User login
Although 2.4 million adults in the United States have been diagnosed with hepatitis C virus (HCV) infection, it remains underdiagnosed and undertreated, particularly among difficult to reach populations, such as persons who inject drugs, marginally housed individuals, correctional populations, and pregnant women.1 Though the US Preventive Services Task Force (USPSTF) broadened HCV screening recommendations to include individuals aged 18 to 79 years, rates of new HCV prescriptions sharply declined during the COVID-19 pandemic.2,3
During the pandemic, many health care systems adopted virtual health care modalities. Within the Veteran Health Administration (VHA), there was an 11-fold increase in virtual encounters. However, veterans aged > 45 years, homeless, and had other insurance were less likely to utilize virtual care.4,5 As health care delivery continues to evolve, health systems must adapt and test innovative models for the treatment of HCV.
There is limited understanding of HCV treatments when exclusively conducted virtually. The aim of this study was to evaluate the effects of the HCV treatment program at the Veterans Affairs Greater Los Angeles Healthcare System (VAGLAHS) during the early phase of the COVID-19 pandemic, when telehealth modalities and mail-order prescriptions were used for HCV diagnosis and treatment. The secondary aim of this study was to understand patient factors associated with treatment initiation and discontinuation for patients using telehealth.
Methods
The VHA is the largest provider of HCV care in the US.6 At VAGLAHS, veterans with HCV are referred for evaluation to a viral hepatitis clinic staffed by gastroenterologists and infectious disease specialists. Veterans with detectable HCV on an HCV RNA test have an additional workup ordered if necessary and are referred to an HCV-specialist pharmacist or physician’s assistant to start treatment. In March 2020, all HCV evaluations and treatment initiation in the viral hepatitis clinic started being conducted exclusively via telehealth. This was the primary modality of HCV evaluations and treatment initiation until COVID-19 restrictions were lifted to permit in-person evaluations. Prescriptions were delivered by mail to patients following treatment initiation appointments.
We retrospectively reviewed electronic health records of veterans referred to start treatment March 1, 2020, through September 30, 2020. The endpoint of the reviewed records was set because during this specific time frame, VAGLAHS used an exclusively telehealth-based model for HCV evaluation and treatment. Patients were followed until June 15, 2021. Due to evolving COVID-19 restrictions at the time, and despite requests received, treatment initiations by the pharmacy team were suspended in March 2020 but HCV treatments resumed in May. Data collected included baseline demographics (age, sex, race, ethnicity, housing status, distance to VAGLAHS), comorbidities (cirrhosis, hepatitis B virus coinfection, HIV coinfection), psychiatric conditions (mood or psychotic disorder, alcohol use disorder [AUD], opioid use disorder), and treatment characteristics (HCV genotype, HCV treatment regimen, baseline viral load). Distance from the patient’s home to VAGLAHS was calculated using CDXZipStream software. Comorbidities and psychiatric conditions were identified by the presence of the appropriate diagnosis via International Statistical Classification of Diseases and Related Health Problems, Tenth Revision codes in the health record and confirmed by review of clinician notes. Active AUD was defined as: (1) the presence of AUD diagnosis code; (2) AUD Identification Test-Consumption (AUDIT-C) score of high or severe risk based on established cutoffs; and (3) active alcohol use noted in the electronic health record. All patients had an AUDIT-C score completed within 1 year of initiating treatment. Opioid use disorder was defined by the presence of diagnostic codes for opioid dependence or opioid abuse.
The reasons for treatment noninitiation and discontinuation were each captured. We calculated descriptive statistics to analyze the frequency distributions of all variables. Independent t tests were used to analyze continuous data and Pearson χ2 test was used to analyze categorical data. Statistical significance was set as P < .05.
Results
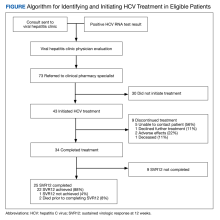
From March 1, 2020, through September 30, 2020, 73 veterans were referred to the HCV clinical pharmacist for treatment (Figure). Forty-three veterans (59%) initiated HCV treatment and 34 (79%) completed the full treatment course (Table 1). Twenty-five patients (65%) had their sustained virologic response at 12 weeks (SVR12) testing and 22 patients achieved SVR12 (88%; 30% of total sample). One patient did not achieve SVR, and 2 patients died (variceal hemorrhage and progression of cerebral amyloidosis/function decline) before the completion of laboratory testing. From March 2020 to May 2020, HCV treatments requests were paused as new COVID-19 policies were being introduced; 33 patients were referred during this time and 21 initiated treatment.
