User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
A hot dog a day takes 36 minutes away
The death ‘dog’
Imagine you’re out in your backyard managing the grill for a big family barbecue. You’ve got a dazzling assortment of meat assorted on your fancy new propane grill, all charring nicely. Naturally, the hot dogs finish first, and as you pull them off, you figure you’ll help yourself to one now. After all, you are the chef, you deserve a reward. But, as you bite into your smoking hot sandwich, a cold, bony finger taps you on the shoulder. You turn and come face to face with the Grim Reaper. “YOU JUST LOST 36 MINUTES,” Death says. “ALSO, MAY I HAVE ONE OF THOSE? THEY LOOK DELICIOUS.”
Nonplussed and moving automatically, you scoop up another hot dog and place it in a bun. “WITH KETCHUP PLEASE,” Death says. “I NEVER CARED FOR MUSTARD.”
“I don’t understand,” you say. “Surely I won’t die at a family barbecue.”
“DO NOT CALL ME SHIRLEY,” Death says. “AND YOU WILL NOT. IT’S PART OF MY NEW CONTRACT.”
A new study, published in Nature Food, found that a person may lose up to 36 minutes for every hot dog consumed. Researchers from the University of Michigan analyzed nearly 6,000 different foods using a new nutritional index to quantify their health effects in minutes of healthy life lost or gained. Eating a serving of nuts adds an extra 26 minutes of life. The researchers determined that replacing just 10% of daily caloric intake from beef and processed foods with fruits, vegetables, and nuts can add 48 minutes per day. It would also reduce the daily carbon footprint by 33%.
“So you go around to everyone eating bad food and tell them how much life they’ve lost?” you ask when the Grim Reaper finishes his story. “Sounds like a drag.”
“IT IS. WE’VE HAD TO HIRE NEW BLOOD.” Death chuckles at its own bad pun. “NOW IF YOU’LL EXCUSE ME, I MUST CHASTISE A MAN IN FLORIDA FOR EATING A WELL-DONE STEAK.”
More stress, less sex
As the world becomes a more stressful place, the human population could face a 50% drop by the end of the century.
Think of stress as a one-two punch to the libido and human fertility. The more people are stressed out, the less likely they are to have quality interactions with others. Many of us would rather be alone with our wine and cheese to watch our favorite show.
Researchers have found that high stress levels have been known to drop sperm count, ovulation, and sexual activity. Guess what? There has been a 50% decrease in sperm counts over the last 50 years. That’s the second punch. But let’s not forget, the times are changing.
“Changes in reproductive behavior that contribute to the population drop include more young couples choosing to be ‘child-free,’ people having fewer children, and couples waiting longer to start families,” said Alexander Suvorov, PhD, of the University of Massachusetts, the paper’s author.
Let’s summarize: The more stress we’re dealing with, the less people want to deal with each other.
Who would have thought the future would be less fun?
‘You are not a horse. You are not a cow. Seriously, y’all. Stop it.’
WARNING: The following descriptions of COVID-19–related insanity may be offensive to some readers.
Greetings, ladies and gentlemen! Welcome to the first round of Pandemic Pandemonium. Let’s get right to the action.
This week’s preshow match-off involves face mask woes. The first comes to us from Alabama, where a woman wore a space helmet to a school board meeting to protest mask mandates. The second comes from Australia, in the form of mischievous magpies. We will explain.
It is not uncommon for magpies to attack those who come too close to their nests in the spring, or “swooping season,” as it’s affectionately called. The magpies are smart enough to recognize the faces of people they see regularly and not attack; however, it’s feared that mask wearing will change this.
While you’re chewing on that exciting appetizer, let’s take a look at our main course, which has a distinct governmental flavor. Jeff Landry is the attorney general of Louisiana, and, like our space-helmet wearer, he’s not a fan of mask mandates. According to Business Insider, Mr. Landry “drafted and distributed sample letters intended to help parents evade mask-wearing ordinances and COVID-19 vaccination requirements for their children in schools.”
Up against him is the Food and Drug Administration’s Twitter account. In an unrelated matter, the agency tweeted, “You are not a horse. You are not a cow. Seriously, y’all. Stop it.” This was in response to people using the nonhuman forms of ivermectin to treat very human COVID-19.
Well, there you have it. Who will win tonight’s exciting edition of Pandemic Pandemonium? The first reader to contact us gets to decide the fate of these worthy contestants.
From venomous poison to heart drug
It’s not likely that anyone who sees a giant, venomous spider is thinking, “Hey! That thing could save my life!” It’s usually quite the opposite. Honestly, we would run away from just about any spider. But what if one of the deadliest spiders in the world could also save you from dying of a heart attack?
You probably don’t believe us, right? That’s fair, but the deadly Fraser Island (K’gari) funnel web spider, might also be the most helpful. Investigators from the University of Queensland in Australia have found a way to extract a molecule from the spider’s venom that might help stop damage from heart attacks and may even preserve hearts being used for transplants. “The Hi1a protein from spider venom blocks acid-sensing ion channels in the heart, so the death message is blocked, cell death is reduced, and we see improved heart cell survival,” Nathan Palpant, PhD, of the university, noted in a written statement.
No one has ever developed a drug to stop the “death signal,” so maybe it’s time to befriend spiders instead of running away from them in horror. Just leave the venom extraction to the professionals.
The death ‘dog’
Imagine you’re out in your backyard managing the grill for a big family barbecue. You’ve got a dazzling assortment of meat assorted on your fancy new propane grill, all charring nicely. Naturally, the hot dogs finish first, and as you pull them off, you figure you’ll help yourself to one now. After all, you are the chef, you deserve a reward. But, as you bite into your smoking hot sandwich, a cold, bony finger taps you on the shoulder. You turn and come face to face with the Grim Reaper. “YOU JUST LOST 36 MINUTES,” Death says. “ALSO, MAY I HAVE ONE OF THOSE? THEY LOOK DELICIOUS.”
Nonplussed and moving automatically, you scoop up another hot dog and place it in a bun. “WITH KETCHUP PLEASE,” Death says. “I NEVER CARED FOR MUSTARD.”
“I don’t understand,” you say. “Surely I won’t die at a family barbecue.”
“DO NOT CALL ME SHIRLEY,” Death says. “AND YOU WILL NOT. IT’S PART OF MY NEW CONTRACT.”
A new study, published in Nature Food, found that a person may lose up to 36 minutes for every hot dog consumed. Researchers from the University of Michigan analyzed nearly 6,000 different foods using a new nutritional index to quantify their health effects in minutes of healthy life lost or gained. Eating a serving of nuts adds an extra 26 minutes of life. The researchers determined that replacing just 10% of daily caloric intake from beef and processed foods with fruits, vegetables, and nuts can add 48 minutes per day. It would also reduce the daily carbon footprint by 33%.
“So you go around to everyone eating bad food and tell them how much life they’ve lost?” you ask when the Grim Reaper finishes his story. “Sounds like a drag.”
“IT IS. WE’VE HAD TO HIRE NEW BLOOD.” Death chuckles at its own bad pun. “NOW IF YOU’LL EXCUSE ME, I MUST CHASTISE A MAN IN FLORIDA FOR EATING A WELL-DONE STEAK.”
More stress, less sex
As the world becomes a more stressful place, the human population could face a 50% drop by the end of the century.
Think of stress as a one-two punch to the libido and human fertility. The more people are stressed out, the less likely they are to have quality interactions with others. Many of us would rather be alone with our wine and cheese to watch our favorite show.
Researchers have found that high stress levels have been known to drop sperm count, ovulation, and sexual activity. Guess what? There has been a 50% decrease in sperm counts over the last 50 years. That’s the second punch. But let’s not forget, the times are changing.
“Changes in reproductive behavior that contribute to the population drop include more young couples choosing to be ‘child-free,’ people having fewer children, and couples waiting longer to start families,” said Alexander Suvorov, PhD, of the University of Massachusetts, the paper’s author.
Let’s summarize: The more stress we’re dealing with, the less people want to deal with each other.
Who would have thought the future would be less fun?
‘You are not a horse. You are not a cow. Seriously, y’all. Stop it.’
WARNING: The following descriptions of COVID-19–related insanity may be offensive to some readers.
Greetings, ladies and gentlemen! Welcome to the first round of Pandemic Pandemonium. Let’s get right to the action.
This week’s preshow match-off involves face mask woes. The first comes to us from Alabama, where a woman wore a space helmet to a school board meeting to protest mask mandates. The second comes from Australia, in the form of mischievous magpies. We will explain.
It is not uncommon for magpies to attack those who come too close to their nests in the spring, or “swooping season,” as it’s affectionately called. The magpies are smart enough to recognize the faces of people they see regularly and not attack; however, it’s feared that mask wearing will change this.
While you’re chewing on that exciting appetizer, let’s take a look at our main course, which has a distinct governmental flavor. Jeff Landry is the attorney general of Louisiana, and, like our space-helmet wearer, he’s not a fan of mask mandates. According to Business Insider, Mr. Landry “drafted and distributed sample letters intended to help parents evade mask-wearing ordinances and COVID-19 vaccination requirements for their children in schools.”
Up against him is the Food and Drug Administration’s Twitter account. In an unrelated matter, the agency tweeted, “You are not a horse. You are not a cow. Seriously, y’all. Stop it.” This was in response to people using the nonhuman forms of ivermectin to treat very human COVID-19.
Well, there you have it. Who will win tonight’s exciting edition of Pandemic Pandemonium? The first reader to contact us gets to decide the fate of these worthy contestants.
From venomous poison to heart drug
It’s not likely that anyone who sees a giant, venomous spider is thinking, “Hey! That thing could save my life!” It’s usually quite the opposite. Honestly, we would run away from just about any spider. But what if one of the deadliest spiders in the world could also save you from dying of a heart attack?
You probably don’t believe us, right? That’s fair, but the deadly Fraser Island (K’gari) funnel web spider, might also be the most helpful. Investigators from the University of Queensland in Australia have found a way to extract a molecule from the spider’s venom that might help stop damage from heart attacks and may even preserve hearts being used for transplants. “The Hi1a protein from spider venom blocks acid-sensing ion channels in the heart, so the death message is blocked, cell death is reduced, and we see improved heart cell survival,” Nathan Palpant, PhD, of the university, noted in a written statement.
No one has ever developed a drug to stop the “death signal,” so maybe it’s time to befriend spiders instead of running away from them in horror. Just leave the venom extraction to the professionals.
The death ‘dog’
Imagine you’re out in your backyard managing the grill for a big family barbecue. You’ve got a dazzling assortment of meat assorted on your fancy new propane grill, all charring nicely. Naturally, the hot dogs finish first, and as you pull them off, you figure you’ll help yourself to one now. After all, you are the chef, you deserve a reward. But, as you bite into your smoking hot sandwich, a cold, bony finger taps you on the shoulder. You turn and come face to face with the Grim Reaper. “YOU JUST LOST 36 MINUTES,” Death says. “ALSO, MAY I HAVE ONE OF THOSE? THEY LOOK DELICIOUS.”
Nonplussed and moving automatically, you scoop up another hot dog and place it in a bun. “WITH KETCHUP PLEASE,” Death says. “I NEVER CARED FOR MUSTARD.”
“I don’t understand,” you say. “Surely I won’t die at a family barbecue.”
“DO NOT CALL ME SHIRLEY,” Death says. “AND YOU WILL NOT. IT’S PART OF MY NEW CONTRACT.”
A new study, published in Nature Food, found that a person may lose up to 36 minutes for every hot dog consumed. Researchers from the University of Michigan analyzed nearly 6,000 different foods using a new nutritional index to quantify their health effects in minutes of healthy life lost or gained. Eating a serving of nuts adds an extra 26 minutes of life. The researchers determined that replacing just 10% of daily caloric intake from beef and processed foods with fruits, vegetables, and nuts can add 48 minutes per day. It would also reduce the daily carbon footprint by 33%.
“So you go around to everyone eating bad food and tell them how much life they’ve lost?” you ask when the Grim Reaper finishes his story. “Sounds like a drag.”
“IT IS. WE’VE HAD TO HIRE NEW BLOOD.” Death chuckles at its own bad pun. “NOW IF YOU’LL EXCUSE ME, I MUST CHASTISE A MAN IN FLORIDA FOR EATING A WELL-DONE STEAK.”
More stress, less sex
As the world becomes a more stressful place, the human population could face a 50% drop by the end of the century.
Think of stress as a one-two punch to the libido and human fertility. The more people are stressed out, the less likely they are to have quality interactions with others. Many of us would rather be alone with our wine and cheese to watch our favorite show.
Researchers have found that high stress levels have been known to drop sperm count, ovulation, and sexual activity. Guess what? There has been a 50% decrease in sperm counts over the last 50 years. That’s the second punch. But let’s not forget, the times are changing.
“Changes in reproductive behavior that contribute to the population drop include more young couples choosing to be ‘child-free,’ people having fewer children, and couples waiting longer to start families,” said Alexander Suvorov, PhD, of the University of Massachusetts, the paper’s author.
Let’s summarize: The more stress we’re dealing with, the less people want to deal with each other.
Who would have thought the future would be less fun?
‘You are not a horse. You are not a cow. Seriously, y’all. Stop it.’
WARNING: The following descriptions of COVID-19–related insanity may be offensive to some readers.
Greetings, ladies and gentlemen! Welcome to the first round of Pandemic Pandemonium. Let’s get right to the action.
This week’s preshow match-off involves face mask woes. The first comes to us from Alabama, where a woman wore a space helmet to a school board meeting to protest mask mandates. The second comes from Australia, in the form of mischievous magpies. We will explain.
It is not uncommon for magpies to attack those who come too close to their nests in the spring, or “swooping season,” as it’s affectionately called. The magpies are smart enough to recognize the faces of people they see regularly and not attack; however, it’s feared that mask wearing will change this.
While you’re chewing on that exciting appetizer, let’s take a look at our main course, which has a distinct governmental flavor. Jeff Landry is the attorney general of Louisiana, and, like our space-helmet wearer, he’s not a fan of mask mandates. According to Business Insider, Mr. Landry “drafted and distributed sample letters intended to help parents evade mask-wearing ordinances and COVID-19 vaccination requirements for their children in schools.”
Up against him is the Food and Drug Administration’s Twitter account. In an unrelated matter, the agency tweeted, “You are not a horse. You are not a cow. Seriously, y’all. Stop it.” This was in response to people using the nonhuman forms of ivermectin to treat very human COVID-19.
Well, there you have it. Who will win tonight’s exciting edition of Pandemic Pandemonium? The first reader to contact us gets to decide the fate of these worthy contestants.
From venomous poison to heart drug
It’s not likely that anyone who sees a giant, venomous spider is thinking, “Hey! That thing could save my life!” It’s usually quite the opposite. Honestly, we would run away from just about any spider. But what if one of the deadliest spiders in the world could also save you from dying of a heart attack?
You probably don’t believe us, right? That’s fair, but the deadly Fraser Island (K’gari) funnel web spider, might also be the most helpful. Investigators from the University of Queensland in Australia have found a way to extract a molecule from the spider’s venom that might help stop damage from heart attacks and may even preserve hearts being used for transplants. “The Hi1a protein from spider venom blocks acid-sensing ion channels in the heart, so the death message is blocked, cell death is reduced, and we see improved heart cell survival,” Nathan Palpant, PhD, of the university, noted in a written statement.
No one has ever developed a drug to stop the “death signal,” so maybe it’s time to befriend spiders instead of running away from them in horror. Just leave the venom extraction to the professionals.
