User login
Anemia and chronic kidney disease: What’s the connection?
• Evaluate for chronic kidney disease (CKD) anemia when a patient has a serum creatinine ≥2 mg/dL and hemoglobin <12 g/dL (adult males and postmenopausal females) or <11 g/dL (premenopausal females). A
• Before you treat CKD anemia, correct any underlying iron deficiency. A
• Start anemia therapy with erythropoietin-stimulating agents when hemoglobin is ≤10 g/dL, and maintain target hemoglobin levels between 11 and 12 g/dL, in accordance with National Kidney Foundation guidelines. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Mary J, a 65-year-old woman with stage 3 chronic kidney disease (CKD), is in your office for a follow-up appointment. Over the past 6 months, she has noticed a decrease in her energy level. On her routine blood work, you see that her hemoglobin has been slowly declining over the past year. It is now 9 g/dL and her estimated glomerular filtration rate (GFR) is 40 mL/min.
How would you evaluate Mary’s anemia, and would you suspect that it was related to her CKD?
Most physicians are aware that CKD—which affects approximately 10% of the US population1—has a deleterious effect on cardiovascular disease, but many fail to recognize the impact it has on the hematopoietic system. Managing the anemia that accompanies CKD in patients like Mary requires a finely tuned diagnostic approach and treatment strategy. This article will help toward that end.
Anemia of CKD: A common problem
Anemia of CKD is one of the first signs of kidney dysfunction, yet it often goes undetected because of its insidious onset. Anemia develops gradually as kidney function declines and the GFR drops to 70 mL/min in male patients and 50 mL/min in females.2 Epidemiologic data indicate that two-thirds of patients in the early stages of kidney failure are also anemic, with a hemoglobin level of less than 11 g/dL, yet only one-third of these patients have ever received erythropoietin-stimulating agents (ESAs) to treat their anemia.1 The National Kidney Foundation Kidney Disease Outcomes Quality Initiative (NKF KDOQI) guidelines recommend that the evaluation of anemia of CKD begin in patients with a serum creatinine ≥2 mg/dL when the hemoglobin is <12 g/dL in adult males and postmenopausal females and <11 g/dL in premenopausal females.3
How kidney failure leads to anemia
Patients like Mary develop anemia of CKD because failing kidneys produce less erythropoietin (EPO) than the body requires for the production of red blood cells. EPO is an endogenous hormone produced by peritubular fibroblasts in the renal cortex.4 Most of this hormone (90%) is produced in the kidney, with the remainder manufactured by hepatocytes.
Erythropoiesis is stimulated by blood loss, decreased oxygen tension, and an increase in oxygen affinity, which leads to an increase in EPO production via upregulation of the EPO gene. In healthy individuals, detection of hypoxia by the kidney can result in a 1000-fold increase in EPO production.5 Patients with CKD don’t have that kind of robust response, and their EPO levels remain normal or below normal even when challenged by lack of oxygen. Anemia in CKD can also be caused by nutritional deficiencies, decreased red blood cell survival because of uremic toxins, oxidative stress, inflammation, and the use of angiotensin-converting enzyme (ACE) inhibitors.
Chronic anemia, CKD, and CV disease: A deadly triad
The leading cause of death in patients with CKD is cardiovascular disease. Patients with cardiorenal anemia syndrome develop a self-perpetuating triad that increases the risk of death when all 3 conditions are present. Anemic patients double their relative risk of death when CKD is present and triple their risk if they have anemia, CKD, and cardiovascular disease.6
Epidemiologic studies suggest an association among anemia, left ventricular hypertrophy (LVH), mortality, and cardiovascular outcomes. One study evaluated 2423 stage 3 and 4 CKD patients with anemia, defined as hemoglobin <13 g/dL in males and <12 g/dL in females. The results showed an increase in composite outcomes of myocardial infarction, stroke, and death.7 A prospective study evaluating 246 people with stages 2 to 4 CKD reported anemia to be an independent risk factor for the development of LVH.8 The stages of CKD are shown in the TABLE.
Suspected mechanisms of cardiovascular disease progression due to chronic anemia include tissue hypoxia, free radical formation, endothelial dysfunction, and vascular damage. Compensatory neurohumeral adaptations result in an increased sympathetic response and upregulation of the reninangiotensin-aldosterone system.9
TABLE
Stages of chronic kidney disease
| Stage | Description | GFR (mL/min/1.73 m2) |
|---|---|---|
| 1 | Kidney damage with normal or increased GFR | ≥90 |
| 2 | Kidney damage with mildly decreased GFR | 60-89 |
| 3 | Moderately decreased GFR | 30-59 |
| 4 | Severely decreased GFR | 15-29 |
| 5 | Kidney failure | <15 or dialysis |
| GFR, glomerular filtration rate. | ||
| Source: KDOQI Clinical Practice Guideline and Clinical Practice Recommendations for anemia in chronic kidney disease: 2007 update of hemoglobin target. Am J Kidney Dis. 2007.3 | ||
Anemia of CKD: A diagnosis of exclusion
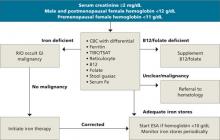
Because anemia can have many causes, other possibilities must be ruled out before a diagnosis of CKD anemia can be made. Testing should be tailored to each individual situation, determined by a thorough history and physical. Steps in the diagnosis are shown in the FLOW CHART. A basic work-up should include complete blood count with differential, iron studies (ferritin, serum Fe, and total iron binding capacity), reticulocyte count, and a guaiac test. Other blood tests, such as thyroid-stimulating hormone (TSH), B12, and folate levels, and a hemolysis panel (lactate dehydrogenase, haptoglobin), should be obtained if the history suggests these disorders. A peripheral blood smear showing normocytic red blood cells with a normochromic pattern would favor the diagnosis of anemia of CKD.
FLOW CHART
A step-by-step guide to CKD anemia diagnosis and treatment
CBC, complete blood count; CKD, chronic kidney disease; ESA, erythropoietin-stimulating agents; R/O, rule out; TIBC/TSAT, total iron-binding capacity/transferrin saturation.
A look at the iron connection
Many patients with CKD anemia have iron deficiency and are unable to produce adequate numbers of red blood cells. Iron deficiency can have many causes: not enough iron-rich food in the diet, chronic bleeding, malabsorption, or an occult gastrointestinal malignancy. Once iron deficiency anemia is diagnosed, a colonoscopy is warranted to rule out occult malignancy. Ferritin, a protein found mostly in macrophages and hepatocytes, stores iron and serves as a marker for total iron stores. Using stored iron requires transferrin, a transporting protein, to shuttle iron from the reticuloendothelial system and gut to the bone marrow. CKD is a pro-inflammatory state that results in a limited ability to use iron stores. For this reason, patients with CKD require higher levels of iron.
Absolute iron deficiency. Iron deficiency in CKD patients with serum ferritin <100 ng/mL and transferrin saturation (TSAT) <20% is characterized as absolute iron deficiency. The TSAT represents the percent of iron bound to transferrin and is a good indicator of the body’s functional capacity to use stored iron.
Relative iron deficiency and iron block. Patients who do not respond to ESA therapy even though they have adequate iron stores are said to have a functional or relative iron deficiency. Iron block is a condition that results in anemia from a chronic inflammatory state such as infection, autoimmune disorders, or malignancies. It resolves once the inflammatory process abates. Both conditions have similar anemia profiles, with a serum ferritin >100 ng/mL and a TSAT <20%. Differentiating between these conditions requires dynamic testing using serial iron studies and observing responses to ESAs and iron supplementation.
Options for correcting iron deficiency
After a thorough history and physical with appropriate screening, you find that Mary has an iron deficiency that must be corrected before her anemia can be treated effectively. Treatment for iron deficiency is usually initiated with oral therapy, at the recommended dose of 200 mg oral elemental iron a day in 3 divided doses.
If the oral therapy does not correct iron deficiency within 3 months, or a patient cannot tolerate the constipation that is often a side effect of this therapy, IV iron administration can be considered. Because CKD patients do not have the ongoing iron losses seen in patients with end-stage renal disease (ESRD), a conservative approach using a single IV dose followed by repeat testing is warranted. The goal is to achieve ferritin levels >100 ng/dL and TSAT >20%. A number of products for IV iron administration are available. The most widely used are iron dextran (INFeD), ferric gluconate (Ferrlecit), and iron sucrose (Venofer).
Iron stores are replenished? Time to treat the anemia
When ferritin levels and TSAT show that iron deficiency has been corrected, ESA treatment for anemia can begin. Two major brands of ESAs currently in use in the United States are a recombinant human erythropoietin (rHuEPO) known as epoetin alfa (Procrit, Epogen), and darbepoetin alpha (Aranesp). Both medications are effective and can be given intravenously or subcutaneously. Subcutaneous darbepoetin alpha has a longer half-life compared with epoetin alpha (70 vs 24 hours), so dosing intervals can be longer.10,11 ESAs should not be started in patients with uncontrolled hypertension until the blood pressure is controlled, or in patients with an active malignancy unless the treatment is directly supervised by an oncologist.
Aim for complete anemia resolution? That’s controversial
Treatment of CKD anemia with ESAs is widely practiced, but controversy over whether it is beneficial to aim for complete resolution of anemia is ongoing. The CREATE (Cardiovascular Risk Reduction by Early Anemia Treatment) and CHOIR (Correction of Hemoglobin and Outcomes in Renal Insufficiency) trials published in 2006 failed to resolve the issue.12,13
In the CREATE trial, patients targeted to achieve normal hemoglobin levels did no better in avoiding cardiovascular events than patients targeted for lower levels. The CHOIR trial was stopped early because of an increased trend toward death and hospitalization for congestive heart failure in the group with therapy targeted to achieve normal hemoglobin levels.