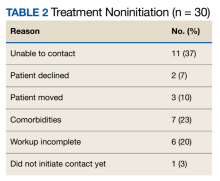
Veterans that did not start HCV treatment had a significantly higher rate of active AUD when compared with those that initiated treatment: 30% vs 9% (P = .02). Of the patients who started and discontinued treatment, none had active AUD. Other baseline demographics, clinical characteristics, and treatment characteristics were similar between the groups. No patient demographic characteristics were significantly associated with HCV treatment discontinuation. We did not observe any major health disparities in initiation or discontinuation by sex, race, ethnicity, or geography. Eleven patients (37%) could not be contacted, which was the most common reason veterans did not initiate treatment (Table 2). Of the 9 patients that did not complete SVR12, 5 patients could not be contacted for follow-up, which was the most common reason veterans discontinued treatment.
Discussion
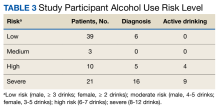
This study highlights the experience of treating patients with HCV with an exclusively telehealth model in the months following implementation of stay-at-home orders from March 19, 2020, to September 30, 2020, during the COVID-19 pandemic at VAGLAHS. We were able to successfully complete treatment for 34 veterans (47%) and achieved SVR rates of 88%. We found that AUD was associated with unsuccessful treatment initiation. There were no statistically significant patient characteristic findings for treatment discontinuation in our study (Table 3). Unhealthy alcohol use and AUD are highly prevalent among veterans with HCV and prior to the pandemic, studies have demonstrated AUD as a barrier to HCV treatment.7
Since worse hepatic outcomes have been observed in veterans with HCV and AUD and increased harmful patterns of drinking occurred during the pandemic, a renewed interest in treating AUD in these veterans during the era of telehealth is critical.8 While we were unable to ascertain whether alcohol misuse in our cohort increased during the pandemic or whether changes in drinking patterns affected HCV treatment outcomes before and after the pandemic, such an association should reinforce the need for clinicians to expeditiously link patients to substance use care. It should also stimulate further considerations of addressing social determinants of health not captured in this study.
During the pandemic, veterans with posttraumatic stress disorder, a history of serving in combat roles, and experiencing related financial stressors had higher risk of AUD.9,10 For veterans with AUD who initiated HCV treatment, none discontinued their therapy, aligning with other studies showed that patients with AUD were able to achieve high rates of SVR and emphasizing that veterans should be treated irrespective of an AUD diagnosis.11 However, more innovative engagement initiatives for veterans with AUD should be explored as we continue to adapt more telehealth-based care for HCV direct-acting antiviral treatments. A more in-depth understanding of how alcohol use relates to treatment noninitiation is warranted, as this may stem from behavioral patterns that could not be captured in the present study.
The inability to reach veterans by telephone was a major reason for noninitiation and discontinuation of treatment. While the expansion of telehealth services has been noted across the VHA, there is still room for improving methods of engaging veterans in health care postpandemic.12 Prior studies in veteran populations that were successful in increasing uptake of HCV treatment have employed telehealth strategies that further emphasizes its integral role in HCV elimination.13 Although our study did not show mental health comorbidities and housing status as statistically significant, it is important to note that 20% of patients referred for HCV treatment had an incomplete evaluation which can lead to potentially unobserved indicators not captured by our study such as quality of linkage to care. It is imperative to stress the best practices for HCV initiation by integrating a multidisciplinary team to address patients’ psychosocial comorbidities.14 Finally, we did not observe any major disparities in treating veterans with HCV during the pandemic. This observation is reassuring and consistent with other VHA data given the heightened recognition of health disparities seen in health care sectors across the country, especially evident during the COVID-19 pandemic and the current era of increased adaptation of telehealth.
Limitations
Limitations to this study include its retrospective nature, small sample size, and short study time frame as a proportion of veterans have yet to complete HCV treatment which can potentially explain how larger studies were able to find other statistically significant patient-related factors impacting treatment initiation compared to ours. Given the lack of universal standardized diagnostic criterion of AUD, this can limit how our study can be compared to others in similar populations. Additionally, this study was conducted at a single facility with a predominantly older male veteran population, which may not be generalizable to other populations.
Conclusions
Treating HCV during the COVID-19 pandemic with telehealth and mail-out medications was feasible and led to high SVR rates, but unhealthy alcohol use and an inability to contact veterans were predominant barriers to success. Future quality improvement efforts should focus on addressing these barriers and exploring the relationship between alcohol use and HCV treatment initiation.
