User login
ACOG: Salpingectomy offers chance to prevent ovarian cancer
Salpingectomy may offer clinicians the chance to prevent ovarian cancer, according to a policy statement from the American College of Obstetricians and Gynecologists’ Committee on Gynecologic Practice, published in Obstetrics and Gynecology on Dec. 22.
Ovarian cancer carries the highest mortality of any gynecologic cancer and is the fifth-leading cause of cancer deaths among women. Overall survival has improved only marginally in the last 50 years, and current attempts at screening are not only unsuccessful but have led to unnecessary surgery and associated complications. The new ACOG committee opinion advises physicians to consider prophylactic salpingectomy as part of a prevention strategy.
The first step for physicians is to discuss salpingectomy with women who are scheduled to undergo routine pelvic surgery, such as tubal sterilization or hysterectomy without oophorectomy. Performing concurrent salpingectomy does not appear to increase complications, compared with tubal ligation or hysterectomy alone, and doesn’t appear to affect ovarian function.
“Given current theories of ovarian carcinogenesis, ovarian conservation and salpingectomy may represent a better option than [bilateral salpingo-oophorectomy] for ovarian cancer risk reduction for most women undergoing other pelvic surgeries for benign disease,” ACOG wrote in the committee opinion (Obstet. Gynecol. 2015;125:279-81).
But the ACOG committee advised physicians to discuss the role of oophorectomy and bilateral salpingo-oophorectomy as part the informed consent discussion.
Salpingectomy also should be discussed when counseling women about laparoscopic sterilization methods since it can be considered a method that provides effective contraception, according to the committee opinion.
Surgeons, however, should not change their approach to hysterectomy or sterilization just because of the “theoretical benefit of salpingectomy,” the committee wrote.
“A vaginal hysterectomy should not be changed to a laparoscopic hysterectomy simply to perform a salpingectomy,” ACOG wrote. “The choice of sterilization procedure should be based on the risks and benefits of the hysteroscopic and laparoscopic approaches.”
The opinion statement also noted that salpingectomy should be performed with “meticulous attention.” The entire fallopian tube should be removed, from the fimbriated end to the uterotubal junction, and any fimbrial attachments on the ovary should be cauterized or removed. “Care should be taken not to interrupt the blood supply to the ovary through the infundibulopelvic ligament because the collateral vasculature from the tubal mesosalpinx is occluded during tube removal.”
And in cases when complete salpingectomy cannot be accomplished, removing as much of the fallopian tubes as possible “still may have value,” ACOG wrote.
Salpingectomy may offer clinicians the chance to prevent ovarian cancer, according to a policy statement from the American College of Obstetricians and Gynecologists’ Committee on Gynecologic Practice, published in Obstetrics and Gynecology on Dec. 22.
Ovarian cancer carries the highest mortality of any gynecologic cancer and is the fifth-leading cause of cancer deaths among women. Overall survival has improved only marginally in the last 50 years, and current attempts at screening are not only unsuccessful but have led to unnecessary surgery and associated complications. The new ACOG committee opinion advises physicians to consider prophylactic salpingectomy as part of a prevention strategy.
The first step for physicians is to discuss salpingectomy with women who are scheduled to undergo routine pelvic surgery, such as tubal sterilization or hysterectomy without oophorectomy. Performing concurrent salpingectomy does not appear to increase complications, compared with tubal ligation or hysterectomy alone, and doesn’t appear to affect ovarian function.
“Given current theories of ovarian carcinogenesis, ovarian conservation and salpingectomy may represent a better option than [bilateral salpingo-oophorectomy] for ovarian cancer risk reduction for most women undergoing other pelvic surgeries for benign disease,” ACOG wrote in the committee opinion (Obstet. Gynecol. 2015;125:279-81).
But the ACOG committee advised physicians to discuss the role of oophorectomy and bilateral salpingo-oophorectomy as part the informed consent discussion.
Salpingectomy also should be discussed when counseling women about laparoscopic sterilization methods since it can be considered a method that provides effective contraception, according to the committee opinion.
Surgeons, however, should not change their approach to hysterectomy or sterilization just because of the “theoretical benefit of salpingectomy,” the committee wrote.
“A vaginal hysterectomy should not be changed to a laparoscopic hysterectomy simply to perform a salpingectomy,” ACOG wrote. “The choice of sterilization procedure should be based on the risks and benefits of the hysteroscopic and laparoscopic approaches.”
The opinion statement also noted that salpingectomy should be performed with “meticulous attention.” The entire fallopian tube should be removed, from the fimbriated end to the uterotubal junction, and any fimbrial attachments on the ovary should be cauterized or removed. “Care should be taken not to interrupt the blood supply to the ovary through the infundibulopelvic ligament because the collateral vasculature from the tubal mesosalpinx is occluded during tube removal.”
And in cases when complete salpingectomy cannot be accomplished, removing as much of the fallopian tubes as possible “still may have value,” ACOG wrote.
Salpingectomy may offer clinicians the chance to prevent ovarian cancer, according to a policy statement from the American College of Obstetricians and Gynecologists’ Committee on Gynecologic Practice, published in Obstetrics and Gynecology on Dec. 22.
Ovarian cancer carries the highest mortality of any gynecologic cancer and is the fifth-leading cause of cancer deaths among women. Overall survival has improved only marginally in the last 50 years, and current attempts at screening are not only unsuccessful but have led to unnecessary surgery and associated complications. The new ACOG committee opinion advises physicians to consider prophylactic salpingectomy as part of a prevention strategy.
The first step for physicians is to discuss salpingectomy with women who are scheduled to undergo routine pelvic surgery, such as tubal sterilization or hysterectomy without oophorectomy. Performing concurrent salpingectomy does not appear to increase complications, compared with tubal ligation or hysterectomy alone, and doesn’t appear to affect ovarian function.
“Given current theories of ovarian carcinogenesis, ovarian conservation and salpingectomy may represent a better option than [bilateral salpingo-oophorectomy] for ovarian cancer risk reduction for most women undergoing other pelvic surgeries for benign disease,” ACOG wrote in the committee opinion (Obstet. Gynecol. 2015;125:279-81).
But the ACOG committee advised physicians to discuss the role of oophorectomy and bilateral salpingo-oophorectomy as part the informed consent discussion.
Salpingectomy also should be discussed when counseling women about laparoscopic sterilization methods since it can be considered a method that provides effective contraception, according to the committee opinion.
Surgeons, however, should not change their approach to hysterectomy or sterilization just because of the “theoretical benefit of salpingectomy,” the committee wrote.
“A vaginal hysterectomy should not be changed to a laparoscopic hysterectomy simply to perform a salpingectomy,” ACOG wrote. “The choice of sterilization procedure should be based on the risks and benefits of the hysteroscopic and laparoscopic approaches.”
The opinion statement also noted that salpingectomy should be performed with “meticulous attention.” The entire fallopian tube should be removed, from the fimbriated end to the uterotubal junction, and any fimbrial attachments on the ovary should be cauterized or removed. “Care should be taken not to interrupt the blood supply to the ovary through the infundibulopelvic ligament because the collateral vasculature from the tubal mesosalpinx is occluded during tube removal.”
And in cases when complete salpingectomy cannot be accomplished, removing as much of the fallopian tubes as possible “still may have value,” ACOG wrote.
FROM OBSTETRICS AND GYNECOLOGY
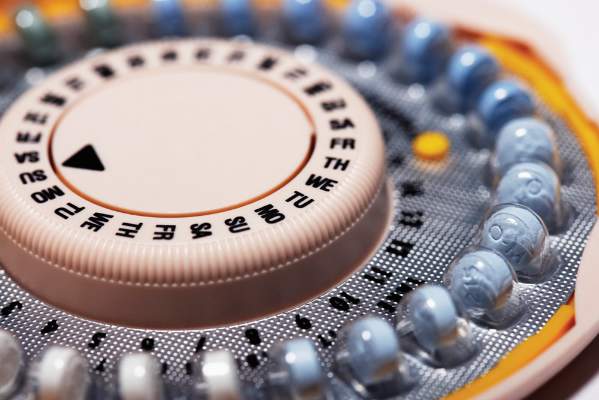
ACOG targets barriers to contraception access
Many barriers still impede women’s access to contraceptive care, and physicians can do more to address them, according to a policy statement and a list of 18 recommendations from the American College of Obstetricians and Gynecologists.
The policy statement, released on Dec. 22 by the ACOG Committee on Health Care for Underserved Women, details many of the reasons why contraceptive care continues to fall short of being an “integral component of women’s health care” and why nearly half of pregnancies in the United States are unintended. High on the list are lack of knowledge, misperceptions, and exaggerated concerns about the safety of contraceptive methods (Obstet. Gynecol. 2015;125:250-5).
These misconceptions exist not just among patients, but among clinicians, the ACOG committee wrote. For instance, many clinicians are “uncertain about the risks and benefits of IUDs and lack knowledge about correct patient selection and contraindications.”
Similarly, many patients have unfounded concerns that oral contraceptives cause major health problems or that IUDs carry a high risk of infection. Many also mistakenly believe that most contraceptives are abortifacients.
Ob.gyns. can do their part to eliminate access barriers by not requiring routine pelvic examinations or cervical cytology before initiating hormonal contraception.
“The prospect of such an examination may deter a woman, especially an adolescent, from having a clinical visit that could facilitate her use of a more effective contraceptive method than those available over the counter,” the committee wrote.
Likewise, it’s excessive to require one appointment to discuss long-acting reversible contraception (LARC) methods and a second appointment to place the device. LARC methods are highly effective but underutilized, especially by adolescent and nulliparous women, according to the committee statement.
Another way that physicians can help prevent unintended pregnancies is to ensure that women are able to undergo permanent sterilization procedures immediately post partum, if they choose. Many women are prevented from doing so because of limited operating room availability, lack of motivation or coordination among the health care team, a mistaken perception of increased risk if such procedures are done in the postpartum setting, and misplacement of sterilization consent forms.
Other major barriers to accessing contraception are related to insurance coverage. High out-of-pocket costs, deductibles, and copayments for contraception limit its use. Even women with good private health insurance that “covers” contraception pay approximately 60% of the cost, compared with only 33% of the cost of other medications. And many insurers limit the number of contraceptive products that can be dispensed at one time.
“Insurance plan restrictions prevent 73% of women from receiving more than a single month’s supply of contraception at a time, yet most women are unable to obtain contraceptive refills on a timely basis,” ACOG wrote.
Additionally, some insurers, clinics, and pharmacies require women to fail on certain contraceptive methods before allowing them to try a more expensive method, such as an IUD or implant.
The opinion statement offers 18 recommendations for improving access to contraception, including full implementation of the Affordable Care Act’s requirement for insurance plans to fully cover all Food and Drug Administration–approved contraceptives, removal of age restrictions to increase access to emergency contraception, and adequate payments to physicians for providing contraceptive services.
Many barriers still impede women’s access to contraceptive care, and physicians can do more to address them, according to a policy statement and a list of 18 recommendations from the American College of Obstetricians and Gynecologists.
