User login
- Use fishing line cut to different lengths (4 cm=10 g; 8 cm=1 g) as substitutes for monofilaments to assist in the diagnosis of diabetic neuropathy. This test is highly specific for neuropathy; using longer lengths of line increases sensitivity.
- Physicians and healthcare providers can use this quick, inexpensive tool for screening neuropathy. Patients can be empowered inexpensively to examine their own feet and reduce the likelihood of developing foot ulcers or amputations.
If there were a less expensive means of reliably performing a standard clinical test in diabetes care, would you want to know about it? If your answer is yes, then the results of this study should be of interest to you. What’s more, your patients can be given the same test to perform at home.
Patients with diabetes have a 15- to 40-times greater risk of leg amputations than those without diabetes, due to loss of protective sensation, ulceration, infection, and gangrene.1-6 Screening for loss of sensation helps prevent foot ulcerations and amputations.
According to the 1988 San Antonio ADA/AAN consensus,7 a diagnosis of diabetic neuropathy (revised in 1992)8 requires that 2 of 4 criteria be met: signs and symptoms, nerve conduction abnormalities, quantitative sensory test (QST) abnormalities, or autonomic test abnormalities. Monofilament testing, part of the QST, can detect loss of pressure sensation on the foot. The 10-g monofilament predicts foot ulcers,9-11 and the 1-g tests sensitivity.12 The reproducibility and predbictive value of monofilaments in identifying diabetes patients at risk for serious limb complications have led the International Diabetes Federation and the World Health Organization to recommend their use.13
Our aim was to demonstrate the utility of 10-g and 1-g monofilaments constructed from fishing line in screening for diabetes neuropathy compared with other QST modalities, including the entire set of monofilaments, in a forced-choice algorithm. We also wanted to show that physicians and patients alike can use this simple, disposable, single-use method for detecting neuropathy.
Methods
We studied 871 subjects (579 normal controls and 292 patients with diagnosed peripheral neuropathy). The study consisted of a 15-item questionnaire that surveyed age, height, weight, race, and diabetes-related issues. We measured random glucose levels with a glucometer to rule out undisclosed hypoglycemia among controls, and performed vibration detection thresholds (VDT) as another confirmatory neuropathy test (values >15 V).
Disposable monofilaments were constructed in our laboratory using commercially available 25-lb “South Bend” brand high-knot-strength fishing line No. M-1425 (South Bend, Inc, North Brook, Ill), which measured 0.020 inches (500 microns) in diameter. We cut the 10-g monofilament to 4 cm in length; the 1-g monofilament to 8 cm. Gripping 1 cm of each monofilament at one end, we used a standard laboratory balance to confirm the buckling force of these lengths. Different brands of fishing line would have to be tested separately for the length needed to create 1- and 10-g monofilaments.
We used a straightforward “yes-no” algorithm in which the subject was asked to identify the presence of sensation correctly on 4 out of 5 trials. One-way analysis of variance (ANOVAR) was used to evaluate interval variables, and likelihood ratio chi-square tests for nominal variables. Statistical significance for all comparisons was accepted at P<.05.
Results
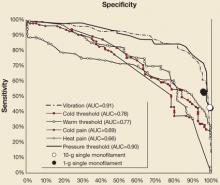
The demographic characteristics of the groups studied are shown in TABLE 1. The sensitivity of the single 10-g and 1-g monofilament was found to be 42.8% and 52.4%, and specificity for each was 99.3% and 96.3%, respectively. TABLE 2 shows the comparison of all of the sensory modalities studied regarding sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV). PPV and NPV were calculated for each modality, with an assumption of disease prevalence of 55% in the diabetic population.14 The 1-g and 10-g monofilaments have very high PPV due to their high specificity (98.7% and 94.6%), but relatively poor NPV (58.7% and 62.3%). The other sensory modalities had good PPV as well (ranging from 81.4% to 91.2%), but a wide range of NPV (53.3% to 75.7%). Lastly, receiver operating characteristic curves (FIGURE) were used to demonstrate the relative value of different sensory modalities, including the single-point values for the 1-g and 10-g monofilaments.
