User login
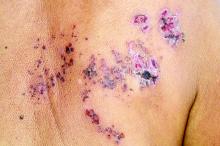
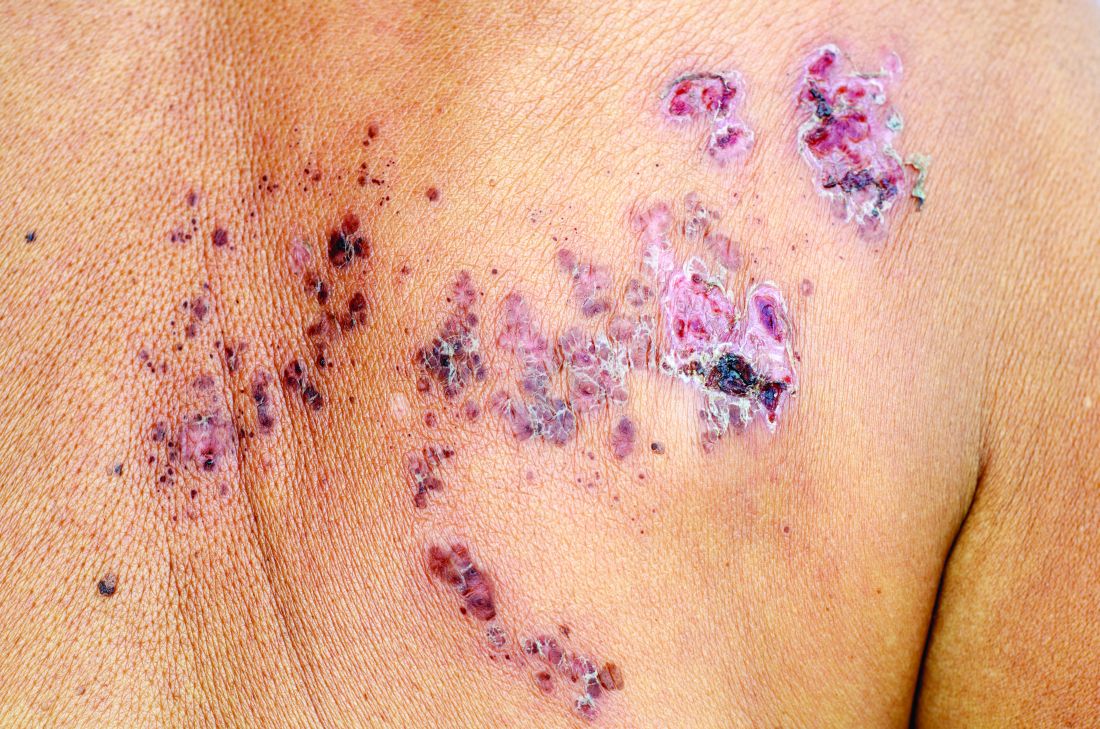
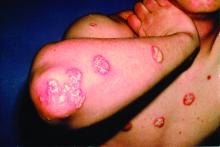
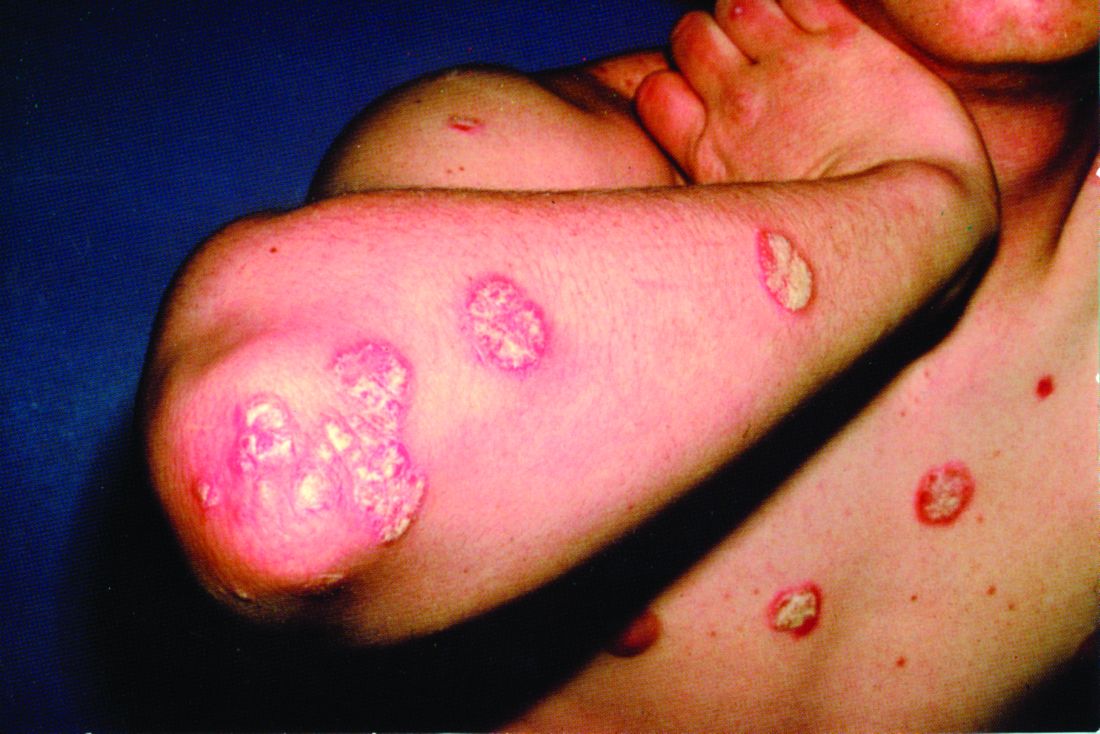
Herpes zoster risk increased with some psoriasis, psoriatic arthritis treatments
All individuals with psoriasis or psoriatic arthritis aged over 50 years should receive the recombinant herpes zoster vaccine, according to a systematic review and consensus recommendations from the National Psoriasis Foundation.
Emily Baumrin, MD, of Brigham and Women’s Hospital, Boston, and her coauthors reviewed 41 studies of herpes zoster in people with psoriasis or psoriatic arthritis according to treatment modality. Their report is in the Journal of the American Academy of Dermatology.
Overall, psoriasis was associated with an increased rate of herpes zoster when compared with the general population: 13.3 cases per 1,000 patient-years for psoriasis and 15.9 for psoriatic arthritis, compared with 8.5 in healthy controls after adjustment for age, sex, and systemic medications. Most of this increased incidence was seen in patients with more severe disease: Those with mild disease who were not receiving systemic therapy had a risk similar to that of healthy controls.
However, one study suggested much of the increased risk of herpes zoster in psoriasis was accounted for by immunosuppressive therapy; when those patients were excluded, there was an 8% increase in risk.
The authors found that people whose psoriasis was treated with tofacitinib (Xeljanz) had a two- to threefold increased risk of herpes zoster, compared with those treated with tumor necrosis factor (TNF) inhibitors or conventional synthetic disease-modifying antirheumatic drugs (DMARDs).
Corticosteroids – either alone or in combination with DMARDs – were also associated with significant increases in the risk of herpes zoster. Patients treated with TNF inhibitor monotherapy had a risk of herpes zoster similar to that of those treated with conventional synthetic DMARDs or no synthetic therapy.
On the question of immunization, the authors pointed to guidelines recommending use of the live attenuated zoster vaccine (Zostavax) in immunocompetent patients or those on low-dose immunosuppression, although they noted that the vaccine is currently contraindicated for patients on biologic DMARDs.
They also examined the evidence for the use of the recently-released non-live recombinant herpes zoster vaccine (Shingrix) in immunocompromised patients, which found no evidence of vaccine-related serious adverse events in individuals with HIV and low CD4 cell counts and in autologous hematopoietic stem cell transplant recipients.
Given this, they recommended that the recombinant vaccine be administered to all patients aged over 50 years with psoriasis or psoriatic arthritis, and to those aged under 50 years who were being treated with tofacitinib, systemic corticosteroids, or combination systemic therapy.
There were insufficient data to draw conclusions about the impact of treatment with the interleukin-12/23 blocker ustekinumab (Stelara) on herpes zoster risk, but the authors noted that there was a trend toward an increased risk. They found no increase in the risk of herpes zoster with interleukin-17 inhibitors (ixekizumab [Taltz], secukinumab [Cosentyx], and brodalumab [Siliq]) and interleukin-23 (p19 subunit) inhibitors (guselkumab [Tremfya], tildrakizumab [Ilumya], and risankizumab) but noted an absence of long-term safety data for these drugs.
Four authors declared advisory, consultancy, or speaker positions with the pharmaceutical sector.
SOURCE: Baumrin E et al. J Am Acad Dermatol. 2019 March 15. doi: 10.1016/j.jaad.2019.03.017.
All individuals with psoriasis or psoriatic arthritis aged over 50 years should receive the recombinant herpes zoster vaccine, according to a systematic review and consensus recommendations from the National Psoriasis Foundation.
Emily Baumrin, MD, of Brigham and Women’s Hospital, Boston, and her coauthors reviewed 41 studies of herpes zoster in people with psoriasis or psoriatic arthritis according to treatment modality. Their report is in the Journal of the American Academy of Dermatology.
Overall, psoriasis was associated with an increased rate of herpes zoster when compared with the general population: 13.3 cases per 1,000 patient-years for psoriasis and 15.9 for psoriatic arthritis, compared with 8.5 in healthy controls after adjustment for age, sex, and systemic medications. Most of this increased incidence was seen in patients with more severe disease: Those with mild disease who were not receiving systemic therapy had a risk similar to that of healthy controls.
However, one study suggested much of the increased risk of herpes zoster in psoriasis was accounted for by immunosuppressive therapy; when those patients were excluded, there was an 8% increase in risk.
The authors found that people whose psoriasis was treated with tofacitinib (Xeljanz) had a two- to threefold increased risk of herpes zoster, compared with those treated with tumor necrosis factor (TNF) inhibitors or conventional synthetic disease-modifying antirheumatic drugs (DMARDs).
Corticosteroids – either alone or in combination with DMARDs – were also associated with significant increases in the risk of herpes zoster. Patients treated with TNF inhibitor monotherapy had a risk of herpes zoster similar to that of those treated with conventional synthetic DMARDs or no synthetic therapy.
On the question of immunization, the authors pointed to guidelines recommending use of the live attenuated zoster vaccine (Zostavax) in immunocompetent patients or those on low-dose immunosuppression, although they noted that the vaccine is currently contraindicated for patients on biologic DMARDs.
They also examined the evidence for the use of the recently-released non-live recombinant herpes zoster vaccine (Shingrix) in immunocompromised patients, which found no evidence of vaccine-related serious adverse events in individuals with HIV and low CD4 cell counts and in autologous hematopoietic stem cell transplant recipients.
Given this, they recommended that the recombinant vaccine be administered to all patients aged over 50 years with psoriasis or psoriatic arthritis, and to those aged under 50 years who were being treated with tofacitinib, systemic corticosteroids, or combination systemic therapy.
There were insufficient data to draw conclusions about the impact of treatment with the interleukin-12/23 blocker ustekinumab (Stelara) on herpes zoster risk, but the authors noted that there was a trend toward an increased risk. They found no increase in the risk of herpes zoster with interleukin-17 inhibitors (ixekizumab [Taltz], secukinumab [Cosentyx], and brodalumab [Siliq]) and interleukin-23 (p19 subunit) inhibitors (guselkumab [Tremfya], tildrakizumab [Ilumya], and risankizumab) but noted an absence of long-term safety data for these drugs.
Four authors declared advisory, consultancy, or speaker positions with the pharmaceutical sector.
SOURCE: Baumrin E et al. J Am Acad Dermatol. 2019 March 15. doi: 10.1016/j.jaad.2019.03.017.
All individuals with psoriasis or psoriatic arthritis aged over 50 years should receive the recombinant herpes zoster vaccine, according to a systematic review and consensus recommendations from the National Psoriasis Foundation.
Emily Baumrin, MD, of Brigham and Women’s Hospital, Boston, and her coauthors reviewed 41 studies of herpes zoster in people with psoriasis or psoriatic arthritis according to treatment modality. Their report is in the Journal of the American Academy of Dermatology.
Overall, psoriasis was associated with an increased rate of herpes zoster when compared with the general population: 13.3 cases per 1,000 patient-years for psoriasis and 15.9 for psoriatic arthritis, compared with 8.5 in healthy controls after adjustment for age, sex, and systemic medications. Most of this increased incidence was seen in patients with more severe disease: Those with mild disease who were not receiving systemic therapy had a risk similar to that of healthy controls.
However, one study suggested much of the increased risk of herpes zoster in psoriasis was accounted for by immunosuppressive therapy; when those patients were excluded, there was an 8% increase in risk.
The authors found that people whose psoriasis was treated with tofacitinib (Xeljanz) had a two- to threefold increased risk of herpes zoster, compared with those treated with tumor necrosis factor (TNF) inhibitors or conventional synthetic disease-modifying antirheumatic drugs (DMARDs).
Corticosteroids – either alone or in combination with DMARDs – were also associated with significant increases in the risk of herpes zoster. Patients treated with TNF inhibitor monotherapy had a risk of herpes zoster similar to that of those treated with conventional synthetic DMARDs or no synthetic therapy.
On the question of immunization, the authors pointed to guidelines recommending use of the live attenuated zoster vaccine (Zostavax) in immunocompetent patients or those on low-dose immunosuppression, although they noted that the vaccine is currently contraindicated for patients on biologic DMARDs.
They also examined the evidence for the use of the recently-released non-live recombinant herpes zoster vaccine (Shingrix) in immunocompromised patients, which found no evidence of vaccine-related serious adverse events in individuals with HIV and low CD4 cell counts and in autologous hematopoietic stem cell transplant recipients.
Given this, they recommended that the recombinant vaccine be administered to all patients aged over 50 years with psoriasis or psoriatic arthritis, and to those aged under 50 years who were being treated with tofacitinib, systemic corticosteroids, or combination systemic therapy.
