User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Q&A: What to know about the new BA 2.86 COVID variant
The Centers for Disease Control and Prevention and the World Health Organization have dubbed the BA 2.86 variant of COVID-19 as a variant to watch.
So far, only 26 cases of “Pirola,” as the new variant is being called, have been identified: 10 in Denmark, four each in Sweden and the United States, three in South Africa, two in Portugal, and one each the United Kingdom, Israel, and Canada. BA 2.86 is a subvariant of Omicron, but according to reports from the CDC, the strain has many more mutations than the ones that came before it.
With so many facts still unknown about this new variant, this news organization asked experts what people need to be aware of as it continues to spread.
What is unique about the BA 2.86 variant?
“It is unique in that it has more than three mutations on the spike protein,” said Purvi S. Parikh, MD, an infectious disease expert at New York University’s Langone Health. The virus uses the spike proteins to enter our cells.
This “may mean it will be more transmissible, cause more severe disease, and/or our vaccines and treatments may not work as well, as compared to other variants,” she said.
What do we need to watch with BA 2.86 going forward?
“We don’t know if this variant will be associated with a change in the disease severity. We currently see increased numbers of cases in general, even though we don’t yet see the BA.2.86 in our system,” said Heba Mostafa, PhD, director of the molecular virology laboratory at Johns Hopkins Hospital in Baltimore.
“It is important to monitor BA.2.86 (and other variants) and understand how its evolution impacts the number of cases and disease outcomes,” she said. “We should all be aware of the current increase in cases, though, and try to get tested and be treated as soon as possible, as antivirals should be effective against the circulating variants.”
What should doctors know?
Dr. Parikh said doctors should generally expect more COVID cases in their clinics and make sure to screen patients even if their symptoms are mild.
“We have tools that can be used – antivirals like Paxlovid are still efficacious with current dominant strains such as EG.5,” she said. “And encourage your patients to get their boosters, mask, wash hands, and social distance.”
How well can our vaccines fight BA 2.86?
“Vaccine coverage for the BA.2.86 is an area of uncertainty right now,” said Dr. Mostafa.
In its report, the CDC said scientists are still figuring out how well the updated COVID vaccine works. It’s expected to be available in the fall, and for now, they believe the new shot will still make infections less severe, new variants and all.
Prior vaccinations and infections have created antibodies in many people, and that will likely provide some protection, Dr. Mostafa said. “When we experienced the Omicron wave in December 2021, even though the variant was distant from what circulated before its emergence and was associated with a very large increase in the number of cases, vaccinations were still protective against severe disease.”
What is the most important thing to keep track of when it comes to this variant?
According to Dr. Parikh, “it’s most important to monitor how transmissible [BA 2.86] is, how severe it is, and if our current treatments and vaccines work.”
Dr. Mostafa said how well the new variants escape existing antibody protection should also be studied and watched closely.
What does this stage of the virus mutation tell us about where we are in the pandemic?
The history of the coronavirus over the past few years shows that variants with many changes evolve and can spread very quickly, Dr. Mostafa said. “Now that the virus is endemic, it is essential to monitor, update vaccinations if necessary, diagnose, treat, and implement infection control measures when necessary.”
With the limited data we have so far, experts seem to agree that while the variant’s makeup raises some red flags, it is too soon to jump to any conclusions about how easy it is to catch it and the ways it may change how the virus impacts those who contract it.
A version of this article first appeared on WebMD.com.
The Centers for Disease Control and Prevention and the World Health Organization have dubbed the BA 2.86 variant of COVID-19 as a variant to watch.
So far, only 26 cases of “Pirola,” as the new variant is being called, have been identified: 10 in Denmark, four each in Sweden and the United States, three in South Africa, two in Portugal, and one each the United Kingdom, Israel, and Canada. BA 2.86 is a subvariant of Omicron, but according to reports from the CDC, the strain has many more mutations than the ones that came before it.
With so many facts still unknown about this new variant, this news organization asked experts what people need to be aware of as it continues to spread.
What is unique about the BA 2.86 variant?
“It is unique in that it has more than three mutations on the spike protein,” said Purvi S. Parikh, MD, an infectious disease expert at New York University’s Langone Health. The virus uses the spike proteins to enter our cells.
This “may mean it will be more transmissible, cause more severe disease, and/or our vaccines and treatments may not work as well, as compared to other variants,” she said.
What do we need to watch with BA 2.86 going forward?
“We don’t know if this variant will be associated with a change in the disease severity. We currently see increased numbers of cases in general, even though we don’t yet see the BA.2.86 in our system,” said Heba Mostafa, PhD, director of the molecular virology laboratory at Johns Hopkins Hospital in Baltimore.
“It is important to monitor BA.2.86 (and other variants) and understand how its evolution impacts the number of cases and disease outcomes,” she said. “We should all be aware of the current increase in cases, though, and try to get tested and be treated as soon as possible, as antivirals should be effective against the circulating variants.”
What should doctors know?
Dr. Parikh said doctors should generally expect more COVID cases in their clinics and make sure to screen patients even if their symptoms are mild.
“We have tools that can be used – antivirals like Paxlovid are still efficacious with current dominant strains such as EG.5,” she said. “And encourage your patients to get their boosters, mask, wash hands, and social distance.”
How well can our vaccines fight BA 2.86?
“Vaccine coverage for the BA.2.86 is an area of uncertainty right now,” said Dr. Mostafa.
In its report, the CDC said scientists are still figuring out how well the updated COVID vaccine works. It’s expected to be available in the fall, and for now, they believe the new shot will still make infections less severe, new variants and all.
Prior vaccinations and infections have created antibodies in many people, and that will likely provide some protection, Dr. Mostafa said. “When we experienced the Omicron wave in December 2021, even though the variant was distant from what circulated before its emergence and was associated with a very large increase in the number of cases, vaccinations were still protective against severe disease.”
What is the most important thing to keep track of when it comes to this variant?
According to Dr. Parikh, “it’s most important to monitor how transmissible [BA 2.86] is, how severe it is, and if our current treatments and vaccines work.”
Dr. Mostafa said how well the new variants escape existing antibody protection should also be studied and watched closely.
What does this stage of the virus mutation tell us about where we are in the pandemic?
The history of the coronavirus over the past few years shows that variants with many changes evolve and can spread very quickly, Dr. Mostafa said. “Now that the virus is endemic, it is essential to monitor, update vaccinations if necessary, diagnose, treat, and implement infection control measures when necessary.”
With the limited data we have so far, experts seem to agree that while the variant’s makeup raises some red flags, it is too soon to jump to any conclusions about how easy it is to catch it and the ways it may change how the virus impacts those who contract it.
A version of this article first appeared on WebMD.com.
The Centers for Disease Control and Prevention and the World Health Organization have dubbed the BA 2.86 variant of COVID-19 as a variant to watch.
So far, only 26 cases of “Pirola,” as the new variant is being called, have been identified: 10 in Denmark, four each in Sweden and the United States, three in South Africa, two in Portugal, and one each the United Kingdom, Israel, and Canada. BA 2.86 is a subvariant of Omicron, but according to reports from the CDC, the strain has many more mutations than the ones that came before it.
With so many facts still unknown about this new variant, this news organization asked experts what people need to be aware of as it continues to spread.
What is unique about the BA 2.86 variant?
“It is unique in that it has more than three mutations on the spike protein,” said Purvi S. Parikh, MD, an infectious disease expert at New York University’s Langone Health. The virus uses the spike proteins to enter our cells.
This “may mean it will be more transmissible, cause more severe disease, and/or our vaccines and treatments may not work as well, as compared to other variants,” she said.
What do we need to watch with BA 2.86 going forward?
“We don’t know if this variant will be associated with a change in the disease severity. We currently see increased numbers of cases in general, even though we don’t yet see the BA.2.86 in our system,” said Heba Mostafa, PhD, director of the molecular virology laboratory at Johns Hopkins Hospital in Baltimore.
“It is important to monitor BA.2.86 (and other variants) and understand how its evolution impacts the number of cases and disease outcomes,” she said. “We should all be aware of the current increase in cases, though, and try to get tested and be treated as soon as possible, as antivirals should be effective against the circulating variants.”
What should doctors know?
Dr. Parikh said doctors should generally expect more COVID cases in their clinics and make sure to screen patients even if their symptoms are mild.
“We have tools that can be used – antivirals like Paxlovid are still efficacious with current dominant strains such as EG.5,” she said. “And encourage your patients to get their boosters, mask, wash hands, and social distance.”
How well can our vaccines fight BA 2.86?
“Vaccine coverage for the BA.2.86 is an area of uncertainty right now,” said Dr. Mostafa.
In its report, the CDC said scientists are still figuring out how well the updated COVID vaccine works. It’s expected to be available in the fall, and for now, they believe the new shot will still make infections less severe, new variants and all.
Prior vaccinations and infections have created antibodies in many people, and that will likely provide some protection, Dr. Mostafa said. “When we experienced the Omicron wave in December 2021, even though the variant was distant from what circulated before its emergence and was associated with a very large increase in the number of cases, vaccinations were still protective against severe disease.”
What is the most important thing to keep track of when it comes to this variant?
According to Dr. Parikh, “it’s most important to monitor how transmissible [BA 2.86] is, how severe it is, and if our current treatments and vaccines work.”
Dr. Mostafa said how well the new variants escape existing antibody protection should also be studied and watched closely.
What does this stage of the virus mutation tell us about where we are in the pandemic?
The history of the coronavirus over the past few years shows that variants with many changes evolve and can spread very quickly, Dr. Mostafa said. “Now that the virus is endemic, it is essential to monitor, update vaccinations if necessary, diagnose, treat, and implement infection control measures when necessary.”
With the limited data we have so far, experts seem to agree that while the variant’s makeup raises some red flags, it is too soon to jump to any conclusions about how easy it is to catch it and the ways it may change how the virus impacts those who contract it.
A version of this article first appeared on WebMD.com.
Unlocking the secrets of brown fat
Brown fat, or thermogenic adipose tissue, appears to act as a “nutrient sink,” consuming glucose and lactate, among other metabolites, say U.S. researchers in a mouse study that supports its potential role in tackling obesity and even cancer.
The research, published recently in Nature Metabolism, was led by David A. Guertin, PhD, of the program in molecular medicine, University of Massachusetts, Worcester.
What is adaptive thermogenesis, and why is it important in temperature regulation?
Adaptive thermogenesis is a physiologic process that occurs in a special type of fat cell, called a brown adipocyte, in which intracellular stored lipids and nutrients taken up from the blood are catabolized to generate heat.
The heat generated by these thermogenic adipocytes is critical for warming the blood and maintaining body temperature in cold environments, and is especially critical in human infants and small mammals, which are more sensitive to low temperatures.
The process is stimulated by the sympathetic nervous system, especially in response to feeling cold, but it can be activated by other stresses as well.
While adaptative thermogenesis is also called nonshivering thermogenesis to distinguish it from muscle shivering, both means of generating heat can work together to maintain body temperature.
Why is it considered a potential target for obesity?
Adult humans have brown adipocytes in specific locations in the body called brown adipose tissues (BAT) or, more simply, “brown fat.”
Intriguingly, clinical data show that the more BAT you have, the more likely you are to be protected against cardiometabolic disorders associated with obesity.
Since obesity results from an imbalance between energy intake and energy expenditure, one model proposes that brown adipocytes rebalance this formula by expending the excess energy (calories) as heat rather than storing it.
This has been referred to as the “nutrient sink” model, and the ability to activate this process therapeutically is a very attractive antiobesity strategy.
Why was it important to understand which circulating metabolites BAT uses for thermogenesis?
It is still not clear why brown fat is so beneficial for human health, and thus there is strong rationale for understanding its metabolism and how it cooperates with other tissues in the body.
For example, prior to our work, the field lacked a broad quantitative picture of how much any individual nutrient from the blood was used by brown fat, or which specific nutrients brown fat prefers to use to make heat – such as lipids, glucose, amino acids, etc. Knowing this information helps us identify more precise strategies to activate brown fat.
In addition, circulating metabolites sometimes also have messenger functions, similar to those of hormones, that stimulate physiologic processes such as adaptative thermogenesis. Highly metabolic tissues also put metabolites back into the blood, which can send messages to the brain and other tissues.
We don’t have a lot of information yet on how brown fat might engage in these processes, and so our study also aimed at finding these special metabolite messengers.
You found that glucose and lactate predominate as BAT fuel sources. What does that tell us?
The major fuels used by brown fat have been debated for a long time.
Our study suggests that BAT in mice mainly prefers glucose and lactate, which is generated from glucose. On one hand, this shows us that thermogenic adipocytes may be especially useful in treating hyperglycemia, or even tumors, by reducing the amount of circulating glucose.
It also tells us that we need to focus more on why brown fat needs so much glucose. Other studies suggest that glucose is not just used as a fuel to generate heat but also may have other important functions in keeping brown adipocytes active and healthy.
We need to know that information so that therapeutic strategies targeting brown adipocytes can be optimized to have the best chance of success.
It’s worth noting that we did our study in mice that had free access to food. If the mice were fasting, they would use more lipids from the blood to supplement for the lack of available glucose, but we think that a baseline amount of glucose is still necessary.
What could be the clinical implications of your results if replicated in humans?
They suggest that glucose is an important resource that thermogenic adipocytes cannot do without, and moreover, that glucose is more than just a carbon source.
Resolving those other functions of glucose may provide insight into mechanisms to stimulate these cells or help explain why overweight or obese people who are insulin resistant have less brown fat activity, as insulin stimulates glucose uptake.
Beyond glucose, if any of these other metabolites made or released by brown fat have beneficial messenger functions, there may be ways to pharmacologically mimic them.
How easily do you think your findings could be applied to humans?
On a fundamental level, the basic cellular mechanisms that drive adaptative thermogenesis are likely the same between mice and humans, but the wiring to the sympathetic nervous system is a bit different.
This is why it’s important to look deeply at brown fat metabolism in mouse models to find pathways fundamental to the basic mechanisms of adaptative thermogenesis in both mice and humans, which could reveal unique therapeutic opportunities.
Another big challenge with comparing humans and mice is that humans typically keep their environment warm, so their brown fat is not that active.
In contrast, mice are often raised their entire lives in a facility kept at room temperature, around 22° C (72° F). While comfortable for the humans working with them, it’s cold for a small mouse, and so mice live with constantly active brown fat.
We can change the mouse environment to alter mouse brown fat activity, but that can’t be done with people. This makes comparative studies difficult.
Nevertheless, studies have shown that people who live in cold climates often have more brown fat, and, conversely, mice raised in warmer environments have brown fat that looks a lot more like human brown fat.
What further research do you have planned, or are looking forward to, in this area?
This is the most fun part of what we do, and I’ve been fortunate to have an amazing team passionately working on these questions.
One is to figure out why glucose is so important for these fascinating cells, which will keep us busy for years. We also need to modify the dietary conditions to determine whether the body prioritizes the use of glucose for adaptive thermogenesis even when there isn’t much available.
Another goal is to test whether any of the other metabolites we identified have bioactive functions. We also discovered a unique role for glutamine metabolism in brown fat, through the consumption of amino acids, that we haven’t yet resolved.
Finally, we want to understand how and why brown fat protects other organs from metabolic diseases, and we are just at the tip of the iceberg here.
The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases; the National Institute on Alcohol Abuse and Alcoholism; the National Heart, Lung, & Blood Institute; the National Institutes of Health; the AASLD Foundation Pinnacle Research Award in Liver Disease; the Edward Mallinckrodt Jr. Foundation Award; and the Basic Science Research Program of the Ministry of Education (South Korea). No relevant financial relationships were disclosed.
A version of this article first appeared on Medscape.com.
Brown fat, or thermogenic adipose tissue, appears to act as a “nutrient sink,” consuming glucose and lactate, among other metabolites, say U.S. researchers in a mouse study that supports its potential role in tackling obesity and even cancer.
The research, published recently in Nature Metabolism, was led by David A. Guertin, PhD, of the program in molecular medicine, University of Massachusetts, Worcester.
What is adaptive thermogenesis, and why is it important in temperature regulation?
Adaptive thermogenesis is a physiologic process that occurs in a special type of fat cell, called a brown adipocyte, in which intracellular stored lipids and nutrients taken up from the blood are catabolized to generate heat.
The heat generated by these thermogenic adipocytes is critical for warming the blood and maintaining body temperature in cold environments, and is especially critical in human infants and small mammals, which are more sensitive to low temperatures.
The process is stimulated by the sympathetic nervous system, especially in response to feeling cold, but it can be activated by other stresses as well.
While adaptative thermogenesis is also called nonshivering thermogenesis to distinguish it from muscle shivering, both means of generating heat can work together to maintain body temperature.
Why is it considered a potential target for obesity?
Adult humans have brown adipocytes in specific locations in the body called brown adipose tissues (BAT) or, more simply, “brown fat.”
Intriguingly, clinical data show that the more BAT you have, the more likely you are to be protected against cardiometabolic disorders associated with obesity.
Since obesity results from an imbalance between energy intake and energy expenditure, one model proposes that brown adipocytes rebalance this formula by expending the excess energy (calories) as heat rather than storing it.
This has been referred to as the “nutrient sink” model, and the ability to activate this process therapeutically is a very attractive antiobesity strategy.
Why was it important to understand which circulating metabolites BAT uses for thermogenesis?
It is still not clear why brown fat is so beneficial for human health, and thus there is strong rationale for understanding its metabolism and how it cooperates with other tissues in the body.
For example, prior to our work, the field lacked a broad quantitative picture of how much any individual nutrient from the blood was used by brown fat, or which specific nutrients brown fat prefers to use to make heat – such as lipids, glucose, amino acids, etc. Knowing this information helps us identify more precise strategies to activate brown fat.
