User login
Type 2 Diabetes Increases the Risk of Parkinson’s Disease
The risk is particularly high among younger patients and those with complications from diabetes.
Patients with type 2 diabetes mellitus have an increased risk of developing Parkinson’s disease later in life, according to an investigation published online ahead of print June 13 in Neurology. The magnitude of risk is greater in younger patients and in patients with complications from diabetes.
Investigators have hypothesized an association between diabetes and the risk of Parkinson’s disease, but studies of the potential link have had conflicting results. Thomas T. Warner, MD, PhD, Professor of Clinical Neurology at University College London (UCL), and colleagues conducted a retrospective cohort study to examine this question anew.
Analyzing a Nationwide Hospital Database
The researchers reviewed English national Hospital Episode Statistics and mortality data collected between 1999 and 2011 and created a cohort of 2,017,115 patients who had been admitted for hospital care with a diagnosis of type 2 diabetes. They created a reference cohort of 6,173,208 patients without diabetes who were admitted for minor medical and surgical procedures. Conditions in this cohort included sprains, inguinal hernia, bruising, and hip replacement. People with Parkinson’s disease, ischemic cerebral infarction, vascular parkinsonism, drug-induced secondary parkinsonism, and normal pressure hydrocephalus were excluded from the study. Dr. Warner and colleagues created multivariable Cox proportional hazard regression models to estimate the risk of subsequent Parkinson’s disease.
Participants with diabetes had a greater risk of a subsequent diagnosis of Parkinson’s disease than patients in the reference cohort (adjusted hazard ratio [HR], 1.32). In subgroup analyses, the researchers found that the risk was substantially higher among patients between ages 25 and 44 (adjusted HR, 3.81) and those with complicated diabetes (adjusted HR, 1.49). Genetic factors may exert a relatively greater effect on younger people, and this difference may account for the increased risk among younger participants with diabetes, said the authors.
The adjusted HR of Parkinson’s disease was 1.40 in patients with diabetes between ages 65 and 74 and 1.18 in those age 75 or older. “The association in elderly patients may be the consequence of disrupted insulin signaling secondary to additional lifestyle and environmental factors causing cumulative pathogenic brain changes,” said Dr. Warner and colleagues.
No Adjustment for Potential Confounders
The large size of the database and the ability to exclude people with cerebrovascular disease and drug-induced and vascular parkinsonisms were among the study’s main strengths, according to the authors. Its weaknesses included an inability to adjust for potential confounders and the lack of clinical information about Parkinson’s disease ascertainment beyond routinely collected data.
The results could help researchers identify “new ways to treat or prevent the development of Parkinson’s disease, such as use of antidiabetes drugs to restore the brain’s insulin signaling,” said Dr. Warner. “A UCL-led study published last year found that a drug commonly used to treat diabetes shows promise in not only relieving Parkinson’s disease symptoms, but potentially altering the course of the disease itself. What we do not know is whether trying to treat people with type 2 diabetes better would reduce the risk of developing Parkinson’s disease.”
—Erik Greb
Suggested Reading
De Pablo-Fernandez E, Goldacre R, Pakpoor J, et al. Association between diabetes and subsequent Parkinson disease: a record-linkage cohort study. Neurology. 2018 Jun 13 [Epub ahead of print].
The risk is particularly high among younger patients and those with complications from diabetes.
The risk is particularly high among younger patients and those with complications from diabetes.
Patients with type 2 diabetes mellitus have an increased risk of developing Parkinson’s disease later in life, according to an investigation published online ahead of print June 13 in Neurology. The magnitude of risk is greater in younger patients and in patients with complications from diabetes.
Investigators have hypothesized an association between diabetes and the risk of Parkinson’s disease, but studies of the potential link have had conflicting results. Thomas T. Warner, MD, PhD, Professor of Clinical Neurology at University College London (UCL), and colleagues conducted a retrospective cohort study to examine this question anew.
Analyzing a Nationwide Hospital Database
The researchers reviewed English national Hospital Episode Statistics and mortality data collected between 1999 and 2011 and created a cohort of 2,017,115 patients who had been admitted for hospital care with a diagnosis of type 2 diabetes. They created a reference cohort of 6,173,208 patients without diabetes who were admitted for minor medical and surgical procedures. Conditions in this cohort included sprains, inguinal hernia, bruising, and hip replacement. People with Parkinson’s disease, ischemic cerebral infarction, vascular parkinsonism, drug-induced secondary parkinsonism, and normal pressure hydrocephalus were excluded from the study. Dr. Warner and colleagues created multivariable Cox proportional hazard regression models to estimate the risk of subsequent Parkinson’s disease.
Participants with diabetes had a greater risk of a subsequent diagnosis of Parkinson’s disease than patients in the reference cohort (adjusted hazard ratio [HR], 1.32). In subgroup analyses, the researchers found that the risk was substantially higher among patients between ages 25 and 44 (adjusted HR, 3.81) and those with complicated diabetes (adjusted HR, 1.49). Genetic factors may exert a relatively greater effect on younger people, and this difference may account for the increased risk among younger participants with diabetes, said the authors.
The adjusted HR of Parkinson’s disease was 1.40 in patients with diabetes between ages 65 and 74 and 1.18 in those age 75 or older. “The association in elderly patients may be the consequence of disrupted insulin signaling secondary to additional lifestyle and environmental factors causing cumulative pathogenic brain changes,” said Dr. Warner and colleagues.
No Adjustment for Potential Confounders
The large size of the database and the ability to exclude people with cerebrovascular disease and drug-induced and vascular parkinsonisms were among the study’s main strengths, according to the authors. Its weaknesses included an inability to adjust for potential confounders and the lack of clinical information about Parkinson’s disease ascertainment beyond routinely collected data.
The results could help researchers identify “new ways to treat or prevent the development of Parkinson’s disease, such as use of antidiabetes drugs to restore the brain’s insulin signaling,” said Dr. Warner. “A UCL-led study published last year found that a drug commonly used to treat diabetes shows promise in not only relieving Parkinson’s disease symptoms, but potentially altering the course of the disease itself. What we do not know is whether trying to treat people with type 2 diabetes better would reduce the risk of developing Parkinson’s disease.”
—Erik Greb
Suggested Reading
De Pablo-Fernandez E, Goldacre R, Pakpoor J, et al. Association between diabetes and subsequent Parkinson disease: a record-linkage cohort study. Neurology. 2018 Jun 13 [Epub ahead of print].
Patients with type 2 diabetes mellitus have an increased risk of developing Parkinson’s disease later in life, according to an investigation published online ahead of print June 13 in Neurology. The magnitude of risk is greater in younger patients and in patients with complications from diabetes.
Investigators have hypothesized an association between diabetes and the risk of Parkinson’s disease, but studies of the potential link have had conflicting results. Thomas T. Warner, MD, PhD, Professor of Clinical Neurology at University College London (UCL), and colleagues conducted a retrospective cohort study to examine this question anew.
Analyzing a Nationwide Hospital Database
The researchers reviewed English national Hospital Episode Statistics and mortality data collected between 1999 and 2011 and created a cohort of 2,017,115 patients who had been admitted for hospital care with a diagnosis of type 2 diabetes. They created a reference cohort of 6,173,208 patients without diabetes who were admitted for minor medical and surgical procedures. Conditions in this cohort included sprains, inguinal hernia, bruising, and hip replacement. People with Parkinson’s disease, ischemic cerebral infarction, vascular parkinsonism, drug-induced secondary parkinsonism, and normal pressure hydrocephalus were excluded from the study. Dr. Warner and colleagues created multivariable Cox proportional hazard regression models to estimate the risk of subsequent Parkinson’s disease.
Participants with diabetes had a greater risk of a subsequent diagnosis of Parkinson’s disease than patients in the reference cohort (adjusted hazard ratio [HR], 1.32). In subgroup analyses, the researchers found that the risk was substantially higher among patients between ages 25 and 44 (adjusted HR, 3.81) and those with complicated diabetes (adjusted HR, 1.49). Genetic factors may exert a relatively greater effect on younger people, and this difference may account for the increased risk among younger participants with diabetes, said the authors.
The adjusted HR of Parkinson’s disease was 1.40 in patients with diabetes between ages 65 and 74 and 1.18 in those age 75 or older. “The association in elderly patients may be the consequence of disrupted insulin signaling secondary to additional lifestyle and environmental factors causing cumulative pathogenic brain changes,” said Dr. Warner and colleagues.
No Adjustment for Potential Confounders
The large size of the database and the ability to exclude people with cerebrovascular disease and drug-induced and vascular parkinsonisms were among the study’s main strengths, according to the authors. Its weaknesses included an inability to adjust for potential confounders and the lack of clinical information about Parkinson’s disease ascertainment beyond routinely collected data.
The results could help researchers identify “new ways to treat or prevent the development of Parkinson’s disease, such as use of antidiabetes drugs to restore the brain’s insulin signaling,” said Dr. Warner. “A UCL-led study published last year found that a drug commonly used to treat diabetes shows promise in not only relieving Parkinson’s disease symptoms, but potentially altering the course of the disease itself. What we do not know is whether trying to treat people with type 2 diabetes better would reduce the risk of developing Parkinson’s disease.”
—Erik Greb
Suggested Reading
De Pablo-Fernandez E, Goldacre R, Pakpoor J, et al. Association between diabetes and subsequent Parkinson disease: a record-linkage cohort study. Neurology. 2018 Jun 13 [Epub ahead of print].
DLBCL survival improved with novel antibody-drug conjugate
STOCKHOLM, SWEDEN – Adding an experimental antibody-drug conjugate to bendamustine and rituximab more than doubled overall survival over bendamustine/rituximab alone in patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), investigators reported.
Among 80 transplant-ineligible patients with relapsed or refractory DLBCL in a phase 2 trial, the combination of the antibody-drug conjugate (ADC) polatuzumab vedotin plus bendamustine/rituximab (BR) was associated with a 40% complete response rate, compared with 15% for BR alone.
More importantly, the ADC was associated with 6.7 months median progression-free survival (PFS) versus 2 months for BR, and 11.8 months median overall survival (OS), versus 4.7 months for BR alone, reported Laurie Sehn, MD, from BC Cancer in Vancouver, British Columbia, Canada.
“This randomized phase 2 trial really is, so far, the only head-to-head comparison of a novel targeted agent against a standard therapy in this patient population that’s ineligible for transplant, and it demonstrated that the combination polatuzumab vedotin with BR significantly improved the response rates and progression-free survival, as well as overall survival,” she said at the annual congress of the European Hematology Association.
However, in a separate cohort of patients with follicular lymphoma in the same trial, there was no difference in either PFS or OS during follow-up to date, Dr. Sehn reported.
Polatuzumab vedotin consists of an antibody targeted against CD79b, an antigenic protein expressed on the surface of normal B cells, as well as DLBCL and follicular lymphoma cells.
Dr. Sehn and her colleagues enrolled 80 patients with DLBCL for whom first-line chemoimmunotherapy had failed and who were ineligible for stem cell transplant due to age and/or comorbidities.
A second cohort included 80 patients with follicular lymphoma. In this group, median PFS with polatuzumab vedotin/BR was 17 months versus 17.3 months for BR alone, and median overall survival had not been reached in either arm at the time of the data cutoff.
In the DLBCL cohort, patients were randomized to receive polatuzumab vedotin 1.8 mg/kg plus bendamustine 90mg/m2 for 2 days and rituximab 375mg/m2) or BR alone for six 21-day cycles.