There were insufficient data to draw conclusions about the impact of treatment with the interleukin-12/23 blocker ustekinumab (Stelara) on herpes zoster risk, but the authors noted that there was a trend toward an increased risk. They found no increase in the risk of herpes zoster with interleukin-17 inhibitors (ixekizumab [Taltz], secukinumab [Cosentyx], and brodalumab [Siliq]) and interleukin-23 (p19 subunit) inhibitors (guselkumab [Tremfya], tildrakizumab [Ilumya], and risankizumab) but noted an absence of long-term safety data for these drugs.
Four authors declared advisory, consultancy, or speaker positions with the pharmaceutical sector.
SOURCE: Baumrin E et al. J Am Acad Dermatol. 2019 March 15. doi: 10.1016/j.jaad.2019.03.017.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Industry-funded rheumatology RCTs are higher quality
MAUI, HAWAII – Industry-funded randomized, controlled clinical trials published in the three top-rated rheumatology journals during the past 20 years are of significantly higher overall quality than the nonindustry-funded ones, Michael Putman, MD, said at the 2019 Rheumatology Winter Clinical Symposium.
Dr. Putman, a second-year rheumatology fellow at Northwestern University, Chicago, analyzed all randomized, controlled trials (RCTs) of pharmacotherapy featuring a comparator – either placebo or an active agent – published in 1998, 2008, and 2018 in Annals of the Rheumatic Diseases, Rheumatology, and Arthritis & Rheumatology.
His main takeaway: “Rheumatologic interventions seem to work pretty well. The mean absolute risk reduction in the trials is 17.5%, so the average number of patients who need to be treated with a rheumatologic intervention is about five. This is why it’s such a great specialty to be a part of: A lot of our patients get better.”
He created an RCT quality rating scale that captured the strength of study design, methodology, and findings based upon whether a randomized trial used a double-blind design; identified a prespecified primary outcome; and featured patient-reported outcomes, power calculations, sensitivity analysis, adjustment for multiple hypotheses, and intention-to-treat analysis. He then applied the rating scale to the 85 published RCTs in the three study years.
Of note, 84% of the trials published in 2018 were industry funded, up from 74% in 2008 and 1998.
“Industry funds the vast majority of studies. Industry studies are significantly more likely to be appropriately double blinded, report patient-reported outcome measures, use intention to treat, and they have a higher overall quality,” according to Dr. Putman.
Indeed, the industry-funded studies averaged a 66% score on his quality grading scale, compared with 45% for nonindustry-funded studies.
Utilization of most of the quality metrics remained stable over time. The exceptions: Incorporation of intent-to-treat analysis increased from 58% in 1998 to 87% in 2018, and sensitivity analysis was employed in just 5% of the trials published in 1998, compared with 37% in 2008 and 26% in 2018.
The most important change over the past 2 decades, in his view, has been the shrinking proportion of RCTs featuring an active-drug, head-to-head comparator arm. In 1998, 42% of studies featured that design; for example, comparing methotrexate to sulfasalazine. By 2018, that figure had dropped to just 13%.
“Most of our trials today compare an active compound, such an interleukin-17 inhibitor, to a placebo. I think that’s a big change in how we do things,” Dr. Putman observed. “With 84% of our studies being funded by industry, the incentives in medicine right now don’t support active comparator research. It’s harder to show a difference between two things that work than it is to show a difference between something and nothing.”
However, he’d welcome a revival of head-to-head active comparator trials.
“I’d really love to have that happen,” he said. “We have basic questions we haven’t answered yet about a lot of our basic drugs: Like in myositis, should you start with Imuran [azathioprine], CellCept [mycophenolate mofetil], or methotrexate?”
Another striking change over time has been the dwindling proportion of published trials with a statistically significant finding for the primary outcome: 79% in 1998, 46% in 2008, and 36% last year. Dr. Putman suspects the explanation lies in the steady improvement in the effectiveness of standard background therapy for many conditions, which makes it tougher to show a striking difference between the add-on study drug and add-on placebo.
“We’re a victim of our own success,” he commented.
In any event, many key secondary outcomes in the RCTs were positive, even when the primary endpoint wasn’t, according to Dr. Putman, and there was a notable dearth of completely negative clinical RCTs published in the three top journals.
“The more cynical interpretation is there’s an incredible amount of publication bias, where we’re only publishing studies that show an effect and the journals or investigators are censoring the ones that don’t. The more charitable explanation, which is probably also true, is that by the time you get to putting on an RCT you kind of think, ‘This thing works.’ You’re not testing random stuff, so your pretest probability of a drug being effective when it enters into an RCT is probably shifted toward effectiveness,” Dr. Putman speculated.
He reported having no financial conflicts regarding his study.
MAUI, HAWAII – Industry-funded randomized, controlled clinical trials published in the three top-rated rheumatology journals during the past 20 years are of significantly higher overall quality than the nonindustry-funded ones, Michael Putman, MD, said at the 2019 Rheumatology Winter Clinical Symposium.
Dr. Putman, a second-year rheumatology fellow at Northwestern University, Chicago, analyzed all randomized, controlled trials (RCTs) of pharmacotherapy featuring a comparator – either placebo or an active agent – published in 1998, 2008, and 2018 in Annals of the Rheumatic Diseases, Rheumatology, and Arthritis & Rheumatology.
His main takeaway: “Rheumatologic interventions seem to work pretty well. The mean absolute risk reduction in the trials is 17.5%, so the average number of patients who need to be treated with a rheumatologic intervention is about five. This is why it’s such a great specialty to be a part of: A lot of our patients get better.”
He created an RCT quality rating scale that captured the strength of study design, methodology, and findings based upon whether a randomized trial used a double-blind design; identified a prespecified primary outcome; and featured patient-reported outcomes, power calculations, sensitivity analysis, adjustment for multiple hypotheses, and intention-to-treat analysis. He then applied the rating scale to the 85 published RCTs in the three study years.
Of note, 84% of the trials published in 2018 were industry funded, up from 74% in 2008 and 1998.
“Industry funds the vast majority of studies. Industry studies are significantly more likely to be appropriately double blinded, report patient-reported outcome measures, use intention to treat, and they have a higher overall quality,” according to Dr. Putman.
Indeed, the industry-funded studies averaged a 66% score on his quality grading scale, compared with 45% for nonindustry-funded studies.
Utilization of most of the quality metrics remained stable over time. The exceptions: Incorporation of intent-to-treat analysis increased from 58% in 1998 to 87% in 2018, and sensitivity analysis was employed in just 5% of the trials published in 1998, compared with 37% in 2008 and 26% in 2018.
The most important change over the past 2 decades, in his view, has been the shrinking proportion of RCTs featuring an active-drug, head-to-head comparator arm. In 1998, 42% of studies featured that design; for example, comparing methotrexate to sulfasalazine. By 2018, that figure had dropped to just 13%.
“Most of our trials today compare an active compound, such an interleukin-17 inhibitor, to a placebo. I think that’s a big change in how we do things,” Dr. Putman observed. “With 84% of our studies being funded by industry, the incentives in medicine right now don’t support active comparator research. It’s harder to show a difference between two things that work than it is to show a difference between something and nothing.”
However, he’d welcome a revival of head-to-head active comparator trials.
“I’d really love to have that happen,” he said. “We have basic questions we haven’t answered yet about a lot of our basic drugs: Like in myositis, should you start with Imuran [azathioprine], CellCept [mycophenolate mofetil], or methotrexate?”
Another striking change over time has been the dwindling proportion of published trials with a statistically significant finding for the primary outcome: 79% in 1998, 46% in 2008, and 36% last year. Dr. Putman suspects the explanation lies in the steady improvement in the effectiveness of standard background therapy for many conditions, which makes it tougher to show a striking difference between the add-on study drug and add-on placebo.
“We’re a victim of our own success,” he commented.
In any event, many key secondary outcomes in the RCTs were positive, even when the primary endpoint wasn’t, according to Dr. Putman, and there was a notable dearth of completely negative clinical RCTs published in the three top journals.
“The more cynical interpretation is there’s an incredible amount of publication bias, where we’re only publishing studies that show an effect and the journals or investigators are censoring the ones that don’t. The more charitable explanation, which is probably also true, is that by the time you get to putting on an RCT you kind of think, ‘This thing works.’ You’re not testing random stuff, so your pretest probability of a drug being effective when it enters into an RCT is probably shifted toward effectiveness,” Dr. Putman speculated.
He reported having no financial conflicts regarding his study.
MAUI, HAWAII – Industry-funded randomized, controlled clinical trials published in the three top-rated rheumatology journals during the past 20 years are of significantly higher overall quality than the nonindustry-funded ones, Michael Putman, MD, said at the 2019 Rheumatology Winter Clinical Symposium.
Dr. Putman, a second-year rheumatology fellow at Northwestern University, Chicago, analyzed all randomized, controlled trials (RCTs) of pharmacotherapy featuring a comparator – either placebo or an active agent – published in 1998, 2008, and 2018 in Annals of the Rheumatic Diseases, Rheumatology, and Arthritis & Rheumatology.
His main takeaway: “Rheumatologic interventions seem to work pretty well. The mean absolute risk reduction in the trials is 17.5%, so the average number of patients who need to be treated with a rheumatologic intervention is about five. This is why it’s such a great specialty to be a part of: A lot of our patients get better.”
He created an RCT quality rating scale that captured the strength of study design, methodology, and findings based upon whether a randomized trial used a double-blind design; identified a prespecified primary outcome; and featured patient-reported outcomes, power calculations, sensitivity analysis, adjustment for multiple hypotheses, and intention-to-treat analysis. He then applied the rating scale to the 85 published RCTs in the three study years.
Of note, 84% of the trials published in 2018 were industry funded, up from 74% in 2008 and 1998.
“Industry funds the vast majority of studies. Industry studies are significantly more likely to be appropriately double blinded, report patient-reported outcome measures, use intention to treat, and they have a higher overall quality,” according to Dr. Putman.
Indeed, the industry-funded studies averaged a 66% score on his quality grading scale, compared with 45% for nonindustry-funded studies.
Utilization of most of the quality metrics remained stable over time. The exceptions: Incorporation of intent-to-treat analysis increased from 58% in 1998 to 87% in 2018, and sensitivity analysis was employed in just 5% of the trials published in 1998, compared with 37% in 2008 and 26% in 2018.
The most important change over the past 2 decades, in his view, has been the shrinking proportion of RCTs featuring an active-drug, head-to-head comparator arm. In 1998, 42% of studies featured that design; for example, comparing methotrexate to sulfasalazine. By 2018, that figure had dropped to just 13%.
“Most of our trials today compare an active compound, such an interleukin-17 inhibitor, to a placebo. I think that’s a big change in how we do things,” Dr. Putman observed. “With 84% of our studies being funded by industry, the incentives in medicine right now don’t support active comparator research. It’s harder to show a difference between two things that work than it is to show a difference between something and nothing.”
However, he’d welcome a revival of head-to-head active comparator trials.
“I’d really love to have that happen,” he said. “We have basic questions we haven’t answered yet about a lot of our basic drugs: Like in myositis, should you start with Imuran [azathioprine], CellCept [mycophenolate mofetil], or methotrexate?”
Another striking change over time has been the dwindling proportion of published trials with a statistically significant finding for the primary outcome: 79% in 1998, 46% in 2008, and 36% last year. Dr. Putman suspects the explanation lies in the steady improvement in the effectiveness of standard background therapy for many conditions, which makes it tougher to show a striking difference between the add-on study drug and add-on placebo.
“We’re a victim of our own success,” he commented.
In any event, many key secondary outcomes in the RCTs were positive, even when the primary endpoint wasn’t, according to Dr. Putman, and there was a notable dearth of completely negative clinical RCTs published in the three top journals.
“The more cynical interpretation is there’s an incredible amount of publication bias, where we’re only publishing studies that show an effect and the journals or investigators are censoring the ones that don’t. The more charitable explanation, which is probably also true, is that by the time you get to putting on an RCT you kind of think, ‘This thing works.’ You’re not testing random stuff, so your pretest probability of a drug being effective when it enters into an RCT is probably shifted toward effectiveness,” Dr. Putman speculated.
He reported having no financial conflicts regarding his study.
REPORTING FROM RWCS 2019
FDA approves patient-controlled injector for guselkumab
The Food and Drug Administration has in adults, the manufacturer announced.
FDA approval is based on results from the phase 3, multicenter, randomized ORION trial, according to a press release issued by Janssen. In a Self-Injection Assessment Questionnaire, patients who received the One-Press injection rated their satisfaction with self-injection a mean score of 9.18 (0 indicated least satisfaction, 10 indicated highest satisfaction) and rated ease of use at 9.24 (10 indicated “very easy”).
In addition, 81% of patients who received One-Press achieved a Investigator’s Global Assessment score of 0 or 1, and 76% achieved a Psoriasis Area Severity Index (PASI) 90 response after 16 weeks; no patients who received the placebo achieved either.
The most common adverse event during the ORION study was injection-site reaction; the most common adverse events associated with guselkumab, an interleukin-23 blocker, include upper respiratory infections, headache, injection-site reactions, joint pain, diarrhea, gastroenteritis, fungal skin infections, and herpes simplex infections.
The Food and Drug Administration has in adults, the manufacturer announced.
FDA approval is based on results from the phase 3, multicenter, randomized ORION trial, according to a press release issued by Janssen. In a Self-Injection Assessment Questionnaire, patients who received the One-Press injection rated their satisfaction with self-injection a mean score of 9.18 (0 indicated least satisfaction, 10 indicated highest satisfaction) and rated ease of use at 9.24 (10 indicated “very easy”).
In addition, 81% of patients who received One-Press achieved a Investigator’s Global Assessment score of 0 or 1, and 76% achieved a Psoriasis Area Severity Index (PASI) 90 response after 16 weeks; no patients who received the placebo achieved either.
