and colleagues, which found that nicotine patches produced slight hemodynamic effects, but to a lesser extent than cigarette smoking.19 For the NRT gum formulation, while a small study found that 4 mg produced coronary vasoconstriction in patients with underlying CAD, a study by Nitenberg and Antony demonstrated that healthy and diseased coronary arteries did not significantly constrict while patients were using nicotine gum both before and after a cold pressor test, suggesting a lesser degree of coronary vasoconstriction than nicotine from cigarette smoking.20,21 Similar findings have been described with the nicotine intranasal spray in a study by Keeley and colleagues, which showed no additional AEs on myocardial demand or vasoconstriction when an intranasal nicotine spray was added to cigarette smoking.22 Importantly, a review of the transdermal and gum formulations found that these less pronounced hemodynamic effects were observed across different doses of NRT; however, further studies are needed to clarify the relationship between NRT dose and cardiovascular effects.23
Overall, NRT does not seem to activate the SNS to the same degree as nicotine obtained via cigarette smoking and likely does not increase the myocardial oxygen demand as much. Additionally, by containing a lower concentration of nicotine, NRT may not impair the myocardium’s ability to supply oxygen to coronary arteries to the same extent as nicotine from cigarette smoking. Therefore, the effects of NRT on MPI using a stress-inducing medication may not be as pronounced. However, due to study limitations, results should be interpreted cautiously.18-23
Conclusions
Because of the close relationship between cigarette smoking and CAD, many patients with underlying CVD are either current smokers or may be using NRT for smoking cessation. Therefore, the question of whether to refrain from nicotine use prior to MPI is clinically relevant. Currently, there is a lack of high-quality studies demonstrating the effects of nicotine and NRT on coronary perfusion. Because of this, the impact of nicotine and NRT use on the accuracy of MPI using stress-inducing medications remains uncertain. Nevertheless, given that nicotine and NRT may largely affect the accuracy of imaging results, several institutions have adopted protocols that prohibit patients from using these drugs on the day of nuclear stress testing.
There are currently no data specifying the number of hours to hold nicotine products prior to cardiac stress testing. It is generally recommended that other medications that affect coronary blood flow be held for 5 half-lives before conducting MPI.4 Following the same guidance for nicotine and NRT may present a reasonable approach to ensure accurate imaging results. Based on the discussed literature, patients should be instructed to refrain from cigarette smoking for at least 5 to 10 hours prior to MPI, given nicotine’s half-life of about 1 to 2 hours.24
The data for NRT are less clear. While use of NRT may not be an absolute contraindication to conducting MPI, it is important to consider that this may affect the accuracy of results. Given this uncertainty, it is likely ideal to hold NRT prior to MPI, based on the specific formulation of NRT and that product's half-life. Further robust studies are needed to analyze the impact of nicotine and NRT on the accuracy of nuclear stress testing using a medication.
Chest pain is one of the most common concerns in patients presenting to the emergency department in the United States, accounting for approximately 7.6 million visits annually.1 Given the high mortality rate associated with acute coronary syndromes, prompt evaluation of chest pain is essential.2 Even in mild cases, recognition of newly onset or worsening coronary artery disease (CAD) is crucial to ensure that patients receive optimal medication therapy.
In symptomatic patients with risk factors for CAD, such as advanced age, hypertension, hyperlipidemia, obesity, and diabetes mellitus, myocardial perfusion imaging (MPI) is frequently used as a modality to assess the presence, location, and severity of ischemic or infarcted myocardium.2 MPI requires administration of a radiopharmaceutical before and after the patient undergoes a form of stress.2 This radiopharmaceutical is then detected in the myocardium with a nuclear camera, and images are obtained of the heart to assess myocardial blood flow.2
MPI can be performed using exercise-induced stress via a treadmill, or medication-induced stress (Table 1). In both strategies, healthy coronary arteries dilate to provide the myocardium with more blood flow to meet the increasing myocardial oxygen demand during this period of stress. While healthy vessels are able to dilate appropriately, coronary arteries with flow-limiting stenoses are unable to dilate to the same extent in response to stress.2 Because radioactive isotope uptake by the myocardium is directly related to arterial blood flow, MPI is able to demonstrate a mismatch in coronary blood flow between healthy and diseased coronary arteries indicated by differences in radioisotope uptake.2 The presence of such a mismatch, in conjunction with clinical history, potentially suggests the presence of CAD.
Prior to conducting MPI with a medication, certain substances should be avoided. For instance, methylxanthines, such as caffeine, aminophylline, and theophylline, antagonize adenosine receptors and can have major drug interactions with regadenoson, adenosine, and dipyridamole. Therefore, it is advised that these substances be stopped for at least 12 hours before testing.3 In some cases, other medications that can affect coronary blood flow, such as long-acting nitrates, β-blockers, and calcium channel blockers, are recommended to be avoided for 12 to 48 hours in order to obtain the most accurate depiction of underlying coronary disease.4
Because nicotine and nicotine replacement therapy (NRT) may have substantial effects on coronary circulation, a current area of controversy is whether these should be stopped prior to the use of a stress-inducing medication during MPI. To date, no formal drug interaction studies have been conducted between nicotine and regadenoson.5 Similarly, the ADVANCE MPI 2 Trial, which led to the US Food and Drug Administration approval of regadenoson, did not specify restrictions on the use of nicotine prior to stress testing in the protocol.6 However, as this trial was multicenter, investigators admit that individual study sites could have had their own restrictions on the use of nicotine prior to stress testing with regadenoson, but this information was not collected.6 The current review focuses on how the simultaneous use of nicotine or NRT during MPI with pharmacologic agents, such as regadenoson, may affect the accuracy of imaging results and the clinical impact of this interaction.
Nicotine Coronary Artery Effect
It is well documented that long-term cigarette smoking is a major risk factor for CAD.7 Compared with nonsmokers, cigarette smokers experience 2 times greater risk of morbidity and mortality from ischemic heart disease.7 There are several mechanisms by which nicotine induces damage to the myocardium (Figure). Nicotine has direct effects on both the sympathetic nervous system (SNS) and myocardial endothelium.8 Together, these factors result in reduced coronary blood flow, leading to less oxygen supply to meet an increased oxygen demand, resulting in myocardial ischemia.
Nicotine’s effect on coronary vasomotor tone occurs primarily through noradrenergic stimulation of α and β receptors associated with coronary vasoconstriction or vasodilation, respectively.9,10 These competing influences on coronary blood flow appear to manifest differently based on whether patients are at rest or in a stressed state. A study by Czerin and colleagues demonstrated that in healthy patients with relatively short smoking histories and in a healthy nonsmoker control group, coronary blood flow increased by 25% and 40%, respectively, with nicotine use at rest.9 However, when these patients were stressed with dipyramidole and while smoking during the examination, myocardial blood flow was reduced by 11% in the study group and 14% in the control group.9 This is likely because the patients studied had relatively healthy coronary arteries that were able to maximally dilate when stressed. In this scenario, nicotine’s dilatory effects are offset by nicotine’s α-receptor–mediated vasoconstriction effects.9 Of note, patients in the study group experienced a somewhat diminished increase in coronary blood flow at rest with nicotine use, suggesting that even a short smoking history may damage the myocardial endothelium, rendering it less responsive to nicotine’s vasodilatory effects.9
These principles similarly apply to patients with underlying moderate-to-severe cardiovascular disease (CVD). With nicotine use at rest, patients with significant CAD do not experience as dramatic of an increase in coronary blood flow, which typically decreases or remains the same despite increased myocardial work.10 This may be because patients with moderate-to-severe CAD often have flow-limiting stenoses and damaged endothelium that do not allow vessels to respond as efficiently to increased myocardial demand or to nicotine’s β-receptor–mediated vasodilatory effects.10,11 Moreover, when stressed, diseased coronary arteries are not able to further dilate and nicotine’s α-receptor–mediated vasoconstriction effects dominate.10,11
In a study by Quillen and colleagues of patients with moderate-to-severe CAD, the mean diameter of proximal coronary artery segments decreased by 5%, the distal coronary diameter decreased 8%, and the coronary vascular resistance increased by 21% while smoking at rest.12 The investigators did not analyze how parameters changed when these diseased coronary arteries were stressed using a medication during MPI. However, it can be predicted that coronary arteries would have constricted to a similar or greater degree than observed in Czerin and colleagues’ study, given that the underlying myocardium was diseased and more susceptible to nicotine’s vasoconstriction effects.9 Importantly, these studies have several limitations, most notably that they are older and have small sample sizes. Additionally, while statistically significant differences were found in the degree of changes in coronary circulation with nicotine use at rest and during stress, it is unclear whether this translates to a clinically significant and impactful finding.9-12
Nicotine Replacement Therapy and Stress Testing
Given the association between cigarette smoking and CAD, medical practitioners strongly encourage patients to quit smoking to reduce their risk of adverse cardiovascular outcomes. Various smoking cessation treatments are available for patients. Common, readily accessible forms of therapy include nicotine replacement products (Table 2).
Early studies of NRT in patients with underlying CVD found an increased risk of cardiovascular events, such as myocardial infarction, presumably due to the nicotine content of these products.13,14 However, the concentration of nicotine in NRT is substantially lower than that found in cigarettes and in some formulations, such as transdermal patches, nicotine is delivered over a prolonged period of time.15 For this reason, NRT is thought to be safe in patients with underlying CVD and stable ischemic heart disease. A recent systematic review and meta-analysis found that while NRT may be associated with tachycardia, it did not increase the risk of more serious cardiovascular adverse effects (AEs).16,17
Given the lower nicotine concentration in NRT products, the associated hemodynamic effect of nicotine also is thought to be less pronounced. In a study conducted by Tzivoni and colleagues in patients with CAD using transdermal nicotine patches, no differences in blood pressure, heart rate, ischemia, or arrhythmias were found from baseline to 2 weeks.18 These findings were further confirmed in a small study by Lucini and colleagues, which found that nicotine patches produced slight hemodynamic effects, but to a lesser extent than cigarette smoking.19 For the NRT gum formulation, while a small study found that 4 mg produced coronary vasoconstriction in patients with underlying CAD, a study by Nitenberg and Antony demonstrated that healthy and diseased coronary arteries did not significantly constrict while patients were using nicotine gum both before and after a cold pressor test, suggesting a lesser degree of coronary vasoconstriction than nicotine from cigarette smoking.20,21 Similar findings have been described with the nicotine intranasal spray in a study by Keeley and colleagues, which showed no additional AEs on myocardial demand or vasoconstriction when an intranasal nicotine spray was added to cigarette smoking.22 Importantly, a review of the transdermal and gum formulations found that these less pronounced hemodynamic effects were observed across different doses of NRT; however, further studies are needed to clarify the relationship between NRT dose and cardiovascular effects.23
Overall, NRT does not seem to activate the SNS to the same degree as nicotine obtained via cigarette smoking and likely does not increase the myocardial oxygen demand as much. Additionally, by containing a lower concentration of nicotine, NRT may not impair the myocardium’s ability to supply oxygen to coronary arteries to the same extent as nicotine from cigarette smoking. Therefore, the effects of NRT on MPI using a stress-inducing medication may not be as pronounced. However, due to study limitations, results should be interpreted cautiously.18-23
Conclusions
Because of the close relationship between cigarette smoking and CAD, many patients with underlying CVD are either current smokers or may be using NRT for smoking cessation. Therefore, the question of whether to refrain from nicotine use prior to MPI is clinically relevant. Currently, there is a lack of high-quality studies demonstrating the effects of nicotine and NRT on coronary perfusion. Because of this, the impact of nicotine and NRT use on the accuracy of MPI using stress-inducing medications remains uncertain. Nevertheless, given that nicotine and NRT may largely affect the accuracy of imaging results, several institutions have adopted protocols that prohibit patients from using these drugs on the day of nuclear stress testing.
There are currently no data specifying the number of hours to hold nicotine products prior to cardiac stress testing. It is generally recommended that other medications that affect coronary blood flow be held for 5 half-lives before conducting MPI.4 Following the same guidance for nicotine and NRT may present a reasonable approach to ensure accurate imaging results. Based on the discussed literature, patients should be instructed to refrain from cigarette smoking for at least 5 to 10 hours prior to MPI, given nicotine’s half-life of about 1 to 2 hours.24
The data for NRT are less clear. While use of NRT may not be an absolute contraindication to conducting MPI, it is important to consider that this may affect the accuracy of results. Given this uncertainty, it is likely ideal to hold NRT prior to MPI, based on the specific formulation of NRT and that product's half-life. Further robust studies are needed to analyze the impact of nicotine and NRT on the accuracy of nuclear stress testing using a medication.
1. Rui P, Kang K, Ashman JJ. National Hospital Ambulatory Medical Care Survey: 2016 emergency department summary tables. Published 2016. Accessed March 30, 2020. https://www.cdc.gov/nchs/data/nhamcs/web_tables/2016_ed_web_tables.pdf
2. Lange RA. Cardiovascular testing. In: DiPiro JT, Talbert RL, Yee GC, Matzke GR, Wells BG, Posey L. eds. Pharmacotherapy: A Pathophysiologic Approach, 10th ed. McGraw Hill; 2017.
3. Mace S. Observation Medicine: Principles and Protocols. Cambridge University Press; 2017.
4. Currie GM. Pharmacology, part 4: nuclear cardiology. J Nucl Med Technol. 2019;47(2):97-110. doi:10.2967/jnmt.118.219675
5. Regadenoson; Package insert. Astellas Pharma US Inc; 2008.
6. Iskandrian AE, Bateman TM, Belardinelli L, et al. Adenosine versus regadenoson comparative evaluation in myocardial perfusion imaging: results of the ADVANCE phase 3 multicenter international trial. J Nucl Cardiol. 2007;14(5):645-658. doi:10.1016/j.nuclcard.2007.06.114
7. Hajar R. Risk factors for coronary artery disease: historical perspectives. Heart Views. 2017;18(3):109-114. doi:10.4103/HEARTVIEWS.HEARTVIEWS_106_17
8. Benowitz NL, Burbank AD. Cardiovascular toxicity of nicotine: implications for electronic cigarette use. Trends Cardiovasc Med. 2016;26(6):515-523. doi:10.1016/j.tcm.2016.03.001
9. Czernin J, Sun K, Brunken R, Böttcher M, Phelps M, Schelbert H. Effect of acute and long-term smoking on myocardial blood flow and flow reserve. Circulation. 1995;91:2891-2897. doi:10.1161/01.CIR.91.12.2891
10. Winniford MD, Wheelan KR, Kremers MS, et al. Smoking-induced coronary vasoconstriction in patients with atherosclerotic coronary artery disease: evidence for adrenergically mediated alterations in coronary artery tone. Circulation. 1986;73(4):662-667. doi:10.1161/01.cir.73.4.662
11. Klein LW, Ambrose J, Pichard A, Holt J, Gorlin R, Teichholz LE. Acute coronary hemodynamic response to cigarette smoking in patients with coronary artery disease. J Am Coll Cardiol. 1984;3(4):879-886. doi:10.1016/s0735-1097(84)80344-7
12. Quillen JE, Rossen JD, Oskarsson HJ, Minor RL Jr, Lopez AG, Winniford MD. Acute effect of cigarette smoking on the coronary circulation: constriction of epicardial and resistance vessels. J Am Coll Cardiol. 1993;22(3):642-647. doi:10.1016/0735-1097(93)90170-6
13. Dacosta A, Guy JM, Tardy B, et al. Myocardial infarction and nicotine patch: a contributing or causative factor?. Eur Heart J. 1993;14(12):1709-1711. doi:10.1093/eurheartj/14.12.1709
14. Ottervanger JP, Festen JM, de Vries AG, Stricker BH. Acute myocardial infarction while using the nicotine patch. Chest. 1995;107(6):1765-1766. doi:10.1378/chest.107.6.1765
15. Dollerup J, Vestbo J, Murray-Thomas T, et al. Cardiovascular risks in smokers treated with nicotine replacement therapy: a historical cohort study. Clin Epidemiol. 2017;9:231-243. Published 2017 Apr 26. doi:10.2147/CLEP.S127775
16. Mills EJ, Wu P, Lockhart I, Wilson K, Ebbert JO. Adverse events associated with nicotine replacement therapy (NRT) for smoking cessation. A systematic review and meta-analysis of one hundred and twenty studies involving 177,390 individuals. Tob Induc Dis. 2010;8(1):8. Published 2010 Jul 13. doi:10.1186/1617-9625-8-8
17. Mills EJ, Thorlund K, Eapen S, Wu P, Prochaska JJ. Cardiovascular events associated with smoking cessation pharmacotherapies: a network meta-analysis. Circulation. 2014;129(1):28-41. doi:10.1161/CIRCULATIONAHA.113.003961
18. Tzivoni D, Keren A, Meyler S, Khoury Z, Lerer T, Brunel P. Cardiovascular safety of transdermal nicotine patches in patients with coronary artery disease who try to quit smoking. Cardiovasc Drugs Ther. 1998;12(3):239-244. doi:10.1023/a:1007757530765
19. Lucini D, Bertocchi F, Malliani A, Pagani M. Autonomic effects of nicotine patch administration in habitual cigarette smokers: a double-blind, placebo-controlled study using spectral analysis of RR interval and systolic arterial pressure variabilities. J Cardiovasc Pharmacol. 1998;31(5):714-720. doi:10.1097/00005344-199805000-00010
20. Kaijser L, Berglund B. Effect of nicotine on coronary blood-flow in man. Clin Physiol. 1985;5(6):541-552. doi:10.1111/j.1475-097x.1985.tb00767.x
21. Nitenberg A, Antony I. Effects of nicotine gum on coronary vasomotor responses during sympathetic stimulation in patients with coronary artery stenosis. J Cardiovasc Pharmacol. 1999;34(5):694-699. doi:10.1097/00005344-199911000-00011
22. Keeley EC, Pirwitz MJ, Landau C, et al. Intranasal nicotine spray does not augment the adverse effects of cigarette smoking on myocardial oxygen demand or coronary arterial dimensions. Am J Med. 1996;101(4):357-363. doi:10.1016/s0002-9343(96)00237-9
23. Benowitz NL, Gourlay SG. Cardiovascular toxicity of nicotine: implications for nicotine replacement therapy. J Am Coll Cardiol. 1997;29(7):1422-1431. doi:10.1016/s0735-1097(97)00079-x
24. Flowers L. Nicotine replacement therapy. Amer J Psych. 2017;11(6):4-7.
25. Adenosine; Package insert. Astellas Pharma US Inc; 1989.
26. Dipyridamole; Package insert. Boehringer Ingelheim Pharmaceuticals Inc; 2019.
27. Dobutamine; Package insert. Baxter Healthcare Corporation; 2012.
1. Rui P, Kang K, Ashman JJ. National Hospital Ambulatory Medical Care Survey: 2016 emergency department summary tables. Published 2016. Accessed March 30, 2020. https://www.cdc.gov/nchs/data/nhamcs/web_tables/2016_ed_web_tables.pdf
2. Lange RA. Cardiovascular testing. In: DiPiro JT, Talbert RL, Yee GC, Matzke GR, Wells BG, Posey L. eds. Pharmacotherapy: A Pathophysiologic Approach, 10th ed. McGraw Hill; 2017.
