User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
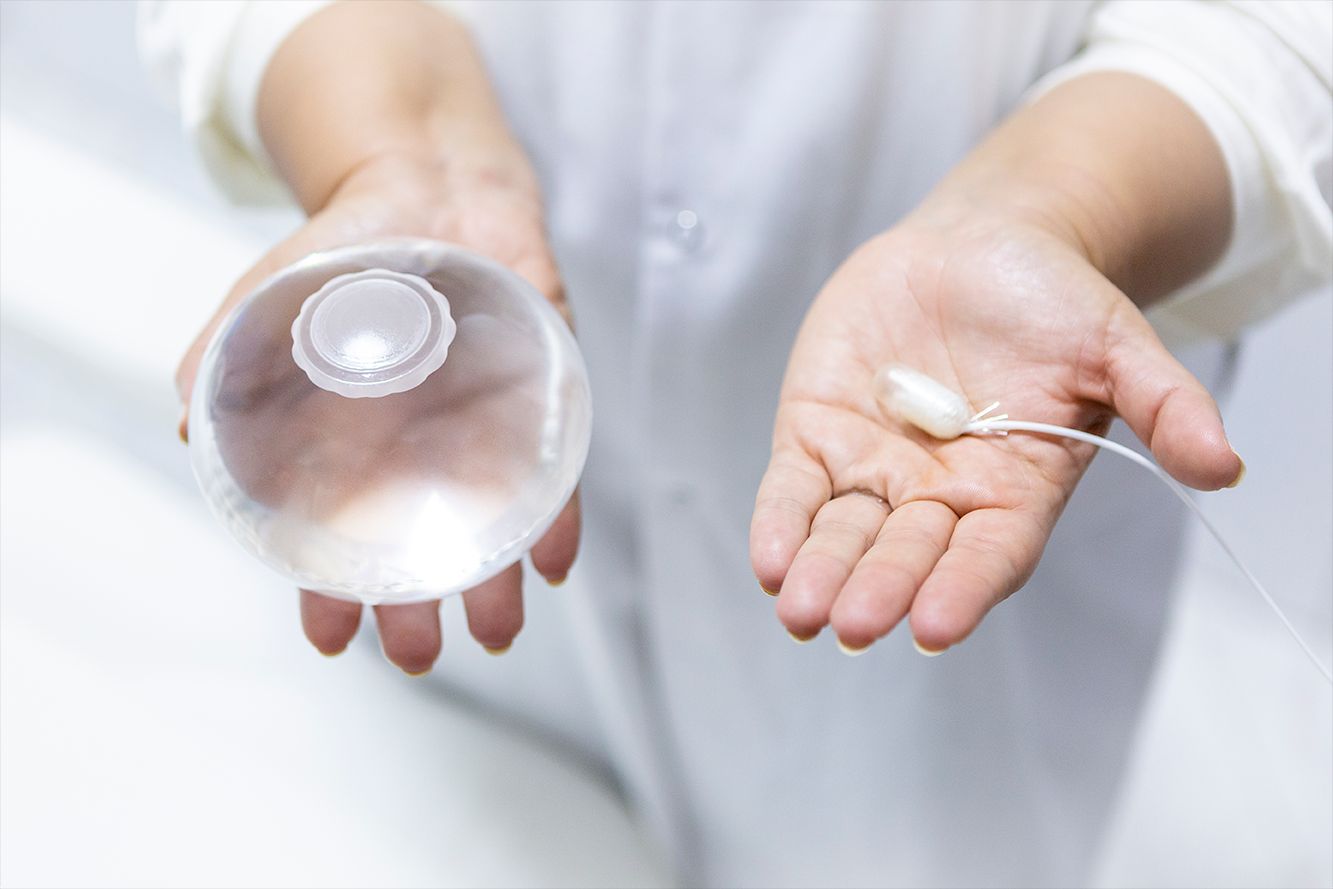
Obesity drug with swallowable balloon boosts weight loss
DUBLIN – A swallowable gastric balloon (Allurion Balloon, formerly known as Elipse) combined with daily subcutaneous injections of the glucagonlike peptide 1 (GLP-1) agonist liraglutide (Saxenda, Novo Nordisk), leads to a significant average total body weight loss of 19% (18 kg or 40 lb) after around 4 months in people with obesity.
said Roberta Ienca, MD, from the Clinica Nuova Villa Claudia, Rome, who presented the findings at this year’s European Congress on Obesity.
“Despite both the balloon and liraglutide working on the early satiety feeling, the introduction of liraglutide around 1 month after [swallowing the balloon] or more frequently after 3-4 months, could sustain these feelings for a longer period of time,” she said in an interview.
“The addition of the GLP-1 agonist therapy (liraglutide) to patients treated with the Allurion program [gastric balloon] is feasible, safe, and effective in those who need additional weight loss,” she emphasized.
The balloon stayed inside participants’ stomachs for an average of 16 weeks and liraglutide was continued for an average of 4 months, resulting in a mean reduction in body mass index (BMI) of 6.4 kg/m2.
The Allurion is the world’s first and only swallowable gastric balloon placed without surgery, endoscopy, or anesthesia, and is excreted naturally after around 16 weeks.
The Allurion program delivered “excellent weight loss in individuals with overweight and obesity without going under the knife, and liraglutide has the potential to further safely enhance weight loss in cases of suboptimal adherence with the program,” Dr. Ienca said. “These two treatment approaches appear to have complementary mechanisms of action in a geographically and demographically diverse population.”
Adelardo Caballero, MD, director of the Institute of Obesity, Madrid, said that he had over 6 years of experience with the Allurion balloon in around 2,500 cases. “Over the last 3 years, we have been using Allurion balloons in combination with GLP-1 agonists. In Europe, use of the swallowable gastric balloon is common, the results are good, and it is a safe tool.”
“Using liraglutide daily in subcutaneous form is authorized in Europe and is useful in overweight and mild obesity, while use in the combination [with the balloon] is also very popular,” he explained. “In the future, the combined use of semaglutide once-weekly GLP-1 agonist or the use of dual GLP-1/gastric inhibitory polypeptide agonists [such as tirzepatide] with the swallowable intragastric balloon Allurion program or endoscopic sleeve gastroplasty will improve results,” he added.
Average 40-lb weight loss with balloon and liraglutide
For the current study, data from three international multidisciplinary obesity centers (in Italy, Spain, and Egypt) were retrospectively analyzed. All 181 patients received the combination of the Allurion balloon and liraglutide, with the latter added 4-16 weeks after swallowing the balloon.
During a 20-minute outpatient visit, participants swallowed the balloon, which was filled with liquid after reaching the stomach, and placement was confirmed by x-ray. The balloon remained inserted for around 15-17 weeks (mean 16 weeks) before natural excretion. All patients received liraglutide once daily for 1-6 months (mean 4 months). After excreting the balloon, patients started the Mediterranean diet for weight maintenance and were followed for at least 6 months.
Patients were monitored for weight loss, percentage total body weight loss, percentage excess weight loss, and BMI reduction. The timing of combining drug therapy with the Allurion program, metabolic results, and adverse event data were collected. However, Dr. Ienca explained that “the study was preliminary and aimed to evaluate feasibility and results of a combined treatment, so we didn’t collect long-term data.”
Liraglutide was mostly added in cases of unsatisfactory weight loss to boost weight reduction in patients with high BMIs, to sustain weight maintenance, and to aid diabetes control in patients with satisfactory weight loss. There were no criteria for time of onset of drug therapy in terms of a time point or percentage weight loss.
Before treatment, mean weight was 94.8 ± 21 kg and mean BMI was 33.7 ± 6.2 kg/m2. After 4 months of balloon treatment, weight loss, percentage total body weight loss, percentage excess weight loss, and decrease in BMI were 13.1 ± 7 kg, 13.9% ± 7.7%, 74.3% ± 57.1%, and 4.5 ±1.4 kg/m2 respectively.
After a mean duration of 4 months of liraglutide treatment (in addition to the gastric balloon), participants lost on average 18.1 ± 12.1 kg overall and 18.7% ± 12% of their initial total body weight. They shed 99.4% ± 84.9% of excess weight and reduced BMI by 6.4 ± 5.9 kg/m2.
Dr. Ienca explained that the study did not explore the separate contributions of the balloon or drug therapy to weight loss. “However, existing literature shows that the Allurion program leads to a weight loss of approximately 14% of total body weight after 4 months, while liraglutide studies report 12% of total body weight loss at 1 year,” he noted.
When describing the mechanism of action, Dr. Ienca said the Allurion balloon induces satiety and delays gastric emptying but the feeling of satiety starts to decrease after the first month. “For a few patients, this feeling of satiety decreases more rapidly or they have more difficulty putting in place new alimentary habits. In these patients, the addition of liraglutide gives an additional boost to support this behavioral change.”
Liraglutide-related adverse events included nausea (16.5%), diarrhea (3.3%), constipation (2.2%), and headache (1.7%), as well as drug discontinuation due to tachycardia/chest pain (1.1%) and gastrointestinal symptoms (1.1%).
Balloon removal because of intolerance occurred in 1.1% of patients, gastric dilation in 0.5%, and early balloon deflation in 0.5%. Other expected balloon-related adverse events included nausea, vomiting, and abdominal cramps.
The researchers note that the Allurion program offers a more acceptable option to balloon placement by endoscopy.
“The ease of use, low rate of adverse events, and potentially lower cost of the Allurion Program could enable much wider application of this critical intervention, and ultimately, help the millions who struggle with obesity and its associated health complications.”
A version of this article originally appeared on Medscape.com.
DUBLIN – A swallowable gastric balloon (Allurion Balloon, formerly known as Elipse) combined with daily subcutaneous injections of the glucagonlike peptide 1 (GLP-1) agonist liraglutide (Saxenda, Novo Nordisk), leads to a significant average total body weight loss of 19% (18 kg or 40 lb) after around 4 months in people with obesity.
said Roberta Ienca, MD, from the Clinica Nuova Villa Claudia, Rome, who presented the findings at this year’s European Congress on Obesity.
“Despite both the balloon and liraglutide working on the early satiety feeling, the introduction of liraglutide around 1 month after [swallowing the balloon] or more frequently after 3-4 months, could sustain these feelings for a longer period of time,” she said in an interview.
“The addition of the GLP-1 agonist therapy (liraglutide) to patients treated with the Allurion program [gastric balloon] is feasible, safe, and effective in those who need additional weight loss,” she emphasized.
The balloon stayed inside participants’ stomachs for an average of 16 weeks and liraglutide was continued for an average of 4 months, resulting in a mean reduction in body mass index (BMI) of 6.4 kg/m2.
The Allurion is the world’s first and only swallowable gastric balloon placed without surgery, endoscopy, or anesthesia, and is excreted naturally after around 16 weeks.
The Allurion program delivered “excellent weight loss in individuals with overweight and obesity without going under the knife, and liraglutide has the potential to further safely enhance weight loss in cases of suboptimal adherence with the program,” Dr. Ienca said. “These two treatment approaches appear to have complementary mechanisms of action in a geographically and demographically diverse population.”
Adelardo Caballero, MD, director of the Institute of Obesity, Madrid, said that he had over 6 years of experience with the Allurion balloon in around 2,500 cases. “Over the last 3 years, we have been using Allurion balloons in combination with GLP-1 agonists. In Europe, use of the swallowable gastric balloon is common, the results are good, and it is a safe tool.”
“Using liraglutide daily in subcutaneous form is authorized in Europe and is useful in overweight and mild obesity, while use in the combination [with the balloon] is also very popular,” he explained. “In the future, the combined use of semaglutide once-weekly GLP-1 agonist or the use of dual GLP-1/gastric inhibitory polypeptide agonists [such as tirzepatide] with the swallowable intragastric balloon Allurion program or endoscopic sleeve gastroplasty will improve results,” he added.
Average 40-lb weight loss with balloon and liraglutide
For the current study, data from three international multidisciplinary obesity centers (in Italy, Spain, and Egypt) were retrospectively analyzed. All 181 patients received the combination of the Allurion balloon and liraglutide, with the latter added 4-16 weeks after swallowing the balloon.
During a 20-minute outpatient visit, participants swallowed the balloon, which was filled with liquid after reaching the stomach, and placement was confirmed by x-ray. The balloon remained inserted for around 15-17 weeks (mean 16 weeks) before natural excretion. All patients received liraglutide once daily for 1-6 months (mean 4 months). After excreting the balloon, patients started the Mediterranean diet for weight maintenance and were followed for at least 6 months.
Patients were monitored for weight loss, percentage total body weight loss, percentage excess weight loss, and BMI reduction. The timing of combining drug therapy with the Allurion program, metabolic results, and adverse event data were collected. However, Dr. Ienca explained that “the study was preliminary and aimed to evaluate feasibility and results of a combined treatment, so we didn’t collect long-term data.”
Liraglutide was mostly added in cases of unsatisfactory weight loss to boost weight reduction in patients with high BMIs, to sustain weight maintenance, and to aid diabetes control in patients with satisfactory weight loss. There were no criteria for time of onset of drug therapy in terms of a time point or percentage weight loss.
Before treatment, mean weight was 94.8 ± 21 kg and mean BMI was 33.7 ± 6.2 kg/m2. After 4 months of balloon treatment, weight loss, percentage total body weight loss, percentage excess weight loss, and decrease in BMI were 13.1 ± 7 kg, 13.9% ± 7.7%, 74.3% ± 57.1%, and 4.5 ±1.4 kg/m2 respectively.
After a mean duration of 4 months of liraglutide treatment (in addition to the gastric balloon), participants lost on average 18.1 ± 12.1 kg overall and 18.7% ± 12% of their initial total body weight. They shed 99.4% ± 84.9% of excess weight and reduced BMI by 6.4 ± 5.9 kg/m2.
Dr. Ienca explained that the study did not explore the separate contributions of the balloon or drug therapy to weight loss. “However, existing literature shows that the Allurion program leads to a weight loss of approximately 14% of total body weight after 4 months, while liraglutide studies report 12% of total body weight loss at 1 year,” he noted.
When describing the mechanism of action, Dr. Ienca said the Allurion balloon induces satiety and delays gastric emptying but the feeling of satiety starts to decrease after the first month. “For a few patients, this feeling of satiety decreases more rapidly or they have more difficulty putting in place new alimentary habits. In these patients, the addition of liraglutide gives an additional boost to support this behavioral change.”
Liraglutide-related adverse events included nausea (16.5%), diarrhea (3.3%), constipation (2.2%), and headache (1.7%), as well as drug discontinuation due to tachycardia/chest pain (1.1%) and gastrointestinal symptoms (1.1%).
Balloon removal because of intolerance occurred in 1.1% of patients, gastric dilation in 0.5%, and early balloon deflation in 0.5%. Other expected balloon-related adverse events included nausea, vomiting, and abdominal cramps.
The researchers note that the Allurion program offers a more acceptable option to balloon placement by endoscopy.
“The ease of use, low rate of adverse events, and potentially lower cost of the Allurion Program could enable much wider application of this critical intervention, and ultimately, help the millions who struggle with obesity and its associated health complications.”
A version of this article originally appeared on Medscape.com.
DUBLIN – A swallowable gastric balloon (Allurion Balloon, formerly known as Elipse) combined with daily subcutaneous injections of the glucagonlike peptide 1 (GLP-1) agonist liraglutide (Saxenda, Novo Nordisk), leads to a significant average total body weight loss of 19% (18 kg or 40 lb) after around 4 months in people with obesity.
said Roberta Ienca, MD, from the Clinica Nuova Villa Claudia, Rome, who presented the findings at this year’s European Congress on Obesity.
“Despite both the balloon and liraglutide working on the early satiety feeling, the introduction of liraglutide around 1 month after [swallowing the balloon] or more frequently after 3-4 months, could sustain these feelings for a longer period of time,” she said in an interview.
“The addition of the GLP-1 agonist therapy (liraglutide) to patients treated with the Allurion program [gastric balloon] is feasible, safe, and effective in those who need additional weight loss,” she emphasized.
The balloon stayed inside participants’ stomachs for an average of 16 weeks and liraglutide was continued for an average of 4 months, resulting in a mean reduction in body mass index (BMI) of 6.4 kg/m2.
The Allurion is the world’s first and only swallowable gastric balloon placed without surgery, endoscopy, or anesthesia, and is excreted naturally after around 16 weeks.
The Allurion program delivered “excellent weight loss in individuals with overweight and obesity without going under the knife, and liraglutide has the potential to further safely enhance weight loss in cases of suboptimal adherence with the program,” Dr. Ienca said. “These two treatment approaches appear to have complementary mechanisms of action in a geographically and demographically diverse population.”
Adelardo Caballero, MD, director of the Institute of Obesity, Madrid, said that he had over 6 years of experience with the Allurion balloon in around 2,500 cases. “Over the last 3 years, we have been using Allurion balloons in combination with GLP-1 agonists. In Europe, use of the swallowable gastric balloon is common, the results are good, and it is a safe tool.”
