User login
The latest research you need to know
In This Edition
- Generic vs. brand-name drugs.
- Rapid-response teams and mortality.
- A new prediction rule for mortality in acute pancreatitis.
- Viral causes of community-acquired pneumonia.
- Intensive insulin therapy in the ICU.
- New preoperative and intraoperative risk factors.
- Timing of ICU feedings and mortality.
- Aspirin as primary prevention in diabetics.
Generic, Brand-Name Drugs Used for Cardiovascular Disease Are Clinically Equivalent
Clinical question: Is there a clinical risk when substituting generic drugs for brand-name drugs in the treatment of cardiovascular disease?
Background: Spending on healthcare in the U.S. has reached critical levels. Increasing prescription drug costs make up a large portion of healthcare expenditures. The high cost of medicines directly affect adherence to treatment regimens and contribute to poor health outcomes. Cardiovascular drugs make up the largest portion of outpatient prescription drug spending.
Study design: Systematic review of relevant articles with a meta-analysis performed to determine an aggregate effect size.
Setting: Multiple locations and varied patient populations.
Synopsis: A total of 47 articles were included in the review, of which 38 were randomized controlled trials (RCTs). The studies measured both clinical efficacy and safety end points. More than half the articles were published prior to 2000. Clinical equivalence was noted in all seven beta-blocker RCTs; 10 of 11 diuretic RCTs; five of seven calcium-channel-blocker RCTs; all three antiplatelet-agent RCTs (clopidogrel, enteric-coated aspirin); two statin RCTs; one ACE-inhibitor RCT; and one alpha-blocker RCT. For drugs with a narrow therapeutic index, clinical equivalence was noted in all five warfarin RCTs and a single Class 1 anti-arrhythmic-agent RCT.
The aggregate effect size was -0.03 (95% CI, -0.15 to 0.08), which indicates nearly complete overlap of the generic and brand-name distributions. The data show no evidence of superiority of brand-name to generic drugs in clinical outcomes measured in the various studies.
In a separate review of editorials addressing generic substitution for cardiovascular drugs, 53% expressed a negative view of generic-drug substitution.
Bottom line: There is clinical equivalency between generic and brand-name drugs used in the treatment of cardiovascular disease. Despite this conclusion, a substantial number of editorials advise against generic substitution, which affects both patient and physician drug preferences.
Citation: Kesselheim A, Misono A, Lee J, et al. Clinical equivalence of generic and brand-name drugs used in cardiovascular disease: a systematic review and meta-analysis. JAMA. 2008; 300(21):2514-2526.
RRT Implement-ation Doesn’t Affect Hospitalwide Code Rates or Mortality
Clinical question: Does the use of a rapid-response team (RRT) affect hospitalwide code rates and mortality?
Background: In the 100,000 Lives campaign, the Institute for Healthcare Improvement recommended that hospitals implement RRTs to help reduce preventable, in-hospital deaths. Studies have demonstrated that rates of non-ICU codes decrease after RRT implementation. It is unclear if this translates to changes in mortality rates.
Study design: Prospective cohort study of adult inpatients.
Setting: Saint Luke’s Hospital, a 404-bed tertiary-care academic hospital in Kansas City, Mo.
Synopsis: The hospital documented a total of 376 RRT activations. After RRT implementation, mean hospitalwide code rates decreased to 7.5 per 1,000 admissions from 11.2 per 1,000 admissions. This was not associated with a statistically significant reduction in hospitalwide code rates (adjusted odds ratio 0.76; 95% CI; 0.57-1.01; P=0.06). Secondary analyses noted lower rates of non-ICU codes (0.59; 95% CI, 0.40-0.89) compared with ICU codes (0.95; 95% CI; 0.64-1.43; P=0.03 for interaction). Finally, the RRT implementation was not associated with lower hospital-wide mortality (0.95; 95% CI; 0.81-1.11; P=0.52).
Secondary analyses also revealed few instances of RRT undertreatment or underutilization that may have affected the mortality numbers.
A limitation of this study is that it was slightly underpowered (78%) to detect a significant mortality difference. The findings also represent a single institution experience, and may not be generalized to other adult hospital settings or RRT programs.
Bottom line: Implementation of an RRT does not confer lower rates of hospital-wide code arrests or mortality.
Citation: Chan P, Khalid A, Longmore L, et al. Hospital-wide code rates and mortality before and after implementation of a rapid response team. JAMA. 2008;300(21):2506-2513.
Simple Scoring System Provides Timely Prediction of Mortality in Acute Pancreatitis
Clinical question: How can physicians predict mortality in acute pancreatitis?
Background: Historical predictors of mortality in acute pancreatitis require up to 48 hours of data, such as with the Ranson Criteria, or extensive amounts of data, such as with the APACHE II score. An easier tool is needed to predict which patients are at higher risk of mortality.
Study design: Retrospective cohort.
Setting: Patients in the Cardinal Health clinical outcomes research database, which supports public reporting of hospital performance.
Synopsis: The authors identified patients with the principal diagnosis of pancreatitis from 2000-2001 and explored numerous diagnostic findings available within the first 24 hours. Ultimately, BUN >25, impaired mental status, presence of SIRS (systemic inflammatory response syndrome), age >60, and presence of a pleural effusion were found to be predictive of mortality. These diagnostic findings correspond to the mnemonic BISAP. The BISAP score was then validated in a second cohort that included patients from 2004-2005.
Each finding in the BISAP score was given one point. A score of less than 2 was present in approximately 60% of patients admitted with acute pancreatitis, and corresponded to a mortality of less than 1%. A score of 2 corresponded to a mortality of 2%. Higher scores were associated with steeply increasing mortality, with a score of 5 corresponding with greater than 20% mortality.
The BISAP score performed similarly to the APACHE II score, but the former is easier to calculate on the day of admission and has fewer parameters. A more challenging research step will be to demonstrate that using the BISAP score to determine treatment strategies can affect patient outcomes.
Bottom line: The easy-to-calculate BISAP score is a new method for predicting mortality in patients with acute pancreatitis. This tool should help hospitalists determine, on the day of admission, to what extent patients with acute pancreatitis need aggressive management, such as ICU admission and early involvement of specialists.
Citation: Wu B, Johannes R, Sun X, Tabak Y, Conwell D, Banks P. The early prediction of mortality in acute pancreatitis: a large population-based study. Gut. 2008;57(12): 1698-1703.
Nasal Swabs Identify Viral Causes in CAP Patients
Clinical question: How often is viral infection associated with community-acquired pneumonia (CAP) in adults?
Background: CAP is a clinically important disease that is associated with significant hospitalization and mortality. CAP treatment guidelines acknowledge respiratory viruses as an etiology for pneumonia, but few recommendations are made regarding management of these viral infections.
Study design: Prospective study.
Setting: Five hospitals in Edmonton, Alberta, from 2004-2006.
Synopsis: The authors enrolled 193 hospitalized adults, median age 71. Nucleic amplification tests (NATs) from nasopharyngeal swab specimens were tested for human metapneumovirus, respiratory syncytial virus, rhinovirus, parainfluenza virus 1-4, coronaviruses, and adenovirus.
Fifteen percent of these patients had a nasal swab positive for a viral infection. Interestingly, 4% of patients had both a positive viral nasal swab and positive bacterial cultures. Compared with bacterial infection, patients with viral infection tended to be older (76 vs. 64 years, P=0.01), have limited ambulation (66% vs. 32%, P=0.006), and have a history of cardiac disease (66% vs. 32%, P=0.02). Patients with bacterial pneumonia showed a statistically significant trend toward having chest pain, an abnormal white blood count, and a lobar infiltrate on chest X-ray.
Further investigations might look at how nasal swab data could be used to improve infection control within the hospital for patients found to have easily transmissible viruses. Further research could explore the feasibility of avoiding antibiotic use in patients found to have viral pneumonia, assuming bacterial co-infection is reliably excluded.
Bottom line: Nasal swabs using NAT technology could play a significant role in identifying pathogens in CAP patients. How this technology should affect clinical decision-making and how it might improve outcomes remains unknown.
Citation: Johnstone J, Majumdar S, Fox J, Marrie T. Viral infection in adults hospitalized with community-acquired pneumonia: prevalence, pathogens, and presentation. Chest. 2008;134(6):1141-1148.
Intensive Insulin Therapy Doesn’t Reduce ICU Mortality
Clinical question: Does the use of intensive insulin therapy decrease mortality in the intensive-care unit (ICU)?
Background: In 2001, Van den Berghe et al (N Engl J Med. 2001;345(19):1359-67) reported a reduction in morbidity and mortality with intensive insulin therapy (IIT) in surgical ICU patients. This study led to the adoption of IIT protocols in many hospitals. Since 2001, further studies have failed to reproduce the same dramatic benefit of IIT.
Study design: Randomized, controlled trial.
Setting: National Guard King Abdulaziz Medical City, a tertiary-care teaching hospital in Saudi Arabia.
Synopsis: Patients were included in this study if they were 18 years or older with serum glucose levels greater than 110 mg/dL during the first 24 hours of ICU admission. There were multiple exclusion criteria, including patients with Type I diabetes, documented hypoglycemia on ICU admission (glucose <40), or diabetic ketoacidosis.
Enrolled patients were randomized to IIT or conventional insulin therapy (CIT). A multidisciplinary team designed the protocols to maintain glucose levels of 80 to 110 mg/dL and 180 to 200 mg/dL in the IIT and the CIT groups, respectively. The primary endpoint measured was ICU mortality.
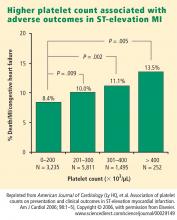
The study did not produce a statistically significant difference in ICU mortality (13.5% for IIT vs. 17.1% for CIT; P=0.30). The adjusted hypoglycemia rate was 6.8 per 100 treatment days with IIT and 0.4 per 100 treatment days with CIT (P<0.0001). Patients with hypoglycemia had higher ICU mortality (23.8% vs. 13.7%, P=0.02).
In the measurement of secondary endpoints, there was a trend toward lower episodes of severe sepsis and septic shock in the IIT group (20.7% in IIT vs. 27.2% in CIT, P=0.08). However, this result was not statistically significant.
Bottom line: This well-designed study failed to show a survival benefit with IIT use in the critical-care setting. Given the findings of this and several other recent studies, one should question whether IIT should be prescribed as the standard of care in all critically-ill patients.
Citation: Arabi Y, Dabbagh O, Tamim H, et al. Intensive versus conventional insulin therapy: a randomized controlled trial in medical and surgical critically ill patients. Crit Care Med. 2008;36(12):3190-3197.
Preoperative and Intraoperative Predictors of Cardiac Adverse Events
Clinical question: What are the incidence and risk factors for perioperative cardiac adverse events after noncardiac surgery?
Background: In the past few decades, the incidence of cardiac adverse events (CAEs) for a general surgery population has remained largely unchanged (approximately 1%). This is in spite of multiple studies evaluating predictive models and attempts at preventive treatment, including preoperative coronary revascularization and perioperative beta blockers.
Study design: Prospective observational study.
Setting: Single, large, tertiary-care university hospital.
Synopsis: A total of 7,740 cases were reviewed in this study, which consisted of general surgery (4,937), vascular surgery (1,846), and urological surgery (957). A trained nurse followed up for perioperative CAEs as many as 30 days after the operation via medical chart review, phone calls, and letters. CAEs were defined as: Q-wave myocardial infarction (MI), non-ST elevation MI, cardiac arrest, or new cardiac dysrhythmia. A total of 83 CAEs (1.1% of patients) had cardiac arrest, with cardiac dysrhythmia being most common.
A total of seven preoperative risk factors were identified as independent predictors for CAEs: age 68, BMI 30, emergent surgery, prior coronary intervention or cardiac surgery, active congestive heart failure, cerebrovascular disease, and hypertension. In addition, two intraoperative risk factors were identified: intraoperative transfusion of packed red blood cells and operative duration of 3.8 hours. (The P value was 0.05 for all independent predictors.)
A further evaluation of intraoperative parameters of high-risk patients experiencing a CAE showed that these patients were more likely to have an episode of mean arterial pressure (MAP) <50 mmHg, an episode of 40% decrease in MAP, and an episode of tachycardia (heart rate >100).
Bottom line: A combination of preoperative predictors and intraoperative elements can help improve risk assessment for perioperative CAEs after noncardiac surgery.
Citation: Kheterpal S, O’Reilly M, Englesbe M, et al. Preoperative and intraoperative predictors of cardiac adverse events after general, vascular, and urological surgery. Anesthesiology. 2009;110(1):58-66.
Early Feeding in the ICU Does Not Affect Hospital Mortality
Clinical question: Will implementing evidence-based feeding guidelines improve feeding practices and reduce mortality in ICU patients?
Background: There is evidence suggesting that providing nutritional support to ICU patients within 24 hours of admission may decrease mortality. It is widely understood that patient care varies between ICUs, and there exist no uniform, evidence-based guidelines for nutritional support. Many patients remain unfed after 48 hours.
Study design: Cluster, randomized-controlled trial.
Setting: ICUs in 27 community and tertiary-care hospitals in Australia and New Zealand.
Synopsis: Hospitals were randomized to intervention or control groups. Dietitian and intensivist co-investigators from intervention ICUs developed guidelines using the Clinical Practice Guideline Development Cycle. Control ICUs were requested to make no new ICU nutritional management changes. The study enrolled 1,118 eligible patients and included adults expected to stay longer than two days. Excluded were patients taking oral diets, patients receiving palliative care or with suspected brain death, and patients sent from other facilities.
Guidelines were implemented using several methods—educational outreach visits, one-on-one conversations, active reminders, passive reminders, and educational in-services. The guidelines were successful in evoking significant practice changes in all of the intervention ICUs. Significantly more patients received nutritional support during their ICU stays in guideline ICUs, and patients in these ICUs were fed significantly earlier. There were, however, no significant differences between guideline and control ICUs with regard to hospital discharge mortality (28.9% vs. 27.4%; 95% CI; -6.3% to 12.0%; P=0.75). The groups also showed no statistical difference in hospital or ICU length of stay.
Bottom line: Significantly more patients in the guideline ICUs were fed within 24 hours, but this did not translate into improvements in mortality or other clinical outcomes.
Citation: Doig G, Simpson F, Finfer S, et al. Effect of evidence-based feeding guidelines on mortality of critically ill adults: a cluster randomized controlled trial. JAMA. 2008;300(23):2731-2741.
Low-Dose Aspirin Doesn’t Reduce Cardiovascular Events in Type 2 Diabetes Patients
Clinical question: Is low-dose aspirin effective for the primary prevention of atherosclerotic events in patients with Type 2 diabetes?
Background: Diabetes is a significant risk factor for cardiovascular events. Several prior studies have shown that aspirin therapy is effective as a secondary prevention strategy for cardiovascular events. The American Diabetes Association also recommends use of aspirin as a primary prevention strategy. Clinical trial data is currently insufficient in this area.
Study design: Prospective, randomized, open-label, controlled trial with blinded endpoint assessment.
Setting: 163 institutions in Japan.
Synopsis: The study enrolled 2,539 diabetic patients between 30 and 85 years old—mean age was 65—and included patients without EKG changes or a significant history or ongoing treatment of atherosclerotic disease. Patients were randomly assigned into an aspirin group (81 mg or 100 mg once daily) or a nonaspirin group. Patients had a median follow up of 4.37 years.
The primary endpoint was any atherosclerotic event, ranging from sudden death to ischemic heart disease or stroke. The incidence of atherosclerotic events was not statistically different in the aspirin group (68 events, 5.4%) than in the nonaspirin group (86 events, 6.7%) (HR, 0.80; 95% CI; 0.58-1.10; log-rank test, P=0.16). However, there was a suggested benefit of primary prevention in the subgroup aged 65 years or older. In addition, the combined endpoint of fatal coronary and cerebrovascular events occurred in one patient in the aspirin group and 10 patients in the nonaspirin group (HR, 0.10; 95 % CI, 0.01-0.79; P=.0037). This study is limited by the low incidence of atherosclerotic disease in Japan.
Bottom line: Low-dose aspirin used in patients with Type 2 diabetes does not significantly demonstrate primary prevention of cardiovascular events.
Citation: Ogawa H, Nakayama M, Morimoto T, et al. Low-dose aspirin for primary prevention of atherosclerotic events in patients with type 2 diabetes: a randomized controlled trial. JAMA. 2008;300(18):2134-2141. TH
In This Edition
- Generic vs. brand-name drugs.
- Rapid-response teams and mortality.
- A new prediction rule for mortality in acute pancreatitis.
- Viral causes of community-acquired pneumonia.
- Intensive insulin therapy in the ICU.
- New preoperative and intraoperative risk factors.
- Timing of ICU feedings and mortality.
- Aspirin as primary prevention in diabetics.
Generic, Brand-Name Drugs Used for Cardiovascular Disease Are Clinically Equivalent
Clinical question: Is there a clinical risk when substituting generic drugs for brand-name drugs in the treatment of cardiovascular disease?
Background: Spending on healthcare in the U.S. has reached critical levels. Increasing prescription drug costs make up a large portion of healthcare expenditures. The high cost of medicines directly affect adherence to treatment regimens and contribute to poor health outcomes. Cardiovascular drugs make up the largest portion of outpatient prescription drug spending.
Study design: Systematic review of relevant articles with a meta-analysis performed to determine an aggregate effect size.
Setting: Multiple locations and varied patient populations.
Synopsis: A total of 47 articles were included in the review, of which 38 were randomized controlled trials (RCTs). The studies measured both clinical efficacy and safety end points. More than half the articles were published prior to 2000. Clinical equivalence was noted in all seven beta-blocker RCTs; 10 of 11 diuretic RCTs; five of seven calcium-channel-blocker RCTs; all three antiplatelet-agent RCTs (clopidogrel, enteric-coated aspirin); two statin RCTs; one ACE-inhibitor RCT; and one alpha-blocker RCT. For drugs with a narrow therapeutic index, clinical equivalence was noted in all five warfarin RCTs and a single Class 1 anti-arrhythmic-agent RCT.
The aggregate effect size was -0.03 (95% CI, -0.15 to 0.08), which indicates nearly complete overlap of the generic and brand-name distributions. The data show no evidence of superiority of brand-name to generic drugs in clinical outcomes measured in the various studies.
In a separate review of editorials addressing generic substitution for cardiovascular drugs, 53% expressed a negative view of generic-drug substitution.
Bottom line: There is clinical equivalency between generic and brand-name drugs used in the treatment of cardiovascular disease. Despite this conclusion, a substantial number of editorials advise against generic substitution, which affects both patient and physician drug preferences.
Citation: Kesselheim A, Misono A, Lee J, et al. Clinical equivalence of generic and brand-name drugs used in cardiovascular disease: a systematic review and meta-analysis. JAMA. 2008; 300(21):2514-2526.
RRT Implement-ation Doesn’t Affect Hospitalwide Code Rates or Mortality
Clinical question: Does the use of a rapid-response team (RRT) affect hospitalwide code rates and mortality?
Background: In the 100,000 Lives campaign, the Institute for Healthcare Improvement recommended that hospitals implement RRTs to help reduce preventable, in-hospital deaths. Studies have demonstrated that rates of non-ICU codes decrease after RRT implementation. It is unclear if this translates to changes in mortality rates.
Study design: Prospective cohort study of adult inpatients.
Setting: Saint Luke’s Hospital, a 404-bed tertiary-care academic hospital in Kansas City, Mo.
Synopsis: The hospital documented a total of 376 RRT activations. After RRT implementation, mean hospitalwide code rates decreased to 7.5 per 1,000 admissions from 11.2 per 1,000 admissions. This was not associated with a statistically significant reduction in hospitalwide code rates (adjusted odds ratio 0.76; 95% CI; 0.57-1.01; P=0.06). Secondary analyses noted lower rates of non-ICU codes (0.59; 95% CI, 0.40-0.89) compared with ICU codes (0.95; 95% CI; 0.64-1.43; P=0.03 for interaction). Finally, the RRT implementation was not associated with lower hospital-wide mortality (0.95; 95% CI; 0.81-1.11; P=0.52).
Secondary analyses also revealed few instances of RRT undertreatment or underutilization that may have affected the mortality numbers.
A limitation of this study is that it was slightly underpowered (78%) to detect a significant mortality difference. The findings also represent a single institution experience, and may not be generalized to other adult hospital settings or RRT programs.
Bottom line: Implementation of an RRT does not confer lower rates of hospital-wide code arrests or mortality.
Citation: Chan P, Khalid A, Longmore L, et al. Hospital-wide code rates and mortality before and after implementation of a rapid response team. JAMA. 2008;300(21):2506-2513.
Simple Scoring System Provides Timely Prediction of Mortality in Acute Pancreatitis
Clinical question: How can physicians predict mortality in acute pancreatitis?
Background: Historical predictors of mortality in acute pancreatitis require up to 48 hours of data, such as with the Ranson Criteria, or extensive amounts of data, such as with the APACHE II score. An easier tool is needed to predict which patients are at higher risk of mortality.
Study design: Retrospective cohort.
Setting: Patients in the Cardinal Health clinical outcomes research database, which supports public reporting of hospital performance.
Synopsis: The authors identified patients with the principal diagnosis of pancreatitis from 2000-2001 and explored numerous diagnostic findings available within the first 24 hours. Ultimately, BUN >25, impaired mental status, presence of SIRS (systemic inflammatory response syndrome), age >60, and presence of a pleural effusion were found to be predictive of mortality. These diagnostic findings correspond to the mnemonic BISAP. The BISAP score was then validated in a second cohort that included patients from 2004-2005.
Each finding in the BISAP score was given one point. A score of less than 2 was present in approximately 60% of patients admitted with acute pancreatitis, and corresponded to a mortality of less than 1%. A score of 2 corresponded to a mortality of 2%. Higher scores were associated with steeply increasing mortality, with a score of 5 corresponding with greater than 20% mortality.
The BISAP score performed similarly to the APACHE II score, but the former is easier to calculate on the day of admission and has fewer parameters. A more challenging research step will be to demonstrate that using the BISAP score to determine treatment strategies can affect patient outcomes.
Bottom line: The easy-to-calculate BISAP score is a new method for predicting mortality in patients with acute pancreatitis. This tool should help hospitalists determine, on the day of admission, to what extent patients with acute pancreatitis need aggressive management, such as ICU admission and early involvement of specialists.
Citation: Wu B, Johannes R, Sun X, Tabak Y, Conwell D, Banks P. The early prediction of mortality in acute pancreatitis: a large population-based study. Gut. 2008;57(12): 1698-1703.
Nasal Swabs Identify Viral Causes in CAP Patients
Clinical question: How often is viral infection associated with community-acquired pneumonia (CAP) in adults?
Background: CAP is a clinically important disease that is associated with significant hospitalization and mortality. CAP treatment guidelines acknowledge respiratory viruses as an etiology for pneumonia, but few recommendations are made regarding management of these viral infections.
Study design: Prospective study.
Setting: Five hospitals in Edmonton, Alberta, from 2004-2006.
Synopsis: The authors enrolled 193 hospitalized adults, median age 71. Nucleic amplification tests (NATs) from nasopharyngeal swab specimens were tested for human metapneumovirus, respiratory syncytial virus, rhinovirus, parainfluenza virus 1-4, coronaviruses, and adenovirus.
Fifteen percent of these patients had a nasal swab positive for a viral infection. Interestingly, 4% of patients had both a positive viral nasal swab and positive bacterial cultures. Compared with bacterial infection, patients with viral infection tended to be older (76 vs. 64 years, P=0.01), have limited ambulation (66% vs. 32%, P=0.006), and have a history of cardiac disease (66% vs. 32%, P=0.02). Patients with bacterial pneumonia showed a statistically significant trend toward having chest pain, an abnormal white blood count, and a lobar infiltrate on chest X-ray.
Further investigations might look at how nasal swab data could be used to improve infection control within the hospital for patients found to have easily transmissible viruses. Further research could explore the feasibility of avoiding antibiotic use in patients found to have viral pneumonia, assuming bacterial co-infection is reliably excluded.
Bottom line: Nasal swabs using NAT technology could play a significant role in identifying pathogens in CAP patients. How this technology should affect clinical decision-making and how it might improve outcomes remains unknown.
Citation: Johnstone J, Majumdar S, Fox J, Marrie T. Viral infection in adults hospitalized with community-acquired pneumonia: prevalence, pathogens, and presentation. Chest. 2008;134(6):1141-1148.
Intensive Insulin Therapy Doesn’t Reduce ICU Mortality
Clinical question: Does the use of intensive insulin therapy decrease mortality in the intensive-care unit (ICU)?
Background: In 2001, Van den Berghe et al (N Engl J Med. 2001;345(19):1359-67) reported a reduction in morbidity and mortality with intensive insulin therapy (IIT) in surgical ICU patients. This study led to the adoption of IIT protocols in many hospitals. Since 2001, further studies have failed to reproduce the same dramatic benefit of IIT.
Study design: Randomized, controlled trial.
Setting: National Guard King Abdulaziz Medical City, a tertiary-care teaching hospital in Saudi Arabia.
Synopsis: Patients were included in this study if they were 18 years or older with serum glucose levels greater than 110 mg/dL during the first 24 hours of ICU admission. There were multiple exclusion criteria, including patients with Type I diabetes, documented hypoglycemia on ICU admission (glucose <40), or diabetic ketoacidosis.
Enrolled patients were randomized to IIT or conventional insulin therapy (CIT). A multidisciplinary team designed the protocols to maintain glucose levels of 80 to 110 mg/dL and 180 to 200 mg/dL in the IIT and the CIT groups, respectively. The primary endpoint measured was ICU mortality.
The study did not produce a statistically significant difference in ICU mortality (13.5% for IIT vs. 17.1% for CIT; P=0.30). The adjusted hypoglycemia rate was 6.8 per 100 treatment days with IIT and 0.4 per 100 treatment days with CIT (P<0.0001). Patients with hypoglycemia had higher ICU mortality (23.8% vs. 13.7%, P=0.02).
In the measurement of secondary endpoints, there was a trend toward lower episodes of severe sepsis and septic shock in the IIT group (20.7% in IIT vs. 27.2% in CIT, P=0.08). However, this result was not statistically significant.
Bottom line: This well-designed study failed to show a survival benefit with IIT use in the critical-care setting. Given the findings of this and several other recent studies, one should question whether IIT should be prescribed as the standard of care in all critically-ill patients.
Citation: Arabi Y, Dabbagh O, Tamim H, et al. Intensive versus conventional insulin therapy: a randomized controlled trial in medical and surgical critically ill patients. Crit Care Med. 2008;36(12):3190-3197.
Preoperative and Intraoperative Predictors of Cardiac Adverse Events
Clinical question: What are the incidence and risk factors for perioperative cardiac adverse events after noncardiac surgery?
Background: In the past few decades, the incidence of cardiac adverse events (CAEs) for a general surgery population has remained largely unchanged (approximately 1%). This is in spite of multiple studies evaluating predictive models and attempts at preventive treatment, including preoperative coronary revascularization and perioperative beta blockers.
Study design: Prospective observational study.
Setting: Single, large, tertiary-care university hospital.
Synopsis: A total of 7,740 cases were reviewed in this study, which consisted of general surgery (4,937), vascular surgery (1,846), and urological surgery (957). A trained nurse followed up for perioperative CAEs as many as 30 days after the operation via medical chart review, phone calls, and letters. CAEs were defined as: Q-wave myocardial infarction (MI), non-ST elevation MI, cardiac arrest, or new cardiac dysrhythmia. A total of 83 CAEs (1.1% of patients) had cardiac arrest, with cardiac dysrhythmia being most common.
A total of seven preoperative risk factors were identified as independent predictors for CAEs: age 68, BMI 30, emergent surgery, prior coronary intervention or cardiac surgery, active congestive heart failure, cerebrovascular disease, and hypertension. In addition, two intraoperative risk factors were identified: intraoperative transfusion of packed red blood cells and operative duration of 3.8 hours. (The P value was 0.05 for all independent predictors.)
A further evaluation of intraoperative parameters of high-risk patients experiencing a CAE showed that these patients were more likely to have an episode of mean arterial pressure (MAP) <50 mmHg, an episode of 40% decrease in MAP, and an episode of tachycardia (heart rate >100).
Bottom line: A combination of preoperative predictors and intraoperative elements can help improve risk assessment for perioperative CAEs after noncardiac surgery.
Citation: Kheterpal S, O’Reilly M, Englesbe M, et al. Preoperative and intraoperative predictors of cardiac adverse events after general, vascular, and urological surgery. Anesthesiology. 2009;110(1):58-66.
Early Feeding in the ICU Does Not Affect Hospital Mortality
Clinical question: Will implementing evidence-based feeding guidelines improve feeding practices and reduce mortality in ICU patients?
Background: There is evidence suggesting that providing nutritional support to ICU patients within 24 hours of admission may decrease mortality. It is widely understood that patient care varies between ICUs, and there exist no uniform, evidence-based guidelines for nutritional support. Many patients remain unfed after 48 hours.
Study design: Cluster, randomized-controlled trial.
Setting: ICUs in 27 community and tertiary-care hospitals in Australia and New Zealand.
Synopsis: Hospitals were randomized to intervention or control groups. Dietitian and intensivist co-investigators from intervention ICUs developed guidelines using the Clinical Practice Guideline Development Cycle. Control ICUs were requested to make no new ICU nutritional management changes. The study enrolled 1,118 eligible patients and included adults expected to stay longer than two days. Excluded were patients taking oral diets, patients receiving palliative care or with suspected brain death, and patients sent from other facilities.
Guidelines were implemented using several methods—educational outreach visits, one-on-one conversations, active reminders, passive reminders, and educational in-services. The guidelines were successful in evoking significant practice changes in all of the intervention ICUs. Significantly more patients received nutritional support during their ICU stays in guideline ICUs, and patients in these ICUs were fed significantly earlier. There were, however, no significant differences between guideline and control ICUs with regard to hospital discharge mortality (28.9% vs. 27.4%; 95% CI; -6.3% to 12.0%; P=0.75). The groups also showed no statistical difference in hospital or ICU length of stay.
Bottom line: Significantly more patients in the guideline ICUs were fed within 24 hours, but this did not translate into improvements in mortality or other clinical outcomes.
Citation: Doig G, Simpson F, Finfer S, et al. Effect of evidence-based feeding guidelines on mortality of critically ill adults: a cluster randomized controlled trial. JAMA. 2008;300(23):2731-2741.
Low-Dose Aspirin Doesn’t Reduce Cardiovascular Events in Type 2 Diabetes Patients
Clinical question: Is low-dose aspirin effective for the primary prevention of atherosclerotic events in patients with Type 2 diabetes?
Background: Diabetes is a significant risk factor for cardiovascular events. Several prior studies have shown that aspirin therapy is effective as a secondary prevention strategy for cardiovascular events. The American Diabetes Association also recommends use of aspirin as a primary prevention strategy. Clinical trial data is currently insufficient in this area.
Study design: Prospective, randomized, open-label, controlled trial with blinded endpoint assessment.
Setting: 163 institutions in Japan.
Synopsis: The study enrolled 2,539 diabetic patients between 30 and 85 years old—mean age was 65—and included patients without EKG changes or a significant history or ongoing treatment of atherosclerotic disease. Patients were randomly assigned into an aspirin group (81 mg or 100 mg once daily) or a nonaspirin group. Patients had a median follow up of 4.37 years.
The primary endpoint was any atherosclerotic event, ranging from sudden death to ischemic heart disease or stroke. The incidence of atherosclerotic events was not statistically different in the aspirin group (68 events, 5.4%) than in the nonaspirin group (86 events, 6.7%) (HR, 0.80; 95% CI; 0.58-1.10; log-rank test, P=0.16). However, there was a suggested benefit of primary prevention in the subgroup aged 65 years or older. In addition, the combined endpoint of fatal coronary and cerebrovascular events occurred in one patient in the aspirin group and 10 patients in the nonaspirin group (HR, 0.10; 95 % CI, 0.01-0.79; P=.0037). This study is limited by the low incidence of atherosclerotic disease in Japan.
Bottom line: Low-dose aspirin used in patients with Type 2 diabetes does not significantly demonstrate primary prevention of cardiovascular events.
Citation: Ogawa H, Nakayama M, Morimoto T, et al. Low-dose aspirin for primary prevention of atherosclerotic events in patients with type 2 diabetes: a randomized controlled trial. JAMA. 2008;300(18):2134-2141. TH
In This Edition
- Generic vs. brand-name drugs.
- Rapid-response teams and mortality.
- A new prediction rule for mortality in acute pancreatitis.
- Viral causes of community-acquired pneumonia.
- Intensive insulin therapy in the ICU.
- New preoperative and intraoperative risk factors.
- Timing of ICU feedings and mortality.
- Aspirin as primary prevention in diabetics.
Generic, Brand-Name Drugs Used for Cardiovascular Disease Are Clinically Equivalent
Clinical question: Is there a clinical risk when substituting generic drugs for brand-name drugs in the treatment of cardiovascular disease?
Background: Spending on healthcare in the U.S. has reached critical levels. Increasing prescription drug costs make up a large portion of healthcare expenditures. The high cost of medicines directly affect adherence to treatment regimens and contribute to poor health outcomes. Cardiovascular drugs make up the largest portion of outpatient prescription drug spending.
Study design: Systematic review of relevant articles with a meta-analysis performed to determine an aggregate effect size.
Setting: Multiple locations and varied patient populations.
Synopsis: A total of 47 articles were included in the review, of which 38 were randomized controlled trials (RCTs). The studies measured both clinical efficacy and safety end points. More than half the articles were published prior to 2000. Clinical equivalence was noted in all seven beta-blocker RCTs; 10 of 11 diuretic RCTs; five of seven calcium-channel-blocker RCTs; all three antiplatelet-agent RCTs (clopidogrel, enteric-coated aspirin); two statin RCTs; one ACE-inhibitor RCT; and one alpha-blocker RCT. For drugs with a narrow therapeutic index, clinical equivalence was noted in all five warfarin RCTs and a single Class 1 anti-arrhythmic-agent RCT.
The aggregate effect size was -0.03 (95% CI, -0.15 to 0.08), which indicates nearly complete overlap of the generic and brand-name distributions. The data show no evidence of superiority of brand-name to generic drugs in clinical outcomes measured in the various studies.
In a separate review of editorials addressing generic substitution for cardiovascular drugs, 53% expressed a negative view of generic-drug substitution.
Bottom line: There is clinical equivalency between generic and brand-name drugs used in the treatment of cardiovascular disease. Despite this conclusion, a substantial number of editorials advise against generic substitution, which affects both patient and physician drug preferences.
