User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Sweeping new vaccine mandates will impact most U.S. workers
, including sweeping vaccine mandates that will affect 100 million American workers, nearly two-thirds of the country’s workforce.
“As your president, I’m announcing tonight a new plan to get more Americans vaccinated to combat those blocking public health,” he said Sept. 9.
As part of a six-part plan unveiled in a speech from the State Dining Room of the White House, President Biden said he would require vaccinations for nearly 4 million federal workers and the employees of companies that contract with the federal government.
He has also directed the Occupational Safety and Health Administration to develop a rule that will require large employers -- those with at least 100 employees -- to ensure their workers are vaccinated or tested weekly.
Nearly 17 million health care workers will face new vaccine mandates as part of the conditions of participation in the Medicare and Medicaid programs.
President Biden said the federal government will require staff at federally funded Head Start programs and schools to be vaccinated. He’s also calling on all states to mandate vaccines for teachers.
“A distinct minority of Americans, supported by a distinct minority of elected officials, are keeping us from turning the corner,” PresidentBiden said. “These pandemic politics, as I refer to them, are making people sick, causing unvaccinated people to die.”
One public health official said he was glad to see the president’s bold action.
“What I saw today was the federal government trying to use its powers to create greater safety in the American population,” said Ashish K. Jha, MD, dean of the school of public health at Brown University, Providence, R.I., in a call with reporters after the speech.
National Nurses United, the largest union of registered nurses in the United States, issued a statement in support of President Biden’s new vaccination requirements, but pushed back on his language.
“…as advocates for public health, registered nurses want to be extremely clear: There is no such thing as a pandemic of only the unvaccinated. The science of epidemiology tells us there is just one deadly, global pandemic that has not yet ended, and we are all in it together. To get out of it, we must act together. All of us,” the statement says.
A host of other professional groups, including the American Medical Association and the Association of State and Territorial Health Officials, also issued statements of support for President Biden’s plan.
But the plan was not well received by all.
“I will pursue every legal option available to the state of Georgia to stop this blatantly unlawful overreach by the Biden Administration,” said Georgia Governor Brian Kemp, a Republican, in a Tweet.
The National Council for Occupational Safety and Health called the plan “a missed opportunity” because it failed to include workplace protections for essential workers such as grocery, postal, and transit workers.
“Social distancing, improved ventilation, shift rotation, and protective equipment to reduce exposure are important components of an overall plan to reduce risk and stop the virus. These tools are missing from the new steps President Biden announced today,” said Jessica Martinez, co-executive director of the group.
In addition to the new vaccination requirements, President Biden said extra doses would be on the way for people who have already been fully vaccinated in order to protect against waning immunity, starting on Sept. 20. But he noted that those plans would be contingent on the Food and Drug Administration’s approval for third doses and the Centers for Disease Control and Prevention’s recommendation of the shots.
President Biden pledged to use the Defense Production Act to ramp up production of at-home tests, which have been selling out across the nation as the Delta variant spreads.
He also announced plans to expand access to COVID-19 testing, including offering testing for free at thousands of pharmacies nationwide and getting major retailers to sell at-home COVID-19 tests at cost.
The BinaxNow test kit, which currently retails for $23.99, will now cost about $15 for two tests at Kroger, Amazon, and Walmart, according to the White House. Food banks and community health centers will get free tests, too.
He called on states to set up COVID-19 testing programs at all schools.
Jha said that in his view, the big, game-changing news out of the president’s speech was the expansion of testing.
“Our country has failed to deploy tests in a way that can really bring this pandemic under control,” Jha said. “There are plenty of reasons, data, experience to indicate that if these were widely available, it would make a dramatic difference in reducing infection numbers across our country.”.
Dr. Jha said the private market had not worked effectively to make testing more widely available, so it was “absolutely a requirement of the federal government to step in and make testing more widely available,” he said.
President Biden also announced new economic stimulus programs, saying he’s expanding loan programs to small businesses and streamlining the loan forgiveness process.
President Biden said he’s boosting help for overburdened hospitals, doubling the number of federal surge response teams sent to hard-hit areas to reduce the strain on local health care workers. He said he would increase the pace of antibody treatments to states by 50%.
“We made so much progress during the past 7 months of this pandemic. Even so, we remain at a critical moment, a critical time,” he said. “We have the tools. Now, we just have to finish the job with truth, with science, with confidence and together as one nation.”
A version of this article first appeared on WebMD.com.
, including sweeping vaccine mandates that will affect 100 million American workers, nearly two-thirds of the country’s workforce.
“As your president, I’m announcing tonight a new plan to get more Americans vaccinated to combat those blocking public health,” he said Sept. 9.
As part of a six-part plan unveiled in a speech from the State Dining Room of the White House, President Biden said he would require vaccinations for nearly 4 million federal workers and the employees of companies that contract with the federal government.
He has also directed the Occupational Safety and Health Administration to develop a rule that will require large employers -- those with at least 100 employees -- to ensure their workers are vaccinated or tested weekly.
Nearly 17 million health care workers will face new vaccine mandates as part of the conditions of participation in the Medicare and Medicaid programs.
President Biden said the federal government will require staff at federally funded Head Start programs and schools to be vaccinated. He’s also calling on all states to mandate vaccines for teachers.
“A distinct minority of Americans, supported by a distinct minority of elected officials, are keeping us from turning the corner,” PresidentBiden said. “These pandemic politics, as I refer to them, are making people sick, causing unvaccinated people to die.”
One public health official said he was glad to see the president’s bold action.
“What I saw today was the federal government trying to use its powers to create greater safety in the American population,” said Ashish K. Jha, MD, dean of the school of public health at Brown University, Providence, R.I., in a call with reporters after the speech.
National Nurses United, the largest union of registered nurses in the United States, issued a statement in support of President Biden’s new vaccination requirements, but pushed back on his language.
“…as advocates for public health, registered nurses want to be extremely clear: There is no such thing as a pandemic of only the unvaccinated. The science of epidemiology tells us there is just one deadly, global pandemic that has not yet ended, and we are all in it together. To get out of it, we must act together. All of us,” the statement says.
A host of other professional groups, including the American Medical Association and the Association of State and Territorial Health Officials, also issued statements of support for President Biden’s plan.
But the plan was not well received by all.
“I will pursue every legal option available to the state of Georgia to stop this blatantly unlawful overreach by the Biden Administration,” said Georgia Governor Brian Kemp, a Republican, in a Tweet.
The National Council for Occupational Safety and Health called the plan “a missed opportunity” because it failed to include workplace protections for essential workers such as grocery, postal, and transit workers.
“Social distancing, improved ventilation, shift rotation, and protective equipment to reduce exposure are important components of an overall plan to reduce risk and stop the virus. These tools are missing from the new steps President Biden announced today,” said Jessica Martinez, co-executive director of the group.
In addition to the new vaccination requirements, President Biden said extra doses would be on the way for people who have already been fully vaccinated in order to protect against waning immunity, starting on Sept. 20. But he noted that those plans would be contingent on the Food and Drug Administration’s approval for third doses and the Centers for Disease Control and Prevention’s recommendation of the shots.
President Biden pledged to use the Defense Production Act to ramp up production of at-home tests, which have been selling out across the nation as the Delta variant spreads.
He also announced plans to expand access to COVID-19 testing, including offering testing for free at thousands of pharmacies nationwide and getting major retailers to sell at-home COVID-19 tests at cost.
The BinaxNow test kit, which currently retails for $23.99, will now cost about $15 for two tests at Kroger, Amazon, and Walmart, according to the White House. Food banks and community health centers will get free tests, too.
He called on states to set up COVID-19 testing programs at all schools.
Jha said that in his view, the big, game-changing news out of the president’s speech was the expansion of testing.
“Our country has failed to deploy tests in a way that can really bring this pandemic under control,” Jha said. “There are plenty of reasons, data, experience to indicate that if these were widely available, it would make a dramatic difference in reducing infection numbers across our country.”.
Dr. Jha said the private market had not worked effectively to make testing more widely available, so it was “absolutely a requirement of the federal government to step in and make testing more widely available,” he said.
President Biden also announced new economic stimulus programs, saying he’s expanding loan programs to small businesses and streamlining the loan forgiveness process.
President Biden said he’s boosting help for overburdened hospitals, doubling the number of federal surge response teams sent to hard-hit areas to reduce the strain on local health care workers. He said he would increase the pace of antibody treatments to states by 50%.
“We made so much progress during the past 7 months of this pandemic. Even so, we remain at a critical moment, a critical time,” he said. “We have the tools. Now, we just have to finish the job with truth, with science, with confidence and together as one nation.”
A version of this article first appeared on WebMD.com.
, including sweeping vaccine mandates that will affect 100 million American workers, nearly two-thirds of the country’s workforce.
“As your president, I’m announcing tonight a new plan to get more Americans vaccinated to combat those blocking public health,” he said Sept. 9.
As part of a six-part plan unveiled in a speech from the State Dining Room of the White House, President Biden said he would require vaccinations for nearly 4 million federal workers and the employees of companies that contract with the federal government.
He has also directed the Occupational Safety and Health Administration to develop a rule that will require large employers -- those with at least 100 employees -- to ensure their workers are vaccinated or tested weekly.
Nearly 17 million health care workers will face new vaccine mandates as part of the conditions of participation in the Medicare and Medicaid programs.
President Biden said the federal government will require staff at federally funded Head Start programs and schools to be vaccinated. He’s also calling on all states to mandate vaccines for teachers.
“A distinct minority of Americans, supported by a distinct minority of elected officials, are keeping us from turning the corner,” PresidentBiden said. “These pandemic politics, as I refer to them, are making people sick, causing unvaccinated people to die.”
One public health official said he was glad to see the president’s bold action.
“What I saw today was the federal government trying to use its powers to create greater safety in the American population,” said Ashish K. Jha, MD, dean of the school of public health at Brown University, Providence, R.I., in a call with reporters after the speech.
National Nurses United, the largest union of registered nurses in the United States, issued a statement in support of President Biden’s new vaccination requirements, but pushed back on his language.
“…as advocates for public health, registered nurses want to be extremely clear: There is no such thing as a pandemic of only the unvaccinated. The science of epidemiology tells us there is just one deadly, global pandemic that has not yet ended, and we are all in it together. To get out of it, we must act together. All of us,” the statement says.
A host of other professional groups, including the American Medical Association and the Association of State and Territorial Health Officials, also issued statements of support for President Biden’s plan.
But the plan was not well received by all.
“I will pursue every legal option available to the state of Georgia to stop this blatantly unlawful overreach by the Biden Administration,” said Georgia Governor Brian Kemp, a Republican, in a Tweet.
The National Council for Occupational Safety and Health called the plan “a missed opportunity” because it failed to include workplace protections for essential workers such as grocery, postal, and transit workers.
“Social distancing, improved ventilation, shift rotation, and protective equipment to reduce exposure are important components of an overall plan to reduce risk and stop the virus. These tools are missing from the new steps President Biden announced today,” said Jessica Martinez, co-executive director of the group.
In addition to the new vaccination requirements, President Biden said extra doses would be on the way for people who have already been fully vaccinated in order to protect against waning immunity, starting on Sept. 20. But he noted that those plans would be contingent on the Food and Drug Administration’s approval for third doses and the Centers for Disease Control and Prevention’s recommendation of the shots.
President Biden pledged to use the Defense Production Act to ramp up production of at-home tests, which have been selling out across the nation as the Delta variant spreads.
He also announced plans to expand access to COVID-19 testing, including offering testing for free at thousands of pharmacies nationwide and getting major retailers to sell at-home COVID-19 tests at cost.
The BinaxNow test kit, which currently retails for $23.99, will now cost about $15 for two tests at Kroger, Amazon, and Walmart, according to the White House. Food banks and community health centers will get free tests, too.
He called on states to set up COVID-19 testing programs at all schools.
Jha said that in his view, the big, game-changing news out of the president’s speech was the expansion of testing.
“Our country has failed to deploy tests in a way that can really bring this pandemic under control,” Jha said. “There are plenty of reasons, data, experience to indicate that if these were widely available, it would make a dramatic difference in reducing infection numbers across our country.”.
Dr. Jha said the private market had not worked effectively to make testing more widely available, so it was “absolutely a requirement of the federal government to step in and make testing more widely available,” he said.
President Biden also announced new economic stimulus programs, saying he’s expanding loan programs to small businesses and streamlining the loan forgiveness process.
President Biden said he’s boosting help for overburdened hospitals, doubling the number of federal surge response teams sent to hard-hit areas to reduce the strain on local health care workers. He said he would increase the pace of antibody treatments to states by 50%.
“We made so much progress during the past 7 months of this pandemic. Even so, we remain at a critical moment, a critical time,” he said. “We have the tools. Now, we just have to finish the job with truth, with science, with confidence and together as one nation.”
A version of this article first appeared on WebMD.com.
COVID vaccine response in patients with solid tumors
Among cancer patients with solid tumors, the response to the COVID-19 vaccine 6 months after receiving a second dose was as good as that of the general population, according to new findings from a case-control study.
The BNT162b2, or Pfizer-BioNTech, vaccine (Comirnaty) was previously shown to have good short-term efficacy, immunogenicity, and safety in cancer patients with solid tumors, but little is known about longer-term efficacy in this population, say the investigators.
They assessed responses 6 months after a second dose of the vaccine.
Serologic tests showed that 122 of 154 patients with solid tumors who were actively undergoing cancer treatment (79%) and 114 of 135 age-matched health care workers who served as control persons (84%) were seropositive at 6 months (P = .32).
Most (81%) of the patients with cancer who were seronegative were receiving chemotherapy, the researchers report.
One case of severe COVID-19 that required hospitalization occurred among the solid-tumor group; none occurred among the control persons, they also note.
The findings were published online on Sept. 2 in Cancer Discovery.
“In our study we saw that in all outcomes, including immunogenicity, infectivity rate throughout the 6-month period, and safety, patients with solid tumors depicted a similar trend as the general population,” commented lead author Irit Ben-Aharon, MD, PhD, director of the division of oncology at Rambam Health Care Campus, Haifa, Israel, in an American Association for Cancer Research press statement.
However, she and her coauthors stressed the importance of continuing to follow guidance, such as social distancing and mask wearing, for reducing COVID-19 transmission. “Due to uncertainty of the extended efficacy of the vaccine in the general population and recent reports on rising infection rates among vaccinated individuals, adherence to health care risk reduction recommendations is cardinal,” they write.
The mean age of the control persons in the study was 63 years, and the mean age of the case patients was 66 years. The most common cancers were gastrointestinal (36%), lung (23%), breast (17%), and genitourinary (11%). Treatment protocols included chemotherapy (62%), biological agents (36%), and immunotherapy (30%). Some patients received more than one type of treatment.
All of the reported adverse effects associated with vaccination had resolved at the time of follow-up.
These data can “help inform recommendations surrounding the prioritization of different groups for booster vaccines,” Dr. Ben-Aharon adds.
In fact, recently updated guidelines from the National Comprehensive Cancer Network state that cancer patients with solid tumors who are receiving treatment within 1 year of their initial vaccine dose should be prioritized for a booster vaccine.
The study was partially supported by the Israel Cancer Research Fund. Serologic testing of the control cohort was supported by the Ministry of Health, Israel. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Among cancer patients with solid tumors, the response to the COVID-19 vaccine 6 months after receiving a second dose was as good as that of the general population, according to new findings from a case-control study.
The BNT162b2, or Pfizer-BioNTech, vaccine (Comirnaty) was previously shown to have good short-term efficacy, immunogenicity, and safety in cancer patients with solid tumors, but little is known about longer-term efficacy in this population, say the investigators.
They assessed responses 6 months after a second dose of the vaccine.
Serologic tests showed that 122 of 154 patients with solid tumors who were actively undergoing cancer treatment (79%) and 114 of 135 age-matched health care workers who served as control persons (84%) were seropositive at 6 months (P = .32).
Most (81%) of the patients with cancer who were seronegative were receiving chemotherapy, the researchers report.
One case of severe COVID-19 that required hospitalization occurred among the solid-tumor group; none occurred among the control persons, they also note.
The findings were published online on Sept. 2 in Cancer Discovery.
“In our study we saw that in all outcomes, including immunogenicity, infectivity rate throughout the 6-month period, and safety, patients with solid tumors depicted a similar trend as the general population,” commented lead author Irit Ben-Aharon, MD, PhD, director of the division of oncology at Rambam Health Care Campus, Haifa, Israel, in an American Association for Cancer Research press statement.
However, she and her coauthors stressed the importance of continuing to follow guidance, such as social distancing and mask wearing, for reducing COVID-19 transmission. “Due to uncertainty of the extended efficacy of the vaccine in the general population and recent reports on rising infection rates among vaccinated individuals, adherence to health care risk reduction recommendations is cardinal,” they write.
The mean age of the control persons in the study was 63 years, and the mean age of the case patients was 66 years. The most common cancers were gastrointestinal (36%), lung (23%), breast (17%), and genitourinary (11%). Treatment protocols included chemotherapy (62%), biological agents (36%), and immunotherapy (30%). Some patients received more than one type of treatment.
All of the reported adverse effects associated with vaccination had resolved at the time of follow-up.
These data can “help inform recommendations surrounding the prioritization of different groups for booster vaccines,” Dr. Ben-Aharon adds.
In fact, recently updated guidelines from the National Comprehensive Cancer Network state that cancer patients with solid tumors who are receiving treatment within 1 year of their initial vaccine dose should be prioritized for a booster vaccine.
The study was partially supported by the Israel Cancer Research Fund. Serologic testing of the control cohort was supported by the Ministry of Health, Israel. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Among cancer patients with solid tumors, the response to the COVID-19 vaccine 6 months after receiving a second dose was as good as that of the general population, according to new findings from a case-control study.
The BNT162b2, or Pfizer-BioNTech, vaccine (Comirnaty) was previously shown to have good short-term efficacy, immunogenicity, and safety in cancer patients with solid tumors, but little is known about longer-term efficacy in this population, say the investigators.
They assessed responses 6 months after a second dose of the vaccine.
Serologic tests showed that 122 of 154 patients with solid tumors who were actively undergoing cancer treatment (79%) and 114 of 135 age-matched health care workers who served as control persons (84%) were seropositive at 6 months (P = .32).
Most (81%) of the patients with cancer who were seronegative were receiving chemotherapy, the researchers report.
One case of severe COVID-19 that required hospitalization occurred among the solid-tumor group; none occurred among the control persons, they also note.
The findings were published online on Sept. 2 in Cancer Discovery.
