User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Headache for inpatients with COVID-19 may predict better survival
, according to recent research published in the journal Headache.
In the systematic review and meta-analysis, Víctor J. Gallardo, MSc, of the headache and neurologic pain research group, Vall d’Hebron Research Institute at the Universitat Autònoma de Barcelona, and colleagues performed a search of studies in PubMed involving headache symptoms, disease survival, and inpatient COVID-19 cases published between December 2019 and December 2020. Overall, 48 studies were identified, consisting of 43,169 inpatients with COVID-19. Using random-effects pooling models, Mr. Gallardo and colleagues estimated the prevalence of headache for inpatients who survived COVID-19, compared with those who did not survive.
Within those studies, 35,132 inpatients (81.4%) survived, while 8,037 inpatients (18.6%) died from COVID-19. The researchers found that inpatients with COVID-19 and headache symptoms had a significantly higher survival rate compared with inpatients with COVID-19 without headache symptoms (risk ratio, 1.90; 95% confidence interval, 1.46-2.47; P < .0001). There was an overall pooled prevalence of headache as a COVID-19 symptom in 10.4% of inpatients, which was reduced to an estimated pooled prevalence of 9.7% after the researchers removed outlier studies in a sensitivity analysis.
Other COVID-19 symptoms that led to improved rates of survival among inpatients were anosmia (RR, 2.94; 95% CI, 1.94-4.45) and myalgia (RR, 1.57; 95% CI, 1.34-1.83) as well as nausea or vomiting (RR, 1.41; 95% CI, 1.08-1.82), while symptoms such as dyspnea, diabetes, chronic liver diseases, chronic respiratory diseases, and chronic kidney diseases were more likely to increase the risk of dying from COVID-19.
The researchers noted several limitations in their meta-analysis that may make their findings less generalizable to future SARS-CoV-2 variants, such as including only studies that were published before COVID-19 vaccines were available and before more infectious SARS-CoV-2 variants like the B.1.617.2 (Delta) variant emerged. They also included studies where inpatients were not tested for COVID-19 because access to testing was not widely available.
“Our meta-analysis points toward a novel possibility: Headache arising secondary to an infection is not a ‘nonspecific’ symptom, but rather it may be a marker of enhanced likelihood of survival. That is, we find that patients reporting headache in the setting of COVID-19 are at reduced risk of death,” Mr. Gallardo and colleagues wrote.
More data needed on association between headache and COVID-19
While headache appeared to affect a small proportion of overall inpatients with COVID-19, the researchers noted this might be because individuals with COVID-19 and headache symptoms are less likely to require hospitalization or a visit to the ED. Another potential explanation is that “people with primary headache disorders, including migraine, may be more likely to report symptoms of COVID-19, but they also may be relatively less likely to experience a life-threatening COVID-19 disease course.”
The researchers said this potential association should be explored in future studies as well as in other viral infections or postviral syndromes such as long COVID. “Defining specific headache mechanisms that could enhance survival from viral infections represents an opportunity for the potential discovery of improved viral therapeutics, as well as for understanding whether, and how, primary headache disorders may be adaptive.”
In a comment, Morris Levin, MD, director of the University of California San Francisco Headache Center, said the findings “of this very thought-provoking review suggest that reporting a headache during a COVID-19 infection seems to be associated with better recovery in hospitalized patients.”
Dr. Levin, who was not involved with the study, acknowledged the researchers’ explanation for the overall low rate of headache in these inpatients as one possible explanation.
“Another could be that sick COVID patients were much more troubled by other symptoms like respiratory distress, which overshadowed their headache symptoms, particularly if they were very ill or if the headache pain was of only mild to moderate severity,” he said. “That could also be an alternate explanation for why less dangerously ill hospitalized patients seemed to have more headaches.”
One limitation he saw in the meta-analysis was how clearly the clinicians characterized headache symptoms in each reviewed study. Dr. Levin suggested a retrospective assessment of premorbid migraine history in hospitalized patients with COVID-19, including survivors and fatalities, might have helped clarify this issue. “The headaches themselves were not characterized so drawing conclusions regarding migraine is challenging.”
Dr. Levin noted it is still not well understood how acute and persistent headaches and other neurological symptoms like mental fog occur in patients with COVID-19. We also do not fully understand the natural history of post-COVID headaches and other neurologic sequelae and the management options for acute and persistent neurological sequelae.
Three authors reported personal and institutional relationships in the form of grants, consultancies, speaker’s bureau positions, guidelines committee member appointments, and editorial board positions for a variety of pharmaceutical companies, agencies, societies, and other organizations. Mr. Gallardo reported no relevant financial disclosures. Dr. Levin reported no relevant financial disclosures.
, according to recent research published in the journal Headache.
In the systematic review and meta-analysis, Víctor J. Gallardo, MSc, of the headache and neurologic pain research group, Vall d’Hebron Research Institute at the Universitat Autònoma de Barcelona, and colleagues performed a search of studies in PubMed involving headache symptoms, disease survival, and inpatient COVID-19 cases published between December 2019 and December 2020. Overall, 48 studies were identified, consisting of 43,169 inpatients with COVID-19. Using random-effects pooling models, Mr. Gallardo and colleagues estimated the prevalence of headache for inpatients who survived COVID-19, compared with those who did not survive.
Within those studies, 35,132 inpatients (81.4%) survived, while 8,037 inpatients (18.6%) died from COVID-19. The researchers found that inpatients with COVID-19 and headache symptoms had a significantly higher survival rate compared with inpatients with COVID-19 without headache symptoms (risk ratio, 1.90; 95% confidence interval, 1.46-2.47; P < .0001). There was an overall pooled prevalence of headache as a COVID-19 symptom in 10.4% of inpatients, which was reduced to an estimated pooled prevalence of 9.7% after the researchers removed outlier studies in a sensitivity analysis.
Other COVID-19 symptoms that led to improved rates of survival among inpatients were anosmia (RR, 2.94; 95% CI, 1.94-4.45) and myalgia (RR, 1.57; 95% CI, 1.34-1.83) as well as nausea or vomiting (RR, 1.41; 95% CI, 1.08-1.82), while symptoms such as dyspnea, diabetes, chronic liver diseases, chronic respiratory diseases, and chronic kidney diseases were more likely to increase the risk of dying from COVID-19.
The researchers noted several limitations in their meta-analysis that may make their findings less generalizable to future SARS-CoV-2 variants, such as including only studies that were published before COVID-19 vaccines were available and before more infectious SARS-CoV-2 variants like the B.1.617.2 (Delta) variant emerged. They also included studies where inpatients were not tested for COVID-19 because access to testing was not widely available.
“Our meta-analysis points toward a novel possibility: Headache arising secondary to an infection is not a ‘nonspecific’ symptom, but rather it may be a marker of enhanced likelihood of survival. That is, we find that patients reporting headache in the setting of COVID-19 are at reduced risk of death,” Mr. Gallardo and colleagues wrote.
More data needed on association between headache and COVID-19
While headache appeared to affect a small proportion of overall inpatients with COVID-19, the researchers noted this might be because individuals with COVID-19 and headache symptoms are less likely to require hospitalization or a visit to the ED. Another potential explanation is that “people with primary headache disorders, including migraine, may be more likely to report symptoms of COVID-19, but they also may be relatively less likely to experience a life-threatening COVID-19 disease course.”
The researchers said this potential association should be explored in future studies as well as in other viral infections or postviral syndromes such as long COVID. “Defining specific headache mechanisms that could enhance survival from viral infections represents an opportunity for the potential discovery of improved viral therapeutics, as well as for understanding whether, and how, primary headache disorders may be adaptive.”
In a comment, Morris Levin, MD, director of the University of California San Francisco Headache Center, said the findings “of this very thought-provoking review suggest that reporting a headache during a COVID-19 infection seems to be associated with better recovery in hospitalized patients.”
Dr. Levin, who was not involved with the study, acknowledged the researchers’ explanation for the overall low rate of headache in these inpatients as one possible explanation.
“Another could be that sick COVID patients were much more troubled by other symptoms like respiratory distress, which overshadowed their headache symptoms, particularly if they were very ill or if the headache pain was of only mild to moderate severity,” he said. “That could also be an alternate explanation for why less dangerously ill hospitalized patients seemed to have more headaches.”
One limitation he saw in the meta-analysis was how clearly the clinicians characterized headache symptoms in each reviewed study. Dr. Levin suggested a retrospective assessment of premorbid migraine history in hospitalized patients with COVID-19, including survivors and fatalities, might have helped clarify this issue. “The headaches themselves were not characterized so drawing conclusions regarding migraine is challenging.”
Dr. Levin noted it is still not well understood how acute and persistent headaches and other neurological symptoms like mental fog occur in patients with COVID-19. We also do not fully understand the natural history of post-COVID headaches and other neurologic sequelae and the management options for acute and persistent neurological sequelae.
Three authors reported personal and institutional relationships in the form of grants, consultancies, speaker’s bureau positions, guidelines committee member appointments, and editorial board positions for a variety of pharmaceutical companies, agencies, societies, and other organizations. Mr. Gallardo reported no relevant financial disclosures. Dr. Levin reported no relevant financial disclosures.
, according to recent research published in the journal Headache.
In the systematic review and meta-analysis, Víctor J. Gallardo, MSc, of the headache and neurologic pain research group, Vall d’Hebron Research Institute at the Universitat Autònoma de Barcelona, and colleagues performed a search of studies in PubMed involving headache symptoms, disease survival, and inpatient COVID-19 cases published between December 2019 and December 2020. Overall, 48 studies were identified, consisting of 43,169 inpatients with COVID-19. Using random-effects pooling models, Mr. Gallardo and colleagues estimated the prevalence of headache for inpatients who survived COVID-19, compared with those who did not survive.
Within those studies, 35,132 inpatients (81.4%) survived, while 8,037 inpatients (18.6%) died from COVID-19. The researchers found that inpatients with COVID-19 and headache symptoms had a significantly higher survival rate compared with inpatients with COVID-19 without headache symptoms (risk ratio, 1.90; 95% confidence interval, 1.46-2.47; P < .0001). There was an overall pooled prevalence of headache as a COVID-19 symptom in 10.4% of inpatients, which was reduced to an estimated pooled prevalence of 9.7% after the researchers removed outlier studies in a sensitivity analysis.
Other COVID-19 symptoms that led to improved rates of survival among inpatients were anosmia (RR, 2.94; 95% CI, 1.94-4.45) and myalgia (RR, 1.57; 95% CI, 1.34-1.83) as well as nausea or vomiting (RR, 1.41; 95% CI, 1.08-1.82), while symptoms such as dyspnea, diabetes, chronic liver diseases, chronic respiratory diseases, and chronic kidney diseases were more likely to increase the risk of dying from COVID-19.
The researchers noted several limitations in their meta-analysis that may make their findings less generalizable to future SARS-CoV-2 variants, such as including only studies that were published before COVID-19 vaccines were available and before more infectious SARS-CoV-2 variants like the B.1.617.2 (Delta) variant emerged. They also included studies where inpatients were not tested for COVID-19 because access to testing was not widely available.
“Our meta-analysis points toward a novel possibility: Headache arising secondary to an infection is not a ‘nonspecific’ symptom, but rather it may be a marker of enhanced likelihood of survival. That is, we find that patients reporting headache in the setting of COVID-19 are at reduced risk of death,” Mr. Gallardo and colleagues wrote.
More data needed on association between headache and COVID-19
While headache appeared to affect a small proportion of overall inpatients with COVID-19, the researchers noted this might be because individuals with COVID-19 and headache symptoms are less likely to require hospitalization or a visit to the ED. Another potential explanation is that “people with primary headache disorders, including migraine, may be more likely to report symptoms of COVID-19, but they also may be relatively less likely to experience a life-threatening COVID-19 disease course.”
The researchers said this potential association should be explored in future studies as well as in other viral infections or postviral syndromes such as long COVID. “Defining specific headache mechanisms that could enhance survival from viral infections represents an opportunity for the potential discovery of improved viral therapeutics, as well as for understanding whether, and how, primary headache disorders may be adaptive.”
In a comment, Morris Levin, MD, director of the University of California San Francisco Headache Center, said the findings “of this very thought-provoking review suggest that reporting a headache during a COVID-19 infection seems to be associated with better recovery in hospitalized patients.”
Dr. Levin, who was not involved with the study, acknowledged the researchers’ explanation for the overall low rate of headache in these inpatients as one possible explanation.
“Another could be that sick COVID patients were much more troubled by other symptoms like respiratory distress, which overshadowed their headache symptoms, particularly if they were very ill or if the headache pain was of only mild to moderate severity,” he said. “That could also be an alternate explanation for why less dangerously ill hospitalized patients seemed to have more headaches.”
One limitation he saw in the meta-analysis was how clearly the clinicians characterized headache symptoms in each reviewed study. Dr. Levin suggested a retrospective assessment of premorbid migraine history in hospitalized patients with COVID-19, including survivors and fatalities, might have helped clarify this issue. “The headaches themselves were not characterized so drawing conclusions regarding migraine is challenging.”
Dr. Levin noted it is still not well understood how acute and persistent headaches and other neurological symptoms like mental fog occur in patients with COVID-19. We also do not fully understand the natural history of post-COVID headaches and other neurologic sequelae and the management options for acute and persistent neurological sequelae.
Three authors reported personal and institutional relationships in the form of grants, consultancies, speaker’s bureau positions, guidelines committee member appointments, and editorial board positions for a variety of pharmaceutical companies, agencies, societies, and other organizations. Mr. Gallardo reported no relevant financial disclosures. Dr. Levin reported no relevant financial disclosures.
FROM HEADACHE
For many, long COVID’s impacts go on and on, major study says
in the same time frame, a large study out of Scotland found.
Multiple studies are evaluating people with long COVID in the hopes of figuring out why some people experience debilitating symptoms long after their primary infection ends and others either do not or recover more quickly.
This current study is notable for its large size – 96,238 people. Researchers checked in with participants at 6, 12, and 18 months, and included a group of people never infected with the coronavirus to help investigators make a stronger case.
“A lot of the symptoms of long COVID are nonspecific and therefore can occur in people never infected,” says senior study author Jill P. Pell, MD, head of the School of Health and Wellbeing at the University of Glasgow in Scotland.
Ruling out coincidence
This study shows that people experienced a wide range of symptoms after becoming infected with COVID-19 at a significantly higher rate than those who were never infected, “thereby confirming that they were genuinely associated with COVID and not merely a coincidence,” she said.
Among 21,525 people who had COVID-19 and had symptoms, tiredness, headache and muscle aches or muscle weakness were the most common ongoing symptoms.
Loss of smell was almost nine times more likely in this group compared to the never-infected group in one analysis where researchers controlled for other possible factors. The risk for loss of taste was almost six times greater, followed by risk of breathlessness at three times higher.
Long COVID risk was highest after a severe original infection and among older people, women, Black, and South Asian populations, people with socioeconomic disadvantages, and those with more than one underlying health condition.
Adding up the 6% with no recovery after 18 months and 42% with partial recovery means that between 6 and 18 months following symptomatic coronavirus infection, almost half of those infected still experience persistent symptoms.
Vaccination validated
On the plus side, people vaccinated against COVID-19 before getting infected had a lower risk for some persistent symptoms. In addition, Dr. Pell and colleagues found no evidence that people who experienced asymptomatic infection were likely to experience long COVID symptoms or challenges with activities of daily living.
The findings of the Long-COVID in Scotland Study (Long-CISS) were published in the journal Nature Communications.
‘More long COVID than ever before’
“Unfortunately, these long COVID symptoms are not getting better as the cases of COVID get milder,” said Thomas Gut, DO, medical director for the post-COVID recovery program at Staten Island (N.Y.) University Hospital. “Quite the opposite – this infection has become so common in a community because it’s so mild and spreading so rapidly that we’re seeing more long COVID symptoms than ever before.”
Although most patients he sees with long COVID resolve their symptoms within 3-6 months, “We do see some patients who require short-term disability because their symptoms continue past 6 months and out to 2 years,” said Dr. Gut, a hospitalist at Staten Island University Hospital, a member hospital of Northwell Health.
Patients with fatigue and neurocognitive symptoms “have a very tough time going back to work. Short-term disability gives them the time and finances to pursue specialty care with cardiology, pulmonary, and neurocognitive testing,” he said.
Support the whole person
The burden of living with long COVID goes beyond the persistent symptoms. “Long COVID can have wide-ranging impacts – not only on health but also quality of life and activities of daily living [including] work, mobility, self-care and more,” Dr. Pell said. “So, people with long COVID need support relevant to their individual needs and this may extend beyond the health care sector, for example including social services, school or workplace.”
Still, Lisa Penziner, RN, founder of the COVID Long Haulers Support Group in Westchester and Long Island, N.Y., said while people with the most severe cases of COVID-19 tended to have the worst long COVID symptoms, they’re not the only ones.
“We saw many post-COVID members who had mild cases and their long-haul symptoms were worse weeks later than the virus itself,” said Md. Penziner.
She estimates that 80%-90% of her support group members recover within 6 months. “However, there are others who were experiencing symptoms for much longer.”
Respiratory treatment, physical therapy, and other follow-up doctor visits are common after 6 months, for example.
“Additionally, there is a mental health component to recovery as well, meaning that the patient must learn to live while experiencing lingering, long-haul COVID symptoms in work and daily life,” said Ms. Penziner, director of special projects at North Westchester Restorative Therapy & Nursing.