The policy statement, released on Dec. 22 by the ACOG Committee on Health Care for Underserved Women, details many of the reasons why contraceptive care continues to fall short of being an “integral component of women’s health care” and why nearly half of pregnancies in the United States are unintended. High on the list are lack of knowledge, misperceptions, and exaggerated concerns about the safety of contraceptive methods (Obstet. Gynecol. 2015;125:250-5).
These misconceptions exist not just among patients, but among clinicians, the ACOG committee wrote. For instance, many clinicians are “uncertain about the risks and benefits of IUDs and lack knowledge about correct patient selection and contraindications.”
Similarly, many patients have unfounded concerns that oral contraceptives cause major health problems or that IUDs carry a high risk of infection. Many also mistakenly believe that most contraceptives are abortifacients.
Ob.gyns. can do their part to eliminate access barriers by not requiring routine pelvic examinations or cervical cytology before initiating hormonal contraception.
“The prospect of such an examination may deter a woman, especially an adolescent, from having a clinical visit that could facilitate her use of a more effective contraceptive method than those available over the counter,” the committee wrote.
Likewise, it’s excessive to require one appointment to discuss long-acting reversible contraception (LARC) methods and a second appointment to place the device. LARC methods are highly effective but underutilized, especially by adolescent and nulliparous women, according to the committee statement.
Another way that physicians can help prevent unintended pregnancies is to ensure that women are able to undergo permanent sterilization procedures immediately post partum, if they choose. Many women are prevented from doing so because of limited operating room availability, lack of motivation or coordination among the health care team, a mistaken perception of increased risk if such procedures are done in the postpartum setting, and misplacement of sterilization consent forms.
Other major barriers to accessing contraception are related to insurance coverage. High out-of-pocket costs, deductibles, and copayments for contraception limit its use. Even women with good private health insurance that “covers” contraception pay approximately 60% of the cost, compared with only 33% of the cost of other medications. And many insurers limit the number of contraceptive products that can be dispensed at one time.
“Insurance plan restrictions prevent 73% of women from receiving more than a single month’s supply of contraception at a time, yet most women are unable to obtain contraceptive refills on a timely basis,” ACOG wrote.
Additionally, some insurers, clinics, and pharmacies require women to fail on certain contraceptive methods before allowing them to try a more expensive method, such as an IUD or implant.
The opinion statement offers 18 recommendations for improving access to contraception, including full implementation of the Affordable Care Act’s requirement for insurance plans to fully cover all Food and Drug Administration–approved contraceptives, removal of age restrictions to increase access to emergency contraception, and adequate payments to physicians for providing contraceptive services.
Many barriers still impede women’s access to contraceptive care, and physicians can do more to address them, according to a policy statement and a list of 18 recommendations from the American College of Obstetricians and Gynecologists.
The policy statement, released on Dec. 22 by the ACOG Committee on Health Care for Underserved Women, details many of the reasons why contraceptive care continues to fall short of being an “integral component of women’s health care” and why nearly half of pregnancies in the United States are unintended. High on the list are lack of knowledge, misperceptions, and exaggerated concerns about the safety of contraceptive methods (Obstet. Gynecol. 2015;125:250-5).
These misconceptions exist not just among patients, but among clinicians, the ACOG committee wrote. For instance, many clinicians are “uncertain about the risks and benefits of IUDs and lack knowledge about correct patient selection and contraindications.”
Similarly, many patients have unfounded concerns that oral contraceptives cause major health problems or that IUDs carry a high risk of infection. Many also mistakenly believe that most contraceptives are abortifacients.
Ob.gyns. can do their part to eliminate access barriers by not requiring routine pelvic examinations or cervical cytology before initiating hormonal contraception.
“The prospect of such an examination may deter a woman, especially an adolescent, from having a clinical visit that could facilitate her use of a more effective contraceptive method than those available over the counter,” the committee wrote.
Likewise, it’s excessive to require one appointment to discuss long-acting reversible contraception (LARC) methods and a second appointment to place the device. LARC methods are highly effective but underutilized, especially by adolescent and nulliparous women, according to the committee statement.
Another way that physicians can help prevent unintended pregnancies is to ensure that women are able to undergo permanent sterilization procedures immediately post partum, if they choose. Many women are prevented from doing so because of limited operating room availability, lack of motivation or coordination among the health care team, a mistaken perception of increased risk if such procedures are done in the postpartum setting, and misplacement of sterilization consent forms.
Other major barriers to accessing contraception are related to insurance coverage. High out-of-pocket costs, deductibles, and copayments for contraception limit its use. Even women with good private health insurance that “covers” contraception pay approximately 60% of the cost, compared with only 33% of the cost of other medications. And many insurers limit the number of contraceptive products that can be dispensed at one time.
“Insurance plan restrictions prevent 73% of women from receiving more than a single month’s supply of contraception at a time, yet most women are unable to obtain contraceptive refills on a timely basis,” ACOG wrote.
Additionally, some insurers, clinics, and pharmacies require women to fail on certain contraceptive methods before allowing them to try a more expensive method, such as an IUD or implant.
The opinion statement offers 18 recommendations for improving access to contraception, including full implementation of the Affordable Care Act’s requirement for insurance plans to fully cover all Food and Drug Administration–approved contraceptives, removal of age restrictions to increase access to emergency contraception, and adequate payments to physicians for providing contraceptive services.
FROM OBSTETRICS AND GYNECOLOGY
Cheaper cytisine topped nicotine replacement therapy for smoking cessation
Cytisine was more effective than nicotine replacement therapy at helping adults quit smoking, with effect sizes similar to those reported in trials of varenicline, according to a report published online Dec. 18 in the New England Journal of Medicine.
Cytisine is a plant-based alkaloid that, like varenicline, is a partial agonist of nicotinic acetylcholine receptors. It remains relatively unknown outside of Eastern Europe, where it has been used successfully for 50 years, the study authors noted, even though it has proved effective there and is inexpensive. Some in the medical community have called for it to be licensed worldwide as a smoking-cessation aid.
The researchers directly compared cytisine with nicotine replacement therapy in an open-label, randomized, noninferiority trial in New Zealand.
A total of 1,310 adults who contacted New Zealand’s Quitline for assistance in smoking cessation were randomly assigned in equal numbers to receive either 25 days’ worth of cytisine tablets or 8 weeks of nicotine replacement therapy (patch and/or gum) plus brief behavioral counseling. They were followed for 6 months.
Cytisine proved significantly superior to nicotine replacement therapy: 40% of patients who took cytisine reported continuous smoking abstinence at 1 month, compared with 31% of those in the control group, said Natalie Walker, Ph.D., of the National Institute for Health Innovation, University of Auckland (New Zealand), and her associates.
Patients in the cytisine group also reported fewer symptoms of tobacco withdrawal than those in the nicotine replacement group.
Among study participants who resumed smoking after completing treatment, the median time to relapse was significantly longer with cytisine (53 days) than with nicotine replacement (11 days). People in the cytisine group also reported smoking fewer cigarettes per day and found smoking less rewarding than it had been before the study, the investigators said (N. Engl. J. Med. 2014;371:2353-62).
However, self-reported adverse events – most commonly nausea, vomiting, and sleep disorders – were almost twice as frequent in the cytisine group as in the control group. Most adverse events were mild to moderate in severity, and only 5% of the study population discontinued treatment because of adverse events.
The findings show that cytisine “is an effective smoking cessation aid for use as a first-line treatment for tobacco dependence,” Dr. Walker and her associates said.
The Health Research Council of New Zealand supported the study. Sopharma, a manufacturer of cytisine (Tabex), supplied the agent at no cost. Dr. Walker reported having no financial conflicts of interest; one of her associates reported receiving lecture fees from Johnson & Johnson and grant support from Pfizer.
Tobacco use is now the leading preventable cause of death worldwide, and the need for smoking cessation aids is urgent.
The compelling rationale for bringing cytisine to market is not that its efficacy is superior to that of current pharmacotherapies, but that the latter are unavailable to many smokers because of their cost. People in low- and middle-income countries would certainly benefit from a less expensive alternative, as would those in high-income countries who seek to contain health care costs.
Dr. Nancy A. Rigotti is in the department of medicine and the Tobacco Research and Treatment Center at Massachusetts General Hospital and Harvard Medical School, Boston. She reported receiving nonfinancial support from Pfizer unrelated to this work. Dr. Rigotti made these remarks in an editorial accompanying Dr. Walker’s report (N. Engl. J. Med. 2014;371:2429-30).
Tobacco use is now the leading preventable cause of death worldwide, and the need for smoking cessation aids is urgent.
The compelling rationale for bringing cytisine to market is not that its efficacy is superior to that of current pharmacotherapies, but that the latter are unavailable to many smokers because of their cost. People in low- and middle-income countries would certainly benefit from a less expensive alternative, as would those in high-income countries who seek to contain health care costs.
Dr. Nancy A. Rigotti is in the department of medicine and the Tobacco Research and Treatment Center at Massachusetts General Hospital and Harvard Medical School, Boston. She reported receiving nonfinancial support from Pfizer unrelated to this work. Dr. Rigotti made these remarks in an editorial accompanying Dr. Walker’s report (N. Engl. J. Med. 2014;371:2429-30).
Tobacco use is now the leading preventable cause of death worldwide, and the need for smoking cessation aids is urgent.
The compelling rationale for bringing cytisine to market is not that its efficacy is superior to that of current pharmacotherapies, but that the latter are unavailable to many smokers because of their cost. People in low- and middle-income countries would certainly benefit from a less expensive alternative, as would those in high-income countries who seek to contain health care costs.
Dr. Nancy A. Rigotti is in the department of medicine and the Tobacco Research and Treatment Center at Massachusetts General Hospital and Harvard Medical School, Boston. She reported receiving nonfinancial support from Pfizer unrelated to this work. Dr. Rigotti made these remarks in an editorial accompanying Dr. Walker’s report (N. Engl. J. Med. 2014;371:2429-30).
Cytisine was more effective than nicotine replacement therapy at helping adults quit smoking, with effect sizes similar to those reported in trials of varenicline, according to a report published online Dec. 18 in the New England Journal of Medicine.
Cytisine is a plant-based alkaloid that, like varenicline, is a partial agonist of nicotinic acetylcholine receptors. It remains relatively unknown outside of Eastern Europe, where it has been used successfully for 50 years, the study authors noted, even though it has proved effective there and is inexpensive. Some in the medical community have called for it to be licensed worldwide as a smoking-cessation aid.
The researchers directly compared cytisine with nicotine replacement therapy in an open-label, randomized, noninferiority trial in New Zealand.
A total of 1,310 adults who contacted New Zealand’s Quitline for assistance in smoking cessation were randomly assigned in equal numbers to receive either 25 days’ worth of cytisine tablets or 8 weeks of nicotine replacement therapy (patch and/or gum) plus brief behavioral counseling. They were followed for 6 months.
Cytisine proved significantly superior to nicotine replacement therapy: 40% of patients who took cytisine reported continuous smoking abstinence at 1 month, compared with 31% of those in the control group, said Natalie Walker, Ph.D., of the National Institute for Health Innovation, University of Auckland (New Zealand), and her associates.
Patients in the cytisine group also reported fewer symptoms of tobacco withdrawal than those in the nicotine replacement group.