3. Mace S. Observation Medicine: Principles and Protocols. Cambridge University Press; 2017.
4. Currie GM. Pharmacology, part 4: nuclear cardiology. J Nucl Med Technol. 2019;47(2):97-110. doi:10.2967/jnmt.118.219675
5. Regadenoson; Package insert. Astellas Pharma US Inc; 2008.
6. Iskandrian AE, Bateman TM, Belardinelli L, et al. Adenosine versus regadenoson comparative evaluation in myocardial perfusion imaging: results of the ADVANCE phase 3 multicenter international trial. J Nucl Cardiol. 2007;14(5):645-658. doi:10.1016/j.nuclcard.2007.06.114
7. Hajar R. Risk factors for coronary artery disease: historical perspectives. Heart Views. 2017;18(3):109-114. doi:10.4103/HEARTVIEWS.HEARTVIEWS_106_17
8. Benowitz NL, Burbank AD. Cardiovascular toxicity of nicotine: implications for electronic cigarette use. Trends Cardiovasc Med. 2016;26(6):515-523. doi:10.1016/j.tcm.2016.03.001
9. Czernin J, Sun K, Brunken R, Böttcher M, Phelps M, Schelbert H. Effect of acute and long-term smoking on myocardial blood flow and flow reserve. Circulation. 1995;91:2891-2897. doi:10.1161/01.CIR.91.12.2891
10. Winniford MD, Wheelan KR, Kremers MS, et al. Smoking-induced coronary vasoconstriction in patients with atherosclerotic coronary artery disease: evidence for adrenergically mediated alterations in coronary artery tone. Circulation. 1986;73(4):662-667. doi:10.1161/01.cir.73.4.662
11. Klein LW, Ambrose J, Pichard A, Holt J, Gorlin R, Teichholz LE. Acute coronary hemodynamic response to cigarette smoking in patients with coronary artery disease. J Am Coll Cardiol. 1984;3(4):879-886. doi:10.1016/s0735-1097(84)80344-7
12. Quillen JE, Rossen JD, Oskarsson HJ, Minor RL Jr, Lopez AG, Winniford MD. Acute effect of cigarette smoking on the coronary circulation: constriction of epicardial and resistance vessels. J Am Coll Cardiol. 1993;22(3):642-647. doi:10.1016/0735-1097(93)90170-6
13. Dacosta A, Guy JM, Tardy B, et al. Myocardial infarction and nicotine patch: a contributing or causative factor?. Eur Heart J. 1993;14(12):1709-1711. doi:10.1093/eurheartj/14.12.1709
14. Ottervanger JP, Festen JM, de Vries AG, Stricker BH. Acute myocardial infarction while using the nicotine patch. Chest. 1995;107(6):1765-1766. doi:10.1378/chest.107.6.1765
15. Dollerup J, Vestbo J, Murray-Thomas T, et al. Cardiovascular risks in smokers treated with nicotine replacement therapy: a historical cohort study. Clin Epidemiol. 2017;9:231-243. Published 2017 Apr 26. doi:10.2147/CLEP.S127775
16. Mills EJ, Wu P, Lockhart I, Wilson K, Ebbert JO. Adverse events associated with nicotine replacement therapy (NRT) for smoking cessation. A systematic review and meta-analysis of one hundred and twenty studies involving 177,390 individuals. Tob Induc Dis. 2010;8(1):8. Published 2010 Jul 13. doi:10.1186/1617-9625-8-8
17. Mills EJ, Thorlund K, Eapen S, Wu P, Prochaska JJ. Cardiovascular events associated with smoking cessation pharmacotherapies: a network meta-analysis. Circulation. 2014;129(1):28-41. doi:10.1161/CIRCULATIONAHA.113.003961
18. Tzivoni D, Keren A, Meyler S, Khoury Z, Lerer T, Brunel P. Cardiovascular safety of transdermal nicotine patches in patients with coronary artery disease who try to quit smoking. Cardiovasc Drugs Ther. 1998;12(3):239-244. doi:10.1023/a:1007757530765
19. Lucini D, Bertocchi F, Malliani A, Pagani M. Autonomic effects of nicotine patch administration in habitual cigarette smokers: a double-blind, placebo-controlled study using spectral analysis of RR interval and systolic arterial pressure variabilities. J Cardiovasc Pharmacol. 1998;31(5):714-720. doi:10.1097/00005344-199805000-00010
20. Kaijser L, Berglund B. Effect of nicotine on coronary blood-flow in man. Clin Physiol. 1985;5(6):541-552. doi:10.1111/j.1475-097x.1985.tb00767.x
21. Nitenberg A, Antony I. Effects of nicotine gum on coronary vasomotor responses during sympathetic stimulation in patients with coronary artery stenosis. J Cardiovasc Pharmacol. 1999;34(5):694-699. doi:10.1097/00005344-199911000-00011
22. Keeley EC, Pirwitz MJ, Landau C, et al. Intranasal nicotine spray does not augment the adverse effects of cigarette smoking on myocardial oxygen demand or coronary arterial dimensions. Am J Med. 1996;101(4):357-363. doi:10.1016/s0002-9343(96)00237-9
23. Benowitz NL, Gourlay SG. Cardiovascular toxicity of nicotine: implications for nicotine replacement therapy. J Am Coll Cardiol. 1997;29(7):1422-1431. doi:10.1016/s0735-1097(97)00079-x
24. Flowers L. Nicotine replacement therapy. Amer J Psych. 2017;11(6):4-7.
25. Adenosine; Package insert. Astellas Pharma US Inc; 1989.
26. Dipyridamole; Package insert. Boehringer Ingelheim Pharmaceuticals Inc; 2019.
27. Dobutamine; Package insert. Baxter Healthcare Corporation; 2012.
A Practical Approach for Primary Care Practitioners to Evaluate and Manage Lower Urinary Tract Symptoms and Benign Prostatic Hyperplasia
Lower urinary tract symptoms (LUTS)are common and tend to increase in frequency with age. Managing LUTS can be complicated, requires an informed discussion between the primary care practitioner (PCP) and patient, and is best achieved by a thorough understanding of the many medical and surgical options available. Over the past 3 decades, medications have become the most common therapy; but recently, newer minimally invasive surgeries have challenged this paradigm. This article provides a comprehensive review for PCPs regarding the evaluation and management of LUTS in men and when to consider a urology referral.
Benign prostatic hyperplasia (BPH) and LUTS are common clinical encounters for most PCPs. About 50% of men will develop LUTS associated with BPH, and symptoms associated with these conditions increase as men age.1,2 Studies have estimated that 90% of men aged 45 to 80 years demonstrate some symptoms of LUTS.3 Strong genetic influence seems to suggest heritability, but BPH also occurs in sporadic forms and is heavily influenced by androgens.4
BPH is a histologic diagnosis, whereas LUTS consists of complex symptomatology related to both static or dynamic components.1 The enlarged prostate gland obstructs the urethra, simultaneously causing an increase in muscle tone and resistance at the bladder neck and prostatic urethra, leading to increased resistance to urine flow. As a result, there is a thickening of the detrusor muscles in the bladder wall and an overall decreased compliance. Urine becomes stored under increased pressure. These changes result in a weak or intermittent urine stream, incomplete emptying of the bladder, postvoid dribble, hesitancy, and irritative symptoms, such as urgency, frequency, and nocturia.
For many patients, BPH associated with LUTS is a quality of life (QOL) issue. The stigma associated with these symptoms often leads to delays in patients seeking care. Many patients do not seek treatment until symptoms have become so severe that changes in bladder health are often irreversible. Early intervention can dramatically improve a patient’s QOL. Also, early intervention has the potential to reduce overall health care expenditures. BPH-related spending exceeds $1 billion each year in the Medicare program alone.5
PCPs are in a unique position to help many patients who present with early-stage LUTS. Given the substantial impact this disease has on QOL, early recognition of symptoms and prompt treatment play a major role. Paramount to this effort is awareness and understanding of various treatments, their advantages, and adverse effects (AEs). This article highlights evidence-based evaluation and treatment of BPH/LUTS for PCPs who treat veterans and recommendations as to when to refer a patient to a urologist.
Evaluation of LUTS and BPH
Evaluation begins with a thorough medical history and physical examination. Particular attention should focus on ruling out other causes of LUTS, such as a urinary tract infection (UTI), acute prostatitis, malignancy, bladder dysfunction, neurogenic bladder, and other obstructive pathology, such as urethral stricture disease. The differential diagnosis of LUTS includes BPH, UTI, bladder neck obstruction, urethral stricture, bladder stones, polydipsia, overactive bladder (OAB), nocturnal polyuria, neurologic disease, genitourinary malignancy, renal failure, and acute/chronic urinary retention.6
Relevant medical history influencing urinary symptoms includes diabetes mellitus, underlying neurologic diseases, previous trauma, sexually transmitted infections, and certain medications. Symptom severity may be obtained using a validated questionnaire, such as the International Prostate Symptom Score (IPSS), which also aids clinicians in assessing the impact of LUTS on QOL. Additionally, urinary frequency or volume records (voiding diary) may help establish the severity of the patient’s symptoms and provide insight into other potential causes for LUTS. Patients with BPH often have concurrent erectile dysfunction (ED) or other sexual dysfunction symptoms. Patients should be evaluated for baseline sexual dysfunction before the initiation of treatment as many therapies worsen symptoms of ED or ejaculatory dysfunction.
A comprehensive physical examination with a focus on the genitourinary system should, at minimum, assess for abnormalities of the urethral meatus, prepuce, penis, groin nodes, and prior surgical scars. A digital rectal examination also should be performed. Although controversial, a digital rectal examination for prostate cancer screening may provide a rough estimate of prostate size, help rule out prostatitis, and detect incident prostate nodules. Prostate size does not necessarily correlate well with the degree of urinary obstruction or LUTS but is an important consideration when deciding among different therapies.1
Laboratory and Adjunctive Tests
A urinalysis with microscopy helps identify other potential causes for urinary symptoms, including infection, proteinuria, or glucosuria. In patients who present with gross or microscopic hematuria, additional consideration should be given to bladder calculi and genitourinary cancer.2 When a reversible source for the hematuria is not identified, these patients require referral to a urologist for a hematuria evaluation.
There is some controversy regarding prostate specific antigen (PSA) testing. Most professional organizations advocate for a shared decision-making approach before testing. The American Cancer Society recommends this informed discussion occur between the patient and the PCP for men aged > 50 years at average risk, men aged > 45 years at high risk of developing prostate cancer (African Americans or first-degree relative with early prostate cancer diagnosis), and aged 40 years for men with more than one first-degree relative with an early prostate cancer diagnosis.7
Adjunctive tests include postvoid residual (PVR), cystoscopy, uroflowmetry, urodynamics, and transrectal ultrasound. However, these are mostly performed by urologists. In some patients with bladder decompensation after prolonged partial bladder outlet obstruction, urodynamics may be used by urologists to determine whether a patient may benefit from an outlet obstruction procedure. Ordering additional imaging or serum studies for the assessment of LUTS is rarely helpful.
Treatment
Treatment includes management with or without lifestyle modification, medication administration, and surgical therapy. New to this paradigm are in-office minimally invasive surgical options. The goal of treatment is not only to reduce patient symptoms and improve QOL, but also to prevent the secondary sequala of urinary retention, bladder failure, and eventual renal impairment.7A basic understanding of these treatments can aid PCPs with appropriate patient counseling and urologic referral.8
Lifestyle and Behavior Modification
Behavior modification is the starting point for all patients with LUTS. Lifestyle modifications for LUTS include avoiding substances that exacerbate symptoms, such as α-agonists (decongestants), caffeine, alcohol, spicy/acidic foods, chocolate, and soda. These substances are known to be bladder irritants. Common medications contributing to LUTS include antidepressants, decongestants, antihistamines, bronchodilators, anticholinergics, and sympathomimetics. To decrease nocturia, behavioral modifications include limiting evening fluid intake, timed diuretic administration for patients already on a diuretic, and elevating legs 1 hour before bedtime. Counseling obese patients to lose weight and increasing physical activity have been linked to reduced LUTS.9 Other behavioral techniques include double voiding: a technique where patients void normally then change positions and return to void to empty the bladder. Another technique is timed voiding: Many patients have impaired sensation when the bladder is full. These patients are encouraged to void at regular intervals.
Complementary and Alternative Medicine
Multiple nutraceutical compounds claim improved urinary health and symptom reduction. These compounds are marketed to patients with little regulation and oversight since supplements are not regulated or held to the same standard as prescription medications. The most popular nutraceutical for prostate health and LUTS is saw palmetto. Despite its common usage for the treatment of LUTS, little data support saw palmetto health claims. In 2012, a systematic review of 32 randomized trials including 5666 patients compared saw palmetto with a placebo. The study found no difference in urinary symptom scores, urinary flow, or prostate size.10,11 Other phytotherapy compounds often considered for urinary symptoms include stinging nettle extract and β-sitosterol compounds. The mechanism of action of these agents is unknown and efficacy data are lacking.
Historically, acupuncture and pelvic floor physical therapy have been used successfully for OAB symptoms. A meta-analysis found positive beneficial effects of acupuncture compared with a sham control for short- and medium-term follow-up in both IPSS and urine flow rates in some studies; however, when combining the studies for more statistical power, the benefits were less clear.12 Physical therapists with specialized training and certification in pelvic health can incorporate certain bladder training techniques. These include voiding positional changes (double voiding and postvoid urethral milking) and timed voiding.13,14 These interventions often address etiologies of LUTS for which medical therapies are not effective as the sole treatment option.
Medication Management
Medical management includes α-blockers, 5-α-reductase inhibitors (5-α-RIs), antimuscarinic or anticholinergic medicines, β-3 agonists, and phosphodiesterase inhibitors (Table). These medications work independently as well as synergistically. The use of medications to improve symptoms must be balanced against potential AEs and the consequences of a lifetime of drug usage, which can be additive.15,16
First-line pharmacological therapy for BPH is α-blockers, which work by blocking α1A receptors in the prostate and bladder neck, leading to smooth muscle relaxation, increased diameter of the channel, and improved urinary flow. α-receptors in the bladder neck and prostate are expressed with increased frequency with age and are a potential cause for worsening symptoms as men age. Studies demonstrate that these medications reduce symptoms by 30 to 40% and increase flow rates by 16 to 25%.17 Commonly prescribed α-blockers include tamsulosin, alfuzosin, silodosin, doxazosin, and terazosin. Doxazosin and terazosin require dose titrations because they may cause significant hypotension. Orthostatic hypotension typically improves with time and is avoided if the patient takes the medication at bedtime. Both doxazosin and terazosin are on the American Geriatric Society’s Beers Criteria list and should be avoided in older patients.18 Tamsulosin, alfuzosin, and silodosin have a standardized dosing regimen and lower rates of hypotension. Significant AEs include ejaculation dysfunction, nasal congestion, and orthostatic hypotension. Duan and colleagues have linked tamsulosin with dementia. However, this association is not causal and further studies are necessary.19,20 Patients who have taken these agents also are at risk for intraoperative floppy iris syndrome (IFIS). Permanent visual problems can arise if the intraoperative management is not managed to account for IFIS. These medications have a rapid onset of action and work immediately. However, to reach maximum benefit, patients must take the medication for several weeks. Unfortunately, up to one-third of patients will have no improvement with α-blocker therapy, and many patients will discontinue these medications because of significant AEs.6,21
5-α-RIs (finasteride and dutasteride) inhibit the conversion of testosterone to more potent dihydrotestosterone. They effectively reduce prostate volume by 25 to 30%.22 The results occur slowly and can take 6 to 12 months to reach the desired outcome. These medications are effective in men with larger prostates and not as effective in men with smaller prostates.23 These medications can improve urinary flow rates by about 10%, reduce IPSS scores by 20 to 30%, reduce the risk of urinary retention by 50%, and reduce the progression of BPH to the point where surgery is required by 50%.24 Furthermore, 5-α-RIs lower PSA by > 50% after 12 months of treatment.25
A baseline PSA should be established before administration and after 6 months of treatment. Any increase in the PSA even if the level is within normal limits should be evaluated for prostate cancer. Sarkar and colleagues recently published a study evaluating prostate cancer diagnosis in patients treated with 5-α-RI and found there was a delay in diagnosing prostate cancer in this population. Controversy also exists as to the potential of these medications increasing the risk for high-grade prostate cancer, which has led to a US Food and Drug Administration (FDA) warning. AEs include decreased libido (1.5%), ejaculatory dysfunction (3.4%), gynecomastia (1.3%), and/or ED (1.6%).26-28 A recent study evaluating 5-α-RIs demonstrated about a 2-fold increased risk of depression.29
There are well-established studies that note increased effectiveness when using combined α-blocker therapy with 5-α-RI medications. The Medical Therapy of Prostate Symptoms (MTOPS) and Combination Avodart and Tamsulosin (CombAT) trials showed that the combination of both medications was more effective in improving voiding symptoms and flow rates than either agent alone.15,16 Combination therapy resulted in a 66% reduction in disease progression, 81% reduction in urinary retention, and a 67% reduction in the need for surgery compared with placebo.
Anticholinergic medication use in BPH with LUTS is well established, and their use is often combined with other therapies. Anticholinergics work by inhibiting muscarinic M3 receptors to reduce detrusor muscle contraction. This effectively decreases bladder contractions and delays the desire to void. Kaplan and colleagues showed that tolterodine significantly improved a patient’s QOL when added to α-blocker therapy.30 Patients reported a positive outcome at 12 weeks, which resulted in a reduction in urgency incontinence, urgency, nocturia, and the overall number of voiding episodes within 24 hours.
β-3 agonists are a class of medications for OAB; mirabegron and vibegron have proven effective in reducing similar symptoms. In phase 3 clinical trials, mirabegron improved urinary incontinence episodes by 50% and reduced the number of voids in 24 hours.31 Mirabegron is well tolerated and avoids many common anticholinergic effects.32 Vibegron is the newest medication in the class and could soon become the preferred agent given it does not have cytochrome P450 interactions and does not cause hypertension like mirabegron.33
Anticholinergics should be used with caution in patients with a history of urinary retention, elevated after-void residual, or other medications with known anticholinergic effects. AEs include sedation, confusion, dry mouth, constipation, and potential falls in older patients.18 Recent studies have noted an association with dementia in the prolonged use of these medications in older patients and should be used cautiously.20
Phosphodiesterase-5 enzyme inhibitors (PDE-5) are adjunctive medications shown to improve LUTS. This class of medication is prescribed mostly for ED. However, tadalafil 5 mg taken daily also is FDA approved for the treatment of LUTS secondary to BPH given its prolonged half-life. The exact mechanism for improved BPH symptoms is unknown. Possibly the effects are due to an increase mediated by PDE-5 in cyclic guanosine monophosphate (cGMP), which increases smooth muscle relaxation and tissue perfusion of the prostate and bladder.34 There have been limited studies on objective improvement in uroflowmetry parameters compared with other treatments. The daily dosing of tadalafil should not be prescribed in men with a creatinine clearance < 30 mL/min.29 Tadalafil is not considered a first-line agent and is usually reserved for patients who experience ED in addition to BPH. When initiating BPH pharmacologic therapy, the PCP should be aware of adherence and high discontinuation rates.35
Surgical Treatments
Surgical treatments are often delayed out of fear of potential AEs or considered a last resort when symptoms are too severe.36 Early intervention is required to prevent irreversible deleterious changes to detrusor muscle structure and function (Figure). Patients fear urinary incontinence, ED or ejaculatory dysfunction, and anesthesia complications associated with surgical interventions.6,37 Multiple studies show that patients fare better with early surgical intervention, experiencing improved IPSS scores, urinary flow, and QOL. The following is an overview of the most popular procedures.
Prostatic urethral lift (PUL) using the UroLift System is an FDA-approved, minimally-invasive treatment of LUTS secondary to BPH. This procedure treats prostates < 80 g with an absent median lobe.6,21,38 Permanent implants are placed per the prostatic urethra to displace obstructing prostate tissue laterally. This opens the urethra directly without cutting, heating, or removing any prostate tissue. This procedure is minimally invasive, often done in the office as an outpatient procedure, and offers better symptom relief than medication with a lower risk profile than transurethral resection of the prostate (TURP).39,40 The LIFT study was a multicenter, randomized, blinded trial; patients were randomized 2:1 to undergo UroLift or a sham operation. At 3 years, average improvements were statistically significant for total IPSS reduction (41%), QOL improvement (49%), and improved maximum flow rates by (51%).41 Risk for urinary incontinence is low, and the procedure has been shown to preserve erectile and ejaculatory function. Furthermore, patients report significant improvement in their QOL without the need for medications. Surgical retreatment rates at 5 years are 13.6%, with an additional 10.7% of subjects back on medication therapy with α-blockers or 5-α-RIs.42
Water vapor thermal therapy or Rez¯um uses steam as thermal energy to destroy obstructing prostate tissue and relieve the obstruction.43 The procedure differs from older conductive heat thermotherapies because the steam penetrates prostate zonal anatomy without affecting areas outside the targeted treatment zone. The procedure is done in the office with local anesthesia and provides long-lasting relief of LUTS with minimal risks. Following the procedure, patients require an indwelling urethral catheter for 3 to 7 days, and most patients begin to experience symptom improvement 2 to 4 weeks following the procedure.44 The procedure received FDA approval in 2015. Four-year data show significant improvement in maximal flow rate (50%), IPSS (47%), and QOL (43%).45 Surgical retreatment rates were 4.4%. Criticisms of this treatment include patient discomfort with the office procedure, the requirement for an indwelling catheter for a short period, and lack of long-term outcomes data. Guidelines support use in prostate volumes > 80 g with or without median lobe anatomy.
TURP is the gold standard to which other treatments are compared.46 The surgery is performed in the operating room where urologists use a rigid cystoscope and resection element to effectively carve out and cauterize obstructing prostate tissue. Patients typically recover for a short period with an indwelling urethral catheter that is often removed 12 to 24 hours after surgery. New research points out that despite increasing mean age (55% of patients are aged > 70 years with associated comorbidities), the morbidity of TURP was < 1% and mortality rate of 0 to 0.3%.47 Postoperative complications include bleeding that requires a transfusion (3%), retrograde ejaculation (65%), and rare urinary incontinence (2%).47 Surgical retreatment rates for patients following a TURP are approximately 13 to 15% at 8 years.34
Laser surgery for BPH includes multiple techniques: photovaporization of the prostate using a Greenlight XPS laser, holmium laser ablation, and holmium laser enucleation (HoLEP). Proponents of these treatments cite lower bleeding risks compared with TURP, but the operation is largely surgeon dependent on the technology chosen. Most studies comparing these technologies with TURP show similar outcomes of IPSS reports, quality of life improvements, and complications.
