User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
In epilepsy, heart issues linked to longer disease duration
, but little is known about how they progress. A new study finds that abnormalities in electrocardiograms are linked to an earlier age of diagnosis and longer epilepsy duration.
The findings could help researchers in the search for biomarkers that could predict later problems in children with epilepsy. “In pediatric neurology I think we’re a little bit removed from some of the cardiovascular complications that can happen within epilepsy, but cardiovascular complications are well established, especially in adults that have epilepsy. Adults with epilepsy are more likely to have coronary artery disease, atherosclerosis, arrhythmias, heart attacks, and sudden cardiac death. It’s a pretty substantial difference compared with their nonepileptic peers. So knowing that, the big question is, how do these changes develop, and how do we really counsel our patients in regards to these complications?” said Brittnie Bartlett, MD, during her presentation of the research at the 2022 annual meeting of the Child Neurology Society.
Identifying factors that increase cardiac complications
Previous studies suggested that epilepsy duration might be linked to cardiovascular complications. In children with Dravet syndrome, epilepsy duration has been shown to be associated with cardiac complications. Pathological T wave alternans, which indicates ventricular instability, has been observed in adults with longstanding epilepsy but not adults with newly diagnosed epilepsy.
“So our question in this preliminary report of our data is: What factors in our general pediatric epilepsy cohort can we identify that put them at a greater risk for having EKG changes, and specifically, we wanted to verify these findings from the other studies that epilepsy duration is, in fact, a risk factor for these EKG changes in general [among children] with epilepsy aside from channelopathies,” said Dr. Bartlett, who is an assistant professor at Baylor College of Medicine and a child neurologist at Texas Children’s Hospital, both in Houston.
She presented a striking finding that cardiovascular changes appear early. “The most important thing I want you all to make note of is the fact that, in this baseline study that we got on these kids, 47% already had changes that we were seeing on their EKGs,” said Dr. Bartlett.
The researchers also looked for factors associated with EKG changes, and found that duration of epilepsy and age at diagnosis were the two salient factors. “Our kids that did have EKG changes present had an average epilepsy duration of 73 months, as opposed to [the children] that did not have EKG changes and had an average epilepsy duration of 46 months,” said Dr. Bartlett.
Other factors, such epilepsy type, etiology, refractory epilepsy, and seizure frequency had no statistically significant association with EKG changes. They also saw no associations with high-risk seizure medications, even though some antiseizure drugs have been shown to be linked to EKG changes.
“We were able to confirm our hypothesis that EKG changes were more prevalent with longer duration of epilepsy. Unfortunately, we weren’t able to find any other clues that would help us counsel our patients, but this is part of a longitudinal prospective study that we’ll be following these kids over a couple of years’ time, so maybe we’ll be able to tease out some of these differences. Ideally, we’d be able to find some kind of a biomarker for future cardiovascular complications, and right now we’re working with some multivariable models to verify some of these findings,” said Dr. Bartlett.
Implications for clinical practice
During the Q&A, Dr. Bartlett was asked if all kids with epilepsy should undergo an EKG. She recommended against it for now. “At this point, I don’t think we have enough clear data to support getting an EKG on every kid with epilepsy. I do think it’s good practice to do them on all kids with channelopathies. As a general practice, I tend to have a low threshold towards many kids with epilepsy, but a lot of these cardiovascular risk factors tend to pop up more in adulthood, so it’s more preventative,” she said.
Grace Gombolay, MD, who moderated the session where the poster was presented, was asked for comment on the study. “What’s surprising about it is that up to half of patients actually had EKG changes, different what from what we see in normal population, and it’s interesting to think about the implications. One of the things that our epilepsy patients are at risk for is SUDEP – sudden, unexplained death in epilepsy. It’s interesting to think about what these EKG changes mean for clinical care. I think it’s too early to say at this time, but this might be one of those markers for SUDEP,” said Dr. Gombolay, who is an assistant professor at Emory University, Atlanta, and director of the Pediatric Neuroimmunology and Multiple Sclerosis Clinic at Children’s Healthcare of Atlanta.
The researchers prospectively studied 213 patients who were recruited. 46% were female, 42% were white, 41% were Hispanic, and 13% were African American. The mean age at enrollment was 116 months, and mean age of seizure onset was 45 months.
The researchers found that 47% had abnormal EKG readings. None of the changes were pathologic, but they may reflect changes to cardiac electrophysiology, according to Dr. Bartlett. Those with abnormal readings were older on average (11.6 vs. 8.3 years; P < .005) and had a longer epilepsy duration (73 vs. 46 months; P = .004).
Dr. Gombolay has no relevant financial disclosures.
, but little is known about how they progress. A new study finds that abnormalities in electrocardiograms are linked to an earlier age of diagnosis and longer epilepsy duration.
The findings could help researchers in the search for biomarkers that could predict later problems in children with epilepsy. “In pediatric neurology I think we’re a little bit removed from some of the cardiovascular complications that can happen within epilepsy, but cardiovascular complications are well established, especially in adults that have epilepsy. Adults with epilepsy are more likely to have coronary artery disease, atherosclerosis, arrhythmias, heart attacks, and sudden cardiac death. It’s a pretty substantial difference compared with their nonepileptic peers. So knowing that, the big question is, how do these changes develop, and how do we really counsel our patients in regards to these complications?” said Brittnie Bartlett, MD, during her presentation of the research at the 2022 annual meeting of the Child Neurology Society.
Identifying factors that increase cardiac complications
Previous studies suggested that epilepsy duration might be linked to cardiovascular complications. In children with Dravet syndrome, epilepsy duration has been shown to be associated with cardiac complications. Pathological T wave alternans, which indicates ventricular instability, has been observed in adults with longstanding epilepsy but not adults with newly diagnosed epilepsy.
“So our question in this preliminary report of our data is: What factors in our general pediatric epilepsy cohort can we identify that put them at a greater risk for having EKG changes, and specifically, we wanted to verify these findings from the other studies that epilepsy duration is, in fact, a risk factor for these EKG changes in general [among children] with epilepsy aside from channelopathies,” said Dr. Bartlett, who is an assistant professor at Baylor College of Medicine and a child neurologist at Texas Children’s Hospital, both in Houston.
She presented a striking finding that cardiovascular changes appear early. “The most important thing I want you all to make note of is the fact that, in this baseline study that we got on these kids, 47% already had changes that we were seeing on their EKGs,” said Dr. Bartlett.
The researchers also looked for factors associated with EKG changes, and found that duration of epilepsy and age at diagnosis were the two salient factors. “Our kids that did have EKG changes present had an average epilepsy duration of 73 months, as opposed to [the children] that did not have EKG changes and had an average epilepsy duration of 46 months,” said Dr. Bartlett.
Other factors, such epilepsy type, etiology, refractory epilepsy, and seizure frequency had no statistically significant association with EKG changes. They also saw no associations with high-risk seizure medications, even though some antiseizure drugs have been shown to be linked to EKG changes.
“We were able to confirm our hypothesis that EKG changes were more prevalent with longer duration of epilepsy. Unfortunately, we weren’t able to find any other clues that would help us counsel our patients, but this is part of a longitudinal prospective study that we’ll be following these kids over a couple of years’ time, so maybe we’ll be able to tease out some of these differences. Ideally, we’d be able to find some kind of a biomarker for future cardiovascular complications, and right now we’re working with some multivariable models to verify some of these findings,” said Dr. Bartlett.
Implications for clinical practice
During the Q&A, Dr. Bartlett was asked if all kids with epilepsy should undergo an EKG. She recommended against it for now. “At this point, I don’t think we have enough clear data to support getting an EKG on every kid with epilepsy. I do think it’s good practice to do them on all kids with channelopathies. As a general practice, I tend to have a low threshold towards many kids with epilepsy, but a lot of these cardiovascular risk factors tend to pop up more in adulthood, so it’s more preventative,” she said.
Grace Gombolay, MD, who moderated the session where the poster was presented, was asked for comment on the study. “What’s surprising about it is that up to half of patients actually had EKG changes, different what from what we see in normal population, and it’s interesting to think about the implications. One of the things that our epilepsy patients are at risk for is SUDEP – sudden, unexplained death in epilepsy. It’s interesting to think about what these EKG changes mean for clinical care. I think it’s too early to say at this time, but this might be one of those markers for SUDEP,” said Dr. Gombolay, who is an assistant professor at Emory University, Atlanta, and director of the Pediatric Neuroimmunology and Multiple Sclerosis Clinic at Children’s Healthcare of Atlanta.
The researchers prospectively studied 213 patients who were recruited. 46% were female, 42% were white, 41% were Hispanic, and 13% were African American. The mean age at enrollment was 116 months, and mean age of seizure onset was 45 months.
The researchers found that 47% had abnormal EKG readings. None of the changes were pathologic, but they may reflect changes to cardiac electrophysiology, according to Dr. Bartlett. Those with abnormal readings were older on average (11.6 vs. 8.3 years; P < .005) and had a longer epilepsy duration (73 vs. 46 months; P = .004).
Dr. Gombolay has no relevant financial disclosures.
, but little is known about how they progress. A new study finds that abnormalities in electrocardiograms are linked to an earlier age of diagnosis and longer epilepsy duration.
The findings could help researchers in the search for biomarkers that could predict later problems in children with epilepsy. “In pediatric neurology I think we’re a little bit removed from some of the cardiovascular complications that can happen within epilepsy, but cardiovascular complications are well established, especially in adults that have epilepsy. Adults with epilepsy are more likely to have coronary artery disease, atherosclerosis, arrhythmias, heart attacks, and sudden cardiac death. It’s a pretty substantial difference compared with their nonepileptic peers. So knowing that, the big question is, how do these changes develop, and how do we really counsel our patients in regards to these complications?” said Brittnie Bartlett, MD, during her presentation of the research at the 2022 annual meeting of the Child Neurology Society.
Identifying factors that increase cardiac complications
Previous studies suggested that epilepsy duration might be linked to cardiovascular complications. In children with Dravet syndrome, epilepsy duration has been shown to be associated with cardiac complications. Pathological T wave alternans, which indicates ventricular instability, has been observed in adults with longstanding epilepsy but not adults with newly diagnosed epilepsy.
“So our question in this preliminary report of our data is: What factors in our general pediatric epilepsy cohort can we identify that put them at a greater risk for having EKG changes, and specifically, we wanted to verify these findings from the other studies that epilepsy duration is, in fact, a risk factor for these EKG changes in general [among children] with epilepsy aside from channelopathies,” said Dr. Bartlett, who is an assistant professor at Baylor College of Medicine and a child neurologist at Texas Children’s Hospital, both in Houston.
She presented a striking finding that cardiovascular changes appear early. “The most important thing I want you all to make note of is the fact that, in this baseline study that we got on these kids, 47% already had changes that we were seeing on their EKGs,” said Dr. Bartlett.
The researchers also looked for factors associated with EKG changes, and found that duration of epilepsy and age at diagnosis were the two salient factors. “Our kids that did have EKG changes present had an average epilepsy duration of 73 months, as opposed to [the children] that did not have EKG changes and had an average epilepsy duration of 46 months,” said Dr. Bartlett.
Other factors, such epilepsy type, etiology, refractory epilepsy, and seizure frequency had no statistically significant association with EKG changes. They also saw no associations with high-risk seizure medications, even though some antiseizure drugs have been shown to be linked to EKG changes.
“We were able to confirm our hypothesis that EKG changes were more prevalent with longer duration of epilepsy. Unfortunately, we weren’t able to find any other clues that would help us counsel our patients, but this is part of a longitudinal prospective study that we’ll be following these kids over a couple of years’ time, so maybe we’ll be able to tease out some of these differences. Ideally, we’d be able to find some kind of a biomarker for future cardiovascular complications, and right now we’re working with some multivariable models to verify some of these findings,” said Dr. Bartlett.
Implications for clinical practice
During the Q&A, Dr. Bartlett was asked if all kids with epilepsy should undergo an EKG. She recommended against it for now. “At this point, I don’t think we have enough clear data to support getting an EKG on every kid with epilepsy. I do think it’s good practice to do them on all kids with channelopathies. As a general practice, I tend to have a low threshold towards many kids with epilepsy, but a lot of these cardiovascular risk factors tend to pop up more in adulthood, so it’s more preventative,” she said.
