User login
Heavy Menstrual Bleeding: How Hematologists Can Help
Heavy menstrual bleeding is more than an inconvenience in adolescents: It often leads to significant medical complications, in addition to disruptions in quality of life. While measuring the true level of bleeding can be a challenge, hematologists say treatments are helpful and can be as simple — and surprising — as doses of aspirin.
About 90% of adolescents with heavy menstrual bleeding will have low ferritin, and 70% will develop anemia, said benign hematologist Juliana Perez Botero, MD, of the Mayo Clinic in Rochester, Minnesota, in a presentation at the American Society of Hematology (ASH) 2024 Annual Meeting. “This is an issue of big magnitude that has public health implications, but it’s also an issue of gender equality and social justice.”
Measuring Menstruation: What Counts as Heavy Bleeding?
According to hematologist Allison Wheeler, MD, of the University of Washington in Seattle, normal menstrual bleeding is defined as lasting for about 5 days with 30-50 mL of blood loss.
“Historically, heavy menstrual bleeding was defined as bleeding as > 7 days or > 80 mL of blood loss,” Wheeler said. “It’s pretty hard to measure those mL. So a more modern definition is increased menstrual blood loss that interferes with a female’s physical, social, emotional, or material quality of life.”
Measuring blood loss during menstruation isn’t simple. The alkaline hematin method, which measures blood in feminine hygiene products, is the “gold standard, but that’s all I’m going to say about that,” Wheeler said.
An alternative is the Pictorial Blood Loss Assessment Chart, a visual tool that patients can use to estimate blood loss, she said, although there have been questions about its accuracy. In addition, it only covers pads and tampons even though other products are now available.
Another option is the Adolescent Menstrual Bleeding Questionnaire. “It captures both how much patients are bleeding and a lot of quality-of-life aspects,” she said.
Non-Steroidal Anti-Inflammatory Drugs (NSAIDs), Surprisingly, Can Be Effective Treatments
It seems counterintuitive that NSAIDs can be helpful in patients with heavy bleeding. “We don’t think about them because Cox inhibition ends up leading to decreases in thromboxane A2, which is going to increase bleeding,” Wheeler said. However, she said, the drugs also decrease prostaglandin within the endometrium, which can improve menstrual bleeding.
“In a meta-analysis, when NSAIDs were compared to placebo, there was decreased menstrual bleeding,” she said. “It was pretty similar to estrogen-progesterone formulations together. And when compared to antifibrinolytics, there was a little more menstrual bleeding with the NSAID use.”
Wheeler cautioned that “it’s important to optimize NSAID dosage. You really can’t use lower doses, and you can’t use sporadic doses.”
Other Options: Tranexamic Acid, Hormone Management
Tranexamic acid is the most common antifibrinolytic treatment for heavy bleeding, Wheeler said. “This is a lysine analog that’s going to inhibit the conversion from plasminogen to plasmin,” she said. “This is going to strengthen and stabilize blood clots that are already formed.”
Multiple clinical trials have studied the treatment in heavy menstrual bleeding, she said. “There’s no increased risk of venous or arterial thrombosis among participants in these clinical trials, but patients with a history of thrombosis were excluded from the majority of them. So we do need to keep that in mind.”
As for adverse effects, they’re mild and sporadic and include headaches and stomach upset, she said.
Another treatment option is estrogen and progesterone therapy using pill, patch, or vaginal ring, Wheeler said. The treatment leads to a “stable and thin very endometrium,” and typically takes 3-6 months to fully kick in. Studies have suggested the therapy can lead to 35%-80% reduction in menstrual blood loss, she said.
Keep in mind, Wheeler noted, “that there are many contraindications associated with estrogen use. Please take note of this, especially in consideration of discussions with patients shifting over to progesterone-only hormonal therapy” — another option to treat excess bleeding.
In most patients, she added, progesterone-only therapy “is going to result in irregular bleeding when you first start taking it. That irregular bleeding can last from up to 3-6 months. It’s something that’s really important to discuss with patients.”
She also noted that this therapy can be given to patients in forms that do not prevent pregnancy.
Botero had no disclosures. Wheeler reported relationships with Novo Nordisk, Bayer, BioMarin, Bioverativ, CSL Behring, Genentech, HEMA, Octapharma, Pfizer, Sanofi-Aventis, Shire North America, Spark, and Takeda.
A version of this article first appeared on Medscape.com.
Heavy menstrual bleeding is more than an inconvenience in adolescents: It often leads to significant medical complications, in addition to disruptions in quality of life. While measuring the true level of bleeding can be a challenge, hematologists say treatments are helpful and can be as simple — and surprising — as doses of aspirin.
About 90% of adolescents with heavy menstrual bleeding will have low ferritin, and 70% will develop anemia, said benign hematologist Juliana Perez Botero, MD, of the Mayo Clinic in Rochester, Minnesota, in a presentation at the American Society of Hematology (ASH) 2024 Annual Meeting. “This is an issue of big magnitude that has public health implications, but it’s also an issue of gender equality and social justice.”
Measuring Menstruation: What Counts as Heavy Bleeding?
According to hematologist Allison Wheeler, MD, of the University of Washington in Seattle, normal menstrual bleeding is defined as lasting for about 5 days with 30-50 mL of blood loss.
“Historically, heavy menstrual bleeding was defined as bleeding as > 7 days or > 80 mL of blood loss,” Wheeler said. “It’s pretty hard to measure those mL. So a more modern definition is increased menstrual blood loss that interferes with a female’s physical, social, emotional, or material quality of life.”
Measuring blood loss during menstruation isn’t simple. The alkaline hematin method, which measures blood in feminine hygiene products, is the “gold standard, but that’s all I’m going to say about that,” Wheeler said.
An alternative is the Pictorial Blood Loss Assessment Chart, a visual tool that patients can use to estimate blood loss, she said, although there have been questions about its accuracy. In addition, it only covers pads and tampons even though other products are now available.
Another option is the Adolescent Menstrual Bleeding Questionnaire. “It captures both how much patients are bleeding and a lot of quality-of-life aspects,” she said.
Non-Steroidal Anti-Inflammatory Drugs (NSAIDs), Surprisingly, Can Be Effective Treatments
It seems counterintuitive that NSAIDs can be helpful in patients with heavy bleeding. “We don’t think about them because Cox inhibition ends up leading to decreases in thromboxane A2, which is going to increase bleeding,” Wheeler said. However, she said, the drugs also decrease prostaglandin within the endometrium, which can improve menstrual bleeding.
“In a meta-analysis, when NSAIDs were compared to placebo, there was decreased menstrual bleeding,” she said. “It was pretty similar to estrogen-progesterone formulations together. And when compared to antifibrinolytics, there was a little more menstrual bleeding with the NSAID use.”
Wheeler cautioned that “it’s important to optimize NSAID dosage. You really can’t use lower doses, and you can’t use sporadic doses.”
Other Options: Tranexamic Acid, Hormone Management
Tranexamic acid is the most common antifibrinolytic treatment for heavy bleeding, Wheeler said. “This is a lysine analog that’s going to inhibit the conversion from plasminogen to plasmin,” she said. “This is going to strengthen and stabilize blood clots that are already formed.”
Multiple clinical trials have studied the treatment in heavy menstrual bleeding, she said. “There’s no increased risk of venous or arterial thrombosis among participants in these clinical trials, but patients with a history of thrombosis were excluded from the majority of them. So we do need to keep that in mind.”
As for adverse effects, they’re mild and sporadic and include headaches and stomach upset, she said.
Another treatment option is estrogen and progesterone therapy using pill, patch, or vaginal ring, Wheeler said. The treatment leads to a “stable and thin very endometrium,” and typically takes 3-6 months to fully kick in. Studies have suggested the therapy can lead to 35%-80% reduction in menstrual blood loss, she said.
Keep in mind, Wheeler noted, “that there are many contraindications associated with estrogen use. Please take note of this, especially in consideration of discussions with patients shifting over to progesterone-only hormonal therapy” — another option to treat excess bleeding.
In most patients, she added, progesterone-only therapy “is going to result in irregular bleeding when you first start taking it. That irregular bleeding can last from up to 3-6 months. It’s something that’s really important to discuss with patients.”
She also noted that this therapy can be given to patients in forms that do not prevent pregnancy.
Botero had no disclosures. Wheeler reported relationships with Novo Nordisk, Bayer, BioMarin, Bioverativ, CSL Behring, Genentech, HEMA, Octapharma, Pfizer, Sanofi-Aventis, Shire North America, Spark, and Takeda.
A version of this article first appeared on Medscape.com.
Heavy menstrual bleeding is more than an inconvenience in adolescents: It often leads to significant medical complications, in addition to disruptions in quality of life. While measuring the true level of bleeding can be a challenge, hematologists say treatments are helpful and can be as simple — and surprising — as doses of aspirin.
About 90% of adolescents with heavy menstrual bleeding will have low ferritin, and 70% will develop anemia, said benign hematologist Juliana Perez Botero, MD, of the Mayo Clinic in Rochester, Minnesota, in a presentation at the American Society of Hematology (ASH) 2024 Annual Meeting. “This is an issue of big magnitude that has public health implications, but it’s also an issue of gender equality and social justice.”
Measuring Menstruation: What Counts as Heavy Bleeding?
According to hematologist Allison Wheeler, MD, of the University of Washington in Seattle, normal menstrual bleeding is defined as lasting for about 5 days with 30-50 mL of blood loss.
“Historically, heavy menstrual bleeding was defined as bleeding as > 7 days or > 80 mL of blood loss,” Wheeler said. “It’s pretty hard to measure those mL. So a more modern definition is increased menstrual blood loss that interferes with a female’s physical, social, emotional, or material quality of life.”
Measuring blood loss during menstruation isn’t simple. The alkaline hematin method, which measures blood in feminine hygiene products, is the “gold standard, but that’s all I’m going to say about that,” Wheeler said.
An alternative is the Pictorial Blood Loss Assessment Chart, a visual tool that patients can use to estimate blood loss, she said, although there have been questions about its accuracy. In addition, it only covers pads and tampons even though other products are now available.
Another option is the Adolescent Menstrual Bleeding Questionnaire. “It captures both how much patients are bleeding and a lot of quality-of-life aspects,” she said.
Non-Steroidal Anti-Inflammatory Drugs (NSAIDs), Surprisingly, Can Be Effective Treatments
It seems counterintuitive that NSAIDs can be helpful in patients with heavy bleeding. “We don’t think about them because Cox inhibition ends up leading to decreases in thromboxane A2, which is going to increase bleeding,” Wheeler said. However, she said, the drugs also decrease prostaglandin within the endometrium, which can improve menstrual bleeding.
“In a meta-analysis, when NSAIDs were compared to placebo, there was decreased menstrual bleeding,” she said. “It was pretty similar to estrogen-progesterone formulations together. And when compared to antifibrinolytics, there was a little more menstrual bleeding with the NSAID use.”
Wheeler cautioned that “it’s important to optimize NSAID dosage. You really can’t use lower doses, and you can’t use sporadic doses.”
Other Options: Tranexamic Acid, Hormone Management
Tranexamic acid is the most common antifibrinolytic treatment for heavy bleeding, Wheeler said. “This is a lysine analog that’s going to inhibit the conversion from plasminogen to plasmin,” she said. “This is going to strengthen and stabilize blood clots that are already formed.”
Multiple clinical trials have studied the treatment in heavy menstrual bleeding, she said. “There’s no increased risk of venous or arterial thrombosis among participants in these clinical trials, but patients with a history of thrombosis were excluded from the majority of them. So we do need to keep that in mind.”
As for adverse effects, they’re mild and sporadic and include headaches and stomach upset, she said.
Another treatment option is estrogen and progesterone therapy using pill, patch, or vaginal ring, Wheeler said. The treatment leads to a “stable and thin very endometrium,” and typically takes 3-6 months to fully kick in. Studies have suggested the therapy can lead to 35%-80% reduction in menstrual blood loss, she said.
Keep in mind, Wheeler noted, “that there are many contraindications associated with estrogen use. Please take note of this, especially in consideration of discussions with patients shifting over to progesterone-only hormonal therapy” — another option to treat excess bleeding.
In most patients, she added, progesterone-only therapy “is going to result in irregular bleeding when you first start taking it. That irregular bleeding can last from up to 3-6 months. It’s something that’s really important to discuss with patients.”
She also noted that this therapy can be given to patients in forms that do not prevent pregnancy.
Botero had no disclosures. Wheeler reported relationships with Novo Nordisk, Bayer, BioMarin, Bioverativ, CSL Behring, Genentech, HEMA, Octapharma, Pfizer, Sanofi-Aventis, Shire North America, Spark, and Takeda.
A version of this article first appeared on Medscape.com.
Patients Have Many Fears, Misconceptions About Radiation Therapy
TOPLINE:
A cross-sectional survey of patients preparing for their first radiation therapy consultation found that many patients worried about the physical effects of radiation therapy, including pain, memory loss, and nausea, and more than 60% said they were concerned about their ability to perform daily activities. Respondents reported a range of other worries, including the financial cost of treatment, transportation to treatment sessions, and the ability to continue working, as well as misconceptions about radiation therapy, such as concerns about emitting radiation to others.
METHODOLOGY:
- Toxicities from cancer therapies can significantly affect patients’ quality of life and may contribute to their apprehensions before starting a new treatment. Some studies have indicated that patients may have misconceptions about chemotherapy, but less is known about patients’ perceptions of radiation therapy.
- Researchers conducted a cross-sectional survey of patients presenting for initial radiation therapy consultation at a single academic institution and analyzed responses from 214 patients (52% men; 51% White individuals) with no prior radiation therapy experience.
- The patients completed a 30-question electronic survey about radiation therapy perceptions and fears or concerns prior to their initial radiation consultation.
- Cancer diagnoses spanned 18 disease sites, with hematologic malignancies (21%), breast cancer (18%), and lung cancer (15%) being the most common.
TAKEAWAY:
- Physical adverse effects were the top concern for patients. These included radiation-induced pain (67%), memory loss (62%), nausea/vomiting (60%), and skin reactions (58%).
- Patients expressed concerns about the impact radiation therapy would have on daily activities, with 62% reporting being moderately or very concerned about their ability to perform daily activities and 37% worried about their ability to continue working. Other concerns included the ability to exercise (over half of respondents), financial cost (36%), and transportation to treatment sessions (26%).
- Misconceptions among patients were also common, with 48% expressing concerns about emitting radiation to others and 45% worrying about excreting radioactive urine or stool.
- Patients had varied levels of prior understanding of radiation therapy. Half of patients reported a complete lack of knowledge about radiation therapy, and 35% said they had read or heard stories about bad adverse effects.
IN PRACTICE:
“Our study suggests that a survey administered prior to radiation oncology consultation can reveal patients’ primary concerns which could promote a more patient-centered discussion that addresses specific concerns and involves appropriate services to help the patient,” the authors wrote.
SOURCE:
This study, led by Jennifer Novak, MD, MS, Department of Radiation Oncology, City of Hope National Medical Center, Duarte, California, was published online in Advances in Radiation Oncology.
LIMITATIONS:
Limitations included response bias and time constraints, which prevented many eligible patients from completing the survey. The single-institution design limits the generalizability of the findings. The survey results also showed a disproportionate focus on physical effects over the social impacts of radiation therapy, which could have limited the comprehensiveness of the findings.
DISCLOSURES:
The authors reported no specific funding for this work and no relevant competing financial interests or personal relationships that could have influenced the work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
A cross-sectional survey of patients preparing for their first radiation therapy consultation found that many patients worried about the physical effects of radiation therapy, including pain, memory loss, and nausea, and more than 60% said they were concerned about their ability to perform daily activities. Respondents reported a range of other worries, including the financial cost of treatment, transportation to treatment sessions, and the ability to continue working, as well as misconceptions about radiation therapy, such as concerns about emitting radiation to others.
METHODOLOGY:
- Toxicities from cancer therapies can significantly affect patients’ quality of life and may contribute to their apprehensions before starting a new treatment. Some studies have indicated that patients may have misconceptions about chemotherapy, but less is known about patients’ perceptions of radiation therapy.
- Researchers conducted a cross-sectional survey of patients presenting for initial radiation therapy consultation at a single academic institution and analyzed responses from 214 patients (52% men; 51% White individuals) with no prior radiation therapy experience.
- The patients completed a 30-question electronic survey about radiation therapy perceptions and fears or concerns prior to their initial radiation consultation.
- Cancer diagnoses spanned 18 disease sites, with hematologic malignancies (21%), breast cancer (18%), and lung cancer (15%) being the most common.
TAKEAWAY:
- Physical adverse effects were the top concern for patients. These included radiation-induced pain (67%), memory loss (62%), nausea/vomiting (60%), and skin reactions (58%).
- Patients expressed concerns about the impact radiation therapy would have on daily activities, with 62% reporting being moderately or very concerned about their ability to perform daily activities and 37% worried about their ability to continue working. Other concerns included the ability to exercise (over half of respondents), financial cost (36%), and transportation to treatment sessions (26%).
- Misconceptions among patients were also common, with 48% expressing concerns about emitting radiation to others and 45% worrying about excreting radioactive urine or stool.
- Patients had varied levels of prior understanding of radiation therapy. Half of patients reported a complete lack of knowledge about radiation therapy, and 35% said they had read or heard stories about bad adverse effects.
IN PRACTICE:
“Our study suggests that a survey administered prior to radiation oncology consultation can reveal patients’ primary concerns which could promote a more patient-centered discussion that addresses specific concerns and involves appropriate services to help the patient,” the authors wrote.
SOURCE:
This study, led by Jennifer Novak, MD, MS, Department of Radiation Oncology, City of Hope National Medical Center, Duarte, California, was published online in Advances in Radiation Oncology.
LIMITATIONS:
Limitations included response bias and time constraints, which prevented many eligible patients from completing the survey. The single-institution design limits the generalizability of the findings. The survey results also showed a disproportionate focus on physical effects over the social impacts of radiation therapy, which could have limited the comprehensiveness of the findings.
DISCLOSURES:
The authors reported no specific funding for this work and no relevant competing financial interests or personal relationships that could have influenced the work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
A cross-sectional survey of patients preparing for their first radiation therapy consultation found that many patients worried about the physical effects of radiation therapy, including pain, memory loss, and nausea, and more than 60% said they were concerned about their ability to perform daily activities. Respondents reported a range of other worries, including the financial cost of treatment, transportation to treatment sessions, and the ability to continue working, as well as misconceptions about radiation therapy, such as concerns about emitting radiation to others.
METHODOLOGY:
- Toxicities from cancer therapies can significantly affect patients’ quality of life and may contribute to their apprehensions before starting a new treatment. Some studies have indicated that patients may have misconceptions about chemotherapy, but less is known about patients’ perceptions of radiation therapy.
- Researchers conducted a cross-sectional survey of patients presenting for initial radiation therapy consultation at a single academic institution and analyzed responses from 214 patients (52% men; 51% White individuals) with no prior radiation therapy experience.
- The patients completed a 30-question electronic survey about radiation therapy perceptions and fears or concerns prior to their initial radiation consultation.
- Cancer diagnoses spanned 18 disease sites, with hematologic malignancies (21%), breast cancer (18%), and lung cancer (15%) being the most common.
TAKEAWAY:
- Physical adverse effects were the top concern for patients. These included radiation-induced pain (67%), memory loss (62%), nausea/vomiting (60%), and skin reactions (58%).
- Patients expressed concerns about the impact radiation therapy would have on daily activities, with 62% reporting being moderately or very concerned about their ability to perform daily activities and 37% worried about their ability to continue working. Other concerns included the ability to exercise (over half of respondents), financial cost (36%), and transportation to treatment sessions (26%).
- Misconceptions among patients were also common, with 48% expressing concerns about emitting radiation to others and 45% worrying about excreting radioactive urine or stool.
- Patients had varied levels of prior understanding of radiation therapy. Half of patients reported a complete lack of knowledge about radiation therapy, and 35% said they had read or heard stories about bad adverse effects.
IN PRACTICE:
“Our study suggests that a survey administered prior to radiation oncology consultation can reveal patients’ primary concerns which could promote a more patient-centered discussion that addresses specific concerns and involves appropriate services to help the patient,” the authors wrote.
SOURCE:
This study, led by Jennifer Novak, MD, MS, Department of Radiation Oncology, City of Hope National Medical Center, Duarte, California, was published online in Advances in Radiation Oncology.
LIMITATIONS:
Limitations included response bias and time constraints, which prevented many eligible patients from completing the survey. The single-institution design limits the generalizability of the findings. The survey results also showed a disproportionate focus on physical effects over the social impacts of radiation therapy, which could have limited the comprehensiveness of the findings.