Patients with extremely large prostates, > 100 g or 4 times the normal size, pose a unique challenge to surgical treatment. Historically, patients were treated with an open simple prostatectomy operation or staged TURP procedures. Today, urologists use newer, safer ways to treat these patients. Both HoLEP and robot-assisted simple prostatectomy work well in relieving urinary symptoms with lower complications compared with older open surgery. Other minimally invasive procedures, such as prostatic artery embolism, have been described for the treatment of BPH specifically in men who may be unfit for surgery.48Future treatments are constantly evolving. Many unanswered questions remain about BPH and the role of inflammation, metabolic dysfunction, obesity, and other genetic factors driving BPH and prostate growth. Pharmaceutical opportunities exist in mechanisms aimed to reduce prostate growth, induce cellular apoptosis, as well as other drugs to reduce bladder symptoms. Newer, minimally invasive therapies also will become more readily available, such as Aquablation, which is the first FDA-granted surgical robot for the autonomous removal of prostatic tissue due to BPH.49 However, the goal of all future therapies should include the balance of alleviating disruptive symptoms while demonstrating a favorable risk profile. Many men discontinue taking medications, yet few present for surgery. Most concerning is the significant population of men who will develop irreversible bladder dysfunction while waiting for the perfect treatment. There are many opportunities for an effective treatment that is less invasive than surgery, provides durable relief, has minimal AEs, and is affordable.
Conclusions
There is no perfect treatment for patients with LUTS. All interventions have potential AEs and associated complications. Medications are often started as first-line therapy but are often discontinued at the onset of significant AEs. This process is often repeated. Many patients will try different medications without any significant improvement in their symptoms or short-term relief, which results in the gradual progression of the disease.
The PCP plays a significant role in the initial evaluation and management of BPH. These frontline clinicians can recognize patients who may already be experiencing sequela of prolonged bladder outlet obstruction and refer these men to urologists promptly. Counseling patients about their treatment options is an important duty for all PCPs.
A clear understanding of the available treatment options will help PCPs counsel patients appropriately about lifestyle modification, medications, and surgical treatment options for their symptoms. The treatment of this disorder is a rapidly evolving topic with the constant introduction of new technologies and medications, which are certain to continue to play an important role for PCPs and urologists.
1. Roehrborn CG. Benign prostatic hyperplasia: an overview. Rev Urol. 2005;7 Suppl 9(Suppl 9):S3-S14
2. McVary KT. Clinical manifestations and diagnostic evaluation of benign prostatic hyperplasia. UpToDate. Updated November 18, 2021. Accessed November 23, 2021. https:// www.uptodate.com/contents/clinical-manifestations-and -diagnostic-evaluation-of-benign-prostatic-hyperplasia
3. McVary KT. BPH: epidemiology and comorbidities. Am J Manag Care. 2006;12(5 Suppl):S122-S128.
4. Ho CK, Habib FK. Estrogen and androgen signaling in the pathogenesis of BPH. Nat Rev Urol. 2011;8(1):29-41. doi:10.1038/nrurol.2010.207
5. Rensing AJ, Kuxhausen A, Vetter J, Strope SA. Differences in the treatment of benign prostatic hyperplasia: comparing the primary care physician and the urologist. Urol Pract. 2017;4(3):193-199. doi:10.1016/j.urpr.2016.07.002
6. Foster HE, Barry MJ, Dahm P, et al. Surgical management of lower urinary tract symptoms attributed to benign prostatic hyperplasia: AUA guideline. J Urol. 2018;200(3):612- 619. doi:10.1016/j.juro.2018.05.048
7. Landau A, Welliver C. Analyzing and characterizing why men seek care for lower urinary tract symptoms. Curr Urol Rep. 2020;21(12):58. Published 2020 Oct 30. doi:10.1007/s11934-020-01006-w
8. Das AK, Leong JY, Roehrborn CG. Office-based therapies for benign prostatic hyperplasia: a review and update. Can J Urol. 2019;26(4 Suppl 1):2-7.
9. Parsons JK, Sarma AV, McVary K, Wei JT. Obesity and benign prostatic hyperplasia: clinical connections, emerging etiological paradigms and future directions. J Urol. 2013;189(1 Suppl):S102-S106. doi:10.1016/j.juro.2012.11.029
10. Pattanaik S, Mavuduru RS, Panda A, et al. Phosphodiesterase inhibitors for lower urinary tract symptoms consistent with benign prostatic hyperplasia. Cochrane Database Syst Rev. 2018;11(11):CD010060. Published 2018 Nov 16. doi:10.1002/14651858.CD010060.pub2
11. McVary KT. Medical treatment of benign prostatic hyperplasia. UpToDate. Updated October 4, 2021. Accessed November 23, 2021. https://www.uptodate.com/contents /medical-treatment-of-benign-prostatic-hyperplasia
12. Zhang W, Ma L, Bauer BA, Liu Z, Lu Y. Acupuncture for benign prostatic hyperplasia: A systematic review and metaanalysis. PLoS One. 2017;12(4):e0174586. Published 2017 Apr 4. doi:10.1371/journal.pone.0174586
13. Newman DK, Guzzo T, Lee D, Jayadevappa R. An evidence- based strategy for the conservative management of the male patient with incontinence. Curr Opin Urol. 2014;24(6):553-559. doi:10.1097/MOU.0000000000000115
14. Newman DK, Wein AJ. Office-based behavioral therapy for management of incontinence and other pelvic disorders. Urol Clin North Am. 2013;40(4):613-635. doi:10.1016/j.ucl.2013.07.010
15. McConnell JD, Roehrborn CG, Bautista OM, et al. The long-term effect of doxazosin, finasteride, and combination therapy on the clinical progression of benign prostatic hyperplasia. N Engl J Med. 2003;349(25):2387-2398. doi:10.1056/NEJMoa030656
16. Roehrborn CG, Barkin J, Siami P, et al. Clinical outcomes after combined therapy with dutasteride plus tamsulosin or either monotherapy in men with benign prostatic hyperplasia (BPH) by baseline characteristics: 4-year results from the randomized, double-blind Combination of Avodart and Tamsulosin (CombAT) trial. BJU Int. 2011;107(6):946-954. doi:10.1111/j.1464-410X.2011.10124.x
17. Djavan B, Marberger M. A meta-analysis on the efficacy and tolerability of alpha1-adrenoceptor antagonists in patients with lower urinary tract symptoms suggestive of benign prostatic obstruction. Eur Urol. 1999;36(1):1-13. doi:10.1159/000019919
18. By the American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American Geriatrics Society 2015 Updated Beers Criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2015;63(11):2227-2246. doi:10.1111/jgs.13702
19. Duan Y, Grady JJ, Albertsen PC, Helen Wu Z. Tamsulosin and the risk of dementia in older men with benign prostatic hyperplasia. Pharmacoepidemiol Drug Saf. 2018;27(3):340- 348. doi:10.1002/pds.4361
20. Coupland CAC, Hill T, Dening T, Morriss R, Moore M, Hippisley-Cox J. Anticholinergic drug exposure and the risk of dementia: a nested case-control study. JAMA Intern Med. 2019;179(8):1084-1093. doi:10.1001/jamainternmed.2019.0677
21. Parsons JK, Dahm P, Köhler TS, Lerner LB, Wilt TJ. Surgical management of lower urinary tract symptoms attributed to benign prostatic hyperplasia: AUA guideline amendment 2020. J Urol. 2020;204(4):799-804. doi:10.1097/JU.0000000000001298
22. Smith AB, Carson CC. Finasteride in the treatment of patients with benign prostatic hyperplasia: a review. Ther Clin Risk Manag. 2009;5(3):535-545. doi:10.2147/tcrm.s6195
23. Andriole GL, Guess HA, Epstein JI, et al. Treatment with finasteride preserves usefulness of prostate-specific antigen in the detection of prostate cancer: results of a randomized, double-blind, placebo-controlled clinical trial. PLESS Study Group. Proscar Long-term Efficacy and Safety Study. Urology. 1998;52(2):195-202. doi:10.1016/s0090-4295(98)00184-8
24. McConnell JD, Bruskewitz R, Walsh P, et al. The effect of finasteride on the risk of acute urinary retention and the need for surgical treatment among men with benign prostatic hyperplasia. Finasteride Long-Term Efficacy and Safety Study Group. N Engl J Med. 1998;338(9):557-563. doi:10.1056/NEJM199802263380901
25. Rittmaster RS. 5alpha-reductase inhibitors in benign prostatic hyperplasia and prostate cancer risk reduction. Best Pract Res Clin Endocrinol Metab. 2008;22(2):389-402. doi:10.1016/j.beem.2008.01.016
26. La Torre A, Giupponi G, Duffy D, Conca A, Cai T, Scardigli A. Sexual dysfunction related to drugs: a critical review. Part V: α-blocker and 5-ARI drugs. Pharmacopsychiatry. 2016;49(1):3-13. doi:10.1055/s-0035-1565100
27. Corona G, Tirabassi G, Santi D, et al. Sexual dysfunction in subjects treated with inhibitors of 5α-reductase for benign prostatic hyperplasia: a comprehensive review and meta-analysis. Andrology. 2017;5(4):671-678. doi:10.1111/andr.12353
28. Trost L, Saitz TR, Hellstrom WJ. Side effects of 5-alpha reductase inhibitors: a comprehensive review. Sex Med Rev. 2013;1(1):24-41. doi:10.1002/smrj.3
29. Welk B, McArthur E, Ordon M, Anderson KK, Hayward J, Dixon S. Association of suicidality and depression with 5α-reductase inhibitors. JAMA Intern Med. 2017;177(5):683-691. doi:10.1001/jamainternmed.2017.0089
30. Kaplan SA, Roehrborn CG, Rovner ES, Carlsson M, Bavendam T, Guan Z. Tolterodine and tamsulosin for treatment of men with lower urinary tract symptoms and overactive bladder: a randomized controlled trial [published correction appears in JAMA. 2007 Mar 21:297(11):1195] [published correction appears in JAMA. 2007 Oct 24;298(16):1864]. JAMA. 2006;296(19):2319-2328. doi:10.1001/jama.296.19.2319
31. Nitti VW, Auerbach S, Martin N, Calhoun A, Lee M, Herschorn S. Results of a randomized phase III trial of mirabegron in patients with overactive bladder. J Urol. 2013;189(4):1388-1395. doi:10.1016/j.juro.2012.10.017
32. Chapple CR, Cardozo L, Nitti VW, Siddiqui E, Michel MC. Mirabegron in overactive bladder: a review of efficacy, safety, and tolerability. Neurourol Urodyn. 2014;33(1):17-30. doi:10.1002/nau.22505
33. Rutman MP, King JR, Bennett N, Ankrom W, Mudd PN. PD14-01 once-daily vibegron, a novel oral β3 agonist does not inhibit CYP2D6, a common pathway for drug metabolism in patients on OAB medications. J Urol. 2019;201(Suppl 4):e231. doi:10.1097/01.JU.0000555478.73162.19
34. Bo K, Frawley HC, Haylen BT, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for the conservative and nonpharmacological management of female pelvic floor dysfunction. Neurourol Urodyn. 2017;36(2):221- 244. doi:10.1002/nau.23107
35. Cindolo L, Pirozzi L, Fanizza C, et al. Drug adherence and clinical outcomes for patients under pharmacological therapy for lower urinary tract symptoms related to benign prostatic hyperplasia: population-based cohort study. Eur Urol. 2015;68(3):418-425. doi:10.1016/j.eururo.2014.11.006
36. Ruhaiyem ME, Alshehri AA, Saade M, Shoabi TA, Zahoor H, Tawfeeq NA. Fear of going under general anesthesia: a cross-sectional study. Saudi J Anaesth. 2016;10(3):317- 321. doi:10.4103/1658-354X.179094
37. Hashim MJ. Patient-centered communication: basic skills. Am Fam Physician. 2017;95(1):29-34.
38. Roehrborn CG, Barkin J, Gange SN, et al. Five year results of the prospective randomized controlled prostatic urethral L.I.F.T. study. Can J Urol. 2017;24(3):8802-8813.
39. Gratzke C, Barber N, Speakman MJ, et al. Prostatic urethral lift vs transurethral resection of the prostate: 2-year results of the BPH6 prospective, multicentre, randomized study. BJU Int. 2017;119(5):767-775.doi:10.1111/bju.13714
40. Sønksen J, Barber NJ, Speakman MJ, et al. Prospective, randomized, multinational study of prostatic urethral lift versus transurethral resection of the prostate: 12-month results from the BPH6 study. Eur Urol. 2015;68(4):643-652. doi:10.1016/j.eururo.2015.04.024
41. Roehrborn CG, Gange SN, Shore ND, et al. The prostatic urethral lift for the treatment of lower urinary tract symptoms associated with prostate enlargement due to benign prostatic hyperplasia: the L.I.F.T. Study. J Urol. 2013;190(6):2161-2167. doi:10.1016/j.juro.2013.05.116
42. McNicholas TA. Benign prostatic hyperplasia and new treatment options - a critical appraisal of the UroLift system. Med Devices (Auckl). 2016;9:115-123. Published 2016 May 19. doi:10.2147/MDER.S60780
43. McVary KT, Rogers T, Roehrborn CG. Rezuˉm Water Vapor thermal therapy for lower urinary tract symptoms associated with benign prostatic hyperplasia: 4-year results from randomized controlled study. Urology. 2019;126:171-179. doi:10.1016/j.urology.2018.12.041
44. Bole R, Gopalakrishna A, Kuang R, et al. Comparative postoperative outcomes of Rezˉum prostate ablation in patients with large versus small glands. J Endourol. 2020;34(7):778-781. doi:10.1089/end.2020.0177
45. Darson MF, Alexander EE, Schiffman ZJ, et al. Procedural techniques and multicenter postmarket experience using minimally invasive convective radiofrequency thermal therapy with Rezˉum system for treatment of lower urinary tract symptoms due to benign prostatic hyperplasia. Res Rep Urol. 2017;9:159-168. Published 2017 Aug 21. doi:10.2147/RRU.S143679
46. Baazeem A, Elhilali MM. Surgical management of benign prostatic hyperplasia: current evidence. Nat Clin Pract Urol. 2008;5(10):540-549. doi:10.1038/ncpuro1214
47. Rassweiler J, Teber D, Kuntz R, Hofmann R. Complications of transurethral resection of the prostate (TURP)- -incidence, management, and prevention. Eur Urol. 2006;50(5):969-980. doi:10.1016/j.eururo.2005.12.042
48. Abt D, Schmid HP, Speakman MJ. Reasons to consider prostatic artery embolization. World J Urol. 2021;39(7):2301-2306. doi:10.1007/s00345-021-03601-z
49. Nguyen DD, Barber N, Bidair M, et al. Waterjet Ablation Therapy for Endoscopic Resection of prostate tissue trial (WATER) vs WATER II: comparing Aquablation therapy for benign prostatic hyperplasia in30-80and80-150mLprostates. BJUInt. 2020;125(1):112-122. doi:10.1111/bju.14917.
Lower urinary tract symptoms (LUTS)are common and tend to increase in frequency with age. Managing LUTS can be complicated, requires an informed discussion between the primary care practitioner (PCP) and patient, and is best achieved by a thorough understanding of the many medical and surgical options available. Over the past 3 decades, medications have become the most common therapy; but recently, newer minimally invasive surgeries have challenged this paradigm. This article provides a comprehensive review for PCPs regarding the evaluation and management of LUTS in men and when to consider a urology referral.
Benign prostatic hyperplasia (BPH) and LUTS are common clinical encounters for most PCPs. About 50% of men will develop LUTS associated with BPH, and symptoms associated with these conditions increase as men age.1,2 Studies have estimated that 90% of men aged 45 to 80 years demonstrate some symptoms of LUTS.3 Strong genetic influence seems to suggest heritability, but BPH also occurs in sporadic forms and is heavily influenced by androgens.4
BPH is a histologic diagnosis, whereas LUTS consists of complex symptomatology related to both static or dynamic components.1 The enlarged prostate gland obstructs the urethra, simultaneously causing an increase in muscle tone and resistance at the bladder neck and prostatic urethra, leading to increased resistance to urine flow. As a result, there is a thickening of the detrusor muscles in the bladder wall and an overall decreased compliance. Urine becomes stored under increased pressure. These changes result in a weak or intermittent urine stream, incomplete emptying of the bladder, postvoid dribble, hesitancy, and irritative symptoms, such as urgency, frequency, and nocturia.
For many patients, BPH associated with LUTS is a quality of life (QOL) issue. The stigma associated with these symptoms often leads to delays in patients seeking care. Many patients do not seek treatment until symptoms have become so severe that changes in bladder health are often irreversible. Early intervention can dramatically improve a patient’s QOL. Also, early intervention has the potential to reduce overall health care expenditures. BPH-related spending exceeds $1 billion each year in the Medicare program alone.5
PCPs are in a unique position to help many patients who present with early-stage LUTS. Given the substantial impact this disease has on QOL, early recognition of symptoms and prompt treatment play a major role. Paramount to this effort is awareness and understanding of various treatments, their advantages, and adverse effects (AEs). This article highlights evidence-based evaluation and treatment of BPH/LUTS for PCPs who treat veterans and recommendations as to when to refer a patient to a urologist.
Evaluation of LUTS and BPH
Evaluation begins with a thorough medical history and physical examination. Particular attention should focus on ruling out other causes of LUTS, such as a urinary tract infection (UTI), acute prostatitis, malignancy, bladder dysfunction, neurogenic bladder, and other obstructive pathology, such as urethral stricture disease. The differential diagnosis of LUTS includes BPH, UTI, bladder neck obstruction, urethral stricture, bladder stones, polydipsia, overactive bladder (OAB), nocturnal polyuria, neurologic disease, genitourinary malignancy, renal failure, and acute/chronic urinary retention.6
Relevant medical history influencing urinary symptoms includes diabetes mellitus, underlying neurologic diseases, previous trauma, sexually transmitted infections, and certain medications. Symptom severity may be obtained using a validated questionnaire, such as the International Prostate Symptom Score (IPSS), which also aids clinicians in assessing the impact of LUTS on QOL. Additionally, urinary frequency or volume records (voiding diary) may help establish the severity of the patient’s symptoms and provide insight into other potential causes for LUTS. Patients with BPH often have concurrent erectile dysfunction (ED) or other sexual dysfunction symptoms. Patients should be evaluated for baseline sexual dysfunction before the initiation of treatment as many therapies worsen symptoms of ED or ejaculatory dysfunction.
A comprehensive physical examination with a focus on the genitourinary system should, at minimum, assess for abnormalities of the urethral meatus, prepuce, penis, groin nodes, and prior surgical scars. A digital rectal examination also should be performed. Although controversial, a digital rectal examination for prostate cancer screening may provide a rough estimate of prostate size, help rule out prostatitis, and detect incident prostate nodules. Prostate size does not necessarily correlate well with the degree of urinary obstruction or LUTS but is an important consideration when deciding among different therapies.1
Laboratory and Adjunctive Tests
A urinalysis with microscopy helps identify other potential causes for urinary symptoms, including infection, proteinuria, or glucosuria. In patients who present with gross or microscopic hematuria, additional consideration should be given to bladder calculi and genitourinary cancer.2 When a reversible source for the hematuria is not identified, these patients require referral to a urologist for a hematuria evaluation.
There is some controversy regarding prostate specific antigen (PSA) testing. Most professional organizations advocate for a shared decision-making approach before testing. The American Cancer Society recommends this informed discussion occur between the patient and the PCP for men aged > 50 years at average risk, men aged > 45 years at high risk of developing prostate cancer (African Americans or first-degree relative with early prostate cancer diagnosis), and aged 40 years for men with more than one first-degree relative with an early prostate cancer diagnosis.7
Adjunctive tests include postvoid residual (PVR), cystoscopy, uroflowmetry, urodynamics, and transrectal ultrasound. However, these are mostly performed by urologists. In some patients with bladder decompensation after prolonged partial bladder outlet obstruction, urodynamics may be used by urologists to determine whether a patient may benefit from an outlet obstruction procedure. Ordering additional imaging or serum studies for the assessment of LUTS is rarely helpful.