Among study participants who resumed smoking after completing treatment, the median time to relapse was significantly longer with cytisine (53 days) than with nicotine replacement (11 days). People in the cytisine group also reported smoking fewer cigarettes per day and found smoking less rewarding than it had been before the study, the investigators said (N. Engl. J. Med. 2014;371:2353-62).
However, self-reported adverse events – most commonly nausea, vomiting, and sleep disorders – were almost twice as frequent in the cytisine group as in the control group. Most adverse events were mild to moderate in severity, and only 5% of the study population discontinued treatment because of adverse events.
The findings show that cytisine “is an effective smoking cessation aid for use as a first-line treatment for tobacco dependence,” Dr. Walker and her associates said.
The Health Research Council of New Zealand supported the study. Sopharma, a manufacturer of cytisine (Tabex), supplied the agent at no cost. Dr. Walker reported having no financial conflicts of interest; one of her associates reported receiving lecture fees from Johnson & Johnson and grant support from Pfizer.
Cytisine was more effective than nicotine replacement therapy at helping adults quit smoking, with effect sizes similar to those reported in trials of varenicline, according to a report published online Dec. 18 in the New England Journal of Medicine.
Cytisine is a plant-based alkaloid that, like varenicline, is a partial agonist of nicotinic acetylcholine receptors. It remains relatively unknown outside of Eastern Europe, where it has been used successfully for 50 years, the study authors noted, even though it has proved effective there and is inexpensive. Some in the medical community have called for it to be licensed worldwide as a smoking-cessation aid.
The researchers directly compared cytisine with nicotine replacement therapy in an open-label, randomized, noninferiority trial in New Zealand.
A total of 1,310 adults who contacted New Zealand’s Quitline for assistance in smoking cessation were randomly assigned in equal numbers to receive either 25 days’ worth of cytisine tablets or 8 weeks of nicotine replacement therapy (patch and/or gum) plus brief behavioral counseling. They were followed for 6 months.
Cytisine proved significantly superior to nicotine replacement therapy: 40% of patients who took cytisine reported continuous smoking abstinence at 1 month, compared with 31% of those in the control group, said Natalie Walker, Ph.D., of the National Institute for Health Innovation, University of Auckland (New Zealand), and her associates.
Patients in the cytisine group also reported fewer symptoms of tobacco withdrawal than those in the nicotine replacement group.
Among study participants who resumed smoking after completing treatment, the median time to relapse was significantly longer with cytisine (53 days) than with nicotine replacement (11 days). People in the cytisine group also reported smoking fewer cigarettes per day and found smoking less rewarding than it had been before the study, the investigators said (N. Engl. J. Med. 2014;371:2353-62).
However, self-reported adverse events – most commonly nausea, vomiting, and sleep disorders – were almost twice as frequent in the cytisine group as in the control group. Most adverse events were mild to moderate in severity, and only 5% of the study population discontinued treatment because of adverse events.
The findings show that cytisine “is an effective smoking cessation aid for use as a first-line treatment for tobacco dependence,” Dr. Walker and her associates said.
The Health Research Council of New Zealand supported the study. Sopharma, a manufacturer of cytisine (Tabex), supplied the agent at no cost. Dr. Walker reported having no financial conflicts of interest; one of her associates reported receiving lecture fees from Johnson & Johnson and grant support from Pfizer.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Cytisine was superior to nicotine replacement therapy for helping adults quit smoking.
Major finding: 40% of patients who took cytisine reported continuous smoking abstinence at 1 month, compared with 31% of patients in the control group.
Data source: An open-label randomized, controlled, noninferiority trial involving 1,310 New Zealand adults followed for 6 months.
Disclosures: The Health Research Council of New Zealand supported the study. Sopharma, a manufacturer of cytisine (Tabex), supplied the agent at no cost. Dr. Walker reported having no financial conflicts of interest; one of her associates reported receiving lecture fees from Johnson & Johnson and grant support from Pfizer.
Intra-arterial therapy improves function after ischemic stroke
Intra-arterial treatment not only enhanced reperfusion but improved patients’ functional recovery after acute ischemic stroke caused by a proximal intracranial arterial occlusion of the anterior circulation in the MR CLEAN trial, a multicenter, open-label, randomized, controlled phase III study.
Contrary to the findings of recent randomized, controlled trials that failed to show such a benefit from intra-arterial therapy, these results clearly demonstrated “a clinically significant increase in functional independence in daily life by 3 months, without any increase in mortality,” said Dr. Olvert A. Berkhemer of the department of radiology at Academic Medical Center, Amsterdam, and the department of neurology at Erasmus University Medical Center, Rotterdam, the Netherlands, and his associates.
They performed the MR CLEAN (Multicenter Randomized Clinical Trial of Endovascular Treatment for Acute Ischemic Stroke in the Netherlands) study to compare intra-arterial treatment – using a microcatheter to deliver a thrombolytic agent, mechanical thrombectomy, or both at the level of occlusion – against usual care in 500 adults aged 23-96 years. All study participants were treated within 6 hours of stroke onset. Usual care could include intravenous alteplase, and mechanical treatment could include thrombus retraction, aspiration, wire disruption, or use of a retrievable stent, all at the discretion of the treating interventionist, the investigators said (N. Engl. J. Med. 2014 Dec. 17 [doi:10.1056/NEJMoa1411587]).
The primary outcome – functional status at 90 days, as measured by the modified Rankin scale – was superior for intra-arterial treatment, with 32.6% of patients achieving functional independence, compared with only 19.1% of the usual-care group. All secondary outcomes also favored intra-arterial treatment, including scores on the National Institutes of Health Stroke Scale (NIHSS) and on measures of activities of daily living and health-related quality of life. In addition, absence of residual occlusion was markedly more frequent with intra-arterial treatment (75.4%) than with usual care (32.9%), and infarct volume was smaller.
There were no significant differences between the two study groups in mortality at 7, 30, or 90 days of follow-up. However, complications related to intra-arterial treatment included embolization into new intracranial territories in 20 patients (8.6%), vessel dissections in 4 patients (1.7%), and vessel perforations in 2 patients (0.9%). In addition, 30 patients who received intra-arterial treatment (13%) also underwent a simultaneous second revascularization, usually cervical carotid stenting, at the discretion of the interventionist. “This complexity needs to be considered when interpreting our trial results,” Dr. Berkhemer and his associates said.
Although MR CLEAN is the first trial to cut through some of the criticisms of previous trials that used intra-arterial therapy by enrolling patients with severe strokes and have proof of proximal vessel occlusion, initiating treatment as early as possible, and using modern thrombectomy devices, it is “premature to conclude that there is no longer equipoise regarding thrombectomy. We need and will get results from other well-designed trials [several of which are ongoing], not only to confirm or refute the results of MR CLEAN but also to look at effects in subgroups (according to stroke severity, occlusion site, or time to treatment initiation), for which most single trials are underpowered,” wrote Dr. Werner Hacke of the department of neurology at University Hospital Heidelberg (Germany) in an accompanying editorial (N. Engl. J. Med. 2014 Dec. 17 [doi:10.1056/NEJMe1413346]).
MR CLEAN was funded by the Dutch Heart Foundation, AngioCare, Covidien/ev3, Medac/Lamepro, and Penumbra. Dr. Berkhemer reported having no financial conflicts of interest, but his associates reported ties to Stryker, BALT, Toshiba, Codman/DePuy Synthes, Sequent Medical, and Covidien/ev3. Dr. Hacke reported grant support from Boehringer Ingelheim, and personal fees from Bayer, Daiichi Sankyo, and Covidien outside his submitted work.
Intra-arterial treatment not only enhanced reperfusion but improved patients’ functional recovery after acute ischemic stroke caused by a proximal intracranial arterial occlusion of the anterior circulation in the MR CLEAN trial, a multicenter, open-label, randomized, controlled phase III study.
Contrary to the findings of recent randomized, controlled trials that failed to show such a benefit from intra-arterial therapy, these results clearly demonstrated “a clinically significant increase in functional independence in daily life by 3 months, without any increase in mortality,” said Dr. Olvert A. Berkhemer of the department of radiology at Academic Medical Center, Amsterdam, and the department of neurology at Erasmus University Medical Center, Rotterdam, the Netherlands, and his associates.
They performed the MR CLEAN (Multicenter Randomized Clinical Trial of Endovascular Treatment for Acute Ischemic Stroke in the Netherlands) study to compare intra-arterial treatment – using a microcatheter to deliver a thrombolytic agent, mechanical thrombectomy, or both at the level of occlusion – against usual care in 500 adults aged 23-96 years. All study participants were treated within 6 hours of stroke onset. Usual care could include intravenous alteplase, and mechanical treatment could include thrombus retraction, aspiration, wire disruption, or use of a retrievable stent, all at the discretion of the treating interventionist, the investigators said (N. Engl. J. Med. 2014 Dec. 17 [doi:10.1056/NEJMoa1411587]).
The primary outcome – functional status at 90 days, as measured by the modified Rankin scale – was superior for intra-arterial treatment, with 32.6% of patients achieving functional independence, compared with only 19.1% of the usual-care group. All secondary outcomes also favored intra-arterial treatment, including scores on the National Institutes of Health Stroke Scale (NIHSS) and on measures of activities of daily living and health-related quality of life. In addition, absence of residual occlusion was markedly more frequent with intra-arterial treatment (75.4%) than with usual care (32.9%), and infarct volume was smaller.
There were no significant differences between the two study groups in mortality at 7, 30, or 90 days of follow-up. However, complications related to intra-arterial treatment included embolization into new intracranial territories in 20 patients (8.6%), vessel dissections in 4 patients (1.7%), and vessel perforations in 2 patients (0.9%). In addition, 30 patients who received intra-arterial treatment (13%) also underwent a simultaneous second revascularization, usually cervical carotid stenting, at the discretion of the interventionist. “This complexity needs to be considered when interpreting our trial results,” Dr. Berkhemer and his associates said.
Although MR CLEAN is the first trial to cut through some of the criticisms of previous trials that used intra-arterial therapy by enrolling patients with severe strokes and have proof of proximal vessel occlusion, initiating treatment as early as possible, and using modern thrombectomy devices, it is “premature to conclude that there is no longer equipoise regarding thrombectomy. We need and will get results from other well-designed trials [several of which are ongoing], not only to confirm or refute the results of MR CLEAN but also to look at effects in subgroups (according to stroke severity, occlusion site, or time to treatment initiation), for which most single trials are underpowered,” wrote Dr. Werner Hacke of the department of neurology at University Hospital Heidelberg (Germany) in an accompanying editorial (N. Engl. J. Med. 2014 Dec. 17 [doi:10.1056/NEJMe1413346]).
MR CLEAN was funded by the Dutch Heart Foundation, AngioCare, Covidien/ev3, Medac/Lamepro, and Penumbra. Dr. Berkhemer reported having no financial conflicts of interest, but his associates reported ties to Stryker, BALT, Toshiba, Codman/DePuy Synthes, Sequent Medical, and Covidien/ev3. Dr. Hacke reported grant support from Boehringer Ingelheim, and personal fees from Bayer, Daiichi Sankyo, and Covidien outside his submitted work.
Intra-arterial treatment not only enhanced reperfusion but improved patients’ functional recovery after acute ischemic stroke caused by a proximal intracranial arterial occlusion of the anterior circulation in the MR CLEAN trial, a multicenter, open-label, randomized, controlled phase III study.