1. Patel AA, Bui A, Prohl E, et al. Innovations in Hepatitis C Screening and Treatment. Hepatol Commun. 2020;5(3):371-386. Published 2020 Dec 7. doi:10.1002/hep4.1646
2. US Preventive Services Task Force, Owens DK, Davidson KW, et al. Screening for Hepatitis C Virus Infection in Adolescents and Adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2020;323(10):970-975. doi:10.1001/jama.2020.1123
3. Kaufman HW, Bull-Otterson L, Meyer WA 3rd, et al. Decreases in Hepatitis C Testing and Treatment During the COVID-19 Pandemic. Am J Prev Med. 2021;61(3):369-376. doi:10.1016/j.amepre.2021.03.011
4. Rosen CS, Morland LA, Glassman LH, et al. Virtual mental health care in the Veterans Health Administration’s immediate response to coronavirus disease-19. Am Psychol. 2021;76(1):26-38. doi:10.1037/amp0000751
5. Balut MD, Wyte-Lake T, Steers WN, et al. Expansion of telemedicine during COVID-19 at a VA specialty clinic. Healthc (Amst). 2022;10(1):100599. doi:10.1016/j.hjdsi.2021.100599
6. Belperio PS, Chartier M, Ross DB, Alaigh P, Shulkin D. Curing Hepatitis C Virus Infection: Best Practices From the U.S. Department of Veterans Affairs. Ann Intern Med. 2017;167(7):499-504. doi:10.7326/M17-1073
7. Lin M, Kramer J, White D, et al. Barriers to hepatitis C treatment in the era of direct-acting anti-viral agents. Aliment Pharmacol Ther. 2017;46(10):992-1000. doi:10.1111/apt.14328
8. Alavi M, Janjua NZ, Chong M, et al. The contribution of alcohol use disorder to decompensated cirrhosis among people with hepatitis C: An international study. J Hepatol. 2018;68(3):393-401. doi:10.1016/j.jhep.2017.10.019
9. Pedersen ER, Davis JP, Fitzke RE, Lee DS, Saba S. American Veterans in the Era of COVID-19: Reactions to the Pandemic, Posttraumatic Stress Disorder, and Substance Use Behaviors. Int J Ment Health Addict. 2023;21(2):767-782. doi:10.1007/s11469-021-00620-0
10. Na PJ, Norman SB, Nichter B, et al. Prevalence, risk and protective factors of alcohol use disorder during the COVID-19 pandemic in U.S. military veterans. Drug Alcohol Depend. 2021;225:108818. doi:10.1016/j.drugalcdep.2021.108818
11. Tsui JI, Williams EC, Green PK, Berry K, Su F, Ioannou GN. Alcohol use and hepatitis C virus treatment outcomes among patients receiving direct antiviral agents. Drug Alcohol Depend. 2016;169:101-109. doi:10.1016/j.drugalcdep.2016.10.021
12. Baum A, Kaboli PJ, Schwartz MD. Reduced In-Person and Increased Telehealth Outpatient Visits During the COVID-19 Pandemic. Ann Intern Med. 2021;174(1):129-131. doi:10.7326/M20-3026
13. Fleming BS, Ifeachor AP, Andres AM, et al. Improving Veteran Access to Treatment for Hepatitis C Virus Infection: Addressing social issues and treatment barriers significantly increases access to HCV care, and many veterans successfully start therapy with the help of additional support staff. Fed Pract. 2017;34(Suppl 4):S24-S28.
14. Belperio PS, Chartier M, Ross DB, Alaigh P, Shulkin D. Curing Hepatitis C Virus Infection: Best Practices From the U.S. Department of Veterans Affairs. Ann Intern Med. 2017;167(7):499-504. doi:10.7326/M17-1073
Although 2.4 million adults in the United States have been diagnosed with hepatitis C virus (HCV) infection, it remains underdiagnosed and undertreated, particularly among difficult to reach populations, such as persons who inject drugs, marginally housed individuals, correctional populations, and pregnant women.1 Though the US Preventive Services Task Force (USPSTF) broadened HCV screening recommendations to include individuals aged 18 to 79 years, rates of new HCV prescriptions sharply declined during the COVID-19 pandemic.2,3
During the pandemic, many health care systems adopted virtual health care modalities. Within the Veteran Health Administration (VHA), there was an 11-fold increase in virtual encounters. However, veterans aged > 45 years, homeless, and had other insurance were less likely to utilize virtual care.4,5 As health care delivery continues to evolve, health systems must adapt and test innovative models for the treatment of HCV.
There is limited understanding of HCV treatments when exclusively conducted virtually. The aim of this study was to evaluate the effects of the HCV treatment program at the Veterans Affairs Greater Los Angeles Healthcare System (VAGLAHS) during the early phase of the COVID-19 pandemic, when telehealth modalities and mail-order prescriptions were used for HCV diagnosis and treatment. The secondary aim of this study was to understand patient factors associated with treatment initiation and discontinuation for patients using telehealth.
