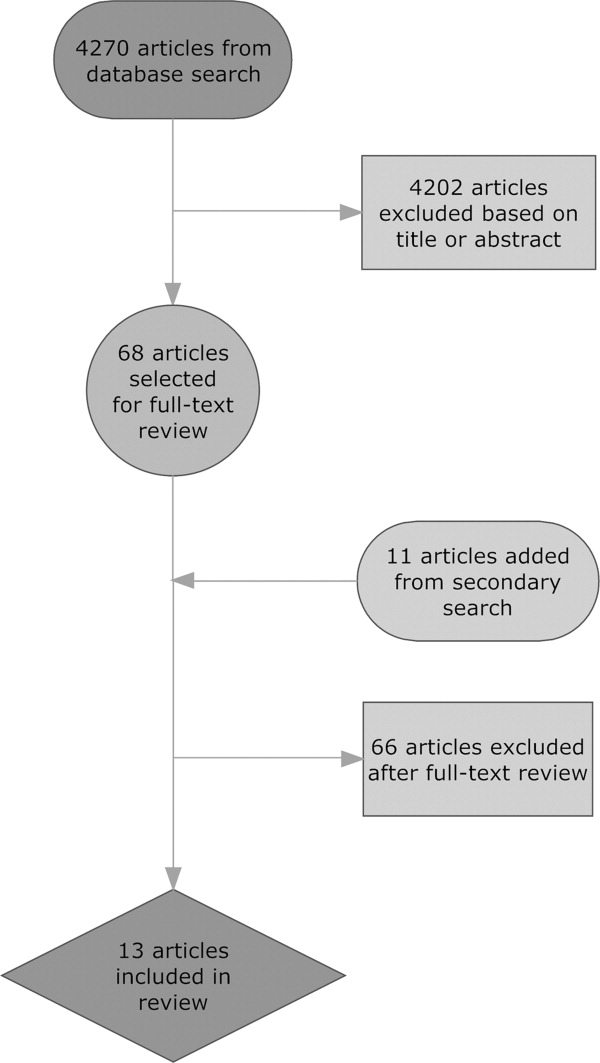
User login
Community Hospitalist Time‐flow
In 2006, after introducing formal hospitalist programs at both an academic hospital and an affiliated community teaching hospital, we conducted a time study to gain insight into the effect of adopting a community model in an academic environment. This evaluation was conducted to identify similarities and differences between the 2 programs and to highlight opportunities for process and quality improvement. The hospitalist case mix index (CMI) was higher at the academic center (1.3) than at the community center (1.1). At both institutions documentation and most order entry were completed on paper, while lab and test results were electronically available. Both hospitalist programs were nonteaching services with day shifts staffed from 7:00 AM to 7:00 PM. At the academic center, a single hospitalist staffed the service for 7 days in a row with an average daily census of 10 patients. At the community hospital, 2 hospitalists carried the service, alternating days as the primary admitter. These hospitalists each carried an average census of 13 patients for 6 days in a row with staggered start/stop dates to ensure service continuity. The years of experience as a practicing hospitalist were similar between the 2 programs (median 4 years and range 1‐10 years for both programs); all hospitalists completed an internal medicine residency.
Methods
A paper‐based tool was used to collect data at 1‐minute intervals into 5 major categories validated through trial observation, content focus groups, and expert opinion. The 5 categories were Direct Patient Care, Indirect Patient Care, Travel, Personal, and Other (Table 1). Communication, a subcategory of Indirect Patient Care, was further classified by the job‐profession category and communication modality of the individual(s) interacting with the hospitalist. The tool allowed for more than 1 task category to be tracked at a time in order to capture multitasking. Three trained industrial engineers shadowed 9 different hospitalists during the day shifts, between 2 and 5 shifts per hospitalist, gathering approximately 355 hours of observational data over the 8 weeks of the study; 4 weeks at each hospital. Weekend and night shift data were not collected due to observer availability. Results for each setting were reported as the mean and standard deviation percentage of physician time observed for each task category. The results were also reported as the mean and standard deviation volume adjusted time per patient for each task category. The adjustment was made by dividing physician time by the number of patient encounters for that observation. Comparative analyses were calculated using a t‐test with a significance level of 0.05 and confidence intervals were reported at a 95% interval. Since this project was a quality improvement initiative analyzing the introduction of a new clinical service, Institutional Review Board (IRB) approval from our institution was not required.
| Category | Definition |
|---|---|
| |
| Direct patient care | Interviewing patient, examining patient, performing procedure on patient, family meeting |
| Indirect patient care | Subcategories listed below |
| Documentation | Writing rules, filling out forms, dictating |
| Orders | Writing paper orders in patient chart, entering orders in CPOE |
| Reviewing records | Looking up medical records in either electronic or paper chart |
| Medical references | Reviewing text books or using computer to consult UpToDate, do literature search, review Micromedix, or use personal digital assistant (PDA) to look up similar information |
| Other indirect patient care | Looking for paper chart, forms, procedural items or work space; waiting for page return, computer to lead, etc |
| Communication | Subcategories listed below |
| Nurse/tech | Nurse or medical technologist |
| Case manager | Case manager or social worker |
| Primary care physician | Talking with the patient's primary care physician |
| Inpatient physician | Specialist attendings, fellows, residents, medical students, other hospitalists |
| Other staff | Pharmacist, therapist, nurse practitioner, physician assistant, unit clerk |
| Phone | On the phone (attribute of Communication) |
| Emailing or text paging (attribute of Communication) | |
| In person | Face to face discussion (attribute of Communication) |
| Personal | Lunch, restroom, calls |
| Travel | Walking between units |
| Other | Meetings, administrative activities |
Results
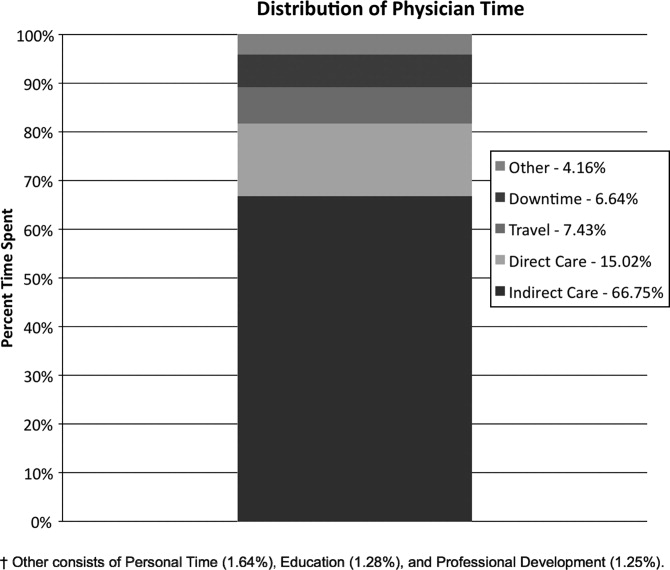
Hospitalist time allocations at the 2 programs were comparatively similar (Table 2). At the academic center, hospitalists spent the majority of their time providing indirect patient care (69.8%, CI: 66.3‐73.3%), followed by direct patient care (13.1%, CI: 11.2‐14.9%), with the remaining time distributed among travel, personal, and other administrative duties. Likewise, the community hospitalists spent the majority of their time providing indirect patient care (68.7%, CI: 63.0‐74.5%), followed by direct patient care (16.7%, CI: 14.1‐19.4%), with travel, personal, and administrative duties completing the day. Additionally, the percent of time spent multitasking, defined as more than 1 task category observed at the same time, was strikingly similar between the 2 groups (Academic: 47.6% 16.5%, Community: 47.9% 9.8%).
| Academic (%) | Community (%) | P Value | |||
|---|---|---|---|---|---|
| Mean | Stdev | Mean | Stdev | ||
| Direct patient care | 13.8 | 4.1 | 17.2 | 6.3 | 0.032 |
| Indirect patient care | 68.2 | 8.0 | 68.0 | 13.2 | 0.756 |
| Documentation | 15.4 | 3.3% | 22.0 | 6.2 | 0.000 |
| Orders | 6.3 | 1.5 | 4.7 | 1.6 | 0.011 |
| Community Rev records | 21.3 | 5.0 | 21.7 | 6.2 | 0.000 |
| Medical refs | l.5 | 0.8 | 0.6 | 0.6 | 0.000 |
| Other indirect patient care | 2.0 | 1.3 | 2.6 | 1.6 | 0.210 |
| Communication | 21.7 | 4.2 | 16.5 | 4.7 | 0.000 |
| Nurse/tech | 5.4 | 2.0 | 5.3 | 2.8 | 0.895 |
| Care manager | 2.8 | 1.8 | 3.4 | 1.7 | 0.229 |
| Primary care physician | 1.1 | 1.2 | 1.1 | 1.1 | 0.818 |
| Inpatient physician | 12.5 | 3.9 | 6.7 | 2.6 | 0.000 |
| Other staff | 6.4 | 9.7 | 2.3 | 1.2 | 0.029 |
| Personal | 4.1 | 2.4 | 2.5 | 1.8 | 0.029 |
| Travel | 4.4 | 1.2 | 3.9 | 1.0 | 0.311 |
| Other | 9.5 | 8.9 | 8.4 | 17.4 | 0.850 |
While the difference in total percent of time spent on direct patient care was statistically significant (P = 0.03), the values converged after adjusting for the differences in average daily census (Table 3). On average, both the academic and community hospitalists spent approximately 10 minutes per patient per day interacting face to face with the patient and/or family (10.0 2.9 minutes and 10.1 3.6 minutes respectively, P = 0.89). However, after volume adjusting, other workflow differences became statistically significant, primarily in indirect patient care (Academic: 54.7 11.1 minutes/patient, Community: 41.9 9.8 minutes/patient, P 0.001). The academic hospitalists spent more time writing orders (4.6 1.3 minutes/patient vs. 2.8 1.1 minutes/patient, P 0.001), looking up and reviewing medical reference materials (1.1 0.6 minutes/patient vs. 0.3 0.4 minutes/patient, P 0.001), and communicating with other providers (20.5 7.7 min/patient vs. 11.1 3.1 min/patient, P 0.001) than their community hospitalist counterparts. Nearly half the time that the academic hospitalists spent communicating was dedicated to speaking with other physicians (9.2 3.5 minutes/patient); more than double that of the community hospitalists (4.0 1.6 minutes). Additionally, the academic hospitalists spent more time speaking with pharmacists (0.7 0.6 minutes vs. 0.1 0.2 minutes, P = 0.001).
| Academic | Community | P Value | |||
|---|---|---|---|---|---|
| Mean | Stdev | Mean | Stdev | ||
| Dir pt care | 10.0 | 2.9 | 10.1 | 3.6 | 0.890 |
| Indirect pt care | 50.1 | 8.4 | 40.5 | 9.8 | 0.000 |
| Documentation | 11.3 | 2.7 | 13.1 | 3.9 | 0.101 |
| Orders | 4.6 | 1.3 | 2.8 | 1.1 | 0.000 |
| Rev records | 15.6 | 4.0 | 13.0 | 4.8 | 0.069 |
| Medial refs | 1.1 | 0.6 | 0.3 | 0.4 | 0.000 |
| Other pt care | 1.5 | 1.0 | 1.5 | 1.0 | 0.833 |
| Communication | 16.0 | 3.8 | 9.7 | 2.8 | 0.000 |
| Nurse/tech | 3.9 | 1.4 | 3.1 | 1.6 | 0.102 |
| Case manager | 2.0 | 1.3 | 2.0 | 1.0 | 0.950 |
| Prim care physician | 0.8 | 0.9 | 0.7 | 0.7 | 0.547 |
| Inpatient physician | 9.2 | 3.5 | 4.0 | 1.6 | 0.000 |
| Other staff | 4.6 | 6.8 | 1.4 | 0.7 | 0.049 |
| Personal | 3.0 | 1.8 | 1.5 | 1.0 | 0.002 |
| Travel | 3.2 | 0.9 | 2.3 | 0.6 | 0.001 |
| Other | 6.8 | 6.0 | 4.4 | 8.4 | 0.306 |
Discussion
In 2006, O'Leary et al.1 demonstrated that academic hospitalists spend approximately 20% of their time engaged in direct patient care. Our results are consistent with these data and further expand these findings to a community setting. Although we found subtle workflow differences between the academic and community programs, their similarities were more striking than their differences. We suspect that these differences can be largely attributed to the higher CMI at the academic program as well as the greater complexity and additional communication hand‐offs inherent to this tertiary academic medical center. For example, at the academic medical center, medicine admissions were screened by a medicine triage resident and subsequently handed off to a hospitalist. In most cases, this system did not preclude the need to speak directly with the emergency department (ED) attending, adding a layer of complexity that did not exist in the community hospital. Finally, in contrast to the community hospital, there was little comanagement at the academic medical center, necessitating frequent transfers to and from medical and subspecialty services.
It appears that hospitalists, irrespective of their work environment, spend far more time documenting, communicating, and coordinating care than at the bedside. It is unclear whether this represents a desirable outcome of hospitalists' role as managers of complex hospital stays or inefficient and ineffective effort that should be mitigated through care delivery redesign. Further research to optimize hospital information management, streamline care processes and eliminate low value‐added effort is clearly needed.
Another notable finding of our study is that hospitalists spend roughly half of their time performing more than 1 work category at the same time deemed as multitasking.2 The prevalence and effects of multitasking are well‐characterized in emergency medicine and likely apply to hospitalists.3, 4 Fractured attention due to multitasking may hamper communication, jeopardize care handoffs, and increase risk for medical errors and litigation.46 While it is likely that multitasking is inherent to the practice of hospital medicine, it is unclear how this could be mitigated or better facilitated. Perhaps this could be done through structured communication and information management. This too merits further investigation.
Lastly, this study found that it takes approximately an hour of a hospitalist's time each day to manage 1 patient's care. This in and of itself, is very important from the standpoint of both billing and workload. In today's professional services fee model, there are a number of components that contribute to the level of service that a hospitalist can bill. One of those components is time, specifically the time spent counseling and/or coordinating care, which as this study suggests, dominates a hospitalist's workday. It is therefore critical that hospitalists accurately and consistently document the amount of time they spend with each patient and specifically describe the counseling and/or activities to coordinate care. Additionally, recognizing how much time is required for a hospitalist to care for a patient has important workload implications. If we assume that it takes approximately an hour per patient and a typical workday is around 11 hours after subtracting personal time, then it would be reasonable to expect that a single hospitalist should have, on average, 11 patient encounters per day. This number is, of course, completely dependent on organizational factors such as a specific hospital's support systems and the mix of admissions, follow‐ups, and discharges on that service.
Our study has several limitations. The time study occurred at 2 hospitals, in 1 mid‐sized Midwestern city, and the results may not be generalizable to other settings. However, the congruence of our findings with those of O'Leary et al.1 suggests that our results maintain external validity. Second, at the time of the study the 2 programs were relatively new and workflows were still evolving. Additionally, the academic and community hospitalist programs were under unified management and 2 of the surveyed hospitalists worked at both programs. This may have artificially homogenized the work patterns observed at both programs. Finally, observing hospitalist activities exclusively during the weekday daytime shifts has the potential to bias the results. However, the night and weekend duties and responsibilities of the 2 programs differed significantly, which would have made it very difficult to derive meaningful comparisons for those observations.
Conclusion
We found that hospitalists in both academic and community settings spend the majority of their time multitasking and engaged in indirect patient care. Further studies are necessary to determine the extent to which this is a necessary feature of the hospitalist care model and whether hospitalists should restructure their workflow to improve outcomes.
- , , .How hospitalists spend their time: Insights on efficiency and safety.J Hosp Med.2006;1:88–93.
- , , , .Emergency department workplace interruptions: Are emergency physicians “interrupt‐driven” and “multitasking”?Acad Emerg Med.2008;7:1239–1243.
- , , , .Work interrupted: a comparison of workplace interruptions in the emergency departments and primary care offices.Ann Emerg Med.2001;38:146–151.
- , , , , , .The multitasking clinician: Decision‐making and cognitive demand during and after team handoffs in emergency care.Int J Med Inform.2007;76:801–811.
- , , , , .Communication loads on clinical staff in the emergency department.Med J Aust.2002;176:415–418.
- , , .Why do people sue doctors? A study of patients and relatives taking legal action.Lancet.1994;343:1609–1613.
In 2006, after introducing formal hospitalist programs at both an academic hospital and an affiliated community teaching hospital, we conducted a time study to gain insight into the effect of adopting a community model in an academic environment. This evaluation was conducted to identify similarities and differences between the 2 programs and to highlight opportunities for process and quality improvement. The hospitalist case mix index (CMI) was higher at the academic center (1.3) than at the community center (1.1). At both institutions documentation and most order entry were completed on paper, while lab and test results were electronically available. Both hospitalist programs were nonteaching services with day shifts staffed from 7:00 AM to 7:00 PM. At the academic center, a single hospitalist staffed the service for 7 days in a row with an average daily census of 10 patients. At the community hospital, 2 hospitalists carried the service, alternating days as the primary admitter. These hospitalists each carried an average census of 13 patients for 6 days in a row with staggered start/stop dates to ensure service continuity. The years of experience as a practicing hospitalist were similar between the 2 programs (median 4 years and range 1‐10 years for both programs); all hospitalists completed an internal medicine residency.
Methods
A paper‐based tool was used to collect data at 1‐minute intervals into 5 major categories validated through trial observation, content focus groups, and expert opinion. The 5 categories were Direct Patient Care, Indirect Patient Care, Travel, Personal, and Other (Table 1). Communication, a subcategory of Indirect Patient Care, was further classified by the job‐profession category and communication modality of the individual(s) interacting with the hospitalist. The tool allowed for more than 1 task category to be tracked at a time in order to capture multitasking. Three trained industrial engineers shadowed 9 different hospitalists during the day shifts, between 2 and 5 shifts per hospitalist, gathering approximately 355 hours of observational data over the 8 weeks of the study; 4 weeks at each hospital. Weekend and night shift data were not collected due to observer availability. Results for each setting were reported as the mean and standard deviation percentage of physician time observed for each task category. The results were also reported as the mean and standard deviation volume adjusted time per patient for each task category. The adjustment was made by dividing physician time by the number of patient encounters for that observation. Comparative analyses were calculated using a t‐test with a significance level of 0.05 and confidence intervals were reported at a 95% interval. Since this project was a quality improvement initiative analyzing the introduction of a new clinical service, Institutional Review Board (IRB) approval from our institution was not required.
| Category | Definition |
|---|---|
| |
| Direct patient care | Interviewing patient, examining patient, performing procedure on patient, family meeting |
| Indirect patient care | Subcategories listed below |
| Documentation | Writing rules, filling out forms, dictating |
| Orders | Writing paper orders in patient chart, entering orders in CPOE |
| Reviewing records | Looking up medical records in either electronic or paper chart |
| Medical references | Reviewing text books or using computer to consult UpToDate, do literature search, review Micromedix, or use personal digital assistant (PDA) to look up similar information |
| Other indirect patient care | Looking for paper chart, forms, procedural items or work space; waiting for page return, computer to lead, etc |
| Communication | Subcategories listed below |
| Nurse/tech | Nurse or medical technologist |
| Case manager | Case manager or social worker |
| Primary care physician | Talking with the patient's primary care physician |
| Inpatient physician | Specialist attendings, fellows, residents, medical students, other hospitalists |
| Other staff | Pharmacist, therapist, nurse practitioner, physician assistant, unit clerk |
| Phone | On the phone (attribute of Communication) |
| Emailing or text paging (attribute of Communication) | |
| In person | Face to face discussion (attribute of Communication) |
| Personal | Lunch, restroom, calls |
| Travel | Walking between units |
| Other | Meetings, administrative activities |
Results
Hospitalist time allocations at the 2 programs were comparatively similar (Table 2). At the academic center, hospitalists spent the majority of their time providing indirect patient care (69.8%, CI: 66.3‐73.3%), followed by direct patient care (13.1%, CI: 11.2‐14.9%), with the remaining time distributed among travel, personal, and other administrative duties. Likewise, the community hospitalists spent the majority of their time providing indirect patient care (68.7%, CI: 63.0‐74.5%), followed by direct patient care (16.7%, CI: 14.1‐19.4%), with travel, personal, and administrative duties completing the day. Additionally, the percent of time spent multitasking, defined as more than 1 task category observed at the same time, was strikingly similar between the 2 groups (Academic: 47.6% 16.5%, Community: 47.9% 9.8%).
| Academic (%) | Community (%) | P Value | |||
|---|---|---|---|---|---|
| Mean | Stdev | Mean | Stdev | ||
| Direct patient care | 13.8 | 4.1 | 17.2 | 6.3 | 0.032 |
| Indirect patient care | 68.2 | 8.0 | 68.0 | 13.2 | 0.756 |
| Documentation | 15.4 | 3.3% | 22.0 | 6.2 | 0.000 |
| Orders | 6.3 | 1.5 | 4.7 | 1.6 | 0.011 |
| Community Rev records | 21.3 | 5.0 | 21.7 | 6.2 | 0.000 |
| Medical refs | l.5 | 0.8 | 0.6 | 0.6 | 0.000 |
| Other indirect patient care | 2.0 | 1.3 | 2.6 | 1.6 | 0.210 |
| Communication | 21.7 | 4.2 | 16.5 | 4.7 | 0.000 |
| Nurse/tech | 5.4 | 2.0 | 5.3 | 2.8 | 0.895 |
| Care manager | 2.8 | 1.8 | 3.4 | 1.7 | 0.229 |
| Primary care physician | 1.1 | 1.2 | 1.1 | 1.1 | 0.818 |
| Inpatient physician | 12.5 | 3.9 | 6.7 | 2.6 | 0.000 |
| Other staff | 6.4 | 9.7 | 2.3 | 1.2 | 0.029 |
| Personal | 4.1 | 2.4 | 2.5 | 1.8 | 0.029 |
| Travel | 4.4 | 1.2 | 3.9 | 1.0 | 0.311 |
| Other | 9.5 | 8.9 | 8.4 | 17.4 | 0.850 |
While the difference in total percent of time spent on direct patient care was statistically significant (P = 0.03), the values converged after adjusting for the differences in average daily census (Table 3). On average, both the academic and community hospitalists spent approximately 10 minutes per patient per day interacting face to face with the patient and/or family (10.0 2.9 minutes and 10.1 3.6 minutes respectively, P = 0.89). However, after volume adjusting, other workflow differences became statistically significant, primarily in indirect patient care (Academic: 54.7 11.1 minutes/patient, Community: 41.9 9.8 minutes/patient, P 0.001). The academic hospitalists spent more time writing orders (4.6 1.3 minutes/patient vs. 2.8 1.1 minutes/patient, P 0.001), looking up and reviewing medical reference materials (1.1 0.6 minutes/patient vs. 0.3 0.4 minutes/patient, P 0.001), and communicating with other providers (20.5 7.7 min/patient vs. 11.1 3.1 min/patient, P 0.001) than their community hospitalist counterparts. Nearly half the time that the academic hospitalists spent communicating was dedicated to speaking with other physicians (9.2 3.5 minutes/patient); more than double that of the community hospitalists (4.0 1.6 minutes). Additionally, the academic hospitalists spent more time speaking with pharmacists (0.7 0.6 minutes vs. 0.1 0.2 minutes, P = 0.001).
| Academic | Community | P Value | |||
|---|---|---|---|---|---|
| Mean | Stdev | Mean | Stdev | ||
| Dir pt care | 10.0 | 2.9 | 10.1 | 3.6 | 0.890 |
| Indirect pt care | 50.1 | 8.4 | 40.5 | 9.8 | 0.000 |
| Documentation | 11.3 | 2.7 | 13.1 | 3.9 | 0.101 |
| Orders | 4.6 | 1.3 | 2.8 | 1.1 | 0.000 |
| Rev records | 15.6 | 4.0 | 13.0 | 4.8 | 0.069 |
| Medial refs | 1.1 | 0.6 | 0.3 | 0.4 | 0.000 |
| Other pt care | 1.5 | 1.0 | 1.5 | 1.0 | 0.833 |
| Communication | 16.0 | 3.8 | 9.7 | 2.8 | 0.000 |
| Nurse/tech | 3.9 | 1.4 | 3.1 | 1.6 | 0.102 |
| Case manager | 2.0 | 1.3 | 2.0 | 1.0 | 0.950 |
| Prim care physician | 0.8 | 0.9 | 0.7 | 0.7 | 0.547 |
| Inpatient physician | 9.2 | 3.5 | 4.0 | 1.6 | 0.000 |
| Other staff | 4.6 | 6.8 | 1.4 | 0.7 | 0.049 |
| Personal | 3.0 | 1.8 | 1.5 | 1.0 | 0.002 |
| Travel | 3.2 | 0.9 | 2.3 | 0.6 | 0.001 |
| Other | 6.8 | 6.0 | 4.4 | 8.4 | 0.306 |
Discussion
In 2006, O'Leary et al.1 demonstrated that academic hospitalists spend approximately 20% of their time engaged in direct patient care. Our results are consistent with these data and further expand these findings to a community setting. Although we found subtle workflow differences between the academic and community programs, their similarities were more striking than their differences. We suspect that these differences can be largely attributed to the higher CMI at the academic program as well as the greater complexity and additional communication hand‐offs inherent to this tertiary academic medical center. For example, at the academic medical center, medicine admissions were screened by a medicine triage resident and subsequently handed off to a hospitalist. In most cases, this system did not preclude the need to speak directly with the emergency department (ED) attending, adding a layer of complexity that did not exist in the community hospital. Finally, in contrast to the community hospital, there was little comanagement at the academic medical center, necessitating frequent transfers to and from medical and subspecialty services.
It appears that hospitalists, irrespective of their work environment, spend far more time documenting, communicating, and coordinating care than at the bedside. It is unclear whether this represents a desirable outcome of hospitalists' role as managers of complex hospital stays or inefficient and ineffective effort that should be mitigated through care delivery redesign. Further research to optimize hospital information management, streamline care processes and eliminate low value‐added effort is clearly needed.
Another notable finding of our study is that hospitalists spend roughly half of their time performing more than 1 work category at the same time deemed as multitasking.2 The prevalence and effects of multitasking are well‐characterized in emergency medicine and likely apply to hospitalists.3, 4 Fractured attention due to multitasking may hamper communication, jeopardize care handoffs, and increase risk for medical errors and litigation.46 While it is likely that multitasking is inherent to the practice of hospital medicine, it is unclear how this could be mitigated or better facilitated. Perhaps this could be done through structured communication and information management. This too merits further investigation.
Lastly, this study found that it takes approximately an hour of a hospitalist's time each day to manage 1 patient's care. This in and of itself, is very important from the standpoint of both billing and workload. In today's professional services fee model, there are a number of components that contribute to the level of service that a hospitalist can bill. One of those components is time, specifically the time spent counseling and/or coordinating care, which as this study suggests, dominates a hospitalist's workday. It is therefore critical that hospitalists accurately and consistently document the amount of time they spend with each patient and specifically describe the counseling and/or activities to coordinate care. Additionally, recognizing how much time is required for a hospitalist to care for a patient has important workload implications. If we assume that it takes approximately an hour per patient and a typical workday is around 11 hours after subtracting personal time, then it would be reasonable to expect that a single hospitalist should have, on average, 11 patient encounters per day. This number is, of course, completely dependent on organizational factors such as a specific hospital's support systems and the mix of admissions, follow‐ups, and discharges on that service.
Our study has several limitations. The time study occurred at 2 hospitals, in 1 mid‐sized Midwestern city, and the results may not be generalizable to other settings. However, the congruence of our findings with those of O'Leary et al.1 suggests that our results maintain external validity. Second, at the time of the study the 2 programs were relatively new and workflows were still evolving. Additionally, the academic and community hospitalist programs were under unified management and 2 of the surveyed hospitalists worked at both programs. This may have artificially homogenized the work patterns observed at both programs. Finally, observing hospitalist activities exclusively during the weekday daytime shifts has the potential to bias the results. However, the night and weekend duties and responsibilities of the 2 programs differed significantly, which would have made it very difficult to derive meaningful comparisons for those observations.
Conclusion
We found that hospitalists in both academic and community settings spend the majority of their time multitasking and engaged in indirect patient care. Further studies are necessary to determine the extent to which this is a necessary feature of the hospitalist care model and whether hospitalists should restructure their workflow to improve outcomes.
In 2006, after introducing formal hospitalist programs at both an academic hospital and an affiliated community teaching hospital, we conducted a time study to gain insight into the effect of adopting a community model in an academic environment. This evaluation was conducted to identify similarities and differences between the 2 programs and to highlight opportunities for process and quality improvement. The hospitalist case mix index (CMI) was higher at the academic center (1.3) than at the community center (1.1). At both institutions documentation and most order entry were completed on paper, while lab and test results were electronically available. Both hospitalist programs were nonteaching services with day shifts staffed from 7:00 AM to 7:00 PM. At the academic center, a single hospitalist staffed the service for 7 days in a row with an average daily census of 10 patients. At the community hospital, 2 hospitalists carried the service, alternating days as the primary admitter. These hospitalists each carried an average census of 13 patients for 6 days in a row with staggered start/stop dates to ensure service continuity. The years of experience as a practicing hospitalist were similar between the 2 programs (median 4 years and range 1‐10 years for both programs); all hospitalists completed an internal medicine residency.
Methods
A paper‐based tool was used to collect data at 1‐minute intervals into 5 major categories validated through trial observation, content focus groups, and expert opinion. The 5 categories were Direct Patient Care, Indirect Patient Care, Travel, Personal, and Other (Table 1). Communication, a subcategory of Indirect Patient Care, was further classified by the job‐profession category and communication modality of the individual(s) interacting with the hospitalist. The tool allowed for more than 1 task category to be tracked at a time in order to capture multitasking. Three trained industrial engineers shadowed 9 different hospitalists during the day shifts, between 2 and 5 shifts per hospitalist, gathering approximately 355 hours of observational data over the 8 weeks of the study; 4 weeks at each hospital. Weekend and night shift data were not collected due to observer availability. Results for each setting were reported as the mean and standard deviation percentage of physician time observed for each task category. The results were also reported as the mean and standard deviation volume adjusted time per patient for each task category. The adjustment was made by dividing physician time by the number of patient encounters for that observation. Comparative analyses were calculated using a t‐test with a significance level of 0.05 and confidence intervals were reported at a 95% interval. Since this project was a quality improvement initiative analyzing the introduction of a new clinical service, Institutional Review Board (IRB) approval from our institution was not required.
| Category | Definition |
|---|---|
| |
| Direct patient care | Interviewing patient, examining patient, performing procedure on patient, family meeting |
| Indirect patient care | Subcategories listed below |
| Documentation | Writing rules, filling out forms, dictating |
| Orders | Writing paper orders in patient chart, entering orders in CPOE |
| Reviewing records | Looking up medical records in either electronic or paper chart |
| Medical references | Reviewing text books or using computer to consult UpToDate, do literature search, review Micromedix, or use personal digital assistant (PDA) to look up similar information |
| Other indirect patient care | Looking for paper chart, forms, procedural items or work space; waiting for page return, computer to lead, etc |
| Communication | Subcategories listed below |
| Nurse/tech | Nurse or medical technologist |
| Case manager | Case manager or social worker |
| Primary care physician | Talking with the patient's primary care physician |
| Inpatient physician | Specialist attendings, fellows, residents, medical students, other hospitalists |
| Other staff | Pharmacist, therapist, nurse practitioner, physician assistant, unit clerk |
| Phone | On the phone (attribute of Communication) |
| Emailing or text paging (attribute of Communication) | |
| In person | Face to face discussion (attribute of Communication) |
| Personal | Lunch, restroom, calls |
| Travel | Walking between units |
| Other | Meetings, administrative activities |
Results
Hospitalist time allocations at the 2 programs were comparatively similar (Table 2). At the academic center, hospitalists spent the majority of their time providing indirect patient care (69.8%, CI: 66.3‐73.3%), followed by direct patient care (13.1%, CI: 11.2‐14.9%), with the remaining time distributed among travel, personal, and other administrative duties. Likewise, the community hospitalists spent the majority of their time providing indirect patient care (68.7%, CI: 63.0‐74.5%), followed by direct patient care (16.7%, CI: 14.1‐19.4%), with travel, personal, and administrative duties completing the day. Additionally, the percent of time spent multitasking, defined as more than 1 task category observed at the same time, was strikingly similar between the 2 groups (Academic: 47.6% 16.5%, Community: 47.9% 9.8%).
| Academic (%) | Community (%) | P Value | |||
|---|---|---|---|---|---|
| Mean | Stdev | Mean | Stdev | ||
| Direct patient care | 13.8 | 4.1 | 17.2 | 6.3 | 0.032 |
| Indirect patient care | 68.2 | 8.0 | 68.0 | 13.2 | 0.756 |
| Documentation | 15.4 | 3.3% | 22.0 | 6.2 | 0.000 |
| Orders | 6.3 | 1.5 | 4.7 | 1.6 | 0.011 |
| Community Rev records | 21.3 | 5.0 | 21.7 | 6.2 | 0.000 |
| Medical refs | l.5 | 0.8 | 0.6 | 0.6 | 0.000 |
| Other indirect patient care | 2.0 | 1.3 | 2.6 | 1.6 | 0.210 |
| Communication | 21.7 | 4.2 | 16.5 | 4.7 | 0.000 |
| Nurse/tech | 5.4 | 2.0 | 5.3 | 2.8 | 0.895 |
| Care manager | 2.8 | 1.8 | 3.4 | 1.7 | 0.229 |
| Primary care physician | 1.1 | 1.2 | 1.1 | 1.1 | 0.818 |
| Inpatient physician | 12.5 | 3.9 | 6.7 | 2.6 | 0.000 |
| Other staff | 6.4 | 9.7 | 2.3 | 1.2 | 0.029 |
| Personal | 4.1 | 2.4 | 2.5 | 1.8 | 0.029 |
| Travel | 4.4 | 1.2 | 3.9 | 1.0 | 0.311 |
| Other | 9.5 | 8.9 | 8.4 | 17.4 | 0.850 |
While the difference in total percent of time spent on direct patient care was statistically significant (P = 0.03), the values converged after adjusting for the differences in average daily census (Table 3). On average, both the academic and community hospitalists spent approximately 10 minutes per patient per day interacting face to face with the patient and/or family (10.0 2.9 minutes and 10.1 3.6 minutes respectively, P = 0.89). However, after volume adjusting, other workflow differences became statistically significant, primarily in indirect patient care (Academic: 54.7 11.1 minutes/patient, Community: 41.9 9.8 minutes/patient, P 0.001). The academic hospitalists spent more time writing orders (4.6 1.3 minutes/patient vs. 2.8 1.1 minutes/patient, P 0.001), looking up and reviewing medical reference materials (1.1 0.6 minutes/patient vs. 0.3 0.4 minutes/patient, P 0.001), and communicating with other providers (20.5 7.7 min/patient vs. 11.1 3.1 min/patient, P 0.001) than their community hospitalist counterparts. Nearly half the time that the academic hospitalists spent communicating was dedicated to speaking with other physicians (9.2 3.5 minutes/patient); more than double that of the community hospitalists (4.0 1.6 minutes). Additionally, the academic hospitalists spent more time speaking with pharmacists (0.7 0.6 minutes vs. 0.1 0.2 minutes, P = 0.001).
| Academic | Community | P Value | |||
|---|---|---|---|---|---|
| Mean | Stdev | Mean | Stdev | ||
| Dir pt care | 10.0 | 2.9 | 10.1 | 3.6 | 0.890 |
| Indirect pt care | 50.1 | 8.4 | 40.5 | 9.8 | 0.000 |
| Documentation | 11.3 | 2.7 | 13.1 | 3.9 | 0.101 |
| Orders | 4.6 | 1.3 | 2.8 | 1.1 | 0.000 |
| Rev records | 15.6 | 4.0 | 13.0 | 4.8 | 0.069 |
| Medial refs | 1.1 | 0.6 | 0.3 | 0.4 | 0.000 |
| Other pt care | 1.5 | 1.0 | 1.5 | 1.0 | 0.833 |
| Communication | 16.0 | 3.8 | 9.7 | 2.8 | 0.000 |
| Nurse/tech | 3.9 | 1.4 | 3.1 | 1.6 | 0.102 |
| Case manager | 2.0 | 1.3 | 2.0 | 1.0 | 0.950 |
| Prim care physician | 0.8 | 0.9 | 0.7 | 0.7 | 0.547 |
| Inpatient physician | 9.2 | 3.5 | 4.0 | 1.6 | 0.000 |
| Other staff | 4.6 | 6.8 | 1.4 | 0.7 | 0.049 |
| Personal | 3.0 | 1.8 | 1.5 | 1.0 | 0.002 |
| Travel | 3.2 | 0.9 | 2.3 | 0.6 | 0.001 |
| Other | 6.8 | 6.0 | 4.4 | 8.4 | 0.306 |
Discussion
In 2006, O'Leary et al.1 demonstrated that academic hospitalists spend approximately 20% of their time engaged in direct patient care. Our results are consistent with these data and further expand these findings to a community setting. Although we found subtle workflow differences between the academic and community programs, their similarities were more striking than their differences. We suspect that these differences can be largely attributed to the higher CMI at the academic program as well as the greater complexity and additional communication hand‐offs inherent to this tertiary academic medical center. For example, at the academic medical center, medicine admissions were screened by a medicine triage resident and subsequently handed off to a hospitalist. In most cases, this system did not preclude the need to speak directly with the emergency department (ED) attending, adding a layer of complexity that did not exist in the community hospital. Finally, in contrast to the community hospital, there was little comanagement at the academic medical center, necessitating frequent transfers to and from medical and subspecialty services.
It appears that hospitalists, irrespective of their work environment, spend far more time documenting, communicating, and coordinating care than at the bedside. It is unclear whether this represents a desirable outcome of hospitalists' role as managers of complex hospital stays or inefficient and ineffective effort that should be mitigated through care delivery redesign. Further research to optimize hospital information management, streamline care processes and eliminate low value‐added effort is clearly needed.
Another notable finding of our study is that hospitalists spend roughly half of their time performing more than 1 work category at the same time deemed as multitasking.2 The prevalence and effects of multitasking are well‐characterized in emergency medicine and likely apply to hospitalists.3, 4 Fractured attention due to multitasking may hamper communication, jeopardize care handoffs, and increase risk for medical errors and litigation.46 While it is likely that multitasking is inherent to the practice of hospital medicine, it is unclear how this could be mitigated or better facilitated. Perhaps this could be done through structured communication and information management. This too merits further investigation.
Lastly, this study found that it takes approximately an hour of a hospitalist's time each day to manage 1 patient's care. This in and of itself, is very important from the standpoint of both billing and workload. In today's professional services fee model, there are a number of components that contribute to the level of service that a hospitalist can bill. One of those components is time, specifically the time spent counseling and/or coordinating care, which as this study suggests, dominates a hospitalist's workday. It is therefore critical that hospitalists accurately and consistently document the amount of time they spend with each patient and specifically describe the counseling and/or activities to coordinate care. Additionally, recognizing how much time is required for a hospitalist to care for a patient has important workload implications. If we assume that it takes approximately an hour per patient and a typical workday is around 11 hours after subtracting personal time, then it would be reasonable to expect that a single hospitalist should have, on average, 11 patient encounters per day. This number is, of course, completely dependent on organizational factors such as a specific hospital's support systems and the mix of admissions, follow‐ups, and discharges on that service.
Our study has several limitations. The time study occurred at 2 hospitals, in 1 mid‐sized Midwestern city, and the results may not be generalizable to other settings. However, the congruence of our findings with those of O'Leary et al.1 suggests that our results maintain external validity. Second, at the time of the study the 2 programs were relatively new and workflows were still evolving. Additionally, the academic and community hospitalist programs were under unified management and 2 of the surveyed hospitalists worked at both programs. This may have artificially homogenized the work patterns observed at both programs. Finally, observing hospitalist activities exclusively during the weekday daytime shifts has the potential to bias the results. However, the night and weekend duties and responsibilities of the 2 programs differed significantly, which would have made it very difficult to derive meaningful comparisons for those observations.
Conclusion
We found that hospitalists in both academic and community settings spend the majority of their time multitasking and engaged in indirect patient care. Further studies are necessary to determine the extent to which this is a necessary feature of the hospitalist care model and whether hospitalists should restructure their workflow to improve outcomes.
- , , .How hospitalists spend their time: Insights on efficiency and safety.J Hosp Med.2006;1:88–93.
- , , , .Emergency department workplace interruptions: Are emergency physicians “interrupt‐driven” and “multitasking”?Acad Emerg Med.2008;7:1239–1243.
- , , , .Work interrupted: a comparison of workplace interruptions in the emergency departments and primary care offices.Ann Emerg Med.2001;38:146–151.
- , , , , , .The multitasking clinician: Decision‐making and cognitive demand during and after team handoffs in emergency care.Int J Med Inform.2007;76:801–811.
- , , , , .Communication loads on clinical staff in the emergency department.Med J Aust.2002;176:415–418.
- , , .Why do people sue doctors? A study of patients and relatives taking legal action.Lancet.1994;343:1609–1613.
- , , .How hospitalists spend their time: Insights on efficiency and safety.J Hosp Med.2006;1:88–93.
- , , , .Emergency department workplace interruptions: Are emergency physicians “interrupt‐driven” and “multitasking”?Acad Emerg Med.2008;7:1239–1243.
- , , , .Work interrupted: a comparison of workplace interruptions in the emergency departments and primary care offices.Ann Emerg Med.2001;38:146–151.
- , , , , , .The multitasking clinician: Decision‐making and cognitive demand during and after team handoffs in emergency care.Int J Med Inform.2007;76:801–811.
- , , , , .Communication loads on clinical staff in the emergency department.Med J Aust.2002;176:415–418.
- , , .Why do people sue doctors? A study of patients and relatives taking legal action.Lancet.1994;343:1609–1613.
Unscripted
A 58‐year old man was admitted with generalized weakness and acute deep venous thrombosis (DVT). His past medical history included hypertension and polymyositis/dermatomyositis (PM/DM) with anti‐synthase syndrome, which had been diagnosed 16 months prior when his creatine kinase (CK) was greater than 12,000 U/L. At that time he also was found to have bilateral lower extremity DVT, and had been treated with warfarin for 1 year. 10 days previously, he had been discharged after a 4‐day hospitalization for a polymyositis flare which was treated with methylprednisolone at 60 mg daily for 5 days. He was discharged home with daily prednisone until this follow‐up a week later, where he reported weakness and bilateral edema. Lower extremity ultrasound demonstrated acute thrombus in the right common femoral vein.
This acute extensive DVT may be a consequence of recent hospitalization and a previously damaged venous system, or may reflect ongoing hypercoagulability from an unresolved condition, such as cancer. Bilateral lower extremity edema may suggest right‐sided heart failure due to progressive interstitial lung disease, which occurs in a subset of patients with PM/DM. Edema may alternatively reflect biventricular heart failure, or liver or kidney disease.
Generalized weakness offers little in the way of focused differential diagnosis until it is characterized as motor weakness (eg, attributed to progression of the myopathy), a dyspnea‐equivalent, or an overall sense of fatigue.
His medications included weekly methotrexate, monthly intravenous immunoglobulin (IVIG) infusions, tacrolimus, hydrochlorothiazide, and aerosolized pentamidine. He had been on varying doses of prednisone for 2 years and his present dose was 40 mg daily. He was allergic to sulfa. He was married and stopped smoking 30 years previously, and did not drink alcohol or use illicit drugs.
Various medication toxicities could account for his presentation. Methotrexate causes interstitial lung disease, and IVIG and tacrolimus may cause renal failure (and fluid overload). The heavy degree of immunosuppression renders him susceptible to a wide range of infections. Aerosolized pentamidine provides incomplete protection against Pneumocystis jirovecii, especially in the lung apices.
Evaluation of the status of his myositis with motor strength assessment is important. In addition associated rashes and signs of malignancy (eg, lymphadenopathy) and infection should be sought. Proximal motor weakness would suggest a myositis flare, although care must be given to exclude competing causes of myopathy, including infections, toxins, or endocrinopathies.
His temperature was 36.2C, pulse 103 beats per minute, blood pressure 156/83 mm Hg, and respiratory rate 18 breaths per minute. He had crackles at both lung bases, and 3+ pitting edema in both lower extremities. On neurological exam his motor strength was found to be diminished at 3/5 in the lower extremities and proximal upper extremities and 4/5 in the distal upper extremities. Reflexes were uniformly at 1+/4 and his cognition was intact. Examinations of his head, skin, heart, and abdomen were normal.
The absence of elevated jugular venous pressure argues against right heart failure. He is afebrile but that is minimally reassuring given the immunosuppression. There are no clues to suggest liver or kidney dysfunction. An unrecognized occlusion of the lower abdominal venous or lymphatic system such as upward extension of the DVT into the inferior vena cava (IVC) or a pelvic obstruction of the lower extremity lymphatic vessels could be considered. It appears that his distal weakness closely mirrors his proximal weakness in distinction to most myopathies which are predominantly proximal (with some exceptions, eg, inclusion body myositis).
