User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Retinopathy ‘emerging decades earlier’ in kids with type 2 diabetes than in adults
Nearly one in four children diagnosed with type 2 diabetes for 5 years or more develop diabetic retinopathy, according to a new report.
The global prevalence of diabetic retinopathy in pediatric patients with type 2 diabetes is about 7%, which appears to increase with age.
“In our clinical practice, we have seen an increase in children presenting with type 2 diabetes over the past few years. These patients present with multiple simultaneous comorbidities and complications like hypertension, fatty liver, and other conditions,” senior author M. Constantine Samaan, MD, told this news organization.
“The exact scale of diabetes-related eye disease was not clear, and we decided to quantify it,” said Dr. Samaan, associate professor of pediatrics at McMaster University and pediatric endocrinologist at McMaster Children’s Hospital in Hamilton, Ont.
“What we found was that in pediatric patients with type 2 diabetes, diabetic retinopathy is present in 1 in 14 youth. The risk of retinopathy increased significantly 5 years after diagnosis to almost one in four,” he noted.
“While we acknowledged that the number of diabetic retinopathy cases was relatively small and there was heterogeneity in studies, we were surprised that retinopathy rates rose so fast in the first few years after diabetes diagnosis,” Dr. Samaan indicated.
The findings signal that the increase in the prevalence of diabetic retinopathy is emerging decades earlier among children compared with adults with type 2 diabetes, the authors wrote in their article published online in JAMA Network Open.
“While the guidelines for eye care in children with type 2 diabetes recommend screening at diagnosis and annually afterward, these recommendations are not followed in almost half of these patients,” Dr. Samaan said. “There is a need to ensure that patients get screened to try and prevent or delay retinopathy onset and progression.”
Analyzing prevalence rates
Diabetic retinopathy is the leading cause of blindness in patients with type 2 diabetes. Between 21% and 39% of adults have diabetic retinopathy at diagnosis, with rates subsequently increasing, the authors wrote.
Dr. Samaan and colleagues conducted a systematic review and meta-analysis to estimate the global prevalence of diabetic retinopathy in pediatric patients with type 2 diabetes. They included studies that had a study population of at least 10 participants diagnosed at age 21 and younger, an observational study design, and prevalence data on diabetic retinopathy.
Among the 29 studies included, 6 were cross-sectional, 13 had a retrospective cohort design, and 10 had a prospective cohort design. Patients were diagnosed between age 6.5 and 21 years, and the diabetes duration ranged from 0 to 15 years after diagnosis.
The overall global prevalence of diabetic retinopathy in 5,924 pediatric patients was 7.0%. Prevalence varied by study design, ranging from 1.1% in cross-sectional studies to 6.5% in prospective cohort studies and 11.3% in retrospective cohort studies.
In the nine studies that reported diabetic retinopathy classification based on criteria, the prevalence of minimal-to-moderate nonproliferative diabetic retinopathy was 11.2%, the prevalence of severe nonproliferative diabetic retinopathy was 2.6%, the prevalence of proliferative diabetic retinopathy was 2.4%, and the prevalence of macular edema was 3.1%.
In the five studies that reported diabetic retinopathy diagnosis using fundoscopy, the prevalence was 0.5%. In the four studies that used 7-field stereoscopic fundus photography, the prevalence was 13.6%.
In the pooled analysis of 27 studies, the prevalence of diabetic retinopathy was 1.8% less than 2.5 years after diabetes diagnosis but more than doubled to 5.1% in years 2.5 to 5 and jumped to 28.8% more than 5 years after diagnosis.
Differences by sex, ethnicity
“We were also surprised that there was very limited evidence to understand the sex and race differences in retinopathy risk,” said Dr. Samaan. “Further research is warranted, considering that more girls develop type 2 diabetes than boys, and the risk of type 2 diabetes is higher in some racial groups.”
In addition, older age, longer diabetes duration, and higher hypertension prevalence were associated with diabetic retinopathy prevalence. There were no associations with obesity prevalence or mean age at diabetes diagnosis. However, patients who developed diabetic retinopathy had a higher mean A1c level of 1.4% compared to those without retinopathy.
Dr. Samaan and colleagues are continuing to research the comorbidities and complications that children with type 2 diabetes face as well as mechanisms that drive diabetes outcomes among children and adolescents.
For now, the findings highlight the importance of retinopathy screening and personalized diabetes treatment to protect vision, Dr. Samaan reiterated.
No funding source for the study was reported. The authors have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Nearly one in four children diagnosed with type 2 diabetes for 5 years or more develop diabetic retinopathy, according to a new report.
The global prevalence of diabetic retinopathy in pediatric patients with type 2 diabetes is about 7%, which appears to increase with age.
“In our clinical practice, we have seen an increase in children presenting with type 2 diabetes over the past few years. These patients present with multiple simultaneous comorbidities and complications like hypertension, fatty liver, and other conditions,” senior author M. Constantine Samaan, MD, told this news organization.
“The exact scale of diabetes-related eye disease was not clear, and we decided to quantify it,” said Dr. Samaan, associate professor of pediatrics at McMaster University and pediatric endocrinologist at McMaster Children’s Hospital in Hamilton, Ont.
“What we found was that in pediatric patients with type 2 diabetes, diabetic retinopathy is present in 1 in 14 youth. The risk of retinopathy increased significantly 5 years after diagnosis to almost one in four,” he noted.
“While we acknowledged that the number of diabetic retinopathy cases was relatively small and there was heterogeneity in studies, we were surprised that retinopathy rates rose so fast in the first few years after diabetes diagnosis,” Dr. Samaan indicated.
The findings signal that the increase in the prevalence of diabetic retinopathy is emerging decades earlier among children compared with adults with type 2 diabetes, the authors wrote in their article published online in JAMA Network Open.
“While the guidelines for eye care in children with type 2 diabetes recommend screening at diagnosis and annually afterward, these recommendations are not followed in almost half of these patients,” Dr. Samaan said. “There is a need to ensure that patients get screened to try and prevent or delay retinopathy onset and progression.”
Analyzing prevalence rates
Diabetic retinopathy is the leading cause of blindness in patients with type 2 diabetes. Between 21% and 39% of adults have diabetic retinopathy at diagnosis, with rates subsequently increasing, the authors wrote.
Dr. Samaan and colleagues conducted a systematic review and meta-analysis to estimate the global prevalence of diabetic retinopathy in pediatric patients with type 2 diabetes. They included studies that had a study population of at least 10 participants diagnosed at age 21 and younger, an observational study design, and prevalence data on diabetic retinopathy.
Among the 29 studies included, 6 were cross-sectional, 13 had a retrospective cohort design, and 10 had a prospective cohort design. Patients were diagnosed between age 6.5 and 21 years, and the diabetes duration ranged from 0 to 15 years after diagnosis.
The overall global prevalence of diabetic retinopathy in 5,924 pediatric patients was 7.0%. Prevalence varied by study design, ranging from 1.1% in cross-sectional studies to 6.5% in prospective cohort studies and 11.3% in retrospective cohort studies.
In the nine studies that reported diabetic retinopathy classification based on criteria, the prevalence of minimal-to-moderate nonproliferative diabetic retinopathy was 11.2%, the prevalence of severe nonproliferative diabetic retinopathy was 2.6%, the prevalence of proliferative diabetic retinopathy was 2.4%, and the prevalence of macular edema was 3.1%.
In the five studies that reported diabetic retinopathy diagnosis using fundoscopy, the prevalence was 0.5%. In the four studies that used 7-field stereoscopic fundus photography, the prevalence was 13.6%.
In the pooled analysis of 27 studies, the prevalence of diabetic retinopathy was 1.8% less than 2.5 years after diabetes diagnosis but more than doubled to 5.1% in years 2.5 to 5 and jumped to 28.8% more than 5 years after diagnosis.
Differences by sex, ethnicity
“We were also surprised that there was very limited evidence to understand the sex and race differences in retinopathy risk,” said Dr. Samaan. “Further research is warranted, considering that more girls develop type 2 diabetes than boys, and the risk of type 2 diabetes is higher in some racial groups.”
In addition, older age, longer diabetes duration, and higher hypertension prevalence were associated with diabetic retinopathy prevalence. There were no associations with obesity prevalence or mean age at diabetes diagnosis. However, patients who developed diabetic retinopathy had a higher mean A1c level of 1.4% compared to those without retinopathy.
Dr. Samaan and colleagues are continuing to research the comorbidities and complications that children with type 2 diabetes face as well as mechanisms that drive diabetes outcomes among children and adolescents.
For now, the findings highlight the importance of retinopathy screening and personalized diabetes treatment to protect vision, Dr. Samaan reiterated.
No funding source for the study was reported. The authors have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Nearly one in four children diagnosed with type 2 diabetes for 5 years or more develop diabetic retinopathy, according to a new report.
The global prevalence of diabetic retinopathy in pediatric patients with type 2 diabetes is about 7%, which appears to increase with age.
“In our clinical practice, we have seen an increase in children presenting with type 2 diabetes over the past few years. These patients present with multiple simultaneous comorbidities and complications like hypertension, fatty liver, and other conditions,” senior author M. Constantine Samaan, MD, told this news organization.
“The exact scale of diabetes-related eye disease was not clear, and we decided to quantify it,” said Dr. Samaan, associate professor of pediatrics at McMaster University and pediatric endocrinologist at McMaster Children’s Hospital in Hamilton, Ont.
“What we found was that in pediatric patients with type 2 diabetes, diabetic retinopathy is present in 1 in 14 youth. The risk of retinopathy increased significantly 5 years after diagnosis to almost one in four,” he noted.
“While we acknowledged that the number of diabetic retinopathy cases was relatively small and there was heterogeneity in studies, we were surprised that retinopathy rates rose so fast in the first few years after diabetes diagnosis,” Dr. Samaan indicated.
The findings signal that the increase in the prevalence of diabetic retinopathy is emerging decades earlier among children compared with adults with type 2 diabetes, the authors wrote in their article published online in JAMA Network Open.
“While the guidelines for eye care in children with type 2 diabetes recommend screening at diagnosis and annually afterward, these recommendations are not followed in almost half of these patients,” Dr. Samaan said. “There is a need to ensure that patients get screened to try and prevent or delay retinopathy onset and progression.”
Analyzing prevalence rates
Diabetic retinopathy is the leading cause of blindness in patients with type 2 diabetes. Between 21% and 39% of adults have diabetic retinopathy at diagnosis, with rates subsequently increasing, the authors wrote.
Dr. Samaan and colleagues conducted a systematic review and meta-analysis to estimate the global prevalence of diabetic retinopathy in pediatric patients with type 2 diabetes. They included studies that had a study population of at least 10 participants diagnosed at age 21 and younger, an observational study design, and prevalence data on diabetic retinopathy.
Among the 29 studies included, 6 were cross-sectional, 13 had a retrospective cohort design, and 10 had a prospective cohort design. Patients were diagnosed between age 6.5 and 21 years, and the diabetes duration ranged from 0 to 15 years after diagnosis.
The overall global prevalence of diabetic retinopathy in 5,924 pediatric patients was 7.0%. Prevalence varied by study design, ranging from 1.1% in cross-sectional studies to 6.5% in prospective cohort studies and 11.3% in retrospective cohort studies.
In the nine studies that reported diabetic retinopathy classification based on criteria, the prevalence of minimal-to-moderate nonproliferative diabetic retinopathy was 11.2%, the prevalence of severe nonproliferative diabetic retinopathy was 2.6%, the prevalence of proliferative diabetic retinopathy was 2.4%, and the prevalence of macular edema was 3.1%.
In the five studies that reported diabetic retinopathy diagnosis using fundoscopy, the prevalence was 0.5%. In the four studies that used 7-field stereoscopic fundus photography, the prevalence was 13.6%.
In the pooled analysis of 27 studies, the prevalence of diabetic retinopathy was 1.8% less than 2.5 years after diabetes diagnosis but more than doubled to 5.1% in years 2.5 to 5 and jumped to 28.8% more than 5 years after diagnosis.
Differences by sex, ethnicity
“We were also surprised that there was very limited evidence to understand the sex and race differences in retinopathy risk,” said Dr. Samaan. “Further research is warranted, considering that more girls develop type 2 diabetes than boys, and the risk of type 2 diabetes is higher in some racial groups.”
In addition, older age, longer diabetes duration, and higher hypertension prevalence were associated with diabetic retinopathy prevalence. There were no associations with obesity prevalence or mean age at diabetes diagnosis. However, patients who developed diabetic retinopathy had a higher mean A1c level of 1.4% compared to those without retinopathy.
Dr. Samaan and colleagues are continuing to research the comorbidities and complications that children with type 2 diabetes face as well as mechanisms that drive diabetes outcomes among children and adolescents.
For now, the findings highlight the importance of retinopathy screening and personalized diabetes treatment to protect vision, Dr. Samaan reiterated.
No funding source for the study was reported. The authors have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Disparities in statin use persist in high-risk Americans
Disparities in statin use in minority populations persist regardless of insurance status and 10-year atherosclerotic cardiovascular disease risk.
Those are among the findings of a study that sampled a national population database and has provided robust data and granular details on those disparities.
The researchers reported in JAMA Cardiology that the overall prevalence of statin use was 25.5%, and that it varied significantly between defined ethnic groups: 20% for Blacks, 15.4% for Hispanics, and 27.9% for Whites (P < .001). Statin use rates by Asian participants, at 25.5%, didn’t differ significantly from use by Whites.
“We know that there are racial and ethnic disparities in the use of guideline-indicated statins after having established heart disease, but it was unknown if these disparities existed in the use of guideline-indicated statins for prevention of heart disease in those who just have risk factors,” lead author Joshua Jacobs, PharmD, a clinical pharmacist of cardiovascular medicine at University of Utah Intermountain Healthcare, said in written comments. “Additionally, race is included in the guideline-recommended risk factor calculation in an effort to reduce these disparities.”
Dr. Jacobs and colleagues evaluated statins for use in primary prevention, building upon previous single-center or diabetes-only cohort studies. What makes their study different from previous studies evaluating disparities in statin use is its use of temporal trends or current 10-year predicted ASCVD risk categorization, he said.
Using data from the National Health and Nutrition Examination Survey (NHANES), the researchers performed a serial, cross-sectional analysis of 3,417 participants that they said represented 39.4 million U.S. adults after applying sampling weights for age, gender, and race and ethnicity. In the weighted sample, 62.2% were men. In terms of self-reported race and ethnicity, 4.2% were of Asian descent, 12.7% were Black, 10.1% were Hispanic, and 73% were White.
Study participants completed a standardized questionnaire given by trained interviewers and also went to mobile examination centers where physical, anthropomorphic, and laboratory measurements, including height, weight, LDL cholesterol, and fasting blood glucose were collected. Pill bottle review also verified participants’ self-reported medication use.
The study noted that for primary prevention of atherosclerotic cardiovascular disease (ASCVD), the 2018 American College of Cardiology/American Heart Association Guideline recommends statins for, among other patient factors, elevated 10-year predicted ASCVD risk. The study divided ASCVD risk strata into three groups – 5% to less than 7.5%, 7.5% to less than 20%, and more than 20% – based on the 2018 ACC/AHA guideline and used pooled cohort equation to calculate 10-year ASCVD risk, which the guideline endorses.
Gaps persist despite ASCVD risk
The analysis found no statistically significant difference within each ASCVD risk strata between the White and Asian groups. But although statin use increased proportionately across each higher risk group, the gap widened noticeably in the highest risk group (more than 20% 10-year risk) between Whites, used as the reference at 37.6%, and Blacks (23.8%; prevalence ratio, .90; 95% confidence interval, .82-.98) and Hispanics (23.9%; PR, .90; 95% CI, .81-.99).
The study also evaluated a number of social determinants of health factors. Health insurance and access to routine health care were significantly associated with greater statin use in Black, Hispanic, and White participants; marital status and food insecurity were not. However, even when variables such as education, household income, and health insurance were applied, statin use was still significantly higher in Whites than in Blacks and Hispanics. For those with health insurance, statin use was 28.6% (95% CI, 25-32), 21.1% (95% CI, 17.3-25.4) and 19.9% (95% CI, 15.9-24.5), respectively.
The study noted that the pooled cohort equation-guided approach to statins for primary prevention, which the 2018 ACC/AHA guideline endorsed, should promote greater use of statins among Black patients. “Equitable use of statin therapy for prevention of heart disease is needed for Black and Hispanic adults,” Dr. Jacobs said. “Improvements in access to care, such as having a routine primary care clinician and health insurance, may decrease these health disparities.”
A goal of the study was to identify if disparities in statin use held up across different risk groups, senior author Ambarish Pandey, MD, said in an interview. Use of the NHANES data makes this study unique among analyses of statin use disparities, he said.
“A lot of the work that has been done previously has focused on secondary prevention among patients who have atherosclerotic cardiovascular disease or have focused on single-center or hospital-based cohorts and have not really focused on a national representative cohort like NHANES,” said Dr. Pandey, of the UT Southwestern Medical Center, Dallas.
The next step is to do community-based participatory research focusing on different implementation strategies to increase the uptake of preventive statin use among Black and Hispanic communities, Dr. Jacobs said.
Dr. Jacobs has no relevant relationships to disclose. Dr. Pandey disclosed relationships with Gilead Sciences, Applied Therapeutics, Myovista, Tricog Health, Eli Lilly, Cytokinetics, Rivus, Roche Diagnostics, Pieces Technologies, Palomarin, Emmi Solutions, and Axon.
Disparities in statin use in minority populations persist regardless of insurance status and 10-year atherosclerotic cardiovascular disease risk.
Those are among the findings of a study that sampled a national population database and has provided robust data and granular details on those disparities.
The researchers reported in JAMA Cardiology that the overall prevalence of statin use was 25.5%, and that it varied significantly between defined ethnic groups: 20% for Blacks, 15.4% for Hispanics, and 27.9% for Whites (P < .001). Statin use rates by Asian participants, at 25.5%, didn’t differ significantly from use by Whites.