Grace Gombolay, MD, who moderated the session where the poster was presented, was asked for comment on the study. “What’s surprising about it is that up to half of patients actually had EKG changes, different what from what we see in normal population, and it’s interesting to think about the implications. One of the things that our epilepsy patients are at risk for is SUDEP – sudden, unexplained death in epilepsy. It’s interesting to think about what these EKG changes mean for clinical care. I think it’s too early to say at this time, but this might be one of those markers for SUDEP,” said Dr. Gombolay, who is an assistant professor at Emory University, Atlanta, and director of the Pediatric Neuroimmunology and Multiple Sclerosis Clinic at Children’s Healthcare of Atlanta.
The researchers prospectively studied 213 patients who were recruited. 46% were female, 42% were white, 41% were Hispanic, and 13% were African American. The mean age at enrollment was 116 months, and mean age of seizure onset was 45 months.
The researchers found that 47% had abnormal EKG readings. None of the changes were pathologic, but they may reflect changes to cardiac electrophysiology, according to Dr. Bartlett. Those with abnormal readings were older on average (11.6 vs. 8.3 years; P < .005) and had a longer epilepsy duration (73 vs. 46 months; P = .004).
Dr. Gombolay has no relevant financial disclosures.
FROM CNS 2022
Ten-day methotrexate pause after COVID vaccine booster enhances immunity against Omicron variant
People taking methotrexate for immunomodulatory diseases can skip one or two scheduled doses after they get an mRNA-based vaccine booster for COVID-19 and achieve a level of immunity against Omicron variants that’s comparable to people who aren’t immunosuppressed, a small observational cohort study from Germany reported.
“In general, the data suggest that pausing methotrexate is feasible, and it’s sufficient if the last dose occurs 1-3 days before the vaccination,” study coauthor Gerd Burmester, MD, a senior professor of rheumatology and immunology at the University of Medicine Berlin, told this news organization. “In pragmatic terms: pausing the methotrexate injection just twice after the vaccine is finished and, interestingly, not prior to the vaccination.”
The study, published online in RMD Open, included a statistical analysis that determined that a 10-day pause after the vaccination would be optimal, Dr. Burmester said.
Dr. Burmester and coauthors claimed this is the first study to evaluate the antibody response in patients on methotrexate against Omicron variants – in this study, variants BA.1 and BA.2 – after getting a COVID-19 mRNA booster. The study compared neutralizing serum activity of 50 patients taking methotrexate – 24 of whom continued treatments uninterrupted and 26 of whom paused treatments after getting a second booster – with 25 nonimmunosuppressed patients who served as controls. A total of 24% of the patients taking methotrexate received the mRNA-1273 vaccine while the entire control group received the Pfizer/BioNTech BNT162b2 vaccine.
The researchers used SARS-CoV-2 pseudovirus neutralization assays to evaluate post-vaccination antibody levels.
The U.S. Centers for Disease Control and Prevention and other government health agencies have recommended that immunocompromised patients get a fourth COVID-19 vaccination. But these vaccines can be problematic in patients taking methotrexate, which was linked to a reduced response after the second and third doses of the COVID-19 vaccine.
Previous studies reported that pausing methotrexate for 10 or 14 days after the first two vaccinations improved the production of neutralizing antibodies. A 2022 study found that a 2-week pause after a booster increased antibody response against S1 RBD (receptor binding domain) of the SARS-CoV-2 spike protein about twofold. Another recently published study of mRNA vaccines found that taking methotrexate with either a biologic or targeted synthetic disease-modifying antirheumatic drug reduces the efficacy of a third (booster) shot of SARS-CoV-2 mRNA vaccine in older adults but not younger patients with RA.
“Our study and also the other studies suggested that you can pause methotrexate treatment safely from a point of view of disease activity of rheumatoid arthritis,” Dr. Burmester said. “If you do the pause just twice or once only, it doesn’t lead to significant flares.”
Study results
The study found that serum neutralizing activity against the Omicron BA.1 variant, measured as geometric mean 50% inhibitory serum dilution (ID50s), wasn’t significantly different between the methotrexate and the nonimmunosuppressed groups before getting their mRNA booster (P = .657). However, 4 weeks after getting the booster, the nonimmunosuppressed group had a 68-fold increase in antibody activity versus a 20-fold increase in the methotrexate patients. After 12 weeks, ID50s in both groups decreased by about half (P = .001).
The methotrexate patients who continued therapy after the booster had significantly lower neutralization against Omicron BA.1 at both 4 weeks and 12 weeks than did their counterparts who paused therapy, as well as control patients.
The results were very similar in the same group comparisons of the serum neutralizing activity against the Omicron BA.2 variant at 4 and 12 weeks after booster vaccination.
Expert commentary
This study is noteworthy because it used SARS-CoV-2 pseudovirus neutralization assays to evaluate antibody levels, Kevin Winthrop, MD, MPH, professor of infectious disease and public health at Oregon Health & Science University, Portland, who was not involved in the study, said. “A lot of studies don’t look at neutralizing antibody titers, and that’s really what we care about,” Dr. Winthrop said. “What we want are functional antibodies that are doing something, and the only way to do that is to test them.”
The study is “confirmatory” of other studies that call for pausing methotrexate after vaccination, Dr. Winthrop said, including a study he coauthored, and which the German researchers cited, that found pausing methotrexate for a week or so after the influenza vaccination in RA patients improved vaccine immunogenicity. He added that the findings with the early Omicron variants are important because the newest boosters target the later Omicron variants, BA.4 and BA.5.
“The bottom line is that when someone comes in for a COVID-19 vaccination, tell them to be off of methotrexate for 7-10 days,” Dr. Winthrop said. “This is for the booster, but it raises the question: If you go out to three, four, or five vaccinations, does this matter anymore? With the flu vaccine, most people are out to 10 or 15 boosters, and we haven’t seen any significant increase in disease flares.”
The study received funding from Medac, Gilead/Galapagos, and Friends and Sponsors of Berlin Charity. Dr. Burmester reported no relevant disclosures. Dr. Winthrop is a research consultant to Pfizer.
A version of this article first appeared on Medscape.com.
People taking methotrexate for immunomodulatory diseases can skip one or two scheduled doses after they get an mRNA-based vaccine booster for COVID-19 and achieve a level of immunity against Omicron variants that’s comparable to people who aren’t immunosuppressed, a small observational cohort study from Germany reported.
“In general, the data suggest that pausing methotrexate is feasible, and it’s sufficient if the last dose occurs 1-3 days before the vaccination,” study coauthor Gerd Burmester, MD, a senior professor of rheumatology and immunology at the University of Medicine Berlin, told this news organization. “In pragmatic terms: pausing the methotrexate injection just twice after the vaccine is finished and, interestingly, not prior to the vaccination.”
The study, published online in RMD Open, included a statistical analysis that determined that a 10-day pause after the vaccination would be optimal, Dr. Burmester said.
Dr. Burmester and coauthors claimed this is the first study to evaluate the antibody response in patients on methotrexate against Omicron variants – in this study, variants BA.1 and BA.2 – after getting a COVID-19 mRNA booster. The study compared neutralizing serum activity of 50 patients taking methotrexate – 24 of whom continued treatments uninterrupted and 26 of whom paused treatments after getting a second booster – with 25 nonimmunosuppressed patients who served as controls. A total of 24% of the patients taking methotrexate received the mRNA-1273 vaccine while the entire control group received the Pfizer/BioNTech BNT162b2 vaccine.
The researchers used SARS-CoV-2 pseudovirus neutralization assays to evaluate post-vaccination antibody levels.
The U.S. Centers for Disease Control and Prevention and other government health agencies have recommended that immunocompromised patients get a fourth COVID-19 vaccination. But these vaccines can be problematic in patients taking methotrexate, which was linked to a reduced response after the second and third doses of the COVID-19 vaccine.
Previous studies reported that pausing methotrexate for 10 or 14 days after the first two vaccinations improved the production of neutralizing antibodies. A 2022 study found that a 2-week pause after a booster increased antibody response against S1 RBD (receptor binding domain) of the SARS-CoV-2 spike protein about twofold. Another recently published study of mRNA vaccines found that taking methotrexate with either a biologic or targeted synthetic disease-modifying antirheumatic drug reduces the efficacy of a third (booster) shot of SARS-CoV-2 mRNA vaccine in older adults but not younger patients with RA.
“Our study and also the other studies suggested that you can pause methotrexate treatment safely from a point of view of disease activity of rheumatoid arthritis,” Dr. Burmester said. “If you do the pause just twice or once only, it doesn’t lead to significant flares.”
Study results
The study found that serum neutralizing activity against the Omicron BA.1 variant, measured as geometric mean 50% inhibitory serum dilution (ID50s), wasn’t significantly different between the methotrexate and the nonimmunosuppressed groups before getting their mRNA booster (P = .657). However, 4 weeks after getting the booster, the nonimmunosuppressed group had a 68-fold increase in antibody activity versus a 20-fold increase in the methotrexate patients. After 12 weeks, ID50s in both groups decreased by about half (P = .001).
The methotrexate patients who continued therapy after the booster had significantly lower neutralization against Omicron BA.1 at both 4 weeks and 12 weeks than did their counterparts who paused therapy, as well as control patients.
The results were very similar in the same group comparisons of the serum neutralizing activity against the Omicron BA.2 variant at 4 and 12 weeks after booster vaccination.
Expert commentary
This study is noteworthy because it used SARS-CoV-2 pseudovirus neutralization assays to evaluate antibody levels, Kevin Winthrop, MD, MPH, professor of infectious disease and public health at Oregon Health & Science University, Portland, who was not involved in the study, said. “A lot of studies don’t look at neutralizing antibody titers, and that’s really what we care about,” Dr. Winthrop said. “What we want are functional antibodies that are doing something, and the only way to do that is to test them.”
The study is “confirmatory” of other studies that call for pausing methotrexate after vaccination, Dr. Winthrop said, including a study he coauthored, and which the German researchers cited, that found pausing methotrexate for a week or so after the influenza vaccination in RA patients improved vaccine immunogenicity. He added that the findings with the early Omicron variants are important because the newest boosters target the later Omicron variants, BA.4 and BA.5.
“The bottom line is that when someone comes in for a COVID-19 vaccination, tell them to be off of methotrexate for 7-10 days,” Dr. Winthrop said. “This is for the booster, but it raises the question: If you go out to three, four, or five vaccinations, does this matter anymore? With the flu vaccine, most people are out to 10 or 15 boosters, and we haven’t seen any significant increase in disease flares.”
The study received funding from Medac, Gilead/Galapagos, and Friends and Sponsors of Berlin Charity. Dr. Burmester reported no relevant disclosures. Dr. Winthrop is a research consultant to Pfizer.
A version of this article first appeared on Medscape.com.
People taking methotrexate for immunomodulatory diseases can skip one or two scheduled doses after they get an mRNA-based vaccine booster for COVID-19 and achieve a level of immunity against Omicron variants that’s comparable to people who aren’t immunosuppressed, a small observational cohort study from Germany reported.
“In general, the data suggest that pausing methotrexate is feasible, and it’s sufficient if the last dose occurs 1-3 days before the vaccination,” study coauthor Gerd Burmester, MD, a senior professor of rheumatology and immunology at the University of Medicine Berlin, told this news organization. “In pragmatic terms: pausing the methotrexate injection just twice after the vaccine is finished and, interestingly, not prior to the vaccination.”
The study, published online in RMD Open, included a statistical analysis that determined that a 10-day pause after the vaccination would be optimal, Dr. Burmester said.
Dr. Burmester and coauthors claimed this is the first study to evaluate the antibody response in patients on methotrexate against Omicron variants – in this study, variants BA.1 and BA.2 – after getting a COVID-19 mRNA booster. The study compared neutralizing serum activity of 50 patients taking methotrexate – 24 of whom continued treatments uninterrupted and 26 of whom paused treatments after getting a second booster – with 25 nonimmunosuppressed patients who served as controls. A total of 24% of the patients taking methotrexate received the mRNA-1273 vaccine while the entire control group received the Pfizer/BioNTech BNT162b2 vaccine.
The researchers used SARS-CoV-2 pseudovirus neutralization assays to evaluate post-vaccination antibody levels.
The U.S. Centers for Disease Control and Prevention and other government health agencies have recommended that immunocompromised patients get a fourth COVID-19 vaccination. But these vaccines can be problematic in patients taking methotrexate, which was linked to a reduced response after the second and third doses of the COVID-19 vaccine.