The most common adverse event during the ORION study was injection-site reaction; the most common adverse events associated with guselkumab, an interleukin-23 blocker, include upper respiratory infections, headache, injection-site reactions, joint pain, diarrhea, gastroenteritis, fungal skin infections, and herpes simplex infections.
The Food and Drug Administration has in adults, the manufacturer announced.
FDA approval is based on results from the phase 3, multicenter, randomized ORION trial, according to a press release issued by Janssen. In a Self-Injection Assessment Questionnaire, patients who received the One-Press injection rated their satisfaction with self-injection a mean score of 9.18 (0 indicated least satisfaction, 10 indicated highest satisfaction) and rated ease of use at 9.24 (10 indicated “very easy”).
In addition, 81% of patients who received One-Press achieved a Investigator’s Global Assessment score of 0 or 1, and 76% achieved a Psoriasis Area Severity Index (PASI) 90 response after 16 weeks; no patients who received the placebo achieved either.
The most common adverse event during the ORION study was injection-site reaction; the most common adverse events associated with guselkumab, an interleukin-23 blocker, include upper respiratory infections, headache, injection-site reactions, joint pain, diarrhea, gastroenteritis, fungal skin infections, and herpes simplex infections.
Golimumab plus methotrexate looks good in early psoriatic arthritis
For patients with early psoriatic arthritis, starting the tumor necrosis factor inhibitor golimumab (Simponi) at the same time as methotrexate nearly doubled the chances of remission, compared with methotrexate monotherapy, researchers reported in Annals of the Rheumatic Diseases.
In this multicenter, double-blind trial, 51 adults with CASPAR-defined psoriatic arthritis who were naive to methotrexate and biologic disease-modifying antirheumatic drugs were randomly assigned to receive monthly golimumab (50 mg subcutaneously) or placebo, in addition to methotrexate (15 mg/week, increased to 25 mg/week over 8 weeks). All patients had current active disease: At baseline, most had at least five swollen joints and at least nine tender joints.
Among 45 patients who completed the study, rates of Disease Activity Score (DAS) remission (DAS C-reactive protein score less than 1.6) at week 22 were 81% for golimumab-methotrexate and 42% for methotrexate-placebo (P = .004). “This difference in DAS remission was already observed at week 8,” wrote Leonieke J.J. van Mens, MD, of AMC/University of Amsterdam and her colleagues.
Golimumab-methotrexate also topped methotrexate monotherapy on secondary outcome measures. By week 22, median swollen joint counts were 0 with combined therapy versus 3 with methotrexate monotherapy (P = .04). Median tender joint counts were 0 and 2, respectively (P = .02). Combined golimumab-methotrexate therapy also produced significantly higher rates of low disease activity based on Disease Activity in Psoriatic Arthritis score (92% vs. 54%, respectively), Minimal Disease Activity (81% vs. 29%), and ACR20, 50, or 70 response (85% vs. 58%, 81% vs. 33%, and 58% vs. 13%, respectively).
Most differences were already statistically significant by week 8, and many were more pronounced by week 22, the researchers said. “It remains unknown if the responses – in particular the stringent responses such as remission – have already plateaued at week 22 or could even further increase over time,” they added. “Similarly, it remains to be determined if the combination of tumor necrosis factor inhibitor and methotrexate is only needed for the induction of remission or is also needed to maintain this state of remission over time.”
They explained that golimumab (or placebo) was stopped at week 22 in patients who achieved DAS CRP remission. An extension of the current study will assess whether methotrexate monotherapy can maintain responses for up to 50 weeks.
The only serious adverse event in the study occurred in the methotrexate arm and consisted of spinal stenosis that was not seen as treatment related. Rates of other adverse events were similar between arms, and those that required a dose halt or dose reduction were related to methotrexate, not golimumab. There were no deaths on trial.
Merck Sharp & Dohme provided medication and unrestricted funding for the study. Dr. van Mens and two coinvestigators reported having no disclosures. Several other coinvestigators disclosed ties to UCB, AbbVie, Novartis, Janssen, Eli Lilly, and other pharmaceutical companies.
SOURCE: van Mens LJJ et al. Ann Rheum Dis. 2019 Feb 26. doi: 10.1136/annrheumdis-2018-214746.
For patients with early psoriatic arthritis, starting the tumor necrosis factor inhibitor golimumab (Simponi) at the same time as methotrexate nearly doubled the chances of remission, compared with methotrexate monotherapy, researchers reported in Annals of the Rheumatic Diseases.
In this multicenter, double-blind trial, 51 adults with CASPAR-defined psoriatic arthritis who were naive to methotrexate and biologic disease-modifying antirheumatic drugs were randomly assigned to receive monthly golimumab (50 mg subcutaneously) or placebo, in addition to methotrexate (15 mg/week, increased to 25 mg/week over 8 weeks). All patients had current active disease: At baseline, most had at least five swollen joints and at least nine tender joints.
Among 45 patients who completed the study, rates of Disease Activity Score (DAS) remission (DAS C-reactive protein score less than 1.6) at week 22 were 81% for golimumab-methotrexate and 42% for methotrexate-placebo (P = .004). “This difference in DAS remission was already observed at week 8,” wrote Leonieke J.J. van Mens, MD, of AMC/University of Amsterdam and her colleagues.
Golimumab-methotrexate also topped methotrexate monotherapy on secondary outcome measures. By week 22, median swollen joint counts were 0 with combined therapy versus 3 with methotrexate monotherapy (P = .04). Median tender joint counts were 0 and 2, respectively (P = .02). Combined golimumab-methotrexate therapy also produced significantly higher rates of low disease activity based on Disease Activity in Psoriatic Arthritis score (92% vs. 54%, respectively), Minimal Disease Activity (81% vs. 29%), and ACR20, 50, or 70 response (85% vs. 58%, 81% vs. 33%, and 58% vs. 13%, respectively).
Most differences were already statistically significant by week 8, and many were more pronounced by week 22, the researchers said. “It remains unknown if the responses – in particular the stringent responses such as remission – have already plateaued at week 22 or could even further increase over time,” they added. “Similarly, it remains to be determined if the combination of tumor necrosis factor inhibitor and methotrexate is only needed for the induction of remission or is also needed to maintain this state of remission over time.”
They explained that golimumab (or placebo) was stopped at week 22 in patients who achieved DAS CRP remission. An extension of the current study will assess whether methotrexate monotherapy can maintain responses for up to 50 weeks.
The only serious adverse event in the study occurred in the methotrexate arm and consisted of spinal stenosis that was not seen as treatment related. Rates of other adverse events were similar between arms, and those that required a dose halt or dose reduction were related to methotrexate, not golimumab. There were no deaths on trial.
Merck Sharp & Dohme provided medication and unrestricted funding for the study. Dr. van Mens and two coinvestigators reported having no disclosures. Several other coinvestigators disclosed ties to UCB, AbbVie, Novartis, Janssen, Eli Lilly, and other pharmaceutical companies.
SOURCE: van Mens LJJ et al. Ann Rheum Dis. 2019 Feb 26. doi: 10.1136/annrheumdis-2018-214746.
For patients with early psoriatic arthritis, starting the tumor necrosis factor inhibitor golimumab (Simponi) at the same time as methotrexate nearly doubled the chances of remission, compared with methotrexate monotherapy, researchers reported in Annals of the Rheumatic Diseases.
In this multicenter, double-blind trial, 51 adults with CASPAR-defined psoriatic arthritis who were naive to methotrexate and biologic disease-modifying antirheumatic drugs were randomly assigned to receive monthly golimumab (50 mg subcutaneously) or placebo, in addition to methotrexate (15 mg/week, increased to 25 mg/week over 8 weeks). All patients had current active disease: At baseline, most had at least five swollen joints and at least nine tender joints.
Among 45 patients who completed the study, rates of Disease Activity Score (DAS) remission (DAS C-reactive protein score less than 1.6) at week 22 were 81% for golimumab-methotrexate and 42% for methotrexate-placebo (P = .004). “This difference in DAS remission was already observed at week 8,” wrote Leonieke J.J. van Mens, MD, of AMC/University of Amsterdam and her colleagues.
Golimumab-methotrexate also topped methotrexate monotherapy on secondary outcome measures. By week 22, median swollen joint counts were 0 with combined therapy versus 3 with methotrexate monotherapy (P = .04). Median tender joint counts were 0 and 2, respectively (P = .02). Combined golimumab-methotrexate therapy also produced significantly higher rates of low disease activity based on Disease Activity in Psoriatic Arthritis score (92% vs. 54%, respectively), Minimal Disease Activity (81% vs. 29%), and ACR20, 50, or 70 response (85% vs. 58%, 81% vs. 33%, and 58% vs. 13%, respectively).
Most differences were already statistically significant by week 8, and many were more pronounced by week 22, the researchers said. “It remains unknown if the responses – in particular the stringent responses such as remission – have already plateaued at week 22 or could even further increase over time,” they added. “Similarly, it remains to be determined if the combination of tumor necrosis factor inhibitor and methotrexate is only needed for the induction of remission or is also needed to maintain this state of remission over time.”
They explained that golimumab (or placebo) was stopped at week 22 in patients who achieved DAS CRP remission. An extension of the current study will assess whether methotrexate monotherapy can maintain responses for up to 50 weeks.
The only serious adverse event in the study occurred in the methotrexate arm and consisted of spinal stenosis that was not seen as treatment related. Rates of other adverse events were similar between arms, and those that required a dose halt or dose reduction were related to methotrexate, not golimumab. There were no deaths on trial.
Merck Sharp & Dohme provided medication and unrestricted funding for the study. Dr. van Mens and two coinvestigators reported having no disclosures. Several other coinvestigators disclosed ties to UCB, AbbVie, Novartis, Janssen, Eli Lilly, and other pharmaceutical companies.
SOURCE: van Mens LJJ et al. Ann Rheum Dis. 2019 Feb 26. doi: 10.1136/annrheumdis-2018-214746.
FROM ANNALS OF THE RHEUMATIC DISEASES
FDA: Safety signal emerged with higher dose of tofacitinib in RA study
the Food and Drug Administration reported.
The trial’s Data Safety and Monitoring Board identified the signal in patients taking a 10-mg dose of tofacitinib twice daily, the FDA said in a safety announcement.
Pfizer, the trial’s sponsor, took “immediate action” to transition patients in the ongoing trial from the 10-mg, twice-daily dose to 5 mg twice daily, which is the approved dose for adult patients with moderate to severe rheumatoid arthritis, the agency said. The 10-mg, twice-daily dose is approved only in the dosing regimen for patients with ulcerative colitis. Xeljanz is also approved to treat psoriatic arthritis. The 11-mg, once-daily dose of Xeljanz XR that is approved to treat rheumatoid arthritis and psoriatic arthritis was not tested in the trial.
The ongoing study was designed to assess risks of cardiovascular events, cancer, and opportunistic infections with tofacitinib 10 mg twice daily or 5 mg twice daily versus the risks in a control group treated with a tumor necrosis factor (TNF) inhibitor, according to the statement.
Patients had to be 50 years of age or older and have at least one cardiovascular risk factor to be eligible for the study, which was required by the agency in 2012 when it approved tofacitinib, the statement says.
The FDA is reviewing trial data and working with Pfizer to better understand the safety signal, its effect on patients, and how tofacitinib should be used, Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research, said in a news release. The trial will continue and is expected to be completed by the end of 2019.
“The agency will take appropriate action, as warranted, to ensure patients enrolled in this and other trials are protected and that health care professionals and clinical trial researchers understand the risks associated with this use,” she added.
Health care professionals should follow tofacitinib prescribing information, monitor patients for the signs and symptoms of pulmonary embolism, and advise patients to seek medical attention immediately if they experience those signs and symptoms, according to the statement.
“We are communicating now, given the serious nature of the safety issue, to ensure that patients taking tofacitinib are aware that the FDA still believes the benefits of taking tofacitinib for its approved uses continue to outweigh the risks,” Dr. Woodcock said in the release.
While not approved in rheumatoid arthritis, the 10-mg, twice-daily dose of tofacitinib is approved in the dosing regimen for patients with ulcerative colitis, the release says.
the Food and Drug Administration reported.
The trial’s Data Safety and Monitoring Board identified the signal in patients taking a 10-mg dose of tofacitinib twice daily, the FDA said in a safety announcement.
Pfizer, the trial’s sponsor, took “immediate action” to transition patients in the ongoing trial from the 10-mg, twice-daily dose to 5 mg twice daily, which is the approved dose for adult patients with moderate to severe rheumatoid arthritis, the agency said. The 10-mg, twice-daily dose is approved only in the dosing regimen for patients with ulcerative colitis. Xeljanz is also approved to treat psoriatic arthritis. The 11-mg, once-daily dose of Xeljanz XR that is approved to treat rheumatoid arthritis and psoriatic arthritis was not tested in the trial.
The ongoing study was designed to assess risks of cardiovascular events, cancer, and opportunistic infections with tofacitinib 10 mg twice daily or 5 mg twice daily versus the risks in a control group treated with a tumor necrosis factor (TNF) inhibitor, according to the statement.
Patients had to be 50 years of age or older and have at least one cardiovascular risk factor to be eligible for the study, which was required by the agency in 2012 when it approved tofacitinib, the statement says.
The FDA is reviewing trial data and working with Pfizer to better understand the safety signal, its effect on patients, and how tofacitinib should be used, Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research, said in a news release. The trial will continue and is expected to be completed by the end of 2019.
“The agency will take appropriate action, as warranted, to ensure patients enrolled in this and other trials are protected and that health care professionals and clinical trial researchers understand the risks associated with this use,” she added.
Health care professionals should follow tofacitinib prescribing information, monitor patients for the signs and symptoms of pulmonary embolism, and advise patients to seek medical attention immediately if they experience those signs and symptoms, according to the statement.
“We are communicating now, given the serious nature of the safety issue, to ensure that patients taking tofacitinib are aware that the FDA still believes the benefits of taking tofacitinib for its approved uses continue to outweigh the risks,” Dr. Woodcock said in the release.
