User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
‘Vaginal dryness’ can be fatal. No, really.
This transcript has been edited for clarity.
What do you mean, Dr. Rubin? How is vaginal dryness killing women? We minimize the term vaginal dryness. When women come to our offices and complain of a little vaginal dryness – or they don’t even come to our office to complain of it because the doctor can’t be bothered with a little vaginal dryness — what they don’t understand is that this “little vaginal dryness” is really something called genitourinary syndrome of menopause (GSM). They don’t know that because they’ve never heard of it, and you may have never heard of it either. In 2014, we changed the terms vaginal dryness and vulvovaginal atrophy or atrophic vaginitis to GSM to make it short and simple.
GSM – what does it mean? It’s not just a little vaginal dryness. It turns out that all of the genital and urinary symptoms from menopause just get worse over time. The bladder, the urethra, and the vagina have lots of hormone receptors, including estrogen and testosterone. When the body no longer makes those hormones, the system doesn’t work very well, and genital and urinary symptoms occur that just get worse over time without treatment. Unlike hot flashes, which tend to go away, GSM does not.
What are the symptoms of GSM? Some are sexual: a little vaginal dryness, pain with sex, and worsening orgasm. But there are also genital and urinary symptoms that get worse: itching, burning irritation, rawness, an awareness of their genitals that the patient has never had before. And as a urologist, we see frequency, urgency, and leakage.
The thing that kills women is recurrent urinary tract infections (UTIs). Did you know that UTIs account for 7 million visits and hospitalizations annually and 25% of all infections in older people? In fact, apparently one-third of the total Medicare expenditure is around UTIs. Not preventing UTIs is costing our health care system an enormous amount of money and resources.
Did you know we’ve had safe and effective treatment options for GSM since the 1970s? Vaginal hormones have existed since the 1970s, but we’re using them only for pain with sex and not for GSM. In fact, data show that by using vaginal hormones, we can prevent UTIs by more than 50%. We can save lives using safe, effective, local, low-dose vaginal hormone strategies. And they are safe and effective for all of our patients in pre- and post menopause.
There are five different treatment options: vaginal estrogen inserts, vaginal estrogen creams, vaginal dehydroepiandrosterone (DHEA), low-dose vaginal estrogen rings, and an oral pill option called ospemifene (Osphena). All are used to treat GSM and will only work if your patient actually uses them and continues to use them.
These treatments are safe. They are effective. They do not increase the level of systemic hormones in the bloodstream. I have many patients with breast cancer who use these products as well. The only patients you may want to talk to your oncology colleagues about is women on active aromatase inhibitors.
We have to understand that UTIs kill people and having GSM is debilitating, often requiring pain medication because it can hurt to sit or to wear pads and our patients’ quality of life is severely affected. So please consider learning how to treat GSM. It turns out you don’t have to do exams. You don’t have to do follow-up. You can give these therapies, and women can use them for life.
Now, if your patient has vaginal bleeding, of course they need to see their gynecologist. But this is something every primary care doctor can and should do. As a urologist, we prescribe a lot of tamsulosin (Flomax) for our male patients to help with urination. Vaginal estrogen or DHEA is basically like Flomax for women, but it prevents UTIs and actually works like sildenafil (Viagra) because it can help orgasm and reduce pain with sex.
You have access to affordable, safe, effective treatment options to treat GSM. So check them out and hopefully change the world.
Dr. Rubin is an assistant clinical professor in the department of urology at Georgetown University, Washington. She reported conflicts of interest with Sprout, Maternal Medical, Absorption Pharmaceuticals, GlaxoSmithKline, and Endo.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
What do you mean, Dr. Rubin? How is vaginal dryness killing women? We minimize the term vaginal dryness. When women come to our offices and complain of a little vaginal dryness – or they don’t even come to our office to complain of it because the doctor can’t be bothered with a little vaginal dryness — what they don’t understand is that this “little vaginal dryness” is really something called genitourinary syndrome of menopause (GSM). They don’t know that because they’ve never heard of it, and you may have never heard of it either. In 2014, we changed the terms vaginal dryness and vulvovaginal atrophy or atrophic vaginitis to GSM to make it short and simple.
GSM – what does it mean? It’s not just a little vaginal dryness. It turns out that all of the genital and urinary symptoms from menopause just get worse over time. The bladder, the urethra, and the vagina have lots of hormone receptors, including estrogen and testosterone. When the body no longer makes those hormones, the system doesn’t work very well, and genital and urinary symptoms occur that just get worse over time without treatment. Unlike hot flashes, which tend to go away, GSM does not.
What are the symptoms of GSM? Some are sexual: a little vaginal dryness, pain with sex, and worsening orgasm. But there are also genital and urinary symptoms that get worse: itching, burning irritation, rawness, an awareness of their genitals that the patient has never had before. And as a urologist, we see frequency, urgency, and leakage.
The thing that kills women is recurrent urinary tract infections (UTIs). Did you know that UTIs account for 7 million visits and hospitalizations annually and 25% of all infections in older people? In fact, apparently one-third of the total Medicare expenditure is around UTIs. Not preventing UTIs is costing our health care system an enormous amount of money and resources.
Did you know we’ve had safe and effective treatment options for GSM since the 1970s? Vaginal hormones have existed since the 1970s, but we’re using them only for pain with sex and not for GSM. In fact, data show that by using vaginal hormones, we can prevent UTIs by more than 50%. We can save lives using safe, effective, local, low-dose vaginal hormone strategies. And they are safe and effective for all of our patients in pre- and post menopause.
There are five different treatment options: vaginal estrogen inserts, vaginal estrogen creams, vaginal dehydroepiandrosterone (DHEA), low-dose vaginal estrogen rings, and an oral pill option called ospemifene (Osphena). All are used to treat GSM and will only work if your patient actually uses them and continues to use them.
These treatments are safe. They are effective. They do not increase the level of systemic hormones in the bloodstream. I have many patients with breast cancer who use these products as well. The only patients you may want to talk to your oncology colleagues about is women on active aromatase inhibitors.
We have to understand that UTIs kill people and having GSM is debilitating, often requiring pain medication because it can hurt to sit or to wear pads and our patients’ quality of life is severely affected. So please consider learning how to treat GSM. It turns out you don’t have to do exams. You don’t have to do follow-up. You can give these therapies, and women can use them for life.
Now, if your patient has vaginal bleeding, of course they need to see their gynecologist. But this is something every primary care doctor can and should do. As a urologist, we prescribe a lot of tamsulosin (Flomax) for our male patients to help with urination. Vaginal estrogen or DHEA is basically like Flomax for women, but it prevents UTIs and actually works like sildenafil (Viagra) because it can help orgasm and reduce pain with sex.
You have access to affordable, safe, effective treatment options to treat GSM. So check them out and hopefully change the world.
Dr. Rubin is an assistant clinical professor in the department of urology at Georgetown University, Washington. She reported conflicts of interest with Sprout, Maternal Medical, Absorption Pharmaceuticals, GlaxoSmithKline, and Endo.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
What do you mean, Dr. Rubin? How is vaginal dryness killing women? We minimize the term vaginal dryness. When women come to our offices and complain of a little vaginal dryness – or they don’t even come to our office to complain of it because the doctor can’t be bothered with a little vaginal dryness — what they don’t understand is that this “little vaginal dryness” is really something called genitourinary syndrome of menopause (GSM). They don’t know that because they’ve never heard of it, and you may have never heard of it either. In 2014, we changed the terms vaginal dryness and vulvovaginal atrophy or atrophic vaginitis to GSM to make it short and simple.
GSM – what does it mean? It’s not just a little vaginal dryness. It turns out that all of the genital and urinary symptoms from menopause just get worse over time. The bladder, the urethra, and the vagina have lots of hormone receptors, including estrogen and testosterone. When the body no longer makes those hormones, the system doesn’t work very well, and genital and urinary symptoms occur that just get worse over time without treatment. Unlike hot flashes, which tend to go away, GSM does not.
What are the symptoms of GSM? Some are sexual: a little vaginal dryness, pain with sex, and worsening orgasm. But there are also genital and urinary symptoms that get worse: itching, burning irritation, rawness, an awareness of their genitals that the patient has never had before. And as a urologist, we see frequency, urgency, and leakage.
The thing that kills women is recurrent urinary tract infections (UTIs). Did you know that UTIs account for 7 million visits and hospitalizations annually and 25% of all infections in older people? In fact, apparently one-third of the total Medicare expenditure is around UTIs. Not preventing UTIs is costing our health care system an enormous amount of money and resources.
Did you know we’ve had safe and effective treatment options for GSM since the 1970s? Vaginal hormones have existed since the 1970s, but we’re using them only for pain with sex and not for GSM. In fact, data show that by using vaginal hormones, we can prevent UTIs by more than 50%. We can save lives using safe, effective, local, low-dose vaginal hormone strategies. And they are safe and effective for all of our patients in pre- and post menopause.
There are five different treatment options: vaginal estrogen inserts, vaginal estrogen creams, vaginal dehydroepiandrosterone (DHEA), low-dose vaginal estrogen rings, and an oral pill option called ospemifene (Osphena). All are used to treat GSM and will only work if your patient actually uses them and continues to use them.
These treatments are safe. They are effective. They do not increase the level of systemic hormones in the bloodstream. I have many patients with breast cancer who use these products as well. The only patients you may want to talk to your oncology colleagues about is women on active aromatase inhibitors.
We have to understand that UTIs kill people and having GSM is debilitating, often requiring pain medication because it can hurt to sit or to wear pads and our patients’ quality of life is severely affected. So please consider learning how to treat GSM. It turns out you don’t have to do exams. You don’t have to do follow-up. You can give these therapies, and women can use them for life.
Now, if your patient has vaginal bleeding, of course they need to see their gynecologist. But this is something every primary care doctor can and should do. As a urologist, we prescribe a lot of tamsulosin (Flomax) for our male patients to help with urination. Vaginal estrogen or DHEA is basically like Flomax for women, but it prevents UTIs and actually works like sildenafil (Viagra) because it can help orgasm and reduce pain with sex.
You have access to affordable, safe, effective treatment options to treat GSM. So check them out and hopefully change the world.
Dr. Rubin is an assistant clinical professor in the department of urology at Georgetown University, Washington. She reported conflicts of interest with Sprout, Maternal Medical, Absorption Pharmaceuticals, GlaxoSmithKline, and Endo.
A version of this article first appeared on Medscape.com.
Nonsurgical option for more large thyroid nodule patients?
WASHINGTON – , compared with only those that were.
While more research is needed, “the risk of false negative FNA results for large nodules may not be as high as reported in previous studies if you include patients who do not have indication for surgery, such as compressive symptoms, suspicious ultrasound features, etc.,” senior author Tracy Tylee, MD, an associate professor of endocrinology at the University of Washington, Seattle, said in an interview.
The implication is that nonsurgical options such as radiofrequency ablation may be appropriate for more patients than realized, she added.
“Clinicians should consider following these patients more conservatively, either with a second FNA to confirm [the] nodule is benign or with ultrasound follow-up for 5 years with intervention only if [there are] significant changes on imaging,” she said.
The findings were presented at the annual meeting of the American Thyroid Association.
Concerns about nodules over 4 cm having high false negative rates
Management of large thyroid nodules over 4 cm that are classified as Bethesda II, indicative of being benign, is complicated by concerns of false negatives in such cases. While the false negative rate for thyroid nodules in general is approximately 3%, the rate for large nodules over 4 cm has been reported as high as 35%.
Importantly, however, most studies evaluating the issue only involve patients who have received thyroid surgery, whereas most benign nodules are not referred for surgery.
“This may overestimate the false negative FNA biopsy risk for this group,” first author Melbin Thomas, MD, also of the University of Washington, said in her talk.
To better assess the false negative rate in the broader context of large nodules that did and did not undergo surgery, Dr. Thomas and her colleagues conducted a retrospective chart review of all patients undergoing FNA biopsy at her center between 2008 and 2014 for thyroid nodules larger than 4 cm and initially classified as Bethesda II, or benign.
With a follow-up of up to 10 years, nodules were considered accurately benign if they showed benign pathology on surgical resection, if they remained benign based on repeat FNA biopsy with Bethesda II results, or if there were no changes on imaging characteristics on ultrasound after at least 2 years.
Overall, 47 nodules over 4 cm and Bethesda II cytology were included, with an average follow-up of 5 years (range 2.2-9.7 years).
Of the nodules, 23 were treated with surgery, two of which were determined to have been malignant (8.7%) and, hence, false negatives. Nine of the nodules had repeat FNA, with none found to be malignant, and 15 received repeat ultrasound, also with no malignancies.
Overall, the false negative rate including all patients was 4.3%.
“False negative FNA biopsy results were not markedly elevated if nodules greater than 4 cm are evaluated, but rates were considerably higher if limited to surgical patients,” Dr. Thomas said.
Clinicians may be compelled to perform more aggressive surgery on large but benign thyroid nodules for a number of reasons, Dr. Tylee noted.
“A concern is that we may discontinue follow-up on these larger nodules and fail to diagnose a cancer early on, before there has been extrathyroidal extension or lymph node metastases,” she said.
In such cases, patients could wind up presenting at a higher stage of disease and require more intensive therapy.
However, with a low false negative rate overall, “all of this can increase the long-term health care costs and anxiety for patients, so having a better understanding of the true benign rate for large nodules is important,” she concluded.
Commenting on the research, Rodis D. Paparodis, MD, chief of Endocrinology, Diabetes, and Metabolism Clinics, in Patras, Greece, said the findings underscore that, as a surgical procedure, “thyroidectomy should be used cautiously, only when the benefit outweighs the risk.”
In his own previous multicenter study, Dr. Paparodis conducted a review of nearly 2,500 thyroidectomies that were performed based on size or longterm slow growth despite preoperative benign FNA findings. The results showed that only 1.9% of patients had any form of thyroid cancer in the nodule that had led to surgery; however, multiple other significant cancers were often present in other locations in the gland.
“Therefore, we suggest that careful sonographic evaluation of all thyroid nodules is warranted prior to deciding and planning the extent of surgical management for multinodular goiter,” he told this news organization.
“In addition, FNA of all suspicious nodules is required as well, to avoid unnecessary surprises in surgical pathology.”
Dr. Tylee, Dr. Thomas, and Dr. Paparodis report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
WASHINGTON – , compared with only those that were.
While more research is needed, “the risk of false negative FNA results for large nodules may not be as high as reported in previous studies if you include patients who do not have indication for surgery, such as compressive symptoms, suspicious ultrasound features, etc.,” senior author Tracy Tylee, MD, an associate professor of endocrinology at the University of Washington, Seattle, said in an interview.
The implication is that nonsurgical options such as radiofrequency ablation may be appropriate for more patients than realized, she added.
“Clinicians should consider following these patients more conservatively, either with a second FNA to confirm [the] nodule is benign or with ultrasound follow-up for 5 years with intervention only if [there are] significant changes on imaging,” she said.
The findings were presented at the annual meeting of the American Thyroid Association.
Concerns about nodules over 4 cm having high false negative rates
Management of large thyroid nodules over 4 cm that are classified as Bethesda II, indicative of being benign, is complicated by concerns of false negatives in such cases. While the false negative rate for thyroid nodules in general is approximately 3%, the rate for large nodules over 4 cm has been reported as high as 35%.
Importantly, however, most studies evaluating the issue only involve patients who have received thyroid surgery, whereas most benign nodules are not referred for surgery.
“This may overestimate the false negative FNA biopsy risk for this group,” first author Melbin Thomas, MD, also of the University of Washington, said in her talk.
To better assess the false negative rate in the broader context of large nodules that did and did not undergo surgery, Dr. Thomas and her colleagues conducted a retrospective chart review of all patients undergoing FNA biopsy at her center between 2008 and 2014 for thyroid nodules larger than 4 cm and initially classified as Bethesda II, or benign.
With a follow-up of up to 10 years, nodules were considered accurately benign if they showed benign pathology on surgical resection, if they remained benign based on repeat FNA biopsy with Bethesda II results, or if there were no changes on imaging characteristics on ultrasound after at least 2 years.
Overall, 47 nodules over 4 cm and Bethesda II cytology were included, with an average follow-up of 5 years (range 2.2-9.7 years).
Of the nodules, 23 were treated with surgery, two of which were determined to have been malignant (8.7%) and, hence, false negatives. Nine of the nodules had repeat FNA, with none found to be malignant, and 15 received repeat ultrasound, also with no malignancies.
Overall, the false negative rate including all patients was 4.3%.
“False negative FNA biopsy results were not markedly elevated if nodules greater than 4 cm are evaluated, but rates were considerably higher if limited to surgical patients,” Dr. Thomas said.
Clinicians may be compelled to perform more aggressive surgery on large but benign thyroid nodules for a number of reasons, Dr. Tylee noted.
“A concern is that we may discontinue follow-up on these larger nodules and fail to diagnose a cancer early on, before there has been extrathyroidal extension or lymph node metastases,” she said.
In such cases, patients could wind up presenting at a higher stage of disease and require more intensive therapy.
However, with a low false negative rate overall, “all of this can increase the long-term health care costs and anxiety for patients, so having a better understanding of the true benign rate for large nodules is important,” she concluded.
Commenting on the research, Rodis D. Paparodis, MD, chief of Endocrinology, Diabetes, and Metabolism Clinics, in Patras, Greece, said the findings underscore that, as a surgical procedure, “thyroidectomy should be used cautiously, only when the benefit outweighs the risk.”
In his own previous multicenter study, Dr. Paparodis conducted a review of nearly 2,500 thyroidectomies that were performed based on size or longterm slow growth despite preoperative benign FNA findings. The results showed that only 1.9% of patients had any form of thyroid cancer in the nodule that had led to surgery; however, multiple other significant cancers were often present in other locations in the gland.
“Therefore, we suggest that careful sonographic evaluation of all thyroid nodules is warranted prior to deciding and planning the extent of surgical management for multinodular goiter,” he told this news organization.
“In addition, FNA of all suspicious nodules is required as well, to avoid unnecessary surprises in surgical pathology.”
Dr. Tylee, Dr. Thomas, and Dr. Paparodis report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
WASHINGTON – , compared with only those that were.
While more research is needed, “the risk of false negative FNA results for large nodules may not be as high as reported in previous studies if you include patients who do not have indication for surgery, such as compressive symptoms, suspicious ultrasound features, etc.,” senior author Tracy Tylee, MD, an associate professor of endocrinology at the University of Washington, Seattle, said in an interview.
The implication is that nonsurgical options such as radiofrequency ablation may be appropriate for more patients than realized, she added.
“Clinicians should consider following these patients more conservatively, either with a second FNA to confirm [the] nodule is benign or with ultrasound follow-up for 5 years with intervention only if [there are] significant changes on imaging,” she said.
The findings were presented at the annual meeting of the American Thyroid Association.
Concerns about nodules over 4 cm having high false negative rates
Management of large thyroid nodules over 4 cm that are classified as Bethesda II, indicative of being benign, is complicated by concerns of false negatives in such cases. While the false negative rate for thyroid nodules in general is approximately 3%, the rate for large nodules over 4 cm has been reported as high as 35%.
Importantly, however, most studies evaluating the issue only involve patients who have received thyroid surgery, whereas most benign nodules are not referred for surgery.
