User login
News and Views that Matter to Pediatricians
The leading independent newspaper covering news and commentary in pediatrics.
Employed physicians: A survival guide
The strike by health care workers at Kaiser Permanente may not involve physicians (yet). But as more doctors in the United States are finding themselves working as salaried employees, physicians can – and probably will – become a powerful force for change in a health care system that has shown itself to be increasingly hostile to employee concerns over issues involving patient care, wages and benefits, safety, and well-being.
Salaried employment has its challenges. Physician-employees may have less autonomy and voice in decision-making that affects patients. They may splinter into fragmented work groups; feel isolated; and have different imperatives based on who they are, what they want, and where they work. They may feel more removed from their patients and struggle to build strong relationships, with their employers in the way.
Yet important opportunities exist for doctors when embracing their employee side. Examples of these interests include adequate compensation, wellness, job security, patient and worker safety, health care quality, reasonable workloads and schedules, and fair treatment by employers, including the need to exhibit a strong collective voice in organizational decision-making.
Some believe that physician-employees must be unionized to maximize their rights and power as employees. Many expect physician unionization to take hold more fully over time. Medical residents, the doctors of tomorrow, are already considering unionization in greater numbers. Some are also doing it in the same employment setting alongside other health professionals, such as nurses.
Having studied doctors and their employment situations for years, I am convinced that whether through unionization or another approach, physicians must also change how they think about control; train and learn alongside other health care workers who share similar interests; and elevate at an early career stage their knowledge of the business side of health care.
Adopt a more pragmatic definition of autonomy
Doctors must embrace an updated definition of autonomy – one that matches their status as highly paid labor.
When I have spoken to physicians in my research about what autonomy means to them, many seem unable to reconceptualize it from a vague and absolute form of their profession’s strategic control over their economic fates and technical skills toward an individualized control that is situation-specific, one centered on winning the daily fights about workplace bread-and-butter issues such as those mentioned above.
But a more pragmatic definition of autonomy could get doctors focused on influencing important issues of the patient-care day and enhance their negotiating power with employers. It would allow physicians to break out of what often seems a paralysis of inaction – waiting for employers, insurers, or the government to reinstate the profession’s idealized version of control by handing it back the keys to the health care system through major regulatory, structural, and reimbursement-related changes. This fantasy is unlikely to become reality.
Physician-employees I’ve talked to over the years understand their everyday challenges. But when it comes to engaging in localized and sustained action to overcome them, they often perform less well, leading to feelings of helplessness and burnout. Valuing tactical control over their jobs and work setting will yield smaller but more impactful wins as employees intent on making their everyday work lives better.
Train alongside other health care professionals
Physicians must accept that how they are trained no longer prepares them for the employee world into which most are dropped. For instance, unless doctors are trained collaboratively alongside other health care professionals – such as nurses – they are less likely to identify closely with these colleagues once in practice. There is strength in numbers, so this mutual identification empowers both groups of employees. Yet, medical education remains largely the same: training young medical students in isolation for the first couple of years, then placing them into clerkships and residencies where true interprofessional care opportunities remain stunted and secondary to the “physician as captain of the team” mantra.
Unfortunately, the “hidden curriculum” of medicine helps convince medical students and residents early in their careers that they are the unquestioned leaders in patient care settings. This hierarchy encourages some doctors to keep their psychological distance from other members of the health care team and to resist sharing power, concerns, or insights with less skilled health care workers. This socialization harms the ability of physicians to act in a unified fashion alongside these other workers. Having physicians learn and train alongside other health professionals yields positive benefits for collective advocacy, including a shared sense of purpose, positive views on collaboration with others in the health setting, and greater development of bonds with nonphysician coworkers.
Integrate business with medical training in real time
Medical students and residents generally lack exposure to the everyday business realities of the U.S. health care system. This gap hinders their ability to understand the employee world and push for the types of changes and work conditions that benefit all health care workers. Formal business and management training should be a required part of every U.S. medical school and residency curriculum from day one. If you see it at all in medical schools now, it is mostly by accident, or given separate treatment in the form of standalone MBA or MPH degrees that rarely integrate organically and in real time with actual medical training. Not every doctor needs an MBA or MPH degree. However, all of them require a stronger contextual understanding of how the medicine they wish to practice is shaped by the economic and fiscal circumstances surrounding it – circumstances they do not control.
This is another reason why young doctors are unhappy and burned out. They cannot push for specific changes or properly critique the pros and cons of how their work is structured because they have not been made aware, in real time as they learn clinical practice, how their jobs are shaped by realities such as insurance coverage and reimbursement, the fragmentation of the care delivery system, their employer’s financial health , and the socioeconomic circumstances of their patients. They aren’t given the methods and tools related to process and quality improvement, budgeting, negotiation, risk management, leadership, and talent management that might help them navigate these undermining forces. They also get little advance exposure in their training to important workplace “soft” skills in such areas as how to work in teams, networking, communication and listening, empathy, and problem-solving – all necessary foci for bringing them closer to other health care workers and advocating alongside them effectively with health care employers.
Now is the time for physicians to embrace their identity as employees. Doing so is in their own best interest as professionals. It will help others in the health care workforce as well as patients. Moreover, it provides a needed counterbalance to the powerful corporate ethos now ascendant in U.S. health care.
Timothy Hoff, PhD, is a professor of management and healthcare systems at Northeastern University, Boston, and an associate fellow at the University of Oxford, England. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
The strike by health care workers at Kaiser Permanente may not involve physicians (yet). But as more doctors in the United States are finding themselves working as salaried employees, physicians can – and probably will – become a powerful force for change in a health care system that has shown itself to be increasingly hostile to employee concerns over issues involving patient care, wages and benefits, safety, and well-being.
Salaried employment has its challenges. Physician-employees may have less autonomy and voice in decision-making that affects patients. They may splinter into fragmented work groups; feel isolated; and have different imperatives based on who they are, what they want, and where they work. They may feel more removed from their patients and struggle to build strong relationships, with their employers in the way.
Yet important opportunities exist for doctors when embracing their employee side. Examples of these interests include adequate compensation, wellness, job security, patient and worker safety, health care quality, reasonable workloads and schedules, and fair treatment by employers, including the need to exhibit a strong collective voice in organizational decision-making.
Some believe that physician-employees must be unionized to maximize their rights and power as employees. Many expect physician unionization to take hold more fully over time. Medical residents, the doctors of tomorrow, are already considering unionization in greater numbers. Some are also doing it in the same employment setting alongside other health professionals, such as nurses.
Having studied doctors and their employment situations for years, I am convinced that whether through unionization or another approach, physicians must also change how they think about control; train and learn alongside other health care workers who share similar interests; and elevate at an early career stage their knowledge of the business side of health care.
Adopt a more pragmatic definition of autonomy
Doctors must embrace an updated definition of autonomy – one that matches their status as highly paid labor.
When I have spoken to physicians in my research about what autonomy means to them, many seem unable to reconceptualize it from a vague and absolute form of their profession’s strategic control over their economic fates and technical skills toward an individualized control that is situation-specific, one centered on winning the daily fights about workplace bread-and-butter issues such as those mentioned above.
But a more pragmatic definition of autonomy could get doctors focused on influencing important issues of the patient-care day and enhance their negotiating power with employers. It would allow physicians to break out of what often seems a paralysis of inaction – waiting for employers, insurers, or the government to reinstate the profession’s idealized version of control by handing it back the keys to the health care system through major regulatory, structural, and reimbursement-related changes. This fantasy is unlikely to become reality.
Physician-employees I’ve talked to over the years understand their everyday challenges. But when it comes to engaging in localized and sustained action to overcome them, they often perform less well, leading to feelings of helplessness and burnout. Valuing tactical control over their jobs and work setting will yield smaller but more impactful wins as employees intent on making their everyday work lives better.
Train alongside other health care professionals
Physicians must accept that how they are trained no longer prepares them for the employee world into which most are dropped. For instance, unless doctors are trained collaboratively alongside other health care professionals – such as nurses – they are less likely to identify closely with these colleagues once in practice. There is strength in numbers, so this mutual identification empowers both groups of employees. Yet, medical education remains largely the same: training young medical students in isolation for the first couple of years, then placing them into clerkships and residencies where true interprofessional care opportunities remain stunted and secondary to the “physician as captain of the team” mantra.
Unfortunately, the “hidden curriculum” of medicine helps convince medical students and residents early in their careers that they are the unquestioned leaders in patient care settings. This hierarchy encourages some doctors to keep their psychological distance from other members of the health care team and to resist sharing power, concerns, or insights with less skilled health care workers. This socialization harms the ability of physicians to act in a unified fashion alongside these other workers. Having physicians learn and train alongside other health professionals yields positive benefits for collective advocacy, including a shared sense of purpose, positive views on collaboration with others in the health setting, and greater development of bonds with nonphysician coworkers.
Integrate business with medical training in real time
Medical students and residents generally lack exposure to the everyday business realities of the U.S. health care system. This gap hinders their ability to understand the employee world and push for the types of changes and work conditions that benefit all health care workers. Formal business and management training should be a required part of every U.S. medical school and residency curriculum from day one. If you see it at all in medical schools now, it is mostly by accident, or given separate treatment in the form of standalone MBA or MPH degrees that rarely integrate organically and in real time with actual medical training. Not every doctor needs an MBA or MPH degree. However, all of them require a stronger contextual understanding of how the medicine they wish to practice is shaped by the economic and fiscal circumstances surrounding it – circumstances they do not control.
This is another reason why young doctors are unhappy and burned out. They cannot push for specific changes or properly critique the pros and cons of how their work is structured because they have not been made aware, in real time as they learn clinical practice, how their jobs are shaped by realities such as insurance coverage and reimbursement, the fragmentation of the care delivery system, their employer’s financial health , and the socioeconomic circumstances of their patients. They aren’t given the methods and tools related to process and quality improvement, budgeting, negotiation, risk management, leadership, and talent management that might help them navigate these undermining forces. They also get little advance exposure in their training to important workplace “soft” skills in such areas as how to work in teams, networking, communication and listening, empathy, and problem-solving – all necessary foci for bringing them closer to other health care workers and advocating alongside them effectively with health care employers.
Now is the time for physicians to embrace their identity as employees. Doing so is in their own best interest as professionals. It will help others in the health care workforce as well as patients. Moreover, it provides a needed counterbalance to the powerful corporate ethos now ascendant in U.S. health care.
Timothy Hoff, PhD, is a professor of management and healthcare systems at Northeastern University, Boston, and an associate fellow at the University of Oxford, England. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
The strike by health care workers at Kaiser Permanente may not involve physicians (yet). But as more doctors in the United States are finding themselves working as salaried employees, physicians can – and probably will – become a powerful force for change in a health care system that has shown itself to be increasingly hostile to employee concerns over issues involving patient care, wages and benefits, safety, and well-being.
Salaried employment has its challenges. Physician-employees may have less autonomy and voice in decision-making that affects patients. They may splinter into fragmented work groups; feel isolated; and have different imperatives based on who they are, what they want, and where they work. They may feel more removed from their patients and struggle to build strong relationships, with their employers in the way.
Yet important opportunities exist for doctors when embracing their employee side. Examples of these interests include adequate compensation, wellness, job security, patient and worker safety, health care quality, reasonable workloads and schedules, and fair treatment by employers, including the need to exhibit a strong collective voice in organizational decision-making.
Some believe that physician-employees must be unionized to maximize their rights and power as employees. Many expect physician unionization to take hold more fully over time. Medical residents, the doctors of tomorrow, are already considering unionization in greater numbers. Some are also doing it in the same employment setting alongside other health professionals, such as nurses.
Having studied doctors and their employment situations for years, I am convinced that whether through unionization or another approach, physicians must also change how they think about control; train and learn alongside other health care workers who share similar interests; and elevate at an early career stage their knowledge of the business side of health care.
Adopt a more pragmatic definition of autonomy
Doctors must embrace an updated definition of autonomy – one that matches their status as highly paid labor.
When I have spoken to physicians in my research about what autonomy means to them, many seem unable to reconceptualize it from a vague and absolute form of their profession’s strategic control over their economic fates and technical skills toward an individualized control that is situation-specific, one centered on winning the daily fights about workplace bread-and-butter issues such as those mentioned above.
But a more pragmatic definition of autonomy could get doctors focused on influencing important issues of the patient-care day and enhance their negotiating power with employers. It would allow physicians to break out of what often seems a paralysis of inaction – waiting for employers, insurers, or the government to reinstate the profession’s idealized version of control by handing it back the keys to the health care system through major regulatory, structural, and reimbursement-related changes. This fantasy is unlikely to become reality.
Physician-employees I’ve talked to over the years understand their everyday challenges. But when it comes to engaging in localized and sustained action to overcome them, they often perform less well, leading to feelings of helplessness and burnout. Valuing tactical control over their jobs and work setting will yield smaller but more impactful wins as employees intent on making their everyday work lives better.
Train alongside other health care professionals
Physicians must accept that how they are trained no longer prepares them for the employee world into which most are dropped. For instance, unless doctors are trained collaboratively alongside other health care professionals – such as nurses – they are less likely to identify closely with these colleagues once in practice. There is strength in numbers, so this mutual identification empowers both groups of employees. Yet, medical education remains largely the same: training young medical students in isolation for the first couple of years, then placing them into clerkships and residencies where true interprofessional care opportunities remain stunted and secondary to the “physician as captain of the team” mantra.
Unfortunately, the “hidden curriculum” of medicine helps convince medical students and residents early in their careers that they are the unquestioned leaders in patient care settings. This hierarchy encourages some doctors to keep their psychological distance from other members of the health care team and to resist sharing power, concerns, or insights with less skilled health care workers. This socialization harms the ability of physicians to act in a unified fashion alongside these other workers. Having physicians learn and train alongside other health professionals yields positive benefits for collective advocacy, including a shared sense of purpose, positive views on collaboration with others in the health setting, and greater development of bonds with nonphysician coworkers.
Integrate business with medical training in real time
Medical students and residents generally lack exposure to the everyday business realities of the U.S. health care system. This gap hinders their ability to understand the employee world and push for the types of changes and work conditions that benefit all health care workers. Formal business and management training should be a required part of every U.S. medical school and residency curriculum from day one. If you see it at all in medical schools now, it is mostly by accident, or given separate treatment in the form of standalone MBA or MPH degrees that rarely integrate organically and in real time with actual medical training. Not every doctor needs an MBA or MPH degree. However, all of them require a stronger contextual understanding of how the medicine they wish to practice is shaped by the economic and fiscal circumstances surrounding it – circumstances they do not control.
This is another reason why young doctors are unhappy and burned out. They cannot push for specific changes or properly critique the pros and cons of how their work is structured because they have not been made aware, in real time as they learn clinical practice, how their jobs are shaped by realities such as insurance coverage and reimbursement, the fragmentation of the care delivery system, their employer’s financial health , and the socioeconomic circumstances of their patients. They aren’t given the methods and tools related to process and quality improvement, budgeting, negotiation, risk management, leadership, and talent management that might help them navigate these undermining forces. They also get little advance exposure in their training to important workplace “soft” skills in such areas as how to work in teams, networking, communication and listening, empathy, and problem-solving – all necessary foci for bringing them closer to other health care workers and advocating alongside them effectively with health care employers.
Now is the time for physicians to embrace their identity as employees. Doing so is in their own best interest as professionals. It will help others in the health care workforce as well as patients. Moreover, it provides a needed counterbalance to the powerful corporate ethos now ascendant in U.S. health care.
Timothy Hoff, PhD, is a professor of management and healthcare systems at Northeastern University, Boston, and an associate fellow at the University of Oxford, England. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
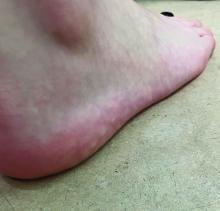
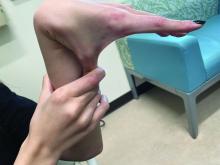
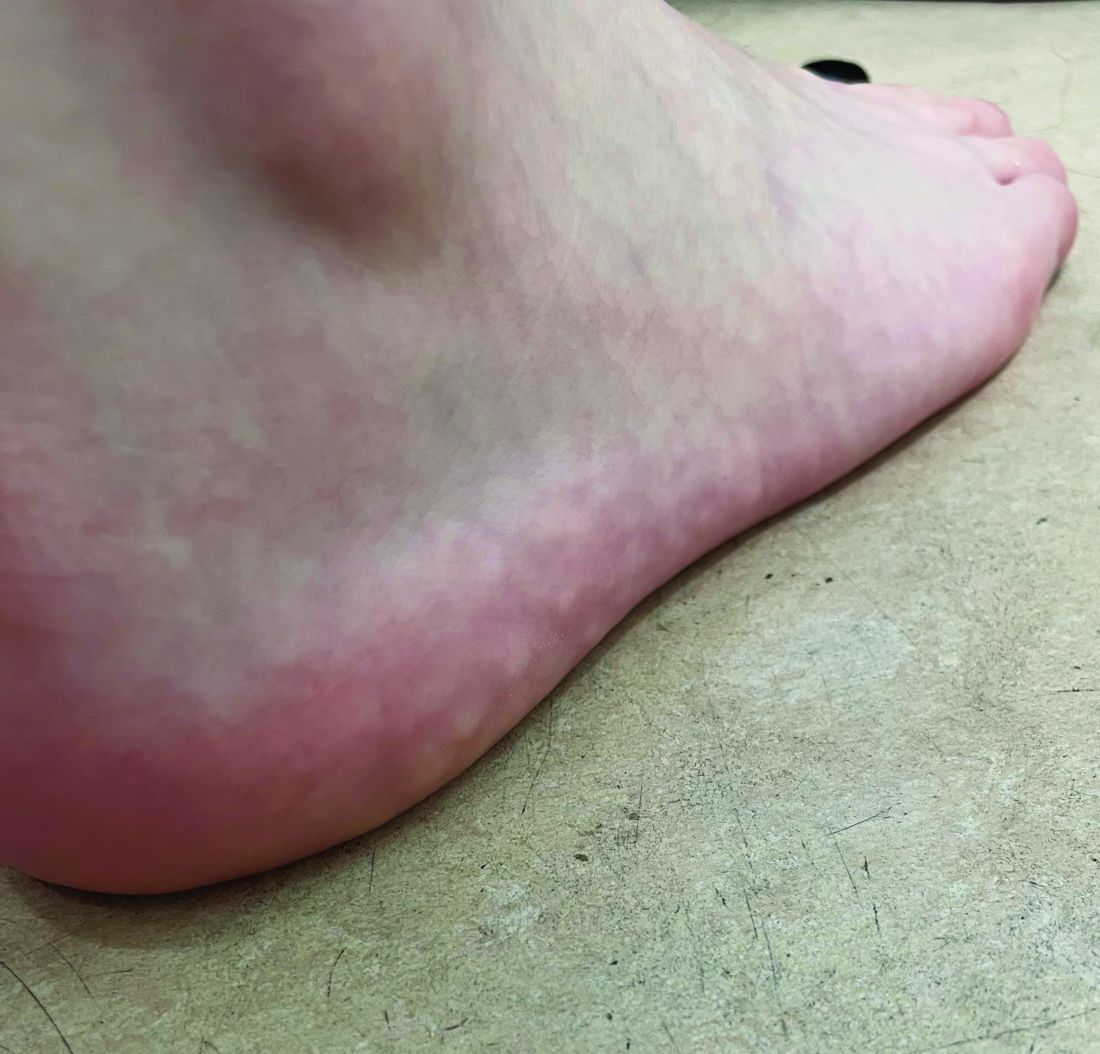
Why legal pot makes this physician sick
Last year, my husband and I took a 16-day road trip from Kentucky through Massachusetts to Maine. On our first morning in Boston, we exited the Park Street Station en route to Boston Common, but instead of being greeted by the aroma of molasses, we were hit full-on with a pungent, repulsive odor. “That’s skunk weed,” my husband chuckled as we stepped right into the middle of the Boston Freedom Rally, a celebration of all things cannabis.
As we boarded a hop-on-hop-off bus, we learned that this was the one week of the year that the city skips testing tour bus drivers for tetrahydrocannabinol (THC), “because we all test positive,” the driver quipped. As our open-air bus circled the Common, a crowd of pot enthusiasts displayed signs in support of relaxed regulation for public consumption.
