User login
News and Views that Matter to Pediatricians
The leading independent newspaper covering news and commentary in pediatrics.
Science lags behind for kids with long COVID
Emma Sherman, a 13-year-old girl in Ascot, England, woke up to a dizzying aura of blind spots and flashing lights in her field of vision. It was May 2020, and she also had crippling nausea and headaches. By August, her dizziness was so overwhelming, she couldn’t hold her head up, lying in her mother’s lap for hours, too fatigued to attend school.
The former competitive gymnast, who had hoped to try out for the cheerleading squad, now used a wheelchair and was a shadow of her former self. She had been diagnosed with COVID-induced postural orthostatic tachycardia syndrome, a condition often caused by an infection that results in a higher heart rate, extreme nausea, dizziness, and fatigue.
“I was so into sports before I got long COVID, and afterwards I could barely walk,” Emma said.
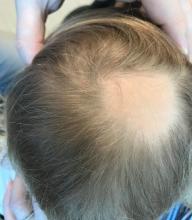
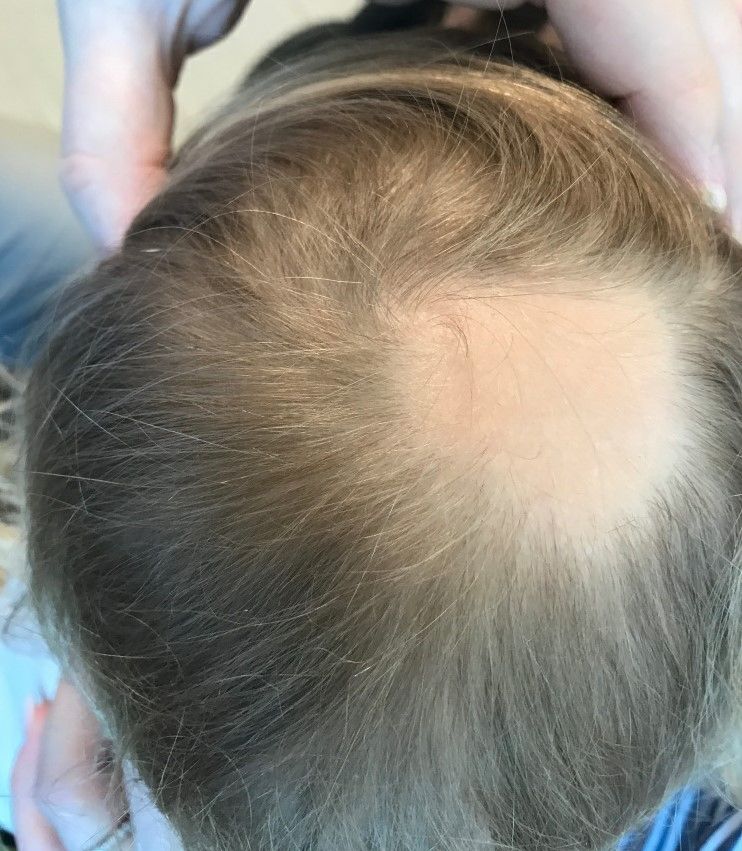
Even minor movements sent her heart rate sky-high. Her long chestnut hair turned gray and fell out in clumps. In the hospital, she was pricked and prodded, her blood tested for numerous conditions.
“They ran every scan known to man and took an MRI of her brain,” said Emma’s mother, Marie Sherman. “All was clear.”
Emma’s pediatrician determined that the teen had long COVID after having had a mild case of the virus in March, about 2 months before her puzzling symptoms began. But beyond a positive antibody test, doctors have found little evidence of what was causing Emma’s symptoms.
For Emma and others with long COVID, there are no medications shown to directly target the condition. Instead, caregivers target their symptoms, which include nausea, dizziness, fatigue, headaches, and a racing heart, said Laura Malone, MD, codirector of the Johns Hopkins Kennedy Krieger Pediatric Post–COVID-19 Rehabilitation Clinic in Baltimore.
“Right now, it’s a rehabilitation-based approach focused on improving symptoms and functioning so that kids can go back to their usual activities as much as possible,” she says.
Depression and anxiety are common, although doctors are struggling to figure out whether COVID is changing the brain or whether mental health symptoms result from all the life disruptions. There’s little research to show how may kids have depression because of long COVID. Dr. Malone said about half of her patients at the Kennedy Krieger Institute›s long COVID clinic are also dealing with mental health issues.
Patients with headaches, dizziness, and nausea are given pain and nausea medications and recommendations for a healthy diet with added fruits and vegetables, monounsaturated fats, lower sodium, unprocessed foods, and whole grains. Kids with irregular or racing heart rates are referred to cardiologists and potentially prescribed beta-blockers to treat their heart arrhythmias, while children with breathing problems may be referred to pulmonologists and those with depression to a psychiatrist.
Still, many patients like Emma go to their doctors with phantom symptoms that don’t show up on scans or blood tests.
“We’re not seeing any evidence of structural damage to the brain, for example,” said Dr. Malone. “When we do MRIs, they often come out normal.”
It’s possible that the virus lingers in some patients, said Rajeev Fernando, MD, an infectious disease specialist and a fellow at Harvard Medical School, Boston. Kids’ strong immune systems often fend off problems that can be noticed. But on the inside, dead fragments of the virus persist, floating in hidden parts of the body and activating the immune system long after the threat has passed.
The virus can be in the gut and in the brain, which may help explain why symptoms like brain fog and nausea can linger in children.
“The immune system doesn’t recognize whether fragments of the virus are dead or alive. It continues to think it’s fighting active COVID,” said Dr. Fernando.
There is little data on how long symptoms last, Dr. Fernando said, as well as how many kids get them and why some are more vulnerable than others. Some research has found that about 5%-15% of children with COVID may get long COVID, but the statistics vary globally.
“Children with long COVID have largely been ignored. And while we’re talking about it now, we’ve got some work to do,” said Dr. Fernando.
As for Emma, she recovered in January of 2021, heading back to school and her friends, although her cardiologist advised her to skip gym classes.
“For the first time in months, I was feeling like myself again,” she said.
But the coronavirus found its way to Emma again. Although she was fully vaccinated in the fall of 2021, when the Omicron variant swept the world late that year, she was infected again.
“When the wave of Omicron descended, Emma was like a sitting duck,” her mother said.
She was bedridden with a high fever and cough. The cold-like symptoms eventually went away, but the issues in her gut stuck around. Since then, Emma has had extreme nausea, losing most of the weight she had gained back.
For her part, Ms. Sherman has found solace in a group called Long COVID Kids, a nonprofit in Europe and the United States. The group is raising awareness about the condition in kids to increase funding, boost understanding, and improve treatment and outcomes.
“There’s nothing worse than watching your child suffer and not being able to do anything about it,” she said. “I tell Emma all the time: If I could just crawl in your body and take it, I would do it in a second.”
Emma is hoping for a fresh start with her family’s move in the coming weeks to Sotogrande in southern Spain.
“I miss the simplest things like going for a run, going to the fair with my friends, and just feeling well,” she said. “I have a long list of things I’ll do once this is all done.”
A version of this article first appeared on WebMD.com.
Emma Sherman, a 13-year-old girl in Ascot, England, woke up to a dizzying aura of blind spots and flashing lights in her field of vision. It was May 2020, and she also had crippling nausea and headaches. By August, her dizziness was so overwhelming, she couldn’t hold her head up, lying in her mother’s lap for hours, too fatigued to attend school.
The former competitive gymnast, who had hoped to try out for the cheerleading squad, now used a wheelchair and was a shadow of her former self. She had been diagnosed with COVID-induced postural orthostatic tachycardia syndrome, a condition often caused by an infection that results in a higher heart rate, extreme nausea, dizziness, and fatigue.
“I was so into sports before I got long COVID, and afterwards I could barely walk,” Emma said.
Even minor movements sent her heart rate sky-high. Her long chestnut hair turned gray and fell out in clumps. In the hospital, she was pricked and prodded, her blood tested for numerous conditions.
“They ran every scan known to man and took an MRI of her brain,” said Emma’s mother, Marie Sherman. “All was clear.”
Emma’s pediatrician determined that the teen had long COVID after having had a mild case of the virus in March, about 2 months before her puzzling symptoms began. But beyond a positive antibody test, doctors have found little evidence of what was causing Emma’s symptoms.
For Emma and others with long COVID, there are no medications shown to directly target the condition. Instead, caregivers target their symptoms, which include nausea, dizziness, fatigue, headaches, and a racing heart, said Laura Malone, MD, codirector of the Johns Hopkins Kennedy Krieger Pediatric Post–COVID-19 Rehabilitation Clinic in Baltimore.
“Right now, it’s a rehabilitation-based approach focused on improving symptoms and functioning so that kids can go back to their usual activities as much as possible,” she says.
Depression and anxiety are common, although doctors are struggling to figure out whether COVID is changing the brain or whether mental health symptoms result from all the life disruptions. There’s little research to show how may kids have depression because of long COVID. Dr. Malone said about half of her patients at the Kennedy Krieger Institute›s long COVID clinic are also dealing with mental health issues.
Patients with headaches, dizziness, and nausea are given pain and nausea medications and recommendations for a healthy diet with added fruits and vegetables, monounsaturated fats, lower sodium, unprocessed foods, and whole grains. Kids with irregular or racing heart rates are referred to cardiologists and potentially prescribed beta-blockers to treat their heart arrhythmias, while children with breathing problems may be referred to pulmonologists and those with depression to a psychiatrist.
Still, many patients like Emma go to their doctors with phantom symptoms that don’t show up on scans or blood tests.
“We’re not seeing any evidence of structural damage to the brain, for example,” said Dr. Malone. “When we do MRIs, they often come out normal.”
It’s possible that the virus lingers in some patients, said Rajeev Fernando, MD, an infectious disease specialist and a fellow at Harvard Medical School, Boston. Kids’ strong immune systems often fend off problems that can be noticed. But on the inside, dead fragments of the virus persist, floating in hidden parts of the body and activating the immune system long after the threat has passed.
The virus can be in the gut and in the brain, which may help explain why symptoms like brain fog and nausea can linger in children.
“The immune system doesn’t recognize whether fragments of the virus are dead or alive. It continues to think it’s fighting active COVID,” said Dr. Fernando.
There is little data on how long symptoms last, Dr. Fernando said, as well as how many kids get them and why some are more vulnerable than others. Some research has found that about 5%-15% of children with COVID may get long COVID, but the statistics vary globally.
“Children with long COVID have largely been ignored. And while we’re talking about it now, we’ve got some work to do,” said Dr. Fernando.
As for Emma, she recovered in January of 2021, heading back to school and her friends, although her cardiologist advised her to skip gym classes.
“For the first time in months, I was feeling like myself again,” she said.
But the coronavirus found its way to Emma again. Although she was fully vaccinated in the fall of 2021, when the Omicron variant swept the world late that year, she was infected again.
“When the wave of Omicron descended, Emma was like a sitting duck,” her mother said.
She was bedridden with a high fever and cough. The cold-like symptoms eventually went away, but the issues in her gut stuck around. Since then, Emma has had extreme nausea, losing most of the weight she had gained back.
For her part, Ms. Sherman has found solace in a group called Long COVID Kids, a nonprofit in Europe and the United States. The group is raising awareness about the condition in kids to increase funding, boost understanding, and improve treatment and outcomes.
“There’s nothing worse than watching your child suffer and not being able to do anything about it,” she said. “I tell Emma all the time: If I could just crawl in your body and take it, I would do it in a second.”
Emma is hoping for a fresh start with her family’s move in the coming weeks to Sotogrande in southern Spain.
“I miss the simplest things like going for a run, going to the fair with my friends, and just feeling well,” she said. “I have a long list of things I’ll do once this is all done.”
A version of this article first appeared on WebMD.com.
Emma Sherman, a 13-year-old girl in Ascot, England, woke up to a dizzying aura of blind spots and flashing lights in her field of vision. It was May 2020, and she also had crippling nausea and headaches. By August, her dizziness was so overwhelming, she couldn’t hold her head up, lying in her mother’s lap for hours, too fatigued to attend school.
The former competitive gymnast, who had hoped to try out for the cheerleading squad, now used a wheelchair and was a shadow of her former self. She had been diagnosed with COVID-induced postural orthostatic tachycardia syndrome, a condition often caused by an infection that results in a higher heart rate, extreme nausea, dizziness, and fatigue.
“I was so into sports before I got long COVID, and afterwards I could barely walk,” Emma said.
Even minor movements sent her heart rate sky-high. Her long chestnut hair turned gray and fell out in clumps. In the hospital, she was pricked and prodded, her blood tested for numerous conditions.
“They ran every scan known to man and took an MRI of her brain,” said Emma’s mother, Marie Sherman. “All was clear.”
Emma’s pediatrician determined that the teen had long COVID after having had a mild case of the virus in March, about 2 months before her puzzling symptoms began. But beyond a positive antibody test, doctors have found little evidence of what was causing Emma’s symptoms.
For Emma and others with long COVID, there are no medications shown to directly target the condition. Instead, caregivers target their symptoms, which include nausea, dizziness, fatigue, headaches, and a racing heart, said Laura Malone, MD, codirector of the Johns Hopkins Kennedy Krieger Pediatric Post–COVID-19 Rehabilitation Clinic in Baltimore.
“Right now, it’s a rehabilitation-based approach focused on improving symptoms and functioning so that kids can go back to their usual activities as much as possible,” she says.
Depression and anxiety are common, although doctors are struggling to figure out whether COVID is changing the brain or whether mental health symptoms result from all the life disruptions. There’s little research to show how may kids have depression because of long COVID. Dr. Malone said about half of her patients at the Kennedy Krieger Institute›s long COVID clinic are also dealing with mental health issues.
Patients with headaches, dizziness, and nausea are given pain and nausea medications and recommendations for a healthy diet with added fruits and vegetables, monounsaturated fats, lower sodium, unprocessed foods, and whole grains. Kids with irregular or racing heart rates are referred to cardiologists and potentially prescribed beta-blockers to treat their heart arrhythmias, while children with breathing problems may be referred to pulmonologists and those with depression to a psychiatrist.
Still, many patients like Emma go to their doctors with phantom symptoms that don’t show up on scans or blood tests.
“We’re not seeing any evidence of structural damage to the brain, for example,” said Dr. Malone. “When we do MRIs, they often come out normal.”
It’s possible that the virus lingers in some patients, said Rajeev Fernando, MD, an infectious disease specialist and a fellow at Harvard Medical School, Boston. Kids’ strong immune systems often fend off problems that can be noticed. But on the inside, dead fragments of the virus persist, floating in hidden parts of the body and activating the immune system long after the threat has passed.
The virus can be in the gut and in the brain, which may help explain why symptoms like brain fog and nausea can linger in children.
“The immune system doesn’t recognize whether fragments of the virus are dead or alive. It continues to think it’s fighting active COVID,” said Dr. Fernando.
There is little data on how long symptoms last, Dr. Fernando said, as well as how many kids get them and why some are more vulnerable than others. Some research has found that about 5%-15% of children with COVID may get long COVID, but the statistics vary globally.
“Children with long COVID have largely been ignored. And while we’re talking about it now, we’ve got some work to do,” said Dr. Fernando.
As for Emma, she recovered in January of 2021, heading back to school and her friends, although her cardiologist advised her to skip gym classes.
“For the first time in months, I was feeling like myself again,” she said.
But the coronavirus found its way to Emma again. Although she was fully vaccinated in the fall of 2021, when the Omicron variant swept the world late that year, she was infected again.
“When the wave of Omicron descended, Emma was like a sitting duck,” her mother said.
She was bedridden with a high fever and cough. The cold-like symptoms eventually went away, but the issues in her gut stuck around. Since then, Emma has had extreme nausea, losing most of the weight she had gained back.
For her part, Ms. Sherman has found solace in a group called Long COVID Kids, a nonprofit in Europe and the United States. The group is raising awareness about the condition in kids to increase funding, boost understanding, and improve treatment and outcomes.
“There’s nothing worse than watching your child suffer and not being able to do anything about it,” she said. “I tell Emma all the time: If I could just crawl in your body and take it, I would do it in a second.”
Emma is hoping for a fresh start with her family’s move in the coming weeks to Sotogrande in southern Spain.
“I miss the simplest things like going for a run, going to the fair with my friends, and just feeling well,” she said. “I have a long list of things I’ll do once this is all done.”
A version of this article first appeared on WebMD.com.
Topical gene therapy for dystrophic epidermolysis bullosa shows promise
INDIANAPOLIS – An investigational compared with placebo, according to results from a small phase 3 study.
DEB is a serious, ultra-rare genetic blistering disease caused by mutations in the COL7A1 gene, encoding for type VII collagen and leading to skin fragility and wounds. No approved therapies are currently available. In the study, treatment was generally well tolerated.
“B-VEC is the first treatment that has not only been shown to be effective, but the first to directly target the defect through topical application,” the study’s principal investigator, Shireen V. Guide, MD, said in an interview during a poster session at the annual meeting of the Society for Pediatric Dermatology. “It delivers type VII collagen gene therapy to these patients, which allows healing in areas that they may have had open since birth. It’s been life-changing for them.”
B-VEC is a herpes simplex virus (HSV-1)-based topical, redosable gene therapy being developed by Krystal Biotech that is designed to restore functional COL7 protein by delivering the COL7A1 gene. For the phase 3, multicenter, double-blind, placebo-controlled study known GEM-3, Dr. Guide, who practices dermatology in Rancho Santa Margarita, Calif., and her colleagues, including Peter Marinkovich, MD, from Stanford (Calif.) University, and Mercedes Gonzalez, MD, from the University of Miami, enrolled 31 patients aged 6 months and older with genetically confirmed DEB. Each patient had one wound treated randomized 1:1 to treatment with B-VEC once a week or placebo for 6 months. The mean age of the 31 study participants was 17 years, 65% were male, 65% were White, and 19% were Asian.
The primary endpoint was complete wound healing (defined as 100% wound closure from exact wound area at baseline, specified as skin re-epithelialization without drainage) at 6 months. Additional endpoints included complete wound healing at 3 months and change in pain associated with wound dressing changes.
At 3 months, 70% of wounds treated with B-VEC met the endpoint of complete wound healing, compared with 20% of wounds treated with placebo (P < .005). At 6 months, 67% of wounds treated with B-VEC met the endpoint of complete wound healing compared with 22% of those treated with placebo (P < .005).
Of the total wounds that closed at 3 months, 67% of wounds treated with B-VEC were also closed at 6 months, compared with 33% of those treated with placebo (P = .02). In other findings, a trend toward decreased pain was observed in wounds treated with B-VEC vs. those treated with placebo.
B-VEC was well tolerated with no treatment-related serious adverse events or discontinuations. Three patients experienced a total of five serious adverse events during the study: anemia (two events), and cellulitis, diarrhea, and positive blood culture (one event each). None were considered related to the study drug.