Better to binge drink than regularly tipple, suggests GI cancer study
When weekly levels are similar
Alcohol use is a known risk factor for gastrointestinal (GI) cancers. Now, new research indicates that this risk is more associated with frequent drinking – even in smaller amounts – compared with higher amounts or binge drinking, given similar weekly levels.
“The novel finding of the current study is that frequent drinking may be more dangerous than binge drinking with regard to GI cancers. Alcohol use is a known risk factor for gastrointestinal (GI) cancers. Now, new research indicates that this risk is more associated with frequent drinking – even in smaller amounts -- compared with higher amounts or binge drinking, given similar weekly levels.” first author Jung Eun Yook, MD, of Seoul (South Korea) National University Hospital, and colleagues reported in an article published Aug. 18, 2021, in JAMA Network Open (doi: 10.1001/jamanetworkopen.2021.20382).
“This finding suggests that repeated alcohol consumption events even at lower amounts of alcohol may have a greater carcinogenic effect on GI organs than the consumption of larger amounts of alcohol at a lower frequency,” the investigators wrote.
A possible reason behind the difference in risk may be that the chronic “carcinogenic insult” from regular alcohol use may promote cancer development, whereas less frequent, episodic alcohol exposures may allow physiologic recovery, said the authors.
The results are from a population-based study that involved 11,737,467 participants in the Korean National Health System database who did not have cancer and who took part in a national screening program between January 2009 and December 2010.
They were followed from the year after their screening until either they had received a diagnosis of a GI cancer, death occurred, or the end of December 2017.
During a median follow-up of 6.4 years, 319,202 (2.7%) of those in the study developed a GI cancer.
The increase in the risk associated with alcohol consumption was dose dependent.
Compared with those who did not consume alcohol, the risk of developing GI cancer was higher for mild drinkers (adjusted hazard ratio, 1.04; 95% confidence interval, 1.03-1.05), moderate drinkers (aHR, 1.14; 95% CI, 1.12-1.15), and heavy drinkers (aHR, 1.28; 95% CI, 1.26-1.29), after adjusting for age, sex, income, smoking status with intensity, regular exercise, body mass index, diabetes, hypertension, and dyslipidemia.
There was a linear association between the frequency of drinking and GI cancer risk, with an aHR of 1.39 for individuals who reported drinking every day (95% CI, 1.36-1.41). The risk for GI cancer increased with consumption of five to seven units per occasion (aHR, 1.15). Notably, there were no similar increases with higher intake, including intake of 8-14 units per occasion (aHR, 1.11; 95% CI, 1.09-1.12), and even up to more than 14 units per occasion (aHR, 1.11; 95%CI, 1.08-1.14), in comparison with an intake of 5-7 units per occasion.
“Given similar weekly alcohol consumption levels, the risk of GI cancer increased with a higher frequency of drinking and decreased with a higher amount per occasion,” the authors write.
“Most previous studies just assess alcohol consumption as a total amount, [such as] drinks per occasion times occasion per week equals drinks per week [and] grams per week,” coauthor Dong Wook Shin, MD, DrPH, Samsung Medical Center, Sungkyunkwan University, Seoul, South Korea, said in an interview.
“But it was not known whether frequent drinking with small amount is more harmful than binge drinking, given a similar level of total drinking,” Dr. Shin said.
The increased risk associated with frequent drinking was generally similar with respect to esophageal, gastric, colorectal, biliary, and pancreatic cancer.
An exception was liver cancer, which showed a slightly decreased risk among mild drinkers (aHR, 0.91; 95% CI, 0.89-0.93).
Of note, the association between alcohol intake and the incidence of GI cancer was lower among women than men in terms of weekly consumption, frequency, and amount of alcohol consumed per occasion.
The associations between drinking and cancer type in terms of esophageal and liver cancers were similar between men and women. However, the alcohol-related risk for colorectal, biliary, and pancreatic cancers was less prominent for women.
Possible mechanisms related to regular drinking
A factor that might account for the increase in GI cancer risk with frequent drinking is that regular alcohol consumption “promotes the accumulation of cell divisions in the stem cells that maintain tissues in homeostasis,” the authors explained.
Another possible explanation is that long-term alcohol exposure may promote carcinogenesis, whereas less frequent exposure might allow “physiological homeostasis,” the authors wrote, adding that in vivo experiments have shown that duration and dose of alcohol exposure have been linked to cancer development.
Importantly, the findings support the importance of reducing the frequency of alcohol use to prevent cancer, the authors noted.
“Alcohol users who have a glass of wine or beer during dinner every day may develop more cancer than people who occasionally consume several drinks,” they cautioned.
Genetics, self-reporting considerations
In a related commentary, John D. Potter, MBBS, PhD, of the Research Center for Hauora and Health, Massey University, Wellington, New Zealand, noted that, in addition to supporting the known link between alcohol and cancers of the esophagus, colorectum, and liver, the study “strengthens evidence for a role of alcohol in stomach, biliary tract, and pancreas cancers.”
In comparison with nondrinkers, those who reported heavy drinking were much more likely to be smokers (51.6% vs. 9.0%); however, the study adjusted for smoking.
“Because the researchers were able to control for tobacco, this last finding [regarding the association with cancers of the stomach, biliary tract, and pancreas] is particularly informative,” Dr. Potter noted.
An important caveat is that more than a quarter of the Korean population is known to have an inactive form of the aldehyde dehydrogenase gene (ALDH2), which could have effects on alcohol metabolism as well as the risk for cancer, Dr. Potter wrote.
“This common polymorphism in ALDH2 (ALDH2 rs671 [c.1510G>A (Glu504Lys)]) has paradoxical effects,” he wrote.
“It increases the level of acetaldehyde in the blood of drinkers, which in turn increases the risk of cancer because acetaldehyde is a key player in the carcinogenicity of alcoholic beverages,” Dr. Potter explained. “On the other hand, the accumulation of acetaldehyde and the resultant flushing response are sufficiently unpleasant that they tend to reduce alcohol consumption among those with the Lys allele.”
The study results may therefore not be generalizable to a population in which the distribution of the variation in the ALDH2 enzyme differs, Dr. Potter added.
The lower prevalence of the inactive form (in North America, for instance) would mean that this lower prevalence was not a constraint on individuals’ drinking behavior as it is for some in Korea, Dr. Potter explained.
He noted another consideration: the underreporting of alcohol use is a well-known limitation of studies involving the assessment of alcohol consumption.
Dr. Shin agreed that underreporting is a limitation.
“People tend to underestimate their alcohol use,” Dr. Shin said in an interview.
However, he noted that “our study participants are health-screening participants aged 40 years and older, [and] people who participate in health screening tend to have higher awareness and better health behavior than nonparticipants.”
The authors and Dr. Potter disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
When weekly levels are similar
When weekly levels are similar
Alcohol use is a known risk factor for gastrointestinal (GI) cancers. Now, new research indicates that this risk is more associated with frequent drinking – even in smaller amounts – compared with higher amounts or binge drinking, given similar weekly levels.
“The novel finding of the current study is that frequent drinking may be more dangerous than binge drinking with regard to GI cancers. Alcohol use is a known risk factor for gastrointestinal (GI) cancers. Now, new research indicates that this risk is more associated with frequent drinking – even in smaller amounts -- compared with higher amounts or binge drinking, given similar weekly levels.” first author Jung Eun Yook, MD, of Seoul (South Korea) National University Hospital, and colleagues reported in an article published Aug. 18, 2021, in JAMA Network Open (doi: 10.1001/jamanetworkopen.2021.20382).
“This finding suggests that repeated alcohol consumption events even at lower amounts of alcohol may have a greater carcinogenic effect on GI organs than the consumption of larger amounts of alcohol at a lower frequency,” the investigators wrote.
A possible reason behind the difference in risk may be that the chronic “carcinogenic insult” from regular alcohol use may promote cancer development, whereas less frequent, episodic alcohol exposures may allow physiologic recovery, said the authors.
The results are from a population-based study that involved 11,737,467 participants in the Korean National Health System database who did not have cancer and who took part in a national screening program between January 2009 and December 2010.
They were followed from the year after their screening until either they had received a diagnosis of a GI cancer, death occurred, or the end of December 2017.
During a median follow-up of 6.4 years, 319,202 (2.7%) of those in the study developed a GI cancer.
The increase in the risk associated with alcohol consumption was dose dependent.
Compared with those who did not consume alcohol, the risk of developing GI cancer was higher for mild drinkers (adjusted hazard ratio, 1.04; 95% confidence interval, 1.03-1.05), moderate drinkers (aHR, 1.14; 95% CI, 1.12-1.15), and heavy drinkers (aHR, 1.28; 95% CI, 1.26-1.29), after adjusting for age, sex, income, smoking status with intensity, regular exercise, body mass index, diabetes, hypertension, and dyslipidemia.
There was a linear association between the frequency of drinking and GI cancer risk, with an aHR of 1.39 for individuals who reported drinking every day (95% CI, 1.36-1.41). The risk for GI cancer increased with consumption of five to seven units per occasion (aHR, 1.15). Notably, there were no similar increases with higher intake, including intake of 8-14 units per occasion (aHR, 1.11; 95% CI, 1.09-1.12), and even up to more than 14 units per occasion (aHR, 1.11; 95%CI, 1.08-1.14), in comparison with an intake of 5-7 units per occasion.
“Given similar weekly alcohol consumption levels, the risk of GI cancer increased with a higher frequency of drinking and decreased with a higher amount per occasion,” the authors write.
“Most previous studies just assess alcohol consumption as a total amount, [such as] drinks per occasion times occasion per week equals drinks per week [and] grams per week,” coauthor Dong Wook Shin, MD, DrPH, Samsung Medical Center, Sungkyunkwan University, Seoul, South Korea, said in an interview.
“But it was not known whether frequent drinking with small amount is more harmful than binge drinking, given a similar level of total drinking,” Dr. Shin said.
The increased risk associated with frequent drinking was generally similar with respect to esophageal, gastric, colorectal, biliary, and pancreatic cancer.
An exception was liver cancer, which showed a slightly decreased risk among mild drinkers (aHR, 0.91; 95% CI, 0.89-0.93).
Of note, the association between alcohol intake and the incidence of GI cancer was lower among women than men in terms of weekly consumption, frequency, and amount of alcohol consumed per occasion.
The associations between drinking and cancer type in terms of esophageal and liver cancers were similar between men and women. However, the alcohol-related risk for colorectal, biliary, and pancreatic cancers was less prominent for women.
Possible mechanisms related to regular drinking
A factor that might account for the increase in GI cancer risk with frequent drinking is that regular alcohol consumption “promotes the accumulation of cell divisions in the stem cells that maintain tissues in homeostasis,” the authors explained.
Another possible explanation is that long-term alcohol exposure may promote carcinogenesis, whereas less frequent exposure might allow “physiological homeostasis,” the authors wrote, adding that in vivo experiments have shown that duration and dose of alcohol exposure have been linked to cancer development.
Importantly, the findings support the importance of reducing the frequency of alcohol use to prevent cancer, the authors noted.
“Alcohol users who have a glass of wine or beer during dinner every day may develop more cancer than people who occasionally consume several drinks,” they cautioned.
Genetics, self-reporting considerations
In a related commentary, John D. Potter, MBBS, PhD, of the Research Center for Hauora and Health, Massey University, Wellington, New Zealand, noted that, in addition to supporting the known link between alcohol and cancers of the esophagus, colorectum, and liver, the study “strengthens evidence for a role of alcohol in stomach, biliary tract, and pancreas cancers.”
In comparison with nondrinkers, those who reported heavy drinking were much more likely to be smokers (51.6% vs. 9.0%); however, the study adjusted for smoking.
“Because the researchers were able to control for tobacco, this last finding [regarding the association with cancers of the stomach, biliary tract, and pancreas] is particularly informative,” Dr. Potter noted.
An important caveat is that more than a quarter of the Korean population is known to have an inactive form of the aldehyde dehydrogenase gene (ALDH2), which could have effects on alcohol metabolism as well as the risk for cancer, Dr. Potter wrote.
“This common polymorphism in ALDH2 (ALDH2 rs671 [c.1510G>A (Glu504Lys)]) has paradoxical effects,” he wrote.
“It increases the level of acetaldehyde in the blood of drinkers, which in turn increases the risk of cancer because acetaldehyde is a key player in the carcinogenicity of alcoholic beverages,” Dr. Potter explained. “On the other hand, the accumulation of acetaldehyde and the resultant flushing response are sufficiently unpleasant that they tend to reduce alcohol consumption among those with the Lys allele.”
The study results may therefore not be generalizable to a population in which the distribution of the variation in the ALDH2 enzyme differs, Dr. Potter added.
The lower prevalence of the inactive form (in North America, for instance) would mean that this lower prevalence was not a constraint on individuals’ drinking behavior as it is for some in Korea, Dr. Potter explained.
He noted another consideration: the underreporting of alcohol use is a well-known limitation of studies involving the assessment of alcohol consumption.
Dr. Shin agreed that underreporting is a limitation.
“People tend to underestimate their alcohol use,” Dr. Shin said in an interview.
However, he noted that “our study participants are health-screening participants aged 40 years and older, [and] people who participate in health screening tend to have higher awareness and better health behavior than nonparticipants.”
The authors and Dr. Potter disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Alcohol use is a known risk factor for gastrointestinal (GI) cancers. Now, new research indicates that this risk is more associated with frequent drinking – even in smaller amounts – compared with higher amounts or binge drinking, given similar weekly levels.
“The novel finding of the current study is that frequent drinking may be more dangerous than binge drinking with regard to GI cancers. Alcohol use is a known risk factor for gastrointestinal (GI) cancers. Now, new research indicates that this risk is more associated with frequent drinking – even in smaller amounts -- compared with higher amounts or binge drinking, given similar weekly levels.” first author Jung Eun Yook, MD, of Seoul (South Korea) National University Hospital, and colleagues reported in an article published Aug. 18, 2021, in JAMA Network Open (doi: 10.1001/jamanetworkopen.2021.20382).
“This finding suggests that repeated alcohol consumption events even at lower amounts of alcohol may have a greater carcinogenic effect on GI organs than the consumption of larger amounts of alcohol at a lower frequency,” the investigators wrote.
A possible reason behind the difference in risk may be that the chronic “carcinogenic insult” from regular alcohol use may promote cancer development, whereas less frequent, episodic alcohol exposures may allow physiologic recovery, said the authors.
The results are from a population-based study that involved 11,737,467 participants in the Korean National Health System database who did not have cancer and who took part in a national screening program between January 2009 and December 2010.
They were followed from the year after their screening until either they had received a diagnosis of a GI cancer, death occurred, or the end of December 2017.
During a median follow-up of 6.4 years, 319,202 (2.7%) of those in the study developed a GI cancer.
The increase in the risk associated with alcohol consumption was dose dependent.
Compared with those who did not consume alcohol, the risk of developing GI cancer was higher for mild drinkers (adjusted hazard ratio, 1.04; 95% confidence interval, 1.03-1.05), moderate drinkers (aHR, 1.14; 95% CI, 1.12-1.15), and heavy drinkers (aHR, 1.28; 95% CI, 1.26-1.29), after adjusting for age, sex, income, smoking status with intensity, regular exercise, body mass index, diabetes, hypertension, and dyslipidemia.