The 34-year-old Boston Freedom Rally is a sign that U.S. culture has transformed forever. Mary Jane is no friend of emergency physicians nor of staff on hospital wards and offices.
Toking boomers and millennials
Researchers at the University of California, San Diego, looked at cannabis-related emergency department visits from all acute-care hospitals in the state from 2005 to 2019 and found an 1,808% increase in patients aged 65 or older (that is not a typo) who were there for complications from cannabis use.
The lead author said in an interview that, “older patients taking marijuana or related products may have dizziness and falls, heart palpitations, panic attacks, confusion, anxiety or worsening of underlying lung diseases, such as asthma or [chronic obstructive pulmonary disease].”
A recent study from Canada suggests that commercialization has been associated with an increase in related hospitalizations, including cannabis-induced psychosis.
According to a National Study of Drug Use and Health, marijuana use in young adults reached an all-time high (pun intended) in 2021. Nearly 10% of eighth graders and 20% of 10th graders reported using marijuana this past year.
The full downside of any drug, legal or illegal, is largely unknown until it infiltrates the mainstream market, but these are the typical cases we see:
Let’s start with the demotivated high school honors student who dropped out of college to work at the local cinema. He stumbled and broke his clavicle outside a bar at 2 AM, but he wasn’t sure if he passed out, so a cardiology consult was requested to “rule out” arrhythmia associated with syncope. He related that his plan to become a railway conductor had been upended because he knew he would be drug tested and just couldn’t give up pot. After a normal cardiac exam, ECG, labs, a Holter, and an echocardiogram were also requested and normal at a significant cost.
Cannabinoid hyperemesis syndrome
One of my Midwest colleagues related her encounter with two middle-aged pot users with ventricular tachycardia (VT). These episodes coincided with potassium levels less than 3.0 mEq/L in the setting of repetitive vomiting. The QTc interval didn’t normalize despite a corrected potassium level in one patient. They were both informed that they should never smoke pot because vomiting would predictably drop their K+ levels again and prolong their QTc intervals. Then began “the circular argument,” as my friend described it. The patient claims, “I smoke pot to relieve my nausea,” to which she explains that “in many folks, pot use induces nausea.” Of course, the classic reply is, “Not me.” Predictably one of these stoners soon returned with more VT, more puking, and more hypokalemia. “Consider yourself ‘allergic’ to pot smoke,” my friend advised, but “was met with no meaningful hint of understanding or hope for transformative change,” she told me.
I’ve seen cannabinoid hyperemesis syndrome several times in the past few years. It occurs in daily to weekly pot users. Very rarely, it can cause cerebral edema, but it is also associated with seizures and dehydration that can lead to hypovolemic shock and kidney failure.
Heart and brain harm
Then there are the young patients who for various reasons have developed heart failure. Unfortunately, some are repetitively tox screen positive with varying trifectas of methamphetamine (meth), cocaine, and THC; opiates, meth, and THC; alcohol, meth, and THC; or heroin, meth, and THC. THC, the ever present and essential third leg of the stool of stupor. These unfortunate patients often need heart failure medications that they can’t afford or won’t take because illicit drug use is expensive and dulls their ability to prioritize their health. Some desperately need a heart transplant, but the necessary negative drug screen is a pipe dream.
And it’s not just the heart that is affected. There are data linking cannabis use to a higher risk for both ischemic and hemorrhagic stroke. A retrospective study published in Stroke, of more than 1,000 people diagnosed with an aneurysmal subarachnoid hemorrhage, found that more than half of the 46 who tested positive for THC at admission developed delayed cerebral ischemia (DCI), which increases the risk for disability or early death. This was after adjusting for several patient characteristics as well as recent exposure to other illicit substances; cocaine, meth, and tobacco use were not associated with DCI.
Natural my ...
I’m certain my anti-cannabis stance will strike a nerve with those who love their recreational THC and push for its legal sale; after all, “It’s perfectly natural.” But I counter with the fact that tornadoes, earthquakes, cyanide, and appendicitis are all natural but certainly not optimal. And what we are seeing in the vascular specialties is completely unnatural. We are treating a different mix of complications than before pot was readily accessible across several states.
Our most effective action is to educate our patients. We should encourage those who don’t currently smoke cannabis to never start and those who do to quit. People who require marijuana for improved quality of life for terminal care or true (not supposed) disorders that mainstream medicine fails should be approached with empathy and caution.
A good rule of thumb is to never breathe anything you can see. Never put anything in your body that comes off the street: Drug dealers who sell cannabis cut with fentanyl will be ecstatic to take someone’s money then merely keep scrolling when their obituary comes up.
Let’s try to reverse the rise of vascular complications, orthopedic injuries, and vomiting across America. We can start by encouraging our patients to avoid “skunk weed” and get back to the sweet smells of nature in our cities and parks.
Some details have been changed to protect the patients’ identities, but the essence of their diagnoses has been preserved.
Dr. Walton-Shirley is a retired clinical cardiologist from Nashville, Tenn. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Last year, my husband and I took a 16-day road trip from Kentucky through Massachusetts to Maine. On our first morning in Boston, we exited the Park Street Station en route to Boston Common, but instead of being greeted by the aroma of molasses, we were hit full-on with a pungent, repulsive odor. “That’s skunk weed,” my husband chuckled as we stepped right into the middle of the Boston Freedom Rally, a celebration of all things cannabis.
As we boarded a hop-on-hop-off bus, we learned that this was the one week of the year that the city skips testing tour bus drivers for tetrahydrocannabinol (THC), “because we all test positive,” the driver quipped. As our open-air bus circled the Common, a crowd of pot enthusiasts displayed signs in support of relaxed regulation for public consumption.
The 34-year-old Boston Freedom Rally is a sign that U.S. culture has transformed forever. Mary Jane is no friend of emergency physicians nor of staff on hospital wards and offices.
Toking boomers and millennials
Researchers at the University of California, San Diego, looked at cannabis-related emergency department visits from all acute-care hospitals in the state from 2005 to 2019 and found an 1,808% increase in patients aged 65 or older (that is not a typo) who were there for complications from cannabis use.
The lead author said in an interview that, “older patients taking marijuana or related products may have dizziness and falls, heart palpitations, panic attacks, confusion, anxiety or worsening of underlying lung diseases, such as asthma or [chronic obstructive pulmonary disease].”
A recent study from Canada suggests that commercialization has been associated with an increase in related hospitalizations, including cannabis-induced psychosis.
According to a National Study of Drug Use and Health, marijuana use in young adults reached an all-time high (pun intended) in 2021. Nearly 10% of eighth graders and 20% of 10th graders reported using marijuana this past year.
The full downside of any drug, legal or illegal, is largely unknown until it infiltrates the mainstream market, but these are the typical cases we see:
Let’s start with the demotivated high school honors student who dropped out of college to work at the local cinema. He stumbled and broke his clavicle outside a bar at 2 AM, but he wasn’t sure if he passed out, so a cardiology consult was requested to “rule out” arrhythmia associated with syncope. He related that his plan to become a railway conductor had been upended because he knew he would be drug tested and just couldn’t give up pot. After a normal cardiac exam, ECG, labs, a Holter, and an echocardiogram were also requested and normal at a significant cost.
Cannabinoid hyperemesis syndrome
One of my Midwest colleagues related her encounter with two middle-aged pot users with ventricular tachycardia (VT). These episodes coincided with potassium levels less than 3.0 mEq/L in the setting of repetitive vomiting. The QTc interval didn’t normalize despite a corrected potassium level in one patient. They were both informed that they should never smoke pot because vomiting would predictably drop their K+ levels again and prolong their QTc intervals. Then began “the circular argument,” as my friend described it. The patient claims, “I smoke pot to relieve my nausea,” to which she explains that “in many folks, pot use induces nausea.” Of course, the classic reply is, “Not me.” Predictably one of these stoners soon returned with more VT, more puking, and more hypokalemia. “Consider yourself ‘allergic’ to pot smoke,” my friend advised, but “was met with no meaningful hint of understanding or hope for transformative change,” she told me.
I’ve seen cannabinoid hyperemesis syndrome several times in the past few years. It occurs in daily to weekly pot users. Very rarely, it can cause cerebral edema, but it is also associated with seizures and dehydration that can lead to hypovolemic shock and kidney failure.
Heart and brain harm
Then there are the young patients who for various reasons have developed heart failure. Unfortunately, some are repetitively tox screen positive with varying trifectas of methamphetamine (meth), cocaine, and THC; opiates, meth, and THC; alcohol, meth, and THC; or heroin, meth, and THC. THC, the ever present and essential third leg of the stool of stupor. These unfortunate patients often need heart failure medications that they can’t afford or won’t take because illicit drug use is expensive and dulls their ability to prioritize their health. Some desperately need a heart transplant, but the necessary negative drug screen is a pipe dream.
And it’s not just the heart that is affected. There are data linking cannabis use to a higher risk for both ischemic and hemorrhagic stroke. A retrospective study published in Stroke, of more than 1,000 people diagnosed with an aneurysmal subarachnoid hemorrhage, found that more than half of the 46 who tested positive for THC at admission developed delayed cerebral ischemia (DCI), which increases the risk for disability or early death. This was after adjusting for several patient characteristics as well as recent exposure to other illicit substances; cocaine, meth, and tobacco use were not associated with DCI.
Natural my ...
I’m certain my anti-cannabis stance will strike a nerve with those who love their recreational THC and push for its legal sale; after all, “It’s perfectly natural.” But I counter with the fact that tornadoes, earthquakes, cyanide, and appendicitis are all natural but certainly not optimal. And what we are seeing in the vascular specialties is completely unnatural. We are treating a different mix of complications than before pot was readily accessible across several states.
Our most effective action is to educate our patients. We should encourage those who don’t currently smoke cannabis to never start and those who do to quit. People who require marijuana for improved quality of life for terminal care or true (not supposed) disorders that mainstream medicine fails should be approached with empathy and caution.
A good rule of thumb is to never breathe anything you can see. Never put anything in your body that comes off the street: Drug dealers who sell cannabis cut with fentanyl will be ecstatic to take someone’s money then merely keep scrolling when their obituary comes up.
Let’s try to reverse the rise of vascular complications, orthopedic injuries, and vomiting across America. We can start by encouraging our patients to avoid “skunk weed” and get back to the sweet smells of nature in our cities and parks.
Some details have been changed to protect the patients’ identities, but the essence of their diagnoses has been preserved.
Dr. Walton-Shirley is a retired clinical cardiologist from Nashville, Tenn. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Last year, my husband and I took a 16-day road trip from Kentucky through Massachusetts to Maine. On our first morning in Boston, we exited the Park Street Station en route to Boston Common, but instead of being greeted by the aroma of molasses, we were hit full-on with a pungent, repulsive odor. “That’s skunk weed,” my husband chuckled as we stepped right into the middle of the Boston Freedom Rally, a celebration of all things cannabis.
As we boarded a hop-on-hop-off bus, we learned that this was the one week of the year that the city skips testing tour bus drivers for tetrahydrocannabinol (THC), “because we all test positive,” the driver quipped. As our open-air bus circled the Common, a crowd of pot enthusiasts displayed signs in support of relaxed regulation for public consumption.
The 34-year-old Boston Freedom Rally is a sign that U.S. culture has transformed forever. Mary Jane is no friend of emergency physicians nor of staff on hospital wards and offices.
Toking boomers and millennials
Researchers at the University of California, San Diego, looked at cannabis-related emergency department visits from all acute-care hospitals in the state from 2005 to 2019 and found an 1,808% increase in patients aged 65 or older (that is not a typo) who were there for complications from cannabis use.
The lead author said in an interview that, “older patients taking marijuana or related products may have dizziness and falls, heart palpitations, panic attacks, confusion, anxiety or worsening of underlying lung diseases, such as asthma or [chronic obstructive pulmonary disease].”
A recent study from Canada suggests that commercialization has been associated with an increase in related hospitalizations, including cannabis-induced psychosis.
According to a National Study of Drug Use and Health, marijuana use in young adults reached an all-time high (pun intended) in 2021. Nearly 10% of eighth graders and 20% of 10th graders reported using marijuana this past year.
The full downside of any drug, legal or illegal, is largely unknown until it infiltrates the mainstream market, but these are the typical cases we see:
Let’s start with the demotivated high school honors student who dropped out of college to work at the local cinema. He stumbled and broke his clavicle outside a bar at 2 AM, but he wasn’t sure if he passed out, so a cardiology consult was requested to “rule out” arrhythmia associated with syncope. He related that his plan to become a railway conductor had been upended because he knew he would be drug tested and just couldn’t give up pot. After a normal cardiac exam, ECG, labs, a Holter, and an echocardiogram were also requested and normal at a significant cost.
Cannabinoid hyperemesis syndrome
One of my Midwest colleagues related her encounter with two middle-aged pot users with ventricular tachycardia (VT). These episodes coincided with potassium levels less than 3.0 mEq/L in the setting of repetitive vomiting. The QTc interval didn’t normalize despite a corrected potassium level in one patient. They were both informed that they should never smoke pot because vomiting would predictably drop their K+ levels again and prolong their QTc intervals. Then began “the circular argument,” as my friend described it. The patient claims, “I smoke pot to relieve my nausea,” to which she explains that “in many folks, pot use induces nausea.” Of course, the classic reply is, “Not me.” Predictably one of these stoners soon returned with more VT, more puking, and more hypokalemia. “Consider yourself ‘allergic’ to pot smoke,” my friend advised, but “was met with no meaningful hint of understanding or hope for transformative change,” she told me.
I’ve seen cannabinoid hyperemesis syndrome several times in the past few years. It occurs in daily to weekly pot users. Very rarely, it can cause cerebral edema, but it is also associated with seizures and dehydration that can lead to hypovolemic shock and kidney failure.
Heart and brain harm
Then there are the young patients who for various reasons have developed heart failure. Unfortunately, some are repetitively tox screen positive with varying trifectas of methamphetamine (meth), cocaine, and THC; opiates, meth, and THC; alcohol, meth, and THC; or heroin, meth, and THC. THC, the ever present and essential third leg of the stool of stupor. These unfortunate patients often need heart failure medications that they can’t afford or won’t take because illicit drug use is expensive and dulls their ability to prioritize their health. Some desperately need a heart transplant, but the necessary negative drug screen is a pipe dream.
And it’s not just the heart that is affected. There are data linking cannabis use to a higher risk for both ischemic and hemorrhagic stroke. A retrospective study published in Stroke, of more than 1,000 people diagnosed with an aneurysmal subarachnoid hemorrhage, found that more than half of the 46 who tested positive for THC at admission developed delayed cerebral ischemia (DCI), which increases the risk for disability or early death. This was after adjusting for several patient characteristics as well as recent exposure to other illicit substances; cocaine, meth, and tobacco use were not associated with DCI.
Natural my ...
I’m certain my anti-cannabis stance will strike a nerve with those who love their recreational THC and push for its legal sale; after all, “It’s perfectly natural.” But I counter with the fact that tornadoes, earthquakes, cyanide, and appendicitis are all natural but certainly not optimal. And what we are seeing in the vascular specialties is completely unnatural. We are treating a different mix of complications than before pot was readily accessible across several states.
Our most effective action is to educate our patients. We should encourage those who don’t currently smoke cannabis to never start and those who do to quit. People who require marijuana for improved quality of life for terminal care or true (not supposed) disorders that mainstream medicine fails should be approached with empathy and caution.
A good rule of thumb is to never breathe anything you can see. Never put anything in your body that comes off the street: Drug dealers who sell cannabis cut with fentanyl will be ecstatic to take someone’s money then merely keep scrolling when their obituary comes up.
Let’s try to reverse the rise of vascular complications, orthopedic injuries, and vomiting across America. We can start by encouraging our patients to avoid “skunk weed” and get back to the sweet smells of nature in our cities and parks.
Some details have been changed to protect the patients’ identities, but the essence of their diagnoses has been preserved.
Dr. Walton-Shirley is a retired clinical cardiologist from Nashville, Tenn. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Once-weekly topical therapy shows promise for moderate to severe acne
TOPLINE:
METHODOLOGY:
- Poor patient compliance with topical acne therapies is a common clinical challenge.
- In a 12-week, randomized, controlled, phase 2b trial of 181 patients 12 years of age and older, researchers investigated the safety, tolerability, and efficacy of DMT310, a powdered mixture of Spongilla lacustris for treating moderate to severe acne. (In vitro studies have found that components of S. lacustris, a freshwater sponge, have effects that include antimicrobial activity against Cutibacterium acnes and anti-inflammatory activity in human keratinocytes).
- The study’s primary efficacy endpoint was the absolute change in inflammatory lesion count from baseline to week 12.
- Endpoint success was defined as an Investigator Global Assessment (IGA) score of 0 or 1 and at least a two-grade improvement from baseline at week 12.
TAKEAWAY:
- Of the 181 patients, 91 received DMT310 (applied once a week to the face and washed off after 10-15 minutes), and 90 received placebo.
- Patients in the DMT310 arm showed a significantly greater mean reduction in the number of inflammatory lesions at week 12, compared with those in the placebo arm (–15.64 vs. –10.84, respectively; P < .001).
- Similarly, patients in the DMT310 arm showed a significantly greater mean reduction in the number of noninflammatory lesions at week 12, compared with those in the placebo arm (–18.26 vs. –12.41, respectively; P < .001).
- At week 12, endpoint success based on IGA scores also significantly favored patients in the DMT310 arm, compared with those in the placebo arm (44.40% vs. 17.78%; P < .001).
IN PRACTICE:
This study is too preliminary to have practice application. The researchers concluded that the findings “support further study of DMT310 in larger, confirmatory phase 3 trials.”
SOURCE:
Lawrence F. Eichenfield, MD, professor of dermatology and pediatrics at the University of California, San Diego, led the research. The study was published online June 7 in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The analysis did not include an active comparator group and it enrolled a limited number of Asian patients.
DISCLOSURES:
Dr. Eichenfield disclosed that he is a consultant to Dermata, which is developing DMT310, as were three other authors of the study. One author is a company employee. The remaining authors disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Poor patient compliance with topical acne therapies is a common clinical challenge.
- In a 12-week, randomized, controlled, phase 2b trial of 181 patients 12 years of age and older, researchers investigated the safety, tolerability, and efficacy of DMT310, a powdered mixture of Spongilla lacustris for treating moderate to severe acne. (In vitro studies have found that components of S. lacustris, a freshwater sponge, have effects that include antimicrobial activity against Cutibacterium acnes and anti-inflammatory activity in human keratinocytes).
- The study’s primary efficacy endpoint was the absolute change in inflammatory lesion count from baseline to week 12.
- Endpoint success was defined as an Investigator Global Assessment (IGA) score of 0 or 1 and at least a two-grade improvement from baseline at week 12.
TAKEAWAY:
- Of the 181 patients, 91 received DMT310 (applied once a week to the face and washed off after 10-15 minutes), and 90 received placebo.
- Patients in the DMT310 arm showed a significantly greater mean reduction in the number of inflammatory lesions at week 12, compared with those in the placebo arm (–15.64 vs. –10.84, respectively; P < .001).
- Similarly, patients in the DMT310 arm showed a significantly greater mean reduction in the number of noninflammatory lesions at week 12, compared with those in the placebo arm (–18.26 vs. –12.41, respectively; P < .001).
- At week 12, endpoint success based on IGA scores also significantly favored patients in the DMT310 arm, compared with those in the placebo arm (44.40% vs. 17.78%; P < .001).
IN PRACTICE:
This study is too preliminary to have practice application. The researchers concluded that the findings “support further study of DMT310 in larger, confirmatory phase 3 trials.”
SOURCE:
Lawrence F. Eichenfield, MD, professor of dermatology and pediatrics at the University of California, San Diego, led the research. The study was published online June 7 in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The analysis did not include an active comparator group and it enrolled a limited number of Asian patients.
DISCLOSURES:
Dr. Eichenfield disclosed that he is a consultant to Dermata, which is developing DMT310, as were three other authors of the study. One author is a company employee. The remaining authors disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Poor patient compliance with topical acne therapies is a common clinical challenge.
- In a 12-week, randomized, controlled, phase 2b trial of 181 patients 12 years of age and older, researchers investigated the safety, tolerability, and efficacy of DMT310, a powdered mixture of Spongilla lacustris for treating moderate to severe acne. (In vitro studies have found that components of S. lacustris, a freshwater sponge, have effects that include antimicrobial activity against Cutibacterium acnes and anti-inflammatory activity in human keratinocytes).