The complete response rate by PET scan – the primary endpoint – was significantly higher with polatuzmab/BR at 40% versus 15% for BR alone (P = .012). Respective overall response rates were 45% versus 18% (P = .008). Also, median PFS with the polatuzmab/BR therapy was 6.7 months versus 2.0 months for BR alone, translating into a hazard ratio of 0.31 (P less than .0001).
Respective median overall survival was 11.8 versus 4.7 months, translating into a hazard ratio for the polatuzmab/BR combination of 0.35 (P = .0008).
The PET complete response rates were higher with polatuzmab/BR regardless of prior lines of therapy or refractory status, Dr. Sehn noted.
“In terms of the safety, I think importantly in the combination there were no unexpected toxicities, so typical to what we would expect with what’s known with this drug alone,” Dr. Sehn said.
Grade 3 or greater toxicities that were higher with the polatuzmab/BR combination included cytopenias, febrile neutropenia, and infections. The single serious adverse event that had a higher incidence in the polatuzumab/BR arm was febrile neutropenia (DLBCL). In total, 12% of patients in the polatuzumab-containing arm and 11% of patients in the BR-only arm died on study. Many of the deaths were due to disease progression.
Anton Hagenbeek, MD, PhD, from the Academic Medical Center at the University of Amsterdam, the Netherlands, who moderated the briefing but was not involved in the study, said that about 20%-30% of patients with relapsed/refractory DLBCL are positive for the CD33 antigen, the target of brentuximab vedotin (Adcetris), and noted that this agent is also being tested in a phase 2 trial.
Martin Hutchings, MD, PhD, from Rigshospitalet in Copenhagen, the Netherlands, who co-moderated the oral abstract session, commented that “it’s not so often that we see significant overall survival differences in a phase 2 study with 80 patients.”
Based on the results of this trial, polatuzumab has been granted breakthrough therapy designation by the U.S. Food and Drug Administration and a PRIME (priority medicine) designation from the European Medicines Agency.
SOURCE: Sehn LH et al. EHA Congress, Abstract S802.
STOCKHOLM, SWEDEN – Adding an experimental antibody-drug conjugate to bendamustine and rituximab more than doubled overall survival over bendamustine/rituximab alone in patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), investigators reported.
Among 80 transplant-ineligible patients with relapsed or refractory DLBCL in a phase 2 trial, the combination of the antibody-drug conjugate (ADC) polatuzumab vedotin plus bendamustine/rituximab (BR) was associated with a 40% complete response rate, compared with 15% for BR alone.
More importantly, the ADC was associated with 6.7 months median progression-free survival (PFS) versus 2 months for BR, and 11.8 months median overall survival (OS), versus 4.7 months for BR alone, reported Laurie Sehn, MD, from BC Cancer in Vancouver, British Columbia, Canada.
“This randomized phase 2 trial really is, so far, the only head-to-head comparison of a novel targeted agent against a standard therapy in this patient population that’s ineligible for transplant, and it demonstrated that the combination polatuzumab vedotin with BR significantly improved the response rates and progression-free survival, as well as overall survival,” she said at the annual congress of the European Hematology Association.
However, in a separate cohort of patients with follicular lymphoma in the same trial, there was no difference in either PFS or OS during follow-up to date, Dr. Sehn reported.
Polatuzumab vedotin consists of an antibody targeted against CD79b, an antigenic protein expressed on the surface of normal B cells, as well as DLBCL and follicular lymphoma cells.
Dr. Sehn and her colleagues enrolled 80 patients with DLBCL for whom first-line chemoimmunotherapy had failed and who were ineligible for stem cell transplant due to age and/or comorbidities.
A second cohort included 80 patients with follicular lymphoma. In this group, median PFS with polatuzumab vedotin/BR was 17 months versus 17.3 months for BR alone, and median overall survival had not been reached in either arm at the time of the data cutoff.
In the DLBCL cohort, patients were randomized to receive polatuzumab vedotin 1.8 mg/kg plus bendamustine 90mg/m2 for 2 days and rituximab 375mg/m2) or BR alone for six 21-day cycles.
The complete response rate by PET scan – the primary endpoint – was significantly higher with polatuzmab/BR at 40% versus 15% for BR alone (P = .012). Respective overall response rates were 45% versus 18% (P = .008). Also, median PFS with the polatuzmab/BR therapy was 6.7 months versus 2.0 months for BR alone, translating into a hazard ratio of 0.31 (P less than .0001).
Respective median overall survival was 11.8 versus 4.7 months, translating into a hazard ratio for the polatuzmab/BR combination of 0.35 (P = .0008).
The PET complete response rates were higher with polatuzmab/BR regardless of prior lines of therapy or refractory status, Dr. Sehn noted.
“In terms of the safety, I think importantly in the combination there were no unexpected toxicities, so typical to what we would expect with what’s known with this drug alone,” Dr. Sehn said.
Grade 3 or greater toxicities that were higher with the polatuzmab/BR combination included cytopenias, febrile neutropenia, and infections. The single serious adverse event that had a higher incidence in the polatuzumab/BR arm was febrile neutropenia (DLBCL). In total, 12% of patients in the polatuzumab-containing arm and 11% of patients in the BR-only arm died on study. Many of the deaths were due to disease progression.
Anton Hagenbeek, MD, PhD, from the Academic Medical Center at the University of Amsterdam, the Netherlands, who moderated the briefing but was not involved in the study, said that about 20%-30% of patients with relapsed/refractory DLBCL are positive for the CD33 antigen, the target of brentuximab vedotin (Adcetris), and noted that this agent is also being tested in a phase 2 trial.
Martin Hutchings, MD, PhD, from Rigshospitalet in Copenhagen, the Netherlands, who co-moderated the oral abstract session, commented that “it’s not so often that we see significant overall survival differences in a phase 2 study with 80 patients.”
Based on the results of this trial, polatuzumab has been granted breakthrough therapy designation by the U.S. Food and Drug Administration and a PRIME (priority medicine) designation from the European Medicines Agency.
SOURCE: Sehn LH et al. EHA Congress, Abstract S802.
STOCKHOLM, SWEDEN – Adding an experimental antibody-drug conjugate to bendamustine and rituximab more than doubled overall survival over bendamustine/rituximab alone in patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), investigators reported.
Among 80 transplant-ineligible patients with relapsed or refractory DLBCL in a phase 2 trial, the combination of the antibody-drug conjugate (ADC) polatuzumab vedotin plus bendamustine/rituximab (BR) was associated with a 40% complete response rate, compared with 15% for BR alone.
More importantly, the ADC was associated with 6.7 months median progression-free survival (PFS) versus 2 months for BR, and 11.8 months median overall survival (OS), versus 4.7 months for BR alone, reported Laurie Sehn, MD, from BC Cancer in Vancouver, British Columbia, Canada.
“This randomized phase 2 trial really is, so far, the only head-to-head comparison of a novel targeted agent against a standard therapy in this patient population that’s ineligible for transplant, and it demonstrated that the combination polatuzumab vedotin with BR significantly improved the response rates and progression-free survival, as well as overall survival,” she said at the annual congress of the European Hematology Association.
However, in a separate cohort of patients with follicular lymphoma in the same trial, there was no difference in either PFS or OS during follow-up to date, Dr. Sehn reported.
Polatuzumab vedotin consists of an antibody targeted against CD79b, an antigenic protein expressed on the surface of normal B cells, as well as DLBCL and follicular lymphoma cells.
Dr. Sehn and her colleagues enrolled 80 patients with DLBCL for whom first-line chemoimmunotherapy had failed and who were ineligible for stem cell transplant due to age and/or comorbidities.
A second cohort included 80 patients with follicular lymphoma. In this group, median PFS with polatuzumab vedotin/BR was 17 months versus 17.3 months for BR alone, and median overall survival had not been reached in either arm at the time of the data cutoff.
In the DLBCL cohort, patients were randomized to receive polatuzumab vedotin 1.8 mg/kg plus bendamustine 90mg/m2 for 2 days and rituximab 375mg/m2) or BR alone for six 21-day cycles.
The complete response rate by PET scan – the primary endpoint – was significantly higher with polatuzmab/BR at 40% versus 15% for BR alone (P = .012). Respective overall response rates were 45% versus 18% (P = .008). Also, median PFS with the polatuzmab/BR therapy was 6.7 months versus 2.0 months for BR alone, translating into a hazard ratio of 0.31 (P less than .0001).
Respective median overall survival was 11.8 versus 4.7 months, translating into a hazard ratio for the polatuzmab/BR combination of 0.35 (P = .0008).
The PET complete response rates were higher with polatuzmab/BR regardless of prior lines of therapy or refractory status, Dr. Sehn noted.
“In terms of the safety, I think importantly in the combination there were no unexpected toxicities, so typical to what we would expect with what’s known with this drug alone,” Dr. Sehn said.
Grade 3 or greater toxicities that were higher with the polatuzmab/BR combination included cytopenias, febrile neutropenia, and infections. The single serious adverse event that had a higher incidence in the polatuzumab/BR arm was febrile neutropenia (DLBCL). In total, 12% of patients in the polatuzumab-containing arm and 11% of patients in the BR-only arm died on study. Many of the deaths were due to disease progression.
Anton Hagenbeek, MD, PhD, from the Academic Medical Center at the University of Amsterdam, the Netherlands, who moderated the briefing but was not involved in the study, said that about 20%-30% of patients with relapsed/refractory DLBCL are positive for the CD33 antigen, the target of brentuximab vedotin (Adcetris), and noted that this agent is also being tested in a phase 2 trial.
Martin Hutchings, MD, PhD, from Rigshospitalet in Copenhagen, the Netherlands, who co-moderated the oral abstract session, commented that “it’s not so often that we see significant overall survival differences in a phase 2 study with 80 patients.”
Based on the results of this trial, polatuzumab has been granted breakthrough therapy designation by the U.S. Food and Drug Administration and a PRIME (priority medicine) designation from the European Medicines Agency.
SOURCE: Sehn LH et al. EHA Congress, Abstract S802.
REPORTING FROM THE EHA CONGRESS
Key clinical point:
Major finding: The complete response rate with polatuzumab vedotin plus bendamustine/rituximab (BR) was 40%, compared with 15% for BR alone.
Study details: Randomized controlled phase 2 trial in 80 patients with relapsed/refractory DLBCL.
Disclosures: The study was funded by Hoffman-La Roche. Dr. Sehn reported ties to Roche/Genentech and others.
Source: Sehn LH et al. EHA Congress, Abstract S802.
‘Excellent’ survival with HCT despite early treatment failure in FL
Autologous and allogeneic hematopoietic stem cell transplantation (HCT) both offer excellent long-term survival in follicular lymphoma (FL) patients who experience early treatment failure, an analysis of a large transplant registry suggests.
Five-year survival rates exceeded 70% for patients who received autologous or matched sibling donor (MSD) transplants, according to the analysis of the Center for International Blood and Marrow Transplant Research (CIBMTR) database. The database included 440 patients who underwent a procedure between 2002 and 2014.
“Until better risk-stratification tools are available for FL, auto-HCT and MSD allo-HCT should be considered as effective treatment options with excellent long-term survival for high-risk patients as defined by early treatment failure,” Sonali M. Smith, MD, of the University of Chicago, and co-investigators wrote in the journal Cancer.
