User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
gambling
compulsive behaviors
ammunition
assault rifle
black jack
Boko Haram
bondage
child abuse
cocaine
Daech
drug paraphernalia
explosion
gun
human trafficking
ISIL
ISIS
Islamic caliphate
Islamic state
mixed martial arts
MMA
molestation
national rifle association
NRA
nsfw
pedophile
pedophilia
poker
porn
pornography
psychedelic drug
recreational drug
sex slave rings
slot machine
terrorism
terrorist
Texas hold 'em
UFC
substance abuse
abuseed
abuseer
abusees
abuseing
abusely
abuses
aeolus
aeolused
aeoluser
aeoluses
aeolusing
aeolusly
aeoluss
ahole
aholeed
aholeer
aholees
aholeing
aholely
aholes
alcohol
alcoholed
alcoholer
alcoholes
alcoholing
alcoholly
alcohols
allman
allmaned
allmaner
allmanes
allmaning
allmanly
allmans
alted
altes
alting
altly
alts
analed
analer
anales
analing
anally
analprobe
analprobeed
analprobeer
analprobees
analprobeing
analprobely
analprobes
anals
anilingus
anilingused
anilinguser
anilinguses
anilingusing
anilingusly
anilinguss
anus
anused
anuser
anuses
anusing
anusly
anuss
areola
areolaed
areolaer
areolaes
areolaing
areolaly
areolas
areole
areoleed
areoleer
areolees
areoleing
areolely
areoles
arian
arianed
arianer
arianes
arianing
arianly
arians
aryan
aryaned
aryaner
aryanes
aryaning
aryanly
aryans
asiaed
asiaer
asiaes
asiaing
asialy
asias
ass
ass hole
ass lick
ass licked
ass licker
ass lickes
ass licking
ass lickly
ass licks
assbang
assbanged
assbangeded
assbangeder
assbangedes
assbangeding
assbangedly
assbangeds
assbanger
assbanges
assbanging
assbangly
assbangs
assbangsed
assbangser
assbangses
assbangsing
assbangsly
assbangss
assed
asser
asses
assesed
asseser
asseses
assesing
assesly
assess
assfuck
assfucked
assfucker
assfuckered
assfuckerer
assfuckeres
assfuckering
assfuckerly
assfuckers
assfuckes
assfucking
assfuckly
assfucks
asshat
asshated
asshater
asshates
asshating
asshatly
asshats
assholeed
assholeer
assholees
assholeing
assholely
assholes
assholesed
assholeser
assholeses
assholesing
assholesly
assholess
assing
assly
assmaster
assmastered
assmasterer
assmasteres
assmastering
assmasterly
assmasters
assmunch
assmunched
assmuncher
assmunches
assmunching
assmunchly
assmunchs
asss
asswipe
asswipeed
asswipeer
asswipees
asswipeing
asswipely
asswipes
asswipesed
asswipeser
asswipeses
asswipesing
asswipesly
asswipess
azz
azzed
azzer
azzes
azzing
azzly
azzs
babeed
babeer
babees
babeing
babely
babes
babesed
babeser
babeses
babesing
babesly
babess
ballsac
ballsaced
ballsacer
ballsaces
ballsacing
ballsack
ballsacked
ballsacker
ballsackes
ballsacking
ballsackly
ballsacks
ballsacly
ballsacs
ballsed
ballser
ballses
ballsing
ballsly
ballss
barf
barfed
barfer
barfes
barfing
barfly
barfs
bastard
bastarded
bastarder
bastardes
bastarding
bastardly
bastards
bastardsed
bastardser
bastardses
bastardsing
bastardsly
bastardss
bawdy
bawdyed
bawdyer
bawdyes
bawdying
bawdyly
bawdys
beaner
beanered
beanerer
beaneres
beanering
beanerly
beaners
beardedclam
beardedclamed
beardedclamer
beardedclames
beardedclaming
beardedclamly
beardedclams
beastiality
beastialityed
beastialityer
beastialityes
beastialitying
beastialityly
beastialitys
beatch
beatched
beatcher
beatches
beatching
beatchly
beatchs
beater
beatered
beaterer
beateres
beatering
beaterly
beaters
beered
beerer
beeres
beering
beerly
beeyotch
beeyotched
beeyotcher
beeyotches
beeyotching
beeyotchly
beeyotchs
beotch
beotched
beotcher
beotches
beotching
beotchly
beotchs
biatch
biatched
biatcher
biatches
biatching
biatchly
biatchs
big tits
big titsed
big titser
big titses
big titsing
big titsly
big titss
bigtits
bigtitsed
bigtitser
bigtitses
bigtitsing
bigtitsly
bigtitss
bimbo
bimboed
bimboer
bimboes
bimboing
bimboly
bimbos
bisexualed
bisexualer
bisexuales
bisexualing
bisexually
bisexuals
bitch
bitched
bitcheded
bitcheder
bitchedes
bitcheding
bitchedly
bitcheds
bitcher
bitches
bitchesed
bitcheser
bitcheses
bitchesing
bitchesly
bitchess
bitching
bitchly
bitchs
bitchy
bitchyed
bitchyer
bitchyes
bitchying
bitchyly
bitchys
bleached
bleacher
bleaches
bleaching
bleachly
bleachs
blow job
blow jobed
blow jober
blow jobes
blow jobing
blow jobly
blow jobs
blowed
blower
blowes
blowing
blowjob
blowjobed
blowjober
blowjobes
blowjobing
blowjobly
blowjobs
blowjobsed
blowjobser
blowjobses
blowjobsing
blowjobsly
blowjobss
blowly
blows
boink
boinked
boinker
boinkes
boinking
boinkly
boinks
bollock
bollocked
bollocker
bollockes
bollocking
bollockly
bollocks
bollocksed
bollockser
bollockses
bollocksing
bollocksly
bollockss
bollok
bolloked
bolloker
bollokes
bolloking
bollokly
bolloks
boner
bonered
bonerer
boneres
bonering
bonerly
boners
bonersed
bonerser
bonerses
bonersing
bonersly
bonerss
bong
bonged
bonger
bonges
bonging
bongly
bongs
boob
boobed
boober
boobes
boobies
boobiesed
boobieser
boobieses
boobiesing
boobiesly
boobiess
boobing
boobly
boobs
boobsed
boobser
boobses
boobsing
boobsly
boobss
booby
boobyed
boobyer
boobyes
boobying
boobyly
boobys
booger
boogered
boogerer
boogeres
boogering
boogerly
boogers
bookie
bookieed
bookieer
bookiees
bookieing
bookiely
bookies
bootee
booteeed
booteeer
booteees
booteeing
booteely
bootees
bootie
bootieed
bootieer
bootiees
bootieing
bootiely
booties
booty
bootyed
bootyer
bootyes
bootying
bootyly
bootys
boozeed
boozeer
boozees
boozeing
boozely
boozer
boozered
boozerer
boozeres
boozering
boozerly
boozers
boozes
boozy
boozyed
boozyer
boozyes
boozying
boozyly
boozys
bosomed
bosomer
bosomes
bosoming
bosomly
bosoms
bosomy
bosomyed
bosomyer
bosomyes
bosomying
bosomyly
bosomys
bugger
buggered
buggerer
buggeres
buggering
buggerly
buggers
bukkake
bukkakeed
bukkakeer
bukkakees
bukkakeing
bukkakely
bukkakes
bull shit
bull shited
bull shiter
bull shites
bull shiting
bull shitly
bull shits
bullshit
bullshited
bullshiter
bullshites
bullshiting
bullshitly
bullshits
bullshitsed
bullshitser
bullshitses
bullshitsing
bullshitsly
bullshitss
bullshitted
bullshitteded
bullshitteder
bullshittedes
bullshitteding
bullshittedly
bullshitteds
bullturds
bullturdsed
bullturdser
bullturdses
bullturdsing
bullturdsly
bullturdss
bung
bunged
bunger
bunges
bunging
bungly
bungs
busty
bustyed
bustyer
bustyes
bustying
bustyly
bustys
butt
butt fuck
butt fucked
butt fucker
butt fuckes
butt fucking
butt fuckly
butt fucks
butted
buttes
buttfuck
buttfucked
buttfucker
buttfuckered
buttfuckerer
buttfuckeres
buttfuckering
buttfuckerly
buttfuckers
buttfuckes
buttfucking
buttfuckly
buttfucks
butting
buttly
buttplug
buttpluged
buttpluger
buttpluges
buttpluging
buttplugly
buttplugs
butts
caca
cacaed
cacaer
cacaes
cacaing
cacaly
cacas
cahone
cahoneed
cahoneer
cahonees
cahoneing
cahonely
cahones
cameltoe
cameltoeed
cameltoeer
cameltoees
cameltoeing
cameltoely
cameltoes
carpetmuncher
carpetmunchered
carpetmuncherer
carpetmuncheres
carpetmunchering
carpetmuncherly
carpetmunchers
cawk
cawked
cawker
cawkes
cawking
cawkly
cawks
chinc
chinced
chincer
chinces
chincing
chincly
chincs
chincsed
chincser
chincses
chincsing
chincsly
chincss
chink
chinked
chinker
chinkes
chinking
chinkly
chinks
chode
chodeed
chodeer
chodees
chodeing
chodely
chodes
chodesed
chodeser
chodeses
chodesing
chodesly
chodess
clit
clited
cliter
clites
cliting
clitly
clitoris
clitorised
clitoriser
clitorises
clitorising
clitorisly
clitoriss
clitorus
clitorused
clitoruser
clitoruses
clitorusing
clitorusly
clitoruss
clits
clitsed
clitser
clitses
clitsing
clitsly
clitss
clitty
clittyed
clittyer
clittyes
clittying
clittyly
clittys
cocain
cocaine
cocained
cocaineed
cocaineer
cocainees
cocaineing
cocainely
cocainer
cocaines
cocaining
cocainly
cocains
cock
cock sucker
cock suckered
cock suckerer
cock suckeres
cock suckering
cock suckerly
cock suckers
cockblock
cockblocked
cockblocker
cockblockes
cockblocking
cockblockly
cockblocks
cocked
cocker
cockes
cockholster
cockholstered
cockholsterer
cockholsteres
cockholstering
cockholsterly
cockholsters
cocking
cockknocker
cockknockered
cockknockerer
cockknockeres
cockknockering
cockknockerly
cockknockers
cockly
cocks
cocksed
cockser
cockses
cocksing
cocksly
cocksmoker
cocksmokered
cocksmokerer
cocksmokeres
cocksmokering
cocksmokerly
cocksmokers
cockss
cocksucker
cocksuckered
cocksuckerer
cocksuckeres
cocksuckering
cocksuckerly
cocksuckers
coital
coitaled
coitaler
coitales
coitaling
coitally
coitals
commie
commieed
commieer
commiees
commieing
commiely
commies
condomed
condomer
condomes
condoming
condomly
condoms
coon
cooned
cooner
coones
cooning
coonly
coons
coonsed
coonser
coonses
coonsing
coonsly
coonss
corksucker
corksuckered
corksuckerer
corksuckeres
corksuckering
corksuckerly
corksuckers
cracked
crackwhore
crackwhoreed
crackwhoreer
crackwhorees
crackwhoreing
crackwhorely
crackwhores
crap
craped
craper
crapes
craping
craply
crappy
crappyed
crappyer
crappyes
crappying
crappyly
crappys
cum
cumed
cumer
cumes
cuming
cumly
cummin
cummined
cumminer
cummines
cumming
cumminged
cumminger
cumminges
cumminging
cummingly
cummings
cummining
cumminly
cummins
cums
cumshot
cumshoted
cumshoter
cumshotes
cumshoting
cumshotly
cumshots
cumshotsed
cumshotser
cumshotses
cumshotsing
cumshotsly
cumshotss
cumslut
cumsluted
cumsluter
cumslutes
cumsluting
cumslutly
cumsluts
cumstain
cumstained
cumstainer
cumstaines
cumstaining
cumstainly
cumstains
cunilingus
cunilingused
cunilinguser
cunilinguses
cunilingusing
cunilingusly
cunilinguss
cunnilingus
cunnilingused
cunnilinguser
cunnilinguses
cunnilingusing
cunnilingusly
cunnilinguss
cunny
cunnyed
cunnyer
cunnyes
cunnying
cunnyly
cunnys
cunt
cunted
cunter
cuntes
cuntface
cuntfaceed
cuntfaceer
cuntfacees
cuntfaceing
cuntfacely
cuntfaces
cunthunter
cunthuntered
cunthunterer
cunthunteres
cunthuntering
cunthunterly
cunthunters
cunting
cuntlick
cuntlicked
cuntlicker
cuntlickered
cuntlickerer
cuntlickeres
cuntlickering
cuntlickerly
cuntlickers
cuntlickes
cuntlicking
cuntlickly
cuntlicks
cuntly
cunts
cuntsed
cuntser
cuntses
cuntsing
cuntsly
cuntss
dago
dagoed
dagoer
dagoes
dagoing
dagoly
dagos
dagosed
dagoser
dagoses
dagosing
dagosly
dagoss
dammit
dammited
dammiter
dammites
dammiting
dammitly
dammits
damn
damned
damneded
damneder
damnedes
damneding
damnedly
damneds
damner
damnes
damning
damnit
damnited
damniter
damnites
damniting
damnitly
damnits
damnly
damns
dick
dickbag
dickbaged
dickbager
dickbages
dickbaging
dickbagly
dickbags
dickdipper
dickdippered
dickdipperer
dickdipperes
dickdippering
dickdipperly
dickdippers
dicked
dicker
dickes
dickface
dickfaceed
dickfaceer
dickfacees
dickfaceing
dickfacely
dickfaces
dickflipper
dickflippered
dickflipperer
dickflipperes
dickflippering
dickflipperly
dickflippers
dickhead
dickheaded
dickheader
dickheades
dickheading
dickheadly
dickheads
dickheadsed
dickheadser
dickheadses
dickheadsing
dickheadsly
dickheadss
dicking
dickish
dickished
dickisher
dickishes
dickishing
dickishly
dickishs
dickly
dickripper
dickrippered
dickripperer
dickripperes
dickrippering
dickripperly
dickrippers
dicks
dicksipper
dicksippered
dicksipperer
dicksipperes
dicksippering
dicksipperly
dicksippers
dickweed
dickweeded
dickweeder
dickweedes
dickweeding
dickweedly
dickweeds
dickwhipper
dickwhippered
dickwhipperer
dickwhipperes
dickwhippering
dickwhipperly
dickwhippers
dickzipper
dickzippered
dickzipperer
dickzipperes
dickzippering
dickzipperly
dickzippers
diddle
diddleed
diddleer
diddlees
diddleing
diddlely
diddles
dike
dikeed
dikeer
dikees
dikeing
dikely
dikes
dildo
dildoed
dildoer
dildoes
dildoing
dildoly
dildos
dildosed
dildoser
dildoses
dildosing
dildosly
dildoss
diligaf
diligafed
diligafer
diligafes
diligafing
diligafly
diligafs
dillweed
dillweeded
dillweeder
dillweedes
dillweeding
dillweedly
dillweeds
dimwit
dimwited
dimwiter
dimwites
dimwiting
dimwitly
dimwits
dingle
dingleed
dingleer
dinglees
dingleing
dinglely
dingles
dipship
dipshiped
dipshiper
dipshipes
dipshiping
dipshiply
dipships
dizzyed
dizzyer
dizzyes
dizzying
dizzyly
dizzys
doggiestyleed
doggiestyleer
doggiestylees
doggiestyleing
doggiestylely
doggiestyles
doggystyleed
doggystyleer
doggystylees
doggystyleing
doggystylely
doggystyles
dong
donged
donger
donges
donging
dongly
dongs
doofus
doofused
doofuser
doofuses
doofusing
doofusly
doofuss
doosh
dooshed
doosher
dooshes
dooshing
dooshly
dooshs
dopeyed
dopeyer
dopeyes
dopeying
dopeyly
dopeys
douchebag
douchebaged
douchebager
douchebages
douchebaging
douchebagly
douchebags
douchebagsed
douchebagser
douchebagses
douchebagsing
douchebagsly
douchebagss
doucheed
doucheer
douchees
doucheing
douchely
douches
douchey
doucheyed
doucheyer
doucheyes
doucheying
doucheyly
doucheys
drunk
drunked
drunker
drunkes
drunking
drunkly
drunks
dumass
dumassed
dumasser
dumasses
dumassing
dumassly
dumasss
dumbass
dumbassed
dumbasser
dumbasses
dumbassesed
dumbasseser
dumbasseses
dumbassesing
dumbassesly
dumbassess
dumbassing
dumbassly
dumbasss
dummy
dummyed
dummyer
dummyes
dummying
dummyly
dummys
dyke
dykeed
dykeer
dykees
dykeing
dykely
dykes
dykesed
dykeser
dykeses
dykesing
dykesly
dykess
erotic
eroticed
eroticer
erotices
eroticing
eroticly
erotics
extacy
extacyed
extacyer
extacyes
extacying
extacyly
extacys
extasy
extasyed
extasyer
extasyes
extasying
extasyly
extasys
fack
facked
facker
fackes
facking
fackly
facks
fag
faged
fager
fages
fagg
fagged
faggeded
faggeder
faggedes
faggeding
faggedly
faggeds
fagger
fagges
fagging
faggit
faggited
faggiter
faggites
faggiting
faggitly
faggits
faggly
faggot
faggoted
faggoter
faggotes
faggoting
faggotly
faggots
faggs
faging
fagly
fagot
fagoted
fagoter
fagotes
fagoting
fagotly
fagots
fags
fagsed
fagser
fagses
fagsing
fagsly
fagss
faig
faiged
faiger
faiges
faiging
faigly
faigs
faigt
faigted
faigter
faigtes
faigting
faigtly
faigts
fannybandit
fannybandited
fannybanditer
fannybandites
fannybanditing
fannybanditly
fannybandits
farted
farter
fartes
farting
fartknocker
fartknockered
fartknockerer
fartknockeres
fartknockering
fartknockerly
fartknockers
fartly
farts
felch
felched
felcher
felchered
felcherer
felcheres
felchering
felcherly
felchers
felches
felching
felchinged
felchinger
felchinges
felchinging
felchingly
felchings
felchly
felchs
fellate
fellateed
fellateer
fellatees
fellateing
fellately
fellates
fellatio
fellatioed
fellatioer
fellatioes
fellatioing
fellatioly
fellatios
feltch
feltched
feltcher
feltchered
feltcherer
feltcheres
feltchering
feltcherly
feltchers
feltches
feltching
feltchly
feltchs
feom
feomed
feomer
feomes
feoming
feomly
feoms
fisted
fisteded
fisteder
fistedes
fisteding
fistedly
fisteds
fisting
fistinged
fistinger
fistinges
fistinging
fistingly
fistings
fisty
fistyed
fistyer
fistyes
fistying
fistyly
fistys
floozy
floozyed
floozyer
floozyes
floozying
floozyly
floozys
foad
foaded
foader
foades
foading
foadly
foads
fondleed
fondleer
fondlees
fondleing
fondlely
fondles
foobar
foobared
foobarer
foobares
foobaring
foobarly
foobars
freex
freexed
freexer
freexes
freexing
freexly
freexs
frigg
frigga
friggaed
friggaer
friggaes
friggaing
friggaly
friggas
frigged
frigger
frigges
frigging
friggly
friggs
fubar
fubared
fubarer
fubares
fubaring
fubarly
fubars
fuck
fuckass
fuckassed
fuckasser
fuckasses
fuckassing
fuckassly
fuckasss
fucked
fuckeded
fuckeder
fuckedes
fuckeding
fuckedly
fuckeds
fucker
fuckered
fuckerer
fuckeres
fuckering
fuckerly
fuckers
fuckes
fuckface
fuckfaceed
fuckfaceer
fuckfacees
fuckfaceing
fuckfacely
fuckfaces
fuckin
fuckined
fuckiner
fuckines
fucking
fuckinged
fuckinger
fuckinges
fuckinging
fuckingly
fuckings
fuckining
fuckinly
fuckins
fuckly
fucknugget
fucknuggeted
fucknuggeter
fucknuggetes
fucknuggeting
fucknuggetly
fucknuggets
fucknut
fucknuted
fucknuter
fucknutes
fucknuting
fucknutly
fucknuts
fuckoff
fuckoffed
fuckoffer
fuckoffes
fuckoffing
fuckoffly
fuckoffs
fucks
fucksed
fuckser
fuckses
fucksing
fucksly
fuckss
fucktard
fucktarded
fucktarder
fucktardes
fucktarding
fucktardly
fucktards
fuckup
fuckuped
fuckuper
fuckupes
fuckuping
fuckuply
fuckups
fuckwad
fuckwaded
fuckwader
fuckwades
fuckwading
fuckwadly
fuckwads
fuckwit
fuckwited
fuckwiter
fuckwites
fuckwiting
fuckwitly
fuckwits
fudgepacker
fudgepackered
fudgepackerer
fudgepackeres
fudgepackering
fudgepackerly
fudgepackers
fuk
fuked
fuker
fukes
fuking
fukly
fuks
fvck
fvcked
fvcker
fvckes
fvcking
fvckly
fvcks
fxck
fxcked
fxcker
fxckes
fxcking
fxckly
fxcks
gae
gaeed
gaeer
gaees
gaeing
gaely
gaes
gai
gaied
gaier
gaies
gaiing
gaily
gais
ganja
ganjaed
ganjaer
ganjaes
ganjaing
ganjaly
ganjas
gayed
gayer
gayes
gaying
gayly
gays
gaysed
gayser
gayses
gaysing
gaysly
gayss
gey
geyed
geyer
geyes
geying
geyly
geys
gfc
gfced
gfcer
gfces
gfcing
gfcly
gfcs
gfy
gfyed
gfyer
gfyes
gfying
gfyly
gfys
ghay
ghayed
ghayer
ghayes
ghaying
ghayly
ghays
ghey
gheyed
gheyer
gheyes
gheying
gheyly
gheys
gigolo
gigoloed
gigoloer
gigoloes
gigoloing
gigololy
gigolos
goatse
goatseed
goatseer
goatsees
goatseing
goatsely
goatses
godamn
godamned
godamner
godamnes
godamning
godamnit
godamnited
godamniter
godamnites
godamniting
godamnitly
godamnits
godamnly
godamns
goddam
goddamed
goddamer
goddames
goddaming
goddamly
goddammit
goddammited
goddammiter
goddammites
goddammiting
goddammitly
goddammits
goddamn
goddamned
goddamner
goddamnes
goddamning
goddamnly
goddamns
goddams
goldenshower
goldenshowered
goldenshowerer
goldenshoweres
goldenshowering
goldenshowerly
goldenshowers
gonad
gonaded
gonader
gonades
gonading
gonadly
gonads
gonadsed
gonadser
gonadses
gonadsing
gonadsly
gonadss
gook
gooked
gooker
gookes
gooking
gookly
gooks
gooksed
gookser
gookses
gooksing
gooksly
gookss
gringo
gringoed
gringoer
gringoes
gringoing
gringoly
gringos
gspot
gspoted
gspoter
gspotes
gspoting
gspotly
gspots
gtfo
gtfoed
gtfoer
gtfoes
gtfoing
gtfoly
gtfos
guido
guidoed
guidoer
guidoes
guidoing
guidoly
guidos
handjob
handjobed
handjober
handjobes
handjobing
handjobly
handjobs
hard on
hard oned
hard oner
hard ones
hard oning
hard only
hard ons
hardknight
hardknighted
hardknighter
hardknightes
hardknighting
hardknightly
hardknights
hebe
hebeed
hebeer
hebees
hebeing
hebely
hebes
heeb
heebed
heeber
heebes
heebing
heebly
heebs
hell
helled
heller
helles
helling
hellly
hells
hemp
hemped
hemper
hempes
hemping
hemply
hemps
heroined
heroiner
heroines
heroining
heroinly
heroins
herp
herped
herper
herpes
herpesed
herpeser
herpeses
herpesing
herpesly
herpess
herping
herply
herps
herpy
herpyed
herpyer
herpyes
herpying
herpyly
herpys
hitler
hitlered
hitlerer
hitleres
hitlering
hitlerly
hitlers
hived
hiver
hives
hiving
hivly
hivs
hobag
hobaged
hobager
hobages
hobaging
hobagly
hobags
homey
homeyed
homeyer
homeyes
homeying
homeyly
homeys
homo
homoed
homoer
homoes
homoey
homoeyed
homoeyer
homoeyes
homoeying
homoeyly
homoeys
homoing
homoly
homos
honky
honkyed
honkyer
honkyes
honkying
honkyly
honkys
hooch
hooched
hoocher
hooches
hooching
hoochly
hoochs
hookah
hookahed
hookaher
hookahes
hookahing
hookahly
hookahs
hooker
hookered
hookerer
hookeres
hookering
hookerly
hookers
hoor
hoored
hoorer
hoores
hooring
hoorly
hoors
hootch
hootched
hootcher
hootches
hootching
hootchly
hootchs
hooter
hootered
hooterer
hooteres
hootering
hooterly
hooters
hootersed
hooterser
hooterses
hootersing
hootersly
hooterss
horny
hornyed
hornyer
hornyes
hornying
hornyly
hornys
houstoned
houstoner
houstones
houstoning
houstonly
houstons
hump
humped
humpeded
humpeder
humpedes
humpeding
humpedly
humpeds
humper
humpes
humping
humpinged
humpinger
humpinges
humpinging
humpingly
humpings
humply
humps
husbanded
husbander
husbandes
husbanding
husbandly
husbands
hussy
hussyed
hussyer
hussyes
hussying
hussyly
hussys
hymened
hymener
hymenes
hymening
hymenly
hymens
inbred
inbreded
inbreder
inbredes
inbreding
inbredly
inbreds
incest
incested
incester
incestes
incesting
incestly
incests
injun
injuned
injuner
injunes
injuning
injunly
injuns
jackass
jackassed
jackasser
jackasses
jackassing
jackassly
jackasss
jackhole
jackholeed
jackholeer
jackholees
jackholeing
jackholely
jackholes
jackoff
jackoffed
jackoffer
jackoffes
jackoffing
jackoffly
jackoffs
jap
japed
japer
japes
japing
japly
japs
japsed
japser
japses
japsing
japsly
japss
jerkoff
jerkoffed
jerkoffer
jerkoffes
jerkoffing
jerkoffly
jerkoffs
jerks
jism
jismed
jismer
jismes
jisming
jismly
jisms
jiz
jized
jizer
jizes
jizing
jizly
jizm
jizmed
jizmer
jizmes
jizming
jizmly
jizms
jizs
jizz
jizzed
jizzeded
jizzeder
jizzedes
jizzeding
jizzedly
jizzeds
jizzer
jizzes
jizzing
jizzly
jizzs
junkie
junkieed
junkieer
junkiees
junkieing
junkiely
junkies
junky
junkyed
junkyer
junkyes
junkying
junkyly
junkys
kike
kikeed
kikeer
kikees
kikeing
kikely
kikes
kikesed
kikeser
kikeses
kikesing
kikesly
kikess
killed
killer
killes
killing
killly
kills
kinky
kinkyed
kinkyer
kinkyes
kinkying
kinkyly
kinkys
kkk
kkked
kkker
kkkes
kkking
kkkly
kkks
klan
klaned
klaner
klanes
klaning
klanly
klans
knobend
knobended
knobender
knobendes
knobending
knobendly
knobends
kooch
kooched
koocher
kooches
koochesed
koocheser
koocheses
koochesing
koochesly
koochess
kooching
koochly
koochs
kootch
kootched
kootcher
kootches
kootching
kootchly
kootchs
kraut
krauted
krauter
krautes
krauting
krautly
krauts
kyke
kykeed
kykeer
kykees
kykeing
kykely
kykes
lech
leched
lecher
leches
leching
lechly
lechs
leper
lepered
leperer
leperes
lepering
leperly
lepers
lesbiansed
lesbianser
lesbianses
lesbiansing
lesbiansly
lesbianss
lesbo
lesboed
lesboer
lesboes
lesboing
lesboly
lesbos
lesbosed
lesboser
lesboses
lesbosing
lesbosly
lesboss
lez
lezbianed
lezbianer
lezbianes
lezbianing
lezbianly
lezbians
lezbiansed
lezbianser
lezbianses
lezbiansing
lezbiansly
lezbianss
lezbo
lezboed
lezboer
lezboes
lezboing
lezboly
lezbos
lezbosed
lezboser
lezboses
lezbosing
lezbosly
lezboss
lezed
lezer
lezes
lezing
lezly
lezs
lezzie
lezzieed
lezzieer
lezziees
lezzieing
lezziely
lezzies
lezziesed
lezzieser
lezzieses
lezziesing
lezziesly
lezziess
lezzy
lezzyed
lezzyer
lezzyes
lezzying
lezzyly
lezzys
lmaoed
lmaoer
lmaoes
lmaoing
lmaoly
lmaos
lmfao
lmfaoed
lmfaoer
lmfaoes
lmfaoing
lmfaoly
lmfaos
loined
loiner
loines
loining
loinly
loins
loinsed
loinser
loinses
loinsing
loinsly
loinss
lubeed
lubeer
lubees
lubeing
lubely
lubes
lusty
lustyed
lustyer
lustyes
lustying
lustyly
lustys
massa
massaed
massaer
massaes
massaing
massaly
massas
masterbate
masterbateed
masterbateer
masterbatees
masterbateing
masterbately
masterbates
masterbating
masterbatinged
masterbatinger
masterbatinges
masterbatinging
masterbatingly
masterbatings
masterbation
masterbationed
masterbationer
masterbationes
masterbationing
masterbationly
masterbations
masturbate
masturbateed
masturbateer
masturbatees
masturbateing
masturbately
masturbates
masturbating
masturbatinged
masturbatinger
masturbatinges
masturbatinging
masturbatingly
masturbatings
masturbation
masturbationed
masturbationer
masturbationes
masturbationing
masturbationly
masturbations