In addition to ongoing medical care, people with long COVID need understanding, she said.
“While long-haul symptoms do not happen to everyone, it is proven that many do experience long-haul symptoms, and the support of the community in understanding is important.”
Limitations of the study
Dr. Pell and colleagues noted some strengths and weaknesses to their study. For example, “as a general population study, our findings provide a better indication of the overall risk and burden of long COVID than hospitalized cohorts,” they noted.
Also, the Scottish population is 96% White, so other long COVID studies with more diverse participants are warranted.
Another potential weakness is the response rate of 16% among those invited to participate in the study, which Dr. Pell and colleagues addressed: “Our cohort included a large sample (33,281) of people previously infected and the response rate of 16% overall and 20% among people who had symptomatic infection was consistent with previous studies that have used SMS text invitations as the sole method of recruitment.”
“We tell patients this should last 3-6 months, but some patients have longer recovery periods,” Dr. Gut said. “We’re here for them. We have a lot of services available to help get them through the recovery process, and we have a lot of options to help support them.”
“What we found most helpful is when there is peer-to-peer support, reaffirming to the member that they are not alone in the long-haul battle, which has been a major benefit of the support group,” Ms. Penziner said.
A version of this article first appeared on WebMD.com.
in the same time frame, a large study out of Scotland found.
Multiple studies are evaluating people with long COVID in the hopes of figuring out why some people experience debilitating symptoms long after their primary infection ends and others either do not or recover more quickly.
This current study is notable for its large size – 96,238 people. Researchers checked in with participants at 6, 12, and 18 months, and included a group of people never infected with the coronavirus to help investigators make a stronger case.
“A lot of the symptoms of long COVID are nonspecific and therefore can occur in people never infected,” says senior study author Jill P. Pell, MD, head of the School of Health and Wellbeing at the University of Glasgow in Scotland.
Ruling out coincidence
This study shows that people experienced a wide range of symptoms after becoming infected with COVID-19 at a significantly higher rate than those who were never infected, “thereby confirming that they were genuinely associated with COVID and not merely a coincidence,” she said.
Among 21,525 people who had COVID-19 and had symptoms, tiredness, headache and muscle aches or muscle weakness were the most common ongoing symptoms.
Loss of smell was almost nine times more likely in this group compared to the never-infected group in one analysis where researchers controlled for other possible factors. The risk for loss of taste was almost six times greater, followed by risk of breathlessness at three times higher.
Long COVID risk was highest after a severe original infection and among older people, women, Black, and South Asian populations, people with socioeconomic disadvantages, and those with more than one underlying health condition.
Adding up the 6% with no recovery after 18 months and 42% with partial recovery means that between 6 and 18 months following symptomatic coronavirus infection, almost half of those infected still experience persistent symptoms.
Vaccination validated
On the plus side, people vaccinated against COVID-19 before getting infected had a lower risk for some persistent symptoms. In addition, Dr. Pell and colleagues found no evidence that people who experienced asymptomatic infection were likely to experience long COVID symptoms or challenges with activities of daily living.
The findings of the Long-COVID in Scotland Study (Long-CISS) were published in the journal Nature Communications.
‘More long COVID than ever before’
“Unfortunately, these long COVID symptoms are not getting better as the cases of COVID get milder,” said Thomas Gut, DO, medical director for the post-COVID recovery program at Staten Island (N.Y.) University Hospital. “Quite the opposite – this infection has become so common in a community because it’s so mild and spreading so rapidly that we’re seeing more long COVID symptoms than ever before.”
Although most patients he sees with long COVID resolve their symptoms within 3-6 months, “We do see some patients who require short-term disability because their symptoms continue past 6 months and out to 2 years,” said Dr. Gut, a hospitalist at Staten Island University Hospital, a member hospital of Northwell Health.
Patients with fatigue and neurocognitive symptoms “have a very tough time going back to work. Short-term disability gives them the time and finances to pursue specialty care with cardiology, pulmonary, and neurocognitive testing,” he said.
Support the whole person
The burden of living with long COVID goes beyond the persistent symptoms. “Long COVID can have wide-ranging impacts – not only on health but also quality of life and activities of daily living [including] work, mobility, self-care and more,” Dr. Pell said. “So, people with long COVID need support relevant to their individual needs and this may extend beyond the health care sector, for example including social services, school or workplace.”
Still, Lisa Penziner, RN, founder of the COVID Long Haulers Support Group in Westchester and Long Island, N.Y., said while people with the most severe cases of COVID-19 tended to have the worst long COVID symptoms, they’re not the only ones.
“We saw many post-COVID members who had mild cases and their long-haul symptoms were worse weeks later than the virus itself,” said Md. Penziner.
She estimates that 80%-90% of her support group members recover within 6 months. “However, there are others who were experiencing symptoms for much longer.”
Respiratory treatment, physical therapy, and other follow-up doctor visits are common after 6 months, for example.
“Additionally, there is a mental health component to recovery as well, meaning that the patient must learn to live while experiencing lingering, long-haul COVID symptoms in work and daily life,” said Ms. Penziner, director of special projects at North Westchester Restorative Therapy & Nursing.
In addition to ongoing medical care, people with long COVID need understanding, she said.
“While long-haul symptoms do not happen to everyone, it is proven that many do experience long-haul symptoms, and the support of the community in understanding is important.”
Limitations of the study
Dr. Pell and colleagues noted some strengths and weaknesses to their study. For example, “as a general population study, our findings provide a better indication of the overall risk and burden of long COVID than hospitalized cohorts,” they noted.
Also, the Scottish population is 96% White, so other long COVID studies with more diverse participants are warranted.
Another potential weakness is the response rate of 16% among those invited to participate in the study, which Dr. Pell and colleagues addressed: “Our cohort included a large sample (33,281) of people previously infected and the response rate of 16% overall and 20% among people who had symptomatic infection was consistent with previous studies that have used SMS text invitations as the sole method of recruitment.”
“We tell patients this should last 3-6 months, but some patients have longer recovery periods,” Dr. Gut said. “We’re here for them. We have a lot of services available to help get them through the recovery process, and we have a lot of options to help support them.”
“What we found most helpful is when there is peer-to-peer support, reaffirming to the member that they are not alone in the long-haul battle, which has been a major benefit of the support group,” Ms. Penziner said.
A version of this article first appeared on WebMD.com.
in the same time frame, a large study out of Scotland found.
Multiple studies are evaluating people with long COVID in the hopes of figuring out why some people experience debilitating symptoms long after their primary infection ends and others either do not or recover more quickly.
This current study is notable for its large size – 96,238 people. Researchers checked in with participants at 6, 12, and 18 months, and included a group of people never infected with the coronavirus to help investigators make a stronger case.
“A lot of the symptoms of long COVID are nonspecific and therefore can occur in people never infected,” says senior study author Jill P. Pell, MD, head of the School of Health and Wellbeing at the University of Glasgow in Scotland.
Ruling out coincidence
This study shows that people experienced a wide range of symptoms after becoming infected with COVID-19 at a significantly higher rate than those who were never infected, “thereby confirming that they were genuinely associated with COVID and not merely a coincidence,” she said.
Among 21,525 people who had COVID-19 and had symptoms, tiredness, headache and muscle aches or muscle weakness were the most common ongoing symptoms.
Loss of smell was almost nine times more likely in this group compared to the never-infected group in one analysis where researchers controlled for other possible factors. The risk for loss of taste was almost six times greater, followed by risk of breathlessness at three times higher.
Long COVID risk was highest after a severe original infection and among older people, women, Black, and South Asian populations, people with socioeconomic disadvantages, and those with more than one underlying health condition.
Adding up the 6% with no recovery after 18 months and 42% with partial recovery means that between 6 and 18 months following symptomatic coronavirus infection, almost half of those infected still experience persistent symptoms.
Vaccination validated
On the plus side, people vaccinated against COVID-19 before getting infected had a lower risk for some persistent symptoms. In addition, Dr. Pell and colleagues found no evidence that people who experienced asymptomatic infection were likely to experience long COVID symptoms or challenges with activities of daily living.
The findings of the Long-COVID in Scotland Study (Long-CISS) were published in the journal Nature Communications.
‘More long COVID than ever before’
“Unfortunately, these long COVID symptoms are not getting better as the cases of COVID get milder,” said Thomas Gut, DO, medical director for the post-COVID recovery program at Staten Island (N.Y.) University Hospital. “Quite the opposite – this infection has become so common in a community because it’s so mild and spreading so rapidly that we’re seeing more long COVID symptoms than ever before.”
Although most patients he sees with long COVID resolve their symptoms within 3-6 months, “We do see some patients who require short-term disability because their symptoms continue past 6 months and out to 2 years,” said Dr. Gut, a hospitalist at Staten Island University Hospital, a member hospital of Northwell Health.
Patients with fatigue and neurocognitive symptoms “have a very tough time going back to work. Short-term disability gives them the time and finances to pursue specialty care with cardiology, pulmonary, and neurocognitive testing,” he said.
Support the whole person
The burden of living with long COVID goes beyond the persistent symptoms. “Long COVID can have wide-ranging impacts – not only on health but also quality of life and activities of daily living [including] work, mobility, self-care and more,” Dr. Pell said. “So, people with long COVID need support relevant to their individual needs and this may extend beyond the health care sector, for example including social services, school or workplace.”
Still, Lisa Penziner, RN, founder of the COVID Long Haulers Support Group in Westchester and Long Island, N.Y., said while people with the most severe cases of COVID-19 tended to have the worst long COVID symptoms, they’re not the only ones.
“We saw many post-COVID members who had mild cases and their long-haul symptoms were worse weeks later than the virus itself,” said Md. Penziner.
She estimates that 80%-90% of her support group members recover within 6 months. “However, there are others who were experiencing symptoms for much longer.”
Respiratory treatment, physical therapy, and other follow-up doctor visits are common after 6 months, for example.
“Additionally, there is a mental health component to recovery as well, meaning that the patient must learn to live while experiencing lingering, long-haul COVID symptoms in work and daily life,” said Ms. Penziner, director of special projects at North Westchester Restorative Therapy & Nursing.
In addition to ongoing medical care, people with long COVID need understanding, she said.
“While long-haul symptoms do not happen to everyone, it is proven that many do experience long-haul symptoms, and the support of the community in understanding is important.”
Limitations of the study
Dr. Pell and colleagues noted some strengths and weaknesses to their study. For example, “as a general population study, our findings provide a better indication of the overall risk and burden of long COVID than hospitalized cohorts,” they noted.
Also, the Scottish population is 96% White, so other long COVID studies with more diverse participants are warranted.
Another potential weakness is the response rate of 16% among those invited to participate in the study, which Dr. Pell and colleagues addressed: “Our cohort included a large sample (33,281) of people previously infected and the response rate of 16% overall and 20% among people who had symptomatic infection was consistent with previous studies that have used SMS text invitations as the sole method of recruitment.”
“We tell patients this should last 3-6 months, but some patients have longer recovery periods,” Dr. Gut said. “We’re here for them. We have a lot of services available to help get them through the recovery process, and we have a lot of options to help support them.”
“What we found most helpful is when there is peer-to-peer support, reaffirming to the member that they are not alone in the long-haul battle, which has been a major benefit of the support group,” Ms. Penziner said.
A version of this article first appeared on WebMD.com.
FROM NATURE COMMUNICATIONS
63% of long COVID patients are women, study says
according to a new study published in JAMA.
The global study also found that about 6% of people with symptomatic infections had long COVID in 2020 and 2021. The risk for long COVID seemed to be greater among those who needed hospitalization, especially those who needed intensive care.
“Quantifying the number of individuals with long COVID may help policy makers ensure adequate access to services to guide people toward recovery, return to the workplace or school, and restore their mental health and social life,” the researchers wrote.
The study team, which included dozens of researchers across nearly every continent, analyzed data from 54 studies and two databases for more than 1 million patients in 22 countries who had symptomatic COVID infections in 2020 and 2021. They looked at three long COVID symptom types: persistent fatigue with bodily pain or mood swings, ongoing respiratory problems, and cognitive issues. The study included people aged 4-66.
Overall, 6.2% of people reported one of the long COVID symptom types, including 3.7% with ongoing respiratory problems, 3.2% with persistent fatigue and bodily pain or mood swings, and 2.2% with cognitive problems. Among those with long COVID, 38% of people reported more than one symptom cluster.
At 3 months after infection, long COVID symptoms were nearly twice as common in women who were at least 20 years old at 10.6%, compared with men who were at least 20 years old at 5.4%.
Children and teens appeared to have lower risks of long COVID. About 2.8% of patients under age 20 with symptomatic infection developed long-term issues.
The estimated average duration of long COVID symptoms was 9 months among hospitalized patients and 4 months among those who weren’t hospitalized. About 15% of people with long COVID symptoms 3 months after the initial infection continued to have symptoms at 12 months.
The study was largely based on detailed data from ongoing COVID-19 studies in the United States, Austria, the Faroe Islands, Germany, Iran, Italy, the Netherlands, Russia, Sweden, and Switzerland, according to UPI. It was supplemented by published data and research conducted as part of the Global Burden of Diseases, Injuries and Risk Factors Study. The dozens of researchers are referred to as “Global Burden of Disease Long COVID Collaborators.”
The study had limitations, the researchers said, including the assumption that long COVID follows a similar course in all countries. Additional studies may show how long COVID symptoms and severity may vary in different countries and continents.
Ultimately, ongoing studies of large numbers of people with long COVID could help scientists and public health officials understand risk factors and ways to treat the debilitating condition, the study authors wrote, noting that “postinfection fatigue syndrome” has been reported before, namely during the 1918 flu pandemic, after the SARS outbreak in 2003, and after the Ebola epidemic in West Africa in 2014.
“Similar symptoms have been reported after other viral infections, including the Epstein-Barr virus, mononucleosis, and dengue, as well as after nonviral infections such as Q fever, Lyme disease and giardiasis,” they wrote.
Several study investigators reported receiving grants and personal fees from a variety of sources.
A version of this article first appeared on Medscape.com.
according to a new study published in JAMA.
The global study also found that about 6% of people with symptomatic infections had long COVID in 2020 and 2021. The risk for long COVID seemed to be greater among those who needed hospitalization, especially those who needed intensive care.
“Quantifying the number of individuals with long COVID may help policy makers ensure adequate access to services to guide people toward recovery, return to the workplace or school, and restore their mental health and social life,” the researchers wrote.
The study team, which included dozens of researchers across nearly every continent, analyzed data from 54 studies and two databases for more than 1 million patients in 22 countries who had symptomatic COVID infections in 2020 and 2021. They looked at three long COVID symptom types: persistent fatigue with bodily pain or mood swings, ongoing respiratory problems, and cognitive issues. The study included people aged 4-66.
Overall, 6.2% of people reported one of the long COVID symptom types, including 3.7% with ongoing respiratory problems, 3.2% with persistent fatigue and bodily pain or mood swings, and 2.2% with cognitive problems. Among those with long COVID, 38% of people reported more than one symptom cluster.
At 3 months after infection, long COVID symptoms were nearly twice as common in women who were at least 20 years old at 10.6%, compared with men who were at least 20 years old at 5.4%.
Children and teens appeared to have lower risks of long COVID. About 2.8% of patients under age 20 with symptomatic infection developed long-term issues.
The estimated average duration of long COVID symptoms was 9 months among hospitalized patients and 4 months among those who weren’t hospitalized. About 15% of people with long COVID symptoms 3 months after the initial infection continued to have symptoms at 12 months.
The study was largely based on detailed data from ongoing COVID-19 studies in the United States, Austria, the Faroe Islands, Germany, Iran, Italy, the Netherlands, Russia, Sweden, and Switzerland, according to UPI. It was supplemented by published data and research conducted as part of the Global Burden of Diseases, Injuries and Risk Factors Study. The dozens of researchers are referred to as “Global Burden of Disease Long COVID Collaborators.”
The study had limitations, the researchers said, including the assumption that long COVID follows a similar course in all countries. Additional studies may show how long COVID symptoms and severity may vary in different countries and continents.
Ultimately, ongoing studies of large numbers of people with long COVID could help scientists and public health officials understand risk factors and ways to treat the debilitating condition, the study authors wrote, noting that “postinfection fatigue syndrome” has been reported before, namely during the 1918 flu pandemic, after the SARS outbreak in 2003, and after the Ebola epidemic in West Africa in 2014.
“Similar symptoms have been reported after other viral infections, including the Epstein-Barr virus, mononucleosis, and dengue, as well as after nonviral infections such as Q fever, Lyme disease and giardiasis,” they wrote.
Several study investigators reported receiving grants and personal fees from a variety of sources.
A version of this article first appeared on Medscape.com.
according to a new study published in JAMA.
The global study also found that about 6% of people with symptomatic infections had long COVID in 2020 and 2021. The risk for long COVID seemed to be greater among those who needed hospitalization, especially those who needed intensive care.
“Quantifying the number of individuals with long COVID may help policy makers ensure adequate access to services to guide people toward recovery, return to the workplace or school, and restore their mental health and social life,” the researchers wrote.
The study team, which included dozens of researchers across nearly every continent, analyzed data from 54 studies and two databases for more than 1 million patients in 22 countries who had symptomatic COVID infections in 2020 and 2021. They looked at three long COVID symptom types: persistent fatigue with bodily pain or mood swings, ongoing respiratory problems, and cognitive issues. The study included people aged 4-66.