In addition, circulating metabolites sometimes also have messenger functions, similar to those of hormones, that stimulate physiologic processes such as adaptative thermogenesis. Highly metabolic tissues also put metabolites back into the blood, which can send messages to the brain and other tissues.
We don’t have a lot of information yet on how brown fat might engage in these processes, and so our study also aimed at finding these special metabolite messengers.
You found that glucose and lactate predominate as BAT fuel sources. What does that tell us?
The major fuels used by brown fat have been debated for a long time.
Our study suggests that BAT in mice mainly prefers glucose and lactate, which is generated from glucose. On one hand, this shows us that thermogenic adipocytes may be especially useful in treating hyperglycemia, or even tumors, by reducing the amount of circulating glucose.
It also tells us that we need to focus more on why brown fat needs so much glucose. Other studies suggest that glucose is not just used as a fuel to generate heat but also may have other important functions in keeping brown adipocytes active and healthy.
We need to know that information so that therapeutic strategies targeting brown adipocytes can be optimized to have the best chance of success.
It’s worth noting that we did our study in mice that had free access to food. If the mice were fasting, they would use more lipids from the blood to supplement for the lack of available glucose, but we think that a baseline amount of glucose is still necessary.
What could be the clinical implications of your results if replicated in humans?
They suggest that glucose is an important resource that thermogenic adipocytes cannot do without, and moreover, that glucose is more than just a carbon source.
Resolving those other functions of glucose may provide insight into mechanisms to stimulate these cells or help explain why overweight or obese people who are insulin resistant have less brown fat activity, as insulin stimulates glucose uptake.
Beyond glucose, if any of these other metabolites made or released by brown fat have beneficial messenger functions, there may be ways to pharmacologically mimic them.
How easily do you think your findings could be applied to humans?
On a fundamental level, the basic cellular mechanisms that drive adaptative thermogenesis are likely the same between mice and humans, but the wiring to the sympathetic nervous system is a bit different.
This is why it’s important to look deeply at brown fat metabolism in mouse models to find pathways fundamental to the basic mechanisms of adaptative thermogenesis in both mice and humans, which could reveal unique therapeutic opportunities.
Another big challenge with comparing humans and mice is that humans typically keep their environment warm, so their brown fat is not that active.
In contrast, mice are often raised their entire lives in a facility kept at room temperature, around 22° C (72° F). While comfortable for the humans working with them, it’s cold for a small mouse, and so mice live with constantly active brown fat.
We can change the mouse environment to alter mouse brown fat activity, but that can’t be done with people. This makes comparative studies difficult.
Nevertheless, studies have shown that people who live in cold climates often have more brown fat, and, conversely, mice raised in warmer environments have brown fat that looks a lot more like human brown fat.
What further research do you have planned, or are looking forward to, in this area?
This is the most fun part of what we do, and I’ve been fortunate to have an amazing team passionately working on these questions.
One is to figure out why glucose is so important for these fascinating cells, which will keep us busy for years. We also need to modify the dietary conditions to determine whether the body prioritizes the use of glucose for adaptive thermogenesis even when there isn’t much available.
Another goal is to test whether any of the other metabolites we identified have bioactive functions. We also discovered a unique role for glutamine metabolism in brown fat, through the consumption of amino acids, that we haven’t yet resolved.
Finally, we want to understand how and why brown fat protects other organs from metabolic diseases, and we are just at the tip of the iceberg here.
The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases; the National Institute on Alcohol Abuse and Alcoholism; the National Heart, Lung, & Blood Institute; the National Institutes of Health; the AASLD Foundation Pinnacle Research Award in Liver Disease; the Edward Mallinckrodt Jr. Foundation Award; and the Basic Science Research Program of the Ministry of Education (South Korea). No relevant financial relationships were disclosed.
A version of this article first appeared on Medscape.com.
Brown fat, or thermogenic adipose tissue, appears to act as a “nutrient sink,” consuming glucose and lactate, among other metabolites, say U.S. researchers in a mouse study that supports its potential role in tackling obesity and even cancer.
The research, published recently in Nature Metabolism, was led by David A. Guertin, PhD, of the program in molecular medicine, University of Massachusetts, Worcester.
What is adaptive thermogenesis, and why is it important in temperature regulation?
Adaptive thermogenesis is a physiologic process that occurs in a special type of fat cell, called a brown adipocyte, in which intracellular stored lipids and nutrients taken up from the blood are catabolized to generate heat.
The heat generated by these thermogenic adipocytes is critical for warming the blood and maintaining body temperature in cold environments, and is especially critical in human infants and small mammals, which are more sensitive to low temperatures.
The process is stimulated by the sympathetic nervous system, especially in response to feeling cold, but it can be activated by other stresses as well.
While adaptative thermogenesis is also called nonshivering thermogenesis to distinguish it from muscle shivering, both means of generating heat can work together to maintain body temperature.
Why is it considered a potential target for obesity?
Adult humans have brown adipocytes in specific locations in the body called brown adipose tissues (BAT) or, more simply, “brown fat.”
Intriguingly, clinical data show that the more BAT you have, the more likely you are to be protected against cardiometabolic disorders associated with obesity.
Since obesity results from an imbalance between energy intake and energy expenditure, one model proposes that brown adipocytes rebalance this formula by expending the excess energy (calories) as heat rather than storing it.
This has been referred to as the “nutrient sink” model, and the ability to activate this process therapeutically is a very attractive antiobesity strategy.
Why was it important to understand which circulating metabolites BAT uses for thermogenesis?
It is still not clear why brown fat is so beneficial for human health, and thus there is strong rationale for understanding its metabolism and how it cooperates with other tissues in the body.
For example, prior to our work, the field lacked a broad quantitative picture of how much any individual nutrient from the blood was used by brown fat, or which specific nutrients brown fat prefers to use to make heat – such as lipids, glucose, amino acids, etc. Knowing this information helps us identify more precise strategies to activate brown fat.
In addition, circulating metabolites sometimes also have messenger functions, similar to those of hormones, that stimulate physiologic processes such as adaptative thermogenesis. Highly metabolic tissues also put metabolites back into the blood, which can send messages to the brain and other tissues.
We don’t have a lot of information yet on how brown fat might engage in these processes, and so our study also aimed at finding these special metabolite messengers.
You found that glucose and lactate predominate as BAT fuel sources. What does that tell us?
The major fuels used by brown fat have been debated for a long time.
Our study suggests that BAT in mice mainly prefers glucose and lactate, which is generated from glucose. On one hand, this shows us that thermogenic adipocytes may be especially useful in treating hyperglycemia, or even tumors, by reducing the amount of circulating glucose.
It also tells us that we need to focus more on why brown fat needs so much glucose. Other studies suggest that glucose is not just used as a fuel to generate heat but also may have other important functions in keeping brown adipocytes active and healthy.
We need to know that information so that therapeutic strategies targeting brown adipocytes can be optimized to have the best chance of success.
It’s worth noting that we did our study in mice that had free access to food. If the mice were fasting, they would use more lipids from the blood to supplement for the lack of available glucose, but we think that a baseline amount of glucose is still necessary.
What could be the clinical implications of your results if replicated in humans?
They suggest that glucose is an important resource that thermogenic adipocytes cannot do without, and moreover, that glucose is more than just a carbon source.
Resolving those other functions of glucose may provide insight into mechanisms to stimulate these cells or help explain why overweight or obese people who are insulin resistant have less brown fat activity, as insulin stimulates glucose uptake.
Beyond glucose, if any of these other metabolites made or released by brown fat have beneficial messenger functions, there may be ways to pharmacologically mimic them.
How easily do you think your findings could be applied to humans?
On a fundamental level, the basic cellular mechanisms that drive adaptative thermogenesis are likely the same between mice and humans, but the wiring to the sympathetic nervous system is a bit different.
This is why it’s important to look deeply at brown fat metabolism in mouse models to find pathways fundamental to the basic mechanisms of adaptative thermogenesis in both mice and humans, which could reveal unique therapeutic opportunities.
Another big challenge with comparing humans and mice is that humans typically keep their environment warm, so their brown fat is not that active.
In contrast, mice are often raised their entire lives in a facility kept at room temperature, around 22° C (72° F). While comfortable for the humans working with them, it’s cold for a small mouse, and so mice live with constantly active brown fat.
We can change the mouse environment to alter mouse brown fat activity, but that can’t be done with people. This makes comparative studies difficult.
Nevertheless, studies have shown that people who live in cold climates often have more brown fat, and, conversely, mice raised in warmer environments have brown fat that looks a lot more like human brown fat.
What further research do you have planned, or are looking forward to, in this area?
This is the most fun part of what we do, and I’ve been fortunate to have an amazing team passionately working on these questions.
One is to figure out why glucose is so important for these fascinating cells, which will keep us busy for years. We also need to modify the dietary conditions to determine whether the body prioritizes the use of glucose for adaptive thermogenesis even when there isn’t much available.
Another goal is to test whether any of the other metabolites we identified have bioactive functions. We also discovered a unique role for glutamine metabolism in brown fat, through the consumption of amino acids, that we haven’t yet resolved.
Finally, we want to understand how and why brown fat protects other organs from metabolic diseases, and we are just at the tip of the iceberg here.
The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases; the National Institute on Alcohol Abuse and Alcoholism; the National Heart, Lung, & Blood Institute; the National Institutes of Health; the AASLD Foundation Pinnacle Research Award in Liver Disease; the Edward Mallinckrodt Jr. Foundation Award; and the Basic Science Research Program of the Ministry of Education (South Korea). No relevant financial relationships were disclosed.
A version of this article first appeared on Medscape.com.
FROM NATURE METABOLISM
Experts debate low-carb diets for people with diabetes
It’s an ongoing debate in the diabetes world: Is it ideal to consume a very-low-carbohydrate diet, or is it better to go with moderate amounts of healthful carbs?
At the annual scientific sessions of the American Diabetes Association, Carol F. Kirkpatrick, PhD, RDN, spoke first, arguing in favor of diets consisting of moderate, high-quality carbohydrates.
Dina Hafez Griauzde, MD, countered that very-low-carbohydrate diets are more beneficial for people with diabetes, primarily type 2 diabetes.
Both speakers based their arguments on published evidence but agreed in the end that discussion with patients about individual dietary preferences should play a major role in the ultimate decision.
Moderate-carbohydrate eating is best
Dr. Kirkpatrick began by explaining that definitions of “low carb” vary in the literature, which makes comparisons between studies difficult. On the basis of a 2019 review that she coauthored, “moderate” carbohydrate consumption was defined as a diet in which 26%-44% of total daily calories are from carbohydrates. “Low” carbohydrate consumption was defined as a diet in which 10%-25% of calories were from carbohydrates. Consuming less than 10% was defined as a very-low-carbohydrate diet (i.e., a ketogenic diet).
Across studies, she noted, the literature shows that within the first 6 months weight loss is typically greater with carbohydrate-restricted diets than with higher-carbohydrate diets, but that by 1 year and beyond weight loss is similar.
“That can be partly due to the difficulty in people maintaining that very severe dietary restriction, although ... we can all acknowledge that it’s difficult for patients to adhere to any dietary pattern, so for sure by 12 months, the difference in the weight loss is gone between the two,” said Dr. Kirkpatrick, of Midwest Biomedical Research, Pocatello, Idaho.
In a recent meta-analysis of 35 trials that examined the dose-dependent effects of carbohydrate restriction for patients with type 2 diabetes, there was a significant decrease in weight as carbohydrates were reduced. But by 12 months (17 trials), the greatest weight reduction was seen at 35% carbohydrate intake.
“It may just be that people were able to adhere to that moderate intake better,” she explained.
Regarding lipids, in her 2019 review and in several meta-analyses since, the effects on low-density lipoprotein cholesterol (LDL-C) varied. For some patients, adhering to a low-carb diet led to reductions in LDL-C, especially if the participants also lost weight, whereas in other patients, a low-carb diet led to an increase in LDL-C.
Either way, a high intake of saturated fatty acids is key to an increase in LDL-C, Dr. Kirkpatrick noted. “So, it’s important that, if a patient chooses to follow a very low carbohydrate diet or any kind of dietary pattern that restricts carbohydrate, that they replace the carbohydrate with unsaturated fat and not saturated fatty acid foods to avoid that increase in LDL-C.”
Generally, the evidence also shows that carbohydrate restriction typically leads to lower triglyceride levels and higher high-density lipoprotein (HDL) cholesterol levels. However, the same meta-analysis showed that the greatest reduction in LDL-C occurred at about 40% carbohydrate consumption.
Another recent meta-analysis showed that LDL-C rose significantly by an average 12.4 mg/dL with very-low-carb (3%-30%) diets, but only slightly, by 0.4 mg/dL, with moderate carb (40%-45%) intake.
Consuming very-low-carb diets did lead to greater reductions in triglycerides, compared with consuming moderate carb diets (23.9 mg/dL vs. 8.9 mg/dL).
“However, in terms of cardiovascular health, we are not entirely sure what that means. ... We have to look at the overall results in the presence of both triglyceride lowering as well as LDL cholesterol,” Dr. Kirkpatrick noted.
Carbohydrate restriction did consistently lead to lower hemoglobin A1c levels by an average of 0.4, 0.6, and 1.0 percentage points at 6 months for diets of 40%, 30%, and 15% carbohydrate, respectively. However, by 12 months, the effect had waned to 0.15, 0.2, and 0.4 A1c percentage points.
“Again, carbohydrate restriction, especially severe, is difficult for people to adhere to, and moderate carbohydrate intake would allow our patients to consume an appropriate amount of carbohydrate and still achieve improved glycemic control,” Dr. Kirkpatrick said.
Two large randomized controlled trials – PREDIMED and CORDIOPREV – examined the effects of the Mediterranean diet on cardiovascular disease prevention. Both showed a decrease in cardiovascular events with the Mediterranean diet, which involves consuming moderate amounts of carbohydrates.
“The Mediterranean dietary pattern has the strongest evidence for benefit, and it’s moderate in carbohydrates,” she concluded.
Very-low-carbohydrate eating is best
Dr. Griauzde was a last-minute replacement speaker for William S. Yancy Jr, MD, of Duke University, Durham, N.C., and presented his slides. She argued that consuming a very low carb diet improves glycemia and that it does not increase but possibly lowers cardiovascular risk.
She began by noting that prior to the discovery of insulin very-low-carb diets had been consumed for over a century to prolong life for people with type 1 diabetes.
“We have long recognized the deleterious role of carbohydrate in type 1 diabetes management, and we have increasingly recognized that role in the management of type 2 diabetes,” said Dr. Griauzde of the University of Michigan in Ann Arbor.
In a small study that compared maintaining a very-low-carb diet for 2 weeks with maintaining a high-carb diet for 2 weeks, total glucose areas under the curve were substantially lower (P < .05) during the low-carb phase, while A1c levels dropped from 7.3% to 6.8% (P = .006).
“We don’t see those outcomes with meds,” Dr. Griauzde noted, adding, “A diet very low in carbohydrates is one of the most potent tools we have to help our patients achieve glycemic control.”
Dr. Griauzde said that the carbohydrate-insulin model provides an explanation for why dietary carbohydrates are particularly obesogenic and metabolically harmful. That model contrasts with the energy balance model, which suggests that all calories are equal.
The rationale of the carbohydrate-insulin model is that dietary carbohydrate – either sugar or starch – raises serum glucose and insulin levels. A carbohydrate-restricted diet therefore reduces the dietary contribution to serum glucose, which then results in lower insulin levels. Insulin is a potent stimulator of lipogenesis (fat storage), and it is a potent inhibitor of lipolysis (the burning of fat). By lowering insulin levels, stored body fat is burned, serum ketone levels increase, and body weight is lowered.
This model suggests that, when insulin levels are chronically high because of excess carbohydrate consumption, circulating fuels are lowered, which leads to an increase in hunger and to overeating. This was demonstrated in a study that compared different levels of isocaloric glycemic index diets in 12 teenage boys with overweight or obesity. The higher-carbohydrate meals led to higher glucose and insulin levels and more food consumption.
In a systematic review of 13 trials of restricted-carbohydrate diets (< 45% carbohydrates) for adults with diabetes, the degree of improvement in A1c level correlated with the degree of carbohydrate restriction over 2-26 weeks (P = .013).
And in a network meta-analysis of 56 trials that compared nine diets among a total of 4,937 participants with type 2 diabetes, one conclusion was that “for reducing A1c, the low-carbohydrate diet was ranked as the best dietary approach (SUCRA: 84%), followed by the Mediterranean diet (80%), and Paleolithic diet (76%), compared with a control diet.”
Regarding the criticism that very-low-carbohydrate diets are high in saturated fat and therefore raise the risk of cardiovascular disease, Dr. Griauzde pointed to another meta-analysis of 21 prospective studies with more than 300,000 participants with 5-25 years of follow-up. In that analysis, the intake of saturated fat was not associated with an increased risk of cardiovascular disease or stroke.
Furthermore, a 12-week randomized controlled trial that involved 40 adults with overweight also suggested that a very-low-carb diet may be superior to a low-fat diet in improving aspects of the metabolic syndrome, including body mass index, lipid levels, and insulin sensitivity. Small LDL particles, which are more atherogenic than larger LDL particles, also decreased despite a threefold increase in saturated fat intake.
Rebuttals: Overall diet, patient preference matter
During the rebuttals, Dr. Kirkpatrick pointed out that large LDL particles are also atherogenic. In addition, she noted that the studies that showed that saturated fat isn’t associated with cardiovascular disease didn’t consider the macronutrients that replaced the saturated fat.
“It really is about increasing consumption of foods that we know are associated with cardiovascular benefit, including plant-based foods that are high quality and not refined carbohydrates ... and healthy protein sources. ... Hopefully we can step away from just looking at macronutrients and look at the total amount of food that people are choosing to eat.”