methed
mether
methes
mething
methly
meths
militaryed
militaryer
militaryes
militarying
militaryly
militarys
mofo
mofoed
mofoer
mofoes
mofoing
mofoly
mofos
molest
molested
molester
molestes
molesting
molestly
molests
moolie
moolieed
moolieer
mooliees
moolieing
mooliely
moolies
moron
moroned
moroner
morones
moroning
moronly
morons
motherfucka
motherfuckaed
motherfuckaer
motherfuckaes
motherfuckaing
motherfuckaly
motherfuckas
motherfucker
motherfuckered
motherfuckerer
motherfuckeres
motherfuckering
motherfuckerly
motherfuckers
motherfucking
motherfuckinged
motherfuckinger
motherfuckinges
motherfuckinging
motherfuckingly
motherfuckings
mtherfucker
mtherfuckered
mtherfuckerer
mtherfuckeres
mtherfuckering
mtherfuckerly
mtherfuckers
mthrfucker
mthrfuckered
mthrfuckerer
mthrfuckeres
mthrfuckering
mthrfuckerly
mthrfuckers
mthrfucking
mthrfuckinged
mthrfuckinger
mthrfuckinges
mthrfuckinging
mthrfuckingly
mthrfuckings
muff
muffdiver
muffdivered
muffdiverer
muffdiveres
muffdivering
muffdiverly
muffdivers
muffed
muffer
muffes
muffing
muffly
muffs
murdered
murderer
murderes
murdering
murderly
murders
muthafuckaz
muthafuckazed
muthafuckazer
muthafuckazes
muthafuckazing
muthafuckazly
muthafuckazs
muthafucker
muthafuckered
muthafuckerer
muthafuckeres
muthafuckering
muthafuckerly
muthafuckers
mutherfucker
mutherfuckered
mutherfuckerer
mutherfuckeres
mutherfuckering
mutherfuckerly
mutherfuckers
mutherfucking
mutherfuckinged
mutherfuckinger
mutherfuckinges
mutherfuckinging
mutherfuckingly
mutherfuckings
muthrfucking
muthrfuckinged
muthrfuckinger
muthrfuckinges
muthrfuckinging
muthrfuckingly
muthrfuckings
nad
naded
nader
nades
nading
nadly
nads
nadsed
nadser
nadses
nadsing
nadsly
nadss
nakeded
nakeder
nakedes
nakeding
nakedly
nakeds
napalm
napalmed
napalmer
napalmes
napalming
napalmly
napalms
nappy
nappyed
nappyer
nappyes
nappying
nappyly
nappys
nazi
nazied
nazier
nazies
naziing
nazily
nazis
nazism
nazismed
nazismer
nazismes
nazisming
nazismly
nazisms
negro
negroed
negroer
negroes
negroing
negroly
negros
nigga
niggaed
niggaer
niggaes
niggah
niggahed
niggaher
niggahes
niggahing
niggahly
niggahs
niggaing
niggaly
niggas
niggased
niggaser
niggases
niggasing
niggasly
niggass
niggaz
niggazed
niggazer
niggazes
niggazing
niggazly
niggazs
nigger
niggered
niggerer
niggeres
niggering
niggerly
niggers
niggersed
niggerser
niggerses
niggersing
niggersly
niggerss
niggle
niggleed
niggleer
nigglees
niggleing
nigglely
niggles
niglet
nigleted
nigleter
nigletes
nigleting
nigletly
niglets
nimrod
nimroded
nimroder
nimrodes
nimroding
nimrodly
nimrods
ninny
ninnyed
ninnyer
ninnyes
ninnying
ninnyly
ninnys
nooky
nookyed
nookyer
nookyes
nookying
nookyly
nookys
nuccitelli
nuccitellied
nuccitellier
nuccitellies
nuccitelliing
nuccitellily
nuccitellis
nympho
nymphoed
nymphoer
nymphoes
nymphoing
nympholy
nymphos
opium
opiumed
opiumer
opiumes
opiuming
opiumly
opiums
orgies
orgiesed
orgieser
orgieses
orgiesing
orgiesly
orgiess
orgy
orgyed
orgyer
orgyes
orgying
orgyly
orgys
paddy
paddyed
paddyer
paddyes
paddying
paddyly
paddys
paki
pakied
pakier
pakies
pakiing
pakily
pakis
pantie
pantieed
pantieer
pantiees
pantieing
pantiely
panties
pantiesed
pantieser
pantieses
pantiesing
pantiesly
pantiess
panty
pantyed
pantyer
pantyes
pantying
pantyly
pantys
pastie
pastieed
pastieer
pastiees
pastieing
pastiely
pasties
pasty
pastyed
pastyer
pastyes
pastying
pastyly
pastys
pecker
peckered
peckerer
peckeres
peckering
peckerly
peckers
pedo
pedoed
pedoer
pedoes
pedoing
pedoly
pedophile
pedophileed
pedophileer
pedophilees
pedophileing
pedophilely
pedophiles
pedophilia
pedophiliac
pedophiliaced
pedophiliacer
pedophiliaces
pedophiliacing
pedophiliacly
pedophiliacs
pedophiliaed
pedophiliaer
pedophiliaes
pedophiliaing
pedophilialy
pedophilias
pedos
penial
penialed
penialer
peniales
penialing
penially
penials
penile
penileed
penileer
penilees
penileing
penilely
peniles
penis
penised
peniser
penises
penising
penisly
peniss
perversion
perversioned
perversioner
perversiones
perversioning
perversionly
perversions
peyote
peyoteed
peyoteer
peyotees
peyoteing
peyotely
peyotes
phuck
phucked
phucker
phuckes
phucking
phuckly
phucks
pillowbiter
pillowbitered
pillowbiterer
pillowbiteres
pillowbitering
pillowbiterly
pillowbiters
pimp
pimped
pimper
pimpes
pimping
pimply
pimps
pinko
pinkoed
pinkoer
pinkoes
pinkoing
pinkoly
pinkos
pissed
pisseded
pisseder
pissedes
pisseding
pissedly
pisseds
pisser
pisses
pissing
pissly
pissoff
pissoffed
pissoffer
pissoffes
pissoffing
pissoffly
pissoffs
pisss
polack
polacked
polacker
polackes
polacking
polackly
polacks
pollock
pollocked
pollocker
pollockes
pollocking
pollockly
pollocks
poon
pooned
pooner
poones
pooning
poonly
poons
poontang
poontanged
poontanger
poontanges
poontanging
poontangly
poontangs
porn
porned
porner
pornes
porning
pornly
porno
pornoed
pornoer
pornoes
pornography
pornographyed
pornographyer
pornographyes
pornographying
pornographyly
pornographys
pornoing
pornoly
pornos
porns
prick
pricked
pricker
prickes
pricking
prickly
pricks
prig
priged
priger
priges
priging
prigly
prigs
prostitute
prostituteed
prostituteer
prostitutees
prostituteing
prostitutely
prostitutes
prude
prudeed
prudeer
prudees
prudeing
prudely
prudes
punkass
punkassed
punkasser
punkasses
punkassing
punkassly
punkasss
punky
punkyed
punkyer
punkyes
punkying
punkyly
punkys
puss
pussed
pusser
pusses
pussies
pussiesed
pussieser
pussieses
pussiesing
pussiesly
pussiess
pussing
pussly
pusss
pussy
pussyed
pussyer
pussyes
pussying
pussyly
pussypounder
pussypoundered
pussypounderer
pussypounderes
pussypoundering
pussypounderly
pussypounders
pussys
puto
putoed
putoer
putoes
putoing
putoly
putos
queaf
queafed
queafer
queafes
queafing
queafly
queafs
queef
queefed
queefer
queefes
queefing
queefly
queefs
queer
queered
queerer
queeres
queering
queerly
queero
queeroed
queeroer
queeroes
queeroing
queeroly
queeros
queers
queersed
queerser
queerses
queersing
queersly
queerss
quicky
quickyed
quickyer
quickyes
quickying
quickyly
quickys
quim
quimed
quimer
quimes
quiming
quimly
quims
racy
racyed
racyer
racyes
racying
racyly
racys
rape
raped
rapeded
rapeder
rapedes
rapeding
rapedly
rapeds
rapeed
rapeer
rapees
rapeing
rapely
raper
rapered
raperer
raperes
rapering
raperly
rapers
rapes
rapist
rapisted
rapister
rapistes
rapisting
rapistly
rapists
raunch
raunched
rauncher
raunches
raunching
raunchly
raunchs
rectus
rectused
rectuser
rectuses
rectusing
rectusly
rectuss
reefer
reefered
reeferer
reeferes
reefering
reeferly
reefers
reetard
reetarded
reetarder
reetardes
reetarding
reetardly
reetards
reich
reiched
reicher
reiches
reiching
reichly
reichs
retard
retarded
retardeded
retardeder
retardedes
retardeding
retardedly
retardeds
retarder
retardes
retarding
retardly
retards
rimjob
rimjobed
rimjober
rimjobes
rimjobing
rimjobly
rimjobs
ritard
ritarded
ritarder
ritardes
ritarding
ritardly
ritards
rtard
rtarded
rtarder
rtardes
rtarding
rtardly
rtards
rum
rumed
rumer
rumes
ruming
rumly
rump
rumped
rumper
rumpes
rumping
rumply
rumprammer
rumprammered
rumprammerer
rumprammeres
rumprammering
rumprammerly
rumprammers
rumps
rums
ruski
ruskied
ruskier
ruskies
ruskiing
ruskily
ruskis
sadism
sadismed
sadismer
sadismes
sadisming
sadismly
sadisms
sadist
sadisted
sadister
sadistes
sadisting
sadistly
sadists
scag
scaged
scager
scages
scaging
scagly
scags
scantily
scantilyed
scantilyer
scantilyes
scantilying
scantilyly
scantilys
schlong
schlonged
schlonger
schlonges
schlonging
schlongly
schlongs
scrog
scroged
scroger
scroges
scroging
scrogly
scrogs
scrot
scrote
scroted
scroteed
scroteer
scrotees
scroteing
scrotely
scroter
scrotes
scroting
scrotly
scrots
scrotum
scrotumed
scrotumer
scrotumes
scrotuming
scrotumly
scrotums
scrud
scruded
scruder
scrudes
scruding
scrudly
scruds
scum
scumed
scumer
scumes
scuming
scumly
scums
seaman
seamaned
seamaner
seamanes
seamaning
seamanly
seamans
seamen
seamened
seamener
seamenes
seamening
seamenly
seamens
seduceed
seduceer
seducees
seduceing
seducely
seduces
semen
semened
semener
semenes
semening
semenly
semens
shamedame
shamedameed
shamedameer
shamedamees
shamedameing
shamedamely
shamedames
shit
shite
shiteater
shiteatered
shiteaterer
shiteateres
shiteatering
shiteaterly
shiteaters
shited
shiteed
shiteer
shitees
shiteing
shitely
shiter
shites
shitface
shitfaceed
shitfaceer
shitfacees
shitfaceing
shitfacely
shitfaces
shithead
shitheaded
shitheader
shitheades
shitheading
shitheadly
shitheads
shithole
shitholeed
shitholeer
shitholees
shitholeing
shitholely
shitholes
shithouse
shithouseed
shithouseer
shithousees
shithouseing
shithousely
shithouses
shiting
shitly
shits
shitsed
shitser
shitses
shitsing
shitsly
shitss
shitt
shitted
shitteded
shitteder
shittedes
shitteding
shittedly
shitteds
shitter
shittered
shitterer
shitteres
shittering
shitterly
shitters
shittes
shitting
shittly
shitts
shitty
shittyed
shittyer
shittyes
shittying
shittyly
shittys
shiz
shized
shizer
shizes
shizing
shizly
shizs
shooted
shooter
shootes
shooting
shootly
shoots
sissy
sissyed
sissyer
sissyes
sissying
sissyly
sissys
skag
skaged
skager
skages
skaging
skagly
skags
skank
skanked
skanker
skankes
skanking
skankly
skanks
slave
slaveed
slaveer
slavees
slaveing
slavely
slaves
sleaze
sleazeed
sleazeer
sleazees
sleazeing
sleazely
sleazes
sleazy
sleazyed
sleazyer
sleazyes
sleazying
sleazyly
sleazys
slut
slutdumper
slutdumpered
slutdumperer
slutdumperes
slutdumpering
slutdumperly
slutdumpers
sluted
sluter
slutes
sluting
slutkiss
slutkissed
slutkisser
slutkisses
slutkissing
slutkissly
slutkisss
slutly
sluts
slutsed
slutser
slutses
slutsing
slutsly
slutss
smegma
smegmaed
smegmaer
smegmaes
smegmaing
smegmaly
smegmas
smut
smuted
smuter
smutes
smuting
smutly
smuts
smutty
smuttyed
smuttyer
smuttyes
smuttying
smuttyly
smuttys
snatch
snatched
snatcher
snatches
snatching
snatchly
snatchs
sniper
snipered
sniperer
sniperes
snipering
sniperly
snipers
snort
snorted
snorter
snortes
snorting
snortly
snorts
snuff
snuffed
snuffer
snuffes
snuffing
snuffly
snuffs
sodom
sodomed
sodomer
sodomes
sodoming
sodomly
sodoms
spic
spiced
spicer
spices
spicing
spick
spicked
spicker
spickes
spicking
spickly
spicks
spicly
spics
spik
spoof
spoofed
spoofer
spoofes
spoofing
spoofly
spoofs
spooge
spoogeed
spoogeer
spoogees
spoogeing
spoogely
spooges
spunk
spunked
spunker
spunkes
spunking
spunkly
spunks
steamyed
steamyer
steamyes
steamying
steamyly
steamys
stfu
stfued
stfuer
stfues
stfuing
stfuly
stfus
stiffy
stiffyed
stiffyer
stiffyes
stiffying
stiffyly
stiffys
stoneded
stoneder
stonedes
stoneding
stonedly
stoneds
stupided
stupider
stupides
stupiding
stupidly
stupids
suckeded
suckeder
suckedes
suckeding
suckedly
suckeds
sucker
suckes
sucking
suckinged
suckinger
suckinges
suckinging
suckingly
suckings
suckly
sucks
sumofabiatch
sumofabiatched
sumofabiatcher
sumofabiatches
sumofabiatching
sumofabiatchly
sumofabiatchs
tard
tarded
tarder
tardes
tarding
tardly
tards
tawdry
tawdryed
tawdryer
tawdryes
tawdrying
tawdryly
tawdrys
teabagging
teabagginged
teabagginger
teabagginges
teabagginging
teabaggingly
teabaggings
terd
terded
terder
terdes
terding
terdly
terds
teste
testee
testeed
testeeed
testeeer
testeees
testeeing
testeely
testeer
testees
testeing
testely
testes
testesed
testeser
testeses
testesing
testesly
testess
testicle
testicleed
testicleer
testiclees
testicleing
testiclely
testicles
testis
testised
testiser
testises
testising
testisly
testiss
thrusted
thruster
thrustes
thrusting
thrustly
thrusts
thug
thuged
thuger
thuges
thuging
thugly
thugs
tinkle
tinkleed
tinkleer
tinklees
tinkleing
tinklely
tinkles
tit
tited
titer
tites
titfuck
titfucked
titfucker
titfuckes
titfucking
titfuckly
titfucks
titi
titied
titier
tities
titiing
titily
titing
titis
titly
tits
titsed
titser
titses
titsing
titsly
titss
tittiefucker
tittiefuckered
tittiefuckerer
tittiefuckeres
tittiefuckering
tittiefuckerly
tittiefuckers
titties
tittiesed
tittieser
tittieses
tittiesing
tittiesly
tittiess
titty
tittyed
tittyer
tittyes
tittyfuck
tittyfucked
tittyfucker
tittyfuckered
tittyfuckerer
tittyfuckeres
tittyfuckering
tittyfuckerly
tittyfuckers
tittyfuckes
tittyfucking
tittyfuckly
tittyfucks
tittying
tittyly
tittys
toke
tokeed
tokeer
tokees
tokeing
tokely
tokes
toots
tootsed
tootser
tootses
tootsing
tootsly
tootss
tramp
tramped
tramper
trampes
tramping
tramply
tramps
transsexualed
transsexualer
transsexuales
transsexualing
transsexually
transsexuals
trashy
trashyed
trashyer
trashyes
trashying
trashyly
trashys
tubgirl
tubgirled
tubgirler
tubgirles
tubgirling
tubgirlly
tubgirls
turd
turded
turder
turdes
turding
turdly
turds
tush
tushed
tusher
tushes
tushing
tushly
tushs
twat
twated
twater
twates
twating
twatly
twats
twatsed
twatser
twatses
twatsing
twatsly
twatss
undies
undiesed
undieser
undieses
undiesing
undiesly
undiess
unweded
unweder
unwedes
unweding
unwedly
unweds
uzi
uzied
uzier
uzies
uziing
uzily
uzis
vag
vaged
vager
vages
vaging
vagly
vags
valium
valiumed
valiumer
valiumes
valiuming
valiumly
valiums
venous
virgined
virginer
virgines
virgining
virginly
virgins
vixen
vixened
vixener
vixenes
vixening
vixenly
vixens
vodkaed
vodkaer
vodkaes
vodkaing
vodkaly
vodkas
voyeur
voyeured
voyeurer
voyeures
voyeuring
voyeurly
voyeurs
vulgar
vulgared
vulgarer
vulgares
vulgaring
vulgarly
vulgars
wang
wanged
wanger
wanges
wanging
wangly
wangs
wank
wanked
wanker
wankered
wankerer
wankeres
wankering
wankerly
wankers
wankes
wanking
wankly
wanks
wazoo
wazooed
wazooer
wazooes
wazooing
wazooly
wazoos
wedgie
wedgieed
wedgieer
wedgiees
wedgieing
wedgiely
wedgies
weeded
weeder
weedes
weeding
weedly
weeds
weenie
weenieed
weenieer
weeniees
weenieing
weeniely
weenies
weewee
weeweeed
weeweeer
weeweees
weeweeing
weeweely
weewees
weiner
weinered
weinerer
weineres
weinering
weinerly
weiners
weirdo
weirdoed
weirdoer
weirdoes
weirdoing
weirdoly
weirdos
wench
wenched
wencher
wenches
wenching
wenchly
wenchs
wetback
wetbacked
wetbacker
wetbackes
wetbacking
wetbackly
wetbacks
whitey
whiteyed
whiteyer
whiteyes
whiteying
whiteyly
whiteys
whiz
whized
whizer
whizes
whizing
whizly
whizs
whoralicious
whoralicioused
whoraliciouser
whoraliciouses
whoraliciousing
whoraliciously
whoraliciouss
whore
whorealicious
whorealicioused
whorealiciouser
whorealiciouses
whorealiciousing
whorealiciously
whorealiciouss
whored
whoreded
whoreder
whoredes
whoreding
whoredly
whoreds
whoreed
whoreer
whorees
whoreface
whorefaceed
whorefaceer
whorefacees
whorefaceing
whorefacely
whorefaces
whorehopper
whorehoppered
whorehopperer
whorehopperes
whorehoppering
whorehopperly
whorehoppers
whorehouse
whorehouseed
whorehouseer
whorehousees
whorehouseing
whorehousely
whorehouses
whoreing
whorely
whores
whoresed
whoreser
whoreses
whoresing
whoresly
whoress
whoring
whoringed
whoringer
whoringes
whoringing
whoringly
whorings
wigger
wiggered
wiggerer
wiggeres
wiggering
wiggerly
wiggers
woody
woodyed
woodyer
woodyes
woodying
woodyly
woodys
wop
woped
woper
wopes
woping
woply
wops
wtf
wtfed
wtfer
wtfes
wtfing
wtfly
wtfs
xxx
xxxed
xxxer
xxxes
xxxing
xxxly
xxxs
yeasty
yeastyed
yeastyer
yeastyes
yeastying
yeastyly
yeastys
yobbo
yobboed
yobboer
yobboes
yobboing
yobboly
yobbos
zoophile
zoophileed
zoophileer
zoophilees
zoophileing
zoophilely
zoophiles
anal
ass
ass lick
balls
ballsac
bisexual
bleach
causas
cheap
cost of miracles
cunt
display network stats
fart
fda and death
fda AND warn
fda AND warning
fda AND warns
feom
fuck
gfc
humira AND expensive
illegal
madvocate
masturbation
nuccitelli
overdose
porn
shit
snort
texarkana
Bipolar depression
Depression
adolescent depression
adolescent major depressive disorder
adolescent schizophrenia
adolescent with major depressive disorder
animals
autism
baby
brexpiprazole
child
child bipolar
child depression
child schizophrenia
children with bipolar disorder
children with depression
children with major depressive disorder
compulsive behaviors
cure
elderly bipolar
elderly depression
elderly major depressive disorder
elderly schizophrenia
elderly with dementia
first break
first episode
gambling
gaming
geriatric depression
geriatric major depressive disorder
geriatric schizophrenia
infant
kid
major depressive disorder
major depressive disorder in adolescents
major depressive disorder in children
parenting
pediatric
pediatric bipolar
pediatric depression
pediatric major depressive disorder
pediatric schizophrenia
pregnancy
pregnant
rexulti
skin care
teen
wine
section[contains(@class, 'nav-hidden')]
footer[@id='footer']
div[contains(@class, 'pane-node-field-article-topics')]
section[contains(@class, 'footer-nav-section-wrapper')]
section[contains(@class, 'content-row')]
div[contains(@class, 'panel-pane pane-article-read-next')]
A peer-reviewed clinical journal serving healthcare professionals working with the Department of Veterans Affairs, the Department of Defense, and the Public Health Service.
The Year of AI: Learning With Machines to Improve Veteran Health Care
The Year of AI: Learning With Machines to Improve Veteran Health Care
We have a tradition at Federal Practitioner where the December editorial usually features some version of the “best and worst” of the last 12 months in government health care. As we close out a difficult year, instead I offer a cautionary yet promising story that epitomizes both risk and benefit.
In some quarters, 2024 has been the year of AI (artificial intelligence).2 While in science fiction, superhuman machines, like the Terminator, are often associated with apocalyptic threats, we often forget the positive models of human-technology interaction, such as the protective robot in Lost in Space. While AI is not yet as advanced as what has already been depicted on the screen, it is inextricably interwoven into the daily fabric of our lives. Almost any website you go to for business or pleasure has a chatbot waiting to help (or frustrate) you. Most of us have Alexa, Siri, or another digital assistant organizing our homes and schedules. When I Google “everyday uses of artificial intelligence,” it is AI that responds with an overview.
Medicine is not immune. Renowned physician and scientist Eric Topol, MD, suggests that AI represents a “fourth industrial revolution in medicine” that can dramatically improve health care.3 The US Department of Veterans Affairs (VA) has been at the forefront of this new space.4 The story recounted below encapsulates the enormous benefits AI can bring to health care and the vigilance we must exercise to anticipate and mitigate risk for this to be an overall positive transition.
The story begins with a key element of AI change—the machine learning predictive algorithm. In this case, the algorithm was designed to predict—and thereby prevent—the top public health priority in federal practice: suicide. The Recovery Engagement and Coordination for Health-Veterans Enhanced Treatment (REACH VET) program was launched in 2017 to assist in identifying the top 0.1% of veterans at the highest risk for suicide.5
At least at this stage of AI in medicine, the safest and most ethical efforts come from collaborations between health care professionals and AI developers that maximize the very different strengths of each partner. REACH VET is an exemplar of this kind of teamwork. Once the algorithm analyzes > 60 variables to identify veterans at high risk for suicide, data are communicated to a REACH VET program coordinator, who then notifies the practitioner responsible for the veteran’s care so they can put into action evidence-based suicide prevention strategies.5
VA researchers in 2021 published a study of 173,313 veterans comparing outcomes before and after entry into the program using a triple differences design. Veterans participating in the program reported an increase in outpatient visits and documentation of safety plans, and a decrease in emergency department visits, inpatient mental health admissions, and recorded suicide attempts.6
A US Government Accounting Office analysis found that “REACH VET had identified veterans who had not been identified through other methods.”7 This was not just an example of AI hype: as a relatively rare and statistically complicated phenomenon, suicide is notoriously difficult to predict and model. Machine learning algorithms like REACH VET have unprecedented potential to assist and augment suicide prevention.8
In 2023, veteran service organizations and journalists raised concerns that the AI algorithm was biased and ignored critical risk factors that put some veterans at increased risk. Based on their analysis, they claimed that the algorithm did not account for risk factors uniquely associated with women veterans, namely military sexual trauma and intimate partner violence.9 Women are the most rapidly growing VA population, yet too often they encounter health care disparities, harassment, and stigmatization when seeking care. The Congressional Veterans Affairs committees investigated and introduced legislation to update the algorithm.10
VA experts dispute these claims, and a computer science PhD may be required to understand the debate. But as the history of medicine has shown us, every treatment and procedure has benefits and risks. No matter how bright and shiny the technology initially appears, a soft scientific underbelly emerges sooner or later. Just as with REACH VET, algorithm bias is often discovered during deployment when the logic of the laboratory encounters the unpredictable variety of humankind.11 Frequently, those problems are—as with REACH VET— not solely or even primarily technical ones. The data mirror society and reflect its biases.
For learning organizations like the VA and the US Department of Defense (DoD), the criticisms of REACH VET signal the need to engage in continuous performance improvement. AI requires the human trainers and supervisors who teach the machines to continuously revise and update their lesson plans. The most recent VA data show that in 2021, 6392 veterans died by suicide.12 In Congressional testimony, VA leaders reported that as of May 2024, REACH VET was operating in 28 VA facilities and had identified 6700 high-risk veterans.13 REACH VET can save veteran’s lives, which is the sine qua non for our federal health care systems.
The algorithm should be improved to identify ALL veterans so they receive lifesaving interventions. Every veteran’s life is sacred; the algorithm that may prevent suicide must be continuously improved. That is why our representatives did not propose to ban REACH VET or enforce an AI winter on the VA and DoD. Instead, they called for an update to the algorithm, underscoring the value of machine learning for suicide prediction and prevention.