The recently published TREAT (Trial to Reduce Cardiovascular Events with Aranesp Therapy) study of patients with type 2 diabetes and CKD showed no reduction in all-cause mortality, cardiovascular morbidity, or ESRD in patients receiving Aranesp targeted to achieve a hemoglobin level of approximately 13 g/dL, compared with placebo.14 The study did demonstrate, however, that patients receiving Aranesp were about twice as likely to have a stroke than the placebo subjects (101 vs 53)—which might lead clinicians to ponder whether the gains, if any, were worth the risk.
Revised labeling. Late last year, the US Food and Drug Administration approved a label change for Procrit and Aranesp, warning that patients with renal failure “experienced greater risks for death and serious cardiovascular events when administered ESAs to target higher vs lower hemoglobin levels” and advising physicians to “individualize dosing to achieve and maintain hemoglobin levels within the range of 10 to 12 g/dL.”10,11 The 2007 NKF KDOQI guidelines suggest maintaining a hemoglobin level between 11 and 12 g/dL and have not incorporated the results of the TREAT trial.
Some patients don’t respond to ESAs
Inadequate response to ESAs is most commonly caused by underdosing or inadequate iron stores. NKF KDOQI guidelines recommend checking TSAT and ferritin prior to initiating therapy and monitoring these levels every 3 months.3 True nonresponders are individuals with good iron stores who are unable to achieve target hemoglobin within 4 to 6 months despite receiving subcutaneous epoetin 300 IU/kg per week. Inadequate response to ESAs can be caused by ongoing occult blood loss, infection, inflammation, nutritional deficiencies, hemolysis, hemoglobinemias, aluminum toxicity, anti-EPO antibody, hyperparathyroidism, multiple myeloma, and bone marrow dysfunction.10,11 If patients do not respond to ESA therapy, the NKF KDOQI guidelines recommend referral to a nephrologist or hematologist.3
How did Mary fare?
Mary did well taking oral iron supplementation. Once her iron deficiency was corrected, you were able to begin treating her anemia. After appropriate titration of her ESA, she was able to maintain a hemoglobin level between 11 and 12 g/dL 4 months into therapy. On a follow-up visit, she had no side effects from the medication and reported an increase in her energy level.
CORRESPONDENCE
Jonathan Taliercio, DO, Cleveland Clinic, Department of Nephrology and Hypertension, 9500 Euclid Avenue, Cleveland, OH 44195; talierj@ccf.org
1. United States Renal Data System, USRDS. 2009 Annual Data Report. Atlas of Chronic Kidney Disease in the United States. Bethesda, MD: National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases; 2009.
2. Hsu CJ, McCulloch CE, Curhan GC. Epidemiology of anemia associated with chronic renal insufficiency among adults in the United States: results from the Third National Health and Nutrition Examination Survey. J Am Soc Nephrol. 2002;13:504-510.
3. KDOQI Clinical Practice Guideline and Clinical Practice Recommendations for anemia in chronic kidney disease: 2007 update of hemoglobin target. Am J Kidney Dis. 2007;50:471-530.
4. Donnelly S. Why is erythropoietin made in the kidney? The kidney functions as a critmeter. Am J Kidney Dis. 2001;38:415-425.
5. Ebert B, Franklin H. Regulation of the erythropoietin gene. Blood. 1999;94:1864-1877.
6. Silverberg D, Wexler D, Blum M, et al. The cardio-renal anaemia syndrome: does it exist? Nephrol Dial Transplant. 2003;18(suppl 8):viii 7-viii 12.
7. Weiner D, Tighiouart H, Vlagopoulos P, et al. Effects of anemia and left ventricular hypertrophy on cardiovascular disease in patients with chronic kidney disease. J Am Soc Nephrol. 2005;16:1803-1810.
8. Levin A, Thompson C, Ethier J, et al. Left ventricular mass index increase in early renal disease: impact of decline in hemoglobin. Am J Kidney Dis. 1999;34:125-134.
9. Rao M, Pereira B. Optimal anemia management reduces cardiovascular morbidity, mortality, and costs in chronic kidney disease. Kidney Int. 2005;68:1432-1438.
10. Amgen. Aranesp (Darbepoetin Alpha) package insert. Available at www.aranesp.com/professional/crf/full_prescribing_info/pi.jsp. Accessed November 16, 2009.
11. Amgen. Procrit (Epoetin Alpha) package insert. Available at www.procrit.com/sites/default/files/shared/OBI/PI/ProcritBooklet.pdf#page=1. Accessed November 16, 2009.
12. Drueke T, Locatelli F, Clyne N, et al. Normalization of hemoglobin level in patients with CKD and anemia. N Engl J Med. 2006;355:2071-2084.
13. Singh A, Szczech L, Tang K. Correction of anemia with epoetin alfa in chronic kidney disease. N Engl J Med. 2006;355:2085-2098.
14. Pfeffer MA, Burdmann EA, Chen CY, et al. A trial of darbepoetin alfa in type 2 diabetes and chronic kidney disease. N Engl J Med. 2009;361:2019-2032.
• Evaluate for chronic kidney disease (CKD) anemia when a patient has a serum creatinine ≥2 mg/dL and hemoglobin <12 g/dL (adult males and postmenopausal females) or <11 g/dL (premenopausal females). A
• Before you treat CKD anemia, correct any underlying iron deficiency. A
• Start anemia therapy with erythropoietin-stimulating agents when hemoglobin is ≤10 g/dL, and maintain target hemoglobin levels between 11 and 12 g/dL, in accordance with National Kidney Foundation guidelines. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Mary J, a 65-year-old woman with stage 3 chronic kidney disease (CKD), is in your office for a follow-up appointment. Over the past 6 months, she has noticed a decrease in her energy level. On her routine blood work, you see that her hemoglobin has been slowly declining over the past year. It is now 9 g/dL and her estimated glomerular filtration rate (GFR) is 40 mL/min.
How would you evaluate Mary’s anemia, and would you suspect that it was related to her CKD?
Most physicians are aware that CKD—which affects approximately 10% of the US population1—has a deleterious effect on cardiovascular disease, but many fail to recognize the impact it has on the hematopoietic system. Managing the anemia that accompanies CKD in patients like Mary requires a finely tuned diagnostic approach and treatment strategy. This article will help toward that end.
Anemia of CKD: A common problem
Anemia of CKD is one of the first signs of kidney dysfunction, yet it often goes undetected because of its insidious onset. Anemia develops gradually as kidney function declines and the GFR drops to 70 mL/min in male patients and 50 mL/min in females.2 Epidemiologic data indicate that two-thirds of patients in the early stages of kidney failure are also anemic, with a hemoglobin level of less than 11 g/dL, yet only one-third of these patients have ever received erythropoietin-stimulating agents (ESAs) to treat their anemia.1 The National Kidney Foundation Kidney Disease Outcomes Quality Initiative (NKF KDOQI) guidelines recommend that the evaluation of anemia of CKD begin in patients with a serum creatinine ≥2 mg/dL when the hemoglobin is <12 g/dL in adult males and postmenopausal females and <11 g/dL in premenopausal females.3
How kidney failure leads to anemia
Patients like Mary develop anemia of CKD because failing kidneys produce less erythropoietin (EPO) than the body requires for the production of red blood cells. EPO is an endogenous hormone produced by peritubular fibroblasts in the renal cortex.4 Most of this hormone (90%) is produced in the kidney, with the remainder manufactured by hepatocytes.
Erythropoiesis is stimulated by blood loss, decreased oxygen tension, and an increase in oxygen affinity, which leads to an increase in EPO production via upregulation of the EPO gene. In healthy individuals, detection of hypoxia by the kidney can result in a 1000-fold increase in EPO production.5 Patients with CKD don’t have that kind of robust response, and their EPO levels remain normal or below normal even when challenged by lack of oxygen. Anemia in CKD can also be caused by nutritional deficiencies, decreased red blood cell survival because of uremic toxins, oxidative stress, inflammation, and the use of angiotensin-converting enzyme (ACE) inhibitors.
Chronic anemia, CKD, and CV disease: A deadly triad
The leading cause of death in patients with CKD is cardiovascular disease. Patients with cardiorenal anemia syndrome develop a self-perpetuating triad that increases the risk of death when all 3 conditions are present. Anemic patients double their relative risk of death when CKD is present and triple their risk if they have anemia, CKD, and cardiovascular disease.6
Epidemiologic studies suggest an association among anemia, left ventricular hypertrophy (LVH), mortality, and cardiovascular outcomes. One study evaluated 2423 stage 3 and 4 CKD patients with anemia, defined as hemoglobin <13 g/dL in males and <12 g/dL in females. The results showed an increase in composite outcomes of myocardial infarction, stroke, and death.7 A prospective study evaluating 246 people with stages 2 to 4 CKD reported anemia to be an independent risk factor for the development of LVH.8 The stages of CKD are shown in the TABLE.
Suspected mechanisms of cardiovascular disease progression due to chronic anemia include tissue hypoxia, free radical formation, endothelial dysfunction, and vascular damage. Compensatory neurohumeral adaptations result in an increased sympathetic response and upregulation of the reninangiotensin-aldosterone system.9
TABLE
Stages of chronic kidney disease
| Stage | Description | GFR (mL/min/1.73 m2) |
|---|---|---|
| 1 | Kidney damage with normal or increased GFR | ≥90 |
| 2 | Kidney damage with mildly decreased GFR | 60-89 |
| 3 | Moderately decreased GFR | 30-59 |
| 4 | Severely decreased GFR | 15-29 |
| 5 | Kidney failure | <15 or dialysis |
| GFR, glomerular filtration rate. | ||
| Source: KDOQI Clinical Practice Guideline and Clinical Practice Recommendations for anemia in chronic kidney disease: 2007 update of hemoglobin target. Am J Kidney Dis. 2007.3 | ||
Anemia of CKD: A diagnosis of exclusion
Because anemia can have many causes, other possibilities must be ruled out before a diagnosis of CKD anemia can be made. Testing should be tailored to each individual situation, determined by a thorough history and physical. Steps in the diagnosis are shown in the FLOW CHART. A basic work-up should include complete blood count with differential, iron studies (ferritin, serum Fe, and total iron binding capacity), reticulocyte count, and a guaiac test. Other blood tests, such as thyroid-stimulating hormone (TSH), B12, and folate levels, and a hemolysis panel (lactate dehydrogenase, haptoglobin), should be obtained if the history suggests these disorders. A peripheral blood smear showing normocytic red blood cells with a normochromic pattern would favor the diagnosis of anemia of CKD.