While not approved in rheumatoid arthritis, the 10-mg, twice-daily dose of tofacitinib is approved in the dosing regimen for patients with ulcerative colitis, the release says.
the Food and Drug Administration reported.
The trial’s Data Safety and Monitoring Board identified the signal in patients taking a 10-mg dose of tofacitinib twice daily, the FDA said in a safety announcement.
Pfizer, the trial’s sponsor, took “immediate action” to transition patients in the ongoing trial from the 10-mg, twice-daily dose to 5 mg twice daily, which is the approved dose for adult patients with moderate to severe rheumatoid arthritis, the agency said. The 10-mg, twice-daily dose is approved only in the dosing regimen for patients with ulcerative colitis. Xeljanz is also approved to treat psoriatic arthritis. The 11-mg, once-daily dose of Xeljanz XR that is approved to treat rheumatoid arthritis and psoriatic arthritis was not tested in the trial.
The ongoing study was designed to assess risks of cardiovascular events, cancer, and opportunistic infections with tofacitinib 10 mg twice daily or 5 mg twice daily versus the risks in a control group treated with a tumor necrosis factor (TNF) inhibitor, according to the statement.
Patients had to be 50 years of age or older and have at least one cardiovascular risk factor to be eligible for the study, which was required by the agency in 2012 when it approved tofacitinib, the statement says.
The FDA is reviewing trial data and working with Pfizer to better understand the safety signal, its effect on patients, and how tofacitinib should be used, Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research, said in a news release. The trial will continue and is expected to be completed by the end of 2019.
“The agency will take appropriate action, as warranted, to ensure patients enrolled in this and other trials are protected and that health care professionals and clinical trial researchers understand the risks associated with this use,” she added.
Health care professionals should follow tofacitinib prescribing information, monitor patients for the signs and symptoms of pulmonary embolism, and advise patients to seek medical attention immediately if they experience those signs and symptoms, according to the statement.
“We are communicating now, given the serious nature of the safety issue, to ensure that patients taking tofacitinib are aware that the FDA still believes the benefits of taking tofacitinib for its approved uses continue to outweigh the risks,” Dr. Woodcock said in the release.
While not approved in rheumatoid arthritis, the 10-mg, twice-daily dose of tofacitinib is approved in the dosing regimen for patients with ulcerative colitis, the release says.
Ixekizumab psoriasis outcomes, sliced and diced
WAIKOLOA, HAWAII – The highly selective interleukin-17A subunit inhibitor in the long-term extension phase of the randomized, controlled UNCOVER-3 (NCT01646177) trial, Craig L. Leonardi, MD, reported at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
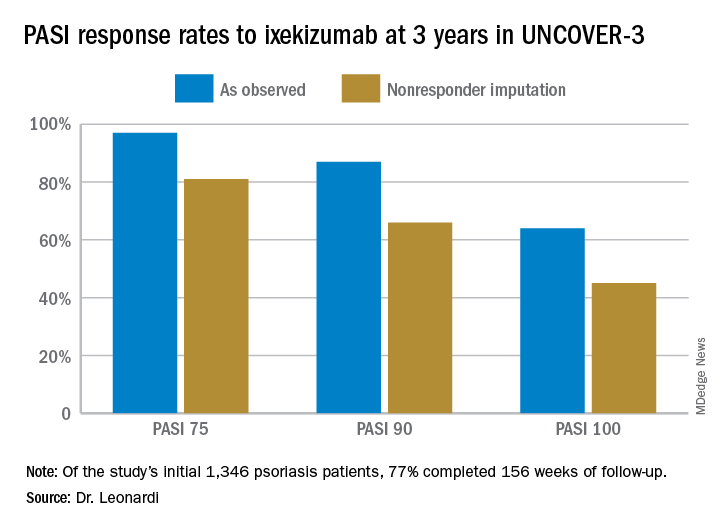
However, the strict inclusion and exclusion criteria employed in randomized trials such as this raise questions about the broader applicability of the results in real-world clinical practice. So separately at the Hawaii seminar, Dr. Leonardi presented a single-center retrospective observational cohort study of the rapidity and duration of response to ixekizumab in his own clinical practice after the biologic received Food and Drug Administration marketing approval. Those results, too, were impressive and, in his view, highly generalizable.
“It is expected that this study cohort is generally representative of patients who are routinely seen at dermatology referral practices in the U.S.,” commented Dr. Leonardi, of Saint Louis University.
UNCOVER-3 included 1,346 psoriasis patients initially randomized 2:2:2:1 to double-blind subcutaneous ixekizumab (Taltz) at 80 mg either every 2 weeks or every 4 weeks after a 160-mg loading dose; subcutaneous etanercept at 50 mg twice weekly; or placebo for 12 weeks, followed by a switch to ixekizumab at 80 mg every 4 weeks from week 12 out to 3 years. The long-term efficacy analysis was restricted to patients who received the biologic according to what ultimately became the approved dosing schedule: a 160-mg loading dose, followed by 80 mg every 2 weeks through week 12, then 80 mg every 4 weeks. The safety analysis, in contrast, included everybody.
Dr. Leonardi presented the efficacy data using several different statistical methodologies, thereby providing an instructive lesson regarding the importance of examining the fine print when viewing clinical trial results. At one extreme is the as-observed analysis. Under this methodology, if a patient dropped out of UNCOVER-3 at, for example, week 11, the last measurement of treatment response, recorded at week 8, is carried forward by investigators and assumed to be valid for the rest of the study. Since week 8 may have been the last time the patient was doing well on the drug, the as-observed analysis can create a distorted overly favorable picture of the drug’s performance.
“Patients fall out because the drug isn’t working well or they’re having a side effect, so over time, you tend to enrich for patients who are doing very well with the as-observed analysis,” the dermatologist explained.
Historically, many industry-sponsored clinical trials reported efficacy outcomes using the as-observed analysis; however, the FDA is increasingly unwilling to accept that approach as the sole analytic method.
At the other extreme is the nonresponder imputation method.
“This is the most stringent statistical package that exists. In fact, when a patient isn’t observed at one of the observation points – for example, at week 8 say the patient has a flat tire and can’t make it to the clinic – they’re counted as a treatment failure. So it’s a very tough statistical package,” according to Dr. Leonardi.
Seventy-seven percent of the initial 1,346 randomized patients in UNCOVER-3 completed 156 weeks of follow-up. To illustrate the importance of paying attention to the details of statistical methodology utilized in reporting efficacy outcomes, he noted that the PASI 75 rate at 156 weeks in study completers on the approved dosing regimen was 97% by the as-observed method, dropping to a still robust 81% by nonresponder imputation. The PASI 90 and -100 rates and static Physician’s Global Assessment (sPGA) results followed suit (see graphic).
Real-world performance
Dr. Leonard’s analysis of ixekizumab’s performance in his own practice included 106 patients placed on the drug following its FDA approval in March 2016, 74% of whom were still on the drug 12 months later. The cohort had a mean disease duration of 15 years. Three-quarters of them had previously received biologic therapy for their psoriasis, most often a tumor necrosis factor inhibitor. The study efficacy endpoints were the sPGA and Dermatology Life Quality Index (DLQI).
Already at 1 month, 30% of ixekizumab-treated patients had an sPGA score of 0, meaning their skin was totally clear. Another 29% had an sPGA of 1, meaning almost clear. At 3 months, 53% of patients had an sPGA of 0 and 21% had an sPGA of 1. Among patients on treatment at 12 months, the rates were 39% and 24% for sPGAs of 0 and 1, respectively. And in patients with an sPGA of 0/1 at 3 months, 73% maintained that score at 12 months, including 47% with an sPGA of 0.
A DLQI score of 0/1, indicative of little or no disease effect upon a patient’s life, was present in 63% of ixekizumab-treated patients at 1 month, 84% at 3 months, and 73% at 12 months.
The value in pushing for PASI 100
The ixekizumab experience in the phase-3 UNCOVER clinical trial program provided the first-ever evidence that incrementally improving psoriasis also provides stepwise improvement in DLQI, a key patient-reported outcome. At week 12 under double-blind conditions, only 4% of ixekizumab-treated patients with less than a PASI 50 response had a DLQI of 0/1. The rate rose to 18.8% in those with a PASI 50 to less than PASI 75 response. In patients with a week-12 PASI 75 to less than PASI 90 response, the DLQI 0/1 rate climbed to 52.3%. At a PASI 90 to less than PASI 100 response, the rate was 66.9%. And 82.9% of patients with a PASI 100 had a DLQI of 0/1. Every step of the way, those DLQI rates were significantly different from each other.
These data are “fascinating,” Dr. Leonardi commented. “If you ever get any inquiries from the friendly insurance carrier and they want to know if you’re improving your patient’s life, this is the kind of data that supports that they’re being improved dramatically.”
Dr. Leonardi noted that ixekizumab isn’t unique in its high rate of clinical effectiveness. That distinction is shared by the other approved IL-17 inhibitors, secukinumab (Cosentyx) and brodalumab (Siliq), as well as the IL-23 inhibitor guselkumab (Tremfya). He refers to these biologics collectively as “high-performance skin-clearance drugs.” He has calculated the number needed to treat (NNT) to achieve a PASI 100 response – complete clearance of the disease – based upon clinical trial data filed with the FDA and/or in the package inserts. The numbers are eye-opening: an NTT of 2.6 for ixekizumab based upon data from the UNCOVER-2 trial, 2.4 for brodalumab, 2.7 for guselkumab, and 3.6 for secukinumab. To help put that into perspective, the NNTs for methotrexate and etanercept (Enbrel) – not so long ago considered state of the art medications for moderate to severe psoriasis – are 25 and 23.3, respectively.
The UNCOVER trial portfolio and Dr. Leonardi’s single-center retrospective study were funded by Eli Lilly, which markets ixekizumab. He reported serving as a consultant to and receiving research funding from that company and more than a dozen others.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The highly selective interleukin-17A subunit inhibitor in the long-term extension phase of the randomized, controlled UNCOVER-3 (NCT01646177) trial, Craig L. Leonardi, MD, reported at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.

However, the strict inclusion and exclusion criteria employed in randomized trials such as this raise questions about the broader applicability of the results in real-world clinical practice. So separately at the Hawaii seminar, Dr. Leonardi presented a single-center retrospective observational cohort study of the rapidity and duration of response to ixekizumab in his own clinical practice after the biologic received Food and Drug Administration marketing approval. Those results, too, were impressive and, in his view, highly generalizable.
“It is expected that this study cohort is generally representative of patients who are routinely seen at dermatology referral practices in the U.S.,” commented Dr. Leonardi, of Saint Louis University.
UNCOVER-3 included 1,346 psoriasis patients initially randomized 2:2:2:1 to double-blind subcutaneous ixekizumab (Taltz) at 80 mg either every 2 weeks or every 4 weeks after a 160-mg loading dose; subcutaneous etanercept at 50 mg twice weekly; or placebo for 12 weeks, followed by a switch to ixekizumab at 80 mg every 4 weeks from week 12 out to 3 years. The long-term efficacy analysis was restricted to patients who received the biologic according to what ultimately became the approved dosing schedule: a 160-mg loading dose, followed by 80 mg every 2 weeks through week 12, then 80 mg every 4 weeks. The safety analysis, in contrast, included everybody.
Dr. Leonardi presented the efficacy data using several different statistical methodologies, thereby providing an instructive lesson regarding the importance of examining the fine print when viewing clinical trial results. At one extreme is the as-observed analysis. Under this methodology, if a patient dropped out of UNCOVER-3 at, for example, week 11, the last measurement of treatment response, recorded at week 8, is carried forward by investigators and assumed to be valid for the rest of the study. Since week 8 may have been the last time the patient was doing well on the drug, the as-observed analysis can create a distorted overly favorable picture of the drug’s performance.
“Patients fall out because the drug isn’t working well or they’re having a side effect, so over time, you tend to enrich for patients who are doing very well with the as-observed analysis,” the dermatologist explained.
Historically, many industry-sponsored clinical trials reported efficacy outcomes using the as-observed analysis; however, the FDA is increasingly unwilling to accept that approach as the sole analytic method.
At the other extreme is the nonresponder imputation method.
“This is the most stringent statistical package that exists. In fact, when a patient isn’t observed at one of the observation points – for example, at week 8 say the patient has a flat tire and can’t make it to the clinic – they’re counted as a treatment failure. So it’s a very tough statistical package,” according to Dr. Leonardi.
Seventy-seven percent of the initial 1,346 randomized patients in UNCOVER-3 completed 156 weeks of follow-up. To illustrate the importance of paying attention to the details of statistical methodology utilized in reporting efficacy outcomes, he noted that the PASI 75 rate at 156 weeks in study completers on the approved dosing regimen was 97% by the as-observed method, dropping to a still robust 81% by nonresponder imputation. The PASI 90 and -100 rates and static Physician’s Global Assessment (sPGA) results followed suit (see graphic).
Real-world performance
Dr. Leonard’s analysis of ixekizumab’s performance in his own practice included 106 patients placed on the drug following its FDA approval in March 2016, 74% of whom were still on the drug 12 months later. The cohort had a mean disease duration of 15 years. Three-quarters of them had previously received biologic therapy for their psoriasis, most often a tumor necrosis factor inhibitor. The study efficacy endpoints were the sPGA and Dermatology Life Quality Index (DLQI).
Already at 1 month, 30% of ixekizumab-treated patients had an sPGA score of 0, meaning their skin was totally clear. Another 29% had an sPGA of 1, meaning almost clear. At 3 months, 53% of patients had an sPGA of 0 and 21% had an sPGA of 1. Among patients on treatment at 12 months, the rates were 39% and 24% for sPGAs of 0 and 1, respectively. And in patients with an sPGA of 0/1 at 3 months, 73% maintained that score at 12 months, including 47% with an sPGA of 0.
A DLQI score of 0/1, indicative of little or no disease effect upon a patient’s life, was present in 63% of ixekizumab-treated patients at 1 month, 84% at 3 months, and 73% at 12 months.