Previous studies reported that pausing methotrexate for 10 or 14 days after the first two vaccinations improved the production of neutralizing antibodies. A 2022 study found that a 2-week pause after a booster increased antibody response against S1 RBD (receptor binding domain) of the SARS-CoV-2 spike protein about twofold. Another recently published study of mRNA vaccines found that taking methotrexate with either a biologic or targeted synthetic disease-modifying antirheumatic drug reduces the efficacy of a third (booster) shot of SARS-CoV-2 mRNA vaccine in older adults but not younger patients with RA.
“Our study and also the other studies suggested that you can pause methotrexate treatment safely from a point of view of disease activity of rheumatoid arthritis,” Dr. Burmester said. “If you do the pause just twice or once only, it doesn’t lead to significant flares.”
Study results
The study found that serum neutralizing activity against the Omicron BA.1 variant, measured as geometric mean 50% inhibitory serum dilution (ID50s), wasn’t significantly different between the methotrexate and the nonimmunosuppressed groups before getting their mRNA booster (P = .657). However, 4 weeks after getting the booster, the nonimmunosuppressed group had a 68-fold increase in antibody activity versus a 20-fold increase in the methotrexate patients. After 12 weeks, ID50s in both groups decreased by about half (P = .001).
The methotrexate patients who continued therapy after the booster had significantly lower neutralization against Omicron BA.1 at both 4 weeks and 12 weeks than did their counterparts who paused therapy, as well as control patients.
The results were very similar in the same group comparisons of the serum neutralizing activity against the Omicron BA.2 variant at 4 and 12 weeks after booster vaccination.
Expert commentary
This study is noteworthy because it used SARS-CoV-2 pseudovirus neutralization assays to evaluate antibody levels, Kevin Winthrop, MD, MPH, professor of infectious disease and public health at Oregon Health & Science University, Portland, who was not involved in the study, said. “A lot of studies don’t look at neutralizing antibody titers, and that’s really what we care about,” Dr. Winthrop said. “What we want are functional antibodies that are doing something, and the only way to do that is to test them.”
The study is “confirmatory” of other studies that call for pausing methotrexate after vaccination, Dr. Winthrop said, including a study he coauthored, and which the German researchers cited, that found pausing methotrexate for a week or so after the influenza vaccination in RA patients improved vaccine immunogenicity. He added that the findings with the early Omicron variants are important because the newest boosters target the later Omicron variants, BA.4 and BA.5.
“The bottom line is that when someone comes in for a COVID-19 vaccination, tell them to be off of methotrexate for 7-10 days,” Dr. Winthrop said. “This is for the booster, but it raises the question: If you go out to three, four, or five vaccinations, does this matter anymore? With the flu vaccine, most people are out to 10 or 15 boosters, and we haven’t seen any significant increase in disease flares.”
The study received funding from Medac, Gilead/Galapagos, and Friends and Sponsors of Berlin Charity. Dr. Burmester reported no relevant disclosures. Dr. Winthrop is a research consultant to Pfizer.
A version of this article first appeared on Medscape.com.
FROM RMD OPEN
Why the 5-day isolation period for COVID makes no sense
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
One of the more baffling decisions the CDC made during this pandemic was when they reduced the duration of isolation after a positive COVID test from 10 days to 5 days and did not require a negative antigen test to end isolation.
Multiple studies had suggested, after all, that positive antigen tests, while not perfect, were a decent proxy for infectivity. And if the purpose of isolation is to keep other community members safe, why not use a readily available test to know when it might be safe to go out in public again?
Also, 5 days just wasn’t that much time. Many individuals are symptomatic long after that point. Many people test positive long after that point. What exactly is the point of the 5-day isolation period?
We got some hard numbers this week to show just how good (or bad) an arbitrary-seeming 5-day isolation period is, thanks to this study from JAMA Network Open, which gives us a low-end estimate for the proportion of people who remain positive on antigen tests, which is to say infectious, after an isolation period.
This study estimates the low end of postisolation infectivity because of the study population: student athletes at an NCAA Division I school, which may or may not be Stanford. These athletes tested positive for COVID after having at least one dose of vaccine from January to May 2022. School protocol was to put the students in isolation for 7 days, at which time they could “test out” with a negative antigen test.
Put simply, these were healthy people. They were young. They were athletes. They were vaccinated. If anyone is going to have a brief, easy COVID course, it would be them. And they are doing at least a week of isolation, not 5 days.
So – isolation for 7 days. Antigen testing on day 7. How many still tested positive? Of 248 individuals tested, 67 (27%) tested positive. One in four.
More than half of those positive on day 7 tested positive on day 8, and more than half of those tested positive again on day 9. By day 10, they were released from isolation without further testing.
So, right there .
There were some predictors of prolonged positivity.
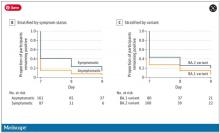
Symptomatic athletes were much more likely to test positive than asymptomatic athletes.
And the particular variant seemed to matter as well. In this time period, BA.1 and BA.2 were dominant, and it was pretty clear that BA.2 persisted longer than BA.1.
This brings me back to my original question: What is the point of the 5-day isolation period? On the basis of this study, you could imagine a guideline based on symptoms: Stay home until you feel better. You could imagine a guideline based on testing: Stay home until you test negative. A guideline based on time alone just doesn’t comport with the data. The benefit of policies based on symptoms or testing are obvious; some people would be out of isolation even before 5 days. But the downside, of course, is that some people would be stuck in isolation for much longer.
Maybe we should just say it. At this point, you could even imagine there being no recommendation at all – no isolation period. Like, you just stay home if you feel like you should stay home. I’m not entirely sure that such a policy would necessarily result in a greater number of infectious people out in the community.
In any case, as the arbitrariness of this particular 5-day isolation policy becomes more clear, the policy itself may be living on borrowed time.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and on Medscape. He tweets @fperrywilson and hosts a repository of his communication work at www.methodsman.com. He disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
One of the more baffling decisions the CDC made during this pandemic was when they reduced the duration of isolation after a positive COVID test from 10 days to 5 days and did not require a negative antigen test to end isolation.
Multiple studies had suggested, after all, that positive antigen tests, while not perfect, were a decent proxy for infectivity. And if the purpose of isolation is to keep other community members safe, why not use a readily available test to know when it might be safe to go out in public again?
Also, 5 days just wasn’t that much time. Many individuals are symptomatic long after that point. Many people test positive long after that point. What exactly is the point of the 5-day isolation period?
We got some hard numbers this week to show just how good (or bad) an arbitrary-seeming 5-day isolation period is, thanks to this study from JAMA Network Open, which gives us a low-end estimate for the proportion of people who remain positive on antigen tests, which is to say infectious, after an isolation period.
This study estimates the low end of postisolation infectivity because of the study population: student athletes at an NCAA Division I school, which may or may not be Stanford. These athletes tested positive for COVID after having at least one dose of vaccine from January to May 2022. School protocol was to put the students in isolation for 7 days, at which time they could “test out” with a negative antigen test.
Put simply, these were healthy people. They were young. They were athletes. They were vaccinated. If anyone is going to have a brief, easy COVID course, it would be them. And they are doing at least a week of isolation, not 5 days.
So – isolation for 7 days. Antigen testing on day 7. How many still tested positive? Of 248 individuals tested, 67 (27%) tested positive. One in four.
More than half of those positive on day 7 tested positive on day 8, and more than half of those tested positive again on day 9. By day 10, they were released from isolation without further testing.
So, right there .
There were some predictors of prolonged positivity.
Symptomatic athletes were much more likely to test positive than asymptomatic athletes.
And the particular variant seemed to matter as well. In this time period, BA.1 and BA.2 were dominant, and it was pretty clear that BA.2 persisted longer than BA.1.
This brings me back to my original question: What is the point of the 5-day isolation period? On the basis of this study, you could imagine a guideline based on symptoms: Stay home until you feel better. You could imagine a guideline based on testing: Stay home until you test negative. A guideline based on time alone just doesn’t comport with the data. The benefit of policies based on symptoms or testing are obvious; some people would be out of isolation even before 5 days. But the downside, of course, is that some people would be stuck in isolation for much longer.
Maybe we should just say it. At this point, you could even imagine there being no recommendation at all – no isolation period. Like, you just stay home if you feel like you should stay home. I’m not entirely sure that such a policy would necessarily result in a greater number of infectious people out in the community.
In any case, as the arbitrariness of this particular 5-day isolation policy becomes more clear, the policy itself may be living on borrowed time.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and on Medscape. He tweets @fperrywilson and hosts a repository of his communication work at www.methodsman.com. He disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
One of the more baffling decisions the CDC made during this pandemic was when they reduced the duration of isolation after a positive COVID test from 10 days to 5 days and did not require a negative antigen test to end isolation.
Multiple studies had suggested, after all, that positive antigen tests, while not perfect, were a decent proxy for infectivity. And if the purpose of isolation is to keep other community members safe, why not use a readily available test to know when it might be safe to go out in public again?
Also, 5 days just wasn’t that much time. Many individuals are symptomatic long after that point. Many people test positive long after that point. What exactly is the point of the 5-day isolation period?
We got some hard numbers this week to show just how good (or bad) an arbitrary-seeming 5-day isolation period is, thanks to this study from JAMA Network Open, which gives us a low-end estimate for the proportion of people who remain positive on antigen tests, which is to say infectious, after an isolation period.
This study estimates the low end of postisolation infectivity because of the study population: student athletes at an NCAA Division I school, which may or may not be Stanford. These athletes tested positive for COVID after having at least one dose of vaccine from January to May 2022. School protocol was to put the students in isolation for 7 days, at which time they could “test out” with a negative antigen test.
Put simply, these were healthy people. They were young. They were athletes. They were vaccinated. If anyone is going to have a brief, easy COVID course, it would be them. And they are doing at least a week of isolation, not 5 days.
So – isolation for 7 days. Antigen testing on day 7. How many still tested positive? Of 248 individuals tested, 67 (27%) tested positive. One in four.
More than half of those positive on day 7 tested positive on day 8, and more than half of those tested positive again on day 9. By day 10, they were released from isolation without further testing.
So, right there .
There were some predictors of prolonged positivity.
Symptomatic athletes were much more likely to test positive than asymptomatic athletes.
And the particular variant seemed to matter as well. In this time period, BA.1 and BA.2 were dominant, and it was pretty clear that BA.2 persisted longer than BA.1.
This brings me back to my original question: What is the point of the 5-day isolation period? On the basis of this study, you could imagine a guideline based on symptoms: Stay home until you feel better. You could imagine a guideline based on testing: Stay home until you test negative. A guideline based on time alone just doesn’t comport with the data. The benefit of policies based on symptoms or testing are obvious; some people would be out of isolation even before 5 days. But the downside, of course, is that some people would be stuck in isolation for much longer.
Maybe we should just say it. At this point, you could even imagine there being no recommendation at all – no isolation period. Like, you just stay home if you feel like you should stay home. I’m not entirely sure that such a policy would necessarily result in a greater number of infectious people out in the community.
In any case, as the arbitrariness of this particular 5-day isolation policy becomes more clear, the policy itself may be living on borrowed time.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and on Medscape. He tweets @fperrywilson and hosts a repository of his communication work at www.methodsman.com. He disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Vaccine adherence hinges on improving science communication
“I’m not getting the vaccine. Nobody knows the long-term effects, and I heard that people are getting clots.”
We were screening patients at a low-cost clinic in Philadelphia for concerns surrounding social determinants of health. During one patient visit, in addition to concerns including housing, medication affordability, and transportation, we found that she had not received the COVID-19 vaccine, and we asked if she was interested in being immunized.
News reports have endlessly covered antivaccine sentiment, but this personal encounter hit home. From simple face masks to groundbreaking vaccines, we failed as physicians to encourage widespread uptake of health-protective measures despite strong scientific backing.
Large swaths of the public deny these tools’ importance or question their safety. This is ultimately rooted in the inability of community leaders and health care professionals to communicate with the public.
Science communication is inherently difficult. Scientists use complex language, and it is hard to evaluate the lay public’s baseline knowledge. Moreover, we are trained to speak with qualifications, encourage doubt, and accept change and evolution of fact. These qualities contrast the definitive messaging necessary in public settings. COVID-19 highlighted these gaps, where regardless of novel scientific solutions, poor communication led to a resistance to accept the tested scientific solution, which ultimately was the rate-limiting factor for overcoming the virus.