“This may overestimate the false negative FNA biopsy risk for this group,” first author Melbin Thomas, MD, also of the University of Washington, said in her talk.
To better assess the false negative rate in the broader context of large nodules that did and did not undergo surgery, Dr. Thomas and her colleagues conducted a retrospective chart review of all patients undergoing FNA biopsy at her center between 2008 and 2014 for thyroid nodules larger than 4 cm and initially classified as Bethesda II, or benign.
With a follow-up of up to 10 years, nodules were considered accurately benign if they showed benign pathology on surgical resection, if they remained benign based on repeat FNA biopsy with Bethesda II results, or if there were no changes on imaging characteristics on ultrasound after at least 2 years.
Overall, 47 nodules over 4 cm and Bethesda II cytology were included, with an average follow-up of 5 years (range 2.2-9.7 years).
Of the nodules, 23 were treated with surgery, two of which were determined to have been malignant (8.7%) and, hence, false negatives. Nine of the nodules had repeat FNA, with none found to be malignant, and 15 received repeat ultrasound, also with no malignancies.
Overall, the false negative rate including all patients was 4.3%.
“False negative FNA biopsy results were not markedly elevated if nodules greater than 4 cm are evaluated, but rates were considerably higher if limited to surgical patients,” Dr. Thomas said.
Clinicians may be compelled to perform more aggressive surgery on large but benign thyroid nodules for a number of reasons, Dr. Tylee noted.
“A concern is that we may discontinue follow-up on these larger nodules and fail to diagnose a cancer early on, before there has been extrathyroidal extension or lymph node metastases,” she said.
In such cases, patients could wind up presenting at a higher stage of disease and require more intensive therapy.
However, with a low false negative rate overall, “all of this can increase the long-term health care costs and anxiety for patients, so having a better understanding of the true benign rate for large nodules is important,” she concluded.
Commenting on the research, Rodis D. Paparodis, MD, chief of Endocrinology, Diabetes, and Metabolism Clinics, in Patras, Greece, said the findings underscore that, as a surgical procedure, “thyroidectomy should be used cautiously, only when the benefit outweighs the risk.”
In his own previous multicenter study, Dr. Paparodis conducted a review of nearly 2,500 thyroidectomies that were performed based on size or longterm slow growth despite preoperative benign FNA findings. The results showed that only 1.9% of patients had any form of thyroid cancer in the nodule that had led to surgery; however, multiple other significant cancers were often present in other locations in the gland.
“Therefore, we suggest that careful sonographic evaluation of all thyroid nodules is warranted prior to deciding and planning the extent of surgical management for multinodular goiter,” he told this news organization.
“In addition, FNA of all suspicious nodules is required as well, to avoid unnecessary surprises in surgical pathology.”
Dr. Tylee, Dr. Thomas, and Dr. Paparodis report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ATA 2023
Many young adults with type 2 diabetes skip medications
Young adults who developed type 2 diabetes as children often do not take medications to control blood pressure or cholesterol, according to a new study in JAMA Network Open. Researchers expressed alarm that young people who forgo these medications increase their chances of developing kidney disease or having a stroke.
“We’re learning more and more that those with youth onset [type 2 diabetes] really differ from those with adult onset: It looks like a more virulent form of the disease because kids are getting complications and comorbidities at much earlier ages and more severe levels,” said study author Paula Trief, PhD, a professor of psychiatry and behavioral science at State University of New York, Syracuse.
Participants in the new study were on average aged 26 years. They also had previously been part of the Treating Options for Type 2 Diabetes in Adolescents and Youth study, known as TODAY, which took place from 2004 to 2011. TODAY enrolled children between ages 10 and 17 years with type 2 diabetes who received either metformin, metformin plus rosiglitazone, or metformin plus a lifestyle intervention.
The study included extensive education and contact from medical professionals to the participants about managing diabetes.
“This cohort was followed a long time and they had a lot of support. It may be better than the real world where people haven’t had the history of this much attention,” said Lorraine Katz, MD, who specializes in endocrinology and diabetes at the Children’s Hospital of Philadelphia. Dr. Katz has enrolled participants in TODAY and published about medication adherence rates but was not part of the recent analysis.
Unannounced pill counts, addressing concerns about medication
The analysis, known as iCount, included 243 participants from the original TODAY study (159 girls) who had hypertension, neuropathy, or dyslipidemia that required ongoing medication. As the TODAY study was concluding between 2017 and 2019, researchers made unannounced phone calls to participants to request the numbers of pills they had prescribed, number of refills, and the refill date. Participants also counted aloud every pill in their possession twice.
Those phone calls continued for 3 consecutive months after iCount began and again at the same intervals 1 year later.
If the number of pills counted at a later time was at least 80% of the starting total, researchers considered this rate as low adherence. Anything less than 80% was considered high adherence.
“That’s kind of an arbitrary cutoff, but it’s one that’s used consistently in the literature” to measure medication adherence for many conditions including cancer and heart disease, Dr. Trief said. Unannounced calls to initiate pill counts were first used to understand how often people took medications for HIV, and this method was found to be a more reliable method than are self-reports.
Of 196 participants with hypertension or neuropathy, 157 (80.1%) had low adherence. And of the 146 people with high cholesterol, 137 (93.8%) had low adherence. Ninety-nine people with high cholesterol also had neuropathy or diabetes.
“This is new to the literature: We don’t really know as much about this age group,” because medication adherence studies of people who have had diabetes for more than a decade and are still in their 20s are rare, Dr. Katz said.
During the core TODAY study period, all medications were provided for free. In contrast, in the current study, participants had to obtain their prescriptions on their own. The researchers found that many participants who showed low adherence to blood pressure medications reported sometimes having trouble obtaining food (n = 62), struggling with securing stable housing (n = 47), or lacking reliable health care insurance (n = 28), all factors linked to medication adherence success, according to the analysis authors.
Researchers also assessed the impact of concerns that taking blood pressure medications may be harmful and found that people with these concerns were 37% less likely to maintain high adherence than others were by the 1-year follow-up point (odds ratio, 0.63; 95% confidence interval, 0.40-0.96; P = .01).
To some extent, the reasons people avoid medications are understandable, according to pediatric endocrinologist Tamara Hannon, MD, of Indiana University, Indianapolis.
“Rather than taking a medicine to feel better, you’re taking one not to have a problem in the future: You might not feel blood pressure, you certainly don’t feel cholesterol,” Dr. Hannon, who was not involved in the analysis, said. “Scolding them or telling them you’re going to be sorry one day doesn’t generally work.”
Dr. Hannon added that education alone about the benefits of medications does not generally drive people to adherence but that adding reminders to their phone calendar when refills are due could help. Or, the clinician could reach out to a trusted person in the patient’s life and enlist their support in taking medications consistently.
Dr. Trief advised that clinicians should carve out time for people to express their concerns about medications rather than simply writing a prescription and sending them on their way and to ask patients open-ended questions.
“If you just say to people do you have any questions, they usually say, ‘no.’ ”
No disclosures were reported.
A version of this article first appeared on Medscape.com.
Young adults who developed type 2 diabetes as children often do not take medications to control blood pressure or cholesterol, according to a new study in JAMA Network Open. Researchers expressed alarm that young people who forgo these medications increase their chances of developing kidney disease or having a stroke.
“We’re learning more and more that those with youth onset [type 2 diabetes] really differ from those with adult onset: It looks like a more virulent form of the disease because kids are getting complications and comorbidities at much earlier ages and more severe levels,” said study author Paula Trief, PhD, a professor of psychiatry and behavioral science at State University of New York, Syracuse.
Participants in the new study were on average aged 26 years. They also had previously been part of the Treating Options for Type 2 Diabetes in Adolescents and Youth study, known as TODAY, which took place from 2004 to 2011. TODAY enrolled children between ages 10 and 17 years with type 2 diabetes who received either metformin, metformin plus rosiglitazone, or metformin plus a lifestyle intervention.
The study included extensive education and contact from medical professionals to the participants about managing diabetes.
“This cohort was followed a long time and they had a lot of support. It may be better than the real world where people haven’t had the history of this much attention,” said Lorraine Katz, MD, who specializes in endocrinology and diabetes at the Children’s Hospital of Philadelphia. Dr. Katz has enrolled participants in TODAY and published about medication adherence rates but was not part of the recent analysis.
Unannounced pill counts, addressing concerns about medication
The analysis, known as iCount, included 243 participants from the original TODAY study (159 girls) who had hypertension, neuropathy, or dyslipidemia that required ongoing medication. As the TODAY study was concluding between 2017 and 2019, researchers made unannounced phone calls to participants to request the numbers of pills they had prescribed, number of refills, and the refill date. Participants also counted aloud every pill in their possession twice.
Those phone calls continued for 3 consecutive months after iCount began and again at the same intervals 1 year later.
If the number of pills counted at a later time was at least 80% of the starting total, researchers considered this rate as low adherence. Anything less than 80% was considered high adherence.
“That’s kind of an arbitrary cutoff, but it’s one that’s used consistently in the literature” to measure medication adherence for many conditions including cancer and heart disease, Dr. Trief said. Unannounced calls to initiate pill counts were first used to understand how often people took medications for HIV, and this method was found to be a more reliable method than are self-reports.
Of 196 participants with hypertension or neuropathy, 157 (80.1%) had low adherence. And of the 146 people with high cholesterol, 137 (93.8%) had low adherence. Ninety-nine people with high cholesterol also had neuropathy or diabetes.
“This is new to the literature: We don’t really know as much about this age group,” because medication adherence studies of people who have had diabetes for more than a decade and are still in their 20s are rare, Dr. Katz said.
During the core TODAY study period, all medications were provided for free. In contrast, in the current study, participants had to obtain their prescriptions on their own. The researchers found that many participants who showed low adherence to blood pressure medications reported sometimes having trouble obtaining food (n = 62), struggling with securing stable housing (n = 47), or lacking reliable health care insurance (n = 28), all factors linked to medication adherence success, according to the analysis authors.
Researchers also assessed the impact of concerns that taking blood pressure medications may be harmful and found that people with these concerns were 37% less likely to maintain high adherence than others were by the 1-year follow-up point (odds ratio, 0.63; 95% confidence interval, 0.40-0.96; P = .01).
To some extent, the reasons people avoid medications are understandable, according to pediatric endocrinologist Tamara Hannon, MD, of Indiana University, Indianapolis.
“Rather than taking a medicine to feel better, you’re taking one not to have a problem in the future: You might not feel blood pressure, you certainly don’t feel cholesterol,” Dr. Hannon, who was not involved in the analysis, said. “Scolding them or telling them you’re going to be sorry one day doesn’t generally work.”
Dr. Hannon added that education alone about the benefits of medications does not generally drive people to adherence but that adding reminders to their phone calendar when refills are due could help. Or, the clinician could reach out to a trusted person in the patient’s life and enlist their support in taking medications consistently.
Dr. Trief advised that clinicians should carve out time for people to express their concerns about medications rather than simply writing a prescription and sending them on their way and to ask patients open-ended questions.
“If you just say to people do you have any questions, they usually say, ‘no.’ ”
No disclosures were reported.
A version of this article first appeared on Medscape.com.
Young adults who developed type 2 diabetes as children often do not take medications to control blood pressure or cholesterol, according to a new study in JAMA Network Open. Researchers expressed alarm that young people who forgo these medications increase their chances of developing kidney disease or having a stroke.
“We’re learning more and more that those with youth onset [type 2 diabetes] really differ from those with adult onset: It looks like a more virulent form of the disease because kids are getting complications and comorbidities at much earlier ages and more severe levels,” said study author Paula Trief, PhD, a professor of psychiatry and behavioral science at State University of New York, Syracuse.
Participants in the new study were on average aged 26 years. They also had previously been part of the Treating Options for Type 2 Diabetes in Adolescents and Youth study, known as TODAY, which took place from 2004 to 2011. TODAY enrolled children between ages 10 and 17 years with type 2 diabetes who received either metformin, metformin plus rosiglitazone, or metformin plus a lifestyle intervention.
The study included extensive education and contact from medical professionals to the participants about managing diabetes.
“This cohort was followed a long time and they had a lot of support. It may be better than the real world where people haven’t had the history of this much attention,” said Lorraine Katz, MD, who specializes in endocrinology and diabetes at the Children’s Hospital of Philadelphia. Dr. Katz has enrolled participants in TODAY and published about medication adherence rates but was not part of the recent analysis.
Unannounced pill counts, addressing concerns about medication
The analysis, known as iCount, included 243 participants from the original TODAY study (159 girls) who had hypertension, neuropathy, or dyslipidemia that required ongoing medication. As the TODAY study was concluding between 2017 and 2019, researchers made unannounced phone calls to participants to request the numbers of pills they had prescribed, number of refills, and the refill date. Participants also counted aloud every pill in their possession twice.
Those phone calls continued for 3 consecutive months after iCount began and again at the same intervals 1 year later.
If the number of pills counted at a later time was at least 80% of the starting total, researchers considered this rate as low adherence. Anything less than 80% was considered high adherence.
“That’s kind of an arbitrary cutoff, but it’s one that’s used consistently in the literature” to measure medication adherence for many conditions including cancer and heart disease, Dr. Trief said. Unannounced calls to initiate pill counts were first used to understand how often people took medications for HIV, and this method was found to be a more reliable method than are self-reports.
Of 196 participants with hypertension or neuropathy, 157 (80.1%) had low adherence. And of the 146 people with high cholesterol, 137 (93.8%) had low adherence. Ninety-nine people with high cholesterol also had neuropathy or diabetes.
“This is new to the literature: We don’t really know as much about this age group,” because medication adherence studies of people who have had diabetes for more than a decade and are still in their 20s are rare, Dr. Katz said.
During the core TODAY study period, all medications were provided for free. In contrast, in the current study, participants had to obtain their prescriptions on their own. The researchers found that many participants who showed low adherence to blood pressure medications reported sometimes having trouble obtaining food (n = 62), struggling with securing stable housing (n = 47), or lacking reliable health care insurance (n = 28), all factors linked to medication adherence success, according to the analysis authors.
Researchers also assessed the impact of concerns that taking blood pressure medications may be harmful and found that people with these concerns were 37% less likely to maintain high adherence than others were by the 1-year follow-up point (odds ratio, 0.63; 95% confidence interval, 0.40-0.96; P = .01).
To some extent, the reasons people avoid medications are understandable, according to pediatric endocrinologist Tamara Hannon, MD, of Indiana University, Indianapolis.
“Rather than taking a medicine to feel better, you’re taking one not to have a problem in the future: You might not feel blood pressure, you certainly don’t feel cholesterol,” Dr. Hannon, who was not involved in the analysis, said. “Scolding them or telling them you’re going to be sorry one day doesn’t generally work.”
Dr. Hannon added that education alone about the benefits of medications does not generally drive people to adherence but that adding reminders to their phone calendar when refills are due could help. Or, the clinician could reach out to a trusted person in the patient’s life and enlist their support in taking medications consistently.
Dr. Trief advised that clinicians should carve out time for people to express their concerns about medications rather than simply writing a prescription and sending them on their way and to ask patients open-ended questions.
“If you just say to people do you have any questions, they usually say, ‘no.’ ”
No disclosures were reported.
A version of this article first appeared on Medscape.com.
Illicit steroids: If MDs don’t ask, patients won’t tell
Before he attended medical school, Thomas O’Connor, MD, had a not-very-well-kept secret: As a competitive powerlifter, he had used steroids to build strength.
Now an internist and clinical instructor of medicine at the University of Connecticut, Farmington, Dr. O’Connor’s practice focuses on the needs of men taking testosterone and other anabolic steroids – a group he feels is poorly understood and largely neglected by conventional medical care, perceptions borne out by a 2020 study of steroid users he helped conduct.
“They felt discriminated against, they did not feel comfortable working with their physicians, and they felt that the doctors did not know what they were doing,” Dr. O’Connor said in an interview. His patients often express anger and frustration with doctors they had seen previously.
Patients turning to home tests
Clients can order a panel of labs designed to screen for health conditions commonly associated with use of AAS, such as dyslipidemia, renal and hepatic dysfunction, polycythemia, thrombosis, and insulin resistance. The panels also include tests for levels of different hormones.
Sales of direct-to-consumer tests topped $3.6 billion in the United States in 2022 and are predicted to grow. Some of that spending is coming from people, mostly men, using illegally obtained steroids to build muscle. Although published data on the size of the bodybuilder market are unavailable, the Internet is a ready source of relatively inexpensive tests aimed at helping individuals monitor their health.
While clinicians may have their doubts about allowing patients to pick and choose tests and interpret their results, proponents claim they empower consumers to take control of their health – and save themselves money in the process.
But a test panel designed to help the user monitor the effects of banned substances is a bit unnerving to many clinicians, including Dr. O’Connor.
“People using anabolic steroids should be aware of the health risks associated with such use and that laboratory analysis is an important step toward improving health outcomes,” he said, “I’m all about open education, but not self-diagnosis and treatment.”
Testosterone and other AAS such as nandrolone, trenbolone, and boldenone are Schedule III controlled substances that have been banned by numerous athletic governing bodies. Yet recreational users can easily obtain them from online international pharmacies without a prescription. Should they be monitoring themselves for side effects?
A basic problem is that few primary care clinicians routinely ask their patients about the use of AAS or feel competent to manage the complications or withdrawal symptoms associated with the agents. And they may have no idea what the average AAS user looks like.
The American College of Sports Medicine updated its statement on the use of AAS in 2021. The statement warned of a growing new segment of users – up to 70% of people who take the drugs do so recreationally in pursuit of a more muscular appearance, rather than competitive athletes seeking enhanced performance.
The ACSM highlighted the syndrome of muscle dysmorphia, also known as “megarexia” or “bigorexia” (think of it as “reverse anorexia”), as a major risk factor for illicit use of AAS.
Stuart Phillips, PhD, professor and director of the department of kinesiology at McMaster University, Hamilton, Ont., coauthored the ACSM guidelines.
“The prince in Snow White circa 1950s was a guy with nice hair,” said Dr. Phillips, pointing to a change in cultural expectations for the male body. “But then fast forward to the prince or hero in any other Disney movie recently – and the guy is jacked.”
Since the last guidelines were published in 1987, Dr. Phillips has seen some cultural shifts. Testosterone has gone from a banned substance no one talked about to a mainstream medical therapy for men with low androgen levels, as any television viewer of primetime sports can attest. “But the other thing that’s changed,” he added, “is that we’ve seen the proliferation of illicit anabolic steroid use solely for the purpose of aesthetics.”
As an adolescent medicine physician who specializes in eating disorders, Jason Nagata, MD, MSc, sees many young men in his practice who engage in different behaviors to increase their muscle mass – from exercising, consuming high protein diets or taking protein supplements, even injecting AAS.