FLOW CHART
A step-by-step guide to CKD anemia diagnosis and treatment
CBC, complete blood count; CKD, chronic kidney disease; ESA, erythropoietin-stimulating agents; R/O, rule out; TIBC/TSAT, total iron-binding capacity/transferrin saturation.
A look at the iron connection
Many patients with CKD anemia have iron deficiency and are unable to produce adequate numbers of red blood cells. Iron deficiency can have many causes: not enough iron-rich food in the diet, chronic bleeding, malabsorption, or an occult gastrointestinal malignancy. Once iron deficiency anemia is diagnosed, a colonoscopy is warranted to rule out occult malignancy. Ferritin, a protein found mostly in macrophages and hepatocytes, stores iron and serves as a marker for total iron stores. Using stored iron requires transferrin, a transporting protein, to shuttle iron from the reticuloendothelial system and gut to the bone marrow. CKD is a pro-inflammatory state that results in a limited ability to use iron stores. For this reason, patients with CKD require higher levels of iron.
Absolute iron deficiency. Iron deficiency in CKD patients with serum ferritin <100 ng/mL and transferrin saturation (TSAT) <20% is characterized as absolute iron deficiency. The TSAT represents the percent of iron bound to transferrin and is a good indicator of the body’s functional capacity to use stored iron.
Relative iron deficiency and iron block. Patients who do not respond to ESA therapy even though they have adequate iron stores are said to have a functional or relative iron deficiency. Iron block is a condition that results in anemia from a chronic inflammatory state such as infection, autoimmune disorders, or malignancies. It resolves once the inflammatory process abates. Both conditions have similar anemia profiles, with a serum ferritin >100 ng/mL and a TSAT <20%. Differentiating between these conditions requires dynamic testing using serial iron studies and observing responses to ESAs and iron supplementation.
Options for correcting iron deficiency
After a thorough history and physical with appropriate screening, you find that Mary has an iron deficiency that must be corrected before her anemia can be treated effectively. Treatment for iron deficiency is usually initiated with oral therapy, at the recommended dose of 200 mg oral elemental iron a day in 3 divided doses.
If the oral therapy does not correct iron deficiency within 3 months, or a patient cannot tolerate the constipation that is often a side effect of this therapy, IV iron administration can be considered. Because CKD patients do not have the ongoing iron losses seen in patients with end-stage renal disease (ESRD), a conservative approach using a single IV dose followed by repeat testing is warranted. The goal is to achieve ferritin levels >100 ng/dL and TSAT >20%. A number of products for IV iron administration are available. The most widely used are iron dextran (INFeD), ferric gluconate (Ferrlecit), and iron sucrose (Venofer).
Iron stores are replenished? Time to treat the anemia
When ferritin levels and TSAT show that iron deficiency has been corrected, ESA treatment for anemia can begin. Two major brands of ESAs currently in use in the United States are a recombinant human erythropoietin (rHuEPO) known as epoetin alfa (Procrit, Epogen), and darbepoetin alpha (Aranesp). Both medications are effective and can be given intravenously or subcutaneously. Subcutaneous darbepoetin alpha has a longer half-life compared with epoetin alpha (70 vs 24 hours), so dosing intervals can be longer.10,11 ESAs should not be started in patients with uncontrolled hypertension until the blood pressure is controlled, or in patients with an active malignancy unless the treatment is directly supervised by an oncologist.
Aim for complete anemia resolution? That’s controversial
Treatment of CKD anemia with ESAs is widely practiced, but controversy over whether it is beneficial to aim for complete resolution of anemia is ongoing. The CREATE (Cardiovascular Risk Reduction by Early Anemia Treatment) and CHOIR (Correction of Hemoglobin and Outcomes in Renal Insufficiency) trials published in 2006 failed to resolve the issue.12,13
In the CREATE trial, patients targeted to achieve normal hemoglobin levels did no better in avoiding cardiovascular events than patients targeted for lower levels. The CHOIR trial was stopped early because of an increased trend toward death and hospitalization for congestive heart failure in the group with therapy targeted to achieve normal hemoglobin levels.
The recently published TREAT (Trial to Reduce Cardiovascular Events with Aranesp Therapy) study of patients with type 2 diabetes and CKD showed no reduction in all-cause mortality, cardiovascular morbidity, or ESRD in patients receiving Aranesp targeted to achieve a hemoglobin level of approximately 13 g/dL, compared with placebo.14 The study did demonstrate, however, that patients receiving Aranesp were about twice as likely to have a stroke than the placebo subjects (101 vs 53)—which might lead clinicians to ponder whether the gains, if any, were worth the risk.
Revised labeling. Late last year, the US Food and Drug Administration approved a label change for Procrit and Aranesp, warning that patients with renal failure “experienced greater risks for death and serious cardiovascular events when administered ESAs to target higher vs lower hemoglobin levels” and advising physicians to “individualize dosing to achieve and maintain hemoglobin levels within the range of 10 to 12 g/dL.”10,11 The 2007 NKF KDOQI guidelines suggest maintaining a hemoglobin level between 11 and 12 g/dL and have not incorporated the results of the TREAT trial.
Some patients don’t respond to ESAs
Inadequate response to ESAs is most commonly caused by underdosing or inadequate iron stores. NKF KDOQI guidelines recommend checking TSAT and ferritin prior to initiating therapy and monitoring these levels every 3 months.3 True nonresponders are individuals with good iron stores who are unable to achieve target hemoglobin within 4 to 6 months despite receiving subcutaneous epoetin 300 IU/kg per week. Inadequate response to ESAs can be caused by ongoing occult blood loss, infection, inflammation, nutritional deficiencies, hemolysis, hemoglobinemias, aluminum toxicity, anti-EPO antibody, hyperparathyroidism, multiple myeloma, and bone marrow dysfunction.10,11 If patients do not respond to ESA therapy, the NKF KDOQI guidelines recommend referral to a nephrologist or hematologist.3
How did Mary fare?
Mary did well taking oral iron supplementation. Once her iron deficiency was corrected, you were able to begin treating her anemia. After appropriate titration of her ESA, she was able to maintain a hemoglobin level between 11 and 12 g/dL 4 months into therapy. On a follow-up visit, she had no side effects from the medication and reported an increase in her energy level.
CORRESPONDENCE
Jonathan Taliercio, DO, Cleveland Clinic, Department of Nephrology and Hypertension, 9500 Euclid Avenue, Cleveland, OH 44195; talierj@ccf.org
• Evaluate for chronic kidney disease (CKD) anemia when a patient has a serum creatinine ≥2 mg/dL and hemoglobin <12 g/dL (adult males and postmenopausal females) or <11 g/dL (premenopausal females). A
• Before you treat CKD anemia, correct any underlying iron deficiency. A
• Start anemia therapy with erythropoietin-stimulating agents when hemoglobin is ≤10 g/dL, and maintain target hemoglobin levels between 11 and 12 g/dL, in accordance with National Kidney Foundation guidelines. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Mary J, a 65-year-old woman with stage 3 chronic kidney disease (CKD), is in your office for a follow-up appointment. Over the past 6 months, she has noticed a decrease in her energy level. On her routine blood work, you see that her hemoglobin has been slowly declining over the past year. It is now 9 g/dL and her estimated glomerular filtration rate (GFR) is 40 mL/min.
How would you evaluate Mary’s anemia, and would you suspect that it was related to her CKD?
Most physicians are aware that CKD—which affects approximately 10% of the US population1—has a deleterious effect on cardiovascular disease, but many fail to recognize the impact it has on the hematopoietic system. Managing the anemia that accompanies CKD in patients like Mary requires a finely tuned diagnostic approach and treatment strategy. This article will help toward that end.
Anemia of CKD: A common problem
Anemia of CKD is one of the first signs of kidney dysfunction, yet it often goes undetected because of its insidious onset. Anemia develops gradually as kidney function declines and the GFR drops to 70 mL/min in male patients and 50 mL/min in females.2 Epidemiologic data indicate that two-thirds of patients in the early stages of kidney failure are also anemic, with a hemoglobin level of less than 11 g/dL, yet only one-third of these patients have ever received erythropoietin-stimulating agents (ESAs) to treat their anemia.1 The National Kidney Foundation Kidney Disease Outcomes Quality Initiative (NKF KDOQI) guidelines recommend that the evaluation of anemia of CKD begin in patients with a serum creatinine ≥2 mg/dL when the hemoglobin is <12 g/dL in adult males and postmenopausal females and <11 g/dL in premenopausal females.3
How kidney failure leads to anemia
Patients like Mary develop anemia of CKD because failing kidneys produce less erythropoietin (EPO) than the body requires for the production of red blood cells. EPO is an endogenous hormone produced by peritubular fibroblasts in the renal cortex.4 Most of this hormone (90%) is produced in the kidney, with the remainder manufactured by hepatocytes.
Erythropoiesis is stimulated by blood loss, decreased oxygen tension, and an increase in oxygen affinity, which leads to an increase in EPO production via upregulation of the EPO gene. In healthy individuals, detection of hypoxia by the kidney can result in a 1000-fold increase in EPO production.5 Patients with CKD don’t have that kind of robust response, and their EPO levels remain normal or below normal even when challenged by lack of oxygen. Anemia in CKD can also be caused by nutritional deficiencies, decreased red blood cell survival because of uremic toxins, oxidative stress, inflammation, and the use of angiotensin-converting enzyme (ACE) inhibitors.