Early treatment failure in FL is associated with worse overall survival. In the National LymphoCare Study (NLCS), patients who received upfront R-CHOP therapy and progressed within 24 months had a 5-year overall survival of 50%, versus 90% for patients without early progression.
By contrast, survival figures in the present study are “provocatively higher” than those in the NLCS, in which only 8 out of 110 patients underwent HCT, Dr Smith and co-authors said.
Dr Smith’s study showed that with a median follow-up of 69 to 73 months, adjusted probability of 5-year overall survival was 70% for autologous and 73% for MSD HCT, versus 49% for matched unrelated donor HCT (P=0.0008).
Ryan C. Lynch, MD, and Ajay K. Gopal, MD, of the Fred Hutchinson Cancer Research Center in Seattle, Washington, said that the finding “convincingly demonstrates” the benefit of transplant in the setting of early treatment failure.
“Select patients (particularly younger patients) with chemoresponsive disease who understand the risk-benefit ratio in comparison with currently approved and experimental therapies still remain good candidates for autologous HCT,” Drs Lynch and Gopal said in an editorial.
“For older patients or patients with comorbidities, we would continue to recommend clinical trials or treatment with an approved PI3K inhibitor,” they added.
The study by Dr Smith and colleagues is not the first to show a benefit of HCT in this clinical scenario. In a recent NLCS/CIBMTR analysis of FL patients, 5-year overall survival was 73% for those undergoing autologous HCT done within a year of early treatment failure, versus 60% for those who did not (P=0.05).
The two studies “collectively suggest that transplantation should be considered in this high-risk group of patients with early relapse,” Dr Smith and co-authors wrote.
Autologous and allogeneic hematopoietic stem cell transplantation (HCT) both offer excellent long-term survival in follicular lymphoma (FL) patients who experience early treatment failure, an analysis of a large transplant registry suggests.
Five-year survival rates exceeded 70% for patients who received autologous or matched sibling donor (MSD) transplants, according to the analysis of the Center for International Blood and Marrow Transplant Research (CIBMTR) database. The database included 440 patients who underwent a procedure between 2002 and 2014.
“Until better risk-stratification tools are available for FL, auto-HCT and MSD allo-HCT should be considered as effective treatment options with excellent long-term survival for high-risk patients as defined by early treatment failure,” Sonali M. Smith, MD, of the University of Chicago, and co-investigators wrote in the journal Cancer.
Early treatment failure in FL is associated with worse overall survival. In the National LymphoCare Study (NLCS), patients who received upfront R-CHOP therapy and progressed within 24 months had a 5-year overall survival of 50%, versus 90% for patients without early progression.
By contrast, survival figures in the present study are “provocatively higher” than those in the NLCS, in which only 8 out of 110 patients underwent HCT, Dr Smith and co-authors said.
Dr Smith’s study showed that with a median follow-up of 69 to 73 months, adjusted probability of 5-year overall survival was 70% for autologous and 73% for MSD HCT, versus 49% for matched unrelated donor HCT (P=0.0008).
Ryan C. Lynch, MD, and Ajay K. Gopal, MD, of the Fred Hutchinson Cancer Research Center in Seattle, Washington, said that the finding “convincingly demonstrates” the benefit of transplant in the setting of early treatment failure.
“Select patients (particularly younger patients) with chemoresponsive disease who understand the risk-benefit ratio in comparison with currently approved and experimental therapies still remain good candidates for autologous HCT,” Drs Lynch and Gopal said in an editorial.
“For older patients or patients with comorbidities, we would continue to recommend clinical trials or treatment with an approved PI3K inhibitor,” they added.
The study by Dr Smith and colleagues is not the first to show a benefit of HCT in this clinical scenario. In a recent NLCS/CIBMTR analysis of FL patients, 5-year overall survival was 73% for those undergoing autologous HCT done within a year of early treatment failure, versus 60% for those who did not (P=0.05).
The two studies “collectively suggest that transplantation should be considered in this high-risk group of patients with early relapse,” Dr Smith and co-authors wrote.
Autologous and allogeneic hematopoietic stem cell transplantation (HCT) both offer excellent long-term survival in follicular lymphoma (FL) patients who experience early treatment failure, an analysis of a large transplant registry suggests.
Five-year survival rates exceeded 70% for patients who received autologous or matched sibling donor (MSD) transplants, according to the analysis of the Center for International Blood and Marrow Transplant Research (CIBMTR) database. The database included 440 patients who underwent a procedure between 2002 and 2014.
“Until better risk-stratification tools are available for FL, auto-HCT and MSD allo-HCT should be considered as effective treatment options with excellent long-term survival for high-risk patients as defined by early treatment failure,” Sonali M. Smith, MD, of the University of Chicago, and co-investigators wrote in the journal Cancer.
Early treatment failure in FL is associated with worse overall survival. In the National LymphoCare Study (NLCS), patients who received upfront R-CHOP therapy and progressed within 24 months had a 5-year overall survival of 50%, versus 90% for patients without early progression.
By contrast, survival figures in the present study are “provocatively higher” than those in the NLCS, in which only 8 out of 110 patients underwent HCT, Dr Smith and co-authors said.
Dr Smith’s study showed that with a median follow-up of 69 to 73 months, adjusted probability of 5-year overall survival was 70% for autologous and 73% for MSD HCT, versus 49% for matched unrelated donor HCT (P=0.0008).
Ryan C. Lynch, MD, and Ajay K. Gopal, MD, of the Fred Hutchinson Cancer Research Center in Seattle, Washington, said that the finding “convincingly demonstrates” the benefit of transplant in the setting of early treatment failure.
“Select patients (particularly younger patients) with chemoresponsive disease who understand the risk-benefit ratio in comparison with currently approved and experimental therapies still remain good candidates for autologous HCT,” Drs Lynch and Gopal said in an editorial.
“For older patients or patients with comorbidities, we would continue to recommend clinical trials or treatment with an approved PI3K inhibitor,” they added.
The study by Dr Smith and colleagues is not the first to show a benefit of HCT in this clinical scenario. In a recent NLCS/CIBMTR analysis of FL patients, 5-year overall survival was 73% for those undergoing autologous HCT done within a year of early treatment failure, versus 60% for those who did not (P=0.05).
The two studies “collectively suggest that transplantation should be considered in this high-risk group of patients with early relapse,” Dr Smith and co-authors wrote.
British good practice paper offers MCL diagnosis pearls
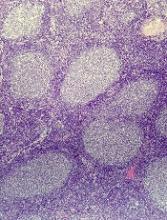
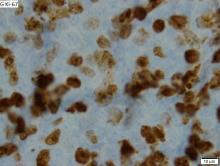
Immunohistochemical panels used in the diagnosis of mantle cell lymphoma should include cyclin D1 and SOX11 immunostaining, according to a good practice paper from the British Society of Haematology.
Pamela McKay, MD, of the Beatson West of Scotland Cancer Centre, Glasgow, and her colleagues provided based on a review of literature from 1980 to 2017. The good practice paper aims to offer best practice advice based on consensus where the evidence is limited. Specifically, the paper incorporates new information on molecular pathology and the use of positron emission tomography/computed tomography (PET/CT) scanning in staging of disease.
The top recommendations related to MCL diagnosis include performing lymph node excision or adequate core biopsy for diagnosis of nodal MCL. For non-nodal presentation, a tissue biopsy or peripheral blood can be used. Additionally, immunohistochemical panels should include cyclin D1 and SOX11 immunostaining.
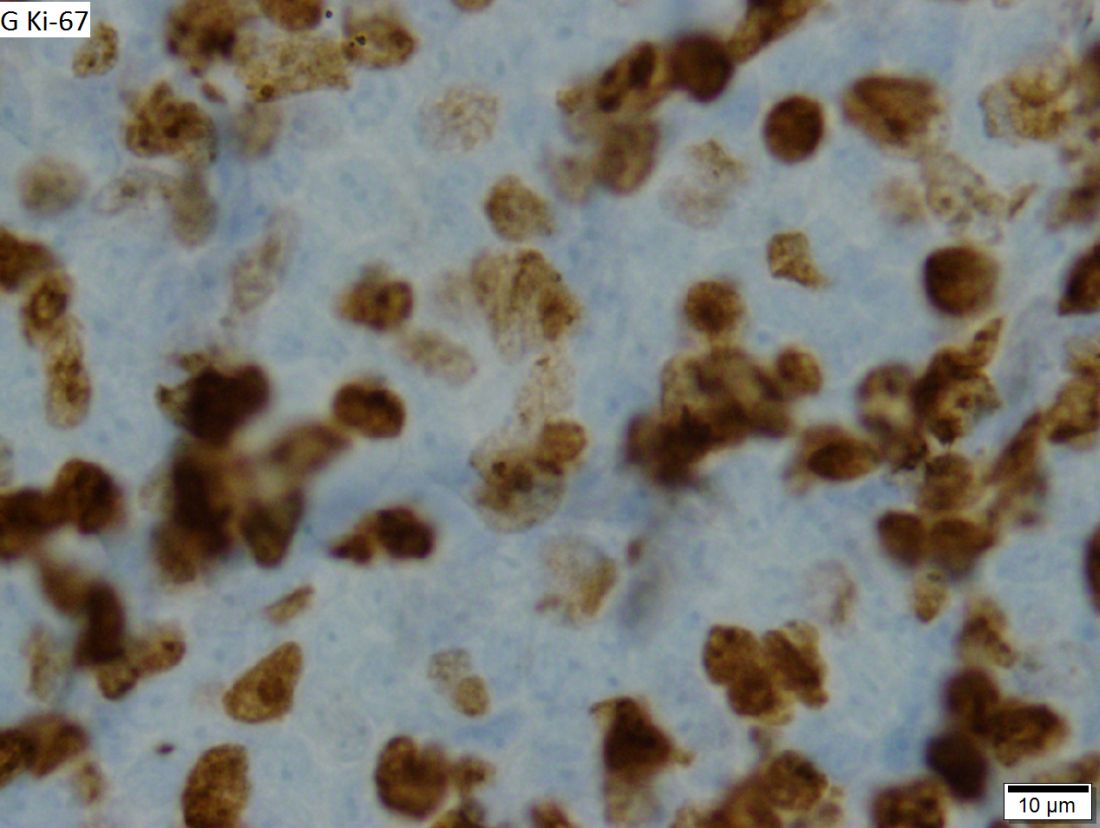
In cases of atypical morphology, aberrant immunophenotype, equivocal cyclin D1 positivity, or unusual clinical presentation, the authors recommended fluorescence in situ hybridization (FISH) to demonstrate the presence of the t(11;14) translocation. They also recommended recording the Ki67 Proliferation Index at baseline, with an index of greater than 30% being indicative of a poorer outcome.
In terms of staging disease, Dr. McKay and her associates recommended that patients undergo staging with CT of the neck, chest, abdomen, and pelvis. They recommended against routine use of fluorodeoxyglucose PET for MCL staging, but said it could be considered if radical radiotherapy is being proposed for early-stage disease.
For cases with suspicion of central nervous system involvement, lumbar puncture with cytospin and immunophenotyping is recommended.
They recommended that all MCL patients have either their simplified or combined MCL international prognostic index score recorded at baseline.
All the authors made a declaration of interest to the British Society of Haematology and task force chairs, which may be viewed on request.
SOURCE: McKay P et al. Br J Haematol. 2018 Jun 8. doi: 10.1111/bjh.15281.