Overall, 6.2% of people reported one of the long COVID symptom types, including 3.7% with ongoing respiratory problems, 3.2% with persistent fatigue and bodily pain or mood swings, and 2.2% with cognitive problems. Among those with long COVID, 38% of people reported more than one symptom cluster.
At 3 months after infection, long COVID symptoms were nearly twice as common in women who were at least 20 years old at 10.6%, compared with men who were at least 20 years old at 5.4%.
Children and teens appeared to have lower risks of long COVID. About 2.8% of patients under age 20 with symptomatic infection developed long-term issues.
The estimated average duration of long COVID symptoms was 9 months among hospitalized patients and 4 months among those who weren’t hospitalized. About 15% of people with long COVID symptoms 3 months after the initial infection continued to have symptoms at 12 months.
The study was largely based on detailed data from ongoing COVID-19 studies in the United States, Austria, the Faroe Islands, Germany, Iran, Italy, the Netherlands, Russia, Sweden, and Switzerland, according to UPI. It was supplemented by published data and research conducted as part of the Global Burden of Diseases, Injuries and Risk Factors Study. The dozens of researchers are referred to as “Global Burden of Disease Long COVID Collaborators.”
The study had limitations, the researchers said, including the assumption that long COVID follows a similar course in all countries. Additional studies may show how long COVID symptoms and severity may vary in different countries and continents.
Ultimately, ongoing studies of large numbers of people with long COVID could help scientists and public health officials understand risk factors and ways to treat the debilitating condition, the study authors wrote, noting that “postinfection fatigue syndrome” has been reported before, namely during the 1918 flu pandemic, after the SARS outbreak in 2003, and after the Ebola epidemic in West Africa in 2014.
“Similar symptoms have been reported after other viral infections, including the Epstein-Barr virus, mononucleosis, and dengue, as well as after nonviral infections such as Q fever, Lyme disease and giardiasis,” they wrote.
Several study investigators reported receiving grants and personal fees from a variety of sources.
A version of this article first appeared on Medscape.com.
FROM JAMA
Why people lie about COVID
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
Have you ever lied about COVID-19?
Before you get upset, before the “how dare you,” I want you to think carefully.
Did you have COVID-19 (or think you did) and not mention it to someone you were going to be with? Did you tell someone you were taking more COVID precautions than you really were? Did you tell someone you were vaccinated when you weren’t? Have you avoided getting a COVID test even though you knew you should have?
Researchers appreciated the fact that public health interventions in COVID are important but are only as good as the percentage of people who actually abide by them. So, they designed a survey to ask the questions that many people don’t want to hear the answer to.
A total of 1,733 participants – 80% of those invited – responded to the survey. By design, approximately one-third of respondents (477) had already had COVID, one-third (499) were vaccinated and not yet infected, and one-third (509) were unvaccinated and not yet infected.
Of those surveyed, 41.6% admitted that they lied about COVID or didn’t adhere to COVID guidelines - a conservative estimate, if you ask me.
Breaking down some of the results, about 20% of people who previously were infected with COVID said they didn’t mention it when meeting with someone. A similar number said they didn’t tell anyone when they were entering a public place. A bit more concerning to me, roughly 20% reported not disclosing their COVID-positive status when going to a health care provider’s office.
About 10% of those who had not been vaccinated reported lying about their vaccination status. That’s actually less than the 15% of vaccinated people who lied and told someone they weren’t vaccinated.
About 17% of people lied about the need to quarantine, and many more broke quarantine rules.
The authors tried to see if certain personal characteristics predicted people who were more likely to lie about COVID-19–related issues. Turns out there was only one thing that predicted honesty: age.
Older people were more honest about their COVID status and COVID habits. Other factors – gender, education, race, political affiliation, COVID-19 conspiracy beliefs, and where you got your COVID information – did not seem to make much of a difference. Why are older people more honest? Because older people take COVID more seriously. And they should; COVID is more severe in older people.
The problem arises, of course, because people who are at lower risk for COVID complications interact with people at higher risk – and in those situations, honesty matters more.
On the other hand, isn’t lying about COVID stuff inevitable? If you know that a positive test means you can’t go to work, and not going to work means you won’t get paid, might you not be more likely to lie about the test? Or not get the test at all?
The authors explored the reasons for dishonesty and they are fairly broad, ranging from the desire for life to feel normal (more than half of people who lied) to not believing that COVID was real (a whopping 30%). Some of the reasons for lying included:
- Wanted life to feel normal (50%).
- Freedom (45%).
- It’s no one’s business (40%).
- COVID isn’t real (30%).
In the end, though, we need to realize that public health recommendations are not going to be universally followed, and people may tell us they are following them when, in fact, they are not.
What this adds is another data point to a trend we’ve seen across the course of the pandemic, a shift from collective to individual responsibility. If you can’t be sure what others are doing in regard to COVID, you need to focus on protecting yourself. Perhaps that shift was inevitable. Doesn’t mean we have to like it.
A version of this article first appeared on Medscape.com.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here on Medscape. He tweets @fperrywilson and hosts a repository of his communication work at www.methodsman.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
Have you ever lied about COVID-19?
Before you get upset, before the “how dare you,” I want you to think carefully.
Did you have COVID-19 (or think you did) and not mention it to someone you were going to be with? Did you tell someone you were taking more COVID precautions than you really were? Did you tell someone you were vaccinated when you weren’t? Have you avoided getting a COVID test even though you knew you should have?
Researchers appreciated the fact that public health interventions in COVID are important but are only as good as the percentage of people who actually abide by them. So, they designed a survey to ask the questions that many people don’t want to hear the answer to.
A total of 1,733 participants – 80% of those invited – responded to the survey. By design, approximately one-third of respondents (477) had already had COVID, one-third (499) were vaccinated and not yet infected, and one-third (509) were unvaccinated and not yet infected.
Of those surveyed, 41.6% admitted that they lied about COVID or didn’t adhere to COVID guidelines - a conservative estimate, if you ask me.
Breaking down some of the results, about 20% of people who previously were infected with COVID said they didn’t mention it when meeting with someone. A similar number said they didn’t tell anyone when they were entering a public place. A bit more concerning to me, roughly 20% reported not disclosing their COVID-positive status when going to a health care provider’s office.
About 10% of those who had not been vaccinated reported lying about their vaccination status. That’s actually less than the 15% of vaccinated people who lied and told someone they weren’t vaccinated.
About 17% of people lied about the need to quarantine, and many more broke quarantine rules.
The authors tried to see if certain personal characteristics predicted people who were more likely to lie about COVID-19–related issues. Turns out there was only one thing that predicted honesty: age.
Older people were more honest about their COVID status and COVID habits. Other factors – gender, education, race, political affiliation, COVID-19 conspiracy beliefs, and where you got your COVID information – did not seem to make much of a difference. Why are older people more honest? Because older people take COVID more seriously. And they should; COVID is more severe in older people.
The problem arises, of course, because people who are at lower risk for COVID complications interact with people at higher risk – and in those situations, honesty matters more.
On the other hand, isn’t lying about COVID stuff inevitable? If you know that a positive test means you can’t go to work, and not going to work means you won’t get paid, might you not be more likely to lie about the test? Or not get the test at all?
The authors explored the reasons for dishonesty and they are fairly broad, ranging from the desire for life to feel normal (more than half of people who lied) to not believing that COVID was real (a whopping 30%). Some of the reasons for lying included:
- Wanted life to feel normal (50%).
- Freedom (45%).
- It’s no one’s business (40%).
- COVID isn’t real (30%).
In the end, though, we need to realize that public health recommendations are not going to be universally followed, and people may tell us they are following them when, in fact, they are not.
What this adds is another data point to a trend we’ve seen across the course of the pandemic, a shift from collective to individual responsibility. If you can’t be sure what others are doing in regard to COVID, you need to focus on protecting yourself. Perhaps that shift was inevitable. Doesn’t mean we have to like it.
A version of this article first appeared on Medscape.com.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here on Medscape. He tweets @fperrywilson and hosts a repository of his communication work at www.methodsman.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
Have you ever lied about COVID-19?
Before you get upset, before the “how dare you,” I want you to think carefully.
Did you have COVID-19 (or think you did) and not mention it to someone you were going to be with? Did you tell someone you were taking more COVID precautions than you really were? Did you tell someone you were vaccinated when you weren’t? Have you avoided getting a COVID test even though you knew you should have?
Researchers appreciated the fact that public health interventions in COVID are important but are only as good as the percentage of people who actually abide by them. So, they designed a survey to ask the questions that many people don’t want to hear the answer to.
A total of 1,733 participants – 80% of those invited – responded to the survey. By design, approximately one-third of respondents (477) had already had COVID, one-third (499) were vaccinated and not yet infected, and one-third (509) were unvaccinated and not yet infected.
Of those surveyed, 41.6% admitted that they lied about COVID or didn’t adhere to COVID guidelines - a conservative estimate, if you ask me.
Breaking down some of the results, about 20% of people who previously were infected with COVID said they didn’t mention it when meeting with someone. A similar number said they didn’t tell anyone when they were entering a public place. A bit more concerning to me, roughly 20% reported not disclosing their COVID-positive status when going to a health care provider’s office.
About 10% of those who had not been vaccinated reported lying about their vaccination status. That’s actually less than the 15% of vaccinated people who lied and told someone they weren’t vaccinated.
About 17% of people lied about the need to quarantine, and many more broke quarantine rules.
The authors tried to see if certain personal characteristics predicted people who were more likely to lie about COVID-19–related issues. Turns out there was only one thing that predicted honesty: age.
Older people were more honest about their COVID status and COVID habits. Other factors – gender, education, race, political affiliation, COVID-19 conspiracy beliefs, and where you got your COVID information – did not seem to make much of a difference. Why are older people more honest? Because older people take COVID more seriously. And they should; COVID is more severe in older people.
The problem arises, of course, because people who are at lower risk for COVID complications interact with people at higher risk – and in those situations, honesty matters more.
On the other hand, isn’t lying about COVID stuff inevitable? If you know that a positive test means you can’t go to work, and not going to work means you won’t get paid, might you not be more likely to lie about the test? Or not get the test at all?
The authors explored the reasons for dishonesty and they are fairly broad, ranging from the desire for life to feel normal (more than half of people who lied) to not believing that COVID was real (a whopping 30%). Some of the reasons for lying included:
- Wanted life to feel normal (50%).
- Freedom (45%).
- It’s no one’s business (40%).
- COVID isn’t real (30%).
In the end, though, we need to realize that public health recommendations are not going to be universally followed, and people may tell us they are following them when, in fact, they are not.
What this adds is another data point to a trend we’ve seen across the course of the pandemic, a shift from collective to individual responsibility. If you can’t be sure what others are doing in regard to COVID, you need to focus on protecting yourself. Perhaps that shift was inevitable. Doesn’t mean we have to like it.
A version of this article first appeared on Medscape.com.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here on Medscape. He tweets @fperrywilson and hosts a repository of his communication work at www.methodsman.com.
Epidemic of brain fog? Long COVID’s effects worry experts
Weeks after Jeannie Volpe caught COVID-19 in November 2020, she could no longer do her job running sexual assault support groups in Anniston, Ala., because she kept forgetting the details that survivors had shared with her. “People were telling me they were having to revisit their traumatic memories, which isn’t fair to anybody,” the 47-year-old says.
Ms. Volpe has been diagnosed with long-COVID autonomic dysfunction, which includes severe muscle pain, depression, anxiety, and a loss of thinking skills. Some of her symptoms are more commonly known as brain fog, and they’re among the most frequent problems reported by people who have long-term issues after a bout of COVID-19.
Many experts and medical professionals say they haven’t even begun to scratch the surface of what impact this will have in years to come.
“I’m very worried that we have an epidemic of neurologic dysfunction coming down the pike,” says Pamela Davis, MD, PhD, a research professor at Case Western Reserve University, Cleveland.
In the 2 years Ms. Volpe has been living with long COVID, her executive function – the mental processes that enable people to focus attention, retain information, and multitask – has been so diminished that she had to relearn to drive. One of the various doctors assessing her has suggested speech therapy to help Ms. Volpe relearn how to form words. “I can see the words I want to say in my mind, but I can’t make them come out of my mouth,” she says in a sluggish voice that gives away her condition.
All of those symptoms make it difficult for her to care for herself. Without a job and health insurance, Ms. Volpe says she’s researched assisted suicide in the states that allow it but has ultimately decided she wants to live.
“People tell you things like you should be grateful you survived it, and you should; but you shouldn’t expect somebody to not grieve after losing their autonomy, their career, their finances.”
The findings of researchers studying the brain effects of COVID-19 reinforce what people with long COVID have been dealing with from the start. Their experiences aren’t imaginary; they’re consistent with neurological disorders – including myalgic encephalomyelitis, also known as chronic fatigue syndrome, or ME/CFS – which carry much more weight in the public imagination than the term brain fog, which can often be used dismissively.
Studies have found that COVID-19 is linked to conditions such as strokes; seizures; and mood, memory, and movement disorders.
While there are still a lot of unanswered questions about exactly how COVID-19 affects the brain and what the long-term effects are, there’s enough reason to suggest people should be trying to avoid both infection and reinfection until researchers get more answers.
Worldwide, it’s estimated that COVID-19 has contributed to more than 40 million new cases of neurological disorders, says Ziyad Al-Aly, MD, a clinical epidemiologist and long COVID researcher at Washington University in St. Louis. In his latest study of 14 million medical records of the U.S. Department of Veterans Affairs, the country’s largest integrated health care system, researchers found that regardless of age, gender, race, and lifestyle,
He noted that some of the conditions, such as headaches and mild decline in memory and sharpness, may improve and go away over time. But others that showed up, such as stroke, encephalitis (inflammation of the brain), and Guillain-Barré syndrome (a rare disorder in which the body’s immune system attacks the nerves), often lead to lasting damage. Dr. Al-Aly’s team found that neurological conditions were 7% more likely in those who had COVID-19 than in those who had never been infected.
What’s more, researchers noticed that compared with control groups, the risk of post-COVID thinking problems was more pronounced in people in their 30s, 40s, and 50s – a group that usually would be very unlikely to have these problems. For those over the age of 60, the risks stood out less because at that stage of life, such thinking problems aren’t as rare.
Another study of the veterans system last year showed that COVID-19 survivors were at a 46% higher risk of considering suicide after 1 year.
“We need to be paying attention to this,” says Dr. Al-Aly. “What we’ve seen is really the tip of the iceberg.” He worries that millions of people, including youths, will lose out on employment and education while dealing with long-term disabilities – and the economic and societal implications of such a fallout. “What we will all be left with is the aftermath of sheer devastation in some people’s lives,” he says.
Igor Koralnik, MD, chief of neuro-infectious disease and global neurology at Northwestern University, Chicago, has been running a specialized long COVID clinic. His team published a paper in March 2021 detailing what they saw in their first 100 patients. “About half the population in the study missed at least 10 days of work. This is going to have persistent impact on the workforce,” Dr. Koralnik said in a podcast posted on the Northwestern website. “We have seen that not only [do] patients have symptoms, but they have decreased quality of life.”
For older people and their caregivers, the risk of potential neurodegenerative diseases that the virus has shown to accelerate, such as dementia, is also a big concern. Alzheimer’s is already the fifth leading cause of death for people 65 and older.
In a recent study of more than 6 million people over the age of 65, Dr. Davis and her team at Case Western found the risk of Alzheimer’s in the year after COVID-19 increased by 50%-80%. The chances were especially high for women older than 85.
To date, there are no good treatments for Alzheimer’s, yet total health care costs for long-term care and hospice services for people with dementia topped $300 billion in 2020. That doesn’t even include the related costs to families.
“The downstream effect of having someone with Alzheimer’s being taken care of by a family member can be devastating on everyone,” she says. “Sometimes the caregivers don’t weather that very well.”
When Dr. Davis’s own father got Alzheimer’s at age 86, her mother took care of him until she had a stroke one morning while making breakfast. Dr. Davis attributes the stroke to the stress of caregiving. That left Dr. Davis no choice but to seek housing where both her parents could get care.
Looking at the broader picture, Dr. Davis believes widespread isolation, loneliness, and grief during the pandemic, and the disease of COVID-19 itself, will continue to have a profound impact on psychiatric diagnoses. This in turn could trigger a wave of new substance abuse as a result of unchecked mental health problems.
Still, not all brain experts are jumping to worst-case scenarios, with a lot yet to be understood before sounding the alarm. Joanna Hellmuth, MD, a neurologist and researcher at the University of California, San Francisco, cautions against reading too much into early data, including any assumptions that COVID-19 causes neurodegeneration or irreversible damage in the brain.
Even with before-and-after brain scans by University of Oxford, England, researchers that show structural changes to the brain after infection, she points out that they didn’t actually study the clinical symptoms of the people in the study, so it’s too soon to reach conclusions about associated cognitive problems.
“It’s an important piece of the puzzle, but we don’t know how that fits together with everything else,” says Dr. Hellmuth. “Some of my patients get better. … I haven’t seen a single person get worse since the pandemic started, and so I’m hopeful.”
A version of this article first appeared on WebMD.com.
Weeks after Jeannie Volpe caught COVID-19 in November 2020, she could no longer do her job running sexual assault support groups in Anniston, Ala., because she kept forgetting the details that survivors had shared with her. “People were telling me they were having to revisit their traumatic memories, which isn’t fair to anybody,” the 47-year-old says.