DISCLOSURES:
The authors reported no specific funding for this work and no relevant competing financial interests or personal relationships that could have influenced the work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Compassion Fatigue: How Oncologists Can Recognize the Signs
Last year, Eric Singhi, MD, a thoracic medical oncologist at MD Anderson Cancer Center in Houston, noticed a shift in his team. The small band of four clinicians, once motivated and enthusiastic, were now withdrawn and distant.
“We had to take a break from clinic one day. We went out, got food and…acknowledged something was off,” Singhi recalled.
That something: Intense emotional exhaustion.
No thoracic oncology team has it easy. Lung cancer remains the leading cause of cancer deaths. But over the last 12 months Singhi’s team had taken on more and more cases in younger patients. Watching patients so close to their own age struggle and ultimately be consumed by this disease took its toll, Singhi said. And the toll was showing up in the lives of his colleagues.
Singhi’s team was suffering from compassion fatigue — the mental, physical, or emotional exhaustion brought on by prolonged exposure to the trauma of people in one’s care. Also referred to secondary trauma, compassion fatigue “leaves someone feeling numb, detached, or like they don’t have emotional resources to keep on giving,” Singhi said.
This fatigue is common among people who work in high-stress caretaking professions. Those involved in cancer care, who frequently find themselves navigating life and death conversations and experiences with patients, are particularly at risk, experts told this news organization.
“I think anyone in medical oncology or hematology who has practiced long enough will feel this,” said Alfred Lee, MD, PhD, burnout researcher and director of the hematology/oncology fellowship at Yale School of Medicine in New Haven, Connecticut. “It’s par for the course, even in training.”
Still, compassion fatigue can be hard to detect without help. Left unaddressed, compassion fatigue can undermine good patient care, even lead to burnout.
“I’ve absolutely treated people leaving the field who think they shouldn’t be a doctor anymore,” said Jennifer Bickel, MD, recently named MD Anderson’s inaugural vice president and chief wellness officer. Those are preventable losses the field can’t afford in a provider shortage, she said.
Recognizing the Signs
Compassion is an essential part of patient care as is empathy. Expressing compassion or feeling empathy toward patients can foster the doctor-patient bond and help both navigate challenging conversations and decisions.
“Compassion and being able to empathize are so important — it’s how you gain trust,” Singhi said. This is especially true for patients who may be underrepresented. “That’s how they become a partner in their care, how they agree to listen about a clinical trial or agree to adhere to their treatment plan,” Singhi noted.
But there’s a risk when clinicians start to give too much.
Overtime, repeated exposure to patients’ trauma can create a sense of heaviness and fatigue, and ultimately cause caregivers to detach, Lee said.
A case study of compassion fatigue, based on the experience of one oncologist, highlighted that the condition parallels posttraumatic stress disorder, aligning on three main symptoms: Feeling hyperarousal, notably hypervigilance or irritability, avoiding stressful situations, and reliving traumatic events.
The initial phases of compassion fatigue, however, can be subtle and easily dismissed. An oncologist may begin to feel a creeping sense of detachment — a divide slowly growing with their patients, a buzz of dissatisfaction on the job, unusual mood changes.
While it’s normal for healthcare workers to experience moments of irritability, dissatisfaction, exhaustion or avoidance, when these experiences become pervasive, compassion fatigue could be the culprit, according to Marra Ackerman, MD, a psychiatrist at NYU Langone Health in New York City.
Compassion fatigue is “a form of coping more than anything,” Ackerman explained. Most of the time, it’s not a conscious choice, she said. When a brain experiences repeated distressing inputs it can shift into self-protection mode. The tipping point is different for everyone, but eventually the brain tries to detach and distance itself from the stressor, Ackerman said. And, in oncology, that stressor is often very sick patients.
If not addressed, the emotional exhaustion can limit a doctor’s efficacy at work. Clinicians suffering from compassion fatigue may find themselves calling in sick more often, making more mistakes, avoiding very sick patients, or having panic attacks at work, said Ackerman.
Clinicians also might use food, drugs, or alcohol to feel better, or detach from their patients, Lee added. “We find if you detach, you can’t provide the care patients need,” he said.
When trying to identify the issue, it’s important to understand that compassion fatigue is not burnout. The two issues can look similar, sharing symptoms like emotional detachment or disillusionment, and may co-exist, but the phenomena stem from different root causes.
While compassion fatigue is rooted in emotions and isn’t necessarily tied to work, burnout is a product of work-related stress. It’s often caused by system-level problems that leave clinicians feeling their work has lost its meaning. Inefficient technology or excessive data entry, for instance, could be drivers of burnout for oncologists because these tasks can pull the doctor away from the meaningful work of treating patients.
What Can Be Done
Oncology comes with its own set of risks and traumas, but for a long time, doctors weren’t equipped or encouraged to see the constant life-or-death tension of the job as a risk factor to their well-being, said Bickel.
“It’s not something we are inherently taught to identify and it’s not something we can identify without time for self-reflection,” Bickel told this news organization. In fact, many people don’t see their own compassion fatigue until a colleague points it out or they’re already past it and have some distance.
Although common among oncology clinicians, compassion fatigue is not inevitable. Oncologists can learn strategies to help manage their exposures to suffering and stave off reverting to coping mechanisms that only perpetuate the problem.
The key, Bickel said, is creating environments where people can feel safe and share their vulnerabilities. Formal peer support check-ins are a good example because they normalize regular communication about day-to-day difficulties as they happen and create space to experience grief. In these groups, no one has to worry that they’re complaining, she said.
Formalized groups also serve as a training ground where staff can learn to be better listeners. Healthcare providers are more likely to reach out to peers because so few people outside their field can relate to the challenges, Bickel explained. That’s why Moffitt Cancer Center, where Bickel recently worked before moving to MD Anderson, offers trainings to help staff learn to listen more effectively. Thirty doctors, nurses and health professionals have received the training so far and serve as a point of contact for colleagues who might be struggling, Bickel said. They can offer their peers an empathic ear, without providing unsolicited advice or commentary.
NYU Langone has a similar offering, Ackerman said. Providers can reach out to fellow nurses or doctors who have more mental health training. The conversation isn’t recorded like an appointment with a therapist may be. It’s more like mentorship where doctors can be open about what they are facing and find some comradery from someone who understands, without fear of stigma.
Talking to colleagues or having debriefs with your care team is critical because it eliminates the isolation that allows compassion fatigue to fester, Lee said. “Reflecting on positive encounters can help inspire us to keep going. Reflecting on the negative can be therapeutic and help find that sense of balance that everyone needs to find.”
As for Singhi’s team, “lots has changed since we’ve become more open,” he said. The team talks about hard patients early and often. These discussions have helped team members keep the challenges at work from seeping into their home life.
Experts agree that the threshold for seeking out help and guidance should be low. Waiting for depression or more severe symptoms to set in is not the best point of intervention, Bickel said. As soon as an oncologist begins to acknowledge a shift in how they connect with patients and the job, that’s enough to reach out.
“It’s not about what you can handle, it’s about how can you thrive and find joy,” she said.
A version of this article first appeared on Medscape.com.
Last year, Eric Singhi, MD, a thoracic medical oncologist at MD Anderson Cancer Center in Houston, noticed a shift in his team. The small band of four clinicians, once motivated and enthusiastic, were now withdrawn and distant.
“We had to take a break from clinic one day. We went out, got food and…acknowledged something was off,” Singhi recalled.
That something: Intense emotional exhaustion.
No thoracic oncology team has it easy. Lung cancer remains the leading cause of cancer deaths. But over the last 12 months Singhi’s team had taken on more and more cases in younger patients. Watching patients so close to their own age struggle and ultimately be consumed by this disease took its toll, Singhi said. And the toll was showing up in the lives of his colleagues.
Singhi’s team was suffering from compassion fatigue — the mental, physical, or emotional exhaustion brought on by prolonged exposure to the trauma of people in one’s care. Also referred to secondary trauma, compassion fatigue “leaves someone feeling numb, detached, or like they don’t have emotional resources to keep on giving,” Singhi said.
This fatigue is common among people who work in high-stress caretaking professions. Those involved in cancer care, who frequently find themselves navigating life and death conversations and experiences with patients, are particularly at risk, experts told this news organization.
“I think anyone in medical oncology or hematology who has practiced long enough will feel this,” said Alfred Lee, MD, PhD, burnout researcher and director of the hematology/oncology fellowship at Yale School of Medicine in New Haven, Connecticut. “It’s par for the course, even in training.”
Still, compassion fatigue can be hard to detect without help. Left unaddressed, compassion fatigue can undermine good patient care, even lead to burnout.
“I’ve absolutely treated people leaving the field who think they shouldn’t be a doctor anymore,” said Jennifer Bickel, MD, recently named MD Anderson’s inaugural vice president and chief wellness officer. Those are preventable losses the field can’t afford in a provider shortage, she said.
Recognizing the Signs
Compassion is an essential part of patient care as is empathy. Expressing compassion or feeling empathy toward patients can foster the doctor-patient bond and help both navigate challenging conversations and decisions.
“Compassion and being able to empathize are so important — it’s how you gain trust,” Singhi said. This is especially true for patients who may be underrepresented. “That’s how they become a partner in their care, how they agree to listen about a clinical trial or agree to adhere to their treatment plan,” Singhi noted.
But there’s a risk when clinicians start to give too much.
Overtime, repeated exposure to patients’ trauma can create a sense of heaviness and fatigue, and ultimately cause caregivers to detach, Lee said.
A case study of compassion fatigue, based on the experience of one oncologist, highlighted that the condition parallels posttraumatic stress disorder, aligning on three main symptoms: Feeling hyperarousal, notably hypervigilance or irritability, avoiding stressful situations, and reliving traumatic events.
The initial phases of compassion fatigue, however, can be subtle and easily dismissed. An oncologist may begin to feel a creeping sense of detachment — a divide slowly growing with their patients, a buzz of dissatisfaction on the job, unusual mood changes.
While it’s normal for healthcare workers to experience moments of irritability, dissatisfaction, exhaustion or avoidance, when these experiences become pervasive, compassion fatigue could be the culprit, according to Marra Ackerman, MD, a psychiatrist at NYU Langone Health in New York City.
Compassion fatigue is “a form of coping more than anything,” Ackerman explained. Most of the time, it’s not a conscious choice, she said. When a brain experiences repeated distressing inputs it can shift into self-protection mode. The tipping point is different for everyone, but eventually the brain tries to detach and distance itself from the stressor, Ackerman said. And, in oncology, that stressor is often very sick patients.
If not addressed, the emotional exhaustion can limit a doctor’s efficacy at work. Clinicians suffering from compassion fatigue may find themselves calling in sick more often, making more mistakes, avoiding very sick patients, or having panic attacks at work, said Ackerman.
Clinicians also might use food, drugs, or alcohol to feel better, or detach from their patients, Lee added. “We find if you detach, you can’t provide the care patients need,” he said.
When trying to identify the issue, it’s important to understand that compassion fatigue is not burnout. The two issues can look similar, sharing symptoms like emotional detachment or disillusionment, and may co-exist, but the phenomena stem from different root causes.
While compassion fatigue is rooted in emotions and isn’t necessarily tied to work, burnout is a product of work-related stress. It’s often caused by system-level problems that leave clinicians feeling their work has lost its meaning. Inefficient technology or excessive data entry, for instance, could be drivers of burnout for oncologists because these tasks can pull the doctor away from the meaningful work of treating patients.
What Can Be Done
Oncology comes with its own set of risks and traumas, but for a long time, doctors weren’t equipped or encouraged to see the constant life-or-death tension of the job as a risk factor to their well-being, said Bickel.
“It’s not something we are inherently taught to identify and it’s not something we can identify without time for self-reflection,” Bickel told this news organization. In fact, many people don’t see their own compassion fatigue until a colleague points it out or they’re already past it and have some distance.
Although common among oncology clinicians, compassion fatigue is not inevitable. Oncologists can learn strategies to help manage their exposures to suffering and stave off reverting to coping mechanisms that only perpetuate the problem.
The key, Bickel said, is creating environments where people can feel safe and share their vulnerabilities. Formal peer support check-ins are a good example because they normalize regular communication about day-to-day difficulties as they happen and create space to experience grief. In these groups, no one has to worry that they’re complaining, she said.
Formalized groups also serve as a training ground where staff can learn to be better listeners. Healthcare providers are more likely to reach out to peers because so few people outside their field can relate to the challenges, Bickel explained. That’s why Moffitt Cancer Center, where Bickel recently worked before moving to MD Anderson, offers trainings to help staff learn to listen more effectively. Thirty doctors, nurses and health professionals have received the training so far and serve as a point of contact for colleagues who might be struggling, Bickel said. They can offer their peers an empathic ear, without providing unsolicited advice or commentary.
NYU Langone has a similar offering, Ackerman said. Providers can reach out to fellow nurses or doctors who have more mental health training. The conversation isn’t recorded like an appointment with a therapist may be. It’s more like mentorship where doctors can be open about what they are facing and find some comradery from someone who understands, without fear of stigma.
Talking to colleagues or having debriefs with your care team is critical because it eliminates the isolation that allows compassion fatigue to fester, Lee said. “Reflecting on positive encounters can help inspire us to keep going. Reflecting on the negative can be therapeutic and help find that sense of balance that everyone needs to find.”
As for Singhi’s team, “lots has changed since we’ve become more open,” he said. The team talks about hard patients early and often. These discussions have helped team members keep the challenges at work from seeping into their home life.
Experts agree that the threshold for seeking out help and guidance should be low. Waiting for depression or more severe symptoms to set in is not the best point of intervention, Bickel said. As soon as an oncologist begins to acknowledge a shift in how they connect with patients and the job, that’s enough to reach out.
“It’s not about what you can handle, it’s about how can you thrive and find joy,” she said.
A version of this article first appeared on Medscape.com.
Last year, Eric Singhi, MD, a thoracic medical oncologist at MD Anderson Cancer Center in Houston, noticed a shift in his team. The small band of four clinicians, once motivated and enthusiastic, were now withdrawn and distant.
“We had to take a break from clinic one day. We went out, got food and…acknowledged something was off,” Singhi recalled.
That something: Intense emotional exhaustion.
No thoracic oncology team has it easy. Lung cancer remains the leading cause of cancer deaths. But over the last 12 months Singhi’s team had taken on more and more cases in younger patients. Watching patients so close to their own age struggle and ultimately be consumed by this disease took its toll, Singhi said. And the toll was showing up in the lives of his colleagues.
Singhi’s team was suffering from compassion fatigue — the mental, physical, or emotional exhaustion brought on by prolonged exposure to the trauma of people in one’s care. Also referred to secondary trauma, compassion fatigue “leaves someone feeling numb, detached, or like they don’t have emotional resources to keep on giving,” Singhi said.
This fatigue is common among people who work in high-stress caretaking professions. Those involved in cancer care, who frequently find themselves navigating life and death conversations and experiences with patients, are particularly at risk, experts told this news organization.
“I think anyone in medical oncology or hematology who has practiced long enough will feel this,” said Alfred Lee, MD, PhD, burnout researcher and director of the hematology/oncology fellowship at Yale School of Medicine in New Haven, Connecticut. “It’s par for the course, even in training.”
Still, compassion fatigue can be hard to detect without help. Left unaddressed, compassion fatigue can undermine good patient care, even lead to burnout.
“I’ve absolutely treated people leaving the field who think they shouldn’t be a doctor anymore,” said Jennifer Bickel, MD, recently named MD Anderson’s inaugural vice president and chief wellness officer. Those are preventable losses the field can’t afford in a provider shortage, she said.
Recognizing the Signs
Compassion is an essential part of patient care as is empathy. Expressing compassion or feeling empathy toward patients can foster the doctor-patient bond and help both navigate challenging conversations and decisions.
“Compassion and being able to empathize are so important — it’s how you gain trust,” Singhi said. This is especially true for patients who may be underrepresented. “That’s how they become a partner in their care, how they agree to listen about a clinical trial or agree to adhere to their treatment plan,” Singhi noted.
But there’s a risk when clinicians start to give too much.
Overtime, repeated exposure to patients’ trauma can create a sense of heaviness and fatigue, and ultimately cause caregivers to detach, Lee said.
A case study of compassion fatigue, based on the experience of one oncologist, highlighted that the condition parallels posttraumatic stress disorder, aligning on three main symptoms: Feeling hyperarousal, notably hypervigilance or irritability, avoiding stressful situations, and reliving traumatic events.
The initial phases of compassion fatigue, however, can be subtle and easily dismissed. An oncologist may begin to feel a creeping sense of detachment — a divide slowly growing with their patients, a buzz of dissatisfaction on the job, unusual mood changes.
While it’s normal for healthcare workers to experience moments of irritability, dissatisfaction, exhaustion or avoidance, when these experiences become pervasive, compassion fatigue could be the culprit, according to Marra Ackerman, MD, a psychiatrist at NYU Langone Health in New York City.
Compassion fatigue is “a form of coping more than anything,” Ackerman explained. Most of the time, it’s not a conscious choice, she said. When a brain experiences repeated distressing inputs it can shift into self-protection mode. The tipping point is different for everyone, but eventually the brain tries to detach and distance itself from the stressor, Ackerman said. And, in oncology, that stressor is often very sick patients.
If not addressed, the emotional exhaustion can limit a doctor’s efficacy at work. Clinicians suffering from compassion fatigue may find themselves calling in sick more often, making more mistakes, avoiding very sick patients, or having panic attacks at work, said Ackerman.
Clinicians also might use food, drugs, or alcohol to feel better, or detach from their patients, Lee added. “We find if you detach, you can’t provide the care patients need,” he said.
When trying to identify the issue, it’s important to understand that compassion fatigue is not burnout. The two issues can look similar, sharing symptoms like emotional detachment or disillusionment, and may co-exist, but the phenomena stem from different root causes.
While compassion fatigue is rooted in emotions and isn’t necessarily tied to work, burnout is a product of work-related stress. It’s often caused by system-level problems that leave clinicians feeling their work has lost its meaning. Inefficient technology or excessive data entry, for instance, could be drivers of burnout for oncologists because these tasks can pull the doctor away from the meaningful work of treating patients.
What Can Be Done
Oncology comes with its own set of risks and traumas, but for a long time, doctors weren’t equipped or encouraged to see the constant life-or-death tension of the job as a risk factor to their well-being, said Bickel.
“It’s not something we are inherently taught to identify and it’s not something we can identify without time for self-reflection,” Bickel told this news organization. In fact, many people don’t see their own compassion fatigue until a colleague points it out or they’re already past it and have some distance.
Although common among oncology clinicians, compassion fatigue is not inevitable. Oncologists can learn strategies to help manage their exposures to suffering and stave off reverting to coping mechanisms that only perpetuate the problem.
The key, Bickel said, is creating environments where people can feel safe and share their vulnerabilities. Formal peer support check-ins are a good example because they normalize regular communication about day-to-day difficulties as they happen and create space to experience grief. In these groups, no one has to worry that they’re complaining, she said.
Formalized groups also serve as a training ground where staff can learn to be better listeners. Healthcare providers are more likely to reach out to peers because so few people outside their field can relate to the challenges, Bickel explained. That’s why Moffitt Cancer Center, where Bickel recently worked before moving to MD Anderson, offers trainings to help staff learn to listen more effectively. Thirty doctors, nurses and health professionals have received the training so far and serve as a point of contact for colleagues who might be struggling, Bickel said. They can offer their peers an empathic ear, without providing unsolicited advice or commentary.
NYU Langone has a similar offering, Ackerman said. Providers can reach out to fellow nurses or doctors who have more mental health training. The conversation isn’t recorded like an appointment with a therapist may be. It’s more like mentorship where doctors can be open about what they are facing and find some comradery from someone who understands, without fear of stigma.
Talking to colleagues or having debriefs with your care team is critical because it eliminates the isolation that allows compassion fatigue to fester, Lee said. “Reflecting on positive encounters can help inspire us to keep going. Reflecting on the negative can be therapeutic and help find that sense of balance that everyone needs to find.”
As for Singhi’s team, “lots has changed since we’ve become more open,” he said. The team talks about hard patients early and often. These discussions have helped team members keep the challenges at work from seeping into their home life.
Experts agree that the threshold for seeking out help and guidance should be low. Waiting for depression or more severe symptoms to set in is not the best point of intervention, Bickel said. As soon as an oncologist begins to acknowledge a shift in how they connect with patients and the job, that’s enough to reach out.
“It’s not about what you can handle, it’s about how can you thrive and find joy,” she said.
A version of this article first appeared on Medscape.com.
Environmental and Socioeconomic Factors Fuel Respiratory Health Disparities in Rural and Urban Areas
In 2016, Brady Scott was in his parents’ home in Garrett, Kentucky, scrolling his Facebook feed when a post from a local newspaper caught his attention. “The article said that if you grew up in the region I grew up in, compared to the richer Central Kentucky region, the life expectancy differed by about 9 years,” he recalled.