The white blood cell count was 26,000/L with normal differential, hemoglobin 11.2 gm/dL, and platelet count was 191,000/L (at recent discharge these values were 23,000, 11.9, and 274,000, respectively). Chemistries were normal except for creatinine of 1.4 mg/dL (baseline 1.2), blood urea nitrogen was 42 mg/dL, albumin 2.6 gm/dL (normal, 3.55.0), and CK 3,710 U/L (20220), decreased from 6,943 U/L at recent discharge. Urine dipstick testing was positive for blood and protein; the urine sediment was unremarkable. Chest radiograph revealed normal lungs and heart.
The white blood cell count is quite elevated, perhaps more so than could be attributed to chronic steroid use, and again raises the concern of an undiagnosed infection. The presence of heme (and protein) in the urine without cells is consistent with pigment nephropathy from the recent rhabdomyolysis.
He was admitted to the hospital. Unfractionated heparin and warfarin were started. No changes were made to his immunosuppressive regimen. Blood cultures were negative after 48 hours. Transthoracic echocardiogram showed an ejection fraction of 60%, normal valves, and right ventricular systolic pressure of 32 mm Hg (normal, 1525 mmHg). On hospital day 3, his platelet count was 147,000/L, and on day 5, 101,000/L. His other laboratory values remained unchanged, and there were no new clinical developments.
A declining platelet count and extensive deep vein thrombosis suggest heparin‐induced thrombocytopenia and thrombosis (HITT), especially with the greater than 50% drop in the setting of IV heparin. His platelets have continued on a downward trajectory that was evident at admission and has progressed during this hospitalization. Assuming this is not due to laboratory error or artifact such as platelet clumping, this decline could have occurred if he was sensitized to heparin during the prior hospitalization, such as for DVT prophylaxis. It is increasingly recognized that HITT can manifest even after exposure to heparin is complete, ie, posthospitalization, and there can be an immediate drop in platelet counts if an unrecognized HITT‐mediated thrombosis is treated with IV heparin. Heparin should be discontinued in favor of a direct thrombin inhibitor and tests for heparin‐induced platelet antibodies (HIPA) and serotonin‐release assay (SRA) sent.
Antiphospholipid antibody syndrome (APLS) is associated with hypercoagulability and thrombocytopenia and is more frequent in patients with autoimmune disorders. The drug list should also be examined for associations with thrombocytopenia. The peripheral smear should be scrutinized and hemoglobin and creatinine followed to exclude thrombotic thrombocytopenic purpura‐hemolytic uremic syndrome (TTP‐HUS).
Heparin was stopped on day 5. Warfarin was continued with a therapeutic international normalized ratio (INR). Tests for antiplatelet factor 4 antibodies, HIPA, and SRA were negative. His weakness and edema improved although his CK remained between 2000 and 4000 U/L. On day 5 he developed mild hemoptysis, and a repeat chest radiograph demonstrated a new left hilar infiltrate. Computed tomography (CT) scan of the chest with contrast demonstrated a left lower lobe consolidation, scattered ground glass opacities in both lung bases, and no pulmonary embolus. He was treated with piperacillin/tazobactam and vancomycin. He remained afebrile. The same day, he erroneously received 125 mg (instead of 12.5 mg) of subcutaneous methotrexate. High‐dose leucovorin was administered on days 5 and 6.
The hemoptysis resolved after 2 days. From days 5 to 9, the platelet count dropped to 80,000/L and his hemoglobin gradually decreased to 7.3 g/dL. Anticoagulation was stopped, vitamin K administered, and an IVC filter placed. Two units of packed red blood cells (RBCs) were transfused.
In suspected HITT (which was not verified here), warfarin is typically withheld until the platelets have recovered and thrombin‐inhibitor anticoagulation has reached a steady state, to avoid the transient hypercoagulability of warfarin initiation.
The unusual time course and the 3 negative tests make HITT unlikely. The continued platelet decline after stopping heparin further supports another etiology. The excess methotrexate dosing complicates interpretation of his thrombocytopenia and anemia, which can be explained by mucosal bleeding, microangiopathic hemolytic anemia (MAHA) such as disseminated intravascular coagulation or TTP‐HUS, or autoimmunity (Evans syndrome). Bone marrow toxicity is also a major effect of methotrexate (in addition to elevation of liver enzymes and acute renal failure); however, there is typically a lag between administration and development of cytopenias. The antibiotics could also account for the ongoing (but not original) thrombocytopenia.
With the new pulmonary infiltrate, infections remain a primary concern and should be evaluated with sputum samples and perhaps bronchoscopy. Given the abnormal urine (even without cells), a pulmonary‐renal inflammatory processes should be considered also to explain the infiltrates and hemoptysis.
Haptoglobin was 20 mg/dL (normal, 37246). The direct antiglobulin test (DAT) was negative. Serum lactate dehydrogenase (LDH) was 1657 U/L (normal, 100220), with elevated LD4 and LD5 isoenzymes. Coagulation studies normalized after the administration of vitamin K. Anti‐nuclear antibody was positive at 8.7 (normal 1.5). Tests for antineutrophil cytoplasmic antibodies were negative. No sputum could be obtained. A pathologist reviewed the blood smear and reported neutrophilic leukocytosis without left shift, and thrombocytopenia with normal platelet morphology.
Low haptoglobin in the setting of an elevated LDH is highly suggestive of hemolysis, particularly the intravascular, microangiopathic varieties. Neutrophilia may reflect infection, a primary myeloproliferative process such as chronic myeloid leukemia, steroid use, or a reactive bone marrow in the setting of acute illness. The negative DAT and significant immunosuppressive regimen makes immune‐mediated hemolysis unlikely, although the history of autoimmunity and the small DAT false‐negative rate leaves Evans syndrome as an outside possibility. Medications such as tacrolimus (causing TTP) or IVIG (given the broad spectrum of antibodies it includes) are other plausible causes of the cytopenias.
At this point, I would analyze the red blood cell (RBC) morphology and check the reticulocyte count to help differentiate between hemolysis and a myelotoxin.
After transfusion, his hemoglobin remained at approximately 8.5 gm/dL and LDH remained elevated but stable. By day 12 the platelet count had fallen to 37,000/L.
With physical therapy the patient gained strength. Antibiotics were discontinued on day 12 and a follow‐up chest x‐ray demonstrated no significant disease. From days 10 to 12, his creatinine rose from 1.5 to 1.9 mg/dL, although urine output remained normal.
A hematologist observed minimal fragmentation of red cells on the blood smear. Commenting on the thrombocytopenia, anemia, and LDH isoenzymes (representative of skeletal/hepatic origin rather than hematologic), and clinical improvement after treatment of a presumed pneumonia, he felt that the continued thrombocytopenia was likely due to drug toxicity, and recommended observation, treatment of renal failure, and discontinuation of tacrolimus.
The failure to increase the hemoglobin after transfusion is consistent with (but not specific for) hemolysis. In conjunction with the progressive thrombocytopenia and persistently elevated LDH, TTP remains a consideration. While TTP can be diagnosed with minimal evidence of schistocytes, the duration of this illness, now spanning almost 2 weeks without significant end organ damagenamely more pronounced renal failure, confusion, or feveris unusual for TTP. Therefore, I think it is reasonable to withhold plasma exchange, although if the cytopenias or renal failure progress after the methotrexate, tacrolimus, and antibiotics are stopped, it may have to be undertaken empirically.
The pulmonary process remains undefined. Edema, pneumonitis (eg, aspiration), a modest pneumonia, or pulmonary hemorrhage could normalize on chest x‐ray after 1 week.
Renal ultrasound was normal. Urinalysis dipstick demonstrated 3+ blood, 3+ protein, and no nitrate or leukocyte esterase. The urine sediment showed only granular casts. Fractional excretion of sodium was 6.7%. Urine protein‐to‐creatinine ratio was 7.5, and urine myoglobin was elevated. Serum C3 and C4 complement levels and cryoglobulins were normal. Reticulocyte count was 8.5% (normal, 0.53.2).
There is significant evidence for intrinsic renal failure, starting with the elevated fractional excretion. Marked proteinuria suggests glomerular damage; nephrotic syndrome could provide an explanation for the recurrent DVT. The 3+ blood without RBCs and the markedly elevated urine myoglobin suggest pigment nephropathy from both myoglobinuria and hemoglobinuria. The elevated reticulocyte count further confirms the impression of hemolysis.
Nephrotic syndrome may result from a primary disease process, such as diabetes, systemic lupus erythematosus (SLE), or amyloidosis, for which there is no evidence to date, or as a consequence of indolent infection, malignancy, or drugs, all of which are reasonable possibilities.
The essential elements at this point include thrombocytopenia, kidney failure with proteinuria, and likely intravascular hemolysis. I would repeat the peripheral smear (looking for schistocytes) and discuss with the rheumatologist if any other medications could be discontinued.
A nephrology consultant diagnosed acute tubular necrosis (ATN) from a combination of insults (intravenous contrast, methotrexate, tacrolimus, and myoglobinuria). Over the next several days, his platelet count rose to approximately 60,000/L. The patient continued to generally feel better but the creatinine steadily increased to 4.9 mg/dL.
The hematologist's reassessment of the smear was unchanged with minimal RBC fragmentation noted. Over the next few days the hemoglobin, creatinine, and platelet count remained stable, and there were no fevers or other clinical developments. On day 21 a kidney biopsy specimen revealed evidence of thrombotic microangiopathy (TMA) and segmental glomerular necrosis, with negative immunofluorescent findings. In addition, the glomerular basement membranes were thickened and effacement of the epithelial foot processes was noted.
TTP (or other MAHA) with only a few schistocytes would be unusual at an advanced stage where organ damage has occurred, although the clinical presentation in drug‐induced variety is variable. TTP is also generally a fatal disease, so relative stability over 3 weeks without definitive therapy is atypical, unless prednisone has served as a temporizing measure. The atypical features raise the possibility of a mimic or variant of TTP such as undiagnosed cancer causing DIC or a medication (eg, tacrolimus)‐associated TTP syndrome.
At least 2 other conditions could account for the hemolysis, thrombocytopenia, and TMA. The positive ANA, glomerular disease, and cytopenias are compatible with SLE, although such progression on an intense immunosuppressive regimen would be unusual. The renal histology in a patient with an autoimmune diathesis warrants reconsideration of antiphospholipid antibody syndrome (APLS), especially in light of the earlier DVT.
Tests for antiphospholipid antibodies were negative. After multidisciplinary deliberation, a diagnosis of TMA due to tacrolimus‐associated TTP/HUS was made. Plasmapheresis was initiated and IVIG and steroids were continued. He had a complicated hospital course and required renal replacement therapy, but with pheresis, his platelet counts and hemoglobin began to recover and he was ultimately discharged in good condition. After he was discharged, testing for ADAMTS13 (a von Willebrand factor‐cleaving protease) activity was reported as 54% (normal, >66%)
Discussion
TMA in the microcirculation is the hallmark pathology of TTP‐HUS but is not specific for this disease. TMA is also seen in disseminated intravascular coagulation, sepsis, cancer, malignant hypertension, human immunodeficiency virus infection, autoimmune disorders, pregnancy‐related conditions, and in association with certain drugs.1 The first pharmacological agent to be associated with TMA was mitomycin in 1971, and since then other drug associations have been described, including antiplatelet medications such as ticlopidine and clopidogrel, antibiotics such as quinine and rifampin, interferon, and immunosuppressants such as cyclosporine and tacrolimus.2 Drug‐induced variants of TTP and TMA are challenging to diagnose because the timing of onset, clinical features, and patient factors (eg, receipt of immunosuppressants) may vary widely and mimic other conditions.2, 3 TMA is a rare complication of tacrolimus and is mostly seen in renal transplant patients at a frequency of 1%. In these patients, renal dysfunction is usually the first herald of TMA and TTP; evidence of hemolysis may be absent.3
The clinical diagnosis of TTP has historically been based on the presence of a classic pentad: MAHA, thrombocytopenia, neurological and renal abnormalities, and fever.4 Elevated levels of LDH and indirect bilirubin and the presence of fragmented RBCs and reticulocytes point toward active intravascular hemolysis. The DAT is usually negative. This textbook illness scriptthe template of a disease that is stored in a clinician's memoryis learned by physicians during training, but undergoes little modification given the limited exposure to a rare disease.
In modern practice, the pentad is rarely seen, and the characteristics of the end‐organ findings may vary substantially. For instance, while neurological symptoms including seizures, coma, and transient confusion occur in 90% of cases, renal involvement is seen in about 50% and fever in only 25% of patients.5 Although the presence of 2 or more schistocytes on the blood smear under 100 microscopy supports the diagnosis of MAHA, cases of TTP without significant schistocytosis have been reported.6
Furthermore, TTP is typically described as acute in onset, but in a quarter of patients the symptoms and signs last for weeks before diagnosis.4 This variability in disease presentation coupled with the high mortality of untreated disease has changed the diagnostic and treatment thresholds for TTP. Trials and expert opinion use MAHA, thrombocytopenia, and the exclusion of alternative causes as sufficient criteria to diagnose TTP and begin treatment.7 The measurement of a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13 (ADAMTS13) activity (a von Willebrand factor‐cleaving protease) for diagnostic purposes remains controversial because assay techniques are not uniform and there is insufficient correlation between levels and clinical disease.810 For instance, the presence of severe ADAMTS 13 deficiency (ie, 5%) along with the presence of an ADAMTS13 inhibitor is considered to be very specific, but not sensitive, for the laboratory diagnosis of idiopathic TTP.11 In cohort studies, the frequency of severe deficiency among patients with idiopathic TTP ranged from 18% to 100%, and the presence of severe deficiency did not predict the development of acute episodes of TTP.9 In a registry study of 142 patients diagnosed with TTP, 81% of patients with secondary TTP (ie, not classified as idiopathic) had ADAMTS13 levels that were normal to subnormal (>25%), and patients with normal ADAMTS13 levels had a higher incidence of acute renal failure, similar to the findings in this patient.10
Untreated TTP has a mortality rate of greater than 90%, but with plasma exchange, survival has improved dramatically.4, 7 Glucocorticoids are often used in addition to plasma exchange, based on case series and reports.9 The addition of cryoprecipitate or fresh frozen plasma to plasmapheresis has not been shown to be beneficial, but rituximab, an anti CD‐20 monoclonal antibody, has shown promise in a small prospective study.12, 13
TTP is a rare disorder with a classic description but substantial variation in clinical presentation. In this case, the background autoimmune myopathy, immunosuppression, coincident acute DVT, unexplained infiltrates, complex medication regimen, and nephrotic range proteinuria (attributed to focal segmental glomerular sclerosis based on the limited evidence available from the biopsy) led the clinicians to ascribe the patient's thrombocytopenia and renal injury to more common conditions and created a challenging environment for the diagnosis of TTP. TTP is a complex disorder and the simplified understanding of the disease and its time course prevented a prompt match between the patient's clinical course and his diagnosis. The combination of a rare condition with inherent variability arising in the setting of medical complexity challenges the processes of problem representation and scripting the answer for even the most seasoned clinician.
The approach to clinical conundrums by an expert clinician is revealed through presentation of an actual patient's case in an approach typical of morning report. Similar to patient care, sequential pieces of information are provided to the clinician who is unfamiliar with the case. The focus is on the thought processes of both the clinical team caring for the patient and the discussant.
Key Teaching Points
-
The classically described pentad of TTP is seldom seen, and the findings of otherwise unexplained MAHA and thrombocytopenia should prompt consideration of TTP.
-
TTP may be acute and idiopathic, or be secondary to drugs, infections, or other conditions. Medication‐induced TTP may present with a wide range of clinical findings.
-
Therapeutic plasma exchange may be life‐saving in cases of TTP, and when appropriate, should be initiated promptly based on clinical suspicion and without waiting to perform tissue biopsy.
- , , .Thrombotic microangiopathies. In: Tischer CC, Brenner BM, eds.Renal Pathology.2nd ed.Philadelphia, PA:JB Lippincott;1994:1154–1184.
- , , .Drug‐induced thrombotic microangiopathy: incidence, prevention and management.Drug Saf.2001;24(7):491–501.
- , , , , .FK 506‐associated thrombotic microangiopathy: report of two cases and review of the literature.Transplantation.1999;67(4):539–544.
- , .Thrombotic Thrombocytopenic purpura: report of 16 cases and review of the literature.Medicine (Baltimore).1966;45:139–159.
- , , , .Thrombotic thrombocytopenic purpura; early and late responders.Am J Hematol.1997;54:102–107.
- .Atypical presentations of thrombotic thrombocytopenic purpura: a review.J Clin Apheresis.2009;24(1)47–52.
- , , , et al.Comparison of plasma exchange with plasma infusion in the treatment of thrombotic thrombocytopenic purpura.N Engl J Med.1991;325:393–397.
- , , , , , .The incidence of thrombotic thrombocytopenic purpura‐hemolytic uremic syndrome: all patients, idiopathic patients, and patients with severe ADAMTS13deficiency.J Thromb Haemost.2005;3:1432–1436.
- .Thrombotic thrombocytopenic purpura.N Engl J Med.2006;354:1927–1935.
- , , , et al.ADAMTS13 activity in thrombotic thrombocytopenic purpura‐hemolytic uremic syndrome: relation to presenting features and clinical outcomes in a prospective cohort of 142 patients.Blood.2003;102:60–68.
- , , .Thrombotic thrombocytopenic purpura.J Thromb Haemost.2005;3:1663–1675.
- , , , et al.Interventions for hemolytic uremic syndrome and thrombotic thrombocytopenic purpura: a systematic review of randomized controlled trials.Am J Kidney Dis.2009;53:259–272.
- , , , et al.:Efficiency of curative and prophylactic treatment with rituximab in ADAMTS13‐deficient TTP: A study of 11 cases.Blood.2005;105:1932–1937.
A 58‐year old man was admitted with generalized weakness and acute deep venous thrombosis (DVT). His past medical history included hypertension and polymyositis/dermatomyositis (PM/DM) with anti‐synthase syndrome, which had been diagnosed 16 months prior when his creatine kinase (CK) was greater than 12,000 U/L. At that time he also was found to have bilateral lower extremity DVT, and had been treated with warfarin for 1 year. 10 days previously, he had been discharged after a 4‐day hospitalization for a polymyositis flare which was treated with methylprednisolone at 60 mg daily for 5 days. He was discharged home with daily prednisone until this follow‐up a week later, where he reported weakness and bilateral edema. Lower extremity ultrasound demonstrated acute thrombus in the right common femoral vein.
This acute extensive DVT may be a consequence of recent hospitalization and a previously damaged venous system, or may reflect ongoing hypercoagulability from an unresolved condition, such as cancer. Bilateral lower extremity edema may suggest right‐sided heart failure due to progressive interstitial lung disease, which occurs in a subset of patients with PM/DM. Edema may alternatively reflect biventricular heart failure, or liver or kidney disease.
Generalized weakness offers little in the way of focused differential diagnosis until it is characterized as motor weakness (eg, attributed to progression of the myopathy), a dyspnea‐equivalent, or an overall sense of fatigue.
His medications included weekly methotrexate, monthly intravenous immunoglobulin (IVIG) infusions, tacrolimus, hydrochlorothiazide, and aerosolized pentamidine. He had been on varying doses of prednisone for 2 years and his present dose was 40 mg daily. He was allergic to sulfa. He was married and stopped smoking 30 years previously, and did not drink alcohol or use illicit drugs.
Various medication toxicities could account for his presentation. Methotrexate causes interstitial lung disease, and IVIG and tacrolimus may cause renal failure (and fluid overload). The heavy degree of immunosuppression renders him susceptible to a wide range of infections. Aerosolized pentamidine provides incomplete protection against Pneumocystis jirovecii, especially in the lung apices.
Evaluation of the status of his myositis with motor strength assessment is important. In addition associated rashes and signs of malignancy (eg, lymphadenopathy) and infection should be sought. Proximal motor weakness would suggest a myositis flare, although care must be given to exclude competing causes of myopathy, including infections, toxins, or endocrinopathies.
His temperature was 36.2C, pulse 103 beats per minute, blood pressure 156/83 mm Hg, and respiratory rate 18 breaths per minute. He had crackles at both lung bases, and 3+ pitting edema in both lower extremities. On neurological exam his motor strength was found to be diminished at 3/5 in the lower extremities and proximal upper extremities and 4/5 in the distal upper extremities. Reflexes were uniformly at 1+/4 and his cognition was intact. Examinations of his head, skin, heart, and abdomen were normal.
The absence of elevated jugular venous pressure argues against right heart failure. He is afebrile but that is minimally reassuring given the immunosuppression. There are no clues to suggest liver or kidney dysfunction. An unrecognized occlusion of the lower abdominal venous or lymphatic system such as upward extension of the DVT into the inferior vena cava (IVC) or a pelvic obstruction of the lower extremity lymphatic vessels could be considered. It appears that his distal weakness closely mirrors his proximal weakness in distinction to most myopathies which are predominantly proximal (with some exceptions, eg, inclusion body myositis).
The white blood cell count was 26,000/L with normal differential, hemoglobin 11.2 gm/dL, and platelet count was 191,000/L (at recent discharge these values were 23,000, 11.9, and 274,000, respectively). Chemistries were normal except for creatinine of 1.4 mg/dL (baseline 1.2), blood urea nitrogen was 42 mg/dL, albumin 2.6 gm/dL (normal, 3.55.0), and CK 3,710 U/L (20220), decreased from 6,943 U/L at recent discharge. Urine dipstick testing was positive for blood and protein; the urine sediment was unremarkable. Chest radiograph revealed normal lungs and heart.
The white blood cell count is quite elevated, perhaps more so than could be attributed to chronic steroid use, and again raises the concern of an undiagnosed infection. The presence of heme (and protein) in the urine without cells is consistent with pigment nephropathy from the recent rhabdomyolysis.
He was admitted to the hospital. Unfractionated heparin and warfarin were started. No changes were made to his immunosuppressive regimen. Blood cultures were negative after 48 hours. Transthoracic echocardiogram showed an ejection fraction of 60%, normal valves, and right ventricular systolic pressure of 32 mm Hg (normal, 1525 mmHg). On hospital day 3, his platelet count was 147,000/L, and on day 5, 101,000/L. His other laboratory values remained unchanged, and there were no new clinical developments.
A declining platelet count and extensive deep vein thrombosis suggest heparin‐induced thrombocytopenia and thrombosis (HITT), especially with the greater than 50% drop in the setting of IV heparin. His platelets have continued on a downward trajectory that was evident at admission and has progressed during this hospitalization. Assuming this is not due to laboratory error or artifact such as platelet clumping, this decline could have occurred if he was sensitized to heparin during the prior hospitalization, such as for DVT prophylaxis. It is increasingly recognized that HITT can manifest even after exposure to heparin is complete, ie, posthospitalization, and there can be an immediate drop in platelet counts if an unrecognized HITT‐mediated thrombosis is treated with IV heparin. Heparin should be discontinued in favor of a direct thrombin inhibitor and tests for heparin‐induced platelet antibodies (HIPA) and serotonin‐release assay (SRA) sent.
Antiphospholipid antibody syndrome (APLS) is associated with hypercoagulability and thrombocytopenia and is more frequent in patients with autoimmune disorders. The drug list should also be examined for associations with thrombocytopenia. The peripheral smear should be scrutinized and hemoglobin and creatinine followed to exclude thrombotic thrombocytopenic purpura‐hemolytic uremic syndrome (TTP‐HUS).
Heparin was stopped on day 5. Warfarin was continued with a therapeutic international normalized ratio (INR). Tests for antiplatelet factor 4 antibodies, HIPA, and SRA were negative. His weakness and edema improved although his CK remained between 2000 and 4000 U/L. On day 5 he developed mild hemoptysis, and a repeat chest radiograph demonstrated a new left hilar infiltrate. Computed tomography (CT) scan of the chest with contrast demonstrated a left lower lobe consolidation, scattered ground glass opacities in both lung bases, and no pulmonary embolus. He was treated with piperacillin/tazobactam and vancomycin. He remained afebrile. The same day, he erroneously received 125 mg (instead of 12.5 mg) of subcutaneous methotrexate. High‐dose leucovorin was administered on days 5 and 6.
The hemoptysis resolved after 2 days. From days 5 to 9, the platelet count dropped to 80,000/L and his hemoglobin gradually decreased to 7.3 g/dL. Anticoagulation was stopped, vitamin K administered, and an IVC filter placed. Two units of packed red blood cells (RBCs) were transfused.
In suspected HITT (which was not verified here), warfarin is typically withheld until the platelets have recovered and thrombin‐inhibitor anticoagulation has reached a steady state, to avoid the transient hypercoagulability of warfarin initiation.
The unusual time course and the 3 negative tests make HITT unlikely. The continued platelet decline after stopping heparin further supports another etiology. The excess methotrexate dosing complicates interpretation of his thrombocytopenia and anemia, which can be explained by mucosal bleeding, microangiopathic hemolytic anemia (MAHA) such as disseminated intravascular coagulation or TTP‐HUS, or autoimmunity (Evans syndrome). Bone marrow toxicity is also a major effect of methotrexate (in addition to elevation of liver enzymes and acute renal failure); however, there is typically a lag between administration and development of cytopenias. The antibiotics could also account for the ongoing (but not original) thrombocytopenia.
With the new pulmonary infiltrate, infections remain a primary concern and should be evaluated with sputum samples and perhaps bronchoscopy. Given the abnormal urine (even without cells), a pulmonary‐renal inflammatory processes should be considered also to explain the infiltrates and hemoptysis.
Haptoglobin was 20 mg/dL (normal, 37246). The direct antiglobulin test (DAT) was negative. Serum lactate dehydrogenase (LDH) was 1657 U/L (normal, 100220), with elevated LD4 and LD5 isoenzymes. Coagulation studies normalized after the administration of vitamin K. Anti‐nuclear antibody was positive at 8.7 (normal 1.5). Tests for antineutrophil cytoplasmic antibodies were negative. No sputum could be obtained. A pathologist reviewed the blood smear and reported neutrophilic leukocytosis without left shift, and thrombocytopenia with normal platelet morphology.
Low haptoglobin in the setting of an elevated LDH is highly suggestive of hemolysis, particularly the intravascular, microangiopathic varieties. Neutrophilia may reflect infection, a primary myeloproliferative process such as chronic myeloid leukemia, steroid use, or a reactive bone marrow in the setting of acute illness. The negative DAT and significant immunosuppressive regimen makes immune‐mediated hemolysis unlikely, although the history of autoimmunity and the small DAT false‐negative rate leaves Evans syndrome as an outside possibility. Medications such as tacrolimus (causing TTP) or IVIG (given the broad spectrum of antibodies it includes) are other plausible causes of the cytopenias.
At this point, I would analyze the red blood cell (RBC) morphology and check the reticulocyte count to help differentiate between hemolysis and a myelotoxin.
After transfusion, his hemoglobin remained at approximately 8.5 gm/dL and LDH remained elevated but stable. By day 12 the platelet count had fallen to 37,000/L.
With physical therapy the patient gained strength. Antibiotics were discontinued on day 12 and a follow‐up chest x‐ray demonstrated no significant disease. From days 10 to 12, his creatinine rose from 1.5 to 1.9 mg/dL, although urine output remained normal.
A hematologist observed minimal fragmentation of red cells on the blood smear. Commenting on the thrombocytopenia, anemia, and LDH isoenzymes (representative of skeletal/hepatic origin rather than hematologic), and clinical improvement after treatment of a presumed pneumonia, he felt that the continued thrombocytopenia was likely due to drug toxicity, and recommended observation, treatment of renal failure, and discontinuation of tacrolimus.
The failure to increase the hemoglobin after transfusion is consistent with (but not specific for) hemolysis. In conjunction with the progressive thrombocytopenia and persistently elevated LDH, TTP remains a consideration. While TTP can be diagnosed with minimal evidence of schistocytes, the duration of this illness, now spanning almost 2 weeks without significant end organ damagenamely more pronounced renal failure, confusion, or feveris unusual for TTP. Therefore, I think it is reasonable to withhold plasma exchange, although if the cytopenias or renal failure progress after the methotrexate, tacrolimus, and antibiotics are stopped, it may have to be undertaken empirically.
The pulmonary process remains undefined. Edema, pneumonitis (eg, aspiration), a modest pneumonia, or pulmonary hemorrhage could normalize on chest x‐ray after 1 week.
Renal ultrasound was normal. Urinalysis dipstick demonstrated 3+ blood, 3+ protein, and no nitrate or leukocyte esterase. The urine sediment showed only granular casts. Fractional excretion of sodium was 6.7%. Urine protein‐to‐creatinine ratio was 7.5, and urine myoglobin was elevated. Serum C3 and C4 complement levels and cryoglobulins were normal. Reticulocyte count was 8.5% (normal, 0.53.2).
There is significant evidence for intrinsic renal failure, starting with the elevated fractional excretion. Marked proteinuria suggests glomerular damage; nephrotic syndrome could provide an explanation for the recurrent DVT. The 3+ blood without RBCs and the markedly elevated urine myoglobin suggest pigment nephropathy from both myoglobinuria and hemoglobinuria. The elevated reticulocyte count further confirms the impression of hemolysis.
Nephrotic syndrome may result from a primary disease process, such as diabetes, systemic lupus erythematosus (SLE), or amyloidosis, for which there is no evidence to date, or as a consequence of indolent infection, malignancy, or drugs, all of which are reasonable possibilities.
The essential elements at this point include thrombocytopenia, kidney failure with proteinuria, and likely intravascular hemolysis. I would repeat the peripheral smear (looking for schistocytes) and discuss with the rheumatologist if any other medications could be discontinued.
A nephrology consultant diagnosed acute tubular necrosis (ATN) from a combination of insults (intravenous contrast, methotrexate, tacrolimus, and myoglobinuria). Over the next several days, his platelet count rose to approximately 60,000/L. The patient continued to generally feel better but the creatinine steadily increased to 4.9 mg/dL.
The hematologist's reassessment of the smear was unchanged with minimal RBC fragmentation noted. Over the next few days the hemoglobin, creatinine, and platelet count remained stable, and there were no fevers or other clinical developments. On day 21 a kidney biopsy specimen revealed evidence of thrombotic microangiopathy (TMA) and segmental glomerular necrosis, with negative immunofluorescent findings. In addition, the glomerular basement membranes were thickened and effacement of the epithelial foot processes was noted.
TTP (or other MAHA) with only a few schistocytes would be unusual at an advanced stage where organ damage has occurred, although the clinical presentation in drug‐induced variety is variable. TTP is also generally a fatal disease, so relative stability over 3 weeks without definitive therapy is atypical, unless prednisone has served as a temporizing measure. The atypical features raise the possibility of a mimic or variant of TTP such as undiagnosed cancer causing DIC or a medication (eg, tacrolimus)‐associated TTP syndrome.
At least 2 other conditions could account for the hemolysis, thrombocytopenia, and TMA. The positive ANA, glomerular disease, and cytopenias are compatible with SLE, although such progression on an intense immunosuppressive regimen would be unusual. The renal histology in a patient with an autoimmune diathesis warrants reconsideration of antiphospholipid antibody syndrome (APLS), especially in light of the earlier DVT.
Tests for antiphospholipid antibodies were negative. After multidisciplinary deliberation, a diagnosis of TMA due to tacrolimus‐associated TTP/HUS was made. Plasmapheresis was initiated and IVIG and steroids were continued. He had a complicated hospital course and required renal replacement therapy, but with pheresis, his platelet counts and hemoglobin began to recover and he was ultimately discharged in good condition. After he was discharged, testing for ADAMTS13 (a von Willebrand factor‐cleaving protease) activity was reported as 54% (normal, >66%)
Discussion
TMA in the microcirculation is the hallmark pathology of TTP‐HUS but is not specific for this disease. TMA is also seen in disseminated intravascular coagulation, sepsis, cancer, malignant hypertension, human immunodeficiency virus infection, autoimmune disorders, pregnancy‐related conditions, and in association with certain drugs.1 The first pharmacological agent to be associated with TMA was mitomycin in 1971, and since then other drug associations have been described, including antiplatelet medications such as ticlopidine and clopidogrel, antibiotics such as quinine and rifampin, interferon, and immunosuppressants such as cyclosporine and tacrolimus.2 Drug‐induced variants of TTP and TMA are challenging to diagnose because the timing of onset, clinical features, and patient factors (eg, receipt of immunosuppressants) may vary widely and mimic other conditions.2, 3 TMA is a rare complication of tacrolimus and is mostly seen in renal transplant patients at a frequency of 1%. In these patients, renal dysfunction is usually the first herald of TMA and TTP; evidence of hemolysis may be absent.3
The clinical diagnosis of TTP has historically been based on the presence of a classic pentad: MAHA, thrombocytopenia, neurological and renal abnormalities, and fever.4 Elevated levels of LDH and indirect bilirubin and the presence of fragmented RBCs and reticulocytes point toward active intravascular hemolysis. The DAT is usually negative. This textbook illness scriptthe template of a disease that is stored in a clinician's memoryis learned by physicians during training, but undergoes little modification given the limited exposure to a rare disease.
In modern practice, the pentad is rarely seen, and the characteristics of the end‐organ findings may vary substantially. For instance, while neurological symptoms including seizures, coma, and transient confusion occur in 90% of cases, renal involvement is seen in about 50% and fever in only 25% of patients.5 Although the presence of 2 or more schistocytes on the blood smear under 100 microscopy supports the diagnosis of MAHA, cases of TTP without significant schistocytosis have been reported.6
Furthermore, TTP is typically described as acute in onset, but in a quarter of patients the symptoms and signs last for weeks before diagnosis.4 This variability in disease presentation coupled with the high mortality of untreated disease has changed the diagnostic and treatment thresholds for TTP. Trials and expert opinion use MAHA, thrombocytopenia, and the exclusion of alternative causes as sufficient criteria to diagnose TTP and begin treatment.7 The measurement of a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13 (ADAMTS13) activity (a von Willebrand factor‐cleaving protease) for diagnostic purposes remains controversial because assay techniques are not uniform and there is insufficient correlation between levels and clinical disease.810 For instance, the presence of severe ADAMTS 13 deficiency (ie, 5%) along with the presence of an ADAMTS13 inhibitor is considered to be very specific, but not sensitive, for the laboratory diagnosis of idiopathic TTP.11 In cohort studies, the frequency of severe deficiency among patients with idiopathic TTP ranged from 18% to 100%, and the presence of severe deficiency did not predict the development of acute episodes of TTP.9 In a registry study of 142 patients diagnosed with TTP, 81% of patients with secondary TTP (ie, not classified as idiopathic) had ADAMTS13 levels that were normal to subnormal (>25%), and patients with normal ADAMTS13 levels had a higher incidence of acute renal failure, similar to the findings in this patient.10
Untreated TTP has a mortality rate of greater than 90%, but with plasma exchange, survival has improved dramatically.4, 7 Glucocorticoids are often used in addition to plasma exchange, based on case series and reports.9 The addition of cryoprecipitate or fresh frozen plasma to plasmapheresis has not been shown to be beneficial, but rituximab, an anti CD‐20 monoclonal antibody, has shown promise in a small prospective study.12, 13
TTP is a rare disorder with a classic description but substantial variation in clinical presentation. In this case, the background autoimmune myopathy, immunosuppression, coincident acute DVT, unexplained infiltrates, complex medication regimen, and nephrotic range proteinuria (attributed to focal segmental glomerular sclerosis based on the limited evidence available from the biopsy) led the clinicians to ascribe the patient's thrombocytopenia and renal injury to more common conditions and created a challenging environment for the diagnosis of TTP. TTP is a complex disorder and the simplified understanding of the disease and its time course prevented a prompt match between the patient's clinical course and his diagnosis. The combination of a rare condition with inherent variability arising in the setting of medical complexity challenges the processes of problem representation and scripting the answer for even the most seasoned clinician.
The approach to clinical conundrums by an expert clinician is revealed through presentation of an actual patient's case in an approach typical of morning report. Similar to patient care, sequential pieces of information are provided to the clinician who is unfamiliar with the case. The focus is on the thought processes of both the clinical team caring for the patient and the discussant.
Key Teaching Points
-
The classically described pentad of TTP is seldom seen, and the findings of otherwise unexplained MAHA and thrombocytopenia should prompt consideration of TTP.
-
TTP may be acute and idiopathic, or be secondary to drugs, infections, or other conditions. Medication‐induced TTP may present with a wide range of clinical findings.
-
Therapeutic plasma exchange may be life‐saving in cases of TTP, and when appropriate, should be initiated promptly based on clinical suspicion and without waiting to perform tissue biopsy.
A 58‐year old man was admitted with generalized weakness and acute deep venous thrombosis (DVT). His past medical history included hypertension and polymyositis/dermatomyositis (PM/DM) with anti‐synthase syndrome, which had been diagnosed 16 months prior when his creatine kinase (CK) was greater than 12,000 U/L. At that time he also was found to have bilateral lower extremity DVT, and had been treated with warfarin for 1 year. 10 days previously, he had been discharged after a 4‐day hospitalization for a polymyositis flare which was treated with methylprednisolone at 60 mg daily for 5 days. He was discharged home with daily prednisone until this follow‐up a week later, where he reported weakness and bilateral edema. Lower extremity ultrasound demonstrated acute thrombus in the right common femoral vein.
This acute extensive DVT may be a consequence of recent hospitalization and a previously damaged venous system, or may reflect ongoing hypercoagulability from an unresolved condition, such as cancer. Bilateral lower extremity edema may suggest right‐sided heart failure due to progressive interstitial lung disease, which occurs in a subset of patients with PM/DM. Edema may alternatively reflect biventricular heart failure, or liver or kidney disease.
Generalized weakness offers little in the way of focused differential diagnosis until it is characterized as motor weakness (eg, attributed to progression of the myopathy), a dyspnea‐equivalent, or an overall sense of fatigue.
His medications included weekly methotrexate, monthly intravenous immunoglobulin (IVIG) infusions, tacrolimus, hydrochlorothiazide, and aerosolized pentamidine. He had been on varying doses of prednisone for 2 years and his present dose was 40 mg daily. He was allergic to sulfa. He was married and stopped smoking 30 years previously, and did not drink alcohol or use illicit drugs.
Various medication toxicities could account for his presentation. Methotrexate causes interstitial lung disease, and IVIG and tacrolimus may cause renal failure (and fluid overload). The heavy degree of immunosuppression renders him susceptible to a wide range of infections. Aerosolized pentamidine provides incomplete protection against Pneumocystis jirovecii, especially in the lung apices.
Evaluation of the status of his myositis with motor strength assessment is important. In addition associated rashes and signs of malignancy (eg, lymphadenopathy) and infection should be sought. Proximal motor weakness would suggest a myositis flare, although care must be given to exclude competing causes of myopathy, including infections, toxins, or endocrinopathies.
His temperature was 36.2C, pulse 103 beats per minute, blood pressure 156/83 mm Hg, and respiratory rate 18 breaths per minute. He had crackles at both lung bases, and 3+ pitting edema in both lower extremities. On neurological exam his motor strength was found to be diminished at 3/5 in the lower extremities and proximal upper extremities and 4/5 in the distal upper extremities. Reflexes were uniformly at 1+/4 and his cognition was intact. Examinations of his head, skin, heart, and abdomen were normal.
The absence of elevated jugular venous pressure argues against right heart failure. He is afebrile but that is minimally reassuring given the immunosuppression. There are no clues to suggest liver or kidney dysfunction. An unrecognized occlusion of the lower abdominal venous or lymphatic system such as upward extension of the DVT into the inferior vena cava (IVC) or a pelvic obstruction of the lower extremity lymphatic vessels could be considered. It appears that his distal weakness closely mirrors his proximal weakness in distinction to most myopathies which are predominantly proximal (with some exceptions, eg, inclusion body myositis).
The white blood cell count was 26,000/L with normal differential, hemoglobin 11.2 gm/dL, and platelet count was 191,000/L (at recent discharge these values were 23,000, 11.9, and 274,000, respectively). Chemistries were normal except for creatinine of 1.4 mg/dL (baseline 1.2), blood urea nitrogen was 42 mg/dL, albumin 2.6 gm/dL (normal, 3.55.0), and CK 3,710 U/L (20220), decreased from 6,943 U/L at recent discharge. Urine dipstick testing was positive for blood and protein; the urine sediment was unremarkable. Chest radiograph revealed normal lungs and heart.
The white blood cell count is quite elevated, perhaps more so than could be attributed to chronic steroid use, and again raises the concern of an undiagnosed infection. The presence of heme (and protein) in the urine without cells is consistent with pigment nephropathy from the recent rhabdomyolysis.
He was admitted to the hospital. Unfractionated heparin and warfarin were started. No changes were made to his immunosuppressive regimen. Blood cultures were negative after 48 hours. Transthoracic echocardiogram showed an ejection fraction of 60%, normal valves, and right ventricular systolic pressure of 32 mm Hg (normal, 1525 mmHg). On hospital day 3, his platelet count was 147,000/L, and on day 5, 101,000/L. His other laboratory values remained unchanged, and there were no new clinical developments.
A declining platelet count and extensive deep vein thrombosis suggest heparin‐induced thrombocytopenia and thrombosis (HITT), especially with the greater than 50% drop in the setting of IV heparin. His platelets have continued on a downward trajectory that was evident at admission and has progressed during this hospitalization. Assuming this is not due to laboratory error or artifact such as platelet clumping, this decline could have occurred if he was sensitized to heparin during the prior hospitalization, such as for DVT prophylaxis. It is increasingly recognized that HITT can manifest even after exposure to heparin is complete, ie, posthospitalization, and there can be an immediate drop in platelet counts if an unrecognized HITT‐mediated thrombosis is treated with IV heparin. Heparin should be discontinued in favor of a direct thrombin inhibitor and tests for heparin‐induced platelet antibodies (HIPA) and serotonin‐release assay (SRA) sent.
Antiphospholipid antibody syndrome (APLS) is associated with hypercoagulability and thrombocytopenia and is more frequent in patients with autoimmune disorders. The drug list should also be examined for associations with thrombocytopenia. The peripheral smear should be scrutinized and hemoglobin and creatinine followed to exclude thrombotic thrombocytopenic purpura‐hemolytic uremic syndrome (TTP‐HUS).
Heparin was stopped on day 5. Warfarin was continued with a therapeutic international normalized ratio (INR). Tests for antiplatelet factor 4 antibodies, HIPA, and SRA were negative. His weakness and edema improved although his CK remained between 2000 and 4000 U/L. On day 5 he developed mild hemoptysis, and a repeat chest radiograph demonstrated a new left hilar infiltrate. Computed tomography (CT) scan of the chest with contrast demonstrated a left lower lobe consolidation, scattered ground glass opacities in both lung bases, and no pulmonary embolus. He was treated with piperacillin/tazobactam and vancomycin. He remained afebrile. The same day, he erroneously received 125 mg (instead of 12.5 mg) of subcutaneous methotrexate. High‐dose leucovorin was administered on days 5 and 6.
The hemoptysis resolved after 2 days. From days 5 to 9, the platelet count dropped to 80,000/L and his hemoglobin gradually decreased to 7.3 g/dL. Anticoagulation was stopped, vitamin K administered, and an IVC filter placed. Two units of packed red blood cells (RBCs) were transfused.
In suspected HITT (which was not verified here), warfarin is typically withheld until the platelets have recovered and thrombin‐inhibitor anticoagulation has reached a steady state, to avoid the transient hypercoagulability of warfarin initiation.
The unusual time course and the 3 negative tests make HITT unlikely. The continued platelet decline after stopping heparin further supports another etiology. The excess methotrexate dosing complicates interpretation of his thrombocytopenia and anemia, which can be explained by mucosal bleeding, microangiopathic hemolytic anemia (MAHA) such as disseminated intravascular coagulation or TTP‐HUS, or autoimmunity (Evans syndrome). Bone marrow toxicity is also a major effect of methotrexate (in addition to elevation of liver enzymes and acute renal failure); however, there is typically a lag between administration and development of cytopenias. The antibiotics could also account for the ongoing (but not original) thrombocytopenia.
With the new pulmonary infiltrate, infections remain a primary concern and should be evaluated with sputum samples and perhaps bronchoscopy. Given the abnormal urine (even without cells), a pulmonary‐renal inflammatory processes should be considered also to explain the infiltrates and hemoptysis.
Haptoglobin was 20 mg/dL (normal, 37246). The direct antiglobulin test (DAT) was negative. Serum lactate dehydrogenase (LDH) was 1657 U/L (normal, 100220), with elevated LD4 and LD5 isoenzymes. Coagulation studies normalized after the administration of vitamin K. Anti‐nuclear antibody was positive at 8.7 (normal 1.5). Tests for antineutrophil cytoplasmic antibodies were negative. No sputum could be obtained. A pathologist reviewed the blood smear and reported neutrophilic leukocytosis without left shift, and thrombocytopenia with normal platelet morphology.