As directors of Physician Executive Leadership, an organization that trains future physicians at Thomas Jefferson University to tackle emerging health care issues, we hosted Paul Offit, MD, a national media figure and vaccine advocate. Dr. Offit shared his personal growth during the pandemic, from being abruptly thrown into the spotlight to eventually honing his communication skills. Dr. Offit discussed the challenges of sharing medical knowledge with laypeople and adaptations that are necessary. We found this transformative, realizing the importance of science communication training early in medical education.
Emphasizing the humanities and building soft skills will improve outcomes and benefit broader society by producing physician-leaders in public health and policy. We hope to improve our own communication skills and work in medical education to incorporate similar training into education paradigms for future students.
As seen in our patient interaction, strong science alone will not drive patient adherence; instead, we must work at personal and system levels to induce change. Physicians have a unique opportunity to generate trust and guide evidence-based policy. We must communicate, whether one-on-one with patients, or to millions of viewers via media or policymaker settings. We hope to not only be doctors, but to be advocates, leaders, and trusted advisers for the public.
Mr. Kieran and Mr. Shah are second-year medical students at Sidney Kimmel Medical College, Philadelphia. Neither disclosed any relevant conflicts of interest. A version of this article first appeared on Medscape.com.
“I’m not getting the vaccine. Nobody knows the long-term effects, and I heard that people are getting clots.”
We were screening patients at a low-cost clinic in Philadelphia for concerns surrounding social determinants of health. During one patient visit, in addition to concerns including housing, medication affordability, and transportation, we found that she had not received the COVID-19 vaccine, and we asked if she was interested in being immunized.
News reports have endlessly covered antivaccine sentiment, but this personal encounter hit home. From simple face masks to groundbreaking vaccines, we failed as physicians to encourage widespread uptake of health-protective measures despite strong scientific backing.
Large swaths of the public deny these tools’ importance or question their safety. This is ultimately rooted in the inability of community leaders and health care professionals to communicate with the public.
Science communication is inherently difficult. Scientists use complex language, and it is hard to evaluate the lay public’s baseline knowledge. Moreover, we are trained to speak with qualifications, encourage doubt, and accept change and evolution of fact. These qualities contrast the definitive messaging necessary in public settings. COVID-19 highlighted these gaps, where regardless of novel scientific solutions, poor communication led to a resistance to accept the tested scientific solution, which ultimately was the rate-limiting factor for overcoming the virus.
As directors of Physician Executive Leadership, an organization that trains future physicians at Thomas Jefferson University to tackle emerging health care issues, we hosted Paul Offit, MD, a national media figure and vaccine advocate. Dr. Offit shared his personal growth during the pandemic, from being abruptly thrown into the spotlight to eventually honing his communication skills. Dr. Offit discussed the challenges of sharing medical knowledge with laypeople and adaptations that are necessary. We found this transformative, realizing the importance of science communication training early in medical education.
Emphasizing the humanities and building soft skills will improve outcomes and benefit broader society by producing physician-leaders in public health and policy. We hope to improve our own communication skills and work in medical education to incorporate similar training into education paradigms for future students.
As seen in our patient interaction, strong science alone will not drive patient adherence; instead, we must work at personal and system levels to induce change. Physicians have a unique opportunity to generate trust and guide evidence-based policy. We must communicate, whether one-on-one with patients, or to millions of viewers via media or policymaker settings. We hope to not only be doctors, but to be advocates, leaders, and trusted advisers for the public.
Mr. Kieran and Mr. Shah are second-year medical students at Sidney Kimmel Medical College, Philadelphia. Neither disclosed any relevant conflicts of interest. A version of this article first appeared on Medscape.com.
“I’m not getting the vaccine. Nobody knows the long-term effects, and I heard that people are getting clots.”
We were screening patients at a low-cost clinic in Philadelphia for concerns surrounding social determinants of health. During one patient visit, in addition to concerns including housing, medication affordability, and transportation, we found that she had not received the COVID-19 vaccine, and we asked if she was interested in being immunized.
News reports have endlessly covered antivaccine sentiment, but this personal encounter hit home. From simple face masks to groundbreaking vaccines, we failed as physicians to encourage widespread uptake of health-protective measures despite strong scientific backing.
Large swaths of the public deny these tools’ importance or question their safety. This is ultimately rooted in the inability of community leaders and health care professionals to communicate with the public.
Science communication is inherently difficult. Scientists use complex language, and it is hard to evaluate the lay public’s baseline knowledge. Moreover, we are trained to speak with qualifications, encourage doubt, and accept change and evolution of fact. These qualities contrast the definitive messaging necessary in public settings. COVID-19 highlighted these gaps, where regardless of novel scientific solutions, poor communication led to a resistance to accept the tested scientific solution, which ultimately was the rate-limiting factor for overcoming the virus.
As directors of Physician Executive Leadership, an organization that trains future physicians at Thomas Jefferson University to tackle emerging health care issues, we hosted Paul Offit, MD, a national media figure and vaccine advocate. Dr. Offit shared his personal growth during the pandemic, from being abruptly thrown into the spotlight to eventually honing his communication skills. Dr. Offit discussed the challenges of sharing medical knowledge with laypeople and adaptations that are necessary. We found this transformative, realizing the importance of science communication training early in medical education.
Emphasizing the humanities and building soft skills will improve outcomes and benefit broader society by producing physician-leaders in public health and policy. We hope to improve our own communication skills and work in medical education to incorporate similar training into education paradigms for future students.
As seen in our patient interaction, strong science alone will not drive patient adherence; instead, we must work at personal and system levels to induce change. Physicians have a unique opportunity to generate trust and guide evidence-based policy. We must communicate, whether one-on-one with patients, or to millions of viewers via media or policymaker settings. We hope to not only be doctors, but to be advocates, leaders, and trusted advisers for the public.
Mr. Kieran and Mr. Shah are second-year medical students at Sidney Kimmel Medical College, Philadelphia. Neither disclosed any relevant conflicts of interest. A version of this article first appeared on Medscape.com.
Pregnancy termination counseling for lung disease requires new caution
NASHVILLE, TENN. – In a growing number of states, , according to a panel of experts assembled for a special session at the annual meeting of the American College of Chest Physicians.
Following the June 24 decision by the U.S. Supreme Court to overturn Roe v. Wade, several states were swift to enact tight restrictions on abortion. These restrictions include bans on elective abortions for almost any reason. Worded in various ways, the new laws typically include exceptions when the health of the mother is threatened, but these exceptions must be navigated carefully.
As a general rule, “there is no clear and specific definition of when the mother’s life is at risk. These laws are vague on purpose,” said Rebecca Cohen, MD, division chief, Complex Family Planning, University of Colorado at Denver, Aurora.
The remarks were relevant to any clinician who advises women regarding pregnancy termination, but Dr. Cohen’s advice was tailored to pulmonologists. Advances have reduced the proportion of women with severe lung diseases, such as pulmonary arterial hypertension or interstitial lung disease, that make pregnancy untenable, but serious risks persist.
Clinicians need to assume a defensive posture, and the first step is to understand the laws, according to Dr. Cohen. For this, she recommended the nongovernmental Guttmacher Institute as a resource. With a focus on sexual and reproductive health, this research institute maintains a state-by-state summary of laws that govern pregnancy termination. The laws are being reconsidered across the country, and Dr. Cohen said the website updates its summaries accordingly.
In states with the most rigorous restrictions, the risks to physicians are substantial. Pulmonologists need to recognize that they might face legal consequences from merely advising a patient to terminate her pregnancy if the medical need is ambiguous or unclear, according to Dr. Cohen.
“If the advice is interpreted as aiding and abetting an elective abortion, it is a felony offense in some states,” Dr. Cohen said.
In states with restrictive laws, pregnancy prevention is the safest approach for women of childbearing age who face life-threatening complications in the event of pregnancy, according to Dr. Cohen. This might reasonably include a step beyond standard contraception. Dr. Cohen mentioned such approaches as period tracking to double down.
In addition, for women of childbearing age with health problems that might result in complications in the event of a pregnancy, it is appropriate to establish this fact in the medical record. This history could prove useful for maximizing options when making decisions in the best interest of the mother’s health in the event of contraception failure.
In addition, pulmonologists who counsel women about the potential for pregnancy termination should consider establishing a relationship with the legal department at the institution where they work, according to Dr. Cohen. In specific cases in which termination is recommended, she further advised building documentation with participation from additional medical specialists, such as an obstetrician who manages high-risk pregnancies.
“There is no guarantee that any given documentation is adequate,” Dr. Cohen warned. She indicated that consensus from multiple clinicians can strengthen the legal defense if one is necessary.
For some serious lung conditions that are incompatible with pregnancy, the threat to the mother’s life can occur early, according to Deborah Jo Levine, MD, a clinical instructor in the division of pulmonary, allergy, and critical care medicine, Stanford (Calif.) University.
As a result, “you need to identify at-risk patients early and develop a plan promptly,” said Dr. Levine, who joined Dr. Cohen on the special panel at the CHEST 2022 meeting. Even when termination is medically appropriate, restrictive laws are making these services harder to find.
In the case of a pregnancy likely to pose a high risk of complications owing to the patient’s having lung disease, “it is important to involve a high-risk ob quickly,” Dr. Levine warned. “In some cases, termination poses less risk if performed early.”
Sunjay R. Devarajan, MD, assistant professor of pulmonary medicine and critical care, Baylor College of Medicine, Houston, has faced this issue in a state that has some of the most restrictive laws. Even when there is no debate about the necessity of a medically indicated abortion, he cautioned that abortion services are becoming harder to find.
“A recent patient who had a complicated unintentional pregnancy on our service had to go out of state for pregnancy termination,” Dr. Devarajan said. He noted that this option is not available to all women, particularly in states such as his own in which most bordering states also now have highly restrictive abortion laws.
On the basis of this experience, he is thinking more defensively. Now that clinicians can be drawn into legal proceedings even when pregnancy termination is indicated, he agreed that clinicians must become familiar with the local laws.
“We are doing better in managing pregnancies in women with serious lung diseases, but termination is still the prudent approach in some cases,” Dr. Devarajan said. He indicated that he considered the advice offered by Dr. Cohen helpful in avoiding complications for the patient and the physician.
Dr. Cohen, Dr. Levine, and Dr. Devarajan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
NASHVILLE, TENN. – In a growing number of states, , according to a panel of experts assembled for a special session at the annual meeting of the American College of Chest Physicians.
Following the June 24 decision by the U.S. Supreme Court to overturn Roe v. Wade, several states were swift to enact tight restrictions on abortion. These restrictions include bans on elective abortions for almost any reason. Worded in various ways, the new laws typically include exceptions when the health of the mother is threatened, but these exceptions must be navigated carefully.
As a general rule, “there is no clear and specific definition of when the mother’s life is at risk. These laws are vague on purpose,” said Rebecca Cohen, MD, division chief, Complex Family Planning, University of Colorado at Denver, Aurora.
The remarks were relevant to any clinician who advises women regarding pregnancy termination, but Dr. Cohen’s advice was tailored to pulmonologists. Advances have reduced the proportion of women with severe lung diseases, such as pulmonary arterial hypertension or interstitial lung disease, that make pregnancy untenable, but serious risks persist.
Clinicians need to assume a defensive posture, and the first step is to understand the laws, according to Dr. Cohen. For this, she recommended the nongovernmental Guttmacher Institute as a resource. With a focus on sexual and reproductive health, this research institute maintains a state-by-state summary of laws that govern pregnancy termination. The laws are being reconsidered across the country, and Dr. Cohen said the website updates its summaries accordingly.
In states with the most rigorous restrictions, the risks to physicians are substantial. Pulmonologists need to recognize that they might face legal consequences from merely advising a patient to terminate her pregnancy if the medical need is ambiguous or unclear, according to Dr. Cohen.
“If the advice is interpreted as aiding and abetting an elective abortion, it is a felony offense in some states,” Dr. Cohen said.
In states with restrictive laws, pregnancy prevention is the safest approach for women of childbearing age who face life-threatening complications in the event of pregnancy, according to Dr. Cohen. This might reasonably include a step beyond standard contraception. Dr. Cohen mentioned such approaches as period tracking to double down.