Methods
The VHA is the largest provider of HCV care in the US.6 At VAGLAHS, veterans with HCV are referred for evaluation to a viral hepatitis clinic staffed by gastroenterologists and infectious disease specialists. Veterans with detectable HCV on an HCV RNA test have an additional workup ordered if necessary and are referred to an HCV-specialist pharmacist or physician’s assistant to start treatment. In March 2020, all HCV evaluations and treatment initiation in the viral hepatitis clinic started being conducted exclusively via telehealth. This was the primary modality of HCV evaluations and treatment initiation until COVID-19 restrictions were lifted to permit in-person evaluations. Prescriptions were delivered by mail to patients following treatment initiation appointments.
We retrospectively reviewed electronic health records of veterans referred to start treatment March 1, 2020, through September 30, 2020. The endpoint of the reviewed records was set because during this specific time frame, VAGLAHS used an exclusively telehealth-based model for HCV evaluation and treatment. Patients were followed until June 15, 2021. Due to evolving COVID-19 restrictions at the time, and despite requests received, treatment initiations by the pharmacy team were suspended in March 2020 but HCV treatments resumed in May. Data collected included baseline demographics (age, sex, race, ethnicity, housing status, distance to VAGLAHS), comorbidities (cirrhosis, hepatitis B virus coinfection, HIV coinfection), psychiatric conditions (mood or psychotic disorder, alcohol use disorder [AUD], opioid use disorder), and treatment characteristics (HCV genotype, HCV treatment regimen, baseline viral load). Distance from the patient’s home to VAGLAHS was calculated using CDXZipStream software. Comorbidities and psychiatric conditions were identified by the presence of the appropriate diagnosis via International Statistical Classification of Diseases and Related Health Problems, Tenth Revision codes in the health record and confirmed by review of clinician notes. Active AUD was defined as: (1) the presence of AUD diagnosis code; (2) AUD Identification Test-Consumption (AUDIT-C) score of high or severe risk based on established cutoffs; and (3) active alcohol use noted in the electronic health record. All patients had an AUDIT-C score completed within 1 year of initiating treatment. Opioid use disorder was defined by the presence of diagnostic codes for opioid dependence or opioid abuse.
The reasons for treatment noninitiation and discontinuation were each captured. We calculated descriptive statistics to analyze the frequency distributions of all variables. Independent t tests were used to analyze continuous data and Pearson χ2 test was used to analyze categorical data. Statistical significance was set as P < .05.
Results
From March 1, 2020, through September 30, 2020, 73 veterans were referred to the HCV clinical pharmacist for treatment (Figure). Forty-three veterans (59%) initiated HCV treatment and 34 (79%) completed the full treatment course (Table 1). Twenty-five patients (65%) had their sustained virologic response at 12 weeks (SVR12) testing and 22 patients achieved SVR12 (88%; 30% of total sample). One patient did not achieve SVR, and 2 patients died (variceal hemorrhage and progression of cerebral amyloidosis/function decline) before the completion of laboratory testing. From March 2020 to May 2020, HCV treatments requests were paused as new COVID-19 policies were being introduced; 33 patients were referred during this time and 21 initiated treatment.
Veterans that did not start HCV treatment had a significantly higher rate of active AUD when compared with those that initiated treatment: 30% vs 9% (P = .02). Of the patients who started and discontinued treatment, none had active AUD. Other baseline demographics, clinical characteristics, and treatment characteristics were similar between the groups. No patient demographic characteristics were significantly associated with HCV treatment discontinuation. We did not observe any major health disparities in initiation or discontinuation by sex, race, ethnicity, or geography. Eleven patients (37%) could not be contacted, which was the most common reason veterans did not initiate treatment (Table 2). Of the 9 patients that did not complete SVR12, 5 patients could not be contacted for follow-up, which was the most common reason veterans discontinued treatment.
Discussion
This study highlights the experience of treating patients with HCV with an exclusively telehealth model in the months following implementation of stay-at-home orders from March 19, 2020, to September 30, 2020, during the COVID-19 pandemic at VAGLAHS. We were able to successfully complete treatment for 34 veterans (47%) and achieved SVR rates of 88%. We found that AUD was associated with unsuccessful treatment initiation. There were no statistically significant patient characteristic findings for treatment discontinuation in our study (Table 3). Unhealthy alcohol use and AUD are highly prevalent among veterans with HCV and prior to the pandemic, studies have demonstrated AUD as a barrier to HCV treatment.7
Since worse hepatic outcomes have been observed in veterans with HCV and AUD and increased harmful patterns of drinking occurred during the pandemic, a renewed interest in treating AUD in these veterans during the era of telehealth is critical.8 While we were unable to ascertain whether alcohol misuse in our cohort increased during the pandemic or whether changes in drinking patterns affected HCV treatment outcomes before and after the pandemic, such an association should reinforce the need for clinicians to expeditiously link patients to substance use care. It should also stimulate further considerations of addressing social determinants of health not captured in this study.