“Using liraglutide daily in subcutaneous form is authorized in Europe and is useful in overweight and mild obesity, while use in the combination [with the balloon] is also very popular,” he explained. “In the future, the combined use of semaglutide once-weekly GLP-1 agonist or the use of dual GLP-1/gastric inhibitory polypeptide agonists [such as tirzepatide] with the swallowable intragastric balloon Allurion program or endoscopic sleeve gastroplasty will improve results,” he added.
Average 40-lb weight loss with balloon and liraglutide
For the current study, data from three international multidisciplinary obesity centers (in Italy, Spain, and Egypt) were retrospectively analyzed. All 181 patients received the combination of the Allurion balloon and liraglutide, with the latter added 4-16 weeks after swallowing the balloon.
During a 20-minute outpatient visit, participants swallowed the balloon, which was filled with liquid after reaching the stomach, and placement was confirmed by x-ray. The balloon remained inserted for around 15-17 weeks (mean 16 weeks) before natural excretion. All patients received liraglutide once daily for 1-6 months (mean 4 months). After excreting the balloon, patients started the Mediterranean diet for weight maintenance and were followed for at least 6 months.
Patients were monitored for weight loss, percentage total body weight loss, percentage excess weight loss, and BMI reduction. The timing of combining drug therapy with the Allurion program, metabolic results, and adverse event data were collected. However, Dr. Ienca explained that “the study was preliminary and aimed to evaluate feasibility and results of a combined treatment, so we didn’t collect long-term data.”
Liraglutide was mostly added in cases of unsatisfactory weight loss to boost weight reduction in patients with high BMIs, to sustain weight maintenance, and to aid diabetes control in patients with satisfactory weight loss. There were no criteria for time of onset of drug therapy in terms of a time point or percentage weight loss.
Before treatment, mean weight was 94.8 ± 21 kg and mean BMI was 33.7 ± 6.2 kg/m2. After 4 months of balloon treatment, weight loss, percentage total body weight loss, percentage excess weight loss, and decrease in BMI were 13.1 ± 7 kg, 13.9% ± 7.7%, 74.3% ± 57.1%, and 4.5 ±1.4 kg/m2 respectively.
After a mean duration of 4 months of liraglutide treatment (in addition to the gastric balloon), participants lost on average 18.1 ± 12.1 kg overall and 18.7% ± 12% of their initial total body weight. They shed 99.4% ± 84.9% of excess weight and reduced BMI by 6.4 ± 5.9 kg/m2.
Dr. Ienca explained that the study did not explore the separate contributions of the balloon or drug therapy to weight loss. “However, existing literature shows that the Allurion program leads to a weight loss of approximately 14% of total body weight after 4 months, while liraglutide studies report 12% of total body weight loss at 1 year,” he noted.
When describing the mechanism of action, Dr. Ienca said the Allurion balloon induces satiety and delays gastric emptying but the feeling of satiety starts to decrease after the first month. “For a few patients, this feeling of satiety decreases more rapidly or they have more difficulty putting in place new alimentary habits. In these patients, the addition of liraglutide gives an additional boost to support this behavioral change.”
Liraglutide-related adverse events included nausea (16.5%), diarrhea (3.3%), constipation (2.2%), and headache (1.7%), as well as drug discontinuation due to tachycardia/chest pain (1.1%) and gastrointestinal symptoms (1.1%).
Balloon removal because of intolerance occurred in 1.1% of patients, gastric dilation in 0.5%, and early balloon deflation in 0.5%. Other expected balloon-related adverse events included nausea, vomiting, and abdominal cramps.
The researchers note that the Allurion program offers a more acceptable option to balloon placement by endoscopy.
“The ease of use, low rate of adverse events, and potentially lower cost of the Allurion Program could enable much wider application of this critical intervention, and ultimately, help the millions who struggle with obesity and its associated health complications.”
A version of this article originally appeared on Medscape.com.
AT ECO 2023
Morning PT
Tuesdays and Fridays are tough. Not so much because of clinic, but rather because of the 32 minutes before clinic that I’m on the Peloton bike. They are the mornings I dedicate to training VO2max.
Training VO2max, or maximal oxygen consumption, is simple. Spin for a leisurely, easy-breathing, 4 minutes, then for 4 minutes push yourself until you see the light of heaven and wish for death to come. Then relax for 4 minutes again. Repeat this cycle four to six times. Done justly, you will dread Tuesdays and Fridays too. The punishing cycle of a 4-minute push, then 4-minute recovery is, however, an excellent way to improve cardiovascular fitness. And no, I’m not training for the Boston Marathon, so why am I working so hard? Because I’m training for marathon clinic days for the next 20 years.
Now more than ever, I feel we have to be physically fit to deal with a physicians’ day’s work. It’s exhausting. The root cause is too much work, yes, but I believe being physically fit could help.
I was talking to an 86-year-old patient about this very topic recently. He was short, with a well-manicured goatee and shiny head. He stuck his arm out to shake my hand. “Glad we’re back to handshakes again, doc.” His grip was that of a 30-year-old. “Buff” you’d likely describe him: He is noticeably muscular, not a skinny old man. He’s an old Navy Master Chief who started a business in wholesale flowers, which distributes all over the United States. And he’s still working full time. Impressed, I asked his secret for such vigor. PT, he replied.
PT, or physical training, is a foundational element of the Navy. Every sailor starts his or her day with morning PT before carrying out their duties. Some 30 years later, this guy is still getting after it. He does push-ups, sit-ups, and pull-ups nearly every morning. Morning PT is what he attributes to his success not only in health, but also business. As he sees it, he has the business savvy and experience of an old guy and the energy and stamina of a college kid. A good combination for a successful life.
I’ve always been pretty fit. Lately, I’ve been trying to take it to the next level, to not just be “physically active,” but rather “high-performance fit.” There are plenty of sources for instruction; how to stay young and healthy isn’t a new idea after all. I mean, Herodotus wrote of finding the Fountain of Youth in the 5th century BCE. A couple thousand years later, it’s still on trend. One of my favorite sages giving health span advice is Peter Attia, MD. I’ve been a fan since I met him at TEDMED in 2013 and I marvel at the astounding body of work he has created since. A Johns Hopkins–trained surgeon, he has spent his career reviewing the scientific literature about longevity and sharing it as actionable content. His book, “Outlive: The Science and Art of Longevity” (New York: Penguin Random House, 2023) is a nice summary of his work. I recommend it.
Right now I’m switching between type 2 muscle fiber work (lots of jumping like my 2-year-old) and cardiovascular training including the aforementioned VO2max work. I cannot say that my patient inbox is any cleaner, or that I’m faster in the office, but I’m not flagging by the end of the day anymore. Master Chief challenged me to match his 10 pull-ups before he returns for his follow up visit. I’ll gladly give up Peloton sprints to work on that.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at dermnews@mdedge.com.
Tuesdays and Fridays are tough. Not so much because of clinic, but rather because of the 32 minutes before clinic that I’m on the Peloton bike. They are the mornings I dedicate to training VO2max.
Training VO2max, or maximal oxygen consumption, is simple. Spin for a leisurely, easy-breathing, 4 minutes, then for 4 minutes push yourself until you see the light of heaven and wish for death to come. Then relax for 4 minutes again. Repeat this cycle four to six times. Done justly, you will dread Tuesdays and Fridays too. The punishing cycle of a 4-minute push, then 4-minute recovery is, however, an excellent way to improve cardiovascular fitness. And no, I’m not training for the Boston Marathon, so why am I working so hard? Because I’m training for marathon clinic days for the next 20 years.
Now more than ever, I feel we have to be physically fit to deal with a physicians’ day’s work. It’s exhausting. The root cause is too much work, yes, but I believe being physically fit could help.
I was talking to an 86-year-old patient about this very topic recently. He was short, with a well-manicured goatee and shiny head. He stuck his arm out to shake my hand. “Glad we’re back to handshakes again, doc.” His grip was that of a 30-year-old. “Buff” you’d likely describe him: He is noticeably muscular, not a skinny old man. He’s an old Navy Master Chief who started a business in wholesale flowers, which distributes all over the United States. And he’s still working full time. Impressed, I asked his secret for such vigor. PT, he replied.
PT, or physical training, is a foundational element of the Navy. Every sailor starts his or her day with morning PT before carrying out their duties. Some 30 years later, this guy is still getting after it. He does push-ups, sit-ups, and pull-ups nearly every morning. Morning PT is what he attributes to his success not only in health, but also business. As he sees it, he has the business savvy and experience of an old guy and the energy and stamina of a college kid. A good combination for a successful life.
I’ve always been pretty fit. Lately, I’ve been trying to take it to the next level, to not just be “physically active,” but rather “high-performance fit.” There are plenty of sources for instruction; how to stay young and healthy isn’t a new idea after all. I mean, Herodotus wrote of finding the Fountain of Youth in the 5th century BCE. A couple thousand years later, it’s still on trend. One of my favorite sages giving health span advice is Peter Attia, MD. I’ve been a fan since I met him at TEDMED in 2013 and I marvel at the astounding body of work he has created since. A Johns Hopkins–trained surgeon, he has spent his career reviewing the scientific literature about longevity and sharing it as actionable content. His book, “Outlive: The Science and Art of Longevity” (New York: Penguin Random House, 2023) is a nice summary of his work. I recommend it.
Right now I’m switching between type 2 muscle fiber work (lots of jumping like my 2-year-old) and cardiovascular training including the aforementioned VO2max work. I cannot say that my patient inbox is any cleaner, or that I’m faster in the office, but I’m not flagging by the end of the day anymore. Master Chief challenged me to match his 10 pull-ups before he returns for his follow up visit. I’ll gladly give up Peloton sprints to work on that.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at dermnews@mdedge.com.
Tuesdays and Fridays are tough. Not so much because of clinic, but rather because of the 32 minutes before clinic that I’m on the Peloton bike. They are the mornings I dedicate to training VO2max.
Training VO2max, or maximal oxygen consumption, is simple. Spin for a leisurely, easy-breathing, 4 minutes, then for 4 minutes push yourself until you see the light of heaven and wish for death to come. Then relax for 4 minutes again. Repeat this cycle four to six times. Done justly, you will dread Tuesdays and Fridays too. The punishing cycle of a 4-minute push, then 4-minute recovery is, however, an excellent way to improve cardiovascular fitness. And no, I’m not training for the Boston Marathon, so why am I working so hard? Because I’m training for marathon clinic days for the next 20 years.
Now more than ever, I feel we have to be physically fit to deal with a physicians’ day’s work. It’s exhausting. The root cause is too much work, yes, but I believe being physically fit could help.
I was talking to an 86-year-old patient about this very topic recently. He was short, with a well-manicured goatee and shiny head. He stuck his arm out to shake my hand. “Glad we’re back to handshakes again, doc.” His grip was that of a 30-year-old. “Buff” you’d likely describe him: He is noticeably muscular, not a skinny old man. He’s an old Navy Master Chief who started a business in wholesale flowers, which distributes all over the United States. And he’s still working full time. Impressed, I asked his secret for such vigor. PT, he replied.
PT, or physical training, is a foundational element of the Navy. Every sailor starts his or her day with morning PT before carrying out their duties. Some 30 years later, this guy is still getting after it. He does push-ups, sit-ups, and pull-ups nearly every morning. Morning PT is what he attributes to his success not only in health, but also business. As he sees it, he has the business savvy and experience of an old guy and the energy and stamina of a college kid. A good combination for a successful life.
I’ve always been pretty fit. Lately, I’ve been trying to take it to the next level, to not just be “physically active,” but rather “high-performance fit.” There are plenty of sources for instruction; how to stay young and healthy isn’t a new idea after all. I mean, Herodotus wrote of finding the Fountain of Youth in the 5th century BCE. A couple thousand years later, it’s still on trend. One of my favorite sages giving health span advice is Peter Attia, MD. I’ve been a fan since I met him at TEDMED in 2013 and I marvel at the astounding body of work he has created since. A Johns Hopkins–trained surgeon, he has spent his career reviewing the scientific literature about longevity and sharing it as actionable content. His book, “Outlive: The Science and Art of Longevity” (New York: Penguin Random House, 2023) is a nice summary of his work. I recommend it.
Right now I’m switching between type 2 muscle fiber work (lots of jumping like my 2-year-old) and cardiovascular training including the aforementioned VO2max work. I cannot say that my patient inbox is any cleaner, or that I’m faster in the office, but I’m not flagging by the end of the day anymore. Master Chief challenged me to match his 10 pull-ups before he returns for his follow up visit. I’ll gladly give up Peloton sprints to work on that.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at dermnews@mdedge.com.
Review supports continued mask-wearing in health care visits
A new study urges people to continue wearing protective masks in medical settings, even though the U.S. public health emergency declaration around COVID-19 has expired.
Masks continue to lower the risk of catching the virus during medical visits, according to the study, published in Annals of Internal Medicine. And there was not much difference between wearing surgical masks and N95 respirators in health care settings.
The researchers reviewed 3 randomized trials and 21 observational studies to compare the effectiveness of those and cloth masks in reducing COVID-19 transmission.
Tara N. Palmore, MD, of George Washington University, Washington, and David K. Henderson, MD, of the National Institutes of Health, Bethesda, Md., wrote in an opinion article accompanying the study.
“In our enthusiasm to return to the appearance and feeling of normalcy, and as institutions decide which mitigation strategies to discontinue, we strongly advocate not discarding this important lesson learned for the sake of our patients’ safety,” Dr. Palmore and Dr. Henderson wrote.
Surgical masks limit the spread of aerosols and droplets from people who have the flu, coronaviruses or other respiratory viruses, CNN reported. And while masks are not 100% effective, they substantially lower the amount of virus put into the air via coughing and talking.
The study said one reason people should wear masks to medical settings is because “health care personnel are notorious for coming to work while ill.” Transmission from patient to staff and staff to patient is still possible, but rare, when both are masked.
The review authors reported no conflicts of interest. Dr. Palmore has received grants from the NIH, Rigel, Gilead, and AbbVie, and Dr. Henderson is a past president of the Society for Healthcare Epidemiology of America.
A version of this article first appeared on WebMD.com.
A new study urges people to continue wearing protective masks in medical settings, even though the U.S. public health emergency declaration around COVID-19 has expired.
Masks continue to lower the risk of catching the virus during medical visits, according to the study, published in Annals of Internal Medicine. And there was not much difference between wearing surgical masks and N95 respirators in health care settings.
The researchers reviewed 3 randomized trials and 21 observational studies to compare the effectiveness of those and cloth masks in reducing COVID-19 transmission.
Tara N. Palmore, MD, of George Washington University, Washington, and David K. Henderson, MD, of the National Institutes of Health, Bethesda, Md., wrote in an opinion article accompanying the study.
“In our enthusiasm to return to the appearance and feeling of normalcy, and as institutions decide which mitigation strategies to discontinue, we strongly advocate not discarding this important lesson learned for the sake of our patients’ safety,” Dr. Palmore and Dr. Henderson wrote.
Surgical masks limit the spread of aerosols and droplets from people who have the flu, coronaviruses or other respiratory viruses, CNN reported. And while masks are not 100% effective, they substantially lower the amount of virus put into the air via coughing and talking.
The study said one reason people should wear masks to medical settings is because “health care personnel are notorious for coming to work while ill.” Transmission from patient to staff and staff to patient is still possible, but rare, when both are masked.
The review authors reported no conflicts of interest. Dr. Palmore has received grants from the NIH, Rigel, Gilead, and AbbVie, and Dr. Henderson is a past president of the Society for Healthcare Epidemiology of America.
A version of this article first appeared on WebMD.com.
A new study urges people to continue wearing protective masks in medical settings, even though the U.S. public health emergency declaration around COVID-19 has expired.
Masks continue to lower the risk of catching the virus during medical visits, according to the study, published in Annals of Internal Medicine. And there was not much difference between wearing surgical masks and N95 respirators in health care settings.
The researchers reviewed 3 randomized trials and 21 observational studies to compare the effectiveness of those and cloth masks in reducing COVID-19 transmission.
Tara N. Palmore, MD, of George Washington University, Washington, and David K. Henderson, MD, of the National Institutes of Health, Bethesda, Md., wrote in an opinion article accompanying the study.
“In our enthusiasm to return to the appearance and feeling of normalcy, and as institutions decide which mitigation strategies to discontinue, we strongly advocate not discarding this important lesson learned for the sake of our patients’ safety,” Dr. Palmore and Dr. Henderson wrote.
Surgical masks limit the spread of aerosols and droplets from people who have the flu, coronaviruses or other respiratory viruses, CNN reported. And while masks are not 100% effective, they substantially lower the amount of virus put into the air via coughing and talking.
The study said one reason people should wear masks to medical settings is because “health care personnel are notorious for coming to work while ill.” Transmission from patient to staff and staff to patient is still possible, but rare, when both are masked.
The review authors reported no conflicts of interest. Dr. Palmore has received grants from the NIH, Rigel, Gilead, and AbbVie, and Dr. Henderson is a past president of the Society for Healthcare Epidemiology of America.
