User login
CAR T-cell therapy found safe, effective for HIV-associated lymphoma
HIV positivity does not preclude chimeric antigen receptor (CAR) T-cell therapy for patients with aggressive lymphoma, a report of two cases suggests. Both of the HIV-positive patients, one of whom had long-term psychiatric comorbidity, achieved durable remission on axicabtagene ciloleucel (Yescarta) without undue toxicity.
“To our knowledge, these are the first reported cases of CAR T-cell therapy administered to HIV-infected patients with lymphoma,” Jeremy S. Abramson, MD, of Massachusetts General Hospital, Boston and his colleagues wrote in Cancer. “Patients with HIV and AIDS, as well as those with preexisting mental illness, should not be considered disqualified from CAR T-cell therapy and deserve ongoing studies to optimize efficacy and safety in this population.”
The Food and Drug Administration has approved two CAR T-cell products that target the B-cell antigen CD19 for the treatment of refractory lymphoma. But their efficacy and safety in HIV-positive patients are unknown because this group has been excluded from pivotal clinical trials.
Dr. Abramson and coauthors detail the two cases of successful anti-CD19 CAR T-cell therapy with axicabtagene ciloleucel in patients with HIV-associated, refractory, high-grade B-cell lymphoma.
The first patient was an HIV-positive man with diffuse large B-cell lymphoma (DLBCL) of germinal center B-cell subtype who was intermittently adherent to antiretroviral therapy. His comorbidities included posttraumatic stress disorder and schizoaffective disorder.
Previous treatments for DLBCL included dose-adjusted etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab (EPOCH-R), and rituximab, ifosfamide, carboplatin, and etoposide (RICE). A recurrence precluded high-dose chemotherapy with autologous stem cell support.
With close multidisciplinary management, including psychiatric consultation, the patient became a candidate for CAR T-cell therapy and received axicabtagene ciloleucel. He experienced grade 2 cytokine release syndrome and grade 3 neurologic toxicity, both of which resolved with treatment. Imaging showed complete remission at approximately 3 months that was sustained at 1 year. Additionally, he had an undetectable HIV viral load and was psychiatrically stable.
The second patient was a man with AIDS-associated, non–germinal center B-cell, Epstein-Barr virus–positive DLBCL who was adherent to antiretroviral therapy. His lymphoma had recurred rapidly after initially responding to dose-adjusted EPOCH-R and then was refractory to combination rituximab and lenalidomide. He previously had hepatitis B virus, cytomegalovirus, and Mycobacterium avium complex infections.
Because of prolonged cytopenias and infectious complications after the previous lymphoma treatments, the patient was considered a poor candidate for high-dose chemotherapy. He underwent CAR T-cell therapy with axicabtagene ciloleucel and had a complete remission on day 28. Additionally, his HIV infection remained well controlled.
“Although much remains to be learned regarding CAR T-cell therapy in patients with refractory hematologic malignancies, with or without HIV infection, the cases presented herein demonstrate that patients with chemotherapy-refractory, high-grade B-cell lymphoma can successfully undergo autologous CAR T-cell manufacturing, and subsequently can safely tolerate CAR T-cell therapy and achieve a durable complete remission,” the researchers wrote. “These cases have further demonstrated the proactive, multidisciplinary care required to navigate a patient with high-risk lymphoma through CAR T-cell therapy with attention to significant medical and psychiatric comorbidities.”
Dr. Abramson reported that he has acted as a paid member of the scientific advisory board and as a paid consultant for Kite Pharma, which markets Yescarta, and several other companies.
SOURCE: Abramson JS et al. Cancer. 2019 Sep 10. doi: 10.1002/cncr.32411.
HIV positivity does not preclude chimeric antigen receptor (CAR) T-cell therapy for patients with aggressive lymphoma, a report of two cases suggests. Both of the HIV-positive patients, one of whom had long-term psychiatric comorbidity, achieved durable remission on axicabtagene ciloleucel (Yescarta) without undue toxicity.
“To our knowledge, these are the first reported cases of CAR T-cell therapy administered to HIV-infected patients with lymphoma,” Jeremy S. Abramson, MD, of Massachusetts General Hospital, Boston and his colleagues wrote in Cancer. “Patients with HIV and AIDS, as well as those with preexisting mental illness, should not be considered disqualified from CAR T-cell therapy and deserve ongoing studies to optimize efficacy and safety in this population.”
The Food and Drug Administration has approved two CAR T-cell products that target the B-cell antigen CD19 for the treatment of refractory lymphoma. But their efficacy and safety in HIV-positive patients are unknown because this group has been excluded from pivotal clinical trials.
Dr. Abramson and coauthors detail the two cases of successful anti-CD19 CAR T-cell therapy with axicabtagene ciloleucel in patients with HIV-associated, refractory, high-grade B-cell lymphoma.
The first patient was an HIV-positive man with diffuse large B-cell lymphoma (DLBCL) of germinal center B-cell subtype who was intermittently adherent to antiretroviral therapy. His comorbidities included posttraumatic stress disorder and schizoaffective disorder.
Previous treatments for DLBCL included dose-adjusted etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab (EPOCH-R), and rituximab, ifosfamide, carboplatin, and etoposide (RICE). A recurrence precluded high-dose chemotherapy with autologous stem cell support.
With close multidisciplinary management, including psychiatric consultation, the patient became a candidate for CAR T-cell therapy and received axicabtagene ciloleucel. He experienced grade 2 cytokine release syndrome and grade 3 neurologic toxicity, both of which resolved with treatment. Imaging showed complete remission at approximately 3 months that was sustained at 1 year. Additionally, he had an undetectable HIV viral load and was psychiatrically stable.
The second patient was a man with AIDS-associated, non–germinal center B-cell, Epstein-Barr virus–positive DLBCL who was adherent to antiretroviral therapy. His lymphoma had recurred rapidly after initially responding to dose-adjusted EPOCH-R and then was refractory to combination rituximab and lenalidomide. He previously had hepatitis B virus, cytomegalovirus, and Mycobacterium avium complex infections.
Because of prolonged cytopenias and infectious complications after the previous lymphoma treatments, the patient was considered a poor candidate for high-dose chemotherapy. He underwent CAR T-cell therapy with axicabtagene ciloleucel and had a complete remission on day 28. Additionally, his HIV infection remained well controlled.
“Although much remains to be learned regarding CAR T-cell therapy in patients with refractory hematologic malignancies, with or without HIV infection, the cases presented herein demonstrate that patients with chemotherapy-refractory, high-grade B-cell lymphoma can successfully undergo autologous CAR T-cell manufacturing, and subsequently can safely tolerate CAR T-cell therapy and achieve a durable complete remission,” the researchers wrote. “These cases have further demonstrated the proactive, multidisciplinary care required to navigate a patient with high-risk lymphoma through CAR T-cell therapy with attention to significant medical and psychiatric comorbidities.”
Dr. Abramson reported that he has acted as a paid member of the scientific advisory board and as a paid consultant for Kite Pharma, which markets Yescarta, and several other companies.
SOURCE: Abramson JS et al. Cancer. 2019 Sep 10. doi: 10.1002/cncr.32411.
HIV positivity does not preclude chimeric antigen receptor (CAR) T-cell therapy for patients with aggressive lymphoma, a report of two cases suggests. Both of the HIV-positive patients, one of whom had long-term psychiatric comorbidity, achieved durable remission on axicabtagene ciloleucel (Yescarta) without undue toxicity.
“To our knowledge, these are the first reported cases of CAR T-cell therapy administered to HIV-infected patients with lymphoma,” Jeremy S. Abramson, MD, of Massachusetts General Hospital, Boston and his colleagues wrote in Cancer. “Patients with HIV and AIDS, as well as those with preexisting mental illness, should not be considered disqualified from CAR T-cell therapy and deserve ongoing studies to optimize efficacy and safety in this population.”
The Food and Drug Administration has approved two CAR T-cell products that target the B-cell antigen CD19 for the treatment of refractory lymphoma. But their efficacy and safety in HIV-positive patients are unknown because this group has been excluded from pivotal clinical trials.
Dr. Abramson and coauthors detail the two cases of successful anti-CD19 CAR T-cell therapy with axicabtagene ciloleucel in patients with HIV-associated, refractory, high-grade B-cell lymphoma.
The first patient was an HIV-positive man with diffuse large B-cell lymphoma (DLBCL) of germinal center B-cell subtype who was intermittently adherent to antiretroviral therapy. His comorbidities included posttraumatic stress disorder and schizoaffective disorder.
Previous treatments for DLBCL included dose-adjusted etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab (EPOCH-R), and rituximab, ifosfamide, carboplatin, and etoposide (RICE). A recurrence precluded high-dose chemotherapy with autologous stem cell support.
With close multidisciplinary management, including psychiatric consultation, the patient became a candidate for CAR T-cell therapy and received axicabtagene ciloleucel. He experienced grade 2 cytokine release syndrome and grade 3 neurologic toxicity, both of which resolved with treatment. Imaging showed complete remission at approximately 3 months that was sustained at 1 year. Additionally, he had an undetectable HIV viral load and was psychiatrically stable.
The second patient was a man with AIDS-associated, non–germinal center B-cell, Epstein-Barr virus–positive DLBCL who was adherent to antiretroviral therapy. His lymphoma had recurred rapidly after initially responding to dose-adjusted EPOCH-R and then was refractory to combination rituximab and lenalidomide. He previously had hepatitis B virus, cytomegalovirus, and Mycobacterium avium complex infections.
Because of prolonged cytopenias and infectious complications after the previous lymphoma treatments, the patient was considered a poor candidate for high-dose chemotherapy. He underwent CAR T-cell therapy with axicabtagene ciloleucel and had a complete remission on day 28. Additionally, his HIV infection remained well controlled.
“Although much remains to be learned regarding CAR T-cell therapy in patients with refractory hematologic malignancies, with or without HIV infection, the cases presented herein demonstrate that patients with chemotherapy-refractory, high-grade B-cell lymphoma can successfully undergo autologous CAR T-cell manufacturing, and subsequently can safely tolerate CAR T-cell therapy and achieve a durable complete remission,” the researchers wrote. “These cases have further demonstrated the proactive, multidisciplinary care required to navigate a patient with high-risk lymphoma through CAR T-cell therapy with attention to significant medical and psychiatric comorbidities.”
Dr. Abramson reported that he has acted as a paid member of the scientific advisory board and as a paid consultant for Kite Pharma, which markets Yescarta, and several other companies.
SOURCE: Abramson JS et al. Cancer. 2019 Sep 10. doi: 10.1002/cncr.32411.
FROM CANCER
Combo could be new standard for transplant-eligible, newly diagnosed myeloma patients
BOSTON — Daratumumab plus bortezomib, lenalidomide, and dexamethasone (D-RVd) may be a new standard of care for transplant-eligible patients with newly diagnosed multiple myeloma, according to a speaker at the International Myeloma Workshop.
In the phase 2 GRIFFIN trial, adding daratumumab to RVd deepened responses at all time points and improved rates of stringent complete response and minimal residual disease (MRD) negativity post consolidation.
These results might convince “early adopters of therapy” to change their practice, said Peter M. Voorhees, MD, of Levine Cancer Institute at Atrium Health in Charlotte, N.C., who presented results from GRIFFIN as a late-breaking abstract at the workshop, which is held by the International Myeloma Society.
“But I think you do have to be careful,” Dr. Voorhees added. “We’ll have to see how these patients do over time. Is the MRD sustained, and does that MRD negativity improve progression-free survival?”
Dr. Voorhees presented data on 207 adults with transplant-eligible, newly diagnosed multiple myeloma who were enrolled in the GRIFFIN trial. The patients received RVd, with or without daratumumab, for induction (cycles 1-4). They received granulocyte colony stimulating factor, with or without plerixafor, for stem cell mobilization, and melphalan for conditioning prior to transplant.
Patients received consolidation with D-RVd or RVd (cycles 5-6) and maintenance with lenalidomide alone or in combination with daratumumab (cycles 7-32). Patients could continue maintenance with lenalidomide alone beyond cycle 32.
The D-RVd arm comprised 104 patients, and the RVd arm comprised 103 patients. Baseline characteristics were well balanced between the treatment arms. The median age was 59 years (range, 29-70 years) in the D-RVd arm and 61 years (range, 40-70 years) in the RVd arm.
Most patients had stage I (47% in the D-RVd arm and 49% in the RVd arm) or stage II disease (39% and 36%, respectively) according to the International Staging System. And most patients had standard risk cytogenetics (84% and 86%, respectively).
Response, MRD, and engraftment
The study’s primary endpoint was stringent complete response by the end of consolidation, which was achieved by 42.4% of patients in the D-RVd arm and 32.0% in the RVd arm (odds ratio [OR] = 1.57; P = .068). The overall response rate at that time point was 99.0% and 91.8%, respectively (P = .0160).
Responses deepened over time, and response rates were greater for D-RVd than for RVd at all time points. The complete response rate was 19.2% in the D-RVd arm and 13.4% in the RVd arm at the end of induction; 27.3% and 19.6%, respectively, at the end of transplant; 51.5% and 42.3%, respectively, at the end of consolidation; and 62.6% and 47.4%, respectively, at the clinical cutoff.
D-RVd also improved MRD negativity (10-5) rates at the end of consolidation. MRD negativity was 44.2% in the D-RVd arm and 14.6% in the RVd arm (OR = 4.70; P less than .0001). The rate of MRD negativity in patients with a complete response or better was 28.8% and 9.7%, respectively (OR = 3.73; P = .0007).
Dr. Voorhees noted that D-RVd was favored across all subgroups for MRD negativity and stringent compete response, except among patients with high-risk cytogenetics and stage III disease.
He also pointed out that stem cell mobilization was “feasible” in the D-RVd arm, and daratumumab did not impact engraftment. The median time to neutrophil engraftment was 12 days in both treatment arms. The median time to platelet engraftment was 13 days in the D-RVd arm and 12 days in the RVd arm.
Safety
“The adverse events are what you would expect,” Dr. Voorhees said. “Grade 3 and 4 neutropenia and thrombocytopenia were seen more often in the dara arm of the trial compared to the RVd arm.”
The most common grade 3/4 treatment-emergent adverse events (in the D-RVd and RVd arms, respectively) were neutropenia (32% and 15%), lymphopenia (23% in both), thrombocytopenia (16% and 8%), and leukopenia (15% and 7%).
“Nonhematologic toxicities were generally equal between the two groups, but I do want to stress that there was a higher rate of infection in the dara arm,” Dr. Voorhees noted.
The incidence of infection was 82% in the D-RVd arm and 55% in the RVd arm, but the rate of grade 3/4 infection was 17% in both arms. The rate of pneumonia was 10% in the D-RVd arm and 9% in the RVd arm.
All-grade infusion-related reactions occurred in 41% of patients in the D-RVd arm, and grade 3/4 infusion-related reactions occurred in 5%.