Immunohistochemical panels used in the diagnosis of mantle cell lymphoma should include cyclin D1 and SOX11 immunostaining, according to a good practice paper from the British Society of Haematology.
Pamela McKay, MD, of the Beatson West of Scotland Cancer Centre, Glasgow, and her colleagues provided based on a review of literature from 1980 to 2017. The good practice paper aims to offer best practice advice based on consensus where the evidence is limited. Specifically, the paper incorporates new information on molecular pathology and the use of positron emission tomography/computed tomography (PET/CT) scanning in staging of disease.
The top recommendations related to MCL diagnosis include performing lymph node excision or adequate core biopsy for diagnosis of nodal MCL. For non-nodal presentation, a tissue biopsy or peripheral blood can be used. Additionally, immunohistochemical panels should include cyclin D1 and SOX11 immunostaining.
In cases of atypical morphology, aberrant immunophenotype, equivocal cyclin D1 positivity, or unusual clinical presentation, the authors recommended fluorescence in situ hybridization (FISH) to demonstrate the presence of the t(11;14) translocation. They also recommended recording the Ki67 Proliferation Index at baseline, with an index of greater than 30% being indicative of a poorer outcome.
In terms of staging disease, Dr. McKay and her associates recommended that patients undergo staging with CT of the neck, chest, abdomen, and pelvis. They recommended against routine use of fluorodeoxyglucose PET for MCL staging, but said it could be considered if radical radiotherapy is being proposed for early-stage disease.
For cases with suspicion of central nervous system involvement, lumbar puncture with cytospin and immunophenotyping is recommended.
They recommended that all MCL patients have either their simplified or combined MCL international prognostic index score recorded at baseline.
All the authors made a declaration of interest to the British Society of Haematology and task force chairs, which may be viewed on request.
SOURCE: McKay P et al. Br J Haematol. 2018 Jun 8. doi: 10.1111/bjh.15281.
Immunohistochemical panels used in the diagnosis of mantle cell lymphoma should include cyclin D1 and SOX11 immunostaining, according to a good practice paper from the British Society of Haematology.
Pamela McKay, MD, of the Beatson West of Scotland Cancer Centre, Glasgow, and her colleagues provided based on a review of literature from 1980 to 2017. The good practice paper aims to offer best practice advice based on consensus where the evidence is limited. Specifically, the paper incorporates new information on molecular pathology and the use of positron emission tomography/computed tomography (PET/CT) scanning in staging of disease.
The top recommendations related to MCL diagnosis include performing lymph node excision or adequate core biopsy for diagnosis of nodal MCL. For non-nodal presentation, a tissue biopsy or peripheral blood can be used. Additionally, immunohistochemical panels should include cyclin D1 and SOX11 immunostaining.
In cases of atypical morphology, aberrant immunophenotype, equivocal cyclin D1 positivity, or unusual clinical presentation, the authors recommended fluorescence in situ hybridization (FISH) to demonstrate the presence of the t(11;14) translocation. They also recommended recording the Ki67 Proliferation Index at baseline, with an index of greater than 30% being indicative of a poorer outcome.
In terms of staging disease, Dr. McKay and her associates recommended that patients undergo staging with CT of the neck, chest, abdomen, and pelvis. They recommended against routine use of fluorodeoxyglucose PET for MCL staging, but said it could be considered if radical radiotherapy is being proposed for early-stage disease.
For cases with suspicion of central nervous system involvement, lumbar puncture with cytospin and immunophenotyping is recommended.
They recommended that all MCL patients have either their simplified or combined MCL international prognostic index score recorded at baseline.
All the authors made a declaration of interest to the British Society of Haematology and task force chairs, which may be viewed on request.
SOURCE: McKay P et al. Br J Haematol. 2018 Jun 8. doi: 10.1111/bjh.15281.
FROM THE BRITISH JOURNAL OF HAEMATOLOGY
Risk of Recurrent ICH Is Higher Among Blacks and Hispanics
The increased severity of hypertension among minorities does not fully account for their increased risk.
Compared with their white peers, black and Hispanic patients with intracerebral hemorrhage (ICH) have a higher risk of recurrence, according to data published online ahead of print June 6 in Neurology. Although black and Hispanic patients have more severe hypertension than whites do, severity of hypertension does not fully account for this increased risk. Future studies should examine whether novel biologic, socioeconomic, or cultural factors play a role, said the researchers.
The scientific literature indicates that blacks and Hispanics have a higher risk of first ICH than whites do. Alessandro Biffi, MD, head of the Aging and Brain Health Research group at Massachusetts General Hospital in Boston, and colleagues hypothesized that hypertension among these populations might contribute toward this increased risk. Because the subject had not been explored previously, Dr. Biffi and colleagues investigated the role of blood pressure and its variability in determining the risk of recurrent ICH among nonwhites.
An Analysis of Two Studies
The authors examined data from a longitudinal study of ICH conducted by Massachusetts General Hospital and from the Ethnic/Racial Variations of Intracerebral Hemorrhage (ERICH) study. They included patients who were 18 or older with a diagnosis of acute primary ICH in their analysis.
At enrollment, participants reported their race or ethnicity during a structured interview and underwent blood pressure measurement. The investigators performed follow-up through phone calls and reviews of medical records. Every six months, investigators recorded at least one blood-pressure measurement and quantified blood-pressure variability. Dr. Biffi and colleagues used Cox regression survival analysis to identify risk factors for ICH recurrence.
Systolic Blood Pressure Was Associated With Recurrence
Of the 2,291 patients included in the analysis, 1,121 were white, 529 were black, 605 were Hispanic, and 36 were of other race or ethnicity. The median systolic blood pressure during follow-up was 149 mm Hg for black participants, 146 mm Hg for Hispanic participants, and 141 mm Hg for white participants. Systolic blood pressure variability also was higher for black participants (median, 3.5%), compared with white participants (median
In all, 26 (1.7%) white participants had ICH recurrence, compared with 35 (6.6%) black participants and 37 (6.1%) Hispanic participants. In univariable analyses, higher systolic blood pressure and greater systolic blood pressure variability were associated with increased ICH recurrence risk. Diastolic blood pressure and diastolic blood pressure variability, however, were not associated with ICH recurrence risk.
In multivariable analyses, black and Hispanic race or ethnicity remained independently associated with increased ICH recurrence risk in both studies, even after adjustment for systolic blood pressure and systolic blood pressure variability. Exposure to antihypertensive agents during follow-up was not associated with ICH recurrence and did not affect the results significantly. The associations were consistent in both studies.
Suggested Reading
Rodriguez-Torres A, Murphy M, Kourkoulis C, et al. Hypertension and intracerebral hemorrhage recurrence among white, black, and Hispanic individuals. Neurology. 2018 Jun 6 [Epub ahead of print].
The increased severity of hypertension among minorities does not fully account for their increased risk.
The increased severity of hypertension among minorities does not fully account for their increased risk.
Compared with their white peers, black and Hispanic patients with intracerebral hemorrhage (ICH) have a higher risk of recurrence, according to data published online ahead of print June 6 in Neurology. Although black and Hispanic patients have more severe hypertension than whites do, severity of hypertension does not fully account for this increased risk. Future studies should examine whether novel biologic, socioeconomic, or cultural factors play a role, said the researchers.
The scientific literature indicates that blacks and Hispanics have a higher risk of first ICH than whites do. Alessandro Biffi, MD, head of the Aging and Brain Health Research group at Massachusetts General Hospital in Boston, and colleagues hypothesized that hypertension among these populations might contribute toward this increased risk. Because the subject had not been explored previously, Dr. Biffi and colleagues investigated the role of blood pressure and its variability in determining the risk of recurrent ICH among nonwhites.
An Analysis of Two Studies
The authors examined data from a longitudinal study of ICH conducted by Massachusetts General Hospital and from the Ethnic/Racial Variations of Intracerebral Hemorrhage (ERICH) study. They included patients who were 18 or older with a diagnosis of acute primary ICH in their analysis.
At enrollment, participants reported their race or ethnicity during a structured interview and underwent blood pressure measurement. The investigators performed follow-up through phone calls and reviews of medical records. Every six months, investigators recorded at least one blood-pressure measurement and quantified blood-pressure variability. Dr. Biffi and colleagues used Cox regression survival analysis to identify risk factors for ICH recurrence.
Systolic Blood Pressure Was Associated With Recurrence
Of the 2,291 patients included in the analysis, 1,121 were white, 529 were black, 605 were Hispanic, and 36 were of other race or ethnicity. The median systolic blood pressure during follow-up was 149 mm Hg for black participants, 146 mm Hg for Hispanic participants, and 141 mm Hg for white participants. Systolic blood pressure variability also was higher for black participants (median, 3.5%), compared with white participants (median
In all, 26 (1.7%) white participants had ICH recurrence, compared with 35 (6.6%) black participants and 37 (6.1%) Hispanic participants. In univariable analyses, higher systolic blood pressure and greater systolic blood pressure variability were associated with increased ICH recurrence risk. Diastolic blood pressure and diastolic blood pressure variability, however, were not associated with ICH recurrence risk.
In multivariable analyses, black and Hispanic race or ethnicity remained independently associated with increased ICH recurrence risk in both studies, even after adjustment for systolic blood pressure and systolic blood pressure variability. Exposure to antihypertensive agents during follow-up was not associated with ICH recurrence and did not affect the results significantly. The associations were consistent in both studies.
Suggested Reading
Rodriguez-Torres A, Murphy M, Kourkoulis C, et al. Hypertension and intracerebral hemorrhage recurrence among white, black, and Hispanic individuals. Neurology. 2018 Jun 6 [Epub ahead of print].
Compared with their white peers, black and Hispanic patients with intracerebral hemorrhage (ICH) have a higher risk of recurrence, according to data published online ahead of print June 6 in Neurology. Although black and Hispanic patients have more severe hypertension than whites do, severity of hypertension does not fully account for this increased risk. Future studies should examine whether novel biologic, socioeconomic, or cultural factors play a role, said the researchers.
The scientific literature indicates that blacks and Hispanics have a higher risk of first ICH than whites do. Alessandro Biffi, MD, head of the Aging and Brain Health Research group at Massachusetts General Hospital in Boston, and colleagues hypothesized that hypertension among these populations might contribute toward this increased risk. Because the subject had not been explored previously, Dr. Biffi and colleagues investigated the role of blood pressure and its variability in determining the risk of recurrent ICH among nonwhites.
An Analysis of Two Studies
The authors examined data from a longitudinal study of ICH conducted by Massachusetts General Hospital and from the Ethnic/Racial Variations of Intracerebral Hemorrhage (ERICH) study. They included patients who were 18 or older with a diagnosis of acute primary ICH in their analysis.
At enrollment, participants reported their race or ethnicity during a structured interview and underwent blood pressure measurement. The investigators performed follow-up through phone calls and reviews of medical records. Every six months, investigators recorded at least one blood-pressure measurement and quantified blood-pressure variability. Dr. Biffi and colleagues used Cox regression survival analysis to identify risk factors for ICH recurrence.
Systolic Blood Pressure Was Associated With Recurrence
Of the 2,291 patients included in the analysis, 1,121 were white, 529 were black, 605 were Hispanic, and 36 were of other race or ethnicity. The median systolic blood pressure during follow-up was 149 mm Hg for black participants, 146 mm Hg for Hispanic participants, and 141 mm Hg for white participants. Systolic blood pressure variability also was higher for black participants (median, 3.5%), compared with white participants (median
In all, 26 (1.7%) white participants had ICH recurrence, compared with 35 (6.6%) black participants and 37 (6.1%) Hispanic participants. In univariable analyses, higher systolic blood pressure and greater systolic blood pressure variability were associated with increased ICH recurrence risk. Diastolic blood pressure and diastolic blood pressure variability, however, were not associated with ICH recurrence risk.