Importantly, Dr. Kirkpatrick said, patients need to be asked about their current dietary patterns and preferences. “Interventions should be patient centered and sensitive to cultural differences. Personalized lifestyle interventions increase the likelihood of success.”
Dr. Griauzde pointed out that newer antiobesity drugs can be added to any diet to decrease appetite and enhance adherence.
Dr. Griauzde also observed, “We can label a very-low-carbohydrate diet ‘extreme,’ but maybe, from the patient’s perspective, it’s extreme to take 200 units of insulin a day. If you can give them the opportunity to discontinue use of the insulin by following a very-low-carbohydrate dietary pattern, that is the opportunity that our patients deserve to have.”
But overall, she agreed with Dr. Kirkpatrick about individualizing any dietary approach: “We will never know from any of the trials that have been done or that will be done in the future what diet is best for an individual patient. ... Our job is to help our patients find the dietary approach that works best for them.”
Dr. Kirkpatrick is a clinical scientist with Midwest Biomedical Research, which has received funding from various food and pharmaceutical companies. She has not received any direct funding. Dr. Yancy is a consultant for The Simply Good Foods Co. Dr. Griauzde has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
It’s an ongoing debate in the diabetes world: Is it ideal to consume a very-low-carbohydrate diet, or is it better to go with moderate amounts of healthful carbs?
At the annual scientific sessions of the American Diabetes Association, Carol F. Kirkpatrick, PhD, RDN, spoke first, arguing in favor of diets consisting of moderate, high-quality carbohydrates.
Dina Hafez Griauzde, MD, countered that very-low-carbohydrate diets are more beneficial for people with diabetes, primarily type 2 diabetes.
Both speakers based their arguments on published evidence but agreed in the end that discussion with patients about individual dietary preferences should play a major role in the ultimate decision.
Moderate-carbohydrate eating is best
Dr. Kirkpatrick began by explaining that definitions of “low carb” vary in the literature, which makes comparisons between studies difficult. On the basis of a 2019 review that she coauthored, “moderate” carbohydrate consumption was defined as a diet in which 26%-44% of total daily calories are from carbohydrates. “Low” carbohydrate consumption was defined as a diet in which 10%-25% of calories were from carbohydrates. Consuming less than 10% was defined as a very-low-carbohydrate diet (i.e., a ketogenic diet).
Across studies, she noted, the literature shows that within the first 6 months weight loss is typically greater with carbohydrate-restricted diets than with higher-carbohydrate diets, but that by 1 year and beyond weight loss is similar.
“That can be partly due to the difficulty in people maintaining that very severe dietary restriction, although ... we can all acknowledge that it’s difficult for patients to adhere to any dietary pattern, so for sure by 12 months, the difference in the weight loss is gone between the two,” said Dr. Kirkpatrick, of Midwest Biomedical Research, Pocatello, Idaho.
In a recent meta-analysis of 35 trials that examined the dose-dependent effects of carbohydrate restriction for patients with type 2 diabetes, there was a significant decrease in weight as carbohydrates were reduced. But by 12 months (17 trials), the greatest weight reduction was seen at 35% carbohydrate intake.
“It may just be that people were able to adhere to that moderate intake better,” she explained.
Regarding lipids, in her 2019 review and in several meta-analyses since, the effects on low-density lipoprotein cholesterol (LDL-C) varied. For some patients, adhering to a low-carb diet led to reductions in LDL-C, especially if the participants also lost weight, whereas in other patients, a low-carb diet led to an increase in LDL-C.
Either way, a high intake of saturated fatty acids is key to an increase in LDL-C, Dr. Kirkpatrick noted. “So, it’s important that, if a patient chooses to follow a very low carbohydrate diet or any kind of dietary pattern that restricts carbohydrate, that they replace the carbohydrate with unsaturated fat and not saturated fatty acid foods to avoid that increase in LDL-C.”
Generally, the evidence also shows that carbohydrate restriction typically leads to lower triglyceride levels and higher high-density lipoprotein (HDL) cholesterol levels. However, the same meta-analysis showed that the greatest reduction in LDL-C occurred at about 40% carbohydrate consumption.
Another recent meta-analysis showed that LDL-C rose significantly by an average 12.4 mg/dL with very-low-carb (3%-30%) diets, but only slightly, by 0.4 mg/dL, with moderate carb (40%-45%) intake.
Consuming very-low-carb diets did lead to greater reductions in triglycerides, compared with consuming moderate carb diets (23.9 mg/dL vs. 8.9 mg/dL).
“However, in terms of cardiovascular health, we are not entirely sure what that means. ... We have to look at the overall results in the presence of both triglyceride lowering as well as LDL cholesterol,” Dr. Kirkpatrick noted.
Carbohydrate restriction did consistently lead to lower hemoglobin A1c levels by an average of 0.4, 0.6, and 1.0 percentage points at 6 months for diets of 40%, 30%, and 15% carbohydrate, respectively. However, by 12 months, the effect had waned to 0.15, 0.2, and 0.4 A1c percentage points.
“Again, carbohydrate restriction, especially severe, is difficult for people to adhere to, and moderate carbohydrate intake would allow our patients to consume an appropriate amount of carbohydrate and still achieve improved glycemic control,” Dr. Kirkpatrick said.
Two large randomized controlled trials – PREDIMED and CORDIOPREV – examined the effects of the Mediterranean diet on cardiovascular disease prevention. Both showed a decrease in cardiovascular events with the Mediterranean diet, which involves consuming moderate amounts of carbohydrates.
“The Mediterranean dietary pattern has the strongest evidence for benefit, and it’s moderate in carbohydrates,” she concluded.
Very-low-carbohydrate eating is best
Dr. Griauzde was a last-minute replacement speaker for William S. Yancy Jr, MD, of Duke University, Durham, N.C., and presented his slides. She argued that consuming a very low carb diet improves glycemia and that it does not increase but possibly lowers cardiovascular risk.
She began by noting that prior to the discovery of insulin very-low-carb diets had been consumed for over a century to prolong life for people with type 1 diabetes.
“We have long recognized the deleterious role of carbohydrate in type 1 diabetes management, and we have increasingly recognized that role in the management of type 2 diabetes,” said Dr. Griauzde of the University of Michigan in Ann Arbor.
In a small study that compared maintaining a very-low-carb diet for 2 weeks with maintaining a high-carb diet for 2 weeks, total glucose areas under the curve were substantially lower (P < .05) during the low-carb phase, while A1c levels dropped from 7.3% to 6.8% (P = .006).
“We don’t see those outcomes with meds,” Dr. Griauzde noted, adding, “A diet very low in carbohydrates is one of the most potent tools we have to help our patients achieve glycemic control.”
Dr. Griauzde said that the carbohydrate-insulin model provides an explanation for why dietary carbohydrates are particularly obesogenic and metabolically harmful. That model contrasts with the energy balance model, which suggests that all calories are equal.
The rationale of the carbohydrate-insulin model is that dietary carbohydrate – either sugar or starch – raises serum glucose and insulin levels. A carbohydrate-restricted diet therefore reduces the dietary contribution to serum glucose, which then results in lower insulin levels. Insulin is a potent stimulator of lipogenesis (fat storage), and it is a potent inhibitor of lipolysis (the burning of fat). By lowering insulin levels, stored body fat is burned, serum ketone levels increase, and body weight is lowered.
This model suggests that, when insulin levels are chronically high because of excess carbohydrate consumption, circulating fuels are lowered, which leads to an increase in hunger and to overeating. This was demonstrated in a study that compared different levels of isocaloric glycemic index diets in 12 teenage boys with overweight or obesity. The higher-carbohydrate meals led to higher glucose and insulin levels and more food consumption.
In a systematic review of 13 trials of restricted-carbohydrate diets (< 45% carbohydrates) for adults with diabetes, the degree of improvement in A1c level correlated with the degree of carbohydrate restriction over 2-26 weeks (P = .013).
And in a network meta-analysis of 56 trials that compared nine diets among a total of 4,937 participants with type 2 diabetes, one conclusion was that “for reducing A1c, the low-carbohydrate diet was ranked as the best dietary approach (SUCRA: 84%), followed by the Mediterranean diet (80%), and Paleolithic diet (76%), compared with a control diet.”
Regarding the criticism that very-low-carbohydrate diets are high in saturated fat and therefore raise the risk of cardiovascular disease, Dr. Griauzde pointed to another meta-analysis of 21 prospective studies with more than 300,000 participants with 5-25 years of follow-up. In that analysis, the intake of saturated fat was not associated with an increased risk of cardiovascular disease or stroke.
Furthermore, a 12-week randomized controlled trial that involved 40 adults with overweight also suggested that a very-low-carb diet may be superior to a low-fat diet in improving aspects of the metabolic syndrome, including body mass index, lipid levels, and insulin sensitivity. Small LDL particles, which are more atherogenic than larger LDL particles, also decreased despite a threefold increase in saturated fat intake.
Rebuttals: Overall diet, patient preference matter
During the rebuttals, Dr. Kirkpatrick pointed out that large LDL particles are also atherogenic. In addition, she noted that the studies that showed that saturated fat isn’t associated with cardiovascular disease didn’t consider the macronutrients that replaced the saturated fat.
“It really is about increasing consumption of foods that we know are associated with cardiovascular benefit, including plant-based foods that are high quality and not refined carbohydrates ... and healthy protein sources. ... Hopefully we can step away from just looking at macronutrients and look at the total amount of food that people are choosing to eat.”
Importantly, Dr. Kirkpatrick said, patients need to be asked about their current dietary patterns and preferences. “Interventions should be patient centered and sensitive to cultural differences. Personalized lifestyle interventions increase the likelihood of success.”
Dr. Griauzde pointed out that newer antiobesity drugs can be added to any diet to decrease appetite and enhance adherence.
Dr. Griauzde also observed, “We can label a very-low-carbohydrate diet ‘extreme,’ but maybe, from the patient’s perspective, it’s extreme to take 200 units of insulin a day. If you can give them the opportunity to discontinue use of the insulin by following a very-low-carbohydrate dietary pattern, that is the opportunity that our patients deserve to have.”
But overall, she agreed with Dr. Kirkpatrick about individualizing any dietary approach: “We will never know from any of the trials that have been done or that will be done in the future what diet is best for an individual patient. ... Our job is to help our patients find the dietary approach that works best for them.”
Dr. Kirkpatrick is a clinical scientist with Midwest Biomedical Research, which has received funding from various food and pharmaceutical companies. She has not received any direct funding. Dr. Yancy is a consultant for The Simply Good Foods Co. Dr. Griauzde has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
It’s an ongoing debate in the diabetes world: Is it ideal to consume a very-low-carbohydrate diet, or is it better to go with moderate amounts of healthful carbs?
At the annual scientific sessions of the American Diabetes Association, Carol F. Kirkpatrick, PhD, RDN, spoke first, arguing in favor of diets consisting of moderate, high-quality carbohydrates.
Dina Hafez Griauzde, MD, countered that very-low-carbohydrate diets are more beneficial for people with diabetes, primarily type 2 diabetes.
Both speakers based their arguments on published evidence but agreed in the end that discussion with patients about individual dietary preferences should play a major role in the ultimate decision.
Moderate-carbohydrate eating is best
Dr. Kirkpatrick began by explaining that definitions of “low carb” vary in the literature, which makes comparisons between studies difficult. On the basis of a 2019 review that she coauthored, “moderate” carbohydrate consumption was defined as a diet in which 26%-44% of total daily calories are from carbohydrates. “Low” carbohydrate consumption was defined as a diet in which 10%-25% of calories were from carbohydrates. Consuming less than 10% was defined as a very-low-carbohydrate diet (i.e., a ketogenic diet).
Across studies, she noted, the literature shows that within the first 6 months weight loss is typically greater with carbohydrate-restricted diets than with higher-carbohydrate diets, but that by 1 year and beyond weight loss is similar.
“That can be partly due to the difficulty in people maintaining that very severe dietary restriction, although ... we can all acknowledge that it’s difficult for patients to adhere to any dietary pattern, so for sure by 12 months, the difference in the weight loss is gone between the two,” said Dr. Kirkpatrick, of Midwest Biomedical Research, Pocatello, Idaho.
In a recent meta-analysis of 35 trials that examined the dose-dependent effects of carbohydrate restriction for patients with type 2 diabetes, there was a significant decrease in weight as carbohydrates were reduced. But by 12 months (17 trials), the greatest weight reduction was seen at 35% carbohydrate intake.
“It may just be that people were able to adhere to that moderate intake better,” she explained.
Regarding lipids, in her 2019 review and in several meta-analyses since, the effects on low-density lipoprotein cholesterol (LDL-C) varied. For some patients, adhering to a low-carb diet led to reductions in LDL-C, especially if the participants also lost weight, whereas in other patients, a low-carb diet led to an increase in LDL-C.
Either way, a high intake of saturated fatty acids is key to an increase in LDL-C, Dr. Kirkpatrick noted. “So, it’s important that, if a patient chooses to follow a very low carbohydrate diet or any kind of dietary pattern that restricts carbohydrate, that they replace the carbohydrate with unsaturated fat and not saturated fatty acid foods to avoid that increase in LDL-C.”
Generally, the evidence also shows that carbohydrate restriction typically leads to lower triglyceride levels and higher high-density lipoprotein (HDL) cholesterol levels. However, the same meta-analysis showed that the greatest reduction in LDL-C occurred at about 40% carbohydrate consumption.
Another recent meta-analysis showed that LDL-C rose significantly by an average 12.4 mg/dL with very-low-carb (3%-30%) diets, but only slightly, by 0.4 mg/dL, with moderate carb (40%-45%) intake.
Consuming very-low-carb diets did lead to greater reductions in triglycerides, compared with consuming moderate carb diets (23.9 mg/dL vs. 8.9 mg/dL).
“However, in terms of cardiovascular health, we are not entirely sure what that means. ... We have to look at the overall results in the presence of both triglyceride lowering as well as LDL cholesterol,” Dr. Kirkpatrick noted.
Carbohydrate restriction did consistently lead to lower hemoglobin A1c levels by an average of 0.4, 0.6, and 1.0 percentage points at 6 months for diets of 40%, 30%, and 15% carbohydrate, respectively. However, by 12 months, the effect had waned to 0.15, 0.2, and 0.4 A1c percentage points.
“Again, carbohydrate restriction, especially severe, is difficult for people to adhere to, and moderate carbohydrate intake would allow our patients to consume an appropriate amount of carbohydrate and still achieve improved glycemic control,” Dr. Kirkpatrick said.
Two large randomized controlled trials – PREDIMED and CORDIOPREV – examined the effects of the Mediterranean diet on cardiovascular disease prevention. Both showed a decrease in cardiovascular events with the Mediterranean diet, which involves consuming moderate amounts of carbohydrates.
“The Mediterranean dietary pattern has the strongest evidence for benefit, and it’s moderate in carbohydrates,” she concluded.
Very-low-carbohydrate eating is best
Dr. Griauzde was a last-minute replacement speaker for William S. Yancy Jr, MD, of Duke University, Durham, N.C., and presented his slides. She argued that consuming a very low carb diet improves glycemia and that it does not increase but possibly lowers cardiovascular risk.
She began by noting that prior to the discovery of insulin very-low-carb diets had been consumed for over a century to prolong life for people with type 1 diabetes.
“We have long recognized the deleterious role of carbohydrate in type 1 diabetes management, and we have increasingly recognized that role in the management of type 2 diabetes,” said Dr. Griauzde of the University of Michigan in Ann Arbor.
In a small study that compared maintaining a very-low-carb diet for 2 weeks with maintaining a high-carb diet for 2 weeks, total glucose areas under the curve were substantially lower (P < .05) during the low-carb phase, while A1c levels dropped from 7.3% to 6.8% (P = .006).
“We don’t see those outcomes with meds,” Dr. Griauzde noted, adding, “A diet very low in carbohydrates is one of the most potent tools we have to help our patients achieve glycemic control.”
Dr. Griauzde said that the carbohydrate-insulin model provides an explanation for why dietary carbohydrates are particularly obesogenic and metabolically harmful. That model contrasts with the energy balance model, which suggests that all calories are equal.
The rationale of the carbohydrate-insulin model is that dietary carbohydrate – either sugar or starch – raises serum glucose and insulin levels. A carbohydrate-restricted diet therefore reduces the dietary contribution to serum glucose, which then results in lower insulin levels. Insulin is a potent stimulator of lipogenesis (fat storage), and it is a potent inhibitor of lipolysis (the burning of fat). By lowering insulin levels, stored body fat is burned, serum ketone levels increase, and body weight is lowered.
This model suggests that, when insulin levels are chronically high because of excess carbohydrate consumption, circulating fuels are lowered, which leads to an increase in hunger and to overeating. This was demonstrated in a study that compared different levels of isocaloric glycemic index diets in 12 teenage boys with overweight or obesity. The higher-carbohydrate meals led to higher glucose and insulin levels and more food consumption.
In a systematic review of 13 trials of restricted-carbohydrate diets (< 45% carbohydrates) for adults with diabetes, the degree of improvement in A1c level correlated with the degree of carbohydrate restriction over 2-26 weeks (P = .013).
And in a network meta-analysis of 56 trials that compared nine diets among a total of 4,937 participants with type 2 diabetes, one conclusion was that “for reducing A1c, the low-carbohydrate diet was ranked as the best dietary approach (SUCRA: 84%), followed by the Mediterranean diet (80%), and Paleolithic diet (76%), compared with a control diet.”
Regarding the criticism that very-low-carbohydrate diets are high in saturated fat and therefore raise the risk of cardiovascular disease, Dr. Griauzde pointed to another meta-analysis of 21 prospective studies with more than 300,000 participants with 5-25 years of follow-up. In that analysis, the intake of saturated fat was not associated with an increased risk of cardiovascular disease or stroke.