Dr. Guide, who is on staff at Children’s Health of Orange County, Orange, Calif., characterized B-VEC as “very novel because it’s very practical.”
To date, all treatments for DEB “have been extremely labor intensive, including skin grafting and hospitalizations. It’s a topical application that can be done in the office and potentially applied at home in the future. It’s also durable. Not only are the [treated] areas closing, but they are staying closed.”
Kalyani S. Marathe, MD, MPH, director of the dermatology division at Cincinnati Children’s Hospital, who was asked to comment on the study, said that topical application of B-VEC “allows the side effect profile to be very favorable. The results are remarkable in the amount of wound healing and reduction in pain.”
The tolerability of this medication “is crucial,” she added. “EB patients have a lot of pain from their wounds and so any treatment needs to be as painless as possible for it to be usable. I’m very excited about the next phase of studies for this medication and hopeful that it heralds new treatments for our EB patients.”
In June 2022, the manufacturer announced that it had submitted a biologics license application to the Food and Drug Administration for approval of B-VEC for the treatment of DEB, and that it anticipates submitting an application for marketing authorization with the European Medical Agency (EMA) in the second half of 2022.
Dr. Guide disclosed that she has served as an investigator for Krystal Biotech, Innovaderm Research, Arcutis, Premier Research, Paidion, and Castle Biosciences. Dr. Marathe disclosed that she has served as an adviser for Verrica, and that Cincinnati Children’s Hospital is a site for the next phase studies for B-VEC.
*This story was updated on July 25.
INDIANAPOLIS – An investigational compared with placebo, according to results from a small phase 3 study.
DEB is a serious, ultra-rare genetic blistering disease caused by mutations in the COL7A1 gene, encoding for type VII collagen and leading to skin fragility and wounds. No approved therapies are currently available. In the study, treatment was generally well tolerated.
“B-VEC is the first treatment that has not only been shown to be effective, but the first to directly target the defect through topical application,” the study’s principal investigator, Shireen V. Guide, MD, said in an interview during a poster session at the annual meeting of the Society for Pediatric Dermatology. “It delivers type VII collagen gene therapy to these patients, which allows healing in areas that they may have had open since birth. It’s been life-changing for them.”
B-VEC is a herpes simplex virus (HSV-1)-based topical, redosable gene therapy being developed by Krystal Biotech that is designed to restore functional COL7 protein by delivering the COL7A1 gene. For the phase 3, multicenter, double-blind, placebo-controlled study known GEM-3, Dr. Guide, who practices dermatology in Rancho Santa Margarita, Calif., and her colleagues, including Peter Marinkovich, MD, from Stanford (Calif.) University, and Mercedes Gonzalez, MD, from the University of Miami, enrolled 31 patients aged 6 months and older with genetically confirmed DEB. Each patient had one wound treated randomized 1:1 to treatment with B-VEC once a week or placebo for 6 months. The mean age of the 31 study participants was 17 years, 65% were male, 65% were White, and 19% were Asian.
The primary endpoint was complete wound healing (defined as 100% wound closure from exact wound area at baseline, specified as skin re-epithelialization without drainage) at 6 months. Additional endpoints included complete wound healing at 3 months and change in pain associated with wound dressing changes.
At 3 months, 70% of wounds treated with B-VEC met the endpoint of complete wound healing, compared with 20% of wounds treated with placebo (P < .005). At 6 months, 67% of wounds treated with B-VEC met the endpoint of complete wound healing compared with 22% of those treated with placebo (P < .005).
Of the total wounds that closed at 3 months, 67% of wounds treated with B-VEC were also closed at 6 months, compared with 33% of those treated with placebo (P = .02). In other findings, a trend toward decreased pain was observed in wounds treated with B-VEC vs. those treated with placebo.
B-VEC was well tolerated with no treatment-related serious adverse events or discontinuations. Three patients experienced a total of five serious adverse events during the study: anemia (two events), and cellulitis, diarrhea, and positive blood culture (one event each). None were considered related to the study drug.
Dr. Guide, who is on staff at Children’s Health of Orange County, Orange, Calif., characterized B-VEC as “very novel because it’s very practical.”
To date, all treatments for DEB “have been extremely labor intensive, including skin grafting and hospitalizations. It’s a topical application that can be done in the office and potentially applied at home in the future. It’s also durable. Not only are the [treated] areas closing, but they are staying closed.”
Kalyani S. Marathe, MD, MPH, director of the dermatology division at Cincinnati Children’s Hospital, who was asked to comment on the study, said that topical application of B-VEC “allows the side effect profile to be very favorable. The results are remarkable in the amount of wound healing and reduction in pain.”
The tolerability of this medication “is crucial,” she added. “EB patients have a lot of pain from their wounds and so any treatment needs to be as painless as possible for it to be usable. I’m very excited about the next phase of studies for this medication and hopeful that it heralds new treatments for our EB patients.”
In June 2022, the manufacturer announced that it had submitted a biologics license application to the Food and Drug Administration for approval of B-VEC for the treatment of DEB, and that it anticipates submitting an application for marketing authorization with the European Medical Agency (EMA) in the second half of 2022.
Dr. Guide disclosed that she has served as an investigator for Krystal Biotech, Innovaderm Research, Arcutis, Premier Research, Paidion, and Castle Biosciences. Dr. Marathe disclosed that she has served as an adviser for Verrica, and that Cincinnati Children’s Hospital is a site for the next phase studies for B-VEC.
*This story was updated on July 25.
INDIANAPOLIS – An investigational compared with placebo, according to results from a small phase 3 study.
DEB is a serious, ultra-rare genetic blistering disease caused by mutations in the COL7A1 gene, encoding for type VII collagen and leading to skin fragility and wounds. No approved therapies are currently available. In the study, treatment was generally well tolerated.
“B-VEC is the first treatment that has not only been shown to be effective, but the first to directly target the defect through topical application,” the study’s principal investigator, Shireen V. Guide, MD, said in an interview during a poster session at the annual meeting of the Society for Pediatric Dermatology. “It delivers type VII collagen gene therapy to these patients, which allows healing in areas that they may have had open since birth. It’s been life-changing for them.”
B-VEC is a herpes simplex virus (HSV-1)-based topical, redosable gene therapy being developed by Krystal Biotech that is designed to restore functional COL7 protein by delivering the COL7A1 gene. For the phase 3, multicenter, double-blind, placebo-controlled study known GEM-3, Dr. Guide, who practices dermatology in Rancho Santa Margarita, Calif., and her colleagues, including Peter Marinkovich, MD, from Stanford (Calif.) University, and Mercedes Gonzalez, MD, from the University of Miami, enrolled 31 patients aged 6 months and older with genetically confirmed DEB. Each patient had one wound treated randomized 1:1 to treatment with B-VEC once a week or placebo for 6 months. The mean age of the 31 study participants was 17 years, 65% were male, 65% were White, and 19% were Asian.
The primary endpoint was complete wound healing (defined as 100% wound closure from exact wound area at baseline, specified as skin re-epithelialization without drainage) at 6 months. Additional endpoints included complete wound healing at 3 months and change in pain associated with wound dressing changes.
At 3 months, 70% of wounds treated with B-VEC met the endpoint of complete wound healing, compared with 20% of wounds treated with placebo (P < .005). At 6 months, 67% of wounds treated with B-VEC met the endpoint of complete wound healing compared with 22% of those treated with placebo (P < .005).
Of the total wounds that closed at 3 months, 67% of wounds treated with B-VEC were also closed at 6 months, compared with 33% of those treated with placebo (P = .02). In other findings, a trend toward decreased pain was observed in wounds treated with B-VEC vs. those treated with placebo.
B-VEC was well tolerated with no treatment-related serious adverse events or discontinuations. Three patients experienced a total of five serious adverse events during the study: anemia (two events), and cellulitis, diarrhea, and positive blood culture (one event each). None were considered related to the study drug.
Dr. Guide, who is on staff at Children’s Health of Orange County, Orange, Calif., characterized B-VEC as “very novel because it’s very practical.”
To date, all treatments for DEB “have been extremely labor intensive, including skin grafting and hospitalizations. It’s a topical application that can be done in the office and potentially applied at home in the future. It’s also durable. Not only are the [treated] areas closing, but they are staying closed.”
Kalyani S. Marathe, MD, MPH, director of the dermatology division at Cincinnati Children’s Hospital, who was asked to comment on the study, said that topical application of B-VEC “allows the side effect profile to be very favorable. The results are remarkable in the amount of wound healing and reduction in pain.”
The tolerability of this medication “is crucial,” she added. “EB patients have a lot of pain from their wounds and so any treatment needs to be as painless as possible for it to be usable. I’m very excited about the next phase of studies for this medication and hopeful that it heralds new treatments for our EB patients.”
In June 2022, the manufacturer announced that it had submitted a biologics license application to the Food and Drug Administration for approval of B-VEC for the treatment of DEB, and that it anticipates submitting an application for marketing authorization with the European Medical Agency (EMA) in the second half of 2022.
Dr. Guide disclosed that she has served as an investigator for Krystal Biotech, Innovaderm Research, Arcutis, Premier Research, Paidion, and Castle Biosciences. Dr. Marathe disclosed that she has served as an adviser for Verrica, and that Cincinnati Children’s Hospital is a site for the next phase studies for B-VEC.
*This story was updated on July 25.
AT SPD 2022
Cue new mothers: Breastfeed infants – but for how long?
How long should mothers breastfeed their babies?
The controversial question has cropped up again after the nation’s leading pediatrics group has issued new recommendations calling for women to breastfeed until their children turn 2, and possibly even longer.
The policy statement, Breastfeeding and the Use of Human Milk, was released on June 27 by the American Academy of Pediatrics. It calls out stigma, lack of support, and workplace barriers that make continued breastfeeding difficult for many mothers.
But the new policy statement isn’t going down smoothly with the Fed Is Best Foundation, a nonprofit group hoping to “debunk and sort out for the public” many of the proclamations in the AAP’s policies, said Christie del Castillo-Hegyi, MD, cofounder of the group and emergency physician at CHI St. Vincent, Little Rock, Ark.
The goal of Fed Is Best is to assist families and health care professionals with current research on the safe feeding of infants – whether with breast milk, formula, or a combination.
The AAP’s previous guidelines, issued in 2012, called for infants to be fed breast milk exclusively for their first 6 months. Continued breastfeeding was recommended while introducing complementary foods for a period of 1 year or longer, the policy stated. The updated policy extends the optimum time line for breastfeeding to up to 2 years, citing the health benefits for babies.
‘Tone deaf and one-sided’
The AAP policy is “tone deaf and one-sided to the 75% of the U.S. mothers who use formula either by necessity or choice,” Dr. del Castillo-Hegyi told this news organization.
She pointed to a long list of factors that could affect the health outcomes of infants with regard to breastfeeding versus formula-feeding. These include socioeconomic status, baseline maternal health and education, maternal genetics, and the effects of developing feeding complications from exclusive breastfeeding for infants whose mothers can’t produce enough milk. These issues can contribute to negative health outcomes and brain development in infants who go on to be formula fed, she said.
She also objected to the fact that the guidelines make little reference to a mother who needs to supplement breast milk with formula within 4 months – and even before that – to meet her infant’s nutritional requirements.
Mothers need to hear “that making sure their infant is adequately fed is the most important goal of any infant feeding recommendation,” Dr. del Castillo-Hegyi said. She noted that the AAP’s rigid guidelines may be impossible for many mothers to follow.
“The pressure to meet [the AAP’s] exceedingly high expectations is causing harm to mothers and babies,” she said, referring to earlier guidelines that contained similar suggestions.
If a mother’s milk is insufficient, babies are at risk for low growth rates, jaundice, and dehydration. Mothers also can be affected if they’re made to feel shame because they cannot provide adequate amounts of breast milk and must supplement their supply with formula.
The blanket nature of the AAPs recommendations is “irresponsible,” given the fact that only about one in four nursing people can produce sufficient breast milk to feed their baby, Dr. del Castillo-Hegyi said.
“Not only is there harm to the infant, who may suffer from developmental problems as a result of the malnutrition they experience, but it harms the mother who believes in the AAP to provide responsible guidelines that help them ensure the best nutrition to their infants,” she said.
Lori Feldman-Winter, MD, chair of the AAP Section on Breastfeeding, defended the updated guidance.
The policy aims “to clarify the evidence that breastfeeding matters and to use the best evidence to equip pediatricians with the ways they can support the mother’s choice,” Dr. Feldman-Winter said in an interview. “The bottom line is that most women can exclusively breastfeed according to our recommendation, but a growing number of women have conditions that make it difficult, such as obesity. Pediatricians are essential in recognizing suboptimal intake in the breastfed infant, and the policy delineates how to do this.”
Dr. Feldman-Winter added that the criticism of the policy “is not unexpected, given the many barriers in our society for women doing the work of mothering and trying to reach their personal breastfeeding goals. We know over 60% of mothers do not reach their intended goals. These barriers are even more apparent for the populations that are underserved and least likely to breastfeed.”
But Dr. del Castillo-Hegyi pushed back on the AAP’s claim that exclusive breastfeeding of infants up to 6 months of age confers significant benefits beyond combination breastfeeding and formula feeding. The policy “fails to address the fact that many mothers do not have the biological capacity to meet the recommendation and are simply unable to exclusively breastfeed their infants” for that length of time, she said.
While the differences of opinion might leave lactating mothers in limbo, another expert pointed out that “support” of mothers is critical.
Jessica Madden, MD, a pediatrician and lactation consultant in Cleveland, Ohio, said advocates should work to normalize extended breastfeeding in the general public.
“I think everyone should work to advocate together,” Dr. Madden said. “From the professional society standpoint, advocacy for extended breastfeeding should come from the Academy of Breastfeeding Medicine and the AAP’s Section on Breastfeeding Medicine.”
She said more emphasis should be focused on the roles that pediatricians and health care providers play, along with insurers and employers, to ensure that moms are confident and comfortable with whatever breastfeeding journey they take.
The AAP will be revisiting the recommendations again soon, Dr. Feldman-Winter said. The U.S. Preventive Services Task Force has completed a systematic review but has not set a date to release findings, she said.
Among the issues the USPSTF will address are whether interventions that support breastfeeding improve outcomes for children and mothers; how to improve the initiation, duration, intensity, and exclusivity of breastfeeding; and the identification of any potential harms of interventions that support breastfeeding.
“The research plan illustrates that breastfeeding is now an active area for research, and we will continue to update our recommendations according to the best evidence,” Dr. Feldman-Winter said.
Dr. del Castillo-Hegyi, Dr. Feldman-Winter, and Dr. Madden have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
How long should mothers breastfeed their babies?
The controversial question has cropped up again after the nation’s leading pediatrics group has issued new recommendations calling for women to breastfeed until their children turn 2, and possibly even longer.
The policy statement, Breastfeeding and the Use of Human Milk, was released on June 27 by the American Academy of Pediatrics. It calls out stigma, lack of support, and workplace barriers that make continued breastfeeding difficult for many mothers.
But the new policy statement isn’t going down smoothly with the Fed Is Best Foundation, a nonprofit group hoping to “debunk and sort out for the public” many of the proclamations in the AAP’s policies, said Christie del Castillo-Hegyi, MD, cofounder of the group and emergency physician at CHI St. Vincent, Little Rock, Ark.
The goal of Fed Is Best is to assist families and health care professionals with current research on the safe feeding of infants – whether with breast milk, formula, or a combination.
The AAP’s previous guidelines, issued in 2012, called for infants to be fed breast milk exclusively for their first 6 months. Continued breastfeeding was recommended while introducing complementary foods for a period of 1 year or longer, the policy stated. The updated policy extends the optimum time line for breastfeeding to up to 2 years, citing the health benefits for babies.
‘Tone deaf and one-sided’
The AAP policy is “tone deaf and one-sided to the 75% of the U.S. mothers who use formula either by necessity or choice,” Dr. del Castillo-Hegyi told this news organization.
She pointed to a long list of factors that could affect the health outcomes of infants with regard to breastfeeding versus formula-feeding. These include socioeconomic status, baseline maternal health and education, maternal genetics, and the effects of developing feeding complications from exclusive breastfeeding for infants whose mothers can’t produce enough milk. These issues can contribute to negative health outcomes and brain development in infants who go on to be formula fed, she said.
She also objected to the fact that the guidelines make little reference to a mother who needs to supplement breast milk with formula within 4 months – and even before that – to meet her infant’s nutritional requirements.
Mothers need to hear “that making sure their infant is adequately fed is the most important goal of any infant feeding recommendation,” Dr. del Castillo-Hegyi said. She noted that the AAP’s rigid guidelines may be impossible for many mothers to follow.
“The pressure to meet [the AAP’s] exceedingly high expectations is causing harm to mothers and babies,” she said, referring to earlier guidelines that contained similar suggestions.
If a mother’s milk is insufficient, babies are at risk for low growth rates, jaundice, and dehydration. Mothers also can be affected if they’re made to feel shame because they cannot provide adequate amounts of breast milk and must supplement their supply with formula.
The blanket nature of the AAPs recommendations is “irresponsible,” given the fact that only about one in four nursing people can produce sufficient breast milk to feed their baby, Dr. del Castillo-Hegyi said.
“Not only is there harm to the infant, who may suffer from developmental problems as a result of the malnutrition they experience, but it harms the mother who believes in the AAP to provide responsible guidelines that help them ensure the best nutrition to their infants,” she said.
Lori Feldman-Winter, MD, chair of the AAP Section on Breastfeeding, defended the updated guidance.
The policy aims “to clarify the evidence that breastfeeding matters and to use the best evidence to equip pediatricians with the ways they can support the mother’s choice,” Dr. Feldman-Winter said in an interview. “The bottom line is that most women can exclusively breastfeed according to our recommendation, but a growing number of women have conditions that make it difficult, such as obesity. Pediatricians are essential in recognizing suboptimal intake in the breastfed infant, and the policy delineates how to do this.”
Dr. Feldman-Winter added that the criticism of the policy “is not unexpected, given the many barriers in our society for women doing the work of mothering and trying to reach their personal breastfeeding goals. We know over 60% of mothers do not reach their intended goals. These barriers are even more apparent for the populations that are underserved and least likely to breastfeed.”
But Dr. del Castillo-Hegyi pushed back on the AAP’s claim that exclusive breastfeeding of infants up to 6 months of age confers significant benefits beyond combination breastfeeding and formula feeding. The policy “fails to address the fact that many mothers do not have the biological capacity to meet the recommendation and are simply unable to exclusively breastfeed their infants” for that length of time, she said.
While the differences of opinion might leave lactating mothers in limbo, another expert pointed out that “support” of mothers is critical.
Jessica Madden, MD, a pediatrician and lactation consultant in Cleveland, Ohio, said advocates should work to normalize extended breastfeeding in the general public.
“I think everyone should work to advocate together,” Dr. Madden said. “From the professional society standpoint, advocacy for extended breastfeeding should come from the Academy of Breastfeeding Medicine and the AAP’s Section on Breastfeeding Medicine.”