The respiratory therapist, then a PhD student at Rush University, Chicago, was struck and began “Googling” to find out why this was the case. Initially, he thought diabetes, smoking, and economic distress — all prevalent problems in the area — were the culprits. However, he soon found that respiratory disease was particularly common in his region.
Now a professor and program director of the Respiratory Care Program at Rush University, Scott has spent several years trying to understand why people in certain regions experience respiratory illness at higher rates than in other places.
The Environment as a Determinant of Health
When Scott began his PhD, the prevalence of asthma in Southeast Kentucky, part of the Appalachian region, was already well-documented. He focused his research on uncontrolled asthma and the triggers that drove asthma exacerbations.
Housing quality emerged as an important factor. He found that exposure to mold, mildew, dust mites, pests, and rodents increased the risk for asthma and exacerbated existing cases. Lower-income families, more likely to live in poor-quality housing, were significantly affected, even in single-family homes.
Wanda Phipatanakul, MD, MS, director of the Division of Immunology Research Center at Boston Children’s Hospital and S. Jean Emans professor of Pediatrics at Harvard Medical School, Boston, has found similar results in urban environments. She said cockroach and mouse allergen exposure is disproportionately prevalent in urban, low-income neighborhoods. These exposures, closely tied to housing conditions, contribute to worse asthma and respiratory problems, particularly in children.
Scott and Phipatanakul agreed that the environment surrounding people’s homes can also exacerbate respiratory disease.
Rural areas present unique risks, such as agricultural activities that release pesticides and other particulates into the air, said Scott. In mountainous areas like Appalachia, mining operations are another significant contributor. For example, blasting mountains with dynamite creates large clouds of dust and pollutants that settle in valleys. Coal-hauling roads contribute to air quality issues, too. And houses near these roads may be exposed to increased levels of particulate matter, he said.
In the city, Phipatanakul has found that historical practices like redlining have systematically denied certain neighborhoods access to resources and investment, leaving a legacy of poor infrastructure, limited resources, and higher exposure to environmental risks. Today, these areas have more highways and fewer green spaces and are disproportionately linked with a higher incidence of respiratory illnesses.
The findings of both Scott and Phipatanakul underscore a critical bottom line: Health disparities are deeply influenced by environmental factors, which are themselves shaped by socioeconomic conditions and historical inequities. Poor housing quality, exposure to allergens, and proximity to environmental hazards disproportionately affect underserved and minority communities, whether in rural or urban settings.
The Role of Green Spaces in Improving Respiratory Health
Restoring and increasing tree cover and green spaces in urban areas can significantly improve respiratory health by addressing environmental challenges and reducing triggers for respiratory issues. Areas with greater greenness tend to have lower levels of pollutants and fewer environmental infestations, such as mice and cockroaches, explained Phipatanakul. Her research highlights that schools in greener areas have fewer airborne pollutants and particles than those in more urbanized, less green areas, which are usually in poorer suburbs.
Trees absorb pollutants such as particulate matter and sulfur dioxide through dry deposition and stomatal uptake, improving air quality. “The question is whether we can use trees as a public health tool, and this is being done in many cities,” said Alessandro Marcon, PhD, a professor of epidemiology and medical statistics at the University of Verona, Verona, Italy, while speaking at the European Respiratory Society conference held in Vienna last September.
A US analysis showed that existing natural vegetation, such as forests and grasslands, absorbs a large portion of emissions. By restoring land cover, pollution from harmful substances like sulfur dioxide and particulate matter could be reduced by about 30%. This approach is often more cost-effective than technological solutions for managing emissions.
Moreover, tree cover contributes to a healthier air microbiome. Research indicates that urban forest areas have lower pathogenic bacteria and fungi concentrations than nearby urban zones.
Another major advantage is the mitigation of the urban heat island effect. A study conducted in Paris found that municipalities with higher tree coverage experienced 20%-30% lower heat-related mortality than those with less greenery. Increasing tree coverage to 30% could reduce up to 40% of excess mortality associated with urban heat islands. Trees achieve this by providing shade and facilitating evapotranspiration, which cools the surrounding air.
Urban environments, unsurprisingly, often have higher levels of air pollution due to increased traffic and industrial activity. However, despite appearing greener, rural environments may harbor less obvious but significant sources of air pollution. “I live in an urban environment now, but I grew up in a rural environment,” Scott said. “Each has its own issues that affect air quality and health.”
Scott, Phipatanakul, and Marcon reported no relevant financial relationships.■
A version of this article first appeared on Medscape.com.
In 2016, Brady Scott was in his parents’ home in Garrett, Kentucky, scrolling his Facebook feed when a post from a local newspaper caught his attention. “The article said that if you grew up in the region I grew up in, compared to the richer Central Kentucky region, the life expectancy differed by about 9 years,” he recalled.
The respiratory therapist, then a PhD student at Rush University, Chicago, was struck and began “Googling” to find out why this was the case. Initially, he thought diabetes, smoking, and economic distress — all prevalent problems in the area — were the culprits. However, he soon found that respiratory disease was particularly common in his region.
Now a professor and program director of the Respiratory Care Program at Rush University, Scott has spent several years trying to understand why people in certain regions experience respiratory illness at higher rates than in other places.
The Environment as a Determinant of Health
When Scott began his PhD, the prevalence of asthma in Southeast Kentucky, part of the Appalachian region, was already well-documented. He focused his research on uncontrolled asthma and the triggers that drove asthma exacerbations.
Housing quality emerged as an important factor. He found that exposure to mold, mildew, dust mites, pests, and rodents increased the risk for asthma and exacerbated existing cases. Lower-income families, more likely to live in poor-quality housing, were significantly affected, even in single-family homes.
Wanda Phipatanakul, MD, MS, director of the Division of Immunology Research Center at Boston Children’s Hospital and S. Jean Emans professor of Pediatrics at Harvard Medical School, Boston, has found similar results in urban environments. She said cockroach and mouse allergen exposure is disproportionately prevalent in urban, low-income neighborhoods. These exposures, closely tied to housing conditions, contribute to worse asthma and respiratory problems, particularly in children.
Scott and Phipatanakul agreed that the environment surrounding people’s homes can also exacerbate respiratory disease.
Rural areas present unique risks, such as agricultural activities that release pesticides and other particulates into the air, said Scott. In mountainous areas like Appalachia, mining operations are another significant contributor. For example, blasting mountains with dynamite creates large clouds of dust and pollutants that settle in valleys. Coal-hauling roads contribute to air quality issues, too. And houses near these roads may be exposed to increased levels of particulate matter, he said.
In the city, Phipatanakul has found that historical practices like redlining have systematically denied certain neighborhoods access to resources and investment, leaving a legacy of poor infrastructure, limited resources, and higher exposure to environmental risks. Today, these areas have more highways and fewer green spaces and are disproportionately linked with a higher incidence of respiratory illnesses.
The findings of both Scott and Phipatanakul underscore a critical bottom line: Health disparities are deeply influenced by environmental factors, which are themselves shaped by socioeconomic conditions and historical inequities. Poor housing quality, exposure to allergens, and proximity to environmental hazards disproportionately affect underserved and minority communities, whether in rural or urban settings.
The Role of Green Spaces in Improving Respiratory Health
Restoring and increasing tree cover and green spaces in urban areas can significantly improve respiratory health by addressing environmental challenges and reducing triggers for respiratory issues. Areas with greater greenness tend to have lower levels of pollutants and fewer environmental infestations, such as mice and cockroaches, explained Phipatanakul. Her research highlights that schools in greener areas have fewer airborne pollutants and particles than those in more urbanized, less green areas, which are usually in poorer suburbs.
Trees absorb pollutants such as particulate matter and sulfur dioxide through dry deposition and stomatal uptake, improving air quality. “The question is whether we can use trees as a public health tool, and this is being done in many cities,” said Alessandro Marcon, PhD, a professor of epidemiology and medical statistics at the University of Verona, Verona, Italy, while speaking at the European Respiratory Society conference held in Vienna last September.
A US analysis showed that existing natural vegetation, such as forests and grasslands, absorbs a large portion of emissions. By restoring land cover, pollution from harmful substances like sulfur dioxide and particulate matter could be reduced by about 30%. This approach is often more cost-effective than technological solutions for managing emissions.
Moreover, tree cover contributes to a healthier air microbiome. Research indicates that urban forest areas have lower pathogenic bacteria and fungi concentrations than nearby urban zones.
Another major advantage is the mitigation of the urban heat island effect. A study conducted in Paris found that municipalities with higher tree coverage experienced 20%-30% lower heat-related mortality than those with less greenery. Increasing tree coverage to 30% could reduce up to 40% of excess mortality associated with urban heat islands. Trees achieve this by providing shade and facilitating evapotranspiration, which cools the surrounding air.
Urban environments, unsurprisingly, often have higher levels of air pollution due to increased traffic and industrial activity. However, despite appearing greener, rural environments may harbor less obvious but significant sources of air pollution. “I live in an urban environment now, but I grew up in a rural environment,” Scott said. “Each has its own issues that affect air quality and health.”
Scott, Phipatanakul, and Marcon reported no relevant financial relationships.■
A version of this article first appeared on Medscape.com.
In 2016, Brady Scott was in his parents’ home in Garrett, Kentucky, scrolling his Facebook feed when a post from a local newspaper caught his attention. “The article said that if you grew up in the region I grew up in, compared to the richer Central Kentucky region, the life expectancy differed by about 9 years,” he recalled.
The respiratory therapist, then a PhD student at Rush University, Chicago, was struck and began “Googling” to find out why this was the case. Initially, he thought diabetes, smoking, and economic distress — all prevalent problems in the area — were the culprits. However, he soon found that respiratory disease was particularly common in his region.
Now a professor and program director of the Respiratory Care Program at Rush University, Scott has spent several years trying to understand why people in certain regions experience respiratory illness at higher rates than in other places.
The Environment as a Determinant of Health
When Scott began his PhD, the prevalence of asthma in Southeast Kentucky, part of the Appalachian region, was already well-documented. He focused his research on uncontrolled asthma and the triggers that drove asthma exacerbations.
Housing quality emerged as an important factor. He found that exposure to mold, mildew, dust mites, pests, and rodents increased the risk for asthma and exacerbated existing cases. Lower-income families, more likely to live in poor-quality housing, were significantly affected, even in single-family homes.
Wanda Phipatanakul, MD, MS, director of the Division of Immunology Research Center at Boston Children’s Hospital and S. Jean Emans professor of Pediatrics at Harvard Medical School, Boston, has found similar results in urban environments. She said cockroach and mouse allergen exposure is disproportionately prevalent in urban, low-income neighborhoods. These exposures, closely tied to housing conditions, contribute to worse asthma and respiratory problems, particularly in children.
Scott and Phipatanakul agreed that the environment surrounding people’s homes can also exacerbate respiratory disease.
Rural areas present unique risks, such as agricultural activities that release pesticides and other particulates into the air, said Scott. In mountainous areas like Appalachia, mining operations are another significant contributor. For example, blasting mountains with dynamite creates large clouds of dust and pollutants that settle in valleys. Coal-hauling roads contribute to air quality issues, too. And houses near these roads may be exposed to increased levels of particulate matter, he said.
In the city, Phipatanakul has found that historical practices like redlining have systematically denied certain neighborhoods access to resources and investment, leaving a legacy of poor infrastructure, limited resources, and higher exposure to environmental risks. Today, these areas have more highways and fewer green spaces and are disproportionately linked with a higher incidence of respiratory illnesses.
The findings of both Scott and Phipatanakul underscore a critical bottom line: Health disparities are deeply influenced by environmental factors, which are themselves shaped by socioeconomic conditions and historical inequities. Poor housing quality, exposure to allergens, and proximity to environmental hazards disproportionately affect underserved and minority communities, whether in rural or urban settings.
The Role of Green Spaces in Improving Respiratory Health
Restoring and increasing tree cover and green spaces in urban areas can significantly improve respiratory health by addressing environmental challenges and reducing triggers for respiratory issues. Areas with greater greenness tend to have lower levels of pollutants and fewer environmental infestations, such as mice and cockroaches, explained Phipatanakul. Her research highlights that schools in greener areas have fewer airborne pollutants and particles than those in more urbanized, less green areas, which are usually in poorer suburbs.
Trees absorb pollutants such as particulate matter and sulfur dioxide through dry deposition and stomatal uptake, improving air quality. “The question is whether we can use trees as a public health tool, and this is being done in many cities,” said Alessandro Marcon, PhD, a professor of epidemiology and medical statistics at the University of Verona, Verona, Italy, while speaking at the European Respiratory Society conference held in Vienna last September.
A US analysis showed that existing natural vegetation, such as forests and grasslands, absorbs a large portion of emissions. By restoring land cover, pollution from harmful substances like sulfur dioxide and particulate matter could be reduced by about 30%. This approach is often more cost-effective than technological solutions for managing emissions.
Moreover, tree cover contributes to a healthier air microbiome. Research indicates that urban forest areas have lower pathogenic bacteria and fungi concentrations than nearby urban zones.
Another major advantage is the mitigation of the urban heat island effect. A study conducted in Paris found that municipalities with higher tree coverage experienced 20%-30% lower heat-related mortality than those with less greenery. Increasing tree coverage to 30% could reduce up to 40% of excess mortality associated with urban heat islands. Trees achieve this by providing shade and facilitating evapotranspiration, which cools the surrounding air.
Urban environments, unsurprisingly, often have higher levels of air pollution due to increased traffic and industrial activity. However, despite appearing greener, rural environments may harbor less obvious but significant sources of air pollution. “I live in an urban environment now, but I grew up in a rural environment,” Scott said. “Each has its own issues that affect air quality and health.”
Scott, Phipatanakul, and Marcon reported no relevant financial relationships.■
A version of this article first appeared on Medscape.com.
Avoid Getting Stuck: A Practical Guide to Managing Chronic Constipation
Introduction
Constipation affects one in six people worldwide and accounts for one third of outpatient visits.1 Chronic constipation is defined by difficult, infrequent, and/or incomplete defecation, quantified by less than three spontaneous bowel movements per week, persisting for at least 3 months. Patients may complain of straining during defecation, incomplete evacuation, hard stools (Bristol stool scale [BSS] type 1-2), and fullness or bloating. Chronic constipation can be subclassified as either a primary or secondary disorder.1,2
Primary Constipation Disorders
Primary constipation includes disorders of the colon or anorectum. This includes irritable bowel syndrome with constipation (IBS-C), chronic idiopathic constipation (CIC), slow transit constipation (STC), dyssynergic defecation, and pelvic floor disorders (see Figure 1).

IBS-C
IBS-C is a chronic disorder of the gut-brain axis with a worldwide prevalence of 1.3% and a prevalence of 6%-16% in the United States, United Kingdom, and Canada, with females more likely to seek care than males.2 The economic impact of IBS-C is estimated to be $1.5 billion–$10 billion per year in the United States alone.3 The distinguishing characteristic is abdominal pain, however IBS-C can present with a constellation of symptoms. The diagnostic paradigm has shifted from IBS being a diagnosis of exclusion to now using a positive diagnostic strategy.2 Using this Rome IV criteria, one can make the diagnosis with > 95% accuracy.2,4
CIC
CIC, previously defined as functional constipation, is a disorder defined by incomplete defecation and difficult or infrequent stool. CIC is diagnosed in patients without an underlying anatomic or structural abnormality. Rome IV Criteria helps further classify the defining characteristics of chronic idiopathic constipation.2
Slow Transit Constipation
STC is characterized by impaired colonic transit time in the absence of pelvic floor dysfunction. It presents with infrequent bowel movements, diminished urgency, and/or straining with defecation.
Defecatory Disorders: Dyssynergic Defecation and Pelvic Floor Dysfunction
Defecatory disorders (DDs) result from alterations in the colonic-neural pathway with an unclear pathogenesis. A firm understanding of colonic physiology is necessary to identify DDs. The right colon helps to store and mix stool contents, the left colon helps add water to the stool, and the anal canal and rectum enable defecation and maintain continence. Any alteration along this physiologic pathway results in DDs.5
DDs primarily develop via maladaptive pelvic floor contraction during defecation or from muscle or nerve injury and include functional outlet obstruction, anorectal dyssynergia, and pelvic floor dysfunction. Increased resistance to defecation results from anismus, paradoxical anal sphincter contraction, or incomplete relaxation of the pelvic floor and external anal sphincter. This muscle incoordination is described as dyssynergia. DDs can involve either muscle or nerve dysfunction or a combination of the two. Reduced rectal sensation caused by reduced sensory triggers can cause stasis of stool, thus propagating the cycle of constipation. Over time, excessive straining can weaken the pelvic floor, increasing the risk of excessive perineal descent, rectal intussusception, solitary rectal ulcer syndrome, and pudendal neuropathy.5 Thus, identification of DDs is crucial in patients with chronic constipation.
Secondary Constipation Disorders
Secondary constipation disorders are a result of an alternate process and warrant a thorough review of outpatient medications and past medical history. Figure 1 outlines the most common causes of secondary constipation, which span a wide differential.
Clinical Evaluation
The evaluation of constipation begins with a thorough history. Description of bowel habits should include frequency, duration, straining, stool consistency using a Bristol stool chart, complete vs incomplete evacuation, pain, bloating, and use of digital maneuvers (vaginal splinting or digital stool removal). One should inquire about back trauma/surgeries and obstetric history to include vaginal forceps injury or episiotomy.
With increased smartphone use, toilet time on average has increased and can contribute to maladaptive bowel habits.6 Patients may not realize they are constipated, so patient education is critical. A patient with daily bowel movements ranging between BSS type 1-6 with incomplete evacuation might complain of diarrhea but may in fact have constipation with overflow diarrhea, for example. Past medical history is also clinically relevant, as systemic conditions can cause secondary constipation. A constipated patient should also be asked what therapies he/she has tried prior to gastroenterology referral as primary care referrals for constipation account for 8 million visits to gastroenterology per year.7
While a sensitive topic, inquire about abuse history, especially in those with childhood constipation symptoms. There is a positive correlation between childhood constipation and physical, emotional, and sexual abuse and, for any number of reasons, your patient may be reluctant to share this or undergo a digital rectal exam (DRE).8 In such cases, be sensitive in asking for this history in private rather than with other family members around and always perform this exam with a chaperone present.
A detailed physical exam is an indispensable tool all gastroenterologists must master when evaluating a constipated patient. Some key exam findings include abdominal distention, high-pitched bowel sounds, and presence of a succussion splash indicating obstructive pathology. Dry skin and brittle hair indicate hypothyroidism while hypermobile joints and skin laxity suggest connective tissue disease. Finally, a physical examination is incomplete without a DRE.
DRE
DRE is an often-overlooked physical exam component which provides helpful insight that can guide management. An informed DRE can help identify structural disorders such as fissure, hemorrhoids, anorectal mass, fecal impaction, rectal prolapse, and excessive perineal descent syndrome.9 Unless contraindicated, DRE should be a standard part of the workup of a patient with chronic constipation.
Workup
Colonoscopy
The role of colonoscopy in chronic constipation is low yield and only indicated if alarm signs are present.2 When no organic causes can be identified, the patient is deemed to have a functional bowel or motility disorder leading to constipation.
Colonic Transit Time
Colonic transit time (CTT) can be evaluated by assessing the presence of radio-opaque sitz markers in the colon with an abdominal x-ray 5 days after ingestion. The presence of five or more sitz markers may indicate STC. However, this can also signal an obstructive defecatory disorder. Colon scintigraphy can determine whether there is diffuse colonic dysmotility or dysfunction in a specific segment of the colon.10
Anorectal Function Testing (AFT)
AFT can evaluate DDs, such as fecal incontinence, dyssynergic defecation, rectal sensory disorders, anorectal pain, and rectal prolapse. AFT comprises three tests: anorectal manometry (ARM), balloon expulsion test (BET), and rectal sensory testing. These assess the defecation, continence, and sensory mechanisms of the rectum, respectively.
ARM testing employs a thin, flexible probe with an attached sensor that is inserted into the rectum to measure internal and external sphincter pressures while at rest, squeezing, and bearing down to give a functional assessment of sphincter tone.11 Cough or party balloon test assesses continence and sphincter strength. Rectal sensation is assessed by inflating a balloon incrementally and asking the patient to indicate first sensation, urgency to defecate, and discomfort. If both ARM and BET are abnormal, the patient meets diagnostic criteria for dyssynergic defecation.12
Pelvic floor disorders can be further assessed by MR defecography or barium defecography. Barium defecography is the more widely available of the two. MR defecography is a dynamic study that directly assesses pelvic floor muscles and endopelvic fascia during various stages of defecation and considered superior. This testing modality can distinguish between functional causes such as dyssynergia or pelvic floor dysfunction and structural causes of obstruction such as rectocele, rectal prolapse, or rectal intussusception. MR defecography is helpful when dyssynergia is suggested by ARM with a normal BET or if there is an absent recto-anal inhibitory reflex on ARM, which may suggest rectal intussusception.