Citation: Kesselheim A, Misono A, Lee J, et al. Clinical equivalence of generic and brand-name drugs used in cardiovascular disease: a systematic review and meta-analysis. JAMA. 2008; 300(21):2514-2526.
RRT Implement-ation Doesn’t Affect Hospitalwide Code Rates or Mortality
Clinical question: Does the use of a rapid-response team (RRT) affect hospitalwide code rates and mortality?
Background: In the 100,000 Lives campaign, the Institute for Healthcare Improvement recommended that hospitals implement RRTs to help reduce preventable, in-hospital deaths. Studies have demonstrated that rates of non-ICU codes decrease after RRT implementation. It is unclear if this translates to changes in mortality rates.
Study design: Prospective cohort study of adult inpatients.
Setting: Saint Luke’s Hospital, a 404-bed tertiary-care academic hospital in Kansas City, Mo.
Synopsis: The hospital documented a total of 376 RRT activations. After RRT implementation, mean hospitalwide code rates decreased to 7.5 per 1,000 admissions from 11.2 per 1,000 admissions. This was not associated with a statistically significant reduction in hospitalwide code rates (adjusted odds ratio 0.76; 95% CI; 0.57-1.01; P=0.06). Secondary analyses noted lower rates of non-ICU codes (0.59; 95% CI, 0.40-0.89) compared with ICU codes (0.95; 95% CI; 0.64-1.43; P=0.03 for interaction). Finally, the RRT implementation was not associated with lower hospital-wide mortality (0.95; 95% CI; 0.81-1.11; P=0.52).
Secondary analyses also revealed few instances of RRT undertreatment or underutilization that may have affected the mortality numbers.
A limitation of this study is that it was slightly underpowered (78%) to detect a significant mortality difference. The findings also represent a single institution experience, and may not be generalized to other adult hospital settings or RRT programs.
Bottom line: Implementation of an RRT does not confer lower rates of hospital-wide code arrests or mortality.
Citation: Chan P, Khalid A, Longmore L, et al. Hospital-wide code rates and mortality before and after implementation of a rapid response team. JAMA. 2008;300(21):2506-2513.
Simple Scoring System Provides Timely Prediction of Mortality in Acute Pancreatitis
Clinical question: How can physicians predict mortality in acute pancreatitis?
Background: Historical predictors of mortality in acute pancreatitis require up to 48 hours of data, such as with the Ranson Criteria, or extensive amounts of data, such as with the APACHE II score. An easier tool is needed to predict which patients are at higher risk of mortality.
Study design: Retrospective cohort.
Setting: Patients in the Cardinal Health clinical outcomes research database, which supports public reporting of hospital performance.
Synopsis: The authors identified patients with the principal diagnosis of pancreatitis from 2000-2001 and explored numerous diagnostic findings available within the first 24 hours. Ultimately, BUN >25, impaired mental status, presence of SIRS (systemic inflammatory response syndrome), age >60, and presence of a pleural effusion were found to be predictive of mortality. These diagnostic findings correspond to the mnemonic BISAP. The BISAP score was then validated in a second cohort that included patients from 2004-2005.
Each finding in the BISAP score was given one point. A score of less than 2 was present in approximately 60% of patients admitted with acute pancreatitis, and corresponded to a mortality of less than 1%. A score of 2 corresponded to a mortality of 2%. Higher scores were associated with steeply increasing mortality, with a score of 5 corresponding with greater than 20% mortality.
The BISAP score performed similarly to the APACHE II score, but the former is easier to calculate on the day of admission and has fewer parameters. A more challenging research step will be to demonstrate that using the BISAP score to determine treatment strategies can affect patient outcomes.
Bottom line: The easy-to-calculate BISAP score is a new method for predicting mortality in patients with acute pancreatitis. This tool should help hospitalists determine, on the day of admission, to what extent patients with acute pancreatitis need aggressive management, such as ICU admission and early involvement of specialists.
Citation: Wu B, Johannes R, Sun X, Tabak Y, Conwell D, Banks P. The early prediction of mortality in acute pancreatitis: a large population-based study. Gut. 2008;57(12): 1698-1703.
Nasal Swabs Identify Viral Causes in CAP Patients
Clinical question: How often is viral infection associated with community-acquired pneumonia (CAP) in adults?
Background: CAP is a clinically important disease that is associated with significant hospitalization and mortality. CAP treatment guidelines acknowledge respiratory viruses as an etiology for pneumonia, but few recommendations are made regarding management of these viral infections.
Study design: Prospective study.
Setting: Five hospitals in Edmonton, Alberta, from 2004-2006.
Synopsis: The authors enrolled 193 hospitalized adults, median age 71. Nucleic amplification tests (NATs) from nasopharyngeal swab specimens were tested for human metapneumovirus, respiratory syncytial virus, rhinovirus, parainfluenza virus 1-4, coronaviruses, and adenovirus.
Fifteen percent of these patients had a nasal swab positive for a viral infection. Interestingly, 4% of patients had both a positive viral nasal swab and positive bacterial cultures. Compared with bacterial infection, patients with viral infection tended to be older (76 vs. 64 years, P=0.01), have limited ambulation (66% vs. 32%, P=0.006), and have a history of cardiac disease (66% vs. 32%, P=0.02). Patients with bacterial pneumonia showed a statistically significant trend toward having chest pain, an abnormal white blood count, and a lobar infiltrate on chest X-ray.
Further investigations might look at how nasal swab data could be used to improve infection control within the hospital for patients found to have easily transmissible viruses. Further research could explore the feasibility of avoiding antibiotic use in patients found to have viral pneumonia, assuming bacterial co-infection is reliably excluded.
Bottom line: Nasal swabs using NAT technology could play a significant role in identifying pathogens in CAP patients. How this technology should affect clinical decision-making and how it might improve outcomes remains unknown.
Citation: Johnstone J, Majumdar S, Fox J, Marrie T. Viral infection in adults hospitalized with community-acquired pneumonia: prevalence, pathogens, and presentation. Chest. 2008;134(6):1141-1148.
Intensive Insulin Therapy Doesn’t Reduce ICU Mortality
Clinical question: Does the use of intensive insulin therapy decrease mortality in the intensive-care unit (ICU)?
Background: In 2001, Van den Berghe et al (N Engl J Med. 2001;345(19):1359-67) reported a reduction in morbidity and mortality with intensive insulin therapy (IIT) in surgical ICU patients. This study led to the adoption of IIT protocols in many hospitals. Since 2001, further studies have failed to reproduce the same dramatic benefit of IIT.
Study design: Randomized, controlled trial.
Setting: National Guard King Abdulaziz Medical City, a tertiary-care teaching hospital in Saudi Arabia.
Synopsis: Patients were included in this study if they were 18 years or older with serum glucose levels greater than 110 mg/dL during the first 24 hours of ICU admission. There were multiple exclusion criteria, including patients with Type I diabetes, documented hypoglycemia on ICU admission (glucose <40), or diabetic ketoacidosis.
Enrolled patients were randomized to IIT or conventional insulin therapy (CIT). A multidisciplinary team designed the protocols to maintain glucose levels of 80 to 110 mg/dL and 180 to 200 mg/dL in the IIT and the CIT groups, respectively. The primary endpoint measured was ICU mortality.
The study did not produce a statistically significant difference in ICU mortality (13.5% for IIT vs. 17.1% for CIT; P=0.30). The adjusted hypoglycemia rate was 6.8 per 100 treatment days with IIT and 0.4 per 100 treatment days with CIT (P<0.0001). Patients with hypoglycemia had higher ICU mortality (23.8% vs. 13.7%, P=0.02).
In the measurement of secondary endpoints, there was a trend toward lower episodes of severe sepsis and septic shock in the IIT group (20.7% in IIT vs. 27.2% in CIT, P=0.08). However, this result was not statistically significant.
Bottom line: This well-designed study failed to show a survival benefit with IIT use in the critical-care setting. Given the findings of this and several other recent studies, one should question whether IIT should be prescribed as the standard of care in all critically-ill patients.
Citation: Arabi Y, Dabbagh O, Tamim H, et al. Intensive versus conventional insulin therapy: a randomized controlled trial in medical and surgical critically ill patients. Crit Care Med. 2008;36(12):3190-3197.
Preoperative and Intraoperative Predictors of Cardiac Adverse Events
Clinical question: What are the incidence and risk factors for perioperative cardiac adverse events after noncardiac surgery?
Background: In the past few decades, the incidence of cardiac adverse events (CAEs) for a general surgery population has remained largely unchanged (approximately 1%). This is in spite of multiple studies evaluating predictive models and attempts at preventive treatment, including preoperative coronary revascularization and perioperative beta blockers.
Study design: Prospective observational study.
Setting: Single, large, tertiary-care university hospital.
Synopsis: A total of 7,740 cases were reviewed in this study, which consisted of general surgery (4,937), vascular surgery (1,846), and urological surgery (957). A trained nurse followed up for perioperative CAEs as many as 30 days after the operation via medical chart review, phone calls, and letters. CAEs were defined as: Q-wave myocardial infarction (MI), non-ST elevation MI, cardiac arrest, or new cardiac dysrhythmia. A total of 83 CAEs (1.1% of patients) had cardiac arrest, with cardiac dysrhythmia being most common.
A total of seven preoperative risk factors were identified as independent predictors for CAEs: age 68, BMI 30, emergent surgery, prior coronary intervention or cardiac surgery, active congestive heart failure, cerebrovascular disease, and hypertension. In addition, two intraoperative risk factors were identified: intraoperative transfusion of packed red blood cells and operative duration of 3.8 hours. (The P value was 0.05 for all independent predictors.)
A further evaluation of intraoperative parameters of high-risk patients experiencing a CAE showed that these patients were more likely to have an episode of mean arterial pressure (MAP) <50 mmHg, an episode of 40% decrease in MAP, and an episode of tachycardia (heart rate >100).
Bottom line: A combination of preoperative predictors and intraoperative elements can help improve risk assessment for perioperative CAEs after noncardiac surgery.
Citation: Kheterpal S, O’Reilly M, Englesbe M, et al. Preoperative and intraoperative predictors of cardiac adverse events after general, vascular, and urological surgery. Anesthesiology. 2009;110(1):58-66.
Early Feeding in the ICU Does Not Affect Hospital Mortality
Clinical question: Will implementing evidence-based feeding guidelines improve feeding practices and reduce mortality in ICU patients?
Background: There is evidence suggesting that providing nutritional support to ICU patients within 24 hours of admission may decrease mortality. It is widely understood that patient care varies between ICUs, and there exist no uniform, evidence-based guidelines for nutritional support. Many patients remain unfed after 48 hours.
Study design: Cluster, randomized-controlled trial.
Setting: ICUs in 27 community and tertiary-care hospitals in Australia and New Zealand.
Synopsis: Hospitals were randomized to intervention or control groups. Dietitian and intensivist co-investigators from intervention ICUs developed guidelines using the Clinical Practice Guideline Development Cycle. Control ICUs were requested to make no new ICU nutritional management changes. The study enrolled 1,118 eligible patients and included adults expected to stay longer than two days. Excluded were patients taking oral diets, patients receiving palliative care or with suspected brain death, and patients sent from other facilities.
Guidelines were implemented using several methods—educational outreach visits, one-on-one conversations, active reminders, passive reminders, and educational in-services. The guidelines were successful in evoking significant practice changes in all of the intervention ICUs. Significantly more patients received nutritional support during their ICU stays in guideline ICUs, and patients in these ICUs were fed significantly earlier. There were, however, no significant differences between guideline and control ICUs with regard to hospital discharge mortality (28.9% vs. 27.4%; 95% CI; -6.3% to 12.0%; P=0.75). The groups also showed no statistical difference in hospital or ICU length of stay.
Bottom line: Significantly more patients in the guideline ICUs were fed within 24 hours, but this did not translate into improvements in mortality or other clinical outcomes.
Citation: Doig G, Simpson F, Finfer S, et al. Effect of evidence-based feeding guidelines on mortality of critically ill adults: a cluster randomized controlled trial. JAMA. 2008;300(23):2731-2741.
Low-Dose Aspirin Doesn’t Reduce Cardiovascular Events in Type 2 Diabetes Patients
Clinical question: Is low-dose aspirin effective for the primary prevention of atherosclerotic events in patients with Type 2 diabetes?
Background: Diabetes is a significant risk factor for cardiovascular events. Several prior studies have shown that aspirin therapy is effective as a secondary prevention strategy for cardiovascular events. The American Diabetes Association also recommends use of aspirin as a primary prevention strategy. Clinical trial data is currently insufficient in this area.
Study design: Prospective, randomized, open-label, controlled trial with blinded endpoint assessment.
Setting: 163 institutions in Japan.
Synopsis: The study enrolled 2,539 diabetic patients between 30 and 85 years old—mean age was 65—and included patients without EKG changes or a significant history or ongoing treatment of atherosclerotic disease. Patients were randomly assigned into an aspirin group (81 mg or 100 mg once daily) or a nonaspirin group. Patients had a median follow up of 4.37 years.
The primary endpoint was any atherosclerotic event, ranging from sudden death to ischemic heart disease or stroke. The incidence of atherosclerotic events was not statistically different in the aspirin group (68 events, 5.4%) than in the nonaspirin group (86 events, 6.7%) (HR, 0.80; 95% CI; 0.58-1.10; log-rank test, P=0.16). However, there was a suggested benefit of primary prevention in the subgroup aged 65 years or older. In addition, the combined endpoint of fatal coronary and cerebrovascular events occurred in one patient in the aspirin group and 10 patients in the nonaspirin group (HR, 0.10; 95 % CI, 0.01-0.79; P=.0037). This study is limited by the low incidence of atherosclerotic disease in Japan.
Bottom line: Low-dose aspirin used in patients with Type 2 diabetes does not significantly demonstrate primary prevention of cardiovascular events.
Citation: Ogawa H, Nakayama M, Morimoto T, et al. Low-dose aspirin for primary prevention of atherosclerotic events in patients with type 2 diabetes: a randomized controlled trial. JAMA. 2008;300(18):2134-2141. TH
See You in Chicago
Having grown up in the shadows of the Windy City, I always wanted to work in the Sears Tower, jog along Lake Shore Drive, catch a comedy show at the Chicago Theater, and—just like every other 11-year-old boy—hop the subway to Wrigley Field for Chicago Cubs’ day games. It’s been nearly two decades since I left Chicago’s suburbs, yet I still refer to the Windy City as my home.
It’s safe to say I’m a little excited about attending my first annual meeting in my favorite U.S. city. An expanded HM09 runs May 14-17 at the Hyatt Regency in downtown Chicago. The 2,000-room base hotel is the perfect location for learning and leisure. It’s just a stone’s throw from Lake Michigan, Navy Pier, museums, great restaurants, and shopping. If you weren’t able to make it to HM05 in Chicago—or aren’t familiar with the stretch of Michigan Avenue known as the Magnificent Mile—it has something for everyone, and it’s easily accessible on foot or by taxi, subway, or horse-drawn carriage.
I know times are tough, but if you can, plan to spend an extra day or two and soak up all that Chicago has to offer. May is a great time to visit Chicago: The temperature should be in the 70s, and thousands of tulips will be in full bloom. The Art Institute of Chicago is opening a new wing and admission will be free May 16-22; the 11th annual Great Chicago Places & Spaces festival is May 16 and offers hundreds of free architectural tours; and Lincoln Park Zoo (a 15-minute cab ride from downtown) is celebrating Bear Awareness Week. For you green thumbs, the Chicago Farmers Market, which offers fresh fruit, vegetables, and flowers, is right around the corner from the Hyatt at Daley Plaza. Feeling ambitious? You can embark on a tour of President Obama’s Chicago; just follow the user-friendly map at www.chicagotribune.com/travel/chi-obama-chicago-htmlstory,0,506256.htmlstory.
Chicago’s nightlife offers something for every taste—beer gardens, IMAX theaters, comedy shows, and live music of all genres. If you’re a sports fan, the Cubs will be in town, and the resurgent Blackhawks could be in the NHL playoffs. If you are looking for a sports bar to watch your favorite team, look no further than Houlihan’s (right next door to the hotel) and Mother Hubbard’s (two blocks north of the hotel).
Dare I forget to mention the main reason HM09 attracts thousands of the nation’s top hospitalists—a world-class continuing medical education lineup, first-rate faculty, and endless networking opportunities? The addition of a fourth meeting day dedicated solely to clinical and practice management precourses not only allows for a less-hectic meeting schedule, but it also cements HM09 as a forward-thinking, education-focused annual conference.
In addition to a pair of powerhouse keynote speakers and the annual Awards of Excellence, SHM will induct its first class of Fellows in Hospital Medicine at HM09. Built using SHM’s Core Competencies, the FHM designation recognizes individuals who have chosen HM as a career and have the credentials to represent the emerging specialty. SHM received more than 600 applications for the FHM designation, and more than 400 hospitalists will be inducted in the first class.
It hardly seems possible, but HM09 is right around the corner. The editorial team here at The Hospitalist has a few aces up our sleeves, too. More on those in the May issue! Also, don’t forget to stop by The Hospitalist booth (listed in the program as the Wiley-Blackwell booth) to introduce yourself, exchange business cards, sign up for a cool prize, and share your ideas on the future of HM. TH
Jason Carris is editor of The Hospitalist.
Having grown up in the shadows of the Windy City, I always wanted to work in the Sears Tower, jog along Lake Shore Drive, catch a comedy show at the Chicago Theater, and—just like every other 11-year-old boy—hop the subway to Wrigley Field for Chicago Cubs’ day games. It’s been nearly two decades since I left Chicago’s suburbs, yet I still refer to the Windy City as my home.
It’s safe to say I’m a little excited about attending my first annual meeting in my favorite U.S. city. An expanded HM09 runs May 14-17 at the Hyatt Regency in downtown Chicago. The 2,000-room base hotel is the perfect location for learning and leisure. It’s just a stone’s throw from Lake Michigan, Navy Pier, museums, great restaurants, and shopping. If you weren’t able to make it to HM05 in Chicago—or aren’t familiar with the stretch of Michigan Avenue known as the Magnificent Mile—it has something for everyone, and it’s easily accessible on foot or by taxi, subway, or horse-drawn carriage.
I know times are tough, but if you can, plan to spend an extra day or two and soak up all that Chicago has to offer. May is a great time to visit Chicago: The temperature should be in the 70s, and thousands of tulips will be in full bloom. The Art Institute of Chicago is opening a new wing and admission will be free May 16-22; the 11th annual Great Chicago Places & Spaces festival is May 16 and offers hundreds of free architectural tours; and Lincoln Park Zoo (a 15-minute cab ride from downtown) is celebrating Bear Awareness Week. For you green thumbs, the Chicago Farmers Market, which offers fresh fruit, vegetables, and flowers, is right around the corner from the Hyatt at Daley Plaza. Feeling ambitious? You can embark on a tour of President Obama’s Chicago; just follow the user-friendly map at www.chicagotribune.com/travel/chi-obama-chicago-htmlstory,0,506256.htmlstory.
Chicago’s nightlife offers something for every taste—beer gardens, IMAX theaters, comedy shows, and live music of all genres. If you’re a sports fan, the Cubs will be in town, and the resurgent Blackhawks could be in the NHL playoffs. If you are looking for a sports bar to watch your favorite team, look no further than Houlihan’s (right next door to the hotel) and Mother Hubbard’s (two blocks north of the hotel).
Dare I forget to mention the main reason HM09 attracts thousands of the nation’s top hospitalists—a world-class continuing medical education lineup, first-rate faculty, and endless networking opportunities? The addition of a fourth meeting day dedicated solely to clinical and practice management precourses not only allows for a less-hectic meeting schedule, but it also cements HM09 as a forward-thinking, education-focused annual conference.
In addition to a pair of powerhouse keynote speakers and the annual Awards of Excellence, SHM will induct its first class of Fellows in Hospital Medicine at HM09. Built using SHM’s Core Competencies, the FHM designation recognizes individuals who have chosen HM as a career and have the credentials to represent the emerging specialty. SHM received more than 600 applications for the FHM designation, and more than 400 hospitalists will be inducted in the first class.
It hardly seems possible, but HM09 is right around the corner. The editorial team here at The Hospitalist has a few aces up our sleeves, too. More on those in the May issue! Also, don’t forget to stop by The Hospitalist booth (listed in the program as the Wiley-Blackwell booth) to introduce yourself, exchange business cards, sign up for a cool prize, and share your ideas on the future of HM. TH
Jason Carris is editor of The Hospitalist.
Having grown up in the shadows of the Windy City, I always wanted to work in the Sears Tower, jog along Lake Shore Drive, catch a comedy show at the Chicago Theater, and—just like every other 11-year-old boy—hop the subway to Wrigley Field for Chicago Cubs’ day games. It’s been nearly two decades since I left Chicago’s suburbs, yet I still refer to the Windy City as my home.
It’s safe to say I’m a little excited about attending my first annual meeting in my favorite U.S. city. An expanded HM09 runs May 14-17 at the Hyatt Regency in downtown Chicago. The 2,000-room base hotel is the perfect location for learning and leisure. It’s just a stone’s throw from Lake Michigan, Navy Pier, museums, great restaurants, and shopping. If you weren’t able to make it to HM05 in Chicago—or aren’t familiar with the stretch of Michigan Avenue known as the Magnificent Mile—it has something for everyone, and it’s easily accessible on foot or by taxi, subway, or horse-drawn carriage.
I know times are tough, but if you can, plan to spend an extra day or two and soak up all that Chicago has to offer. May is a great time to visit Chicago: The temperature should be in the 70s, and thousands of tulips will be in full bloom. The Art Institute of Chicago is opening a new wing and admission will be free May 16-22; the 11th annual Great Chicago Places & Spaces festival is May 16 and offers hundreds of free architectural tours; and Lincoln Park Zoo (a 15-minute cab ride from downtown) is celebrating Bear Awareness Week. For you green thumbs, the Chicago Farmers Market, which offers fresh fruit, vegetables, and flowers, is right around the corner from the Hyatt at Daley Plaza. Feeling ambitious? You can embark on a tour of President Obama’s Chicago; just follow the user-friendly map at www.chicagotribune.com/travel/chi-obama-chicago-htmlstory,0,506256.htmlstory.
Chicago’s nightlife offers something for every taste—beer gardens, IMAX theaters, comedy shows, and live music of all genres. If you’re a sports fan, the Cubs will be in town, and the resurgent Blackhawks could be in the NHL playoffs. If you are looking for a sports bar to watch your favorite team, look no further than Houlihan’s (right next door to the hotel) and Mother Hubbard’s (two blocks north of the hotel).
Dare I forget to mention the main reason HM09 attracts thousands of the nation’s top hospitalists—a world-class continuing medical education lineup, first-rate faculty, and endless networking opportunities? The addition of a fourth meeting day dedicated solely to clinical and practice management precourses not only allows for a less-hectic meeting schedule, but it also cements HM09 as a forward-thinking, education-focused annual conference.
In addition to a pair of powerhouse keynote speakers and the annual Awards of Excellence, SHM will induct its first class of Fellows in Hospital Medicine at HM09. Built using SHM’s Core Competencies, the FHM designation recognizes individuals who have chosen HM as a career and have the credentials to represent the emerging specialty. SHM received more than 600 applications for the FHM designation, and more than 400 hospitalists will be inducted in the first class.
It hardly seems possible, but HM09 is right around the corner. The editorial team here at The Hospitalist has a few aces up our sleeves, too. More on those in the May issue! Also, don’t forget to stop by The Hospitalist booth (listed in the program as the Wiley-Blackwell booth) to introduce yourself, exchange business cards, sign up for a cool prize, and share your ideas on the future of HM. TH
Jason Carris is editor of The Hospitalist.
Telemedicine Can Help Solve Intensivist Shortage
Having spent my medical career in the ICU and the hospital, I have followed the recent articles on the struggle to care for ICU patients with interest. Gretchen Henkel’s article on hospitalists filling ICU manpower gaps (“The New Intensivists,” October 2008, p. 1) poses a very real question for community hospitals, which face the greatest challenges in this area. Two issues are common: 1) difficulty in providing 24/7 ICU coverage and 2) the competing priorities that ICU medical leaders face. For these challenges, telemedicine offers a possible solution.
This unique, high-intensity, multidisciplinary approach to the patient population—an integral part of intensivist training—is a proven process shown to have meaningful results in the ICU. However, the team approach to managing ICU patients can be hard to come by. As suggested in Henkel’s article, there are several ways to approach this, but simply having a hospitalist consult on an ICU patient is not, I believe, a solution. Not only can this add to the strain on a hospitalist team, but a proactive approach to the ICU patient also can be hampered by the need for hospitalists to be present in the medical-surgical areas. Ideally, an intensivist should lead a multiprofessional team; however, there is a tremendous intensivist shortage, with less than 20% of ICUs staffed with them. Telemedicine offers a way to bridge the gap of expertise and manpower in many settings, bringing intensivists to the forefront of the ICU multidisciplinary team.
For the multidisciplinary approach to be effective, a physician must be committed to creating the team and identifying the measures that it will impact. In many community hospitals, this is the ICU medical director. However, competing priorities can make this directive difficult to achieve. Teleintensivists, intensivists that practice medicine via telemedicine, proactively establish best practices and a multidisciplinary approach, thus dramatically affecting the quality and financial metrics of the ICU.
Lack of 24/7 ICU coverage is another big challenge for community hospitals. Trying to meet this challenge by simply adding intensivists is likely to be met with defeat, given the shortage of hospitalists. And adding more hospitalists in the ICU continues to drive the hospitalist shortage. There is a variety of solutions for bedside procedures; however, the constant need to respond to phone calls and unpredictable patient interventions remains. While utilizing midlevel providers can help, this approach is not likely to support the demand of the aging population.
Hospitals increasingly are considering telemedicine to meet the 24/7 need. Teleintensivists have risen to add manpower and immediate response to ICU patients. Without the distraction of constant interruptions and with a process to manage the deluge of data, community hospitals with teleintensivist programs are seeing a drop in ventilator-associated pneumonia, better blood glucose management, and compliance with sepsis and other bundles.
This proactive approach to ICU patient care has led to significant decreases in mortality and lengths of stay.
A recent article in The New York Times focused on “disruptive innovation” in healthcare.1 Given the pressing issues facing the industry, disruptive innovation―at the bedside as well as with telemedicine technology―will be a key factor in meeting our ICU needs successfully.
Mary Jo Gorman, MD, MBA
Editor’s note: Dr. Gorman, a former SHM president, is the CEO of St. Louis-based Advanced ICU Care, which provides intensivists to community hospitals using telemedicine. TH
Reference
1. Rae-Dupree J. Disruptive innovation, applied to health care. The New York Times Web site. Available at: www.nytimes.com/2009/02/01/business/01unbox.html?scp=1&sq=disruptive%20innovation&st=cse. Accessed March 3, 2009.
Having spent my medical career in the ICU and the hospital, I have followed the recent articles on the struggle to care for ICU patients with interest. Gretchen Henkel’s article on hospitalists filling ICU manpower gaps (“The New Intensivists,” October 2008, p. 1) poses a very real question for community hospitals, which face the greatest challenges in this area. Two issues are common: 1) difficulty in providing 24/7 ICU coverage and 2) the competing priorities that ICU medical leaders face. For these challenges, telemedicine offers a possible solution.
This unique, high-intensity, multidisciplinary approach to the patient population—an integral part of intensivist training—is a proven process shown to have meaningful results in the ICU. However, the team approach to managing ICU patients can be hard to come by. As suggested in Henkel’s article, there are several ways to approach this, but simply having a hospitalist consult on an ICU patient is not, I believe, a solution. Not only can this add to the strain on a hospitalist team, but a proactive approach to the ICU patient also can be hampered by the need for hospitalists to be present in the medical-surgical areas. Ideally, an intensivist should lead a multiprofessional team; however, there is a tremendous intensivist shortage, with less than 20% of ICUs staffed with them. Telemedicine offers a way to bridge the gap of expertise and manpower in many settings, bringing intensivists to the forefront of the ICU multidisciplinary team.
For the multidisciplinary approach to be effective, a physician must be committed to creating the team and identifying the measures that it will impact. In many community hospitals, this is the ICU medical director. However, competing priorities can make this directive difficult to achieve. Teleintensivists, intensivists that practice medicine via telemedicine, proactively establish best practices and a multidisciplinary approach, thus dramatically affecting the quality and financial metrics of the ICU.
Lack of 24/7 ICU coverage is another big challenge for community hospitals. Trying to meet this challenge by simply adding intensivists is likely to be met with defeat, given the shortage of hospitalists. And adding more hospitalists in the ICU continues to drive the hospitalist shortage. There is a variety of solutions for bedside procedures; however, the constant need to respond to phone calls and unpredictable patient interventions remains. While utilizing midlevel providers can help, this approach is not likely to support the demand of the aging population.
Hospitals increasingly are considering telemedicine to meet the 24/7 need. Teleintensivists have risen to add manpower and immediate response to ICU patients. Without the distraction of constant interruptions and with a process to manage the deluge of data, community hospitals with teleintensivist programs are seeing a drop in ventilator-associated pneumonia, better blood glucose management, and compliance with sepsis and other bundles.
This proactive approach to ICU patient care has led to significant decreases in mortality and lengths of stay.
A recent article in The New York Times focused on “disruptive innovation” in healthcare.1 Given the pressing issues facing the industry, disruptive innovation―at the bedside as well as with telemedicine technology―will be a key factor in meeting our ICU needs successfully.
Mary Jo Gorman, MD, MBA
Editor’s note: Dr. Gorman, a former SHM president, is the CEO of St. Louis-based Advanced ICU Care, which provides intensivists to community hospitals using telemedicine. TH
Reference
1. Rae-Dupree J. Disruptive innovation, applied to health care. The New York Times Web site. Available at: www.nytimes.com/2009/02/01/business/01unbox.html?scp=1&sq=disruptive%20innovation&st=cse. Accessed March 3, 2009.
Having spent my medical career in the ICU and the hospital, I have followed the recent articles on the struggle to care for ICU patients with interest. Gretchen Henkel’s article on hospitalists filling ICU manpower gaps (“The New Intensivists,” October 2008, p. 1) poses a very real question for community hospitals, which face the greatest challenges in this area. Two issues are common: 1) difficulty in providing 24/7 ICU coverage and 2) the competing priorities that ICU medical leaders face. For these challenges, telemedicine offers a possible solution.
This unique, high-intensity, multidisciplinary approach to the patient population—an integral part of intensivist training—is a proven process shown to have meaningful results in the ICU. However, the team approach to managing ICU patients can be hard to come by. As suggested in Henkel’s article, there are several ways to approach this, but simply having a hospitalist consult on an ICU patient is not, I believe, a solution. Not only can this add to the strain on a hospitalist team, but a proactive approach to the ICU patient also can be hampered by the need for hospitalists to be present in the medical-surgical areas. Ideally, an intensivist should lead a multiprofessional team; however, there is a tremendous intensivist shortage, with less than 20% of ICUs staffed with them. Telemedicine offers a way to bridge the gap of expertise and manpower in many settings, bringing intensivists to the forefront of the ICU multidisciplinary team.
For the multidisciplinary approach to be effective, a physician must be committed to creating the team and identifying the measures that it will impact. In many community hospitals, this is the ICU medical director. However, competing priorities can make this directive difficult to achieve. Teleintensivists, intensivists that practice medicine via telemedicine, proactively establish best practices and a multidisciplinary approach, thus dramatically affecting the quality and financial metrics of the ICU.
Lack of 24/7 ICU coverage is another big challenge for community hospitals. Trying to meet this challenge by simply adding intensivists is likely to be met with defeat, given the shortage of hospitalists. And adding more hospitalists in the ICU continues to drive the hospitalist shortage. There is a variety of solutions for bedside procedures; however, the constant need to respond to phone calls and unpredictable patient interventions remains. While utilizing midlevel providers can help, this approach is not likely to support the demand of the aging population.
Hospitals increasingly are considering telemedicine to meet the 24/7 need. Teleintensivists have risen to add manpower and immediate response to ICU patients. Without the distraction of constant interruptions and with a process to manage the deluge of data, community hospitals with teleintensivist programs are seeing a drop in ventilator-associated pneumonia, better blood glucose management, and compliance with sepsis and other bundles.
This proactive approach to ICU patient care has led to significant decreases in mortality and lengths of stay.
A recent article in The New York Times focused on “disruptive innovation” in healthcare.1 Given the pressing issues facing the industry, disruptive innovation―at the bedside as well as with telemedicine technology―will be a key factor in meeting our ICU needs successfully.
Mary Jo Gorman, MD, MBA
Editor’s note: Dr. Gorman, a former SHM president, is the CEO of St. Louis-based Advanced ICU Care, which provides intensivists to community hospitals using telemedicine. TH
Reference
1. Rae-Dupree J. Disruptive innovation, applied to health care. The New York Times Web site. Available at: www.nytimes.com/2009/02/01/business/01unbox.html?scp=1&sq=disruptive%20innovation&st=cse. Accessed March 3, 2009.
Non-Physician Providers: Vital HM Resources
Enter text here
Enter text here
Enter text here
Project BOOST Expands
SHM’s Project BOOST (Better Outcomes for Older Adults through Safe Transitions) is an initiative to improve practices in transition care and reduce readmission rates for hospitals across the country. The project’s toolkit, mentoring program, and national advocacy efforts have proven so successful that the program is expanding this year.
In 2008, SHM began the first round of the Project BOOST mentoring program in six pilot hospitals. The first full cycle of Project BOOST mentoring sites began in March at 24 sites. The Hospitalist will feature updates on the full cycle of Project BOOST later this year. For more information about Project BOOST, visit www.hospitalmedicine.org/BOOST or e-mail BOOST@hospitalmedicine.org.
As a pilot site, Southwestern Vermont Medical Center in Bennington has worked with mentors for the past six months. We caught up with project leader Jennifer Fells, RN, MS, to discuss the institution’s participation.
—Jennifer Fells, RN, MS, Southwestern Vermont Medical Center, Bennington
Question: Why did your group choose to participate in the mentoring program?
Answer: We wanted to reduce our readmission rate, and we knew we weren’t doing a service to patients. This was also a goal of our organization overall; it’s not only a benefit to the hospital, but a larger value when the patient goes back to the community.
Q: How has the BOOST mentoring program benefited your program?
A: It helped us get organized by beginning the process and affirmed our belief that there were ways to address readmissions. The toolkit has proved to be invaluable. The mentors helped us keep on track and offered us guidance. They share the experiences of the other Project BOOST teams, and we benefit from that information.
The changes in the approach to the discharge process were a surprise to us. After we developed our team, we discovered how fragmented the discharge process was throughout the entire organization. Discharges were handled over multiple disciplines, and it was fragmented by design.