“In our study we saw that in all outcomes, including immunogenicity, infectivity rate throughout the 6-month period, and safety, patients with solid tumors depicted a similar trend as the general population,” commented lead author Irit Ben-Aharon, MD, PhD, director of the division of oncology at Rambam Health Care Campus, Haifa, Israel, in an American Association for Cancer Research press statement.
However, she and her coauthors stressed the importance of continuing to follow guidance, such as social distancing and mask wearing, for reducing COVID-19 transmission. “Due to uncertainty of the extended efficacy of the vaccine in the general population and recent reports on rising infection rates among vaccinated individuals, adherence to health care risk reduction recommendations is cardinal,” they write.
The mean age of the control persons in the study was 63 years, and the mean age of the case patients was 66 years. The most common cancers were gastrointestinal (36%), lung (23%), breast (17%), and genitourinary (11%). Treatment protocols included chemotherapy (62%), biological agents (36%), and immunotherapy (30%). Some patients received more than one type of treatment.
All of the reported adverse effects associated with vaccination had resolved at the time of follow-up.
These data can “help inform recommendations surrounding the prioritization of different groups for booster vaccines,” Dr. Ben-Aharon adds.
In fact, recently updated guidelines from the National Comprehensive Cancer Network state that cancer patients with solid tumors who are receiving treatment within 1 year of their initial vaccine dose should be prioritized for a booster vaccine.
The study was partially supported by the Israel Cancer Research Fund. Serologic testing of the control cohort was supported by the Ministry of Health, Israel. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Long COVID could spell kidney troubles down the line
Physicians caring for COVID-19 survivors should routinely check kidney function, which is often damaged by the SARS-CoV-2 virus months after both severe and milder cases, new research indicates.
The largest study to date with the longest follow-up of COVID-19-related kidney outcomes also found that every type of kidney problem, including end-stage kidney disease (ESKD), was far more common in COVID-19 survivors who were admitted to the ICU or experienced acute kidney injury (AKI) while hospitalized.
Researchers analyzed U.S. Veterans Health Administration data from more than 1.7 million patients, including more than 89,000 who tested positive for COVID-19, for the study, which was published online Sept. 1, 2021, in the Journal of the American Society of Nephrology.
The risk of kidney problems “is more robust or pronounced in people who have had severe infection, but present in even asymptomatic and mild disease, which shouldn’t be discounted. Those people represent the majority of those with COVID-19,” said senior author Ziyad Al-Aly, MD, of the Veteran Affairs St. Louis Health Care System.
“That’s why the results are important, because even in people with mild disease to start with, the risk of kidney problems is not trivial,” he told this news organization. “It’s smaller than in people who were in the ICU, but it’s not ... zero.”
Experts aren’t yet certain how COVID-19 can damage the kidneys, hypothesizing that several factors may be at play. The virus may directly infect kidney cells rich in ACE2 receptors, which are key to infection, said nephrologist F. Perry Wilson, MD, of Yale University, New Haven, Conn., and a member of Medscape’s advisory board.
Kidneys might also be particularly vulnerable to the inflammatory cascade or blood clotting often seen in COVID-19, Dr. Al-Aly and Wilson both suggested.
COVID-19 survivors more likely to have kidney damage than controls
“A lot of health systems either have or are establishing post-COVID care clinics, which we think should definitely incorporate a kidney component,” Dr. Al-Aly advised. “They should check patients’ blood and urine for kidney problems.”
This is particularly important because “kidney problems, for the most part, are painless and silent,” he added.
“Realizing 2 years down the road that someone has ESKD, where they need dialysis or a kidney transplant, is what we don’t want. We don’t want this to be unrecognized, uncared for, unattended to,” he said.
Dr. Al-Aly and colleagues evaluated VA health system records, including data from 89,216 patients who tested positive for COVID-19 between March 2020 and March 2021, as well as 1.7 million controls who did not have COVID-19. Over a median follow-up of about 5.5 months, participants’ estimated glomerular filtration rate and serum creatinine levels were tracked to assess kidney health and outcomes according to infection severity.
Results were striking, with COVID-19 survivors about one-third more likely than controls to have kidney damage or significant declines in kidney function between 1 and 6 months after infection. More than 4,700 COVID-19 survivors had lost at least 30% of their kidney function within a year, and these patients were 25% more likely to reach that level of decline than controls.
Additionally, COVID-19 survivors were nearly twice as likely to experience AKI and almost three times as likely to be diagnosed with ESKD as controls.
If your patient had COVID-19, ‘it’s reasonable to check kidney function’
“This information tells us that if your patient was sick with COVID-19 and comes for follow-up visits, it’s reasonable to check their kidney function,” Dr. Wilson, who was not involved with the research, told this news organization.
“Even for patients who were not hospitalized, if they were laid low or dehydrated ... it should be part of the post-COVID care package,” he said.
If just a fraction of the millions of COVID-19 survivors in the United States develop long-term kidney problems, the ripple effect on American health care could be substantial, Dr. Wilson and Dr. Al-Aly agreed.
“We’re still living in a pandemic, so it’s hard to tell the total impact,” Dr. Al-Aly said. “But this ultimately will contribute to a rise in burden of kidney disease. This and other long COVID manifestations are going to alter the landscape of clinical care and health care in the United States for a decade or more.”
Because renal problems can limit a patient’s treatment options for other major diseases, including diabetes and cancer, COVID-related kidney damage can ultimately impact survivability.
“There are a lot of medications you can’t use in people with advanced kidney problems,” Dr. Al-Aly said.
The main study limitation was that patients were mostly older White men (median age, 68 years), although more than 9,000 women were included in the VA data, Dr. Al-Aly noted. Additionally, controls were more likely to be younger, Black, living in long-term care, and have higher rates of chronic health conditions and medication use.
The experts agreed that ongoing research tracking kidney outcomes is crucial for years to come.
“We also need to be following a cohort of these patients as part of a research protocol where they come in every 6 months for a standard set of lab tests to really understand what’s going on with their kidneys,” Dr. Wilson said.
“Lastly – and a much tougher sell – is we need biopsies. It’s very hard to infer what’s going on in complex disease with the kidneys without biopsy tissue,” he added.
The study was funded by the American Society of Nephrology and the Department of Veterans Affairs. Dr. Al-Aly and Dr. Wilson reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Physicians caring for COVID-19 survivors should routinely check kidney function, which is often damaged by the SARS-CoV-2 virus months after both severe and milder cases, new research indicates.
The largest study to date with the longest follow-up of COVID-19-related kidney outcomes also found that every type of kidney problem, including end-stage kidney disease (ESKD), was far more common in COVID-19 survivors who were admitted to the ICU or experienced acute kidney injury (AKI) while hospitalized.
Researchers analyzed U.S. Veterans Health Administration data from more than 1.7 million patients, including more than 89,000 who tested positive for COVID-19, for the study, which was published online Sept. 1, 2021, in the Journal of the American Society of Nephrology.
The risk of kidney problems “is more robust or pronounced in people who have had severe infection, but present in even asymptomatic and mild disease, which shouldn’t be discounted. Those people represent the majority of those with COVID-19,” said senior author Ziyad Al-Aly, MD, of the Veteran Affairs St. Louis Health Care System.
“That’s why the results are important, because even in people with mild disease to start with, the risk of kidney problems is not trivial,” he told this news organization. “It’s smaller than in people who were in the ICU, but it’s not ... zero.”
Experts aren’t yet certain how COVID-19 can damage the kidneys, hypothesizing that several factors may be at play. The virus may directly infect kidney cells rich in ACE2 receptors, which are key to infection, said nephrologist F. Perry Wilson, MD, of Yale University, New Haven, Conn., and a member of Medscape’s advisory board.
Kidneys might also be particularly vulnerable to the inflammatory cascade or blood clotting often seen in COVID-19, Dr. Al-Aly and Wilson both suggested.
COVID-19 survivors more likely to have kidney damage than controls
“A lot of health systems either have or are establishing post-COVID care clinics, which we think should definitely incorporate a kidney component,” Dr. Al-Aly advised. “They should check patients’ blood and urine for kidney problems.”
This is particularly important because “kidney problems, for the most part, are painless and silent,” he added.
“Realizing 2 years down the road that someone has ESKD, where they need dialysis or a kidney transplant, is what we don’t want. We don’t want this to be unrecognized, uncared for, unattended to,” he said.
Dr. Al-Aly and colleagues evaluated VA health system records, including data from 89,216 patients who tested positive for COVID-19 between March 2020 and March 2021, as well as 1.7 million controls who did not have COVID-19. Over a median follow-up of about 5.5 months, participants’ estimated glomerular filtration rate and serum creatinine levels were tracked to assess kidney health and outcomes according to infection severity.
Results were striking, with COVID-19 survivors about one-third more likely than controls to have kidney damage or significant declines in kidney function between 1 and 6 months after infection. More than 4,700 COVID-19 survivors had lost at least 30% of their kidney function within a year, and these patients were 25% more likely to reach that level of decline than controls.
Additionally, COVID-19 survivors were nearly twice as likely to experience AKI and almost three times as likely to be diagnosed with ESKD as controls.
If your patient had COVID-19, ‘it’s reasonable to check kidney function’
“This information tells us that if your patient was sick with COVID-19 and comes for follow-up visits, it’s reasonable to check their kidney function,” Dr. Wilson, who was not involved with the research, told this news organization.
“Even for patients who were not hospitalized, if they were laid low or dehydrated ... it should be part of the post-COVID care package,” he said.
If just a fraction of the millions of COVID-19 survivors in the United States develop long-term kidney problems, the ripple effect on American health care could be substantial, Dr. Wilson and Dr. Al-Aly agreed.
“We’re still living in a pandemic, so it’s hard to tell the total impact,” Dr. Al-Aly said. “But this ultimately will contribute to a rise in burden of kidney disease. This and other long COVID manifestations are going to alter the landscape of clinical care and health care in the United States for a decade or more.”
Because renal problems can limit a patient’s treatment options for other major diseases, including diabetes and cancer, COVID-related kidney damage can ultimately impact survivability.
“There are a lot of medications you can’t use in people with advanced kidney problems,” Dr. Al-Aly said.
The main study limitation was that patients were mostly older White men (median age, 68 years), although more than 9,000 women were included in the VA data, Dr. Al-Aly noted. Additionally, controls were more likely to be younger, Black, living in long-term care, and have higher rates of chronic health conditions and medication use.
The experts agreed that ongoing research tracking kidney outcomes is crucial for years to come.
“We also need to be following a cohort of these patients as part of a research protocol where they come in every 6 months for a standard set of lab tests to really understand what’s going on with their kidneys,” Dr. Wilson said.
“Lastly – and a much tougher sell – is we need biopsies. It’s very hard to infer what’s going on in complex disease with the kidneys without biopsy tissue,” he added.
The study was funded by the American Society of Nephrology and the Department of Veterans Affairs. Dr. Al-Aly and Dr. Wilson reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Physicians caring for COVID-19 survivors should routinely check kidney function, which is often damaged by the SARS-CoV-2 virus months after both severe and milder cases, new research indicates.
The largest study to date with the longest follow-up of COVID-19-related kidney outcomes also found that every type of kidney problem, including end-stage kidney disease (ESKD), was far more common in COVID-19 survivors who were admitted to the ICU or experienced acute kidney injury (AKI) while hospitalized.
Researchers analyzed U.S. Veterans Health Administration data from more than 1.7 million patients, including more than 89,000 who tested positive for COVID-19, for the study, which was published online Sept. 1, 2021, in the Journal of the American Society of Nephrology.
The risk of kidney problems “is more robust or pronounced in people who have had severe infection, but present in even asymptomatic and mild disease, which shouldn’t be discounted. Those people represent the majority of those with COVID-19,” said senior author Ziyad Al-Aly, MD, of the Veteran Affairs St. Louis Health Care System.
“That’s why the results are important, because even in people with mild disease to start with, the risk of kidney problems is not trivial,” he told this news organization. “It’s smaller than in people who were in the ICU, but it’s not ... zero.”
Experts aren’t yet certain how COVID-19 can damage the kidneys, hypothesizing that several factors may be at play. The virus may directly infect kidney cells rich in ACE2 receptors, which are key to infection, said nephrologist F. Perry Wilson, MD, of Yale University, New Haven, Conn., and a member of Medscape’s advisory board.
Kidneys might also be particularly vulnerable to the inflammatory cascade or blood clotting often seen in COVID-19, Dr. Al-Aly and Wilson both suggested.
COVID-19 survivors more likely to have kidney damage than controls
“A lot of health systems either have or are establishing post-COVID care clinics, which we think should definitely incorporate a kidney component,” Dr. Al-Aly advised. “They should check patients’ blood and urine for kidney problems.”
This is particularly important because “kidney problems, for the most part, are painless and silent,” he added.
“Realizing 2 years down the road that someone has ESKD, where they need dialysis or a kidney transplant, is what we don’t want. We don’t want this to be unrecognized, uncared for, unattended to,” he said.
Dr. Al-Aly and colleagues evaluated VA health system records, including data from 89,216 patients who tested positive for COVID-19 between March 2020 and March 2021, as well as 1.7 million controls who did not have COVID-19. Over a median follow-up of about 5.5 months, participants’ estimated glomerular filtration rate and serum creatinine levels were tracked to assess kidney health and outcomes according to infection severity.
Results were striking, with COVID-19 survivors about one-third more likely than controls to have kidney damage or significant declines in kidney function between 1 and 6 months after infection. More than 4,700 COVID-19 survivors had lost at least 30% of their kidney function within a year, and these patients were 25% more likely to reach that level of decline than controls.
Additionally, COVID-19 survivors were nearly twice as likely to experience AKI and almost three times as likely to be diagnosed with ESKD as controls.
If your patient had COVID-19, ‘it’s reasonable to check kidney function’
“This information tells us that if your patient was sick with COVID-19 and comes for follow-up visits, it’s reasonable to check their kidney function,” Dr. Wilson, who was not involved with the research, told this news organization.
“Even for patients who were not hospitalized, if they were laid low or dehydrated ... it should be part of the post-COVID care package,” he said.
If just a fraction of the millions of COVID-19 survivors in the United States develop long-term kidney problems, the ripple effect on American health care could be substantial, Dr. Wilson and Dr. Al-Aly agreed.
“We’re still living in a pandemic, so it’s hard to tell the total impact,” Dr. Al-Aly said. “But this ultimately will contribute to a rise in burden of kidney disease. This and other long COVID manifestations are going to alter the landscape of clinical care and health care in the United States for a decade or more.”
Because renal problems can limit a patient’s treatment options for other major diseases, including diabetes and cancer, COVID-related kidney damage can ultimately impact survivability.
“There are a lot of medications you can’t use in people with advanced kidney problems,” Dr. Al-Aly said.
The main study limitation was that patients were mostly older White men (median age, 68 years), although more than 9,000 women were included in the VA data, Dr. Al-Aly noted. Additionally, controls were more likely to be younger, Black, living in long-term care, and have higher rates of chronic health conditions and medication use.
The experts agreed that ongoing research tracking kidney outcomes is crucial for years to come.
“We also need to be following a cohort of these patients as part of a research protocol where they come in every 6 months for a standard set of lab tests to really understand what’s going on with their kidneys,” Dr. Wilson said.
“Lastly – and a much tougher sell – is we need biopsies. It’s very hard to infer what’s going on in complex disease with the kidneys without biopsy tissue,” he added.
The study was funded by the American Society of Nephrology and the Department of Veterans Affairs. Dr. Al-Aly and Dr. Wilson reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Elderly mice receive the gift of warmth
Steal from the warm, give to the cold
If there’s one constant in life other than taxes, it’s elderly people moving to Florida. The Sunshine State’s reputation as a giant retirement home needs no elaboration, but why do senior citizens gravitate there? Well, many reasons, but a big one is that, the older you get, the more susceptible and sensitive you are to the cold. And now, according to a new study, we may have identified a culprit.
Researchers from Yale University examined a group of mice and found that the older ones lacked ICL2 cells in their fatty tissue. These cells, at least in younger mice, help restore body heat when exposed to cold temperatures. Lacking these cells meant that older mice had a limited ability to burn their fat and raise their temperature in response to cold.
Well, job done, all we need to do now is stimulate production of ICL2 cells in elderly people, and they’ll be able to go outside in 80-degree weather without a sweater again. Except there’s a problem. In a cruel twist of fate, when the elderly mice were given a molecule to boost ICL2 cell production, they actually became less tolerant of the cold than at baseline. Oops.
The scientists didn’t give up though, and gave their elderly mice ICL2 cells from young mice. This finally did the trick, though we have to admit, if that treatment does eventually scale up to humans, the prospect of a bunch of senior citizens taking ICL2 cells from young people to stay warm does sound a bit like a bad vampire movie premise. “I vant to suck your immune cell group 2 innate lymphoid cells!” Not the most pithy catch phrase in the world.
Grocery store tapping your subconscious? It’s a good thing
We all know there’s marketing and functionality elements to grocery stores and how they’re set up for your shopping pleasure. But what if I told you that the good old supermarket subconscious trick works on how healthy food decisions are?
In a recent study, researchers at the University of Southampton in England found that if you placed a wider selection of fruits and vegetables near the entrances and more nonfood items near checkouts, sales decreased on the sweets and increased on the produce. “The findings of our study suggest that a healthier store layout could lead to nearly 10,000 extra portions of fruit and vegetables and approximately 1,500 fewer portions of confectionery being sold on a weekly basis in each store,” lead author Dr. Christina Vogel explained.
You’re probably thinking that food placement studies aren’t new. That’s true, but this one went above and beyond. Instead of just looking at the influence placement has on purchase, this one took it further by trying to reduce the consumers’ “calorie opportunities” and examining the effect on sales. Also, customer loyalty, patterns, and diets were taken into account across multiple household members.
The researchers think shifting the layouts in grocery stores could shift people’s food choices, producing a domino effect on the population’s overall diet. With obesity, diabetes, and cardiology concerns always looming, swaying consumers toward healthier food choices makes for better public health overall.
So if you feel like you’re being subconsciously assaulted by veggies every time you walk into Trader Joe’s, just know it’s for your own good.
TikTokers take on tics
We know TikTok is what makes a lot of teens and young adults tick, but what if TikTokers are actually catching tic disorders from other TikTokers?
TikTok blew up during the pandemic. Many people were stuck at home and had nothing better to do than make and watch TikTok videos. The pandemic brought isolation, uncertainty, and anxiety. The stress that followed may have caused many people, mostly women and young girls, to develop tic disorders.