In multivariable analyses, black and Hispanic race or ethnicity remained independently associated with increased ICH recurrence risk in both studies, even after adjustment for systolic blood pressure and systolic blood pressure variability. Exposure to antihypertensive agents during follow-up was not associated with ICH recurrence and did not affect the results significantly. The associations were consistent in both studies.
Suggested Reading
Rodriguez-Torres A, Murphy M, Kourkoulis C, et al. Hypertension and intracerebral hemorrhage recurrence among white, black, and Hispanic individuals. Neurology. 2018 Jun 6 [Epub ahead of print].
Chemo-free combo provides potential first-line option for FL
CHICAGO—A chemotherapy-free combination of lenalidomide plus rituximab shows similar efficacy and a different safety profile to chemotherapy plus rituximab (R-chemo) followed by rituximab maintenance in patients with previously untreated follicular lymphoma (FL).
According to investigators, the multicenter, international phase 3 RELEVANCE trial is the first to evaluate the chemo-free combination against the standard of care, R-chemo with rituximab maintenance.
“These results show that lenalidomide plus rituximab, a novel immunomodulatory approach, is a potential first-line option for patients with FL requiring treatment,” said investigator Nathan H. Fowler, MD, of the University of Texas MD Anderson Cancer Center in Houston.
Dr Fowler presented the results of the study at the 2018 ASCO Annual Meeting (abstract 7500).
The current standard of care in previously untreated symptomatic FL is immunochemotherapy induction followed by rituximab maintenance.
The immunomodulatory agent lenalidomide has complementary mechanisms with rituximab. Phase 2 studies of combined immunotherapy with lenalidomide and rituximab demonstrated 3-year progression-free survival (PFS) of 79%-81% in previously untreated FL, Dr Fowler said.
Phase 3 RELEVANCE trial (NCT01650701)
Investigators evaluated 1030 previously untreated grade 1-3a FL patients who required therapy.
Patients in the lenalidomide-rituximab group (n=513) received lenalidomide doses of 20 mg per day on days 2 to 22 and 28 for 6 to 12 cycles. Responders continued on therapy at 10 mg per day for a total of 18 cycles.
The rituximab dose was 375 mg/m2 weekly in cycle 1 and day 1 in cycles 2 to 6 and continued in responders for 12 additional cycles.
Patients in the R-chemo arm (n=517) received the investigator’s choice of standard rituximab-CHOP, rituximab-bendamustine, or rituximab-CVP, followed by 12 cycles of rituximab.
Most patients (72%) in the R-chemo arm received R-CHOP.
Baseline characteristics were similar in both groups, Dr Fowler said.
Co-primary endpoints were complete remission/complete remission unconfirmed (CR/Cru) at 120 weeks and PFS.
Results
At a median follow-up of 37.9 months, the superiority for lenalidomide and rituximab over rituximab-chemotherapy was not established.
For the lenalidomide-rituximab patients, the CR/Cru was 48% and 3-year PFS was 77% as compared to 53% and 78%, respectively, for rituximab-chemotherapy patients, as assessed by an independent review committee.
Overall survival was 94% in both groups.
Safety
“Important differences in safety profiles were observed between the arms,” Dr Fowler said.
Rituximab-chemotherapy patients had more frequent neutropenia, febrile neutropenia, growth factor usage, nausea, vomiting, neuropathy, and alopecia.
Lenalidomide and rituximab showed more cutaneous reactions, tumor flare, and diarrhea.
Toxicity profiles differed, with higher grade 4 neutropenia (31% vs 8%) and febrile neutropenia (7% vs 2%) with rituximab-chemotherapy compared with lenalidomide-rituximab, respectively.
More patients experienced grade 3/4 cutaneous events (7% vs 1%) with lenalidomide-rituximab.
Second primary malignancies were slightly higher with rituximab-chemotherapy (10%) than with lenalidomide-rituximab (7%). Grade 5 adverse events were 1% in both groups.
About 70% of patients completed treatment in both groups.
“Lenalidomide and rituximab was not superior to rituximab-chemotherapy based on mature CR/Cru at 120 weeks and interim PFS,” Dr Fowler said. “Both treatments showed similar efficacy results. Treatment effects on PFS were consistent across pre-specified subgroups.”
Dr Fowler presented data as of May 31, 2017. Continued follow-up on PFS and OS is ongoing.
The study is sponsored by Celgene Corporation and the Lymphoma Academic Research Organisation (LYSARC).
CHICAGO—A chemotherapy-free combination of lenalidomide plus rituximab shows similar efficacy and a different safety profile to chemotherapy plus rituximab (R-chemo) followed by rituximab maintenance in patients with previously untreated follicular lymphoma (FL).
According to investigators, the multicenter, international phase 3 RELEVANCE trial is the first to evaluate the chemo-free combination against the standard of care, R-chemo with rituximab maintenance.
“These results show that lenalidomide plus rituximab, a novel immunomodulatory approach, is a potential first-line option for patients with FL requiring treatment,” said investigator Nathan H. Fowler, MD, of the University of Texas MD Anderson Cancer Center in Houston.
Dr Fowler presented the results of the study at the 2018 ASCO Annual Meeting (abstract 7500).
The current standard of care in previously untreated symptomatic FL is immunochemotherapy induction followed by rituximab maintenance.
The immunomodulatory agent lenalidomide has complementary mechanisms with rituximab. Phase 2 studies of combined immunotherapy with lenalidomide and rituximab demonstrated 3-year progression-free survival (PFS) of 79%-81% in previously untreated FL, Dr Fowler said.
Phase 3 RELEVANCE trial (NCT01650701)
Investigators evaluated 1030 previously untreated grade 1-3a FL patients who required therapy.
Patients in the lenalidomide-rituximab group (n=513) received lenalidomide doses of 20 mg per day on days 2 to 22 and 28 for 6 to 12 cycles. Responders continued on therapy at 10 mg per day for a total of 18 cycles.
The rituximab dose was 375 mg/m2 weekly in cycle 1 and day 1 in cycles 2 to 6 and continued in responders for 12 additional cycles.
Patients in the R-chemo arm (n=517) received the investigator’s choice of standard rituximab-CHOP, rituximab-bendamustine, or rituximab-CVP, followed by 12 cycles of rituximab.
Most patients (72%) in the R-chemo arm received R-CHOP.
Baseline characteristics were similar in both groups, Dr Fowler said.
Co-primary endpoints were complete remission/complete remission unconfirmed (CR/Cru) at 120 weeks and PFS.
Results
At a median follow-up of 37.9 months, the superiority for lenalidomide and rituximab over rituximab-chemotherapy was not established.
For the lenalidomide-rituximab patients, the CR/Cru was 48% and 3-year PFS was 77% as compared to 53% and 78%, respectively, for rituximab-chemotherapy patients, as assessed by an independent review committee.
Overall survival was 94% in both groups.
Safety
“Important differences in safety profiles were observed between the arms,” Dr Fowler said.
Rituximab-chemotherapy patients had more frequent neutropenia, febrile neutropenia, growth factor usage, nausea, vomiting, neuropathy, and alopecia.
Lenalidomide and rituximab showed more cutaneous reactions, tumor flare, and diarrhea.
Toxicity profiles differed, with higher grade 4 neutropenia (31% vs 8%) and febrile neutropenia (7% vs 2%) with rituximab-chemotherapy compared with lenalidomide-rituximab, respectively.
More patients experienced grade 3/4 cutaneous events (7% vs 1%) with lenalidomide-rituximab.
Second primary malignancies were slightly higher with rituximab-chemotherapy (10%) than with lenalidomide-rituximab (7%). Grade 5 adverse events were 1% in both groups.
About 70% of patients completed treatment in both groups.
“Lenalidomide and rituximab was not superior to rituximab-chemotherapy based on mature CR/Cru at 120 weeks and interim PFS,” Dr Fowler said. “Both treatments showed similar efficacy results. Treatment effects on PFS were consistent across pre-specified subgroups.”
Dr Fowler presented data as of May 31, 2017. Continued follow-up on PFS and OS is ongoing.
The study is sponsored by Celgene Corporation and the Lymphoma Academic Research Organisation (LYSARC).
CHICAGO—A chemotherapy-free combination of lenalidomide plus rituximab shows similar efficacy and a different safety profile to chemotherapy plus rituximab (R-chemo) followed by rituximab maintenance in patients with previously untreated follicular lymphoma (FL).
According to investigators, the multicenter, international phase 3 RELEVANCE trial is the first to evaluate the chemo-free combination against the standard of care, R-chemo with rituximab maintenance.
“These results show that lenalidomide plus rituximab, a novel immunomodulatory approach, is a potential first-line option for patients with FL requiring treatment,” said investigator Nathan H. Fowler, MD, of the University of Texas MD Anderson Cancer Center in Houston.
Dr Fowler presented the results of the study at the 2018 ASCO Annual Meeting (abstract 7500).
The current standard of care in previously untreated symptomatic FL is immunochemotherapy induction followed by rituximab maintenance.
The immunomodulatory agent lenalidomide has complementary mechanisms with rituximab. Phase 2 studies of combined immunotherapy with lenalidomide and rituximab demonstrated 3-year progression-free survival (PFS) of 79%-81% in previously untreated FL, Dr Fowler said.
Phase 3 RELEVANCE trial (NCT01650701)
Investigators evaluated 1030 previously untreated grade 1-3a FL patients who required therapy.
Patients in the lenalidomide-rituximab group (n=513) received lenalidomide doses of 20 mg per day on days 2 to 22 and 28 for 6 to 12 cycles. Responders continued on therapy at 10 mg per day for a total of 18 cycles.
The rituximab dose was 375 mg/m2 weekly in cycle 1 and day 1 in cycles 2 to 6 and continued in responders for 12 additional cycles.
Patients in the R-chemo arm (n=517) received the investigator’s choice of standard rituximab-CHOP, rituximab-bendamustine, or rituximab-CVP, followed by 12 cycles of rituximab.
Most patients (72%) in the R-chemo arm received R-CHOP.
Baseline characteristics were similar in both groups, Dr Fowler said.
Co-primary endpoints were complete remission/complete remission unconfirmed (CR/Cru) at 120 weeks and PFS.
Results
At a median follow-up of 37.9 months, the superiority for lenalidomide and rituximab over rituximab-chemotherapy was not established.
For the lenalidomide-rituximab patients, the CR/Cru was 48% and 3-year PFS was 77% as compared to 53% and 78%, respectively, for rituximab-chemotherapy patients, as assessed by an independent review committee.
Overall survival was 94% in both groups.
Safety
“Important differences in safety profiles were observed between the arms,” Dr Fowler said.
Rituximab-chemotherapy patients had more frequent neutropenia, febrile neutropenia, growth factor usage, nausea, vomiting, neuropathy, and alopecia.
Lenalidomide and rituximab showed more cutaneous reactions, tumor flare, and diarrhea.
Toxicity profiles differed, with higher grade 4 neutropenia (31% vs 8%) and febrile neutropenia (7% vs 2%) with rituximab-chemotherapy compared with lenalidomide-rituximab, respectively.