“A third of teenage boys across the U.S. report they’re trying to gain weight to bulk up and gain muscle,” said Dr. Nagata, an associate professor of pediatrics at the University of California, San Francisco.
In a 2020 study published in JAMA Pediatrics, Dr. Nagata and colleagues found that use of legal performance-enhancing substances in young men aged 18-26 years was associated with a higher odds of using AAS 7 years later (adjusted odds ratio, 3.18; 95% confidence interval, 1.90-5.32). “Some of the legal performance-enhancing substances like the protein powders or creatine may serve as a gateway to use of AAS,” Dr. Nagata said.
Another important factor is exposure to AAS use on social media, where muscular influencers gain huge followings. Dr. Nagata said most of the research on eating disorders and social media has examined the role of media on weight loss in girls.
“Although there’s less research on the social media impact on boys and men, a few studies have shown links between more Instagram use and muscle dissatisfaction, as well as thinking about using steroids,” he said.
The number of people using AAS is not trivial. In a longitudinal study (led by Nagata) of young U.S. adults surveyed multiple times between 1994 and 2002, a total of 2.7% of 18- to 26-year-old men and 0.4% of women reported using AAS. In a more recent cohort of adolescents in Minnesota aged 14-22 years followed between 2010 and 2018, a total of 2.2% of males and 1% of females initiated AAS use.
The Endocrine Society has estimated that between 2.9 and 4 million Americans have used an AAS at some point in their lives. Given that use is illegal without a prescription, a limitation of any survey is that participants may not be willing to disclose their AAS habit, leading to an underestimate of the actual number.
Nor are the complications of AAS use negligible. The drugs can have wide-ranging effects on the body, potentially affecting the brain, heart, liver, kidneys, musculoskeletal system, immune system, and reproductive systems. And individuals might unknowingly expose themselves to AAS: A recent literature review found that over a quarter of dietary supplements tested were found to contain undeclared substances that are on the World Anti-Doping Agency’s list of banned agents.
Alarming mistrust of MDs
Dr. O’Connor’s study shed some light on why AAS users might resort to surfing the Internet looking for a way to diagnose their own complications from steroid use. The web-based survey of nearly 2,400 men who said they took the drugs found that participants considered physicians to be the worst source of information, ranking them below coaches, online bodybuilding forums and sites, other AAS users, and bodybuilding books or magazines. The majority (56%) did not reveal AAS use to their clinicians. Of those who did, 55% reported feeling discriminated against for the admission.
Dr. O’Connor said physicians receive scant education on the many different drugs and regimens used by bodybuilders and have no idea how to a manage withdrawal syndrome for people trying to get off steroids. He urged the medical community to develop an educational campaign for clinicians, similar to those from public health officials aimed at combating the opioid epidemic: “Let’s educate med students and the residents. Let’s put [steroid use] on our agenda.”
Consumer testing evangelist ... or physician nemesis?
Nelson Vergel, BSChE, MBA, is on a mission to make medical lab testing affordable and accessible to everyone. The chemical engineer founded Discounted Labs 8 years ago, offering commonly ordered tests, such as complete blood counts, liver function tests, and cholesterol levels.
Mr. Vergel has advocated for the use of hormones to treat HIV-wasting disease for nearly 40 years, after his own diagnosis of the infection in 1986. After losing 40 pounds, steroids saved his life, he said.
Mr. Vergel said he was shocked to learn about the lack of continuing medical education on AAS for physicians and agreed with Dr. O’Connor that more training is needed for the medical profession. He also recognized that stigma on the part of clinicians is a huge barrier for many AAS users.
“We have to accept the fact that people are using them instead of demonizing them,” Mr. Vergel said. “What I was seeing is that there was so much stigma – and patients would not even talk to their doctors about their use.”
After reviewing Google analytics for his lab’s website and seeing how often “bodybuilder” came up as a search term, he added a panel of labs a year ago that allows AAS users to monitor themselves for adverse events.
Although he doesn’t condone the use of AAS without a medical indication and advises customers to discuss their results with a doctor, “we have to make sure people are reducing their harm or risk,” he said. “That’s really my goal.”
Many health care professionals would disagree with that statement. Dr. Nagata said he was concerned that management of side effects is too complicated. “There are a lot of nuances in the interpretation of these tests.” Arriving at the correct interpretation of the results requires a clinician’s thorough review of each patient’s health history, family history, and mental health history along with lab results.
‘I’m concerned’
In a second article outlining harm-reduction strategies designed to improve care for patients using AAS, Dr. O’Connor and colleagues outlined an approach for talking with patients who are concerned about their health and are seeking guidance from a clinician.
The first step is to work on developing a rapport, and not to demand that patients stop their use of AAS. His recommended opening line is: “I want to be honest with you – I’m concerned.”
The initial interaction is an opportunity to find out why the person uses AAS, what health concerns they have at present, and why they are seeking care. Open-ended questions may reveal concerns that the patient has about fertility or side effects.
Consistent with harm-reduction approaches used for other public health epidemics – such as opioid abuse and blood-borne pathogens among people who inject drugs – follow-up visits can include nonjudgmental discussions about decreasing or stopping their use.
Ultimately, minimizing the harms of AAS use can serve as a bridge to their cessation, but the medical community needs to build up trust with a community of users who currently rely more on each other and the Internet for guidance than their primary care physicians. “We need more education. We’re going to need resources to do it,” Dr. O’Connell said. “And we’re going to have to do it.”
Dr. Phillips and Dr. Nagata have no financial disclosures. Mr. Vergel is the owner and founder of Discounted Labs but reported no other financial conflicts. Dr. O’Connor owns Anabolic Doc but has no additional financial disclosures.
A version of this article first appeared on Medscape.com.
Before he attended medical school, Thomas O’Connor, MD, had a not-very-well-kept secret: As a competitive powerlifter, he had used steroids to build strength.
Now an internist and clinical instructor of medicine at the University of Connecticut, Farmington, Dr. O’Connor’s practice focuses on the needs of men taking testosterone and other anabolic steroids – a group he feels is poorly understood and largely neglected by conventional medical care, perceptions borne out by a 2020 study of steroid users he helped conduct.
“They felt discriminated against, they did not feel comfortable working with their physicians, and they felt that the doctors did not know what they were doing,” Dr. O’Connor said in an interview. His patients often express anger and frustration with doctors they had seen previously.
Patients turning to home tests
Clients can order a panel of labs designed to screen for health conditions commonly associated with use of AAS, such as dyslipidemia, renal and hepatic dysfunction, polycythemia, thrombosis, and insulin resistance. The panels also include tests for levels of different hormones.
Sales of direct-to-consumer tests topped $3.6 billion in the United States in 2022 and are predicted to grow. Some of that spending is coming from people, mostly men, using illegally obtained steroids to build muscle. Although published data on the size of the bodybuilder market are unavailable, the Internet is a ready source of relatively inexpensive tests aimed at helping individuals monitor their health.
While clinicians may have their doubts about allowing patients to pick and choose tests and interpret their results, proponents claim they empower consumers to take control of their health – and save themselves money in the process.
But a test panel designed to help the user monitor the effects of banned substances is a bit unnerving to many clinicians, including Dr. O’Connor.
“People using anabolic steroids should be aware of the health risks associated with such use and that laboratory analysis is an important step toward improving health outcomes,” he said, “I’m all about open education, but not self-diagnosis and treatment.”
Testosterone and other AAS such as nandrolone, trenbolone, and boldenone are Schedule III controlled substances that have been banned by numerous athletic governing bodies. Yet recreational users can easily obtain them from online international pharmacies without a prescription. Should they be monitoring themselves for side effects?
A basic problem is that few primary care clinicians routinely ask their patients about the use of AAS or feel competent to manage the complications or withdrawal symptoms associated with the agents. And they may have no idea what the average AAS user looks like.
The American College of Sports Medicine updated its statement on the use of AAS in 2021. The statement warned of a growing new segment of users – up to 70% of people who take the drugs do so recreationally in pursuit of a more muscular appearance, rather than competitive athletes seeking enhanced performance.
The ACSM highlighted the syndrome of muscle dysmorphia, also known as “megarexia” or “bigorexia” (think of it as “reverse anorexia”), as a major risk factor for illicit use of AAS.
Stuart Phillips, PhD, professor and director of the department of kinesiology at McMaster University, Hamilton, Ont., coauthored the ACSM guidelines.
“The prince in Snow White circa 1950s was a guy with nice hair,” said Dr. Phillips, pointing to a change in cultural expectations for the male body. “But then fast forward to the prince or hero in any other Disney movie recently – and the guy is jacked.”
Since the last guidelines were published in 1987, Dr. Phillips has seen some cultural shifts. Testosterone has gone from a banned substance no one talked about to a mainstream medical therapy for men with low androgen levels, as any television viewer of primetime sports can attest. “But the other thing that’s changed,” he added, “is that we’ve seen the proliferation of illicit anabolic steroid use solely for the purpose of aesthetics.”
As an adolescent medicine physician who specializes in eating disorders, Jason Nagata, MD, MSc, sees many young men in his practice who engage in different behaviors to increase their muscle mass – from exercising, consuming high protein diets or taking protein supplements, even injecting AAS.
“A third of teenage boys across the U.S. report they’re trying to gain weight to bulk up and gain muscle,” said Dr. Nagata, an associate professor of pediatrics at the University of California, San Francisco.
In a 2020 study published in JAMA Pediatrics, Dr. Nagata and colleagues found that use of legal performance-enhancing substances in young men aged 18-26 years was associated with a higher odds of using AAS 7 years later (adjusted odds ratio, 3.18; 95% confidence interval, 1.90-5.32). “Some of the legal performance-enhancing substances like the protein powders or creatine may serve as a gateway to use of AAS,” Dr. Nagata said.
Another important factor is exposure to AAS use on social media, where muscular influencers gain huge followings. Dr. Nagata said most of the research on eating disorders and social media has examined the role of media on weight loss in girls.
“Although there’s less research on the social media impact on boys and men, a few studies have shown links between more Instagram use and muscle dissatisfaction, as well as thinking about using steroids,” he said.
The number of people using AAS is not trivial. In a longitudinal study (led by Nagata) of young U.S. adults surveyed multiple times between 1994 and 2002, a total of 2.7% of 18- to 26-year-old men and 0.4% of women reported using AAS. In a more recent cohort of adolescents in Minnesota aged 14-22 years followed between 2010 and 2018, a total of 2.2% of males and 1% of females initiated AAS use.
The Endocrine Society has estimated that between 2.9 and 4 million Americans have used an AAS at some point in their lives. Given that use is illegal without a prescription, a limitation of any survey is that participants may not be willing to disclose their AAS habit, leading to an underestimate of the actual number.
Nor are the complications of AAS use negligible. The drugs can have wide-ranging effects on the body, potentially affecting the brain, heart, liver, kidneys, musculoskeletal system, immune system, and reproductive systems. And individuals might unknowingly expose themselves to AAS: A recent literature review found that over a quarter of dietary supplements tested were found to contain undeclared substances that are on the World Anti-Doping Agency’s list of banned agents.
Alarming mistrust of MDs
Dr. O’Connor’s study shed some light on why AAS users might resort to surfing the Internet looking for a way to diagnose their own complications from steroid use. The web-based survey of nearly 2,400 men who said they took the drugs found that participants considered physicians to be the worst source of information, ranking them below coaches, online bodybuilding forums and sites, other AAS users, and bodybuilding books or magazines. The majority (56%) did not reveal AAS use to their clinicians. Of those who did, 55% reported feeling discriminated against for the admission.
Dr. O’Connor said physicians receive scant education on the many different drugs and regimens used by bodybuilders and have no idea how to a manage withdrawal syndrome for people trying to get off steroids. He urged the medical community to develop an educational campaign for clinicians, similar to those from public health officials aimed at combating the opioid epidemic: “Let’s educate med students and the residents. Let’s put [steroid use] on our agenda.”
Consumer testing evangelist ... or physician nemesis?
Nelson Vergel, BSChE, MBA, is on a mission to make medical lab testing affordable and accessible to everyone. The chemical engineer founded Discounted Labs 8 years ago, offering commonly ordered tests, such as complete blood counts, liver function tests, and cholesterol levels.
Mr. Vergel has advocated for the use of hormones to treat HIV-wasting disease for nearly 40 years, after his own diagnosis of the infection in 1986. After losing 40 pounds, steroids saved his life, he said.
Mr. Vergel said he was shocked to learn about the lack of continuing medical education on AAS for physicians and agreed with Dr. O’Connor that more training is needed for the medical profession. He also recognized that stigma on the part of clinicians is a huge barrier for many AAS users.
“We have to accept the fact that people are using them instead of demonizing them,” Mr. Vergel said. “What I was seeing is that there was so much stigma – and patients would not even talk to their doctors about their use.”
After reviewing Google analytics for his lab’s website and seeing how often “bodybuilder” came up as a search term, he added a panel of labs a year ago that allows AAS users to monitor themselves for adverse events.
Although he doesn’t condone the use of AAS without a medical indication and advises customers to discuss their results with a doctor, “we have to make sure people are reducing their harm or risk,” he said. “That’s really my goal.”
Many health care professionals would disagree with that statement. Dr. Nagata said he was concerned that management of side effects is too complicated. “There are a lot of nuances in the interpretation of these tests.” Arriving at the correct interpretation of the results requires a clinician’s thorough review of each patient’s health history, family history, and mental health history along with lab results.
‘I’m concerned’
In a second article outlining harm-reduction strategies designed to improve care for patients using AAS, Dr. O’Connor and colleagues outlined an approach for talking with patients who are concerned about their health and are seeking guidance from a clinician.
The first step is to work on developing a rapport, and not to demand that patients stop their use of AAS. His recommended opening line is: “I want to be honest with you – I’m concerned.”
The initial interaction is an opportunity to find out why the person uses AAS, what health concerns they have at present, and why they are seeking care. Open-ended questions may reveal concerns that the patient has about fertility or side effects.
Consistent with harm-reduction approaches used for other public health epidemics – such as opioid abuse and blood-borne pathogens among people who inject drugs – follow-up visits can include nonjudgmental discussions about decreasing or stopping their use.
Ultimately, minimizing the harms of AAS use can serve as a bridge to their cessation, but the medical community needs to build up trust with a community of users who currently rely more on each other and the Internet for guidance than their primary care physicians. “We need more education. We’re going to need resources to do it,” Dr. O’Connell said. “And we’re going to have to do it.”
Dr. Phillips and Dr. Nagata have no financial disclosures. Mr. Vergel is the owner and founder of Discounted Labs but reported no other financial conflicts. Dr. O’Connor owns Anabolic Doc but has no additional financial disclosures.
A version of this article first appeared on Medscape.com.
Before he attended medical school, Thomas O’Connor, MD, had a not-very-well-kept secret: As a competitive powerlifter, he had used steroids to build strength.
Now an internist and clinical instructor of medicine at the University of Connecticut, Farmington, Dr. O’Connor’s practice focuses on the needs of men taking testosterone and other anabolic steroids – a group he feels is poorly understood and largely neglected by conventional medical care, perceptions borne out by a 2020 study of steroid users he helped conduct.
“They felt discriminated against, they did not feel comfortable working with their physicians, and they felt that the doctors did not know what they were doing,” Dr. O’Connor said in an interview. His patients often express anger and frustration with doctors they had seen previously.
Patients turning to home tests
Clients can order a panel of labs designed to screen for health conditions commonly associated with use of AAS, such as dyslipidemia, renal and hepatic dysfunction, polycythemia, thrombosis, and insulin resistance. The panels also include tests for levels of different hormones.
Sales of direct-to-consumer tests topped $3.6 billion in the United States in 2022 and are predicted to grow. Some of that spending is coming from people, mostly men, using illegally obtained steroids to build muscle. Although published data on the size of the bodybuilder market are unavailable, the Internet is a ready source of relatively inexpensive tests aimed at helping individuals monitor their health.
While clinicians may have their doubts about allowing patients to pick and choose tests and interpret their results, proponents claim they empower consumers to take control of their health – and save themselves money in the process.
But a test panel designed to help the user monitor the effects of banned substances is a bit unnerving to many clinicians, including Dr. O’Connor.
“People using anabolic steroids should be aware of the health risks associated with such use and that laboratory analysis is an important step toward improving health outcomes,” he said, “I’m all about open education, but not self-diagnosis and treatment.”
Testosterone and other AAS such as nandrolone, trenbolone, and boldenone are Schedule III controlled substances that have been banned by numerous athletic governing bodies. Yet recreational users can easily obtain them from online international pharmacies without a prescription. Should they be monitoring themselves for side effects?
A basic problem is that few primary care clinicians routinely ask their patients about the use of AAS or feel competent to manage the complications or withdrawal symptoms associated with the agents. And they may have no idea what the average AAS user looks like.
The American College of Sports Medicine updated its statement on the use of AAS in 2021. The statement warned of a growing new segment of users – up to 70% of people who take the drugs do so recreationally in pursuit of a more muscular appearance, rather than competitive athletes seeking enhanced performance.
The ACSM highlighted the syndrome of muscle dysmorphia, also known as “megarexia” or “bigorexia” (think of it as “reverse anorexia”), as a major risk factor for illicit use of AAS.
Stuart Phillips, PhD, professor and director of the department of kinesiology at McMaster University, Hamilton, Ont., coauthored the ACSM guidelines.
“The prince in Snow White circa 1950s was a guy with nice hair,” said Dr. Phillips, pointing to a change in cultural expectations for the male body. “But then fast forward to the prince or hero in any other Disney movie recently – and the guy is jacked.”
Since the last guidelines were published in 1987, Dr. Phillips has seen some cultural shifts. Testosterone has gone from a banned substance no one talked about to a mainstream medical therapy for men with low androgen levels, as any television viewer of primetime sports can attest. “But the other thing that’s changed,” he added, “is that we’ve seen the proliferation of illicit anabolic steroid use solely for the purpose of aesthetics.”
As an adolescent medicine physician who specializes in eating disorders, Jason Nagata, MD, MSc, sees many young men in his practice who engage in different behaviors to increase their muscle mass – from exercising, consuming high protein diets or taking protein supplements, even injecting AAS.
“A third of teenage boys across the U.S. report they’re trying to gain weight to bulk up and gain muscle,” said Dr. Nagata, an associate professor of pediatrics at the University of California, San Francisco.
In a 2020 study published in JAMA Pediatrics, Dr. Nagata and colleagues found that use of legal performance-enhancing substances in young men aged 18-26 years was associated with a higher odds of using AAS 7 years later (adjusted odds ratio, 3.18; 95% confidence interval, 1.90-5.32). “Some of the legal performance-enhancing substances like the protein powders or creatine may serve as a gateway to use of AAS,” Dr. Nagata said.
Another important factor is exposure to AAS use on social media, where muscular influencers gain huge followings. Dr. Nagata said most of the research on eating disorders and social media has examined the role of media on weight loss in girls.
“Although there’s less research on the social media impact on boys and men, a few studies have shown links between more Instagram use and muscle dissatisfaction, as well as thinking about using steroids,” he said.
The number of people using AAS is not trivial. In a longitudinal study (led by Nagata) of young U.S. adults surveyed multiple times between 1994 and 2002, a total of 2.7% of 18- to 26-year-old men and 0.4% of women reported using AAS. In a more recent cohort of adolescents in Minnesota aged 14-22 years followed between 2010 and 2018, a total of 2.2% of males and 1% of females initiated AAS use.