For example, our documentation, discharge plans, discharge recommendations, and patient-education materials are in different parts of our documentation system.
Q: What did you learn about your program through the initial Project BOOST step: analyze care delivery?
A: We realized that disciplines were not coordinated with one another and there was not enough time for the physician to complete the discharge plan. The process was cumbersome, awkward, and very time-consuming. We were looking to create efficiency in the information that needs to be coordinated to do those discharge orders.
Q: What additional changes do you hope to see in the remaining time with BOOST mentors?
A: We definitely hope to improve our process. We want to have implemented the tools and have a coordinated discharge process, and a centralized way to communicate the discharge plan. We found a lack of communication creates a barrier among disciplines, and we hope to correct that and become more customer-friendly to patients.
Q: How did your site’s BOOST mentor assist in the implementation process? What was the outcome?
A: Our site mentor helped with the clarification of data to be collected for measurement, keeping us on track with the toolkit, and served as another set of eyes. When you are in an organization, and even when you have the appropriate team, you always need somebody to say, “What do you think about this?” or “Did you think about this item?”
It’s another perspective, sharing gained knowledge from other organizations. That’s very critical.
SHM marketing coordinator Nadia Clenending contributed to this report.
SHM’s Project BOOST (Better Outcomes for Older Adults through Safe Transitions) is an initiative to improve practices in transition care and reduce readmission rates for hospitals across the country. The project’s toolkit, mentoring program, and national advocacy efforts have proven so successful that the program is expanding this year.
In 2008, SHM began the first round of the Project BOOST mentoring program in six pilot hospitals. The first full cycle of Project BOOST mentoring sites began in March at 24 sites. The Hospitalist will feature updates on the full cycle of Project BOOST later this year. For more information about Project BOOST, visit www.hospitalmedicine.org/BOOST or e-mail BOOST@hospitalmedicine.org.
As a pilot site, Southwestern Vermont Medical Center in Bennington has worked with mentors for the past six months. We caught up with project leader Jennifer Fells, RN, MS, to discuss the institution’s participation.
—Jennifer Fells, RN, MS, Southwestern Vermont Medical Center, Bennington
Question: Why did your group choose to participate in the mentoring program?
Answer: We wanted to reduce our readmission rate, and we knew we weren’t doing a service to patients. This was also a goal of our organization overall; it’s not only a benefit to the hospital, but a larger value when the patient goes back to the community.
Q: How has the BOOST mentoring program benefited your program?
A: It helped us get organized by beginning the process and affirmed our belief that there were ways to address readmissions. The toolkit has proved to be invaluable. The mentors helped us keep on track and offered us guidance. They share the experiences of the other Project BOOST teams, and we benefit from that information.
The changes in the approach to the discharge process were a surprise to us. After we developed our team, we discovered how fragmented the discharge process was throughout the entire organization. Discharges were handled over multiple disciplines, and it was fragmented by design.
For example, our documentation, discharge plans, discharge recommendations, and patient-education materials are in different parts of our documentation system.
Q: What did you learn about your program through the initial Project BOOST step: analyze care delivery?
A: We realized that disciplines were not coordinated with one another and there was not enough time for the physician to complete the discharge plan. The process was cumbersome, awkward, and very time-consuming. We were looking to create efficiency in the information that needs to be coordinated to do those discharge orders.
Q: What additional changes do you hope to see in the remaining time with BOOST mentors?
A: We definitely hope to improve our process. We want to have implemented the tools and have a coordinated discharge process, and a centralized way to communicate the discharge plan. We found a lack of communication creates a barrier among disciplines, and we hope to correct that and become more customer-friendly to patients.
Q: How did your site’s BOOST mentor assist in the implementation process? What was the outcome?
A: Our site mentor helped with the clarification of data to be collected for measurement, keeping us on track with the toolkit, and served as another set of eyes. When you are in an organization, and even when you have the appropriate team, you always need somebody to say, “What do you think about this?” or “Did you think about this item?”
It’s another perspective, sharing gained knowledge from other organizations. That’s very critical.
SHM marketing coordinator Nadia Clenending contributed to this report.
SHM’s Project BOOST (Better Outcomes for Older Adults through Safe Transitions) is an initiative to improve practices in transition care and reduce readmission rates for hospitals across the country. The project’s toolkit, mentoring program, and national advocacy efforts have proven so successful that the program is expanding this year.
In 2008, SHM began the first round of the Project BOOST mentoring program in six pilot hospitals. The first full cycle of Project BOOST mentoring sites began in March at 24 sites. The Hospitalist will feature updates on the full cycle of Project BOOST later this year. For more information about Project BOOST, visit www.hospitalmedicine.org/BOOST or e-mail BOOST@hospitalmedicine.org.
As a pilot site, Southwestern Vermont Medical Center in Bennington has worked with mentors for the past six months. We caught up with project leader Jennifer Fells, RN, MS, to discuss the institution’s participation.
—Jennifer Fells, RN, MS, Southwestern Vermont Medical Center, Bennington
Question: Why did your group choose to participate in the mentoring program?
Answer: We wanted to reduce our readmission rate, and we knew we weren’t doing a service to patients. This was also a goal of our organization overall; it’s not only a benefit to the hospital, but a larger value when the patient goes back to the community.
Q: How has the BOOST mentoring program benefited your program?
A: It helped us get organized by beginning the process and affirmed our belief that there were ways to address readmissions. The toolkit has proved to be invaluable. The mentors helped us keep on track and offered us guidance. They share the experiences of the other Project BOOST teams, and we benefit from that information.
The changes in the approach to the discharge process were a surprise to us. After we developed our team, we discovered how fragmented the discharge process was throughout the entire organization. Discharges were handled over multiple disciplines, and it was fragmented by design.
For example, our documentation, discharge plans, discharge recommendations, and patient-education materials are in different parts of our documentation system.
Q: What did you learn about your program through the initial Project BOOST step: analyze care delivery?
A: We realized that disciplines were not coordinated with one another and there was not enough time for the physician to complete the discharge plan. The process was cumbersome, awkward, and very time-consuming. We were looking to create efficiency in the information that needs to be coordinated to do those discharge orders.
Q: What additional changes do you hope to see in the remaining time with BOOST mentors?
A: We definitely hope to improve our process. We want to have implemented the tools and have a coordinated discharge process, and a centralized way to communicate the discharge plan. We found a lack of communication creates a barrier among disciplines, and we hope to correct that and become more customer-friendly to patients.
Q: How did your site’s BOOST mentor assist in the implementation process? What was the outcome?
A: Our site mentor helped with the clarification of data to be collected for measurement, keeping us on track with the toolkit, and served as another set of eyes. When you are in an organization, and even when you have the appropriate team, you always need somebody to say, “What do you think about this?” or “Did you think about this item?”
It’s another perspective, sharing gained knowledge from other organizations. That’s very critical.
SHM marketing coordinator Nadia Clenending contributed to this report.
Consumer-Driven Healthcare
Joseph Forrester, DO, a critical-care hospitalist and pulmonologist in Denver, discovered firsthand how scoping out and paying for healthcare now resembles shopping for other big-ticket items. Unlike purchasing and paying down a diamond ring or a 60-inch flat-screen TV, for which the final price and payment is duly noted and balances promptly adjusted, hospital billing attempted to overcharge Dr. Forrester by 500%. He already had paid $4,000 toward his 2008 out-of-pocket deductible for medical expenses and was surprised when the hospital said he would have to pay the full $5,000 deductible before he could receive care. The savvy doc went to a real-time claims adjudication tool that he uses in his own practice to show the hospital he’d already satisfied the first 80% of the $5,000 deductible.
The hospital’s billing department listened and responded.
Dr. Forrester paid the remaining $1,000 to fulfill his deductible, and his insurer covered the rest of the treatment cost. “Having access to this information allowed him to receive care immediately, without having to wait weeks for the hospital to correct its mistaken information,” says Chris Stanley, MD, a medical director with United Healthcare.
High-deductible health plans (HDHPs) like Dr. Forrester’s have been growing in popularity since the establishment of health savings accounts (HSAs). Designed to help individuals save for future medical and retiree health expenses on a tax-free basis, HSAs were signed into law by President Bush in December 2003. These products are just beginning to influence how hospitals collect fees and how patients negotiate with their physicians—including hospitalists—about which medications, tests, and procedures they’re willing to pay for.
Break from Tradition
According to the U.S. Treasury Department Web site, HSAs allow individuals to “own and control the money in your HSA. Decisions on how to spend the money are made by the consumer without relying on a third party or a health insurer. Consumers will also decide what types of investments to make with the money in the account in order to make it grow.”
HSAs only are available to individuals covered solely through an HDHP. Individuals receiving veterans benefits or already on Medicare are not eligible; however, if they establish an HSA before enrolling in Medicare, they can keep it—but not add to it.
HDHPs offer consumers—especially young, healthy individuals—low premiums and high deductibles (between $1,150 and $2,900 for individuals and $2,300 to $5,800 for family plans). In addition to paying a low premium, consumers can put money into an HSA to pay for out-of-pocket expenses, including deductibles, co-pays, and co-insurance. The maximum amount of tax-free money a consumer can stash in an HSA this year is $5,800 for individuals and $11,600 for families. (Those 55 and older can contribute an additional $1,000 annually to their HSAs to accelerate their savings rates.)
Consumers can access HSA funds through a debit card, or they can pay for a service, then file for reimbursement.
An HSA should not be confused with a flexible spending account (FSA). Both are paid for by employees with pre-tax dollars; however, FSAs:
- Carry no insurance requirements;
- Are capped at $5,000 in annual contributions;
- Do not pay interest on the account balance; and
- Must be used—or forfeited—by the end of the plan year.
In contrast, an HSA:
- Is funded by the employee or jointly by employer and employee (known as a health reimbursement account, or HRA);
- Has insurance requirements on deductibles and out-of-pocket contributions;
- Pays the provider directly and submits receipts to the account administrator;
- Accumulates interest through a financial institution; and
- Allows unused funds to be carried forward.
Easy Business Decision
Employers are jumping on the HDHP bandwagon, largely to shift more health insurance costs to employees. HDHPs allow consumers to save on upfront costs (e.g., premiums and routine medical expenses) while allowing them to partner with their physicians when deciding how and when they will spend their HSA dollars.
Allowing patients to be involved in the testing, medication, and length-of-stay decisions relative to their care is a reversal from the status quo. Physicians working with hospitalized patients aren’t used to patients questioning treatment or asking for a cost analysis of medications. Another way to think about working with patients who have nontraditional plans: If you were eating at a soup kitchen, you probably wouldn’t complain about having to eat off of paper plates. But if you were dining at a five-star restaurant, you’d freely complain to the maître d’ if your soup was cold or the salad limp.
The 2008 National Study of Employer-Sponsored Health Plans, conducted by international human resources consulting firm Mercer, reported consumer-directed health plans, coupled with either an HSA or an HRA, are offered by 45% of companies with 25,000 or more employees (up from 22% in 2005). Nine percent of companies with 10 to 499 employees offer consumer-directed health plans, up from 2% in 2005.
Mercer partner Blaine Bos notes that raising deductibles is the fallback for employers faced with medical cost increases they can’t—or won’t—absorb. “The introduction of HSAs may have changed employers’ thinking on just how high a deductible can go without causing employees to revolt,” Bos says in the survey analysis. He predicts bad economic times will accelerate consumer-directed health plan uptake in small and large firms because they deliver substantially lower costs than PPOs and HMOs. In 2008, CDHP costs averaged $6,207 per employee, compared with $7,815 for PPOs and $7,768 for HMOs.
Data from ehealthinsurance.com indicate consumers are taking full advantage of HDHPs: Fifteen percent choose the highest deductibles, 48% the mid-range, and 37% the lowest deductible.
Still on HM’s Horizon
Consumer-driven healthcare has yet to significantly affect hospitals and—by extension—HM groups, although hospital admissions were down 2% nationally in 2008 and hospital debts are climbing. Adam Singer, MD, CEO of IPC: The Hospitalist Company, says the impact of HSAs on HM isn’t noticeable yet because patients haven’t adapted to the new model. “Consumers usually aren’t price-shopping the facility, because they’re committed to their physicians and will go to the hospital where their physician has privileges,” Dr. Singer says. “Additionally, the patients hospitalists see are very sick. Many came in through the ED and had no choice about how they got there. They certainly don’t pick their hospitalists.”
By extension, the ED isn’t immune to market forces driving consumer-directed care. Karen McConnell, PhD, director of the Oregon Health and Science University’s Center for Policy and Research in Emergency Medicine, posits that rapid adoption of high-deductible plans could change ED utilization (Ann Emerg Med. 2005;46(6):536-40). Although the ED may be insulated from extensive shopping and price negotiation because visits generally are for urgent conditions, Dr. McConnell says, ED utilization patterns may change if cost-conscious HSA holders forego other necessary medical care or seek substitutes for less-urgent problems.
The reality of consumer-directed healthcare and patients footing more of their own medical bills could eventually have a significant impact on HM programs. “Hospitals are under attack,” Dr. Singer says. “The 20% to 50% of hospital medicine program revenues received as support payments from their hospitals may drop as high-deductible plans with HSAs drive down hospital revenues. So as hospital revenues fall, the subsidies—particularly some of the more absurd, seven-figure subsidies that hospitalists enjoy—are vulnerable.”
Patients Take Charge
With their own money at stake, HSA consumers are engaged in the decision to spend—or save—their healthcare dollars. Although the shift in the doctor-patient decision-making process has slowly found its way into the hospital, it is playing out in doctor’s offices—one of several pipelines for hospital admissions. As a consumer, Linda Waldmann, manager of MyCost, a real-time claims-adjudication tool offered by Alegent Health, introduced cost when making treatment decisions after she was diagnosed with carpal tunnel syndrome. She asked her orthopedic surgeon lots of questions, absorbed what he said, then made her own treatment choices.
“My orthopedist wanted me to have three tests, but I elected to postpone one test until my HSA replenished the following year,” Waldmann says. “Doctors are still hesitant about negotiating with patients, but this one understood my concern.”
Blue Cross/Blue Shield of Tennessee’s (BCBST) Maggie Fox, director of application systems, saw a large jump (to 33% in 2009 from 8% in 2008) among BCBST’s 5,000 employees opting for HSAs. The Tennessee company emphasizes prevention, education, and wellness as critical components of consumer-directed care, and the company’s HSA consumers are offered a variety of discounts for adhering to healthy lifestyles.
At BCBST, HSAs have opened dialogue between patients, physicians, and hospitals. Through a Web-based portal called “Blue Access,” providers receive information on a patient’s financial liability in as little as 10 seconds. “HSAs and HRAs have created a patient liability that never existed before,” Fox says. “Higher out-of-pocket costs change everything. Providers need to collect payment at the point of care, whether that’s the office or hospital. There’s more work at checkout, but at least patients and providers know the exact amount the care costs and how much the patient has to pay.”
Davis Liu, MD, a Wharton School of Business graduate and family physician with Northern California Permanente Medical Group, advises hospitalists to be ready for patients with HSAs to challenge treatment decisions because of the cost. He says hospitalists must prepare to communicate clearly and effectively with HSA patients, especially when it comes to necessary testing and medications. The task might be difficult because information about testing costs and procedures is limited, and prices vary dramatically by hospital and region. “While it is extremely unlikely that patients will refuse testing when hospitalized, hospitalists must be acutely aware that these patients may skip follow-up appointments, testing, and surgeries,” Dr. Liu says.
United Healthcare’s Dr. Stanley sees physician decision-making evolving as HSA patients become more aware of the economics of treatment options. “Patients are already questioning doctors who order four tests when they’re only willing to pay for three, wanting to postpone procedures, and asking about costs for additional tests and procedures,” he says. “Eventually, cost consciousness will impact group practices. They will have to decrease overhead, revamp collection processes, and strive for administrative simplicity.” Woe to the physician who believes cost isn’t their responsibility, Dr. Stanley says, as they “must realize they’re small-business owners and act accordingly.”
The Future
Consumer-driven healthcare might have little effect on hospitals right now, but change is on the horizon, according to Greg Scandlen, president and CEO of Consumers for Health Care Choice in Hagerstown, Md. He cites the 2008 National Health Interview Survey conducted by the Centers for Disease Control and Prevention, which shows 20% of Americans have an HDHP, as proof positive these new plans are increasing market penetration.
“We’re at a tipping point where every provider will have to deal with cash-paying clients,” he says. “Hospitals with Chargemaster pricing [the list price for services and procedures charged to self-pay and other uninsured clients, usually three to three and a half times the normal Medicare reimbursement] won’t get away with that much longer. They’ll have to charge reasonable, negotiated rates rather than slamming self-pay patients.”
It looks as though the days when patients entered the hospital worried about getting well and dealing with the bills later will soon be in the rearview mirror. Not far into the future, individuals with self-pay components to their health insurance might demand and receive full financial disclosure of all their expected hospital costs (see “Patients Can Shop Around,” p. 29) at the time of admission, with necessary adjustments at discharge.
“Hospitals can’t have secret pricing in a transparent, consumer-driven world,” says Scandlen. “The HDHP model hasn’t fully hit hospitals yet, but they’ll get clobbered in about two years if they don’t adapt.” TH
Marlene Piturro is a freelance writer based in Hastings-on-Hudson, NY.
Joseph Forrester, DO, a critical-care hospitalist and pulmonologist in Denver, discovered firsthand how scoping out and paying for healthcare now resembles shopping for other big-ticket items. Unlike purchasing and paying down a diamond ring or a 60-inch flat-screen TV, for which the final price and payment is duly noted and balances promptly adjusted, hospital billing attempted to overcharge Dr. Forrester by 500%. He already had paid $4,000 toward his 2008 out-of-pocket deductible for medical expenses and was surprised when the hospital said he would have to pay the full $5,000 deductible before he could receive care. The savvy doc went to a real-time claims adjudication tool that he uses in his own practice to show the hospital he’d already satisfied the first 80% of the $5,000 deductible.
The hospital’s billing department listened and responded.
Dr. Forrester paid the remaining $1,000 to fulfill his deductible, and his insurer covered the rest of the treatment cost. “Having access to this information allowed him to receive care immediately, without having to wait weeks for the hospital to correct its mistaken information,” says Chris Stanley, MD, a medical director with United Healthcare.
High-deductible health plans (HDHPs) like Dr. Forrester’s have been growing in popularity since the establishment of health savings accounts (HSAs). Designed to help individuals save for future medical and retiree health expenses on a tax-free basis, HSAs were signed into law by President Bush in December 2003. These products are just beginning to influence how hospitals collect fees and how patients negotiate with their physicians—including hospitalists—about which medications, tests, and procedures they’re willing to pay for.
Break from Tradition
According to the U.S. Treasury Department Web site, HSAs allow individuals to “own and control the money in your HSA. Decisions on how to spend the money are made by the consumer without relying on a third party or a health insurer. Consumers will also decide what types of investments to make with the money in the account in order to make it grow.”
HSAs only are available to individuals covered solely through an HDHP. Individuals receiving veterans benefits or already on Medicare are not eligible; however, if they establish an HSA before enrolling in Medicare, they can keep it—but not add to it.
HDHPs offer consumers—especially young, healthy individuals—low premiums and high deductibles (between $1,150 and $2,900 for individuals and $2,300 to $5,800 for family plans). In addition to paying a low premium, consumers can put money into an HSA to pay for out-of-pocket expenses, including deductibles, co-pays, and co-insurance. The maximum amount of tax-free money a consumer can stash in an HSA this year is $5,800 for individuals and $11,600 for families. (Those 55 and older can contribute an additional $1,000 annually to their HSAs to accelerate their savings rates.)
Consumers can access HSA funds through a debit card, or they can pay for a service, then file for reimbursement.
An HSA should not be confused with a flexible spending account (FSA). Both are paid for by employees with pre-tax dollars; however, FSAs:
- Carry no insurance requirements;
- Are capped at $5,000 in annual contributions;
- Do not pay interest on the account balance; and
- Must be used—or forfeited—by the end of the plan year.
In contrast, an HSA:
- Is funded by the employee or jointly by employer and employee (known as a health reimbursement account, or HRA);
- Has insurance requirements on deductibles and out-of-pocket contributions;
- Pays the provider directly and submits receipts to the account administrator;
- Accumulates interest through a financial institution; and
- Allows unused funds to be carried forward.
Easy Business Decision
Employers are jumping on the HDHP bandwagon, largely to shift more health insurance costs to employees. HDHPs allow consumers to save on upfront costs (e.g., premiums and routine medical expenses) while allowing them to partner with their physicians when deciding how and when they will spend their HSA dollars.
Allowing patients to be involved in the testing, medication, and length-of-stay decisions relative to their care is a reversal from the status quo. Physicians working with hospitalized patients aren’t used to patients questioning treatment or asking for a cost analysis of medications. Another way to think about working with patients who have nontraditional plans: If you were eating at a soup kitchen, you probably wouldn’t complain about having to eat off of paper plates. But if you were dining at a five-star restaurant, you’d freely complain to the maître d’ if your soup was cold or the salad limp.
The 2008 National Study of Employer-Sponsored Health Plans, conducted by international human resources consulting firm Mercer, reported consumer-directed health plans, coupled with either an HSA or an HRA, are offered by 45% of companies with 25,000 or more employees (up from 22% in 2005). Nine percent of companies with 10 to 499 employees offer consumer-directed health plans, up from 2% in 2005.
Mercer partner Blaine Bos notes that raising deductibles is the fallback for employers faced with medical cost increases they can’t—or won’t—absorb. “The introduction of HSAs may have changed employers’ thinking on just how high a deductible can go without causing employees to revolt,” Bos says in the survey analysis. He predicts bad economic times will accelerate consumer-directed health plan uptake in small and large firms because they deliver substantially lower costs than PPOs and HMOs. In 2008, CDHP costs averaged $6,207 per employee, compared with $7,815 for PPOs and $7,768 for HMOs.
Data from ehealthinsurance.com indicate consumers are taking full advantage of HDHPs: Fifteen percent choose the highest deductibles, 48% the mid-range, and 37% the lowest deductible.
Still on HM’s Horizon
Consumer-driven healthcare has yet to significantly affect hospitals and—by extension—HM groups, although hospital admissions were down 2% nationally in 2008 and hospital debts are climbing. Adam Singer, MD, CEO of IPC: The Hospitalist Company, says the impact of HSAs on HM isn’t noticeable yet because patients haven’t adapted to the new model. “Consumers usually aren’t price-shopping the facility, because they’re committed to their physicians and will go to the hospital where their physician has privileges,” Dr. Singer says. “Additionally, the patients hospitalists see are very sick. Many came in through the ED and had no choice about how they got there. They certainly don’t pick their hospitalists.”
By extension, the ED isn’t immune to market forces driving consumer-directed care. Karen McConnell, PhD, director of the Oregon Health and Science University’s Center for Policy and Research in Emergency Medicine, posits that rapid adoption of high-deductible plans could change ED utilization (Ann Emerg Med. 2005;46(6):536-40). Although the ED may be insulated from extensive shopping and price negotiation because visits generally are for urgent conditions, Dr. McConnell says, ED utilization patterns may change if cost-conscious HSA holders forego other necessary medical care or seek substitutes for less-urgent problems.
The reality of consumer-directed healthcare and patients footing more of their own medical bills could eventually have a significant impact on HM programs. “Hospitals are under attack,” Dr. Singer says. “The 20% to 50% of hospital medicine program revenues received as support payments from their hospitals may drop as high-deductible plans with HSAs drive down hospital revenues. So as hospital revenues fall, the subsidies—particularly some of the more absurd, seven-figure subsidies that hospitalists enjoy—are vulnerable.”
Patients Take Charge
With their own money at stake, HSA consumers are engaged in the decision to spend—or save—their healthcare dollars. Although the shift in the doctor-patient decision-making process has slowly found its way into the hospital, it is playing out in doctor’s offices—one of several pipelines for hospital admissions. As a consumer, Linda Waldmann, manager of MyCost, a real-time claims-adjudication tool offered by Alegent Health, introduced cost when making treatment decisions after she was diagnosed with carpal tunnel syndrome. She asked her orthopedic surgeon lots of questions, absorbed what he said, then made her own treatment choices.
“My orthopedist wanted me to have three tests, but I elected to postpone one test until my HSA replenished the following year,” Waldmann says. “Doctors are still hesitant about negotiating with patients, but this one understood my concern.”
Blue Cross/Blue Shield of Tennessee’s (BCBST) Maggie Fox, director of application systems, saw a large jump (to 33% in 2009 from 8% in 2008) among BCBST’s 5,000 employees opting for HSAs. The Tennessee company emphasizes prevention, education, and wellness as critical components of consumer-directed care, and the company’s HSA consumers are offered a variety of discounts for adhering to healthy lifestyles.
At BCBST, HSAs have opened dialogue between patients, physicians, and hospitals. Through a Web-based portal called “Blue Access,” providers receive information on a patient’s financial liability in as little as 10 seconds. “HSAs and HRAs have created a patient liability that never existed before,” Fox says. “Higher out-of-pocket costs change everything. Providers need to collect payment at the point of care, whether that’s the office or hospital. There’s more work at checkout, but at least patients and providers know the exact amount the care costs and how much the patient has to pay.”
Davis Liu, MD, a Wharton School of Business graduate and family physician with Northern California Permanente Medical Group, advises hospitalists to be ready for patients with HSAs to challenge treatment decisions because of the cost. He says hospitalists must prepare to communicate clearly and effectively with HSA patients, especially when it comes to necessary testing and medications. The task might be difficult because information about testing costs and procedures is limited, and prices vary dramatically by hospital and region. “While it is extremely unlikely that patients will refuse testing when hospitalized, hospitalists must be acutely aware that these patients may skip follow-up appointments, testing, and surgeries,” Dr. Liu says.
United Healthcare’s Dr. Stanley sees physician decision-making evolving as HSA patients become more aware of the economics of treatment options. “Patients are already questioning doctors who order four tests when they’re only willing to pay for three, wanting to postpone procedures, and asking about costs for additional tests and procedures,” he says. “Eventually, cost consciousness will impact group practices. They will have to decrease overhead, revamp collection processes, and strive for administrative simplicity.” Woe to the physician who believes cost isn’t their responsibility, Dr. Stanley says, as they “must realize they’re small-business owners and act accordingly.”
The Future
Consumer-driven healthcare might have little effect on hospitals right now, but change is on the horizon, according to Greg Scandlen, president and CEO of Consumers for Health Care Choice in Hagerstown, Md. He cites the 2008 National Health Interview Survey conducted by the Centers for Disease Control and Prevention, which shows 20% of Americans have an HDHP, as proof positive these new plans are increasing market penetration.
“We’re at a tipping point where every provider will have to deal with cash-paying clients,” he says. “Hospitals with Chargemaster pricing [the list price for services and procedures charged to self-pay and other uninsured clients, usually three to three and a half times the normal Medicare reimbursement] won’t get away with that much longer. They’ll have to charge reasonable, negotiated rates rather than slamming self-pay patients.”
It looks as though the days when patients entered the hospital worried about getting well and dealing with the bills later will soon be in the rearview mirror. Not far into the future, individuals with self-pay components to their health insurance might demand and receive full financial disclosure of all their expected hospital costs (see “Patients Can Shop Around,” p. 29) at the time of admission, with necessary adjustments at discharge.
“Hospitals can’t have secret pricing in a transparent, consumer-driven world,” says Scandlen. “The HDHP model hasn’t fully hit hospitals yet, but they’ll get clobbered in about two years if they don’t adapt.” TH
Marlene Piturro is a freelance writer based in Hastings-on-Hudson, NY.
Joseph Forrester, DO, a critical-care hospitalist and pulmonologist in Denver, discovered firsthand how scoping out and paying for healthcare now resembles shopping for other big-ticket items. Unlike purchasing and paying down a diamond ring or a 60-inch flat-screen TV, for which the final price and payment is duly noted and balances promptly adjusted, hospital billing attempted to overcharge Dr. Forrester by 500%. He already had paid $4,000 toward his 2008 out-of-pocket deductible for medical expenses and was surprised when the hospital said he would have to pay the full $5,000 deductible before he could receive care. The savvy doc went to a real-time claims adjudication tool that he uses in his own practice to show the hospital he’d already satisfied the first 80% of the $5,000 deductible.
The hospital’s billing department listened and responded.
Dr. Forrester paid the remaining $1,000 to fulfill his deductible, and his insurer covered the rest of the treatment cost. “Having access to this information allowed him to receive care immediately, without having to wait weeks for the hospital to correct its mistaken information,” says Chris Stanley, MD, a medical director with United Healthcare.
High-deductible health plans (HDHPs) like Dr. Forrester’s have been growing in popularity since the establishment of health savings accounts (HSAs). Designed to help individuals save for future medical and retiree health expenses on a tax-free basis, HSAs were signed into law by President Bush in December 2003. These products are just beginning to influence how hospitals collect fees and how patients negotiate with their physicians—including hospitalists—about which medications, tests, and procedures they’re willing to pay for.
Break from Tradition
According to the U.S. Treasury Department Web site, HSAs allow individuals to “own and control the money in your HSA. Decisions on how to spend the money are made by the consumer without relying on a third party or a health insurer. Consumers will also decide what types of investments to make with the money in the account in order to make it grow.”
HSAs only are available to individuals covered solely through an HDHP. Individuals receiving veterans benefits or already on Medicare are not eligible; however, if they establish an HSA before enrolling in Medicare, they can keep it—but not add to it.
HDHPs offer consumers—especially young, healthy individuals—low premiums and high deductibles (between $1,150 and $2,900 for individuals and $2,300 to $5,800 for family plans). In addition to paying a low premium, consumers can put money into an HSA to pay for out-of-pocket expenses, including deductibles, co-pays, and co-insurance. The maximum amount of tax-free money a consumer can stash in an HSA this year is $5,800 for individuals and $11,600 for families. (Those 55 and older can contribute an additional $1,000 annually to their HSAs to accelerate their savings rates.)
Consumers can access HSA funds through a debit card, or they can pay for a service, then file for reimbursement.
An HSA should not be confused with a flexible spending account (FSA). Both are paid for by employees with pre-tax dollars; however, FSAs:
- Carry no insurance requirements;
- Are capped at $5,000 in annual contributions;
- Do not pay interest on the account balance; and
- Must be used—or forfeited—by the end of the plan year.
In contrast, an HSA:
- Is funded by the employee or jointly by employer and employee (known as a health reimbursement account, or HRA);
- Has insurance requirements on deductibles and out-of-pocket contributions;
- Pays the provider directly and submits receipts to the account administrator;
- Accumulates interest through a financial institution; and
- Allows unused funds to be carried forward.
Easy Business Decision
Employers are jumping on the HDHP bandwagon, largely to shift more health insurance costs to employees. HDHPs allow consumers to save on upfront costs (e.g., premiums and routine medical expenses) while allowing them to partner with their physicians when deciding how and when they will spend their HSA dollars.
Allowing patients to be involved in the testing, medication, and length-of-stay decisions relative to their care is a reversal from the status quo. Physicians working with hospitalized patients aren’t used to patients questioning treatment or asking for a cost analysis of medications. Another way to think about working with patients who have nontraditional plans: If you were eating at a soup kitchen, you probably wouldn’t complain about having to eat off of paper plates. But if you were dining at a five-star restaurant, you’d freely complain to the maître d’ if your soup was cold or the salad limp.
The 2008 National Study of Employer-Sponsored Health Plans, conducted by international human resources consulting firm Mercer, reported consumer-directed health plans, coupled with either an HSA or an HRA, are offered by 45% of companies with 25,000 or more employees (up from 22% in 2005). Nine percent of companies with 10 to 499 employees offer consumer-directed health plans, up from 2% in 2005.
Mercer partner Blaine Bos notes that raising deductibles is the fallback for employers faced with medical cost increases they can’t—or won’t—absorb. “The introduction of HSAs may have changed employers’ thinking on just how high a deductible can go without causing employees to revolt,” Bos says in the survey analysis. He predicts bad economic times will accelerate consumer-directed health plan uptake in small and large firms because they deliver substantially lower costs than PPOs and HMOs. In 2008, CDHP costs averaged $6,207 per employee, compared with $7,815 for PPOs and $7,768 for HMOs.
Data from ehealthinsurance.com indicate consumers are taking full advantage of HDHPs: Fifteen percent choose the highest deductibles, 48% the mid-range, and 37% the lowest deductible.
Still on HM’s Horizon
Consumer-driven healthcare has yet to significantly affect hospitals and—by extension—HM groups, although hospital admissions were down 2% nationally in 2008 and hospital debts are climbing. Adam Singer, MD, CEO of IPC: The Hospitalist Company, says the impact of HSAs on HM isn’t noticeable yet because patients haven’t adapted to the new model. “Consumers usually aren’t price-shopping the facility, because they’re committed to their physicians and will go to the hospital where their physician has privileges,” Dr. Singer says. “Additionally, the patients hospitalists see are very sick. Many came in through the ED and had no choice about how they got there. They certainly don’t pick their hospitalists.”
By extension, the ED isn’t immune to market forces driving consumer-directed care. Karen McConnell, PhD, director of the Oregon Health and Science University’s Center for Policy and Research in Emergency Medicine, posits that rapid adoption of high-deductible plans could change ED utilization (Ann Emerg Med. 2005;46(6):536-40). Although the ED may be insulated from extensive shopping and price negotiation because visits generally are for urgent conditions, Dr. McConnell says, ED utilization patterns may change if cost-conscious HSA holders forego other necessary medical care or seek substitutes for less-urgent problems.
The reality of consumer-directed healthcare and patients footing more of their own medical bills could eventually have a significant impact on HM programs. “Hospitals are under attack,” Dr. Singer says. “The 20% to 50% of hospital medicine program revenues received as support payments from their hospitals may drop as high-deductible plans with HSAs drive down hospital revenues. So as hospital revenues fall, the subsidies—particularly some of the more absurd, seven-figure subsidies that hospitalists enjoy—are vulnerable.”
Patients Take Charge
With their own money at stake, HSA consumers are engaged in the decision to spend—or save—their healthcare dollars. Although the shift in the doctor-patient decision-making process has slowly found its way into the hospital, it is playing out in doctor’s offices—one of several pipelines for hospital admissions. As a consumer, Linda Waldmann, manager of MyCost, a real-time claims-adjudication tool offered by Alegent Health, introduced cost when making treatment decisions after she was diagnosed with carpal tunnel syndrome. She asked her orthopedic surgeon lots of questions, absorbed what he said, then made her own treatment choices.
“My orthopedist wanted me to have three tests, but I elected to postpone one test until my HSA replenished the following year,” Waldmann says. “Doctors are still hesitant about negotiating with patients, but this one understood my concern.”