Treatment
Treatment includes management with or without lifestyle modification, medication administration, and surgical therapy. New to this paradigm are in-office minimally invasive surgical options. The goal of treatment is not only to reduce patient symptoms and improve QOL, but also to prevent the secondary sequala of urinary retention, bladder failure, and eventual renal impairment.7A basic understanding of these treatments can aid PCPs with appropriate patient counseling and urologic referral.8
Lifestyle and Behavior Modification
Behavior modification is the starting point for all patients with LUTS. Lifestyle modifications for LUTS include avoiding substances that exacerbate symptoms, such as α-agonists (decongestants), caffeine, alcohol, spicy/acidic foods, chocolate, and soda. These substances are known to be bladder irritants. Common medications contributing to LUTS include antidepressants, decongestants, antihistamines, bronchodilators, anticholinergics, and sympathomimetics. To decrease nocturia, behavioral modifications include limiting evening fluid intake, timed diuretic administration for patients already on a diuretic, and elevating legs 1 hour before bedtime. Counseling obese patients to lose weight and increasing physical activity have been linked to reduced LUTS.9 Other behavioral techniques include double voiding: a technique where patients void normally then change positions and return to void to empty the bladder. Another technique is timed voiding: Many patients have impaired sensation when the bladder is full. These patients are encouraged to void at regular intervals.
Complementary and Alternative Medicine
Multiple nutraceutical compounds claim improved urinary health and symptom reduction. These compounds are marketed to patients with little regulation and oversight since supplements are not regulated or held to the same standard as prescription medications. The most popular nutraceutical for prostate health and LUTS is saw palmetto. Despite its common usage for the treatment of LUTS, little data support saw palmetto health claims. In 2012, a systematic review of 32 randomized trials including 5666 patients compared saw palmetto with a placebo. The study found no difference in urinary symptom scores, urinary flow, or prostate size.10,11 Other phytotherapy compounds often considered for urinary symptoms include stinging nettle extract and β-sitosterol compounds. The mechanism of action of these agents is unknown and efficacy data are lacking.
Historically, acupuncture and pelvic floor physical therapy have been used successfully for OAB symptoms. A meta-analysis found positive beneficial effects of acupuncture compared with a sham control for short- and medium-term follow-up in both IPSS and urine flow rates in some studies; however, when combining the studies for more statistical power, the benefits were less clear.12 Physical therapists with specialized training and certification in pelvic health can incorporate certain bladder training techniques. These include voiding positional changes (double voiding and postvoid urethral milking) and timed voiding.13,14 These interventions often address etiologies of LUTS for which medical therapies are not effective as the sole treatment option.
Medication Management
Medical management includes α-blockers, 5-α-reductase inhibitors (5-α-RIs), antimuscarinic or anticholinergic medicines, β-3 agonists, and phosphodiesterase inhibitors (Table). These medications work independently as well as synergistically. The use of medications to improve symptoms must be balanced against potential AEs and the consequences of a lifetime of drug usage, which can be additive.15,16
First-line pharmacological therapy for BPH is α-blockers, which work by blocking α1A receptors in the prostate and bladder neck, leading to smooth muscle relaxation, increased diameter of the channel, and improved urinary flow. α-receptors in the bladder neck and prostate are expressed with increased frequency with age and are a potential cause for worsening symptoms as men age. Studies demonstrate that these medications reduce symptoms by 30 to 40% and increase flow rates by 16 to 25%.17 Commonly prescribed α-blockers include tamsulosin, alfuzosin, silodosin, doxazosin, and terazosin. Doxazosin and terazosin require dose titrations because they may cause significant hypotension. Orthostatic hypotension typically improves with time and is avoided if the patient takes the medication at bedtime. Both doxazosin and terazosin are on the American Geriatric Society’s Beers Criteria list and should be avoided in older patients.18 Tamsulosin, alfuzosin, and silodosin have a standardized dosing regimen and lower rates of hypotension. Significant AEs include ejaculation dysfunction, nasal congestion, and orthostatic hypotension. Duan and colleagues have linked tamsulosin with dementia. However, this association is not causal and further studies are necessary.19,20 Patients who have taken these agents also are at risk for intraoperative floppy iris syndrome (IFIS). Permanent visual problems can arise if the intraoperative management is not managed to account for IFIS. These medications have a rapid onset of action and work immediately. However, to reach maximum benefit, patients must take the medication for several weeks. Unfortunately, up to one-third of patients will have no improvement with α-blocker therapy, and many patients will discontinue these medications because of significant AEs.6,21
5-α-RIs (finasteride and dutasteride) inhibit the conversion of testosterone to more potent dihydrotestosterone. They effectively reduce prostate volume by 25 to 30%.22 The results occur slowly and can take 6 to 12 months to reach the desired outcome. These medications are effective in men with larger prostates and not as effective in men with smaller prostates.23 These medications can improve urinary flow rates by about 10%, reduce IPSS scores by 20 to 30%, reduce the risk of urinary retention by 50%, and reduce the progression of BPH to the point where surgery is required by 50%.24 Furthermore, 5-α-RIs lower PSA by > 50% after 12 months of treatment.25
A baseline PSA should be established before administration and after 6 months of treatment. Any increase in the PSA even if the level is within normal limits should be evaluated for prostate cancer. Sarkar and colleagues recently published a study evaluating prostate cancer diagnosis in patients treated with 5-α-RI and found there was a delay in diagnosing prostate cancer in this population. Controversy also exists as to the potential of these medications increasing the risk for high-grade prostate cancer, which has led to a US Food and Drug Administration (FDA) warning. AEs include decreased libido (1.5%), ejaculatory dysfunction (3.4%), gynecomastia (1.3%), and/or ED (1.6%).26-28 A recent study evaluating 5-α-RIs demonstrated about a 2-fold increased risk of depression.29
There are well-established studies that note increased effectiveness when using combined α-blocker therapy with 5-α-RI medications. The Medical Therapy of Prostate Symptoms (MTOPS) and Combination Avodart and Tamsulosin (CombAT) trials showed that the combination of both medications was more effective in improving voiding symptoms and flow rates than either agent alone.15,16 Combination therapy resulted in a 66% reduction in disease progression, 81% reduction in urinary retention, and a 67% reduction in the need for surgery compared with placebo.
Anticholinergic medication use in BPH with LUTS is well established, and their use is often combined with other therapies. Anticholinergics work by inhibiting muscarinic M3 receptors to reduce detrusor muscle contraction. This effectively decreases bladder contractions and delays the desire to void. Kaplan and colleagues showed that tolterodine significantly improved a patient’s QOL when added to α-blocker therapy.30 Patients reported a positive outcome at 12 weeks, which resulted in a reduction in urgency incontinence, urgency, nocturia, and the overall number of voiding episodes within 24 hours.
β-3 agonists are a class of medications for OAB; mirabegron and vibegron have proven effective in reducing similar symptoms. In phase 3 clinical trials, mirabegron improved urinary incontinence episodes by 50% and reduced the number of voids in 24 hours.31 Mirabegron is well tolerated and avoids many common anticholinergic effects.32 Vibegron is the newest medication in the class and could soon become the preferred agent given it does not have cytochrome P450 interactions and does not cause hypertension like mirabegron.33
Anticholinergics should be used with caution in patients with a history of urinary retention, elevated after-void residual, or other medications with known anticholinergic effects. AEs include sedation, confusion, dry mouth, constipation, and potential falls in older patients.18 Recent studies have noted an association with dementia in the prolonged use of these medications in older patients and should be used cautiously.20
Phosphodiesterase-5 enzyme inhibitors (PDE-5) are adjunctive medications shown to improve LUTS. This class of medication is prescribed mostly for ED. However, tadalafil 5 mg taken daily also is FDA approved for the treatment of LUTS secondary to BPH given its prolonged half-life. The exact mechanism for improved BPH symptoms is unknown. Possibly the effects are due to an increase mediated by PDE-5 in cyclic guanosine monophosphate (cGMP), which increases smooth muscle relaxation and tissue perfusion of the prostate and bladder.34 There have been limited studies on objective improvement in uroflowmetry parameters compared with other treatments. The daily dosing of tadalafil should not be prescribed in men with a creatinine clearance < 30 mL/min.29 Tadalafil is not considered a first-line agent and is usually reserved for patients who experience ED in addition to BPH. When initiating BPH pharmacologic therapy, the PCP should be aware of adherence and high discontinuation rates.35
Surgical Treatments
Surgical treatments are often delayed out of fear of potential AEs or considered a last resort when symptoms are too severe.36 Early intervention is required to prevent irreversible deleterious changes to detrusor muscle structure and function (Figure). Patients fear urinary incontinence, ED or ejaculatory dysfunction, and anesthesia complications associated with surgical interventions.6,37 Multiple studies show that patients fare better with early surgical intervention, experiencing improved IPSS scores, urinary flow, and QOL. The following is an overview of the most popular procedures.
Prostatic urethral lift (PUL) using the UroLift System is an FDA-approved, minimally-invasive treatment of LUTS secondary to BPH. This procedure treats prostates < 80 g with an absent median lobe.6,21,38 Permanent implants are placed per the prostatic urethra to displace obstructing prostate tissue laterally. This opens the urethra directly without cutting, heating, or removing any prostate tissue. This procedure is minimally invasive, often done in the office as an outpatient procedure, and offers better symptom relief than medication with a lower risk profile than transurethral resection of the prostate (TURP).39,40 The LIFT study was a multicenter, randomized, blinded trial; patients were randomized 2:1 to undergo UroLift or a sham operation. At 3 years, average improvements were statistically significant for total IPSS reduction (41%), QOL improvement (49%), and improved maximum flow rates by (51%).41 Risk for urinary incontinence is low, and the procedure has been shown to preserve erectile and ejaculatory function. Furthermore, patients report significant improvement in their QOL without the need for medications. Surgical retreatment rates at 5 years are 13.6%, with an additional 10.7% of subjects back on medication therapy with α-blockers or 5-α-RIs.42
Water vapor thermal therapy or Rez¯um uses steam as thermal energy to destroy obstructing prostate tissue and relieve the obstruction.43 The procedure differs from older conductive heat thermotherapies because the steam penetrates prostate zonal anatomy without affecting areas outside the targeted treatment zone. The procedure is done in the office with local anesthesia and provides long-lasting relief of LUTS with minimal risks. Following the procedure, patients require an indwelling urethral catheter for 3 to 7 days, and most patients begin to experience symptom improvement 2 to 4 weeks following the procedure.44 The procedure received FDA approval in 2015. Four-year data show significant improvement in maximal flow rate (50%), IPSS (47%), and QOL (43%).45 Surgical retreatment rates were 4.4%. Criticisms of this treatment include patient discomfort with the office procedure, the requirement for an indwelling catheter for a short period, and lack of long-term outcomes data. Guidelines support use in prostate volumes > 80 g with or without median lobe anatomy.
TURP is the gold standard to which other treatments are compared.46 The surgery is performed in the operating room where urologists use a rigid cystoscope and resection element to effectively carve out and cauterize obstructing prostate tissue. Patients typically recover for a short period with an indwelling urethral catheter that is often removed 12 to 24 hours after surgery. New research points out that despite increasing mean age (55% of patients are aged > 70 years with associated comorbidities), the morbidity of TURP was < 1% and mortality rate of 0 to 0.3%.47 Postoperative complications include bleeding that requires a transfusion (3%), retrograde ejaculation (65%), and rare urinary incontinence (2%).47 Surgical retreatment rates for patients following a TURP are approximately 13 to 15% at 8 years.34
Laser surgery for BPH includes multiple techniques: photovaporization of the prostate using a Greenlight XPS laser, holmium laser ablation, and holmium laser enucleation (HoLEP). Proponents of these treatments cite lower bleeding risks compared with TURP, but the operation is largely surgeon dependent on the technology chosen. Most studies comparing these technologies with TURP show similar outcomes of IPSS reports, quality of life improvements, and complications.
Patients with extremely large prostates, > 100 g or 4 times the normal size, pose a unique challenge to surgical treatment. Historically, patients were treated with an open simple prostatectomy operation or staged TURP procedures. Today, urologists use newer, safer ways to treat these patients. Both HoLEP and robot-assisted simple prostatectomy work well in relieving urinary symptoms with lower complications compared with older open surgery. Other minimally invasive procedures, such as prostatic artery embolism, have been described for the treatment of BPH specifically in men who may be unfit for surgery.48Future treatments are constantly evolving. Many unanswered questions remain about BPH and the role of inflammation, metabolic dysfunction, obesity, and other genetic factors driving BPH and prostate growth. Pharmaceutical opportunities exist in mechanisms aimed to reduce prostate growth, induce cellular apoptosis, as well as other drugs to reduce bladder symptoms. Newer, minimally invasive therapies also will become more readily available, such as Aquablation, which is the first FDA-granted surgical robot for the autonomous removal of prostatic tissue due to BPH.49 However, the goal of all future therapies should include the balance of alleviating disruptive symptoms while demonstrating a favorable risk profile. Many men discontinue taking medications, yet few present for surgery. Most concerning is the significant population of men who will develop irreversible bladder dysfunction while waiting for the perfect treatment. There are many opportunities for an effective treatment that is less invasive than surgery, provides durable relief, has minimal AEs, and is affordable.
Conclusions
There is no perfect treatment for patients with LUTS. All interventions have potential AEs and associated complications. Medications are often started as first-line therapy but are often discontinued at the onset of significant AEs. This process is often repeated. Many patients will try different medications without any significant improvement in their symptoms or short-term relief, which results in the gradual progression of the disease.
The PCP plays a significant role in the initial evaluation and management of BPH. These frontline clinicians can recognize patients who may already be experiencing sequela of prolonged bladder outlet obstruction and refer these men to urologists promptly. Counseling patients about their treatment options is an important duty for all PCPs.
A clear understanding of the available treatment options will help PCPs counsel patients appropriately about lifestyle modification, medications, and surgical treatment options for their symptoms. The treatment of this disorder is a rapidly evolving topic with the constant introduction of new technologies and medications, which are certain to continue to play an important role for PCPs and urologists.
Lower urinary tract symptoms (LUTS)are common and tend to increase in frequency with age. Managing LUTS can be complicated, requires an informed discussion between the primary care practitioner (PCP) and patient, and is best achieved by a thorough understanding of the many medical and surgical options available. Over the past 3 decades, medications have become the most common therapy; but recently, newer minimally invasive surgeries have challenged this paradigm. This article provides a comprehensive review for PCPs regarding the evaluation and management of LUTS in men and when to consider a urology referral.
Benign prostatic hyperplasia (BPH) and LUTS are common clinical encounters for most PCPs. About 50% of men will develop LUTS associated with BPH, and symptoms associated with these conditions increase as men age.1,2 Studies have estimated that 90% of men aged 45 to 80 years demonstrate some symptoms of LUTS.3 Strong genetic influence seems to suggest heritability, but BPH also occurs in sporadic forms and is heavily influenced by androgens.4
BPH is a histologic diagnosis, whereas LUTS consists of complex symptomatology related to both static or dynamic components.1 The enlarged prostate gland obstructs the urethra, simultaneously causing an increase in muscle tone and resistance at the bladder neck and prostatic urethra, leading to increased resistance to urine flow. As a result, there is a thickening of the detrusor muscles in the bladder wall and an overall decreased compliance. Urine becomes stored under increased pressure. These changes result in a weak or intermittent urine stream, incomplete emptying of the bladder, postvoid dribble, hesitancy, and irritative symptoms, such as urgency, frequency, and nocturia.
For many patients, BPH associated with LUTS is a quality of life (QOL) issue. The stigma associated with these symptoms often leads to delays in patients seeking care. Many patients do not seek treatment until symptoms have become so severe that changes in bladder health are often irreversible. Early intervention can dramatically improve a patient’s QOL. Also, early intervention has the potential to reduce overall health care expenditures. BPH-related spending exceeds $1 billion each year in the Medicare program alone.5
PCPs are in a unique position to help many patients who present with early-stage LUTS. Given the substantial impact this disease has on QOL, early recognition of symptoms and prompt treatment play a major role. Paramount to this effort is awareness and understanding of various treatments, their advantages, and adverse effects (AEs). This article highlights evidence-based evaluation and treatment of BPH/LUTS for PCPs who treat veterans and recommendations as to when to refer a patient to a urologist.
Evaluation of LUTS and BPH
Evaluation begins with a thorough medical history and physical examination. Particular attention should focus on ruling out other causes of LUTS, such as a urinary tract infection (UTI), acute prostatitis, malignancy, bladder dysfunction, neurogenic bladder, and other obstructive pathology, such as urethral stricture disease. The differential diagnosis of LUTS includes BPH, UTI, bladder neck obstruction, urethral stricture, bladder stones, polydipsia, overactive bladder (OAB), nocturnal polyuria, neurologic disease, genitourinary malignancy, renal failure, and acute/chronic urinary retention.6
Relevant medical history influencing urinary symptoms includes diabetes mellitus, underlying neurologic diseases, previous trauma, sexually transmitted infections, and certain medications. Symptom severity may be obtained using a validated questionnaire, such as the International Prostate Symptom Score (IPSS), which also aids clinicians in assessing the impact of LUTS on QOL. Additionally, urinary frequency or volume records (voiding diary) may help establish the severity of the patient’s symptoms and provide insight into other potential causes for LUTS. Patients with BPH often have concurrent erectile dysfunction (ED) or other sexual dysfunction symptoms. Patients should be evaluated for baseline sexual dysfunction before the initiation of treatment as many therapies worsen symptoms of ED or ejaculatory dysfunction.
A comprehensive physical examination with a focus on the genitourinary system should, at minimum, assess for abnormalities of the urethral meatus, prepuce, penis, groin nodes, and prior surgical scars. A digital rectal examination also should be performed. Although controversial, a digital rectal examination for prostate cancer screening may provide a rough estimate of prostate size, help rule out prostatitis, and detect incident prostate nodules. Prostate size does not necessarily correlate well with the degree of urinary obstruction or LUTS but is an important consideration when deciding among different therapies.1
Laboratory and Adjunctive Tests
A urinalysis with microscopy helps identify other potential causes for urinary symptoms, including infection, proteinuria, or glucosuria. In patients who present with gross or microscopic hematuria, additional consideration should be given to bladder calculi and genitourinary cancer.2 When a reversible source for the hematuria is not identified, these patients require referral to a urologist for a hematuria evaluation.
There is some controversy regarding prostate specific antigen (PSA) testing. Most professional organizations advocate for a shared decision-making approach before testing. The American Cancer Society recommends this informed discussion occur between the patient and the PCP for men aged > 50 years at average risk, men aged > 45 years at high risk of developing prostate cancer (African Americans or first-degree relative with early prostate cancer diagnosis), and aged 40 years for men with more than one first-degree relative with an early prostate cancer diagnosis.7
Adjunctive tests include postvoid residual (PVR), cystoscopy, uroflowmetry, urodynamics, and transrectal ultrasound. However, these are mostly performed by urologists. In some patients with bladder decompensation after prolonged partial bladder outlet obstruction, urodynamics may be used by urologists to determine whether a patient may benefit from an outlet obstruction procedure. Ordering additional imaging or serum studies for the assessment of LUTS is rarely helpful.
Treatment
Treatment includes management with or without lifestyle modification, medication administration, and surgical therapy. New to this paradigm are in-office minimally invasive surgical options. The goal of treatment is not only to reduce patient symptoms and improve QOL, but also to prevent the secondary sequala of urinary retention, bladder failure, and eventual renal impairment.7A basic understanding of these treatments can aid PCPs with appropriate patient counseling and urologic referral.8
Lifestyle and Behavior Modification
Behavior modification is the starting point for all patients with LUTS. Lifestyle modifications for LUTS include avoiding substances that exacerbate symptoms, such as α-agonists (decongestants), caffeine, alcohol, spicy/acidic foods, chocolate, and soda. These substances are known to be bladder irritants. Common medications contributing to LUTS include antidepressants, decongestants, antihistamines, bronchodilators, anticholinergics, and sympathomimetics. To decrease nocturia, behavioral modifications include limiting evening fluid intake, timed diuretic administration for patients already on a diuretic, and elevating legs 1 hour before bedtime. Counseling obese patients to lose weight and increasing physical activity have been linked to reduced LUTS.9 Other behavioral techniques include double voiding: a technique where patients void normally then change positions and return to void to empty the bladder. Another technique is timed voiding: Many patients have impaired sensation when the bladder is full. These patients are encouraged to void at regular intervals.
Complementary and Alternative Medicine
Multiple nutraceutical compounds claim improved urinary health and symptom reduction. These compounds are marketed to patients with little regulation and oversight since supplements are not regulated or held to the same standard as prescription medications. The most popular nutraceutical for prostate health and LUTS is saw palmetto. Despite its common usage for the treatment of LUTS, little data support saw palmetto health claims. In 2012, a systematic review of 32 randomized trials including 5666 patients compared saw palmetto with a placebo. The study found no difference in urinary symptom scores, urinary flow, or prostate size.10,11 Other phytotherapy compounds often considered for urinary symptoms include stinging nettle extract and β-sitosterol compounds. The mechanism of action of these agents is unknown and efficacy data are lacking.