Ms. Volpe has been diagnosed with long-COVID autonomic dysfunction, which includes severe muscle pain, depression, anxiety, and a loss of thinking skills. Some of her symptoms are more commonly known as brain fog, and they’re among the most frequent problems reported by people who have long-term issues after a bout of COVID-19.
Many experts and medical professionals say they haven’t even begun to scratch the surface of what impact this will have in years to come.
“I’m very worried that we have an epidemic of neurologic dysfunction coming down the pike,” says Pamela Davis, MD, PhD, a research professor at Case Western Reserve University, Cleveland.
In the 2 years Ms. Volpe has been living with long COVID, her executive function – the mental processes that enable people to focus attention, retain information, and multitask – has been so diminished that she had to relearn to drive. One of the various doctors assessing her has suggested speech therapy to help Ms. Volpe relearn how to form words. “I can see the words I want to say in my mind, but I can’t make them come out of my mouth,” she says in a sluggish voice that gives away her condition.
All of those symptoms make it difficult for her to care for herself. Without a job and health insurance, Ms. Volpe says she’s researched assisted suicide in the states that allow it but has ultimately decided she wants to live.
“People tell you things like you should be grateful you survived it, and you should; but you shouldn’t expect somebody to not grieve after losing their autonomy, their career, their finances.”
The findings of researchers studying the brain effects of COVID-19 reinforce what people with long COVID have been dealing with from the start. Their experiences aren’t imaginary; they’re consistent with neurological disorders – including myalgic encephalomyelitis, also known as chronic fatigue syndrome, or ME/CFS – which carry much more weight in the public imagination than the term brain fog, which can often be used dismissively.
Studies have found that COVID-19 is linked to conditions such as strokes; seizures; and mood, memory, and movement disorders.
While there are still a lot of unanswered questions about exactly how COVID-19 affects the brain and what the long-term effects are, there’s enough reason to suggest people should be trying to avoid both infection and reinfection until researchers get more answers.
Worldwide, it’s estimated that COVID-19 has contributed to more than 40 million new cases of neurological disorders, says Ziyad Al-Aly, MD, a clinical epidemiologist and long COVID researcher at Washington University in St. Louis. In his latest study of 14 million medical records of the U.S. Department of Veterans Affairs, the country’s largest integrated health care system, researchers found that regardless of age, gender, race, and lifestyle,
He noted that some of the conditions, such as headaches and mild decline in memory and sharpness, may improve and go away over time. But others that showed up, such as stroke, encephalitis (inflammation of the brain), and Guillain-Barré syndrome (a rare disorder in which the body’s immune system attacks the nerves), often lead to lasting damage. Dr. Al-Aly’s team found that neurological conditions were 7% more likely in those who had COVID-19 than in those who had never been infected.
What’s more, researchers noticed that compared with control groups, the risk of post-COVID thinking problems was more pronounced in people in their 30s, 40s, and 50s – a group that usually would be very unlikely to have these problems. For those over the age of 60, the risks stood out less because at that stage of life, such thinking problems aren’t as rare.
Another study of the veterans system last year showed that COVID-19 survivors were at a 46% higher risk of considering suicide after 1 year.
“We need to be paying attention to this,” says Dr. Al-Aly. “What we’ve seen is really the tip of the iceberg.” He worries that millions of people, including youths, will lose out on employment and education while dealing with long-term disabilities – and the economic and societal implications of such a fallout. “What we will all be left with is the aftermath of sheer devastation in some people’s lives,” he says.
Igor Koralnik, MD, chief of neuro-infectious disease and global neurology at Northwestern University, Chicago, has been running a specialized long COVID clinic. His team published a paper in March 2021 detailing what they saw in their first 100 patients. “About half the population in the study missed at least 10 days of work. This is going to have persistent impact on the workforce,” Dr. Koralnik said in a podcast posted on the Northwestern website. “We have seen that not only [do] patients have symptoms, but they have decreased quality of life.”
For older people and their caregivers, the risk of potential neurodegenerative diseases that the virus has shown to accelerate, such as dementia, is also a big concern. Alzheimer’s is already the fifth leading cause of death for people 65 and older.
In a recent study of more than 6 million people over the age of 65, Dr. Davis and her team at Case Western found the risk of Alzheimer’s in the year after COVID-19 increased by 50%-80%. The chances were especially high for women older than 85.
To date, there are no good treatments for Alzheimer’s, yet total health care costs for long-term care and hospice services for people with dementia topped $300 billion in 2020. That doesn’t even include the related costs to families.
“The downstream effect of having someone with Alzheimer’s being taken care of by a family member can be devastating on everyone,” she says. “Sometimes the caregivers don’t weather that very well.”
When Dr. Davis’s own father got Alzheimer’s at age 86, her mother took care of him until she had a stroke one morning while making breakfast. Dr. Davis attributes the stroke to the stress of caregiving. That left Dr. Davis no choice but to seek housing where both her parents could get care.
Looking at the broader picture, Dr. Davis believes widespread isolation, loneliness, and grief during the pandemic, and the disease of COVID-19 itself, will continue to have a profound impact on psychiatric diagnoses. This in turn could trigger a wave of new substance abuse as a result of unchecked mental health problems.
Still, not all brain experts are jumping to worst-case scenarios, with a lot yet to be understood before sounding the alarm. Joanna Hellmuth, MD, a neurologist and researcher at the University of California, San Francisco, cautions against reading too much into early data, including any assumptions that COVID-19 causes neurodegeneration or irreversible damage in the brain.
Even with before-and-after brain scans by University of Oxford, England, researchers that show structural changes to the brain after infection, she points out that they didn’t actually study the clinical symptoms of the people in the study, so it’s too soon to reach conclusions about associated cognitive problems.
“It’s an important piece of the puzzle, but we don’t know how that fits together with everything else,” says Dr. Hellmuth. “Some of my patients get better. … I haven’t seen a single person get worse since the pandemic started, and so I’m hopeful.”
A version of this article first appeared on WebMD.com.
Weeks after Jeannie Volpe caught COVID-19 in November 2020, she could no longer do her job running sexual assault support groups in Anniston, Ala., because she kept forgetting the details that survivors had shared with her. “People were telling me they were having to revisit their traumatic memories, which isn’t fair to anybody,” the 47-year-old says.
Ms. Volpe has been diagnosed with long-COVID autonomic dysfunction, which includes severe muscle pain, depression, anxiety, and a loss of thinking skills. Some of her symptoms are more commonly known as brain fog, and they’re among the most frequent problems reported by people who have long-term issues after a bout of COVID-19.
Many experts and medical professionals say they haven’t even begun to scratch the surface of what impact this will have in years to come.
“I’m very worried that we have an epidemic of neurologic dysfunction coming down the pike,” says Pamela Davis, MD, PhD, a research professor at Case Western Reserve University, Cleveland.
In the 2 years Ms. Volpe has been living with long COVID, her executive function – the mental processes that enable people to focus attention, retain information, and multitask – has been so diminished that she had to relearn to drive. One of the various doctors assessing her has suggested speech therapy to help Ms. Volpe relearn how to form words. “I can see the words I want to say in my mind, but I can’t make them come out of my mouth,” she says in a sluggish voice that gives away her condition.
All of those symptoms make it difficult for her to care for herself. Without a job and health insurance, Ms. Volpe says she’s researched assisted suicide in the states that allow it but has ultimately decided she wants to live.
“People tell you things like you should be grateful you survived it, and you should; but you shouldn’t expect somebody to not grieve after losing their autonomy, their career, their finances.”
The findings of researchers studying the brain effects of COVID-19 reinforce what people with long COVID have been dealing with from the start. Their experiences aren’t imaginary; they’re consistent with neurological disorders – including myalgic encephalomyelitis, also known as chronic fatigue syndrome, or ME/CFS – which carry much more weight in the public imagination than the term brain fog, which can often be used dismissively.
Studies have found that COVID-19 is linked to conditions such as strokes; seizures; and mood, memory, and movement disorders.
While there are still a lot of unanswered questions about exactly how COVID-19 affects the brain and what the long-term effects are, there’s enough reason to suggest people should be trying to avoid both infection and reinfection until researchers get more answers.
Worldwide, it’s estimated that COVID-19 has contributed to more than 40 million new cases of neurological disorders, says Ziyad Al-Aly, MD, a clinical epidemiologist and long COVID researcher at Washington University in St. Louis. In his latest study of 14 million medical records of the U.S. Department of Veterans Affairs, the country’s largest integrated health care system, researchers found that regardless of age, gender, race, and lifestyle,
He noted that some of the conditions, such as headaches and mild decline in memory and sharpness, may improve and go away over time. But others that showed up, such as stroke, encephalitis (inflammation of the brain), and Guillain-Barré syndrome (a rare disorder in which the body’s immune system attacks the nerves), often lead to lasting damage. Dr. Al-Aly’s team found that neurological conditions were 7% more likely in those who had COVID-19 than in those who had never been infected.
What’s more, researchers noticed that compared with control groups, the risk of post-COVID thinking problems was more pronounced in people in their 30s, 40s, and 50s – a group that usually would be very unlikely to have these problems. For those over the age of 60, the risks stood out less because at that stage of life, such thinking problems aren’t as rare.
Another study of the veterans system last year showed that COVID-19 survivors were at a 46% higher risk of considering suicide after 1 year.
“We need to be paying attention to this,” says Dr. Al-Aly. “What we’ve seen is really the tip of the iceberg.” He worries that millions of people, including youths, will lose out on employment and education while dealing with long-term disabilities – and the economic and societal implications of such a fallout. “What we will all be left with is the aftermath of sheer devastation in some people’s lives,” he says.
Igor Koralnik, MD, chief of neuro-infectious disease and global neurology at Northwestern University, Chicago, has been running a specialized long COVID clinic. His team published a paper in March 2021 detailing what they saw in their first 100 patients. “About half the population in the study missed at least 10 days of work. This is going to have persistent impact on the workforce,” Dr. Koralnik said in a podcast posted on the Northwestern website. “We have seen that not only [do] patients have symptoms, but they have decreased quality of life.”
For older people and their caregivers, the risk of potential neurodegenerative diseases that the virus has shown to accelerate, such as dementia, is also a big concern. Alzheimer’s is already the fifth leading cause of death for people 65 and older.
In a recent study of more than 6 million people over the age of 65, Dr. Davis and her team at Case Western found the risk of Alzheimer’s in the year after COVID-19 increased by 50%-80%. The chances were especially high for women older than 85.
To date, there are no good treatments for Alzheimer’s, yet total health care costs for long-term care and hospice services for people with dementia topped $300 billion in 2020. That doesn’t even include the related costs to families.
“The downstream effect of having someone with Alzheimer’s being taken care of by a family member can be devastating on everyone,” she says. “Sometimes the caregivers don’t weather that very well.”
When Dr. Davis’s own father got Alzheimer’s at age 86, her mother took care of him until she had a stroke one morning while making breakfast. Dr. Davis attributes the stroke to the stress of caregiving. That left Dr. Davis no choice but to seek housing where both her parents could get care.
Looking at the broader picture, Dr. Davis believes widespread isolation, loneliness, and grief during the pandemic, and the disease of COVID-19 itself, will continue to have a profound impact on psychiatric diagnoses. This in turn could trigger a wave of new substance abuse as a result of unchecked mental health problems.
Still, not all brain experts are jumping to worst-case scenarios, with a lot yet to be understood before sounding the alarm. Joanna Hellmuth, MD, a neurologist and researcher at the University of California, San Francisco, cautions against reading too much into early data, including any assumptions that COVID-19 causes neurodegeneration or irreversible damage in the brain.
Even with before-and-after brain scans by University of Oxford, England, researchers that show structural changes to the brain after infection, she points out that they didn’t actually study the clinical symptoms of the people in the study, so it’s too soon to reach conclusions about associated cognitive problems.
“It’s an important piece of the puzzle, but we don’t know how that fits together with everything else,” says Dr. Hellmuth. “Some of my patients get better. … I haven’t seen a single person get worse since the pandemic started, and so I’m hopeful.”
A version of this article first appeared on WebMD.com.
At the front lines of long COVID, local clinics prove vital
Big-name hospital chains across the United States are opening dedicated centers to help patients dealing with long COVID. But so are the lower-profile clinics and hospitals run by cities, counties and states – including Harborview Medical Center in Seattle.
They serve areas ranging from Campbell County, Wyo., with 47,000 residents, to New York City, with its 8.4 million people. Many providers working there are searching for innovative ways to approach this lingering illness with its variety of symptoms, from brain fog to shortness of breath to depression and more.
Their efforts often fall below the radar, with still-scant serious media attention to long COVID or the public health employees working to treat ailing patients.
Why are state and local health agencies taking on these duties?
They’re leading the way in part because the federal government has made only limited efforts, said Lisa McCorkell, a cofounder of the Patient-Led Research Collaborative. The international group was founded in spring 2020 by researchers who are also long COVID patients.
“It’s a big reason why long COVID isn’t talked about as much,” Ms. McCorkell said. “It’s definitely a national issue. But it trickles down to state and local health departments, and there’s not enough resources.”
The government clinics may be accessible to people without insurance and often are cheaper than clinics at private hospitals.
Harborview has treated more than 1,000 patients with long COVID, and another 200 patients are awaiting treatment, said Jessica Bender, MD, a codirector of the University of Washington Post-COVID Rehabilitation and Recovery Clinic in Seattle’s First Hill neighborhood.
The group Survivor Corps offers lists by states of clinics. While the publicly run clinics may be less expensive or even free for some patients, methods of payment vary from clinic to clinic. Federally qualified health clinics offer treatment on a sliding scale. For instance, the Riverside University Health System in California has federally qualified centers. And other providers who are not federally qualified also offer care paid for on a sliding scale. They include Campbell County Health, where some residents are eligible for discounts of 25%-100%, said spokesperson Norberto Orellana.
At Harborview, Dr. Bender said the public hospital’s post-COVID clinic initially began with a staff of rehabilitation doctors but expanded in 2021 to include family and internal medicine doctors. And it offers mental health programs with rehabilitation psychologists who instruct on how to deal with doctors or loved ones who don’t believe that long COVID exists.
“I have patients who really have been devastated by the lack of support from coworkers [and] family,” Dr. Bender said.
In Campbell County, Wyo., the pandemic surge did not arrive in earnest until late 2021. Physical therapists at Campbell County’s Health Rehabilitation Services organized a rehabilitation program for residents with long COVID after recognizing the need, said Shannon Sorensen, rehabilitation director at Campbell County Health.
“We had patients coming in showing chest pain, or heart palpitations. There were people trying to get back to work. They were frustrated,” Ms. Sorensen said.
Myalgic encephalomyelitis and chronic fatigue syndrome activists have embraced the fight to recognize and help long COVID patients, noting the similarities between the conditions, and hope to help kickstart more organized research, treatment and benefits for long COVID sufferers and myalgic encephalomyelitis/chronic fatigue syndrome patients alike.
In Ft. Collins, Colo., disability activist Alison Sbrana has long had myalgic encephalomyelitis. She and other members of the local chapter of ME Action have met with state officials for several years and are finally seeing the results of those efforts.
Colorado Gov. Jared Polis has created the full-time position of policy adviser for long COVID and post–viral infection planning.
“This is one way forward of how state governments are (finally) paying attention to infection-triggered chronic illnesses and starting to think ahead on them,” Ms. Sbrana said.
New York City’s Health + Hospitals launched what may be the most expansive long COVID treatment program in the nation in April 2021. Called AfterCare, it provides physical and mental health services as well as community support systems and financial assistance.
A persistent issue for patients is that there isn’t yet a test for long COVID, like there is for COVID-19, said Amanda Johnson, MD, assistant vice president for ambulatory care and population health at New York Health + Hospitals. “It’s in many ways a diagnosis of exclusion. You have to make sure their shortness of breath isn’t caused by something else. The same with anemia,” she said.
California’s Department of Public Health has a detailed website devoted to the topic, including videos of “long haulers” describing their experiences.
Vermont is one of several states studying long COVID, said Mark Levine, MD, the state health commissioner. The state, in collaboration with the University of Vermont, has established a surveillance project to determine how many people have long COVID, as well as how severe it is, how long it lasts, and potential predispositions.
The University of Utah, Salt Lake City, established a comprehensive COVID-19 clinic more than a year ago that also handles long COVID patients, said Jeannette Brown, MD, PhD, an associate professor at the school and director of the COVID-19 clinic.
Jennifer Chevinsky, MD, MPH, already had a deep understanding of long COVID when she landed in Riverside County, Calif., in the summer of 2021. She came from Atlanta, where as part of her job as an epidemic intelligence service officer at the CDC, she heard stories of COVID-19 patients who were not getting better.
Now she is a deputy public health officer for Riverside County, in a region known for its deserts, sizzling summer temperatures and diverse populations. She said her department has helped launch programs such as post–COVID-19 follow-up phone calls and long COVID training programs that reach out to the many Latino residents in this county of 2.4 million people. It also includes Black and Native American residents.
“We’re making sure information is circulated with community and faith-based organizations, and community health workers,” she said.
Ms. McCorkell said there is still much work to do to raise public awareness of the risks of long COVID and how to obtain care for patients. She would like to see a national public health campaign about long COVID, possibly spearheaded by the Centers for Disease Control and Prevention in partnership with local health workers and community-based organizations.