During the pandemic, veterans with posttraumatic stress disorder, a history of serving in combat roles, and experiencing related financial stressors had higher risk of AUD.9,10 For veterans with AUD who initiated HCV treatment, none discontinued their therapy, aligning with other studies showed that patients with AUD were able to achieve high rates of SVR and emphasizing that veterans should be treated irrespective of an AUD diagnosis.11 However, more innovative engagement initiatives for veterans with AUD should be explored as we continue to adapt more telehealth-based care for HCV direct-acting antiviral treatments. A more in-depth understanding of how alcohol use relates to treatment noninitiation is warranted, as this may stem from behavioral patterns that could not be captured in the present study.
The inability to reach veterans by telephone was a major reason for noninitiation and discontinuation of treatment. While the expansion of telehealth services has been noted across the VHA, there is still room for improving methods of engaging veterans in health care postpandemic.12 Prior studies in veteran populations that were successful in increasing uptake of HCV treatment have employed telehealth strategies that further emphasizes its integral role in HCV elimination.13 Although our study did not show mental health comorbidities and housing status as statistically significant, it is important to note that 20% of patients referred for HCV treatment had an incomplete evaluation which can lead to potentially unobserved indicators not captured by our study such as quality of linkage to care. It is imperative to stress the best practices for HCV initiation by integrating a multidisciplinary team to address patients’ psychosocial comorbidities.14 Finally, we did not observe any major disparities in treating veterans with HCV during the pandemic. This observation is reassuring and consistent with other VHA data given the heightened recognition of health disparities seen in health care sectors across the country, especially evident during the COVID-19 pandemic and the current era of increased adaptation of telehealth.
Limitations
Limitations to this study include its retrospective nature, small sample size, and short study time frame as a proportion of veterans have yet to complete HCV treatment which can potentially explain how larger studies were able to find other statistically significant patient-related factors impacting treatment initiation compared to ours. Given the lack of universal standardized diagnostic criterion of AUD, this can limit how our study can be compared to others in similar populations. Additionally, this study was conducted at a single facility with a predominantly older male veteran population, which may not be generalizable to other populations.
Conclusions
Treating HCV during the COVID-19 pandemic with telehealth and mail-out medications was feasible and led to high SVR rates, but unhealthy alcohol use and an inability to contact veterans were predominant barriers to success. Future quality improvement efforts should focus on addressing these barriers and exploring the relationship between alcohol use and HCV treatment initiation.
Although 2.4 million adults in the United States have been diagnosed with hepatitis C virus (HCV) infection, it remains underdiagnosed and undertreated, particularly among difficult to reach populations, such as persons who inject drugs, marginally housed individuals, correctional populations, and pregnant women.1 Though the US Preventive Services Task Force (USPSTF) broadened HCV screening recommendations to include individuals aged 18 to 79 years, rates of new HCV prescriptions sharply declined during the COVID-19 pandemic.2,3
During the pandemic, many health care systems adopted virtual health care modalities. Within the Veteran Health Administration (VHA), there was an 11-fold increase in virtual encounters. However, veterans aged > 45 years, homeless, and had other insurance were less likely to utilize virtual care.4,5 As health care delivery continues to evolve, health systems must adapt and test innovative models for the treatment of HCV.
There is limited understanding of HCV treatments when exclusively conducted virtually. The aim of this study was to evaluate the effects of the HCV treatment program at the Veterans Affairs Greater Los Angeles Healthcare System (VAGLAHS) during the early phase of the COVID-19 pandemic, when telehealth modalities and mail-order prescriptions were used for HCV diagnosis and treatment. The secondary aim of this study was to understand patient factors associated with treatment initiation and discontinuation for patients using telehealth.
Methods
The VHA is the largest provider of HCV care in the US.6 At VAGLAHS, veterans with HCV are referred for evaluation to a viral hepatitis clinic staffed by gastroenterologists and infectious disease specialists. Veterans with detectable HCV on an HCV RNA test have an additional workup ordered if necessary and are referred to an HCV-specialist pharmacist or physician’s assistant to start treatment. In March 2020, all HCV evaluations and treatment initiation in the viral hepatitis clinic started being conducted exclusively via telehealth. This was the primary modality of HCV evaluations and treatment initiation until COVID-19 restrictions were lifted to permit in-person evaluations. Prescriptions were delivered by mail to patients following treatment initiation appointments.