Furthermore, a 12-week randomized controlled trial that involved 40 adults with overweight also suggested that a very-low-carb diet may be superior to a low-fat diet in improving aspects of the metabolic syndrome, including body mass index, lipid levels, and insulin sensitivity. Small LDL particles, which are more atherogenic than larger LDL particles, also decreased despite a threefold increase in saturated fat intake.
Rebuttals: Overall diet, patient preference matter
During the rebuttals, Dr. Kirkpatrick pointed out that large LDL particles are also atherogenic. In addition, she noted that the studies that showed that saturated fat isn’t associated with cardiovascular disease didn’t consider the macronutrients that replaced the saturated fat.
“It really is about increasing consumption of foods that we know are associated with cardiovascular benefit, including plant-based foods that are high quality and not refined carbohydrates ... and healthy protein sources. ... Hopefully we can step away from just looking at macronutrients and look at the total amount of food that people are choosing to eat.”
Importantly, Dr. Kirkpatrick said, patients need to be asked about their current dietary patterns and preferences. “Interventions should be patient centered and sensitive to cultural differences. Personalized lifestyle interventions increase the likelihood of success.”
Dr. Griauzde pointed out that newer antiobesity drugs can be added to any diet to decrease appetite and enhance adherence.
Dr. Griauzde also observed, “We can label a very-low-carbohydrate diet ‘extreme,’ but maybe, from the patient’s perspective, it’s extreme to take 200 units of insulin a day. If you can give them the opportunity to discontinue use of the insulin by following a very-low-carbohydrate dietary pattern, that is the opportunity that our patients deserve to have.”
But overall, she agreed with Dr. Kirkpatrick about individualizing any dietary approach: “We will never know from any of the trials that have been done or that will be done in the future what diet is best for an individual patient. ... Our job is to help our patients find the dietary approach that works best for them.”
Dr. Kirkpatrick is a clinical scientist with Midwest Biomedical Research, which has received funding from various food and pharmaceutical companies. She has not received any direct funding. Dr. Yancy is a consultant for The Simply Good Foods Co. Dr. Griauzde has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ADA 2023
Weight loss linked to mortality risk in older women
Weight loss of at least 5% over a 3-year period was associated with significantly increased mortality in women at age 90, 95, and 100 years compared with those whose weight remained stable, based on data from more than 50,000 individuals.
Previous studies of later-life weight changes and mortality have yielded inconsistent results driven by considerations of weight loss intentionality, and data on older adults in particular are limited, wrote Aladdin H. Shadyab, PhD, of the University of California, San Diego, and colleagues.
In a study published in the Journals of Gerontology: Medical Sciences, the researchers reviewed data from the Women’s Health Initiative, a prospective study of factors affecting chronic disease development in postmenopausal women. The study population included 54,437 women who entered the WHI between 1993 and 1998 at ages 50-79 years. The mean baseline age was 69.8 years; 89.5% of the participants were White, 5.7% were Black, 2.7% were Asian, 2.5% were Hispanic/Latino, and the remaining 1.0% were multiracial, American Indian/Alaskan Native, Native Hawaiian/Other Pacific Islander, or unknown.
The primary outcomes were the associations of short-term (3-year) and long-term (10-year) weight changes with survival to ages 90, 95, and 100 years.
A total of 30,647 women survived to at least 90 years (56.3%).
Overall, women with a short-term weight loss of 5% or more of body weight were 33% less likely to survive to age 90 years, 35% less likely to survive to age 95 years, and 38% less likely to survive to age 100 years than were those whose weight remained stable (odds ratios, 0.67, 0.65, and 0.62, respectively).
The associations were stronger in cases of unintentional short-term weight loss. Intentional weight loss from baseline to year 3 was associated with 17% lower odds of survival to age 90 compared to stable weight (OR, 0.83), but unintentional weight loss was associated with 51% lower odds of survival to age 90 (OR, 0.49).
Similarly, women with 10-year weight loss of at least 5% were 40% less likely to survive to 90 years and 49% less likely to survive to 95 years (OR, 0.60 and OR, 0.51, respectively). The sample size was too small to assess the relation of 10-year weight loss with survival to 100 years, and intentionality was not assessed for 10-year weight changes.
By contrast, weight gain of at least 5% had no significant effect on survival to ages 90, 95, or 100 years, but stable weight over time increased the odds of living to ages 90 to 100 years by 1.2-fold to 2-fold compared to either intentional or unintentional weight loss of at least 5%.
The trends in results were similar across body weight categories (normal weight, overweight, and obese as defined by body mass index). Baseline age and smoking status had no significant effect on the results.
Some of the proportion of self-reported intentional weight loss in the study population may have been unintentional, the researchers wrote in their discussion.
“It is important to note that perceived intentionality of weight loss may be influenced by the many societal pressures to lose weight, especially among women, and therefore overestimate the behavioral changes underlying experienced weight loss in older adults,” they said.
The findings were limited by several factors including the potential for inaccurate self-reported weight loss intention, and the likelihood that the mean older age of the population at baseline (older than 60 years) meant that they were more likely to live longer regardless of weight changes, the researchers noted. Other limitations included the primarily White study population, and other residual confounding factors such as ill health that might drive weight loss, the researchers noted.
However, the results were strengthened by the large sample size and long follow-up period, and suggest that “blanket recommendations for weight loss in older women are unlikely to lead to better survival at advanced ages,” they concluded.
Data support weight monitoring
The investigators acknowledged that their data do not affect clinical recommendations for moderate weight loss in older women to improve health outcomes, especially in those with overweight or obesity, but instead “support close monitoring of the amount and speed of weight loss, particularly when unintentional, as an indicator of underlying poor health and predictor of decreased lifespan in older women.”
Neil Skolnik, MD, professor of family and community medicine at the Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, agreed with this conclusion. The current study suggests that when older women lose a significant amount of weight unintentionally, it could be a sign of failing health, he said.
Weight gain or loss in old age is very different from weight issues in younger people, where clinicians may be encouraging weight loss to improve health outcomes, Dr. Skolnik said in an interview.
A key take-home message for clinicians, in addition to monitoring weight in older patients, is to emphasize nutrition for individuals in their 80s, 90s, and beyond, he said.
The study was supported by the National Heart, Lung, and Blood Institute. Dr. Shadyab had no financial conflicts to disclose. Dr. Skolnik had no financial conflicts to disclose and serves on the editorial advisory board of Family Practice News.
Weight loss of at least 5% over a 3-year period was associated with significantly increased mortality in women at age 90, 95, and 100 years compared with those whose weight remained stable, based on data from more than 50,000 individuals.
Previous studies of later-life weight changes and mortality have yielded inconsistent results driven by considerations of weight loss intentionality, and data on older adults in particular are limited, wrote Aladdin H. Shadyab, PhD, of the University of California, San Diego, and colleagues.
In a study published in the Journals of Gerontology: Medical Sciences, the researchers reviewed data from the Women’s Health Initiative, a prospective study of factors affecting chronic disease development in postmenopausal women. The study population included 54,437 women who entered the WHI between 1993 and 1998 at ages 50-79 years. The mean baseline age was 69.8 years; 89.5% of the participants were White, 5.7% were Black, 2.7% were Asian, 2.5% were Hispanic/Latino, and the remaining 1.0% were multiracial, American Indian/Alaskan Native, Native Hawaiian/Other Pacific Islander, or unknown.
The primary outcomes were the associations of short-term (3-year) and long-term (10-year) weight changes with survival to ages 90, 95, and 100 years.
A total of 30,647 women survived to at least 90 years (56.3%).
Overall, women with a short-term weight loss of 5% or more of body weight were 33% less likely to survive to age 90 years, 35% less likely to survive to age 95 years, and 38% less likely to survive to age 100 years than were those whose weight remained stable (odds ratios, 0.67, 0.65, and 0.62, respectively).
The associations were stronger in cases of unintentional short-term weight loss. Intentional weight loss from baseline to year 3 was associated with 17% lower odds of survival to age 90 compared to stable weight (OR, 0.83), but unintentional weight loss was associated with 51% lower odds of survival to age 90 (OR, 0.49).
Similarly, women with 10-year weight loss of at least 5% were 40% less likely to survive to 90 years and 49% less likely to survive to 95 years (OR, 0.60 and OR, 0.51, respectively). The sample size was too small to assess the relation of 10-year weight loss with survival to 100 years, and intentionality was not assessed for 10-year weight changes.
By contrast, weight gain of at least 5% had no significant effect on survival to ages 90, 95, or 100 years, but stable weight over time increased the odds of living to ages 90 to 100 years by 1.2-fold to 2-fold compared to either intentional or unintentional weight loss of at least 5%.
The trends in results were similar across body weight categories (normal weight, overweight, and obese as defined by body mass index). Baseline age and smoking status had no significant effect on the results.
Some of the proportion of self-reported intentional weight loss in the study population may have been unintentional, the researchers wrote in their discussion.
“It is important to note that perceived intentionality of weight loss may be influenced by the many societal pressures to lose weight, especially among women, and therefore overestimate the behavioral changes underlying experienced weight loss in older adults,” they said.
The findings were limited by several factors including the potential for inaccurate self-reported weight loss intention, and the likelihood that the mean older age of the population at baseline (older than 60 years) meant that they were more likely to live longer regardless of weight changes, the researchers noted. Other limitations included the primarily White study population, and other residual confounding factors such as ill health that might drive weight loss, the researchers noted.
However, the results were strengthened by the large sample size and long follow-up period, and suggest that “blanket recommendations for weight loss in older women are unlikely to lead to better survival at advanced ages,” they concluded.
Data support weight monitoring
The investigators acknowledged that their data do not affect clinical recommendations for moderate weight loss in older women to improve health outcomes, especially in those with overweight or obesity, but instead “support close monitoring of the amount and speed of weight loss, particularly when unintentional, as an indicator of underlying poor health and predictor of decreased lifespan in older women.”
Neil Skolnik, MD, professor of family and community medicine at the Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, agreed with this conclusion. The current study suggests that when older women lose a significant amount of weight unintentionally, it could be a sign of failing health, he said.
Weight gain or loss in old age is very different from weight issues in younger people, where clinicians may be encouraging weight loss to improve health outcomes, Dr. Skolnik said in an interview.
A key take-home message for clinicians, in addition to monitoring weight in older patients, is to emphasize nutrition for individuals in their 80s, 90s, and beyond, he said.
The study was supported by the National Heart, Lung, and Blood Institute. Dr. Shadyab had no financial conflicts to disclose. Dr. Skolnik had no financial conflicts to disclose and serves on the editorial advisory board of Family Practice News.
Weight loss of at least 5% over a 3-year period was associated with significantly increased mortality in women at age 90, 95, and 100 years compared with those whose weight remained stable, based on data from more than 50,000 individuals.
Previous studies of later-life weight changes and mortality have yielded inconsistent results driven by considerations of weight loss intentionality, and data on older adults in particular are limited, wrote Aladdin H. Shadyab, PhD, of the University of California, San Diego, and colleagues.
In a study published in the Journals of Gerontology: Medical Sciences, the researchers reviewed data from the Women’s Health Initiative, a prospective study of factors affecting chronic disease development in postmenopausal women. The study population included 54,437 women who entered the WHI between 1993 and 1998 at ages 50-79 years. The mean baseline age was 69.8 years; 89.5% of the participants were White, 5.7% were Black, 2.7% were Asian, 2.5% were Hispanic/Latino, and the remaining 1.0% were multiracial, American Indian/Alaskan Native, Native Hawaiian/Other Pacific Islander, or unknown.
The primary outcomes were the associations of short-term (3-year) and long-term (10-year) weight changes with survival to ages 90, 95, and 100 years.
A total of 30,647 women survived to at least 90 years (56.3%).
Overall, women with a short-term weight loss of 5% or more of body weight were 33% less likely to survive to age 90 years, 35% less likely to survive to age 95 years, and 38% less likely to survive to age 100 years than were those whose weight remained stable (odds ratios, 0.67, 0.65, and 0.62, respectively).
The associations were stronger in cases of unintentional short-term weight loss. Intentional weight loss from baseline to year 3 was associated with 17% lower odds of survival to age 90 compared to stable weight (OR, 0.83), but unintentional weight loss was associated with 51% lower odds of survival to age 90 (OR, 0.49).
Similarly, women with 10-year weight loss of at least 5% were 40% less likely to survive to 90 years and 49% less likely to survive to 95 years (OR, 0.60 and OR, 0.51, respectively). The sample size was too small to assess the relation of 10-year weight loss with survival to 100 years, and intentionality was not assessed for 10-year weight changes.
By contrast, weight gain of at least 5% had no significant effect on survival to ages 90, 95, or 100 years, but stable weight over time increased the odds of living to ages 90 to 100 years by 1.2-fold to 2-fold compared to either intentional or unintentional weight loss of at least 5%.
The trends in results were similar across body weight categories (normal weight, overweight, and obese as defined by body mass index). Baseline age and smoking status had no significant effect on the results.
Some of the proportion of self-reported intentional weight loss in the study population may have been unintentional, the researchers wrote in their discussion.
“It is important to note that perceived intentionality of weight loss may be influenced by the many societal pressures to lose weight, especially among women, and therefore overestimate the behavioral changes underlying experienced weight loss in older adults,” they said.
The findings were limited by several factors including the potential for inaccurate self-reported weight loss intention, and the likelihood that the mean older age of the population at baseline (older than 60 years) meant that they were more likely to live longer regardless of weight changes, the researchers noted. Other limitations included the primarily White study population, and other residual confounding factors such as ill health that might drive weight loss, the researchers noted.
However, the results were strengthened by the large sample size and long follow-up period, and suggest that “blanket recommendations for weight loss in older women are unlikely to lead to better survival at advanced ages,” they concluded.
Data support weight monitoring
The investigators acknowledged that their data do not affect clinical recommendations for moderate weight loss in older women to improve health outcomes, especially in those with overweight or obesity, but instead “support close monitoring of the amount and speed of weight loss, particularly when unintentional, as an indicator of underlying poor health and predictor of decreased lifespan in older women.”
Neil Skolnik, MD, professor of family and community medicine at the Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, agreed with this conclusion. The current study suggests that when older women lose a significant amount of weight unintentionally, it could be a sign of failing health, he said.
Weight gain or loss in old age is very different from weight issues in younger people, where clinicians may be encouraging weight loss to improve health outcomes, Dr. Skolnik said in an interview.
A key take-home message for clinicians, in addition to monitoring weight in older patients, is to emphasize nutrition for individuals in their 80s, 90s, and beyond, he said.
The study was supported by the National Heart, Lung, and Blood Institute. Dr. Shadyab had no financial conflicts to disclose. Dr. Skolnik had no financial conflicts to disclose and serves on the editorial advisory board of Family Practice News.
FROM THE JOURNALS OF GERONTOLOGY: MEDICAL SCIENCES
Diabetes drug class appears to reduce recurrent gout flares
The glucose-lowering drug class sodium-glucose cotransporter 2 (SGLT2) inhibitors appear to reduce the risk for recurrent gout flares in people with gout and type 2 diabetes, and to lessen excess mortality in those individuals, compared with those who initiated other types of glucose-lowering medications, new data suggest.
Among nearly 6,000 adults with both type 2 diabetes and gout from a U.K. primary care database, initiation of SGLT2 inhibitor treatment was associated with 19% fewer recurrent gout flares and 29% lower mortality.
Moreover, unlike other urate-lowering therapies, there were no apparent transient increases in the risk of gout flares after initiating therapy, Jie Wei, PhD, of Health Management Center, Xiangya Hospital, Central South University, Changsha, China, and colleagues reported in JAMA Network Open.
These results are important because current management of gout is suboptimal. Many patients either don’t receive adequate urate-lowering therapies such as allopurinol or stop taking them, Dr. Wei and colleagues said.
In addition to lowering glucose, SGLT2 inhibitors also reduce the risk for major adverse cardiovascular events and all-cause mortality in people regardless of their diabetes status. Previous studies have also found that SGLT2 inhibitors reduce the risk for developing gout and of gout flares.
Asked to comment, gout specialist John D. FitzGerald, MD, PhD, clinical chief of rheumatology at the University of California, Los Angeles, said in an interview: “I think it’s a well-done paper, with a large dataset. I think it just reinforces the findings from the other papers. Mostly anything that lowers uric acid levels is going to lower recurrent gout attacks, so it all makes sense.”
However, while Dr. FitzGerald thinks the drug class is a good option for people with diabetes or cardiorenal indications for them who also have gout, he doesn’t envision it as first-line for most other patients with gout. “The current treatments are very effective. Allopurinol brings down uric acid levels by 5-7 points. There are patients who fail allopurinol, but those are less than 5%.”
The most common reason patients stop taking allopurinol is the frequent initial gout flare. But that’s preventable, Dr. FitzGerald said, either by titrating up slowly, or by adding colchicine along with it. “By going slowly, you can avoid that flare risk. I think that’s what’s going on with the SGLT2 inhibitor. It’s not a dramatic urate-lowering drug, but it is clinically meaningful. I think that’s what this paper is showing.”
But, he noted, “I think there are so many reasons to start the SGLT2 inhibitors that if somebody also has gout, all the better. And, if somebody is on the margin with diabetes and gout control and can’t go with allopurinol, it would be great to add for both conditions.”
Less gout recurrence, lower mortality
The retrospective study was conducted from Jan. 1, 2013, to March 31, 2022. Among 5,931 patients with both type 2 diabetes and gout, 1,548 (26.1%) initiated an SGLT2 inhibitor (dapagliflozin, empagliflozin, or canagliflozin), while 4,383 (73.9%) initiated treatment with other active comparators, mostly (92.6%) dipeptidyl peptidase–4 inhibitors.