The trial was sponsored by Janssen. Dr. Voorhees reported relationships with Janssen and several other companies.
SOURCE: Voorhees PM et al. IMW 2019. Abstract OAB-087.
BOSTON — Daratumumab plus bortezomib, lenalidomide, and dexamethasone (D-RVd) may be a new standard of care for transplant-eligible patients with newly diagnosed multiple myeloma, according to a speaker at the International Myeloma Workshop.
In the phase 2 GRIFFIN trial, adding daratumumab to RVd deepened responses at all time points and improved rates of stringent complete response and minimal residual disease (MRD) negativity post consolidation.
These results might convince “early adopters of therapy” to change their practice, said Peter M. Voorhees, MD, of Levine Cancer Institute at Atrium Health in Charlotte, N.C., who presented results from GRIFFIN as a late-breaking abstract at the workshop, which is held by the International Myeloma Society.
“But I think you do have to be careful,” Dr. Voorhees added. “We’ll have to see how these patients do over time. Is the MRD sustained, and does that MRD negativity improve progression-free survival?”
Dr. Voorhees presented data on 207 adults with transplant-eligible, newly diagnosed multiple myeloma who were enrolled in the GRIFFIN trial. The patients received RVd, with or without daratumumab, for induction (cycles 1-4). They received granulocyte colony stimulating factor, with or without plerixafor, for stem cell mobilization, and melphalan for conditioning prior to transplant.
Patients received consolidation with D-RVd or RVd (cycles 5-6) and maintenance with lenalidomide alone or in combination with daratumumab (cycles 7-32). Patients could continue maintenance with lenalidomide alone beyond cycle 32.
The D-RVd arm comprised 104 patients, and the RVd arm comprised 103 patients. Baseline characteristics were well balanced between the treatment arms. The median age was 59 years (range, 29-70 years) in the D-RVd arm and 61 years (range, 40-70 years) in the RVd arm.
Most patients had stage I (47% in the D-RVd arm and 49% in the RVd arm) or stage II disease (39% and 36%, respectively) according to the International Staging System. And most patients had standard risk cytogenetics (84% and 86%, respectively).
Response, MRD, and engraftment
The study’s primary endpoint was stringent complete response by the end of consolidation, which was achieved by 42.4% of patients in the D-RVd arm and 32.0% in the RVd arm (odds ratio [OR] = 1.57; P = .068). The overall response rate at that time point was 99.0% and 91.8%, respectively (P = .0160).
Responses deepened over time, and response rates were greater for D-RVd than for RVd at all time points. The complete response rate was 19.2% in the D-RVd arm and 13.4% in the RVd arm at the end of induction; 27.3% and 19.6%, respectively, at the end of transplant; 51.5% and 42.3%, respectively, at the end of consolidation; and 62.6% and 47.4%, respectively, at the clinical cutoff.
D-RVd also improved MRD negativity (10-5) rates at the end of consolidation. MRD negativity was 44.2% in the D-RVd arm and 14.6% in the RVd arm (OR = 4.70; P less than .0001). The rate of MRD negativity in patients with a complete response or better was 28.8% and 9.7%, respectively (OR = 3.73; P = .0007).
Dr. Voorhees noted that D-RVd was favored across all subgroups for MRD negativity and stringent compete response, except among patients with high-risk cytogenetics and stage III disease.
He also pointed out that stem cell mobilization was “feasible” in the D-RVd arm, and daratumumab did not impact engraftment. The median time to neutrophil engraftment was 12 days in both treatment arms. The median time to platelet engraftment was 13 days in the D-RVd arm and 12 days in the RVd arm.
Safety
“The adverse events are what you would expect,” Dr. Voorhees said. “Grade 3 and 4 neutropenia and thrombocytopenia were seen more often in the dara arm of the trial compared to the RVd arm.”
The most common grade 3/4 treatment-emergent adverse events (in the D-RVd and RVd arms, respectively) were neutropenia (32% and 15%), lymphopenia (23% in both), thrombocytopenia (16% and 8%), and leukopenia (15% and 7%).
“Nonhematologic toxicities were generally equal between the two groups, but I do want to stress that there was a higher rate of infection in the dara arm,” Dr. Voorhees noted.
The incidence of infection was 82% in the D-RVd arm and 55% in the RVd arm, but the rate of grade 3/4 infection was 17% in both arms. The rate of pneumonia was 10% in the D-RVd arm and 9% in the RVd arm.
All-grade infusion-related reactions occurred in 41% of patients in the D-RVd arm, and grade 3/4 infusion-related reactions occurred in 5%.
The trial was sponsored by Janssen. Dr. Voorhees reported relationships with Janssen and several other companies.
SOURCE: Voorhees PM et al. IMW 2019. Abstract OAB-087.
BOSTON — Daratumumab plus bortezomib, lenalidomide, and dexamethasone (D-RVd) may be a new standard of care for transplant-eligible patients with newly diagnosed multiple myeloma, according to a speaker at the International Myeloma Workshop.
In the phase 2 GRIFFIN trial, adding daratumumab to RVd deepened responses at all time points and improved rates of stringent complete response and minimal residual disease (MRD) negativity post consolidation.
These results might convince “early adopters of therapy” to change their practice, said Peter M. Voorhees, MD, of Levine Cancer Institute at Atrium Health in Charlotte, N.C., who presented results from GRIFFIN as a late-breaking abstract at the workshop, which is held by the International Myeloma Society.
“But I think you do have to be careful,” Dr. Voorhees added. “We’ll have to see how these patients do over time. Is the MRD sustained, and does that MRD negativity improve progression-free survival?”
Dr. Voorhees presented data on 207 adults with transplant-eligible, newly diagnosed multiple myeloma who were enrolled in the GRIFFIN trial. The patients received RVd, with or without daratumumab, for induction (cycles 1-4). They received granulocyte colony stimulating factor, with or without plerixafor, for stem cell mobilization, and melphalan for conditioning prior to transplant.
Patients received consolidation with D-RVd or RVd (cycles 5-6) and maintenance with lenalidomide alone or in combination with daratumumab (cycles 7-32). Patients could continue maintenance with lenalidomide alone beyond cycle 32.
The D-RVd arm comprised 104 patients, and the RVd arm comprised 103 patients. Baseline characteristics were well balanced between the treatment arms. The median age was 59 years (range, 29-70 years) in the D-RVd arm and 61 years (range, 40-70 years) in the RVd arm.
Most patients had stage I (47% in the D-RVd arm and 49% in the RVd arm) or stage II disease (39% and 36%, respectively) according to the International Staging System. And most patients had standard risk cytogenetics (84% and 86%, respectively).
Response, MRD, and engraftment
The study’s primary endpoint was stringent complete response by the end of consolidation, which was achieved by 42.4% of patients in the D-RVd arm and 32.0% in the RVd arm (odds ratio [OR] = 1.57; P = .068). The overall response rate at that time point was 99.0% and 91.8%, respectively (P = .0160).
Responses deepened over time, and response rates were greater for D-RVd than for RVd at all time points. The complete response rate was 19.2% in the D-RVd arm and 13.4% in the RVd arm at the end of induction; 27.3% and 19.6%, respectively, at the end of transplant; 51.5% and 42.3%, respectively, at the end of consolidation; and 62.6% and 47.4%, respectively, at the clinical cutoff.
D-RVd also improved MRD negativity (10-5) rates at the end of consolidation. MRD negativity was 44.2% in the D-RVd arm and 14.6% in the RVd arm (OR = 4.70; P less than .0001). The rate of MRD negativity in patients with a complete response or better was 28.8% and 9.7%, respectively (OR = 3.73; P = .0007).
Dr. Voorhees noted that D-RVd was favored across all subgroups for MRD negativity and stringent compete response, except among patients with high-risk cytogenetics and stage III disease.
He also pointed out that stem cell mobilization was “feasible” in the D-RVd arm, and daratumumab did not impact engraftment. The median time to neutrophil engraftment was 12 days in both treatment arms. The median time to platelet engraftment was 13 days in the D-RVd arm and 12 days in the RVd arm.
Safety
“The adverse events are what you would expect,” Dr. Voorhees said. “Grade 3 and 4 neutropenia and thrombocytopenia were seen more often in the dara arm of the trial compared to the RVd arm.”
The most common grade 3/4 treatment-emergent adverse events (in the D-RVd and RVd arms, respectively) were neutropenia (32% and 15%), lymphopenia (23% in both), thrombocytopenia (16% and 8%), and leukopenia (15% and 7%).
“Nonhematologic toxicities were generally equal between the two groups, but I do want to stress that there was a higher rate of infection in the dara arm,” Dr. Voorhees noted.
The incidence of infection was 82% in the D-RVd arm and 55% in the RVd arm, but the rate of grade 3/4 infection was 17% in both arms. The rate of pneumonia was 10% in the D-RVd arm and 9% in the RVd arm.
All-grade infusion-related reactions occurred in 41% of patients in the D-RVd arm, and grade 3/4 infusion-related reactions occurred in 5%.
The trial was sponsored by Janssen. Dr. Voorhees reported relationships with Janssen and several other companies.
SOURCE: Voorhees PM et al. IMW 2019. Abstract OAB-087.
FROM IMW 2019
Adding elotuzumab to lenalidomide/dexamethasone can prolong survival in relapsed/refractory myeloma
BOSTON – Adding elotuzumab to lenalidomide and dexamethasone can prolong overall survival in patients with relapsed/refractory multiple myeloma, according to final results from the ELOQUENT-2 trial.
At a minimum follow-up of 6 years, elotuzumab plus lenalidomide/dexamethasone (ELd) reduced the risk of death by 18% and prolonged the median overall survival by 8.7 months when compared to treatment with lenalidomide/dexamethasone (Ld).
“The combination of elotuzumab with lenalidomide and dexamethasone demonstrated a statistically significant and clinically meaningful 18% reduction in the risk of death,” said Meletios A. Dimopoulos, MD, PhD, of the National and Kapodistrian University of Athens. “This treatment combination is the only approved antibody-based regimen shown to prolong overall survival significantly in patients with relapsed or refractory myeloma in the context of a large, prospective, randomized trial.”
Dr. Dimopoulos presented results from this phase 3 trial at the International Myeloma Workshop, held by the International Myeloma Society.
The final analysis of ELOQUENT-2 included 646 patients with relapsed/refractory multiple myeloma who had received one to three prior lines of therapy at baseline. There were 321 patients randomized to ELd and 325 randomized to Ld. Baseline characteristics were well balanced between the treatment arms.
At the data cutoff of Oct. 3, 2018, 319 patients in the ELd arm and 316 in the Ld arm had received their assigned treatment. Ten percent (n = 33) of patients in the ELd arm and 4% (n = 14) in the Ld arm were still receiving their assigned treatment at the cutoff date.
The median number of treatment cycles was 19 (range, 9-42) in the ELd arm and 14 (range, 6-25) in the Ld arm. Most patients discontinued treatment due to disease progression (56% in the ELd arm and 57% in the Ld arm) or treatment-related toxicity (12% and 14%, respectively).
Survival
At a minimum follow-up of 71 months, the median overall survival was 48.3 months in the ELd arm and 39.6 months in the Ld arm. The hazard ratio was 0.82 (P = .0408).
The overall survival advantage with ELd was observed in all prespecified patient subgroups, Dr. Dimopoulos said. For example, overall survival favored ELd in patients aged 75 years and older (HR, 0.69), patients with International Staging System stage III disease at enrollment (HR, 0.74), those who had received two to three prior lines of therapy (HR, 0.71), and patients with del(17p) (HR, 0.71).
There were 212 deaths in the ELd arm and 225 in the Ld arm (67% and 71%, respectively). The most common causes of death were disease progression (41% in the ELd arm and 45% in the Ld arm), infection (9% and 6%, respectively), and “other” or unknown causes (7% and 9%, respectively). Two percent of patients in each arm (n = 7 in both) died from study treatment–related toxicity.
Dr. Dimopoulos pointed out that there were no imbalances in subsequent therapies between the ELd and Ld arms. The most common subsequent therapies (in the ELd and Ld arms, respectively) were bortezomib (38% and 42%), cyclophosphamide (30% in both arms), pomalidomide (26% and 29%), and lenalidomide (18% and 22%).
Safety
“Most of the adverse events which occurred throughout the study were due to the known side effects of lenalidomide and dexamethasone,” Dr. Dimopoulos said. “The contribution of elotuzumab to any kind of toxicity was minimal.”
Nearly all patients in both arms (99%) experienced adverse events. Serious adverse events were observed in 75% of patients in the ELd arm and 61% in the Ld arm. Adverse events leading to treatment discontinuation occurred in 36% and 33%, respectively, and grade 3-4 events leading to discontinuation occurred in 21% and 20%, respectively.
Grade 3-4 adverse events of special interest (in the ELd and Ld arms, respectively) were infections (35% and 27%), renal and urinary disorders (5% in both), cardiac disorders (6% and 8%), and lymphopenia (8% and 4%). Second primary malignancies occurred in 12% of patients in the ELd arm and 9% in the Ld arm.
This trial was sponsored by Bristol-Myers Squibb in collaboration with AbbVie. Dr. Dimopoulos reported relationships with Bristol-Myers Squibb, Amgen, Celgene, Janssen, and Takeda.
SOURCE: Dimopoulos MA et al. IMW 2019, Abstract OAB-021.
BOSTON – Adding elotuzumab to lenalidomide and dexamethasone can prolong overall survival in patients with relapsed/refractory multiple myeloma, according to final results from the ELOQUENT-2 trial.
At a minimum follow-up of 6 years, elotuzumab plus lenalidomide/dexamethasone (ELd) reduced the risk of death by 18% and prolonged the median overall survival by 8.7 months when compared to treatment with lenalidomide/dexamethasone (Ld).
“The combination of elotuzumab with lenalidomide and dexamethasone demonstrated a statistically significant and clinically meaningful 18% reduction in the risk of death,” said Meletios A. Dimopoulos, MD, PhD, of the National and Kapodistrian University of Athens. “This treatment combination is the only approved antibody-based regimen shown to prolong overall survival significantly in patients with relapsed or refractory myeloma in the context of a large, prospective, randomized trial.”
Dr. Dimopoulos presented results from this phase 3 trial at the International Myeloma Workshop, held by the International Myeloma Society.
The final analysis of ELOQUENT-2 included 646 patients with relapsed/refractory multiple myeloma who had received one to three prior lines of therapy at baseline. There were 321 patients randomized to ELd and 325 randomized to Ld. Baseline characteristics were well balanced between the treatment arms.
At the data cutoff of Oct. 3, 2018, 319 patients in the ELd arm and 316 in the Ld arm had received their assigned treatment. Ten percent (n = 33) of patients in the ELd arm and 4% (n = 14) in the Ld arm were still receiving their assigned treatment at the cutoff date.