There’s a TikTok for everything, whether it’s a new dance or a recipe. Many people even use TikTok to speak out about their illnesses. Several TikTokers have Tourette’s syndrome and show their tics on their videos. It appears that some audience members actually “catch” the tics from watching the videos and are then unable to stop certain jerking movements or saying specific words.
Neurologists at the University of Calgary (Alta.), who were hearing from colleagues and getting referrals of such patients, called it “an epidemic within the pandemic.” The behavior is not actually Tourette’s, they told Vice, but the patients “cannot stop, and we have absolutely witnessed that.”
There is, of course, controversy over the issue. One individual with the condition said, “I feel like there’s a lot of really weird, backwards stigma on TikTok about tic disorders. Like, you aren’t allowed to have one unless it’s this one.”
Who would have guessed that people would disagree over stuff on the Internet?
Look on the bright side: Obesity edition
The pandemic may have postponed “Top Gun: Maverick” and “The Marvelous Mrs. Maisel” until who-knows-when, but we here at LOTME are happy to announce the nearly-as-anticipated return of Bacteria vs. the World.
As you may recall from our last edition of BVTW, bacteria battled the ghost of Charles Darwin, who had taken the earthly form of antibiotics capable of stopping bacterial evolution. Tonight, our prokaryotic protagonists take on an equally relentless and ubiquitous challenger: obesity.
Specifically, we’re putting bacteria up against the obesity survival paradox, that phenomenon in which obesity and overweight seem to protect against – yes, you guessed it – bacterial infections.
A Swedish research team observed a group of 2,196 individual adults who received care for suspected severe bacterial infection at Skaraborg Hospital in Skövde. One year after hospitalization, 26% of normal-weight (body mass index, 18.5-24.99) patients were dead, compared with 17% of overweight (BMI, 25.0-29.99), 16% of obese (BMI, 30.0-34.99), and 9% of very obese (BMI >35) patients.
These results confirm the obesity survival paradox, but “what we don’t know is how being overweight can benefit the patient with a bacterial infection, or whether it’s connected with functions in the immune system and how they’re regulated,” lead author Dr. Åsa Alsiö said in a written statement.
A spokes-cell for the bacteria disputed the results and challenged the legitimacy of the investigators. When asked if there should be some sort of reexamination of the findings, he/she/it replied: “You bet your flagella.” We then pointed out that humans don’t have flagellum, and the representative raised his/her/its flagella in what could only be considered an obscene gesture.
Steal from the warm, give to the cold
If there’s one constant in life other than taxes, it’s elderly people moving to Florida. The Sunshine State’s reputation as a giant retirement home needs no elaboration, but why do senior citizens gravitate there? Well, many reasons, but a big one is that, the older you get, the more susceptible and sensitive you are to the cold. And now, according to a new study, we may have identified a culprit.
Researchers from Yale University examined a group of mice and found that the older ones lacked ICL2 cells in their fatty tissue. These cells, at least in younger mice, help restore body heat when exposed to cold temperatures. Lacking these cells meant that older mice had a limited ability to burn their fat and raise their temperature in response to cold.
Well, job done, all we need to do now is stimulate production of ICL2 cells in elderly people, and they’ll be able to go outside in 80-degree weather without a sweater again. Except there’s a problem. In a cruel twist of fate, when the elderly mice were given a molecule to boost ICL2 cell production, they actually became less tolerant of the cold than at baseline. Oops.
The scientists didn’t give up though, and gave their elderly mice ICL2 cells from young mice. This finally did the trick, though we have to admit, if that treatment does eventually scale up to humans, the prospect of a bunch of senior citizens taking ICL2 cells from young people to stay warm does sound a bit like a bad vampire movie premise. “I vant to suck your immune cell group 2 innate lymphoid cells!” Not the most pithy catch phrase in the world.
Grocery store tapping your subconscious? It’s a good thing
We all know there’s marketing and functionality elements to grocery stores and how they’re set up for your shopping pleasure. But what if I told you that the good old supermarket subconscious trick works on how healthy food decisions are?
In a recent study, researchers at the University of Southampton in England found that if you placed a wider selection of fruits and vegetables near the entrances and more nonfood items near checkouts, sales decreased on the sweets and increased on the produce. “The findings of our study suggest that a healthier store layout could lead to nearly 10,000 extra portions of fruit and vegetables and approximately 1,500 fewer portions of confectionery being sold on a weekly basis in each store,” lead author Dr. Christina Vogel explained.
You’re probably thinking that food placement studies aren’t new. That’s true, but this one went above and beyond. Instead of just looking at the influence placement has on purchase, this one took it further by trying to reduce the consumers’ “calorie opportunities” and examining the effect on sales. Also, customer loyalty, patterns, and diets were taken into account across multiple household members.
The researchers think shifting the layouts in grocery stores could shift people’s food choices, producing a domino effect on the population’s overall diet. With obesity, diabetes, and cardiology concerns always looming, swaying consumers toward healthier food choices makes for better public health overall.
So if you feel like you’re being subconsciously assaulted by veggies every time you walk into Trader Joe’s, just know it’s for your own good.
TikTokers take on tics
We know TikTok is what makes a lot of teens and young adults tick, but what if TikTokers are actually catching tic disorders from other TikTokers?
TikTok blew up during the pandemic. Many people were stuck at home and had nothing better to do than make and watch TikTok videos. The pandemic brought isolation, uncertainty, and anxiety. The stress that followed may have caused many people, mostly women and young girls, to develop tic disorders.
There’s a TikTok for everything, whether it’s a new dance or a recipe. Many people even use TikTok to speak out about their illnesses. Several TikTokers have Tourette’s syndrome and show their tics on their videos. It appears that some audience members actually “catch” the tics from watching the videos and are then unable to stop certain jerking movements or saying specific words.
Neurologists at the University of Calgary (Alta.), who were hearing from colleagues and getting referrals of such patients, called it “an epidemic within the pandemic.” The behavior is not actually Tourette’s, they told Vice, but the patients “cannot stop, and we have absolutely witnessed that.”
There is, of course, controversy over the issue. One individual with the condition said, “I feel like there’s a lot of really weird, backwards stigma on TikTok about tic disorders. Like, you aren’t allowed to have one unless it’s this one.”
Who would have guessed that people would disagree over stuff on the Internet?
Look on the bright side: Obesity edition
The pandemic may have postponed “Top Gun: Maverick” and “The Marvelous Mrs. Maisel” until who-knows-when, but we here at LOTME are happy to announce the nearly-as-anticipated return of Bacteria vs. the World.
As you may recall from our last edition of BVTW, bacteria battled the ghost of Charles Darwin, who had taken the earthly form of antibiotics capable of stopping bacterial evolution. Tonight, our prokaryotic protagonists take on an equally relentless and ubiquitous challenger: obesity.
Specifically, we’re putting bacteria up against the obesity survival paradox, that phenomenon in which obesity and overweight seem to protect against – yes, you guessed it – bacterial infections.
A Swedish research team observed a group of 2,196 individual adults who received care for suspected severe bacterial infection at Skaraborg Hospital in Skövde. One year after hospitalization, 26% of normal-weight (body mass index, 18.5-24.99) patients were dead, compared with 17% of overweight (BMI, 25.0-29.99), 16% of obese (BMI, 30.0-34.99), and 9% of very obese (BMI >35) patients.
These results confirm the obesity survival paradox, but “what we don’t know is how being overweight can benefit the patient with a bacterial infection, or whether it’s connected with functions in the immune system and how they’re regulated,” lead author Dr. Åsa Alsiö said in a written statement.
A spokes-cell for the bacteria disputed the results and challenged the legitimacy of the investigators. When asked if there should be some sort of reexamination of the findings, he/she/it replied: “You bet your flagella.” We then pointed out that humans don’t have flagellum, and the representative raised his/her/its flagella in what could only be considered an obscene gesture.
Steal from the warm, give to the cold
If there’s one constant in life other than taxes, it’s elderly people moving to Florida. The Sunshine State’s reputation as a giant retirement home needs no elaboration, but why do senior citizens gravitate there? Well, many reasons, but a big one is that, the older you get, the more susceptible and sensitive you are to the cold. And now, according to a new study, we may have identified a culprit.
Researchers from Yale University examined a group of mice and found that the older ones lacked ICL2 cells in their fatty tissue. These cells, at least in younger mice, help restore body heat when exposed to cold temperatures. Lacking these cells meant that older mice had a limited ability to burn their fat and raise their temperature in response to cold.
Well, job done, all we need to do now is stimulate production of ICL2 cells in elderly people, and they’ll be able to go outside in 80-degree weather without a sweater again. Except there’s a problem. In a cruel twist of fate, when the elderly mice were given a molecule to boost ICL2 cell production, they actually became less tolerant of the cold than at baseline. Oops.
The scientists didn’t give up though, and gave their elderly mice ICL2 cells from young mice. This finally did the trick, though we have to admit, if that treatment does eventually scale up to humans, the prospect of a bunch of senior citizens taking ICL2 cells from young people to stay warm does sound a bit like a bad vampire movie premise. “I vant to suck your immune cell group 2 innate lymphoid cells!” Not the most pithy catch phrase in the world.
Grocery store tapping your subconscious? It’s a good thing
We all know there’s marketing and functionality elements to grocery stores and how they’re set up for your shopping pleasure. But what if I told you that the good old supermarket subconscious trick works on how healthy food decisions are?
In a recent study, researchers at the University of Southampton in England found that if you placed a wider selection of fruits and vegetables near the entrances and more nonfood items near checkouts, sales decreased on the sweets and increased on the produce. “The findings of our study suggest that a healthier store layout could lead to nearly 10,000 extra portions of fruit and vegetables and approximately 1,500 fewer portions of confectionery being sold on a weekly basis in each store,” lead author Dr. Christina Vogel explained.
You’re probably thinking that food placement studies aren’t new. That’s true, but this one went above and beyond. Instead of just looking at the influence placement has on purchase, this one took it further by trying to reduce the consumers’ “calorie opportunities” and examining the effect on sales. Also, customer loyalty, patterns, and diets were taken into account across multiple household members.
The researchers think shifting the layouts in grocery stores could shift people’s food choices, producing a domino effect on the population’s overall diet. With obesity, diabetes, and cardiology concerns always looming, swaying consumers toward healthier food choices makes for better public health overall.
So if you feel like you’re being subconsciously assaulted by veggies every time you walk into Trader Joe’s, just know it’s for your own good.
TikTokers take on tics
We know TikTok is what makes a lot of teens and young adults tick, but what if TikTokers are actually catching tic disorders from other TikTokers?
TikTok blew up during the pandemic. Many people were stuck at home and had nothing better to do than make and watch TikTok videos. The pandemic brought isolation, uncertainty, and anxiety. The stress that followed may have caused many people, mostly women and young girls, to develop tic disorders.
There’s a TikTok for everything, whether it’s a new dance or a recipe. Many people even use TikTok to speak out about their illnesses. Several TikTokers have Tourette’s syndrome and show their tics on their videos. It appears that some audience members actually “catch” the tics from watching the videos and are then unable to stop certain jerking movements or saying specific words.
Neurologists at the University of Calgary (Alta.), who were hearing from colleagues and getting referrals of such patients, called it “an epidemic within the pandemic.” The behavior is not actually Tourette’s, they told Vice, but the patients “cannot stop, and we have absolutely witnessed that.”
There is, of course, controversy over the issue. One individual with the condition said, “I feel like there’s a lot of really weird, backwards stigma on TikTok about tic disorders. Like, you aren’t allowed to have one unless it’s this one.”
Who would have guessed that people would disagree over stuff on the Internet?
Look on the bright side: Obesity edition
The pandemic may have postponed “Top Gun: Maverick” and “The Marvelous Mrs. Maisel” until who-knows-when, but we here at LOTME are happy to announce the nearly-as-anticipated return of Bacteria vs. the World.
As you may recall from our last edition of BVTW, bacteria battled the ghost of Charles Darwin, who had taken the earthly form of antibiotics capable of stopping bacterial evolution. Tonight, our prokaryotic protagonists take on an equally relentless and ubiquitous challenger: obesity.
Specifically, we’re putting bacteria up against the obesity survival paradox, that phenomenon in which obesity and overweight seem to protect against – yes, you guessed it – bacterial infections.
A Swedish research team observed a group of 2,196 individual adults who received care for suspected severe bacterial infection at Skaraborg Hospital in Skövde. One year after hospitalization, 26% of normal-weight (body mass index, 18.5-24.99) patients were dead, compared with 17% of overweight (BMI, 25.0-29.99), 16% of obese (BMI, 30.0-34.99), and 9% of very obese (BMI >35) patients.
These results confirm the obesity survival paradox, but “what we don’t know is how being overweight can benefit the patient with a bacterial infection, or whether it’s connected with functions in the immune system and how they’re regulated,” lead author Dr. Åsa Alsiö said in a written statement.
A spokes-cell for the bacteria disputed the results and challenged the legitimacy of the investigators. When asked if there should be some sort of reexamination of the findings, he/she/it replied: “You bet your flagella.” We then pointed out that humans don’t have flagellum, and the representative raised his/her/its flagella in what could only be considered an obscene gesture.
Choosing Wisely campaign targets waste and overuse in hospital pediatrics
“Health care spending and health care waste is a huge problem in the U.S., including for children,” Vivian Lee, MD, of Children’s Hospital, Los Angeles, said in a presentation at the 2021 virtual Pediatric Hospital Medicine conference.
Data from a 2019 study suggested that approximately 25% of health care spending in the United States qualifies as “wasteful spending,” in categories such as overtesting, and unnecessary hospitalization, Dr. Lee said. “It is essential for physicians in hospitals to be stewards of high-value care,” she emphasized.
To combat wasteful spending and control health care costs, the Choosing Wisely campaign was created in 2012 as an initiative from the American Board of Internal Medicine Foundation. An ongoing goal of the campaign is to raise awareness among physicians and patients about potential areas of low-value services and overuse. The overall campaign includes clinician-driven recommendations from multiple medical organizations.
The PHM produced its first set of five recommendations in 2012, Dr. Lee said. These recommendations, titled “Five Things Physicians and Patients Should Question,” have been updated for 2021. The updated recommendations were created as a partnership among the Academic Pediatric Association, the American Academy of Pediatrics, and the Society of Hospital Medicine. A joint committee reviewed the latest evidence, and the updates were approved by the societies and published by the ABIM in January 2021.
“We think these recommendations truly reflect an exciting and evolving landscape for pediatric hospitalists,” Dr. Lee said. “There is a greater focus on opportunities to transition out of the hospital sooner, or avoid hospitalization altogether. There is an emphasis on antibiotic stewardship and a growing recognition of the impact that overuse may have on our vulnerable neonatal population,” she said. Several members of the Choosing Wisely panel presented the recommendations during the virtual presentation.
Revised recommendations
The new “Five Things Physicians and Patients Should Question” are as follows:
1. Do not prescribe IV antibiotics for predetermined durations for patients hospitalized with infections such as pyelonephritis, osteomyelitis, and complicated pneumonia. Consider early transition to oral antibiotics.
Many antibiotic doses used in clinical practice are preset durations that are not based on high-quality evidence, said Mike Tchou, MD, of Children’s Hospital of Colorado in Aurora. However, studies now show that earlier transition to enteral antibiotics can improve a range of outcomes including neonatal UTIs, osteomyelitis, and complicated pneumonia, he said. Considering early transition based on a patient’s response can decrease adverse events, pain, length of stay, and health care costs, he explained.
2. Do not continue hospitalization in well-appearing febrile infants once bacterial cultures (i.e., blood, cerebrospinal, and/or urine) have been confirmed negative for 24-36 hours, if adequate outpatient follow-up can be assured.
Recent data indicate that continuing hospitalization beyond 24-36 hours of confirmed negative bacterial cultures does not improve clinical outcomes for well-appearing infants admitted for concern of serious bacterial infection, said Paula Soung, MD, of Children’s Wisconsin in Milwaukee. In fact, “blood culture yield is highest in the first 12-36 hours after incubation with multiple studies demonstrating > 90% of pathogen cultures being positive by 24 hours,” Dr. Soung said. “If adequate outpatient follow-up can be assured, discharging well-appearing febrile infants at 24-36 hours after confirming cultures are negative has many positive outcomes,” she said.
3. Do not initiate phototherapy in term or late preterm well-appearing infants with neonatal hyperbilirubinemia if their bilirubin is below levels at which the AAP guidelines recommend treatment.
In making this recommendation, “we considered that the risk of kernicterus and cerebral palsy is extremely low in otherwise healthy term and late preterm newborns,” said Allison Holmes, MD, of Children’s Hospital at Dartmouth-Hitchcock, Manchester, N.H. “Subthreshold phototherapy leads to unnecessary hospitalization and its associated costs and harms,” and data show that kernicterus generally occurs close to 40 mg/dL and occurs most often in infants with hemolysis, she added.
The evidence for the recommendations included data showing that, among other factors, 8.6 of 100,000 babies have a bilirubin greater than 30 mg/dL, said Dr. Holmes. Risks of using subthreshold phototherapy include increased length of stay, increased readmissions, and increased costs, as well as decreased breastfeeding, bonding with parents, and increased parental anxiety. “Adding prolonged hospitalization for an intervention that might not be necessary can be stressful for parents,” she said.
4. Do not use broad-spectrum antibiotics such as ceftriaxone for children hospitalized with uncomplicated community-acquired pneumonia. Use narrow-spectrum antibiotics such as penicillin, ampicillin, or amoxicillin.
Michelle Lossius, MD, of the Shands Hospital for Children at the University of Florida, Gainesville, noted that the recommendations reflect IDSA guidelines from 2011 advising the use of ampicillin or penicillin for this population of children. More recent studies with large populations support the ability of narrow-spectrum antibiotics to limit the development of resistant organisms while achieving the same or better outcomes for children hospitalized with CAP, she said.
5. Do not start IV antibiotic therapy on well-appearing newborn infants with isolated risk factors for sepsis such as maternal chorioamnionitis, prolonged rupture of membranes, or untreated group-B streptococcal colonization. Use clinical tools such as an evidence-based sepsis risk calculator to guide management.
“This recommendation combines other recommendations,” said Prabi Rajbhandari, MD, of Akron (Ohio) Children’s Hospital. The evidence is ample, as the Centers for Disease Control and Prevention recommends the use of sepsis calculators to guide clinical management in sepsis patients, she said.
Data comparing periods before and after the adoption of a sepsis risk calculator showed a significant reduction in the use of blood cultures and antibiotics, she noted. Other risks of jumping to IV antibiotics include increased hospital stay, increased parental anxiety, and decreased parental bonding, Dr. Rajbhandari added.