She said more emphasis should be focused on the roles that pediatricians and health care providers play, along with insurers and employers, to ensure that moms are confident and comfortable with whatever breastfeeding journey they take.
The AAP will be revisiting the recommendations again soon, Dr. Feldman-Winter said. The U.S. Preventive Services Task Force has completed a systematic review but has not set a date to release findings, she said.
Among the issues the USPSTF will address are whether interventions that support breastfeeding improve outcomes for children and mothers; how to improve the initiation, duration, intensity, and exclusivity of breastfeeding; and the identification of any potential harms of interventions that support breastfeeding.
“The research plan illustrates that breastfeeding is now an active area for research, and we will continue to update our recommendations according to the best evidence,” Dr. Feldman-Winter said.
Dr. del Castillo-Hegyi, Dr. Feldman-Winter, and Dr. Madden have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
How long should mothers breastfeed their babies?
The controversial question has cropped up again after the nation’s leading pediatrics group has issued new recommendations calling for women to breastfeed until their children turn 2, and possibly even longer.
The policy statement, Breastfeeding and the Use of Human Milk, was released on June 27 by the American Academy of Pediatrics. It calls out stigma, lack of support, and workplace barriers that make continued breastfeeding difficult for many mothers.
But the new policy statement isn’t going down smoothly with the Fed Is Best Foundation, a nonprofit group hoping to “debunk and sort out for the public” many of the proclamations in the AAP’s policies, said Christie del Castillo-Hegyi, MD, cofounder of the group and emergency physician at CHI St. Vincent, Little Rock, Ark.
The goal of Fed Is Best is to assist families and health care professionals with current research on the safe feeding of infants – whether with breast milk, formula, or a combination.
The AAP’s previous guidelines, issued in 2012, called for infants to be fed breast milk exclusively for their first 6 months. Continued breastfeeding was recommended while introducing complementary foods for a period of 1 year or longer, the policy stated. The updated policy extends the optimum time line for breastfeeding to up to 2 years, citing the health benefits for babies.
‘Tone deaf and one-sided’
The AAP policy is “tone deaf and one-sided to the 75% of the U.S. mothers who use formula either by necessity or choice,” Dr. del Castillo-Hegyi told this news organization.
She pointed to a long list of factors that could affect the health outcomes of infants with regard to breastfeeding versus formula-feeding. These include socioeconomic status, baseline maternal health and education, maternal genetics, and the effects of developing feeding complications from exclusive breastfeeding for infants whose mothers can’t produce enough milk. These issues can contribute to negative health outcomes and brain development in infants who go on to be formula fed, she said.
She also objected to the fact that the guidelines make little reference to a mother who needs to supplement breast milk with formula within 4 months – and even before that – to meet her infant’s nutritional requirements.
Mothers need to hear “that making sure their infant is adequately fed is the most important goal of any infant feeding recommendation,” Dr. del Castillo-Hegyi said. She noted that the AAP’s rigid guidelines may be impossible for many mothers to follow.
“The pressure to meet [the AAP’s] exceedingly high expectations is causing harm to mothers and babies,” she said, referring to earlier guidelines that contained similar suggestions.
If a mother’s milk is insufficient, babies are at risk for low growth rates, jaundice, and dehydration. Mothers also can be affected if they’re made to feel shame because they cannot provide adequate amounts of breast milk and must supplement their supply with formula.
The blanket nature of the AAPs recommendations is “irresponsible,” given the fact that only about one in four nursing people can produce sufficient breast milk to feed their baby, Dr. del Castillo-Hegyi said.
“Not only is there harm to the infant, who may suffer from developmental problems as a result of the malnutrition they experience, but it harms the mother who believes in the AAP to provide responsible guidelines that help them ensure the best nutrition to their infants,” she said.
Lori Feldman-Winter, MD, chair of the AAP Section on Breastfeeding, defended the updated guidance.
The policy aims “to clarify the evidence that breastfeeding matters and to use the best evidence to equip pediatricians with the ways they can support the mother’s choice,” Dr. Feldman-Winter said in an interview. “The bottom line is that most women can exclusively breastfeed according to our recommendation, but a growing number of women have conditions that make it difficult, such as obesity. Pediatricians are essential in recognizing suboptimal intake in the breastfed infant, and the policy delineates how to do this.”
Dr. Feldman-Winter added that the criticism of the policy “is not unexpected, given the many barriers in our society for women doing the work of mothering and trying to reach their personal breastfeeding goals. We know over 60% of mothers do not reach their intended goals. These barriers are even more apparent for the populations that are underserved and least likely to breastfeed.”
But Dr. del Castillo-Hegyi pushed back on the AAP’s claim that exclusive breastfeeding of infants up to 6 months of age confers significant benefits beyond combination breastfeeding and formula feeding. The policy “fails to address the fact that many mothers do not have the biological capacity to meet the recommendation and are simply unable to exclusively breastfeed their infants” for that length of time, she said.
While the differences of opinion might leave lactating mothers in limbo, another expert pointed out that “support” of mothers is critical.
Jessica Madden, MD, a pediatrician and lactation consultant in Cleveland, Ohio, said advocates should work to normalize extended breastfeeding in the general public.
“I think everyone should work to advocate together,” Dr. Madden said. “From the professional society standpoint, advocacy for extended breastfeeding should come from the Academy of Breastfeeding Medicine and the AAP’s Section on Breastfeeding Medicine.”
She said more emphasis should be focused on the roles that pediatricians and health care providers play, along with insurers and employers, to ensure that moms are confident and comfortable with whatever breastfeeding journey they take.
The AAP will be revisiting the recommendations again soon, Dr. Feldman-Winter said. The U.S. Preventive Services Task Force has completed a systematic review but has not set a date to release findings, she said.
Among the issues the USPSTF will address are whether interventions that support breastfeeding improve outcomes for children and mothers; how to improve the initiation, duration, intensity, and exclusivity of breastfeeding; and the identification of any potential harms of interventions that support breastfeeding.
“The research plan illustrates that breastfeeding is now an active area for research, and we will continue to update our recommendations according to the best evidence,” Dr. Feldman-Winter said.
Dr. del Castillo-Hegyi, Dr. Feldman-Winter, and Dr. Madden have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Moms’ cooing swapped with morphine for newborns in withdrawal
Four years ago, Atrium Health, in Charlotte, N.C., embarked on a dramatic change in how it cares for newborns exposed to opioids in the womb.
Until then, most of the 700 or so babies who underwent opioid withdrawal each year in the hospital system spent their first weeks in a neonatal intensive care unit (NICU), isolated from their parents and treated with regular doses of morphine to ease their symptoms.
Now, most babies stay in the hospital for just a few days under a new approach called Eat, Sleep, Console. These young patients stay in private rooms where they can bond with their parents and volunteer caregivers. The usual course of treatment is no longer extended therapy with opioid replacements. Instead, mothers are encouraged to stay overnight and are taught how to sooth their babies with swaddling, rocking, and cooing.
As a result, the average length of stay for newborns with neonatal abstinence syndrome (NAS) has dropped from 12 days to 6. Use of morphine has fallen by 79%, from 2.25 to 0.45 mg/kg per stay, according to results of a quality improvement pilot project at one of Atrium’s community hospitals.
Similar outcomes from other hospitals around the country have led to widespread uptake of Eat, Sleep, Console since its advent in 2017. That year, according to federal data, seven newborns were diagnosed with NAS for every 1,000 births.
Advocates say the family-centric model helps parents feel less stigmatized and more confident in their ability to care for their babies, who can have symptoms such as irritability and difficulty feeding for months.
The approach “really empowers families to do what they do best, which is take care of each other,” Douglas Dodds, MD, a pediatrician who led the effort at Atrium, told this news organization.
Questioning the old protocols
Numerous state perinatal collaboratives, hospital associations, and health systems say the program is the new standard of care for infants with NAS and neonatal opioid withdrawal syndrome (NOWS).
Twenty-six hospitals have adopted Eat, Sleep, Console as part of a clinical trial sponsored by the National Institutes of Health and a program called Advancing Clinical Trials in Neonatal Opioid Withdrawal Syndrome (ACT NOW). Researchers are comparing the approach to previous care protocols in regard to 12 outcomes, including time to medical readiness for discharge, frequency of opioid replacement therapy, and safety problems, such as seizures during treatment.
The transition has been swift. Less than a decade ago, most hospitals used the Finnegan Neonatal Abstinence Scoring System, which was developed in the 1970s to assess babies whose mothers had used heroin during pregnancy.
The Finnegan score entails monitoring babies every 3 hours for 21 symptoms, including high-pitched crying, sneezing, gastrointestinal problems, and yawning. If a baby scores an 8 or more three times in a row, most protocols using the traditional Finnegan approach recommend that providers move infants to an NICU, where they receive morphine or methadone. Once opioid replacement therapy is started, the protocols require a gradual weaning that lasts 3-4 weeks.
As the opioid epidemic grew and NICUs around the country began to fill with babies experiencing NAS or NOW, some clinicians began to question the Finnegan-driven approach.
“You have these miserable babies who are going through this really tough experience, and our first move is to separate them from their moms,” said Matthew Grossman, MD, a pediatric hospitalist at Yale New Haven Children’s Hospital, New Haven, Conn., who created Eat, Sleep, Console.
Dr. Grossman, associate professor and vice chair for quality in the department of pediatrics at Yale University, said he noticed that when mothers stayed overnight with their babies, the infants tended to have fewer withdrawal symptoms. Indeed, previous studies had demonstrated the benefits of breastfeeding and allowing mothers and babies to share a room.
“If you think of mom as a medicine, then you can’t put the baby in a unit where the mom can’t be there,” Dr. Grossman told this news organization. “It would be like taking a kid with pneumonia and putting him in a unit that doesn’t have antibiotics.”
Despite its prominence, the Finnegan score has never been validated for guiding the treatment of NAS. In addition, Finnegan scores can be inconsistent, and the assessment requires disturbing an infant to check signs such as its startle reflex, which, as Dr. Grossman and his fellow researchers pointed out, flies in the face of American Academy of Pediatrics’ recommendations to prioritize swaddling and minimize stimulation for infants with NAS.
By contrast, Eat, Sleep, Console offers a simplified assessment. Interventions are called for if a baby eats less than an ounce of food at a time/does not breastfeed, sleeps less than an hour at a stretch, or takes more than 10 minutes to be consoled. After nonpharmacologic interventions have been tried, doses of medication are used as needed. Babies who are doing well can be discharged in as few as 4 days.
Quashing bias against parents with substance abuse disorder
Even with the promise of shorter stays and better care, switching to nonpharmacologic care presents hurdles for hospitals. Among these is a lack of physical space for mothers to room with their babies in a quiet environment.
“In many community hospitals, the only place for infants to go is a neonatal intensive care unit, outside of the newborn nursery,” said Stephen Patrick, MD, MPH, associate professor and director of the Center for Child Health Policy at Vanderbilt University, Nashville, Tenn., who researches stigma associated with opioid use during pregnancy.
Administrators at SSM St. Mary’s Hospital in St. Louis initially balked at providing private rooms for mothers and their babies with NAS and NOWS, according to Kimberly Spence, MD, a neonatologist at SSM Health. She said the initial plan was to put the babies in a busy, brightly lit nursery.
But resistance waned as the hospital convinced health plans to pay for private rooms for the 5-7 days it typically takes a baby to go through withdrawal, said Dr. Spence, associate professor of pediatrics at Saint Louis University.
“We were able to provide enough data that this is evidence-based medicine and babies do better with their moms, and that ethically, this is the right thing to do, to reduce transfers to an NICU,” she said.
In addition, news stories about the family-centric approach and shorter stays for infants, along with SSM’s launch of an outpatient clinic to treat pregnant women with opioid use disorder, helped the system to attract more patients and increase its market share, said Dr. Spence.
Another challenge was getting physicians and nurses to set aside any judgments of parents with substance abuse disorder, according to Dr. Grossman and others.
“A lot of faculty and staff on the medical team didn’t feel like we should trust moms with their babies’ medical care” at SSM, Dr. Spence said.
Some hospitals conduct anti-bias training to teach providers that substance abuse is a disease that deserves proper medical treatment and not the moral failing of a patient. Such education may involve explaining that babies’ outcomes are improved when women undergo treatment with methadone or buprenorphine during pregnancy, even though use of those medications does pose a risk of NAS.
Creating a system that supports parents with substance abuse disorders may help to change perceptions. At Atrium Health, some staff members now enjoy working with these families because they can make a profound impact, Dr. Dodds said. He said they’ve learned that families suffering from substance abuse disorder “are not that different than any other family.”
Dr. Dodds, Dr. Patrick, Dr. Spence, and Dr. Grossman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Four years ago, Atrium Health, in Charlotte, N.C., embarked on a dramatic change in how it cares for newborns exposed to opioids in the womb.
Until then, most of the 700 or so babies who underwent opioid withdrawal each year in the hospital system spent their first weeks in a neonatal intensive care unit (NICU), isolated from their parents and treated with regular doses of morphine to ease their symptoms.
Now, most babies stay in the hospital for just a few days under a new approach called Eat, Sleep, Console. These young patients stay in private rooms where they can bond with their parents and volunteer caregivers. The usual course of treatment is no longer extended therapy with opioid replacements. Instead, mothers are encouraged to stay overnight and are taught how to sooth their babies with swaddling, rocking, and cooing.
As a result, the average length of stay for newborns with neonatal abstinence syndrome (NAS) has dropped from 12 days to 6. Use of morphine has fallen by 79%, from 2.25 to 0.45 mg/kg per stay, according to results of a quality improvement pilot project at one of Atrium’s community hospitals.
Similar outcomes from other hospitals around the country have led to widespread uptake of Eat, Sleep, Console since its advent in 2017. That year, according to federal data, seven newborns were diagnosed with NAS for every 1,000 births.
Advocates say the family-centric model helps parents feel less stigmatized and more confident in their ability to care for their babies, who can have symptoms such as irritability and difficulty feeding for months.
The approach “really empowers families to do what they do best, which is take care of each other,” Douglas Dodds, MD, a pediatrician who led the effort at Atrium, told this news organization.
Questioning the old protocols
Numerous state perinatal collaboratives, hospital associations, and health systems say the program is the new standard of care for infants with NAS and neonatal opioid withdrawal syndrome (NOWS).
Twenty-six hospitals have adopted Eat, Sleep, Console as part of a clinical trial sponsored by the National Institutes of Health and a program called Advancing Clinical Trials in Neonatal Opioid Withdrawal Syndrome (ACT NOW). Researchers are comparing the approach to previous care protocols in regard to 12 outcomes, including time to medical readiness for discharge, frequency of opioid replacement therapy, and safety problems, such as seizures during treatment.
The transition has been swift. Less than a decade ago, most hospitals used the Finnegan Neonatal Abstinence Scoring System, which was developed in the 1970s to assess babies whose mothers had used heroin during pregnancy.
The Finnegan score entails monitoring babies every 3 hours for 21 symptoms, including high-pitched crying, sneezing, gastrointestinal problems, and yawning. If a baby scores an 8 or more three times in a row, most protocols using the traditional Finnegan approach recommend that providers move infants to an NICU, where they receive morphine or methadone. Once opioid replacement therapy is started, the protocols require a gradual weaning that lasts 3-4 weeks.
As the opioid epidemic grew and NICUs around the country began to fill with babies experiencing NAS or NOW, some clinicians began to question the Finnegan-driven approach.
“You have these miserable babies who are going through this really tough experience, and our first move is to separate them from their moms,” said Matthew Grossman, MD, a pediatric hospitalist at Yale New Haven Children’s Hospital, New Haven, Conn., who created Eat, Sleep, Console.
Dr. Grossman, associate professor and vice chair for quality in the department of pediatrics at Yale University, said he noticed that when mothers stayed overnight with their babies, the infants tended to have fewer withdrawal symptoms. Indeed, previous studies had demonstrated the benefits of breastfeeding and allowing mothers and babies to share a room.
“If you think of mom as a medicine, then you can’t put the baby in a unit where the mom can’t be there,” Dr. Grossman told this news organization. “It would be like taking a kid with pneumonia and putting him in a unit that doesn’t have antibiotics.”
Despite its prominence, the Finnegan score has never been validated for guiding the treatment of NAS. In addition, Finnegan scores can be inconsistent, and the assessment requires disturbing an infant to check signs such as its startle reflex, which, as Dr. Grossman and his fellow researchers pointed out, flies in the face of American Academy of Pediatrics’ recommendations to prioritize swaddling and minimize stimulation for infants with NAS.
By contrast, Eat, Sleep, Console offers a simplified assessment. Interventions are called for if a baby eats less than an ounce of food at a time/does not breastfeed, sleeps less than an hour at a stretch, or takes more than 10 minutes to be consoled. After nonpharmacologic interventions have been tried, doses of medication are used as needed. Babies who are doing well can be discharged in as few as 4 days.
Quashing bias against parents with substance abuse disorder
Even with the promise of shorter stays and better care, switching to nonpharmacologic care presents hurdles for hospitals. Among these is a lack of physical space for mothers to room with their babies in a quiet environment.
“In many community hospitals, the only place for infants to go is a neonatal intensive care unit, outside of the newborn nursery,” said Stephen Patrick, MD, MPH, associate professor and director of the Center for Child Health Policy at Vanderbilt University, Nashville, Tenn., who researches stigma associated with opioid use during pregnancy.
Administrators at SSM St. Mary’s Hospital in St. Louis initially balked at providing private rooms for mothers and their babies with NAS and NOWS, according to Kimberly Spence, MD, a neonatologist at SSM Health. She said the initial plan was to put the babies in a busy, brightly lit nursery.
But resistance waned as the hospital convinced health plans to pay for private rooms for the 5-7 days it typically takes a baby to go through withdrawal, said Dr. Spence, associate professor of pediatrics at Saint Louis University.
“We were able to provide enough data that this is evidence-based medicine and babies do better with their moms, and that ethically, this is the right thing to do, to reduce transfers to an NICU,” she said.
In addition, news stories about the family-centric approach and shorter stays for infants, along with SSM’s launch of an outpatient clinic to treat pregnant women with opioid use disorder, helped the system to attract more patients and increase its market share, said Dr. Spence.
Another challenge was getting physicians and nurses to set aside any judgments of parents with substance abuse disorder, according to Dr. Grossman and others.
“A lot of faculty and staff on the medical team didn’t feel like we should trust moms with their babies’ medical care” at SSM, Dr. Spence said.
Some hospitals conduct anti-bias training to teach providers that substance abuse is a disease that deserves proper medical treatment and not the moral failing of a patient. Such education may involve explaining that babies’ outcomes are improved when women undergo treatment with methadone or buprenorphine during pregnancy, even though use of those medications does pose a risk of NAS.
Creating a system that supports parents with substance abuse disorders may help to change perceptions. At Atrium Health, some staff members now enjoy working with these families because they can make a profound impact, Dr. Dodds said. He said they’ve learned that families suffering from substance abuse disorder “are not that different than any other family.”