“That,” she said, “could make a big difference.”
A version of this article first appeared on WebMD.com.
Big-name hospital chains across the United States are opening dedicated centers to help patients dealing with long COVID. But so are the lower-profile clinics and hospitals run by cities, counties and states – including Harborview Medical Center in Seattle.
They serve areas ranging from Campbell County, Wyo., with 47,000 residents, to New York City, with its 8.4 million people. Many providers working there are searching for innovative ways to approach this lingering illness with its variety of symptoms, from brain fog to shortness of breath to depression and more.
Their efforts often fall below the radar, with still-scant serious media attention to long COVID or the public health employees working to treat ailing patients.
Why are state and local health agencies taking on these duties?
They’re leading the way in part because the federal government has made only limited efforts, said Lisa McCorkell, a cofounder of the Patient-Led Research Collaborative. The international group was founded in spring 2020 by researchers who are also long COVID patients.
“It’s a big reason why long COVID isn’t talked about as much,” Ms. McCorkell said. “It’s definitely a national issue. But it trickles down to state and local health departments, and there’s not enough resources.”
The government clinics may be accessible to people without insurance and often are cheaper than clinics at private hospitals.
Harborview has treated more than 1,000 patients with long COVID, and another 200 patients are awaiting treatment, said Jessica Bender, MD, a codirector of the University of Washington Post-COVID Rehabilitation and Recovery Clinic in Seattle’s First Hill neighborhood.
The group Survivor Corps offers lists by states of clinics. While the publicly run clinics may be less expensive or even free for some patients, methods of payment vary from clinic to clinic. Federally qualified health clinics offer treatment on a sliding scale. For instance, the Riverside University Health System in California has federally qualified centers. And other providers who are not federally qualified also offer care paid for on a sliding scale. They include Campbell County Health, where some residents are eligible for discounts of 25%-100%, said spokesperson Norberto Orellana.
At Harborview, Dr. Bender said the public hospital’s post-COVID clinic initially began with a staff of rehabilitation doctors but expanded in 2021 to include family and internal medicine doctors. And it offers mental health programs with rehabilitation psychologists who instruct on how to deal with doctors or loved ones who don’t believe that long COVID exists.
“I have patients who really have been devastated by the lack of support from coworkers [and] family,” Dr. Bender said.
In Campbell County, Wyo., the pandemic surge did not arrive in earnest until late 2021. Physical therapists at Campbell County’s Health Rehabilitation Services organized a rehabilitation program for residents with long COVID after recognizing the need, said Shannon Sorensen, rehabilitation director at Campbell County Health.
“We had patients coming in showing chest pain, or heart palpitations. There were people trying to get back to work. They were frustrated,” Ms. Sorensen said.
Myalgic encephalomyelitis and chronic fatigue syndrome activists have embraced the fight to recognize and help long COVID patients, noting the similarities between the conditions, and hope to help kickstart more organized research, treatment and benefits for long COVID sufferers and myalgic encephalomyelitis/chronic fatigue syndrome patients alike.
In Ft. Collins, Colo., disability activist Alison Sbrana has long had myalgic encephalomyelitis. She and other members of the local chapter of ME Action have met with state officials for several years and are finally seeing the results of those efforts.
Colorado Gov. Jared Polis has created the full-time position of policy adviser for long COVID and post–viral infection planning.
“This is one way forward of how state governments are (finally) paying attention to infection-triggered chronic illnesses and starting to think ahead on them,” Ms. Sbrana said.
New York City’s Health + Hospitals launched what may be the most expansive long COVID treatment program in the nation in April 2021. Called AfterCare, it provides physical and mental health services as well as community support systems and financial assistance.
A persistent issue for patients is that there isn’t yet a test for long COVID, like there is for COVID-19, said Amanda Johnson, MD, assistant vice president for ambulatory care and population health at New York Health + Hospitals. “It’s in many ways a diagnosis of exclusion. You have to make sure their shortness of breath isn’t caused by something else. The same with anemia,” she said.
California’s Department of Public Health has a detailed website devoted to the topic, including videos of “long haulers” describing their experiences.
Vermont is one of several states studying long COVID, said Mark Levine, MD, the state health commissioner. The state, in collaboration with the University of Vermont, has established a surveillance project to determine how many people have long COVID, as well as how severe it is, how long it lasts, and potential predispositions.
The University of Utah, Salt Lake City, established a comprehensive COVID-19 clinic more than a year ago that also handles long COVID patients, said Jeannette Brown, MD, PhD, an associate professor at the school and director of the COVID-19 clinic.
Jennifer Chevinsky, MD, MPH, already had a deep understanding of long COVID when she landed in Riverside County, Calif., in the summer of 2021. She came from Atlanta, where as part of her job as an epidemic intelligence service officer at the CDC, she heard stories of COVID-19 patients who were not getting better.
Now she is a deputy public health officer for Riverside County, in a region known for its deserts, sizzling summer temperatures and diverse populations. She said her department has helped launch programs such as post–COVID-19 follow-up phone calls and long COVID training programs that reach out to the many Latino residents in this county of 2.4 million people. It also includes Black and Native American residents.
“We’re making sure information is circulated with community and faith-based organizations, and community health workers,” she said.
Ms. McCorkell said there is still much work to do to raise public awareness of the risks of long COVID and how to obtain care for patients. She would like to see a national public health campaign about long COVID, possibly spearheaded by the Centers for Disease Control and Prevention in partnership with local health workers and community-based organizations.
“That,” she said, “could make a big difference.”
A version of this article first appeared on WebMD.com.
Big-name hospital chains across the United States are opening dedicated centers to help patients dealing with long COVID. But so are the lower-profile clinics and hospitals run by cities, counties and states – including Harborview Medical Center in Seattle.
They serve areas ranging from Campbell County, Wyo., with 47,000 residents, to New York City, with its 8.4 million people. Many providers working there are searching for innovative ways to approach this lingering illness with its variety of symptoms, from brain fog to shortness of breath to depression and more.
Their efforts often fall below the radar, with still-scant serious media attention to long COVID or the public health employees working to treat ailing patients.
Why are state and local health agencies taking on these duties?
They’re leading the way in part because the federal government has made only limited efforts, said Lisa McCorkell, a cofounder of the Patient-Led Research Collaborative. The international group was founded in spring 2020 by researchers who are also long COVID patients.
“It’s a big reason why long COVID isn’t talked about as much,” Ms. McCorkell said. “It’s definitely a national issue. But it trickles down to state and local health departments, and there’s not enough resources.”
The government clinics may be accessible to people without insurance and often are cheaper than clinics at private hospitals.
Harborview has treated more than 1,000 patients with long COVID, and another 200 patients are awaiting treatment, said Jessica Bender, MD, a codirector of the University of Washington Post-COVID Rehabilitation and Recovery Clinic in Seattle’s First Hill neighborhood.
The group Survivor Corps offers lists by states of clinics. While the publicly run clinics may be less expensive or even free for some patients, methods of payment vary from clinic to clinic. Federally qualified health clinics offer treatment on a sliding scale. For instance, the Riverside University Health System in California has federally qualified centers. And other providers who are not federally qualified also offer care paid for on a sliding scale. They include Campbell County Health, where some residents are eligible for discounts of 25%-100%, said spokesperson Norberto Orellana.
At Harborview, Dr. Bender said the public hospital’s post-COVID clinic initially began with a staff of rehabilitation doctors but expanded in 2021 to include family and internal medicine doctors. And it offers mental health programs with rehabilitation psychologists who instruct on how to deal with doctors or loved ones who don’t believe that long COVID exists.
“I have patients who really have been devastated by the lack of support from coworkers [and] family,” Dr. Bender said.
In Campbell County, Wyo., the pandemic surge did not arrive in earnest until late 2021. Physical therapists at Campbell County’s Health Rehabilitation Services organized a rehabilitation program for residents with long COVID after recognizing the need, said Shannon Sorensen, rehabilitation director at Campbell County Health.
“We had patients coming in showing chest pain, or heart palpitations. There were people trying to get back to work. They were frustrated,” Ms. Sorensen said.
Myalgic encephalomyelitis and chronic fatigue syndrome activists have embraced the fight to recognize and help long COVID patients, noting the similarities between the conditions, and hope to help kickstart more organized research, treatment and benefits for long COVID sufferers and myalgic encephalomyelitis/chronic fatigue syndrome patients alike.
In Ft. Collins, Colo., disability activist Alison Sbrana has long had myalgic encephalomyelitis. She and other members of the local chapter of ME Action have met with state officials for several years and are finally seeing the results of those efforts.
Colorado Gov. Jared Polis has created the full-time position of policy adviser for long COVID and post–viral infection planning.
“This is one way forward of how state governments are (finally) paying attention to infection-triggered chronic illnesses and starting to think ahead on them,” Ms. Sbrana said.
New York City’s Health + Hospitals launched what may be the most expansive long COVID treatment program in the nation in April 2021. Called AfterCare, it provides physical and mental health services as well as community support systems and financial assistance.
A persistent issue for patients is that there isn’t yet a test for long COVID, like there is for COVID-19, said Amanda Johnson, MD, assistant vice president for ambulatory care and population health at New York Health + Hospitals. “It’s in many ways a diagnosis of exclusion. You have to make sure their shortness of breath isn’t caused by something else. The same with anemia,” she said.
California’s Department of Public Health has a detailed website devoted to the topic, including videos of “long haulers” describing their experiences.
Vermont is one of several states studying long COVID, said Mark Levine, MD, the state health commissioner. The state, in collaboration with the University of Vermont, has established a surveillance project to determine how many people have long COVID, as well as how severe it is, how long it lasts, and potential predispositions.
The University of Utah, Salt Lake City, established a comprehensive COVID-19 clinic more than a year ago that also handles long COVID patients, said Jeannette Brown, MD, PhD, an associate professor at the school and director of the COVID-19 clinic.
Jennifer Chevinsky, MD, MPH, already had a deep understanding of long COVID when she landed in Riverside County, Calif., in the summer of 2021. She came from Atlanta, where as part of her job as an epidemic intelligence service officer at the CDC, she heard stories of COVID-19 patients who were not getting better.
Now she is a deputy public health officer for Riverside County, in a region known for its deserts, sizzling summer temperatures and diverse populations. She said her department has helped launch programs such as post–COVID-19 follow-up phone calls and long COVID training programs that reach out to the many Latino residents in this county of 2.4 million people. It also includes Black and Native American residents.
“We’re making sure information is circulated with community and faith-based organizations, and community health workers,” she said.
Ms. McCorkell said there is still much work to do to raise public awareness of the risks of long COVID and how to obtain care for patients. She would like to see a national public health campaign about long COVID, possibly spearheaded by the Centers for Disease Control and Prevention in partnership with local health workers and community-based organizations.
“That,” she said, “could make a big difference.”
A version of this article first appeared on WebMD.com.
Children and COVID: Downward trend reverses with small increase in new cases
A small increase in new cases brought COVID-19’s latest losing streak to an end at 4 weeks, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
The 40,656 new cases reported bring the U.S. cumulative count of child COVID-19 cases to over 14.8 million since the pandemic began, which represents 18.4% of all cases, the AAP and CHA said in their weekly report based on state-level data.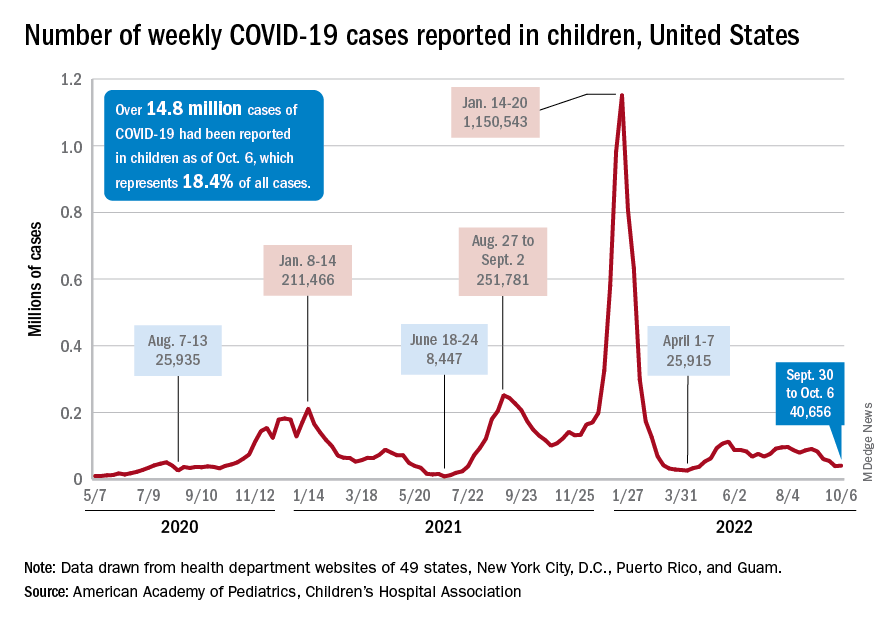
The increase in new cases was not reflected in emergency department visits or hospital admissions, which both continued sustained declines that started in August. In the week from Sept. 27 to Oct. 4, the 7-day averages for ED visits with diagnosed COVID were down by 21.5% (age 0-11), 27.3% (12-15), and 18.2% (16-17), the Centers for Disease Control and Prevention said, while the most recent 7-day average for new admissions – 127 per day for Oct. 2-8 – among children aged 0-17 years with confirmed COVID was down from 161 per day the previous week, a drop of over 21%.
The state-level data that are currently available (several states are no longer reporting) show Alaska (25.5%) and Vermont (25.4%) have the highest proportions of cumulative cases in children, and Florida (12.3%) and Utah (13.5%) have the lowest. Rhode Island has the highest rate of COVID-19 per 100,000 children at 40,427, while Missouri has the lowest at 14,252. The national average is 19,687 per 100,000, the AAP and CHA reported.
Taking a look at vaccination
Vaccinations were up slightly in children aged 12-17 years, as 20,000 initial doses were given during the week of Sept. 29 to Oct. 5, compared with 17,000 and 18,000 the previous 2 weeks. Initial vaccinations in younger children, however, continued declines dating back to August, the AAP said in its weekly vaccination trends report.
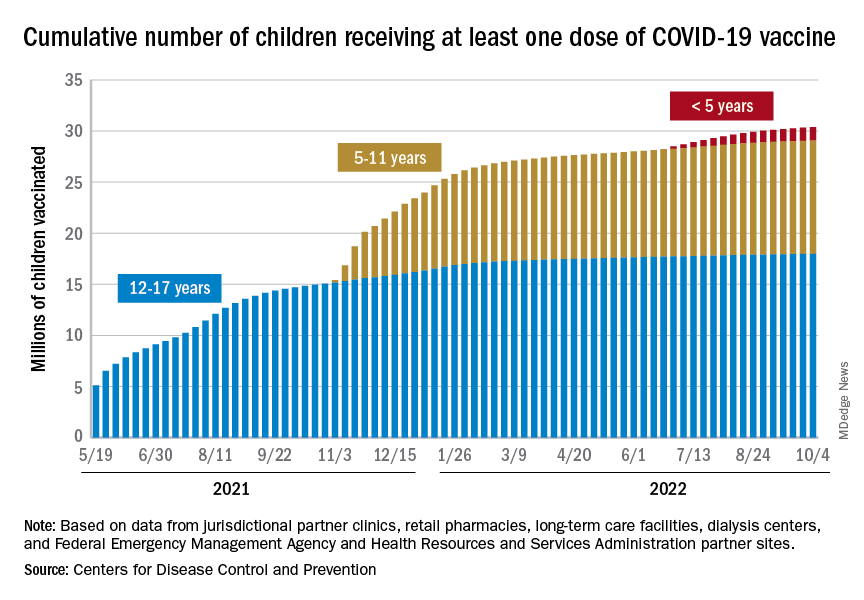
The District of Columbia and Massachusetts have the most highly vaccinated groups of 12- to 17-year-olds, as 100% and 95%, respectively, have received initial doses, while Wyoming (39%) and Idaho (42%) have the lowest. D.C. (73%) and Vermont (68%) have the highest proportions of vaccinated 5- to 11-year-olds, and Alabama (17%) and Mississippi (18%) have the lowest. For children under age 5 years, those in D.C. (33%) and Vermont (26%) are the most likely to have received an initial COVID vaccination, while Alabama, Louisiana, and Mississippi share national-low rates of 2%, the AAP said its report, which is based on CDC data.
When all states and territories are combined, 71% of children aged 12-17 have received at least one dose of vaccine, as have 38.6% of all children 5-11 years old and 6.7% of those under age 5. Almost 61% of the nation’s 16- to 17-year-olds have been fully vaccinated, along with 31.5% of those aged 5-11 and 2.4% of children younger than 5 years, the CDC said on its COVID Data Tracker.
About 42 million children – 58% of the population under the age of 18 years – have not received any vaccine yet, the AAP noted. Meanwhile, CDC data indicate that 36 children died of COVID in the last week, with pediatric deaths now totaling 1,781 over the course of the pandemic.
A small increase in new cases brought COVID-19’s latest losing streak to an end at 4 weeks, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
The 40,656 new cases reported bring the U.S. cumulative count of child COVID-19 cases to over 14.8 million since the pandemic began, which represents 18.4% of all cases, the AAP and CHA said in their weekly report based on state-level data.