Next steps include how to prioritize implementation, as well as deimplementation of outdated practices, said Francisco Alvarez, MD, of Lucile Packard Children’s Hospital, Palo Alto, Calif. “A lot of our practices were started without good evidence for why they should be done,” he said. Other steps include value improvement research; use of dashboards and benchmarking; involving other stakeholders including patients, families, and other health care providers; and addressing racial disparities, he concluded.
The presenters had no financial conflicts to disclose. The conference was sponsored by the Academic Pediatric Association, the American Academy of Pediatrics, and the Society of Hospital Medicine.
“Health care spending and health care waste is a huge problem in the U.S., including for children,” Vivian Lee, MD, of Children’s Hospital, Los Angeles, said in a presentation at the 2021 virtual Pediatric Hospital Medicine conference.
Data from a 2019 study suggested that approximately 25% of health care spending in the United States qualifies as “wasteful spending,” in categories such as overtesting, and unnecessary hospitalization, Dr. Lee said. “It is essential for physicians in hospitals to be stewards of high-value care,” she emphasized.
To combat wasteful spending and control health care costs, the Choosing Wisely campaign was created in 2012 as an initiative from the American Board of Internal Medicine Foundation. An ongoing goal of the campaign is to raise awareness among physicians and patients about potential areas of low-value services and overuse. The overall campaign includes clinician-driven recommendations from multiple medical organizations.
The PHM produced its first set of five recommendations in 2012, Dr. Lee said. These recommendations, titled “Five Things Physicians and Patients Should Question,” have been updated for 2021. The updated recommendations were created as a partnership among the Academic Pediatric Association, the American Academy of Pediatrics, and the Society of Hospital Medicine. A joint committee reviewed the latest evidence, and the updates were approved by the societies and published by the ABIM in January 2021.
“We think these recommendations truly reflect an exciting and evolving landscape for pediatric hospitalists,” Dr. Lee said. “There is a greater focus on opportunities to transition out of the hospital sooner, or avoid hospitalization altogether. There is an emphasis on antibiotic stewardship and a growing recognition of the impact that overuse may have on our vulnerable neonatal population,” she said. Several members of the Choosing Wisely panel presented the recommendations during the virtual presentation.
Revised recommendations
The new “Five Things Physicians and Patients Should Question” are as follows:
1. Do not prescribe IV antibiotics for predetermined durations for patients hospitalized with infections such as pyelonephritis, osteomyelitis, and complicated pneumonia. Consider early transition to oral antibiotics.
Many antibiotic doses used in clinical practice are preset durations that are not based on high-quality evidence, said Mike Tchou, MD, of Children’s Hospital of Colorado in Aurora. However, studies now show that earlier transition to enteral antibiotics can improve a range of outcomes including neonatal UTIs, osteomyelitis, and complicated pneumonia, he said. Considering early transition based on a patient’s response can decrease adverse events, pain, length of stay, and health care costs, he explained.
2. Do not continue hospitalization in well-appearing febrile infants once bacterial cultures (i.e., blood, cerebrospinal, and/or urine) have been confirmed negative for 24-36 hours, if adequate outpatient follow-up can be assured.
Recent data indicate that continuing hospitalization beyond 24-36 hours of confirmed negative bacterial cultures does not improve clinical outcomes for well-appearing infants admitted for concern of serious bacterial infection, said Paula Soung, MD, of Children’s Wisconsin in Milwaukee. In fact, “blood culture yield is highest in the first 12-36 hours after incubation with multiple studies demonstrating > 90% of pathogen cultures being positive by 24 hours,” Dr. Soung said. “If adequate outpatient follow-up can be assured, discharging well-appearing febrile infants at 24-36 hours after confirming cultures are negative has many positive outcomes,” she said.
3. Do not initiate phototherapy in term or late preterm well-appearing infants with neonatal hyperbilirubinemia if their bilirubin is below levels at which the AAP guidelines recommend treatment.
In making this recommendation, “we considered that the risk of kernicterus and cerebral palsy is extremely low in otherwise healthy term and late preterm newborns,” said Allison Holmes, MD, of Children’s Hospital at Dartmouth-Hitchcock, Manchester, N.H. “Subthreshold phototherapy leads to unnecessary hospitalization and its associated costs and harms,” and data show that kernicterus generally occurs close to 40 mg/dL and occurs most often in infants with hemolysis, she added.
The evidence for the recommendations included data showing that, among other factors, 8.6 of 100,000 babies have a bilirubin greater than 30 mg/dL, said Dr. Holmes. Risks of using subthreshold phototherapy include increased length of stay, increased readmissions, and increased costs, as well as decreased breastfeeding, bonding with parents, and increased parental anxiety. “Adding prolonged hospitalization for an intervention that might not be necessary can be stressful for parents,” she said.
4. Do not use broad-spectrum antibiotics such as ceftriaxone for children hospitalized with uncomplicated community-acquired pneumonia. Use narrow-spectrum antibiotics such as penicillin, ampicillin, or amoxicillin.
Michelle Lossius, MD, of the Shands Hospital for Children at the University of Florida, Gainesville, noted that the recommendations reflect IDSA guidelines from 2011 advising the use of ampicillin or penicillin for this population of children. More recent studies with large populations support the ability of narrow-spectrum antibiotics to limit the development of resistant organisms while achieving the same or better outcomes for children hospitalized with CAP, she said.
5. Do not start IV antibiotic therapy on well-appearing newborn infants with isolated risk factors for sepsis such as maternal chorioamnionitis, prolonged rupture of membranes, or untreated group-B streptococcal colonization. Use clinical tools such as an evidence-based sepsis risk calculator to guide management.
“This recommendation combines other recommendations,” said Prabi Rajbhandari, MD, of Akron (Ohio) Children’s Hospital. The evidence is ample, as the Centers for Disease Control and Prevention recommends the use of sepsis calculators to guide clinical management in sepsis patients, she said.
Data comparing periods before and after the adoption of a sepsis risk calculator showed a significant reduction in the use of blood cultures and antibiotics, she noted. Other risks of jumping to IV antibiotics include increased hospital stay, increased parental anxiety, and decreased parental bonding, Dr. Rajbhandari added.
Next steps include how to prioritize implementation, as well as deimplementation of outdated practices, said Francisco Alvarez, MD, of Lucile Packard Children’s Hospital, Palo Alto, Calif. “A lot of our practices were started without good evidence for why they should be done,” he said. Other steps include value improvement research; use of dashboards and benchmarking; involving other stakeholders including patients, families, and other health care providers; and addressing racial disparities, he concluded.
The presenters had no financial conflicts to disclose. The conference was sponsored by the Academic Pediatric Association, the American Academy of Pediatrics, and the Society of Hospital Medicine.
“Health care spending and health care waste is a huge problem in the U.S., including for children,” Vivian Lee, MD, of Children’s Hospital, Los Angeles, said in a presentation at the 2021 virtual Pediatric Hospital Medicine conference.
Data from a 2019 study suggested that approximately 25% of health care spending in the United States qualifies as “wasteful spending,” in categories such as overtesting, and unnecessary hospitalization, Dr. Lee said. “It is essential for physicians in hospitals to be stewards of high-value care,” she emphasized.
To combat wasteful spending and control health care costs, the Choosing Wisely campaign was created in 2012 as an initiative from the American Board of Internal Medicine Foundation. An ongoing goal of the campaign is to raise awareness among physicians and patients about potential areas of low-value services and overuse. The overall campaign includes clinician-driven recommendations from multiple medical organizations.
The PHM produced its first set of five recommendations in 2012, Dr. Lee said. These recommendations, titled “Five Things Physicians and Patients Should Question,” have been updated for 2021. The updated recommendations were created as a partnership among the Academic Pediatric Association, the American Academy of Pediatrics, and the Society of Hospital Medicine. A joint committee reviewed the latest evidence, and the updates were approved by the societies and published by the ABIM in January 2021.
“We think these recommendations truly reflect an exciting and evolving landscape for pediatric hospitalists,” Dr. Lee said. “There is a greater focus on opportunities to transition out of the hospital sooner, or avoid hospitalization altogether. There is an emphasis on antibiotic stewardship and a growing recognition of the impact that overuse may have on our vulnerable neonatal population,” she said. Several members of the Choosing Wisely panel presented the recommendations during the virtual presentation.
Revised recommendations
The new “Five Things Physicians and Patients Should Question” are as follows:
1. Do not prescribe IV antibiotics for predetermined durations for patients hospitalized with infections such as pyelonephritis, osteomyelitis, and complicated pneumonia. Consider early transition to oral antibiotics.
Many antibiotic doses used in clinical practice are preset durations that are not based on high-quality evidence, said Mike Tchou, MD, of Children’s Hospital of Colorado in Aurora. However, studies now show that earlier transition to enteral antibiotics can improve a range of outcomes including neonatal UTIs, osteomyelitis, and complicated pneumonia, he said. Considering early transition based on a patient’s response can decrease adverse events, pain, length of stay, and health care costs, he explained.
2. Do not continue hospitalization in well-appearing febrile infants once bacterial cultures (i.e., blood, cerebrospinal, and/or urine) have been confirmed negative for 24-36 hours, if adequate outpatient follow-up can be assured.
Recent data indicate that continuing hospitalization beyond 24-36 hours of confirmed negative bacterial cultures does not improve clinical outcomes for well-appearing infants admitted for concern of serious bacterial infection, said Paula Soung, MD, of Children’s Wisconsin in Milwaukee. In fact, “blood culture yield is highest in the first 12-36 hours after incubation with multiple studies demonstrating > 90% of pathogen cultures being positive by 24 hours,” Dr. Soung said. “If adequate outpatient follow-up can be assured, discharging well-appearing febrile infants at 24-36 hours after confirming cultures are negative has many positive outcomes,” she said.
3. Do not initiate phototherapy in term or late preterm well-appearing infants with neonatal hyperbilirubinemia if their bilirubin is below levels at which the AAP guidelines recommend treatment.
In making this recommendation, “we considered that the risk of kernicterus and cerebral palsy is extremely low in otherwise healthy term and late preterm newborns,” said Allison Holmes, MD, of Children’s Hospital at Dartmouth-Hitchcock, Manchester, N.H. “Subthreshold phototherapy leads to unnecessary hospitalization and its associated costs and harms,” and data show that kernicterus generally occurs close to 40 mg/dL and occurs most often in infants with hemolysis, she added.
The evidence for the recommendations included data showing that, among other factors, 8.6 of 100,000 babies have a bilirubin greater than 30 mg/dL, said Dr. Holmes. Risks of using subthreshold phototherapy include increased length of stay, increased readmissions, and increased costs, as well as decreased breastfeeding, bonding with parents, and increased parental anxiety. “Adding prolonged hospitalization for an intervention that might not be necessary can be stressful for parents,” she said.
4. Do not use broad-spectrum antibiotics such as ceftriaxone for children hospitalized with uncomplicated community-acquired pneumonia. Use narrow-spectrum antibiotics such as penicillin, ampicillin, or amoxicillin.
Michelle Lossius, MD, of the Shands Hospital for Children at the University of Florida, Gainesville, noted that the recommendations reflect IDSA guidelines from 2011 advising the use of ampicillin or penicillin for this population of children. More recent studies with large populations support the ability of narrow-spectrum antibiotics to limit the development of resistant organisms while achieving the same or better outcomes for children hospitalized with CAP, she said.
5. Do not start IV antibiotic therapy on well-appearing newborn infants with isolated risk factors for sepsis such as maternal chorioamnionitis, prolonged rupture of membranes, or untreated group-B streptococcal colonization. Use clinical tools such as an evidence-based sepsis risk calculator to guide management.
“This recommendation combines other recommendations,” said Prabi Rajbhandari, MD, of Akron (Ohio) Children’s Hospital. The evidence is ample, as the Centers for Disease Control and Prevention recommends the use of sepsis calculators to guide clinical management in sepsis patients, she said.
Data comparing periods before and after the adoption of a sepsis risk calculator showed a significant reduction in the use of blood cultures and antibiotics, she noted. Other risks of jumping to IV antibiotics include increased hospital stay, increased parental anxiety, and decreased parental bonding, Dr. Rajbhandari added.
Next steps include how to prioritize implementation, as well as deimplementation of outdated practices, said Francisco Alvarez, MD, of Lucile Packard Children’s Hospital, Palo Alto, Calif. “A lot of our practices were started without good evidence for why they should be done,” he said. Other steps include value improvement research; use of dashboards and benchmarking; involving other stakeholders including patients, families, and other health care providers; and addressing racial disparities, he concluded.
The presenters had no financial conflicts to disclose. The conference was sponsored by the Academic Pediatric Association, the American Academy of Pediatrics, and the Society of Hospital Medicine.
FROM PHM 2021
United States reaches 5 million cases of child COVID
Cases of child COVID-19 set a new 1-week record and the total number of children infected during the pandemic passed 5 million, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
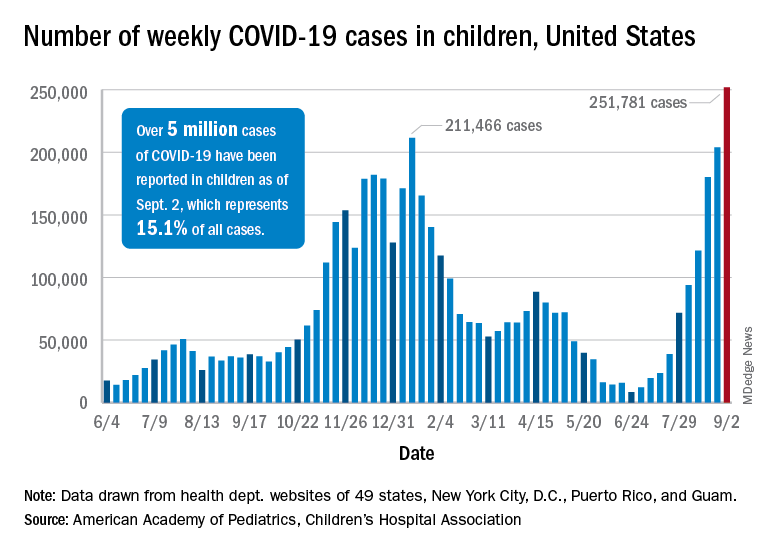
The nearly 282,000 new cases reported in the United States during the week ending Sept. 2 broke the record of 211,000 set in mid-January and brought the cumulative count to 5,049,465 children with COVID-19 since the pandemic began, the AAP and the CHA said in their weekly COVID report.
Hospitalizations in children aged 0-17 years have also reached record levels in recent days. The highest daily admission rate since the pandemic began, 0.51 per 100,000 population, was recorded on Sept. 2, less than 2 months after the nation saw its lowest child COVID admission rate for 1 day: 0.07 per 100,000 on July 4. That’s an increase of 629%, according to data from the Centers for Disease Control and Prevention.
Vaccinations in children, however, did not follow suit. New vaccinations in children aged 12-17 years dropped by 4.5% for the week ending Sept. 6, compared with the week before. Initiations were actually up almost 12% for children aged 16-17, but that was not enough to overcome the continued decline among 12- to 15-year-olds, the CDC said on its COVID Data Tracker.
Despite the decline in new vaccinations, those younger children passed a noteworthy group milestone: 50.9% of all 12- to 15-year-olds now have received at least one dose, with 38.6% having completed the regimen. The 16- to 17-year-olds got an earlier start and have reached 58.9% coverage for one dose and 47.6% for two, the CDC said.
A total of 12.2 million children aged 12-17 years had received at least one dose of COVID vaccine as of Sept. 6, of whom almost 9.5 million are fully vaccinated, based on the CDC data.
At the state level, Vermont has the highest rates for vaccine initiation (75%) and full vaccination (65%), with Massachusetts (75%/62%) and Connecticut (73%/59%) just behind. The other end of the scale is occupied by Wyoming (28% initiation/19% full vaccination), Alabama (32%/19%), and North Dakota (32%/23%), the AAP said in a separate report.
In a recent letter to the Food and Drug Administration, AAP President Lee Savio Beers, MD, said that the “Delta variant is surging at extremely alarming rates in every region of America. This surge is seriously impacting all populations, including children.” Dr. Beers urged the FDA to work “aggressively toward authorizing safe and effective COVID-19 vaccines for children under age 12 as soon as possible.”
Cases of child COVID-19 set a new 1-week record and the total number of children infected during the pandemic passed 5 million, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The nearly 282,000 new cases reported in the United States during the week ending Sept. 2 broke the record of 211,000 set in mid-January and brought the cumulative count to 5,049,465 children with COVID-19 since the pandemic began, the AAP and the CHA said in their weekly COVID report.
Hospitalizations in children aged 0-17 years have also reached record levels in recent days. The highest daily admission rate since the pandemic began, 0.51 per 100,000 population, was recorded on Sept. 2, less than 2 months after the nation saw its lowest child COVID admission rate for 1 day: 0.07 per 100,000 on July 4. That’s an increase of 629%, according to data from the Centers for Disease Control and Prevention.
Vaccinations in children, however, did not follow suit. New vaccinations in children aged 12-17 years dropped by 4.5% for the week ending Sept. 6, compared with the week before. Initiations were actually up almost 12% for children aged 16-17, but that was not enough to overcome the continued decline among 12- to 15-year-olds, the CDC said on its COVID Data Tracker.
Despite the decline in new vaccinations, those younger children passed a noteworthy group milestone: 50.9% of all 12- to 15-year-olds now have received at least one dose, with 38.6% having completed the regimen. The 16- to 17-year-olds got an earlier start and have reached 58.9% coverage for one dose and 47.6% for two, the CDC said.
A total of 12.2 million children aged 12-17 years had received at least one dose of COVID vaccine as of Sept. 6, of whom almost 9.5 million are fully vaccinated, based on the CDC data.
At the state level, Vermont has the highest rates for vaccine initiation (75%) and full vaccination (65%), with Massachusetts (75%/62%) and Connecticut (73%/59%) just behind. The other end of the scale is occupied by Wyoming (28% initiation/19% full vaccination), Alabama (32%/19%), and North Dakota (32%/23%), the AAP said in a separate report.
In a recent letter to the Food and Drug Administration, AAP President Lee Savio Beers, MD, said that the “Delta variant is surging at extremely alarming rates in every region of America. This surge is seriously impacting all populations, including children.” Dr. Beers urged the FDA to work “aggressively toward authorizing safe and effective COVID-19 vaccines for children under age 12 as soon as possible.”