The Endocrine Society has estimated that between 2.9 and 4 million Americans have used an AAS at some point in their lives. Given that use is illegal without a prescription, a limitation of any survey is that participants may not be willing to disclose their AAS habit, leading to an underestimate of the actual number.
Nor are the complications of AAS use negligible. The drugs can have wide-ranging effects on the body, potentially affecting the brain, heart, liver, kidneys, musculoskeletal system, immune system, and reproductive systems. And individuals might unknowingly expose themselves to AAS: A recent literature review found that over a quarter of dietary supplements tested were found to contain undeclared substances that are on the World Anti-Doping Agency’s list of banned agents.
Alarming mistrust of MDs
Dr. O’Connor’s study shed some light on why AAS users might resort to surfing the Internet looking for a way to diagnose their own complications from steroid use. The web-based survey of nearly 2,400 men who said they took the drugs found that participants considered physicians to be the worst source of information, ranking them below coaches, online bodybuilding forums and sites, other AAS users, and bodybuilding books or magazines. The majority (56%) did not reveal AAS use to their clinicians. Of those who did, 55% reported feeling discriminated against for the admission.
Dr. O’Connor said physicians receive scant education on the many different drugs and regimens used by bodybuilders and have no idea how to a manage withdrawal syndrome for people trying to get off steroids. He urged the medical community to develop an educational campaign for clinicians, similar to those from public health officials aimed at combating the opioid epidemic: “Let’s educate med students and the residents. Let’s put [steroid use] on our agenda.”
Consumer testing evangelist ... or physician nemesis?
Nelson Vergel, BSChE, MBA, is on a mission to make medical lab testing affordable and accessible to everyone. The chemical engineer founded Discounted Labs 8 years ago, offering commonly ordered tests, such as complete blood counts, liver function tests, and cholesterol levels.
Mr. Vergel has advocated for the use of hormones to treat HIV-wasting disease for nearly 40 years, after his own diagnosis of the infection in 1986. After losing 40 pounds, steroids saved his life, he said.
Mr. Vergel said he was shocked to learn about the lack of continuing medical education on AAS for physicians and agreed with Dr. O’Connor that more training is needed for the medical profession. He also recognized that stigma on the part of clinicians is a huge barrier for many AAS users.
“We have to accept the fact that people are using them instead of demonizing them,” Mr. Vergel said. “What I was seeing is that there was so much stigma – and patients would not even talk to their doctors about their use.”
After reviewing Google analytics for his lab’s website and seeing how often “bodybuilder” came up as a search term, he added a panel of labs a year ago that allows AAS users to monitor themselves for adverse events.
Although he doesn’t condone the use of AAS without a medical indication and advises customers to discuss their results with a doctor, “we have to make sure people are reducing their harm or risk,” he said. “That’s really my goal.”
Many health care professionals would disagree with that statement. Dr. Nagata said he was concerned that management of side effects is too complicated. “There are a lot of nuances in the interpretation of these tests.” Arriving at the correct interpretation of the results requires a clinician’s thorough review of each patient’s health history, family history, and mental health history along with lab results.
‘I’m concerned’
In a second article outlining harm-reduction strategies designed to improve care for patients using AAS, Dr. O’Connor and colleagues outlined an approach for talking with patients who are concerned about their health and are seeking guidance from a clinician.
The first step is to work on developing a rapport, and not to demand that patients stop their use of AAS. His recommended opening line is: “I want to be honest with you – I’m concerned.”
The initial interaction is an opportunity to find out why the person uses AAS, what health concerns they have at present, and why they are seeking care. Open-ended questions may reveal concerns that the patient has about fertility or side effects.
Consistent with harm-reduction approaches used for other public health epidemics – such as opioid abuse and blood-borne pathogens among people who inject drugs – follow-up visits can include nonjudgmental discussions about decreasing or stopping their use.
Ultimately, minimizing the harms of AAS use can serve as a bridge to their cessation, but the medical community needs to build up trust with a community of users who currently rely more on each other and the Internet for guidance than their primary care physicians. “We need more education. We’re going to need resources to do it,” Dr. O’Connell said. “And we’re going to have to do it.”
Dr. Phillips and Dr. Nagata have no financial disclosures. Mr. Vergel is the owner and founder of Discounted Labs but reported no other financial conflicts. Dr. O’Connor owns Anabolic Doc but has no additional financial disclosures.
A version of this article first appeared on Medscape.com.
Redefining CVD risk: Cardiovascular-kidney-metabolic (CKM) syndrome
“This work was prompted by the fact that CKM syndrome leads to premature morbidity and mortality, primarily because of a higher burden of CVD,” writing committee chair Chiadi Ndumele, MD, PhD, said in an interview.
“While CKM syndrome is a public health emergency, there is also great potential for improving CKM health in the population, with an increasing number of therapies that favorably impact metabolic risk factors, risk for adverse kidney events, or both, which also protect against CVD,” added Dr. Ndumele, director of obesity and cardiometabolic research in the division of cardiology at Johns Hopkins University, Baltimore.
The AHA presidential advisory and accompanying scientific statement, which provides a synopsis of evidence for the science and clinical management of CKM, were published online in the journal Circulation.
CKM syndrome staging
According to the AHA, one in three U.S. adults have three or more risk factors that contribute to CVD, metabolic disorders, and/or kidney disease.
In addition to defining CKM syndrome, the advisory provides a “staging construct, to be used in both adults and youth, that reflects the progressive pathophysiology and risk within CKM syndrome, with therapeutic guidance tied to CKM stages,” Dr. Ndumele told this news organization.
The AHA outlines four stages of CKM syndrome:
Stage 0: At this stage, no CKM risk factors are present, and the goal is to prevent CKM syndrome (particularly unhealthy weight gain) by achieving and maintaining ideal health based on the AHA’s Life’s Essential 8 recommendations. Adults in this stage should be screened every 3-5 years to assess lipids, blood pressure, and blood sugar.
Stage 1: At this stage, excess weight, abdominal obesity, or dysfunctional adipose tissue (clinically manifest as impaired glucose tolerance or prediabetes) is present without other metabolic risk factors or CVD. Management includes providing support for healthy lifestyle changes (healthy eating and regular physical activity), with a goal of at least 5% weight loss and addressing glucose intolerance if needed. Screening adults with stage 1 CKM every 2-3 years is advised to assess blood pressure, triglycerides, cholesterol, and blood sugar.
Stage 2: At this stage, metabolic risk factors (hypertriglyceridemia, hypertension, metabolic syndrome, diabetes) and kidney disease are present. The goal is to address risk factors to prevent progression to CVD and kidney failure. Screening for stage 2 CKM syndrome aligns with AHA/ACC guidelines, which include yearly assessment of blood pressure, triglycerides, cholesterol, blood sugar, and kidney function. More frequent kidney screening is recommended for individuals with increased risk of kidney failure based on kidney function assessments.
Stage 3: This stage describes individuals with subclinical CVD with metabolic risk factors or kidney disease or those at high predicted risk for CVD. The goal is to intensify efforts to prevent progression to symptomatic CVD and kidney failure. This may involve increasing or changing medications, and additional focus on lifestyle changes. Coronary artery calcium (CAC) measurement in some adults is recommended to assess narrowing of the arteries when treatment decisions are unclear.
Stage 4: Individuals with stage 4 CKM syndrome have symptomatic CVD, excess body fat, metabolic risk factors, or kidney disease. Stage 4 CKM syndrome is divided into two subcategories: (4a) no kidney failure and (4b) kidney failure. In this stage, patients may have already had a myocardial infarction (MI) or stroke or may already have heart failure. They also may have additional CV conditions such as peripheral artery disease or atrial fibrillation. The goal of care is individualized treatment for CVD with consideration for CKM syndrome conditions.
The advisory also describes CKM syndrome regression, “an important concept and public health message in which people making healthy lifestyle changes and achieving weight loss may regress to lower CKM syndrome stages and a better state of health,” the AHA says in a news release.
They note that a “critical” next step is to update the pooled cohort equation (PCE) risk prediction algorithm to include measures of kidney function, type 2 diabetes control, and social determinants of health for a more comprehensive risk estimate.
The advisory also recommends risk calculator updates be expanded to assess risk in people as young as age 30 and to calculate both 10- and 30-year CVD risk.
“Clearly defining the patient with CKM syndrome, and providing new approaches for CKM syndrome staging and risk prediction, will help health care professionals to identify these individuals earlier and to provide timely, holistic, and patient-centered care,” Dr. Ndumele said.
This presidential advisory was prepared by the volunteer writing group on behalf of the AHA . The authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
“This work was prompted by the fact that CKM syndrome leads to premature morbidity and mortality, primarily because of a higher burden of CVD,” writing committee chair Chiadi Ndumele, MD, PhD, said in an interview.
“While CKM syndrome is a public health emergency, there is also great potential for improving CKM health in the population, with an increasing number of therapies that favorably impact metabolic risk factors, risk for adverse kidney events, or both, which also protect against CVD,” added Dr. Ndumele, director of obesity and cardiometabolic research in the division of cardiology at Johns Hopkins University, Baltimore.
The AHA presidential advisory and accompanying scientific statement, which provides a synopsis of evidence for the science and clinical management of CKM, were published online in the journal Circulation.
CKM syndrome staging
According to the AHA, one in three U.S. adults have three or more risk factors that contribute to CVD, metabolic disorders, and/or kidney disease.
In addition to defining CKM syndrome, the advisory provides a “staging construct, to be used in both adults and youth, that reflects the progressive pathophysiology and risk within CKM syndrome, with therapeutic guidance tied to CKM stages,” Dr. Ndumele told this news organization.
The AHA outlines four stages of CKM syndrome:
Stage 0: At this stage, no CKM risk factors are present, and the goal is to prevent CKM syndrome (particularly unhealthy weight gain) by achieving and maintaining ideal health based on the AHA’s Life’s Essential 8 recommendations. Adults in this stage should be screened every 3-5 years to assess lipids, blood pressure, and blood sugar.
Stage 1: At this stage, excess weight, abdominal obesity, or dysfunctional adipose tissue (clinically manifest as impaired glucose tolerance or prediabetes) is present without other metabolic risk factors or CVD. Management includes providing support for healthy lifestyle changes (healthy eating and regular physical activity), with a goal of at least 5% weight loss and addressing glucose intolerance if needed. Screening adults with stage 1 CKM every 2-3 years is advised to assess blood pressure, triglycerides, cholesterol, and blood sugar.
Stage 2: At this stage, metabolic risk factors (hypertriglyceridemia, hypertension, metabolic syndrome, diabetes) and kidney disease are present. The goal is to address risk factors to prevent progression to CVD and kidney failure. Screening for stage 2 CKM syndrome aligns with AHA/ACC guidelines, which include yearly assessment of blood pressure, triglycerides, cholesterol, blood sugar, and kidney function. More frequent kidney screening is recommended for individuals with increased risk of kidney failure based on kidney function assessments.
Stage 3: This stage describes individuals with subclinical CVD with metabolic risk factors or kidney disease or those at high predicted risk for CVD. The goal is to intensify efforts to prevent progression to symptomatic CVD and kidney failure. This may involve increasing or changing medications, and additional focus on lifestyle changes. Coronary artery calcium (CAC) measurement in some adults is recommended to assess narrowing of the arteries when treatment decisions are unclear.
Stage 4: Individuals with stage 4 CKM syndrome have symptomatic CVD, excess body fat, metabolic risk factors, or kidney disease. Stage 4 CKM syndrome is divided into two subcategories: (4a) no kidney failure and (4b) kidney failure. In this stage, patients may have already had a myocardial infarction (MI) or stroke or may already have heart failure. They also may have additional CV conditions such as peripheral artery disease or atrial fibrillation. The goal of care is individualized treatment for CVD with consideration for CKM syndrome conditions.
The advisory also describes CKM syndrome regression, “an important concept and public health message in which people making healthy lifestyle changes and achieving weight loss may regress to lower CKM syndrome stages and a better state of health,” the AHA says in a news release.
They note that a “critical” next step is to update the pooled cohort equation (PCE) risk prediction algorithm to include measures of kidney function, type 2 diabetes control, and social determinants of health for a more comprehensive risk estimate.
The advisory also recommends risk calculator updates be expanded to assess risk in people as young as age 30 and to calculate both 10- and 30-year CVD risk.
“Clearly defining the patient with CKM syndrome, and providing new approaches for CKM syndrome staging and risk prediction, will help health care professionals to identify these individuals earlier and to provide timely, holistic, and patient-centered care,” Dr. Ndumele said.
This presidential advisory was prepared by the volunteer writing group on behalf of the AHA . The authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
“This work was prompted by the fact that CKM syndrome leads to premature morbidity and mortality, primarily because of a higher burden of CVD,” writing committee chair Chiadi Ndumele, MD, PhD, said in an interview.
“While CKM syndrome is a public health emergency, there is also great potential for improving CKM health in the population, with an increasing number of therapies that favorably impact metabolic risk factors, risk for adverse kidney events, or both, which also protect against CVD,” added Dr. Ndumele, director of obesity and cardiometabolic research in the division of cardiology at Johns Hopkins University, Baltimore.
The AHA presidential advisory and accompanying scientific statement, which provides a synopsis of evidence for the science and clinical management of CKM, were published online in the journal Circulation.
CKM syndrome staging
According to the AHA, one in three U.S. adults have three or more risk factors that contribute to CVD, metabolic disorders, and/or kidney disease.
In addition to defining CKM syndrome, the advisory provides a “staging construct, to be used in both adults and youth, that reflects the progressive pathophysiology and risk within CKM syndrome, with therapeutic guidance tied to CKM stages,” Dr. Ndumele told this news organization.
The AHA outlines four stages of CKM syndrome:
Stage 0: At this stage, no CKM risk factors are present, and the goal is to prevent CKM syndrome (particularly unhealthy weight gain) by achieving and maintaining ideal health based on the AHA’s Life’s Essential 8 recommendations. Adults in this stage should be screened every 3-5 years to assess lipids, blood pressure, and blood sugar.
Stage 1: At this stage, excess weight, abdominal obesity, or dysfunctional adipose tissue (clinically manifest as impaired glucose tolerance or prediabetes) is present without other metabolic risk factors or CVD. Management includes providing support for healthy lifestyle changes (healthy eating and regular physical activity), with a goal of at least 5% weight loss and addressing glucose intolerance if needed. Screening adults with stage 1 CKM every 2-3 years is advised to assess blood pressure, triglycerides, cholesterol, and blood sugar.
Stage 2: At this stage, metabolic risk factors (hypertriglyceridemia, hypertension, metabolic syndrome, diabetes) and kidney disease are present. The goal is to address risk factors to prevent progression to CVD and kidney failure. Screening for stage 2 CKM syndrome aligns with AHA/ACC guidelines, which include yearly assessment of blood pressure, triglycerides, cholesterol, blood sugar, and kidney function. More frequent kidney screening is recommended for individuals with increased risk of kidney failure based on kidney function assessments.
Stage 3: This stage describes individuals with subclinical CVD with metabolic risk factors or kidney disease or those at high predicted risk for CVD. The goal is to intensify efforts to prevent progression to symptomatic CVD and kidney failure. This may involve increasing or changing medications, and additional focus on lifestyle changes. Coronary artery calcium (CAC) measurement in some adults is recommended to assess narrowing of the arteries when treatment decisions are unclear.
Stage 4: Individuals with stage 4 CKM syndrome have symptomatic CVD, excess body fat, metabolic risk factors, or kidney disease. Stage 4 CKM syndrome is divided into two subcategories: (4a) no kidney failure and (4b) kidney failure. In this stage, patients may have already had a myocardial infarction (MI) or stroke or may already have heart failure. They also may have additional CV conditions such as peripheral artery disease or atrial fibrillation. The goal of care is individualized treatment for CVD with consideration for CKM syndrome conditions.
The advisory also describes CKM syndrome regression, “an important concept and public health message in which people making healthy lifestyle changes and achieving weight loss may regress to lower CKM syndrome stages and a better state of health,” the AHA says in a news release.
They note that a “critical” next step is to update the pooled cohort equation (PCE) risk prediction algorithm to include measures of kidney function, type 2 diabetes control, and social determinants of health for a more comprehensive risk estimate.
The advisory also recommends risk calculator updates be expanded to assess risk in people as young as age 30 and to calculate both 10- and 30-year CVD risk.
“Clearly defining the patient with CKM syndrome, and providing new approaches for CKM syndrome staging and risk prediction, will help health care professionals to identify these individuals earlier and to provide timely, holistic, and patient-centered care,” Dr. Ndumele said.
This presidential advisory was prepared by the volunteer writing group on behalf of the AHA . The authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CIRCULATION
Every click you make, the EHR is watching you
This transcript has been edited for clarity.
When I close my eyes and imagine what it is I do for a living, I see a computer screen.
I’m primarily a clinical researcher, so much of what I do is looking at statistical software, or, more recently, writing grant applications. But even when I think of my clinical duties, I see that computer screen.
The reason? The electronic health record (EHR) – the hot, beating heart of medical care in the modern era. Our most powerful tool and our greatest enemy.
The EHR records everything – not just the vital signs and lab values of our patients, not just our notes and billing codes. Everything. Every interaction we have is tracked and can be analyzed. The EHR is basically Sting in the song “Every Breath You Take.” Every click you make, it is watching you.
Researchers are leveraging that panopticon to give insight into something we don’t talk about frequently: the issue of racial bias in medicine. Is our true nature revealed by our interactions with the EHR?
We’re talking about this study in JAMA Network Open.
Researchers leveraged huge amounts of EHR data from two big academic medical centers, Vanderbilt University Medical Center and Northwestern University Medical Center. All told, there are data from nearly 250,000 hospitalizations here.
The researchers created a metric for EHR engagement. Basically, they summed the amount of clicks and other EHR interactions that occurred during the hospitalization, divided by the length of stay in days, to create a sort of average “engagement per day” metric. This number was categorized into four groups: low engagement, medium engagement, high engagement, and very high engagement.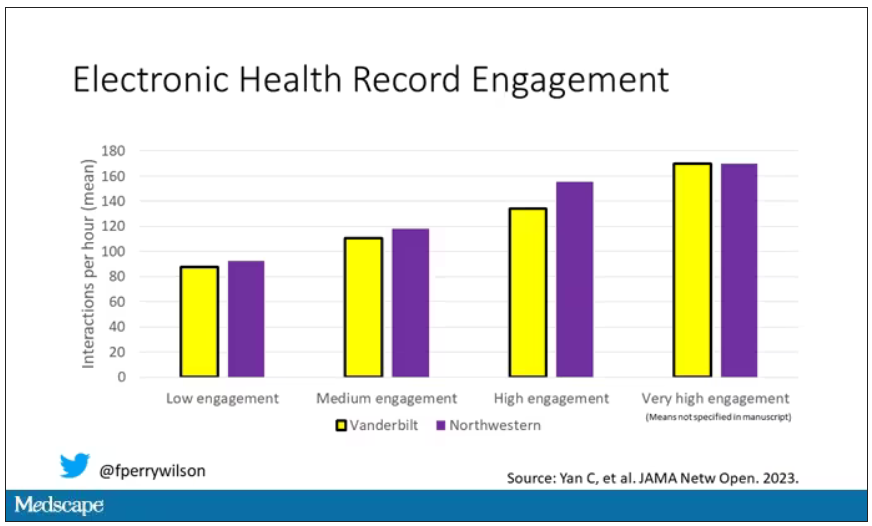
What factors would predict higher engagement? Well, , except among Black patients who actually got a bit more engagement.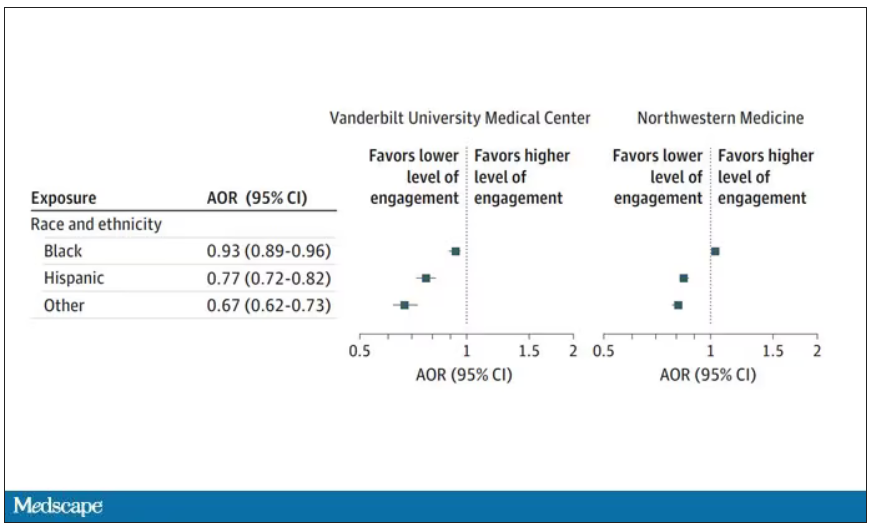
So, right away we need to be concerned about the obvious implications. Less engagement with the EHR may mean lower-quality care, right? Less attention to medical issues. And if that differs systematically by race, that’s a problem.
But we need to be careful here, because engagement in the health record is not random. Many factors would lead you to spend more time in one patient’s chart vs. another. Medical complexity is the most obvious one. The authors did their best to account for this, adjusting for patients’ age, sex, insurance status, comorbidity score, and social deprivation index based on their ZIP code. But notably, they did not account for the acuity of illness during the hospitalization. If individuals identifying as a minority were, all else being equal, less likely to be severely ill by the time they were hospitalized, you might see results like this.
The authors also restrict their analysis to individuals who were discharged alive. I’m not entirely clear why they made this choice. Most people don’t die in the hospital; the inpatient mortality rate at most centers is 1%-1.5%. But excluding those patients could potentially bias these results, especially if race is, all else being equal, a predictor of inpatient mortality, as some studies have shown.
But the truth is, these data aren’t coming out of nowhere; they don’t exist in a vacuum. Numerous studies demonstrate different intensity of care among minority vs. nonminority individuals. There is this study, which shows that minority populations are less likely to be placed on the liver transplant waitlist.