Low haptoglobin in the setting of an elevated LDH is highly suggestive of hemolysis, particularly the intravascular, microangiopathic varieties. Neutrophilia may reflect infection, a primary myeloproliferative process such as chronic myeloid leukemia, steroid use, or a reactive bone marrow in the setting of acute illness. The negative DAT and significant immunosuppressive regimen makes immune‐mediated hemolysis unlikely, although the history of autoimmunity and the small DAT false‐negative rate leaves Evans syndrome as an outside possibility. Medications such as tacrolimus (causing TTP) or IVIG (given the broad spectrum of antibodies it includes) are other plausible causes of the cytopenias.
At this point, I would analyze the red blood cell (RBC) morphology and check the reticulocyte count to help differentiate between hemolysis and a myelotoxin.
After transfusion, his hemoglobin remained at approximately 8.5 gm/dL and LDH remained elevated but stable. By day 12 the platelet count had fallen to 37,000/L.
With physical therapy the patient gained strength. Antibiotics were discontinued on day 12 and a follow‐up chest x‐ray demonstrated no significant disease. From days 10 to 12, his creatinine rose from 1.5 to 1.9 mg/dL, although urine output remained normal.
A hematologist observed minimal fragmentation of red cells on the blood smear. Commenting on the thrombocytopenia, anemia, and LDH isoenzymes (representative of skeletal/hepatic origin rather than hematologic), and clinical improvement after treatment of a presumed pneumonia, he felt that the continued thrombocytopenia was likely due to drug toxicity, and recommended observation, treatment of renal failure, and discontinuation of tacrolimus.
The failure to increase the hemoglobin after transfusion is consistent with (but not specific for) hemolysis. In conjunction with the progressive thrombocytopenia and persistently elevated LDH, TTP remains a consideration. While TTP can be diagnosed with minimal evidence of schistocytes, the duration of this illness, now spanning almost 2 weeks without significant end organ damagenamely more pronounced renal failure, confusion, or feveris unusual for TTP. Therefore, I think it is reasonable to withhold plasma exchange, although if the cytopenias or renal failure progress after the methotrexate, tacrolimus, and antibiotics are stopped, it may have to be undertaken empirically.
The pulmonary process remains undefined. Edema, pneumonitis (eg, aspiration), a modest pneumonia, or pulmonary hemorrhage could normalize on chest x‐ray after 1 week.
Renal ultrasound was normal. Urinalysis dipstick demonstrated 3+ blood, 3+ protein, and no nitrate or leukocyte esterase. The urine sediment showed only granular casts. Fractional excretion of sodium was 6.7%. Urine protein‐to‐creatinine ratio was 7.5, and urine myoglobin was elevated. Serum C3 and C4 complement levels and cryoglobulins were normal. Reticulocyte count was 8.5% (normal, 0.53.2).
There is significant evidence for intrinsic renal failure, starting with the elevated fractional excretion. Marked proteinuria suggests glomerular damage; nephrotic syndrome could provide an explanation for the recurrent DVT. The 3+ blood without RBCs and the markedly elevated urine myoglobin suggest pigment nephropathy from both myoglobinuria and hemoglobinuria. The elevated reticulocyte count further confirms the impression of hemolysis.
Nephrotic syndrome may result from a primary disease process, such as diabetes, systemic lupus erythematosus (SLE), or amyloidosis, for which there is no evidence to date, or as a consequence of indolent infection, malignancy, or drugs, all of which are reasonable possibilities.
The essential elements at this point include thrombocytopenia, kidney failure with proteinuria, and likely intravascular hemolysis. I would repeat the peripheral smear (looking for schistocytes) and discuss with the rheumatologist if any other medications could be discontinued.
A nephrology consultant diagnosed acute tubular necrosis (ATN) from a combination of insults (intravenous contrast, methotrexate, tacrolimus, and myoglobinuria). Over the next several days, his platelet count rose to approximately 60,000/L. The patient continued to generally feel better but the creatinine steadily increased to 4.9 mg/dL.
The hematologist's reassessment of the smear was unchanged with minimal RBC fragmentation noted. Over the next few days the hemoglobin, creatinine, and platelet count remained stable, and there were no fevers or other clinical developments. On day 21 a kidney biopsy specimen revealed evidence of thrombotic microangiopathy (TMA) and segmental glomerular necrosis, with negative immunofluorescent findings. In addition, the glomerular basement membranes were thickened and effacement of the epithelial foot processes was noted.
TTP (or other MAHA) with only a few schistocytes would be unusual at an advanced stage where organ damage has occurred, although the clinical presentation in drug‐induced variety is variable. TTP is also generally a fatal disease, so relative stability over 3 weeks without definitive therapy is atypical, unless prednisone has served as a temporizing measure. The atypical features raise the possibility of a mimic or variant of TTP such as undiagnosed cancer causing DIC or a medication (eg, tacrolimus)‐associated TTP syndrome.
At least 2 other conditions could account for the hemolysis, thrombocytopenia, and TMA. The positive ANA, glomerular disease, and cytopenias are compatible with SLE, although such progression on an intense immunosuppressive regimen would be unusual. The renal histology in a patient with an autoimmune diathesis warrants reconsideration of antiphospholipid antibody syndrome (APLS), especially in light of the earlier DVT.
Tests for antiphospholipid antibodies were negative. After multidisciplinary deliberation, a diagnosis of TMA due to tacrolimus‐associated TTP/HUS was made. Plasmapheresis was initiated and IVIG and steroids were continued. He had a complicated hospital course and required renal replacement therapy, but with pheresis, his platelet counts and hemoglobin began to recover and he was ultimately discharged in good condition. After he was discharged, testing for ADAMTS13 (a von Willebrand factor‐cleaving protease) activity was reported as 54% (normal, >66%)
Discussion
TMA in the microcirculation is the hallmark pathology of TTP‐HUS but is not specific for this disease. TMA is also seen in disseminated intravascular coagulation, sepsis, cancer, malignant hypertension, human immunodeficiency virus infection, autoimmune disorders, pregnancy‐related conditions, and in association with certain drugs.1 The first pharmacological agent to be associated with TMA was mitomycin in 1971, and since then other drug associations have been described, including antiplatelet medications such as ticlopidine and clopidogrel, antibiotics such as quinine and rifampin, interferon, and immunosuppressants such as cyclosporine and tacrolimus.2 Drug‐induced variants of TTP and TMA are challenging to diagnose because the timing of onset, clinical features, and patient factors (eg, receipt of immunosuppressants) may vary widely and mimic other conditions.2, 3 TMA is a rare complication of tacrolimus and is mostly seen in renal transplant patients at a frequency of 1%. In these patients, renal dysfunction is usually the first herald of TMA and TTP; evidence of hemolysis may be absent.3
The clinical diagnosis of TTP has historically been based on the presence of a classic pentad: MAHA, thrombocytopenia, neurological and renal abnormalities, and fever.4 Elevated levels of LDH and indirect bilirubin and the presence of fragmented RBCs and reticulocytes point toward active intravascular hemolysis. The DAT is usually negative. This textbook illness scriptthe template of a disease that is stored in a clinician's memoryis learned by physicians during training, but undergoes little modification given the limited exposure to a rare disease.
In modern practice, the pentad is rarely seen, and the characteristics of the end‐organ findings may vary substantially. For instance, while neurological symptoms including seizures, coma, and transient confusion occur in 90% of cases, renal involvement is seen in about 50% and fever in only 25% of patients.5 Although the presence of 2 or more schistocytes on the blood smear under 100 microscopy supports the diagnosis of MAHA, cases of TTP without significant schistocytosis have been reported.6
Furthermore, TTP is typically described as acute in onset, but in a quarter of patients the symptoms and signs last for weeks before diagnosis.4 This variability in disease presentation coupled with the high mortality of untreated disease has changed the diagnostic and treatment thresholds for TTP. Trials and expert opinion use MAHA, thrombocytopenia, and the exclusion of alternative causes as sufficient criteria to diagnose TTP and begin treatment.7 The measurement of a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13 (ADAMTS13) activity (a von Willebrand factor‐cleaving protease) for diagnostic purposes remains controversial because assay techniques are not uniform and there is insufficient correlation between levels and clinical disease.810 For instance, the presence of severe ADAMTS 13 deficiency (ie, 5%) along with the presence of an ADAMTS13 inhibitor is considered to be very specific, but not sensitive, for the laboratory diagnosis of idiopathic TTP.11 In cohort studies, the frequency of severe deficiency among patients with idiopathic TTP ranged from 18% to 100%, and the presence of severe deficiency did not predict the development of acute episodes of TTP.9 In a registry study of 142 patients diagnosed with TTP, 81% of patients with secondary TTP (ie, not classified as idiopathic) had ADAMTS13 levels that were normal to subnormal (>25%), and patients with normal ADAMTS13 levels had a higher incidence of acute renal failure, similar to the findings in this patient.10
Untreated TTP has a mortality rate of greater than 90%, but with plasma exchange, survival has improved dramatically.4, 7 Glucocorticoids are often used in addition to plasma exchange, based on case series and reports.9 The addition of cryoprecipitate or fresh frozen plasma to plasmapheresis has not been shown to be beneficial, but rituximab, an anti CD‐20 monoclonal antibody, has shown promise in a small prospective study.12, 13
TTP is a rare disorder with a classic description but substantial variation in clinical presentation. In this case, the background autoimmune myopathy, immunosuppression, coincident acute DVT, unexplained infiltrates, complex medication regimen, and nephrotic range proteinuria (attributed to focal segmental glomerular sclerosis based on the limited evidence available from the biopsy) led the clinicians to ascribe the patient's thrombocytopenia and renal injury to more common conditions and created a challenging environment for the diagnosis of TTP. TTP is a complex disorder and the simplified understanding of the disease and its time course prevented a prompt match between the patient's clinical course and his diagnosis. The combination of a rare condition with inherent variability arising in the setting of medical complexity challenges the processes of problem representation and scripting the answer for even the most seasoned clinician.
The approach to clinical conundrums by an expert clinician is revealed through presentation of an actual patient's case in an approach typical of morning report. Similar to patient care, sequential pieces of information are provided to the clinician who is unfamiliar with the case. The focus is on the thought processes of both the clinical team caring for the patient and the discussant.
Key Teaching Points
-
The classically described pentad of TTP is seldom seen, and the findings of otherwise unexplained MAHA and thrombocytopenia should prompt consideration of TTP.
-
TTP may be acute and idiopathic, or be secondary to drugs, infections, or other conditions. Medication‐induced TTP may present with a wide range of clinical findings.
-
Therapeutic plasma exchange may be life‐saving in cases of TTP, and when appropriate, should be initiated promptly based on clinical suspicion and without waiting to perform tissue biopsy.
- , , .Thrombotic microangiopathies. In: Tischer CC, Brenner BM, eds.Renal Pathology.2nd ed.Philadelphia, PA:JB Lippincott;1994:1154–1184.
- , , .Drug‐induced thrombotic microangiopathy: incidence, prevention and management.Drug Saf.2001;24(7):491–501.
- , , , , .FK 506‐associated thrombotic microangiopathy: report of two cases and review of the literature.Transplantation.1999;67(4):539–544.
- , .Thrombotic Thrombocytopenic purpura: report of 16 cases and review of the literature.Medicine (Baltimore).1966;45:139–159.
- , , , .Thrombotic thrombocytopenic purpura; early and late responders.Am J Hematol.1997;54:102–107.
- .Atypical presentations of thrombotic thrombocytopenic purpura: a review.J Clin Apheresis.2009;24(1)47–52.
- , , , et al.Comparison of plasma exchange with plasma infusion in the treatment of thrombotic thrombocytopenic purpura.N Engl J Med.1991;325:393–397.
- , , , , , .The incidence of thrombotic thrombocytopenic purpura‐hemolytic uremic syndrome: all patients, idiopathic patients, and patients with severe ADAMTS13deficiency.J Thromb Haemost.2005;3:1432–1436.
- .Thrombotic thrombocytopenic purpura.N Engl J Med.2006;354:1927–1935.
- , , , et al.ADAMTS13 activity in thrombotic thrombocytopenic purpura‐hemolytic uremic syndrome: relation to presenting features and clinical outcomes in a prospective cohort of 142 patients.Blood.2003;102:60–68.
- , , .Thrombotic thrombocytopenic purpura.J Thromb Haemost.2005;3:1663–1675.
- , , , et al.Interventions for hemolytic uremic syndrome and thrombotic thrombocytopenic purpura: a systematic review of randomized controlled trials.Am J Kidney Dis.2009;53:259–272.
- , , , et al.:Efficiency of curative and prophylactic treatment with rituximab in ADAMTS13‐deficient TTP: A study of 11 cases.Blood.2005;105:1932–1937.
- , , .Thrombotic microangiopathies. In: Tischer CC, Brenner BM, eds.Renal Pathology.2nd ed.Philadelphia, PA:JB Lippincott;1994:1154–1184.
- , , .Drug‐induced thrombotic microangiopathy: incidence, prevention and management.Drug Saf.2001;24(7):491–501.
- , , , , .FK 506‐associated thrombotic microangiopathy: report of two cases and review of the literature.Transplantation.1999;67(4):539–544.
- , .Thrombotic Thrombocytopenic purpura: report of 16 cases and review of the literature.Medicine (Baltimore).1966;45:139–159.
- , , , .Thrombotic thrombocytopenic purpura; early and late responders.Am J Hematol.1997;54:102–107.
- .Atypical presentations of thrombotic thrombocytopenic purpura: a review.J Clin Apheresis.2009;24(1)47–52.
- , , , et al.Comparison of plasma exchange with plasma infusion in the treatment of thrombotic thrombocytopenic purpura.N Engl J Med.1991;325:393–397.
- , , , , , .The incidence of thrombotic thrombocytopenic purpura‐hemolytic uremic syndrome: all patients, idiopathic patients, and patients with severe ADAMTS13deficiency.J Thromb Haemost.2005;3:1432–1436.
- .Thrombotic thrombocytopenic purpura.N Engl J Med.2006;354:1927–1935.
- , , , et al.ADAMTS13 activity in thrombotic thrombocytopenic purpura‐hemolytic uremic syndrome: relation to presenting features and clinical outcomes in a prospective cohort of 142 patients.Blood.2003;102:60–68.
- , , .Thrombotic thrombocytopenic purpura.J Thromb Haemost.2005;3:1663–1675.
- , , , et al.Interventions for hemolytic uremic syndrome and thrombotic thrombocytopenic purpura: a systematic review of randomized controlled trials.Am J Kidney Dis.2009;53:259–272.
- , , , et al.:Efficiency of curative and prophylactic treatment with rituximab in ADAMTS13‐deficient TTP: A study of 11 cases.Blood.2005;105:1932–1937.
Hospitalists: Lean leaders for hospitals
Unsustainable increases in health care costs mandate efforts at cost reduction.1 Such efforts necessitate enhanced productivity, especially given the specter of an aging population afflicted by a burgeoning chronic disease burden.2 Productivity is less a choice than an imperative forced upon hospitals and health systems as they attempt to address the competing requirements of diminished resources and increased demands. While the traditional mindset treats the goals of cost reduction and improving quality as tradeoffs, the methodology and philosophy known as Lean provides a proven approach for simultaneously improving both factors.3 Ideally, improved quality should lead to lower cost, and improved productivity should lead to better quality outcomes for patients.
This issue of the Journal of Hospital Medicine (JHM) describes multiple efforts to assess the activities of hospitalists and other hospital‐based physicians through use of time‐flow measurement.47 Understanding how health care workers spend their time and on which tasks that time is spent are essential steps toward applying Lean methodology at the point of care, or gembaa Japanese word that means the place where the work is actually done.8 At many health care institutions this gemba focus has not been integral to healthcare management models, and likely is a contributing factor to the cost and quality levels that exist today. The studies directly observing care delivery published in this issue of JHM provide invaluable lessons on how we might both improve productivity and quality of care delivery in the hospital. In this editorial, we review essential components of Lean methodology and propose how hospitalists and hospitals can benefit from its application.9
Value and Waste
In the Lean model, work and activity are broken down into the general categories of value and waste. The time and activities, as viewed from the customer's (ie, patient in the hospital) perspective, can also be categorized in a similar way. The goal in a Lean environment is to maximize value to the customer while reducing activities that are not value (ie, activities lacking value are waste).
Some define value as the simple mathematical equation of quality divided by cost.10 Better quality and/or lower cost means more value. A classical Lean definition of value requires three criteria to be met.11 First, the customer (patient) must be willing to pay for the given activity, directly or indirectly. When a hospitalist initiates care in the Emergency Department by placing admitting orders for a patient, the patient would view this activity as value because it progressed the care of the patient. However, if the patient is forced to wait 5 hours in the Emergency Room for an available inpatient bed while receiving minimal care, the patient may likely view that time as waste. Second, the activity must move the process forward toward the desired outcome in a meaningful way. Testing and exam activity that leads to a diagnosis would meet this criterion, while unnecessary CT scans might not. Third, the activity must be done properly the first time so as to minimize any rework, an important core quality component of the Lean approach.
All hospitalists perform activities that represent value and others that represent waste during their day. The nomenclature is not meant to be a value judgment on the clinician or their role. Lean provides a formal framework to describe waste in 8 key categories (Table 1), all meant to look at the system related elements of waste instead of the blaming of an individual.12 Common applications of Lean in healthcare focus on reducing waste to free up more time to deliver value, or to ensure that the value work is done at the highest possible level of quality. When hospitalists must take time to locate a colleague or a piece of information, that hunting and gathering time is waste. It distracts them from providing value. Too much waste within a fixed time period may lead to corners being cut or a lack of responsiveness to patient needsresulting in degradation in the quality of care and outcomes.
|
| Defects (correction, rework) |
| Overproduction |
| Transportation |
| Waiting |
| Inventory |
| Motion |
| Overprocessing |
| Human talent |
A simpler way of looking at activity for hospitalists and the care team often classifies any time spent in the patient room or at the bedside as direct value. This time can include clinical activities or time spent simply communicating with a patient and their families about their care or concerns. There may be activity in the room that could be considered waste (searching for information in the EMR), but proximity to patients is often considered valuable for other reasons. In the field of nursing, multiple studies in the past few years focused on identifying the percentage of time that nurses spend in patient rooms (consistently in the 3035% range across health systems and continents).13 The problem of waste is a long‐standing one in hospitals. In 1922 Henry Ford wrote, In the ordinary hospital the nurses must make useless steps. More of their time is spent in walking than in caring for the patient. [A hospital in Detroit] is designed to save steps we have tried to eliminate waste motion in the hospital.14
Activity outside of the patient room may be sometimes considered of indirect value, but this is often a gray area. Charting and medical decision‐making may benefit the patient and move the care process forward, and thus be of clear value. Yet, such activity may have questionable patient value if undertaken solely for billing or regulatory reasons. Effectively coordinating care between different members of the care team from both inside the hospital as well as beyond its walls does have value, but waste typically occurs when information is transferred incompletely or inaccurately.
Reducing waste often requires systemic changes to processes, workflow, and physical space. Motion (walking and searching) is a common form of waste in healthcare. Systemic Lean improvements might include changing the location of equipment and medication storage, or even patients.15 Uneven workloads often cannot be addressed by an individualthere must be a systemic effort to level workloads (the Lean term being heijunka), for example, leveling patient discharges throughout the day instead of doing them all in the late afternoon.
Lean also focuses on not wasting human talent or professional potential, often referred to in the literature as the eighth type of waste because it is missing from some Lean reference books.11 When hospitalists perform work that could be done by a midlevel provider (ie, physician assistant or nurse practitioner), or when a nurse performs work that could be done by a tech, the hospital wastes a scarce resource, human capital. Of note, changing these roles and responsibilities requires systemic effort rather than people just quitting a certain activity because it is below their pay grade; eg, it is better for the wrong person to be taking vital signs than to not have them documented at all.
Subject or Scientist
Toyota describes its management system, the Toyota Way, as having 2 equally important pillars: continuous improvement and what they call respect for humanity.16
If hospitals focus only on the improvement pillar, they run the risk of alienating the clinicians and staff members, undercutting any attempts at quality or productivity improvement. Respect for humanity is a much more sophisticated concept than just making employees happy in a superficial way. Respect, in a Lean sense, includes not robbing people of the opportunity to improve their own work. As participation increases the pride people feel in their work, more improvement resultsa virtuous cycle.17
Importantly, the Lean approach to quality improvement does not mirror the classical approach to improving productivity in a factory. Frederick Taylor (18561915) and Frank Gilbreth (18681924) are considered the fathers of Industrial Engineering, but their philosophy promulgated the belief that workers are not smart enough to participate in improvement.18 While they contributed a number of work analysis and process improvement methods that we use to this day, their philosophy is not one that fits with the respect for humanity principle of a modern professional workplace. Taylor believed a primary workplace problem was that people loafed and did not work hard enough; a seeming defect in their character as opposed to something that management should investigate and understand (for example, asking Why are people no longer motivated?).19 Taylor stood over workers, timing and watching their efforts, devising methods that workers should use to maximize their productivity. The term Taylorist is often used to describe this forced separation between working and thinking. The modern approach to Lean management draws more on the philosophy of Demingpeople want to do quality work, but the system gets in the way. The modern Lean approach emphasizes that every employee has 2 jobsboth to do the work and to improve it. The daily practice of kaizen, or continuous improvement, engages every employee in a problem‐solving dialogue with their leaders. In a Lean hospital, everybody deserves respect for their role, from a night‐time hospitalist to patient transporters, and all can play a role in process improvement.
Having research assistants shadow hospitalists could be done in a Taylorist or Deming way. Ideally, the role of a Lean improvement professional would be to teach those doing the work how to identify waste, allowing the hospitalists to develop and test their own improvements based on their existing professional knowledge combined with Lean principles. While the time‐flow studies published in this issue of JHM identified how the hospital system can be a barrier to hospitalist efficiency, this also potentially represents a wasted opportunity. Ideally, if the observers had been Lean improvement professionals they would not have just shadowed hospitalists without talking to or engaging them. They would have helped identify batching in a process or teaching the hospitalists why that practice is often not optimal. Future research should focus on applying this approach to time‐flow analysis in the hospital.
Simply putLean and process improvement techniques run the risk of being disrespectful, ineffective, and unsustainable when they are done to somebody, (the Taylor/Gilbreth approach) instead of utilized to both assess activities and glean learning from the front‐line staff. To be sustainable, effective, and respectful, hospitals should strive to truly engage in process improvement the people who are actually performing the work. Instead of efficiency experts, we need skilled coaches and mentors who can guide people towards generating their own improvements. Finally, when we have experts like Taylor or Gilbreth leading process improvement, those experts become a crutch and a bottleneck. Only by teaching the clinicians and staff members these skills, combined with patient focus and respect for humanity, can we begin moving a hospital's culture to one of true continuous improvementleading to better patient safety and quality, better access, lower costs, and better staff morale.
Conclusion
Hospitalists seem to be ideal leaders in efforts to generate ideas for improvement to remove waste from the health care system. Efficiency, value, and quality will be the mantra as we head into an era of healthcare where every action will be analyzed as to whether the action provides value to the patient. Hospitalists are well poised as Dr. Peter Pronovost recently stated. I think hospitalists' roles are going to go up dramatically, and I hope the field responds by making sure they put out people who have the skills to lead.20 Hospitalists experience and see waste in the processes of care. Yet, as Lord Kelvin is credited with the saying, If you can not measure it, you can not improve it and future time‐flow studies of hospitalists must take advantage of opportunities to also measure waste and not just document activity.
- .The cost implications of health care reform.N Engl J Med.2010 (in press).
- . The cost of inaction: the urgent need for health reform. Available at: http://www.healthreform.gov/reports/inaction/inactionreportprintmarch2009.pdf. Accessed May2010.
- .Writing the new playbook for U.S. health care: lessons from Wisconsin.Health Aff (Millwood).2009;28(5):1343–1350.
- , , , , .Systematic review of time studies evaluating physicians in the hospital setting.J Hosp Med.2010;5(6):353–359.
- , , , et al.Where did the day go?—A time‐motion study of hospitalists.J Hosp Med.2010;5(6):323–328.
- , , , , .Comparing Academic and Community‐Based Hospitalists.J Hosp Med.2010;5(6):349–352.
- , , , , .Hospitalist time usage and cyclicality: opportunities to improve efficiency.J Hosp Med.2010;5(6):329–334.
- , .Lean Lexicon: A Graphical Glossary for Lean Thinkers.Cambridge, MA:Lean Enterprise Institute;2003.
- , , , .Lean health care: what can hospitals learn from a world‐class automaker?J Hosp Med.2006;1(3):191–199.
- . Evaluating the Impact of Value‐Based Purchasing: A Guide for Purchasers. Available at: http://www.ahrq.gov/about/cods/valuebased/evalvbp1.htm. Accessed May2010.
- .Lean Hospitals: Improving Quality, Patient Safety, and Employee Satisfaction.New York:Productivity Press;2008.
- , .Lean Thinking.New York:Simon and Schuster;1996.
- , .Creating an environment for caring using lean principles of the Virginia Mason Production System.J Nurs Adm.2007;37(6):287–294.
- , .My Life and Work.Garden City, NY:Garden City Publishing;1922.
- , , , et al.Impact of localizing physicians to hospital units on nurse‐physician communication and agreement on the plan of care.J Gen Intern Med.2009;24(11):1223–1227.
- .Toyota Production System: Beyond Large‐Scale Production.New York:Productivity Press;1988.
- .Out of the Crisis.Cambridge:Massachusetts Institute of Technology—Center of Advanced Educational Services;1982.
- .Frederick Taylor and Frank Gilbreth: competition in scientific management.Bus Hist Rev.1957;31(1):23–34.
- .The Principles of Scientific Management.Norwood, MA:The Plimpton Press;1911.
- .The Year Ahead.The hospitalist.2010;14(2):1,4–5.
Unsustainable increases in health care costs mandate efforts at cost reduction.1 Such efforts necessitate enhanced productivity, especially given the specter of an aging population afflicted by a burgeoning chronic disease burden.2 Productivity is less a choice than an imperative forced upon hospitals and health systems as they attempt to address the competing requirements of diminished resources and increased demands. While the traditional mindset treats the goals of cost reduction and improving quality as tradeoffs, the methodology and philosophy known as Lean provides a proven approach for simultaneously improving both factors.3 Ideally, improved quality should lead to lower cost, and improved productivity should lead to better quality outcomes for patients.
This issue of the Journal of Hospital Medicine (JHM) describes multiple efforts to assess the activities of hospitalists and other hospital‐based physicians through use of time‐flow measurement.47 Understanding how health care workers spend their time and on which tasks that time is spent are essential steps toward applying Lean methodology at the point of care, or gembaa Japanese word that means the place where the work is actually done.8 At many health care institutions this gemba focus has not been integral to healthcare management models, and likely is a contributing factor to the cost and quality levels that exist today. The studies directly observing care delivery published in this issue of JHM provide invaluable lessons on how we might both improve productivity and quality of care delivery in the hospital. In this editorial, we review essential components of Lean methodology and propose how hospitalists and hospitals can benefit from its application.9
Value and Waste
In the Lean model, work and activity are broken down into the general categories of value and waste. The time and activities, as viewed from the customer's (ie, patient in the hospital) perspective, can also be categorized in a similar way. The goal in a Lean environment is to maximize value to the customer while reducing activities that are not value (ie, activities lacking value are waste).
Some define value as the simple mathematical equation of quality divided by cost.10 Better quality and/or lower cost means more value. A classical Lean definition of value requires three criteria to be met.11 First, the customer (patient) must be willing to pay for the given activity, directly or indirectly. When a hospitalist initiates care in the Emergency Department by placing admitting orders for a patient, the patient would view this activity as value because it progressed the care of the patient. However, if the patient is forced to wait 5 hours in the Emergency Room for an available inpatient bed while receiving minimal care, the patient may likely view that time as waste. Second, the activity must move the process forward toward the desired outcome in a meaningful way. Testing and exam activity that leads to a diagnosis would meet this criterion, while unnecessary CT scans might not. Third, the activity must be done properly the first time so as to minimize any rework, an important core quality component of the Lean approach.
All hospitalists perform activities that represent value and others that represent waste during their day. The nomenclature is not meant to be a value judgment on the clinician or their role. Lean provides a formal framework to describe waste in 8 key categories (Table 1), all meant to look at the system related elements of waste instead of the blaming of an individual.12 Common applications of Lean in healthcare focus on reducing waste to free up more time to deliver value, or to ensure that the value work is done at the highest possible level of quality. When hospitalists must take time to locate a colleague or a piece of information, that hunting and gathering time is waste. It distracts them from providing value. Too much waste within a fixed time period may lead to corners being cut or a lack of responsiveness to patient needsresulting in degradation in the quality of care and outcomes.
|
| Defects (correction, rework) |
| Overproduction |
| Transportation |
| Waiting |
| Inventory |
| Motion |
| Overprocessing |
| Human talent |
A simpler way of looking at activity for hospitalists and the care team often classifies any time spent in the patient room or at the bedside as direct value. This time can include clinical activities or time spent simply communicating with a patient and their families about their care or concerns. There may be activity in the room that could be considered waste (searching for information in the EMR), but proximity to patients is often considered valuable for other reasons. In the field of nursing, multiple studies in the past few years focused on identifying the percentage of time that nurses spend in patient rooms (consistently in the 3035% range across health systems and continents).13 The problem of waste is a long‐standing one in hospitals. In 1922 Henry Ford wrote, In the ordinary hospital the nurses must make useless steps. More of their time is spent in walking than in caring for the patient. [A hospital in Detroit] is designed to save steps we have tried to eliminate waste motion in the hospital.14
Activity outside of the patient room may be sometimes considered of indirect value, but this is often a gray area. Charting and medical decision‐making may benefit the patient and move the care process forward, and thus be of clear value. Yet, such activity may have questionable patient value if undertaken solely for billing or regulatory reasons. Effectively coordinating care between different members of the care team from both inside the hospital as well as beyond its walls does have value, but waste typically occurs when information is transferred incompletely or inaccurately.
Reducing waste often requires systemic changes to processes, workflow, and physical space. Motion (walking and searching) is a common form of waste in healthcare. Systemic Lean improvements might include changing the location of equipment and medication storage, or even patients.15 Uneven workloads often cannot be addressed by an individualthere must be a systemic effort to level workloads (the Lean term being heijunka), for example, leveling patient discharges throughout the day instead of doing them all in the late afternoon.
Lean also focuses on not wasting human talent or professional potential, often referred to in the literature as the eighth type of waste because it is missing from some Lean reference books.11 When hospitalists perform work that could be done by a midlevel provider (ie, physician assistant or nurse practitioner), or when a nurse performs work that could be done by a tech, the hospital wastes a scarce resource, human capital. Of note, changing these roles and responsibilities requires systemic effort rather than people just quitting a certain activity because it is below their pay grade; eg, it is better for the wrong person to be taking vital signs than to not have them documented at all.
Subject or Scientist
Toyota describes its management system, the Toyota Way, as having 2 equally important pillars: continuous improvement and what they call respect for humanity.16
If hospitals focus only on the improvement pillar, they run the risk of alienating the clinicians and staff members, undercutting any attempts at quality or productivity improvement. Respect for humanity is a much more sophisticated concept than just making employees happy in a superficial way. Respect, in a Lean sense, includes not robbing people of the opportunity to improve their own work. As participation increases the pride people feel in their work, more improvement resultsa virtuous cycle.17
Importantly, the Lean approach to quality improvement does not mirror the classical approach to improving productivity in a factory. Frederick Taylor (18561915) and Frank Gilbreth (18681924) are considered the fathers of Industrial Engineering, but their philosophy promulgated the belief that workers are not smart enough to participate in improvement.18 While they contributed a number of work analysis and process improvement methods that we use to this day, their philosophy is not one that fits with the respect for humanity principle of a modern professional workplace. Taylor believed a primary workplace problem was that people loafed and did not work hard enough; a seeming defect in their character as opposed to something that management should investigate and understand (for example, asking Why are people no longer motivated?).19 Taylor stood over workers, timing and watching their efforts, devising methods that workers should use to maximize their productivity. The term Taylorist is often used to describe this forced separation between working and thinking. The modern approach to Lean management draws more on the philosophy of Demingpeople want to do quality work, but the system gets in the way. The modern Lean approach emphasizes that every employee has 2 jobsboth to do the work and to improve it. The daily practice of kaizen, or continuous improvement, engages every employee in a problem‐solving dialogue with their leaders. In a Lean hospital, everybody deserves respect for their role, from a night‐time hospitalist to patient transporters, and all can play a role in process improvement.
Having research assistants shadow hospitalists could be done in a Taylorist or Deming way. Ideally, the role of a Lean improvement professional would be to teach those doing the work how to identify waste, allowing the hospitalists to develop and test their own improvements based on their existing professional knowledge combined with Lean principles. While the time‐flow studies published in this issue of JHM identified how the hospital system can be a barrier to hospitalist efficiency, this also potentially represents a wasted opportunity. Ideally, if the observers had been Lean improvement professionals they would not have just shadowed hospitalists without talking to or engaging them. They would have helped identify batching in a process or teaching the hospitalists why that practice is often not optimal. Future research should focus on applying this approach to time‐flow analysis in the hospital.
Simply putLean and process improvement techniques run the risk of being disrespectful, ineffective, and unsustainable when they are done to somebody, (the Taylor/Gilbreth approach) instead of utilized to both assess activities and glean learning from the front‐line staff. To be sustainable, effective, and respectful, hospitals should strive to truly engage in process improvement the people who are actually performing the work. Instead of efficiency experts, we need skilled coaches and mentors who can guide people towards generating their own improvements. Finally, when we have experts like Taylor or Gilbreth leading process improvement, those experts become a crutch and a bottleneck. Only by teaching the clinicians and staff members these skills, combined with patient focus and respect for humanity, can we begin moving a hospital's culture to one of true continuous improvementleading to better patient safety and quality, better access, lower costs, and better staff morale.
Conclusion
Hospitalists seem to be ideal leaders in efforts to generate ideas for improvement to remove waste from the health care system. Efficiency, value, and quality will be the mantra as we head into an era of healthcare where every action will be analyzed as to whether the action provides value to the patient. Hospitalists are well poised as Dr. Peter Pronovost recently stated. I think hospitalists' roles are going to go up dramatically, and I hope the field responds by making sure they put out people who have the skills to lead.20 Hospitalists experience and see waste in the processes of care. Yet, as Lord Kelvin is credited with the saying, If you can not measure it, you can not improve it and future time‐flow studies of hospitalists must take advantage of opportunities to also measure waste and not just document activity.
Unsustainable increases in health care costs mandate efforts at cost reduction.1 Such efforts necessitate enhanced productivity, especially given the specter of an aging population afflicted by a burgeoning chronic disease burden.2 Productivity is less a choice than an imperative forced upon hospitals and health systems as they attempt to address the competing requirements of diminished resources and increased demands. While the traditional mindset treats the goals of cost reduction and improving quality as tradeoffs, the methodology and philosophy known as Lean provides a proven approach for simultaneously improving both factors.3 Ideally, improved quality should lead to lower cost, and improved productivity should lead to better quality outcomes for patients.
This issue of the Journal of Hospital Medicine (JHM) describes multiple efforts to assess the activities of hospitalists and other hospital‐based physicians through use of time‐flow measurement.47 Understanding how health care workers spend their time and on which tasks that time is spent are essential steps toward applying Lean methodology at the point of care, or gembaa Japanese word that means the place where the work is actually done.8 At many health care institutions this gemba focus has not been integral to healthcare management models, and likely is a contributing factor to the cost and quality levels that exist today. The studies directly observing care delivery published in this issue of JHM provide invaluable lessons on how we might both improve productivity and quality of care delivery in the hospital. In this editorial, we review essential components of Lean methodology and propose how hospitalists and hospitals can benefit from its application.9
Value and Waste
In the Lean model, work and activity are broken down into the general categories of value and waste. The time and activities, as viewed from the customer's (ie, patient in the hospital) perspective, can also be categorized in a similar way. The goal in a Lean environment is to maximize value to the customer while reducing activities that are not value (ie, activities lacking value are waste).
Some define value as the simple mathematical equation of quality divided by cost.10 Better quality and/or lower cost means more value. A classical Lean definition of value requires three criteria to be met.11 First, the customer (patient) must be willing to pay for the given activity, directly or indirectly. When a hospitalist initiates care in the Emergency Department by placing admitting orders for a patient, the patient would view this activity as value because it progressed the care of the patient. However, if the patient is forced to wait 5 hours in the Emergency Room for an available inpatient bed while receiving minimal care, the patient may likely view that time as waste. Second, the activity must move the process forward toward the desired outcome in a meaningful way. Testing and exam activity that leads to a diagnosis would meet this criterion, while unnecessary CT scans might not. Third, the activity must be done properly the first time so as to minimize any rework, an important core quality component of the Lean approach.
All hospitalists perform activities that represent value and others that represent waste during their day. The nomenclature is not meant to be a value judgment on the clinician or their role. Lean provides a formal framework to describe waste in 8 key categories (Table 1), all meant to look at the system related elements of waste instead of the blaming of an individual.12 Common applications of Lean in healthcare focus on reducing waste to free up more time to deliver value, or to ensure that the value work is done at the highest possible level of quality. When hospitalists must take time to locate a colleague or a piece of information, that hunting and gathering time is waste. It distracts them from providing value. Too much waste within a fixed time period may lead to corners being cut or a lack of responsiveness to patient needsresulting in degradation in the quality of care and outcomes.
|
| Defects (correction, rework) |
| Overproduction |
| Transportation |
| Waiting |
| Inventory |
| Motion |
| Overprocessing |
| Human talent |
A simpler way of looking at activity for hospitalists and the care team often classifies any time spent in the patient room or at the bedside as direct value. This time can include clinical activities or time spent simply communicating with a patient and their families about their care or concerns. There may be activity in the room that could be considered waste (searching for information in the EMR), but proximity to patients is often considered valuable for other reasons. In the field of nursing, multiple studies in the past few years focused on identifying the percentage of time that nurses spend in patient rooms (consistently in the 3035% range across health systems and continents).13 The problem of waste is a long‐standing one in hospitals. In 1922 Henry Ford wrote, In the ordinary hospital the nurses must make useless steps. More of their time is spent in walking than in caring for the patient. [A hospital in Detroit] is designed to save steps we have tried to eliminate waste motion in the hospital.14
Activity outside of the patient room may be sometimes considered of indirect value, but this is often a gray area. Charting and medical decision‐making may benefit the patient and move the care process forward, and thus be of clear value. Yet, such activity may have questionable patient value if undertaken solely for billing or regulatory reasons. Effectively coordinating care between different members of the care team from both inside the hospital as well as beyond its walls does have value, but waste typically occurs when information is transferred incompletely or inaccurately.
Reducing waste often requires systemic changes to processes, workflow, and physical space. Motion (walking and searching) is a common form of waste in healthcare. Systemic Lean improvements might include changing the location of equipment and medication storage, or even patients.15 Uneven workloads often cannot be addressed by an individualthere must be a systemic effort to level workloads (the Lean term being heijunka), for example, leveling patient discharges throughout the day instead of doing them all in the late afternoon.
Lean also focuses on not wasting human talent or professional potential, often referred to in the literature as the eighth type of waste because it is missing from some Lean reference books.11 When hospitalists perform work that could be done by a midlevel provider (ie, physician assistant or nurse practitioner), or when a nurse performs work that could be done by a tech, the hospital wastes a scarce resource, human capital. Of note, changing these roles and responsibilities requires systemic effort rather than people just quitting a certain activity because it is below their pay grade; eg, it is better for the wrong person to be taking vital signs than to not have them documented at all.
Subject or Scientist
Toyota describes its management system, the Toyota Way, as having 2 equally important pillars: continuous improvement and what they call respect for humanity.16
If hospitals focus only on the improvement pillar, they run the risk of alienating the clinicians and staff members, undercutting any attempts at quality or productivity improvement. Respect for humanity is a much more sophisticated concept than just making employees happy in a superficial way. Respect, in a Lean sense, includes not robbing people of the opportunity to improve their own work. As participation increases the pride people feel in their work, more improvement resultsa virtuous cycle.17
Importantly, the Lean approach to quality improvement does not mirror the classical approach to improving productivity in a factory. Frederick Taylor (18561915) and Frank Gilbreth (18681924) are considered the fathers of Industrial Engineering, but their philosophy promulgated the belief that workers are not smart enough to participate in improvement.18 While they contributed a number of work analysis and process improvement methods that we use to this day, their philosophy is not one that fits with the respect for humanity principle of a modern professional workplace. Taylor believed a primary workplace problem was that people loafed and did not work hard enough; a seeming defect in their character as opposed to something that management should investigate and understand (for example, asking Why are people no longer motivated?).19 Taylor stood over workers, timing and watching their efforts, devising methods that workers should use to maximize their productivity. The term Taylorist is often used to describe this forced separation between working and thinking. The modern approach to Lean management draws more on the philosophy of Demingpeople want to do quality work, but the system gets in the way. The modern Lean approach emphasizes that every employee has 2 jobsboth to do the work and to improve it. The daily practice of kaizen, or continuous improvement, engages every employee in a problem‐solving dialogue with their leaders. In a Lean hospital, everybody deserves respect for their role, from a night‐time hospitalist to patient transporters, and all can play a role in process improvement.
Having research assistants shadow hospitalists could be done in a Taylorist or Deming way. Ideally, the role of a Lean improvement professional would be to teach those doing the work how to identify waste, allowing the hospitalists to develop and test their own improvements based on their existing professional knowledge combined with Lean principles. While the time‐flow studies published in this issue of JHM identified how the hospital system can be a barrier to hospitalist efficiency, this also potentially represents a wasted opportunity. Ideally, if the observers had been Lean improvement professionals they would not have just shadowed hospitalists without talking to or engaging them. They would have helped identify batching in a process or teaching the hospitalists why that practice is often not optimal. Future research should focus on applying this approach to time‐flow analysis in the hospital.
Simply putLean and process improvement techniques run the risk of being disrespectful, ineffective, and unsustainable when they are done to somebody, (the Taylor/Gilbreth approach) instead of utilized to both assess activities and glean learning from the front‐line staff. To be sustainable, effective, and respectful, hospitals should strive to truly engage in process improvement the people who are actually performing the work. Instead of efficiency experts, we need skilled coaches and mentors who can guide people towards generating their own improvements. Finally, when we have experts like Taylor or Gilbreth leading process improvement, those experts become a crutch and a bottleneck. Only by teaching the clinicians and staff members these skills, combined with patient focus and respect for humanity, can we begin moving a hospital's culture to one of true continuous improvementleading to better patient safety and quality, better access, lower costs, and better staff morale.
Conclusion
Hospitalists seem to be ideal leaders in efforts to generate ideas for improvement to remove waste from the health care system. Efficiency, value, and quality will be the mantra as we head into an era of healthcare where every action will be analyzed as to whether the action provides value to the patient. Hospitalists are well poised as Dr. Peter Pronovost recently stated. I think hospitalists' roles are going to go up dramatically, and I hope the field responds by making sure they put out people who have the skills to lead.20 Hospitalists experience and see waste in the processes of care. Yet, as Lord Kelvin is credited with the saying, If you can not measure it, you can not improve it and future time‐flow studies of hospitalists must take advantage of opportunities to also measure waste and not just document activity.
- .The cost implications of health care reform.N Engl J Med.2010 (in press).
- . The cost of inaction: the urgent need for health reform. Available at: http://www.healthreform.gov/reports/inaction/inactionreportprintmarch2009.pdf. Accessed May2010.
- .Writing the new playbook for U.S. health care: lessons from Wisconsin.Health Aff (Millwood).2009;28(5):1343–1350.
- , , , , .Systematic review of time studies evaluating physicians in the hospital setting.J Hosp Med.2010;5(6):353–359.
- , , , et al.Where did the day go?—A time‐motion study of hospitalists.J Hosp Med.2010;5(6):323–328.
- , , , , .Comparing Academic and Community‐Based Hospitalists.J Hosp Med.2010;5(6):349–352.
- , , , , .Hospitalist time usage and cyclicality: opportunities to improve efficiency.J Hosp Med.2010;5(6):329–334.
- , .Lean Lexicon: A Graphical Glossary for Lean Thinkers.Cambridge, MA:Lean Enterprise Institute;2003.
- , , , .Lean health care: what can hospitals learn from a world‐class automaker?J Hosp Med.2006;1(3):191–199.
- . Evaluating the Impact of Value‐Based Purchasing: A Guide for Purchasers. Available at: http://www.ahrq.gov/about/cods/valuebased/evalvbp1.htm. Accessed May2010.
- .Lean Hospitals: Improving Quality, Patient Safety, and Employee Satisfaction.New York:Productivity Press;2008.
- , .Lean Thinking.New York:Simon and Schuster;1996.
- , .Creating an environment for caring using lean principles of the Virginia Mason Production System.J Nurs Adm.2007;37(6):287–294.
- , .My Life and Work.Garden City, NY:Garden City Publishing;1922.
- , , , et al.Impact of localizing physicians to hospital units on nurse‐physician communication and agreement on the plan of care.J Gen Intern Med.2009;24(11):1223–1227.
- .Toyota Production System: Beyond Large‐Scale Production.New York:Productivity Press;1988.