The median number of treatment cycles was 19 (range, 9-42) in the ELd arm and 14 (range, 6-25) in the Ld arm. Most patients discontinued treatment due to disease progression (56% in the ELd arm and 57% in the Ld arm) or treatment-related toxicity (12% and 14%, respectively).
Survival
At a minimum follow-up of 71 months, the median overall survival was 48.3 months in the ELd arm and 39.6 months in the Ld arm. The hazard ratio was 0.82 (P = .0408).
The overall survival advantage with ELd was observed in all prespecified patient subgroups, Dr. Dimopoulos said. For example, overall survival favored ELd in patients aged 75 years and older (HR, 0.69), patients with International Staging System stage III disease at enrollment (HR, 0.74), those who had received two to three prior lines of therapy (HR, 0.71), and patients with del(17p) (HR, 0.71).
There were 212 deaths in the ELd arm and 225 in the Ld arm (67% and 71%, respectively). The most common causes of death were disease progression (41% in the ELd arm and 45% in the Ld arm), infection (9% and 6%, respectively), and “other” or unknown causes (7% and 9%, respectively). Two percent of patients in each arm (n = 7 in both) died from study treatment–related toxicity.
Dr. Dimopoulos pointed out that there were no imbalances in subsequent therapies between the ELd and Ld arms. The most common subsequent therapies (in the ELd and Ld arms, respectively) were bortezomib (38% and 42%), cyclophosphamide (30% in both arms), pomalidomide (26% and 29%), and lenalidomide (18% and 22%).
Safety
“Most of the adverse events which occurred throughout the study were due to the known side effects of lenalidomide and dexamethasone,” Dr. Dimopoulos said. “The contribution of elotuzumab to any kind of toxicity was minimal.”
Nearly all patients in both arms (99%) experienced adverse events. Serious adverse events were observed in 75% of patients in the ELd arm and 61% in the Ld arm. Adverse events leading to treatment discontinuation occurred in 36% and 33%, respectively, and grade 3-4 events leading to discontinuation occurred in 21% and 20%, respectively.
Grade 3-4 adverse events of special interest (in the ELd and Ld arms, respectively) were infections (35% and 27%), renal and urinary disorders (5% in both), cardiac disorders (6% and 8%), and lymphopenia (8% and 4%). Second primary malignancies occurred in 12% of patients in the ELd arm and 9% in the Ld arm.
This trial was sponsored by Bristol-Myers Squibb in collaboration with AbbVie. Dr. Dimopoulos reported relationships with Bristol-Myers Squibb, Amgen, Celgene, Janssen, and Takeda.
SOURCE: Dimopoulos MA et al. IMW 2019, Abstract OAB-021.
BOSTON – Adding elotuzumab to lenalidomide and dexamethasone can prolong overall survival in patients with relapsed/refractory multiple myeloma, according to final results from the ELOQUENT-2 trial.
At a minimum follow-up of 6 years, elotuzumab plus lenalidomide/dexamethasone (ELd) reduced the risk of death by 18% and prolonged the median overall survival by 8.7 months when compared to treatment with lenalidomide/dexamethasone (Ld).
“The combination of elotuzumab with lenalidomide and dexamethasone demonstrated a statistically significant and clinically meaningful 18% reduction in the risk of death,” said Meletios A. Dimopoulos, MD, PhD, of the National and Kapodistrian University of Athens. “This treatment combination is the only approved antibody-based regimen shown to prolong overall survival significantly in patients with relapsed or refractory myeloma in the context of a large, prospective, randomized trial.”
Dr. Dimopoulos presented results from this phase 3 trial at the International Myeloma Workshop, held by the International Myeloma Society.
The final analysis of ELOQUENT-2 included 646 patients with relapsed/refractory multiple myeloma who had received one to three prior lines of therapy at baseline. There were 321 patients randomized to ELd and 325 randomized to Ld. Baseline characteristics were well balanced between the treatment arms.
At the data cutoff of Oct. 3, 2018, 319 patients in the ELd arm and 316 in the Ld arm had received their assigned treatment. Ten percent (n = 33) of patients in the ELd arm and 4% (n = 14) in the Ld arm were still receiving their assigned treatment at the cutoff date.
The median number of treatment cycles was 19 (range, 9-42) in the ELd arm and 14 (range, 6-25) in the Ld arm. Most patients discontinued treatment due to disease progression (56% in the ELd arm and 57% in the Ld arm) or treatment-related toxicity (12% and 14%, respectively).
Survival
At a minimum follow-up of 71 months, the median overall survival was 48.3 months in the ELd arm and 39.6 months in the Ld arm. The hazard ratio was 0.82 (P = .0408).
The overall survival advantage with ELd was observed in all prespecified patient subgroups, Dr. Dimopoulos said. For example, overall survival favored ELd in patients aged 75 years and older (HR, 0.69), patients with International Staging System stage III disease at enrollment (HR, 0.74), those who had received two to three prior lines of therapy (HR, 0.71), and patients with del(17p) (HR, 0.71).
There were 212 deaths in the ELd arm and 225 in the Ld arm (67% and 71%, respectively). The most common causes of death were disease progression (41% in the ELd arm and 45% in the Ld arm), infection (9% and 6%, respectively), and “other” or unknown causes (7% and 9%, respectively). Two percent of patients in each arm (n = 7 in both) died from study treatment–related toxicity.
Dr. Dimopoulos pointed out that there were no imbalances in subsequent therapies between the ELd and Ld arms. The most common subsequent therapies (in the ELd and Ld arms, respectively) were bortezomib (38% and 42%), cyclophosphamide (30% in both arms), pomalidomide (26% and 29%), and lenalidomide (18% and 22%).
Safety
“Most of the adverse events which occurred throughout the study were due to the known side effects of lenalidomide and dexamethasone,” Dr. Dimopoulos said. “The contribution of elotuzumab to any kind of toxicity was minimal.”
Nearly all patients in both arms (99%) experienced adverse events. Serious adverse events were observed in 75% of patients in the ELd arm and 61% in the Ld arm. Adverse events leading to treatment discontinuation occurred in 36% and 33%, respectively, and grade 3-4 events leading to discontinuation occurred in 21% and 20%, respectively.
Grade 3-4 adverse events of special interest (in the ELd and Ld arms, respectively) were infections (35% and 27%), renal and urinary disorders (5% in both), cardiac disorders (6% and 8%), and lymphopenia (8% and 4%). Second primary malignancies occurred in 12% of patients in the ELd arm and 9% in the Ld arm.
This trial was sponsored by Bristol-Myers Squibb in collaboration with AbbVie. Dr. Dimopoulos reported relationships with Bristol-Myers Squibb, Amgen, Celgene, Janssen, and Takeda.
SOURCE: Dimopoulos MA et al. IMW 2019, Abstract OAB-021.
FROM IMW 2019
CT103A elicits responses after prior CAR T-cell relapse
BOSTON – CT103A, a chimeric antigen receptor (CAR) T-cell therapy, is “active and effective” in patients with relapsed/refractory multiple myeloma, according to a speaker at the International Myeloma Workshop, held by the International Myeloma Society.
The anti–B-cell maturation antigen (BCMA) CAR T-cell therapy produced a 100% response rate in patients with heavily pretreated multiple myeloma, and three of four patients who had failed a prior CAR T-cell therapy achieved a stringent complete response after CT103A.
Chunrui Li, MD, PhD, of Tongji Hospital and Tongji Medical College, Huazhong University of Science, Wuhan, China, presented these results at the workshop.
Dr. Li noted that anti-BCMA CAR T-cell therapy has produced responses in myeloma patients, but approximately half of patients typically relapse in about a year. CAR T-cell infusions after relapse have not been effective in these patients.
In an effort to change that, Dr. Li and his colleagues developed CT103A, a lentiviral vector containing a CAR structure with a fully human single-chain fragment variant; CD8a hinger; and transmembrane, 4-1BB co-stimulatory, and CD3z activation domains.
Dr. Li and his colleagues evaluated CT103A in a phase 0 trial (ChiCTR1800018137) of 18 patients who had received at least three prior lines of therapy and had disease refractory to a proteasome inhibitor and an immunomodulatory agent.
The patients’ median age was 53.3 years (range, 38-66 years), and their median time since diagnosis was 32 months (range, 8-92 months). They had received a median of 4 (range, 3-6) prior therapies. All had received prior bortezomib and lenalidomide, seven had undergone a transplant, and four had been treated on a trial of murine anti-BCMA CAR T-cell therapy.
For the current trial, patients received lymphodepletion with cyclophosphamide and fludarabine, followed by CT103A at 1x106, 3x106, or 6x106 CAR T cells/kg.
There was one dose-limiting toxicity at the highest dose level – grade 4 cytokine release syndrome (CRS) in a patient who died at day 19 after CT103A infusion.
In all, 17 patients developed CRS, four with grade 1, eight with grade 2, four with grade 3, and one with grade 4 CRS. None of the patients developed neurologic toxicity.
Serious adverse events related to lymphodepletion and/or CT103A included prolonged cytopenia (n = 3), pulmonary infection (n = 2), herpes zoster (n = 1), pleuritis (n = 1), and hypoxemia (n = 1).
There were 17 patients evaluable for efficacy, and all of them achieved a response at some point. In eight patients, responses have lasted more than 200 days.
At the data cutoff, there were 10 stringent complete responses, two complete responses, and three very good partial responses. One patient progressed after achieving a very good partial response, and one patient achieved a partial response but ultimately died (likely of respiratory failure attributable to a lung infection).
Of the four patients who had previously received murine CAR T-cell therapy, one progressed, and three achieved a stringent complete response.
This study was funded by Nanjing Iaso Biotherapeutics. Dr. Li did not disclose any conflicts of interest.
SOURCE: Li C et al. IMW 2019, Abstract OAB-033.
BOSTON – CT103A, a chimeric antigen receptor (CAR) T-cell therapy, is “active and effective” in patients with relapsed/refractory multiple myeloma, according to a speaker at the International Myeloma Workshop, held by the International Myeloma Society.
The anti–B-cell maturation antigen (BCMA) CAR T-cell therapy produced a 100% response rate in patients with heavily pretreated multiple myeloma, and three of four patients who had failed a prior CAR T-cell therapy achieved a stringent complete response after CT103A.
Chunrui Li, MD, PhD, of Tongji Hospital and Tongji Medical College, Huazhong University of Science, Wuhan, China, presented these results at the workshop.
Dr. Li noted that anti-BCMA CAR T-cell therapy has produced responses in myeloma patients, but approximately half of patients typically relapse in about a year. CAR T-cell infusions after relapse have not been effective in these patients.
In an effort to change that, Dr. Li and his colleagues developed CT103A, a lentiviral vector containing a CAR structure with a fully human single-chain fragment variant; CD8a hinger; and transmembrane, 4-1BB co-stimulatory, and CD3z activation domains.
Dr. Li and his colleagues evaluated CT103A in a phase 0 trial (ChiCTR1800018137) of 18 patients who had received at least three prior lines of therapy and had disease refractory to a proteasome inhibitor and an immunomodulatory agent.
The patients’ median age was 53.3 years (range, 38-66 years), and their median time since diagnosis was 32 months (range, 8-92 months). They had received a median of 4 (range, 3-6) prior therapies. All had received prior bortezomib and lenalidomide, seven had undergone a transplant, and four had been treated on a trial of murine anti-BCMA CAR T-cell therapy.
For the current trial, patients received lymphodepletion with cyclophosphamide and fludarabine, followed by CT103A at 1x106, 3x106, or 6x106 CAR T cells/kg.
There was one dose-limiting toxicity at the highest dose level – grade 4 cytokine release syndrome (CRS) in a patient who died at day 19 after CT103A infusion.
In all, 17 patients developed CRS, four with grade 1, eight with grade 2, four with grade 3, and one with grade 4 CRS. None of the patients developed neurologic toxicity.
Serious adverse events related to lymphodepletion and/or CT103A included prolonged cytopenia (n = 3), pulmonary infection (n = 2), herpes zoster (n = 1), pleuritis (n = 1), and hypoxemia (n = 1).
There were 17 patients evaluable for efficacy, and all of them achieved a response at some point. In eight patients, responses have lasted more than 200 days.
At the data cutoff, there were 10 stringent complete responses, two complete responses, and three very good partial responses. One patient progressed after achieving a very good partial response, and one patient achieved a partial response but ultimately died (likely of respiratory failure attributable to a lung infection).
Of the four patients who had previously received murine CAR T-cell therapy, one progressed, and three achieved a stringent complete response.
This study was funded by Nanjing Iaso Biotherapeutics. Dr. Li did not disclose any conflicts of interest.
SOURCE: Li C et al. IMW 2019, Abstract OAB-033.
BOSTON – CT103A, a chimeric antigen receptor (CAR) T-cell therapy, is “active and effective” in patients with relapsed/refractory multiple myeloma, according to a speaker at the International Myeloma Workshop, held by the International Myeloma Society.
The anti–B-cell maturation antigen (BCMA) CAR T-cell therapy produced a 100% response rate in patients with heavily pretreated multiple myeloma, and three of four patients who had failed a prior CAR T-cell therapy achieved a stringent complete response after CT103A.
Chunrui Li, MD, PhD, of Tongji Hospital and Tongji Medical College, Huazhong University of Science, Wuhan, China, presented these results at the workshop.
Dr. Li noted that anti-BCMA CAR T-cell therapy has produced responses in myeloma patients, but approximately half of patients typically relapse in about a year. CAR T-cell infusions after relapse have not been effective in these patients.
In an effort to change that, Dr. Li and his colleagues developed CT103A, a lentiviral vector containing a CAR structure with a fully human single-chain fragment variant; CD8a hinger; and transmembrane, 4-1BB co-stimulatory, and CD3z activation domains.
Dr. Li and his colleagues evaluated CT103A in a phase 0 trial (ChiCTR1800018137) of 18 patients who had received at least three prior lines of therapy and had disease refractory to a proteasome inhibitor and an immunomodulatory agent.
The patients’ median age was 53.3 years (range, 38-66 years), and their median time since diagnosis was 32 months (range, 8-92 months). They had received a median of 4 (range, 3-6) prior therapies. All had received prior bortezomib and lenalidomide, seven had undergone a transplant, and four had been treated on a trial of murine anti-BCMA CAR T-cell therapy.
For the current trial, patients received lymphodepletion with cyclophosphamide and fludarabine, followed by CT103A at 1x106, 3x106, or 6x106 CAR T cells/kg.
There was one dose-limiting toxicity at the highest dose level – grade 4 cytokine release syndrome (CRS) in a patient who died at day 19 after CT103A infusion.
In all, 17 patients developed CRS, four with grade 1, eight with grade 2, four with grade 3, and one with grade 4 CRS. None of the patients developed neurologic toxicity.
Serious adverse events related to lymphodepletion and/or CT103A included prolonged cytopenia (n = 3), pulmonary infection (n = 2), herpes zoster (n = 1), pleuritis (n = 1), and hypoxemia (n = 1).
