User login
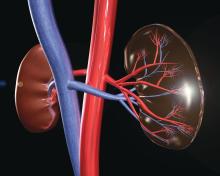
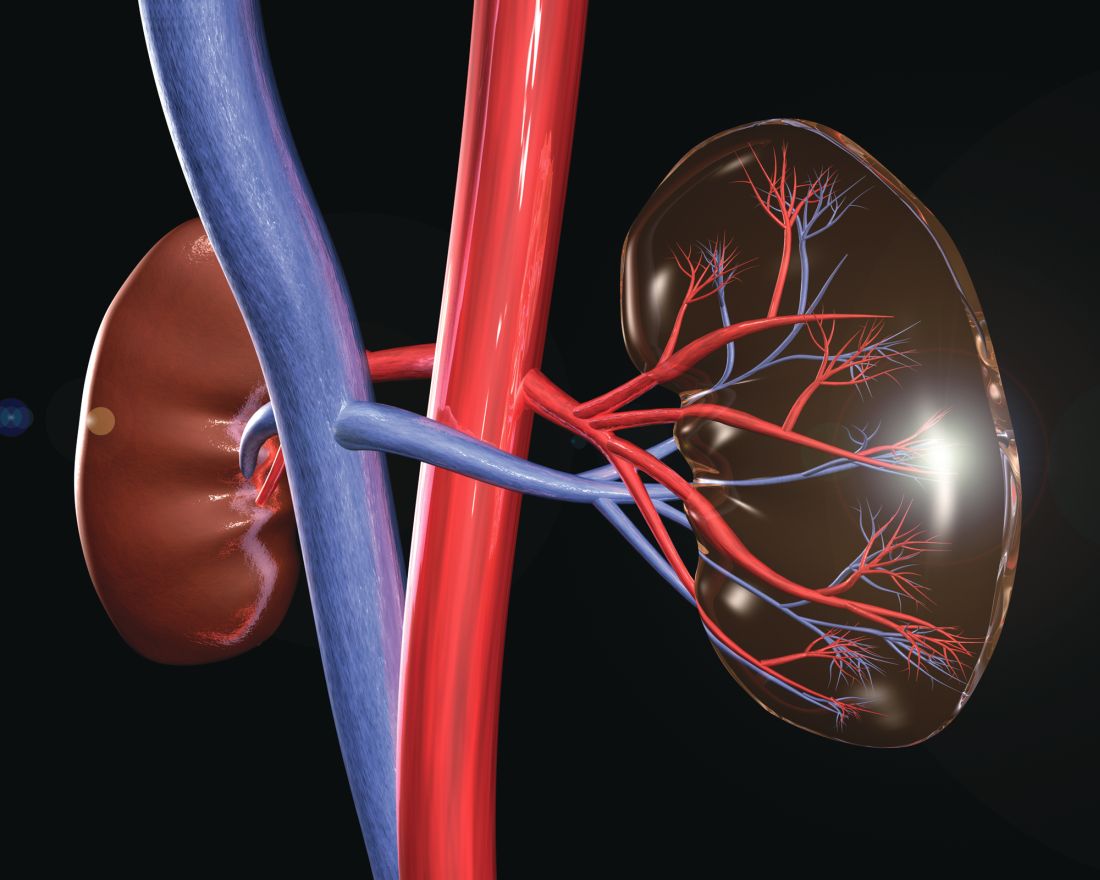
The HOPE in Action Multicenter Kidney Study, the first large-scale clinical trial to study kidney transplantations between people with HIV, has begun at clinical centers across the United States, according to an announcement by the National Institutes of Health. The study, sponsored by the National Institute of Allergy and Infectious Diseases and NIH, will assess the safety of HIV-positive donor to HIV-positive recipient kidney transplantation. This form of transplantation was made legal by the passage of the HOPE (HIV Organ Policy Equity) Act, which permits the transplantation of organs from donors with HIV into qualified HIV-positive recipients with end-stage organ failure.
Outcomes to be studied in the multicenter trial will include potential transplant-related and HIV-related complications following surgery, as well as organ rejection, organ failure, and failure of previously effective HIV medications. The study is currently enrolling and is expected to include 360 participants, with an estimated completion date of Aug. 1, 2022.
“The HOPE Act of 2013 opened the door for researchers to explore a potential new source of donor organs for those living with HIV – a population with a significant and growing need for transplants. This study offers a chance to improve the health of those living with HIV, and increase the overall supply of transplantable organs,” said NIAID Director Anthony S. Fauci, MD, in the NIH news release. Transplantation of organs from HIV-positive donors to HIV-negative recipients remains illegal in the United States.
More information about the study can be found at ClinicalTrials.gov using the identifier NCT03500315. A similar trial to examine liver transplantation, the HOPE in Action Multicenter Liver Study, is expected to launch later in 2018.
The HOPE in Action Multicenter Kidney Study, the first large-scale clinical trial to study kidney transplantations between people with HIV, has begun at clinical centers across the United States, according to an announcement by the National Institutes of Health. The study, sponsored by the National Institute of Allergy and Infectious Diseases and NIH, will assess the safety of HIV-positive donor to HIV-positive recipient kidney transplantation. This form of transplantation was made legal by the passage of the HOPE (HIV Organ Policy Equity) Act, which permits the transplantation of organs from donors with HIV into qualified HIV-positive recipients with end-stage organ failure.
Outcomes to be studied in the multicenter trial will include potential transplant-related and HIV-related complications following surgery, as well as organ rejection, organ failure, and failure of previously effective HIV medications. The study is currently enrolling and is expected to include 360 participants, with an estimated completion date of Aug. 1, 2022.
“The HOPE Act of 2013 opened the door for researchers to explore a potential new source of donor organs for those living with HIV – a population with a significant and growing need for transplants. This study offers a chance to improve the health of those living with HIV, and increase the overall supply of transplantable organs,” said NIAID Director Anthony S. Fauci, MD, in the NIH news release. Transplantation of organs from HIV-positive donors to HIV-negative recipients remains illegal in the United States.
More information about the study can be found at ClinicalTrials.gov using the identifier NCT03500315. A similar trial to examine liver transplantation, the HOPE in Action Multicenter Liver Study, is expected to launch later in 2018.
The HOPE in Action Multicenter Kidney Study, the first large-scale clinical trial to study kidney transplantations between people with HIV, has begun at clinical centers across the United States, according to an announcement by the National Institutes of Health. The study, sponsored by the National Institute of Allergy and Infectious Diseases and NIH, will assess the safety of HIV-positive donor to HIV-positive recipient kidney transplantation. This form of transplantation was made legal by the passage of the HOPE (HIV Organ Policy Equity) Act, which permits the transplantation of organs from donors with HIV into qualified HIV-positive recipients with end-stage organ failure.
Outcomes to be studied in the multicenter trial will include potential transplant-related and HIV-related complications following surgery, as well as organ rejection, organ failure, and failure of previously effective HIV medications. The study is currently enrolling and is expected to include 360 participants, with an estimated completion date of Aug. 1, 2022.
“The HOPE Act of 2013 opened the door for researchers to explore a potential new source of donor organs for those living with HIV – a population with a significant and growing need for transplants. This study offers a chance to improve the health of those living with HIV, and increase the overall supply of transplantable organs,” said NIAID Director Anthony S. Fauci, MD, in the NIH news release. Transplantation of organs from HIV-positive donors to HIV-negative recipients remains illegal in the United States.
More information about the study can be found at ClinicalTrials.gov using the identifier NCT03500315. A similar trial to examine liver transplantation, the HOPE in Action Multicenter Liver Study, is expected to launch later in 2018.