The epigraph from one of the top AI ethicists and scientists in the world makes the point that AI is not the moral agent here: it is fallible humans who must keep learning along with machines. That is why, at the end of 2024, VA experts are revising the algorithm so REACH VET can help prevent even more veteran suicides in 2025 and beyond.14
- Waikar S. Health care’s AI future: a conversation with Fei Fei Li and Andrew Ng. HAI Stanford University. May 10, 2021. Accessed November 13, 2024. https://hai.stanford.edu/news/health-cares-ai-future-conversation-fei-fei-li-and-andrew-ng
- Johnson E, Forbes Technology Council. 2023 Was the Year of AI Hype—2024 is the Year of AI Practicality. Forbes. April 2, 2024. Accessed November 13, 2024. https://www.forbes.com/councils/forbestechcouncil/2024/04/02/2023-was-the-year-of-ai-hype-2024-is-the-year-of-ai-practicality/
- Topol E. Deep Medicine: How Artificial Intelligence Can Make Healthcare Human Again. Basic Books; 2019.
- Perlis R. The VA was an early adopter of artificial intelligence to improve care-here’s what they learned. JAMA. 2024;332(17):1411-1414. doi:10.1001/jama.2024.20563
- VA REACH VET initiative helps save lives [press release]. April 3, 2017. Accessed November 13, 2024. https://news.va.gov/36714/va-reach-vet-initiative-helps-save-veterans-lives/
- McCarthy JF, Cooper SA, Dent KR, et al. Evaluation of the recovery engagement and coordination for health-veterans enhanced treatment suicide risk modeling clinical program in the Veterans Health Administration. JAMA Netw Open. 2021;4(10):e2129900. doi:10.1001/jamanetworkopen.2021.29900
- US Government Office of Accountability. Veteran suicide: VA efforts to identify veterans at risk through analysis of health record information. September 14, 2022. Accessed November 13, 2024. https://www.gao.gov/products/gao-22-105165
- Pigoni A, Delvecchio G, Turtulici N, et al. Machine learning and the prediction of suicide in psychiatric populations: a systematic review. Transl Psychiatry. 2024;14(1):140. doi:10.1038/s41398-024-02852-9
- Glantz A. VA veteran suicide prevention algorithm favors men. Military.com. May 23, 2024. Accessed November 13, 2024. https://www.military.com/daily-news/2024/05/23/vas-veteran-suicide-prevention-algorithm-favors-men.html
- S.5210 BRAVE Act of 2024. 118th Congress. https://www.congress.gov/bill/118th-congress/senate-bill/5210/text
- Ratwani RM, Sutton K, and Galarrga JE. Addressing algorithmic bias in health care. JAMA. 2024;332(13):1051-1052. doi:10.1001/jama.2024.1348/
- US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention. 2023 national veteran suicide prevention annual report. November 2023 Accessed November 13, 2024. https://www.mentalhealth.va.gov/docs/data-sheets/2023/2023-National-Veteran-Suicide-Prevention-Annual-Report-FINAL-508.pdf
- House Committee on Veterans Affairs. Health Chairwoman Miller-Meeks opens Iowa field hearing on breakthroughs in VA healthcare. May 13, 2024. Accessed November 13, 2024. https://veterans.house.gov/news/documentsingle.aspx?DocumentID=6452
- Graham E. VA is updating its AI suicide risk model to reach more women. NEXTGOV/FCW. October 18, 2024. Accessed November 13, 2024. https://www.nextgov.com/artificial-intelligence/2024/10/va-updating-its-ai-suicide-risk-model-reach-more-women/400377/
We have a tradition at Federal Practitioner where the December editorial usually features some version of the “best and worst” of the last 12 months in government health care. As we close out a difficult year, instead I offer a cautionary yet promising story that epitomizes both risk and benefit.
In some quarters, 2024 has been the year of AI (artificial intelligence).2 While in science fiction, superhuman machines, like the Terminator, are often associated with apocalyptic threats, we often forget the positive models of human-technology interaction, such as the protective robot in Lost in Space. While AI is not yet as advanced as what has already been depicted on the screen, it is inextricably interwoven into the daily fabric of our lives. Almost any website you go to for business or pleasure has a chatbot waiting to help (or frustrate) you. Most of us have Alexa, Siri, or another digital assistant organizing our homes and schedules. When I Google “everyday uses of artificial intelligence,” it is AI that responds with an overview.
Medicine is not immune. Renowned physician and scientist Eric Topol, MD, suggests that AI represents a “fourth industrial revolution in medicine” that can dramatically improve health care.3 The US Department of Veterans Affairs (VA) has been at the forefront of this new space.4 The story recounted below encapsulates the enormous benefits AI can bring to health care and the vigilance we must exercise to anticipate and mitigate risk for this to be an overall positive transition.
The story begins with a key element of AI change—the machine learning predictive algorithm. In this case, the algorithm was designed to predict—and thereby prevent—the top public health priority in federal practice: suicide. The Recovery Engagement and Coordination for Health-Veterans Enhanced Treatment (REACH VET) program was launched in 2017 to assist in identifying the top 0.1% of veterans at the highest risk for suicide.5
At least at this stage of AI in medicine, the safest and most ethical efforts come from collaborations between health care professionals and AI developers that maximize the very different strengths of each partner. REACH VET is an exemplar of this kind of teamwork. Once the algorithm analyzes > 60 variables to identify veterans at high risk for suicide, data are communicated to a REACH VET program coordinator, who then notifies the practitioner responsible for the veteran’s care so they can put into action evidence-based suicide prevention strategies.5
VA researchers in 2021 published a study of 173,313 veterans comparing outcomes before and after entry into the program using a triple differences design. Veterans participating in the program reported an increase in outpatient visits and documentation of safety plans, and a decrease in emergency department visits, inpatient mental health admissions, and recorded suicide attempts.6
A US Government Accounting Office analysis found that “REACH VET had identified veterans who had not been identified through other methods.”7 This was not just an example of AI hype: as a relatively rare and statistically complicated phenomenon, suicide is notoriously difficult to predict and model. Machine learning algorithms like REACH VET have unprecedented potential to assist and augment suicide prevention.8
In 2023, veteran service organizations and journalists raised concerns that the AI algorithm was biased and ignored critical risk factors that put some veterans at increased risk. Based on their analysis, they claimed that the algorithm did not account for risk factors uniquely associated with women veterans, namely military sexual trauma and intimate partner violence.9 Women are the most rapidly growing VA population, yet too often they encounter health care disparities, harassment, and stigmatization when seeking care. The Congressional Veterans Affairs committees investigated and introduced legislation to update the algorithm.10
VA experts dispute these claims, and a computer science PhD may be required to understand the debate. But as the history of medicine has shown us, every treatment and procedure has benefits and risks. No matter how bright and shiny the technology initially appears, a soft scientific underbelly emerges sooner or later. Just as with REACH VET, algorithm bias is often discovered during deployment when the logic of the laboratory encounters the unpredictable variety of humankind.11 Frequently, those problems are—as with REACH VET— not solely or even primarily technical ones. The data mirror society and reflect its biases.
For learning organizations like the VA and the US Department of Defense (DoD), the criticisms of REACH VET signal the need to engage in continuous performance improvement. AI requires the human trainers and supervisors who teach the machines to continuously revise and update their lesson plans. The most recent VA data show that in 2021, 6392 veterans died by suicide.12 In Congressional testimony, VA leaders reported that as of May 2024, REACH VET was operating in 28 VA facilities and had identified 6700 high-risk veterans.13 REACH VET can save veteran’s lives, which is the sine qua non for our federal health care systems.
The algorithm should be improved to identify ALL veterans so they receive lifesaving interventions. Every veteran’s life is sacred; the algorithm that may prevent suicide must be continuously improved. That is why our representatives did not propose to ban REACH VET or enforce an AI winter on the VA and DoD. Instead, they called for an update to the algorithm, underscoring the value of machine learning for suicide prediction and prevention.
The epigraph from one of the top AI ethicists and scientists in the world makes the point that AI is not the moral agent here: it is fallible humans who must keep learning along with machines. That is why, at the end of 2024, VA experts are revising the algorithm so REACH VET can help prevent even more veteran suicides in 2025 and beyond.14
We have a tradition at Federal Practitioner where the December editorial usually features some version of the “best and worst” of the last 12 months in government health care. As we close out a difficult year, instead I offer a cautionary yet promising story that epitomizes both risk and benefit.
In some quarters, 2024 has been the year of AI (artificial intelligence).2 While in science fiction, superhuman machines, like the Terminator, are often associated with apocalyptic threats, we often forget the positive models of human-technology interaction, such as the protective robot in Lost in Space. While AI is not yet as advanced as what has already been depicted on the screen, it is inextricably interwoven into the daily fabric of our lives. Almost any website you go to for business or pleasure has a chatbot waiting to help (or frustrate) you. Most of us have Alexa, Siri, or another digital assistant organizing our homes and schedules. When I Google “everyday uses of artificial intelligence,” it is AI that responds with an overview.
Medicine is not immune. Renowned physician and scientist Eric Topol, MD, suggests that AI represents a “fourth industrial revolution in medicine” that can dramatically improve health care.3 The US Department of Veterans Affairs (VA) has been at the forefront of this new space.4 The story recounted below encapsulates the enormous benefits AI can bring to health care and the vigilance we must exercise to anticipate and mitigate risk for this to be an overall positive transition.
The story begins with a key element of AI change—the machine learning predictive algorithm. In this case, the algorithm was designed to predict—and thereby prevent—the top public health priority in federal practice: suicide. The Recovery Engagement and Coordination for Health-Veterans Enhanced Treatment (REACH VET) program was launched in 2017 to assist in identifying the top 0.1% of veterans at the highest risk for suicide.5
At least at this stage of AI in medicine, the safest and most ethical efforts come from collaborations between health care professionals and AI developers that maximize the very different strengths of each partner. REACH VET is an exemplar of this kind of teamwork. Once the algorithm analyzes > 60 variables to identify veterans at high risk for suicide, data are communicated to a REACH VET program coordinator, who then notifies the practitioner responsible for the veteran’s care so they can put into action evidence-based suicide prevention strategies.5
VA researchers in 2021 published a study of 173,313 veterans comparing outcomes before and after entry into the program using a triple differences design. Veterans participating in the program reported an increase in outpatient visits and documentation of safety plans, and a decrease in emergency department visits, inpatient mental health admissions, and recorded suicide attempts.6
A US Government Accounting Office analysis found that “REACH VET had identified veterans who had not been identified through other methods.”7 This was not just an example of AI hype: as a relatively rare and statistically complicated phenomenon, suicide is notoriously difficult to predict and model. Machine learning algorithms like REACH VET have unprecedented potential to assist and augment suicide prevention.8
In 2023, veteran service organizations and journalists raised concerns that the AI algorithm was biased and ignored critical risk factors that put some veterans at increased risk. Based on their analysis, they claimed that the algorithm did not account for risk factors uniquely associated with women veterans, namely military sexual trauma and intimate partner violence.9 Women are the most rapidly growing VA population, yet too often they encounter health care disparities, harassment, and stigmatization when seeking care. The Congressional Veterans Affairs committees investigated and introduced legislation to update the algorithm.10
VA experts dispute these claims, and a computer science PhD may be required to understand the debate. But as the history of medicine has shown us, every treatment and procedure has benefits and risks. No matter how bright and shiny the technology initially appears, a soft scientific underbelly emerges sooner or later. Just as with REACH VET, algorithm bias is often discovered during deployment when the logic of the laboratory encounters the unpredictable variety of humankind.11 Frequently, those problems are—as with REACH VET— not solely or even primarily technical ones. The data mirror society and reflect its biases.
For learning organizations like the VA and the US Department of Defense (DoD), the criticisms of REACH VET signal the need to engage in continuous performance improvement. AI requires the human trainers and supervisors who teach the machines to continuously revise and update their lesson plans. The most recent VA data show that in 2021, 6392 veterans died by suicide.12 In Congressional testimony, VA leaders reported that as of May 2024, REACH VET was operating in 28 VA facilities and had identified 6700 high-risk veterans.13 REACH VET can save veteran’s lives, which is the sine qua non for our federal health care systems.
The algorithm should be improved to identify ALL veterans so they receive lifesaving interventions. Every veteran’s life is sacred; the algorithm that may prevent suicide must be continuously improved. That is why our representatives did not propose to ban REACH VET or enforce an AI winter on the VA and DoD. Instead, they called for an update to the algorithm, underscoring the value of machine learning for suicide prediction and prevention.
The epigraph from one of the top AI ethicists and scientists in the world makes the point that AI is not the moral agent here: it is fallible humans who must keep learning along with machines. That is why, at the end of 2024, VA experts are revising the algorithm so REACH VET can help prevent even more veteran suicides in 2025 and beyond.14
- Waikar S. Health care’s AI future: a conversation with Fei Fei Li and Andrew Ng. HAI Stanford University. May 10, 2021. Accessed November 13, 2024. https://hai.stanford.edu/news/health-cares-ai-future-conversation-fei-fei-li-and-andrew-ng
- Johnson E, Forbes Technology Council. 2023 Was the Year of AI Hype—2024 is the Year of AI Practicality. Forbes. April 2, 2024. Accessed November 13, 2024. https://www.forbes.com/councils/forbestechcouncil/2024/04/02/2023-was-the-year-of-ai-hype-2024-is-the-year-of-ai-practicality/
- Topol E. Deep Medicine: How Artificial Intelligence Can Make Healthcare Human Again. Basic Books; 2019.
- Perlis R. The VA was an early adopter of artificial intelligence to improve care-here’s what they learned. JAMA. 2024;332(17):1411-1414. doi:10.1001/jama.2024.20563
- VA REACH VET initiative helps save lives [press release]. April 3, 2017. Accessed November 13, 2024. https://news.va.gov/36714/va-reach-vet-initiative-helps-save-veterans-lives/
- McCarthy JF, Cooper SA, Dent KR, et al. Evaluation of the recovery engagement and coordination for health-veterans enhanced treatment suicide risk modeling clinical program in the Veterans Health Administration. JAMA Netw Open. 2021;4(10):e2129900. doi:10.1001/jamanetworkopen.2021.29900
- US Government Office of Accountability. Veteran suicide: VA efforts to identify veterans at risk through analysis of health record information. September 14, 2022. Accessed November 13, 2024. https://www.gao.gov/products/gao-22-105165
- Pigoni A, Delvecchio G, Turtulici N, et al. Machine learning and the prediction of suicide in psychiatric populations: a systematic review. Transl Psychiatry. 2024;14(1):140. doi:10.1038/s41398-024-02852-9
- Glantz A. VA veteran suicide prevention algorithm favors men. Military.com. May 23, 2024. Accessed November 13, 2024. https://www.military.com/daily-news/2024/05/23/vas-veteran-suicide-prevention-algorithm-favors-men.html
- S.5210 BRAVE Act of 2024. 118th Congress. https://www.congress.gov/bill/118th-congress/senate-bill/5210/text
- Ratwani RM, Sutton K, and Galarrga JE. Addressing algorithmic bias in health care. JAMA. 2024;332(13):1051-1052. doi:10.1001/jama.2024.1348/
- US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention. 2023 national veteran suicide prevention annual report. November 2023 Accessed November 13, 2024. https://www.mentalhealth.va.gov/docs/data-sheets/2023/2023-National-Veteran-Suicide-Prevention-Annual-Report-FINAL-508.pdf
- House Committee on Veterans Affairs. Health Chairwoman Miller-Meeks opens Iowa field hearing on breakthroughs in VA healthcare. May 13, 2024. Accessed November 13, 2024. https://veterans.house.gov/news/documentsingle.aspx?DocumentID=6452
- Graham E. VA is updating its AI suicide risk model to reach more women. NEXTGOV/FCW. October 18, 2024. Accessed November 13, 2024. https://www.nextgov.com/artificial-intelligence/2024/10/va-updating-its-ai-suicide-risk-model-reach-more-women/400377/
- Waikar S. Health care’s AI future: a conversation with Fei Fei Li and Andrew Ng. HAI Stanford University. May 10, 2021. Accessed November 13, 2024. https://hai.stanford.edu/news/health-cares-ai-future-conversation-fei-fei-li-and-andrew-ng
- Johnson E, Forbes Technology Council. 2023 Was the Year of AI Hype—2024 is the Year of AI Practicality. Forbes. April 2, 2024. Accessed November 13, 2024. https://www.forbes.com/councils/forbestechcouncil/2024/04/02/2023-was-the-year-of-ai-hype-2024-is-the-year-of-ai-practicality/
- Topol E. Deep Medicine: How Artificial Intelligence Can Make Healthcare Human Again. Basic Books; 2019.
- Perlis R. The VA was an early adopter of artificial intelligence to improve care-here’s what they learned. JAMA. 2024;332(17):1411-1414. doi:10.1001/jama.2024.20563
- VA REACH VET initiative helps save lives [press release]. April 3, 2017. Accessed November 13, 2024. https://news.va.gov/36714/va-reach-vet-initiative-helps-save-veterans-lives/
- McCarthy JF, Cooper SA, Dent KR, et al. Evaluation of the recovery engagement and coordination for health-veterans enhanced treatment suicide risk modeling clinical program in the Veterans Health Administration. JAMA Netw Open. 2021;4(10):e2129900. doi:10.1001/jamanetworkopen.2021.29900
- US Government Office of Accountability. Veteran suicide: VA efforts to identify veterans at risk through analysis of health record information. September 14, 2022. Accessed November 13, 2024. https://www.gao.gov/products/gao-22-105165
- Pigoni A, Delvecchio G, Turtulici N, et al. Machine learning and the prediction of suicide in psychiatric populations: a systematic review. Transl Psychiatry. 2024;14(1):140. doi:10.1038/s41398-024-02852-9
- Glantz A. VA veteran suicide prevention algorithm favors men. Military.com. May 23, 2024. Accessed November 13, 2024. https://www.military.com/daily-news/2024/05/23/vas-veteran-suicide-prevention-algorithm-favors-men.html
- S.5210 BRAVE Act of 2024. 118th Congress. https://www.congress.gov/bill/118th-congress/senate-bill/5210/text
- Ratwani RM, Sutton K, and Galarrga JE. Addressing algorithmic bias in health care. JAMA. 2024;332(13):1051-1052. doi:10.1001/jama.2024.1348/
- US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention. 2023 national veteran suicide prevention annual report. November 2023 Accessed November 13, 2024. https://www.mentalhealth.va.gov/docs/data-sheets/2023/2023-National-Veteran-Suicide-Prevention-Annual-Report-FINAL-508.pdf
- House Committee on Veterans Affairs. Health Chairwoman Miller-Meeks opens Iowa field hearing on breakthroughs in VA healthcare. May 13, 2024. Accessed November 13, 2024. https://veterans.house.gov/news/documentsingle.aspx?DocumentID=6452
- Graham E. VA is updating its AI suicide risk model to reach more women. NEXTGOV/FCW. October 18, 2024. Accessed November 13, 2024. https://www.nextgov.com/artificial-intelligence/2024/10/va-updating-its-ai-suicide-risk-model-reach-more-women/400377/
The Year of AI: Learning With Machines to Improve Veteran Health Care
The Year of AI: Learning With Machines to Improve Veteran Health Care
Could Diet and Gut Bacteria Be Fueling Early CRC?
This transcript has been edited for clarity.
I’d like to reflect a little on the ever-rising incidence of early-onset colorectal cancer. I saw two patients in the clinic on Friday, both in their early thirties, presenting with stage IV disease. Both had young families — a disaster.
This is an issue that we must address, I think, epidemiologically. We know that and currently, around 200,000 such cases are diagnosed every year, but it is said to increase unquestionably.
The epidemiologists, I think, correctly have identified that this sharp, rapid increase does imply that there is a new environmental change that is underpinning or underscoring this rise in early-onset disease.
There’s a fantastic team that has been put together by Paul Brennan, Mike Stratton, and colleagues, a collaborative group of epidemiologists, geneticists, and bioinformaticians, who are looking at a global study to try to understand the basis of early-onset colorectal cancer. Their approach is to combine conventional epidemiology, genomics, and fantastic computational support to try to unpick the mutational signatures involved.
The dominant hypothesis is that, over the past 20-25 years or so, there has been a change in diet that has allowed an alteration in the gut microbiome such that we now harbor, in some cases, more bacteria capable of manufacturing, synthesizing, and releasing mutagenic chemicals. There’s a subtype of Escherichia coli which manufactures one such mutagen called colibactin.
Again, through some of the painstaking, extraordinary work that Mike Stratton and colleagues have done at the Sanger Institute, they have managed to, using a variety of different techniques — in vitro, observational, and so on — relate exposure to the mutagen colibactin to a particular mutational signature.
They plan to do a large global study — one of the strengths — involving many different countries around the globe, collect material from older colorectal cancer patients and early-onset colorectal cancer patients, and undertake a staggeringly large mutational study to see if the mutational signature associated with colibactin is more highly represented in these early-onset cases. The hypothesis is that, if you’re exposed to this mutagen in childhood, then it increases the tumor mutational burden and therefore the likelihood of developing cancer at an earlier age.
All of us believe that converting a normal cell into a tumor cell usually requires five or six or seven separate mutational events occurring at random. The earlier these occur, the greater the tumor, the greater the normal single-cellular mutational burden, and the more likely it is to develop cancer sooner rather than later.
This is a fantastically interesting study, and it’s the way ahead with modern genetic epidemiology, one would say. We wish them well. This will be a 3- to 5-year truly international effort, bringing together a genuinely internationally outstanding research team. We hope that they are able to shed more light on the epidemiology of this early-onset disease, because only by understanding can we deflect and deal with it.
Knowledge is power, as I’ve said many times before. If we understand the underlying epidemiology, that will allow us to intervene, one would hope, and avoid the chaotic disaster of my clinic on Friday, with these two young patients with an extremely limited lifespan and large families who will be left bereft in having lost a parent.
More power to the team. We wish them well with the study, but again, this is a pointer to the future, one would hope, of modern genetic computational epidemiology.
I’d be really interested in any ideas or comments that you might have. Are you in the field? Are you seeing more young patients? Do you have any ideas or hypotheses of your own around the microbiome and what bugs might be involved and so on?
Dr. Kerr, Professor, Nuffield Department of Clinical Laboratory Science, University of Oxford, England; Professor of Cancer Medicine, Oxford Cancer Centre, Oxford, United Kingdom, has disclosed relevant financial relationships with Celleron Therapeutics, Oxford Cancer Biomarkers, Afrox, GlaxoSmithKline, Bayer, Genomic Health, Merck Serono, and Roche.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I’d like to reflect a little on the ever-rising incidence of early-onset colorectal cancer. I saw two patients in the clinic on Friday, both in their early thirties, presenting with stage IV disease. Both had young families — a disaster.
This is an issue that we must address, I think, epidemiologically. We know that and currently, around 200,000 such cases are diagnosed every year, but it is said to increase unquestionably.
The epidemiologists, I think, correctly have identified that this sharp, rapid increase does imply that there is a new environmental change that is underpinning or underscoring this rise in early-onset disease.
There’s a fantastic team that has been put together by Paul Brennan, Mike Stratton, and colleagues, a collaborative group of epidemiologists, geneticists, and bioinformaticians, who are looking at a global study to try to understand the basis of early-onset colorectal cancer. Their approach is to combine conventional epidemiology, genomics, and fantastic computational support to try to unpick the mutational signatures involved.
The dominant hypothesis is that, over the past 20-25 years or so, there has been a change in diet that has allowed an alteration in the gut microbiome such that we now harbor, in some cases, more bacteria capable of manufacturing, synthesizing, and releasing mutagenic chemicals. There’s a subtype of Escherichia coli which manufactures one such mutagen called colibactin.
Again, through some of the painstaking, extraordinary work that Mike Stratton and colleagues have done at the Sanger Institute, they have managed to, using a variety of different techniques — in vitro, observational, and so on — relate exposure to the mutagen colibactin to a particular mutational signature.
They plan to do a large global study — one of the strengths — involving many different countries around the globe, collect material from older colorectal cancer patients and early-onset colorectal cancer patients, and undertake a staggeringly large mutational study to see if the mutational signature associated with colibactin is more highly represented in these early-onset cases. The hypothesis is that, if you’re exposed to this mutagen in childhood, then it increases the tumor mutational burden and therefore the likelihood of developing cancer at an earlier age.
All of us believe that converting a normal cell into a tumor cell usually requires five or six or seven separate mutational events occurring at random. The earlier these occur, the greater the tumor, the greater the normal single-cellular mutational burden, and the more likely it is to develop cancer sooner rather than later.
This is a fantastically interesting study, and it’s the way ahead with modern genetic epidemiology, one would say. We wish them well. This will be a 3- to 5-year truly international effort, bringing together a genuinely internationally outstanding research team. We hope that they are able to shed more light on the epidemiology of this early-onset disease, because only by understanding can we deflect and deal with it.
Knowledge is power, as I’ve said many times before. If we understand the underlying epidemiology, that will allow us to intervene, one would hope, and avoid the chaotic disaster of my clinic on Friday, with these two young patients with an extremely limited lifespan and large families who will be left bereft in having lost a parent.
More power to the team. We wish them well with the study, but again, this is a pointer to the future, one would hope, of modern genetic computational epidemiology.
I’d be really interested in any ideas or comments that you might have. Are you in the field? Are you seeing more young patients? Do you have any ideas or hypotheses of your own around the microbiome and what bugs might be involved and so on?
Dr. Kerr, Professor, Nuffield Department of Clinical Laboratory Science, University of Oxford, England; Professor of Cancer Medicine, Oxford Cancer Centre, Oxford, United Kingdom, has disclosed relevant financial relationships with Celleron Therapeutics, Oxford Cancer Biomarkers, Afrox, GlaxoSmithKline, Bayer, Genomic Health, Merck Serono, and Roche.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I’d like to reflect a little on the ever-rising incidence of early-onset colorectal cancer. I saw two patients in the clinic on Friday, both in their early thirties, presenting with stage IV disease. Both had young families — a disaster.
This is an issue that we must address, I think, epidemiologically. We know that and currently, around 200,000 such cases are diagnosed every year, but it is said to increase unquestionably.
The epidemiologists, I think, correctly have identified that this sharp, rapid increase does imply that there is a new environmental change that is underpinning or underscoring this rise in early-onset disease.
There’s a fantastic team that has been put together by Paul Brennan, Mike Stratton, and colleagues, a collaborative group of epidemiologists, geneticists, and bioinformaticians, who are looking at a global study to try to understand the basis of early-onset colorectal cancer. Their approach is to combine conventional epidemiology, genomics, and fantastic computational support to try to unpick the mutational signatures involved.
The dominant hypothesis is that, over the past 20-25 years or so, there has been a change in diet that has allowed an alteration in the gut microbiome such that we now harbor, in some cases, more bacteria capable of manufacturing, synthesizing, and releasing mutagenic chemicals. There’s a subtype of Escherichia coli which manufactures one such mutagen called colibactin.
Again, through some of the painstaking, extraordinary work that Mike Stratton and colleagues have done at the Sanger Institute, they have managed to, using a variety of different techniques — in vitro, observational, and so on — relate exposure to the mutagen colibactin to a particular mutational signature.
They plan to do a large global study — one of the strengths — involving many different countries around the globe, collect material from older colorectal cancer patients and early-onset colorectal cancer patients, and undertake a staggeringly large mutational study to see if the mutational signature associated with colibactin is more highly represented in these early-onset cases. The hypothesis is that, if you’re exposed to this mutagen in childhood, then it increases the tumor mutational burden and therefore the likelihood of developing cancer at an earlier age.
All of us believe that converting a normal cell into a tumor cell usually requires five or six or seven separate mutational events occurring at random. The earlier these occur, the greater the tumor, the greater the normal single-cellular mutational burden, and the more likely it is to develop cancer sooner rather than later.