“We know that there are racial and ethnic disparities in the use of guideline-indicated statins after having established heart disease, but it was unknown if these disparities existed in the use of guideline-indicated statins for prevention of heart disease in those who just have risk factors,” lead author Joshua Jacobs, PharmD, a clinical pharmacist of cardiovascular medicine at University of Utah Intermountain Healthcare, said in written comments. “Additionally, race is included in the guideline-recommended risk factor calculation in an effort to reduce these disparities.”
Dr. Jacobs and colleagues evaluated statins for use in primary prevention, building upon previous single-center or diabetes-only cohort studies. What makes their study different from previous studies evaluating disparities in statin use is its use of temporal trends or current 10-year predicted ASCVD risk categorization, he said.
Using data from the National Health and Nutrition Examination Survey (NHANES), the researchers performed a serial, cross-sectional analysis of 3,417 participants that they said represented 39.4 million U.S. adults after applying sampling weights for age, gender, and race and ethnicity. In the weighted sample, 62.2% were men. In terms of self-reported race and ethnicity, 4.2% were of Asian descent, 12.7% were Black, 10.1% were Hispanic, and 73% were White.
Study participants completed a standardized questionnaire given by trained interviewers and also went to mobile examination centers where physical, anthropomorphic, and laboratory measurements, including height, weight, LDL cholesterol, and fasting blood glucose were collected. Pill bottle review also verified participants’ self-reported medication use.
The study noted that for primary prevention of atherosclerotic cardiovascular disease (ASCVD), the 2018 American College of Cardiology/American Heart Association Guideline recommends statins for, among other patient factors, elevated 10-year predicted ASCVD risk. The study divided ASCVD risk strata into three groups – 5% to less than 7.5%, 7.5% to less than 20%, and more than 20% – based on the 2018 ACC/AHA guideline and used pooled cohort equation to calculate 10-year ASCVD risk, which the guideline endorses.
Gaps persist despite ASCVD risk
The analysis found no statistically significant difference within each ASCVD risk strata between the White and Asian groups. But although statin use increased proportionately across each higher risk group, the gap widened noticeably in the highest risk group (more than 20% 10-year risk) between Whites, used as the reference at 37.6%, and Blacks (23.8%; prevalence ratio, .90; 95% confidence interval, .82-.98) and Hispanics (23.9%; PR, .90; 95% CI, .81-.99).
The study also evaluated a number of social determinants of health factors. Health insurance and access to routine health care were significantly associated with greater statin use in Black, Hispanic, and White participants; marital status and food insecurity were not. However, even when variables such as education, household income, and health insurance were applied, statin use was still significantly higher in Whites than in Blacks and Hispanics. For those with health insurance, statin use was 28.6% (95% CI, 25-32), 21.1% (95% CI, 17.3-25.4) and 19.9% (95% CI, 15.9-24.5), respectively.
The study noted that the pooled cohort equation-guided approach to statins for primary prevention, which the 2018 ACC/AHA guideline endorsed, should promote greater use of statins among Black patients. “Equitable use of statin therapy for prevention of heart disease is needed for Black and Hispanic adults,” Dr. Jacobs said. “Improvements in access to care, such as having a routine primary care clinician and health insurance, may decrease these health disparities.”
A goal of the study was to identify if disparities in statin use held up across different risk groups, senior author Ambarish Pandey, MD, said in an interview. Use of the NHANES data makes this study unique among analyses of statin use disparities, he said.
“A lot of the work that has been done previously has focused on secondary prevention among patients who have atherosclerotic cardiovascular disease or have focused on single-center or hospital-based cohorts and have not really focused on a national representative cohort like NHANES,” said Dr. Pandey, of the UT Southwestern Medical Center, Dallas.
The next step is to do community-based participatory research focusing on different implementation strategies to increase the uptake of preventive statin use among Black and Hispanic communities, Dr. Jacobs said.
Dr. Jacobs has no relevant relationships to disclose. Dr. Pandey disclosed relationships with Gilead Sciences, Applied Therapeutics, Myovista, Tricog Health, Eli Lilly, Cytokinetics, Rivus, Roche Diagnostics, Pieces Technologies, Palomarin, Emmi Solutions, and Axon.
Disparities in statin use in minority populations persist regardless of insurance status and 10-year atherosclerotic cardiovascular disease risk.
Those are among the findings of a study that sampled a national population database and has provided robust data and granular details on those disparities.
The researchers reported in JAMA Cardiology that the overall prevalence of statin use was 25.5%, and that it varied significantly between defined ethnic groups: 20% for Blacks, 15.4% for Hispanics, and 27.9% for Whites (P < .001). Statin use rates by Asian participants, at 25.5%, didn’t differ significantly from use by Whites.
“We know that there are racial and ethnic disparities in the use of guideline-indicated statins after having established heart disease, but it was unknown if these disparities existed in the use of guideline-indicated statins for prevention of heart disease in those who just have risk factors,” lead author Joshua Jacobs, PharmD, a clinical pharmacist of cardiovascular medicine at University of Utah Intermountain Healthcare, said in written comments. “Additionally, race is included in the guideline-recommended risk factor calculation in an effort to reduce these disparities.”
Dr. Jacobs and colleagues evaluated statins for use in primary prevention, building upon previous single-center or diabetes-only cohort studies. What makes their study different from previous studies evaluating disparities in statin use is its use of temporal trends or current 10-year predicted ASCVD risk categorization, he said.
Using data from the National Health and Nutrition Examination Survey (NHANES), the researchers performed a serial, cross-sectional analysis of 3,417 participants that they said represented 39.4 million U.S. adults after applying sampling weights for age, gender, and race and ethnicity. In the weighted sample, 62.2% were men. In terms of self-reported race and ethnicity, 4.2% were of Asian descent, 12.7% were Black, 10.1% were Hispanic, and 73% were White.
Study participants completed a standardized questionnaire given by trained interviewers and also went to mobile examination centers where physical, anthropomorphic, and laboratory measurements, including height, weight, LDL cholesterol, and fasting blood glucose were collected. Pill bottle review also verified participants’ self-reported medication use.
The study noted that for primary prevention of atherosclerotic cardiovascular disease (ASCVD), the 2018 American College of Cardiology/American Heart Association Guideline recommends statins for, among other patient factors, elevated 10-year predicted ASCVD risk. The study divided ASCVD risk strata into three groups – 5% to less than 7.5%, 7.5% to less than 20%, and more than 20% – based on the 2018 ACC/AHA guideline and used pooled cohort equation to calculate 10-year ASCVD risk, which the guideline endorses.
Gaps persist despite ASCVD risk
The analysis found no statistically significant difference within each ASCVD risk strata between the White and Asian groups. But although statin use increased proportionately across each higher risk group, the gap widened noticeably in the highest risk group (more than 20% 10-year risk) between Whites, used as the reference at 37.6%, and Blacks (23.8%; prevalence ratio, .90; 95% confidence interval, .82-.98) and Hispanics (23.9%; PR, .90; 95% CI, .81-.99).
The study also evaluated a number of social determinants of health factors. Health insurance and access to routine health care were significantly associated with greater statin use in Black, Hispanic, and White participants; marital status and food insecurity were not. However, even when variables such as education, household income, and health insurance were applied, statin use was still significantly higher in Whites than in Blacks and Hispanics. For those with health insurance, statin use was 28.6% (95% CI, 25-32), 21.1% (95% CI, 17.3-25.4) and 19.9% (95% CI, 15.9-24.5), respectively.
The study noted that the pooled cohort equation-guided approach to statins for primary prevention, which the 2018 ACC/AHA guideline endorsed, should promote greater use of statins among Black patients. “Equitable use of statin therapy for prevention of heart disease is needed for Black and Hispanic adults,” Dr. Jacobs said. “Improvements in access to care, such as having a routine primary care clinician and health insurance, may decrease these health disparities.”
A goal of the study was to identify if disparities in statin use held up across different risk groups, senior author Ambarish Pandey, MD, said in an interview. Use of the NHANES data makes this study unique among analyses of statin use disparities, he said.
“A lot of the work that has been done previously has focused on secondary prevention among patients who have atherosclerotic cardiovascular disease or have focused on single-center or hospital-based cohorts and have not really focused on a national representative cohort like NHANES,” said Dr. Pandey, of the UT Southwestern Medical Center, Dallas.
The next step is to do community-based participatory research focusing on different implementation strategies to increase the uptake of preventive statin use among Black and Hispanic communities, Dr. Jacobs said.
Dr. Jacobs has no relevant relationships to disclose. Dr. Pandey disclosed relationships with Gilead Sciences, Applied Therapeutics, Myovista, Tricog Health, Eli Lilly, Cytokinetics, Rivus, Roche Diagnostics, Pieces Technologies, Palomarin, Emmi Solutions, and Axon.
FROM JAMA CARDIOLOGY
Luxe vacations, private jets: Medical device maker, surgeon to pay $46 million penalty in kickback scheme
according to experts familiar with the federal Anti-Kickback Statute.
Historically, enforcement actions have primarily focused on the person or organization offering the perks – and not necessarily the physicians accepting it, Steven W. Ortquist, founder and principal of Arete Compliance Solutions, LLC, in Phoenix, told this news organization.
But that’s changing.
“In recent years, we are seeing a trend toward holding physicians and others on the receiving end of the inducement accountable as well,” said Mr. Ortquist, who is a past board member and president of the Health Care Compliance Association. He noted that authorities usually pursue the inducing company first before moving on to individual clinicians or practices.
The Department of Justice followed a similar pattern in a recently announced kickback settlement that ensnared an intraocular lens distributor, an ophthalmology equipment supplier, two CEOs, and a surgeon. Precision Lens must pay more than $43 million for offering high-end vacations and other expensive perks to surgeons who used its cataract products.
The verdict marks the end of a 6-week civil jury trial, where evidence emerged that Paul Ehlen, owner of Precision Lens and its parent company, Cameron-Ehlen Group, maintained a secret “slush fund” for paying kickbacks to ophthalmic surgeons. The inducement scheme netted the Minnesota-based company millions in sales and led to the submission of 64,575 false Medicare claims from 2006 to 2015, a violation of the Anti-Kickback Statute and the False Claims Act.
According to court documents, physicians received luxury travel and entertainment packages, including skiing, fishing, and golfing excursions at exclusive destinations, often traveling via private jet to attend Broadway musicals and major sporting events. Mr. Ehlen and company representatives also sold frequent flyer miles to physicians at a steep discount, allowing them to take personal and business trips below fair market value.
Federal authorities initially announced an investigation into the business practices of Precision Lens in 2017 after receiving a whistleblower complaint from Kipp Fesenmaier, a former executive at Sightpath Medical, an ophthalmology supplier and “corporate partner” of Precision Lens. Mr. Fesenmaier alleged that both companies were involved in an inducement scheme.
Sightpath Medical and its CEO, James Tiffany, agreed to a $12 million settlement to resolve the kickback allegations.
The Department of Justice subsequently investigated Jitendra Swarup, MD, an ophthalmologist and cataract surgeon who allegedly received “unlawful remuneration from Sightpath, Precision, and Ehlen” and filed false insurance claims. In addition to accepting expensive hunting and fishing trips from the medical device companies, Dr. Swarup was paid more than $100,000 per year for consulting services he did not fully render.
Dr. Swarup agreed to a nearly $3 million settlement and participation in a 3-year corporate integrity agreement with the Office of Inspector General. In exchange for compliance with such contracts, the OIG permits physicians to continue participating in Medicare, Medicaid, and other federal health care programs.
In a statement from attorneys, Precision Lens and Mr. Ehlen pledged to appeal the verdict and “defend ... our wholly appropriate actions” while remaining focused on their commitment to health care clinicians and manufacturers.
‘Endless’ opportunities for inducement
Unfortunately, opportunities for inducement are “endless,” experts say. Extravagant trips, dinners, and gifts can trigger a violation, but so can nearly anything of value.
Just last year, Biotronik reached a $12.95 million settlement amid allegations that company representatives wined and dined physicians to induce their use of its pacemakers and defibrillators. To date, no physicians have been charged.
But after a record-breaking number of whistleblower judgments last fiscal year totaling more than $2 billion, physicians should take note, Radha Bhatnagar, Esq, director of compliance at The CM Group, told the news organization.
“When manufacturers offer physicians kickbacks with the added element of fraudulent Medicare or Medicaid reimbursements, that is typically when manufacturers and individuals face civil and criminal liability,” said Ms. Bhatnagar, something the Department of Justice alluded to when announcing a settlement involving 15 Texas physicians last year.
In another case, Kingsley R. Chin, an orthopedic surgeon and designer of a spinal implant, was indicted in 2021 for paying millions of dollars in sham consulting fees to physicians who used his products. At least six surgeons who accepted money from Dr. Chin were later named in a civil case and ordered to pay $3.3 million in penalties.
Jason Montone, DO, an orthopedic surgeon who accepted the illicit payments, agreed to a plea deal with a reduced prison sentence, 1 year of supervised release, and a fine of $379,000.
Although Dr. Chin’s sentencing hasn’t been announced, violating kickback laws can result in a sentence of up to 10 years.
A version of this article originally appeared on Medscape.com.
according to experts familiar with the federal Anti-Kickback Statute.
Historically, enforcement actions have primarily focused on the person or organization offering the perks – and not necessarily the physicians accepting it, Steven W. Ortquist, founder and principal of Arete Compliance Solutions, LLC, in Phoenix, told this news organization.
But that’s changing.
“In recent years, we are seeing a trend toward holding physicians and others on the receiving end of the inducement accountable as well,” said Mr. Ortquist, who is a past board member and president of the Health Care Compliance Association. He noted that authorities usually pursue the inducing company first before moving on to individual clinicians or practices.
The Department of Justice followed a similar pattern in a recently announced kickback settlement that ensnared an intraocular lens distributor, an ophthalmology equipment supplier, two CEOs, and a surgeon. Precision Lens must pay more than $43 million for offering high-end vacations and other expensive perks to surgeons who used its cataract products.
The verdict marks the end of a 6-week civil jury trial, where evidence emerged that Paul Ehlen, owner of Precision Lens and its parent company, Cameron-Ehlen Group, maintained a secret “slush fund” for paying kickbacks to ophthalmic surgeons. The inducement scheme netted the Minnesota-based company millions in sales and led to the submission of 64,575 false Medicare claims from 2006 to 2015, a violation of the Anti-Kickback Statute and the False Claims Act.
According to court documents, physicians received luxury travel and entertainment packages, including skiing, fishing, and golfing excursions at exclusive destinations, often traveling via private jet to attend Broadway musicals and major sporting events. Mr. Ehlen and company representatives also sold frequent flyer miles to physicians at a steep discount, allowing them to take personal and business trips below fair market value.
Federal authorities initially announced an investigation into the business practices of Precision Lens in 2017 after receiving a whistleblower complaint from Kipp Fesenmaier, a former executive at Sightpath Medical, an ophthalmology supplier and “corporate partner” of Precision Lens. Mr. Fesenmaier alleged that both companies were involved in an inducement scheme.
Sightpath Medical and its CEO, James Tiffany, agreed to a $12 million settlement to resolve the kickback allegations.
The Department of Justice subsequently investigated Jitendra Swarup, MD, an ophthalmologist and cataract surgeon who allegedly received “unlawful remuneration from Sightpath, Precision, and Ehlen” and filed false insurance claims. In addition to accepting expensive hunting and fishing trips from the medical device companies, Dr. Swarup was paid more than $100,000 per year for consulting services he did not fully render.
Dr. Swarup agreed to a nearly $3 million settlement and participation in a 3-year corporate integrity agreement with the Office of Inspector General. In exchange for compliance with such contracts, the OIG permits physicians to continue participating in Medicare, Medicaid, and other federal health care programs.
In a statement from attorneys, Precision Lens and Mr. Ehlen pledged to appeal the verdict and “defend ... our wholly appropriate actions” while remaining focused on their commitment to health care clinicians and manufacturers.
‘Endless’ opportunities for inducement
Unfortunately, opportunities for inducement are “endless,” experts say. Extravagant trips, dinners, and gifts can trigger a violation, but so can nearly anything of value.
Just last year, Biotronik reached a $12.95 million settlement amid allegations that company representatives wined and dined physicians to induce their use of its pacemakers and defibrillators. To date, no physicians have been charged.
But after a record-breaking number of whistleblower judgments last fiscal year totaling more than $2 billion, physicians should take note, Radha Bhatnagar, Esq, director of compliance at The CM Group, told the news organization.
“When manufacturers offer physicians kickbacks with the added element of fraudulent Medicare or Medicaid reimbursements, that is typically when manufacturers and individuals face civil and criminal liability,” said Ms. Bhatnagar, something the Department of Justice alluded to when announcing a settlement involving 15 Texas physicians last year.
In another case, Kingsley R. Chin, an orthopedic surgeon and designer of a spinal implant, was indicted in 2021 for paying millions of dollars in sham consulting fees to physicians who used his products. At least six surgeons who accepted money from Dr. Chin were later named in a civil case and ordered to pay $3.3 million in penalties.
Jason Montone, DO, an orthopedic surgeon who accepted the illicit payments, agreed to a plea deal with a reduced prison sentence, 1 year of supervised release, and a fine of $379,000.
Although Dr. Chin’s sentencing hasn’t been announced, violating kickback laws can result in a sentence of up to 10 years.
A version of this article originally appeared on Medscape.com.
according to experts familiar with the federal Anti-Kickback Statute.
Historically, enforcement actions have primarily focused on the person or organization offering the perks – and not necessarily the physicians accepting it, Steven W. Ortquist, founder and principal of Arete Compliance Solutions, LLC, in Phoenix, told this news organization.
But that’s changing.
“In recent years, we are seeing a trend toward holding physicians and others on the receiving end of the inducement accountable as well,” said Mr. Ortquist, who is a past board member and president of the Health Care Compliance Association. He noted that authorities usually pursue the inducing company first before moving on to individual clinicians or practices.