More patients experienced grade 3/4 cutaneous events (7% vs 1%) with lenalidomide-rituximab.
Second primary malignancies were slightly higher with rituximab-chemotherapy (10%) than with lenalidomide-rituximab (7%). Grade 5 adverse events were 1% in both groups.
About 70% of patients completed treatment in both groups.
“Lenalidomide and rituximab was not superior to rituximab-chemotherapy based on mature CR/Cru at 120 weeks and interim PFS,” Dr Fowler said. “Both treatments showed similar efficacy results. Treatment effects on PFS were consistent across pre-specified subgroups.”
Dr Fowler presented data as of May 31, 2017. Continued follow-up on PFS and OS is ongoing.
The study is sponsored by Celgene Corporation and the Lymphoma Academic Research Organisation (LYSARC).
FDA grants regular approval to venetoclax for CLL/SLL
Venetoclax (Venclexta) has received regular approval from the Food and Drug Administration for the treatment of patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL), with or without 17p deletion, who have received at least one prior therapy.
The approval was based results from the MURANO trial of 389 patients, which was a randomized, multicenter, open-label trial of venetoclax plus rituximab versus bendamustine plus rituximab.
Neutropenia, diarrhea, upper respiratory tract infection, fatigue, cough, and nausea were the most common adverse events seen in the venetoclax arm. Grade 3 or 4 neutropenia developed in 64% of those patients, and grade 4 in 31%. The most common infection in venetoclax patients was pneumonia, but overall, 21% of patients in that arm experienced some kind of infection.
Because of the rapid reduction in tumor size, tumor lysis syndrome is possible with venetoclax treatment, the FDA noted.
In 2016, the FDA granted accelerated approval to venetoclax for treatment of patients with CLL with 17d deletion who had received at least one prior line of therapy.
Venetoclax (Venclexta) has received regular approval from the Food and Drug Administration for the treatment of patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL), with or without 17p deletion, who have received at least one prior therapy.
The approval was based results from the MURANO trial of 389 patients, which was a randomized, multicenter, open-label trial of venetoclax plus rituximab versus bendamustine plus rituximab.
Neutropenia, diarrhea, upper respiratory tract infection, fatigue, cough, and nausea were the most common adverse events seen in the venetoclax arm. Grade 3 or 4 neutropenia developed in 64% of those patients, and grade 4 in 31%. The most common infection in venetoclax patients was pneumonia, but overall, 21% of patients in that arm experienced some kind of infection.
Because of the rapid reduction in tumor size, tumor lysis syndrome is possible with venetoclax treatment, the FDA noted.
In 2016, the FDA granted accelerated approval to venetoclax for treatment of patients with CLL with 17d deletion who had received at least one prior line of therapy.
Venetoclax (Venclexta) has received regular approval from the Food and Drug Administration for the treatment of patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL), with or without 17p deletion, who have received at least one prior therapy.
The approval was based results from the MURANO trial of 389 patients, which was a randomized, multicenter, open-label trial of venetoclax plus rituximab versus bendamustine plus rituximab.
Neutropenia, diarrhea, upper respiratory tract infection, fatigue, cough, and nausea were the most common adverse events seen in the venetoclax arm. Grade 3 or 4 neutropenia developed in 64% of those patients, and grade 4 in 31%. The most common infection in venetoclax patients was pneumonia, but overall, 21% of patients in that arm experienced some kind of infection.
Because of the rapid reduction in tumor size, tumor lysis syndrome is possible with venetoclax treatment, the FDA noted.
In 2016, the FDA granted accelerated approval to venetoclax for treatment of patients with CLL with 17d deletion who had received at least one prior line of therapy.
Venetoclax plus ibrutinib yields encouraging MRD results in first-line CLL
CHICAGO – The combination of ibrutinib plus venetoclax yielded a high rate of undetectable minimal residual disease (MRD) when used as first-line treatment for chronic lymphocytic leukemia (CLL), according to preliminary results of the CAPTIVATE trial.
Of the first 30 patients in the trial, 23 (77%) had undetectable blood MRD after just six cycles of combined treatment, said investigator William G. Wierda, MD, PhD, of the University of Texas MD Anderson Cancer Center, Houston.
“These early results show a highly active and safe treatment with 12 cycles of combined treatment with ibrutinib and venetoclax,” Dr. Wierda said in a presentation of the CAPTIVATE results at the annual meeting of the American Society of Clinical Oncology.
Those MRD results are “at least as good as we can achieve with chemoimmunotherapy,” Bruce D. Cheson, MD, head of hematology at Georgetown University, Washington, said during a discussion of the CAPTIVATE study results.
Dr. Cheson referenced MRD results from a 2016 analysis of the CLL8 and CLL10 trials, which included patients treated with fludarabine, cyclophosphamide, and rituximab (FCR) and bendamustine plus rituximab (BR). In that analysis, 33.6% of patients achieved MRD-negative complete response and 29.1% achieved MRD-negative partial response.
In CAPTIVATE, by contrast, all of the complete remissions were MRD negative, as were a majority of the partial responders, Dr. Cheson noted.
Venetoclax and ibrutinib have “clinically complimentary activity” that provided a rationale for combining the two, Dr. Wierda said at ASCO. Ibrutinib is a BTK inhibitor that has a high rate of response and durable disease control, though continuous treatment is indicated, he said, because most patients achieve partial remissions as best response and continue to have residual disease in blood or bone marrow. Venetoclax, he added, is a BCL-2 inhibitor that produces durable partial remissions, though “residual disease is typically present in the form of persistently enlarged lymph nodes,” he said. “Venetoclax is highly effective at clearing disease from blood and bone marrow.”
The phase 2 CAPTIVATE trial includes a total of 164 patients younger than 70 years of age who receive a 3-cycle ibrutinib lead-in, followed by ibrutinib plus venetoclax for 12 cycles. At that point, patients are randomized according to MRD status. Patients with confirmed undetectable MRD are randomized to further treatment with ibrutinib or placebo, and those with undetectable MRD not confirmed are randomized to ibrutinib versus ibrutinib plus venetoclax.
In addition to early efficacy data, Dr. Wierda also reported some safety data. Compared with the single-agent ibrutinib lead-in period, combined ibrutinib plus venetoclax treatment had more gastrointestinal-associated events and neutropenia. Almost half of patients (45%) have had a treatment-related grade 3-4 adverse event, though just 18 (11%) have had treatment-related adverse events classified as serious, and there have been no adverse event-related deaths on study.
The high activity of ibrutinib plus venetoclax in CAPTIVATE supports further study of the combination, Dr. Wierda said. A randomized, open-label phase 3 trial of ibrutinib plus venetoclax versus chlorambucil plus obinutuzumab as first-line treatment for CLL is currently recruiting.
The study was sponsored by Pharmacyclics, an AbbVie company. Dr. Wierda reported consulting and research funding from Pharmacyclics, AbbVie, and several other companies.
SOURCE: Wierda WG et al. ASCO 2018, Abstract 7502.
CHICAGO – The combination of ibrutinib plus venetoclax yielded a high rate of undetectable minimal residual disease (MRD) when used as first-line treatment for chronic lymphocytic leukemia (CLL), according to preliminary results of the CAPTIVATE trial.
Of the first 30 patients in the trial, 23 (77%) had undetectable blood MRD after just six cycles of combined treatment, said investigator William G. Wierda, MD, PhD, of the University of Texas MD Anderson Cancer Center, Houston.
“These early results show a highly active and safe treatment with 12 cycles of combined treatment with ibrutinib and venetoclax,” Dr. Wierda said in a presentation of the CAPTIVATE results at the annual meeting of the American Society of Clinical Oncology.
Those MRD results are “at least as good as we can achieve with chemoimmunotherapy,” Bruce D. Cheson, MD, head of hematology at Georgetown University, Washington, said during a discussion of the CAPTIVATE study results.
Dr. Cheson referenced MRD results from a 2016 analysis of the CLL8 and CLL10 trials, which included patients treated with fludarabine, cyclophosphamide, and rituximab (FCR) and bendamustine plus rituximab (BR). In that analysis, 33.6% of patients achieved MRD-negative complete response and 29.1% achieved MRD-negative partial response.
In CAPTIVATE, by contrast, all of the complete remissions were MRD negative, as were a majority of the partial responders, Dr. Cheson noted.
Venetoclax and ibrutinib have “clinically complimentary activity” that provided a rationale for combining the two, Dr. Wierda said at ASCO. Ibrutinib is a BTK inhibitor that has a high rate of response and durable disease control, though continuous treatment is indicated, he said, because most patients achieve partial remissions as best response and continue to have residual disease in blood or bone marrow. Venetoclax, he added, is a BCL-2 inhibitor that produces durable partial remissions, though “residual disease is typically present in the form of persistently enlarged lymph nodes,” he said. “Venetoclax is highly effective at clearing disease from blood and bone marrow.”
The phase 2 CAPTIVATE trial includes a total of 164 patients younger than 70 years of age who receive a 3-cycle ibrutinib lead-in, followed by ibrutinib plus venetoclax for 12 cycles. At that point, patients are randomized according to MRD status. Patients with confirmed undetectable MRD are randomized to further treatment with ibrutinib or placebo, and those with undetectable MRD not confirmed are randomized to ibrutinib versus ibrutinib plus venetoclax.
In addition to early efficacy data, Dr. Wierda also reported some safety data. Compared with the single-agent ibrutinib lead-in period, combined ibrutinib plus venetoclax treatment had more gastrointestinal-associated events and neutropenia. Almost half of patients (45%) have had a treatment-related grade 3-4 adverse event, though just 18 (11%) have had treatment-related adverse events classified as serious, and there have been no adverse event-related deaths on study.
The high activity of ibrutinib plus venetoclax in CAPTIVATE supports further study of the combination, Dr. Wierda said. A randomized, open-label phase 3 trial of ibrutinib plus venetoclax versus chlorambucil plus obinutuzumab as first-line treatment for CLL is currently recruiting.
The study was sponsored by Pharmacyclics, an AbbVie company. Dr. Wierda reported consulting and research funding from Pharmacyclics, AbbVie, and several other companies.
SOURCE: Wierda WG et al. ASCO 2018, Abstract 7502.
CHICAGO – The combination of ibrutinib plus venetoclax yielded a high rate of undetectable minimal residual disease (MRD) when used as first-line treatment for chronic lymphocytic leukemia (CLL), according to preliminary results of the CAPTIVATE trial.
Of the first 30 patients in the trial, 23 (77%) had undetectable blood MRD after just six cycles of combined treatment, said investigator William G. Wierda, MD, PhD, of the University of Texas MD Anderson Cancer Center, Houston.
“These early results show a highly active and safe treatment with 12 cycles of combined treatment with ibrutinib and venetoclax,” Dr. Wierda said in a presentation of the CAPTIVATE results at the annual meeting of the American Society of Clinical Oncology.
Those MRD results are “at least as good as we can achieve with chemoimmunotherapy,” Bruce D. Cheson, MD, head of hematology at Georgetown University, Washington, said during a discussion of the CAPTIVATE study results.
Dr. Cheson referenced MRD results from a 2016 analysis of the CLL8 and CLL10 trials, which included patients treated with fludarabine, cyclophosphamide, and rituximab (FCR) and bendamustine plus rituximab (BR). In that analysis, 33.6% of patients achieved MRD-negative complete response and 29.1% achieved MRD-negative partial response.