In addition, for women of childbearing age with health problems that might result in complications in the event of a pregnancy, it is appropriate to establish this fact in the medical record. This history could prove useful for maximizing options when making decisions in the best interest of the mother’s health in the event of contraception failure.
In addition, pulmonologists who counsel women about the potential for pregnancy termination should consider establishing a relationship with the legal department at the institution where they work, according to Dr. Cohen. In specific cases in which termination is recommended, she further advised building documentation with participation from additional medical specialists, such as an obstetrician who manages high-risk pregnancies.
“There is no guarantee that any given documentation is adequate,” Dr. Cohen warned. She indicated that consensus from multiple clinicians can strengthen the legal defense if one is necessary.
For some serious lung conditions that are incompatible with pregnancy, the threat to the mother’s life can occur early, according to Deborah Jo Levine, MD, a clinical instructor in the division of pulmonary, allergy, and critical care medicine, Stanford (Calif.) University.
As a result, “you need to identify at-risk patients early and develop a plan promptly,” said Dr. Levine, who joined Dr. Cohen on the special panel at the CHEST 2022 meeting. Even when termination is medically appropriate, restrictive laws are making these services harder to find.
In the case of a pregnancy likely to pose a high risk of complications owing to the patient’s having lung disease, “it is important to involve a high-risk ob quickly,” Dr. Levine warned. “In some cases, termination poses less risk if performed early.”
Sunjay R. Devarajan, MD, assistant professor of pulmonary medicine and critical care, Baylor College of Medicine, Houston, has faced this issue in a state that has some of the most restrictive laws. Even when there is no debate about the necessity of a medically indicated abortion, he cautioned that abortion services are becoming harder to find.
“A recent patient who had a complicated unintentional pregnancy on our service had to go out of state for pregnancy termination,” Dr. Devarajan said. He noted that this option is not available to all women, particularly in states such as his own in which most bordering states also now have highly restrictive abortion laws.
On the basis of this experience, he is thinking more defensively. Now that clinicians can be drawn into legal proceedings even when pregnancy termination is indicated, he agreed that clinicians must become familiar with the local laws.
“We are doing better in managing pregnancies in women with serious lung diseases, but termination is still the prudent approach in some cases,” Dr. Devarajan said. He indicated that he considered the advice offered by Dr. Cohen helpful in avoiding complications for the patient and the physician.
Dr. Cohen, Dr. Levine, and Dr. Devarajan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
NASHVILLE, TENN. – In a growing number of states, , according to a panel of experts assembled for a special session at the annual meeting of the American College of Chest Physicians.
Following the June 24 decision by the U.S. Supreme Court to overturn Roe v. Wade, several states were swift to enact tight restrictions on abortion. These restrictions include bans on elective abortions for almost any reason. Worded in various ways, the new laws typically include exceptions when the health of the mother is threatened, but these exceptions must be navigated carefully.
As a general rule, “there is no clear and specific definition of when the mother’s life is at risk. These laws are vague on purpose,” said Rebecca Cohen, MD, division chief, Complex Family Planning, University of Colorado at Denver, Aurora.
The remarks were relevant to any clinician who advises women regarding pregnancy termination, but Dr. Cohen’s advice was tailored to pulmonologists. Advances have reduced the proportion of women with severe lung diseases, such as pulmonary arterial hypertension or interstitial lung disease, that make pregnancy untenable, but serious risks persist.
Clinicians need to assume a defensive posture, and the first step is to understand the laws, according to Dr. Cohen. For this, she recommended the nongovernmental Guttmacher Institute as a resource. With a focus on sexual and reproductive health, this research institute maintains a state-by-state summary of laws that govern pregnancy termination. The laws are being reconsidered across the country, and Dr. Cohen said the website updates its summaries accordingly.
In states with the most rigorous restrictions, the risks to physicians are substantial. Pulmonologists need to recognize that they might face legal consequences from merely advising a patient to terminate her pregnancy if the medical need is ambiguous or unclear, according to Dr. Cohen.
“If the advice is interpreted as aiding and abetting an elective abortion, it is a felony offense in some states,” Dr. Cohen said.
In states with restrictive laws, pregnancy prevention is the safest approach for women of childbearing age who face life-threatening complications in the event of pregnancy, according to Dr. Cohen. This might reasonably include a step beyond standard contraception. Dr. Cohen mentioned such approaches as period tracking to double down.
In addition, for women of childbearing age with health problems that might result in complications in the event of a pregnancy, it is appropriate to establish this fact in the medical record. This history could prove useful for maximizing options when making decisions in the best interest of the mother’s health in the event of contraception failure.
In addition, pulmonologists who counsel women about the potential for pregnancy termination should consider establishing a relationship with the legal department at the institution where they work, according to Dr. Cohen. In specific cases in which termination is recommended, she further advised building documentation with participation from additional medical specialists, such as an obstetrician who manages high-risk pregnancies.
“There is no guarantee that any given documentation is adequate,” Dr. Cohen warned. She indicated that consensus from multiple clinicians can strengthen the legal defense if one is necessary.
For some serious lung conditions that are incompatible with pregnancy, the threat to the mother’s life can occur early, according to Deborah Jo Levine, MD, a clinical instructor in the division of pulmonary, allergy, and critical care medicine, Stanford (Calif.) University.
As a result, “you need to identify at-risk patients early and develop a plan promptly,” said Dr. Levine, who joined Dr. Cohen on the special panel at the CHEST 2022 meeting. Even when termination is medically appropriate, restrictive laws are making these services harder to find.
In the case of a pregnancy likely to pose a high risk of complications owing to the patient’s having lung disease, “it is important to involve a high-risk ob quickly,” Dr. Levine warned. “In some cases, termination poses less risk if performed early.”
Sunjay R. Devarajan, MD, assistant professor of pulmonary medicine and critical care, Baylor College of Medicine, Houston, has faced this issue in a state that has some of the most restrictive laws. Even when there is no debate about the necessity of a medically indicated abortion, he cautioned that abortion services are becoming harder to find.
“A recent patient who had a complicated unintentional pregnancy on our service had to go out of state for pregnancy termination,” Dr. Devarajan said. He noted that this option is not available to all women, particularly in states such as his own in which most bordering states also now have highly restrictive abortion laws.
On the basis of this experience, he is thinking more defensively. Now that clinicians can be drawn into legal proceedings even when pregnancy termination is indicated, he agreed that clinicians must become familiar with the local laws.
“We are doing better in managing pregnancies in women with serious lung diseases, but termination is still the prudent approach in some cases,” Dr. Devarajan said. He indicated that he considered the advice offered by Dr. Cohen helpful in avoiding complications for the patient and the physician.
Dr. Cohen, Dr. Levine, and Dr. Devarajan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Research fails to justify post-COVID-19 wave of new-onset parkinsonism
, a multinational team of researchers reported at the International Congress of Parkinson’s Disease and Movement Disorders.
SARS-CoV-2 led to numerous discussions about a potential post–COVID-19 emergence of new-onset parkinsonism in susceptible individuals, often referred to in the literature as a “perfect storm” or a “wave” of parkinsonism, according to lead study author Iro Boura, MD.
Postviral precedence
“Although pathogens have been associated both with parkinsonism cases and Parkinson’s disease pathogenesis, the main concern of a potential connection between COVID-19 and new-onset parkinsonism arose from the historically documented parkinsonism cases appearing with encephalitis lethargica,” said Dr. Boura, a PhD candidate with the University of Crete in Greece and ex-fellow at King’s College London.
Encephalitis lethargica appeared between 1916 and 1930 and has been epidemiologically related to the Spanish influenza pandemic, “although this link has been strongly debated by other researchers,” she added.
Because the connection of COVID-19 and parkinsonism seemed highly speculative, Dr. Boura and movement disorder specialist Kallol Ray Chaudhuri DSc, FRCP, MD, decided to search for any data supporting this notion. “Such a possibility would have a significant impact on everyday practice, including long follow-up neurological assessments of COVID-19 patients, along with greater vigilance in recognizing potential symptoms,” said Dr. Boura.
They found no organized research exploring this link, aside from published case reports.
Scant evidence of a parkinsonism wave
The investigators conducted a review of the literature up to February 2022 to identify and analyze published cases of new-onset parkinsonism following a confirmed SARS-CoV-2 infection in otherwise healthy individuals. They ended up with 20 such cases.
Although some cases presented during or shortly after a COVID-19 infection, “the numbers are currently quite low to draw safe conclusions and generalize these findings as a risk of parkinsonism for the general population,” said Dr. Boura. Overall, parkinsonism appeared in the context of encephalopathy in 11 patients. Four patients developed postinfectious parkinsonism without encephalopathy. Another four had phenotypic similarities to idiopathic Parkinson’s disease.
Nine patients were responsive to levodopa, while four required immunomodulatory treatment.
Although cases have already been reported, current data do not yet justify the concept of a post–COVID-19 parkinsonism wave. However, long-term surveillance is crucial to ensure that reports of further cases are carefully documented and analyzed.
Dr. Chaudhuri’s research team recently wrote a book exploring the numerous aspects of COVID-19 and parkinsonism, including Parkinson’s disease, said Dr. Boura.
“Moreover, the COVID-19 Clinical Neuroscience Study (COVID-CNS), with serial follow-up visits for COVID-19 patients, including imaging, is currently running in the United Kingdom with the active participation of Prof Chaudhuri’s team, aiming at revealing any potential parkinsonism cases after a COVID-19 infection,” she said.
, a multinational team of researchers reported at the International Congress of Parkinson’s Disease and Movement Disorders.
SARS-CoV-2 led to numerous discussions about a potential post–COVID-19 emergence of new-onset parkinsonism in susceptible individuals, often referred to in the literature as a “perfect storm” or a “wave” of parkinsonism, according to lead study author Iro Boura, MD.
Postviral precedence
“Although pathogens have been associated both with parkinsonism cases and Parkinson’s disease pathogenesis, the main concern of a potential connection between COVID-19 and new-onset parkinsonism arose from the historically documented parkinsonism cases appearing with encephalitis lethargica,” said Dr. Boura, a PhD candidate with the University of Crete in Greece and ex-fellow at King’s College London.
Encephalitis lethargica appeared between 1916 and 1930 and has been epidemiologically related to the Spanish influenza pandemic, “although this link has been strongly debated by other researchers,” she added.
Because the connection of COVID-19 and parkinsonism seemed highly speculative, Dr. Boura and movement disorder specialist Kallol Ray Chaudhuri DSc, FRCP, MD, decided to search for any data supporting this notion. “Such a possibility would have a significant impact on everyday practice, including long follow-up neurological assessments of COVID-19 patients, along with greater vigilance in recognizing potential symptoms,” said Dr. Boura.
They found no organized research exploring this link, aside from published case reports.
Scant evidence of a parkinsonism wave
The investigators conducted a review of the literature up to February 2022 to identify and analyze published cases of new-onset parkinsonism following a confirmed SARS-CoV-2 infection in otherwise healthy individuals. They ended up with 20 such cases.
Although some cases presented during or shortly after a COVID-19 infection, “the numbers are currently quite low to draw safe conclusions and generalize these findings as a risk of parkinsonism for the general population,” said Dr. Boura. Overall, parkinsonism appeared in the context of encephalopathy in 11 patients. Four patients developed postinfectious parkinsonism without encephalopathy. Another four had phenotypic similarities to idiopathic Parkinson’s disease.
Nine patients were responsive to levodopa, while four required immunomodulatory treatment.
Although cases have already been reported, current data do not yet justify the concept of a post–COVID-19 parkinsonism wave. However, long-term surveillance is crucial to ensure that reports of further cases are carefully documented and analyzed.
Dr. Chaudhuri’s research team recently wrote a book exploring the numerous aspects of COVID-19 and parkinsonism, including Parkinson’s disease, said Dr. Boura.
“Moreover, the COVID-19 Clinical Neuroscience Study (COVID-CNS), with serial follow-up visits for COVID-19 patients, including imaging, is currently running in the United Kingdom with the active participation of Prof Chaudhuri’s team, aiming at revealing any potential parkinsonism cases after a COVID-19 infection,” she said.
, a multinational team of researchers reported at the International Congress of Parkinson’s Disease and Movement Disorders.
SARS-CoV-2 led to numerous discussions about a potential post–COVID-19 emergence of new-onset parkinsonism in susceptible individuals, often referred to in the literature as a “perfect storm” or a “wave” of parkinsonism, according to lead study author Iro Boura, MD.