The Department of Justice followed a similar pattern in a recently announced kickback settlement that ensnared an intraocular lens distributor, an ophthalmology equipment supplier, two CEOs, and a surgeon. Precision Lens must pay more than $43 million for offering high-end vacations and other expensive perks to surgeons who used its cataract products.
The verdict marks the end of a 6-week civil jury trial, where evidence emerged that Paul Ehlen, owner of Precision Lens and its parent company, Cameron-Ehlen Group, maintained a secret “slush fund” for paying kickbacks to ophthalmic surgeons. The inducement scheme netted the Minnesota-based company millions in sales and led to the submission of 64,575 false Medicare claims from 2006 to 2015, a violation of the Anti-Kickback Statute and the False Claims Act.
According to court documents, physicians received luxury travel and entertainment packages, including skiing, fishing, and golfing excursions at exclusive destinations, often traveling via private jet to attend Broadway musicals and major sporting events. Mr. Ehlen and company representatives also sold frequent flyer miles to physicians at a steep discount, allowing them to take personal and business trips below fair market value.
Federal authorities initially announced an investigation into the business practices of Precision Lens in 2017 after receiving a whistleblower complaint from Kipp Fesenmaier, a former executive at Sightpath Medical, an ophthalmology supplier and “corporate partner” of Precision Lens. Mr. Fesenmaier alleged that both companies were involved in an inducement scheme.
Sightpath Medical and its CEO, James Tiffany, agreed to a $12 million settlement to resolve the kickback allegations.
The Department of Justice subsequently investigated Jitendra Swarup, MD, an ophthalmologist and cataract surgeon who allegedly received “unlawful remuneration from Sightpath, Precision, and Ehlen” and filed false insurance claims. In addition to accepting expensive hunting and fishing trips from the medical device companies, Dr. Swarup was paid more than $100,000 per year for consulting services he did not fully render.
Dr. Swarup agreed to a nearly $3 million settlement and participation in a 3-year corporate integrity agreement with the Office of Inspector General. In exchange for compliance with such contracts, the OIG permits physicians to continue participating in Medicare, Medicaid, and other federal health care programs.
In a statement from attorneys, Precision Lens and Mr. Ehlen pledged to appeal the verdict and “defend ... our wholly appropriate actions” while remaining focused on their commitment to health care clinicians and manufacturers.
‘Endless’ opportunities for inducement
Unfortunately, opportunities for inducement are “endless,” experts say. Extravagant trips, dinners, and gifts can trigger a violation, but so can nearly anything of value.
Just last year, Biotronik reached a $12.95 million settlement amid allegations that company representatives wined and dined physicians to induce their use of its pacemakers and defibrillators. To date, no physicians have been charged.
But after a record-breaking number of whistleblower judgments last fiscal year totaling more than $2 billion, physicians should take note, Radha Bhatnagar, Esq, director of compliance at The CM Group, told the news organization.
“When manufacturers offer physicians kickbacks with the added element of fraudulent Medicare or Medicaid reimbursements, that is typically when manufacturers and individuals face civil and criminal liability,” said Ms. Bhatnagar, something the Department of Justice alluded to when announcing a settlement involving 15 Texas physicians last year.
In another case, Kingsley R. Chin, an orthopedic surgeon and designer of a spinal implant, was indicted in 2021 for paying millions of dollars in sham consulting fees to physicians who used his products. At least six surgeons who accepted money from Dr. Chin were later named in a civil case and ordered to pay $3.3 million in penalties.
Jason Montone, DO, an orthopedic surgeon who accepted the illicit payments, agreed to a plea deal with a reduced prison sentence, 1 year of supervised release, and a fine of $379,000.
Although Dr. Chin’s sentencing hasn’t been announced, violating kickback laws can result in a sentence of up to 10 years.
A version of this article originally appeared on Medscape.com.
What happens when newer weight loss meds are stopped?
Some of these medicines are approved for treating obesity (Wegovy), whereas others are approved for type 2 diabetes (Ozempic and Mounjaro). Tirzepatide (Mounjaro) has been fast-tracked for approval for weight loss by the U.S. Food and Drug Administration this year, and in the first of the series of studies looking at its effect on obesity, the SURMOUNT-1 trial, tirzepatide demonstrated a mean weight loss of around 22% in people without diabetes, spurring significant off-label use.
Our offices are full of patients who have taken these medications, with unprecedented improvements in their weight, cardiometabolic health, and quality of life. What happens when patients stop taking these medications? Or more importantly, why stop them?
Although these drugs are very effective for weight loss and treating diabetes, there can be adverse effects, primarily gastrointestinal, that limit treatment continuation. Nausea is the most common side effect and usually diminishes over time. Slow dose titration and dietary modification can minimize unwanted gastrointestinal side effects.
Drug-induced acute pancreatitis, a rare adverse event requiring patients to stop therapy, was seen in approximately 0.2% of people in clinical trials.
Medications effective but cost prohibitive?
Beyond adverse effects, patients may be forced to stop treatment because of medication cost, changes in insurance coverage, or issues with drug availability.
Two incretin therapies currently approved for treating obesity – liraglutide (Saxenda) and semaglutide (Wegovy) – cost around $1,400 per month. Insurance coverage and manufacturer discounts can make treatment affordable, but anti-obesity medicines aren’t covered by Medicare or by many employer-sponsored commercial plans.
Changes in employment or insurance coverage, or expiration of manufacturer copay cards, may require patients to stop or change therapies. The increased prescribing and overall expense of these drugs have prompted insurance plans and self-insured groups to consider whether providing coverage for these medications is sustainable.
Limited coverage has led to significant off-label prescribing of incretin therapies that aren’t approved for treating obesity (for instance, Ozempic and Mounjaro) and compounding pharmacies selling peptides that allegedly contain the active pharmaceutical ingredients. High demand for these medications has created significant supply shortages over the past year, causing many people to be without treatment for significant periods of time, as reported by this news organization.
Recently, I saw a patient who lost more than 30 pounds with semaglutide (Wegovy). She then changed employers and the medication was no longer covered. She gained back almost 10 pounds over 3 months and was prescribed tirzepatide (Mounjaro) off-label for weight loss by another provider, using a manufacturer discount card to make the medication affordable. The patient did well with the new regimen and lost about 20 pounds, but the pharmacy stopped filling the prescription when changes were made to the discount card. Afraid of regaining the weight, she came to see us as a new patient to discuss her options with her lack of coverage for anti-obesity medications.
Stopping equals weight regain
Obesity is a chronic disease like hypertension. It responds to treatment and when people stop taking these anti-obesity medications, this is generally associated with increased appetite and less satiety, and there is subsequent weight regain and a recurrence in excess weight-related complications.
The STEP-1 trial extension showed an initial mean body weight reduction of 17.3% with weekly semaglutide 2.4 mg over 1 year. On average, two-thirds of the weight lost was regained by participants within 1 year of stopping semaglutide and the study’s lifestyle intervention. Many of the improvements seen in cardiometabolic variables, like blood glucose and blood pressure, similarly reverted to baseline.
There are also 2-year data from the STEP-5 trial with semaglutide; 3-year data from the SCALE trial with liraglutide; and 5-year nonrandomized data with multiple agents that show durable, clinically significant weight loss from medical therapies for obesity.
These data together demonstrate that medications are effective for durable weight loss if they are continued. However, this is not how obesity is currently treated. Anti-obesity medications are prescribed to less than 3% of eligible people in the United States, and the average duration of therapy is less than 90 days. This treatment length isn’t sufficient to see the full benefits most medications offer and certainly doesn’t support long-term weight maintenance.
A recent study showed that, in addition to maintaining weight loss from medical therapies, incretin-containing anti-obesity medication regimens were effective for treating weight regain and facilitating healthier weight after bariatric surgery.
Chronic therapy is needed for weight maintenance because several neurohormonal changes occur owing to weight loss. Metabolic adaptation is the relative reduction in energy expenditure, below what would be expected, in people after weight loss. When this is combined with physiologic changes that increase appetite and decrease satiety, many people create a positive energy balance that results in weight regain. This has been observed in reality TV shows such as “The Biggest Loser”: It’s biology, not willpower.
Unfortunately, many people – including health care providers – don’t understand how these changes promote weight regain and patients are too often blamed when their weight goes back up after medications are stopped. This blame is greatly misinformed by weight-biased beliefs that people with obesity are lazy and lack self-control for weight loss or maintenance. Nobody would be surprised if someone’s blood pressure went up if their antihypertensive medications were stopped. Why do we think so differently when treating obesity?
The prevalence of obesity in the United States is over 40% and growing. We are fortunate to have new medications that on average lead to 15% or greater weight loss when combined with lifestyle modification.
However, these medications are expensive and the limited insurance coverage currently available may not improve. From a patient experience perspective, it’s distressing to have to discontinue treatments that have helped to achieve a healthier weight and then experience regain.
People need better access to evidence-based treatments for obesity, which include lifestyle interventions, anti-obesity medications, and bariatric procedures. Successful treatment of obesity should include a personalized, patient-centered approach that may require a combination of therapies, such as medications and surgery, for lasting weight control.
Dr. Almandoz is associate professor, department of internal medicine, division of endocrinology; medical director, weight wellness program, University of Texas Southwestern, Dallas. He disclosed ties with Novo Nordisk and Eli Lilly. Follow Dr. Almandoz on Twitter: @JaimeAlmandoz.
A version of this article originally appeared on Medscape.com.
Some of these medicines are approved for treating obesity (Wegovy), whereas others are approved for type 2 diabetes (Ozempic and Mounjaro). Tirzepatide (Mounjaro) has been fast-tracked for approval for weight loss by the U.S. Food and Drug Administration this year, and in the first of the series of studies looking at its effect on obesity, the SURMOUNT-1 trial, tirzepatide demonstrated a mean weight loss of around 22% in people without diabetes, spurring significant off-label use.
Our offices are full of patients who have taken these medications, with unprecedented improvements in their weight, cardiometabolic health, and quality of life. What happens when patients stop taking these medications? Or more importantly, why stop them?
Although these drugs are very effective for weight loss and treating diabetes, there can be adverse effects, primarily gastrointestinal, that limit treatment continuation. Nausea is the most common side effect and usually diminishes over time. Slow dose titration and dietary modification can minimize unwanted gastrointestinal side effects.
Drug-induced acute pancreatitis, a rare adverse event requiring patients to stop therapy, was seen in approximately 0.2% of people in clinical trials.
Medications effective but cost prohibitive?
Beyond adverse effects, patients may be forced to stop treatment because of medication cost, changes in insurance coverage, or issues with drug availability.
Two incretin therapies currently approved for treating obesity – liraglutide (Saxenda) and semaglutide (Wegovy) – cost around $1,400 per month. Insurance coverage and manufacturer discounts can make treatment affordable, but anti-obesity medicines aren’t covered by Medicare or by many employer-sponsored commercial plans.
Changes in employment or insurance coverage, or expiration of manufacturer copay cards, may require patients to stop or change therapies. The increased prescribing and overall expense of these drugs have prompted insurance plans and self-insured groups to consider whether providing coverage for these medications is sustainable.
Limited coverage has led to significant off-label prescribing of incretin therapies that aren’t approved for treating obesity (for instance, Ozempic and Mounjaro) and compounding pharmacies selling peptides that allegedly contain the active pharmaceutical ingredients. High demand for these medications has created significant supply shortages over the past year, causing many people to be without treatment for significant periods of time, as reported by this news organization.
Recently, I saw a patient who lost more than 30 pounds with semaglutide (Wegovy). She then changed employers and the medication was no longer covered. She gained back almost 10 pounds over 3 months and was prescribed tirzepatide (Mounjaro) off-label for weight loss by another provider, using a manufacturer discount card to make the medication affordable. The patient did well with the new regimen and lost about 20 pounds, but the pharmacy stopped filling the prescription when changes were made to the discount card. Afraid of regaining the weight, she came to see us as a new patient to discuss her options with her lack of coverage for anti-obesity medications.
Stopping equals weight regain
Obesity is a chronic disease like hypertension. It responds to treatment and when people stop taking these anti-obesity medications, this is generally associated with increased appetite and less satiety, and there is subsequent weight regain and a recurrence in excess weight-related complications.
The STEP-1 trial extension showed an initial mean body weight reduction of 17.3% with weekly semaglutide 2.4 mg over 1 year. On average, two-thirds of the weight lost was regained by participants within 1 year of stopping semaglutide and the study’s lifestyle intervention. Many of the improvements seen in cardiometabolic variables, like blood glucose and blood pressure, similarly reverted to baseline.
There are also 2-year data from the STEP-5 trial with semaglutide; 3-year data from the SCALE trial with liraglutide; and 5-year nonrandomized data with multiple agents that show durable, clinically significant weight loss from medical therapies for obesity.
These data together demonstrate that medications are effective for durable weight loss if they are continued. However, this is not how obesity is currently treated. Anti-obesity medications are prescribed to less than 3% of eligible people in the United States, and the average duration of therapy is less than 90 days. This treatment length isn’t sufficient to see the full benefits most medications offer and certainly doesn’t support long-term weight maintenance.
A recent study showed that, in addition to maintaining weight loss from medical therapies, incretin-containing anti-obesity medication regimens were effective for treating weight regain and facilitating healthier weight after bariatric surgery.
Chronic therapy is needed for weight maintenance because several neurohormonal changes occur owing to weight loss. Metabolic adaptation is the relative reduction in energy expenditure, below what would be expected, in people after weight loss. When this is combined with physiologic changes that increase appetite and decrease satiety, many people create a positive energy balance that results in weight regain. This has been observed in reality TV shows such as “The Biggest Loser”: It’s biology, not willpower.
Unfortunately, many people – including health care providers – don’t understand how these changes promote weight regain and patients are too often blamed when their weight goes back up after medications are stopped. This blame is greatly misinformed by weight-biased beliefs that people with obesity are lazy and lack self-control for weight loss or maintenance. Nobody would be surprised if someone’s blood pressure went up if their antihypertensive medications were stopped. Why do we think so differently when treating obesity?
The prevalence of obesity in the United States is over 40% and growing. We are fortunate to have new medications that on average lead to 15% or greater weight loss when combined with lifestyle modification.
However, these medications are expensive and the limited insurance coverage currently available may not improve. From a patient experience perspective, it’s distressing to have to discontinue treatments that have helped to achieve a healthier weight and then experience regain.
People need better access to evidence-based treatments for obesity, which include lifestyle interventions, anti-obesity medications, and bariatric procedures. Successful treatment of obesity should include a personalized, patient-centered approach that may require a combination of therapies, such as medications and surgery, for lasting weight control.
Dr. Almandoz is associate professor, department of internal medicine, division of endocrinology; medical director, weight wellness program, University of Texas Southwestern, Dallas. He disclosed ties with Novo Nordisk and Eli Lilly. Follow Dr. Almandoz on Twitter: @JaimeAlmandoz.
A version of this article originally appeared on Medscape.com.
Some of these medicines are approved for treating obesity (Wegovy), whereas others are approved for type 2 diabetes (Ozempic and Mounjaro). Tirzepatide (Mounjaro) has been fast-tracked for approval for weight loss by the U.S. Food and Drug Administration this year, and in the first of the series of studies looking at its effect on obesity, the SURMOUNT-1 trial, tirzepatide demonstrated a mean weight loss of around 22% in people without diabetes, spurring significant off-label use.
Our offices are full of patients who have taken these medications, with unprecedented improvements in their weight, cardiometabolic health, and quality of life. What happens when patients stop taking these medications? Or more importantly, why stop them?
Although these drugs are very effective for weight loss and treating diabetes, there can be adverse effects, primarily gastrointestinal, that limit treatment continuation. Nausea is the most common side effect and usually diminishes over time. Slow dose titration and dietary modification can minimize unwanted gastrointestinal side effects.
Drug-induced acute pancreatitis, a rare adverse event requiring patients to stop therapy, was seen in approximately 0.2% of people in clinical trials.
Medications effective but cost prohibitive?
Beyond adverse effects, patients may be forced to stop treatment because of medication cost, changes in insurance coverage, or issues with drug availability.
Two incretin therapies currently approved for treating obesity – liraglutide (Saxenda) and semaglutide (Wegovy) – cost around $1,400 per month. Insurance coverage and manufacturer discounts can make treatment affordable, but anti-obesity medicines aren’t covered by Medicare or by many employer-sponsored commercial plans.
Changes in employment or insurance coverage, or expiration of manufacturer copay cards, may require patients to stop or change therapies. The increased prescribing and overall expense of these drugs have prompted insurance plans and self-insured groups to consider whether providing coverage for these medications is sustainable.
Limited coverage has led to significant off-label prescribing of incretin therapies that aren’t approved for treating obesity (for instance, Ozempic and Mounjaro) and compounding pharmacies selling peptides that allegedly contain the active pharmaceutical ingredients. High demand for these medications has created significant supply shortages over the past year, causing many people to be without treatment for significant periods of time, as reported by this news organization.
Recently, I saw a patient who lost more than 30 pounds with semaglutide (Wegovy). She then changed employers and the medication was no longer covered. She gained back almost 10 pounds over 3 months and was prescribed tirzepatide (Mounjaro) off-label for weight loss by another provider, using a manufacturer discount card to make the medication affordable. The patient did well with the new regimen and lost about 20 pounds, but the pharmacy stopped filling the prescription when changes were made to the discount card. Afraid of regaining the weight, she came to see us as a new patient to discuss her options with her lack of coverage for anti-obesity medications.
Stopping equals weight regain
Obesity is a chronic disease like hypertension. It responds to treatment and when people stop taking these anti-obesity medications, this is generally associated with increased appetite and less satiety, and there is subsequent weight regain and a recurrence in excess weight-related complications.