TABLE 1
Epidemiology and group characteristics of the cohort
| CONTROLS | DIABETIC NEUROPATHS | P VALUE | TEST | |
|---|---|---|---|---|
| n | 579 | 292 | ||
| Age (years±SE) | 38.5±0.7 | 61.4±0.8 | <.0001 | ANOVA |
| Gender (M : F) | 200:376 | 169:123 | <.0001 | χ2 |
| Race (n, % of group) | <.0001 | χ2 | ||
| White | 198 (38%) | 226 (87%) | ||
| Black | 274 (53%) | 27 (10%) | ||
| Asian/Pacific Area | 24 (5%) | 1 (<1%) | ||
| Hispanic | 14 (3%) | 2 (1%) | ||
| Native American | 10 (2%) | 0 | ||
| Other | 1 (<1%) | 3 (1%) | ||
| Height | 66.7±0.2 | 67.4±0.3 | .0837 | ANOVA |
| Weight | 168.8±1.9 | 200.8±4.2 | <.0001 | ANOVA |
| Body-mass index | 26.6±0.3 | 30.5±0.7 | <.0001 | ANOVA |
| Random glucose (finger stick) | 95.3±0.8 | 138.2±4.9 | <.0001 | ANOVA |
TABLE 2
Sensitivity and specificity of sensory modalities*
| DIAGNOSTIC CUTOFF LEVEL | SENSITIVITY | SPECIFICITY | PPV† | NPV† | |
|---|---|---|---|---|---|
| 10-g single monofilament | 10 g | 42.8% | 99.3% | 98.7% | 58.7% |
| 1-g single monofilament | 1 g | 52.4% | 96.3% | 94.6% | 62.3% |
| Pressure threshold (multiple monofilaments) | 0.98 g | 76.3% | 90.6% | 90.8% | 75.7% |
| Vibration threshold | 5.1 volts | 76.0% | 91.0% | 91.2% | 75.6% |
| Cold detection threshold | 1.6°C | 44.8% | 90.0% | 84.6% | 57.2% |
| Warm detection threshold | 1.6°C | 46.6% | 92.1% | 87.9% | 58.5% |
| Cold pain threshold | 0.0°C | 35.3% | 90.2% | 81.4% | 53.3% |
| Heat pain threshold | 17.8°C | 35.5% | 90.2% | 81.5% | 53.4% |
| * Includes pressure monofilaments (multiple monofilaments using a forced-choice algorithm), the 10-g and 1-g monofilament, and other sensory modalities. The diagnostic cutoff level for each sensory continuum was set at 90% specificity. | |||||
| † Positive predictive value (PPV) and negative predictive value (NPV) assume presence of diabetes and 55% disease prevalence of neuropathy in the diabetic population. | |||||
FIGURE
Evaluation of neuropathy: ROC curve analysis
Receiver operating characteristics (ROC) of all of the sensory modalities across their spectrum of measurement, expressed here as sensitivity and specificity (instead of the more customary 1–specificity used in signal detection theory) for clinical usefulness. AUC denotes area under the curve for each modality.
Conclusions
The 10-g monofilament is not as sensitive as the 1-g filament, VDT, or the fully quantitative pressure threshold for detecting neuropathy. It is, however, highly specific (99.3%). The 1-g monofilament has nearly the same specificity (96.3%), but has about 10% higher sensitivity at 52.4%. These results support the notion that the 1-g monofilament offers a more sensitive screening tool for diabetic neuropathy than the 10-g filament, without sacrificing specificity.
The use of single monofilaments tested at a single site yielded similar results as a number of other studies that examined multiple testing sites. Studies found that combination tests (2 sites combined) yielded a PPV of only 84%,15 whereas our results with the same 10-g filament at a single site yielded a PPV of 98%, and the 1-gram filament further improved sensitivity with a PPV of 94%.
Some methodological issues are potential sources of error in this study. We constructed filaments with readily available materials and a standardized method. Therefore, we were able to produce large quantities of inexpensive and disposable monofilaments. Studies have shown that the actual buckling force of commercially supplied filaments can vary considerably.16 Therefore, especially with fishing line, be sure to measure and confirm buckling force. Using our technique, we were able to consistently validate the buckling force of each filament.
Furthermore, monofilaments age and lose their compressible strength with repeated use. Our method eliminated this drawback by creating disposable, single-use monofilaments.
In addition, the experience of Litzelman and colleagues17 indicates that giving patients monofilaments encourages self examination, engenders behavioral change, and reduces ulcer rate by almost 60%.
In summary, we found that a single homemade monofilament using fishing line has considerable clinical utility. It is an economical, quick, and simple test to perform at the bedside or even in the field. The 1-g filament is considerably more sensitive than the commonly used 10-g version without sacrificing specificity. Both can be constructed from 25-lb fishing line cut to 8- or 4-cm, respectively. For these reasons we recommend its use as a screening tool for physicians, health care providers, and patients in detecting diabetic neuropathy. We do not, however, recommend that the monofilament be used as a substitute for quantitative tests for vibration and pressure,18,19 both of which are sensitive to early and milder changes in sensory function.
CORRESPONDENCE
Aaron I. Vinik, MD, PhD, Eastern Virginia Medical School, Strelitz Diabetes Institutes 855 W. Brambleton Avenue, Norfolk, VA 23510. E-mail: vinikai@evms.edu
1. Preventive foot care in diabetes. Diabetes Care 2004;27:63S-664.
2. Boulton AJ, Vinik A, Arezzo J, et al. Position statement: diabetic neuropathies. Diabetes Care 2005;28:956-962.
3. Birke JA, Rolfsen RJ. Evaluation of a self-administered sensory testing tool to identify patients at risk of diabetes-related foot problems. Diabetes Care 1998;21:23-25.