There was a linear association between the frequency of drinking and GI cancer risk, with an aHR of 1.39 for individuals who reported drinking every day (95% CI, 1.36-1.41). The risk for GI cancer increased with consumption of five to seven units per occasion (aHR, 1.15). Notably, there were no similar increases with higher intake, including intake of 8-14 units per occasion (aHR, 1.11; 95% CI, 1.09-1.12), and even up to more than 14 units per occasion (aHR, 1.11; 95%CI, 1.08-1.14), in comparison with an intake of 5-7 units per occasion.
“Given similar weekly alcohol consumption levels, the risk of GI cancer increased with a higher frequency of drinking and decreased with a higher amount per occasion,” the authors write.
“Most previous studies just assess alcohol consumption as a total amount, [such as] drinks per occasion times occasion per week equals drinks per week [and] grams per week,” coauthor Dong Wook Shin, MD, DrPH, Samsung Medical Center, Sungkyunkwan University, Seoul, South Korea, said in an interview.
“But it was not known whether frequent drinking with small amount is more harmful than binge drinking, given a similar level of total drinking,” Dr. Shin said.
The increased risk associated with frequent drinking was generally similar with respect to esophageal, gastric, colorectal, biliary, and pancreatic cancer.
An exception was liver cancer, which showed a slightly decreased risk among mild drinkers (aHR, 0.91; 95% CI, 0.89-0.93).
Of note, the association between alcohol intake and the incidence of GI cancer was lower among women than men in terms of weekly consumption, frequency, and amount of alcohol consumed per occasion.
The associations between drinking and cancer type in terms of esophageal and liver cancers were similar between men and women. However, the alcohol-related risk for colorectal, biliary, and pancreatic cancers was less prominent for women.
Possible mechanisms related to regular drinking
A factor that might account for the increase in GI cancer risk with frequent drinking is that regular alcohol consumption “promotes the accumulation of cell divisions in the stem cells that maintain tissues in homeostasis,” the authors explained.
Another possible explanation is that long-term alcohol exposure may promote carcinogenesis, whereas less frequent exposure might allow “physiological homeostasis,” the authors wrote, adding that in vivo experiments have shown that duration and dose of alcohol exposure have been linked to cancer development.
Importantly, the findings support the importance of reducing the frequency of alcohol use to prevent cancer, the authors noted.
“Alcohol users who have a glass of wine or beer during dinner every day may develop more cancer than people who occasionally consume several drinks,” they cautioned.
Genetics, self-reporting considerations
In a related commentary, John D. Potter, MBBS, PhD, of the Research Center for Hauora and Health, Massey University, Wellington, New Zealand, noted that, in addition to supporting the known link between alcohol and cancers of the esophagus, colorectum, and liver, the study “strengthens evidence for a role of alcohol in stomach, biliary tract, and pancreas cancers.”
In comparison with nondrinkers, those who reported heavy drinking were much more likely to be smokers (51.6% vs. 9.0%); however, the study adjusted for smoking.
“Because the researchers were able to control for tobacco, this last finding [regarding the association with cancers of the stomach, biliary tract, and pancreas] is particularly informative,” Dr. Potter noted.
An important caveat is that more than a quarter of the Korean population is known to have an inactive form of the aldehyde dehydrogenase gene (ALDH2), which could have effects on alcohol metabolism as well as the risk for cancer, Dr. Potter wrote.
“This common polymorphism in ALDH2 (ALDH2 rs671 [c.1510G>A (Glu504Lys)]) has paradoxical effects,” he wrote.
“It increases the level of acetaldehyde in the blood of drinkers, which in turn increases the risk of cancer because acetaldehyde is a key player in the carcinogenicity of alcoholic beverages,” Dr. Potter explained. “On the other hand, the accumulation of acetaldehyde and the resultant flushing response are sufficiently unpleasant that they tend to reduce alcohol consumption among those with the Lys allele.”
The study results may therefore not be generalizable to a population in which the distribution of the variation in the ALDH2 enzyme differs, Dr. Potter added.
The lower prevalence of the inactive form (in North America, for instance) would mean that this lower prevalence was not a constraint on individuals’ drinking behavior as it is for some in Korea, Dr. Potter explained.
He noted another consideration: the underreporting of alcohol use is a well-known limitation of studies involving the assessment of alcohol consumption.
Dr. Shin agreed that underreporting is a limitation.
“People tend to underestimate their alcohol use,” Dr. Shin said in an interview.
However, he noted that “our study participants are health-screening participants aged 40 years and older, [and] people who participate in health screening tend to have higher awareness and better health behavior than nonparticipants.”
The authors and Dr. Potter disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Fauci corrects prediction on when pandemic will be under control
The United States could get the COVID-19 pandemic under control by the spring of 2022 if enough Americans become vaccinated, Anthony S. Fauci, MD, said.
Speaking to Anderson Cooper on CNN, Dr. Fauci corrected the timeline he gave in an interview earlier with Mary Louise Kelly of NPR.
In the NPR interview, he had said that if “the overwhelming majority of the people vaccinated, I think as we get into the fall and the winter, we could start to really get some good control over this as we get into 2022.”
Dr. Fauci told Mr. Cooper that he listened to a recording of the NPR interview later and realized his mistake.
“I meant to say the spring of 2022,” Dr. Fauci told CNN. “I misspoke. My bad.”
Dr. Fauci, the head of the National Institute of Allergy and Infectious Diseases and the chief White House medical adviser, said the pandemic will be under control when the large majority of Americans have gotten vaccinated or been infected with COVID-19 and recovered, which offers some protection against the virus.
People who have been infected and recovered should still get vaccinated, he said.
“The degree of protection you could induce in someone who’s been infected and then recovered and then vaccinated is an enormous increase in the degree of protection,” Dr. Fauci said.
“I think we can get a degree of overall blanket protection of the community that as we get into the early part of 2022 ... we could start getting back to a degree of normality.”
A version of this article first appeared on WebMD.com.
The United States could get the COVID-19 pandemic under control by the spring of 2022 if enough Americans become vaccinated, Anthony S. Fauci, MD, said.
Speaking to Anderson Cooper on CNN, Dr. Fauci corrected the timeline he gave in an interview earlier with Mary Louise Kelly of NPR.
In the NPR interview, he had said that if “the overwhelming majority of the people vaccinated, I think as we get into the fall and the winter, we could start to really get some good control over this as we get into 2022.”
Dr. Fauci told Mr. Cooper that he listened to a recording of the NPR interview later and realized his mistake.
“I meant to say the spring of 2022,” Dr. Fauci told CNN. “I misspoke. My bad.”
Dr. Fauci, the head of the National Institute of Allergy and Infectious Diseases and the chief White House medical adviser, said the pandemic will be under control when the large majority of Americans have gotten vaccinated or been infected with COVID-19 and recovered, which offers some protection against the virus.
People who have been infected and recovered should still get vaccinated, he said.
“The degree of protection you could induce in someone who’s been infected and then recovered and then vaccinated is an enormous increase in the degree of protection,” Dr. Fauci said.
“I think we can get a degree of overall blanket protection of the community that as we get into the early part of 2022 ... we could start getting back to a degree of normality.”
A version of this article first appeared on WebMD.com.
The United States could get the COVID-19 pandemic under control by the spring of 2022 if enough Americans become vaccinated, Anthony S. Fauci, MD, said.
Speaking to Anderson Cooper on CNN, Dr. Fauci corrected the timeline he gave in an interview earlier with Mary Louise Kelly of NPR.
In the NPR interview, he had said that if “the overwhelming majority of the people vaccinated, I think as we get into the fall and the winter, we could start to really get some good control over this as we get into 2022.”
Dr. Fauci told Mr. Cooper that he listened to a recording of the NPR interview later and realized his mistake.
“I meant to say the spring of 2022,” Dr. Fauci told CNN. “I misspoke. My bad.”
Dr. Fauci, the head of the National Institute of Allergy and Infectious Diseases and the chief White House medical adviser, said the pandemic will be under control when the large majority of Americans have gotten vaccinated or been infected with COVID-19 and recovered, which offers some protection against the virus.
People who have been infected and recovered should still get vaccinated, he said.
“The degree of protection you could induce in someone who’s been infected and then recovered and then vaccinated is an enormous increase in the degree of protection,” Dr. Fauci said.
“I think we can get a degree of overall blanket protection of the community that as we get into the early part of 2022 ... we could start getting back to a degree of normality.”
A version of this article first appeared on WebMD.com.
COVID booster may benefit active-treatment cancer patients
A COVID-19 booster shot may be beneficial for patients with cancer who are undergoing treatment, according to new findings from an Israeli case-control study.
The seropositivity rate among the patients with cancer remained high (87%) about 4 months after the patients had received the second BNT162b2 (Pfizer/BioNTech) vaccination. However, the median IgG titer in the patients and the control persons who were without cancer decreased over time. Notably, in a previous analysis that the authors conducted and in the current one, the IgG titers were statistically significantly lower in the patients with cancer as compared to control persons.
The correlation between antibody levels following vaccination and clinical protection has yet to be proven, but the accumulating evidence supports antibody response as a possible correlate of disease protection.
“Our data can’t predict if a third booster dose is necessary,” said study author Salomon M. Stemmer, MD, professor at the Institute of Oncology of Rabin Medical Center, Petah Tikva, Israel. “It does seem quite logical that a booster dose will cause an increase in IgG levels.”
The findings were published Aug. 11, 2021, in a research letter in JAMA Oncology.
In their previous study, Dr. Stemmer and colleagues compared the rates of anti–spike antibody response to the initial shot of the BNT162b2 vaccine among 102 adults with solid-tumor cancers who were undergoing treatment with that of 78 healthy control persons. They found that a high percentage of patients undergoing treatment for cancer (90%) achieved a sufficient antibody response to the BNT162b2 vaccine.
Booster endorsed
Responses to COVID-19 vaccination have varied among patients with cancer. For patients with solid tumors, responses have been good even while the patients were receiving systemic therapy. However, among patients with blood cancers, particularly those receiving immunosuppressive therapies, responses have been poor. Studies have identified factors associated with a poor response, but it has been unclear whether to recommend booster shots.
In August the Food and Drug Administration authorized a third dose of either the Pfizer or the Moderna COVID-19 vaccine for all individuals with compromised immune systems. Those eligible for a third dose include solid-organ transplant recipients, those undergoing cancer treatments, and people with autoimmune diseases that suppress their immune systems.
IgG titers lower in cancer patients
In the current analysis, the authors evaluated the anti-S response in the patients with cancer approximately 4 months after they had received the second vaccine dose. They compared the responses in those patients with the responses in a control group.
The cohort included 95 patients from the prior study and 66 control persons. The most common malignancies were gastrointestinal (26%), lung (25%), and breast (18%).
All patients were receiving systemic therapy. Chemotherapy was the most common (28%), followed by immunotherapy (21%) and combination chemotherapy/biological therapy (20%).
At a median of 123 days after the second vaccination, 83 patients with cancer (87%) and all of the control patients (100%) were seropositive for anti-S IgG antibodies. The median titer levels were significantly lower among case patients as compared with control patients (417 AU/mL [interquartile range, 136-895] vs. 1,220 AU/mL [IQR, 588-1,987]; P < .001)
There was a 3.6-fold range in median titer values across tumor types and an even wider range (8.8-fold) across the different types of treatment. The lowest titers were observed among patients who had received immunotherapy plus chemotherapy/biological therapy (median [IQR], 94.4 [49.4-191] AU/mL vs. 147 [62.8-339] AU/mL).
In an exploratory multivariable analysis, treatments with chemotherapy plus immunotherapy and immunotherapy plus biological therapy were significantly associated with lower IgG titers.
No downside for cancer patients
The Biden administration announced a plan to begin booster COVID-19 vaccinations for all American adults in September, with recommendations that the third vaccine be given at least 8 months after the second mRNA vaccine dose.
Jeremy M. Levin, DPhil, the chairman and CEO of Ovid Therapeutics, explained that, concerning boosters, “it is inconceivable that we will have all data at this stage.
“Knowledge about how boosters work and don’t work and when you should ideally have them is imperfect,” he told this news organization. “However, we can have a lot of confidence in the fact that hundreds of millions of people have received the vaccine, so we know a lot about the safety and efficacy.”
Immunocompromised adults represent less than 5% of the total population, and most of the available data on vaccination are from patients who have undergone solid-organ transplant, Dr. Levin explained. Studies have shown that their response is less robust to vaccination in comparison with adults in the general population.
“Although it is still preliminary, the strongest data come from Israel,” he said, “where they found that the booster was highly effective and doubled the number of transplant patients who developed antibodies.”
But data are not yet available in the setting of cancer. “But even though we don’t have the data yet, the answer is that no matter, the booster process is essential,” he said. “The evidence we have is that boosters raise the immune response, and it is the best data we have now.”
Martin J. Edelman, MD, chair, department of hematology/oncology, Fox Chase Cancer Center, Philadelphia, noted that the current recommendation is that patients who are immunocompromised receive a booster immediately.
At his health system, this is interpreted to include patients who have undergone the following treatments: Transplant (solid-organ and bone marrow transplant), hemodialysis, hematologic malignancy treatment, active immunosuppressive (chemotherapy, chemoimmunotherapy, and nonhormonal or single-agent immunotherapy) treatment, rheumatology treatments, and high-dose steroids.
“As for cancer patients, we are making arrangements to vaccinate patients who meet the above criteria now,” he said. “There is no known downside to receiving booster immediately. While there may be less of a response than waiting for completion of treatment, we know that patients on active therapy are frequently able to mount a response, and any response is better than none.”
Dr. Edelman added that this area is changing very rapidly. “We will modify our approach as information and guidance from appropriate organizations, such as the FDA and CDC, become available.”
Dr. Stemmer has received institutional research grants from CAN-FITE, AstraZeneca, Bioline RX, BMS, Halozyme, Clovis Oncology, CTG Pharma, Exelixis, Geicam, Incyte, Lilly, Moderna, Teva Pharmaceuticals, and Roche, and owns stocks and options in CTG Pharma, DocBoxMD, Tyrnovo, VYPE, Cytora, and CAN-FITE. Dr. Edelman has received personal fees and other compensation from Windmil, Biomarker Strategies, AstraZeneca, Takeda, GlaxoSmithKline, Apexigen, Nektar, Bristol-Myers Squibb, Armo, Bergen Bio, and Apexigen outside the submitted work. He has submitted a patent for epigenetic modifications to increase susceptibility to radiopharmaceuticals and is a paid adviser for Kanaph and Flame. Dr. Levin is chairman and CEO of Ovid Therapeutics.
A version of this article first appeared on Medscape.com.
A COVID-19 booster shot may be beneficial for patients with cancer who are undergoing treatment, according to new findings from an Israeli case-control study.
The seropositivity rate among the patients with cancer remained high (87%) about 4 months after the patients had received the second BNT162b2 (Pfizer/BioNTech) vaccination. However, the median IgG titer in the patients and the control persons who were without cancer decreased over time. Notably, in a previous analysis that the authors conducted and in the current one, the IgG titers were statistically significantly lower in the patients with cancer as compared to control persons.
The correlation between antibody levels following vaccination and clinical protection has yet to be proven, but the accumulating evidence supports antibody response as a possible correlate of disease protection.
“Our data can’t predict if a third booster dose is necessary,” said study author Salomon M. Stemmer, MD, professor at the Institute of Oncology of Rabin Medical Center, Petah Tikva, Israel. “It does seem quite logical that a booster dose will cause an increase in IgG levels.”