Blue Cross/Blue Shield of Tennessee’s (BCBST) Maggie Fox, director of application systems, saw a large jump (to 33% in 2009 from 8% in 2008) among BCBST’s 5,000 employees opting for HSAs. The Tennessee company emphasizes prevention, education, and wellness as critical components of consumer-directed care, and the company’s HSA consumers are offered a variety of discounts for adhering to healthy lifestyles.
At BCBST, HSAs have opened dialogue between patients, physicians, and hospitals. Through a Web-based portal called “Blue Access,” providers receive information on a patient’s financial liability in as little as 10 seconds. “HSAs and HRAs have created a patient liability that never existed before,” Fox says. “Higher out-of-pocket costs change everything. Providers need to collect payment at the point of care, whether that’s the office or hospital. There’s more work at checkout, but at least patients and providers know the exact amount the care costs and how much the patient has to pay.”
Davis Liu, MD, a Wharton School of Business graduate and family physician with Northern California Permanente Medical Group, advises hospitalists to be ready for patients with HSAs to challenge treatment decisions because of the cost. He says hospitalists must prepare to communicate clearly and effectively with HSA patients, especially when it comes to necessary testing and medications. The task might be difficult because information about testing costs and procedures is limited, and prices vary dramatically by hospital and region. “While it is extremely unlikely that patients will refuse testing when hospitalized, hospitalists must be acutely aware that these patients may skip follow-up appointments, testing, and surgeries,” Dr. Liu says.
United Healthcare’s Dr. Stanley sees physician decision-making evolving as HSA patients become more aware of the economics of treatment options. “Patients are already questioning doctors who order four tests when they’re only willing to pay for three, wanting to postpone procedures, and asking about costs for additional tests and procedures,” he says. “Eventually, cost consciousness will impact group practices. They will have to decrease overhead, revamp collection processes, and strive for administrative simplicity.” Woe to the physician who believes cost isn’t their responsibility, Dr. Stanley says, as they “must realize they’re small-business owners and act accordingly.”
The Future
Consumer-driven healthcare might have little effect on hospitals right now, but change is on the horizon, according to Greg Scandlen, president and CEO of Consumers for Health Care Choice in Hagerstown, Md. He cites the 2008 National Health Interview Survey conducted by the Centers for Disease Control and Prevention, which shows 20% of Americans have an HDHP, as proof positive these new plans are increasing market penetration.
“We’re at a tipping point where every provider will have to deal with cash-paying clients,” he says. “Hospitals with Chargemaster pricing [the list price for services and procedures charged to self-pay and other uninsured clients, usually three to three and a half times the normal Medicare reimbursement] won’t get away with that much longer. They’ll have to charge reasonable, negotiated rates rather than slamming self-pay patients.”
It looks as though the days when patients entered the hospital worried about getting well and dealing with the bills later will soon be in the rearview mirror. Not far into the future, individuals with self-pay components to their health insurance might demand and receive full financial disclosure of all their expected hospital costs (see “Patients Can Shop Around,” p. 29) at the time of admission, with necessary adjustments at discharge.
“Hospitals can’t have secret pricing in a transparent, consumer-driven world,” says Scandlen. “The HDHP model hasn’t fully hit hospitals yet, but they’ll get clobbered in about two years if they don’t adapt.” TH
Marlene Piturro is a freelance writer based in Hastings-on-Hudson, NY.
Proceedings of the 2008 Heart-Brain Summit
Supplement Editor:
Marc S. Penn, MD, PhD
Contents
Preface
Earl E. Bakken, MD, HonC, Hon DSc (3), Hon DHL (2)
Introduction: Heart-brain medicine: Update 2008
Marc S. Penn, MD, PhD, and Earl E. Bakken, MD, HonC, Hon DSc (3), Hon DHL (2)
Bakken Lecture: The brain, the heart, and therapeutic hypothermia
Patrick M. Kochanek, MD
Session 1: Pathways Involved in Neuromodulation of Risks in Coronary Artery Disease
Depression and heart rate variability in patients with coronary heart disease
Robert M. Carney, PhD, and Kenneth E. Freedland, PhD
Autonomic function and prognosis
Michael S. Lauer, MD
Vagal tone and the inflammatory reflex
Julian F. Thayer, PhD
Inflammation, atherosclerosis, and arterial thrombosis: Role of the scavenger receptors CD36
Roy L. Silverstein, MD
Pioneer Award Address: Ignorance isn't biased: Comments on receiving the Pioneer Award
David S. Goldstein, MD, PhD
Session II: Measures and Strategies for Modulation of Heart-Brain Interactions
Heart rate variability with deep breathing as a clinical test of cardiovagal function
Robert W. Shields, Jr, MD
Basic research models for the study of underlying mechanisms of electrical neuromodulation and ischemi heart-brain interactions
Mike J.L. DeJongste, MD, PhD, FESC; Gert J. TerHorst, PhD; and Robert D. Foreman, PhD
Session III: Annual Review of Key Papers in Heart-Brain Medicine
Key papers in the field published during the year prior to the Summit were discussed; two of those papers are reported here.
Cardiac sympathetic denervation preceding motor signs in Parkinson disease
David S. Goldstein, MD, PhD; Yehonatan Sharabi, MD; Barbara I. Karp, MD; Oladi Bentho; Ahmed Saleem, MD; Karel Pacak, MD, PhD; and Graeme Eisenhofer, PhD
Supine low-frequency power of heart rate variability reflects baroreflex function, not cardiac sympathetic innervation
Jeffrey P. Moak, MD; David S. Goldstein, MD, PhD; Basil A. Eldadah, MD, PhD; Ahmed Saleem, MD; Courtney Holmes, CMT; Sandra Pechnik, RN; and Yehonatan Sharabi, MD
Session IV: Code Lavender—Strategies for Implementing Heart-Brain Medicine
Is posttraumatic stress disorder related to development of heart disease? An update
Laura D. Kubzansky, PhD, and Karestan C. Koenen, PhD
Creating a healing environment: Rationale and research overview
Jone Geimer-Flanders, DO
Redesigning the neurocritical care unit to enhance family participation and improve outcomes
Owen Samuels, MD
Session V: Insights into Neuromodulation of Cardiovascular Function
Neuromodulation of cardiac pain and cerebral vasculature: Neural mechanisms
Robert D. Foreman, PhD, and Chao Qin, MD, PhD
Pinacidil induces vascular dilation and hyperemia in vivo and does not impact biophysical properties of neurons and astrocytes in vitro
Rosa Cao; Bryan T. Higashikubo; Jessica Cardin; Ulf Knoblich; Raddy Ramos, PhD; Mark T. Nelson, PhD; Christopher I. Moore, PhD; and Joshua C. Brumberg, PhD
The polyvagal theory: New insights into adaptive reactions of the autonomic nervous system
Stephen W. Porges, PhD
Poster Abstracts
Abstract 1: Insulin use does not protect against restenosis in diabetic patients presenting with acute coronary syndrome or symptomatic angina
Matthew C. Becker, MD; John M. Galla, MD; Saif Anwaruddin, MD; Samir Kapadia, MD; and Richard A. Krasuski, MD
Abstract 2: Postoperative statin use and lower LDL cholesterol concentration are associated with reduced incidence of stroke
Matthew C. Becker, John M. Galla, Ryan P. Daly, Femi Philip, Peter Zimbwa, Stephen O. Chen, Chen H. Chow, Tingfei Hu, Richard A. Krasuski, and Arman T. Askari
Abstract 3: Brain edema and blood-brain barrier leakage influence antiepileptic drug levels
Giulia Betto, Vincent Fazio, Damir Janigro, and Chaitali Ghosh
Abstract 4: CPAP treatment vs conservative treatment in mild obstructive sleep apnea: Implications on cardiovascular morbidity
Kumar Budur, MD, and Nattapong Jaimchariyatam, MD
Abstract 5: New bioinformatics program identifies behavioral medicine interventions for epidemic cardiovascular disease in the developing world: Analysis of multidisciplinary findings for launching a new global public health initiative in heart-brain medicine
William C. Bushell, PhD
Abstract 6: Do systemic inflammation and blood-brain barrier failure play a role in pediatric psychosis?
Erin Carlton, Tatiana Falcone, Ayush Batra, Vince Fazio, Kathleen Franco, and Damir Janigro
Abstract 7: Brain, heart, and education
Linda Bryant Caviness, PhD
Abstract 8: Tobacco smoke mediates a monocytic and endothelial proinflammatory activation that synergistically affects BBB integrity
L. Cucullo, T. Sathe, M. Hossain, and D. Janigro
Abstract 9: Dynamic changes in ECG predict poor outcome after aneurysmal subarachnoid hemorrhage (aSAH)
H.A. Elsharkawy, MD; S.M. El Hadi, MD, PhD; J.E. Tetzlaff, MD; and J.J. Provencio, MD, FCCM
Abstract 10: Mechanism studies of malformation of cortical development by prenatal exposure of combined methylazoxymethanol and thalidomide
Q. Fan, S. Ramakrishna, N. Marchi, V. Fazio, K. Hallene, and D. Janigro
Abstract 11: Proapolipoprotein A1 demonstrates improved potential as a serum marker for brain metastases without vascular disease interference
Vince Fazio, Peter Mazzone, Nicola Marchi, Thomas Masaryk, and Damir Janigro
Abstract 12: Biofeedback-assisted stress management training to reverse myocardial remodeling in patients with end-stage heart failure
Dana L. Frank, BS; Mary E. Klecka, BA; Jerry Kiffer, MA; Heather Henrickson, PhD; Michael G. McKee, PhD; and Christine S. Moravec, PhD
Abstract 13: Nitric oxide and arginine metabolism in depression: Effect of a serotonin-norepinephrine reuptake inhibitor
Angelos Halaris, John Piletz, Omer Iqbal, Debra Hoppensteadt, Jawed Fareed, He Zhu, James Sinacore, and C. Lindsay DeVane
Abstract 14: Association between excessive daytime sleepiness and oxygen desaturation in obstructive sleep apnea syndrome: Nadir oxygen saturation vs mean oxygen saturation vs time spent below 90% oxygen saturation—which is important?
Nattapong Jaimchariyatam, MD, and Kumar Budur, MD
Abstract 15: Endotoxin preconditioning of the CNS: Microglia activation and neuroprotection
Walid Jalabi, Ranjan Dutta, Yongming Jin, Gerson Criste, Xinghua Yin, Grahame J. Kidd, and Bruce D. Trapp
Abstract 16: Pilot of stress reduction strategies for patients after a coronary event
R. Lindquist, D. Windenburg, K. Savik, and U. Bronas
Abstract 17: Cerebrovascular substrates of seizures after cardiopulmonary bypass
Rebecca O’Dwyer, Tim Wehner, Dileep Nair, Giulia Betto, Nicola Marchi, and Damir Janigro
Abstract 18: Depression and whole blood serotonin in patients with coronary heart disease from the heart and soul study
Lawson Wulsin, Dominique Musselman, Christian Otte, Erica Bruce, Sadia Ali, and Mary Whooley
Abstract 19: Gender differences prominent in linking anxiety to long-term mortality among the elderly
Jianping Zhang, MD, PhD; Boaz Kahana, PhD; Eva Kahana, PhD; Bo Hu, PhD; and Leo Pozuelo, MD
Abstract 20: Temporal lobe and sinus node: A case report provides evidence for bidirectional effects
Rebecca O’Dwyer, MD; Andreas Alexopoulos, MD, MPH; Walid Saliba, MD; Imad Najm, MD; and Richard Burgess, MD, PhD
Supplement Editor:
Marc S. Penn, MD, PhD
Contents
Preface
Earl E. Bakken, MD, HonC, Hon DSc (3), Hon DHL (2)
Introduction: Heart-brain medicine: Update 2008
Marc S. Penn, MD, PhD, and Earl E. Bakken, MD, HonC, Hon DSc (3), Hon DHL (2)
Bakken Lecture: The brain, the heart, and therapeutic hypothermia
Patrick M. Kochanek, MD
Session 1: Pathways Involved in Neuromodulation of Risks in Coronary Artery Disease
Depression and heart rate variability in patients with coronary heart disease
Robert M. Carney, PhD, and Kenneth E. Freedland, PhD
Autonomic function and prognosis
Michael S. Lauer, MD
Vagal tone and the inflammatory reflex
Julian F. Thayer, PhD
Inflammation, atherosclerosis, and arterial thrombosis: Role of the scavenger receptors CD36
Roy L. Silverstein, MD
Pioneer Award Address: Ignorance isn't biased: Comments on receiving the Pioneer Award
David S. Goldstein, MD, PhD
Session II: Measures and Strategies for Modulation of Heart-Brain Interactions
Heart rate variability with deep breathing as a clinical test of cardiovagal function
Robert W. Shields, Jr, MD
Basic research models for the study of underlying mechanisms of electrical neuromodulation and ischemi heart-brain interactions
Mike J.L. DeJongste, MD, PhD, FESC; Gert J. TerHorst, PhD; and Robert D. Foreman, PhD
Session III: Annual Review of Key Papers in Heart-Brain Medicine
Key papers in the field published during the year prior to the Summit were discussed; two of those papers are reported here.
Cardiac sympathetic denervation preceding motor signs in Parkinson disease
David S. Goldstein, MD, PhD; Yehonatan Sharabi, MD; Barbara I. Karp, MD; Oladi Bentho; Ahmed Saleem, MD; Karel Pacak, MD, PhD; and Graeme Eisenhofer, PhD
Supine low-frequency power of heart rate variability reflects baroreflex function, not cardiac sympathetic innervation
Jeffrey P. Moak, MD; David S. Goldstein, MD, PhD; Basil A. Eldadah, MD, PhD; Ahmed Saleem, MD; Courtney Holmes, CMT; Sandra Pechnik, RN; and Yehonatan Sharabi, MD
Session IV: Code Lavender—Strategies for Implementing Heart-Brain Medicine
Is posttraumatic stress disorder related to development of heart disease? An update
Laura D. Kubzansky, PhD, and Karestan C. Koenen, PhD
Creating a healing environment: Rationale and research overview
Jone Geimer-Flanders, DO
Redesigning the neurocritical care unit to enhance family participation and improve outcomes
Owen Samuels, MD
Session V: Insights into Neuromodulation of Cardiovascular Function
Neuromodulation of cardiac pain and cerebral vasculature: Neural mechanisms
Robert D. Foreman, PhD, and Chao Qin, MD, PhD
Pinacidil induces vascular dilation and hyperemia in vivo and does not impact biophysical properties of neurons and astrocytes in vitro
Rosa Cao; Bryan T. Higashikubo; Jessica Cardin; Ulf Knoblich; Raddy Ramos, PhD; Mark T. Nelson, PhD; Christopher I. Moore, PhD; and Joshua C. Brumberg, PhD
The polyvagal theory: New insights into adaptive reactions of the autonomic nervous system
Stephen W. Porges, PhD
Poster Abstracts
Abstract 1: Insulin use does not protect against restenosis in diabetic patients presenting with acute coronary syndrome or symptomatic angina
Matthew C. Becker, MD; John M. Galla, MD; Saif Anwaruddin, MD; Samir Kapadia, MD; and Richard A. Krasuski, MD
Abstract 2: Postoperative statin use and lower LDL cholesterol concentration are associated with reduced incidence of stroke
Matthew C. Becker, John M. Galla, Ryan P. Daly, Femi Philip, Peter Zimbwa, Stephen O. Chen, Chen H. Chow, Tingfei Hu, Richard A. Krasuski, and Arman T. Askari
Abstract 3: Brain edema and blood-brain barrier leakage influence antiepileptic drug levels
Giulia Betto, Vincent Fazio, Damir Janigro, and Chaitali Ghosh
Abstract 4: CPAP treatment vs conservative treatment in mild obstructive sleep apnea: Implications on cardiovascular morbidity
Kumar Budur, MD, and Nattapong Jaimchariyatam, MD
Abstract 5: New bioinformatics program identifies behavioral medicine interventions for epidemic cardiovascular disease in the developing world: Analysis of multidisciplinary findings for launching a new global public health initiative in heart-brain medicine
William C. Bushell, PhD
Abstract 6: Do systemic inflammation and blood-brain barrier failure play a role in pediatric psychosis?
Erin Carlton, Tatiana Falcone, Ayush Batra, Vince Fazio, Kathleen Franco, and Damir Janigro
Abstract 7: Brain, heart, and education
Linda Bryant Caviness, PhD
Abstract 8: Tobacco smoke mediates a monocytic and endothelial proinflammatory activation that synergistically affects BBB integrity
L. Cucullo, T. Sathe, M. Hossain, and D. Janigro
Abstract 9: Dynamic changes in ECG predict poor outcome after aneurysmal subarachnoid hemorrhage (aSAH)
H.A. Elsharkawy, MD; S.M. El Hadi, MD, PhD; J.E. Tetzlaff, MD; and J.J. Provencio, MD, FCCM
Abstract 10: Mechanism studies of malformation of cortical development by prenatal exposure of combined methylazoxymethanol and thalidomide
Q. Fan, S. Ramakrishna, N. Marchi, V. Fazio, K. Hallene, and D. Janigro
Abstract 11: Proapolipoprotein A1 demonstrates improved potential as a serum marker for brain metastases without vascular disease interference
Vince Fazio, Peter Mazzone, Nicola Marchi, Thomas Masaryk, and Damir Janigro
Abstract 12: Biofeedback-assisted stress management training to reverse myocardial remodeling in patients with end-stage heart failure
Dana L. Frank, BS; Mary E. Klecka, BA; Jerry Kiffer, MA; Heather Henrickson, PhD; Michael G. McKee, PhD; and Christine S. Moravec, PhD
Abstract 13: Nitric oxide and arginine metabolism in depression: Effect of a serotonin-norepinephrine reuptake inhibitor
Angelos Halaris, John Piletz, Omer Iqbal, Debra Hoppensteadt, Jawed Fareed, He Zhu, James Sinacore, and C. Lindsay DeVane
Abstract 14: Association between excessive daytime sleepiness and oxygen desaturation in obstructive sleep apnea syndrome: Nadir oxygen saturation vs mean oxygen saturation vs time spent below 90% oxygen saturation—which is important?
Nattapong Jaimchariyatam, MD, and Kumar Budur, MD
Abstract 15: Endotoxin preconditioning of the CNS: Microglia activation and neuroprotection
Walid Jalabi, Ranjan Dutta, Yongming Jin, Gerson Criste, Xinghua Yin, Grahame J. Kidd, and Bruce D. Trapp
Abstract 16: Pilot of stress reduction strategies for patients after a coronary event
R. Lindquist, D. Windenburg, K. Savik, and U. Bronas
Abstract 17: Cerebrovascular substrates of seizures after cardiopulmonary bypass
Rebecca O’Dwyer, Tim Wehner, Dileep Nair, Giulia Betto, Nicola Marchi, and Damir Janigro
Abstract 18: Depression and whole blood serotonin in patients with coronary heart disease from the heart and soul study
Lawson Wulsin, Dominique Musselman, Christian Otte, Erica Bruce, Sadia Ali, and Mary Whooley
Abstract 19: Gender differences prominent in linking anxiety to long-term mortality among the elderly
Jianping Zhang, MD, PhD; Boaz Kahana, PhD; Eva Kahana, PhD; Bo Hu, PhD; and Leo Pozuelo, MD
Abstract 20: Temporal lobe and sinus node: A case report provides evidence for bidirectional effects
Rebecca O’Dwyer, MD; Andreas Alexopoulos, MD, MPH; Walid Saliba, MD; Imad Najm, MD; and Richard Burgess, MD, PhD
Supplement Editor:
Marc S. Penn, MD, PhD
Contents
Preface
Earl E. Bakken, MD, HonC, Hon DSc (3), Hon DHL (2)
Introduction: Heart-brain medicine: Update 2008
Marc S. Penn, MD, PhD, and Earl E. Bakken, MD, HonC, Hon DSc (3), Hon DHL (2)
Bakken Lecture: The brain, the heart, and therapeutic hypothermia
Patrick M. Kochanek, MD
Session 1: Pathways Involved in Neuromodulation of Risks in Coronary Artery Disease
Depression and heart rate variability in patients with coronary heart disease
Robert M. Carney, PhD, and Kenneth E. Freedland, PhD
Autonomic function and prognosis
Michael S. Lauer, MD
Vagal tone and the inflammatory reflex
Julian F. Thayer, PhD
Inflammation, atherosclerosis, and arterial thrombosis: Role of the scavenger receptors CD36
Roy L. Silverstein, MD
Pioneer Award Address: Ignorance isn't biased: Comments on receiving the Pioneer Award
David S. Goldstein, MD, PhD
Session II: Measures and Strategies for Modulation of Heart-Brain Interactions
Heart rate variability with deep breathing as a clinical test of cardiovagal function
Robert W. Shields, Jr, MD
Basic research models for the study of underlying mechanisms of electrical neuromodulation and ischemi heart-brain interactions
Mike J.L. DeJongste, MD, PhD, FESC; Gert J. TerHorst, PhD; and Robert D. Foreman, PhD
Session III: Annual Review of Key Papers in Heart-Brain Medicine
Key papers in the field published during the year prior to the Summit were discussed; two of those papers are reported here.
Cardiac sympathetic denervation preceding motor signs in Parkinson disease
David S. Goldstein, MD, PhD; Yehonatan Sharabi, MD; Barbara I. Karp, MD; Oladi Bentho; Ahmed Saleem, MD; Karel Pacak, MD, PhD; and Graeme Eisenhofer, PhD
Supine low-frequency power of heart rate variability reflects baroreflex function, not cardiac sympathetic innervation
Jeffrey P. Moak, MD; David S. Goldstein, MD, PhD; Basil A. Eldadah, MD, PhD; Ahmed Saleem, MD; Courtney Holmes, CMT; Sandra Pechnik, RN; and Yehonatan Sharabi, MD
Session IV: Code Lavender—Strategies for Implementing Heart-Brain Medicine
Is posttraumatic stress disorder related to development of heart disease? An update
Laura D. Kubzansky, PhD, and Karestan C. Koenen, PhD
Creating a healing environment: Rationale and research overview
Jone Geimer-Flanders, DO
Redesigning the neurocritical care unit to enhance family participation and improve outcomes
Owen Samuels, MD
Session V: Insights into Neuromodulation of Cardiovascular Function
Neuromodulation of cardiac pain and cerebral vasculature: Neural mechanisms
Robert D. Foreman, PhD, and Chao Qin, MD, PhD
Pinacidil induces vascular dilation and hyperemia in vivo and does not impact biophysical properties of neurons and astrocytes in vitro
Rosa Cao; Bryan T. Higashikubo; Jessica Cardin; Ulf Knoblich; Raddy Ramos, PhD; Mark T. Nelson, PhD; Christopher I. Moore, PhD; and Joshua C. Brumberg, PhD
The polyvagal theory: New insights into adaptive reactions of the autonomic nervous system
Stephen W. Porges, PhD
Poster Abstracts
Abstract 1: Insulin use does not protect against restenosis in diabetic patients presenting with acute coronary syndrome or symptomatic angina
Matthew C. Becker, MD; John M. Galla, MD; Saif Anwaruddin, MD; Samir Kapadia, MD; and Richard A. Krasuski, MD
Abstract 2: Postoperative statin use and lower LDL cholesterol concentration are associated with reduced incidence of stroke
Matthew C. Becker, John M. Galla, Ryan P. Daly, Femi Philip, Peter Zimbwa, Stephen O. Chen, Chen H. Chow, Tingfei Hu, Richard A. Krasuski, and Arman T. Askari
Abstract 3: Brain edema and blood-brain barrier leakage influence antiepileptic drug levels
Giulia Betto, Vincent Fazio, Damir Janigro, and Chaitali Ghosh
Abstract 4: CPAP treatment vs conservative treatment in mild obstructive sleep apnea: Implications on cardiovascular morbidity
Kumar Budur, MD, and Nattapong Jaimchariyatam, MD
Abstract 5: New bioinformatics program identifies behavioral medicine interventions for epidemic cardiovascular disease in the developing world: Analysis of multidisciplinary findings for launching a new global public health initiative in heart-brain medicine
William C. Bushell, PhD
Abstract 6: Do systemic inflammation and blood-brain barrier failure play a role in pediatric psychosis?
Erin Carlton, Tatiana Falcone, Ayush Batra, Vince Fazio, Kathleen Franco, and Damir Janigro
Abstract 7: Brain, heart, and education
Linda Bryant Caviness, PhD
Abstract 8: Tobacco smoke mediates a monocytic and endothelial proinflammatory activation that synergistically affects BBB integrity
L. Cucullo, T. Sathe, M. Hossain, and D. Janigro
Abstract 9: Dynamic changes in ECG predict poor outcome after aneurysmal subarachnoid hemorrhage (aSAH)
H.A. Elsharkawy, MD; S.M. El Hadi, MD, PhD; J.E. Tetzlaff, MD; and J.J. Provencio, MD, FCCM
Abstract 10: Mechanism studies of malformation of cortical development by prenatal exposure of combined methylazoxymethanol and thalidomide
Q. Fan, S. Ramakrishna, N. Marchi, V. Fazio, K. Hallene, and D. Janigro
Abstract 11: Proapolipoprotein A1 demonstrates improved potential as a serum marker for brain metastases without vascular disease interference
Vince Fazio, Peter Mazzone, Nicola Marchi, Thomas Masaryk, and Damir Janigro
Abstract 12: Biofeedback-assisted stress management training to reverse myocardial remodeling in patients with end-stage heart failure
Dana L. Frank, BS; Mary E. Klecka, BA; Jerry Kiffer, MA; Heather Henrickson, PhD; Michael G. McKee, PhD; and Christine S. Moravec, PhD
Abstract 13: Nitric oxide and arginine metabolism in depression: Effect of a serotonin-norepinephrine reuptake inhibitor
Angelos Halaris, John Piletz, Omer Iqbal, Debra Hoppensteadt, Jawed Fareed, He Zhu, James Sinacore, and C. Lindsay DeVane
Abstract 14: Association between excessive daytime sleepiness and oxygen desaturation in obstructive sleep apnea syndrome: Nadir oxygen saturation vs mean oxygen saturation vs time spent below 90% oxygen saturation—which is important?
Nattapong Jaimchariyatam, MD, and Kumar Budur, MD
Abstract 15: Endotoxin preconditioning of the CNS: Microglia activation and neuroprotection
Walid Jalabi, Ranjan Dutta, Yongming Jin, Gerson Criste, Xinghua Yin, Grahame J. Kidd, and Bruce D. Trapp
Abstract 16: Pilot of stress reduction strategies for patients after a coronary event
R. Lindquist, D. Windenburg, K. Savik, and U. Bronas
Abstract 17: Cerebrovascular substrates of seizures after cardiopulmonary bypass
Rebecca O’Dwyer, Tim Wehner, Dileep Nair, Giulia Betto, Nicola Marchi, and Damir Janigro
Abstract 18: Depression and whole blood serotonin in patients with coronary heart disease from the heart and soul study
Lawson Wulsin, Dominique Musselman, Christian Otte, Erica Bruce, Sadia Ali, and Mary Whooley
Abstract 19: Gender differences prominent in linking anxiety to long-term mortality among the elderly
Jianping Zhang, MD, PhD; Boaz Kahana, PhD; Eva Kahana, PhD; Bo Hu, PhD; and Leo Pozuelo, MD
Abstract 20: Temporal lobe and sinus node: A case report provides evidence for bidirectional effects
Rebecca O’Dwyer, MD; Andreas Alexopoulos, MD, MPH; Walid Saliba, MD; Imad Najm, MD; and Richard Burgess, MD, PhD
Platelet Response in Acute Coronary Syndromes
Supplement Editors:
Deepak L. Bhatt, MD, MPH, and W. Frank Peacock, MD
Contents
Importance of platelets and platelet response in acute coronary syndromes
Kandice Kottke-Marchant, MD, PhD
Novel antiplatelet strategies in acute coronary syndromes
Marc S. Sabatine, MD, MPH
The current state of antiplatelet therapy in acute coronary syndromes: The data and the real word
John H. Alexander, MD, MHSc
Platelet response in practice: Applying new insights and tools for testing and treatment
Deepak L. Bhatt, MD, MPH; Kandice Kottke-Marchant, MD, PhD; John H. Alexander, MD, MHSc; W. Frank Peacock, MD; and Marc S. Sabatine, MD, MPH
Supplement Editors:
Deepak L. Bhatt, MD, MPH, and W. Frank Peacock, MD
Contents
Importance of platelets and platelet response in acute coronary syndromes
Kandice Kottke-Marchant, MD, PhD
Novel antiplatelet strategies in acute coronary syndromes
Marc S. Sabatine, MD, MPH
The current state of antiplatelet therapy in acute coronary syndromes: The data and the real word
John H. Alexander, MD, MHSc
Platelet response in practice: Applying new insights and tools for testing and treatment
Deepak L. Bhatt, MD, MPH; Kandice Kottke-Marchant, MD, PhD; John H. Alexander, MD, MHSc; W. Frank Peacock, MD; and Marc S. Sabatine, MD, MPH
Supplement Editors:
Deepak L. Bhatt, MD, MPH, and W. Frank Peacock, MD
Contents
Importance of platelets and platelet response in acute coronary syndromes
Kandice Kottke-Marchant, MD, PhD
Novel antiplatelet strategies in acute coronary syndromes
Marc S. Sabatine, MD, MPH
The current state of antiplatelet therapy in acute coronary syndromes: The data and the real word
John H. Alexander, MD, MHSc
Platelet response in practice: Applying new insights and tools for testing and treatment
Deepak L. Bhatt, MD, MPH; Kandice Kottke-Marchant, MD, PhD; John H. Alexander, MD, MHSc; W. Frank Peacock, MD; and Marc S. Sabatine, MD, MPH
Importance of platelets and platelet response in acute coronary syndromes
PLATELET FUNCTION
Platelets are non-nucleated cells produced by megakaryocytes, which are very large cells (50 to 100 μm in diameter) found in bone marrow. The megakaryocyte surface membrane forms protoplatelet extensions from which platelets “bud off” and are emitted into the circulation, where they number approximately 200,000 to 400,000 per microliter of blood.
Platelet activation
Platelets play a crucial role in the vascular response to injury, and activation of platelets has long been recognized as an important step. Platelets release dense granules that contain the nucleotide adenosine diphosphate (ADP), which activates other platelets. They also possess alpha granules, which contain proteins and protein mediators (eg, platelet-derived growth factor, platelet factor 4) that are involved in inflammatory processes. The platelet surface is coated with hundreds of thousands of receptors for other cells, including activated vascular wall cells and extracellular matrix proteins. Platelets possess an affinity for adherence, especially to injured vessel walls, where they release their granule contents and then aggregate. These properties promote platelets’ involvement in many vascular processes, including ACS, as will be explored below.
Platelets exist in a nonactivated state and are drawn passively into areas of vascular injury. Initially, they adhere to proteins such as von Willebrand factor, which is a large extracellular matrix protein produced by endothelial cells. The platelet glycoprotein Ib/IX/V binds to von Willebrand factor, forming a loose association that results in platelets rolling on the surface of the vessel wall. As a multimer, von Willebrand factor exists in one subunit that is dimerized and then polymerized, making it an ideal substrate for platelets because of the multiple substrates to which platelets can adhere.
Platelet fibrinogen receptor
The platelet fibrinogen receptor (glycoprotein IIb/IIIa receptor) is an αIIbβ3 integrin that binds to arginine-glycine-aspartic acid (RGD) epitopes of proteins, such as fibrinogen. Fibrinogen has a two-dimensional symmetry, with RGD groups on both ends of the molecule, which makes it an ideal molecule for linking platelet to platelet.
von Willebrand factor has RGD groups, as do both fibronectin and glycoprotein IIb/IIIa vitronectin, and can therefore bind to many plasma and extracellular matrix proteins. The glycoprotein IIb/IIIa receptor is inactive in resting platelets. It becomes activated during the platelet activation process and binds to fibrinogen, which bridges to other platelets, causing aggregation.
ADP receptors
Various receptors on platelet surfaces are responsible for platelet activation. One is a family of receptors for ADP. As ADP is released from platelets, it can then activate other platelets by binding to the receptors. The ADP receptor P2Y12 signals through G protein pathways and is coupled to adenylate cyclase, an enzyme that catalyzes the conversion of adenosine triphosphate to cyclic adenosine monophosphate (cAMP). High levels of cAMP inhibit platelet function; ADP binding to P2Y12 shuts down adenylate cyclase, which leads to phosphoinositide 3-kinase activation and accelerated aggregation and platelet release.
A final notable factor in the mediation of platelet activation and aggregation is phospholipase A2, which liberates arachidonic acid from the platelet membrane, metabolizing it through cyclooxygenase and thromboxane synthase to generate thromoboxane A2, which leads to release of platelet granule contents and aggregation of other platelets.
PLATELET FUNCTION TESTS
Platelet function assays are inherently variable because they measure cell function rather than a single analyte. Several new platelet testing devices have come to market with the goal of ease of use; many can now be used at the bedside to measure platelet function.
Platelet count
In my view, the platelet count remains one of the best tests for assessing bleeding risk, as a low platelet count is one of the most common causes of bleeding. However, the platelet count is not a functional assay because it does not evaluate other platelet functions.
Screening tests
Screening tests, or global tests for platelet function, do not identify specific causes of platelet dysfunction but combine measurement of many different aspects of platelet function, such as adhesion, aggregation and granule release.
Bleeding time. The bleeding time is an archaic test because of the poor correlation between bleeding time and bleeding disorders or thrombotic disorders. Its utility in measuring platelet function is therefore highly limited.
PFA-100. The PFA-100 Platelet Function Analyzer system (PFA-100) is one example of a global platelet function assay that measures multiple platelet functions, including platelet adhesion and aggregation. The instrument, which is about the size of a bread box, uses a citrate-anticoagulated whole blood specimen to measure platelet reaction in a high-shear environment. Blood travels at high shear rates through membranes coated with either collagen and ADP or collagen and epinephrine (epinephrine receptors exist on platelet surfaces). Platelets adhere to the membranes and then activate, aggregate, and occlude a small aperture in the center of each membrane, yielding a measurable closure time.
Since the PFA-100 was developed before the availability of the thienopyridine antiplatelet drugs, its utility lies not in monitoring the effects of those agents but in its ability to detect aspirin-induced platelet dysfunction or intrinsic platelet function disorders. An abnormal epinephrine cartridge closure time in the presence of a normal ADP cartridge closure time indicates aspirin-induced platelet dysfunction. An abnormal closure time on both measures is indicative of von Willebrand disease or a platelet defect such as Glanzmann thrombasthenia or Bernard-Soulier syndrome.