4. McGill M, Molyneaux L, Spencer R, Heng LF, Yue DK. Possible sources of discrepancies in the use of the Semmes-Weinstein monofilament. Diabetes Care 1999;22:598-602.
5. Adler AI, Boyko EJ, Ahroni JH, Smith DG. Lower-extremity amputation in diabetes. The independent effects of peripheral vascular disease, sensory neuropathy, and foot ulcers. Diabetes Care 1999;22:1029-1035.
6. Armstrong DG, Lavery LA, Vela SA, Quebedeaux TL, Fleischli JG. Choosing a practical screening instrument to identify patients at risk for diabetic foot ulceration. Arch Intern Med 1998;158:289-292.
7. Consensus State: Report and recommendations of the San Antonio conference on diabetic neuropathy. American Diabetes Association American Academy of Neurology. Diabetes Care 1988;11:592-597.
8. Consensus State: Proceedings of a consensus development conference on standardized measures in diabetic neuropathy. Muscle Nerve 1992;15:1143-1170.
9. McGill M, Molyneaux L, Yue DK. Use of the Semmes-Weinstein 5.07/10 gram monofilament: the long and the short of it. Diabetes Med 1998;15:615-617.
10. Mueller MJ. Identifying patients with diabetes mellitus who are at risk for lower-extremity complications: use of Semmes-Weinstein monofilaments. Phys Ther 1996;76:68-71.
11. Kumar S, Fernando DJ, Veves A, Knowles EA, Young MJ, Boulton AJ. Semmes-Weinstein monofilaments: a simple, effective and inexpensive screening device for identifying diabetic patients at risk of foot ulceration. Diabetes Res Clin Pract 1991;13:63-67.
12. Vinik AI, Suwanwalaikorn S, Stansberry KB, Holland MT, McNitt PM, Colen LE. Quantitative measurement of cutaneous perception in diabetic neuropathy. Muscle Nerve 1995;18:574-584.
13. Krans HH, Perta M, Keen K. The St Vincent declaration action programme. Eurodiabcare 1992; Diabetes Care and Research in Europe. WHO.
14. Pirart J. Diabetes mellitus and its degenerative complications: a prospective study of 4,400 patients observed between 1947 and 1973. Diabetes Care 1978;1:252-263.
15. McGill M, Molyneaux L, Spencer R, Heng LF, Yue DK. Possible sources of discrepancies in the use of the Semmes-Weinstein monofilament. Impact on prevalence of insensate foot and workload requirements. Diabetes Care 1999;22:598-602.
16. McGill M, Molyneaux L, Yue DK. Use of the Semmes-Weinstein 5.07/10 gram monofilament: the long and the short of it. Diabetes Med 1998;15:615-617.
17. Litzelman DK, Marriott DJ, Vinicor F. Independent physiological predictors of foot lesions in patients with NIDDM. Diabetes Care 1997;20:1273-1278.
18. Shearer A, Scuffham P, Gordois A, Oglesby A. Predicted costs and outcomes from reduced vibration detection in people with diabetes in the U.S. Diabetes Care 2003;26:2305-2310.
19. Siao P, Cros DP. Quantitative sensory testing. Phys Med Rehabil Clin N Am 2003;14:261-286.
- Use fishing line cut to different lengths (4 cm=10 g; 8 cm=1 g) as substitutes for monofilaments to assist in the diagnosis of diabetic neuropathy. This test is highly specific for neuropathy; using longer lengths of line increases sensitivity.
- Physicians and healthcare providers can use this quick, inexpensive tool for screening neuropathy. Patients can be empowered inexpensively to examine their own feet and reduce the likelihood of developing foot ulcers or amputations.
If there were a less expensive means of reliably performing a standard clinical test in diabetes care, would you want to know about it? If your answer is yes, then the results of this study should be of interest to you. What’s more, your patients can be given the same test to perform at home.
Patients with diabetes have a 15- to 40-times greater risk of leg amputations than those without diabetes, due to loss of protective sensation, ulceration, infection, and gangrene.1-6 Screening for loss of sensation helps prevent foot ulcerations and amputations.
According to the 1988 San Antonio ADA/AAN consensus,7 a diagnosis of diabetic neuropathy (revised in 1992)8 requires that 2 of 4 criteria be met: signs and symptoms, nerve conduction abnormalities, quantitative sensory test (QST) abnormalities, or autonomic test abnormalities. Monofilament testing, part of the QST, can detect loss of pressure sensation on the foot. The 10-g monofilament predicts foot ulcers,9-11 and the 1-g tests sensitivity.12 The reproducibility and predbictive value of monofilaments in identifying diabetes patients at risk for serious limb complications have led the International Diabetes Federation and the World Health Organization to recommend their use.13
Our aim was to demonstrate the utility of 10-g and 1-g monofilaments constructed from fishing line in screening for diabetes neuropathy compared with other QST modalities, including the entire set of monofilaments, in a forced-choice algorithm. We also wanted to show that physicians and patients alike can use this simple, disposable, single-use method for detecting neuropathy.