A version of this article first appeared on WebMD.com.
FROM ANNALS OF INTERNAL MEDICINE
The antimicrobial peptide that even Pharma can love
Fastest peptide north, south, east, aaaaand west of the Pecos
Bacterial infections are supposed to be simple. You get infected, you get an antibiotic to treat it. Easy. Some bacteria, though, don’t play by the rules. Those antibiotics may kill 99.9% of germs, but what about the 0.1% that gets left behind? With their fallen comrades out of the way, the accidentally drug resistant species are free to inherit the Earth.
Antibiotic resistance is thus a major concern for the medical community. Naturally, anything that prevents doctors from successfully curing sick people is a priority. Unless you’re a major pharmaceutical company that has been loath to develop new drugs that can beat antibiotic-resistant bacteria. Blah blah, time and money, blah blah, long time between development and market application, blah blah, no profit. We all know the story with pharmaceutical companies.
Research from other sources has continued, however, and Brazilian scientists recently published research involving a peptide known as plantaricin 149. This peptide, derived from the bacterium Lactobacillus plantarum, has been known for nearly 30 years to have antibacterial properties. Pln149 in its natural state, though, is not particularly efficient at bacteria-killing. Fortunately, we have science and technology on our side.
The researchers synthesized 20 analogs of Pln149, of which Pln149-PEP20 had the best results. The elegantly named compound is less than half the size of the original peptide, less toxic, and far better at killing any and all drug-resistant bacteria the researchers threw at it. How much better? Pln149-PEP20 started killing bacteria less than an hour after being introduced in lab trials.
The research is just in its early days – just because something is less toxic doesn’t necessarily mean you want to go and help yourself to it – but we can only hope that those lovely pharmaceutical companies deign to look down upon us and actually develop a drug utilizing Pln149-PEP20 to, you know, actually help sick people, instead of trying to build monopolies or avoiding paying billions in taxes. Yeah, we couldn’t keep a straight face through that last sentence either.
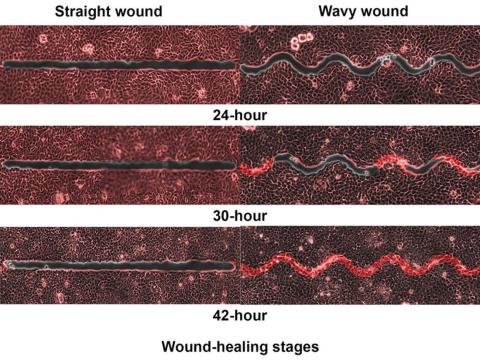
Speed healing: The wavy wound gets the swirl
Did you know that wavy wounds heal faster than straight wounds? Well, we didn’t, but apparently quite a few people did, because somebody has been trying to figure out why wavy wounds heal faster than straight ones. Do the surgeons know about this? How about you dermatologists? Wavy over straight? We’re the media. We’re supposed to report this kind of stuff. Maybe hit us with a tweet next time you do something important, or push a TikTok our way, okay?
You could be more like the investigators at Nanyang Technological University in Singapore, who figured out the why and then released a statement about it.
They created synthetic wounds – some straight, some wavy – in micropatterned hydrogel substrates that mimicked human skin. Then they used an advanced optical technique known as particle image velocimetry to measure fluid flow and learn how cells moved to close the wound gaps.
The wavy wounds “induced more complex collective cell movements, such as a swirly, vortex-like motion,” according to the written statement from NTU Singapore. In the straight wounds, cell movements paralleled the wound front, “moving in straight lines like a marching band,” they pointed out, unlike some researchers who never call us unless they need money.
Complex epithelial cell movements are better, it turns out. Over an observation period of 64 hours the NTU team found that the healing efficiency of wavy gaps – measured by the area covered by the cells over time – is nearly five times faster than straight gaps.
The complex motion “enabled cells to quickly connect with similar cells on the opposite site of the wound edge, forming a bridge and closing the wavy wound gaps faster than straight gaps,” explained lead author Xu Hongmei, a doctoral student at NTU’s School of Mechanical and Aerospace Engineering, who seems to have time to toss out a tumblr or two to keep the press informed.
As for the rest of you, would it kill you to pick up a phone once in a while? Maybe let a journalist know that you’re still alive? We have feelings too, you know, and we worry.
A little Jekyll, a little Hyde, and a little shop of horrors
More “Little Shop of Horrors” references are coming, so be prepared.
We begin with Triphyophyllum peltatum. This woody vine is of great interest to medical and pharmaceutical researchers because its constituents have shown promise against pancreatic cancer and leukemia cells, among others, along with the pathogens that cause malaria and other diseases. There is another side, however. T. peltatum also has a tendency to turn into a realistic Audrey II when deprived.
No, of course they’re not craving human flesh, but it does become … carnivorous in its appetite.
T. peltatum, native to the West African tropics and not found in a New York florist shop, has the unique ability to change its diet and development based on the environmental circumstances. For some unknown reason, the leaves would develop adhesive traps in the form of sticky drops that capture insect prey. The plant is notoriously hard to grow, however, so no one could study the transformation under lab conditions. Until now.
A group of German scientists “exposed the plant to different stress factors, including deficiencies of various nutrients, and studied how it responded to each,” said Dr. Traud Winkelmann of Leibniz University Hannover. “Only in one case were we able to observe the formation of traps: in the case of a lack of phosphorus.”
Well, there you have it: phosphorus. We need it for healthy bones and teeth, which this plant doesn’t have to worry about, unlike its Tony Award–nominated counterpart. The investigators hope that their findings could lead to “future molecular analyses that will help understand the origins of carnivory,” but we’re guessing that a certain singing alien species will be left out of that research.
Fastest peptide north, south, east, aaaaand west of the Pecos
Bacterial infections are supposed to be simple. You get infected, you get an antibiotic to treat it. Easy. Some bacteria, though, don’t play by the rules. Those antibiotics may kill 99.9% of germs, but what about the 0.1% that gets left behind? With their fallen comrades out of the way, the accidentally drug resistant species are free to inherit the Earth.
Antibiotic resistance is thus a major concern for the medical community. Naturally, anything that prevents doctors from successfully curing sick people is a priority. Unless you’re a major pharmaceutical company that has been loath to develop new drugs that can beat antibiotic-resistant bacteria. Blah blah, time and money, blah blah, long time between development and market application, blah blah, no profit. We all know the story with pharmaceutical companies.
Research from other sources has continued, however, and Brazilian scientists recently published research involving a peptide known as plantaricin 149. This peptide, derived from the bacterium Lactobacillus plantarum, has been known for nearly 30 years to have antibacterial properties. Pln149 in its natural state, though, is not particularly efficient at bacteria-killing. Fortunately, we have science and technology on our side.
The researchers synthesized 20 analogs of Pln149, of which Pln149-PEP20 had the best results. The elegantly named compound is less than half the size of the original peptide, less toxic, and far better at killing any and all drug-resistant bacteria the researchers threw at it. How much better? Pln149-PEP20 started killing bacteria less than an hour after being introduced in lab trials.
The research is just in its early days – just because something is less toxic doesn’t necessarily mean you want to go and help yourself to it – but we can only hope that those lovely pharmaceutical companies deign to look down upon us and actually develop a drug utilizing Pln149-PEP20 to, you know, actually help sick people, instead of trying to build monopolies or avoiding paying billions in taxes. Yeah, we couldn’t keep a straight face through that last sentence either.
Speed healing: The wavy wound gets the swirl
Did you know that wavy wounds heal faster than straight wounds? Well, we didn’t, but apparently quite a few people did, because somebody has been trying to figure out why wavy wounds heal faster than straight ones. Do the surgeons know about this? How about you dermatologists? Wavy over straight? We’re the media. We’re supposed to report this kind of stuff. Maybe hit us with a tweet next time you do something important, or push a TikTok our way, okay?
You could be more like the investigators at Nanyang Technological University in Singapore, who figured out the why and then released a statement about it.
They created synthetic wounds – some straight, some wavy – in micropatterned hydrogel substrates that mimicked human skin. Then they used an advanced optical technique known as particle image velocimetry to measure fluid flow and learn how cells moved to close the wound gaps.
The wavy wounds “induced more complex collective cell movements, such as a swirly, vortex-like motion,” according to the written statement from NTU Singapore. In the straight wounds, cell movements paralleled the wound front, “moving in straight lines like a marching band,” they pointed out, unlike some researchers who never call us unless they need money.
Complex epithelial cell movements are better, it turns out. Over an observation period of 64 hours the NTU team found that the healing efficiency of wavy gaps – measured by the area covered by the cells over time – is nearly five times faster than straight gaps.
The complex motion “enabled cells to quickly connect with similar cells on the opposite site of the wound edge, forming a bridge and closing the wavy wound gaps faster than straight gaps,” explained lead author Xu Hongmei, a doctoral student at NTU’s School of Mechanical and Aerospace Engineering, who seems to have time to toss out a tumblr or two to keep the press informed.
As for the rest of you, would it kill you to pick up a phone once in a while? Maybe let a journalist know that you’re still alive? We have feelings too, you know, and we worry.
A little Jekyll, a little Hyde, and a little shop of horrors
More “Little Shop of Horrors” references are coming, so be prepared.
We begin with Triphyophyllum peltatum. This woody vine is of great interest to medical and pharmaceutical researchers because its constituents have shown promise against pancreatic cancer and leukemia cells, among others, along with the pathogens that cause malaria and other diseases. There is another side, however. T. peltatum also has a tendency to turn into a realistic Audrey II when deprived.
No, of course they’re not craving human flesh, but it does become … carnivorous in its appetite.
T. peltatum, native to the West African tropics and not found in a New York florist shop, has the unique ability to change its diet and development based on the environmental circumstances. For some unknown reason, the leaves would develop adhesive traps in the form of sticky drops that capture insect prey. The plant is notoriously hard to grow, however, so no one could study the transformation under lab conditions. Until now.
A group of German scientists “exposed the plant to different stress factors, including deficiencies of various nutrients, and studied how it responded to each,” said Dr. Traud Winkelmann of Leibniz University Hannover. “Only in one case were we able to observe the formation of traps: in the case of a lack of phosphorus.”
Well, there you have it: phosphorus. We need it for healthy bones and teeth, which this plant doesn’t have to worry about, unlike its Tony Award–nominated counterpart. The investigators hope that their findings could lead to “future molecular analyses that will help understand the origins of carnivory,” but we’re guessing that a certain singing alien species will be left out of that research.
Fastest peptide north, south, east, aaaaand west of the Pecos
Bacterial infections are supposed to be simple. You get infected, you get an antibiotic to treat it. Easy. Some bacteria, though, don’t play by the rules. Those antibiotics may kill 99.9% of germs, but what about the 0.1% that gets left behind? With their fallen comrades out of the way, the accidentally drug resistant species are free to inherit the Earth.
Antibiotic resistance is thus a major concern for the medical community. Naturally, anything that prevents doctors from successfully curing sick people is a priority. Unless you’re a major pharmaceutical company that has been loath to develop new drugs that can beat antibiotic-resistant bacteria. Blah blah, time and money, blah blah, long time between development and market application, blah blah, no profit. We all know the story with pharmaceutical companies.
Research from other sources has continued, however, and Brazilian scientists recently published research involving a peptide known as plantaricin 149. This peptide, derived from the bacterium Lactobacillus plantarum, has been known for nearly 30 years to have antibacterial properties. Pln149 in its natural state, though, is not particularly efficient at bacteria-killing. Fortunately, we have science and technology on our side.
The researchers synthesized 20 analogs of Pln149, of which Pln149-PEP20 had the best results. The elegantly named compound is less than half the size of the original peptide, less toxic, and far better at killing any and all drug-resistant bacteria the researchers threw at it. How much better? Pln149-PEP20 started killing bacteria less than an hour after being introduced in lab trials.
The research is just in its early days – just because something is less toxic doesn’t necessarily mean you want to go and help yourself to it – but we can only hope that those lovely pharmaceutical companies deign to look down upon us and actually develop a drug utilizing Pln149-PEP20 to, you know, actually help sick people, instead of trying to build monopolies or avoiding paying billions in taxes. Yeah, we couldn’t keep a straight face through that last sentence either.
Speed healing: The wavy wound gets the swirl
Did you know that wavy wounds heal faster than straight wounds? Well, we didn’t, but apparently quite a few people did, because somebody has been trying to figure out why wavy wounds heal faster than straight ones. Do the surgeons know about this? How about you dermatologists? Wavy over straight? We’re the media. We’re supposed to report this kind of stuff. Maybe hit us with a tweet next time you do something important, or push a TikTok our way, okay?
You could be more like the investigators at Nanyang Technological University in Singapore, who figured out the why and then released a statement about it.
They created synthetic wounds – some straight, some wavy – in micropatterned hydrogel substrates that mimicked human skin. Then they used an advanced optical technique known as particle image velocimetry to measure fluid flow and learn how cells moved to close the wound gaps.
The wavy wounds “induced more complex collective cell movements, such as a swirly, vortex-like motion,” according to the written statement from NTU Singapore. In the straight wounds, cell movements paralleled the wound front, “moving in straight lines like a marching band,” they pointed out, unlike some researchers who never call us unless they need money.
Complex epithelial cell movements are better, it turns out. Over an observation period of 64 hours the NTU team found that the healing efficiency of wavy gaps – measured by the area covered by the cells over time – is nearly five times faster than straight gaps.
The complex motion “enabled cells to quickly connect with similar cells on the opposite site of the wound edge, forming a bridge and closing the wavy wound gaps faster than straight gaps,” explained lead author Xu Hongmei, a doctoral student at NTU’s School of Mechanical and Aerospace Engineering, who seems to have time to toss out a tumblr or two to keep the press informed.
As for the rest of you, would it kill you to pick up a phone once in a while? Maybe let a journalist know that you’re still alive? We have feelings too, you know, and we worry.
A little Jekyll, a little Hyde, and a little shop of horrors
More “Little Shop of Horrors” references are coming, so be prepared.
We begin with Triphyophyllum peltatum. This woody vine is of great interest to medical and pharmaceutical researchers because its constituents have shown promise against pancreatic cancer and leukemia cells, among others, along with the pathogens that cause malaria and other diseases. There is another side, however. T. peltatum also has a tendency to turn into a realistic Audrey II when deprived.
No, of course they’re not craving human flesh, but it does become … carnivorous in its appetite.
T. peltatum, native to the West African tropics and not found in a New York florist shop, has the unique ability to change its diet and development based on the environmental circumstances. For some unknown reason, the leaves would develop adhesive traps in the form of sticky drops that capture insect prey. The plant is notoriously hard to grow, however, so no one could study the transformation under lab conditions. Until now.
A group of German scientists “exposed the plant to different stress factors, including deficiencies of various nutrients, and studied how it responded to each,” said Dr. Traud Winkelmann of Leibniz University Hannover. “Only in one case were we able to observe the formation of traps: in the case of a lack of phosphorus.”
Well, there you have it: phosphorus. We need it for healthy bones and teeth, which this plant doesn’t have to worry about, unlike its Tony Award–nominated counterpart. The investigators hope that their findings could lead to “future molecular analyses that will help understand the origins of carnivory,” but we’re guessing that a certain singing alien species will be left out of that research.
Combination Therapies for Patients With Type 2 Diabetes
The treatment of type 2 diabetes (T2D) has traditionally followed a stepwise approach, beginning with metformin and continuing with the addition of other agents to achieve target glycemic control.
In this ReCAP, Dr Chika Anekwe, of the Massachusetts General Hospital Weight Center in Boston, Massachusetts, discusses categories for antidiabetic therapies, when to combine medications that have differing mechanisms, and the clinical benefits of combination therapy, such as a stronger response in the reduction of hemoglobin A1c as well as loss of body weight.
She also discusses tirzepatide, a dual therapy combining glucagon-like peptide-1 agonist and glucose-dependent insulinotropic polypeptide (GIP) agonist, which has shown clinically meaningful results for both blood sugar reduction and body weight reduction for patients with T2D.
--
Chika V. Anekwe, MD, MPH, Obesity Medicine Physician, Massachusetts General Hospital Weight Center, Boston, Massachusetts
Chika V. Anekwe, MD, MPH, has disclosed no relevant financial relationships.
The treatment of type 2 diabetes (T2D) has traditionally followed a stepwise approach, beginning with metformin and continuing with the addition of other agents to achieve target glycemic control.
In this ReCAP, Dr Chika Anekwe, of the Massachusetts General Hospital Weight Center in Boston, Massachusetts, discusses categories for antidiabetic therapies, when to combine medications that have differing mechanisms, and the clinical benefits of combination therapy, such as a stronger response in the reduction of hemoglobin A1c as well as loss of body weight.
She also discusses tirzepatide, a dual therapy combining glucagon-like peptide-1 agonist and glucose-dependent insulinotropic polypeptide (GIP) agonist, which has shown clinically meaningful results for both blood sugar reduction and body weight reduction for patients with T2D.