Cases of child COVID-19 set a new 1-week record and the total number of children infected during the pandemic passed 5 million, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The nearly 282,000 new cases reported in the United States during the week ending Sept. 2 broke the record of 211,000 set in mid-January and brought the cumulative count to 5,049,465 children with COVID-19 since the pandemic began, the AAP and the CHA said in their weekly COVID report.
Hospitalizations in children aged 0-17 years have also reached record levels in recent days. The highest daily admission rate since the pandemic began, 0.51 per 100,000 population, was recorded on Sept. 2, less than 2 months after the nation saw its lowest child COVID admission rate for 1 day: 0.07 per 100,000 on July 4. That’s an increase of 629%, according to data from the Centers for Disease Control and Prevention.
Vaccinations in children, however, did not follow suit. New vaccinations in children aged 12-17 years dropped by 4.5% for the week ending Sept. 6, compared with the week before. Initiations were actually up almost 12% for children aged 16-17, but that was not enough to overcome the continued decline among 12- to 15-year-olds, the CDC said on its COVID Data Tracker.
Despite the decline in new vaccinations, those younger children passed a noteworthy group milestone: 50.9% of all 12- to 15-year-olds now have received at least one dose, with 38.6% having completed the regimen. The 16- to 17-year-olds got an earlier start and have reached 58.9% coverage for one dose and 47.6% for two, the CDC said.
A total of 12.2 million children aged 12-17 years had received at least one dose of COVID vaccine as of Sept. 6, of whom almost 9.5 million are fully vaccinated, based on the CDC data.
At the state level, Vermont has the highest rates for vaccine initiation (75%) and full vaccination (65%), with Massachusetts (75%/62%) and Connecticut (73%/59%) just behind. The other end of the scale is occupied by Wyoming (28% initiation/19% full vaccination), Alabama (32%/19%), and North Dakota (32%/23%), the AAP said in a separate report.
In a recent letter to the Food and Drug Administration, AAP President Lee Savio Beers, MD, said that the “Delta variant is surging at extremely alarming rates in every region of America. This surge is seriously impacting all populations, including children.” Dr. Beers urged the FDA to work “aggressively toward authorizing safe and effective COVID-19 vaccines for children under age 12 as soon as possible.”
Anakinra improved survival in hospitalized COVID-19 patients
Hospitalized COVID-19 patients at increased risk for respiratory failure showed significant improvement after treatment with anakinra, compared with placebo, based on data from a phase 3, randomized trial of nearly 600 patients who also received standard of care treatment.
Anakinra, a recombinant interleukin (IL)-1 receptor antagonist that blocks activity for both IL-1 alpha and beta, showed a 70% decrease in the risk of progression to severe respiratory failure in a prior open-label, phase 2, proof-of-concept study, wrote Evdoxia Kyriazopoulou, MD, PhD, of National and Kapodistrian University of Athens, and colleagues.
Previous research has shown that soluble urokinase plasminogen activator receptor (suPAR) serum levels can signal increased risk of progression to severe disease and respiratory failure in COVID-19 patients, they noted.
Supported by these early findings, “the SAVE-MORE study (suPAR-guided anakinra treatment for validation of the risk and early management of severe respiratory failure by COVID-19) is a pivotal, confirmatory, phase 3, double-blind, randomized controlled trial that evaluated the efficacy and safety of early initiation of anakinra treatment in hospitalized patients with moderate or severe COVID-19,” the researchers said.
In the SAVE-MORE study published Sept. 3 in Nature Medicine, the researchers identified 594 adults with COVID-19 who were hospitalized at 37 centers in Greece and Italy and at risk of progressing to respiratory failure based on plasma suPAR levels of at least 6 ng/mL.
The primary objective was to assess the impact of early anakinra treatment on the clinical status of COVID-19 patients at risk for severe disease according to the 11-point, ordinal World Health Organization Clinical Progression Scale (WHO-CPS) at 28 days after starting treatment. All patients received standard of care, which consisted of regular monitoring of physical signs, oximetry, and anticoagulation. Patients with severe disease by the WHO definition were also received 6 mg of dexamethasone intravenously daily for 10 days. A total of 405 were randomized to anakinra and 189 to placebo. Approximately 92% of the study participants had severe pneumonia according to the WHO classification for COVID-19. The average age of the patients was 62 years, 58% were male, and the average body mass index was 29.5 kg/m2.
At 28 days, 204 (50.4%) of the anakinra-treated patients had fully recovered, with no detectable viral RNA, compared with 50 (26.5%) of the placebo-treated patients (P < .0001). In addition, significantly fewer patients in the anakinra group had died by 28 days (13 patients, 3.2%), compared with patients in the placebo group (13 patients, 6.9%).
The median decrease in WHO-CPS scores from baseline to 28 days was 4 points in the anakinra group and 3 points in the placebo group, a statistically significant difference (P < .0001).
“Overall, the unadjusted proportional odds of having a worse score on the 11-point WHO-CPS at day 28 with anakinra was 0.36 versus placebo,” and this number remained the same in adjusted analysis, the researchers wrote.
All five secondary endpoints on the WHO-CPS showed significant benefits of anakinra, compared with placebo. These included an absolute decrease of WHO-CPS at day 28 and day 14 from baseline; an absolute decrease of Sequential Organ Failure Assessment scores at day 7 from baseline; and a significantly shorter mean time to both hospital and ICU discharge (1 day and 4 days, respectively) with anakinra versus placebo.
Follow-up laboratory data showed a significant increase in absolute lymphocyte count at 7 days, a significant decrease in circulating IL-6 levels at 4 and 7 days, and significantly decreased plasma C-reactive protein (CRP) levels at 7 days.
Serious treatment-emergent adverse events were reported in 16% with anakinra and in 21.7% with placebo; the most common of these events were infections (8.4% with anakinra and 15.9% with placebo). The next most common serious treatment-emergent adverse events were ventilator-associated pneumonia, septic shock and multiple organ dysfunction, bloodstream infections, and pulmonary embolism. The most common nonserious treatment-emergent adverse events were an increase of liver function tests and hyperglycemia (similar in anakinra and placebo groups) and nonserious anemia (lower in the anakinra group).
The study findings were limited by several factors, including the lack of patients with critical COVID-19 disease and the challenge of application of suPAR in all hospital settings, the researchers noted. However, “the results validate the findings of the previous SAVE open-label phase 2 trial,” they said. The results suggest “that suPAR should be measured upon admission of all patients with COVID-19 who do not need oxygen or who need nasal or mask oxygen, and that, if suPAR levels are 6 ng/mL or higher, anakinra treatment might be a suitable therapy,” they concluded.
Cytokine storm syndrome remains a treatment challenge
“Many who die from COVID-19 suffer hyperinflammation with features of cytokine storm syndrome (CSS) and associated acute respiratory distress syndrome,” wrote Randy Q. Cron, MD, and W. Winn Chatham, MD, of the University of Alabama at Birmingham, and Roberto Caricchio, MD, of Temple University, Philadelphia, in an accompanying editorial. They noted that the SAVE-MORE trial results contrast with another recent randomized trial of canakinumab, which failed to show notable benefits, compared with placebo, in treating hospitalized patients with COVID-19 pneumonia.
“There are some key differences between these trials, one being that anakinra blocks signaling of both IL-1 alpha and IL-1 beta, whereas canakinumab binds only IL-1 beta,” the editorialists explained. “SARS-CoV-2–infected endothelium may be a particularly important source of IL-1 alpha that is not targeted by canakinumab,” they noted.
Additional studies have examined IL-6 inhibition to treat COVID-19 patients, but data have been inconsistent, the editorialists said.
“One thing that is clearly emerging from this pandemic is that the CSS associated with COVID-19 is relatively unique, with only modestly elevated levels of IL-6, CRP, and ferritin, for example,” they noted. However, the SAVE-MORE study suggests that more targeted approaches, such as anakinra, “may allow earlier introduction of anticytokine treatment” and support the use of IL-1 blockade with anakinra for cases of severe COVID-19 pneumonia.
Predicting risk for severe disease
“One of the major challenges in the management of patients with COVID-19 is identifying patients at risk of severe disease who would warrant early intervention with anti-inflammatory therapy,” said Salim Hayek, MD, medical director of the University of Michigan’s Frankel Cardiovascular Center Clinics, in an interview. “We and others had found that soluble urokinase plasminogen activator receptor (suPAR) levels are the strongest predictor of severe disease amongst biomarkers of inflammation,” he said. “In this study, patients with high suPAR levels derived benefit from anakinra, compared to those with placebo. This study is a great example of how suPAR levels could be used to identify high-risk patients that would benefit from therapies targeting inflammation,” Dr. Hayek emphasized.
“The findings are in line with the hypothesis that patients with the highest degrees of inflammation would benefit the best from targeting the hyperinflammatory cascade using anakinra or other interleukin antagonists,” Dr. Hayek said. “Given suPAR levels are the best predictors of high-risk disease, it is not surprising to see that patients with high levels benefit from targeting inflammation,” he noted.
The take-home message for clinicians at this time is that anakinra effectively improves outcomes in COVID-19 patients with high suPAR levels, Dr. Hayek said. “SuPAR can be measured easily at the point of care. Thus, a targeted strategy using suPAR to identify patients who would benefit from anakinra appears to be viable,” he explained.
However, “Whether anakinra is effective in patients with lower suPAR levels (<6 ng/mL) is unclear and was not answered by this study,” he said. “We eagerly await results of other trials to make that determination. Whether suPAR levels can also help guide the use of other therapies for COVID-19 should be explored and would enhance the personalization of treatment for COVID-19 according to the underlying inflammatory state,” he added.
The SAVE-MORE study was funded by the Hellenic Institute for the Study of Sepsis and Sobi, which manufactures anakinra. Some of the study authors reported financial relationships with Sobi and other pharmaceutical companies.
Dr. Cron disclosed serving as a consultant to Sobi, Novartis, Pfizer, and Sironax. Dr. Cron and Dr. Chatham disclosed having received grant support from Sobi for investigator-initiated clinical trials, and Dr. Caricchio disclosed serving as a consultant to GlaxoSmithKline, Johnson & Johnson, Aurinia, and Bristol-Myers Squibb. Dr. Hayek had no relevant financial conflicts to disclose.
Hospitalized COVID-19 patients at increased risk for respiratory failure showed significant improvement after treatment with anakinra, compared with placebo, based on data from a phase 3, randomized trial of nearly 600 patients who also received standard of care treatment.
Anakinra, a recombinant interleukin (IL)-1 receptor antagonist that blocks activity for both IL-1 alpha and beta, showed a 70% decrease in the risk of progression to severe respiratory failure in a prior open-label, phase 2, proof-of-concept study, wrote Evdoxia Kyriazopoulou, MD, PhD, of National and Kapodistrian University of Athens, and colleagues.
Previous research has shown that soluble urokinase plasminogen activator receptor (suPAR) serum levels can signal increased risk of progression to severe disease and respiratory failure in COVID-19 patients, they noted.
Supported by these early findings, “the SAVE-MORE study (suPAR-guided anakinra treatment for validation of the risk and early management of severe respiratory failure by COVID-19) is a pivotal, confirmatory, phase 3, double-blind, randomized controlled trial that evaluated the efficacy and safety of early initiation of anakinra treatment in hospitalized patients with moderate or severe COVID-19,” the researchers said.
In the SAVE-MORE study published Sept. 3 in Nature Medicine, the researchers identified 594 adults with COVID-19 who were hospitalized at 37 centers in Greece and Italy and at risk of progressing to respiratory failure based on plasma suPAR levels of at least 6 ng/mL.
The primary objective was to assess the impact of early anakinra treatment on the clinical status of COVID-19 patients at risk for severe disease according to the 11-point, ordinal World Health Organization Clinical Progression Scale (WHO-CPS) at 28 days after starting treatment. All patients received standard of care, which consisted of regular monitoring of physical signs, oximetry, and anticoagulation. Patients with severe disease by the WHO definition were also received 6 mg of dexamethasone intravenously daily for 10 days. A total of 405 were randomized to anakinra and 189 to placebo. Approximately 92% of the study participants had severe pneumonia according to the WHO classification for COVID-19. The average age of the patients was 62 years, 58% were male, and the average body mass index was 29.5 kg/m2.
At 28 days, 204 (50.4%) of the anakinra-treated patients had fully recovered, with no detectable viral RNA, compared with 50 (26.5%) of the placebo-treated patients (P < .0001). In addition, significantly fewer patients in the anakinra group had died by 28 days (13 patients, 3.2%), compared with patients in the placebo group (13 patients, 6.9%).
The median decrease in WHO-CPS scores from baseline to 28 days was 4 points in the anakinra group and 3 points in the placebo group, a statistically significant difference (P < .0001).
“Overall, the unadjusted proportional odds of having a worse score on the 11-point WHO-CPS at day 28 with anakinra was 0.36 versus placebo,” and this number remained the same in adjusted analysis, the researchers wrote.
All five secondary endpoints on the WHO-CPS showed significant benefits of anakinra, compared with placebo. These included an absolute decrease of WHO-CPS at day 28 and day 14 from baseline; an absolute decrease of Sequential Organ Failure Assessment scores at day 7 from baseline; and a significantly shorter mean time to both hospital and ICU discharge (1 day and 4 days, respectively) with anakinra versus placebo.
Follow-up laboratory data showed a significant increase in absolute lymphocyte count at 7 days, a significant decrease in circulating IL-6 levels at 4 and 7 days, and significantly decreased plasma C-reactive protein (CRP) levels at 7 days.
Serious treatment-emergent adverse events were reported in 16% with anakinra and in 21.7% with placebo; the most common of these events were infections (8.4% with anakinra and 15.9% with placebo). The next most common serious treatment-emergent adverse events were ventilator-associated pneumonia, septic shock and multiple organ dysfunction, bloodstream infections, and pulmonary embolism. The most common nonserious treatment-emergent adverse events were an increase of liver function tests and hyperglycemia (similar in anakinra and placebo groups) and nonserious anemia (lower in the anakinra group).
The study findings were limited by several factors, including the lack of patients with critical COVID-19 disease and the challenge of application of suPAR in all hospital settings, the researchers noted. However, “the results validate the findings of the previous SAVE open-label phase 2 trial,” they said. The results suggest “that suPAR should be measured upon admission of all patients with COVID-19 who do not need oxygen or who need nasal or mask oxygen, and that, if suPAR levels are 6 ng/mL or higher, anakinra treatment might be a suitable therapy,” they concluded.
Cytokine storm syndrome remains a treatment challenge
“Many who die from COVID-19 suffer hyperinflammation with features of cytokine storm syndrome (CSS) and associated acute respiratory distress syndrome,” wrote Randy Q. Cron, MD, and W. Winn Chatham, MD, of the University of Alabama at Birmingham, and Roberto Caricchio, MD, of Temple University, Philadelphia, in an accompanying editorial. They noted that the SAVE-MORE trial results contrast with another recent randomized trial of canakinumab, which failed to show notable benefits, compared with placebo, in treating hospitalized patients with COVID-19 pneumonia.
“There are some key differences between these trials, one being that anakinra blocks signaling of both IL-1 alpha and IL-1 beta, whereas canakinumab binds only IL-1 beta,” the editorialists explained. “SARS-CoV-2–infected endothelium may be a particularly important source of IL-1 alpha that is not targeted by canakinumab,” they noted.
Additional studies have examined IL-6 inhibition to treat COVID-19 patients, but data have been inconsistent, the editorialists said.
“One thing that is clearly emerging from this pandemic is that the CSS associated with COVID-19 is relatively unique, with only modestly elevated levels of IL-6, CRP, and ferritin, for example,” they noted. However, the SAVE-MORE study suggests that more targeted approaches, such as anakinra, “may allow earlier introduction of anticytokine treatment” and support the use of IL-1 blockade with anakinra for cases of severe COVID-19 pneumonia.
Predicting risk for severe disease
“One of the major challenges in the management of patients with COVID-19 is identifying patients at risk of severe disease who would warrant early intervention with anti-inflammatory therapy,” said Salim Hayek, MD, medical director of the University of Michigan’s Frankel Cardiovascular Center Clinics, in an interview. “We and others had found that soluble urokinase plasminogen activator receptor (suPAR) levels are the strongest predictor of severe disease amongst biomarkers of inflammation,” he said. “In this study, patients with high suPAR levels derived benefit from anakinra, compared to those with placebo. This study is a great example of how suPAR levels could be used to identify high-risk patients that would benefit from therapies targeting inflammation,” Dr. Hayek emphasized.
“The findings are in line with the hypothesis that patients with the highest degrees of inflammation would benefit the best from targeting the hyperinflammatory cascade using anakinra or other interleukin antagonists,” Dr. Hayek said. “Given suPAR levels are the best predictors of high-risk disease, it is not surprising to see that patients with high levels benefit from targeting inflammation,” he noted.
The take-home message for clinicians at this time is that anakinra effectively improves outcomes in COVID-19 patients with high suPAR levels, Dr. Hayek said. “SuPAR can be measured easily at the point of care. Thus, a targeted strategy using suPAR to identify patients who would benefit from anakinra appears to be viable,” he explained.
However, “Whether anakinra is effective in patients with lower suPAR levels (<6 ng/mL) is unclear and was not answered by this study,” he said. “We eagerly await results of other trials to make that determination. Whether suPAR levels can also help guide the use of other therapies for COVID-19 should be explored and would enhance the personalization of treatment for COVID-19 according to the underlying inflammatory state,” he added.
The SAVE-MORE study was funded by the Hellenic Institute for the Study of Sepsis and Sobi, which manufactures anakinra. Some of the study authors reported financial relationships with Sobi and other pharmaceutical companies.
Dr. Cron disclosed serving as a consultant to Sobi, Novartis, Pfizer, and Sironax. Dr. Cron and Dr. Chatham disclosed having received grant support from Sobi for investigator-initiated clinical trials, and Dr. Caricchio disclosed serving as a consultant to GlaxoSmithKline, Johnson & Johnson, Aurinia, and Bristol-Myers Squibb. Dr. Hayek had no relevant financial conflicts to disclose.
Hospitalized COVID-19 patients at increased risk for respiratory failure showed significant improvement after treatment with anakinra, compared with placebo, based on data from a phase 3, randomized trial of nearly 600 patients who also received standard of care treatment.
Anakinra, a recombinant interleukin (IL)-1 receptor antagonist that blocks activity for both IL-1 alpha and beta, showed a 70% decrease in the risk of progression to severe respiratory failure in a prior open-label, phase 2, proof-of-concept study, wrote Evdoxia Kyriazopoulou, MD, PhD, of National and Kapodistrian University of Athens, and colleagues.