Management
CIC
Incorporating 20-30 g of total soluble fiber, such as psyllium in individuals with low dietary fiber intake is the first-line recommendation for CIC.13 If response to a trial of fiber supplementation is inadequate, over-the-counter (OTC) osmotic laxatives such as polyethylene glycol and magnesium oxide can be incorporated. In the event of failure of OTC osmotic laxatives, lactulose can be considered. Stimulant laxatives such as senna, bisacodyl, or sodium picosulfate can be added as an adjunctive measure for short periods of time, defined as daily for 4 weeks or less.
If these measures are inadequate, pharmacological therapy with secretagogues and 5HT agonists can be considered. Prucalopride, a selective agonist of serotonin 5-HT4 receptors, is approved for CIC, prescribed 2 mg daily.14 It can also be used in patients with global motility delays, such as gastroparetics with constipation. The mechanism of action of secretagogues and specific dosing of these medications are discussed in Figure 2.15 Vibrant is a non-pharmacologic, orally ingested, vibrating, and programmable capsule device that has recently received Food and Drug Administration approval for treatment of chronic constipation by stimulating the intestinal wall, thereby promoting colonic contractile activity to achieve more spontaneous bowel movements. Further studies are required to assess its efficacy.16 Additionally, if there is inadequate response to all the above, it would be prudent to evaluate for the presence of pelvic floor dysfunction as well.
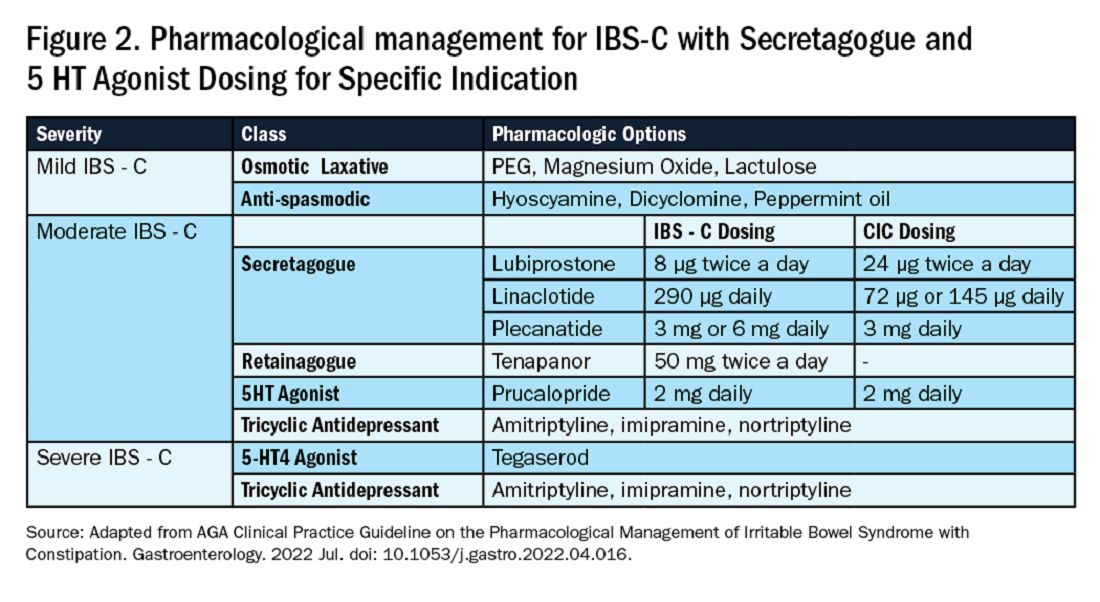
IBS-C
Similar to CIC, treatment for mild IBS-C starts with osmotic laxatives with the additional component of pain control. Antispasmodics can be used to manage the abdominal pain, cramping, and spasms associated with IBS-C. Antispasmodics available in the United States include anticholinergic agents that cause smooth muscle relaxation, such as dicyclomine or hyoscyamine or direct smooth muscle relaxants such as peppermint oil.17 IBS-C patients with moderate symptoms may need escalation of therapy to secretagogues or 5HT agonists (see Figure 2). Secretagogues increase fluid retention in the colonic lumen to promote bowel movements and improve visceral hypersensitivity. Lubiprostone is an intestinal chloride channel activator, indicated only for adult women with IBS-C. Linaclotide and plecanatide are guanylate cyclase-C activators which increase intestinal chloride and bicarbonate secretion, and both are indicated in IBS-C and CIC. Tenapanor inhibits the sodium/hydrogen exchanger in the GI tract, leading to increased water secretion, and is recommended for IBS-C in adults who have failed secretagogues.
All four of these drugs can be considered for moderate to severe IBS-C symptoms. In the case of severe IBS-C symptoms, Tegaserod, a 5-HT4 receptor partial agonist has been approved in women under 65 without significant cardiovascular or cerebrovascular disease.18 Regardless of IBS-C symptom severity, persistent visceral hypersensitivity can be treated with low-dose neuromodulators.19 Figure 2 provides treatment recommendations for IBS-C based on symptom severity.
Opioid-Induced Constipation (OIC)
In patients with OIC, peripherally acting mu-opioid receptor antagonists such as methylnaltrexone and naloxegol can be beneficial where stimulant laxatives are insufficient. Additionally, lubiprostone is indicated in OIC in non-cancer patients. At present, there are no head-to-head trials comparing efficacy of these medications.
Defecatory Disorders
Biofeedback therapy is the cornerstone of treatment for dyssynergic defecation, focusing on neuromuscular training to restore a normal pattern of defecation by teaching patients to tense the abdomen and relax the pelvic floor muscles and anal sphincter. It retrains the body to coordinate abdominal, rectal, and anal muscles to achieve synchronous contraction to achieve complete evacuation. It also increases awareness and response to rectal fullness or the need to defecate.
Biofeedback makes patients aware of counterproductive subconscious actions such as contracting of their anal sphincter during defecation followed by simulated defecation training with focus on how to tighten abdominal muscles and relax pelvic floor muscles to initiate and complete defecation.20 This is performed in the office with a physiotherapist or trained nurse for at least six sessions or at home where patients are encouraged to perform the exercises for 20 minutes, twice a day. These sessions utilize tools such as manometry probes, electromyography probes, simulated balloon, or home biofeedback training devices to provide visual feedback while practicing abdominophrenic breathing. Biofeedback is particularly helpful in patients suffering from constipation. Patients with defecatory disorders can also benefit from pelvic floor physical therapy which focuses on strengthening the pelvic and puborectal muscles, external anal sphincter, and pelvic muscles. This is more useful in patients with fecal incontinence. Despite all these treatments, a subset of patients may still not respond and may qualify for surgical evaluation.
Conclusion
While constipation is seldom life-threatening, it has a negative impact on patient quality of life and poses a significant financial burden on our overall healthcare system. The complexity of this condition should be appreciated and understood in order for a complete and thorough evaluation. We trust that our practical guide should serve as a useful tool in the evaluation of a chronically constipated patient.
Dr. Salim (@hamsalim07 on X) is based in the Department of Internal Medicine, University of Texas Medical Branch, Galveston. Dr. Chowdhury (annicho.med on Instagram) is a fellow in the Department of Gastroenterology, Hepatology, and Nutrition, University of Texas MD Anderson Cancer Center, Houston. Dr. Viswanathan (@LavanyaMD on X) is Associate Professor, University of Texas MD Anderson Cancer Center. The authors declare no conflict of interest.
References
1. Mugie S et al. Best Pract Res Clin Gastroenterol. 2011 Feb. doi: 10.1016/j.bpg.2010.12.010.
2. Almario CV et al. Gastroenterology. 2023 Dec. doi: 10.1053/j.gastro.2023.08.010.
3. Canavan C et al. Clin Epidemiol. 2014 Feb. doi: 10.2147/CLEP.S40245.
4. Rao SSC. Gastroenterol Clin North Am. 2007 Sep. doi: 10.1016/j.gtc.2007.07.013.
5. Bharucha AE et al. Gastroenterology. 2020 Apr. doi: 10.1053/j.gastro.2019.12.034.
6. Cinquetti M et al. Clin Exp Pediatr. 2021 Sep. doi: 10.3345/cep.2020.01326.
7. Shah ND et al. Am J Gastroenterol. 2008 Jul. doi: 10.1111/j.1572-0241.2008.01910.x.
8. Rajindrajith S et al. J Pediatr Gastroenterol Nutr. 2014 Apr. doi: 10.1097/MPG.0000000000000249.
9. Talley NJ. Am J Gastroenterol. 2008 Apr. doi: 10.1111/j.1572-0241.2008.01832.x.
10. Maurer AH. J Nucl Med. 2015 Sep. doi: 10.2967/jnumed.113.134551.
11. Frye J et al. Am J Gastroenterol. 2024 Aug. doi: 10.14309/ajg.0000000000002670.
12. Rao SSC et al. J Neurogastroenterol Motil. 2016 Jun. doi: 10.5056/jnm16060.
13. Chang L et al. Gastroenterology. 2023 Jun. doi: 10.1053/j.gastro.2023.03.214.
14. Brenner DM et al. Am J Gastroenterol. 2021 Aug. doi: 10.14309/ajg.0000000000001266.
15. Chang L et al. Gastroenterology. 2022 Jul. doi: 10.1053/j.gastro.2022.04.016.
16. Rao SSC et al. Gastroenterology. 2023 Jun. doi: 10.1053/j.gastro.2023.02.013.
17. Lacy BE et al. Am J Gastroenterol. 2021 Jan. doi: 10.14309/ajg.0000000000001036.
18. Anderson JL et al. J Cardiovasc Pharmacol Ther. 2009 Sep. doi: 10.1177/1074248409340158.
19. Rahimi R et al. World J Gastroenterol. 2009 Apr. doi: 10.3748/wjg.15.1548.
20. Rao SSC. Best Pract Res Clin Gastroenterol. 2011 Feb. doi: 10.1016/j.bpg.2011.01.004.
Introduction
Constipation affects one in six people worldwide and accounts for one third of outpatient visits.1 Chronic constipation is defined by difficult, infrequent, and/or incomplete defecation, quantified by less than three spontaneous bowel movements per week, persisting for at least 3 months. Patients may complain of straining during defecation, incomplete evacuation, hard stools (Bristol stool scale [BSS] type 1-2), and fullness or bloating. Chronic constipation can be subclassified as either a primary or secondary disorder.1,2
Primary Constipation Disorders
Primary constipation includes disorders of the colon or anorectum. This includes irritable bowel syndrome with constipation (IBS-C), chronic idiopathic constipation (CIC), slow transit constipation (STC), dyssynergic defecation, and pelvic floor disorders (see Figure 1).

IBS-C
IBS-C is a chronic disorder of the gut-brain axis with a worldwide prevalence of 1.3% and a prevalence of 6%-16% in the United States, United Kingdom, and Canada, with females more likely to seek care than males.2 The economic impact of IBS-C is estimated to be $1.5 billion–$10 billion per year in the United States alone.3 The distinguishing characteristic is abdominal pain, however IBS-C can present with a constellation of symptoms. The diagnostic paradigm has shifted from IBS being a diagnosis of exclusion to now using a positive diagnostic strategy.2 Using this Rome IV criteria, one can make the diagnosis with > 95% accuracy.2,4
CIC
CIC, previously defined as functional constipation, is a disorder defined by incomplete defecation and difficult or infrequent stool. CIC is diagnosed in patients without an underlying anatomic or structural abnormality. Rome IV Criteria helps further classify the defining characteristics of chronic idiopathic constipation.2
Slow Transit Constipation
STC is characterized by impaired colonic transit time in the absence of pelvic floor dysfunction. It presents with infrequent bowel movements, diminished urgency, and/or straining with defecation.
Defecatory Disorders: Dyssynergic Defecation and Pelvic Floor Dysfunction
Defecatory disorders (DDs) result from alterations in the colonic-neural pathway with an unclear pathogenesis. A firm understanding of colonic physiology is necessary to identify DDs. The right colon helps to store and mix stool contents, the left colon helps add water to the stool, and the anal canal and rectum enable defecation and maintain continence. Any alteration along this physiologic pathway results in DDs.5
DDs primarily develop via maladaptive pelvic floor contraction during defecation or from muscle or nerve injury and include functional outlet obstruction, anorectal dyssynergia, and pelvic floor dysfunction. Increased resistance to defecation results from anismus, paradoxical anal sphincter contraction, or incomplete relaxation of the pelvic floor and external anal sphincter. This muscle incoordination is described as dyssynergia. DDs can involve either muscle or nerve dysfunction or a combination of the two. Reduced rectal sensation caused by reduced sensory triggers can cause stasis of stool, thus propagating the cycle of constipation. Over time, excessive straining can weaken the pelvic floor, increasing the risk of excessive perineal descent, rectal intussusception, solitary rectal ulcer syndrome, and pudendal neuropathy.5 Thus, identification of DDs is crucial in patients with chronic constipation.
Secondary Constipation Disorders
Secondary constipation disorders are a result of an alternate process and warrant a thorough review of outpatient medications and past medical history. Figure 1 outlines the most common causes of secondary constipation, which span a wide differential.
Clinical Evaluation
The evaluation of constipation begins with a thorough history. Description of bowel habits should include frequency, duration, straining, stool consistency using a Bristol stool chart, complete vs incomplete evacuation, pain, bloating, and use of digital maneuvers (vaginal splinting or digital stool removal). One should inquire about back trauma/surgeries and obstetric history to include vaginal forceps injury or episiotomy.
With increased smartphone use, toilet time on average has increased and can contribute to maladaptive bowel habits.6 Patients may not realize they are constipated, so patient education is critical. A patient with daily bowel movements ranging between BSS type 1-6 with incomplete evacuation might complain of diarrhea but may in fact have constipation with overflow diarrhea, for example. Past medical history is also clinically relevant, as systemic conditions can cause secondary constipation. A constipated patient should also be asked what therapies he/she has tried prior to gastroenterology referral as primary care referrals for constipation account for 8 million visits to gastroenterology per year.7
While a sensitive topic, inquire about abuse history, especially in those with childhood constipation symptoms. There is a positive correlation between childhood constipation and physical, emotional, and sexual abuse and, for any number of reasons, your patient may be reluctant to share this or undergo a digital rectal exam (DRE).8 In such cases, be sensitive in asking for this history in private rather than with other family members around and always perform this exam with a chaperone present.
A detailed physical exam is an indispensable tool all gastroenterologists must master when evaluating a constipated patient. Some key exam findings include abdominal distention, high-pitched bowel sounds, and presence of a succussion splash indicating obstructive pathology. Dry skin and brittle hair indicate hypothyroidism while hypermobile joints and skin laxity suggest connective tissue disease. Finally, a physical examination is incomplete without a DRE.
DRE
DRE is an often-overlooked physical exam component which provides helpful insight that can guide management. An informed DRE can help identify structural disorders such as fissure, hemorrhoids, anorectal mass, fecal impaction, rectal prolapse, and excessive perineal descent syndrome.9 Unless contraindicated, DRE should be a standard part of the workup of a patient with chronic constipation.
Workup
Colonoscopy
The role of colonoscopy in chronic constipation is low yield and only indicated if alarm signs are present.2 When no organic causes can be identified, the patient is deemed to have a functional bowel or motility disorder leading to constipation.
Colonic Transit Time
Colonic transit time (CTT) can be evaluated by assessing the presence of radio-opaque sitz markers in the colon with an abdominal x-ray 5 days after ingestion. The presence of five or more sitz markers may indicate STC. However, this can also signal an obstructive defecatory disorder. Colon scintigraphy can determine whether there is diffuse colonic dysmotility or dysfunction in a specific segment of the colon.10
Anorectal Function Testing (AFT)
AFT can evaluate DDs, such as fecal incontinence, dyssynergic defecation, rectal sensory disorders, anorectal pain, and rectal prolapse. AFT comprises three tests: anorectal manometry (ARM), balloon expulsion test (BET), and rectal sensory testing. These assess the defecation, continence, and sensory mechanisms of the rectum, respectively.
ARM testing employs a thin, flexible probe with an attached sensor that is inserted into the rectum to measure internal and external sphincter pressures while at rest, squeezing, and bearing down to give a functional assessment of sphincter tone.11 Cough or party balloon test assesses continence and sphincter strength. Rectal sensation is assessed by inflating a balloon incrementally and asking the patient to indicate first sensation, urgency to defecate, and discomfort. If both ARM and BET are abnormal, the patient meets diagnostic criteria for dyssynergic defecation.12
Pelvic floor disorders can be further assessed by MR defecography or barium defecography. Barium defecography is the more widely available of the two. MR defecography is a dynamic study that directly assesses pelvic floor muscles and endopelvic fascia during various stages of defecation and considered superior. This testing modality can distinguish between functional causes such as dyssynergia or pelvic floor dysfunction and structural causes of obstruction such as rectocele, rectal prolapse, or rectal intussusception. MR defecography is helpful when dyssynergia is suggested by ARM with a normal BET or if there is an absent recto-anal inhibitory reflex on ARM, which may suggest rectal intussusception.
Management
CIC
Incorporating 20-30 g of total soluble fiber, such as psyllium in individuals with low dietary fiber intake is the first-line recommendation for CIC.13 If response to a trial of fiber supplementation is inadequate, over-the-counter (OTC) osmotic laxatives such as polyethylene glycol and magnesium oxide can be incorporated. In the event of failure of OTC osmotic laxatives, lactulose can be considered. Stimulant laxatives such as senna, bisacodyl, or sodium picosulfate can be added as an adjunctive measure for short periods of time, defined as daily for 4 weeks or less.
If these measures are inadequate, pharmacological therapy with secretagogues and 5HT agonists can be considered. Prucalopride, a selective agonist of serotonin 5-HT4 receptors, is approved for CIC, prescribed 2 mg daily.14 It can also be used in patients with global motility delays, such as gastroparetics with constipation. The mechanism of action of secretagogues and specific dosing of these medications are discussed in Figure 2.15 Vibrant is a non-pharmacologic, orally ingested, vibrating, and programmable capsule device that has recently received Food and Drug Administration approval for treatment of chronic constipation by stimulating the intestinal wall, thereby promoting colonic contractile activity to achieve more spontaneous bowel movements. Further studies are required to assess its efficacy.16 Additionally, if there is inadequate response to all the above, it would be prudent to evaluate for the presence of pelvic floor dysfunction as well.

IBS-C
Similar to CIC, treatment for mild IBS-C starts with osmotic laxatives with the additional component of pain control. Antispasmodics can be used to manage the abdominal pain, cramping, and spasms associated with IBS-C. Antispasmodics available in the United States include anticholinergic agents that cause smooth muscle relaxation, such as dicyclomine or hyoscyamine or direct smooth muscle relaxants such as peppermint oil.17 IBS-C patients with moderate symptoms may need escalation of therapy to secretagogues or 5HT agonists (see Figure 2). Secretagogues increase fluid retention in the colonic lumen to promote bowel movements and improve visceral hypersensitivity. Lubiprostone is an intestinal chloride channel activator, indicated only for adult women with IBS-C. Linaclotide and plecanatide are guanylate cyclase-C activators which increase intestinal chloride and bicarbonate secretion, and both are indicated in IBS-C and CIC. Tenapanor inhibits the sodium/hydrogen exchanger in the GI tract, leading to increased water secretion, and is recommended for IBS-C in adults who have failed secretagogues.
All four of these drugs can be considered for moderate to severe IBS-C symptoms. In the case of severe IBS-C symptoms, Tegaserod, a 5-HT4 receptor partial agonist has been approved in women under 65 without significant cardiovascular or cerebrovascular disease.18 Regardless of IBS-C symptom severity, persistent visceral hypersensitivity can be treated with low-dose neuromodulators.19 Figure 2 provides treatment recommendations for IBS-C based on symptom severity.
Opioid-Induced Constipation (OIC)
In patients with OIC, peripherally acting mu-opioid receptor antagonists such as methylnaltrexone and naloxegol can be beneficial where stimulant laxatives are insufficient. Additionally, lubiprostone is indicated in OIC in non-cancer patients. At present, there are no head-to-head trials comparing efficacy of these medications.
Defecatory Disorders
Biofeedback therapy is the cornerstone of treatment for dyssynergic defecation, focusing on neuromuscular training to restore a normal pattern of defecation by teaching patients to tense the abdomen and relax the pelvic floor muscles and anal sphincter. It retrains the body to coordinate abdominal, rectal, and anal muscles to achieve synchronous contraction to achieve complete evacuation. It also increases awareness and response to rectal fullness or the need to defecate.