The value in pushing for PASI 100
The ixekizumab experience in the phase-3 UNCOVER clinical trial program provided the first-ever evidence that incrementally improving psoriasis also provides stepwise improvement in DLQI, a key patient-reported outcome. At week 12 under double-blind conditions, only 4% of ixekizumab-treated patients with less than a PASI 50 response had a DLQI of 0/1. The rate rose to 18.8% in those with a PASI 50 to less than PASI 75 response. In patients with a week-12 PASI 75 to less than PASI 90 response, the DLQI 0/1 rate climbed to 52.3%. At a PASI 90 to less than PASI 100 response, the rate was 66.9%. And 82.9% of patients with a PASI 100 had a DLQI of 0/1. Every step of the way, those DLQI rates were significantly different from each other.
These data are “fascinating,” Dr. Leonardi commented. “If you ever get any inquiries from the friendly insurance carrier and they want to know if you’re improving your patient’s life, this is the kind of data that supports that they’re being improved dramatically.”
Dr. Leonardi noted that ixekizumab isn’t unique in its high rate of clinical effectiveness. That distinction is shared by the other approved IL-17 inhibitors, secukinumab (Cosentyx) and brodalumab (Siliq), as well as the IL-23 inhibitor guselkumab (Tremfya). He refers to these biologics collectively as “high-performance skin-clearance drugs.” He has calculated the number needed to treat (NNT) to achieve a PASI 100 response – complete clearance of the disease – based upon clinical trial data filed with the FDA and/or in the package inserts. The numbers are eye-opening: an NTT of 2.6 for ixekizumab based upon data from the UNCOVER-2 trial, 2.4 for brodalumab, 2.7 for guselkumab, and 3.6 for secukinumab. To help put that into perspective, the NNTs for methotrexate and etanercept (Enbrel) – not so long ago considered state of the art medications for moderate to severe psoriasis – are 25 and 23.3, respectively.
The UNCOVER trial portfolio and Dr. Leonardi’s single-center retrospective study were funded by Eli Lilly, which markets ixekizumab. He reported serving as a consultant to and receiving research funding from that company and more than a dozen others.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The highly selective interleukin-17A subunit inhibitor in the long-term extension phase of the randomized, controlled UNCOVER-3 (NCT01646177) trial, Craig L. Leonardi, MD, reported at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.

However, the strict inclusion and exclusion criteria employed in randomized trials such as this raise questions about the broader applicability of the results in real-world clinical practice. So separately at the Hawaii seminar, Dr. Leonardi presented a single-center retrospective observational cohort study of the rapidity and duration of response to ixekizumab in his own clinical practice after the biologic received Food and Drug Administration marketing approval. Those results, too, were impressive and, in his view, highly generalizable.
“It is expected that this study cohort is generally representative of patients who are routinely seen at dermatology referral practices in the U.S.,” commented Dr. Leonardi, of Saint Louis University.
UNCOVER-3 included 1,346 psoriasis patients initially randomized 2:2:2:1 to double-blind subcutaneous ixekizumab (Taltz) at 80 mg either every 2 weeks or every 4 weeks after a 160-mg loading dose; subcutaneous etanercept at 50 mg twice weekly; or placebo for 12 weeks, followed by a switch to ixekizumab at 80 mg every 4 weeks from week 12 out to 3 years. The long-term efficacy analysis was restricted to patients who received the biologic according to what ultimately became the approved dosing schedule: a 160-mg loading dose, followed by 80 mg every 2 weeks through week 12, then 80 mg every 4 weeks. The safety analysis, in contrast, included everybody.
Dr. Leonardi presented the efficacy data using several different statistical methodologies, thereby providing an instructive lesson regarding the importance of examining the fine print when viewing clinical trial results. At one extreme is the as-observed analysis. Under this methodology, if a patient dropped out of UNCOVER-3 at, for example, week 11, the last measurement of treatment response, recorded at week 8, is carried forward by investigators and assumed to be valid for the rest of the study. Since week 8 may have been the last time the patient was doing well on the drug, the as-observed analysis can create a distorted overly favorable picture of the drug’s performance.
“Patients fall out because the drug isn’t working well or they’re having a side effect, so over time, you tend to enrich for patients who are doing very well with the as-observed analysis,” the dermatologist explained.
Historically, many industry-sponsored clinical trials reported efficacy outcomes using the as-observed analysis; however, the FDA is increasingly unwilling to accept that approach as the sole analytic method.
At the other extreme is the nonresponder imputation method.
“This is the most stringent statistical package that exists. In fact, when a patient isn’t observed at one of the observation points – for example, at week 8 say the patient has a flat tire and can’t make it to the clinic – they’re counted as a treatment failure. So it’s a very tough statistical package,” according to Dr. Leonardi.
Seventy-seven percent of the initial 1,346 randomized patients in UNCOVER-3 completed 156 weeks of follow-up. To illustrate the importance of paying attention to the details of statistical methodology utilized in reporting efficacy outcomes, he noted that the PASI 75 rate at 156 weeks in study completers on the approved dosing regimen was 97% by the as-observed method, dropping to a still robust 81% by nonresponder imputation. The PASI 90 and -100 rates and static Physician’s Global Assessment (sPGA) results followed suit (see graphic).
Real-world performance
Dr. Leonard’s analysis of ixekizumab’s performance in his own practice included 106 patients placed on the drug following its FDA approval in March 2016, 74% of whom were still on the drug 12 months later. The cohort had a mean disease duration of 15 years. Three-quarters of them had previously received biologic therapy for their psoriasis, most often a tumor necrosis factor inhibitor. The study efficacy endpoints were the sPGA and Dermatology Life Quality Index (DLQI).
Already at 1 month, 30% of ixekizumab-treated patients had an sPGA score of 0, meaning their skin was totally clear. Another 29% had an sPGA of 1, meaning almost clear. At 3 months, 53% of patients had an sPGA of 0 and 21% had an sPGA of 1. Among patients on treatment at 12 months, the rates were 39% and 24% for sPGAs of 0 and 1, respectively. And in patients with an sPGA of 0/1 at 3 months, 73% maintained that score at 12 months, including 47% with an sPGA of 0.
A DLQI score of 0/1, indicative of little or no disease effect upon a patient’s life, was present in 63% of ixekizumab-treated patients at 1 month, 84% at 3 months, and 73% at 12 months.
The value in pushing for PASI 100
The ixekizumab experience in the phase-3 UNCOVER clinical trial program provided the first-ever evidence that incrementally improving psoriasis also provides stepwise improvement in DLQI, a key patient-reported outcome. At week 12 under double-blind conditions, only 4% of ixekizumab-treated patients with less than a PASI 50 response had a DLQI of 0/1. The rate rose to 18.8% in those with a PASI 50 to less than PASI 75 response. In patients with a week-12 PASI 75 to less than PASI 90 response, the DLQI 0/1 rate climbed to 52.3%. At a PASI 90 to less than PASI 100 response, the rate was 66.9%. And 82.9% of patients with a PASI 100 had a DLQI of 0/1. Every step of the way, those DLQI rates were significantly different from each other.
These data are “fascinating,” Dr. Leonardi commented. “If you ever get any inquiries from the friendly insurance carrier and they want to know if you’re improving your patient’s life, this is the kind of data that supports that they’re being improved dramatically.”
Dr. Leonardi noted that ixekizumab isn’t unique in its high rate of clinical effectiveness. That distinction is shared by the other approved IL-17 inhibitors, secukinumab (Cosentyx) and brodalumab (Siliq), as well as the IL-23 inhibitor guselkumab (Tremfya). He refers to these biologics collectively as “high-performance skin-clearance drugs.” He has calculated the number needed to treat (NNT) to achieve a PASI 100 response – complete clearance of the disease – based upon clinical trial data filed with the FDA and/or in the package inserts. The numbers are eye-opening: an NTT of 2.6 for ixekizumab based upon data from the UNCOVER-2 trial, 2.4 for brodalumab, 2.7 for guselkumab, and 3.6 for secukinumab. To help put that into perspective, the NNTs for methotrexate and etanercept (Enbrel) – not so long ago considered state of the art medications for moderate to severe psoriasis – are 25 and 23.3, respectively.
The UNCOVER trial portfolio and Dr. Leonardi’s single-center retrospective study were funded by Eli Lilly, which markets ixekizumab. He reported serving as a consultant to and receiving research funding from that company and more than a dozen others.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
REPORTING FROM SDEF HAWAII DERMATOLOGY SEMINAR
AAD, NPF release two joint guidelines on treatment, management of psoriasis
The .
These guidelines are the first of two papers to be published in the Journal of the American Academy of Dermatology (JAAD), with four more guidelines on psoriasis to be published later this year in JAAD on phototherapy, topical therapy, nonbiologic systemic medications, and treatment of pediatric patients.
The guideline on biologics updates the 2008 AAD guidelines on psoriasis. In an interview, Alan Menter, MD, cochair of the guidelines work group and lead author of the biologics paper, said the guidelines for biologics were needed because of major advances with the availability of new biologics over the last decade. For example, three tumor necrosis factor–alpha (TNF-alpha) inhibitors were available in 2008, but that number has increased to 10 biologics and now includes agents such as those targeting interleukin (IL)-12/IL-23, IL-17 and IL-23.
In addition, the new guidelines from AAD were developed to represent improvements in the management of patients with moderate to severe psoriasis as well as the relationship between psoriasis and related comorbidities.
“Major advances in new biologic drugs [are] now available to patients, plus [there have been] significant advances in our understanding of comorbid conditions,” such as cardiovascular comorbidities, said Dr. Menter, chairman of the division of dermatology, Baylor University Medical Center, and clinical professor of dermatology, University of Texas, both in Dallas.
The working group for each set of guidelines consisted of dermatologists, patient representatives, a cardiologist, and a rheumatologist. The biologic guidelines working group analyzed studies published between January 2008 and December 2018 and issued a series of recommendations based on published evidence for the effectiveness, adverse events, and switching for Food and Drug Administration–approved TNF-alpha inhibitors (etanercept, infliximab, adalimumab, certolizumab, and TNF-alpha biosimilars); IL-12/IL-23 inhibitors (ustekinumab); IL-17 inhibitors (secukinumab, ixekizumab, and brodalumab); and IL-23 inhibitors (guselkumab and tildrakizumab, and risankizumab, which is still under FDA review) for monotherapy or combination therapy in patients with moderate to severe psoriasis.
The biologic guidelines noted that, while FDA-approved biologics were deemed safe overall for patients with moderate to severe psoriasis, dermatologists should recognize the adverse effects of these therapies, monitor for infections, and counsel their patients against modifying or discontinuing therapy without first consulting a dermatologist. In general, the working group noted that failure with one biologic does not necessarily mean that a patient will experience failure with a different biologic, even among TNF-alpha and IL-12/IL-23 inhibitors. However, reduced efficacy for a patient receiving a specific TNF-alpha inhibitor may predict reduced efficacy when switching to a different TNF-alpha inhibitor, they said.
In the psoriasis comorbidity guideline, the working group examined the therapeutic interventions for psoriasis-related comorbidities such as psoriatic arthritis (PsA), cardiovascular disease, metabolic syndrome, and inflammatory bowel disease. They also provided recommendations on the effect of psoriasis on mental health, quality of life, and lifestyle choices such as smoking and alcohol use.
With respect to cardiovascular disease, the dermatologist should ensure that patients are aware of the association between risk factors for cardiovascular disease and psoriasis, and that they undergo screening for these risk factors, consider lifestyle changes to reduce risk of cardiovascular disease, and consult with cardiologists and primary care providers based on individual risk, the guideline states. The working group recommended that patients with psoriasis undergo screening for hypertension, diabetes, and hyperlipidemia based on national guidelines, with more frequent screening recommended for patients with psoriasis greater than 10% body surface area or who are eligible for systemic or phototherapy.
In both the biologic and the comorbidity guidelines, the working groups stressed the importance of patient education and the role of the dermatologist in educating patients so that shared decision-making can occur. They noted that education was related to improved quality of life for these patients.
“Both the comorbidities guidelines and the biologic guidelines will help educate the psoriasis population with input from dermatologists in clinical practices,” Dr. Menter said.
However, both working groups noted there are still significant gaps in research, such as the effects of treatment combinations for new biologics and the lack of biomarkers that would identify which biologics are best suited for individual psoriasis patients.
There is also little known about the complex relationship between psoriasis and its comorbidities, and how psoriasis treatment can potentially prevent future disease. To ensure treatment of psoriasis-related comorbidities, dermatologists should consider psoriasis as a systemic disease with multiple comorbidities and interact with primary care doctors, cardiologists, and other providers involved in the care of the patients, Dr. Menter said.
There were no specific funding sources reported for the guidelines. Several authors reported relationships with industry, including pharmaceutical companies with drugs and products involving psoriasis, during the development of the guidelines. If a potential conflict was noted, the working group member recused himself or herself from discussion and drafting of recommendations, according to the paper. Dr. Menter’s disclosure includes serving as a consultant, speaker, investigator, and adviser, and receiving honoraria, from multiple pharmaceutical companies.
SOURCE: Menter A et al. J Am Acad Dermatol. 2019 Feb 13. doi: 10.1016/j.jaad.2018.11.057. Elmets CA et al. J Am Acad Dermatol. 2019 Feb 13. doi: 10.1016/j.jaad.2018.11.058.
The .
These guidelines are the first of two papers to be published in the Journal of the American Academy of Dermatology (JAAD), with four more guidelines on psoriasis to be published later this year in JAAD on phototherapy, topical therapy, nonbiologic systemic medications, and treatment of pediatric patients.
The guideline on biologics updates the 2008 AAD guidelines on psoriasis. In an interview, Alan Menter, MD, cochair of the guidelines work group and lead author of the biologics paper, said the guidelines for biologics were needed because of major advances with the availability of new biologics over the last decade. For example, three tumor necrosis factor–alpha (TNF-alpha) inhibitors were available in 2008, but that number has increased to 10 biologics and now includes agents such as those targeting interleukin (IL)-12/IL-23, IL-17 and IL-23.