There were 17 patients evaluable for efficacy, and all of them achieved a response at some point. In eight patients, responses have lasted more than 200 days.
At the data cutoff, there were 10 stringent complete responses, two complete responses, and three very good partial responses. One patient progressed after achieving a very good partial response, and one patient achieved a partial response but ultimately died (likely of respiratory failure attributable to a lung infection).
Of the four patients who had previously received murine CAR T-cell therapy, one progressed, and three achieved a stringent complete response.
This study was funded by Nanjing Iaso Biotherapeutics. Dr. Li did not disclose any conflicts of interest.
SOURCE: Li C et al. IMW 2019, Abstract OAB-033.
REPORTING FROM IMW 2019
Worse PFS when radiation is withheld in early-stage Hodgkin lymphoma
Radiotherapy appears to be an essential component of the optimal treatment regimen for adults with early-stage favorable Hodgkin lymphoma, investigators in a randomized phase 3 trial asserted.
Among more than 600 patients with early-stage Hodgkin lymphoma who were positron-emission tomography (PET)–negative after two cycles of standard chemotherapy, 5-year progression-free survival was significantly better for patients who had also received involved-field radiotherapy, compared with those who had received chemotherapy alone.
The HD16 trial was designed to show whether using PET findings to opt for consolidation radiotherapy could be noninferior to the use of combined modality therapy (CMT) with ABVD (doxorubicin, bleomycin, vinblastine and dacarbazine) and radiation for all patients with early-stage favorable-risk disease.
“However, we failed to meet the primary objective of the trial as PET-guided omission of radiotherapy results in poorer tumor control compared with CMT. We therefore recommend proceeding with consolidation radiotherapy as a standard of care for patients achieving a metabolic response after two cycles of ABVD,” Michael Fuchs, MD, from the University Hospital of Cologne, Germany, and colleagues in the German Hodgkin Study Group wrote in the Journal of Clinical Oncology.
The investigators also found that patients who remained PET-positive after two cycles of ABVD were at high risk for treatment failure, particularly when a Deauville score of 4 was used as the minimum threshold for positivity.
Although CMT is associated with high cure rates for patients with Hodgkin lymphoma, clinicians are concerned about long-term toxicities and risk for second malignancies, which prompted investigators to see whether radiotherapy could be safely eliminated in some cases.
The HD16 investigators enrolled 1,150 patients aged 18-75 years with early-stage favorable Hodgkin lymphoma, and randomly assigned them to receive two cycles of ABVD with either 20 Gy of involved-field radiotherapy or PET-guided treatment in which involved-field radiation was eliminated for those patients who were PET-negative after chemotherapy, with PET negativity defined as a Deauville score less than 3.
After a median follow-up of 47 months, the 5-year progression-free survival rates among 628 PET-negative patients were 93.4% for those assigned to CMT, versus 86.1% for patients assigned to chemotherapy alone, which translated into a hazard ratio HR of 1.78 (95% confidence interval, 1.02-3.12; P = .040). The upper limit of the confidence interval exceeds the predefined noninferiority margin of 3.01, which indicates that eliminating radiation was clinically inferior.
The difference in progression-free survival rates between the treatment arms was primarily caused by a significant increase in disease recurrence in what would have been the involved field for patients in the ABVD-alone group (in-field recurrence rate, 9% vs. 2% for patients who received CMT; P = .0003). In contrast, there were no significant differences between the groups in out-of-field recurrences (5% vs. 4%, respectively).
Five-year overall survival rates were virtually identical between the treatment arms among PET-negative patients.
When the investigators compared all PET-negative with PET-positive patients (Deauville score 4), they saw that 5-year estimated progression-free survival rates were 93.1% vs. 80.9%, respectively, an absolute difference of 12.1% that translated into a HR of 2.94 (P less than .001). There were no significant differences by PET status in 5-year overall survival, however.
“We assume that the small radiation fields and doses used in our HD16 trial will induce fewer late adverse events than those reported in the literature. However, we cannot exclude an increased risk for certain late effects, such as breast cancer in very young women, as the risk for this specific second malignancy increases with younger age,” the researchers wrote.
The study was supported by grants from Deutsche Krebshilfe and the Swiss State Secretariat for Education, Research, and Innovation. Dr. Fuchs reported honoraria from Amgen, Affimed, Celgene, and Takeda. Multiple coauthors reported industry funding.
SOURCE: Fuchs M et al. J Clin Oncol. 2019 Sep 10. doi: 10.1200/JCO.19.00964.
Radiotherapy appears to be an essential component of the optimal treatment regimen for adults with early-stage favorable Hodgkin lymphoma, investigators in a randomized phase 3 trial asserted.
Among more than 600 patients with early-stage Hodgkin lymphoma who were positron-emission tomography (PET)–negative after two cycles of standard chemotherapy, 5-year progression-free survival was significantly better for patients who had also received involved-field radiotherapy, compared with those who had received chemotherapy alone.
The HD16 trial was designed to show whether using PET findings to opt for consolidation radiotherapy could be noninferior to the use of combined modality therapy (CMT) with ABVD (doxorubicin, bleomycin, vinblastine and dacarbazine) and radiation for all patients with early-stage favorable-risk disease.
“However, we failed to meet the primary objective of the trial as PET-guided omission of radiotherapy results in poorer tumor control compared with CMT. We therefore recommend proceeding with consolidation radiotherapy as a standard of care for patients achieving a metabolic response after two cycles of ABVD,” Michael Fuchs, MD, from the University Hospital of Cologne, Germany, and colleagues in the German Hodgkin Study Group wrote in the Journal of Clinical Oncology.
The investigators also found that patients who remained PET-positive after two cycles of ABVD were at high risk for treatment failure, particularly when a Deauville score of 4 was used as the minimum threshold for positivity.
Although CMT is associated with high cure rates for patients with Hodgkin lymphoma, clinicians are concerned about long-term toxicities and risk for second malignancies, which prompted investigators to see whether radiotherapy could be safely eliminated in some cases.
The HD16 investigators enrolled 1,150 patients aged 18-75 years with early-stage favorable Hodgkin lymphoma, and randomly assigned them to receive two cycles of ABVD with either 20 Gy of involved-field radiotherapy or PET-guided treatment in which involved-field radiation was eliminated for those patients who were PET-negative after chemotherapy, with PET negativity defined as a Deauville score less than 3.
After a median follow-up of 47 months, the 5-year progression-free survival rates among 628 PET-negative patients were 93.4% for those assigned to CMT, versus 86.1% for patients assigned to chemotherapy alone, which translated into a hazard ratio HR of 1.78 (95% confidence interval, 1.02-3.12; P = .040). The upper limit of the confidence interval exceeds the predefined noninferiority margin of 3.01, which indicates that eliminating radiation was clinically inferior.
The difference in progression-free survival rates between the treatment arms was primarily caused by a significant increase in disease recurrence in what would have been the involved field for patients in the ABVD-alone group (in-field recurrence rate, 9% vs. 2% for patients who received CMT; P = .0003). In contrast, there were no significant differences between the groups in out-of-field recurrences (5% vs. 4%, respectively).
Five-year overall survival rates were virtually identical between the treatment arms among PET-negative patients.
When the investigators compared all PET-negative with PET-positive patients (Deauville score 4), they saw that 5-year estimated progression-free survival rates were 93.1% vs. 80.9%, respectively, an absolute difference of 12.1% that translated into a HR of 2.94 (P less than .001). There were no significant differences by PET status in 5-year overall survival, however.
“We assume that the small radiation fields and doses used in our HD16 trial will induce fewer late adverse events than those reported in the literature. However, we cannot exclude an increased risk for certain late effects, such as breast cancer in very young women, as the risk for this specific second malignancy increases with younger age,” the researchers wrote.
The study was supported by grants from Deutsche Krebshilfe and the Swiss State Secretariat for Education, Research, and Innovation. Dr. Fuchs reported honoraria from Amgen, Affimed, Celgene, and Takeda. Multiple coauthors reported industry funding.
SOURCE: Fuchs M et al. J Clin Oncol. 2019 Sep 10. doi: 10.1200/JCO.19.00964.
Radiotherapy appears to be an essential component of the optimal treatment regimen for adults with early-stage favorable Hodgkin lymphoma, investigators in a randomized phase 3 trial asserted.
Among more than 600 patients with early-stage Hodgkin lymphoma who were positron-emission tomography (PET)–negative after two cycles of standard chemotherapy, 5-year progression-free survival was significantly better for patients who had also received involved-field radiotherapy, compared with those who had received chemotherapy alone.
The HD16 trial was designed to show whether using PET findings to opt for consolidation radiotherapy could be noninferior to the use of combined modality therapy (CMT) with ABVD (doxorubicin, bleomycin, vinblastine and dacarbazine) and radiation for all patients with early-stage favorable-risk disease.
“However, we failed to meet the primary objective of the trial as PET-guided omission of radiotherapy results in poorer tumor control compared with CMT. We therefore recommend proceeding with consolidation radiotherapy as a standard of care for patients achieving a metabolic response after two cycles of ABVD,” Michael Fuchs, MD, from the University Hospital of Cologne, Germany, and colleagues in the German Hodgkin Study Group wrote in the Journal of Clinical Oncology.
The investigators also found that patients who remained PET-positive after two cycles of ABVD were at high risk for treatment failure, particularly when a Deauville score of 4 was used as the minimum threshold for positivity.
Although CMT is associated with high cure rates for patients with Hodgkin lymphoma, clinicians are concerned about long-term toxicities and risk for second malignancies, which prompted investigators to see whether radiotherapy could be safely eliminated in some cases.
The HD16 investigators enrolled 1,150 patients aged 18-75 years with early-stage favorable Hodgkin lymphoma, and randomly assigned them to receive two cycles of ABVD with either 20 Gy of involved-field radiotherapy or PET-guided treatment in which involved-field radiation was eliminated for those patients who were PET-negative after chemotherapy, with PET negativity defined as a Deauville score less than 3.
After a median follow-up of 47 months, the 5-year progression-free survival rates among 628 PET-negative patients were 93.4% for those assigned to CMT, versus 86.1% for patients assigned to chemotherapy alone, which translated into a hazard ratio HR of 1.78 (95% confidence interval, 1.02-3.12; P = .040). The upper limit of the confidence interval exceeds the predefined noninferiority margin of 3.01, which indicates that eliminating radiation was clinically inferior.
The difference in progression-free survival rates between the treatment arms was primarily caused by a significant increase in disease recurrence in what would have been the involved field for patients in the ABVD-alone group (in-field recurrence rate, 9% vs. 2% for patients who received CMT; P = .0003). In contrast, there were no significant differences between the groups in out-of-field recurrences (5% vs. 4%, respectively).
Five-year overall survival rates were virtually identical between the treatment arms among PET-negative patients.
When the investigators compared all PET-negative with PET-positive patients (Deauville score 4), they saw that 5-year estimated progression-free survival rates were 93.1% vs. 80.9%, respectively, an absolute difference of 12.1% that translated into a HR of 2.94 (P less than .001). There were no significant differences by PET status in 5-year overall survival, however.
“We assume that the small radiation fields and doses used in our HD16 trial will induce fewer late adverse events than those reported in the literature. However, we cannot exclude an increased risk for certain late effects, such as breast cancer in very young women, as the risk for this specific second malignancy increases with younger age,” the researchers wrote.
The study was supported by grants from Deutsche Krebshilfe and the Swiss State Secretariat for Education, Research, and Innovation. Dr. Fuchs reported honoraria from Amgen, Affimed, Celgene, and Takeda. Multiple coauthors reported industry funding.
SOURCE: Fuchs M et al. J Clin Oncol. 2019 Sep 10. doi: 10.1200/JCO.19.00964.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Novel gene therapies show promise for sickle cell cure
Early results indicate experimental gene therapies could illicit a cure for sickle cell disease (SCD), but many barriers to access remain, namely cost, experts reported during a recent webinar sponsored by the National Heart, Lung, and Blood Institute.
At present, allogeneic hematopoietic stem cell transplant remains the only curative therapy available for patients with SCD. Newer transplant techniques include the use of mobilized blood stem cells, where stem cells are collected from the circulation using blood cell growth factors, explained Mark Walters, MD, of UCSF Benioff Children’s Hospital Oakland in California.
The most promising experimental gene therapies currently undergoing clinical development are gene-addition and gene-editing therapies, he said. Another technique, in vivo gene editing to correct the sickle mutation, is also being investigated, but has not yet reached clinical development.
Gene-addition therapy
Gene-addition therapy is a technique where a fetal hemoglobin (HbF) or anti-sickling beta-hemoglobin gene is inserted into a hematopoietic stem cell to illicit a curative effect. In this technique, the corrective gene is harvested from a patient’s own blood stem cells.
In patients with SCD, when HbF levels are elevated, the likelihood of sickling is reduced, resulting in a milder form of disease. As a result, raising HbF levels is a therapeutic target that forms the basis of several ongoing clinical studies.
The technique involves packaging an HbF rescue gene into a viral vector and coincubating the vector with a patient’s own blood stem cells. Subsequently, the corrected stem cells are injected back into the patient to produce higher levels of HbF.
The ongoing phase 1/2 HGB-206 clinical study is evaluating this technique in patients aged 12-50 years with severe SCD in multiple centers throughout Europe and the United States.
In those treated thus far, initial results appear promising, Dr. Walters reported, with one patient experiencing a rise in Hb levels from 10.7 g/dL at 3 months to 15.0 g/dL at 15 months follow-up.
Dr. Walters also reported that some of these patients no longer exhibit any signs or symptoms of SCD, such as anemia or painful adverse events. While these initial findings are compelling, whether these benefits will be maintained is still unknown.
“While it’s too early to call this a cure, if [these results] could be extended for 5, 10, or 15 years, I think everyone would agree that this would be a cure,” he said.
This technique could be universally available, he said, since a patient’s own blood stem cells are used. Other complications, such as graft-versus-host disease (GVHD) or immune-related reactions, are negated with this form of therapy, he said.
Recent evidence has demonstrated that only about 20% of donor stem cells need to be corrected to illicit a very strong effect. This principle is now being applied in gene-editing techniques, as correcting every gene in every stem cell would be very challenging, Dr. Walters explained.
Gene editing
Another technique being investigated in SCD is gene editing, in which the fetal hemoglobin gene is “reawakened,” or other techniques are used to correct the sickle gene directly, such as CRISPR-Cas9 technology, Dr. Walters said.
In this technique, the Cas9 protein makes a cut and repairs an individual’s genomic DNA by inserting a strand of corrected donor DNA. The novel technology would allow for targeted genome editing that is specific to the SCD patient.
Currently, this experimental therapy is being investigated in preclinical studies. Dr. Walters said that he and his colleagues hope to begin enrolling patients in clinical trials within the next 1-2 years.
But while some gene therapies have been approved in other disorders, such as spinal muscle atrophy, a limiting factor to widespread availability is cost. Despite promising initial results in SCD, the affordability of future gene therapies will be a key factor to universal access, Dr. Walters said.