The STEP-1 trial extension showed an initial mean body weight reduction of 17.3% with weekly semaglutide 2.4 mg over 1 year. On average, two-thirds of the weight lost was regained by participants within 1 year of stopping semaglutide and the study’s lifestyle intervention. Many of the improvements seen in cardiometabolic variables, like blood glucose and blood pressure, similarly reverted to baseline.
There are also 2-year data from the STEP-5 trial with semaglutide; 3-year data from the SCALE trial with liraglutide; and 5-year nonrandomized data with multiple agents that show durable, clinically significant weight loss from medical therapies for obesity.
These data together demonstrate that medications are effective for durable weight loss if they are continued. However, this is not how obesity is currently treated. Anti-obesity medications are prescribed to less than 3% of eligible people in the United States, and the average duration of therapy is less than 90 days. This treatment length isn’t sufficient to see the full benefits most medications offer and certainly doesn’t support long-term weight maintenance.
A recent study showed that, in addition to maintaining weight loss from medical therapies, incretin-containing anti-obesity medication regimens were effective for treating weight regain and facilitating healthier weight after bariatric surgery.
Chronic therapy is needed for weight maintenance because several neurohormonal changes occur owing to weight loss. Metabolic adaptation is the relative reduction in energy expenditure, below what would be expected, in people after weight loss. When this is combined with physiologic changes that increase appetite and decrease satiety, many people create a positive energy balance that results in weight regain. This has been observed in reality TV shows such as “The Biggest Loser”: It’s biology, not willpower.
Unfortunately, many people – including health care providers – don’t understand how these changes promote weight regain and patients are too often blamed when their weight goes back up after medications are stopped. This blame is greatly misinformed by weight-biased beliefs that people with obesity are lazy and lack self-control for weight loss or maintenance. Nobody would be surprised if someone’s blood pressure went up if their antihypertensive medications were stopped. Why do we think so differently when treating obesity?
The prevalence of obesity in the United States is over 40% and growing. We are fortunate to have new medications that on average lead to 15% or greater weight loss when combined with lifestyle modification.
However, these medications are expensive and the limited insurance coverage currently available may not improve. From a patient experience perspective, it’s distressing to have to discontinue treatments that have helped to achieve a healthier weight and then experience regain.
People need better access to evidence-based treatments for obesity, which include lifestyle interventions, anti-obesity medications, and bariatric procedures. Successful treatment of obesity should include a personalized, patient-centered approach that may require a combination of therapies, such as medications and surgery, for lasting weight control.
Dr. Almandoz is associate professor, department of internal medicine, division of endocrinology; medical director, weight wellness program, University of Texas Southwestern, Dallas. He disclosed ties with Novo Nordisk and Eli Lilly. Follow Dr. Almandoz on Twitter: @JaimeAlmandoz.
A version of this article originally appeared on Medscape.com.
Osteoporosis drugs may extend life after fracture
Long-term osteoporosis medications are associated with a reduced mortality risk following a fracture, new data suggest.
The findings, from nearly 50,000 individuals in a nationwide Taiwanese database from 2009 until 2018, suggest that alendronate/risedronate, denosumab, and zoledronic acid all result in a significantly lower mortality risk post fracture of 17%-22%, compared with raloxifene and bazedoxifene.
“Treatment for osteoporosis has the potential to minimize mortality risk in people of all ages and sexes for any type of fracture. The longer-acting treatments could lower mortality risk,” wrote Chih-Hsing Wu, MD, of the Institute of Gerontology at National Cheng Kung University, Tainan, Taiwan, and colleagues.
The findings have been published online in the Journal of Clinical Endocrinology and Metabolism.
Robert A. Adler, MD, who is chief of endocrinology at the Central Virginia Veterans Affairs Health Care System, Richmond, told this news organization that he hopes these new findings from a “really good database ... may be helpful in talking to a patient about the pros and cons of taking these drugs.”
“Patients have been made very fearful of the unusual side effects, particularly of the antiresorptive drugs,” which he notes include the rare adverse effects of jaw necrosis and atypical femoral fracture, which occur in about 1 per 10,000 patient-years.
“And because of that we have a hard time convincing people to want to take the drug in the first place or to stay on the drug once they start,” said Dr. Adler, who stressed that his viewpoints are his own and not representative of the VA.
“These data should help reinforce the advice already given in professional guidelines that their benefit outweighs any risks,” he stresses.
Dr. Adler also pointed out that both bisphosphonates included in the study, alendronate and zoledronic acid, are now available as generics and therefore inexpensive, but the latter can be subject to facility fees depending on where the infusion is delivered.
He added that hip fracture, in particular, triples the overall 1-year mortality risk in women aged 75-84 years and quadruples the risk in men. The study’s findings suggest that bisphosphonates, in particular, have pleiotropic effects beyond the bone; however, the underlying mechanisms are hard to determine.
“We don’t know all the reasons why people die after a fracture. These are older people who often have multiple medical problems, so it’s hard to dissect that out,” he said.
But whatever the mechanism for the salutary effect of the drugs, Dr. Adler said: “This is one other factor that might change people’s minds. You’re less likely to die. Well, that’s pretty good.”
‘Denosumab is a more potent antiresorptive than bisphosphonates’
Dr. Wu and colleagues analyzed data for individuals from Taiwan’s National Health Insurance Research Database. Between 2009 and 2017, 219,461 individuals had been newly diagnosed with an osteoporotic fracture. Of those, 46,729 were aged 40 and older and had been prescribed at least one anti-osteoporosis medication.
Participants were a mean age of 74.5 years, were 80% women, and 32% died during a mean follow-up of 4.7 years. The most commonly used anti-osteoporosis medications were the bisphosphonates alendronate or risedronate, followed by denosumab and the selective estrogen-receptor modulators (SERMs) daily oral raloxifene or bazedoxifene.
Patients treated with SERMs were used as the reference group because those drugs have been shown to have a neutral effect on mortality.
After adjustments, all but one of the medications had significantly lower mortality risks during follow-up, compared with raloxifene and bazedoxifene.
Compared with SERMs, at all fracture sites, the hazard ratios for mortality were 0.83 for alendronate/risedronate, 0.86 for denosumab, and 0.78 for zoledronic acid. Only ibandronate did not show the same protective effect.
Similar results were found for hip and vertebral fractures analyzed individually.
Women had a lower mortality risk than men.
Dr. Adler wrote an accompanying editorial for the article by Dr. Wu and colleagues.
Regarding the finding of benefit for denosumab, Dr. Adler notes: “I don’t know of another study that found denosumab leads to lower mortality. On the other hand, denosumab is a more potent antiresorptive than bisphosphonates.”
The study was funded by research grants from the Ministry of Science and Technology, Taiwan, partially supported by a research grant from the Taiwanese Osteoporosis Association and grants from National Cheng Kung University Hospital, Taiwan. Dr. Wu has reported receiving honoraria for lectures, attending meetings, and/or travel from Eli Lilly, Roche, Amgen, Merck, Servier, GE Lunar, Harvester, TCM Biotech, and Alvogen/Lotus. Dr. Adler has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Long-term osteoporosis medications are associated with a reduced mortality risk following a fracture, new data suggest.
The findings, from nearly 50,000 individuals in a nationwide Taiwanese database from 2009 until 2018, suggest that alendronate/risedronate, denosumab, and zoledronic acid all result in a significantly lower mortality risk post fracture of 17%-22%, compared with raloxifene and bazedoxifene.
“Treatment for osteoporosis has the potential to minimize mortality risk in people of all ages and sexes for any type of fracture. The longer-acting treatments could lower mortality risk,” wrote Chih-Hsing Wu, MD, of the Institute of Gerontology at National Cheng Kung University, Tainan, Taiwan, and colleagues.
The findings have been published online in the Journal of Clinical Endocrinology and Metabolism.
Robert A. Adler, MD, who is chief of endocrinology at the Central Virginia Veterans Affairs Health Care System, Richmond, told this news organization that he hopes these new findings from a “really good database ... may be helpful in talking to a patient about the pros and cons of taking these drugs.”
“Patients have been made very fearful of the unusual side effects, particularly of the antiresorptive drugs,” which he notes include the rare adverse effects of jaw necrosis and atypical femoral fracture, which occur in about 1 per 10,000 patient-years.
“And because of that we have a hard time convincing people to want to take the drug in the first place or to stay on the drug once they start,” said Dr. Adler, who stressed that his viewpoints are his own and not representative of the VA.
“These data should help reinforce the advice already given in professional guidelines that their benefit outweighs any risks,” he stresses.
Dr. Adler also pointed out that both bisphosphonates included in the study, alendronate and zoledronic acid, are now available as generics and therefore inexpensive, but the latter can be subject to facility fees depending on where the infusion is delivered.
He added that hip fracture, in particular, triples the overall 1-year mortality risk in women aged 75-84 years and quadruples the risk in men. The study’s findings suggest that bisphosphonates, in particular, have pleiotropic effects beyond the bone; however, the underlying mechanisms are hard to determine.
“We don’t know all the reasons why people die after a fracture. These are older people who often have multiple medical problems, so it’s hard to dissect that out,” he said.
But whatever the mechanism for the salutary effect of the drugs, Dr. Adler said: “This is one other factor that might change people’s minds. You’re less likely to die. Well, that’s pretty good.”
‘Denosumab is a more potent antiresorptive than bisphosphonates’
Dr. Wu and colleagues analyzed data for individuals from Taiwan’s National Health Insurance Research Database. Between 2009 and 2017, 219,461 individuals had been newly diagnosed with an osteoporotic fracture. Of those, 46,729 were aged 40 and older and had been prescribed at least one anti-osteoporosis medication.
Participants were a mean age of 74.5 years, were 80% women, and 32% died during a mean follow-up of 4.7 years. The most commonly used anti-osteoporosis medications were the bisphosphonates alendronate or risedronate, followed by denosumab and the selective estrogen-receptor modulators (SERMs) daily oral raloxifene or bazedoxifene.
Patients treated with SERMs were used as the reference group because those drugs have been shown to have a neutral effect on mortality.
After adjustments, all but one of the medications had significantly lower mortality risks during follow-up, compared with raloxifene and bazedoxifene.
Compared with SERMs, at all fracture sites, the hazard ratios for mortality were 0.83 for alendronate/risedronate, 0.86 for denosumab, and 0.78 for zoledronic acid. Only ibandronate did not show the same protective effect.
Similar results were found for hip and vertebral fractures analyzed individually.
Women had a lower mortality risk than men.
Dr. Adler wrote an accompanying editorial for the article by Dr. Wu and colleagues.
Regarding the finding of benefit for denosumab, Dr. Adler notes: “I don’t know of another study that found denosumab leads to lower mortality. On the other hand, denosumab is a more potent antiresorptive than bisphosphonates.”
The study was funded by research grants from the Ministry of Science and Technology, Taiwan, partially supported by a research grant from the Taiwanese Osteoporosis Association and grants from National Cheng Kung University Hospital, Taiwan. Dr. Wu has reported receiving honoraria for lectures, attending meetings, and/or travel from Eli Lilly, Roche, Amgen, Merck, Servier, GE Lunar, Harvester, TCM Biotech, and Alvogen/Lotus. Dr. Adler has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Long-term osteoporosis medications are associated with a reduced mortality risk following a fracture, new data suggest.
The findings, from nearly 50,000 individuals in a nationwide Taiwanese database from 2009 until 2018, suggest that alendronate/risedronate, denosumab, and zoledronic acid all result in a significantly lower mortality risk post fracture of 17%-22%, compared with raloxifene and bazedoxifene.
“Treatment for osteoporosis has the potential to minimize mortality risk in people of all ages and sexes for any type of fracture. The longer-acting treatments could lower mortality risk,” wrote Chih-Hsing Wu, MD, of the Institute of Gerontology at National Cheng Kung University, Tainan, Taiwan, and colleagues.
The findings have been published online in the Journal of Clinical Endocrinology and Metabolism.
Robert A. Adler, MD, who is chief of endocrinology at the Central Virginia Veterans Affairs Health Care System, Richmond, told this news organization that he hopes these new findings from a “really good database ... may be helpful in talking to a patient about the pros and cons of taking these drugs.”
“Patients have been made very fearful of the unusual side effects, particularly of the antiresorptive drugs,” which he notes include the rare adverse effects of jaw necrosis and atypical femoral fracture, which occur in about 1 per 10,000 patient-years.
“And because of that we have a hard time convincing people to want to take the drug in the first place or to stay on the drug once they start,” said Dr. Adler, who stressed that his viewpoints are his own and not representative of the VA.
“These data should help reinforce the advice already given in professional guidelines that their benefit outweighs any risks,” he stresses.
Dr. Adler also pointed out that both bisphosphonates included in the study, alendronate and zoledronic acid, are now available as generics and therefore inexpensive, but the latter can be subject to facility fees depending on where the infusion is delivered.
He added that hip fracture, in particular, triples the overall 1-year mortality risk in women aged 75-84 years and quadruples the risk in men. The study’s findings suggest that bisphosphonates, in particular, have pleiotropic effects beyond the bone; however, the underlying mechanisms are hard to determine.
“We don’t know all the reasons why people die after a fracture. These are older people who often have multiple medical problems, so it’s hard to dissect that out,” he said.
But whatever the mechanism for the salutary effect of the drugs, Dr. Adler said: “This is one other factor that might change people’s minds. You’re less likely to die. Well, that’s pretty good.”
‘Denosumab is a more potent antiresorptive than bisphosphonates’
Dr. Wu and colleagues analyzed data for individuals from Taiwan’s National Health Insurance Research Database. Between 2009 and 2017, 219,461 individuals had been newly diagnosed with an osteoporotic fracture. Of those, 46,729 were aged 40 and older and had been prescribed at least one anti-osteoporosis medication.
Participants were a mean age of 74.5 years, were 80% women, and 32% died during a mean follow-up of 4.7 years. The most commonly used anti-osteoporosis medications were the bisphosphonates alendronate or risedronate, followed by denosumab and the selective estrogen-receptor modulators (SERMs) daily oral raloxifene or bazedoxifene.
Patients treated with SERMs were used as the reference group because those drugs have been shown to have a neutral effect on mortality.
After adjustments, all but one of the medications had significantly lower mortality risks during follow-up, compared with raloxifene and bazedoxifene.
Compared with SERMs, at all fracture sites, the hazard ratios for mortality were 0.83 for alendronate/risedronate, 0.86 for denosumab, and 0.78 for zoledronic acid. Only ibandronate did not show the same protective effect.
Similar results were found for hip and vertebral fractures analyzed individually.
Women had a lower mortality risk than men.
Dr. Adler wrote an accompanying editorial for the article by Dr. Wu and colleagues.
Regarding the finding of benefit for denosumab, Dr. Adler notes: “I don’t know of another study that found denosumab leads to lower mortality. On the other hand, denosumab is a more potent antiresorptive than bisphosphonates.”
The study was funded by research grants from the Ministry of Science and Technology, Taiwan, partially supported by a research grant from the Taiwanese Osteoporosis Association and grants from National Cheng Kung University Hospital, Taiwan. Dr. Wu has reported receiving honoraria for lectures, attending meetings, and/or travel from Eli Lilly, Roche, Amgen, Merck, Servier, GE Lunar, Harvester, TCM Biotech, and Alvogen/Lotus. Dr. Adler has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Dapagliflozin’s HFpEF benefit tied to lower filling pressure
NEW ORLEANS – Treatment of patients with heart failure with preserved ejection fraction (HFpEF) with the SGLT2 inhibitor dapagliflozin (Farxiga) for 24 weeks produced significant and beneficial reductions in left-heart filling pressures in a mechanistic, randomized clinical study.
The findings “provide new insight into the mechanisms underlying the favorable clinical effects of dapagliflozin in patients with HFpEF,” Barry A. Borlaug, MD, said at the joint scientific sessions of the American College of Cardiology and the World Heart Federation. “Elevations in left heart filling pressures at rest and during exercise are fundamental pathophysiologic features of HFpEF,” he noted.
Results from prior studies documented the benefit of dapagliflozin for improving clinical outcomes in patients with HFpEF in the DELIVER trial, and for the related sodium-glucose cotransporter 2 (SGLT2) inhibitor empagliflozin (Jardiance) in the EMPEROR-Preserved trial. The new findings presented by Dr. Borlaug provide evidence from a placebo-controlled, prospective study for one way by which these SGLT2 inhibitors exert this benefit in patients with HFpEF.
The results of his single-center study showed that, in patients with HFpEF who also exhibited “severe” elevations in pulmonary capillary wedge pressure (PCWP) during exercise, 24 weeks of treatment with dapagliflozin led to a significant reduction in PCWP during exercise. The treatment produced an average 6.1–mm Hg drop from baseline compared with control patients who received placebo. A similar pattern occurred when these patients were at rest, when dapagliflozin treatment linked with a significant average reduction in PCWP from baseline of 3.5 mm Hg compared with controls.
Improving a ‘specific and fundamental’ feature of HFpEF
“This fantastic study looked at one of the fundamental aspects of HFpEF,” said John R. Teerlink, MD, designated discussant for the study. “You’ve shown that dapagliflozin targets a specific and fundamental” manifestation of HFpEF by lowering PCWP, said Dr. Teerlink, director of Heart Failure at the San Francisco Veterans Affairs Medical Center.
However, Dr. Teerlink added, the study did not directly address the related question of what physiologic action of dapagliflozin produces this notable drop in PCWP.
“We’re just starting to look at that,” replied Dr. Borlaug, a cardiologist and professor at the Mayo Clinic in Rochester, Minn.
He reported finding an intriguing correlate in the current study linked to the cut in PCWP with dapagliflozin treatment. The SGLT2 inhibitor at a standard daily 10-mg dose produced an average 3.5-kg drop in body weight in the dapagliflozin-treated patients that significantly linked with the changes in PCWP both at rest and during exercise. Dapagliflozin-treated patients also showed a significant reduction from their baseline plasma volume compared with placebo-treated patients, but this “poorly correlated” with the dapagliflozin-linked cuts in PCWP, Dr. Borlaug said.