Gout flares were identified in the charts for a total of 86% of the participants. The weighted incidence rates for the first recurrent flare were 32.4 versus 41.2 per 1,000 person-years in the SGLT2 inhibitor versus comparator groups, with a weighted absolute rate difference of –8.8/1,000 and weighted hazard ratio of 0.81, a significant difference.
All-cause mortality was 18.8 versus 24.9 per 1,000 person-years, respectively, giving an HR of 0.71 at 5-year follow-up.
Dr. FitzGerald, who chaired the American College of Rheumatology’s 2020 gout guidelines, said he anticipates that the SGLT2 inhibitors will be mentioned in the next update to the ACR’s now “living” guidelines, although he was not speaking on the organization’s behalf.
“We talk about losartan in the current [ACR guidelines], about its specific uric acid–lowering effect. Drugs can make uric acid worse or better. For example, thiazides make it higher. I think the SGLT2 [inhibitors] are important, but I don’t think they’re huge. The study is great, and I think the drugs are great, but I don’t think they will change the way gout is managed.”
This work was supported by grants from the National Key Research and Development Plan, the National Natural Science Foundation of China, the Project Program of National Clinical Research Center for Geriatric Disorders, and from the Natural Science Foundation of Hunan Province. Dr. Wei reported receiving grant funding from Xiangya Hospital Central South University Project Program of National Clinical Research Center for Geriatric Disorders and the Science and Technology Department of Hunan Province, the Natural Science Foundation of Hunan Province, during the conduct of the study. Dr. FitzGerald reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The glucose-lowering drug class sodium-glucose cotransporter 2 (SGLT2) inhibitors appear to reduce the risk for recurrent gout flares in people with gout and type 2 diabetes, and to lessen excess mortality in those individuals, compared with those who initiated other types of glucose-lowering medications, new data suggest.
Among nearly 6,000 adults with both type 2 diabetes and gout from a U.K. primary care database, initiation of SGLT2 inhibitor treatment was associated with 19% fewer recurrent gout flares and 29% lower mortality.
Moreover, unlike other urate-lowering therapies, there were no apparent transient increases in the risk of gout flares after initiating therapy, Jie Wei, PhD, of Health Management Center, Xiangya Hospital, Central South University, Changsha, China, and colleagues reported in JAMA Network Open.
These results are important because current management of gout is suboptimal. Many patients either don’t receive adequate urate-lowering therapies such as allopurinol or stop taking them, Dr. Wei and colleagues said.
In addition to lowering glucose, SGLT2 inhibitors also reduce the risk for major adverse cardiovascular events and all-cause mortality in people regardless of their diabetes status. Previous studies have also found that SGLT2 inhibitors reduce the risk for developing gout and of gout flares.
Asked to comment, gout specialist John D. FitzGerald, MD, PhD, clinical chief of rheumatology at the University of California, Los Angeles, said in an interview: “I think it’s a well-done paper, with a large dataset. I think it just reinforces the findings from the other papers. Mostly anything that lowers uric acid levels is going to lower recurrent gout attacks, so it all makes sense.”
However, while Dr. FitzGerald thinks the drug class is a good option for people with diabetes or cardiorenal indications for them who also have gout, he doesn’t envision it as first-line for most other patients with gout. “The current treatments are very effective. Allopurinol brings down uric acid levels by 5-7 points. There are patients who fail allopurinol, but those are less than 5%.”
The most common reason patients stop taking allopurinol is the frequent initial gout flare. But that’s preventable, Dr. FitzGerald said, either by titrating up slowly, or by adding colchicine along with it. “By going slowly, you can avoid that flare risk. I think that’s what’s going on with the SGLT2 inhibitor. It’s not a dramatic urate-lowering drug, but it is clinically meaningful. I think that’s what this paper is showing.”
But, he noted, “I think there are so many reasons to start the SGLT2 inhibitors that if somebody also has gout, all the better. And, if somebody is on the margin with diabetes and gout control and can’t go with allopurinol, it would be great to add for both conditions.”
Less gout recurrence, lower mortality
The retrospective study was conducted from Jan. 1, 2013, to March 31, 2022. Among 5,931 patients with both type 2 diabetes and gout, 1,548 (26.1%) initiated an SGLT2 inhibitor (dapagliflozin, empagliflozin, or canagliflozin), while 4,383 (73.9%) initiated treatment with other active comparators, mostly (92.6%) dipeptidyl peptidase–4 inhibitors.
Gout flares were identified in the charts for a total of 86% of the participants. The weighted incidence rates for the first recurrent flare were 32.4 versus 41.2 per 1,000 person-years in the SGLT2 inhibitor versus comparator groups, with a weighted absolute rate difference of –8.8/1,000 and weighted hazard ratio of 0.81, a significant difference.
All-cause mortality was 18.8 versus 24.9 per 1,000 person-years, respectively, giving an HR of 0.71 at 5-year follow-up.
Dr. FitzGerald, who chaired the American College of Rheumatology’s 2020 gout guidelines, said he anticipates that the SGLT2 inhibitors will be mentioned in the next update to the ACR’s now “living” guidelines, although he was not speaking on the organization’s behalf.
“We talk about losartan in the current [ACR guidelines], about its specific uric acid–lowering effect. Drugs can make uric acid worse or better. For example, thiazides make it higher. I think the SGLT2 [inhibitors] are important, but I don’t think they’re huge. The study is great, and I think the drugs are great, but I don’t think they will change the way gout is managed.”
This work was supported by grants from the National Key Research and Development Plan, the National Natural Science Foundation of China, the Project Program of National Clinical Research Center for Geriatric Disorders, and from the Natural Science Foundation of Hunan Province. Dr. Wei reported receiving grant funding from Xiangya Hospital Central South University Project Program of National Clinical Research Center for Geriatric Disorders and the Science and Technology Department of Hunan Province, the Natural Science Foundation of Hunan Province, during the conduct of the study. Dr. FitzGerald reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The glucose-lowering drug class sodium-glucose cotransporter 2 (SGLT2) inhibitors appear to reduce the risk for recurrent gout flares in people with gout and type 2 diabetes, and to lessen excess mortality in those individuals, compared with those who initiated other types of glucose-lowering medications, new data suggest.
Among nearly 6,000 adults with both type 2 diabetes and gout from a U.K. primary care database, initiation of SGLT2 inhibitor treatment was associated with 19% fewer recurrent gout flares and 29% lower mortality.
Moreover, unlike other urate-lowering therapies, there were no apparent transient increases in the risk of gout flares after initiating therapy, Jie Wei, PhD, of Health Management Center, Xiangya Hospital, Central South University, Changsha, China, and colleagues reported in JAMA Network Open.
These results are important because current management of gout is suboptimal. Many patients either don’t receive adequate urate-lowering therapies such as allopurinol or stop taking them, Dr. Wei and colleagues said.
In addition to lowering glucose, SGLT2 inhibitors also reduce the risk for major adverse cardiovascular events and all-cause mortality in people regardless of their diabetes status. Previous studies have also found that SGLT2 inhibitors reduce the risk for developing gout and of gout flares.
Asked to comment, gout specialist John D. FitzGerald, MD, PhD, clinical chief of rheumatology at the University of California, Los Angeles, said in an interview: “I think it’s a well-done paper, with a large dataset. I think it just reinforces the findings from the other papers. Mostly anything that lowers uric acid levels is going to lower recurrent gout attacks, so it all makes sense.”
However, while Dr. FitzGerald thinks the drug class is a good option for people with diabetes or cardiorenal indications for them who also have gout, he doesn’t envision it as first-line for most other patients with gout. “The current treatments are very effective. Allopurinol brings down uric acid levels by 5-7 points. There are patients who fail allopurinol, but those are less than 5%.”
The most common reason patients stop taking allopurinol is the frequent initial gout flare. But that’s preventable, Dr. FitzGerald said, either by titrating up slowly, or by adding colchicine along with it. “By going slowly, you can avoid that flare risk. I think that’s what’s going on with the SGLT2 inhibitor. It’s not a dramatic urate-lowering drug, but it is clinically meaningful. I think that’s what this paper is showing.”
But, he noted, “I think there are so many reasons to start the SGLT2 inhibitors that if somebody also has gout, all the better. And, if somebody is on the margin with diabetes and gout control and can’t go with allopurinol, it would be great to add for both conditions.”
Less gout recurrence, lower mortality
The retrospective study was conducted from Jan. 1, 2013, to March 31, 2022. Among 5,931 patients with both type 2 diabetes and gout, 1,548 (26.1%) initiated an SGLT2 inhibitor (dapagliflozin, empagliflozin, or canagliflozin), while 4,383 (73.9%) initiated treatment with other active comparators, mostly (92.6%) dipeptidyl peptidase–4 inhibitors.
Gout flares were identified in the charts for a total of 86% of the participants. The weighted incidence rates for the first recurrent flare were 32.4 versus 41.2 per 1,000 person-years in the SGLT2 inhibitor versus comparator groups, with a weighted absolute rate difference of –8.8/1,000 and weighted hazard ratio of 0.81, a significant difference.
All-cause mortality was 18.8 versus 24.9 per 1,000 person-years, respectively, giving an HR of 0.71 at 5-year follow-up.
Dr. FitzGerald, who chaired the American College of Rheumatology’s 2020 gout guidelines, said he anticipates that the SGLT2 inhibitors will be mentioned in the next update to the ACR’s now “living” guidelines, although he was not speaking on the organization’s behalf.
“We talk about losartan in the current [ACR guidelines], about its specific uric acid–lowering effect. Drugs can make uric acid worse or better. For example, thiazides make it higher. I think the SGLT2 [inhibitors] are important, but I don’t think they’re huge. The study is great, and I think the drugs are great, but I don’t think they will change the way gout is managed.”
This work was supported by grants from the National Key Research and Development Plan, the National Natural Science Foundation of China, the Project Program of National Clinical Research Center for Geriatric Disorders, and from the Natural Science Foundation of Hunan Province. Dr. Wei reported receiving grant funding from Xiangya Hospital Central South University Project Program of National Clinical Research Center for Geriatric Disorders and the Science and Technology Department of Hunan Province, the Natural Science Foundation of Hunan Province, during the conduct of the study. Dr. FitzGerald reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
NPs, PAs, and physicians hope to join doctors’ union in rare alliance
Advanced practice providers (APPs) such as nurse practitioners (NPs) and physician assistants (PAs) have long been at odds with doctor groups over scope of practice issues. But in a rare alliance, in late September. If successful, the Allina group will join the Doctors Council SEIU, Local 10MD.
The Allina health care providers share concerns about their working conditions, such as understaffing and inadequate resources, limited decision-making authority, and health systems valuing productivity and profit over patient care.
Although doctors and APPs have said that they generally work well together, the relationship has been strained in recent years as APPs argue for greater scope of practice. Meanwhile, physician groups, such as the American Medical Association, believe that APPs need more oversight.
An Allina union organizer, Britta V. Kasmarik, CNP, acknowledges the tension between physicians and APPs. But she said in an interview that the union effort helped bond this group of health care providers. “We share common goals of providing high-quality care for patients in a safe way, and we see the same things day in and day out with our patients.”
Matt Hoffman, MD, a primary care physician at Allina, told this news organization that APPs in his specialty perform the same job as doctors “and the working conditions are really identical. In our view, that means we should be unionizing together.”
The decision to hold a union vote follows similar action by nearly 150 Allina Mercy Hospital physicians in March. Allina Health appealed the vote.
In response to a New York Times investigation, the Minnesota Attorney General’s office began reviewing reports of aggressive billing practices and denied care at Allina Health.
The Allina Health system, which reports $4 billion in annual revenue, cut off nonemergency services to patients, including children, if their medical debt exceeded $4,500, according to the New York Times article. For Allina’s physicians and APPs, that meant leaving patients’ illnesses untreated.
Less than a week after the attorney general announced its investigation, the health system ended this practice.
In a prepared statement to this news organization, Allina Health said that its providers are “critical members of our teams. … We deeply value and share their commitment to providing high-quality care to our patients.”
The health system said it planned to make operational improvements, implement new communication tools, and provide additional well-being resources and enhanced employee benefits “to improve the provider experience.” In addition, it hoped to continue to “foster a culture of collaboration with all our employees.”
Having a union will allow health care providers to advocate for their patients and give health care providers more decision-making power instead of corporate leaders maintaining full authority, Ms. Kasmarik told this news organization.
Union organizers are also concerned with changes to the daily practice of medicine. “We don’t want to be spending our time doing paperwork and calling insurance companies and filling out forms,” said Dr. Hoffman. “We want to be in the exam room with a patient.”
The Allina providers organized after multiple requests to corporate managers failed to address their concerns. Their demands include increased staffing and help with nonclinical work so that clinicians can spend more time with their patients.
“What I’m really excited about is that we will be able to work with the other unionized groups to make change ... by being involved in health care policy at a state or national level,” Dr. Hoffman said. For example, that involvement might include challenging insurance company decisions.
Doctors Council bills itself as the largest union for attending physicians in the country, with 3,500 members, according to Joe Crane, national organizing director.
Despite an increase in union efforts since the pandemic, health care workers – particularly doctors – have been slow to join unions. Mr. Crane estimated that only about 3% of U.S. physicians are currently union members. He cited union campaigns in Massachusetts, New York, and Washington, DC. For comparison, a minority of advanced practice registered nurses (APRNs) (9%) report union membership, according to Medscape’s APRN compensation report last year.
Dr. Hoffman is confident the Allina health care providers will have enough votes to win the election to join the union. “We should have done this years ago.”
A version of this article appeared on Medscape.com.
Advanced practice providers (APPs) such as nurse practitioners (NPs) and physician assistants (PAs) have long been at odds with doctor groups over scope of practice issues. But in a rare alliance, in late September. If successful, the Allina group will join the Doctors Council SEIU, Local 10MD.
The Allina health care providers share concerns about their working conditions, such as understaffing and inadequate resources, limited decision-making authority, and health systems valuing productivity and profit over patient care.
Although doctors and APPs have said that they generally work well together, the relationship has been strained in recent years as APPs argue for greater scope of practice. Meanwhile, physician groups, such as the American Medical Association, believe that APPs need more oversight.
An Allina union organizer, Britta V. Kasmarik, CNP, acknowledges the tension between physicians and APPs. But she said in an interview that the union effort helped bond this group of health care providers. “We share common goals of providing high-quality care for patients in a safe way, and we see the same things day in and day out with our patients.”
Matt Hoffman, MD, a primary care physician at Allina, told this news organization that APPs in his specialty perform the same job as doctors “and the working conditions are really identical. In our view, that means we should be unionizing together.”
The decision to hold a union vote follows similar action by nearly 150 Allina Mercy Hospital physicians in March. Allina Health appealed the vote.
In response to a New York Times investigation, the Minnesota Attorney General’s office began reviewing reports of aggressive billing practices and denied care at Allina Health.
The Allina Health system, which reports $4 billion in annual revenue, cut off nonemergency services to patients, including children, if their medical debt exceeded $4,500, according to the New York Times article. For Allina’s physicians and APPs, that meant leaving patients’ illnesses untreated.
Less than a week after the attorney general announced its investigation, the health system ended this practice.
In a prepared statement to this news organization, Allina Health said that its providers are “critical members of our teams. … We deeply value and share their commitment to providing high-quality care to our patients.”
The health system said it planned to make operational improvements, implement new communication tools, and provide additional well-being resources and enhanced employee benefits “to improve the provider experience.” In addition, it hoped to continue to “foster a culture of collaboration with all our employees.”
Having a union will allow health care providers to advocate for their patients and give health care providers more decision-making power instead of corporate leaders maintaining full authority, Ms. Kasmarik told this news organization.
Union organizers are also concerned with changes to the daily practice of medicine. “We don’t want to be spending our time doing paperwork and calling insurance companies and filling out forms,” said Dr. Hoffman. “We want to be in the exam room with a patient.”
The Allina providers organized after multiple requests to corporate managers failed to address their concerns. Their demands include increased staffing and help with nonclinical work so that clinicians can spend more time with their patients.
“What I’m really excited about is that we will be able to work with the other unionized groups to make change ... by being involved in health care policy at a state or national level,” Dr. Hoffman said. For example, that involvement might include challenging insurance company decisions.
Doctors Council bills itself as the largest union for attending physicians in the country, with 3,500 members, according to Joe Crane, national organizing director.
Despite an increase in union efforts since the pandemic, health care workers – particularly doctors – have been slow to join unions. Mr. Crane estimated that only about 3% of U.S. physicians are currently union members. He cited union campaigns in Massachusetts, New York, and Washington, DC. For comparison, a minority of advanced practice registered nurses (APRNs) (9%) report union membership, according to Medscape’s APRN compensation report last year.
Dr. Hoffman is confident the Allina health care providers will have enough votes to win the election to join the union. “We should have done this years ago.”
A version of this article appeared on Medscape.com.
Advanced practice providers (APPs) such as nurse practitioners (NPs) and physician assistants (PAs) have long been at odds with doctor groups over scope of practice issues. But in a rare alliance, in late September. If successful, the Allina group will join the Doctors Council SEIU, Local 10MD.
The Allina health care providers share concerns about their working conditions, such as understaffing and inadequate resources, limited decision-making authority, and health systems valuing productivity and profit over patient care.
Although doctors and APPs have said that they generally work well together, the relationship has been strained in recent years as APPs argue for greater scope of practice. Meanwhile, physician groups, such as the American Medical Association, believe that APPs need more oversight.
An Allina union organizer, Britta V. Kasmarik, CNP, acknowledges the tension between physicians and APPs. But she said in an interview that the union effort helped bond this group of health care providers. “We share common goals of providing high-quality care for patients in a safe way, and we see the same things day in and day out with our patients.”
Matt Hoffman, MD, a primary care physician at Allina, told this news organization that APPs in his specialty perform the same job as doctors “and the working conditions are really identical. In our view, that means we should be unionizing together.”