Specific functional tests
Whole blood platelet aggregation is typically a high-complexity laboratory test. Recently, self-contained assay platforms that can measure whole blood aggregation have been developed. These are applicable for smaller hospitals and near-patient settings. One such rapid platelet function analyzer, known commercially as VerifyNow, offers point-of-care assessment of platelet function. The instrument, which is the size of a telephone answering machine, operates by a principle similar to that of optical platelet aggregation: platelet function is measured by the rate and extent of change in light transmittance in response to the introduction of agonists specific to various antiplatelet medications. Low light transmittance indicates a blood sample with inhibited platelet function; high light transmittance indicates normal platelet function.
Measurement of VASP phosphorylation. Vasodilator-stimulated phosphoprotein (VASP) is an intracellular platelet protein that is nonphosphorylated in basal state. The phosphorylation of VASP depends on the level of activation of the P2Y12 receptor, a target of thienopyridine drugs. Thus, measuring VASP phosphorylation by flow cytometry using citrated whole blood can be a highly specific indicator of the action and efficacy of clopidogrel and other thienopyridine drugs.
A flow cytometry assay that measures VASP phosphorylation requires a whole blood sample that is incubated with ADP to measure what is called the platelet reactivity index. Adding ADP to whole blood stimulates adenylate cyclase, lowering cAMP and shutting off protein kinase, which results in low levels of VASP phosphorylation. Thus, if VASP is phosphorylated, the platelets are inhibited; if VASP is not phosphorylated, the platelets are activated. A satisfactory therapeutic response to clopidogrel or another thienopyridine drug produces a low platelet reactivity index, reflecting platelet inhibition.
ROLE OF PLATELETS IN ATHEROSCLEROSIS
Platelets serve major functions in three key aspects of atherosclerosis: atherogenesis, inflammation, and atherothrombosis.
Atherogenesis
Platelets play a pivotal role in atherogenesis.1 They release matrix metalloproteinases that are involved in degrading the matrix in atherosclerotic plaques. Moreover, they contain and release chemokines and growth factors, including:
- RANTES, a chemokine that stimulates monocytes and T cells to increase the production of monocyte inflammatory mediators
- Platelet-derived growth factor, which stimulates the migration and proliferation of smooth muscle cells
- Transforming growth factor–β, which also stimulates proliferation of smooth muscle cells.
Inflammation
Activated platelets release inflammatory mediators and thereby change the adhesive and chemotactic properties of endothelial cells. Likewise, mediators derived from inflammatory cells (neutrophils) can affect platelet function.
Platelet-derived mediators include the following:
- Pro‑interleukin (IL)-β, which triggers the synthesis of E-selectin that enables endothelial cells to interact with leukocytes
- Thromboxane A2, which increases neutrophil adhesion to facilitate platelet aggregation
- Platelet-derived growth factor and platelet factor 4, which increase neutrophil chemotaxis (the ability of neutrophils to infiltrate atherosclerotic plaque)
- CD40 ligand, a protein expressed on platelets that induces inflammatory responses in the endothelium
- P-selectin, a cell adhesion molecule expressed on activated platelets that enhances the adhesion of monocytes on activated endothelial cells.
Among the neutrophil-derived mediators, some—such as superoxide and leukotrienes—enhance platelet activation, whereas elastases inhibit platelet activation.
Overall, once inflammation begins in an atherosclerotic plaque, much reciprocal platelet activation can occur, so that the inflammatory process can become a feed-forward loop to eventually promote atherothrombosis.
Atherothrombosis
In the last stage of the atherosclerotic process, platelet enzymes that degrade the matrix may make plaques vulnerable to rupture by creating fissures in the fibrous plaque cap. This exposes the lipid-rich core, which contains a significant amount of thromboplastin. Exposure to the extracellular matrix can lead to further platelet adhesion, activation, and aggregation. The development of a platelet thrombus is usually one of the ultimate steps in atherothrombosis leading to ACS, including MI.
ROLE OF PLATELETS IN ACUTE CORONARY SYNDROMES: WHAT IS THE EVIDENCE?
How predictive is an elevated platelet count?
However, another study conducted in a slightly different population—1,616 patients with non‑ST-segment-elevation MI/unstable angina—found no correlation between platelet count (by quintiles) and death at 60 months.3 The lowest mortality was observed in patients with a platelet count in the second-lowest quintile, although the highest mortality was indeed observed in the quintile of patients with the lowest platelet counts.3
The differing results in the above two studies suggest that additional platelet factors, beyond platelet count, contribute to the risk of adverse outcomes following ACS.
Platelet hyperreactivity and outcomes in ACS
Platelet hyperreactivity—ie, residual platelet activity despite antiplatelet therapy—appears to be involved in the spectrum of ACS. A recent study evaluated the association between hyperreactivity of platelets to ADP and outcomes in 600 patients with stable cardiovascular disease who were on aspirin therapy.4 Hyperreactivity was defined as a collagen/ADP closure time of less than 90 seconds on the PFA-100 system (short collagen/ADP closure time). On receiver operating characteristic (ROC) curve analysis, a short collagen/ADP closure time served as a significant predictor of recurrent events (relative risk [RR] = 3.65; 95% CI, 1.76–7.57) and death (RR = 6.56; 95% CI, 1.93–22.35) compared with a closure time of 90 seconds or greater. The authors concluded that there appears to be a subgroup of patients with stable cardiovascular disease who have an increased risk of major adverse events associated with platelet hyperreactivity.4
An earlier study by Harrison et al assessed platelet function using the PFA-100 in 78 patients presenting with acute chest pain classified as MI, unstable angina, or nonspecific chest pain.5 Using the PFA-100, they found shorter collagen/ADP closure times and higher levels of von Willebrand factor in subjects with MI compared with those who had unstable angina or nonspecific chest pain.5 Fuchs et al reported a similar association between von Willebrand factor and outcomes in 208 patients with ACS,6 raising the possibility that von Willebrand factor, through its association with increased platelet adhesion and activation, may be a major contributor to risk in ACS.
Similarly, an association between platelet hyperreactivity and cardiovascular events has been suggested in patients with type 2 diabetes. In a 2007 study of 173 patients with type 2 diabetes and coronary artery disease receiving dual antiplatelet therapy (aspirin plus clopidogrel), the 2-year risk of major cardiovascular events was significantly higher in those in the highest quartile of platelet aggregation compared with those in the lower three quartiles (hazard ratio = 3.35; 95% CI, 1.68–6.66).7 In a separate study, Serebruany et al measured platelet activity by five different testing methods in 822 patients with coronary artery disease and found significantly higher platelet hyperreactivity by all methods in those patients who had diabetes (n = 257) than in those who did not (n = 565).8
Marcucci et al recently examined the relationship between clinical characteristics and residual platelet activity in 386 patients with ACS on dual antiplatelet therapy (aspirin plus clopidogrel).9 The presence of residual platelet activity (determined by platelet aggregation in response to the agonists arachidonic acid and ADP, as well as by the PFA-100) was associated with significantly higher inflammatory status, as determined by leukocyte count and erythrocyte sedimentation rate. The same association was observed among a subset of patients in this study undergoing percutaneous coronary intervention (PCI) who were receiving dual antiplatelet therapy; additionally, residual platelet activity was associated with a significantly higher incidence of diabetes and a significantly lower ejection fraction in this subset.9
Platelet hyperreactivity while on dual antiplatelet therapy (aspirin plus clopidogrel) was also found to be predictive of clinical outcome in a study of 195 patients with non-ST-elevation MI undergoing PCI.10 Hyporesponse to antiplatelet therapy, as measured by a high VASP platelet reactivity index (PRI), predicted an increased risk of recurrent ischemic events within 30 days of PCI. Using ROC curve analysis, the investigators found that a VASP PRI cutoff value of 53% (ie, a high PRI [> 53%] indicates residual platelet activity despite clopidogrel) had a sensitivity of 93%, a specificity of 50%, a positive predictive value of 12%, and a negative predictive value of 99% for ischemic events.10 Similarly, among 144 patients undergoing PCI assessed for decreased platelet reactivity to a loading dose of clopidogrel, Bonello et al also found that a VASP PRI greater than 50% was optimal for predicting major adverse cardiovascular events: all 21 events in the study occurred among patients whose VASP PRI was in the highest four quintiles.11
CONCLUSIONS AND GENERAL ASSESSMENT OF PLATELET FUNCTION TESTS
Platelets clearly are involved in the pathogenesis of atherothrombosis. Accumulating evidence suggests that both an elevated platelet count and platelet hyperreactivity (residual platelet activity despite dual antiplatelet therapy) may be associated with adverse cardiovascular events in patients with ACS.
Platelet function can be measured using several different assays and measures of platelet activation. The best assays for measuring residual platelet activity in the setting of antiplatelet therapy are still being defined, as are their predictive values. Platelet aggregation remains the gold standard. The PFA-100 may detect overall platelet hyperreactivity despite the use of antiplatelet therapy, and is attracting increasing use for this purpose. VASP phosphorylation may be a good assay for detecting P2Y12 inhibition but is limited to thienopyridines in terms of detecting platelet hyperreactivity. For predicting adverse cardiac events, ROC curve analysis should be used to objectively define cutoff values for platelet hyperreactivity as opposed to reliance on arbitrarily defined cutoff values.
Moving forward, standard testing protocols for platelet aggregation clearly are needed to achieve consistency among studies.
- Davì G, Patrono C. Platelet activation and atherothrombosis. N Engl J Med 2007; 357:2482–2494.
- Ly HQ, Kirtane AJ, Murphy SA, et al. Association of platelet counts on presentation and clinical outcomes in ST-elevation myocardial infarction (from the TIMI Trials). Am J Cardiol 2006; 98:1–5.
- Mueller C, Neumann FJ, Hochholzer W, et al. The impact of platelet count on mortality in unstable angina/non-ST-segment elevation myocardial infarction. Am Heart J 2006; 151:1214.e1–7.
- Christie DJ, Kottke-Marchant K, Gorman RT. Hypersensitivity of platelets to adenosine diphosphate in patients with stable cardiovascular disease predicts major adverse events despite antiplatelet therapy. Platelets 2008; 19:104–110.
- Harrison P, Mackie I, Mathur A, et al. Platelet hyperfunction in acute coronary syndromes. Blood Coagul Fibrinolysis 2005; 16:557–562.
- Fuchs I, Frossard M, Spiel A, Riedmüller E, Laggner AN, Jilma B. Platelet function in patients with acute coronary syndrome (ACS) predicts recurrent ACS. J Thromb Haemost 2006; 4:2547–2552.
- Angiolillo DJ, Bernardo E, Sabaté M, et al. Impact of platelet reactivity on cardiovascular outcomes in patients with type 2 diabetes mellitus and coronary artery disease. J Am Coll Cardiol 2007; 50:1541–1547.
- Serebruany V, Pokov I, Kuliczkowski W, Chesebro J, Badimon J. Baseline platelet activity and response after clopidogrel in 257 diabetics among 822 patients with coronary artery disease. Thromb Haemost 2008; 100:76–82.
- Marcucci R, Gori AM, Paniccia R, et al. Residual platelet reactivity is associated with clinical and laboratory characteristics in patients with ischemic heart disease undergoing PCI on dual antiplatelet therapy. Atherosclerosis 2007; 195:e217–e223.
- Frere C, Cuisset T, Quilici J, et al. ADP-induced platelet aggregation and platelet reactivity index VASP are good predictive markers for clinical outcomes in non-ST elevation acute coronary syndrome. Thromb Haemost 2007; 98:838–843.
- Bonello L, Paganelli F, Arpin-Bornet M, et al. Vasodilator-stimulated phosphoprotein phosphorylation analysis prior to percutaneous coronary intervention for exclusion of postprocedural major adverse cardiovascular events. J Thromb Haemost 2007; 5:1630–1636.
PLATELET FUNCTION
Platelets are non-nucleated cells produced by megakaryocytes, which are very large cells (50 to 100 μm in diameter) found in bone marrow. The megakaryocyte surface membrane forms protoplatelet extensions from which platelets “bud off” and are emitted into the circulation, where they number approximately 200,000 to 400,000 per microliter of blood.
Platelet activation
Platelets play a crucial role in the vascular response to injury, and activation of platelets has long been recognized as an important step. Platelets release dense granules that contain the nucleotide adenosine diphosphate (ADP), which activates other platelets. They also possess alpha granules, which contain proteins and protein mediators (eg, platelet-derived growth factor, platelet factor 4) that are involved in inflammatory processes. The platelet surface is coated with hundreds of thousands of receptors for other cells, including activated vascular wall cells and extracellular matrix proteins. Platelets possess an affinity for adherence, especially to injured vessel walls, where they release their granule contents and then aggregate. These properties promote platelets’ involvement in many vascular processes, including ACS, as will be explored below.
Platelets exist in a nonactivated state and are drawn passively into areas of vascular injury. Initially, they adhere to proteins such as von Willebrand factor, which is a large extracellular matrix protein produced by endothelial cells. The platelet glycoprotein Ib/IX/V binds to von Willebrand factor, forming a loose association that results in platelets rolling on the surface of the vessel wall. As a multimer, von Willebrand factor exists in one subunit that is dimerized and then polymerized, making it an ideal substrate for platelets because of the multiple substrates to which platelets can adhere.
Platelet fibrinogen receptor
The platelet fibrinogen receptor (glycoprotein IIb/IIIa receptor) is an αIIbβ3 integrin that binds to arginine-glycine-aspartic acid (RGD) epitopes of proteins, such as fibrinogen. Fibrinogen has a two-dimensional symmetry, with RGD groups on both ends of the molecule, which makes it an ideal molecule for linking platelet to platelet.
von Willebrand factor has RGD groups, as do both fibronectin and glycoprotein IIb/IIIa vitronectin, and can therefore bind to many plasma and extracellular matrix proteins. The glycoprotein IIb/IIIa receptor is inactive in resting platelets. It becomes activated during the platelet activation process and binds to fibrinogen, which bridges to other platelets, causing aggregation.
ADP receptors
Various receptors on platelet surfaces are responsible for platelet activation. One is a family of receptors for ADP. As ADP is released from platelets, it can then activate other platelets by binding to the receptors. The ADP receptor P2Y12 signals through G protein pathways and is coupled to adenylate cyclase, an enzyme that catalyzes the conversion of adenosine triphosphate to cyclic adenosine monophosphate (cAMP). High levels of cAMP inhibit platelet function; ADP binding to P2Y12 shuts down adenylate cyclase, which leads to phosphoinositide 3-kinase activation and accelerated aggregation and platelet release.
A final notable factor in the mediation of platelet activation and aggregation is phospholipase A2, which liberates arachidonic acid from the platelet membrane, metabolizing it through cyclooxygenase and thromboxane synthase to generate thromoboxane A2, which leads to release of platelet granule contents and aggregation of other platelets.
PLATELET FUNCTION TESTS
Platelet function assays are inherently variable because they measure cell function rather than a single analyte. Several new platelet testing devices have come to market with the goal of ease of use; many can now be used at the bedside to measure platelet function.
Platelet count
In my view, the platelet count remains one of the best tests for assessing bleeding risk, as a low platelet count is one of the most common causes of bleeding. However, the platelet count is not a functional assay because it does not evaluate other platelet functions.
Screening tests
Screening tests, or global tests for platelet function, do not identify specific causes of platelet dysfunction but combine measurement of many different aspects of platelet function, such as adhesion, aggregation and granule release.
Bleeding time. The bleeding time is an archaic test because of the poor correlation between bleeding time and bleeding disorders or thrombotic disorders. Its utility in measuring platelet function is therefore highly limited.
PFA-100. The PFA-100 Platelet Function Analyzer system (PFA-100) is one example of a global platelet function assay that measures multiple platelet functions, including platelet adhesion and aggregation. The instrument, which is about the size of a bread box, uses a citrate-anticoagulated whole blood specimen to measure platelet reaction in a high-shear environment. Blood travels at high shear rates through membranes coated with either collagen and ADP or collagen and epinephrine (epinephrine receptors exist on platelet surfaces). Platelets adhere to the membranes and then activate, aggregate, and occlude a small aperture in the center of each membrane, yielding a measurable closure time.
Since the PFA-100 was developed before the availability of the thienopyridine antiplatelet drugs, its utility lies not in monitoring the effects of those agents but in its ability to detect aspirin-induced platelet dysfunction or intrinsic platelet function disorders. An abnormal epinephrine cartridge closure time in the presence of a normal ADP cartridge closure time indicates aspirin-induced platelet dysfunction. An abnormal closure time on both measures is indicative of von Willebrand disease or a platelet defect such as Glanzmann thrombasthenia or Bernard-Soulier syndrome.
Specific functional tests
Whole blood platelet aggregation is typically a high-complexity laboratory test. Recently, self-contained assay platforms that can measure whole blood aggregation have been developed. These are applicable for smaller hospitals and near-patient settings. One such rapid platelet function analyzer, known commercially as VerifyNow, offers point-of-care assessment of platelet function. The instrument, which is the size of a telephone answering machine, operates by a principle similar to that of optical platelet aggregation: platelet function is measured by the rate and extent of change in light transmittance in response to the introduction of agonists specific to various antiplatelet medications. Low light transmittance indicates a blood sample with inhibited platelet function; high light transmittance indicates normal platelet function.
Measurement of VASP phosphorylation. Vasodilator-stimulated phosphoprotein (VASP) is an intracellular platelet protein that is nonphosphorylated in basal state. The phosphorylation of VASP depends on the level of activation of the P2Y12 receptor, a target of thienopyridine drugs. Thus, measuring VASP phosphorylation by flow cytometry using citrated whole blood can be a highly specific indicator of the action and efficacy of clopidogrel and other thienopyridine drugs.
A flow cytometry assay that measures VASP phosphorylation requires a whole blood sample that is incubated with ADP to measure what is called the platelet reactivity index. Adding ADP to whole blood stimulates adenylate cyclase, lowering cAMP and shutting off protein kinase, which results in low levels of VASP phosphorylation. Thus, if VASP is phosphorylated, the platelets are inhibited; if VASP is not phosphorylated, the platelets are activated. A satisfactory therapeutic response to clopidogrel or another thienopyridine drug produces a low platelet reactivity index, reflecting platelet inhibition.
ROLE OF PLATELETS IN ATHEROSCLEROSIS
Platelets serve major functions in three key aspects of atherosclerosis: atherogenesis, inflammation, and atherothrombosis.
Atherogenesis
Platelets play a pivotal role in atherogenesis.1 They release matrix metalloproteinases that are involved in degrading the matrix in atherosclerotic plaques. Moreover, they contain and release chemokines and growth factors, including:
- RANTES, a chemokine that stimulates monocytes and T cells to increase the production of monocyte inflammatory mediators
- Platelet-derived growth factor, which stimulates the migration and proliferation of smooth muscle cells
- Transforming growth factor–β, which also stimulates proliferation of smooth muscle cells.
Inflammation
Activated platelets release inflammatory mediators and thereby change the adhesive and chemotactic properties of endothelial cells. Likewise, mediators derived from inflammatory cells (neutrophils) can affect platelet function.
Platelet-derived mediators include the following:
- Pro‑interleukin (IL)-β, which triggers the synthesis of E-selectin that enables endothelial cells to interact with leukocytes
- Thromboxane A2, which increases neutrophil adhesion to facilitate platelet aggregation
- Platelet-derived growth factor and platelet factor 4, which increase neutrophil chemotaxis (the ability of neutrophils to infiltrate atherosclerotic plaque)
- CD40 ligand, a protein expressed on platelets that induces inflammatory responses in the endothelium
- P-selectin, a cell adhesion molecule expressed on activated platelets that enhances the adhesion of monocytes on activated endothelial cells.
Among the neutrophil-derived mediators, some—such as superoxide and leukotrienes—enhance platelet activation, whereas elastases inhibit platelet activation.
Overall, once inflammation begins in an atherosclerotic plaque, much reciprocal platelet activation can occur, so that the inflammatory process can become a feed-forward loop to eventually promote atherothrombosis.
Atherothrombosis
In the last stage of the atherosclerotic process, platelet enzymes that degrade the matrix may make plaques vulnerable to rupture by creating fissures in the fibrous plaque cap. This exposes the lipid-rich core, which contains a significant amount of thromboplastin. Exposure to the extracellular matrix can lead to further platelet adhesion, activation, and aggregation. The development of a platelet thrombus is usually one of the ultimate steps in atherothrombosis leading to ACS, including MI.
ROLE OF PLATELETS IN ACUTE CORONARY SYNDROMES: WHAT IS THE EVIDENCE?
How predictive is an elevated platelet count?
However, another study conducted in a slightly different population—1,616 patients with non‑ST-segment-elevation MI/unstable angina—found no correlation between platelet count (by quintiles) and death at 60 months.3 The lowest mortality was observed in patients with a platelet count in the second-lowest quintile, although the highest mortality was indeed observed in the quintile of patients with the lowest platelet counts.3
The differing results in the above two studies suggest that additional platelet factors, beyond platelet count, contribute to the risk of adverse outcomes following ACS.
Platelet hyperreactivity and outcomes in ACS
Platelet hyperreactivity—ie, residual platelet activity despite antiplatelet therapy—appears to be involved in the spectrum of ACS. A recent study evaluated the association between hyperreactivity of platelets to ADP and outcomes in 600 patients with stable cardiovascular disease who were on aspirin therapy.4 Hyperreactivity was defined as a collagen/ADP closure time of less than 90 seconds on the PFA-100 system (short collagen/ADP closure time). On receiver operating characteristic (ROC) curve analysis, a short collagen/ADP closure time served as a significant predictor of recurrent events (relative risk [RR] = 3.65; 95% CI, 1.76–7.57) and death (RR = 6.56; 95% CI, 1.93–22.35) compared with a closure time of 90 seconds or greater. The authors concluded that there appears to be a subgroup of patients with stable cardiovascular disease who have an increased risk of major adverse events associated with platelet hyperreactivity.4
An earlier study by Harrison et al assessed platelet function using the PFA-100 in 78 patients presenting with acute chest pain classified as MI, unstable angina, or nonspecific chest pain.5 Using the PFA-100, they found shorter collagen/ADP closure times and higher levels of von Willebrand factor in subjects with MI compared with those who had unstable angina or nonspecific chest pain.5 Fuchs et al reported a similar association between von Willebrand factor and outcomes in 208 patients with ACS,6 raising the possibility that von Willebrand factor, through its association with increased platelet adhesion and activation, may be a major contributor to risk in ACS.
Similarly, an association between platelet hyperreactivity and cardiovascular events has been suggested in patients with type 2 diabetes. In a 2007 study of 173 patients with type 2 diabetes and coronary artery disease receiving dual antiplatelet therapy (aspirin plus clopidogrel), the 2-year risk of major cardiovascular events was significantly higher in those in the highest quartile of platelet aggregation compared with those in the lower three quartiles (hazard ratio = 3.35; 95% CI, 1.68–6.66).7 In a separate study, Serebruany et al measured platelet activity by five different testing methods in 822 patients with coronary artery disease and found significantly higher platelet hyperreactivity by all methods in those patients who had diabetes (n = 257) than in those who did not (n = 565).8
Marcucci et al recently examined the relationship between clinical characteristics and residual platelet activity in 386 patients with ACS on dual antiplatelet therapy (aspirin plus clopidogrel).9 The presence of residual platelet activity (determined by platelet aggregation in response to the agonists arachidonic acid and ADP, as well as by the PFA-100) was associated with significantly higher inflammatory status, as determined by leukocyte count and erythrocyte sedimentation rate. The same association was observed among a subset of patients in this study undergoing percutaneous coronary intervention (PCI) who were receiving dual antiplatelet therapy; additionally, residual platelet activity was associated with a significantly higher incidence of diabetes and a significantly lower ejection fraction in this subset.9
Platelet hyperreactivity while on dual antiplatelet therapy (aspirin plus clopidogrel) was also found to be predictive of clinical outcome in a study of 195 patients with non-ST-elevation MI undergoing PCI.10 Hyporesponse to antiplatelet therapy, as measured by a high VASP platelet reactivity index (PRI), predicted an increased risk of recurrent ischemic events within 30 days of PCI. Using ROC curve analysis, the investigators found that a VASP PRI cutoff value of 53% (ie, a high PRI [> 53%] indicates residual platelet activity despite clopidogrel) had a sensitivity of 93%, a specificity of 50%, a positive predictive value of 12%, and a negative predictive value of 99% for ischemic events.10 Similarly, among 144 patients undergoing PCI assessed for decreased platelet reactivity to a loading dose of clopidogrel, Bonello et al also found that a VASP PRI greater than 50% was optimal for predicting major adverse cardiovascular events: all 21 events in the study occurred among patients whose VASP PRI was in the highest four quintiles.11
CONCLUSIONS AND GENERAL ASSESSMENT OF PLATELET FUNCTION TESTS
Platelets clearly are involved in the pathogenesis of atherothrombosis. Accumulating evidence suggests that both an elevated platelet count and platelet hyperreactivity (residual platelet activity despite dual antiplatelet therapy) may be associated with adverse cardiovascular events in patients with ACS.
Platelet function can be measured using several different assays and measures of platelet activation. The best assays for measuring residual platelet activity in the setting of antiplatelet therapy are still being defined, as are their predictive values. Platelet aggregation remains the gold standard. The PFA-100 may detect overall platelet hyperreactivity despite the use of antiplatelet therapy, and is attracting increasing use for this purpose. VASP phosphorylation may be a good assay for detecting P2Y12 inhibition but is limited to thienopyridines in terms of detecting platelet hyperreactivity. For predicting adverse cardiac events, ROC curve analysis should be used to objectively define cutoff values for platelet hyperreactivity as opposed to reliance on arbitrarily defined cutoff values.
Moving forward, standard testing protocols for platelet aggregation clearly are needed to achieve consistency among studies.
PLATELET FUNCTION
Platelets are non-nucleated cells produced by megakaryocytes, which are very large cells (50 to 100 μm in diameter) found in bone marrow. The megakaryocyte surface membrane forms protoplatelet extensions from which platelets “bud off” and are emitted into the circulation, where they number approximately 200,000 to 400,000 per microliter of blood.
Platelet activation
Platelets play a crucial role in the vascular response to injury, and activation of platelets has long been recognized as an important step. Platelets release dense granules that contain the nucleotide adenosine diphosphate (ADP), which activates other platelets. They also possess alpha granules, which contain proteins and protein mediators (eg, platelet-derived growth factor, platelet factor 4) that are involved in inflammatory processes. The platelet surface is coated with hundreds of thousands of receptors for other cells, including activated vascular wall cells and extracellular matrix proteins. Platelets possess an affinity for adherence, especially to injured vessel walls, where they release their granule contents and then aggregate. These properties promote platelets’ involvement in many vascular processes, including ACS, as will be explored below.
Platelets exist in a nonactivated state and are drawn passively into areas of vascular injury. Initially, they adhere to proteins such as von Willebrand factor, which is a large extracellular matrix protein produced by endothelial cells. The platelet glycoprotein Ib/IX/V binds to von Willebrand factor, forming a loose association that results in platelets rolling on the surface of the vessel wall. As a multimer, von Willebrand factor exists in one subunit that is dimerized and then polymerized, making it an ideal substrate for platelets because of the multiple substrates to which platelets can adhere.
Platelet fibrinogen receptor
The platelet fibrinogen receptor (glycoprotein IIb/IIIa receptor) is an αIIbβ3 integrin that binds to arginine-glycine-aspartic acid (RGD) epitopes of proteins, such as fibrinogen. Fibrinogen has a two-dimensional symmetry, with RGD groups on both ends of the molecule, which makes it an ideal molecule for linking platelet to platelet.
von Willebrand factor has RGD groups, as do both fibronectin and glycoprotein IIb/IIIa vitronectin, and can therefore bind to many plasma and extracellular matrix proteins. The glycoprotein IIb/IIIa receptor is inactive in resting platelets. It becomes activated during the platelet activation process and binds to fibrinogen, which bridges to other platelets, causing aggregation.
ADP receptors
Various receptors on platelet surfaces are responsible for platelet activation. One is a family of receptors for ADP. As ADP is released from platelets, it can then activate other platelets by binding to the receptors. The ADP receptor P2Y12 signals through G protein pathways and is coupled to adenylate cyclase, an enzyme that catalyzes the conversion of adenosine triphosphate to cyclic adenosine monophosphate (cAMP). High levels of cAMP inhibit platelet function; ADP binding to P2Y12 shuts down adenylate cyclase, which leads to phosphoinositide 3-kinase activation and accelerated aggregation and platelet release.
A final notable factor in the mediation of platelet activation and aggregation is phospholipase A2, which liberates arachidonic acid from the platelet membrane, metabolizing it through cyclooxygenase and thromboxane synthase to generate thromoboxane A2, which leads to release of platelet granule contents and aggregation of other platelets.
PLATELET FUNCTION TESTS
Platelet function assays are inherently variable because they measure cell function rather than a single analyte. Several new platelet testing devices have come to market with the goal of ease of use; many can now be used at the bedside to measure platelet function.
Platelet count
In my view, the platelet count remains one of the best tests for assessing bleeding risk, as a low platelet count is one of the most common causes of bleeding. However, the platelet count is not a functional assay because it does not evaluate other platelet functions.
Screening tests
Screening tests, or global tests for platelet function, do not identify specific causes of platelet dysfunction but combine measurement of many different aspects of platelet function, such as adhesion, aggregation and granule release.
Bleeding time. The bleeding time is an archaic test because of the poor correlation between bleeding time and bleeding disorders or thrombotic disorders. Its utility in measuring platelet function is therefore highly limited.
PFA-100. The PFA-100 Platelet Function Analyzer system (PFA-100) is one example of a global platelet function assay that measures multiple platelet functions, including platelet adhesion and aggregation. The instrument, which is about the size of a bread box, uses a citrate-anticoagulated whole blood specimen to measure platelet reaction in a high-shear environment. Blood travels at high shear rates through membranes coated with either collagen and ADP or collagen and epinephrine (epinephrine receptors exist on platelet surfaces). Platelets adhere to the membranes and then activate, aggregate, and occlude a small aperture in the center of each membrane, yielding a measurable closure time.
Since the PFA-100 was developed before the availability of the thienopyridine antiplatelet drugs, its utility lies not in monitoring the effects of those agents but in its ability to detect aspirin-induced platelet dysfunction or intrinsic platelet function disorders. An abnormal epinephrine cartridge closure time in the presence of a normal ADP cartridge closure time indicates aspirin-induced platelet dysfunction. An abnormal closure time on both measures is indicative of von Willebrand disease or a platelet defect such as Glanzmann thrombasthenia or Bernard-Soulier syndrome.
Specific functional tests
Whole blood platelet aggregation is typically a high-complexity laboratory test. Recently, self-contained assay platforms that can measure whole blood aggregation have been developed. These are applicable for smaller hospitals and near-patient settings. One such rapid platelet function analyzer, known commercially as VerifyNow, offers point-of-care assessment of platelet function. The instrument, which is the size of a telephone answering machine, operates by a principle similar to that of optical platelet aggregation: platelet function is measured by the rate and extent of change in light transmittance in response to the introduction of agonists specific to various antiplatelet medications. Low light transmittance indicates a blood sample with inhibited platelet function; high light transmittance indicates normal platelet function.
Measurement of VASP phosphorylation. Vasodilator-stimulated phosphoprotein (VASP) is an intracellular platelet protein that is nonphosphorylated in basal state. The phosphorylation of VASP depends on the level of activation of the P2Y12 receptor, a target of thienopyridine drugs. Thus, measuring VASP phosphorylation by flow cytometry using citrated whole blood can be a highly specific indicator of the action and efficacy of clopidogrel and other thienopyridine drugs.
A flow cytometry assay that measures VASP phosphorylation requires a whole blood sample that is incubated with ADP to measure what is called the platelet reactivity index. Adding ADP to whole blood stimulates adenylate cyclase, lowering cAMP and shutting off protein kinase, which results in low levels of VASP phosphorylation. Thus, if VASP is phosphorylated, the platelets are inhibited; if VASP is not phosphorylated, the platelets are activated. A satisfactory therapeutic response to clopidogrel or another thienopyridine drug produces a low platelet reactivity index, reflecting platelet inhibition.
ROLE OF PLATELETS IN ATHEROSCLEROSIS
Platelets serve major functions in three key aspects of atherosclerosis: atherogenesis, inflammation, and atherothrombosis.
Atherogenesis
Platelets play a pivotal role in atherogenesis.1 They release matrix metalloproteinases that are involved in degrading the matrix in atherosclerotic plaques. Moreover, they contain and release chemokines and growth factors, including:
- RANTES, a chemokine that stimulates monocytes and T cells to increase the production of monocyte inflammatory mediators
- Platelet-derived growth factor, which stimulates the migration and proliferation of smooth muscle cells
- Transforming growth factor–β, which also stimulates proliferation of smooth muscle cells.
Inflammation
Activated platelets release inflammatory mediators and thereby change the adhesive and chemotactic properties of endothelial cells. Likewise, mediators derived from inflammatory cells (neutrophils) can affect platelet function.
Platelet-derived mediators include the following:
- Pro‑interleukin (IL)-β, which triggers the synthesis of E-selectin that enables endothelial cells to interact with leukocytes
- Thromboxane A2, which increases neutrophil adhesion to facilitate platelet aggregation
- Platelet-derived growth factor and platelet factor 4, which increase neutrophil chemotaxis (the ability of neutrophils to infiltrate atherosclerotic plaque)
- CD40 ligand, a protein expressed on platelets that induces inflammatory responses in the endothelium
- P-selectin, a cell adhesion molecule expressed on activated platelets that enhances the adhesion of monocytes on activated endothelial cells.
Among the neutrophil-derived mediators, some—such as superoxide and leukotrienes—enhance platelet activation, whereas elastases inhibit platelet activation.
Overall, once inflammation begins in an atherosclerotic plaque, much reciprocal platelet activation can occur, so that the inflammatory process can become a feed-forward loop to eventually promote atherothrombosis.
Atherothrombosis
In the last stage of the atherosclerotic process, platelet enzymes that degrade the matrix may make plaques vulnerable to rupture by creating fissures in the fibrous plaque cap. This exposes the lipid-rich core, which contains a significant amount of thromboplastin. Exposure to the extracellular matrix can lead to further platelet adhesion, activation, and aggregation. The development of a platelet thrombus is usually one of the ultimate steps in atherothrombosis leading to ACS, including MI.
ROLE OF PLATELETS IN ACUTE CORONARY SYNDROMES: WHAT IS THE EVIDENCE?
How predictive is an elevated platelet count?