The Cure Sickle Cell Initiative
Traci Mondoro, PhD, chief of the Translational Blood Science and Resources Branch at NHLBI, explained that the NHLBI has funded a large proportion of the research that has formed the basis of several genetically based clinical studies.
One of the primary goals of the Cure Sickle Cell Initiative is to bridge the gap between new research and the SCD community. Their aim is to improve access for patients to participate in genetically based studies to advance cures.
The comprehensive approach is intended to fill in existing gaps by funding breakthrough research in both academic and private settings.
By establishing partnerships with key stakeholders, institutions, and patient groups, Dr. Mondoro said they hope to increase patient participation in clinical trials involving curative therapies. In the future, they also intend to establish a large body of evidence to provide adequate safety data to study these therapies in pediatric populations.
Dr. Walters and Dr. Mondoro did not provide information on financial disclosures.
Early results indicate experimental gene therapies could illicit a cure for sickle cell disease (SCD), but many barriers to access remain, namely cost, experts reported during a recent webinar sponsored by the National Heart, Lung, and Blood Institute.
At present, allogeneic hematopoietic stem cell transplant remains the only curative therapy available for patients with SCD. Newer transplant techniques include the use of mobilized blood stem cells, where stem cells are collected from the circulation using blood cell growth factors, explained Mark Walters, MD, of UCSF Benioff Children’s Hospital Oakland in California.
The most promising experimental gene therapies currently undergoing clinical development are gene-addition and gene-editing therapies, he said. Another technique, in vivo gene editing to correct the sickle mutation, is also being investigated, but has not yet reached clinical development.
Gene-addition therapy
Gene-addition therapy is a technique where a fetal hemoglobin (HbF) or anti-sickling beta-hemoglobin gene is inserted into a hematopoietic stem cell to illicit a curative effect. In this technique, the corrective gene is harvested from a patient’s own blood stem cells.
In patients with SCD, when HbF levels are elevated, the likelihood of sickling is reduced, resulting in a milder form of disease. As a result, raising HbF levels is a therapeutic target that forms the basis of several ongoing clinical studies.
The technique involves packaging an HbF rescue gene into a viral vector and coincubating the vector with a patient’s own blood stem cells. Subsequently, the corrected stem cells are injected back into the patient to produce higher levels of HbF.
The ongoing phase 1/2 HGB-206 clinical study is evaluating this technique in patients aged 12-50 years with severe SCD in multiple centers throughout Europe and the United States.
In those treated thus far, initial results appear promising, Dr. Walters reported, with one patient experiencing a rise in Hb levels from 10.7 g/dL at 3 months to 15.0 g/dL at 15 months follow-up.
Dr. Walters also reported that some of these patients no longer exhibit any signs or symptoms of SCD, such as anemia or painful adverse events. While these initial findings are compelling, whether these benefits will be maintained is still unknown.
“While it’s too early to call this a cure, if [these results] could be extended for 5, 10, or 15 years, I think everyone would agree that this would be a cure,” he said.
This technique could be universally available, he said, since a patient’s own blood stem cells are used. Other complications, such as graft-versus-host disease (GVHD) or immune-related reactions, are negated with this form of therapy, he said.
Recent evidence has demonstrated that only about 20% of donor stem cells need to be corrected to illicit a very strong effect. This principle is now being applied in gene-editing techniques, as correcting every gene in every stem cell would be very challenging, Dr. Walters explained.
Gene editing
Another technique being investigated in SCD is gene editing, in which the fetal hemoglobin gene is “reawakened,” or other techniques are used to correct the sickle gene directly, such as CRISPR-Cas9 technology, Dr. Walters said.
In this technique, the Cas9 protein makes a cut and repairs an individual’s genomic DNA by inserting a strand of corrected donor DNA. The novel technology would allow for targeted genome editing that is specific to the SCD patient.
Currently, this experimental therapy is being investigated in preclinical studies. Dr. Walters said that he and his colleagues hope to begin enrolling patients in clinical trials within the next 1-2 years.
But while some gene therapies have been approved in other disorders, such as spinal muscle atrophy, a limiting factor to widespread availability is cost. Despite promising initial results in SCD, the affordability of future gene therapies will be a key factor to universal access, Dr. Walters said.
The Cure Sickle Cell Initiative
Traci Mondoro, PhD, chief of the Translational Blood Science and Resources Branch at NHLBI, explained that the NHLBI has funded a large proportion of the research that has formed the basis of several genetically based clinical studies.
One of the primary goals of the Cure Sickle Cell Initiative is to bridge the gap between new research and the SCD community. Their aim is to improve access for patients to participate in genetically based studies to advance cures.
The comprehensive approach is intended to fill in existing gaps by funding breakthrough research in both academic and private settings.
By establishing partnerships with key stakeholders, institutions, and patient groups, Dr. Mondoro said they hope to increase patient participation in clinical trials involving curative therapies. In the future, they also intend to establish a large body of evidence to provide adequate safety data to study these therapies in pediatric populations.
Dr. Walters and Dr. Mondoro did not provide information on financial disclosures.
Early results indicate experimental gene therapies could illicit a cure for sickle cell disease (SCD), but many barriers to access remain, namely cost, experts reported during a recent webinar sponsored by the National Heart, Lung, and Blood Institute.
At present, allogeneic hematopoietic stem cell transplant remains the only curative therapy available for patients with SCD. Newer transplant techniques include the use of mobilized blood stem cells, where stem cells are collected from the circulation using blood cell growth factors, explained Mark Walters, MD, of UCSF Benioff Children’s Hospital Oakland in California.
The most promising experimental gene therapies currently undergoing clinical development are gene-addition and gene-editing therapies, he said. Another technique, in vivo gene editing to correct the sickle mutation, is also being investigated, but has not yet reached clinical development.
Gene-addition therapy
Gene-addition therapy is a technique where a fetal hemoglobin (HbF) or anti-sickling beta-hemoglobin gene is inserted into a hematopoietic stem cell to illicit a curative effect. In this technique, the corrective gene is harvested from a patient’s own blood stem cells.
In patients with SCD, when HbF levels are elevated, the likelihood of sickling is reduced, resulting in a milder form of disease. As a result, raising HbF levels is a therapeutic target that forms the basis of several ongoing clinical studies.
The technique involves packaging an HbF rescue gene into a viral vector and coincubating the vector with a patient’s own blood stem cells. Subsequently, the corrected stem cells are injected back into the patient to produce higher levels of HbF.
The ongoing phase 1/2 HGB-206 clinical study is evaluating this technique in patients aged 12-50 years with severe SCD in multiple centers throughout Europe and the United States.
In those treated thus far, initial results appear promising, Dr. Walters reported, with one patient experiencing a rise in Hb levels from 10.7 g/dL at 3 months to 15.0 g/dL at 15 months follow-up.
Dr. Walters also reported that some of these patients no longer exhibit any signs or symptoms of SCD, such as anemia or painful adverse events. While these initial findings are compelling, whether these benefits will be maintained is still unknown.
“While it’s too early to call this a cure, if [these results] could be extended for 5, 10, or 15 years, I think everyone would agree that this would be a cure,” he said.
This technique could be universally available, he said, since a patient’s own blood stem cells are used. Other complications, such as graft-versus-host disease (GVHD) or immune-related reactions, are negated with this form of therapy, he said.
Recent evidence has demonstrated that only about 20% of donor stem cells need to be corrected to illicit a very strong effect. This principle is now being applied in gene-editing techniques, as correcting every gene in every stem cell would be very challenging, Dr. Walters explained.
Gene editing
Another technique being investigated in SCD is gene editing, in which the fetal hemoglobin gene is “reawakened,” or other techniques are used to correct the sickle gene directly, such as CRISPR-Cas9 technology, Dr. Walters said.
In this technique, the Cas9 protein makes a cut and repairs an individual’s genomic DNA by inserting a strand of corrected donor DNA. The novel technology would allow for targeted genome editing that is specific to the SCD patient.
Currently, this experimental therapy is being investigated in preclinical studies. Dr. Walters said that he and his colleagues hope to begin enrolling patients in clinical trials within the next 1-2 years.
But while some gene therapies have been approved in other disorders, such as spinal muscle atrophy, a limiting factor to widespread availability is cost. Despite promising initial results in SCD, the affordability of future gene therapies will be a key factor to universal access, Dr. Walters said.
The Cure Sickle Cell Initiative
Traci Mondoro, PhD, chief of the Translational Blood Science and Resources Branch at NHLBI, explained that the NHLBI has funded a large proportion of the research that has formed the basis of several genetically based clinical studies.
One of the primary goals of the Cure Sickle Cell Initiative is to bridge the gap between new research and the SCD community. Their aim is to improve access for patients to participate in genetically based studies to advance cures.
The comprehensive approach is intended to fill in existing gaps by funding breakthrough research in both academic and private settings.
By establishing partnerships with key stakeholders, institutions, and patient groups, Dr. Mondoro said they hope to increase patient participation in clinical trials involving curative therapies. In the future, they also intend to establish a large body of evidence to provide adequate safety data to study these therapies in pediatric populations.
Dr. Walters and Dr. Mondoro did not provide information on financial disclosures.
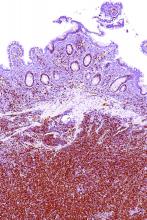
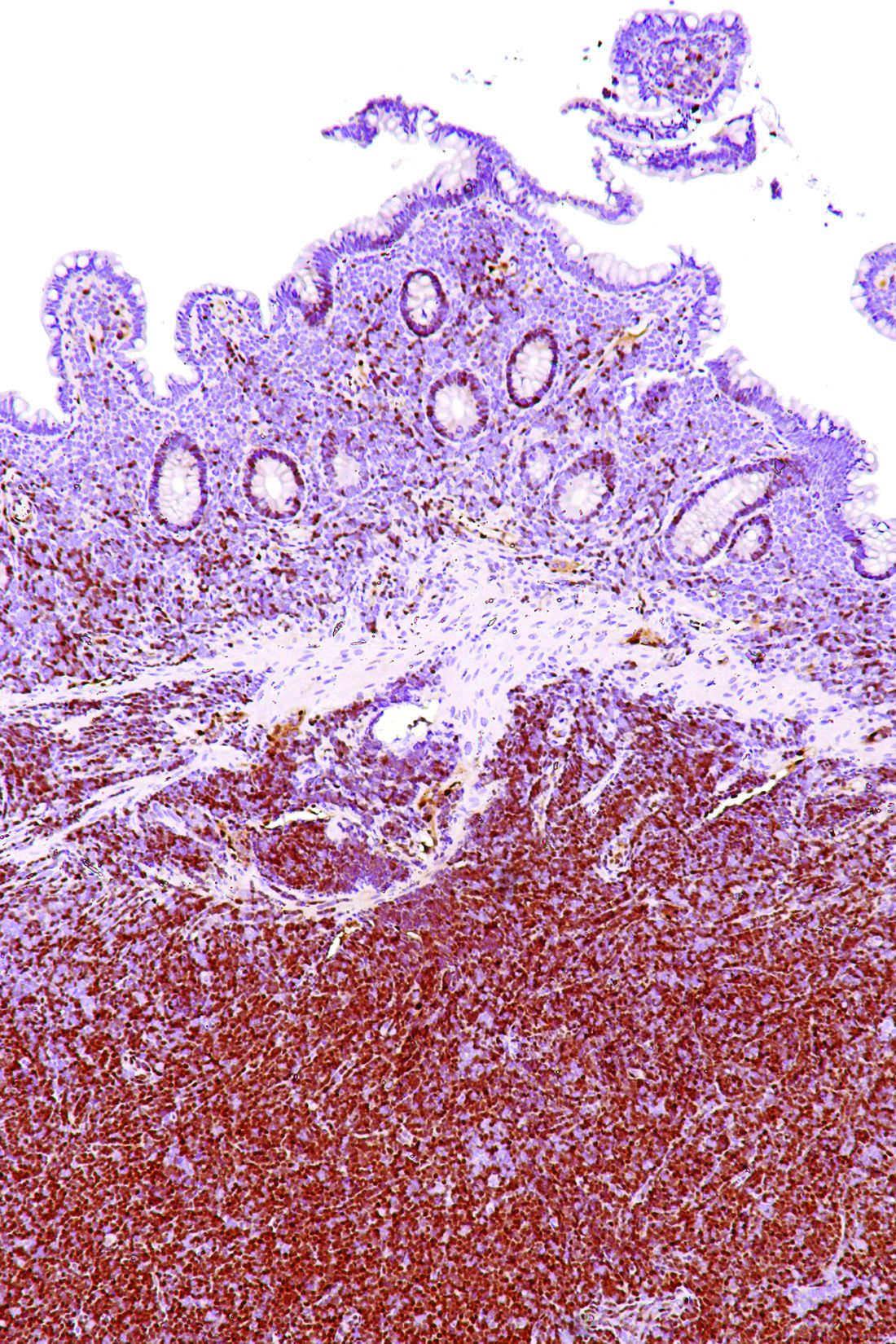
Staging PET/CT better defines extent of mantle cell lymphoma
Use of staging PET/CT better defines the extent of mantle cell lymphoma (MCL) in certain compartments, adding important information for treatment decisions, a cohort study suggests. However, patient selection and evaluation criteria are important for its utility.
“A correct and early identification of initial disease could be crucial because it could affect patient management and therapeutic choice,” Domenico Albano, MD, a nuclear medicine physician at the University of Brescia (Italy) and Spedali Civili Brescia, and colleagues wrote in Clinical Lymphoma, Myeloma, & Leukemia.
Using retrospective data from two centers and 122 patients with MCL, the investigators compared the utility of staging 18F-fluorodeoxyglucose (18F-FDG) PET/CT with that of other modalities used for detecting disease in specific compartments. They also assessed its impact on patient management.
The study results showed that, with the exception of a single patient having bone marrow involvement, all patients had positive PET/CT results, with detection of at least one hypermetabolic lesion.
For assessing nodal involvement, compared with CT alone, PET/CT detected additional lesions in 21% of patients. Splenic lesions were detected by PET/CT in 49% of patients and by CT alone in 47%.
For assessing bone marrow involvement, compared with bone marrow biopsy, PET/CT had a sensitivity of 52%, a specificity of 98%, a positive predictive value of 97%, a negative predictive value of 65%, and an overall accuracy of 74%.
For gastrointestinal involvement, compared with endoscopy, PET/CT had a sensitivity of 64% (78% after excluding diabetic patients taking metformin), a specificity of 91% (92%), a positive predictive value of 69% (72%), a negative predictive value of 90% (94%), and an overall accuracy of 85% (89%).
Ultimately, relative to CT alone, PET/CT altered stage and management in 19% of patients. Specifically, this imaging led to up-staging in 17% of patients, prompting clinicians to switch to more-aggressive chemotherapy, and down-staging in 2% of patients, prompting clinicians to skip unnecessary invasive therapies.