In CAPTIVATE, by contrast, all of the complete remissions were MRD negative, as were a majority of the partial responders, Dr. Cheson noted.
Venetoclax and ibrutinib have “clinically complimentary activity” that provided a rationale for combining the two, Dr. Wierda said at ASCO. Ibrutinib is a BTK inhibitor that has a high rate of response and durable disease control, though continuous treatment is indicated, he said, because most patients achieve partial remissions as best response and continue to have residual disease in blood or bone marrow. Venetoclax, he added, is a BCL-2 inhibitor that produces durable partial remissions, though “residual disease is typically present in the form of persistently enlarged lymph nodes,” he said. “Venetoclax is highly effective at clearing disease from blood and bone marrow.”
The phase 2 CAPTIVATE trial includes a total of 164 patients younger than 70 years of age who receive a 3-cycle ibrutinib lead-in, followed by ibrutinib plus venetoclax for 12 cycles. At that point, patients are randomized according to MRD status. Patients with confirmed undetectable MRD are randomized to further treatment with ibrutinib or placebo, and those with undetectable MRD not confirmed are randomized to ibrutinib versus ibrutinib plus venetoclax.
In addition to early efficacy data, Dr. Wierda also reported some safety data. Compared with the single-agent ibrutinib lead-in period, combined ibrutinib plus venetoclax treatment had more gastrointestinal-associated events and neutropenia. Almost half of patients (45%) have had a treatment-related grade 3-4 adverse event, though just 18 (11%) have had treatment-related adverse events classified as serious, and there have been no adverse event-related deaths on study.
The high activity of ibrutinib plus venetoclax in CAPTIVATE supports further study of the combination, Dr. Wierda said. A randomized, open-label phase 3 trial of ibrutinib plus venetoclax versus chlorambucil plus obinutuzumab as first-line treatment for CLL is currently recruiting.
The study was sponsored by Pharmacyclics, an AbbVie company. Dr. Wierda reported consulting and research funding from Pharmacyclics, AbbVie, and several other companies.
SOURCE: Wierda WG et al. ASCO 2018, Abstract 7502.
REPORTING FROM ASCO 2018
Key clinical point:
Major finding: Of 14 patients, 12 (86%) who completed 12 cycles of treatment had undetectable bone marrow MRD.
Study details: Early results of the phase 2 CAPTIVATE trial including 164 patients younger than 70 years of age with previously untreated CLL.
Disclosures: The study was sponsored by Pharmacyclics, an Abbvie company. Dr. Wierda reported consulting and research funding from Pharmacyclics, AbbVie, and several other companies.
Source: Wierda WG et al. ASCO 2018, Abstract 7502.
Polatuzumab plus BR improves efficacy in DLBCL
CHICAGO—Polatuzumab vedotin, when added to bendamustine (B) and rituximab (R), significantly improved response and survival rates in a cohort of patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL), according to a phase 2 study.
By contrast, there were no such improvements in a cohort of follicular lymphoma (FL) patients, at least in short-term follow-up, investigator Laurie Helen Sehn, MD, of the BC Cancer Agency in Vancouver, Canada, said at the 2018 ASCO Annual Meeting.
However, the improvement in overall survival in DLBCL patients is “remarkable,” Dr Sehn affirmed in an oral presentation (abstract 7507).
“Based on these encouraging results, polatuzumab vedotin has received breakthrough therapy designation and priority medicines designation by the FDA and EMA for patients with relapsed or refractory DLBCL,” she said.
Polatuzumab-BR study (NCT02257567)
The study by Dr Sehn and colleagues included a cohort of 80 DLBCL patients randomized to BR or polatuzumab-BR for 6 planned 21-day cycles.
Investigators randomized another cohort of 80 FL patients to BR or polatuzumab-BR for 6 planned 28-day cycles.
The primary endpoint was complete response (CR) assessed by fluorodeoxyglucose positron emission tomography (FDG-PET) at 6 to 8 weeks after the end of treatment.
DLBCL patients
A total of 40% of polatuzumab-BR-treated DLBCL patients achieved CR at the end of treatment, versus 15% of BR-treated patients (P=0.012).
That CR improvement translated into a significantly higher progression-free survival (PFS) (6.7 months for polatuzumab-BR vs 2.0 months for BR, P<0.0001) and overall survival (11.8 months versus 4.7 months, P=0.0008), according to Dr Sehn.
The FDG-PET CR rates were higher in the polatuzumab-BR arm regardless of the number of prior lines of treatment for DLBCL, and regardless of relapsed versus refractory status, Dr. Sehn added.
FL patients
By contrast, in the FL cohort, the FDG-PET CR rate was high for both arms, at 69% for polatuzumab-BR and 63% for BR.
And there was no significant difference in progression-free survival (P=0.58) with “relatively short-term follow-up,” she said.
Adverse events
The most common grades 3 – 5 adverse events for both DLBCL and FL patients were higher in the polatuzumab-BR arm than the BR arm and included cytopenias, febrile neutropenia, and infections.
Serious AEs were also higher in the polatuzumab-BR arm and included febrile neutropenia for both FL and DLBCL patients and infection for FL patients.
Five percent of FL patients and 18% of DLBCL had a grade 5 event.
Commentary
Whether polatuzumab vedotin will change treatment paradigms for DLBCL patients may be answered by the ongoing POLARIX study, according to Alison Moskowitz, MD, of Memorial Sloan Kettering Cancer Center in New York, NY.
The randomized phase 3 POLARIX study (abstract TPS7589) is comparing polatuzumab plus R-CHP to R-CHOP in patients with previously untreated DLBCL.
“Certainly, there are patients who do very well with R-CHOP chemotherapy alone, and so we need to learn whether this is necessary for all patients, or only the high-risk patients,” Dr Moskowitz said in a talk at ASCO commenting on the results of the polatuzumab-BR study.
Hoffman-LaRoche is the sponsor of the study.
CHICAGO—Polatuzumab vedotin, when added to bendamustine (B) and rituximab (R), significantly improved response and survival rates in a cohort of patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL), according to a phase 2 study.
By contrast, there were no such improvements in a cohort of follicular lymphoma (FL) patients, at least in short-term follow-up, investigator Laurie Helen Sehn, MD, of the BC Cancer Agency in Vancouver, Canada, said at the 2018 ASCO Annual Meeting.
However, the improvement in overall survival in DLBCL patients is “remarkable,” Dr Sehn affirmed in an oral presentation (abstract 7507).
“Based on these encouraging results, polatuzumab vedotin has received breakthrough therapy designation and priority medicines designation by the FDA and EMA for patients with relapsed or refractory DLBCL,” she said.
Polatuzumab-BR study (NCT02257567)
The study by Dr Sehn and colleagues included a cohort of 80 DLBCL patients randomized to BR or polatuzumab-BR for 6 planned 21-day cycles.
Investigators randomized another cohort of 80 FL patients to BR or polatuzumab-BR for 6 planned 28-day cycles.
The primary endpoint was complete response (CR) assessed by fluorodeoxyglucose positron emission tomography (FDG-PET) at 6 to 8 weeks after the end of treatment.
DLBCL patients
A total of 40% of polatuzumab-BR-treated DLBCL patients achieved CR at the end of treatment, versus 15% of BR-treated patients (P=0.012).
That CR improvement translated into a significantly higher progression-free survival (PFS) (6.7 months for polatuzumab-BR vs 2.0 months for BR, P<0.0001) and overall survival (11.8 months versus 4.7 months, P=0.0008), according to Dr Sehn.
The FDG-PET CR rates were higher in the polatuzumab-BR arm regardless of the number of prior lines of treatment for DLBCL, and regardless of relapsed versus refractory status, Dr. Sehn added.
FL patients
By contrast, in the FL cohort, the FDG-PET CR rate was high for both arms, at 69% for polatuzumab-BR and 63% for BR.
And there was no significant difference in progression-free survival (P=0.58) with “relatively short-term follow-up,” she said.
Adverse events
The most common grades 3 – 5 adverse events for both DLBCL and FL patients were higher in the polatuzumab-BR arm than the BR arm and included cytopenias, febrile neutropenia, and infections.
Serious AEs were also higher in the polatuzumab-BR arm and included febrile neutropenia for both FL and DLBCL patients and infection for FL patients.
Five percent of FL patients and 18% of DLBCL had a grade 5 event.
Commentary
Whether polatuzumab vedotin will change treatment paradigms for DLBCL patients may be answered by the ongoing POLARIX study, according to Alison Moskowitz, MD, of Memorial Sloan Kettering Cancer Center in New York, NY.
The randomized phase 3 POLARIX study (abstract TPS7589) is comparing polatuzumab plus R-CHP to R-CHOP in patients with previously untreated DLBCL.
“Certainly, there are patients who do very well with R-CHOP chemotherapy alone, and so we need to learn whether this is necessary for all patients, or only the high-risk patients,” Dr Moskowitz said in a talk at ASCO commenting on the results of the polatuzumab-BR study.
Hoffman-LaRoche is the sponsor of the study.
CHICAGO—Polatuzumab vedotin, when added to bendamustine (B) and rituximab (R), significantly improved response and survival rates in a cohort of patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL), according to a phase 2 study.
By contrast, there were no such improvements in a cohort of follicular lymphoma (FL) patients, at least in short-term follow-up, investigator Laurie Helen Sehn, MD, of the BC Cancer Agency in Vancouver, Canada, said at the 2018 ASCO Annual Meeting.
However, the improvement in overall survival in DLBCL patients is “remarkable,” Dr Sehn affirmed in an oral presentation (abstract 7507).
“Based on these encouraging results, polatuzumab vedotin has received breakthrough therapy designation and priority medicines designation by the FDA and EMA for patients with relapsed or refractory DLBCL,” she said.
Polatuzumab-BR study (NCT02257567)
The study by Dr Sehn and colleagues included a cohort of 80 DLBCL patients randomized to BR or polatuzumab-BR for 6 planned 21-day cycles.
Investigators randomized another cohort of 80 FL patients to BR or polatuzumab-BR for 6 planned 28-day cycles.
The primary endpoint was complete response (CR) assessed by fluorodeoxyglucose positron emission tomography (FDG-PET) at 6 to 8 weeks after the end of treatment.
DLBCL patients
A total of 40% of polatuzumab-BR-treated DLBCL patients achieved CR at the end of treatment, versus 15% of BR-treated patients (P=0.012).
That CR improvement translated into a significantly higher progression-free survival (PFS) (6.7 months for polatuzumab-BR vs 2.0 months for BR, P<0.0001) and overall survival (11.8 months versus 4.7 months, P=0.0008), according to Dr Sehn.
The FDG-PET CR rates were higher in the polatuzumab-BR arm regardless of the number of prior lines of treatment for DLBCL, and regardless of relapsed versus refractory status, Dr. Sehn added.
FL patients
By contrast, in the FL cohort, the FDG-PET CR rate was high for both arms, at 69% for polatuzumab-BR and 63% for BR.
And there was no significant difference in progression-free survival (P=0.58) with “relatively short-term follow-up,” she said.
Adverse events
The most common grades 3 – 5 adverse events for both DLBCL and FL patients were higher in the polatuzumab-BR arm than the BR arm and included cytopenias, febrile neutropenia, and infections.
Serious AEs were also higher in the polatuzumab-BR arm and included febrile neutropenia for both FL and DLBCL patients and infection for FL patients.
Five percent of FL patients and 18% of DLBCL had a grade 5 event.