- .Out of the Crisis.Cambridge:Massachusetts Institute of Technology—Center of Advanced Educational Services;1982.
- .Frederick Taylor and Frank Gilbreth: competition in scientific management.Bus Hist Rev.1957;31(1):23–34.
- .The Principles of Scientific Management.Norwood, MA:The Plimpton Press;1911.
- .The Year Ahead.The hospitalist.2010;14(2):1,4–5.
- .The cost implications of health care reform.N Engl J Med.2010 (in press).
- . The cost of inaction: the urgent need for health reform. Available at: http://www.healthreform.gov/reports/inaction/inactionreportprintmarch2009.pdf. Accessed May2010.
- .Writing the new playbook for U.S. health care: lessons from Wisconsin.Health Aff (Millwood).2009;28(5):1343–1350.
- , , , , .Systematic review of time studies evaluating physicians in the hospital setting.J Hosp Med.2010;5(6):353–359.
- , , , et al.Where did the day go?—A time‐motion study of hospitalists.J Hosp Med.2010;5(6):323–328.
- , , , , .Comparing Academic and Community‐Based Hospitalists.J Hosp Med.2010;5(6):349–352.
- , , , , .Hospitalist time usage and cyclicality: opportunities to improve efficiency.J Hosp Med.2010;5(6):329–334.
- , .Lean Lexicon: A Graphical Glossary for Lean Thinkers.Cambridge, MA:Lean Enterprise Institute;2003.
- , , , .Lean health care: what can hospitals learn from a world‐class automaker?J Hosp Med.2006;1(3):191–199.
- . Evaluating the Impact of Value‐Based Purchasing: A Guide for Purchasers. Available at: http://www.ahrq.gov/about/cods/valuebased/evalvbp1.htm. Accessed May2010.
- .Lean Hospitals: Improving Quality, Patient Safety, and Employee Satisfaction.New York:Productivity Press;2008.
- , .Lean Thinking.New York:Simon and Schuster;1996.
- , .Creating an environment for caring using lean principles of the Virginia Mason Production System.J Nurs Adm.2007;37(6):287–294.
- , .My Life and Work.Garden City, NY:Garden City Publishing;1922.
- , , , et al.Impact of localizing physicians to hospital units on nurse‐physician communication and agreement on the plan of care.J Gen Intern Med.2009;24(11):1223–1227.
- .Toyota Production System: Beyond Large‐Scale Production.New York:Productivity Press;1988.
- .Out of the Crisis.Cambridge:Massachusetts Institute of Technology—Center of Advanced Educational Services;1982.
- .Frederick Taylor and Frank Gilbreth: competition in scientific management.Bus Hist Rev.1957;31(1):23–34.
- .The Principles of Scientific Management.Norwood, MA:The Plimpton Press;1911.
- .The Year Ahead.The hospitalist.2010;14(2):1,4–5.
Time Studies of Hospital Physicians
Time‐motion studies, introduced by Frederick W. Taylor in the 1880s,1 have been widely implemented across the business world; a Google search of time‐motion study returns approximately 32 million results. Such studies continuously document how workers spend their time and then use this information to identify and eliminate inefficient practices. Work‐sampling is a similar methodology introduced by L.H.C. Tippett in 1935.2 Work‐sampling utilizes a trained observer to document activities at predetermined or random intervals of time. Given a large enough number of observations, this method can be comparable to the continuous observation method used in time‐motion studies.3
Healthcare has begun to utilize these time‐study methodologies to evaluate the activities of physicians and nurses. Researchers have successfully used time‐study methodology in the emergency department, intensive care unit, and ambulatory and surgical settings in the U.S. and around the world to better understand physician activities and to design and assess interventions to improve efficiency.49 Hospitals are also eager to enhance efficiency in the inpatient setting given the current economic environment. Hospitalizations account for over a third of healthcare costs in the United States, making them an attractive target for cost‐cutting measures.10 Acknowledging that healthcare expenditures cannot continue to rise,11 insurers, particularly the Centers for Medicare and Medicaid Services (CMS), increasingly seek to reduce payments to hospitals.12 Compounding these pressures, a major supply of relatively inexpensive labor shrank with the decision by the Accreditation Council for Graduate Medicine Education (ACGME) to restrict the number of hours residents are allowed to work. Efficiency concerns gain new urgency as hospitals scramble to cover their patient loads with reduced physician availability.13
We undertook a systematic review of time‐motion and work‐sampling studies performed in the hospital setting to better understand the available literature describing the activities of physicians caring for hospitalized patients. An additional goal of this review was to determine the extent of available time‐flow literature describing the activity of hospitalists. The hospitalist movement provided one viable solution to the gap between demand for hospital patient care and the reduced supply of available physicianstypically primary care physicians in community hospitals and residents in teaching hospitals.14 Hospital medicine is the fastest‐growing specialty in the history of American medicine.15 More than half of American hospitals now have hospital medicine programs with a total of greater than 25,000 hospitalists in the U.S.15 This popularity has been driven by hospitalists' ability to increase efficiency through decreasing overall cost and length of stay for patients without increasing readmission rates or reducing primary care physician satisfaction.1619 However, exactly how hospitalists accomplish this increase in efficiency is still the subject of debate. One time‐motion study provides a glimpse into the activities of hospitalists at an academic urban hospital,20 but may not be applicable to many other hospitals.
Methods
Data Sources
With assistance from a medical librarian, we searched for English‐language articles published between 1965 and June 2009 using the MEDLINE (
Study Selection
We selected articles that met the following criteria: (1) explicit use of time‐motion methodology or work‐sampling performed via direct observation; (2) study populations including physicians, medical residents, or interns; (3) performance sites on an inpatient hospital ward (ie, not outpatient within the hospital, emergency room (ER), or operating room (OR)); and (4) observation of at least half of a shift. Titles and abstracts of the retrieved citations were first reviewed to identify studies that could potentially meet our criteria. Full‐text versions of the selected articles were then retrieved and analyzed by at least 2 of 4 authors (V.F., K.E., D.M., and M.T.) to determine the final list of articles. Articles that failed to provide sufficient information for one or more criteria were excluded.
Data Extraction
Each article was independently reviewed by at least 2 of 3 authors (V.F., D.M., and M.T.) using a standardized data abstraction form. The form included the following categories: sample population, sample size, hospital type, data collection tool type, time‐motion/observation duration, key categories of activity, and key results. If an article included additional data beyond the scope of this review (eg, data from surgical residents in the OR as well as internal medicine residents) only the qualifying portion of the study was included. Disagreements were resolved through discussion and consensus. Data were then compiled into tables.
Results
Our database search yielded 4270 potential articles. We then reviewed the title and abstract of each of these articles to identify studies that evaluated physicians, were performed on a hospital ward, and explicitly used time‐motion or direct‐observation work‐sampling methodology. For articles lacking an abstract but having a relevant title, we obtained the full text to determine eligibility for additional review. Sixty‐eight articles from this original search were selected for full‐text review. Ten of these articles met the selection criteria. Most of the articles excluded in this step were either conducted in an outpatient OR or ER setting, or used self‐report data instead of direct‐observation data. A secondary search using the reference lists of all obtained articles as well as consultation with experts in the field yielded 11 additional articles of interest. Three of these 11 articles were found to meet our criteria, bringing the total to 13 articles for review (Fig. 1).
These 13 articles included several types of physicians in their samples. Eleven included interns,2131 7 included residents,2123, 2628, 31 and 4 included attending physicians20, 23, 26, 32 (Table 1). Six articles included more than 1 type of subject.2123, 26, 28, 31 The main focus of these articles also varied. Nine of the 13 studies were designed to simply describe how residents, physicians and nurses spend their time.20, 2227, 29, 31 Three studies were primarily concerned with comparing groups from different intern programs, residency rotations, hospital types, or shifts.28, 30, 32 The remaining study attempted to quantify the amount of time physicians spent on tasks that could be performed by non‐physician staff.21 Only 2 articles evaluated hospitalists,20, 32 and we found no articles studying hospitalists in a community, non‐teaching setting. The studies were performed as early as 1961 and as recently as 2009. Just 5 of the 13 articles were published within the last 10 years. 0
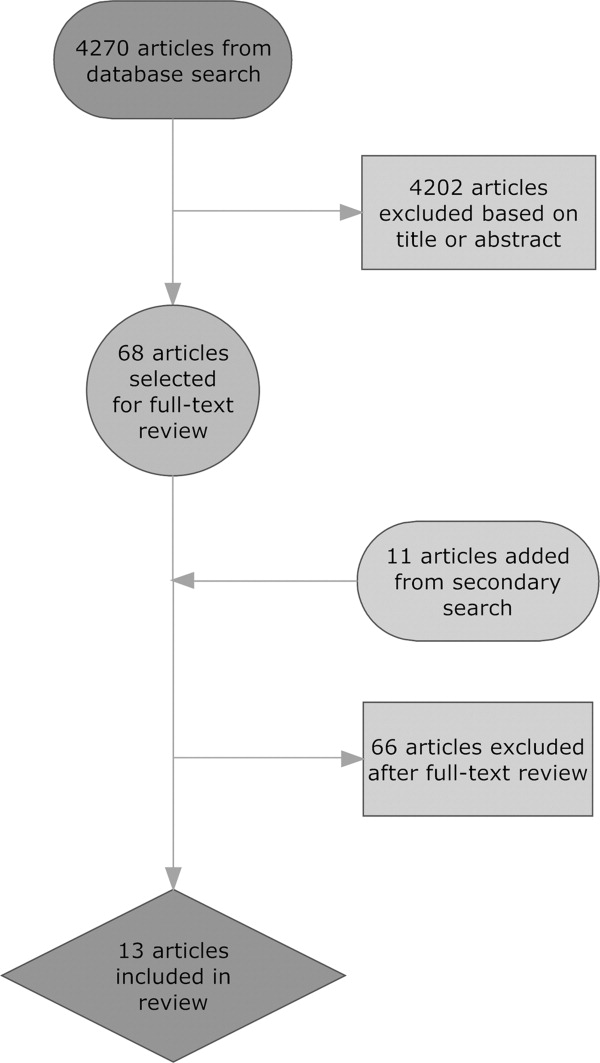
| Reference (Year) | Type of Hospital | Data Collection Tool | Direct/Indirect Care Estimates | Key Activities Reported and Percentage of Time Spent on Each, and Other Results |
|---|---|---|---|---|
| ||||
| Ammenwerth and Spotl (2009) | Academic | Work sampling: paper and stopwatch | Direct care 27.5%; indirect care 62.8% | Documentation tasks 26.6%; direct care 27.5%; communication 36.2%; other tasks 9.7%. Approximately 16% of documentation time was administrative documentation. |
| Arthurson et al. (1976) | Academic | Paper and stopwatch | Direct care 40.8%; indirect care 45.5% | Medical intern: patient care 40.8%; clerical 25.5%; telephone 10%; professional discussion 10.5%; transit/waiting 6.5%, personal 7% |
| Gabow et al. (2006) | Academic | Paper and stopwatch with pedometer | Direct care 19.5%; indirect care: 32% | Attend to/assess patient 17%; charting 9.5%; consult with MD/nurse 9.5%; downtime 6.5%; educational activity 2.5%; family interaction 0.5%; paging/phone 3.5%; procedure 1.5%; review films/laboratory results 9%; rounds 15.5%; sleep 21.5%; travel 3.5% |
| Gillanders et al. (1971) | Academic | Paper and stopwatch | Direct care 19%; indirect care 42.5% | Individual interaction 14.5%; nonverbal communication 20.5%; procedures 10.0%; laboratory work 3.5%; interpersonal communication 18.5%; rounds 9%; direction and supervision 0.5%; nonmedical talk 2%; education 8.5%; ancillary activities 5.5%; personal 7% |
| Knickman et al. (1992) | Academic | Paper and stopwatch | Direct care 8.5%; indirect care 37.7% | Education 20.7%; information gathering 13.7%; personal 13.3%; testing 12%; consulting 12%; documenting 9.8%; transit 8.2%; procedures 5.4%; interacting with patients 3.1%; administration 1.8%. 46.7% of residents' time was spent on tasks that required a physician. |
| Lurie et al. (1989) | Academic | Paper and stopwatch | Direct care 17.4%; indirect care 39.3% | Procedures 3%; patient evaluation 20%; communication 27%; basic 40%; miscellaneous 10%. On average, doctors were interrupted 9 minutes into an H&P, got 230 minutes of sleep per night, and slept 59 minutes before being woken up by some interruption. |
| Magnusson et al. (1998) | Academic | Paper and stopwatch | Could not be determined | Clinical 54%; education 28%; personal 18%. The 3 specialties did not differ significantly in time spent on these categories except for education time: emergency 24%; internal 28%; surgery 18%. |
| Malkenson (unpublished data) | 1 Community; 1 Academic | Paper and stopwatch | Academic: direct care 19%; indirect care 56%. Community: direct care 25%; indirect care 55%. | Direct patient care (25% community, 19% academic); indirect patient care (55% community, 56% academic); personal time (4% community, 6% academic); travel time (10% community, 10% academic); other activities (10% community, 13% academic) |
| Nerenz et al. (1990) | Academic | Work sampling: paper and stopwatch | Direct care 18.9%; indirect care could not be determined | Interns averaged 21 pages over 30 hours of observation, and slept an average of 2.5 hours with 2 interruptions. Attending physicians interacted with the interns for an average of 139 minutes per shift. |
| O'Leary et al. (2006) | Academic | Paper and stopwatch | Direct care 18%; indirect care 69% | Indirect patient care occupied 69% of hospitalists' time. Indirect care included: documentation 37%; communication 35%; reviewing results 21%; orders 7%. Direct care occupied 18%, and included: history and physical 18%; follow‐up visits 53%; family meetings 13%; discharge instructions 16%. Remaining time was spent on personal activities 4%; professional development 3%; education 3%; travel 3%. |
| Parenti et al. (1993) | Academic | Paper and stopwatch | Interns: direct care 39%; indirect care 51%. Residents: direct care 40%; indirect care 47%. | Interns: procedures 4%; patient evaluation 35%; communication 42%; basics 11%; miscellaneous 8%. Residents: procedures 2%; patient evaluation 38%; communication 35%; basics 12%; miscellaneous 13%. |
| Payson et al. (1961) | Academic | Paper and stopwatch | Could not be determined | Communication with staff took up the largest amount of time. Remaining time was evenly distributed between the categories of personal activities, ancillary duties, patient and relative contact, and intravenous therapy. Overall percentages of time were not reported. |
| Westbrook et al. (2008) | Academic | PDA | Attending physicians: direct care 18.0%; indirect care 63.5%. Residents: direct care 16.0%; indirect care 66.7%. Interns: direct care 11%; indirect care 85%. | Communication 33%; social activities 17%; indirect care 17%; direct care 15%; documentation 9%; medication tasks 7%; supervision or education 7%; transit 6%; discharge summary 5%; administrative tasks 2%; answering pager 0.8% |
Methodological quality also varied. Of the 11 time‐motion studies, the total amount of time subjects were observed in the studies ranged from 48 to 720 hours, with a mean of 254 hours. The number of subjects observed varied between 1 and 35, with a mean of 12 subjects. Average time observed per subject ranged from 8 hours to 113.5 hours, with a median of 26 hours. Six of the 11 studies observed subjects continuously for an entire shift.22, 25, 2831 Four studies covered an entire shift over the course of several days, using shorter observation periods.20, 21, 26, 27 One study observed subjects for only part of a shift.32 Ten of the time‐flow articles reported collecting data with a stopwatch and paper‐and‐pencil form2022, 25, 2732 and 1 used a handheld computer system.26 Two studies utilized work‐sampling techniques, both using paper‐and‐pencil forms to collect data during a full shift. Ammenwerth and Spotl23 studied 8 physicians for a total of 40 hours, collecting 5500 observation points. Nerenz et al.24 studied 11 interns for a total of approximately 330 hours, and collected 7858 observations. Both of these studies collected sufficiently large samples to satisfy the power requirements described by Sittig.3
Study sites were relatively uniform. Only one study evaluated physicians at both a teaching community hospital and an academic hospital.32 The remaining 12 observed physicians only in academic hospitals. Two studies were conducted in Australia,25, 26 1 in Austria,23 and the remaining 10 were conducted in the United States.
To provide a rough estimate of the amount of time physicians spend on direct care activities at the patients' bedside vs. indirect care activities, we attempted to calculate these figures for each article using a common definition. For the sake of consistency and to allow us to include as many studies as possible, we used the broadest definition of indirect care found among the articles, which included activities such as professional communication, medication review, documentation, and reviewing test results. Three articles did not provide enough information to calculate these values.24, 27, 29
All 10 articles that did provide sufficient information found that indirect care activities consumed the greater portion of time. Indirect care occupied an average of 50% of physicians' time, ranging from 32% to 69%. Direct care, on the other hand, accounted for an average of 23% of physicians' time, and ranged from 8.5% to 41%. Three articles that included data specific to attending physicians or hospitalists demonstrated an even larger disparity between direct and indirect care.20, 26, 32 In these articles, physicians spent an average of 19% of their time on direct care and 64% on indirect care, suggesting that senior physicians in the academic setting spend less time with patients and more time on care activities away from patients.
Four studies recorded various forms of interruptions of work flow.20, 24, 26, 31 Lurie et al.31 found that interns and residents were interrupted approximately 9 minutes into the performance of every history and physical (H&P). Westbrook et al.26 found that residents were interrupted on average every 21 minutes regardless of the task being performed. Nerenz et al.24 reported that interns received an average of 21 pages over the course of a 30 hour shift. They also noted that, on average, 12 of these pages were merely transient distractions, but 9 pages required some action on the part of the intern.24 Finally, O'leary et al.20 found that hospitalists received an average of 3.5 pages an hour and that 7% of their day was spent returning pages. Two articles recorded events of multitasking. Westbrook et al.26 found that 20% of physicians' time was spent performing more than one activity. Similarly, O'Leary et al.20 reported that 21% of hospitalists' time was spent multitasking. Neither study reported the types of activity performed during multitasking.
One article considered the amount of time physicians spend performing tasks that could be performed by non‐physician staff. Knickman et al.21 reported that in the traditional physician‐centered model of care, approximately 19% of a resident's time is spent on tasks that could be performed by non‐physician staff. They suggested that switching to a mid‐level provider model of care could significantly reduce the impact of resident work hour restrictions.21
Parenti and Lurie28 examined internal medicine residents on both day and night shifts.31 These authors concluded that residents on the night shift have an easier time because they see fewer patients and have more down time than residents on day shifts.28 Additionally, Lurie et al. found that residents got an average of 230 minutes (3.8 hours) of sleep per night and slept, on average, 59 minutes before being awakened by an interruption.31 However, these studies preceded work hour regulations.
Discussion
This systematic review of time studies set in the hospital, the first of which we are aware, revealed a sizable number (13) of articles evaluating physicians. However, the studies almost exclusively focused on academic hospitals (92%) and the majority (69%) analyzed only the activities of physicians in training. The studies were diverse in their methodology, subject populations, and, not surprisingly, their results. Even those studies designed simply to document the activities of physicians in the hospital report widely varying findings. For example, the percentage of time physicians spent on direct‐care activities varied from 8.5% to 41%, while indirect‐care time varied from 32% to 69%. These results likely reflect the heterogeneity of the hospital environment and differences among hospitals, as well as variations in the design and quality of the studies.
Despite this variability, a few observations appear consistent. Physicians perform many tasks that may be readily accomplished by less costly staff. This could partly explain why far more time is spent on activities indirectly related to a patient's care (eg, documentation and coordinating tests), instead of directly interacting with hospitalized patients. Additionally, physicians caring for hospitalized patients experience multiple interruptions and must regularly multitask. Unfortunately, very little research in the hospital setting has evaluated the impact of these interruptions on work efficiency, medical errors, or adverse events.
With the intense national interest in improving the value of healthcare by both enhancing quality and reducing costs, further efforts to optimize the efficiency of hospitalists will be needed.33 As hospitals and hospitalists aim to enhance the efficiency of care delivery to hospitalized patients, and also are increasingly asked to expend time to optimize the hospital discharge process to reduce readmissions,34, 35 time‐motion and work‐sampling studies can provide guidance.
One of the principal difficulties in aggregating data from time studies is the variety of approaches used to analyze activities. Lack of standardization in the approach to assessing physician activities (eg, use of a stopwatch with paper documentation vs. computer) and dissimilar categorizations inhibit efforts to summarize the findings across studies. Categories of activity were generally selected with the specific goals of the study in mind, instead of utilizing a readily available standardized approach. Moreover, the lack of detailed definitions of categories and sub‐categories, along with data for each, produces a significant barrier to comparison. Based on this review of available literature and our own experience conducting time‐motion evaluation of hospitalists, we propose the basic activity categorization in Table 2. Future researchers would be able to more readily compare their findings to other time‐motion studies by utilizing such a standardized approach to categorizing physician activities. Adding custom sub‐categories within this basic set would allow researchers to explore more specific time‐flow questions while maintaining comparability of most data. Electronic data collection tools (eg, handheld or tablet computers) could also facilitate the collection of more detailed and accurate data to increase study reliability.
| Primary | Secondary | Tertiary |
|---|---|---|
| Direct patient care | Daily rounds | Evaluation |
| Education | ||
| Admission history and physical | ||
| Consultation history and physical | ||
| Discharge | Evaluation | |
| Education | ||
| Procedures | ||
| Indirect care | Reviewing test results | |
| Documentation | Orders | |
| History and physical | ||
| Progress notes | ||
| Discharge paperwork | ||
| Communication | Paging | |
| Patient relatives | ||
| Other physicians | ||
| Nurse | ||
| Ancillary staff | ||
| Other | Education | |
| Transit/travel | ||
| Personal (eg, eating, restroom) | ||
| Miscellaneous |
Our systematic review is limited in its scope, as we focused only on the activities of physicians working in the hospital. Our exclusion criteria also eliminated several more focused time studies that evaluated only one small part of a physician's workflow, such as Amusan et al.'s36 evaluation of EMR and CPOE implementation during morning rounds. The available literature itself is also lacking in several important ways. Much of the literature is now limited by its age. The constant advance of medical technology, changes in work hour regulations, and new reimbursement structures have all affected physician workflow, and likely contributed to the variability of time study findings. Additionally, the available literature focuses almost exclusively on academic hospitals and teaching services. All but 1 of the studies collected data exclusively in academic hospitals, despite the fact that more than 90% of hospital care delivery in the U.S. occurs in a non‐academic hospital setting.20, 37 Just 1 study evaluated the activity of hospitalists directly caring for patients without assistance from residents.20 The significantly different workforce composition in community hospitals could mean that most findings are not relevant to the vast majority of U.S. hospitals. For example, the studies documenting that physicians in training (ie, residents) perform many activities that could be performed by a non‐physician are likely not applicable to the community hospital setting. Thus, additional research is needed to better understand how hospitalists can deliver care more efficiently, particularly in the community hospital setting and in the current technological and structural environment of healthcare.
This systematic review of the literature provides insight into published studies attempting to evaluate physician activities in the hospital through time‐motion and work‐sampling studies. Published research to date appears extremely variable in quality, limiting our ability to draw firm conclusions. However, it appears that hospital‐based physicians spend most of their time not interacting with patients, and non‐physician staff could readily complete a sizable portion of their tasks. Given the necessity for multitasking by hospitalists, better documentation of its frequency and impact is needed, as well as information about the types of tasks performed while multitasking, which has yet to be reported. Additionally, the effect of interruptions (including, but not limited to paging) needs further evaluation.
When properly performed, time‐study methodology represents a powerful approach to understanding the activities of hospitalists and how we might reengineer hospital care delivery to be more efficient. Efforts to standardize healthcare delivery and integrate health information technology could benefit dramatically from detailed information regarding physician activities and empirical testing of quality improvement initiatives. Future research using time‐motion or work‐sampling methodology should be careful to define and report categories of activity with enough detail that comparisons with other studies are possible.
Acknowledgements
The authors acknowledge the assistance of Linda O'Dwyer, MA MSLIS, research librarian at the Northwestern University Feinberg School of Medicine for her assistance with the search of the medical literature.
- .Motion and Time Study: Design and Measurement of Work.6th ed.New York:Wiley;1968.
- .Statistical methods in textile research. Uses of the binomial and poissant distributions.J Textile Inst Trans.1935;26:51–55.
- .Work‐sampling: a statistical approach to evaluation of the effect of computers on work patterns in the healthcare industry.Proc Annu Symp Comput Appl Med Care.1992:537–541.
- , , , , , .Time motion study in a pediatric emergency department before and after computer physician order entry.Ann Emerg Med.2009;53(4):462–468, e461.
- , , , .A “time‐and‐motion” study of endoscopic practice: strategies to enhance efficiency.Gastrointest Endosc.2008;68(6):1043–1050.
- , , , , , .Workflow in intensive care unit remote monitoring: A time‐and‐motion study.Crit Care Med.2007;35(9):2057–2063.
- , , , et al.Workflow analysis of medical staff in surgical wards based on time‐motion study data.Jpn Hosp.2008(27):75–80.
- , , , , , .General and visceral surgery practice in German hospitals: a real‐time work analysis on surgeons' work flow.Langenbecks Arch Surg.2010;395(1):81–87.
- , , , et al.Electronic health records in specialty care: a time‐motion study.J Am Med Inform Assoc.2007;14(5):609–615.
- , , , .National health spending in 2007: slower drug spending contributes to lowest rate of overall growth since 1998.Health Aff (Millwood).2009;28(1):246–261.
- , .The challenge of rising health care costs–a view from the Congressional Budget Office.N Engl J Med.2007;357(18):1793–1795.
- .Nonpayment for performance? Medicare's new reimbursement rule.N Engl J Med.2007;357(16):1573–1575.
- , .Hospitalists in teaching hospitals: opportunities but not without danger.J Gen Intern Med.2004;19:392–393.
- .The future of hospital medicine: evolution or revolution?Am J Med.2004;117:446–450.
- , .The evolution and future of hospital medicine.Mt Sinai J Med.2008;75(5):418–423.
- , , , , , .Outcomes of care by hospitalists, general internists, and family physicians.[see comment].N Engl J Med.2007;357(25):2589–2600.
- , .The hospitalist movement 5 years later.JAMA.2002;287:487–494.
- , .The impact of hospitalists on the cost and quality of inpatient care in the United States: a research synthesis.Med Care Res Rev.2005;62:379–406.
- .Hospitalists and the hospital medicine system of care are good for patient care.Arch Intern Med.2008;168(12):1254–1256; discussion 1259–1260.
- , , .How hospitalists spend their time: insights on efficiency and safety.J Hosp Med.2006;1(2):88–93.
- , , , , .The potential for using non‐physicians to compensate for the reduced availability of residents.Acad Med.1992;67(7):429–438.
- , , , , , .Observations of residents' work activities for 24 consecutive hours: Implications for workflow redesign.Acad Med.2006;81(8):766–775.
- , .The time needed for clinical documentation versus direct patient care. A work‐sampling analysis of physicians' activities.Methods Inf Med.2009;48(1):84–91.
- , , , et al.The on‐call experience of interns in internal medicine. Medical Education Task Force of Henry Ford Hospital.Arch Intern Med.1990;150(11):2294–2297.
- , , .What does the intern do?Med J Aust.1976;1(3):63–65.
- , , , .All in a day's work: an observational study to quantify how and with whom doctors on hospital wards spend their time.Med J Aust.2008;188(9):506–509.
- , , , .Resident educational time study: a tale of three specialties.Acad Emerg Med.1998;5(7):718–725.
- , .Are things different in the light of day? A time study of internal medicine house staff days.Am J Med.1993;94(6):654–658.
- , , .Time study of an internship on a university medical service.N Engl J Med.1961;264:439–443.
- , .Time study comparisons of 3 intern programs.J Med Educ.1971;46(2):142–149.
- , , , , .How do house officers spend their nights? A time study of internal medicine house staff on call.N Engl J Med.1989;320(25):1673–1677.
- .The Effect of a Non‐teaching Hospitalist Service in an Academic Hospital Setting: A Comparative Time‐motion and Clinical Outcomes Study. Unpublished Manuscript. Vol2006.
- .A strategy for health care reform—toward a value‐based system.N Engl J Med.2009;361(2):109–112.
- , , .Rehospitalizations among patients in the fee‐for‐service medicare program.N Engl J Med.2009;360(14):1418–1428.
- , .BOOSTing the hospital discharge.J Hosp Med.2009;4(4):209–210.
- , , , .A time‐motion study to evaluate the impact of EMR and CPOE implementation on physician efficiency.J Healthc Inf Manag.2008;22(4):31–37.
- , , , , .The ecology of medical care revisited.N Engl J Med.2001;344(26):2021–2025.
Time‐motion studies, introduced by Frederick W. Taylor in the 1880s,1 have been widely implemented across the business world; a Google search of time‐motion study returns approximately 32 million results. Such studies continuously document how workers spend their time and then use this information to identify and eliminate inefficient practices. Work‐sampling is a similar methodology introduced by L.H.C. Tippett in 1935.2 Work‐sampling utilizes a trained observer to document activities at predetermined or random intervals of time. Given a large enough number of observations, this method can be comparable to the continuous observation method used in time‐motion studies.3
Healthcare has begun to utilize these time‐study methodologies to evaluate the activities of physicians and nurses. Researchers have successfully used time‐study methodology in the emergency department, intensive care unit, and ambulatory and surgical settings in the U.S. and around the world to better understand physician activities and to design and assess interventions to improve efficiency.49 Hospitals are also eager to enhance efficiency in the inpatient setting given the current economic environment. Hospitalizations account for over a third of healthcare costs in the United States, making them an attractive target for cost‐cutting measures.10 Acknowledging that healthcare expenditures cannot continue to rise,11 insurers, particularly the Centers for Medicare and Medicaid Services (CMS), increasingly seek to reduce payments to hospitals.12 Compounding these pressures, a major supply of relatively inexpensive labor shrank with the decision by the Accreditation Council for Graduate Medicine Education (ACGME) to restrict the number of hours residents are allowed to work. Efficiency concerns gain new urgency as hospitals scramble to cover their patient loads with reduced physician availability.13
We undertook a systematic review of time‐motion and work‐sampling studies performed in the hospital setting to better understand the available literature describing the activities of physicians caring for hospitalized patients. An additional goal of this review was to determine the extent of available time‐flow literature describing the activity of hospitalists. The hospitalist movement provided one viable solution to the gap between demand for hospital patient care and the reduced supply of available physicianstypically primary care physicians in community hospitals and residents in teaching hospitals.14 Hospital medicine is the fastest‐growing specialty in the history of American medicine.15 More than half of American hospitals now have hospital medicine programs with a total of greater than 25,000 hospitalists in the U.S.15 This popularity has been driven by hospitalists' ability to increase efficiency through decreasing overall cost and length of stay for patients without increasing readmission rates or reducing primary care physician satisfaction.1619 However, exactly how hospitalists accomplish this increase in efficiency is still the subject of debate. One time‐motion study provides a glimpse into the activities of hospitalists at an academic urban hospital,20 but may not be applicable to many other hospitals.
Methods
Data Sources
With assistance from a medical librarian, we searched for English‐language articles published between 1965 and June 2009 using the MEDLINE (
Study Selection
We selected articles that met the following criteria: (1) explicit use of time‐motion methodology or work‐sampling performed via direct observation; (2) study populations including physicians, medical residents, or interns; (3) performance sites on an inpatient hospital ward (ie, not outpatient within the hospital, emergency room (ER), or operating room (OR)); and (4) observation of at least half of a shift. Titles and abstracts of the retrieved citations were first reviewed to identify studies that could potentially meet our criteria. Full‐text versions of the selected articles were then retrieved and analyzed by at least 2 of 4 authors (V.F., K.E., D.M., and M.T.) to determine the final list of articles. Articles that failed to provide sufficient information for one or more criteria were excluded.
Data Extraction
Each article was independently reviewed by at least 2 of 3 authors (V.F., D.M., and M.T.) using a standardized data abstraction form. The form included the following categories: sample population, sample size, hospital type, data collection tool type, time‐motion/observation duration, key categories of activity, and key results. If an article included additional data beyond the scope of this review (eg, data from surgical residents in the OR as well as internal medicine residents) only the qualifying portion of the study was included. Disagreements were resolved through discussion and consensus. Data were then compiled into tables.
Results
Our database search yielded 4270 potential articles. We then reviewed the title and abstract of each of these articles to identify studies that evaluated physicians, were performed on a hospital ward, and explicitly used time‐motion or direct‐observation work‐sampling methodology. For articles lacking an abstract but having a relevant title, we obtained the full text to determine eligibility for additional review. Sixty‐eight articles from this original search were selected for full‐text review. Ten of these articles met the selection criteria. Most of the articles excluded in this step were either conducted in an outpatient OR or ER setting, or used self‐report data instead of direct‐observation data. A secondary search using the reference lists of all obtained articles as well as consultation with experts in the field yielded 11 additional articles of interest. Three of these 11 articles were found to meet our criteria, bringing the total to 13 articles for review (Fig. 1).
These 13 articles included several types of physicians in their samples. Eleven included interns,2131 7 included residents,2123, 2628, 31 and 4 included attending physicians20, 23, 26, 32 (Table 1). Six articles included more than 1 type of subject.2123, 26, 28, 31 The main focus of these articles also varied. Nine of the 13 studies were designed to simply describe how residents, physicians and nurses spend their time.20, 2227, 29, 31 Three studies were primarily concerned with comparing groups from different intern programs, residency rotations, hospital types, or shifts.28, 30, 32 The remaining study attempted to quantify the amount of time physicians spent on tasks that could be performed by non‐physician staff.21 Only 2 articles evaluated hospitalists,20, 32 and we found no articles studying hospitalists in a community, non‐teaching setting. The studies were performed as early as 1961 and as recently as 2009. Just 5 of the 13 articles were published within the last 10 years. 0

| Reference (Year) | Type of Hospital | Data Collection Tool | Direct/Indirect Care Estimates | Key Activities Reported and Percentage of Time Spent on Each, and Other Results |
|---|---|---|---|---|
| ||||
| Ammenwerth and Spotl (2009) | Academic | Work sampling: paper and stopwatch | Direct care 27.5%; indirect care 62.8% | Documentation tasks 26.6%; direct care 27.5%; communication 36.2%; other tasks 9.7%. Approximately 16% of documentation time was administrative documentation. |
| Arthurson et al. (1976) | Academic | Paper and stopwatch | Direct care 40.8%; indirect care 45.5% | Medical intern: patient care 40.8%; clerical 25.5%; telephone 10%; professional discussion 10.5%; transit/waiting 6.5%, personal 7% |
| Gabow et al. (2006) | Academic | Paper and stopwatch with pedometer | Direct care 19.5%; indirect care: 32% | Attend to/assess patient 17%; charting 9.5%; consult with MD/nurse 9.5%; downtime 6.5%; educational activity 2.5%; family interaction 0.5%; paging/phone 3.5%; procedure 1.5%; review films/laboratory results 9%; rounds 15.5%; sleep 21.5%; travel 3.5% |
| Gillanders et al. (1971) | Academic | Paper and stopwatch | Direct care 19%; indirect care 42.5% | Individual interaction 14.5%; nonverbal communication 20.5%; procedures 10.0%; laboratory work 3.5%; interpersonal communication 18.5%; rounds 9%; direction and supervision 0.5%; nonmedical talk 2%; education 8.5%; ancillary activities 5.5%; personal 7% |
| Knickman et al. (1992) | Academic | Paper and stopwatch | Direct care 8.5%; indirect care 37.7% | Education 20.7%; information gathering 13.7%; personal 13.3%; testing 12%; consulting 12%; documenting 9.8%; transit 8.2%; procedures 5.4%; interacting with patients 3.1%; administration 1.8%. 46.7% of residents' time was spent on tasks that required a physician. |
| Lurie et al. (1989) | Academic | Paper and stopwatch | Direct care 17.4%; indirect care 39.3% | Procedures 3%; patient evaluation 20%; communication 27%; basic 40%; miscellaneous 10%. On average, doctors were interrupted 9 minutes into an H&P, got 230 minutes of sleep per night, and slept 59 minutes before being woken up by some interruption. |
| Magnusson et al. (1998) | Academic | Paper and stopwatch | Could not be determined | Clinical 54%; education 28%; personal 18%. The 3 specialties did not differ significantly in time spent on these categories except for education time: emergency 24%; internal 28%; surgery 18%. |
| Malkenson (unpublished data) | 1 Community; 1 Academic | Paper and stopwatch | Academic: direct care 19%; indirect care 56%. Community: direct care 25%; indirect care 55%. | Direct patient care (25% community, 19% academic); indirect patient care (55% community, 56% academic); personal time (4% community, 6% academic); travel time (10% community, 10% academic); other activities (10% community, 13% academic) |
| Nerenz et al. (1990) | Academic | Work sampling: paper and stopwatch | Direct care 18.9%; indirect care could not be determined | Interns averaged 21 pages over 30 hours of observation, and slept an average of 2.5 hours with 2 interruptions. Attending physicians interacted with the interns for an average of 139 minutes per shift. |
| O'Leary et al. (2006) | Academic | Paper and stopwatch | Direct care 18%; indirect care 69% | Indirect patient care occupied 69% of hospitalists' time. Indirect care included: documentation 37%; communication 35%; reviewing results 21%; orders 7%. Direct care occupied 18%, and included: history and physical 18%; follow‐up visits 53%; family meetings 13%; discharge instructions 16%. Remaining time was spent on personal activities 4%; professional development 3%; education 3%; travel 3%. |
| Parenti et al. (1993) | Academic | Paper and stopwatch | Interns: direct care 39%; indirect care 51%. Residents: direct care 40%; indirect care 47%. | Interns: procedures 4%; patient evaluation 35%; communication 42%; basics 11%; miscellaneous 8%. Residents: procedures 2%; patient evaluation 38%; communication 35%; basics 12%; miscellaneous 13%. |
| Payson et al. (1961) | Academic | Paper and stopwatch | Could not be determined | Communication with staff took up the largest amount of time. Remaining time was evenly distributed between the categories of personal activities, ancillary duties, patient and relative contact, and intravenous therapy. Overall percentages of time were not reported. |
| Westbrook et al. (2008) | Academic | PDA | Attending physicians: direct care 18.0%; indirect care 63.5%. Residents: direct care 16.0%; indirect care 66.7%. Interns: direct care 11%; indirect care 85%. | Communication 33%; social activities 17%; indirect care 17%; direct care 15%; documentation 9%; medication tasks 7%; supervision or education 7%; transit 6%; discharge summary 5%; administrative tasks 2%; answering pager 0.8% |
Methodological quality also varied. Of the 11 time‐motion studies, the total amount of time subjects were observed in the studies ranged from 48 to 720 hours, with a mean of 254 hours. The number of subjects observed varied between 1 and 35, with a mean of 12 subjects. Average time observed per subject ranged from 8 hours to 113.5 hours, with a median of 26 hours. Six of the 11 studies observed subjects continuously for an entire shift.22, 25, 2831 Four studies covered an entire shift over the course of several days, using shorter observation periods.20, 21, 26, 27 One study observed subjects for only part of a shift.32 Ten of the time‐flow articles reported collecting data with a stopwatch and paper‐and‐pencil form2022, 25, 2732 and 1 used a handheld computer system.26 Two studies utilized work‐sampling techniques, both using paper‐and‐pencil forms to collect data during a full shift. Ammenwerth and Spotl23 studied 8 physicians for a total of 40 hours, collecting 5500 observation points. Nerenz et al.24 studied 11 interns for a total of approximately 330 hours, and collected 7858 observations. Both of these studies collected sufficiently large samples to satisfy the power requirements described by Sittig.3
Study sites were relatively uniform. Only one study evaluated physicians at both a teaching community hospital and an academic hospital.32 The remaining 12 observed physicians only in academic hospitals. Two studies were conducted in Australia,25, 26 1 in Austria,23 and the remaining 10 were conducted in the United States.
To provide a rough estimate of the amount of time physicians spend on direct care activities at the patients' bedside vs. indirect care activities, we attempted to calculate these figures for each article using a common definition. For the sake of consistency and to allow us to include as many studies as possible, we used the broadest definition of indirect care found among the articles, which included activities such as professional communication, medication review, documentation, and reviewing test results. Three articles did not provide enough information to calculate these values.24, 27, 29
All 10 articles that did provide sufficient information found that indirect care activities consumed the greater portion of time. Indirect care occupied an average of 50% of physicians' time, ranging from 32% to 69%. Direct care, on the other hand, accounted for an average of 23% of physicians' time, and ranged from 8.5% to 41%. Three articles that included data specific to attending physicians or hospitalists demonstrated an even larger disparity between direct and indirect care.20, 26, 32 In these articles, physicians spent an average of 19% of their time on direct care and 64% on indirect care, suggesting that senior physicians in the academic setting spend less time with patients and more time on care activities away from patients.
Four studies recorded various forms of interruptions of work flow.20, 24, 26, 31 Lurie et al.31 found that interns and residents were interrupted approximately 9 minutes into the performance of every history and physical (H&P). Westbrook et al.26 found that residents were interrupted on average every 21 minutes regardless of the task being performed. Nerenz et al.24 reported that interns received an average of 21 pages over the course of a 30 hour shift. They also noted that, on average, 12 of these pages were merely transient distractions, but 9 pages required some action on the part of the intern.24 Finally, O'leary et al.20 found that hospitalists received an average of 3.5 pages an hour and that 7% of their day was spent returning pages. Two articles recorded events of multitasking. Westbrook et al.26 found that 20% of physicians' time was spent performing more than one activity. Similarly, O'Leary et al.20 reported that 21% of hospitalists' time was spent multitasking. Neither study reported the types of activity performed during multitasking.
One article considered the amount of time physicians spend performing tasks that could be performed by non‐physician staff. Knickman et al.21 reported that in the traditional physician‐centered model of care, approximately 19% of a resident's time is spent on tasks that could be performed by non‐physician staff. They suggested that switching to a mid‐level provider model of care could significantly reduce the impact of resident work hour restrictions.21
Parenti and Lurie28 examined internal medicine residents on both day and night shifts.31 These authors concluded that residents on the night shift have an easier time because they see fewer patients and have more down time than residents on day shifts.28 Additionally, Lurie et al. found that residents got an average of 230 minutes (3.8 hours) of sleep per night and slept, on average, 59 minutes before being awakened by an interruption.31 However, these studies preceded work hour regulations.
Discussion
This systematic review of time studies set in the hospital, the first of which we are aware, revealed a sizable number (13) of articles evaluating physicians. However, the studies almost exclusively focused on academic hospitals (92%) and the majority (69%) analyzed only the activities of physicians in training. The studies were diverse in their methodology, subject populations, and, not surprisingly, their results. Even those studies designed simply to document the activities of physicians in the hospital report widely varying findings. For example, the percentage of time physicians spent on direct‐care activities varied from 8.5% to 41%, while indirect‐care time varied from 32% to 69%. These results likely reflect the heterogeneity of the hospital environment and differences among hospitals, as well as variations in the design and quality of the studies.
Despite this variability, a few observations appear consistent. Physicians perform many tasks that may be readily accomplished by less costly staff. This could partly explain why far more time is spent on activities indirectly related to a patient's care (eg, documentation and coordinating tests), instead of directly interacting with hospitalized patients. Additionally, physicians caring for hospitalized patients experience multiple interruptions and must regularly multitask. Unfortunately, very little research in the hospital setting has evaluated the impact of these interruptions on work efficiency, medical errors, or adverse events.
With the intense national interest in improving the value of healthcare by both enhancing quality and reducing costs, further efforts to optimize the efficiency of hospitalists will be needed.33 As hospitals and hospitalists aim to enhance the efficiency of care delivery to hospitalized patients, and also are increasingly asked to expend time to optimize the hospital discharge process to reduce readmissions,34, 35 time‐motion and work‐sampling studies can provide guidance.