“We demonstrated that 18F-FDG pathologic uptake in MCL occurred in almost all patients, with excellent detection rate in nodal and splenic disease – indeed, better than CT,” the researchers wrote. “When we analyzed bone marrow and gastrointestinal involvement, the selection of patients excluding all conditions potentially affecting organ uptake and the use of specific criteria for the evaluation of these organs seemed to be crucial. Within these caveats, PET/CT showed good specificity for bone marrow and gastrointestinal evaluation.”
Study funding was not disclosed. Dr. Albano reported having no relevant conflicts of interest.
SOURCE: Albano D et al. Clin Lymphoma Myeloma Leuk. 2019 Aug;19(8):e457-64.
Use of staging PET/CT better defines the extent of mantle cell lymphoma (MCL) in certain compartments, adding important information for treatment decisions, a cohort study suggests. However, patient selection and evaluation criteria are important for its utility.
“A correct and early identification of initial disease could be crucial because it could affect patient management and therapeutic choice,” Domenico Albano, MD, a nuclear medicine physician at the University of Brescia (Italy) and Spedali Civili Brescia, and colleagues wrote in Clinical Lymphoma, Myeloma, & Leukemia.
Using retrospective data from two centers and 122 patients with MCL, the investigators compared the utility of staging 18F-fluorodeoxyglucose (18F-FDG) PET/CT with that of other modalities used for detecting disease in specific compartments. They also assessed its impact on patient management.
The study results showed that, with the exception of a single patient having bone marrow involvement, all patients had positive PET/CT results, with detection of at least one hypermetabolic lesion.
For assessing nodal involvement, compared with CT alone, PET/CT detected additional lesions in 21% of patients. Splenic lesions were detected by PET/CT in 49% of patients and by CT alone in 47%.
For assessing bone marrow involvement, compared with bone marrow biopsy, PET/CT had a sensitivity of 52%, a specificity of 98%, a positive predictive value of 97%, a negative predictive value of 65%, and an overall accuracy of 74%.
For gastrointestinal involvement, compared with endoscopy, PET/CT had a sensitivity of 64% (78% after excluding diabetic patients taking metformin), a specificity of 91% (92%), a positive predictive value of 69% (72%), a negative predictive value of 90% (94%), and an overall accuracy of 85% (89%).
Ultimately, relative to CT alone, PET/CT altered stage and management in 19% of patients. Specifically, this imaging led to up-staging in 17% of patients, prompting clinicians to switch to more-aggressive chemotherapy, and down-staging in 2% of patients, prompting clinicians to skip unnecessary invasive therapies.
“We demonstrated that 18F-FDG pathologic uptake in MCL occurred in almost all patients, with excellent detection rate in nodal and splenic disease – indeed, better than CT,” the researchers wrote. “When we analyzed bone marrow and gastrointestinal involvement, the selection of patients excluding all conditions potentially affecting organ uptake and the use of specific criteria for the evaluation of these organs seemed to be crucial. Within these caveats, PET/CT showed good specificity for bone marrow and gastrointestinal evaluation.”
Study funding was not disclosed. Dr. Albano reported having no relevant conflicts of interest.
SOURCE: Albano D et al. Clin Lymphoma Myeloma Leuk. 2019 Aug;19(8):e457-64.
Use of staging PET/CT better defines the extent of mantle cell lymphoma (MCL) in certain compartments, adding important information for treatment decisions, a cohort study suggests. However, patient selection and evaluation criteria are important for its utility.
“A correct and early identification of initial disease could be crucial because it could affect patient management and therapeutic choice,” Domenico Albano, MD, a nuclear medicine physician at the University of Brescia (Italy) and Spedali Civili Brescia, and colleagues wrote in Clinical Lymphoma, Myeloma, & Leukemia.
Using retrospective data from two centers and 122 patients with MCL, the investigators compared the utility of staging 18F-fluorodeoxyglucose (18F-FDG) PET/CT with that of other modalities used for detecting disease in specific compartments. They also assessed its impact on patient management.
The study results showed that, with the exception of a single patient having bone marrow involvement, all patients had positive PET/CT results, with detection of at least one hypermetabolic lesion.
For assessing nodal involvement, compared with CT alone, PET/CT detected additional lesions in 21% of patients. Splenic lesions were detected by PET/CT in 49% of patients and by CT alone in 47%.
For assessing bone marrow involvement, compared with bone marrow biopsy, PET/CT had a sensitivity of 52%, a specificity of 98%, a positive predictive value of 97%, a negative predictive value of 65%, and an overall accuracy of 74%.
For gastrointestinal involvement, compared with endoscopy, PET/CT had a sensitivity of 64% (78% after excluding diabetic patients taking metformin), a specificity of 91% (92%), a positive predictive value of 69% (72%), a negative predictive value of 90% (94%), and an overall accuracy of 85% (89%).
Ultimately, relative to CT alone, PET/CT altered stage and management in 19% of patients. Specifically, this imaging led to up-staging in 17% of patients, prompting clinicians to switch to more-aggressive chemotherapy, and down-staging in 2% of patients, prompting clinicians to skip unnecessary invasive therapies.
“We demonstrated that 18F-FDG pathologic uptake in MCL occurred in almost all patients, with excellent detection rate in nodal and splenic disease – indeed, better than CT,” the researchers wrote. “When we analyzed bone marrow and gastrointestinal involvement, the selection of patients excluding all conditions potentially affecting organ uptake and the use of specific criteria for the evaluation of these organs seemed to be crucial. Within these caveats, PET/CT showed good specificity for bone marrow and gastrointestinal evaluation.”
Study funding was not disclosed. Dr. Albano reported having no relevant conflicts of interest.
SOURCE: Albano D et al. Clin Lymphoma Myeloma Leuk. 2019 Aug;19(8):e457-64.
FROM CLINICAL LYMPHOMA, MYELOMA, & LEUKEMIA
Melflufen-dexamethasone active in patients with relapsed/refractory myeloma and EMD
BOSTON – Melflufen plus dexamethasone is active in patients with relapsed/refractory multiple myeloma, whether or not they have extramedullary disease (EMD), a phase 2 trial suggests.
In the HORIZON trial, melflufen-dexamethasone produced an overall response rate of 23% in patients with EMD and 27% in those without EMD.
Paul G. Richardson, MD, of Harvard Medical School and the Dana-Farber Cancer Institute, both in Boston, presented these results as a late-breaking abstract at the International Myeloma Workshop, held by the International Myeloma Society.
As of July 30, 2019, 136 patients had been treated on the HORIZON trial. The trial is enrolling patients with relapsed/refractory multiple myeloma refractory to pomalidomide, an anti-CD38 monoclonal antibody (mAb), or both. The patients must have received at least two prior lines of therapy, including a proteasome inhibitor (PI) and an immunomodulatory agent (IMiD).
Dr. Richardson presented results for 130 patients, 44 with EMD and 86 without it. The median age at baseline was 64 years in the EMD and non-EMD groups (overall range, 35-86 years). More than half of patients had high-risk cytogenetics (52% in the EMD group and 57% in the non-EMD group).
The median number of prior therapies was five in both the EMD and non-EMD groups. Most patients had received at least one prior transplant (73% in the EMD group and 69% in the non-EMD group). Most patients in both groups were refractory to an anti-CD38 mAb (93% EMD and 72% non-EMD); an IMiD and a PI (93% EMD and 90% non-EMD); an IMiD, a PI, and an anti-CD38 mAb (91% EMD and 63% non-EMD); and their last therapy (100% EMD and 95% non-EMD).
Dr. Richardson pointed out that the incidence of EMD in this trial is higher than has been reported previously. Of the 44 EMD patients, 26 had soft-tissue EMD, and 18 had bone-related EMD. Five patients had CNS involvement.
Another key finding, according to Dr. Richardson, was that EMD appeared to be associated with prior anti-CD38 therapy. Specifically, 40% of patients exposed to an anti-CD38 mAb had EMD, compared with 11% of patients who had not received an anti-CD38 mAb (P = .01).
“I don’t for a minute want to say that CD38-targeted therapy engenders extramedullary disease,” Dr. Richardson said. “I think what we can say, though, is that, once CD38 treatment fails a patient, extramedullary disease … is a very real challenge. Therefore, we need rationally targeted approaches, ideally in combination, to meet that challenge.”
The overall response rate was similar for EMD and non-EMD patients – 23% and 27%, respectively. The median duration of response was 3.4 months in the EMD patients and 4.4 months in the non-EMD group. The clinical benefit rate was 30% and 45%, respectively.
In the EMD group, the overall response rate was 19% in patients with soft-tissue EMD and 28% in bone-related EMD. None of the patients with CNS disease responded.
The median progression-free survival was 2.9 months for patients with EMD and 4.6 months for those without EMD. The median overall survival was 5.8 month and 11.6 months, respectively.
The median overall survival was 18.5 months in EMD responders and 17.2 months in non-EMD responders. The median overall survival was 5.1 months in EMD nonresponders and 8.5 months in non-EMD nonresponders.
In all, 54% of patients received subsequent therapy. There were no significant differences in outcomes between EMD and non-EMD patients.
The safety profiles were similar for EMD and non-EMD patients, Dr. Richardson said. Melflufen-dexamethasone was considered well tolerated overall, and there were no treatment-related deaths.
The most common treatment-emergent adverse events (grade 3 and 4, respectively) were thrombocytopenia (22% and 46%), neutropenia (32% and 35%), anemia (35% and 1%), white blood cell count decrease (10% and 7%), and pneumonia (7% and 1%).
This trial is sponsored by Oncopeptides. Dr. Richardson reported an advisory role and research funding from Oncopeptides.
SOURCE: Richardson PG et al. IMW 2019, Abstract OAB-086.
BOSTON – Melflufen plus dexamethasone is active in patients with relapsed/refractory multiple myeloma, whether or not they have extramedullary disease (EMD), a phase 2 trial suggests.
In the HORIZON trial, melflufen-dexamethasone produced an overall response rate of 23% in patients with EMD and 27% in those without EMD.
Paul G. Richardson, MD, of Harvard Medical School and the Dana-Farber Cancer Institute, both in Boston, presented these results as a late-breaking abstract at the International Myeloma Workshop, held by the International Myeloma Society.
As of July 30, 2019, 136 patients had been treated on the HORIZON trial. The trial is enrolling patients with relapsed/refractory multiple myeloma refractory to pomalidomide, an anti-CD38 monoclonal antibody (mAb), or both. The patients must have received at least two prior lines of therapy, including a proteasome inhibitor (PI) and an immunomodulatory agent (IMiD).
Dr. Richardson presented results for 130 patients, 44 with EMD and 86 without it. The median age at baseline was 64 years in the EMD and non-EMD groups (overall range, 35-86 years). More than half of patients had high-risk cytogenetics (52% in the EMD group and 57% in the non-EMD group).
The median number of prior therapies was five in both the EMD and non-EMD groups. Most patients had received at least one prior transplant (73% in the EMD group and 69% in the non-EMD group). Most patients in both groups were refractory to an anti-CD38 mAb (93% EMD and 72% non-EMD); an IMiD and a PI (93% EMD and 90% non-EMD); an IMiD, a PI, and an anti-CD38 mAb (91% EMD and 63% non-EMD); and their last therapy (100% EMD and 95% non-EMD).
Dr. Richardson pointed out that the incidence of EMD in this trial is higher than has been reported previously. Of the 44 EMD patients, 26 had soft-tissue EMD, and 18 had bone-related EMD. Five patients had CNS involvement.
Another key finding, according to Dr. Richardson, was that EMD appeared to be associated with prior anti-CD38 therapy. Specifically, 40% of patients exposed to an anti-CD38 mAb had EMD, compared with 11% of patients who had not received an anti-CD38 mAb (P = .01).
“I don’t for a minute want to say that CD38-targeted therapy engenders extramedullary disease,” Dr. Richardson said. “I think what we can say, though, is that, once CD38 treatment fails a patient, extramedullary disease … is a very real challenge. Therefore, we need rationally targeted approaches, ideally in combination, to meet that challenge.”
The overall response rate was similar for EMD and non-EMD patients – 23% and 27%, respectively. The median duration of response was 3.4 months in the EMD patients and 4.4 months in the non-EMD group. The clinical benefit rate was 30% and 45%, respectively.
In the EMD group, the overall response rate was 19% in patients with soft-tissue EMD and 28% in bone-related EMD. None of the patients with CNS disease responded.
The median progression-free survival was 2.9 months for patients with EMD and 4.6 months for those without EMD. The median overall survival was 5.8 month and 11.6 months, respectively.
The median overall survival was 18.5 months in EMD responders and 17.2 months in non-EMD responders. The median overall survival was 5.1 months in EMD nonresponders and 8.5 months in non-EMD nonresponders.
In all, 54% of patients received subsequent therapy. There were no significant differences in outcomes between EMD and non-EMD patients.
The safety profiles were similar for EMD and non-EMD patients, Dr. Richardson said. Melflufen-dexamethasone was considered well tolerated overall, and there were no treatment-related deaths.
The most common treatment-emergent adverse events (grade 3 and 4, respectively) were thrombocytopenia (22% and 46%), neutropenia (32% and 35%), anemia (35% and 1%), white blood cell count decrease (10% and 7%), and pneumonia (7% and 1%).
This trial is sponsored by Oncopeptides. Dr. Richardson reported an advisory role and research funding from Oncopeptides.
SOURCE: Richardson PG et al. IMW 2019, Abstract OAB-086.
BOSTON – Melflufen plus dexamethasone is active in patients with relapsed/refractory multiple myeloma, whether or not they have extramedullary disease (EMD), a phase 2 trial suggests.
In the HORIZON trial, melflufen-dexamethasone produced an overall response rate of 23% in patients with EMD and 27% in those without EMD.
Paul G. Richardson, MD, of Harvard Medical School and the Dana-Farber Cancer Institute, both in Boston, presented these results as a late-breaking abstract at the International Myeloma Workshop, held by the International Myeloma Society.
As of July 30, 2019, 136 patients had been treated on the HORIZON trial. The trial is enrolling patients with relapsed/refractory multiple myeloma refractory to pomalidomide, an anti-CD38 monoclonal antibody (mAb), or both. The patients must have received at least two prior lines of therapy, including a proteasome inhibitor (PI) and an immunomodulatory agent (IMiD).
Dr. Richardson presented results for 130 patients, 44 with EMD and 86 without it. The median age at baseline was 64 years in the EMD and non-EMD groups (overall range, 35-86 years). More than half of patients had high-risk cytogenetics (52% in the EMD group and 57% in the non-EMD group).
The median number of prior therapies was five in both the EMD and non-EMD groups. Most patients had received at least one prior transplant (73% in the EMD group and 69% in the non-EMD group). Most patients in both groups were refractory to an anti-CD38 mAb (93% EMD and 72% non-EMD); an IMiD and a PI (93% EMD and 90% non-EMD); an IMiD, a PI, and an anti-CD38 mAb (91% EMD and 63% non-EMD); and their last therapy (100% EMD and 95% non-EMD).