In addition, the new guidelines from AAD were developed to represent improvements in the management of patients with moderate to severe psoriasis as well as the relationship between psoriasis and related comorbidities.
“Major advances in new biologic drugs [are] now available to patients, plus [there have been] significant advances in our understanding of comorbid conditions,” such as cardiovascular comorbidities, said Dr. Menter, chairman of the division of dermatology, Baylor University Medical Center, and clinical professor of dermatology, University of Texas, both in Dallas.
The working group for each set of guidelines consisted of dermatologists, patient representatives, a cardiologist, and a rheumatologist. The biologic guidelines working group analyzed studies published between January 2008 and December 2018 and issued a series of recommendations based on published evidence for the effectiveness, adverse events, and switching for Food and Drug Administration–approved TNF-alpha inhibitors (etanercept, infliximab, adalimumab, certolizumab, and TNF-alpha biosimilars); IL-12/IL-23 inhibitors (ustekinumab); IL-17 inhibitors (secukinumab, ixekizumab, and brodalumab); and IL-23 inhibitors (guselkumab and tildrakizumab, and risankizumab, which is still under FDA review) for monotherapy or combination therapy in patients with moderate to severe psoriasis.
The biologic guidelines noted that, while FDA-approved biologics were deemed safe overall for patients with moderate to severe psoriasis, dermatologists should recognize the adverse effects of these therapies, monitor for infections, and counsel their patients against modifying or discontinuing therapy without first consulting a dermatologist. In general, the working group noted that failure with one biologic does not necessarily mean that a patient will experience failure with a different biologic, even among TNF-alpha and IL-12/IL-23 inhibitors. However, reduced efficacy for a patient receiving a specific TNF-alpha inhibitor may predict reduced efficacy when switching to a different TNF-alpha inhibitor, they said.
In the psoriasis comorbidity guideline, the working group examined the therapeutic interventions for psoriasis-related comorbidities such as psoriatic arthritis (PsA), cardiovascular disease, metabolic syndrome, and inflammatory bowel disease. They also provided recommendations on the effect of psoriasis on mental health, quality of life, and lifestyle choices such as smoking and alcohol use.
With respect to cardiovascular disease, the dermatologist should ensure that patients are aware of the association between risk factors for cardiovascular disease and psoriasis, and that they undergo screening for these risk factors, consider lifestyle changes to reduce risk of cardiovascular disease, and consult with cardiologists and primary care providers based on individual risk, the guideline states. The working group recommended that patients with psoriasis undergo screening for hypertension, diabetes, and hyperlipidemia based on national guidelines, with more frequent screening recommended for patients with psoriasis greater than 10% body surface area or who are eligible for systemic or phototherapy.
In both the biologic and the comorbidity guidelines, the working groups stressed the importance of patient education and the role of the dermatologist in educating patients so that shared decision-making can occur. They noted that education was related to improved quality of life for these patients.
“Both the comorbidities guidelines and the biologic guidelines will help educate the psoriasis population with input from dermatologists in clinical practices,” Dr. Menter said.
However, both working groups noted there are still significant gaps in research, such as the effects of treatment combinations for new biologics and the lack of biomarkers that would identify which biologics are best suited for individual psoriasis patients.
There is also little known about the complex relationship between psoriasis and its comorbidities, and how psoriasis treatment can potentially prevent future disease. To ensure treatment of psoriasis-related comorbidities, dermatologists should consider psoriasis as a systemic disease with multiple comorbidities and interact with primary care doctors, cardiologists, and other providers involved in the care of the patients, Dr. Menter said.
There were no specific funding sources reported for the guidelines. Several authors reported relationships with industry, including pharmaceutical companies with drugs and products involving psoriasis, during the development of the guidelines. If a potential conflict was noted, the working group member recused himself or herself from discussion and drafting of recommendations, according to the paper. Dr. Menter’s disclosure includes serving as a consultant, speaker, investigator, and adviser, and receiving honoraria, from multiple pharmaceutical companies.
SOURCE: Menter A et al. J Am Acad Dermatol. 2019 Feb 13. doi: 10.1016/j.jaad.2018.11.057. Elmets CA et al. J Am Acad Dermatol. 2019 Feb 13. doi: 10.1016/j.jaad.2018.11.058.
The .
These guidelines are the first of two papers to be published in the Journal of the American Academy of Dermatology (JAAD), with four more guidelines on psoriasis to be published later this year in JAAD on phototherapy, topical therapy, nonbiologic systemic medications, and treatment of pediatric patients.
The guideline on biologics updates the 2008 AAD guidelines on psoriasis. In an interview, Alan Menter, MD, cochair of the guidelines work group and lead author of the biologics paper, said the guidelines for biologics were needed because of major advances with the availability of new biologics over the last decade. For example, three tumor necrosis factor–alpha (TNF-alpha) inhibitors were available in 2008, but that number has increased to 10 biologics and now includes agents such as those targeting interleukin (IL)-12/IL-23, IL-17 and IL-23.
In addition, the new guidelines from AAD were developed to represent improvements in the management of patients with moderate to severe psoriasis as well as the relationship between psoriasis and related comorbidities.
“Major advances in new biologic drugs [are] now available to patients, plus [there have been] significant advances in our understanding of comorbid conditions,” such as cardiovascular comorbidities, said Dr. Menter, chairman of the division of dermatology, Baylor University Medical Center, and clinical professor of dermatology, University of Texas, both in Dallas.
The working group for each set of guidelines consisted of dermatologists, patient representatives, a cardiologist, and a rheumatologist. The biologic guidelines working group analyzed studies published between January 2008 and December 2018 and issued a series of recommendations based on published evidence for the effectiveness, adverse events, and switching for Food and Drug Administration–approved TNF-alpha inhibitors (etanercept, infliximab, adalimumab, certolizumab, and TNF-alpha biosimilars); IL-12/IL-23 inhibitors (ustekinumab); IL-17 inhibitors (secukinumab, ixekizumab, and brodalumab); and IL-23 inhibitors (guselkumab and tildrakizumab, and risankizumab, which is still under FDA review) for monotherapy or combination therapy in patients with moderate to severe psoriasis.
The biologic guidelines noted that, while FDA-approved biologics were deemed safe overall for patients with moderate to severe psoriasis, dermatologists should recognize the adverse effects of these therapies, monitor for infections, and counsel their patients against modifying or discontinuing therapy without first consulting a dermatologist. In general, the working group noted that failure with one biologic does not necessarily mean that a patient will experience failure with a different biologic, even among TNF-alpha and IL-12/IL-23 inhibitors. However, reduced efficacy for a patient receiving a specific TNF-alpha inhibitor may predict reduced efficacy when switching to a different TNF-alpha inhibitor, they said.
In the psoriasis comorbidity guideline, the working group examined the therapeutic interventions for psoriasis-related comorbidities such as psoriatic arthritis (PsA), cardiovascular disease, metabolic syndrome, and inflammatory bowel disease. They also provided recommendations on the effect of psoriasis on mental health, quality of life, and lifestyle choices such as smoking and alcohol use.
With respect to cardiovascular disease, the dermatologist should ensure that patients are aware of the association between risk factors for cardiovascular disease and psoriasis, and that they undergo screening for these risk factors, consider lifestyle changes to reduce risk of cardiovascular disease, and consult with cardiologists and primary care providers based on individual risk, the guideline states. The working group recommended that patients with psoriasis undergo screening for hypertension, diabetes, and hyperlipidemia based on national guidelines, with more frequent screening recommended for patients with psoriasis greater than 10% body surface area or who are eligible for systemic or phototherapy.
In both the biologic and the comorbidity guidelines, the working groups stressed the importance of patient education and the role of the dermatologist in educating patients so that shared decision-making can occur. They noted that education was related to improved quality of life for these patients.
“Both the comorbidities guidelines and the biologic guidelines will help educate the psoriasis population with input from dermatologists in clinical practices,” Dr. Menter said.
However, both working groups noted there are still significant gaps in research, such as the effects of treatment combinations for new biologics and the lack of biomarkers that would identify which biologics are best suited for individual psoriasis patients.
There is also little known about the complex relationship between psoriasis and its comorbidities, and how psoriasis treatment can potentially prevent future disease. To ensure treatment of psoriasis-related comorbidities, dermatologists should consider psoriasis as a systemic disease with multiple comorbidities and interact with primary care doctors, cardiologists, and other providers involved in the care of the patients, Dr. Menter said.
There were no specific funding sources reported for the guidelines. Several authors reported relationships with industry, including pharmaceutical companies with drugs and products involving psoriasis, during the development of the guidelines. If a potential conflict was noted, the working group member recused himself or herself from discussion and drafting of recommendations, according to the paper. Dr. Menter’s disclosure includes serving as a consultant, speaker, investigator, and adviser, and receiving honoraria, from multiple pharmaceutical companies.
SOURCE: Menter A et al. J Am Acad Dermatol. 2019 Feb 13. doi: 10.1016/j.jaad.2018.11.057. Elmets CA et al. J Am Acad Dermatol. 2019 Feb 13. doi: 10.1016/j.jaad.2018.11.058.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
What’s new with adalimumab? Plenty
WAIKOLOA, HAWAII – A flurry of recent impressive , identifies a simple biomarker predictive of the likelihood of a favorable PASI 75 response, and highlights a disconnect in psoriatic arthritis (PsA) patients between clinical response as reflected in disease activity and radiographic progression of joint disease, according to Kristina C. Duffin, MD.
Also, a new citrate-free version of adalimumab (Humira) is available. It requires a new prescription, and an additional prior authorization is mandated by some insurers. But this is a welcome innovation for patients bothered by significant burning and stinging with their injections of classic adalimumab, Dr. Duffin, cochair of the department of dermatology at the University of Utah, Salt Lake City, said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
New long-term safety data
Adalimumab is a market leader in biologic therapy for psoriasis. But the long-term experience with biologics in dermatology is still relatively limited, so the recent publication of two large studies providing encouraging evidence of the long-term safety of adalimumab is noteworthy.
Craig L. Leonardi, MD, of Saint Louis University, St. Louis, Mo., was first author of an analysis of long-term safety data from 18 clinical trials in adults with moderate to severe plaque psoriasis. The key takeaway, in Dr. Duffin’s view, was that the rate of adverse events, including serious infections and malignancies other than nonmelanoma skin cancer, remained stable over time out to 240 weeks of follow-up in patients on continuous treatment, with no new safety signals emerging (Br J Dermatol. 2019 Jan;180[1]:76-85).
However, randomized clinical trials often paint an overly rosy safety picture because of their strict inclusion and exclusion criteria.
“We single out patients for clinical trials because they’re especially healthy. That doesn’t happen in real-world registries,” she noted.
That’s why a systematic review of adalimumab’s safety performance in 10 real-world registries of adalimumab-treated psoriasis patients is particularly informative. The registries included in the systematic review, led by Bruce E. Strober, MD, PhD, professor of dermatology at the University of Connecticut, Farmington, didn’t all measure the same outcomes. But the three registries that documented major adverse cardiovascular events showed rates of less than 0.1 to less than 1 per 100 patient-years. Rates of malignancies other than nonmelanoma skin cancer were consistently in the 0.3-0.6 events per 100 patient-years range, similar to what has been reported in studies of other systemic psoriasis therapies, biologic as well as nonbiologic (J Eur Acad Dermatol Venereol. 2018 Dec;32[12]:2126-33).
Overall infection rates reported in the real-world registries ranged from 7.7 to 14.7 events per 100 patient-years, which is actually considerably lower than in the clinical trials. Rates of serious infections ranged from less than one up to two events per 100 patient-years, with the most common ones being cellulitis and pneumonia, consistent with the randomized trial experience.
Predicting response to adalimumab
A prospective, multicenter, observational cohort study of 544 psoriasis patients on adalimumab monotherapy conducted by U.K. investigators concluded that a patient’s serum drug level is the single most important predictor of treatment response. A cut point of 3.2 mcg/mL, which is considered the minimal effective circulating drug level, was associated with a 65% probability of a 75% improvement in Psoriasis Area and Severity Index from baseline, or PASI 75 response. The higher the serum drug level, the greater the likelihood of a PASI 75 response, up to a serum level of 7 mcg/mL, which was associated with an 81% probability of achieving PASI 75. Beyond 7 mcg/mL, however, the relationship with treatment response plateaued. Importantly, drug levels measured early on – at 1-12 weeks into therapy – were predictive of response 6 months later. So were steady-state levels (J Invest Dermatol. 2019 Jan;139[1]:115-23).
This is clinically useful information, Dr. Duffin observed.
“I’m hoping we’re going to see more real-world use of checking drug levels,” she said.
Indeed, even though the approved dosing of adalimumab for psoriasis is 40 mg by subcutaneous injection every 2 weeks, the new American Academy of Dermatology/National Psoriasis Foundation joint guidelines for treatment of psoriasis with biologics declare that “a maintenance dose of adalimumab at 40 mg/week is recommended for better disease control in some patients” (J Am Acad Dermatol. 2019 Feb 7. doi: 10.1016/j.jaad.2018.11.057. [Epub ahead of print]).
The new guidelines provide support for dermatologists who decide weekly therapy is best for a given patient, and adalimumab drug levels could prove useful in identifying the patient subgroup likely to benefit.
Dr. Duffin is often consulted by other physicians as to whether they should check for neutralizing antibodies in patients who appear to be losing therapeutic efficacy on a given biologic. She’s not a fan of the practice.
“There are commercial assays out there, but it’s very hard to interpret them because we don’t really know if they’re truly measuring neutralizing antibodies. And the cost is not insignificant; it can be hundreds of dollars,” she noted.
She believes a straightforward measurement of the serum biologic level is a better strategy.
“It makes sense: This is an indirect way of determining if there’s been neutralization of the drug, rather than trying to check the antibody that’s doing it, which is fraught with problems,” Dr. Duffin said.