Biofeedback makes patients aware of counterproductive subconscious actions such as contracting of their anal sphincter during defecation followed by simulated defecation training with focus on how to tighten abdominal muscles and relax pelvic floor muscles to initiate and complete defecation.20 This is performed in the office with a physiotherapist or trained nurse for at least six sessions or at home where patients are encouraged to perform the exercises for 20 minutes, twice a day. These sessions utilize tools such as manometry probes, electromyography probes, simulated balloon, or home biofeedback training devices to provide visual feedback while practicing abdominophrenic breathing. Biofeedback is particularly helpful in patients suffering from constipation. Patients with defecatory disorders can also benefit from pelvic floor physical therapy which focuses on strengthening the pelvic and puborectal muscles, external anal sphincter, and pelvic muscles. This is more useful in patients with fecal incontinence. Despite all these treatments, a subset of patients may still not respond and may qualify for surgical evaluation.
Conclusion
While constipation is seldom life-threatening, it has a negative impact on patient quality of life and poses a significant financial burden on our overall healthcare system. The complexity of this condition should be appreciated and understood in order for a complete and thorough evaluation. We trust that our practical guide should serve as a useful tool in the evaluation of a chronically constipated patient.
Dr. Salim (@hamsalim07 on X) is based in the Department of Internal Medicine, University of Texas Medical Branch, Galveston. Dr. Chowdhury (annicho.med on Instagram) is a fellow in the Department of Gastroenterology, Hepatology, and Nutrition, University of Texas MD Anderson Cancer Center, Houston. Dr. Viswanathan (@LavanyaMD on X) is Associate Professor, University of Texas MD Anderson Cancer Center. The authors declare no conflict of interest.
References
1. Mugie S et al. Best Pract Res Clin Gastroenterol. 2011 Feb. doi: 10.1016/j.bpg.2010.12.010.
2. Almario CV et al. Gastroenterology. 2023 Dec. doi: 10.1053/j.gastro.2023.08.010.
3. Canavan C et al. Clin Epidemiol. 2014 Feb. doi: 10.2147/CLEP.S40245.
4. Rao SSC. Gastroenterol Clin North Am. 2007 Sep. doi: 10.1016/j.gtc.2007.07.013.
5. Bharucha AE et al. Gastroenterology. 2020 Apr. doi: 10.1053/j.gastro.2019.12.034.
6. Cinquetti M et al. Clin Exp Pediatr. 2021 Sep. doi: 10.3345/cep.2020.01326.
7. Shah ND et al. Am J Gastroenterol. 2008 Jul. doi: 10.1111/j.1572-0241.2008.01910.x.
8. Rajindrajith S et al. J Pediatr Gastroenterol Nutr. 2014 Apr. doi: 10.1097/MPG.0000000000000249.
9. Talley NJ. Am J Gastroenterol. 2008 Apr. doi: 10.1111/j.1572-0241.2008.01832.x.
10. Maurer AH. J Nucl Med. 2015 Sep. doi: 10.2967/jnumed.113.134551.
11. Frye J et al. Am J Gastroenterol. 2024 Aug. doi: 10.14309/ajg.0000000000002670.
12. Rao SSC et al. J Neurogastroenterol Motil. 2016 Jun. doi: 10.5056/jnm16060.
13. Chang L et al. Gastroenterology. 2023 Jun. doi: 10.1053/j.gastro.2023.03.214.
14. Brenner DM et al. Am J Gastroenterol. 2021 Aug. doi: 10.14309/ajg.0000000000001266.
15. Chang L et al. Gastroenterology. 2022 Jul. doi: 10.1053/j.gastro.2022.04.016.
16. Rao SSC et al. Gastroenterology. 2023 Jun. doi: 10.1053/j.gastro.2023.02.013.
17. Lacy BE et al. Am J Gastroenterol. 2021 Jan. doi: 10.14309/ajg.0000000000001036.
18. Anderson JL et al. J Cardiovasc Pharmacol Ther. 2009 Sep. doi: 10.1177/1074248409340158.
19. Rahimi R et al. World J Gastroenterol. 2009 Apr. doi: 10.3748/wjg.15.1548.
20. Rao SSC. Best Pract Res Clin Gastroenterol. 2011 Feb. doi: 10.1016/j.bpg.2011.01.004.
Introduction
Constipation affects one in six people worldwide and accounts for one third of outpatient visits.1 Chronic constipation is defined by difficult, infrequent, and/or incomplete defecation, quantified by less than three spontaneous bowel movements per week, persisting for at least 3 months. Patients may complain of straining during defecation, incomplete evacuation, hard stools (Bristol stool scale [BSS] type 1-2), and fullness or bloating. Chronic constipation can be subclassified as either a primary or secondary disorder.1,2
Primary Constipation Disorders
Primary constipation includes disorders of the colon or anorectum. This includes irritable bowel syndrome with constipation (IBS-C), chronic idiopathic constipation (CIC), slow transit constipation (STC), dyssynergic defecation, and pelvic floor disorders (see Figure 1).

IBS-C
IBS-C is a chronic disorder of the gut-brain axis with a worldwide prevalence of 1.3% and a prevalence of 6%-16% in the United States, United Kingdom, and Canada, with females more likely to seek care than males.2 The economic impact of IBS-C is estimated to be $1.5 billion–$10 billion per year in the United States alone.3 The distinguishing characteristic is abdominal pain, however IBS-C can present with a constellation of symptoms. The diagnostic paradigm has shifted from IBS being a diagnosis of exclusion to now using a positive diagnostic strategy.2 Using this Rome IV criteria, one can make the diagnosis with > 95% accuracy.2,4
CIC
CIC, previously defined as functional constipation, is a disorder defined by incomplete defecation and difficult or infrequent stool. CIC is diagnosed in patients without an underlying anatomic or structural abnormality. Rome IV Criteria helps further classify the defining characteristics of chronic idiopathic constipation.2
Slow Transit Constipation
STC is characterized by impaired colonic transit time in the absence of pelvic floor dysfunction. It presents with infrequent bowel movements, diminished urgency, and/or straining with defecation.
Defecatory Disorders: Dyssynergic Defecation and Pelvic Floor Dysfunction
Defecatory disorders (DDs) result from alterations in the colonic-neural pathway with an unclear pathogenesis. A firm understanding of colonic physiology is necessary to identify DDs. The right colon helps to store and mix stool contents, the left colon helps add water to the stool, and the anal canal and rectum enable defecation and maintain continence. Any alteration along this physiologic pathway results in DDs.5
DDs primarily develop via maladaptive pelvic floor contraction during defecation or from muscle or nerve injury and include functional outlet obstruction, anorectal dyssynergia, and pelvic floor dysfunction. Increased resistance to defecation results from anismus, paradoxical anal sphincter contraction, or incomplete relaxation of the pelvic floor and external anal sphincter. This muscle incoordination is described as dyssynergia. DDs can involve either muscle or nerve dysfunction or a combination of the two. Reduced rectal sensation caused by reduced sensory triggers can cause stasis of stool, thus propagating the cycle of constipation. Over time, excessive straining can weaken the pelvic floor, increasing the risk of excessive perineal descent, rectal intussusception, solitary rectal ulcer syndrome, and pudendal neuropathy.5 Thus, identification of DDs is crucial in patients with chronic constipation.
Secondary Constipation Disorders
Secondary constipation disorders are a result of an alternate process and warrant a thorough review of outpatient medications and past medical history. Figure 1 outlines the most common causes of secondary constipation, which span a wide differential.
Clinical Evaluation
The evaluation of constipation begins with a thorough history. Description of bowel habits should include frequency, duration, straining, stool consistency using a Bristol stool chart, complete vs incomplete evacuation, pain, bloating, and use of digital maneuvers (vaginal splinting or digital stool removal). One should inquire about back trauma/surgeries and obstetric history to include vaginal forceps injury or episiotomy.
With increased smartphone use, toilet time on average has increased and can contribute to maladaptive bowel habits.6 Patients may not realize they are constipated, so patient education is critical. A patient with daily bowel movements ranging between BSS type 1-6 with incomplete evacuation might complain of diarrhea but may in fact have constipation with overflow diarrhea, for example. Past medical history is also clinically relevant, as systemic conditions can cause secondary constipation. A constipated patient should also be asked what therapies he/she has tried prior to gastroenterology referral as primary care referrals for constipation account for 8 million visits to gastroenterology per year.7
While a sensitive topic, inquire about abuse history, especially in those with childhood constipation symptoms. There is a positive correlation between childhood constipation and physical, emotional, and sexual abuse and, for any number of reasons, your patient may be reluctant to share this or undergo a digital rectal exam (DRE).8 In such cases, be sensitive in asking for this history in private rather than with other family members around and always perform this exam with a chaperone present.
A detailed physical exam is an indispensable tool all gastroenterologists must master when evaluating a constipated patient. Some key exam findings include abdominal distention, high-pitched bowel sounds, and presence of a succussion splash indicating obstructive pathology. Dry skin and brittle hair indicate hypothyroidism while hypermobile joints and skin laxity suggest connective tissue disease. Finally, a physical examination is incomplete without a DRE.
DRE
DRE is an often-overlooked physical exam component which provides helpful insight that can guide management. An informed DRE can help identify structural disorders such as fissure, hemorrhoids, anorectal mass, fecal impaction, rectal prolapse, and excessive perineal descent syndrome.9 Unless contraindicated, DRE should be a standard part of the workup of a patient with chronic constipation.
Workup
Colonoscopy
The role of colonoscopy in chronic constipation is low yield and only indicated if alarm signs are present.2 When no organic causes can be identified, the patient is deemed to have a functional bowel or motility disorder leading to constipation.
Colonic Transit Time
Colonic transit time (CTT) can be evaluated by assessing the presence of radio-opaque sitz markers in the colon with an abdominal x-ray 5 days after ingestion. The presence of five or more sitz markers may indicate STC. However, this can also signal an obstructive defecatory disorder. Colon scintigraphy can determine whether there is diffuse colonic dysmotility or dysfunction in a specific segment of the colon.10
Anorectal Function Testing (AFT)
AFT can evaluate DDs, such as fecal incontinence, dyssynergic defecation, rectal sensory disorders, anorectal pain, and rectal prolapse. AFT comprises three tests: anorectal manometry (ARM), balloon expulsion test (BET), and rectal sensory testing. These assess the defecation, continence, and sensory mechanisms of the rectum, respectively.
ARM testing employs a thin, flexible probe with an attached sensor that is inserted into the rectum to measure internal and external sphincter pressures while at rest, squeezing, and bearing down to give a functional assessment of sphincter tone.11 Cough or party balloon test assesses continence and sphincter strength. Rectal sensation is assessed by inflating a balloon incrementally and asking the patient to indicate first sensation, urgency to defecate, and discomfort. If both ARM and BET are abnormal, the patient meets diagnostic criteria for dyssynergic defecation.12
Pelvic floor disorders can be further assessed by MR defecography or barium defecography. Barium defecography is the more widely available of the two. MR defecography is a dynamic study that directly assesses pelvic floor muscles and endopelvic fascia during various stages of defecation and considered superior. This testing modality can distinguish between functional causes such as dyssynergia or pelvic floor dysfunction and structural causes of obstruction such as rectocele, rectal prolapse, or rectal intussusception. MR defecography is helpful when dyssynergia is suggested by ARM with a normal BET or if there is an absent recto-anal inhibitory reflex on ARM, which may suggest rectal intussusception.
Management
CIC
Incorporating 20-30 g of total soluble fiber, such as psyllium in individuals with low dietary fiber intake is the first-line recommendation for CIC.13 If response to a trial of fiber supplementation is inadequate, over-the-counter (OTC) osmotic laxatives such as polyethylene glycol and magnesium oxide can be incorporated. In the event of failure of OTC osmotic laxatives, lactulose can be considered. Stimulant laxatives such as senna, bisacodyl, or sodium picosulfate can be added as an adjunctive measure for short periods of time, defined as daily for 4 weeks or less.
If these measures are inadequate, pharmacological therapy with secretagogues and 5HT agonists can be considered. Prucalopride, a selective agonist of serotonin 5-HT4 receptors, is approved for CIC, prescribed 2 mg daily.14 It can also be used in patients with global motility delays, such as gastroparetics with constipation. The mechanism of action of secretagogues and specific dosing of these medications are discussed in Figure 2.15 Vibrant is a non-pharmacologic, orally ingested, vibrating, and programmable capsule device that has recently received Food and Drug Administration approval for treatment of chronic constipation by stimulating the intestinal wall, thereby promoting colonic contractile activity to achieve more spontaneous bowel movements. Further studies are required to assess its efficacy.16 Additionally, if there is inadequate response to all the above, it would be prudent to evaluate for the presence of pelvic floor dysfunction as well.

IBS-C
Similar to CIC, treatment for mild IBS-C starts with osmotic laxatives with the additional component of pain control. Antispasmodics can be used to manage the abdominal pain, cramping, and spasms associated with IBS-C. Antispasmodics available in the United States include anticholinergic agents that cause smooth muscle relaxation, such as dicyclomine or hyoscyamine or direct smooth muscle relaxants such as peppermint oil.17 IBS-C patients with moderate symptoms may need escalation of therapy to secretagogues or 5HT agonists (see Figure 2). Secretagogues increase fluid retention in the colonic lumen to promote bowel movements and improve visceral hypersensitivity. Lubiprostone is an intestinal chloride channel activator, indicated only for adult women with IBS-C. Linaclotide and plecanatide are guanylate cyclase-C activators which increase intestinal chloride and bicarbonate secretion, and both are indicated in IBS-C and CIC. Tenapanor inhibits the sodium/hydrogen exchanger in the GI tract, leading to increased water secretion, and is recommended for IBS-C in adults who have failed secretagogues.
All four of these drugs can be considered for moderate to severe IBS-C symptoms. In the case of severe IBS-C symptoms, Tegaserod, a 5-HT4 receptor partial agonist has been approved in women under 65 without significant cardiovascular or cerebrovascular disease.18 Regardless of IBS-C symptom severity, persistent visceral hypersensitivity can be treated with low-dose neuromodulators.19 Figure 2 provides treatment recommendations for IBS-C based on symptom severity.
Opioid-Induced Constipation (OIC)
In patients with OIC, peripherally acting mu-opioid receptor antagonists such as methylnaltrexone and naloxegol can be beneficial where stimulant laxatives are insufficient. Additionally, lubiprostone is indicated in OIC in non-cancer patients. At present, there are no head-to-head trials comparing efficacy of these medications.
Defecatory Disorders
Biofeedback therapy is the cornerstone of treatment for dyssynergic defecation, focusing on neuromuscular training to restore a normal pattern of defecation by teaching patients to tense the abdomen and relax the pelvic floor muscles and anal sphincter. It retrains the body to coordinate abdominal, rectal, and anal muscles to achieve synchronous contraction to achieve complete evacuation. It also increases awareness and response to rectal fullness or the need to defecate.
Biofeedback makes patients aware of counterproductive subconscious actions such as contracting of their anal sphincter during defecation followed by simulated defecation training with focus on how to tighten abdominal muscles and relax pelvic floor muscles to initiate and complete defecation.20 This is performed in the office with a physiotherapist or trained nurse for at least six sessions or at home where patients are encouraged to perform the exercises for 20 minutes, twice a day. These sessions utilize tools such as manometry probes, electromyography probes, simulated balloon, or home biofeedback training devices to provide visual feedback while practicing abdominophrenic breathing. Biofeedback is particularly helpful in patients suffering from constipation. Patients with defecatory disorders can also benefit from pelvic floor physical therapy which focuses on strengthening the pelvic and puborectal muscles, external anal sphincter, and pelvic muscles. This is more useful in patients with fecal incontinence. Despite all these treatments, a subset of patients may still not respond and may qualify for surgical evaluation.
Conclusion
While constipation is seldom life-threatening, it has a negative impact on patient quality of life and poses a significant financial burden on our overall healthcare system. The complexity of this condition should be appreciated and understood in order for a complete and thorough evaluation. We trust that our practical guide should serve as a useful tool in the evaluation of a chronically constipated patient.
Dr. Salim (@hamsalim07 on X) is based in the Department of Internal Medicine, University of Texas Medical Branch, Galveston. Dr. Chowdhury (annicho.med on Instagram) is a fellow in the Department of Gastroenterology, Hepatology, and Nutrition, University of Texas MD Anderson Cancer Center, Houston. Dr. Viswanathan (@LavanyaMD on X) is Associate Professor, University of Texas MD Anderson Cancer Center. The authors declare no conflict of interest.
References
1. Mugie S et al. Best Pract Res Clin Gastroenterol. 2011 Feb. doi: 10.1016/j.bpg.2010.12.010.
2. Almario CV et al. Gastroenterology. 2023 Dec. doi: 10.1053/j.gastro.2023.08.010.
3. Canavan C et al. Clin Epidemiol. 2014 Feb. doi: 10.2147/CLEP.S40245.
4. Rao SSC. Gastroenterol Clin North Am. 2007 Sep. doi: 10.1016/j.gtc.2007.07.013.
5. Bharucha AE et al. Gastroenterology. 2020 Apr. doi: 10.1053/j.gastro.2019.12.034.
6. Cinquetti M et al. Clin Exp Pediatr. 2021 Sep. doi: 10.3345/cep.2020.01326.
7. Shah ND et al. Am J Gastroenterol. 2008 Jul. doi: 10.1111/j.1572-0241.2008.01910.x.
8. Rajindrajith S et al. J Pediatr Gastroenterol Nutr. 2014 Apr. doi: 10.1097/MPG.0000000000000249.
9. Talley NJ. Am J Gastroenterol. 2008 Apr. doi: 10.1111/j.1572-0241.2008.01832.x.
10. Maurer AH. J Nucl Med. 2015 Sep. doi: 10.2967/jnumed.113.134551.
11. Frye J et al. Am J Gastroenterol. 2024 Aug. doi: 10.14309/ajg.0000000000002670.
12. Rao SSC et al. J Neurogastroenterol Motil. 2016 Jun. doi: 10.5056/jnm16060.
13. Chang L et al. Gastroenterology. 2023 Jun. doi: 10.1053/j.gastro.2023.03.214.
14. Brenner DM et al. Am J Gastroenterol. 2021 Aug. doi: 10.14309/ajg.0000000000001266.
15. Chang L et al. Gastroenterology. 2022 Jul. doi: 10.1053/j.gastro.2022.04.016.
16. Rao SSC et al. Gastroenterology. 2023 Jun. doi: 10.1053/j.gastro.2023.02.013.
17. Lacy BE et al. Am J Gastroenterol. 2021 Jan. doi: 10.14309/ajg.0000000000001036.
18. Anderson JL et al. J Cardiovasc Pharmacol Ther. 2009 Sep. doi: 10.1177/1074248409340158.
19. Rahimi R et al. World J Gastroenterol. 2009 Apr. doi: 10.3748/wjg.15.1548.
20. Rao SSC. Best Pract Res Clin Gastroenterol. 2011 Feb. doi: 10.1016/j.bpg.2011.01.004.
Random Biopsy Improves IBD Dysplasia Detection, With Caveats
according to a recent review and meta-analysis.
Random biopsies collected in studies after 2011 provided limited additional yield, suggesting that high-definition equipment alone may be sufficient to achieve a high detection rate, lead author Li Gao, MD, of Air Force Medical University, Xi’an, China, and colleagues reported. In contrast, patients with primary sclerosing cholangitis (PSC) consistently benefited from random biopsy, offering clearer support for use in this subgroup.
“Random biopsy has been proposed as a strategy that may detect dysplastic lesions that cannot be identified endoscopically, thus minimizing the occurrence of missed colitis-associated dysplasia during colonoscopy,” the investigators wrote in Clinical Gastroenterology and Hepatology. However, the role of random biopsies in colonoscopic surveillance for patients with IBD remains a topic of ongoing debate.”
The SCENIC guidelines remain inconclusive on the role of random biopsy in IBD surveillance, the investigators noted, while other guidelines recommend random biopsy with high-definition white light endoscopy, but not chromoendoscopy.
The present meta-analysis aimed to characterize the impact of random biopsy on dysplasia detection. The investigators aggregated prospective and retrospective studies published in English through September 2023, all of which compared random biopsy with other surveillance techniques and reported the proportion of dysplasia detected exclusively through random biopsy.
“To the best of our knowledge, this systematic review and meta-analysis was the first comprehensive summary of the additional yield of random biopsy during colorectal cancer surveillance in patients with IBD,” Dr. Gao and colleagues noted.
The final dataset comprised 37 studies with 9,051 patients undergoing colorectal cancer surveillance for IBD. Patients had diverse baseline characteristics, including different proportions of ulcerative colitis and Crohn’s disease, as well as varying prevalence of PSC, a known risk factor for colorectal neoplasia.