This is a fantastically interesting study, and it’s the way ahead with modern genetic epidemiology, one would say. We wish them well. This will be a 3- to 5-year truly international effort, bringing together a genuinely internationally outstanding research team. We hope that they are able to shed more light on the epidemiology of this early-onset disease, because only by understanding can we deflect and deal with it.
Knowledge is power, as I’ve said many times before. If we understand the underlying epidemiology, that will allow us to intervene, one would hope, and avoid the chaotic disaster of my clinic on Friday, with these two young patients with an extremely limited lifespan and large families who will be left bereft in having lost a parent.
More power to the team. We wish them well with the study, but again, this is a pointer to the future, one would hope, of modern genetic computational epidemiology.
I’d be really interested in any ideas or comments that you might have. Are you in the field? Are you seeing more young patients? Do you have any ideas or hypotheses of your own around the microbiome and what bugs might be involved and so on?
Dr. Kerr, Professor, Nuffield Department of Clinical Laboratory Science, University of Oxford, England; Professor of Cancer Medicine, Oxford Cancer Centre, Oxford, United Kingdom, has disclosed relevant financial relationships with Celleron Therapeutics, Oxford Cancer Biomarkers, Afrox, GlaxoSmithKline, Bayer, Genomic Health, Merck Serono, and Roche.
A version of this article appeared on Medscape.com.
Pharmacist-Driven Deprescribing to Reduce Anticholinergic Burden in Veterans With Dementia
Pharmacist-Driven Deprescribing to Reduce Anticholinergic Burden in Veterans With Dementia
Anticholinergic medications block the activity of the neurotransmitter acetylcholine by binding to either muscarinic or nicotinic receptors in both the peripheral and central nervous system. Anticholinergic medications typically refer to antimuscarinic medications and have been prescribed to treat a variety of conditions common in older adults, including overactive bladder, allergies, muscle spasms, and sleep disorders.1,2 Since muscarinic receptors are present throughout the body, anticholinergic medications are associated with many adverse effects (AEs), including constipation, urinary retention, xerostomia, and delirium. Older adults are more sensitive to these AEs due to physiological changes associated with aging.1
The American Geriatric Society Beers Criteria for Potentially Inappropriate Medications Use in Older Adults identifies drugs with strong anticholinergic properties. The Beers Criteria strongly recommends avoiding these medications in patients with dementia or cognitive impairment due to the risk of central nervous system AEs. In the updated 2023 Beers Criteria, the rationale was expanded to recognize the risks of the cumulative anticholinergic burden associated with concurrent anticholinergic use.3,4
Given the prevalent use of anticholinergic medications in older adults, there has been significant research demonstrating their AEs, specifically delirium and cognitive impairment in geriatric patients. A systematic review of 14 articles conducted in 7 different countries of patients with median age of 76.4 to 86.1 years reviewed clinical outcomes of anticholinergic use in patients with dementia. Five studies found anticholinergics were associated with increased all-cause mortality in patients with dementia, and 3 studies found anticholinergics were associated with longer hospital stays. Other studies found that anticholinergics were associated with delirium and reduced health-related quality of life.5
About 35% of veterans with dementia have been prescribed a medication regimen with a high anticholinergic burden.6 In 2018, the US Department of Veterans Affairs (VA) Pharmacy Benfits Management Center for Medical Safety completed a centrally aggregated medication use evaluation (CAMUE) to assess the appropriateness of anticholinergic medication use in patients with dementia. The retrospective chart review included 1094 veterans from 19 sites. Overall, about 15% of the veterans experienced new falls, delirium, or worsening dementia within 30 days of starting an anticholinergic medication. Furthermore, < 40% had documentation of a nonanticholinergic alternative medication trial, and < 20% had documented nonpharmacologic therapy. The documentation of risk-benefit assessment acknowledging the risks of anticholinergic medication use in veterans with dementia occurred only about 13% of the time. The CAMUE concluded that the risks of initiating an anticholinergic medication in veterans with dementia are likely underdocumented and possibly under considered by prescribers.7
Developed within the Veterans Health Administration (VHA), VIONE (Vital, Important, Optional, Not Indicated, Every medication has an indication) is a medication management methodology that aims to reduce polypharmacy and improve patient safety consistent with high-reliability organizations. Since it launched in 2016, VIONE has gradually been implemented at many VHA facilities. The VIONE deprescribing dashboard had not been used at the VA Louisville Healthcare System prior to this quality improvement project.
This dashboard uses the Beers Criteria to identify potentially inappropriate anticholinergic medications. It uses the Anticholinergic Cognitive Burden (ACB) scale to calculate the cumulative anticholinergic risk for each patient. Medications with an ACB score of 2 or 3 have clinically relevant cognitive effects such as delirium and dementia (Table 1). For each point increase in total ACB score, a decline in mini-mental state examination score of 0.33 points over 2 years has been shown. Each point increase has also been correlated with a 26% increase in risk of death.8-10
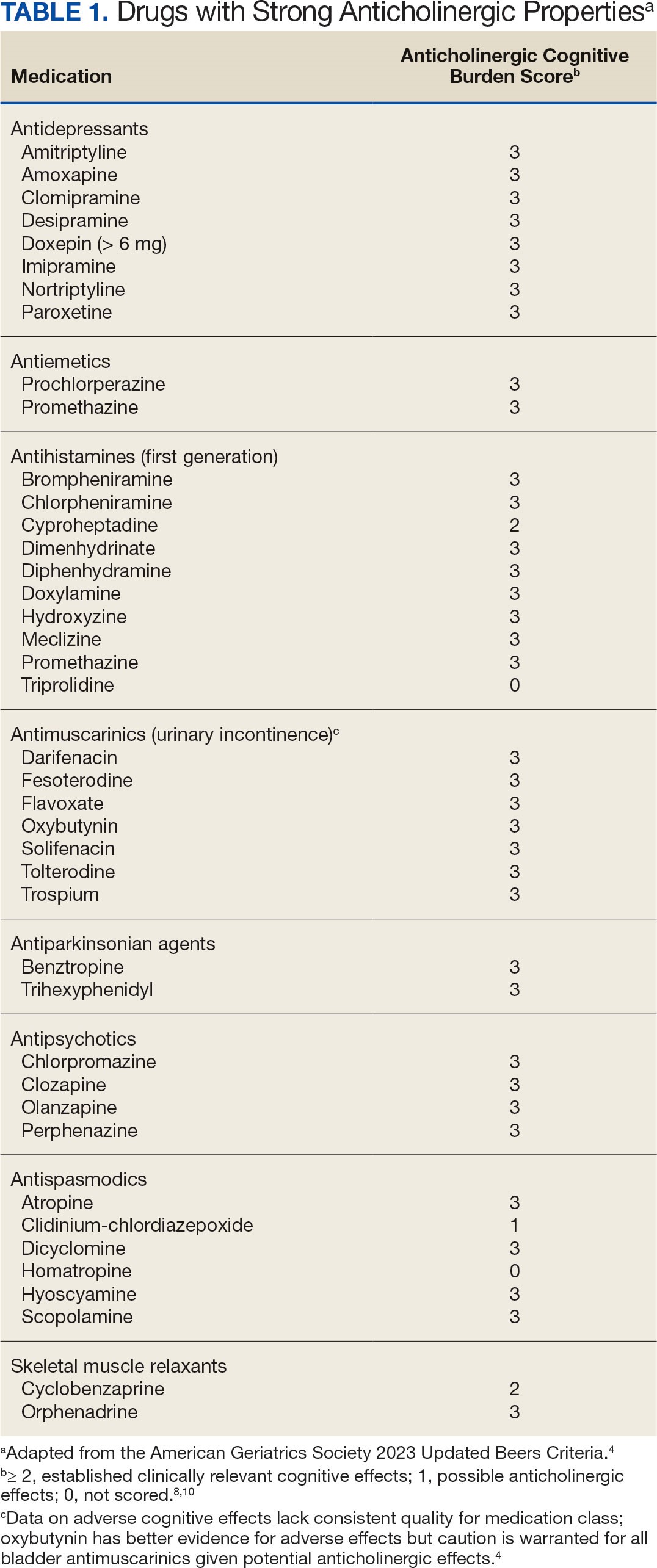
Methods
The purpose of this quality improvement project was to determine the impact of pharmacist-driven deprescribing on the anticholinergic burden in veterans with dementia at VA Louisville Healthcare System. Data were obtained through the Computerized Patient Record System (CPRS) and VIONE deprescribing dashboard and entered in a secure Microsoft Excel spreadsheet. Pharmacist deprescribing steps were entered as CPRS progress notes. A deprescribing note template was created, and 11 templates with indication-specific recommendations were created for each anticholinergic indication identified (contact authors for deprescribing note template examples). Usage of anticholinergic medications was reexamined 3 months after the deprescribing note was entered.
Eligible patients identified in the VIONE deprescribing dashboard had an outpatient order for a medication with strong anticholinergic properties as identified using the Beers Criteria and were aged ≥ 65 years. Patients also had to be diagnosed with dementia or cognitive impairment. Patients were excluded if they were receiving hospice care or if the anticholinergic medication was from a non-VA prescriber or filled at a non-VA pharmacy. The VIONE deprescribing dashboard also excluded skeletal muscle relaxants if the patient had a spinal cord-related visit in the previous 2 years, first-generation antihistamines if the patient had a vertigo diagnosis, hydroxyzine if the indication was for anxiety, trospium if the indication was for overactive bladder, and antipsychotics if the patient had been diagnosed with schizophrenia or bipolar disorder. The following were included in the deprescribing recommendations if the dashboard identified the patient due to receiving a second strongly anticholinergic medication: first generation antihistamines if the patient was diagnosed with vertigo and hydroxyzine if the indication is for anxiety.
Each eligible patient received a focused medication review by a pharmacist via electronic chart review and a templated CPRS progress note with patient-specific recommendations. The prescriber and the patient’s primary care practitioner were recommended to perform a patient-specific risk-benefit assessment, deprescribe potentially inappropriate anticholinergic medications, and consider nonanticholinergic alternatives (both pharmacologic and nonpharmacologic). Data collected included baseline age, sex, prespecified comorbidities (type of dementia, cognitive impairment, delirium, benign prostatic hyperplasia/lower urinary tract symptoms), duration of prescribed anticholinergic medication, indication and deprescribing rate for each anticholinergic agent, and concurrent dementia medications (acetylcholinesterase inhibitors, memantine, or both).
The primary outcome was the number of patients that had = 1 medication with strong anticholinergic properties deprescribed. Deprescribing was defined as medication discontinuation or reduction of total daily dose. Secondary outcomes were the mean change in ACB scale, the number of patients with dose tapering, documented patient-specific risk-benefit assessment, and initiated nonanticholinergic alternative per pharmacist recommendation.
Results
The VIONE deprescribing dashboard identified 121 patients; 45 were excluded for non-VA prescriber or pharmacy, and 8 patients were excluded for other reasons. Sixty-eight patients were included in the deprescribing initiative. The mean age was 73.4 years (range, 67-93), 65 (96%) were male, and 34 (50%) had unspecified dementia (Table 2). Thirty-one patients (46%) had concurrent cholinesterase inhibitor prescriptions for dementia. The median duration of use of a strong anticholinergic medication was 11 months.
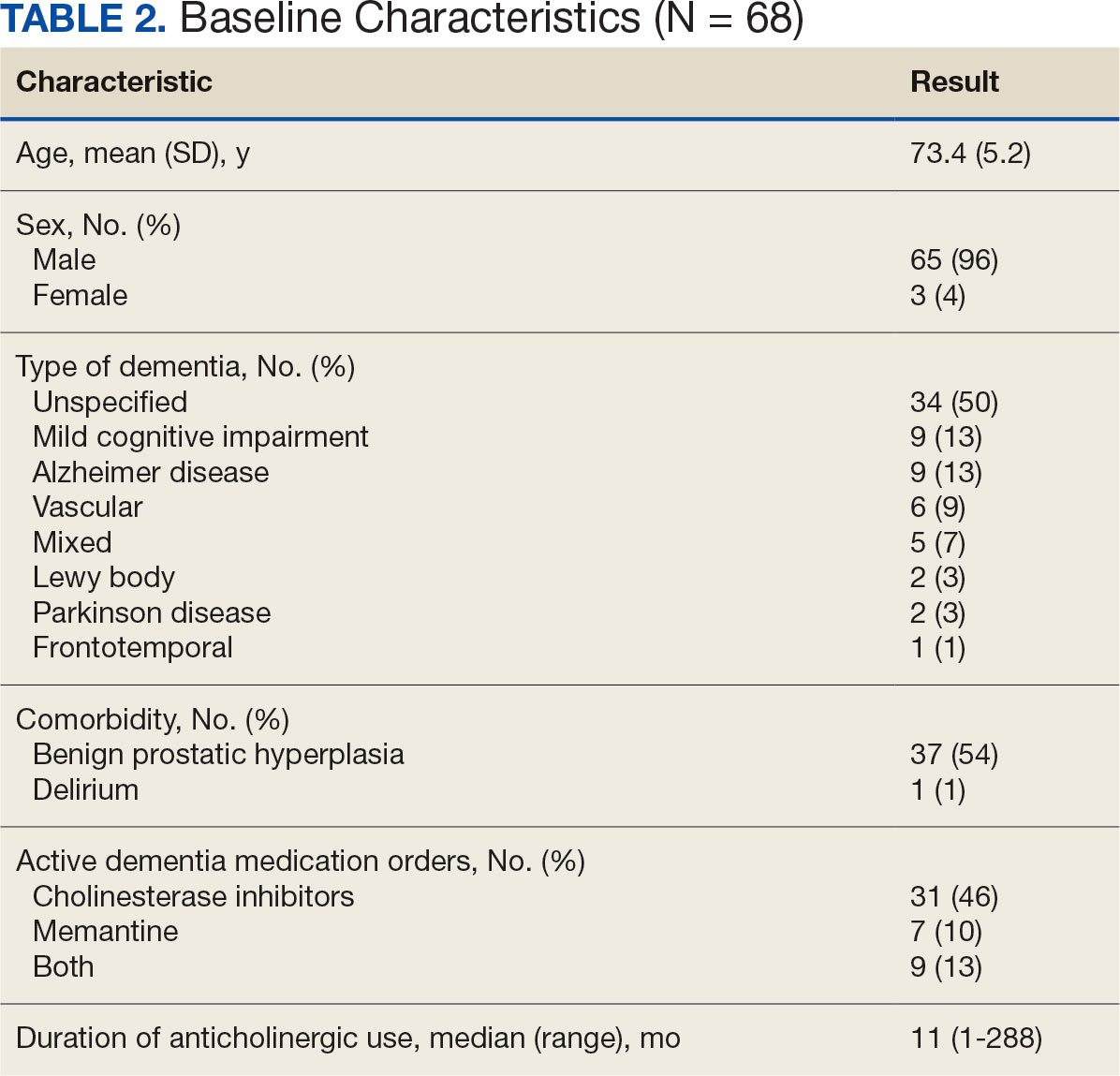
Twenty-nine patients (43%) had ≥ 1 medication with strong anticholinergic properties deprescribed. Anticholinergic medication was discontinued for 26 patients, and the dose was decreased for 3 patients. ACB score fell by a mean of 1.1 per patient. There was an increase in the documented risk-benefit assessment for anticholinergic medications from a baseline of 4 (6%) to 19 (28%) 3 months after the deprescribing note. Cyclobenzaprine, paroxetine, and oxybutynin were deprescribed the most, and amitriptyline had the lowest rate of deprescribing (Table 3). Thirty patients (44%) had a pharmacologic, nonanticholinergic alternative initiated per pharmacist recommendation, and 6 patients (9%) had a nonpharmacologic alternative initiated per pharmacist recommendation.
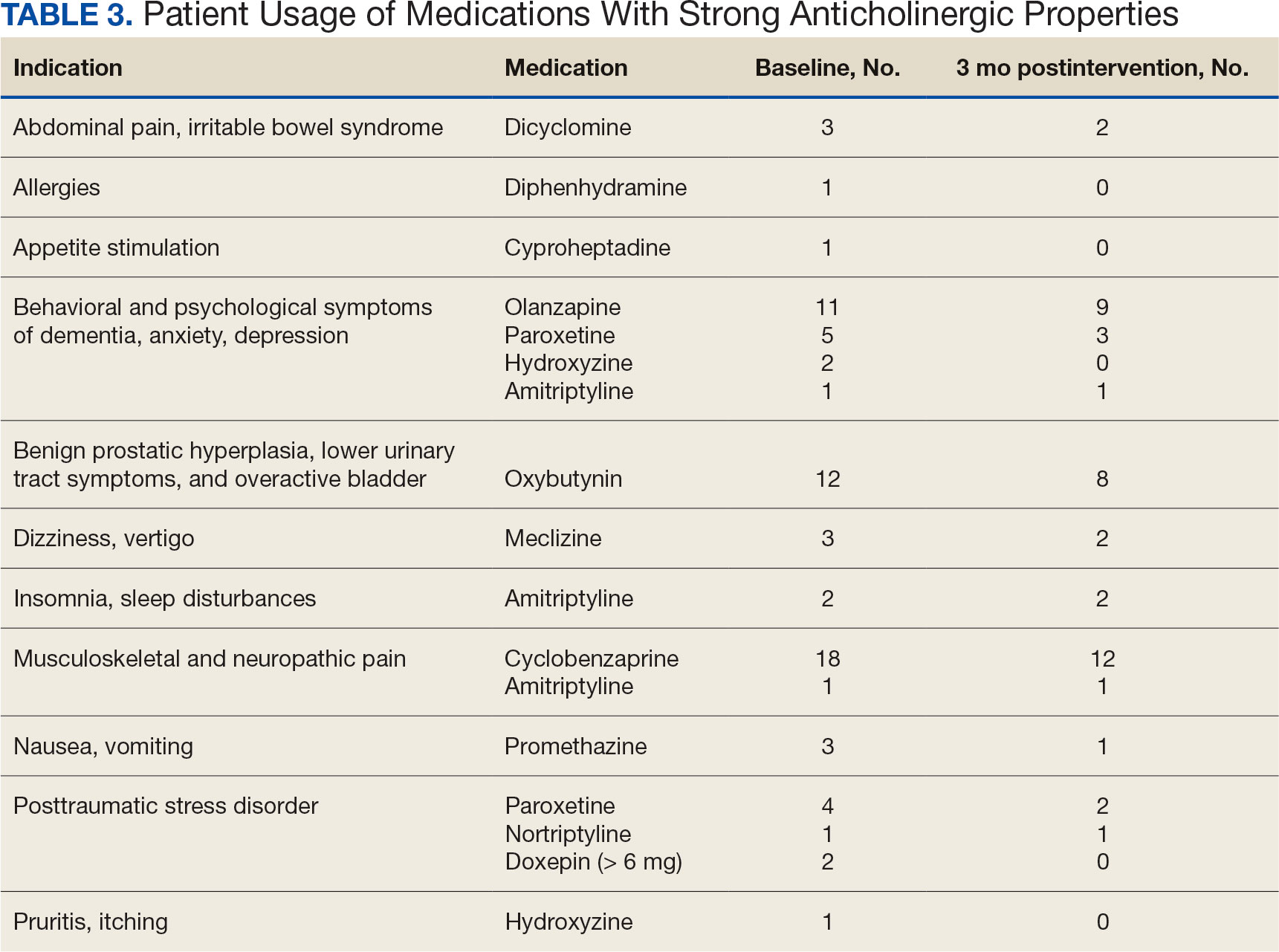
Discussion
This quality improvement project suggests that with the use of population health management tools such as the VIONE deprescribing dashboard, pharmacists can help identify and deprescribe strong anticholinergic medications in patients with cognitive impairment or dementia. Pharmacists can also aid in deprescribing through evidence-based recommendations to guide risk-benefit discussion and consider safer, nonanticholinergic alternatives. The authors were able to help reduce anticholinergic cognitive burden in 43% of patients in this sample. The mean 1.1 ACB score reduction was considered clinically significant based on prior studies that found that each 1-point increase in ACB score correlated with declined cognition and increased mortality.8,10 The VIONE deprescribing dashboard provided real-time patient data and helped target patients at the highest risk of anticholinergic AEs. The creation of the note templates based on the indication helped streamline recommendations. Typically, the prescriber addressed the recommendations at a routine follow-up appointment. The deprescribing method used in this project was time-efficient and could be easily replicated once the CPRS note templates were created. Future deprescribing projects could consider more direct pharmacist intervention and medication management.
Limitations
There was no direct assessment of clinical outcomes such as change in cognition using cognitive function tests. However, multiple studies have demonstrated AEs associated with strong anticholinergic medication use and additive anticholinergic burden in patients with dementia or cognitive impairment.1,5 Also, the 3-month follow-up period was relatively short. The pharmacist’s deprescribing recommendations may have been accepted after 3 months, or patients could have restarted their anticholinergic medications. Longer follow-up time could provide more robust results and conclusions. Thirdly, there was no formal definition of what constituted a risk-benefit assessment of anticholinergic medications. The risk-benefit assessment was determined at the discretion of the authors, which was subjective and allowed for bias. Finally, 6 patients died during the 3-month follow-up. The data for these patients were included in the baseline characteristics but not in the study outcomes. If these patients had been excluded from the results, a higher percentage of patients (47%) would have had ≥ 1 anticholinergic medication deprescribed.
Conclusions
In collaboration with the interdisciplinary team, pharmacist recommendations resulted in deprescribing of anticholinergic medications in veterans with dementia or cognitive impairment. The VIONE deprescribing dashboard, an easily accessible population health management tool, can identify patients prescribed potentially inappropriate medications and help target patients at the highest risk of anticholinergic AEs. To prevent worsening cognitive impairment, delirium, falls, and other AEs, this deprescribing initiative can be replicated at other VHA facilities. Future projects could have a longer follow-up period, incorporate more direct pharmacist intervention, and assess clinical outcomes of deprescribing.
- Gray SL, Hanlon JT. Anticholinergic medication use and dementia: latest evidence and clinical implications. Ther Adv Drug Saf. 2016;7(5):217-224. doi:10.1177/2042098616658399
- Kersten H, Wyller TB. Anticholinergic drug burden in older people’s brain - how well is it measured? Basic Clin Pharmacol Toxicol. 2014;114(2):151-159. doi:10.1111/bcpt.12140
- By the 2019 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2019 updated AGS beers criteria® for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2019;67(4):674-694. doi:10.1111/jgs.15767
- By the 2023 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2023 updated AGS Beers Criteria® for potentially inappropriate medication use in older adults J Am Geriatr Soc. 2023;71(7):2052-2081. doi:10.1111/jgs.18372
- Wang K, Alan J, Page AT, Dimopoulos E, Etherton-Beer C. Anticholinergics and clinical outcomes amongst people with pre-existing dementia: a systematic review. Maturitas. 2021;151:1-14. doi:10.1016/j.maturitas.2021.06.004
- Thorpe JM, Thorpe CT, Gellad WF, et al. Dual health care system use and high-risk prescribing in patients with dementia: a national cohort study. Ann Intern Med. 2017;166(3):157-163. doi:10.7326/M16-0551
- McCarren M, Burk M, Carico R, Glassman P, Good CB, Cunningham F. Design of a centrally aggregated medication use evaluation (CAMUE): anticholinergics in dementia. Presented at: 2019 HSR&D/QUERI National Conference; October 29-31, 2019; Washington, DC. https://www.hsrd.research.va.gov/meetings/2019/abstract-display.cfm?AbsNum=4027
- Boustani, M, Campbell, N, Munger S, et al. Impact of anticholinergics on the aging brain: a review and practical application. Aging Health. 2008;4(3):311-320. doi:10.2217/1745509.x
- Constantino-Corpuz JK, Alonso MTD. Assessment of a medication deprescribing tool on polypharmacy and cost avoidance. Fed Pract. 2021;38(7):332-336. doi:10.12788/fp.0146
- Fox C, Richardson K, Maidment ID, et al. Anticholinergic medication use and cognitive impairment in the older population: the medical research council cognitive function and ageing study. J Am Geriatr Soc. 2011;59(8):1477-1483. doi:10.1111/j.1532-5415.2011.03491.x
Anticholinergic medications block the activity of the neurotransmitter acetylcholine by binding to either muscarinic or nicotinic receptors in both the peripheral and central nervous system. Anticholinergic medications typically refer to antimuscarinic medications and have been prescribed to treat a variety of conditions common in older adults, including overactive bladder, allergies, muscle spasms, and sleep disorders.1,2 Since muscarinic receptors are present throughout the body, anticholinergic medications are associated with many adverse effects (AEs), including constipation, urinary retention, xerostomia, and delirium. Older adults are more sensitive to these AEs due to physiological changes associated with aging.1
The American Geriatric Society Beers Criteria for Potentially Inappropriate Medications Use in Older Adults identifies drugs with strong anticholinergic properties. The Beers Criteria strongly recommends avoiding these medications in patients with dementia or cognitive impairment due to the risk of central nervous system AEs. In the updated 2023 Beers Criteria, the rationale was expanded to recognize the risks of the cumulative anticholinergic burden associated with concurrent anticholinergic use.3,4
Given the prevalent use of anticholinergic medications in older adults, there has been significant research demonstrating their AEs, specifically delirium and cognitive impairment in geriatric patients. A systematic review of 14 articles conducted in 7 different countries of patients with median age of 76.4 to 86.1 years reviewed clinical outcomes of anticholinergic use in patients with dementia. Five studies found anticholinergics were associated with increased all-cause mortality in patients with dementia, and 3 studies found anticholinergics were associated with longer hospital stays. Other studies found that anticholinergics were associated with delirium and reduced health-related quality of life.5
About 35% of veterans with dementia have been prescribed a medication regimen with a high anticholinergic burden.6 In 2018, the US Department of Veterans Affairs (VA) Pharmacy Benfits Management Center for Medical Safety completed a centrally aggregated medication use evaluation (CAMUE) to assess the appropriateness of anticholinergic medication use in patients with dementia. The retrospective chart review included 1094 veterans from 19 sites. Overall, about 15% of the veterans experienced new falls, delirium, or worsening dementia within 30 days of starting an anticholinergic medication. Furthermore, < 40% had documentation of a nonanticholinergic alternative medication trial, and < 20% had documented nonpharmacologic therapy. The documentation of risk-benefit assessment acknowledging the risks of anticholinergic medication use in veterans with dementia occurred only about 13% of the time. The CAMUE concluded that the risks of initiating an anticholinergic medication in veterans with dementia are likely underdocumented and possibly under considered by prescribers.7
Developed within the Veterans Health Administration (VHA), VIONE (Vital, Important, Optional, Not Indicated, Every medication has an indication) is a medication management methodology that aims to reduce polypharmacy and improve patient safety consistent with high-reliability organizations. Since it launched in 2016, VIONE has gradually been implemented at many VHA facilities. The VIONE deprescribing dashboard had not been used at the VA Louisville Healthcare System prior to this quality improvement project.
This dashboard uses the Beers Criteria to identify potentially inappropriate anticholinergic medications. It uses the Anticholinergic Cognitive Burden (ACB) scale to calculate the cumulative anticholinergic risk for each patient. Medications with an ACB score of 2 or 3 have clinically relevant cognitive effects such as delirium and dementia (Table 1). For each point increase in total ACB score, a decline in mini-mental state examination score of 0.33 points over 2 years has been shown. Each point increase has also been correlated with a 26% increase in risk of death.8-10

Methods
The purpose of this quality improvement project was to determine the impact of pharmacist-driven deprescribing on the anticholinergic burden in veterans with dementia at VA Louisville Healthcare System. Data were obtained through the Computerized Patient Record System (CPRS) and VIONE deprescribing dashboard and entered in a secure Microsoft Excel spreadsheet. Pharmacist deprescribing steps were entered as CPRS progress notes. A deprescribing note template was created, and 11 templates with indication-specific recommendations were created for each anticholinergic indication identified (contact authors for deprescribing note template examples). Usage of anticholinergic medications was reexamined 3 months after the deprescribing note was entered.