Dr. Dodds, Dr. Patrick, Dr. Spence, and Dr. Grossman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Four years ago, Atrium Health, in Charlotte, N.C., embarked on a dramatic change in how it cares for newborns exposed to opioids in the womb.
Until then, most of the 700 or so babies who underwent opioid withdrawal each year in the hospital system spent their first weeks in a neonatal intensive care unit (NICU), isolated from their parents and treated with regular doses of morphine to ease their symptoms.
Now, most babies stay in the hospital for just a few days under a new approach called Eat, Sleep, Console. These young patients stay in private rooms where they can bond with their parents and volunteer caregivers. The usual course of treatment is no longer extended therapy with opioid replacements. Instead, mothers are encouraged to stay overnight and are taught how to sooth their babies with swaddling, rocking, and cooing.
As a result, the average length of stay for newborns with neonatal abstinence syndrome (NAS) has dropped from 12 days to 6. Use of morphine has fallen by 79%, from 2.25 to 0.45 mg/kg per stay, according to results of a quality improvement pilot project at one of Atrium’s community hospitals.
Similar outcomes from other hospitals around the country have led to widespread uptake of Eat, Sleep, Console since its advent in 2017. That year, according to federal data, seven newborns were diagnosed with NAS for every 1,000 births.
Advocates say the family-centric model helps parents feel less stigmatized and more confident in their ability to care for their babies, who can have symptoms such as irritability and difficulty feeding for months.
The approach “really empowers families to do what they do best, which is take care of each other,” Douglas Dodds, MD, a pediatrician who led the effort at Atrium, told this news organization.
Questioning the old protocols
Numerous state perinatal collaboratives, hospital associations, and health systems say the program is the new standard of care for infants with NAS and neonatal opioid withdrawal syndrome (NOWS).
Twenty-six hospitals have adopted Eat, Sleep, Console as part of a clinical trial sponsored by the National Institutes of Health and a program called Advancing Clinical Trials in Neonatal Opioid Withdrawal Syndrome (ACT NOW). Researchers are comparing the approach to previous care protocols in regard to 12 outcomes, including time to medical readiness for discharge, frequency of opioid replacement therapy, and safety problems, such as seizures during treatment.
The transition has been swift. Less than a decade ago, most hospitals used the Finnegan Neonatal Abstinence Scoring System, which was developed in the 1970s to assess babies whose mothers had used heroin during pregnancy.
The Finnegan score entails monitoring babies every 3 hours for 21 symptoms, including high-pitched crying, sneezing, gastrointestinal problems, and yawning. If a baby scores an 8 or more three times in a row, most protocols using the traditional Finnegan approach recommend that providers move infants to an NICU, where they receive morphine or methadone. Once opioid replacement therapy is started, the protocols require a gradual weaning that lasts 3-4 weeks.
As the opioid epidemic grew and NICUs around the country began to fill with babies experiencing NAS or NOW, some clinicians began to question the Finnegan-driven approach.
“You have these miserable babies who are going through this really tough experience, and our first move is to separate them from their moms,” said Matthew Grossman, MD, a pediatric hospitalist at Yale New Haven Children’s Hospital, New Haven, Conn., who created Eat, Sleep, Console.
Dr. Grossman, associate professor and vice chair for quality in the department of pediatrics at Yale University, said he noticed that when mothers stayed overnight with their babies, the infants tended to have fewer withdrawal symptoms. Indeed, previous studies had demonstrated the benefits of breastfeeding and allowing mothers and babies to share a room.
“If you think of mom as a medicine, then you can’t put the baby in a unit where the mom can’t be there,” Dr. Grossman told this news organization. “It would be like taking a kid with pneumonia and putting him in a unit that doesn’t have antibiotics.”
Despite its prominence, the Finnegan score has never been validated for guiding the treatment of NAS. In addition, Finnegan scores can be inconsistent, and the assessment requires disturbing an infant to check signs such as its startle reflex, which, as Dr. Grossman and his fellow researchers pointed out, flies in the face of American Academy of Pediatrics’ recommendations to prioritize swaddling and minimize stimulation for infants with NAS.
By contrast, Eat, Sleep, Console offers a simplified assessment. Interventions are called for if a baby eats less than an ounce of food at a time/does not breastfeed, sleeps less than an hour at a stretch, or takes more than 10 minutes to be consoled. After nonpharmacologic interventions have been tried, doses of medication are used as needed. Babies who are doing well can be discharged in as few as 4 days.
Quashing bias against parents with substance abuse disorder
Even with the promise of shorter stays and better care, switching to nonpharmacologic care presents hurdles for hospitals. Among these is a lack of physical space for mothers to room with their babies in a quiet environment.
“In many community hospitals, the only place for infants to go is a neonatal intensive care unit, outside of the newborn nursery,” said Stephen Patrick, MD, MPH, associate professor and director of the Center for Child Health Policy at Vanderbilt University, Nashville, Tenn., who researches stigma associated with opioid use during pregnancy.
Administrators at SSM St. Mary’s Hospital in St. Louis initially balked at providing private rooms for mothers and their babies with NAS and NOWS, according to Kimberly Spence, MD, a neonatologist at SSM Health. She said the initial plan was to put the babies in a busy, brightly lit nursery.
But resistance waned as the hospital convinced health plans to pay for private rooms for the 5-7 days it typically takes a baby to go through withdrawal, said Dr. Spence, associate professor of pediatrics at Saint Louis University.
“We were able to provide enough data that this is evidence-based medicine and babies do better with their moms, and that ethically, this is the right thing to do, to reduce transfers to an NICU,” she said.
In addition, news stories about the family-centric approach and shorter stays for infants, along with SSM’s launch of an outpatient clinic to treat pregnant women with opioid use disorder, helped the system to attract more patients and increase its market share, said Dr. Spence.
Another challenge was getting physicians and nurses to set aside any judgments of parents with substance abuse disorder, according to Dr. Grossman and others.
“A lot of faculty and staff on the medical team didn’t feel like we should trust moms with their babies’ medical care” at SSM, Dr. Spence said.
Some hospitals conduct anti-bias training to teach providers that substance abuse is a disease that deserves proper medical treatment and not the moral failing of a patient. Such education may involve explaining that babies’ outcomes are improved when women undergo treatment with methadone or buprenorphine during pregnancy, even though use of those medications does pose a risk of NAS.
Creating a system that supports parents with substance abuse disorders may help to change perceptions. At Atrium Health, some staff members now enjoy working with these families because they can make a profound impact, Dr. Dodds said. He said they’ve learned that families suffering from substance abuse disorder “are not that different than any other family.”
Dr. Dodds, Dr. Patrick, Dr. Spence, and Dr. Grossman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Heart health poor for many U.S. children
U.S. children appear to be failing an important test – of their hearts, not minds.
New research from the Ann & Robert H. Lurie Children’s Hospital of Chicago shows that heart health is a concern for many long before adulthood because fewer than one-third of children aged 2-19 years scored highly on the American Heart Association’s checklist for ideal cardiovascular fitness.
“This study gives us a new baseline for children’s heart health in the United States,” said Amanda Perak, MD, pediatric cardiologist at Ann & Robert H. Lurie Children’s Hospital of Chicago and a coauthor of the study.
Dr. Perak and colleagues published their findings in the journal Circulation.
The researchers identified 9888 children who completed the Centers for Disease Control and Prevention’s National Health and Nutrition Examination Survey between 2013 and 2018. They analyzed the available data using the AHA’s Life’s Essential 8 – a 100-point assessment of eight predictors for measuring heart health, including sleep, nicotine exposure, and blood glucose.
Data for only three metrics were available for all children in the study: diet, physical activity, and body mass index. As children aged, more metrics were averaged to obtain the overall cardiovascular health score. For instance, cholesterol/lipid levels become available at age 6 years, and blood pressure can be measured starting at age 8 years.
Only 2.2% of children in the study had optimal heart health, according to the Life’s Essential 8 scoring system, which spans poor (0-49), moderate (50-79), and high (80-100). Fewer than one in three (29.1%) overall had high scores, and scores worsened with age.
In the 2- to 5-year age group, over half (56.5%) of the children had good heart health. However, only one-third (33.5%) of 6- to 11-year-olds scored highly. Meanwhile, only 14% of adolescents had good heart scores, Dr. Perak’s group found.
Heart health scores based on diet were lowest for every age group. In the youngest age group, the average cardiovascular health (CVH) score was about 61. In the 12- to 19-year age group, however, the average CVH score decreased to 28.5, the lowest measured score for any group in the study.
With such worrisome diet scores for the 12- to 19-year-old group, public health policies need to focus on changes, like removing sugar-sweetened beverage options from schools, according to Joseph Mahgerefteh, MD, director of preventive cardiology at the Mount Sinai Kravis Children’s Heart Center, New York. He added that parents and their children also have a role to play.
“Some of our teenagers forget they can drink water when they are thirsty, and it is not necessary to drink sugar-sweetened beverages for thirst,” Dr. Mahgerefteh, who was not involved in the study, said in an interview. “Fresh vegetable intake is so low to a degree that some of our patients refuse to have any type of vegetable in their diet.”
“As a physician community caring for these patients, we need to be much more aggressive with our counseling and referral of these patients,” added Barry Love, MD, director of the congenital cardiac catheterization program at the Mount Sinai Kravis Children’s Heart Center. “These youngsters will inevitably encounter the effect of these conditions – coronary artery disease and stroke – at a much earlier adult age.”
Dr. Perak, Dr. Mahgerefteh, and Dr. Love reported no relevant financial conflicts of interest.
A version of this article first appeared on Medscape.com.
U.S. children appear to be failing an important test – of their hearts, not minds.
New research from the Ann & Robert H. Lurie Children’s Hospital of Chicago shows that heart health is a concern for many long before adulthood because fewer than one-third of children aged 2-19 years scored highly on the American Heart Association’s checklist for ideal cardiovascular fitness.
“This study gives us a new baseline for children’s heart health in the United States,” said Amanda Perak, MD, pediatric cardiologist at Ann & Robert H. Lurie Children’s Hospital of Chicago and a coauthor of the study.
Dr. Perak and colleagues published their findings in the journal Circulation.
The researchers identified 9888 children who completed the Centers for Disease Control and Prevention’s National Health and Nutrition Examination Survey between 2013 and 2018. They analyzed the available data using the AHA’s Life’s Essential 8 – a 100-point assessment of eight predictors for measuring heart health, including sleep, nicotine exposure, and blood glucose.
Data for only three metrics were available for all children in the study: diet, physical activity, and body mass index. As children aged, more metrics were averaged to obtain the overall cardiovascular health score. For instance, cholesterol/lipid levels become available at age 6 years, and blood pressure can be measured starting at age 8 years.
Only 2.2% of children in the study had optimal heart health, according to the Life’s Essential 8 scoring system, which spans poor (0-49), moderate (50-79), and high (80-100). Fewer than one in three (29.1%) overall had high scores, and scores worsened with age.
In the 2- to 5-year age group, over half (56.5%) of the children had good heart health. However, only one-third (33.5%) of 6- to 11-year-olds scored highly. Meanwhile, only 14% of adolescents had good heart scores, Dr. Perak’s group found.
Heart health scores based on diet were lowest for every age group. In the youngest age group, the average cardiovascular health (CVH) score was about 61. In the 12- to 19-year age group, however, the average CVH score decreased to 28.5, the lowest measured score for any group in the study.
With such worrisome diet scores for the 12- to 19-year-old group, public health policies need to focus on changes, like removing sugar-sweetened beverage options from schools, according to Joseph Mahgerefteh, MD, director of preventive cardiology at the Mount Sinai Kravis Children’s Heart Center, New York. He added that parents and their children also have a role to play.
“Some of our teenagers forget they can drink water when they are thirsty, and it is not necessary to drink sugar-sweetened beverages for thirst,” Dr. Mahgerefteh, who was not involved in the study, said in an interview. “Fresh vegetable intake is so low to a degree that some of our patients refuse to have any type of vegetable in their diet.”
“As a physician community caring for these patients, we need to be much more aggressive with our counseling and referral of these patients,” added Barry Love, MD, director of the congenital cardiac catheterization program at the Mount Sinai Kravis Children’s Heart Center. “These youngsters will inevitably encounter the effect of these conditions – coronary artery disease and stroke – at a much earlier adult age.”
Dr. Perak, Dr. Mahgerefteh, and Dr. Love reported no relevant financial conflicts of interest.
A version of this article first appeared on Medscape.com.
U.S. children appear to be failing an important test – of their hearts, not minds.
New research from the Ann & Robert H. Lurie Children’s Hospital of Chicago shows that heart health is a concern for many long before adulthood because fewer than one-third of children aged 2-19 years scored highly on the American Heart Association’s checklist for ideal cardiovascular fitness.
“This study gives us a new baseline for children’s heart health in the United States,” said Amanda Perak, MD, pediatric cardiologist at Ann & Robert H. Lurie Children’s Hospital of Chicago and a coauthor of the study.
Dr. Perak and colleagues published their findings in the journal Circulation.
The researchers identified 9888 children who completed the Centers for Disease Control and Prevention’s National Health and Nutrition Examination Survey between 2013 and 2018. They analyzed the available data using the AHA’s Life’s Essential 8 – a 100-point assessment of eight predictors for measuring heart health, including sleep, nicotine exposure, and blood glucose.
Data for only three metrics were available for all children in the study: diet, physical activity, and body mass index. As children aged, more metrics were averaged to obtain the overall cardiovascular health score. For instance, cholesterol/lipid levels become available at age 6 years, and blood pressure can be measured starting at age 8 years.
Only 2.2% of children in the study had optimal heart health, according to the Life’s Essential 8 scoring system, which spans poor (0-49), moderate (50-79), and high (80-100). Fewer than one in three (29.1%) overall had high scores, and scores worsened with age.
In the 2- to 5-year age group, over half (56.5%) of the children had good heart health. However, only one-third (33.5%) of 6- to 11-year-olds scored highly. Meanwhile, only 14% of adolescents had good heart scores, Dr. Perak’s group found.
Heart health scores based on diet were lowest for every age group. In the youngest age group, the average cardiovascular health (CVH) score was about 61. In the 12- to 19-year age group, however, the average CVH score decreased to 28.5, the lowest measured score for any group in the study.
With such worrisome diet scores for the 12- to 19-year-old group, public health policies need to focus on changes, like removing sugar-sweetened beverage options from schools, according to Joseph Mahgerefteh, MD, director of preventive cardiology at the Mount Sinai Kravis Children’s Heart Center, New York. He added that parents and their children also have a role to play.
“Some of our teenagers forget they can drink water when they are thirsty, and it is not necessary to drink sugar-sweetened beverages for thirst,” Dr. Mahgerefteh, who was not involved in the study, said in an interview. “Fresh vegetable intake is so low to a degree that some of our patients refuse to have any type of vegetable in their diet.”
“As a physician community caring for these patients, we need to be much more aggressive with our counseling and referral of these patients,” added Barry Love, MD, director of the congenital cardiac catheterization program at the Mount Sinai Kravis Children’s Heart Center. “These youngsters will inevitably encounter the effect of these conditions – coronary artery disease and stroke – at a much earlier adult age.”
Dr. Perak, Dr. Mahgerefteh, and Dr. Love reported no relevant financial conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM CIRCULATION
One thing is certain, says survey: Doctors hate taxes
For the Medscape Physicians and Taxes Report 2022, physicians shared information about their tax debt as well as how they feel about the U.S. tax code, audits, and the prospects for the future.
Even though it may not always seem that way to physicians, their family tax bills – around $75,406 on average – are in line with the other top 10% of U.S. taxpayers, according to an examination of IRS data by the Tax Foundation. However, when it comes to local taxes, the Tax Foundation found that physicians pay more than average. (Forty-three states collect tax on individual incomes.)
The average physician’s family pays a 35% marginal tax rate, compared with the top marginal tax rate in the United States of 37%. (The marginal tax rate is the highest amount of tax charged on each additional dollar after the IRS bracket rates are applied to your income.)
According to Alexis Gallati, founder of Cerebral Tax Advisors, a Knoxville, Tenn.–based firm that caters to medical professionals, doctors also should pay attention to their effective tax rate, or the percentage of income they pay in taxes. It takes into account differing tax rates on ordinary income, capital gains, and other income sources, she says. “It gives a better 30,000-foot view of your tax situation.”
Some high-income families are required to pay the Alternative Minimum Tax (AMT), though in 2019 that applied to only one-tenth of U.S. households. The AMT is designed to make sure that high earners with many options for exemptions and deductions still contribute a minimum amount of tax. Only 13% of physicians surveyed said they paid the AMT, though 29% were unsure.
Filing taxes as painful as paying them
According to a 2021 Gallup poll, 50% of Americans think they pay too much tax. (About 44% think their tax bill is about right, and a kindhearted 4% think they pay too little.) Doctors are outliers on this one, with 75% saying they pay too much in taxes.
When asked what they would do to fix the tax system, the physicians in the Medscape survey had a wide array of proposed solutions, from “drop the corporate tax rate to nearly nothing to stimulate the economy” to “everyone should pay equitably. There are too many loopholes for the very wealthy.”
Some of the complaints were less about tax rates than the process of filing. One respondent said: “I would love for this system to not be our personal responsibility. Why should it be my duty to pay someone every year to do my taxes?”
About 48% of physicians prepare their own taxes (about the same percentage as the rest of the population), with most of those filing electronically, primarily because it saves time and the software is easy to use. Intuit TurboTax was the most popular online software, with 22% of respondents saying they currently used this product.
Of those who did pay someone to prepare their taxes, the complexity of their taxes cost them; the average respondent paid about three times the average rate for the service. In the long run, the cost might have been recouped.
Navjeet Chahal, managing partner and CEO of Chahal and Associates, a San Francisco–area firm specializing in working with physicians, points out that tax advisors don’t just fill out the forms; they proactively advise physicians about how they can limit their taxes. And indeed, most respondents feel that they got their money’s worth, with 70% saying their tax preparers charged a fair fee.
Though the physicians surveyed tended to think they pay too much tax, and several mentioned particular gripes with the system, the complexity of the tax code didn’t seem to be a big issue. While 82% of Americans polled in 2021 by Pew Research said they were bothered “a lot” or “some” by the complexity of the tax system, 68% of physicians agreed or slightly agreed that the U.S. tax system “makes sense.”
Gimme a break
Physicians are the beneficiaries of several types of tax breaks. Contributing to a pretax 401(k) account was the most common exemption, with 60% of physicians surveyed using this plan. Other tax breaks cited by respondents were: contributing to charity (54%), home mortgage interest (46%), and writing off business expenses (39%).
About one in five physicians has experienced an audit, but that risk has declined significantly in recent years, thanks to tighter IRS budgets. Overall, only about 1 in 167 U.S. taxpayers were audited in 2020, according to the IRS. Even for taxpayers reporting $5 million or more in income, the audit rate is only about 0.25%, the Government Accountability Office says.