“I don’t think this means weight loss is the cause of the hemodynamic benefit, but maybe it’s an indicator. When patients [with HFpEF] lose weight, they are in a metabolic state that leads to good changes in hemodynamics,” he suggested. “My guess is that there is probably a combination of many different little things [caused by dapagliflozin treatment of patients with HFpEF] that together result in the 20%-25% relative improvement we see in filling pressure.”
An ‘obese, cardiometabolic’ HFpEF phenotype
The study enrolled patients with HFpEF and a left ventricular ejection fraction of at least 50%, a New York Heart Association functional class of 2 or 3, and a PCWP during exercise of at least 25 mm Hg. Of the 37 evaluable patients, about two-thirds of the patients were women, more than two-thirds were in functional class 3, about 70% were obese, and their average ejection fraction was about 62%. The study excluded patients with HFpEF who also had type 1 diabetes, cardiomyopathy, pericardial disease, or other causes of dyspnea or heart failure.
Dr. Teerlink asked about the generalizability of the findings, as the study cohort seemed to differ in certain respects from the patients enrolled in the DELIVER trial, and because of the many apparently distinct patient phenotypes that exist within the scope of HFpEF.
An “obese, cardiometabolic phenotype” predominated the study cohort, Dr. Borlaug said. “The patients we enrolled look like the HFpEF patients seen in U.S. clinics.” However, he added that “in reality, many [HFpEF phenotypes] coexist in one patient. It’s not that simple,” that every patient with HFpEF can be categorized into a single HFpEF phenotype.
The researchers monitored PCWP invasively with high-fidelity micromanometer catheters.
The study was sponsored by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Borlaug has received research funding from AstraZeneca, as well as from Corvia, GlaxoSmithKline, Medtronic, Mesoblast, Novo Nordisk, and Tenax. Dr. Teerlink has had financial relationships with AstraZeneca, as well as with Amgen, Bayer, Boehringer Ingelheim, Bristol Myers Squibb, Cytokinetics, Medtronic, Merck, Novartis, Servier, and Windtree Therapeutics.
NEW ORLEANS – Treatment of patients with heart failure with preserved ejection fraction (HFpEF) with the SGLT2 inhibitor dapagliflozin (Farxiga) for 24 weeks produced significant and beneficial reductions in left-heart filling pressures in a mechanistic, randomized clinical study.
The findings “provide new insight into the mechanisms underlying the favorable clinical effects of dapagliflozin in patients with HFpEF,” Barry A. Borlaug, MD, said at the joint scientific sessions of the American College of Cardiology and the World Heart Federation. “Elevations in left heart filling pressures at rest and during exercise are fundamental pathophysiologic features of HFpEF,” he noted.
Results from prior studies documented the benefit of dapagliflozin for improving clinical outcomes in patients with HFpEF in the DELIVER trial, and for the related sodium-glucose cotransporter 2 (SGLT2) inhibitor empagliflozin (Jardiance) in the EMPEROR-Preserved trial. The new findings presented by Dr. Borlaug provide evidence from a placebo-controlled, prospective study for one way by which these SGLT2 inhibitors exert this benefit in patients with HFpEF.
The results of his single-center study showed that, in patients with HFpEF who also exhibited “severe” elevations in pulmonary capillary wedge pressure (PCWP) during exercise, 24 weeks of treatment with dapagliflozin led to a significant reduction in PCWP during exercise. The treatment produced an average 6.1–mm Hg drop from baseline compared with control patients who received placebo. A similar pattern occurred when these patients were at rest, when dapagliflozin treatment linked with a significant average reduction in PCWP from baseline of 3.5 mm Hg compared with controls.
Improving a ‘specific and fundamental’ feature of HFpEF
“This fantastic study looked at one of the fundamental aspects of HFpEF,” said John R. Teerlink, MD, designated discussant for the study. “You’ve shown that dapagliflozin targets a specific and fundamental” manifestation of HFpEF by lowering PCWP, said Dr. Teerlink, director of Heart Failure at the San Francisco Veterans Affairs Medical Center.
However, Dr. Teerlink added, the study did not directly address the related question of what physiologic action of dapagliflozin produces this notable drop in PCWP.
“We’re just starting to look at that,” replied Dr. Borlaug, a cardiologist and professor at the Mayo Clinic in Rochester, Minn.
He reported finding an intriguing correlate in the current study linked to the cut in PCWP with dapagliflozin treatment. The SGLT2 inhibitor at a standard daily 10-mg dose produced an average 3.5-kg drop in body weight in the dapagliflozin-treated patients that significantly linked with the changes in PCWP both at rest and during exercise. Dapagliflozin-treated patients also showed a significant reduction from their baseline plasma volume compared with placebo-treated patients, but this “poorly correlated” with the dapagliflozin-linked cuts in PCWP, Dr. Borlaug said.
“I don’t think this means weight loss is the cause of the hemodynamic benefit, but maybe it’s an indicator. When patients [with HFpEF] lose weight, they are in a metabolic state that leads to good changes in hemodynamics,” he suggested. “My guess is that there is probably a combination of many different little things [caused by dapagliflozin treatment of patients with HFpEF] that together result in the 20%-25% relative improvement we see in filling pressure.”
An ‘obese, cardiometabolic’ HFpEF phenotype
The study enrolled patients with HFpEF and a left ventricular ejection fraction of at least 50%, a New York Heart Association functional class of 2 or 3, and a PCWP during exercise of at least 25 mm Hg. Of the 37 evaluable patients, about two-thirds of the patients were women, more than two-thirds were in functional class 3, about 70% were obese, and their average ejection fraction was about 62%. The study excluded patients with HFpEF who also had type 1 diabetes, cardiomyopathy, pericardial disease, or other causes of dyspnea or heart failure.
Dr. Teerlink asked about the generalizability of the findings, as the study cohort seemed to differ in certain respects from the patients enrolled in the DELIVER trial, and because of the many apparently distinct patient phenotypes that exist within the scope of HFpEF.
An “obese, cardiometabolic phenotype” predominated the study cohort, Dr. Borlaug said. “The patients we enrolled look like the HFpEF patients seen in U.S. clinics.” However, he added that “in reality, many [HFpEF phenotypes] coexist in one patient. It’s not that simple,” that every patient with HFpEF can be categorized into a single HFpEF phenotype.
The researchers monitored PCWP invasively with high-fidelity micromanometer catheters.
The study was sponsored by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Borlaug has received research funding from AstraZeneca, as well as from Corvia, GlaxoSmithKline, Medtronic, Mesoblast, Novo Nordisk, and Tenax. Dr. Teerlink has had financial relationships with AstraZeneca, as well as with Amgen, Bayer, Boehringer Ingelheim, Bristol Myers Squibb, Cytokinetics, Medtronic, Merck, Novartis, Servier, and Windtree Therapeutics.
NEW ORLEANS – Treatment of patients with heart failure with preserved ejection fraction (HFpEF) with the SGLT2 inhibitor dapagliflozin (Farxiga) for 24 weeks produced significant and beneficial reductions in left-heart filling pressures in a mechanistic, randomized clinical study.
The findings “provide new insight into the mechanisms underlying the favorable clinical effects of dapagliflozin in patients with HFpEF,” Barry A. Borlaug, MD, said at the joint scientific sessions of the American College of Cardiology and the World Heart Federation. “Elevations in left heart filling pressures at rest and during exercise are fundamental pathophysiologic features of HFpEF,” he noted.
Results from prior studies documented the benefit of dapagliflozin for improving clinical outcomes in patients with HFpEF in the DELIVER trial, and for the related sodium-glucose cotransporter 2 (SGLT2) inhibitor empagliflozin (Jardiance) in the EMPEROR-Preserved trial. The new findings presented by Dr. Borlaug provide evidence from a placebo-controlled, prospective study for one way by which these SGLT2 inhibitors exert this benefit in patients with HFpEF.
The results of his single-center study showed that, in patients with HFpEF who also exhibited “severe” elevations in pulmonary capillary wedge pressure (PCWP) during exercise, 24 weeks of treatment with dapagliflozin led to a significant reduction in PCWP during exercise. The treatment produced an average 6.1–mm Hg drop from baseline compared with control patients who received placebo. A similar pattern occurred when these patients were at rest, when dapagliflozin treatment linked with a significant average reduction in PCWP from baseline of 3.5 mm Hg compared with controls.
Improving a ‘specific and fundamental’ feature of HFpEF
“This fantastic study looked at one of the fundamental aspects of HFpEF,” said John R. Teerlink, MD, designated discussant for the study. “You’ve shown that dapagliflozin targets a specific and fundamental” manifestation of HFpEF by lowering PCWP, said Dr. Teerlink, director of Heart Failure at the San Francisco Veterans Affairs Medical Center.
However, Dr. Teerlink added, the study did not directly address the related question of what physiologic action of dapagliflozin produces this notable drop in PCWP.
“We’re just starting to look at that,” replied Dr. Borlaug, a cardiologist and professor at the Mayo Clinic in Rochester, Minn.
He reported finding an intriguing correlate in the current study linked to the cut in PCWP with dapagliflozin treatment. The SGLT2 inhibitor at a standard daily 10-mg dose produced an average 3.5-kg drop in body weight in the dapagliflozin-treated patients that significantly linked with the changes in PCWP both at rest and during exercise. Dapagliflozin-treated patients also showed a significant reduction from their baseline plasma volume compared with placebo-treated patients, but this “poorly correlated” with the dapagliflozin-linked cuts in PCWP, Dr. Borlaug said.
“I don’t think this means weight loss is the cause of the hemodynamic benefit, but maybe it’s an indicator. When patients [with HFpEF] lose weight, they are in a metabolic state that leads to good changes in hemodynamics,” he suggested. “My guess is that there is probably a combination of many different little things [caused by dapagliflozin treatment of patients with HFpEF] that together result in the 20%-25% relative improvement we see in filling pressure.”
An ‘obese, cardiometabolic’ HFpEF phenotype
The study enrolled patients with HFpEF and a left ventricular ejection fraction of at least 50%, a New York Heart Association functional class of 2 or 3, and a PCWP during exercise of at least 25 mm Hg. Of the 37 evaluable patients, about two-thirds of the patients were women, more than two-thirds were in functional class 3, about 70% were obese, and their average ejection fraction was about 62%. The study excluded patients with HFpEF who also had type 1 diabetes, cardiomyopathy, pericardial disease, or other causes of dyspnea or heart failure.
Dr. Teerlink asked about the generalizability of the findings, as the study cohort seemed to differ in certain respects from the patients enrolled in the DELIVER trial, and because of the many apparently distinct patient phenotypes that exist within the scope of HFpEF.
An “obese, cardiometabolic phenotype” predominated the study cohort, Dr. Borlaug said. “The patients we enrolled look like the HFpEF patients seen in U.S. clinics.” However, he added that “in reality, many [HFpEF phenotypes] coexist in one patient. It’s not that simple,” that every patient with HFpEF can be categorized into a single HFpEF phenotype.
The researchers monitored PCWP invasively with high-fidelity micromanometer catheters.
The study was sponsored by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Borlaug has received research funding from AstraZeneca, as well as from Corvia, GlaxoSmithKline, Medtronic, Mesoblast, Novo Nordisk, and Tenax. Dr. Teerlink has had financial relationships with AstraZeneca, as well as with Amgen, Bayer, Boehringer Ingelheim, Bristol Myers Squibb, Cytokinetics, Medtronic, Merck, Novartis, Servier, and Windtree Therapeutics.
AT ACC 2023
Even small changes in fitness tied to lower mortality risk
Even relatively small changes in cardiorespiratory fitness (CRF) are associated with “considerable” impact on clinical symptoms and mortality risk among individuals with and without cardiovascular disease, new observational data in United States veterans suggest.
“We had a few surprises,” Peter Kokkinos, PhD, Robert Wood Johnson Medical School, New Brunswick, N. J., and the VA Medical Center, Washington, told this news organization. “First, the mortality risk was greatly attenuated in those who were moderate- and high-fit at baseline, despite a decline in fitness over time. In fact, in those with no CVD, the risk was not significantly elevated even when CRF declined by at least one MET [metabolic equivalent of task] for the moderate-fit and two or more METs for the high-fit group.”
“Second,” he said, “Our findings suggest that the impact of CRF on human health is not ephemeral, but rather carries a certain protection over time. Third, the changes in CRF necessary to impact mortality risk are relatively small (> 1.0 METs). This has a substantial clinical and public health significance.”
The study was published online in the Journal of the American College of Cardiology.
CRF up, mortality risk down
Dr. Kokkinos and colleagues analyzed data from 93,060 U.S. veterans; of these, 95% were men (mean age, 61.4 years) and 5% were women (mean age, 57.1 years). Overall, 72% of participants were White; 19.8%, African American; 5.2%, Hispanic; 1.9%, Native American, Asian, or Hawaiian; and 1.2%, unknown.
Participants were assigned to age-specific fitness quartiles based on peak METs achieved on a baseline exercise treadmill test (ETT). Each CRF quartile was stratified based on CRF changes (increase, decrease, no change) on the final ETT, with at least two ETT assessments at least 1 year apart.
The mean follow-up was 5.8 years (663,522 person-years), during which 18,302 deaths (19.7%) occurred, for an average annual mortality rate of 27.6 events per 1,000 person-years.
CRF was unchanged in 25.1% of the cohort, increased in 29.3%, and decreased in 45.6%. The trend was similar for those with and without CVD.
Significant differences were seen in all variables across CRF categories. In general, body weight, body mass index, CVD risk factors, and overall disease burden were progressively more unfavorable for those in the lowest CRF categories.
Conversely, medication use was progressively higher among those in low CRF categories.
After adjustment, higher CRF was inversely related to mortality risk for the entire cohort, with and without CVD. Cumulative survival rates across CRF categories declined progressively with increased fitness.
For patients with CVD (hazard ratio, 1.11), other significant predictors of all-cause mortality for patients were age (HR, 1.07), body mass index (HR, 0.98), chronic kidney disease (HR, 1.85), smoking (HR, 1.57), type 2 diabetes (HR, 1.42), hypertension (HR, 1.39), and cancers (HR, 1.37).
Generally, changes in CRF of at least 1.0 MET were associated with inverse and proportionate changes in mortality risk, regardless of baseline CRF status. For example, they note, a CRF decline of > 2.0 METs was associated with a 74% increased mortality risk for low-fit individuals with CVD, and a 69% increase for those without CVD.
A second analysis was done after excluding patients whose CRF declined and who died within 2 years of their last ETT, to account for the possibility that higher mortality rates and CRF declines were consequences of underlying disease (reverse causality). The association between changes in CRF and mortality risk persisted and remained similar to that observed in the entire cohort.
The authors add, “It is noteworthy that CRF increased by at least 1 MET in approximately 29% of the participants in the current study and decreased in approximately 46% of participants. This finding underscores the need to promote physical activity to maintain or increase CRF levels in middle-aged and older individuals.”
“Our findings make a persuasive argument that CRF is a strong and independent determinant of all-cause mortality risk, independent of genetic factors,” Dr. Kokkinos said. “We know that CRF is determined to some degree by genetic factors. However, improvements in aerobic capacity or CRF over time are largely the outcomes of regular engagement in aerobic activities of adequate intensity and volume.”
“Conversely,” he said, “a decline in CRF is likely the result of sedentary behavior, the onset of a chronic condition, or aging.”
If genetics were the sole contributor to mortality risk, then changes in CRF would not influence mortality risk, he concluded.
CRF impact “woefully underestimated”
Barry A. Franklin, PhD, past chair of both the American Heart Association’s Council on Physical Activity and Metabolism and the National Advocacy Committee, said the study substantiates previous smaller studies and is a “seminal” work.
“CRF is woefully underestimated as an index of health outcomes and survival,” said Dr. Franklin, director of preventive cardiology and cardiac rehabilitation at Beaumont Health in Royal Oak, Mich. “Moderate to vigorous physical activity should be regularly promoted by the medical community.”
Dr. Franklin’s recent review, published in Mayo Clinic Proceedings, provides evidence for other exercise benefits that clinicians may not be aware of, he noted. These include:
- Each 1 MET increase in CRF is generally associated with approximately 16% reduction in mortality.
- At any given risk factor profile or coronary calcium score, unfit people have 2-3 times the mortality as their fit counterparts.
- Fitness is inversely related to annual health care costs (each 1 MET increase in CRF is associated with approximately 6% lower annual health care costs).
- Physically active people hospitalized with acute coronary syndromes have better short-term outcomes (likely because of a phenomenon called ‘exercise preconditioning’).
- Fit people who undergo elective or emergent surgical procedures have better outcomes.
- Regular physical activity is a common characteristic in population subsets who routinely live into their 90s and to 100+.
Dr. Franklin had this advice for clinicians seeking to promote CRF increases of 1 MET or more among patients: “Sedentary people who embark on a walking program, who over time increase their walking speed to 3 mph or faster, invariably show at least a 1 MET increase in CRF during subsequent peak or symptom-limited treadmill testing.”
“Another general rule is that if an exercise program decreases heart rate at a given or fixed workload by about 10 beats per minute [bpm], the same treadmill workload that initially was accomplished at a heart rate of 120 bpm is now being accomplished at a heart rate of 110 bpm,” likely resulting in about a 1 MET increase in fitness.
“Accordingly,” he added, “a 20-bpm decrease would suggest a 2 MET increase in fitness!”
In a related editorial, Leonard A. Kaminsky, Ball State University, Muncie, Ind. and colleagues, write, “We agree with and believe the conclusion, reached by Kokkinos et al., bears repeating. We (again) call on both clinicians and public health professionals to adopt CRF as a key health indicator.”
“This should be done by coupling routine assessments of CRF with continued advocacy for promoting physical activity as an essential healthy lifestyle behavior,” they write.
No funding or relevant financial relationships were disclosed.
A version of this article first appeared on Medscape.com.
Even relatively small changes in cardiorespiratory fitness (CRF) are associated with “considerable” impact on clinical symptoms and mortality risk among individuals with and without cardiovascular disease, new observational data in United States veterans suggest.