Contrary to the findings of recent randomized, controlled trials that failed to show such a benefit from intra-arterial therapy, these results clearly demonstrated “a clinically significant increase in functional independence in daily life by 3 months, without any increase in mortality,” said Dr. Olvert A. Berkhemer of the department of radiology at Academic Medical Center, Amsterdam, and the department of neurology at Erasmus University Medical Center, Rotterdam, the Netherlands, and his associates.
They performed the MR CLEAN (Multicenter Randomized Clinical Trial of Endovascular Treatment for Acute Ischemic Stroke in the Netherlands) study to compare intra-arterial treatment – using a microcatheter to deliver a thrombolytic agent, mechanical thrombectomy, or both at the level of occlusion – against usual care in 500 adults aged 23-96 years. All study participants were treated within 6 hours of stroke onset. Usual care could include intravenous alteplase, and mechanical treatment could include thrombus retraction, aspiration, wire disruption, or use of a retrievable stent, all at the discretion of the treating interventionist, the investigators said (N. Engl. J. Med. 2014 Dec. 17 [doi:10.1056/NEJMoa1411587]).
The primary outcome – functional status at 90 days, as measured by the modified Rankin scale – was superior for intra-arterial treatment, with 32.6% of patients achieving functional independence, compared with only 19.1% of the usual-care group. All secondary outcomes also favored intra-arterial treatment, including scores on the National Institutes of Health Stroke Scale (NIHSS) and on measures of activities of daily living and health-related quality of life. In addition, absence of residual occlusion was markedly more frequent with intra-arterial treatment (75.4%) than with usual care (32.9%), and infarct volume was smaller.
There were no significant differences between the two study groups in mortality at 7, 30, or 90 days of follow-up. However, complications related to intra-arterial treatment included embolization into new intracranial territories in 20 patients (8.6%), vessel dissections in 4 patients (1.7%), and vessel perforations in 2 patients (0.9%). In addition, 30 patients who received intra-arterial treatment (13%) also underwent a simultaneous second revascularization, usually cervical carotid stenting, at the discretion of the interventionist. “This complexity needs to be considered when interpreting our trial results,” Dr. Berkhemer and his associates said.
Although MR CLEAN is the first trial to cut through some of the criticisms of previous trials that used intra-arterial therapy by enrolling patients with severe strokes and have proof of proximal vessel occlusion, initiating treatment as early as possible, and using modern thrombectomy devices, it is “premature to conclude that there is no longer equipoise regarding thrombectomy. We need and will get results from other well-designed trials [several of which are ongoing], not only to confirm or refute the results of MR CLEAN but also to look at effects in subgroups (according to stroke severity, occlusion site, or time to treatment initiation), for which most single trials are underpowered,” wrote Dr. Werner Hacke of the department of neurology at University Hospital Heidelberg (Germany) in an accompanying editorial (N. Engl. J. Med. 2014 Dec. 17 [doi:10.1056/NEJMe1413346]).
MR CLEAN was funded by the Dutch Heart Foundation, AngioCare, Covidien/ev3, Medac/Lamepro, and Penumbra. Dr. Berkhemer reported having no financial conflicts of interest, but his associates reported ties to Stryker, BALT, Toshiba, Codman/DePuy Synthes, Sequent Medical, and Covidien/ev3. Dr. Hacke reported grant support from Boehringer Ingelheim, and personal fees from Bayer, Daiichi Sankyo, and Covidien outside his submitted work.
FROM NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Intra-arterial therapy improves patient function, not just reperfusion, after ischemic stroke caused by proximal intracranial arterial occlusion.
Major finding: A total of 32.6% of patients receiving intra-arterial treatment achieved functional independence at 90 days, compared with only 19.1% of the usual-care group
Data source: MR CLEAN is a multicenter, phase III, open-label, randomized, controlled trial involving 500 Dutch patients with acute ischemic stroke.
Disclosures: MR CLEAN was funded by the Dutch Heart Foundation, AngioCare, Covidien/ev3, Medac/Lamepro, and Penumbra. Dr. Berkhemer reported having no financial conflicts of interest, but his associates reported ties to Stryker, BALT, Toshiba, Codman/DePuy Synthes, Sequent Medical, and Covidien/ev3.
Glycemic Index Doesn’t Affect Insulin Sensitivity, Lipid Levels
A healthy diet that was modified to have a low glycemic index did not improve insulin sensitivity, lipid levels, or blood pressure in overweight and obese adults, compared with one with a high glycemic index, according to a report published online Dec. 16 in JAMA.
This unexpected finding indicates that, against a background diet that is already healthy, “using glycemic index to select specific foods may not improve cardiovascular risk factors or insulin resistance,” said Dr. Frank M. Sacks of the department of nutrition, Harvard School of Public Health, Boston, and his associates.
The investigators performed a randomized, crossover feeding study in which 163 people aged at least 30 years were randomly assigned to one of four study diets for 5 weeks, then switched to a different diet for a further 5 weeks, after a 2-week washout period.
“We studied diets that had a large contrast in glycemic index, while at the same time we controlled intake of total carbohydrates and other key nutrients such as fatty acids, potassium, and sodium and maintained body weight. The background diets in which we manipulated glycemic index were healthful dietary patterns established in the Dietary Approaches to Stop Hypertension (DASH) and Optimal Macronutrient Intake to Prevent Heart Disease (OmniHeart) studies that are being recommended in dietary guidelines to prevent CVD,” they noted. About half of the study population were female and half were black, and there were high prevalences of obesity (56%), hypertension (26%), high cholesterol (68%), and impaired fasting glucose (30%). All meals, snacks, and beverages were provided on site. Adherence, which was closely monitored, was high: All study foods and no nonstudy foods were consumed on 96% of person-days on each diet.
Contrary to expectations, the two low-glycemic-index diets failed to improve insulin sensitivity, cholesterol levels, or blood pressure when compared with the two high-glycemic-index diets. “Composing a DASH-type diet with low-glycemic-index foods ... does not improve CVD risk factors and may in fact reduce insulin sensitivity and increase LDL cholesterol,” Dr. Sacks and his associates wrote (JAMA 2014 Dec. 16 [doi:10.1001/jama.2014.16658]).
They noted that these findings are especially important given that some nutrition policies advocate replacing high-glycemic-index with low-glycemic-index foods. Even though some experts go so far as to promote that all foods be labeled with their glycemic-index values, “the benefits of glycemic index are uncertain, especially with persons are already consuming a healthful diet rich in whole grains, vegetables, and fruits.”
The investigators emphasized that none of the study participants had type 2 diabetes, so these findings do not apply to patients with that disease.
The unexpected findings by Sacks et al. suggest that glycemic index is less important than previously thought, especially in the context of an overall healthy diet, as tested in this study.
These results should direct attention back to the importance of maintaining an overall heart-healthy lifestyle rather than focusing on a narrow concept such as the glycemic index of individual foods. It isn’t a matter of good foods and bad foods, but the overall dietary pattern that is important.
Dr. Robert H. Eckel of the University of Colorado, Aurora, made these remarks in an editorial accompanying Dr. Sacks’s report (JAMA 2014;312:2508-9). He reported having no financial disclosures.
The unexpected findings by Sacks et al. suggest that glycemic index is less important than previously thought, especially in the context of an overall healthy diet, as tested in this study.
These results should direct attention back to the importance of maintaining an overall heart-healthy lifestyle rather than focusing on a narrow concept such as the glycemic index of individual foods. It isn’t a matter of good foods and bad foods, but the overall dietary pattern that is important.
Dr. Robert H. Eckel of the University of Colorado, Aurora, made these remarks in an editorial accompanying Dr. Sacks’s report (JAMA 2014;312:2508-9). He reported having no financial disclosures.
The unexpected findings by Sacks et al. suggest that glycemic index is less important than previously thought, especially in the context of an overall healthy diet, as tested in this study.
These results should direct attention back to the importance of maintaining an overall heart-healthy lifestyle rather than focusing on a narrow concept such as the glycemic index of individual foods. It isn’t a matter of good foods and bad foods, but the overall dietary pattern that is important.
Dr. Robert H. Eckel of the University of Colorado, Aurora, made these remarks in an editorial accompanying Dr. Sacks’s report (JAMA 2014;312:2508-9). He reported having no financial disclosures.
A healthy diet that was modified to have a low glycemic index did not improve insulin sensitivity, lipid levels, or blood pressure in overweight and obese adults, compared with one with a high glycemic index, according to a report published online Dec. 16 in JAMA.
This unexpected finding indicates that, against a background diet that is already healthy, “using glycemic index to select specific foods may not improve cardiovascular risk factors or insulin resistance,” said Dr. Frank M. Sacks of the department of nutrition, Harvard School of Public Health, Boston, and his associates.
The investigators performed a randomized, crossover feeding study in which 163 people aged at least 30 years were randomly assigned to one of four study diets for 5 weeks, then switched to a different diet for a further 5 weeks, after a 2-week washout period.
“We studied diets that had a large contrast in glycemic index, while at the same time we controlled intake of total carbohydrates and other key nutrients such as fatty acids, potassium, and sodium and maintained body weight. The background diets in which we manipulated glycemic index were healthful dietary patterns established in the Dietary Approaches to Stop Hypertension (DASH) and Optimal Macronutrient Intake to Prevent Heart Disease (OmniHeart) studies that are being recommended in dietary guidelines to prevent CVD,” they noted. About half of the study population were female and half were black, and there were high prevalences of obesity (56%), hypertension (26%), high cholesterol (68%), and impaired fasting glucose (30%). All meals, snacks, and beverages were provided on site. Adherence, which was closely monitored, was high: All study foods and no nonstudy foods were consumed on 96% of person-days on each diet.
Contrary to expectations, the two low-glycemic-index diets failed to improve insulin sensitivity, cholesterol levels, or blood pressure when compared with the two high-glycemic-index diets. “Composing a DASH-type diet with low-glycemic-index foods ... does not improve CVD risk factors and may in fact reduce insulin sensitivity and increase LDL cholesterol,” Dr. Sacks and his associates wrote (JAMA 2014 Dec. 16 [doi:10.1001/jama.2014.16658]).
They noted that these findings are especially important given that some nutrition policies advocate replacing high-glycemic-index with low-glycemic-index foods. Even though some experts go so far as to promote that all foods be labeled with their glycemic-index values, “the benefits of glycemic index are uncertain, especially with persons are already consuming a healthful diet rich in whole grains, vegetables, and fruits.”
The investigators emphasized that none of the study participants had type 2 diabetes, so these findings do not apply to patients with that disease.
A healthy diet that was modified to have a low glycemic index did not improve insulin sensitivity, lipid levels, or blood pressure in overweight and obese adults, compared with one with a high glycemic index, according to a report published online Dec. 16 in JAMA.
This unexpected finding indicates that, against a background diet that is already healthy, “using glycemic index to select specific foods may not improve cardiovascular risk factors or insulin resistance,” said Dr. Frank M. Sacks of the department of nutrition, Harvard School of Public Health, Boston, and his associates.
The investigators performed a randomized, crossover feeding study in which 163 people aged at least 30 years were randomly assigned to one of four study diets for 5 weeks, then switched to a different diet for a further 5 weeks, after a 2-week washout period.