The odds of a physician being summoned to a meeting with an auditor probably won’t increase for a few years, Mr. Gallati said. But the good news for doctors is bad news for lower-income Americans. “The IRS is woefully understaffed and underfunded, with the result that the agency is going for lower-hanging fruit and auditing more people in lower income brackets,” she said in an interview.
While one respondent described his experience with the IRS as “the audit from hell,” others thought it not so bad, with 72% saying the auditors treated them fairly. One respondent described the audit as “boring, short, and successful for me. The IRS owed me money.”
When it comes to taxes, physician respondents, on the whole, did not seem to be optimistic about the future. About 61% expect an increase in their tax rate because of Biden administration policies. One respondent veered into hyperbole with the comment: “I believe taxes will increase for physicians until they have no more money!”
Mr. Chahal doesn’t see it that way. He pointed out that recent attempts to raise taxes completely failed. “I personally don’t see that happening unless there’s a significant shift in the House and the Senate.”
A version of this article first appeared on Medscape.com.
For the Medscape Physicians and Taxes Report 2022, physicians shared information about their tax debt as well as how they feel about the U.S. tax code, audits, and the prospects for the future.
Even though it may not always seem that way to physicians, their family tax bills – around $75,406 on average – are in line with the other top 10% of U.S. taxpayers, according to an examination of IRS data by the Tax Foundation. However, when it comes to local taxes, the Tax Foundation found that physicians pay more than average. (Forty-three states collect tax on individual incomes.)
The average physician’s family pays a 35% marginal tax rate, compared with the top marginal tax rate in the United States of 37%. (The marginal tax rate is the highest amount of tax charged on each additional dollar after the IRS bracket rates are applied to your income.)
According to Alexis Gallati, founder of Cerebral Tax Advisors, a Knoxville, Tenn.–based firm that caters to medical professionals, doctors also should pay attention to their effective tax rate, or the percentage of income they pay in taxes. It takes into account differing tax rates on ordinary income, capital gains, and other income sources, she says. “It gives a better 30,000-foot view of your tax situation.”
Some high-income families are required to pay the Alternative Minimum Tax (AMT), though in 2019 that applied to only one-tenth of U.S. households. The AMT is designed to make sure that high earners with many options for exemptions and deductions still contribute a minimum amount of tax. Only 13% of physicians surveyed said they paid the AMT, though 29% were unsure.
Filing taxes as painful as paying them
According to a 2021 Gallup poll, 50% of Americans think they pay too much tax. (About 44% think their tax bill is about right, and a kindhearted 4% think they pay too little.) Doctors are outliers on this one, with 75% saying they pay too much in taxes.
When asked what they would do to fix the tax system, the physicians in the Medscape survey had a wide array of proposed solutions, from “drop the corporate tax rate to nearly nothing to stimulate the economy” to “everyone should pay equitably. There are too many loopholes for the very wealthy.”
Some of the complaints were less about tax rates than the process of filing. One respondent said: “I would love for this system to not be our personal responsibility. Why should it be my duty to pay someone every year to do my taxes?”
About 48% of physicians prepare their own taxes (about the same percentage as the rest of the population), with most of those filing electronically, primarily because it saves time and the software is easy to use. Intuit TurboTax was the most popular online software, with 22% of respondents saying they currently used this product.
Of those who did pay someone to prepare their taxes, the complexity of their taxes cost them; the average respondent paid about three times the average rate for the service. In the long run, the cost might have been recouped.
Navjeet Chahal, managing partner and CEO of Chahal and Associates, a San Francisco–area firm specializing in working with physicians, points out that tax advisors don’t just fill out the forms; they proactively advise physicians about how they can limit their taxes. And indeed, most respondents feel that they got their money’s worth, with 70% saying their tax preparers charged a fair fee.
Though the physicians surveyed tended to think they pay too much tax, and several mentioned particular gripes with the system, the complexity of the tax code didn’t seem to be a big issue. While 82% of Americans polled in 2021 by Pew Research said they were bothered “a lot” or “some” by the complexity of the tax system, 68% of physicians agreed or slightly agreed that the U.S. tax system “makes sense.”
Gimme a break
Physicians are the beneficiaries of several types of tax breaks. Contributing to a pretax 401(k) account was the most common exemption, with 60% of physicians surveyed using this plan. Other tax breaks cited by respondents were: contributing to charity (54%), home mortgage interest (46%), and writing off business expenses (39%).
About one in five physicians has experienced an audit, but that risk has declined significantly in recent years, thanks to tighter IRS budgets. Overall, only about 1 in 167 U.S. taxpayers were audited in 2020, according to the IRS. Even for taxpayers reporting $5 million or more in income, the audit rate is only about 0.25%, the Government Accountability Office says.
The odds of a physician being summoned to a meeting with an auditor probably won’t increase for a few years, Mr. Gallati said. But the good news for doctors is bad news for lower-income Americans. “The IRS is woefully understaffed and underfunded, with the result that the agency is going for lower-hanging fruit and auditing more people in lower income brackets,” she said in an interview.
While one respondent described his experience with the IRS as “the audit from hell,” others thought it not so bad, with 72% saying the auditors treated them fairly. One respondent described the audit as “boring, short, and successful for me. The IRS owed me money.”
When it comes to taxes, physician respondents, on the whole, did not seem to be optimistic about the future. About 61% expect an increase in their tax rate because of Biden administration policies. One respondent veered into hyperbole with the comment: “I believe taxes will increase for physicians until they have no more money!”
Mr. Chahal doesn’t see it that way. He pointed out that recent attempts to raise taxes completely failed. “I personally don’t see that happening unless there’s a significant shift in the House and the Senate.”
A version of this article first appeared on Medscape.com.
For the Medscape Physicians and Taxes Report 2022, physicians shared information about their tax debt as well as how they feel about the U.S. tax code, audits, and the prospects for the future.
Even though it may not always seem that way to physicians, their family tax bills – around $75,406 on average – are in line with the other top 10% of U.S. taxpayers, according to an examination of IRS data by the Tax Foundation. However, when it comes to local taxes, the Tax Foundation found that physicians pay more than average. (Forty-three states collect tax on individual incomes.)
The average physician’s family pays a 35% marginal tax rate, compared with the top marginal tax rate in the United States of 37%. (The marginal tax rate is the highest amount of tax charged on each additional dollar after the IRS bracket rates are applied to your income.)
According to Alexis Gallati, founder of Cerebral Tax Advisors, a Knoxville, Tenn.–based firm that caters to medical professionals, doctors also should pay attention to their effective tax rate, or the percentage of income they pay in taxes. It takes into account differing tax rates on ordinary income, capital gains, and other income sources, she says. “It gives a better 30,000-foot view of your tax situation.”
Some high-income families are required to pay the Alternative Minimum Tax (AMT), though in 2019 that applied to only one-tenth of U.S. households. The AMT is designed to make sure that high earners with many options for exemptions and deductions still contribute a minimum amount of tax. Only 13% of physicians surveyed said they paid the AMT, though 29% were unsure.
Filing taxes as painful as paying them
According to a 2021 Gallup poll, 50% of Americans think they pay too much tax. (About 44% think their tax bill is about right, and a kindhearted 4% think they pay too little.) Doctors are outliers on this one, with 75% saying they pay too much in taxes.
When asked what they would do to fix the tax system, the physicians in the Medscape survey had a wide array of proposed solutions, from “drop the corporate tax rate to nearly nothing to stimulate the economy” to “everyone should pay equitably. There are too many loopholes for the very wealthy.”
Some of the complaints were less about tax rates than the process of filing. One respondent said: “I would love for this system to not be our personal responsibility. Why should it be my duty to pay someone every year to do my taxes?”
About 48% of physicians prepare their own taxes (about the same percentage as the rest of the population), with most of those filing electronically, primarily because it saves time and the software is easy to use. Intuit TurboTax was the most popular online software, with 22% of respondents saying they currently used this product.
Of those who did pay someone to prepare their taxes, the complexity of their taxes cost them; the average respondent paid about three times the average rate for the service. In the long run, the cost might have been recouped.
Navjeet Chahal, managing partner and CEO of Chahal and Associates, a San Francisco–area firm specializing in working with physicians, points out that tax advisors don’t just fill out the forms; they proactively advise physicians about how they can limit their taxes. And indeed, most respondents feel that they got their money’s worth, with 70% saying their tax preparers charged a fair fee.
Though the physicians surveyed tended to think they pay too much tax, and several mentioned particular gripes with the system, the complexity of the tax code didn’t seem to be a big issue. While 82% of Americans polled in 2021 by Pew Research said they were bothered “a lot” or “some” by the complexity of the tax system, 68% of physicians agreed or slightly agreed that the U.S. tax system “makes sense.”
Gimme a break
Physicians are the beneficiaries of several types of tax breaks. Contributing to a pretax 401(k) account was the most common exemption, with 60% of physicians surveyed using this plan. Other tax breaks cited by respondents were: contributing to charity (54%), home mortgage interest (46%), and writing off business expenses (39%).
About one in five physicians has experienced an audit, but that risk has declined significantly in recent years, thanks to tighter IRS budgets. Overall, only about 1 in 167 U.S. taxpayers were audited in 2020, according to the IRS. Even for taxpayers reporting $5 million or more in income, the audit rate is only about 0.25%, the Government Accountability Office says.
The odds of a physician being summoned to a meeting with an auditor probably won’t increase for a few years, Mr. Gallati said. But the good news for doctors is bad news for lower-income Americans. “The IRS is woefully understaffed and underfunded, with the result that the agency is going for lower-hanging fruit and auditing more people in lower income brackets,” she said in an interview.
While one respondent described his experience with the IRS as “the audit from hell,” others thought it not so bad, with 72% saying the auditors treated them fairly. One respondent described the audit as “boring, short, and successful for me. The IRS owed me money.”
When it comes to taxes, physician respondents, on the whole, did not seem to be optimistic about the future. About 61% expect an increase in their tax rate because of Biden administration policies. One respondent veered into hyperbole with the comment: “I believe taxes will increase for physicians until they have no more money!”
Mr. Chahal doesn’t see it that way. He pointed out that recent attempts to raise taxes completely failed. “I personally don’t see that happening unless there’s a significant shift in the House and the Senate.”
A version of this article first appeared on Medscape.com.
Clinical characteristics of recurrent RIME elucidated in chart review
INDIANAPOLIS – , in a single-center retrospective study. In addition, 71% of patients with recurrent disease experienced 1-2 recurrences – episodes that were generally milder and occurred at variable intervals.
Those are among key findings from the study of 50 patients with RIME, presented by Catherina X. Pan at the annual meeting of the Society for Pediatric Dermatology.
Reactive infectious mucocutaneous eruption (RIME) is a novel term encompassing an array of rare, parainfectious mucositis diseases, noted Ms. Pan, a fourth-year medical student at Harvard Medical School, Boston. Previously known as Mycoplasma pneumoniae-induced rash and mucositis (MIRM), common clinical characteristics of RIME include less than 10% body surface area involvement of polymorphic skin lesions (vesiculobullous or targetoid macules/papules); erosive oral, genital, and/or ocular mucositis involving more than two sites, and evidence of prior infection including but not limited to upper respiratory infection, fever, and cough.
In addition to M. pneumoniae, other pathogens have been implicated, she said. “While the underlying etiology of the disease is not entirely clear, it’s become increasingly known that RIME tends to recur in a subset of patients.”
A cohort study of 13 patients with RIME found that Black race, male sex, and older age were predominant among the five patients who developed recurrent disease.
The estimated recurrence rate is between 8% and 38%, but the clinical characteristics of patients who develop recurrent RIME tend to be poorly understood, Ms. Pan said.
Along with her mentor, Sadaf Hussain, MD, of the department of dermatology at Boston Children’s Hospital, Ms. Pan conducted a retrospective chart review to characterize the clinical history and course of disease in patients diagnosed with recurrent RIME. They extracted data between January of 2000 and March of 2022 using ICD-10 codes used by board-certified dermatologists at Boston Children’s Hospital, as well as a text search for RIME or MIRM in the dermatology notes. Patients were included if they had a RIME/MIRM diagnosis by a board-certified dermatologist and/or infection on PCR/serology and mucositis involvement with limited skin involvement.
The study population included 50 patients: 24 with recurrent RIME and 26 with isolated RIME. The majority (66%) were male, and the mean age of RIME onset was between 11 and 12 years old, which is up to two years younger than previously reported in the case series of 13 patients. Most of the study participants (79%) were White, but there were no significant differences in patients who had recurrent RIME and those who had isolated RIME in terms of age, sex, or race.
Isolated vs. recurrent RIME
However, compared with patients who had isolated RIME, a greater proportion of those with recurrent RIME had a history of atopic disease (46% vs. 23%, respectively; P = .136), as well as a history of tonsillectomy and adenoidectomy (25% vs. 4%; P = .045). “This has not been previously observed, but it may generate a hypothesis that patients with a history of frequent infection as well as amplified immune responses may be associated with disease recurrence,” Ms. Pan said.
The average number of episodes among patients with recurrent RIME was 3.5 and the interval between episodes was variable, at a mean of 10.2 months. Ms. Pan reported that 71% of recurrent RIME patients experienced 1-2 episodes, although one patient experienced 9 episodes.
Clinically, episodes among all patients with RIME were characterized by infectious prodromal symptoms (69%), oral lesions (95%), ocular lesions (60%), genital lesions (41%) and cutaneous lesions (40%). However, RIME recurrences were less severe and more atypical, with 49% involving only one mucosal surface and 29% involving two mucosal surfaces. Also, except for oral lesions, rates of infectious prodromal symptoms and other lesions significantly decreased among recurrences compared with initial RIME.
“Notably, we found that M. pneumoniae was the most common known cause of RIME, particularly among the initial episodes,” Ms. Pan said. “However, 61% of recurrent RIME episodes did not have a known cause in terms of infectious etiology. And, concordant with prior studies, we also found decreased severity [of RIME recurrences] as indicated by decreased rates of emergency department presentation, hospitalization, and duration of hospitalization.”
In other findings, psychiatric complications such as anxiety and depression followed the onset of RIME in 33% of those with recurrent disease and 22% of those with isolated disease. In addition, the three most common treatments among all 50 patients were systemic steroids, topical steroids, and M. pneumoniae-specific antibiotics.
“While RIME is considered as typically milder than Stevens-Johnson syndrome and toxic epidermal necrolysis with low mortality rates, it can lead to severe complications including conjunctival shrinkage, corneal ulceration and scarring, blindness, and oral, ocular, urogenital synechiae,” Ms. Pan noted. “Increased use of corticosteroids and steroid-sparing agents such as IVIG have also been observed. Multidisciplinary care with ophthalmology, urology, and mental health services is critical.”
She acknowledged certain limitations of the study, including its retrospective, single-center design, and the possibility that milder cases may have been excluded due to a lack of accurate diagnosis or referral.
Carrie C. Coughlin, MD, who was asked to comment on the study results, pointed out that nearly half (24) of patients in the cohort experienced recurrent RIME. “This is a high proportion, suggesting counseling about the possibility of recurrence is more important than previously thought,” said Dr. Coughlin, director of the section of pediatric dermatology Washington University/St. Louis Children’s Hospital.
“Fortunately, recurrent cases tended to be less severe. However, many patients had more than one recurrence, making this challenging for affected patients.”
The researchers reported having no financial disclosures. Dr. Coughlin is on the board of the Pediatric Dermatology Research Alliance (PeDRA) and the International Immunosuppression and Transplant Skin Cancer Collaborative.
INDIANAPOLIS – , in a single-center retrospective study. In addition, 71% of patients with recurrent disease experienced 1-2 recurrences – episodes that were generally milder and occurred at variable intervals.
Those are among key findings from the study of 50 patients with RIME, presented by Catherina X. Pan at the annual meeting of the Society for Pediatric Dermatology.
Reactive infectious mucocutaneous eruption (RIME) is a novel term encompassing an array of rare, parainfectious mucositis diseases, noted Ms. Pan, a fourth-year medical student at Harvard Medical School, Boston. Previously known as Mycoplasma pneumoniae-induced rash and mucositis (MIRM), common clinical characteristics of RIME include less than 10% body surface area involvement of polymorphic skin lesions (vesiculobullous or targetoid macules/papules); erosive oral, genital, and/or ocular mucositis involving more than two sites, and evidence of prior infection including but not limited to upper respiratory infection, fever, and cough.
In addition to M. pneumoniae, other pathogens have been implicated, she said. “While the underlying etiology of the disease is not entirely clear, it’s become increasingly known that RIME tends to recur in a subset of patients.”
A cohort study of 13 patients with RIME found that Black race, male sex, and older age were predominant among the five patients who developed recurrent disease.
The estimated recurrence rate is between 8% and 38%, but the clinical characteristics of patients who develop recurrent RIME tend to be poorly understood, Ms. Pan said.
Along with her mentor, Sadaf Hussain, MD, of the department of dermatology at Boston Children’s Hospital, Ms. Pan conducted a retrospective chart review to characterize the clinical history and course of disease in patients diagnosed with recurrent RIME. They extracted data between January of 2000 and March of 2022 using ICD-10 codes used by board-certified dermatologists at Boston Children’s Hospital, as well as a text search for RIME or MIRM in the dermatology notes. Patients were included if they had a RIME/MIRM diagnosis by a board-certified dermatologist and/or infection on PCR/serology and mucositis involvement with limited skin involvement.
The study population included 50 patients: 24 with recurrent RIME and 26 with isolated RIME. The majority (66%) were male, and the mean age of RIME onset was between 11 and 12 years old, which is up to two years younger than previously reported in the case series of 13 patients. Most of the study participants (79%) were White, but there were no significant differences in patients who had recurrent RIME and those who had isolated RIME in terms of age, sex, or race.
Isolated vs. recurrent RIME
However, compared with patients who had isolated RIME, a greater proportion of those with recurrent RIME had a history of atopic disease (46% vs. 23%, respectively; P = .136), as well as a history of tonsillectomy and adenoidectomy (25% vs. 4%; P = .045). “This has not been previously observed, but it may generate a hypothesis that patients with a history of frequent infection as well as amplified immune responses may be associated with disease recurrence,” Ms. Pan said.
The average number of episodes among patients with recurrent RIME was 3.5 and the interval between episodes was variable, at a mean of 10.2 months. Ms. Pan reported that 71% of recurrent RIME patients experienced 1-2 episodes, although one patient experienced 9 episodes.
Clinically, episodes among all patients with RIME were characterized by infectious prodromal symptoms (69%), oral lesions (95%), ocular lesions (60%), genital lesions (41%) and cutaneous lesions (40%). However, RIME recurrences were less severe and more atypical, with 49% involving only one mucosal surface and 29% involving two mucosal surfaces. Also, except for oral lesions, rates of infectious prodromal symptoms and other lesions significantly decreased among recurrences compared with initial RIME.