--
Chika V. Anekwe, MD, MPH, Obesity Medicine Physician, Massachusetts General Hospital Weight Center, Boston, Massachusetts
Chika V. Anekwe, MD, MPH, has disclosed no relevant financial relationships.
The treatment of type 2 diabetes (T2D) has traditionally followed a stepwise approach, beginning with metformin and continuing with the addition of other agents to achieve target glycemic control.
In this ReCAP, Dr Chika Anekwe, of the Massachusetts General Hospital Weight Center in Boston, Massachusetts, discusses categories for antidiabetic therapies, when to combine medications that have differing mechanisms, and the clinical benefits of combination therapy, such as a stronger response in the reduction of hemoglobin A1c as well as loss of body weight.
She also discusses tirzepatide, a dual therapy combining glucagon-like peptide-1 agonist and glucose-dependent insulinotropic polypeptide (GIP) agonist, which has shown clinically meaningful results for both blood sugar reduction and body weight reduction for patients with T2D.
--
Chika V. Anekwe, MD, MPH, Obesity Medicine Physician, Massachusetts General Hospital Weight Center, Boston, Massachusetts
Chika V. Anekwe, MD, MPH, has disclosed no relevant financial relationships.

Emotional eating isn’t all emotional
“Food gives me ‘hugs,’ ” Ms. S* said as her eyes lit up. Finally, after weeks of working together, she could articulate her complex relationship with food. She had been struggling to explain why she continued to eat when she was full or consumed foods she knew wouldn’t help her health.
Like millions of people struggling with their weight or the disease of obesity, Ms. S had tried multiple diets and programs but continued to return to unhelpful eating patterns. Ms. S was an emotional eater, and the pandemic only worsened her emotional eating. As a single professional forced to work from home during the pandemic, she became lonely. She went from working in a busy downtown office, training for half-marathons, and teaching live workout sessions to being alone daily. Her only “real” human interaction was when she ordered daily delivery meals of her favorite comfort foods. As a person with type 2 diabetes, she knew that her delivery habit was wrecking her health, but willpower wasn’t enough to make her stop.
Her psychologist referred her to our virtual integrative obesity practice to help her lose weight and find long-term solutions. Ms. S admitted that she knew what she was doing as an emotional eater. But like many emotional eaters, she didn’t know why or how to switch from emotional eating to eating based on her biological hunger signals. As a trained obesity expert and recovering emotional eater of 8 years, personally and professionally I can appreciate the challenges of emotional eating and how it can sabotage even the best weight loss plan. In this article, I will share facts and feelings that drive emotional eating. I aim to empower clinicians seeking to help patients with emotional eating.
Fact: Emotional eating isn’t all emotional
It’s important not to dismiss emotional eating as all emotion driven. Recall that hunger is hormonally regulated. There are two main hunger pathways: the homeostatic pathway and the hedonic pathway. The homeostatic pathway is our biological hunger pathway and is driven by the need for energy in calories. Conversely, hedonic eating is pleasure-driven and uses emotional stimuli to “bypass” the physical hunger/satisfaction signals.
Emotional eating falls under the hedonic pathway. As clinicians, the first step in helping a patient struggling with emotional eating is empathetically listening, then assessing for any physiologic causes.
Several factors can disrupt physiologic appetite regulation, such as sleep disturbances; high stress levels; and many medical conditions, including but not limited to obesity, diabetes, and polycystic ovarian syndrome. Such factors as insulin resistance and inflammation are a common link in these conditions. Both contribute to the pathophysiology of the changes in appetite and can influence other hormones that lead to reduced satisfaction after eating. Furthermore, mental health conditions may disrupt levels of neurotransmitters such as serotonin and dopamine, which can also cause appetite changes.
These settings of physiologically disrupted appetite can trigger hedonic eating. But the relationship is complex. For example, one way to research hedonic eating is by using the Power of Food Scale. Functional MRI studies show that people with higher Power of Food Scale readings have more brain activity in the visual cortex when they see highly palatable foods. While more studies are needed to better understand the clinical implications of this finding, it’s yet another indicator that “emotional” eating isn’t all emotional. It’s also physiologic.
Feelings: Patterns, personality, places, psychological factors
Physiology only explains part of emotional eating. Like Ms. S, emotional eaters have strong emotional connections to food and behavior patterns. Often, physiologic cues have been coupled with psychological habits.
For example, menses is a common physiologic trigger for stress-eating for many of my patients. Studies have shown that in addition to iron levels changing during menses, calcium, magnesium, and phosphorous levels also change. Emotionally, the discomfort of “that time of the month” can lead to solace in comfort foods such as chocolate in different forms. But this isn’t surprising, as cacao and its derivative, chocolate, are rich in iron and other minerals. The chocolate is actually addressing a physical and emotional need. It can be helpful to point out this association to your patients. Suggest choosing a lower-sugar form of chocolate, such as dark chocolate, or even trying cacao nibs, while addressing any emotions.
But physiologic conditions and patterns aren’t the only emotional eating triggers. Places and psychological conditions can also trigger emotional eating.
Places and people
Celebrations, vacations, proximity to certain restaurants, exposure to food marketing, and major life shifts can lead to increased hedonic eating. Helping patients recognize this connection opens the door to advance preparation for these situations.
Psychological conditions can be connected to emotional eating. It’s important to screen for mental health conditions and past traumas. For example, emotional eating could be a symptom of binge eating disorder, major depression, or generalized anxiety disorder. Childhood trauma is associated with disordered eating. The adverse childhood events quiz can be used clinically.
Emotional eating can lead to feelings of guilt, shame, and negative self-talk. It’s helpful to offer patients reassurance and encourage self-compassion. After all, it’s natural to eat. The goal isn’t to stop eating but to eat on the basis of physiologic needs.
Putting it together: Addressing the facts and feelings of emotional eating
1. Treat biological causes that impact physiologic hunger and trigger emotional eating.
2. Triggers: Address patterns, places/people, psychological events.
3. Transition to non-food rewards; the key to emotional eating is eating. While healthier substitutes can be a short-term solution for improving eating behaviors, ultimately, helping patients find non-food ways to address emotions is invaluable.
4. Stress management: Offer your patients ways to decrease stress levels through mindfulness and other techniques.
5. Professional support: Creating a multidisciplinary team is helpful, given the complexity of emotional eating. In addition to the primary care physician/clinician, other team members may include:
- Psychologist
- Psychiatrist
- coach and/or certified wellness coaches
- Obesity specialist
Back to Ms. S
Ms. S is doing well. We started her on a GLP-1 agonist to address her underlying insulin resistance. Together we’ve found creative ways to satisfy her loneliness, such as volunteering and teaching virtual workout classes. Her emotional eating has decreased by over 60%, and we continue to discover new strategies to address her emotional eating triggers.
Conclusion
Despite being common, the impact of emotional eating is often minimized. With no DSM-5 criteria or ICD-11 code, it’s easy to dismiss emotional eating clinically. However, emotional eating is common and associated with weight gain.
In light of the obesity epidemic, this significance can’t be overlooked. Thankfully we have groundbreaking medications to address the homeostatic hunger pathway and physiologic drivers of emotional eating, but they’re not a substitute for addressing the psychosocial components of emotional eating.
As clinicians, we can have a meaningful impact on our patients’ lives beyond writing a prescription.
*Name/initial changed for privacy.
Sylvia Gonsahn-Bollie, MD, DipABOM, is an integrative obesity specialist focused on individualized solutions for emotional and biological overeating.
A version of this article first appeared on Medscape.com.
“Food gives me ‘hugs,’ ” Ms. S* said as her eyes lit up. Finally, after weeks of working together, she could articulate her complex relationship with food. She had been struggling to explain why she continued to eat when she was full or consumed foods she knew wouldn’t help her health.
Like millions of people struggling with their weight or the disease of obesity, Ms. S had tried multiple diets and programs but continued to return to unhelpful eating patterns. Ms. S was an emotional eater, and the pandemic only worsened her emotional eating. As a single professional forced to work from home during the pandemic, she became lonely. She went from working in a busy downtown office, training for half-marathons, and teaching live workout sessions to being alone daily. Her only “real” human interaction was when she ordered daily delivery meals of her favorite comfort foods. As a person with type 2 diabetes, she knew that her delivery habit was wrecking her health, but willpower wasn’t enough to make her stop.
Her psychologist referred her to our virtual integrative obesity practice to help her lose weight and find long-term solutions. Ms. S admitted that she knew what she was doing as an emotional eater. But like many emotional eaters, she didn’t know why or how to switch from emotional eating to eating based on her biological hunger signals. As a trained obesity expert and recovering emotional eater of 8 years, personally and professionally I can appreciate the challenges of emotional eating and how it can sabotage even the best weight loss plan. In this article, I will share facts and feelings that drive emotional eating. I aim to empower clinicians seeking to help patients with emotional eating.
Fact: Emotional eating isn’t all emotional
It’s important not to dismiss emotional eating as all emotion driven. Recall that hunger is hormonally regulated. There are two main hunger pathways: the homeostatic pathway and the hedonic pathway. The homeostatic pathway is our biological hunger pathway and is driven by the need for energy in calories. Conversely, hedonic eating is pleasure-driven and uses emotional stimuli to “bypass” the physical hunger/satisfaction signals.
Emotional eating falls under the hedonic pathway. As clinicians, the first step in helping a patient struggling with emotional eating is empathetically listening, then assessing for any physiologic causes.
Several factors can disrupt physiologic appetite regulation, such as sleep disturbances; high stress levels; and many medical conditions, including but not limited to obesity, diabetes, and polycystic ovarian syndrome. Such factors as insulin resistance and inflammation are a common link in these conditions. Both contribute to the pathophysiology of the changes in appetite and can influence other hormones that lead to reduced satisfaction after eating. Furthermore, mental health conditions may disrupt levels of neurotransmitters such as serotonin and dopamine, which can also cause appetite changes.
These settings of physiologically disrupted appetite can trigger hedonic eating. But the relationship is complex. For example, one way to research hedonic eating is by using the Power of Food Scale. Functional MRI studies show that people with higher Power of Food Scale readings have more brain activity in the visual cortex when they see highly palatable foods. While more studies are needed to better understand the clinical implications of this finding, it’s yet another indicator that “emotional” eating isn’t all emotional. It’s also physiologic.
Feelings: Patterns, personality, places, psychological factors
Physiology only explains part of emotional eating. Like Ms. S, emotional eaters have strong emotional connections to food and behavior patterns. Often, physiologic cues have been coupled with psychological habits.
For example, menses is a common physiologic trigger for stress-eating for many of my patients. Studies have shown that in addition to iron levels changing during menses, calcium, magnesium, and phosphorous levels also change. Emotionally, the discomfort of “that time of the month” can lead to solace in comfort foods such as chocolate in different forms. But this isn’t surprising, as cacao and its derivative, chocolate, are rich in iron and other minerals. The chocolate is actually addressing a physical and emotional need. It can be helpful to point out this association to your patients. Suggest choosing a lower-sugar form of chocolate, such as dark chocolate, or even trying cacao nibs, while addressing any emotions.
But physiologic conditions and patterns aren’t the only emotional eating triggers. Places and psychological conditions can also trigger emotional eating.
Places and people
Celebrations, vacations, proximity to certain restaurants, exposure to food marketing, and major life shifts can lead to increased hedonic eating. Helping patients recognize this connection opens the door to advance preparation for these situations.
Psychological conditions can be connected to emotional eating. It’s important to screen for mental health conditions and past traumas. For example, emotional eating could be a symptom of binge eating disorder, major depression, or generalized anxiety disorder. Childhood trauma is associated with disordered eating. The adverse childhood events quiz can be used clinically.
Emotional eating can lead to feelings of guilt, shame, and negative self-talk. It’s helpful to offer patients reassurance and encourage self-compassion. After all, it’s natural to eat. The goal isn’t to stop eating but to eat on the basis of physiologic needs.
Putting it together: Addressing the facts and feelings of emotional eating
1. Treat biological causes that impact physiologic hunger and trigger emotional eating.
2. Triggers: Address patterns, places/people, psychological events.
3. Transition to non-food rewards; the key to emotional eating is eating. While healthier substitutes can be a short-term solution for improving eating behaviors, ultimately, helping patients find non-food ways to address emotions is invaluable.
4. Stress management: Offer your patients ways to decrease stress levels through mindfulness and other techniques.
5. Professional support: Creating a multidisciplinary team is helpful, given the complexity of emotional eating. In addition to the primary care physician/clinician, other team members may include:
- Psychologist
- Psychiatrist
- coach and/or certified wellness coaches
- Obesity specialist
Back to Ms. S
Ms. S is doing well. We started her on a GLP-1 agonist to address her underlying insulin resistance. Together we’ve found creative ways to satisfy her loneliness, such as volunteering and teaching virtual workout classes. Her emotional eating has decreased by over 60%, and we continue to discover new strategies to address her emotional eating triggers.
Conclusion
Despite being common, the impact of emotional eating is often minimized. With no DSM-5 criteria or ICD-11 code, it’s easy to dismiss emotional eating clinically. However, emotional eating is common and associated with weight gain.
In light of the obesity epidemic, this significance can’t be overlooked. Thankfully we have groundbreaking medications to address the homeostatic hunger pathway and physiologic drivers of emotional eating, but they’re not a substitute for addressing the psychosocial components of emotional eating.
As clinicians, we can have a meaningful impact on our patients’ lives beyond writing a prescription.
*Name/initial changed for privacy.
Sylvia Gonsahn-Bollie, MD, DipABOM, is an integrative obesity specialist focused on individualized solutions for emotional and biological overeating.
A version of this article first appeared on Medscape.com.
“Food gives me ‘hugs,’ ” Ms. S* said as her eyes lit up. Finally, after weeks of working together, she could articulate her complex relationship with food. She had been struggling to explain why she continued to eat when she was full or consumed foods she knew wouldn’t help her health.
Like millions of people struggling with their weight or the disease of obesity, Ms. S had tried multiple diets and programs but continued to return to unhelpful eating patterns. Ms. S was an emotional eater, and the pandemic only worsened her emotional eating. As a single professional forced to work from home during the pandemic, she became lonely. She went from working in a busy downtown office, training for half-marathons, and teaching live workout sessions to being alone daily. Her only “real” human interaction was when she ordered daily delivery meals of her favorite comfort foods. As a person with type 2 diabetes, she knew that her delivery habit was wrecking her health, but willpower wasn’t enough to make her stop.
Her psychologist referred her to our virtual integrative obesity practice to help her lose weight and find long-term solutions. Ms. S admitted that she knew what she was doing as an emotional eater. But like many emotional eaters, she didn’t know why or how to switch from emotional eating to eating based on her biological hunger signals. As a trained obesity expert and recovering emotional eater of 8 years, personally and professionally I can appreciate the challenges of emotional eating and how it can sabotage even the best weight loss plan. In this article, I will share facts and feelings that drive emotional eating. I aim to empower clinicians seeking to help patients with emotional eating.
Fact: Emotional eating isn’t all emotional
It’s important not to dismiss emotional eating as all emotion driven. Recall that hunger is hormonally regulated. There are two main hunger pathways: the homeostatic pathway and the hedonic pathway. The homeostatic pathway is our biological hunger pathway and is driven by the need for energy in calories. Conversely, hedonic eating is pleasure-driven and uses emotional stimuli to “bypass” the physical hunger/satisfaction signals.
Emotional eating falls under the hedonic pathway. As clinicians, the first step in helping a patient struggling with emotional eating is empathetically listening, then assessing for any physiologic causes.
Several factors can disrupt physiologic appetite regulation, such as sleep disturbances; high stress levels; and many medical conditions, including but not limited to obesity, diabetes, and polycystic ovarian syndrome. Such factors as insulin resistance and inflammation are a common link in these conditions. Both contribute to the pathophysiology of the changes in appetite and can influence other hormones that lead to reduced satisfaction after eating. Furthermore, mental health conditions may disrupt levels of neurotransmitters such as serotonin and dopamine, which can also cause appetite changes.
These settings of physiologically disrupted appetite can trigger hedonic eating. But the relationship is complex. For example, one way to research hedonic eating is by using the Power of Food Scale. Functional MRI studies show that people with higher Power of Food Scale readings have more brain activity in the visual cortex when they see highly palatable foods. While more studies are needed to better understand the clinical implications of this finding, it’s yet another indicator that “emotional” eating isn’t all emotional. It’s also physiologic.
Feelings: Patterns, personality, places, psychological factors
Physiology only explains part of emotional eating. Like Ms. S, emotional eaters have strong emotional connections to food and behavior patterns. Often, physiologic cues have been coupled with psychological habits.
For example, menses is a common physiologic trigger for stress-eating for many of my patients. Studies have shown that in addition to iron levels changing during menses, calcium, magnesium, and phosphorous levels also change. Emotionally, the discomfort of “that time of the month” can lead to solace in comfort foods such as chocolate in different forms. But this isn’t surprising, as cacao and its derivative, chocolate, are rich in iron and other minerals. The chocolate is actually addressing a physical and emotional need. It can be helpful to point out this association to your patients. Suggest choosing a lower-sugar form of chocolate, such as dark chocolate, or even trying cacao nibs, while addressing any emotions.