“We had a few surprises,” Peter Kokkinos, PhD, Robert Wood Johnson Medical School, New Brunswick, N. J., and the VA Medical Center, Washington, told this news organization. “First, the mortality risk was greatly attenuated in those who were moderate- and high-fit at baseline, despite a decline in fitness over time. In fact, in those with no CVD, the risk was not significantly elevated even when CRF declined by at least one MET [metabolic equivalent of task] for the moderate-fit and two or more METs for the high-fit group.”
“Second,” he said, “Our findings suggest that the impact of CRF on human health is not ephemeral, but rather carries a certain protection over time. Third, the changes in CRF necessary to impact mortality risk are relatively small (> 1.0 METs). This has a substantial clinical and public health significance.”
The study was published online in the Journal of the American College of Cardiology.
CRF up, mortality risk down
Dr. Kokkinos and colleagues analyzed data from 93,060 U.S. veterans; of these, 95% were men (mean age, 61.4 years) and 5% were women (mean age, 57.1 years). Overall, 72% of participants were White; 19.8%, African American; 5.2%, Hispanic; 1.9%, Native American, Asian, or Hawaiian; and 1.2%, unknown.
Participants were assigned to age-specific fitness quartiles based on peak METs achieved on a baseline exercise treadmill test (ETT). Each CRF quartile was stratified based on CRF changes (increase, decrease, no change) on the final ETT, with at least two ETT assessments at least 1 year apart.
The mean follow-up was 5.8 years (663,522 person-years), during which 18,302 deaths (19.7%) occurred, for an average annual mortality rate of 27.6 events per 1,000 person-years.
CRF was unchanged in 25.1% of the cohort, increased in 29.3%, and decreased in 45.6%. The trend was similar for those with and without CVD.
Significant differences were seen in all variables across CRF categories. In general, body weight, body mass index, CVD risk factors, and overall disease burden were progressively more unfavorable for those in the lowest CRF categories.
Conversely, medication use was progressively higher among those in low CRF categories.
After adjustment, higher CRF was inversely related to mortality risk for the entire cohort, with and without CVD. Cumulative survival rates across CRF categories declined progressively with increased fitness.
For patients with CVD (hazard ratio, 1.11), other significant predictors of all-cause mortality for patients were age (HR, 1.07), body mass index (HR, 0.98), chronic kidney disease (HR, 1.85), smoking (HR, 1.57), type 2 diabetes (HR, 1.42), hypertension (HR, 1.39), and cancers (HR, 1.37).
Generally, changes in CRF of at least 1.0 MET were associated with inverse and proportionate changes in mortality risk, regardless of baseline CRF status. For example, they note, a CRF decline of > 2.0 METs was associated with a 74% increased mortality risk for low-fit individuals with CVD, and a 69% increase for those without CVD.
A second analysis was done after excluding patients whose CRF declined and who died within 2 years of their last ETT, to account for the possibility that higher mortality rates and CRF declines were consequences of underlying disease (reverse causality). The association between changes in CRF and mortality risk persisted and remained similar to that observed in the entire cohort.
The authors add, “It is noteworthy that CRF increased by at least 1 MET in approximately 29% of the participants in the current study and decreased in approximately 46% of participants. This finding underscores the need to promote physical activity to maintain or increase CRF levels in middle-aged and older individuals.”
“Our findings make a persuasive argument that CRF is a strong and independent determinant of all-cause mortality risk, independent of genetic factors,” Dr. Kokkinos said. “We know that CRF is determined to some degree by genetic factors. However, improvements in aerobic capacity or CRF over time are largely the outcomes of regular engagement in aerobic activities of adequate intensity and volume.”
“Conversely,” he said, “a decline in CRF is likely the result of sedentary behavior, the onset of a chronic condition, or aging.”
If genetics were the sole contributor to mortality risk, then changes in CRF would not influence mortality risk, he concluded.
CRF impact “woefully underestimated”
Barry A. Franklin, PhD, past chair of both the American Heart Association’s Council on Physical Activity and Metabolism and the National Advocacy Committee, said the study substantiates previous smaller studies and is a “seminal” work.
“CRF is woefully underestimated as an index of health outcomes and survival,” said Dr. Franklin, director of preventive cardiology and cardiac rehabilitation at Beaumont Health in Royal Oak, Mich. “Moderate to vigorous physical activity should be regularly promoted by the medical community.”
Dr. Franklin’s recent review, published in Mayo Clinic Proceedings, provides evidence for other exercise benefits that clinicians may not be aware of, he noted. These include:
- Each 1 MET increase in CRF is generally associated with approximately 16% reduction in mortality.
- At any given risk factor profile or coronary calcium score, unfit people have 2-3 times the mortality as their fit counterparts.
- Fitness is inversely related to annual health care costs (each 1 MET increase in CRF is associated with approximately 6% lower annual health care costs).
- Physically active people hospitalized with acute coronary syndromes have better short-term outcomes (likely because of a phenomenon called ‘exercise preconditioning’).
- Fit people who undergo elective or emergent surgical procedures have better outcomes.
- Regular physical activity is a common characteristic in population subsets who routinely live into their 90s and to 100+.
Dr. Franklin had this advice for clinicians seeking to promote CRF increases of 1 MET or more among patients: “Sedentary people who embark on a walking program, who over time increase their walking speed to 3 mph or faster, invariably show at least a 1 MET increase in CRF during subsequent peak or symptom-limited treadmill testing.”
“Another general rule is that if an exercise program decreases heart rate at a given or fixed workload by about 10 beats per minute [bpm], the same treadmill workload that initially was accomplished at a heart rate of 120 bpm is now being accomplished at a heart rate of 110 bpm,” likely resulting in about a 1 MET increase in fitness.
“Accordingly,” he added, “a 20-bpm decrease would suggest a 2 MET increase in fitness!”
In a related editorial, Leonard A. Kaminsky, Ball State University, Muncie, Ind. and colleagues, write, “We agree with and believe the conclusion, reached by Kokkinos et al., bears repeating. We (again) call on both clinicians and public health professionals to adopt CRF as a key health indicator.”
“This should be done by coupling routine assessments of CRF with continued advocacy for promoting physical activity as an essential healthy lifestyle behavior,” they write.
No funding or relevant financial relationships were disclosed.
A version of this article first appeared on Medscape.com.
Even relatively small changes in cardiorespiratory fitness (CRF) are associated with “considerable” impact on clinical symptoms and mortality risk among individuals with and without cardiovascular disease, new observational data in United States veterans suggest.
“We had a few surprises,” Peter Kokkinos, PhD, Robert Wood Johnson Medical School, New Brunswick, N. J., and the VA Medical Center, Washington, told this news organization. “First, the mortality risk was greatly attenuated in those who were moderate- and high-fit at baseline, despite a decline in fitness over time. In fact, in those with no CVD, the risk was not significantly elevated even when CRF declined by at least one MET [metabolic equivalent of task] for the moderate-fit and two or more METs for the high-fit group.”
“Second,” he said, “Our findings suggest that the impact of CRF on human health is not ephemeral, but rather carries a certain protection over time. Third, the changes in CRF necessary to impact mortality risk are relatively small (> 1.0 METs). This has a substantial clinical and public health significance.”
The study was published online in the Journal of the American College of Cardiology.
CRF up, mortality risk down
Dr. Kokkinos and colleagues analyzed data from 93,060 U.S. veterans; of these, 95% were men (mean age, 61.4 years) and 5% were women (mean age, 57.1 years). Overall, 72% of participants were White; 19.8%, African American; 5.2%, Hispanic; 1.9%, Native American, Asian, or Hawaiian; and 1.2%, unknown.
Participants were assigned to age-specific fitness quartiles based on peak METs achieved on a baseline exercise treadmill test (ETT). Each CRF quartile was stratified based on CRF changes (increase, decrease, no change) on the final ETT, with at least two ETT assessments at least 1 year apart.
The mean follow-up was 5.8 years (663,522 person-years), during which 18,302 deaths (19.7%) occurred, for an average annual mortality rate of 27.6 events per 1,000 person-years.
CRF was unchanged in 25.1% of the cohort, increased in 29.3%, and decreased in 45.6%. The trend was similar for those with and without CVD.
Significant differences were seen in all variables across CRF categories. In general, body weight, body mass index, CVD risk factors, and overall disease burden were progressively more unfavorable for those in the lowest CRF categories.
Conversely, medication use was progressively higher among those in low CRF categories.
After adjustment, higher CRF was inversely related to mortality risk for the entire cohort, with and without CVD. Cumulative survival rates across CRF categories declined progressively with increased fitness.
For patients with CVD (hazard ratio, 1.11), other significant predictors of all-cause mortality for patients were age (HR, 1.07), body mass index (HR, 0.98), chronic kidney disease (HR, 1.85), smoking (HR, 1.57), type 2 diabetes (HR, 1.42), hypertension (HR, 1.39), and cancers (HR, 1.37).
Generally, changes in CRF of at least 1.0 MET were associated with inverse and proportionate changes in mortality risk, regardless of baseline CRF status. For example, they note, a CRF decline of > 2.0 METs was associated with a 74% increased mortality risk for low-fit individuals with CVD, and a 69% increase for those without CVD.
A second analysis was done after excluding patients whose CRF declined and who died within 2 years of their last ETT, to account for the possibility that higher mortality rates and CRF declines were consequences of underlying disease (reverse causality). The association between changes in CRF and mortality risk persisted and remained similar to that observed in the entire cohort.
The authors add, “It is noteworthy that CRF increased by at least 1 MET in approximately 29% of the participants in the current study and decreased in approximately 46% of participants. This finding underscores the need to promote physical activity to maintain or increase CRF levels in middle-aged and older individuals.”
“Our findings make a persuasive argument that CRF is a strong and independent determinant of all-cause mortality risk, independent of genetic factors,” Dr. Kokkinos said. “We know that CRF is determined to some degree by genetic factors. However, improvements in aerobic capacity or CRF over time are largely the outcomes of regular engagement in aerobic activities of adequate intensity and volume.”
“Conversely,” he said, “a decline in CRF is likely the result of sedentary behavior, the onset of a chronic condition, or aging.”
If genetics were the sole contributor to mortality risk, then changes in CRF would not influence mortality risk, he concluded.
CRF impact “woefully underestimated”
Barry A. Franklin, PhD, past chair of both the American Heart Association’s Council on Physical Activity and Metabolism and the National Advocacy Committee, said the study substantiates previous smaller studies and is a “seminal” work.
“CRF is woefully underestimated as an index of health outcomes and survival,” said Dr. Franklin, director of preventive cardiology and cardiac rehabilitation at Beaumont Health in Royal Oak, Mich. “Moderate to vigorous physical activity should be regularly promoted by the medical community.”
Dr. Franklin’s recent review, published in Mayo Clinic Proceedings, provides evidence for other exercise benefits that clinicians may not be aware of, he noted. These include:
- Each 1 MET increase in CRF is generally associated with approximately 16% reduction in mortality.
- At any given risk factor profile or coronary calcium score, unfit people have 2-3 times the mortality as their fit counterparts.
- Fitness is inversely related to annual health care costs (each 1 MET increase in CRF is associated with approximately 6% lower annual health care costs).
- Physically active people hospitalized with acute coronary syndromes have better short-term outcomes (likely because of a phenomenon called ‘exercise preconditioning’).
- Fit people who undergo elective or emergent surgical procedures have better outcomes.
- Regular physical activity is a common characteristic in population subsets who routinely live into their 90s and to 100+.
Dr. Franklin had this advice for clinicians seeking to promote CRF increases of 1 MET or more among patients: “Sedentary people who embark on a walking program, who over time increase their walking speed to 3 mph or faster, invariably show at least a 1 MET increase in CRF during subsequent peak or symptom-limited treadmill testing.”
“Another general rule is that if an exercise program decreases heart rate at a given or fixed workload by about 10 beats per minute [bpm], the same treadmill workload that initially was accomplished at a heart rate of 120 bpm is now being accomplished at a heart rate of 110 bpm,” likely resulting in about a 1 MET increase in fitness.
“Accordingly,” he added, “a 20-bpm decrease would suggest a 2 MET increase in fitness!”
In a related editorial, Leonard A. Kaminsky, Ball State University, Muncie, Ind. and colleagues, write, “We agree with and believe the conclusion, reached by Kokkinos et al., bears repeating. We (again) call on both clinicians and public health professionals to adopt CRF as a key health indicator.”
“This should be done by coupling routine assessments of CRF with continued advocacy for promoting physical activity as an essential healthy lifestyle behavior,” they write.
No funding or relevant financial relationships were disclosed.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Tooth loss and diabetes together hasten mental decline
most specifically in those 65-74 years of age, new findings suggest.
The data come from a 12-year follow-up of older adults in a nationally representative U.S. survey.
“From a clinical perspective, our study demonstrates the importance of improving access to dental health care and integrating primary dental and medical care. Health care professionals and family caregivers should pay close attention to the cognitive status of diabetic older adults with poor oral health status,” lead author Bei Wu, PhD, of New York University, said in an interview. Dr. Wu is the Dean’s Professor in Global Health and codirector of the NYU Aging Incubator.
Moreover, said Dr. Wu: “For individuals with both poor oral health and diabetes, regular dental visits should be encouraged in addition to adherence to the diabetes self-care protocol.”
Diabetes has long been recognized as a risk factor for cognitive decline, but the findings have been inconsistent for different age groups. Tooth loss has also been linked to cognitive decline and dementia, as well as diabetes.
The mechanisms aren’t entirely clear, but “co-occurring diabetes and poor oral health may increase the risk for dementia, possibly via the potentially interrelated pathways of chronic inflammation and cardiovascular risk factors,” Dr. Wu said.
The new study, published in the Journal of Dental Research, is the first to examine the relationships between all three conditions by age group.
Diabetes, edentulism, and cognitive decline
The data came from a total of 9,948 participants in the Health and Retirement Study (HRS) from 2006 to 2018. At baseline, 5,440 participants were aged 65-74 years, 3,300 were aged 75-84, and 1,208 were aged 85 years or older.
They were assessed every 2 years using the 35-point Telephone Survey for Cognitive Status, which included tests of immediate and delayed word recall, repeated subtracting by 7, counting backward from 20, naming objects, and naming the president and vice president of the U.S. As might be expected, the youngest group scored the highest, averaging 23 points, while the oldest group scored lowest, at 18.5 points.
Participants were also asked if they had ever been told by a doctor that they have diabetes. Another question was: “Have you lost all of your upper and lower natural permanent teeth?”
The condition of having no teeth is known as edentulism.
The percentages of participants who reported having both diabetes and edentulism were 6.0%, 6.7%, and 5.0% for those aged 65-74 years, 75-84 years, and 85 years or older, respectively. The proportions with neither of those conditions were 63.5%, 60.4%, and 58.3% in those three age groups, respectively (P < .001).
Compared with their counterparts with neither diabetes nor edentulism at baseline, older adults with both conditions aged 65-74 years (P < .001) and aged 75-84 years had worse cognitive function (P < .001).
In terms of the rate of cognitive decline, compared with those with neither condition from the same age cohort, older adults aged 65-74 years with both conditions declined at a higher rate (P < .001).
Having diabetes alone led to accelerated cognitive decline in older adults aged 65-74 years (P < .001). Having edentulism alone led to accelerated decline in older adults aged 65-74 years (P < .001) and older adults aged 75-84 years (P < 0.01).
“Our study finds the co-occurrence of diabetes and edentulism led to a worse cognitive function and a faster cognitive decline in older adults aged 65-74 years,” say Wu and colleagues.
Study limitations: Better data needed
The study has several limitations, most of them due to the data source. For example, while the HRS collects detailed information on cognitive status, edentulism is its only measure of oral health. There were no data on whether individuals had replacements such as dentures or implants that would affect their ability to eat, which could influence other health factors.
“I have made repeated appeals for federal funding to collect more oral health-related information in large national surveys,” Dr. Wu told this news organization.
Similarly, assessments of diabetes status such as hemoglobin A1c were only available for small subsets and not sufficient to demonstrate statistical significance, she explained.
Dr. Wu suggested that both oral health and cognitive screening might be included in the “Welcome to Medicare” preventive visit. In addition, “Oral hygiene practice should also be highlighted to improve cognitive health. Developing dental care interventions and programs are needed for reducing the societal cost of dementia.”
The study was partially supported by the National Institutes of Health. The authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
most specifically in those 65-74 years of age, new findings suggest.
The data come from a 12-year follow-up of older adults in a nationally representative U.S. survey.
“From a clinical perspective, our study demonstrates the importance of improving access to dental health care and integrating primary dental and medical care. Health care professionals and family caregivers should pay close attention to the cognitive status of diabetic older adults with poor oral health status,” lead author Bei Wu, PhD, of New York University, said in an interview. Dr. Wu is the Dean’s Professor in Global Health and codirector of the NYU Aging Incubator.
Moreover, said Dr. Wu: “For individuals with both poor oral health and diabetes, regular dental visits should be encouraged in addition to adherence to the diabetes self-care protocol.”
Diabetes has long been recognized as a risk factor for cognitive decline, but the findings have been inconsistent for different age groups. Tooth loss has also been linked to cognitive decline and dementia, as well as diabetes.
The mechanisms aren’t entirely clear, but “co-occurring diabetes and poor oral health may increase the risk for dementia, possibly via the potentially interrelated pathways of chronic inflammation and cardiovascular risk factors,” Dr. Wu said.
The new study, published in the Journal of Dental Research, is the first to examine the relationships between all three conditions by age group.
Diabetes, edentulism, and cognitive decline
The data came from a total of 9,948 participants in the Health and Retirement Study (HRS) from 2006 to 2018. At baseline, 5,440 participants were aged 65-74 years, 3,300 were aged 75-84, and 1,208 were aged 85 years or older.