The increase in new cases was not reflected in emergency department visits or hospital admissions, which both continued sustained declines that started in August. In the week from Sept. 27 to Oct. 4, the 7-day averages for ED visits with diagnosed COVID were down by 21.5% (age 0-11), 27.3% (12-15), and 18.2% (16-17), the Centers for Disease Control and Prevention said, while the most recent 7-day average for new admissions – 127 per day for Oct. 2-8 – among children aged 0-17 years with confirmed COVID was down from 161 per day the previous week, a drop of over 21%.
The state-level data that are currently available (several states are no longer reporting) show Alaska (25.5%) and Vermont (25.4%) have the highest proportions of cumulative cases in children, and Florida (12.3%) and Utah (13.5%) have the lowest. Rhode Island has the highest rate of COVID-19 per 100,000 children at 40,427, while Missouri has the lowest at 14,252. The national average is 19,687 per 100,000, the AAP and CHA reported.
Taking a look at vaccination
Vaccinations were up slightly in children aged 12-17 years, as 20,000 initial doses were given during the week of Sept. 29 to Oct. 5, compared with 17,000 and 18,000 the previous 2 weeks. Initial vaccinations in younger children, however, continued declines dating back to August, the AAP said in its weekly vaccination trends report.

The District of Columbia and Massachusetts have the most highly vaccinated groups of 12- to 17-year-olds, as 100% and 95%, respectively, have received initial doses, while Wyoming (39%) and Idaho (42%) have the lowest. D.C. (73%) and Vermont (68%) have the highest proportions of vaccinated 5- to 11-year-olds, and Alabama (17%) and Mississippi (18%) have the lowest. For children under age 5 years, those in D.C. (33%) and Vermont (26%) are the most likely to have received an initial COVID vaccination, while Alabama, Louisiana, and Mississippi share national-low rates of 2%, the AAP said its report, which is based on CDC data.
When all states and territories are combined, 71% of children aged 12-17 have received at least one dose of vaccine, as have 38.6% of all children 5-11 years old and 6.7% of those under age 5. Almost 61% of the nation’s 16- to 17-year-olds have been fully vaccinated, along with 31.5% of those aged 5-11 and 2.4% of children younger than 5 years, the CDC said on its COVID Data Tracker.
About 42 million children – 58% of the population under the age of 18 years – have not received any vaccine yet, the AAP noted. Meanwhile, CDC data indicate that 36 children died of COVID in the last week, with pediatric deaths now totaling 1,781 over the course of the pandemic.
A small increase in new cases brought COVID-19’s latest losing streak to an end at 4 weeks, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
The 40,656 new cases reported bring the U.S. cumulative count of child COVID-19 cases to over 14.8 million since the pandemic began, which represents 18.4% of all cases, the AAP and CHA said in their weekly report based on state-level data.
The increase in new cases was not reflected in emergency department visits or hospital admissions, which both continued sustained declines that started in August. In the week from Sept. 27 to Oct. 4, the 7-day averages for ED visits with diagnosed COVID were down by 21.5% (age 0-11), 27.3% (12-15), and 18.2% (16-17), the Centers for Disease Control and Prevention said, while the most recent 7-day average for new admissions – 127 per day for Oct. 2-8 – among children aged 0-17 years with confirmed COVID was down from 161 per day the previous week, a drop of over 21%.
The state-level data that are currently available (several states are no longer reporting) show Alaska (25.5%) and Vermont (25.4%) have the highest proportions of cumulative cases in children, and Florida (12.3%) and Utah (13.5%) have the lowest. Rhode Island has the highest rate of COVID-19 per 100,000 children at 40,427, while Missouri has the lowest at 14,252. The national average is 19,687 per 100,000, the AAP and CHA reported.
Taking a look at vaccination
Vaccinations were up slightly in children aged 12-17 years, as 20,000 initial doses were given during the week of Sept. 29 to Oct. 5, compared with 17,000 and 18,000 the previous 2 weeks. Initial vaccinations in younger children, however, continued declines dating back to August, the AAP said in its weekly vaccination trends report.

The District of Columbia and Massachusetts have the most highly vaccinated groups of 12- to 17-year-olds, as 100% and 95%, respectively, have received initial doses, while Wyoming (39%) and Idaho (42%) have the lowest. D.C. (73%) and Vermont (68%) have the highest proportions of vaccinated 5- to 11-year-olds, and Alabama (17%) and Mississippi (18%) have the lowest. For children under age 5 years, those in D.C. (33%) and Vermont (26%) are the most likely to have received an initial COVID vaccination, while Alabama, Louisiana, and Mississippi share national-low rates of 2%, the AAP said its report, which is based on CDC data.
When all states and territories are combined, 71% of children aged 12-17 have received at least one dose of vaccine, as have 38.6% of all children 5-11 years old and 6.7% of those under age 5. Almost 61% of the nation’s 16- to 17-year-olds have been fully vaccinated, along with 31.5% of those aged 5-11 and 2.4% of children younger than 5 years, the CDC said on its COVID Data Tracker.
About 42 million children – 58% of the population under the age of 18 years – have not received any vaccine yet, the AAP noted. Meanwhile, CDC data indicate that 36 children died of COVID in the last week, with pediatric deaths now totaling 1,781 over the course of the pandemic.
Previous endemic coronavirus encounters linked with long COVID
People who develop long COVID may be responding more strongly to a non–SARS-CoV-2 virus they encountered in the past than to SARS-CoV-2, a study by researchers at Harvard Medical School suggests.
Long COVID, also called postacute sequelae of COVID-19 (PASC), causes various symptoms that persist at least 4 weeks after the initial SARS-CoV-2 infection, they write in the preprint server medRxiv. Four authors explained their research into possible mechanisms of long COVID in an interview.
“Immunity to non-COVID endemic coronaviruses may play a role in who develops PASC,” co–lead author Jonathan D. Herman, MD, PhD, said. “There’s still so much more we need to understand, but it is striking that back-boosting of immune responses to coronavirus OC43 was uniquely enriched in individuals with PASC.”
“In the study, individuals with PASC preferentially generated stronger responses to previously encountered cold-causing coronaviruses,” co–senior author Galit Alter, PhD, said.
“Instead of generating strong SARS-CoV-2 immunity, they bolstered a response to a different coronavirus, potentially making their response less effective in clearing SARS-CoV-2. Surprisingly, most of the individuals had been vaccinated – and they still maintained this unusual antibody response – pointing to new therapeutic pathways to treat PASC,” Dr. Alter said.
Humoral immunity offers a clue to long-COVID origins
One-fifth of COVID-19 patients progress to long COVID, but which patients develop PASC and why are not well understood, the authors write.
“Antibodies represent powerful biomarkers that have been used for decades to diagnose disease. However, antibodies also provide a powerful source of information on previous infections. The use of antibody profiling, here, pointed to the presence of incomplete antibody responses to SARS-CoV-2 in individuals with PASC,” Dr. Alter said.
The researchers reviewed the medical records of patients in the Mass General Brigham health care system in Boston, including referrals from rheumatologists of participants diagnosed with COVID-19 outside the MGB system, starting on March 1, 2020.
They focused on patients with systemic autoimmune rheumatic diseases (SARDs) because their tendency toward inflammation and autoantibody production may make them more susceptible to PASC and enrich for specific inflammatory-driven endotypes.
All 43 participants had COVID-19 without hospital admission and SARDs. Patients treated only for fibromyalgia, osteoarthritis, mechanical back pain, gout, or pseudogout without a SARD were excluded from the study.
Overall, 79% of participants were female, 35% had rheumatoid arthritis, 19% had psoriatic arthritis, and 95% had received a COVID-19 vaccine.
The researchers used systems serology to perform comprehensive antibody profiling against SARS-CoV-2 and a panel of endemic pathogens or routine vaccine antigens.
Long-COVID patients had a distinct immune response
Overall, 17 patients developed PASC and 26 did not, and in those with PASC, they found a distinct humoral immune response. Patients with PASC:
- harbored less inflamed and weaker Fc-gamma receptor–binding anti–SARS-CoV-2 antibodies;
- showed a significantly expanded and more inflamed antibody response against endemic coronavirus OC43; and
- mounted more avid IgM responses and developed expanded inflammatory OC43 S2–specific Fc-receptor–binding responses, which were linked to cross reactivity across SARS-CoV-2 and common coronaviruses.
“Strengths of the study include the detailed phenotypes of cases after COVID-19, particularly to classify PASC presence or absence, as well as the depth and breadth of antibody profiling. This allowed us to identify a humoral immune signature of PASC,” said co–senior author Jeffrey A. Sparks, MD, MMSc.
“However, the study was limited in its size to investigate different types of PASC, such as fatigue or lung symptoms, that may have biologic differences. Also, all patients in the study had a preexisting rheumatic disease,” he acknowledged.
“A substantial portion of patients with COVID-19 will develop PASC, which can have substantial impact on health and quality of life,” said co–senior author Zachary S. Wallace, MD, MS. “Given the higher risk of COVID-19 in many patients with rheumatic disease, it is important to understand the etiology of PASC in this vulnerable population, to enable future diagnostic and therapeutic advances.”
Davey Smith, MD, professor of medicine and head of infectious diseases and global public health at the University of California, San Diego, in La Jolla, who was not involved in the study, called the findings interesting even though the results will not immediately affect patient care.
“There may be a link between previous non–SARS-CoV-2 coronavirus infection and PASC,” he added. “Perhaps, by understanding why some people do and do not get PASC, we can develop treatments for the condition.
“This paper is a preprint and will need to go through peer review,” Dr. Smith said. “There are many elements that need to be scrutinized. For example, there is no definition of PASC that is universally accepted, so how did that play into this study?”
Mark Cameron, PhD, associate professor in the department of population and quantitative health sciences at Case Western Reserve University, Cleveland, called this a strong study from a strong group, although it is a preprint prior to peer review.
“In this initial study, the scientists focused on people who had rheumatic disease before getting COVID-19, knowing they are at higher risk for lasting complications and hopefully are more immunologically similar when diagnosed with long COVID – a single ‘endotype’ or group of patients with similar clinical symptoms and background,” he noted.
“Our immune system’s memory sometimes fails to effectively fight a new virus that looks too much like a virus it saw before. This ineffective immune response can set up various problems, including the poor recoveries we see in people with long COVID,” he said.
“OC43 probably emerged in the late 1800s and probably caused a pandemic of severe respiratory illness between 1889 and 1890, previously thought to be a flu,” Dr. Cameron recalled. “OC43 is still around as an endemic coronavirus, usually causing mild or moderate upper-respiratory infections.”
COVID-19 immunity is complex, and previous SARS-CoV-2 infection doesn’t guarantee we won't get COVID-19 again, especially as new variants emerge, added Dr. Cameron, who also was not involved in the study.
“This study may help us better understand the risks and possible mechanisms associated with COVID-19 and long COVID in the face of previous coronavirus infections,” he said. “It may also help guide future COVID-19 therapies and vaccines.”
The authors plan further related research.
The study received grant support and an anonymous donation. Dr. Alter, Dr. Sparks, and Dr. Wallace report financial relationships with the pharmaceutical industry. All other authors, and Dr. Davey and Dr. Cameron, report no conflicts of interest with the study. All experts commented by email.
* This story was updated 10/12/2022.
People who develop long COVID may be responding more strongly to a non–SARS-CoV-2 virus they encountered in the past than to SARS-CoV-2, a study by researchers at Harvard Medical School suggests.
Long COVID, also called postacute sequelae of COVID-19 (PASC), causes various symptoms that persist at least 4 weeks after the initial SARS-CoV-2 infection, they write in the preprint server medRxiv. Four authors explained their research into possible mechanisms of long COVID in an interview.
“Immunity to non-COVID endemic coronaviruses may play a role in who develops PASC,” co–lead author Jonathan D. Herman, MD, PhD, said. “There’s still so much more we need to understand, but it is striking that back-boosting of immune responses to coronavirus OC43 was uniquely enriched in individuals with PASC.”
“In the study, individuals with PASC preferentially generated stronger responses to previously encountered cold-causing coronaviruses,” co–senior author Galit Alter, PhD, said.
“Instead of generating strong SARS-CoV-2 immunity, they bolstered a response to a different coronavirus, potentially making their response less effective in clearing SARS-CoV-2. Surprisingly, most of the individuals had been vaccinated – and they still maintained this unusual antibody response – pointing to new therapeutic pathways to treat PASC,” Dr. Alter said.
Humoral immunity offers a clue to long-COVID origins
One-fifth of COVID-19 patients progress to long COVID, but which patients develop PASC and why are not well understood, the authors write.
“Antibodies represent powerful biomarkers that have been used for decades to diagnose disease. However, antibodies also provide a powerful source of information on previous infections. The use of antibody profiling, here, pointed to the presence of incomplete antibody responses to SARS-CoV-2 in individuals with PASC,” Dr. Alter said.
The researchers reviewed the medical records of patients in the Mass General Brigham health care system in Boston, including referrals from rheumatologists of participants diagnosed with COVID-19 outside the MGB system, starting on March 1, 2020.
They focused on patients with systemic autoimmune rheumatic diseases (SARDs) because their tendency toward inflammation and autoantibody production may make them more susceptible to PASC and enrich for specific inflammatory-driven endotypes.
All 43 participants had COVID-19 without hospital admission and SARDs. Patients treated only for fibromyalgia, osteoarthritis, mechanical back pain, gout, or pseudogout without a SARD were excluded from the study.
Overall, 79% of participants were female, 35% had rheumatoid arthritis, 19% had psoriatic arthritis, and 95% had received a COVID-19 vaccine.
The researchers used systems serology to perform comprehensive antibody profiling against SARS-CoV-2 and a panel of endemic pathogens or routine vaccine antigens.
Long-COVID patients had a distinct immune response
Overall, 17 patients developed PASC and 26 did not, and in those with PASC, they found a distinct humoral immune response. Patients with PASC:
- harbored less inflamed and weaker Fc-gamma receptor–binding anti–SARS-CoV-2 antibodies;
- showed a significantly expanded and more inflamed antibody response against endemic coronavirus OC43; and
- mounted more avid IgM responses and developed expanded inflammatory OC43 S2–specific Fc-receptor–binding responses, which were linked to cross reactivity across SARS-CoV-2 and common coronaviruses.
“Strengths of the study include the detailed phenotypes of cases after COVID-19, particularly to classify PASC presence or absence, as well as the depth and breadth of antibody profiling. This allowed us to identify a humoral immune signature of PASC,” said co–senior author Jeffrey A. Sparks, MD, MMSc.
“However, the study was limited in its size to investigate different types of PASC, such as fatigue or lung symptoms, that may have biologic differences. Also, all patients in the study had a preexisting rheumatic disease,” he acknowledged.
“A substantial portion of patients with COVID-19 will develop PASC, which can have substantial impact on health and quality of life,” said co–senior author Zachary S. Wallace, MD, MS. “Given the higher risk of COVID-19 in many patients with rheumatic disease, it is important to understand the etiology of PASC in this vulnerable population, to enable future diagnostic and therapeutic advances.”
Davey Smith, MD, professor of medicine and head of infectious diseases and global public health at the University of California, San Diego, in La Jolla, who was not involved in the study, called the findings interesting even though the results will not immediately affect patient care.
“There may be a link between previous non–SARS-CoV-2 coronavirus infection and PASC,” he added. “Perhaps, by understanding why some people do and do not get PASC, we can develop treatments for the condition.
“This paper is a preprint and will need to go through peer review,” Dr. Smith said. “There are many elements that need to be scrutinized. For example, there is no definition of PASC that is universally accepted, so how did that play into this study?”
Mark Cameron, PhD, associate professor in the department of population and quantitative health sciences at Case Western Reserve University, Cleveland, called this a strong study from a strong group, although it is a preprint prior to peer review.
“In this initial study, the scientists focused on people who had rheumatic disease before getting COVID-19, knowing they are at higher risk for lasting complications and hopefully are more immunologically similar when diagnosed with long COVID – a single ‘endotype’ or group of patients with similar clinical symptoms and background,” he noted.
“Our immune system’s memory sometimes fails to effectively fight a new virus that looks too much like a virus it saw before. This ineffective immune response can set up various problems, including the poor recoveries we see in people with long COVID,” he said.
“OC43 probably emerged in the late 1800s and probably caused a pandemic of severe respiratory illness between 1889 and 1890, previously thought to be a flu,” Dr. Cameron recalled. “OC43 is still around as an endemic coronavirus, usually causing mild or moderate upper-respiratory infections.”
COVID-19 immunity is complex, and previous SARS-CoV-2 infection doesn’t guarantee we won't get COVID-19 again, especially as new variants emerge, added Dr. Cameron, who also was not involved in the study.
“This study may help us better understand the risks and possible mechanisms associated with COVID-19 and long COVID in the face of previous coronavirus infections,” he said. “It may also help guide future COVID-19 therapies and vaccines.”
The authors plan further related research.
The study received grant support and an anonymous donation. Dr. Alter, Dr. Sparks, and Dr. Wallace report financial relationships with the pharmaceutical industry. All other authors, and Dr. Davey and Dr. Cameron, report no conflicts of interest with the study. All experts commented by email.
* This story was updated 10/12/2022.
People who develop long COVID may be responding more strongly to a non–SARS-CoV-2 virus they encountered in the past than to SARS-CoV-2, a study by researchers at Harvard Medical School suggests.