Historically, acupuncture and pelvic floor physical therapy have been used successfully for OAB symptoms. A meta-analysis found positive beneficial effects of acupuncture compared with a sham control for short- and medium-term follow-up in both IPSS and urine flow rates in some studies; however, when combining the studies for more statistical power, the benefits were less clear.12 Physical therapists with specialized training and certification in pelvic health can incorporate certain bladder training techniques. These include voiding positional changes (double voiding and postvoid urethral milking) and timed voiding.13,14 These interventions often address etiologies of LUTS for which medical therapies are not effective as the sole treatment option.
Medication Management
Medical management includes α-blockers, 5-α-reductase inhibitors (5-α-RIs), antimuscarinic or anticholinergic medicines, β-3 agonists, and phosphodiesterase inhibitors (Table). These medications work independently as well as synergistically. The use of medications to improve symptoms must be balanced against potential AEs and the consequences of a lifetime of drug usage, which can be additive.15,16
First-line pharmacological therapy for BPH is α-blockers, which work by blocking α1A receptors in the prostate and bladder neck, leading to smooth muscle relaxation, increased diameter of the channel, and improved urinary flow. α-receptors in the bladder neck and prostate are expressed with increased frequency with age and are a potential cause for worsening symptoms as men age. Studies demonstrate that these medications reduce symptoms by 30 to 40% and increase flow rates by 16 to 25%.17 Commonly prescribed α-blockers include tamsulosin, alfuzosin, silodosin, doxazosin, and terazosin. Doxazosin and terazosin require dose titrations because they may cause significant hypotension. Orthostatic hypotension typically improves with time and is avoided if the patient takes the medication at bedtime. Both doxazosin and terazosin are on the American Geriatric Society’s Beers Criteria list and should be avoided in older patients.18 Tamsulosin, alfuzosin, and silodosin have a standardized dosing regimen and lower rates of hypotension. Significant AEs include ejaculation dysfunction, nasal congestion, and orthostatic hypotension. Duan and colleagues have linked tamsulosin with dementia. However, this association is not causal and further studies are necessary.19,20 Patients who have taken these agents also are at risk for intraoperative floppy iris syndrome (IFIS). Permanent visual problems can arise if the intraoperative management is not managed to account for IFIS. These medications have a rapid onset of action and work immediately. However, to reach maximum benefit, patients must take the medication for several weeks. Unfortunately, up to one-third of patients will have no improvement with α-blocker therapy, and many patients will discontinue these medications because of significant AEs.6,21
5-α-RIs (finasteride and dutasteride) inhibit the conversion of testosterone to more potent dihydrotestosterone. They effectively reduce prostate volume by 25 to 30%.22 The results occur slowly and can take 6 to 12 months to reach the desired outcome. These medications are effective in men with larger prostates and not as effective in men with smaller prostates.23 These medications can improve urinary flow rates by about 10%, reduce IPSS scores by 20 to 30%, reduce the risk of urinary retention by 50%, and reduce the progression of BPH to the point where surgery is required by 50%.24 Furthermore, 5-α-RIs lower PSA by > 50% after 12 months of treatment.25
A baseline PSA should be established before administration and after 6 months of treatment. Any increase in the PSA even if the level is within normal limits should be evaluated for prostate cancer. Sarkar and colleagues recently published a study evaluating prostate cancer diagnosis in patients treated with 5-α-RI and found there was a delay in diagnosing prostate cancer in this population. Controversy also exists as to the potential of these medications increasing the risk for high-grade prostate cancer, which has led to a US Food and Drug Administration (FDA) warning. AEs include decreased libido (1.5%), ejaculatory dysfunction (3.4%), gynecomastia (1.3%), and/or ED (1.6%).26-28 A recent study evaluating 5-α-RIs demonstrated about a 2-fold increased risk of depression.29
There are well-established studies that note increased effectiveness when using combined α-blocker therapy with 5-α-RI medications. The Medical Therapy of Prostate Symptoms (MTOPS) and Combination Avodart and Tamsulosin (CombAT) trials showed that the combination of both medications was more effective in improving voiding symptoms and flow rates than either agent alone.15,16 Combination therapy resulted in a 66% reduction in disease progression, 81% reduction in urinary retention, and a 67% reduction in the need for surgery compared with placebo.
Anticholinergic medication use in BPH with LUTS is well established, and their use is often combined with other therapies. Anticholinergics work by inhibiting muscarinic M3 receptors to reduce detrusor muscle contraction. This effectively decreases bladder contractions and delays the desire to void. Kaplan and colleagues showed that tolterodine significantly improved a patient’s QOL when added to α-blocker therapy.30 Patients reported a positive outcome at 12 weeks, which resulted in a reduction in urgency incontinence, urgency, nocturia, and the overall number of voiding episodes within 24 hours.
β-3 agonists are a class of medications for OAB; mirabegron and vibegron have proven effective in reducing similar symptoms. In phase 3 clinical trials, mirabegron improved urinary incontinence episodes by 50% and reduced the number of voids in 24 hours.31 Mirabegron is well tolerated and avoids many common anticholinergic effects.32 Vibegron is the newest medication in the class and could soon become the preferred agent given it does not have cytochrome P450 interactions and does not cause hypertension like mirabegron.33
Anticholinergics should be used with caution in patients with a history of urinary retention, elevated after-void residual, or other medications with known anticholinergic effects. AEs include sedation, confusion, dry mouth, constipation, and potential falls in older patients.18 Recent studies have noted an association with dementia in the prolonged use of these medications in older patients and should be used cautiously.20
Phosphodiesterase-5 enzyme inhibitors (PDE-5) are adjunctive medications shown to improve LUTS. This class of medication is prescribed mostly for ED. However, tadalafil 5 mg taken daily also is FDA approved for the treatment of LUTS secondary to BPH given its prolonged half-life. The exact mechanism for improved BPH symptoms is unknown. Possibly the effects are due to an increase mediated by PDE-5 in cyclic guanosine monophosphate (cGMP), which increases smooth muscle relaxation and tissue perfusion of the prostate and bladder.34 There have been limited studies on objective improvement in uroflowmetry parameters compared with other treatments. The daily dosing of tadalafil should not be prescribed in men with a creatinine clearance < 30 mL/min.29 Tadalafil is not considered a first-line agent and is usually reserved for patients who experience ED in addition to BPH. When initiating BPH pharmacologic therapy, the PCP should be aware of adherence and high discontinuation rates.35
Surgical Treatments
Surgical treatments are often delayed out of fear of potential AEs or considered a last resort when symptoms are too severe.36 Early intervention is required to prevent irreversible deleterious changes to detrusor muscle structure and function (Figure). Patients fear urinary incontinence, ED or ejaculatory dysfunction, and anesthesia complications associated with surgical interventions.6,37 Multiple studies show that patients fare better with early surgical intervention, experiencing improved IPSS scores, urinary flow, and QOL. The following is an overview of the most popular procedures.
Prostatic urethral lift (PUL) using the UroLift System is an FDA-approved, minimally-invasive treatment of LUTS secondary to BPH. This procedure treats prostates < 80 g with an absent median lobe.6,21,38 Permanent implants are placed per the prostatic urethra to displace obstructing prostate tissue laterally. This opens the urethra directly without cutting, heating, or removing any prostate tissue. This procedure is minimally invasive, often done in the office as an outpatient procedure, and offers better symptom relief than medication with a lower risk profile than transurethral resection of the prostate (TURP).39,40 The LIFT study was a multicenter, randomized, blinded trial; patients were randomized 2:1 to undergo UroLift or a sham operation. At 3 years, average improvements were statistically significant for total IPSS reduction (41%), QOL improvement (49%), and improved maximum flow rates by (51%).41 Risk for urinary incontinence is low, and the procedure has been shown to preserve erectile and ejaculatory function. Furthermore, patients report significant improvement in their QOL without the need for medications. Surgical retreatment rates at 5 years are 13.6%, with an additional 10.7% of subjects back on medication therapy with α-blockers or 5-α-RIs.42
Water vapor thermal therapy or Rez¯um uses steam as thermal energy to destroy obstructing prostate tissue and relieve the obstruction.43 The procedure differs from older conductive heat thermotherapies because the steam penetrates prostate zonal anatomy without affecting areas outside the targeted treatment zone. The procedure is done in the office with local anesthesia and provides long-lasting relief of LUTS with minimal risks. Following the procedure, patients require an indwelling urethral catheter for 3 to 7 days, and most patients begin to experience symptom improvement 2 to 4 weeks following the procedure.44 The procedure received FDA approval in 2015. Four-year data show significant improvement in maximal flow rate (50%), IPSS (47%), and QOL (43%).45 Surgical retreatment rates were 4.4%. Criticisms of this treatment include patient discomfort with the office procedure, the requirement for an indwelling catheter for a short period, and lack of long-term outcomes data. Guidelines support use in prostate volumes > 80 g with or without median lobe anatomy.
TURP is the gold standard to which other treatments are compared.46 The surgery is performed in the operating room where urologists use a rigid cystoscope and resection element to effectively carve out and cauterize obstructing prostate tissue. Patients typically recover for a short period with an indwelling urethral catheter that is often removed 12 to 24 hours after surgery. New research points out that despite increasing mean age (55% of patients are aged > 70 years with associated comorbidities), the morbidity of TURP was < 1% and mortality rate of 0 to 0.3%.47 Postoperative complications include bleeding that requires a transfusion (3%), retrograde ejaculation (65%), and rare urinary incontinence (2%).47 Surgical retreatment rates for patients following a TURP are approximately 13 to 15% at 8 years.34
Laser surgery for BPH includes multiple techniques: photovaporization of the prostate using a Greenlight XPS laser, holmium laser ablation, and holmium laser enucleation (HoLEP). Proponents of these treatments cite lower bleeding risks compared with TURP, but the operation is largely surgeon dependent on the technology chosen. Most studies comparing these technologies with TURP show similar outcomes of IPSS reports, quality of life improvements, and complications.
Patients with extremely large prostates, > 100 g or 4 times the normal size, pose a unique challenge to surgical treatment. Historically, patients were treated with an open simple prostatectomy operation or staged TURP procedures. Today, urologists use newer, safer ways to treat these patients. Both HoLEP and robot-assisted simple prostatectomy work well in relieving urinary symptoms with lower complications compared with older open surgery. Other minimally invasive procedures, such as prostatic artery embolism, have been described for the treatment of BPH specifically in men who may be unfit for surgery.48Future treatments are constantly evolving. Many unanswered questions remain about BPH and the role of inflammation, metabolic dysfunction, obesity, and other genetic factors driving BPH and prostate growth. Pharmaceutical opportunities exist in mechanisms aimed to reduce prostate growth, induce cellular apoptosis, as well as other drugs to reduce bladder symptoms. Newer, minimally invasive therapies also will become more readily available, such as Aquablation, which is the first FDA-granted surgical robot for the autonomous removal of prostatic tissue due to BPH.49 However, the goal of all future therapies should include the balance of alleviating disruptive symptoms while demonstrating a favorable risk profile. Many men discontinue taking medications, yet few present for surgery. Most concerning is the significant population of men who will develop irreversible bladder dysfunction while waiting for the perfect treatment. There are many opportunities for an effective treatment that is less invasive than surgery, provides durable relief, has minimal AEs, and is affordable.
Conclusions
There is no perfect treatment for patients with LUTS. All interventions have potential AEs and associated complications. Medications are often started as first-line therapy but are often discontinued at the onset of significant AEs. This process is often repeated. Many patients will try different medications without any significant improvement in their symptoms or short-term relief, which results in the gradual progression of the disease.
The PCP plays a significant role in the initial evaluation and management of BPH. These frontline clinicians can recognize patients who may already be experiencing sequela of prolonged bladder outlet obstruction and refer these men to urologists promptly. Counseling patients about their treatment options is an important duty for all PCPs.
A clear understanding of the available treatment options will help PCPs counsel patients appropriately about lifestyle modification, medications, and surgical treatment options for their symptoms. The treatment of this disorder is a rapidly evolving topic with the constant introduction of new technologies and medications, which are certain to continue to play an important role for PCPs and urologists.
1. Roehrborn CG. Benign prostatic hyperplasia: an overview. Rev Urol. 2005;7 Suppl 9(Suppl 9):S3-S14
2. McVary KT. Clinical manifestations and diagnostic evaluation of benign prostatic hyperplasia. UpToDate. Updated November 18, 2021. Accessed November 23, 2021. https:// www.uptodate.com/contents/clinical-manifestations-and -diagnostic-evaluation-of-benign-prostatic-hyperplasia
3. McVary KT. BPH: epidemiology and comorbidities. Am J Manag Care. 2006;12(5 Suppl):S122-S128.
4. Ho CK, Habib FK. Estrogen and androgen signaling in the pathogenesis of BPH. Nat Rev Urol. 2011;8(1):29-41. doi:10.1038/nrurol.2010.207
5. Rensing AJ, Kuxhausen A, Vetter J, Strope SA. Differences in the treatment of benign prostatic hyperplasia: comparing the primary care physician and the urologist. Urol Pract. 2017;4(3):193-199. doi:10.1016/j.urpr.2016.07.002
6. Foster HE, Barry MJ, Dahm P, et al. Surgical management of lower urinary tract symptoms attributed to benign prostatic hyperplasia: AUA guideline. J Urol. 2018;200(3):612- 619. doi:10.1016/j.juro.2018.05.048
7. Landau A, Welliver C. Analyzing and characterizing why men seek care for lower urinary tract symptoms. Curr Urol Rep. 2020;21(12):58. Published 2020 Oct 30. doi:10.1007/s11934-020-01006-w
8. Das AK, Leong JY, Roehrborn CG. Office-based therapies for benign prostatic hyperplasia: a review and update. Can J Urol. 2019;26(4 Suppl 1):2-7.
9. Parsons JK, Sarma AV, McVary K, Wei JT. Obesity and benign prostatic hyperplasia: clinical connections, emerging etiological paradigms and future directions. J Urol. 2013;189(1 Suppl):S102-S106. doi:10.1016/j.juro.2012.11.029
10. Pattanaik S, Mavuduru RS, Panda A, et al. Phosphodiesterase inhibitors for lower urinary tract symptoms consistent with benign prostatic hyperplasia. Cochrane Database Syst Rev. 2018;11(11):CD010060. Published 2018 Nov 16. doi:10.1002/14651858.CD010060.pub2
11. McVary KT. Medical treatment of benign prostatic hyperplasia. UpToDate. Updated October 4, 2021. Accessed November 23, 2021. https://www.uptodate.com/contents /medical-treatment-of-benign-prostatic-hyperplasia
12. Zhang W, Ma L, Bauer BA, Liu Z, Lu Y. Acupuncture for benign prostatic hyperplasia: A systematic review and metaanalysis. PLoS One. 2017;12(4):e0174586. Published 2017 Apr 4. doi:10.1371/journal.pone.0174586
13. Newman DK, Guzzo T, Lee D, Jayadevappa R. An evidence- based strategy for the conservative management of the male patient with incontinence. Curr Opin Urol. 2014;24(6):553-559. doi:10.1097/MOU.0000000000000115
14. Newman DK, Wein AJ. Office-based behavioral therapy for management of incontinence and other pelvic disorders. Urol Clin North Am. 2013;40(4):613-635. doi:10.1016/j.ucl.2013.07.010
15. McConnell JD, Roehrborn CG, Bautista OM, et al. The long-term effect of doxazosin, finasteride, and combination therapy on the clinical progression of benign prostatic hyperplasia. N Engl J Med. 2003;349(25):2387-2398. doi:10.1056/NEJMoa030656
16. Roehrborn CG, Barkin J, Siami P, et al. Clinical outcomes after combined therapy with dutasteride plus tamsulosin or either monotherapy in men with benign prostatic hyperplasia (BPH) by baseline characteristics: 4-year results from the randomized, double-blind Combination of Avodart and Tamsulosin (CombAT) trial. BJU Int. 2011;107(6):946-954. doi:10.1111/j.1464-410X.2011.10124.x
17. Djavan B, Marberger M. A meta-analysis on the efficacy and tolerability of alpha1-adrenoceptor antagonists in patients with lower urinary tract symptoms suggestive of benign prostatic obstruction. Eur Urol. 1999;36(1):1-13. doi:10.1159/000019919
18. By the American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American Geriatrics Society 2015 Updated Beers Criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2015;63(11):2227-2246. doi:10.1111/jgs.13702
19. Duan Y, Grady JJ, Albertsen PC, Helen Wu Z. Tamsulosin and the risk of dementia in older men with benign prostatic hyperplasia. Pharmacoepidemiol Drug Saf. 2018;27(3):340- 348. doi:10.1002/pds.4361
20. Coupland CAC, Hill T, Dening T, Morriss R, Moore M, Hippisley-Cox J. Anticholinergic drug exposure and the risk of dementia: a nested case-control study. JAMA Intern Med. 2019;179(8):1084-1093. doi:10.1001/jamainternmed.2019.0677
21. Parsons JK, Dahm P, Köhler TS, Lerner LB, Wilt TJ. Surgical management of lower urinary tract symptoms attributed to benign prostatic hyperplasia: AUA guideline amendment 2020. J Urol. 2020;204(4):799-804. doi:10.1097/JU.0000000000001298
22. Smith AB, Carson CC. Finasteride in the treatment of patients with benign prostatic hyperplasia: a review. Ther Clin Risk Manag. 2009;5(3):535-545. doi:10.2147/tcrm.s6195
23. Andriole GL, Guess HA, Epstein JI, et al. Treatment with finasteride preserves usefulness of prostate-specific antigen in the detection of prostate cancer: results of a randomized, double-blind, placebo-controlled clinical trial. PLESS Study Group. Proscar Long-term Efficacy and Safety Study. Urology. 1998;52(2):195-202. doi:10.1016/s0090-4295(98)00184-8
24. McConnell JD, Bruskewitz R, Walsh P, et al. The effect of finasteride on the risk of acute urinary retention and the need for surgical treatment among men with benign prostatic hyperplasia. Finasteride Long-Term Efficacy and Safety Study Group. N Engl J Med. 1998;338(9):557-563. doi:10.1056/NEJM199802263380901
25. Rittmaster RS. 5alpha-reductase inhibitors in benign prostatic hyperplasia and prostate cancer risk reduction. Best Pract Res Clin Endocrinol Metab. 2008;22(2):389-402. doi:10.1016/j.beem.2008.01.016
26. La Torre A, Giupponi G, Duffy D, Conca A, Cai T, Scardigli A. Sexual dysfunction related to drugs: a critical review. Part V: α-blocker and 5-ARI drugs. Pharmacopsychiatry. 2016;49(1):3-13. doi:10.1055/s-0035-1565100
27. Corona G, Tirabassi G, Santi D, et al. Sexual dysfunction in subjects treated with inhibitors of 5α-reductase for benign prostatic hyperplasia: a comprehensive review and meta-analysis. Andrology. 2017;5(4):671-678. doi:10.1111/andr.12353
28. Trost L, Saitz TR, Hellstrom WJ. Side effects of 5-alpha reductase inhibitors: a comprehensive review. Sex Med Rev. 2013;1(1):24-41. doi:10.1002/smrj.3
29. Welk B, McArthur E, Ordon M, Anderson KK, Hayward J, Dixon S. Association of suicidality and depression with 5α-reductase inhibitors. JAMA Intern Med. 2017;177(5):683-691. doi:10.1001/jamainternmed.2017.0089
30. Kaplan SA, Roehrborn CG, Rovner ES, Carlsson M, Bavendam T, Guan Z. Tolterodine and tamsulosin for treatment of men with lower urinary tract symptoms and overactive bladder: a randomized controlled trial [published correction appears in JAMA. 2007 Mar 21:297(11):1195] [published correction appears in JAMA. 2007 Oct 24;298(16):1864]. JAMA. 2006;296(19):2319-2328. doi:10.1001/jama.296.19.2319
31. Nitti VW, Auerbach S, Martin N, Calhoun A, Lee M, Herschorn S. Results of a randomized phase III trial of mirabegron in patients with overactive bladder. J Urol. 2013;189(4):1388-1395. doi:10.1016/j.juro.2012.10.017
32. Chapple CR, Cardozo L, Nitti VW, Siddiqui E, Michel MC. Mirabegron in overactive bladder: a review of efficacy, safety, and tolerability. Neurourol Urodyn. 2014;33(1):17-30. doi:10.1002/nau.22505
33. Rutman MP, King JR, Bennett N, Ankrom W, Mudd PN. PD14-01 once-daily vibegron, a novel oral β3 agonist does not inhibit CYP2D6, a common pathway for drug metabolism in patients on OAB medications. J Urol. 2019;201(Suppl 4):e231. doi:10.1097/01.JU.0000555478.73162.19
34. Bo K, Frawley HC, Haylen BT, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for the conservative and nonpharmacological management of female pelvic floor dysfunction. Neurourol Urodyn. 2017;36(2):221- 244. doi:10.1002/nau.23107
35. Cindolo L, Pirozzi L, Fanizza C, et al. Drug adherence and clinical outcomes for patients under pharmacological therapy for lower urinary tract symptoms related to benign prostatic hyperplasia: population-based cohort study. Eur Urol. 2015;68(3):418-425. doi:10.1016/j.eururo.2014.11.006
36. Ruhaiyem ME, Alshehri AA, Saade M, Shoabi TA, Zahoor H, Tawfeeq NA. Fear of going under general anesthesia: a cross-sectional study. Saudi J Anaesth. 2016;10(3):317- 321. doi:10.4103/1658-354X.179094
37. Hashim MJ. Patient-centered communication: basic skills. Am Fam Physician. 2017;95(1):29-34.
38. Roehrborn CG, Barkin J, Gange SN, et al. Five year results of the prospective randomized controlled prostatic urethral L.I.F.T. study. Can J Urol. 2017;24(3):8802-8813.