We retrospectively reviewed electronic health records of veterans referred to start treatment March 1, 2020, through September 30, 2020. The endpoint of the reviewed records was set because during this specific time frame, VAGLAHS used an exclusively telehealth-based model for HCV evaluation and treatment. Patients were followed until June 15, 2021. Due to evolving COVID-19 restrictions at the time, and despite requests received, treatment initiations by the pharmacy team were suspended in March 2020 but HCV treatments resumed in May. Data collected included baseline demographics (age, sex, race, ethnicity, housing status, distance to VAGLAHS), comorbidities (cirrhosis, hepatitis B virus coinfection, HIV coinfection), psychiatric conditions (mood or psychotic disorder, alcohol use disorder [AUD], opioid use disorder), and treatment characteristics (HCV genotype, HCV treatment regimen, baseline viral load). Distance from the patient’s home to VAGLAHS was calculated using CDXZipStream software. Comorbidities and psychiatric conditions were identified by the presence of the appropriate diagnosis via International Statistical Classification of Diseases and Related Health Problems, Tenth Revision codes in the health record and confirmed by review of clinician notes. Active AUD was defined as: (1) the presence of AUD diagnosis code; (2) AUD Identification Test-Consumption (AUDIT-C) score of high or severe risk based on established cutoffs; and (3) active alcohol use noted in the electronic health record. All patients had an AUDIT-C score completed within 1 year of initiating treatment. Opioid use disorder was defined by the presence of diagnostic codes for opioid dependence or opioid abuse.
The reasons for treatment noninitiation and discontinuation were each captured. We calculated descriptive statistics to analyze the frequency distributions of all variables. Independent t tests were used to analyze continuous data and Pearson χ2 test was used to analyze categorical data. Statistical significance was set as P < .05.
Results
From March 1, 2020, through September 30, 2020, 73 veterans were referred to the HCV clinical pharmacist for treatment (Figure). Forty-three veterans (59%) initiated HCV treatment and 34 (79%) completed the full treatment course (Table 1). Twenty-five patients (65%) had their sustained virologic response at 12 weeks (SVR12) testing and 22 patients achieved SVR12 (88%; 30% of total sample). One patient did not achieve SVR, and 2 patients died (variceal hemorrhage and progression of cerebral amyloidosis/function decline) before the completion of laboratory testing. From March 2020 to May 2020, HCV treatments requests were paused as new COVID-19 policies were being introduced; 33 patients were referred during this time and 21 initiated treatment.
Veterans that did not start HCV treatment had a significantly higher rate of active AUD when compared with those that initiated treatment: 30% vs 9% (P = .02). Of the patients who started and discontinued treatment, none had active AUD. Other baseline demographics, clinical characteristics, and treatment characteristics were similar between the groups. No patient demographic characteristics were significantly associated with HCV treatment discontinuation. We did not observe any major health disparities in initiation or discontinuation by sex, race, ethnicity, or geography. Eleven patients (37%) could not be contacted, which was the most common reason veterans did not initiate treatment (Table 2). Of the 9 patients that did not complete SVR12, 5 patients could not be contacted for follow-up, which was the most common reason veterans discontinued treatment.
Discussion
This study highlights the experience of treating patients with HCV with an exclusively telehealth model in the months following implementation of stay-at-home orders from March 19, 2020, to September 30, 2020, during the COVID-19 pandemic at VAGLAHS. We were able to successfully complete treatment for 34 veterans (47%) and achieved SVR rates of 88%. We found that AUD was associated with unsuccessful treatment initiation. There were no statistically significant patient characteristic findings for treatment discontinuation in our study (Table 3). Unhealthy alcohol use and AUD are highly prevalent among veterans with HCV and prior to the pandemic, studies have demonstrated AUD as a barrier to HCV treatment.7
Since worse hepatic outcomes have been observed in veterans with HCV and AUD and increased harmful patterns of drinking occurred during the pandemic, a renewed interest in treating AUD in these veterans during the era of telehealth is critical.8 While we were unable to ascertain whether alcohol misuse in our cohort increased during the pandemic or whether changes in drinking patterns affected HCV treatment outcomes before and after the pandemic, such an association should reinforce the need for clinicians to expeditiously link patients to substance use care. It should also stimulate further considerations of addressing social determinants of health not captured in this study.
During the pandemic, veterans with posttraumatic stress disorder, a history of serving in combat roles, and experiencing related financial stressors had higher risk of AUD.9,10 For veterans with AUD who initiated HCV treatment, none discontinued their therapy, aligning with other studies showed that patients with AUD were able to achieve high rates of SVR and emphasizing that veterans should be treated irrespective of an AUD diagnosis.11 However, more innovative engagement initiatives for veterans with AUD should be explored as we continue to adapt more telehealth-based care for HCV direct-acting antiviral treatments. A more in-depth understanding of how alcohol use relates to treatment noninitiation is warranted, as this may stem from behavioral patterns that could not be captured in the present study.