Methods
We studied 871 subjects (579 normal controls and 292 patients with diagnosed peripheral neuropathy). The study consisted of a 15-item questionnaire that surveyed age, height, weight, race, and diabetes-related issues. We measured random glucose levels with a glucometer to rule out undisclosed hypoglycemia among controls, and performed vibration detection thresholds (VDT) as another confirmatory neuropathy test (values >15 V).
Disposable monofilaments were constructed in our laboratory using commercially available 25-lb “South Bend” brand high-knot-strength fishing line No. M-1425 (South Bend, Inc, North Brook, Ill), which measured 0.020 inches (500 microns) in diameter. We cut the 10-g monofilament to 4 cm in length; the 1-g monofilament to 8 cm. Gripping 1 cm of each monofilament at one end, we used a standard laboratory balance to confirm the buckling force of these lengths. Different brands of fishing line would have to be tested separately for the length needed to create 1- and 10-g monofilaments.
We used a straightforward “yes-no” algorithm in which the subject was asked to identify the presence of sensation correctly on 4 out of 5 trials. One-way analysis of variance (ANOVAR) was used to evaluate interval variables, and likelihood ratio chi-square tests for nominal variables. Statistical significance for all comparisons was accepted at P<.05.
Results
The demographic characteristics of the groups studied are shown in TABLE 1. The sensitivity of the single 10-g and 1-g monofilament was found to be 42.8% and 52.4%, and specificity for each was 99.3% and 96.3%, respectively. TABLE 2 shows the comparison of all of the sensory modalities studied regarding sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV). PPV and NPV were calculated for each modality, with an assumption of disease prevalence of 55% in the diabetic population.14 The 1-g and 10-g monofilaments have very high PPV due to their high specificity (98.7% and 94.6%), but relatively poor NPV (58.7% and 62.3%). The other sensory modalities had good PPV as well (ranging from 81.4% to 91.2%), but a wide range of NPV (53.3% to 75.7%). Lastly, receiver operating characteristic curves (FIGURE) were used to demonstrate the relative value of different sensory modalities, including the single-point values for the 1-g and 10-g monofilaments.
TABLE 1
Epidemiology and group characteristics of the cohort
| CONTROLS | DIABETIC NEUROPATHS | P VALUE | TEST | |
|---|---|---|---|---|
| n | 579 | 292 | ||
| Age (years±SE) | 38.5±0.7 | 61.4±0.8 | <.0001 | ANOVA |
| Gender (M : F) | 200:376 | 169:123 | <.0001 | χ2 |
| Race (n, % of group) | <.0001 | χ2 | ||
| White | 198 (38%) | 226 (87%) | ||
| Black | 274 (53%) | 27 (10%) | ||
| Asian/Pacific Area | 24 (5%) | 1 (<1%) | ||
| Hispanic | 14 (3%) | 2 (1%) | ||
| Native American | 10 (2%) | 0 | ||
| Other | 1 (<1%) | 3 (1%) | ||
| Height | 66.7±0.2 | 67.4±0.3 | .0837 | ANOVA |
| Weight | 168.8±1.9 | 200.8±4.2 | <.0001 | ANOVA |
| Body-mass index | 26.6±0.3 | 30.5±0.7 | <.0001 | ANOVA |
| Random glucose (finger stick) | 95.3±0.8 | 138.2±4.9 | <.0001 | ANOVA |
TABLE 2
Sensitivity and specificity of sensory modalities*
| DIAGNOSTIC CUTOFF LEVEL | SENSITIVITY | SPECIFICITY | PPV† | NPV† | |
|---|---|---|---|---|---|
| 10-g single monofilament | 10 g | 42.8% | 99.3% | 98.7% | 58.7% |
| 1-g single monofilament | 1 g | 52.4% | 96.3% | 94.6% | 62.3% |
| Pressure threshold (multiple monofilaments) | 0.98 g | 76.3% | 90.6% | 90.8% | 75.7% |
| Vibration threshold | 5.1 volts | 76.0% | 91.0% | 91.2% | 75.6% |
| Cold detection threshold | 1.6°C | 44.8% | 90.0% | 84.6% | 57.2% |
| Warm detection threshold | 1.6°C | 46.6% | 92.1% | 87.9% | 58.5% |
| Cold pain threshold | 0.0°C | 35.3% | 90.2% | 81.4% | 53.3% |
| Heat pain threshold | 17.8°C | 35.5% | 90.2% | 81.5% | 53.4% |
| * Includes pressure monofilaments (multiple monofilaments using a forced-choice algorithm), the 10-g and 1-g monofilament, and other sensory modalities. The diagnostic cutoff level for each sensory continuum was set at 90% specificity. | |||||
| † Positive predictive value (PPV) and negative predictive value (NPV) assume presence of diabetes and 55% disease prevalence of neuropathy in the diabetic population. | |||||
FIGURE
Evaluation of neuropathy: ROC curve analysis
Receiver operating characteristics (ROC) of all of the sensory modalities across their spectrum of measurement, expressed here as sensitivity and specificity (instead of the more customary 1–specificity used in signal detection theory) for clinical usefulness. AUC denotes area under the curve for each modality.