Chronic anemia, CKD, and CV disease: A deadly triad
The leading cause of death in patients with CKD is cardiovascular disease. Patients with cardiorenal anemia syndrome develop a self-perpetuating triad that increases the risk of death when all 3 conditions are present. Anemic patients double their relative risk of death when CKD is present and triple their risk if they have anemia, CKD, and cardiovascular disease.6
Epidemiologic studies suggest an association among anemia, left ventricular hypertrophy (LVH), mortality, and cardiovascular outcomes. One study evaluated 2423 stage 3 and 4 CKD patients with anemia, defined as hemoglobin <13 g/dL in males and <12 g/dL in females. The results showed an increase in composite outcomes of myocardial infarction, stroke, and death.7 A prospective study evaluating 246 people with stages 2 to 4 CKD reported anemia to be an independent risk factor for the development of LVH.8 The stages of CKD are shown in the TABLE.
Suspected mechanisms of cardiovascular disease progression due to chronic anemia include tissue hypoxia, free radical formation, endothelial dysfunction, and vascular damage. Compensatory neurohumeral adaptations result in an increased sympathetic response and upregulation of the reninangiotensin-aldosterone system.9
TABLE
Stages of chronic kidney disease
| Stage | Description | GFR (mL/min/1.73 m2) |
|---|---|---|
| 1 | Kidney damage with normal or increased GFR | ≥90 |
| 2 | Kidney damage with mildly decreased GFR | 60-89 |
| 3 | Moderately decreased GFR | 30-59 |
| 4 | Severely decreased GFR | 15-29 |
| 5 | Kidney failure | <15 or dialysis |
| GFR, glomerular filtration rate. | ||
| Source: KDOQI Clinical Practice Guideline and Clinical Practice Recommendations for anemia in chronic kidney disease: 2007 update of hemoglobin target. Am J Kidney Dis. 2007.3 | ||
Anemia of CKD: A diagnosis of exclusion
Because anemia can have many causes, other possibilities must be ruled out before a diagnosis of CKD anemia can be made. Testing should be tailored to each individual situation, determined by a thorough history and physical. Steps in the diagnosis are shown in the FLOW CHART. A basic work-up should include complete blood count with differential, iron studies (ferritin, serum Fe, and total iron binding capacity), reticulocyte count, and a guaiac test. Other blood tests, such as thyroid-stimulating hormone (TSH), B12, and folate levels, and a hemolysis panel (lactate dehydrogenase, haptoglobin), should be obtained if the history suggests these disorders. A peripheral blood smear showing normocytic red blood cells with a normochromic pattern would favor the diagnosis of anemia of CKD.
FLOW CHART
A step-by-step guide to CKD anemia diagnosis and treatment
CBC, complete blood count; CKD, chronic kidney disease; ESA, erythropoietin-stimulating agents; R/O, rule out; TIBC/TSAT, total iron-binding capacity/transferrin saturation.
A look at the iron connection
Many patients with CKD anemia have iron deficiency and are unable to produce adequate numbers of red blood cells. Iron deficiency can have many causes: not enough iron-rich food in the diet, chronic bleeding, malabsorption, or an occult gastrointestinal malignancy. Once iron deficiency anemia is diagnosed, a colonoscopy is warranted to rule out occult malignancy. Ferritin, a protein found mostly in macrophages and hepatocytes, stores iron and serves as a marker for total iron stores. Using stored iron requires transferrin, a transporting protein, to shuttle iron from the reticuloendothelial system and gut to the bone marrow. CKD is a pro-inflammatory state that results in a limited ability to use iron stores. For this reason, patients with CKD require higher levels of iron.
Absolute iron deficiency. Iron deficiency in CKD patients with serum ferritin <100 ng/mL and transferrin saturation (TSAT) <20% is characterized as absolute iron deficiency. The TSAT represents the percent of iron bound to transferrin and is a good indicator of the body’s functional capacity to use stored iron.
Relative iron deficiency and iron block. Patients who do not respond to ESA therapy even though they have adequate iron stores are said to have a functional or relative iron deficiency. Iron block is a condition that results in anemia from a chronic inflammatory state such as infection, autoimmune disorders, or malignancies. It resolves once the inflammatory process abates. Both conditions have similar anemia profiles, with a serum ferritin >100 ng/mL and a TSAT <20%. Differentiating between these conditions requires dynamic testing using serial iron studies and observing responses to ESAs and iron supplementation.
Options for correcting iron deficiency
After a thorough history and physical with appropriate screening, you find that Mary has an iron deficiency that must be corrected before her anemia can be treated effectively. Treatment for iron deficiency is usually initiated with oral therapy, at the recommended dose of 200 mg oral elemental iron a day in 3 divided doses.
If the oral therapy does not correct iron deficiency within 3 months, or a patient cannot tolerate the constipation that is often a side effect of this therapy, IV iron administration can be considered. Because CKD patients do not have the ongoing iron losses seen in patients with end-stage renal disease (ESRD), a conservative approach using a single IV dose followed by repeat testing is warranted. The goal is to achieve ferritin levels >100 ng/dL and TSAT >20%. A number of products for IV iron administration are available. The most widely used are iron dextran (INFeD), ferric gluconate (Ferrlecit), and iron sucrose (Venofer).
Iron stores are replenished? Time to treat the anemia
When ferritin levels and TSAT show that iron deficiency has been corrected, ESA treatment for anemia can begin. Two major brands of ESAs currently in use in the United States are a recombinant human erythropoietin (rHuEPO) known as epoetin alfa (Procrit, Epogen), and darbepoetin alpha (Aranesp). Both medications are effective and can be given intravenously or subcutaneously. Subcutaneous darbepoetin alpha has a longer half-life compared with epoetin alpha (70 vs 24 hours), so dosing intervals can be longer.10,11 ESAs should not be started in patients with uncontrolled hypertension until the blood pressure is controlled, or in patients with an active malignancy unless the treatment is directly supervised by an oncologist.
Aim for complete anemia resolution? That’s controversial
Treatment of CKD anemia with ESAs is widely practiced, but controversy over whether it is beneficial to aim for complete resolution of anemia is ongoing. The CREATE (Cardiovascular Risk Reduction by Early Anemia Treatment) and CHOIR (Correction of Hemoglobin and Outcomes in Renal Insufficiency) trials published in 2006 failed to resolve the issue.12,13
In the CREATE trial, patients targeted to achieve normal hemoglobin levels did no better in avoiding cardiovascular events than patients targeted for lower levels. The CHOIR trial was stopped early because of an increased trend toward death and hospitalization for congestive heart failure in the group with therapy targeted to achieve normal hemoglobin levels.
The recently published TREAT (Trial to Reduce Cardiovascular Events with Aranesp Therapy) study of patients with type 2 diabetes and CKD showed no reduction in all-cause mortality, cardiovascular morbidity, or ESRD in patients receiving Aranesp targeted to achieve a hemoglobin level of approximately 13 g/dL, compared with placebo.14 The study did demonstrate, however, that patients receiving Aranesp were about twice as likely to have a stroke than the placebo subjects (101 vs 53)—which might lead clinicians to ponder whether the gains, if any, were worth the risk.
Revised labeling. Late last year, the US Food and Drug Administration approved a label change for Procrit and Aranesp, warning that patients with renal failure “experienced greater risks for death and serious cardiovascular events when administered ESAs to target higher vs lower hemoglobin levels” and advising physicians to “individualize dosing to achieve and maintain hemoglobin levels within the range of 10 to 12 g/dL.”10,11 The 2007 NKF KDOQI guidelines suggest maintaining a hemoglobin level between 11 and 12 g/dL and have not incorporated the results of the TREAT trial.
Some patients don’t respond to ESAs
Inadequate response to ESAs is most commonly caused by underdosing or inadequate iron stores. NKF KDOQI guidelines recommend checking TSAT and ferritin prior to initiating therapy and monitoring these levels every 3 months.3 True nonresponders are individuals with good iron stores who are unable to achieve target hemoglobin within 4 to 6 months despite receiving subcutaneous epoetin 300 IU/kg per week. Inadequate response to ESAs can be caused by ongoing occult blood loss, infection, inflammation, nutritional deficiencies, hemolysis, hemoglobinemias, aluminum toxicity, anti-EPO antibody, hyperparathyroidism, multiple myeloma, and bone marrow dysfunction.10,11 If patients do not respond to ESA therapy, the NKF KDOQI guidelines recommend referral to a nephrologist or hematologist.3
How did Mary fare?
Mary did well taking oral iron supplementation. Once her iron deficiency was corrected, you were able to begin treating her anemia. After appropriate titration of her ESA, she was able to maintain a hemoglobin level between 11 and 12 g/dL 4 months into therapy. On a follow-up visit, she had no side effects from the medication and reported an increase in her energy level.
CORRESPONDENCE
Jonathan Taliercio, DO, Cleveland Clinic, Department of Nephrology and Hypertension, 9500 Euclid Avenue, Cleveland, OH 44195; talierj@ccf.org
1. United States Renal Data System, USRDS. 2009 Annual Data Report. Atlas of Chronic Kidney Disease in the United States. Bethesda, MD: National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases; 2009.
2. Hsu CJ, McCulloch CE, Curhan GC. Epidemiology of anemia associated with chronic renal insufficiency among adults in the United States: results from the Third National Health and Nutrition Examination Survey. J Am Soc Nephrol. 2002;13:504-510.
3. KDOQI Clinical Practice Guideline and Clinical Practice Recommendations for anemia in chronic kidney disease: 2007 update of hemoglobin target. Am J Kidney Dis. 2007;50:471-530.