The decision to hold a union vote follows similar action by nearly 150 Allina Mercy Hospital physicians in March. Allina Health appealed the vote.
In response to a New York Times investigation, the Minnesota Attorney General’s office began reviewing reports of aggressive billing practices and denied care at Allina Health.
The Allina Health system, which reports $4 billion in annual revenue, cut off nonemergency services to patients, including children, if their medical debt exceeded $4,500, according to the New York Times article. For Allina’s physicians and APPs, that meant leaving patients’ illnesses untreated.
Less than a week after the attorney general announced its investigation, the health system ended this practice.
In a prepared statement to this news organization, Allina Health said that its providers are “critical members of our teams. … We deeply value and share their commitment to providing high-quality care to our patients.”
The health system said it planned to make operational improvements, implement new communication tools, and provide additional well-being resources and enhanced employee benefits “to improve the provider experience.” In addition, it hoped to continue to “foster a culture of collaboration with all our employees.”
Having a union will allow health care providers to advocate for their patients and give health care providers more decision-making power instead of corporate leaders maintaining full authority, Ms. Kasmarik told this news organization.
Union organizers are also concerned with changes to the daily practice of medicine. “We don’t want to be spending our time doing paperwork and calling insurance companies and filling out forms,” said Dr. Hoffman. “We want to be in the exam room with a patient.”
The Allina providers organized after multiple requests to corporate managers failed to address their concerns. Their demands include increased staffing and help with nonclinical work so that clinicians can spend more time with their patients.
“What I’m really excited about is that we will be able to work with the other unionized groups to make change ... by being involved in health care policy at a state or national level,” Dr. Hoffman said. For example, that involvement might include challenging insurance company decisions.
Doctors Council bills itself as the largest union for attending physicians in the country, with 3,500 members, according to Joe Crane, national organizing director.
Despite an increase in union efforts since the pandemic, health care workers – particularly doctors – have been slow to join unions. Mr. Crane estimated that only about 3% of U.S. physicians are currently union members. He cited union campaigns in Massachusetts, New York, and Washington, DC. For comparison, a minority of advanced practice registered nurses (APRNs) (9%) report union membership, according to Medscape’s APRN compensation report last year.
Dr. Hoffman is confident the Allina health care providers will have enough votes to win the election to join the union. “We should have done this years ago.”
A version of this article appeared on Medscape.com.
FDA to step up oversight of cosmetics, assess ‘forever chemicals’
They are also preparing to assess potential risks of so-called forever chemicals in these products.
The Food and Drug Administration last year gained new authority over cosmetics when Congress passed the Modernization of Cosmetics Regulation Act of 2022 (MoCRA) by adding this bill to a December budget package.
“On average, consumers in the U.S. use six to 12 cosmetics products daily. But, until recently the FDA didn’t have the authority to require manufacturers to submit cosmetic product listings, including a list of ingredients used in these products, or register the facilities where they were produced,” Namandjé Bumpus, PhD, FDA’s chief scientist, said in a press release.
In the statement, the FDA announced the release of a draft guidance document that is intended to help companies comply with the transparency requirements slated to kick in this December. The agency is accepting comments on this draft guidance through Sept. 7.
“Later this year, registration and listing of cosmetic product facilities and products will become a requirement, making information about cosmetic products, including the ingredients used in products and the facilities where they are produced, readily available to the agency,” Dr. Bumpus said.
The products, according to the FDA statement, include makeup, nail polishes, shaving creams, other grooming products, perfumes, face and body cleansers, hair products, moisturizers, and other skin care items.
MoCRA “represents a sea change in how FDA regulates the cosmetics industry,” attorneys Frederick R. Ball, Alyson Walker Lotman, and Kelly A. Bonner, wrote in an article for the Food and Drug Law Institute published in spring 2023.
The FDA has called the MoCRA law “the most significant expansion” of its authority to regulate cosmetics since the Federal Food, Drug, and Cosmetic Act was passed in 1938.
The agency is in the process of expanding its staff to carry out newly authorized duties, including the tracking of adverse events. The FDA budget request for fiscal 2024, which begins Oct. 1, seeks $5 million for work needed to implement MoCRA.
PFAS, or ‘forever chemicals’
Some of the requested FDA funding is intended to prepare the agency to assess the use of per-and polyfluoroalkyl substances (PFAS) in cosmetics.
MoCRA sets a 3-year deadline for the FDA to issue an assessment of the use and potential risks of PFAS in cosmetics products. PFAS are sometimes added as ingredients in some cosmetic products, including lotions, cleansers, nail polish, shaving cream, foundation, lipstick, eyeliner, eyeshadow, and mascara, according to the FDA. Sometimes the presence of PFAS in cosmetics is unintentional and is the result of impurities in raw materials or is due to the breakdown of ingredients, the FDA said.
The FDA’s website says that so far, the available research doesn’t allow for “definitive conclusions about the potential health risks of PFAS in cosmetics.”
The Centers for Disease Control and Prevention has stated that research has suggested potential links between high levels of certain PFAS, in general, with increased cholesterol levels, changes in liver enzyme levels, increased risk of hypertension or preeclampsia in pregnant women, and increased risk of kidney or testicular cancer.
PFAS compounds often are used to resist grease, oil, water, and heat in industrial settings. They are used in thousands of products, from nonstick cookware to firefighting foams and protective gear, because they can reduce friction, according to a National Academies of Sciences, Engineering, and Medicine report on PFAS that was issued last year.
PFAS are known as “forever chemicals” because they contain a carbon-fluorine bond, which does not break naturally. Even when PFAS are transformed in the body, they can assume other forms of PFAS that preserve the troublesome carbon-fluorine bond. With PFAS, the human body is confronted with a substance it doesn’t have the tools to process.
This is in contrast to proteins and carbohydrates, which are in a sense prepackaged for relatively easy disassembly in the human body. Many of these compounds have weak links that enzymes and stomach acid can take apart, such as sulfur-to-sulfur (disulfide) bonds. That’s why protein-based biotech drugs are injected instead of administered as pills. The ultimate goal of this digestion is for the body to gain energy from these compounds.
But with PFAS, the body faces the challenge of carbon-fluorine bonds that are very hard to break down, and there is no payoff for these efforts, Graham F. Peaslee, PhD, professor of physics at the University of Notre Dame (Indiana), told this news organization.
“Nothing will naturally eat it because when you break the bond, it’s like eating celery,” he said. “You use more calories to eat the celery than you gain back from it.”
Interest from a U.S. senator
Dr. Peaslee was one of the authors of a 2021 article about PFAS in cosmetics that appeared in the journal Environmental Science and Technology Letters.
In the article, Dr. Peaslee and colleagues reported on their screening of 231 cosmetic products purchased in the United States and Canada using particle-induced gamma-ray emission spectroscopy. They found cases of undisclosed PFAS in cosmetic products. Foundations, mascaras, and lip products were noted as being especially problematic.
Sen. Susan Collins (R-ME) cited Dr. Peaslee’s article in a 2021 floor speech as she argued for having the FDA ban the intentional addition of PFAS to cosmetics.
“The findings of this study are particularly alarming, as many of these products are subject to direct human exposure,” Sen. Collins said. “For example, lipstick is often inadvertently ingested, and mascara is sometimes absorbed through tear ducts.”
In addition, workers at cosmetics plants may be exposed to PFAS and discarded cosmetics that have these compounds, which could potentially contaminate drinking water, Sen. Collins said. In 2021, she introduced legislation seeking a ban on PFAS that are intentionally added to cosmetics. That legislation did not advance through the Senate.
But the Senate Appropriations Committee, on which Sen. Collins is the ranking Republican, wants the FDA to keep a ban on PFAS in mind.
The Senate Agriculture Appropriations subcommittee, which oversees the FDA’s budget, raised the issue of PFAS and cosmetics in a June report. The FDA should develop a plan outlining research needed to inform “regulatory decision making, including potential development of a proposed rule to ban intentionally added PFAS substances in cosmetics,” the subcommittee said.
A version of this article first appeared on Medscape.com.
They are also preparing to assess potential risks of so-called forever chemicals in these products.
The Food and Drug Administration last year gained new authority over cosmetics when Congress passed the Modernization of Cosmetics Regulation Act of 2022 (MoCRA) by adding this bill to a December budget package.
“On average, consumers in the U.S. use six to 12 cosmetics products daily. But, until recently the FDA didn’t have the authority to require manufacturers to submit cosmetic product listings, including a list of ingredients used in these products, or register the facilities where they were produced,” Namandjé Bumpus, PhD, FDA’s chief scientist, said in a press release.
In the statement, the FDA announced the release of a draft guidance document that is intended to help companies comply with the transparency requirements slated to kick in this December. The agency is accepting comments on this draft guidance through Sept. 7.
“Later this year, registration and listing of cosmetic product facilities and products will become a requirement, making information about cosmetic products, including the ingredients used in products and the facilities where they are produced, readily available to the agency,” Dr. Bumpus said.
The products, according to the FDA statement, include makeup, nail polishes, shaving creams, other grooming products, perfumes, face and body cleansers, hair products, moisturizers, and other skin care items.
MoCRA “represents a sea change in how FDA regulates the cosmetics industry,” attorneys Frederick R. Ball, Alyson Walker Lotman, and Kelly A. Bonner, wrote in an article for the Food and Drug Law Institute published in spring 2023.
The FDA has called the MoCRA law “the most significant expansion” of its authority to regulate cosmetics since the Federal Food, Drug, and Cosmetic Act was passed in 1938.
The agency is in the process of expanding its staff to carry out newly authorized duties, including the tracking of adverse events. The FDA budget request for fiscal 2024, which begins Oct. 1, seeks $5 million for work needed to implement MoCRA.
PFAS, or ‘forever chemicals’
Some of the requested FDA funding is intended to prepare the agency to assess the use of per-and polyfluoroalkyl substances (PFAS) in cosmetics.
MoCRA sets a 3-year deadline for the FDA to issue an assessment of the use and potential risks of PFAS in cosmetics products. PFAS are sometimes added as ingredients in some cosmetic products, including lotions, cleansers, nail polish, shaving cream, foundation, lipstick, eyeliner, eyeshadow, and mascara, according to the FDA. Sometimes the presence of PFAS in cosmetics is unintentional and is the result of impurities in raw materials or is due to the breakdown of ingredients, the FDA said.
The FDA’s website says that so far, the available research doesn’t allow for “definitive conclusions about the potential health risks of PFAS in cosmetics.”
The Centers for Disease Control and Prevention has stated that research has suggested potential links between high levels of certain PFAS, in general, with increased cholesterol levels, changes in liver enzyme levels, increased risk of hypertension or preeclampsia in pregnant women, and increased risk of kidney or testicular cancer.
PFAS compounds often are used to resist grease, oil, water, and heat in industrial settings. They are used in thousands of products, from nonstick cookware to firefighting foams and protective gear, because they can reduce friction, according to a National Academies of Sciences, Engineering, and Medicine report on PFAS that was issued last year.
PFAS are known as “forever chemicals” because they contain a carbon-fluorine bond, which does not break naturally. Even when PFAS are transformed in the body, they can assume other forms of PFAS that preserve the troublesome carbon-fluorine bond. With PFAS, the human body is confronted with a substance it doesn’t have the tools to process.
This is in contrast to proteins and carbohydrates, which are in a sense prepackaged for relatively easy disassembly in the human body. Many of these compounds have weak links that enzymes and stomach acid can take apart, such as sulfur-to-sulfur (disulfide) bonds. That’s why protein-based biotech drugs are injected instead of administered as pills. The ultimate goal of this digestion is for the body to gain energy from these compounds.
But with PFAS, the body faces the challenge of carbon-fluorine bonds that are very hard to break down, and there is no payoff for these efforts, Graham F. Peaslee, PhD, professor of physics at the University of Notre Dame (Indiana), told this news organization.
“Nothing will naturally eat it because when you break the bond, it’s like eating celery,” he said. “You use more calories to eat the celery than you gain back from it.”
Interest from a U.S. senator
Dr. Peaslee was one of the authors of a 2021 article about PFAS in cosmetics that appeared in the journal Environmental Science and Technology Letters.
In the article, Dr. Peaslee and colleagues reported on their screening of 231 cosmetic products purchased in the United States and Canada using particle-induced gamma-ray emission spectroscopy. They found cases of undisclosed PFAS in cosmetic products. Foundations, mascaras, and lip products were noted as being especially problematic.
Sen. Susan Collins (R-ME) cited Dr. Peaslee’s article in a 2021 floor speech as she argued for having the FDA ban the intentional addition of PFAS to cosmetics.
“The findings of this study are particularly alarming, as many of these products are subject to direct human exposure,” Sen. Collins said. “For example, lipstick is often inadvertently ingested, and mascara is sometimes absorbed through tear ducts.”
In addition, workers at cosmetics plants may be exposed to PFAS and discarded cosmetics that have these compounds, which could potentially contaminate drinking water, Sen. Collins said. In 2021, she introduced legislation seeking a ban on PFAS that are intentionally added to cosmetics. That legislation did not advance through the Senate.
But the Senate Appropriations Committee, on which Sen. Collins is the ranking Republican, wants the FDA to keep a ban on PFAS in mind.
The Senate Agriculture Appropriations subcommittee, which oversees the FDA’s budget, raised the issue of PFAS and cosmetics in a June report. The FDA should develop a plan outlining research needed to inform “regulatory decision making, including potential development of a proposed rule to ban intentionally added PFAS substances in cosmetics,” the subcommittee said.
A version of this article first appeared on Medscape.com.
They are also preparing to assess potential risks of so-called forever chemicals in these products.
The Food and Drug Administration last year gained new authority over cosmetics when Congress passed the Modernization of Cosmetics Regulation Act of 2022 (MoCRA) by adding this bill to a December budget package.
“On average, consumers in the U.S. use six to 12 cosmetics products daily. But, until recently the FDA didn’t have the authority to require manufacturers to submit cosmetic product listings, including a list of ingredients used in these products, or register the facilities where they were produced,” Namandjé Bumpus, PhD, FDA’s chief scientist, said in a press release.
In the statement, the FDA announced the release of a draft guidance document that is intended to help companies comply with the transparency requirements slated to kick in this December. The agency is accepting comments on this draft guidance through Sept. 7.
“Later this year, registration and listing of cosmetic product facilities and products will become a requirement, making information about cosmetic products, including the ingredients used in products and the facilities where they are produced, readily available to the agency,” Dr. Bumpus said.
The products, according to the FDA statement, include makeup, nail polishes, shaving creams, other grooming products, perfumes, face and body cleansers, hair products, moisturizers, and other skin care items.
MoCRA “represents a sea change in how FDA regulates the cosmetics industry,” attorneys Frederick R. Ball, Alyson Walker Lotman, and Kelly A. Bonner, wrote in an article for the Food and Drug Law Institute published in spring 2023.
The FDA has called the MoCRA law “the most significant expansion” of its authority to regulate cosmetics since the Federal Food, Drug, and Cosmetic Act was passed in 1938.
The agency is in the process of expanding its staff to carry out newly authorized duties, including the tracking of adverse events. The FDA budget request for fiscal 2024, which begins Oct. 1, seeks $5 million for work needed to implement MoCRA.
PFAS, or ‘forever chemicals’
Some of the requested FDA funding is intended to prepare the agency to assess the use of per-and polyfluoroalkyl substances (PFAS) in cosmetics.
MoCRA sets a 3-year deadline for the FDA to issue an assessment of the use and potential risks of PFAS in cosmetics products. PFAS are sometimes added as ingredients in some cosmetic products, including lotions, cleansers, nail polish, shaving cream, foundation, lipstick, eyeliner, eyeshadow, and mascara, according to the FDA. Sometimes the presence of PFAS in cosmetics is unintentional and is the result of impurities in raw materials or is due to the breakdown of ingredients, the FDA said.
The FDA’s website says that so far, the available research doesn’t allow for “definitive conclusions about the potential health risks of PFAS in cosmetics.”
The Centers for Disease Control and Prevention has stated that research has suggested potential links between high levels of certain PFAS, in general, with increased cholesterol levels, changes in liver enzyme levels, increased risk of hypertension or preeclampsia in pregnant women, and increased risk of kidney or testicular cancer.
PFAS compounds often are used to resist grease, oil, water, and heat in industrial settings. They are used in thousands of products, from nonstick cookware to firefighting foams and protective gear, because they can reduce friction, according to a National Academies of Sciences, Engineering, and Medicine report on PFAS that was issued last year.
PFAS are known as “forever chemicals” because they contain a carbon-fluorine bond, which does not break naturally. Even when PFAS are transformed in the body, they can assume other forms of PFAS that preserve the troublesome carbon-fluorine bond. With PFAS, the human body is confronted with a substance it doesn’t have the tools to process.
This is in contrast to proteins and carbohydrates, which are in a sense prepackaged for relatively easy disassembly in the human body. Many of these compounds have weak links that enzymes and stomach acid can take apart, such as sulfur-to-sulfur (disulfide) bonds. That’s why protein-based biotech drugs are injected instead of administered as pills. The ultimate goal of this digestion is for the body to gain energy from these compounds.
But with PFAS, the body faces the challenge of carbon-fluorine bonds that are very hard to break down, and there is no payoff for these efforts, Graham F. Peaslee, PhD, professor of physics at the University of Notre Dame (Indiana), told this news organization.
“Nothing will naturally eat it because when you break the bond, it’s like eating celery,” he said. “You use more calories to eat the celery than you gain back from it.”
Interest from a U.S. senator
Dr. Peaslee was one of the authors of a 2021 article about PFAS in cosmetics that appeared in the journal Environmental Science and Technology Letters.
In the article, Dr. Peaslee and colleagues reported on their screening of 231 cosmetic products purchased in the United States and Canada using particle-induced gamma-ray emission spectroscopy. They found cases of undisclosed PFAS in cosmetic products. Foundations, mascaras, and lip products were noted as being especially problematic.