Dr. Richardson pointed out that the incidence of EMD in this trial is higher than has been reported previously. Of the 44 EMD patients, 26 had soft-tissue EMD, and 18 had bone-related EMD. Five patients had CNS involvement.
Another key finding, according to Dr. Richardson, was that EMD appeared to be associated with prior anti-CD38 therapy. Specifically, 40% of patients exposed to an anti-CD38 mAb had EMD, compared with 11% of patients who had not received an anti-CD38 mAb (P = .01).
“I don’t for a minute want to say that CD38-targeted therapy engenders extramedullary disease,” Dr. Richardson said. “I think what we can say, though, is that, once CD38 treatment fails a patient, extramedullary disease … is a very real challenge. Therefore, we need rationally targeted approaches, ideally in combination, to meet that challenge.”
The overall response rate was similar for EMD and non-EMD patients – 23% and 27%, respectively. The median duration of response was 3.4 months in the EMD patients and 4.4 months in the non-EMD group. The clinical benefit rate was 30% and 45%, respectively.
In the EMD group, the overall response rate was 19% in patients with soft-tissue EMD and 28% in bone-related EMD. None of the patients with CNS disease responded.
The median progression-free survival was 2.9 months for patients with EMD and 4.6 months for those without EMD. The median overall survival was 5.8 month and 11.6 months, respectively.
The median overall survival was 18.5 months in EMD responders and 17.2 months in non-EMD responders. The median overall survival was 5.1 months in EMD nonresponders and 8.5 months in non-EMD nonresponders.
In all, 54% of patients received subsequent therapy. There were no significant differences in outcomes between EMD and non-EMD patients.
The safety profiles were similar for EMD and non-EMD patients, Dr. Richardson said. Melflufen-dexamethasone was considered well tolerated overall, and there were no treatment-related deaths.
The most common treatment-emergent adverse events (grade 3 and 4, respectively) were thrombocytopenia (22% and 46%), neutropenia (32% and 35%), anemia (35% and 1%), white blood cell count decrease (10% and 7%), and pneumonia (7% and 1%).
This trial is sponsored by Oncopeptides. Dr. Richardson reported an advisory role and research funding from Oncopeptides.
SOURCE: Richardson PG et al. IMW 2019, Abstract OAB-086.
REPORTING FROM IMW 2019
Genetic analysis highlights value of germline variants in MDS, AML
Germline DDX41 mutations were found to be relatively prevalent and showed favorable outcomes in a cohort of patients with myelodysplastic syndromes or acute myeloid leukemia, according to a genetic analysis.
The results demonstrate that systematic genetic testing for DDX41 mutations may aid in clinical decision making for adults with myelodysplastic syndromes (MDS) or acute myeloid leukemia (AML).
“We screened a large, unselected cohort of adult patients diagnosed with MDS/AML to analyze the biological and clinical features of DDX41-related myeloid malignancies,” wrote Marie Sébert, MD, PhD, of Hôpital Saint Louis in Paris and colleagues. The results were published in Blood.
The researchers used next-generation sequencing to analyze blood and bone marrow samples from 1,385 patients with MDS or AML to detect DDX41 mutations. A variant allele frequency of greater than 0.4 was regarded as indicative of a germline origin, and only specific variants (minor allele frequency of less than 0.01) were included.
The team analyzed various parameters relevant to DDX41-related myeloid disorders, including patient demographics, karyotyping, and treatment response rates, in addition to the prevalence of DDX41-related malignancies.
A total of 28 distinct germline DDX41 variants were detected among 43 unrelated patients. The researchers classified 21 of the variants as causal, with the rest being of unknown significance.
“We focused on the 33 patients having causal variants, representing 2.4% of our cohort,” they wrote.
The majority of patients with DDX41-related MDS/AML were male, with a median age of 69 years. Few patients (27%) had a family history of blood malignancies, while the majority of patients (85%) had a normal karyotype.
With respect to treatment, most high-risk patients received either azacitidine or intensive chemotherapy, with overall response rates of 73% and 100%, respectively. The median overall survival was 5.2 years.
There are currently no consensus recommendations on genetic counseling and follow-up of asymptomatic carriers and more studies are needed to refine clinical management and genetic counseling, the researchers wrote. “However, in our experience, DDX41-mutated patients frequently presented mild cytopenias years before overt hematological myeloid malignancy, suggesting that watchful surveillance would allow the detection of disease evolution.”
No funding sources were reported, and the authors reported having no conflicts of interest.
SOURCE: Sébert M et al. Blood. 2019 Sep 4. doi: 10.1182/blood.2019000909.
Germline DDX41 mutations were found to be relatively prevalent and showed favorable outcomes in a cohort of patients with myelodysplastic syndromes or acute myeloid leukemia, according to a genetic analysis.
The results demonstrate that systematic genetic testing for DDX41 mutations may aid in clinical decision making for adults with myelodysplastic syndromes (MDS) or acute myeloid leukemia (AML).
“We screened a large, unselected cohort of adult patients diagnosed with MDS/AML to analyze the biological and clinical features of DDX41-related myeloid malignancies,” wrote Marie Sébert, MD, PhD, of Hôpital Saint Louis in Paris and colleagues. The results were published in Blood.
The researchers used next-generation sequencing to analyze blood and bone marrow samples from 1,385 patients with MDS or AML to detect DDX41 mutations. A variant allele frequency of greater than 0.4 was regarded as indicative of a germline origin, and only specific variants (minor allele frequency of less than 0.01) were included.
The team analyzed various parameters relevant to DDX41-related myeloid disorders, including patient demographics, karyotyping, and treatment response rates, in addition to the prevalence of DDX41-related malignancies.
A total of 28 distinct germline DDX41 variants were detected among 43 unrelated patients. The researchers classified 21 of the variants as causal, with the rest being of unknown significance.
“We focused on the 33 patients having causal variants, representing 2.4% of our cohort,” they wrote.
The majority of patients with DDX41-related MDS/AML were male, with a median age of 69 years. Few patients (27%) had a family history of blood malignancies, while the majority of patients (85%) had a normal karyotype.
With respect to treatment, most high-risk patients received either azacitidine or intensive chemotherapy, with overall response rates of 73% and 100%, respectively. The median overall survival was 5.2 years.
There are currently no consensus recommendations on genetic counseling and follow-up of asymptomatic carriers and more studies are needed to refine clinical management and genetic counseling, the researchers wrote. “However, in our experience, DDX41-mutated patients frequently presented mild cytopenias years before overt hematological myeloid malignancy, suggesting that watchful surveillance would allow the detection of disease evolution.”
No funding sources were reported, and the authors reported having no conflicts of interest.
SOURCE: Sébert M et al. Blood. 2019 Sep 4. doi: 10.1182/blood.2019000909.
Germline DDX41 mutations were found to be relatively prevalent and showed favorable outcomes in a cohort of patients with myelodysplastic syndromes or acute myeloid leukemia, according to a genetic analysis.
The results demonstrate that systematic genetic testing for DDX41 mutations may aid in clinical decision making for adults with myelodysplastic syndromes (MDS) or acute myeloid leukemia (AML).
“We screened a large, unselected cohort of adult patients diagnosed with MDS/AML to analyze the biological and clinical features of DDX41-related myeloid malignancies,” wrote Marie Sébert, MD, PhD, of Hôpital Saint Louis in Paris and colleagues. The results were published in Blood.
The researchers used next-generation sequencing to analyze blood and bone marrow samples from 1,385 patients with MDS or AML to detect DDX41 mutations. A variant allele frequency of greater than 0.4 was regarded as indicative of a germline origin, and only specific variants (minor allele frequency of less than 0.01) were included.
The team analyzed various parameters relevant to DDX41-related myeloid disorders, including patient demographics, karyotyping, and treatment response rates, in addition to the prevalence of DDX41-related malignancies.
A total of 28 distinct germline DDX41 variants were detected among 43 unrelated patients. The researchers classified 21 of the variants as causal, with the rest being of unknown significance.
“We focused on the 33 patients having causal variants, representing 2.4% of our cohort,” they wrote.
The majority of patients with DDX41-related MDS/AML were male, with a median age of 69 years. Few patients (27%) had a family history of blood malignancies, while the majority of patients (85%) had a normal karyotype.
With respect to treatment, most high-risk patients received either azacitidine or intensive chemotherapy, with overall response rates of 73% and 100%, respectively. The median overall survival was 5.2 years.
There are currently no consensus recommendations on genetic counseling and follow-up of asymptomatic carriers and more studies are needed to refine clinical management and genetic counseling, the researchers wrote. “However, in our experience, DDX41-mutated patients frequently presented mild cytopenias years before overt hematological myeloid malignancy, suggesting that watchful surveillance would allow the detection of disease evolution.”
No funding sources were reported, and the authors reported having no conflicts of interest.
SOURCE: Sébert M et al. Blood. 2019 Sep 4. doi: 10.1182/blood.2019000909.
FROM BLOOD
Could home care replace inpatient HSCT?
Can receiving all posttransplant care at home benefit patients undergoing hematopoietic stem cell transplant (HSCT)? Researchers are conducting phase 2 trials to find out.
Nelson Chao, MD, and colleagues at Duke University in Durham, N.C., completed a phase 1 trial that suggested post-HSCT care at home was feasible and safe (Blood. 2017;130:745).
Now, the team is conducting phase 2 trials – NCT01725022 and NCT02218151 – comparing patients who receive all posttransplant care at home with patients treated in the hospital or in the outpatient setting with daily visits to the clinic.
The main goal is to determine if allogeneic HSCT recipients treated at home can maintain their normal microbiome and, as a result, have a lower risk of graft-versus-host disease (GVHD). The researchers are also looking at other outcomes such as quality of life, treatment-related morbidities and mortality, and the cost of care for both allogeneic and autologous transplant recipients.
To be eligible for home care after HSCT, a patient must live within a 90-minute driving distance of Duke and have a caregiver available at home. The patient’s home must pass an inspection, showing it to be free of sources for potential infection, such as mold or pets that sleep in the patient’s bed.
When the time comes for treatment, the patient receives conditioning at the hospital but can return home the day before or the day of transplant. After discharge, the patient is visited by a nurse practitioner or physician assistant each morning for a physical examination and blood draw.
In the afternoon, the patient is visited by a clinic nurse who brings any necessary supplies or treatments, such as blood products or intravenous antibiotics. The patient also has daily video calls with an attending physician and can be admitted to the hospital for any events that cannot be managed in the home setting.
Patients can have visitors and spend time away from home, but precautions are necessary. Friends or family who are sick should not be allowed to visit, and patients should avoid crowds when they go out.

Initial findings
The Duke team has treated 41 HSCT recipients at home so far. Dr. Chao said it’s still too early to draw any conclusions about differences in outcomes between home care and inpatient/outpatient HSCT.
However, a preliminary analysis of costs suggests home care is cheaper than inpatient HSCT. The researchers found that, for the first several transplants, at day 60, the cost of home care was roughly half that of inpatient HSCT.
In addition, patients seem to be happy with posttransplant care at home.
“The patients love being at home, in their own environment, with their families,” Dr. Chao said. “Almost every single patient [in the phase 1 trial] said that he or she liked it much better. There was one patient in the phase 1 that felt a little isolated, and I can see why because we say, ‘You can stay home, but don’t have a whole lot of people in.’ ”
One patient’s experience
Beth Vanderkin said it was “a blessing” to receive care at home after undergoing HSCT at Duke.
Ms. Vanderkin was diagnosed with diffuse large B-cell lymphoma in 2014. After two chemotherapy regimens failed to shrink the tumor in her chest, she underwent radiotherapy and responded well. When a PET scan revealed the tumor had gone completely, she proceeded to transplant.
She received a haploidentical HSCT using cells donated by her eldest daughter, Hannah Eichhorst. Ms. Vanderkin received the transplant in the hospital, and for 2 weeks after that, she made daily visits to the transplant clinic.
After those 2 weeks, Ms. Vanderkin continued her treatment at home. Like other patients eligible for home care, Ms. Vanderkin lived close to Duke, had a caregiver available, and had passed a home inspection. The Duke team shipped the needed medical supplies to her house and arranged twice-daily visits from nurses and daily video calls with a doctor.
Ms. Vanderkin said receiving care at home was “a game changer.” She derived comfort from recovering in her own environment, could spend more time with her family, and didn’t have to miss special events. While receiving care at home, Ms. Vanderkin attended the homecoming event where her son, Josiah, was part of the court. Wearing a face mask and carrying a portable pump in her purse, Ms. Vanderkin joined other mothers in escorting their children onto the football field.
“I got to escort my son out onto the field, and he was crowned king that night,” Ms. Vanderkin said. “I didn’t do a lot of things [while receiving care at home], but there were things I didn’t have to miss because I was at home and not in the hospital.”
Ms. Vanderkin said home care was also beneficial for her husband, who was her caregiver. Thomas Vanderkin was able to work from home while caring for his wife, and the daily nurses’ visits allowed him to run errands without having to leave Ms. Vanderkin alone.
Since her experience with home care, Ms. Vanderkin has spent many more days in the hospital and clinic. She experienced a relapse after the transplant and went on to receive more chemotherapy as well as ipilimumab. She responded to that treatment and has now been cancer-free for 3 years.
The ipilimumab did cause side effects, including intestinal problems that resulted in the need for parenteral nutrition. This side effect was made more bearable, Ms. Vanderkin said, because she was able to receive the parenteral nutrition at home. She and her husband were comfortable with additional home care because of their positive experience with posttransplant care.
“I think we’re conditioned to think that, to receive the best care, we have to be sitting in a hospital room or a clinic, but I think there’s a lot of things we can probably do at home,” Ms. Vanderkin said. “And we might fare a lot better as patients if we’re in an environment that we feel comfortable in.”
Experience at other centers
The team at Duke is not the first to study HSCT care at home. In fact, researchers in Sweden have been studying posttransplant home care since 1998.
A pilot trial the group published in 2000 suggested that home care was safe and, in some ways, superior to inpatient HSCT (Bone Marrow Transplant. 2000 Nov;26[10]:1057-60). Patients treated at home had a lower rate of bacteremia, fewer days of total parenteral nutrition, fewer erythrocyte transfusions, and fewer days on antibiotics and analgesics. Rates of fever, engraftment time, and acute GVHD were similar between the inpatient and home-care groups.
A study published by the same researchers in 2002 showed that patients who received home care had lower rates of grade 2-4 acute GVHD and transplant-related mortality compared to inpatients (Blood. 2002 Dec 15;100[13]:4317-24). Two-year overall survival was superior with home care as well.
On the other hand, a study the group published in 2013 showed no significant differences in 5-year survival, transplant-related mortality, relapse, or chronic GVHD between inpatients and those who received care at home (Biol Blood Marrow Transplant. 2013. doi: 10.1016/j.bbmt.2012.11.5189).