- The study’s primary efficacy endpoint was the absolute change in inflammatory lesion count from baseline to week 12.
- Endpoint success was defined as an Investigator Global Assessment (IGA) score of 0 or 1 and at least a two-grade improvement from baseline at week 12.
TAKEAWAY:
- Of the 181 patients, 91 received DMT310 (applied once a week to the face and washed off after 10-15 minutes), and 90 received placebo.
- Patients in the DMT310 arm showed a significantly greater mean reduction in the number of inflammatory lesions at week 12, compared with those in the placebo arm (–15.64 vs. –10.84, respectively; P < .001).
- Similarly, patients in the DMT310 arm showed a significantly greater mean reduction in the number of noninflammatory lesions at week 12, compared with those in the placebo arm (–18.26 vs. –12.41, respectively; P < .001).
- At week 12, endpoint success based on IGA scores also significantly favored patients in the DMT310 arm, compared with those in the placebo arm (44.40% vs. 17.78%; P < .001).
IN PRACTICE:
This study is too preliminary to have practice application. The researchers concluded that the findings “support further study of DMT310 in larger, confirmatory phase 3 trials.”
SOURCE:
Lawrence F. Eichenfield, MD, professor of dermatology and pediatrics at the University of California, San Diego, led the research. The study was published online June 7 in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The analysis did not include an active comparator group and it enrolled a limited number of Asian patients.
DISCLOSURES:
Dr. Eichenfield disclosed that he is a consultant to Dermata, which is developing DMT310, as were three other authors of the study. One author is a company employee. The remaining authors disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Artificial intelligence in the office: Part 2
In the year since generative artificial intelligence (AI) software first began to emerge for use, the staggering pace and breadth of development has condensed years of growth and change into months and weeks. Among the settings where these tools may find the greatest straight-line relevance is private medical practice.
. A multitude of generative AI products with potential medical applications are now available, with new ones appearing almost weekly. (As always, I have no financial interest in any product or service mentioned in this column.)
Last month, I discussed ChatGPT, the best-known AI algorithm, and some of its applications in clinical practice, such as generating website, video, and blog content. ChatGPT can also provide rapid and concise answers to general medical questions, like a search engine – but with more natural language processing and contextual understanding. Additionally, the algorithm can draft generic medical documents, including templates for after-visit summaries, postprocedure instructions, referrals, prior authorization appeal letters, and educational handouts.
Another useful feature of ChatGPT is its ability to provide accurate and conversational language translations, thus serving as an interpreter during clinic visits in situations where a human translator is not available. It also has potential uses in clinical research by finding resources, formulating hypotheses, drafting study protocols, and collecting large amounts of data in short periods of time. Other possibilities include survey administration, clinical trial recruitment, and automatic medication monitoring.
GPT-4, the latest version of ChatGPT, is reported to have greater problem-solving abilities and an even broader knowledge base. Among its claimed skills are the ability to find the latest literature in a given area, write a discharge summary for a patient following an uncomplicated surgery, and an image analysis feature to identify objects in photos. GPT-4 has been praised as having “the potential to help drive medical innovation, from aiding with patient discharge notes, summarizing recent clinical trials, providing information on ethical guidelines, and much more.”
Bard, an AI “chat bot” introduced by Google earlier this year, intends to leverage Google’s enormous database to compete with ChatGPT in providing answers to medical questions. Bard also hopes to play a pivotal role in expanding telemedicine and remote care via Google’s secure connections and access to patient records and medical history, and “facilitate seamless communication through appointment scheduling, messaging, and sharing medical images,” according to PackT, a website for IT professionals. The company claims that Bard’s integration of AI and machine learning capabilities will serve to elevate health care efficiency and patient outcomes, PackT says, and “the platform’s AI system quickly and accurately analyzes patient records, identifies patterns and trends, and aids medical professionals in developing effective treatment plans.”
Doximity has introduced an AI engine called DocsGPT, an encrypted, HIPAA-compliant writing assistant that, the company says, can draft any form of professional correspondence, including prior authorization letters, insurance appeals, patient support letters, and patient education materials. The service is available at no charge to all U.S. physicians and medical students through their Doximity accounts.
Microsoft has introduced several AI products. BioGPT is a language model specifically designed for health care. Compared with GPT models that are trained on more general text data, BioGPT is purported to have a deeper understanding of the language used in biomedical research and can generate more accurate and relevant outputs for biomedical tasks, such as drug discovery, disease classification, and clinical decision support. Fabric is another health care–specific data and analytics platform the company described in an announcement in May. It can combine data from sources such as electronic health records, images, lab systems, medical devices, and claims systems so hospitals and offices can standardize it and access it in the same place. Microsoft said the new tools will help eliminate the “time-consuming” process of searching through these sources one by one. Microsoft will also offer a new generative AI chatbot called the Azure Health Bot, which can pull information from a health organization’s own internal data as well as reputable external sources such as the Food and Drug Administration and the National Institutes of Health.
Several other AI products are available for clinicians. Tana served as an administrative aid and a clinical helper during the height of the COVID-19 pandemic, answering frequently asked questions, facilitating appointment management, and gathering preliminary medical information prior to teleconsultations. Dougall GPT is another AI chatbot tailored for health care professionals. It provides clinicians with AI-tuned answers to their queries, augmented by links to relevant, up-to-date, authoritative resources. It also assists in drafting patient instructions, consultation summaries, speeches, and professional correspondence. Wang has created Clinical Camel, an open-source health care–focused chatbot that assembles medical data with a combination of user-shared conversations and synthetic conversations derived from curated clinical articles. The Chinese company Baidu has rolled out Ernie as a potential rival to ChatGPT. You get the idea.
Of course, the inherent drawbacks of AI, such as producing false or biased information, perpetuating harmful stereotypes, and presenting information that has since been proven inaccurate or out-of-date, must always be kept in mind. All AI algorithms have been criticized for giving wrong answers, as their datasets are generally culled from information published in 2021 or earlier. Several of them have been shown to fabricate information – a phenomenon labeled “artificial hallucinations” in one article. “The scientific community must be vigilant in verifying the accuracy and reliability of the information provided by AI tools,” wrote the authors of that paper. “Researchers should use AI as an aid rather than a replacement for critical thinking and fact-checking.”
In the year since generative artificial intelligence (AI) software first began to emerge for use, the staggering pace and breadth of development has condensed years of growth and change into months and weeks. Among the settings where these tools may find the greatest straight-line relevance is private medical practice.
. A multitude of generative AI products with potential medical applications are now available, with new ones appearing almost weekly. (As always, I have no financial interest in any product or service mentioned in this column.)
Last month, I discussed ChatGPT, the best-known AI algorithm, and some of its applications in clinical practice, such as generating website, video, and blog content. ChatGPT can also provide rapid and concise answers to general medical questions, like a search engine – but with more natural language processing and contextual understanding. Additionally, the algorithm can draft generic medical documents, including templates for after-visit summaries, postprocedure instructions, referrals, prior authorization appeal letters, and educational handouts.
Another useful feature of ChatGPT is its ability to provide accurate and conversational language translations, thus serving as an interpreter during clinic visits in situations where a human translator is not available. It also has potential uses in clinical research by finding resources, formulating hypotheses, drafting study protocols, and collecting large amounts of data in short periods of time. Other possibilities include survey administration, clinical trial recruitment, and automatic medication monitoring.
GPT-4, the latest version of ChatGPT, is reported to have greater problem-solving abilities and an even broader knowledge base. Among its claimed skills are the ability to find the latest literature in a given area, write a discharge summary for a patient following an uncomplicated surgery, and an image analysis feature to identify objects in photos. GPT-4 has been praised as having “the potential to help drive medical innovation, from aiding with patient discharge notes, summarizing recent clinical trials, providing information on ethical guidelines, and much more.”
Bard, an AI “chat bot” introduced by Google earlier this year, intends to leverage Google’s enormous database to compete with ChatGPT in providing answers to medical questions. Bard also hopes to play a pivotal role in expanding telemedicine and remote care via Google’s secure connections and access to patient records and medical history, and “facilitate seamless communication through appointment scheduling, messaging, and sharing medical images,” according to PackT, a website for IT professionals. The company claims that Bard’s integration of AI and machine learning capabilities will serve to elevate health care efficiency and patient outcomes, PackT says, and “the platform’s AI system quickly and accurately analyzes patient records, identifies patterns and trends, and aids medical professionals in developing effective treatment plans.”
Doximity has introduced an AI engine called DocsGPT, an encrypted, HIPAA-compliant writing assistant that, the company says, can draft any form of professional correspondence, including prior authorization letters, insurance appeals, patient support letters, and patient education materials. The service is available at no charge to all U.S. physicians and medical students through their Doximity accounts.
Microsoft has introduced several AI products. BioGPT is a language model specifically designed for health care. Compared with GPT models that are trained on more general text data, BioGPT is purported to have a deeper understanding of the language used in biomedical research and can generate more accurate and relevant outputs for biomedical tasks, such as drug discovery, disease classification, and clinical decision support. Fabric is another health care–specific data and analytics platform the company described in an announcement in May. It can combine data from sources such as electronic health records, images, lab systems, medical devices, and claims systems so hospitals and offices can standardize it and access it in the same place. Microsoft said the new tools will help eliminate the “time-consuming” process of searching through these sources one by one. Microsoft will also offer a new generative AI chatbot called the Azure Health Bot, which can pull information from a health organization’s own internal data as well as reputable external sources such as the Food and Drug Administration and the National Institutes of Health.
Several other AI products are available for clinicians. Tana served as an administrative aid and a clinical helper during the height of the COVID-19 pandemic, answering frequently asked questions, facilitating appointment management, and gathering preliminary medical information prior to teleconsultations. Dougall GPT is another AI chatbot tailored for health care professionals. It provides clinicians with AI-tuned answers to their queries, augmented by links to relevant, up-to-date, authoritative resources. It also assists in drafting patient instructions, consultation summaries, speeches, and professional correspondence. Wang has created Clinical Camel, an open-source health care–focused chatbot that assembles medical data with a combination of user-shared conversations and synthetic conversations derived from curated clinical articles. The Chinese company Baidu has rolled out Ernie as a potential rival to ChatGPT. You get the idea.
Of course, the inherent drawbacks of AI, such as producing false or biased information, perpetuating harmful stereotypes, and presenting information that has since been proven inaccurate or out-of-date, must always be kept in mind. All AI algorithms have been criticized for giving wrong answers, as their datasets are generally culled from information published in 2021 or earlier. Several of them have been shown to fabricate information – a phenomenon labeled “artificial hallucinations” in one article. “The scientific community must be vigilant in verifying the accuracy and reliability of the information provided by AI tools,” wrote the authors of that paper. “Researchers should use AI as an aid rather than a replacement for critical thinking and fact-checking.”
In the year since generative artificial intelligence (AI) software first began to emerge for use, the staggering pace and breadth of development has condensed years of growth and change into months and weeks. Among the settings where these tools may find the greatest straight-line relevance is private medical practice.
. A multitude of generative AI products with potential medical applications are now available, with new ones appearing almost weekly. (As always, I have no financial interest in any product or service mentioned in this column.)
Last month, I discussed ChatGPT, the best-known AI algorithm, and some of its applications in clinical practice, such as generating website, video, and blog content. ChatGPT can also provide rapid and concise answers to general medical questions, like a search engine – but with more natural language processing and contextual understanding. Additionally, the algorithm can draft generic medical documents, including templates for after-visit summaries, postprocedure instructions, referrals, prior authorization appeal letters, and educational handouts.
Another useful feature of ChatGPT is its ability to provide accurate and conversational language translations, thus serving as an interpreter during clinic visits in situations where a human translator is not available. It also has potential uses in clinical research by finding resources, formulating hypotheses, drafting study protocols, and collecting large amounts of data in short periods of time. Other possibilities include survey administration, clinical trial recruitment, and automatic medication monitoring.
GPT-4, the latest version of ChatGPT, is reported to have greater problem-solving abilities and an even broader knowledge base. Among its claimed skills are the ability to find the latest literature in a given area, write a discharge summary for a patient following an uncomplicated surgery, and an image analysis feature to identify objects in photos. GPT-4 has been praised as having “the potential to help drive medical innovation, from aiding with patient discharge notes, summarizing recent clinical trials, providing information on ethical guidelines, and much more.”
Bard, an AI “chat bot” introduced by Google earlier this year, intends to leverage Google’s enormous database to compete with ChatGPT in providing answers to medical questions. Bard also hopes to play a pivotal role in expanding telemedicine and remote care via Google’s secure connections and access to patient records and medical history, and “facilitate seamless communication through appointment scheduling, messaging, and sharing medical images,” according to PackT, a website for IT professionals. The company claims that Bard’s integration of AI and machine learning capabilities will serve to elevate health care efficiency and patient outcomes, PackT says, and “the platform’s AI system quickly and accurately analyzes patient records, identifies patterns and trends, and aids medical professionals in developing effective treatment plans.”
Doximity has introduced an AI engine called DocsGPT, an encrypted, HIPAA-compliant writing assistant that, the company says, can draft any form of professional correspondence, including prior authorization letters, insurance appeals, patient support letters, and patient education materials. The service is available at no charge to all U.S. physicians and medical students through their Doximity accounts.
Microsoft has introduced several AI products. BioGPT is a language model specifically designed for health care. Compared with GPT models that are trained on more general text data, BioGPT is purported to have a deeper understanding of the language used in biomedical research and can generate more accurate and relevant outputs for biomedical tasks, such as drug discovery, disease classification, and clinical decision support. Fabric is another health care–specific data and analytics platform the company described in an announcement in May. It can combine data from sources such as electronic health records, images, lab systems, medical devices, and claims systems so hospitals and offices can standardize it and access it in the same place. Microsoft said the new tools will help eliminate the “time-consuming” process of searching through these sources one by one. Microsoft will also offer a new generative AI chatbot called the Azure Health Bot, which can pull information from a health organization’s own internal data as well as reputable external sources such as the Food and Drug Administration and the National Institutes of Health.
Several other AI products are available for clinicians. Tana served as an administrative aid and a clinical helper during the height of the COVID-19 pandemic, answering frequently asked questions, facilitating appointment management, and gathering preliminary medical information prior to teleconsultations. Dougall GPT is another AI chatbot tailored for health care professionals. It provides clinicians with AI-tuned answers to their queries, augmented by links to relevant, up-to-date, authoritative resources. It also assists in drafting patient instructions, consultation summaries, speeches, and professional correspondence. Wang has created Clinical Camel, an open-source health care–focused chatbot that assembles medical data with a combination of user-shared conversations and synthetic conversations derived from curated clinical articles. The Chinese company Baidu has rolled out Ernie as a potential rival to ChatGPT. You get the idea.
Of course, the inherent drawbacks of AI, such as producing false or biased information, perpetuating harmful stereotypes, and presenting information that has since been proven inaccurate or out-of-date, must always be kept in mind. All AI algorithms have been criticized for giving wrong answers, as their datasets are generally culled from information published in 2021 or earlier. Several of them have been shown to fabricate information – a phenomenon labeled “artificial hallucinations” in one article. “The scientific community must be vigilant in verifying the accuracy and reliability of the information provided by AI tools,” wrote the authors of that paper. “Researchers should use AI as an aid rather than a replacement for critical thinking and fact-checking.”
Prior authorization software: Saves time but hurdles remain
New England Baptist Hospital has been grappling with a serious problem facing health care today: insurers demanding prior authorizations for services ordered by physicians. Meeting payers’ requirements eats up time, delays treatment, and can be a costly drain on doctors’ practices.
To deal with this problem, the Boston orthopedic hospital has opted to automate submission of prior authorization requests on behalf of more than 100 mostly orthopedic surgeons on staff.
After 5 years using this system, “we can say that automation definitely works,” said Lidiya Hadzhieva, director of patient access at the hospital. The software has reduced write-offs by 30% and staff costs by 25%. Prior authorization gets approved 3 days after scheduling, compared with 11 days previously, she said.
“This software not only saves staff time, but it can also more accurately predict when prior authorization is needed,” she added.
For practices deluged with required prior authorizations by insurers, automation is emerging as a way for practices to make the process less time-consuming and save money. However, the software can be costly and may not be adoptable to many practices, and many physicians are not even aware it exists.
So far, the software is mainly used at large organizations like hospital systems. But as word gets out and the software becomes easier to use, private practices and other smaller entities may join the automation trend.
There is definitely a need to automate prior authorization. The American Medical Association reports that physicians spend 16 hours per week on prior authorizations. In a recent AMA survey, more than 60% of physicians indicated that it’s difficult to know when prior authorization is needed. And 93% of physicians reported care delays while waiting for authorization, the AMA said.
Experts estimate that 80% of prior authorization work could be automated, but most practices still use the phone or fax, even as numbers of prior authorizations continue to increase.
How it works
Automation software connects directly to the practice’s electronic health record (EHR). “When the doctor places an order in the EHR, the process starts automatically,” Ms. Hadzhieva said. “The doctor may not even notice it.”
In addition to using an EHR connection, many software products can communicate with the payer through its portal or by fax or phone, while still automating other parts of the process.
The software’s first step is to decide whether prior authorization is needed. This requires having an updated list of the rules that each payer uses for prior authorization. Manually keeping track of payer rules is very time-consuming, but automation uses bots to visit each payer site to look for rules changes. One vendor, Infinitus, uses a voice-based bot called Eva that calls up each payer and speaks with a representative.
“Automatically updating payer rules is not a new technology,” said YiDing Yu, MD, chief product officer at Olive, the automation vendor for New England Baptist. “What is new in the last 5 years is extracting the information needed for the prior authorization out of the clinical notes.”
This is challenging because each doctor has different ways to describe each step of clinical work. To identify this shorthand, Dr. Yu said Olive uses natural language processing, which is a form of artificial intelligence that learns how each doctor describes things.
Dr. Yu asserts that Olive is actually better than a practice’s staff at digging out clinical information. She said staff without much clinical training may miss terms that the software can catch, and they don’t have the time to go back many months into the record to find valuable information. But automation can do that.
In some instances, however, the software may not be able to find the information, in which case it alerts staff through a prompt in the EHR and the information is retrieved manually, Dr. Yu said.
Next, the Olive software puts the information it found into the request form and sends it to the payer. After submission, the software constantly checks on the status of each request, again visiting payer sites with a bot.
At New England Baptist, the software is used mainly by physicians in fairly small private practices who are on staff. They are using the software on the hospital’s dime, but it only works inside the hospital, Ms. Hadzhieva said. For their work outside of the hospital, they would have to purchase the Olive software on their own, she said.
Automation hasn’t spread to practices yet
Despite the promising outcomes for products like Olive, automation software is still primarily used by large organizations. Vendors say very few private practices have bought it yet. “The technology works, but it is still in the early-adopter phase,” Dr. Yu said.
For one thing, the software can be expensive. Very few vendors reveal their prices, but Dr. Yu did so. She said Olive normally costs about $50,000 a year for even a small organization. She insisted, however, that the savings from avoiding just one denial each month for a hip surgery would justify the expense.
On the other hand, some automation software is free, such as the Surescripts product for prior authorization of prescriptions. But it is unclear whether Surescripts does as much as Olive. Vendors’ descriptions of their products tend to be vague.
Also, Surescripts and Olive have entirely separate functions. Dr. Yu said Olive is limited to procedures, so it benefits specialties like oncology, neurosurgery, colorectal surgery, vascular surgery, and cardiology. Olive does not cover prescriptions, because they operate on a different technology.
Dr. Yu said another hurdle for adopting the software is the kind of EHR systems that doctors use. At this point, only a few EHR systems – such as Epic, Cerner, and Athena – are compatible with Olive. Large organizations tend to use Epic and Cerner, while many practices often use Athena or a variety of other systems, she said.
Despite stunted demand, there is no shortage of companies offering automation software for medical (that is, non-prescription) prior authorization. One compilation lists 25 such vendors, including companies like Myndshft, Rhyme, Infinitus, Infinx, and Waystar. As with any start-up technology, companies occasionally buy each other out.
In addition to issues like cost, specialty, and EHR compatibility, another hurdle is that few doctors even know the technology exists. Vendors say marketing focuses on larger provider organizations, not smaller practices.