39. Gratzke C, Barber N, Speakman MJ, et al. Prostatic urethral lift vs transurethral resection of the prostate: 2-year results of the BPH6 prospective, multicentre, randomized study. BJU Int. 2017;119(5):767-775.doi:10.1111/bju.13714
40. Sønksen J, Barber NJ, Speakman MJ, et al. Prospective, randomized, multinational study of prostatic urethral lift versus transurethral resection of the prostate: 12-month results from the BPH6 study. Eur Urol. 2015;68(4):643-652. doi:10.1016/j.eururo.2015.04.024
41. Roehrborn CG, Gange SN, Shore ND, et al. The prostatic urethral lift for the treatment of lower urinary tract symptoms associated with prostate enlargement due to benign prostatic hyperplasia: the L.I.F.T. Study. J Urol. 2013;190(6):2161-2167. doi:10.1016/j.juro.2013.05.116
42. McNicholas TA. Benign prostatic hyperplasia and new treatment options - a critical appraisal of the UroLift system. Med Devices (Auckl). 2016;9:115-123. Published 2016 May 19. doi:10.2147/MDER.S60780
43. McVary KT, Rogers T, Roehrborn CG. Rezuˉm Water Vapor thermal therapy for lower urinary tract symptoms associated with benign prostatic hyperplasia: 4-year results from randomized controlled study. Urology. 2019;126:171-179. doi:10.1016/j.urology.2018.12.041
44. Bole R, Gopalakrishna A, Kuang R, et al. Comparative postoperative outcomes of Rezˉum prostate ablation in patients with large versus small glands. J Endourol. 2020;34(7):778-781. doi:10.1089/end.2020.0177
45. Darson MF, Alexander EE, Schiffman ZJ, et al. Procedural techniques and multicenter postmarket experience using minimally invasive convective radiofrequency thermal therapy with Rezˉum system for treatment of lower urinary tract symptoms due to benign prostatic hyperplasia. Res Rep Urol. 2017;9:159-168. Published 2017 Aug 21. doi:10.2147/RRU.S143679
46. Baazeem A, Elhilali MM. Surgical management of benign prostatic hyperplasia: current evidence. Nat Clin Pract Urol. 2008;5(10):540-549. doi:10.1038/ncpuro1214
47. Rassweiler J, Teber D, Kuntz R, Hofmann R. Complications of transurethral resection of the prostate (TURP)- -incidence, management, and prevention. Eur Urol. 2006;50(5):969-980. doi:10.1016/j.eururo.2005.12.042
48. Abt D, Schmid HP, Speakman MJ. Reasons to consider prostatic artery embolization. World J Urol. 2021;39(7):2301-2306. doi:10.1007/s00345-021-03601-z
49. Nguyen DD, Barber N, Bidair M, et al. Waterjet Ablation Therapy for Endoscopic Resection of prostate tissue trial (WATER) vs WATER II: comparing Aquablation therapy for benign prostatic hyperplasia in30-80and80-150mLprostates. BJUInt. 2020;125(1):112-122. doi:10.1111/bju.14917.
1. Roehrborn CG. Benign prostatic hyperplasia: an overview. Rev Urol. 2005;7 Suppl 9(Suppl 9):S3-S14
2. McVary KT. Clinical manifestations and diagnostic evaluation of benign prostatic hyperplasia. UpToDate. Updated November 18, 2021. Accessed November 23, 2021. https:// www.uptodate.com/contents/clinical-manifestations-and -diagnostic-evaluation-of-benign-prostatic-hyperplasia
3. McVary KT. BPH: epidemiology and comorbidities. Am J Manag Care. 2006;12(5 Suppl):S122-S128.
4. Ho CK, Habib FK. Estrogen and androgen signaling in the pathogenesis of BPH. Nat Rev Urol. 2011;8(1):29-41. doi:10.1038/nrurol.2010.207
5. Rensing AJ, Kuxhausen A, Vetter J, Strope SA. Differences in the treatment of benign prostatic hyperplasia: comparing the primary care physician and the urologist. Urol Pract. 2017;4(3):193-199. doi:10.1016/j.urpr.2016.07.002
6. Foster HE, Barry MJ, Dahm P, et al. Surgical management of lower urinary tract symptoms attributed to benign prostatic hyperplasia: AUA guideline. J Urol. 2018;200(3):612- 619. doi:10.1016/j.juro.2018.05.048
7. Landau A, Welliver C. Analyzing and characterizing why men seek care for lower urinary tract symptoms. Curr Urol Rep. 2020;21(12):58. Published 2020 Oct 30. doi:10.1007/s11934-020-01006-w
8. Das AK, Leong JY, Roehrborn CG. Office-based therapies for benign prostatic hyperplasia: a review and update. Can J Urol. 2019;26(4 Suppl 1):2-7.
9. Parsons JK, Sarma AV, McVary K, Wei JT. Obesity and benign prostatic hyperplasia: clinical connections, emerging etiological paradigms and future directions. J Urol. 2013;189(1 Suppl):S102-S106. doi:10.1016/j.juro.2012.11.029
10. Pattanaik S, Mavuduru RS, Panda A, et al. Phosphodiesterase inhibitors for lower urinary tract symptoms consistent with benign prostatic hyperplasia. Cochrane Database Syst Rev. 2018;11(11):CD010060. Published 2018 Nov 16. doi:10.1002/14651858.CD010060.pub2
11. McVary KT. Medical treatment of benign prostatic hyperplasia. UpToDate. Updated October 4, 2021. Accessed November 23, 2021. https://www.uptodate.com/contents /medical-treatment-of-benign-prostatic-hyperplasia
12. Zhang W, Ma L, Bauer BA, Liu Z, Lu Y. Acupuncture for benign prostatic hyperplasia: A systematic review and metaanalysis. PLoS One. 2017;12(4):e0174586. Published 2017 Apr 4. doi:10.1371/journal.pone.0174586
13. Newman DK, Guzzo T, Lee D, Jayadevappa R. An evidence- based strategy for the conservative management of the male patient with incontinence. Curr Opin Urol. 2014;24(6):553-559. doi:10.1097/MOU.0000000000000115
14. Newman DK, Wein AJ. Office-based behavioral therapy for management of incontinence and other pelvic disorders. Urol Clin North Am. 2013;40(4):613-635. doi:10.1016/j.ucl.2013.07.010
15. McConnell JD, Roehrborn CG, Bautista OM, et al. The long-term effect of doxazosin, finasteride, and combination therapy on the clinical progression of benign prostatic hyperplasia. N Engl J Med. 2003;349(25):2387-2398. doi:10.1056/NEJMoa030656
16. Roehrborn CG, Barkin J, Siami P, et al. Clinical outcomes after combined therapy with dutasteride plus tamsulosin or either monotherapy in men with benign prostatic hyperplasia (BPH) by baseline characteristics: 4-year results from the randomized, double-blind Combination of Avodart and Tamsulosin (CombAT) trial. BJU Int. 2011;107(6):946-954. doi:10.1111/j.1464-410X.2011.10124.x
17. Djavan B, Marberger M. A meta-analysis on the efficacy and tolerability of alpha1-adrenoceptor antagonists in patients with lower urinary tract symptoms suggestive of benign prostatic obstruction. Eur Urol. 1999;36(1):1-13. doi:10.1159/000019919
18. By the American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American Geriatrics Society 2015 Updated Beers Criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2015;63(11):2227-2246. doi:10.1111/jgs.13702
19. Duan Y, Grady JJ, Albertsen PC, Helen Wu Z. Tamsulosin and the risk of dementia in older men with benign prostatic hyperplasia. Pharmacoepidemiol Drug Saf. 2018;27(3):340- 348. doi:10.1002/pds.4361
20. Coupland CAC, Hill T, Dening T, Morriss R, Moore M, Hippisley-Cox J. Anticholinergic drug exposure and the risk of dementia: a nested case-control study. JAMA Intern Med. 2019;179(8):1084-1093. doi:10.1001/jamainternmed.2019.0677
21. Parsons JK, Dahm P, Köhler TS, Lerner LB, Wilt TJ. Surgical management of lower urinary tract symptoms attributed to benign prostatic hyperplasia: AUA guideline amendment 2020. J Urol. 2020;204(4):799-804. doi:10.1097/JU.0000000000001298
22. Smith AB, Carson CC. Finasteride in the treatment of patients with benign prostatic hyperplasia: a review. Ther Clin Risk Manag. 2009;5(3):535-545. doi:10.2147/tcrm.s6195
23. Andriole GL, Guess HA, Epstein JI, et al. Treatment with finasteride preserves usefulness of prostate-specific antigen in the detection of prostate cancer: results of a randomized, double-blind, placebo-controlled clinical trial. PLESS Study Group. Proscar Long-term Efficacy and Safety Study. Urology. 1998;52(2):195-202. doi:10.1016/s0090-4295(98)00184-8
24. McConnell JD, Bruskewitz R, Walsh P, et al. The effect of finasteride on the risk of acute urinary retention and the need for surgical treatment among men with benign prostatic hyperplasia. Finasteride Long-Term Efficacy and Safety Study Group. N Engl J Med. 1998;338(9):557-563. doi:10.1056/NEJM199802263380901
25. Rittmaster RS. 5alpha-reductase inhibitors in benign prostatic hyperplasia and prostate cancer risk reduction. Best Pract Res Clin Endocrinol Metab. 2008;22(2):389-402. doi:10.1016/j.beem.2008.01.016
26. La Torre A, Giupponi G, Duffy D, Conca A, Cai T, Scardigli A. Sexual dysfunction related to drugs: a critical review. Part V: α-blocker and 5-ARI drugs. Pharmacopsychiatry. 2016;49(1):3-13. doi:10.1055/s-0035-1565100
27. Corona G, Tirabassi G, Santi D, et al. Sexual dysfunction in subjects treated with inhibitors of 5α-reductase for benign prostatic hyperplasia: a comprehensive review and meta-analysis. Andrology. 2017;5(4):671-678. doi:10.1111/andr.12353
28. Trost L, Saitz TR, Hellstrom WJ. Side effects of 5-alpha reductase inhibitors: a comprehensive review. Sex Med Rev. 2013;1(1):24-41. doi:10.1002/smrj.3
29. Welk B, McArthur E, Ordon M, Anderson KK, Hayward J, Dixon S. Association of suicidality and depression with 5α-reductase inhibitors. JAMA Intern Med. 2017;177(5):683-691. doi:10.1001/jamainternmed.2017.0089
30. Kaplan SA, Roehrborn CG, Rovner ES, Carlsson M, Bavendam T, Guan Z. Tolterodine and tamsulosin for treatment of men with lower urinary tract symptoms and overactive bladder: a randomized controlled trial [published correction appears in JAMA. 2007 Mar 21:297(11):1195] [published correction appears in JAMA. 2007 Oct 24;298(16):1864]. JAMA. 2006;296(19):2319-2328. doi:10.1001/jama.296.19.2319
31. Nitti VW, Auerbach S, Martin N, Calhoun A, Lee M, Herschorn S. Results of a randomized phase III trial of mirabegron in patients with overactive bladder. J Urol. 2013;189(4):1388-1395. doi:10.1016/j.juro.2012.10.017
32. Chapple CR, Cardozo L, Nitti VW, Siddiqui E, Michel MC. Mirabegron in overactive bladder: a review of efficacy, safety, and tolerability. Neurourol Urodyn. 2014;33(1):17-30. doi:10.1002/nau.22505
33. Rutman MP, King JR, Bennett N, Ankrom W, Mudd PN. PD14-01 once-daily vibegron, a novel oral β3 agonist does not inhibit CYP2D6, a common pathway for drug metabolism in patients on OAB medications. J Urol. 2019;201(Suppl 4):e231. doi:10.1097/01.JU.0000555478.73162.19
34. Bo K, Frawley HC, Haylen BT, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for the conservative and nonpharmacological management of female pelvic floor dysfunction. Neurourol Urodyn. 2017;36(2):221- 244. doi:10.1002/nau.23107
35. Cindolo L, Pirozzi L, Fanizza C, et al. Drug adherence and clinical outcomes for patients under pharmacological therapy for lower urinary tract symptoms related to benign prostatic hyperplasia: population-based cohort study. Eur Urol. 2015;68(3):418-425. doi:10.1016/j.eururo.2014.11.006
36. Ruhaiyem ME, Alshehri AA, Saade M, Shoabi TA, Zahoor H, Tawfeeq NA. Fear of going under general anesthesia: a cross-sectional study. Saudi J Anaesth. 2016;10(3):317- 321. doi:10.4103/1658-354X.179094
37. Hashim MJ. Patient-centered communication: basic skills. Am Fam Physician. 2017;95(1):29-34.
38. Roehrborn CG, Barkin J, Gange SN, et al. Five year results of the prospective randomized controlled prostatic urethral L.I.F.T. study. Can J Urol. 2017;24(3):8802-8813.
39. Gratzke C, Barber N, Speakman MJ, et al. Prostatic urethral lift vs transurethral resection of the prostate: 2-year results of the BPH6 prospective, multicentre, randomized study. BJU Int. 2017;119(5):767-775.doi:10.1111/bju.13714
40. Sønksen J, Barber NJ, Speakman MJ, et al. Prospective, randomized, multinational study of prostatic urethral lift versus transurethral resection of the prostate: 12-month results from the BPH6 study. Eur Urol. 2015;68(4):643-652. doi:10.1016/j.eururo.2015.04.024
41. Roehrborn CG, Gange SN, Shore ND, et al. The prostatic urethral lift for the treatment of lower urinary tract symptoms associated with prostate enlargement due to benign prostatic hyperplasia: the L.I.F.T. Study. J Urol. 2013;190(6):2161-2167. doi:10.1016/j.juro.2013.05.116
42. McNicholas TA. Benign prostatic hyperplasia and new treatment options - a critical appraisal of the UroLift system. Med Devices (Auckl). 2016;9:115-123. Published 2016 May 19. doi:10.2147/MDER.S60780
43. McVary KT, Rogers T, Roehrborn CG. Rezuˉm Water Vapor thermal therapy for lower urinary tract symptoms associated with benign prostatic hyperplasia: 4-year results from randomized controlled study. Urology. 2019;126:171-179. doi:10.1016/j.urology.2018.12.041
44. Bole R, Gopalakrishna A, Kuang R, et al. Comparative postoperative outcomes of Rezˉum prostate ablation in patients with large versus small glands. J Endourol. 2020;34(7):778-781. doi:10.1089/end.2020.0177
45. Darson MF, Alexander EE, Schiffman ZJ, et al. Procedural techniques and multicenter postmarket experience using minimally invasive convective radiofrequency thermal therapy with Rezˉum system for treatment of lower urinary tract symptoms due to benign prostatic hyperplasia. Res Rep Urol. 2017;9:159-168. Published 2017 Aug 21. doi:10.2147/RRU.S143679
46. Baazeem A, Elhilali MM. Surgical management of benign prostatic hyperplasia: current evidence. Nat Clin Pract Urol. 2008;5(10):540-549. doi:10.1038/ncpuro1214
47. Rassweiler J, Teber D, Kuntz R, Hofmann R. Complications of transurethral resection of the prostate (TURP)- -incidence, management, and prevention. Eur Urol. 2006;50(5):969-980. doi:10.1016/j.eururo.2005.12.042
48. Abt D, Schmid HP, Speakman MJ. Reasons to consider prostatic artery embolization. World J Urol. 2021;39(7):2301-2306. doi:10.1007/s00345-021-03601-z
49. Nguyen DD, Barber N, Bidair M, et al. Waterjet Ablation Therapy for Endoscopic Resection of prostate tissue trial (WATER) vs WATER II: comparing Aquablation therapy for benign prostatic hyperplasia in30-80and80-150mLprostates. BJUInt. 2020;125(1):112-122. doi:10.1111/bju.14917.
2021 Update on bone health
Recently, the National Osteoporosis Foundation (NOF) changed its name to the Bone Health and Osteoporosis Foundation (BHOF). Several years ago, in 2016 at my urging, this column was renamed from “Update on osteoporosis” to “Update on bone health.” I believe we were on the leading edge of this movement. As expressed in last year’s Update, our patients’ bone health must be emphasized more than it has been in the past.1
Consider that localized breast cancer carries a 5-year survival rate of 99%.2 Most of my patients are keenly aware that periodic competent breast imaging is the key to the earliest possible diagnosis. By contrast, in this country a hip fracture carries a mortality in the first year of 21%!3 Furthermore, approximately one-third of women who fracture their hip do not have osteoporosis.4 While the risk of hip fracture is greatest in women with osteoporosis, it is not absent in those without the condition. Finally, the role of muscle mass, strength, and performance in bone health is a rapidly emerging topic and one that constitutes the core of this year’s Update.
Muscle mass and strength play key role in bone health
de Villiers TJ, Goldstein SR. Update on bone health: the International Menopause Society white paper 2021. Climacteric. 2021;24:498-504. doi:10.1080/13697137.2021.1950967.
Recently, de Villiers and Goldstein offered an overview of osteoporosis.5 What is worthy of reporting here is the role of muscle in bone health.
The bone-muscle relationship
Most clinicians know that osteoporosis and osteopenia are well-defined conditions with known risks associated with fracture. According to a review of PubMed, the first article with the keyword “osteoporosis” was published in 1894; through May 2020, 93,335 articles used that keyword. “Osteoporosis” is derived from the Greek osteon (bone) and poros (little hole). Thus, osteoporosis means “porous bone.”
Sarcopenia is characterized by progressive and generalized loss of skeletal muscle mass, strength, and function, and the condition is associated with a risk of adverse outcomes that include physical disabilities, poor quality of life, and death.6,7 “Sarcopenia” has its roots in the Greek words sarx (flesh) and penia (loss), and the term was coined in 1989.8 A PubMed review that included “sarcopenia” as the keyword revealed that the first article was published in 1993, with 12,068 articles published through May 2020.
Notably, muscle accounts for about 60% of the body’s protein. Muscle mass decreases with age, but younger patients with malnutrition, cachexia, or inflammatory diseases are also prone to decreased muscle mass. While osteoporosis has a well-accepted definition based on dual-energy x-ray absorptiometry (DXA) measurements, sarcopenia has no universally accepted definition, consensus diagnostic criteria, or treatment guidelines. In 2016, however, the International Classification of Diseases, Tenth Revision, Clinical Modification (CD-10-CM) finally recognized sarcopenia as a disease entity.
Currently, the most widely accepted definition comes from the European Working Group on Sarcopenia in Older People, which labeled presarcopenia as low muscle mass without impact on muscle strength or performance; sarcopenia as low muscle mass with either low muscle strength or low physical performance; and severe sarcopenia has all 3 criteria being present.9
When osteosarcopenia (osteoporosis or osteopenia combined with sarcopenia) exists, it can result in a threefold increase in risk of falls and a fourfold increase in fracture risk compared with women who have osteopenia or osteoporosis alone.10
The morbidity and mortality from fragility fractures are well known. Initially, diagnosis of risk seemed to be mainly T-scores on bone mineral density (BMD) testing (normal, osteopenic, osteoporosis). The FRAX fracture risk assessment tool, which includes a number of variables, further refined risk assessment. Increasingly, there is evidence of crosstalk between muscle and bone. Sarcopenia, the loss of skeletal muscle mass, strength, and performance, appears to play an important role as well for fracture risk. Simple tools to evaluate a patient’s muscle status exist. At the very least, resistance and balance exercises should be part of all clinicians’ patient counseling for bone health.
Continue to: Denosumab decreased falls risk, improved sarcopenia measures vs comparator antiresorptives...