The findings were published Aug. 11, 2021, in a research letter in JAMA Oncology.
In their previous study, Dr. Stemmer and colleagues compared the rates of anti–spike antibody response to the initial shot of the BNT162b2 vaccine among 102 adults with solid-tumor cancers who were undergoing treatment with that of 78 healthy control persons. They found that a high percentage of patients undergoing treatment for cancer (90%) achieved a sufficient antibody response to the BNT162b2 vaccine.
Booster endorsed
Responses to COVID-19 vaccination have varied among patients with cancer. For patients with solid tumors, responses have been good even while the patients were receiving systemic therapy. However, among patients with blood cancers, particularly those receiving immunosuppressive therapies, responses have been poor. Studies have identified factors associated with a poor response, but it has been unclear whether to recommend booster shots.
In August the Food and Drug Administration authorized a third dose of either the Pfizer or the Moderna COVID-19 vaccine for all individuals with compromised immune systems. Those eligible for a third dose include solid-organ transplant recipients, those undergoing cancer treatments, and people with autoimmune diseases that suppress their immune systems.
IgG titers lower in cancer patients
In the current analysis, the authors evaluated the anti-S response in the patients with cancer approximately 4 months after they had received the second vaccine dose. They compared the responses in those patients with the responses in a control group.
The cohort included 95 patients from the prior study and 66 control persons. The most common malignancies were gastrointestinal (26%), lung (25%), and breast (18%).
All patients were receiving systemic therapy. Chemotherapy was the most common (28%), followed by immunotherapy (21%) and combination chemotherapy/biological therapy (20%).
At a median of 123 days after the second vaccination, 83 patients with cancer (87%) and all of the control patients (100%) were seropositive for anti-S IgG antibodies. The median titer levels were significantly lower among case patients as compared with control patients (417 AU/mL [interquartile range, 136-895] vs. 1,220 AU/mL [IQR, 588-1,987]; P < .001)
There was a 3.6-fold range in median titer values across tumor types and an even wider range (8.8-fold) across the different types of treatment. The lowest titers were observed among patients who had received immunotherapy plus chemotherapy/biological therapy (median [IQR], 94.4 [49.4-191] AU/mL vs. 147 [62.8-339] AU/mL).
In an exploratory multivariable analysis, treatments with chemotherapy plus immunotherapy and immunotherapy plus biological therapy were significantly associated with lower IgG titers.
No downside for cancer patients
The Biden administration announced a plan to begin booster COVID-19 vaccinations for all American adults in September, with recommendations that the third vaccine be given at least 8 months after the second mRNA vaccine dose.
Jeremy M. Levin, DPhil, the chairman and CEO of Ovid Therapeutics, explained that, concerning boosters, “it is inconceivable that we will have all data at this stage.
“Knowledge about how boosters work and don’t work and when you should ideally have them is imperfect,” he told this news organization. “However, we can have a lot of confidence in the fact that hundreds of millions of people have received the vaccine, so we know a lot about the safety and efficacy.”
Immunocompromised adults represent less than 5% of the total population, and most of the available data on vaccination are from patients who have undergone solid-organ transplant, Dr. Levin explained. Studies have shown that their response is less robust to vaccination in comparison with adults in the general population.
“Although it is still preliminary, the strongest data come from Israel,” he said, “where they found that the booster was highly effective and doubled the number of transplant patients who developed antibodies.”
But data are not yet available in the setting of cancer. “But even though we don’t have the data yet, the answer is that no matter, the booster process is essential,” he said. “The evidence we have is that boosters raise the immune response, and it is the best data we have now.”
Martin J. Edelman, MD, chair, department of hematology/oncology, Fox Chase Cancer Center, Philadelphia, noted that the current recommendation is that patients who are immunocompromised receive a booster immediately.
At his health system, this is interpreted to include patients who have undergone the following treatments: Transplant (solid-organ and bone marrow transplant), hemodialysis, hematologic malignancy treatment, active immunosuppressive (chemotherapy, chemoimmunotherapy, and nonhormonal or single-agent immunotherapy) treatment, rheumatology treatments, and high-dose steroids.
“As for cancer patients, we are making arrangements to vaccinate patients who meet the above criteria now,” he said. “There is no known downside to receiving booster immediately. While there may be less of a response than waiting for completion of treatment, we know that patients on active therapy are frequently able to mount a response, and any response is better than none.”
Dr. Edelman added that this area is changing very rapidly. “We will modify our approach as information and guidance from appropriate organizations, such as the FDA and CDC, become available.”
Dr. Stemmer has received institutional research grants from CAN-FITE, AstraZeneca, Bioline RX, BMS, Halozyme, Clovis Oncology, CTG Pharma, Exelixis, Geicam, Incyte, Lilly, Moderna, Teva Pharmaceuticals, and Roche, and owns stocks and options in CTG Pharma, DocBoxMD, Tyrnovo, VYPE, Cytora, and CAN-FITE. Dr. Edelman has received personal fees and other compensation from Windmil, Biomarker Strategies, AstraZeneca, Takeda, GlaxoSmithKline, Apexigen, Nektar, Bristol-Myers Squibb, Armo, Bergen Bio, and Apexigen outside the submitted work. He has submitted a patent for epigenetic modifications to increase susceptibility to radiopharmaceuticals and is a paid adviser for Kanaph and Flame. Dr. Levin is chairman and CEO of Ovid Therapeutics.
A version of this article first appeared on Medscape.com.
A COVID-19 booster shot may be beneficial for patients with cancer who are undergoing treatment, according to new findings from an Israeli case-control study.
The seropositivity rate among the patients with cancer remained high (87%) about 4 months after the patients had received the second BNT162b2 (Pfizer/BioNTech) vaccination. However, the median IgG titer in the patients and the control persons who were without cancer decreased over time. Notably, in a previous analysis that the authors conducted and in the current one, the IgG titers were statistically significantly lower in the patients with cancer as compared to control persons.
The correlation between antibody levels following vaccination and clinical protection has yet to be proven, but the accumulating evidence supports antibody response as a possible correlate of disease protection.
“Our data can’t predict if a third booster dose is necessary,” said study author Salomon M. Stemmer, MD, professor at the Institute of Oncology of Rabin Medical Center, Petah Tikva, Israel. “It does seem quite logical that a booster dose will cause an increase in IgG levels.”
The findings were published Aug. 11, 2021, in a research letter in JAMA Oncology.
In their previous study, Dr. Stemmer and colleagues compared the rates of anti–spike antibody response to the initial shot of the BNT162b2 vaccine among 102 adults with solid-tumor cancers who were undergoing treatment with that of 78 healthy control persons. They found that a high percentage of patients undergoing treatment for cancer (90%) achieved a sufficient antibody response to the BNT162b2 vaccine.
Booster endorsed
Responses to COVID-19 vaccination have varied among patients with cancer. For patients with solid tumors, responses have been good even while the patients were receiving systemic therapy. However, among patients with blood cancers, particularly those receiving immunosuppressive therapies, responses have been poor. Studies have identified factors associated with a poor response, but it has been unclear whether to recommend booster shots.
In August the Food and Drug Administration authorized a third dose of either the Pfizer or the Moderna COVID-19 vaccine for all individuals with compromised immune systems. Those eligible for a third dose include solid-organ transplant recipients, those undergoing cancer treatments, and people with autoimmune diseases that suppress their immune systems.
IgG titers lower in cancer patients
In the current analysis, the authors evaluated the anti-S response in the patients with cancer approximately 4 months after they had received the second vaccine dose. They compared the responses in those patients with the responses in a control group.
The cohort included 95 patients from the prior study and 66 control persons. The most common malignancies were gastrointestinal (26%), lung (25%), and breast (18%).
All patients were receiving systemic therapy. Chemotherapy was the most common (28%), followed by immunotherapy (21%) and combination chemotherapy/biological therapy (20%).
At a median of 123 days after the second vaccination, 83 patients with cancer (87%) and all of the control patients (100%) were seropositive for anti-S IgG antibodies. The median titer levels were significantly lower among case patients as compared with control patients (417 AU/mL [interquartile range, 136-895] vs. 1,220 AU/mL [IQR, 588-1,987]; P < .001)
There was a 3.6-fold range in median titer values across tumor types and an even wider range (8.8-fold) across the different types of treatment. The lowest titers were observed among patients who had received immunotherapy plus chemotherapy/biological therapy (median [IQR], 94.4 [49.4-191] AU/mL vs. 147 [62.8-339] AU/mL).
In an exploratory multivariable analysis, treatments with chemotherapy plus immunotherapy and immunotherapy plus biological therapy were significantly associated with lower IgG titers.
No downside for cancer patients
The Biden administration announced a plan to begin booster COVID-19 vaccinations for all American adults in September, with recommendations that the third vaccine be given at least 8 months after the second mRNA vaccine dose.
Jeremy M. Levin, DPhil, the chairman and CEO of Ovid Therapeutics, explained that, concerning boosters, “it is inconceivable that we will have all data at this stage.
“Knowledge about how boosters work and don’t work and when you should ideally have them is imperfect,” he told this news organization. “However, we can have a lot of confidence in the fact that hundreds of millions of people have received the vaccine, so we know a lot about the safety and efficacy.”
Immunocompromised adults represent less than 5% of the total population, and most of the available data on vaccination are from patients who have undergone solid-organ transplant, Dr. Levin explained. Studies have shown that their response is less robust to vaccination in comparison with adults in the general population.
“Although it is still preliminary, the strongest data come from Israel,” he said, “where they found that the booster was highly effective and doubled the number of transplant patients who developed antibodies.”
But data are not yet available in the setting of cancer. “But even though we don’t have the data yet, the answer is that no matter, the booster process is essential,” he said. “The evidence we have is that boosters raise the immune response, and it is the best data we have now.”
Martin J. Edelman, MD, chair, department of hematology/oncology, Fox Chase Cancer Center, Philadelphia, noted that the current recommendation is that patients who are immunocompromised receive a booster immediately.
At his health system, this is interpreted to include patients who have undergone the following treatments: Transplant (solid-organ and bone marrow transplant), hemodialysis, hematologic malignancy treatment, active immunosuppressive (chemotherapy, chemoimmunotherapy, and nonhormonal or single-agent immunotherapy) treatment, rheumatology treatments, and high-dose steroids.
“As for cancer patients, we are making arrangements to vaccinate patients who meet the above criteria now,” he said. “There is no known downside to receiving booster immediately. While there may be less of a response than waiting for completion of treatment, we know that patients on active therapy are frequently able to mount a response, and any response is better than none.”
Dr. Edelman added that this area is changing very rapidly. “We will modify our approach as information and guidance from appropriate organizations, such as the FDA and CDC, become available.”
Dr. Stemmer has received institutional research grants from CAN-FITE, AstraZeneca, Bioline RX, BMS, Halozyme, Clovis Oncology, CTG Pharma, Exelixis, Geicam, Incyte, Lilly, Moderna, Teva Pharmaceuticals, and Roche, and owns stocks and options in CTG Pharma, DocBoxMD, Tyrnovo, VYPE, Cytora, and CAN-FITE. Dr. Edelman has received personal fees and other compensation from Windmil, Biomarker Strategies, AstraZeneca, Takeda, GlaxoSmithKline, Apexigen, Nektar, Bristol-Myers Squibb, Armo, Bergen Bio, and Apexigen outside the submitted work. He has submitted a patent for epigenetic modifications to increase susceptibility to radiopharmaceuticals and is a paid adviser for Kanaph and Flame. Dr. Levin is chairman and CEO of Ovid Therapeutics.
A version of this article first appeared on Medscape.com.
Children and COVID: New cases soar to near-record level
Weekly cases of COVID-19 in children jumped by nearly 50% in the United States, posting the highest count since hitting a pandemic high back in mid-January, a new report shows.
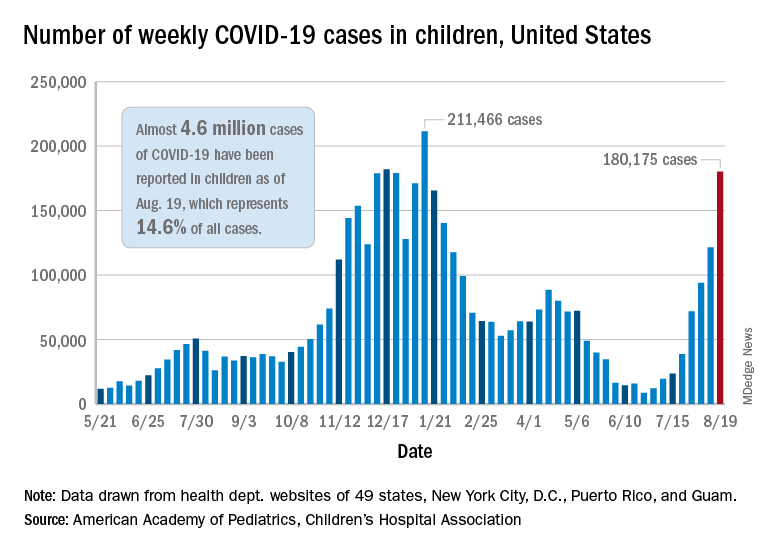
The latest weekly figure represents a 48% increase over the previous week and an increase of over 2,000% in the 8 weeks since the national count dropped to a low of 8,500 cases for the week of June 18-24, the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID report.
Vaccinations, in the meantime, appear to be headed in the opposite direction. Vaccine initiations were down for the second consecutive week, falling by 18% among 12- to 15-year-olds and by 15% in those aged 16-17 years, according to data from the Centers for Disease Control and Prevention.
Nationally, about 47% of children aged 12-15 and 56% of those aged 16-17 have received at least one dose of COVID vaccine as of Aug. 23, with 34% and 44%, respectively, reaching full vaccination. The total number of children with at least one dose is 11.6 million, including a relatively small number (about 200,000) of children under age 12 years, the CDC said on its COVID Data Tracker.
At the state level, vaccination is a source of considerable disparity. In Vermont, 73% of children aged 12-17 had received at least one dose by Aug. 18, and 63% were fully vaccinated. In Wyoming, however, just 25% of children had received at least one dose (17% are fully vaccinated), while Alabama has a lowest-in-the-nation full vaccination rate of 14%, based on a separate AAP analysis of CDC data.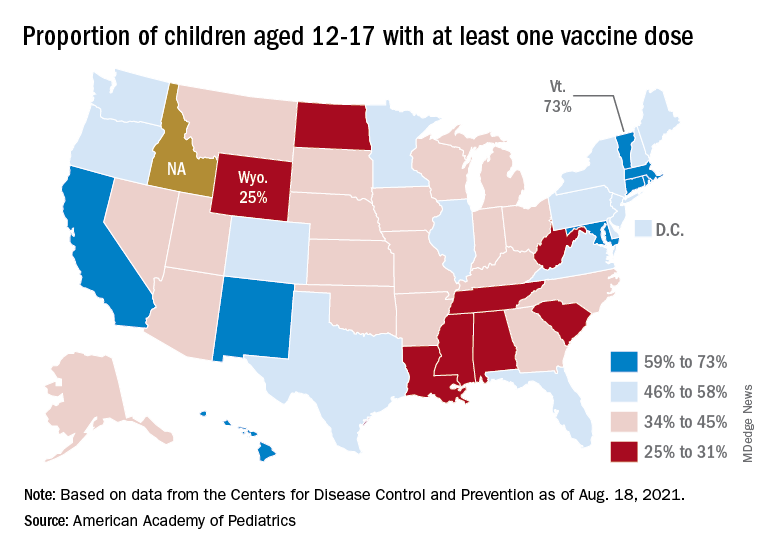
There are seven states in which over 60% of 12- to 17-year-olds have at least started the vaccine regimen and five states where less than 30% have received at least one dose, the AAP noted.