But physiologic conditions and patterns aren’t the only emotional eating triggers. Places and psychological conditions can also trigger emotional eating.
Places and people
Celebrations, vacations, proximity to certain restaurants, exposure to food marketing, and major life shifts can lead to increased hedonic eating. Helping patients recognize this connection opens the door to advance preparation for these situations.
Psychological conditions can be connected to emotional eating. It’s important to screen for mental health conditions and past traumas. For example, emotional eating could be a symptom of binge eating disorder, major depression, or generalized anxiety disorder. Childhood trauma is associated with disordered eating. The adverse childhood events quiz can be used clinically.
Emotional eating can lead to feelings of guilt, shame, and negative self-talk. It’s helpful to offer patients reassurance and encourage self-compassion. After all, it’s natural to eat. The goal isn’t to stop eating but to eat on the basis of physiologic needs.
Putting it together: Addressing the facts and feelings of emotional eating
1. Treat biological causes that impact physiologic hunger and trigger emotional eating.
2. Triggers: Address patterns, places/people, psychological events.
3. Transition to non-food rewards; the key to emotional eating is eating. While healthier substitutes can be a short-term solution for improving eating behaviors, ultimately, helping patients find non-food ways to address emotions is invaluable.
4. Stress management: Offer your patients ways to decrease stress levels through mindfulness and other techniques.
5. Professional support: Creating a multidisciplinary team is helpful, given the complexity of emotional eating. In addition to the primary care physician/clinician, other team members may include:
- Psychologist
- Psychiatrist
- coach and/or certified wellness coaches
- Obesity specialist
Back to Ms. S
Ms. S is doing well. We started her on a GLP-1 agonist to address her underlying insulin resistance. Together we’ve found creative ways to satisfy her loneliness, such as volunteering and teaching virtual workout classes. Her emotional eating has decreased by over 60%, and we continue to discover new strategies to address her emotional eating triggers.
Conclusion
Despite being common, the impact of emotional eating is often minimized. With no DSM-5 criteria or ICD-11 code, it’s easy to dismiss emotional eating clinically. However, emotional eating is common and associated with weight gain.
In light of the obesity epidemic, this significance can’t be overlooked. Thankfully we have groundbreaking medications to address the homeostatic hunger pathway and physiologic drivers of emotional eating, but they’re not a substitute for addressing the psychosocial components of emotional eating.
As clinicians, we can have a meaningful impact on our patients’ lives beyond writing a prescription.
*Name/initial changed for privacy.
Sylvia Gonsahn-Bollie, MD, DipABOM, is an integrative obesity specialist focused on individualized solutions for emotional and biological overeating.
A version of this article first appeared on Medscape.com.
TransCon PTH nears U.S. approval for hypoparathyroidism?
SEATTLE –
Findings from 110-week phase 2 data for the once-daily investigational parathyroid hormone (PTH) replacement drug were recently presented at the annual scientific & clinical congress of the American Association of Clinical Endocrinology.
Overall, the drug was associated with independence from conventional calcium and active vitamin D therapy in most patients at 110 weeks, with no discontinuations due to adverse effects.
“Patients with hypoparathyroidism have low serum calcium levels and struggle with quality of life and biochemical abnormalities. The data from the TransCon PTH studies seem to show that a lot of these abnormalities can be reversed,” presenter Mishaela R. Rubin, MD, said in an interview.
Other PTH replacement therapies such as Nupara (now discontinued) and teriparatide (off-label) have been used in some patients with hypoparathyroidism.
However, “[TransCon PTH] is delivered in such a way as to have a prolonged half-life, so that’s kind of a special benefit that it has,” added Dr. Rubin of the division of endocrinology and metabolic bone disease, department of medicine, Columbia University, New York.
Asked to comment, session moderator Thanh Hoang, DO, of Walter Reed National Military Medical Center, Silver Spring, Md., said: “I think it’s a very promising medication because right now we don’t have a lot of options ... I think it would help a lot of patients.”
Approval denied, company addressing concerns
On May 1, the Food and Drug Administration issued a complete response letter, signaling denial of approval for the TransCon PTH, citing concerns related to manufacturing control of the product’s drug/device combination product, but not about the product’s safety and efficacy, according to an Ascendis statement.
The company is now working with the FDA to address these issues and is awaiting a European Union decision later this year.
The FDA did not request that the company conduct further clinical trials of TransCon PTH, which now include published 26-week phase 2 and phase 3 data along with the current longer-term phase 2 data presented at AACE.
“The company has said that they’re hopeful the issues will be addressable and that the FDA did not have any concerns about safety,” Dr. Rubin said in an interview.
Calcium normalized, bone turnover improved
Dr. Rubin presented long-term efficacy and safety data from the Phase 2 PaTH Forward trial, which involved 57 of the initial 59 participants who completed week 110 of an open-label extension of the trial.
During the first 4 weeks, patients had been randomized to TransCon PTH at fixed doses of 15 µg/day, 18 µg/day, 21 µg/day, or placebo. After week 4, all patients switched to TransCon PTH titrated to doses of 6-60 µg/day along with conventional therapy, with the goal of maintaining normocalcemia.
Participants were a mean age of 50 years, 81% were women, and 92% were White. Causes of hypoparathyroidism were neck surgery in 80%, autoimmune disease in 2%, and idiopathic disease in 19%. Disease duration was 12 years (range 1-39), and all were taking conventional therapy including calcium and active vitamin D (calcitriol or alfacaldiol).
At 110 weeks, all 57 patients were able to stop taking active vitamin D, and 53 of the 57 (93%) patients achieved independence from conventional therapy, defined as taking 0 µg/day of active vitamin D and no more than 600 mg/day of calcium (the dietary supplement dose). A total of 44 (77%) patients were not taking any calcium or active vitamin D.
“This really establishes the durability up to 2 years of keeping people off conventional therapy,” Dr. Rubin said during her presentation.
There was an initial uptick to 9.4 mg/dL in mean serum calcium, as some participants were still taking active vitamin D, but that dropped to 8.9 mg/dL by week 26. Mean 24-hour urine calcium dropped from 428 mg/day at baseline to 173 mg/day by week 26. Both serum calcium and urine calcium remained in the normal range through week 110 in all patients, at 8.6 mg/dL and 167 mg/day, respectively.
“This is a really important outcome because we know that high urine calcium in these patients sets them at risk for going on to develop nephrocalcinosis, nephrolithiasis, and ultimately, chronic kidney disease,” Dr. Rubin said.
Serum levels of two bone formation markers peaked at 12 weeks after initiation of TransCon PTH. Both trended downward thereafter through week 110 to levels approximating those of age- and sex-matched controls.
“Both markers started off low, consistent with hypoparathyroidism, but with initiation of TransCon PTH we see a robust increase in bone turnover markers, almost as if the bone is ‘waking up,’ if you will. And this is consistent with calcium being mobilized from the skeleton and going into the circulation,” Dr. Rubin explained.
Bone mineral density assessed by dual-energy x-ray absorptiometry normalized, primarily in the first 26 weeks. For lumbar spine L1-L4, mean Z-scores dropped from 1.6 to 1.0 at 26 weeks and down to 0.7 by week 100. For total hip, those values were 1.0, 0.6, and 0.4, respectively. The values approached age- and sex-matched norms, Dr. Rubin noted, to “perhaps where their skeleton would be if they hadn’t had hypoparathyroidism.”
Overall 56 of the 57 (94.9%) patients reported treatment-emergent adverse events, of which 25 (42.4%) were treatment related and none were deemed serious. There were no treatment-emergent adverse events related to hypercalcemia or hypocalcemia leading to health care visits or hospitalization, none leading to discontinuation of study drug, and none to death.
“So overall, a reassuring safety profile,” Dr. Rubin said. “We look forward to presenting the next 2 years’ worth of data to the end of the open-label extension study.”
Dr. Rubin is a paid researcher for Ascendis, which funded the study. Dr. Hoang has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
SEATTLE –
Findings from 110-week phase 2 data for the once-daily investigational parathyroid hormone (PTH) replacement drug were recently presented at the annual scientific & clinical congress of the American Association of Clinical Endocrinology.
Overall, the drug was associated with independence from conventional calcium and active vitamin D therapy in most patients at 110 weeks, with no discontinuations due to adverse effects.
“Patients with hypoparathyroidism have low serum calcium levels and struggle with quality of life and biochemical abnormalities. The data from the TransCon PTH studies seem to show that a lot of these abnormalities can be reversed,” presenter Mishaela R. Rubin, MD, said in an interview.
Other PTH replacement therapies such as Nupara (now discontinued) and teriparatide (off-label) have been used in some patients with hypoparathyroidism.
However, “[TransCon PTH] is delivered in such a way as to have a prolonged half-life, so that’s kind of a special benefit that it has,” added Dr. Rubin of the division of endocrinology and metabolic bone disease, department of medicine, Columbia University, New York.
Asked to comment, session moderator Thanh Hoang, DO, of Walter Reed National Military Medical Center, Silver Spring, Md., said: “I think it’s a very promising medication because right now we don’t have a lot of options ... I think it would help a lot of patients.”
Approval denied, company addressing concerns
On May 1, the Food and Drug Administration issued a complete response letter, signaling denial of approval for the TransCon PTH, citing concerns related to manufacturing control of the product’s drug/device combination product, but not about the product’s safety and efficacy, according to an Ascendis statement.
The company is now working with the FDA to address these issues and is awaiting a European Union decision later this year.
The FDA did not request that the company conduct further clinical trials of TransCon PTH, which now include published 26-week phase 2 and phase 3 data along with the current longer-term phase 2 data presented at AACE.
“The company has said that they’re hopeful the issues will be addressable and that the FDA did not have any concerns about safety,” Dr. Rubin said in an interview.
Calcium normalized, bone turnover improved
Dr. Rubin presented long-term efficacy and safety data from the Phase 2 PaTH Forward trial, which involved 57 of the initial 59 participants who completed week 110 of an open-label extension of the trial.
During the first 4 weeks, patients had been randomized to TransCon PTH at fixed doses of 15 µg/day, 18 µg/day, 21 µg/day, or placebo. After week 4, all patients switched to TransCon PTH titrated to doses of 6-60 µg/day along with conventional therapy, with the goal of maintaining normocalcemia.
Participants were a mean age of 50 years, 81% were women, and 92% were White. Causes of hypoparathyroidism were neck surgery in 80%, autoimmune disease in 2%, and idiopathic disease in 19%. Disease duration was 12 years (range 1-39), and all were taking conventional therapy including calcium and active vitamin D (calcitriol or alfacaldiol).
At 110 weeks, all 57 patients were able to stop taking active vitamin D, and 53 of the 57 (93%) patients achieved independence from conventional therapy, defined as taking 0 µg/day of active vitamin D and no more than 600 mg/day of calcium (the dietary supplement dose). A total of 44 (77%) patients were not taking any calcium or active vitamin D.
“This really establishes the durability up to 2 years of keeping people off conventional therapy,” Dr. Rubin said during her presentation.
There was an initial uptick to 9.4 mg/dL in mean serum calcium, as some participants were still taking active vitamin D, but that dropped to 8.9 mg/dL by week 26. Mean 24-hour urine calcium dropped from 428 mg/day at baseline to 173 mg/day by week 26. Both serum calcium and urine calcium remained in the normal range through week 110 in all patients, at 8.6 mg/dL and 167 mg/day, respectively.
“This is a really important outcome because we know that high urine calcium in these patients sets them at risk for going on to develop nephrocalcinosis, nephrolithiasis, and ultimately, chronic kidney disease,” Dr. Rubin said.
Serum levels of two bone formation markers peaked at 12 weeks after initiation of TransCon PTH. Both trended downward thereafter through week 110 to levels approximating those of age- and sex-matched controls.
“Both markers started off low, consistent with hypoparathyroidism, but with initiation of TransCon PTH we see a robust increase in bone turnover markers, almost as if the bone is ‘waking up,’ if you will. And this is consistent with calcium being mobilized from the skeleton and going into the circulation,” Dr. Rubin explained.
Bone mineral density assessed by dual-energy x-ray absorptiometry normalized, primarily in the first 26 weeks. For lumbar spine L1-L4, mean Z-scores dropped from 1.6 to 1.0 at 26 weeks and down to 0.7 by week 100. For total hip, those values were 1.0, 0.6, and 0.4, respectively. The values approached age- and sex-matched norms, Dr. Rubin noted, to “perhaps where their skeleton would be if they hadn’t had hypoparathyroidism.”
Overall 56 of the 57 (94.9%) patients reported treatment-emergent adverse events, of which 25 (42.4%) were treatment related and none were deemed serious. There were no treatment-emergent adverse events related to hypercalcemia or hypocalcemia leading to health care visits or hospitalization, none leading to discontinuation of study drug, and none to death.
“So overall, a reassuring safety profile,” Dr. Rubin said. “We look forward to presenting the next 2 years’ worth of data to the end of the open-label extension study.”
Dr. Rubin is a paid researcher for Ascendis, which funded the study. Dr. Hoang has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
SEATTLE –
Findings from 110-week phase 2 data for the once-daily investigational parathyroid hormone (PTH) replacement drug were recently presented at the annual scientific & clinical congress of the American Association of Clinical Endocrinology.
Overall, the drug was associated with independence from conventional calcium and active vitamin D therapy in most patients at 110 weeks, with no discontinuations due to adverse effects.
“Patients with hypoparathyroidism have low serum calcium levels and struggle with quality of life and biochemical abnormalities. The data from the TransCon PTH studies seem to show that a lot of these abnormalities can be reversed,” presenter Mishaela R. Rubin, MD, said in an interview.
Other PTH replacement therapies such as Nupara (now discontinued) and teriparatide (off-label) have been used in some patients with hypoparathyroidism.
However, “[TransCon PTH] is delivered in such a way as to have a prolonged half-life, so that’s kind of a special benefit that it has,” added Dr. Rubin of the division of endocrinology and metabolic bone disease, department of medicine, Columbia University, New York.
Asked to comment, session moderator Thanh Hoang, DO, of Walter Reed National Military Medical Center, Silver Spring, Md., said: “I think it’s a very promising medication because right now we don’t have a lot of options ... I think it would help a lot of patients.”
Approval denied, company addressing concerns
On May 1, the Food and Drug Administration issued a complete response letter, signaling denial of approval for the TransCon PTH, citing concerns related to manufacturing control of the product’s drug/device combination product, but not about the product’s safety and efficacy, according to an Ascendis statement.
The company is now working with the FDA to address these issues and is awaiting a European Union decision later this year.
The FDA did not request that the company conduct further clinical trials of TransCon PTH, which now include published 26-week phase 2 and phase 3 data along with the current longer-term phase 2 data presented at AACE.
“The company has said that they’re hopeful the issues will be addressable and that the FDA did not have any concerns about safety,” Dr. Rubin said in an interview.
Calcium normalized, bone turnover improved
Dr. Rubin presented long-term efficacy and safety data from the Phase 2 PaTH Forward trial, which involved 57 of the initial 59 participants who completed week 110 of an open-label extension of the trial.
During the first 4 weeks, patients had been randomized to TransCon PTH at fixed doses of 15 µg/day, 18 µg/day, 21 µg/day, or placebo. After week 4, all patients switched to TransCon PTH titrated to doses of 6-60 µg/day along with conventional therapy, with the goal of maintaining normocalcemia.
Participants were a mean age of 50 years, 81% were women, and 92% were White. Causes of hypoparathyroidism were neck surgery in 80%, autoimmune disease in 2%, and idiopathic disease in 19%. Disease duration was 12 years (range 1-39), and all were taking conventional therapy including calcium and active vitamin D (calcitriol or alfacaldiol).
At 110 weeks, all 57 patients were able to stop taking active vitamin D, and 53 of the 57 (93%) patients achieved independence from conventional therapy, defined as taking 0 µg/day of active vitamin D and no more than 600 mg/day of calcium (the dietary supplement dose). A total of 44 (77%) patients were not taking any calcium or active vitamin D.
“This really establishes the durability up to 2 years of keeping people off conventional therapy,” Dr. Rubin said during her presentation.
There was an initial uptick to 9.4 mg/dL in mean serum calcium, as some participants were still taking active vitamin D, but that dropped to 8.9 mg/dL by week 26. Mean 24-hour urine calcium dropped from 428 mg/day at baseline to 173 mg/day by week 26. Both serum calcium and urine calcium remained in the normal range through week 110 in all patients, at 8.6 mg/dL and 167 mg/day, respectively.
“This is a really important outcome because we know that high urine calcium in these patients sets them at risk for going on to develop nephrocalcinosis, nephrolithiasis, and ultimately, chronic kidney disease,” Dr. Rubin said.
Serum levels of two bone formation markers peaked at 12 weeks after initiation of TransCon PTH. Both trended downward thereafter through week 110 to levels approximating those of age- and sex-matched controls.