However, another study conducted in a slightly different population—1,616 patients with non‑ST-segment-elevation MI/unstable angina—found no correlation between platelet count (by quintiles) and death at 60 months.3 The lowest mortality was observed in patients with a platelet count in the second-lowest quintile, although the highest mortality was indeed observed in the quintile of patients with the lowest platelet counts.3
The differing results in the above two studies suggest that additional platelet factors, beyond platelet count, contribute to the risk of adverse outcomes following ACS.
Platelet hyperreactivity and outcomes in ACS
Platelet hyperreactivity—ie, residual platelet activity despite antiplatelet therapy—appears to be involved in the spectrum of ACS. A recent study evaluated the association between hyperreactivity of platelets to ADP and outcomes in 600 patients with stable cardiovascular disease who were on aspirin therapy.4 Hyperreactivity was defined as a collagen/ADP closure time of less than 90 seconds on the PFA-100 system (short collagen/ADP closure time). On receiver operating characteristic (ROC) curve analysis, a short collagen/ADP closure time served as a significant predictor of recurrent events (relative risk [RR] = 3.65; 95% CI, 1.76–7.57) and death (RR = 6.56; 95% CI, 1.93–22.35) compared with a closure time of 90 seconds or greater. The authors concluded that there appears to be a subgroup of patients with stable cardiovascular disease who have an increased risk of major adverse events associated with platelet hyperreactivity.4
An earlier study by Harrison et al assessed platelet function using the PFA-100 in 78 patients presenting with acute chest pain classified as MI, unstable angina, or nonspecific chest pain.5 Using the PFA-100, they found shorter collagen/ADP closure times and higher levels of von Willebrand factor in subjects with MI compared with those who had unstable angina or nonspecific chest pain.5 Fuchs et al reported a similar association between von Willebrand factor and outcomes in 208 patients with ACS,6 raising the possibility that von Willebrand factor, through its association with increased platelet adhesion and activation, may be a major contributor to risk in ACS.
Similarly, an association between platelet hyperreactivity and cardiovascular events has been suggested in patients with type 2 diabetes. In a 2007 study of 173 patients with type 2 diabetes and coronary artery disease receiving dual antiplatelet therapy (aspirin plus clopidogrel), the 2-year risk of major cardiovascular events was significantly higher in those in the highest quartile of platelet aggregation compared with those in the lower three quartiles (hazard ratio = 3.35; 95% CI, 1.68–6.66).7 In a separate study, Serebruany et al measured platelet activity by five different testing methods in 822 patients with coronary artery disease and found significantly higher platelet hyperreactivity by all methods in those patients who had diabetes (n = 257) than in those who did not (n = 565).8
Marcucci et al recently examined the relationship between clinical characteristics and residual platelet activity in 386 patients with ACS on dual antiplatelet therapy (aspirin plus clopidogrel).9 The presence of residual platelet activity (determined by platelet aggregation in response to the agonists arachidonic acid and ADP, as well as by the PFA-100) was associated with significantly higher inflammatory status, as determined by leukocyte count and erythrocyte sedimentation rate. The same association was observed among a subset of patients in this study undergoing percutaneous coronary intervention (PCI) who were receiving dual antiplatelet therapy; additionally, residual platelet activity was associated with a significantly higher incidence of diabetes and a significantly lower ejection fraction in this subset.9
Platelet hyperreactivity while on dual antiplatelet therapy (aspirin plus clopidogrel) was also found to be predictive of clinical outcome in a study of 195 patients with non-ST-elevation MI undergoing PCI.10 Hyporesponse to antiplatelet therapy, as measured by a high VASP platelet reactivity index (PRI), predicted an increased risk of recurrent ischemic events within 30 days of PCI. Using ROC curve analysis, the investigators found that a VASP PRI cutoff value of 53% (ie, a high PRI [> 53%] indicates residual platelet activity despite clopidogrel) had a sensitivity of 93%, a specificity of 50%, a positive predictive value of 12%, and a negative predictive value of 99% for ischemic events.10 Similarly, among 144 patients undergoing PCI assessed for decreased platelet reactivity to a loading dose of clopidogrel, Bonello et al also found that a VASP PRI greater than 50% was optimal for predicting major adverse cardiovascular events: all 21 events in the study occurred among patients whose VASP PRI was in the highest four quintiles.11
CONCLUSIONS AND GENERAL ASSESSMENT OF PLATELET FUNCTION TESTS
Platelets clearly are involved in the pathogenesis of atherothrombosis. Accumulating evidence suggests that both an elevated platelet count and platelet hyperreactivity (residual platelet activity despite dual antiplatelet therapy) may be associated with adverse cardiovascular events in patients with ACS.
Platelet function can be measured using several different assays and measures of platelet activation. The best assays for measuring residual platelet activity in the setting of antiplatelet therapy are still being defined, as are their predictive values. Platelet aggregation remains the gold standard. The PFA-100 may detect overall platelet hyperreactivity despite the use of antiplatelet therapy, and is attracting increasing use for this purpose. VASP phosphorylation may be a good assay for detecting P2Y12 inhibition but is limited to thienopyridines in terms of detecting platelet hyperreactivity. For predicting adverse cardiac events, ROC curve analysis should be used to objectively define cutoff values for platelet hyperreactivity as opposed to reliance on arbitrarily defined cutoff values.
Moving forward, standard testing protocols for platelet aggregation clearly are needed to achieve consistency among studies.
- Davì G, Patrono C. Platelet activation and atherothrombosis. N Engl J Med 2007; 357:2482–2494.
- Ly HQ, Kirtane AJ, Murphy SA, et al. Association of platelet counts on presentation and clinical outcomes in ST-elevation myocardial infarction (from the TIMI Trials). Am J Cardiol 2006; 98:1–5.
- Mueller C, Neumann FJ, Hochholzer W, et al. The impact of platelet count on mortality in unstable angina/non-ST-segment elevation myocardial infarction. Am Heart J 2006; 151:1214.e1–7.
- Christie DJ, Kottke-Marchant K, Gorman RT. Hypersensitivity of platelets to adenosine diphosphate in patients with stable cardiovascular disease predicts major adverse events despite antiplatelet therapy. Platelets 2008; 19:104–110.
- Harrison P, Mackie I, Mathur A, et al. Platelet hyperfunction in acute coronary syndromes. Blood Coagul Fibrinolysis 2005; 16:557–562.
- Fuchs I, Frossard M, Spiel A, Riedmüller E, Laggner AN, Jilma B. Platelet function in patients with acute coronary syndrome (ACS) predicts recurrent ACS. J Thromb Haemost 2006; 4:2547–2552.
- Angiolillo DJ, Bernardo E, Sabaté M, et al. Impact of platelet reactivity on cardiovascular outcomes in patients with type 2 diabetes mellitus and coronary artery disease. J Am Coll Cardiol 2007; 50:1541–1547.
- Serebruany V, Pokov I, Kuliczkowski W, Chesebro J, Badimon J. Baseline platelet activity and response after clopidogrel in 257 diabetics among 822 patients with coronary artery disease. Thromb Haemost 2008; 100:76–82.
- Marcucci R, Gori AM, Paniccia R, et al. Residual platelet reactivity is associated with clinical and laboratory characteristics in patients with ischemic heart disease undergoing PCI on dual antiplatelet therapy. Atherosclerosis 2007; 195:e217–e223.
- Frere C, Cuisset T, Quilici J, et al. ADP-induced platelet aggregation and platelet reactivity index VASP are good predictive markers for clinical outcomes in non-ST elevation acute coronary syndrome. Thromb Haemost 2007; 98:838–843.
- Bonello L, Paganelli F, Arpin-Bornet M, et al. Vasodilator-stimulated phosphoprotein phosphorylation analysis prior to percutaneous coronary intervention for exclusion of postprocedural major adverse cardiovascular events. J Thromb Haemost 2007; 5:1630–1636.
- Davì G, Patrono C. Platelet activation and atherothrombosis. N Engl J Med 2007; 357:2482–2494.
- Ly HQ, Kirtane AJ, Murphy SA, et al. Association of platelet counts on presentation and clinical outcomes in ST-elevation myocardial infarction (from the TIMI Trials). Am J Cardiol 2006; 98:1–5.
- Mueller C, Neumann FJ, Hochholzer W, et al. The impact of platelet count on mortality in unstable angina/non-ST-segment elevation myocardial infarction. Am Heart J 2006; 151:1214.e1–7.
- Christie DJ, Kottke-Marchant K, Gorman RT. Hypersensitivity of platelets to adenosine diphosphate in patients with stable cardiovascular disease predicts major adverse events despite antiplatelet therapy. Platelets 2008; 19:104–110.
- Harrison P, Mackie I, Mathur A, et al. Platelet hyperfunction in acute coronary syndromes. Blood Coagul Fibrinolysis 2005; 16:557–562.
- Fuchs I, Frossard M, Spiel A, Riedmüller E, Laggner AN, Jilma B. Platelet function in patients with acute coronary syndrome (ACS) predicts recurrent ACS. J Thromb Haemost 2006; 4:2547–2552.
- Angiolillo DJ, Bernardo E, Sabaté M, et al. Impact of platelet reactivity on cardiovascular outcomes in patients with type 2 diabetes mellitus and coronary artery disease. J Am Coll Cardiol 2007; 50:1541–1547.
- Serebruany V, Pokov I, Kuliczkowski W, Chesebro J, Badimon J. Baseline platelet activity and response after clopidogrel in 257 diabetics among 822 patients with coronary artery disease. Thromb Haemost 2008; 100:76–82.
- Marcucci R, Gori AM, Paniccia R, et al. Residual platelet reactivity is associated with clinical and laboratory characteristics in patients with ischemic heart disease undergoing PCI on dual antiplatelet therapy. Atherosclerosis 2007; 195:e217–e223.
- Frere C, Cuisset T, Quilici J, et al. ADP-induced platelet aggregation and platelet reactivity index VASP are good predictive markers for clinical outcomes in non-ST elevation acute coronary syndrome. Thromb Haemost 2007; 98:838–843.
- Bonello L, Paganelli F, Arpin-Bornet M, et al. Vasodilator-stimulated phosphoprotein phosphorylation analysis prior to percutaneous coronary intervention for exclusion of postprocedural major adverse cardiovascular events. J Thromb Haemost 2007; 5:1630–1636.
KEY POINTS
- Platelet function assays are inherently variable because they measure cell function rather than a single analyte.
- Screening tests, or global tests for platelet function, do not identify specific causes of platelet dysfunction but combine measurement of different aspects of platelet function.
- There appears to be a subgroup of patients with stable cardiovascular disease who have an increased risk of major cardiac events associated with platelet hyperreactivity.
- For predicting cardiac events, receiver operating characteristic (ROC) curve analysis should be used to objectively define cutoff values for platelet hyperreactivity as opposed to reliance on arbitrary cutoff values.
Novel antiplatelet strategies in acute coronary syndromes
An enhanced understanding of platelet biology, as reviewed in the previous article in this supplement, has made it possible to identify a wide variety of platelet agonists. This knowledge has fostered the development of a host of pharmacologic strategies to block agonists such as cyclooxygenase, thromboxane, adenosine diphosphate (ADP), and thrombin, among others. This article will discuss the pharmacologic properties of novel antiplatelet agents, as well as alternative dosing of the established antiplatelet agent clopidogrel, and will review data from available comparative and placebo-controlled trials of these agents. The article concludes with comparative perspectives on the potential roles and relative advantages of these agents in the evolving management of patients with acute coronary syndromes (ACS).
CLOPIDOGREL AND THE CHALLENGE OF VARIABLE RESPONSE
Clopidogrel, a member of the thienopyridine class of ADP receptor inhibitors, is well established for use in patients with ACS at a loading dose of 300 mg followed by a maintenance dose of 75 mg/day. At this loading dose, inhibition of platelet aggregation to ADP is approximately 30%, and the time to peak effect is approximately 4 to 6 hours.1
As with most other drugs, the response to clopidogrel is variable. However, in contrast to the accepted measures of response to antihypertensive or lipid-lowering drugs, there are no routinely used tests for measuring response to antiplatelet therapies. As a result, a “one size fits all” strategy in the dosing of clopidogrel has prevailed.
This variability in response is clinically relevant. In a study assessing clopidogrel responsiveness by ADP-induced platelet aggregation in 60 patients who experienced ST-segment-elevation myocardial infarction (MI), Matetzky et al found that the lowest levels of clopidogrel responsiveness were associated with a significantly elevated rate (P = .007) of recurrent cardiovascular events 6 months after the MI.3 Gurbel et al found a similar association between clopidogrel responsiveness and subacute stent thrombosis in a study of 120 patients using two different methods—light transmission aggregotomy to 5 μmol/L of ADP, and the ratio of vasodilator-stimulated phosphoprotein reactivity—to assess clopidogrel responsiveness.4
Increasing the loading dose raises response rates
One proposed method for boosting responsiveness to clopidogrel in suboptimal responders is the use of a higher dose. In a study of 190 patients undergoing coronary stenting, increasing the loading dose from 300 mg to 600 mg reduced the rate of clopidogrel resistance (defined as a < 10% absolute change in aggregation to 5 μM of ADP at 24 hours) from 28% to 8% (P < .001),5 a finding that supports the notion of enhanced response at doses up to 600 mg. Single loading doses in excess of 600 mg yield diminishing returns in terms of platelet inhibition, most likely as a result of clopidogrel pharmacokinetics.6
Compared with 300 mg of clopidogrel, the more potent platelet inhibitory effect of a 600-mg dose translated to a two-thirds reduction (P = .041) in the composite end point of death, MI, or target vessel revascularization at 30 days in a study of 255 patients with stable coronary artery disease undergoing percutaneous coronary intervention (PCI).7 The reduction in this composite end point with high-dose clopidogrel was driven by a reduction in the incidence of periprocedural MI.
In a separate study of 292 patients with non‑ST-segment-elevation ACS who were scheduled for PCI, the superior platelet response to 600 mg versus 300 mg of clopidogrel translated to a 60% reduction in adverse thrombotic events (P = .02), and this benefit extended beyond rates of periprocedural MI.8
Similar results with increased maintenance dose
Similarly, emerging data suggest that raising the maintenance dose of clopidogrel can also raise response rates. In a study of 60 patients, doubling the maintenance dose of clopidogrel after PCI from 75 mg/day to 150 mg/day resulted in improved platelet inhibition as assessed by rapid platelet function analysis.9 Likewise, a 150-mg/day maintenance dose of clopidogrel was associated with a superior antiplatelet effect compared with 75 mg/day in a study of 40 patients with type 2 diabetes.10
Large definitive trial is under way
In the wake of these smaller trials, a large randomized trial known as CURRENT is comparing a strategy of high-dose clopidogrel with standard-dose clopidogrel in patients with ACS for whom an early invasive management strategy is planned.11 The high-dose regimen involves a 600-mg loading dose followed by 150 mg/day for 1 week and then 75 mg/day for 3 weeks, whereas the standard-dose regimen involves a 300-mg loading dose followed by 75 mg/day for 4 weeks. Both groups are being further randomized to low-dose aspirin (75 to 100 mg/day) or high-dose aspirin (300 to 325 mg/day) for 30 days after PCI. With a target enrollment well beyond 10,000 patients, CURRENT should definitively clarify the relative efficacy and safety of high-dose clopidogrel in this setting.
Tailoring clopidogrel therapy
Investigators have explored tailoring the dosing of clopidogrel around the time of PCI based on the degree of platelet inhibition. In one study, administering additional loading doses of clopidogrel, up to a total of 2,400 mg, before PCI in patients with a suboptimal degree of platelet inhibition resulted in a lower rate of ischemic complications following PCI.12
PRASUGREL, A NOVEL THIENOPYRIDINE
Prasugrel is an investigational third-generation thienopyridine currently under US Food and Drug Administration (FDA) review for use in patients with ACS being managed with PCI. Like clopidogrel, prasugrel is a prodrug that requires conversion to an active metabolite prior to binding to the platelet P2Y12 receptor for ADP to confer antiplatelet activity. Prasugrel is metabolized more efficiently than clopidogrel, allowing for faster activation and superior bioavailability to produce a greater and more consistent antiplatelet effect.1,13
The active metabolites of clopidogrel and prasugrel are no different in their ability to inhibit platelet aggregation, but approximately 85% of clopidogrel is inactivated by esterases, with the remaining 15% being converted to the active metabolite using the cytochrome P450 pathway via two successive oxidative steps in the liver.14 In contrast, esterases facilitate the transformation of prasugrel to its active metabolite.14 This activation requires only one oxidative step that can occur in either the liver or the gut through cytochrome P450.
Both prasugrel and clopidogrel are irreversible P2Y12 receptor blockers. For this reason, one must wait approximately 5 days after the last dose of either medication for generation of a sufficient number of new platelets to allow restoration of normal platelet-mediated hemostasis.
Inhibition of platelet aggregation relative to clopidogrel
In a study among healthy volunteers, inhibition of platelet aggregation was significantly higher after a 60-mg loading dose of prasugrel compared with a 300-mg loading dose of clopidogrel.13 Further, suboptimal responders to clopidogrel who crossed over to prasugrel had levels of platelet inhibition as high as 80% following prasugrel administration. The time to peak effect of prasugrel was about 1 hour. Inhibition of platelet aggregation was more consistent following dosing of prasugrel compared with clopidogrel.13
In a study of 201 patients undergoing cardiac catheterization with planned PCI, Wiviott et al demonstrated better levels of inhibition of platelet aggregation at 6 hours after a 60-mg loading dose of prasugrel than after a 600-mg loading dose of clopidogrel (P < .0001).1
Clinical effects relative to clopidogrel: TRITON-TIMI 38
A large phase 3 clinical trial—the Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel–Thrombolysis in Myocardial Infarction (TRITON-TIMI) 38—was conducted to compare the effects of prasugrel and standard-dose clopidogrel on death and ischemic end points in 13,608 patients with ACS scheduled to undergo PCI.15 Patients randomized to clopidogrel were given the standard regimen of a 300-mg loading dose followed by a 75-mg daily maintenance dose; those randomized to prasugrel were given a 60-mg loading dose followed by a 10-mg daily maintenance dose. The study drug was typically given immediately before PCI, a time frame that may mimic real-life use but that favored the faster-onset prasugrel over the slower-onset clopidogrel. Both groups also received low-dose aspirin. Approximately half of the patients in each group were treated with a glycoprotein IIb/IIIa inhibitor. The median duration of therapy was approximately 15 months.
Efficacy. The primary end point—a composite of cardiovascular death, MI, or stroke—occurred in 9.9% of patients randomized to prasugrel compared with 12.1% of those randomized to clopidogrel, corresponding to a 19% relative risk reduction (P = .0004) with prasugrel. Based on these results, 46 patients would need to be treated with prasugrel rather than with clopidogrel to prevent 1 additional cardiovascular death, MI, or stroke.15
The reduction in the primary end point with prasugrel was driven primarily by a reduction in nonfatal MI; nonsignificant trends favored prasugrel over clopidogrel on rates of cardiovascular death and all-cause mortality, but there was no difference in stroke rates. Prasugrel’s effect was consistent across subgroups based on MI type, sex, age, the type of stent used, adjunctive antithrombotic therapy, and renal function.15
In the subgroup of patients with diabetes, the relative reduction in the primary end point with prasugrel compared with clopidogrel was 30% (P < .001), and the respective relative reduction among patients with diabetes who required insulin was 37%.16
Safety. Higher antiplatelet potency carries the trade-off of increased bleeding, and this trade-off was apparent with prasugrel in TRITON-TIMI 38.15 TIMI major bleeding (not counting bleeding related to coronary artery bypass graft surgery [CABG]) occurred significantly more often in prasugrel-treated subjects than in those receiving clopidogrel (2.4% vs 1.8%; P = .03), as did life-threatening bleeds (1.4% vs 0.9%; P = .01). Because absolute rates of major bleeding were low in each treatment group, based on these results, 167 patients would need to be treated with prasugrel rather than clopidogrel to result in 1 excess non-CABG-related major bleeding episode. Rates of intracranial hemorrhage were identical in the two treatment groups.15
Net clinical outcome and therapeutic considerations. Overall analysis of the balance of efficacy and safety in TRITON-TIMI 38 revealed that 138 events were prevented with randomization to prasugrel instead of clopidogrel, at a cost of 35 additional TIMI major bleeds (Figure 2).15
In a post hoc analysis of net clinical outcome, in which major bleeding events were added to the primary composite efficacy end point, prasugrel was associated with a 13% relative risk reduction (P = .004).15 Twenty-three MIs were prevented per 1,000 treated patients with the use of prasugrel instead of clopidogrel, at a cost of 6 excess non-CABG-related major bleeds.15
Another post hoc assessment identified three subgroups who had a significantly increased risk of TIMI major bleeds with randomization to prasugrel15:
- Patients aged 75 years or older
- Patients with a body weight less than 60 kg
- Patients with a history of stroke or transient ischemic attack (TIA).
In these three subgroups, the net clinical effect either was neutral (for those aged ≥ 75years and for those weighing < 60 kg) or favored clopidogrel (for those with a history of stroke or TIA). The group with a history of stroke or TIA represented 4% of the entire cohort, and the TRITON-TIMI 38 investigators recommended avoiding prasugrel in patients with a history of these events. The other two subgroups with a significantly increased bleeding risk with prasugrel represented 16% of the entire cohort, and in these two groups the investigators suggested a pharmacokinetics-guided reduction in the maintenance dose of prasugrel, although a recommendation for such dosing is based on modeling and not actual outcomes data.15
Stent thrombosis. A subanalysis of TRITON-TIMI 38 examined the risk of stent thrombosis in the 12,844 patients enrolled in the trial who had stents implanted.17 Stent thrombosis was assessed using the Academic Research Consortium definitions of definite, probable, and possible stent thrombosis.18 The risk of definite or probable stent thrombosis was halved (hazard ratio = 0.48; P < .0001) with the use of prasugrel compared with clopidogrel, and the reduction was highly significant regardless of the type of stent implanted or the way stent thrombosis was defined. Significant reductions in both early (within the first 30 days) stent thrombosis (P < .0001) and late (beyond 30 days) stent thrombosis (P = .03) were observed in the prasugrel arm compared with the clopidogrel arm.17
AZD6140, A REVERSIBLE P2Y12 RECEPTOR ANTAGONIST
AZD6140, another investigational antiplatelet agent, is an orally active reversible P2Y12 receptor antagonist, in contrast to the thienopyridines, which are irreversible inhibitors. A member of the cyclo-pentyl-triazolo-pyrimidine (CPTP) class, AZD6140 has a rapid onset of action (≤ 2 hours) and does not require metabolic activation. Its plasma half-life is approximately 12 hours, which translates to twice-daily dosing.
Inhibition of platelet aggregation relative to clopidogrel
In a study of clopidogrel-naïve patients with ACS, inhibition of platelet aggregation 12 hours after administration of AZD6140 was approximately 75% with 90-mg, 180-mg, and 270-mg doses, significantly greater than the 30% inhibition achieved after administration of 300 mg of clopidogrel (P < .0002 for all doses of AZD6140 vs clopidogrel).19 Whereas steady state was achieved in approximately 4 to 6 hours with clopidogrel, it was achieved in approximately 2 hours or less with AZD6140.
Clinical safety and efficacy relative to clopidogrel
In a dose-ranging study of AZD6140, adjudicated bleeding rates were similar among two different doses of AZD6140 (90 mg twice daily and 180 mg twice daily) and clopidogrel 75 mg once daily, with no evidence of a dose effect for major bleeding with AZD6140.20 Although this study, conducted in 990 patients with ACS, was underpowered for efficacy end points, rates of adjudicated MI were numerically lower in each of the AZD6140 groups than in the clopidogrel group.
A more definitive evaluation of the relative effcicacy and safety of AZD6140 is expected from the ongoing PLATO trial, which is comparing 90 mg of AZD6140 twice daily with clopidogrel 75 mg/day among 18,000 patients randomized to one of the two treatments within 24 hours of an index ACS event.21
CANGRELOR, A RAPID PARENTERAL P2Y12 RECEPTOR ANTAGONIST
Cangrelor (formerly known as AR-C69931MX) is an intravenously (IV) administered P2Y12 receptor antagonist under investigation for treatment of ACS and use during PCI and other coronary procedures. The compound is an adenosine triphosphate analogue with a plasma half-life of 5 to 9 minutes. Cangrelor is highly reversible, as platelet function returns to normal within 20 minutes of dosing. Within 15 minutes of initiation, cangrelor produces profound platelet inhibition and rapidly achieves steady state; peak effect occurs within minutes.22 The response to cangrelor is highly consistent, with virtually all recipients achieving the same degree of platelet inhibition. Platelet response approaches baseline 15 minutes after termination.22
If approved by the FDA, cangrelor would be administered similar to the way that glycoprotein IIb/IIIa inhibitors are, as it would be used primarily in the catheterization laboratory and then discontinued after the procedure, at which point transition to a long-term oral therapy would be necessary.
Clinical effects relative to abciximab
Cangrelor has been compared with the glycoprotein IIb/IIIa inhibitor abciximab and placebo in 249 patients undergoing elective or urgent PCI.22 Rates of the combined end point of death, MI, or need for repeat revascularization at 30 days were similar with cangrelor and abciximab (5.7% vs 5.4%, respectively; P = NS), both of which were lower than the rate with placebo (10.0%). Major or minor bleeding through 7 days occurred in numerically fewer cangrelor recipients compared with abciximab recipients (7.0% vs 9.0%), although the small sample size precluded evaluation for statistical significance.
Clinical effects relative to clopidogrel—the CHAMPION trials
A phase 3 trial program consisting of two multinational studies of cangrelor—the Cangrelor Versus Standard Therapy to Achieve Optimal Management of Platelet Inhibition (CHAMPION) program—is currently under way.
CHAMPION-PCI is enrolling 9,000 patients presenting with ACS who are being randomized in a double-blind fashion at the start of PCI to a 600-mg loading dose of clopidogrel or to cangrelor given as an IV bolus of 30 μg/kg followed by an IV infusion of 4 μg/kg/min. The primary end point is a composite of all-cause mortality, MI, or ischemia-driven revascularization in the 48 hours following randomization. Secondary end points include rates of all-cause mortality and MI at 48 hours.23
CHAMPION-PLATFORM is enrolling 4,400 patients scheduled for PCI as a result of ACS who are being randomized in a double-blind, double-dummy manner to (1) cangrelor bolus and infusion plus oral placebo or (2) oral clopidogrel plus placebo bolus and infusion before their index procedures. Dosages of the two agents are the same as in CHAMPION-PCI. The primary end point is a composite of death, MI, or urgent target vessel revascularization at 48 hours. Secondary end points include 30-day and 1-year clinical outcomes.23
The rationale for the CHAMPION investigations stems from the need to initiate clopidogrel before a patient is taken to the catheterization laboratory, owing to the inability to achieve a high degree of platelet inhibition until 4 to 6 hours after clopidogrel administration. Although this strategy can be undertaken without complication for most patients, a subset of patients with three-vessel disease or left-main disease will require CABG, which then must be delayed several days until clopidogrel’s platelet-inhibiting effect diminishes. A rapid-acting IV inhibitor of the P2Y12 receptor such as cangrelor would obviate this concern.
THROMBIN INHIBITORS
Thrombin plays an important role in platelet activation, and thrombin receptor antagonists may represent a safer means of inhibiting platelet activation relative to traditional antiplatelet agents. This theoretical safety advantage stems from the notion that blocking the action of platelets at the thrombin receptor would preserve platelets’ function as mediators of primary hemostasis. Because thrombin’s activation of platelets should occur only during clot formation, blocking platelet activation at the thrombin receptor would interrupt thrombin’s ability to propagate platelet activation during formation of coronary artery clots.
One agent in this class that is being studied extensively is SCH 530348, an oral thrombin receptor antagonist with potent antiplatelet activity. Its peak antiplatelet potency is achieved within hours when a loading dose is given, and within days without a loading dose. Wearing-off of the action of SCH 530348 takes weeks.24
Inhibition of platelet aggregation with thrombin receptor antagonists is measured in response to the thrombin receptor antagonist peptide (TRAP), not ADP. The proportion of subjects treated with SCH 530348 who achieve greater than 80% inhibition of platelet aggregation to 15 μM of TRAP ranges from 91% (with 0.5 mg of SCH 530348) to 100% (with 1.0 mg and 2.5 mg) at both 30 days and 60 days.25
Clinical effects in placebo-controlled trials
SCH 530348 was studied in the Thrombin Receptor Antagonist (TRA)–PCI trial, a dose-ranging study in which patients were randomized to one of three oral loading doses of the study drug (10 mg, 20 mg, or 40 mg) on top of a clopidogrel loading dose before undergoing cardiac catheterization for planned PCI; patients were then randomized to one of three maintenance doses of SCH 530348 (0.5 mg, 1.0 mg, or 2.5 mg) or placebo (depending on loading therapy) for 60 days.25
Among the 573 patients undergoing PCI , the rate of TIMI major or minor bleeding was not significantly higher with any dose of SCH 530348 compared with placebo,25 supporting the hypothesis that thrombin receptor antagonism inhibits platelet aggregation without a significant increase in bleeding.
Although the TRA-PCI study was not powered to detect differences in clinical event rates, a reduction in the rate of major adverse cardiovascular events was observed in a dose-dependent manner with SCH 530348 compared with placebo in the PCI cohort.25
On the basis of the TRA-PCI trial, a pair of phase 3 trials of SCH 530348 have been launched—the Thrombin Receptor Antagonist in Secondary Prevention of Atherothrombotic Ischemic Events (TRA 2°P-TIMI 50) study and the Thrombin Receptor Antagonist for Clinical Event Reduction in ACS (TRA-CER) study.
TRA 2°P-TIMI 50 is a multinational double-blind study enrolling 19,500 patients with prior MI or stroke or with existing peripheral arterial disease. Patients are being randomized to placebo plus standard medical care (including aspirin and clopidogrel) or to 2.5 mg of SCH 530348 once daily plus standard medical care. The primary end point is the composite of cardiovascular death, MI, urgent coronary revascularization, or stroke.26
TRA-CER is a multinational double-blind study with planned enrollment of 10,000 patients with non-ST-segment-elevation MI. Patients are being randomized to placebo plus standard medical care (including aspirin or clopidogrel) or to SCH 530348 (using the oral 40-mg loading dose and a maintenance dose of 2.5 mg once daily) plus standard medical care. The primary end point is the composite of cardiovascular death, MI, rehospitalization for ACS, urgent coronary revascularization, or stroke. The key secondary end point is the composite of cardiovascular death, MI, or stroke.27
COMPARATIVE CONSIDERATIONS
Inhibition of platelet aggregation
Clopidogrel achieves about 30% inhibition of platelet aggregation to ADP at its current FDA-approved loading dose of 300 mg and about 40% inhibition when its dose is doubled to 600 mg. These levels of inhibition are increased to 75% to 80% by clopidogrel’s fellow thienopyridine prasugrel, and this increase is attributable to prasugrel’s more efficient metabolism from prodrug to active metabolite. The reversible P2Y12 receptor antagonist AZD6140 achieves a comparable 75% to 80% inhibition of platelet aggregation. The parenterally administered P2Y12 receptor antagonist cangrelor achieves greater than 90% inhibition, as does the oral thrombin receptor antagonist SCH 530348, although the latter agent’s inhibition is to the agonist TRAP rather than ADP.
Time to peak effect
The time to peak effect with clopidogrel is approximately 4 hours regardless of the loading dose used (300 mg or 600 mg); this is substantially reduced with all of the investigational agents except SCH 530348. The novel agents’ reduced time to peak effect can offer advantages in speeding patients’ readiness to undergo catheterization procedures. This is particularly true for the IV agent cangrelor, which achieves its peak effect within minutes, although the 1-hour to 2-hour time frame with oral agents prasugrel and AZD6140 also would usually obviate any need to delay catheterization.
Consistency of platelet response
Standard-dose clopidogrel has the least consistency of platelet response among the therapies reviewed. Although increasing the clopidogrel dose yields somewhat greater consistency in response, it is still lower than the very high degrees of consistency observed with all of the novel compounds, each of which appears to achieve the same degree of inhibition of aggregation in virtually all patients.
Offset of effect
Both of the thienopyridines—clopidogrel and prasugrel—have an offset of effect of about 5 days, which requires delay of surgery, if possible, for several days in patients taking these agents. This is not an issue for the reversible oral agent AZD6140, whose offset of action takes just 1 to 2 days. While this rapid wearing-off of effect translates to a potential advantage for AZD6140, it also poses the potential drawback that a missed dose or two may leave the patient exposed to the risk of a thrombotic event. Cangrelor’s rapid offset of 20 minutes promotes its envisioned use as a catheterization lab–based medication like the glycoprotein IIb/IIIa inhibitors that can be started right before a PCI procedure and stopped immediately afterward. Because SCH 530348 has a very long half-life and thus a weeks-long washout period, the practicality of its use may depend on the hypothesis that thrombin receptor antagonists do not interfere with primary hemostasis, which is supported by data to date but remains to be definitively confirmed.
CONCLUSIONS
Clopidogrel achieves modest platelet inhibition with wide variability in response. Higher doses of clopidogrel achieve modestly greater degrees of inhibition than standard doses, and appear to result in a decreased rate of ischemic events. Although higher doses of clopidogrel have been embraced by some clinicians, we await definitive phase 3 trial evidence of net benefit before making high-dose clopidogrel the new standard of care.
Compared with clopidogrel, the investigational thienopyridine prasugrel is a more potent and consistent blocker of the ADP receptor. It results in a decreased rate of ischemic events relative to clopidogrel, including a 50% reduction in the rate of stent thrombosis, but is associated with an increased rate of bleeding. If prasugrel is approved for marketing, its use should be avoided in patients with a history of stroke or TIA, and avoidance or dose adjustment may be necessary in patients aged 75 years or older and in patients weighing less than 60 kg.
Other novel antiplatelet agents being evaluated for use in patients with ACS—the reversible oral ADP receptor blocker AZD6140, the rapid-acting IV ADP receptor blocker cangrelor, and oral thrombin receptor antagonists—offer potential advantages that need to be examined in the context of large-scale clinical trials.
- Wiviott SD, Trenk D, Frelinger AL, et al. Prasugrel compared with high loading- and maintenance-dose clopidogrel in patients with planned percutaneous coronary intervention: the Prasugrel in Comparison to Clopidogrel for Inhibition of Platelet Activation and Aggregation–Thrombolysis in Myocardial Infarction 44 trial. Circulation 2007; 116:2923–2932.
- Serebruany VL, Steinhubl SR, Berger PB, Malinin AI, Bhatt DL, Topol EJ. Variability in platelet responsiveness to clopidogrel among 544 individuals. J Am Coll Cardiol 2005; 45:246–251.