Even many tech-savvy doctors, like Adam Bruggeman, MD, an orthopedist and CEO of Texas Spine Care Center in San Antonio, say they know little about the technology. “There is definitely a need to automate prior authorization,” he said. “But I don’t know of any colleagues who use it.” He has only just begun to explore vendors, he said.
Many medical practice consultants also have not yet explored the technology. “Automation makes a lot of sense, because there are a lot of repetitive tasks in prior authorization,” said Jill Arena, CEO of Portland, Ore.–based Health e Practices. “But I haven’t looked into it yet, and none of my clients has even asked about it.”
“I could see how it could be an easier sell for large organizations,” she added. “They have an IT person and a CFO who can explore the issue. Smaller practices usually don’t have that kind of expertise.”
Where does automation go from here?
Until now, clinicians who want to fully automate prior authorizations would have to buy two products – one for medical procedures and one for prescriptions. This has to do with incompatible electronic transmission standards, which are used to digitize information, said Susan Lawson-Dawson, content marketing strategist for the vendor Myndshft Health.
Myndshft has long been selling automation software for medical prior authorizations, but now it is introducing a product for prescriptions, Ms. Lawson-Dawson said. She said Myndshft will then be the only vendor to automate both kinds of prior authorizations.
Ms. Lawson-Dawson said Myndshft has 685 customers to date and is looking for more business. Recently the company entered the Google Cloud Marketplace. Google Cloud customers can now direct their committed spend with Google to purchasing Myndshft, meaning they could get it at a discount.
Software like Olive and Myndshft can operate independently of payers, but a vendor called Rhyme depends on payers for its software to function, said Rhyme CEO Joe Anstine. He said more than 300 payers have agreed to install the Rhyme system, and Rhyme has signed up a number of large health systems to use the product. Initially, he said, clinicians paid for the service, but now Rhyme is beginning to find payers to foot the costs and to let clinicians use it for free, which would open Rhyme up to smaller practices.
EHR companies themselves are beginning to offer automation, too. Epic, for example, has created a tool for prior authorization as part of its Epic Payer Platform. Like Rhyme, it requires payer cooperation, because information goes back and forth between clinician and payer in what is called bi-directional exchange.
The Epic product is still in its pilot phase. Epic reported that several large health systems were using its product in conjunction with a specific payer – for instance, Mayo Clinic with Blue Cross and Blue Shield of Minnesota and Ochsner Health with Humana. According to Epic, the arrangement reduced Mayo’s denials due to additional documentation requests by 63% for professional billing.
Automating with just one payer still means the clinician has to deal with manual processes at other payers, but a large clinician could have sufficient volume with that one payer to make the arrangement useful.
Will payers automate prior authorization?
Ultimately, payers may take the automation business away from vendors, offering a free product to all clinicians. But don’t hold your breath. Payers first have to rebuild their electronic systems to accommodate an electronic connection with providers. Even then, some payers might hold back from automating, forcing practices to continue manually processing some prior authorizations.
Efforts are underway, however, to mandate payers to support prior authorization automation. For this to happen, payers would have to revamp their data so that it could be easily read by practices’ EHRs. This would mean adopting a specific interoperability standard called Health Level 7 Fast Healthcare Interoperability Resources (FHIR).
Toward this goal, the Centers for Medicare & Medicaid Services proposes to require payers to adopt FHIR by January 2026. (CMS still has to finalize the rule.) Experts say the two-year ramp-up time is needed because it takes extensive work for payers to translate their data into FHIR.
The only payer so far to switch to FHIR for prior authorization is Regence in Washington state. In a pilot project, it has automated prior authorization with just one provider, MultiCare Connected Care, an accountable care organization (ACO), also in Washington state.
Anna Taylor, associate vice president of population health and value-based care at MultiCare, explained how the arrangement works. “Two separate entities are sharing one operational process,” she told this news organization. “That means they can have a digital conversation back and forth, so it is much easier to resolve prior authorization issues.”
Unlike many vendor products, the Regence service is free. And while the vendors market only to large organizations, most doctors in the MultiCare arrangement are in independent practices. Ms. Taylor said these doctors have been “enthusiastic” about the arrangement.
The results of the pilot are impressive. Ms. Taylor said automation has resulted in a 233% productivity gain for MultiCare clinicians, and 89% of submissions to Regence get an immediate response.
There is a potential downside, however, to working directly with payers. A direct connection to clinicians allows payers to access the doctor’s clinical notes, which could make many doctors uneasy. But Ms. Taylor said Regence only has access to the “discrete data fields” on MultiCare’s EHR dashboard, not to the notes themselves.
The ultimate goal of the Regence-Multicare project is to include more payers and clinicians. Ms. Taylor said two of the 27 other payers that MultiCare works with are “highly interested,” but it would take a lot of work for them to get connected with practices and other clinicians.
Ultimately, payers could offer automation and third-party vendors might then fade away. However, physicians may resist working directly with payers if the arrangement requires full access to their medical records.
A version of this article first appeared on Medscape.com.
New England Baptist Hospital has been grappling with a serious problem facing health care today: insurers demanding prior authorizations for services ordered by physicians. Meeting payers’ requirements eats up time, delays treatment, and can be a costly drain on doctors’ practices.
To deal with this problem, the Boston orthopedic hospital has opted to automate submission of prior authorization requests on behalf of more than 100 mostly orthopedic surgeons on staff.
After 5 years using this system, “we can say that automation definitely works,” said Lidiya Hadzhieva, director of patient access at the hospital. The software has reduced write-offs by 30% and staff costs by 25%. Prior authorization gets approved 3 days after scheduling, compared with 11 days previously, she said.
“This software not only saves staff time, but it can also more accurately predict when prior authorization is needed,” she added.
For practices deluged with required prior authorizations by insurers, automation is emerging as a way for practices to make the process less time-consuming and save money. However, the software can be costly and may not be adoptable to many practices, and many physicians are not even aware it exists.
So far, the software is mainly used at large organizations like hospital systems. But as word gets out and the software becomes easier to use, private practices and other smaller entities may join the automation trend.
There is definitely a need to automate prior authorization. The American Medical Association reports that physicians spend 16 hours per week on prior authorizations. In a recent AMA survey, more than 60% of physicians indicated that it’s difficult to know when prior authorization is needed. And 93% of physicians reported care delays while waiting for authorization, the AMA said.
Experts estimate that 80% of prior authorization work could be automated, but most practices still use the phone or fax, even as numbers of prior authorizations continue to increase.
How it works
Automation software connects directly to the practice’s electronic health record (EHR). “When the doctor places an order in the EHR, the process starts automatically,” Ms. Hadzhieva said. “The doctor may not even notice it.”
In addition to using an EHR connection, many software products can communicate with the payer through its portal or by fax or phone, while still automating other parts of the process.
The software’s first step is to decide whether prior authorization is needed. This requires having an updated list of the rules that each payer uses for prior authorization. Manually keeping track of payer rules is very time-consuming, but automation uses bots to visit each payer site to look for rules changes. One vendor, Infinitus, uses a voice-based bot called Eva that calls up each payer and speaks with a representative.
“Automatically updating payer rules is not a new technology,” said YiDing Yu, MD, chief product officer at Olive, the automation vendor for New England Baptist. “What is new in the last 5 years is extracting the information needed for the prior authorization out of the clinical notes.”
This is challenging because each doctor has different ways to describe each step of clinical work. To identify this shorthand, Dr. Yu said Olive uses natural language processing, which is a form of artificial intelligence that learns how each doctor describes things.
Dr. Yu asserts that Olive is actually better than a practice’s staff at digging out clinical information. She said staff without much clinical training may miss terms that the software can catch, and they don’t have the time to go back many months into the record to find valuable information. But automation can do that.
In some instances, however, the software may not be able to find the information, in which case it alerts staff through a prompt in the EHR and the information is retrieved manually, Dr. Yu said.
Next, the Olive software puts the information it found into the request form and sends it to the payer. After submission, the software constantly checks on the status of each request, again visiting payer sites with a bot.
At New England Baptist, the software is used mainly by physicians in fairly small private practices who are on staff. They are using the software on the hospital’s dime, but it only works inside the hospital, Ms. Hadzhieva said. For their work outside of the hospital, they would have to purchase the Olive software on their own, she said.
Automation hasn’t spread to practices yet
Despite the promising outcomes for products like Olive, automation software is still primarily used by large organizations. Vendors say very few private practices have bought it yet. “The technology works, but it is still in the early-adopter phase,” Dr. Yu said.
For one thing, the software can be expensive. Very few vendors reveal their prices, but Dr. Yu did so. She said Olive normally costs about $50,000 a year for even a small organization. She insisted, however, that the savings from avoiding just one denial each month for a hip surgery would justify the expense.
On the other hand, some automation software is free, such as the Surescripts product for prior authorization of prescriptions. But it is unclear whether Surescripts does as much as Olive. Vendors’ descriptions of their products tend to be vague.
Also, Surescripts and Olive have entirely separate functions. Dr. Yu said Olive is limited to procedures, so it benefits specialties like oncology, neurosurgery, colorectal surgery, vascular surgery, and cardiology. Olive does not cover prescriptions, because they operate on a different technology.
Dr. Yu said another hurdle for adopting the software is the kind of EHR systems that doctors use. At this point, only a few EHR systems – such as Epic, Cerner, and Athena – are compatible with Olive. Large organizations tend to use Epic and Cerner, while many practices often use Athena or a variety of other systems, she said.
Despite stunted demand, there is no shortage of companies offering automation software for medical (that is, non-prescription) prior authorization. One compilation lists 25 such vendors, including companies like Myndshft, Rhyme, Infinitus, Infinx, and Waystar. As with any start-up technology, companies occasionally buy each other out.
In addition to issues like cost, specialty, and EHR compatibility, another hurdle is that few doctors even know the technology exists. Vendors say marketing focuses on larger provider organizations, not smaller practices.
Even many tech-savvy doctors, like Adam Bruggeman, MD, an orthopedist and CEO of Texas Spine Care Center in San Antonio, say they know little about the technology. “There is definitely a need to automate prior authorization,” he said. “But I don’t know of any colleagues who use it.” He has only just begun to explore vendors, he said.
Many medical practice consultants also have not yet explored the technology. “Automation makes a lot of sense, because there are a lot of repetitive tasks in prior authorization,” said Jill Arena, CEO of Portland, Ore.–based Health e Practices. “But I haven’t looked into it yet, and none of my clients has even asked about it.”
“I could see how it could be an easier sell for large organizations,” she added. “They have an IT person and a CFO who can explore the issue. Smaller practices usually don’t have that kind of expertise.”
Where does automation go from here?
Until now, clinicians who want to fully automate prior authorizations would have to buy two products – one for medical procedures and one for prescriptions. This has to do with incompatible electronic transmission standards, which are used to digitize information, said Susan Lawson-Dawson, content marketing strategist for the vendor Myndshft Health.
Myndshft has long been selling automation software for medical prior authorizations, but now it is introducing a product for prescriptions, Ms. Lawson-Dawson said. She said Myndshft will then be the only vendor to automate both kinds of prior authorizations.
Ms. Lawson-Dawson said Myndshft has 685 customers to date and is looking for more business. Recently the company entered the Google Cloud Marketplace. Google Cloud customers can now direct their committed spend with Google to purchasing Myndshft, meaning they could get it at a discount.
Software like Olive and Myndshft can operate independently of payers, but a vendor called Rhyme depends on payers for its software to function, said Rhyme CEO Joe Anstine. He said more than 300 payers have agreed to install the Rhyme system, and Rhyme has signed up a number of large health systems to use the product. Initially, he said, clinicians paid for the service, but now Rhyme is beginning to find payers to foot the costs and to let clinicians use it for free, which would open Rhyme up to smaller practices.
EHR companies themselves are beginning to offer automation, too. Epic, for example, has created a tool for prior authorization as part of its Epic Payer Platform. Like Rhyme, it requires payer cooperation, because information goes back and forth between clinician and payer in what is called bi-directional exchange.
The Epic product is still in its pilot phase. Epic reported that several large health systems were using its product in conjunction with a specific payer – for instance, Mayo Clinic with Blue Cross and Blue Shield of Minnesota and Ochsner Health with Humana. According to Epic, the arrangement reduced Mayo’s denials due to additional documentation requests by 63% for professional billing.
Automating with just one payer still means the clinician has to deal with manual processes at other payers, but a large clinician could have sufficient volume with that one payer to make the arrangement useful.
Will payers automate prior authorization?
Ultimately, payers may take the automation business away from vendors, offering a free product to all clinicians. But don’t hold your breath. Payers first have to rebuild their electronic systems to accommodate an electronic connection with providers. Even then, some payers might hold back from automating, forcing practices to continue manually processing some prior authorizations.
Efforts are underway, however, to mandate payers to support prior authorization automation. For this to happen, payers would have to revamp their data so that it could be easily read by practices’ EHRs. This would mean adopting a specific interoperability standard called Health Level 7 Fast Healthcare Interoperability Resources (FHIR).
Toward this goal, the Centers for Medicare & Medicaid Services proposes to require payers to adopt FHIR by January 2026. (CMS still has to finalize the rule.) Experts say the two-year ramp-up time is needed because it takes extensive work for payers to translate their data into FHIR.
The only payer so far to switch to FHIR for prior authorization is Regence in Washington state. In a pilot project, it has automated prior authorization with just one provider, MultiCare Connected Care, an accountable care organization (ACO), also in Washington state.
Anna Taylor, associate vice president of population health and value-based care at MultiCare, explained how the arrangement works. “Two separate entities are sharing one operational process,” she told this news organization. “That means they can have a digital conversation back and forth, so it is much easier to resolve prior authorization issues.”
Unlike many vendor products, the Regence service is free. And while the vendors market only to large organizations, most doctors in the MultiCare arrangement are in independent practices. Ms. Taylor said these doctors have been “enthusiastic” about the arrangement.
The results of the pilot are impressive. Ms. Taylor said automation has resulted in a 233% productivity gain for MultiCare clinicians, and 89% of submissions to Regence get an immediate response.
There is a potential downside, however, to working directly with payers. A direct connection to clinicians allows payers to access the doctor’s clinical notes, which could make many doctors uneasy. But Ms. Taylor said Regence only has access to the “discrete data fields” on MultiCare’s EHR dashboard, not to the notes themselves.
The ultimate goal of the Regence-Multicare project is to include more payers and clinicians. Ms. Taylor said two of the 27 other payers that MultiCare works with are “highly interested,” but it would take a lot of work for them to get connected with practices and other clinicians.
Ultimately, payers could offer automation and third-party vendors might then fade away. However, physicians may resist working directly with payers if the arrangement requires full access to their medical records.
A version of this article first appeared on Medscape.com.
New England Baptist Hospital has been grappling with a serious problem facing health care today: insurers demanding prior authorizations for services ordered by physicians. Meeting payers’ requirements eats up time, delays treatment, and can be a costly drain on doctors’ practices.
To deal with this problem, the Boston orthopedic hospital has opted to automate submission of prior authorization requests on behalf of more than 100 mostly orthopedic surgeons on staff.
After 5 years using this system, “we can say that automation definitely works,” said Lidiya Hadzhieva, director of patient access at the hospital. The software has reduced write-offs by 30% and staff costs by 25%. Prior authorization gets approved 3 days after scheduling, compared with 11 days previously, she said.
“This software not only saves staff time, but it can also more accurately predict when prior authorization is needed,” she added.
For practices deluged with required prior authorizations by insurers, automation is emerging as a way for practices to make the process less time-consuming and save money. However, the software can be costly and may not be adoptable to many practices, and many physicians are not even aware it exists.
So far, the software is mainly used at large organizations like hospital systems. But as word gets out and the software becomes easier to use, private practices and other smaller entities may join the automation trend.
There is definitely a need to automate prior authorization. The American Medical Association reports that physicians spend 16 hours per week on prior authorizations. In a recent AMA survey, more than 60% of physicians indicated that it’s difficult to know when prior authorization is needed. And 93% of physicians reported care delays while waiting for authorization, the AMA said.
Experts estimate that 80% of prior authorization work could be automated, but most practices still use the phone or fax, even as numbers of prior authorizations continue to increase.
How it works
Automation software connects directly to the practice’s electronic health record (EHR). “When the doctor places an order in the EHR, the process starts automatically,” Ms. Hadzhieva said. “The doctor may not even notice it.”
In addition to using an EHR connection, many software products can communicate with the payer through its portal or by fax or phone, while still automating other parts of the process.
The software’s first step is to decide whether prior authorization is needed. This requires having an updated list of the rules that each payer uses for prior authorization. Manually keeping track of payer rules is very time-consuming, but automation uses bots to visit each payer site to look for rules changes. One vendor, Infinitus, uses a voice-based bot called Eva that calls up each payer and speaks with a representative.
“Automatically updating payer rules is not a new technology,” said YiDing Yu, MD, chief product officer at Olive, the automation vendor for New England Baptist. “What is new in the last 5 years is extracting the information needed for the prior authorization out of the clinical notes.”
This is challenging because each doctor has different ways to describe each step of clinical work. To identify this shorthand, Dr. Yu said Olive uses natural language processing, which is a form of artificial intelligence that learns how each doctor describes things.
Dr. Yu asserts that Olive is actually better than a practice’s staff at digging out clinical information. She said staff without much clinical training may miss terms that the software can catch, and they don’t have the time to go back many months into the record to find valuable information. But automation can do that.
In some instances, however, the software may not be able to find the information, in which case it alerts staff through a prompt in the EHR and the information is retrieved manually, Dr. Yu said.
Next, the Olive software puts the information it found into the request form and sends it to the payer. After submission, the software constantly checks on the status of each request, again visiting payer sites with a bot.
At New England Baptist, the software is used mainly by physicians in fairly small private practices who are on staff. They are using the software on the hospital’s dime, but it only works inside the hospital, Ms. Hadzhieva said. For their work outside of the hospital, they would have to purchase the Olive software on their own, she said.
Automation hasn’t spread to practices yet
Despite the promising outcomes for products like Olive, automation software is still primarily used by large organizations. Vendors say very few private practices have bought it yet. “The technology works, but it is still in the early-adopter phase,” Dr. Yu said.
For one thing, the software can be expensive. Very few vendors reveal their prices, but Dr. Yu did so. She said Olive normally costs about $50,000 a year for even a small organization. She insisted, however, that the savings from avoiding just one denial each month for a hip surgery would justify the expense.
On the other hand, some automation software is free, such as the Surescripts product for prior authorization of prescriptions. But it is unclear whether Surescripts does as much as Olive. Vendors’ descriptions of their products tend to be vague.
Also, Surescripts and Olive have entirely separate functions. Dr. Yu said Olive is limited to procedures, so it benefits specialties like oncology, neurosurgery, colorectal surgery, vascular surgery, and cardiology. Olive does not cover prescriptions, because they operate on a different technology.
Dr. Yu said another hurdle for adopting the software is the kind of EHR systems that doctors use. At this point, only a few EHR systems – such as Epic, Cerner, and Athena – are compatible with Olive. Large organizations tend to use Epic and Cerner, while many practices often use Athena or a variety of other systems, she said.
Despite stunted demand, there is no shortage of companies offering automation software for medical (that is, non-prescription) prior authorization. One compilation lists 25 such vendors, including companies like Myndshft, Rhyme, Infinitus, Infinx, and Waystar. As with any start-up technology, companies occasionally buy each other out.
In addition to issues like cost, specialty, and EHR compatibility, another hurdle is that few doctors even know the technology exists. Vendors say marketing focuses on larger provider organizations, not smaller practices.
Even many tech-savvy doctors, like Adam Bruggeman, MD, an orthopedist and CEO of Texas Spine Care Center in San Antonio, say they know little about the technology. “There is definitely a need to automate prior authorization,” he said. “But I don’t know of any colleagues who use it.” He has only just begun to explore vendors, he said.
Many medical practice consultants also have not yet explored the technology. “Automation makes a lot of sense, because there are a lot of repetitive tasks in prior authorization,” said Jill Arena, CEO of Portland, Ore.–based Health e Practices. “But I haven’t looked into it yet, and none of my clients has even asked about it.”
“I could see how it could be an easier sell for large organizations,” she added. “They have an IT person and a CFO who can explore the issue. Smaller practices usually don’t have that kind of expertise.”
Where does automation go from here?
Until now, clinicians who want to fully automate prior authorizations would have to buy two products – one for medical procedures and one for prescriptions. This has to do with incompatible electronic transmission standards, which are used to digitize information, said Susan Lawson-Dawson, content marketing strategist for the vendor Myndshft Health.