Eligible patients identified in the VIONE deprescribing dashboard had an outpatient order for a medication with strong anticholinergic properties as identified using the Beers Criteria and were aged ≥ 65 years. Patients also had to be diagnosed with dementia or cognitive impairment. Patients were excluded if they were receiving hospice care or if the anticholinergic medication was from a non-VA prescriber or filled at a non-VA pharmacy. The VIONE deprescribing dashboard also excluded skeletal muscle relaxants if the patient had a spinal cord-related visit in the previous 2 years, first-generation antihistamines if the patient had a vertigo diagnosis, hydroxyzine if the indication was for anxiety, trospium if the indication was for overactive bladder, and antipsychotics if the patient had been diagnosed with schizophrenia or bipolar disorder. The following were included in the deprescribing recommendations if the dashboard identified the patient due to receiving a second strongly anticholinergic medication: first generation antihistamines if the patient was diagnosed with vertigo and hydroxyzine if the indication is for anxiety.
Each eligible patient received a focused medication review by a pharmacist via electronic chart review and a templated CPRS progress note with patient-specific recommendations. The prescriber and the patient’s primary care practitioner were recommended to perform a patient-specific risk-benefit assessment, deprescribe potentially inappropriate anticholinergic medications, and consider nonanticholinergic alternatives (both pharmacologic and nonpharmacologic). Data collected included baseline age, sex, prespecified comorbidities (type of dementia, cognitive impairment, delirium, benign prostatic hyperplasia/lower urinary tract symptoms), duration of prescribed anticholinergic medication, indication and deprescribing rate for each anticholinergic agent, and concurrent dementia medications (acetylcholinesterase inhibitors, memantine, or both).
The primary outcome was the number of patients that had = 1 medication with strong anticholinergic properties deprescribed. Deprescribing was defined as medication discontinuation or reduction of total daily dose. Secondary outcomes were the mean change in ACB scale, the number of patients with dose tapering, documented patient-specific risk-benefit assessment, and initiated nonanticholinergic alternative per pharmacist recommendation.
Results
The VIONE deprescribing dashboard identified 121 patients; 45 were excluded for non-VA prescriber or pharmacy, and 8 patients were excluded for other reasons. Sixty-eight patients were included in the deprescribing initiative. The mean age was 73.4 years (range, 67-93), 65 (96%) were male, and 34 (50%) had unspecified dementia (Table 2). Thirty-one patients (46%) had concurrent cholinesterase inhibitor prescriptions for dementia. The median duration of use of a strong anticholinergic medication was 11 months.

Twenty-nine patients (43%) had ≥ 1 medication with strong anticholinergic properties deprescribed. Anticholinergic medication was discontinued for 26 patients, and the dose was decreased for 3 patients. ACB score fell by a mean of 1.1 per patient. There was an increase in the documented risk-benefit assessment for anticholinergic medications from a baseline of 4 (6%) to 19 (28%) 3 months after the deprescribing note. Cyclobenzaprine, paroxetine, and oxybutynin were deprescribed the most, and amitriptyline had the lowest rate of deprescribing (Table 3). Thirty patients (44%) had a pharmacologic, nonanticholinergic alternative initiated per pharmacist recommendation, and 6 patients (9%) had a nonpharmacologic alternative initiated per pharmacist recommendation.

Discussion
This quality improvement project suggests that with the use of population health management tools such as the VIONE deprescribing dashboard, pharmacists can help identify and deprescribe strong anticholinergic medications in patients with cognitive impairment or dementia. Pharmacists can also aid in deprescribing through evidence-based recommendations to guide risk-benefit discussion and consider safer, nonanticholinergic alternatives. The authors were able to help reduce anticholinergic cognitive burden in 43% of patients in this sample. The mean 1.1 ACB score reduction was considered clinically significant based on prior studies that found that each 1-point increase in ACB score correlated with declined cognition and increased mortality.8,10 The VIONE deprescribing dashboard provided real-time patient data and helped target patients at the highest risk of anticholinergic AEs. The creation of the note templates based on the indication helped streamline recommendations. Typically, the prescriber addressed the recommendations at a routine follow-up appointment. The deprescribing method used in this project was time-efficient and could be easily replicated once the CPRS note templates were created. Future deprescribing projects could consider more direct pharmacist intervention and medication management.
Limitations
There was no direct assessment of clinical outcomes such as change in cognition using cognitive function tests. However, multiple studies have demonstrated AEs associated with strong anticholinergic medication use and additive anticholinergic burden in patients with dementia or cognitive impairment.1,5 Also, the 3-month follow-up period was relatively short. The pharmacist’s deprescribing recommendations may have been accepted after 3 months, or patients could have restarted their anticholinergic medications. Longer follow-up time could provide more robust results and conclusions. Thirdly, there was no formal definition of what constituted a risk-benefit assessment of anticholinergic medications. The risk-benefit assessment was determined at the discretion of the authors, which was subjective and allowed for bias. Finally, 6 patients died during the 3-month follow-up. The data for these patients were included in the baseline characteristics but not in the study outcomes. If these patients had been excluded from the results, a higher percentage of patients (47%) would have had ≥ 1 anticholinergic medication deprescribed.
Conclusions
In collaboration with the interdisciplinary team, pharmacist recommendations resulted in deprescribing of anticholinergic medications in veterans with dementia or cognitive impairment. The VIONE deprescribing dashboard, an easily accessible population health management tool, can identify patients prescribed potentially inappropriate medications and help target patients at the highest risk of anticholinergic AEs. To prevent worsening cognitive impairment, delirium, falls, and other AEs, this deprescribing initiative can be replicated at other VHA facilities. Future projects could have a longer follow-up period, incorporate more direct pharmacist intervention, and assess clinical outcomes of deprescribing.
Anticholinergic medications block the activity of the neurotransmitter acetylcholine by binding to either muscarinic or nicotinic receptors in both the peripheral and central nervous system. Anticholinergic medications typically refer to antimuscarinic medications and have been prescribed to treat a variety of conditions common in older adults, including overactive bladder, allergies, muscle spasms, and sleep disorders.1,2 Since muscarinic receptors are present throughout the body, anticholinergic medications are associated with many adverse effects (AEs), including constipation, urinary retention, xerostomia, and delirium. Older adults are more sensitive to these AEs due to physiological changes associated with aging.1
The American Geriatric Society Beers Criteria for Potentially Inappropriate Medications Use in Older Adults identifies drugs with strong anticholinergic properties. The Beers Criteria strongly recommends avoiding these medications in patients with dementia or cognitive impairment due to the risk of central nervous system AEs. In the updated 2023 Beers Criteria, the rationale was expanded to recognize the risks of the cumulative anticholinergic burden associated with concurrent anticholinergic use.3,4
Given the prevalent use of anticholinergic medications in older adults, there has been significant research demonstrating their AEs, specifically delirium and cognitive impairment in geriatric patients. A systematic review of 14 articles conducted in 7 different countries of patients with median age of 76.4 to 86.1 years reviewed clinical outcomes of anticholinergic use in patients with dementia. Five studies found anticholinergics were associated with increased all-cause mortality in patients with dementia, and 3 studies found anticholinergics were associated with longer hospital stays. Other studies found that anticholinergics were associated with delirium and reduced health-related quality of life.5
About 35% of veterans with dementia have been prescribed a medication regimen with a high anticholinergic burden.6 In 2018, the US Department of Veterans Affairs (VA) Pharmacy Benfits Management Center for Medical Safety completed a centrally aggregated medication use evaluation (CAMUE) to assess the appropriateness of anticholinergic medication use in patients with dementia. The retrospective chart review included 1094 veterans from 19 sites. Overall, about 15% of the veterans experienced new falls, delirium, or worsening dementia within 30 days of starting an anticholinergic medication. Furthermore, < 40% had documentation of a nonanticholinergic alternative medication trial, and < 20% had documented nonpharmacologic therapy. The documentation of risk-benefit assessment acknowledging the risks of anticholinergic medication use in veterans with dementia occurred only about 13% of the time. The CAMUE concluded that the risks of initiating an anticholinergic medication in veterans with dementia are likely underdocumented and possibly under considered by prescribers.7
Developed within the Veterans Health Administration (VHA), VIONE (Vital, Important, Optional, Not Indicated, Every medication has an indication) is a medication management methodology that aims to reduce polypharmacy and improve patient safety consistent with high-reliability organizations. Since it launched in 2016, VIONE has gradually been implemented at many VHA facilities. The VIONE deprescribing dashboard had not been used at the VA Louisville Healthcare System prior to this quality improvement project.
This dashboard uses the Beers Criteria to identify potentially inappropriate anticholinergic medications. It uses the Anticholinergic Cognitive Burden (ACB) scale to calculate the cumulative anticholinergic risk for each patient. Medications with an ACB score of 2 or 3 have clinically relevant cognitive effects such as delirium and dementia (Table 1). For each point increase in total ACB score, a decline in mini-mental state examination score of 0.33 points over 2 years has been shown. Each point increase has also been correlated with a 26% increase in risk of death.8-10

Methods
The purpose of this quality improvement project was to determine the impact of pharmacist-driven deprescribing on the anticholinergic burden in veterans with dementia at VA Louisville Healthcare System. Data were obtained through the Computerized Patient Record System (CPRS) and VIONE deprescribing dashboard and entered in a secure Microsoft Excel spreadsheet. Pharmacist deprescribing steps were entered as CPRS progress notes. A deprescribing note template was created, and 11 templates with indication-specific recommendations were created for each anticholinergic indication identified (contact authors for deprescribing note template examples). Usage of anticholinergic medications was reexamined 3 months after the deprescribing note was entered.
Eligible patients identified in the VIONE deprescribing dashboard had an outpatient order for a medication with strong anticholinergic properties as identified using the Beers Criteria and were aged ≥ 65 years. Patients also had to be diagnosed with dementia or cognitive impairment. Patients were excluded if they were receiving hospice care or if the anticholinergic medication was from a non-VA prescriber or filled at a non-VA pharmacy. The VIONE deprescribing dashboard also excluded skeletal muscle relaxants if the patient had a spinal cord-related visit in the previous 2 years, first-generation antihistamines if the patient had a vertigo diagnosis, hydroxyzine if the indication was for anxiety, trospium if the indication was for overactive bladder, and antipsychotics if the patient had been diagnosed with schizophrenia or bipolar disorder. The following were included in the deprescribing recommendations if the dashboard identified the patient due to receiving a second strongly anticholinergic medication: first generation antihistamines if the patient was diagnosed with vertigo and hydroxyzine if the indication is for anxiety.
Each eligible patient received a focused medication review by a pharmacist via electronic chart review and a templated CPRS progress note with patient-specific recommendations. The prescriber and the patient’s primary care practitioner were recommended to perform a patient-specific risk-benefit assessment, deprescribe potentially inappropriate anticholinergic medications, and consider nonanticholinergic alternatives (both pharmacologic and nonpharmacologic). Data collected included baseline age, sex, prespecified comorbidities (type of dementia, cognitive impairment, delirium, benign prostatic hyperplasia/lower urinary tract symptoms), duration of prescribed anticholinergic medication, indication and deprescribing rate for each anticholinergic agent, and concurrent dementia medications (acetylcholinesterase inhibitors, memantine, or both).
The primary outcome was the number of patients that had = 1 medication with strong anticholinergic properties deprescribed. Deprescribing was defined as medication discontinuation or reduction of total daily dose. Secondary outcomes were the mean change in ACB scale, the number of patients with dose tapering, documented patient-specific risk-benefit assessment, and initiated nonanticholinergic alternative per pharmacist recommendation.
Results
The VIONE deprescribing dashboard identified 121 patients; 45 were excluded for non-VA prescriber or pharmacy, and 8 patients were excluded for other reasons. Sixty-eight patients were included in the deprescribing initiative. The mean age was 73.4 years (range, 67-93), 65 (96%) were male, and 34 (50%) had unspecified dementia (Table 2). Thirty-one patients (46%) had concurrent cholinesterase inhibitor prescriptions for dementia. The median duration of use of a strong anticholinergic medication was 11 months.

Twenty-nine patients (43%) had ≥ 1 medication with strong anticholinergic properties deprescribed. Anticholinergic medication was discontinued for 26 patients, and the dose was decreased for 3 patients. ACB score fell by a mean of 1.1 per patient. There was an increase in the documented risk-benefit assessment for anticholinergic medications from a baseline of 4 (6%) to 19 (28%) 3 months after the deprescribing note. Cyclobenzaprine, paroxetine, and oxybutynin were deprescribed the most, and amitriptyline had the lowest rate of deprescribing (Table 3). Thirty patients (44%) had a pharmacologic, nonanticholinergic alternative initiated per pharmacist recommendation, and 6 patients (9%) had a nonpharmacologic alternative initiated per pharmacist recommendation.

Discussion
This quality improvement project suggests that with the use of population health management tools such as the VIONE deprescribing dashboard, pharmacists can help identify and deprescribe strong anticholinergic medications in patients with cognitive impairment or dementia. Pharmacists can also aid in deprescribing through evidence-based recommendations to guide risk-benefit discussion and consider safer, nonanticholinergic alternatives. The authors were able to help reduce anticholinergic cognitive burden in 43% of patients in this sample. The mean 1.1 ACB score reduction was considered clinically significant based on prior studies that found that each 1-point increase in ACB score correlated with declined cognition and increased mortality.8,10 The VIONE deprescribing dashboard provided real-time patient data and helped target patients at the highest risk of anticholinergic AEs. The creation of the note templates based on the indication helped streamline recommendations. Typically, the prescriber addressed the recommendations at a routine follow-up appointment. The deprescribing method used in this project was time-efficient and could be easily replicated once the CPRS note templates were created. Future deprescribing projects could consider more direct pharmacist intervention and medication management.
Limitations
There was no direct assessment of clinical outcomes such as change in cognition using cognitive function tests. However, multiple studies have demonstrated AEs associated with strong anticholinergic medication use and additive anticholinergic burden in patients with dementia or cognitive impairment.1,5 Also, the 3-month follow-up period was relatively short. The pharmacist’s deprescribing recommendations may have been accepted after 3 months, or patients could have restarted their anticholinergic medications. Longer follow-up time could provide more robust results and conclusions. Thirdly, there was no formal definition of what constituted a risk-benefit assessment of anticholinergic medications. The risk-benefit assessment was determined at the discretion of the authors, which was subjective and allowed for bias. Finally, 6 patients died during the 3-month follow-up. The data for these patients were included in the baseline characteristics but not in the study outcomes. If these patients had been excluded from the results, a higher percentage of patients (47%) would have had ≥ 1 anticholinergic medication deprescribed.
Conclusions
In collaboration with the interdisciplinary team, pharmacist recommendations resulted in deprescribing of anticholinergic medications in veterans with dementia or cognitive impairment. The VIONE deprescribing dashboard, an easily accessible population health management tool, can identify patients prescribed potentially inappropriate medications and help target patients at the highest risk of anticholinergic AEs. To prevent worsening cognitive impairment, delirium, falls, and other AEs, this deprescribing initiative can be replicated at other VHA facilities. Future projects could have a longer follow-up period, incorporate more direct pharmacist intervention, and assess clinical outcomes of deprescribing.
- Gray SL, Hanlon JT. Anticholinergic medication use and dementia: latest evidence and clinical implications. Ther Adv Drug Saf. 2016;7(5):217-224. doi:10.1177/2042098616658399
- Kersten H, Wyller TB. Anticholinergic drug burden in older people’s brain - how well is it measured? Basic Clin Pharmacol Toxicol. 2014;114(2):151-159. doi:10.1111/bcpt.12140
- By the 2019 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2019 updated AGS beers criteria® for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2019;67(4):674-694. doi:10.1111/jgs.15767
- By the 2023 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2023 updated AGS Beers Criteria® for potentially inappropriate medication use in older adults J Am Geriatr Soc. 2023;71(7):2052-2081. doi:10.1111/jgs.18372
- Wang K, Alan J, Page AT, Dimopoulos E, Etherton-Beer C. Anticholinergics and clinical outcomes amongst people with pre-existing dementia: a systematic review. Maturitas. 2021;151:1-14. doi:10.1016/j.maturitas.2021.06.004
- Thorpe JM, Thorpe CT, Gellad WF, et al. Dual health care system use and high-risk prescribing in patients with dementia: a national cohort study. Ann Intern Med. 2017;166(3):157-163. doi:10.7326/M16-0551
- McCarren M, Burk M, Carico R, Glassman P, Good CB, Cunningham F. Design of a centrally aggregated medication use evaluation (CAMUE): anticholinergics in dementia. Presented at: 2019 HSR&D/QUERI National Conference; October 29-31, 2019; Washington, DC. https://www.hsrd.research.va.gov/meetings/2019/abstract-display.cfm?AbsNum=4027
- Boustani, M, Campbell, N, Munger S, et al. Impact of anticholinergics on the aging brain: a review and practical application. Aging Health. 2008;4(3):311-320. doi:10.2217/1745509.x
- Constantino-Corpuz JK, Alonso MTD. Assessment of a medication deprescribing tool on polypharmacy and cost avoidance. Fed Pract. 2021;38(7):332-336. doi:10.12788/fp.0146
- Fox C, Richardson K, Maidment ID, et al. Anticholinergic medication use and cognitive impairment in the older population: the medical research council cognitive function and ageing study. J Am Geriatr Soc. 2011;59(8):1477-1483. doi:10.1111/j.1532-5415.2011.03491.x
- Gray SL, Hanlon JT. Anticholinergic medication use and dementia: latest evidence and clinical implications. Ther Adv Drug Saf. 2016;7(5):217-224. doi:10.1177/2042098616658399
- Kersten H, Wyller TB. Anticholinergic drug burden in older people’s brain - how well is it measured? Basic Clin Pharmacol Toxicol. 2014;114(2):151-159. doi:10.1111/bcpt.12140
- By the 2019 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2019 updated AGS beers criteria® for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2019;67(4):674-694. doi:10.1111/jgs.15767
- By the 2023 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2023 updated AGS Beers Criteria® for potentially inappropriate medication use in older adults J Am Geriatr Soc. 2023;71(7):2052-2081. doi:10.1111/jgs.18372
- Wang K, Alan J, Page AT, Dimopoulos E, Etherton-Beer C. Anticholinergics and clinical outcomes amongst people with pre-existing dementia: a systematic review. Maturitas. 2021;151:1-14. doi:10.1016/j.maturitas.2021.06.004
- Thorpe JM, Thorpe CT, Gellad WF, et al. Dual health care system use and high-risk prescribing in patients with dementia: a national cohort study. Ann Intern Med. 2017;166(3):157-163. doi:10.7326/M16-0551
- McCarren M, Burk M, Carico R, Glassman P, Good CB, Cunningham F. Design of a centrally aggregated medication use evaluation (CAMUE): anticholinergics in dementia. Presented at: 2019 HSR&D/QUERI National Conference; October 29-31, 2019; Washington, DC. https://www.hsrd.research.va.gov/meetings/2019/abstract-display.cfm?AbsNum=4027
- Boustani, M, Campbell, N, Munger S, et al. Impact of anticholinergics on the aging brain: a review and practical application. Aging Health. 2008;4(3):311-320. doi:10.2217/1745509.x
- Constantino-Corpuz JK, Alonso MTD. Assessment of a medication deprescribing tool on polypharmacy and cost avoidance. Fed Pract. 2021;38(7):332-336. doi:10.12788/fp.0146
- Fox C, Richardson K, Maidment ID, et al. Anticholinergic medication use and cognitive impairment in the older population: the medical research council cognitive function and ageing study. J Am Geriatr Soc. 2011;59(8):1477-1483. doi:10.1111/j.1532-5415.2011.03491.x
Pharmacist-Driven Deprescribing to Reduce Anticholinergic Burden in Veterans With Dementia
Pharmacist-Driven Deprescribing to Reduce Anticholinergic Burden in Veterans With Dementia
New Cancer Vaccines on the Horizon: Renewed Hope or Hype?
Vaccines for treating and preventing cancer have long been considered a holy grail in oncology.
But aside from a few notable exceptions — including the human papillomavirus (HPV) vaccine, which has dramatically reduced the incidence of HPV-related cancers, and a Bacillus Calmette-Guerin vaccine, which helps prevent early-stage bladder cancer recurrence — most have failed to deliver.
Following a string of disappointments over the past decade, recent advances in the immunotherapy space are bringing renewed hope for progress.
In an American Association for Cancer Research (AACR) series earlier in 2024, Catherine J. Wu, MD, predicted big strides for cancer vaccines, especially for personalized vaccines that target patient-specific neoantigens — the proteins that form on cancer cells — as well as vaccines that can treat diverse tumor types.
said Wu, the Lavine Family Chair of Preventative Cancer Therapies at Dana-Farber Cancer Institute and a professor of medicine at Harvard Medical School, both in Boston, Massachusetts.
A prime example is a personalized, messenger RNA (mRNA)–based vaccine designed to prevent melanoma recurrence. The mRNA-4157 vaccine encodes up to 34 different patient-specific neoantigens.
“This is one of the most exciting developments in modern cancer therapy,” said Lawrence Young, a virologist and professor of molecular oncology at the University of Warwick, Coventry, England, who commented on the investigational vaccine via the UK-based Science Media Centre.
Other promising options are on the horizon as well. In August, BioNTech announced a phase 1 global trial to study BNT116 — a vaccine to treat non–small cell lung cancer (NSCLC). BNT116, like mRNA-4157, targets specific antigens in the lung cancer cells.
“This technology is the next big phase of cancer treatment,” Siow Ming Lee, MD, a consultant medical oncologist at University College London Hospitals in England, which is leading the UK trial for the lung cancer and melanoma vaccines, told The Guardian. “We are now entering this very exciting new era of mRNA-based immunotherapy clinical trials to investigate the treatment of lung cancer.”
Still, these predictions have a familiar ring. While the prospects are exciting, delivering on them is another story. There are simply no guarantees these strategies will work as hoped.
Then: Where We Were
Cancer vaccine research began to ramp up in the 2000s, and in 2006, the first-generation HPV vaccine, Gardasil, was approved. Gardasil prevents infection from four strains of HPV that cause about 80% of cervical cancer cases.
In 2010, the Food and Drug Administration approved sipuleucel-T, the first therapeutic cancer vaccine, which improved overall survival in patients with hormone-refractory prostate cancer.
Researchers predicted this approval would “pave the way for developing innovative, next generation of vaccines with enhanced antitumor potency.”
In a 2015 AACR research forecast report, Drew Pardoll, MD, PhD, co-director of the Cancer Immunology and Hematopoiesis Program at Johns Hopkins University, Baltimore, Maryland, said that “we can expect to see encouraging results from studies using cancer vaccines.”
Despite the excitement surrounding cancer vaccines alongside a few successes, the next decade brought a longer string of late-phase disappointments.
In 2016, the phase 3 ACT IV trial of a therapeutic vaccine to treat glioblastoma multiforme (CDX-110) was terminated after it failed to demonstrate improved survival.
In 2017, a phase 3 trial of the therapeutic pancreatic cancer vaccine, GVAX, was stopped early for lack of efficacy.
That year, an attenuated Listeria monocytogenes vaccine to treat pancreatic cancer and mesothelioma also failed to come to fruition. In late 2017, concerns over listeria infections prompted Aduro Biotech to cancel its listeria-based cancer treatment program.
In 2018, a phase 3 trial of belagenpumatucel-L, a therapeutic NSCLC vaccine, failed to demonstrate a significant improvement in survival and further study was discontinued.
And in 2019, a vaccine targeting MAGE-A3, a cancer-testis antigen present in multiple tumor types, failed to meet endpoints for improved survival in a phase 3 trial, leading to discontinuation of the vaccine program.
But these disappointments and failures are normal parts of medical research and drug development and have allowed for incremental advances that helped fuel renewed interest and hope for cancer vaccines, when the timing was right, explained vaccine pioneer Larry W. Kwak, MD, PhD, deputy director of the Comprehensive Cancer Center at City of Hope, Duarte, California.
When it comes to vaccine progress, timing makes a difference. In 2011, Kwak and colleagues published promising phase 3 trial results on a personalized vaccine. The vaccine was a patient-specific tumor-derived antigen for patients with follicular lymphoma in their first remission following chemotherapy. Patients who received the vaccine demonstrated significantly longer disease-free survival.
But, at the time, personalized vaccines faced strong headwinds due, largely, to high costs, and commercial interest failed to materialize. “That’s been the major hurdle for a long time,” said Kwak.
Now, however, interest has returned alongside advances in technology and research. The big shift has been the emergence of lower-cost rapid-production mRNA and DNA platforms and a better understanding of how vaccines and potent immune stimulants, like checkpoint inhibitors, can work together to improve outcomes, he explained.
“The timing wasn’t right” back then, Kwak noted. “Now, it’s a different environment and a different time.”
A Turning Point?
Indeed, a decade later, cancer vaccine development appears to be headed in a more promising direction.
Among key cancer vaccines to watch is the mRNA-4157 vaccine, developed by Merck and Moderna, designed to prevent melanoma recurrence. In a recent phase 2 study, patients receiving the mRNA-4157 vaccine alongside pembrolizumab had nearly half the risk for melanoma recurrence or death at 3 years compared with those receiving pembrolizumab alone. Investigators are now evaluating the vaccine in a global phase 3 study in patients with high-risk, stage IIB to IV melanoma following surgery.
Another one to watch is the BNT116 NSCLC vaccine from BioNTech. This vaccine presents the immune system with NSCLC tumor markers to encourage the body to fight cancer cells expressing those markers while ignoring healthy cells. BioNTech also launched a global clinical trial for its vaccine this year.
Other notables include a pancreatic cancer mRNA vaccine, which has shown promising early results in a small trial of 16 patients. Of 16 patients who received the vaccine alongside chemotherapy and after surgery and immunotherapy, 8 responded. Of these eight, six remained recurrence free at 3 years. Investigators noted that the vaccine appeared to stimulate a durable T-cell response in patients who responded.
Kwak has also continued his work on lymphoma vaccines. In August, his team published promising first-in-human data on the use of personalized neoantigen vaccines as an early intervention in untreated patients with lymphoplasmacytic lymphoma. Among nine asymptomatic patients who received the vaccine, all achieved stable disease or better, with no dose-limiting toxicities. One patient had a minor response, and the median time to progression was greater than 72 months.
“The current setting is more for advanced disease,” Kwak explained. “It’s a tougher task, but combined with checkpoint blockade, it may be potent enough to work.”
Still, caution is important. Despite early promise, it’s too soon to tell which, if any, of these investigational vaccines will pan out in the long run. Like investigational drugs, cancer vaccines may show big promising initially but then fail in larger trials.
One key to success, according to Kwak, is to design trials so that even negative results will inform next steps.
But, he noted, failures in large clinical trials will “put a chilling effect on cancer vaccine research again.”
“That’s what keeps me up at night,” he said. “We know the science is fundamentally sound and we have seen glimpses over decades of research that cancer vaccines can work, so it’s really just a matter of tweaking things to optimize trial design.”
Companies tend to design trials to test if a vaccine works or not, without trying to understand why, he said.
“What we need to do is design those so that we can learn from negative results,” he said. That’s what he and his colleagues attempted to do in their recent trial. “We didn’t just look at clinical results; we’re interrogating the actual tumor environment to understand what worked and didn’t and how to tweak that for the next trial.”
Kwak and his colleagues found, for instance, that the vaccine had a greater effect on B cell–derived tumor cells than on cells of plasma origin, so “the most rational design for the next iteration is to combine the vaccine with agents that work directly against plasma cells,” he explained.
As for what’s next, Kwak said: “We’re just focused on trying to do good science and understand. We’ve seen glimpses of success. That’s where we are.”
A version of this article first appeared on Medscape.com.