Denosumab decreased falls risk, improved sarcopenia measures vs comparator antiresorptives
El Miedany Y, El Gaafary M, Toth M, et al; Egyptian Academy of Bone Health, Metabolic Bone Diseases. Is there a potential dual effect of denosumab for treatment of osteoporosis and sarcopenia? Clin Rheumatol. 2021;40:4225-4232. doi: 10.1007/s10067-021 -05757-w.
Osteosarcopenia, the combination of osteoporosis or osteopenia with sarcopenia, has been shown to increase the overall rate of falls and fracture when compared with fall and fracture rates in women with osteopenia or osteoporosis alone.10 A study by El Miedany and colleagues examined whether denosumab treatment had a possible dual therapeutic effect on osteoporosis and sarcopenia.11
Study details
The investigators looked at 135 patients diagnosed with postmenopausal osteoporosis and who were prescribed denosumab and compared them with a control group of 272 patients stratified into 2 subgroups: 136 were prescribed alendronate and 136 were prescribed zoledronate.
Assessments were performed for all participants for BMD (DXA), fall risk (falls risk assessment score [FRAS]), fracture risk (FRAX assessment tool), and sarcopenia measures. Reassessments were conducted after 5 years of denosumab or alendronate therapy, 3 years of zoledronate therapy, and 1 year after stopping the osteoporosis therapy.
The FRAS uses the clinical variables of history of falls in the last 12 months, impaired sight, weak hand grip, history of loss of balance in the last 12 months, and slowing of the walking speed/change in gait to yield a percent chance of sustaining a fall.12 Sarcopenic measures include grip strength, timed up and go (TUG) mobility test, and gait speed. There were no significant demographic differences between the 3 groups.
Denosumab reduced risk of falls and positively affected muscle strength
On completion of the 5-year denosumab therapy, falls risk was significantly decreased (P = .001) and significant improvements were seen in all sarcopenia measures (P = .01). One year after denosumab was discontinued, a significant worsening of both falls risk and sarcopenia measures (P = .01) occurred. This was in contrast to results in both control groups (alendronate and zoledronate), in which there was an improvement, although less robust in gait speed and the TUG test (P = .05) but no improvement in risk of falls. Thus, the results of this study showed that denosumab not only improved bone mass but also reduced falls risk.
Compared with bisphosphonates, denosumab showed the highest significant positive effect on both physical performance and skeletal muscle strength. This is evidenced by improvement of the gait speed, TUG test, and 4-m walk test (P<.001) in the denosumab group versus in the alendronate and zoledronate group (P<.05).
These results agree with the outcomes of the FREEDOM (Fracture Reduction Evaluation of Denosumab in Osteoporosis 6 months) trial, which revealed that not only did denosumab treatment reduce the risk of vertebral, nonvertebral, and hip fracture over 36 months, but also that the denosumab-treated group had fewer falls (4.5%) compared with the other groups (5.7%) (P = .02).13
These data highlight that osteoporosis and sarcopenia may share similar underlying risk factors and that muscle-bone interactions are important to minimize the risk of falls, fractures, and hospitalizations. While all 3 antiresorptives (denosumab, alendronate, zoledronate) improved measures of BMD and sarcopenia, only denosumab resulted in a reduction in the FRAS risk of falls score.
Continue to: Estrogen’s role in bone health and its therapeutic potential in osteosarcopenia...
Estrogen’s role in bone health and its therapeutic potential in osteosarcopenia
Mandelli A, Tacconi E, Levinger I, et al. The role of estrogens in osteosarcopenia: from biology to potential dual therapeutic effects. Climacteric. 2021;1-7. doi: 10.1080/13697137.2021.1965118.
Osteosarcopenia is a particular term used to describe the coexistence of 2 pathologies, osteopenia/ osteoporosis and sarcopenia.14 Sarcopenia is characterized by a loss of muscle mass, strength, and performance. Numerous studies indicate that higher lean body mass is related to increased BMD and reduced fracture risk, especially in postmenopausal women.15
Menopause, muscle, and estrogen’s physiologic effects
Estrogens play a critical role in maintaining bone and muscle mass in women. Women experience a decline in musculoskeletal quantity and quality at the onset of menopause.16 Muscle mass and strength decrease rapidly after menopause, which suggests that degradation of muscle protein begins to exert a more significant effect due to a decrease in protein synthesis. Indeed, a reduced response to anabolic stimuli has been shown in postmenopausal women.17 Normalization of the protein synthesis response after restoring estrogen levels with estrogen therapy supports this hypothesis.18
In a meta-analysis to identify the role of estrogen therapy on muscle strength, the authors concluded that estrogens benefit muscle strength not by increasing the skeletal mass but by improving muscle quality and its ability to generate force.19 In addition, however, it has been demonstrated that exercise prevents and delays the onset of osteosarcopenia.20
Estrogens play a crucial role in maintaining bone and skeletal muscle health in women. Estrogen therapy is an accepted treatment for osteoporosis, whereas its effects on sarcopenia, although promising, indicate that additional studies are required before it can be recommended solely for that purpose. Given the well-described benefits of exercise on muscle and bone health, postmenopausal women should be encouraged to engage in regular physical exercise as a preventive or disease-modifying treatment for osteosarcopenia.
When should bone mass be measured in premenopausal women?
Conradie M, de Villiers T. Premenopausal osteoporosis. Climacteric. 2021:1-14. doi: 10.1080/13697137 .2021.1926974.
Most women’s clinicians are somewhat well acquainted with the increasing importance of preventing, diagnosing, and treating postmenopausal osteoporosis, which predisposes to fragility fracture and the morbidity and even mortality that brings. Increasingly, some younger women are asking for and receiving both bone mass measurements that may be inappropriately ordered and/or wrongly interpreted. Conradie and de Villiers provided an overview of premenopausal osteoporosis, containing important facts that all clinicians who care for women should be aware of.21
Indications for testing
BMD testing is only indicated in younger women in settings in which the result may influence management decisions, such as:
- a history of fragility fracture
- diseases associated with low bone mass, such as anorexia nervosa, hypogonadism, hyperparathyroidism, hyperthyroidism, celiac disease, irritable bowel disease, rheumatoid arthritis, lupus, renal disease, Marfan syndrome
- medications, such as glucocorticoids, aromatase inhibitors, premenopausal tamoxifen, excess thyroid hormone replacement, progesterone contraception
- excessive alcohol consumption, heavy smoking, vitamin D deficiency, calcium deficiency, occasionally veganism or vegetarianism.
BMD interpretation in premenopausal women does not use the T-scores developed for postmenopausal women in which standard deviations (SD) from the mean for a young reference population are employed. In that population, the normal range is up to -1.0 SD; osteopenia > -1.0 < -2.5 SD; and osteoporosis > -2.5 SD. Instead, in premenopausal patients, Z-scores, which compare the measured bone mass to an age- and gender-matched cohort, are employed. Z-scores > 2 SD below the matched population should be used rather than the T-scores that are already familiar to most clinicians.
Up to 90% of these premenopausal women with such skeletal fragility will display the secondary causes described above. ●
Very specific indications are required to consider bone mass measurements in premenopausal women. When measurements are indicated, the values are evaluated by Z-scores that compare them to those of matched-aged women and not by T-scores meant for postmenopausal women. When fragility or low-trauma fractures or Z-scores more than 2 SD below their peers are present, secondary causes of premenopausal osteoporosis include a variety of disease states, medications, and lifestyle situations. When such factors are present, many general women’s health clinicians may want to refer patients for consultation to a metabolic bone specialist for workup and management.
- Goldstein SR. Update on bone health. OBG Manag. 2020;32:16-20, 22-23.
- American Cancer Society. Cancer Facts & Figures 2020. Atlanta, GA: American Cancer Society; 2020. https://www .cancer.org/content/dam/cancer-org/research/cancer-facts -and-statistics/annual-cancer-facts-and-figures/2020/cancer -facts-and-figures-2020.pdf. Accessed November 11, 2021.
- Downey C, Kelly M, Quinlan JF. Changing trends in the mortality rate at 1-year post hip fracture—a systematic review. World J Orthop. 2019;10:166-175.
- Schuit SC, van der Klift M, Weel AE, et al. Fracture incidence and association with bone mineral density in elderly men and women: the Rotterdam Study. Bone. 2004;34:195-202.
- de Villiers, TJ, Goldstein SR. Update on bone health: the International Menopause Society white paper 2021. Climacteric. 2021;24:498-504.
- Goodpaster BH, Park SW, Harris TB, et al. The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J Gerontol A Biol Sci Med Sci. 2006;61:1059-1064.
- Santilli V, Bernetti A, Mangone M, et al. Clinical definition of sarcopenia. Clin Cases Miner Bone Metab. 2014;11:177-180.
- Rosenberg I. Epidemiological and methodological problems in determining nutritional status of older persons. Proceedings of a conference. Albuquerque, New Mexico, October 19-21, 1989. Am J Clin Nutr. 1989;50:1231-1233.
- Cruz-Jentoft AJ, Baeyens JP, Bauer JM, et al; European Working Group on Sarcopenia in Older People. Sarcopenia: European consensus on definition and diagnosis—report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010;39:412-423.
- Sepúlveda-Loyola W, Phu S, Bani Hassan E, et al. The joint occurrence of osteoporosis and sarcopenia (osteosarcopenia): definitions and characteristics. J Am Med Dir Assoc. 2020;21:220-225.
- El Miedany Y, El Gaafary M, Toth M, et al; Egyptian Academy of Bone Health, Metabolic Bone Diseases. Is there a potential dual effect of denosumab for treatment of osteoporosis and sarcopenia? Clin Rheumatol. 2021;40:4225-4232.
- El Miedany Y, El Gaafary M, Toth M, et al. Falls risk assessment score (FRAS): time to rethink. J Clin Gerontol Geriatr. 2011;21-26.
- Cummings SR, Martin JS, McClung MR, et al; FREEDOM Trial. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med. 2009;361: 756-765.
- Inoue T, Maeda K, Nagano A, et al. Related factors and clinical outcomes of osteosarcopenia: a narrative review. Nutrients. 2021;13:291.
- Kaji H. Linkage between muscle and bone: common catabolic signals resulting in osteoporosis and sarcopenia. Curr Opin Clin Nutr Metab Care. 2013;16:272-277.
- Sipilä S, Törmäkangas T, Sillanpää E, et al. Muscle and bone mass in middle‐aged women: role of menopausal status and physical activity. J Cachexia Sarcopenia Muscle. 2020;11: 698-709.
- Bamman MM, Hill VJ, Adams GR, et al. Gender differences in resistance-training-induced myofiber hypertrophy among older adults. J Gerontol A Biol Sci Med Sci. 2003;58:108-116.
- Hansen M, Skovgaard D, Reitelseder S, et al. Effects of estrogen replacement and lower androgen status on skeletal muscle collagen and myofibrillar protein synthesis in postmenopausal women. J Gerontol A Biol Sci Med Sci. 2012;67:1005-1013.
- Greising SM, Baltgalvis KA, Lowe DA, et al. Hormone therapy and skeletal muscle strength: a meta-analysis. J Gerontol A Biol Sci Med Sci. 2009;64:1071-1081.
- Cariati I, Bonanni R, Onorato F, et al. Role of physical activity in bone-muscle crosstalk: biological aspects and clinical implications. J Funct Morphol Kinesiol. 2021;6:55.
- Conradie M, de Villiers T. Premenopausal osteoporosis. Climacteric. 2021:1-14.
Recently, the National Osteoporosis Foundation (NOF) changed its name to the Bone Health and Osteoporosis Foundation (BHOF). Several years ago, in 2016 at my urging, this column was renamed from “Update on osteoporosis” to “Update on bone health.” I believe we were on the leading edge of this movement. As expressed in last year’s Update, our patients’ bone health must be emphasized more than it has been in the past.1
Consider that localized breast cancer carries a 5-year survival rate of 99%.2 Most of my patients are keenly aware that periodic competent breast imaging is the key to the earliest possible diagnosis. By contrast, in this country a hip fracture carries a mortality in the first year of 21%!3 Furthermore, approximately one-third of women who fracture their hip do not have osteoporosis.4 While the risk of hip fracture is greatest in women with osteoporosis, it is not absent in those without the condition. Finally, the role of muscle mass, strength, and performance in bone health is a rapidly emerging topic and one that constitutes the core of this year’s Update.
Muscle mass and strength play key role in bone health
de Villiers TJ, Goldstein SR. Update on bone health: the International Menopause Society white paper 2021. Climacteric. 2021;24:498-504. doi:10.1080/13697137.2021.1950967.
Recently, de Villiers and Goldstein offered an overview of osteoporosis.5 What is worthy of reporting here is the role of muscle in bone health.
The bone-muscle relationship
Most clinicians know that osteoporosis and osteopenia are well-defined conditions with known risks associated with fracture. According to a review of PubMed, the first article with the keyword “osteoporosis” was published in 1894; through May 2020, 93,335 articles used that keyword. “Osteoporosis” is derived from the Greek osteon (bone) and poros (little hole). Thus, osteoporosis means “porous bone.”
Sarcopenia is characterized by progressive and generalized loss of skeletal muscle mass, strength, and function, and the condition is associated with a risk of adverse outcomes that include physical disabilities, poor quality of life, and death.6,7 “Sarcopenia” has its roots in the Greek words sarx (flesh) and penia (loss), and the term was coined in 1989.8 A PubMed review that included “sarcopenia” as the keyword revealed that the first article was published in 1993, with 12,068 articles published through May 2020.
Notably, muscle accounts for about 60% of the body’s protein. Muscle mass decreases with age, but younger patients with malnutrition, cachexia, or inflammatory diseases are also prone to decreased muscle mass. While osteoporosis has a well-accepted definition based on dual-energy x-ray absorptiometry (DXA) measurements, sarcopenia has no universally accepted definition, consensus diagnostic criteria, or treatment guidelines. In 2016, however, the International Classification of Diseases, Tenth Revision, Clinical Modification (CD-10-CM) finally recognized sarcopenia as a disease entity.
Currently, the most widely accepted definition comes from the European Working Group on Sarcopenia in Older People, which labeled presarcopenia as low muscle mass without impact on muscle strength or performance; sarcopenia as low muscle mass with either low muscle strength or low physical performance; and severe sarcopenia has all 3 criteria being present.9
When osteosarcopenia (osteoporosis or osteopenia combined with sarcopenia) exists, it can result in a threefold increase in risk of falls and a fourfold increase in fracture risk compared with women who have osteopenia or osteoporosis alone.10
The morbidity and mortality from fragility fractures are well known. Initially, diagnosis of risk seemed to be mainly T-scores on bone mineral density (BMD) testing (normal, osteopenic, osteoporosis). The FRAX fracture risk assessment tool, which includes a number of variables, further refined risk assessment. Increasingly, there is evidence of crosstalk between muscle and bone. Sarcopenia, the loss of skeletal muscle mass, strength, and performance, appears to play an important role as well for fracture risk. Simple tools to evaluate a patient’s muscle status exist. At the very least, resistance and balance exercises should be part of all clinicians’ patient counseling for bone health.
Continue to: Denosumab decreased falls risk, improved sarcopenia measures vs comparator antiresorptives...
Denosumab decreased falls risk, improved sarcopenia measures vs comparator antiresorptives
El Miedany Y, El Gaafary M, Toth M, et al; Egyptian Academy of Bone Health, Metabolic Bone Diseases. Is there a potential dual effect of denosumab for treatment of osteoporosis and sarcopenia? Clin Rheumatol. 2021;40:4225-4232. doi: 10.1007/s10067-021 -05757-w.
Osteosarcopenia, the combination of osteoporosis or osteopenia with sarcopenia, has been shown to increase the overall rate of falls and fracture when compared with fall and fracture rates in women with osteopenia or osteoporosis alone.10 A study by El Miedany and colleagues examined whether denosumab treatment had a possible dual therapeutic effect on osteoporosis and sarcopenia.11
Study details
The investigators looked at 135 patients diagnosed with postmenopausal osteoporosis and who were prescribed denosumab and compared them with a control group of 272 patients stratified into 2 subgroups: 136 were prescribed alendronate and 136 were prescribed zoledronate.
Assessments were performed for all participants for BMD (DXA), fall risk (falls risk assessment score [FRAS]), fracture risk (FRAX assessment tool), and sarcopenia measures. Reassessments were conducted after 5 years of denosumab or alendronate therapy, 3 years of zoledronate therapy, and 1 year after stopping the osteoporosis therapy.
The FRAS uses the clinical variables of history of falls in the last 12 months, impaired sight, weak hand grip, history of loss of balance in the last 12 months, and slowing of the walking speed/change in gait to yield a percent chance of sustaining a fall.12 Sarcopenic measures include grip strength, timed up and go (TUG) mobility test, and gait speed. There were no significant demographic differences between the 3 groups.
Denosumab reduced risk of falls and positively affected muscle strength
On completion of the 5-year denosumab therapy, falls risk was significantly decreased (P = .001) and significant improvements were seen in all sarcopenia measures (P = .01). One year after denosumab was discontinued, a significant worsening of both falls risk and sarcopenia measures (P = .01) occurred. This was in contrast to results in both control groups (alendronate and zoledronate), in which there was an improvement, although less robust in gait speed and the TUG test (P = .05) but no improvement in risk of falls. Thus, the results of this study showed that denosumab not only improved bone mass but also reduced falls risk.
Compared with bisphosphonates, denosumab showed the highest significant positive effect on both physical performance and skeletal muscle strength. This is evidenced by improvement of the gait speed, TUG test, and 4-m walk test (P<.001) in the denosumab group versus in the alendronate and zoledronate group (P<.05).
These results agree with the outcomes of the FREEDOM (Fracture Reduction Evaluation of Denosumab in Osteoporosis 6 months) trial, which revealed that not only did denosumab treatment reduce the risk of vertebral, nonvertebral, and hip fracture over 36 months, but also that the denosumab-treated group had fewer falls (4.5%) compared with the other groups (5.7%) (P = .02).13
These data highlight that osteoporosis and sarcopenia may share similar underlying risk factors and that muscle-bone interactions are important to minimize the risk of falls, fractures, and hospitalizations. While all 3 antiresorptives (denosumab, alendronate, zoledronate) improved measures of BMD and sarcopenia, only denosumab resulted in a reduction in the FRAS risk of falls score.
Continue to: Estrogen’s role in bone health and its therapeutic potential in osteosarcopenia...
Estrogen’s role in bone health and its therapeutic potential in osteosarcopenia
Mandelli A, Tacconi E, Levinger I, et al. The role of estrogens in osteosarcopenia: from biology to potential dual therapeutic effects. Climacteric. 2021;1-7. doi: 10.1080/13697137.2021.1965118.
Osteosarcopenia is a particular term used to describe the coexistence of 2 pathologies, osteopenia/ osteoporosis and sarcopenia.14 Sarcopenia is characterized by a loss of muscle mass, strength, and performance. Numerous studies indicate that higher lean body mass is related to increased BMD and reduced fracture risk, especially in postmenopausal women.15
Menopause, muscle, and estrogen’s physiologic effects
Estrogens play a critical role in maintaining bone and muscle mass in women. Women experience a decline in musculoskeletal quantity and quality at the onset of menopause.16 Muscle mass and strength decrease rapidly after menopause, which suggests that degradation of muscle protein begins to exert a more significant effect due to a decrease in protein synthesis. Indeed, a reduced response to anabolic stimuli has been shown in postmenopausal women.17 Normalization of the protein synthesis response after restoring estrogen levels with estrogen therapy supports this hypothesis.18
In a meta-analysis to identify the role of estrogen therapy on muscle strength, the authors concluded that estrogens benefit muscle strength not by increasing the skeletal mass but by improving muscle quality and its ability to generate force.19 In addition, however, it has been demonstrated that exercise prevents and delays the onset of osteosarcopenia.20
Estrogens play a crucial role in maintaining bone and skeletal muscle health in women. Estrogen therapy is an accepted treatment for osteoporosis, whereas its effects on sarcopenia, although promising, indicate that additional studies are required before it can be recommended solely for that purpose. Given the well-described benefits of exercise on muscle and bone health, postmenopausal women should be encouraged to engage in regular physical exercise as a preventive or disease-modifying treatment for osteosarcopenia.
When should bone mass be measured in premenopausal women?
Conradie M, de Villiers T. Premenopausal osteoporosis. Climacteric. 2021:1-14. doi: 10.1080/13697137 .2021.1926974.