The pooled additional yield of random biopsy was 10.34% in per-patient analysis and 16.20% in per-lesion analysis, meaning that approximately 1 in 10 patients and 1 in 6 lesions were detected exclusively through random biopsy. Despite these benefits, detection rates were relatively low: 1.31% per patient and 2.82% per lesion.
Subgroup analyses showed a decline in random biopsy additional yield over time. Studies conducted before 2011 reported an additional yield of 14.43% in per-patient analysis, compared to just 0.42% in studies conducted after 2011. This decline coincided with the widespread adoption of high-definition endoscopy.
PSC status strongly influenced detection rates throughout the study period. In patients without PSC (0%-10% PSC prevalence), the additional yield of random biopsy was 4.83% in per-patient analysis and 11.23% in per-lesion analysis. In studies where all patients had PSC, the additional yield increased dramatically to 56.05% and 45.22%, respectively.
“These findings highlight the incremental benefits of random biopsy and provide valuable insights into the management of endoscopic surveillance in patients with IBD,” the investigators wrote. “Considering the decreased additional yields in studies initiated after 2011, and the influence of PSC, endoscopy centers lacking full high-definition equipment should consider incorporating random biopsy in the standard colonoscopy surveillance for IBD patients, especially in those with PSC.”This study was supported by the National Key R&D Program of China, the Key Research and Development Program of Shaanxi Province, and the Nanchang High-Level Scientific and Technological Innovation Talents “Double Hundred Plan” project. The investigators disclosed no conflicts of interest.
Patients with inflammatory bowel diseases (IBD) with colonic involvement are at two- to threefold increased risk of colorectal cancer (CRC), compared with the general population. The development and progression of dysplasia in these patients with IBD does not follow the typical adenoma-carcinoma sequence; rather, patients with IBD at increased risk of colorectal cancer may have field cancerization changes. Historically, these mucosal changes have been difficult to visualize endoscopically, at least with standard definition endoscopes. As a result, systematic, four-quadrant, random biopsies — 8 in each segment of the colon, totaling to 32 biopsies — are recommended for dysplasia detection. The practice has been adopted and accepted widely. Over time, there have been significant advancements in the management of IBD, with improved colonoscopic resolution, adjunct surveillance techniques, focus on quality of colonoscopic exams and evolution of treatments and treatment targets, and these have resulted in a reduction in the risk of CRC in patients with IBD. The value of random biopsies for dysplasia surveillance in patients with colonic IBD has been questioned.
In this context, the systematic review and meta-analysis from Gao and colleagues provides critical insights into the yield of random biopsies for dysplasia surveillance in patients with IBD. Through a detailed analysis of 37 studies published between 2003 to 2023, with 9051 patients who underwent dysplasia surveillance with random biopsies, they ascertained the incremental yield of random biopsies. Overall, 1.3% of patients who underwent random biopsies were detected to have dysplasia. Of these, 1 in 10 patients were detected to have dysplasia only on random biopsies. On per-lesion analysis, one in six dysplastic lesions were only detected on random biopsies. Interestingly, this yield of random biopsies varied markedly depending on the era, as a surrogate for quality of colonoscopies. In studies that fully enrolled and published before 2011 (majority of patients recruited in the 1990s to early 2000s), the per-patient incremental yield of random biopsies was 14%; this dropped precipitously to 0.4% in studies published after 2011 (majority of patients recruited in late 2000s to 2010s). The incremental yield of random biopsies remained markedly high in studies with a high proportion of patients with primary sclerosing cholangitis (PSC), a condition consistently associated with a four- to sixfold higher risk of CRC in patients with IBD.
These findings lend support to the notion that improvements in endoscopy equipment with wide adoption of high-definition white-light colonoscopes and an emphasis on quality of endoscopic examination may be leading to better endoscopic detection of previously “invisible” dysplastic lesions, leading to a markedly lower incremental yield of random biopsies in the current era. This questions the utility of routinely collecting 32 random biopsies during a surveillance exam for a patient with IBD at increased risk of CRC (as long as a thorough high-quality exam is being performed), though there may be subpopulations such as patients with PSC where there may be benefit. Large ongoing trials comparing the yield of targeted biopsies vs random and targeted biopsies in patients with IBD undergoing dysplasia surveillance with high-definition colonoscopes will help to definitively address this question.
Siddharth Singh, MD, MS, is associate professor of medicine and director of the UCSD IBD Center in the division of gastroenterology, University of California, San Diego. He declares no conflicts of interest relative to this article.
Patients with inflammatory bowel diseases (IBD) with colonic involvement are at two- to threefold increased risk of colorectal cancer (CRC), compared with the general population. The development and progression of dysplasia in these patients with IBD does not follow the typical adenoma-carcinoma sequence; rather, patients with IBD at increased risk of colorectal cancer may have field cancerization changes. Historically, these mucosal changes have been difficult to visualize endoscopically, at least with standard definition endoscopes. As a result, systematic, four-quadrant, random biopsies — 8 in each segment of the colon, totaling to 32 biopsies — are recommended for dysplasia detection. The practice has been adopted and accepted widely. Over time, there have been significant advancements in the management of IBD, with improved colonoscopic resolution, adjunct surveillance techniques, focus on quality of colonoscopic exams and evolution of treatments and treatment targets, and these have resulted in a reduction in the risk of CRC in patients with IBD. The value of random biopsies for dysplasia surveillance in patients with colonic IBD has been questioned.
In this context, the systematic review and meta-analysis from Gao and colleagues provides critical insights into the yield of random biopsies for dysplasia surveillance in patients with IBD. Through a detailed analysis of 37 studies published between 2003 to 2023, with 9051 patients who underwent dysplasia surveillance with random biopsies, they ascertained the incremental yield of random biopsies. Overall, 1.3% of patients who underwent random biopsies were detected to have dysplasia. Of these, 1 in 10 patients were detected to have dysplasia only on random biopsies. On per-lesion analysis, one in six dysplastic lesions were only detected on random biopsies. Interestingly, this yield of random biopsies varied markedly depending on the era, as a surrogate for quality of colonoscopies. In studies that fully enrolled and published before 2011 (majority of patients recruited in the 1990s to early 2000s), the per-patient incremental yield of random biopsies was 14%; this dropped precipitously to 0.4% in studies published after 2011 (majority of patients recruited in late 2000s to 2010s). The incremental yield of random biopsies remained markedly high in studies with a high proportion of patients with primary sclerosing cholangitis (PSC), a condition consistently associated with a four- to sixfold higher risk of CRC in patients with IBD.
These findings lend support to the notion that improvements in endoscopy equipment with wide adoption of high-definition white-light colonoscopes and an emphasis on quality of endoscopic examination may be leading to better endoscopic detection of previously “invisible” dysplastic lesions, leading to a markedly lower incremental yield of random biopsies in the current era. This questions the utility of routinely collecting 32 random biopsies during a surveillance exam for a patient with IBD at increased risk of CRC (as long as a thorough high-quality exam is being performed), though there may be subpopulations such as patients with PSC where there may be benefit. Large ongoing trials comparing the yield of targeted biopsies vs random and targeted biopsies in patients with IBD undergoing dysplasia surveillance with high-definition colonoscopes will help to definitively address this question.
Siddharth Singh, MD, MS, is associate professor of medicine and director of the UCSD IBD Center in the division of gastroenterology, University of California, San Diego. He declares no conflicts of interest relative to this article.
Patients with inflammatory bowel diseases (IBD) with colonic involvement are at two- to threefold increased risk of colorectal cancer (CRC), compared with the general population. The development and progression of dysplasia in these patients with IBD does not follow the typical adenoma-carcinoma sequence; rather, patients with IBD at increased risk of colorectal cancer may have field cancerization changes. Historically, these mucosal changes have been difficult to visualize endoscopically, at least with standard definition endoscopes. As a result, systematic, four-quadrant, random biopsies — 8 in each segment of the colon, totaling to 32 biopsies — are recommended for dysplasia detection. The practice has been adopted and accepted widely. Over time, there have been significant advancements in the management of IBD, with improved colonoscopic resolution, adjunct surveillance techniques, focus on quality of colonoscopic exams and evolution of treatments and treatment targets, and these have resulted in a reduction in the risk of CRC in patients with IBD. The value of random biopsies for dysplasia surveillance in patients with colonic IBD has been questioned.
In this context, the systematic review and meta-analysis from Gao and colleagues provides critical insights into the yield of random biopsies for dysplasia surveillance in patients with IBD. Through a detailed analysis of 37 studies published between 2003 to 2023, with 9051 patients who underwent dysplasia surveillance with random biopsies, they ascertained the incremental yield of random biopsies. Overall, 1.3% of patients who underwent random biopsies were detected to have dysplasia. Of these, 1 in 10 patients were detected to have dysplasia only on random biopsies. On per-lesion analysis, one in six dysplastic lesions were only detected on random biopsies. Interestingly, this yield of random biopsies varied markedly depending on the era, as a surrogate for quality of colonoscopies. In studies that fully enrolled and published before 2011 (majority of patients recruited in the 1990s to early 2000s), the per-patient incremental yield of random biopsies was 14%; this dropped precipitously to 0.4% in studies published after 2011 (majority of patients recruited in late 2000s to 2010s). The incremental yield of random biopsies remained markedly high in studies with a high proportion of patients with primary sclerosing cholangitis (PSC), a condition consistently associated with a four- to sixfold higher risk of CRC in patients with IBD.
These findings lend support to the notion that improvements in endoscopy equipment with wide adoption of high-definition white-light colonoscopes and an emphasis on quality of endoscopic examination may be leading to better endoscopic detection of previously “invisible” dysplastic lesions, leading to a markedly lower incremental yield of random biopsies in the current era. This questions the utility of routinely collecting 32 random biopsies during a surveillance exam for a patient with IBD at increased risk of CRC (as long as a thorough high-quality exam is being performed), though there may be subpopulations such as patients with PSC where there may be benefit. Large ongoing trials comparing the yield of targeted biopsies vs random and targeted biopsies in patients with IBD undergoing dysplasia surveillance with high-definition colonoscopes will help to definitively address this question.
Siddharth Singh, MD, MS, is associate professor of medicine and director of the UCSD IBD Center in the division of gastroenterology, University of California, San Diego. He declares no conflicts of interest relative to this article.
according to a recent review and meta-analysis.
Random biopsies collected in studies after 2011 provided limited additional yield, suggesting that high-definition equipment alone may be sufficient to achieve a high detection rate, lead author Li Gao, MD, of Air Force Medical University, Xi’an, China, and colleagues reported. In contrast, patients with primary sclerosing cholangitis (PSC) consistently benefited from random biopsy, offering clearer support for use in this subgroup.
“Random biopsy has been proposed as a strategy that may detect dysplastic lesions that cannot be identified endoscopically, thus minimizing the occurrence of missed colitis-associated dysplasia during colonoscopy,” the investigators wrote in Clinical Gastroenterology and Hepatology. However, the role of random biopsies in colonoscopic surveillance for patients with IBD remains a topic of ongoing debate.”
The SCENIC guidelines remain inconclusive on the role of random biopsy in IBD surveillance, the investigators noted, while other guidelines recommend random biopsy with high-definition white light endoscopy, but not chromoendoscopy.
The present meta-analysis aimed to characterize the impact of random biopsy on dysplasia detection. The investigators aggregated prospective and retrospective studies published in English through September 2023, all of which compared random biopsy with other surveillance techniques and reported the proportion of dysplasia detected exclusively through random biopsy.
“To the best of our knowledge, this systematic review and meta-analysis was the first comprehensive summary of the additional yield of random biopsy during colorectal cancer surveillance in patients with IBD,” Dr. Gao and colleagues noted.
The final dataset comprised 37 studies with 9,051 patients undergoing colorectal cancer surveillance for IBD. Patients had diverse baseline characteristics, including different proportions of ulcerative colitis and Crohn’s disease, as well as varying prevalence of PSC, a known risk factor for colorectal neoplasia.
The pooled additional yield of random biopsy was 10.34% in per-patient analysis and 16.20% in per-lesion analysis, meaning that approximately 1 in 10 patients and 1 in 6 lesions were detected exclusively through random biopsy. Despite these benefits, detection rates were relatively low: 1.31% per patient and 2.82% per lesion.
Subgroup analyses showed a decline in random biopsy additional yield over time. Studies conducted before 2011 reported an additional yield of 14.43% in per-patient analysis, compared to just 0.42% in studies conducted after 2011. This decline coincided with the widespread adoption of high-definition endoscopy.
PSC status strongly influenced detection rates throughout the study period. In patients without PSC (0%-10% PSC prevalence), the additional yield of random biopsy was 4.83% in per-patient analysis and 11.23% in per-lesion analysis. In studies where all patients had PSC, the additional yield increased dramatically to 56.05% and 45.22%, respectively.
“These findings highlight the incremental benefits of random biopsy and provide valuable insights into the management of endoscopic surveillance in patients with IBD,” the investigators wrote. “Considering the decreased additional yields in studies initiated after 2011, and the influence of PSC, endoscopy centers lacking full high-definition equipment should consider incorporating random biopsy in the standard colonoscopy surveillance for IBD patients, especially in those with PSC.”This study was supported by the National Key R&D Program of China, the Key Research and Development Program of Shaanxi Province, and the Nanchang High-Level Scientific and Technological Innovation Talents “Double Hundred Plan” project. The investigators disclosed no conflicts of interest.
according to a recent review and meta-analysis.
Random biopsies collected in studies after 2011 provided limited additional yield, suggesting that high-definition equipment alone may be sufficient to achieve a high detection rate, lead author Li Gao, MD, of Air Force Medical University, Xi’an, China, and colleagues reported. In contrast, patients with primary sclerosing cholangitis (PSC) consistently benefited from random biopsy, offering clearer support for use in this subgroup.
“Random biopsy has been proposed as a strategy that may detect dysplastic lesions that cannot be identified endoscopically, thus minimizing the occurrence of missed colitis-associated dysplasia during colonoscopy,” the investigators wrote in Clinical Gastroenterology and Hepatology. However, the role of random biopsies in colonoscopic surveillance for patients with IBD remains a topic of ongoing debate.”
The SCENIC guidelines remain inconclusive on the role of random biopsy in IBD surveillance, the investigators noted, while other guidelines recommend random biopsy with high-definition white light endoscopy, but not chromoendoscopy.
The present meta-analysis aimed to characterize the impact of random biopsy on dysplasia detection. The investigators aggregated prospective and retrospective studies published in English through September 2023, all of which compared random biopsy with other surveillance techniques and reported the proportion of dysplasia detected exclusively through random biopsy.
“To the best of our knowledge, this systematic review and meta-analysis was the first comprehensive summary of the additional yield of random biopsy during colorectal cancer surveillance in patients with IBD,” Dr. Gao and colleagues noted.
The final dataset comprised 37 studies with 9,051 patients undergoing colorectal cancer surveillance for IBD. Patients had diverse baseline characteristics, including different proportions of ulcerative colitis and Crohn’s disease, as well as varying prevalence of PSC, a known risk factor for colorectal neoplasia.
The pooled additional yield of random biopsy was 10.34% in per-patient analysis and 16.20% in per-lesion analysis, meaning that approximately 1 in 10 patients and 1 in 6 lesions were detected exclusively through random biopsy. Despite these benefits, detection rates were relatively low: 1.31% per patient and 2.82% per lesion.
Subgroup analyses showed a decline in random biopsy additional yield over time. Studies conducted before 2011 reported an additional yield of 14.43% in per-patient analysis, compared to just 0.42% in studies conducted after 2011. This decline coincided with the widespread adoption of high-definition endoscopy.
PSC status strongly influenced detection rates throughout the study period. In patients without PSC (0%-10% PSC prevalence), the additional yield of random biopsy was 4.83% in per-patient analysis and 11.23% in per-lesion analysis. In studies where all patients had PSC, the additional yield increased dramatically to 56.05% and 45.22%, respectively.
“These findings highlight the incremental benefits of random biopsy and provide valuable insights into the management of endoscopic surveillance in patients with IBD,” the investigators wrote. “Considering the decreased additional yields in studies initiated after 2011, and the influence of PSC, endoscopy centers lacking full high-definition equipment should consider incorporating random biopsy in the standard colonoscopy surveillance for IBD patients, especially in those with PSC.”This study was supported by the National Key R&D Program of China, the Key Research and Development Program of Shaanxi Province, and the Nanchang High-Level Scientific and Technological Innovation Talents “Double Hundred Plan” project. The investigators disclosed no conflicts of interest.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Suboptimal Diets Tied to Global Doubling of GI Cancer Cases
according to a recent study.
Writing in Gastroenterology, researchers led by Li Liu, PhD, of the department of epidemiology and biostatistics, Ministry of Education Key Lab of Environment and Health, School of Public Health, Tongji Medical College, Huazhong University of Science and Technology in Wuhan, China, reported that excessive consumption of processed meats (the biggest culprit), insufficient fruit intake, and insufficient whole grain intake were the leading dietary risk factors. In addition, the number of diet-related cases doubled from 1990 to 2018.
“In regions with limited access to healthy foods, policy interventions like taxing unhealthy foods and subsidizing nutritious options may help shift dietary patterns and reduce cancer risk,” Liu said in an interview.
The study examined meta-analyses from 184 countries in seven regions for the period 1990-2018 looking at rates of six major GI cancers: colorectal, liver, esophageal, pancreatic, and gallbladder/biliary tract. Among these, the age-standardized incidence of liver, pancreatic, and colorectal increased significantly over the past 3 decades.
The research team used a comparative risk assessment model to estimate the impact of diet on GI cancer independent of energy intake and adiposity. Although the principal dietary risk factors varied across individual cancers, suboptimal intake of the three aforementioned components was responsible for 66.51% of all diet-attributable GI cancers in 2018. The global mean processed meat consumption was 17 g/d in 2018, falling to a low in South Asia of 3 g/d.
The investigators also found diet-linked cancer incidence positively correlated with the Sociodemographic Index (SDI), an integrated measure of national development, income, and fertility. Incidence varied across world regions, with the highest proportion of cases in Central and Eastern Europe, Central Asia, Latin America, the Caribbean, and in high-income countries. The findings support the development of targeted diet-related public health interventions in various regions and nations to reduce GI cancer incidence, the authors wrote.
Among the study’s specific findings:
- In 2018, 21.5% (95% uncertainty interval [UI], 19.1-24.5) of incident GI cancer cases globally were attributable to suboptimal diets, a relatively stable proportion since 1990 (22.4%; 95% UI, 19.7-25.6).
- Absolute diet-attributable cases doubled from 580,862 (95% UI, 510,658-664,076) in 1990 to 1,039,877 (95% UI, 923,482-1,187,244) in 2018.
- Excessive processed meat consumption (5.9%; 95% UI, 4.2%-7.9%), insufficient fruit intake (4.8%; 95% UI, 3.8%-5.9%), and insufficient whole grain intake (3.6%; 95% UI, 2.8%-5.1%) were the most significant dietary risk factors in 2018 — a shift from 1990 when the third major concern was insufficient non-starchy vegetable intake.
Given the well-established link between diet and GI cancers, the incidence findings came as no surprise. “However, the dramatic doubling of diet-attributable cases over the past few decades was truly unexpected,” Liu said. “This increase can likely be attributed to global population growth and aging. While aging is an irreversible process, we can still reduce the growing burden of diet-related GI cancers by focusing on modifiable behaviors, particularly through targeted dietary interventions.”
A Modifiable Risk Factor
Commenting on the analysis but not involved in it, Andrew T. Chan, MD, MPH, a professor of medicine at Harvard Medical School and a gastroenterologist at Massachusetts General Hospital, both in Boston, noted that his own group’s studies also support the association of diet with an increased risk for GI cancers, particularly colorectal cancers.
“Although much work needs to be done to clarify the precise mechanisms underlying this association, there are substantial data that diet may cause changes in the gut microbiome, which in turn promotes cancer,” Chan said in an interview. “Going forward, we are working to develop strategies in which diet is modified to mitigate the risk of cancer associated with suboptimal diets.”
In other study findings, Liu’s group observed that two regional groups, Central and Eastern Europe, Central Asia, Latin America, and the Caribbean, as well as high-income countries, bore the top three diet-attributable burdens worldwide in 2018, all driven mostly by an upward-trending excess of processed meat.
By regions, Central and Eastern Europe and Central Asia experienced the highest attributable burden across regions in 1990 (31.6%; UI, 27.0%-37.4%) and 2018 (31.6%; UI, 27.3%-36.5%).
As for the impact of the SDI, the authors explained that diet-attributable GI cancer burden was higher among adults with higher education and living in urban areas than among those with lower education and rural residency. “Some dietary habits tended to be worse in higher-SDI countries, specifically, higher consumption of processed meats,” they wrote.