There is this study, which found that minority kids with type 1 diabetes were less likely to get insulin pumps than were their White counterparts. And this one, which showed that kids with acute appendicitis were less likely to get pain-control medications if they were Black.
This study shows that although life expectancy decreased across all races during the pandemic, it decreased the most among minority populations.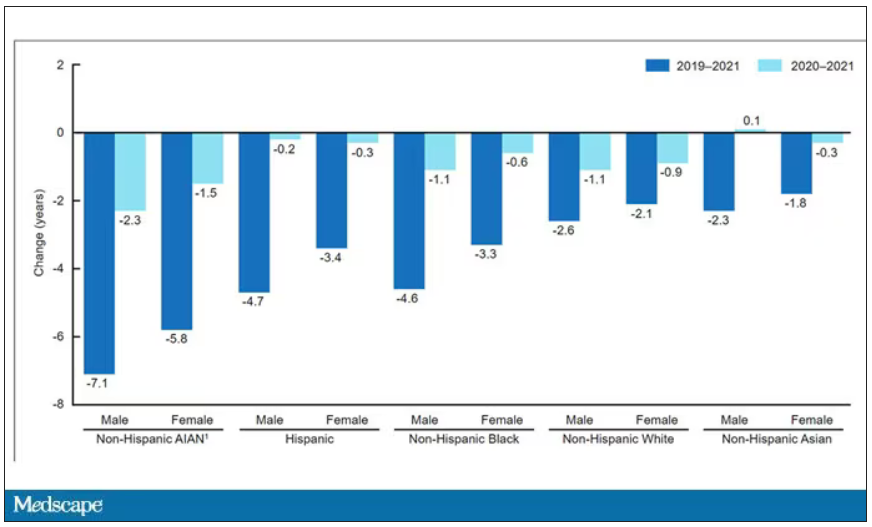
This list goes on. It’s why the CDC has called racism a “fundamental cause of ... disease.”
So, yes, it is clear that there are racial disparities in health care outcomes. It is clear that there are racial disparities in treatments. It is also clear that virtually every physician believes they deliver equitable care. Somewhere, this disconnect arises. Could the actions we take in the EHR reveal the unconscious biases we have? Does the all-seeing eye of the EHR see not only into our brains but into our hearts? And if it can, are we ready to confront what it sees?
F. Perry Wilson, MD, MSCE, is associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator in New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
When I close my eyes and imagine what it is I do for a living, I see a computer screen.
I’m primarily a clinical researcher, so much of what I do is looking at statistical software, or, more recently, writing grant applications. But even when I think of my clinical duties, I see that computer screen.
The reason? The electronic health record (EHR) – the hot, beating heart of medical care in the modern era. Our most powerful tool and our greatest enemy.
The EHR records everything – not just the vital signs and lab values of our patients, not just our notes and billing codes. Everything. Every interaction we have is tracked and can be analyzed. The EHR is basically Sting in the song “Every Breath You Take.” Every click you make, it is watching you.
Researchers are leveraging that panopticon to give insight into something we don’t talk about frequently: the issue of racial bias in medicine. Is our true nature revealed by our interactions with the EHR?
We’re talking about this study in JAMA Network Open.
Researchers leveraged huge amounts of EHR data from two big academic medical centers, Vanderbilt University Medical Center and Northwestern University Medical Center. All told, there are data from nearly 250,000 hospitalizations here.
The researchers created a metric for EHR engagement. Basically, they summed the amount of clicks and other EHR interactions that occurred during the hospitalization, divided by the length of stay in days, to create a sort of average “engagement per day” metric. This number was categorized into four groups: low engagement, medium engagement, high engagement, and very high engagement.
What factors would predict higher engagement? Well, , except among Black patients who actually got a bit more engagement.
So, right away we need to be concerned about the obvious implications. Less engagement with the EHR may mean lower-quality care, right? Less attention to medical issues. And if that differs systematically by race, that’s a problem.
But we need to be careful here, because engagement in the health record is not random. Many factors would lead you to spend more time in one patient’s chart vs. another. Medical complexity is the most obvious one. The authors did their best to account for this, adjusting for patients’ age, sex, insurance status, comorbidity score, and social deprivation index based on their ZIP code. But notably, they did not account for the acuity of illness during the hospitalization. If individuals identifying as a minority were, all else being equal, less likely to be severely ill by the time they were hospitalized, you might see results like this.
The authors also restrict their analysis to individuals who were discharged alive. I’m not entirely clear why they made this choice. Most people don’t die in the hospital; the inpatient mortality rate at most centers is 1%-1.5%. But excluding those patients could potentially bias these results, especially if race is, all else being equal, a predictor of inpatient mortality, as some studies have shown.
But the truth is, these data aren’t coming out of nowhere; they don’t exist in a vacuum. Numerous studies demonstrate different intensity of care among minority vs. nonminority individuals. There is this study, which shows that minority populations are less likely to be placed on the liver transplant waitlist.
There is this study, which found that minority kids with type 1 diabetes were less likely to get insulin pumps than were their White counterparts. And this one, which showed that kids with acute appendicitis were less likely to get pain-control medications if they were Black.
This study shows that although life expectancy decreased across all races during the pandemic, it decreased the most among minority populations.
This list goes on. It’s why the CDC has called racism a “fundamental cause of ... disease.”
So, yes, it is clear that there are racial disparities in health care outcomes. It is clear that there are racial disparities in treatments. It is also clear that virtually every physician believes they deliver equitable care. Somewhere, this disconnect arises. Could the actions we take in the EHR reveal the unconscious biases we have? Does the all-seeing eye of the EHR see not only into our brains but into our hearts? And if it can, are we ready to confront what it sees?
F. Perry Wilson, MD, MSCE, is associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator in New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
When I close my eyes and imagine what it is I do for a living, I see a computer screen.
I’m primarily a clinical researcher, so much of what I do is looking at statistical software, or, more recently, writing grant applications. But even when I think of my clinical duties, I see that computer screen.
The reason? The electronic health record (EHR) – the hot, beating heart of medical care in the modern era. Our most powerful tool and our greatest enemy.
The EHR records everything – not just the vital signs and lab values of our patients, not just our notes and billing codes. Everything. Every interaction we have is tracked and can be analyzed. The EHR is basically Sting in the song “Every Breath You Take.” Every click you make, it is watching you.
Researchers are leveraging that panopticon to give insight into something we don’t talk about frequently: the issue of racial bias in medicine. Is our true nature revealed by our interactions with the EHR?
We’re talking about this study in JAMA Network Open.
Researchers leveraged huge amounts of EHR data from two big academic medical centers, Vanderbilt University Medical Center and Northwestern University Medical Center. All told, there are data from nearly 250,000 hospitalizations here.
The researchers created a metric for EHR engagement. Basically, they summed the amount of clicks and other EHR interactions that occurred during the hospitalization, divided by the length of stay in days, to create a sort of average “engagement per day” metric. This number was categorized into four groups: low engagement, medium engagement, high engagement, and very high engagement.
What factors would predict higher engagement? Well, , except among Black patients who actually got a bit more engagement.
So, right away we need to be concerned about the obvious implications. Less engagement with the EHR may mean lower-quality care, right? Less attention to medical issues. And if that differs systematically by race, that’s a problem.
But we need to be careful here, because engagement in the health record is not random. Many factors would lead you to spend more time in one patient’s chart vs. another. Medical complexity is the most obvious one. The authors did their best to account for this, adjusting for patients’ age, sex, insurance status, comorbidity score, and social deprivation index based on their ZIP code. But notably, they did not account for the acuity of illness during the hospitalization. If individuals identifying as a minority were, all else being equal, less likely to be severely ill by the time they were hospitalized, you might see results like this.
The authors also restrict their analysis to individuals who were discharged alive. I’m not entirely clear why they made this choice. Most people don’t die in the hospital; the inpatient mortality rate at most centers is 1%-1.5%. But excluding those patients could potentially bias these results, especially if race is, all else being equal, a predictor of inpatient mortality, as some studies have shown.
But the truth is, these data aren’t coming out of nowhere; they don’t exist in a vacuum. Numerous studies demonstrate different intensity of care among minority vs. nonminority individuals. There is this study, which shows that minority populations are less likely to be placed on the liver transplant waitlist.
There is this study, which found that minority kids with type 1 diabetes were less likely to get insulin pumps than were their White counterparts. And this one, which showed that kids with acute appendicitis were less likely to get pain-control medications if they were Black.
This study shows that although life expectancy decreased across all races during the pandemic, it decreased the most among minority populations.
This list goes on. It’s why the CDC has called racism a “fundamental cause of ... disease.”
So, yes, it is clear that there are racial disparities in health care outcomes. It is clear that there are racial disparities in treatments. It is also clear that virtually every physician believes they deliver equitable care. Somewhere, this disconnect arises. Could the actions we take in the EHR reveal the unconscious biases we have? Does the all-seeing eye of the EHR see not only into our brains but into our hearts? And if it can, are we ready to confront what it sees?
F. Perry Wilson, MD, MSCE, is associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator in New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
New hyperglycemia emergency guidance updates DKA definition
HAMBURG, GERMANY – along with many other updates to the last statement on the topic, published 14 years ago.
Based on extensive literature reviews and observations of current trends, the new document – due to be published soon – will cover diagnosis and management of the two most serious acute hyperglycemic emergencies seen in adults, DKA and hyperosmolar hyperglycemic state (HHS).
New to the 2023 version will be a strong emphasis on the excess morbidity and mortality risks associated with the increasingly common “hybrid” presentation of the two conditions together, now seen in about a third of cases.
The new report will also more strongly urge clinicians to investigate why the person experienced the emergency.
While new-onset diabetes and infection are recognized precipitating causes for DKA, insulin omission related to finances, mental health, and social determinants should be identified, and patients directed to appropriate resources, said experts previewing the upcoming new report at the annual meeting of the European Association for the Study of Diabetes.
“The challenge is, although we were making progress for a long time in terms of those hyperglycemic crises, we’ve really plateaued and there are still people being admitted in large numbers, and when you look more globally even more so,” said American Diabetes Association Chief Science and Medical Officer Robert A. Gabbay, MD, PhD.
The new consensus report will be jointly endorsed by the ADA, the EASD, the American Association of Clinical Endocrinology, the Diabetes Technology Society, and the Joint British Diabetes Societies for Inpatient Care. The previous consensus statement on the subject was published in 2009 by the ADA alone.
New DKA and HHS definitions reflect emerging trends
The statement will revise the definition of DKA, partly spurred by the increasing occurrence and recognition of euglycemic ketoacidosis arising from the use of sodium-glucose cotransporter 2 (SGLT2) inhibitors. For all patients with hyperglycemic crisis, the hyperglycemia cutoff is now lowered to 200 mg/dL (11.1 mmol/L) from the previous 250 mg/dL.
However, the glucose cutoff has been removed entirely for people with a history of diabetes.
“Both of these changes are recognizing the wide range of glucose levels at the presence of DKA. Approximately 10% of DKA occurs with euglycemia or near-normoglycemia,” noted coauthor Shivani Misra, MD, PhD, senior clinical lecturer and honorary consultant in Metabolic Medicine at Imperial College, London.
For assessing ketosis in DKA, the new statement strongly recommends use of beta-hydroxybutyrate – either via point-of-care test or serum level measured in a laboratory – with a low cutoff of ≥ 3.0 mmol/L. Alternatively, a urine ketone strip value of 2+ or greater can be used.
However, beta-hydroxybutyrate testing is more widely available now than it was in 2009 and is strongly preferred over urine ketone measurement because it’s the predominant ketone during acidosis. Moreover, urine acetoacetate – measured by the strips – paradoxically increases during resolution of DKA, and drug interferences can occur with urine ketone measurement, Dr. Misra noted.
Metabolic acidosis is now defined as a pH < 7.3 and/or a bicarbonate concentration < 18 mmol/L, up from 15 in some prior guidelines including the United Kingdom’s. Also, anion gap has been removed from the main definition but, the document will say, can still be used in settings where ketone testing is unavailable.
As previously, the new statement will classify DKA by mild, moderate, and severe but now for the first time there are recommendations of care for each of those levels, as well as for HHS.
For HHS, the glucose cutoff of ≥ 600 mg/dL will stay the same. But now, the effective serum osmolality has been lowered from > 320 to > 300 mOsml/L to account for the effect of dehydration, along with an alternative criteria of total serum osmolality > 320 mOsm/L. The same two changes as with DKA for both ketones and acidosis have also been included for HHS.
Asked to comment, session audience member and independent diabetes industry consultant Charles Alexander, MD, told this news organization, “I liked the proposal to eliminate the anion gap in decision-making and to focus on measurement of blood ketones, principally beta-hydroxybutyrate, in the diagnosis of DKA and monitoring the effect of treatment.
“If someone is on an SGLT2 inhibitor, there is no need to look at blood glucose levels, which may be normal or near normal in the setting of DKA.”
But Dr. Alexander thinks that they should have eliminated glucose levels entirely as part of the DKA/HHS definition even for people without diabetes.
“The problem is that medical education for many years has taught us that DKA is a condition of high blood glucose, but it may not be. It is good that they said blood glucose levels were not important if the patient had a history of diabetes. However, a glucose of 200mg/dL may not be low enough if someone is on an SGLT2 inhibitor. There needs to be a much lower threshold for measuring blood ketones in anyone with nausea, vomiting, and abdominal pain, regardless of the blood glucose level.”
Acute management: IV fluids, insulin, and potassium
Like the 2009 statement, the new one will include detailed management flowcharts for DKA and HHS, but this time in color. This new statement includes individual algorithms for management with intravenous fluids, insulin, and potassium. Bicarbonate has been removed and relegated to a note at the bottom saying that it should only be considered if pH is < 7.0.
Under fluid treatment, the new statement offers more information about using crystalloids to treat dehydration and a recommendation to add dextrose to IV fluid therapy as a substrate when the glucose drops below 250 mg/dL, in order to prevent hypoglycemia. For euglycemic DKA, the recommendation is to include dextrose and normal saline simultaneously.
And for the first time, subcutaneous rather than IV insulin is considered acceptable for mild, but not moderate or severe, DKA.
Two options are suggested for IV insulin in HHS: The fluid can be given first and low-dose fixed-rate insulin infusion added, or fluids and insulin can be given at the same time.
Criteria for resolution of DKA are a venous pH of ≥ 7.3 or bicarbonate > 18 mmol/L, ketones < 0.6 mmol/L, and glucose ideally < 200 mg/dL (11.0 mmol/L). For HHS, resolution is suggested when the measured or calculated serum osmolality falls to < 300 mosm/kg, blood glucose is < 250mg/dL (13.9 mmol/L), urine output > 0.5 mL/kg/hour, and cognitive status is improved.
The statement also will provide detailed recommended options for transitioning from IV to subcutaneous insulin, but defers to clinical judgment for deciding when the patient can be discharged. The initiation or continuation of SGLT2 inhibitors is not recommended at any time during hospitalization for hyperglycemic crises.
Mitigating complications, preventing recurrence
In addition to listing potential complications of treating hyperglycemic crises, just as the 2009 statement did, the new one will offer mitigation strategies for some of the more common ones. For preventing hypoglycemia, frequent blood glucose monitoring is advised along with adding dextrose to the IV fluids when glucose drops below 250 mg/dL.
For prevention of hypokalemia, which occurs in about half of patients treated for DKA and HHS, the statement recommends potassium monitoring every 4 hours and replacement added to fluids.
Acute kidney injury, also occurring in about half of people treated for DKA and/or HHS, usually resolves with hydration. Daily renal function monitoring is advised.
Preventing recurrence: Many factors beyond clinical
Prevention of recurrence with readmission for DKA and/or HHS, occurring in up to 22% of U.S. patients within 30 days, entails close follow-up within 2-4 weeks after discharge (including via telemedicine), and assessment of possible causes, including mental health disorders and social determinants of health.
Appropriate education should be provided, including “structured education” involving problem-solving, sick day rules, injection techniques, a review of insulin doses, consideration of continuous glucose monitoring (CGM), and home ketone testing.
Patients should be provided with an adequate supply of insulin and durable diabetes equipment, along with contact information for health care professionals who can assist them. Social service professionals can be helpful for patients who lack reliable access.
Dr. Gabbay told this news organization, “The eye-opening thing is we tend to typically think of DKA as how people tend to get diagnosed with diabetes and, yes, that’s true, but that’s only a minority of people. Those might be preventable by early screening, but all these other people and the number of recurrent episodes, that’s an area where it’s really a failure of the system where we can do better in ensuring that doesn’t happen.”
Education is only part of it, he stressed. “It’s not just an intelligence thing. It’s social factors, and there can be complex psychological issues and mental health issues. We need to screen for those things when we see someone coming back the second, third, fifth, or sixth time. We’ve all seen that. Just educating them to take their insulin is not the answer. …You’ve got to ask the questions and engage them to go a little deeper.”
Dr. Gabbay is an employee of the ADA. Dr. Alexander has reported being a nonpaid advisor for diaTribe and a consultant for Kinexum. Dr. Misra has received speaker fees from Sanofi and ABCD and an investigator-initiated research grant from Dexcom, and is a trustee for the Diabetes Research and Wellness Foundation in the United Kingdom.
A version of this article appeared on Medscape.com.
HAMBURG, GERMANY – along with many other updates to the last statement on the topic, published 14 years ago.
Based on extensive literature reviews and observations of current trends, the new document – due to be published soon – will cover diagnosis and management of the two most serious acute hyperglycemic emergencies seen in adults, DKA and hyperosmolar hyperglycemic state (HHS).
New to the 2023 version will be a strong emphasis on the excess morbidity and mortality risks associated with the increasingly common “hybrid” presentation of the two conditions together, now seen in about a third of cases.
The new report will also more strongly urge clinicians to investigate why the person experienced the emergency.
While new-onset diabetes and infection are recognized precipitating causes for DKA, insulin omission related to finances, mental health, and social determinants should be identified, and patients directed to appropriate resources, said experts previewing the upcoming new report at the annual meeting of the European Association for the Study of Diabetes.
“The challenge is, although we were making progress for a long time in terms of those hyperglycemic crises, we’ve really plateaued and there are still people being admitted in large numbers, and when you look more globally even more so,” said American Diabetes Association Chief Science and Medical Officer Robert A. Gabbay, MD, PhD.
The new consensus report will be jointly endorsed by the ADA, the EASD, the American Association of Clinical Endocrinology, the Diabetes Technology Society, and the Joint British Diabetes Societies for Inpatient Care. The previous consensus statement on the subject was published in 2009 by the ADA alone.
New DKA and HHS definitions reflect emerging trends
The statement will revise the definition of DKA, partly spurred by the increasing occurrence and recognition of euglycemic ketoacidosis arising from the use of sodium-glucose cotransporter 2 (SGLT2) inhibitors. For all patients with hyperglycemic crisis, the hyperglycemia cutoff is now lowered to 200 mg/dL (11.1 mmol/L) from the previous 250 mg/dL.
However, the glucose cutoff has been removed entirely for people with a history of diabetes.
“Both of these changes are recognizing the wide range of glucose levels at the presence of DKA. Approximately 10% of DKA occurs with euglycemia or near-normoglycemia,” noted coauthor Shivani Misra, MD, PhD, senior clinical lecturer and honorary consultant in Metabolic Medicine at Imperial College, London.
For assessing ketosis in DKA, the new statement strongly recommends use of beta-hydroxybutyrate – either via point-of-care test or serum level measured in a laboratory – with a low cutoff of ≥ 3.0 mmol/L. Alternatively, a urine ketone strip value of 2+ or greater can be used.
However, beta-hydroxybutyrate testing is more widely available now than it was in 2009 and is strongly preferred over urine ketone measurement because it’s the predominant ketone during acidosis. Moreover, urine acetoacetate – measured by the strips – paradoxically increases during resolution of DKA, and drug interferences can occur with urine ketone measurement, Dr. Misra noted.
Metabolic acidosis is now defined as a pH < 7.3 and/or a bicarbonate concentration < 18 mmol/L, up from 15 in some prior guidelines including the United Kingdom’s. Also, anion gap has been removed from the main definition but, the document will say, can still be used in settings where ketone testing is unavailable.
As previously, the new statement will classify DKA by mild, moderate, and severe but now for the first time there are recommendations of care for each of those levels, as well as for HHS.
For HHS, the glucose cutoff of ≥ 600 mg/dL will stay the same. But now, the effective serum osmolality has been lowered from > 320 to > 300 mOsml/L to account for the effect of dehydration, along with an alternative criteria of total serum osmolality > 320 mOsm/L. The same two changes as with DKA for both ketones and acidosis have also been included for HHS.