Postviral precedence
“Although pathogens have been associated both with parkinsonism cases and Parkinson’s disease pathogenesis, the main concern of a potential connection between COVID-19 and new-onset parkinsonism arose from the historically documented parkinsonism cases appearing with encephalitis lethargica,” said Dr. Boura, a PhD candidate with the University of Crete in Greece and ex-fellow at King’s College London.
Encephalitis lethargica appeared between 1916 and 1930 and has been epidemiologically related to the Spanish influenza pandemic, “although this link has been strongly debated by other researchers,” she added.
Because the connection of COVID-19 and parkinsonism seemed highly speculative, Dr. Boura and movement disorder specialist Kallol Ray Chaudhuri DSc, FRCP, MD, decided to search for any data supporting this notion. “Such a possibility would have a significant impact on everyday practice, including long follow-up neurological assessments of COVID-19 patients, along with greater vigilance in recognizing potential symptoms,” said Dr. Boura.
They found no organized research exploring this link, aside from published case reports.
Scant evidence of a parkinsonism wave
The investigators conducted a review of the literature up to February 2022 to identify and analyze published cases of new-onset parkinsonism following a confirmed SARS-CoV-2 infection in otherwise healthy individuals. They ended up with 20 such cases.
Although some cases presented during or shortly after a COVID-19 infection, “the numbers are currently quite low to draw safe conclusions and generalize these findings as a risk of parkinsonism for the general population,” said Dr. Boura. Overall, parkinsonism appeared in the context of encephalopathy in 11 patients. Four patients developed postinfectious parkinsonism without encephalopathy. Another four had phenotypic similarities to idiopathic Parkinson’s disease.
Nine patients were responsive to levodopa, while four required immunomodulatory treatment.
Although cases have already been reported, current data do not yet justify the concept of a post–COVID-19 parkinsonism wave. However, long-term surveillance is crucial to ensure that reports of further cases are carefully documented and analyzed.
Dr. Chaudhuri’s research team recently wrote a book exploring the numerous aspects of COVID-19 and parkinsonism, including Parkinson’s disease, said Dr. Boura.
“Moreover, the COVID-19 Clinical Neuroscience Study (COVID-CNS), with serial follow-up visits for COVID-19 patients, including imaging, is currently running in the United Kingdom with the active participation of Prof Chaudhuri’s team, aiming at revealing any potential parkinsonism cases after a COVID-19 infection,” she said.
FROM MDS 2022
Iron deficiency may protect against bacterial pneumonia
Patients with iron deficiency anemia who developed bacterial pneumonia showed improved outcomes compared to those without iron deficiency anemia, based on data from more than 450,000 individuals in the National Inpatient Sample.
Iron deficiency is the most common nutritional deficiency worldwide, and can lead to anemia, but iron also has been identified as essential to the survival and growth of pathogenic organisms, Mubarak Yusuf, MD, said in a presentation at the annual meeting of the American College of Chest Physicians (CHEST).
However, the specific impact of iron deficiency anemia (IDA) on outcomes in patients hospitalized with acute bacterial infections has not been explored, said Dr. Yusuf, a third-year internal medicine resident at Lincoln Medical Center in New York.
In the study, Dr. Yusuf and colleagues reviewed data from the Nationwide Inpatient Sample (NIS) Database for 2016-2019. They identified 452,040 adults aged 18 or older with a primary diagnosis of bacterial pneumonia based on ICD-10 codes. Patients with a principal diagnosis other than bacterial pneumonia were excluded.
Of these, 5.5% had a secondary diagnosis of IDA. The mean age of the study population was similar between the IDA and non-IDA groups (68 years) and racial distribution was similar, with a White majority of approximately 77%. Slightly more patients in the IDA group were women (58.5% vs. 51.6%) and this difference was statistically significant (P < .00001). Most of the patients (94.6%) in the IDA group had at least three comorbidities, as did 78.1% of the non-IDA group.
The primary outcome was mortality, and the overall mortality in the study population was 2.89%. Although the mortality percentage was higher in the IDA group compared to the non-IDA group (3.25% vs. 2.87%), “when we adjusted for confounders, we noticed a decreased odds of mortality in the IDA group” with an adjusted odds ratio of 0.74 (P = .001), Dr. Yusuf said.
In addition, secondary outcomes of septic shock, acute respiratory failure, and cardiac arrest were lower in the IDA group in a regression analysis, with adjusted odds ratios of 0.71, 0.78, and 0.57, respectively.
The mean length of stay was 0.3 days higher in the IDA group, and the researchers found a nonsignificant increase in total hospital costs of $402.5 for IDA patients compared to those without IDA, said Dr. Yusuf.
The take-home message from the study is actually a question to the clinician, Dr. Yusuf said. “Should you consider a delay in treatment [of iron deficiency anemia] if the patient is not symptomatic?” he asked.
More research is needed to investigate the improved outcomes in the iron deficient population, but the large sample size supports an association that is worth exploring, he concluded.
“The findings of this research may suggest a protective effect of iron deficiency in acute bacterial pneumonia,” Dr. Yusuf said in a press release accompanying the meeting presentation. “More research is needed to elucidate the improved outcomes found in this population, but this research may lead clinicians to consider a delay in treatment of nonsymptomatic iron deficiency in acute bacterial infection,” he added.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Patients with iron deficiency anemia who developed bacterial pneumonia showed improved outcomes compared to those without iron deficiency anemia, based on data from more than 450,000 individuals in the National Inpatient Sample.
Iron deficiency is the most common nutritional deficiency worldwide, and can lead to anemia, but iron also has been identified as essential to the survival and growth of pathogenic organisms, Mubarak Yusuf, MD, said in a presentation at the annual meeting of the American College of Chest Physicians (CHEST).
However, the specific impact of iron deficiency anemia (IDA) on outcomes in patients hospitalized with acute bacterial infections has not been explored, said Dr. Yusuf, a third-year internal medicine resident at Lincoln Medical Center in New York.
In the study, Dr. Yusuf and colleagues reviewed data from the Nationwide Inpatient Sample (NIS) Database for 2016-2019. They identified 452,040 adults aged 18 or older with a primary diagnosis of bacterial pneumonia based on ICD-10 codes. Patients with a principal diagnosis other than bacterial pneumonia were excluded.
Of these, 5.5% had a secondary diagnosis of IDA. The mean age of the study population was similar between the IDA and non-IDA groups (68 years) and racial distribution was similar, with a White majority of approximately 77%. Slightly more patients in the IDA group were women (58.5% vs. 51.6%) and this difference was statistically significant (P < .00001). Most of the patients (94.6%) in the IDA group had at least three comorbidities, as did 78.1% of the non-IDA group.
The primary outcome was mortality, and the overall mortality in the study population was 2.89%. Although the mortality percentage was higher in the IDA group compared to the non-IDA group (3.25% vs. 2.87%), “when we adjusted for confounders, we noticed a decreased odds of mortality in the IDA group” with an adjusted odds ratio of 0.74 (P = .001), Dr. Yusuf said.
In addition, secondary outcomes of septic shock, acute respiratory failure, and cardiac arrest were lower in the IDA group in a regression analysis, with adjusted odds ratios of 0.71, 0.78, and 0.57, respectively.
The mean length of stay was 0.3 days higher in the IDA group, and the researchers found a nonsignificant increase in total hospital costs of $402.5 for IDA patients compared to those without IDA, said Dr. Yusuf.
The take-home message from the study is actually a question to the clinician, Dr. Yusuf said. “Should you consider a delay in treatment [of iron deficiency anemia] if the patient is not symptomatic?” he asked.
More research is needed to investigate the improved outcomes in the iron deficient population, but the large sample size supports an association that is worth exploring, he concluded.
“The findings of this research may suggest a protective effect of iron deficiency in acute bacterial pneumonia,” Dr. Yusuf said in a press release accompanying the meeting presentation. “More research is needed to elucidate the improved outcomes found in this population, but this research may lead clinicians to consider a delay in treatment of nonsymptomatic iron deficiency in acute bacterial infection,” he added.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Patients with iron deficiency anemia who developed bacterial pneumonia showed improved outcomes compared to those without iron deficiency anemia, based on data from more than 450,000 individuals in the National Inpatient Sample.
Iron deficiency is the most common nutritional deficiency worldwide, and can lead to anemia, but iron also has been identified as essential to the survival and growth of pathogenic organisms, Mubarak Yusuf, MD, said in a presentation at the annual meeting of the American College of Chest Physicians (CHEST).
However, the specific impact of iron deficiency anemia (IDA) on outcomes in patients hospitalized with acute bacterial infections has not been explored, said Dr. Yusuf, a third-year internal medicine resident at Lincoln Medical Center in New York.
In the study, Dr. Yusuf and colleagues reviewed data from the Nationwide Inpatient Sample (NIS) Database for 2016-2019. They identified 452,040 adults aged 18 or older with a primary diagnosis of bacterial pneumonia based on ICD-10 codes. Patients with a principal diagnosis other than bacterial pneumonia were excluded.
Of these, 5.5% had a secondary diagnosis of IDA. The mean age of the study population was similar between the IDA and non-IDA groups (68 years) and racial distribution was similar, with a White majority of approximately 77%. Slightly more patients in the IDA group were women (58.5% vs. 51.6%) and this difference was statistically significant (P < .00001). Most of the patients (94.6%) in the IDA group had at least three comorbidities, as did 78.1% of the non-IDA group.
The primary outcome was mortality, and the overall mortality in the study population was 2.89%. Although the mortality percentage was higher in the IDA group compared to the non-IDA group (3.25% vs. 2.87%), “when we adjusted for confounders, we noticed a decreased odds of mortality in the IDA group” with an adjusted odds ratio of 0.74 (P = .001), Dr. Yusuf said.
In addition, secondary outcomes of septic shock, acute respiratory failure, and cardiac arrest were lower in the IDA group in a regression analysis, with adjusted odds ratios of 0.71, 0.78, and 0.57, respectively.
The mean length of stay was 0.3 days higher in the IDA group, and the researchers found a nonsignificant increase in total hospital costs of $402.5 for IDA patients compared to those without IDA, said Dr. Yusuf.
The take-home message from the study is actually a question to the clinician, Dr. Yusuf said. “Should you consider a delay in treatment [of iron deficiency anemia] if the patient is not symptomatic?” he asked.
More research is needed to investigate the improved outcomes in the iron deficient population, but the large sample size supports an association that is worth exploring, he concluded.
“The findings of this research may suggest a protective effect of iron deficiency in acute bacterial pneumonia,” Dr. Yusuf said in a press release accompanying the meeting presentation. “More research is needed to elucidate the improved outcomes found in this population, but this research may lead clinicians to consider a delay in treatment of nonsymptomatic iron deficiency in acute bacterial infection,” he added.
The study received no outside funding. The researchers had no financial conflicts to disclose.
FROM CHEST 2022
Reminder that COVID-19 and cancer can be a deadly combo
A new study underscores the importance of COVID-19 and regular COVID-19 testing among adults with a recent cancer diagnosis.
The Indiana statewide study, conducted at the beginning of the pandemic, found that
“This analysis provides additional empirical evidence on the magnitude of risk to patients with cancer whose immune systems are often weakened either by the disease or treatment,” the study team wrote.
The study was published online in JMIR Cancer.
Although evidence has consistently revealed similar findings, the risk of death among unvaccinated people with cancer and COVID-19 has not been nearly as high in previous studies, lead author Brian E. Dixon, PhD, MBA, with Indiana University Richard M. Fairbanks School of Public Health, Indianapolis, said in a statement. Previous studies from China, for instance, reported a two- to threefold greater risk of all-cause mortality among unvaccinated adults with cancer and COVID-19.
A potential reason for this discrepancy, Dr. Dixon noted, is that earlier studies were “generally smaller and made calculations based on data from a single cancer center or health system.”
Another reason is testing for COVID-19 early in the pandemic was limited to symptomatic individuals who may have had more severe infections, possibly leading to an overestimate of the association between SARS-CoV-2 infection, cancer, and all-cause mortality.
In the current analysis, researchers used electronic health records linked to Indiana’s statewide SARS-CoV-2 testing database and state vital records to evaluate the association between SARS-CoV-2 infection and all-cause mortality among 41,924 adults newly diagnosed with cancer between Jan. 1, 2019, and Dec. 31, 2020.