- Matetzky S, Shenkman B, Guetta V, et al. Clopidogrel resistance is associated with increased risk of recurrent atherothrombotic events in patients with acute myocardial infarction. Circulation 2004; 109:3171–3175.
- Gurbel PA, Bliden KP, Samara W, et al. Clopidogrel effect on platelet reactivity in patients with stent thrombosis: results of the CREST Study. J Am Coll Cardiol 2005; 46:1827–1832.
- Gurbel PA, Bliden KP, Hayes KM, Yoho JA, Herzog WR, Tantry US. The relation of dosing to clopidogrel responsiveness and the incidence of high post-treatment platelet aggregation in patients undergoing coronary stenting. J Am Coll Cardiol 2005; 45:1392–1396.
- von Beckerath N, Taubert D, Pogatsa-Murray G, Schömig E, Kastrati A, Schömig A. Absorption, metabolization, and antiplatelet effects of 300-, 600-, and 900-mg loading doses of clopidogrel: results of the ISAR-CHOICE (Intracoronary Stenting and Antithrombotic Regimen: Choose Between 3 High Oral Doses for Immediate Clopidogrel Effect) Trial. Circulation 2005; 112:2946–2950.
- Patti G, Colonna G, Pasceri V, Pepe LL, Montinaro A, Di Sciascio G. Randomized trial of high loading dose of clopidogrel for reduction of periprocedural myocardial infarction in patients undergoing coronary intervention: results from the ARMYDA-2 (Antiplatelet therapy for Reduction of MYocardial Damage during Angioplasty) study. Circulation 2005; 111:2099–2106.
- Cuisset T, Frere C, Quilici J, et al. Benefit of a 600-mg loading dose of clopidogrel on platelet reactivity and clinical outcomes in patients with non-ST-segment elevation acute coronary syndrome undergoing coronary stenting. J Am Coll Cardiol 2006; 48:1339–1345.
- von Beckerath N, Kastrati A, Wieczorek A, et al. A double-blind, randomized study on platelet aggregation in patients treated with a daily dose of 150 or 75 mg of clopidogrel for 30 days. Eur Heart J 2007; 28:1814–1819.
- Angiolillo DJ, Shoemaker SB, Desai B, et al. Randomized comparison of a high clopidogrel maintenance dose in patients with diabetes mellitus and coronary artery disease: results of the Optimizing Antiplatelet Therapy in Diabetes Mellitus (OPTIMUS) study. Circulation 2007; 115:708–716.
- Clopidogrel optimal loading dose usage to reduce recurrent events/optimal antiplatelet strategy for interventions (CURRENT/OASIS7). Clinical Trials.gov Web site. http://clinicaltrials.gov/ct2/show/NCT00335452. Updated September 1, 2008. Accessed December 16, 2008.
- Bonello L, Camoin-Jau L, Arques S, et al. Adjusted clopidogrel loading doses according to vasodilator-stimulated phosphoprotein phosphorylation index decrease rate of major adverse cardiovascular events in patients with clopidogrel resistance: a multicenter randomized prospective study. J Am Coll Cardiol 2008; 51:1404–1411.
- Brandt JT, Payne CD, Wiviott SD, et al. A comparison of prasugrel and clopidogrel loading doses on platelet function: magnitude of platelet inhibition is related to active metabolic formation. Am Heart J 2007; 153:66.e9–e16.
- Herbert JM, Savi P. P2Y12, a new platelet ADP receptor, target of clopidogrel. Semin Vasc Med 2003; 3:113–122.
- Wiviott SD, Braunwald E, McCabe CH, et al. Prasugrel versus clopidogrel in patients with acute coronary syndromes. N Engl J Med 2007; 357:2001–2015.
- Wiviott SD, Braunwald E, Angiolillo DJ, et al. Greater clinical benefit of more intensive oral antiplatelet therapy with prasugrel in patients with diabetes mellitus in the Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel–Thrombolysis in Myocardial Infarction 38. Circulation 2008; 118:1626–1636.
- Wiviott SD, Braunwald E, McCabe CH, et al. Intensive oral antiplatelet therapy for reduction of ischaemic events including stent thrombosis in patients with acute coronary syndromes treated with percutaneous coronary intervention and stenting in the TRITON-TIMI 38 trial: a subanalysis of a randomised trial. Lancet 2008; 371:1353–1363.
- Mauri L, Hsieh WH, Massaro JM, Ho KK, D’Agostino R, Cutlip DE. Stent thrombosis in randomized clinical trials of drug-eluting stents. N Engl J Med 2007; 356:1020–1029.
- Storey RF, Husted S, Harrington RA, et al. Inhibition of platelet aggregation by AZD6140, a reversible oral P2Y12 receptor antagonist, compared with clopidogrel in patients with acute coronary syndromes. J Am Coll Cardiol 2007; 50:1852–1856.
- Cannon CP, Husted S, Harrington RA, et al. Safety, tolerability, and initial efficacy of AZD6140, the first reversible oral adenosine diphosphate receptor antagonist, compared with clopidogrel, in patients with non-ST-segment elevation acute coronary syndrome: primary results of the DISPERSE-2 trial. J Am Coll Cardiol 2007; 50:1844–1851.
- A comparison of AZD6140 and clopidogrel in patients with acute coronary syndrome (PLATO). Clinical Trials.gov Web site. http://clinicaltrials.gov/ct2/show/NCT00391872. Updated December 3, 2008. Accessed December 5, 2008.
- Greenbaum AB, Grines CL, Bittl JA, et al. Initial experience with an intravenous P2Y12 platelet receptor antagonist in patients undergoing percutaneous coronary intervention: results from a 2-part, phase II, multicenter, randomized, placebo- and active-controlled trial. Am Heart J 2006; 151:689.e1–689.e10.
- A clinical trial to demonstrate the efficacy of cangrelor (PCI). Clinical Trials.gov Web site. http://www.clinicaltrials.gov/ct/show/nct00305162. Updated December 3, 2008. Accessed December 5, 2008.
- Bhatt DL, Topol EJ. Scientific and therapeutic advances in antiplatelet therapy. Nat Rev Drug Discov 2003; 2:15–28.
- Moliterno DJ, Becker RC, Jennings LK, et al; TRA-PCI Study Investigators. Results of a multinational randomized, double-blind, placebo-controlled study of a novel thrombin receptor antagonist (SCH 530348) in percutaneous coronary intervention. Presented at: 56th Annual Scientific Session of the American College of Cardiology; March 24–27, 2007; New Orleans, LA.
- Trial to assess the effects of SCH 530348 in preventing heart attack and stroke in patients with atherosclerosis (TRA 2°P-TIMI 50). Clinical Trials.gov Web site. http://clinicaltrials.gov/ct2/show/NCT00526474. Updated November 13, 2008. Accessed December 16, 2008.
- Trial to assess the effects of SCH 530348 in preventing heart attack and stroke in patients with acute coronary syndrome (TRA-CER). Clinical Trials.gov Web site. http://clinicaltrials.gov/ct2/show/NCT00527943. Updated November 12, 2008. Accessed December 16, 2008.
An enhanced understanding of platelet biology, as reviewed in the previous article in this supplement, has made it possible to identify a wide variety of platelet agonists. This knowledge has fostered the development of a host of pharmacologic strategies to block agonists such as cyclooxygenase, thromboxane, adenosine diphosphate (ADP), and thrombin, among others. This article will discuss the pharmacologic properties of novel antiplatelet agents, as well as alternative dosing of the established antiplatelet agent clopidogrel, and will review data from available comparative and placebo-controlled trials of these agents. The article concludes with comparative perspectives on the potential roles and relative advantages of these agents in the evolving management of patients with acute coronary syndromes (ACS).
CLOPIDOGREL AND THE CHALLENGE OF VARIABLE RESPONSE
Clopidogrel, a member of the thienopyridine class of ADP receptor inhibitors, is well established for use in patients with ACS at a loading dose of 300 mg followed by a maintenance dose of 75 mg/day. At this loading dose, inhibition of platelet aggregation to ADP is approximately 30%, and the time to peak effect is approximately 4 to 6 hours.1
As with most other drugs, the response to clopidogrel is variable. However, in contrast to the accepted measures of response to antihypertensive or lipid-lowering drugs, there are no routinely used tests for measuring response to antiplatelet therapies. As a result, a “one size fits all” strategy in the dosing of clopidogrel has prevailed.
This variability in response is clinically relevant. In a study assessing clopidogrel responsiveness by ADP-induced platelet aggregation in 60 patients who experienced ST-segment-elevation myocardial infarction (MI), Matetzky et al found that the lowest levels of clopidogrel responsiveness were associated with a significantly elevated rate (P = .007) of recurrent cardiovascular events 6 months after the MI.3 Gurbel et al found a similar association between clopidogrel responsiveness and subacute stent thrombosis in a study of 120 patients using two different methods—light transmission aggregotomy to 5 μmol/L of ADP, and the ratio of vasodilator-stimulated phosphoprotein reactivity—to assess clopidogrel responsiveness.4
Increasing the loading dose raises response rates
One proposed method for boosting responsiveness to clopidogrel in suboptimal responders is the use of a higher dose. In a study of 190 patients undergoing coronary stenting, increasing the loading dose from 300 mg to 600 mg reduced the rate of clopidogrel resistance (defined as a < 10% absolute change in aggregation to 5 μM of ADP at 24 hours) from 28% to 8% (P < .001),5 a finding that supports the notion of enhanced response at doses up to 600 mg. Single loading doses in excess of 600 mg yield diminishing returns in terms of platelet inhibition, most likely as a result of clopidogrel pharmacokinetics.6
Compared with 300 mg of clopidogrel, the more potent platelet inhibitory effect of a 600-mg dose translated to a two-thirds reduction (P = .041) in the composite end point of death, MI, or target vessel revascularization at 30 days in a study of 255 patients with stable coronary artery disease undergoing percutaneous coronary intervention (PCI).7 The reduction in this composite end point with high-dose clopidogrel was driven by a reduction in the incidence of periprocedural MI.
In a separate study of 292 patients with non‑ST-segment-elevation ACS who were scheduled for PCI, the superior platelet response to 600 mg versus 300 mg of clopidogrel translated to a 60% reduction in adverse thrombotic events (P = .02), and this benefit extended beyond rates of periprocedural MI.8
Similar results with increased maintenance dose
Similarly, emerging data suggest that raising the maintenance dose of clopidogrel can also raise response rates. In a study of 60 patients, doubling the maintenance dose of clopidogrel after PCI from 75 mg/day to 150 mg/day resulted in improved platelet inhibition as assessed by rapid platelet function analysis.9 Likewise, a 150-mg/day maintenance dose of clopidogrel was associated with a superior antiplatelet effect compared with 75 mg/day in a study of 40 patients with type 2 diabetes.10
Large definitive trial is under way
In the wake of these smaller trials, a large randomized trial known as CURRENT is comparing a strategy of high-dose clopidogrel with standard-dose clopidogrel in patients with ACS for whom an early invasive management strategy is planned.11 The high-dose regimen involves a 600-mg loading dose followed by 150 mg/day for 1 week and then 75 mg/day for 3 weeks, whereas the standard-dose regimen involves a 300-mg loading dose followed by 75 mg/day for 4 weeks. Both groups are being further randomized to low-dose aspirin (75 to 100 mg/day) or high-dose aspirin (300 to 325 mg/day) for 30 days after PCI. With a target enrollment well beyond 10,000 patients, CURRENT should definitively clarify the relative efficacy and safety of high-dose clopidogrel in this setting.
Tailoring clopidogrel therapy
Investigators have explored tailoring the dosing of clopidogrel around the time of PCI based on the degree of platelet inhibition. In one study, administering additional loading doses of clopidogrel, up to a total of 2,400 mg, before PCI in patients with a suboptimal degree of platelet inhibition resulted in a lower rate of ischemic complications following PCI.12
PRASUGREL, A NOVEL THIENOPYRIDINE
Prasugrel is an investigational third-generation thienopyridine currently under US Food and Drug Administration (FDA) review for use in patients with ACS being managed with PCI. Like clopidogrel, prasugrel is a prodrug that requires conversion to an active metabolite prior to binding to the platelet P2Y12 receptor for ADP to confer antiplatelet activity. Prasugrel is metabolized more efficiently than clopidogrel, allowing for faster activation and superior bioavailability to produce a greater and more consistent antiplatelet effect.1,13
The active metabolites of clopidogrel and prasugrel are no different in their ability to inhibit platelet aggregation, but approximately 85% of clopidogrel is inactivated by esterases, with the remaining 15% being converted to the active metabolite using the cytochrome P450 pathway via two successive oxidative steps in the liver.14 In contrast, esterases facilitate the transformation of prasugrel to its active metabolite.14 This activation requires only one oxidative step that can occur in either the liver or the gut through cytochrome P450.
Both prasugrel and clopidogrel are irreversible P2Y12 receptor blockers. For this reason, one must wait approximately 5 days after the last dose of either medication for generation of a sufficient number of new platelets to allow restoration of normal platelet-mediated hemostasis.
Inhibition of platelet aggregation relative to clopidogrel
In a study among healthy volunteers, inhibition of platelet aggregation was significantly higher after a 60-mg loading dose of prasugrel compared with a 300-mg loading dose of clopidogrel.13 Further, suboptimal responders to clopidogrel who crossed over to prasugrel had levels of platelet inhibition as high as 80% following prasugrel administration. The time to peak effect of prasugrel was about 1 hour. Inhibition of platelet aggregation was more consistent following dosing of prasugrel compared with clopidogrel.13
In a study of 201 patients undergoing cardiac catheterization with planned PCI, Wiviott et al demonstrated better levels of inhibition of platelet aggregation at 6 hours after a 60-mg loading dose of prasugrel than after a 600-mg loading dose of clopidogrel (P < .0001).1
Clinical effects relative to clopidogrel: TRITON-TIMI 38
A large phase 3 clinical trial—the Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel–Thrombolysis in Myocardial Infarction (TRITON-TIMI) 38—was conducted to compare the effects of prasugrel and standard-dose clopidogrel on death and ischemic end points in 13,608 patients with ACS scheduled to undergo PCI.15 Patients randomized to clopidogrel were given the standard regimen of a 300-mg loading dose followed by a 75-mg daily maintenance dose; those randomized to prasugrel were given a 60-mg loading dose followed by a 10-mg daily maintenance dose. The study drug was typically given immediately before PCI, a time frame that may mimic real-life use but that favored the faster-onset prasugrel over the slower-onset clopidogrel. Both groups also received low-dose aspirin. Approximately half of the patients in each group were treated with a glycoprotein IIb/IIIa inhibitor. The median duration of therapy was approximately 15 months.
Efficacy. The primary end point—a composite of cardiovascular death, MI, or stroke—occurred in 9.9% of patients randomized to prasugrel compared with 12.1% of those randomized to clopidogrel, corresponding to a 19% relative risk reduction (P = .0004) with prasugrel. Based on these results, 46 patients would need to be treated with prasugrel rather than with clopidogrel to prevent 1 additional cardiovascular death, MI, or stroke.15
The reduction in the primary end point with prasugrel was driven primarily by a reduction in nonfatal MI; nonsignificant trends favored prasugrel over clopidogrel on rates of cardiovascular death and all-cause mortality, but there was no difference in stroke rates. Prasugrel’s effect was consistent across subgroups based on MI type, sex, age, the type of stent used, adjunctive antithrombotic therapy, and renal function.15
In the subgroup of patients with diabetes, the relative reduction in the primary end point with prasugrel compared with clopidogrel was 30% (P < .001), and the respective relative reduction among patients with diabetes who required insulin was 37%.16
Safety. Higher antiplatelet potency carries the trade-off of increased bleeding, and this trade-off was apparent with prasugrel in TRITON-TIMI 38.15 TIMI major bleeding (not counting bleeding related to coronary artery bypass graft surgery [CABG]) occurred significantly more often in prasugrel-treated subjects than in those receiving clopidogrel (2.4% vs 1.8%; P = .03), as did life-threatening bleeds (1.4% vs 0.9%; P = .01). Because absolute rates of major bleeding were low in each treatment group, based on these results, 167 patients would need to be treated with prasugrel rather than clopidogrel to result in 1 excess non-CABG-related major bleeding episode. Rates of intracranial hemorrhage were identical in the two treatment groups.15
Net clinical outcome and therapeutic considerations. Overall analysis of the balance of efficacy and safety in TRITON-TIMI 38 revealed that 138 events were prevented with randomization to prasugrel instead of clopidogrel, at a cost of 35 additional TIMI major bleeds (Figure 2).15
In a post hoc analysis of net clinical outcome, in which major bleeding events were added to the primary composite efficacy end point, prasugrel was associated with a 13% relative risk reduction (P = .004).15 Twenty-three MIs were prevented per 1,000 treated patients with the use of prasugrel instead of clopidogrel, at a cost of 6 excess non-CABG-related major bleeds.15
Another post hoc assessment identified three subgroups who had a significantly increased risk of TIMI major bleeds with randomization to prasugrel15:
- Patients aged 75 years or older
- Patients with a body weight less than 60 kg
- Patients with a history of stroke or transient ischemic attack (TIA).
In these three subgroups, the net clinical effect either was neutral (for those aged ≥ 75years and for those weighing < 60 kg) or favored clopidogrel (for those with a history of stroke or TIA). The group with a history of stroke or TIA represented 4% of the entire cohort, and the TRITON-TIMI 38 investigators recommended avoiding prasugrel in patients with a history of these events. The other two subgroups with a significantly increased bleeding risk with prasugrel represented 16% of the entire cohort, and in these two groups the investigators suggested a pharmacokinetics-guided reduction in the maintenance dose of prasugrel, although a recommendation for such dosing is based on modeling and not actual outcomes data.15
Stent thrombosis. A subanalysis of TRITON-TIMI 38 examined the risk of stent thrombosis in the 12,844 patients enrolled in the trial who had stents implanted.17 Stent thrombosis was assessed using the Academic Research Consortium definitions of definite, probable, and possible stent thrombosis.18 The risk of definite or probable stent thrombosis was halved (hazard ratio = 0.48; P < .0001) with the use of prasugrel compared with clopidogrel, and the reduction was highly significant regardless of the type of stent implanted or the way stent thrombosis was defined. Significant reductions in both early (within the first 30 days) stent thrombosis (P < .0001) and late (beyond 30 days) stent thrombosis (P = .03) were observed in the prasugrel arm compared with the clopidogrel arm.17
AZD6140, A REVERSIBLE P2Y12 RECEPTOR ANTAGONIST
AZD6140, another investigational antiplatelet agent, is an orally active reversible P2Y12 receptor antagonist, in contrast to the thienopyridines, which are irreversible inhibitors. A member of the cyclo-pentyl-triazolo-pyrimidine (CPTP) class, AZD6140 has a rapid onset of action (≤ 2 hours) and does not require metabolic activation. Its plasma half-life is approximately 12 hours, which translates to twice-daily dosing.
Inhibition of platelet aggregation relative to clopidogrel
In a study of clopidogrel-naïve patients with ACS, inhibition of platelet aggregation 12 hours after administration of AZD6140 was approximately 75% with 90-mg, 180-mg, and 270-mg doses, significantly greater than the 30% inhibition achieved after administration of 300 mg of clopidogrel (P < .0002 for all doses of AZD6140 vs clopidogrel).19 Whereas steady state was achieved in approximately 4 to 6 hours with clopidogrel, it was achieved in approximately 2 hours or less with AZD6140.
Clinical safety and efficacy relative to clopidogrel
In a dose-ranging study of AZD6140, adjudicated bleeding rates were similar among two different doses of AZD6140 (90 mg twice daily and 180 mg twice daily) and clopidogrel 75 mg once daily, with no evidence of a dose effect for major bleeding with AZD6140.20 Although this study, conducted in 990 patients with ACS, was underpowered for efficacy end points, rates of adjudicated MI were numerically lower in each of the AZD6140 groups than in the clopidogrel group.
A more definitive evaluation of the relative effcicacy and safety of AZD6140 is expected from the ongoing PLATO trial, which is comparing 90 mg of AZD6140 twice daily with clopidogrel 75 mg/day among 18,000 patients randomized to one of the two treatments within 24 hours of an index ACS event.21
CANGRELOR, A RAPID PARENTERAL P2Y12 RECEPTOR ANTAGONIST
Cangrelor (formerly known as AR-C69931MX) is an intravenously (IV) administered P2Y12 receptor antagonist under investigation for treatment of ACS and use during PCI and other coronary procedures. The compound is an adenosine triphosphate analogue with a plasma half-life of 5 to 9 minutes. Cangrelor is highly reversible, as platelet function returns to normal within 20 minutes of dosing. Within 15 minutes of initiation, cangrelor produces profound platelet inhibition and rapidly achieves steady state; peak effect occurs within minutes.22 The response to cangrelor is highly consistent, with virtually all recipients achieving the same degree of platelet inhibition. Platelet response approaches baseline 15 minutes after termination.22
If approved by the FDA, cangrelor would be administered similar to the way that glycoprotein IIb/IIIa inhibitors are, as it would be used primarily in the catheterization laboratory and then discontinued after the procedure, at which point transition to a long-term oral therapy would be necessary.
Clinical effects relative to abciximab
Cangrelor has been compared with the glycoprotein IIb/IIIa inhibitor abciximab and placebo in 249 patients undergoing elective or urgent PCI.22 Rates of the combined end point of death, MI, or need for repeat revascularization at 30 days were similar with cangrelor and abciximab (5.7% vs 5.4%, respectively; P = NS), both of which were lower than the rate with placebo (10.0%). Major or minor bleeding through 7 days occurred in numerically fewer cangrelor recipients compared with abciximab recipients (7.0% vs 9.0%), although the small sample size precluded evaluation for statistical significance.
Clinical effects relative to clopidogrel—the CHAMPION trials
A phase 3 trial program consisting of two multinational studies of cangrelor—the Cangrelor Versus Standard Therapy to Achieve Optimal Management of Platelet Inhibition (CHAMPION) program—is currently under way.
CHAMPION-PCI is enrolling 9,000 patients presenting with ACS who are being randomized in a double-blind fashion at the start of PCI to a 600-mg loading dose of clopidogrel or to cangrelor given as an IV bolus of 30 μg/kg followed by an IV infusion of 4 μg/kg/min. The primary end point is a composite of all-cause mortality, MI, or ischemia-driven revascularization in the 48 hours following randomization. Secondary end points include rates of all-cause mortality and MI at 48 hours.23
CHAMPION-PLATFORM is enrolling 4,400 patients scheduled for PCI as a result of ACS who are being randomized in a double-blind, double-dummy manner to (1) cangrelor bolus and infusion plus oral placebo or (2) oral clopidogrel plus placebo bolus and infusion before their index procedures. Dosages of the two agents are the same as in CHAMPION-PCI. The primary end point is a composite of death, MI, or urgent target vessel revascularization at 48 hours. Secondary end points include 30-day and 1-year clinical outcomes.23
The rationale for the CHAMPION investigations stems from the need to initiate clopidogrel before a patient is taken to the catheterization laboratory, owing to the inability to achieve a high degree of platelet inhibition until 4 to 6 hours after clopidogrel administration. Although this strategy can be undertaken without complication for most patients, a subset of patients with three-vessel disease or left-main disease will require CABG, which then must be delayed several days until clopidogrel’s platelet-inhibiting effect diminishes. A rapid-acting IV inhibitor of the P2Y12 receptor such as cangrelor would obviate this concern.
THROMBIN INHIBITORS
Thrombin plays an important role in platelet activation, and thrombin receptor antagonists may represent a safer means of inhibiting platelet activation relative to traditional antiplatelet agents. This theoretical safety advantage stems from the notion that blocking the action of platelets at the thrombin receptor would preserve platelets’ function as mediators of primary hemostasis. Because thrombin’s activation of platelets should occur only during clot formation, blocking platelet activation at the thrombin receptor would interrupt thrombin’s ability to propagate platelet activation during formation of coronary artery clots.
One agent in this class that is being studied extensively is SCH 530348, an oral thrombin receptor antagonist with potent antiplatelet activity. Its peak antiplatelet potency is achieved within hours when a loading dose is given, and within days without a loading dose. Wearing-off of the action of SCH 530348 takes weeks.24
Inhibition of platelet aggregation with thrombin receptor antagonists is measured in response to the thrombin receptor antagonist peptide (TRAP), not ADP. The proportion of subjects treated with SCH 530348 who achieve greater than 80% inhibition of platelet aggregation to 15 μM of TRAP ranges from 91% (with 0.5 mg of SCH 530348) to 100% (with 1.0 mg and 2.5 mg) at both 30 days and 60 days.25
Clinical effects in placebo-controlled trials
SCH 530348 was studied in the Thrombin Receptor Antagonist (TRA)–PCI trial, a dose-ranging study in which patients were randomized to one of three oral loading doses of the study drug (10 mg, 20 mg, or 40 mg) on top of a clopidogrel loading dose before undergoing cardiac catheterization for planned PCI; patients were then randomized to one of three maintenance doses of SCH 530348 (0.5 mg, 1.0 mg, or 2.5 mg) or placebo (depending on loading therapy) for 60 days.25
Among the 573 patients undergoing PCI , the rate of TIMI major or minor bleeding was not significantly higher with any dose of SCH 530348 compared with placebo,25 supporting the hypothesis that thrombin receptor antagonism inhibits platelet aggregation without a significant increase in bleeding.
Although the TRA-PCI study was not powered to detect differences in clinical event rates, a reduction in the rate of major adverse cardiovascular events was observed in a dose-dependent manner with SCH 530348 compared with placebo in the PCI cohort.25
On the basis of the TRA-PCI trial, a pair of phase 3 trials of SCH 530348 have been launched—the Thrombin Receptor Antagonist in Secondary Prevention of Atherothrombotic Ischemic Events (TRA 2°P-TIMI 50) study and the Thrombin Receptor Antagonist for Clinical Event Reduction in ACS (TRA-CER) study.
TRA 2°P-TIMI 50 is a multinational double-blind study enrolling 19,500 patients with prior MI or stroke or with existing peripheral arterial disease. Patients are being randomized to placebo plus standard medical care (including aspirin and clopidogrel) or to 2.5 mg of SCH 530348 once daily plus standard medical care. The primary end point is the composite of cardiovascular death, MI, urgent coronary revascularization, or stroke.26
TRA-CER is a multinational double-blind study with planned enrollment of 10,000 patients with non-ST-segment-elevation MI. Patients are being randomized to placebo plus standard medical care (including aspirin or clopidogrel) or to SCH 530348 (using the oral 40-mg loading dose and a maintenance dose of 2.5 mg once daily) plus standard medical care. The primary end point is the composite of cardiovascular death, MI, rehospitalization for ACS, urgent coronary revascularization, or stroke. The key secondary end point is the composite of cardiovascular death, MI, or stroke.27
COMPARATIVE CONSIDERATIONS
Inhibition of platelet aggregation
Clopidogrel achieves about 30% inhibition of platelet aggregation to ADP at its current FDA-approved loading dose of 300 mg and about 40% inhibition when its dose is doubled to 600 mg. These levels of inhibition are increased to 75% to 80% by clopidogrel’s fellow thienopyridine prasugrel, and this increase is attributable to prasugrel’s more efficient metabolism from prodrug to active metabolite. The reversible P2Y12 receptor antagonist AZD6140 achieves a comparable 75% to 80% inhibition of platelet aggregation. The parenterally administered P2Y12 receptor antagonist cangrelor achieves greater than 90% inhibition, as does the oral thrombin receptor antagonist SCH 530348, although the latter agent’s inhibition is to the agonist TRAP rather than ADP.
Time to peak effect
The time to peak effect with clopidogrel is approximately 4 hours regardless of the loading dose used (300 mg or 600 mg); this is substantially reduced with all of the investigational agents except SCH 530348. The novel agents’ reduced time to peak effect can offer advantages in speeding patients’ readiness to undergo catheterization procedures. This is particularly true for the IV agent cangrelor, which achieves its peak effect within minutes, although the 1-hour to 2-hour time frame with oral agents prasugrel and AZD6140 also would usually obviate any need to delay catheterization.
Consistency of platelet response
Standard-dose clopidogrel has the least consistency of platelet response among the therapies reviewed. Although increasing the clopidogrel dose yields somewhat greater consistency in response, it is still lower than the very high degrees of consistency observed with all of the novel compounds, each of which appears to achieve the same degree of inhibition of aggregation in virtually all patients.
Offset of effect
Both of the thienopyridines—clopidogrel and prasugrel—have an offset of effect of about 5 days, which requires delay of surgery, if possible, for several days in patients taking these agents. This is not an issue for the reversible oral agent AZD6140, whose offset of action takes just 1 to 2 days. While this rapid wearing-off of effect translates to a potential advantage for AZD6140, it also poses the potential drawback that a missed dose or two may leave the patient exposed to the risk of a thrombotic event. Cangrelor’s rapid offset of 20 minutes promotes its envisioned use as a catheterization lab–based medication like the glycoprotein IIb/IIIa inhibitors that can be started right before a PCI procedure and stopped immediately afterward. Because SCH 530348 has a very long half-life and thus a weeks-long washout period, the practicality of its use may depend on the hypothesis that thrombin receptor antagonists do not interfere with primary hemostasis, which is supported by data to date but remains to be definitively confirmed.
CONCLUSIONS
Clopidogrel achieves modest platelet inhibition with wide variability in response. Higher doses of clopidogrel achieve modestly greater degrees of inhibition than standard doses, and appear to result in a decreased rate of ischemic events. Although higher doses of clopidogrel have been embraced by some clinicians, we await definitive phase 3 trial evidence of net benefit before making high-dose clopidogrel the new standard of care.
Compared with clopidogrel, the investigational thienopyridine prasugrel is a more potent and consistent blocker of the ADP receptor. It results in a decreased rate of ischemic events relative to clopidogrel, including a 50% reduction in the rate of stent thrombosis, but is associated with an increased rate of bleeding. If prasugrel is approved for marketing, its use should be avoided in patients with a history of stroke or TIA, and avoidance or dose adjustment may be necessary in patients aged 75 years or older and in patients weighing less than 60 kg.
Other novel antiplatelet agents being evaluated for use in patients with ACS—the reversible oral ADP receptor blocker AZD6140, the rapid-acting IV ADP receptor blocker cangrelor, and oral thrombin receptor antagonists—offer potential advantages that need to be examined in the context of large-scale clinical trials.
An enhanced understanding of platelet biology, as reviewed in the previous article in this supplement, has made it possible to identify a wide variety of platelet agonists. This knowledge has fostered the development of a host of pharmacologic strategies to block agonists such as cyclooxygenase, thromboxane, adenosine diphosphate (ADP), and thrombin, among others. This article will discuss the pharmacologic properties of novel antiplatelet agents, as well as alternative dosing of the established antiplatelet agent clopidogrel, and will review data from available comparative and placebo-controlled trials of these agents. The article concludes with comparative perspectives on the potential roles and relative advantages of these agents in the evolving management of patients with acute coronary syndromes (ACS).
CLOPIDOGREL AND THE CHALLENGE OF VARIABLE RESPONSE
Clopidogrel, a member of the thienopyridine class of ADP receptor inhibitors, is well established for use in patients with ACS at a loading dose of 300 mg followed by a maintenance dose of 75 mg/day. At this loading dose, inhibition of platelet aggregation to ADP is approximately 30%, and the time to peak effect is approximately 4 to 6 hours.1
As with most other drugs, the response to clopidogrel is variable. However, in contrast to the accepted measures of response to antihypertensive or lipid-lowering drugs, there are no routinely used tests for measuring response to antiplatelet therapies. As a result, a “one size fits all” strategy in the dosing of clopidogrel has prevailed.
This variability in response is clinically relevant. In a study assessing clopidogrel responsiveness by ADP-induced platelet aggregation in 60 patients who experienced ST-segment-elevation myocardial infarction (MI), Matetzky et al found that the lowest levels of clopidogrel responsiveness were associated with a significantly elevated rate (P = .007) of recurrent cardiovascular events 6 months after the MI.3 Gurbel et al found a similar association between clopidogrel responsiveness and subacute stent thrombosis in a study of 120 patients using two different methods—light transmission aggregotomy to 5 μmol/L of ADP, and the ratio of vasodilator-stimulated phosphoprotein reactivity—to assess clopidogrel responsiveness.4
Increasing the loading dose raises response rates
One proposed method for boosting responsiveness to clopidogrel in suboptimal responders is the use of a higher dose. In a study of 190 patients undergoing coronary stenting, increasing the loading dose from 300 mg to 600 mg reduced the rate of clopidogrel resistance (defined as a < 10% absolute change in aggregation to 5 μM of ADP at 24 hours) from 28% to 8% (P < .001),5 a finding that supports the notion of enhanced response at doses up to 600 mg. Single loading doses in excess of 600 mg yield diminishing returns in terms of platelet inhibition, most likely as a result of clopidogrel pharmacokinetics.6
Compared with 300 mg of clopidogrel, the more potent platelet inhibitory effect of a 600-mg dose translated to a two-thirds reduction (P = .041) in the composite end point of death, MI, or target vessel revascularization at 30 days in a study of 255 patients with stable coronary artery disease undergoing percutaneous coronary intervention (PCI).7 The reduction in this composite end point with high-dose clopidogrel was driven by a reduction in the incidence of periprocedural MI.
In a separate study of 292 patients with non‑ST-segment-elevation ACS who were scheduled for PCI, the superior platelet response to 600 mg versus 300 mg of clopidogrel translated to a 60% reduction in adverse thrombotic events (P = .02), and this benefit extended beyond rates of periprocedural MI.8
Similar results with increased maintenance dose
Similarly, emerging data suggest that raising the maintenance dose of clopidogrel can also raise response rates. In a study of 60 patients, doubling the maintenance dose of clopidogrel after PCI from 75 mg/day to 150 mg/day resulted in improved platelet inhibition as assessed by rapid platelet function analysis.9 Likewise, a 150-mg/day maintenance dose of clopidogrel was associated with a superior antiplatelet effect compared with 75 mg/day in a study of 40 patients with type 2 diabetes.10
Large definitive trial is under way
In the wake of these smaller trials, a large randomized trial known as CURRENT is comparing a strategy of high-dose clopidogrel with standard-dose clopidogrel in patients with ACS for whom an early invasive management strategy is planned.11 The high-dose regimen involves a 600-mg loading dose followed by 150 mg/day for 1 week and then 75 mg/day for 3 weeks, whereas the standard-dose regimen involves a 300-mg loading dose followed by 75 mg/day for 4 weeks. Both groups are being further randomized to low-dose aspirin (75 to 100 mg/day) or high-dose aspirin (300 to 325 mg/day) for 30 days after PCI. With a target enrollment well beyond 10,000 patients, CURRENT should definitively clarify the relative efficacy and safety of high-dose clopidogrel in this setting.