Asked to comment, session audience member and independent diabetes industry consultant Charles Alexander, MD, told this news organization, “I liked the proposal to eliminate the anion gap in decision-making and to focus on measurement of blood ketones, principally beta-hydroxybutyrate, in the diagnosis of DKA and monitoring the effect of treatment.
“If someone is on an SGLT2 inhibitor, there is no need to look at blood glucose levels, which may be normal or near normal in the setting of DKA.”
But Dr. Alexander thinks that they should have eliminated glucose levels entirely as part of the DKA/HHS definition even for people without diabetes.
“The problem is that medical education for many years has taught us that DKA is a condition of high blood glucose, but it may not be. It is good that they said blood glucose levels were not important if the patient had a history of diabetes. However, a glucose of 200mg/dL may not be low enough if someone is on an SGLT2 inhibitor. There needs to be a much lower threshold for measuring blood ketones in anyone with nausea, vomiting, and abdominal pain, regardless of the blood glucose level.”
Acute management: IV fluids, insulin, and potassium
Like the 2009 statement, the new one will include detailed management flowcharts for DKA and HHS, but this time in color. This new statement includes individual algorithms for management with intravenous fluids, insulin, and potassium. Bicarbonate has been removed and relegated to a note at the bottom saying that it should only be considered if pH is < 7.0.
Under fluid treatment, the new statement offers more information about using crystalloids to treat dehydration and a recommendation to add dextrose to IV fluid therapy as a substrate when the glucose drops below 250 mg/dL, in order to prevent hypoglycemia. For euglycemic DKA, the recommendation is to include dextrose and normal saline simultaneously.
And for the first time, subcutaneous rather than IV insulin is considered acceptable for mild, but not moderate or severe, DKA.
Two options are suggested for IV insulin in HHS: The fluid can be given first and low-dose fixed-rate insulin infusion added, or fluids and insulin can be given at the same time.
Criteria for resolution of DKA are a venous pH of ≥ 7.3 or bicarbonate > 18 mmol/L, ketones < 0.6 mmol/L, and glucose ideally < 200 mg/dL (11.0 mmol/L). For HHS, resolution is suggested when the measured or calculated serum osmolality falls to < 300 mosm/kg, blood glucose is < 250mg/dL (13.9 mmol/L), urine output > 0.5 mL/kg/hour, and cognitive status is improved.
The statement also will provide detailed recommended options for transitioning from IV to subcutaneous insulin, but defers to clinical judgment for deciding when the patient can be discharged. The initiation or continuation of SGLT2 inhibitors is not recommended at any time during hospitalization for hyperglycemic crises.
Mitigating complications, preventing recurrence
In addition to listing potential complications of treating hyperglycemic crises, just as the 2009 statement did, the new one will offer mitigation strategies for some of the more common ones. For preventing hypoglycemia, frequent blood glucose monitoring is advised along with adding dextrose to the IV fluids when glucose drops below 250 mg/dL.
For prevention of hypokalemia, which occurs in about half of patients treated for DKA and HHS, the statement recommends potassium monitoring every 4 hours and replacement added to fluids.
Acute kidney injury, also occurring in about half of people treated for DKA and/or HHS, usually resolves with hydration. Daily renal function monitoring is advised.
Preventing recurrence: Many factors beyond clinical
Prevention of recurrence with readmission for DKA and/or HHS, occurring in up to 22% of U.S. patients within 30 days, entails close follow-up within 2-4 weeks after discharge (including via telemedicine), and assessment of possible causes, including mental health disorders and social determinants of health.
Appropriate education should be provided, including “structured education” involving problem-solving, sick day rules, injection techniques, a review of insulin doses, consideration of continuous glucose monitoring (CGM), and home ketone testing.
Patients should be provided with an adequate supply of insulin and durable diabetes equipment, along with contact information for health care professionals who can assist them. Social service professionals can be helpful for patients who lack reliable access.
Dr. Gabbay told this news organization, “The eye-opening thing is we tend to typically think of DKA as how people tend to get diagnosed with diabetes and, yes, that’s true, but that’s only a minority of people. Those might be preventable by early screening, but all these other people and the number of recurrent episodes, that’s an area where it’s really a failure of the system where we can do better in ensuring that doesn’t happen.”
Education is only part of it, he stressed. “It’s not just an intelligence thing. It’s social factors, and there can be complex psychological issues and mental health issues. We need to screen for those things when we see someone coming back the second, third, fifth, or sixth time. We’ve all seen that. Just educating them to take their insulin is not the answer. …You’ve got to ask the questions and engage them to go a little deeper.”
Dr. Gabbay is an employee of the ADA. Dr. Alexander has reported being a nonpaid advisor for diaTribe and a consultant for Kinexum. Dr. Misra has received speaker fees from Sanofi and ABCD and an investigator-initiated research grant from Dexcom, and is a trustee for the Diabetes Research and Wellness Foundation in the United Kingdom.
A version of this article appeared on Medscape.com.
HAMBURG, GERMANY – along with many other updates to the last statement on the topic, published 14 years ago.
Based on extensive literature reviews and observations of current trends, the new document – due to be published soon – will cover diagnosis and management of the two most serious acute hyperglycemic emergencies seen in adults, DKA and hyperosmolar hyperglycemic state (HHS).
New to the 2023 version will be a strong emphasis on the excess morbidity and mortality risks associated with the increasingly common “hybrid” presentation of the two conditions together, now seen in about a third of cases.
The new report will also more strongly urge clinicians to investigate why the person experienced the emergency.
While new-onset diabetes and infection are recognized precipitating causes for DKA, insulin omission related to finances, mental health, and social determinants should be identified, and patients directed to appropriate resources, said experts previewing the upcoming new report at the annual meeting of the European Association for the Study of Diabetes.
“The challenge is, although we were making progress for a long time in terms of those hyperglycemic crises, we’ve really plateaued and there are still people being admitted in large numbers, and when you look more globally even more so,” said American Diabetes Association Chief Science and Medical Officer Robert A. Gabbay, MD, PhD.
The new consensus report will be jointly endorsed by the ADA, the EASD, the American Association of Clinical Endocrinology, the Diabetes Technology Society, and the Joint British Diabetes Societies for Inpatient Care. The previous consensus statement on the subject was published in 2009 by the ADA alone.
New DKA and HHS definitions reflect emerging trends
The statement will revise the definition of DKA, partly spurred by the increasing occurrence and recognition of euglycemic ketoacidosis arising from the use of sodium-glucose cotransporter 2 (SGLT2) inhibitors. For all patients with hyperglycemic crisis, the hyperglycemia cutoff is now lowered to 200 mg/dL (11.1 mmol/L) from the previous 250 mg/dL.
However, the glucose cutoff has been removed entirely for people with a history of diabetes.
“Both of these changes are recognizing the wide range of glucose levels at the presence of DKA. Approximately 10% of DKA occurs with euglycemia or near-normoglycemia,” noted coauthor Shivani Misra, MD, PhD, senior clinical lecturer and honorary consultant in Metabolic Medicine at Imperial College, London.
For assessing ketosis in DKA, the new statement strongly recommends use of beta-hydroxybutyrate – either via point-of-care test or serum level measured in a laboratory – with a low cutoff of ≥ 3.0 mmol/L. Alternatively, a urine ketone strip value of 2+ or greater can be used.
However, beta-hydroxybutyrate testing is more widely available now than it was in 2009 and is strongly preferred over urine ketone measurement because it’s the predominant ketone during acidosis. Moreover, urine acetoacetate – measured by the strips – paradoxically increases during resolution of DKA, and drug interferences can occur with urine ketone measurement, Dr. Misra noted.
Metabolic acidosis is now defined as a pH < 7.3 and/or a bicarbonate concentration < 18 mmol/L, up from 15 in some prior guidelines including the United Kingdom’s. Also, anion gap has been removed from the main definition but, the document will say, can still be used in settings where ketone testing is unavailable.
As previously, the new statement will classify DKA by mild, moderate, and severe but now for the first time there are recommendations of care for each of those levels, as well as for HHS.
For HHS, the glucose cutoff of ≥ 600 mg/dL will stay the same. But now, the effective serum osmolality has been lowered from > 320 to > 300 mOsml/L to account for the effect of dehydration, along with an alternative criteria of total serum osmolality > 320 mOsm/L. The same two changes as with DKA for both ketones and acidosis have also been included for HHS.
Asked to comment, session audience member and independent diabetes industry consultant Charles Alexander, MD, told this news organization, “I liked the proposal to eliminate the anion gap in decision-making and to focus on measurement of blood ketones, principally beta-hydroxybutyrate, in the diagnosis of DKA and monitoring the effect of treatment.
“If someone is on an SGLT2 inhibitor, there is no need to look at blood glucose levels, which may be normal or near normal in the setting of DKA.”
But Dr. Alexander thinks that they should have eliminated glucose levels entirely as part of the DKA/HHS definition even for people without diabetes.
“The problem is that medical education for many years has taught us that DKA is a condition of high blood glucose, but it may not be. It is good that they said blood glucose levels were not important if the patient had a history of diabetes. However, a glucose of 200mg/dL may not be low enough if someone is on an SGLT2 inhibitor. There needs to be a much lower threshold for measuring blood ketones in anyone with nausea, vomiting, and abdominal pain, regardless of the blood glucose level.”
Acute management: IV fluids, insulin, and potassium
Like the 2009 statement, the new one will include detailed management flowcharts for DKA and HHS, but this time in color. This new statement includes individual algorithms for management with intravenous fluids, insulin, and potassium. Bicarbonate has been removed and relegated to a note at the bottom saying that it should only be considered if pH is < 7.0.
Under fluid treatment, the new statement offers more information about using crystalloids to treat dehydration and a recommendation to add dextrose to IV fluid therapy as a substrate when the glucose drops below 250 mg/dL, in order to prevent hypoglycemia. For euglycemic DKA, the recommendation is to include dextrose and normal saline simultaneously.
And for the first time, subcutaneous rather than IV insulin is considered acceptable for mild, but not moderate or severe, DKA.
Two options are suggested for IV insulin in HHS: The fluid can be given first and low-dose fixed-rate insulin infusion added, or fluids and insulin can be given at the same time.
Criteria for resolution of DKA are a venous pH of ≥ 7.3 or bicarbonate > 18 mmol/L, ketones < 0.6 mmol/L, and glucose ideally < 200 mg/dL (11.0 mmol/L). For HHS, resolution is suggested when the measured or calculated serum osmolality falls to < 300 mosm/kg, blood glucose is < 250mg/dL (13.9 mmol/L), urine output > 0.5 mL/kg/hour, and cognitive status is improved.
The statement also will provide detailed recommended options for transitioning from IV to subcutaneous insulin, but defers to clinical judgment for deciding when the patient can be discharged. The initiation or continuation of SGLT2 inhibitors is not recommended at any time during hospitalization for hyperglycemic crises.
Mitigating complications, preventing recurrence
In addition to listing potential complications of treating hyperglycemic crises, just as the 2009 statement did, the new one will offer mitigation strategies for some of the more common ones. For preventing hypoglycemia, frequent blood glucose monitoring is advised along with adding dextrose to the IV fluids when glucose drops below 250 mg/dL.
For prevention of hypokalemia, which occurs in about half of patients treated for DKA and HHS, the statement recommends potassium monitoring every 4 hours and replacement added to fluids.
Acute kidney injury, also occurring in about half of people treated for DKA and/or HHS, usually resolves with hydration. Daily renal function monitoring is advised.
Preventing recurrence: Many factors beyond clinical
Prevention of recurrence with readmission for DKA and/or HHS, occurring in up to 22% of U.S. patients within 30 days, entails close follow-up within 2-4 weeks after discharge (including via telemedicine), and assessment of possible causes, including mental health disorders and social determinants of health.
Appropriate education should be provided, including “structured education” involving problem-solving, sick day rules, injection techniques, a review of insulin doses, consideration of continuous glucose monitoring (CGM), and home ketone testing.
Patients should be provided with an adequate supply of insulin and durable diabetes equipment, along with contact information for health care professionals who can assist them. Social service professionals can be helpful for patients who lack reliable access.
Dr. Gabbay told this news organization, “The eye-opening thing is we tend to typically think of DKA as how people tend to get diagnosed with diabetes and, yes, that’s true, but that’s only a minority of people. Those might be preventable by early screening, but all these other people and the number of recurrent episodes, that’s an area where it’s really a failure of the system where we can do better in ensuring that doesn’t happen.”
Education is only part of it, he stressed. “It’s not just an intelligence thing. It’s social factors, and there can be complex psychological issues and mental health issues. We need to screen for those things when we see someone coming back the second, third, fifth, or sixth time. We’ve all seen that. Just educating them to take their insulin is not the answer. …You’ve got to ask the questions and engage them to go a little deeper.”
Dr. Gabbay is an employee of the ADA. Dr. Alexander has reported being a nonpaid advisor for diaTribe and a consultant for Kinexum. Dr. Misra has received speaker fees from Sanofi and ABCD and an investigator-initiated research grant from Dexcom, and is a trustee for the Diabetes Research and Wellness Foundation in the United Kingdom.
A version of this article appeared on Medscape.com.
AT EASD 2023
Semaglutide win in HFpEF with obesity regardless of ejection fraction: STEP-HFpEF
CLEVELAND – independently of baseline left-ventricular ejection fraction (LVEF).
The finding comes from a prespecified secondary analysis of the STEP-HFpEF trial of more than 500 nondiabetic patients with obesity and HF with an initial LVEF of 45% or greater.
They suggest that for patients with the obesity phenotype of HFpEF, semaglutide (Wegovy) could potentially join SGLT2 inhibitors on the short list of meds with consistent treatment effects whether LVEF is mildly reduced, preserved, or in the normal range.
That would distinguish the drug, a glucagon-like peptide-1 (GLP-1) receptor agonist, from mineralocorticoid receptor antagonists (MRA), sacubitril-valsartan (Entresto), and other renin-angiotensin-system inhibitors (RASi), whose benefits tend to taper off with rising LVEF.
The patients assigned to semaglutide showed significant improvement in both primary endpoints – change in Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ-CSS) and change in body weight at 52 weeks – whether their baseline LVEF was 45%-49%, 50%-59%, or 60% or greater.
Results were similar for improvements in 6-minute walk distance (6MWD) and levels of NT-terminal pro–brain natriuretic peptide (NT-proBNP) and C-reactive protein, observed Javed Butler, MD, when presenting the analysis at the annual meeting of the Heart Failure Society of America, Cleveland.
Dr. Butler, of Baylor Scott and White Research Institute, Dallas, and the University of Mississippi, Jackson, is also lead author of the study, which was published on the same day in the Journal of the American College of Cardiology.
In his presentation, Dr. Butler singled out the NT-proBNP finding as “very meaningful” with respect to understanding potential mechanisms of the drug effects observed in the trial.
For example, people with obesity tend to have lower than average natriuretic peptide levels that “actually go up a bit” when they lose weight, he observed. But in the trial, “we saw a reduction in NT-proBNP in spite of the weight loss,” regardless of LVEF category.
John McMurray, MD, University of Glasgow, the invited discussant for Dr. Butler’s presentation, agreed that it raises the question whether weight loss was the sole semaglutide effect responsible for the improvement in heart failure status and biomarkers. The accompanying NT-proBNP reductions – when the opposite might otherwise have been expected – may point to a possible mechanism of action that is “something more than just weight loss,” he said. “If that were the case, it becomes very important, because it means that this treatment might do good things in non-obese patients or might do good things in patients with other types of heart failure.”
‘Vital reassurance’
More definitive trials are needed “to clarify safety and efficacy of obesity-targeted therapeutics in HF across the ejection fraction spectrum,” according to an accompanying editorial).
Still, the STEP-HFpEF analysis “strengthens the role of GLP-1 [receptor agonists] to ameliorate health status” for patients with obesity and HF with mildly reduced or preserved ejection fraction, write Muthiah Vaduganathan, MD, MPH, and John W. Ostrominski, MD, Brigham and Women’s Hospital and Harvard Medical School, both in Boston.
Its findings “provide vital reassurance” on semaglutide safety and efficacy in HF with below-normal LVEF and “tentatively support the existence of a more general, LVEF-independent, obesity-related HF phenotype capable of favorable modification with incretin-based therapies.”
The lack of heterogeneity in treatment effects across LVEF subgroups “is not surprising,” but “the findings reinforce that the benefits of this therapy in those meeting trial criteria do not vary by left ventricular ejection fraction,” Gregg C. Fonarow, MD, University of California, Los Angeles, Medical Center, said in an interview.
It remains unknown, however, “whether the improvement in health status, functional status, and reduced inflammation” will translate to reduced risk of cardiovascular death or HF hospitalization, said Dr. Fonarow, who isn’t connected to STEP-HFpEF.
It’s a question for future studies, he agreed, whether semaglutide would confer similar benefits for patients with obesity and HF with LVEF less than 45% or in non-obese HF patients.
Dr. McMurray proposed that future GLP-1 receptor agonist heart-failure trials should include non-obese patients to determine whether the effects seen in STEP-HFpEF were due to something more than weight loss. Trials in patients with obesity and HF with reduced LVEF would also be important.
“If it turns out just to be about weight loss, then we need to think about the alternatives,” including diet, exercise, and bariatric surgery but also, potentially, weight-loss drugs other than semaglutide, he said.
No heterogeneity by LVEF
STEP-HFpEF randomly assigned 529 patients free of diabetes with an LVEF greater than or equal to 45%, a body mass index (BMI) of at least 30 kg/m2, and NYHA functional status of 2-4 to either a placebo injection or 2.4-mg semaglutide subcutaneously once a week (the dose used for weight reduction) atop standard care.
As previously reported, those assigned to semaglutide showed significant improvements at 1 year in symptoms and in physical limitation, per changes in KCCQ-CSS, and weight loss, compared with the control group. Their exercise capacity, as measured by 6MWD, also improved.
The more weight patients lost while taking semaglutide, the better their KCCQ-CSS and 6MWD outcomes, a prior secondary analysis suggested. But the STEP-HFpEF researchers said weight loss did not appear to explain all of their gains, compared with usual care.
For the current analysis, the 263 patients assigned to receive semaglutide and 266 control patients were divided into three groups by baseline LVEF and compared for the same outcomes.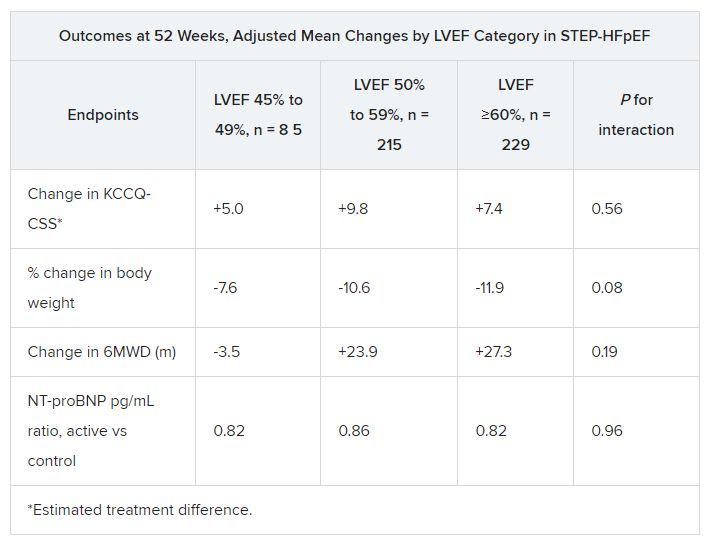
The semaglutide group, compared with control patients, also showed a significantly increased hierarchical composite win ratio, 1.72 (95% CI, 1.37-2.15; P < .001), that was consistent across LVEF categories and that accounted for all-cause mortality, HF events, KCCQ-CSS and 6MWD changes, and change in CRP.
Limitations make it hard to generalize the results, the authors caution. Well over 90% of the participants were White patients, for example, and the overall trial was not powered to show subgroup differences.
Given the many patients with HFpEF who have a cardiometabolic phenotype and are with overweight or obesity, write Dr. Butler and colleagues, their treatment approach “may ultimately include combination therapy with SGLT2 inhibitors and GLP-1 receptor agonists, given their non-overlapping and complementary mechanisms of action.”
Dr. Fonarow noted that both MRAs and sacubitril-valsartan offer clinical benefits for patients with HF and LVEF “in the 41%-60% range” that are evident “across BMI categories.”
So it’s likely, he said, that those medications as well as SGLT2 inhibitors will be used along with GLP-1 receptor agonists for patients with HFpEF and obesity.
STEP-HFpEF was funded by Novo Nordisk. Dr. Butler and the other authors disclose consulting for many companies, a list of which can be found in the report. Dr. Fonarow reports consulting for multiple companies. Dr. McMurray discloses consulting for AstraZeneca. Dr. Ostrominski reports no relevant disclosures. Dr. Vaduganathan discloses receiving grant support, serving on advisory boards, or speaking for multiple companies and serving on committees for studies sponsored by AstraZeneca, Galmed, Novartis, Bayer AG, Occlutech, and Impulse Dynamics.
A version of this article appeared on Medscape.com.
CLEVELAND – independently of baseline left-ventricular ejection fraction (LVEF).
The finding comes from a prespecified secondary analysis of the STEP-HFpEF trial of more than 500 nondiabetic patients with obesity and HF with an initial LVEF of 45% or greater.
They suggest that for patients with the obesity phenotype of HFpEF, semaglutide (Wegovy) could potentially join SGLT2 inhibitors on the short list of meds with consistent treatment effects whether LVEF is mildly reduced, preserved, or in the normal range.