Myndshft has long been selling automation software for medical prior authorizations, but now it is introducing a product for prescriptions, Ms. Lawson-Dawson said. She said Myndshft will then be the only vendor to automate both kinds of prior authorizations.
Ms. Lawson-Dawson said Myndshft has 685 customers to date and is looking for more business. Recently the company entered the Google Cloud Marketplace. Google Cloud customers can now direct their committed spend with Google to purchasing Myndshft, meaning they could get it at a discount.
Software like Olive and Myndshft can operate independently of payers, but a vendor called Rhyme depends on payers for its software to function, said Rhyme CEO Joe Anstine. He said more than 300 payers have agreed to install the Rhyme system, and Rhyme has signed up a number of large health systems to use the product. Initially, he said, clinicians paid for the service, but now Rhyme is beginning to find payers to foot the costs and to let clinicians use it for free, which would open Rhyme up to smaller practices.
EHR companies themselves are beginning to offer automation, too. Epic, for example, has created a tool for prior authorization as part of its Epic Payer Platform. Like Rhyme, it requires payer cooperation, because information goes back and forth between clinician and payer in what is called bi-directional exchange.
The Epic product is still in its pilot phase. Epic reported that several large health systems were using its product in conjunction with a specific payer – for instance, Mayo Clinic with Blue Cross and Blue Shield of Minnesota and Ochsner Health with Humana. According to Epic, the arrangement reduced Mayo’s denials due to additional documentation requests by 63% for professional billing.
Automating with just one payer still means the clinician has to deal with manual processes at other payers, but a large clinician could have sufficient volume with that one payer to make the arrangement useful.
Will payers automate prior authorization?
Ultimately, payers may take the automation business away from vendors, offering a free product to all clinicians. But don’t hold your breath. Payers first have to rebuild their electronic systems to accommodate an electronic connection with providers. Even then, some payers might hold back from automating, forcing practices to continue manually processing some prior authorizations.
Efforts are underway, however, to mandate payers to support prior authorization automation. For this to happen, payers would have to revamp their data so that it could be easily read by practices’ EHRs. This would mean adopting a specific interoperability standard called Health Level 7 Fast Healthcare Interoperability Resources (FHIR).
Toward this goal, the Centers for Medicare & Medicaid Services proposes to require payers to adopt FHIR by January 2026. (CMS still has to finalize the rule.) Experts say the two-year ramp-up time is needed because it takes extensive work for payers to translate their data into FHIR.
The only payer so far to switch to FHIR for prior authorization is Regence in Washington state. In a pilot project, it has automated prior authorization with just one provider, MultiCare Connected Care, an accountable care organization (ACO), also in Washington state.
Anna Taylor, associate vice president of population health and value-based care at MultiCare, explained how the arrangement works. “Two separate entities are sharing one operational process,” she told this news organization. “That means they can have a digital conversation back and forth, so it is much easier to resolve prior authorization issues.”
Unlike many vendor products, the Regence service is free. And while the vendors market only to large organizations, most doctors in the MultiCare arrangement are in independent practices. Ms. Taylor said these doctors have been “enthusiastic” about the arrangement.
The results of the pilot are impressive. Ms. Taylor said automation has resulted in a 233% productivity gain for MultiCare clinicians, and 89% of submissions to Regence get an immediate response.
There is a potential downside, however, to working directly with payers. A direct connection to clinicians allows payers to access the doctor’s clinical notes, which could make many doctors uneasy. But Ms. Taylor said Regence only has access to the “discrete data fields” on MultiCare’s EHR dashboard, not to the notes themselves.
The ultimate goal of the Regence-Multicare project is to include more payers and clinicians. Ms. Taylor said two of the 27 other payers that MultiCare works with are “highly interested,” but it would take a lot of work for them to get connected with practices and other clinicians.
Ultimately, payers could offer automation and third-party vendors might then fade away. However, physicians may resist working directly with payers if the arrangement requires full access to their medical records.
A version of this article first appeared on Medscape.com.
Maternal perinatal mortality: A pediatric issue
Checking on the well-being of mothers is one of the important acknowledged aspects of primary pediatric care. “How are you doing?” directed to the child’s mother has long been considered an appropriate question. The AAP recommends several checks in the Bright Futures Guidelines, including conducting several formal screens for depression and asking about “getting time alone with your partner” as well as other supports.
But I have recently become aware of new data that changes my ideas about what we pediatricians need to be doing as part of our care for children and their families, especially in the first year: Considering the risks to the mother of dying.
Maternal mortality increased by 26.6% from 2000 to 2014 across the United States such that it is higher now than it was for our own mothers. The U.S. now has the highest rates of maternal mortality among high-income nations, especially for Black, American Indian, or Alaska Native women, those of lower socioeconomic status, and those under 18 or over 35 years old.
You may be thinking, well, that is an issue for ob.gyns. Indeed, the most common reasons for maternal death are cardiovascular: hemorrhage, hypertensive disorders, deep vein thrombosis, and stroke, all usually occurring at or in the first week after birth. You may have heard about sudden unexpected heart failure from postpartum cardiomyopathy, although rare (1 in 1,000-4,000), presenting from 1 month pre birth to 5 months post delivery, which is when we may be the main clinicians seeing the mother, not the ob.gyns. This can be easily missed since it presents with shortness of breath and decreased exercise tolerance, fatigue, palpitations, and/or leg swelling. Serious eclampsia may have only symptoms of headache or abdominal pain. All of these may easily be mistaken for lingering pregnancy symptoms. But in higher income countries, such as the U.S., 38% of maternal deaths occur from 8 to 42 days after birth, the period for fatal infections as well as cardiac complications. Elevated risk for all of these causes of mortality include Black race, obesity, tobacco use, congenital heart disease, and being older than 40.
As pediatric providers, we may see mothers along with their infants as newborns in the hospital, at day 2, at 2 weeks, or even at 1-2 months after birth, potentially before their one recommended postnatal obstetric visit at 3-8 weeks. Asking the mother how she is feeling at those times should not just be a social nicety but rather an additional check for serious postnatal complications.
Additional concerns
But wait, it gets worse.
Did you know that the leading cause of maternal death from pregnancy up to 1 year after a birth is homicide?
Maternal perinatal mortality figures have not usually included “perinatal-associated” deaths, a maternal death attributable to a condition that is unaffected by the pregnancy and occurring within 1 year of delivery (that I will cite as perinatal henceforth). While half of maternal deaths occur during pregnancy, another half occur in the year following. There were 3.62 homicides per 100,000 live births among females who were pregnant or within 1 year postpartum, 16% more than for similarly aged nonpregnant and nonpostpartum women (3.12 deaths/100,000 population, P < .05). Homicides made up 8.4% of reported perinatal maternal deaths from all causes, with a rate of 1.7 per 100,000 live births, twice the rate of any one of the other leading causes noted above. Black women had seven times the risk of perinatal homicide as that of White women. Females under 20, many of them our own pediatric patients, had a greater than six times higher risk and those aged 20-24 had a 65% higher risk of pregnancy-associated homicide across race and ethnic groups. Homicide is most likely before 21 weeks of pregnancy, decreases in the third trimester, but increases again after birth. Two-thirds of pregnancy-associated homicide deaths occurred in the home, with the perpetrator a current or prior partner (> 59%, with 98% being male), 45%-50% were associated with reported intimate partner violence (IPV), and the most common method was a firearm (55%). Often the same women had histories of substance abuse, serious mental illness, and/or prior IPV, all risk factors for pregnancy-associated deaths, including from homicide.
Homicide? “Not the mothers in my practice,” you may say, but, if not homicide, drug-related deaths (3.68 per 100,000 person-years) and suicide (1.42 per 100,000 person-years) together comprise 18% of all maternal deaths. Non-Hispanic White women, Medicaid-insured women, and women residing in smaller cities were especially likely to die from drugs or suicide. More than half (54.3%) of perinatal suicides involve intimate partner conflict, which increases the risk ninefold. Perinatal mood disorders, affecting up to 15% of pregnant and postpartum U.S. women, is also a risk factor in substance abuse, opioid overdose death, and suicide.
And substance use has gotten more dangerous with the increase in fentanyl lacing. Pregnancy-associated deaths (4%-10% of deaths) involving opioids more than doubled between 2007 and 2016, and, although the rates are higher for Black women, the increase has been greater for non-Hispanic White women. Two-thirds of those deaths occur between 6 and 12 months postpartum, on our watch. Although many women decrease substance use during pregnancy, they may fall back into substance use (rates increase 4 times by 7-12 months after delivery) and not continue to receive treatment. Although pharmacotherapy (e.g., methadone, buprenorphine treatment) is the current standard of care for opioid use disorder (OUD) during pregnancy, nearly half receiving treatment in publicly funded centers are not receiving these medications and others may lose insurance or access to pregnancy-related treatment programs after delivery, increasing risk of relapse. Stigma, and punitive or discriminatory approaches to pregnant women with OUD (e.g., jail, removal of children) can dissuade them from participating in treatment, increasing overdose risk.
It is important to note that in more than half of the 41 deaths from violent trauma in one study (including 22 homicides), obstetrical providers knew of or suspected IPV. Also, the vast majority (74%) of those who died by drugs or suicide had made one or more emergency department or hospital visit between their delivery and death, and 39% had made three or more visits. Without knowing if anything was done in those cases, we also know that, in addition to thorough, compassionate providers, there is sometimes segmentation of responsibility, insensitivity, discrimination, racism, stigma, inequity, lack of resources, lack of access, lack of payment mechanisms, legal issues for immigrants, time constraints, and other systemic deficits that may hinder effective care for these and subsequent women.
Awareness and action
What should we, who are primary care pediatric providers, do about these threats to the mothers and pregnant young women we care for? Clearly, their children, our main patients, would be terribly and permanently hurt by harm coming to their mothers – the extreme adverse childhood experiences and social determinants of health to which we are already committed.
I hope this article will help alert pediatric providers to what is being published, mainly as women’s health and public health issues.
First, we need awareness of the physical symptoms that may come up in our interactions with pregnant and postpartum women so that we can educate them and expedite any indicated emergency care.
Next, we need to expand our routine screening of mothers and pregnant women from just the most impactful social determinants of health (including depression, substance use, and IPV) to include anxiety, past suicide attempts and current suicidal ideation, and the presence of firearms, early and repeatedly in the first year of the child’s life. Adults and teens are more likely to disclose risk for sensitive issues through questionnaires than through interviews, perhaps even more so when the identified patient is their child rather than themselves. Any screen can have false negatives, so asking directly when risk is suspected is important. The reason for screening could be framed as caring for the caregiver who is the most important person for the child. It could be accompanied by acknowledging that pregnancy and the first year of life can be difficult for mothers and their partners and that we want to support them and connect them to resources, if needed. When substance use disorder is acknowledged, we should prescribe and teach about Narcan for overdose. When there is IPV, we should discuss firearm removal/locking as well as counseling on a personal safety plan.
Working as part of an on-site or virtual team that includes professionals who know about community resources and can coordinate care is essential, in addition to educating about 211 for services and 988 for suicide risk.
Finally, we can advocate and vote for programs, people, and laws that support and safeguard women and families, address substance use, and reduce access to firearms.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS (www.CHADIS.com). She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at pdnews@mdedge.com.
Checking on the well-being of mothers is one of the important acknowledged aspects of primary pediatric care. “How are you doing?” directed to the child’s mother has long been considered an appropriate question. The AAP recommends several checks in the Bright Futures Guidelines, including conducting several formal screens for depression and asking about “getting time alone with your partner” as well as other supports.
But I have recently become aware of new data that changes my ideas about what we pediatricians need to be doing as part of our care for children and their families, especially in the first year: Considering the risks to the mother of dying.
Maternal mortality increased by 26.6% from 2000 to 2014 across the United States such that it is higher now than it was for our own mothers. The U.S. now has the highest rates of maternal mortality among high-income nations, especially for Black, American Indian, or Alaska Native women, those of lower socioeconomic status, and those under 18 or over 35 years old.
You may be thinking, well, that is an issue for ob.gyns. Indeed, the most common reasons for maternal death are cardiovascular: hemorrhage, hypertensive disorders, deep vein thrombosis, and stroke, all usually occurring at or in the first week after birth. You may have heard about sudden unexpected heart failure from postpartum cardiomyopathy, although rare (1 in 1,000-4,000), presenting from 1 month pre birth to 5 months post delivery, which is when we may be the main clinicians seeing the mother, not the ob.gyns. This can be easily missed since it presents with shortness of breath and decreased exercise tolerance, fatigue, palpitations, and/or leg swelling. Serious eclampsia may have only symptoms of headache or abdominal pain. All of these may easily be mistaken for lingering pregnancy symptoms. But in higher income countries, such as the U.S., 38% of maternal deaths occur from 8 to 42 days after birth, the period for fatal infections as well as cardiac complications. Elevated risk for all of these causes of mortality include Black race, obesity, tobacco use, congenital heart disease, and being older than 40.
As pediatric providers, we may see mothers along with their infants as newborns in the hospital, at day 2, at 2 weeks, or even at 1-2 months after birth, potentially before their one recommended postnatal obstetric visit at 3-8 weeks. Asking the mother how she is feeling at those times should not just be a social nicety but rather an additional check for serious postnatal complications.
Additional concerns
But wait, it gets worse.
Did you know that the leading cause of maternal death from pregnancy up to 1 year after a birth is homicide?
Maternal perinatal mortality figures have not usually included “perinatal-associated” deaths, a maternal death attributable to a condition that is unaffected by the pregnancy and occurring within 1 year of delivery (that I will cite as perinatal henceforth). While half of maternal deaths occur during pregnancy, another half occur in the year following. There were 3.62 homicides per 100,000 live births among females who were pregnant or within 1 year postpartum, 16% more than for similarly aged nonpregnant and nonpostpartum women (3.12 deaths/100,000 population, P < .05). Homicides made up 8.4% of reported perinatal maternal deaths from all causes, with a rate of 1.7 per 100,000 live births, twice the rate of any one of the other leading causes noted above. Black women had seven times the risk of perinatal homicide as that of White women. Females under 20, many of them our own pediatric patients, had a greater than six times higher risk and those aged 20-24 had a 65% higher risk of pregnancy-associated homicide across race and ethnic groups. Homicide is most likely before 21 weeks of pregnancy, decreases in the third trimester, but increases again after birth. Two-thirds of pregnancy-associated homicide deaths occurred in the home, with the perpetrator a current or prior partner (> 59%, with 98% being male), 45%-50% were associated with reported intimate partner violence (IPV), and the most common method was a firearm (55%). Often the same women had histories of substance abuse, serious mental illness, and/or prior IPV, all risk factors for pregnancy-associated deaths, including from homicide.
Homicide? “Not the mothers in my practice,” you may say, but, if not homicide, drug-related deaths (3.68 per 100,000 person-years) and suicide (1.42 per 100,000 person-years) together comprise 18% of all maternal deaths. Non-Hispanic White women, Medicaid-insured women, and women residing in smaller cities were especially likely to die from drugs or suicide. More than half (54.3%) of perinatal suicides involve intimate partner conflict, which increases the risk ninefold. Perinatal mood disorders, affecting up to 15% of pregnant and postpartum U.S. women, is also a risk factor in substance abuse, opioid overdose death, and suicide.
And substance use has gotten more dangerous with the increase in fentanyl lacing. Pregnancy-associated deaths (4%-10% of deaths) involving opioids more than doubled between 2007 and 2016, and, although the rates are higher for Black women, the increase has been greater for non-Hispanic White women. Two-thirds of those deaths occur between 6 and 12 months postpartum, on our watch. Although many women decrease substance use during pregnancy, they may fall back into substance use (rates increase 4 times by 7-12 months after delivery) and not continue to receive treatment. Although pharmacotherapy (e.g., methadone, buprenorphine treatment) is the current standard of care for opioid use disorder (OUD) during pregnancy, nearly half receiving treatment in publicly funded centers are not receiving these medications and others may lose insurance or access to pregnancy-related treatment programs after delivery, increasing risk of relapse. Stigma, and punitive or discriminatory approaches to pregnant women with OUD (e.g., jail, removal of children) can dissuade them from participating in treatment, increasing overdose risk.
It is important to note that in more than half of the 41 deaths from violent trauma in one study (including 22 homicides), obstetrical providers knew of or suspected IPV. Also, the vast majority (74%) of those who died by drugs or suicide had made one or more emergency department or hospital visit between their delivery and death, and 39% had made three or more visits. Without knowing if anything was done in those cases, we also know that, in addition to thorough, compassionate providers, there is sometimes segmentation of responsibility, insensitivity, discrimination, racism, stigma, inequity, lack of resources, lack of access, lack of payment mechanisms, legal issues for immigrants, time constraints, and other systemic deficits that may hinder effective care for these and subsequent women.
Awareness and action
What should we, who are primary care pediatric providers, do about these threats to the mothers and pregnant young women we care for? Clearly, their children, our main patients, would be terribly and permanently hurt by harm coming to their mothers – the extreme adverse childhood experiences and social determinants of health to which we are already committed.
I hope this article will help alert pediatric providers to what is being published, mainly as women’s health and public health issues.
First, we need awareness of the physical symptoms that may come up in our interactions with pregnant and postpartum women so that we can educate them and expedite any indicated emergency care.
Next, we need to expand our routine screening of mothers and pregnant women from just the most impactful social determinants of health (including depression, substance use, and IPV) to include anxiety, past suicide attempts and current suicidal ideation, and the presence of firearms, early and repeatedly in the first year of the child’s life. Adults and teens are more likely to disclose risk for sensitive issues through questionnaires than through interviews, perhaps even more so when the identified patient is their child rather than themselves. Any screen can have false negatives, so asking directly when risk is suspected is important. The reason for screening could be framed as caring for the caregiver who is the most important person for the child. It could be accompanied by acknowledging that pregnancy and the first year of life can be difficult for mothers and their partners and that we want to support them and connect them to resources, if needed. When substance use disorder is acknowledged, we should prescribe and teach about Narcan for overdose. When there is IPV, we should discuss firearm removal/locking as well as counseling on a personal safety plan.
Working as part of an on-site or virtual team that includes professionals who know about community resources and can coordinate care is essential, in addition to educating about 211 for services and 988 for suicide risk.
Finally, we can advocate and vote for programs, people, and laws that support and safeguard women and families, address substance use, and reduce access to firearms.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS (www.CHADIS.com). She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at pdnews@mdedge.com.
Checking on the well-being of mothers is one of the important acknowledged aspects of primary pediatric care. “How are you doing?” directed to the child’s mother has long been considered an appropriate question. The AAP recommends several checks in the Bright Futures Guidelines, including conducting several formal screens for depression and asking about “getting time alone with your partner” as well as other supports.
But I have recently become aware of new data that changes my ideas about what we pediatricians need to be doing as part of our care for children and their families, especially in the first year: Considering the risks to the mother of dying.
Maternal mortality increased by 26.6% from 2000 to 2014 across the United States such that it is higher now than it was for our own mothers. The U.S. now has the highest rates of maternal mortality among high-income nations, especially for Black, American Indian, or Alaska Native women, those of lower socioeconomic status, and those under 18 or over 35 years old.
You may be thinking, well, that is an issue for ob.gyns. Indeed, the most common reasons for maternal death are cardiovascular: hemorrhage, hypertensive disorders, deep vein thrombosis, and stroke, all usually occurring at or in the first week after birth. You may have heard about sudden unexpected heart failure from postpartum cardiomyopathy, although rare (1 in 1,000-4,000), presenting from 1 month pre birth to 5 months post delivery, which is when we may be the main clinicians seeing the mother, not the ob.gyns. This can be easily missed since it presents with shortness of breath and decreased exercise tolerance, fatigue, palpitations, and/or leg swelling. Serious eclampsia may have only symptoms of headache or abdominal pain. All of these may easily be mistaken for lingering pregnancy symptoms. But in higher income countries, such as the U.S., 38% of maternal deaths occur from 8 to 42 days after birth, the period for fatal infections as well as cardiac complications. Elevated risk for all of these causes of mortality include Black race, obesity, tobacco use, congenital heart disease, and being older than 40.