“Both markers started off low, consistent with hypoparathyroidism, but with initiation of TransCon PTH we see a robust increase in bone turnover markers, almost as if the bone is ‘waking up,’ if you will. And this is consistent with calcium being mobilized from the skeleton and going into the circulation,” Dr. Rubin explained.
Bone mineral density assessed by dual-energy x-ray absorptiometry normalized, primarily in the first 26 weeks. For lumbar spine L1-L4, mean Z-scores dropped from 1.6 to 1.0 at 26 weeks and down to 0.7 by week 100. For total hip, those values were 1.0, 0.6, and 0.4, respectively. The values approached age- and sex-matched norms, Dr. Rubin noted, to “perhaps where their skeleton would be if they hadn’t had hypoparathyroidism.”
Overall 56 of the 57 (94.9%) patients reported treatment-emergent adverse events, of which 25 (42.4%) were treatment related and none were deemed serious. There were no treatment-emergent adverse events related to hypercalcemia or hypocalcemia leading to health care visits or hospitalization, none leading to discontinuation of study drug, and none to death.
“So overall, a reassuring safety profile,” Dr. Rubin said. “We look forward to presenting the next 2 years’ worth of data to the end of the open-label extension study.”
Dr. Rubin is a paid researcher for Ascendis, which funded the study. Dr. Hoang has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
AT AACE 2023
Docs fervently hope federal ban on noncompete clauses goes through
The Federal Trade Commission’s proposed regulation that would ban noncompete agreements across the country seems like potential good news for doctors. Of course, many hospitals and employers are against it. As a result, the FTC’s sweeping proposal has tongues wagging on both sides of the issue.
Many physicians are thrilled that they may soon have more control over their career and not be stuck in jobs where they feel frustrated, underpaid, or blocked in their progress.
As of 2018, as many as 45% of primary care physicians had inked such agreements with their employers.
Typically, the agreements prevent physicians from practicing medicine with a new employer for a defined period within a specific geographic area. No matter how attractive an alternate offer of employment might be, doctors are bound by the agreements to say no if the offer exists in that defined area and time period.
The period for public comment on the proposed regulation ended on April 19, and there is currently no set date for a decision.
In a Medscape poll of 558 physicians, more than 9 out of 10 respondents said that they were either currently bound by a noncompete clause or that they had been bound by one in the past that had forced them to temporarily stop working, commute long distances, move to a different area, or switch fields.
The new proposal would make it illegal for an employer, such as a hospital or large group, to enter a noncompete with a worker; maintain a noncompete with a worker; or represent to a worker, under certain circumstances, that the worker is subject to a noncompete.
It also would not only ban future noncompete agreements but also retroactively invalidate existing ones. The FTC reasons that noncompete clauses could potentially increase worker earnings as well as lower health care costs by billions of dollars. If the ruling were to move forward, it would represent part of President Biden’s “worker-forward” priorities, focusing on how competition can be a good thing for employees. The President billed the FTC’s announcement as a “huge win for workers.”
In its statements on the proposed ban, the FTC claimed that it could lower consumer prices across the board by as much as $150 billion per year and return nearly $300 million to workers each year.
However, even if passed, the draft rule would keep in place nonsolicitation rules that many health care organizations have put into place. That means that, if a physician leaves an employer, he or she cannot reach out to former patients and colleagues to bring them along or invite them to switch to him or her in the new job.
Within that clause, however, the FTC has specified that if such nonsolicitation agreement has the “equivalent effect” of a noncompete, the agency would deem it such. That means, even if that rule stays, it could be contested and may be interpreted as violating the noncompete law. So there’s value in reading all the fine print should the ban move forward.
Could the ban bring potential downsides?
Most physicians view the potential to break free of a noncompete agreement as a victory. Peter Glennon, an employment litigation attorney with The Glennon Law Firm in Rochester, N.Y., says not so fast. “If you ask anyone if they’d prefer a noncompete agreement, of course they’re going to say no,” he said in an interview. “It sounds like a restriction, one that can hold you back.”
Mr. Glennon believes that there are actually upsides to physician noncompetes. For instance, many noncompetes come with sign-on bonuses that could potentially disappear without the agreements. There’s also the fact that when some physicians sign a noncompete agreement, they then receive pro bono training and continuing education along with marketing and promotion of their skills. Without signing a noncompete, employers may be less incentivized to provide all those benefits to their physician employers.
Those benefits – and the noncompetes – also vary by specialty, Mr. Glennon said. “In 2021, Washington, DC, banned noncompetes for doctors making less than $250,000. So, most generalists there can walk across the street and get a new job. For specialists like cardiologists or neurosurgeons, however, advanced training and marketing benefits matter, so many of them don’t want to lose noncompetes.”
Still, most physicians hope that the FTC’s ban takes hold. Manan Shah, MD, founder, and chief medical officer at Wyndly, an allergy relief startup practice, is one of them.
“Initially, it might disincentivize hospital systems from helping new physicians build up their name and practice because they might be concerned about a physician leaving and starting anew,” he said. “But in the long term, hospitals require physicians to bring their patients to them for care, so the best hospitals will always compete for the best physicians and support them as they build up their practice.”
Dr. Shah views noncompetes as overly prohibitive to physicians. “Right now, if a physician starts a job at a large hospital system and realizes they want to switch jobs, the noncompete distances are so wide they often have to move cities to continue practicing,” he said. “Picking up and starting over in a new city isn’t an option for everyone and can be especially difficult for someone with a family.”
Where Mr. Glennon argued that a physician leaving a team-based practice might harm patients, Shah takes a different perspective. “Imagine you have a doctor whom you trust and have been working with,” he said. “If something changes at their hospital and they decide to move, you literally have to find a new doctor instead of just being able to see them at another location down the street.”
Another potential burden of the noncompete agreements is that they could possibly squelch doctor’s desires to hang up their own shingle. According to Dr. Shah, the agreements make it so that if a physician wants to work independently, it’s nearly impossible to fly solo. “This is frustrating because independent practices have been shown to be more cost effective and allow patients to build better relationships with their doctors,” he claimed.
A 2016 study from Annals of Family Medicine supports that claim, at least for small general practices. Another study appearing in JAMA concurred. It does point out, however, that the cost equation is nuanced and that benefits of larger systems include more resilience to economic downturns and can provide more specialized care.
Will nonprofit hospitals be subject to this noncompete ban?
Further complicating the noncompete ban issue is how it might impact nonprofit institutions versus their for-profit peers. Most hospitals structured as nonprofits would be exempt from the rule because the FTC Act provides that it can enforce against “persons, partnerships, or corporations,” which are further defined as entities “organized to carry on business for their own profit or that of their members.”
The fallout from this, said Dr. Shah, is that it “would disproportionately affect health care providers, since many hospital systems are nonprofits. This is disconcerting because we know that many nonprofit systems make large profits anyway and can offer executive teams’ lucrative packages, while the nurses, assistants, and physicians providing the care are generally not well compensated.”
So far, about nine states plus Washington, D.C., have already put noncompete bans in place, and they may serve as a harbinger of things to come should the federal ban go into effect. Each varies in its specifics. Some, like Indiana, outright ban them, whereas others limit them based on variables like income and industry. “We’re seeing these states responding to local market conditions,” said Darryl Drevna, senior director of regulatory affairs at the American Medical Group Association. “Health care is a hyperlocal market. Depending on the situation, the bans adapt and respond specific to those states.”
Should the federal ban take hold, however, it will supersede whatever rules the individual states have in place.
Some opponents of the federal ban proposal question its authority to begin with, however, Mr. Glennon included. “Many people believe the FTC is overstepping,” he said. “Some people believe that Section 5 of the FTC Act does not give it the authority to police labor markets.”
Mr. Drevna noted that the FTC has taken an aggressive stance, one that will ultimately wind up in the courts. “How it works out is anyone’s guess,” he said. “Ideally, the FTC will consider the comments and concerns of groups like AMGA and realize that states are best suited to regulate in this area.”
In general, the ban’s supporters are employees/physicians; those who oppose it are their employers. Joining the AMGA in speaking out against the noncompete ban is the American Hospital Association, whereas the American College of Emergency Physicians has come out largely in support of the ban.
Still, doctors like Dr. Shah remain hopeful. “I am optimistic that perhaps my colleagues will not continue to be stuck in overrestrictive noncompetes, but I am also realistic,” he said. “Hospital systems are already coming out strongly against this and they have deep pockets, so I won’t be surprised if it does not come to pass.”
A version of this article first appeared on Medscape.com.
The Federal Trade Commission’s proposed regulation that would ban noncompete agreements across the country seems like potential good news for doctors. Of course, many hospitals and employers are against it. As a result, the FTC’s sweeping proposal has tongues wagging on both sides of the issue.
Many physicians are thrilled that they may soon have more control over their career and not be stuck in jobs where they feel frustrated, underpaid, or blocked in their progress.
As of 2018, as many as 45% of primary care physicians had inked such agreements with their employers.
Typically, the agreements prevent physicians from practicing medicine with a new employer for a defined period within a specific geographic area. No matter how attractive an alternate offer of employment might be, doctors are bound by the agreements to say no if the offer exists in that defined area and time period.
The period for public comment on the proposed regulation ended on April 19, and there is currently no set date for a decision.
In a Medscape poll of 558 physicians, more than 9 out of 10 respondents said that they were either currently bound by a noncompete clause or that they had been bound by one in the past that had forced them to temporarily stop working, commute long distances, move to a different area, or switch fields.
The new proposal would make it illegal for an employer, such as a hospital or large group, to enter a noncompete with a worker; maintain a noncompete with a worker; or represent to a worker, under certain circumstances, that the worker is subject to a noncompete.
It also would not only ban future noncompete agreements but also retroactively invalidate existing ones. The FTC reasons that noncompete clauses could potentially increase worker earnings as well as lower health care costs by billions of dollars. If the ruling were to move forward, it would represent part of President Biden’s “worker-forward” priorities, focusing on how competition can be a good thing for employees. The President billed the FTC’s announcement as a “huge win for workers.”
In its statements on the proposed ban, the FTC claimed that it could lower consumer prices across the board by as much as $150 billion per year and return nearly $300 million to workers each year.
However, even if passed, the draft rule would keep in place nonsolicitation rules that many health care organizations have put into place. That means that, if a physician leaves an employer, he or she cannot reach out to former patients and colleagues to bring them along or invite them to switch to him or her in the new job.
Within that clause, however, the FTC has specified that if such nonsolicitation agreement has the “equivalent effect” of a noncompete, the agency would deem it such. That means, even if that rule stays, it could be contested and may be interpreted as violating the noncompete law. So there’s value in reading all the fine print should the ban move forward.
Could the ban bring potential downsides?
Most physicians view the potential to break free of a noncompete agreement as a victory. Peter Glennon, an employment litigation attorney with The Glennon Law Firm in Rochester, N.Y., says not so fast. “If you ask anyone if they’d prefer a noncompete agreement, of course they’re going to say no,” he said in an interview. “It sounds like a restriction, one that can hold you back.”
Mr. Glennon believes that there are actually upsides to physician noncompetes. For instance, many noncompetes come with sign-on bonuses that could potentially disappear without the agreements. There’s also the fact that when some physicians sign a noncompete agreement, they then receive pro bono training and continuing education along with marketing and promotion of their skills. Without signing a noncompete, employers may be less incentivized to provide all those benefits to their physician employers.
Those benefits – and the noncompetes – also vary by specialty, Mr. Glennon said. “In 2021, Washington, DC, banned noncompetes for doctors making less than $250,000. So, most generalists there can walk across the street and get a new job. For specialists like cardiologists or neurosurgeons, however, advanced training and marketing benefits matter, so many of them don’t want to lose noncompetes.”
Still, most physicians hope that the FTC’s ban takes hold. Manan Shah, MD, founder, and chief medical officer at Wyndly, an allergy relief startup practice, is one of them.
“Initially, it might disincentivize hospital systems from helping new physicians build up their name and practice because they might be concerned about a physician leaving and starting anew,” he said. “But in the long term, hospitals require physicians to bring their patients to them for care, so the best hospitals will always compete for the best physicians and support them as they build up their practice.”
Dr. Shah views noncompetes as overly prohibitive to physicians. “Right now, if a physician starts a job at a large hospital system and realizes they want to switch jobs, the noncompete distances are so wide they often have to move cities to continue practicing,” he said. “Picking up and starting over in a new city isn’t an option for everyone and can be especially difficult for someone with a family.”
Where Mr. Glennon argued that a physician leaving a team-based practice might harm patients, Shah takes a different perspective. “Imagine you have a doctor whom you trust and have been working with,” he said. “If something changes at their hospital and they decide to move, you literally have to find a new doctor instead of just being able to see them at another location down the street.”
Another potential burden of the noncompete agreements is that they could possibly squelch doctor’s desires to hang up their own shingle. According to Dr. Shah, the agreements make it so that if a physician wants to work independently, it’s nearly impossible to fly solo. “This is frustrating because independent practices have been shown to be more cost effective and allow patients to build better relationships with their doctors,” he claimed.
A 2016 study from Annals of Family Medicine supports that claim, at least for small general practices. Another study appearing in JAMA concurred. It does point out, however, that the cost equation is nuanced and that benefits of larger systems include more resilience to economic downturns and can provide more specialized care.
Will nonprofit hospitals be subject to this noncompete ban?
Further complicating the noncompete ban issue is how it might impact nonprofit institutions versus their for-profit peers. Most hospitals structured as nonprofits would be exempt from the rule because the FTC Act provides that it can enforce against “persons, partnerships, or corporations,” which are further defined as entities “organized to carry on business for their own profit or that of their members.”
The fallout from this, said Dr. Shah, is that it “would disproportionately affect health care providers, since many hospital systems are nonprofits. This is disconcerting because we know that many nonprofit systems make large profits anyway and can offer executive teams’ lucrative packages, while the nurses, assistants, and physicians providing the care are generally not well compensated.”
So far, about nine states plus Washington, D.C., have already put noncompete bans in place, and they may serve as a harbinger of things to come should the federal ban go into effect. Each varies in its specifics. Some, like Indiana, outright ban them, whereas others limit them based on variables like income and industry. “We’re seeing these states responding to local market conditions,” said Darryl Drevna, senior director of regulatory affairs at the American Medical Group Association. “Health care is a hyperlocal market. Depending on the situation, the bans adapt and respond specific to those states.”
Should the federal ban take hold, however, it will supersede whatever rules the individual states have in place.
Some opponents of the federal ban proposal question its authority to begin with, however, Mr. Glennon included. “Many people believe the FTC is overstepping,” he said. “Some people believe that Section 5 of the FTC Act does not give it the authority to police labor markets.”
Mr. Drevna noted that the FTC has taken an aggressive stance, one that will ultimately wind up in the courts. “How it works out is anyone’s guess,” he said. “Ideally, the FTC will consider the comments and concerns of groups like AMGA and realize that states are best suited to regulate in this area.”
In general, the ban’s supporters are employees/physicians; those who oppose it are their employers. Joining the AMGA in speaking out against the noncompete ban is the American Hospital Association, whereas the American College of Emergency Physicians has come out largely in support of the ban.
Still, doctors like Dr. Shah remain hopeful. “I am optimistic that perhaps my colleagues will not continue to be stuck in overrestrictive noncompetes, but I am also realistic,” he said. “Hospital systems are already coming out strongly against this and they have deep pockets, so I won’t be surprised if it does not come to pass.”
A version of this article first appeared on Medscape.com.
The Federal Trade Commission’s proposed regulation that would ban noncompete agreements across the country seems like potential good news for doctors. Of course, many hospitals and employers are against it. As a result, the FTC’s sweeping proposal has tongues wagging on both sides of the issue.
Many physicians are thrilled that they may soon have more control over their career and not be stuck in jobs where they feel frustrated, underpaid, or blocked in their progress.
As of 2018, as many as 45% of primary care physicians had inked such agreements with their employers.
Typically, the agreements prevent physicians from practicing medicine with a new employer for a defined period within a specific geographic area. No matter how attractive an alternate offer of employment might be, doctors are bound by the agreements to say no if the offer exists in that defined area and time period.
The period for public comment on the proposed regulation ended on April 19, and there is currently no set date for a decision.
In a Medscape poll of 558 physicians, more than 9 out of 10 respondents said that they were either currently bound by a noncompete clause or that they had been bound by one in the past that had forced them to temporarily stop working, commute long distances, move to a different area, or switch fields.
The new proposal would make it illegal for an employer, such as a hospital or large group, to enter a noncompete with a worker; maintain a noncompete with a worker; or represent to a worker, under certain circumstances, that the worker is subject to a noncompete.
It also would not only ban future noncompete agreements but also retroactively invalidate existing ones. The FTC reasons that noncompete clauses could potentially increase worker earnings as well as lower health care costs by billions of dollars. If the ruling were to move forward, it would represent part of President Biden’s “worker-forward” priorities, focusing on how competition can be a good thing for employees. The President billed the FTC’s announcement as a “huge win for workers.”