Radiographic progression and clinical PsA activity on adalimumab don’t always correlate
A post hoc analysis of the randomized, double-blind, placebo-controlled ADEPT trial in PsA patients demonstrated that inhibition of radiographic progression as measured by change in modified total Sharp score from baseline through 24 weeks of adalimumab therapy was greater than expected based upon control of clinical disease activity (Rheumatology [Oxford]. 2019 Jan 3. doi: 10.1093/rheumatology/key417. [Epub ahead of print]).
One implication of the disconnect between radiographic progression and clinical disease documented in this study is that a dermatologist shouldn’t be too quick to change from adalimumab to another biologic just because a patient with PsA reports continued but bearable joint pain. And the converse is also true.
“I think that we as dermatologists probably shouldn’t be reassured when a patient says, ‘My joints feel great!” That’s because you may not necessarily be able to predict lack of progression in Sharp score based upon clinical response,” Dr. Duffin cautioned. “I think you should still have a rheumatologist check in with the patient and do x-rays periodically. The rheumatologist I work with does that, usually about on a yearly basis.”
Another key finding in the ADEPT analysis was that concomitant methotrexate had no added effect in terms of preventing joint destruction. This underscores the prescience of the first-ever collaborative American College of Rheumatology/National Psoriasis Foundation guidelines for the treatment of PsA (Arthritis Care Res (Hoboken). 2019 Jan;71[1]:2-29).
The new guidelines recommend that, in a psoriasis patient with confirmed PsA, the first-line treatment is a tumor necrosis factor (TNF) inhibitor. Agents from this class are preferred over other biologics because they are backed by a larger body of data regarding inhibition of joint disease progression. If the patient fails on the first TNF inhibitor prescribed, second-line therapy is another TNF inhibitor. So is third-line therapy.
Adalimumab citrate free
Not only does this new iteration of adalimumab do away with citrate as a buffer because it can cause pain and burning, it also utilizes a thinner 29-gauge needle rather than the standard 27-gauge. And the needle cover isn’t made with natural rubber latex. Also, both the pen and prefilled syringe contain half the volume of liquid, compared with the classic version of the biologic, so it’s 40 mg of drug in 0.4 mL rather than in 0.8 mL.
The packaging of adalimumab citrate free is different. It comes in a blue box to distinguish the product from the classic version.
Dr. Duffin reported receiving research grants from and serving as a consultant to AbbVie, which markets adalimumab, as well as close to a dozen other pharmaceutical companies.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – A flurry of recent impressive , identifies a simple biomarker predictive of the likelihood of a favorable PASI 75 response, and highlights a disconnect in psoriatic arthritis (PsA) patients between clinical response as reflected in disease activity and radiographic progression of joint disease, according to Kristina C. Duffin, MD.
Also, a new citrate-free version of adalimumab (Humira) is available. It requires a new prescription, and an additional prior authorization is mandated by some insurers. But this is a welcome innovation for patients bothered by significant burning and stinging with their injections of classic adalimumab, Dr. Duffin, cochair of the department of dermatology at the University of Utah, Salt Lake City, said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
New long-term safety data
Adalimumab is a market leader in biologic therapy for psoriasis. But the long-term experience with biologics in dermatology is still relatively limited, so the recent publication of two large studies providing encouraging evidence of the long-term safety of adalimumab is noteworthy.
Craig L. Leonardi, MD, of Saint Louis University, St. Louis, Mo., was first author of an analysis of long-term safety data from 18 clinical trials in adults with moderate to severe plaque psoriasis. The key takeaway, in Dr. Duffin’s view, was that the rate of adverse events, including serious infections and malignancies other than nonmelanoma skin cancer, remained stable over time out to 240 weeks of follow-up in patients on continuous treatment, with no new safety signals emerging (Br J Dermatol. 2019 Jan;180[1]:76-85).
However, randomized clinical trials often paint an overly rosy safety picture because of their strict inclusion and exclusion criteria.
“We single out patients for clinical trials because they’re especially healthy. That doesn’t happen in real-world registries,” she noted.
That’s why a systematic review of adalimumab’s safety performance in 10 real-world registries of adalimumab-treated psoriasis patients is particularly informative. The registries included in the systematic review, led by Bruce E. Strober, MD, PhD, professor of dermatology at the University of Connecticut, Farmington, didn’t all measure the same outcomes. But the three registries that documented major adverse cardiovascular events showed rates of less than 0.1 to less than 1 per 100 patient-years. Rates of malignancies other than nonmelanoma skin cancer were consistently in the 0.3-0.6 events per 100 patient-years range, similar to what has been reported in studies of other systemic psoriasis therapies, biologic as well as nonbiologic (J Eur Acad Dermatol Venereol. 2018 Dec;32[12]:2126-33).
Overall infection rates reported in the real-world registries ranged from 7.7 to 14.7 events per 100 patient-years, which is actually considerably lower than in the clinical trials. Rates of serious infections ranged from less than one up to two events per 100 patient-years, with the most common ones being cellulitis and pneumonia, consistent with the randomized trial experience.
Predicting response to adalimumab
A prospective, multicenter, observational cohort study of 544 psoriasis patients on adalimumab monotherapy conducted by U.K. investigators concluded that a patient’s serum drug level is the single most important predictor of treatment response. A cut point of 3.2 mcg/mL, which is considered the minimal effective circulating drug level, was associated with a 65% probability of a 75% improvement in Psoriasis Area and Severity Index from baseline, or PASI 75 response. The higher the serum drug level, the greater the likelihood of a PASI 75 response, up to a serum level of 7 mcg/mL, which was associated with an 81% probability of achieving PASI 75. Beyond 7 mcg/mL, however, the relationship with treatment response plateaued. Importantly, drug levels measured early on – at 1-12 weeks into therapy – were predictive of response 6 months later. So were steady-state levels (J Invest Dermatol. 2019 Jan;139[1]:115-23).
This is clinically useful information, Dr. Duffin observed.
“I’m hoping we’re going to see more real-world use of checking drug levels,” she said.
Indeed, even though the approved dosing of adalimumab for psoriasis is 40 mg by subcutaneous injection every 2 weeks, the new American Academy of Dermatology/National Psoriasis Foundation joint guidelines for treatment of psoriasis with biologics declare that “a maintenance dose of adalimumab at 40 mg/week is recommended for better disease control in some patients” (J Am Acad Dermatol. 2019 Feb 7. doi: 10.1016/j.jaad.2018.11.057. [Epub ahead of print]).
The new guidelines provide support for dermatologists who decide weekly therapy is best for a given patient, and adalimumab drug levels could prove useful in identifying the patient subgroup likely to benefit.
Dr. Duffin is often consulted by other physicians as to whether they should check for neutralizing antibodies in patients who appear to be losing therapeutic efficacy on a given biologic. She’s not a fan of the practice.
“There are commercial assays out there, but it’s very hard to interpret them because we don’t really know if they’re truly measuring neutralizing antibodies. And the cost is not insignificant; it can be hundreds of dollars,” she noted.
She believes a straightforward measurement of the serum biologic level is a better strategy.
“It makes sense: This is an indirect way of determining if there’s been neutralization of the drug, rather than trying to check the antibody that’s doing it, which is fraught with problems,” Dr. Duffin said.
Radiographic progression and clinical PsA activity on adalimumab don’t always correlate
A post hoc analysis of the randomized, double-blind, placebo-controlled ADEPT trial in PsA patients demonstrated that inhibition of radiographic progression as measured by change in modified total Sharp score from baseline through 24 weeks of adalimumab therapy was greater than expected based upon control of clinical disease activity (Rheumatology [Oxford]. 2019 Jan 3. doi: 10.1093/rheumatology/key417. [Epub ahead of print]).
One implication of the disconnect between radiographic progression and clinical disease documented in this study is that a dermatologist shouldn’t be too quick to change from adalimumab to another biologic just because a patient with PsA reports continued but bearable joint pain. And the converse is also true.
“I think that we as dermatologists probably shouldn’t be reassured when a patient says, ‘My joints feel great!” That’s because you may not necessarily be able to predict lack of progression in Sharp score based upon clinical response,” Dr. Duffin cautioned. “I think you should still have a rheumatologist check in with the patient and do x-rays periodically. The rheumatologist I work with does that, usually about on a yearly basis.”
Another key finding in the ADEPT analysis was that concomitant methotrexate had no added effect in terms of preventing joint destruction. This underscores the prescience of the first-ever collaborative American College of Rheumatology/National Psoriasis Foundation guidelines for the treatment of PsA (Arthritis Care Res (Hoboken). 2019 Jan;71[1]:2-29).
The new guidelines recommend that, in a psoriasis patient with confirmed PsA, the first-line treatment is a tumor necrosis factor (TNF) inhibitor. Agents from this class are preferred over other biologics because they are backed by a larger body of data regarding inhibition of joint disease progression. If the patient fails on the first TNF inhibitor prescribed, second-line therapy is another TNF inhibitor. So is third-line therapy.
Adalimumab citrate free
Not only does this new iteration of adalimumab do away with citrate as a buffer because it can cause pain and burning, it also utilizes a thinner 29-gauge needle rather than the standard 27-gauge. And the needle cover isn’t made with natural rubber latex. Also, both the pen and prefilled syringe contain half the volume of liquid, compared with the classic version of the biologic, so it’s 40 mg of drug in 0.4 mL rather than in 0.8 mL.
The packaging of adalimumab citrate free is different. It comes in a blue box to distinguish the product from the classic version.
Dr. Duffin reported receiving research grants from and serving as a consultant to AbbVie, which markets adalimumab, as well as close to a dozen other pharmaceutical companies.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – A flurry of recent impressive , identifies a simple biomarker predictive of the likelihood of a favorable PASI 75 response, and highlights a disconnect in psoriatic arthritis (PsA) patients between clinical response as reflected in disease activity and radiographic progression of joint disease, according to Kristina C. Duffin, MD.
Also, a new citrate-free version of adalimumab (Humira) is available. It requires a new prescription, and an additional prior authorization is mandated by some insurers. But this is a welcome innovation for patients bothered by significant burning and stinging with their injections of classic adalimumab, Dr. Duffin, cochair of the department of dermatology at the University of Utah, Salt Lake City, said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
New long-term safety data
Adalimumab is a market leader in biologic therapy for psoriasis. But the long-term experience with biologics in dermatology is still relatively limited, so the recent publication of two large studies providing encouraging evidence of the long-term safety of adalimumab is noteworthy.
Craig L. Leonardi, MD, of Saint Louis University, St. Louis, Mo., was first author of an analysis of long-term safety data from 18 clinical trials in adults with moderate to severe plaque psoriasis. The key takeaway, in Dr. Duffin’s view, was that the rate of adverse events, including serious infections and malignancies other than nonmelanoma skin cancer, remained stable over time out to 240 weeks of follow-up in patients on continuous treatment, with no new safety signals emerging (Br J Dermatol. 2019 Jan;180[1]:76-85).
However, randomized clinical trials often paint an overly rosy safety picture because of their strict inclusion and exclusion criteria.
“We single out patients for clinical trials because they’re especially healthy. That doesn’t happen in real-world registries,” she noted.
That’s why a systematic review of adalimumab’s safety performance in 10 real-world registries of adalimumab-treated psoriasis patients is particularly informative. The registries included in the systematic review, led by Bruce E. Strober, MD, PhD, professor of dermatology at the University of Connecticut, Farmington, didn’t all measure the same outcomes. But the three registries that documented major adverse cardiovascular events showed rates of less than 0.1 to less than 1 per 100 patient-years. Rates of malignancies other than nonmelanoma skin cancer were consistently in the 0.3-0.6 events per 100 patient-years range, similar to what has been reported in studies of other systemic psoriasis therapies, biologic as well as nonbiologic (J Eur Acad Dermatol Venereol. 2018 Dec;32[12]:2126-33).
Overall infection rates reported in the real-world registries ranged from 7.7 to 14.7 events per 100 patient-years, which is actually considerably lower than in the clinical trials. Rates of serious infections ranged from less than one up to two events per 100 patient-years, with the most common ones being cellulitis and pneumonia, consistent with the randomized trial experience.
Predicting response to adalimumab
A prospective, multicenter, observational cohort study of 544 psoriasis patients on adalimumab monotherapy conducted by U.K. investigators concluded that a patient’s serum drug level is the single most important predictor of treatment response. A cut point of 3.2 mcg/mL, which is considered the minimal effective circulating drug level, was associated with a 65% probability of a 75% improvement in Psoriasis Area and Severity Index from baseline, or PASI 75 response. The higher the serum drug level, the greater the likelihood of a PASI 75 response, up to a serum level of 7 mcg/mL, which was associated with an 81% probability of achieving PASI 75. Beyond 7 mcg/mL, however, the relationship with treatment response plateaued. Importantly, drug levels measured early on – at 1-12 weeks into therapy – were predictive of response 6 months later. So were steady-state levels (J Invest Dermatol. 2019 Jan;139[1]:115-23).
This is clinically useful information, Dr. Duffin observed.
“I’m hoping we’re going to see more real-world use of checking drug levels,” she said.
Indeed, even though the approved dosing of adalimumab for psoriasis is 40 mg by subcutaneous injection every 2 weeks, the new American Academy of Dermatology/National Psoriasis Foundation joint guidelines for treatment of psoriasis with biologics declare that “a maintenance dose of adalimumab at 40 mg/week is recommended for better disease control in some patients” (J Am Acad Dermatol. 2019 Feb 7. doi: 10.1016/j.jaad.2018.11.057. [Epub ahead of print]).
The new guidelines provide support for dermatologists who decide weekly therapy is best for a given patient, and adalimumab drug levels could prove useful in identifying the patient subgroup likely to benefit.
Dr. Duffin is often consulted by other physicians as to whether they should check for neutralizing antibodies in patients who appear to be losing therapeutic efficacy on a given biologic. She’s not a fan of the practice.
“There are commercial assays out there, but it’s very hard to interpret them because we don’t really know if they’re truly measuring neutralizing antibodies. And the cost is not insignificant; it can be hundreds of dollars,” she noted.
She believes a straightforward measurement of the serum biologic level is a better strategy.