Back on the incidence side of the pandemic, Mississippi and Hawaii had the largest increases in new cases over the past 2 weeks, followed by Florida and West Virginia. Cumulative figures show that California has had the most cases overall in children (550,337), Vermont has the highest proportion of all cases in children (22.9%), and Rhode Island has the highest rate of cases per 100,000 (10,636), the AAP and CHA said in the joint report based on data from 49 states, the District of Columbia, New York City, Puerto Rico, and Guam.
Add up all those jurisdictions, and it works out to 4.6 million children infected with SARS-CoV-2 as of Aug. 19, with children representing 14.6% of all cases since the start of the pandemic. There have been over 18,000 hospitalizations so far, which is just 2.3% of the total for all ages in the 23 states (and New York City) that are reporting such data on their health department websites, the AAP and CHA said.
The number of COVID-related deaths in children is now 402 after the largest 1-week increase (24) since late May of 2020, when the AAP/CHA coverage began. Mortality data by age are available from 44 states, New York City, Puerto Rico, and Guam.
Weekly cases of COVID-19 in children jumped by nearly 50% in the United States, posting the highest count since hitting a pandemic high back in mid-January, a new report shows.

The latest weekly figure represents a 48% increase over the previous week and an increase of over 2,000% in the 8 weeks since the national count dropped to a low of 8,500 cases for the week of June 18-24, the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID report.
Vaccinations, in the meantime, appear to be headed in the opposite direction. Vaccine initiations were down for the second consecutive week, falling by 18% among 12- to 15-year-olds and by 15% in those aged 16-17 years, according to data from the Centers for Disease Control and Prevention.
Nationally, about 47% of children aged 12-15 and 56% of those aged 16-17 have received at least one dose of COVID vaccine as of Aug. 23, with 34% and 44%, respectively, reaching full vaccination. The total number of children with at least one dose is 11.6 million, including a relatively small number (about 200,000) of children under age 12 years, the CDC said on its COVID Data Tracker.
At the state level, vaccination is a source of considerable disparity. In Vermont, 73% of children aged 12-17 had received at least one dose by Aug. 18, and 63% were fully vaccinated. In Wyoming, however, just 25% of children had received at least one dose (17% are fully vaccinated), while Alabama has a lowest-in-the-nation full vaccination rate of 14%, based on a separate AAP analysis of CDC data.
There are seven states in which over 60% of 12- to 17-year-olds have at least started the vaccine regimen and five states where less than 30% have received at least one dose, the AAP noted.
Back on the incidence side of the pandemic, Mississippi and Hawaii had the largest increases in new cases over the past 2 weeks, followed by Florida and West Virginia. Cumulative figures show that California has had the most cases overall in children (550,337), Vermont has the highest proportion of all cases in children (22.9%), and Rhode Island has the highest rate of cases per 100,000 (10,636), the AAP and CHA said in the joint report based on data from 49 states, the District of Columbia, New York City, Puerto Rico, and Guam.
Add up all those jurisdictions, and it works out to 4.6 million children infected with SARS-CoV-2 as of Aug. 19, with children representing 14.6% of all cases since the start of the pandemic. There have been over 18,000 hospitalizations so far, which is just 2.3% of the total for all ages in the 23 states (and New York City) that are reporting such data on their health department websites, the AAP and CHA said.
The number of COVID-related deaths in children is now 402 after the largest 1-week increase (24) since late May of 2020, when the AAP/CHA coverage began. Mortality data by age are available from 44 states, New York City, Puerto Rico, and Guam.
Weekly cases of COVID-19 in children jumped by nearly 50% in the United States, posting the highest count since hitting a pandemic high back in mid-January, a new report shows.

The latest weekly figure represents a 48% increase over the previous week and an increase of over 2,000% in the 8 weeks since the national count dropped to a low of 8,500 cases for the week of June 18-24, the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID report.
Vaccinations, in the meantime, appear to be headed in the opposite direction. Vaccine initiations were down for the second consecutive week, falling by 18% among 12- to 15-year-olds and by 15% in those aged 16-17 years, according to data from the Centers for Disease Control and Prevention.
Nationally, about 47% of children aged 12-15 and 56% of those aged 16-17 have received at least one dose of COVID vaccine as of Aug. 23, with 34% and 44%, respectively, reaching full vaccination. The total number of children with at least one dose is 11.6 million, including a relatively small number (about 200,000) of children under age 12 years, the CDC said on its COVID Data Tracker.
At the state level, vaccination is a source of considerable disparity. In Vermont, 73% of children aged 12-17 had received at least one dose by Aug. 18, and 63% were fully vaccinated. In Wyoming, however, just 25% of children had received at least one dose (17% are fully vaccinated), while Alabama has a lowest-in-the-nation full vaccination rate of 14%, based on a separate AAP analysis of CDC data.
There are seven states in which over 60% of 12- to 17-year-olds have at least started the vaccine regimen and five states where less than 30% have received at least one dose, the AAP noted.
Back on the incidence side of the pandemic, Mississippi and Hawaii had the largest increases in new cases over the past 2 weeks, followed by Florida and West Virginia. Cumulative figures show that California has had the most cases overall in children (550,337), Vermont has the highest proportion of all cases in children (22.9%), and Rhode Island has the highest rate of cases per 100,000 (10,636), the AAP and CHA said in the joint report based on data from 49 states, the District of Columbia, New York City, Puerto Rico, and Guam.
Add up all those jurisdictions, and it works out to 4.6 million children infected with SARS-CoV-2 as of Aug. 19, with children representing 14.6% of all cases since the start of the pandemic. There have been over 18,000 hospitalizations so far, which is just 2.3% of the total for all ages in the 23 states (and New York City) that are reporting such data on their health department websites, the AAP and CHA said.
The number of COVID-related deaths in children is now 402 after the largest 1-week increase (24) since late May of 2020, when the AAP/CHA coverage began. Mortality data by age are available from 44 states, New York City, Puerto Rico, and Guam.
Prevalence of high-risk HPV types dwindled since vaccine approval
Young women who received the quadrivalent human papillomavirus (HPV) vaccine had fewer and fewer infections with high-risk HPV strains covered by the vaccine year after year, but the incidence of high-risk strains that were not covered by the vaccine increased over the same 12-year period, researchers report in a study published August 23 in JAMA Open Network.
“One of the unique contributions that this study provides is the evaluation of a real-world example of the HPV infection rates following immunization in a population of adolescent girls and young adult women at a single health center in a large U.S. city, reflecting strong evidence of vaccine effectiveness,” write Nicolas F. Schlecht, PhD, a professor of oncology at Roswell Park Comprehensive Cancer Center, Buffalo, and his colleagues. “Previous surveillance studies from the U.S. have involved older women and populations with relatively low vaccine coverage.”
In addition to supporting the value of continuing to vaccinate teens against HPV, the findings underscore the importance of continuing to screen women for cervical cancer, Dr. Schlecht said in an interview.
“HPV has not and is not going away,” he said. “We need to keep on our toes with screening and other measures to continue to prevent the development of cervix cancer,” including monitoring different high-risk HPV types and keeping a close eye on cervical precancer rates, particularly CIN3 and cervix cancer, he said. “The vaccines are definitely a good thing. Just getting rid of HPV16 is an amazing accomplishment.”
Kevin Ault, MD, a professor of ob/gyn and academic specialist director of clinical and translational research at the University of Kansas, Kansas City, told this news organization that other studies have had similar findings, but this one is larger with longer follow-up.
“The take-home message is that vaccines work, and this is especially true for the HPV vaccine,” said Dr. Ault, who was not involved in the research. “The vaccine prevents HPV infections and the consequences of these infections, such as cervical cancer. The results are consistent with other studies in different settings, so they are likely generalizable.”
The researchers collected data from October 2007, shortly after the vaccine was approved, through September 2019 on sexually active adolescent and young women aged 13 to 21 years who had received the HPV vaccine and had agreed to follow-up assessments every 6 months until they turned 26. Each follow-up included the collecting of samples of cervical and anal cells for polymerase chain reaction testing for the presence of HPV types.
More than half of the 1,453 participants were Hispanic (58.8%), and half were Black (50.4%), including 15% Hispanic and Black patients. The average age of the participants was 18 years. They were tracked for a median 2.4 years. Nearly half the participants (48%) received the HPV vaccine prior to sexual debut.
For the longitudinal study, the researchers adjusted for participants’ age, the year they received the vaccine, and the years since they were vaccinated. They also tracked breakthrough infections for the four types of HPV covered by the vaccine in participants who received the vaccine before sexual debut.
“We evaluated whether infection rates for HPV have changed since the administration of the vaccine by assessing longitudinally the probability of HPV detection over time among vaccinated participants while adjusting for changes in cohort characteristics over time,” the researchers write. In their statistical analysis, they made adjustments for the number of vaccine doses participants received before their first study visit, age at sexual debut, age at first vaccine dose, number of sexual partners in the preceding 6 months, consistency of condom use during sex, history of a positive chlamydia test, and, for anal HPV analyses, whether the participants had had anal sex in the previous 6 months.
The average age at first intercourse remained steady at 15 years throughout the study, but the average age of vaccination dropped from 18 years in 2008 to 12 years in 2019 (P < .001). More than half the participants (64%) had had at least three lifetime sexual partners at baseline.
After adjustment for age, the researchers found that the incidence of the four HPV types covered by the vaccine – HPV-6, HPV-11, HPV-16, and HPV-18 – dropped more each year, shifting from 9.1% from 2008-2010 to 4.7% from 2017-2019. The effect was even greater among those vaccinated prior to sexual debut; for those patients, the incidence of the four vaccine types dropped from 8.8% to 1.7% over the course of the study. Declines over time also occurred for anal types HPV-31 (adjusted odds ratio [aOR] = 0.76) and HPV-45 (aOR = 0.77). Those vaccinated prior to any sexual intercourse had 19% lower odds of infection per year with a vaccine-covered HPV type.
“We were really excited to see that the types targeted by the vaccines were considerably lower over time in our population,” Dr. Schlecht told this news organization. “This is an important observation, since most of these types are the most worrisome for cervical cancer.”
They were surprised, however, to see overall HPV prevalence increase over time, particularly with the high-risk HPV types that were not covered by the quadrivalent vaccine.
Prevalence of cervical high-risk types not in the vaccine increased from 25.1% from 2008-2010 to 30.5% from 2017-2019. Odds of detection of high-risk HPV types not covered by the vaccine increased 8% each year, particularly for HPV-56 and HPV-68; anal HPV types increased 11% each year. Neither age nor recent number of sexual partners affected the findings.
“The underlying mechanisms for the observed increased detection of specific non-vaccine HPV types over time are not yet clear.”
“We hope this doesn’t translate into some increase in cervical neoplasia that is unanticipated,” Dr. Schlecht said. He noted that the attributable risks for cancer associated with nonvaccine high-risk HPV types remain low. “Theoretical concerns are one thing; actual data is what drives the show,” he said.
The research was funded by the National Institutes of Health and the Icahn School of Medicine at Mount Sinai, New York. Dr. Schlecht has served on advisory boards for Merck, GlaxoSmithKline (GSK), and PDS Biotechnology. One author previously served on a GSK advisory board, and another worked with Merck on an early vaccine trial. Dr. Ault has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Young women who received the quadrivalent human papillomavirus (HPV) vaccine had fewer and fewer infections with high-risk HPV strains covered by the vaccine year after year, but the incidence of high-risk strains that were not covered by the vaccine increased over the same 12-year period, researchers report in a study published August 23 in JAMA Open Network.
“One of the unique contributions that this study provides is the evaluation of a real-world example of the HPV infection rates following immunization in a population of adolescent girls and young adult women at a single health center in a large U.S. city, reflecting strong evidence of vaccine effectiveness,” write Nicolas F. Schlecht, PhD, a professor of oncology at Roswell Park Comprehensive Cancer Center, Buffalo, and his colleagues. “Previous surveillance studies from the U.S. have involved older women and populations with relatively low vaccine coverage.”
In addition to supporting the value of continuing to vaccinate teens against HPV, the findings underscore the importance of continuing to screen women for cervical cancer, Dr. Schlecht said in an interview.
“HPV has not and is not going away,” he said. “We need to keep on our toes with screening and other measures to continue to prevent the development of cervix cancer,” including monitoring different high-risk HPV types and keeping a close eye on cervical precancer rates, particularly CIN3 and cervix cancer, he said. “The vaccines are definitely a good thing. Just getting rid of HPV16 is an amazing accomplishment.”
Kevin Ault, MD, a professor of ob/gyn and academic specialist director of clinical and translational research at the University of Kansas, Kansas City, told this news organization that other studies have had similar findings, but this one is larger with longer follow-up.
“The take-home message is that vaccines work, and this is especially true for the HPV vaccine,” said Dr. Ault, who was not involved in the research. “The vaccine prevents HPV infections and the consequences of these infections, such as cervical cancer. The results are consistent with other studies in different settings, so they are likely generalizable.”
The researchers collected data from October 2007, shortly after the vaccine was approved, through September 2019 on sexually active adolescent and young women aged 13 to 21 years who had received the HPV vaccine and had agreed to follow-up assessments every 6 months until they turned 26. Each follow-up included the collecting of samples of cervical and anal cells for polymerase chain reaction testing for the presence of HPV types.
More than half of the 1,453 participants were Hispanic (58.8%), and half were Black (50.4%), including 15% Hispanic and Black patients. The average age of the participants was 18 years. They were tracked for a median 2.4 years. Nearly half the participants (48%) received the HPV vaccine prior to sexual debut.
For the longitudinal study, the researchers adjusted for participants’ age, the year they received the vaccine, and the years since they were vaccinated. They also tracked breakthrough infections for the four types of HPV covered by the vaccine in participants who received the vaccine before sexual debut.
“We evaluated whether infection rates for HPV have changed since the administration of the vaccine by assessing longitudinally the probability of HPV detection over time among vaccinated participants while adjusting for changes in cohort characteristics over time,” the researchers write. In their statistical analysis, they made adjustments for the number of vaccine doses participants received before their first study visit, age at sexual debut, age at first vaccine dose, number of sexual partners in the preceding 6 months, consistency of condom use during sex, history of a positive chlamydia test, and, for anal HPV analyses, whether the participants had had anal sex in the previous 6 months.
The average age at first intercourse remained steady at 15 years throughout the study, but the average age of vaccination dropped from 18 years in 2008 to 12 years in 2019 (P < .001). More than half the participants (64%) had had at least three lifetime sexual partners at baseline.
After adjustment for age, the researchers found that the incidence of the four HPV types covered by the vaccine – HPV-6, HPV-11, HPV-16, and HPV-18 – dropped more each year, shifting from 9.1% from 2008-2010 to 4.7% from 2017-2019. The effect was even greater among those vaccinated prior to sexual debut; for those patients, the incidence of the four vaccine types dropped from 8.8% to 1.7% over the course of the study. Declines over time also occurred for anal types HPV-31 (adjusted odds ratio [aOR] = 0.76) and HPV-45 (aOR = 0.77). Those vaccinated prior to any sexual intercourse had 19% lower odds of infection per year with a vaccine-covered HPV type.