Most people with cancer were White (78.4%) and about half were male. At the time of diagnosis, 17% had one comorbid condition and about 10% had two or more. Most patients had breast cancer (14%), prostate cancer (13%), or melanoma (13%).
During the study period, 2,894 patients (7%) tested positive for SARS-CoV-2.
In multivariate adjusted analysis, the risk of death among those newly diagnosed with cancer increased by 91% (adjusted hazard ratio, 1.91) during the first year of the pandemic before vaccines were available, compared with the year before (January 2019 to Jan. 14, 2020).
During the pandemic period, the risk of death was roughly threefold higher among adults 65 years old and older, compared with adults 18-44 years old (aHR, 3.35).
When looking at the time from a cancer diagnosis to SARS-CoV-2 infection, infection was associated with an almost sevenfold increase in all-cause mortality (aHR, 6.91). Adults 65 years old and older had an almost threefold increased risk of dying, compared with their younger peers (aHR, 2.74).
Dr. Dixon and colleagues also observed an increased risk of death in men with cancer and COVID, compared with women (aHR, 1.23) and those with at least two comorbid conditions versus none (aHR, 2.12). In addition, the risk of dying was 9% higher among Indiana’s rural population than urban dwellers.
Compared with other cancer types, individuals with lung cancer and other digestive cancers had the highest risk of death after SARS-CoV-2 infection (aHR, 1.45 and 1.80, respectively).
“Our findings highlight the increased risk of death for adult cancer patients who test positive for COVID and underscore the importance to cancer patients – including those in remission – of vaccinations, boosters, and regular COVID testing,” Dr. Dixon commented.
“Our results should encourage individuals diagnosed with cancer not only to take preventive action, but also to expeditiously seek out treatments available in the marketplace should they test positive for COVID,” he added.
Support for the study was provided by Indiana University Simon Cancer Center and the Centers for Disease Control and Prevention. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study underscores the importance of COVID-19 and regular COVID-19 testing among adults with a recent cancer diagnosis.
The Indiana statewide study, conducted at the beginning of the pandemic, found that
“This analysis provides additional empirical evidence on the magnitude of risk to patients with cancer whose immune systems are often weakened either by the disease or treatment,” the study team wrote.
The study was published online in JMIR Cancer.
Although evidence has consistently revealed similar findings, the risk of death among unvaccinated people with cancer and COVID-19 has not been nearly as high in previous studies, lead author Brian E. Dixon, PhD, MBA, with Indiana University Richard M. Fairbanks School of Public Health, Indianapolis, said in a statement. Previous studies from China, for instance, reported a two- to threefold greater risk of all-cause mortality among unvaccinated adults with cancer and COVID-19.
A potential reason for this discrepancy, Dr. Dixon noted, is that earlier studies were “generally smaller and made calculations based on data from a single cancer center or health system.”
Another reason is testing for COVID-19 early in the pandemic was limited to symptomatic individuals who may have had more severe infections, possibly leading to an overestimate of the association between SARS-CoV-2 infection, cancer, and all-cause mortality.
In the current analysis, researchers used electronic health records linked to Indiana’s statewide SARS-CoV-2 testing database and state vital records to evaluate the association between SARS-CoV-2 infection and all-cause mortality among 41,924 adults newly diagnosed with cancer between Jan. 1, 2019, and Dec. 31, 2020.
Most people with cancer were White (78.4%) and about half were male. At the time of diagnosis, 17% had one comorbid condition and about 10% had two or more. Most patients had breast cancer (14%), prostate cancer (13%), or melanoma (13%).
During the study period, 2,894 patients (7%) tested positive for SARS-CoV-2.
In multivariate adjusted analysis, the risk of death among those newly diagnosed with cancer increased by 91% (adjusted hazard ratio, 1.91) during the first year of the pandemic before vaccines were available, compared with the year before (January 2019 to Jan. 14, 2020).
During the pandemic period, the risk of death was roughly threefold higher among adults 65 years old and older, compared with adults 18-44 years old (aHR, 3.35).
When looking at the time from a cancer diagnosis to SARS-CoV-2 infection, infection was associated with an almost sevenfold increase in all-cause mortality (aHR, 6.91). Adults 65 years old and older had an almost threefold increased risk of dying, compared with their younger peers (aHR, 2.74).
Dr. Dixon and colleagues also observed an increased risk of death in men with cancer and COVID, compared with women (aHR, 1.23) and those with at least two comorbid conditions versus none (aHR, 2.12). In addition, the risk of dying was 9% higher among Indiana’s rural population than urban dwellers.
Compared with other cancer types, individuals with lung cancer and other digestive cancers had the highest risk of death after SARS-CoV-2 infection (aHR, 1.45 and 1.80, respectively).
“Our findings highlight the increased risk of death for adult cancer patients who test positive for COVID and underscore the importance to cancer patients – including those in remission – of vaccinations, boosters, and regular COVID testing,” Dr. Dixon commented.
“Our results should encourage individuals diagnosed with cancer not only to take preventive action, but also to expeditiously seek out treatments available in the marketplace should they test positive for COVID,” he added.
Support for the study was provided by Indiana University Simon Cancer Center and the Centers for Disease Control and Prevention. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study underscores the importance of COVID-19 and regular COVID-19 testing among adults with a recent cancer diagnosis.
The Indiana statewide study, conducted at the beginning of the pandemic, found that
“This analysis provides additional empirical evidence on the magnitude of risk to patients with cancer whose immune systems are often weakened either by the disease or treatment,” the study team wrote.
The study was published online in JMIR Cancer.
Although evidence has consistently revealed similar findings, the risk of death among unvaccinated people with cancer and COVID-19 has not been nearly as high in previous studies, lead author Brian E. Dixon, PhD, MBA, with Indiana University Richard M. Fairbanks School of Public Health, Indianapolis, said in a statement. Previous studies from China, for instance, reported a two- to threefold greater risk of all-cause mortality among unvaccinated adults with cancer and COVID-19.
A potential reason for this discrepancy, Dr. Dixon noted, is that earlier studies were “generally smaller and made calculations based on data from a single cancer center or health system.”
Another reason is testing for COVID-19 early in the pandemic was limited to symptomatic individuals who may have had more severe infections, possibly leading to an overestimate of the association between SARS-CoV-2 infection, cancer, and all-cause mortality.
In the current analysis, researchers used electronic health records linked to Indiana’s statewide SARS-CoV-2 testing database and state vital records to evaluate the association between SARS-CoV-2 infection and all-cause mortality among 41,924 adults newly diagnosed with cancer between Jan. 1, 2019, and Dec. 31, 2020.
Most people with cancer were White (78.4%) and about half were male. At the time of diagnosis, 17% had one comorbid condition and about 10% had two or more. Most patients had breast cancer (14%), prostate cancer (13%), or melanoma (13%).
During the study period, 2,894 patients (7%) tested positive for SARS-CoV-2.
In multivariate adjusted analysis, the risk of death among those newly diagnosed with cancer increased by 91% (adjusted hazard ratio, 1.91) during the first year of the pandemic before vaccines were available, compared with the year before (January 2019 to Jan. 14, 2020).
During the pandemic period, the risk of death was roughly threefold higher among adults 65 years old and older, compared with adults 18-44 years old (aHR, 3.35).
When looking at the time from a cancer diagnosis to SARS-CoV-2 infection, infection was associated with an almost sevenfold increase in all-cause mortality (aHR, 6.91). Adults 65 years old and older had an almost threefold increased risk of dying, compared with their younger peers (aHR, 2.74).
Dr. Dixon and colleagues also observed an increased risk of death in men with cancer and COVID, compared with women (aHR, 1.23) and those with at least two comorbid conditions versus none (aHR, 2.12). In addition, the risk of dying was 9% higher among Indiana’s rural population than urban dwellers.
Compared with other cancer types, individuals with lung cancer and other digestive cancers had the highest risk of death after SARS-CoV-2 infection (aHR, 1.45 and 1.80, respectively).
“Our findings highlight the increased risk of death for adult cancer patients who test positive for COVID and underscore the importance to cancer patients – including those in remission – of vaccinations, boosters, and regular COVID testing,” Dr. Dixon commented.
“Our results should encourage individuals diagnosed with cancer not only to take preventive action, but also to expeditiously seek out treatments available in the marketplace should they test positive for COVID,” he added.
Support for the study was provided by Indiana University Simon Cancer Center and the Centers for Disease Control and Prevention. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JMIR CANCER
COVID tied to spike in deaths in chronic liver disease with diabetes
The COVID-19 pandemic fueled a sharp uptick in deaths related to chronic liver disease and cirrhosis among people with diabetes, largely owing to nonalcoholic fatty liver disease (NAFLD) and alcohol-related liver disease (ALD), new data show.
“Our observations confirm that COVID-19 had a higher likelihood of impacting vulnerable populations with pre-existing chronic liver diseases and diabetes, with a death rate as high as 10% in individuals with co-existing chronic liver disease and diabetes,” write the authors.
“The inability to attend regular outpatient clinics for close monitoring and treatment accompanied by diversion of health care resources to COVID-19 care may have resulted in the suboptimal or delayed clinical care of individuals with diabetes and chronic liver disease during the COVID-19 pandemic,” they add.
Donghee Kim, MD, PhD, with the Division of Gastroenterology and Hepatology, Stanford (Calif.) University School of Medicine, and colleagues report their findings in the journal Digestive and Liver Disease.
Vulnerable group
The researchers used U.S. national mortality data (2017-2020) to estimate chronic liver disease–related mortality trends among individuals with diabetes before and during the COVID-19 pandemic.
Before the pandemic, the quarterly mortality for chronic liver disease remained stable (quarterly percentage change, 0.6%) but then sharply increased during the pandemic (QPC, 8.6%).
A similar trend was seen with cirrhosis-related mortality (QPC, 0.3% before the pandemic vs. 8.4% during the pandemic).
NAFLD and ALD mortality among individuals with diabetes was steadily increasing before the pandemic (QPC, 4.2% and 3.5%, respectively) but showed a more rapid increase during the pandemic (QPC, 9.6% and 7.7%, respectively).
ALD-related mortality in men was more than threefold higher than in women, while NAFLD-related mortality in women was more than twofold higher than in men.
Mortality for hepatitis C virus infection declined before the pandemic (QPC, −3.3%) and remained stable during the pandemic.
COVID-19–related mortality among adults with chronic liver disease and diabetes also rose sharply during the pandemic – from 0.4% in the first quarter of 2020 to 12.9% in the last quarter of 2020 – with no considerable difference between men and women.
Blame it on lockdowns?
Dr. Kim and colleagues say research is needed to better understand the direct and indirect influence of COVID-19 on coexisting chronic liver disease and diabetes.
“It is plausible that psychosocial stress and a higher predisposition to psychiatric disorders during the COVID-19 pandemic can increase the risk of alcohol use disorder and ALD,” they write.
“Furthermore, it is prudent to suspect that COVID-19–related lockdowns may increase the risk of obesity, leading to a higher risk of insulin resistance and metabolic complications, including diabetes and NAFLD. Future studies are needed to improve our understanding of these possible pathogenetic links. More importantly, emergency preparedness or contingency plans must be in place to continue and provide uninterrupted care for chronic ailments during times of disaster,” they add.
The study had no specific funding. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The COVID-19 pandemic fueled a sharp uptick in deaths related to chronic liver disease and cirrhosis among people with diabetes, largely owing to nonalcoholic fatty liver disease (NAFLD) and alcohol-related liver disease (ALD), new data show.
“Our observations confirm that COVID-19 had a higher likelihood of impacting vulnerable populations with pre-existing chronic liver diseases and diabetes, with a death rate as high as 10% in individuals with co-existing chronic liver disease and diabetes,” write the authors.
“The inability to attend regular outpatient clinics for close monitoring and treatment accompanied by diversion of health care resources to COVID-19 care may have resulted in the suboptimal or delayed clinical care of individuals with diabetes and chronic liver disease during the COVID-19 pandemic,” they add.
Donghee Kim, MD, PhD, with the Division of Gastroenterology and Hepatology, Stanford (Calif.) University School of Medicine, and colleagues report their findings in the journal Digestive and Liver Disease.
Vulnerable group
The researchers used U.S. national mortality data (2017-2020) to estimate chronic liver disease–related mortality trends among individuals with diabetes before and during the COVID-19 pandemic.