“We studied diets that had a large contrast in glycemic index, while at the same time we controlled intake of total carbohydrates and other key nutrients such as fatty acids, potassium, and sodium and maintained body weight. The background diets in which we manipulated glycemic index were healthful dietary patterns established in the Dietary Approaches to Stop Hypertension (DASH) and Optimal Macronutrient Intake to Prevent Heart Disease (OmniHeart) studies that are being recommended in dietary guidelines to prevent CVD,” they noted. About half of the study population were female and half were black, and there were high prevalences of obesity (56%), hypertension (26%), high cholesterol (68%), and impaired fasting glucose (30%). All meals, snacks, and beverages were provided on site. Adherence, which was closely monitored, was high: All study foods and no nonstudy foods were consumed on 96% of person-days on each diet.
Contrary to expectations, the two low-glycemic-index diets failed to improve insulin sensitivity, cholesterol levels, or blood pressure when compared with the two high-glycemic-index diets. “Composing a DASH-type diet with low-glycemic-index foods ... does not improve CVD risk factors and may in fact reduce insulin sensitivity and increase LDL cholesterol,” Dr. Sacks and his associates wrote (JAMA 2014 Dec. 16 [doi:10.1001/jama.2014.16658]).
They noted that these findings are especially important given that some nutrition policies advocate replacing high-glycemic-index with low-glycemic-index foods. Even though some experts go so far as to promote that all foods be labeled with their glycemic-index values, “the benefits of glycemic index are uncertain, especially with persons are already consuming a healthful diet rich in whole grains, vegetables, and fruits.”
The investigators emphasized that none of the study participants had type 2 diabetes, so these findings do not apply to patients with that disease.
Glycemic index doesn’t affect insulin sensitivity, lipid levels
A healthy diet that was modified to have a low glycemic index did not improve insulin sensitivity, lipid levels, or blood pressure in overweight and obese adults, compared with one with a high glycemic index, according to a report published online Dec. 16 in JAMA.
This unexpected finding indicates that, against a background diet that is already healthy, “using glycemic index to select specific foods may not improve cardiovascular risk factors or insulin resistance,” said Dr. Frank M. Sacks of the department of nutrition, Harvard School of Public Health, Boston, and his associates.
The investigators performed a randomized, crossover feeding study in which 163 people aged at least 30 years were randomly assigned to one of four study diets for 5 weeks, then switched to a different diet for a further 5 weeks, after a 2-week washout period.
“We studied diets that had a large contrast in glycemic index, while at the same time we controlled intake of total carbohydrates and other key nutrients such as fatty acids, potassium, and sodium and maintained body weight. The background diets in which we manipulated glycemic index were healthful dietary patterns established in the Dietary Approaches to Stop Hypertension (DASH) and Optimal Macronutrient Intake to Prevent Heart Disease (OmniHeart) studies that are being recommended in dietary guidelines to prevent CVD,” they noted. About half of the study population were female and half were black, and there were high prevalences of obesity (56%), hypertension (26%), high cholesterol (68%), and impaired fasting glucose (30%). All meals, snacks, and beverages were provided on site. Adherence, which was closely monitored, was high: All study foods and no nonstudy foods were consumed on 96% of person-days on each diet.
Contrary to expectations, the two low-glycemic-index diets failed to improve insulin sensitivity, cholesterol levels, or blood pressure when compared with the two high-glycemic-index diets. “Composing a DASH-type diet with low-glycemic-index foods ... does not improve CVD risk factors and may in fact reduce insulin sensitivity and increase LDL cholesterol,” Dr. Sacks and his associates wrote (JAMA 2014 Dec. 16 [doi:10.1001/jama.2014.16658]).
They noted that these findings are especially important given that some nutrition policies advocate replacing high-glycemic-index with low-glycemic-index foods. Even though some experts go so far as to promote that all foods be labeled with their glycemic-index values, “the benefits of glycemic index are uncertain, especially with persons are already consuming a healthful diet rich in whole grains, vegetables, and fruits.”
The investigators emphasized that none of the study participants had type 2 diabetes, so these findings do not apply to patients with that disease.
The unexpected findings by Sacks et al. suggest that glycemic index is less important than previously thought, especially in the context of an overall healthy diet, as tested in this study.
These results should direct attention back to the importance of maintaining an overall heart-healthy lifestyle rather than focusing on a narrow concept such as the glycemic index of individual foods. It isn’t a matter of good foods and bad foods, but the overall dietary pattern that is important.
Dr. Robert H. Eckel of the University of Colorado, Aurora, made these remarks in an editorial accompanying Dr. Sacks’s report (JAMA 2014;312:2508-9). He reported having no financial disclosures.
The unexpected findings by Sacks et al. suggest that glycemic index is less important than previously thought, especially in the context of an overall healthy diet, as tested in this study.
These results should direct attention back to the importance of maintaining an overall heart-healthy lifestyle rather than focusing on a narrow concept such as the glycemic index of individual foods. It isn’t a matter of good foods and bad foods, but the overall dietary pattern that is important.
Dr. Robert H. Eckel of the University of Colorado, Aurora, made these remarks in an editorial accompanying Dr. Sacks’s report (JAMA 2014;312:2508-9). He reported having no financial disclosures.
The unexpected findings by Sacks et al. suggest that glycemic index is less important than previously thought, especially in the context of an overall healthy diet, as tested in this study.
These results should direct attention back to the importance of maintaining an overall heart-healthy lifestyle rather than focusing on a narrow concept such as the glycemic index of individual foods. It isn’t a matter of good foods and bad foods, but the overall dietary pattern that is important.
Dr. Robert H. Eckel of the University of Colorado, Aurora, made these remarks in an editorial accompanying Dr. Sacks’s report (JAMA 2014;312:2508-9). He reported having no financial disclosures.
A healthy diet that was modified to have a low glycemic index did not improve insulin sensitivity, lipid levels, or blood pressure in overweight and obese adults, compared with one with a high glycemic index, according to a report published online Dec. 16 in JAMA.
This unexpected finding indicates that, against a background diet that is already healthy, “using glycemic index to select specific foods may not improve cardiovascular risk factors or insulin resistance,” said Dr. Frank M. Sacks of the department of nutrition, Harvard School of Public Health, Boston, and his associates.
The investigators performed a randomized, crossover feeding study in which 163 people aged at least 30 years were randomly assigned to one of four study diets for 5 weeks, then switched to a different diet for a further 5 weeks, after a 2-week washout period.
“We studied diets that had a large contrast in glycemic index, while at the same time we controlled intake of total carbohydrates and other key nutrients such as fatty acids, potassium, and sodium and maintained body weight. The background diets in which we manipulated glycemic index were healthful dietary patterns established in the Dietary Approaches to Stop Hypertension (DASH) and Optimal Macronutrient Intake to Prevent Heart Disease (OmniHeart) studies that are being recommended in dietary guidelines to prevent CVD,” they noted. About half of the study population were female and half were black, and there were high prevalences of obesity (56%), hypertension (26%), high cholesterol (68%), and impaired fasting glucose (30%). All meals, snacks, and beverages were provided on site. Adherence, which was closely monitored, was high: All study foods and no nonstudy foods were consumed on 96% of person-days on each diet.
Contrary to expectations, the two low-glycemic-index diets failed to improve insulin sensitivity, cholesterol levels, or blood pressure when compared with the two high-glycemic-index diets. “Composing a DASH-type diet with low-glycemic-index foods ... does not improve CVD risk factors and may in fact reduce insulin sensitivity and increase LDL cholesterol,” Dr. Sacks and his associates wrote (JAMA 2014 Dec. 16 [doi:10.1001/jama.2014.16658]).
They noted that these findings are especially important given that some nutrition policies advocate replacing high-glycemic-index with low-glycemic-index foods. Even though some experts go so far as to promote that all foods be labeled with their glycemic-index values, “the benefits of glycemic index are uncertain, especially with persons are already consuming a healthful diet rich in whole grains, vegetables, and fruits.”
The investigators emphasized that none of the study participants had type 2 diabetes, so these findings do not apply to patients with that disease.
A healthy diet that was modified to have a low glycemic index did not improve insulin sensitivity, lipid levels, or blood pressure in overweight and obese adults, compared with one with a high glycemic index, according to a report published online Dec. 16 in JAMA.
This unexpected finding indicates that, against a background diet that is already healthy, “using glycemic index to select specific foods may not improve cardiovascular risk factors or insulin resistance,” said Dr. Frank M. Sacks of the department of nutrition, Harvard School of Public Health, Boston, and his associates.
The investigators performed a randomized, crossover feeding study in which 163 people aged at least 30 years were randomly assigned to one of four study diets for 5 weeks, then switched to a different diet for a further 5 weeks, after a 2-week washout period.
“We studied diets that had a large contrast in glycemic index, while at the same time we controlled intake of total carbohydrates and other key nutrients such as fatty acids, potassium, and sodium and maintained body weight. The background diets in which we manipulated glycemic index were healthful dietary patterns established in the Dietary Approaches to Stop Hypertension (DASH) and Optimal Macronutrient Intake to Prevent Heart Disease (OmniHeart) studies that are being recommended in dietary guidelines to prevent CVD,” they noted. About half of the study population were female and half were black, and there were high prevalences of obesity (56%), hypertension (26%), high cholesterol (68%), and impaired fasting glucose (30%). All meals, snacks, and beverages were provided on site. Adherence, which was closely monitored, was high: All study foods and no nonstudy foods were consumed on 96% of person-days on each diet.
Contrary to expectations, the two low-glycemic-index diets failed to improve insulin sensitivity, cholesterol levels, or blood pressure when compared with the two high-glycemic-index diets. “Composing a DASH-type diet with low-glycemic-index foods ... does not improve CVD risk factors and may in fact reduce insulin sensitivity and increase LDL cholesterol,” Dr. Sacks and his associates wrote (JAMA 2014 Dec. 16 [doi:10.1001/jama.2014.16658]).
They noted that these findings are especially important given that some nutrition policies advocate replacing high-glycemic-index with low-glycemic-index foods. Even though some experts go so far as to promote that all foods be labeled with their glycemic-index values, “the benefits of glycemic index are uncertain, especially with persons are already consuming a healthful diet rich in whole grains, vegetables, and fruits.”
The investigators emphasized that none of the study participants had type 2 diabetes, so these findings do not apply to patients with that disease.
Key clinical point: A low-glycemic-index diet failed to improve insulin sensitivity, lipid levels, or blood pressure, compared with a high-glycemic-index diet.
Major finding: Contrary to expectations, the two low-glycemic-index diets failed to improve insulin sensitivity, cholesterol levels, or blood pressure when compared with the two high-glycemic-index diets.
Data source: A 5-week randomized crossover controlled-feeding study involving 163 overweight or obese adults with high rates of hypertension, hypercholesterolemia, and impaired fasting glucose.
Disclosures: This study was supported by the National Institutes of Health, the Harvard Clinical and Translational Science Center, the National Center for Advancing Translational Science, and Brigham and Women’s Hospital. Dr. Sacks and his associates reported no relevant financial disclosures.