Most women’s clinicians are somewhat well acquainted with the increasing importance of preventing, diagnosing, and treating postmenopausal osteoporosis, which predisposes to fragility fracture and the morbidity and even mortality that brings. Increasingly, some younger women are asking for and receiving both bone mass measurements that may be inappropriately ordered and/or wrongly interpreted. Conradie and de Villiers provided an overview of premenopausal osteoporosis, containing important facts that all clinicians who care for women should be aware of.21
Indications for testing
BMD testing is only indicated in younger women in settings in which the result may influence management decisions, such as:
- a history of fragility fracture
- diseases associated with low bone mass, such as anorexia nervosa, hypogonadism, hyperparathyroidism, hyperthyroidism, celiac disease, irritable bowel disease, rheumatoid arthritis, lupus, renal disease, Marfan syndrome
- medications, such as glucocorticoids, aromatase inhibitors, premenopausal tamoxifen, excess thyroid hormone replacement, progesterone contraception
- excessive alcohol consumption, heavy smoking, vitamin D deficiency, calcium deficiency, occasionally veganism or vegetarianism.
BMD interpretation in premenopausal women does not use the T-scores developed for postmenopausal women in which standard deviations (SD) from the mean for a young reference population are employed. In that population, the normal range is up to -1.0 SD; osteopenia > -1.0 < -2.5 SD; and osteoporosis > -2.5 SD. Instead, in premenopausal patients, Z-scores, which compare the measured bone mass to an age- and gender-matched cohort, are employed. Z-scores > 2 SD below the matched population should be used rather than the T-scores that are already familiar to most clinicians.
Up to 90% of these premenopausal women with such skeletal fragility will display the secondary causes described above. ●
Very specific indications are required to consider bone mass measurements in premenopausal women. When measurements are indicated, the values are evaluated by Z-scores that compare them to those of matched-aged women and not by T-scores meant for postmenopausal women. When fragility or low-trauma fractures or Z-scores more than 2 SD below their peers are present, secondary causes of premenopausal osteoporosis include a variety of disease states, medications, and lifestyle situations. When such factors are present, many general women’s health clinicians may want to refer patients for consultation to a metabolic bone specialist for workup and management.
Recently, the National Osteoporosis Foundation (NOF) changed its name to the Bone Health and Osteoporosis Foundation (BHOF). Several years ago, in 2016 at my urging, this column was renamed from “Update on osteoporosis” to “Update on bone health.” I believe we were on the leading edge of this movement. As expressed in last year’s Update, our patients’ bone health must be emphasized more than it has been in the past.1
Consider that localized breast cancer carries a 5-year survival rate of 99%.2 Most of my patients are keenly aware that periodic competent breast imaging is the key to the earliest possible diagnosis. By contrast, in this country a hip fracture carries a mortality in the first year of 21%!3 Furthermore, approximately one-third of women who fracture their hip do not have osteoporosis.4 While the risk of hip fracture is greatest in women with osteoporosis, it is not absent in those without the condition. Finally, the role of muscle mass, strength, and performance in bone health is a rapidly emerging topic and one that constitutes the core of this year’s Update.
Muscle mass and strength play key role in bone health
de Villiers TJ, Goldstein SR. Update on bone health: the International Menopause Society white paper 2021. Climacteric. 2021;24:498-504. doi:10.1080/13697137.2021.1950967.
Recently, de Villiers and Goldstein offered an overview of osteoporosis.5 What is worthy of reporting here is the role of muscle in bone health.
The bone-muscle relationship
Most clinicians know that osteoporosis and osteopenia are well-defined conditions with known risks associated with fracture. According to a review of PubMed, the first article with the keyword “osteoporosis” was published in 1894; through May 2020, 93,335 articles used that keyword. “Osteoporosis” is derived from the Greek osteon (bone) and poros (little hole). Thus, osteoporosis means “porous bone.”
Sarcopenia is characterized by progressive and generalized loss of skeletal muscle mass, strength, and function, and the condition is associated with a risk of adverse outcomes that include physical disabilities, poor quality of life, and death.6,7 “Sarcopenia” has its roots in the Greek words sarx (flesh) and penia (loss), and the term was coined in 1989.8 A PubMed review that included “sarcopenia” as the keyword revealed that the first article was published in 1993, with 12,068 articles published through May 2020.
Notably, muscle accounts for about 60% of the body’s protein. Muscle mass decreases with age, but younger patients with malnutrition, cachexia, or inflammatory diseases are also prone to decreased muscle mass. While osteoporosis has a well-accepted definition based on dual-energy x-ray absorptiometry (DXA) measurements, sarcopenia has no universally accepted definition, consensus diagnostic criteria, or treatment guidelines. In 2016, however, the International Classification of Diseases, Tenth Revision, Clinical Modification (CD-10-CM) finally recognized sarcopenia as a disease entity.
Currently, the most widely accepted definition comes from the European Working Group on Sarcopenia in Older People, which labeled presarcopenia as low muscle mass without impact on muscle strength or performance; sarcopenia as low muscle mass with either low muscle strength or low physical performance; and severe sarcopenia has all 3 criteria being present.9
When osteosarcopenia (osteoporosis or osteopenia combined with sarcopenia) exists, it can result in a threefold increase in risk of falls and a fourfold increase in fracture risk compared with women who have osteopenia or osteoporosis alone.10
The morbidity and mortality from fragility fractures are well known. Initially, diagnosis of risk seemed to be mainly T-scores on bone mineral density (BMD) testing (normal, osteopenic, osteoporosis). The FRAX fracture risk assessment tool, which includes a number of variables, further refined risk assessment. Increasingly, there is evidence of crosstalk between muscle and bone. Sarcopenia, the loss of skeletal muscle mass, strength, and performance, appears to play an important role as well for fracture risk. Simple tools to evaluate a patient’s muscle status exist. At the very least, resistance and balance exercises should be part of all clinicians’ patient counseling for bone health.
Continue to: Denosumab decreased falls risk, improved sarcopenia measures vs comparator antiresorptives...
Denosumab decreased falls risk, improved sarcopenia measures vs comparator antiresorptives
El Miedany Y, El Gaafary M, Toth M, et al; Egyptian Academy of Bone Health, Metabolic Bone Diseases. Is there a potential dual effect of denosumab for treatment of osteoporosis and sarcopenia? Clin Rheumatol. 2021;40:4225-4232. doi: 10.1007/s10067-021 -05757-w.
Osteosarcopenia, the combination of osteoporosis or osteopenia with sarcopenia, has been shown to increase the overall rate of falls and fracture when compared with fall and fracture rates in women with osteopenia or osteoporosis alone.10 A study by El Miedany and colleagues examined whether denosumab treatment had a possible dual therapeutic effect on osteoporosis and sarcopenia.11
Study details
The investigators looked at 135 patients diagnosed with postmenopausal osteoporosis and who were prescribed denosumab and compared them with a control group of 272 patients stratified into 2 subgroups: 136 were prescribed alendronate and 136 were prescribed zoledronate.
Assessments were performed for all participants for BMD (DXA), fall risk (falls risk assessment score [FRAS]), fracture risk (FRAX assessment tool), and sarcopenia measures. Reassessments were conducted after 5 years of denosumab or alendronate therapy, 3 years of zoledronate therapy, and 1 year after stopping the osteoporosis therapy.
The FRAS uses the clinical variables of history of falls in the last 12 months, impaired sight, weak hand grip, history of loss of balance in the last 12 months, and slowing of the walking speed/change in gait to yield a percent chance of sustaining a fall.12 Sarcopenic measures include grip strength, timed up and go (TUG) mobility test, and gait speed. There were no significant demographic differences between the 3 groups.
Denosumab reduced risk of falls and positively affected muscle strength
On completion of the 5-year denosumab therapy, falls risk was significantly decreased (P = .001) and significant improvements were seen in all sarcopenia measures (P = .01). One year after denosumab was discontinued, a significant worsening of both falls risk and sarcopenia measures (P = .01) occurred. This was in contrast to results in both control groups (alendronate and zoledronate), in which there was an improvement, although less robust in gait speed and the TUG test (P = .05) but no improvement in risk of falls. Thus, the results of this study showed that denosumab not only improved bone mass but also reduced falls risk.
Compared with bisphosphonates, denosumab showed the highest significant positive effect on both physical performance and skeletal muscle strength. This is evidenced by improvement of the gait speed, TUG test, and 4-m walk test (P<.001) in the denosumab group versus in the alendronate and zoledronate group (P<.05).
These results agree with the outcomes of the FREEDOM (Fracture Reduction Evaluation of Denosumab in Osteoporosis 6 months) trial, which revealed that not only did denosumab treatment reduce the risk of vertebral, nonvertebral, and hip fracture over 36 months, but also that the denosumab-treated group had fewer falls (4.5%) compared with the other groups (5.7%) (P = .02).13
These data highlight that osteoporosis and sarcopenia may share similar underlying risk factors and that muscle-bone interactions are important to minimize the risk of falls, fractures, and hospitalizations. While all 3 antiresorptives (denosumab, alendronate, zoledronate) improved measures of BMD and sarcopenia, only denosumab resulted in a reduction in the FRAS risk of falls score.
Continue to: Estrogen’s role in bone health and its therapeutic potential in osteosarcopenia...
Estrogen’s role in bone health and its therapeutic potential in osteosarcopenia
Mandelli A, Tacconi E, Levinger I, et al. The role of estrogens in osteosarcopenia: from biology to potential dual therapeutic effects. Climacteric. 2021;1-7. doi: 10.1080/13697137.2021.1965118.
Osteosarcopenia is a particular term used to describe the coexistence of 2 pathologies, osteopenia/ osteoporosis and sarcopenia.14 Sarcopenia is characterized by a loss of muscle mass, strength, and performance. Numerous studies indicate that higher lean body mass is related to increased BMD and reduced fracture risk, especially in postmenopausal women.15
Menopause, muscle, and estrogen’s physiologic effects
Estrogens play a critical role in maintaining bone and muscle mass in women. Women experience a decline in musculoskeletal quantity and quality at the onset of menopause.16 Muscle mass and strength decrease rapidly after menopause, which suggests that degradation of muscle protein begins to exert a more significant effect due to a decrease in protein synthesis. Indeed, a reduced response to anabolic stimuli has been shown in postmenopausal women.17 Normalization of the protein synthesis response after restoring estrogen levels with estrogen therapy supports this hypothesis.18
In a meta-analysis to identify the role of estrogen therapy on muscle strength, the authors concluded that estrogens benefit muscle strength not by increasing the skeletal mass but by improving muscle quality and its ability to generate force.19 In addition, however, it has been demonstrated that exercise prevents and delays the onset of osteosarcopenia.20
Estrogens play a crucial role in maintaining bone and skeletal muscle health in women. Estrogen therapy is an accepted treatment for osteoporosis, whereas its effects on sarcopenia, although promising, indicate that additional studies are required before it can be recommended solely for that purpose. Given the well-described benefits of exercise on muscle and bone health, postmenopausal women should be encouraged to engage in regular physical exercise as a preventive or disease-modifying treatment for osteosarcopenia.
When should bone mass be measured in premenopausal women?
Conradie M, de Villiers T. Premenopausal osteoporosis. Climacteric. 2021:1-14. doi: 10.1080/13697137 .2021.1926974.
Most women’s clinicians are somewhat well acquainted with the increasing importance of preventing, diagnosing, and treating postmenopausal osteoporosis, which predisposes to fragility fracture and the morbidity and even mortality that brings. Increasingly, some younger women are asking for and receiving both bone mass measurements that may be inappropriately ordered and/or wrongly interpreted. Conradie and de Villiers provided an overview of premenopausal osteoporosis, containing important facts that all clinicians who care for women should be aware of.21
Indications for testing
BMD testing is only indicated in younger women in settings in which the result may influence management decisions, such as:
- a history of fragility fracture
- diseases associated with low bone mass, such as anorexia nervosa, hypogonadism, hyperparathyroidism, hyperthyroidism, celiac disease, irritable bowel disease, rheumatoid arthritis, lupus, renal disease, Marfan syndrome
- medications, such as glucocorticoids, aromatase inhibitors, premenopausal tamoxifen, excess thyroid hormone replacement, progesterone contraception
- excessive alcohol consumption, heavy smoking, vitamin D deficiency, calcium deficiency, occasionally veganism or vegetarianism.
BMD interpretation in premenopausal women does not use the T-scores developed for postmenopausal women in which standard deviations (SD) from the mean for a young reference population are employed. In that population, the normal range is up to -1.0 SD; osteopenia > -1.0 < -2.5 SD; and osteoporosis > -2.5 SD. Instead, in premenopausal patients, Z-scores, which compare the measured bone mass to an age- and gender-matched cohort, are employed. Z-scores > 2 SD below the matched population should be used rather than the T-scores that are already familiar to most clinicians.
Up to 90% of these premenopausal women with such skeletal fragility will display the secondary causes described above. ●
Very specific indications are required to consider bone mass measurements in premenopausal women. When measurements are indicated, the values are evaluated by Z-scores that compare them to those of matched-aged women and not by T-scores meant for postmenopausal women. When fragility or low-trauma fractures or Z-scores more than 2 SD below their peers are present, secondary causes of premenopausal osteoporosis include a variety of disease states, medications, and lifestyle situations. When such factors are present, many general women’s health clinicians may want to refer patients for consultation to a metabolic bone specialist for workup and management.
- Goldstein SR. Update on bone health. OBG Manag. 2020;32:16-20, 22-23.
- American Cancer Society. Cancer Facts & Figures 2020. Atlanta, GA: American Cancer Society; 2020. https://www .cancer.org/content/dam/cancer-org/research/cancer-facts -and-statistics/annual-cancer-facts-and-figures/2020/cancer -facts-and-figures-2020.pdf. Accessed November 11, 2021.
- Downey C, Kelly M, Quinlan JF. Changing trends in the mortality rate at 1-year post hip fracture—a systematic review. World J Orthop. 2019;10:166-175.
- Schuit SC, van der Klift M, Weel AE, et al. Fracture incidence and association with bone mineral density in elderly men and women: the Rotterdam Study. Bone. 2004;34:195-202.
- de Villiers, TJ, Goldstein SR. Update on bone health: the International Menopause Society white paper 2021. Climacteric. 2021;24:498-504.
- Goodpaster BH, Park SW, Harris TB, et al. The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J Gerontol A Biol Sci Med Sci. 2006;61:1059-1064.
- Santilli V, Bernetti A, Mangone M, et al. Clinical definition of sarcopenia. Clin Cases Miner Bone Metab. 2014;11:177-180.
- Rosenberg I. Epidemiological and methodological problems in determining nutritional status of older persons. Proceedings of a conference. Albuquerque, New Mexico, October 19-21, 1989. Am J Clin Nutr. 1989;50:1231-1233.
- Cruz-Jentoft AJ, Baeyens JP, Bauer JM, et al; European Working Group on Sarcopenia in Older People. Sarcopenia: European consensus on definition and diagnosis—report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010;39:412-423.
- Sepúlveda-Loyola W, Phu S, Bani Hassan E, et al. The joint occurrence of osteoporosis and sarcopenia (osteosarcopenia): definitions and characteristics. J Am Med Dir Assoc. 2020;21:220-225.
- El Miedany Y, El Gaafary M, Toth M, et al; Egyptian Academy of Bone Health, Metabolic Bone Diseases. Is there a potential dual effect of denosumab for treatment of osteoporosis and sarcopenia? Clin Rheumatol. 2021;40:4225-4232.
- El Miedany Y, El Gaafary M, Toth M, et al. Falls risk assessment score (FRAS): time to rethink. J Clin Gerontol Geriatr. 2011;21-26.
- Cummings SR, Martin JS, McClung MR, et al; FREEDOM Trial. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med. 2009;361: 756-765.
- Inoue T, Maeda K, Nagano A, et al. Related factors and clinical outcomes of osteosarcopenia: a narrative review. Nutrients. 2021;13:291.
- Kaji H. Linkage between muscle and bone: common catabolic signals resulting in osteoporosis and sarcopenia. Curr Opin Clin Nutr Metab Care. 2013;16:272-277.
- Sipilä S, Törmäkangas T, Sillanpää E, et al. Muscle and bone mass in middle‐aged women: role of menopausal status and physical activity. J Cachexia Sarcopenia Muscle. 2020;11: 698-709.
- Bamman MM, Hill VJ, Adams GR, et al. Gender differences in resistance-training-induced myofiber hypertrophy among older adults. J Gerontol A Biol Sci Med Sci. 2003;58:108-116.
- Hansen M, Skovgaard D, Reitelseder S, et al. Effects of estrogen replacement and lower androgen status on skeletal muscle collagen and myofibrillar protein synthesis in postmenopausal women. J Gerontol A Biol Sci Med Sci. 2012;67:1005-1013.
- Greising SM, Baltgalvis KA, Lowe DA, et al. Hormone therapy and skeletal muscle strength: a meta-analysis. J Gerontol A Biol Sci Med Sci. 2009;64:1071-1081.
- Cariati I, Bonanni R, Onorato F, et al. Role of physical activity in bone-muscle crosstalk: biological aspects and clinical implications. J Funct Morphol Kinesiol. 2021;6:55.
- Conradie M, de Villiers T. Premenopausal osteoporosis. Climacteric. 2021:1-14.
- Goldstein SR. Update on bone health. OBG Manag. 2020;32:16-20, 22-23.
- American Cancer Society. Cancer Facts & Figures 2020. Atlanta, GA: American Cancer Society; 2020. https://www .cancer.org/content/dam/cancer-org/research/cancer-facts -and-statistics/annual-cancer-facts-and-figures/2020/cancer -facts-and-figures-2020.pdf. Accessed November 11, 2021.
- Downey C, Kelly M, Quinlan JF. Changing trends in the mortality rate at 1-year post hip fracture—a systematic review. World J Orthop. 2019;10:166-175.
- Schuit SC, van der Klift M, Weel AE, et al. Fracture incidence and association with bone mineral density in elderly men and women: the Rotterdam Study. Bone. 2004;34:195-202.
- de Villiers, TJ, Goldstein SR. Update on bone health: the International Menopause Society white paper 2021. Climacteric. 2021;24:498-504.
- Goodpaster BH, Park SW, Harris TB, et al. The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J Gerontol A Biol Sci Med Sci. 2006;61:1059-1064.
- Santilli V, Bernetti A, Mangone M, et al. Clinical definition of sarcopenia. Clin Cases Miner Bone Metab. 2014;11:177-180.
- Rosenberg I. Epidemiological and methodological problems in determining nutritional status of older persons. Proceedings of a conference. Albuquerque, New Mexico, October 19-21, 1989. Am J Clin Nutr. 1989;50:1231-1233.
- Cruz-Jentoft AJ, Baeyens JP, Bauer JM, et al; European Working Group on Sarcopenia in Older People. Sarcopenia: European consensus on definition and diagnosis—report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010;39:412-423.
- Sepúlveda-Loyola W, Phu S, Bani Hassan E, et al. The joint occurrence of osteoporosis and sarcopenia (osteosarcopenia): definitions and characteristics. J Am Med Dir Assoc. 2020;21:220-225.
- El Miedany Y, El Gaafary M, Toth M, et al; Egyptian Academy of Bone Health, Metabolic Bone Diseases. Is there a potential dual effect of denosumab for treatment of osteoporosis and sarcopenia? Clin Rheumatol. 2021;40:4225-4232.
- El Miedany Y, El Gaafary M, Toth M, et al. Falls risk assessment score (FRAS): time to rethink. J Clin Gerontol Geriatr. 2011;21-26.
- Cummings SR, Martin JS, McClung MR, et al; FREEDOM Trial. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med. 2009;361: 756-765.
- Inoue T, Maeda K, Nagano A, et al. Related factors and clinical outcomes of osteosarcopenia: a narrative review. Nutrients. 2021;13:291.
- Kaji H. Linkage between muscle and bone: common catabolic signals resulting in osteoporosis and sarcopenia. Curr Opin Clin Nutr Metab Care. 2013;16:272-277.
- Sipilä S, Törmäkangas T, Sillanpää E, et al. Muscle and bone mass in middle‐aged women: role of menopausal status and physical activity. J Cachexia Sarcopenia Muscle. 2020;11: 698-709.
- Bamman MM, Hill VJ, Adams GR, et al. Gender differences in resistance-training-induced myofiber hypertrophy among older adults. J Gerontol A Biol Sci Med Sci. 2003;58:108-116.
- Hansen M, Skovgaard D, Reitelseder S, et al. Effects of estrogen replacement and lower androgen status on skeletal muscle collagen and myofibrillar protein synthesis in postmenopausal women. J Gerontol A Biol Sci Med Sci. 2012;67:1005-1013.
- Greising SM, Baltgalvis KA, Lowe DA, et al. Hormone therapy and skeletal muscle strength: a meta-analysis. J Gerontol A Biol Sci Med Sci. 2009;64:1071-1081.
- Cariati I, Bonanni R, Onorato F, et al. Role of physical activity in bone-muscle crosstalk: biological aspects and clinical implications. J Funct Morphol Kinesiol. 2021;6:55.
- Conradie M, de Villiers T. Premenopausal osteoporosis. Climacteric. 2021:1-14.