Vaccines for treating and preventing cancer have long been considered a holy grail in oncology.
But aside from a few notable exceptions — including the human papillomavirus (HPV) vaccine, which has dramatically reduced the incidence of HPV-related cancers, and a Bacillus Calmette-Guerin vaccine, which helps prevent early-stage bladder cancer recurrence — most have failed to deliver.
Following a string of disappointments over the past decade, recent advances in the immunotherapy space are bringing renewed hope for progress.
In an American Association for Cancer Research (AACR) series earlier in 2024, Catherine J. Wu, MD, predicted big strides for cancer vaccines, especially for personalized vaccines that target patient-specific neoantigens — the proteins that form on cancer cells — as well as vaccines that can treat diverse tumor types.
said Wu, the Lavine Family Chair of Preventative Cancer Therapies at Dana-Farber Cancer Institute and a professor of medicine at Harvard Medical School, both in Boston, Massachusetts.
A prime example is a personalized, messenger RNA (mRNA)–based vaccine designed to prevent melanoma recurrence. The mRNA-4157 vaccine encodes up to 34 different patient-specific neoantigens.
“This is one of the most exciting developments in modern cancer therapy,” said Lawrence Young, a virologist and professor of molecular oncology at the University of Warwick, Coventry, England, who commented on the investigational vaccine via the UK-based Science Media Centre.
Other promising options are on the horizon as well. In August, BioNTech announced a phase 1 global trial to study BNT116 — a vaccine to treat non–small cell lung cancer (NSCLC). BNT116, like mRNA-4157, targets specific antigens in the lung cancer cells.
“This technology is the next big phase of cancer treatment,” Siow Ming Lee, MD, a consultant medical oncologist at University College London Hospitals in England, which is leading the UK trial for the lung cancer and melanoma vaccines, told The Guardian. “We are now entering this very exciting new era of mRNA-based immunotherapy clinical trials to investigate the treatment of lung cancer.”
Still, these predictions have a familiar ring. While the prospects are exciting, delivering on them is another story. There are simply no guarantees these strategies will work as hoped.
Then: Where We Were
Cancer vaccine research began to ramp up in the 2000s, and in 2006, the first-generation HPV vaccine, Gardasil, was approved. Gardasil prevents infection from four strains of HPV that cause about 80% of cervical cancer cases.
In 2010, the Food and Drug Administration approved sipuleucel-T, the first therapeutic cancer vaccine, which improved overall survival in patients with hormone-refractory prostate cancer.
Researchers predicted this approval would “pave the way for developing innovative, next generation of vaccines with enhanced antitumor potency.”
In a 2015 AACR research forecast report, Drew Pardoll, MD, PhD, co-director of the Cancer Immunology and Hematopoiesis Program at Johns Hopkins University, Baltimore, Maryland, said that “we can expect to see encouraging results from studies using cancer vaccines.”
Despite the excitement surrounding cancer vaccines alongside a few successes, the next decade brought a longer string of late-phase disappointments.
In 2016, the phase 3 ACT IV trial of a therapeutic vaccine to treat glioblastoma multiforme (CDX-110) was terminated after it failed to demonstrate improved survival.
In 2017, a phase 3 trial of the therapeutic pancreatic cancer vaccine, GVAX, was stopped early for lack of efficacy.
That year, an attenuated Listeria monocytogenes vaccine to treat pancreatic cancer and mesothelioma also failed to come to fruition. In late 2017, concerns over listeria infections prompted Aduro Biotech to cancel its listeria-based cancer treatment program.
In 2018, a phase 3 trial of belagenpumatucel-L, a therapeutic NSCLC vaccine, failed to demonstrate a significant improvement in survival and further study was discontinued.
And in 2019, a vaccine targeting MAGE-A3, a cancer-testis antigen present in multiple tumor types, failed to meet endpoints for improved survival in a phase 3 trial, leading to discontinuation of the vaccine program.
But these disappointments and failures are normal parts of medical research and drug development and have allowed for incremental advances that helped fuel renewed interest and hope for cancer vaccines, when the timing was right, explained vaccine pioneer Larry W. Kwak, MD, PhD, deputy director of the Comprehensive Cancer Center at City of Hope, Duarte, California.
When it comes to vaccine progress, timing makes a difference. In 2011, Kwak and colleagues published promising phase 3 trial results on a personalized vaccine. The vaccine was a patient-specific tumor-derived antigen for patients with follicular lymphoma in their first remission following chemotherapy. Patients who received the vaccine demonstrated significantly longer disease-free survival.
But, at the time, personalized vaccines faced strong headwinds due, largely, to high costs, and commercial interest failed to materialize. “That’s been the major hurdle for a long time,” said Kwak.
Now, however, interest has returned alongside advances in technology and research. The big shift has been the emergence of lower-cost rapid-production mRNA and DNA platforms and a better understanding of how vaccines and potent immune stimulants, like checkpoint inhibitors, can work together to improve outcomes, he explained.
“The timing wasn’t right” back then, Kwak noted. “Now, it’s a different environment and a different time.”
A Turning Point?
Indeed, a decade later, cancer vaccine development appears to be headed in a more promising direction.
Among key cancer vaccines to watch is the mRNA-4157 vaccine, developed by Merck and Moderna, designed to prevent melanoma recurrence. In a recent phase 2 study, patients receiving the mRNA-4157 vaccine alongside pembrolizumab had nearly half the risk for melanoma recurrence or death at 3 years compared with those receiving pembrolizumab alone. Investigators are now evaluating the vaccine in a global phase 3 study in patients with high-risk, stage IIB to IV melanoma following surgery.
Another one to watch is the BNT116 NSCLC vaccine from BioNTech. This vaccine presents the immune system with NSCLC tumor markers to encourage the body to fight cancer cells expressing those markers while ignoring healthy cells. BioNTech also launched a global clinical trial for its vaccine this year.
Other notables include a pancreatic cancer mRNA vaccine, which has shown promising early results in a small trial of 16 patients. Of 16 patients who received the vaccine alongside chemotherapy and after surgery and immunotherapy, 8 responded. Of these eight, six remained recurrence free at 3 years. Investigators noted that the vaccine appeared to stimulate a durable T-cell response in patients who responded.
Kwak has also continued his work on lymphoma vaccines. In August, his team published promising first-in-human data on the use of personalized neoantigen vaccines as an early intervention in untreated patients with lymphoplasmacytic lymphoma. Among nine asymptomatic patients who received the vaccine, all achieved stable disease or better, with no dose-limiting toxicities. One patient had a minor response, and the median time to progression was greater than 72 months.
“The current setting is more for advanced disease,” Kwak explained. “It’s a tougher task, but combined with checkpoint blockade, it may be potent enough to work.”
Still, caution is important. Despite early promise, it’s too soon to tell which, if any, of these investigational vaccines will pan out in the long run. Like investigational drugs, cancer vaccines may show big promising initially but then fail in larger trials.
One key to success, according to Kwak, is to design trials so that even negative results will inform next steps.
But, he noted, failures in large clinical trials will “put a chilling effect on cancer vaccine research again.”
“That’s what keeps me up at night,” he said. “We know the science is fundamentally sound and we have seen glimpses over decades of research that cancer vaccines can work, so it’s really just a matter of tweaking things to optimize trial design.”
Companies tend to design trials to test if a vaccine works or not, without trying to understand why, he said.
“What we need to do is design those so that we can learn from negative results,” he said. That’s what he and his colleagues attempted to do in their recent trial. “We didn’t just look at clinical results; we’re interrogating the actual tumor environment to understand what worked and didn’t and how to tweak that for the next trial.”
Kwak and his colleagues found, for instance, that the vaccine had a greater effect on B cell–derived tumor cells than on cells of plasma origin, so “the most rational design for the next iteration is to combine the vaccine with agents that work directly against plasma cells,” he explained.
As for what’s next, Kwak said: “We’re just focused on trying to do good science and understand. We’ve seen glimpses of success. That’s where we are.”
A version of this article first appeared on Medscape.com.
Vaccines for treating and preventing cancer have long been considered a holy grail in oncology.
But aside from a few notable exceptions — including the human papillomavirus (HPV) vaccine, which has dramatically reduced the incidence of HPV-related cancers, and a Bacillus Calmette-Guerin vaccine, which helps prevent early-stage bladder cancer recurrence — most have failed to deliver.
Following a string of disappointments over the past decade, recent advances in the immunotherapy space are bringing renewed hope for progress.
In an American Association for Cancer Research (AACR) series earlier in 2024, Catherine J. Wu, MD, predicted big strides for cancer vaccines, especially for personalized vaccines that target patient-specific neoantigens — the proteins that form on cancer cells — as well as vaccines that can treat diverse tumor types.
said Wu, the Lavine Family Chair of Preventative Cancer Therapies at Dana-Farber Cancer Institute and a professor of medicine at Harvard Medical School, both in Boston, Massachusetts.
A prime example is a personalized, messenger RNA (mRNA)–based vaccine designed to prevent melanoma recurrence. The mRNA-4157 vaccine encodes up to 34 different patient-specific neoantigens.
“This is one of the most exciting developments in modern cancer therapy,” said Lawrence Young, a virologist and professor of molecular oncology at the University of Warwick, Coventry, England, who commented on the investigational vaccine via the UK-based Science Media Centre.
Other promising options are on the horizon as well. In August, BioNTech announced a phase 1 global trial to study BNT116 — a vaccine to treat non–small cell lung cancer (NSCLC). BNT116, like mRNA-4157, targets specific antigens in the lung cancer cells.
“This technology is the next big phase of cancer treatment,” Siow Ming Lee, MD, a consultant medical oncologist at University College London Hospitals in England, which is leading the UK trial for the lung cancer and melanoma vaccines, told The Guardian. “We are now entering this very exciting new era of mRNA-based immunotherapy clinical trials to investigate the treatment of lung cancer.”
Still, these predictions have a familiar ring. While the prospects are exciting, delivering on them is another story. There are simply no guarantees these strategies will work as hoped.
Then: Where We Were
Cancer vaccine research began to ramp up in the 2000s, and in 2006, the first-generation HPV vaccine, Gardasil, was approved. Gardasil prevents infection from four strains of HPV that cause about 80% of cervical cancer cases.
In 2010, the Food and Drug Administration approved sipuleucel-T, the first therapeutic cancer vaccine, which improved overall survival in patients with hormone-refractory prostate cancer.
Researchers predicted this approval would “pave the way for developing innovative, next generation of vaccines with enhanced antitumor potency.”
In a 2015 AACR research forecast report, Drew Pardoll, MD, PhD, co-director of the Cancer Immunology and Hematopoiesis Program at Johns Hopkins University, Baltimore, Maryland, said that “we can expect to see encouraging results from studies using cancer vaccines.”
Despite the excitement surrounding cancer vaccines alongside a few successes, the next decade brought a longer string of late-phase disappointments.
In 2016, the phase 3 ACT IV trial of a therapeutic vaccine to treat glioblastoma multiforme (CDX-110) was terminated after it failed to demonstrate improved survival.
In 2017, a phase 3 trial of the therapeutic pancreatic cancer vaccine, GVAX, was stopped early for lack of efficacy.
That year, an attenuated Listeria monocytogenes vaccine to treat pancreatic cancer and mesothelioma also failed to come to fruition. In late 2017, concerns over listeria infections prompted Aduro Biotech to cancel its listeria-based cancer treatment program.
In 2018, a phase 3 trial of belagenpumatucel-L, a therapeutic NSCLC vaccine, failed to demonstrate a significant improvement in survival and further study was discontinued.
And in 2019, a vaccine targeting MAGE-A3, a cancer-testis antigen present in multiple tumor types, failed to meet endpoints for improved survival in a phase 3 trial, leading to discontinuation of the vaccine program.
But these disappointments and failures are normal parts of medical research and drug development and have allowed for incremental advances that helped fuel renewed interest and hope for cancer vaccines, when the timing was right, explained vaccine pioneer Larry W. Kwak, MD, PhD, deputy director of the Comprehensive Cancer Center at City of Hope, Duarte, California.
When it comes to vaccine progress, timing makes a difference. In 2011, Kwak and colleagues published promising phase 3 trial results on a personalized vaccine. The vaccine was a patient-specific tumor-derived antigen for patients with follicular lymphoma in their first remission following chemotherapy. Patients who received the vaccine demonstrated significantly longer disease-free survival.
But, at the time, personalized vaccines faced strong headwinds due, largely, to high costs, and commercial interest failed to materialize. “That’s been the major hurdle for a long time,” said Kwak.
Now, however, interest has returned alongside advances in technology and research. The big shift has been the emergence of lower-cost rapid-production mRNA and DNA platforms and a better understanding of how vaccines and potent immune stimulants, like checkpoint inhibitors, can work together to improve outcomes, he explained.
“The timing wasn’t right” back then, Kwak noted. “Now, it’s a different environment and a different time.”
A Turning Point?
Indeed, a decade later, cancer vaccine development appears to be headed in a more promising direction.
Among key cancer vaccines to watch is the mRNA-4157 vaccine, developed by Merck and Moderna, designed to prevent melanoma recurrence. In a recent phase 2 study, patients receiving the mRNA-4157 vaccine alongside pembrolizumab had nearly half the risk for melanoma recurrence or death at 3 years compared with those receiving pembrolizumab alone. Investigators are now evaluating the vaccine in a global phase 3 study in patients with high-risk, stage IIB to IV melanoma following surgery.
Another one to watch is the BNT116 NSCLC vaccine from BioNTech. This vaccine presents the immune system with NSCLC tumor markers to encourage the body to fight cancer cells expressing those markers while ignoring healthy cells. BioNTech also launched a global clinical trial for its vaccine this year.
Other notables include a pancreatic cancer mRNA vaccine, which has shown promising early results in a small trial of 16 patients. Of 16 patients who received the vaccine alongside chemotherapy and after surgery and immunotherapy, 8 responded. Of these eight, six remained recurrence free at 3 years. Investigators noted that the vaccine appeared to stimulate a durable T-cell response in patients who responded.
Kwak has also continued his work on lymphoma vaccines. In August, his team published promising first-in-human data on the use of personalized neoantigen vaccines as an early intervention in untreated patients with lymphoplasmacytic lymphoma. Among nine asymptomatic patients who received the vaccine, all achieved stable disease or better, with no dose-limiting toxicities. One patient had a minor response, and the median time to progression was greater than 72 months.
“The current setting is more for advanced disease,” Kwak explained. “It’s a tougher task, but combined with checkpoint blockade, it may be potent enough to work.”
Still, caution is important. Despite early promise, it’s too soon to tell which, if any, of these investigational vaccines will pan out in the long run. Like investigational drugs, cancer vaccines may show big promising initially but then fail in larger trials.
One key to success, according to Kwak, is to design trials so that even negative results will inform next steps.
But, he noted, failures in large clinical trials will “put a chilling effect on cancer vaccine research again.”
“That’s what keeps me up at night,” he said. “We know the science is fundamentally sound and we have seen glimpses over decades of research that cancer vaccines can work, so it’s really just a matter of tweaking things to optimize trial design.”
Companies tend to design trials to test if a vaccine works or not, without trying to understand why, he said.
“What we need to do is design those so that we can learn from negative results,” he said. That’s what he and his colleagues attempted to do in their recent trial. “We didn’t just look at clinical results; we’re interrogating the actual tumor environment to understand what worked and didn’t and how to tweak that for the next trial.”
Kwak and his colleagues found, for instance, that the vaccine had a greater effect on B cell–derived tumor cells than on cells of plasma origin, so “the most rational design for the next iteration is to combine the vaccine with agents that work directly against plasma cells,” he explained.
As for what’s next, Kwak said: “We’re just focused on trying to do good science and understand. We’ve seen glimpses of success. That’s where we are.”
A version of this article first appeared on Medscape.com.
Identifying the Best Upfront Regimen for Unresectable CRC Liver Metastasis
A new report demonstrated why patients benefit most from starting on a two-drug chemotherapy regimen — FOLFOX or FOLFIRI — instead of a three-drug regimen — FOLFOXIRI.
The CAIRO5 trial compared overall survival among 294 patients with right sided tumors and/or RAS/BRAF mutations who received FOLFOXIRI (5-fluorouracil [FU], oxaliplatin, irinotecan, plus folinic acid as an enhancer) or investigators’ choice of FOLFOX (5-FU, oxaliplatin, and folinic acid) or FOLFIRI (5-FU, irinotecan, and folinic acid). All patients also received bevacizumab.
In a post hoc analysis, researchers found no overall survival benefit among patients receiving the three-drug regimen. At a median follow-up of just over 5 years, the median overall survival was 23.6 months with FOLFOX or FOLFIRI vs 24.1 months with FOLFOXIRI (P = .44).
The finding means that patients can avoid the extra toxicity associated with combining oxaliplatin and irinotecan without compromising overall survival, Alan P. Venook, MD, a gastrointestinal medical oncologist at the University of California San Francisco, told Medscape Medical News.
The analysis did not stop there in defining the optimal upfront therapy for this patient population.
In a second arm of the analysis, researchers looked at whether swapping in panitumumab for bevacizumab offered any benefit in 236 patients with left-sided tumors and wild-type RAS/BRAF who received either of the two-drug regimens.
Here, the authors also found no benefit of using panitumumab with FOLFOX or FOLFIRI instead of bevacizumab, reporting a median overall survival of 38.3 months with panitumumab vs 39.9 months with bevacizumab.
In addition to avoiding upfront FOLFOXIRI, patients can also avoid the skin reactions and other toxicities associated with panitumumab, including “horrible acne,” Venook said.
Overall, the results support the use of FOLFOX or FOLFIRI with bevacizumab “irrespective of RAS/BRAFV600E status and tumor sidedness” as the initial treatment for CRC with liver-only metastases, concluded the study investigators, from the University Medical Center Utrecht in the Netherlands.
Why Does This Clarity Matter?
The study confirms the standard practice in the United States of starting patients on a two-drug chemotherapy with bevacizumab for the indication and highlights “why we don’t go all in right at the beginning” with a three-drug regimen, Venook said.
In short, more drugs upfront isn’t going to change patients’ long-term survival outcome. Plus, using FOLFOXIRI upfront means “you’ve really pretty much used up all your guns for early treatment,” Venook said.
As for bevacizumab vs panitumumab, most practitioners in the United States favor bevacizumab because of the rash many patients on epidermal growth factor receptor blockers like panitumumab and cetuximab get, Venook said.
Because FOLFOX and FOLFIRI are equally effective on the overall survival front, the decision between them comes down to a balance between patient comorbidities and side effect profiles. Neuropathy, for instance, is more common with FOLFOX, whereas diarrhea is more likely with FOLFIRI, Venook said.
Venook favors FOLFIRI because “almost every patient will develop neuropathy” after seven or eight doses of FOLFOX, which limits its use. “You’re expecting that first treatment to give you the most mileage,” so starting with a treatment “you’re going to get limited use out of ... never made sense to me,” he said.
Venook noted that the results apply only to the older patients studied in CAIRO5 and not necessarily to the ever-growing population of younger people with CRC. Patients in the trial had a median age of 62 years with a performance status of 0-1, a median of 12 liver lesions with no metastases outside the liver, and no contraindications to local or systemic treatment.
The CAIRO5 analysis also looked at what happens after upfront chemotherapy, with the goal being to shrink liver lesions so the lesions can be surgically removed or treated with thermal ablation.
Almost half the patients ultimately underwent resection or ablation, and 39% of those in the FOLFOX or FOLFIRI plus bevacizumab group and 49% in the FOLFOX or FOLFIRI plus panitumumab group then went on to receive adjuvant chemotherapy (ACT) to reduce the risk for recurrence. ACT was recommended in the study, but not required, and consisted of chemotherapy minus bevacizumab or panitumumab.
Overall survival was longest among patients who had complete local treatment without recurrences for at least 6 months (64.3 months) or who had salvage local treatment after early recurrence (58.9 months). Median overall survival was 30.5 months for patients with complete local treatment without salvage after early recurrence, and 28.7 months for patients with incomplete local treatment. Overall survival was worst in patients who remained unresectable (18.3 months).
ACT was associated with improved overall and relapse-free survival, justifying discussing the option with patients who have completed local treatment, the study team said.
CAIRO5 was funded by Roche and Amgen, makers of bevacizumab and panitumumab, respectively. Bond and Venook didn’t have any disclosures.
A version of this article first appeared on Medscape.com.
A new report demonstrated why patients benefit most from starting on a two-drug chemotherapy regimen — FOLFOX or FOLFIRI — instead of a three-drug regimen — FOLFOXIRI.
The CAIRO5 trial compared overall survival among 294 patients with right sided tumors and/or RAS/BRAF mutations who received FOLFOXIRI (5-fluorouracil [FU], oxaliplatin, irinotecan, plus folinic acid as an enhancer) or investigators’ choice of FOLFOX (5-FU, oxaliplatin, and folinic acid) or FOLFIRI (5-FU, irinotecan, and folinic acid). All patients also received bevacizumab.
In a post hoc analysis, researchers found no overall survival benefit among patients receiving the three-drug regimen. At a median follow-up of just over 5 years, the median overall survival was 23.6 months with FOLFOX or FOLFIRI vs 24.1 months with FOLFOXIRI (P = .44).
The finding means that patients can avoid the extra toxicity associated with combining oxaliplatin and irinotecan without compromising overall survival, Alan P. Venook, MD, a gastrointestinal medical oncologist at the University of California San Francisco, told Medscape Medical News.
The analysis did not stop there in defining the optimal upfront therapy for this patient population.
In a second arm of the analysis, researchers looked at whether swapping in panitumumab for bevacizumab offered any benefit in 236 patients with left-sided tumors and wild-type RAS/BRAF who received either of the two-drug regimens.
Here, the authors also found no benefit of using panitumumab with FOLFOX or FOLFIRI instead of bevacizumab, reporting a median overall survival of 38.3 months with panitumumab vs 39.9 months with bevacizumab.
In addition to avoiding upfront FOLFOXIRI, patients can also avoid the skin reactions and other toxicities associated with panitumumab, including “horrible acne,” Venook said.
Overall, the results support the use of FOLFOX or FOLFIRI with bevacizumab “irrespective of RAS/BRAFV600E status and tumor sidedness” as the initial treatment for CRC with liver-only metastases, concluded the study investigators, from the University Medical Center Utrecht in the Netherlands.
Why Does This Clarity Matter?
The study confirms the standard practice in the United States of starting patients on a two-drug chemotherapy with bevacizumab for the indication and highlights “why we don’t go all in right at the beginning” with a three-drug regimen, Venook said.
In short, more drugs upfront isn’t going to change patients’ long-term survival outcome. Plus, using FOLFOXIRI upfront means “you’ve really pretty much used up all your guns for early treatment,” Venook said.
As for bevacizumab vs panitumumab, most practitioners in the United States favor bevacizumab because of the rash many patients on epidermal growth factor receptor blockers like panitumumab and cetuximab get, Venook said.
Because FOLFOX and FOLFIRI are equally effective on the overall survival front, the decision between them comes down to a balance between patient comorbidities and side effect profiles. Neuropathy, for instance, is more common with FOLFOX, whereas diarrhea is more likely with FOLFIRI, Venook said.
Venook favors FOLFIRI because “almost every patient will develop neuropathy” after seven or eight doses of FOLFOX, which limits its use. “You’re expecting that first treatment to give you the most mileage,” so starting with a treatment “you’re going to get limited use out of ... never made sense to me,” he said.
Venook noted that the results apply only to the older patients studied in CAIRO5 and not necessarily to the ever-growing population of younger people with CRC. Patients in the trial had a median age of 62 years with a performance status of 0-1, a median of 12 liver lesions with no metastases outside the liver, and no contraindications to local or systemic treatment.
The CAIRO5 analysis also looked at what happens after upfront chemotherapy, with the goal being to shrink liver lesions so the lesions can be surgically removed or treated with thermal ablation.
Almost half the patients ultimately underwent resection or ablation, and 39% of those in the FOLFOX or FOLFIRI plus bevacizumab group and 49% in the FOLFOX or FOLFIRI plus panitumumab group then went on to receive adjuvant chemotherapy (ACT) to reduce the risk for recurrence. ACT was recommended in the study, but not required, and consisted of chemotherapy minus bevacizumab or panitumumab.
Overall survival was longest among patients who had complete local treatment without recurrences for at least 6 months (64.3 months) or who had salvage local treatment after early recurrence (58.9 months). Median overall survival was 30.5 months for patients with complete local treatment without salvage after early recurrence, and 28.7 months for patients with incomplete local treatment. Overall survival was worst in patients who remained unresectable (18.3 months).
ACT was associated with improved overall and relapse-free survival, justifying discussing the option with patients who have completed local treatment, the study team said.
CAIRO5 was funded by Roche and Amgen, makers of bevacizumab and panitumumab, respectively. Bond and Venook didn’t have any disclosures.
A version of this article first appeared on Medscape.com.
A new report demonstrated why patients benefit most from starting on a two-drug chemotherapy regimen — FOLFOX or FOLFIRI — instead of a three-drug regimen — FOLFOXIRI.
The CAIRO5 trial compared overall survival among 294 patients with right sided tumors and/or RAS/BRAF mutations who received FOLFOXIRI (5-fluorouracil [FU], oxaliplatin, irinotecan, plus folinic acid as an enhancer) or investigators’ choice of FOLFOX (5-FU, oxaliplatin, and folinic acid) or FOLFIRI (5-FU, irinotecan, and folinic acid). All patients also received bevacizumab.
In a post hoc analysis, researchers found no overall survival benefit among patients receiving the three-drug regimen. At a median follow-up of just over 5 years, the median overall survival was 23.6 months with FOLFOX or FOLFIRI vs 24.1 months with FOLFOXIRI (P = .44).
The finding means that patients can avoid the extra toxicity associated with combining oxaliplatin and irinotecan without compromising overall survival, Alan P. Venook, MD, a gastrointestinal medical oncologist at the University of California San Francisco, told Medscape Medical News.
The analysis did not stop there in defining the optimal upfront therapy for this patient population.
In a second arm of the analysis, researchers looked at whether swapping in panitumumab for bevacizumab offered any benefit in 236 patients with left-sided tumors and wild-type RAS/BRAF who received either of the two-drug regimens.
Here, the authors also found no benefit of using panitumumab with FOLFOX or FOLFIRI instead of bevacizumab, reporting a median overall survival of 38.3 months with panitumumab vs 39.9 months with bevacizumab.
In addition to avoiding upfront FOLFOXIRI, patients can also avoid the skin reactions and other toxicities associated with panitumumab, including “horrible acne,” Venook said.
Overall, the results support the use of FOLFOX or FOLFIRI with bevacizumab “irrespective of RAS/BRAFV600E status and tumor sidedness” as the initial treatment for CRC with liver-only metastases, concluded the study investigators, from the University Medical Center Utrecht in the Netherlands.
Why Does This Clarity Matter?
The study confirms the standard practice in the United States of starting patients on a two-drug chemotherapy with bevacizumab for the indication and highlights “why we don’t go all in right at the beginning” with a three-drug regimen, Venook said.
In short, more drugs upfront isn’t going to change patients’ long-term survival outcome. Plus, using FOLFOXIRI upfront means “you’ve really pretty much used up all your guns for early treatment,” Venook said.
As for bevacizumab vs panitumumab, most practitioners in the United States favor bevacizumab because of the rash many patients on epidermal growth factor receptor blockers like panitumumab and cetuximab get, Venook said.
Because FOLFOX and FOLFIRI are equally effective on the overall survival front, the decision between them comes down to a balance between patient comorbidities and side effect profiles. Neuropathy, for instance, is more common with FOLFOX, whereas diarrhea is more likely with FOLFIRI, Venook said.
Venook favors FOLFIRI because “almost every patient will develop neuropathy” after seven or eight doses of FOLFOX, which limits its use. “You’re expecting that first treatment to give you the most mileage,” so starting with a treatment “you’re going to get limited use out of ... never made sense to me,” he said.