They were assessed every 2 years using the 35-point Telephone Survey for Cognitive Status, which included tests of immediate and delayed word recall, repeated subtracting by 7, counting backward from 20, naming objects, and naming the president and vice president of the U.S. As might be expected, the youngest group scored the highest, averaging 23 points, while the oldest group scored lowest, at 18.5 points.
Participants were also asked if they had ever been told by a doctor that they have diabetes. Another question was: “Have you lost all of your upper and lower natural permanent teeth?”
The condition of having no teeth is known as edentulism.
The percentages of participants who reported having both diabetes and edentulism were 6.0%, 6.7%, and 5.0% for those aged 65-74 years, 75-84 years, and 85 years or older, respectively. The proportions with neither of those conditions were 63.5%, 60.4%, and 58.3% in those three age groups, respectively (P < .001).
Compared with their counterparts with neither diabetes nor edentulism at baseline, older adults with both conditions aged 65-74 years (P < .001) and aged 75-84 years had worse cognitive function (P < .001).
In terms of the rate of cognitive decline, compared with those with neither condition from the same age cohort, older adults aged 65-74 years with both conditions declined at a higher rate (P < .001).
Having diabetes alone led to accelerated cognitive decline in older adults aged 65-74 years (P < .001). Having edentulism alone led to accelerated decline in older adults aged 65-74 years (P < .001) and older adults aged 75-84 years (P < 0.01).
“Our study finds the co-occurrence of diabetes and edentulism led to a worse cognitive function and a faster cognitive decline in older adults aged 65-74 years,” say Wu and colleagues.
Study limitations: Better data needed
The study has several limitations, most of them due to the data source. For example, while the HRS collects detailed information on cognitive status, edentulism is its only measure of oral health. There were no data on whether individuals had replacements such as dentures or implants that would affect their ability to eat, which could influence other health factors.
“I have made repeated appeals for federal funding to collect more oral health-related information in large national surveys,” Dr. Wu told this news organization.
Similarly, assessments of diabetes status such as hemoglobin A1c were only available for small subsets and not sufficient to demonstrate statistical significance, she explained.
Dr. Wu suggested that both oral health and cognitive screening might be included in the “Welcome to Medicare” preventive visit. In addition, “Oral hygiene practice should also be highlighted to improve cognitive health. Developing dental care interventions and programs are needed for reducing the societal cost of dementia.”
The study was partially supported by the National Institutes of Health. The authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
most specifically in those 65-74 years of age, new findings suggest.
The data come from a 12-year follow-up of older adults in a nationally representative U.S. survey.
“From a clinical perspective, our study demonstrates the importance of improving access to dental health care and integrating primary dental and medical care. Health care professionals and family caregivers should pay close attention to the cognitive status of diabetic older adults with poor oral health status,” lead author Bei Wu, PhD, of New York University, said in an interview. Dr. Wu is the Dean’s Professor in Global Health and codirector of the NYU Aging Incubator.
Moreover, said Dr. Wu: “For individuals with both poor oral health and diabetes, regular dental visits should be encouraged in addition to adherence to the diabetes self-care protocol.”
Diabetes has long been recognized as a risk factor for cognitive decline, but the findings have been inconsistent for different age groups. Tooth loss has also been linked to cognitive decline and dementia, as well as diabetes.
The mechanisms aren’t entirely clear, but “co-occurring diabetes and poor oral health may increase the risk for dementia, possibly via the potentially interrelated pathways of chronic inflammation and cardiovascular risk factors,” Dr. Wu said.
The new study, published in the Journal of Dental Research, is the first to examine the relationships between all three conditions by age group.
Diabetes, edentulism, and cognitive decline
The data came from a total of 9,948 participants in the Health and Retirement Study (HRS) from 2006 to 2018. At baseline, 5,440 participants were aged 65-74 years, 3,300 were aged 75-84, and 1,208 were aged 85 years or older.
They were assessed every 2 years using the 35-point Telephone Survey for Cognitive Status, which included tests of immediate and delayed word recall, repeated subtracting by 7, counting backward from 20, naming objects, and naming the president and vice president of the U.S. As might be expected, the youngest group scored the highest, averaging 23 points, while the oldest group scored lowest, at 18.5 points.
Participants were also asked if they had ever been told by a doctor that they have diabetes. Another question was: “Have you lost all of your upper and lower natural permanent teeth?”
The condition of having no teeth is known as edentulism.
The percentages of participants who reported having both diabetes and edentulism were 6.0%, 6.7%, and 5.0% for those aged 65-74 years, 75-84 years, and 85 years or older, respectively. The proportions with neither of those conditions were 63.5%, 60.4%, and 58.3% in those three age groups, respectively (P < .001).
Compared with their counterparts with neither diabetes nor edentulism at baseline, older adults with both conditions aged 65-74 years (P < .001) and aged 75-84 years had worse cognitive function (P < .001).
In terms of the rate of cognitive decline, compared with those with neither condition from the same age cohort, older adults aged 65-74 years with both conditions declined at a higher rate (P < .001).
Having diabetes alone led to accelerated cognitive decline in older adults aged 65-74 years (P < .001). Having edentulism alone led to accelerated decline in older adults aged 65-74 years (P < .001) and older adults aged 75-84 years (P < 0.01).
“Our study finds the co-occurrence of diabetes and edentulism led to a worse cognitive function and a faster cognitive decline in older adults aged 65-74 years,” say Wu and colleagues.
Study limitations: Better data needed
The study has several limitations, most of them due to the data source. For example, while the HRS collects detailed information on cognitive status, edentulism is its only measure of oral health. There were no data on whether individuals had replacements such as dentures or implants that would affect their ability to eat, which could influence other health factors.
“I have made repeated appeals for federal funding to collect more oral health-related information in large national surveys,” Dr. Wu told this news organization.
Similarly, assessments of diabetes status such as hemoglobin A1c were only available for small subsets and not sufficient to demonstrate statistical significance, she explained.
Dr. Wu suggested that both oral health and cognitive screening might be included in the “Welcome to Medicare” preventive visit. In addition, “Oral hygiene practice should also be highlighted to improve cognitive health. Developing dental care interventions and programs are needed for reducing the societal cost of dementia.”
The study was partially supported by the National Institutes of Health. The authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF DENTAL RESEARCH
Celebrity death finally solved – with locks of hair
This transcript has been edited for clarity.
I’m going to open this week with a case.
A 56-year-old musician presents with diffuse abdominal pain, cramping, and jaundice. His medical history is notable for years of diffuse abdominal complaints, characterized by disabling bouts of diarrhea.
In addition to the jaundice, this acute illness was accompanied by fever as well as diffuse edema and ascites. The patient underwent several abdominal paracenteses to drain excess fluid. One consulting physician administered alcohol to relieve pain, to little avail.
The patient succumbed to his illness. An autopsy showed diffuse liver injury, as well as papillary necrosis of the kidneys. Notably, the nerves of his auditory canal were noted to be thickened, along with the bony part of the skull, consistent with Paget disease of the bone and explaining, potentially, why the talented musician had gone deaf at such a young age.
An interesting note on social history: The patient had apparently developed some feelings for the niece of that doctor who prescribed alcohol. Her name was Therese, perhaps mistranscribed as Elise, and it seems that he may have written this song for her.
We’re talking about this paper in Current Biology, by Tristan Begg and colleagues, which gives us a look into the very genome of what some would argue is the world’s greatest composer.
The ability to extract DNA from older specimens has transformed the fields of anthropology, archaeology, and history, and now, perhaps, musicology as well.
The researchers identified eight locks of hair in private and public collections, all attributed to the maestro.
Four of the samples had an intact chain of custody from the time the hair was cut. DNA sequencing on these four and an additional one of the eight locks came from the same individual, a male of European heritage.
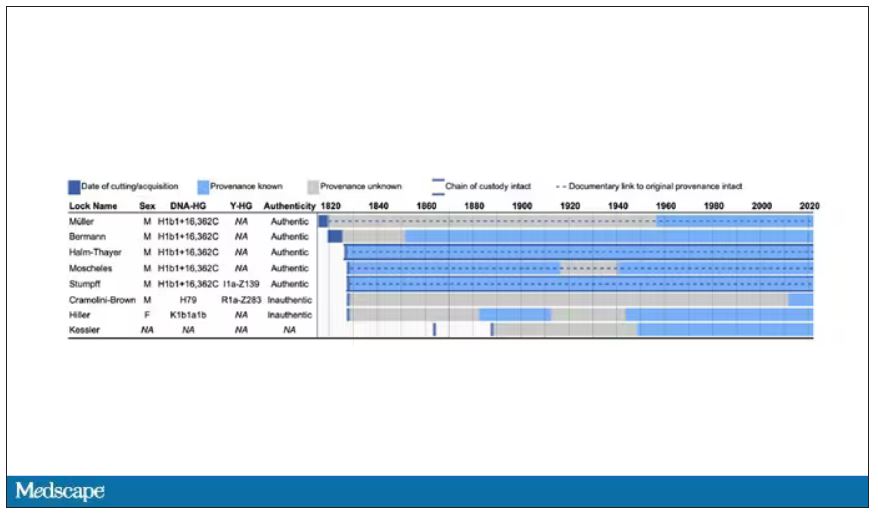
The three locks with less documentation came from three other unrelated individuals. Interestingly, analysis of one of those hair samples – the so-called Hiller Lock – had shown high levels of lead, leading historians to speculate that lead poisoning could account for some of Beethoven’s symptoms.

DNA analysis of that hair reveals it to have come from a woman likely of North African, Middle Eastern, or Jewish ancestry. We can no longer presume that plumbism was involved in Beethoven’s death. Beethoven’s ancestry turns out to be less exotic and maps quite well to ethnic German populations today.
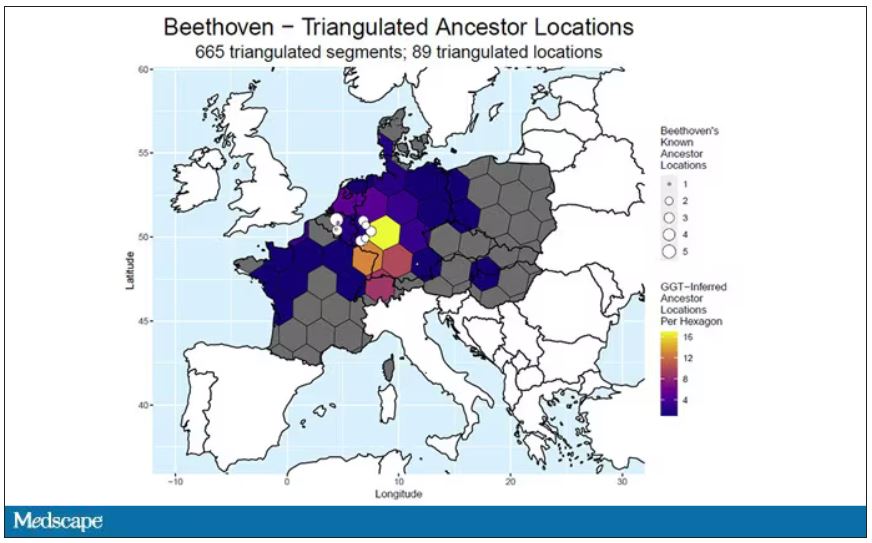
In fact, there are van Beethovens alive as we speak, primarily in Belgium. Genealogic records suggest that these van Beethovens share a common ancestor with the virtuoso composer, a man by the name of Aert van Beethoven.
But the DNA reveals a scandal.
The Y-chromosome that Beethoven inherited was not Aert van Beethoven’s. Questions of Beethoven’s paternity have been raised before, but this evidence strongly suggests an extramarital paternity event, at least in the generations preceding his birth. That’s right – Beethoven may not have been a Beethoven.
With five locks now essentially certain to have come from Beethoven himself, the authors could use DNA analysis to try to explain three significant health problems he experienced throughout his life and death: his hearing loss, his terrible gastrointestinal issues, and his liver failure.
Let’s start with the most disappointing results, explanations for his hearing loss. No genetic cause was forthcoming, though the authors note that they have little to go on in regard to the genetic risk for otosclerosis, to which his hearing loss has often been attributed. Lead poisoning is, of course, possible here, though this report focuses only on genetics – there was no testing for lead – and as I mentioned, the lock that was strongly lead-positive in prior studies is almost certainly inauthentic.
What about his lifelong GI complaints? Some have suggested celiac disease or lactose intolerance as explanations. These can essentially be ruled out by the genetic analysis, which shows no risk alleles for celiac disease and the presence of the lactase-persistence gene which confers the ability to metabolize lactose throughout one’s life. IBS is harder to assess genetically, but for what it’s worth, he scored quite low on a polygenic risk score for the condition, in just the 9th percentile of risk. We should probably be looking elsewhere to explain the GI distress.
The genetic information bore much more fruit in regard to his liver disease. Remember that Beethoven’s autopsy showed cirrhosis. His polygenic risk score for liver cirrhosis puts him in the 96th percentile of risk. He was also heterozygous for two variants that can cause hereditary hemochromatosis. The risk for cirrhosis among those with these variants is increased by the use of alcohol. And historical accounts are quite clear that Beethoven consumed more than his share.
But it wasn’t just Beethoven’s DNA in these hair follicles. Analysis of a follicle from later in his life revealed the unmistakable presence of hepatitis B virus. Endemic in Europe at the time, this was a common cause of liver failure and is likely to have contributed to, if not directly caused, Beethoven’s demise.
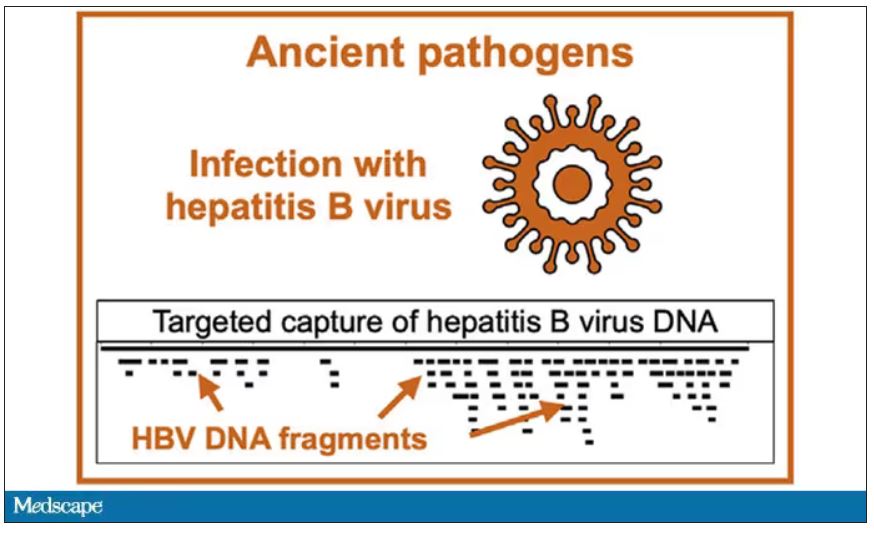
It’s hard to read these results and not marvel at the fact that, two centuries after his death, our fascination with Beethoven has led us to probe every corner of his life – his letters, his writings, his medical records, and now his very DNA. What are we actually looking for? Is it relevant to us today what caused his hearing loss? His stomach troubles? Even his death? Will it help any patients in the future? I propose that what we are actually trying to understand is something ineffable: Genius of magnitude that is rarely seen in one or many lifetimes. And our scientific tools, as sharp as they may have become, are still far too blunt to probe the depths of that transcendence.
In any case, friends, no more of these sounds. Let us sing more cheerful songs, more full of joy.
For Medscape, I’m Perry Wilson.
Dr. Wilson is associate professor, department of medicine, and director, Clinical and Translational Research Accelerator, at Yale University, New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’m going to open this week with a case.
A 56-year-old musician presents with diffuse abdominal pain, cramping, and jaundice. His medical history is notable for years of diffuse abdominal complaints, characterized by disabling bouts of diarrhea.
In addition to the jaundice, this acute illness was accompanied by fever as well as diffuse edema and ascites. The patient underwent several abdominal paracenteses to drain excess fluid. One consulting physician administered alcohol to relieve pain, to little avail.
The patient succumbed to his illness. An autopsy showed diffuse liver injury, as well as papillary necrosis of the kidneys. Notably, the nerves of his auditory canal were noted to be thickened, along with the bony part of the skull, consistent with Paget disease of the bone and explaining, potentially, why the talented musician had gone deaf at such a young age.
An interesting note on social history: The patient had apparently developed some feelings for the niece of that doctor who prescribed alcohol. Her name was Therese, perhaps mistranscribed as Elise, and it seems that he may have written this song for her.
We’re talking about this paper in Current Biology, by Tristan Begg and colleagues, which gives us a look into the very genome of what some would argue is the world’s greatest composer.
The ability to extract DNA from older specimens has transformed the fields of anthropology, archaeology, and history, and now, perhaps, musicology as well.
The researchers identified eight locks of hair in private and public collections, all attributed to the maestro.
Four of the samples had an intact chain of custody from the time the hair was cut. DNA sequencing on these four and an additional one of the eight locks came from the same individual, a male of European heritage.

The three locks with less documentation came from three other unrelated individuals. Interestingly, analysis of one of those hair samples – the so-called Hiller Lock – had shown high levels of lead, leading historians to speculate that lead poisoning could account for some of Beethoven’s symptoms.

DNA analysis of that hair reveals it to have come from a woman likely of North African, Middle Eastern, or Jewish ancestry. We can no longer presume that plumbism was involved in Beethoven’s death. Beethoven’s ancestry turns out to be less exotic and maps quite well to ethnic German populations today.

In fact, there are van Beethovens alive as we speak, primarily in Belgium. Genealogic records suggest that these van Beethovens share a common ancestor with the virtuoso composer, a man by the name of Aert van Beethoven.
But the DNA reveals a scandal.
The Y-chromosome that Beethoven inherited was not Aert van Beethoven’s. Questions of Beethoven’s paternity have been raised before, but this evidence strongly suggests an extramarital paternity event, at least in the generations preceding his birth. That’s right – Beethoven may not have been a Beethoven.