“Notably, we found that M. pneumoniae was the most common known cause of RIME, particularly among the initial episodes,” Ms. Pan said. “However, 61% of recurrent RIME episodes did not have a known cause in terms of infectious etiology. And, concordant with prior studies, we also found decreased severity [of RIME recurrences] as indicated by decreased rates of emergency department presentation, hospitalization, and duration of hospitalization.”
In other findings, psychiatric complications such as anxiety and depression followed the onset of RIME in 33% of those with recurrent disease and 22% of those with isolated disease. In addition, the three most common treatments among all 50 patients were systemic steroids, topical steroids, and M. pneumoniae-specific antibiotics.
“While RIME is considered as typically milder than Stevens-Johnson syndrome and toxic epidermal necrolysis with low mortality rates, it can lead to severe complications including conjunctival shrinkage, corneal ulceration and scarring, blindness, and oral, ocular, urogenital synechiae,” Ms. Pan noted. “Increased use of corticosteroids and steroid-sparing agents such as IVIG have also been observed. Multidisciplinary care with ophthalmology, urology, and mental health services is critical.”
She acknowledged certain limitations of the study, including its retrospective, single-center design, and the possibility that milder cases may have been excluded due to a lack of accurate diagnosis or referral.
Carrie C. Coughlin, MD, who was asked to comment on the study results, pointed out that nearly half (24) of patients in the cohort experienced recurrent RIME. “This is a high proportion, suggesting counseling about the possibility of recurrence is more important than previously thought,” said Dr. Coughlin, director of the section of pediatric dermatology Washington University/St. Louis Children’s Hospital.
“Fortunately, recurrent cases tended to be less severe. However, many patients had more than one recurrence, making this challenging for affected patients.”
The researchers reported having no financial disclosures. Dr. Coughlin is on the board of the Pediatric Dermatology Research Alliance (PeDRA) and the International Immunosuppression and Transplant Skin Cancer Collaborative.
INDIANAPOLIS – , in a single-center retrospective study. In addition, 71% of patients with recurrent disease experienced 1-2 recurrences – episodes that were generally milder and occurred at variable intervals.
Those are among key findings from the study of 50 patients with RIME, presented by Catherina X. Pan at the annual meeting of the Society for Pediatric Dermatology.
Reactive infectious mucocutaneous eruption (RIME) is a novel term encompassing an array of rare, parainfectious mucositis diseases, noted Ms. Pan, a fourth-year medical student at Harvard Medical School, Boston. Previously known as Mycoplasma pneumoniae-induced rash and mucositis (MIRM), common clinical characteristics of RIME include less than 10% body surface area involvement of polymorphic skin lesions (vesiculobullous or targetoid macules/papules); erosive oral, genital, and/or ocular mucositis involving more than two sites, and evidence of prior infection including but not limited to upper respiratory infection, fever, and cough.
In addition to M. pneumoniae, other pathogens have been implicated, she said. “While the underlying etiology of the disease is not entirely clear, it’s become increasingly known that RIME tends to recur in a subset of patients.”
A cohort study of 13 patients with RIME found that Black race, male sex, and older age were predominant among the five patients who developed recurrent disease.
The estimated recurrence rate is between 8% and 38%, but the clinical characteristics of patients who develop recurrent RIME tend to be poorly understood, Ms. Pan said.
Along with her mentor, Sadaf Hussain, MD, of the department of dermatology at Boston Children’s Hospital, Ms. Pan conducted a retrospective chart review to characterize the clinical history and course of disease in patients diagnosed with recurrent RIME. They extracted data between January of 2000 and March of 2022 using ICD-10 codes used by board-certified dermatologists at Boston Children’s Hospital, as well as a text search for RIME or MIRM in the dermatology notes. Patients were included if they had a RIME/MIRM diagnosis by a board-certified dermatologist and/or infection on PCR/serology and mucositis involvement with limited skin involvement.
The study population included 50 patients: 24 with recurrent RIME and 26 with isolated RIME. The majority (66%) were male, and the mean age of RIME onset was between 11 and 12 years old, which is up to two years younger than previously reported in the case series of 13 patients. Most of the study participants (79%) were White, but there were no significant differences in patients who had recurrent RIME and those who had isolated RIME in terms of age, sex, or race.
Isolated vs. recurrent RIME
However, compared with patients who had isolated RIME, a greater proportion of those with recurrent RIME had a history of atopic disease (46% vs. 23%, respectively; P = .136), as well as a history of tonsillectomy and adenoidectomy (25% vs. 4%; P = .045). “This has not been previously observed, but it may generate a hypothesis that patients with a history of frequent infection as well as amplified immune responses may be associated with disease recurrence,” Ms. Pan said.
The average number of episodes among patients with recurrent RIME was 3.5 and the interval between episodes was variable, at a mean of 10.2 months. Ms. Pan reported that 71% of recurrent RIME patients experienced 1-2 episodes, although one patient experienced 9 episodes.
Clinically, episodes among all patients with RIME were characterized by infectious prodromal symptoms (69%), oral lesions (95%), ocular lesions (60%), genital lesions (41%) and cutaneous lesions (40%). However, RIME recurrences were less severe and more atypical, with 49% involving only one mucosal surface and 29% involving two mucosal surfaces. Also, except for oral lesions, rates of infectious prodromal symptoms and other lesions significantly decreased among recurrences compared with initial RIME.
“Notably, we found that M. pneumoniae was the most common known cause of RIME, particularly among the initial episodes,” Ms. Pan said. “However, 61% of recurrent RIME episodes did not have a known cause in terms of infectious etiology. And, concordant with prior studies, we also found decreased severity [of RIME recurrences] as indicated by decreased rates of emergency department presentation, hospitalization, and duration of hospitalization.”
In other findings, psychiatric complications such as anxiety and depression followed the onset of RIME in 33% of those with recurrent disease and 22% of those with isolated disease. In addition, the three most common treatments among all 50 patients were systemic steroids, topical steroids, and M. pneumoniae-specific antibiotics.
“While RIME is considered as typically milder than Stevens-Johnson syndrome and toxic epidermal necrolysis with low mortality rates, it can lead to severe complications including conjunctival shrinkage, corneal ulceration and scarring, blindness, and oral, ocular, urogenital synechiae,” Ms. Pan noted. “Increased use of corticosteroids and steroid-sparing agents such as IVIG have also been observed. Multidisciplinary care with ophthalmology, urology, and mental health services is critical.”
She acknowledged certain limitations of the study, including its retrospective, single-center design, and the possibility that milder cases may have been excluded due to a lack of accurate diagnosis or referral.
Carrie C. Coughlin, MD, who was asked to comment on the study results, pointed out that nearly half (24) of patients in the cohort experienced recurrent RIME. “This is a high proportion, suggesting counseling about the possibility of recurrence is more important than previously thought,” said Dr. Coughlin, director of the section of pediatric dermatology Washington University/St. Louis Children’s Hospital.
“Fortunately, recurrent cases tended to be less severe. However, many patients had more than one recurrence, making this challenging for affected patients.”
The researchers reported having no financial disclosures. Dr. Coughlin is on the board of the Pediatric Dermatology Research Alliance (PeDRA) and the International Immunosuppression and Transplant Skin Cancer Collaborative.
AT SPD 2022
CAR T-cell therapy turns 10 and finally earns the word ‘cure’
Ten years ago, Stephan Grupp, MD, PhD, plunged into an unexplored area of pediatric cancer treatment with a 6-year-old patient for whom every treatment available for her acute lymphoblastic leukemia (ALL) had been exhausted.
Dr. Grupp, a pioneer in cellular immunotherapy at Children’s Hospital of Philadelphia, had just got the green light to launch the first phase 1 trial of chimeric antigen receptor (CAR) T-cell therapy for children.
“The trial opened at the absolute last possible moment that it could have been helpful to her,” he said in an interview. “There was nothing else to do to temporize her further. ... It had to open then or never.”
The patient was Emily Whitehead, who has since become a poster girl for the dramatic results that can be achieved with these novel therapies. After that one CAR T-cell treatment back in 2012, she has been free of her leukemia and has remained in remission for more than 10 years.
Dr. Grupp said that he is, at last, starting to use the “cure” word.
“I’m not just a doctor, I’m a scientist – and one case isn’t enough to have confidence about anything,” he said. “We wanted more patients to be out longer to be able to say that thing which we have for a long time called the ‘c word.’
“CAR T-cell therapy has now been given to hundreds of patients at CHOP, and – we are unique in this – we have a couple dozen patients who are 5, 6, 7, 9 years out or more without further therapy. That feels like a cure to me,” he commented.
First patient with ALL
Emily was the first patient with ALL to receive the novel treatment, and also the first child.
There was a precedent, however. After having been “stuck” for decades, the CAR T-cell field had recently made a breakthrough, thanks to research by Dr. Grupp’s colleague Carl June, MD, and associates at the University of Pennsylvania, Philadelphia. By tweaking two key steps in the genetic modification of T cells, Dr. June’s team had successfully treated three adults with chronic lymphocytic leukemia (CLL), two of whom were in complete remission.
But using the treatment for a child and for a different type of leukemia was a daunting prospect. Dr. Grupp said that he was candid with Emily’s parents, Tom and Kari Whitehead, emphasizing that there are no guarantees in cancer treatment, particularly in a phase 1 trial.
But the Whiteheads had no time to waste and nowhere else to turn. Her father, Tom, recalled saying: “This is something outside the box, this is going to give her a chance.”
Dr. Grupp, who described himself as being “on the cowboy end” of oncology care, was ready to take the plunge.
Little did any of them know that the treatment would make Emily even sicker than she already was, putting her in intensive care. But thanks to a combination of several lucky breaks and a lot of brain power, she would make a breathtakingly rapid recovery.
The ‘magic formula’
CAR T-cell therapy involves harvesting a patient’s T cells and modifying them in the lab with a chimeric antigen receptor to target CD19, a protein found on the surface of ALL cancer cells.
Before the University of Pennsylvania team tweaked the process, clinical trials of the therapy yielded only modest results because the modified T cells “were very powerful in the short term but had almost no proliferative capacity” once they were infused back into the patient, Dr. Grupp explained.
“It does not matter how many cells you give to a patient, what matters is that the cells grow in the patient to the level needed to control the leukemia,” he said.
Dr. June’s team came up with what Dr. Grupp calls “the magic formula”: A bead-based manufacturing process that produced younger T-cell phenotypes with “enormous” proliferative capacity, and a lentiviral approach to the genetic modification, enabling prolonged expression of the CAR-T molecule.
“Was it rogue? Absolutely, positively not,” said Dr. Grupp, thinking back to the day he enrolled Emily in the trial. “Was it risky? Obviously ... we all dived into this pool without knowing what was under the water, so I would say, rogue, no, risky, yes. And I would say we didn’t know nearly enough about the risks.”
Cytokine storm
The gravest risk that Dr. Grupp and his team encountered was something they had not anticipated. At the time, they had no name for it.
The three adults with CLL who had received CAR T-cell therapy had experienced a mild version that the researchers referred to as “tumor lysis syndrome”.
But for Emily, on day 3 of her CAR T-cell infusion, there was a ferocious reaction storm that later came to be called cytokine release syndrome.
“The wheels just came off then,” said Mr. Whitehead. “I remember her blood pressure was 53 over 29. They took her to the ICU, induced a coma, and put her on a ventilator. It was brutal to watch. The oscillatory ventilator just pounds on you, and there was blood bubbling out around the hose in her mouth.
“I remember the third or fourth night, a doctor took me in the hallway and said, ‘There’s a one-in-a-thousand chance your daughter is alive when the sun comes up,’” Mr. Whitehead said in an interview. “And I said: ‘All right, I’ll see you at rounds tomorrow, because she’ll still be here.’ ”
“We had some vague notion of toxicity ... but it turned out not nearly enough,” said Dr. Grupp. The ICU “worked flat out” to save her life. “They had deployed everything they had to keep a human being alive and they had nothing more to add. At some point, you run out of things that you can do, and we had run out.”
On the fly
It was then that the team ran into some good luck. The first break was when they decided to look at her cytokines. “Our whole knowledge base came together in the moment, on the fly, at the exact moment when Emily was so very sick,” he recalled. “Could we get the result fast enough? The lab dropped everything to run the test.”
They ordered a broad cytokine panel that included 30 analytes. The results showed that a number of cytokines “were just unbelievably elevated,” he said. Among them was interleukin-6.
“IL-6 isn’t even made by T cells, so nobody in the world would have guessed that this would have mattered. If we’d ordered a smaller panel, it might not even have been on it. Yet this was the one cytokine we had a drug for – tocilizumab – so that was chance. And then, another chance was that the drug was at the hospital, because there are rheumatology patients who get it.
“So, we went from making the determination that IL-6 was high and figuring out there was a drug for it at 3:00 o’clock to giving the drug to her at 8:00 o’clock, and then her clinical situation turned around so quickly – I mean hours later.”
Emily woke up from a 14-day medically induced coma on her seventh birthday.
Eight days later, her bone marrow showed complete remission. “The doctors said, ‘We’ve never seen anyone this sick get better any faster,’ ” Mr. Whitehead said.
She had already been through a battery of treatments for her leukemia. “It was 22 months of failed, standard treatment, and then just 23 days after they gave her the first dose of CAR T-cells that she was cancer free,” he added.
Talking about ‘cure’
Now that Emily, 17, has remained in remission for 10 years, Dr. Grupp is finally willing to use the word “cure” – but it has taken him a long time.
Now, he says, the challenge from the bedside is to keep parents’ and patients’ expectations realistic about what they see as a miracle cure.
“It’s not a miracle. We can get patients into remission 90-plus percent of the time – but some patients do relapse – and then there are the risks [of the cytokine storm, which can be life-threatening].
“Right now, our experience is that about 12% of patients end up in the ICU, but they hardly ever end up as sick as Emily ... because now we’re giving the tocilizumab much earlier,” Dr. Grupp said.
Hearing whispers
Since their daughter’s recovery, Tom and Kari Whitehead have dedicated much of their time to spreading the word about the treatment that saved Emily’s life. Mr. Whitehead testified at the Food and Drug Administration’s advisory committee meeting in 2017 when approval was being considered for the CAR T-cell product that Emily received. The product was tisagenlecleucel-T (Novartis); at that meeting, there was a unanimous vote to recommend approval. This was the first CAR T cell to reach the market.
As cofounders of the Emily Whitehead Foundation, Emily’s parents have helped raise more than $2 million to support research in the field, and they travel around the world telling their story to “move this revolution forward.”
Despite their fierce belief in the science that saved Emily, they also acknowledge there was luck – and faith. Early in their journey, when Emily experienced relapse after her initial treatments, Mr. Whitehead drew comfort from two visions, which he calls “whispers,” that guided them through several forks in the road and through tough decisions about Emily’s treatment.
Several times the parents refused treatment that was offered to Emily, and once they had her discharged against medical advice. “I told Kari she’s definitely going to beat her cancer – I saw it. I don’t know how it’s going to happen, but we’re going to be in the bone marrow transplant hallway [at CHOP] teaching her to walk again. I know a lot of doctors don’t want to hear anything about ‘a sign,’ or what guided us, but I don’t think you have to separate faith and science, I think it takes everything to make something like this to happen.”
Enduring effect
The key to the CAR T-cell breakthrough that gave rise to Emily’s therapy was cell proliferation, and the effect is enduring, beyond all expectations, said Dr. Grupp. The modified T cells are still detectable in Emily and other patients in long-term remission.
“The fundamental question is, are the cells still working, or are the patients cured and they don’t need them?” said Dr. Grupp. “I think it’s the latter. The data that we have from several large datasets that we developed with Novartis are that, if you get to a year and your minimal residual disease testing both by flow and by next-generation sequencing is negative and you still have B-cell aplasia, the relapse risk is close to zero at that point.”
While it’s still not clear if and when that risk will ever get to zero, Emily and Dr. Grupp have successfully closed the chapter.
“Oncologists have different notions of what the word ‘cure’ means. If your attitude is you’re not cured until you’ve basically reached the end of your life and you haven’t relapsed, well, that’s an impossible bar to hit. My attitude is, if your likelihood of having a disease recurrence is lower than the other risks in your life, like getting into your car and driving to your appointment, then that’s what a functional cure looks like,” he said.
“I’m probably the doctor that still sees her the most, but honestly, the whole conversation is not about leukemia at all. She has B-cell aplasia, so we have to treat that, and then it’s about making sure there’s no long-term side effects from the totality of her treatment. Generally, for a patient who’s gotten a moderate amount of chemotherapy and CAR T, that should not interfere with fertility. Has any patient in the history of the world ever relapsed more than 5 years out from their therapy? Of course. Is that incredibly rare? Yes, it is. You can be paralyzed by that, or you can compartmentalize it.”
As for the Whiteheads, they are focused on Emily’s college applications, her new driver’s license, and her project to cowrite a film about her story with a Hollywood filmmaker.
Mr. Whitehead said the one thing he hopes clinicians take away from their story is that sometimes a parent’s instinct transcends science.
A version of this article first appeared on Medscape.com.
Ten years ago, Stephan Grupp, MD, PhD, plunged into an unexplored area of pediatric cancer treatment with a 6-year-old patient for whom every treatment available for her acute lymphoblastic leukemia (ALL) had been exhausted.
Dr. Grupp, a pioneer in cellular immunotherapy at Children’s Hospital of Philadelphia, had just got the green light to launch the first phase 1 trial of chimeric antigen receptor (CAR) T-cell therapy for children.
“The trial opened at the absolute last possible moment that it could have been helpful to her,” he said in an interview. “There was nothing else to do to temporize her further. ... It had to open then or never.”
The patient was Emily Whitehead, who has since become a poster girl for the dramatic results that can be achieved with these novel therapies. After that one CAR T-cell treatment back in 2012, she has been free of her leukemia and has remained in remission for more than 10 years.
Dr. Grupp said that he is, at last, starting to use the “cure” word.
“I’m not just a doctor, I’m a scientist – and one case isn’t enough to have confidence about anything,” he said. “We wanted more patients to be out longer to be able to say that thing which we have for a long time called the ‘c word.’
“CAR T-cell therapy has now been given to hundreds of patients at CHOP, and – we are unique in this – we have a couple dozen patients who are 5, 6, 7, 9 years out or more without further therapy. That feels like a cure to me,” he commented.
First patient with ALL
Emily was the first patient with ALL to receive the novel treatment, and also the first child.
There was a precedent, however. After having been “stuck” for decades, the CAR T-cell field had recently made a breakthrough, thanks to research by Dr. Grupp’s colleague Carl June, MD, and associates at the University of Pennsylvania, Philadelphia. By tweaking two key steps in the genetic modification of T cells, Dr. June’s team had successfully treated three adults with chronic lymphocytic leukemia (CLL), two of whom were in complete remission.
But using the treatment for a child and for a different type of leukemia was a daunting prospect. Dr. Grupp said that he was candid with Emily’s parents, Tom and Kari Whitehead, emphasizing that there are no guarantees in cancer treatment, particularly in a phase 1 trial.