4. Donnelly S. Why is erythropoietin made in the kidney? The kidney functions as a critmeter. Am J Kidney Dis. 2001;38:415-425.
5. Ebert B, Franklin H. Regulation of the erythropoietin gene. Blood. 1999;94:1864-1877.
6. Silverberg D, Wexler D, Blum M, et al. The cardio-renal anaemia syndrome: does it exist? Nephrol Dial Transplant. 2003;18(suppl 8):viii 7-viii 12.
7. Weiner D, Tighiouart H, Vlagopoulos P, et al. Effects of anemia and left ventricular hypertrophy on cardiovascular disease in patients with chronic kidney disease. J Am Soc Nephrol. 2005;16:1803-1810.
8. Levin A, Thompson C, Ethier J, et al. Left ventricular mass index increase in early renal disease: impact of decline in hemoglobin. Am J Kidney Dis. 1999;34:125-134.
9. Rao M, Pereira B. Optimal anemia management reduces cardiovascular morbidity, mortality, and costs in chronic kidney disease. Kidney Int. 2005;68:1432-1438.
10. Amgen. Aranesp (Darbepoetin Alpha) package insert. Available at www.aranesp.com/professional/crf/full_prescribing_info/pi.jsp. Accessed November 16, 2009.
11. Amgen. Procrit (Epoetin Alpha) package insert. Available at www.procrit.com/sites/default/files/shared/OBI/PI/ProcritBooklet.pdf#page=1. Accessed November 16, 2009.
12. Drueke T, Locatelli F, Clyne N, et al. Normalization of hemoglobin level in patients with CKD and anemia. N Engl J Med. 2006;355:2071-2084.
13. Singh A, Szczech L, Tang K. Correction of anemia with epoetin alfa in chronic kidney disease. N Engl J Med. 2006;355:2085-2098.
14. Pfeffer MA, Burdmann EA, Chen CY, et al. A trial of darbepoetin alfa in type 2 diabetes and chronic kidney disease. N Engl J Med. 2009;361:2019-2032.
1. United States Renal Data System, USRDS. 2009 Annual Data Report. Atlas of Chronic Kidney Disease in the United States. Bethesda, MD: National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases; 2009.
2. Hsu CJ, McCulloch CE, Curhan GC. Epidemiology of anemia associated with chronic renal insufficiency among adults in the United States: results from the Third National Health and Nutrition Examination Survey. J Am Soc Nephrol. 2002;13:504-510.
3. KDOQI Clinical Practice Guideline and Clinical Practice Recommendations for anemia in chronic kidney disease: 2007 update of hemoglobin target. Am J Kidney Dis. 2007;50:471-530.
4. Donnelly S. Why is erythropoietin made in the kidney? The kidney functions as a critmeter. Am J Kidney Dis. 2001;38:415-425.
5. Ebert B, Franklin H. Regulation of the erythropoietin gene. Blood. 1999;94:1864-1877.
6. Silverberg D, Wexler D, Blum M, et al. The cardio-renal anaemia syndrome: does it exist? Nephrol Dial Transplant. 2003;18(suppl 8):viii 7-viii 12.
7. Weiner D, Tighiouart H, Vlagopoulos P, et al. Effects of anemia and left ventricular hypertrophy on cardiovascular disease in patients with chronic kidney disease. J Am Soc Nephrol. 2005;16:1803-1810.
8. Levin A, Thompson C, Ethier J, et al. Left ventricular mass index increase in early renal disease: impact of decline in hemoglobin. Am J Kidney Dis. 1999;34:125-134.
9. Rao M, Pereira B. Optimal anemia management reduces cardiovascular morbidity, mortality, and costs in chronic kidney disease. Kidney Int. 2005;68:1432-1438.
10. Amgen. Aranesp (Darbepoetin Alpha) package insert. Available at www.aranesp.com/professional/crf/full_prescribing_info/pi.jsp. Accessed November 16, 2009.
11. Amgen. Procrit (Epoetin Alpha) package insert. Available at www.procrit.com/sites/default/files/shared/OBI/PI/ProcritBooklet.pdf#page=1. Accessed November 16, 2009.
12. Drueke T, Locatelli F, Clyne N, et al. Normalization of hemoglobin level in patients with CKD and anemia. N Engl J Med. 2006;355:2071-2084.
13. Singh A, Szczech L, Tang K. Correction of anemia with epoetin alfa in chronic kidney disease. N Engl J Med. 2006;355:2085-2098.
14. Pfeffer MA, Burdmann EA, Chen CY, et al. A trial of darbepoetin alfa in type 2 diabetes and chronic kidney disease. N Engl J Med. 2009;361:2019-2032.
The Child With Familial Hyperlipidemia
Start with the most important thing—education of the child and the family. This condition is familial, so relatives are more likely to have this form of hyperlipidemia as well.
Ask families about relevant history of early heart disease. “Early familial heart disease” is defined as a father or grandfather younger than age 55 years and/or mother or grandmother younger than 65 years with known heart disease.
We recommend screening all children by the age of 2 years for relevant family history. Studies now indicate lipid deposits can start as early as this age.
Clinical intervention often is more about prevention than treatment. Unless children are homozygous for one of the genetic defects associated with familial hyperlipidemia, they may not have signs or symptoms until they reach their twenties or thirties.
It is appropriate for you to begin lifestyle recommendations with any overweight or obese child. Counsel the patient and family about better diet and exercise regimens. For example, instruct them to avoid fried foods and if they need to cook with oil, to use vegetable oil.
Recommend 60 minutes of moderate exercise daily. This does not have to be an hour all at once—it can be 20 minutes in the morning before the school bus comes, 20 minutes in the afternoon, and another 20 minutes in the evening. The physical activity does not have to be on the soccer field either. The patient can exercise by climbing the stairs or participating in a scavenger hunt at the mall.
The essential thing is getting the child off the couch and away from the computer. This is particularly important because many schools are cutting their physical education programs in this economy.
Emphasize to parents that familial hyperlipidemia is one of the preventable forms of heart disease. Parents have a choice if they want their children to lead long, healthy lives.
Monitor the child's growth. If the child exceeds the 95th percentile on the growth chart, draw cholesterol levels. If the numbers are high, initiate at least a 6-month trial of diet and exercise. If, after this time, the cholesterol levels remain high, consider prescribing a low-dose statin. If medication fails to reduce high cholesterol after 2 months, I recommend these children see a subspecialist like myself.
For the most part, they come to me obese and/or with high cholesterol. I lecture them like you cannot believe, and their weight and cholesterol numbers improve. For this reason, I have very few patients for whom I have to start medication.
The cholesterol assay you do has to be a fasting lipid profile, not a random cholesterol reading. A random test does not provide the most appropriate information. Use common sense regarding when to test kids. In other words, do not test cholesterol levels the day after their birthday, right after Halloween, or anytime between Thanksgiving and Christmas. Testing cholesterol at any time during spring and summer, if possible, is preferable.
You don't need to refer most children with familial hyperlipidemia for cardiac stress testing. Stress testing is generally reserved for treatment-refractory patients with established high cholesterol. This provides useful baseline information for children we cannot control well.
Start with the most important thing—education of the child and the family. This condition is familial, so relatives are more likely to have this form of hyperlipidemia as well.
Ask families about relevant history of early heart disease. “Early familial heart disease” is defined as a father or grandfather younger than age 55 years and/or mother or grandmother younger than 65 years with known heart disease.
We recommend screening all children by the age of 2 years for relevant family history. Studies now indicate lipid deposits can start as early as this age.
Clinical intervention often is more about prevention than treatment. Unless children are homozygous for one of the genetic defects associated with familial hyperlipidemia, they may not have signs or symptoms until they reach their twenties or thirties.
It is appropriate for you to begin lifestyle recommendations with any overweight or obese child. Counsel the patient and family about better diet and exercise regimens. For example, instruct them to avoid fried foods and if they need to cook with oil, to use vegetable oil.
Recommend 60 minutes of moderate exercise daily. This does not have to be an hour all at once—it can be 20 minutes in the morning before the school bus comes, 20 minutes in the afternoon, and another 20 minutes in the evening. The physical activity does not have to be on the soccer field either. The patient can exercise by climbing the stairs or participating in a scavenger hunt at the mall.
The essential thing is getting the child off the couch and away from the computer. This is particularly important because many schools are cutting their physical education programs in this economy.
Emphasize to parents that familial hyperlipidemia is one of the preventable forms of heart disease. Parents have a choice if they want their children to lead long, healthy lives.
Monitor the child's growth. If the child exceeds the 95th percentile on the growth chart, draw cholesterol levels. If the numbers are high, initiate at least a 6-month trial of diet and exercise. If, after this time, the cholesterol levels remain high, consider prescribing a low-dose statin. If medication fails to reduce high cholesterol after 2 months, I recommend these children see a subspecialist like myself.
For the most part, they come to me obese and/or with high cholesterol. I lecture them like you cannot believe, and their weight and cholesterol numbers improve. For this reason, I have very few patients for whom I have to start medication.
The cholesterol assay you do has to be a fasting lipid profile, not a random cholesterol reading. A random test does not provide the most appropriate information. Use common sense regarding when to test kids. In other words, do not test cholesterol levels the day after their birthday, right after Halloween, or anytime between Thanksgiving and Christmas. Testing cholesterol at any time during spring and summer, if possible, is preferable.
You don't need to refer most children with familial hyperlipidemia for cardiac stress testing. Stress testing is generally reserved for treatment-refractory patients with established high cholesterol. This provides useful baseline information for children we cannot control well.
Start with the most important thing—education of the child and the family. This condition is familial, so relatives are more likely to have this form of hyperlipidemia as well.
Ask families about relevant history of early heart disease. “Early familial heart disease” is defined as a father or grandfather younger than age 55 years and/or mother or grandmother younger than 65 years with known heart disease.