As pediatric providers, we may see mothers along with their infants as newborns in the hospital, at day 2, at 2 weeks, or even at 1-2 months after birth, potentially before their one recommended postnatal obstetric visit at 3-8 weeks. Asking the mother how she is feeling at those times should not just be a social nicety but rather an additional check for serious postnatal complications.
Additional concerns
But wait, it gets worse.
Did you know that the leading cause of maternal death from pregnancy up to 1 year after a birth is homicide?
Maternal perinatal mortality figures have not usually included “perinatal-associated” deaths, a maternal death attributable to a condition that is unaffected by the pregnancy and occurring within 1 year of delivery (that I will cite as perinatal henceforth). While half of maternal deaths occur during pregnancy, another half occur in the year following. There were 3.62 homicides per 100,000 live births among females who were pregnant or within 1 year postpartum, 16% more than for similarly aged nonpregnant and nonpostpartum women (3.12 deaths/100,000 population, P < .05). Homicides made up 8.4% of reported perinatal maternal deaths from all causes, with a rate of 1.7 per 100,000 live births, twice the rate of any one of the other leading causes noted above. Black women had seven times the risk of perinatal homicide as that of White women. Females under 20, many of them our own pediatric patients, had a greater than six times higher risk and those aged 20-24 had a 65% higher risk of pregnancy-associated homicide across race and ethnic groups. Homicide is most likely before 21 weeks of pregnancy, decreases in the third trimester, but increases again after birth. Two-thirds of pregnancy-associated homicide deaths occurred in the home, with the perpetrator a current or prior partner (> 59%, with 98% being male), 45%-50% were associated with reported intimate partner violence (IPV), and the most common method was a firearm (55%). Often the same women had histories of substance abuse, serious mental illness, and/or prior IPV, all risk factors for pregnancy-associated deaths, including from homicide.
Homicide? “Not the mothers in my practice,” you may say, but, if not homicide, drug-related deaths (3.68 per 100,000 person-years) and suicide (1.42 per 100,000 person-years) together comprise 18% of all maternal deaths. Non-Hispanic White women, Medicaid-insured women, and women residing in smaller cities were especially likely to die from drugs or suicide. More than half (54.3%) of perinatal suicides involve intimate partner conflict, which increases the risk ninefold. Perinatal mood disorders, affecting up to 15% of pregnant and postpartum U.S. women, is also a risk factor in substance abuse, opioid overdose death, and suicide.
And substance use has gotten more dangerous with the increase in fentanyl lacing. Pregnancy-associated deaths (4%-10% of deaths) involving opioids more than doubled between 2007 and 2016, and, although the rates are higher for Black women, the increase has been greater for non-Hispanic White women. Two-thirds of those deaths occur between 6 and 12 months postpartum, on our watch. Although many women decrease substance use during pregnancy, they may fall back into substance use (rates increase 4 times by 7-12 months after delivery) and not continue to receive treatment. Although pharmacotherapy (e.g., methadone, buprenorphine treatment) is the current standard of care for opioid use disorder (OUD) during pregnancy, nearly half receiving treatment in publicly funded centers are not receiving these medications and others may lose insurance or access to pregnancy-related treatment programs after delivery, increasing risk of relapse. Stigma, and punitive or discriminatory approaches to pregnant women with OUD (e.g., jail, removal of children) can dissuade them from participating in treatment, increasing overdose risk.
It is important to note that in more than half of the 41 deaths from violent trauma in one study (including 22 homicides), obstetrical providers knew of or suspected IPV. Also, the vast majority (74%) of those who died by drugs or suicide had made one or more emergency department or hospital visit between their delivery and death, and 39% had made three or more visits. Without knowing if anything was done in those cases, we also know that, in addition to thorough, compassionate providers, there is sometimes segmentation of responsibility, insensitivity, discrimination, racism, stigma, inequity, lack of resources, lack of access, lack of payment mechanisms, legal issues for immigrants, time constraints, and other systemic deficits that may hinder effective care for these and subsequent women.
Awareness and action
What should we, who are primary care pediatric providers, do about these threats to the mothers and pregnant young women we care for? Clearly, their children, our main patients, would be terribly and permanently hurt by harm coming to their mothers – the extreme adverse childhood experiences and social determinants of health to which we are already committed.
I hope this article will help alert pediatric providers to what is being published, mainly as women’s health and public health issues.
First, we need awareness of the physical symptoms that may come up in our interactions with pregnant and postpartum women so that we can educate them and expedite any indicated emergency care.
Next, we need to expand our routine screening of mothers and pregnant women from just the most impactful social determinants of health (including depression, substance use, and IPV) to include anxiety, past suicide attempts and current suicidal ideation, and the presence of firearms, early and repeatedly in the first year of the child’s life. Adults and teens are more likely to disclose risk for sensitive issues through questionnaires than through interviews, perhaps even more so when the identified patient is their child rather than themselves. Any screen can have false negatives, so asking directly when risk is suspected is important. The reason for screening could be framed as caring for the caregiver who is the most important person for the child. It could be accompanied by acknowledging that pregnancy and the first year of life can be difficult for mothers and their partners and that we want to support them and connect them to resources, if needed. When substance use disorder is acknowledged, we should prescribe and teach about Narcan for overdose. When there is IPV, we should discuss firearm removal/locking as well as counseling on a personal safety plan.
Working as part of an on-site or virtual team that includes professionals who know about community resources and can coordinate care is essential, in addition to educating about 211 for services and 988 for suicide risk.
Finally, we can advocate and vote for programs, people, and laws that support and safeguard women and families, address substance use, and reduce access to firearms.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS (www.CHADIS.com). She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at pdnews@mdedge.com.
AI in medicine has a major Cassandra problem
This transcript has been edited for clarity.
Today I’m going to talk to you about a study at the cutting edge of modern medicine, one that uses an artificial intelligence (AI) model to guide care. But before I do, I need to take you back to the late Bronze Age, to a city located on the coast of what is now Turkey.
Troy’s towering walls made it seem unassailable, but that would not stop the Achaeans and their fleet of black ships from making landfall, and, after a siege, destroying the city. The destruction of Troy, as told in the Iliad and the Aeneid, was foretold by Cassandra, the daughter of King Priam and Priestess of Troy.
Cassandra had been given the gift of prophecy by the god Apollo in exchange for her favors. But after the gift was bestowed, she rejected the bright god and, in his rage, he added a curse to her blessing: that no one would ever believe her prophecies.
Thus it was that when her brother Paris set off to Sparta to abduct Helen, she warned him that his actions would lead to the downfall of their great city. He, of course, ignored her.
And you know the rest of the story.
Why am I telling you the story of Cassandra of Troy when we’re supposed to be talking about AI in medicine? Because AI has a major Cassandra problem.
The recent history of AI, and particularly the subset of AI known as machine learning in medicine, has been characterized by an accuracy arms race.
The electronic health record allows for the collection of volumes of data orders of magnitude greater than what we have ever been able to collect before. And all that data can be crunched by various algorithms to make predictions about, well, anything – whether a patient will be transferred to the intensive care unit, whether a GI bleed will need an intervention, whether someone will die in the next year.
Studies in this area tend to rely on retrospective datasets, and as time has gone on, better algorithms and more data have led to better and better predictions. In some simpler cases, machine-learning models have achieved near-perfect accuracy – Cassandra-level accuracy – as in the reading of chest x-rays for pneumonia, for example.
But as Cassandra teaches us, even perfect prediction is useless if no one believes you, if they don’t change their behavior. And this is the central problem of AI in medicine today. Many people are focusing on accuracy of the prediction but have forgotten that high accuracy is just table stakes for an AI model to be useful. It has to not only be accurate, but its use also has to change outcomes for patients. We need to be able to save Troy.
The best way to determine whether an AI model will help patients is to treat a model like we treat a new medication and evaluate it through a randomized trial. That’s what researchers, led by Shannon Walker of Vanderbilt University, Nashville, Tenn., did in a paper appearing in JAMA Network Open.
The model in question was one that predicted venous thromboembolism – blood clots – in hospitalized children. The model took in a variety of data points from the health record: a history of blood clot, history of cancer, presence of a central line, a variety of lab values. And the predictive model was very good – maybe not Cassandra good, but it achieved an AUC of 0.90, which means it had very high accuracy.
But again, accuracy is just table stakes.
The authors deployed the model in the live health record and recorded the results. For half of the kids, that was all that happened; no one actually saw the predictions. For those randomized to the intervention, the hematology team would be notified when the risk for clot was calculated to be greater than 2.5%. The hematology team would then contact the primary team to discuss prophylactic anticoagulation.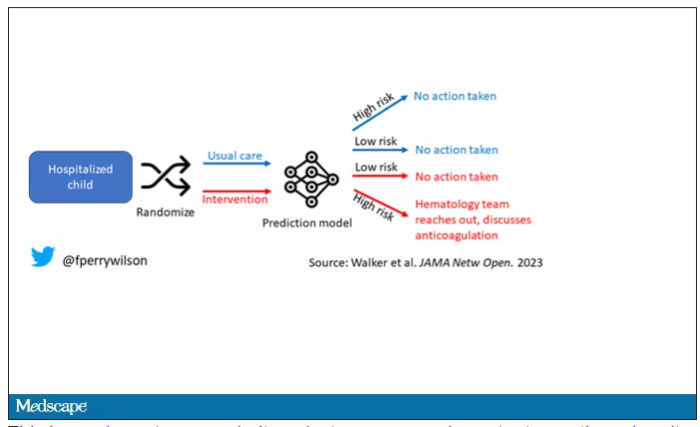
This is an elegant approach.
Let’s start with those table stakes – accuracy. The predictions were, by and large, pretty accurate in this trial. Of the 135 kids who developed blood clots, 121 had been flagged by the model in advance. That’s about 90%. The model flagged about 10% of kids who didn’t get a blood clot as well, but that’s not entirely surprising since the threshold for flagging was a 2.5% risk.
Given that the model preidentified almost every kid who would go on to develop a blood clot, it would make sense that kids randomized to the intervention would do better; after all, Cassandra was calling out her warnings.
But those kids didn’t do better. The rate of blood clot was no different between the group that used the accurate prediction model and the group that did not.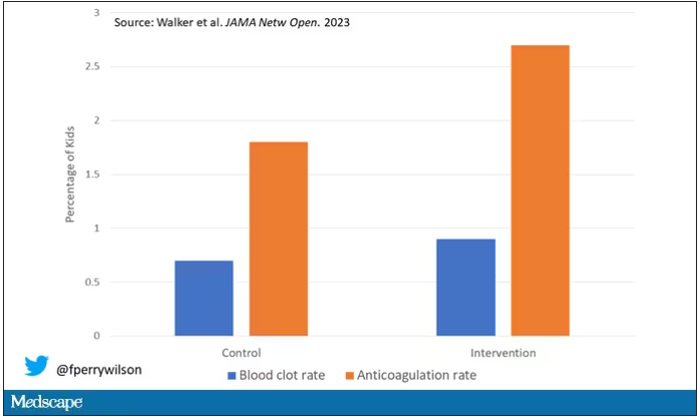
Why? Why does the use of an accurate model not necessarily improve outcomes?
First of all, a warning must lead to some change in management. Indeed, the kids in the intervention group were more likely to receive anticoagulation, but barely so. There were lots of reasons for this: physician preference, imminent discharge, active bleeding, and so on.
But let’s take a look at the 77 kids in the intervention arm who developed blood clots, because I think this is an instructive analysis.
Six of them did not meet the 2.5% threshold criteria, a case where the model missed its mark. Again, accuracy is table stakes.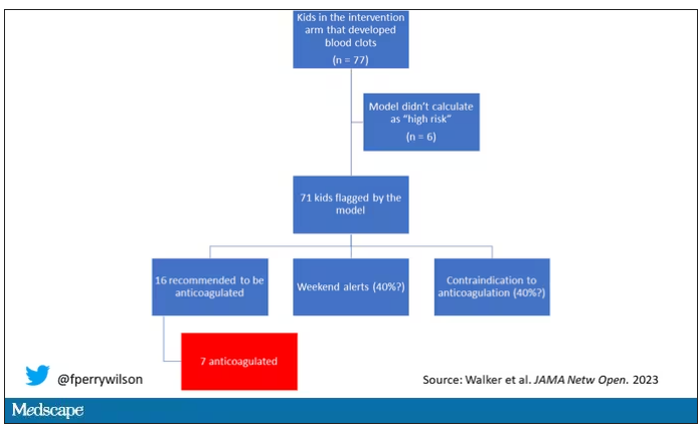
Of the remaining 71, only 16 got a recommendation from the hematologist to start anticoagulation. Why not more? Well, the model identified some of the high-risk kids on the weekend, and it seems that the study team did not contact treatment teams during that time. That may account for about 40% of these cases. The remainder had some contraindication to anticoagulation.
Most tellingly, of the 16 who did get a recommendation to start anticoagulation, the recommendation was followed in only seven patients.
This is the gap between accurate prediction and the ability to change outcomes for patients. A prediction is useless if it is wrong, for sure. But it’s also useless if you don’t tell anyone about it. It’s useless if you tell someone but they can’t do anything about it. And it’s useless if they could do something about it but choose not to.
That’s the gulf that these models need to cross at this point. So, the next time some slick company tells you how accurate their AI model is, ask them if accuracy is really the most important thing. If they say, “Well, yes, of course,” then tell them about Cassandra.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Today I’m going to talk to you about a study at the cutting edge of modern medicine, one that uses an artificial intelligence (AI) model to guide care. But before I do, I need to take you back to the late Bronze Age, to a city located on the coast of what is now Turkey.
Troy’s towering walls made it seem unassailable, but that would not stop the Achaeans and their fleet of black ships from making landfall, and, after a siege, destroying the city. The destruction of Troy, as told in the Iliad and the Aeneid, was foretold by Cassandra, the daughter of King Priam and Priestess of Troy.
Cassandra had been given the gift of prophecy by the god Apollo in exchange for her favors. But after the gift was bestowed, she rejected the bright god and, in his rage, he added a curse to her blessing: that no one would ever believe her prophecies.
Thus it was that when her brother Paris set off to Sparta to abduct Helen, she warned him that his actions would lead to the downfall of their great city. He, of course, ignored her.
And you know the rest of the story.
Why am I telling you the story of Cassandra of Troy when we’re supposed to be talking about AI in medicine? Because AI has a major Cassandra problem.
The recent history of AI, and particularly the subset of AI known as machine learning in medicine, has been characterized by an accuracy arms race.
The electronic health record allows for the collection of volumes of data orders of magnitude greater than what we have ever been able to collect before. And all that data can be crunched by various algorithms to make predictions about, well, anything – whether a patient will be transferred to the intensive care unit, whether a GI bleed will need an intervention, whether someone will die in the next year.
Studies in this area tend to rely on retrospective datasets, and as time has gone on, better algorithms and more data have led to better and better predictions. In some simpler cases, machine-learning models have achieved near-perfect accuracy – Cassandra-level accuracy – as in the reading of chest x-rays for pneumonia, for example.
But as Cassandra teaches us, even perfect prediction is useless if no one believes you, if they don’t change their behavior. And this is the central problem of AI in medicine today. Many people are focusing on accuracy of the prediction but have forgotten that high accuracy is just table stakes for an AI model to be useful. It has to not only be accurate, but its use also has to change outcomes for patients. We need to be able to save Troy.
The best way to determine whether an AI model will help patients is to treat a model like we treat a new medication and evaluate it through a randomized trial. That’s what researchers, led by Shannon Walker of Vanderbilt University, Nashville, Tenn., did in a paper appearing in JAMA Network Open.
The model in question was one that predicted venous thromboembolism – blood clots – in hospitalized children. The model took in a variety of data points from the health record: a history of blood clot, history of cancer, presence of a central line, a variety of lab values. And the predictive model was very good – maybe not Cassandra good, but it achieved an AUC of 0.90, which means it had very high accuracy.
But again, accuracy is just table stakes.
The authors deployed the model in the live health record and recorded the results. For half of the kids, that was all that happened; no one actually saw the predictions. For those randomized to the intervention, the hematology team would be notified when the risk for clot was calculated to be greater than 2.5%. The hematology team would then contact the primary team to discuss prophylactic anticoagulation.
This is an elegant approach.
Let’s start with those table stakes – accuracy. The predictions were, by and large, pretty accurate in this trial. Of the 135 kids who developed blood clots, 121 had been flagged by the model in advance. That’s about 90%. The model flagged about 10% of kids who didn’t get a blood clot as well, but that’s not entirely surprising since the threshold for flagging was a 2.5% risk.
Given that the model preidentified almost every kid who would go on to develop a blood clot, it would make sense that kids randomized to the intervention would do better; after all, Cassandra was calling out her warnings.
But those kids didn’t do better. The rate of blood clot was no different between the group that used the accurate prediction model and the group that did not.
Why? Why does the use of an accurate model not necessarily improve outcomes?
First of all, a warning must lead to some change in management. Indeed, the kids in the intervention group were more likely to receive anticoagulation, but barely so. There were lots of reasons for this: physician preference, imminent discharge, active bleeding, and so on.
But let’s take a look at the 77 kids in the intervention arm who developed blood clots, because I think this is an instructive analysis.
Six of them did not meet the 2.5% threshold criteria, a case where the model missed its mark. Again, accuracy is table stakes.
Of the remaining 71, only 16 got a recommendation from the hematologist to start anticoagulation. Why not more? Well, the model identified some of the high-risk kids on the weekend, and it seems that the study team did not contact treatment teams during that time. That may account for about 40% of these cases. The remainder had some contraindication to anticoagulation.
Most tellingly, of the 16 who did get a recommendation to start anticoagulation, the recommendation was followed in only seven patients.
This is the gap between accurate prediction and the ability to change outcomes for patients. A prediction is useless if it is wrong, for sure. But it’s also useless if you don’t tell anyone about it. It’s useless if you tell someone but they can’t do anything about it. And it’s useless if they could do something about it but choose not to.
That’s the gulf that these models need to cross at this point. So, the next time some slick company tells you how accurate their AI model is, ask them if accuracy is really the most important thing. If they say, “Well, yes, of course,” then tell them about Cassandra.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Today I’m going to talk to you about a study at the cutting edge of modern medicine, one that uses an artificial intelligence (AI) model to guide care. But before I do, I need to take you back to the late Bronze Age, to a city located on the coast of what is now Turkey.
Troy’s towering walls made it seem unassailable, but that would not stop the Achaeans and their fleet of black ships from making landfall, and, after a siege, destroying the city. The destruction of Troy, as told in the Iliad and the Aeneid, was foretold by Cassandra, the daughter of King Priam and Priestess of Troy.
Cassandra had been given the gift of prophecy by the god Apollo in exchange for her favors. But after the gift was bestowed, she rejected the bright god and, in his rage, he added a curse to her blessing: that no one would ever believe her prophecies.
Thus it was that when her brother Paris set off to Sparta to abduct Helen, she warned him that his actions would lead to the downfall of their great city. He, of course, ignored her.
And you know the rest of the story.
Why am I telling you the story of Cassandra of Troy when we’re supposed to be talking about AI in medicine? Because AI has a major Cassandra problem.
The recent history of AI, and particularly the subset of AI known as machine learning in medicine, has been characterized by an accuracy arms race.
The electronic health record allows for the collection of volumes of data orders of magnitude greater than what we have ever been able to collect before. And all that data can be crunched by various algorithms to make predictions about, well, anything – whether a patient will be transferred to the intensive care unit, whether a GI bleed will need an intervention, whether someone will die in the next year.
Studies in this area tend to rely on retrospective datasets, and as time has gone on, better algorithms and more data have led to better and better predictions. In some simpler cases, machine-learning models have achieved near-perfect accuracy – Cassandra-level accuracy – as in the reading of chest x-rays for pneumonia, for example.
But as Cassandra teaches us, even perfect prediction is useless if no one believes you, if they don’t change their behavior. And this is the central problem of AI in medicine today. Many people are focusing on accuracy of the prediction but have forgotten that high accuracy is just table stakes for an AI model to be useful. It has to not only be accurate, but its use also has to change outcomes for patients. We need to be able to save Troy.
The best way to determine whether an AI model will help patients is to treat a model like we treat a new medication and evaluate it through a randomized trial. That’s what researchers, led by Shannon Walker of Vanderbilt University, Nashville, Tenn., did in a paper appearing in JAMA Network Open.