That would distinguish the drug, a glucagon-like peptide-1 (GLP-1) receptor agonist, from mineralocorticoid receptor antagonists (MRA), sacubitril-valsartan (Entresto), and other renin-angiotensin-system inhibitors (RASi), whose benefits tend to taper off with rising LVEF.
The patients assigned to semaglutide showed significant improvement in both primary endpoints – change in Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ-CSS) and change in body weight at 52 weeks – whether their baseline LVEF was 45%-49%, 50%-59%, or 60% or greater.
Results were similar for improvements in 6-minute walk distance (6MWD) and levels of NT-terminal pro–brain natriuretic peptide (NT-proBNP) and C-reactive protein, observed Javed Butler, MD, when presenting the analysis at the annual meeting of the Heart Failure Society of America, Cleveland.
Dr. Butler, of Baylor Scott and White Research Institute, Dallas, and the University of Mississippi, Jackson, is also lead author of the study, which was published on the same day in the Journal of the American College of Cardiology.
In his presentation, Dr. Butler singled out the NT-proBNP finding as “very meaningful” with respect to understanding potential mechanisms of the drug effects observed in the trial.
For example, people with obesity tend to have lower than average natriuretic peptide levels that “actually go up a bit” when they lose weight, he observed. But in the trial, “we saw a reduction in NT-proBNP in spite of the weight loss,” regardless of LVEF category.
John McMurray, MD, University of Glasgow, the invited discussant for Dr. Butler’s presentation, agreed that it raises the question whether weight loss was the sole semaglutide effect responsible for the improvement in heart failure status and biomarkers. The accompanying NT-proBNP reductions – when the opposite might otherwise have been expected – may point to a possible mechanism of action that is “something more than just weight loss,” he said. “If that were the case, it becomes very important, because it means that this treatment might do good things in non-obese patients or might do good things in patients with other types of heart failure.”
‘Vital reassurance’
More definitive trials are needed “to clarify safety and efficacy of obesity-targeted therapeutics in HF across the ejection fraction spectrum,” according to an accompanying editorial).
Still, the STEP-HFpEF analysis “strengthens the role of GLP-1 [receptor agonists] to ameliorate health status” for patients with obesity and HF with mildly reduced or preserved ejection fraction, write Muthiah Vaduganathan, MD, MPH, and John W. Ostrominski, MD, Brigham and Women’s Hospital and Harvard Medical School, both in Boston.
Its findings “provide vital reassurance” on semaglutide safety and efficacy in HF with below-normal LVEF and “tentatively support the existence of a more general, LVEF-independent, obesity-related HF phenotype capable of favorable modification with incretin-based therapies.”
The lack of heterogeneity in treatment effects across LVEF subgroups “is not surprising,” but “the findings reinforce that the benefits of this therapy in those meeting trial criteria do not vary by left ventricular ejection fraction,” Gregg C. Fonarow, MD, University of California, Los Angeles, Medical Center, said in an interview.
It remains unknown, however, “whether the improvement in health status, functional status, and reduced inflammation” will translate to reduced risk of cardiovascular death or HF hospitalization, said Dr. Fonarow, who isn’t connected to STEP-HFpEF.
It’s a question for future studies, he agreed, whether semaglutide would confer similar benefits for patients with obesity and HF with LVEF less than 45% or in non-obese HF patients.
Dr. McMurray proposed that future GLP-1 receptor agonist heart-failure trials should include non-obese patients to determine whether the effects seen in STEP-HFpEF were due to something more than weight loss. Trials in patients with obesity and HF with reduced LVEF would also be important.
“If it turns out just to be about weight loss, then we need to think about the alternatives,” including diet, exercise, and bariatric surgery but also, potentially, weight-loss drugs other than semaglutide, he said.
No heterogeneity by LVEF
STEP-HFpEF randomly assigned 529 patients free of diabetes with an LVEF greater than or equal to 45%, a body mass index (BMI) of at least 30 kg/m2, and NYHA functional status of 2-4 to either a placebo injection or 2.4-mg semaglutide subcutaneously once a week (the dose used for weight reduction) atop standard care.
As previously reported, those assigned to semaglutide showed significant improvements at 1 year in symptoms and in physical limitation, per changes in KCCQ-CSS, and weight loss, compared with the control group. Their exercise capacity, as measured by 6MWD, also improved.
The more weight patients lost while taking semaglutide, the better their KCCQ-CSS and 6MWD outcomes, a prior secondary analysis suggested. But the STEP-HFpEF researchers said weight loss did not appear to explain all of their gains, compared with usual care.
For the current analysis, the 263 patients assigned to receive semaglutide and 266 control patients were divided into three groups by baseline LVEF and compared for the same outcomes.
The semaglutide group, compared with control patients, also showed a significantly increased hierarchical composite win ratio, 1.72 (95% CI, 1.37-2.15; P < .001), that was consistent across LVEF categories and that accounted for all-cause mortality, HF events, KCCQ-CSS and 6MWD changes, and change in CRP.
Limitations make it hard to generalize the results, the authors caution. Well over 90% of the participants were White patients, for example, and the overall trial was not powered to show subgroup differences.
Given the many patients with HFpEF who have a cardiometabolic phenotype and are with overweight or obesity, write Dr. Butler and colleagues, their treatment approach “may ultimately include combination therapy with SGLT2 inhibitors and GLP-1 receptor agonists, given their non-overlapping and complementary mechanisms of action.”
Dr. Fonarow noted that both MRAs and sacubitril-valsartan offer clinical benefits for patients with HF and LVEF “in the 41%-60% range” that are evident “across BMI categories.”
So it’s likely, he said, that those medications as well as SGLT2 inhibitors will be used along with GLP-1 receptor agonists for patients with HFpEF and obesity.
STEP-HFpEF was funded by Novo Nordisk. Dr. Butler and the other authors disclose consulting for many companies, a list of which can be found in the report. Dr. Fonarow reports consulting for multiple companies. Dr. McMurray discloses consulting for AstraZeneca. Dr. Ostrominski reports no relevant disclosures. Dr. Vaduganathan discloses receiving grant support, serving on advisory boards, or speaking for multiple companies and serving on committees for studies sponsored by AstraZeneca, Galmed, Novartis, Bayer AG, Occlutech, and Impulse Dynamics.
A version of this article appeared on Medscape.com.
CLEVELAND – independently of baseline left-ventricular ejection fraction (LVEF).
The finding comes from a prespecified secondary analysis of the STEP-HFpEF trial of more than 500 nondiabetic patients with obesity and HF with an initial LVEF of 45% or greater.
They suggest that for patients with the obesity phenotype of HFpEF, semaglutide (Wegovy) could potentially join SGLT2 inhibitors on the short list of meds with consistent treatment effects whether LVEF is mildly reduced, preserved, or in the normal range.
That would distinguish the drug, a glucagon-like peptide-1 (GLP-1) receptor agonist, from mineralocorticoid receptor antagonists (MRA), sacubitril-valsartan (Entresto), and other renin-angiotensin-system inhibitors (RASi), whose benefits tend to taper off with rising LVEF.
The patients assigned to semaglutide showed significant improvement in both primary endpoints – change in Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ-CSS) and change in body weight at 52 weeks – whether their baseline LVEF was 45%-49%, 50%-59%, or 60% or greater.
Results were similar for improvements in 6-minute walk distance (6MWD) and levels of NT-terminal pro–brain natriuretic peptide (NT-proBNP) and C-reactive protein, observed Javed Butler, MD, when presenting the analysis at the annual meeting of the Heart Failure Society of America, Cleveland.
Dr. Butler, of Baylor Scott and White Research Institute, Dallas, and the University of Mississippi, Jackson, is also lead author of the study, which was published on the same day in the Journal of the American College of Cardiology.
In his presentation, Dr. Butler singled out the NT-proBNP finding as “very meaningful” with respect to understanding potential mechanisms of the drug effects observed in the trial.
For example, people with obesity tend to have lower than average natriuretic peptide levels that “actually go up a bit” when they lose weight, he observed. But in the trial, “we saw a reduction in NT-proBNP in spite of the weight loss,” regardless of LVEF category.
John McMurray, MD, University of Glasgow, the invited discussant for Dr. Butler’s presentation, agreed that it raises the question whether weight loss was the sole semaglutide effect responsible for the improvement in heart failure status and biomarkers. The accompanying NT-proBNP reductions – when the opposite might otherwise have been expected – may point to a possible mechanism of action that is “something more than just weight loss,” he said. “If that were the case, it becomes very important, because it means that this treatment might do good things in non-obese patients or might do good things in patients with other types of heart failure.”
‘Vital reassurance’
More definitive trials are needed “to clarify safety and efficacy of obesity-targeted therapeutics in HF across the ejection fraction spectrum,” according to an accompanying editorial).
Still, the STEP-HFpEF analysis “strengthens the role of GLP-1 [receptor agonists] to ameliorate health status” for patients with obesity and HF with mildly reduced or preserved ejection fraction, write Muthiah Vaduganathan, MD, MPH, and John W. Ostrominski, MD, Brigham and Women’s Hospital and Harvard Medical School, both in Boston.
Its findings “provide vital reassurance” on semaglutide safety and efficacy in HF with below-normal LVEF and “tentatively support the existence of a more general, LVEF-independent, obesity-related HF phenotype capable of favorable modification with incretin-based therapies.”
The lack of heterogeneity in treatment effects across LVEF subgroups “is not surprising,” but “the findings reinforce that the benefits of this therapy in those meeting trial criteria do not vary by left ventricular ejection fraction,” Gregg C. Fonarow, MD, University of California, Los Angeles, Medical Center, said in an interview.
It remains unknown, however, “whether the improvement in health status, functional status, and reduced inflammation” will translate to reduced risk of cardiovascular death or HF hospitalization, said Dr. Fonarow, who isn’t connected to STEP-HFpEF.
It’s a question for future studies, he agreed, whether semaglutide would confer similar benefits for patients with obesity and HF with LVEF less than 45% or in non-obese HF patients.
Dr. McMurray proposed that future GLP-1 receptor agonist heart-failure trials should include non-obese patients to determine whether the effects seen in STEP-HFpEF were due to something more than weight loss. Trials in patients with obesity and HF with reduced LVEF would also be important.
“If it turns out just to be about weight loss, then we need to think about the alternatives,” including diet, exercise, and bariatric surgery but also, potentially, weight-loss drugs other than semaglutide, he said.
No heterogeneity by LVEF
STEP-HFpEF randomly assigned 529 patients free of diabetes with an LVEF greater than or equal to 45%, a body mass index (BMI) of at least 30 kg/m2, and NYHA functional status of 2-4 to either a placebo injection or 2.4-mg semaglutide subcutaneously once a week (the dose used for weight reduction) atop standard care.
As previously reported, those assigned to semaglutide showed significant improvements at 1 year in symptoms and in physical limitation, per changes in KCCQ-CSS, and weight loss, compared with the control group. Their exercise capacity, as measured by 6MWD, also improved.
The more weight patients lost while taking semaglutide, the better their KCCQ-CSS and 6MWD outcomes, a prior secondary analysis suggested. But the STEP-HFpEF researchers said weight loss did not appear to explain all of their gains, compared with usual care.
For the current analysis, the 263 patients assigned to receive semaglutide and 266 control patients were divided into three groups by baseline LVEF and compared for the same outcomes.
The semaglutide group, compared with control patients, also showed a significantly increased hierarchical composite win ratio, 1.72 (95% CI, 1.37-2.15; P < .001), that was consistent across LVEF categories and that accounted for all-cause mortality, HF events, KCCQ-CSS and 6MWD changes, and change in CRP.
Limitations make it hard to generalize the results, the authors caution. Well over 90% of the participants were White patients, for example, and the overall trial was not powered to show subgroup differences.
Given the many patients with HFpEF who have a cardiometabolic phenotype and are with overweight or obesity, write Dr. Butler and colleagues, their treatment approach “may ultimately include combination therapy with SGLT2 inhibitors and GLP-1 receptor agonists, given their non-overlapping and complementary mechanisms of action.”
Dr. Fonarow noted that both MRAs and sacubitril-valsartan offer clinical benefits for patients with HF and LVEF “in the 41%-60% range” that are evident “across BMI categories.”
So it’s likely, he said, that those medications as well as SGLT2 inhibitors will be used along with GLP-1 receptor agonists for patients with HFpEF and obesity.
STEP-HFpEF was funded by Novo Nordisk. Dr. Butler and the other authors disclose consulting for many companies, a list of which can be found in the report. Dr. Fonarow reports consulting for multiple companies. Dr. McMurray discloses consulting for AstraZeneca. Dr. Ostrominski reports no relevant disclosures. Dr. Vaduganathan discloses receiving grant support, serving on advisory boards, or speaking for multiple companies and serving on committees for studies sponsored by AstraZeneca, Galmed, Novartis, Bayer AG, Occlutech, and Impulse Dynamics.
A version of this article appeared on Medscape.com.
AT HFSA 2023
MASLD linked to increased risk for various clinical outcomes
TOPLINE:
Metabolic dysfunction–associated steatotic liver disease (MASLD) significantly raises the risk for cardiovascular, metabolic, oncologic, and other outcomes, a new meta-analysis suggests.
METHODOLOGY:
- Researchers conducted a comprehensive meta-analysis of studies to investigate longitudinal clinical outcomes associated with MASLD, previously known as nonalcoholic fatty liver disease.
- They identified 129 original studies that evaluated the longitudinal risks for incident clinical outcomes in patients with MASLD vs those without the disease.
- Investigators calculated pooled risk estimates for clinical outcomes in patients with MASLD and those without MASLD, with MASLD being diagnosed by imaging, biopsy, blood tests, or ICD codes.
TAKEAWAY:
- MASLD was associated with a significant increased risk for cardiovascular disease outcomes (hazard ratio, 1.43), metabolic outcomes such as incident hypertension (HR, 1.75), prediabetes (HR, 1.69), diabetes (HR, 2.56), metabolic syndrome (HR, 2.57), chronic kidney disease (HR, 1.38), and various liver-related outcomes (HR, 3.92).
- Patients with advanced MASLD had a significantly greater risk (P = .02) of developing diabetes than did their peers with less severe MASLD (HR, 1.63), compared with persons without MASLD.
- MASLD was also associated with all cancers (HR, 1.54); the highest risk was seen for hepatocellular carcinoma (HR, 4.37).
- Subgroup analyses stratified by sex found no significant differences in the risks observed between men and women with MASLD.
IN PRACTICE:
“It is imperative to understand that MASLD is a complex and multifaceted condition that requires a comprehensive approach to recognition and treatment beyond that of the hepatologist alone,” the authors wrote. “The growing prevalence of MASLD will remain a major global health threat that requires effective disease management frameworks to be put in place.”
SOURCE:
The study, with co–first authors Kai En Chan and Elden Yen Hng Ong, National University of Singapore, was published online in Clinical Gastroenterology and Hepatology.
LIMITATIONS:
The results depend on the validity of the original studies, and residual confounding factors may have biased the reported results. The study is also limited in its inclusion of large population-based studies using ICD codes that may result in misclassification bias. There was no examination of longitudinal outcomes in patients with histologically confirmed MASLD.
DISCLOSURES:
The study had no funding. Some authors reported research support, consulting fees, or stock options from pharmaceutical companies.
A version of this article appeared on Medscape.com.
TOPLINE:
Metabolic dysfunction–associated steatotic liver disease (MASLD) significantly raises the risk for cardiovascular, metabolic, oncologic, and other outcomes, a new meta-analysis suggests.
METHODOLOGY:
- Researchers conducted a comprehensive meta-analysis of studies to investigate longitudinal clinical outcomes associated with MASLD, previously known as nonalcoholic fatty liver disease.
- They identified 129 original studies that evaluated the longitudinal risks for incident clinical outcomes in patients with MASLD vs those without the disease.
- Investigators calculated pooled risk estimates for clinical outcomes in patients with MASLD and those without MASLD, with MASLD being diagnosed by imaging, biopsy, blood tests, or ICD codes.
TAKEAWAY:
- MASLD was associated with a significant increased risk for cardiovascular disease outcomes (hazard ratio, 1.43), metabolic outcomes such as incident hypertension (HR, 1.75), prediabetes (HR, 1.69), diabetes (HR, 2.56), metabolic syndrome (HR, 2.57), chronic kidney disease (HR, 1.38), and various liver-related outcomes (HR, 3.92).
- Patients with advanced MASLD had a significantly greater risk (P = .02) of developing diabetes than did their peers with less severe MASLD (HR, 1.63), compared with persons without MASLD.
- MASLD was also associated with all cancers (HR, 1.54); the highest risk was seen for hepatocellular carcinoma (HR, 4.37).
- Subgroup analyses stratified by sex found no significant differences in the risks observed between men and women with MASLD.
IN PRACTICE:
“It is imperative to understand that MASLD is a complex and multifaceted condition that requires a comprehensive approach to recognition and treatment beyond that of the hepatologist alone,” the authors wrote. “The growing prevalence of MASLD will remain a major global health threat that requires effective disease management frameworks to be put in place.”
SOURCE:
The study, with co–first authors Kai En Chan and Elden Yen Hng Ong, National University of Singapore, was published online in Clinical Gastroenterology and Hepatology.
LIMITATIONS:
The results depend on the validity of the original studies, and residual confounding factors may have biased the reported results. The study is also limited in its inclusion of large population-based studies using ICD codes that may result in misclassification bias. There was no examination of longitudinal outcomes in patients with histologically confirmed MASLD.
DISCLOSURES:
The study had no funding. Some authors reported research support, consulting fees, or stock options from pharmaceutical companies.
A version of this article appeared on Medscape.com.
TOPLINE:
Metabolic dysfunction–associated steatotic liver disease (MASLD) significantly raises the risk for cardiovascular, metabolic, oncologic, and other outcomes, a new meta-analysis suggests.
METHODOLOGY:
- Researchers conducted a comprehensive meta-analysis of studies to investigate longitudinal clinical outcomes associated with MASLD, previously known as nonalcoholic fatty liver disease.
- They identified 129 original studies that evaluated the longitudinal risks for incident clinical outcomes in patients with MASLD vs those without the disease.
- Investigators calculated pooled risk estimates for clinical outcomes in patients with MASLD and those without MASLD, with MASLD being diagnosed by imaging, biopsy, blood tests, or ICD codes.
TAKEAWAY:
- MASLD was associated with a significant increased risk for cardiovascular disease outcomes (hazard ratio, 1.43), metabolic outcomes such as incident hypertension (HR, 1.75), prediabetes (HR, 1.69), diabetes (HR, 2.56), metabolic syndrome (HR, 2.57), chronic kidney disease (HR, 1.38), and various liver-related outcomes (HR, 3.92).
- Patients with advanced MASLD had a significantly greater risk (P = .02) of developing diabetes than did their peers with less severe MASLD (HR, 1.63), compared with persons without MASLD.
- MASLD was also associated with all cancers (HR, 1.54); the highest risk was seen for hepatocellular carcinoma (HR, 4.37).
- Subgroup analyses stratified by sex found no significant differences in the risks observed between men and women with MASLD.
IN PRACTICE:
“It is imperative to understand that MASLD is a complex and multifaceted condition that requires a comprehensive approach to recognition and treatment beyond that of the hepatologist alone,” the authors wrote. “The growing prevalence of MASLD will remain a major global health threat that requires effective disease management frameworks to be put in place.”
SOURCE:
The study, with co–first authors Kai En Chan and Elden Yen Hng Ong, National University of Singapore, was published online in Clinical Gastroenterology and Hepatology.
LIMITATIONS:
The results depend on the validity of the original studies, and residual confounding factors may have biased the reported results. The study is also limited in its inclusion of large population-based studies using ICD codes that may result in misclassification bias. There was no examination of longitudinal outcomes in patients with histologically confirmed MASLD.
DISCLOSURES:
The study had no funding. Some authors reported research support, consulting fees, or stock options from pharmaceutical companies.
A version of this article appeared on Medscape.com.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Difficult-to-Control Diabetes: Is Cortisol at Play?
Is Cortisol at Play?
Daniel Einhorn, MD; John Buse, MD, PhD; Ralph DeFronzo, MD; Juan Pablo Frias, MD, and Christopher Lucci, MD, share their insights and perspectives on the connection between difficult-to control T2DM and untreated hypercortisolism:
• Almost a quarter of patients with type 2 diabetes (T2DM) require 3 or more medications to manage their disease, and even then, many patients have difficulty getting their diabetes under control1
• Elevated cortisol activity can exacerbate the pathophysiology of T2DM and can counter the impact of traditional anti-diabetic medications, making diabetes control challenging2-6
• Studies emerging over the last two decades suggest that up to 10% of patients with T2DM may have hypercortisolism7-11
• Patients with treatment-resistant T2DM should therefore be evaluated for hypercortisolism
• Treating the underlying hypercortisolism is important in these patients because managing comorbidities (alone) has not significantly reduced morbidity and mortality12,13