We recommend screening all children by the age of 2 years for relevant family history. Studies now indicate lipid deposits can start as early as this age.
Clinical intervention often is more about prevention than treatment. Unless children are homozygous for one of the genetic defects associated with familial hyperlipidemia, they may not have signs or symptoms until they reach their twenties or thirties.
It is appropriate for you to begin lifestyle recommendations with any overweight or obese child. Counsel the patient and family about better diet and exercise regimens. For example, instruct them to avoid fried foods and if they need to cook with oil, to use vegetable oil.
Recommend 60 minutes of moderate exercise daily. This does not have to be an hour all at once—it can be 20 minutes in the morning before the school bus comes, 20 minutes in the afternoon, and another 20 minutes in the evening. The physical activity does not have to be on the soccer field either. The patient can exercise by climbing the stairs or participating in a scavenger hunt at the mall.
The essential thing is getting the child off the couch and away from the computer. This is particularly important because many schools are cutting their physical education programs in this economy.
Emphasize to parents that familial hyperlipidemia is one of the preventable forms of heart disease. Parents have a choice if they want their children to lead long, healthy lives.
Monitor the child's growth. If the child exceeds the 95th percentile on the growth chart, draw cholesterol levels. If the numbers are high, initiate at least a 6-month trial of diet and exercise. If, after this time, the cholesterol levels remain high, consider prescribing a low-dose statin. If medication fails to reduce high cholesterol after 2 months, I recommend these children see a subspecialist like myself.
For the most part, they come to me obese and/or with high cholesterol. I lecture them like you cannot believe, and their weight and cholesterol numbers improve. For this reason, I have very few patients for whom I have to start medication.
The cholesterol assay you do has to be a fasting lipid profile, not a random cholesterol reading. A random test does not provide the most appropriate information. Use common sense regarding when to test kids. In other words, do not test cholesterol levels the day after their birthday, right after Halloween, or anytime between Thanksgiving and Christmas. Testing cholesterol at any time during spring and summer, if possible, is preferable.
You don't need to refer most children with familial hyperlipidemia for cardiac stress testing. Stress testing is generally reserved for treatment-refractory patients with established high cholesterol. This provides useful baseline information for children we cannot control well.
Managing Acid-Related Disorders Through the Ages of Mankind
A supplement to Internal Medicine News supported by a restricted grant from TAP Pharmaceutical Products, Inc.
Symposium Highlights of articles based on presentations given at a continuing medical education symposium held on October 22, 2002, in Seattle, Wash.

To view the supplement, click the image above.
A supplement to Internal Medicine News supported by a restricted grant from TAP Pharmaceutical Products, Inc.
Symposium Highlights of articles based on presentations given at a continuing medical education symposium held on October 22, 2002, in Seattle, Wash.

To view the supplement, click the image above.
A supplement to Internal Medicine News supported by a restricted grant from TAP Pharmaceutical Products, Inc.
Symposium Highlights of articles based on presentations given at a continuing medical education symposium held on October 22, 2002, in Seattle, Wash.

To view the supplement, click the image above.
Increased Elderly Care Costs Linked to Length of Stay
The latest wave of data confirming that end-of-life care is an outsized driver of healthcare costs comes with one caveat from the chair of SHM’s research committee.
“It’s not always obvious you’re providing end-of-life care when you’re providing it,” says David Meltzer, MD, PhD, FHM, FACP, chief of the section of hospital medicine and associate professor in the Department of Medicine and the Graduate School of Public Policy Studies at the University of Chicago. “You have to be really careful not to conflate age and proximity to death.”
Dr. Meltzer was one of the lead investigators who helped develop the university’s Curriculum for the Hospitalized Aging Medical Patient (CHAMP) and has studied the costs of healthcare delivery to the elderly. He cautions hospitalists against couching cost calculations in terms of the age of their patient census—and reviewing instead what would improve a patient’s quality of life.
“There are times when it’s appropriate to discuss the goals of care,” Dr. Meltzer says. “I doubt the answer to that question is defined by age.”
Accordingly, a statistical brief released last month by the federal Agency of Healthcare Research and Quality (AHRQ) focused on cost drivers, not age. The data show the inpatient death rate in 2007 was 1.9%. “However, these hospital stays ending in death were responsible for 5.2% ($20 billion) of all hospital inpatient costs,” the brief concluded. In what is probably no surprise to hospitalists, much of that cost is traced to length of stay, which averaged 8.8 days for patients who died and 4.5 days for those who lived. The data is for 2007, the latest year available.
To counter rising costs, Dr. Meltzer recommends:
- Review a patient’s condition holistically, looking past age;
- Have an upfront discussion with the patient about what their expectations of care are. Avoid excess care that is not in line with the patient’s wishes; and
- Talk with hospital administration to ensure that your HM group has a say in care decisions, effectively giving hospitalists an opportunity to act as a change agent.
“It’s not so much measuring against cost; it’s measuring against the patient’s preferences,” Dr. Meltzer says. “Ultimately, the physician is the patient’s agent.”
The latest wave of data confirming that end-of-life care is an outsized driver of healthcare costs comes with one caveat from the chair of SHM’s research committee.
“It’s not always obvious you’re providing end-of-life care when you’re providing it,” says David Meltzer, MD, PhD, FHM, FACP, chief of the section of hospital medicine and associate professor in the Department of Medicine and the Graduate School of Public Policy Studies at the University of Chicago. “You have to be really careful not to conflate age and proximity to death.”
Dr. Meltzer was one of the lead investigators who helped develop the university’s Curriculum for the Hospitalized Aging Medical Patient (CHAMP) and has studied the costs of healthcare delivery to the elderly. He cautions hospitalists against couching cost calculations in terms of the age of their patient census—and reviewing instead what would improve a patient’s quality of life.
“There are times when it’s appropriate to discuss the goals of care,” Dr. Meltzer says. “I doubt the answer to that question is defined by age.”
Accordingly, a statistical brief released last month by the federal Agency of Healthcare Research and Quality (AHRQ) focused on cost drivers, not age. The data show the inpatient death rate in 2007 was 1.9%. “However, these hospital stays ending in death were responsible for 5.2% ($20 billion) of all hospital inpatient costs,” the brief concluded. In what is probably no surprise to hospitalists, much of that cost is traced to length of stay, which averaged 8.8 days for patients who died and 4.5 days for those who lived. The data is for 2007, the latest year available.
To counter rising costs, Dr. Meltzer recommends:
- Review a patient’s condition holistically, looking past age;
- Have an upfront discussion with the patient about what their expectations of care are. Avoid excess care that is not in line with the patient’s wishes; and
- Talk with hospital administration to ensure that your HM group has a say in care decisions, effectively giving hospitalists an opportunity to act as a change agent.
“It’s not so much measuring against cost; it’s measuring against the patient’s preferences,” Dr. Meltzer says. “Ultimately, the physician is the patient’s agent.”
The latest wave of data confirming that end-of-life care is an outsized driver of healthcare costs comes with one caveat from the chair of SHM’s research committee.
“It’s not always obvious you’re providing end-of-life care when you’re providing it,” says David Meltzer, MD, PhD, FHM, FACP, chief of the section of hospital medicine and associate professor in the Department of Medicine and the Graduate School of Public Policy Studies at the University of Chicago. “You have to be really careful not to conflate age and proximity to death.”
Dr. Meltzer was one of the lead investigators who helped develop the university’s Curriculum for the Hospitalized Aging Medical Patient (CHAMP) and has studied the costs of healthcare delivery to the elderly. He cautions hospitalists against couching cost calculations in terms of the age of their patient census—and reviewing instead what would improve a patient’s quality of life.
“There are times when it’s appropriate to discuss the goals of care,” Dr. Meltzer says. “I doubt the answer to that question is defined by age.”
Accordingly, a statistical brief released last month by the federal Agency of Healthcare Research and Quality (AHRQ) focused on cost drivers, not age. The data show the inpatient death rate in 2007 was 1.9%. “However, these hospital stays ending in death were responsible for 5.2% ($20 billion) of all hospital inpatient costs,” the brief concluded. In what is probably no surprise to hospitalists, much of that cost is traced to length of stay, which averaged 8.8 days for patients who died and 4.5 days for those who lived. The data is for 2007, the latest year available.
To counter rising costs, Dr. Meltzer recommends:
- Review a patient’s condition holistically, looking past age;
- Have an upfront discussion with the patient about what their expectations of care are. Avoid excess care that is not in line with the patient’s wishes; and
- Talk with hospital administration to ensure that your HM group has a say in care decisions, effectively giving hospitalists an opportunity to act as a change agent.
“It’s not so much measuring against cost; it’s measuring against the patient’s preferences,” Dr. Meltzer says. “Ultimately, the physician is the patient’s agent.”
In the Literature: The Latest Research You Need to Know
Clinical question: Does the new antiarrhythmic agent dronedarone, an amiodarone derivative, reduce the incidence of stroke in patients with persistent or paroxysmal atrial fibrillation and at least one risk factor for vascular events?
Background: Antiarrhythmic therapy has not been shown to reduce stroke risk in the treatment of chronic atrial fibrillation. A sub-group analysis of a recent multicentered, international, industry-sponsored, randomized trial of the new antiarrhythmic dronedarone, suggested a potential reduction in stroke risk when compared with placebo.
Study design: Post-hoc analysis of a previous prospective, randomized, placebo-controlled, double-blinded, parallel-arm trial.
Setting: 551 centers in 37 countries.
Synopsis: In addition to anticoagulation and rate control, 4,628 patients with atrial fibrillation and at least one risk factor for cardiovascular (CV) hospitalization were randomized to dronedarone versus placebo. The study assessed the primary outcomes of stroke, acute coronary syndromes, CV, and total mortality.