AI-induced symptoms don’t predict survival
Vasomotor and musculoskeletal symptoms associated with aromatase inhibitor adjuvant therapy do not signal a more intense treatment response and thus do not predict better survival in women with breast cancer, according to a report published online Dec. 15 in Journal of Clinical Oncology.
Even though several retrospective exploratory analyses have suggested that treatment-emergent symptoms may be associated with improved survival in this patient population, this secondary analysis of data in a large international prospective randomized clinical trial argues otherwise. “It is premature to counsel patients on whether to continue or change their adjuvant AI therapy based on symptoms,” said Dr. Vered Stearns of the Kimmel Cancer Center, Johns Hopkins University, Baltimore, and her associates.
To investigate whether adverse effects might be useful in informing treatment decisions, the investigators analyzed data from the NCIC Clinical Trials Group MA.27 trial – an open-label phase III study conducted in 2003-2008 and involving 7,567 postmenopausal women who had hormone receptor–positive breast cancer. After completing their initial treatment and a washout period to allow for any adverse effects related to it, these participants were randomly assigned to receive adjuvant therapy with either anastrozole (3,787 women) or exemestane (3,789 women) and followed for a median of 4 years. To collect treatment-related symptoms in a standardized manner, they completed Common Terminology Criteria for Adverse Events questionnaires at baseline and at regular intervals afterward.
A relatively high number of these participants (34%) reported already having such symptoms at baseline, which may reflect rebound symptoms from hormone therapy withdrawal during the washout period. For women who had no symptoms at baseline, 25% reported them at 6 months and 52% reported them by 12 months.
At 3-month follow-up, 96 of the 403 women with severe symptoms discontinued treatment; the rate of treatment discontinuation was 12% at 6 months and 10% at 12 months. The most common reason cited for discontinuation was joint pain.
The emergence of new or worsening vasomotor and musculoskeletal symptoms showed no association with recurrence-free survival. There was no correlation between recurrence-free survival and symptoms at baseline or symptoms induced by AI therapy at 3 months, 6 months, or 12 months in the overall study population or in any subgroup of patients, Dr. Stearns and her associates said (J. Clin. Oncol. 2014 Dec. 15 [doi:10.1200;JCO.2014.57.6926]).
These findings indicate that treatment-emergent symptoms should be managed as effectively as possible but not held up as evidence that adjuvant therapy is working. “Women should be supported through treatment and encouraged to remain on their AI regardless of their symptoms,” the investigators added.
The notion that toxicity related to treatment can be a surrogate for treatment efficacy has long been embedded in the psyche of patients. Oncologists also can mistakenly believe that with cancer, which warrants the most aggressive possible therapy, the strength of a treatment is reflected in the adverse effects it produces.

|
Dr. William J. Gradishar |
But toxicity must not be considered an automatic surrogate measure of clinical benefit. Much of cancer treatments’ toxicity results from collateral damage: nontargeted effects to normal tissue that may or may not share features of the target cancer cell.
Dr. William J. Gradishar is at Northwestern University, Chicago. His financial disclosures are available at www.jco.org. Dr. Gradishar made these remarks in an editorial accompanying Dr. Stearns’s report (J. Clin. Oncol. 2014 Dec. 15 [doi:10.1200/JCO.2014.59.0919]).
The notion that toxicity related to treatment can be a surrogate for treatment efficacy has long been embedded in the psyche of patients. Oncologists also can mistakenly believe that with cancer, which warrants the most aggressive possible therapy, the strength of a treatment is reflected in the adverse effects it produces.

|
Dr. William J. Gradishar |
But toxicity must not be considered an automatic surrogate measure of clinical benefit. Much of cancer treatments’ toxicity results from collateral damage: nontargeted effects to normal tissue that may or may not share features of the target cancer cell.
Dr. William J. Gradishar is at Northwestern University, Chicago. His financial disclosures are available at www.jco.org. Dr. Gradishar made these remarks in an editorial accompanying Dr. Stearns’s report (J. Clin. Oncol. 2014 Dec. 15 [doi:10.1200/JCO.2014.59.0919]).
The notion that toxicity related to treatment can be a surrogate for treatment efficacy has long been embedded in the psyche of patients. Oncologists also can mistakenly believe that with cancer, which warrants the most aggressive possible therapy, the strength of a treatment is reflected in the adverse effects it produces.

|
Dr. William J. Gradishar |
But toxicity must not be considered an automatic surrogate measure of clinical benefit. Much of cancer treatments’ toxicity results from collateral damage: nontargeted effects to normal tissue that may or may not share features of the target cancer cell.
Dr. William J. Gradishar is at Northwestern University, Chicago. His financial disclosures are available at www.jco.org. Dr. Gradishar made these remarks in an editorial accompanying Dr. Stearns’s report (J. Clin. Oncol. 2014 Dec. 15 [doi:10.1200/JCO.2014.59.0919]).
Vasomotor and musculoskeletal symptoms associated with aromatase inhibitor adjuvant therapy do not signal a more intense treatment response and thus do not predict better survival in women with breast cancer, according to a report published online Dec. 15 in Journal of Clinical Oncology.
Even though several retrospective exploratory analyses have suggested that treatment-emergent symptoms may be associated with improved survival in this patient population, this secondary analysis of data in a large international prospective randomized clinical trial argues otherwise. “It is premature to counsel patients on whether to continue or change their adjuvant AI therapy based on symptoms,” said Dr. Vered Stearns of the Kimmel Cancer Center, Johns Hopkins University, Baltimore, and her associates.
To investigate whether adverse effects might be useful in informing treatment decisions, the investigators analyzed data from the NCIC Clinical Trials Group MA.27 trial – an open-label phase III study conducted in 2003-2008 and involving 7,567 postmenopausal women who had hormone receptor–positive breast cancer. After completing their initial treatment and a washout period to allow for any adverse effects related to it, these participants were randomly assigned to receive adjuvant therapy with either anastrozole (3,787 women) or exemestane (3,789 women) and followed for a median of 4 years. To collect treatment-related symptoms in a standardized manner, they completed Common Terminology Criteria for Adverse Events questionnaires at baseline and at regular intervals afterward.
A relatively high number of these participants (34%) reported already having such symptoms at baseline, which may reflect rebound symptoms from hormone therapy withdrawal during the washout period. For women who had no symptoms at baseline, 25% reported them at 6 months and 52% reported them by 12 months.
At 3-month follow-up, 96 of the 403 women with severe symptoms discontinued treatment; the rate of treatment discontinuation was 12% at 6 months and 10% at 12 months. The most common reason cited for discontinuation was joint pain.
The emergence of new or worsening vasomotor and musculoskeletal symptoms showed no association with recurrence-free survival. There was no correlation between recurrence-free survival and symptoms at baseline or symptoms induced by AI therapy at 3 months, 6 months, or 12 months in the overall study population or in any subgroup of patients, Dr. Stearns and her associates said (J. Clin. Oncol. 2014 Dec. 15 [doi:10.1200;JCO.2014.57.6926]).
These findings indicate that treatment-emergent symptoms should be managed as effectively as possible but not held up as evidence that adjuvant therapy is working. “Women should be supported through treatment and encouraged to remain on their AI regardless of their symptoms,” the investigators added.
Vasomotor and musculoskeletal symptoms associated with aromatase inhibitor adjuvant therapy do not signal a more intense treatment response and thus do not predict better survival in women with breast cancer, according to a report published online Dec. 15 in Journal of Clinical Oncology.
Even though several retrospective exploratory analyses have suggested that treatment-emergent symptoms may be associated with improved survival in this patient population, this secondary analysis of data in a large international prospective randomized clinical trial argues otherwise. “It is premature to counsel patients on whether to continue or change their adjuvant AI therapy based on symptoms,” said Dr. Vered Stearns of the Kimmel Cancer Center, Johns Hopkins University, Baltimore, and her associates.
To investigate whether adverse effects might be useful in informing treatment decisions, the investigators analyzed data from the NCIC Clinical Trials Group MA.27 trial – an open-label phase III study conducted in 2003-2008 and involving 7,567 postmenopausal women who had hormone receptor–positive breast cancer. After completing their initial treatment and a washout period to allow for any adverse effects related to it, these participants were randomly assigned to receive adjuvant therapy with either anastrozole (3,787 women) or exemestane (3,789 women) and followed for a median of 4 years. To collect treatment-related symptoms in a standardized manner, they completed Common Terminology Criteria for Adverse Events questionnaires at baseline and at regular intervals afterward.
A relatively high number of these participants (34%) reported already having such symptoms at baseline, which may reflect rebound symptoms from hormone therapy withdrawal during the washout period. For women who had no symptoms at baseline, 25% reported them at 6 months and 52% reported them by 12 months.
At 3-month follow-up, 96 of the 403 women with severe symptoms discontinued treatment; the rate of treatment discontinuation was 12% at 6 months and 10% at 12 months. The most common reason cited for discontinuation was joint pain.
The emergence of new or worsening vasomotor and musculoskeletal symptoms showed no association with recurrence-free survival. There was no correlation between recurrence-free survival and symptoms at baseline or symptoms induced by AI therapy at 3 months, 6 months, or 12 months in the overall study population or in any subgroup of patients, Dr. Stearns and her associates said (J. Clin. Oncol. 2014 Dec. 15 [doi:10.1200;JCO.2014.57.6926]).
These findings indicate that treatment-emergent symptoms should be managed as effectively as possible but not held up as evidence that adjuvant therapy is working. “Women should be supported through treatment and encouraged to remain on their AI regardless of their symptoms,” the investigators added.
Key clinical point: Vasomotor and musculoskeletal symptoms induced by aromatase inhibitors do not signal a more-intense treatment response or improved survival in breast cancer.
Major finding:There was no correlation between recurrence-free survival and symptoms at baseline or symptoms induced by AI therapy at 3 months, 6 months, or 12 months in the overall study population or in any subgroup of patients.
Data source: A secondary analysis of data in a large international randomized phase III clinical trial involving 7,576 postmenopausal women with hormone receptor–positive breast cancer who were followed for a median of 4 years.
Disclosures: This study was supported by the NCIC Clinical Trials Group, Canada; the Canadian Cancer Society Research Institute; the U.S. National Cancer Institute; the International Breast Cancer Study Group; Pfizer; the Susan G. Komen for the Cure; and the Avon Foundation, New York. Dr. Stearns reported receiving research funding from AbbVie, Celgene, Merck, Novartis, Pfizer, MedImmune, and Puma, and her associates reported ties to numerous industry sources.
New agent extends progression-free survival in advanced pancreatic cancer
Adding an investigational hypoxia-activated cytotoxic agent, TH-302, to standard gemcitabine chemotherapy significantly improved progression-free survival and the tumor response rate in a phase II clinical trial of adults with advanced or metastatic pancreatic cancer, according to a report published online Dec. 15 in the Journal of Clinical Oncology.
The encouraging results of this industry-sponsored open-label trial (NCT01144455) spurred the initiation of an international phase III study of TH-302 (NCT01746979), which “has the potential to provide an alternative regimen to the current standard-of-care regimens,” as well as a phase I/II study combining TH-302 with gemcitabine plus nab-paclitaxel (NCT02047500) for pancreatic cancer, said Dr. Mitesh J. Borad of the Mayo Clinic, Scottsdale, Ariz., and his associates.