In its statements on the proposed ban, the FTC claimed that it could lower consumer prices across the board by as much as $150 billion per year and return nearly $300 million to workers each year.
However, even if passed, the draft rule would keep in place nonsolicitation rules that many health care organizations have put into place. That means that, if a physician leaves an employer, he or she cannot reach out to former patients and colleagues to bring them along or invite them to switch to him or her in the new job.
Within that clause, however, the FTC has specified that if such nonsolicitation agreement has the “equivalent effect” of a noncompete, the agency would deem it such. That means, even if that rule stays, it could be contested and may be interpreted as violating the noncompete law. So there’s value in reading all the fine print should the ban move forward.
Could the ban bring potential downsides?
Most physicians view the potential to break free of a noncompete agreement as a victory. Peter Glennon, an employment litigation attorney with The Glennon Law Firm in Rochester, N.Y., says not so fast. “If you ask anyone if they’d prefer a noncompete agreement, of course they’re going to say no,” he said in an interview. “It sounds like a restriction, one that can hold you back.”
Mr. Glennon believes that there are actually upsides to physician noncompetes. For instance, many noncompetes come with sign-on bonuses that could potentially disappear without the agreements. There’s also the fact that when some physicians sign a noncompete agreement, they then receive pro bono training and continuing education along with marketing and promotion of their skills. Without signing a noncompete, employers may be less incentivized to provide all those benefits to their physician employers.
Those benefits – and the noncompetes – also vary by specialty, Mr. Glennon said. “In 2021, Washington, DC, banned noncompetes for doctors making less than $250,000. So, most generalists there can walk across the street and get a new job. For specialists like cardiologists or neurosurgeons, however, advanced training and marketing benefits matter, so many of them don’t want to lose noncompetes.”
Still, most physicians hope that the FTC’s ban takes hold. Manan Shah, MD, founder, and chief medical officer at Wyndly, an allergy relief startup practice, is one of them.
“Initially, it might disincentivize hospital systems from helping new physicians build up their name and practice because they might be concerned about a physician leaving and starting anew,” he said. “But in the long term, hospitals require physicians to bring their patients to them for care, so the best hospitals will always compete for the best physicians and support them as they build up their practice.”
Dr. Shah views noncompetes as overly prohibitive to physicians. “Right now, if a physician starts a job at a large hospital system and realizes they want to switch jobs, the noncompete distances are so wide they often have to move cities to continue practicing,” he said. “Picking up and starting over in a new city isn’t an option for everyone and can be especially difficult for someone with a family.”
Where Mr. Glennon argued that a physician leaving a team-based practice might harm patients, Shah takes a different perspective. “Imagine you have a doctor whom you trust and have been working with,” he said. “If something changes at their hospital and they decide to move, you literally have to find a new doctor instead of just being able to see them at another location down the street.”
Another potential burden of the noncompete agreements is that they could possibly squelch doctor’s desires to hang up their own shingle. According to Dr. Shah, the agreements make it so that if a physician wants to work independently, it’s nearly impossible to fly solo. “This is frustrating because independent practices have been shown to be more cost effective and allow patients to build better relationships with their doctors,” he claimed.
A 2016 study from Annals of Family Medicine supports that claim, at least for small general practices. Another study appearing in JAMA concurred. It does point out, however, that the cost equation is nuanced and that benefits of larger systems include more resilience to economic downturns and can provide more specialized care.
Will nonprofit hospitals be subject to this noncompete ban?
Further complicating the noncompete ban issue is how it might impact nonprofit institutions versus their for-profit peers. Most hospitals structured as nonprofits would be exempt from the rule because the FTC Act provides that it can enforce against “persons, partnerships, or corporations,” which are further defined as entities “organized to carry on business for their own profit or that of their members.”
The fallout from this, said Dr. Shah, is that it “would disproportionately affect health care providers, since many hospital systems are nonprofits. This is disconcerting because we know that many nonprofit systems make large profits anyway and can offer executive teams’ lucrative packages, while the nurses, assistants, and physicians providing the care are generally not well compensated.”
So far, about nine states plus Washington, D.C., have already put noncompete bans in place, and they may serve as a harbinger of things to come should the federal ban go into effect. Each varies in its specifics. Some, like Indiana, outright ban them, whereas others limit them based on variables like income and industry. “We’re seeing these states responding to local market conditions,” said Darryl Drevna, senior director of regulatory affairs at the American Medical Group Association. “Health care is a hyperlocal market. Depending on the situation, the bans adapt and respond specific to those states.”
Should the federal ban take hold, however, it will supersede whatever rules the individual states have in place.
Some opponents of the federal ban proposal question its authority to begin with, however, Mr. Glennon included. “Many people believe the FTC is overstepping,” he said. “Some people believe that Section 5 of the FTC Act does not give it the authority to police labor markets.”
Mr. Drevna noted that the FTC has taken an aggressive stance, one that will ultimately wind up in the courts. “How it works out is anyone’s guess,” he said. “Ideally, the FTC will consider the comments and concerns of groups like AMGA and realize that states are best suited to regulate in this area.”
In general, the ban’s supporters are employees/physicians; those who oppose it are their employers. Joining the AMGA in speaking out against the noncompete ban is the American Hospital Association, whereas the American College of Emergency Physicians has come out largely in support of the ban.
Still, doctors like Dr. Shah remain hopeful. “I am optimistic that perhaps my colleagues will not continue to be stuck in overrestrictive noncompetes, but I am also realistic,” he said. “Hospital systems are already coming out strongly against this and they have deep pockets, so I won’t be surprised if it does not come to pass.”
A version of this article first appeared on Medscape.com.
How BMI over time impacts GI cancer risk
according to new data from the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial.
The researchers found that being overweight or obese in early and middle adulthood was associated with an increased risk for colorectal cancer (CRC) and noncolorectal GI cancers. Maintaining or increasing BMI over time among overweight or obese individuals was also associated with an increased GI cancer risk.
Aspirin use did not significantly modify these associations, suggesting that aspirin may not be as effective for cancer prevention among overweight or obese individuals.
The results provide “relatively consistent messaging that overweight or obesity from early to later adulthood as well as BMI increases throughout adulthood were associated with increased risk of GI cancers, especially CRC,” the authors of an editorial accompanying the study wrote.
These “important findings highlight the unmet need to identify the critical time window linking adiposity and GI cancer,” said editorialists Mengyao Shi, MBBS, MPD, and Yin Cao, ScD, MPH, of Washington University in St. Louis.
The analysis was published online in JAMA Network Open.
A growing body of evidence has revealed a strong association between obesity and GI cancers, with chronic inflammation being a likely cause. As rates of overweight and obesity continue to grow, better understanding of the association between obesity and cancer has become increasingly important.
In the analysis, Holli A. Loomans-Kropp, PhD, MPH, with Ohio State University, Columbus, and Asad Umar, PhD, DVM, with the National Cancer Institute, Rockville, Md., explored associations between BMI in early adulthood (age 20), middle adulthood (age 50) and later adulthood (age 55 and over) and GI cancer risk in 135,161 adults from the PLCO Cancer Screening Trial.
BMI was determined using self-reported height and weight at each age time point. The median age at enrollment was 62 years, and 50% of participants were women. Overweight BMI was 25.0-29.9 kg/m2 and obese BMI was 30 or higher.
During as many as 21 years of follow-up, 2,803 individuals developed CRC and 2,285 developed non-CRC GI cancers (esophageal, liver, gastric, and pancreatic).
Overweight BMI in early, middle, and later adulthood was associated with an increased risk of CRC (hazard ratio, 1.23 for early and middle years; HR, 1.21 for later years). Obese BMI in middle and later adulthood was also associated with increased risk of CRC (HR, 1.55 and 1.39, respectively).
The authors observed similar associations between BMI in middle and later adulthood and overall GI and non-CRC GI risk.
“When modeled continuously, we observed 2%-4% increased risk of both CRC and noncolorectal GI cancer with each 1-unit increase in BMI across all time points,” the researchers said.
Their data also suggest that BMI over time may be associated with GI cancer risk. Adults who exhibited no change in overweight or obese BMIs between early and later adulthood and those who exhibited increases in BMI from underweight or normal in early adulthood to overweight or obese BMI in later adulthood had a significantly higher risk for CRC and noncolorectal GI cancer.
Among frequent aspirin users, those with overweight or obese BMIs in early, middle, and later adulthood still had an increased risk for CRC and noncolorectal GI cancer (hazard ratios, 1.44, 1.45, and 1.43, respectively).
The finding that regular weekly aspirin use did not modify GI cancer risk suggests that obesity may alter the cancer-preventive effect of aspirin, the researchers suggested. Individuals with obesity may need to increase aspirin frequency or dosage to see an effect, but upping the dose comes with its own risks, including GI bleeding.
Overall, until now, most epidemiologic studies have examined BMI at one time point, “missing the opportunity to delineate the contribution of adiposity throughout the life course,” Dr. Shi and Dr. Cao wrote.
“As we continue to investigate precision-based interventions to intercept the link between obesity and cancer, it is imperative to reiterate the importance of maintaining a healthy weight and lifestyle from an early age and incorporate it widely into cancer prevention strategies at all levels with immediate implementation,” the editorialists concluded.
This study was supported in part by funds from Ohio State University and the NIH. The study authors reported no relevant financial relationships. Dr. Cao has received personal fees from Geneoscopy for consulting.
A version of this article originally appeared on Medscape.com.
according to new data from the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial.
The researchers found that being overweight or obese in early and middle adulthood was associated with an increased risk for colorectal cancer (CRC) and noncolorectal GI cancers. Maintaining or increasing BMI over time among overweight or obese individuals was also associated with an increased GI cancer risk.
Aspirin use did not significantly modify these associations, suggesting that aspirin may not be as effective for cancer prevention among overweight or obese individuals.
The results provide “relatively consistent messaging that overweight or obesity from early to later adulthood as well as BMI increases throughout adulthood were associated with increased risk of GI cancers, especially CRC,” the authors of an editorial accompanying the study wrote.
These “important findings highlight the unmet need to identify the critical time window linking adiposity and GI cancer,” said editorialists Mengyao Shi, MBBS, MPD, and Yin Cao, ScD, MPH, of Washington University in St. Louis.
The analysis was published online in JAMA Network Open.
A growing body of evidence has revealed a strong association between obesity and GI cancers, with chronic inflammation being a likely cause. As rates of overweight and obesity continue to grow, better understanding of the association between obesity and cancer has become increasingly important.
In the analysis, Holli A. Loomans-Kropp, PhD, MPH, with Ohio State University, Columbus, and Asad Umar, PhD, DVM, with the National Cancer Institute, Rockville, Md., explored associations between BMI in early adulthood (age 20), middle adulthood (age 50) and later adulthood (age 55 and over) and GI cancer risk in 135,161 adults from the PLCO Cancer Screening Trial.
BMI was determined using self-reported height and weight at each age time point. The median age at enrollment was 62 years, and 50% of participants were women. Overweight BMI was 25.0-29.9 kg/m2 and obese BMI was 30 or higher.
During as many as 21 years of follow-up, 2,803 individuals developed CRC and 2,285 developed non-CRC GI cancers (esophageal, liver, gastric, and pancreatic).
Overweight BMI in early, middle, and later adulthood was associated with an increased risk of CRC (hazard ratio, 1.23 for early and middle years; HR, 1.21 for later years). Obese BMI in middle and later adulthood was also associated with increased risk of CRC (HR, 1.55 and 1.39, respectively).
The authors observed similar associations between BMI in middle and later adulthood and overall GI and non-CRC GI risk.
“When modeled continuously, we observed 2%-4% increased risk of both CRC and noncolorectal GI cancer with each 1-unit increase in BMI across all time points,” the researchers said.
Their data also suggest that BMI over time may be associated with GI cancer risk. Adults who exhibited no change in overweight or obese BMIs between early and later adulthood and those who exhibited increases in BMI from underweight or normal in early adulthood to overweight or obese BMI in later adulthood had a significantly higher risk for CRC and noncolorectal GI cancer.
Among frequent aspirin users, those with overweight or obese BMIs in early, middle, and later adulthood still had an increased risk for CRC and noncolorectal GI cancer (hazard ratios, 1.44, 1.45, and 1.43, respectively).
The finding that regular weekly aspirin use did not modify GI cancer risk suggests that obesity may alter the cancer-preventive effect of aspirin, the researchers suggested. Individuals with obesity may need to increase aspirin frequency or dosage to see an effect, but upping the dose comes with its own risks, including GI bleeding.
Overall, until now, most epidemiologic studies have examined BMI at one time point, “missing the opportunity to delineate the contribution of adiposity throughout the life course,” Dr. Shi and Dr. Cao wrote.
“As we continue to investigate precision-based interventions to intercept the link between obesity and cancer, it is imperative to reiterate the importance of maintaining a healthy weight and lifestyle from an early age and incorporate it widely into cancer prevention strategies at all levels with immediate implementation,” the editorialists concluded.
This study was supported in part by funds from Ohio State University and the NIH. The study authors reported no relevant financial relationships. Dr. Cao has received personal fees from Geneoscopy for consulting.
A version of this article originally appeared on Medscape.com.
according to new data from the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial.
The researchers found that being overweight or obese in early and middle adulthood was associated with an increased risk for colorectal cancer (CRC) and noncolorectal GI cancers. Maintaining or increasing BMI over time among overweight or obese individuals was also associated with an increased GI cancer risk.
Aspirin use did not significantly modify these associations, suggesting that aspirin may not be as effective for cancer prevention among overweight or obese individuals.
The results provide “relatively consistent messaging that overweight or obesity from early to later adulthood as well as BMI increases throughout adulthood were associated with increased risk of GI cancers, especially CRC,” the authors of an editorial accompanying the study wrote.
These “important findings highlight the unmet need to identify the critical time window linking adiposity and GI cancer,” said editorialists Mengyao Shi, MBBS, MPD, and Yin Cao, ScD, MPH, of Washington University in St. Louis.
The analysis was published online in JAMA Network Open.
A growing body of evidence has revealed a strong association between obesity and GI cancers, with chronic inflammation being a likely cause. As rates of overweight and obesity continue to grow, better understanding of the association between obesity and cancer has become increasingly important.
In the analysis, Holli A. Loomans-Kropp, PhD, MPH, with Ohio State University, Columbus, and Asad Umar, PhD, DVM, with the National Cancer Institute, Rockville, Md., explored associations between BMI in early adulthood (age 20), middle adulthood (age 50) and later adulthood (age 55 and over) and GI cancer risk in 135,161 adults from the PLCO Cancer Screening Trial.
BMI was determined using self-reported height and weight at each age time point. The median age at enrollment was 62 years, and 50% of participants were women. Overweight BMI was 25.0-29.9 kg/m2 and obese BMI was 30 or higher.
During as many as 21 years of follow-up, 2,803 individuals developed CRC and 2,285 developed non-CRC GI cancers (esophageal, liver, gastric, and pancreatic).
Overweight BMI in early, middle, and later adulthood was associated with an increased risk of CRC (hazard ratio, 1.23 for early and middle years; HR, 1.21 for later years). Obese BMI in middle and later adulthood was also associated with increased risk of CRC (HR, 1.55 and 1.39, respectively).
The authors observed similar associations between BMI in middle and later adulthood and overall GI and non-CRC GI risk.
“When modeled continuously, we observed 2%-4% increased risk of both CRC and noncolorectal GI cancer with each 1-unit increase in BMI across all time points,” the researchers said.
Their data also suggest that BMI over time may be associated with GI cancer risk. Adults who exhibited no change in overweight or obese BMIs between early and later adulthood and those who exhibited increases in BMI from underweight or normal in early adulthood to overweight or obese BMI in later adulthood had a significantly higher risk for CRC and noncolorectal GI cancer.
Among frequent aspirin users, those with overweight or obese BMIs in early, middle, and later adulthood still had an increased risk for CRC and noncolorectal GI cancer (hazard ratios, 1.44, 1.45, and 1.43, respectively).
The finding that regular weekly aspirin use did not modify GI cancer risk suggests that obesity may alter the cancer-preventive effect of aspirin, the researchers suggested. Individuals with obesity may need to increase aspirin frequency or dosage to see an effect, but upping the dose comes with its own risks, including GI bleeding.
Overall, until now, most epidemiologic studies have examined BMI at one time point, “missing the opportunity to delineate the contribution of adiposity throughout the life course,” Dr. Shi and Dr. Cao wrote.
“As we continue to investigate precision-based interventions to intercept the link between obesity and cancer, it is imperative to reiterate the importance of maintaining a healthy weight and lifestyle from an early age and incorporate it widely into cancer prevention strategies at all levels with immediate implementation,” the editorialists concluded.
This study was supported in part by funds from Ohio State University and the NIH. The study authors reported no relevant financial relationships. Dr. Cao has received personal fees from Geneoscopy for consulting.
A version of this article originally appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Could vitamin D supplementation help in long COVID?
, in a retrospective, case-matched study.
The lower levels of vitamin D in patients with long COVID were most notable in those with brain fog.