The dronedarone arm experienced a 0.6% absolute risk reduction (ARR) of stroke {(number-needed-to-treat (NNT) 167); relative risk reduction (RRR) 33%; P=0.027}; 0.7% ARR of “stroke or transient ischemic attack” (NNT 143; RRR 30%; P=0.031); 1.7% ARR of “stroke, ACS, or CV death,” (NNT 59; RRR 31%; P<0.001); and a 1.6% ARR of “stroke, ACS, or all-cause mortality” (NNT 63; RRR 24%; P=0.002). In further subgroup analysis, only the 65% of patients had CHADS2 scores =2, benefited (RR stroke 0.50; 95% CI, 0.32-0.79).
The results are thought-provoking, and suggest there could be more than antiarrhythmic effects of dronedarone (such as antithrombotic, antihypertensive, or neuroprotective effects). However, given the limitations of the study (post-hoc analysis, an unclear mechanism of action, and strokes were not adjudicated) and suggests somewhat different conclusions than the prior AFFIRM trial (NEJM 2002;347:1825), which looked at rate versus rhythm control in patients on the parent drug amiodarone, and was not associated with decreased risk of stroke, more research is needed.
Bottom line: At present, the mainstay of stroke prevention in atrial fibrillation should remain rate control, anticoagulants, and antiplatelet agents.
Citation: Connolly SJ, Crijns HJ, Torp-Pedersen C, et al. Analysis of stroke in ATHENA: a placebo-controlled, double-blinded, parallel-arm trial to assess the efficacy of dronedarone 400 mg BID for the prevention of cardiovascular hospitalization or death from any cause in patients with atrial fibrillation/atrial flutter. Circulation. 2009;120(13):1174-1180.
Reviewed for TH eWire by Bhaskar Arora, MD, Thomas Barrett, MD, MCR, FHM, Honora Englander, MD, Stephanie Halvorson, MD, Alan J. Hunter, MD, David Kagen, MD, Blake Lesselroth, MD, MBI, Portland Veterans Affairs Medical Center and Division of Hospital Medicine, Oregon Health & Science University.
Clinical question: Does the new antiarrhythmic agent dronedarone, an amiodarone derivative, reduce the incidence of stroke in patients with persistent or paroxysmal atrial fibrillation and at least one risk factor for vascular events?
Background: Antiarrhythmic therapy has not been shown to reduce stroke risk in the treatment of chronic atrial fibrillation. A sub-group analysis of a recent multicentered, international, industry-sponsored, randomized trial of the new antiarrhythmic dronedarone, suggested a potential reduction in stroke risk when compared with placebo.
Study design: Post-hoc analysis of a previous prospective, randomized, placebo-controlled, double-blinded, parallel-arm trial.
Setting: 551 centers in 37 countries.
Synopsis: In addition to anticoagulation and rate control, 4,628 patients with atrial fibrillation and at least one risk factor for cardiovascular (CV) hospitalization were randomized to dronedarone versus placebo. The study assessed the primary outcomes of stroke, acute coronary syndromes, CV, and total mortality.
The dronedarone arm experienced a 0.6% absolute risk reduction (ARR) of stroke {(number-needed-to-treat (NNT) 167); relative risk reduction (RRR) 33%; P=0.027}; 0.7% ARR of “stroke or transient ischemic attack” (NNT 143; RRR 30%; P=0.031); 1.7% ARR of “stroke, ACS, or CV death,” (NNT 59; RRR 31%; P<0.001); and a 1.6% ARR of “stroke, ACS, or all-cause mortality” (NNT 63; RRR 24%; P=0.002). In further subgroup analysis, only the 65% of patients had CHADS2 scores =2, benefited (RR stroke 0.50; 95% CI, 0.32-0.79).
The results are thought-provoking, and suggest there could be more than antiarrhythmic effects of dronedarone (such as antithrombotic, antihypertensive, or neuroprotective effects). However, given the limitations of the study (post-hoc analysis, an unclear mechanism of action, and strokes were not adjudicated) and suggests somewhat different conclusions than the prior AFFIRM trial (NEJM 2002;347:1825), which looked at rate versus rhythm control in patients on the parent drug amiodarone, and was not associated with decreased risk of stroke, more research is needed.
Bottom line: At present, the mainstay of stroke prevention in atrial fibrillation should remain rate control, anticoagulants, and antiplatelet agents.
Citation: Connolly SJ, Crijns HJ, Torp-Pedersen C, et al. Analysis of stroke in ATHENA: a placebo-controlled, double-blinded, parallel-arm trial to assess the efficacy of dronedarone 400 mg BID for the prevention of cardiovascular hospitalization or death from any cause in patients with atrial fibrillation/atrial flutter. Circulation. 2009;120(13):1174-1180.
Reviewed for TH eWire by Bhaskar Arora, MD, Thomas Barrett, MD, MCR, FHM, Honora Englander, MD, Stephanie Halvorson, MD, Alan J. Hunter, MD, David Kagen, MD, Blake Lesselroth, MD, MBI, Portland Veterans Affairs Medical Center and Division of Hospital Medicine, Oregon Health & Science University.
Clinical question: Does the new antiarrhythmic agent dronedarone, an amiodarone derivative, reduce the incidence of stroke in patients with persistent or paroxysmal atrial fibrillation and at least one risk factor for vascular events?
Background: Antiarrhythmic therapy has not been shown to reduce stroke risk in the treatment of chronic atrial fibrillation. A sub-group analysis of a recent multicentered, international, industry-sponsored, randomized trial of the new antiarrhythmic dronedarone, suggested a potential reduction in stroke risk when compared with placebo.
Study design: Post-hoc analysis of a previous prospective, randomized, placebo-controlled, double-blinded, parallel-arm trial.
Setting: 551 centers in 37 countries.
Synopsis: In addition to anticoagulation and rate control, 4,628 patients with atrial fibrillation and at least one risk factor for cardiovascular (CV) hospitalization were randomized to dronedarone versus placebo. The study assessed the primary outcomes of stroke, acute coronary syndromes, CV, and total mortality.
The dronedarone arm experienced a 0.6% absolute risk reduction (ARR) of stroke {(number-needed-to-treat (NNT) 167); relative risk reduction (RRR) 33%; P=0.027}; 0.7% ARR of “stroke or transient ischemic attack” (NNT 143; RRR 30%; P=0.031); 1.7% ARR of “stroke, ACS, or CV death,” (NNT 59; RRR 31%; P<0.001); and a 1.6% ARR of “stroke, ACS, or all-cause mortality” (NNT 63; RRR 24%; P=0.002). In further subgroup analysis, only the 65% of patients had CHADS2 scores =2, benefited (RR stroke 0.50; 95% CI, 0.32-0.79).
The results are thought-provoking, and suggest there could be more than antiarrhythmic effects of dronedarone (such as antithrombotic, antihypertensive, or neuroprotective effects). However, given the limitations of the study (post-hoc analysis, an unclear mechanism of action, and strokes were not adjudicated) and suggests somewhat different conclusions than the prior AFFIRM trial (NEJM 2002;347:1825), which looked at rate versus rhythm control in patients on the parent drug amiodarone, and was not associated with decreased risk of stroke, more research is needed.
Bottom line: At present, the mainstay of stroke prevention in atrial fibrillation should remain rate control, anticoagulants, and antiplatelet agents.
Citation: Connolly SJ, Crijns HJ, Torp-Pedersen C, et al. Analysis of stroke in ATHENA: a placebo-controlled, double-blinded, parallel-arm trial to assess the efficacy of dronedarone 400 mg BID for the prevention of cardiovascular hospitalization or death from any cause in patients with atrial fibrillation/atrial flutter. Circulation. 2009;120(13):1174-1180.
Reviewed for TH eWire by Bhaskar Arora, MD, Thomas Barrett, MD, MCR, FHM, Honora Englander, MD, Stephanie Halvorson, MD, Alan J. Hunter, MD, David Kagen, MD, Blake Lesselroth, MD, MBI, Portland Veterans Affairs Medical Center and Division of Hospital Medicine, Oregon Health & Science University.
Congress Gets Defensive with Medicare Payments
Congressional action has delayed a potentially devastating cut in Medicare physician reimbursements for at least two more months, while a separate attempt to delay the looming elimination of Medicare’s consultation billing codes now seems increasingly unlikely to succeed.
In November, the House of Representatives passed a bill that would have rescinded the 21.2% cut to Medicare’s physician fee schedule for 2010 (dictated by the current formula’s sustainable growth rate, or SGR). But the Senate balked at the expected $247 billion price tag, and was unable to muster enough votes to avert a filibuster.
Trying a different tack, House Democrats used the must-pass Defense appropriations bill to push through an amendment freezing Medicare payments at current levels through February, buying Congress more time to find a better solution. The Senate followed suit by approving the bill on Saturday, though a longer-term fix is still in flux. A joint letter by SHM, the American Medical Association (AMA), and other physicians groups calls for a permanent end to the SGR formula—a potentially contentious issue that will await Congress in 2010.
Meanwhile, a request from the AMA and other physician groups to delay the elimination of Medicare consultation codes for a year to allow more time for guidance and ironing out technical issues has yielded no guarantees from the Centers for Medicare and Medicaid Services (CMS). Sen. Arlen Specter (D-Penn.) had offered an amendment seeking such a delay to the Senate’s healthcare reform legislation, but a spokesperson from Spector's office said the amendment did not move forward with the Senate bill—an exclusion that now makes a last-minute reprieve unlikely.
In the interim, CMS has released a 29-page transmittal explaining how the eliminated codes will be replaced by existing evaluation and management codes. Click here to download a PDF of the transmittal.
Congressional action has delayed a potentially devastating cut in Medicare physician reimbursements for at least two more months, while a separate attempt to delay the looming elimination of Medicare’s consultation billing codes now seems increasingly unlikely to succeed.