Previous research has shown that soluble urokinase plasminogen activator receptor (suPAR) serum levels can signal increased risk of progression to severe disease and respiratory failure in COVID-19 patients, they noted.
Supported by these early findings, “the SAVE-MORE study (suPAR-guided anakinra treatment for validation of the risk and early management of severe respiratory failure by COVID-19) is a pivotal, confirmatory, phase 3, double-blind, randomized controlled trial that evaluated the efficacy and safety of early initiation of anakinra treatment in hospitalized patients with moderate or severe COVID-19,” the researchers said.
In the SAVE-MORE study published Sept. 3 in Nature Medicine, the researchers identified 594 adults with COVID-19 who were hospitalized at 37 centers in Greece and Italy and at risk of progressing to respiratory failure based on plasma suPAR levels of at least 6 ng/mL.
The primary objective was to assess the impact of early anakinra treatment on the clinical status of COVID-19 patients at risk for severe disease according to the 11-point, ordinal World Health Organization Clinical Progression Scale (WHO-CPS) at 28 days after starting treatment. All patients received standard of care, which consisted of regular monitoring of physical signs, oximetry, and anticoagulation. Patients with severe disease by the WHO definition were also received 6 mg of dexamethasone intravenously daily for 10 days. A total of 405 were randomized to anakinra and 189 to placebo. Approximately 92% of the study participants had severe pneumonia according to the WHO classification for COVID-19. The average age of the patients was 62 years, 58% were male, and the average body mass index was 29.5 kg/m2.
At 28 days, 204 (50.4%) of the anakinra-treated patients had fully recovered, with no detectable viral RNA, compared with 50 (26.5%) of the placebo-treated patients (P < .0001). In addition, significantly fewer patients in the anakinra group had died by 28 days (13 patients, 3.2%), compared with patients in the placebo group (13 patients, 6.9%).
The median decrease in WHO-CPS scores from baseline to 28 days was 4 points in the anakinra group and 3 points in the placebo group, a statistically significant difference (P < .0001).
“Overall, the unadjusted proportional odds of having a worse score on the 11-point WHO-CPS at day 28 with anakinra was 0.36 versus placebo,” and this number remained the same in adjusted analysis, the researchers wrote.
All five secondary endpoints on the WHO-CPS showed significant benefits of anakinra, compared with placebo. These included an absolute decrease of WHO-CPS at day 28 and day 14 from baseline; an absolute decrease of Sequential Organ Failure Assessment scores at day 7 from baseline; and a significantly shorter mean time to both hospital and ICU discharge (1 day and 4 days, respectively) with anakinra versus placebo.
Follow-up laboratory data showed a significant increase in absolute lymphocyte count at 7 days, a significant decrease in circulating IL-6 levels at 4 and 7 days, and significantly decreased plasma C-reactive protein (CRP) levels at 7 days.
Serious treatment-emergent adverse events were reported in 16% with anakinra and in 21.7% with placebo; the most common of these events were infections (8.4% with anakinra and 15.9% with placebo). The next most common serious treatment-emergent adverse events were ventilator-associated pneumonia, septic shock and multiple organ dysfunction, bloodstream infections, and pulmonary embolism. The most common nonserious treatment-emergent adverse events were an increase of liver function tests and hyperglycemia (similar in anakinra and placebo groups) and nonserious anemia (lower in the anakinra group).
The study findings were limited by several factors, including the lack of patients with critical COVID-19 disease and the challenge of application of suPAR in all hospital settings, the researchers noted. However, “the results validate the findings of the previous SAVE open-label phase 2 trial,” they said. The results suggest “that suPAR should be measured upon admission of all patients with COVID-19 who do not need oxygen or who need nasal or mask oxygen, and that, if suPAR levels are 6 ng/mL or higher, anakinra treatment might be a suitable therapy,” they concluded.
Cytokine storm syndrome remains a treatment challenge
“Many who die from COVID-19 suffer hyperinflammation with features of cytokine storm syndrome (CSS) and associated acute respiratory distress syndrome,” wrote Randy Q. Cron, MD, and W. Winn Chatham, MD, of the University of Alabama at Birmingham, and Roberto Caricchio, MD, of Temple University, Philadelphia, in an accompanying editorial. They noted that the SAVE-MORE trial results contrast with another recent randomized trial of canakinumab, which failed to show notable benefits, compared with placebo, in treating hospitalized patients with COVID-19 pneumonia.
“There are some key differences between these trials, one being that anakinra blocks signaling of both IL-1 alpha and IL-1 beta, whereas canakinumab binds only IL-1 beta,” the editorialists explained. “SARS-CoV-2–infected endothelium may be a particularly important source of IL-1 alpha that is not targeted by canakinumab,” they noted.
Additional studies have examined IL-6 inhibition to treat COVID-19 patients, but data have been inconsistent, the editorialists said.
“One thing that is clearly emerging from this pandemic is that the CSS associated with COVID-19 is relatively unique, with only modestly elevated levels of IL-6, CRP, and ferritin, for example,” they noted. However, the SAVE-MORE study suggests that more targeted approaches, such as anakinra, “may allow earlier introduction of anticytokine treatment” and support the use of IL-1 blockade with anakinra for cases of severe COVID-19 pneumonia.
Predicting risk for severe disease
“One of the major challenges in the management of patients with COVID-19 is identifying patients at risk of severe disease who would warrant early intervention with anti-inflammatory therapy,” said Salim Hayek, MD, medical director of the University of Michigan’s Frankel Cardiovascular Center Clinics, in an interview. “We and others had found that soluble urokinase plasminogen activator receptor (suPAR) levels are the strongest predictor of severe disease amongst biomarkers of inflammation,” he said. “In this study, patients with high suPAR levels derived benefit from anakinra, compared to those with placebo. This study is a great example of how suPAR levels could be used to identify high-risk patients that would benefit from therapies targeting inflammation,” Dr. Hayek emphasized.
“The findings are in line with the hypothesis that patients with the highest degrees of inflammation would benefit the best from targeting the hyperinflammatory cascade using anakinra or other interleukin antagonists,” Dr. Hayek said. “Given suPAR levels are the best predictors of high-risk disease, it is not surprising to see that patients with high levels benefit from targeting inflammation,” he noted.
The take-home message for clinicians at this time is that anakinra effectively improves outcomes in COVID-19 patients with high suPAR levels, Dr. Hayek said. “SuPAR can be measured easily at the point of care. Thus, a targeted strategy using suPAR to identify patients who would benefit from anakinra appears to be viable,” he explained.
However, “Whether anakinra is effective in patients with lower suPAR levels (<6 ng/mL) is unclear and was not answered by this study,” he said. “We eagerly await results of other trials to make that determination. Whether suPAR levels can also help guide the use of other therapies for COVID-19 should be explored and would enhance the personalization of treatment for COVID-19 according to the underlying inflammatory state,” he added.
The SAVE-MORE study was funded by the Hellenic Institute for the Study of Sepsis and Sobi, which manufactures anakinra. Some of the study authors reported financial relationships with Sobi and other pharmaceutical companies.
Dr. Cron disclosed serving as a consultant to Sobi, Novartis, Pfizer, and Sironax. Dr. Cron and Dr. Chatham disclosed having received grant support from Sobi for investigator-initiated clinical trials, and Dr. Caricchio disclosed serving as a consultant to GlaxoSmithKline, Johnson & Johnson, Aurinia, and Bristol-Myers Squibb. Dr. Hayek had no relevant financial conflicts to disclose.
FROM NATURE MEDICINE
Large study affirms what we already know: Masks work to prevent COVID-19
It also shows that surgical masks are more effective than cloth face coverings.
The study, which was published ahead of peer review, demonstrates the power of careful investigation and offers a host of lessons about mask wearing that will be important worldwide. One key finding of the study, for example, is that wearing a mask doesn’t lead people to abandon social distancing, something public health officials had feared might happen if masks gave people a false sense of security.
“What we really were able to achieve is to demonstrate that masks are effective against COVID-19, even under a rigorous and systematic evaluation that was done in the throes of the pandemic,” said Ashley Styczynski, MD, who was an infectious disease fellow at Stanford (Calif.) University when she collaborated on the study with other colleagues at Stanford, Yale, and Innovations for Poverty Action, a large research and policy nonprofit organization that currently works in 22 countries.
“And so, I think people who have been holding out on wearing masks because [they] felt like there wasn’t enough evidence for it, we’re hoping this will really help bridge that gap for them,” she said.
It included more than 600 unions – or local governmental districts in Bangladesh – and roughly 340,000 people.
Half of the districts were given cloth or surgical face masks along with continual reminders to wear them properly; the other half were tracked with no intervention. Blood tests of people who developed symptoms during the study verified their infections.
Compared to villages that didn’t mask, those in which masks of any type were worn had about 9% fewer symptomatic cases of COVID-19. The finding was statistically significant and was unlikely to have occurred by chance alone.
“Somebody could read this study and say, ‘OK, you reduced COVID-19 by 9%. Big deal.’ And what I would respond to that would be that, if anything, we think that that is a substantial underestimate,” Dr. Styczynski said.
One reason they think they underestimated the effectiveness of masks is that they tested only people who were having symptoms, so people who had only very mild or asymptomatic infections were missed.
Another reason is that, among people who had symptoms, only one-third agreed to undergo a blood test. The effect may have been bigger had participation been universal.
Local transmission may have played a role, too. Rates of COVID-19 in Bangladesh were relatively low during the study. Most infections were caused by the B.1.1.7, or Alpha, variant.
Since then, Delta has taken over. Delta is thought to be more transmissible, and some studies have suggested that people infected with Delta shed more viral particles. Masks may be more effective when more virus is circulating.
The investigators also found important differences by age and by the type of mask. Villages in which surgical masks were worn had 11% fewer COVID-19 cases than villages in which masks were not worn. In villages in which cloth masks were worn, on the other hand, infections were reduced by only 5%.
The cloth masks were substantial. Each had three layers – two layers of fabric with an outer layer of polypropylene. On testing, the filtration efficiency of the cloth masks was only about 37%, compared with 95% for the three-layer surgical masks, which were also made of polypropylene.
Masks were most effective for older individuals. People aged 50-60 years who wore surgical masks were 23% less likely to test positive for COVID, compared with their peers who didn’t wear masks. For people older than 60, the reduction in risk was greater – 35%.
Rigorous research
The study took place over a period of 8 weeks in each district. The interventions were rolled out in waves, with the first starting in November 2020 and the last in January 2021.
Investigators gave each household free cloth or surgical face masks and showed families a video about proper mask wearing with promotional messages from the prime minister, a head imam, and a national cricket star. They also handed out free masks.
Previous studies have shown that people aren’t always truthful about wearing masks in public. In Kenya, for example, 88% of people answering a phone survey said that they wore masks regularly, but researchers determined that only 10% of them actually did so.
Investigators in the Bangladesh study didn’t just ask people if they’d worn masks, they stationed themselves in public markets, mosques, tea stalls, and on roads that were the main entrances to the villages and took notes.
They also tested various ways to educate people and to remind them to wear masks. They found that four factors were effective at promoting the wearing of masks, and they gave them an acronym – NORM.
- N for no-cost masks.
- O for offering information through the video and local leaders.
- R for regular reminders to people by investigators who stand in public markets and offer masks or encourage anyone who wasn’t wearing one or wasn’t wearing it correctly.
- M for modeling, in which local leaders, such as imams, wear masks and remind their followers to wear them.
These four measures tripled the wearing of masks in the intervention communities, from a baseline level of 13% to 42%. People continued to wear their masks properly for about 2 weeks after the study ended, indicating that they’d gotten used to wearing them.
Dr. Styczynski said that nothing else – not text message reminders, or signs posted in public places, or local incentives – moved the needle on mask wearing.
Saved lives and money
The study found that the strategy was cost effective, too. Giving masks to a large population and getting people to use them costs about $10,000 per life saved from COVID, on par with the cost of deploying mosquito nets to save people from malaria, Dr. Styczynski said.
“I think that what we’ve been able to show is that this is a really important tool to be used globally, especially as countries have delays in getting access to vaccines and rolling them out,” she said.
Dr. Styczynski said masks will continue to be important even in countries such as the United States, where vaccines aren’t stopping transmission 100% and there are still large portions of the population who are unvaccinated, such as children.
“If we want to reduce COVID-19 here, it’s really important that we consider the ongoing utility of masks, in addition to vaccines, and not really thinking of them as one or the other,” she said.
The study was funded by a grant from GiveWell.org. The funder had no role in the study design, interpretation, or the decision to publish.
A version of this article first appeared on Medscape.com.
It also shows that surgical masks are more effective than cloth face coverings.
The study, which was published ahead of peer review, demonstrates the power of careful investigation and offers a host of lessons about mask wearing that will be important worldwide. One key finding of the study, for example, is that wearing a mask doesn’t lead people to abandon social distancing, something public health officials had feared might happen if masks gave people a false sense of security.
“What we really were able to achieve is to demonstrate that masks are effective against COVID-19, even under a rigorous and systematic evaluation that was done in the throes of the pandemic,” said Ashley Styczynski, MD, who was an infectious disease fellow at Stanford (Calif.) University when she collaborated on the study with other colleagues at Stanford, Yale, and Innovations for Poverty Action, a large research and policy nonprofit organization that currently works in 22 countries.
“And so, I think people who have been holding out on wearing masks because [they] felt like there wasn’t enough evidence for it, we’re hoping this will really help bridge that gap for them,” she said.
It included more than 600 unions – or local governmental districts in Bangladesh – and roughly 340,000 people.
Half of the districts were given cloth or surgical face masks along with continual reminders to wear them properly; the other half were tracked with no intervention. Blood tests of people who developed symptoms during the study verified their infections.
Compared to villages that didn’t mask, those in which masks of any type were worn had about 9% fewer symptomatic cases of COVID-19. The finding was statistically significant and was unlikely to have occurred by chance alone.
“Somebody could read this study and say, ‘OK, you reduced COVID-19 by 9%. Big deal.’ And what I would respond to that would be that, if anything, we think that that is a substantial underestimate,” Dr. Styczynski said.
One reason they think they underestimated the effectiveness of masks is that they tested only people who were having symptoms, so people who had only very mild or asymptomatic infections were missed.
Another reason is that, among people who had symptoms, only one-third agreed to undergo a blood test. The effect may have been bigger had participation been universal.
Local transmission may have played a role, too. Rates of COVID-19 in Bangladesh were relatively low during the study. Most infections were caused by the B.1.1.7, or Alpha, variant.
Since then, Delta has taken over. Delta is thought to be more transmissible, and some studies have suggested that people infected with Delta shed more viral particles. Masks may be more effective when more virus is circulating.
The investigators also found important differences by age and by the type of mask. Villages in which surgical masks were worn had 11% fewer COVID-19 cases than villages in which masks were not worn. In villages in which cloth masks were worn, on the other hand, infections were reduced by only 5%.
The cloth masks were substantial. Each had three layers – two layers of fabric with an outer layer of polypropylene. On testing, the filtration efficiency of the cloth masks was only about 37%, compared with 95% for the three-layer surgical masks, which were also made of polypropylene.
Masks were most effective for older individuals. People aged 50-60 years who wore surgical masks were 23% less likely to test positive for COVID, compared with their peers who didn’t wear masks. For people older than 60, the reduction in risk was greater – 35%.
Rigorous research
The study took place over a period of 8 weeks in each district. The interventions were rolled out in waves, with the first starting in November 2020 and the last in January 2021.
Investigators gave each household free cloth or surgical face masks and showed families a video about proper mask wearing with promotional messages from the prime minister, a head imam, and a national cricket star. They also handed out free masks.
Previous studies have shown that people aren’t always truthful about wearing masks in public. In Kenya, for example, 88% of people answering a phone survey said that they wore masks regularly, but researchers determined that only 10% of them actually did so.
Investigators in the Bangladesh study didn’t just ask people if they’d worn masks, they stationed themselves in public markets, mosques, tea stalls, and on roads that were the main entrances to the villages and took notes.
They also tested various ways to educate people and to remind them to wear masks. They found that four factors were effective at promoting the wearing of masks, and they gave them an acronym – NORM.
- N for no-cost masks.
- O for offering information through the video and local leaders.
- R for regular reminders to people by investigators who stand in public markets and offer masks or encourage anyone who wasn’t wearing one or wasn’t wearing it correctly.
- M for modeling, in which local leaders, such as imams, wear masks and remind their followers to wear them.
These four measures tripled the wearing of masks in the intervention communities, from a baseline level of 13% to 42%. People continued to wear their masks properly for about 2 weeks after the study ended, indicating that they’d gotten used to wearing them.
Dr. Styczynski said that nothing else – not text message reminders, or signs posted in public places, or local incentives – moved the needle on mask wearing.
Saved lives and money
The study found that the strategy was cost effective, too. Giving masks to a large population and getting people to use them costs about $10,000 per life saved from COVID, on par with the cost of deploying mosquito nets to save people from malaria, Dr. Styczynski said.
“I think that what we’ve been able to show is that this is a really important tool to be used globally, especially as countries have delays in getting access to vaccines and rolling them out,” she said.
Dr. Styczynski said masks will continue to be important even in countries such as the United States, where vaccines aren’t stopping transmission 100% and there are still large portions of the population who are unvaccinated, such as children.
“If we want to reduce COVID-19 here, it’s really important that we consider the ongoing utility of masks, in addition to vaccines, and not really thinking of them as one or the other,” she said.
The study was funded by a grant from GiveWell.org. The funder had no role in the study design, interpretation, or the decision to publish.
A version of this article first appeared on Medscape.com.
It also shows that surgical masks are more effective than cloth face coverings.
The study, which was published ahead of peer review, demonstrates the power of careful investigation and offers a host of lessons about mask wearing that will be important worldwide. One key finding of the study, for example, is that wearing a mask doesn’t lead people to abandon social distancing, something public health officials had feared might happen if masks gave people a false sense of security.
“What we really were able to achieve is to demonstrate that masks are effective against COVID-19, even under a rigorous and systematic evaluation that was done in the throes of the pandemic,” said Ashley Styczynski, MD, who was an infectious disease fellow at Stanford (Calif.) University when she collaborated on the study with other colleagues at Stanford, Yale, and Innovations for Poverty Action, a large research and policy nonprofit organization that currently works in 22 countries.
“And so, I think people who have been holding out on wearing masks because [they] felt like there wasn’t enough evidence for it, we’re hoping this will really help bridge that gap for them,” she said.
It included more than 600 unions – or local governmental districts in Bangladesh – and roughly 340,000 people.