One of the principal difficulties in aggregating data from time studies is the variety of approaches used to analyze activities. Lack of standardization in the approach to assessing physician activities (eg, use of a stopwatch with paper documentation vs. computer) and dissimilar categorizations inhibit efforts to summarize the findings across studies. Categories of activity were generally selected with the specific goals of the study in mind, instead of utilizing a readily available standardized approach. Moreover, the lack of detailed definitions of categories and sub‐categories, along with data for each, produces a significant barrier to comparison. Based on this review of available literature and our own experience conducting time‐motion evaluation of hospitalists, we propose the basic activity categorization in Table 2. Future researchers would be able to more readily compare their findings to other time‐motion studies by utilizing such a standardized approach to categorizing physician activities. Adding custom sub‐categories within this basic set would allow researchers to explore more specific time‐flow questions while maintaining comparability of most data. Electronic data collection tools (eg, handheld or tablet computers) could also facilitate the collection of more detailed and accurate data to increase study reliability.
| Primary | Secondary | Tertiary |
|---|---|---|
| Direct patient care | Daily rounds | Evaluation |
| Education | ||
| Admission history and physical | ||
| Consultation history and physical | ||
| Discharge | Evaluation | |
| Education | ||
| Procedures | ||
| Indirect care | Reviewing test results | |
| Documentation | Orders | |
| History and physical | ||
| Progress notes | ||
| Discharge paperwork | ||
| Communication | Paging | |
| Patient relatives | ||
| Other physicians | ||
| Nurse | ||
| Ancillary staff | ||
| Other | Education | |
| Transit/travel | ||
| Personal (eg, eating, restroom) | ||
| Miscellaneous |
Our systematic review is limited in its scope, as we focused only on the activities of physicians working in the hospital. Our exclusion criteria also eliminated several more focused time studies that evaluated only one small part of a physician's workflow, such as Amusan et al.'s36 evaluation of EMR and CPOE implementation during morning rounds. The available literature itself is also lacking in several important ways. Much of the literature is now limited by its age. The constant advance of medical technology, changes in work hour regulations, and new reimbursement structures have all affected physician workflow, and likely contributed to the variability of time study findings. Additionally, the available literature focuses almost exclusively on academic hospitals and teaching services. All but 1 of the studies collected data exclusively in academic hospitals, despite the fact that more than 90% of hospital care delivery in the U.S. occurs in a non‐academic hospital setting.20, 37 Just 1 study evaluated the activity of hospitalists directly caring for patients without assistance from residents.20 The significantly different workforce composition in community hospitals could mean that most findings are not relevant to the vast majority of U.S. hospitals. For example, the studies documenting that physicians in training (ie, residents) perform many activities that could be performed by a non‐physician are likely not applicable to the community hospital setting. Thus, additional research is needed to better understand how hospitalists can deliver care more efficiently, particularly in the community hospital setting and in the current technological and structural environment of healthcare.
This systematic review of the literature provides insight into published studies attempting to evaluate physician activities in the hospital through time‐motion and work‐sampling studies. Published research to date appears extremely variable in quality, limiting our ability to draw firm conclusions. However, it appears that hospital‐based physicians spend most of their time not interacting with patients, and non‐physician staff could readily complete a sizable portion of their tasks. Given the necessity for multitasking by hospitalists, better documentation of its frequency and impact is needed, as well as information about the types of tasks performed while multitasking, which has yet to be reported. Additionally, the effect of interruptions (including, but not limited to paging) needs further evaluation.
When properly performed, time‐study methodology represents a powerful approach to understanding the activities of hospitalists and how we might reengineer hospital care delivery to be more efficient. Efforts to standardize healthcare delivery and integrate health information technology could benefit dramatically from detailed information regarding physician activities and empirical testing of quality improvement initiatives. Future research using time‐motion or work‐sampling methodology should be careful to define and report categories of activity with enough detail that comparisons with other studies are possible.
Acknowledgements
The authors acknowledge the assistance of Linda O'Dwyer, MA MSLIS, research librarian at the Northwestern University Feinberg School of Medicine for her assistance with the search of the medical literature.
Time‐motion studies, introduced by Frederick W. Taylor in the 1880s,1 have been widely implemented across the business world; a Google search of time‐motion study returns approximately 32 million results. Such studies continuously document how workers spend their time and then use this information to identify and eliminate inefficient practices. Work‐sampling is a similar methodology introduced by L.H.C. Tippett in 1935.2 Work‐sampling utilizes a trained observer to document activities at predetermined or random intervals of time. Given a large enough number of observations, this method can be comparable to the continuous observation method used in time‐motion studies.3
Healthcare has begun to utilize these time‐study methodologies to evaluate the activities of physicians and nurses. Researchers have successfully used time‐study methodology in the emergency department, intensive care unit, and ambulatory and surgical settings in the U.S. and around the world to better understand physician activities and to design and assess interventions to improve efficiency.49 Hospitals are also eager to enhance efficiency in the inpatient setting given the current economic environment. Hospitalizations account for over a third of healthcare costs in the United States, making them an attractive target for cost‐cutting measures.10 Acknowledging that healthcare expenditures cannot continue to rise,11 insurers, particularly the Centers for Medicare and Medicaid Services (CMS), increasingly seek to reduce payments to hospitals.12 Compounding these pressures, a major supply of relatively inexpensive labor shrank with the decision by the Accreditation Council for Graduate Medicine Education (ACGME) to restrict the number of hours residents are allowed to work. Efficiency concerns gain new urgency as hospitals scramble to cover their patient loads with reduced physician availability.13
We undertook a systematic review of time‐motion and work‐sampling studies performed in the hospital setting to better understand the available literature describing the activities of physicians caring for hospitalized patients. An additional goal of this review was to determine the extent of available time‐flow literature describing the activity of hospitalists. The hospitalist movement provided one viable solution to the gap between demand for hospital patient care and the reduced supply of available physicianstypically primary care physicians in community hospitals and residents in teaching hospitals.14 Hospital medicine is the fastest‐growing specialty in the history of American medicine.15 More than half of American hospitals now have hospital medicine programs with a total of greater than 25,000 hospitalists in the U.S.15 This popularity has been driven by hospitalists' ability to increase efficiency through decreasing overall cost and length of stay for patients without increasing readmission rates or reducing primary care physician satisfaction.1619 However, exactly how hospitalists accomplish this increase in efficiency is still the subject of debate. One time‐motion study provides a glimpse into the activities of hospitalists at an academic urban hospital,20 but may not be applicable to many other hospitals.
Methods
Data Sources
With assistance from a medical librarian, we searched for English‐language articles published between 1965 and June 2009 using the MEDLINE (
Study Selection
We selected articles that met the following criteria: (1) explicit use of time‐motion methodology or work‐sampling performed via direct observation; (2) study populations including physicians, medical residents, or interns; (3) performance sites on an inpatient hospital ward (ie, not outpatient within the hospital, emergency room (ER), or operating room (OR)); and (4) observation of at least half of a shift. Titles and abstracts of the retrieved citations were first reviewed to identify studies that could potentially meet our criteria. Full‐text versions of the selected articles were then retrieved and analyzed by at least 2 of 4 authors (V.F., K.E., D.M., and M.T.) to determine the final list of articles. Articles that failed to provide sufficient information for one or more criteria were excluded.
Data Extraction
Each article was independently reviewed by at least 2 of 3 authors (V.F., D.M., and M.T.) using a standardized data abstraction form. The form included the following categories: sample population, sample size, hospital type, data collection tool type, time‐motion/observation duration, key categories of activity, and key results. If an article included additional data beyond the scope of this review (eg, data from surgical residents in the OR as well as internal medicine residents) only the qualifying portion of the study was included. Disagreements were resolved through discussion and consensus. Data were then compiled into tables.
Results
Our database search yielded 4270 potential articles. We then reviewed the title and abstract of each of these articles to identify studies that evaluated physicians, were performed on a hospital ward, and explicitly used time‐motion or direct‐observation work‐sampling methodology. For articles lacking an abstract but having a relevant title, we obtained the full text to determine eligibility for additional review. Sixty‐eight articles from this original search were selected for full‐text review. Ten of these articles met the selection criteria. Most of the articles excluded in this step were either conducted in an outpatient OR or ER setting, or used self‐report data instead of direct‐observation data. A secondary search using the reference lists of all obtained articles as well as consultation with experts in the field yielded 11 additional articles of interest. Three of these 11 articles were found to meet our criteria, bringing the total to 13 articles for review (Fig. 1).
These 13 articles included several types of physicians in their samples. Eleven included interns,2131 7 included residents,2123, 2628, 31 and 4 included attending physicians20, 23, 26, 32 (Table 1). Six articles included more than 1 type of subject.2123, 26, 28, 31 The main focus of these articles also varied. Nine of the 13 studies were designed to simply describe how residents, physicians and nurses spend their time.20, 2227, 29, 31 Three studies were primarily concerned with comparing groups from different intern programs, residency rotations, hospital types, or shifts.28, 30, 32 The remaining study attempted to quantify the amount of time physicians spent on tasks that could be performed by non‐physician staff.21 Only 2 articles evaluated hospitalists,20, 32 and we found no articles studying hospitalists in a community, non‐teaching setting. The studies were performed as early as 1961 and as recently as 2009. Just 5 of the 13 articles were published within the last 10 years. 0

| Reference (Year) | Type of Hospital | Data Collection Tool | Direct/Indirect Care Estimates | Key Activities Reported and Percentage of Time Spent on Each, and Other Results |
|---|---|---|---|---|
| ||||
| Ammenwerth and Spotl (2009) | Academic | Work sampling: paper and stopwatch | Direct care 27.5%; indirect care 62.8% | Documentation tasks 26.6%; direct care 27.5%; communication 36.2%; other tasks 9.7%. Approximately 16% of documentation time was administrative documentation. |
| Arthurson et al. (1976) | Academic | Paper and stopwatch | Direct care 40.8%; indirect care 45.5% | Medical intern: patient care 40.8%; clerical 25.5%; telephone 10%; professional discussion 10.5%; transit/waiting 6.5%, personal 7% |
| Gabow et al. (2006) | Academic | Paper and stopwatch with pedometer | Direct care 19.5%; indirect care: 32% | Attend to/assess patient 17%; charting 9.5%; consult with MD/nurse 9.5%; downtime 6.5%; educational activity 2.5%; family interaction 0.5%; paging/phone 3.5%; procedure 1.5%; review films/laboratory results 9%; rounds 15.5%; sleep 21.5%; travel 3.5% |
| Gillanders et al. (1971) | Academic | Paper and stopwatch | Direct care 19%; indirect care 42.5% | Individual interaction 14.5%; nonverbal communication 20.5%; procedures 10.0%; laboratory work 3.5%; interpersonal communication 18.5%; rounds 9%; direction and supervision 0.5%; nonmedical talk 2%; education 8.5%; ancillary activities 5.5%; personal 7% |
| Knickman et al. (1992) | Academic | Paper and stopwatch | Direct care 8.5%; indirect care 37.7% | Education 20.7%; information gathering 13.7%; personal 13.3%; testing 12%; consulting 12%; documenting 9.8%; transit 8.2%; procedures 5.4%; interacting with patients 3.1%; administration 1.8%. 46.7% of residents' time was spent on tasks that required a physician. |
| Lurie et al. (1989) | Academic | Paper and stopwatch | Direct care 17.4%; indirect care 39.3% | Procedures 3%; patient evaluation 20%; communication 27%; basic 40%; miscellaneous 10%. On average, doctors were interrupted 9 minutes into an H&P, got 230 minutes of sleep per night, and slept 59 minutes before being woken up by some interruption. |
| Magnusson et al. (1998) | Academic | Paper and stopwatch | Could not be determined | Clinical 54%; education 28%; personal 18%. The 3 specialties did not differ significantly in time spent on these categories except for education time: emergency 24%; internal 28%; surgery 18%. |
| Malkenson (unpublished data) | 1 Community; 1 Academic | Paper and stopwatch | Academic: direct care 19%; indirect care 56%. Community: direct care 25%; indirect care 55%. | Direct patient care (25% community, 19% academic); indirect patient care (55% community, 56% academic); personal time (4% community, 6% academic); travel time (10% community, 10% academic); other activities (10% community, 13% academic) |
| Nerenz et al. (1990) | Academic | Work sampling: paper and stopwatch | Direct care 18.9%; indirect care could not be determined | Interns averaged 21 pages over 30 hours of observation, and slept an average of 2.5 hours with 2 interruptions. Attending physicians interacted with the interns for an average of 139 minutes per shift. |
| O'Leary et al. (2006) | Academic | Paper and stopwatch | Direct care 18%; indirect care 69% | Indirect patient care occupied 69% of hospitalists' time. Indirect care included: documentation 37%; communication 35%; reviewing results 21%; orders 7%. Direct care occupied 18%, and included: history and physical 18%; follow‐up visits 53%; family meetings 13%; discharge instructions 16%. Remaining time was spent on personal activities 4%; professional development 3%; education 3%; travel 3%. |
| Parenti et al. (1993) | Academic | Paper and stopwatch | Interns: direct care 39%; indirect care 51%. Residents: direct care 40%; indirect care 47%. | Interns: procedures 4%; patient evaluation 35%; communication 42%; basics 11%; miscellaneous 8%. Residents: procedures 2%; patient evaluation 38%; communication 35%; basics 12%; miscellaneous 13%. |
| Payson et al. (1961) | Academic | Paper and stopwatch | Could not be determined | Communication with staff took up the largest amount of time. Remaining time was evenly distributed between the categories of personal activities, ancillary duties, patient and relative contact, and intravenous therapy. Overall percentages of time were not reported. |
| Westbrook et al. (2008) | Academic | PDA | Attending physicians: direct care 18.0%; indirect care 63.5%. Residents: direct care 16.0%; indirect care 66.7%. Interns: direct care 11%; indirect care 85%. | Communication 33%; social activities 17%; indirect care 17%; direct care 15%; documentation 9%; medication tasks 7%; supervision or education 7%; transit 6%; discharge summary 5%; administrative tasks 2%; answering pager 0.8% |
Methodological quality also varied. Of the 11 time‐motion studies, the total amount of time subjects were observed in the studies ranged from 48 to 720 hours, with a mean of 254 hours. The number of subjects observed varied between 1 and 35, with a mean of 12 subjects. Average time observed per subject ranged from 8 hours to 113.5 hours, with a median of 26 hours. Six of the 11 studies observed subjects continuously for an entire shift.22, 25, 2831 Four studies covered an entire shift over the course of several days, using shorter observation periods.20, 21, 26, 27 One study observed subjects for only part of a shift.32 Ten of the time‐flow articles reported collecting data with a stopwatch and paper‐and‐pencil form2022, 25, 2732 and 1 used a handheld computer system.26 Two studies utilized work‐sampling techniques, both using paper‐and‐pencil forms to collect data during a full shift. Ammenwerth and Spotl23 studied 8 physicians for a total of 40 hours, collecting 5500 observation points. Nerenz et al.24 studied 11 interns for a total of approximately 330 hours, and collected 7858 observations. Both of these studies collected sufficiently large samples to satisfy the power requirements described by Sittig.3
Study sites were relatively uniform. Only one study evaluated physicians at both a teaching community hospital and an academic hospital.32 The remaining 12 observed physicians only in academic hospitals. Two studies were conducted in Australia,25, 26 1 in Austria,23 and the remaining 10 were conducted in the United States.
To provide a rough estimate of the amount of time physicians spend on direct care activities at the patients' bedside vs. indirect care activities, we attempted to calculate these figures for each article using a common definition. For the sake of consistency and to allow us to include as many studies as possible, we used the broadest definition of indirect care found among the articles, which included activities such as professional communication, medication review, documentation, and reviewing test results. Three articles did not provide enough information to calculate these values.24, 27, 29
All 10 articles that did provide sufficient information found that indirect care activities consumed the greater portion of time. Indirect care occupied an average of 50% of physicians' time, ranging from 32% to 69%. Direct care, on the other hand, accounted for an average of 23% of physicians' time, and ranged from 8.5% to 41%. Three articles that included data specific to attending physicians or hospitalists demonstrated an even larger disparity between direct and indirect care.20, 26, 32 In these articles, physicians spent an average of 19% of their time on direct care and 64% on indirect care, suggesting that senior physicians in the academic setting spend less time with patients and more time on care activities away from patients.
Four studies recorded various forms of interruptions of work flow.20, 24, 26, 31 Lurie et al.31 found that interns and residents were interrupted approximately 9 minutes into the performance of every history and physical (H&P). Westbrook et al.26 found that residents were interrupted on average every 21 minutes regardless of the task being performed. Nerenz et al.24 reported that interns received an average of 21 pages over the course of a 30 hour shift. They also noted that, on average, 12 of these pages were merely transient distractions, but 9 pages required some action on the part of the intern.24 Finally, O'leary et al.20 found that hospitalists received an average of 3.5 pages an hour and that 7% of their day was spent returning pages. Two articles recorded events of multitasking. Westbrook et al.26 found that 20% of physicians' time was spent performing more than one activity. Similarly, O'Leary et al.20 reported that 21% of hospitalists' time was spent multitasking. Neither study reported the types of activity performed during multitasking.
One article considered the amount of time physicians spend performing tasks that could be performed by non‐physician staff. Knickman et al.21 reported that in the traditional physician‐centered model of care, approximately 19% of a resident's time is spent on tasks that could be performed by non‐physician staff. They suggested that switching to a mid‐level provider model of care could significantly reduce the impact of resident work hour restrictions.21
Parenti and Lurie28 examined internal medicine residents on both day and night shifts.31 These authors concluded that residents on the night shift have an easier time because they see fewer patients and have more down time than residents on day shifts.28 Additionally, Lurie et al. found that residents got an average of 230 minutes (3.8 hours) of sleep per night and slept, on average, 59 minutes before being awakened by an interruption.31 However, these studies preceded work hour regulations.
Discussion
This systematic review of time studies set in the hospital, the first of which we are aware, revealed a sizable number (13) of articles evaluating physicians. However, the studies almost exclusively focused on academic hospitals (92%) and the majority (69%) analyzed only the activities of physicians in training. The studies were diverse in their methodology, subject populations, and, not surprisingly, their results. Even those studies designed simply to document the activities of physicians in the hospital report widely varying findings. For example, the percentage of time physicians spent on direct‐care activities varied from 8.5% to 41%, while indirect‐care time varied from 32% to 69%. These results likely reflect the heterogeneity of the hospital environment and differences among hospitals, as well as variations in the design and quality of the studies.
Despite this variability, a few observations appear consistent. Physicians perform many tasks that may be readily accomplished by less costly staff. This could partly explain why far more time is spent on activities indirectly related to a patient's care (eg, documentation and coordinating tests), instead of directly interacting with hospitalized patients. Additionally, physicians caring for hospitalized patients experience multiple interruptions and must regularly multitask. Unfortunately, very little research in the hospital setting has evaluated the impact of these interruptions on work efficiency, medical errors, or adverse events.
With the intense national interest in improving the value of healthcare by both enhancing quality and reducing costs, further efforts to optimize the efficiency of hospitalists will be needed.33 As hospitals and hospitalists aim to enhance the efficiency of care delivery to hospitalized patients, and also are increasingly asked to expend time to optimize the hospital discharge process to reduce readmissions,34, 35 time‐motion and work‐sampling studies can provide guidance.
One of the principal difficulties in aggregating data from time studies is the variety of approaches used to analyze activities. Lack of standardization in the approach to assessing physician activities (eg, use of a stopwatch with paper documentation vs. computer) and dissimilar categorizations inhibit efforts to summarize the findings across studies. Categories of activity were generally selected with the specific goals of the study in mind, instead of utilizing a readily available standardized approach. Moreover, the lack of detailed definitions of categories and sub‐categories, along with data for each, produces a significant barrier to comparison. Based on this review of available literature and our own experience conducting time‐motion evaluation of hospitalists, we propose the basic activity categorization in Table 2. Future researchers would be able to more readily compare their findings to other time‐motion studies by utilizing such a standardized approach to categorizing physician activities. Adding custom sub‐categories within this basic set would allow researchers to explore more specific time‐flow questions while maintaining comparability of most data. Electronic data collection tools (eg, handheld or tablet computers) could also facilitate the collection of more detailed and accurate data to increase study reliability.
| Primary | Secondary | Tertiary |
|---|---|---|
| Direct patient care | Daily rounds | Evaluation |
| Education | ||
| Admission history and physical | ||
| Consultation history and physical | ||
| Discharge | Evaluation | |
| Education | ||
| Procedures | ||
| Indirect care | Reviewing test results | |
| Documentation | Orders | |
| History and physical | ||
| Progress notes | ||
| Discharge paperwork | ||
| Communication | Paging | |
| Patient relatives | ||
| Other physicians | ||
| Nurse | ||
| Ancillary staff | ||
| Other | Education | |
| Transit/travel | ||
| Personal (eg, eating, restroom) | ||
| Miscellaneous |
Our systematic review is limited in its scope, as we focused only on the activities of physicians working in the hospital. Our exclusion criteria also eliminated several more focused time studies that evaluated only one small part of a physician's workflow, such as Amusan et al.'s36 evaluation of EMR and CPOE implementation during morning rounds. The available literature itself is also lacking in several important ways. Much of the literature is now limited by its age. The constant advance of medical technology, changes in work hour regulations, and new reimbursement structures have all affected physician workflow, and likely contributed to the variability of time study findings. Additionally, the available literature focuses almost exclusively on academic hospitals and teaching services. All but 1 of the studies collected data exclusively in academic hospitals, despite the fact that more than 90% of hospital care delivery in the U.S. occurs in a non‐academic hospital setting.20, 37 Just 1 study evaluated the activity of hospitalists directly caring for patients without assistance from residents.20 The significantly different workforce composition in community hospitals could mean that most findings are not relevant to the vast majority of U.S. hospitals. For example, the studies documenting that physicians in training (ie, residents) perform many activities that could be performed by a non‐physician are likely not applicable to the community hospital setting. Thus, additional research is needed to better understand how hospitalists can deliver care more efficiently, particularly in the community hospital setting and in the current technological and structural environment of healthcare.
This systematic review of the literature provides insight into published studies attempting to evaluate physician activities in the hospital through time‐motion and work‐sampling studies. Published research to date appears extremely variable in quality, limiting our ability to draw firm conclusions. However, it appears that hospital‐based physicians spend most of their time not interacting with patients, and non‐physician staff could readily complete a sizable portion of their tasks. Given the necessity for multitasking by hospitalists, better documentation of its frequency and impact is needed, as well as information about the types of tasks performed while multitasking, which has yet to be reported. Additionally, the effect of interruptions (including, but not limited to paging) needs further evaluation.
When properly performed, time‐study methodology represents a powerful approach to understanding the activities of hospitalists and how we might reengineer hospital care delivery to be more efficient. Efforts to standardize healthcare delivery and integrate health information technology could benefit dramatically from detailed information regarding physician activities and empirical testing of quality improvement initiatives. Future research using time‐motion or work‐sampling methodology should be careful to define and report categories of activity with enough detail that comparisons with other studies are possible.
Acknowledgements
The authors acknowledge the assistance of Linda O'Dwyer, MA MSLIS, research librarian at the Northwestern University Feinberg School of Medicine for her assistance with the search of the medical literature.
- .Motion and Time Study: Design and Measurement of Work.6th ed.New York:Wiley;1968.
- .Statistical methods in textile research. Uses of the binomial and poissant distributions.J Textile Inst Trans.1935;26:51–55.
- .Work‐sampling: a statistical approach to evaluation of the effect of computers on work patterns in the healthcare industry.Proc Annu Symp Comput Appl Med Care.1992:537–541.
- , , , , , .Time motion study in a pediatric emergency department before and after computer physician order entry.Ann Emerg Med.2009;53(4):462–468, e461.
- , , , .A “time‐and‐motion” study of endoscopic practice: strategies to enhance efficiency.Gastrointest Endosc.2008;68(6):1043–1050.
- , , , , , .Workflow in intensive care unit remote monitoring: A time‐and‐motion study.Crit Care Med.2007;35(9):2057–2063.
- , , , et al.Workflow analysis of medical staff in surgical wards based on time‐motion study data.Jpn Hosp.2008(27):75–80.
- , , , , , .General and visceral surgery practice in German hospitals: a real‐time work analysis on surgeons' work flow.Langenbecks Arch Surg.2010;395(1):81–87.
- , , , et al.Electronic health records in specialty care: a time‐motion study.J Am Med Inform Assoc.2007;14(5):609–615.
- , , , .National health spending in 2007: slower drug spending contributes to lowest rate of overall growth since 1998.Health Aff (Millwood).2009;28(1):246–261.
- , .The challenge of rising health care costs–a view from the Congressional Budget Office.N Engl J Med.2007;357(18):1793–1795.
- .Nonpayment for performance? Medicare's new reimbursement rule.N Engl J Med.2007;357(16):1573–1575.
- , .Hospitalists in teaching hospitals: opportunities but not without danger.J Gen Intern Med.2004;19:392–393.
- .The future of hospital medicine: evolution or revolution?Am J Med.2004;117:446–450.
- , .The evolution and future of hospital medicine.Mt Sinai J Med.2008;75(5):418–423.
- , , , , , .Outcomes of care by hospitalists, general internists, and family physicians.[see comment].N Engl J Med.2007;357(25):2589–2600.
- , .The hospitalist movement 5 years later.JAMA.2002;287:487–494.
- , .The impact of hospitalists on the cost and quality of inpatient care in the United States: a research synthesis.Med Care Res Rev.2005;62:379–406.
- .Hospitalists and the hospital medicine system of care are good for patient care.Arch Intern Med.2008;168(12):1254–1256; discussion 1259–1260.
- , , .How hospitalists spend their time: insights on efficiency and safety.J Hosp Med.2006;1(2):88–93.
- , , , , .The potential for using non‐physicians to compensate for the reduced availability of residents.Acad Med.1992;67(7):429–438.
- , , , , , .Observations of residents' work activities for 24 consecutive hours: Implications for workflow redesign.Acad Med.2006;81(8):766–775.
- , .The time needed for clinical documentation versus direct patient care. A work‐sampling analysis of physicians' activities.Methods Inf Med.2009;48(1):84–91.
- , , , et al.The on‐call experience of interns in internal medicine. Medical Education Task Force of Henry Ford Hospital.Arch Intern Med.1990;150(11):2294–2297.
- , , .What does the intern do?Med J Aust.1976;1(3):63–65.
- , , , .All in a day's work: an observational study to quantify how and with whom doctors on hospital wards spend their time.Med J Aust.2008;188(9):506–509.
- , , , .Resident educational time study: a tale of three specialties.Acad Emerg Med.1998;5(7):718–725.
- , .Are things different in the light of day? A time study of internal medicine house staff days.Am J Med.1993;94(6):654–658.
- , , .Time study of an internship on a university medical service.N Engl J Med.1961;264:439–443.
- , .Time study comparisons of 3 intern programs.J Med Educ.1971;46(2):142–149.
- , , , , .How do house officers spend their nights? A time study of internal medicine house staff on call.N Engl J Med.1989;320(25):1673–1677.
- .The Effect of a Non‐teaching Hospitalist Service in an Academic Hospital Setting: A Comparative Time‐motion and Clinical Outcomes Study. Unpublished Manuscript. Vol2006.
- .A strategy for health care reform—toward a value‐based system.N Engl J Med.2009;361(2):109–112.
- , , .Rehospitalizations among patients in the fee‐for‐service medicare program.N Engl J Med.2009;360(14):1418–1428.
- , .BOOSTing the hospital discharge.J Hosp Med.2009;4(4):209–210.
- , , , .A time‐motion study to evaluate the impact of EMR and CPOE implementation on physician efficiency.J Healthc Inf Manag.2008;22(4):31–37.
- , , , , .The ecology of medical care revisited.N Engl J Med.2001;344(26):2021–2025.
- .Motion and Time Study: Design and Measurement of Work.6th ed.New York:Wiley;1968.
- .Statistical methods in textile research. Uses of the binomial and poissant distributions.J Textile Inst Trans.1935;26:51–55.
- .Work‐sampling: a statistical approach to evaluation of the effect of computers on work patterns in the healthcare industry.Proc Annu Symp Comput Appl Med Care.1992:537–541.
- , , , , , .Time motion study in a pediatric emergency department before and after computer physician order entry.Ann Emerg Med.2009;53(4):462–468, e461.
- , , , .A “time‐and‐motion” study of endoscopic practice: strategies to enhance efficiency.Gastrointest Endosc.2008;68(6):1043–1050.
- , , , , , .Workflow in intensive care unit remote monitoring: A time‐and‐motion study.Crit Care Med.2007;35(9):2057–2063.
- , , , et al.Workflow analysis of medical staff in surgical wards based on time‐motion study data.Jpn Hosp.2008(27):75–80.
- , , , , , .General and visceral surgery practice in German hospitals: a real‐time work analysis on surgeons' work flow.Langenbecks Arch Surg.2010;395(1):81–87.
- , , , et al.Electronic health records in specialty care: a time‐motion study.J Am Med Inform Assoc.2007;14(5):609–615.
- , , , .National health spending in 2007: slower drug spending contributes to lowest rate of overall growth since 1998.Health Aff (Millwood).2009;28(1):246–261.
- , .The challenge of rising health care costs–a view from the Congressional Budget Office.N Engl J Med.2007;357(18):1793–1795.
- .Nonpayment for performance? Medicare's new reimbursement rule.N Engl J Med.2007;357(16):1573–1575.
- , .Hospitalists in teaching hospitals: opportunities but not without danger.J Gen Intern Med.2004;19:392–393.
- .The future of hospital medicine: evolution or revolution?Am J Med.2004;117:446–450.
- , .The evolution and future of hospital medicine.Mt Sinai J Med.2008;75(5):418–423.
- , , , , , .Outcomes of care by hospitalists, general internists, and family physicians.[see comment].N Engl J Med.2007;357(25):2589–2600.
- , .The hospitalist movement 5 years later.JAMA.2002;287:487–494.
- , .The impact of hospitalists on the cost and quality of inpatient care in the United States: a research synthesis.Med Care Res Rev.2005;62:379–406.
- .Hospitalists and the hospital medicine system of care are good for patient care.Arch Intern Med.2008;168(12):1254–1256; discussion 1259–1260.
- , , .How hospitalists spend their time: insights on efficiency and safety.J Hosp Med.2006;1(2):88–93.
- , , , , .The potential for using non‐physicians to compensate for the reduced availability of residents.Acad Med.1992;67(7):429–438.
- , , , , , .Observations of residents' work activities for 24 consecutive hours: Implications for workflow redesign.Acad Med.2006;81(8):766–775.
- , .The time needed for clinical documentation versus direct patient care. A work‐sampling analysis of physicians' activities.Methods Inf Med.2009;48(1):84–91.
- , , , et al.The on‐call experience of interns in internal medicine. Medical Education Task Force of Henry Ford Hospital.Arch Intern Med.1990;150(11):2294–2297.
- , , .What does the intern do?Med J Aust.1976;1(3):63–65.
- , , , .All in a day's work: an observational study to quantify how and with whom doctors on hospital wards spend their time.Med J Aust.2008;188(9):506–509.
- , , , .Resident educational time study: a tale of three specialties.Acad Emerg Med.1998;5(7):718–725.
- , .Are things different in the light of day? A time study of internal medicine house staff days.Am J Med.1993;94(6):654–658.
- , , .Time study of an internship on a university medical service.N Engl J Med.1961;264:439–443.
- , .Time study comparisons of 3 intern programs.J Med Educ.1971;46(2):142–149.
- , , , , .How do house officers spend their nights? A time study of internal medicine house staff on call.N Engl J Med.1989;320(25):1673–1677.
- .The Effect of a Non‐teaching Hospitalist Service in an Academic Hospital Setting: A Comparative Time‐motion and Clinical Outcomes Study. Unpublished Manuscript. Vol2006.
- .A strategy for health care reform—toward a value‐based system.N Engl J Med.2009;361(2):109–112.
- , , .Rehospitalizations among patients in the fee‐for‐service medicare program.N Engl J Med.2009;360(14):1418–1428.
- , .BOOSTing the hospital discharge.J Hosp Med.2009;4(4):209–210.
- , , , .A time‐motion study to evaluate the impact of EMR and CPOE implementation on physician efficiency.J Healthc Inf Manag.2008;22(4):31–37.
- , , , , .The ecology of medical care revisited.N Engl J Med.2001;344(26):2021–2025.
Hospitalist Time Usage and Cyclicality
Many academic medical centers (AMCs) employ hospitalists to provide care for patients on resident services as supervising attendings,1, 2 as well as on nonresident services.3 The number of hospitalists working on nonresident services at AMCs has grown exponentially, as the Accreditation Council for Graduate Medical Education (ACGME) implemented duty‐hour standards for residents.3 According to the latest Society of Hospital Medicine (SHM) estimates, the number of practicing hospitalists is projected to grow to 30,000 by 2010.4 As astonishing as this growth may sound, it is anticipated that more hospitalists will be needed to meet the demand for these physicians.5 Further, as financial realities require AMCs to be increasingly efficient without compromising patient care, and hospitalists provide a greater range of clinical services, it is important to better understand how hospitalists spend their time in the hospital. Understanding the daily work flow of hospitalists can identify how these physicians can be better supported. A previous report by O'Leary et al.6 highlighted how hospitalists spent their time during their usual day shifts at an AMC. It is important to validate their study to determine broadly applicable findings. We performed a time‐motion study where we followed the admitting hospitalists during the day and night shifts. We felt it was important to focus on hospitalists who are admitting patients, as this has potential patient safety and quality implications related to multitasking, triaging, and helping patients navigate through a complex admission process involving multiple clinical services. Our goal was to better understand how the flow of patients impacted these physicians, and determine how our hospitalists spent their time providing direct and indirect patient care‐related activities. In addition, we looked for predictable variations in activities throughout the day that might be associated with the timely care of patients.
Materials and Methods
Setting
The University of Michigan Health System (UMHS) is a tertiary care AMC, with more than 800 beds, and over 34,000 annual adult discharges. Internal Medicine services comprise a large proportion of those discharged, accounting for over 17,000 discharges per year; and is projected to grow at an annual rate of 4%. As service caps and work‐hour restrictions have limited the total number of patients that medical residents are able to care for, our hospitalist group has increased the number of physicians on the nonresident hospitalist service. At the time of the study, there were 23 hospitalists, equivalent to 18.25 full‐time equivalents (FTEs), staffing the service. The hospitalists provide in‐house patient care 24 hours a day and 7 days a week. Hospitalists also provide general medicine consult services, surgical comanagement and perioperative care, procedures, inpatient cardiopulmonary arrest response, rapid response team supervision, and observation care; and are also the primary inpatient physicians for many of the hospitalized interventional radiology and dermatology patients. These direct patient care activities account for 4500 annual discharges from the nonresident service.
Data Collection
Four university undergraduate business administration program students shadowed 11 hospitalists over a 3‐week period in 4‐hour to 12‐hour time blocks. The students followed the hospitalist on the shift that was taking admission calls, during day and night. A data collection tool was designed to track physicians' actions in 1‐minute increments, using categories similar to those used in a previously published time‐motion study of hospitalists' activities (Table 1).6 Physicians' activities each minute were assigned to a single category that most represented their action during that time period. At our AMC, 6 hospitalists work during the day shifts, and 2 on the night shifts. Our hospitalists may have patients in any of the 14 general care units in the hospital, as our hospitalists' services are not geographically based. The day hospitalists' shifts are scheduled from 7 AM to 7 PM. Two of the 6 hospitalists rotate through a 3‐day cycle as the admitting physician. Their duties include triaging and admitting patients until 2 PM, providing the day‐to‐day care for their patients until 7 PM, and occasionally cross‐covering for the other day‐shift hospitalists that have left for the day. The 4 other day‐shift hospitalists, not on their rotation as the admitting physician, may sign out and leave as early as 4 PM if their work for the day is done. At 2 PM, a separate swing‐shift hospitalist takes over the role of triaging and admitting until 7 PM. During the day shift, consults and perioperative management of patients are provided by a separate hospitalist on the consult service. At 7 PM, 2 nocturnists arrive for their 7 PM to 7 AM shift. The nocturnists, in addition to cross‐covering service patients, admit a maximum of 6 patients each, or until midnightwhichever comes first.
| Category | Code | Description |
|---|---|---|
| Direct patient care | DPIH | Initial history |
| DPDI | Discharge instructions | |
| DPFM | Family meetings | |
| DPRV | Revisit | |
| DPCC | Cross‐cover | |
| Indirect patient care | ||
| Documentation | IDGD | General documentation |
| IDDN | Daily notes | |
| IDDD | Discharge navigator | |
| Records/Results | IPMR | Review medical records |
| Communication | ICHH | Patient handoffs |
| ICFF | Face‐to‐face | |
| ICIP | Incoming page | |
| ICOP | Outgoing page | |
| ICIC | Incoming call | |
| ICOC | Outgoing call | |
| ICEE | E‐mail communications | |
| ICDP | Discharge planner | |
| Orders | IOWO | Writing orders |
| Professional development | PDRR | Reading articles, textbooks, references |
| Education | EEWR | Teaching during work rounds |
| Travel | TTTT | Travel |
| Personal | PPPP | Personal |
| Down time | DDDD | Downtime |
The students observed 11 different hospitalists, and followed these physicians during 9 weekday shifts, 5 weekday swing shifts, 10 weekday night shifts, and 4 weekend night shifts. The variance in the number of each type of shifts monitored was likely due to scheduling limitations of the students. In total, they collected data on 8,915 minutes of hospitalists' activities. The students monitored the hospitalists representing time periods from 7 AM to 2 AM. Analysis from 2 AM to 7 AM was excluded, because after 2 AM the hospitalists did not routinely evaluate new patients with the exception of emergent requests. New admissions after midnight are handled by a night float service staffed by residents.
Results
Overall, time spent on patient care activities comprised the bulk of hospitalists' shifts (82%) (Figure 1). Patient care activities were further categorized as direct patient caredefined as face‐to‐face patient or family time; and indirect patient caredefined as activities related to patient care, but without patient or family contact. Direct and indirect patient care accounted for 15% and 67% of the hospitalists' time, respectively. The other 18% of the hospitalists' time spent in the hospital were broadly categorized into: professional development, education, personal, downtime, and travel. Professional development included activities such as looking up information (eg, literature search); education included times that hospitalists spent with residents or medical students; personal time included only restroom and food breaks; and travel included time spent moving from 1 area to the next during their shift.
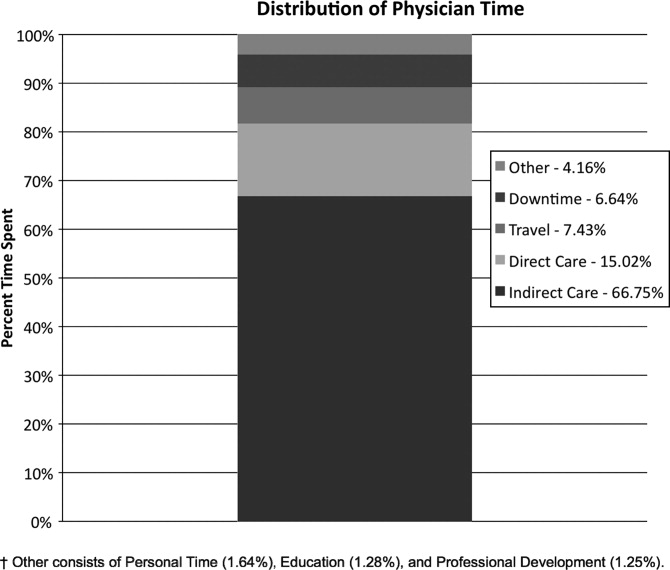
The majority of the hospitalists' direct patient care time was spent on evaluating new patients (79%). Significantly smaller amounts of time were spent on other direct care activities: cross‐covering other patients (8%), follow‐up visits (7%), family meetings (4%), and discharge instructions (2%) (Figure 2).
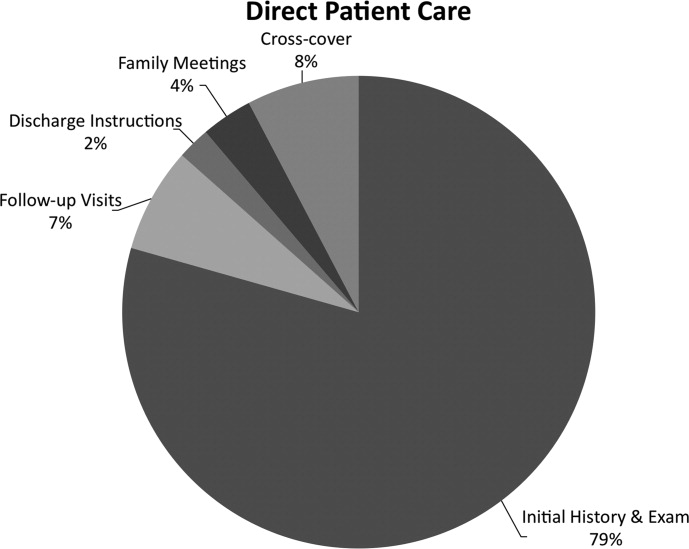
Indirect patient care activities included, 41% of time used to communicate with other healthcare providers, 26% on medical documentation, 20% reviewing medical records and results, and 13% of time writing orders (Figure 3). Communication accounted for a large proportion of a hospitalists' work, and included telephone conversations with Emergency Department (ED) or other admitting providers, handoffs, paging, face‐to‐face conversations with consultants and other support staff, and e‐mail.
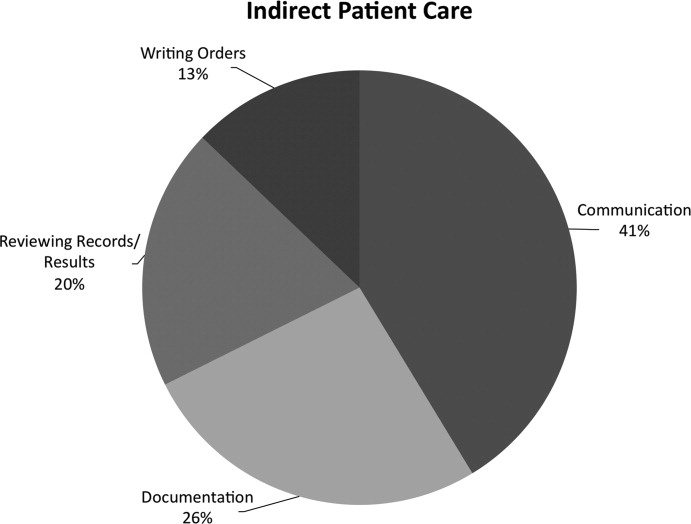
Figure 4 shows the hourly distribution of time spent on direct and indirect patient care by a hospitalist throughout the day. The day‐time hospitalists pick up their signout from the nocturnists at 7 AM to begin their shift. The swing hospitalists arrive at 2 PM during the weekdays, and their primary duty is to triage and admit patients until 7 PM. The nocturnists start their shift at 7 PM, at which time the daytime and swing‐shift hospitalists all sign out for the night.
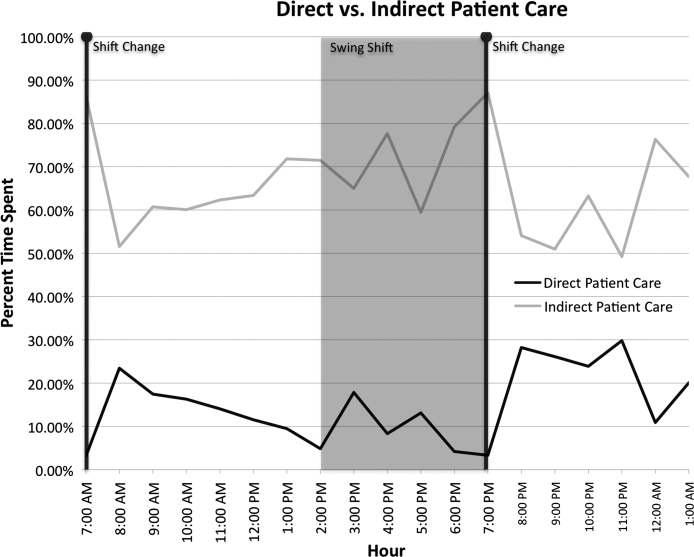
Discussion
Hospitalists on the nonresident service at our AMC utilize about 15% of their time on face‐to‐face patient care activities, 67% on indirect patient care activities, and 7% of time on moving from 1 part of the hospital to another. Hospitalists are valuable members of the physician work force who address the increasing patient care demands in the face of increasing limitations on residency work‐hours, a growing aging population, and existing inefficiencies in AMCs. The only other work‐flow study of hospitalists of which we are aware provided a single institution's perspective on time utilization by hospitalists. Our study in a different AMC setting revealed strong consistency with the O'Leary et al.6 study in the fraction of time hospitalists spent on direct patient care (15% and 18%, respectively), indirect patient care (67% and 69%); and within indirect patient care the time spent on documentation (26% and 37% of total time) and communications (41% and 35%). While travel in the O'Leary et al.6 study took up only 3% of hospitalists' time, the conclusions in that paper clearly suggest that the authors consider it an area of concern. Our study found that travel accounted for over 7% of hospitalists' time, confirming that intuition. The significant travel time may in part reflect the effects of a non‐geographically‐located hospitalist service. From these 2 studies we can be more confident that in large, tertiary care AMCs the time hospitalists spend on indirect patient care dominates that for direct patient care (by a factor of 4 in these studies), that within indirect patient care documentation and communication are dominant activities, and that travel can take a significant amount of time when patients are dispersed throughout the facility.
Both studies demonstrated that communication accounted for a significant proportion of a hospitalist's time. In our study communication accounted for 28% of their total time in the hospital, and 41% of the indirect patient care portion (Figure 3). A closer look within our communication category revealed that phone calls and handoffs accounted for two‐thirds of all communication time observed. As the hospitalists who carry the admitting pager, they receive the pages to take admission calls, but also take calls from consultants who have recommendations, as well as from nursing and other hospital staff. Depending on the nature of the conversation, the phone calls can last several minutes. While ensuring the communication between health care providers is complete and thorough, there may be opportunities to develop novel approaches to the way hospitalists communicate with other care providers. For example, at the UMHS, alternative communication methods with nursing staff have been proposed such as utilizing a website or a handheld device to help hospitalists prioritize their communications back to the nursing staff7; while standardizing the intake information from the ED or other admitting providers may help reduce the total time spent on phone calls. We will need to further explore the potential benefits of these ideas in future work.