The inability to reach veterans by telephone was a major reason for noninitiation and discontinuation of treatment. While the expansion of telehealth services has been noted across the VHA, there is still room for improving methods of engaging veterans in health care postpandemic.12 Prior studies in veteran populations that were successful in increasing uptake of HCV treatment have employed telehealth strategies that further emphasizes its integral role in HCV elimination.13 Although our study did not show mental health comorbidities and housing status as statistically significant, it is important to note that 20% of patients referred for HCV treatment had an incomplete evaluation which can lead to potentially unobserved indicators not captured by our study such as quality of linkage to care. It is imperative to stress the best practices for HCV initiation by integrating a multidisciplinary team to address patients’ psychosocial comorbidities.14 Finally, we did not observe any major disparities in treating veterans with HCV during the pandemic. This observation is reassuring and consistent with other VHA data given the heightened recognition of health disparities seen in health care sectors across the country, especially evident during the COVID-19 pandemic and the current era of increased adaptation of telehealth.
Limitations
Limitations to this study include its retrospective nature, small sample size, and short study time frame as a proportion of veterans have yet to complete HCV treatment which can potentially explain how larger studies were able to find other statistically significant patient-related factors impacting treatment initiation compared to ours. Given the lack of universal standardized diagnostic criterion of AUD, this can limit how our study can be compared to others in similar populations. Additionally, this study was conducted at a single facility with a predominantly older male veteran population, which may not be generalizable to other populations.
Conclusions
Treating HCV during the COVID-19 pandemic with telehealth and mail-out medications was feasible and led to high SVR rates, but unhealthy alcohol use and an inability to contact veterans were predominant barriers to success. Future quality improvement efforts should focus on addressing these barriers and exploring the relationship between alcohol use and HCV treatment initiation.
1. Patel AA, Bui A, Prohl E, et al. Innovations in Hepatitis C Screening and Treatment. Hepatol Commun. 2020;5(3):371-386. Published 2020 Dec 7. doi:10.1002/hep4.1646
2. US Preventive Services Task Force, Owens DK, Davidson KW, et al. Screening for Hepatitis C Virus Infection in Adolescents and Adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2020;323(10):970-975. doi:10.1001/jama.2020.1123
3. Kaufman HW, Bull-Otterson L, Meyer WA 3rd, et al. Decreases in Hepatitis C Testing and Treatment During the COVID-19 Pandemic. Am J Prev Med. 2021;61(3):369-376. doi:10.1016/j.amepre.2021.03.011
4. Rosen CS, Morland LA, Glassman LH, et al. Virtual mental health care in the Veterans Health Administration’s immediate response to coronavirus disease-19. Am Psychol. 2021;76(1):26-38. doi:10.1037/amp0000751
5. Balut MD, Wyte-Lake T, Steers WN, et al. Expansion of telemedicine during COVID-19 at a VA specialty clinic. Healthc (Amst). 2022;10(1):100599. doi:10.1016/j.hjdsi.2021.100599
6. Belperio PS, Chartier M, Ross DB, Alaigh P, Shulkin D. Curing Hepatitis C Virus Infection: Best Practices From the U.S. Department of Veterans Affairs. Ann Intern Med. 2017;167(7):499-504. doi:10.7326/M17-1073
7. Lin M, Kramer J, White D, et al. Barriers to hepatitis C treatment in the era of direct-acting anti-viral agents. Aliment Pharmacol Ther. 2017;46(10):992-1000. doi:10.1111/apt.14328
8. Alavi M, Janjua NZ, Chong M, et al. The contribution of alcohol use disorder to decompensated cirrhosis among people with hepatitis C: An international study. J Hepatol. 2018;68(3):393-401. doi:10.1016/j.jhep.2017.10.019
9. Pedersen ER, Davis JP, Fitzke RE, Lee DS, Saba S. American Veterans in the Era of COVID-19: Reactions to the Pandemic, Posttraumatic Stress Disorder, and Substance Use Behaviors. Int J Ment Health Addict. 2023;21(2):767-782. doi:10.1007/s11469-021-00620-0
10. Na PJ, Norman SB, Nichter B, et al. Prevalence, risk and protective factors of alcohol use disorder during the COVID-19 pandemic in U.S. military veterans. Drug Alcohol Depend. 2021;225:108818. doi:10.1016/j.drugalcdep.2021.108818
11. Tsui JI, Williams EC, Green PK, Berry K, Su F, Ioannou GN. Alcohol use and hepatitis C virus treatment outcomes among patients receiving direct antiviral agents. Drug Alcohol Depend. 2016;169:101-109. doi:10.1016/j.drugalcdep.2016.10.021
12. Baum A, Kaboli PJ, Schwartz MD. Reduced In-Person and Increased Telehealth Outpatient Visits During the COVID-19 Pandemic. Ann Intern Med. 2021;174(1):129-131. doi:10.7326/M20-3026
13. Fleming BS, Ifeachor AP, Andres AM, et al. Improving Veteran Access to Treatment for Hepatitis C Virus Infection: Addressing social issues and treatment barriers significantly increases access to HCV care, and many veterans successfully start therapy with the help of additional support staff. Fed Pract. 2017;34(Suppl 4):S24-S28.