“We were really excited to see that the types targeted by the vaccines were considerably lower over time in our population,” Dr. Schlecht told this news organization. “This is an important observation, since most of these types are the most worrisome for cervical cancer.”
They were surprised, however, to see overall HPV prevalence increase over time, particularly with the high-risk HPV types that were not covered by the quadrivalent vaccine.
Prevalence of cervical high-risk types not in the vaccine increased from 25.1% from 2008-2010 to 30.5% from 2017-2019. Odds of detection of high-risk HPV types not covered by the vaccine increased 8% each year, particularly for HPV-56 and HPV-68; anal HPV types increased 11% each year. Neither age nor recent number of sexual partners affected the findings.
“The underlying mechanisms for the observed increased detection of specific non-vaccine HPV types over time are not yet clear.”
“We hope this doesn’t translate into some increase in cervical neoplasia that is unanticipated,” Dr. Schlecht said. He noted that the attributable risks for cancer associated with nonvaccine high-risk HPV types remain low. “Theoretical concerns are one thing; actual data is what drives the show,” he said.
The research was funded by the National Institutes of Health and the Icahn School of Medicine at Mount Sinai, New York. Dr. Schlecht has served on advisory boards for Merck, GlaxoSmithKline (GSK), and PDS Biotechnology. One author previously served on a GSK advisory board, and another worked with Merck on an early vaccine trial. Dr. Ault has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Young women who received the quadrivalent human papillomavirus (HPV) vaccine had fewer and fewer infections with high-risk HPV strains covered by the vaccine year after year, but the incidence of high-risk strains that were not covered by the vaccine increased over the same 12-year period, researchers report in a study published August 23 in JAMA Open Network.
“One of the unique contributions that this study provides is the evaluation of a real-world example of the HPV infection rates following immunization in a population of adolescent girls and young adult women at a single health center in a large U.S. city, reflecting strong evidence of vaccine effectiveness,” write Nicolas F. Schlecht, PhD, a professor of oncology at Roswell Park Comprehensive Cancer Center, Buffalo, and his colleagues. “Previous surveillance studies from the U.S. have involved older women and populations with relatively low vaccine coverage.”
In addition to supporting the value of continuing to vaccinate teens against HPV, the findings underscore the importance of continuing to screen women for cervical cancer, Dr. Schlecht said in an interview.
“HPV has not and is not going away,” he said. “We need to keep on our toes with screening and other measures to continue to prevent the development of cervix cancer,” including monitoring different high-risk HPV types and keeping a close eye on cervical precancer rates, particularly CIN3 and cervix cancer, he said. “The vaccines are definitely a good thing. Just getting rid of HPV16 is an amazing accomplishment.”
Kevin Ault, MD, a professor of ob/gyn and academic specialist director of clinical and translational research at the University of Kansas, Kansas City, told this news organization that other studies have had similar findings, but this one is larger with longer follow-up.
“The take-home message is that vaccines work, and this is especially true for the HPV vaccine,” said Dr. Ault, who was not involved in the research. “The vaccine prevents HPV infections and the consequences of these infections, such as cervical cancer. The results are consistent with other studies in different settings, so they are likely generalizable.”
The researchers collected data from October 2007, shortly after the vaccine was approved, through September 2019 on sexually active adolescent and young women aged 13 to 21 years who had received the HPV vaccine and had agreed to follow-up assessments every 6 months until they turned 26. Each follow-up included the collecting of samples of cervical and anal cells for polymerase chain reaction testing for the presence of HPV types.
More than half of the 1,453 participants were Hispanic (58.8%), and half were Black (50.4%), including 15% Hispanic and Black patients. The average age of the participants was 18 years. They were tracked for a median 2.4 years. Nearly half the participants (48%) received the HPV vaccine prior to sexual debut.
For the longitudinal study, the researchers adjusted for participants’ age, the year they received the vaccine, and the years since they were vaccinated. They also tracked breakthrough infections for the four types of HPV covered by the vaccine in participants who received the vaccine before sexual debut.
“We evaluated whether infection rates for HPV have changed since the administration of the vaccine by assessing longitudinally the probability of HPV detection over time among vaccinated participants while adjusting for changes in cohort characteristics over time,” the researchers write. In their statistical analysis, they made adjustments for the number of vaccine doses participants received before their first study visit, age at sexual debut, age at first vaccine dose, number of sexual partners in the preceding 6 months, consistency of condom use during sex, history of a positive chlamydia test, and, for anal HPV analyses, whether the participants had had anal sex in the previous 6 months.
The average age at first intercourse remained steady at 15 years throughout the study, but the average age of vaccination dropped from 18 years in 2008 to 12 years in 2019 (P < .001). More than half the participants (64%) had had at least three lifetime sexual partners at baseline.
After adjustment for age, the researchers found that the incidence of the four HPV types covered by the vaccine – HPV-6, HPV-11, HPV-16, and HPV-18 – dropped more each year, shifting from 9.1% from 2008-2010 to 4.7% from 2017-2019. The effect was even greater among those vaccinated prior to sexual debut; for those patients, the incidence of the four vaccine types dropped from 8.8% to 1.7% over the course of the study. Declines over time also occurred for anal types HPV-31 (adjusted odds ratio [aOR] = 0.76) and HPV-45 (aOR = 0.77). Those vaccinated prior to any sexual intercourse had 19% lower odds of infection per year with a vaccine-covered HPV type.
“We were really excited to see that the types targeted by the vaccines were considerably lower over time in our population,” Dr. Schlecht told this news organization. “This is an important observation, since most of these types are the most worrisome for cervical cancer.”
They were surprised, however, to see overall HPV prevalence increase over time, particularly with the high-risk HPV types that were not covered by the quadrivalent vaccine.
Prevalence of cervical high-risk types not in the vaccine increased from 25.1% from 2008-2010 to 30.5% from 2017-2019. Odds of detection of high-risk HPV types not covered by the vaccine increased 8% each year, particularly for HPV-56 and HPV-68; anal HPV types increased 11% each year. Neither age nor recent number of sexual partners affected the findings.
“The underlying mechanisms for the observed increased detection of specific non-vaccine HPV types over time are not yet clear.”
“We hope this doesn’t translate into some increase in cervical neoplasia that is unanticipated,” Dr. Schlecht said. He noted that the attributable risks for cancer associated with nonvaccine high-risk HPV types remain low. “Theoretical concerns are one thing; actual data is what drives the show,” he said.
The research was funded by the National Institutes of Health and the Icahn School of Medicine at Mount Sinai, New York. Dr. Schlecht has served on advisory boards for Merck, GlaxoSmithKline (GSK), and PDS Biotechnology. One author previously served on a GSK advisory board, and another worked with Merck on an early vaccine trial. Dr. Ault has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Health care workers eager for COVID booster shots
As COVID vaccine boosters move closer to reality, most physicians and nurses are ready and willing to get another shot in the arm, according to a new Medscape survey.
Altogether, 93% of physicians and 87% of nurses/advanced practice nurses (APNs) said they wanted to get a booster, although the timing of when they wanted the shots differed somewhat between the two groups surveyed Aug. 4-15.
Among the 732 physicians polled, 50% wanted to get their shot immediately, compared with 38% of the 1,193 nurses/APNs who responded, while 44% of physicians and 50% of nurses/APNs said that they would wait until the vaccine booster was authorized and recommended.
At this point in time, almost all of the health care workers surveyed – 98% of physicians and 94% of nurses/APNs – have been fully vaccinated against COVID-19. A small proportion of each group, however, received the Johnson & Johnson vaccine (1% of physicians and 3% of nurses) and are not included in the current plan for booster shots.
The Medscape survey sample did include one group that is already eligible for a third dose: About 20% of physicians and 26% of nurses/ANPs said they have a condition or take a medication that compromises their immune system.
Respondents’ experiences with patient requests for boosters suggest a somewhat lower level of interest. About two-thirds of the health care workers (69% of physicians and 63% of nurses) said that patients frequently or sometimes asked about COVID boosters, compared with 13% (physicians) and 19% (nurses) who said their patients had never asked.
Interest lower among general population
In a separate survey conducted by WebMD, 82% of those who have been at least partially vaccinated said they want to get a COVID vaccine booster (14% immediately and 68% after authorization and recommendation). Of the remaining vaccinees, 7% said they do not want to get a booster and 11% were unsure.
The full sample of 592 respondents surveyed Aug. 5-10, however, included 19% who do not plan to get vaccinated and 6% who are planning to be vaccinated but have not yet done so.
The proportion of immunocompromised individuals in the two survey groups was similar, with about 25% of those in the WebMD survey reporting they have a condition or take a medication that compromises their immune system. Those respondents were more than twice as likely to want to get a booster immediately, compared to those with an uncompromised immune system (24% vs. 11%).
The distribution of vaccines received by brand was also comparable between the two groups surveyed. Of health care workers and readers, over half of each group received the Pfizer/BioNTech vaccine (59% vs. 54%), followed by Moderna (38% vs. 40%) and Johnson & Johnson (3% vs. 5%).
A version of this article first appeared on Medscape.com.
As COVID vaccine boosters move closer to reality, most physicians and nurses are ready and willing to get another shot in the arm, according to a new Medscape survey.
Altogether, 93% of physicians and 87% of nurses/advanced practice nurses (APNs) said they wanted to get a booster, although the timing of when they wanted the shots differed somewhat between the two groups surveyed Aug. 4-15.
Among the 732 physicians polled, 50% wanted to get their shot immediately, compared with 38% of the 1,193 nurses/APNs who responded, while 44% of physicians and 50% of nurses/APNs said that they would wait until the vaccine booster was authorized and recommended.
At this point in time, almost all of the health care workers surveyed – 98% of physicians and 94% of nurses/APNs – have been fully vaccinated against COVID-19. A small proportion of each group, however, received the Johnson & Johnson vaccine (1% of physicians and 3% of nurses) and are not included in the current plan for booster shots.
The Medscape survey sample did include one group that is already eligible for a third dose: About 20% of physicians and 26% of nurses/ANPs said they have a condition or take a medication that compromises their immune system.
Respondents’ experiences with patient requests for boosters suggest a somewhat lower level of interest. About two-thirds of the health care workers (69% of physicians and 63% of nurses) said that patients frequently or sometimes asked about COVID boosters, compared with 13% (physicians) and 19% (nurses) who said their patients had never asked.
Interest lower among general population
In a separate survey conducted by WebMD, 82% of those who have been at least partially vaccinated said they want to get a COVID vaccine booster (14% immediately and 68% after authorization and recommendation). Of the remaining vaccinees, 7% said they do not want to get a booster and 11% were unsure.
The full sample of 592 respondents surveyed Aug. 5-10, however, included 19% who do not plan to get vaccinated and 6% who are planning to be vaccinated but have not yet done so.
The proportion of immunocompromised individuals in the two survey groups was similar, with about 25% of those in the WebMD survey reporting they have a condition or take a medication that compromises their immune system. Those respondents were more than twice as likely to want to get a booster immediately, compared to those with an uncompromised immune system (24% vs. 11%).
The distribution of vaccines received by brand was also comparable between the two groups surveyed. Of health care workers and readers, over half of each group received the Pfizer/BioNTech vaccine (59% vs. 54%), followed by Moderna (38% vs. 40%) and Johnson & Johnson (3% vs. 5%).
A version of this article first appeared on Medscape.com.
As COVID vaccine boosters move closer to reality, most physicians and nurses are ready and willing to get another shot in the arm, according to a new Medscape survey.
Altogether, 93% of physicians and 87% of nurses/advanced practice nurses (APNs) said they wanted to get a booster, although the timing of when they wanted the shots differed somewhat between the two groups surveyed Aug. 4-15.
Among the 732 physicians polled, 50% wanted to get their shot immediately, compared with 38% of the 1,193 nurses/APNs who responded, while 44% of physicians and 50% of nurses/APNs said that they would wait until the vaccine booster was authorized and recommended.
At this point in time, almost all of the health care workers surveyed – 98% of physicians and 94% of nurses/APNs – have been fully vaccinated against COVID-19. A small proportion of each group, however, received the Johnson & Johnson vaccine (1% of physicians and 3% of nurses) and are not included in the current plan for booster shots.
The Medscape survey sample did include one group that is already eligible for a third dose: About 20% of physicians and 26% of nurses/ANPs said they have a condition or take a medication that compromises their immune system.
Respondents’ experiences with patient requests for boosters suggest a somewhat lower level of interest. About two-thirds of the health care workers (69% of physicians and 63% of nurses) said that patients frequently or sometimes asked about COVID boosters, compared with 13% (physicians) and 19% (nurses) who said their patients had never asked.
Interest lower among general population
In a separate survey conducted by WebMD, 82% of those who have been at least partially vaccinated said they want to get a COVID vaccine booster (14% immediately and 68% after authorization and recommendation). Of the remaining vaccinees, 7% said they do not want to get a booster and 11% were unsure.
The full sample of 592 respondents surveyed Aug. 5-10, however, included 19% who do not plan to get vaccinated and 6% who are planning to be vaccinated but have not yet done so.
The proportion of immunocompromised individuals in the two survey groups was similar, with about 25% of those in the WebMD survey reporting they have a condition or take a medication that compromises their immune system. Those respondents were more than twice as likely to want to get a booster immediately, compared to those with an uncompromised immune system (24% vs. 11%).
The distribution of vaccines received by brand was also comparable between the two groups surveyed. Of health care workers and readers, over half of each group received the Pfizer/BioNTech vaccine (59% vs. 54%), followed by Moderna (38% vs. 40%) and Johnson & Johnson (3% vs. 5%).
A version of this article first appeared on Medscape.com.
Guidance on additional COVID-19 vaccine dose for MS patients
Patients aged 12 years and older with multiple sclerosis (MS) who are fully immunized against COVID-19 with either the Pfizer-BioNTech or Moderna mRNA vaccine may be eligible to receive an additional dose now, the National Multiple Sclerosis Society has announced.
New guidance, which is “based on available data from studies and expert consensus opinion” by a panel of MS neurologists and experts, was published Aug. 19 on the organization’s website.
The Food and Drug Administration has authorized an additional dose of the coronavirus vaccine for patients who are expected to not have a normal or adequate immune response to the first two doses. Patients with MS who use certain treatments have a reduced or absent antibody response to the vaccine, according to recent data.
“We want people living with MS to be aware of this additional dose and discuss when they need an additional dose or booster dose with their health care provider,” Julie Fiol, RN, MSW, associate vice president of health care access, National MS Society, said in an interview.
Those who may benefit from an additional dose include patients with MS who use sphingosine 1-phosphate receptor modulators, anti-CD20 monoclonal antibodies, or alemtuzumab (Lemtrada), the National MS Society noted. These particular disease modifying therapies (DMTs) have a stronger effect on the immune system than do other treatments.
Protecting ‘the most vulnerable’
Sphingosine 1-phosphate receptor modulators include fingolimod (Gilenya), siponimod (Mayzent), ozanimod (Zeposia), and ponesimod (Ponvory).
Anti-CD20 monoclonal antibodies include ocrelizumab (Ocrevus), ofatumumab (Kesimpta), rituximab (Rituxan), and corresponding biosimilars.
Current data do not support an additional dose for immunocompromised patients who received the Johnson & Johnson vaccine. The FDA and the Centers for Disease Control and Prevention are developing recommendations for these patients, and the National MS Society will update its guidance as needed, the organization noted in its statement.