The model in question was one that predicted venous thromboembolism – blood clots – in hospitalized children. The model took in a variety of data points from the health record: a history of blood clot, history of cancer, presence of a central line, a variety of lab values. And the predictive model was very good – maybe not Cassandra good, but it achieved an AUC of 0.90, which means it had very high accuracy.
But again, accuracy is just table stakes.
The authors deployed the model in the live health record and recorded the results. For half of the kids, that was all that happened; no one actually saw the predictions. For those randomized to the intervention, the hematology team would be notified when the risk for clot was calculated to be greater than 2.5%. The hematology team would then contact the primary team to discuss prophylactic anticoagulation.
This is an elegant approach.
Let’s start with those table stakes – accuracy. The predictions were, by and large, pretty accurate in this trial. Of the 135 kids who developed blood clots, 121 had been flagged by the model in advance. That’s about 90%. The model flagged about 10% of kids who didn’t get a blood clot as well, but that’s not entirely surprising since the threshold for flagging was a 2.5% risk.
Given that the model preidentified almost every kid who would go on to develop a blood clot, it would make sense that kids randomized to the intervention would do better; after all, Cassandra was calling out her warnings.
But those kids didn’t do better. The rate of blood clot was no different between the group that used the accurate prediction model and the group that did not.
Why? Why does the use of an accurate model not necessarily improve outcomes?
First of all, a warning must lead to some change in management. Indeed, the kids in the intervention group were more likely to receive anticoagulation, but barely so. There were lots of reasons for this: physician preference, imminent discharge, active bleeding, and so on.
But let’s take a look at the 77 kids in the intervention arm who developed blood clots, because I think this is an instructive analysis.
Six of them did not meet the 2.5% threshold criteria, a case where the model missed its mark. Again, accuracy is table stakes.
Of the remaining 71, only 16 got a recommendation from the hematologist to start anticoagulation. Why not more? Well, the model identified some of the high-risk kids on the weekend, and it seems that the study team did not contact treatment teams during that time. That may account for about 40% of these cases. The remainder had some contraindication to anticoagulation.
Most tellingly, of the 16 who did get a recommendation to start anticoagulation, the recommendation was followed in only seven patients.
This is the gap between accurate prediction and the ability to change outcomes for patients. A prediction is useless if it is wrong, for sure. But it’s also useless if you don’t tell anyone about it. It’s useless if you tell someone but they can’t do anything about it. And it’s useless if they could do something about it but choose not to.
That’s the gulf that these models need to cross at this point. So, the next time some slick company tells you how accurate their AI model is, ask them if accuracy is really the most important thing. If they say, “Well, yes, of course,” then tell them about Cassandra.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Are migraine preventives underused in young adults?
, according to recent research published in the journal Headache.
“Approximately two-fifths of young adults with migraine were prescribed preventive medications, and this did not differ between pediatric and adult neurologists,” Hannah F. J. Shapiro MD, of the department of neurology at the University of California, San Francisco, and the UCSF Benioff Children’s Hospitals, and colleagues wrote in their study. “This finding suggests that pediatric neurologists are providing comparable care to adult neurologists for young adults with migraine; however, this may represent the underuse of preventive medications in this patient population.”
Dr. Shapiro and colleagues conducted a retrospective study of 767 patients (mean age 20.3 years) at Mass General Brigham Hospital in Boston between 2017 and 2021 who received care from a pediatric or adult neurologist for episodic migraine. The majority of patients in the study were white (72.2%), non-Hispanic (82.1%) women (80.3%) with episodic migraine (72.8%), some of whom experienced a psychiatric comorbidity (12.7%), and had a 3.88 mean clinic visits for migraine. Researchers assessed prescription of migraine preventive medication as a primary outcome, with a secondary outcome of comparing the rate of migraine preventive prescriptions written by pediatric and adult neurologists.
Overall, 290 patients (37.8%) received care from a pediatric neurologist, and 131 of those 290 patients (45.2%) received preventive medications (95% confidence interval, 39.5%-51.0%). The remaining 477 patients received care from an adult neurologist; of these, 206 patients (43.2%) received preventive medications (95% CI, 39.0%-47.7%; P = .591). The most common preventive medication prescribed was topiramate, which was prescribed in 19.1% of cases by adult neurologists and 15.2% of cases by pediatric neurologists. Other preventive medications included tricyclic antidepressants such as amitriptyline and nortriptyline; pediatric neurologists prescribed amitriptyline more often than adult neurologists (14.5% vs. 5.5%; P < .001), and adult neurologists prescribed nortriptyline more often than pediatric neurologists (12.8% vs. 2.4%; P < .001).
Dr. Shapiro and colleagues performed a mixed effects logistic regression analysis of potential confounders, and found no significant association between clinician specialty and use of preventive medication (adjusted odds ratio, 1.20; 95% CI, 0.62-2.31), while factors such as female sex (aOR, 1.69; 95% CI, 1.07-2.66) and number of visits (aOR, 1.64; 95% CI, 1.49-1.80) carried associations with preventive medication use.
The finding that pediatric and adult neurologists use similar preventive medications is a positive one because “patients who continue care into adulthood with a pediatric neurologist should receive comparable care to the care they would receive with an adult neurologist,” Dr. Shapiro and colleagues said. “It is even more pertinent now for pediatric neurologists to have comfort prescribing preventive medication to young adults, as the newer calcitonin gene-related peptide (CGRP) pathway antagonists are currently only FDA approved for use in patients aged 18 years or older.”
Roadblocks may prevent adoption
M. Cristina Victorio, MD, a pediatric neurologist and director of the headache program at Akron (Ohio) Children’s, said in an interview that the study is well-designed, but the results cannot be generalized as the study is retrospective, was conducted at a single institution, and data about nutraceuticals and drug-free neuromodulation devices were excluded from the analysis.
Another aspect of the study to consider is that episodic migraine, defined as between 0 and 14 migraine days per month, comprised most of the diagnoses in this study, while preventive medication is usually considered in patients with migraines occurring at least 6 days per month. “[I]f migraine is only once every other month or once a month, preventive treatment may not be recommended,” she said.
There is also the element of patient preference, which is “difficult to obtain” in a retrospective study, she noted.
Citing the authors’ comments about pediatric neurologists’ comfortability prescribing preventive medications, including CGRP antagonists, Dr. Victorio said she offers CGRP antagonists to “young adult patients who have failed at least two of the guideline-recommended preventive medications.”
However, pediatric neurologists may encounter roadblocks to prescribing these medications. “A big challenge is access, as it requires prior authorization as well as writing a letter of appeal or medical necessity, which can be a nuisance for clinicians who are already inundated with clinical responsibilities,” she said.
More education is needed
“As a pediatric headache specialist and knowing the results of this study, my colleagues and I have a role in educating all clinicians as well as trainees on headache management to improve and provide optimal care for young adult patients with migraine,” Dr. Victorio said.
In her experience, more clinic visits usually mean a need for preventative medication, and psychiatric morbidities are common. “I differ in the sense that as a headache specialist I am comfortable offering various preventive treatment options when indicated, so I do not believe I am underutilizing,” she said.
Dr. Victorio said she prescribes topiramate, amitriptyline, and propranolol as migraine preventatives for adolescents and young adults, but recommends cyproheptadine for younger children “due to lesser side effects, tolerability, and convenience of formulation (both liquid and tablet forms are available), which can be challenging for younger children who are unable to swallow pills.”
“Cognizant that there are patients who are reluctant to take daily prescription medication and that consideration for preventive treatment includes patient’s preference, I include the use of nutraceuticals and drug-free neuromodulation devices when discussing preventive treatment options,” she added, noting that children and adolescents “[m]ore often than not” prefer nutraceuticals like magnesium and vitamin B2.
“I think the bottom line is that all clinicians managing young adults with migraine should know when to consider starting preventive migraine medication,” Dr. Victorio said. “Not offering preventive treatment to young adults specifically for those who have frequent migraine attacks, or those who have severe migraine despite adequate acute treatment, or those with significant adverse reactions to acute medications will only put these patients at risk to progression to chronic migraine (meaning having migraine more often than not – at least 15 days per month), and increases headache-related disability and reduces quality of life.”
The authors report no relevant financial disclosures. This study was supported by Harvard University and an award from the National Institutes of Health. Dr. Victorio reports being on the advisory board for Theranica Bio-electronics, has received honorarium serving as an author of the Merck Manual, and is involved in industry-sponsored clinical trials through Akron Children’s Hospital.
, according to recent research published in the journal Headache.
“Approximately two-fifths of young adults with migraine were prescribed preventive medications, and this did not differ between pediatric and adult neurologists,” Hannah F. J. Shapiro MD, of the department of neurology at the University of California, San Francisco, and the UCSF Benioff Children’s Hospitals, and colleagues wrote in their study. “This finding suggests that pediatric neurologists are providing comparable care to adult neurologists for young adults with migraine; however, this may represent the underuse of preventive medications in this patient population.”
Dr. Shapiro and colleagues conducted a retrospective study of 767 patients (mean age 20.3 years) at Mass General Brigham Hospital in Boston between 2017 and 2021 who received care from a pediatric or adult neurologist for episodic migraine. The majority of patients in the study were white (72.2%), non-Hispanic (82.1%) women (80.3%) with episodic migraine (72.8%), some of whom experienced a psychiatric comorbidity (12.7%), and had a 3.88 mean clinic visits for migraine. Researchers assessed prescription of migraine preventive medication as a primary outcome, with a secondary outcome of comparing the rate of migraine preventive prescriptions written by pediatric and adult neurologists.
Overall, 290 patients (37.8%) received care from a pediatric neurologist, and 131 of those 290 patients (45.2%) received preventive medications (95% confidence interval, 39.5%-51.0%). The remaining 477 patients received care from an adult neurologist; of these, 206 patients (43.2%) received preventive medications (95% CI, 39.0%-47.7%; P = .591). The most common preventive medication prescribed was topiramate, which was prescribed in 19.1% of cases by adult neurologists and 15.2% of cases by pediatric neurologists. Other preventive medications included tricyclic antidepressants such as amitriptyline and nortriptyline; pediatric neurologists prescribed amitriptyline more often than adult neurologists (14.5% vs. 5.5%; P < .001), and adult neurologists prescribed nortriptyline more often than pediatric neurologists (12.8% vs. 2.4%; P < .001).
Dr. Shapiro and colleagues performed a mixed effects logistic regression analysis of potential confounders, and found no significant association between clinician specialty and use of preventive medication (adjusted odds ratio, 1.20; 95% CI, 0.62-2.31), while factors such as female sex (aOR, 1.69; 95% CI, 1.07-2.66) and number of visits (aOR, 1.64; 95% CI, 1.49-1.80) carried associations with preventive medication use.
The finding that pediatric and adult neurologists use similar preventive medications is a positive one because “patients who continue care into adulthood with a pediatric neurologist should receive comparable care to the care they would receive with an adult neurologist,” Dr. Shapiro and colleagues said. “It is even more pertinent now for pediatric neurologists to have comfort prescribing preventive medication to young adults, as the newer calcitonin gene-related peptide (CGRP) pathway antagonists are currently only FDA approved for use in patients aged 18 years or older.”
Roadblocks may prevent adoption
M. Cristina Victorio, MD, a pediatric neurologist and director of the headache program at Akron (Ohio) Children’s, said in an interview that the study is well-designed, but the results cannot be generalized as the study is retrospective, was conducted at a single institution, and data about nutraceuticals and drug-free neuromodulation devices were excluded from the analysis.
Another aspect of the study to consider is that episodic migraine, defined as between 0 and 14 migraine days per month, comprised most of the diagnoses in this study, while preventive medication is usually considered in patients with migraines occurring at least 6 days per month. “[I]f migraine is only once every other month or once a month, preventive treatment may not be recommended,” she said.
There is also the element of patient preference, which is “difficult to obtain” in a retrospective study, she noted.
Citing the authors’ comments about pediatric neurologists’ comfortability prescribing preventive medications, including CGRP antagonists, Dr. Victorio said she offers CGRP antagonists to “young adult patients who have failed at least two of the guideline-recommended preventive medications.”
However, pediatric neurologists may encounter roadblocks to prescribing these medications. “A big challenge is access, as it requires prior authorization as well as writing a letter of appeal or medical necessity, which can be a nuisance for clinicians who are already inundated with clinical responsibilities,” she said.
More education is needed
“As a pediatric headache specialist and knowing the results of this study, my colleagues and I have a role in educating all clinicians as well as trainees on headache management to improve and provide optimal care for young adult patients with migraine,” Dr. Victorio said.
In her experience, more clinic visits usually mean a need for preventative medication, and psychiatric morbidities are common. “I differ in the sense that as a headache specialist I am comfortable offering various preventive treatment options when indicated, so I do not believe I am underutilizing,” she said.
Dr. Victorio said she prescribes topiramate, amitriptyline, and propranolol as migraine preventatives for adolescents and young adults, but recommends cyproheptadine for younger children “due to lesser side effects, tolerability, and convenience of formulation (both liquid and tablet forms are available), which can be challenging for younger children who are unable to swallow pills.”
“Cognizant that there are patients who are reluctant to take daily prescription medication and that consideration for preventive treatment includes patient’s preference, I include the use of nutraceuticals and drug-free neuromodulation devices when discussing preventive treatment options,” she added, noting that children and adolescents “[m]ore often than not” prefer nutraceuticals like magnesium and vitamin B2.
“I think the bottom line is that all clinicians managing young adults with migraine should know when to consider starting preventive migraine medication,” Dr. Victorio said. “Not offering preventive treatment to young adults specifically for those who have frequent migraine attacks, or those who have severe migraine despite adequate acute treatment, or those with significant adverse reactions to acute medications will only put these patients at risk to progression to chronic migraine (meaning having migraine more often than not – at least 15 days per month), and increases headache-related disability and reduces quality of life.”
The authors report no relevant financial disclosures. This study was supported by Harvard University and an award from the National Institutes of Health. Dr. Victorio reports being on the advisory board for Theranica Bio-electronics, has received honorarium serving as an author of the Merck Manual, and is involved in industry-sponsored clinical trials through Akron Children’s Hospital.
, according to recent research published in the journal Headache.
“Approximately two-fifths of young adults with migraine were prescribed preventive medications, and this did not differ between pediatric and adult neurologists,” Hannah F. J. Shapiro MD, of the department of neurology at the University of California, San Francisco, and the UCSF Benioff Children’s Hospitals, and colleagues wrote in their study. “This finding suggests that pediatric neurologists are providing comparable care to adult neurologists for young adults with migraine; however, this may represent the underuse of preventive medications in this patient population.”
Dr. Shapiro and colleagues conducted a retrospective study of 767 patients (mean age 20.3 years) at Mass General Brigham Hospital in Boston between 2017 and 2021 who received care from a pediatric or adult neurologist for episodic migraine. The majority of patients in the study were white (72.2%), non-Hispanic (82.1%) women (80.3%) with episodic migraine (72.8%), some of whom experienced a psychiatric comorbidity (12.7%), and had a 3.88 mean clinic visits for migraine. Researchers assessed prescription of migraine preventive medication as a primary outcome, with a secondary outcome of comparing the rate of migraine preventive prescriptions written by pediatric and adult neurologists.
Overall, 290 patients (37.8%) received care from a pediatric neurologist, and 131 of those 290 patients (45.2%) received preventive medications (95% confidence interval, 39.5%-51.0%). The remaining 477 patients received care from an adult neurologist; of these, 206 patients (43.2%) received preventive medications (95% CI, 39.0%-47.7%; P = .591). The most common preventive medication prescribed was topiramate, which was prescribed in 19.1% of cases by adult neurologists and 15.2% of cases by pediatric neurologists. Other preventive medications included tricyclic antidepressants such as amitriptyline and nortriptyline; pediatric neurologists prescribed amitriptyline more often than adult neurologists (14.5% vs. 5.5%; P < .001), and adult neurologists prescribed nortriptyline more often than pediatric neurologists (12.8% vs. 2.4%; P < .001).
Dr. Shapiro and colleagues performed a mixed effects logistic regression analysis of potential confounders, and found no significant association between clinician specialty and use of preventive medication (adjusted odds ratio, 1.20; 95% CI, 0.62-2.31), while factors such as female sex (aOR, 1.69; 95% CI, 1.07-2.66) and number of visits (aOR, 1.64; 95% CI, 1.49-1.80) carried associations with preventive medication use.
The finding that pediatric and adult neurologists use similar preventive medications is a positive one because “patients who continue care into adulthood with a pediatric neurologist should receive comparable care to the care they would receive with an adult neurologist,” Dr. Shapiro and colleagues said. “It is even more pertinent now for pediatric neurologists to have comfort prescribing preventive medication to young adults, as the newer calcitonin gene-related peptide (CGRP) pathway antagonists are currently only FDA approved for use in patients aged 18 years or older.”
Roadblocks may prevent adoption
M. Cristina Victorio, MD, a pediatric neurologist and director of the headache program at Akron (Ohio) Children’s, said in an interview that the study is well-designed, but the results cannot be generalized as the study is retrospective, was conducted at a single institution, and data about nutraceuticals and drug-free neuromodulation devices were excluded from the analysis.
Another aspect of the study to consider is that episodic migraine, defined as between 0 and 14 migraine days per month, comprised most of the diagnoses in this study, while preventive medication is usually considered in patients with migraines occurring at least 6 days per month. “[I]f migraine is only once every other month or once a month, preventive treatment may not be recommended,” she said.
There is also the element of patient preference, which is “difficult to obtain” in a retrospective study, she noted.
Citing the authors’ comments about pediatric neurologists’ comfortability prescribing preventive medications, including CGRP antagonists, Dr. Victorio said she offers CGRP antagonists to “young adult patients who have failed at least two of the guideline-recommended preventive medications.”
However, pediatric neurologists may encounter roadblocks to prescribing these medications. “A big challenge is access, as it requires prior authorization as well as writing a letter of appeal or medical necessity, which can be a nuisance for clinicians who are already inundated with clinical responsibilities,” she said.
More education is needed
“As a pediatric headache specialist and knowing the results of this study, my colleagues and I have a role in educating all clinicians as well as trainees on headache management to improve and provide optimal care for young adult patients with migraine,” Dr. Victorio said.
In her experience, more clinic visits usually mean a need for preventative medication, and psychiatric morbidities are common. “I differ in the sense that as a headache specialist I am comfortable offering various preventive treatment options when indicated, so I do not believe I am underutilizing,” she said.
Dr. Victorio said she prescribes topiramate, amitriptyline, and propranolol as migraine preventatives for adolescents and young adults, but recommends cyproheptadine for younger children “due to lesser side effects, tolerability, and convenience of formulation (both liquid and tablet forms are available), which can be challenging for younger children who are unable to swallow pills.”
“Cognizant that there are patients who are reluctant to take daily prescription medication and that consideration for preventive treatment includes patient’s preference, I include the use of nutraceuticals and drug-free neuromodulation devices when discussing preventive treatment options,” she added, noting that children and adolescents “[m]ore often than not” prefer nutraceuticals like magnesium and vitamin B2.
“I think the bottom line is that all clinicians managing young adults with migraine should know when to consider starting preventive migraine medication,” Dr. Victorio said. “Not offering preventive treatment to young adults specifically for those who have frequent migraine attacks, or those who have severe migraine despite adequate acute treatment, or those with significant adverse reactions to acute medications will only put these patients at risk to progression to chronic migraine (meaning having migraine more often than not – at least 15 days per month), and increases headache-related disability and reduces quality of life.”
The authors report no relevant financial disclosures. This study was supported by Harvard University and an award from the National Institutes of Health. Dr. Victorio reports being on the advisory board for Theranica Bio-electronics, has received honorarium serving as an author of the Merck Manual, and is involved in industry-sponsored clinical trials through Akron Children’s Hospital.
FROM HEADACHE
Metformin, weight management to stop type 2 diabetes in kids
TOPLINE:
Nearly one in five adolescents are living with prediabetes, a condition where blood glucose levels are elevated, but are not high enough for a type 2 diabetes (T2D) diagnosis. According to a new study, higher levels of nonfasting glucose and hemoglobin A1c, and worsening obesity are important predictors of progression to T2D. In addition, metformin and weight stabilization may prove to be important interventions for preventing T2D in kids.
METHODOLOGY:
- Researchers did a retrospective chart review of patient data from Vanderbilt University Medical Center Pediatric Prediabetes Clinic, Nashville, Tenn., from May 2015 to August 2022.