Long COVID, also called postacute sequelae of COVID-19 (PASC), causes various symptoms that persist at least 4 weeks after the initial SARS-CoV-2 infection, they write in the preprint server medRxiv. Four authors explained their research into possible mechanisms of long COVID in an interview.
“Immunity to non-COVID endemic coronaviruses may play a role in who develops PASC,” co–lead author Jonathan D. Herman, MD, PhD, said. “There’s still so much more we need to understand, but it is striking that back-boosting of immune responses to coronavirus OC43 was uniquely enriched in individuals with PASC.”
“In the study, individuals with PASC preferentially generated stronger responses to previously encountered cold-causing coronaviruses,” co–senior author Galit Alter, PhD, said.
“Instead of generating strong SARS-CoV-2 immunity, they bolstered a response to a different coronavirus, potentially making their response less effective in clearing SARS-CoV-2. Surprisingly, most of the individuals had been vaccinated – and they still maintained this unusual antibody response – pointing to new therapeutic pathways to treat PASC,” Dr. Alter said.
Humoral immunity offers a clue to long-COVID origins
One-fifth of COVID-19 patients progress to long COVID, but which patients develop PASC and why are not well understood, the authors write.
“Antibodies represent powerful biomarkers that have been used for decades to diagnose disease. However, antibodies also provide a powerful source of information on previous infections. The use of antibody profiling, here, pointed to the presence of incomplete antibody responses to SARS-CoV-2 in individuals with PASC,” Dr. Alter said.
The researchers reviewed the medical records of patients in the Mass General Brigham health care system in Boston, including referrals from rheumatologists of participants diagnosed with COVID-19 outside the MGB system, starting on March 1, 2020.
They focused on patients with systemic autoimmune rheumatic diseases (SARDs) because their tendency toward inflammation and autoantibody production may make them more susceptible to PASC and enrich for specific inflammatory-driven endotypes.
All 43 participants had COVID-19 without hospital admission and SARDs. Patients treated only for fibromyalgia, osteoarthritis, mechanical back pain, gout, or pseudogout without a SARD were excluded from the study.
Overall, 79% of participants were female, 35% had rheumatoid arthritis, 19% had psoriatic arthritis, and 95% had received a COVID-19 vaccine.
The researchers used systems serology to perform comprehensive antibody profiling against SARS-CoV-2 and a panel of endemic pathogens or routine vaccine antigens.
Long-COVID patients had a distinct immune response
Overall, 17 patients developed PASC and 26 did not, and in those with PASC, they found a distinct humoral immune response. Patients with PASC:
- harbored less inflamed and weaker Fc-gamma receptor–binding anti–SARS-CoV-2 antibodies;
- showed a significantly expanded and more inflamed antibody response against endemic coronavirus OC43; and
- mounted more avid IgM responses and developed expanded inflammatory OC43 S2–specific Fc-receptor–binding responses, which were linked to cross reactivity across SARS-CoV-2 and common coronaviruses.
“Strengths of the study include the detailed phenotypes of cases after COVID-19, particularly to classify PASC presence or absence, as well as the depth and breadth of antibody profiling. This allowed us to identify a humoral immune signature of PASC,” said co–senior author Jeffrey A. Sparks, MD, MMSc.
“However, the study was limited in its size to investigate different types of PASC, such as fatigue or lung symptoms, that may have biologic differences. Also, all patients in the study had a preexisting rheumatic disease,” he acknowledged.
“A substantial portion of patients with COVID-19 will develop PASC, which can have substantial impact on health and quality of life,” said co–senior author Zachary S. Wallace, MD, MS. “Given the higher risk of COVID-19 in many patients with rheumatic disease, it is important to understand the etiology of PASC in this vulnerable population, to enable future diagnostic and therapeutic advances.”
Davey Smith, MD, professor of medicine and head of infectious diseases and global public health at the University of California, San Diego, in La Jolla, who was not involved in the study, called the findings interesting even though the results will not immediately affect patient care.
“There may be a link between previous non–SARS-CoV-2 coronavirus infection and PASC,” he added. “Perhaps, by understanding why some people do and do not get PASC, we can develop treatments for the condition.
“This paper is a preprint and will need to go through peer review,” Dr. Smith said. “There are many elements that need to be scrutinized. For example, there is no definition of PASC that is universally accepted, so how did that play into this study?”
Mark Cameron, PhD, associate professor in the department of population and quantitative health sciences at Case Western Reserve University, Cleveland, called this a strong study from a strong group, although it is a preprint prior to peer review.
“In this initial study, the scientists focused on people who had rheumatic disease before getting COVID-19, knowing they are at higher risk for lasting complications and hopefully are more immunologically similar when diagnosed with long COVID – a single ‘endotype’ or group of patients with similar clinical symptoms and background,” he noted.
“Our immune system’s memory sometimes fails to effectively fight a new virus that looks too much like a virus it saw before. This ineffective immune response can set up various problems, including the poor recoveries we see in people with long COVID,” he said.
“OC43 probably emerged in the late 1800s and probably caused a pandemic of severe respiratory illness between 1889 and 1890, previously thought to be a flu,” Dr. Cameron recalled. “OC43 is still around as an endemic coronavirus, usually causing mild or moderate upper-respiratory infections.”
COVID-19 immunity is complex, and previous SARS-CoV-2 infection doesn’t guarantee we won't get COVID-19 again, especially as new variants emerge, added Dr. Cameron, who also was not involved in the study.
“This study may help us better understand the risks and possible mechanisms associated with COVID-19 and long COVID in the face of previous coronavirus infections,” he said. “It may also help guide future COVID-19 therapies and vaccines.”
The authors plan further related research.
The study received grant support and an anonymous donation. Dr. Alter, Dr. Sparks, and Dr. Wallace report financial relationships with the pharmaceutical industry. All other authors, and Dr. Davey and Dr. Cameron, report no conflicts of interest with the study. All experts commented by email.
* This story was updated 10/12/2022.
FROM MEDRXIV
Sigmoidoscopy screening cuts CRC mortality, incidence
Although endoscopic screening provides an opportunity for early identification and removal of premalignant polyps, data quantifying the long-term effects of sigmoidoscopy screening are lacking, corresponding author Frederik E. Juul, MD, said in an interview.
“Sigmoidoscopy screening have been shown to reduce colorectal cancer incidence and mortality, but it was unknown how long-lasting the effects were, and whether the effect differed by sex or age,” Dr. Juul said.
“For the first time, we were able to pool data from all four randomized sigmoidoscopy screening trials and include data from recent updates from two of the trials (U.S. and Italy), which means that we were able to answer these questions better than ever before,” he said.
In the pooled analysis, published in Annals of Internal Medicine, researchers from Norway, the United States, Italy, and the United Kingdom reviewed data from four studies with at least 15 years of follow-up. The analysis included 137,493 individuals randomized to at least one sigmoidoscopy screening and 137,459 randomized to usual care.
The primary outcomes were the incidence and mortality of CRC after sigmoidoscopy screening, compared with usual care, in adults with average CRC risk aged 55-64 years. Secondary outcomes included CRC incidence and mortality based on distal versus proximal colon, sex, and older versus younger age group (55-59 years vs. 60-64 years at study enrollment).
After 15 years’ follow-up, the pooled cumulative incidence of CRC was 1.84 cases per 100 persons in the screening group versus 2.35 cases per 100 persons in the usual-care group, representing a 21% reduction in incidence among those who were screened.
The pooled cumulative CRC mortality was 0.51 deaths per 100 persons in the screening group versus 0.65 deaths per 100 persons in the usual-care group, representing a 20% reduction in CRC mortality for those who were screened, the researchers noted. The all-cause mortality was reduced by 2% in the screening group compared with usual care; the pooled cumulative all-cause mortality was 14.3 deaths per 100 persons in the screening group versus 14.6 deaths per 100 persons in the usual-care group.
In terms of secondary outcomes, the significant reductions in CRC incidence and mortality were confined to the distal colon, with no significant differences observed in the proximal colon, the researchers noted. The reasons for this difference are unclear. Previous studies of three of the four trials showed a small reduction in CRC in the proximal colon, but may be related to the longer follow-up in the analysis of four trials.
The incidence of CRC varied by gender, with an incidence reduction of 25% for men versus 16% for women. The reasons for the gender difference are yet to be undetermined, but may include differences in the quality of bowel preparation, the greater technical challenge of screening women, and the higher incidence and proportion of proximal colon cancer versus distal colon cancer in women, the researchers noted.
“The long-term benefit of one single procedure was probably what surprised us the most,” Dr. Juul said in an interview. “Not only were the cumulative incidence and mortality lower in screened individuals 15 years after the procedure, but the yearly incidence was consistently lower in screened individuals compared to usual care, even at the end of the follow-up period.
“Although a previous study in Norway had indicated a sex difference in effect, we were surprised to see this in a pooled analysis across trials in four different countries,” he added.
Data may drive screening guidelines
The main finding of the study is that sigmoidoscopy screening with investigation of the distal colon provides at least 15 years of protection against colorectal cancer; “this may have an impact on how often average-risk individuals needs to be screened,” Dr. Juul said in an interview.
As for additional research, ongoing studies are examining primary colonoscopy screening, including a study recently published in the New England Journal of Medicine, Dr. Juul said.“Our study investigating sigmoidoscopy screening has a longer follow-up and it will be interesting to see if primary colonoscopy screening is equally or more effective as sigmoidoscopy at 15-years follow-up.”
More research is needed on direct comparisons of different colorectal cancer screening methods such as sigmoidoscopy and colonoscopy, said Dr. Juul. In addition, “The optimal surveillance interval in individuals identified at screening to be low- or high-risk of developing colorectal cancer are unknown,” he said.
“Our research group is involved in trials [the EPoS trials] looking into this question, but there are still years until we have the final results,” he added.
The findings were limited by several factors including the variation in methodology among the four trials and the lower number of individuals referred for colonoscopy in the U.K. and Italian trials, lack of analysis of potential confounding variables, and less granular data from the U.K. trial because of privacy regulations, the researchers wrote.
However, the findings were strengthened by the large study population, long-term follow-up, and detailed data, and they indicate a “significant and sustained” effect of screening sigmoidoscopy for the long-term reduction of CRC incidence and mortality, the authors concluded.
Findings can inform shared decision-making
“Colon cancer is the third-leading cause of death in the United States in men and women, and the second-leading cause of cancer deaths if we were to combine both genders,” Noel Deep, MD, said in an interview. “Sigmoidoscopy is more acceptable as a screening tool compared to a colonoscopy because of the lower risk of bowel injury, fewer side effects and less of a bowel prep, and also less need for sedation. This current study confirms prior data, including the 2012 PLCO trial, that it [sigmoidoscopy] reduces the incidence and mortality from colorectal cancer.”
The study findings were not surprising, given the prior knowledge and evidence of the benefits of sigmoidoscopy, Dr. Deep said, who was not involved in the study. However, “the fact that a single sigmoidoscopy led to decreased incidence and decreased mortality at 15 years was surprising to me, as current models suggest increasing incidence of proximal colon adenomas and cancers, which did not seem to be the case in this study.”
The current study can help primary care physicians and advance practice clinicians in patient counseling by supporting sigmoidoscopy as an option for patients who are unwilling to commit to a full colonoscopy, Dr. Deep said. However, “the patients should be advised that abnormal findings on the sigmoidoscopy would necessitate them being referred for a colonoscopy, and also the limitations of a sigmoidoscopy in detecting polyps or cancers in the cecum, ascending colon, transverse colon and descending colon.”
Looking ahead, “I would like to see research into the appropriate age for colorectal cancer screening using sigmoidoscopy and any benefit in offering this option at an earlier age,” Dr. Deep said. He also expressed a wish to know more about the reasons for the decreased benefit of screening sigmoidoscopy in women, and the reasons for the observed difference in all-cause mortality between genders.
“I would also like to see what the results of screening colonoscopies in a general population would reveal, and if it would reveal similar benefits, and also if there would be a gender difference or age-based difference in outcomes,” he said.
The study was supported by the Health Fund of South-East Norway. The researchers had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the editorial advisory board of Internal Medicine News.
Although endoscopic screening provides an opportunity for early identification and removal of premalignant polyps, data quantifying the long-term effects of sigmoidoscopy screening are lacking, corresponding author Frederik E. Juul, MD, said in an interview.
“Sigmoidoscopy screening have been shown to reduce colorectal cancer incidence and mortality, but it was unknown how long-lasting the effects were, and whether the effect differed by sex or age,” Dr. Juul said.
“For the first time, we were able to pool data from all four randomized sigmoidoscopy screening trials and include data from recent updates from two of the trials (U.S. and Italy), which means that we were able to answer these questions better than ever before,” he said.
In the pooled analysis, published in Annals of Internal Medicine, researchers from Norway, the United States, Italy, and the United Kingdom reviewed data from four studies with at least 15 years of follow-up. The analysis included 137,493 individuals randomized to at least one sigmoidoscopy screening and 137,459 randomized to usual care.
The primary outcomes were the incidence and mortality of CRC after sigmoidoscopy screening, compared with usual care, in adults with average CRC risk aged 55-64 years. Secondary outcomes included CRC incidence and mortality based on distal versus proximal colon, sex, and older versus younger age group (55-59 years vs. 60-64 years at study enrollment).
After 15 years’ follow-up, the pooled cumulative incidence of CRC was 1.84 cases per 100 persons in the screening group versus 2.35 cases per 100 persons in the usual-care group, representing a 21% reduction in incidence among those who were screened.
The pooled cumulative CRC mortality was 0.51 deaths per 100 persons in the screening group versus 0.65 deaths per 100 persons in the usual-care group, representing a 20% reduction in CRC mortality for those who were screened, the researchers noted. The all-cause mortality was reduced by 2% in the screening group compared with usual care; the pooled cumulative all-cause mortality was 14.3 deaths per 100 persons in the screening group versus 14.6 deaths per 100 persons in the usual-care group.
In terms of secondary outcomes, the significant reductions in CRC incidence and mortality were confined to the distal colon, with no significant differences observed in the proximal colon, the researchers noted. The reasons for this difference are unclear. Previous studies of three of the four trials showed a small reduction in CRC in the proximal colon, but may be related to the longer follow-up in the analysis of four trials.
The incidence of CRC varied by gender, with an incidence reduction of 25% for men versus 16% for women. The reasons for the gender difference are yet to be undetermined, but may include differences in the quality of bowel preparation, the greater technical challenge of screening women, and the higher incidence and proportion of proximal colon cancer versus distal colon cancer in women, the researchers noted.
“The long-term benefit of one single procedure was probably what surprised us the most,” Dr. Juul said in an interview. “Not only were the cumulative incidence and mortality lower in screened individuals 15 years after the procedure, but the yearly incidence was consistently lower in screened individuals compared to usual care, even at the end of the follow-up period.
“Although a previous study in Norway had indicated a sex difference in effect, we were surprised to see this in a pooled analysis across trials in four different countries,” he added.
Data may drive screening guidelines
The main finding of the study is that sigmoidoscopy screening with investigation of the distal colon provides at least 15 years of protection against colorectal cancer; “this may have an impact on how often average-risk individuals needs to be screened,” Dr. Juul said in an interview.
As for additional research, ongoing studies are examining primary colonoscopy screening, including a study recently published in the New England Journal of Medicine, Dr. Juul said.“Our study investigating sigmoidoscopy screening has a longer follow-up and it will be interesting to see if primary colonoscopy screening is equally or more effective as sigmoidoscopy at 15-years follow-up.”
More research is needed on direct comparisons of different colorectal cancer screening methods such as sigmoidoscopy and colonoscopy, said Dr. Juul. In addition, “The optimal surveillance interval in individuals identified at screening to be low- or high-risk of developing colorectal cancer are unknown,” he said.
“Our research group is involved in trials [the EPoS trials] looking into this question, but there are still years until we have the final results,” he added.
The findings were limited by several factors including the variation in methodology among the four trials and the lower number of individuals referred for colonoscopy in the U.K. and Italian trials, lack of analysis of potential confounding variables, and less granular data from the U.K. trial because of privacy regulations, the researchers wrote.
However, the findings were strengthened by the large study population, long-term follow-up, and detailed data, and they indicate a “significant and sustained” effect of screening sigmoidoscopy for the long-term reduction of CRC incidence and mortality, the authors concluded.
Findings can inform shared decision-making
“Colon cancer is the third-leading cause of death in the United States in men and women, and the second-leading cause of cancer deaths if we were to combine both genders,” Noel Deep, MD, said in an interview. “Sigmoidoscopy is more acceptable as a screening tool compared to a colonoscopy because of the lower risk of bowel injury, fewer side effects and less of a bowel prep, and also less need for sedation. This current study confirms prior data, including the 2012 PLCO trial, that it [sigmoidoscopy] reduces the incidence and mortality from colorectal cancer.”
The study findings were not surprising, given the prior knowledge and evidence of the benefits of sigmoidoscopy, Dr. Deep said, who was not involved in the study. However, “the fact that a single sigmoidoscopy led to decreased incidence and decreased mortality at 15 years was surprising to me, as current models suggest increasing incidence of proximal colon adenomas and cancers, which did not seem to be the case in this study.”
The current study can help primary care physicians and advance practice clinicians in patient counseling by supporting sigmoidoscopy as an option for patients who are unwilling to commit to a full colonoscopy, Dr. Deep said. However, “the patients should be advised that abnormal findings on the sigmoidoscopy would necessitate them being referred for a colonoscopy, and also the limitations of a sigmoidoscopy in detecting polyps or cancers in the cecum, ascending colon, transverse colon and descending colon.”