The phase 2 trials at Duke should provide more insight into patient outcomes, but results probably won’t be available for 2 more years, Dr. Chao said.
In the meantime, other U.S. researchers are studying home care as well. Memorial Sloan Kettering Cancer Center is conducting a pilot study to determine if HSCT care at home is feasible (NCT02671448).
Dr. Chao said home care should be possible for other centers, particularly those that already perform outpatient HSCT.
“Having the outpatient infrastructure to support these patients is a big step,” he said. “And I think we were able to do that mainly because we do most of our transplants in the outpatient setting already. So that jump to the home is a little less compared to a center that does no outpatient transplants.”
He added, “There’s a certain amount of inertia to overcome and a certain amount of apprehension from the caregivers initially because [patients aren’t] sitting in your unit all the time, but I don’t see this as a huge barrier.”
In fact, Dr. Chao said, if results with home care are favorable, it could potentially replace inpatient HSCT for certain patients.
Dr. Chao’s research is supported by Duke University, and he reported having no relevant financial disclosures.
Can receiving all posttransplant care at home benefit patients undergoing hematopoietic stem cell transplant (HSCT)? Researchers are conducting phase 2 trials to find out.
Nelson Chao, MD, and colleagues at Duke University in Durham, N.C., completed a phase 1 trial that suggested post-HSCT care at home was feasible and safe (Blood. 2017;130:745).
Now, the team is conducting phase 2 trials – NCT01725022 and NCT02218151 – comparing patients who receive all posttransplant care at home with patients treated in the hospital or in the outpatient setting with daily visits to the clinic.
The main goal is to determine if allogeneic HSCT recipients treated at home can maintain their normal microbiome and, as a result, have a lower risk of graft-versus-host disease (GVHD). The researchers are also looking at other outcomes such as quality of life, treatment-related morbidities and mortality, and the cost of care for both allogeneic and autologous transplant recipients.
To be eligible for home care after HSCT, a patient must live within a 90-minute driving distance of Duke and have a caregiver available at home. The patient’s home must pass an inspection, showing it to be free of sources for potential infection, such as mold or pets that sleep in the patient’s bed.
When the time comes for treatment, the patient receives conditioning at the hospital but can return home the day before or the day of transplant. After discharge, the patient is visited by a nurse practitioner or physician assistant each morning for a physical examination and blood draw.
In the afternoon, the patient is visited by a clinic nurse who brings any necessary supplies or treatments, such as blood products or intravenous antibiotics. The patient also has daily video calls with an attending physician and can be admitted to the hospital for any events that cannot be managed in the home setting.
Patients can have visitors and spend time away from home, but precautions are necessary. Friends or family who are sick should not be allowed to visit, and patients should avoid crowds when they go out.

Initial findings
The Duke team has treated 41 HSCT recipients at home so far. Dr. Chao said it’s still too early to draw any conclusions about differences in outcomes between home care and inpatient/outpatient HSCT.
However, a preliminary analysis of costs suggests home care is cheaper than inpatient HSCT. The researchers found that, for the first several transplants, at day 60, the cost of home care was roughly half that of inpatient HSCT.
In addition, patients seem to be happy with posttransplant care at home.
“The patients love being at home, in their own environment, with their families,” Dr. Chao said. “Almost every single patient [in the phase 1 trial] said that he or she liked it much better. There was one patient in the phase 1 that felt a little isolated, and I can see why because we say, ‘You can stay home, but don’t have a whole lot of people in.’ ”
One patient’s experience
Beth Vanderkin said it was “a blessing” to receive care at home after undergoing HSCT at Duke.
Ms. Vanderkin was diagnosed with diffuse large B-cell lymphoma in 2014. After two chemotherapy regimens failed to shrink the tumor in her chest, she underwent radiotherapy and responded well. When a PET scan revealed the tumor had gone completely, she proceeded to transplant.
She received a haploidentical HSCT using cells donated by her eldest daughter, Hannah Eichhorst. Ms. Vanderkin received the transplant in the hospital, and for 2 weeks after that, she made daily visits to the transplant clinic.
After those 2 weeks, Ms. Vanderkin continued her treatment at home. Like other patients eligible for home care, Ms. Vanderkin lived close to Duke, had a caregiver available, and had passed a home inspection. The Duke team shipped the needed medical supplies to her house and arranged twice-daily visits from nurses and daily video calls with a doctor.
Ms. Vanderkin said receiving care at home was “a game changer.” She derived comfort from recovering in her own environment, could spend more time with her family, and didn’t have to miss special events. While receiving care at home, Ms. Vanderkin attended the homecoming event where her son, Josiah, was part of the court. Wearing a face mask and carrying a portable pump in her purse, Ms. Vanderkin joined other mothers in escorting their children onto the football field.
“I got to escort my son out onto the field, and he was crowned king that night,” Ms. Vanderkin said. “I didn’t do a lot of things [while receiving care at home], but there were things I didn’t have to miss because I was at home and not in the hospital.”
Ms. Vanderkin said home care was also beneficial for her husband, who was her caregiver. Thomas Vanderkin was able to work from home while caring for his wife, and the daily nurses’ visits allowed him to run errands without having to leave Ms. Vanderkin alone.
Since her experience with home care, Ms. Vanderkin has spent many more days in the hospital and clinic. She experienced a relapse after the transplant and went on to receive more chemotherapy as well as ipilimumab. She responded to that treatment and has now been cancer-free for 3 years.
The ipilimumab did cause side effects, including intestinal problems that resulted in the need for parenteral nutrition. This side effect was made more bearable, Ms. Vanderkin said, because she was able to receive the parenteral nutrition at home. She and her husband were comfortable with additional home care because of their positive experience with posttransplant care.
“I think we’re conditioned to think that, to receive the best care, we have to be sitting in a hospital room or a clinic, but I think there’s a lot of things we can probably do at home,” Ms. Vanderkin said. “And we might fare a lot better as patients if we’re in an environment that we feel comfortable in.”
Experience at other centers
The team at Duke is not the first to study HSCT care at home. In fact, researchers in Sweden have been studying posttransplant home care since 1998.
A pilot trial the group published in 2000 suggested that home care was safe and, in some ways, superior to inpatient HSCT (Bone Marrow Transplant. 2000 Nov;26[10]:1057-60). Patients treated at home had a lower rate of bacteremia, fewer days of total parenteral nutrition, fewer erythrocyte transfusions, and fewer days on antibiotics and analgesics. Rates of fever, engraftment time, and acute GVHD were similar between the inpatient and home-care groups.
A study published by the same researchers in 2002 showed that patients who received home care had lower rates of grade 2-4 acute GVHD and transplant-related mortality compared to inpatients (Blood. 2002 Dec 15;100[13]:4317-24). Two-year overall survival was superior with home care as well.
On the other hand, a study the group published in 2013 showed no significant differences in 5-year survival, transplant-related mortality, relapse, or chronic GVHD between inpatients and those who received care at home (Biol Blood Marrow Transplant. 2013. doi: 10.1016/j.bbmt.2012.11.5189).
The phase 2 trials at Duke should provide more insight into patient outcomes, but results probably won’t be available for 2 more years, Dr. Chao said.
In the meantime, other U.S. researchers are studying home care as well. Memorial Sloan Kettering Cancer Center is conducting a pilot study to determine if HSCT care at home is feasible (NCT02671448).
Dr. Chao said home care should be possible for other centers, particularly those that already perform outpatient HSCT.
“Having the outpatient infrastructure to support these patients is a big step,” he said. “And I think we were able to do that mainly because we do most of our transplants in the outpatient setting already. So that jump to the home is a little less compared to a center that does no outpatient transplants.”
He added, “There’s a certain amount of inertia to overcome and a certain amount of apprehension from the caregivers initially because [patients aren’t] sitting in your unit all the time, but I don’t see this as a huge barrier.”
In fact, Dr. Chao said, if results with home care are favorable, it could potentially replace inpatient HSCT for certain patients.
Dr. Chao’s research is supported by Duke University, and he reported having no relevant financial disclosures.
Can receiving all posttransplant care at home benefit patients undergoing hematopoietic stem cell transplant (HSCT)? Researchers are conducting phase 2 trials to find out.
Nelson Chao, MD, and colleagues at Duke University in Durham, N.C., completed a phase 1 trial that suggested post-HSCT care at home was feasible and safe (Blood. 2017;130:745).
Now, the team is conducting phase 2 trials – NCT01725022 and NCT02218151 – comparing patients who receive all posttransplant care at home with patients treated in the hospital or in the outpatient setting with daily visits to the clinic.
The main goal is to determine if allogeneic HSCT recipients treated at home can maintain their normal microbiome and, as a result, have a lower risk of graft-versus-host disease (GVHD). The researchers are also looking at other outcomes such as quality of life, treatment-related morbidities and mortality, and the cost of care for both allogeneic and autologous transplant recipients.
To be eligible for home care after HSCT, a patient must live within a 90-minute driving distance of Duke and have a caregiver available at home. The patient’s home must pass an inspection, showing it to be free of sources for potential infection, such as mold or pets that sleep in the patient’s bed.
When the time comes for treatment, the patient receives conditioning at the hospital but can return home the day before or the day of transplant. After discharge, the patient is visited by a nurse practitioner or physician assistant each morning for a physical examination and blood draw.
In the afternoon, the patient is visited by a clinic nurse who brings any necessary supplies or treatments, such as blood products or intravenous antibiotics. The patient also has daily video calls with an attending physician and can be admitted to the hospital for any events that cannot be managed in the home setting.
Patients can have visitors and spend time away from home, but precautions are necessary. Friends or family who are sick should not be allowed to visit, and patients should avoid crowds when they go out.

Initial findings
The Duke team has treated 41 HSCT recipients at home so far. Dr. Chao said it’s still too early to draw any conclusions about differences in outcomes between home care and inpatient/outpatient HSCT.
However, a preliminary analysis of costs suggests home care is cheaper than inpatient HSCT. The researchers found that, for the first several transplants, at day 60, the cost of home care was roughly half that of inpatient HSCT.
In addition, patients seem to be happy with posttransplant care at home.
“The patients love being at home, in their own environment, with their families,” Dr. Chao said. “Almost every single patient [in the phase 1 trial] said that he or she liked it much better. There was one patient in the phase 1 that felt a little isolated, and I can see why because we say, ‘You can stay home, but don’t have a whole lot of people in.’ ”
One patient’s experience
Beth Vanderkin said it was “a blessing” to receive care at home after undergoing HSCT at Duke.
Ms. Vanderkin was diagnosed with diffuse large B-cell lymphoma in 2014. After two chemotherapy regimens failed to shrink the tumor in her chest, she underwent radiotherapy and responded well. When a PET scan revealed the tumor had gone completely, she proceeded to transplant.
She received a haploidentical HSCT using cells donated by her eldest daughter, Hannah Eichhorst. Ms. Vanderkin received the transplant in the hospital, and for 2 weeks after that, she made daily visits to the transplant clinic.
After those 2 weeks, Ms. Vanderkin continued her treatment at home. Like other patients eligible for home care, Ms. Vanderkin lived close to Duke, had a caregiver available, and had passed a home inspection. The Duke team shipped the needed medical supplies to her house and arranged twice-daily visits from nurses and daily video calls with a doctor.
Ms. Vanderkin said receiving care at home was “a game changer.” She derived comfort from recovering in her own environment, could spend more time with her family, and didn’t have to miss special events. While receiving care at home, Ms. Vanderkin attended the homecoming event where her son, Josiah, was part of the court. Wearing a face mask and carrying a portable pump in her purse, Ms. Vanderkin joined other mothers in escorting their children onto the football field.
“I got to escort my son out onto the field, and he was crowned king that night,” Ms. Vanderkin said. “I didn’t do a lot of things [while receiving care at home], but there were things I didn’t have to miss because I was at home and not in the hospital.”
Ms. Vanderkin said home care was also beneficial for her husband, who was her caregiver. Thomas Vanderkin was able to work from home while caring for his wife, and the daily nurses’ visits allowed him to run errands without having to leave Ms. Vanderkin alone.
Since her experience with home care, Ms. Vanderkin has spent many more days in the hospital and clinic. She experienced a relapse after the transplant and went on to receive more chemotherapy as well as ipilimumab. She responded to that treatment and has now been cancer-free for 3 years.
The ipilimumab did cause side effects, including intestinal problems that resulted in the need for parenteral nutrition. This side effect was made more bearable, Ms. Vanderkin said, because she was able to receive the parenteral nutrition at home. She and her husband were comfortable with additional home care because of their positive experience with posttransplant care.
“I think we’re conditioned to think that, to receive the best care, we have to be sitting in a hospital room or a clinic, but I think there’s a lot of things we can probably do at home,” Ms. Vanderkin said. “And we might fare a lot better as patients if we’re in an environment that we feel comfortable in.”
Experience at other centers
The team at Duke is not the first to study HSCT care at home. In fact, researchers in Sweden have been studying posttransplant home care since 1998.
A pilot trial the group published in 2000 suggested that home care was safe and, in some ways, superior to inpatient HSCT (Bone Marrow Transplant. 2000 Nov;26[10]:1057-60). Patients treated at home had a lower rate of bacteremia, fewer days of total parenteral nutrition, fewer erythrocyte transfusions, and fewer days on antibiotics and analgesics. Rates of fever, engraftment time, and acute GVHD were similar between the inpatient and home-care groups.
A study published by the same researchers in 2002 showed that patients who received home care had lower rates of grade 2-4 acute GVHD and transplant-related mortality compared to inpatients (Blood. 2002 Dec 15;100[13]:4317-24). Two-year overall survival was superior with home care as well.
On the other hand, a study the group published in 2013 showed no significant differences in 5-year survival, transplant-related mortality, relapse, or chronic GVHD between inpatients and those who received care at home (Biol Blood Marrow Transplant. 2013. doi: 10.1016/j.bbmt.2012.11.5189).
The phase 2 trials at Duke should provide more insight into patient outcomes, but results probably won’t be available for 2 more years, Dr. Chao said.
In the meantime, other U.S. researchers are studying home care as well. Memorial Sloan Kettering Cancer Center is conducting a pilot study to determine if HSCT care at home is feasible (NCT02671448).
Dr. Chao said home care should be possible for other centers, particularly those that already perform outpatient HSCT.
“Having the outpatient infrastructure to support these patients is a big step,” he said. “And I think we were able to do that mainly because we do most of our transplants in the outpatient setting already. So that jump to the home is a little less compared to a center that does no outpatient transplants.”
He added, “There’s a certain amount of inertia to overcome and a certain amount of apprehension from the caregivers initially because [patients aren’t] sitting in your unit all the time, but I don’t see this as a huge barrier.”
In fact, Dr. Chao said, if results with home care are favorable, it could potentially replace inpatient HSCT for certain patients.
Dr. Chao’s research is supported by Duke University, and he reported having no relevant financial disclosures.