“Like other medical decisions, the decision to get an additional dose is best made in partnership with your health care provider,” said Ms. Fiol. “Talk to your MS health care provider to determine what is best for you.”
MS itself does not compromise the immune system, but some MS therapies alter the immune system and reduce the body’s response to vaccination. Patients with MS who use B cell-depleting therapies have a better antibody response when they receive the vaccine 3 months or more after the last dose of MS therapy, according to the National MS Society.
Data suggest that patients with MS are not more susceptible to COVID-19 infection, severe illness, or death than are patients without MS. However, certain groups of patients with MS, such as those who receive B cell-depleting treatments, are more susceptible to having a severe case of COVID-19.
That said, “everyone will need a booster at some point. Those who take DMTs that have greater impact on the immune system are the most urgent need now,” the organization noted.
“Vaccination against COVID-19 is critical for public safety and, especially, the safety of the most vulnerable among us,” said Ms. Fiol. “We encourage everyone with MS get vaccinated.”
A version of this article first appeared on Medscape.com.
Patients aged 12 years and older with multiple sclerosis (MS) who are fully immunized against COVID-19 with either the Pfizer-BioNTech or Moderna mRNA vaccine may be eligible to receive an additional dose now, the National Multiple Sclerosis Society has announced.
New guidance, which is “based on available data from studies and expert consensus opinion” by a panel of MS neurologists and experts, was published Aug. 19 on the organization’s website.
The Food and Drug Administration has authorized an additional dose of the coronavirus vaccine for patients who are expected to not have a normal or adequate immune response to the first two doses. Patients with MS who use certain treatments have a reduced or absent antibody response to the vaccine, according to recent data.
“We want people living with MS to be aware of this additional dose and discuss when they need an additional dose or booster dose with their health care provider,” Julie Fiol, RN, MSW, associate vice president of health care access, National MS Society, said in an interview.
Those who may benefit from an additional dose include patients with MS who use sphingosine 1-phosphate receptor modulators, anti-CD20 monoclonal antibodies, or alemtuzumab (Lemtrada), the National MS Society noted. These particular disease modifying therapies (DMTs) have a stronger effect on the immune system than do other treatments.
Protecting ‘the most vulnerable’
Sphingosine 1-phosphate receptor modulators include fingolimod (Gilenya), siponimod (Mayzent), ozanimod (Zeposia), and ponesimod (Ponvory).
Anti-CD20 monoclonal antibodies include ocrelizumab (Ocrevus), ofatumumab (Kesimpta), rituximab (Rituxan), and corresponding biosimilars.
Current data do not support an additional dose for immunocompromised patients who received the Johnson & Johnson vaccine. The FDA and the Centers for Disease Control and Prevention are developing recommendations for these patients, and the National MS Society will update its guidance as needed, the organization noted in its statement.
“Like other medical decisions, the decision to get an additional dose is best made in partnership with your health care provider,” said Ms. Fiol. “Talk to your MS health care provider to determine what is best for you.”
MS itself does not compromise the immune system, but some MS therapies alter the immune system and reduce the body’s response to vaccination. Patients with MS who use B cell-depleting therapies have a better antibody response when they receive the vaccine 3 months or more after the last dose of MS therapy, according to the National MS Society.
Data suggest that patients with MS are not more susceptible to COVID-19 infection, severe illness, or death than are patients without MS. However, certain groups of patients with MS, such as those who receive B cell-depleting treatments, are more susceptible to having a severe case of COVID-19.
That said, “everyone will need a booster at some point. Those who take DMTs that have greater impact on the immune system are the most urgent need now,” the organization noted.
“Vaccination against COVID-19 is critical for public safety and, especially, the safety of the most vulnerable among us,” said Ms. Fiol. “We encourage everyone with MS get vaccinated.”
A version of this article first appeared on Medscape.com.
Patients aged 12 years and older with multiple sclerosis (MS) who are fully immunized against COVID-19 with either the Pfizer-BioNTech or Moderna mRNA vaccine may be eligible to receive an additional dose now, the National Multiple Sclerosis Society has announced.
New guidance, which is “based on available data from studies and expert consensus opinion” by a panel of MS neurologists and experts, was published Aug. 19 on the organization’s website.
The Food and Drug Administration has authorized an additional dose of the coronavirus vaccine for patients who are expected to not have a normal or adequate immune response to the first two doses. Patients with MS who use certain treatments have a reduced or absent antibody response to the vaccine, according to recent data.
“We want people living with MS to be aware of this additional dose and discuss when they need an additional dose or booster dose with their health care provider,” Julie Fiol, RN, MSW, associate vice president of health care access, National MS Society, said in an interview.
Those who may benefit from an additional dose include patients with MS who use sphingosine 1-phosphate receptor modulators, anti-CD20 monoclonal antibodies, or alemtuzumab (Lemtrada), the National MS Society noted. These particular disease modifying therapies (DMTs) have a stronger effect on the immune system than do other treatments.
Protecting ‘the most vulnerable’
Sphingosine 1-phosphate receptor modulators include fingolimod (Gilenya), siponimod (Mayzent), ozanimod (Zeposia), and ponesimod (Ponvory).
Anti-CD20 monoclonal antibodies include ocrelizumab (Ocrevus), ofatumumab (Kesimpta), rituximab (Rituxan), and corresponding biosimilars.
Current data do not support an additional dose for immunocompromised patients who received the Johnson & Johnson vaccine. The FDA and the Centers for Disease Control and Prevention are developing recommendations for these patients, and the National MS Society will update its guidance as needed, the organization noted in its statement.
“Like other medical decisions, the decision to get an additional dose is best made in partnership with your health care provider,” said Ms. Fiol. “Talk to your MS health care provider to determine what is best for you.”
MS itself does not compromise the immune system, but some MS therapies alter the immune system and reduce the body’s response to vaccination. Patients with MS who use B cell-depleting therapies have a better antibody response when they receive the vaccine 3 months or more after the last dose of MS therapy, according to the National MS Society.
Data suggest that patients with MS are not more susceptible to COVID-19 infection, severe illness, or death than are patients without MS. However, certain groups of patients with MS, such as those who receive B cell-depleting treatments, are more susceptible to having a severe case of COVID-19.
That said, “everyone will need a booster at some point. Those who take DMTs that have greater impact on the immune system are the most urgent need now,” the organization noted.
“Vaccination against COVID-19 is critical for public safety and, especially, the safety of the most vulnerable among us,” said Ms. Fiol. “We encourage everyone with MS get vaccinated.”
A version of this article first appeared on Medscape.com.
Q&A: Get flu shot early this year? Same time as COVID vaccine?
With first-time COVID-19 immunizations continuing and the plan to offer booster vaccines to most Americans starting next month, what are the considerations for getting COVID-19 and flu shots at the same time?
This news organization asked Andrew T. Pavia, MD, for his advice. He is the George and Esther Gross Presidential Professor and chief of the division of pediatric infectious diseases at the University of Utah, Salt Lake City, and a fellow of the Infectious Diseases Society of America.
Q: With COVID-19 cases surging, is it a good idea to get the flu shot early this season?
Dr. Pavia: I don’t think there is a rush to do it in August, but it is a good idea to get a flu shot this season. The consequences of getting the flu while COVID is circulating are serious.
Q: What are the implications?
There are some we know and some we don’t know. If you develop flu-like symptoms, you’re going to have to get tested. You’re going to have to stay home quite a bit longer if you get a definitive (positive COVID-19) test than you would simply with flu symptoms. Also, you’re probably going to miss work when your workplace is very stressed or your children are stressed by having COVID circulating in schools.
The part we know less about are the implications of getting the flu and COVID together. There is some reason to believe if you get them together, the illness will be more severe. We are seeing that with RSV (respiratory syncytial virus) and parainfluenza and COVID coinfections in children. They appear to be quite severe.
But for flu, we just don’t have the data yet. That’s because there really was no cocirculation of COVID and influenza with the exception of parts of China for a brief part of February and March.
Q: Will the planned administration of booster COVID-19 shots this fall affect the number of people who get the flu vaccine or how it’s distributed?
It creates a lot of logistical challenges, particularly for hospitals and other places that need to vaccinate a large number of their employees for flu and that will need to give COVID boosters at about the same time period. It also creates logistical challenges for doctors’ offices.
But we don’t know of any reason why you can’t give the two shots together.
Q: Is it possible flu season will be more severe because we isolated and wore masks, etc., last winter? Any science behind that?
The more you study flu, the less you can predict, and I’ve been studying flu for a long time. There are reasons that might suggest a severe flu season – there has been limited immunity, and some people are not wearing masks effectively and they are gathering again. Those are things we believe protected us from influenza last season.
But we have not seen flu emerge yet. Normally we look to Australia, New Zealand, and South Africa during their winter – which is our summer – to get some idea of what is over the horizon for the Northern Hemisphere. Flu activity in Australia has been very modest this year.
That might mean flu may not show up for a while, but I would be loathe to make a prediction.
Q: What are the chances we’ll see a flu outbreak like we’re seeing with RSV, which is normally a winter illness?
The fact that we had a summer RSV surge just gives you an idea of how the normal epidemiology of viral infections has been disrupted. It means anything could happen with influenza. It could show up late summer or fall or wait until next spring.
We really don’t understand how those interactions work. When a new flu strain emerges, it often ignores the traditional behavior and shows up in the spring or fall. It happened in the 2009 pandemic, it happened in 1918.
The one thing I would safely predict about the next flu wave is that it will surprise us.
Q: Are you hopeful that combination vaccines in development from a number of companies, such as Moderna, Novavax, and Vivaldi, will be effective?
It is beginning to look like COVID will be with us for the foreseeable future – maybe as a seasonal virus or maybe as an ongoing pandemic. We are going to need to protect (ourselves) simultaneously against the flu and COVID. A single shot is a great way to do that – nobody wants two needles; nobody wants two trips to get vaccinated.
An effective combination vaccine would be a really great tool.
We have to wait to see what the science shows us, because they are quite different viruses. We won’t know if a combination vaccine works well and has acceptable side effects until we do those studies.
Q. Do you know at this point whether the side effects from two vaccines would be additive? Is there any way to predict that?
There is no way to predict. There are so many things that go into whether someone has side effects that we don’t understand. With fairly reactogenic vaccines like the mRNA vaccines, lots of people have no side effects whatsoever and others are really uncomfortable for 24 hours.
Flu is generally a better tolerated vaccine. There are still people who get muscle aches and very sore arms. I don’t think we can predict if getting two will be additive or just the same as getting one vaccine.
Q: Other than convenience and the benefit for people who are needle-phobic, are there any other advantages of combining them into one shot?
The logistics alone are enough to justify having one effective product if we can make one. It should reduce the overall cost of administration and reduce time off from work.
The combination vaccines given by pediatricians have been very successful. They reduce the number of needles for kids and make it much easier for parents and the pediatricians administering them. The same principle should apply to adults, who sometimes are less brave about needles than kids are.
Historically, combined vaccines in general have worked as well as vaccines given alone, but there have been exceptions. We just have to see what the products look like.
Q: For now, the flu vaccine and COVID-19 vaccine are single products. If you get them separately, is it better to put some time between the two?
We don’t know. There are studies that probably won’t be out in time to decide in September. They are looking at whether you get an equivalent immune response if you give them together or apart.
For now, I would say the advantage of getting them together is if you do get side effects, you’ll only get them once – one day to suffer through them. Also, it’s one trip to the doctor.
The potential advantage of separating them is that is how we developed and tested the vaccines. If you do react to them, side effects could be milder, but it will be on two separate days.
I would recommend doing whatever works so that you get both vaccines in a timely manner.
I’m going to get my flu shot as soon as it’s available. If I’m due for a COVID booster at that time, I would probably do them together.
Q: Do you foresee a point in the future when the predominant strain of SARS-CoV-2 will be one of the components of a flu vaccine, like we did in the past with H1N1, etc?
It really remains to be seen, but it is very conceivable it could happen. The same companies that developed COVID-19 vaccines are working on flu vaccines.
Q: Any other advice for people concerned about getting immunized against both COVID-19 and influenza in the coming months?
There is no side effect of the vaccine that begins to approach the risk you face from either disease. It’s really one of the best things you can do to protect yourself is to get vaccinated.
In the case of flu, the vaccine is only modestly effective, but it still saves tens of thousands of lives each year. The SARS-CoV-2 vaccine is a much better vaccine and a deadlier disease.
Dr. Pavia consulted for GlaxoSmithKline on influenza testing.
A version of this article first appeared on Medscape.com.
With first-time COVID-19 immunizations continuing and the plan to offer booster vaccines to most Americans starting next month, what are the considerations for getting COVID-19 and flu shots at the same time?
This news organization asked Andrew T. Pavia, MD, for his advice. He is the George and Esther Gross Presidential Professor and chief of the division of pediatric infectious diseases at the University of Utah, Salt Lake City, and a fellow of the Infectious Diseases Society of America.
Q: With COVID-19 cases surging, is it a good idea to get the flu shot early this season?
Dr. Pavia: I don’t think there is a rush to do it in August, but it is a good idea to get a flu shot this season. The consequences of getting the flu while COVID is circulating are serious.
Q: What are the implications?
There are some we know and some we don’t know. If you develop flu-like symptoms, you’re going to have to get tested. You’re going to have to stay home quite a bit longer if you get a definitive (positive COVID-19) test than you would simply with flu symptoms. Also, you’re probably going to miss work when your workplace is very stressed or your children are stressed by having COVID circulating in schools.
The part we know less about are the implications of getting the flu and COVID together. There is some reason to believe if you get them together, the illness will be more severe. We are seeing that with RSV (respiratory syncytial virus) and parainfluenza and COVID coinfections in children. They appear to be quite severe.
But for flu, we just don’t have the data yet. That’s because there really was no cocirculation of COVID and influenza with the exception of parts of China for a brief part of February and March.
Q: Will the planned administration of booster COVID-19 shots this fall affect the number of people who get the flu vaccine or how it’s distributed?
It creates a lot of logistical challenges, particularly for hospitals and other places that need to vaccinate a large number of their employees for flu and that will need to give COVID boosters at about the same time period. It also creates logistical challenges for doctors’ offices.
But we don’t know of any reason why you can’t give the two shots together.
Q: Is it possible flu season will be more severe because we isolated and wore masks, etc., last winter? Any science behind that?
The more you study flu, the less you can predict, and I’ve been studying flu for a long time. There are reasons that might suggest a severe flu season – there has been limited immunity, and some people are not wearing masks effectively and they are gathering again. Those are things we believe protected us from influenza last season.
But we have not seen flu emerge yet. Normally we look to Australia, New Zealand, and South Africa during their winter – which is our summer – to get some idea of what is over the horizon for the Northern Hemisphere. Flu activity in Australia has been very modest this year.
That might mean flu may not show up for a while, but I would be loathe to make a prediction.
Q: What are the chances we’ll see a flu outbreak like we’re seeing with RSV, which is normally a winter illness?
The fact that we had a summer RSV surge just gives you an idea of how the normal epidemiology of viral infections has been disrupted. It means anything could happen with influenza. It could show up late summer or fall or wait until next spring.
We really don’t understand how those interactions work. When a new flu strain emerges, it often ignores the traditional behavior and shows up in the spring or fall. It happened in the 2009 pandemic, it happened in 1918.
The one thing I would safely predict about the next flu wave is that it will surprise us.
Q: Are you hopeful that combination vaccines in development from a number of companies, such as Moderna, Novavax, and Vivaldi, will be effective?