“It makes sense: This is an indirect way of determining if there’s been neutralization of the drug, rather than trying to check the antibody that’s doing it, which is fraught with problems,” Dr. Duffin said.
Radiographic progression and clinical PsA activity on adalimumab don’t always correlate
A post hoc analysis of the randomized, double-blind, placebo-controlled ADEPT trial in PsA patients demonstrated that inhibition of radiographic progression as measured by change in modified total Sharp score from baseline through 24 weeks of adalimumab therapy was greater than expected based upon control of clinical disease activity (Rheumatology [Oxford]. 2019 Jan 3. doi: 10.1093/rheumatology/key417. [Epub ahead of print]).
One implication of the disconnect between radiographic progression and clinical disease documented in this study is that a dermatologist shouldn’t be too quick to change from adalimumab to another biologic just because a patient with PsA reports continued but bearable joint pain. And the converse is also true.
“I think that we as dermatologists probably shouldn’t be reassured when a patient says, ‘My joints feel great!” That’s because you may not necessarily be able to predict lack of progression in Sharp score based upon clinical response,” Dr. Duffin cautioned. “I think you should still have a rheumatologist check in with the patient and do x-rays periodically. The rheumatologist I work with does that, usually about on a yearly basis.”
Another key finding in the ADEPT analysis was that concomitant methotrexate had no added effect in terms of preventing joint destruction. This underscores the prescience of the first-ever collaborative American College of Rheumatology/National Psoriasis Foundation guidelines for the treatment of PsA (Arthritis Care Res (Hoboken). 2019 Jan;71[1]:2-29).
The new guidelines recommend that, in a psoriasis patient with confirmed PsA, the first-line treatment is a tumor necrosis factor (TNF) inhibitor. Agents from this class are preferred over other biologics because they are backed by a larger body of data regarding inhibition of joint disease progression. If the patient fails on the first TNF inhibitor prescribed, second-line therapy is another TNF inhibitor. So is third-line therapy.
Adalimumab citrate free
Not only does this new iteration of adalimumab do away with citrate as a buffer because it can cause pain and burning, it also utilizes a thinner 29-gauge needle rather than the standard 27-gauge. And the needle cover isn’t made with natural rubber latex. Also, both the pen and prefilled syringe contain half the volume of liquid, compared with the classic version of the biologic, so it’s 40 mg of drug in 0.4 mL rather than in 0.8 mL.
The packaging of adalimumab citrate free is different. It comes in a blue box to distinguish the product from the classic version.
Dr. Duffin reported receiving research grants from and serving as a consultant to AbbVie, which markets adalimumab, as well as close to a dozen other pharmaceutical companies.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
REPORTING FROM SDEF HAWAII DERMATOLOGY SEMINAR
Don’t miss early joint involvement in psoriasis
WAIKOLOA, HAWAII – advised Alan Menter, MD.
About a third of patients with psoriasis will go on to develop joint involvement, and about half of those will go on to develop permanent joint destruction “if left untreated,” he noted. But early joint involvement has to be caught first, and dermatologists aren’t doing a very good job at early detection, according to Dr. Menter, clinical professor of dermatology at the University of Texas, Dallas.
The consequences, including arthritis mutilans, can be devastating. “It’s vitally important for us to prevent any permanent joint disease by” picking it up early, he said. “Our job as dermatologists is to diagnose it early.”
It’s not hard to do, just a few extra questions and a few extra steps on the physical exam, which takes a minute or two during each visit with psoriasis patients, are needed, he said.
Dr. Menter reviewed questions to ask patients, and explained how to examine patients for joint involvement and alter treatment when it’s found, in an interview at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – advised Alan Menter, MD.
About a third of patients with psoriasis will go on to develop joint involvement, and about half of those will go on to develop permanent joint destruction “if left untreated,” he noted. But early joint involvement has to be caught first, and dermatologists aren’t doing a very good job at early detection, according to Dr. Menter, clinical professor of dermatology at the University of Texas, Dallas.
The consequences, including arthritis mutilans, can be devastating. “It’s vitally important for us to prevent any permanent joint disease by” picking it up early, he said. “Our job as dermatologists is to diagnose it early.”
It’s not hard to do, just a few extra questions and a few extra steps on the physical exam, which takes a minute or two during each visit with psoriasis patients, are needed, he said.
Dr. Menter reviewed questions to ask patients, and explained how to examine patients for joint involvement and alter treatment when it’s found, in an interview at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – advised Alan Menter, MD.
About a third of patients with psoriasis will go on to develop joint involvement, and about half of those will go on to develop permanent joint destruction “if left untreated,” he noted. But early joint involvement has to be caught first, and dermatologists aren’t doing a very good job at early detection, according to Dr. Menter, clinical professor of dermatology at the University of Texas, Dallas.
The consequences, including arthritis mutilans, can be devastating. “It’s vitally important for us to prevent any permanent joint disease by” picking it up early, he said. “Our job as dermatologists is to diagnose it early.”
It’s not hard to do, just a few extra questions and a few extra steps on the physical exam, which takes a minute or two during each visit with psoriasis patients, are needed, he said.
Dr. Menter reviewed questions to ask patients, and explained how to examine patients for joint involvement and alter treatment when it’s found, in an interview at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Biologics curb coronary artery plaques in severe psoriasis
Treatment with biologic therapy significantly improves coronary plaque profiles in patients with severe psoriasis, based on data from 121 adult patients who completed a year of follow-up.
A previous study showed a reduced rate of nonfatal myocardial infarction, nonfatal stroke, and cardiovascular death among individuals treated with biologic therapies, wrote Youssef A. Elnabawi, MD, of the National Heart, Lung, and Blood Institute in Bethesda, Md., and his colleagues.
Psoriasis “provides a reliable model to study inflammatory atherogenesis and the longitudinal impact of modulating specific cytokines on vascular behavior, while treating the primary skin disease with [Food and Drug Administration]–approved biologic therapies,” the researchers said.
In a study published in Cardiovascular Research, patients given biologics showed a 5% reduction in total coronary plaque burden after 1 year, as well as a 64% improvement in Psoriasis Area Severity Index scores. In addition, the decrease in noncalcified plaque burden in the biologics group was significantly greater, compared with the nonbiologics group (P =.03), and remained significant after controlling for standard cardiovascular risk factors.
When broken down by biologic, “we observed the greatest percent reduction of noncalcified plaque burden in patients on [anti-interleukin (IL)–17] therapy with a reduction in necrotic core suggesting a potential role for IL-17 in atherosclerotic pathways,” Dr. Elnabawi and his colleagues wrote.
(from 2.0 mg/dL to 1.4 mg/dL), but no change in the nonbiologics group.
The study population included patients naive to biologic or systemic psoriasis therapies who were assessed via clinical and laboratory data and coronary computed tomography angiography at baseline and after 1 year. A total of 89 participants with moderate to severe psoriasis received biologics, including adalimumab, etanercept, ustekinumab, secukinumab, and ixekizumab; 32 psoriasis patients received no biologics and served as a reference group. The average age of the patients was 50 years, and 58% were male. At baseline, patients had low cardiovascular risk based on Framingham scores, and moderate to severe skin disease.
The findings were limited by several factors, including the observational nature of the study, small study population, and the open-label use of biologics, as well as the use of coronary indices, rather than actual cardiovascular events, to assess cardiovascular disease risk, the researchers noted.
However, the results, combined with results from previous studies in animal models, “support further investigation of IL-17 blockade on coronary disease in humans,” they said.
The study was supported by the National Heart, Lung, and Blood Institute, with additional support from the National Institutes of Health Medical Research Scholars Program, the Doris Duke Charitable Foundation, the American Association for Dental Research, the Colgate-Palmolive Company, Genentech, Elsevier, and other private donors. Dr. Elnabawi had no financial conflicts to disclose.
SOURCE: Elnabawi YA et al. Cardiovasc Res. 2019. doi: 10.1093/cvr/cvz009.
Treatment with biologic therapy significantly improves coronary plaque profiles in patients with severe psoriasis, based on data from 121 adult patients who completed a year of follow-up.
A previous study showed a reduced rate of nonfatal myocardial infarction, nonfatal stroke, and cardiovascular death among individuals treated with biologic therapies, wrote Youssef A. Elnabawi, MD, of the National Heart, Lung, and Blood Institute in Bethesda, Md., and his colleagues.
Psoriasis “provides a reliable model to study inflammatory atherogenesis and the longitudinal impact of modulating specific cytokines on vascular behavior, while treating the primary skin disease with [Food and Drug Administration]–approved biologic therapies,” the researchers said.
In a study published in Cardiovascular Research, patients given biologics showed a 5% reduction in total coronary plaque burden after 1 year, as well as a 64% improvement in Psoriasis Area Severity Index scores. In addition, the decrease in noncalcified plaque burden in the biologics group was significantly greater, compared with the nonbiologics group (P =.03), and remained significant after controlling for standard cardiovascular risk factors.
When broken down by biologic, “we observed the greatest percent reduction of noncalcified plaque burden in patients on [anti-interleukin (IL)–17] therapy with a reduction in necrotic core suggesting a potential role for IL-17 in atherosclerotic pathways,” Dr. Elnabawi and his colleagues wrote.
(from 2.0 mg/dL to 1.4 mg/dL), but no change in the nonbiologics group.
The study population included patients naive to biologic or systemic psoriasis therapies who were assessed via clinical and laboratory data and coronary computed tomography angiography at baseline and after 1 year. A total of 89 participants with moderate to severe psoriasis received biologics, including adalimumab, etanercept, ustekinumab, secukinumab, and ixekizumab; 32 psoriasis patients received no biologics and served as a reference group. The average age of the patients was 50 years, and 58% were male. At baseline, patients had low cardiovascular risk based on Framingham scores, and moderate to severe skin disease.
The findings were limited by several factors, including the observational nature of the study, small study population, and the open-label use of biologics, as well as the use of coronary indices, rather than actual cardiovascular events, to assess cardiovascular disease risk, the researchers noted.
However, the results, combined with results from previous studies in animal models, “support further investigation of IL-17 blockade on coronary disease in humans,” they said.
The study was supported by the National Heart, Lung, and Blood Institute, with additional support from the National Institutes of Health Medical Research Scholars Program, the Doris Duke Charitable Foundation, the American Association for Dental Research, the Colgate-Palmolive Company, Genentech, Elsevier, and other private donors. Dr. Elnabawi had no financial conflicts to disclose.
SOURCE: Elnabawi YA et al. Cardiovasc Res. 2019. doi: 10.1093/cvr/cvz009.
Treatment with biologic therapy significantly improves coronary plaque profiles in patients with severe psoriasis, based on data from 121 adult patients who completed a year of follow-up.
A previous study showed a reduced rate of nonfatal myocardial infarction, nonfatal stroke, and cardiovascular death among individuals treated with biologic therapies, wrote Youssef A. Elnabawi, MD, of the National Heart, Lung, and Blood Institute in Bethesda, Md., and his colleagues.
Psoriasis “provides a reliable model to study inflammatory atherogenesis and the longitudinal impact of modulating specific cytokines on vascular behavior, while treating the primary skin disease with [Food and Drug Administration]–approved biologic therapies,” the researchers said.
In a study published in Cardiovascular Research, patients given biologics showed a 5% reduction in total coronary plaque burden after 1 year, as well as a 64% improvement in Psoriasis Area Severity Index scores. In addition, the decrease in noncalcified plaque burden in the biologics group was significantly greater, compared with the nonbiologics group (P =.03), and remained significant after controlling for standard cardiovascular risk factors.
When broken down by biologic, “we observed the greatest percent reduction of noncalcified plaque burden in patients on [anti-interleukin (IL)–17] therapy with a reduction in necrotic core suggesting a potential role for IL-17 in atherosclerotic pathways,” Dr. Elnabawi and his colleagues wrote.
(from 2.0 mg/dL to 1.4 mg/dL), but no change in the nonbiologics group.
The study population included patients naive to biologic or systemic psoriasis therapies who were assessed via clinical and laboratory data and coronary computed tomography angiography at baseline and after 1 year. A total of 89 participants with moderate to severe psoriasis received biologics, including adalimumab, etanercept, ustekinumab, secukinumab, and ixekizumab; 32 psoriasis patients received no biologics and served as a reference group. The average age of the patients was 50 years, and 58% were male. At baseline, patients had low cardiovascular risk based on Framingham scores, and moderate to severe skin disease.
The findings were limited by several factors, including the observational nature of the study, small study population, and the open-label use of biologics, as well as the use of coronary indices, rather than actual cardiovascular events, to assess cardiovascular disease risk, the researchers noted.
However, the results, combined with results from previous studies in animal models, “support further investigation of IL-17 blockade on coronary disease in humans,” they said.
The study was supported by the National Heart, Lung, and Blood Institute, with additional support from the National Institutes of Health Medical Research Scholars Program, the Doris Duke Charitable Foundation, the American Association for Dental Research, the Colgate-Palmolive Company, Genentech, Elsevier, and other private donors. Dr. Elnabawi had no financial conflicts to disclose.
SOURCE: Elnabawi YA et al. Cardiovasc Res. 2019. doi: 10.1093/cvr/cvz009.
FROM CARDIOVASCULAR RESEARCH
Key clinical point: Psoriasis patients treated with biologics also showed improvement in coronary artery profiles after 1 year, compared with patients not treated with biologics.
Major finding: Biologic therapy was associated with a 5% reduction in total coronary plaque burden from baseline.
Study details: The data come from 121 psoriasis patients in a prospective, observational study.
Disclosures: The study was supported by the National Heart, Lung, and Blood Institute, with additional support from the National Institutes of Health Medical Research Scholars Program, the Doris Duke Charitable Foundation, the American Association for Dental Research, the Colgate-Palmolive Company, Genentech, Elsevier, and other private donors. Dr. Elnabawi had no financial conflicts to disclose.
Source: Elnabawi YA et al. Cardiovasc Res. 2019. doi: 10.1093/cvr/cvz009.