Sen. Susan Collins (R-ME) cited Dr. Peaslee’s article in a 2021 floor speech as she argued for having the FDA ban the intentional addition of PFAS to cosmetics.
“The findings of this study are particularly alarming, as many of these products are subject to direct human exposure,” Sen. Collins said. “For example, lipstick is often inadvertently ingested, and mascara is sometimes absorbed through tear ducts.”
In addition, workers at cosmetics plants may be exposed to PFAS and discarded cosmetics that have these compounds, which could potentially contaminate drinking water, Sen. Collins said. In 2021, she introduced legislation seeking a ban on PFAS that are intentionally added to cosmetics. That legislation did not advance through the Senate.
But the Senate Appropriations Committee, on which Sen. Collins is the ranking Republican, wants the FDA to keep a ban on PFAS in mind.
The Senate Agriculture Appropriations subcommittee, which oversees the FDA’s budget, raised the issue of PFAS and cosmetics in a June report. The FDA should develop a plan outlining research needed to inform “regulatory decision making, including potential development of a proposed rule to ban intentionally added PFAS substances in cosmetics,” the subcommittee said.
A version of this article first appeared on Medscape.com.
FDA clears new capabilities for diabetes app BlueStar
The latest clearance, announced on Aug. 23, enables the app-based platform to provide bolus insulin dose recommendations that are based on glucose and trend data from a compatible continuous glucose monitoring (CGM) device. On Aug. 15, the FDA cleared the BlueStar to use connected insulin dosing data in personalized bolus insulin dosing recommendations.
“Welldoc is the first company to receive clearance for a CGM-informed bolus calculator specifically designed for adults who manage their diabetes with multiple daily injections of insulin,” according to a company statement.
“With this clearance, Welldoc is filling a significant gap for people who require complex insulin regimens. By connecting directly with CGM data and using both glucose values and trend arrows, the BlueStar solution will provide precise and in-the-moment insulin dosing guidance directly to individuals, helping them reach their glucose targets,” endocrinologist Grazia Aleppo, MD, of Northwestern University, Chicago, said in the statement.
The new features extend the platform’s existing digital diet and lifestyle coaching capabilities. Previous FDA clearances included expansions to use most types of available insulins, including bolus and premixed insulin titration for patients with type 2 diabetes, in September 2021 and for basal insulin adjustment in June 2020.
Dr. Aleppo was a principal investigator in Welldoc’s clinical validation study for BlueStar.
A version of this article first appeared on Medscape.com.
The latest clearance, announced on Aug. 23, enables the app-based platform to provide bolus insulin dose recommendations that are based on glucose and trend data from a compatible continuous glucose monitoring (CGM) device. On Aug. 15, the FDA cleared the BlueStar to use connected insulin dosing data in personalized bolus insulin dosing recommendations.
“Welldoc is the first company to receive clearance for a CGM-informed bolus calculator specifically designed for adults who manage their diabetes with multiple daily injections of insulin,” according to a company statement.
“With this clearance, Welldoc is filling a significant gap for people who require complex insulin regimens. By connecting directly with CGM data and using both glucose values and trend arrows, the BlueStar solution will provide precise and in-the-moment insulin dosing guidance directly to individuals, helping them reach their glucose targets,” endocrinologist Grazia Aleppo, MD, of Northwestern University, Chicago, said in the statement.
The new features extend the platform’s existing digital diet and lifestyle coaching capabilities. Previous FDA clearances included expansions to use most types of available insulins, including bolus and premixed insulin titration for patients with type 2 diabetes, in September 2021 and for basal insulin adjustment in June 2020.
Dr. Aleppo was a principal investigator in Welldoc’s clinical validation study for BlueStar.
A version of this article first appeared on Medscape.com.
The latest clearance, announced on Aug. 23, enables the app-based platform to provide bolus insulin dose recommendations that are based on glucose and trend data from a compatible continuous glucose monitoring (CGM) device. On Aug. 15, the FDA cleared the BlueStar to use connected insulin dosing data in personalized bolus insulin dosing recommendations.
“Welldoc is the first company to receive clearance for a CGM-informed bolus calculator specifically designed for adults who manage their diabetes with multiple daily injections of insulin,” according to a company statement.
“With this clearance, Welldoc is filling a significant gap for people who require complex insulin regimens. By connecting directly with CGM data and using both glucose values and trend arrows, the BlueStar solution will provide precise and in-the-moment insulin dosing guidance directly to individuals, helping them reach their glucose targets,” endocrinologist Grazia Aleppo, MD, of Northwestern University, Chicago, said in the statement.
The new features extend the platform’s existing digital diet and lifestyle coaching capabilities. Previous FDA clearances included expansions to use most types of available insulins, including bolus and premixed insulin titration for patients with type 2 diabetes, in September 2021 and for basal insulin adjustment in June 2020.
Dr. Aleppo was a principal investigator in Welldoc’s clinical validation study for BlueStar.
A version of this article first appeared on Medscape.com.
Gender-affirming surgeries nearly tripled between 2016 and 2019: Study
a new study published in JAMA Network Open found.
Breast and chest surgeries were the most common procedures performed, and the number of surgical procedures carried out increased with age. The researchers said that, in addition to legal shifts, the established safety of the surgeries and resulting increase in quality of life may also help explain the increase.
“The point of this is to raise awareness and to really document the patterns of care in the United States,” said Jason Wright, MD, an associate professor at Columbia University, New York. “We hope that people understand that these procedures are being performed more commonly and they’re out there.”
A study published in 2022 in JAMA Pediatrics found that the number of chest reconstruction surgeries among U.S. adolescents rose fourfold between 2016 and 2019.
The new study included data from 2016 to 2020 in the Nationwide Ambulatory Surgery Sample and the National Inpatient Sample. More than 48,000 patients with diagnosis codes for gender identity disorder, transsexualism, or a personal history of sex reassignment were identified. Age ranges were grouped as 12-18 (7.7%), 19-30 (52.3%), and 31-40 (21.8%).
The number of gender-affirming procedures rose from 4,552 in 2016 to a peak of 13,011 in 2019. (A slight decline to 12,818 procedures in 2020 was attributed to the COVID-19 pandemic.) The surgeries were grouped into three categories: breast and chest procedures, which occurred in 56.6% of patients; genital reconstructive surgeries (35.1%), and other facial cosmetic procedures (13.9%).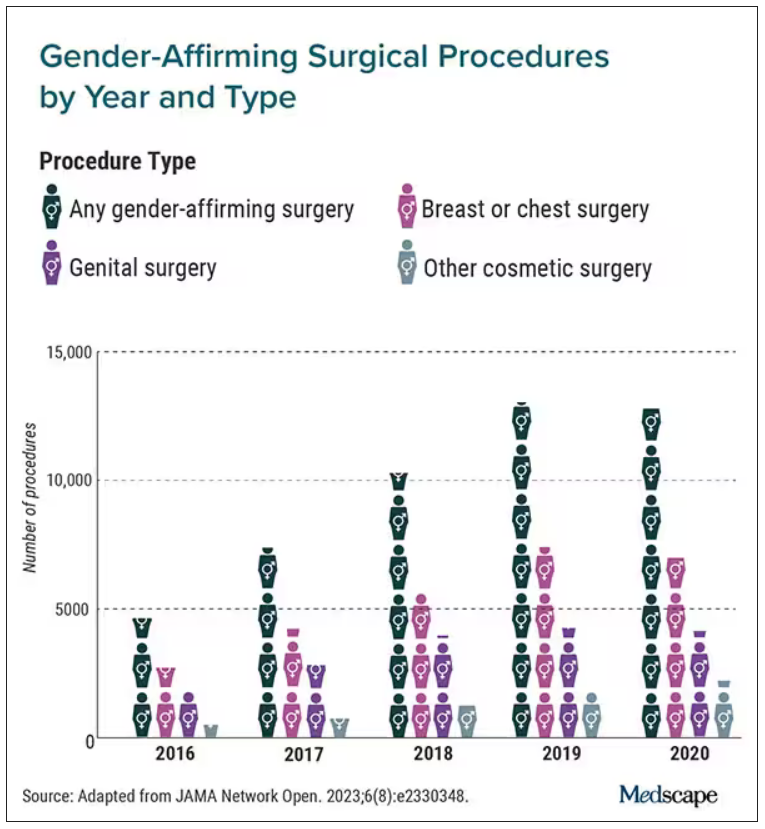
“We really wanted to try to make this as representative as we could,” Dr. Wright said. “I think this is really the best estimates that are available to date.”
Chest and breast procedures made up a higher percentage of surgeries in younger patients, while genital surgical procedures made up a higher percentage in older patients. For example, patients aged 19-30 made up 59.1% of breast or chest surgeries and 44.2% of genital surgeries. However, those aged 31-40 accounted for 26.2% of genital surgeries and 18.1% of breast or chest surgeries. For ages 41-50, the spread was more than double, accounting for 12.8% of genital surgeries and only 6.1% of breast or chest surgeries, according to the researchers.
Undocumented uptick
In addition to more inclusive health insurance, Dr. Wright said the increase in these procedures can also be attributed to studies showing their safety and the long-term association with high patient satisfaction.
Kevin Wang, MD, medical director of Providence–Swedish Health Services’ LGBTQIA+ program in Seattle, agreed that changes in health insurance coverage for gender-affirming surgery likely account in part for their increase. But he added that more clinicians are performing these procedures.
He said gender-affirming surgeries improve quality of life for the people who undergo them. The American Academy of Pediatrics has said it would be conducting a thorough review of the effects of transgender care on youth. A 2018 policy statement from the group said transgender youth should “have access to comprehensive, gender-affirming, and developmentally appropriate health care that is provided in a safe and inclusive clinical space.”
Dr. Wright cited several limitations to his group’s study that may result in the undercapture of transgender individuals and gender-affirming surgery; in particular, while the study captured inpatient and ambulatory surgical procedures in large, nationwide datasets, a small number of the procedures could have been performed in other settings.
Guiding a patient through gender-affirming care and surgical procedures can be an arduous process, including understanding their goals, using hormone therapy, and making referrals to specialists. Dr. Wang said he works to maximize his patients’ physical, mental, and emotional health, and helps them understand the risks.
He cited the double standard of a cisgender woman wanting breast augmentation without justification, but someone who identifies as transgender has many more boxes to check – for example, seeing a behavior health specialist to demonstrate they understand the risks and securing a letter of support from their primary care physician to undergo a similar procedure.
“It’s just interesting how the transgender community has to jump through so many more barriers and hoops for affirming, lifesaving procedures where you have other people who are doing it for aesthetic purposes and do not require any type of authorization,” Dr. Wang said.
Dr. Wright said he hopes the findings call attention to the need for more professionals working in the gender-affirming care field.
“I think for the medical community, it’s important to raise the idea that these procedures are becoming more common,” Dr. Wright said. “We are going to need specialists who have expertise in transgender care and surgeons who have the ability to perform these operations. Hopefully, this sheds light on the resources that are going to be required to care for these patients going forward.”
Dr. Wright reported receiving grants from Merck and personal fees from UpToDate outside the submitted work. No other disclosures were reported.
A version of this article first appeared on Medscape.com.
a new study published in JAMA Network Open found.
Breast and chest surgeries were the most common procedures performed, and the number of surgical procedures carried out increased with age. The researchers said that, in addition to legal shifts, the established safety of the surgeries and resulting increase in quality of life may also help explain the increase.
“The point of this is to raise awareness and to really document the patterns of care in the United States,” said Jason Wright, MD, an associate professor at Columbia University, New York. “We hope that people understand that these procedures are being performed more commonly and they’re out there.”
A study published in 2022 in JAMA Pediatrics found that the number of chest reconstruction surgeries among U.S. adolescents rose fourfold between 2016 and 2019.
The new study included data from 2016 to 2020 in the Nationwide Ambulatory Surgery Sample and the National Inpatient Sample. More than 48,000 patients with diagnosis codes for gender identity disorder, transsexualism, or a personal history of sex reassignment were identified. Age ranges were grouped as 12-18 (7.7%), 19-30 (52.3%), and 31-40 (21.8%).
The number of gender-affirming procedures rose from 4,552 in 2016 to a peak of 13,011 in 2019. (A slight decline to 12,818 procedures in 2020 was attributed to the COVID-19 pandemic.) The surgeries were grouped into three categories: breast and chest procedures, which occurred in 56.6% of patients; genital reconstructive surgeries (35.1%), and other facial cosmetic procedures (13.9%).
“We really wanted to try to make this as representative as we could,” Dr. Wright said. “I think this is really the best estimates that are available to date.”
Chest and breast procedures made up a higher percentage of surgeries in younger patients, while genital surgical procedures made up a higher percentage in older patients. For example, patients aged 19-30 made up 59.1% of breast or chest surgeries and 44.2% of genital surgeries. However, those aged 31-40 accounted for 26.2% of genital surgeries and 18.1% of breast or chest surgeries. For ages 41-50, the spread was more than double, accounting for 12.8% of genital surgeries and only 6.1% of breast or chest surgeries, according to the researchers.
Undocumented uptick
In addition to more inclusive health insurance, Dr. Wright said the increase in these procedures can also be attributed to studies showing their safety and the long-term association with high patient satisfaction.
Kevin Wang, MD, medical director of Providence–Swedish Health Services’ LGBTQIA+ program in Seattle, agreed that changes in health insurance coverage for gender-affirming surgery likely account in part for their increase. But he added that more clinicians are performing these procedures.
He said gender-affirming surgeries improve quality of life for the people who undergo them. The American Academy of Pediatrics has said it would be conducting a thorough review of the effects of transgender care on youth. A 2018 policy statement from the group said transgender youth should “have access to comprehensive, gender-affirming, and developmentally appropriate health care that is provided in a safe and inclusive clinical space.”
Dr. Wright cited several limitations to his group’s study that may result in the undercapture of transgender individuals and gender-affirming surgery; in particular, while the study captured inpatient and ambulatory surgical procedures in large, nationwide datasets, a small number of the procedures could have been performed in other settings.
Guiding a patient through gender-affirming care and surgical procedures can be an arduous process, including understanding their goals, using hormone therapy, and making referrals to specialists. Dr. Wang said he works to maximize his patients’ physical, mental, and emotional health, and helps them understand the risks.
He cited the double standard of a cisgender woman wanting breast augmentation without justification, but someone who identifies as transgender has many more boxes to check – for example, seeing a behavior health specialist to demonstrate they understand the risks and securing a letter of support from their primary care physician to undergo a similar procedure.
“It’s just interesting how the transgender community has to jump through so many more barriers and hoops for affirming, lifesaving procedures where you have other people who are doing it for aesthetic purposes and do not require any type of authorization,” Dr. Wang said.
Dr. Wright said he hopes the findings call attention to the need for more professionals working in the gender-affirming care field.
“I think for the medical community, it’s important to raise the idea that these procedures are becoming more common,” Dr. Wright said. “We are going to need specialists who have expertise in transgender care and surgeons who have the ability to perform these operations. Hopefully, this sheds light on the resources that are going to be required to care for these patients going forward.”
Dr. Wright reported receiving grants from Merck and personal fees from UpToDate outside the submitted work. No other disclosures were reported.
A version of this article first appeared on Medscape.com.
a new study published in JAMA Network Open found.
Breast and chest surgeries were the most common procedures performed, and the number of surgical procedures carried out increased with age. The researchers said that, in addition to legal shifts, the established safety of the surgeries and resulting increase in quality of life may also help explain the increase.
“The point of this is to raise awareness and to really document the patterns of care in the United States,” said Jason Wright, MD, an associate professor at Columbia University, New York. “We hope that people understand that these procedures are being performed more commonly and they’re out there.”
A study published in 2022 in JAMA Pediatrics found that the number of chest reconstruction surgeries among U.S. adolescents rose fourfold between 2016 and 2019.
The new study included data from 2016 to 2020 in the Nationwide Ambulatory Surgery Sample and the National Inpatient Sample. More than 48,000 patients with diagnosis codes for gender identity disorder, transsexualism, or a personal history of sex reassignment were identified. Age ranges were grouped as 12-18 (7.7%), 19-30 (52.3%), and 31-40 (21.8%).
The number of gender-affirming procedures rose from 4,552 in 2016 to a peak of 13,011 in 2019. (A slight decline to 12,818 procedures in 2020 was attributed to the COVID-19 pandemic.) The surgeries were grouped into three categories: breast and chest procedures, which occurred in 56.6% of patients; genital reconstructive surgeries (35.1%), and other facial cosmetic procedures (13.9%).
“We really wanted to try to make this as representative as we could,” Dr. Wright said. “I think this is really the best estimates that are available to date.”
Chest and breast procedures made up a higher percentage of surgeries in younger patients, while genital surgical procedures made up a higher percentage in older patients. For example, patients aged 19-30 made up 59.1% of breast or chest surgeries and 44.2% of genital surgeries. However, those aged 31-40 accounted for 26.2% of genital surgeries and 18.1% of breast or chest surgeries. For ages 41-50, the spread was more than double, accounting for 12.8% of genital surgeries and only 6.1% of breast or chest surgeries, according to the researchers.
Undocumented uptick
In addition to more inclusive health insurance, Dr. Wright said the increase in these procedures can also be attributed to studies showing their safety and the long-term association with high patient satisfaction.
Kevin Wang, MD, medical director of Providence–Swedish Health Services’ LGBTQIA+ program in Seattle, agreed that changes in health insurance coverage for gender-affirming surgery likely account in part for their increase. But he added that more clinicians are performing these procedures.
He said gender-affirming surgeries improve quality of life for the people who undergo them. The American Academy of Pediatrics has said it would be conducting a thorough review of the effects of transgender care on youth. A 2018 policy statement from the group said transgender youth should “have access to comprehensive, gender-affirming, and developmentally appropriate health care that is provided in a safe and inclusive clinical space.”