- The study included 552 children with prediabetes, defined as abnormal blood glucose (fasting plasma glucose [FPG] ≥ 100 mg/dL, random glucose ≥ 150 mg/dL), or hemoglobin A1c equal to or greater than 5.9%.
- Based on follow-up visits, patients were classified as having progressed to T2D, or nonprogression.
- Researchers analyzed the patients’ initial visit A1c, fasting C-peptide, 2-hour glucose, fasting glucose, and body mass index (BMI), among other baseline characteristics.
TAKEAWAY:
- Thirty-six children (6.5%) progressed to T2D during the duration of the study period.
- The average time to T2D diagnosis was much longer in patients taking metformin (43 months), compared with those not taking the prescribed medication (28 months).
- Worsening obesity was strongly associated with T2D progression – patients who progressed to T2D had a higher BMI at baseline and had continued weight gain.
- A higher baseline A1c, fasting C-peptide, and 2-hour glucose were also associated with progression to T2D.
- In the multivariable analysis, both A1c and 2-hour glucose were strong independent predictors of progression.
- Fasting plasma glucose was not associated with progression to T2D.
IN PRACTICE:
“Weight stabilization and metformin therapy could be important interventions for diabetes prevention in children,” study author Ashley H. Shoemaker, MD, MSci, a pediatric endocrinologist at Vanderbilt University Medical Center in Nashville, Tenn., said in a press release.
In addition, A1c plus a nonfasting glucose may be a feasible way to identify high-risk pediatric patients in a clinical setting.
SOURCE:
This study was performed by Natasha Belsky, Jaclyn Tamaroff, and Ashley H. Shoemaker of the Vanderbilt University Medical Center and the Vanderbilt University School of Medicine in Nashville, Tenn. It was published October 12, 2023, in the Journal of the Endocrine Society
LIMITATIONS:
Additional patients who developed T2D may have been lost to follow-up, since the authors did not contact patients to confirm their disease status. The authors were also unable to establish racial differences in the progression to T2D because of missing data.
DISCLOSURES:
Funding for this study was provided by the National Center for Advancing Translational Sciences. One author has research contracts with Novo Nordisk and Boehringer Ingelheim.
A version of this article first appeared on Medscape.com.
TOPLINE:
Nearly one in five adolescents are living with prediabetes, a condition where blood glucose levels are elevated, but are not high enough for a type 2 diabetes (T2D) diagnosis. According to a new study, higher levels of nonfasting glucose and hemoglobin A1c, and worsening obesity are important predictors of progression to T2D. In addition, metformin and weight stabilization may prove to be important interventions for preventing T2D in kids.
METHODOLOGY:
- Researchers did a retrospective chart review of patient data from Vanderbilt University Medical Center Pediatric Prediabetes Clinic, Nashville, Tenn., from May 2015 to August 2022.
- The study included 552 children with prediabetes, defined as abnormal blood glucose (fasting plasma glucose [FPG] ≥ 100 mg/dL, random glucose ≥ 150 mg/dL), or hemoglobin A1c equal to or greater than 5.9%.
- Based on follow-up visits, patients were classified as having progressed to T2D, or nonprogression.
- Researchers analyzed the patients’ initial visit A1c, fasting C-peptide, 2-hour glucose, fasting glucose, and body mass index (BMI), among other baseline characteristics.
TAKEAWAY:
- Thirty-six children (6.5%) progressed to T2D during the duration of the study period.
- The average time to T2D diagnosis was much longer in patients taking metformin (43 months), compared with those not taking the prescribed medication (28 months).
- Worsening obesity was strongly associated with T2D progression – patients who progressed to T2D had a higher BMI at baseline and had continued weight gain.
- A higher baseline A1c, fasting C-peptide, and 2-hour glucose were also associated with progression to T2D.
- In the multivariable analysis, both A1c and 2-hour glucose were strong independent predictors of progression.
- Fasting plasma glucose was not associated with progression to T2D.
IN PRACTICE:
“Weight stabilization and metformin therapy could be important interventions for diabetes prevention in children,” study author Ashley H. Shoemaker, MD, MSci, a pediatric endocrinologist at Vanderbilt University Medical Center in Nashville, Tenn., said in a press release.
In addition, A1c plus a nonfasting glucose may be a feasible way to identify high-risk pediatric patients in a clinical setting.
SOURCE:
This study was performed by Natasha Belsky, Jaclyn Tamaroff, and Ashley H. Shoemaker of the Vanderbilt University Medical Center and the Vanderbilt University School of Medicine in Nashville, Tenn. It was published October 12, 2023, in the Journal of the Endocrine Society
LIMITATIONS:
Additional patients who developed T2D may have been lost to follow-up, since the authors did not contact patients to confirm their disease status. The authors were also unable to establish racial differences in the progression to T2D because of missing data.
DISCLOSURES:
Funding for this study was provided by the National Center for Advancing Translational Sciences. One author has research contracts with Novo Nordisk and Boehringer Ingelheim.
A version of this article first appeared on Medscape.com.
TOPLINE:
Nearly one in five adolescents are living with prediabetes, a condition where blood glucose levels are elevated, but are not high enough for a type 2 diabetes (T2D) diagnosis. According to a new study, higher levels of nonfasting glucose and hemoglobin A1c, and worsening obesity are important predictors of progression to T2D. In addition, metformin and weight stabilization may prove to be important interventions for preventing T2D in kids.
METHODOLOGY:
- Researchers did a retrospective chart review of patient data from Vanderbilt University Medical Center Pediatric Prediabetes Clinic, Nashville, Tenn., from May 2015 to August 2022.
- The study included 552 children with prediabetes, defined as abnormal blood glucose (fasting plasma glucose [FPG] ≥ 100 mg/dL, random glucose ≥ 150 mg/dL), or hemoglobin A1c equal to or greater than 5.9%.
- Based on follow-up visits, patients were classified as having progressed to T2D, or nonprogression.
- Researchers analyzed the patients’ initial visit A1c, fasting C-peptide, 2-hour glucose, fasting glucose, and body mass index (BMI), among other baseline characteristics.
TAKEAWAY:
- Thirty-six children (6.5%) progressed to T2D during the duration of the study period.
- The average time to T2D diagnosis was much longer in patients taking metformin (43 months), compared with those not taking the prescribed medication (28 months).
- Worsening obesity was strongly associated with T2D progression – patients who progressed to T2D had a higher BMI at baseline and had continued weight gain.
- A higher baseline A1c, fasting C-peptide, and 2-hour glucose were also associated with progression to T2D.
- In the multivariable analysis, both A1c and 2-hour glucose were strong independent predictors of progression.
- Fasting plasma glucose was not associated with progression to T2D.
IN PRACTICE:
“Weight stabilization and metformin therapy could be important interventions for diabetes prevention in children,” study author Ashley H. Shoemaker, MD, MSci, a pediatric endocrinologist at Vanderbilt University Medical Center in Nashville, Tenn., said in a press release.
In addition, A1c plus a nonfasting glucose may be a feasible way to identify high-risk pediatric patients in a clinical setting.
SOURCE:
This study was performed by Natasha Belsky, Jaclyn Tamaroff, and Ashley H. Shoemaker of the Vanderbilt University Medical Center and the Vanderbilt University School of Medicine in Nashville, Tenn. It was published October 12, 2023, in the Journal of the Endocrine Society
LIMITATIONS:
Additional patients who developed T2D may have been lost to follow-up, since the authors did not contact patients to confirm their disease status. The authors were also unable to establish racial differences in the progression to T2D because of missing data.
DISCLOSURES:
Funding for this study was provided by the National Center for Advancing Translational Sciences. One author has research contracts with Novo Nordisk and Boehringer Ingelheim.
A version of this article first appeared on Medscape.com.
What's the diagnosis?
The lesions on the heels are consistent with piezogenic pedal papules. They seem to be more common in women and have been described in families, though a genetic link hasn’t been elucidated. PPP manifests as small, soft, compressible papules on the lateral aspects of the skin on the heels, more noticeable when the patient is standing, and can also present on the wrists and legs. While they may not be a cause for serious concern, understanding their causes, associated conditions, and management is important.
Piezogenic pedal papules are flesh-colored or slightly reddish and can range in size from a few millimeters to a centimeter or more. They are described as benign herniations of elastic tissue and subcutaneous fat through the reticular dermis. The lesions are triggered by increased pressure and compression, such as standing or the application of pressure on the heel. The exact etiology is not known. While piezogenic pedal papules are often asymptomatic, some individuals experience discomfort, itching, or mild pain, particularly when walking or applying pressure to the affected area, especially in patients with Ehlers-Danlos syndrome (EDS).
Individuals who may be at risk of developing these lesions include obese patients, individuals with pes planus, and people who have occupations that require long periods of standing. It can also be seen in athletes who participate in long-distance running or high-impact sports. Piezogenic pedal papules have been described as one of the core skin findings in patients with hypermobile Ehlers-Danlos syndrome (hEDS), which also includes skin hyperextensibility, joint hypermobility, tissue fragility with atrophic cutaneous scars, and abnormal bruising and bleeding. Our patient presented with some of these characteristics (piezogenic papules, soft elastic skin, and some joint hypermobility) but did not fulfill all the criteria for the diagnosis of hEDS or other types of EDS.
The diagnosis of hEDS is based on the 2017 diagnostic criteria checklist. To be diagnosed with hEDS, the patient may have all three parts of the diagnostic criteria. The three domains include generalized joint hypermobility (partially met by our patient), evidence of syndromic features, musculoskeletal complications, and/or family history (she had a few of these criteria, including piezogenic papules and striae), and the exclusion of alternative diagnoses (see references for the PDF checklist). As she does have some features, we diagnosed her with hypermobility spectrum disorder. There is no genetic testing available for the hypermobile spectrum disorder or the hypermobile type of EDS. Given that these patients can present with mitral valve prolapse, she was referred to a cardiac echocardiogram, which was reported as normal.
The diagnosis of PPP is made clinically, and rarely a biopsy is required. Biopsies of the lesions show hyperkeratosis, degeneration of the thin fibrous septa between fat lobules, and subsequent coalescence of fat. If the presentation is atypical, a high-frequency ultrasound can be requested to confirm the physical exam findings.
If the lesions are fixed, firm, and solitary, a diagnosis to consider is juvenile aponeurotic fibroma, which occurs more often in children and adolescents on the wrists and is less common on the ankles. If there is suspicion for this condition, a plain radiograph will show stippled calcifications.
PPP are usually asymptomatic and need no further treatment. When they are symptomatic, conservative management should be considered first, which includes behavioral modifications, weight loss, avoidance of prolonged standing, and reduced foot trauma. If these are not successful, compression socks, heel cups, and other orthotics can be recommended. Intralesional injections of betamethasone and bupivacaine have been reported as an option in patients with symptomatic PPP and a history of EDS.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
Suggested reading
Edimo CO et al. Int J Womens Dermatol. 2021 Jan. doi: 10.1016/j.ijwd.2021.01.020. Erratum in: Int J Womens Dermatol. 2021 Jul 31;7(5Part B):869-70.
Brown F, Cook C. Piezogenic Pedal Papule. 2023 Aug 16. In: StatPearls [Internet]. Treasure Island, Fla.: StatPearls Publishing, 2023. PMID: 29489228.
Levy HP. Hypermobile Ehlers-Danlos Syndrome. 2004 Oct 22 [Updated 2018 Jun 21]. In: Adam MP, Mirzaa GM, Pagon RA, et al., editors. GeneReviews® [Internet]. Seattle: University of Washington, Seattle; 1993-2023. Available from: www.ncbi.nlm.nih.gov/books/NBK1279/.
The International Consortium on Ehlers-Danlos Syndrome & Related Disorders. Diagnostic Criteria for Hypermobile Ehlers-Danlos Syndrome (hEDS). www.ehlers-danlos.com/wp-content/uploads/2017/05/hEDS-Dx-Criteria-checklist-1.pdf.
The lesions on the heels are consistent with piezogenic pedal papules. They seem to be more common in women and have been described in families, though a genetic link hasn’t been elucidated. PPP manifests as small, soft, compressible papules on the lateral aspects of the skin on the heels, more noticeable when the patient is standing, and can also present on the wrists and legs. While they may not be a cause for serious concern, understanding their causes, associated conditions, and management is important.
Piezogenic pedal papules are flesh-colored or slightly reddish and can range in size from a few millimeters to a centimeter or more. They are described as benign herniations of elastic tissue and subcutaneous fat through the reticular dermis. The lesions are triggered by increased pressure and compression, such as standing or the application of pressure on the heel. The exact etiology is not known. While piezogenic pedal papules are often asymptomatic, some individuals experience discomfort, itching, or mild pain, particularly when walking or applying pressure to the affected area, especially in patients with Ehlers-Danlos syndrome (EDS).
Individuals who may be at risk of developing these lesions include obese patients, individuals with pes planus, and people who have occupations that require long periods of standing. It can also be seen in athletes who participate in long-distance running or high-impact sports. Piezogenic pedal papules have been described as one of the core skin findings in patients with hypermobile Ehlers-Danlos syndrome (hEDS), which also includes skin hyperextensibility, joint hypermobility, tissue fragility with atrophic cutaneous scars, and abnormal bruising and bleeding. Our patient presented with some of these characteristics (piezogenic papules, soft elastic skin, and some joint hypermobility) but did not fulfill all the criteria for the diagnosis of hEDS or other types of EDS.
The diagnosis of hEDS is based on the 2017 diagnostic criteria checklist. To be diagnosed with hEDS, the patient may have all three parts of the diagnostic criteria. The three domains include generalized joint hypermobility (partially met by our patient), evidence of syndromic features, musculoskeletal complications, and/or family history (she had a few of these criteria, including piezogenic papules and striae), and the exclusion of alternative diagnoses (see references for the PDF checklist). As she does have some features, we diagnosed her with hypermobility spectrum disorder. There is no genetic testing available for the hypermobile spectrum disorder or the hypermobile type of EDS. Given that these patients can present with mitral valve prolapse, she was referred to a cardiac echocardiogram, which was reported as normal.
The diagnosis of PPP is made clinically, and rarely a biopsy is required. Biopsies of the lesions show hyperkeratosis, degeneration of the thin fibrous septa between fat lobules, and subsequent coalescence of fat. If the presentation is atypical, a high-frequency ultrasound can be requested to confirm the physical exam findings.
If the lesions are fixed, firm, and solitary, a diagnosis to consider is juvenile aponeurotic fibroma, which occurs more often in children and adolescents on the wrists and is less common on the ankles. If there is suspicion for this condition, a plain radiograph will show stippled calcifications.
PPP are usually asymptomatic and need no further treatment. When they are symptomatic, conservative management should be considered first, which includes behavioral modifications, weight loss, avoidance of prolonged standing, and reduced foot trauma. If these are not successful, compression socks, heel cups, and other orthotics can be recommended. Intralesional injections of betamethasone and bupivacaine have been reported as an option in patients with symptomatic PPP and a history of EDS.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
Suggested reading
Edimo CO et al. Int J Womens Dermatol. 2021 Jan. doi: 10.1016/j.ijwd.2021.01.020. Erratum in: Int J Womens Dermatol. 2021 Jul 31;7(5Part B):869-70.
Brown F, Cook C. Piezogenic Pedal Papule. 2023 Aug 16. In: StatPearls [Internet]. Treasure Island, Fla.: StatPearls Publishing, 2023. PMID: 29489228.
Levy HP. Hypermobile Ehlers-Danlos Syndrome. 2004 Oct 22 [Updated 2018 Jun 21]. In: Adam MP, Mirzaa GM, Pagon RA, et al., editors. GeneReviews® [Internet]. Seattle: University of Washington, Seattle; 1993-2023. Available from: www.ncbi.nlm.nih.gov/books/NBK1279/.
The International Consortium on Ehlers-Danlos Syndrome & Related Disorders. Diagnostic Criteria for Hypermobile Ehlers-Danlos Syndrome (hEDS). www.ehlers-danlos.com/wp-content/uploads/2017/05/hEDS-Dx-Criteria-checklist-1.pdf.
The lesions on the heels are consistent with piezogenic pedal papules. They seem to be more common in women and have been described in families, though a genetic link hasn’t been elucidated. PPP manifests as small, soft, compressible papules on the lateral aspects of the skin on the heels, more noticeable when the patient is standing, and can also present on the wrists and legs. While they may not be a cause for serious concern, understanding their causes, associated conditions, and management is important.
Piezogenic pedal papules are flesh-colored or slightly reddish and can range in size from a few millimeters to a centimeter or more. They are described as benign herniations of elastic tissue and subcutaneous fat through the reticular dermis. The lesions are triggered by increased pressure and compression, such as standing or the application of pressure on the heel. The exact etiology is not known. While piezogenic pedal papules are often asymptomatic, some individuals experience discomfort, itching, or mild pain, particularly when walking or applying pressure to the affected area, especially in patients with Ehlers-Danlos syndrome (EDS).
Individuals who may be at risk of developing these lesions include obese patients, individuals with pes planus, and people who have occupations that require long periods of standing. It can also be seen in athletes who participate in long-distance running or high-impact sports. Piezogenic pedal papules have been described as one of the core skin findings in patients with hypermobile Ehlers-Danlos syndrome (hEDS), which also includes skin hyperextensibility, joint hypermobility, tissue fragility with atrophic cutaneous scars, and abnormal bruising and bleeding. Our patient presented with some of these characteristics (piezogenic papules, soft elastic skin, and some joint hypermobility) but did not fulfill all the criteria for the diagnosis of hEDS or other types of EDS.
The diagnosis of hEDS is based on the 2017 diagnostic criteria checklist. To be diagnosed with hEDS, the patient may have all three parts of the diagnostic criteria. The three domains include generalized joint hypermobility (partially met by our patient), evidence of syndromic features, musculoskeletal complications, and/or family history (she had a few of these criteria, including piezogenic papules and striae), and the exclusion of alternative diagnoses (see references for the PDF checklist). As she does have some features, we diagnosed her with hypermobility spectrum disorder. There is no genetic testing available for the hypermobile spectrum disorder or the hypermobile type of EDS. Given that these patients can present with mitral valve prolapse, she was referred to a cardiac echocardiogram, which was reported as normal.
The diagnosis of PPP is made clinically, and rarely a biopsy is required. Biopsies of the lesions show hyperkeratosis, degeneration of the thin fibrous septa between fat lobules, and subsequent coalescence of fat. If the presentation is atypical, a high-frequency ultrasound can be requested to confirm the physical exam findings.
If the lesions are fixed, firm, and solitary, a diagnosis to consider is juvenile aponeurotic fibroma, which occurs more often in children and adolescents on the wrists and is less common on the ankles. If there is suspicion for this condition, a plain radiograph will show stippled calcifications.
PPP are usually asymptomatic and need no further treatment. When they are symptomatic, conservative management should be considered first, which includes behavioral modifications, weight loss, avoidance of prolonged standing, and reduced foot trauma. If these are not successful, compression socks, heel cups, and other orthotics can be recommended. Intralesional injections of betamethasone and bupivacaine have been reported as an option in patients with symptomatic PPP and a history of EDS.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
Suggested reading
Edimo CO et al. Int J Womens Dermatol. 2021 Jan. doi: 10.1016/j.ijwd.2021.01.020. Erratum in: Int J Womens Dermatol. 2021 Jul 31;7(5Part B):869-70.
Brown F, Cook C. Piezogenic Pedal Papule. 2023 Aug 16. In: StatPearls [Internet]. Treasure Island, Fla.: StatPearls Publishing, 2023. PMID: 29489228.
Levy HP. Hypermobile Ehlers-Danlos Syndrome. 2004 Oct 22 [Updated 2018 Jun 21]. In: Adam MP, Mirzaa GM, Pagon RA, et al., editors. GeneReviews® [Internet]. Seattle: University of Washington, Seattle; 1993-2023. Available from: www.ncbi.nlm.nih.gov/books/NBK1279/.
The International Consortium on Ehlers-Danlos Syndrome & Related Disorders. Diagnostic Criteria for Hypermobile Ehlers-Danlos Syndrome (hEDS). www.ehlers-danlos.com/wp-content/uploads/2017/05/hEDS-Dx-Criteria-checklist-1.pdf.
On a physical exam, she has several skin-colored soft papules on the heels when she stands up (Picture 1).
She is not able to touch the floor without bending her knees, and she has normal extension of her arms and knees. She has no bruises or abnormal scars and has some striae on her legs.