In this trial, 214 patients who hadn’t received any systemic therapy for pancreatic cancer were enrolled during a 1-year period and treated at 45 U.S. medical centers. The study participants were randomly assigned to receive gemcitabine alone, which was the standard of care at the time (69 patients); a lower dose of TH-302 (240 mg/m2) plus gemcitabine (71 patients); or a higher dose of TH-302 (340 mg/m2) plus gemcitabine (74 patients). Any patients in the gemcitabine-only group whose disease progressed were allowed to cross over and be randomly assigned to one of the other study groups, and 26 patients did so.
The primary efficacy endpoint, progression-free survival, was significantly prolonged with the addition of TH-302 (median, 5.6 months), compared with gemcitabine alone (median, 3.6 months). The rate of complete or partial tumor response also was significantly higher with the addition of lower-dose TH-302 (17%) or higher-dose TH-302 (26%) than with gemcitabine alone (12%).
The median overall survival was longer with lower-dose TH-302 (8.7 months) or higher-dose TH-302 (9.2 months) than with gemcitabine alone (6.9 months), but this difference did not attain statistical significance. However, the study wasn’t sufficiently powered to compare overall survival, which would have required a larger sample size. And permitting crossover from the control to the TH-302 groups also disallowed valid comparisons in overall survival, Dr. Borad and his associates said (J. Clin. Oncol. 2014 Dec. 15 [doi:10.1200/JCO.2014.55.7504]).
As expected, skin toxicity, mucosal toxicity, and enhanced myelosuppression were the most frequent adverse events related to TH-302. However, these effects were “manageable,” given that the rate of treatment discontinuation was no higher among patients who received the drug than among those who did not, the investigators added.
Adding an investigational hypoxia-activated cytotoxic agent, TH-302, to standard gemcitabine chemotherapy significantly improved progression-free survival and the tumor response rate in a phase II clinical trial of adults with advanced or metastatic pancreatic cancer, according to a report published online Dec. 15 in the Journal of Clinical Oncology.
The encouraging results of this industry-sponsored open-label trial (NCT01144455) spurred the initiation of an international phase III study of TH-302 (NCT01746979), which “has the potential to provide an alternative regimen to the current standard-of-care regimens,” as well as a phase I/II study combining TH-302 with gemcitabine plus nab-paclitaxel (NCT02047500) for pancreatic cancer, said Dr. Mitesh J. Borad of the Mayo Clinic, Scottsdale, Ariz., and his associates.
In this trial, 214 patients who hadn’t received any systemic therapy for pancreatic cancer were enrolled during a 1-year period and treated at 45 U.S. medical centers. The study participants were randomly assigned to receive gemcitabine alone, which was the standard of care at the time (69 patients); a lower dose of TH-302 (240 mg/m2) plus gemcitabine (71 patients); or a higher dose of TH-302 (340 mg/m2) plus gemcitabine (74 patients). Any patients in the gemcitabine-only group whose disease progressed were allowed to cross over and be randomly assigned to one of the other study groups, and 26 patients did so.
The primary efficacy endpoint, progression-free survival, was significantly prolonged with the addition of TH-302 (median, 5.6 months), compared with gemcitabine alone (median, 3.6 months). The rate of complete or partial tumor response also was significantly higher with the addition of lower-dose TH-302 (17%) or higher-dose TH-302 (26%) than with gemcitabine alone (12%).
The median overall survival was longer with lower-dose TH-302 (8.7 months) or higher-dose TH-302 (9.2 months) than with gemcitabine alone (6.9 months), but this difference did not attain statistical significance. However, the study wasn’t sufficiently powered to compare overall survival, which would have required a larger sample size. And permitting crossover from the control to the TH-302 groups also disallowed valid comparisons in overall survival, Dr. Borad and his associates said (J. Clin. Oncol. 2014 Dec. 15 [doi:10.1200/JCO.2014.55.7504]).
As expected, skin toxicity, mucosal toxicity, and enhanced myelosuppression were the most frequent adverse events related to TH-302. However, these effects were “manageable,” given that the rate of treatment discontinuation was no higher among patients who received the drug than among those who did not, the investigators added.
Adding an investigational hypoxia-activated cytotoxic agent, TH-302, to standard gemcitabine chemotherapy significantly improved progression-free survival and the tumor response rate in a phase II clinical trial of adults with advanced or metastatic pancreatic cancer, according to a report published online Dec. 15 in the Journal of Clinical Oncology.
The encouraging results of this industry-sponsored open-label trial (NCT01144455) spurred the initiation of an international phase III study of TH-302 (NCT01746979), which “has the potential to provide an alternative regimen to the current standard-of-care regimens,” as well as a phase I/II study combining TH-302 with gemcitabine plus nab-paclitaxel (NCT02047500) for pancreatic cancer, said Dr. Mitesh J. Borad of the Mayo Clinic, Scottsdale, Ariz., and his associates.
In this trial, 214 patients who hadn’t received any systemic therapy for pancreatic cancer were enrolled during a 1-year period and treated at 45 U.S. medical centers. The study participants were randomly assigned to receive gemcitabine alone, which was the standard of care at the time (69 patients); a lower dose of TH-302 (240 mg/m2) plus gemcitabine (71 patients); or a higher dose of TH-302 (340 mg/m2) plus gemcitabine (74 patients). Any patients in the gemcitabine-only group whose disease progressed were allowed to cross over and be randomly assigned to one of the other study groups, and 26 patients did so.
The primary efficacy endpoint, progression-free survival, was significantly prolonged with the addition of TH-302 (median, 5.6 months), compared with gemcitabine alone (median, 3.6 months). The rate of complete or partial tumor response also was significantly higher with the addition of lower-dose TH-302 (17%) or higher-dose TH-302 (26%) than with gemcitabine alone (12%).
The median overall survival was longer with lower-dose TH-302 (8.7 months) or higher-dose TH-302 (9.2 months) than with gemcitabine alone (6.9 months), but this difference did not attain statistical significance. However, the study wasn’t sufficiently powered to compare overall survival, which would have required a larger sample size. And permitting crossover from the control to the TH-302 groups also disallowed valid comparisons in overall survival, Dr. Borad and his associates said (J. Clin. Oncol. 2014 Dec. 15 [doi:10.1200/JCO.2014.55.7504]).
As expected, skin toxicity, mucosal toxicity, and enhanced myelosuppression were the most frequent adverse events related to TH-302. However, these effects were “manageable,” given that the rate of treatment discontinuation was no higher among patients who received the drug than among those who did not, the investigators added.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: The investigational cytotoxic agent TH-302 extended progression-free survival and improved the tumor response rate in advanced/metastatic pancreatic cancer.
Major finding: The primary efficacy endpoint, progression-free survival, was significantly prolonged with the addition of TH-302 (median, 5.6 months), compared with gemcitabine alone (median, 3.6 months).
Data source: A 1-year, open-label, multicenter, phase II randomized clinical trial involving 214 adults treated at 45 U.S. medical centers.
Disclosures: This study was supported by Threshold Pharmaceuticals and Merck KGaA, makers of TH-320. Dr. Borad’s associates reported ties to Eli Lilly and MedImmune.
Prostate cancer surveillance appears safe through 15 years
Active surveillance appears to be safe through 15 years of follow-up for men who have low-risk prostate cancer, according to a report published online Dec. 15 in the Journal of Clinical Oncology.
In extended follow-up of a prospective cohort study begun in 1995, 993 men (current median age, 68 years; range, 41-89 years) with low-risk prostate cancer were assessed. Active surveillance consisted of PSA testing every 3 months for the first 2 years after diagnosis and then every 6 months thereafter, with repeat biopsy at 1 year and then every 3-4 years until the age of 80. These study participants were offered radical intervention only if the disease showed signs of progression, said Dr. Laurence Klotz of Sunnybrook Health Sciences Centre, University of Toronto, and his associates.
A total of 149 patients died, 819 were alive, and 25 were lost to follow-up. Only 15 men (1.5%) died from prostate cancer, and an additional 13 men with confirmed metastases either are alive (9 patients) or died from other causes (4 patients). Overall, the risk of dying from another cause was nearly 10 times greater than that for dying from prostate cancer (HR, 9.2). Even among men younger than 70, who had lower competing risks of death from other causes than older men, the risk of death from another cause was almost six times greater than that for death from prostate cancer (HR, 5.8), the investigators said (J. Clin. Oncol. 2014 Dec. 15 [doi:10.1200/JCO.2014.55.1192]).
The rate of patients who developed PSA failure during follow-up was 2.8% at 5 years and 10.2% at 10 years after diagnosis. These outcomes are consistent with those in low-risk patients managed with initial definitive intervention such as radiotherapy and surgery, Dr. Klotz and his associates added.
This study was supported by the Prostate Cancer Research Foundation of Canada. Dr. Klotz and his associates reported having no financial conflicts of interest.
This report from Dr. Klotz and his associates gives us the longest follow-up to date in one of the largest extant active surveillance cohorts.
The overall rate of metastasis was 2.8%. Before proponents of immediate definitive treatment argue that this rate is too high, they should note that it is not dissimilar from the risk of lethal disease among men who receive such immediate definitive treatment for low-risk tumors.
Dr. Matthew R. Cooperberg is with the University of California, San Francisco, and the Helen Diller Family Comprehensive Cancer Center. He reported receiving research funding from Genomic Health, Myriad Genetics, and GenomeDx Biosciences. Dr. Cooperberg made these remarks in an editorial accompanying the report (J. Clin. Oncol. 2014 Dec. 15 [doi:10.1200/jco.2014.59.2329]).
This report from Dr. Klotz and his associates gives us the longest follow-up to date in one of the largest extant active surveillance cohorts.
The overall rate of metastasis was 2.8%. Before proponents of immediate definitive treatment argue that this rate is too high, they should note that it is not dissimilar from the risk of lethal disease among men who receive such immediate definitive treatment for low-risk tumors.
Dr. Matthew R. Cooperberg is with the University of California, San Francisco, and the Helen Diller Family Comprehensive Cancer Center. He reported receiving research funding from Genomic Health, Myriad Genetics, and GenomeDx Biosciences. Dr. Cooperberg made these remarks in an editorial accompanying the report (J. Clin. Oncol. 2014 Dec. 15 [doi:10.1200/jco.2014.59.2329]).
This report from Dr. Klotz and his associates gives us the longest follow-up to date in one of the largest extant active surveillance cohorts.
The overall rate of metastasis was 2.8%. Before proponents of immediate definitive treatment argue that this rate is too high, they should note that it is not dissimilar from the risk of lethal disease among men who receive such immediate definitive treatment for low-risk tumors.
Dr. Matthew R. Cooperberg is with the University of California, San Francisco, and the Helen Diller Family Comprehensive Cancer Center. He reported receiving research funding from Genomic Health, Myriad Genetics, and GenomeDx Biosciences. Dr. Cooperberg made these remarks in an editorial accompanying the report (J. Clin. Oncol. 2014 Dec. 15 [doi:10.1200/jco.2014.59.2329]).
Active surveillance appears to be safe through 15 years of follow-up for men who have low-risk prostate cancer, according to a report published online Dec. 15 in the Journal of Clinical Oncology.