14. Belperio PS, Chartier M, Ross DB, Alaigh P, Shulkin D. Curing Hepatitis C Virus Infection: Best Practices From the U.S. Department of Veterans Affairs. Ann Intern Med. 2017;167(7):499-504. doi:10.7326/M17-1073
1. Patel AA, Bui A, Prohl E, et al. Innovations in Hepatitis C Screening and Treatment. Hepatol Commun. 2020;5(3):371-386. Published 2020 Dec 7. doi:10.1002/hep4.1646
2. US Preventive Services Task Force, Owens DK, Davidson KW, et al. Screening for Hepatitis C Virus Infection in Adolescents and Adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2020;323(10):970-975. doi:10.1001/jama.2020.1123
3. Kaufman HW, Bull-Otterson L, Meyer WA 3rd, et al. Decreases in Hepatitis C Testing and Treatment During the COVID-19 Pandemic. Am J Prev Med. 2021;61(3):369-376. doi:10.1016/j.amepre.2021.03.011
4. Rosen CS, Morland LA, Glassman LH, et al. Virtual mental health care in the Veterans Health Administration’s immediate response to coronavirus disease-19. Am Psychol. 2021;76(1):26-38. doi:10.1037/amp0000751
5. Balut MD, Wyte-Lake T, Steers WN, et al. Expansion of telemedicine during COVID-19 at a VA specialty clinic. Healthc (Amst). 2022;10(1):100599. doi:10.1016/j.hjdsi.2021.100599
6. Belperio PS, Chartier M, Ross DB, Alaigh P, Shulkin D. Curing Hepatitis C Virus Infection: Best Practices From the U.S. Department of Veterans Affairs. Ann Intern Med. 2017;167(7):499-504. doi:10.7326/M17-1073
7. Lin M, Kramer J, White D, et al. Barriers to hepatitis C treatment in the era of direct-acting anti-viral agents. Aliment Pharmacol Ther. 2017;46(10):992-1000. doi:10.1111/apt.14328
8. Alavi M, Janjua NZ, Chong M, et al. The contribution of alcohol use disorder to decompensated cirrhosis among people with hepatitis C: An international study. J Hepatol. 2018;68(3):393-401. doi:10.1016/j.jhep.2017.10.019
9. Pedersen ER, Davis JP, Fitzke RE, Lee DS, Saba S. American Veterans in the Era of COVID-19: Reactions to the Pandemic, Posttraumatic Stress Disorder, and Substance Use Behaviors. Int J Ment Health Addict. 2023;21(2):767-782. doi:10.1007/s11469-021-00620-0
10. Na PJ, Norman SB, Nichter B, et al. Prevalence, risk and protective factors of alcohol use disorder during the COVID-19 pandemic in U.S. military veterans. Drug Alcohol Depend. 2021;225:108818. doi:10.1016/j.drugalcdep.2021.108818
11. Tsui JI, Williams EC, Green PK, Berry K, Su F, Ioannou GN. Alcohol use and hepatitis C virus treatment outcomes among patients receiving direct antiviral agents. Drug Alcohol Depend. 2016;169:101-109. doi:10.1016/j.drugalcdep.2016.10.021
12. Baum A, Kaboli PJ, Schwartz MD. Reduced In-Person and Increased Telehealth Outpatient Visits During the COVID-19 Pandemic. Ann Intern Med. 2021;174(1):129-131. doi:10.7326/M20-3026
13. Fleming BS, Ifeachor AP, Andres AM, et al. Improving Veteran Access to Treatment for Hepatitis C Virus Infection: Addressing social issues and treatment barriers significantly increases access to HCV care, and many veterans successfully start therapy with the help of additional support staff. Fed Pract. 2017;34(Suppl 4):S24-S28.
14. Belperio PS, Chartier M, Ross DB, Alaigh P, Shulkin D. Curing Hepatitis C Virus Infection: Best Practices From the U.S. Department of Veterans Affairs. Ann Intern Med. 2017;167(7):499-504. doi:10.7326/M17-1073