Conclusions
The 10-g monofilament is not as sensitive as the 1-g filament, VDT, or the fully quantitative pressure threshold for detecting neuropathy. It is, however, highly specific (99.3%). The 1-g monofilament has nearly the same specificity (96.3%), but has about 10% higher sensitivity at 52.4%. These results support the notion that the 1-g monofilament offers a more sensitive screening tool for diabetic neuropathy than the 10-g filament, without sacrificing specificity.
The use of single monofilaments tested at a single site yielded similar results as a number of other studies that examined multiple testing sites. Studies found that combination tests (2 sites combined) yielded a PPV of only 84%,15 whereas our results with the same 10-g filament at a single site yielded a PPV of 98%, and the 1-gram filament further improved sensitivity with a PPV of 94%.
Some methodological issues are potential sources of error in this study. We constructed filaments with readily available materials and a standardized method. Therefore, we were able to produce large quantities of inexpensive and disposable monofilaments. Studies have shown that the actual buckling force of commercially supplied filaments can vary considerably.16 Therefore, especially with fishing line, be sure to measure and confirm buckling force. Using our technique, we were able to consistently validate the buckling force of each filament.
Furthermore, monofilaments age and lose their compressible strength with repeated use. Our method eliminated this drawback by creating disposable, single-use monofilaments.
In addition, the experience of Litzelman and colleagues17 indicates that giving patients monofilaments encourages self examination, engenders behavioral change, and reduces ulcer rate by almost 60%.
In summary, we found that a single homemade monofilament using fishing line has considerable clinical utility. It is an economical, quick, and simple test to perform at the bedside or even in the field. The 1-g filament is considerably more sensitive than the commonly used 10-g version without sacrificing specificity. Both can be constructed from 25-lb fishing line cut to 8- or 4-cm, respectively. For these reasons we recommend its use as a screening tool for physicians, health care providers, and patients in detecting diabetic neuropathy. We do not, however, recommend that the monofilament be used as a substitute for quantitative tests for vibration and pressure,18,19 both of which are sensitive to early and milder changes in sensory function.
CORRESPONDENCE
Aaron I. Vinik, MD, PhD, Eastern Virginia Medical School, Strelitz Diabetes Institutes 855 W. Brambleton Avenue, Norfolk, VA 23510. E-mail: vinikai@evms.edu
- Use fishing line cut to different lengths (4 cm=10 g; 8 cm=1 g) as substitutes for monofilaments to assist in the diagnosis of diabetic neuropathy. This test is highly specific for neuropathy; using longer lengths of line increases sensitivity.
- Physicians and healthcare providers can use this quick, inexpensive tool for screening neuropathy. Patients can be empowered inexpensively to examine their own feet and reduce the likelihood of developing foot ulcers or amputations.
If there were a less expensive means of reliably performing a standard clinical test in diabetes care, would you want to know about it? If your answer is yes, then the results of this study should be of interest to you. What’s more, your patients can be given the same test to perform at home.
Patients with diabetes have a 15- to 40-times greater risk of leg amputations than those without diabetes, due to loss of protective sensation, ulceration, infection, and gangrene.1-6 Screening for loss of sensation helps prevent foot ulcerations and amputations.
According to the 1988 San Antonio ADA/AAN consensus,7 a diagnosis of diabetic neuropathy (revised in 1992)8 requires that 2 of 4 criteria be met: signs and symptoms, nerve conduction abnormalities, quantitative sensory test (QST) abnormalities, or autonomic test abnormalities. Monofilament testing, part of the QST, can detect loss of pressure sensation on the foot. The 10-g monofilament predicts foot ulcers,9-11 and the 1-g tests sensitivity.12 The reproducibility and predbictive value of monofilaments in identifying diabetes patients at risk for serious limb complications have led the International Diabetes Federation and the World Health Organization to recommend their use.13
Our aim was to demonstrate the utility of 10-g and 1-g monofilaments constructed from fishing line in screening for diabetes neuropathy compared with other QST modalities, including the entire set of monofilaments, in a forced-choice algorithm. We also wanted to show that physicians and patients alike can use this simple, disposable, single-use method for detecting neuropathy.