In extended follow-up of a prospective cohort study begun in 1995, 993 men (current median age, 68 years; range, 41-89 years) with low-risk prostate cancer were assessed. Active surveillance consisted of PSA testing every 3 months for the first 2 years after diagnosis and then every 6 months thereafter, with repeat biopsy at 1 year and then every 3-4 years until the age of 80. These study participants were offered radical intervention only if the disease showed signs of progression, said Dr. Laurence Klotz of Sunnybrook Health Sciences Centre, University of Toronto, and his associates.
A total of 149 patients died, 819 were alive, and 25 were lost to follow-up. Only 15 men (1.5%) died from prostate cancer, and an additional 13 men with confirmed metastases either are alive (9 patients) or died from other causes (4 patients). Overall, the risk of dying from another cause was nearly 10 times greater than that for dying from prostate cancer (HR, 9.2). Even among men younger than 70, who had lower competing risks of death from other causes than older men, the risk of death from another cause was almost six times greater than that for death from prostate cancer (HR, 5.8), the investigators said (J. Clin. Oncol. 2014 Dec. 15 [doi:10.1200/JCO.2014.55.1192]).
The rate of patients who developed PSA failure during follow-up was 2.8% at 5 years and 10.2% at 10 years after diagnosis. These outcomes are consistent with those in low-risk patients managed with initial definitive intervention such as radiotherapy and surgery, Dr. Klotz and his associates added.
This study was supported by the Prostate Cancer Research Foundation of Canada. Dr. Klotz and his associates reported having no financial conflicts of interest.
Active surveillance appears to be safe through 15 years of follow-up for men who have low-risk prostate cancer, according to a report published online Dec. 15 in the Journal of Clinical Oncology.
In extended follow-up of a prospective cohort study begun in 1995, 993 men (current median age, 68 years; range, 41-89 years) with low-risk prostate cancer were assessed. Active surveillance consisted of PSA testing every 3 months for the first 2 years after diagnosis and then every 6 months thereafter, with repeat biopsy at 1 year and then every 3-4 years until the age of 80. These study participants were offered radical intervention only if the disease showed signs of progression, said Dr. Laurence Klotz of Sunnybrook Health Sciences Centre, University of Toronto, and his associates.
A total of 149 patients died, 819 were alive, and 25 were lost to follow-up. Only 15 men (1.5%) died from prostate cancer, and an additional 13 men with confirmed metastases either are alive (9 patients) or died from other causes (4 patients). Overall, the risk of dying from another cause was nearly 10 times greater than that for dying from prostate cancer (HR, 9.2). Even among men younger than 70, who had lower competing risks of death from other causes than older men, the risk of death from another cause was almost six times greater than that for death from prostate cancer (HR, 5.8), the investigators said (J. Clin. Oncol. 2014 Dec. 15 [doi:10.1200/JCO.2014.55.1192]).
The rate of patients who developed PSA failure during follow-up was 2.8% at 5 years and 10.2% at 10 years after diagnosis. These outcomes are consistent with those in low-risk patients managed with initial definitive intervention such as radiotherapy and surgery, Dr. Klotz and his associates added.
This study was supported by the Prostate Cancer Research Foundation of Canada. Dr. Klotz and his associates reported having no financial conflicts of interest.
Key clinical point: Active surveillance for low-risk prostate cancer appears safe through 15 years of follow-up.
Major finding: Only 1.5% of men died from prostate cancer during follow-up, and the risk of dying from another cause was nearly 10 times greater than that for dying from prostate cancer (HR, 9.2).
Data source: Extended (15-year) follow-up of a cohort of 993 men with low-risk prostate cancer who only underwent treatment if the disease showed signs of progression.
Disclosures: This study was supported by the Prostate Cancer Research Foundation of Canada. Dr. Klotz and his associates reported having no financial conflicts of interest.
Antifungal formulation improves penetration through nail plate
A new formulation of topical efinaconazole allows the antifungal to penetrate through the nail plate much better than do existing treatments for onychomycosis, according to a report in the December issue of Journal of Drugs in Dermatology.
Poor nail penetration and diffusion through the thick, keratin-rich, lipid-containing barrier of the nail is the main factor limiting the effectiveness of current antifungals. A formulation with reduced keratin binding and lipophilicity would be more successful, particularly one with low surface tension to facilitate the transfer, wetting, spreading, and adhesion of the active ingredient under the surface of the nail, said Dr. Leon H. Kircik of Mount Sinai Medical Center, New York; Indiana University, Indianapolis; and Physician Skin Care PLLC, Louisville, Ky.
Dr. Kircik tested 41 possible liquid formulations of efinaconazole, assessed eight prototypes on nail samples from cadavers and in a guinea pig model, and settled on one formulation – (14C)-efinaconazole 10% topical solution – with the best penetrative and spreading properties to test in human subjects. This formulation contained ethanol as the primary vehicle and solvent, cyclomethicone as a wetting agent that reduces surface tension, esters to keep the efinaconazole in solution after the alcohol evaporates, preservatives, and water. When the solution is applied to the affected nail and the solvents evaporate, the concentration of the active ingredient increases to approximately 40%, Dr. Kircik noted.
Dr. Kircik then tested this formulation in 40 patients with toenail onychomycosis (mean age, 67 years), who applied it daily for 28 days and were followed for an additional 2 weeks. The concentrations of efinaconazole in the nail were 4 orders of magnitude higher than the MIC values of efinaconazole against dermatophytes. Drug concentrations were not affected by nail thickness or the severity of nail disease, and were maintained throughout follow-up. The findings indicate that after treatment is completed, “the nail plate and nail bed continue to be exposed to inhibitory drug concentrations for a period of time,” Dr. Kircik said (J. Drugs Dermatol. 2014;13:1457-61).
In 11 patients, Dr. Kircik assessed the ability of the active drug to reach the site of infection by spreading into the space between the nail plate and the nail bed, rather than directly through the nail plate. Fluorescein was incorporated into the topical preparation and applied distally between the nail and nail bed, without touching the surface of the toenail. Then visualization under UV light showed the spreading of the formulation to the nail bed, a finding that was confirmed after nail clipping.
A new formulation of topical efinaconazole allows the antifungal to penetrate through the nail plate much better than do existing treatments for onychomycosis, according to a report in the December issue of Journal of Drugs in Dermatology.
Poor nail penetration and diffusion through the thick, keratin-rich, lipid-containing barrier of the nail is the main factor limiting the effectiveness of current antifungals. A formulation with reduced keratin binding and lipophilicity would be more successful, particularly one with low surface tension to facilitate the transfer, wetting, spreading, and adhesion of the active ingredient under the surface of the nail, said Dr. Leon H. Kircik of Mount Sinai Medical Center, New York; Indiana University, Indianapolis; and Physician Skin Care PLLC, Louisville, Ky.
Dr. Kircik tested 41 possible liquid formulations of efinaconazole, assessed eight prototypes on nail samples from cadavers and in a guinea pig model, and settled on one formulation – (14C)-efinaconazole 10% topical solution – with the best penetrative and spreading properties to test in human subjects. This formulation contained ethanol as the primary vehicle and solvent, cyclomethicone as a wetting agent that reduces surface tension, esters to keep the efinaconazole in solution after the alcohol evaporates, preservatives, and water. When the solution is applied to the affected nail and the solvents evaporate, the concentration of the active ingredient increases to approximately 40%, Dr. Kircik noted.
Dr. Kircik then tested this formulation in 40 patients with toenail onychomycosis (mean age, 67 years), who applied it daily for 28 days and were followed for an additional 2 weeks. The concentrations of efinaconazole in the nail were 4 orders of magnitude higher than the MIC values of efinaconazole against dermatophytes. Drug concentrations were not affected by nail thickness or the severity of nail disease, and were maintained throughout follow-up. The findings indicate that after treatment is completed, “the nail plate and nail bed continue to be exposed to inhibitory drug concentrations for a period of time,” Dr. Kircik said (J. Drugs Dermatol. 2014;13:1457-61).
In 11 patients, Dr. Kircik assessed the ability of the active drug to reach the site of infection by spreading into the space between the nail plate and the nail bed, rather than directly through the nail plate. Fluorescein was incorporated into the topical preparation and applied distally between the nail and nail bed, without touching the surface of the toenail. Then visualization under UV light showed the spreading of the formulation to the nail bed, a finding that was confirmed after nail clipping.
A new formulation of topical efinaconazole allows the antifungal to penetrate through the nail plate much better than do existing treatments for onychomycosis, according to a report in the December issue of Journal of Drugs in Dermatology.
Poor nail penetration and diffusion through the thick, keratin-rich, lipid-containing barrier of the nail is the main factor limiting the effectiveness of current antifungals. A formulation with reduced keratin binding and lipophilicity would be more successful, particularly one with low surface tension to facilitate the transfer, wetting, spreading, and adhesion of the active ingredient under the surface of the nail, said Dr. Leon H. Kircik of Mount Sinai Medical Center, New York; Indiana University, Indianapolis; and Physician Skin Care PLLC, Louisville, Ky.
Dr. Kircik tested 41 possible liquid formulations of efinaconazole, assessed eight prototypes on nail samples from cadavers and in a guinea pig model, and settled on one formulation – (14C)-efinaconazole 10% topical solution – with the best penetrative and spreading properties to test in human subjects. This formulation contained ethanol as the primary vehicle and solvent, cyclomethicone as a wetting agent that reduces surface tension, esters to keep the efinaconazole in solution after the alcohol evaporates, preservatives, and water. When the solution is applied to the affected nail and the solvents evaporate, the concentration of the active ingredient increases to approximately 40%, Dr. Kircik noted.
Dr. Kircik then tested this formulation in 40 patients with toenail onychomycosis (mean age, 67 years), who applied it daily for 28 days and were followed for an additional 2 weeks. The concentrations of efinaconazole in the nail were 4 orders of magnitude higher than the MIC values of efinaconazole against dermatophytes. Drug concentrations were not affected by nail thickness or the severity of nail disease, and were maintained throughout follow-up. The findings indicate that after treatment is completed, “the nail plate and nail bed continue to be exposed to inhibitory drug concentrations for a period of time,” Dr. Kircik said (J. Drugs Dermatol. 2014;13:1457-61).
In 11 patients, Dr. Kircik assessed the ability of the active drug to reach the site of infection by spreading into the space between the nail plate and the nail bed, rather than directly through the nail plate. Fluorescein was incorporated into the topical preparation and applied distally between the nail and nail bed, without touching the surface of the toenail. Then visualization under UV light showed the spreading of the formulation to the nail bed, a finding that was confirmed after nail clipping.
Key clinical point: A new topical efinaconazole formulation allows better penetration of the active drug through the nail plate and into the space between the nail and the nail bed.
Major finding: In 40 patients with toenail onychomycosis who applied the new formulation daily for 28 days, the concentrations of efinaconazole in the nail were 4 orders of magnitude higher than the MIC values of efinaconazole against dermatophytes.
Data source: Preclinical studies and a small phase II study testing the penetrance of efinaconazole through the nail plate in patients with onychomycosis.
Disclosures: This work was sponsored by Valeant Pharmaceuticals. Dr. Kircik reported serving as an investigator, consultant, speaker, and advisory board member for Valeant, Merz, and Anacor.