But the Whiteheads had no time to waste and nowhere else to turn. Her father, Tom, recalled saying: “This is something outside the box, this is going to give her a chance.”
Dr. Grupp, who described himself as being “on the cowboy end” of oncology care, was ready to take the plunge.
Little did any of them know that the treatment would make Emily even sicker than she already was, putting her in intensive care. But thanks to a combination of several lucky breaks and a lot of brain power, she would make a breathtakingly rapid recovery.
The ‘magic formula’
CAR T-cell therapy involves harvesting a patient’s T cells and modifying them in the lab with a chimeric antigen receptor to target CD19, a protein found on the surface of ALL cancer cells.
Before the University of Pennsylvania team tweaked the process, clinical trials of the therapy yielded only modest results because the modified T cells “were very powerful in the short term but had almost no proliferative capacity” once they were infused back into the patient, Dr. Grupp explained.
“It does not matter how many cells you give to a patient, what matters is that the cells grow in the patient to the level needed to control the leukemia,” he said.
Dr. June’s team came up with what Dr. Grupp calls “the magic formula”: A bead-based manufacturing process that produced younger T-cell phenotypes with “enormous” proliferative capacity, and a lentiviral approach to the genetic modification, enabling prolonged expression of the CAR-T molecule.
“Was it rogue? Absolutely, positively not,” said Dr. Grupp, thinking back to the day he enrolled Emily in the trial. “Was it risky? Obviously ... we all dived into this pool without knowing what was under the water, so I would say, rogue, no, risky, yes. And I would say we didn’t know nearly enough about the risks.”
Cytokine storm
The gravest risk that Dr. Grupp and his team encountered was something they had not anticipated. At the time, they had no name for it.
The three adults with CLL who had received CAR T-cell therapy had experienced a mild version that the researchers referred to as “tumor lysis syndrome”.
But for Emily, on day 3 of her CAR T-cell infusion, there was a ferocious reaction storm that later came to be called cytokine release syndrome.
“The wheels just came off then,” said Mr. Whitehead. “I remember her blood pressure was 53 over 29. They took her to the ICU, induced a coma, and put her on a ventilator. It was brutal to watch. The oscillatory ventilator just pounds on you, and there was blood bubbling out around the hose in her mouth.
“I remember the third or fourth night, a doctor took me in the hallway and said, ‘There’s a one-in-a-thousand chance your daughter is alive when the sun comes up,’” Mr. Whitehead said in an interview. “And I said: ‘All right, I’ll see you at rounds tomorrow, because she’ll still be here.’ ”
“We had some vague notion of toxicity ... but it turned out not nearly enough,” said Dr. Grupp. The ICU “worked flat out” to save her life. “They had deployed everything they had to keep a human being alive and they had nothing more to add. At some point, you run out of things that you can do, and we had run out.”
On the fly
It was then that the team ran into some good luck. The first break was when they decided to look at her cytokines. “Our whole knowledge base came together in the moment, on the fly, at the exact moment when Emily was so very sick,” he recalled. “Could we get the result fast enough? The lab dropped everything to run the test.”
They ordered a broad cytokine panel that included 30 analytes. The results showed that a number of cytokines “were just unbelievably elevated,” he said. Among them was interleukin-6.
“IL-6 isn’t even made by T cells, so nobody in the world would have guessed that this would have mattered. If we’d ordered a smaller panel, it might not even have been on it. Yet this was the one cytokine we had a drug for – tocilizumab – so that was chance. And then, another chance was that the drug was at the hospital, because there are rheumatology patients who get it.
“So, we went from making the determination that IL-6 was high and figuring out there was a drug for it at 3:00 o’clock to giving the drug to her at 8:00 o’clock, and then her clinical situation turned around so quickly – I mean hours later.”
Emily woke up from a 14-day medically induced coma on her seventh birthday.
Eight days later, her bone marrow showed complete remission. “The doctors said, ‘We’ve never seen anyone this sick get better any faster,’ ” Mr. Whitehead said.
She had already been through a battery of treatments for her leukemia. “It was 22 months of failed, standard treatment, and then just 23 days after they gave her the first dose of CAR T-cells that she was cancer free,” he added.
Talking about ‘cure’
Now that Emily, 17, has remained in remission for 10 years, Dr. Grupp is finally willing to use the word “cure” – but it has taken him a long time.
Now, he says, the challenge from the bedside is to keep parents’ and patients’ expectations realistic about what they see as a miracle cure.
“It’s not a miracle. We can get patients into remission 90-plus percent of the time – but some patients do relapse – and then there are the risks [of the cytokine storm, which can be life-threatening].
“Right now, our experience is that about 12% of patients end up in the ICU, but they hardly ever end up as sick as Emily ... because now we’re giving the tocilizumab much earlier,” Dr. Grupp said.
Hearing whispers
Since their daughter’s recovery, Tom and Kari Whitehead have dedicated much of their time to spreading the word about the treatment that saved Emily’s life. Mr. Whitehead testified at the Food and Drug Administration’s advisory committee meeting in 2017 when approval was being considered for the CAR T-cell product that Emily received. The product was tisagenlecleucel-T (Novartis); at that meeting, there was a unanimous vote to recommend approval. This was the first CAR T cell to reach the market.
As cofounders of the Emily Whitehead Foundation, Emily’s parents have helped raise more than $2 million to support research in the field, and they travel around the world telling their story to “move this revolution forward.”
Despite their fierce belief in the science that saved Emily, they also acknowledge there was luck – and faith. Early in their journey, when Emily experienced relapse after her initial treatments, Mr. Whitehead drew comfort from two visions, which he calls “whispers,” that guided them through several forks in the road and through tough decisions about Emily’s treatment.
Several times the parents refused treatment that was offered to Emily, and once they had her discharged against medical advice. “I told Kari she’s definitely going to beat her cancer – I saw it. I don’t know how it’s going to happen, but we’re going to be in the bone marrow transplant hallway [at CHOP] teaching her to walk again. I know a lot of doctors don’t want to hear anything about ‘a sign,’ or what guided us, but I don’t think you have to separate faith and science, I think it takes everything to make something like this to happen.”
Enduring effect
The key to the CAR T-cell breakthrough that gave rise to Emily’s therapy was cell proliferation, and the effect is enduring, beyond all expectations, said Dr. Grupp. The modified T cells are still detectable in Emily and other patients in long-term remission.
“The fundamental question is, are the cells still working, or are the patients cured and they don’t need them?” said Dr. Grupp. “I think it’s the latter. The data that we have from several large datasets that we developed with Novartis are that, if you get to a year and your minimal residual disease testing both by flow and by next-generation sequencing is negative and you still have B-cell aplasia, the relapse risk is close to zero at that point.”
While it’s still not clear if and when that risk will ever get to zero, Emily and Dr. Grupp have successfully closed the chapter.
“Oncologists have different notions of what the word ‘cure’ means. If your attitude is you’re not cured until you’ve basically reached the end of your life and you haven’t relapsed, well, that’s an impossible bar to hit. My attitude is, if your likelihood of having a disease recurrence is lower than the other risks in your life, like getting into your car and driving to your appointment, then that’s what a functional cure looks like,” he said.
“I’m probably the doctor that still sees her the most, but honestly, the whole conversation is not about leukemia at all. She has B-cell aplasia, so we have to treat that, and then it’s about making sure there’s no long-term side effects from the totality of her treatment. Generally, for a patient who’s gotten a moderate amount of chemotherapy and CAR T, that should not interfere with fertility. Has any patient in the history of the world ever relapsed more than 5 years out from their therapy? Of course. Is that incredibly rare? Yes, it is. You can be paralyzed by that, or you can compartmentalize it.”
As for the Whiteheads, they are focused on Emily’s college applications, her new driver’s license, and her project to cowrite a film about her story with a Hollywood filmmaker.
Mr. Whitehead said the one thing he hopes clinicians take away from their story is that sometimes a parent’s instinct transcends science.
A version of this article first appeared on Medscape.com.
Ten years ago, Stephan Grupp, MD, PhD, plunged into an unexplored area of pediatric cancer treatment with a 6-year-old patient for whom every treatment available for her acute lymphoblastic leukemia (ALL) had been exhausted.
Dr. Grupp, a pioneer in cellular immunotherapy at Children’s Hospital of Philadelphia, had just got the green light to launch the first phase 1 trial of chimeric antigen receptor (CAR) T-cell therapy for children.
“The trial opened at the absolute last possible moment that it could have been helpful to her,” he said in an interview. “There was nothing else to do to temporize her further. ... It had to open then or never.”
The patient was Emily Whitehead, who has since become a poster girl for the dramatic results that can be achieved with these novel therapies. After that one CAR T-cell treatment back in 2012, she has been free of her leukemia and has remained in remission for more than 10 years.
Dr. Grupp said that he is, at last, starting to use the “cure” word.
“I’m not just a doctor, I’m a scientist – and one case isn’t enough to have confidence about anything,” he said. “We wanted more patients to be out longer to be able to say that thing which we have for a long time called the ‘c word.’
“CAR T-cell therapy has now been given to hundreds of patients at CHOP, and – we are unique in this – we have a couple dozen patients who are 5, 6, 7, 9 years out or more without further therapy. That feels like a cure to me,” he commented.
First patient with ALL
Emily was the first patient with ALL to receive the novel treatment, and also the first child.
There was a precedent, however. After having been “stuck” for decades, the CAR T-cell field had recently made a breakthrough, thanks to research by Dr. Grupp’s colleague Carl June, MD, and associates at the University of Pennsylvania, Philadelphia. By tweaking two key steps in the genetic modification of T cells, Dr. June’s team had successfully treated three adults with chronic lymphocytic leukemia (CLL), two of whom were in complete remission.
But using the treatment for a child and for a different type of leukemia was a daunting prospect. Dr. Grupp said that he was candid with Emily’s parents, Tom and Kari Whitehead, emphasizing that there are no guarantees in cancer treatment, particularly in a phase 1 trial.
But the Whiteheads had no time to waste and nowhere else to turn. Her father, Tom, recalled saying: “This is something outside the box, this is going to give her a chance.”
Dr. Grupp, who described himself as being “on the cowboy end” of oncology care, was ready to take the plunge.
Little did any of them know that the treatment would make Emily even sicker than she already was, putting her in intensive care. But thanks to a combination of several lucky breaks and a lot of brain power, she would make a breathtakingly rapid recovery.
The ‘magic formula’
CAR T-cell therapy involves harvesting a patient’s T cells and modifying them in the lab with a chimeric antigen receptor to target CD19, a protein found on the surface of ALL cancer cells.
Before the University of Pennsylvania team tweaked the process, clinical trials of the therapy yielded only modest results because the modified T cells “were very powerful in the short term but had almost no proliferative capacity” once they were infused back into the patient, Dr. Grupp explained.
“It does not matter how many cells you give to a patient, what matters is that the cells grow in the patient to the level needed to control the leukemia,” he said.
Dr. June’s team came up with what Dr. Grupp calls “the magic formula”: A bead-based manufacturing process that produced younger T-cell phenotypes with “enormous” proliferative capacity, and a lentiviral approach to the genetic modification, enabling prolonged expression of the CAR-T molecule.
“Was it rogue? Absolutely, positively not,” said Dr. Grupp, thinking back to the day he enrolled Emily in the trial. “Was it risky? Obviously ... we all dived into this pool without knowing what was under the water, so I would say, rogue, no, risky, yes. And I would say we didn’t know nearly enough about the risks.”
Cytokine storm
The gravest risk that Dr. Grupp and his team encountered was something they had not anticipated. At the time, they had no name for it.
The three adults with CLL who had received CAR T-cell therapy had experienced a mild version that the researchers referred to as “tumor lysis syndrome”.
But for Emily, on day 3 of her CAR T-cell infusion, there was a ferocious reaction storm that later came to be called cytokine release syndrome.
“The wheels just came off then,” said Mr. Whitehead. “I remember her blood pressure was 53 over 29. They took her to the ICU, induced a coma, and put her on a ventilator. It was brutal to watch. The oscillatory ventilator just pounds on you, and there was blood bubbling out around the hose in her mouth.
“I remember the third or fourth night, a doctor took me in the hallway and said, ‘There’s a one-in-a-thousand chance your daughter is alive when the sun comes up,’” Mr. Whitehead said in an interview. “And I said: ‘All right, I’ll see you at rounds tomorrow, because she’ll still be here.’ ”
“We had some vague notion of toxicity ... but it turned out not nearly enough,” said Dr. Grupp. The ICU “worked flat out” to save her life. “They had deployed everything they had to keep a human being alive and they had nothing more to add. At some point, you run out of things that you can do, and we had run out.”
On the fly
It was then that the team ran into some good luck. The first break was when they decided to look at her cytokines. “Our whole knowledge base came together in the moment, on the fly, at the exact moment when Emily was so very sick,” he recalled. “Could we get the result fast enough? The lab dropped everything to run the test.”
They ordered a broad cytokine panel that included 30 analytes. The results showed that a number of cytokines “were just unbelievably elevated,” he said. Among them was interleukin-6.
“IL-6 isn’t even made by T cells, so nobody in the world would have guessed that this would have mattered. If we’d ordered a smaller panel, it might not even have been on it. Yet this was the one cytokine we had a drug for – tocilizumab – so that was chance. And then, another chance was that the drug was at the hospital, because there are rheumatology patients who get it.
“So, we went from making the determination that IL-6 was high and figuring out there was a drug for it at 3:00 o’clock to giving the drug to her at 8:00 o’clock, and then her clinical situation turned around so quickly – I mean hours later.”
Emily woke up from a 14-day medically induced coma on her seventh birthday.
Eight days later, her bone marrow showed complete remission. “The doctors said, ‘We’ve never seen anyone this sick get better any faster,’ ” Mr. Whitehead said.
She had already been through a battery of treatments for her leukemia. “It was 22 months of failed, standard treatment, and then just 23 days after they gave her the first dose of CAR T-cells that she was cancer free,” he added.
Talking about ‘cure’
Now that Emily, 17, has remained in remission for 10 years, Dr. Grupp is finally willing to use the word “cure” – but it has taken him a long time.
Now, he says, the challenge from the bedside is to keep parents’ and patients’ expectations realistic about what they see as a miracle cure.
“It’s not a miracle. We can get patients into remission 90-plus percent of the time – but some patients do relapse – and then there are the risks [of the cytokine storm, which can be life-threatening].
“Right now, our experience is that about 12% of patients end up in the ICU, but they hardly ever end up as sick as Emily ... because now we’re giving the tocilizumab much earlier,” Dr. Grupp said.
Hearing whispers
Since their daughter’s recovery, Tom and Kari Whitehead have dedicated much of their time to spreading the word about the treatment that saved Emily’s life. Mr. Whitehead testified at the Food and Drug Administration’s advisory committee meeting in 2017 when approval was being considered for the CAR T-cell product that Emily received. The product was tisagenlecleucel-T (Novartis); at that meeting, there was a unanimous vote to recommend approval. This was the first CAR T cell to reach the market.
As cofounders of the Emily Whitehead Foundation, Emily’s parents have helped raise more than $2 million to support research in the field, and they travel around the world telling their story to “move this revolution forward.”
Despite their fierce belief in the science that saved Emily, they also acknowledge there was luck – and faith. Early in their journey, when Emily experienced relapse after her initial treatments, Mr. Whitehead drew comfort from two visions, which he calls “whispers,” that guided them through several forks in the road and through tough decisions about Emily’s treatment.
Several times the parents refused treatment that was offered to Emily, and once they had her discharged against medical advice. “I told Kari she’s definitely going to beat her cancer – I saw it. I don’t know how it’s going to happen, but we’re going to be in the bone marrow transplant hallway [at CHOP] teaching her to walk again. I know a lot of doctors don’t want to hear anything about ‘a sign,’ or what guided us, but I don’t think you have to separate faith and science, I think it takes everything to make something like this to happen.”
Enduring effect
The key to the CAR T-cell breakthrough that gave rise to Emily’s therapy was cell proliferation, and the effect is enduring, beyond all expectations, said Dr. Grupp. The modified T cells are still detectable in Emily and other patients in long-term remission.
“The fundamental question is, are the cells still working, or are the patients cured and they don’t need them?” said Dr. Grupp. “I think it’s the latter. The data that we have from several large datasets that we developed with Novartis are that, if you get to a year and your minimal residual disease testing both by flow and by next-generation sequencing is negative and you still have B-cell aplasia, the relapse risk is close to zero at that point.”
While it’s still not clear if and when that risk will ever get to zero, Emily and Dr. Grupp have successfully closed the chapter.
“Oncologists have different notions of what the word ‘cure’ means. If your attitude is you’re not cured until you’ve basically reached the end of your life and you haven’t relapsed, well, that’s an impossible bar to hit. My attitude is, if your likelihood of having a disease recurrence is lower than the other risks in your life, like getting into your car and driving to your appointment, then that’s what a functional cure looks like,” he said.
“I’m probably the doctor that still sees her the most, but honestly, the whole conversation is not about leukemia at all. She has B-cell aplasia, so we have to treat that, and then it’s about making sure there’s no long-term side effects from the totality of her treatment. Generally, for a patient who’s gotten a moderate amount of chemotherapy and CAR T, that should not interfere with fertility. Has any patient in the history of the world ever relapsed more than 5 years out from their therapy? Of course. Is that incredibly rare? Yes, it is. You can be paralyzed by that, or you can compartmentalize it.”
As for the Whiteheads, they are focused on Emily’s college applications, her new driver’s license, and her project to cowrite a film about her story with a Hollywood filmmaker.
Mr. Whitehead said the one thing he hopes clinicians take away from their story is that sometimes a parent’s instinct transcends science.
A version of this article first appeared on Medscape.com.
A toddler presents with patchy hair loss
Given the history of sudden hair loss, with the exam revealing a well-circumscribed patch of focal alopecia without cutaneous inflammation, hairs with a narrow base and broad distal shaft, the diagnosis is alopecia areata (AA).
Alopecia areata (AA) is a nonscarring alopecia, within a set of diseases characterized by the preservation of hair follicles and therefore the potential for future hair regrowth.1 AA is believed to be caused by a breakdown of the immune-privileged nature of hair follicles, resulting in T-lymphocytes targeting the hair follicle directly, shifting follicles to early catagen or telogen phase, but sparing follicular stem cells, thereby allowing the follicle to regenerate in the future.1-3 Risk factors include family history of AA, thyroid disorders, as well as iron and vitamin D deficiency.4,5 It characteristically presents with focal, well-demarcated patches of hair loss in the scalp, typically with background skin normal to slightly pink.3,6 Exam can show “exclamation point” hairs consisting of hairs that are narrow at their base and wide at the distal end.3,7 Patients may also exhibit eyebrow and eyelash loss as well as nail changes including nail pitting and splitting.8 Diagnosis is typically made clinically but is supported by a positive hair pull test, where hairs are pulled from the periphery of an alopecic lesion; the presence of greater than 10% of hairs plucked from the scalp indicates a positive result.9,10
What’s the differential diagnosis?