Although the proportional attributable GI incidence remains relatively stable, they added, the doubling of absolute cases from 1990 to 2018, along with the discrepancies between urbanicity and countries/regions, supports more targeted preventive measures.
And while the diet-GI cancer connection is clear, they agreed with Chan in that “the precise pathogenesis from suboptimal diets to these cancers remains unclear and requires further basic studies to clarify the mechanism.”
In the meantime, the findings “underscore the urgent need for proactive public health interventions. Diet, as a modifiable risk factor, still offers substantial potential for improvement,” Liu said.
This study was funded by the National Natural Science Foundation of China and the American Cancer Society. The authors and Chan disclosed no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
according to a recent study.
Writing in Gastroenterology, researchers led by Li Liu, PhD, of the department of epidemiology and biostatistics, Ministry of Education Key Lab of Environment and Health, School of Public Health, Tongji Medical College, Huazhong University of Science and Technology in Wuhan, China, reported that excessive consumption of processed meats (the biggest culprit), insufficient fruit intake, and insufficient whole grain intake were the leading dietary risk factors. In addition, the number of diet-related cases doubled from 1990 to 2018.
“In regions with limited access to healthy foods, policy interventions like taxing unhealthy foods and subsidizing nutritious options may help shift dietary patterns and reduce cancer risk,” Liu said in an interview.
The study examined meta-analyses from 184 countries in seven regions for the period 1990-2018 looking at rates of six major GI cancers: colorectal, liver, esophageal, pancreatic, and gallbladder/biliary tract. Among these, the age-standardized incidence of liver, pancreatic, and colorectal increased significantly over the past 3 decades.
The research team used a comparative risk assessment model to estimate the impact of diet on GI cancer independent of energy intake and adiposity. Although the principal dietary risk factors varied across individual cancers, suboptimal intake of the three aforementioned components was responsible for 66.51% of all diet-attributable GI cancers in 2018. The global mean processed meat consumption was 17 g/d in 2018, falling to a low in South Asia of 3 g/d.
The investigators also found diet-linked cancer incidence positively correlated with the Sociodemographic Index (SDI), an integrated measure of national development, income, and fertility. Incidence varied across world regions, with the highest proportion of cases in Central and Eastern Europe, Central Asia, Latin America, the Caribbean, and in high-income countries. The findings support the development of targeted diet-related public health interventions in various regions and nations to reduce GI cancer incidence, the authors wrote.
Among the study’s specific findings:
- In 2018, 21.5% (95% uncertainty interval [UI], 19.1-24.5) of incident GI cancer cases globally were attributable to suboptimal diets, a relatively stable proportion since 1990 (22.4%; 95% UI, 19.7-25.6).
- Absolute diet-attributable cases doubled from 580,862 (95% UI, 510,658-664,076) in 1990 to 1,039,877 (95% UI, 923,482-1,187,244) in 2018.
- Excessive processed meat consumption (5.9%; 95% UI, 4.2%-7.9%), insufficient fruit intake (4.8%; 95% UI, 3.8%-5.9%), and insufficient whole grain intake (3.6%; 95% UI, 2.8%-5.1%) were the most significant dietary risk factors in 2018 — a shift from 1990 when the third major concern was insufficient non-starchy vegetable intake.
Given the well-established link between diet and GI cancers, the incidence findings came as no surprise. “However, the dramatic doubling of diet-attributable cases over the past few decades was truly unexpected,” Liu said. “This increase can likely be attributed to global population growth and aging. While aging is an irreversible process, we can still reduce the growing burden of diet-related GI cancers by focusing on modifiable behaviors, particularly through targeted dietary interventions.”
A Modifiable Risk Factor
Commenting on the analysis but not involved in it, Andrew T. Chan, MD, MPH, a professor of medicine at Harvard Medical School and a gastroenterologist at Massachusetts General Hospital, both in Boston, noted that his own group’s studies also support the association of diet with an increased risk for GI cancers, particularly colorectal cancers.
“Although much work needs to be done to clarify the precise mechanisms underlying this association, there are substantial data that diet may cause changes in the gut microbiome, which in turn promotes cancer,” Chan said in an interview. “Going forward, we are working to develop strategies in which diet is modified to mitigate the risk of cancer associated with suboptimal diets.”
In other study findings, Liu’s group observed that two regional groups, Central and Eastern Europe, Central Asia, Latin America, and the Caribbean, as well as high-income countries, bore the top three diet-attributable burdens worldwide in 2018, all driven mostly by an upward-trending excess of processed meat.
By regions, Central and Eastern Europe and Central Asia experienced the highest attributable burden across regions in 1990 (31.6%; UI, 27.0%-37.4%) and 2018 (31.6%; UI, 27.3%-36.5%).
As for the impact of the SDI, the authors explained that diet-attributable GI cancer burden was higher among adults with higher education and living in urban areas than among those with lower education and rural residency. “Some dietary habits tended to be worse in higher-SDI countries, specifically, higher consumption of processed meats,” they wrote.
Although the proportional attributable GI incidence remains relatively stable, they added, the doubling of absolute cases from 1990 to 2018, along with the discrepancies between urbanicity and countries/regions, supports more targeted preventive measures.
And while the diet-GI cancer connection is clear, they agreed with Chan in that “the precise pathogenesis from suboptimal diets to these cancers remains unclear and requires further basic studies to clarify the mechanism.”
In the meantime, the findings “underscore the urgent need for proactive public health interventions. Diet, as a modifiable risk factor, still offers substantial potential for improvement,” Liu said.
This study was funded by the National Natural Science Foundation of China and the American Cancer Society. The authors and Chan disclosed no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
according to a recent study.
Writing in Gastroenterology, researchers led by Li Liu, PhD, of the department of epidemiology and biostatistics, Ministry of Education Key Lab of Environment and Health, School of Public Health, Tongji Medical College, Huazhong University of Science and Technology in Wuhan, China, reported that excessive consumption of processed meats (the biggest culprit), insufficient fruit intake, and insufficient whole grain intake were the leading dietary risk factors. In addition, the number of diet-related cases doubled from 1990 to 2018.
“In regions with limited access to healthy foods, policy interventions like taxing unhealthy foods and subsidizing nutritious options may help shift dietary patterns and reduce cancer risk,” Liu said in an interview.
The study examined meta-analyses from 184 countries in seven regions for the period 1990-2018 looking at rates of six major GI cancers: colorectal, liver, esophageal, pancreatic, and gallbladder/biliary tract. Among these, the age-standardized incidence of liver, pancreatic, and colorectal increased significantly over the past 3 decades.
The research team used a comparative risk assessment model to estimate the impact of diet on GI cancer independent of energy intake and adiposity. Although the principal dietary risk factors varied across individual cancers, suboptimal intake of the three aforementioned components was responsible for 66.51% of all diet-attributable GI cancers in 2018. The global mean processed meat consumption was 17 g/d in 2018, falling to a low in South Asia of 3 g/d.
The investigators also found diet-linked cancer incidence positively correlated with the Sociodemographic Index (SDI), an integrated measure of national development, income, and fertility. Incidence varied across world regions, with the highest proportion of cases in Central and Eastern Europe, Central Asia, Latin America, the Caribbean, and in high-income countries. The findings support the development of targeted diet-related public health interventions in various regions and nations to reduce GI cancer incidence, the authors wrote.
Among the study’s specific findings:
- In 2018, 21.5% (95% uncertainty interval [UI], 19.1-24.5) of incident GI cancer cases globally were attributable to suboptimal diets, a relatively stable proportion since 1990 (22.4%; 95% UI, 19.7-25.6).
- Absolute diet-attributable cases doubled from 580,862 (95% UI, 510,658-664,076) in 1990 to 1,039,877 (95% UI, 923,482-1,187,244) in 2018.
- Excessive processed meat consumption (5.9%; 95% UI, 4.2%-7.9%), insufficient fruit intake (4.8%; 95% UI, 3.8%-5.9%), and insufficient whole grain intake (3.6%; 95% UI, 2.8%-5.1%) were the most significant dietary risk factors in 2018 — a shift from 1990 when the third major concern was insufficient non-starchy vegetable intake.
Given the well-established link between diet and GI cancers, the incidence findings came as no surprise. “However, the dramatic doubling of diet-attributable cases over the past few decades was truly unexpected,” Liu said. “This increase can likely be attributed to global population growth and aging. While aging is an irreversible process, we can still reduce the growing burden of diet-related GI cancers by focusing on modifiable behaviors, particularly through targeted dietary interventions.”
A Modifiable Risk Factor
Commenting on the analysis but not involved in it, Andrew T. Chan, MD, MPH, a professor of medicine at Harvard Medical School and a gastroenterologist at Massachusetts General Hospital, both in Boston, noted that his own group’s studies also support the association of diet with an increased risk for GI cancers, particularly colorectal cancers.
“Although much work needs to be done to clarify the precise mechanisms underlying this association, there are substantial data that diet may cause changes in the gut microbiome, which in turn promotes cancer,” Chan said in an interview. “Going forward, we are working to develop strategies in which diet is modified to mitigate the risk of cancer associated with suboptimal diets.”
In other study findings, Liu’s group observed that two regional groups, Central and Eastern Europe, Central Asia, Latin America, and the Caribbean, as well as high-income countries, bore the top three diet-attributable burdens worldwide in 2018, all driven mostly by an upward-trending excess of processed meat.
By regions, Central and Eastern Europe and Central Asia experienced the highest attributable burden across regions in 1990 (31.6%; UI, 27.0%-37.4%) and 2018 (31.6%; UI, 27.3%-36.5%).
As for the impact of the SDI, the authors explained that diet-attributable GI cancer burden was higher among adults with higher education and living in urban areas than among those with lower education and rural residency. “Some dietary habits tended to be worse in higher-SDI countries, specifically, higher consumption of processed meats,” they wrote.
Although the proportional attributable GI incidence remains relatively stable, they added, the doubling of absolute cases from 1990 to 2018, along with the discrepancies between urbanicity and countries/regions, supports more targeted preventive measures.
And while the diet-GI cancer connection is clear, they agreed with Chan in that “the precise pathogenesis from suboptimal diets to these cancers remains unclear and requires further basic studies to clarify the mechanism.”
In the meantime, the findings “underscore the urgent need for proactive public health interventions. Diet, as a modifiable risk factor, still offers substantial potential for improvement,” Liu said.
This study was funded by the National Natural Science Foundation of China and the American Cancer Society. The authors and Chan disclosed no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
FROM GASTROENTEROLOGY
Reports Find Room for Improvement in VA Suicide-Risk Screening
About 18 veterans die by suicide daily, and while many received health care services in the year prior to their death, half did not receive a mental health diagnosis.
To address this, the Veterans Health Administration (VHA) has updated or initiated programs and policies aimed at identifying at-risk veterans. Since May 2018, the VHA introduced the Suicide Risk Identification Strategy (Risk ID) program, which includes screening patients using the Columbia-Suicide Severity Rating Scale (C-SSRS). Positive screenings call for a licensed independent clinician to document a comprehensive suicide risk evaluation.
Despite these measures, challenges persist in implementation and effectiveness, outlined in reports issue by the VA Office of Inspector General (OIG) during the Biden Administration. Michael Missal, who had served as VA Inspector General since 2016 was recently dismissed by President Trump.
Risk ID
The OIG report surveyed 137 facilities regarding Risk ID processes, training, and monitoring. Findings from that review revealed gaps in training: suicide prevention training does not adequately address Risk ID requirements, leaving staff unprepared to conduct screenings and evaluations. Although the VHA has developed additional training related to Risk ID, the training is not required and the VHA does not monitor staff training completion.
The VHA requires annual screening for all patients and has established a screening clinical reminder in patients’ electronic health records. Despite this, the national screening metric remained below 60% in 2023. Conversely, same-day evaluations after positive screenings reached 82%, though this metric excludes patients who were not screened. In 2024, the VHA added Risk ID evaluation metrics to leadership performance plans, aiming to clarify standards and promote adherence.
Mental Health Treatment Coordinators
A second OIG investigation from December 2024 reviewed VHA requirements related to suicide risk identification processes and also evaluated national compliance with mental health treatment coordinator (MHTC) role requirements.
Suicide risks peaks after discharge from mental health units, with 40% of suicidal behaviors occurring within 90 days. The VHA requires suicide risk screening within 24 hours of discharge and safety plans for high-risk patients using the C-SSRS, but the OIG found adherence issues. In a review of 200 patients discharged between October 2019 and September 2020, staff failed to complete the required screening for 27% of patients and safety plans for 12% of patients.
The VHA also requires clinicians to develop a safety plan with patients who recently attempted suicide or expressed suicidal ideation, are at risk of suicide prior to mental health unit discharge, or are determined to be at “high or intermediate acute or chronic risk” of suicide. For those patients, staff must flag the electronic health record.
OIG also found that over half of surveyed patients with an assigned MHTC were not able to identify the MHTC or another VHA staff member to contact for help with care. One-third of assigned MHTCs did not participate in patients’ transitions from inpatient to outpatient care. Despite the VHA no longer requiring 7-day follow-up appointments as of 2023, the OIG emphasized the need for guidance on scheduling postdischarge mental health appointments to promote engagement.
Consistent with VHA’s discontinuation of a required 7-day follow-up appointment, the OIG recognizes that postdischarge follow-up appointments are most effectively scheduled in consideration of a patient’s treatment needs, preferences, and availability rather than an arbitrary timeliness expectation. Patients flagged as high-risk must attend 4 mental health visits within 30 days of discharge. However, the OIG found that only 48% met this requirement, while 34% attended 1 to 3 appointments, and 18% attended none. Among surveyed patients, self-motivation and encouragement from family or friends were key drivers of attendance.
The OIG concluded that failures in suicide risk identification and care coordination could lead to underestimated suicide risk, overestimated discharge readiness, and unmitigated risks. Inadequate safety planning may also leave patients ill-equipped to manage crises. While the VHA has updated guidelines for MHTC involvement, these measures have not significantly improved continuity of care.
About 18 veterans die by suicide daily, and while many received health care services in the year prior to their death, half did not receive a mental health diagnosis.
To address this, the Veterans Health Administration (VHA) has updated or initiated programs and policies aimed at identifying at-risk veterans. Since May 2018, the VHA introduced the Suicide Risk Identification Strategy (Risk ID) program, which includes screening patients using the Columbia-Suicide Severity Rating Scale (C-SSRS). Positive screenings call for a licensed independent clinician to document a comprehensive suicide risk evaluation.
Despite these measures, challenges persist in implementation and effectiveness, outlined in reports issue by the VA Office of Inspector General (OIG) during the Biden Administration. Michael Missal, who had served as VA Inspector General since 2016 was recently dismissed by President Trump.
Risk ID
The OIG report surveyed 137 facilities regarding Risk ID processes, training, and monitoring. Findings from that review revealed gaps in training: suicide prevention training does not adequately address Risk ID requirements, leaving staff unprepared to conduct screenings and evaluations. Although the VHA has developed additional training related to Risk ID, the training is not required and the VHA does not monitor staff training completion.
The VHA requires annual screening for all patients and has established a screening clinical reminder in patients’ electronic health records. Despite this, the national screening metric remained below 60% in 2023. Conversely, same-day evaluations after positive screenings reached 82%, though this metric excludes patients who were not screened. In 2024, the VHA added Risk ID evaluation metrics to leadership performance plans, aiming to clarify standards and promote adherence.
Mental Health Treatment Coordinators
A second OIG investigation from December 2024 reviewed VHA requirements related to suicide risk identification processes and also evaluated national compliance with mental health treatment coordinator (MHTC) role requirements.
Suicide risks peaks after discharge from mental health units, with 40% of suicidal behaviors occurring within 90 days. The VHA requires suicide risk screening within 24 hours of discharge and safety plans for high-risk patients using the C-SSRS, but the OIG found adherence issues. In a review of 200 patients discharged between October 2019 and September 2020, staff failed to complete the required screening for 27% of patients and safety plans for 12% of patients.
The VHA also requires clinicians to develop a safety plan with patients who recently attempted suicide or expressed suicidal ideation, are at risk of suicide prior to mental health unit discharge, or are determined to be at “high or intermediate acute or chronic risk” of suicide. For those patients, staff must flag the electronic health record.
OIG also found that over half of surveyed patients with an assigned MHTC were not able to identify the MHTC or another VHA staff member to contact for help with care. One-third of assigned MHTCs did not participate in patients’ transitions from inpatient to outpatient care. Despite the VHA no longer requiring 7-day follow-up appointments as of 2023, the OIG emphasized the need for guidance on scheduling postdischarge mental health appointments to promote engagement.
Consistent with VHA’s discontinuation of a required 7-day follow-up appointment, the OIG recognizes that postdischarge follow-up appointments are most effectively scheduled in consideration of a patient’s treatment needs, preferences, and availability rather than an arbitrary timeliness expectation. Patients flagged as high-risk must attend 4 mental health visits within 30 days of discharge. However, the OIG found that only 48% met this requirement, while 34% attended 1 to 3 appointments, and 18% attended none. Among surveyed patients, self-motivation and encouragement from family or friends were key drivers of attendance.
The OIG concluded that failures in suicide risk identification and care coordination could lead to underestimated suicide risk, overestimated discharge readiness, and unmitigated risks. Inadequate safety planning may also leave patients ill-equipped to manage crises. While the VHA has updated guidelines for MHTC involvement, these measures have not significantly improved continuity of care.
About 18 veterans die by suicide daily, and while many received health care services in the year prior to their death, half did not receive a mental health diagnosis.
To address this, the Veterans Health Administration (VHA) has updated or initiated programs and policies aimed at identifying at-risk veterans. Since May 2018, the VHA introduced the Suicide Risk Identification Strategy (Risk ID) program, which includes screening patients using the Columbia-Suicide Severity Rating Scale (C-SSRS). Positive screenings call for a licensed independent clinician to document a comprehensive suicide risk evaluation.
Despite these measures, challenges persist in implementation and effectiveness, outlined in reports issue by the VA Office of Inspector General (OIG) during the Biden Administration. Michael Missal, who had served as VA Inspector General since 2016 was recently dismissed by President Trump.
Risk ID
The OIG report surveyed 137 facilities regarding Risk ID processes, training, and monitoring. Findings from that review revealed gaps in training: suicide prevention training does not adequately address Risk ID requirements, leaving staff unprepared to conduct screenings and evaluations. Although the VHA has developed additional training related to Risk ID, the training is not required and the VHA does not monitor staff training completion.
The VHA requires annual screening for all patients and has established a screening clinical reminder in patients’ electronic health records. Despite this, the national screening metric remained below 60% in 2023. Conversely, same-day evaluations after positive screenings reached 82%, though this metric excludes patients who were not screened. In 2024, the VHA added Risk ID evaluation metrics to leadership performance plans, aiming to clarify standards and promote adherence.
Mental Health Treatment Coordinators
A second OIG investigation from December 2024 reviewed VHA requirements related to suicide risk identification processes and also evaluated national compliance with mental health treatment coordinator (MHTC) role requirements.
Suicide risks peaks after discharge from mental health units, with 40% of suicidal behaviors occurring within 90 days. The VHA requires suicide risk screening within 24 hours of discharge and safety plans for high-risk patients using the C-SSRS, but the OIG found adherence issues. In a review of 200 patients discharged between October 2019 and September 2020, staff failed to complete the required screening for 27% of patients and safety plans for 12% of patients.
The VHA also requires clinicians to develop a safety plan with patients who recently attempted suicide or expressed suicidal ideation, are at risk of suicide prior to mental health unit discharge, or are determined to be at “high or intermediate acute or chronic risk” of suicide. For those patients, staff must flag the electronic health record.
OIG also found that over half of surveyed patients with an assigned MHTC were not able to identify the MHTC or another VHA staff member to contact for help with care. One-third of assigned MHTCs did not participate in patients’ transitions from inpatient to outpatient care. Despite the VHA no longer requiring 7-day follow-up appointments as of 2023, the OIG emphasized the need for guidance on scheduling postdischarge mental health appointments to promote engagement.
Consistent with VHA’s discontinuation of a required 7-day follow-up appointment, the OIG recognizes that postdischarge follow-up appointments are most effectively scheduled in consideration of a patient’s treatment needs, preferences, and availability rather than an arbitrary timeliness expectation. Patients flagged as high-risk must attend 4 mental health visits within 30 days of discharge. However, the OIG found that only 48% met this requirement, while 34% attended 1 to 3 appointments, and 18% attended none. Among surveyed patients, self-motivation and encouragement from family or friends were key drivers of attendance.
The OIG concluded that failures in suicide risk identification and care coordination could lead to underestimated suicide risk, overestimated discharge readiness, and unmitigated risks. Inadequate safety planning may also leave patients ill-equipped to manage crises. While the VHA has updated guidelines for MHTC involvement, these measures have not significantly improved continuity of care.