Tailoring clopidogrel therapy
Investigators have explored tailoring the dosing of clopidogrel around the time of PCI based on the degree of platelet inhibition. In one study, administering additional loading doses of clopidogrel, up to a total of 2,400 mg, before PCI in patients with a suboptimal degree of platelet inhibition resulted in a lower rate of ischemic complications following PCI.12
PRASUGREL, A NOVEL THIENOPYRIDINE
Prasugrel is an investigational third-generation thienopyridine currently under US Food and Drug Administration (FDA) review for use in patients with ACS being managed with PCI. Like clopidogrel, prasugrel is a prodrug that requires conversion to an active metabolite prior to binding to the platelet P2Y12 receptor for ADP to confer antiplatelet activity. Prasugrel is metabolized more efficiently than clopidogrel, allowing for faster activation and superior bioavailability to produce a greater and more consistent antiplatelet effect.1,13
The active metabolites of clopidogrel and prasugrel are no different in their ability to inhibit platelet aggregation, but approximately 85% of clopidogrel is inactivated by esterases, with the remaining 15% being converted to the active metabolite using the cytochrome P450 pathway via two successive oxidative steps in the liver.14 In contrast, esterases facilitate the transformation of prasugrel to its active metabolite.14 This activation requires only one oxidative step that can occur in either the liver or the gut through cytochrome P450.
Both prasugrel and clopidogrel are irreversible P2Y12 receptor blockers. For this reason, one must wait approximately 5 days after the last dose of either medication for generation of a sufficient number of new platelets to allow restoration of normal platelet-mediated hemostasis.
Inhibition of platelet aggregation relative to clopidogrel
In a study among healthy volunteers, inhibition of platelet aggregation was significantly higher after a 60-mg loading dose of prasugrel compared with a 300-mg loading dose of clopidogrel.13 Further, suboptimal responders to clopidogrel who crossed over to prasugrel had levels of platelet inhibition as high as 80% following prasugrel administration. The time to peak effect of prasugrel was about 1 hour. Inhibition of platelet aggregation was more consistent following dosing of prasugrel compared with clopidogrel.13
In a study of 201 patients undergoing cardiac catheterization with planned PCI, Wiviott et al demonstrated better levels of inhibition of platelet aggregation at 6 hours after a 60-mg loading dose of prasugrel than after a 600-mg loading dose of clopidogrel (P < .0001).1
Clinical effects relative to clopidogrel: TRITON-TIMI 38
A large phase 3 clinical trial—the Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel–Thrombolysis in Myocardial Infarction (TRITON-TIMI) 38—was conducted to compare the effects of prasugrel and standard-dose clopidogrel on death and ischemic end points in 13,608 patients with ACS scheduled to undergo PCI.15 Patients randomized to clopidogrel were given the standard regimen of a 300-mg loading dose followed by a 75-mg daily maintenance dose; those randomized to prasugrel were given a 60-mg loading dose followed by a 10-mg daily maintenance dose. The study drug was typically given immediately before PCI, a time frame that may mimic real-life use but that favored the faster-onset prasugrel over the slower-onset clopidogrel. Both groups also received low-dose aspirin. Approximately half of the patients in each group were treated with a glycoprotein IIb/IIIa inhibitor. The median duration of therapy was approximately 15 months.
Efficacy. The primary end point—a composite of cardiovascular death, MI, or stroke—occurred in 9.9% of patients randomized to prasugrel compared with 12.1% of those randomized to clopidogrel, corresponding to a 19% relative risk reduction (P = .0004) with prasugrel. Based on these results, 46 patients would need to be treated with prasugrel rather than with clopidogrel to prevent 1 additional cardiovascular death, MI, or stroke.15
The reduction in the primary end point with prasugrel was driven primarily by a reduction in nonfatal MI; nonsignificant trends favored prasugrel over clopidogrel on rates of cardiovascular death and all-cause mortality, but there was no difference in stroke rates. Prasugrel’s effect was consistent across subgroups based on MI type, sex, age, the type of stent used, adjunctive antithrombotic therapy, and renal function.15
In the subgroup of patients with diabetes, the relative reduction in the primary end point with prasugrel compared with clopidogrel was 30% (P < .001), and the respective relative reduction among patients with diabetes who required insulin was 37%.16
Safety. Higher antiplatelet potency carries the trade-off of increased bleeding, and this trade-off was apparent with prasugrel in TRITON-TIMI 38.15 TIMI major bleeding (not counting bleeding related to coronary artery bypass graft surgery [CABG]) occurred significantly more often in prasugrel-treated subjects than in those receiving clopidogrel (2.4% vs 1.8%; P = .03), as did life-threatening bleeds (1.4% vs 0.9%; P = .01). Because absolute rates of major bleeding were low in each treatment group, based on these results, 167 patients would need to be treated with prasugrel rather than clopidogrel to result in 1 excess non-CABG-related major bleeding episode. Rates of intracranial hemorrhage were identical in the two treatment groups.15
Net clinical outcome and therapeutic considerations. Overall analysis of the balance of efficacy and safety in TRITON-TIMI 38 revealed that 138 events were prevented with randomization to prasugrel instead of clopidogrel, at a cost of 35 additional TIMI major bleeds (Figure 2).15
In a post hoc analysis of net clinical outcome, in which major bleeding events were added to the primary composite efficacy end point, prasugrel was associated with a 13% relative risk reduction (P = .004).15 Twenty-three MIs were prevented per 1,000 treated patients with the use of prasugrel instead of clopidogrel, at a cost of 6 excess non-CABG-related major bleeds.15
Another post hoc assessment identified three subgroups who had a significantly increased risk of TIMI major bleeds with randomization to prasugrel15:
- Patients aged 75 years or older
- Patients with a body weight less than 60 kg
- Patients with a history of stroke or transient ischemic attack (TIA).
In these three subgroups, the net clinical effect either was neutral (for those aged ≥ 75years and for those weighing < 60 kg) or favored clopidogrel (for those with a history of stroke or TIA). The group with a history of stroke or TIA represented 4% of the entire cohort, and the TRITON-TIMI 38 investigators recommended avoiding prasugrel in patients with a history of these events. The other two subgroups with a significantly increased bleeding risk with prasugrel represented 16% of the entire cohort, and in these two groups the investigators suggested a pharmacokinetics-guided reduction in the maintenance dose of prasugrel, although a recommendation for such dosing is based on modeling and not actual outcomes data.15
Stent thrombosis. A subanalysis of TRITON-TIMI 38 examined the risk of stent thrombosis in the 12,844 patients enrolled in the trial who had stents implanted.17 Stent thrombosis was assessed using the Academic Research Consortium definitions of definite, probable, and possible stent thrombosis.18 The risk of definite or probable stent thrombosis was halved (hazard ratio = 0.48; P < .0001) with the use of prasugrel compared with clopidogrel, and the reduction was highly significant regardless of the type of stent implanted or the way stent thrombosis was defined. Significant reductions in both early (within the first 30 days) stent thrombosis (P < .0001) and late (beyond 30 days) stent thrombosis (P = .03) were observed in the prasugrel arm compared with the clopidogrel arm.17
AZD6140, A REVERSIBLE P2Y12 RECEPTOR ANTAGONIST
AZD6140, another investigational antiplatelet agent, is an orally active reversible P2Y12 receptor antagonist, in contrast to the thienopyridines, which are irreversible inhibitors. A member of the cyclo-pentyl-triazolo-pyrimidine (CPTP) class, AZD6140 has a rapid onset of action (≤ 2 hours) and does not require metabolic activation. Its plasma half-life is approximately 12 hours, which translates to twice-daily dosing.
Inhibition of platelet aggregation relative to clopidogrel
In a study of clopidogrel-naïve patients with ACS, inhibition of platelet aggregation 12 hours after administration of AZD6140 was approximately 75% with 90-mg, 180-mg, and 270-mg doses, significantly greater than the 30% inhibition achieved after administration of 300 mg of clopidogrel (P < .0002 for all doses of AZD6140 vs clopidogrel).19 Whereas steady state was achieved in approximately 4 to 6 hours with clopidogrel, it was achieved in approximately 2 hours or less with AZD6140.
Clinical safety and efficacy relative to clopidogrel
In a dose-ranging study of AZD6140, adjudicated bleeding rates were similar among two different doses of AZD6140 (90 mg twice daily and 180 mg twice daily) and clopidogrel 75 mg once daily, with no evidence of a dose effect for major bleeding with AZD6140.20 Although this study, conducted in 990 patients with ACS, was underpowered for efficacy end points, rates of adjudicated MI were numerically lower in each of the AZD6140 groups than in the clopidogrel group.
A more definitive evaluation of the relative effcicacy and safety of AZD6140 is expected from the ongoing PLATO trial, which is comparing 90 mg of AZD6140 twice daily with clopidogrel 75 mg/day among 18,000 patients randomized to one of the two treatments within 24 hours of an index ACS event.21
CANGRELOR, A RAPID PARENTERAL P2Y12 RECEPTOR ANTAGONIST
Cangrelor (formerly known as AR-C69931MX) is an intravenously (IV) administered P2Y12 receptor antagonist under investigation for treatment of ACS and use during PCI and other coronary procedures. The compound is an adenosine triphosphate analogue with a plasma half-life of 5 to 9 minutes. Cangrelor is highly reversible, as platelet function returns to normal within 20 minutes of dosing. Within 15 minutes of initiation, cangrelor produces profound platelet inhibition and rapidly achieves steady state; peak effect occurs within minutes.22 The response to cangrelor is highly consistent, with virtually all recipients achieving the same degree of platelet inhibition. Platelet response approaches baseline 15 minutes after termination.22
If approved by the FDA, cangrelor would be administered similar to the way that glycoprotein IIb/IIIa inhibitors are, as it would be used primarily in the catheterization laboratory and then discontinued after the procedure, at which point transition to a long-term oral therapy would be necessary.
Clinical effects relative to abciximab
Cangrelor has been compared with the glycoprotein IIb/IIIa inhibitor abciximab and placebo in 249 patients undergoing elective or urgent PCI.22 Rates of the combined end point of death, MI, or need for repeat revascularization at 30 days were similar with cangrelor and abciximab (5.7% vs 5.4%, respectively; P = NS), both of which were lower than the rate with placebo (10.0%). Major or minor bleeding through 7 days occurred in numerically fewer cangrelor recipients compared with abciximab recipients (7.0% vs 9.0%), although the small sample size precluded evaluation for statistical significance.
Clinical effects relative to clopidogrel—the CHAMPION trials
A phase 3 trial program consisting of two multinational studies of cangrelor—the Cangrelor Versus Standard Therapy to Achieve Optimal Management of Platelet Inhibition (CHAMPION) program—is currently under way.
CHAMPION-PCI is enrolling 9,000 patients presenting with ACS who are being randomized in a double-blind fashion at the start of PCI to a 600-mg loading dose of clopidogrel or to cangrelor given as an IV bolus of 30 μg/kg followed by an IV infusion of 4 μg/kg/min. The primary end point is a composite of all-cause mortality, MI, or ischemia-driven revascularization in the 48 hours following randomization. Secondary end points include rates of all-cause mortality and MI at 48 hours.23
CHAMPION-PLATFORM is enrolling 4,400 patients scheduled for PCI as a result of ACS who are being randomized in a double-blind, double-dummy manner to (1) cangrelor bolus and infusion plus oral placebo or (2) oral clopidogrel plus placebo bolus and infusion before their index procedures. Dosages of the two agents are the same as in CHAMPION-PCI. The primary end point is a composite of death, MI, or urgent target vessel revascularization at 48 hours. Secondary end points include 30-day and 1-year clinical outcomes.23
The rationale for the CHAMPION investigations stems from the need to initiate clopidogrel before a patient is taken to the catheterization laboratory, owing to the inability to achieve a high degree of platelet inhibition until 4 to 6 hours after clopidogrel administration. Although this strategy can be undertaken without complication for most patients, a subset of patients with three-vessel disease or left-main disease will require CABG, which then must be delayed several days until clopidogrel’s platelet-inhibiting effect diminishes. A rapid-acting IV inhibitor of the P2Y12 receptor such as cangrelor would obviate this concern.
THROMBIN INHIBITORS
Thrombin plays an important role in platelet activation, and thrombin receptor antagonists may represent a safer means of inhibiting platelet activation relative to traditional antiplatelet agents. This theoretical safety advantage stems from the notion that blocking the action of platelets at the thrombin receptor would preserve platelets’ function as mediators of primary hemostasis. Because thrombin’s activation of platelets should occur only during clot formation, blocking platelet activation at the thrombin receptor would interrupt thrombin’s ability to propagate platelet activation during formation of coronary artery clots.
One agent in this class that is being studied extensively is SCH 530348, an oral thrombin receptor antagonist with potent antiplatelet activity. Its peak antiplatelet potency is achieved within hours when a loading dose is given, and within days without a loading dose. Wearing-off of the action of SCH 530348 takes weeks.24
Inhibition of platelet aggregation with thrombin receptor antagonists is measured in response to the thrombin receptor antagonist peptide (TRAP), not ADP. The proportion of subjects treated with SCH 530348 who achieve greater than 80% inhibition of platelet aggregation to 15 μM of TRAP ranges from 91% (with 0.5 mg of SCH 530348) to 100% (with 1.0 mg and 2.5 mg) at both 30 days and 60 days.25
Clinical effects in placebo-controlled trials
SCH 530348 was studied in the Thrombin Receptor Antagonist (TRA)–PCI trial, a dose-ranging study in which patients were randomized to one of three oral loading doses of the study drug (10 mg, 20 mg, or 40 mg) on top of a clopidogrel loading dose before undergoing cardiac catheterization for planned PCI; patients were then randomized to one of three maintenance doses of SCH 530348 (0.5 mg, 1.0 mg, or 2.5 mg) or placebo (depending on loading therapy) for 60 days.25
Among the 573 patients undergoing PCI , the rate of TIMI major or minor bleeding was not significantly higher with any dose of SCH 530348 compared with placebo,25 supporting the hypothesis that thrombin receptor antagonism inhibits platelet aggregation without a significant increase in bleeding.
Although the TRA-PCI study was not powered to detect differences in clinical event rates, a reduction in the rate of major adverse cardiovascular events was observed in a dose-dependent manner with SCH 530348 compared with placebo in the PCI cohort.25
On the basis of the TRA-PCI trial, a pair of phase 3 trials of SCH 530348 have been launched—the Thrombin Receptor Antagonist in Secondary Prevention of Atherothrombotic Ischemic Events (TRA 2°P-TIMI 50) study and the Thrombin Receptor Antagonist for Clinical Event Reduction in ACS (TRA-CER) study.
TRA 2°P-TIMI 50 is a multinational double-blind study enrolling 19,500 patients with prior MI or stroke or with existing peripheral arterial disease. Patients are being randomized to placebo plus standard medical care (including aspirin and clopidogrel) or to 2.5 mg of SCH 530348 once daily plus standard medical care. The primary end point is the composite of cardiovascular death, MI, urgent coronary revascularization, or stroke.26
TRA-CER is a multinational double-blind study with planned enrollment of 10,000 patients with non-ST-segment-elevation MI. Patients are being randomized to placebo plus standard medical care (including aspirin or clopidogrel) or to SCH 530348 (using the oral 40-mg loading dose and a maintenance dose of 2.5 mg once daily) plus standard medical care. The primary end point is the composite of cardiovascular death, MI, rehospitalization for ACS, urgent coronary revascularization, or stroke. The key secondary end point is the composite of cardiovascular death, MI, or stroke.27
COMPARATIVE CONSIDERATIONS
Inhibition of platelet aggregation
Clopidogrel achieves about 30% inhibition of platelet aggregation to ADP at its current FDA-approved loading dose of 300 mg and about 40% inhibition when its dose is doubled to 600 mg. These levels of inhibition are increased to 75% to 80% by clopidogrel’s fellow thienopyridine prasugrel, and this increase is attributable to prasugrel’s more efficient metabolism from prodrug to active metabolite. The reversible P2Y12 receptor antagonist AZD6140 achieves a comparable 75% to 80% inhibition of platelet aggregation. The parenterally administered P2Y12 receptor antagonist cangrelor achieves greater than 90% inhibition, as does the oral thrombin receptor antagonist SCH 530348, although the latter agent’s inhibition is to the agonist TRAP rather than ADP.
Time to peak effect
The time to peak effect with clopidogrel is approximately 4 hours regardless of the loading dose used (300 mg or 600 mg); this is substantially reduced with all of the investigational agents except SCH 530348. The novel agents’ reduced time to peak effect can offer advantages in speeding patients’ readiness to undergo catheterization procedures. This is particularly true for the IV agent cangrelor, which achieves its peak effect within minutes, although the 1-hour to 2-hour time frame with oral agents prasugrel and AZD6140 also would usually obviate any need to delay catheterization.
Consistency of platelet response
Standard-dose clopidogrel has the least consistency of platelet response among the therapies reviewed. Although increasing the clopidogrel dose yields somewhat greater consistency in response, it is still lower than the very high degrees of consistency observed with all of the novel compounds, each of which appears to achieve the same degree of inhibition of aggregation in virtually all patients.
Offset of effect
Both of the thienopyridines—clopidogrel and prasugrel—have an offset of effect of about 5 days, which requires delay of surgery, if possible, for several days in patients taking these agents. This is not an issue for the reversible oral agent AZD6140, whose offset of action takes just 1 to 2 days. While this rapid wearing-off of effect translates to a potential advantage for AZD6140, it also poses the potential drawback that a missed dose or two may leave the patient exposed to the risk of a thrombotic event. Cangrelor’s rapid offset of 20 minutes promotes its envisioned use as a catheterization lab–based medication like the glycoprotein IIb/IIIa inhibitors that can be started right before a PCI procedure and stopped immediately afterward. Because SCH 530348 has a very long half-life and thus a weeks-long washout period, the practicality of its use may depend on the hypothesis that thrombin receptor antagonists do not interfere with primary hemostasis, which is supported by data to date but remains to be definitively confirmed.
CONCLUSIONS
Clopidogrel achieves modest platelet inhibition with wide variability in response. Higher doses of clopidogrel achieve modestly greater degrees of inhibition than standard doses, and appear to result in a decreased rate of ischemic events. Although higher doses of clopidogrel have been embraced by some clinicians, we await definitive phase 3 trial evidence of net benefit before making high-dose clopidogrel the new standard of care.
Compared with clopidogrel, the investigational thienopyridine prasugrel is a more potent and consistent blocker of the ADP receptor. It results in a decreased rate of ischemic events relative to clopidogrel, including a 50% reduction in the rate of stent thrombosis, but is associated with an increased rate of bleeding. If prasugrel is approved for marketing, its use should be avoided in patients with a history of stroke or TIA, and avoidance or dose adjustment may be necessary in patients aged 75 years or older and in patients weighing less than 60 kg.
Other novel antiplatelet agents being evaluated for use in patients with ACS—the reversible oral ADP receptor blocker AZD6140, the rapid-acting IV ADP receptor blocker cangrelor, and oral thrombin receptor antagonists—offer potential advantages that need to be examined in the context of large-scale clinical trials.
- Wiviott SD, Trenk D, Frelinger AL, et al. Prasugrel compared with high loading- and maintenance-dose clopidogrel in patients with planned percutaneous coronary intervention: the Prasugrel in Comparison to Clopidogrel for Inhibition of Platelet Activation and Aggregation–Thrombolysis in Myocardial Infarction 44 trial. Circulation 2007; 116:2923–2932.
- Serebruany VL, Steinhubl SR, Berger PB, Malinin AI, Bhatt DL, Topol EJ. Variability in platelet responsiveness to clopidogrel among 544 individuals. J Am Coll Cardiol 2005; 45:246–251.
- Matetzky S, Shenkman B, Guetta V, et al. Clopidogrel resistance is associated with increased risk of recurrent atherothrombotic events in patients with acute myocardial infarction. Circulation 2004; 109:3171–3175.
- Gurbel PA, Bliden KP, Samara W, et al. Clopidogrel effect on platelet reactivity in patients with stent thrombosis: results of the CREST Study. J Am Coll Cardiol 2005; 46:1827–1832.
- Gurbel PA, Bliden KP, Hayes KM, Yoho JA, Herzog WR, Tantry US. The relation of dosing to clopidogrel responsiveness and the incidence of high post-treatment platelet aggregation in patients undergoing coronary stenting. J Am Coll Cardiol 2005; 45:1392–1396.
- von Beckerath N, Taubert D, Pogatsa-Murray G, Schömig E, Kastrati A, Schömig A. Absorption, metabolization, and antiplatelet effects of 300-, 600-, and 900-mg loading doses of clopidogrel: results of the ISAR-CHOICE (Intracoronary Stenting and Antithrombotic Regimen: Choose Between 3 High Oral Doses for Immediate Clopidogrel Effect) Trial. Circulation 2005; 112:2946–2950.
- Patti G, Colonna G, Pasceri V, Pepe LL, Montinaro A, Di Sciascio G. Randomized trial of high loading dose of clopidogrel for reduction of periprocedural myocardial infarction in patients undergoing coronary intervention: results from the ARMYDA-2 (Antiplatelet therapy for Reduction of MYocardial Damage during Angioplasty) study. Circulation 2005; 111:2099–2106.
- Cuisset T, Frere C, Quilici J, et al. Benefit of a 600-mg loading dose of clopidogrel on platelet reactivity and clinical outcomes in patients with non-ST-segment elevation acute coronary syndrome undergoing coronary stenting. J Am Coll Cardiol 2006; 48:1339–1345.
- von Beckerath N, Kastrati A, Wieczorek A, et al. A double-blind, randomized study on platelet aggregation in patients treated with a daily dose of 150 or 75 mg of clopidogrel for 30 days. Eur Heart J 2007; 28:1814–1819.
- Angiolillo DJ, Shoemaker SB, Desai B, et al. Randomized comparison of a high clopidogrel maintenance dose in patients with diabetes mellitus and coronary artery disease: results of the Optimizing Antiplatelet Therapy in Diabetes Mellitus (OPTIMUS) study. Circulation 2007; 115:708–716.
- Clopidogrel optimal loading dose usage to reduce recurrent events/optimal antiplatelet strategy for interventions (CURRENT/OASIS7). Clinical Trials.gov Web site. http://clinicaltrials.gov/ct2/show/NCT00335452. Updated September 1, 2008. Accessed December 16, 2008.
- Bonello L, Camoin-Jau L, Arques S, et al. Adjusted clopidogrel loading doses according to vasodilator-stimulated phosphoprotein phosphorylation index decrease rate of major adverse cardiovascular events in patients with clopidogrel resistance: a multicenter randomized prospective study. J Am Coll Cardiol 2008; 51:1404–1411.
- Brandt JT, Payne CD, Wiviott SD, et al. A comparison of prasugrel and clopidogrel loading doses on platelet function: magnitude of platelet inhibition is related to active metabolic formation. Am Heart J 2007; 153:66.e9–e16.
- Herbert JM, Savi P. P2Y12, a new platelet ADP receptor, target of clopidogrel. Semin Vasc Med 2003; 3:113–122.
- Wiviott SD, Braunwald E, McCabe CH, et al. Prasugrel versus clopidogrel in patients with acute coronary syndromes. N Engl J Med 2007; 357:2001–2015.
- Wiviott SD, Braunwald E, Angiolillo DJ, et al. Greater clinical benefit of more intensive oral antiplatelet therapy with prasugrel in patients with diabetes mellitus in the Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel–Thrombolysis in Myocardial Infarction 38. Circulation 2008; 118:1626–1636.
- Wiviott SD, Braunwald E, McCabe CH, et al. Intensive oral antiplatelet therapy for reduction of ischaemic events including stent thrombosis in patients with acute coronary syndromes treated with percutaneous coronary intervention and stenting in the TRITON-TIMI 38 trial: a subanalysis of a randomised trial. Lancet 2008; 371:1353–1363.
- Mauri L, Hsieh WH, Massaro JM, Ho KK, D’Agostino R, Cutlip DE. Stent thrombosis in randomized clinical trials of drug-eluting stents. N Engl J Med 2007; 356:1020–1029.
- Storey RF, Husted S, Harrington RA, et al. Inhibition of platelet aggregation by AZD6140, a reversible oral P2Y12 receptor antagonist, compared with clopidogrel in patients with acute coronary syndromes. J Am Coll Cardiol 2007; 50:1852–1856.
- Cannon CP, Husted S, Harrington RA, et al. Safety, tolerability, and initial efficacy of AZD6140, the first reversible oral adenosine diphosphate receptor antagonist, compared with clopidogrel, in patients with non-ST-segment elevation acute coronary syndrome: primary results of the DISPERSE-2 trial. J Am Coll Cardiol 2007; 50:1844–1851.
- A comparison of AZD6140 and clopidogrel in patients with acute coronary syndrome (PLATO). Clinical Trials.gov Web site. http://clinicaltrials.gov/ct2/show/NCT00391872. Updated December 3, 2008. Accessed December 5, 2008.
- Greenbaum AB, Grines CL, Bittl JA, et al. Initial experience with an intravenous P2Y12 platelet receptor antagonist in patients undergoing percutaneous coronary intervention: results from a 2-part, phase II, multicenter, randomized, placebo- and active-controlled trial. Am Heart J 2006; 151:689.e1–689.e10.
- A clinical trial to demonstrate the efficacy of cangrelor (PCI). Clinical Trials.gov Web site. http://www.clinicaltrials.gov/ct/show/nct00305162. Updated December 3, 2008. Accessed December 5, 2008.
- Bhatt DL, Topol EJ. Scientific and therapeutic advances in antiplatelet therapy. Nat Rev Drug Discov 2003; 2:15–28.
- Moliterno DJ, Becker RC, Jennings LK, et al; TRA-PCI Study Investigators. Results of a multinational randomized, double-blind, placebo-controlled study of a novel thrombin receptor antagonist (SCH 530348) in percutaneous coronary intervention. Presented at: 56th Annual Scientific Session of the American College of Cardiology; March 24–27, 2007; New Orleans, LA.
- Trial to assess the effects of SCH 530348 in preventing heart attack and stroke in patients with atherosclerosis (TRA 2°P-TIMI 50). Clinical Trials.gov Web site. http://clinicaltrials.gov/ct2/show/NCT00526474. Updated November 13, 2008. Accessed December 16, 2008.
- Trial to assess the effects of SCH 530348 in preventing heart attack and stroke in patients with acute coronary syndrome (TRA-CER). Clinical Trials.gov Web site. http://clinicaltrials.gov/ct2/show/NCT00527943. Updated November 12, 2008. Accessed December 16, 2008.
- Wiviott SD, Trenk D, Frelinger AL, et al. Prasugrel compared with high loading- and maintenance-dose clopidogrel in patients with planned percutaneous coronary intervention: the Prasugrel in Comparison to Clopidogrel for Inhibition of Platelet Activation and Aggregation–Thrombolysis in Myocardial Infarction 44 trial. Circulation 2007; 116:2923–2932.
- Serebruany VL, Steinhubl SR, Berger PB, Malinin AI, Bhatt DL, Topol EJ. Variability in platelet responsiveness to clopidogrel among 544 individuals. J Am Coll Cardiol 2005; 45:246–251.
- Matetzky S, Shenkman B, Guetta V, et al. Clopidogrel resistance is associated with increased risk of recurrent atherothrombotic events in patients with acute myocardial infarction. Circulation 2004; 109:3171–3175.
- Gurbel PA, Bliden KP, Samara W, et al. Clopidogrel effect on platelet reactivity in patients with stent thrombosis: results of the CREST Study. J Am Coll Cardiol 2005; 46:1827–1832.
- Gurbel PA, Bliden KP, Hayes KM, Yoho JA, Herzog WR, Tantry US. The relation of dosing to clopidogrel responsiveness and the incidence of high post-treatment platelet aggregation in patients undergoing coronary stenting. J Am Coll Cardiol 2005; 45:1392–1396.
- von Beckerath N, Taubert D, Pogatsa-Murray G, Schömig E, Kastrati A, Schömig A. Absorption, metabolization, and antiplatelet effects of 300-, 600-, and 900-mg loading doses of clopidogrel: results of the ISAR-CHOICE (Intracoronary Stenting and Antithrombotic Regimen: Choose Between 3 High Oral Doses for Immediate Clopidogrel Effect) Trial. Circulation 2005; 112:2946–2950.
- Patti G, Colonna G, Pasceri V, Pepe LL, Montinaro A, Di Sciascio G. Randomized trial of high loading dose of clopidogrel for reduction of periprocedural myocardial infarction in patients undergoing coronary intervention: results from the ARMYDA-2 (Antiplatelet therapy for Reduction of MYocardial Damage during Angioplasty) study. Circulation 2005; 111:2099–2106.
- Cuisset T, Frere C, Quilici J, et al. Benefit of a 600-mg loading dose of clopidogrel on platelet reactivity and clinical outcomes in patients with non-ST-segment elevation acute coronary syndrome undergoing coronary stenting. J Am Coll Cardiol 2006; 48:1339–1345.
- von Beckerath N, Kastrati A, Wieczorek A, et al. A double-blind, randomized study on platelet aggregation in patients treated with a daily dose of 150 or 75 mg of clopidogrel for 30 days. Eur Heart J 2007; 28:1814–1819.
- Angiolillo DJ, Shoemaker SB, Desai B, et al. Randomized comparison of a high clopidogrel maintenance dose in patients with diabetes mellitus and coronary artery disease: results of the Optimizing Antiplatelet Therapy in Diabetes Mellitus (OPTIMUS) study. Circulation 2007; 115:708–716.
- Clopidogrel optimal loading dose usage to reduce recurrent events/optimal antiplatelet strategy for interventions (CURRENT/OASIS7). Clinical Trials.gov Web site. http://clinicaltrials.gov/ct2/show/NCT00335452. Updated September 1, 2008. Accessed December 16, 2008.
- Bonello L, Camoin-Jau L, Arques S, et al. Adjusted clopidogrel loading doses according to vasodilator-stimulated phosphoprotein phosphorylation index decrease rate of major adverse cardiovascular events in patients with clopidogrel resistance: a multicenter randomized prospective study. J Am Coll Cardiol 2008; 51:1404–1411.
- Brandt JT, Payne CD, Wiviott SD, et al. A comparison of prasugrel and clopidogrel loading doses on platelet function: magnitude of platelet inhibition is related to active metabolic formation. Am Heart J 2007; 153:66.e9–e16.
- Herbert JM, Savi P. P2Y12, a new platelet ADP receptor, target of clopidogrel. Semin Vasc Med 2003; 3:113–122.
- Wiviott SD, Braunwald E, McCabe CH, et al. Prasugrel versus clopidogrel in patients with acute coronary syndromes. N Engl J Med 2007; 357:2001–2015.
- Wiviott SD, Braunwald E, Angiolillo DJ, et al. Greater clinical benefit of more intensive oral antiplatelet therapy with prasugrel in patients with diabetes mellitus in the Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel–Thrombolysis in Myocardial Infarction 38. Circulation 2008; 118:1626–1636.
- Wiviott SD, Braunwald E, McCabe CH, et al. Intensive oral antiplatelet therapy for reduction of ischaemic events including stent thrombosis in patients with acute coronary syndromes treated with percutaneous coronary intervention and stenting in the TRITON-TIMI 38 trial: a subanalysis of a randomised trial. Lancet 2008; 371:1353–1363.
- Mauri L, Hsieh WH, Massaro JM, Ho KK, D’Agostino R, Cutlip DE. Stent thrombosis in randomized clinical trials of drug-eluting stents. N Engl J Med 2007; 356:1020–1029.
- Storey RF, Husted S, Harrington RA, et al. Inhibition of platelet aggregation by AZD6140, a reversible oral P2Y12 receptor antagonist, compared with clopidogrel in patients with acute coronary syndromes. J Am Coll Cardiol 2007; 50:1852–1856.
- Cannon CP, Husted S, Harrington RA, et al. Safety, tolerability, and initial efficacy of AZD6140, the first reversible oral adenosine diphosphate receptor antagonist, compared with clopidogrel, in patients with non-ST-segment elevation acute coronary syndrome: primary results of the DISPERSE-2 trial. J Am Coll Cardiol 2007; 50:1844–1851.
- A comparison of AZD6140 and clopidogrel in patients with acute coronary syndrome (PLATO). Clinical Trials.gov Web site. http://clinicaltrials.gov/ct2/show/NCT00391872. Updated December 3, 2008. Accessed December 5, 2008.
- Greenbaum AB, Grines CL, Bittl JA, et al. Initial experience with an intravenous P2Y12 platelet receptor antagonist in patients undergoing percutaneous coronary intervention: results from a 2-part, phase II, multicenter, randomized, placebo- and active-controlled trial. Am Heart J 2006; 151:689.e1–689.e10.
- A clinical trial to demonstrate the efficacy of cangrelor (PCI). Clinical Trials.gov Web site. http://www.clinicaltrials.gov/ct/show/nct00305162. Updated December 3, 2008. Accessed December 5, 2008.
- Bhatt DL, Topol EJ. Scientific and therapeutic advances in antiplatelet therapy. Nat Rev Drug Discov 2003; 2:15–28.
- Moliterno DJ, Becker RC, Jennings LK, et al; TRA-PCI Study Investigators. Results of a multinational randomized, double-blind, placebo-controlled study of a novel thrombin receptor antagonist (SCH 530348) in percutaneous coronary intervention. Presented at: 56th Annual Scientific Session of the American College of Cardiology; March 24–27, 2007; New Orleans, LA.
- Trial to assess the effects of SCH 530348 in preventing heart attack and stroke in patients with atherosclerosis (TRA 2°P-TIMI 50). Clinical Trials.gov Web site. http://clinicaltrials.gov/ct2/show/NCT00526474. Updated November 13, 2008. Accessed December 16, 2008.
- Trial to assess the effects of SCH 530348 in preventing heart attack and stroke in patients with acute coronary syndrome (TRA-CER). Clinical Trials.gov Web site. http://clinicaltrials.gov/ct2/show/NCT00527943. Updated November 12, 2008. Accessed December 16, 2008.
KEY POINTS
- There is substantial interpatient variability in the response to clopidogrel.
- In the large TRITON-TIMI 38 trial, the composite rate of death, myocardial infarction, or stroke was reduced by 19% and the rate of stent thrombosis was halved in patients receiving prasugrel compared with standard-dose clopidogrel.
- The risk of major bleeding with prasugrel is highest in patients aged 75 or older, those weighing less than 60 kg, and those with a history of stroke or transient ischemic attack.
- Thrombin receptor antagonists are being studied to see if their use can reduce ischemic events without increasing bleeding.