Our data also reveal an interesting cyclicality of daily activities for the hospitalists, as shown in Figure 4. We identified batching behaviors throughout the day, which cause delays in seeing patients and can be deleterious to smooth workflows in support services. Spikes in indirect patient care, followed closely by spikes in direct patient care, occur regularly at shift changes (7 AM, 2 PM, and 7 PM). Also, in the night shift, indirect patient care drops to its lowest levels (in % of time spent) throughout the day, and direct patient care reaches its highest levels. The day‐shift indirect care profile is counter‐cyclical with direct care, as the hospitalist shifts between direct care and indirect care depending on the time of the day. We discuss these phenomena in turn.
It is known that variability in any operation causes congestion and delay, as an unavoidable consequence of the physics of material and information flows.8 Indeed, an entire subindustry based on Lean manufacturing principles has evolved from the Toyota Production System based on the elimination of unnecessary variability in operations.9 Lean processes have been ongoing in manufacturing facilities for decades, and these efforts are just recently being embraced by the service sector in general, and health care specifically.10, 11 Batching is an extreme form of variability, where there is a lull in the amount of work being done and then a burst of work is done over a short period of time. This means that jobs pile up in the queue waiting for the next spike of activity. Our data indicate batching seems to be a common phenomenon for our hospitalists. The majority of the patients admitted to our hospitalist service are unscheduled admissions that arrive primarily through the ED. One potential result of the unscheduled admissions is that patients could be referred to our hospitalist service at a pace that is not well predictable on an hour‐to‐hour basis. This could lead to an unintended result of multiple patients admitted over a short period of time. This means that many patients wait for intake, delaying the onset of their care by the inpatient physician. Also, since an initial exam often results in orders for laboratory tests and studies, batching on the floor will translate into batching of orders going to nursing, pathology, radiology, and other hospital support services. This imposes the cost of variability on these other services in the hospital. From a systems perspective, efficiency will improve if these activities can be smoothed throughout the day. This may suggest opportunities to work with the ED, to help smooth the inflow of patients into the hospital system.
Within the hospital, all of the day‐shift hospitalists can be reached about the needs of their respective patients, however, the physician carrying the admission pager also fields calls for admissions, and acts as the default contact person for the hospitalist group. As this hospitalist receives information on new admissions, he/she is aware of patients ready for intake but cannot evaluate them at the rate they are being referred, so the queue builds. This continues into the swing shift, which also fields referrals faster than they can attend to them. The volatility in indirect care during the swing shift, 2 PM to 7 PM, reflects a significant amount of triaging and fielding general calls about hospitalist patients. These activities further reduce the swing shift's ability to clear the intake queue. The night shift finally gets to these patients and, eventually, clears the queue. There may be an opportunity to consider the use of multiple input pagers or other process changes that can smooth this flow and rationalize the recurring tasks of finding patients and the responsible physician.
Another concept in Lean thinking is that variability is costly when it represents a mismatch between demand for a service and the capacity to serve. With regards to admitted patients, when demand outpaces capacity, patients will wait. When capacity outpaces demand, there is excess capacity in the system. The ideal is to match demand and capacity at all times, so nobody waits and the system carries no costly excess capacity. As the intake providers for admitted patients, we can attack this problem from the capacity side. Here, 2 generic Lean tactics are to: (1) reallocate resources to a bottleneck that is holding up the entire system, and (2) relieve workers of time‐consuming but non‐value‐adding work so they have more capacity to devote to serving demand. In our study, carrying multiple input pagers is an example of tactic (1), and efficient communication technologies and practices that reduce indirect time is an example of (2). Systemwide improvements would require further investigation by working with the variability on the input side (eg, ED admissions).
Our study also found that a significant percent of the time observed was spent traveling (7.4%) from room to room between different floors in the hospital. Travel time, which is non‐value‐adding, is one of the major forms of waste Lean thinking.12 Our hospitalists can provide care to patients at any of the general medical‐surgical beds we have available at our health system. These beds are distributed across 14 units on 5 different floors, as well as in the ED if a bed is not available for an admitted patient. In hospitals routinely operating at high occupancy, such as our AMC, patients often get distributed throughout the facility for lack of beds on the appropriate service's ward. One cost for this is a potential mismatch between a patient's needs and floor nurses' training. Our study reveals another cost, and that is its contribution to the significant amount of time hospitalists spent on travel, which is largely driven by the need to see dispersed patients. Reducing this cost requires a systemic, rather than service‐specific, solution. Our AMC is adding observation‐status beds to relieve some of the pressure on licensed beds, and considering bed management (including parts of the admissions and discharge processes) changes designed to promote better collocation of patients with services. Further study on these and other collocation tactics is warranted.
The spike in indirect activities at 4 PM represents, in part, an early signout by 1 or more of the hospitalists who are not scheduled to hold the admission pager, and have completed their work for the day. This handoff will be replicated at 7 PM when the nocturnists arrive for their night shift. In addition to a significant indirect load on physicians, multiple handoffs have been associated with decreased quality of care.13 Again, it is worthwhile considering the feasibility of alternative shift schedules that can minimize handoffs.
Finally, our findings revealed that a low percentage of time was dedicated to providing discharge instructions (2.24% of direct patient care time, and 0.34% of total time). Because the task of discharging patients falls primarily on the day‐shift hospitalists, when combined with swing‐shift and night‐shift hospitalists' data, the low percentage measured on discharge instructions may have been diluted. Nonetheless, this may point to the need for further investigation on how hospitalists provide direct patient encounter time during this critical phase of transition out of the hospital.
Our study is not without limitations. The student observers shadowed a representative group of hospitalists, but they were not able to follow everyone in the group. More specifically, their observations were made on the hospitalist who was carrying the primary hospitalist service admitting pager. Although it was the intent of our study to focus on the hospitalists we felt would be the busiest, our results may not be generalizable to all hospitalists. Although our research supports the previous findings by O'Leary et al.,6 a second limitation to our study is that our analysis was done at a single hospitalist group in an AMC, and hence the results may not be generalizable to other hospitalist groups. Another limitation may be that we did not do an evaluation of the hours between 2 AM to 7 AM. This period of time is used to catch up on medical documentation and to be available for medical emergencies. As more hospitalist programs are employing the use of nocturnists, it may be informative to have this time period tracked for activities.
Conclusions
Our study supports the broad allocation of hospitalist time found in an earlier study at a different AMC,6 suggesting that these might be generally representative in other AMCs. We found that travel constitutes a significant claim in hospitalists' time, due in part to the inability to collocate hospitalist service patients. Remedies are not likely to be service‐specific, but will require systemwide analyses of admission and discharge processes. Communication takes a significant amount of hospitalist time, with pages and phone calls related to handoffs accounting for most of the total communication time. As hospitalists working at non‐AMC settings may experience different work flow issues, we would like to see time‐motion studies of hospitalists in other types of hospitals. Future studies should also seek to better understand the how hospitals at high occupancy may reduce batching and streamline both the discharge and admission process, determine the factors that account for the significant communication time and how these processes could be streamlined, and evaluate the potential benefits of geographical localization of hospitalists' patients.
Acknowledgements
The authors thank Tracey Jackson, Michael Paulsen, Deepak Srinivasin, and Ryan Werblow, who were students in the undergraduate business school program, for their invaluable contribution in shadowing hospitalists to collect the time study data.
- , , , .Where should hospitalists sit within the academic medical center?J Gen Intern Med.2008;23:1269–1272.
- , .Hospitalists in teaching hospitals: opportunities but not without danger.J Gen Intern Med.2004;19:392–393.
- , , , , .Non‐housestaff medicine services in academic centers: models and challenges.J Hosp Med.2008;3:247–255.
- Society of Hospital Medicine. Society of Hospital Medicine Releases Results of the 2007–2008 Survey on the State of the Hospital Medicine Movement.2008. Available at: http://www.hospitalmedicine.org/AM/Template.cfm? Section=Press_Releases3:398–402.
- , , .How hospitalists spend their time: insights on efficiency and safety.J Hosp Med.2006;1:88–93.
- , .MCOMM: Redefining Medical Communication in the 21st Century, University of Michigan Health System. In: Society of Hospital Medicine Annual Meeting, Best of Innovations Presentation; 2009; Chicago, IL;2009.
- , .Factory Physics: Foundations of Manufacturing Management.Boston:Irwin, McGraw‐Hill;1996.
- .The Toyota Way.1st ed.Madison, WI:McGraw‐Hill;2004.
- Going Lean in Health Care.White Paper.Boston, MA:Institute for Healthcare Improvement;2005 January and February, 2005. Available at: http://www.ihconline.org/toolkits/LeanInHealthcare/GoingLeaninHealth CareWhitePaper.pdf. Accessed September 2009.
- , , , .Lean health care: what can hospitals learn from a world‐class automaker?J Hosp Med.2006;1:191–199.
- , , , , .Managing Business Process Flows.Upper Saddle River, NJ:Prentice Hall;2006.
- , .The patient handoff: medicine's Formula One moment.Chest.2008;134:9–12.
Many academic medical centers (AMCs) employ hospitalists to provide care for patients on resident services as supervising attendings,1, 2 as well as on nonresident services.3 The number of hospitalists working on nonresident services at AMCs has grown exponentially, as the Accreditation Council for Graduate Medical Education (ACGME) implemented duty‐hour standards for residents.3 According to the latest Society of Hospital Medicine (SHM) estimates, the number of practicing hospitalists is projected to grow to 30,000 by 2010.4 As astonishing as this growth may sound, it is anticipated that more hospitalists will be needed to meet the demand for these physicians.5 Further, as financial realities require AMCs to be increasingly efficient without compromising patient care, and hospitalists provide a greater range of clinical services, it is important to better understand how hospitalists spend their time in the hospital. Understanding the daily work flow of hospitalists can identify how these physicians can be better supported. A previous report by O'Leary et al.6 highlighted how hospitalists spent their time during their usual day shifts at an AMC. It is important to validate their study to determine broadly applicable findings. We performed a time‐motion study where we followed the admitting hospitalists during the day and night shifts. We felt it was important to focus on hospitalists who are admitting patients, as this has potential patient safety and quality implications related to multitasking, triaging, and helping patients navigate through a complex admission process involving multiple clinical services. Our goal was to better understand how the flow of patients impacted these physicians, and determine how our hospitalists spent their time providing direct and indirect patient care‐related activities. In addition, we looked for predictable variations in activities throughout the day that might be associated with the timely care of patients.
Materials and Methods
Setting
The University of Michigan Health System (UMHS) is a tertiary care AMC, with more than 800 beds, and over 34,000 annual adult discharges. Internal Medicine services comprise a large proportion of those discharged, accounting for over 17,000 discharges per year; and is projected to grow at an annual rate of 4%. As service caps and work‐hour restrictions have limited the total number of patients that medical residents are able to care for, our hospitalist group has increased the number of physicians on the nonresident hospitalist service. At the time of the study, there were 23 hospitalists, equivalent to 18.25 full‐time equivalents (FTEs), staffing the service. The hospitalists provide in‐house patient care 24 hours a day and 7 days a week. Hospitalists also provide general medicine consult services, surgical comanagement and perioperative care, procedures, inpatient cardiopulmonary arrest response, rapid response team supervision, and observation care; and are also the primary inpatient physicians for many of the hospitalized interventional radiology and dermatology patients. These direct patient care activities account for 4500 annual discharges from the nonresident service.
Data Collection
Four university undergraduate business administration program students shadowed 11 hospitalists over a 3‐week period in 4‐hour to 12‐hour time blocks. The students followed the hospitalist on the shift that was taking admission calls, during day and night. A data collection tool was designed to track physicians' actions in 1‐minute increments, using categories similar to those used in a previously published time‐motion study of hospitalists' activities (Table 1).6 Physicians' activities each minute were assigned to a single category that most represented their action during that time period. At our AMC, 6 hospitalists work during the day shifts, and 2 on the night shifts. Our hospitalists may have patients in any of the 14 general care units in the hospital, as our hospitalists' services are not geographically based. The day hospitalists' shifts are scheduled from 7 AM to 7 PM. Two of the 6 hospitalists rotate through a 3‐day cycle as the admitting physician. Their duties include triaging and admitting patients until 2 PM, providing the day‐to‐day care for their patients until 7 PM, and occasionally cross‐covering for the other day‐shift hospitalists that have left for the day. The 4 other day‐shift hospitalists, not on their rotation as the admitting physician, may sign out and leave as early as 4 PM if their work for the day is done. At 2 PM, a separate swing‐shift hospitalist takes over the role of triaging and admitting until 7 PM. During the day shift, consults and perioperative management of patients are provided by a separate hospitalist on the consult service. At 7 PM, 2 nocturnists arrive for their 7 PM to 7 AM shift. The nocturnists, in addition to cross‐covering service patients, admit a maximum of 6 patients each, or until midnightwhichever comes first.
| Category | Code | Description |
|---|---|---|
| Direct patient care | DPIH | Initial history |
| DPDI | Discharge instructions | |
| DPFM | Family meetings | |
| DPRV | Revisit | |
| DPCC | Cross‐cover | |
| Indirect patient care | ||
| Documentation | IDGD | General documentation |
| IDDN | Daily notes | |
| IDDD | Discharge navigator | |
| Records/Results | IPMR | Review medical records |
| Communication | ICHH | Patient handoffs |
| ICFF | Face‐to‐face | |
| ICIP | Incoming page | |
| ICOP | Outgoing page | |
| ICIC | Incoming call | |
| ICOC | Outgoing call | |
| ICEE | E‐mail communications | |
| ICDP | Discharge planner | |
| Orders | IOWO | Writing orders |
| Professional development | PDRR | Reading articles, textbooks, references |
| Education | EEWR | Teaching during work rounds |
| Travel | TTTT | Travel |
| Personal | PPPP | Personal |
| Down time | DDDD | Downtime |
The students observed 11 different hospitalists, and followed these physicians during 9 weekday shifts, 5 weekday swing shifts, 10 weekday night shifts, and 4 weekend night shifts. The variance in the number of each type of shifts monitored was likely due to scheduling limitations of the students. In total, they collected data on 8,915 minutes of hospitalists' activities. The students monitored the hospitalists representing time periods from 7 AM to 2 AM. Analysis from 2 AM to 7 AM was excluded, because after 2 AM the hospitalists did not routinely evaluate new patients with the exception of emergent requests. New admissions after midnight are handled by a night float service staffed by residents.
Results
Overall, time spent on patient care activities comprised the bulk of hospitalists' shifts (82%) (Figure 1). Patient care activities were further categorized as direct patient caredefined as face‐to‐face patient or family time; and indirect patient caredefined as activities related to patient care, but without patient or family contact. Direct and indirect patient care accounted for 15% and 67% of the hospitalists' time, respectively. The other 18% of the hospitalists' time spent in the hospital were broadly categorized into: professional development, education, personal, downtime, and travel. Professional development included activities such as looking up information (eg, literature search); education included times that hospitalists spent with residents or medical students; personal time included only restroom and food breaks; and travel included time spent moving from 1 area to the next during their shift.

The majority of the hospitalists' direct patient care time was spent on evaluating new patients (79%). Significantly smaller amounts of time were spent on other direct care activities: cross‐covering other patients (8%), follow‐up visits (7%), family meetings (4%), and discharge instructions (2%) (Figure 2).

Indirect patient care activities included, 41% of time used to communicate with other healthcare providers, 26% on medical documentation, 20% reviewing medical records and results, and 13% of time writing orders (Figure 3). Communication accounted for a large proportion of a hospitalists' work, and included telephone conversations with Emergency Department (ED) or other admitting providers, handoffs, paging, face‐to‐face conversations with consultants and other support staff, and e‐mail.

Figure 4 shows the hourly distribution of time spent on direct and indirect patient care by a hospitalist throughout the day. The day‐time hospitalists pick up their signout from the nocturnists at 7 AM to begin their shift. The swing hospitalists arrive at 2 PM during the weekdays, and their primary duty is to triage and admit patients until 7 PM. The nocturnists start their shift at 7 PM, at which time the daytime and swing‐shift hospitalists all sign out for the night.

Discussion
Hospitalists on the nonresident service at our AMC utilize about 15% of their time on face‐to‐face patient care activities, 67% on indirect patient care activities, and 7% of time on moving from 1 part of the hospital to another. Hospitalists are valuable members of the physician work force who address the increasing patient care demands in the face of increasing limitations on residency work‐hours, a growing aging population, and existing inefficiencies in AMCs. The only other work‐flow study of hospitalists of which we are aware provided a single institution's perspective on time utilization by hospitalists. Our study in a different AMC setting revealed strong consistency with the O'Leary et al.6 study in the fraction of time hospitalists spent on direct patient care (15% and 18%, respectively), indirect patient care (67% and 69%); and within indirect patient care the time spent on documentation (26% and 37% of total time) and communications (41% and 35%). While travel in the O'Leary et al.6 study took up only 3% of hospitalists' time, the conclusions in that paper clearly suggest that the authors consider it an area of concern. Our study found that travel accounted for over 7% of hospitalists' time, confirming that intuition. The significant travel time may in part reflect the effects of a non‐geographically‐located hospitalist service. From these 2 studies we can be more confident that in large, tertiary care AMCs the time hospitalists spend on indirect patient care dominates that for direct patient care (by a factor of 4 in these studies), that within indirect patient care documentation and communication are dominant activities, and that travel can take a significant amount of time when patients are dispersed throughout the facility.
Both studies demonstrated that communication accounted for a significant proportion of a hospitalist's time. In our study communication accounted for 28% of their total time in the hospital, and 41% of the indirect patient care portion (Figure 3). A closer look within our communication category revealed that phone calls and handoffs accounted for two‐thirds of all communication time observed. As the hospitalists who carry the admitting pager, they receive the pages to take admission calls, but also take calls from consultants who have recommendations, as well as from nursing and other hospital staff. Depending on the nature of the conversation, the phone calls can last several minutes. While ensuring the communication between health care providers is complete and thorough, there may be opportunities to develop novel approaches to the way hospitalists communicate with other care providers. For example, at the UMHS, alternative communication methods with nursing staff have been proposed such as utilizing a website or a handheld device to help hospitalists prioritize their communications back to the nursing staff7; while standardizing the intake information from the ED or other admitting providers may help reduce the total time spent on phone calls. We will need to further explore the potential benefits of these ideas in future work.
Our data also reveal an interesting cyclicality of daily activities for the hospitalists, as shown in Figure 4. We identified batching behaviors throughout the day, which cause delays in seeing patients and can be deleterious to smooth workflows in support services. Spikes in indirect patient care, followed closely by spikes in direct patient care, occur regularly at shift changes (7 AM, 2 PM, and 7 PM). Also, in the night shift, indirect patient care drops to its lowest levels (in % of time spent) throughout the day, and direct patient care reaches its highest levels. The day‐shift indirect care profile is counter‐cyclical with direct care, as the hospitalist shifts between direct care and indirect care depending on the time of the day. We discuss these phenomena in turn.
It is known that variability in any operation causes congestion and delay, as an unavoidable consequence of the physics of material and information flows.8 Indeed, an entire subindustry based on Lean manufacturing principles has evolved from the Toyota Production System based on the elimination of unnecessary variability in operations.9 Lean processes have been ongoing in manufacturing facilities for decades, and these efforts are just recently being embraced by the service sector in general, and health care specifically.10, 11 Batching is an extreme form of variability, where there is a lull in the amount of work being done and then a burst of work is done over a short period of time. This means that jobs pile up in the queue waiting for the next spike of activity. Our data indicate batching seems to be a common phenomenon for our hospitalists. The majority of the patients admitted to our hospitalist service are unscheduled admissions that arrive primarily through the ED. One potential result of the unscheduled admissions is that patients could be referred to our hospitalist service at a pace that is not well predictable on an hour‐to‐hour basis. This could lead to an unintended result of multiple patients admitted over a short period of time. This means that many patients wait for intake, delaying the onset of their care by the inpatient physician. Also, since an initial exam often results in orders for laboratory tests and studies, batching on the floor will translate into batching of orders going to nursing, pathology, radiology, and other hospital support services. This imposes the cost of variability on these other services in the hospital. From a systems perspective, efficiency will improve if these activities can be smoothed throughout the day. This may suggest opportunities to work with the ED, to help smooth the inflow of patients into the hospital system.
Within the hospital, all of the day‐shift hospitalists can be reached about the needs of their respective patients, however, the physician carrying the admission pager also fields calls for admissions, and acts as the default contact person for the hospitalist group. As this hospitalist receives information on new admissions, he/she is aware of patients ready for intake but cannot evaluate them at the rate they are being referred, so the queue builds. This continues into the swing shift, which also fields referrals faster than they can attend to them. The volatility in indirect care during the swing shift, 2 PM to 7 PM, reflects a significant amount of triaging and fielding general calls about hospitalist patients. These activities further reduce the swing shift's ability to clear the intake queue. The night shift finally gets to these patients and, eventually, clears the queue. There may be an opportunity to consider the use of multiple input pagers or other process changes that can smooth this flow and rationalize the recurring tasks of finding patients and the responsible physician.
Another concept in Lean thinking is that variability is costly when it represents a mismatch between demand for a service and the capacity to serve. With regards to admitted patients, when demand outpaces capacity, patients will wait. When capacity outpaces demand, there is excess capacity in the system. The ideal is to match demand and capacity at all times, so nobody waits and the system carries no costly excess capacity. As the intake providers for admitted patients, we can attack this problem from the capacity side. Here, 2 generic Lean tactics are to: (1) reallocate resources to a bottleneck that is holding up the entire system, and (2) relieve workers of time‐consuming but non‐value‐adding work so they have more capacity to devote to serving demand. In our study, carrying multiple input pagers is an example of tactic (1), and efficient communication technologies and practices that reduce indirect time is an example of (2). Systemwide improvements would require further investigation by working with the variability on the input side (eg, ED admissions).
Our study also found that a significant percent of the time observed was spent traveling (7.4%) from room to room between different floors in the hospital. Travel time, which is non‐value‐adding, is one of the major forms of waste Lean thinking.12 Our hospitalists can provide care to patients at any of the general medical‐surgical beds we have available at our health system. These beds are distributed across 14 units on 5 different floors, as well as in the ED if a bed is not available for an admitted patient. In hospitals routinely operating at high occupancy, such as our AMC, patients often get distributed throughout the facility for lack of beds on the appropriate service's ward. One cost for this is a potential mismatch between a patient's needs and floor nurses' training. Our study reveals another cost, and that is its contribution to the significant amount of time hospitalists spent on travel, which is largely driven by the need to see dispersed patients. Reducing this cost requires a systemic, rather than service‐specific, solution. Our AMC is adding observation‐status beds to relieve some of the pressure on licensed beds, and considering bed management (including parts of the admissions and discharge processes) changes designed to promote better collocation of patients with services. Further study on these and other collocation tactics is warranted.
The spike in indirect activities at 4 PM represents, in part, an early signout by 1 or more of the hospitalists who are not scheduled to hold the admission pager, and have completed their work for the day. This handoff will be replicated at 7 PM when the nocturnists arrive for their night shift. In addition to a significant indirect load on physicians, multiple handoffs have been associated with decreased quality of care.13 Again, it is worthwhile considering the feasibility of alternative shift schedules that can minimize handoffs.
Finally, our findings revealed that a low percentage of time was dedicated to providing discharge instructions (2.24% of direct patient care time, and 0.34% of total time). Because the task of discharging patients falls primarily on the day‐shift hospitalists, when combined with swing‐shift and night‐shift hospitalists' data, the low percentage measured on discharge instructions may have been diluted. Nonetheless, this may point to the need for further investigation on how hospitalists provide direct patient encounter time during this critical phase of transition out of the hospital.
Our study is not without limitations. The student observers shadowed a representative group of hospitalists, but they were not able to follow everyone in the group. More specifically, their observations were made on the hospitalist who was carrying the primary hospitalist service admitting pager. Although it was the intent of our study to focus on the hospitalists we felt would be the busiest, our results may not be generalizable to all hospitalists. Although our research supports the previous findings by O'Leary et al.,6 a second limitation to our study is that our analysis was done at a single hospitalist group in an AMC, and hence the results may not be generalizable to other hospitalist groups. Another limitation may be that we did not do an evaluation of the hours between 2 AM to 7 AM. This period of time is used to catch up on medical documentation and to be available for medical emergencies. As more hospitalist programs are employing the use of nocturnists, it may be informative to have this time period tracked for activities.
Conclusions
Our study supports the broad allocation of hospitalist time found in an earlier study at a different AMC,6 suggesting that these might be generally representative in other AMCs. We found that travel constitutes a significant claim in hospitalists' time, due in part to the inability to collocate hospitalist service patients. Remedies are not likely to be service‐specific, but will require systemwide analyses of admission and discharge processes. Communication takes a significant amount of hospitalist time, with pages and phone calls related to handoffs accounting for most of the total communication time. As hospitalists working at non‐AMC settings may experience different work flow issues, we would like to see time‐motion studies of hospitalists in other types of hospitals. Future studies should also seek to better understand the how hospitals at high occupancy may reduce batching and streamline both the discharge and admission process, determine the factors that account for the significant communication time and how these processes could be streamlined, and evaluate the potential benefits of geographical localization of hospitalists' patients.
Acknowledgements
The authors thank Tracey Jackson, Michael Paulsen, Deepak Srinivasin, and Ryan Werblow, who were students in the undergraduate business school program, for their invaluable contribution in shadowing hospitalists to collect the time study data.
Many academic medical centers (AMCs) employ hospitalists to provide care for patients on resident services as supervising attendings,1, 2 as well as on nonresident services.3 The number of hospitalists working on nonresident services at AMCs has grown exponentially, as the Accreditation Council for Graduate Medical Education (ACGME) implemented duty‐hour standards for residents.3 According to the latest Society of Hospital Medicine (SHM) estimates, the number of practicing hospitalists is projected to grow to 30,000 by 2010.4 As astonishing as this growth may sound, it is anticipated that more hospitalists will be needed to meet the demand for these physicians.5 Further, as financial realities require AMCs to be increasingly efficient without compromising patient care, and hospitalists provide a greater range of clinical services, it is important to better understand how hospitalists spend their time in the hospital. Understanding the daily work flow of hospitalists can identify how these physicians can be better supported. A previous report by O'Leary et al.6 highlighted how hospitalists spent their time during their usual day shifts at an AMC. It is important to validate their study to determine broadly applicable findings. We performed a time‐motion study where we followed the admitting hospitalists during the day and night shifts. We felt it was important to focus on hospitalists who are admitting patients, as this has potential patient safety and quality implications related to multitasking, triaging, and helping patients navigate through a complex admission process involving multiple clinical services. Our goal was to better understand how the flow of patients impacted these physicians, and determine how our hospitalists spent their time providing direct and indirect patient care‐related activities. In addition, we looked for predictable variations in activities throughout the day that might be associated with the timely care of patients.
Materials and Methods
Setting
The University of Michigan Health System (UMHS) is a tertiary care AMC, with more than 800 beds, and over 34,000 annual adult discharges. Internal Medicine services comprise a large proportion of those discharged, accounting for over 17,000 discharges per year; and is projected to grow at an annual rate of 4%. As service caps and work‐hour restrictions have limited the total number of patients that medical residents are able to care for, our hospitalist group has increased the number of physicians on the nonresident hospitalist service. At the time of the study, there were 23 hospitalists, equivalent to 18.25 full‐time equivalents (FTEs), staffing the service. The hospitalists provide in‐house patient care 24 hours a day and 7 days a week. Hospitalists also provide general medicine consult services, surgical comanagement and perioperative care, procedures, inpatient cardiopulmonary arrest response, rapid response team supervision, and observation care; and are also the primary inpatient physicians for many of the hospitalized interventional radiology and dermatology patients. These direct patient care activities account for 4500 annual discharges from the nonresident service.
Data Collection
Four university undergraduate business administration program students shadowed 11 hospitalists over a 3‐week period in 4‐hour to 12‐hour time blocks. The students followed the hospitalist on the shift that was taking admission calls, during day and night. A data collection tool was designed to track physicians' actions in 1‐minute increments, using categories similar to those used in a previously published time‐motion study of hospitalists' activities (Table 1).6 Physicians' activities each minute were assigned to a single category that most represented their action during that time period. At our AMC, 6 hospitalists work during the day shifts, and 2 on the night shifts. Our hospitalists may have patients in any of the 14 general care units in the hospital, as our hospitalists' services are not geographically based. The day hospitalists' shifts are scheduled from 7 AM to 7 PM. Two of the 6 hospitalists rotate through a 3‐day cycle as the admitting physician. Their duties include triaging and admitting patients until 2 PM, providing the day‐to‐day care for their patients until 7 PM, and occasionally cross‐covering for the other day‐shift hospitalists that have left for the day. The 4 other day‐shift hospitalists, not on their rotation as the admitting physician, may sign out and leave as early as 4 PM if their work for the day is done. At 2 PM, a separate swing‐shift hospitalist takes over the role of triaging and admitting until 7 PM. During the day shift, consults and perioperative management of patients are provided by a separate hospitalist on the consult service. At 7 PM, 2 nocturnists arrive for their 7 PM to 7 AM shift. The nocturnists, in addition to cross‐covering service patients, admit a maximum of 6 patients each, or until midnightwhichever comes first.
| Category | Code | Description |
|---|---|---|
| Direct patient care | DPIH | Initial history |
| DPDI | Discharge instructions | |
| DPFM | Family meetings | |
| DPRV | Revisit | |
| DPCC | Cross‐cover | |
| Indirect patient care | ||
| Documentation | IDGD | General documentation |
| IDDN | Daily notes | |
| IDDD | Discharge navigator | |
| Records/Results | IPMR | Review medical records |
| Communication | ICHH | Patient handoffs |
| ICFF | Face‐to‐face | |
| ICIP | Incoming page | |
| ICOP | Outgoing page | |
| ICIC | Incoming call | |
| ICOC | Outgoing call | |
| ICEE | E‐mail communications | |
| ICDP | Discharge planner | |
| Orders | IOWO | Writing orders |
| Professional development | PDRR | Reading articles, textbooks, references |
| Education | EEWR | Teaching during work rounds |
| Travel | TTTT | Travel |
| Personal | PPPP | Personal |
| Down time | DDDD | Downtime |
The students observed 11 different hospitalists, and followed these physicians during 9 weekday shifts, 5 weekday swing shifts, 10 weekday night shifts, and 4 weekend night shifts. The variance in the number of each type of shifts monitored was likely due to scheduling limitations of the students. In total, they collected data on 8,915 minutes of hospitalists' activities. The students monitored the hospitalists representing time periods from 7 AM to 2 AM. Analysis from 2 AM to 7 AM was excluded, because after 2 AM the hospitalists did not routinely evaluate new patients with the exception of emergent requests. New admissions after midnight are handled by a night float service staffed by residents.
Results
Overall, time spent on patient care activities comprised the bulk of hospitalists' shifts (82%) (Figure 1). Patient care activities were further categorized as direct patient caredefined as face‐to‐face patient or family time; and indirect patient caredefined as activities related to patient care, but without patient or family contact. Direct and indirect patient care accounted for 15% and 67% of the hospitalists' time, respectively. The other 18% of the hospitalists' time spent in the hospital were broadly categorized into: professional development, education, personal, downtime, and travel. Professional development included activities such as looking up information (eg, literature search); education included times that hospitalists spent with residents or medical students; personal time included only restroom and food breaks; and travel included time spent moving from 1 area to the next during their shift.

The majority of the hospitalists' direct patient care time was spent on evaluating new patients (79%). Significantly smaller amounts of time were spent on other direct care activities: cross‐covering other patients (8%), follow‐up visits (7%), family meetings (4%), and discharge instructions (2%) (Figure 2).

Indirect patient care activities included, 41% of time used to communicate with other healthcare providers, 26% on medical documentation, 20% reviewing medical records and results, and 13% of time writing orders (Figure 3). Communication accounted for a large proportion of a hospitalists' work, and included telephone conversations with Emergency Department (ED) or other admitting providers, handoffs, paging, face‐to‐face conversations with consultants and other support staff, and e‐mail.

Figure 4 shows the hourly distribution of time spent on direct and indirect patient care by a hospitalist throughout the day. The day‐time hospitalists pick up their signout from the nocturnists at 7 AM to begin their shift. The swing hospitalists arrive at 2 PM during the weekdays, and their primary duty is to triage and admit patients until 7 PM. The nocturnists start their shift at 7 PM, at which time the daytime and swing‐shift hospitalists all sign out for the night.

Discussion
Hospitalists on the nonresident service at our AMC utilize about 15% of their time on face‐to‐face patient care activities, 67% on indirect patient care activities, and 7% of time on moving from 1 part of the hospital to another. Hospitalists are valuable members of the physician work force who address the increasing patient care demands in the face of increasing limitations on residency work‐hours, a growing aging population, and existing inefficiencies in AMCs. The only other work‐flow study of hospitalists of which we are aware provided a single institution's perspective on time utilization by hospitalists. Our study in a different AMC setting revealed strong consistency with the O'Leary et al.6 study in the fraction of time hospitalists spent on direct patient care (15% and 18%, respectively), indirect patient care (67% and 69%); and within indirect patient care the time spent on documentation (26% and 37% of total time) and communications (41% and 35%). While travel in the O'Leary et al.6 study took up only 3% of hospitalists' time, the conclusions in that paper clearly suggest that the authors consider it an area of concern. Our study found that travel accounted for over 7% of hospitalists' time, confirming that intuition. The significant travel time may in part reflect the effects of a non‐geographically‐located hospitalist service. From these 2 studies we can be more confident that in large, tertiary care AMCs the time hospitalists spend on indirect patient care dominates that for direct patient care (by a factor of 4 in these studies), that within indirect patient care documentation and communication are dominant activities, and that travel can take a significant amount of time when patients are dispersed throughout the facility.
Both studies demonstrated that communication accounted for a significant proportion of a hospitalist's time. In our study communication accounted for 28% of their total time in the hospital, and 41% of the indirect patient care portion (Figure 3). A closer look within our communication category revealed that phone calls and handoffs accounted for two‐thirds of all communication time observed. As the hospitalists who carry the admitting pager, they receive the pages to take admission calls, but also take calls from consultants who have recommendations, as well as from nursing and other hospital staff. Depending on the nature of the conversation, the phone calls can last several minutes. While ensuring the communication between health care providers is complete and thorough, there may be opportunities to develop novel approaches to the way hospitalists communicate with other care providers. For example, at the UMHS, alternative communication methods with nursing staff have been proposed such as utilizing a website or a handheld device to help hospitalists prioritize their communications back to the nursing staff7; while standardizing the intake information from the ED or other admitting providers may help reduce the total time spent on phone calls. We will need to further explore the potential benefits of these ideas in future work.
Our data also reveal an interesting cyclicality of daily activities for the hospitalists, as shown in Figure 4. We identified batching behaviors throughout the day, which cause delays in seeing patients and can be deleterious to smooth workflows in support services. Spikes in indirect patient care, followed closely by spikes in direct patient care, occur regularly at shift changes (7 AM, 2 PM, and 7 PM). Also, in the night shift, indirect patient care drops to its lowest levels (in % of time spent) throughout the day, and direct patient care reaches its highest levels. The day‐shift indirect care profile is counter‐cyclical with direct care, as the hospitalist shifts between direct care and indirect care depending on the time of the day. We discuss these phenomena in turn.
It is known that variability in any operation causes congestion and delay, as an unavoidable consequence of the physics of material and information flows.8 Indeed, an entire subindustry based on Lean manufacturing principles has evolved from the Toyota Production System based on the elimination of unnecessary variability in operations.9 Lean processes have been ongoing in manufacturing facilities for decades, and these efforts are just recently being embraced by the service sector in general, and health care specifically.10, 11 Batching is an extreme form of variability, where there is a lull in the amount of work being done and then a burst of work is done over a short period of time. This means that jobs pile up in the queue waiting for the next spike of activity. Our data indicate batching seems to be a common phenomenon for our hospitalists. The majority of the patients admitted to our hospitalist service are unscheduled admissions that arrive primarily through the ED. One potential result of the unscheduled admissions is that patients could be referred to our hospitalist service at a pace that is not well predictable on an hour‐to‐hour basis. This could lead to an unintended result of multiple patients admitted over a short period of time. This means that many patients wait for intake, delaying the onset of their care by the inpatient physician. Also, since an initial exam often results in orders for laboratory tests and studies, batching on the floor will translate into batching of orders going to nursing, pathology, radiology, and other hospital support services. This imposes the cost of variability on these other services in the hospital. From a systems perspective, efficiency will improve if these activities can be smoothed throughout the day. This may suggest opportunities to work with the ED, to help smooth the inflow of patients into the hospital system.
Within the hospital, all of the day‐shift hospitalists can be reached about the needs of their respective patients, however, the physician carrying the admission pager also fields calls for admissions, and acts as the default contact person for the hospitalist group. As this hospitalist receives information on new admissions, he/she is aware of patients ready for intake but cannot evaluate them at the rate they are being referred, so the queue builds. This continues into the swing shift, which also fields referrals faster than they can attend to them. The volatility in indirect care during the swing shift, 2 PM to 7 PM, reflects a significant amount of triaging and fielding general calls about hospitalist patients. These activities further reduce the swing shift's ability to clear the intake queue. The night shift finally gets to these patients and, eventually, clears the queue. There may be an opportunity to consider the use of multiple input pagers or other process changes that can smooth this flow and rationalize the recurring tasks of finding patients and the responsible physician.
Another concept in Lean thinking is that variability is costly when it represents a mismatch between demand for a service and the capacity to serve. With regards to admitted patients, when demand outpaces capacity, patients will wait. When capacity outpaces demand, there is excess capacity in the system. The ideal is to match demand and capacity at all times, so nobody waits and the system carries no costly excess capacity. As the intake providers for admitted patients, we can attack this problem from the capacity side. Here, 2 generic Lean tactics are to: (1) reallocate resources to a bottleneck that is holding up the entire system, and (2) relieve workers of time‐consuming but non‐value‐adding work so they have more capacity to devote to serving demand. In our study, carrying multiple input pagers is an example of tactic (1), and efficient communication technologies and practices that reduce indirect time is an example of (2). Systemwide improvements would require further investigation by working with the variability on the input side (eg, ED admissions).
Our study also found that a significant percent of the time observed was spent traveling (7.4%) from room to room between different floors in the hospital. Travel time, which is non‐value‐adding, is one of the major forms of waste Lean thinking.12 Our hospitalists can provide care to patients at any of the general medical‐surgical beds we have available at our health system. These beds are distributed across 14 units on 5 different floors, as well as in the ED if a bed is not available for an admitted patient. In hospitals routinely operating at high occupancy, such as our AMC, patients often get distributed throughout the facility for lack of beds on the appropriate service's ward. One cost for this is a potential mismatch between a patient's needs and floor nurses' training. Our study reveals another cost, and that is its contribution to the significant amount of time hospitalists spent on travel, which is largely driven by the need to see dispersed patients. Reducing this cost requires a systemic, rather than service‐specific, solution. Our AMC is adding observation‐status beds to relieve some of the pressure on licensed beds, and considering bed management (including parts of the admissions and discharge processes) changes designed to promote better collocation of patients with services. Further study on these and other collocation tactics is warranted.
The spike in indirect activities at 4 PM represents, in part, an early signout by 1 or more of the hospitalists who are not scheduled to hold the admission pager, and have completed their work for the day. This handoff will be replicated at 7 PM when the nocturnists arrive for their night shift. In addition to a significant indirect load on physicians, multiple handoffs have been associated with decreased quality of care.13 Again, it is worthwhile considering the feasibility of alternative shift schedules that can minimize handoffs.
Finally, our findings revealed that a low percentage of time was dedicated to providing discharge instructions (2.24% of direct patient care time, and 0.34% of total time). Because the task of discharging patients falls primarily on the day‐shift hospitalists, when combined with swing‐shift and night‐shift hospitalists' data, the low percentage measured on discharge instructions may have been diluted. Nonetheless, this may point to the need for further investigation on how hospitalists provide direct patient encounter time during this critical phase of transition out of the hospital.
Our study is not without limitations. The student observers shadowed a representative group of hospitalists, but they were not able to follow everyone in the group. More specifically, their observations were made on the hospitalist who was carrying the primary hospitalist service admitting pager. Although it was the intent of our study to focus on the hospitalists we felt would be the busiest, our results may not be generalizable to all hospitalists. Although our research supports the previous findings by O'Leary et al.,6 a second limitation to our study is that our analysis was done at a single hospitalist group in an AMC, and hence the results may not be generalizable to other hospitalist groups. Another limitation may be that we did not do an evaluation of the hours between 2 AM to 7 AM. This period of time is used to catch up on medical documentation and to be available for medical emergencies. As more hospitalist programs are employing the use of nocturnists, it may be informative to have this time period tracked for activities.
Conclusions
Our study supports the broad allocation of hospitalist time found in an earlier study at a different AMC,6 suggesting that these might be generally representative in other AMCs. We found that travel constitutes a significant claim in hospitalists' time, due in part to the inability to collocate hospitalist service patients. Remedies are not likely to be service‐specific, but will require systemwide analyses of admission and discharge processes. Communication takes a significant amount of hospitalist time, with pages and phone calls related to handoffs accounting for most of the total communication time. As hospitalists working at non‐AMC settings may experience different work flow issues, we would like to see time‐motion studies of hospitalists in other types of hospitals. Future studies should also seek to better understand the how hospitals at high occupancy may reduce batching and streamline both the discharge and admission process, determine the factors that account for the significant communication time and how these processes could be streamlined, and evaluate the potential benefits of geographical localization of hospitalists' patients.
Acknowledgements
The authors thank Tracey Jackson, Michael Paulsen, Deepak Srinivasin, and Ryan Werblow, who were students in the undergraduate business school program, for their invaluable contribution in shadowing hospitalists to collect the time study data.
- , , , .Where should hospitalists sit within the academic medical center?J Gen Intern Med.2008;23:1269–1272.
- , .Hospitalists in teaching hospitals: opportunities but not without danger.J Gen Intern Med.2004;19:392–393.
- , , , , .Non‐housestaff medicine services in academic centers: models and challenges.J Hosp Med.2008;3:247–255.
- Society of Hospital Medicine. Society of Hospital Medicine Releases Results of the 2007–2008 Survey on the State of the Hospital Medicine Movement.2008. Available at: http://www.hospitalmedicine.org/AM/Template.cfm? Section=Press_Releases3:398–402.
- , , .How hospitalists spend their time: insights on efficiency and safety.J Hosp Med.2006;1:88–93.
- , .MCOMM: Redefining Medical Communication in the 21st Century, University of Michigan Health System. In: Society of Hospital Medicine Annual Meeting, Best of Innovations Presentation; 2009; Chicago, IL;2009.
- , .Factory Physics: Foundations of Manufacturing Management.Boston:Irwin, McGraw‐Hill;1996.
- .The Toyota Way.1st ed.Madison, WI:McGraw‐Hill;2004.
- Going Lean in Health Care.White Paper.Boston, MA:Institute for Healthcare Improvement;2005 January and February, 2005. Available at: http://www.ihconline.org/toolkits/LeanInHealthcare/GoingLeaninHealth CareWhitePaper.pdf. Accessed September 2009.
- , , , .Lean health care: what can hospitals learn from a world‐class automaker?J Hosp Med.2006;1:191–199.
- , , , , .Managing Business Process Flows.Upper Saddle River, NJ:Prentice Hall;2006.
- , .The patient handoff: medicine's Formula One moment.Chest.2008;134:9–12.
- , , , .Where should hospitalists sit within the academic medical center?J Gen Intern Med.2008;23:1269–1272.
- , .Hospitalists in teaching hospitals: opportunities but not without danger.J Gen Intern Med.2004;19:392–393.
- , , , , .Non‐housestaff medicine services in academic centers: models and challenges.J Hosp Med.2008;3:247–255.
- Society of Hospital Medicine. Society of Hospital Medicine Releases Results of the 2007–2008 Survey on the State of the Hospital Medicine Movement.2008. Available at: http://www.hospitalmedicine.org/AM/Template.cfm? Section=Press_Releases3:398–402.
- , , .How hospitalists spend their time: insights on efficiency and safety.J Hosp Med.2006;1:88–93.
- , .MCOMM: Redefining Medical Communication in the 21st Century, University of Michigan Health System. In: Society of Hospital Medicine Annual Meeting, Best of Innovations Presentation; 2009; Chicago, IL;2009.
- , .Factory Physics: Foundations of Manufacturing Management.Boston:Irwin, McGraw‐Hill;1996.
- .The Toyota Way.1st ed.Madison, WI:McGraw‐Hill;2004.
- Going Lean in Health Care.White Paper.Boston, MA:Institute for Healthcare Improvement;2005 January and February, 2005. Available at: http://www.ihconline.org/toolkits/LeanInHealthcare/GoingLeaninHealth CareWhitePaper.pdf. Accessed September 2009.