These findings, by Luigi di Filippo, MD, and colleagues, were recently presented at the European Congress of Endocrinology and published in the Journal of Clinical Endocrinology & Metabolism.
“Our data suggest that vitamin D levels should be evaluated in COVID-19 patients after hospital discharge,” wrote the researchers, from San Raffaele Hospital, Milan.
“The role of vitamin D supplementation as a preventive strategy of COVID-19 sequelae should be tested in randomized controlled trials,” they urged.
The researchers also stressed that this was a controlled study in a homogeneous population, it included multiple signs and symptoms of long COVID, and it had a longer follow-up than most previous studies (6 vs. 3 months).
“The highly controlled nature of our study helps us better understand the role of vitamin D deficiency in long COVID and establish that there is likely a link between vitamin D deficiency and long COVID,” senior author Andrea Giustina, MD, said in a press release from the ECE.
“Our study shows that COVID-19 patients with low vitamin D levels are more likely to develop long COVID, but it is not yet known whether vitamin D supplements could improve the symptoms or reduce this risk altogether,” he cautioned.
“If confirmed in large, interventional, randomized controlled trials, [our data suggest] that vitamin D supplementation could represent a possible preventive strategy in reducing the burden of COVID-19 sequelae,” Dr. Giustina and colleagues wrote.
Reasonable to test vitamin D levels, consider supplementation
Invited to comment, Amiel Dror, MD, PhD, who led a related study that showed that people with a vitamin D deficiency were more likely to have severe COVID-19, agreed.
“The novelty and significance of this [new] study lie in the fact that it expands on our current understanding of the interplay between vitamin D and COVID-19, taking it beyond the acute phase of the disease,” said Dr. Dror, from Bar-Ilan University, Safed, Israel.
“It’s striking to see how vitamin D levels continue to influence patients’ health even after recovery from the initial infection,” he noted.
“The findings certainly add weight to the argument for conducting a randomized control trial [RCT],” he continued, which “would enable us to conclusively determine whether vitamin D supplementation can effectively reduce the risk or severity of long COVID.”
“In the interim,” Dr. Dror said, “given the safety profile of vitamin D and its broad health benefits, it could be reasonable to test for vitamin D levels in patients admitted with COVID-19. If levels are found to be low, supplementation could be considered.”
“However, it’s important to note that this should be done under medical supervision,” he cautioned, “and further studies are needed to establish the optimal timing and dosage of supplementation.”
“I anticipate that we’ll see more RCTs [of this] in the future,” he speculated.
Low vitamin D and risk of long COVID
Long COVID is an emerging syndrome that affects 50%-70% of COVID-19 survivors.
Low levels of vitamin D have been associated with increased likelihood of needing mechanical ventilation and worse survival in patients hospitalized with COVID-19, but the risk of long COVID associated with vitamin D has not been known.
Researchers analyzed data from adults aged 18 and older hospitalized at San Raffaele Hospital with a confirmed diagnosis of COVID-19 and discharged during the first pandemic wave, from March to May 2020, and then seen 6-months later for follow-up.
Patients were excluded if they had been admitted to the intensive care unit during hospitalization or had missing medical data or blood samples available to determine (OH) vitamin D levels, at admission and the 6-month follow-up.
Long COVID-19 was defined based on the U.K. National Institute for Health and Care Excellence guidelines as the concomitant presence of at least two or more of 17 signs and symptoms that were absent prior to the COVID-19 infection and could only be attributed to that acute disease.
Researchers identified 50 patients with long COVID at the 6-month follow-up and matched them with 50 patients without long COVID at that time point, based on age, sex, concomitant comorbidities, need for noninvasive mechanical ventilation, and week of evaluation.
Patients were a mean age of 61 years (range, 51-73) and 56% were men; 28% had been on a ventilator during hospitalization for COVID-19.
The most frequent signs and symptoms at 6 months in the patients with long COVID were asthenia (weakness, 38% of patients), dysgeusia (bad taste in the mouth, 34%), dyspnea (shortness of breath, 34%), and anosmia (loss of sense of smell, 24%).
Most symptoms were related to the cardiorespiratory system (42%), the feeling of well-being (42%), or the senses (36%), and fewer patients had symptoms related to neurocognitive impairment (headache or brain fog, 14%), or ear, nose, and throat (12%), or gastrointestinal system (4%).
Patients with long COVID had lower mean 25(OH) vitamin D levels than patients without long COVID (20.1 vs 23.2 ng/mL; P = .03). However, actual vitamin D deficiency levels were similar in both groups.
Two-thirds of patients with low vitamin D levels at hospital admission still presented with low levels at the 6-month follow-up.
Vitamin D levels were significantly lower in patients with neurocognitive symptoms at follow-up (n = 7) than in those without such symptoms (n = 93) (14.6 vs. 20.6 ng/mL; P = .042).
In patients with vitamin D deficiency (< 20 ng/mL) at admission and at follow-up (n = 42), those with long COVID (n = 22) had lower vitamin D levels at follow-up than those without long COVID (n = 20) (12.7 vs. 15.2 ng/mL; P = .041).
And in multiple regression analyses, a lower 25(OH) vitamin D level at follow-up was the only variable that was significantly associated with long COVID (odds ratio, 1.09; 95% confidence interval, 1.01-1.16; P = .008).
The findings “strongly reinforce the clinical usefulness of 25(OH) vitamin D evaluation as a possible modifiable pathophysiological factor underlying this emerging worldwide critical health issue,” the researchers concluded.
The study was supported by Abiogen Pharma. One study author is an employee at Abiogen. Dr. Giustina has reported being a consultant for Abiogen and Takeda and receiving a research grant to his institution from Takeda. Dr. Di Filippo and the other authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, in a retrospective, case-matched study.
The lower levels of vitamin D in patients with long COVID were most notable in those with brain fog.
These findings, by Luigi di Filippo, MD, and colleagues, were recently presented at the European Congress of Endocrinology and published in the Journal of Clinical Endocrinology & Metabolism.
“Our data suggest that vitamin D levels should be evaluated in COVID-19 patients after hospital discharge,” wrote the researchers, from San Raffaele Hospital, Milan.
“The role of vitamin D supplementation as a preventive strategy of COVID-19 sequelae should be tested in randomized controlled trials,” they urged.
The researchers also stressed that this was a controlled study in a homogeneous population, it included multiple signs and symptoms of long COVID, and it had a longer follow-up than most previous studies (6 vs. 3 months).
“The highly controlled nature of our study helps us better understand the role of vitamin D deficiency in long COVID and establish that there is likely a link between vitamin D deficiency and long COVID,” senior author Andrea Giustina, MD, said in a press release from the ECE.
“Our study shows that COVID-19 patients with low vitamin D levels are more likely to develop long COVID, but it is not yet known whether vitamin D supplements could improve the symptoms or reduce this risk altogether,” he cautioned.
“If confirmed in large, interventional, randomized controlled trials, [our data suggest] that vitamin D supplementation could represent a possible preventive strategy in reducing the burden of COVID-19 sequelae,” Dr. Giustina and colleagues wrote.
Reasonable to test vitamin D levels, consider supplementation
Invited to comment, Amiel Dror, MD, PhD, who led a related study that showed that people with a vitamin D deficiency were more likely to have severe COVID-19, agreed.
“The novelty and significance of this [new] study lie in the fact that it expands on our current understanding of the interplay between vitamin D and COVID-19, taking it beyond the acute phase of the disease,” said Dr. Dror, from Bar-Ilan University, Safed, Israel.
“It’s striking to see how vitamin D levels continue to influence patients’ health even after recovery from the initial infection,” he noted.
“The findings certainly add weight to the argument for conducting a randomized control trial [RCT],” he continued, which “would enable us to conclusively determine whether vitamin D supplementation can effectively reduce the risk or severity of long COVID.”
“In the interim,” Dr. Dror said, “given the safety profile of vitamin D and its broad health benefits, it could be reasonable to test for vitamin D levels in patients admitted with COVID-19. If levels are found to be low, supplementation could be considered.”
“However, it’s important to note that this should be done under medical supervision,” he cautioned, “and further studies are needed to establish the optimal timing and dosage of supplementation.”
“I anticipate that we’ll see more RCTs [of this] in the future,” he speculated.
Low vitamin D and risk of long COVID
Long COVID is an emerging syndrome that affects 50%-70% of COVID-19 survivors.
Low levels of vitamin D have been associated with increased likelihood of needing mechanical ventilation and worse survival in patients hospitalized with COVID-19, but the risk of long COVID associated with vitamin D has not been known.
Researchers analyzed data from adults aged 18 and older hospitalized at San Raffaele Hospital with a confirmed diagnosis of COVID-19 and discharged during the first pandemic wave, from March to May 2020, and then seen 6-months later for follow-up.
Patients were excluded if they had been admitted to the intensive care unit during hospitalization or had missing medical data or blood samples available to determine (OH) vitamin D levels, at admission and the 6-month follow-up.
Long COVID-19 was defined based on the U.K. National Institute for Health and Care Excellence guidelines as the concomitant presence of at least two or more of 17 signs and symptoms that were absent prior to the COVID-19 infection and could only be attributed to that acute disease.
Researchers identified 50 patients with long COVID at the 6-month follow-up and matched them with 50 patients without long COVID at that time point, based on age, sex, concomitant comorbidities, need for noninvasive mechanical ventilation, and week of evaluation.
Patients were a mean age of 61 years (range, 51-73) and 56% were men; 28% had been on a ventilator during hospitalization for COVID-19.
The most frequent signs and symptoms at 6 months in the patients with long COVID were asthenia (weakness, 38% of patients), dysgeusia (bad taste in the mouth, 34%), dyspnea (shortness of breath, 34%), and anosmia (loss of sense of smell, 24%).
Most symptoms were related to the cardiorespiratory system (42%), the feeling of well-being (42%), or the senses (36%), and fewer patients had symptoms related to neurocognitive impairment (headache or brain fog, 14%), or ear, nose, and throat (12%), or gastrointestinal system (4%).
Patients with long COVID had lower mean 25(OH) vitamin D levels than patients without long COVID (20.1 vs 23.2 ng/mL; P = .03). However, actual vitamin D deficiency levels were similar in both groups.
Two-thirds of patients with low vitamin D levels at hospital admission still presented with low levels at the 6-month follow-up.
Vitamin D levels were significantly lower in patients with neurocognitive symptoms at follow-up (n = 7) than in those without such symptoms (n = 93) (14.6 vs. 20.6 ng/mL; P = .042).
In patients with vitamin D deficiency (< 20 ng/mL) at admission and at follow-up (n = 42), those with long COVID (n = 22) had lower vitamin D levels at follow-up than those without long COVID (n = 20) (12.7 vs. 15.2 ng/mL; P = .041).
And in multiple regression analyses, a lower 25(OH) vitamin D level at follow-up was the only variable that was significantly associated with long COVID (odds ratio, 1.09; 95% confidence interval, 1.01-1.16; P = .008).
The findings “strongly reinforce the clinical usefulness of 25(OH) vitamin D evaluation as a possible modifiable pathophysiological factor underlying this emerging worldwide critical health issue,” the researchers concluded.
The study was supported by Abiogen Pharma. One study author is an employee at Abiogen. Dr. Giustina has reported being a consultant for Abiogen and Takeda and receiving a research grant to his institution from Takeda. Dr. Di Filippo and the other authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, in a retrospective, case-matched study.
The lower levels of vitamin D in patients with long COVID were most notable in those with brain fog.
These findings, by Luigi di Filippo, MD, and colleagues, were recently presented at the European Congress of Endocrinology and published in the Journal of Clinical Endocrinology & Metabolism.
“Our data suggest that vitamin D levels should be evaluated in COVID-19 patients after hospital discharge,” wrote the researchers, from San Raffaele Hospital, Milan.
“The role of vitamin D supplementation as a preventive strategy of COVID-19 sequelae should be tested in randomized controlled trials,” they urged.
The researchers also stressed that this was a controlled study in a homogeneous population, it included multiple signs and symptoms of long COVID, and it had a longer follow-up than most previous studies (6 vs. 3 months).
“The highly controlled nature of our study helps us better understand the role of vitamin D deficiency in long COVID and establish that there is likely a link between vitamin D deficiency and long COVID,” senior author Andrea Giustina, MD, said in a press release from the ECE.
“Our study shows that COVID-19 patients with low vitamin D levels are more likely to develop long COVID, but it is not yet known whether vitamin D supplements could improve the symptoms or reduce this risk altogether,” he cautioned.
“If confirmed in large, interventional, randomized controlled trials, [our data suggest] that vitamin D supplementation could represent a possible preventive strategy in reducing the burden of COVID-19 sequelae,” Dr. Giustina and colleagues wrote.
Reasonable to test vitamin D levels, consider supplementation
Invited to comment, Amiel Dror, MD, PhD, who led a related study that showed that people with a vitamin D deficiency were more likely to have severe COVID-19, agreed.
“The novelty and significance of this [new] study lie in the fact that it expands on our current understanding of the interplay between vitamin D and COVID-19, taking it beyond the acute phase of the disease,” said Dr. Dror, from Bar-Ilan University, Safed, Israel.
“It’s striking to see how vitamin D levels continue to influence patients’ health even after recovery from the initial infection,” he noted.
“The findings certainly add weight to the argument for conducting a randomized control trial [RCT],” he continued, which “would enable us to conclusively determine whether vitamin D supplementation can effectively reduce the risk or severity of long COVID.”
“In the interim,” Dr. Dror said, “given the safety profile of vitamin D and its broad health benefits, it could be reasonable to test for vitamin D levels in patients admitted with COVID-19. If levels are found to be low, supplementation could be considered.”
“However, it’s important to note that this should be done under medical supervision,” he cautioned, “and further studies are needed to establish the optimal timing and dosage of supplementation.”
“I anticipate that we’ll see more RCTs [of this] in the future,” he speculated.
Low vitamin D and risk of long COVID
Long COVID is an emerging syndrome that affects 50%-70% of COVID-19 survivors.
Low levels of vitamin D have been associated with increased likelihood of needing mechanical ventilation and worse survival in patients hospitalized with COVID-19, but the risk of long COVID associated with vitamin D has not been known.
Researchers analyzed data from adults aged 18 and older hospitalized at San Raffaele Hospital with a confirmed diagnosis of COVID-19 and discharged during the first pandemic wave, from March to May 2020, and then seen 6-months later for follow-up.
Patients were excluded if they had been admitted to the intensive care unit during hospitalization or had missing medical data or blood samples available to determine (OH) vitamin D levels, at admission and the 6-month follow-up.
Long COVID-19 was defined based on the U.K. National Institute for Health and Care Excellence guidelines as the concomitant presence of at least two or more of 17 signs and symptoms that were absent prior to the COVID-19 infection and could only be attributed to that acute disease.
Researchers identified 50 patients with long COVID at the 6-month follow-up and matched them with 50 patients without long COVID at that time point, based on age, sex, concomitant comorbidities, need for noninvasive mechanical ventilation, and week of evaluation.
Patients were a mean age of 61 years (range, 51-73) and 56% were men; 28% had been on a ventilator during hospitalization for COVID-19.
The most frequent signs and symptoms at 6 months in the patients with long COVID were asthenia (weakness, 38% of patients), dysgeusia (bad taste in the mouth, 34%), dyspnea (shortness of breath, 34%), and anosmia (loss of sense of smell, 24%).
Most symptoms were related to the cardiorespiratory system (42%), the feeling of well-being (42%), or the senses (36%), and fewer patients had symptoms related to neurocognitive impairment (headache or brain fog, 14%), or ear, nose, and throat (12%), or gastrointestinal system (4%).
Patients with long COVID had lower mean 25(OH) vitamin D levels than patients without long COVID (20.1 vs 23.2 ng/mL; P = .03). However, actual vitamin D deficiency levels were similar in both groups.
Two-thirds of patients with low vitamin D levels at hospital admission still presented with low levels at the 6-month follow-up.
Vitamin D levels were significantly lower in patients with neurocognitive symptoms at follow-up (n = 7) than in those without such symptoms (n = 93) (14.6 vs. 20.6 ng/mL; P = .042).
In patients with vitamin D deficiency (< 20 ng/mL) at admission and at follow-up (n = 42), those with long COVID (n = 22) had lower vitamin D levels at follow-up than those without long COVID (n = 20) (12.7 vs. 15.2 ng/mL; P = .041).
And in multiple regression analyses, a lower 25(OH) vitamin D level at follow-up was the only variable that was significantly associated with long COVID (odds ratio, 1.09; 95% confidence interval, 1.01-1.16; P = .008).
The findings “strongly reinforce the clinical usefulness of 25(OH) vitamin D evaluation as a possible modifiable pathophysiological factor underlying this emerging worldwide critical health issue,” the researchers concluded.
The study was supported by Abiogen Pharma. One study author is an employee at Abiogen. Dr. Giustina has reported being a consultant for Abiogen and Takeda and receiving a research grant to his institution from Takeda. Dr. Di Filippo and the other authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ECE 2023