In November, the House of Representatives passed a bill that would have rescinded the 21.2% cut to Medicare’s physician fee schedule for 2010 (dictated by the current formula’s sustainable growth rate, or SGR). But the Senate balked at the expected $247 billion price tag, and was unable to muster enough votes to avert a filibuster.
Trying a different tack, House Democrats used the must-pass Defense appropriations bill to push through an amendment freezing Medicare payments at current levels through February, buying Congress more time to find a better solution. The Senate followed suit by approving the bill on Saturday, though a longer-term fix is still in flux. A joint letter by SHM, the American Medical Association (AMA), and other physicians groups calls for a permanent end to the SGR formula—a potentially contentious issue that will await Congress in 2010.
Meanwhile, a request from the AMA and other physician groups to delay the elimination of Medicare consultation codes for a year to allow more time for guidance and ironing out technical issues has yielded no guarantees from the Centers for Medicare and Medicaid Services (CMS). Sen. Arlen Specter (D-Penn.) had offered an amendment seeking such a delay to the Senate’s healthcare reform legislation, but a spokesperson from Spector's office said the amendment did not move forward with the Senate bill—an exclusion that now makes a last-minute reprieve unlikely.
In the interim, CMS has released a 29-page transmittal explaining how the eliminated codes will be replaced by existing evaluation and management codes. Click here to download a PDF of the transmittal.
Congressional action has delayed a potentially devastating cut in Medicare physician reimbursements for at least two more months, while a separate attempt to delay the looming elimination of Medicare’s consultation billing codes now seems increasingly unlikely to succeed.
In November, the House of Representatives passed a bill that would have rescinded the 21.2% cut to Medicare’s physician fee schedule for 2010 (dictated by the current formula’s sustainable growth rate, or SGR). But the Senate balked at the expected $247 billion price tag, and was unable to muster enough votes to avert a filibuster.
Trying a different tack, House Democrats used the must-pass Defense appropriations bill to push through an amendment freezing Medicare payments at current levels through February, buying Congress more time to find a better solution. The Senate followed suit by approving the bill on Saturday, though a longer-term fix is still in flux. A joint letter by SHM, the American Medical Association (AMA), and other physicians groups calls for a permanent end to the SGR formula—a potentially contentious issue that will await Congress in 2010.
Meanwhile, a request from the AMA and other physician groups to delay the elimination of Medicare consultation codes for a year to allow more time for guidance and ironing out technical issues has yielded no guarantees from the Centers for Medicare and Medicaid Services (CMS). Sen. Arlen Specter (D-Penn.) had offered an amendment seeking such a delay to the Senate’s healthcare reform legislation, but a spokesperson from Spector's office said the amendment did not move forward with the Senate bill—an exclusion that now makes a last-minute reprieve unlikely.
In the interim, CMS has released a 29-page transmittal explaining how the eliminated codes will be replaced by existing evaluation and management codes. Click here to download a PDF of the transmittal.
Project BOOST Expansion Planned
Project BOOST (Better Outcomes for Older Adults through Safer Transitions) is getting, well, a pretty big boost in 2010.
The pilot transitional-care program implemented at 30 sites in the past 18 months pairs SHM mentors with hospitalists to improve care via a discharge planning toolkit. Preliminary plans call for up to 45 additional sites staggered over three cohorts in the next 12 months.
Tina Budnitz, MPH, SHM senior advisor and director of Project BOOST, says the initiative also is working to create a tuition-based model, planning an educational Web-based seminar and standardized “teachback” materials. The BOOST team also is promoting an Internet listserv that hospitalists at the debut sites have used as a communal communication tool.
“It is a big jump,” Budnitz acknowledges. “We think the secret sauce that’s made it work so far will be able to scale.”
Quantifiable data from the first six sites, which were selected for the program in the summer of 2008, could be released this spring. A lack of metrics, however, has done little to stymie the initiative’s growth. Two sites—Hospital of the University of Pennsylvania in Philadelphia and St. Mary’s Health Center in St. Louis, Mo.—already have won safety awards related to the program.
One of the program’s mentors says the early success can be tied to hospitalists’ commitment to using the toolkit as the basis of a transitional-care upgrade, instead of expecting the program’s presence alone to improve care.
“It’s way more than what is BOOST and ‘how are we going to implement?’ ” says Janet Nagamine, MD, FHM, hospitalist at Kaiser Permanente in Santa Clara, Calif., SHM board member, and chair of SHM’s Hospital Quality and Patient Safety Committee. “They have really looked at their entire discharge process and done a lot of looking at their individual and specific challenges and how to overcome them.”
Project BOOST (Better Outcomes for Older Adults through Safer Transitions) is getting, well, a pretty big boost in 2010.
The pilot transitional-care program implemented at 30 sites in the past 18 months pairs SHM mentors with hospitalists to improve care via a discharge planning toolkit. Preliminary plans call for up to 45 additional sites staggered over three cohorts in the next 12 months.
Tina Budnitz, MPH, SHM senior advisor and director of Project BOOST, says the initiative also is working to create a tuition-based model, planning an educational Web-based seminar and standardized “teachback” materials. The BOOST team also is promoting an Internet listserv that hospitalists at the debut sites have used as a communal communication tool.
“It is a big jump,” Budnitz acknowledges. “We think the secret sauce that’s made it work so far will be able to scale.”
Quantifiable data from the first six sites, which were selected for the program in the summer of 2008, could be released this spring. A lack of metrics, however, has done little to stymie the initiative’s growth. Two sites—Hospital of the University of Pennsylvania in Philadelphia and St. Mary’s Health Center in St. Louis, Mo.—already have won safety awards related to the program.
One of the program’s mentors says the early success can be tied to hospitalists’ commitment to using the toolkit as the basis of a transitional-care upgrade, instead of expecting the program’s presence alone to improve care.
“It’s way more than what is BOOST and ‘how are we going to implement?’ ” says Janet Nagamine, MD, FHM, hospitalist at Kaiser Permanente in Santa Clara, Calif., SHM board member, and chair of SHM’s Hospital Quality and Patient Safety Committee. “They have really looked at their entire discharge process and done a lot of looking at their individual and specific challenges and how to overcome them.”
Project BOOST (Better Outcomes for Older Adults through Safer Transitions) is getting, well, a pretty big boost in 2010.
The pilot transitional-care program implemented at 30 sites in the past 18 months pairs SHM mentors with hospitalists to improve care via a discharge planning toolkit. Preliminary plans call for up to 45 additional sites staggered over three cohorts in the next 12 months.
Tina Budnitz, MPH, SHM senior advisor and director of Project BOOST, says the initiative also is working to create a tuition-based model, planning an educational Web-based seminar and standardized “teachback” materials. The BOOST team also is promoting an Internet listserv that hospitalists at the debut sites have used as a communal communication tool.
“It is a big jump,” Budnitz acknowledges. “We think the secret sauce that’s made it work so far will be able to scale.”
Quantifiable data from the first six sites, which were selected for the program in the summer of 2008, could be released this spring. A lack of metrics, however, has done little to stymie the initiative’s growth. Two sites—Hospital of the University of Pennsylvania in Philadelphia and St. Mary’s Health Center in St. Louis, Mo.—already have won safety awards related to the program.
One of the program’s mentors says the early success can be tied to hospitalists’ commitment to using the toolkit as the basis of a transitional-care upgrade, instead of expecting the program’s presence alone to improve care.
“It’s way more than what is BOOST and ‘how are we going to implement?’ ” says Janet Nagamine, MD, FHM, hospitalist at Kaiser Permanente in Santa Clara, Calif., SHM board member, and chair of SHM’s Hospital Quality and Patient Safety Committee. “They have really looked at their entire discharge process and done a lot of looking at their individual and specific challenges and how to overcome them.”
CML Treatment: Old Drug, New Name
There's a new old drug for chronic myeloid leukemia, and it seems to work in patients who don't respond to any of the approved therapies for this disease. Dr. Jorge Cortes discusses the potential of omacetaxine mepesuccinate at the annual meeting of the American Society of Hematology.
There's a new old drug for chronic myeloid leukemia, and it seems to work in patients who don't respond to any of the approved therapies for this disease. Dr. Jorge Cortes discusses the potential of omacetaxine mepesuccinate at the annual meeting of the American Society of Hematology.
There's a new old drug for chronic myeloid leukemia, and it seems to work in patients who don't respond to any of the approved therapies for this disease. Dr. Jorge Cortes discusses the potential of omacetaxine mepesuccinate at the annual meeting of the American Society of Hematology.
Multiple Myeloma in the Elderly
In the VISTA trial, the Spanish Myeloma Group created an induction regimen of bortezomib, melphalan, and prednisone that elderly patients could tolerate. This year the group took that regimen a step further.
In the VISTA trial, the Spanish Myeloma Group created an induction regimen of bortezomib, melphalan, and prednisone that elderly patients could tolerate. This year the group took that regimen a step further.
In the VISTA trial, the Spanish Myeloma Group created an induction regimen of bortezomib, melphalan, and prednisone that elderly patients could tolerate. This year the group took that regimen a step further.
First-Line Lymphoma Treatments
Bendamustine plus rituximab has the potential to replace standard CHOP chemotherapy plus rituximab in the first-line treatment of advanced lymphomas. Dr. Mathias J. Rummel discusses data presented at the annual meeting of the American Society of Hematology.
Bendamustine plus rituximab has the potential to replace standard CHOP chemotherapy plus rituximab in the first-line treatment of advanced lymphomas. Dr. Mathias J. Rummel discusses data presented at the annual meeting of the American Society of Hematology.
Bendamustine plus rituximab has the potential to replace standard CHOP chemotherapy plus rituximab in the first-line treatment of advanced lymphomas. Dr. Mathias J. Rummel discusses data presented at the annual meeting of the American Society of Hematology.