Half of the districts were given cloth or surgical face masks along with continual reminders to wear them properly; the other half were tracked with no intervention. Blood tests of people who developed symptoms during the study verified their infections.
Compared to villages that didn’t mask, those in which masks of any type were worn had about 9% fewer symptomatic cases of COVID-19. The finding was statistically significant and was unlikely to have occurred by chance alone.
“Somebody could read this study and say, ‘OK, you reduced COVID-19 by 9%. Big deal.’ And what I would respond to that would be that, if anything, we think that that is a substantial underestimate,” Dr. Styczynski said.
One reason they think they underestimated the effectiveness of masks is that they tested only people who were having symptoms, so people who had only very mild or asymptomatic infections were missed.
Another reason is that, among people who had symptoms, only one-third agreed to undergo a blood test. The effect may have been bigger had participation been universal.
Local transmission may have played a role, too. Rates of COVID-19 in Bangladesh were relatively low during the study. Most infections were caused by the B.1.1.7, or Alpha, variant.
Since then, Delta has taken over. Delta is thought to be more transmissible, and some studies have suggested that people infected with Delta shed more viral particles. Masks may be more effective when more virus is circulating.
The investigators also found important differences by age and by the type of mask. Villages in which surgical masks were worn had 11% fewer COVID-19 cases than villages in which masks were not worn. In villages in which cloth masks were worn, on the other hand, infections were reduced by only 5%.
The cloth masks were substantial. Each had three layers – two layers of fabric with an outer layer of polypropylene. On testing, the filtration efficiency of the cloth masks was only about 37%, compared with 95% for the three-layer surgical masks, which were also made of polypropylene.
Masks were most effective for older individuals. People aged 50-60 years who wore surgical masks were 23% less likely to test positive for COVID, compared with their peers who didn’t wear masks. For people older than 60, the reduction in risk was greater – 35%.
Rigorous research
The study took place over a period of 8 weeks in each district. The interventions were rolled out in waves, with the first starting in November 2020 and the last in January 2021.
Investigators gave each household free cloth or surgical face masks and showed families a video about proper mask wearing with promotional messages from the prime minister, a head imam, and a national cricket star. They also handed out free masks.
Previous studies have shown that people aren’t always truthful about wearing masks in public. In Kenya, for example, 88% of people answering a phone survey said that they wore masks regularly, but researchers determined that only 10% of them actually did so.
Investigators in the Bangladesh study didn’t just ask people if they’d worn masks, they stationed themselves in public markets, mosques, tea stalls, and on roads that were the main entrances to the villages and took notes.
They also tested various ways to educate people and to remind them to wear masks. They found that four factors were effective at promoting the wearing of masks, and they gave them an acronym – NORM.
- N for no-cost masks.
- O for offering information through the video and local leaders.
- R for regular reminders to people by investigators who stand in public markets and offer masks or encourage anyone who wasn’t wearing one or wasn’t wearing it correctly.
- M for modeling, in which local leaders, such as imams, wear masks and remind their followers to wear them.
These four measures tripled the wearing of masks in the intervention communities, from a baseline level of 13% to 42%. People continued to wear their masks properly for about 2 weeks after the study ended, indicating that they’d gotten used to wearing them.
Dr. Styczynski said that nothing else – not text message reminders, or signs posted in public places, or local incentives – moved the needle on mask wearing.
Saved lives and money
The study found that the strategy was cost effective, too. Giving masks to a large population and getting people to use them costs about $10,000 per life saved from COVID, on par with the cost of deploying mosquito nets to save people from malaria, Dr. Styczynski said.
“I think that what we’ve been able to show is that this is a really important tool to be used globally, especially as countries have delays in getting access to vaccines and rolling them out,” she said.
Dr. Styczynski said masks will continue to be important even in countries such as the United States, where vaccines aren’t stopping transmission 100% and there are still large portions of the population who are unvaccinated, such as children.
“If we want to reduce COVID-19 here, it’s really important that we consider the ongoing utility of masks, in addition to vaccines, and not really thinking of them as one or the other,” she said.
The study was funded by a grant from GiveWell.org. The funder had no role in the study design, interpretation, or the decision to publish.
A version of this article first appeared on Medscape.com.
Health care–associated infections spiked in 2020 in U.S. hospitals
Several health care-associated infections in U.S. hospitals spiked in 2020 compared to the previous year, according to a Centers for Disease Control and Prevention analysis published Sept. 2 in Infection Control and Hospital Epidemiology. Soaring hospitalization rates, sicker patients who required more frequent and intense care, and staffing and supply shortages caused by the COVID-19 pandemic are thought to have contributed to this increase.
This is the first increase in health care–associated infections since 2015.
These findings “are a reflection of the enormous stress that COVID has placed on our health care system,” Arjun Srinivasan, MD (Capt, USPHS), the associate director of the CDC’s Health care-Associated Infection Prevention Programs, Atlanta, told this news organization. He was not an author of the article, but he supervised the research. “We don’t want anyone to read this report and think that it represents a failure of the individual provider or a failure of health care providers in this country in their care of COVID patients,” he said. He noted that health care professionals have provided “tremendously good care to patients under extremely difficult circumstances.”
“People don’t fail – systems fail – and that’s what happened here,” he said. “Our systems that we need to have in place to prevent health care–associated infection simply were not as strong as they needed to be to survive this challenge.”
In the study, researchers used data reported to the National Healthcare Safety Network, the CDC’s tracking system for health care–associated infections. The team compared national standard infection ratios – calculated by dividing the number of reported infections by the number of predicted infections – between 2019 and 2020 for six routinely tracked events:
- Central line–associated bloodstream infections.
- Catheter-associated urinary tract infections (CAUTIs).
- Ventilator-associated events (VAEs).
- Infections associated with colon surgery and abdominal hysterectomy.
- Clostridioides difficile infections.
- Methicillin-resistant Staphylococcus aureus (MRSA) infections.
Infections were estimated using regression models created with baseline data from 2015.
“The new report highlights the need for health care facilities to strengthen their infection prevention programs and support them with adequate resources so that they can handle emerging threats to public health, while at the same time ensuring that gains made in combating HAIs [health care–associated infections] are not lost,” said the Association for Professionals in Infection Control and Epidemiology in a statement.
The analysis revealed significant national increases in central line–associated bloodstream infections, CAUTIs, VAEs, and MRSA infections in 2020 compared to 2019. Among all infection types, the greatest increase was in central-line infections, which were 46% to 47% higher in the third quarter and fourth quarter (Q4) of 2020 relative to the same periods the previous year. VAEs rose by 45%, MRSA infections increased by 34%, and CAUTIs increased by 19% in Q4 of 2020 compared to 2019.
The influx of sicker patients in hospitals throughout 2020 led to more frequent and longer use of medical devices such as catheters and ventilators. The use of these devices increases risk for infection, David P. Calfee, MD, chief medical epidemiologist at the New York–Presbyterian/Weill Cornell Medical Center, said in an interview. He is an editor of Infection Control and Hospital Epidemiology and was not involved with the study. Shortages in personal protective equipment and crowded intensive care units could also have affected how care was delivered, he said. These factors could have led to “reductions in the ability to provide some of the types of care that are needed to optimally reduce the risk of infection.”
There was either no change or decreases in infections associated with colon surgery or abdominal hysterectomy, likely because there were fewer elective surgeries performed, said Dr. Srinivasan. C. difficile–associated infections also decreased throughout 2020 compared to the previous year. Common practices to prevent the spread of COVID-19 in hospitals, such as environmental cleaning, use of personal protective equipment, and patient isolation, likely helped to curb the spread of C. difficile. Although these mitigating procedures do help protect against MRSA infection, many other factors, notably, the use of medical devices such as ventilators and catheters, can increase the risk for MRSA infection, Dr. Srinivasan added.
Although more research is needed to identify the reasons for these spikes in infection, the findings help quantify the scope of these increases across the United States, Dr. Calfee said. The data allow hospitals and health care professionals to “look back at what we did and then think forward in terms of what we can do different in the future,” he added, “so that these stresses to the system have less of an impact on how we are able to provide care.”
Dr. Srinivasan and Dr. Calfee report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Several health care-associated infections in U.S. hospitals spiked in 2020 compared to the previous year, according to a Centers for Disease Control and Prevention analysis published Sept. 2 in Infection Control and Hospital Epidemiology. Soaring hospitalization rates, sicker patients who required more frequent and intense care, and staffing and supply shortages caused by the COVID-19 pandemic are thought to have contributed to this increase.
This is the first increase in health care–associated infections since 2015.
These findings “are a reflection of the enormous stress that COVID has placed on our health care system,” Arjun Srinivasan, MD (Capt, USPHS), the associate director of the CDC’s Health care-Associated Infection Prevention Programs, Atlanta, told this news organization. He was not an author of the article, but he supervised the research. “We don’t want anyone to read this report and think that it represents a failure of the individual provider or a failure of health care providers in this country in their care of COVID patients,” he said. He noted that health care professionals have provided “tremendously good care to patients under extremely difficult circumstances.”
“People don’t fail – systems fail – and that’s what happened here,” he said. “Our systems that we need to have in place to prevent health care–associated infection simply were not as strong as they needed to be to survive this challenge.”
In the study, researchers used data reported to the National Healthcare Safety Network, the CDC’s tracking system for health care–associated infections. The team compared national standard infection ratios – calculated by dividing the number of reported infections by the number of predicted infections – between 2019 and 2020 for six routinely tracked events:
- Central line–associated bloodstream infections.
- Catheter-associated urinary tract infections (CAUTIs).
- Ventilator-associated events (VAEs).
- Infections associated with colon surgery and abdominal hysterectomy.
- Clostridioides difficile infections.
- Methicillin-resistant Staphylococcus aureus (MRSA) infections.
Infections were estimated using regression models created with baseline data from 2015.
“The new report highlights the need for health care facilities to strengthen their infection prevention programs and support them with adequate resources so that they can handle emerging threats to public health, while at the same time ensuring that gains made in combating HAIs [health care–associated infections] are not lost,” said the Association for Professionals in Infection Control and Epidemiology in a statement.
The analysis revealed significant national increases in central line–associated bloodstream infections, CAUTIs, VAEs, and MRSA infections in 2020 compared to 2019. Among all infection types, the greatest increase was in central-line infections, which were 46% to 47% higher in the third quarter and fourth quarter (Q4) of 2020 relative to the same periods the previous year. VAEs rose by 45%, MRSA infections increased by 34%, and CAUTIs increased by 19% in Q4 of 2020 compared to 2019.
The influx of sicker patients in hospitals throughout 2020 led to more frequent and longer use of medical devices such as catheters and ventilators. The use of these devices increases risk for infection, David P. Calfee, MD, chief medical epidemiologist at the New York–Presbyterian/Weill Cornell Medical Center, said in an interview. He is an editor of Infection Control and Hospital Epidemiology and was not involved with the study. Shortages in personal protective equipment and crowded intensive care units could also have affected how care was delivered, he said. These factors could have led to “reductions in the ability to provide some of the types of care that are needed to optimally reduce the risk of infection.”
There was either no change or decreases in infections associated with colon surgery or abdominal hysterectomy, likely because there were fewer elective surgeries performed, said Dr. Srinivasan. C. difficile–associated infections also decreased throughout 2020 compared to the previous year. Common practices to prevent the spread of COVID-19 in hospitals, such as environmental cleaning, use of personal protective equipment, and patient isolation, likely helped to curb the spread of C. difficile. Although these mitigating procedures do help protect against MRSA infection, many other factors, notably, the use of medical devices such as ventilators and catheters, can increase the risk for MRSA infection, Dr. Srinivasan added.
Although more research is needed to identify the reasons for these spikes in infection, the findings help quantify the scope of these increases across the United States, Dr. Calfee said. The data allow hospitals and health care professionals to “look back at what we did and then think forward in terms of what we can do different in the future,” he added, “so that these stresses to the system have less of an impact on how we are able to provide care.”
Dr. Srinivasan and Dr. Calfee report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Several health care-associated infections in U.S. hospitals spiked in 2020 compared to the previous year, according to a Centers for Disease Control and Prevention analysis published Sept. 2 in Infection Control and Hospital Epidemiology. Soaring hospitalization rates, sicker patients who required more frequent and intense care, and staffing and supply shortages caused by the COVID-19 pandemic are thought to have contributed to this increase.
This is the first increase in health care–associated infections since 2015.
These findings “are a reflection of the enormous stress that COVID has placed on our health care system,” Arjun Srinivasan, MD (Capt, USPHS), the associate director of the CDC’s Health care-Associated Infection Prevention Programs, Atlanta, told this news organization. He was not an author of the article, but he supervised the research. “We don’t want anyone to read this report and think that it represents a failure of the individual provider or a failure of health care providers in this country in their care of COVID patients,” he said. He noted that health care professionals have provided “tremendously good care to patients under extremely difficult circumstances.”
“People don’t fail – systems fail – and that’s what happened here,” he said. “Our systems that we need to have in place to prevent health care–associated infection simply were not as strong as they needed to be to survive this challenge.”
In the study, researchers used data reported to the National Healthcare Safety Network, the CDC’s tracking system for health care–associated infections. The team compared national standard infection ratios – calculated by dividing the number of reported infections by the number of predicted infections – between 2019 and 2020 for six routinely tracked events:
- Central line–associated bloodstream infections.
- Catheter-associated urinary tract infections (CAUTIs).
- Ventilator-associated events (VAEs).
- Infections associated with colon surgery and abdominal hysterectomy.
- Clostridioides difficile infections.
- Methicillin-resistant Staphylococcus aureus (MRSA) infections.
Infections were estimated using regression models created with baseline data from 2015.
“The new report highlights the need for health care facilities to strengthen their infection prevention programs and support them with adequate resources so that they can handle emerging threats to public health, while at the same time ensuring that gains made in combating HAIs [health care–associated infections] are not lost,” said the Association for Professionals in Infection Control and Epidemiology in a statement.
The analysis revealed significant national increases in central line–associated bloodstream infections, CAUTIs, VAEs, and MRSA infections in 2020 compared to 2019. Among all infection types, the greatest increase was in central-line infections, which were 46% to 47% higher in the third quarter and fourth quarter (Q4) of 2020 relative to the same periods the previous year. VAEs rose by 45%, MRSA infections increased by 34%, and CAUTIs increased by 19% in Q4 of 2020 compared to 2019.
The influx of sicker patients in hospitals throughout 2020 led to more frequent and longer use of medical devices such as catheters and ventilators. The use of these devices increases risk for infection, David P. Calfee, MD, chief medical epidemiologist at the New York–Presbyterian/Weill Cornell Medical Center, said in an interview. He is an editor of Infection Control and Hospital Epidemiology and was not involved with the study. Shortages in personal protective equipment and crowded intensive care units could also have affected how care was delivered, he said. These factors could have led to “reductions in the ability to provide some of the types of care that are needed to optimally reduce the risk of infection.”
There was either no change or decreases in infections associated with colon surgery or abdominal hysterectomy, likely because there were fewer elective surgeries performed, said Dr. Srinivasan. C. difficile–associated infections also decreased throughout 2020 compared to the previous year. Common practices to prevent the spread of COVID-19 in hospitals, such as environmental cleaning, use of personal protective equipment, and patient isolation, likely helped to curb the spread of C. difficile. Although these mitigating procedures do help protect against MRSA infection, many other factors, notably, the use of medical devices such as ventilators and catheters, can increase the risk for MRSA infection, Dr. Srinivasan added.
Although more research is needed to identify the reasons for these spikes in infection, the findings help quantify the scope of these increases across the United States, Dr. Calfee said. The data allow hospitals and health care professionals to “look back at what we did and then think forward in terms of what we can do different in the future,” he added, “so that these stresses to the system have less of an impact on how we are able to provide care.”
Dr. Srinivasan and Dr. Calfee report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
NIH on HIV vaccine failure: ‘Get your HIV-negative, at-risk patients on PrEP tomorrow’
Last year, Katherine Gill, MBChB, an HIV prevention researcher in Cape Town, South Africa, realized how jaded she’d become to vaccine research when the Pfizer COVID-19 vaccine came back as 95% effective. In her career conducting HIV clinical trials, she had never seen anything like it. Even HIV prevention methods she had studied that had worked, such as the dapivirine ring, had an overall efficacy of 30%.
The COVID-19 success story started to soften her views toward another vaccine trial she was helping to conduct, a trial in HIV that used the same platform as Johnson & Johnson’s successful COVID-19 vaccine.
“When the COVID vaccine was cracked so quickly and seemingly quite easily, I did start to think, ‘Well, maybe … maybe this will work for HIV,’ “ she said in an interview.
That turned out to be false hope. The National Institutes of Health (NIH) announced that the trial Dr. Gill was helping to conduct, HVTN 705, was stopping early because it hadn’t generated enough of an immune response in participants to justify continuing. It was the second HIV vaccine to fail in the last year. It’s also the latest in what has been a litany of disappointments in the attempt to boost the human immune system to fight HIV without the need for ongoing HIV treatment.
In HVTN 705, known as the Imbokodo study (imbokodo is a Zulu word that’s part of a saying about women being strong as rocks), researchers used the platform made up of a common cold virus, adenovirus 26, to deliver a computer-generated mosaic of HIV antigens to participants’ immune systems. That mosaic of antigens is meant to goose the immune system into recognizing HIV if it were exposed to it.
When HIV enters the body, it infiltrates immune cells and replicates within them. To the rest of the immune system, those cells still register as just typical T-cells. The rest of the immune system can’t see that the virus is spreading through the very cells meant to protect the body from illness. That, plus the armor of sugary glycoproteins encasing the virus, has made HIV nearly impervious to vaccination.
Then, those so-called “prime” shots were followed by a second shot that targets glycoprotein 140, on the most common HIV subtype (or clade) in Africa, clade C. In the Imbokodo trial, a total of 2,637 women from five sub-Saharan African countries received shots at baseline, 3 months, 6 months, and 1 year. Then researchers followed the women from month 7 to 2 years after their third dose, testing their blood to see if their immune systems had generated the immune response the vaccine was meant to induce – and whether such immune response was associated with lower rates of HIV.
When researchers looked at the first 2 years of data, they learned that the vaccine was safe. And they found a total of 114 new cases of HIV – 51 among women who received the vaccine and 63 among those who received a placebo. That’s a 25.2% efficacy rate – but it wasn’t statistically significant.
The results are frustrating, said Carl Dieffenbach, PhD, director of the AIDS division at the National Institute of Allergy and Infectious Diseases (NIAID). NIAID is one of the funders of the study.