Before the pandemic, the quarterly mortality for chronic liver disease remained stable (quarterly percentage change, 0.6%) but then sharply increased during the pandemic (QPC, 8.6%).
A similar trend was seen with cirrhosis-related mortality (QPC, 0.3% before the pandemic vs. 8.4% during the pandemic).
NAFLD and ALD mortality among individuals with diabetes was steadily increasing before the pandemic (QPC, 4.2% and 3.5%, respectively) but showed a more rapid increase during the pandemic (QPC, 9.6% and 7.7%, respectively).
ALD-related mortality in men was more than threefold higher than in women, while NAFLD-related mortality in women was more than twofold higher than in men.
Mortality for hepatitis C virus infection declined before the pandemic (QPC, −3.3%) and remained stable during the pandemic.
COVID-19–related mortality among adults with chronic liver disease and diabetes also rose sharply during the pandemic – from 0.4% in the first quarter of 2020 to 12.9% in the last quarter of 2020 – with no considerable difference between men and women.
Blame it on lockdowns?
Dr. Kim and colleagues say research is needed to better understand the direct and indirect influence of COVID-19 on coexisting chronic liver disease and diabetes.
“It is plausible that psychosocial stress and a higher predisposition to psychiatric disorders during the COVID-19 pandemic can increase the risk of alcohol use disorder and ALD,” they write.
“Furthermore, it is prudent to suspect that COVID-19–related lockdowns may increase the risk of obesity, leading to a higher risk of insulin resistance and metabolic complications, including diabetes and NAFLD. Future studies are needed to improve our understanding of these possible pathogenetic links. More importantly, emergency preparedness or contingency plans must be in place to continue and provide uninterrupted care for chronic ailments during times of disaster,” they add.
The study had no specific funding. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The COVID-19 pandemic fueled a sharp uptick in deaths related to chronic liver disease and cirrhosis among people with diabetes, largely owing to nonalcoholic fatty liver disease (NAFLD) and alcohol-related liver disease (ALD), new data show.
“Our observations confirm that COVID-19 had a higher likelihood of impacting vulnerable populations with pre-existing chronic liver diseases and diabetes, with a death rate as high as 10% in individuals with co-existing chronic liver disease and diabetes,” write the authors.
“The inability to attend regular outpatient clinics for close monitoring and treatment accompanied by diversion of health care resources to COVID-19 care may have resulted in the suboptimal or delayed clinical care of individuals with diabetes and chronic liver disease during the COVID-19 pandemic,” they add.
Donghee Kim, MD, PhD, with the Division of Gastroenterology and Hepatology, Stanford (Calif.) University School of Medicine, and colleagues report their findings in the journal Digestive and Liver Disease.
Vulnerable group
The researchers used U.S. national mortality data (2017-2020) to estimate chronic liver disease–related mortality trends among individuals with diabetes before and during the COVID-19 pandemic.
Before the pandemic, the quarterly mortality for chronic liver disease remained stable (quarterly percentage change, 0.6%) but then sharply increased during the pandemic (QPC, 8.6%).
A similar trend was seen with cirrhosis-related mortality (QPC, 0.3% before the pandemic vs. 8.4% during the pandemic).
NAFLD and ALD mortality among individuals with diabetes was steadily increasing before the pandemic (QPC, 4.2% and 3.5%, respectively) but showed a more rapid increase during the pandemic (QPC, 9.6% and 7.7%, respectively).
ALD-related mortality in men was more than threefold higher than in women, while NAFLD-related mortality in women was more than twofold higher than in men.
Mortality for hepatitis C virus infection declined before the pandemic (QPC, −3.3%) and remained stable during the pandemic.
COVID-19–related mortality among adults with chronic liver disease and diabetes also rose sharply during the pandemic – from 0.4% in the first quarter of 2020 to 12.9% in the last quarter of 2020 – with no considerable difference between men and women.
Blame it on lockdowns?
Dr. Kim and colleagues say research is needed to better understand the direct and indirect influence of COVID-19 on coexisting chronic liver disease and diabetes.
“It is plausible that psychosocial stress and a higher predisposition to psychiatric disorders during the COVID-19 pandemic can increase the risk of alcohol use disorder and ALD,” they write.
“Furthermore, it is prudent to suspect that COVID-19–related lockdowns may increase the risk of obesity, leading to a higher risk of insulin resistance and metabolic complications, including diabetes and NAFLD. Future studies are needed to improve our understanding of these possible pathogenetic links. More importantly, emergency preparedness or contingency plans must be in place to continue and provide uninterrupted care for chronic ailments during times of disaster,” they add.
The study had no specific funding. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Preexisting mental illness symptoms spiked during pandemic
“Those with preexisting mental health conditions may be particularly vulnerable to these effects because they are more susceptible to experiencing high levels of stress during a crisis and are more likely to experience isolation/despair during confinement compared to the general population,” wrote Danna Ramirez of The Menninger Clinic, Houston, and colleagues.
In a study published in Psychiatry Research , the investigators compared data from 142 adolescents aged 12-17 years and 470 adults aged 18-79 years who were admitted to an inpatient psychiatric hospital in Houston. Of these, 65 adolescents and 235 adults were admitted before the pandemic, and 77 adolescents and 235 adults were admitted during the pandemic.
Clinical outcomes were scores on the Generalized Anxiety Disorder Scale (GAD-7), the Patient Health Questionnaire (PHQ-9), the Patient Health Questionnaire for Adolescents (PHQ-A), the Difficulties in Emotion Regulation Scale–Short Form (DERS-SF), the World Health Organization Disability Assessment Scale (WHODAS), the World Health Organization Alcohol, Smoking, and Substance Involvement Screening Test (WHOASSIST), the Pittsburgh Sleep Quality Index (PSQI), the Disturbing Dream and Nightmare Severity Index (DDNSI), and the Suicide Behaviors Questionnaire–Revised (SBQ-R).
Overall, adults admitted during the pandemic had significantly higher levels of anxiety, depression, emotional dysregulation, and disability (P < .001 for all) as well as nightmares (P = .013) compared to those admitted prior to the pandemic.
Among adolescents, measures of anxiety, depression, and sleep quality were significantly higher at admission during the pandemic compared to prior to the pandemic (P = .005, P = .005, and P = .011, respectively)
Reasons for the increase in symptom severity remain unclear, but include the possibility that individuals with preexisting mental illness simply became more ill; or that individuals with symptoms delayed hospital admission out of fear of exposure to COVID-19, which resulted in more severe symptoms at admission, the researchers wrote in their discussion.
The findings were limited by several factors, including the primarily White population and the reliance on self-reports, the researchers noted. Another limitation was the lack of differentiation between patients who may have had COVID-19 before hospitalization and those who did not, so the researchers could not determine whether the virus itself played a biological role in symptom severity.
However, the results support data from previous studies and identify increased psychiatry symptom severity for patients admitted for inpatient psychiatry care during the pandemic, they said. Although resources are scarce, the findings emphasize that mental health needs, especially for those with preexisting conditions, should not be overlooked, and continuity and expansion of access to mental health care for all should be prioritized, they concluded.
The study was supported by The Menninger Clinic and The Menninger Clinic Foundation. The researchers had no financial conflicts to disclose.
“Those with preexisting mental health conditions may be particularly vulnerable to these effects because they are more susceptible to experiencing high levels of stress during a crisis and are more likely to experience isolation/despair during confinement compared to the general population,” wrote Danna Ramirez of The Menninger Clinic, Houston, and colleagues.
In a study published in Psychiatry Research , the investigators compared data from 142 adolescents aged 12-17 years and 470 adults aged 18-79 years who were admitted to an inpatient psychiatric hospital in Houston. Of these, 65 adolescents and 235 adults were admitted before the pandemic, and 77 adolescents and 235 adults were admitted during the pandemic.
Clinical outcomes were scores on the Generalized Anxiety Disorder Scale (GAD-7), the Patient Health Questionnaire (PHQ-9), the Patient Health Questionnaire for Adolescents (PHQ-A), the Difficulties in Emotion Regulation Scale–Short Form (DERS-SF), the World Health Organization Disability Assessment Scale (WHODAS), the World Health Organization Alcohol, Smoking, and Substance Involvement Screening Test (WHOASSIST), the Pittsburgh Sleep Quality Index (PSQI), the Disturbing Dream and Nightmare Severity Index (DDNSI), and the Suicide Behaviors Questionnaire–Revised (SBQ-R).
Overall, adults admitted during the pandemic had significantly higher levels of anxiety, depression, emotional dysregulation, and disability (P < .001 for all) as well as nightmares (P = .013) compared to those admitted prior to the pandemic.
Among adolescents, measures of anxiety, depression, and sleep quality were significantly higher at admission during the pandemic compared to prior to the pandemic (P = .005, P = .005, and P = .011, respectively)
Reasons for the increase in symptom severity remain unclear, but include the possibility that individuals with preexisting mental illness simply became more ill; or that individuals with symptoms delayed hospital admission out of fear of exposure to COVID-19, which resulted in more severe symptoms at admission, the researchers wrote in their discussion.
The findings were limited by several factors, including the primarily White population and the reliance on self-reports, the researchers noted. Another limitation was the lack of differentiation between patients who may have had COVID-19 before hospitalization and those who did not, so the researchers could not determine whether the virus itself played a biological role in symptom severity.
However, the results support data from previous studies and identify increased psychiatry symptom severity for patients admitted for inpatient psychiatry care during the pandemic, they said. Although resources are scarce, the findings emphasize that mental health needs, especially for those with preexisting conditions, should not be overlooked, and continuity and expansion of access to mental health care for all should be prioritized, they concluded.
The study was supported by The Menninger Clinic and The Menninger Clinic Foundation. The researchers had no financial conflicts to disclose.
“Those with preexisting mental health conditions may be particularly vulnerable to these effects because they are more susceptible to experiencing high levels of stress during a crisis and are more likely to experience isolation/despair during confinement compared to the general population,” wrote Danna Ramirez of The Menninger Clinic, Houston, and colleagues.
In a study published in Psychiatry Research , the investigators compared data from 142 adolescents aged 12-17 years and 470 adults aged 18-79 years who were admitted to an inpatient psychiatric hospital in Houston. Of these, 65 adolescents and 235 adults were admitted before the pandemic, and 77 adolescents and 235 adults were admitted during the pandemic.
Clinical outcomes were scores on the Generalized Anxiety Disorder Scale (GAD-7), the Patient Health Questionnaire (PHQ-9), the Patient Health Questionnaire for Adolescents (PHQ-A), the Difficulties in Emotion Regulation Scale–Short Form (DERS-SF), the World Health Organization Disability Assessment Scale (WHODAS), the World Health Organization Alcohol, Smoking, and Substance Involvement Screening Test (WHOASSIST), the Pittsburgh Sleep Quality Index (PSQI), the Disturbing Dream and Nightmare Severity Index (DDNSI), and the Suicide Behaviors Questionnaire–Revised (SBQ-R).
Overall, adults admitted during the pandemic had significantly higher levels of anxiety, depression, emotional dysregulation, and disability (P < .001 for all) as well as nightmares (P = .013) compared to those admitted prior to the pandemic.
Among adolescents, measures of anxiety, depression, and sleep quality were significantly higher at admission during the pandemic compared to prior to the pandemic (P = .005, P = .005, and P = .011, respectively)
Reasons for the increase in symptom severity remain unclear, but include the possibility that individuals with preexisting mental illness simply became more ill; or that individuals with symptoms delayed hospital admission out of fear of exposure to COVID-19, which resulted in more severe symptoms at admission, the researchers wrote in their discussion.
The findings were limited by several factors, including the primarily White population and the reliance on self-reports, the researchers noted. Another limitation was the lack of differentiation between patients who may have had COVID-19 before hospitalization and those who did not, so the researchers could not determine whether the virus itself played a biological role in symptom severity.
However, the results support data from previous studies and identify increased psychiatry symptom severity for patients admitted for inpatient psychiatry care during the pandemic, they said. Although resources are scarce, the findings emphasize that mental health needs, especially for those with preexisting conditions, should not be overlooked, and continuity and expansion of access to mental health care for all should be prioritized, they concluded.
The study was supported by The Menninger Clinic and The Menninger Clinic Foundation. The researchers had no financial conflicts to disclose.
FROM PSYCHIATRY RESEARCH