Venook noted that the results apply only to the older patients studied in CAIRO5 and not necessarily to the ever-growing population of younger people with CRC. Patients in the trial had a median age of 62 years with a performance status of 0-1, a median of 12 liver lesions with no metastases outside the liver, and no contraindications to local or systemic treatment.
The CAIRO5 analysis also looked at what happens after upfront chemotherapy, with the goal being to shrink liver lesions so the lesions can be surgically removed or treated with thermal ablation.
Almost half the patients ultimately underwent resection or ablation, and 39% of those in the FOLFOX or FOLFIRI plus bevacizumab group and 49% in the FOLFOX or FOLFIRI plus panitumumab group then went on to receive adjuvant chemotherapy (ACT) to reduce the risk for recurrence. ACT was recommended in the study, but not required, and consisted of chemotherapy minus bevacizumab or panitumumab.
Overall survival was longest among patients who had complete local treatment without recurrences for at least 6 months (64.3 months) or who had salvage local treatment after early recurrence (58.9 months). Median overall survival was 30.5 months for patients with complete local treatment without salvage after early recurrence, and 28.7 months for patients with incomplete local treatment. Overall survival was worst in patients who remained unresectable (18.3 months).
ACT was associated with improved overall and relapse-free survival, justifying discussing the option with patients who have completed local treatment, the study team said.
CAIRO5 was funded by Roche and Amgen, makers of bevacizumab and panitumumab, respectively. Bond and Venook didn’t have any disclosures.
A version of this article first appeared on Medscape.com.
FROM JAMA ONCOLOGY
Drugs Targeting Osteoarthritis Pain: What’s in Development?
WASHINGTON — Investigational treatments aimed specifically at reducing pain in knee osteoarthritis (OA) are moving forward in parallel with disease-modifying approaches.
“We still have very few treatments for the pain of osteoarthritis…It worries me that people think the only way forward is structure modification. I think while we’re waiting for some drugs to be structure modifying, we still need more pain relief. About 70% of people can’t tolerate or shouldn’t be on a [nonsteroidal anti-inflammatory drug], and that leaves a large number of people with pain,” Philip Conaghan, MBBS, PhD, Chair of Musculoskeletal Medicine at the University of Leeds in England, said in an interview.
At the annual meeting of the American College of Rheumatology, Conaghan, who is also honorary consultant rheumatologist for the Leeds Teaching Hospitals NHS Trust, presented new data for two novel approaches, both targeting peripheral nociceptive pain signaling.
In a late-breaking poster, he presented phase 2 trial data on RTX-GRT7039 (resiniferatoxin [RTX]), an agonist of the transient receptor potential vanilloid 1 that is a driver of OA pain. The trial investigated the efficacy and safety of a single intra-articular injection of RTX-GRT7039 in people with knee OA.
And separately, in a late-breaking oral abstract session, Conaghan presented phase 2 trial safety and efficacy data for another investigational agent called LEVI-04, a first-in-class neurotrophin receptor fusion protein (p75NTR-Fc) that supplements the endogenous protein and provides analgesia via inhibition of NT-3 activity.
“I think both have potential to provide good pain relief, through slightly different mechanisms,” Conaghan said in an interview.
Asked to comment, session moderator Gregory C. Gardner, MD, emeritus professor in the Division of Rheumatology at the University of Washington, Seattle, said in an interview: “I think the results are really exciting terms of the ability to control pain to a significant degree in patients with osteoarthritis.”
However, Gardner also said, “The molecules can be very expensive ... so who do we give them to? Will insurance companies pay for this simply for OA pain? They improve function ... so clearly, [they] will be a boon to treating osteoarthritis, but do we give them to people with only more advanced forms of osteoarthritis or earlier on?”
Moreover, Gardner said, “One of my concerns about treating osteoarthritis is I don’t want to do too good of a job treating pain in somebody who has a biomechanically abnormal joint. ... You’ve got a knee that’s worn out some of the cartilage, and now you feel like you can go out and play soccer again. That’s not a good thing. That joint will wear out very quickly, even though it doesn’t feel pain.”
Another OA expert, Matlock Jeffries, MD, director of the Arthritis Research Unit at the Oklahoma Medical Research Foundation, Oklahoma City, said in an interview, “I think we don’t focus nearly enough on pain, and that’s [partly] because the [Food and Drug Administration] has defined endpoints for knee OA trials that are radiographic. ... Patients do not care what their joint space narrowing is. They care what their pain is. And joint space changes and pain do not correlate in knee OA. ... About 20% or 30% of patients who have completely normal x-rays have a lot of pain…I hope that we’ll have some new OA pain therapeutics in the future because that’s what patients actually care about.”
But Jeffries noted that it will be very important to ensure that these agents don’t produce significant side effects, as had been seen previously in several large industry-sponsored trials of drugs targeting nerve growth factors.
“The big concern that we have in the field ... is that the nerve growth factor antibody trials were all stopped because there was a low but persistent risk of rapidly progressive OA in a small percent of patients. I think one of the questions in the field is whether targeting other things having to do with OA pain is going to result in similar bad outcomes. I think the answer is probably not, but that’s one thing that people do worry about, and they never really figured out why the [rapidly progressive OA] was happening.”
‘Potential to Provide Meaningful and Sustained Analgesia’
The phase 2 trial of RTX-GRT7039, funded by manufacturer Grünenthal, enrolled 40 patients with a baseline visual analog pain score (VAS) of > 40 mm on motion for average joint pain in the target knee over the past 2 days with or without analgesic medication and Kellgren-Lawrence grades 2-4.
They were randomized to receive a single intra-articular injection of 2 mg or 4 mg RTX-GRT7039 within 1 minute after receiving 5 mL ropivacaine (0.5%) or 4 mg or 8 mg RTX-GRT7039 administered 15 minutes after 5 mL ropivacaine pretreatment, or equivalent placebo treatments plus ropivacaine.
Plasma samples were collected for up to 2 hours, and VAS pain scores were collected for up to 3 hours post injection.
Reductions in VAS scores from baseline in the treated knee were seen in all RTX treatment groups as early as day 8 post injection and were maintained up to 6 months, while no reductions in VAS pain on motion scores were seen in the placebo group.
At 3 months, the absolute baseline-adjusted reductions in VAS scores were similar for RTX 2 mg (–39.75), RTX 4 mg (–40.20), and RTX 8 mg (–30.25), while the reduction in the placebo group was just –8.50. At 6 months, the mean absolute reduction in VAS score was numerically greater in the RTX 2-mg (–46.49), RTX 4-mg (–43.40), and RTX 8-mg (–38.60) groups vs the group that received RTX 4 mg within 1 minute after receiving ropivacaine (–22.00).
At both 3 and 6 months, a higher proportion of patients receiving any dose of RTX-GRT7039 achieved ≥ 50% and ≥ 70% reduction in pain on motion, compared with those who received placebo. All RTX-GRT7039 treatment groups reported a greater improvement in Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) total score than the placebo group at both 3 and 6 months.
Rates of treatment-emergent adverse events were similar between the RTX groups (85.7%-90.9%) and placebo (85.7%) and slightly lower in the group that received RTX 4 mg within 1 minute of receiving ropivacaine (60.0%).
There was a trend toward greater procedural/injection site pain in the RTX treatment groups, compared with placebo, most commonly arthralgia (37.5%), headache (17.5%), and back pain (10%). This tended to peak around 0.5 hours post injection and resolve by 1.5-3.0 hours.
No treatment-related serious adverse events occurred, and no treatment-emergent adverse events led to discontinuation or death.
“This early-phase trial indicates that RTX-GRT7039 has the potential to provide meaningful and sustained analgesia for patients with knee OA pain,” Conaghan and colleagues wrote in their poster.
The drug is now being evaluated in three phase 3 trials (NCT05248386, NCT05449132, and NCT05377489).
LEVI-04: Modulation of NT-3 Appears to Work Safely
LEVI-04 was evaluated in a phase 2, 20-week, 13-center (Europe and Hong Kong) randomized, double-blind, placebo-controlled trial in 518 people with knee OA who had WOMAC pain subscale scores ≥ 20, mean average daily pain numeric rating scale score of 4-9, and radiographic Kellgren-Lawrence grade ≥ 2.
They were randomized to a total of five infusions of placebo or 0.3 mg/kg, 0.1 mg/kg, or 2 mg/kg LEVI-04 from baseline through week 16, with safety follow-up to week 30.
The primary endpoint, change in WOMAC pain from baseline to weeks 5 and 17, was met for all three doses. At 17 weeks, those were –2.79, –2.89, and –3.08 for 0.3 mg, 1.0 mg, and 2 mg, respectively, vs –2.28 for placebo (all P < .05).
Secondary endpoints, including WOMAC physical function, WOMAC stiffness, and Patient Global Assessment, and > 50% pain responders, were also all met at weeks 5 and 17. More than 50% of the LEVI-04–treated patients reported ≥ 50% reduction in pain, and > 25% reported ≥ 75% reduction at weeks 5 and 17.
“So, this modulation of NT-3 is working,” Conaghan commented.
There were no increased incidences of severe adverse events, treatment-emergent adverse events, or joint pathologies, including rapidly progressive OA, compared with placebo.
There were more paresthesias reported with the active drug, 2-4 vs 1 with placebo. “That says to me that the drug is working and that it’s having an effect on peripheral nerves, but luckily these were all mild or moderate and didn’t lead to any study withdrawal or discontinuation,” Conaghan said.
Phase 3 trials are in the planning stages, he noted.
Other Approaches to Treating OA Pain
Other approaches to treating OA pain have included methotrexate, for which Conaghan was also a coauthor on one paper that came out earlier in 2024. “This presumably works by treating inflammation, but it’s not clear if that is within-joint inflammation or systemic inflammation,” he said in an interview.
Another approach, using the weight loss drug semaglutide, was presented in April 2024 at the 2024 World Congress on Osteoarthritis annual meeting and published in October 2024 in The New England Journal of Medicine
The trial involving RTX-GRT7039 was funded by Grünenthal, and some study coauthors are employees of the company. The trial involving LEVI-04 was funded by Levicept, and some study coauthors are employees of the company. Conaghan is a consultant and/or speaker for Eli Lilly, Eupraxia Pharmaceuticals, Formation Bio, Galapagos, Genascence, GlaxoSmithKline, Grünenthal, Janssen Pharmaceuticals, Kolon TissueGene, Levicept, Medipost, Moebius, Novartis, Pacira, Sandoz, Stryker Corporation, and Takeda. Gardner and Jeffries had no disclosures.
A version of this article appeared on Medscape.com.
WASHINGTON — Investigational treatments aimed specifically at reducing pain in knee osteoarthritis (OA) are moving forward in parallel with disease-modifying approaches.
“We still have very few treatments for the pain of osteoarthritis…It worries me that people think the only way forward is structure modification. I think while we’re waiting for some drugs to be structure modifying, we still need more pain relief. About 70% of people can’t tolerate or shouldn’t be on a [nonsteroidal anti-inflammatory drug], and that leaves a large number of people with pain,” Philip Conaghan, MBBS, PhD, Chair of Musculoskeletal Medicine at the University of Leeds in England, said in an interview.
At the annual meeting of the American College of Rheumatology, Conaghan, who is also honorary consultant rheumatologist for the Leeds Teaching Hospitals NHS Trust, presented new data for two novel approaches, both targeting peripheral nociceptive pain signaling.
In a late-breaking poster, he presented phase 2 trial data on RTX-GRT7039 (resiniferatoxin [RTX]), an agonist of the transient receptor potential vanilloid 1 that is a driver of OA pain. The trial investigated the efficacy and safety of a single intra-articular injection of RTX-GRT7039 in people with knee OA.
And separately, in a late-breaking oral abstract session, Conaghan presented phase 2 trial safety and efficacy data for another investigational agent called LEVI-04, a first-in-class neurotrophin receptor fusion protein (p75NTR-Fc) that supplements the endogenous protein and provides analgesia via inhibition of NT-3 activity.
“I think both have potential to provide good pain relief, through slightly different mechanisms,” Conaghan said in an interview.
Asked to comment, session moderator Gregory C. Gardner, MD, emeritus professor in the Division of Rheumatology at the University of Washington, Seattle, said in an interview: “I think the results are really exciting terms of the ability to control pain to a significant degree in patients with osteoarthritis.”
However, Gardner also said, “The molecules can be very expensive ... so who do we give them to? Will insurance companies pay for this simply for OA pain? They improve function ... so clearly, [they] will be a boon to treating osteoarthritis, but do we give them to people with only more advanced forms of osteoarthritis or earlier on?”
Moreover, Gardner said, “One of my concerns about treating osteoarthritis is I don’t want to do too good of a job treating pain in somebody who has a biomechanically abnormal joint. ... You’ve got a knee that’s worn out some of the cartilage, and now you feel like you can go out and play soccer again. That’s not a good thing. That joint will wear out very quickly, even though it doesn’t feel pain.”
Another OA expert, Matlock Jeffries, MD, director of the Arthritis Research Unit at the Oklahoma Medical Research Foundation, Oklahoma City, said in an interview, “I think we don’t focus nearly enough on pain, and that’s [partly] because the [Food and Drug Administration] has defined endpoints for knee OA trials that are radiographic. ... Patients do not care what their joint space narrowing is. They care what their pain is. And joint space changes and pain do not correlate in knee OA. ... About 20% or 30% of patients who have completely normal x-rays have a lot of pain…I hope that we’ll have some new OA pain therapeutics in the future because that’s what patients actually care about.”
But Jeffries noted that it will be very important to ensure that these agents don’t produce significant side effects, as had been seen previously in several large industry-sponsored trials of drugs targeting nerve growth factors.
“The big concern that we have in the field ... is that the nerve growth factor antibody trials were all stopped because there was a low but persistent risk of rapidly progressive OA in a small percent of patients. I think one of the questions in the field is whether targeting other things having to do with OA pain is going to result in similar bad outcomes. I think the answer is probably not, but that’s one thing that people do worry about, and they never really figured out why the [rapidly progressive OA] was happening.”
‘Potential to Provide Meaningful and Sustained Analgesia’
The phase 2 trial of RTX-GRT7039, funded by manufacturer Grünenthal, enrolled 40 patients with a baseline visual analog pain score (VAS) of > 40 mm on motion for average joint pain in the target knee over the past 2 days with or without analgesic medication and Kellgren-Lawrence grades 2-4.
They were randomized to receive a single intra-articular injection of 2 mg or 4 mg RTX-GRT7039 within 1 minute after receiving 5 mL ropivacaine (0.5%) or 4 mg or 8 mg RTX-GRT7039 administered 15 minutes after 5 mL ropivacaine pretreatment, or equivalent placebo treatments plus ropivacaine.
Plasma samples were collected for up to 2 hours, and VAS pain scores were collected for up to 3 hours post injection.
Reductions in VAS scores from baseline in the treated knee were seen in all RTX treatment groups as early as day 8 post injection and were maintained up to 6 months, while no reductions in VAS pain on motion scores were seen in the placebo group.
At 3 months, the absolute baseline-adjusted reductions in VAS scores were similar for RTX 2 mg (–39.75), RTX 4 mg (–40.20), and RTX 8 mg (–30.25), while the reduction in the placebo group was just –8.50. At 6 months, the mean absolute reduction in VAS score was numerically greater in the RTX 2-mg (–46.49), RTX 4-mg (–43.40), and RTX 8-mg (–38.60) groups vs the group that received RTX 4 mg within 1 minute after receiving ropivacaine (–22.00).
At both 3 and 6 months, a higher proportion of patients receiving any dose of RTX-GRT7039 achieved ≥ 50% and ≥ 70% reduction in pain on motion, compared with those who received placebo. All RTX-GRT7039 treatment groups reported a greater improvement in Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) total score than the placebo group at both 3 and 6 months.
Rates of treatment-emergent adverse events were similar between the RTX groups (85.7%-90.9%) and placebo (85.7%) and slightly lower in the group that received RTX 4 mg within 1 minute of receiving ropivacaine (60.0%).
There was a trend toward greater procedural/injection site pain in the RTX treatment groups, compared with placebo, most commonly arthralgia (37.5%), headache (17.5%), and back pain (10%). This tended to peak around 0.5 hours post injection and resolve by 1.5-3.0 hours.
No treatment-related serious adverse events occurred, and no treatment-emergent adverse events led to discontinuation or death.
“This early-phase trial indicates that RTX-GRT7039 has the potential to provide meaningful and sustained analgesia for patients with knee OA pain,” Conaghan and colleagues wrote in their poster.
The drug is now being evaluated in three phase 3 trials (NCT05248386, NCT05449132, and NCT05377489).
LEVI-04: Modulation of NT-3 Appears to Work Safely
LEVI-04 was evaluated in a phase 2, 20-week, 13-center (Europe and Hong Kong) randomized, double-blind, placebo-controlled trial in 518 people with knee OA who had WOMAC pain subscale scores ≥ 20, mean average daily pain numeric rating scale score of 4-9, and radiographic Kellgren-Lawrence grade ≥ 2.
They were randomized to a total of five infusions of placebo or 0.3 mg/kg, 0.1 mg/kg, or 2 mg/kg LEVI-04 from baseline through week 16, with safety follow-up to week 30.
The primary endpoint, change in WOMAC pain from baseline to weeks 5 and 17, was met for all three doses. At 17 weeks, those were –2.79, –2.89, and –3.08 for 0.3 mg, 1.0 mg, and 2 mg, respectively, vs –2.28 for placebo (all P < .05).
Secondary endpoints, including WOMAC physical function, WOMAC stiffness, and Patient Global Assessment, and > 50% pain responders, were also all met at weeks 5 and 17. More than 50% of the LEVI-04–treated patients reported ≥ 50% reduction in pain, and > 25% reported ≥ 75% reduction at weeks 5 and 17.
“So, this modulation of NT-3 is working,” Conaghan commented.
There were no increased incidences of severe adverse events, treatment-emergent adverse events, or joint pathologies, including rapidly progressive OA, compared with placebo.
There were more paresthesias reported with the active drug, 2-4 vs 1 with placebo. “That says to me that the drug is working and that it’s having an effect on peripheral nerves, but luckily these were all mild or moderate and didn’t lead to any study withdrawal or discontinuation,” Conaghan said.
Phase 3 trials are in the planning stages, he noted.
Other Approaches to Treating OA Pain
Other approaches to treating OA pain have included methotrexate, for which Conaghan was also a coauthor on one paper that came out earlier in 2024. “This presumably works by treating inflammation, but it’s not clear if that is within-joint inflammation or systemic inflammation,” he said in an interview.
Another approach, using the weight loss drug semaglutide, was presented in April 2024 at the 2024 World Congress on Osteoarthritis annual meeting and published in October 2024 in The New England Journal of Medicine
The trial involving RTX-GRT7039 was funded by Grünenthal, and some study coauthors are employees of the company. The trial involving LEVI-04 was funded by Levicept, and some study coauthors are employees of the company. Conaghan is a consultant and/or speaker for Eli Lilly, Eupraxia Pharmaceuticals, Formation Bio, Galapagos, Genascence, GlaxoSmithKline, Grünenthal, Janssen Pharmaceuticals, Kolon TissueGene, Levicept, Medipost, Moebius, Novartis, Pacira, Sandoz, Stryker Corporation, and Takeda. Gardner and Jeffries had no disclosures.
A version of this article appeared on Medscape.com.
WASHINGTON — Investigational treatments aimed specifically at reducing pain in knee osteoarthritis (OA) are moving forward in parallel with disease-modifying approaches.
“We still have very few treatments for the pain of osteoarthritis…It worries me that people think the only way forward is structure modification. I think while we’re waiting for some drugs to be structure modifying, we still need more pain relief. About 70% of people can’t tolerate or shouldn’t be on a [nonsteroidal anti-inflammatory drug], and that leaves a large number of people with pain,” Philip Conaghan, MBBS, PhD, Chair of Musculoskeletal Medicine at the University of Leeds in England, said in an interview.
At the annual meeting of the American College of Rheumatology, Conaghan, who is also honorary consultant rheumatologist for the Leeds Teaching Hospitals NHS Trust, presented new data for two novel approaches, both targeting peripheral nociceptive pain signaling.
In a late-breaking poster, he presented phase 2 trial data on RTX-GRT7039 (resiniferatoxin [RTX]), an agonist of the transient receptor potential vanilloid 1 that is a driver of OA pain. The trial investigated the efficacy and safety of a single intra-articular injection of RTX-GRT7039 in people with knee OA.
And separately, in a late-breaking oral abstract session, Conaghan presented phase 2 trial safety and efficacy data for another investigational agent called LEVI-04, a first-in-class neurotrophin receptor fusion protein (p75NTR-Fc) that supplements the endogenous protein and provides analgesia via inhibition of NT-3 activity.
“I think both have potential to provide good pain relief, through slightly different mechanisms,” Conaghan said in an interview.
Asked to comment, session moderator Gregory C. Gardner, MD, emeritus professor in the Division of Rheumatology at the University of Washington, Seattle, said in an interview: “I think the results are really exciting terms of the ability to control pain to a significant degree in patients with osteoarthritis.”
However, Gardner also said, “The molecules can be very expensive ... so who do we give them to? Will insurance companies pay for this simply for OA pain? They improve function ... so clearly, [they] will be a boon to treating osteoarthritis, but do we give them to people with only more advanced forms of osteoarthritis or earlier on?”
Moreover, Gardner said, “One of my concerns about treating osteoarthritis is I don’t want to do too good of a job treating pain in somebody who has a biomechanically abnormal joint. ... You’ve got a knee that’s worn out some of the cartilage, and now you feel like you can go out and play soccer again. That’s not a good thing. That joint will wear out very quickly, even though it doesn’t feel pain.”
Another OA expert, Matlock Jeffries, MD, director of the Arthritis Research Unit at the Oklahoma Medical Research Foundation, Oklahoma City, said in an interview, “I think we don’t focus nearly enough on pain, and that’s [partly] because the [Food and Drug Administration] has defined endpoints for knee OA trials that are radiographic. ... Patients do not care what their joint space narrowing is. They care what their pain is. And joint space changes and pain do not correlate in knee OA. ... About 20% or 30% of patients who have completely normal x-rays have a lot of pain…I hope that we’ll have some new OA pain therapeutics in the future because that’s what patients actually care about.”
But Jeffries noted that it will be very important to ensure that these agents don’t produce significant side effects, as had been seen previously in several large industry-sponsored trials of drugs targeting nerve growth factors.
“The big concern that we have in the field ... is that the nerve growth factor antibody trials were all stopped because there was a low but persistent risk of rapidly progressive OA in a small percent of patients. I think one of the questions in the field is whether targeting other things having to do with OA pain is going to result in similar bad outcomes. I think the answer is probably not, but that’s one thing that people do worry about, and they never really figured out why the [rapidly progressive OA] was happening.”
‘Potential to Provide Meaningful and Sustained Analgesia’
The phase 2 trial of RTX-GRT7039, funded by manufacturer Grünenthal, enrolled 40 patients with a baseline visual analog pain score (VAS) of > 40 mm on motion for average joint pain in the target knee over the past 2 days with or without analgesic medication and Kellgren-Lawrence grades 2-4.
They were randomized to receive a single intra-articular injection of 2 mg or 4 mg RTX-GRT7039 within 1 minute after receiving 5 mL ropivacaine (0.5%) or 4 mg or 8 mg RTX-GRT7039 administered 15 minutes after 5 mL ropivacaine pretreatment, or equivalent placebo treatments plus ropivacaine.
Plasma samples were collected for up to 2 hours, and VAS pain scores were collected for up to 3 hours post injection.
Reductions in VAS scores from baseline in the treated knee were seen in all RTX treatment groups as early as day 8 post injection and were maintained up to 6 months, while no reductions in VAS pain on motion scores were seen in the placebo group.
At 3 months, the absolute baseline-adjusted reductions in VAS scores were similar for RTX 2 mg (–39.75), RTX 4 mg (–40.20), and RTX 8 mg (–30.25), while the reduction in the placebo group was just –8.50. At 6 months, the mean absolute reduction in VAS score was numerically greater in the RTX 2-mg (–46.49), RTX 4-mg (–43.40), and RTX 8-mg (–38.60) groups vs the group that received RTX 4 mg within 1 minute after receiving ropivacaine (–22.00).
At both 3 and 6 months, a higher proportion of patients receiving any dose of RTX-GRT7039 achieved ≥ 50% and ≥ 70% reduction in pain on motion, compared with those who received placebo. All RTX-GRT7039 treatment groups reported a greater improvement in Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) total score than the placebo group at both 3 and 6 months.
Rates of treatment-emergent adverse events were similar between the RTX groups (85.7%-90.9%) and placebo (85.7%) and slightly lower in the group that received RTX 4 mg within 1 minute of receiving ropivacaine (60.0%).
There was a trend toward greater procedural/injection site pain in the RTX treatment groups, compared with placebo, most commonly arthralgia (37.5%), headache (17.5%), and back pain (10%). This tended to peak around 0.5 hours post injection and resolve by 1.5-3.0 hours.
No treatment-related serious adverse events occurred, and no treatment-emergent adverse events led to discontinuation or death.
“This early-phase trial indicates that RTX-GRT7039 has the potential to provide meaningful and sustained analgesia for patients with knee OA pain,” Conaghan and colleagues wrote in their poster.
The drug is now being evaluated in three phase 3 trials (NCT05248386, NCT05449132, and NCT05377489).
LEVI-04: Modulation of NT-3 Appears to Work Safely
LEVI-04 was evaluated in a phase 2, 20-week, 13-center (Europe and Hong Kong) randomized, double-blind, placebo-controlled trial in 518 people with knee OA who had WOMAC pain subscale scores ≥ 20, mean average daily pain numeric rating scale score of 4-9, and radiographic Kellgren-Lawrence grade ≥ 2.
They were randomized to a total of five infusions of placebo or 0.3 mg/kg, 0.1 mg/kg, or 2 mg/kg LEVI-04 from baseline through week 16, with safety follow-up to week 30.
The primary endpoint, change in WOMAC pain from baseline to weeks 5 and 17, was met for all three doses. At 17 weeks, those were –2.79, –2.89, and –3.08 for 0.3 mg, 1.0 mg, and 2 mg, respectively, vs –2.28 for placebo (all P < .05).
Secondary endpoints, including WOMAC physical function, WOMAC stiffness, and Patient Global Assessment, and > 50% pain responders, were also all met at weeks 5 and 17. More than 50% of the LEVI-04–treated patients reported ≥ 50% reduction in pain, and > 25% reported ≥ 75% reduction at weeks 5 and 17.
“So, this modulation of NT-3 is working,” Conaghan commented.
There were no increased incidences of severe adverse events, treatment-emergent adverse events, or joint pathologies, including rapidly progressive OA, compared with placebo.
There were more paresthesias reported with the active drug, 2-4 vs 1 with placebo. “That says to me that the drug is working and that it’s having an effect on peripheral nerves, but luckily these were all mild or moderate and didn’t lead to any study withdrawal or discontinuation,” Conaghan said.
Phase 3 trials are in the planning stages, he noted.
Other Approaches to Treating OA Pain
Other approaches to treating OA pain have included methotrexate, for which Conaghan was also a coauthor on one paper that came out earlier in 2024. “This presumably works by treating inflammation, but it’s not clear if that is within-joint inflammation or systemic inflammation,” he said in an interview.
Another approach, using the weight loss drug semaglutide, was presented in April 2024 at the 2024 World Congress on Osteoarthritis annual meeting and published in October 2024 in The New England Journal of Medicine
The trial involving RTX-GRT7039 was funded by Grünenthal, and some study coauthors are employees of the company. The trial involving LEVI-04 was funded by Levicept, and some study coauthors are employees of the company. Conaghan is a consultant and/or speaker for Eli Lilly, Eupraxia Pharmaceuticals, Formation Bio, Galapagos, Genascence, GlaxoSmithKline, Grünenthal, Janssen Pharmaceuticals, Kolon TissueGene, Levicept, Medipost, Moebius, Novartis, Pacira, Sandoz, Stryker Corporation, and Takeda. Gardner and Jeffries had no disclosures.