“This [trial] is a little more confounding, in that there is this low level of statistically insignificant difference between vaccine and placebo that starts somewhere around month 9 and then just kind of indolently is maintained over the next 15 months,” he said in an interview. “That’s kind of frustrating. Does it mean there’s a signal or is this just chance? Because that’s what statistics tell us, not to believe your last data point.”
What this means for the future direction of vaccine research is unclear. A sister study to Imbokodo, called Mosaico, recently finished enrolling participants. Mosaico uses the same adenovirus 26 platform, but it’s loaded with different antigens and targets a different glycoprotein for a different HIV subtype. If that trial shows success, it could mean that the platform is right, but the targets in the Imbokodo vaccine were wrong.
Dr. Dieffenbach said that before NIAID decides what to do with Mosaico they’ve asked researchers to analyze the data on the people who did respond, to see if those people have some specific variant of HIV or some other biomarker that could be used to form the next iteration of an HIV vaccine candidate. Only after that will they make a decision about Mosaico.
But he added that it does make him wonder if vaccine approaches that rely on nonneutralizing antibodies like this one have a ceiling of effectiveness that’s just too low to alter the course of the epidemic.
“I think we’ve discovered that there’s not a floor to [these nonneutralizing approaches], but there probably is a ceiling,” he said. “I don’t know if we’re going to get better than” a 25%-29% efficacy rate with those approaches.
The Imbokodo findings reminded Mitchell Warren, executive director of the global HIV prevention nonprofit, AVAC, of the data released in January from the Antibody Mediated Prevention (AMP) trial. That trial pitted the broadly neutralizing antibody (bNAb) VCR01 against HIV – and mostly, it lost.
VCR01 worked only on HIV variants that 30% of participants had. But in those 30%, it was 75% effective at preventing HIV. Now you have Imbokodo, with its potential 25% activity against HIV, something that may have been a fluke. This, to Mr. Warren, requires a rethinking of the whole HIV vaccine enterprise while “doubling, tripling, quadrupling down” on the HIV prevention methods we know work, such as preexposure prophylaxis (PrEP).
Dr. Dieffenbach agreed. To clinicians, Dr. Dieffenbach said the message of this HIV vaccine trial is flush with urgency: “Get your HIV-negative, at-risk people on PrEP tomorrow.”
There are now two pills approved for HIV prevention, both of which have been found to be up to 99% effective when taken consistently. A third option, injectable cabotegravir (Vocabria), has been submitted to the Food and Drug Administration for approval. The federal Ready, Set, PrEP program makes the pill available for free for those who qualify, and recently the Biden administration reaffirmed that, under the Affordable Care Act, insurance companies should cover all costs associated with PrEP, including lab work and exam visits.
But for the 157 women who participated in the trial at Dr. Gill’s site in Masiphumelele, on the southwestern tip of South Africa, the trial was personal, said Jason Naidoo, community liaison officer at the Desmond Tutu HIV Foundation, which conducted a portion of the study. These were women whose parents, siblings, or children were living with HIV or had died from AIDS-defining illnesses, he said. Their lives were chaotic, traveling at a moment’s notice to hometowns on the Eastern Cape, an 11-hour car ride away – longer by bus – for traditional prayers, funerals, and other important events.
Mr. Naidoo remembers arranging buses for the women to return for scheduled clinic visits, leaving the Eastern Cape in the afternoon and arriving in Masiphumelele in the early morning hours, just to keep the clinic appointment. Then, they’d turn around and return east.
They did this for 3 years, he said.
“The fact that these participants have stuck to this and been dedicated amidst all of the chaos talks about their commitment to actually having a vaccine for HIV,” he said. “They know their own risk profile as young Black women in South Africa, and they understand the need for an intervention for the future generations.
“So you can understand the emotion and the sense of sadness, the disappointment – the incredible [dis]belief that this [the failure of the vaccine] could have happened, because the expectations are so, so high.”
For Dr. Gill, who is lead investigator for Imbokodo in Masiphumelele, the weariness toward vaccines is back. Another trial is underway for an HIV vaccine with a platform that was successful in COVID-19 – using messenger RNA (mRNA), like the Pfizer and Moderna COVID-19 vaccines did.
“I think we need to be careful,” she said, “thinking that the mRNA vaccines are going to crack it.”
Dr. Dieffenbach, Dr. Gill, and Mr. Naidoo have disclosed no relevant financial relationships. The study was funded by Janssen, a Johnson & Johnson company, with NIAID and the Bill and Melinda Gates Foundation.
A version of this article first appeared on Medscape.com.
Last year, Katherine Gill, MBChB, an HIV prevention researcher in Cape Town, South Africa, realized how jaded she’d become to vaccine research when the Pfizer COVID-19 vaccine came back as 95% effective. In her career conducting HIV clinical trials, she had never seen anything like it. Even HIV prevention methods she had studied that had worked, such as the dapivirine ring, had an overall efficacy of 30%.
The COVID-19 success story started to soften her views toward another vaccine trial she was helping to conduct, a trial in HIV that used the same platform as Johnson & Johnson’s successful COVID-19 vaccine.
“When the COVID vaccine was cracked so quickly and seemingly quite easily, I did start to think, ‘Well, maybe … maybe this will work for HIV,’ “ she said in an interview.
That turned out to be false hope. The National Institutes of Health (NIH) announced that the trial Dr. Gill was helping to conduct, HVTN 705, was stopping early because it hadn’t generated enough of an immune response in participants to justify continuing. It was the second HIV vaccine to fail in the last year. It’s also the latest in what has been a litany of disappointments in the attempt to boost the human immune system to fight HIV without the need for ongoing HIV treatment.
In HVTN 705, known as the Imbokodo study (imbokodo is a Zulu word that’s part of a saying about women being strong as rocks), researchers used the platform made up of a common cold virus, adenovirus 26, to deliver a computer-generated mosaic of HIV antigens to participants’ immune systems. That mosaic of antigens is meant to goose the immune system into recognizing HIV if it were exposed to it.
When HIV enters the body, it infiltrates immune cells and replicates within them. To the rest of the immune system, those cells still register as just typical T-cells. The rest of the immune system can’t see that the virus is spreading through the very cells meant to protect the body from illness. That, plus the armor of sugary glycoproteins encasing the virus, has made HIV nearly impervious to vaccination.
Then, those so-called “prime” shots were followed by a second shot that targets glycoprotein 140, on the most common HIV subtype (or clade) in Africa, clade C. In the Imbokodo trial, a total of 2,637 women from five sub-Saharan African countries received shots at baseline, 3 months, 6 months, and 1 year. Then researchers followed the women from month 7 to 2 years after their third dose, testing their blood to see if their immune systems had generated the immune response the vaccine was meant to induce – and whether such immune response was associated with lower rates of HIV.
When researchers looked at the first 2 years of data, they learned that the vaccine was safe. And they found a total of 114 new cases of HIV – 51 among women who received the vaccine and 63 among those who received a placebo. That’s a 25.2% efficacy rate – but it wasn’t statistically significant.
The results are frustrating, said Carl Dieffenbach, PhD, director of the AIDS division at the National Institute of Allergy and Infectious Diseases (NIAID). NIAID is one of the funders of the study.
“This [trial] is a little more confounding, in that there is this low level of statistically insignificant difference between vaccine and placebo that starts somewhere around month 9 and then just kind of indolently is maintained over the next 15 months,” he said in an interview. “That’s kind of frustrating. Does it mean there’s a signal or is this just chance? Because that’s what statistics tell us, not to believe your last data point.”
What this means for the future direction of vaccine research is unclear. A sister study to Imbokodo, called Mosaico, recently finished enrolling participants. Mosaico uses the same adenovirus 26 platform, but it’s loaded with different antigens and targets a different glycoprotein for a different HIV subtype. If that trial shows success, it could mean that the platform is right, but the targets in the Imbokodo vaccine were wrong.
Dr. Dieffenbach said that before NIAID decides what to do with Mosaico they’ve asked researchers to analyze the data on the people who did respond, to see if those people have some specific variant of HIV or some other biomarker that could be used to form the next iteration of an HIV vaccine candidate. Only after that will they make a decision about Mosaico.
But he added that it does make him wonder if vaccine approaches that rely on nonneutralizing antibodies like this one have a ceiling of effectiveness that’s just too low to alter the course of the epidemic.
“I think we’ve discovered that there’s not a floor to [these nonneutralizing approaches], but there probably is a ceiling,” he said. “I don’t know if we’re going to get better than” a 25%-29% efficacy rate with those approaches.
The Imbokodo findings reminded Mitchell Warren, executive director of the global HIV prevention nonprofit, AVAC, of the data released in January from the Antibody Mediated Prevention (AMP) trial. That trial pitted the broadly neutralizing antibody (bNAb) VCR01 against HIV – and mostly, it lost.
VCR01 worked only on HIV variants that 30% of participants had. But in those 30%, it was 75% effective at preventing HIV. Now you have Imbokodo, with its potential 25% activity against HIV, something that may have been a fluke. This, to Mr. Warren, requires a rethinking of the whole HIV vaccine enterprise while “doubling, tripling, quadrupling down” on the HIV prevention methods we know work, such as preexposure prophylaxis (PrEP).
Dr. Dieffenbach agreed. To clinicians, Dr. Dieffenbach said the message of this HIV vaccine trial is flush with urgency: “Get your HIV-negative, at-risk people on PrEP tomorrow.”
There are now two pills approved for HIV prevention, both of which have been found to be up to 99% effective when taken consistently. A third option, injectable cabotegravir (Vocabria), has been submitted to the Food and Drug Administration for approval. The federal Ready, Set, PrEP program makes the pill available for free for those who qualify, and recently the Biden administration reaffirmed that, under the Affordable Care Act, insurance companies should cover all costs associated with PrEP, including lab work and exam visits.
But for the 157 women who participated in the trial at Dr. Gill’s site in Masiphumelele, on the southwestern tip of South Africa, the trial was personal, said Jason Naidoo, community liaison officer at the Desmond Tutu HIV Foundation, which conducted a portion of the study. These were women whose parents, siblings, or children were living with HIV or had died from AIDS-defining illnesses, he said. Their lives were chaotic, traveling at a moment’s notice to hometowns on the Eastern Cape, an 11-hour car ride away – longer by bus – for traditional prayers, funerals, and other important events.
Mr. Naidoo remembers arranging buses for the women to return for scheduled clinic visits, leaving the Eastern Cape in the afternoon and arriving in Masiphumelele in the early morning hours, just to keep the clinic appointment. Then, they’d turn around and return east.
They did this for 3 years, he said.
“The fact that these participants have stuck to this and been dedicated amidst all of the chaos talks about their commitment to actually having a vaccine for HIV,” he said. “They know their own risk profile as young Black women in South Africa, and they understand the need for an intervention for the future generations.
“So you can understand the emotion and the sense of sadness, the disappointment – the incredible [dis]belief that this [the failure of the vaccine] could have happened, because the expectations are so, so high.”
For Dr. Gill, who is lead investigator for Imbokodo in Masiphumelele, the weariness toward vaccines is back. Another trial is underway for an HIV vaccine with a platform that was successful in COVID-19 – using messenger RNA (mRNA), like the Pfizer and Moderna COVID-19 vaccines did.
“I think we need to be careful,” she said, “thinking that the mRNA vaccines are going to crack it.”
Dr. Dieffenbach, Dr. Gill, and Mr. Naidoo have disclosed no relevant financial relationships. The study was funded by Janssen, a Johnson & Johnson company, with NIAID and the Bill and Melinda Gates Foundation.
A version of this article first appeared on Medscape.com.
Last year, Katherine Gill, MBChB, an HIV prevention researcher in Cape Town, South Africa, realized how jaded she’d become to vaccine research when the Pfizer COVID-19 vaccine came back as 95% effective. In her career conducting HIV clinical trials, she had never seen anything like it. Even HIV prevention methods she had studied that had worked, such as the dapivirine ring, had an overall efficacy of 30%.
The COVID-19 success story started to soften her views toward another vaccine trial she was helping to conduct, a trial in HIV that used the same platform as Johnson & Johnson’s successful COVID-19 vaccine.
“When the COVID vaccine was cracked so quickly and seemingly quite easily, I did start to think, ‘Well, maybe … maybe this will work for HIV,’ “ she said in an interview.
That turned out to be false hope. The National Institutes of Health (NIH) announced that the trial Dr. Gill was helping to conduct, HVTN 705, was stopping early because it hadn’t generated enough of an immune response in participants to justify continuing. It was the second HIV vaccine to fail in the last year. It’s also the latest in what has been a litany of disappointments in the attempt to boost the human immune system to fight HIV without the need for ongoing HIV treatment.
In HVTN 705, known as the Imbokodo study (imbokodo is a Zulu word that’s part of a saying about women being strong as rocks), researchers used the platform made up of a common cold virus, adenovirus 26, to deliver a computer-generated mosaic of HIV antigens to participants’ immune systems. That mosaic of antigens is meant to goose the immune system into recognizing HIV if it were exposed to it.
When HIV enters the body, it infiltrates immune cells and replicates within them. To the rest of the immune system, those cells still register as just typical T-cells. The rest of the immune system can’t see that the virus is spreading through the very cells meant to protect the body from illness. That, plus the armor of sugary glycoproteins encasing the virus, has made HIV nearly impervious to vaccination.
Then, those so-called “prime” shots were followed by a second shot that targets glycoprotein 140, on the most common HIV subtype (or clade) in Africa, clade C. In the Imbokodo trial, a total of 2,637 women from five sub-Saharan African countries received shots at baseline, 3 months, 6 months, and 1 year. Then researchers followed the women from month 7 to 2 years after their third dose, testing their blood to see if their immune systems had generated the immune response the vaccine was meant to induce – and whether such immune response was associated with lower rates of HIV.
When researchers looked at the first 2 years of data, they learned that the vaccine was safe. And they found a total of 114 new cases of HIV – 51 among women who received the vaccine and 63 among those who received a placebo. That’s a 25.2% efficacy rate – but it wasn’t statistically significant.
The results are frustrating, said Carl Dieffenbach, PhD, director of the AIDS division at the National Institute of Allergy and Infectious Diseases (NIAID). NIAID is one of the funders of the study.
“This [trial] is a little more confounding, in that there is this low level of statistically insignificant difference between vaccine and placebo that starts somewhere around month 9 and then just kind of indolently is maintained over the next 15 months,” he said in an interview. “That’s kind of frustrating. Does it mean there’s a signal or is this just chance? Because that’s what statistics tell us, not to believe your last data point.”
What this means for the future direction of vaccine research is unclear. A sister study to Imbokodo, called Mosaico, recently finished enrolling participants. Mosaico uses the same adenovirus 26 platform, but it’s loaded with different antigens and targets a different glycoprotein for a different HIV subtype. If that trial shows success, it could mean that the platform is right, but the targets in the Imbokodo vaccine were wrong.
Dr. Dieffenbach said that before NIAID decides what to do with Mosaico they’ve asked researchers to analyze the data on the people who did respond, to see if those people have some specific variant of HIV or some other biomarker that could be used to form the next iteration of an HIV vaccine candidate. Only after that will they make a decision about Mosaico.
But he added that it does make him wonder if vaccine approaches that rely on nonneutralizing antibodies like this one have a ceiling of effectiveness that’s just too low to alter the course of the epidemic.
“I think we’ve discovered that there’s not a floor to [these nonneutralizing approaches], but there probably is a ceiling,” he said. “I don’t know if we’re going to get better than” a 25%-29% efficacy rate with those approaches.
The Imbokodo findings reminded Mitchell Warren, executive director of the global HIV prevention nonprofit, AVAC, of the data released in January from the Antibody Mediated Prevention (AMP) trial. That trial pitted the broadly neutralizing antibody (bNAb) VCR01 against HIV – and mostly, it lost.
VCR01 worked only on HIV variants that 30% of participants had. But in those 30%, it was 75% effective at preventing HIV. Now you have Imbokodo, with its potential 25% activity against HIV, something that may have been a fluke. This, to Mr. Warren, requires a rethinking of the whole HIV vaccine enterprise while “doubling, tripling, quadrupling down” on the HIV prevention methods we know work, such as preexposure prophylaxis (PrEP).
Dr. Dieffenbach agreed. To clinicians, Dr. Dieffenbach said the message of this HIV vaccine trial is flush with urgency: “Get your HIV-negative, at-risk people on PrEP tomorrow.”
There are now two pills approved for HIV prevention, both of which have been found to be up to 99% effective when taken consistently. A third option, injectable cabotegravir (Vocabria), has been submitted to the Food and Drug Administration for approval. The federal Ready, Set, PrEP program makes the pill available for free for those who qualify, and recently the Biden administration reaffirmed that, under the Affordable Care Act, insurance companies should cover all costs associated with PrEP, including lab work and exam visits.
But for the 157 women who participated in the trial at Dr. Gill’s site in Masiphumelele, on the southwestern tip of South Africa, the trial was personal, said Jason Naidoo, community liaison officer at the Desmond Tutu HIV Foundation, which conducted a portion of the study. These were women whose parents, siblings, or children were living with HIV or had died from AIDS-defining illnesses, he said. Their lives were chaotic, traveling at a moment’s notice to hometowns on the Eastern Cape, an 11-hour car ride away – longer by bus – for traditional prayers, funerals, and other important events.
Mr. Naidoo remembers arranging buses for the women to return for scheduled clinic visits, leaving the Eastern Cape in the afternoon and arriving in Masiphumelele in the early morning hours, just to keep the clinic appointment. Then, they’d turn around and return east.
They did this for 3 years, he said.
“The fact that these participants have stuck to this and been dedicated amidst all of the chaos talks about their commitment to actually having a vaccine for HIV,” he said. “They know their own risk profile as young Black women in South Africa, and they understand the need for an intervention for the future generations.
“So you can understand the emotion and the sense of sadness, the disappointment – the incredible [dis]belief that this [the failure of the vaccine] could have happened, because the expectations are so, so high.”
For Dr. Gill, who is lead investigator for Imbokodo in Masiphumelele, the weariness toward vaccines is back. Another trial is underway for an HIV vaccine with a platform that was successful in COVID-19 – using messenger RNA (mRNA), like the Pfizer and Moderna COVID-19 vaccines did.
“I think we need to be careful,” she said, “thinking that the mRNA vaccines are going to crack it.”
Dr. Dieffenbach, Dr. Gill, and Mr. Naidoo have disclosed no relevant financial relationships. The study was funded by Janssen, a Johnson & Johnson company, with NIAID and the Bill and Melinda Gates Foundation.
A version of this article first appeared on Medscape.com.