Daniel Einhorn, MD
Meeting Moderator
Vice President
Endocrine Strategy
Corcept Therapeutics
Menlo Park, CA

John Buse, MD, PhD
University of North Carolina
School of Medicine
UNC Diabetes and Endocrinology Clinic
Chapel Hill, NC

Ralph DeFronzo, MD
University of Texas
Health Science Center
San Antonio, TX

Juan Pablo Frias, MD
Velocity Clinical Research
Los Angeles, CA

Christopher Lucci, MD
Diabetes and Cardiovascular of Rockport
Rockport, TX
Click HERE to read the supplement.
References
- Fang M, et al. N Engl J Med. 2021;384(23):2219-2228.
- Scaroni C, et al. Endocr Rev. 2017;38(3):189-219.
- Mazziotti G, et al. Trends Endocrinol Metab. 2011;22(12):499-506.
- Pivonello R, et al. Neuroendocrinology. 2010;92(suppl 1):77-81.
- Mason IC, et al. Diabetologia. 2020;63(3):462-472.
- Thau L, et al. StatPearls [Internet]. Updated August 29, 2022. Accessed February 3, 2023. https://www.ncbi.nlm.nih.gov/books/NBK538239/
- Chiodini I, et al. Eur J Endocrinol. 2005;153(6):837-844.
- Catargi B, et al. J Clin Endocrinol Metab. 2003;88(12):5808-5813.
- Costa DS, et al. J Diabetes Complications. 2016;30(6):1032-1038.
- Leon-Justel A, et al. J Clin Endocrinol Metab. 2016;101(10):3747-3754.
- Steffensen C, et al. Horm Metab Res. 2019;51(1):62-68.
- Petramala L, et al. Endocrine. 2020;70(1):150-163.
- Morelli V, et al. Front Endocrinol (Lausanne). 2022;13:898084.
©2023 Corcept Therapeutics Incorporated. All Rights Reserved. DSE-01055 SEP 2023
Daniel Einhorn, MD; John Buse, MD, PhD; Ralph DeFronzo, MD; Juan Pablo Frias, MD, and Christopher Lucci, MD, share their insights and perspectives on the connection between difficult-to control T2DM and untreated hypercortisolism:
• Almost a quarter of patients with type 2 diabetes (T2DM) require 3 or more medications to manage their disease, and even then, many patients have difficulty getting their diabetes under control1
• Elevated cortisol activity can exacerbate the pathophysiology of T2DM and can counter the impact of traditional anti-diabetic medications, making diabetes control challenging2-6
• Studies emerging over the last two decades suggest that up to 10% of patients with T2DM may have hypercortisolism7-11
• Patients with treatment-resistant T2DM should therefore be evaluated for hypercortisolism
• Treating the underlying hypercortisolism is important in these patients because managing comorbidities (alone) has not significantly reduced morbidity and mortality12,13

Daniel Einhorn, MD
Meeting Moderator
Vice President
Endocrine Strategy
Corcept Therapeutics
Menlo Park, CA

John Buse, MD, PhD
University of North Carolina
School of Medicine
UNC Diabetes and Endocrinology Clinic
Chapel Hill, NC

Ralph DeFronzo, MD
University of Texas
Health Science Center
San Antonio, TX

Juan Pablo Frias, MD
Velocity Clinical Research
Los Angeles, CA

Christopher Lucci, MD
Diabetes and Cardiovascular of Rockport
Rockport, TX
Click HERE to read the supplement.
References
- Fang M, et al. N Engl J Med. 2021;384(23):2219-2228.
- Scaroni C, et al. Endocr Rev. 2017;38(3):189-219.
- Mazziotti G, et al. Trends Endocrinol Metab. 2011;22(12):499-506.
- Pivonello R, et al. Neuroendocrinology. 2010;92(suppl 1):77-81.
- Mason IC, et al. Diabetologia. 2020;63(3):462-472.
- Thau L, et al. StatPearls [Internet]. Updated August 29, 2022. Accessed February 3, 2023. https://www.ncbi.nlm.nih.gov/books/NBK538239/
- Chiodini I, et al. Eur J Endocrinol. 2005;153(6):837-844.
- Catargi B, et al. J Clin Endocrinol Metab. 2003;88(12):5808-5813.
- Costa DS, et al. J Diabetes Complications. 2016;30(6):1032-1038.
- Leon-Justel A, et al. J Clin Endocrinol Metab. 2016;101(10):3747-3754.
- Steffensen C, et al. Horm Metab Res. 2019;51(1):62-68.
- Petramala L, et al. Endocrine. 2020;70(1):150-163.
- Morelli V, et al. Front Endocrinol (Lausanne). 2022;13:898084.
©2023 Corcept Therapeutics Incorporated. All Rights Reserved. DSE-01055 SEP 2023
Daniel Einhorn, MD; John Buse, MD, PhD; Ralph DeFronzo, MD; Juan Pablo Frias, MD, and Christopher Lucci, MD, share their insights and perspectives on the connection between difficult-to control T2DM and untreated hypercortisolism:
• Almost a quarter of patients with type 2 diabetes (T2DM) require 3 or more medications to manage their disease, and even then, many patients have difficulty getting their diabetes under control1
• Elevated cortisol activity can exacerbate the pathophysiology of T2DM and can counter the impact of traditional anti-diabetic medications, making diabetes control challenging2-6
• Studies emerging over the last two decades suggest that up to 10% of patients with T2DM may have hypercortisolism7-11
• Patients with treatment-resistant T2DM should therefore be evaluated for hypercortisolism
• Treating the underlying hypercortisolism is important in these patients because managing comorbidities (alone) has not significantly reduced morbidity and mortality12,13

Daniel Einhorn, MD
Meeting Moderator
Vice President
Endocrine Strategy
Corcept Therapeutics
Menlo Park, CA

John Buse, MD, PhD
University of North Carolina
School of Medicine
UNC Diabetes and Endocrinology Clinic
Chapel Hill, NC

Ralph DeFronzo, MD
University of Texas
Health Science Center
San Antonio, TX

Juan Pablo Frias, MD
Velocity Clinical Research
Los Angeles, CA

Christopher Lucci, MD
Diabetes and Cardiovascular of Rockport
Rockport, TX
Click HERE to read the supplement.
References
- Fang M, et al. N Engl J Med. 2021;384(23):2219-2228.
- Scaroni C, et al. Endocr Rev. 2017;38(3):189-219.
- Mazziotti G, et al. Trends Endocrinol Metab. 2011;22(12):499-506.
- Pivonello R, et al. Neuroendocrinology. 2010;92(suppl 1):77-81.
- Mason IC, et al. Diabetologia. 2020;63(3):462-472.
- Thau L, et al. StatPearls [Internet]. Updated August 29, 2022. Accessed February 3, 2023. https://www.ncbi.nlm.nih.gov/books/NBK538239/
- Chiodini I, et al. Eur J Endocrinol. 2005;153(6):837-844.
- Catargi B, et al. J Clin Endocrinol Metab. 2003;88(12):5808-5813.
- Costa DS, et al. J Diabetes Complications. 2016;30(6):1032-1038.
- Leon-Justel A, et al. J Clin Endocrinol Metab. 2016;101(10):3747-3754.
- Steffensen C, et al. Horm Metab Res. 2019;51(1):62-68.
- Petramala L, et al. Endocrine. 2020;70(1):150-163.
- Morelli V, et al. Front Endocrinol (Lausanne). 2022;13:898084.
©2023 Corcept Therapeutics Incorporated. All Rights Reserved. DSE-01055 SEP 2023
Is Cortisol at Play?
Is Cortisol at Play?