- , , , .Lean health care: what can hospitals learn from a world‐class automaker?J Hosp Med.2006;1:191–199.
- , , , , .Managing Business Process Flows.Upper Saddle River, NJ:Prentice Hall;2006.
- , .The patient handoff: medicine's Formula One moment.Chest.2008;134:9–12.
Copyright © 2010 Society of Hospital Medicine
The renal failure that vanished
A 35‐year‐old African American female presented to her primary care provider with a 4‐day history of progressive nausea, vomiting, and generalized malaise. The patient had been in her usual state of health prior to the onset of these symptoms and had no history of prior hospitalization. She denied any fevers, chills, abdominal pain, or change in diet prior to the onset of her symptoms. She also had no recent exposure to sick contacts, human immunodeficiency virus (HIV) risk factors, or history of recent travel. One week prior to her presentation, the patient had been prescribed rifampin for treatment of chronic hidradenitis suppurativa. She had been taking rifampin for 5 days until she developed her current symptoms. The patient was not taking any other medications and had no other medical problems.
On presentation, the patient was afebrile and her vital signs were within normal limits. She was alert and oriented with no scleral icterus. Cardiopulmonary exam was within normal limits. Her abdomen was nondistended with diffuse nonlocalizing tenderness, normal bowel sounds, and no signs of acute abdomen. No hepatomegaly was noted, and stool was negative for occult blood. No rashes or joint abnormalities were noted on exam, but multiple nodulocystic lesions were noted bilaterally in her axillae. Laboratory findings on presentation were most notable for a blood urea nitrogen level of 38 mg/dL, a creatinine of 5.3 mg/dL, and a calculated fractional excretion of sodium of 2.6%. Urine analysis revealed no significant hematuria, proteinuria, or red blood cell casts, but did demonstrate white blood cells, white blood cell casts, and eosinophils. Blood cultures drawn on admission were negative and the patient had a normal leukocyte count.
The patient was admitted to the general medicine service and the causes of her acute renal failure were explored. She was treated with intravenous fluids because a component of prerenal azotemia was initially suspected. Rifampin was discontinued. Despite significant hydration, the patient remained oliguric. She was challenged with high‐dose loop diuretics for 3 days but still remained oliguric. Renal ultrasound showed moderately echogenic, large 16‐cm kidneys bilaterally, with no evidence of hydronephrosis or renal calculi. Laboratory evaluation for diabetes and infiltrative disease of the kidneys such as HIV, amyloidosis, and nonspecific gammopathies were negative. The patient's creatinine level steadily increased and eventually peaked at 14.2 mg/dL. When the patient began to develop shortness of breath, lower extremity edema, and abdominal distension on hospital day 4, hemodialysis was initiated. On hospital day 6, the patient underwent a renal biopsy (Figure 1) that demonstrated patchy inflammatory infiltrates with scattered eosinophils and evidence of interstitial edema and tubulitis. Congo red staining was negative for amyloid and no immune deposits were noted. A diagnosis of acute interstitial nephritis (AIN) was made and the patient was started on high‐dose prednisone.
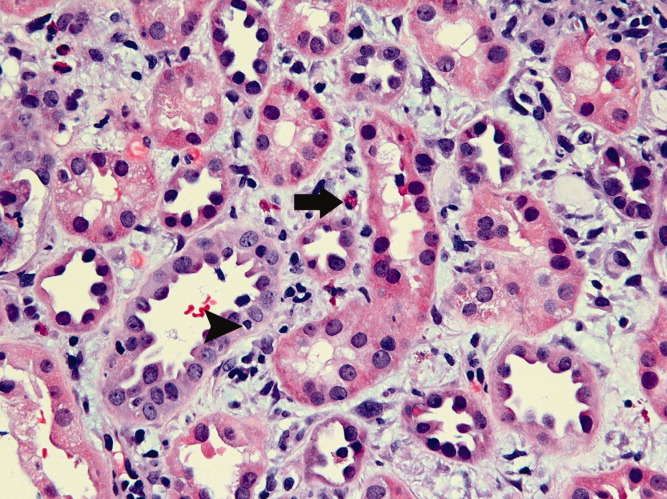
Over the 48 hours following initiation of prednisone therapy, the patient's urine output gradually began to improve and the patient was producing over 2 liters of urine per day. In addition, the patient's axillary cystic lesions became less inflamed and painful. The patient was discharged home with plans to continue hemodialysis as an outpatient. Three days after discharge, when the patient presented for hemodialysis, her creatinine was noted to be 1.2 mg/dL. Due to her improved creatinine and maintenance of good urine output, hemodialysis was discontinued. The patient was slowly tapered off her prednisone over the next several weeks. One month later her creatinine was 0.9 mg/dL. She had required no further hemodialysis since her hospitalization.
DISCUSSION
AIN is an uncommon but significant cause of acute renal failure, and accounts for 2% to 3% of all renal biopsies performed.1 AIN is thought to be an immune‐mediated process, and drug‐induced hypersensitivity is the most common cause of AIN. Nonsteroidal antiinflammatory drug (NSAID) use, antibiotics, proton pump inhibitors, and several other medications have been implicated in the pathogenesis of AIN. Rifampin is a medication that has a known association with AIN, with most cases being described in regions where treatment of endemic tuberculosis is common. The majority of cases of rifampin‐induced AIN occur in the setting of drug reexposure, due to an immunologically‐mediated process that causes tubulointerstitial injury.2
Patients with drug‐induced AIN typically present with oliguria secondary to an acute decline in renal function. The classic clinical triad of fever, rash, and arthralgias is uncommon, and all 3 occur in only 30% of all cases.3 More commonly, patients typically present with vague flu‐like and gastrointestinal symptoms, including fever, abdominal pain, nausea, and vomiting. Urinalysis may be helpful, but hematuria occurs in less than one‐half of all cases, and sterile pyuria is common but not always present. It has been suggested that the presence of eosinophiluria may lead to high suspicion of AIN, but the sensitivity and specificity of eosinophiluria are low, at 40% and 72%, respectively.3 Thus, renal biopsy is often performed to make a confirmatory diagnosis of AIN in the appropriate clinical setting. Histopathologically, the presence of inflammatory infiltrates in the renal tubules and interstitium with conservation of the glomerular structures is visualized.2, 3
A large number of patients who present with AIN may require temporary renal replacement therapy; however, most patients have been observed to recover full renal function. Despite this, review of the literature shows that many patients may have persistent elevations in their serum creatinine. Corticosteroid therapy, although controversial, has commonly been initiated in patients whose renal function does not improve with conservative therapy. To date there are no prospective randomized clinical trials, and data guiding optimal management in AIN is sparse. Some studies have demonstrated no benefit in corticosteroid therapy in lowering serum creatinine levels in patients with AIN,4 but others have observed a significantly increased risk of interstitial fibrosis and failure to return to baseline creatinine in those patients that received delayed treatment with corticosteroids more than 1 week after the withdrawal of the offending agent.5
The patient described in our case did not present with the classic symptoms noted in AIN. Yet she had evidence of eosinophiluria, which increased our suspicion for AIN. Although other potential etiologies of this patient's acute renal failure were considered, given her negative serologic studies and the results of her renal biopsy, AIN was considered the leading diagnosis. Since AIN was recognized early in this patient, the offending medication was discontinued promptly, prednisone therapy was initiated appropriately, and the renal failure that had developed quickly vanished.
- ,.Acute interstitial nephritis.J Am Soc Nephrol.1998;9(3):506–515.
- ,,,,.Rifampicin‐associated acute renal failure: pathophysiologic, immunologic, and clinical features.Am J Kidney Dis.1998;31(1):108–115.
- ,.A reversible cause of acute renal failure.Postgrad Med J.2001;77(909):478–480.
- ,,, et al.Acute interstitial nephritis: clinical features and response to corticosteroid therapy.Nephrol Dial Transplant.2004;19:2778–2783.
- , ,, et al.Early steroid treatment improves the recovery of renal function in patients with drug‐induced acute interstitial nephritis.Kidney Int.2008;73(8):940–946.
A 35‐year‐old African American female presented to her primary care provider with a 4‐day history of progressive nausea, vomiting, and generalized malaise. The patient had been in her usual state of health prior to the onset of these symptoms and had no history of prior hospitalization. She denied any fevers, chills, abdominal pain, or change in diet prior to the onset of her symptoms. She also had no recent exposure to sick contacts, human immunodeficiency virus (HIV) risk factors, or history of recent travel. One week prior to her presentation, the patient had been prescribed rifampin for treatment of chronic hidradenitis suppurativa. She had been taking rifampin for 5 days until she developed her current symptoms. The patient was not taking any other medications and had no other medical problems.
On presentation, the patient was afebrile and her vital signs were within normal limits. She was alert and oriented with no scleral icterus. Cardiopulmonary exam was within normal limits. Her abdomen was nondistended with diffuse nonlocalizing tenderness, normal bowel sounds, and no signs of acute abdomen. No hepatomegaly was noted, and stool was negative for occult blood. No rashes or joint abnormalities were noted on exam, but multiple nodulocystic lesions were noted bilaterally in her axillae. Laboratory findings on presentation were most notable for a blood urea nitrogen level of 38 mg/dL, a creatinine of 5.3 mg/dL, and a calculated fractional excretion of sodium of 2.6%. Urine analysis revealed no significant hematuria, proteinuria, or red blood cell casts, but did demonstrate white blood cells, white blood cell casts, and eosinophils. Blood cultures drawn on admission were negative and the patient had a normal leukocyte count.
The patient was admitted to the general medicine service and the causes of her acute renal failure were explored. She was treated with intravenous fluids because a component of prerenal azotemia was initially suspected. Rifampin was discontinued. Despite significant hydration, the patient remained oliguric. She was challenged with high‐dose loop diuretics for 3 days but still remained oliguric. Renal ultrasound showed moderately echogenic, large 16‐cm kidneys bilaterally, with no evidence of hydronephrosis or renal calculi. Laboratory evaluation for diabetes and infiltrative disease of the kidneys such as HIV, amyloidosis, and nonspecific gammopathies were negative. The patient's creatinine level steadily increased and eventually peaked at 14.2 mg/dL. When the patient began to develop shortness of breath, lower extremity edema, and abdominal distension on hospital day 4, hemodialysis was initiated. On hospital day 6, the patient underwent a renal biopsy (Figure 1) that demonstrated patchy inflammatory infiltrates with scattered eosinophils and evidence of interstitial edema and tubulitis. Congo red staining was negative for amyloid and no immune deposits were noted. A diagnosis of acute interstitial nephritis (AIN) was made and the patient was started on high‐dose prednisone.

Over the 48 hours following initiation of prednisone therapy, the patient's urine output gradually began to improve and the patient was producing over 2 liters of urine per day. In addition, the patient's axillary cystic lesions became less inflamed and painful. The patient was discharged home with plans to continue hemodialysis as an outpatient. Three days after discharge, when the patient presented for hemodialysis, her creatinine was noted to be 1.2 mg/dL. Due to her improved creatinine and maintenance of good urine output, hemodialysis was discontinued. The patient was slowly tapered off her prednisone over the next several weeks. One month later her creatinine was 0.9 mg/dL. She had required no further hemodialysis since her hospitalization.
DISCUSSION
AIN is an uncommon but significant cause of acute renal failure, and accounts for 2% to 3% of all renal biopsies performed.1 AIN is thought to be an immune‐mediated process, and drug‐induced hypersensitivity is the most common cause of AIN. Nonsteroidal antiinflammatory drug (NSAID) use, antibiotics, proton pump inhibitors, and several other medications have been implicated in the pathogenesis of AIN. Rifampin is a medication that has a known association with AIN, with most cases being described in regions where treatment of endemic tuberculosis is common. The majority of cases of rifampin‐induced AIN occur in the setting of drug reexposure, due to an immunologically‐mediated process that causes tubulointerstitial injury.2
Patients with drug‐induced AIN typically present with oliguria secondary to an acute decline in renal function. The classic clinical triad of fever, rash, and arthralgias is uncommon, and all 3 occur in only 30% of all cases.3 More commonly, patients typically present with vague flu‐like and gastrointestinal symptoms, including fever, abdominal pain, nausea, and vomiting. Urinalysis may be helpful, but hematuria occurs in less than one‐half of all cases, and sterile pyuria is common but not always present. It has been suggested that the presence of eosinophiluria may lead to high suspicion of AIN, but the sensitivity and specificity of eosinophiluria are low, at 40% and 72%, respectively.3 Thus, renal biopsy is often performed to make a confirmatory diagnosis of AIN in the appropriate clinical setting. Histopathologically, the presence of inflammatory infiltrates in the renal tubules and interstitium with conservation of the glomerular structures is visualized.2, 3
A large number of patients who present with AIN may require temporary renal replacement therapy; however, most patients have been observed to recover full renal function. Despite this, review of the literature shows that many patients may have persistent elevations in their serum creatinine. Corticosteroid therapy, although controversial, has commonly been initiated in patients whose renal function does not improve with conservative therapy. To date there are no prospective randomized clinical trials, and data guiding optimal management in AIN is sparse. Some studies have demonstrated no benefit in corticosteroid therapy in lowering serum creatinine levels in patients with AIN,4 but others have observed a significantly increased risk of interstitial fibrosis and failure to return to baseline creatinine in those patients that received delayed treatment with corticosteroids more than 1 week after the withdrawal of the offending agent.5
The patient described in our case did not present with the classic symptoms noted in AIN. Yet she had evidence of eosinophiluria, which increased our suspicion for AIN. Although other potential etiologies of this patient's acute renal failure were considered, given her negative serologic studies and the results of her renal biopsy, AIN was considered the leading diagnosis. Since AIN was recognized early in this patient, the offending medication was discontinued promptly, prednisone therapy was initiated appropriately, and the renal failure that had developed quickly vanished.
A 35‐year‐old African American female presented to her primary care provider with a 4‐day history of progressive nausea, vomiting, and generalized malaise. The patient had been in her usual state of health prior to the onset of these symptoms and had no history of prior hospitalization. She denied any fevers, chills, abdominal pain, or change in diet prior to the onset of her symptoms. She also had no recent exposure to sick contacts, human immunodeficiency virus (HIV) risk factors, or history of recent travel. One week prior to her presentation, the patient had been prescribed rifampin for treatment of chronic hidradenitis suppurativa. She had been taking rifampin for 5 days until she developed her current symptoms. The patient was not taking any other medications and had no other medical problems.
On presentation, the patient was afebrile and her vital signs were within normal limits. She was alert and oriented with no scleral icterus. Cardiopulmonary exam was within normal limits. Her abdomen was nondistended with diffuse nonlocalizing tenderness, normal bowel sounds, and no signs of acute abdomen. No hepatomegaly was noted, and stool was negative for occult blood. No rashes or joint abnormalities were noted on exam, but multiple nodulocystic lesions were noted bilaterally in her axillae. Laboratory findings on presentation were most notable for a blood urea nitrogen level of 38 mg/dL, a creatinine of 5.3 mg/dL, and a calculated fractional excretion of sodium of 2.6%. Urine analysis revealed no significant hematuria, proteinuria, or red blood cell casts, but did demonstrate white blood cells, white blood cell casts, and eosinophils. Blood cultures drawn on admission were negative and the patient had a normal leukocyte count.
The patient was admitted to the general medicine service and the causes of her acute renal failure were explored. She was treated with intravenous fluids because a component of prerenal azotemia was initially suspected. Rifampin was discontinued. Despite significant hydration, the patient remained oliguric. She was challenged with high‐dose loop diuretics for 3 days but still remained oliguric. Renal ultrasound showed moderately echogenic, large 16‐cm kidneys bilaterally, with no evidence of hydronephrosis or renal calculi. Laboratory evaluation for diabetes and infiltrative disease of the kidneys such as HIV, amyloidosis, and nonspecific gammopathies were negative. The patient's creatinine level steadily increased and eventually peaked at 14.2 mg/dL. When the patient began to develop shortness of breath, lower extremity edema, and abdominal distension on hospital day 4, hemodialysis was initiated. On hospital day 6, the patient underwent a renal biopsy (Figure 1) that demonstrated patchy inflammatory infiltrates with scattered eosinophils and evidence of interstitial edema and tubulitis. Congo red staining was negative for amyloid and no immune deposits were noted. A diagnosis of acute interstitial nephritis (AIN) was made and the patient was started on high‐dose prednisone.

Over the 48 hours following initiation of prednisone therapy, the patient's urine output gradually began to improve and the patient was producing over 2 liters of urine per day. In addition, the patient's axillary cystic lesions became less inflamed and painful. The patient was discharged home with plans to continue hemodialysis as an outpatient. Three days after discharge, when the patient presented for hemodialysis, her creatinine was noted to be 1.2 mg/dL. Due to her improved creatinine and maintenance of good urine output, hemodialysis was discontinued. The patient was slowly tapered off her prednisone over the next several weeks. One month later her creatinine was 0.9 mg/dL. She had required no further hemodialysis since her hospitalization.
DISCUSSION
AIN is an uncommon but significant cause of acute renal failure, and accounts for 2% to 3% of all renal biopsies performed.1 AIN is thought to be an immune‐mediated process, and drug‐induced hypersensitivity is the most common cause of AIN. Nonsteroidal antiinflammatory drug (NSAID) use, antibiotics, proton pump inhibitors, and several other medications have been implicated in the pathogenesis of AIN. Rifampin is a medication that has a known association with AIN, with most cases being described in regions where treatment of endemic tuberculosis is common. The majority of cases of rifampin‐induced AIN occur in the setting of drug reexposure, due to an immunologically‐mediated process that causes tubulointerstitial injury.2
Patients with drug‐induced AIN typically present with oliguria secondary to an acute decline in renal function. The classic clinical triad of fever, rash, and arthralgias is uncommon, and all 3 occur in only 30% of all cases.3 More commonly, patients typically present with vague flu‐like and gastrointestinal symptoms, including fever, abdominal pain, nausea, and vomiting. Urinalysis may be helpful, but hematuria occurs in less than one‐half of all cases, and sterile pyuria is common but not always present. It has been suggested that the presence of eosinophiluria may lead to high suspicion of AIN, but the sensitivity and specificity of eosinophiluria are low, at 40% and 72%, respectively.3 Thus, renal biopsy is often performed to make a confirmatory diagnosis of AIN in the appropriate clinical setting. Histopathologically, the presence of inflammatory infiltrates in the renal tubules and interstitium with conservation of the glomerular structures is visualized.2, 3
A large number of patients who present with AIN may require temporary renal replacement therapy; however, most patients have been observed to recover full renal function. Despite this, review of the literature shows that many patients may have persistent elevations in their serum creatinine. Corticosteroid therapy, although controversial, has commonly been initiated in patients whose renal function does not improve with conservative therapy. To date there are no prospective randomized clinical trials, and data guiding optimal management in AIN is sparse. Some studies have demonstrated no benefit in corticosteroid therapy in lowering serum creatinine levels in patients with AIN,4 but others have observed a significantly increased risk of interstitial fibrosis and failure to return to baseline creatinine in those patients that received delayed treatment with corticosteroids more than 1 week after the withdrawal of the offending agent.5
The patient described in our case did not present with the classic symptoms noted in AIN. Yet she had evidence of eosinophiluria, which increased our suspicion for AIN. Although other potential etiologies of this patient's acute renal failure were considered, given her negative serologic studies and the results of her renal biopsy, AIN was considered the leading diagnosis. Since AIN was recognized early in this patient, the offending medication was discontinued promptly, prednisone therapy was initiated appropriately, and the renal failure that had developed quickly vanished.
- ,.Acute interstitial nephritis.J Am Soc Nephrol.1998;9(3):506–515.
- ,,,,.Rifampicin‐associated acute renal failure: pathophysiologic, immunologic, and clinical features.Am J Kidney Dis.1998;31(1):108–115.
- ,.A reversible cause of acute renal failure.Postgrad Med J.2001;77(909):478–480.
- ,,, et al.Acute interstitial nephritis: clinical features and response to corticosteroid therapy.Nephrol Dial Transplant.2004;19:2778–2783.
- , ,, et al.Early steroid treatment improves the recovery of renal function in patients with drug‐induced acute interstitial nephritis.Kidney Int.2008;73(8):940–946.
- ,.Acute interstitial nephritis.J Am Soc Nephrol.1998;9(3):506–515.
- ,,,,.Rifampicin‐associated acute renal failure: pathophysiologic, immunologic, and clinical features.Am J Kidney Dis.1998;31(1):108–115.
- ,.A reversible cause of acute renal failure.Postgrad Med J.2001;77(909):478–480.
- ,,, et al.Acute interstitial nephritis: clinical features and response to corticosteroid therapy.Nephrol Dial Transplant.2004;19:2778–2783.
- , ,, et al.Early steroid treatment improves the recovery of renal function in patients with drug‐induced acute interstitial nephritis.Kidney Int.2008;73(8):940–946.
A fall to remember
A healthy 44‐year‐old man presented to the emergency room with buttock pain, 2 days after falling from a 4‐foot table onto a cement floor, striking his right buttock. On presentation he was in moderate distress, with heart rate = 130 beats per minute (bpm) and blood pressure = 80/40 mm Hg. There was a 2‐cm area of erythema on the right flank, and mild tenderness of the right buttock. There was no evidence of skin anesthesia or crepitance. Initial laboratory results were notable for creatinine = 1.5 mg/dl, creatine phosphokinase = 11,500 U/L, lactate = 6.1 mmo/L, and white blood cell count = 10 k/UL, with 93% neutrophils. Plain radiography of the hip and spine were negative for fractures and soft tissue gas. Despite receiving multiple doses of narcotic analgesia, the patient continued to complain of severe pain. Within 2 hours of presentation, the right flank erythema extended proximally up the back and distally to the right thigh (Figure 1). The patient was taken to the operating room for surgical exploration and was diagnosed with necrotizing fasciitis extending from his posterior neck to the right popliteal fossa (Figures 2 and 3). Intraoperative cultures grew Group A streptococcus, confirming a diagnosis of Type II necrotizing fasciitis. The patient required multiple debridements and subsequent reconstructive procedures. After more than 3 months in the hospital and acute rehabilitation, he returned home.
Necrotizing fasciitis is a rapidly progressive infection that spreads along fascial planes, causing necrosis of subcutaneous tissues. Type II necrotizing fasciitis is a monomicrobial infection and occurs in healthy patients, with blunt trauma being a known precipitant. The classic finding in necrotizing fasciitis is pain out of proportion to the physical examination. Additionally, patients will typically present with septic shock and end‐organ dysfunction. Diagnosis requires a strong index of suspicion and surgical exploration is necessary for diagnosis and treatment.
A healthy 44‐year‐old man presented to the emergency room with buttock pain, 2 days after falling from a 4‐foot table onto a cement floor, striking his right buttock. On presentation he was in moderate distress, with heart rate = 130 beats per minute (bpm) and blood pressure = 80/40 mm Hg. There was a 2‐cm area of erythema on the right flank, and mild tenderness of the right buttock. There was no evidence of skin anesthesia or crepitance. Initial laboratory results were notable for creatinine = 1.5 mg/dl, creatine phosphokinase = 11,500 U/L, lactate = 6.1 mmo/L, and white blood cell count = 10 k/UL, with 93% neutrophils. Plain radiography of the hip and spine were negative for fractures and soft tissue gas. Despite receiving multiple doses of narcotic analgesia, the patient continued to complain of severe pain. Within 2 hours of presentation, the right flank erythema extended proximally up the back and distally to the right thigh (Figure 1). The patient was taken to the operating room for surgical exploration and was diagnosed with necrotizing fasciitis extending from his posterior neck to the right popliteal fossa (Figures 2 and 3). Intraoperative cultures grew Group A streptococcus, confirming a diagnosis of Type II necrotizing fasciitis. The patient required multiple debridements and subsequent reconstructive procedures. After more than 3 months in the hospital and acute rehabilitation, he returned home.



Necrotizing fasciitis is a rapidly progressive infection that spreads along fascial planes, causing necrosis of subcutaneous tissues. Type II necrotizing fasciitis is a monomicrobial infection and occurs in healthy patients, with blunt trauma being a known precipitant. The classic finding in necrotizing fasciitis is pain out of proportion to the physical examination. Additionally, patients will typically present with septic shock and end‐organ dysfunction. Diagnosis requires a strong index of suspicion and surgical exploration is necessary for diagnosis and treatment.
A healthy 44‐year‐old man presented to the emergency room with buttock pain, 2 days after falling from a 4‐foot table onto a cement floor, striking his right buttock. On presentation he was in moderate distress, with heart rate = 130 beats per minute (bpm) and blood pressure = 80/40 mm Hg. There was a 2‐cm area of erythema on the right flank, and mild tenderness of the right buttock. There was no evidence of skin anesthesia or crepitance. Initial laboratory results were notable for creatinine = 1.5 mg/dl, creatine phosphokinase = 11,500 U/L, lactate = 6.1 mmo/L, and white blood cell count = 10 k/UL, with 93% neutrophils. Plain radiography of the hip and spine were negative for fractures and soft tissue gas. Despite receiving multiple doses of narcotic analgesia, the patient continued to complain of severe pain. Within 2 hours of presentation, the right flank erythema extended proximally up the back and distally to the right thigh (Figure 1). The patient was taken to the operating room for surgical exploration and was diagnosed with necrotizing fasciitis extending from his posterior neck to the right popliteal fossa (Figures 2 and 3). Intraoperative cultures grew Group A streptococcus, confirming a diagnosis of Type II necrotizing fasciitis. The patient required multiple debridements and subsequent reconstructive procedures. After more than 3 months in the hospital and acute rehabilitation, he returned home.



Necrotizing fasciitis is a rapidly progressive infection that spreads along fascial planes, causing necrosis of subcutaneous tissues. Type II necrotizing fasciitis is a monomicrobial infection and occurs in healthy patients, with blunt trauma being a known precipitant. The classic finding in necrotizing fasciitis is pain out of proportion to the physical examination. Additionally, patients will typically present with septic shock and end‐organ dysfunction. Diagnosis requires a strong index of suspicion and surgical exploration is necessary for diagnosis and treatment.
Pediatric Hospital Medicine Core Competencies
Introduction
The Society of Hospital Medicine (SHM) defines hospitalists as physicians whose primary professional focus is the comprehensive general medical care of hospitalized patients. Their activities include patient care, teaching, research, and leadership related to Hospital Medicine.1 It is estimated that there are up to 2500 pediatric hospitalists in the United States, with continued growth due to the converging needs for a dedicated focus on patient safety, quality improvement, hospital throughput, and inpatient teaching.2‐9 (Pediatric Hospital Medicine (PHM), as defined today, has been practiced in the United States for at least 30 years10 and continues to evolve as an area of specialization, with the refinement of a distinct knowledgebase and skill set focused on the provision of high quality general pediatric care in the inpatient setting. PHM is the latest site‐specific specialty to emerge from the field of general pediatrics it's development analogous to the evolution of critical care or emergency medicine during previous decades.11 Adult hospital medicine has defined itself within the field of general internal medicine12 and has recently received approval to provide a recognized focus of practice exam in 2010 for those re‐certifying with the American Board of Internal Medicine,13 PHM is creating an identity as a subspecialty practice with distinct focus on inpatient care for children within the larger context of general pediatric care.8, 14
The Pediatric Hospital Medicine Core Competencies were created to help define the roles and expectations for pediatric hospitalists, regardless of practice setting. The intent is to provide a unified approach toward identifying the specific body of knowledge and measurable skills needed to assure delivery of the highest quality of care for all hospitalized pediatric patients. Most children requiring hospitalization in the United States are hospitalized in community settings where subspecialty support is more limited and many pediatric services may be unavailable. Children with complex, chronic medical problems, however, are more likely to be hospitalized at a tertiary care or academic institutions. In order to unify pediatric hospitalists who work in different practice environments, the PHM Core Competencies were constructed to represent the knowledge, skills, attitudes, and systems improvements that all pediatric hospitalists can be expected to acquire and maintain.
Furthermore, the content of the PHM Core Competencies reflect the fact that children are a vulnerable population. Their care requires attention to many elements which distinguishes it from that given to the majority of the adult population: dependency, differences in developmental physiology and behavior, occurrence of congenital genetic disorders and age‐based clinical conditions, impact of chronic disease states on whole child development, and weight‐based medication dosing often with limited guidance from pediatric studies, to name a few. Awareness of these needs must be heightened when a child enters the hospital where diagnoses, procedures, and treatments often include use of high‐risk modalities and require coordination of care across multiple providers.
Pediatric hospitalists commonly work to improve the systems of care in which they operate and therefore both clinical and non‐clinical topics are included. The 54 chapters address the fundamental and most common components of inpatient care but are not an extensive review of all aspects of inpatient medicine encountered by those caring for hospitalized children. Finally, the PHM Core Competencies are not intended for use in assessing proficiency immediately post‐residency, but do provide a framework for the education and evaluation of both physicians‐in‐training and practicing hospitalists. Meeting these competencies is anticipated to take from one to three years of active practice in pediatric hospital medicine, and may be reached through a combination of practice experience, course work, self‐directed work, and/or formalized training.
Methods
Timeline
In 2002, SHM convened an educational summit from which there was a resolution to create core competencies. Following the summit, the SHM Pediatric Core Curriculum Task Force (CCTF) was created, which included 12 pediatric hospitalists practicing in academic and community facilities, as well as teaching and non‐teaching settings, and occupying leadership positions within institutions of varied size and geographic location. Shortly thereafter, in November 2003, approximately 130 pediatric hospitalists attended the first PHM meeting in San Antonio, Texas.11 At this meeting, with support from leaders in pediatric emergency medicine, first discussions regarding PHM scope of practice were held.
Formal development of the competencies began in 2005 in parallel to but distinct from SHM's adult work, which culminated in The Core Competencies in Hospital Medicine: A Framework for Curriculum Development published in 2006. The CCTF divided into three groups, focused on clinical, procedural, and systems‐based topics. Face‐to‐face meetings were held at the SHM annual meetings, with most work being completed by phone and electronically in the interim periods. In 2007, due to the overlapping interests of the three core pediatric societies, the work was transferred to leaders within the APA. In 2008 the work was transferred back to the leadership within SHM. Since that time, external reviewers were solicited, new chapters created, sections re‐aligned, internal and external reviewer comments incorporated, and final edits for taxonomy, content, and formatting were completed (Table 1).
| Date | Event |
|---|---|
| Feb 2002 | SHM Educational Summit held and CCTF created |
| Oct 2003 | 1st PHM meeting held in San Antonio |
| 2003‐2007 | Chapter focus determined; contributors engaged |
| 2007‐2008 | APA PHM Special Interest Group (SIG) review; creation of separate PHM Fellowship Competencies (not in this document) |
| Aug 2008‐Oct 2008 | SHM Pediatric Committee and CCTF members resume work; editorial review |
| Oct 2008‐Mar 2009 | Internal review: PHM Fellowship Director, AAP, APA, and SHM section/committee leader, and key national PHM leader reviews solicited and returned |
| Mar 2009 | PHM Fellowship Director comments addressed; editorial review |
| Mar‐Apr 2009 | External reviewers solicited from national agencies and societies relevant to PHM |
| Apr‐July 2009 | External reviewer comments returned |
| July‐Oct 2009 | Contributor review of all comments; editorial review, sections revised |
| Oct 2009 | Final review: Chapters to SHM subcommittees and Board |
Areas of Focused Practice
The PHM Core Competencies were conceptualized similarly to the SHM adult core competencies. Initial sections were divided into clinical conditions, procedures, and systems. However as content developed and reviewer comments were addressed, the four final sections were modified to those noted in Table 2. For the Common Clinical Diagnoses and Conditions, the goal was to select conditions most commonly encountered by pediatric hospitalists. Non‐surgical diagnosis‐related group (DRG) conditions were selected from the following sources: The Joint Commission's (TJC) Oryx Performance Measures Report15‐16 (asthma, abdominal pain, acute gastroenteritis, simple pneumonia); Child Health Corporation of America's Pediatric Health Information System Dataset (CHCA PHIS, Shawnee Mission, KS), and relevant publications on common pediatric hospitalizations.17 These data were compared to billing data from randomly‐selected practicing hospitalists representing free‐standing children's and community hospitals, teaching and non‐teaching settings, and urban and rural locations. The 22 clinical conditions chosen by the CCTF were those most relevant to the practice of pediatric hospital medicine.
| Common Clinical Diagnoses and Conditions | Specialized Clinical Services | Core Skills | Healthcare Systems: Supporting and Advancing Child Health | |
|---|---|---|---|---|
| Acute abdominal pain and the acute abdomen | Neonatal fever | Child abuse and neglect | Bladder catheterization/suprapubic bladder tap | Advocacy |
| Apparent life‐threatening event | Neonatal jaundice | Hospice and palliative care | Electrocardiogram interpretation | Business practices |
| Asthma | Pneumonia | Leading a healthcare team | Feeding tubes | Communication |
| Bone and joint infections | Respiratory failure | Newborn care and delivery room management | Fluids and electrolyte management | Continuous quality improvement |
| Bronchiolitis | Seizures | Technology‐dependent children | Intravenous access and phlebotomy | Cost‐effective care |
| Central nervous system infections | Shock | Transport of the critically ill child | Lumbar puncture | Education |
| Diabetes mellitus | Sickle cell disease | Non‐invasive monitoring | Ethics | |
| Failure to thrive | Skin and soft tissue infection | Nutrition | Evidence‐based medicine | |
| Fever of unknown origin | Toxic ingestion | Oxygen delivery and airway management | Health information systems | |
| Gastroenteritis | Upper airway infections | Pain management | Legal issues/risk management | |
| Kawasaki disease | Urinary tract infections | Pediatric advanced life support | Patient safety |
The Specialized Clinical Servicessection addresses important components of care that are not DRG‐based and reflect the unique needs of hospitalized children, as assessed by the CCTF, editors, and contributors. Core Skillswere chosen based on the HCUP Factbook 2 Procedures,18 billing data from randomly‐selected practicing hospitalists representing the same settings listed above, and critical input from reviewers. Depending on the individual setting, pediatric hospitalists may require skills in areas not found in these 11 chapters, such as chest tube placement or ventilator management. The list is therefore not exhaustive, but rather representative of skills most pediatric hospitalists should maintain.
The Healthcare Systems: Supporting and Advancing Child Healthchapters are likely the most dissimilar to any core content taught in traditional residency programs. While residency graduates are versed in some components listed in these chapters, comprehensive education in most of these competencies is currently lacking. Improvement of healthcare systems is an essential element of pediatric hospital medicine, and unifies all pediatric hospitalists regardless of practice environment or patient population. Therefore, this section includes chapters that not only focus on systems of care, but also on advancing child health through advocacy, research, education, evidence‐based medicine, and ethical practice. These chapters were drawn from a combination of several sources: expectations of external agencies (TJC, Center for Medicaid and Medicare) related to the specific nonclinical work in which pediatric hospitalists are integrally involved; expectations for advocacy as best defined by the AAP and the National Association of Children's Hospitals and Related Institutions (NACHRI); the six core competency domains mandated by the Accrediting Council on Graduate Medical Education (ACGME), the American Board of Pediatrics (ABP), and hospital medical staff offices as part of Focused Professional Practice Evaluation (FPPE) and Ongoing Professional Practice Evaluation (OPPE)16; and assessment of responsibilities and leadership roles fulfilled by pediatric hospitalists in all venues. In keeping with the intent of the competencies to be timeless, the competency elements call out the need to attend to the changing goals of these groups as well as those of the Institute of Healthcare Improvement (IHI), the Alliance for Pediatric Quality (which consists of ABP, AAP, TJC, CHCA, NACHRI), and local hospital systems leaders.
Contributors and Review
The CCTF selected section (associate) editors from SHM based on established expertise in each area, with input from the SHM Pediatric and Education Committees and the SHM Board. As a collaborative effort, authors for various chapters were solicited in consultation with experts from the AAP, APA, and SHM, and included non‐hospitalists with reputations as experts in various fields. Numerous SHM Pediatric Committee and CCTF conference calls were held to review hospital and academic appointments, presentations given, and affiliations relevant to the practice of pediatric hospital medicine. This vetting process resulted in a robust author list representing diverse geographic and practice settings. Contributors were provided with structure (Knowledge, Skills, Attitudes, and Systems subsections) and content (timeless, competency based) guidelines.
The review process was rigorous, and included both internal and external reviewers. The APA review in 2007 included the PHM Special Interest Group as well as the PHM Fellowship Directors (Table 1). After return to SHM and further editing, the internal review commenced which focused on content and scope. The editors addressed the resulting suggestions and worked to standardize formatting and use of Bloom's taxonomy.19 A list of common terms and phrases were created to add consistency between chapters. External reviewers were first mailed a letter requesting interest, which was followed up by emails, letters, and phone calls to encourage feedback. External review included 29 solicited agencies and societies (Table 3), with overall response rate of 66% (41% for Groups I and II). Individual contributors then reviewed comments specific to their chapters, with associate editor overview of their respective sections. The editors reviewed each chapter individually multiple times throughout the 2007‐2009 years, contacting individual contributors and reviewers by email and phone. Editors concluded a final comprehensive review of all chapters in late 2009.
| I. Academic and certifying societies |
| Academic Pediatric Association |
| Accreditation Council for Graduate Medical Education, Pediatric Residency Review Committee |
| American Academy of Family Physicians |
| American Academy of Pediatrics Board |
| American Academy of Pediatrics National Committee on Hospital Care |
| American Association of Critical Care Nursing |
| American Board of Family Medicine |
| American Board of Pediatrics |
| American College of Emergency Physicians |
| American Pediatric Society |
| Association of American Medical Colleges |
| Association of Medical School Pediatric Department Chairs (AMSPDC) |
| Association of Pediatric Program Directors |
| Council on Teaching Hospitals |
| Society of Pediatric Research |
| II. Stakeholder agencies |
| Agency for Healthcare Research and Quality |
| American Association of Critical Care Nursing |
| American College of Emergency Physicians |
| American Hospital Association (AHA) |
| American Nurses Association |
| American Society of Health‐System Pharmacists |
| Child Health Corporation of America (CHCA) |
| Institute for Healthcare Improvement |
| National Association for Children's Hospitals and Related Institutions (NACHRI) |
| National Association of Pediatric Nurse Practitioners (NAPNAP) |
| National Initiative for Children's Healthcare Quality (NICHQ) |
| National Quality Forum |
| Quality Resources International |
| Robert Wood Johnson Foundation |
| The Joint Commission for Accreditation of Hospitals and Organizations (TJC) |
| III. Pediatric hospital medicine fellowship directors |
| Boston Children's |
| Children's Hospital Los Angeles |
| Children's National D.C. |
| Emory |
| Hospital for Sick Kids Toronto |
| Rady Children's San Diego University of California San Diego |
| Riley Children's Hospital Indiana |
| University of South Florida, All Children's Hospital |
| Texas Children's Hospital, Baylor College of Medicine |
| IV. SHM, APA, AAP Leadership and committee chairs |
| American Academy of Pediatrics Section on Hospital Medicine |
| Academic Pediatric Association PHM Special Interest Group |
| SHM Board |
| SHM Education Committee |
| SHM Family Practice Committee |
| SHM Hospital Quality and Patient Safety Committee |
| SHM IT Task Force |
| SHM Journal Editorial Board |
| SHM Palliative Care Task Force |
| SHM Practice Analysis Committee |
| SHM Public Policy Committee |
| SHM Research Committee |
Chapter Content
Each of the 54 chapters within the four sections of these competencies is presented in the educational theory of learning domains: Knowledge, Skills, Attitudes, with a final Systems domain added to reflect the emphasis of hospitalist practice on improving healthcare systems. Each chapter is designed to stand alone, which may assist with development of curriculum at individual practice locations. Certain key phrases are apparent throughout, such as lead, coordinate, or participate in and work with hospital and community leaders to which were designed to note the varied roles in different practice settings. Some chapters specifically comment on the application of competency bullets given the unique and differing roles and expectations of pediatric hospitalists, such as research and education. Chapters state specific proficiencies expected wherever possible, with phrases and wording selected to help guide learning activities to achieve the competency.
Application and Future Directions
Although pediatric hospitalists care for children in many settings, these core competencies address the common expectations for any venue. Pediatric hospital medicine requires skills in acute care clinical medicine that attend to the changing needs of hospitalized children. The core of pediatric hospital medicine is dedicated to the care of children in the geographic hospital environment between emergency medicine and tertiary pediatric and neonatal intensive care units. Pediatric hospitalists provide care in related clinical service programs that are linked to hospital systems. In performing these activities, pediatric hospitalists consistently partner with ambulatory providers and subspecialists to render coordinated care across the continuum for a given child. Pediatric hospital medicine is an interdisciplinary practice, with focus on processes of care and clinical quality outcomes based in evidence. Engagement in local, state, and national initiatives to improve child health outcomes is a cornerstone of pediatric hospitalists' practice. These competencies provide the framework for creation of curricula that can reflect local issues and react to changing evidence.
As providers of systems‐based care, pediatric hospitalists are called upon more and more to render care and provide leadership in clinical arenas that are integral to healthcare organizations, such as home health care, sub‐acute care facilities, and hospice and palliative care programs. The practice of pediatric hospital medicine has evolved to its current state through efforts of many represented in the competencies as contributors, associate editors, editors, and reviewers. Pediatric hospitalists are committed to leading change in healthcare for hospitalized children, and are positioned well to address the interests and needs of community and urban, teaching and non‐teaching facilities, and the children and families they serve. These competencies reflect the areas of focused practice which, similar to pediatric emergency medicine, will no doubt be refined but not fundamentally changed in future years. The intent, we hope, is clear: to provide excellence in clinical care, accountability for practice, and lead improvements in healthcare for hospitalized children.
- Society of Hospital Medicine (SHM). Definition of a Hospitalist. http://www.hospitalmedicine.org/AM/Template.cfm?Section=General_Information 2009.
- .Pediatric Hospitalists Membership Numbers. In.Philadelphia:Society of Hospital Medicine, PA 19130;2009.
- , .The emerging role of “hospitalists” in the American health care system.N Engl J Med.1996;335:514–517.
- .The future of hospital medicine: evolution or revolution?.Am J Med.2004;117:446–450.
- , .The hospitalist movement 5 years later.JAMA.2002;287:487–494.
- , , , , .Variation in pediatric hospitalists' use of proven and unproven therapies: A study from the Pediatric Research in Inpatient Settings (PRIS) network.J Hosp Med.2008;3(4):292–298.
- , , .Pediatric hospitalists: Training, current practice, and career goals.J Hosp Med.2009;4(3):179–186.
- , .Standardize to excellence: improving the quality and safety of care with clinical pathways.Pediatr Clin North Am.2009;56(4):893–904.
- .Evolution of a new specialty ‐ a twenty year pediatric hospitalist experience [Abstract]. In:National Association of Inpatient Physicians (now Society of Hospital Medicine).New Orleans, Louisiana;1999.
- , , , , , , et al.Pediatric hospitalists: report of a leadership conference.Pediatrics.2006;117(4):1122–1130.
- , , , , e.The core competencies in hospital medicine: a framework for curriculum development.J Hosp Med.2006;1(Suppl 1).
- American Board of Internal Medicine. Questions and answers regarding ABIM recognition of focused practice in hospital medicine through maintenance of certification. http://www.abim.org/news/news/focused‐practice‐hospital‐medicine‐qa.aspx. Published 2010. Accessed January 6,2010.
- .Comprehensive pediatric hospital medicine.N Engl J Med.2008;358(21):2301–2302.
- The Joint Commission. Performance measurement initiatives. http://www. jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/. Published 2010. Accessed December 5,2010.
- The Joint Commission. Standards frequently asked questions: comprehensive accreditation manual for critical access hospitals (CAMCAH). http://www.jointcommission.org/AccreditationPrograms/CriticalAccess Hospitals/Standards/09_FAQs/default.htm. Accessed December 5,2008; December 14, 2009.
- , , , , .Infectious disease hospitalizations among infants in the United States.Pediatrics.2008;121(2):244–252.
- , , , .Procedures in U.S. hospitals, 1997.HCUP fact book no. 2. In:agency for healthcare research and quality,Rockville, MD;2001.
- Anderson L, Krathwohl DR, Airasian PW, Cruikshank KA, Mayer RE, Pintrich PR, et al., editors.A taxonomy for learning, teaching, and assessing. In: A Revision of Bloom's Taxonomy of Educational Objectives.Upper Saddle River, NJ: Addison Wesley Longman, Inc. Pearson Education USA;2001.