The differential diagnosis of AA includes other nonscarring alopecias such as trichotillomania and telogen effluvium. Other possible diagnoses include lichen planopilaris and tinea capitis.
Trichotillomania results in irregularly bordered hair loss and broken hairs of different lengths because of an internal urge to remove one’s hair, resulting in nonscarring alopecia. It can be associated with obsessive-compulsive disorder, anxiety, or other body-altering behaviors like skin picking and nail biting (characterized as body-focused repetitive behavior disorders). Treatments include reassurance and education, behavior modification, or systemic therapy including tricyclic antidepressants or SSRIs. Toddlers can engage in hair pulling behavior and trichotillomania can be difficult to differentiate from AA. However, the absence of broken hairs of varying lengths makes trichotillomania less likely in this patient.
Telogen effluvium is another form of nonscarring alopecia that presents as diffuse hair thinning across the entire scalp in response to acute psychological or physiological stress, hormonal changes, certain medications, systemic illness, or nutritional deficiency. The timing between the triggering event and hair loss can vary from weeks to months. Diagnosis requires detailed history-taking and may include evaluation for endocrinologic hair thinning (e.g. thyroid function tests) to identify reversible causes. Treatment involves directing therapy to the underlying etiology and most cases of telogen effluvium are self-limited. The presence of a well-circumscribed patch of hair loss in this patient makes AA more likely.
Lichen planopilaris (LPP) is a scarring, irreversible alopecia caused by T-lymphocytes attacking follicular hair stem cells. It is characterized by hair loss, pruritus, burning pain, scalp scaling, and multifocal scarring. Exam shows patches of alopecia with loss of follicular ostia centrally and perifollicular scale and erythema at the borders. Diagnosis is aided by biopsy of the affected scalp. Treatment of LPP requires the use of potent and superpotent topical corticosteroids and intralesional corticosteroids to decrease scalp inflammation and prevent further progression. The presence of follicular ostia and absence of perifollicular scale in this patient makes LPP highly unlikely.
Tinea capitis is a fungal infection of the scalp caused by dermatophytes including Trychophyton tonsurans and Microsporum canis. It presents with patches of alopecia with overlying scale and broken hairs and can have associated cervical and occipital lymphadenopathy. Diagnosis can involve skin scraping and KOH prep to visualize branching hyphae as well as fungal culture to identify the causative organism. Because dermatophytes in tinea capitis invade hair follicles, topical antifungals are ineffective because of their lack of penetration. Therefore, systemic antifungals including oral terbinafine and griseofulvin are considered first-line agents for treatment.
What’s the management plan?
The diagnosis of AA is usually a clinical one, though assessment of alternative diagnoses is appropriate dependent on signs and symptoms. Workup of AA can include thyroid studies because of the association with autoimmune thyroid disease, though studies suggest limited screening benefits in children.11 Given its variable and unpredictable course, management can include “watchful waiting” because of its potential for spontaneous remission.6 For limited patchy loss, active treatment with mid to superpotent topical steroids or intralesional triamcinolone acetonide in older children and adolescents is reasonable.12 Other treatment options include topical or low-dose oral minoxidil and immunotherapy with diphenylcyclopropenone or squaric acid (inducing an allergic contact dermatitis).12 Management of therapies for more extensive AA is evolving, with ongoing studies of oral JAK-inhibitors and biologic agents.12,13
Our patient was started on topical fluocinonide 0.05% solution and achieved good disease control and hair regrowth over the course of 3 months.
Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Haft is an inflammatory skin disease fellow in the division of pediatric and adolescent dermatology at the university and Rady Children’s Hospital. They had no disclosures.
References
1. Bernardez C et al. Actas Dermosifiliogr. 2015;106(3):158-67.
2. Rajabi F et al. Br J Dermatol. 2018;179(5):1033-48.
3. Strazzulla LC et al. J Am Acad Dermatol. 2018;78(1):1-12.
4. Lee S et al. J Am Acad Dermatol. 2019;80(2):466-77 e16.
5. MacLean KJ and Tidman MJ. Practitioner. 2013;257(1764):29-32, 3.
6. Pratt CH et al. Nat Rev Dis Primers. 2017;3:17011.
7. Gilhar A et al. N Engl J Med. 2012;366(16):1515-25.
8. Wyrwich KW et al. Am J Clin Dermatol. 2020;21(5):725-32.
9. Spano F and Donovan JC. Can Fam Physician. 2015;61(9):751-5.
10. Mounsey AL and Reed SW. Am Fam Physician. 2009;80(4):356-62.
11. Hordinsky MK. J Investig Dermatol Symp Proc. 2015;17(2):44-6.
12. Strazzulla LC et al. J Am Acad Dermatol. 2018;78(1):15-24.
13. Zhou C et al. Clin Rev Allergy Immunol. 2021;61(3):403-23.
Given the history of sudden hair loss, with the exam revealing a well-circumscribed patch of focal alopecia without cutaneous inflammation, hairs with a narrow base and broad distal shaft, the diagnosis is alopecia areata (AA).
Alopecia areata (AA) is a nonscarring alopecia, within a set of diseases characterized by the preservation of hair follicles and therefore the potential for future hair regrowth.1 AA is believed to be caused by a breakdown of the immune-privileged nature of hair follicles, resulting in T-lymphocytes targeting the hair follicle directly, shifting follicles to early catagen or telogen phase, but sparing follicular stem cells, thereby allowing the follicle to regenerate in the future.1-3 Risk factors include family history of AA, thyroid disorders, as well as iron and vitamin D deficiency.4,5 It characteristically presents with focal, well-demarcated patches of hair loss in the scalp, typically with background skin normal to slightly pink.3,6 Exam can show “exclamation point” hairs consisting of hairs that are narrow at their base and wide at the distal end.3,7 Patients may also exhibit eyebrow and eyelash loss as well as nail changes including nail pitting and splitting.8 Diagnosis is typically made clinically but is supported by a positive hair pull test, where hairs are pulled from the periphery of an alopecic lesion; the presence of greater than 10% of hairs plucked from the scalp indicates a positive result.9,10
What’s the differential diagnosis?
The differential diagnosis of AA includes other nonscarring alopecias such as trichotillomania and telogen effluvium. Other possible diagnoses include lichen planopilaris and tinea capitis.
Trichotillomania results in irregularly bordered hair loss and broken hairs of different lengths because of an internal urge to remove one’s hair, resulting in nonscarring alopecia. It can be associated with obsessive-compulsive disorder, anxiety, or other body-altering behaviors like skin picking and nail biting (characterized as body-focused repetitive behavior disorders). Treatments include reassurance and education, behavior modification, or systemic therapy including tricyclic antidepressants or SSRIs. Toddlers can engage in hair pulling behavior and trichotillomania can be difficult to differentiate from AA. However, the absence of broken hairs of varying lengths makes trichotillomania less likely in this patient.
Telogen effluvium is another form of nonscarring alopecia that presents as diffuse hair thinning across the entire scalp in response to acute psychological or physiological stress, hormonal changes, certain medications, systemic illness, or nutritional deficiency. The timing between the triggering event and hair loss can vary from weeks to months. Diagnosis requires detailed history-taking and may include evaluation for endocrinologic hair thinning (e.g. thyroid function tests) to identify reversible causes. Treatment involves directing therapy to the underlying etiology and most cases of telogen effluvium are self-limited. The presence of a well-circumscribed patch of hair loss in this patient makes AA more likely.
Lichen planopilaris (LPP) is a scarring, irreversible alopecia caused by T-lymphocytes attacking follicular hair stem cells. It is characterized by hair loss, pruritus, burning pain, scalp scaling, and multifocal scarring. Exam shows patches of alopecia with loss of follicular ostia centrally and perifollicular scale and erythema at the borders. Diagnosis is aided by biopsy of the affected scalp. Treatment of LPP requires the use of potent and superpotent topical corticosteroids and intralesional corticosteroids to decrease scalp inflammation and prevent further progression. The presence of follicular ostia and absence of perifollicular scale in this patient makes LPP highly unlikely.
Tinea capitis is a fungal infection of the scalp caused by dermatophytes including Trychophyton tonsurans and Microsporum canis. It presents with patches of alopecia with overlying scale and broken hairs and can have associated cervical and occipital lymphadenopathy. Diagnosis can involve skin scraping and KOH prep to visualize branching hyphae as well as fungal culture to identify the causative organism. Because dermatophytes in tinea capitis invade hair follicles, topical antifungals are ineffective because of their lack of penetration. Therefore, systemic antifungals including oral terbinafine and griseofulvin are considered first-line agents for treatment.
What’s the management plan?
The diagnosis of AA is usually a clinical one, though assessment of alternative diagnoses is appropriate dependent on signs and symptoms. Workup of AA can include thyroid studies because of the association with autoimmune thyroid disease, though studies suggest limited screening benefits in children.11 Given its variable and unpredictable course, management can include “watchful waiting” because of its potential for spontaneous remission.6 For limited patchy loss, active treatment with mid to superpotent topical steroids or intralesional triamcinolone acetonide in older children and adolescents is reasonable.12 Other treatment options include topical or low-dose oral minoxidil and immunotherapy with diphenylcyclopropenone or squaric acid (inducing an allergic contact dermatitis).12 Management of therapies for more extensive AA is evolving, with ongoing studies of oral JAK-inhibitors and biologic agents.12,13
Our patient was started on topical fluocinonide 0.05% solution and achieved good disease control and hair regrowth over the course of 3 months.
Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Haft is an inflammatory skin disease fellow in the division of pediatric and adolescent dermatology at the university and Rady Children’s Hospital. They had no disclosures.
References
1. Bernardez C et al. Actas Dermosifiliogr. 2015;106(3):158-67.
2. Rajabi F et al. Br J Dermatol. 2018;179(5):1033-48.
3. Strazzulla LC et al. J Am Acad Dermatol. 2018;78(1):1-12.
4. Lee S et al. J Am Acad Dermatol. 2019;80(2):466-77 e16.
5. MacLean KJ and Tidman MJ. Practitioner. 2013;257(1764):29-32, 3.
6. Pratt CH et al. Nat Rev Dis Primers. 2017;3:17011.
7. Gilhar A et al. N Engl J Med. 2012;366(16):1515-25.
8. Wyrwich KW et al. Am J Clin Dermatol. 2020;21(5):725-32.
9. Spano F and Donovan JC. Can Fam Physician. 2015;61(9):751-5.
10. Mounsey AL and Reed SW. Am Fam Physician. 2009;80(4):356-62.
11. Hordinsky MK. J Investig Dermatol Symp Proc. 2015;17(2):44-6.
12. Strazzulla LC et al. J Am Acad Dermatol. 2018;78(1):15-24.
13. Zhou C et al. Clin Rev Allergy Immunol. 2021;61(3):403-23.
Given the history of sudden hair loss, with the exam revealing a well-circumscribed patch of focal alopecia without cutaneous inflammation, hairs with a narrow base and broad distal shaft, the diagnosis is alopecia areata (AA).
Alopecia areata (AA) is a nonscarring alopecia, within a set of diseases characterized by the preservation of hair follicles and therefore the potential for future hair regrowth.1 AA is believed to be caused by a breakdown of the immune-privileged nature of hair follicles, resulting in T-lymphocytes targeting the hair follicle directly, shifting follicles to early catagen or telogen phase, but sparing follicular stem cells, thereby allowing the follicle to regenerate in the future.1-3 Risk factors include family history of AA, thyroid disorders, as well as iron and vitamin D deficiency.4,5 It characteristically presents with focal, well-demarcated patches of hair loss in the scalp, typically with background skin normal to slightly pink.3,6 Exam can show “exclamation point” hairs consisting of hairs that are narrow at their base and wide at the distal end.3,7 Patients may also exhibit eyebrow and eyelash loss as well as nail changes including nail pitting and splitting.8 Diagnosis is typically made clinically but is supported by a positive hair pull test, where hairs are pulled from the periphery of an alopecic lesion; the presence of greater than 10% of hairs plucked from the scalp indicates a positive result.9,10
What’s the differential diagnosis?
The differential diagnosis of AA includes other nonscarring alopecias such as trichotillomania and telogen effluvium. Other possible diagnoses include lichen planopilaris and tinea capitis.
Trichotillomania results in irregularly bordered hair loss and broken hairs of different lengths because of an internal urge to remove one’s hair, resulting in nonscarring alopecia. It can be associated with obsessive-compulsive disorder, anxiety, or other body-altering behaviors like skin picking and nail biting (characterized as body-focused repetitive behavior disorders). Treatments include reassurance and education, behavior modification, or systemic therapy including tricyclic antidepressants or SSRIs. Toddlers can engage in hair pulling behavior and trichotillomania can be difficult to differentiate from AA. However, the absence of broken hairs of varying lengths makes trichotillomania less likely in this patient.
Telogen effluvium is another form of nonscarring alopecia that presents as diffuse hair thinning across the entire scalp in response to acute psychological or physiological stress, hormonal changes, certain medications, systemic illness, or nutritional deficiency. The timing between the triggering event and hair loss can vary from weeks to months. Diagnosis requires detailed history-taking and may include evaluation for endocrinologic hair thinning (e.g. thyroid function tests) to identify reversible causes. Treatment involves directing therapy to the underlying etiology and most cases of telogen effluvium are self-limited. The presence of a well-circumscribed patch of hair loss in this patient makes AA more likely.
Lichen planopilaris (LPP) is a scarring, irreversible alopecia caused by T-lymphocytes attacking follicular hair stem cells. It is characterized by hair loss, pruritus, burning pain, scalp scaling, and multifocal scarring. Exam shows patches of alopecia with loss of follicular ostia centrally and perifollicular scale and erythema at the borders. Diagnosis is aided by biopsy of the affected scalp. Treatment of LPP requires the use of potent and superpotent topical corticosteroids and intralesional corticosteroids to decrease scalp inflammation and prevent further progression. The presence of follicular ostia and absence of perifollicular scale in this patient makes LPP highly unlikely.
Tinea capitis is a fungal infection of the scalp caused by dermatophytes including Trychophyton tonsurans and Microsporum canis. It presents with patches of alopecia with overlying scale and broken hairs and can have associated cervical and occipital lymphadenopathy. Diagnosis can involve skin scraping and KOH prep to visualize branching hyphae as well as fungal culture to identify the causative organism. Because dermatophytes in tinea capitis invade hair follicles, topical antifungals are ineffective because of their lack of penetration. Therefore, systemic antifungals including oral terbinafine and griseofulvin are considered first-line agents for treatment.
What’s the management plan?
The diagnosis of AA is usually a clinical one, though assessment of alternative diagnoses is appropriate dependent on signs and symptoms. Workup of AA can include thyroid studies because of the association with autoimmune thyroid disease, though studies suggest limited screening benefits in children.11 Given its variable and unpredictable course, management can include “watchful waiting” because of its potential for spontaneous remission.6 For limited patchy loss, active treatment with mid to superpotent topical steroids or intralesional triamcinolone acetonide in older children and adolescents is reasonable.12 Other treatment options include topical or low-dose oral minoxidil and immunotherapy with diphenylcyclopropenone or squaric acid (inducing an allergic contact dermatitis).12 Management of therapies for more extensive AA is evolving, with ongoing studies of oral JAK-inhibitors and biologic agents.12,13
Our patient was started on topical fluocinonide 0.05% solution and achieved good disease control and hair regrowth over the course of 3 months.
Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Haft is an inflammatory skin disease fellow in the division of pediatric and adolescent dermatology at the university and Rady Children’s Hospital. They had no disclosures.
References
1. Bernardez C et al. Actas Dermosifiliogr. 2015;106(3):158-67.
2. Rajabi F et al. Br J Dermatol. 2018;179(5):1033-48.
3. Strazzulla LC et al. J Am Acad Dermatol. 2018;78(1):1-12.
4. Lee S et al. J Am Acad Dermatol. 2019;80(2):466-77 e16.
5. MacLean KJ and Tidman MJ. Practitioner. 2013;257(1764):29-32, 3.
6. Pratt CH et al. Nat Rev Dis Primers. 2017;3:17011.
7. Gilhar A et al. N Engl J Med. 2012;366(16):1515-25.
8. Wyrwich KW et al. Am J Clin Dermatol. 2020;21(5):725-32.
9. Spano F and Donovan JC. Can Fam Physician. 2015;61(9):751-5.
10. Mounsey AL and Reed SW. Am Fam Physician. 2009;80(4):356-62.
11. Hordinsky MK. J Investig Dermatol Symp Proc. 2015;17(2):44-6.
12. Strazzulla LC et al. J Am Acad Dermatol. 2018;78(1):15-24.
13. Zhou C et al. Clin Rev Allergy Immunol. 2021;61(3):403-23.
Examination findings of the scalp demonstrate a well-circumscribed alopecic patch on the vertex scalp without erythema or scale. Closer inspection of the patch with magnification or 'dermoscopy' reveals hair follicle ostia and hairs that are broader distally and narrower at their base. Nails and rest of the skin exam are unremarkable.
Biden tests positive for COVID-19: White House
Biden, 79, is experiencing “very mild” symptoms, White House Press Secretary Karine Jean-Pierre said in a statement. The president is fully vaccinated and has been boosted twice and has started taking the antiviral Paxlovid since testing positive, Ms. Jean-Pierre said.
President Biden plans to isolate at the White House and “will continue to carry out all of his duties fully during that time,” the statement said.
“He has been in contact with members of the White House staff by phone this morning, and will participate in his planned meetings at the White House this morning via phone and Zoom from the residence.”
President Biden will return to in-person work after he tests negative.
This is a developing story. Please check back for updates. A version of this article first appeared on WebMD.com .
Biden, 79, is experiencing “very mild” symptoms, White House Press Secretary Karine Jean-Pierre said in a statement. The president is fully vaccinated and has been boosted twice and has started taking the antiviral Paxlovid since testing positive, Ms. Jean-Pierre said.
President Biden plans to isolate at the White House and “will continue to carry out all of his duties fully during that time,” the statement said.
“He has been in contact with members of the White House staff by phone this morning, and will participate in his planned meetings at the White House this morning via phone and Zoom from the residence.”
President Biden will return to in-person work after he tests negative.
This is a developing story. Please check back for updates. A version of this article first appeared on WebMD.com .
Biden, 79, is experiencing “very mild” symptoms, White House Press Secretary Karine Jean-Pierre said in a statement. The president is fully vaccinated and has been boosted twice and has started taking the antiviral Paxlovid since testing positive, Ms. Jean-Pierre said.
President Biden plans to isolate at the White House and “will continue to carry out all of his duties fully during that time,” the statement said.
“He has been in contact with members of the White House staff by phone this morning, and will participate in his planned meetings at the White House this morning via phone and Zoom from the residence.”
President Biden will return to in-person work after he tests negative.
This is a developing story. Please check back for updates. A version of this article first appeared on WebMD.com .