Methods
We studied 871 subjects (579 normal controls and 292 patients with diagnosed peripheral neuropathy). The study consisted of a 15-item questionnaire that surveyed age, height, weight, race, and diabetes-related issues. We measured random glucose levels with a glucometer to rule out undisclosed hypoglycemia among controls, and performed vibration detection thresholds (VDT) as another confirmatory neuropathy test (values >15 V).
Disposable monofilaments were constructed in our laboratory using commercially available 25-lb “South Bend” brand high-knot-strength fishing line No. M-1425 (South Bend, Inc, North Brook, Ill), which measured 0.020 inches (500 microns) in diameter. We cut the 10-g monofilament to 4 cm in length; the 1-g monofilament to 8 cm. Gripping 1 cm of each monofilament at one end, we used a standard laboratory balance to confirm the buckling force of these lengths. Different brands of fishing line would have to be tested separately for the length needed to create 1- and 10-g monofilaments.
We used a straightforward “yes-no” algorithm in which the subject was asked to identify the presence of sensation correctly on 4 out of 5 trials. One-way analysis of variance (ANOVAR) was used to evaluate interval variables, and likelihood ratio chi-square tests for nominal variables. Statistical significance for all comparisons was accepted at P<.05.
Results
The demographic characteristics of the groups studied are shown in TABLE 1. The sensitivity of the single 10-g and 1-g monofilament was found to be 42.8% and 52.4%, and specificity for each was 99.3% and 96.3%, respectively. TABLE 2 shows the comparison of all of the sensory modalities studied regarding sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV). PPV and NPV were calculated for each modality, with an assumption of disease prevalence of 55% in the diabetic population.14 The 1-g and 10-g monofilaments have very high PPV due to their high specificity (98.7% and 94.6%), but relatively poor NPV (58.7% and 62.3%). The other sensory modalities had good PPV as well (ranging from 81.4% to 91.2%), but a wide range of NPV (53.3% to 75.7%). Lastly, receiver operating characteristic curves (FIGURE) were used to demonstrate the relative value of different sensory modalities, including the single-point values for the 1-g and 10-g monofilaments.
TABLE 1
Epidemiology and group characteristics of the cohort
| CONTROLS | DIABETIC NEUROPATHS | P VALUE | TEST | |
|---|---|---|---|---|
| n | 579 | 292 | ||
| Age (years±SE) | 38.5±0.7 | 61.4±0.8 | <.0001 | ANOVA |
| Gender (M : F) | 200:376 | 169:123 | <.0001 | χ2 |
| Race (n, % of group) | <.0001 | χ2 | ||
| White | 198 (38%) | 226 (87%) | ||
| Black | 274 (53%) | 27 (10%) | ||
| Asian/Pacific Area | 24 (5%) | 1 (<1%) | ||
| Hispanic | 14 (3%) | 2 (1%) | ||
| Native American | 10 (2%) | 0 | ||
| Other | 1 (<1%) | 3 (1%) | ||
| Height | 66.7±0.2 | 67.4±0.3 | .0837 | ANOVA |
| Weight | 168.8±1.9 | 200.8±4.2 | <.0001 | ANOVA |
| Body-mass index | 26.6±0.3 | 30.5±0.7 | <.0001 | ANOVA |
| Random glucose (finger stick) | 95.3±0.8 | 138.2±4.9 | <.0001 | ANOVA |
TABLE 2
Sensitivity and specificity of sensory modalities*
| DIAGNOSTIC CUTOFF LEVEL | SENSITIVITY | SPECIFICITY | PPV† | NPV† | |
|---|---|---|---|---|---|
| 10-g single monofilament | 10 g | 42.8% | 99.3% | 98.7% | 58.7% |
| 1-g single monofilament | 1 g | 52.4% | 96.3% | 94.6% | 62.3% |
| Pressure threshold (multiple monofilaments) | 0.98 g | 76.3% | 90.6% | 90.8% | 75.7% |
| Vibration threshold | 5.1 volts | 76.0% | 91.0% | 91.2% | 75.6% |
| Cold detection threshold | 1.6°C | 44.8% | 90.0% | 84.6% | 57.2% |
| Warm detection threshold | 1.6°C | 46.6% | 92.1% | 87.9% | 58.5% |
| Cold pain threshold | 0.0°C | 35.3% | 90.2% | 81.4% | 53.3% |
| Heat pain threshold | 17.8°C | 35.5% | 90.2% | 81.5% | 53.4% |
| * Includes pressure monofilaments (multiple monofilaments using a forced-choice algorithm), the 10-g and 1-g monofilament, and other sensory modalities. The diagnostic cutoff level for each sensory continuum was set at 90% specificity. | |||||
| † Positive predictive value (PPV) and negative predictive value (NPV) assume presence of diabetes and 55% disease prevalence of neuropathy in the diabetic population. | |||||
FIGURE
Evaluation of neuropathy: ROC curve analysis
Receiver operating characteristics (ROC) of all of the sensory modalities across their spectrum of measurement, expressed here as sensitivity and specificity (instead of the more customary 1–specificity used in signal detection theory) for clinical usefulness. AUC denotes area under the curve for each modality.