A version of this article appeared on Medscape.com.
FROM ACR 2024
Iron Overload: The Silent Bone Breaker
TOPLINE:
Patients with serum ferritin levels higher than 1000 μg/L show a 91% increased risk for any fracture, with a doubled risk for vertebral and humerus fractures compared with those without iron overload.
METHODOLOGY:
- Iron overload’s association with decreased bone mineral density is established, but its relationship to osteoporotic fracture risk has remained understudied and inconsistent across fracture sites.
- Researchers conducted a population-based cohort study using a UK general practice database to evaluate the fracture risk in 20,264 patients with iron overload and 192,956 matched controls without elevated ferritin (mean age, 57 years; about 40% women).
- Patients with iron overload were identified as those with laboratory-confirmed iron overload (serum ferritin levels > 1000 μg/L; n = 13,510) or a diagnosis of an iron overloading disorder, such as thalassemia major, sickle cell disease, or hemochromatosis (n = 6754).
- The primary outcome of interest was the first occurrence of an osteoporotic fracture after the diagnosis of iron overload or first record of high ferritin.
- A sensitivity analysis was conducted to check the impact of laboratory-confirmed iron overload on the risk for osteoporotic fracture compared with a diagnosis code without elevated ferritin.
TAKEAWAY:
- In the overall cohort, patients with iron overload had a 55% higher risk for any osteoporotic fracture than control individuals (adjusted hazard ratio [aHR], 1.55; 95% CI, 1.42-1.68), with the highest risk observed for vertebral fractures (aHR, 1.97; 95% CI, 1.63-2.37) and humerus fractures (aHR, 1.91; 95% CI, 1.61-2.26).
- Patients with laboratory-confirmed iron overload showed a 91% increased risk for any fracture (aHR, 1.91; 95% CI, 1.73-2.10), with a 2.5-fold higher risk observed for vertebral fractures (aHR, 2.51; 95% CI, 2.01-3.12), followed by humerus fractures (aHR, 2.41; 95% CI, 1.96-2.95).
- There was no increased risk for fracture at any site in patients with a diagnosis of an iron overloading disorder but no laboratory-confirmed iron overload.
- No sex-specific differences were identified in the association between iron overload and fracture risk.
IN PRACTICE:
“The main clinical message from our findings is that clinicians should consider iron overloading as a risk factor for fracture. Importantly, among high-risk patients presenting with serum ferritin values exceeding 1000 μg/L, osteoporosis screening and treatment strategies should be initiated in accordance with the guidelines for patients with hepatic disease,” the authors wrote.
SOURCE:
The study was led by Andrea Michelle Burden, PhD, Institute of Pharmaceutical Sciences, Department of Chemistry and Applied Biosciences, ETH Zürich in Switzerland, and was published online in The Journal of Clinical Endocrinology & Metabolism.
LIMITATIONS:
The study could not assess the duration of iron overload on fracture risk, and thus, patients could enter the cohort with a single elevated serum ferritin value that may not have reflected systemic iron overload. The authors also acknowledged potential exposure misclassification among matched control individuals because only 2.9% had a serum ferritin value available at baseline. Also, researchers were unable to adjust for inflammation status due to the limited availability of C-reactive protein measurements and the lack of leukocyte count data in primary care settings.
DISCLOSURES:
This study received support through grants from the German Research Foundation. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Patients with serum ferritin levels higher than 1000 μg/L show a 91% increased risk for any fracture, with a doubled risk for vertebral and humerus fractures compared with those without iron overload.
METHODOLOGY:
- Iron overload’s association with decreased bone mineral density is established, but its relationship to osteoporotic fracture risk has remained understudied and inconsistent across fracture sites.
- Researchers conducted a population-based cohort study using a UK general practice database to evaluate the fracture risk in 20,264 patients with iron overload and 192,956 matched controls without elevated ferritin (mean age, 57 years; about 40% women).
- Patients with iron overload were identified as those with laboratory-confirmed iron overload (serum ferritin levels > 1000 μg/L; n = 13,510) or a diagnosis of an iron overloading disorder, such as thalassemia major, sickle cell disease, or hemochromatosis (n = 6754).
- The primary outcome of interest was the first occurrence of an osteoporotic fracture after the diagnosis of iron overload or first record of high ferritin.
- A sensitivity analysis was conducted to check the impact of laboratory-confirmed iron overload on the risk for osteoporotic fracture compared with a diagnosis code without elevated ferritin.
TAKEAWAY:
- In the overall cohort, patients with iron overload had a 55% higher risk for any osteoporotic fracture than control individuals (adjusted hazard ratio [aHR], 1.55; 95% CI, 1.42-1.68), with the highest risk observed for vertebral fractures (aHR, 1.97; 95% CI, 1.63-2.37) and humerus fractures (aHR, 1.91; 95% CI, 1.61-2.26).
- Patients with laboratory-confirmed iron overload showed a 91% increased risk for any fracture (aHR, 1.91; 95% CI, 1.73-2.10), with a 2.5-fold higher risk observed for vertebral fractures (aHR, 2.51; 95% CI, 2.01-3.12), followed by humerus fractures (aHR, 2.41; 95% CI, 1.96-2.95).
- There was no increased risk for fracture at any site in patients with a diagnosis of an iron overloading disorder but no laboratory-confirmed iron overload.
- No sex-specific differences were identified in the association between iron overload and fracture risk.
IN PRACTICE:
“The main clinical message from our findings is that clinicians should consider iron overloading as a risk factor for fracture. Importantly, among high-risk patients presenting with serum ferritin values exceeding 1000 μg/L, osteoporosis screening and treatment strategies should be initiated in accordance with the guidelines for patients with hepatic disease,” the authors wrote.
SOURCE:
The study was led by Andrea Michelle Burden, PhD, Institute of Pharmaceutical Sciences, Department of Chemistry and Applied Biosciences, ETH Zürich in Switzerland, and was published online in The Journal of Clinical Endocrinology & Metabolism.
LIMITATIONS:
The study could not assess the duration of iron overload on fracture risk, and thus, patients could enter the cohort with a single elevated serum ferritin value that may not have reflected systemic iron overload. The authors also acknowledged potential exposure misclassification among matched control individuals because only 2.9% had a serum ferritin value available at baseline. Also, researchers were unable to adjust for inflammation status due to the limited availability of C-reactive protein measurements and the lack of leukocyte count data in primary care settings.
DISCLOSURES:
This study received support through grants from the German Research Foundation. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Patients with serum ferritin levels higher than 1000 μg/L show a 91% increased risk for any fracture, with a doubled risk for vertebral and humerus fractures compared with those without iron overload.
METHODOLOGY:
- Iron overload’s association with decreased bone mineral density is established, but its relationship to osteoporotic fracture risk has remained understudied and inconsistent across fracture sites.
- Researchers conducted a population-based cohort study using a UK general practice database to evaluate the fracture risk in 20,264 patients with iron overload and 192,956 matched controls without elevated ferritin (mean age, 57 years; about 40% women).
- Patients with iron overload were identified as those with laboratory-confirmed iron overload (serum ferritin levels > 1000 μg/L; n = 13,510) or a diagnosis of an iron overloading disorder, such as thalassemia major, sickle cell disease, or hemochromatosis (n = 6754).
- The primary outcome of interest was the first occurrence of an osteoporotic fracture after the diagnosis of iron overload or first record of high ferritin.
- A sensitivity analysis was conducted to check the impact of laboratory-confirmed iron overload on the risk for osteoporotic fracture compared with a diagnosis code without elevated ferritin.
TAKEAWAY:
- In the overall cohort, patients with iron overload had a 55% higher risk for any osteoporotic fracture than control individuals (adjusted hazard ratio [aHR], 1.55; 95% CI, 1.42-1.68), with the highest risk observed for vertebral fractures (aHR, 1.97; 95% CI, 1.63-2.37) and humerus fractures (aHR, 1.91; 95% CI, 1.61-2.26).
- Patients with laboratory-confirmed iron overload showed a 91% increased risk for any fracture (aHR, 1.91; 95% CI, 1.73-2.10), with a 2.5-fold higher risk observed for vertebral fractures (aHR, 2.51; 95% CI, 2.01-3.12), followed by humerus fractures (aHR, 2.41; 95% CI, 1.96-2.95).
- There was no increased risk for fracture at any site in patients with a diagnosis of an iron overloading disorder but no laboratory-confirmed iron overload.
- No sex-specific differences were identified in the association between iron overload and fracture risk.
IN PRACTICE:
“The main clinical message from our findings is that clinicians should consider iron overloading as a risk factor for fracture. Importantly, among high-risk patients presenting with serum ferritin values exceeding 1000 μg/L, osteoporosis screening and treatment strategies should be initiated in accordance with the guidelines for patients with hepatic disease,” the authors wrote.
SOURCE:
The study was led by Andrea Michelle Burden, PhD, Institute of Pharmaceutical Sciences, Department of Chemistry and Applied Biosciences, ETH Zürich in Switzerland, and was published online in The Journal of Clinical Endocrinology & Metabolism.
LIMITATIONS:
The study could not assess the duration of iron overload on fracture risk, and thus, patients could enter the cohort with a single elevated serum ferritin value that may not have reflected systemic iron overload. The authors also acknowledged potential exposure misclassification among matched control individuals because only 2.9% had a serum ferritin value available at baseline. Also, researchers were unable to adjust for inflammation status due to the limited availability of C-reactive protein measurements and the lack of leukocyte count data in primary care settings.
DISCLOSURES:
This study received support through grants from the German Research Foundation. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Most US Adults Plan to Skip Annual COVID Vaccines
Most US adults continue to plan on skipping an annual COVID vaccine.
according to results of a new survey from the Pew Research Center.
When asked why people wouldn’t get an updated COVID vaccine, 61% said a major reason was that they don’t think they need it, and 60% said a major reason is that they are concerned about side effects. Cost was a factor for 14% of people, and 46% of people said they don’t get vaccines in general.
There were some differences in intention to get vaccinated based on a person’s age. Among people ages 65 and older, 27% said they had already gotten the vaccine, and another 27% said they probably will get the shot, leaving 45% who said they probably won’t roll up their sleeves. People ages 30-49 years old were the least likely to plan on getting a COVID shot – 66% said they probably won’t get one.
Public health officials say everyone should get an annual COVID vaccine, just as they should get a flu shot, because the vaccines are formulated each year to target virus strains predicted to be in wide circulation. Also, immunity – either from past vaccination or past infection – wanes over time.
Research shows that the vaccines reduce the likelihood of hospitalization or death caused by severe illness, particularly among people who have risk factors, like being over age 65 or having health issues that are becoming increasingly common in the United States, like diabetes, heart problems, and lung conditions.
The survey included 9,593 adults who were asked about their COVID vaccine intentions with this question: “Public health officials recently recommended an updated vaccine for COVID-19. Do you think you will probably get an updated vaccine, probably not get an updated vaccine, or have you already received an updated vaccine?” The survey was done online and by telephone from October 21 to October 27.
So far in 2024, the CDC’s ongoing immunization survey shows that 17% of adults say that, as of November 2, they have gotten vaccinated for COVID-19 this season, and 14% said they will definitely get vaccinated. The Pew Research Center survey found that 15% of people said they’ve already gotten the shot this season.
Reports of positive COVID tests, emergency room visits, and hospitalizations remain very low. About 3.6% of test results shared with the CDC were positive for COVID the week ending November 9. Less than 1% of ER visits involve a COVID diagnosis, and hospitalizations are well below the rate seen at this time last year. Last year, COVID activity in the United States began rising around Thanksgiving and continued upward, peaking in early January.
The protection from a COVID-19 vaccination usually fully kicks in about 2 weeks after you get the shot, and the vaccines are most effective for the following 3 months.
A version of this article first appeared on WebMD.com.
Most US adults continue to plan on skipping an annual COVID vaccine.
according to results of a new survey from the Pew Research Center.
When asked why people wouldn’t get an updated COVID vaccine, 61% said a major reason was that they don’t think they need it, and 60% said a major reason is that they are concerned about side effects. Cost was a factor for 14% of people, and 46% of people said they don’t get vaccines in general.
There were some differences in intention to get vaccinated based on a person’s age. Among people ages 65 and older, 27% said they had already gotten the vaccine, and another 27% said they probably will get the shot, leaving 45% who said they probably won’t roll up their sleeves. People ages 30-49 years old were the least likely to plan on getting a COVID shot – 66% said they probably won’t get one.
Public health officials say everyone should get an annual COVID vaccine, just as they should get a flu shot, because the vaccines are formulated each year to target virus strains predicted to be in wide circulation. Also, immunity – either from past vaccination or past infection – wanes over time.
Research shows that the vaccines reduce the likelihood of hospitalization or death caused by severe illness, particularly among people who have risk factors, like being over age 65 or having health issues that are becoming increasingly common in the United States, like diabetes, heart problems, and lung conditions.
The survey included 9,593 adults who were asked about their COVID vaccine intentions with this question: “Public health officials recently recommended an updated vaccine for COVID-19. Do you think you will probably get an updated vaccine, probably not get an updated vaccine, or have you already received an updated vaccine?” The survey was done online and by telephone from October 21 to October 27.
So far in 2024, the CDC’s ongoing immunization survey shows that 17% of adults say that, as of November 2, they have gotten vaccinated for COVID-19 this season, and 14% said they will definitely get vaccinated. The Pew Research Center survey found that 15% of people said they’ve already gotten the shot this season.
Reports of positive COVID tests, emergency room visits, and hospitalizations remain very low. About 3.6% of test results shared with the CDC were positive for COVID the week ending November 9. Less than 1% of ER visits involve a COVID diagnosis, and hospitalizations are well below the rate seen at this time last year. Last year, COVID activity in the United States began rising around Thanksgiving and continued upward, peaking in early January.
The protection from a COVID-19 vaccination usually fully kicks in about 2 weeks after you get the shot, and the vaccines are most effective for the following 3 months.
A version of this article first appeared on WebMD.com.
Most US adults continue to plan on skipping an annual COVID vaccine.
according to results of a new survey from the Pew Research Center.
When asked why people wouldn’t get an updated COVID vaccine, 61% said a major reason was that they don’t think they need it, and 60% said a major reason is that they are concerned about side effects. Cost was a factor for 14% of people, and 46% of people said they don’t get vaccines in general.
There were some differences in intention to get vaccinated based on a person’s age. Among people ages 65 and older, 27% said they had already gotten the vaccine, and another 27% said they probably will get the shot, leaving 45% who said they probably won’t roll up their sleeves. People ages 30-49 years old were the least likely to plan on getting a COVID shot – 66% said they probably won’t get one.
Public health officials say everyone should get an annual COVID vaccine, just as they should get a flu shot, because the vaccines are formulated each year to target virus strains predicted to be in wide circulation. Also, immunity – either from past vaccination or past infection – wanes over time.
Research shows that the vaccines reduce the likelihood of hospitalization or death caused by severe illness, particularly among people who have risk factors, like being over age 65 or having health issues that are becoming increasingly common in the United States, like diabetes, heart problems, and lung conditions.
The survey included 9,593 adults who were asked about their COVID vaccine intentions with this question: “Public health officials recently recommended an updated vaccine for COVID-19. Do you think you will probably get an updated vaccine, probably not get an updated vaccine, or have you already received an updated vaccine?” The survey was done online and by telephone from October 21 to October 27.
So far in 2024, the CDC’s ongoing immunization survey shows that 17% of adults say that, as of November 2, they have gotten vaccinated for COVID-19 this season, and 14% said they will definitely get vaccinated. The Pew Research Center survey found that 15% of people said they’ve already gotten the shot this season.
Reports of positive COVID tests, emergency room visits, and hospitalizations remain very low. About 3.6% of test results shared with the CDC were positive for COVID the week ending November 9. Less than 1% of ER visits involve a COVID diagnosis, and hospitalizations are well below the rate seen at this time last year. Last year, COVID activity in the United States began rising around Thanksgiving and continued upward, peaking in early January.
The protection from a COVID-19 vaccination usually fully kicks in about 2 weeks after you get the shot, and the vaccines are most effective for the following 3 months.
A version of this article first appeared on WebMD.com.
Pertussis Cases Spike in November
Six times as many cases of pertussis were reported in the United States for the week ending November 16, 2024, as the same week in 2023, according to new data from the Centers for Disease Control and Prevention (CDC).
Of the 434 cases reported for the week ending November 16, 2024, a majority (109) occurred in the East North Central region, mostly in Ohio (93). Another 70 cases occurred in the West North Central region, with 32 cases and 37 cases in Missouri and Nebraska, respectively.
None of the 75 cases in the Middle Atlantic region occurred in New Jersey or New York City; 38 were reported elsewhere in New York, and 37 in Pennsylvania. The South Atlantic region reported 55 cases, including 29 in Florida. The East South Central and West South Central regions reported 11 and 20 cases, respectively. The Mountain and Pacific regions reported 31 (20 in Arizona) and 47 (20 in Washington State) cases, respectively.
The CDC tracks pertussis cases through a national surveillance system, but many cases are likely unrecognized and unreported, according to the CDC.
Although vaccines for pertussis (whooping cough) provide protection, their effectiveness decreases over time, and the CDC expects rates to increase in vaccinated and unvaccinated populations as case levels rebound with the lifting of pandemic mitigation strategies such as masking and remote learning.
Recent CDC data reported by Medscape Medical News showed an association between lower vaccination rates and 2024’s uptick in pertussis cases.
A version of this article first appeared on Medscape.com.
Six times as many cases of pertussis were reported in the United States for the week ending November 16, 2024, as the same week in 2023, according to new data from the Centers for Disease Control and Prevention (CDC).
Of the 434 cases reported for the week ending November 16, 2024, a majority (109) occurred in the East North Central region, mostly in Ohio (93). Another 70 cases occurred in the West North Central region, with 32 cases and 37 cases in Missouri and Nebraska, respectively.
None of the 75 cases in the Middle Atlantic region occurred in New Jersey or New York City; 38 were reported elsewhere in New York, and 37 in Pennsylvania. The South Atlantic region reported 55 cases, including 29 in Florida. The East South Central and West South Central regions reported 11 and 20 cases, respectively. The Mountain and Pacific regions reported 31 (20 in Arizona) and 47 (20 in Washington State) cases, respectively.
The CDC tracks pertussis cases through a national surveillance system, but many cases are likely unrecognized and unreported, according to the CDC.
Although vaccines for pertussis (whooping cough) provide protection, their effectiveness decreases over time, and the CDC expects rates to increase in vaccinated and unvaccinated populations as case levels rebound with the lifting of pandemic mitigation strategies such as masking and remote learning.
Recent CDC data reported by Medscape Medical News showed an association between lower vaccination rates and 2024’s uptick in pertussis cases.
A version of this article first appeared on Medscape.com.
Six times as many cases of pertussis were reported in the United States for the week ending November 16, 2024, as the same week in 2023, according to new data from the Centers for Disease Control and Prevention (CDC).
Of the 434 cases reported for the week ending November 16, 2024, a majority (109) occurred in the East North Central region, mostly in Ohio (93). Another 70 cases occurred in the West North Central region, with 32 cases and 37 cases in Missouri and Nebraska, respectively.
None of the 75 cases in the Middle Atlantic region occurred in New Jersey or New York City; 38 were reported elsewhere in New York, and 37 in Pennsylvania. The South Atlantic region reported 55 cases, including 29 in Florida. The East South Central and West South Central regions reported 11 and 20 cases, respectively. The Mountain and Pacific regions reported 31 (20 in Arizona) and 47 (20 in Washington State) cases, respectively.
The CDC tracks pertussis cases through a national surveillance system, but many cases are likely unrecognized and unreported, according to the CDC.
Although vaccines for pertussis (whooping cough) provide protection, their effectiveness decreases over time, and the CDC expects rates to increase in vaccinated and unvaccinated populations as case levels rebound with the lifting of pandemic mitigation strategies such as masking and remote learning.
Recent CDC data reported by Medscape Medical News showed an association between lower vaccination rates and 2024’s uptick in pertussis cases.
A version of this article first appeared on Medscape.com.
BCG Vaccine May Protect Against Long COVID Symptoms
TOPLINE:
METHODOLOGY:
- A phase 3 clinical trial initiated in early 2020 investigated the effect of the BCG vaccine injected during active infection on COVID-19 progression in adults with mild or moderate COVID-19. The current study summarizes the 6- and 12-month follow-up data with a focus on long-COVID symptoms.
- Patients who tested positive for severe acute respiratory syndrome coronavirus 2 were randomly assigned to receive either 0.1 mL of intradermal BCG (n = 191) or 0.9% saline placebo (n = 202) within 14 days of symptom onset and were followed up at 7, 14, 21, and 45 days and at 6 and 12 months postinjection.
- Overall, 157 BCG (median age, 40 years; 54.1% women) and 142 placebo (median age, 41 years; 65.5% women) recipients completed the 6-month follow-up, and 97 BCG (median age, 37 years; 49.5% women) and 95 placebo (median age, 40 years; 67.4% women) recipients completed the 12-month follow-up.
- The researchers primarily assessed the effect of the BCG vaccine on the development of the symptoms of long COVID at 6 and 12 months.
TAKEAWAY:
- Hearing problems were less frequent among BCG recipients at 6 months compared with those who received placebo (odds ratio [OR], 0.26; 95% CI, 0.045-1.0; P = .044).
- At 12 months, participants who received the BCG vaccine exhibited fewer issues with sleeping (P = .027), concentration (P = .009), memory (P = .009), and vision (P = .022) along with a lower long-COVID score (one-sided Wilcoxon test, P = .002) than those who received placebo.
- At 6 months, BCG demonstrated a sex-specific paradoxical effect on hair loss, decreasing it in men (P = .031), while causing a slight, though statistically nonsignificant, increase in women.
- Male sex was the strongest predictive factor for long COVID, cognitive dysfunction, and cardiopulmonary scores at both follow-up assessments.
IN PRACTICE:
“[The study] findings suggest that BCG immunotherapy for an existing ailment may be superior to prophylaxis in healthy individuals,” the authors wrote.
SOURCE:
The study was led by Mehrsa Jalalizadeh and Keini Buosi, UroScience, State University of Campinas, Unicamp, São Paulo, Brazil. It was published online on November 19, 2024, in the Journal of Internal Medicine.
LIMITATIONS:
Previous mycobacterial exposure was not tested among the study participants. A notable loss to follow-up, particularly at 12 months, may have introduced bias into the results.
DISCLOSURES:
The study was supported by the Coordination for the Improvement of Higher Education Personnel, Federal Government of Brazil, the General Coordination of the National Immunization Program, Ministry of Health (Brazil), and the National Council for Scientific and Technological Development-Research Productivity. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- A phase 3 clinical trial initiated in early 2020 investigated the effect of the BCG vaccine injected during active infection on COVID-19 progression in adults with mild or moderate COVID-19. The current study summarizes the 6- and 12-month follow-up data with a focus on long-COVID symptoms.
- Patients who tested positive for severe acute respiratory syndrome coronavirus 2 were randomly assigned to receive either 0.1 mL of intradermal BCG (n = 191) or 0.9% saline placebo (n = 202) within 14 days of symptom onset and were followed up at 7, 14, 21, and 45 days and at 6 and 12 months postinjection.
- Overall, 157 BCG (median age, 40 years; 54.1% women) and 142 placebo (median age, 41 years; 65.5% women) recipients completed the 6-month follow-up, and 97 BCG (median age, 37 years; 49.5% women) and 95 placebo (median age, 40 years; 67.4% women) recipients completed the 12-month follow-up.
- The researchers primarily assessed the effect of the BCG vaccine on the development of the symptoms of long COVID at 6 and 12 months.
TAKEAWAY:
- Hearing problems were less frequent among BCG recipients at 6 months compared with those who received placebo (odds ratio [OR], 0.26; 95% CI, 0.045-1.0; P = .044).
- At 12 months, participants who received the BCG vaccine exhibited fewer issues with sleeping (P = .027), concentration (P = .009), memory (P = .009), and vision (P = .022) along with a lower long-COVID score (one-sided Wilcoxon test, P = .002) than those who received placebo.
- At 6 months, BCG demonstrated a sex-specific paradoxical effect on hair loss, decreasing it in men (P = .031), while causing a slight, though statistically nonsignificant, increase in women.
- Male sex was the strongest predictive factor for long COVID, cognitive dysfunction, and cardiopulmonary scores at both follow-up assessments.
IN PRACTICE:
“[The study] findings suggest that BCG immunotherapy for an existing ailment may be superior to prophylaxis in healthy individuals,” the authors wrote.
SOURCE:
The study was led by Mehrsa Jalalizadeh and Keini Buosi, UroScience, State University of Campinas, Unicamp, São Paulo, Brazil. It was published online on November 19, 2024, in the Journal of Internal Medicine.
LIMITATIONS:
Previous mycobacterial exposure was not tested among the study participants. A notable loss to follow-up, particularly at 12 months, may have introduced bias into the results.
DISCLOSURES:
The study was supported by the Coordination for the Improvement of Higher Education Personnel, Federal Government of Brazil, the General Coordination of the National Immunization Program, Ministry of Health (Brazil), and the National Council for Scientific and Technological Development-Research Productivity. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- A phase 3 clinical trial initiated in early 2020 investigated the effect of the BCG vaccine injected during active infection on COVID-19 progression in adults with mild or moderate COVID-19. The current study summarizes the 6- and 12-month follow-up data with a focus on long-COVID symptoms.
- Patients who tested positive for severe acute respiratory syndrome coronavirus 2 were randomly assigned to receive either 0.1 mL of intradermal BCG (n = 191) or 0.9% saline placebo (n = 202) within 14 days of symptom onset and were followed up at 7, 14, 21, and 45 days and at 6 and 12 months postinjection.
- Overall, 157 BCG (median age, 40 years; 54.1% women) and 142 placebo (median age, 41 years; 65.5% women) recipients completed the 6-month follow-up, and 97 BCG (median age, 37 years; 49.5% women) and 95 placebo (median age, 40 years; 67.4% women) recipients completed the 12-month follow-up.
- The researchers primarily assessed the effect of the BCG vaccine on the development of the symptoms of long COVID at 6 and 12 months.
TAKEAWAY:
- Hearing problems were less frequent among BCG recipients at 6 months compared with those who received placebo (odds ratio [OR], 0.26; 95% CI, 0.045-1.0; P = .044).
- At 12 months, participants who received the BCG vaccine exhibited fewer issues with sleeping (P = .027), concentration (P = .009), memory (P = .009), and vision (P = .022) along with a lower long-COVID score (one-sided Wilcoxon test, P = .002) than those who received placebo.
- At 6 months, BCG demonstrated a sex-specific paradoxical effect on hair loss, decreasing it in men (P = .031), while causing a slight, though statistically nonsignificant, increase in women.
- Male sex was the strongest predictive factor for long COVID, cognitive dysfunction, and cardiopulmonary scores at both follow-up assessments.
IN PRACTICE:
“[The study] findings suggest that BCG immunotherapy for an existing ailment may be superior to prophylaxis in healthy individuals,” the authors wrote.
SOURCE:
The study was led by Mehrsa Jalalizadeh and Keini Buosi, UroScience, State University of Campinas, Unicamp, São Paulo, Brazil. It was published online on November 19, 2024, in the Journal of Internal Medicine.
LIMITATIONS:
Previous mycobacterial exposure was not tested among the study participants. A notable loss to follow-up, particularly at 12 months, may have introduced bias into the results.
DISCLOSURES:
The study was supported by the Coordination for the Improvement of Higher Education Personnel, Federal Government of Brazil, the General Coordination of the National Immunization Program, Ministry of Health (Brazil), and the National Council for Scientific and Technological Development-Research Productivity. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.