Commentary
Whether polatuzumab vedotin will change treatment paradigms for DLBCL patients may be answered by the ongoing POLARIX study, according to Alison Moskowitz, MD, of Memorial Sloan Kettering Cancer Center in New York, NY.
The randomized phase 3 POLARIX study (abstract TPS7589) is comparing polatuzumab plus R-CHP to R-CHOP in patients with previously untreated DLBCL.
“Certainly, there are patients who do very well with R-CHOP chemotherapy alone, and so we need to learn whether this is necessary for all patients, or only the high-risk patients,” Dr Moskowitz said in a talk at ASCO commenting on the results of the polatuzumab-BR study.
Hoffman-LaRoche is the sponsor of the study.
Chemo-free regimen appears viable in previously untreated FL
CHICAGO – Lenalidomide plus rituximab (R2) had comparable efficacy versus standard chemoimmunotherapy in patients with previously untreated follicular lymphoma, according to results from a phase 3 trial.
RELEVANCE is the first randomized, phase 3 trial to examine a chemotherapy-free regimen in this setting.
Response and progression-free survival (PFS) results were similar for patients who received R2 followed by rituximab maintenance and patients assigned to chemotherapy plus rituximab and rituximab maintenance, in study results presented at the annual meeting of the American Society of Clinical Oncology.
“These results show that lenalidomide plus rituximab, which is a novel immunomodulatory approach, is a potential first-line option for patients with follicular lymphoma that require treatment,” said investigator Nathan H. Fowler, MD, of the University of Texas MD Anderson Cancer Center, Houston.
But since the study was designed as a superiority trial, rather than a noninferiority trial, and it failed to meet its primary endpoint of superior complete remission (CR) or CR unconfirmed (CRu) at 120 weeks, said Bruce D. Cheson, MD, head of hematology at Georgetown University, Washington.
R2 had a similar PFS overall and in all major patient subgroups, similar overall survival, less nonhematologic toxicity aside from rash, less neutropenia, and fewer infections despite increased use of growth factors in the chemoimmunotherapy arm, Dr. Cheson said in a presentation commenting on the results. “Therefore, I agree with Dr. Fowler’s conclusion that R2 can be considered as an option for the front-line therapy of patients with follicular lymphoma,” Dr. Cheson said.
The RELEVANCE study included 1,030 patients (median age, 59 years) with previously untreated, advanced follicular lymphoma requiring treatment. They were randomized 1:1 to either lenalidomide plus rituximab followed by rituximab maintenance, or R-chemotherapy followed by rituximab maintenance.
For patients randomly assigned to R-chemotherapy, physicians could choose among three standard regimens: rituximab plus bendamustine (R-B), rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), or rituximab plus cyclophosphamide, vincristine, and prednisone (R-CVP).
There was no statistical difference between treatment approaches in CR/CRu at 120 weeks, which was 48% in the R2 arm and 53% in the R-chemotherapy arm (P = 0.13). Best CR/CRu also was not statistically different between arms (59% and 67%, respectively), as was best overall response rate (84% and 89%). The 3-year duration of response was 77% in the R2 arm and 74% for R-chemotherapy.
With 37.9 months median follow-up, progression-free survival was “nearly identical” between the two groups, Dr. Fowler said, at 77% for R2 and 78% for R-chemotherapy (P = 0.48). The 3-year overall survival was 94% in both the R2 and R-chemotherapy arms, though survival data are still immature, Dr. Fowler noted.
Grade 3/4 neutropenia was more common in the R-chemotherapy arm, resulting in higher rates of febrile neutropenia, according to Dr. Fowler, who also noted that rash and cutaneous reactions were more common with R2. About 70% of patients in each arm were able to tolerate treatment, and reasons for discontinuation were “fairly similar” between arms, Dr. Fowler added.
Second primary malignancies occurred in 7% of patients in the R2 arm and 10% of the R-chemotherapy arm.
The study was sponsored was Celgene and the Lymphoma Academic Research Organisation. Dr. Fowler reported disclosures related to Abbvie, Celgene, Janssen, Merck, and Roche.
SOURCE: Fowler NH et al. ASCO 2018, Abstract 7500.
CHICAGO – Lenalidomide plus rituximab (R2) had comparable efficacy versus standard chemoimmunotherapy in patients with previously untreated follicular lymphoma, according to results from a phase 3 trial.
RELEVANCE is the first randomized, phase 3 trial to examine a chemotherapy-free regimen in this setting.
Response and progression-free survival (PFS) results were similar for patients who received R2 followed by rituximab maintenance and patients assigned to chemotherapy plus rituximab and rituximab maintenance, in study results presented at the annual meeting of the American Society of Clinical Oncology.
“These results show that lenalidomide plus rituximab, which is a novel immunomodulatory approach, is a potential first-line option for patients with follicular lymphoma that require treatment,” said investigator Nathan H. Fowler, MD, of the University of Texas MD Anderson Cancer Center, Houston.
But since the study was designed as a superiority trial, rather than a noninferiority trial, and it failed to meet its primary endpoint of superior complete remission (CR) or CR unconfirmed (CRu) at 120 weeks, said Bruce D. Cheson, MD, head of hematology at Georgetown University, Washington.
R2 had a similar PFS overall and in all major patient subgroups, similar overall survival, less nonhematologic toxicity aside from rash, less neutropenia, and fewer infections despite increased use of growth factors in the chemoimmunotherapy arm, Dr. Cheson said in a presentation commenting on the results. “Therefore, I agree with Dr. Fowler’s conclusion that R2 can be considered as an option for the front-line therapy of patients with follicular lymphoma,” Dr. Cheson said.
The RELEVANCE study included 1,030 patients (median age, 59 years) with previously untreated, advanced follicular lymphoma requiring treatment. They were randomized 1:1 to either lenalidomide plus rituximab followed by rituximab maintenance, or R-chemotherapy followed by rituximab maintenance.
For patients randomly assigned to R-chemotherapy, physicians could choose among three standard regimens: rituximab plus bendamustine (R-B), rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), or rituximab plus cyclophosphamide, vincristine, and prednisone (R-CVP).
There was no statistical difference between treatment approaches in CR/CRu at 120 weeks, which was 48% in the R2 arm and 53% in the R-chemotherapy arm (P = 0.13). Best CR/CRu also was not statistically different between arms (59% and 67%, respectively), as was best overall response rate (84% and 89%). The 3-year duration of response was 77% in the R2 arm and 74% for R-chemotherapy.
With 37.9 months median follow-up, progression-free survival was “nearly identical” between the two groups, Dr. Fowler said, at 77% for R2 and 78% for R-chemotherapy (P = 0.48). The 3-year overall survival was 94% in both the R2 and R-chemotherapy arms, though survival data are still immature, Dr. Fowler noted.
Grade 3/4 neutropenia was more common in the R-chemotherapy arm, resulting in higher rates of febrile neutropenia, according to Dr. Fowler, who also noted that rash and cutaneous reactions were more common with R2. About 70% of patients in each arm were able to tolerate treatment, and reasons for discontinuation were “fairly similar” between arms, Dr. Fowler added.
Second primary malignancies occurred in 7% of patients in the R2 arm and 10% of the R-chemotherapy arm.
The study was sponsored was Celgene and the Lymphoma Academic Research Organisation. Dr. Fowler reported disclosures related to Abbvie, Celgene, Janssen, Merck, and Roche.
SOURCE: Fowler NH et al. ASCO 2018, Abstract 7500.
CHICAGO – Lenalidomide plus rituximab (R2) had comparable efficacy versus standard chemoimmunotherapy in patients with previously untreated follicular lymphoma, according to results from a phase 3 trial.
RELEVANCE is the first randomized, phase 3 trial to examine a chemotherapy-free regimen in this setting.
Response and progression-free survival (PFS) results were similar for patients who received R2 followed by rituximab maintenance and patients assigned to chemotherapy plus rituximab and rituximab maintenance, in study results presented at the annual meeting of the American Society of Clinical Oncology.
“These results show that lenalidomide plus rituximab, which is a novel immunomodulatory approach, is a potential first-line option for patients with follicular lymphoma that require treatment,” said investigator Nathan H. Fowler, MD, of the University of Texas MD Anderson Cancer Center, Houston.
But since the study was designed as a superiority trial, rather than a noninferiority trial, and it failed to meet its primary endpoint of superior complete remission (CR) or CR unconfirmed (CRu) at 120 weeks, said Bruce D. Cheson, MD, head of hematology at Georgetown University, Washington.
R2 had a similar PFS overall and in all major patient subgroups, similar overall survival, less nonhematologic toxicity aside from rash, less neutropenia, and fewer infections despite increased use of growth factors in the chemoimmunotherapy arm, Dr. Cheson said in a presentation commenting on the results. “Therefore, I agree with Dr. Fowler’s conclusion that R2 can be considered as an option for the front-line therapy of patients with follicular lymphoma,” Dr. Cheson said.
The RELEVANCE study included 1,030 patients (median age, 59 years) with previously untreated, advanced follicular lymphoma requiring treatment. They were randomized 1:1 to either lenalidomide plus rituximab followed by rituximab maintenance, or R-chemotherapy followed by rituximab maintenance.
For patients randomly assigned to R-chemotherapy, physicians could choose among three standard regimens: rituximab plus bendamustine (R-B), rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), or rituximab plus cyclophosphamide, vincristine, and prednisone (R-CVP).
There was no statistical difference between treatment approaches in CR/CRu at 120 weeks, which was 48% in the R2 arm and 53% in the R-chemotherapy arm (P = 0.13). Best CR/CRu also was not statistically different between arms (59% and 67%, respectively), as was best overall response rate (84% and 89%). The 3-year duration of response was 77% in the R2 arm and 74% for R-chemotherapy.
With 37.9 months median follow-up, progression-free survival was “nearly identical” between the two groups, Dr. Fowler said, at 77% for R2 and 78% for R-chemotherapy (P = 0.48). The 3-year overall survival was 94% in both the R2 and R-chemotherapy arms, though survival data are still immature, Dr. Fowler noted.
Grade 3/4 neutropenia was more common in the R-chemotherapy arm, resulting in higher rates of febrile neutropenia, according to Dr. Fowler, who also noted that rash and cutaneous reactions were more common with R2. About 70% of patients in each arm were able to tolerate treatment, and reasons for discontinuation were “fairly similar” between arms, Dr. Fowler added.
Second primary malignancies occurred in 7% of patients in the R2 arm and 10% of the R-chemotherapy arm.
The study was sponsored was Celgene and the Lymphoma Academic Research Organisation. Dr. Fowler reported disclosures related to Abbvie, Celgene, Janssen, Merck, and Roche.
SOURCE: Fowler NH et al. ASCO 2018, Abstract 7500.
REPORTING FROM ASCO 2018
Key clinical point: in patients with previously untreated follicular lymphoma.
Major finding: With 37.9 months’ median follow-up, progression-free survival was “nearly identical” between the two groups, at 77% for R2 and 78% for rituximab chemotherapy (P = 0.48).
Study details: RELEVANCE, a phase 3, randomized clinical trial including 1,030 patients with previously untreated, advanced follicular lymphoma requiring treatment.
Disclosures: The study was sponsored was Celgene and the Lymphoma Academic Research Organisation. Dr. Fowler reported disclosures related to AbbVie, Celgene, Janssen, Merck, and Roche.
Source: Fowler NH et al. ASCO 2018, Abstract 7500.