It is beginning to look like COVID will be with us for the foreseeable future – maybe as a seasonal virus or maybe as an ongoing pandemic. We are going to need to protect (ourselves) simultaneously against the flu and COVID. A single shot is a great way to do that – nobody wants two needles; nobody wants two trips to get vaccinated.
An effective combination vaccine would be a really great tool.
We have to wait to see what the science shows us, because they are quite different viruses. We won’t know if a combination vaccine works well and has acceptable side effects until we do those studies.
Q. Do you know at this point whether the side effects from two vaccines would be additive? Is there any way to predict that?
There is no way to predict. There are so many things that go into whether someone has side effects that we don’t understand. With fairly reactogenic vaccines like the mRNA vaccines, lots of people have no side effects whatsoever and others are really uncomfortable for 24 hours.
Flu is generally a better tolerated vaccine. There are still people who get muscle aches and very sore arms. I don’t think we can predict if getting two will be additive or just the same as getting one vaccine.
Q: Other than convenience and the benefit for people who are needle-phobic, are there any other advantages of combining them into one shot?
The logistics alone are enough to justify having one effective product if we can make one. It should reduce the overall cost of administration and reduce time off from work.
The combination vaccines given by pediatricians have been very successful. They reduce the number of needles for kids and make it much easier for parents and the pediatricians administering them. The same principle should apply to adults, who sometimes are less brave about needles than kids are.
Historically, combined vaccines in general have worked as well as vaccines given alone, but there have been exceptions. We just have to see what the products look like.
Q: For now, the flu vaccine and COVID-19 vaccine are single products. If you get them separately, is it better to put some time between the two?
We don’t know. There are studies that probably won’t be out in time to decide in September. They are looking at whether you get an equivalent immune response if you give them together or apart.
For now, I would say the advantage of getting them together is if you do get side effects, you’ll only get them once – one day to suffer through them. Also, it’s one trip to the doctor.
The potential advantage of separating them is that is how we developed and tested the vaccines. If you do react to them, side effects could be milder, but it will be on two separate days.
I would recommend doing whatever works so that you get both vaccines in a timely manner.
I’m going to get my flu shot as soon as it’s available. If I’m due for a COVID booster at that time, I would probably do them together.
Q: Do you foresee a point in the future when the predominant strain of SARS-CoV-2 will be one of the components of a flu vaccine, like we did in the past with H1N1, etc?
It really remains to be seen, but it is very conceivable it could happen. The same companies that developed COVID-19 vaccines are working on flu vaccines.
Q: Any other advice for people concerned about getting immunized against both COVID-19 and influenza in the coming months?
There is no side effect of the vaccine that begins to approach the risk you face from either disease. It’s really one of the best things you can do to protect yourself is to get vaccinated.
In the case of flu, the vaccine is only modestly effective, but it still saves tens of thousands of lives each year. The SARS-CoV-2 vaccine is a much better vaccine and a deadlier disease.
Dr. Pavia consulted for GlaxoSmithKline on influenza testing.
A version of this article first appeared on Medscape.com.
With first-time COVID-19 immunizations continuing and the plan to offer booster vaccines to most Americans starting next month, what are the considerations for getting COVID-19 and flu shots at the same time?
This news organization asked Andrew T. Pavia, MD, for his advice. He is the George and Esther Gross Presidential Professor and chief of the division of pediatric infectious diseases at the University of Utah, Salt Lake City, and a fellow of the Infectious Diseases Society of America.
Q: With COVID-19 cases surging, is it a good idea to get the flu shot early this season?
Dr. Pavia: I don’t think there is a rush to do it in August, but it is a good idea to get a flu shot this season. The consequences of getting the flu while COVID is circulating are serious.
Q: What are the implications?
There are some we know and some we don’t know. If you develop flu-like symptoms, you’re going to have to get tested. You’re going to have to stay home quite a bit longer if you get a definitive (positive COVID-19) test than you would simply with flu symptoms. Also, you’re probably going to miss work when your workplace is very stressed or your children are stressed by having COVID circulating in schools.
The part we know less about are the implications of getting the flu and COVID together. There is some reason to believe if you get them together, the illness will be more severe. We are seeing that with RSV (respiratory syncytial virus) and parainfluenza and COVID coinfections in children. They appear to be quite severe.
But for flu, we just don’t have the data yet. That’s because there really was no cocirculation of COVID and influenza with the exception of parts of China for a brief part of February and March.
Q: Will the planned administration of booster COVID-19 shots this fall affect the number of people who get the flu vaccine or how it’s distributed?
It creates a lot of logistical challenges, particularly for hospitals and other places that need to vaccinate a large number of their employees for flu and that will need to give COVID boosters at about the same time period. It also creates logistical challenges for doctors’ offices.
But we don’t know of any reason why you can’t give the two shots together.
Q: Is it possible flu season will be more severe because we isolated and wore masks, etc., last winter? Any science behind that?
The more you study flu, the less you can predict, and I’ve been studying flu for a long time. There are reasons that might suggest a severe flu season – there has been limited immunity, and some people are not wearing masks effectively and they are gathering again. Those are things we believe protected us from influenza last season.
But we have not seen flu emerge yet. Normally we look to Australia, New Zealand, and South Africa during their winter – which is our summer – to get some idea of what is over the horizon for the Northern Hemisphere. Flu activity in Australia has been very modest this year.
That might mean flu may not show up for a while, but I would be loathe to make a prediction.
Q: What are the chances we’ll see a flu outbreak like we’re seeing with RSV, which is normally a winter illness?
The fact that we had a summer RSV surge just gives you an idea of how the normal epidemiology of viral infections has been disrupted. It means anything could happen with influenza. It could show up late summer or fall or wait until next spring.
We really don’t understand how those interactions work. When a new flu strain emerges, it often ignores the traditional behavior and shows up in the spring or fall. It happened in the 2009 pandemic, it happened in 1918.
The one thing I would safely predict about the next flu wave is that it will surprise us.
Q: Are you hopeful that combination vaccines in development from a number of companies, such as Moderna, Novavax, and Vivaldi, will be effective?
It is beginning to look like COVID will be with us for the foreseeable future – maybe as a seasonal virus or maybe as an ongoing pandemic. We are going to need to protect (ourselves) simultaneously against the flu and COVID. A single shot is a great way to do that – nobody wants two needles; nobody wants two trips to get vaccinated.
An effective combination vaccine would be a really great tool.
We have to wait to see what the science shows us, because they are quite different viruses. We won’t know if a combination vaccine works well and has acceptable side effects until we do those studies.
Q. Do you know at this point whether the side effects from two vaccines would be additive? Is there any way to predict that?
There is no way to predict. There are so many things that go into whether someone has side effects that we don’t understand. With fairly reactogenic vaccines like the mRNA vaccines, lots of people have no side effects whatsoever and others are really uncomfortable for 24 hours.
Flu is generally a better tolerated vaccine. There are still people who get muscle aches and very sore arms. I don’t think we can predict if getting two will be additive or just the same as getting one vaccine.
Q: Other than convenience and the benefit for people who are needle-phobic, are there any other advantages of combining them into one shot?
The logistics alone are enough to justify having one effective product if we can make one. It should reduce the overall cost of administration and reduce time off from work.
The combination vaccines given by pediatricians have been very successful. They reduce the number of needles for kids and make it much easier for parents and the pediatricians administering them. The same principle should apply to adults, who sometimes are less brave about needles than kids are.
Historically, combined vaccines in general have worked as well as vaccines given alone, but there have been exceptions. We just have to see what the products look like.
Q: For now, the flu vaccine and COVID-19 vaccine are single products. If you get them separately, is it better to put some time between the two?
We don’t know. There are studies that probably won’t be out in time to decide in September. They are looking at whether you get an equivalent immune response if you give them together or apart.
For now, I would say the advantage of getting them together is if you do get side effects, you’ll only get them once – one day to suffer through them. Also, it’s one trip to the doctor.
The potential advantage of separating them is that is how we developed and tested the vaccines. If you do react to them, side effects could be milder, but it will be on two separate days.
I would recommend doing whatever works so that you get both vaccines in a timely manner.
I’m going to get my flu shot as soon as it’s available. If I’m due for a COVID booster at that time, I would probably do them together.
Q: Do you foresee a point in the future when the predominant strain of SARS-CoV-2 will be one of the components of a flu vaccine, like we did in the past with H1N1, etc?
It really remains to be seen, but it is very conceivable it could happen. The same companies that developed COVID-19 vaccines are working on flu vaccines.
Q: Any other advice for people concerned about getting immunized against both COVID-19 and influenza in the coming months?
There is no side effect of the vaccine that begins to approach the risk you face from either disease. It’s really one of the best things you can do to protect yourself is to get vaccinated.
In the case of flu, the vaccine is only modestly effective, but it still saves tens of thousands of lives each year. The SARS-CoV-2 vaccine is a much better vaccine and a deadlier disease.
Dr. Pavia consulted for GlaxoSmithKline on influenza testing.
A version of this article first appeared on Medscape.com.
Plastic barriers may not stop COVID-19 spread, experts say
Plastic barriers that separate people in stores, restaurants, and classrooms may not be as effective at stopping the spread of COVID-19 as originally thought, according to The New York Times.
Scientists who study air flow, ventilation, and aerosol droplets say the barriers may not help, and in fact, could make the situation worse by blocking normal air flow, the newspaper reported.
Typically, as people interact and breathe in a room, currents and ventilation systems recirculate the air and disperse the exhaled particles. With plastic barriers, however, particles could get trapped in “dead zones” and build up.
“If you have a forest of barriers in a classroom, it’s going to interfere with proper ventilation of that room,” Linsey Marr, professor of civil and environmental engineering at Virginia Tech, told the newspaper.
“Everybody’s aerosols are going to be trapped and stuck there and building up, and they will end up spreading beyond your own desk,” she said.
Several variables factor into the efficacy of plastic barriers, The New York Times reported. Shields may stop big respiratory droplets from coughs and sneezes, for instance, but they may not do much to prevent small aerosol particles from viruses such as COVID-19 from spreading.
“We have shown this effect of blocking larger particles, but also that the smaller aerosols travel over the screen and become mixed in the room air within about 5 minutes,” Catherine Noakes, professor of environment engineering at the University of Leeds, told the newspaper.
“This means if people are interacting for more than a few minutes, they would likely be exposed to the virus regardless of the screen,” she said.
The effectiveness of plastic barriers likely also depends on the location and setup, the newspaper reported. A bus driver with a large barrier, for instance, may be able to avoid inhaling the particles that passengers are exhaling. A bank cashier or store clerk behind a large barrier may also be partly protected.
Even still, scientists say more research is needed. For instance, taller barriers are more likely to be effective. However, a large number of barriers in one room could likely block air flow.
Researchers have recommended that schools and offices focus on ventilation, masks, and vaccines to slow the spread of the coronavirus.
“Air flow in rooms is pretty complicated,” Richard Corsi, dean of engineering at the University of California at Davis, told the newspaper.
“Every room is different in terms of the arrangement of furniture, the height of the walls and ceilings, the vents, where the bookshelves are,” he said. “All of these things have a huge impact on the actual flow and air distribution in a room.”
A version of this article first appeared on WebMD.com.
Plastic barriers that separate people in stores, restaurants, and classrooms may not be as effective at stopping the spread of COVID-19 as originally thought, according to The New York Times.
Scientists who study air flow, ventilation, and aerosol droplets say the barriers may not help, and in fact, could make the situation worse by blocking normal air flow, the newspaper reported.
Typically, as people interact and breathe in a room, currents and ventilation systems recirculate the air and disperse the exhaled particles. With plastic barriers, however, particles could get trapped in “dead zones” and build up.
“If you have a forest of barriers in a classroom, it’s going to interfere with proper ventilation of that room,” Linsey Marr, professor of civil and environmental engineering at Virginia Tech, told the newspaper.
“Everybody’s aerosols are going to be trapped and stuck there and building up, and they will end up spreading beyond your own desk,” she said.
Several variables factor into the efficacy of plastic barriers, The New York Times reported. Shields may stop big respiratory droplets from coughs and sneezes, for instance, but they may not do much to prevent small aerosol particles from viruses such as COVID-19 from spreading.
“We have shown this effect of blocking larger particles, but also that the smaller aerosols travel over the screen and become mixed in the room air within about 5 minutes,” Catherine Noakes, professor of environment engineering at the University of Leeds, told the newspaper.
“This means if people are interacting for more than a few minutes, they would likely be exposed to the virus regardless of the screen,” she said.
The effectiveness of plastic barriers likely also depends on the location and setup, the newspaper reported. A bus driver with a large barrier, for instance, may be able to avoid inhaling the particles that passengers are exhaling. A bank cashier or store clerk behind a large barrier may also be partly protected.
Even still, scientists say more research is needed. For instance, taller barriers are more likely to be effective. However, a large number of barriers in one room could likely block air flow.
Researchers have recommended that schools and offices focus on ventilation, masks, and vaccines to slow the spread of the coronavirus.
“Air flow in rooms is pretty complicated,” Richard Corsi, dean of engineering at the University of California at Davis, told the newspaper.
“Every room is different in terms of the arrangement of furniture, the height of the walls and ceilings, the vents, where the bookshelves are,” he said. “All of these things have a huge impact on the actual flow and air distribution in a room.”
A version of this article first appeared on WebMD.com.
Plastic barriers that separate people in stores, restaurants, and classrooms may not be as effective at stopping the spread of COVID-19 as originally thought, according to The New York Times.
Scientists who study air flow, ventilation, and aerosol droplets say the barriers may not help, and in fact, could make the situation worse by blocking normal air flow, the newspaper reported.
Typically, as people interact and breathe in a room, currents and ventilation systems recirculate the air and disperse the exhaled particles. With plastic barriers, however, particles could get trapped in “dead zones” and build up.
“If you have a forest of barriers in a classroom, it’s going to interfere with proper ventilation of that room,” Linsey Marr, professor of civil and environmental engineering at Virginia Tech, told the newspaper.
“Everybody’s aerosols are going to be trapped and stuck there and building up, and they will end up spreading beyond your own desk,” she said.
Several variables factor into the efficacy of plastic barriers, The New York Times reported. Shields may stop big respiratory droplets from coughs and sneezes, for instance, but they may not do much to prevent small aerosol particles from viruses such as COVID-19 from spreading.
“We have shown this effect of blocking larger particles, but also that the smaller aerosols travel over the screen and become mixed in the room air within about 5 minutes,” Catherine Noakes, professor of environment engineering at the University of Leeds, told the newspaper.
“This means if people are interacting for more than a few minutes, they would likely be exposed to the virus regardless of the screen,” she said.
The effectiveness of plastic barriers likely also depends on the location and setup, the newspaper reported. A bus driver with a large barrier, for instance, may be able to avoid inhaling the particles that passengers are exhaling. A bank cashier or store clerk behind a large barrier may also be partly protected.
Even still, scientists say more research is needed. For instance, taller barriers are more likely to be effective. However, a large number of barriers in one room could likely block air flow.
Researchers have recommended that schools and offices focus on ventilation, masks, and vaccines to slow the spread of the coronavirus.
“Air flow in rooms is pretty complicated,” Richard Corsi, dean of engineering at the University of California at Davis, told the newspaper.
“Every room is different in terms of the arrangement of furniture, the height of the walls and ceilings, the vents, where the bookshelves are,” he said. “All of these things have a huge impact on the actual flow and air distribution in a room.”
A version of this article first appeared on WebMD.com.