Dr. Wright cited several limitations to his group’s study that may result in the undercapture of transgender individuals and gender-affirming surgery; in particular, while the study captured inpatient and ambulatory surgical procedures in large, nationwide datasets, a small number of the procedures could have been performed in other settings.
Guiding a patient through gender-affirming care and surgical procedures can be an arduous process, including understanding their goals, using hormone therapy, and making referrals to specialists. Dr. Wang said he works to maximize his patients’ physical, mental, and emotional health, and helps them understand the risks.
He cited the double standard of a cisgender woman wanting breast augmentation without justification, but someone who identifies as transgender has many more boxes to check – for example, seeing a behavior health specialist to demonstrate they understand the risks and securing a letter of support from their primary care physician to undergo a similar procedure.
“It’s just interesting how the transgender community has to jump through so many more barriers and hoops for affirming, lifesaving procedures where you have other people who are doing it for aesthetic purposes and do not require any type of authorization,” Dr. Wang said.
Dr. Wright said he hopes the findings call attention to the need for more professionals working in the gender-affirming care field.
“I think for the medical community, it’s important to raise the idea that these procedures are becoming more common,” Dr. Wright said. “We are going to need specialists who have expertise in transgender care and surgeons who have the ability to perform these operations. Hopefully, this sheds light on the resources that are going to be required to care for these patients going forward.”
Dr. Wright reported receiving grants from Merck and personal fees from UpToDate outside the submitted work. No other disclosures were reported.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Medicare announces 10 drugs targeted for price cuts in 2026
People on Medicare may in 2026 see prices drop for 10 medicines, including pricey diabetes, cancer, blood clot, and arthritis treatments, if advocates for federal drug-price negotiations can implement their plans amid tough opposition.
It’s unclear at this time, though, how these negotiations will play out. The Chamber of Commerce has sided with pharmaceutical companies in bids to block direct Medicare negotiation of drug prices. Many influential Republicans in Congress oppose this plan, which has deep support from both Democrats and AARP.
While facing strong opposition to negotiations, the Centers for Medicare & Medicaid Services sought in its announcement to illustrate the high costs of the selected medicines.
CMS provided data on total Part D costs for selected medicines for the period from June 2022 to May 2023, along with tallies of the number of people taking these drugs. The 10 selected medicines are as follows:
- Eliquis (generic name: apixaban), used to prevent and treat serious blood clots. It is taken by about 3.7 million people through Part D plans. The estimated cost is $16.4 billion.
- Jardiance (generic name: empagliflozin), used for diabetes and heart failure. It is taken by almost 1.6 million people through Part D plans. The estimated cost is $7.06 billion.
- Xarelto (generic name: rivaroxaban), used for blood clots. It is taken by about 1.3 million people through Part D plans. The estimated cost is $6 billion.
- Januvia (generic name: sitagliptin), used for diabetes. It is taken by about 869,00 people through Part D plans. The estimated cost is $4.1 billion.
- Farxiga (generic name: dapagliflozin), used for diabetes, heart failure, and chronic kidney disease. It is taken by about 799,000 people through Part D plans. The estimated cost is almost $3.3 billion.
- Entresto (generic name: sacubitril/valsartan), used to treat heart failure. It is taken by 587,000 people through Part D plans. The estimated cost is $2.9 billion.
- Enbrel( generic name: etanercept), used for rheumatoid arthritis, psoriasis, and psoriatic arthritis. It is taken by 48,000 people through Part D plans. The estimated cost is $2.8 billion.
- Imbruvica (generic name: ibrutinib), used to treat some blood cancers. It is taken by about 20,000 people in Part D plans. The estimated cost is $2.7 billion.
- Stelara (generic name: ustekinumab), used to treat plaque psoriasis, psoriatic arthritis, or certain bowel conditions (Crohn’s disease, ulcerative colitis). It is used by about 22,000 people through Part D plans. The estimated cost is $2.6 billion.
- Fiasp; Fiasp FlexTouch; Fiasp PenFill; NovoLog; NovoLog FlexPen; NovoLog PenFill. These are forms of insulin used to treat diabetes. They are used by about 777,000 people through Part D plans. The estimated cost is $2.6 billion.
A vocal critic of Medicare drug negotiations, Joel White, president of the Council for Affordable Health Coverage, called the announcement of the 10 drugs selected for negotiation “a hollow victory lap.” A former Republican staffer on the House Ways and Means Committee, Mr. White aided with the development of the Medicare Part D plans and has kept tabs on the pharmacy programs since its launch in 2006.
“No one’s costs will go down now or for years because of this announcement” about Part D negotiations, Mr. White said in a statement.
According to its website, CAHC includes among its members the American Academy of Ophthalmology as well as some patient groups, drugmakers, such as Johnson & Johnson, and insurers and industry groups, such as the National Association of Manufacturers.
Separately, the influential Chamber of Commerce is making a strong push to at least delay the implementation of the Medicare Part D drug negotiations. On Aug. 28, the chamber released a letter sent to the Biden administration, raising concerns about a “rush” to implement the provisions of the Inflation Reduction Act.
The chamber also has filed suit to challenge the drug negotiation provisions of the Inflation Reduction Act, requesting that the court issue a preliminary injunction by Oct. 1, 2023.
Other pending legal challenges to direct Medicare drug negotiations include suits filed by Merck, Bristol-Myers Squibb, Johnson & Johnson, Boehringer Ingelheim, and AstraZeneca, according to an email from Pharmaceutical Research and Manufacturers of America. PhRMA also said it is a party to a case.
In addition, the three congressional Republicans with most direct influence over Medicare policy issued on Aug. 29 a joint statement outlining their objections to the planned negotiations on drug prices.
This drug-negotiation proposal is “an unworkable, legally dubious scheme that will lead to higher prices for new drugs coming to market, stifle the development of new cures, and destroy jobs,” said House Energy and Commerce Committee Chair Cathy McMorris Rodgers (R-Wash.), House Ways and Means Committee Chair Jason Smith (R-Mo.), and Senate Finance Committee Ranking Member Mike Crapo (R-Idaho).
Democrats were equally firm and vocal in their support of the negotiations. Senate Finance Chairman Ron Wyden (D-Ore.) issued a statement on Aug. 29 that said the release of the list of the 10 drugs selected for Medicare drug negotiations is part of a “seismic shift in the relationship between Big Pharma, the federal government, and seniors who are counting on lower prices.
“I will be following the negotiation process closely and will fight any attempt by Big Pharma to undo or undermine the progress that’s been made,” Mr. Wyden said.
In addition, AARP issued a statement of its continued support for Medicare drug negotiations.
“The No. 1 reason seniors skip or ration their prescriptions is because they can’t afford them. This must stop,” said AARP executive vice president and chief advocacy and engagement officer Nancy LeaMond in the statement. “The big drug companies and their allies continue suing to overturn the Medicare drug price negotiation program to keep up their price gouging. We can’t allow seniors to be Big Pharma’s cash machine anymore.”
A version of this article first appeared on Medscape.com.
People on Medicare may in 2026 see prices drop for 10 medicines, including pricey diabetes, cancer, blood clot, and arthritis treatments, if advocates for federal drug-price negotiations can implement their plans amid tough opposition.
It’s unclear at this time, though, how these negotiations will play out. The Chamber of Commerce has sided with pharmaceutical companies in bids to block direct Medicare negotiation of drug prices. Many influential Republicans in Congress oppose this plan, which has deep support from both Democrats and AARP.
While facing strong opposition to negotiations, the Centers for Medicare & Medicaid Services sought in its announcement to illustrate the high costs of the selected medicines.
CMS provided data on total Part D costs for selected medicines for the period from June 2022 to May 2023, along with tallies of the number of people taking these drugs. The 10 selected medicines are as follows:
- Eliquis (generic name: apixaban), used to prevent and treat serious blood clots. It is taken by about 3.7 million people through Part D plans. The estimated cost is $16.4 billion.
- Jardiance (generic name: empagliflozin), used for diabetes and heart failure. It is taken by almost 1.6 million people through Part D plans. The estimated cost is $7.06 billion.
- Xarelto (generic name: rivaroxaban), used for blood clots. It is taken by about 1.3 million people through Part D plans. The estimated cost is $6 billion.
- Januvia (generic name: sitagliptin), used for diabetes. It is taken by about 869,00 people through Part D plans. The estimated cost is $4.1 billion.
- Farxiga (generic name: dapagliflozin), used for diabetes, heart failure, and chronic kidney disease. It is taken by about 799,000 people through Part D plans. The estimated cost is almost $3.3 billion.
- Entresto (generic name: sacubitril/valsartan), used to treat heart failure. It is taken by 587,000 people through Part D plans. The estimated cost is $2.9 billion.
- Enbrel( generic name: etanercept), used for rheumatoid arthritis, psoriasis, and psoriatic arthritis. It is taken by 48,000 people through Part D plans. The estimated cost is $2.8 billion.
- Imbruvica (generic name: ibrutinib), used to treat some blood cancers. It is taken by about 20,000 people in Part D plans. The estimated cost is $2.7 billion.
- Stelara (generic name: ustekinumab), used to treat plaque psoriasis, psoriatic arthritis, or certain bowel conditions (Crohn’s disease, ulcerative colitis). It is used by about 22,000 people through Part D plans. The estimated cost is $2.6 billion.
- Fiasp; Fiasp FlexTouch; Fiasp PenFill; NovoLog; NovoLog FlexPen; NovoLog PenFill. These are forms of insulin used to treat diabetes. They are used by about 777,000 people through Part D plans. The estimated cost is $2.6 billion.
A vocal critic of Medicare drug negotiations, Joel White, president of the Council for Affordable Health Coverage, called the announcement of the 10 drugs selected for negotiation “a hollow victory lap.” A former Republican staffer on the House Ways and Means Committee, Mr. White aided with the development of the Medicare Part D plans and has kept tabs on the pharmacy programs since its launch in 2006.
“No one’s costs will go down now or for years because of this announcement” about Part D negotiations, Mr. White said in a statement.
According to its website, CAHC includes among its members the American Academy of Ophthalmology as well as some patient groups, drugmakers, such as Johnson & Johnson, and insurers and industry groups, such as the National Association of Manufacturers.
Separately, the influential Chamber of Commerce is making a strong push to at least delay the implementation of the Medicare Part D drug negotiations. On Aug. 28, the chamber released a letter sent to the Biden administration, raising concerns about a “rush” to implement the provisions of the Inflation Reduction Act.
The chamber also has filed suit to challenge the drug negotiation provisions of the Inflation Reduction Act, requesting that the court issue a preliminary injunction by Oct. 1, 2023.
Other pending legal challenges to direct Medicare drug negotiations include suits filed by Merck, Bristol-Myers Squibb, Johnson & Johnson, Boehringer Ingelheim, and AstraZeneca, according to an email from Pharmaceutical Research and Manufacturers of America. PhRMA also said it is a party to a case.
In addition, the three congressional Republicans with most direct influence over Medicare policy issued on Aug. 29 a joint statement outlining their objections to the planned negotiations on drug prices.
This drug-negotiation proposal is “an unworkable, legally dubious scheme that will lead to higher prices for new drugs coming to market, stifle the development of new cures, and destroy jobs,” said House Energy and Commerce Committee Chair Cathy McMorris Rodgers (R-Wash.), House Ways and Means Committee Chair Jason Smith (R-Mo.), and Senate Finance Committee Ranking Member Mike Crapo (R-Idaho).
Democrats were equally firm and vocal in their support of the negotiations. Senate Finance Chairman Ron Wyden (D-Ore.) issued a statement on Aug. 29 that said the release of the list of the 10 drugs selected for Medicare drug negotiations is part of a “seismic shift in the relationship between Big Pharma, the federal government, and seniors who are counting on lower prices.
“I will be following the negotiation process closely and will fight any attempt by Big Pharma to undo or undermine the progress that’s been made,” Mr. Wyden said.
In addition, AARP issued a statement of its continued support for Medicare drug negotiations.
“The No. 1 reason seniors skip or ration their prescriptions is because they can’t afford them. This must stop,” said AARP executive vice president and chief advocacy and engagement officer Nancy LeaMond in the statement. “The big drug companies and their allies continue suing to overturn the Medicare drug price negotiation program to keep up their price gouging. We can’t allow seniors to be Big Pharma’s cash machine anymore.”
A version of this article first appeared on Medscape.com.
People on Medicare may in 2026 see prices drop for 10 medicines, including pricey diabetes, cancer, blood clot, and arthritis treatments, if advocates for federal drug-price negotiations can implement their plans amid tough opposition.
It’s unclear at this time, though, how these negotiations will play out. The Chamber of Commerce has sided with pharmaceutical companies in bids to block direct Medicare negotiation of drug prices. Many influential Republicans in Congress oppose this plan, which has deep support from both Democrats and AARP.
While facing strong opposition to negotiations, the Centers for Medicare & Medicaid Services sought in its announcement to illustrate the high costs of the selected medicines.
CMS provided data on total Part D costs for selected medicines for the period from June 2022 to May 2023, along with tallies of the number of people taking these drugs. The 10 selected medicines are as follows:
- Eliquis (generic name: apixaban), used to prevent and treat serious blood clots. It is taken by about 3.7 million people through Part D plans. The estimated cost is $16.4 billion.
- Jardiance (generic name: empagliflozin), used for diabetes and heart failure. It is taken by almost 1.6 million people through Part D plans. The estimated cost is $7.06 billion.
- Xarelto (generic name: rivaroxaban), used for blood clots. It is taken by about 1.3 million people through Part D plans. The estimated cost is $6 billion.
- Januvia (generic name: sitagliptin), used for diabetes. It is taken by about 869,00 people through Part D plans. The estimated cost is $4.1 billion.
- Farxiga (generic name: dapagliflozin), used for diabetes, heart failure, and chronic kidney disease. It is taken by about 799,000 people through Part D plans. The estimated cost is almost $3.3 billion.
- Entresto (generic name: sacubitril/valsartan), used to treat heart failure. It is taken by 587,000 people through Part D plans. The estimated cost is $2.9 billion.
- Enbrel( generic name: etanercept), used for rheumatoid arthritis, psoriasis, and psoriatic arthritis. It is taken by 48,000 people through Part D plans. The estimated cost is $2.8 billion.
- Imbruvica (generic name: ibrutinib), used to treat some blood cancers. It is taken by about 20,000 people in Part D plans. The estimated cost is $2.7 billion.
- Stelara (generic name: ustekinumab), used to treat plaque psoriasis, psoriatic arthritis, or certain bowel conditions (Crohn’s disease, ulcerative colitis). It is used by about 22,000 people through Part D plans. The estimated cost is $2.6 billion.
- Fiasp; Fiasp FlexTouch; Fiasp PenFill; NovoLog; NovoLog FlexPen; NovoLog PenFill. These are forms of insulin used to treat diabetes. They are used by about 777,000 people through Part D plans. The estimated cost is $2.6 billion.
A vocal critic of Medicare drug negotiations, Joel White, president of the Council for Affordable Health Coverage, called the announcement of the 10 drugs selected for negotiation “a hollow victory lap.” A former Republican staffer on the House Ways and Means Committee, Mr. White aided with the development of the Medicare Part D plans and has kept tabs on the pharmacy programs since its launch in 2006.
“No one’s costs will go down now or for years because of this announcement” about Part D negotiations, Mr. White said in a statement.
According to its website, CAHC includes among its members the American Academy of Ophthalmology as well as some patient groups, drugmakers, such as Johnson & Johnson, and insurers and industry groups, such as the National Association of Manufacturers.
Separately, the influential Chamber of Commerce is making a strong push to at least delay the implementation of the Medicare Part D drug negotiations. On Aug. 28, the chamber released a letter sent to the Biden administration, raising concerns about a “rush” to implement the provisions of the Inflation Reduction Act.
The chamber also has filed suit to challenge the drug negotiation provisions of the Inflation Reduction Act, requesting that the court issue a preliminary injunction by Oct. 1, 2023.
Other pending legal challenges to direct Medicare drug negotiations include suits filed by Merck, Bristol-Myers Squibb, Johnson & Johnson, Boehringer Ingelheim, and AstraZeneca, according to an email from Pharmaceutical Research and Manufacturers of America. PhRMA also said it is a party to a case.
In addition, the three congressional Republicans with most direct influence over Medicare policy issued on Aug. 29 a joint statement outlining their objections to the planned negotiations on drug prices.
This drug-negotiation proposal is “an unworkable, legally dubious scheme that will lead to higher prices for new drugs coming to market, stifle the development of new cures, and destroy jobs,” said House Energy and Commerce Committee Chair Cathy McMorris Rodgers (R-Wash.), House Ways and Means Committee Chair Jason Smith (R-Mo.), and Senate Finance Committee Ranking Member Mike Crapo (R-Idaho).
Democrats were equally firm and vocal in their support of the negotiations. Senate Finance Chairman Ron Wyden (D-Ore.) issued a statement on Aug. 29 that said the release of the list of the 10 drugs selected for Medicare drug negotiations is part of a “seismic shift in the relationship between Big Pharma, the federal government, and seniors who are counting on lower prices.
“I will be following the negotiation process closely and will fight any attempt by Big Pharma to undo or undermine the progress that’s been made,” Mr. Wyden said.
In addition, AARP issued a statement of its continued support for Medicare drug negotiations.
“The No. 1 reason seniors skip or ration their prescriptions is because they can’t afford them. This must stop,” said AARP executive vice president and chief advocacy and engagement officer Nancy LeaMond in the statement. “The big drug companies and their allies continue suing to overturn the Medicare drug price negotiation program to keep up their price gouging. We can’t allow seniors to be Big Pharma’s cash machine anymore.”
A version of this article first appeared on Medscape.com.