Promising New Blood Test for Colorectal Cancer Screening
, the largest study of any blood-based CRC screening test.
With continued optimization, this blood test “may provide a convenient, effective option for colorectal cancer screening in the intended use population,” said Aasma Shaukat, MD, MPH, AGAF, with New York University Grossman School of Medicine in New York City.
CRC screening rates remain suboptimal, with nearly 40% of eligible adults in the United States not up to date with screening, noted Shaukat, who presented the study results at the American Society of Clinical Oncology (ASCO) Gastrointestinal Cancers Symposium 2025 in San Francisco.
Blood-based testing offers a promising complementary approach that may boost patient adherence among unscreened individuals, she added.
The study evaluated the clinical performance of the investigational blood-based screening test in 27,010 adults aged ≥ 45 years at average-risk for CRC. Patients with no personal history of cancer, colorectal adenoma, or inflammatory bowel disease, as well as no family history of CRC or hereditary gastrointestinal cancer syndromes were eligible for the study.
Participants had blood drawn before bowel preparation for colonoscopy, and the blood test results were measured against colonoscopy findings.
The primary endpoints included sensitivity for CRC, specificity for advanced colorectal neoplasia, and negative and positive predictive value for advanced colorectal neoplasia. A secondary endpoint was sensitivity for advanced precancerous lesions.
The blood-based screening test met all primary endpoints, with a sensitivity for CRC of 79.2% and a specificity for advanced colorectal neoplasia of 91.5%, Shaukat reported at a conference briefing. Negative predictive value for advanced colorectal neoplasia was 90.8%, though the positive predictive value was only 15.5% and sensitivity for advanced precancerous lesions, a secondary endpoint, was 12.5%.
Similar results were achieved in a prespecified analysis in which performance of the blood test was weighted to match US census data for sex and age distributions. Sensitivity for CRC was 81.1%, specificity was 90.4%, and negative predictive value for advanced colorectal neoplasia was 90.5%; but the positive predictive value was 15.5%, and the sensitivity for advanced precancerous lesions was 13.7%.
This type of analysis provides “a snapshot of how [the test] would perform in the US population,” Shaukat explained, adding that the sensitivity for CRC and advanced precursor lesions was “lower than expected and will continue to be optimized in future research and development.”
It will also be important to determine when the test should be repeated and how often and to look at the determinants around cost and comparative effectiveness, she said. Modeling and other outcome studies — which will be forthcoming in the future years — could help shed some light on these questions.
Briefing moderator Julie Gralow, MD, ASCO chief medical officer, said it will be important to compare how this new blood test compares with Guardant Health’s Shield CRC blood test that was approved in 2024. Although there’s no study directly comparing the new blood test to Shield, data from the ECLIPSE study reported that Shield had 83% sensitivity for CRC and 90% specificity for advanced neoplasia, but only 13% sensitivity for advanced precancerous lesions — which appears similar, so far, to findings reported for the new blood test.
But any screening is better than no screening, and with further study, this blood test may “add another tool to our toolkit,” said Pamela Kunz, MD, director of the Center for Gastrointestinal Cancers at Smilow Cancer Hospital and Yale Cancer Center, New Haven, Connecticut, who spoke at the briefing. Kunz said she is eager to see some of the future work optimizing the sensitivity and subsequent analyses that look at differences by race and ethnicity.
The study had no specific funding. Shaukat consults for Freenome Holdings and Iterative Health. Kunz declared ties with Ipsen, Novartis, Genentech/Roche, Amgen, Crinetics Pharmaceuticals, Natera, HUTCHMED, and ITM Isotope Technologies Munich. Gralow declared consulting or advisory roles with Genentech/Roche.
A version of this article appeared on Medscape.com .
, the largest study of any blood-based CRC screening test.
With continued optimization, this blood test “may provide a convenient, effective option for colorectal cancer screening in the intended use population,” said Aasma Shaukat, MD, MPH, AGAF, with New York University Grossman School of Medicine in New York City.
CRC screening rates remain suboptimal, with nearly 40% of eligible adults in the United States not up to date with screening, noted Shaukat, who presented the study results at the American Society of Clinical Oncology (ASCO) Gastrointestinal Cancers Symposium 2025 in San Francisco.
Blood-based testing offers a promising complementary approach that may boost patient adherence among unscreened individuals, she added.
The study evaluated the clinical performance of the investigational blood-based screening test in 27,010 adults aged ≥ 45 years at average-risk for CRC. Patients with no personal history of cancer, colorectal adenoma, or inflammatory bowel disease, as well as no family history of CRC or hereditary gastrointestinal cancer syndromes were eligible for the study.
Participants had blood drawn before bowel preparation for colonoscopy, and the blood test results were measured against colonoscopy findings.
The primary endpoints included sensitivity for CRC, specificity for advanced colorectal neoplasia, and negative and positive predictive value for advanced colorectal neoplasia. A secondary endpoint was sensitivity for advanced precancerous lesions.
The blood-based screening test met all primary endpoints, with a sensitivity for CRC of 79.2% and a specificity for advanced colorectal neoplasia of 91.5%, Shaukat reported at a conference briefing. Negative predictive value for advanced colorectal neoplasia was 90.8%, though the positive predictive value was only 15.5% and sensitivity for advanced precancerous lesions, a secondary endpoint, was 12.5%.
Similar results were achieved in a prespecified analysis in which performance of the blood test was weighted to match US census data for sex and age distributions. Sensitivity for CRC was 81.1%, specificity was 90.4%, and negative predictive value for advanced colorectal neoplasia was 90.5%; but the positive predictive value was 15.5%, and the sensitivity for advanced precancerous lesions was 13.7%.
This type of analysis provides “a snapshot of how [the test] would perform in the US population,” Shaukat explained, adding that the sensitivity for CRC and advanced precursor lesions was “lower than expected and will continue to be optimized in future research and development.”
It will also be important to determine when the test should be repeated and how often and to look at the determinants around cost and comparative effectiveness, she said. Modeling and other outcome studies — which will be forthcoming in the future years — could help shed some light on these questions.
Briefing moderator Julie Gralow, MD, ASCO chief medical officer, said it will be important to compare how this new blood test compares with Guardant Health’s Shield CRC blood test that was approved in 2024. Although there’s no study directly comparing the new blood test to Shield, data from the ECLIPSE study reported that Shield had 83% sensitivity for CRC and 90% specificity for advanced neoplasia, but only 13% sensitivity for advanced precancerous lesions — which appears similar, so far, to findings reported for the new blood test.
But any screening is better than no screening, and with further study, this blood test may “add another tool to our toolkit,” said Pamela Kunz, MD, director of the Center for Gastrointestinal Cancers at Smilow Cancer Hospital and Yale Cancer Center, New Haven, Connecticut, who spoke at the briefing. Kunz said she is eager to see some of the future work optimizing the sensitivity and subsequent analyses that look at differences by race and ethnicity.
The study had no specific funding. Shaukat consults for Freenome Holdings and Iterative Health. Kunz declared ties with Ipsen, Novartis, Genentech/Roche, Amgen, Crinetics Pharmaceuticals, Natera, HUTCHMED, and ITM Isotope Technologies Munich. Gralow declared consulting or advisory roles with Genentech/Roche.
A version of this article appeared on Medscape.com .
, the largest study of any blood-based CRC screening test.
With continued optimization, this blood test “may provide a convenient, effective option for colorectal cancer screening in the intended use population,” said Aasma Shaukat, MD, MPH, AGAF, with New York University Grossman School of Medicine in New York City.
CRC screening rates remain suboptimal, with nearly 40% of eligible adults in the United States not up to date with screening, noted Shaukat, who presented the study results at the American Society of Clinical Oncology (ASCO) Gastrointestinal Cancers Symposium 2025 in San Francisco.
Blood-based testing offers a promising complementary approach that may boost patient adherence among unscreened individuals, she added.
The study evaluated the clinical performance of the investigational blood-based screening test in 27,010 adults aged ≥ 45 years at average-risk for CRC. Patients with no personal history of cancer, colorectal adenoma, or inflammatory bowel disease, as well as no family history of CRC or hereditary gastrointestinal cancer syndromes were eligible for the study.
Participants had blood drawn before bowel preparation for colonoscopy, and the blood test results were measured against colonoscopy findings.
The primary endpoints included sensitivity for CRC, specificity for advanced colorectal neoplasia, and negative and positive predictive value for advanced colorectal neoplasia. A secondary endpoint was sensitivity for advanced precancerous lesions.
The blood-based screening test met all primary endpoints, with a sensitivity for CRC of 79.2% and a specificity for advanced colorectal neoplasia of 91.5%, Shaukat reported at a conference briefing. Negative predictive value for advanced colorectal neoplasia was 90.8%, though the positive predictive value was only 15.5% and sensitivity for advanced precancerous lesions, a secondary endpoint, was 12.5%.
Similar results were achieved in a prespecified analysis in which performance of the blood test was weighted to match US census data for sex and age distributions. Sensitivity for CRC was 81.1%, specificity was 90.4%, and negative predictive value for advanced colorectal neoplasia was 90.5%; but the positive predictive value was 15.5%, and the sensitivity for advanced precancerous lesions was 13.7%.
This type of analysis provides “a snapshot of how [the test] would perform in the US population,” Shaukat explained, adding that the sensitivity for CRC and advanced precursor lesions was “lower than expected and will continue to be optimized in future research and development.”
It will also be important to determine when the test should be repeated and how often and to look at the determinants around cost and comparative effectiveness, she said. Modeling and other outcome studies — which will be forthcoming in the future years — could help shed some light on these questions.
Briefing moderator Julie Gralow, MD, ASCO chief medical officer, said it will be important to compare how this new blood test compares with Guardant Health’s Shield CRC blood test that was approved in 2024. Although there’s no study directly comparing the new blood test to Shield, data from the ECLIPSE study reported that Shield had 83% sensitivity for CRC and 90% specificity for advanced neoplasia, but only 13% sensitivity for advanced precancerous lesions — which appears similar, so far, to findings reported for the new blood test.
But any screening is better than no screening, and with further study, this blood test may “add another tool to our toolkit,” said Pamela Kunz, MD, director of the Center for Gastrointestinal Cancers at Smilow Cancer Hospital and Yale Cancer Center, New Haven, Connecticut, who spoke at the briefing. Kunz said she is eager to see some of the future work optimizing the sensitivity and subsequent analyses that look at differences by race and ethnicity.
The study had no specific funding. Shaukat consults for Freenome Holdings and Iterative Health. Kunz declared ties with Ipsen, Novartis, Genentech/Roche, Amgen, Crinetics Pharmaceuticals, Natera, HUTCHMED, and ITM Isotope Technologies Munich. Gralow declared consulting or advisory roles with Genentech/Roche.
A version of this article appeared on Medscape.com .
FROM ASCO GI 2025
Low-Dose Aspirin Cuts CRC Recurrence
according to findings from the phase 3 ALASCCA trial.
These results stress “the importance of upfront genomic testing” in patients with CRC, said Anna Martling, MD, PhD, from Karolinska Institutet, Stockholm, Sweden, who reported the findings at the American Society of Clinical Oncology (ASCO) Gastrointestinal Cancers Symposium 2025 in San Francisco.
This is the first trial to show that mutations in the PI3K signaling pathway, beyond PIK3CA alterations, predict aspirin response, “expanding the targetable patient population substantially,” Martling added. Genetic mutations along the PI3K signaling pathway are found in about 30% of CRCs.
While aspirin as chemoprevention in CRC has been studied, data confirming its effectiveness as well as uptake of this approach in practice have been lacking, explained ASCO expert commenter Pamela Kunz, MD, with Smilow Cancer Hospital and Yale Cancer Center, New Haven, Connecticut.
“It’s really clear that this is a practice-changing study,” said Kunz. The findings indicate that this approach “checks all of the boxes: It’s effective, it’s low risk, it’s inexpensive, and it’s easy to administer.”
The trial included 626 patients (median age, 66 years; 52% women) with stages II-III colon cancer (67%) or stages I-III rectal cancer (33%) across 33 hospitals in Sweden, Denmark, Finland, and Norway.
Patients were stratified into two groups based on specific PI3K pathway alterations Group A (n = 314) included patients with PIK3CA mutations in exon 9 and/or 20, and group B (n = 312) included those with other PI3K pathway mutations, including PIK3CA mutations outside exon 9/20, or mutations in PIK3R1 or PTEN genes.
Participants in both groups were randomly allocated 1:1 to 160 mg/d of aspirin or placebo for 3 years. The primary outcome was CRC recurrence; disease-free survival was a secondary outcome.
Compared with placebo, aspirin reduced the risk for recurrence by 51% (hazard ratio [HR], 0.49) in patients with PIK3CA mutations, with a 3-year recurrence rate of 7.7% in those taking aspirin vs 14.1% in the placebo group.
“Interestingly,” Martling noted, in the exploratory arm that included other mutations along the PIK3 pathway beyond PIK3CA (Group B), the effect was even stronger. Patients in this group had a 58% (HR, 0.42) lower risk for recurrence than those in the placebo group, with a 3-year recurrence rate of 7.7% in the aspirin group vs 16.8% recurrence rate in the placebo group.
Aspirin also had a disease-free survival benefit in both groups, but it was significant only in group B.
While the study was not specifically designed for subgroup analysis, the benefit of aspirin was observed in all subgroups examined, including men and women with colon or rectal cancer, those who did and did not receive neoadjuvant or adjuvant treatment, and those with stages I-III disease.
The incidence of adverse events was as expected and severe side effects associated with 160 mg/d aspirin were rare, Martling said.
Both Martling and Kunz predicted that these findings will change clinical practice. “I anticipate that we’ll be seeing adoption of this [strategy],” Kunz said.
This study received funding from the Swedish Research Council, Swedish Cancer Society, ALF (a regional agreement on medical training and clinical research between the Stockholm County Council and Karolinska Institutet), and the Stockholm Cancer Society. Martling disclosed various relationships with Bactiguard, Smartcella, CarpoNovum and Pfizer. Kunz disclosed relationships with Bristol-Myers Squibb, Novartis and TayzeBio.
A version of this article appeared on Medscape.com .
according to findings from the phase 3 ALASCCA trial.
These results stress “the importance of upfront genomic testing” in patients with CRC, said Anna Martling, MD, PhD, from Karolinska Institutet, Stockholm, Sweden, who reported the findings at the American Society of Clinical Oncology (ASCO) Gastrointestinal Cancers Symposium 2025 in San Francisco.
This is the first trial to show that mutations in the PI3K signaling pathway, beyond PIK3CA alterations, predict aspirin response, “expanding the targetable patient population substantially,” Martling added. Genetic mutations along the PI3K signaling pathway are found in about 30% of CRCs.
While aspirin as chemoprevention in CRC has been studied, data confirming its effectiveness as well as uptake of this approach in practice have been lacking, explained ASCO expert commenter Pamela Kunz, MD, with Smilow Cancer Hospital and Yale Cancer Center, New Haven, Connecticut.
“It’s really clear that this is a practice-changing study,” said Kunz. The findings indicate that this approach “checks all of the boxes: It’s effective, it’s low risk, it’s inexpensive, and it’s easy to administer.”
The trial included 626 patients (median age, 66 years; 52% women) with stages II-III colon cancer (67%) or stages I-III rectal cancer (33%) across 33 hospitals in Sweden, Denmark, Finland, and Norway.
Patients were stratified into two groups based on specific PI3K pathway alterations Group A (n = 314) included patients with PIK3CA mutations in exon 9 and/or 20, and group B (n = 312) included those with other PI3K pathway mutations, including PIK3CA mutations outside exon 9/20, or mutations in PIK3R1 or PTEN genes.
Participants in both groups were randomly allocated 1:1 to 160 mg/d of aspirin or placebo for 3 years. The primary outcome was CRC recurrence; disease-free survival was a secondary outcome.
Compared with placebo, aspirin reduced the risk for recurrence by 51% (hazard ratio [HR], 0.49) in patients with PIK3CA mutations, with a 3-year recurrence rate of 7.7% in those taking aspirin vs 14.1% in the placebo group.
“Interestingly,” Martling noted, in the exploratory arm that included other mutations along the PIK3 pathway beyond PIK3CA (Group B), the effect was even stronger. Patients in this group had a 58% (HR, 0.42) lower risk for recurrence than those in the placebo group, with a 3-year recurrence rate of 7.7% in the aspirin group vs 16.8% recurrence rate in the placebo group.
Aspirin also had a disease-free survival benefit in both groups, but it was significant only in group B.
While the study was not specifically designed for subgroup analysis, the benefit of aspirin was observed in all subgroups examined, including men and women with colon or rectal cancer, those who did and did not receive neoadjuvant or adjuvant treatment, and those with stages I-III disease.
The incidence of adverse events was as expected and severe side effects associated with 160 mg/d aspirin were rare, Martling said.
Both Martling and Kunz predicted that these findings will change clinical practice. “I anticipate that we’ll be seeing adoption of this [strategy],” Kunz said.
This study received funding from the Swedish Research Council, Swedish Cancer Society, ALF (a regional agreement on medical training and clinical research between the Stockholm County Council and Karolinska Institutet), and the Stockholm Cancer Society. Martling disclosed various relationships with Bactiguard, Smartcella, CarpoNovum and Pfizer. Kunz disclosed relationships with Bristol-Myers Squibb, Novartis and TayzeBio.
A version of this article appeared on Medscape.com .
according to findings from the phase 3 ALASCCA trial.
These results stress “the importance of upfront genomic testing” in patients with CRC, said Anna Martling, MD, PhD, from Karolinska Institutet, Stockholm, Sweden, who reported the findings at the American Society of Clinical Oncology (ASCO) Gastrointestinal Cancers Symposium 2025 in San Francisco.
This is the first trial to show that mutations in the PI3K signaling pathway, beyond PIK3CA alterations, predict aspirin response, “expanding the targetable patient population substantially,” Martling added. Genetic mutations along the PI3K signaling pathway are found in about 30% of CRCs.
While aspirin as chemoprevention in CRC has been studied, data confirming its effectiveness as well as uptake of this approach in practice have been lacking, explained ASCO expert commenter Pamela Kunz, MD, with Smilow Cancer Hospital and Yale Cancer Center, New Haven, Connecticut.
“It’s really clear that this is a practice-changing study,” said Kunz. The findings indicate that this approach “checks all of the boxes: It’s effective, it’s low risk, it’s inexpensive, and it’s easy to administer.”
The trial included 626 patients (median age, 66 years; 52% women) with stages II-III colon cancer (67%) or stages I-III rectal cancer (33%) across 33 hospitals in Sweden, Denmark, Finland, and Norway.
Patients were stratified into two groups based on specific PI3K pathway alterations Group A (n = 314) included patients with PIK3CA mutations in exon 9 and/or 20, and group B (n = 312) included those with other PI3K pathway mutations, including PIK3CA mutations outside exon 9/20, or mutations in PIK3R1 or PTEN genes.
Participants in both groups were randomly allocated 1:1 to 160 mg/d of aspirin or placebo for 3 years. The primary outcome was CRC recurrence; disease-free survival was a secondary outcome.
Compared with placebo, aspirin reduced the risk for recurrence by 51% (hazard ratio [HR], 0.49) in patients with PIK3CA mutations, with a 3-year recurrence rate of 7.7% in those taking aspirin vs 14.1% in the placebo group.
“Interestingly,” Martling noted, in the exploratory arm that included other mutations along the PIK3 pathway beyond PIK3CA (Group B), the effect was even stronger. Patients in this group had a 58% (HR, 0.42) lower risk for recurrence than those in the placebo group, with a 3-year recurrence rate of 7.7% in the aspirin group vs 16.8% recurrence rate in the placebo group.
Aspirin also had a disease-free survival benefit in both groups, but it was significant only in group B.
While the study was not specifically designed for subgroup analysis, the benefit of aspirin was observed in all subgroups examined, including men and women with colon or rectal cancer, those who did and did not receive neoadjuvant or adjuvant treatment, and those with stages I-III disease.
The incidence of adverse events was as expected and severe side effects associated with 160 mg/d aspirin were rare, Martling said.
Both Martling and Kunz predicted that these findings will change clinical practice. “I anticipate that we’ll be seeing adoption of this [strategy],” Kunz said.
This study received funding from the Swedish Research Council, Swedish Cancer Society, ALF (a regional agreement on medical training and clinical research between the Stockholm County Council and Karolinska Institutet), and the Stockholm Cancer Society. Martling disclosed various relationships with Bactiguard, Smartcella, CarpoNovum and Pfizer. Kunz disclosed relationships with Bristol-Myers Squibb, Novartis and TayzeBio.
A version of this article appeared on Medscape.com .
FROM ASCO GI 2025