Introduction
The Society of Hospital Medicine (SHM) defines hospitalists as physicians whose primary professional focus is the comprehensive general medical care of hospitalized patients. Their activities include patient care, teaching, research, and leadership related to Hospital Medicine.1 It is estimated that there are up to 2500 pediatric hospitalists in the United States, with continued growth due to the converging needs for a dedicated focus on patient safety, quality improvement, hospital throughput, and inpatient teaching.2‐9 (Pediatric Hospital Medicine (PHM), as defined today, has been practiced in the United States for at least 30 years10 and continues to evolve as an area of specialization, with the refinement of a distinct knowledgebase and skill set focused on the provision of high quality general pediatric care in the inpatient setting. PHM is the latest site‐specific specialty to emerge from the field of general pediatrics it's development analogous to the evolution of critical care or emergency medicine during previous decades.11 Adult hospital medicine has defined itself within the field of general internal medicine12 and has recently received approval to provide a recognized focus of practice exam in 2010 for those re‐certifying with the American Board of Internal Medicine,13 PHM is creating an identity as a subspecialty practice with distinct focus on inpatient care for children within the larger context of general pediatric care.8, 14
The Pediatric Hospital Medicine Core Competencies were created to help define the roles and expectations for pediatric hospitalists, regardless of practice setting. The intent is to provide a unified approach toward identifying the specific body of knowledge and measurable skills needed to assure delivery of the highest quality of care for all hospitalized pediatric patients. Most children requiring hospitalization in the United States are hospitalized in community settings where subspecialty support is more limited and many pediatric services may be unavailable. Children with complex, chronic medical problems, however, are more likely to be hospitalized at a tertiary care or academic institutions. In order to unify pediatric hospitalists who work in different practice environments, the PHM Core Competencies were constructed to represent the knowledge, skills, attitudes, and systems improvements that all pediatric hospitalists can be expected to acquire and maintain.
Furthermore, the content of the PHM Core Competencies reflect the fact that children are a vulnerable population. Their care requires attention to many elements which distinguishes it from that given to the majority of the adult population: dependency, differences in developmental physiology and behavior, occurrence of congenital genetic disorders and age‐based clinical conditions, impact of chronic disease states on whole child development, and weight‐based medication dosing often with limited guidance from pediatric studies, to name a few. Awareness of these needs must be heightened when a child enters the hospital where diagnoses, procedures, and treatments often include use of high‐risk modalities and require coordination of care across multiple providers.
Pediatric hospitalists commonly work to improve the systems of care in which they operate and therefore both clinical and non‐clinical topics are included. The 54 chapters address the fundamental and most common components of inpatient care but are not an extensive review of all aspects of inpatient medicine encountered by those caring for hospitalized children. Finally, the PHM Core Competencies are not intended for use in assessing proficiency immediately post‐residency, but do provide a framework for the education and evaluation of both physicians‐in‐training and practicing hospitalists. Meeting these competencies is anticipated to take from one to three years of active practice in pediatric hospital medicine, and may be reached through a combination of practice experience, course work, self‐directed work, and/or formalized training.
Methods
Timeline
In 2002, SHM convened an educational summit from which there was a resolution to create core competencies. Following the summit, the SHM Pediatric Core Curriculum Task Force (CCTF) was created, which included 12 pediatric hospitalists practicing in academic and community facilities, as well as teaching and non‐teaching settings, and occupying leadership positions within institutions of varied size and geographic location. Shortly thereafter, in November 2003, approximately 130 pediatric hospitalists attended the first PHM meeting in San Antonio, Texas.11 At this meeting, with support from leaders in pediatric emergency medicine, first discussions regarding PHM scope of practice were held.
Formal development of the competencies began in 2005 in parallel to but distinct from SHM's adult work, which culminated in The Core Competencies in Hospital Medicine: A Framework for Curriculum Development published in 2006. The CCTF divided into three groups, focused on clinical, procedural, and systems‐based topics. Face‐to‐face meetings were held at the SHM annual meetings, with most work being completed by phone and electronically in the interim periods. In 2007, due to the overlapping interests of the three core pediatric societies, the work was transferred to leaders within the APA. In 2008 the work was transferred back to the leadership within SHM. Since that time, external reviewers were solicited, new chapters created, sections re‐aligned, internal and external reviewer comments incorporated, and final edits for taxonomy, content, and formatting were completed (Table 1).
| Date | Event |
|---|---|
| Feb 2002 | SHM Educational Summit held and CCTF created |
| Oct 2003 | 1st PHM meeting held in San Antonio |
| 2003‐2007 | Chapter focus determined; contributors engaged |
| 2007‐2008 | APA PHM Special Interest Group (SIG) review; creation of separate PHM Fellowship Competencies (not in this document) |
| Aug 2008‐Oct 2008 | SHM Pediatric Committee and CCTF members resume work; editorial review |
| Oct 2008‐Mar 2009 | Internal review: PHM Fellowship Director, AAP, APA, and SHM section/committee leader, and key national PHM leader reviews solicited and returned |
| Mar 2009 | PHM Fellowship Director comments addressed; editorial review |
| Mar‐Apr 2009 | External reviewers solicited from national agencies and societies relevant to PHM |
| Apr‐July 2009 | External reviewer comments returned |
| July‐Oct 2009 | Contributor review of all comments; editorial review, sections revised |
| Oct 2009 | Final review: Chapters to SHM subcommittees and Board |
Areas of Focused Practice
The PHM Core Competencies were conceptualized similarly to the SHM adult core competencies. Initial sections were divided into clinical conditions, procedures, and systems. However as content developed and reviewer comments were addressed, the four final sections were modified to those noted in Table 2. For the Common Clinical Diagnoses and Conditions, the goal was to select conditions most commonly encountered by pediatric hospitalists. Non‐surgical diagnosis‐related group (DRG) conditions were selected from the following sources: The Joint Commission's (TJC) Oryx Performance Measures Report15‐16 (asthma, abdominal pain, acute gastroenteritis, simple pneumonia); Child Health Corporation of America's Pediatric Health Information System Dataset (CHCA PHIS, Shawnee Mission, KS), and relevant publications on common pediatric hospitalizations.17 These data were compared to billing data from randomly‐selected practicing hospitalists representing free‐standing children's and community hospitals, teaching and non‐teaching settings, and urban and rural locations. The 22 clinical conditions chosen by the CCTF were those most relevant to the practice of pediatric hospital medicine.
| Common Clinical Diagnoses and Conditions | Specialized Clinical Services | Core Skills | Healthcare Systems: Supporting and Advancing Child Health | |
|---|---|---|---|---|
| Acute abdominal pain and the acute abdomen | Neonatal fever | Child abuse and neglect | Bladder catheterization/suprapubic bladder tap | Advocacy |
| Apparent life‐threatening event | Neonatal jaundice | Hospice and palliative care | Electrocardiogram interpretation | Business practices |
| Asthma | Pneumonia | Leading a healthcare team | Feeding tubes | Communication |
| Bone and joint infections | Respiratory failure | Newborn care and delivery room management | Fluids and electrolyte management | Continuous quality improvement |
| Bronchiolitis | Seizures | Technology‐dependent children | Intravenous access and phlebotomy | Cost‐effective care |
| Central nervous system infections | Shock | Transport of the critically ill child | Lumbar puncture | Education |
| Diabetes mellitus | Sickle cell disease | Non‐invasive monitoring | Ethics | |
| Failure to thrive | Skin and soft tissue infection | Nutrition | Evidence‐based medicine | |
| Fever of unknown origin | Toxic ingestion | Oxygen delivery and airway management | Health information systems | |
| Gastroenteritis | Upper airway infections | Pain management | Legal issues/risk management | |
| Kawasaki disease | Urinary tract infections | Pediatric advanced life support | Patient safety |
The Specialized Clinical Servicessection addresses important components of care that are not DRG‐based and reflect the unique needs of hospitalized children, as assessed by the CCTF, editors, and contributors. Core Skillswere chosen based on the HCUP Factbook 2 Procedures,18 billing data from randomly‐selected practicing hospitalists representing the same settings listed above, and critical input from reviewers. Depending on the individual setting, pediatric hospitalists may require skills in areas not found in these 11 chapters, such as chest tube placement or ventilator management. The list is therefore not exhaustive, but rather representative of skills most pediatric hospitalists should maintain.
The Healthcare Systems: Supporting and Advancing Child Healthchapters are likely the most dissimilar to any core content taught in traditional residency programs. While residency graduates are versed in some components listed in these chapters, comprehensive education in most of these competencies is currently lacking. Improvement of healthcare systems is an essential element of pediatric hospital medicine, and unifies all pediatric hospitalists regardless of practice environment or patient population. Therefore, this section includes chapters that not only focus on systems of care, but also on advancing child health through advocacy, research, education, evidence‐based medicine, and ethical practice. These chapters were drawn from a combination of several sources: expectations of external agencies (TJC, Center for Medicaid and Medicare) related to the specific nonclinical work in which pediatric hospitalists are integrally involved; expectations for advocacy as best defined by the AAP and the National Association of Children's Hospitals and Related Institutions (NACHRI); the six core competency domains mandated by the Accrediting Council on Graduate Medical Education (ACGME), the American Board of Pediatrics (ABP), and hospital medical staff offices as part of Focused Professional Practice Evaluation (FPPE) and Ongoing Professional Practice Evaluation (OPPE)16; and assessment of responsibilities and leadership roles fulfilled by pediatric hospitalists in all venues. In keeping with the intent of the competencies to be timeless, the competency elements call out the need to attend to the changing goals of these groups as well as those of the Institute of Healthcare Improvement (IHI), the Alliance for Pediatric Quality (which consists of ABP, AAP, TJC, CHCA, NACHRI), and local hospital systems leaders.
Contributors and Review
The CCTF selected section (associate) editors from SHM based on established expertise in each area, with input from the SHM Pediatric and Education Committees and the SHM Board. As a collaborative effort, authors for various chapters were solicited in consultation with experts from the AAP, APA, and SHM, and included non‐hospitalists with reputations as experts in various fields. Numerous SHM Pediatric Committee and CCTF conference calls were held to review hospital and academic appointments, presentations given, and affiliations relevant to the practice of pediatric hospital medicine. This vetting process resulted in a robust author list representing diverse geographic and practice settings. Contributors were provided with structure (Knowledge, Skills, Attitudes, and Systems subsections) and content (timeless, competency based) guidelines.
The review process was rigorous, and included both internal and external reviewers. The APA review in 2007 included the PHM Special Interest Group as well as the PHM Fellowship Directors (Table 1). After return to SHM and further editing, the internal review commenced which focused on content and scope. The editors addressed the resulting suggestions and worked to standardize formatting and use of Bloom's taxonomy.19 A list of common terms and phrases were created to add consistency between chapters. External reviewers were first mailed a letter requesting interest, which was followed up by emails, letters, and phone calls to encourage feedback. External review included 29 solicited agencies and societies (Table 3), with overall response rate of 66% (41% for Groups I and II). Individual contributors then reviewed comments specific to their chapters, with associate editor overview of their respective sections. The editors reviewed each chapter individually multiple times throughout the 2007‐2009 years, contacting individual contributors and reviewers by email and phone. Editors concluded a final comprehensive review of all chapters in late 2009.
| I. Academic and certifying societies |
| Academic Pediatric Association |
| Accreditation Council for Graduate Medical Education, Pediatric Residency Review Committee |
| American Academy of Family Physicians |
| American Academy of Pediatrics Board |
| American Academy of Pediatrics National Committee on Hospital Care |
| American Association of Critical Care Nursing |
| American Board of Family Medicine |
| American Board of Pediatrics |
| American College of Emergency Physicians |
| American Pediatric Society |
| Association of American Medical Colleges |
| Association of Medical School Pediatric Department Chairs (AMSPDC) |
| Association of Pediatric Program Directors |
| Council on Teaching Hospitals |
| Society of Pediatric Research |
| II. Stakeholder agencies |
| Agency for Healthcare Research and Quality |
| American Association of Critical Care Nursing |
| American College of Emergency Physicians |
| American Hospital Association (AHA) |
| American Nurses Association |
| American Society of Health‐System Pharmacists |
| Child Health Corporation of America (CHCA) |
| Institute for Healthcare Improvement |
| National Association for Children's Hospitals and Related Institutions (NACHRI) |
| National Association of Pediatric Nurse Practitioners (NAPNAP) |
| National Initiative for Children's Healthcare Quality (NICHQ) |
| National Quality Forum |
| Quality Resources International |
| Robert Wood Johnson Foundation |
| The Joint Commission for Accreditation of Hospitals and Organizations (TJC) |
| III. Pediatric hospital medicine fellowship directors |
| Boston Children's |
| Children's Hospital Los Angeles |
| Children's National D.C. |
| Emory |
| Hospital for Sick Kids Toronto |
| Rady Children's San Diego University of California San Diego |
| Riley Children's Hospital Indiana |
| University of South Florida, All Children's Hospital |
| Texas Children's Hospital, Baylor College of Medicine |
| IV. SHM, APA, AAP Leadership and committee chairs |
| American Academy of Pediatrics Section on Hospital Medicine |
| Academic Pediatric Association PHM Special Interest Group |
| SHM Board |
| SHM Education Committee |
| SHM Family Practice Committee |
| SHM Hospital Quality and Patient Safety Committee |
| SHM IT Task Force |
| SHM Journal Editorial Board |
| SHM Palliative Care Task Force |
| SHM Practice Analysis Committee |
| SHM Public Policy Committee |
| SHM Research Committee |
Chapter Content
Each of the 54 chapters within the four sections of these competencies is presented in the educational theory of learning domains: Knowledge, Skills, Attitudes, with a final Systems domain added to reflect the emphasis of hospitalist practice on improving healthcare systems. Each chapter is designed to stand alone, which may assist with development of curriculum at individual practice locations. Certain key phrases are apparent throughout, such as lead, coordinate, or participate in and work with hospital and community leaders to which were designed to note the varied roles in different practice settings. Some chapters specifically comment on the application of competency bullets given the unique and differing roles and expectations of pediatric hospitalists, such as research and education. Chapters state specific proficiencies expected wherever possible, with phrases and wording selected to help guide learning activities to achieve the competency.
Application and Future Directions
Although pediatric hospitalists care for children in many settings, these core competencies address the common expectations for any venue. Pediatric hospital medicine requires skills in acute care clinical medicine that attend to the changing needs of hospitalized children. The core of pediatric hospital medicine is dedicated to the care of children in the geographic hospital environment between emergency medicine and tertiary pediatric and neonatal intensive care units. Pediatric hospitalists provide care in related clinical service programs that are linked to hospital systems. In performing these activities, pediatric hospitalists consistently partner with ambulatory providers and subspecialists to render coordinated care across the continuum for a given child. Pediatric hospital medicine is an interdisciplinary practice, with focus on processes of care and clinical quality outcomes based in evidence. Engagement in local, state, and national initiatives to improve child health outcomes is a cornerstone of pediatric hospitalists' practice. These competencies provide the framework for creation of curricula that can reflect local issues and react to changing evidence.
As providers of systems‐based care, pediatric hospitalists are called upon more and more to render care and provide leadership in clinical arenas that are integral to healthcare organizations, such as home health care, sub‐acute care facilities, and hospice and palliative care programs. The practice of pediatric hospital medicine has evolved to its current state through efforts of many represented in the competencies as contributors, associate editors, editors, and reviewers. Pediatric hospitalists are committed to leading change in healthcare for hospitalized children, and are positioned well to address the interests and needs of community and urban, teaching and non‐teaching facilities, and the children and families they serve. These competencies reflect the areas of focused practice which, similar to pediatric emergency medicine, will no doubt be refined but not fundamentally changed in future years. The intent, we hope, is clear: to provide excellence in clinical care, accountability for practice, and lead improvements in healthcare for hospitalized children.
Introduction
The Society of Hospital Medicine (SHM) defines hospitalists as physicians whose primary professional focus is the comprehensive general medical care of hospitalized patients. Their activities include patient care, teaching, research, and leadership related to Hospital Medicine.1 It is estimated that there are up to 2500 pediatric hospitalists in the United States, with continued growth due to the converging needs for a dedicated focus on patient safety, quality improvement, hospital throughput, and inpatient teaching.2‐9 (Pediatric Hospital Medicine (PHM), as defined today, has been practiced in the United States for at least 30 years10 and continues to evolve as an area of specialization, with the refinement of a distinct knowledgebase and skill set focused on the provision of high quality general pediatric care in the inpatient setting. PHM is the latest site‐specific specialty to emerge from the field of general pediatrics it's development analogous to the evolution of critical care or emergency medicine during previous decades.11 Adult hospital medicine has defined itself within the field of general internal medicine12 and has recently received approval to provide a recognized focus of practice exam in 2010 for those re‐certifying with the American Board of Internal Medicine,13 PHM is creating an identity as a subspecialty practice with distinct focus on inpatient care for children within the larger context of general pediatric care.8, 14
The Pediatric Hospital Medicine Core Competencies were created to help define the roles and expectations for pediatric hospitalists, regardless of practice setting. The intent is to provide a unified approach toward identifying the specific body of knowledge and measurable skills needed to assure delivery of the highest quality of care for all hospitalized pediatric patients. Most children requiring hospitalization in the United States are hospitalized in community settings where subspecialty support is more limited and many pediatric services may be unavailable. Children with complex, chronic medical problems, however, are more likely to be hospitalized at a tertiary care or academic institutions. In order to unify pediatric hospitalists who work in different practice environments, the PHM Core Competencies were constructed to represent the knowledge, skills, attitudes, and systems improvements that all pediatric hospitalists can be expected to acquire and maintain.
Furthermore, the content of the PHM Core Competencies reflect the fact that children are a vulnerable population. Their care requires attention to many elements which distinguishes it from that given to the majority of the adult population: dependency, differences in developmental physiology and behavior, occurrence of congenital genetic disorders and age‐based clinical conditions, impact of chronic disease states on whole child development, and weight‐based medication dosing often with limited guidance from pediatric studies, to name a few. Awareness of these needs must be heightened when a child enters the hospital where diagnoses, procedures, and treatments often include use of high‐risk modalities and require coordination of care across multiple providers.
Pediatric hospitalists commonly work to improve the systems of care in which they operate and therefore both clinical and non‐clinical topics are included. The 54 chapters address the fundamental and most common components of inpatient care but are not an extensive review of all aspects of inpatient medicine encountered by those caring for hospitalized children. Finally, the PHM Core Competencies are not intended for use in assessing proficiency immediately post‐residency, but do provide a framework for the education and evaluation of both physicians‐in‐training and practicing hospitalists. Meeting these competencies is anticipated to take from one to three years of active practice in pediatric hospital medicine, and may be reached through a combination of practice experience, course work, self‐directed work, and/or formalized training.
Methods
Timeline
In 2002, SHM convened an educational summit from which there was a resolution to create core competencies. Following the summit, the SHM Pediatric Core Curriculum Task Force (CCTF) was created, which included 12 pediatric hospitalists practicing in academic and community facilities, as well as teaching and non‐teaching settings, and occupying leadership positions within institutions of varied size and geographic location. Shortly thereafter, in November 2003, approximately 130 pediatric hospitalists attended the first PHM meeting in San Antonio, Texas.11 At this meeting, with support from leaders in pediatric emergency medicine, first discussions regarding PHM scope of practice were held.
Formal development of the competencies began in 2005 in parallel to but distinct from SHM's adult work, which culminated in The Core Competencies in Hospital Medicine: A Framework for Curriculum Development published in 2006. The CCTF divided into three groups, focused on clinical, procedural, and systems‐based topics. Face‐to‐face meetings were held at the SHM annual meetings, with most work being completed by phone and electronically in the interim periods. In 2007, due to the overlapping interests of the three core pediatric societies, the work was transferred to leaders within the APA. In 2008 the work was transferred back to the leadership within SHM. Since that time, external reviewers were solicited, new chapters created, sections re‐aligned, internal and external reviewer comments incorporated, and final edits for taxonomy, content, and formatting were completed (Table 1).
| Date | Event |
|---|---|
| Feb 2002 | SHM Educational Summit held and CCTF created |
| Oct 2003 | 1st PHM meeting held in San Antonio |
| 2003‐2007 | Chapter focus determined; contributors engaged |
| 2007‐2008 | APA PHM Special Interest Group (SIG) review; creation of separate PHM Fellowship Competencies (not in this document) |
| Aug 2008‐Oct 2008 | SHM Pediatric Committee and CCTF members resume work; editorial review |
| Oct 2008‐Mar 2009 | Internal review: PHM Fellowship Director, AAP, APA, and SHM section/committee leader, and key national PHM leader reviews solicited and returned |
| Mar 2009 | PHM Fellowship Director comments addressed; editorial review |
| Mar‐Apr 2009 | External reviewers solicited from national agencies and societies relevant to PHM |
| Apr‐July 2009 | External reviewer comments returned |
| July‐Oct 2009 | Contributor review of all comments; editorial review, sections revised |
| Oct 2009 | Final review: Chapters to SHM subcommittees and Board |
Areas of Focused Practice
The PHM Core Competencies were conceptualized similarly to the SHM adult core competencies. Initial sections were divided into clinical conditions, procedures, and systems. However as content developed and reviewer comments were addressed, the four final sections were modified to those noted in Table 2. For the Common Clinical Diagnoses and Conditions, the goal was to select conditions most commonly encountered by pediatric hospitalists. Non‐surgical diagnosis‐related group (DRG) conditions were selected from the following sources: The Joint Commission's (TJC) Oryx Performance Measures Report15‐16 (asthma, abdominal pain, acute gastroenteritis, simple pneumonia); Child Health Corporation of America's Pediatric Health Information System Dataset (CHCA PHIS, Shawnee Mission, KS), and relevant publications on common pediatric hospitalizations.17 These data were compared to billing data from randomly‐selected practicing hospitalists representing free‐standing children's and community hospitals, teaching and non‐teaching settings, and urban and rural locations. The 22 clinical conditions chosen by the CCTF were those most relevant to the practice of pediatric hospital medicine.
| Common Clinical Diagnoses and Conditions | Specialized Clinical Services | Core Skills | Healthcare Systems: Supporting and Advancing Child Health | |
|---|---|---|---|---|
| Acute abdominal pain and the acute abdomen | Neonatal fever | Child abuse and neglect | Bladder catheterization/suprapubic bladder tap | Advocacy |
| Apparent life‐threatening event | Neonatal jaundice | Hospice and palliative care | Electrocardiogram interpretation | Business practices |
| Asthma | Pneumonia | Leading a healthcare team | Feeding tubes | Communication |
| Bone and joint infections | Respiratory failure | Newborn care and delivery room management | Fluids and electrolyte management | Continuous quality improvement |
| Bronchiolitis | Seizures | Technology‐dependent children | Intravenous access and phlebotomy | Cost‐effective care |
| Central nervous system infections | Shock | Transport of the critically ill child | Lumbar puncture | Education |
| Diabetes mellitus | Sickle cell disease | Non‐invasive monitoring | Ethics | |
| Failure to thrive | Skin and soft tissue infection | Nutrition | Evidence‐based medicine | |
| Fever of unknown origin | Toxic ingestion | Oxygen delivery and airway management | Health information systems | |
| Gastroenteritis | Upper airway infections | Pain management | Legal issues/risk management | |
| Kawasaki disease | Urinary tract infections | Pediatric advanced life support | Patient safety |
The Specialized Clinical Servicessection addresses important components of care that are not DRG‐based and reflect the unique needs of hospitalized children, as assessed by the CCTF, editors, and contributors. Core Skillswere chosen based on the HCUP Factbook 2 Procedures,18 billing data from randomly‐selected practicing hospitalists representing the same settings listed above, and critical input from reviewers. Depending on the individual setting, pediatric hospitalists may require skills in areas not found in these 11 chapters, such as chest tube placement or ventilator management. The list is therefore not exhaustive, but rather representative of skills most pediatric hospitalists should maintain.
The Healthcare Systems: Supporting and Advancing Child Healthchapters are likely the most dissimilar to any core content taught in traditional residency programs. While residency graduates are versed in some components listed in these chapters, comprehensive education in most of these competencies is currently lacking. Improvement of healthcare systems is an essential element of pediatric hospital medicine, and unifies all pediatric hospitalists regardless of practice environment or patient population. Therefore, this section includes chapters that not only focus on systems of care, but also on advancing child health through advocacy, research, education, evidence‐based medicine, and ethical practice. These chapters were drawn from a combination of several sources: expectations of external agencies (TJC, Center for Medicaid and Medicare) related to the specific nonclinical work in which pediatric hospitalists are integrally involved; expectations for advocacy as best defined by the AAP and the National Association of Children's Hospitals and Related Institutions (NACHRI); the six core competency domains mandated by the Accrediting Council on Graduate Medical Education (ACGME), the American Board of Pediatrics (ABP), and hospital medical staff offices as part of Focused Professional Practice Evaluation (FPPE) and Ongoing Professional Practice Evaluation (OPPE)16; and assessment of responsibilities and leadership roles fulfilled by pediatric hospitalists in all venues. In keeping with the intent of the competencies to be timeless, the competency elements call out the need to attend to the changing goals of these groups as well as those of the Institute of Healthcare Improvement (IHI), the Alliance for Pediatric Quality (which consists of ABP, AAP, TJC, CHCA, NACHRI), and local hospital systems leaders.
Contributors and Review
The CCTF selected section (associate) editors from SHM based on established expertise in each area, with input from the SHM Pediatric and Education Committees and the SHM Board. As a collaborative effort, authors for various chapters were solicited in consultation with experts from the AAP, APA, and SHM, and included non‐hospitalists with reputations as experts in various fields. Numerous SHM Pediatric Committee and CCTF conference calls were held to review hospital and academic appointments, presentations given, and affiliations relevant to the practice of pediatric hospital medicine. This vetting process resulted in a robust author list representing diverse geographic and practice settings. Contributors were provided with structure (Knowledge, Skills, Attitudes, and Systems subsections) and content (timeless, competency based) guidelines.
The review process was rigorous, and included both internal and external reviewers. The APA review in 2007 included the PHM Special Interest Group as well as the PHM Fellowship Directors (Table 1). After return to SHM and further editing, the internal review commenced which focused on content and scope. The editors addressed the resulting suggestions and worked to standardize formatting and use of Bloom's taxonomy.19 A list of common terms and phrases were created to add consistency between chapters. External reviewers were first mailed a letter requesting interest, which was followed up by emails, letters, and phone calls to encourage feedback. External review included 29 solicited agencies and societies (Table 3), with overall response rate of 66% (41% for Groups I and II). Individual contributors then reviewed comments specific to their chapters, with associate editor overview of their respective sections. The editors reviewed each chapter individually multiple times throughout the 2007‐2009 years, contacting individual contributors and reviewers by email and phone. Editors concluded a final comprehensive review of all chapters in late 2009.
| I. Academic and certifying societies |
| Academic Pediatric Association |
| Accreditation Council for Graduate Medical Education, Pediatric Residency Review Committee |
| American Academy of Family Physicians |
| American Academy of Pediatrics Board |
| American Academy of Pediatrics National Committee on Hospital Care |
| American Association of Critical Care Nursing |
| American Board of Family Medicine |
| American Board of Pediatrics |
| American College of Emergency Physicians |
| American Pediatric Society |
| Association of American Medical Colleges |
| Association of Medical School Pediatric Department Chairs (AMSPDC) |
| Association of Pediatric Program Directors |
| Council on Teaching Hospitals |
| Society of Pediatric Research |
| II. Stakeholder agencies |
| Agency for Healthcare Research and Quality |
| American Association of Critical Care Nursing |
| American College of Emergency Physicians |
| American Hospital Association (AHA) |
| American Nurses Association |
| American Society of Health‐System Pharmacists |
| Child Health Corporation of America (CHCA) |
| Institute for Healthcare Improvement |
| National Association for Children's Hospitals and Related Institutions (NACHRI) |
| National Association of Pediatric Nurse Practitioners (NAPNAP) |
| National Initiative for Children's Healthcare Quality (NICHQ) |
| National Quality Forum |
| Quality Resources International |
| Robert Wood Johnson Foundation |
| The Joint Commission for Accreditation of Hospitals and Organizations (TJC) |
| III. Pediatric hospital medicine fellowship directors |
| Boston Children's |
| Children's Hospital Los Angeles |
| Children's National D.C. |
| Emory |
| Hospital for Sick Kids Toronto |
| Rady Children's San Diego University of California San Diego |
| Riley Children's Hospital Indiana |
| University of South Florida, All Children's Hospital |
| Texas Children's Hospital, Baylor College of Medicine |
| IV. SHM, APA, AAP Leadership and committee chairs |
| American Academy of Pediatrics Section on Hospital Medicine |
| Academic Pediatric Association PHM Special Interest Group |
| SHM Board |
| SHM Education Committee |
| SHM Family Practice Committee |
| SHM Hospital Quality and Patient Safety Committee |
| SHM IT Task Force |
| SHM Journal Editorial Board |
| SHM Palliative Care Task Force |
| SHM Practice Analysis Committee |
| SHM Public Policy Committee |
| SHM Research Committee |
Chapter Content
Each of the 54 chapters within the four sections of these competencies is presented in the educational theory of learning domains: Knowledge, Skills, Attitudes, with a final Systems domain added to reflect the emphasis of hospitalist practice on improving healthcare systems. Each chapter is designed to stand alone, which may assist with development of curriculum at individual practice locations. Certain key phrases are apparent throughout, such as lead, coordinate, or participate in and work with hospital and community leaders to which were designed to note the varied roles in different practice settings. Some chapters specifically comment on the application of competency bullets given the unique and differing roles and expectations of pediatric hospitalists, such as research and education. Chapters state specific proficiencies expected wherever possible, with phrases and wording selected to help guide learning activities to achieve the competency.
Application and Future Directions
Although pediatric hospitalists care for children in many settings, these core competencies address the common expectations for any venue. Pediatric hospital medicine requires skills in acute care clinical medicine that attend to the changing needs of hospitalized children. The core of pediatric hospital medicine is dedicated to the care of children in the geographic hospital environment between emergency medicine and tertiary pediatric and neonatal intensive care units. Pediatric hospitalists provide care in related clinical service programs that are linked to hospital systems. In performing these activities, pediatric hospitalists consistently partner with ambulatory providers and subspecialists to render coordinated care across the continuum for a given child. Pediatric hospital medicine is an interdisciplinary practice, with focus on processes of care and clinical quality outcomes based in evidence. Engagement in local, state, and national initiatives to improve child health outcomes is a cornerstone of pediatric hospitalists' practice. These competencies provide the framework for creation of curricula that can reflect local issues and react to changing evidence.
As providers of systems‐based care, pediatric hospitalists are called upon more and more to render care and provide leadership in clinical arenas that are integral to healthcare organizations, such as home health care, sub‐acute care facilities, and hospice and palliative care programs. The practice of pediatric hospital medicine has evolved to its current state through efforts of many represented in the competencies as contributors, associate editors, editors, and reviewers. Pediatric hospitalists are committed to leading change in healthcare for hospitalized children, and are positioned well to address the interests and needs of community and urban, teaching and non‐teaching facilities, and the children and families they serve. These competencies reflect the areas of focused practice which, similar to pediatric emergency medicine, will no doubt be refined but not fundamentally changed in future years. The intent, we hope, is clear: to provide excellence in clinical care, accountability for practice, and lead improvements in healthcare for hospitalized children.
- Society of Hospital Medicine (SHM). Definition of a Hospitalist. http://www.hospitalmedicine.org/AM/Template.cfm?Section=General_Information 2009.
- .Pediatric Hospitalists Membership Numbers. In.Philadelphia:Society of Hospital Medicine, PA 19130;2009.
- , .The emerging role of “hospitalists” in the American health care system.N Engl J Med.1996;335:514–517.
- .The future of hospital medicine: evolution or revolution?.Am J Med.2004;117:446–450.
- , .The hospitalist movement 5 years later.JAMA.2002;287:487–494.
- , , , , .Variation in pediatric hospitalists' use of proven and unproven therapies: A study from the Pediatric Research in Inpatient Settings (PRIS) network.J Hosp Med.2008;3(4):292–298.
- , , .Pediatric hospitalists: Training, current practice, and career goals.J Hosp Med.2009;4(3):179–186.
- , .Standardize to excellence: improving the quality and safety of care with clinical pathways.Pediatr Clin North Am.2009;56(4):893–904.
- .Evolution of a new specialty ‐ a twenty year pediatric hospitalist experience [Abstract]. In:National Association of Inpatient Physicians (now Society of Hospital Medicine).New Orleans, Louisiana;1999.
- , , , , , , et al.Pediatric hospitalists: report of a leadership conference.Pediatrics.2006;117(4):1122–1130.
- , , , , e.The core competencies in hospital medicine: a framework for curriculum development.J Hosp Med.2006;1(Suppl 1).
- American Board of Internal Medicine. Questions and answers regarding ABIM recognition of focused practice in hospital medicine through maintenance of certification. http://www.abim.org/news/news/focused‐practice‐hospital‐medicine‐qa.aspx. Published 2010. Accessed January 6,2010.
- .Comprehensive pediatric hospital medicine.N Engl J Med.2008;358(21):2301–2302.
- The Joint Commission. Performance measurement initiatives. http://www. jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/. Published 2010. Accessed December 5,2010.
- The Joint Commission. Standards frequently asked questions: comprehensive accreditation manual for critical access hospitals (CAMCAH). http://www.jointcommission.org/AccreditationPrograms/CriticalAccess Hospitals/Standards/09_FAQs/default.htm. Accessed December 5,2008; December 14, 2009.
- , , , , .Infectious disease hospitalizations among infants in the United States.Pediatrics.2008;121(2):244–252.
- , , , .Procedures in U.S. hospitals, 1997.HCUP fact book no. 2. In:agency for healthcare research and quality,Rockville, MD;2001.
- Anderson L, Krathwohl DR, Airasian PW, Cruikshank KA, Mayer RE, Pintrich PR, et al., editors.A taxonomy for learning, teaching, and assessing. In: A Revision of Bloom's Taxonomy of Educational Objectives.Upper Saddle River, NJ: Addison Wesley Longman, Inc. Pearson Education USA;2001.
- Society of Hospital Medicine (SHM). Definition of a Hospitalist. http://www.hospitalmedicine.org/AM/Template.cfm?Section=General_Information 2009.
- .Pediatric Hospitalists Membership Numbers. In.Philadelphia:Society of Hospital Medicine, PA 19130;2009.
- , .The emerging role of “hospitalists” in the American health care system.N Engl J Med.1996;335:514–517.
- .The future of hospital medicine: evolution or revolution?.Am J Med.2004;117:446–450.
- , .The hospitalist movement 5 years later.JAMA.2002;287:487–494.
- , , , , .Variation in pediatric hospitalists' use of proven and unproven therapies: A study from the Pediatric Research in Inpatient Settings (PRIS) network.J Hosp Med.2008;3(4):292–298.
- , , .Pediatric hospitalists: Training, current practice, and career goals.J Hosp Med.2009;4(3):179–186.
- , .Standardize to excellence: improving the quality and safety of care with clinical pathways.Pediatr Clin North Am.2009;56(4):893–904.
- .Evolution of a new specialty ‐ a twenty year pediatric hospitalist experience [Abstract]. In:National Association of Inpatient Physicians (now Society of Hospital Medicine).New Orleans, Louisiana;1999.
- , , , , , , et al.Pediatric hospitalists: report of a leadership conference.Pediatrics.2006;117(4):1122–1130.
- , , , , e.The core competencies in hospital medicine: a framework for curriculum development.J Hosp Med.2006;1(Suppl 1).
- American Board of Internal Medicine. Questions and answers regarding ABIM recognition of focused practice in hospital medicine through maintenance of certification. http://www.abim.org/news/news/focused‐practice‐hospital‐medicine‐qa.aspx. Published 2010. Accessed January 6,2010.
- .Comprehensive pediatric hospital medicine.N Engl J Med.2008;358(21):2301–2302.
- The Joint Commission. Performance measurement initiatives. http://www. jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/. Published 2010. Accessed December 5,2010.
- The Joint Commission. Standards frequently asked questions: comprehensive accreditation manual for critical access hospitals (CAMCAH). http://www.jointcommission.org/AccreditationPrograms/CriticalAccess Hospitals/Standards/09_FAQs/default.htm. Accessed December 5,2008; December 14, 2009.
- , , , , .Infectious disease hospitalizations among infants in the United States.Pediatrics.2008;121(2):244–252.
- , , , .Procedures in U.S. hospitals, 1997.HCUP fact book no. 2. In:agency for healthcare research and quality,Rockville, MD;2001.
- Anderson L, Krathwohl DR, Airasian PW, Cruikshank KA, Mayer RE, Pintrich PR, et al., editors.A taxonomy for learning, teaching, and assessing. In: A Revision of Bloom's Taxonomy of Educational Objectives.Upper Saddle River, NJ: Addison Wesley Longman, Inc. Pearson Education USA;2001.
Copyright © 2010 Society of Hospital Medicine
Electronic Health Records Get the Green Light
Now that the Centers for Medicare and Medicaid Services (CMS) has defined "meaningful use" of electronic health records (EHR), hospitalists know what's expected of them in order to grab a piece of the $20 billion set aside for doctors and hospitals that adopt new technologies.
CMS’ final rule (PDF) is less restrictive than the proposed rule put forth in January, but it still challenges HM groups and their respective institutions to meet new guidelines to make digital record-keeping routine. Stage-one rules, which take effect next year, require eligible physicians (EPs) and eligible hospitals to meet goals in 15 and 14 categories, respectively. Up to five goals can be deferred, according to CMS. The CMS timeline includes second and third stages, each of which will require goals that are even more advanced.
Some hospitalists feared the rules in stage one would be punitively strict, says Robert Lineberger MD FHM, medical information officer at Durham (N.C.) Regional Hospital, part of the Duke University Health System. “What it means is the government is serious about helping people instead of being as strict as it appeared they were going to be,” says Dr. Lineberger, who serves on SHM's IT Core Committee. “I think people are overall pretty pleased there was a relaxation.”
The road to full adoption of EHR is far from complete, and hospitals that have yet to put in place even the most basic electronic infrastructure might struggle to meet even the lowest thresholds.
And while specific standards for future stages have not been codified, “like everything else that goes on in the hospital, [HM] should be in the middle of that,” Dr. Lineberger says.
Now that the Centers for Medicare and Medicaid Services (CMS) has defined "meaningful use" of electronic health records (EHR), hospitalists know what's expected of them in order to grab a piece of the $20 billion set aside for doctors and hospitals that adopt new technologies.
CMS’ final rule (PDF) is less restrictive than the proposed rule put forth in January, but it still challenges HM groups and their respective institutions to meet new guidelines to make digital record-keeping routine. Stage-one rules, which take effect next year, require eligible physicians (EPs) and eligible hospitals to meet goals in 15 and 14 categories, respectively. Up to five goals can be deferred, according to CMS. The CMS timeline includes second and third stages, each of which will require goals that are even more advanced.
Some hospitalists feared the rules in stage one would be punitively strict, says Robert Lineberger MD FHM, medical information officer at Durham (N.C.) Regional Hospital, part of the Duke University Health System. “What it means is the government is serious about helping people instead of being as strict as it appeared they were going to be,” says Dr. Lineberger, who serves on SHM's IT Core Committee. “I think people are overall pretty pleased there was a relaxation.”
The road to full adoption of EHR is far from complete, and hospitals that have yet to put in place even the most basic electronic infrastructure might struggle to meet even the lowest thresholds.
And while specific standards for future stages have not been codified, “like everything else that goes on in the hospital, [HM] should be in the middle of that,” Dr. Lineberger says.
Now that the Centers for Medicare and Medicaid Services (CMS) has defined "meaningful use" of electronic health records (EHR), hospitalists know what's expected of them in order to grab a piece of the $20 billion set aside for doctors and hospitals that adopt new technologies.
CMS’ final rule (PDF) is less restrictive than the proposed rule put forth in January, but it still challenges HM groups and their respective institutions to meet new guidelines to make digital record-keeping routine. Stage-one rules, which take effect next year, require eligible physicians (EPs) and eligible hospitals to meet goals in 15 and 14 categories, respectively. Up to five goals can be deferred, according to CMS. The CMS timeline includes second and third stages, each of which will require goals that are even more advanced.
Some hospitalists feared the rules in stage one would be punitively strict, says Robert Lineberger MD FHM, medical information officer at Durham (N.C.) Regional Hospital, part of the Duke University Health System. “What it means is the government is serious about helping people instead of being as strict as it appeared they were going to be,” says Dr. Lineberger, who serves on SHM's IT Core Committee. “I think people are overall pretty pleased there was a relaxation.”
The road to full adoption of EHR is far from complete, and hospitals that have yet to put in place even the most basic electronic infrastructure might struggle to meet even the lowest thresholds.
And while specific standards for future stages have not been codified, “like everything else that goes on in the hospital, [HM] should be in the middle of that,” Dr. Lineberger says.
New Study Rebuffs Physician Training Misperceptions
A new study comparing physicians who were trained in the U.S. with those trained in medical schools abroad should offer reassurance to patients, families, and professional peers who hold biases against foreign-born or foreign-trained physicians, the lead author says.
John Norcini, PhD, CEO of the Foundation for Advancement of International Medical Education and Research, and colleagues studied 244,000 hospitalized Pennsylvania patients with congestive heart failure and acute myocardial infarction. They found that mortality rates were slightly lower for physicians who were trained abroad and were not U.S. citizens when they entered medical school. The study showed higher rates for U.S. citizens who went overseas for their medical training.
The Norcini study (Health Affairs. 2010;29:1461-1468) focused on family medicine, internal medicine, and cardiology physicians, but it did not identify hospitalists. One-quarter of all physicians practicing in the U.S. are foreign-trained; however, a greater proportion are found in primary care and internal medicine. For hospitalists, the foreign-trained percentage might be even higher, 40% according to Philip Miller of the physician recruiting firm Merritt Hawkins.
One thing that can be said about international medical graduates is that they are a “crucial and growing part of the hospital medicine workforce, and we welcome them,” says Winthrop Whitcomb, MD, MHM, former SHM president and medical director of healthcare quality at Baystate Medical Center in Springfield, Mass. “I find, having worked with physicians trained all over the world, that for the best ones, it’s what they do every day, not where they came from. Are they consistent, careful, compassionate and committed to improving day by day?”
The challenge for hospitalist groups, he adds is to clearly state expectations for physicians, hold them accountable, make sure they understand the group’s goals and standards, and offer the tools they need to improve. An example could be access to English as a Second Language instruction to enhance communication.
A new study comparing physicians who were trained in the U.S. with those trained in medical schools abroad should offer reassurance to patients, families, and professional peers who hold biases against foreign-born or foreign-trained physicians, the lead author says.
John Norcini, PhD, CEO of the Foundation for Advancement of International Medical Education and Research, and colleagues studied 244,000 hospitalized Pennsylvania patients with congestive heart failure and acute myocardial infarction. They found that mortality rates were slightly lower for physicians who were trained abroad and were not U.S. citizens when they entered medical school. The study showed higher rates for U.S. citizens who went overseas for their medical training.
The Norcini study (Health Affairs. 2010;29:1461-1468) focused on family medicine, internal medicine, and cardiology physicians, but it did not identify hospitalists. One-quarter of all physicians practicing in the U.S. are foreign-trained; however, a greater proportion are found in primary care and internal medicine. For hospitalists, the foreign-trained percentage might be even higher, 40% according to Philip Miller of the physician recruiting firm Merritt Hawkins.
One thing that can be said about international medical graduates is that they are a “crucial and growing part of the hospital medicine workforce, and we welcome them,” says Winthrop Whitcomb, MD, MHM, former SHM president and medical director of healthcare quality at Baystate Medical Center in Springfield, Mass. “I find, having worked with physicians trained all over the world, that for the best ones, it’s what they do every day, not where they came from. Are they consistent, careful, compassionate and committed to improving day by day?”
The challenge for hospitalist groups, he adds is to clearly state expectations for physicians, hold them accountable, make sure they understand the group’s goals and standards, and offer the tools they need to improve. An example could be access to English as a Second Language instruction to enhance communication.
A new study comparing physicians who were trained in the U.S. with those trained in medical schools abroad should offer reassurance to patients, families, and professional peers who hold biases against foreign-born or foreign-trained physicians, the lead author says.
John Norcini, PhD, CEO of the Foundation for Advancement of International Medical Education and Research, and colleagues studied 244,000 hospitalized Pennsylvania patients with congestive heart failure and acute myocardial infarction. They found that mortality rates were slightly lower for physicians who were trained abroad and were not U.S. citizens when they entered medical school. The study showed higher rates for U.S. citizens who went overseas for their medical training.
The Norcini study (Health Affairs. 2010;29:1461-1468) focused on family medicine, internal medicine, and cardiology physicians, but it did not identify hospitalists. One-quarter of all physicians practicing in the U.S. are foreign-trained; however, a greater proportion are found in primary care and internal medicine. For hospitalists, the foreign-trained percentage might be even higher, 40% according to Philip Miller of the physician recruiting firm Merritt Hawkins.
One thing that can be said about international medical graduates is that they are a “crucial and growing part of the hospital medicine workforce, and we welcome them,” says Winthrop Whitcomb, MD, MHM, former SHM president and medical director of healthcare quality at Baystate Medical Center in Springfield, Mass. “I find, having worked with physicians trained all over the world, that for the best ones, it’s what they do every day, not where they came from. Are they consistent, careful, compassionate and committed to improving day by day?”
The challenge for hospitalist groups, he adds is to clearly state expectations for physicians, hold them accountable, make sure they understand the group’s goals and standards, and offer the tools they need to improve. An example could be access to English as a Second Language instruction to enhance communication.