Conclusions
The 10-g monofilament is not as sensitive as the 1-g filament, VDT, or the fully quantitative pressure threshold for detecting neuropathy. It is, however, highly specific (99.3%). The 1-g monofilament has nearly the same specificity (96.3%), but has about 10% higher sensitivity at 52.4%. These results support the notion that the 1-g monofilament offers a more sensitive screening tool for diabetic neuropathy than the 10-g filament, without sacrificing specificity.
The use of single monofilaments tested at a single site yielded similar results as a number of other studies that examined multiple testing sites. Studies found that combination tests (2 sites combined) yielded a PPV of only 84%,15 whereas our results with the same 10-g filament at a single site yielded a PPV of 98%, and the 1-gram filament further improved sensitivity with a PPV of 94%.
Some methodological issues are potential sources of error in this study. We constructed filaments with readily available materials and a standardized method. Therefore, we were able to produce large quantities of inexpensive and disposable monofilaments. Studies have shown that the actual buckling force of commercially supplied filaments can vary considerably.16 Therefore, especially with fishing line, be sure to measure and confirm buckling force. Using our technique, we were able to consistently validate the buckling force of each filament.
Furthermore, monofilaments age and lose their compressible strength with repeated use. Our method eliminated this drawback by creating disposable, single-use monofilaments.
In addition, the experience of Litzelman and colleagues17 indicates that giving patients monofilaments encourages self examination, engenders behavioral change, and reduces ulcer rate by almost 60%.
In summary, we found that a single homemade monofilament using fishing line has considerable clinical utility. It is an economical, quick, and simple test to perform at the bedside or even in the field. The 1-g filament is considerably more sensitive than the commonly used 10-g version without sacrificing specificity. Both can be constructed from 25-lb fishing line cut to 8- or 4-cm, respectively. For these reasons we recommend its use as a screening tool for physicians, health care providers, and patients in detecting diabetic neuropathy. We do not, however, recommend that the monofilament be used as a substitute for quantitative tests for vibration and pressure,18,19 both of which are sensitive to early and milder changes in sensory function.
CORRESPONDENCE
Aaron I. Vinik, MD, PhD, Eastern Virginia Medical School, Strelitz Diabetes Institutes 855 W. Brambleton Avenue, Norfolk, VA 23510. E-mail: vinikai@evms.edu
1. Preventive foot care in diabetes. Diabetes Care 2004;27:63S-664.
2. Boulton AJ, Vinik A, Arezzo J, et al. Position statement: diabetic neuropathies. Diabetes Care 2005;28:956-962.
3. Birke JA, Rolfsen RJ. Evaluation of a self-administered sensory testing tool to identify patients at risk of diabetes-related foot problems. Diabetes Care 1998;21:23-25.
4. McGill M, Molyneaux L, Spencer R, Heng LF, Yue DK. Possible sources of discrepancies in the use of the Semmes-Weinstein monofilament. Diabetes Care 1999;22:598-602.
5. Adler AI, Boyko EJ, Ahroni JH, Smith DG. Lower-extremity amputation in diabetes. The independent effects of peripheral vascular disease, sensory neuropathy, and foot ulcers. Diabetes Care 1999;22:1029-1035.
6. Armstrong DG, Lavery LA, Vela SA, Quebedeaux TL, Fleischli JG. Choosing a practical screening instrument to identify patients at risk for diabetic foot ulceration. Arch Intern Med 1998;158:289-292.
7. Consensus State: Report and recommendations of the San Antonio conference on diabetic neuropathy. American Diabetes Association American Academy of Neurology. Diabetes Care 1988;11:592-597.
8. Consensus State: Proceedings of a consensus development conference on standardized measures in diabetic neuropathy. Muscle Nerve 1992;15:1143-1170.
9. McGill M, Molyneaux L, Yue DK. Use of the Semmes-Weinstein 5.07/10 gram monofilament: the long and the short of it. Diabetes Med 1998;15:615-617.
10. Mueller MJ. Identifying patients with diabetes mellitus who are at risk for lower-extremity complications: use of Semmes-Weinstein monofilaments. Phys Ther 1996;76:68-71.
11. Kumar S, Fernando DJ, Veves A, Knowles EA, Young MJ, Boulton AJ. Semmes-Weinstein monofilaments: a simple, effective and inexpensive screening device for identifying diabetic patients at risk of foot ulceration. Diabetes Res Clin Pract 1991;13:63-67.
12. Vinik AI, Suwanwalaikorn S, Stansberry KB, Holland MT, McNitt PM, Colen LE. Quantitative measurement of cutaneous perception in diabetic neuropathy. Muscle Nerve 1995;18:574-584.