With five locks now essentially certain to have come from Beethoven himself, the authors could use DNA analysis to try to explain three significant health problems he experienced throughout his life and death: his hearing loss, his terrible gastrointestinal issues, and his liver failure.
Let’s start with the most disappointing results, explanations for his hearing loss. No genetic cause was forthcoming, though the authors note that they have little to go on in regard to the genetic risk for otosclerosis, to which his hearing loss has often been attributed. Lead poisoning is, of course, possible here, though this report focuses only on genetics – there was no testing for lead – and as I mentioned, the lock that was strongly lead-positive in prior studies is almost certainly inauthentic.
What about his lifelong GI complaints? Some have suggested celiac disease or lactose intolerance as explanations. These can essentially be ruled out by the genetic analysis, which shows no risk alleles for celiac disease and the presence of the lactase-persistence gene which confers the ability to metabolize lactose throughout one’s life. IBS is harder to assess genetically, but for what it’s worth, he scored quite low on a polygenic risk score for the condition, in just the 9th percentile of risk. We should probably be looking elsewhere to explain the GI distress.
The genetic information bore much more fruit in regard to his liver disease. Remember that Beethoven’s autopsy showed cirrhosis. His polygenic risk score for liver cirrhosis puts him in the 96th percentile of risk. He was also heterozygous for two variants that can cause hereditary hemochromatosis. The risk for cirrhosis among those with these variants is increased by the use of alcohol. And historical accounts are quite clear that Beethoven consumed more than his share.
But it wasn’t just Beethoven’s DNA in these hair follicles. Analysis of a follicle from later in his life revealed the unmistakable presence of hepatitis B virus. Endemic in Europe at the time, this was a common cause of liver failure and is likely to have contributed to, if not directly caused, Beethoven’s demise.

It’s hard to read these results and not marvel at the fact that, two centuries after his death, our fascination with Beethoven has led us to probe every corner of his life – his letters, his writings, his medical records, and now his very DNA. What are we actually looking for? Is it relevant to us today what caused his hearing loss? His stomach troubles? Even his death? Will it help any patients in the future? I propose that what we are actually trying to understand is something ineffable: Genius of magnitude that is rarely seen in one or many lifetimes. And our scientific tools, as sharp as they may have become, are still far too blunt to probe the depths of that transcendence.
In any case, friends, no more of these sounds. Let us sing more cheerful songs, more full of joy.
For Medscape, I’m Perry Wilson.
Dr. Wilson is associate professor, department of medicine, and director, Clinical and Translational Research Accelerator, at Yale University, New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’m going to open this week with a case.
A 56-year-old musician presents with diffuse abdominal pain, cramping, and jaundice. His medical history is notable for years of diffuse abdominal complaints, characterized by disabling bouts of diarrhea.
In addition to the jaundice, this acute illness was accompanied by fever as well as diffuse edema and ascites. The patient underwent several abdominal paracenteses to drain excess fluid. One consulting physician administered alcohol to relieve pain, to little avail.
The patient succumbed to his illness. An autopsy showed diffuse liver injury, as well as papillary necrosis of the kidneys. Notably, the nerves of his auditory canal were noted to be thickened, along with the bony part of the skull, consistent with Paget disease of the bone and explaining, potentially, why the talented musician had gone deaf at such a young age.
An interesting note on social history: The patient had apparently developed some feelings for the niece of that doctor who prescribed alcohol. Her name was Therese, perhaps mistranscribed as Elise, and it seems that he may have written this song for her.
We’re talking about this paper in Current Biology, by Tristan Begg and colleagues, which gives us a look into the very genome of what some would argue is the world’s greatest composer.
The ability to extract DNA from older specimens has transformed the fields of anthropology, archaeology, and history, and now, perhaps, musicology as well.
The researchers identified eight locks of hair in private and public collections, all attributed to the maestro.
Four of the samples had an intact chain of custody from the time the hair was cut. DNA sequencing on these four and an additional one of the eight locks came from the same individual, a male of European heritage.

The three locks with less documentation came from three other unrelated individuals. Interestingly, analysis of one of those hair samples – the so-called Hiller Lock – had shown high levels of lead, leading historians to speculate that lead poisoning could account for some of Beethoven’s symptoms.

DNA analysis of that hair reveals it to have come from a woman likely of North African, Middle Eastern, or Jewish ancestry. We can no longer presume that plumbism was involved in Beethoven’s death. Beethoven’s ancestry turns out to be less exotic and maps quite well to ethnic German populations today.

In fact, there are van Beethovens alive as we speak, primarily in Belgium. Genealogic records suggest that these van Beethovens share a common ancestor with the virtuoso composer, a man by the name of Aert van Beethoven.
But the DNA reveals a scandal.
The Y-chromosome that Beethoven inherited was not Aert van Beethoven’s. Questions of Beethoven’s paternity have been raised before, but this evidence strongly suggests an extramarital paternity event, at least in the generations preceding his birth. That’s right – Beethoven may not have been a Beethoven.
With five locks now essentially certain to have come from Beethoven himself, the authors could use DNA analysis to try to explain three significant health problems he experienced throughout his life and death: his hearing loss, his terrible gastrointestinal issues, and his liver failure.
Let’s start with the most disappointing results, explanations for his hearing loss. No genetic cause was forthcoming, though the authors note that they have little to go on in regard to the genetic risk for otosclerosis, to which his hearing loss has often been attributed. Lead poisoning is, of course, possible here, though this report focuses only on genetics – there was no testing for lead – and as I mentioned, the lock that was strongly lead-positive in prior studies is almost certainly inauthentic.
What about his lifelong GI complaints? Some have suggested celiac disease or lactose intolerance as explanations. These can essentially be ruled out by the genetic analysis, which shows no risk alleles for celiac disease and the presence of the lactase-persistence gene which confers the ability to metabolize lactose throughout one’s life. IBS is harder to assess genetically, but for what it’s worth, he scored quite low on a polygenic risk score for the condition, in just the 9th percentile of risk. We should probably be looking elsewhere to explain the GI distress.
The genetic information bore much more fruit in regard to his liver disease. Remember that Beethoven’s autopsy showed cirrhosis. His polygenic risk score for liver cirrhosis puts him in the 96th percentile of risk. He was also heterozygous for two variants that can cause hereditary hemochromatosis. The risk for cirrhosis among those with these variants is increased by the use of alcohol. And historical accounts are quite clear that Beethoven consumed more than his share.
But it wasn’t just Beethoven’s DNA in these hair follicles. Analysis of a follicle from later in his life revealed the unmistakable presence of hepatitis B virus. Endemic in Europe at the time, this was a common cause of liver failure and is likely to have contributed to, if not directly caused, Beethoven’s demise.

It’s hard to read these results and not marvel at the fact that, two centuries after his death, our fascination with Beethoven has led us to probe every corner of his life – his letters, his writings, his medical records, and now his very DNA. What are we actually looking for? Is it relevant to us today what caused his hearing loss? His stomach troubles? Even his death? Will it help any patients in the future? I propose that what we are actually trying to understand is something ineffable: Genius of magnitude that is rarely seen in one or many lifetimes. And our scientific tools, as sharp as they may have become, are still far too blunt to probe the depths of that transcendence.
In any case, friends, no more of these sounds. Let us sing more cheerful songs, more full of joy.
For Medscape, I’m Perry Wilson.
Dr. Wilson is associate professor, department of medicine, and director, Clinical and Translational Research Accelerator, at Yale University, New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Nurse makes millions selling her licensing exam study sheets
Ms. Beggs, 28, sells one-page study sheets or bundles of sheets, sometimes with colorful drawings, conversation bubbles and underlining, that boil down concepts for particular conditions into easy-to-understand language.
The biggest seller on Ms. Beggs’ online marketplace Etsy site, RNExplained, is a bundle of study guides covering eight core nursing classes. The notes range in price from $2 to $150. More than 70,000 customers have bought the $60 bundle, according to the website.
Ms. Beggs’ business developed in a “very unintentional” way when COVID hit with just months left in her nursing program at Mount Saint Mary’s University, Los Angeles, she told this news organization.
Classes had switched to Zoom, and she had no one to study with as she prepared to take her board exams.
“The best way I know how to study is to teach things out loud. But because I had nobody to teach out loud to, I would literally teach them to the wall,” Ms. Beggs said. “I would record myself so I could play it back and teach myself these topics that were hard for me to understand.”
Just for fun, she says, she posted them on TikTok and the responses started flowing in, with followers asking where she was selling the sheets. She now has more than 660,000 TikTok followers and 9 million likes.
Ms. Beggs said that every sheet highlights a condition, and she has made 308 of them.
Traditional classroom lessons typically teach one medical condition in 5-6 pages, Ms. Beggs said. “I go straight to the point.”
One reviewer on Ms. Beggs’ Etsy site appreciated the handwritten notes, calling them “simplified and concise.” Another commented: “Definitely helped me pass my last exam.”
Ms. Beggs says that her notes may seem simple, but each page represents comprehensive research.
“I have to go through not just one source of information to make sure my information is factual,” Ms. Beggs says. “What you teach in California might be a little different than what you teach in Florida. It’s very meticulous. The lab values will be a little different everywhere you go.”
She acknowledges her competition, noting that there are many other study guides for the NCLEX and nursing courses.
Nursing groups weigh in
Dawn Kappel, spokesperson for the National Council of State Boards of Nursing, which oversees NCLEX, said in an interview that “NCSBN has no issue with the current content of Stephanee Beggs’ business venture.”
For many students, the study guides will be helpful, especially for visual learners, said Carole Kenner, PhD, RN, dean and professor in the School of Nursing and Health Sciences at The College of New Jersey.
But for students “who are less confident in their knowledge, I would want to see a lot more in-depth explanation and rationale,” Dr. Kenner said.
“Since the NCLEX is moving to more cased-based scenarios, the next-gen unfolding cases, you really have to understand a lot of the rationale.”
The notes remind Dr. Kenner of traditional flash cards. “I don’t think it will work for all students, but even the fanciest of onsite review courses are useful to everyone,” she said.
‘Not cutting corners’
As an emergency nurse, Ms. Beggs said, “I have the experience as a nurse to show people that what you are learning will be seen in real life.”
“The way I teach my brand is not to take shortcuts. I love to teach to understand rather than teaching to memorize for an exam.”
She said she sees her guides as a supplement to learning, not a replacement.
“It’s not cutting corners,” she says. “I condense a medical condition that could take a very long time to understand and break it into layman’s terms.”
Ms. Beggs said when people hear about the $2 million, they often ask her whether she plans to give up her shifts in the emergency department for the more lucrative venture.
The answer is no, at least not yet.
“Aside from teaching, I genuinely love being at the bedside,” Ms. Beggs said. “I don’t foresee myself leaving that for good for as long as I can handle both.” She acknowledged, though, that her business now takes up most of her time.
“I love everything about both aspects, so it’s hard for me to choose.”
A version of this article first appeared on Medscape.com.
Ms. Beggs, 28, sells one-page study sheets or bundles of sheets, sometimes with colorful drawings, conversation bubbles and underlining, that boil down concepts for particular conditions into easy-to-understand language.
The biggest seller on Ms. Beggs’ online marketplace Etsy site, RNExplained, is a bundle of study guides covering eight core nursing classes. The notes range in price from $2 to $150. More than 70,000 customers have bought the $60 bundle, according to the website.
Ms. Beggs’ business developed in a “very unintentional” way when COVID hit with just months left in her nursing program at Mount Saint Mary’s University, Los Angeles, she told this news organization.
Classes had switched to Zoom, and she had no one to study with as she prepared to take her board exams.
“The best way I know how to study is to teach things out loud. But because I had nobody to teach out loud to, I would literally teach them to the wall,” Ms. Beggs said. “I would record myself so I could play it back and teach myself these topics that were hard for me to understand.”
Just for fun, she says, she posted them on TikTok and the responses started flowing in, with followers asking where she was selling the sheets. She now has more than 660,000 TikTok followers and 9 million likes.
Ms. Beggs said that every sheet highlights a condition, and she has made 308 of them.
Traditional classroom lessons typically teach one medical condition in 5-6 pages, Ms. Beggs said. “I go straight to the point.”
One reviewer on Ms. Beggs’ Etsy site appreciated the handwritten notes, calling them “simplified and concise.” Another commented: “Definitely helped me pass my last exam.”
Ms. Beggs says that her notes may seem simple, but each page represents comprehensive research.
“I have to go through not just one source of information to make sure my information is factual,” Ms. Beggs says. “What you teach in California might be a little different than what you teach in Florida. It’s very meticulous. The lab values will be a little different everywhere you go.”
She acknowledges her competition, noting that there are many other study guides for the NCLEX and nursing courses.
Nursing groups weigh in
Dawn Kappel, spokesperson for the National Council of State Boards of Nursing, which oversees NCLEX, said in an interview that “NCSBN has no issue with the current content of Stephanee Beggs’ business venture.”
For many students, the study guides will be helpful, especially for visual learners, said Carole Kenner, PhD, RN, dean and professor in the School of Nursing and Health Sciences at The College of New Jersey.
But for students “who are less confident in their knowledge, I would want to see a lot more in-depth explanation and rationale,” Dr. Kenner said.
“Since the NCLEX is moving to more cased-based scenarios, the next-gen unfolding cases, you really have to understand a lot of the rationale.”
The notes remind Dr. Kenner of traditional flash cards. “I don’t think it will work for all students, but even the fanciest of onsite review courses are useful to everyone,” she said.
‘Not cutting corners’
As an emergency nurse, Ms. Beggs said, “I have the experience as a nurse to show people that what you are learning will be seen in real life.”
“The way I teach my brand is not to take shortcuts. I love to teach to understand rather than teaching to memorize for an exam.”
She said she sees her guides as a supplement to learning, not a replacement.
“It’s not cutting corners,” she says. “I condense a medical condition that could take a very long time to understand and break it into layman’s terms.”
Ms. Beggs said when people hear about the $2 million, they often ask her whether she plans to give up her shifts in the emergency department for the more lucrative venture.
The answer is no, at least not yet.
“Aside from teaching, I genuinely love being at the bedside,” Ms. Beggs said. “I don’t foresee myself leaving that for good for as long as I can handle both.” She acknowledged, though, that her business now takes up most of her time.
“I love everything about both aspects, so it’s hard for me to choose.”
A version of this article first appeared on Medscape.com.
Ms. Beggs, 28, sells one-page study sheets or bundles of sheets, sometimes with colorful drawings, conversation bubbles and underlining, that boil down concepts for particular conditions into easy-to-understand language.
The biggest seller on Ms. Beggs’ online marketplace Etsy site, RNExplained, is a bundle of study guides covering eight core nursing classes. The notes range in price from $2 to $150. More than 70,000 customers have bought the $60 bundle, according to the website.
Ms. Beggs’ business developed in a “very unintentional” way when COVID hit with just months left in her nursing program at Mount Saint Mary’s University, Los Angeles, she told this news organization.
Classes had switched to Zoom, and she had no one to study with as she prepared to take her board exams.
“The best way I know how to study is to teach things out loud. But because I had nobody to teach out loud to, I would literally teach them to the wall,” Ms. Beggs said. “I would record myself so I could play it back and teach myself these topics that were hard for me to understand.”
Just for fun, she says, she posted them on TikTok and the responses started flowing in, with followers asking where she was selling the sheets. She now has more than 660,000 TikTok followers and 9 million likes.
Ms. Beggs said that every sheet highlights a condition, and she has made 308 of them.
Traditional classroom lessons typically teach one medical condition in 5-6 pages, Ms. Beggs said. “I go straight to the point.”
One reviewer on Ms. Beggs’ Etsy site appreciated the handwritten notes, calling them “simplified and concise.” Another commented: “Definitely helped me pass my last exam.”
Ms. Beggs says that her notes may seem simple, but each page represents comprehensive research.
“I have to go through not just one source of information to make sure my information is factual,” Ms. Beggs says. “What you teach in California might be a little different than what you teach in Florida. It’s very meticulous. The lab values will be a little different everywhere you go.”
She acknowledges her competition, noting that there are many other study guides for the NCLEX and nursing courses.
Nursing groups weigh in
Dawn Kappel, spokesperson for the National Council of State Boards of Nursing, which oversees NCLEX, said in an interview that “NCSBN has no issue with the current content of Stephanee Beggs’ business venture.”
For many students, the study guides will be helpful, especially for visual learners, said Carole Kenner, PhD, RN, dean and professor in the School of Nursing and Health Sciences at The College of New Jersey.
But for students “who are less confident in their knowledge, I would want to see a lot more in-depth explanation and rationale,” Dr. Kenner said.
“Since the NCLEX is moving to more cased-based scenarios, the next-gen unfolding cases, you really have to understand a lot of the rationale.”
The notes remind Dr. Kenner of traditional flash cards. “I don’t think it will work for all students, but even the fanciest of onsite review courses are useful to everyone,” she said.
‘Not cutting corners’
As an emergency nurse, Ms. Beggs said, “I have the experience as a nurse to show people that what you are learning will be seen in real life.”
“The way I teach my brand is not to take shortcuts. I love to teach to understand rather than teaching to memorize for an exam.”
She said she sees her guides as a supplement to learning, not a replacement.
“It’s not cutting corners,” she says. “I condense a medical condition that could take a very long time to understand and break it into layman’s terms.”
Ms. Beggs said when people hear about the $2 million, they often ask her whether she plans to give up her shifts in the emergency department for the more lucrative venture.
The answer is no, at least not yet.
“Aside from teaching, I genuinely love being at the bedside,” Ms. Beggs said. “I don’t foresee myself leaving that for good for as long as I can handle both.” She acknowledged, though, that her business now takes up most of her time.
“I love everything about both aspects, so it’s hard for me to choose.”
A version of this article first appeared on Medscape.com.