Looking ahead, “I would like to see research into the appropriate age for colorectal cancer screening using sigmoidoscopy and any benefit in offering this option at an earlier age,” Dr. Deep said. He also expressed a wish to know more about the reasons for the decreased benefit of screening sigmoidoscopy in women, and the reasons for the observed difference in all-cause mortality between genders.
“I would also like to see what the results of screening colonoscopies in a general population would reveal, and if it would reveal similar benefits, and also if there would be a gender difference or age-based difference in outcomes,” he said.
The study was supported by the Health Fund of South-East Norway. The researchers had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the editorial advisory board of Internal Medicine News.
Although endoscopic screening provides an opportunity for early identification and removal of premalignant polyps, data quantifying the long-term effects of sigmoidoscopy screening are lacking, corresponding author Frederik E. Juul, MD, said in an interview.
“Sigmoidoscopy screening have been shown to reduce colorectal cancer incidence and mortality, but it was unknown how long-lasting the effects were, and whether the effect differed by sex or age,” Dr. Juul said.
“For the first time, we were able to pool data from all four randomized sigmoidoscopy screening trials and include data from recent updates from two of the trials (U.S. and Italy), which means that we were able to answer these questions better than ever before,” he said.
In the pooled analysis, published in Annals of Internal Medicine, researchers from Norway, the United States, Italy, and the United Kingdom reviewed data from four studies with at least 15 years of follow-up. The analysis included 137,493 individuals randomized to at least one sigmoidoscopy screening and 137,459 randomized to usual care.
The primary outcomes were the incidence and mortality of CRC after sigmoidoscopy screening, compared with usual care, in adults with average CRC risk aged 55-64 years. Secondary outcomes included CRC incidence and mortality based on distal versus proximal colon, sex, and older versus younger age group (55-59 years vs. 60-64 years at study enrollment).
After 15 years’ follow-up, the pooled cumulative incidence of CRC was 1.84 cases per 100 persons in the screening group versus 2.35 cases per 100 persons in the usual-care group, representing a 21% reduction in incidence among those who were screened.
The pooled cumulative CRC mortality was 0.51 deaths per 100 persons in the screening group versus 0.65 deaths per 100 persons in the usual-care group, representing a 20% reduction in CRC mortality for those who were screened, the researchers noted. The all-cause mortality was reduced by 2% in the screening group compared with usual care; the pooled cumulative all-cause mortality was 14.3 deaths per 100 persons in the screening group versus 14.6 deaths per 100 persons in the usual-care group.
In terms of secondary outcomes, the significant reductions in CRC incidence and mortality were confined to the distal colon, with no significant differences observed in the proximal colon, the researchers noted. The reasons for this difference are unclear. Previous studies of three of the four trials showed a small reduction in CRC in the proximal colon, but may be related to the longer follow-up in the analysis of four trials.
The incidence of CRC varied by gender, with an incidence reduction of 25% for men versus 16% for women. The reasons for the gender difference are yet to be undetermined, but may include differences in the quality of bowel preparation, the greater technical challenge of screening women, and the higher incidence and proportion of proximal colon cancer versus distal colon cancer in women, the researchers noted.
“The long-term benefit of one single procedure was probably what surprised us the most,” Dr. Juul said in an interview. “Not only were the cumulative incidence and mortality lower in screened individuals 15 years after the procedure, but the yearly incidence was consistently lower in screened individuals compared to usual care, even at the end of the follow-up period.
“Although a previous study in Norway had indicated a sex difference in effect, we were surprised to see this in a pooled analysis across trials in four different countries,” he added.
Data may drive screening guidelines
The main finding of the study is that sigmoidoscopy screening with investigation of the distal colon provides at least 15 years of protection against colorectal cancer; “this may have an impact on how often average-risk individuals needs to be screened,” Dr. Juul said in an interview.
As for additional research, ongoing studies are examining primary colonoscopy screening, including a study recently published in the New England Journal of Medicine, Dr. Juul said.“Our study investigating sigmoidoscopy screening has a longer follow-up and it will be interesting to see if primary colonoscopy screening is equally or more effective as sigmoidoscopy at 15-years follow-up.”
More research is needed on direct comparisons of different colorectal cancer screening methods such as sigmoidoscopy and colonoscopy, said Dr. Juul. In addition, “The optimal surveillance interval in individuals identified at screening to be low- or high-risk of developing colorectal cancer are unknown,” he said.
“Our research group is involved in trials [the EPoS trials] looking into this question, but there are still years until we have the final results,” he added.
The findings were limited by several factors including the variation in methodology among the four trials and the lower number of individuals referred for colonoscopy in the U.K. and Italian trials, lack of analysis of potential confounding variables, and less granular data from the U.K. trial because of privacy regulations, the researchers wrote.
However, the findings were strengthened by the large study population, long-term follow-up, and detailed data, and they indicate a “significant and sustained” effect of screening sigmoidoscopy for the long-term reduction of CRC incidence and mortality, the authors concluded.
Findings can inform shared decision-making
“Colon cancer is the third-leading cause of death in the United States in men and women, and the second-leading cause of cancer deaths if we were to combine both genders,” Noel Deep, MD, said in an interview. “Sigmoidoscopy is more acceptable as a screening tool compared to a colonoscopy because of the lower risk of bowel injury, fewer side effects and less of a bowel prep, and also less need for sedation. This current study confirms prior data, including the 2012 PLCO trial, that it [sigmoidoscopy] reduces the incidence and mortality from colorectal cancer.”
The study findings were not surprising, given the prior knowledge and evidence of the benefits of sigmoidoscopy, Dr. Deep said, who was not involved in the study. However, “the fact that a single sigmoidoscopy led to decreased incidence and decreased mortality at 15 years was surprising to me, as current models suggest increasing incidence of proximal colon adenomas and cancers, which did not seem to be the case in this study.”
The current study can help primary care physicians and advance practice clinicians in patient counseling by supporting sigmoidoscopy as an option for patients who are unwilling to commit to a full colonoscopy, Dr. Deep said. However, “the patients should be advised that abnormal findings on the sigmoidoscopy would necessitate them being referred for a colonoscopy, and also the limitations of a sigmoidoscopy in detecting polyps or cancers in the cecum, ascending colon, transverse colon and descending colon.”
Looking ahead, “I would like to see research into the appropriate age for colorectal cancer screening using sigmoidoscopy and any benefit in offering this option at an earlier age,” Dr. Deep said. He also expressed a wish to know more about the reasons for the decreased benefit of screening sigmoidoscopy in women, and the reasons for the observed difference in all-cause mortality between genders.
“I would also like to see what the results of screening colonoscopies in a general population would reveal, and if it would reveal similar benefits, and also if there would be a gender difference or age-based difference in outcomes,” he said.
The study was supported by the Health Fund of South-East Norway. The researchers had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the editorial advisory board of Internal Medicine News.
FROM ANNALS OF INTERNAL MEDICINE
Longer boarding times predict patient processing in ED
on the basis of data from nearly 900 facilities presented at the annual meeting of the American College of Emergency Physicians.
The study was important to conduct at this time because ED boarding is significantly limiting ED physicians to provide optimal care, said Camila Tyminski, MD, of Brown University, Providence, R.I., who presented the findings at the meeting.
“Boarding had steadily been rising prior to the COVID-19 pandemic due to increased ED use. As our data show, boarding had a detrimental impact on ED throughput measures, including increased door to provider time, increased length of stay of the patient discharged from the ED, and increased rate of patients that left before completion of treatment,” she said.
“It was important to understand these trends prior to 2019-2020 because the COVID-19 pandemic and national nursing shortage have drastically worsened boarding. This study provided a framework for future studies on boarding across ED’s nationally since the start of the pandemic,” she added.
“Post-pandemic, we have hit a crisis point,” lead author Anthony Napoli, MD, also of Brown University, said in an interview. “Boarding is largely a hospital capacity problem, but one key fix germane to EM [emergency medicine] is the provider in triage model (PIT). While PIT has been shown to improve efficiency of ED care, a single institution study demonstrated that it was unable to mitigate the effects of boarding. The study of the association of boarding and efficiency of ED operations and intake needed to be shown on a national scale,” he said.
The researchers reviewed cross-sectional ED operational data from the ED Department Benchmarking Alliance (EDBA), a voluntary database that includes self-reports of operational metrics from approximately half of EDs in the United States.
The data set included 892 EDs; freestanding and pediatric EDs, as well as those with missing boarding data, were excluded.
The primary outcome was boarding time, door-to-provider time (D2P), length of stay for discharged patients (LOSD) and the percentage of patients who left the hospital before treatment was complete (LBTC).
In a multivariate analysis, increased boarding time was significantly associated with longer D2P time, LOSD time, and rates of LBTC.
Overall, D2P and LOSD increased by 0.8 minutes and 2.8 minutes, respectively, for each additional 10 minutes of boarding time. LBTC rates increased by 0.1% for each additional 10 minutes of boarding time.
However, boarding did not have a significant impact on operational metrics among hospitals with fewer than 20,000 visits per year.
Although more research is needed, the results indicate that boarding reduces the throughput of nonboarded patients at a ratio of approximately 4:1. The limited impact of ED efficiency measures on operations highlights the need for hospital-based solutions to boarding, Dr. Tyminski concluded.
“Overall, we expected that there would be an association between boarding and reductions in ED intake and operational efficiency,” said Dr. Napoli in an interview. “However, we were surprised the relationship continued to be as strong in a national study of nearly a quarter of all EDs, as it did in our prior local study,” he said. “Every 10 minutes of boarding in an ED is associated with an approximate 0.1% increase in LWBS and a 3-minute increase in LOSD. Extrapolating this association across the country, we predicted that nearly one million patients may have potentially not received ED care due to boarding,” he explained. “Not only does this potentially have a huge impact on hospital finances but also the overall health of our patients,” he added.
The key takeaway from the study is that boarding is a hospital capacity management issue, said Dr. Napoli. Hospital leadership must be directly involved in plans to mitigate or eliminate it to the extent possible; until then, boarding will continue to result in inefficient ED operations, he explained.
“As ED providers, we are limited in what we can do, but one area where we might be able to make the most impact is to optimize the care and throughput of the LOSD patients,” Dr. Tyminski said. More research is needed to see if interventions to reduce boarding correspond with equivalent improvements in emergency department intake and improved ED throughput, she noted.
The study received no outside funding. The researchers disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
on the basis of data from nearly 900 facilities presented at the annual meeting of the American College of Emergency Physicians.
The study was important to conduct at this time because ED boarding is significantly limiting ED physicians to provide optimal care, said Camila Tyminski, MD, of Brown University, Providence, R.I., who presented the findings at the meeting.
“Boarding had steadily been rising prior to the COVID-19 pandemic due to increased ED use. As our data show, boarding had a detrimental impact on ED throughput measures, including increased door to provider time, increased length of stay of the patient discharged from the ED, and increased rate of patients that left before completion of treatment,” she said.
“It was important to understand these trends prior to 2019-2020 because the COVID-19 pandemic and national nursing shortage have drastically worsened boarding. This study provided a framework for future studies on boarding across ED’s nationally since the start of the pandemic,” she added.
“Post-pandemic, we have hit a crisis point,” lead author Anthony Napoli, MD, also of Brown University, said in an interview. “Boarding is largely a hospital capacity problem, but one key fix germane to EM [emergency medicine] is the provider in triage model (PIT). While PIT has been shown to improve efficiency of ED care, a single institution study demonstrated that it was unable to mitigate the effects of boarding. The study of the association of boarding and efficiency of ED operations and intake needed to be shown on a national scale,” he said.
The researchers reviewed cross-sectional ED operational data from the ED Department Benchmarking Alliance (EDBA), a voluntary database that includes self-reports of operational metrics from approximately half of EDs in the United States.
The data set included 892 EDs; freestanding and pediatric EDs, as well as those with missing boarding data, were excluded.
The primary outcome was boarding time, door-to-provider time (D2P), length of stay for discharged patients (LOSD) and the percentage of patients who left the hospital before treatment was complete (LBTC).
In a multivariate analysis, increased boarding time was significantly associated with longer D2P time, LOSD time, and rates of LBTC.
Overall, D2P and LOSD increased by 0.8 minutes and 2.8 minutes, respectively, for each additional 10 minutes of boarding time. LBTC rates increased by 0.1% for each additional 10 minutes of boarding time.
However, boarding did not have a significant impact on operational metrics among hospitals with fewer than 20,000 visits per year.
Although more research is needed, the results indicate that boarding reduces the throughput of nonboarded patients at a ratio of approximately 4:1. The limited impact of ED efficiency measures on operations highlights the need for hospital-based solutions to boarding, Dr. Tyminski concluded.
“Overall, we expected that there would be an association between boarding and reductions in ED intake and operational efficiency,” said Dr. Napoli in an interview. “However, we were surprised the relationship continued to be as strong in a national study of nearly a quarter of all EDs, as it did in our prior local study,” he said. “Every 10 minutes of boarding in an ED is associated with an approximate 0.1% increase in LWBS and a 3-minute increase in LOSD. Extrapolating this association across the country, we predicted that nearly one million patients may have potentially not received ED care due to boarding,” he explained. “Not only does this potentially have a huge impact on hospital finances but also the overall health of our patients,” he added.
The key takeaway from the study is that boarding is a hospital capacity management issue, said Dr. Napoli. Hospital leadership must be directly involved in plans to mitigate or eliminate it to the extent possible; until then, boarding will continue to result in inefficient ED operations, he explained.
“As ED providers, we are limited in what we can do, but one area where we might be able to make the most impact is to optimize the care and throughput of the LOSD patients,” Dr. Tyminski said. More research is needed to see if interventions to reduce boarding correspond with equivalent improvements in emergency department intake and improved ED throughput, she noted.
The study received no outside funding. The researchers disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
on the basis of data from nearly 900 facilities presented at the annual meeting of the American College of Emergency Physicians.
The study was important to conduct at this time because ED boarding is significantly limiting ED physicians to provide optimal care, said Camila Tyminski, MD, of Brown University, Providence, R.I., who presented the findings at the meeting.
“Boarding had steadily been rising prior to the COVID-19 pandemic due to increased ED use. As our data show, boarding had a detrimental impact on ED throughput measures, including increased door to provider time, increased length of stay of the patient discharged from the ED, and increased rate of patients that left before completion of treatment,” she said.
“It was important to understand these trends prior to 2019-2020 because the COVID-19 pandemic and national nursing shortage have drastically worsened boarding. This study provided a framework for future studies on boarding across ED’s nationally since the start of the pandemic,” she added.
“Post-pandemic, we have hit a crisis point,” lead author Anthony Napoli, MD, also of Brown University, said in an interview. “Boarding is largely a hospital capacity problem, but one key fix germane to EM [emergency medicine] is the provider in triage model (PIT). While PIT has been shown to improve efficiency of ED care, a single institution study demonstrated that it was unable to mitigate the effects of boarding. The study of the association of boarding and efficiency of ED operations and intake needed to be shown on a national scale,” he said.
The researchers reviewed cross-sectional ED operational data from the ED Department Benchmarking Alliance (EDBA), a voluntary database that includes self-reports of operational metrics from approximately half of EDs in the United States.
The data set included 892 EDs; freestanding and pediatric EDs, as well as those with missing boarding data, were excluded.
The primary outcome was boarding time, door-to-provider time (D2P), length of stay for discharged patients (LOSD) and the percentage of patients who left the hospital before treatment was complete (LBTC).
In a multivariate analysis, increased boarding time was significantly associated with longer D2P time, LOSD time, and rates of LBTC.
Overall, D2P and LOSD increased by 0.8 minutes and 2.8 minutes, respectively, for each additional 10 minutes of boarding time. LBTC rates increased by 0.1% for each additional 10 minutes of boarding time.
However, boarding did not have a significant impact on operational metrics among hospitals with fewer than 20,000 visits per year.
Although more research is needed, the results indicate that boarding reduces the throughput of nonboarded patients at a ratio of approximately 4:1. The limited impact of ED efficiency measures on operations highlights the need for hospital-based solutions to boarding, Dr. Tyminski concluded.
“Overall, we expected that there would be an association between boarding and reductions in ED intake and operational efficiency,” said Dr. Napoli in an interview. “However, we were surprised the relationship continued to be as strong in a national study of nearly a quarter of all EDs, as it did in our prior local study,” he said. “Every 10 minutes of boarding in an ED is associated with an approximate 0.1% increase in LWBS and a 3-minute increase in LOSD. Extrapolating this association across the country, we predicted that nearly one million patients may have potentially not received ED care due to boarding,” he explained. “Not only does this potentially have a huge impact on hospital finances but also the overall health of our patients,” he added.
The key takeaway from the study is that boarding is a hospital capacity management issue, said Dr. Napoli. Hospital leadership must be directly involved in plans to mitigate or eliminate it to the extent possible; until then, boarding will continue to result in inefficient ED operations, he explained.
“As ED providers, we are limited in what we can do, but one area where we might be able to make the most impact is to optimize the care and throughput of the LOSD patients,” Dr. Tyminski said. More research is needed to see if interventions to reduce boarding correspond with equivalent improvements in emergency department intake and improved ED throughput, she noted.
The study received no outside funding. The researchers disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ACEP 2022