13. Krans HH, Perta M, Keen K. The St Vincent declaration action programme. Eurodiabcare 1992; Diabetes Care and Research in Europe. WHO.
14. Pirart J. Diabetes mellitus and its degenerative complications: a prospective study of 4,400 patients observed between 1947 and 1973. Diabetes Care 1978;1:252-263.
15. McGill M, Molyneaux L, Spencer R, Heng LF, Yue DK. Possible sources of discrepancies in the use of the Semmes-Weinstein monofilament. Impact on prevalence of insensate foot and workload requirements. Diabetes Care 1999;22:598-602.
16. McGill M, Molyneaux L, Yue DK. Use of the Semmes-Weinstein 5.07/10 gram monofilament: the long and the short of it. Diabetes Med 1998;15:615-617.
17. Litzelman DK, Marriott DJ, Vinicor F. Independent physiological predictors of foot lesions in patients with NIDDM. Diabetes Care 1997;20:1273-1278.
18. Shearer A, Scuffham P, Gordois A, Oglesby A. Predicted costs and outcomes from reduced vibration detection in people with diabetes in the U.S. Diabetes Care 2003;26:2305-2310.
19. Siao P, Cros DP. Quantitative sensory testing. Phys Med Rehabil Clin N Am 2003;14:261-286.
1. Preventive foot care in diabetes. Diabetes Care 2004;27:63S-664.
2. Boulton AJ, Vinik A, Arezzo J, et al. Position statement: diabetic neuropathies. Diabetes Care 2005;28:956-962.
3. Birke JA, Rolfsen RJ. Evaluation of a self-administered sensory testing tool to identify patients at risk of diabetes-related foot problems. Diabetes Care 1998;21:23-25.
4. McGill M, Molyneaux L, Spencer R, Heng LF, Yue DK. Possible sources of discrepancies in the use of the Semmes-Weinstein monofilament. Diabetes Care 1999;22:598-602.
5. Adler AI, Boyko EJ, Ahroni JH, Smith DG. Lower-extremity amputation in diabetes. The independent effects of peripheral vascular disease, sensory neuropathy, and foot ulcers. Diabetes Care 1999;22:1029-1035.
6. Armstrong DG, Lavery LA, Vela SA, Quebedeaux TL, Fleischli JG. Choosing a practical screening instrument to identify patients at risk for diabetic foot ulceration. Arch Intern Med 1998;158:289-292.
7. Consensus State: Report and recommendations of the San Antonio conference on diabetic neuropathy. American Diabetes Association American Academy of Neurology. Diabetes Care 1988;11:592-597.
8. Consensus State: Proceedings of a consensus development conference on standardized measures in diabetic neuropathy. Muscle Nerve 1992;15:1143-1170.
9. McGill M, Molyneaux L, Yue DK. Use of the Semmes-Weinstein 5.07/10 gram monofilament: the long and the short of it. Diabetes Med 1998;15:615-617.
10. Mueller MJ. Identifying patients with diabetes mellitus who are at risk for lower-extremity complications: use of Semmes-Weinstein monofilaments. Phys Ther 1996;76:68-71.
11. Kumar S, Fernando DJ, Veves A, Knowles EA, Young MJ, Boulton AJ. Semmes-Weinstein monofilaments: a simple, effective and inexpensive screening device for identifying diabetic patients at risk of foot ulceration. Diabetes Res Clin Pract 1991;13:63-67.
12. Vinik AI, Suwanwalaikorn S, Stansberry KB, Holland MT, McNitt PM, Colen LE. Quantitative measurement of cutaneous perception in diabetic neuropathy. Muscle Nerve 1995;18:574-584.
13. Krans HH, Perta M, Keen K. The St Vincent declaration action programme. Eurodiabcare 1992; Diabetes Care and Research in Europe. WHO.
14. Pirart J. Diabetes mellitus and its degenerative complications: a prospective study of 4,400 patients observed between 1947 and 1973. Diabetes Care 1978;1:252-263.
15. McGill M, Molyneaux L, Spencer R, Heng LF, Yue DK. Possible sources of discrepancies in the use of the Semmes-Weinstein monofilament. Impact on prevalence of insensate foot and workload requirements. Diabetes Care 1999;22:598-602.
16. McGill M, Molyneaux L, Yue DK. Use of the Semmes-Weinstein 5.07/10 gram monofilament: the long and the short of it. Diabetes Med 1998;15:615-617.
17. Litzelman DK, Marriott DJ, Vinicor F. Independent physiological predictors of foot lesions in patients with NIDDM. Diabetes Care 1997;20:1273-1278.
18. Shearer A, Scuffham P, Gordois A, Oglesby A. Predicted costs and outcomes from reduced vibration detection in people with diabetes in the U.S. Diabetes Care 2003;26:2305-2310.
19. Siao P, Cros DP. Quantitative sensory testing. Phys Med Rehabil Clin N Am 2003;14:261-286.