User login
Capturing Pathology Workload Associated With Precision Oncology
Precision oncology (PO) is cancer treatment individualized to the special characteristics of a patient’s tumor. It has become standard care for most patients with advanced cancer. Advances in molecular cell biology and immunology have identified numerous targets and many therapies have been developed as a result. Molecular testing and targeted therapy are typically covered by insurance, even when inflation-adjusted price growth is considered.1 Barriers remain, however, and pathologists are uniquely qualified to address some of the challenges.2
Most US laboratories do not perform molecular diagnostic tests for PO, particularly comprehensive evaluation of multiple targets by next-generation sequencing, or other techniques. Instead, these tests are sent to reference laboratories. The workload associated with referral testing is an obstacle to increased use of such tests. Despite guideline recommendations, a minority of indicated tests are performed.3 This is true even when testing costs are covered by clinical trials or grants, such as those in the Veterans Health Administration (VHA).
The main characteristic of successful PO programs is a multidisciplinary commitment, including pathology involvement in molecular tumor boards and assistance with test selection, tissue collection, and result interpretation.2 This, however, adds to the workload for the pathology department, an underappreciated phenomenon in the context of pathology workforce shortages.4
Workforce shortages impact all occupations in the laboratory setting. Though the shortage of medical technologists in clinical pathology laboratories has repeatedly been identified as critical at the VHA as well as in the private sector, the same cannot be said for staff shortages in anatomic pathology laboratories. Thus, the hospital laboratory divisions are concerned with biopsy or resection tissue specimens as opposed to the bodily fluid specimens that predominate in clinical laboratories.5 The lack of accurate data on histopathology technicians and technologists has precluded the degree of recognition seen for medical technologists. In labor statistics, these occupations are often obscured by inclusion with other jobs in broad categories such as medical and clinical laboratory technologists and technicians.6 Vacancy—the principal metric used to assess medical laboratory workforce shortage—fails to account for positions that are eventually eliminated after remaining vacant for prolonged periods, positions not replaced as a result of ambitious efficiency measures, or positions that were never created due to insufficient funding, reasons for administrative disapproval, or coverage by laboratory professionals working extra shifts or second jobs.7
Increased demand for pathologists is suggested by a 42% increase in workload per pathologist over the last decade, while a shortage is suggested by decreases in absolute and population-adjusted numbers of pathologists.8,9 An influx of pathologists is not an expected remedy due to the global decline in medical graduates pursuing careers in the field.8
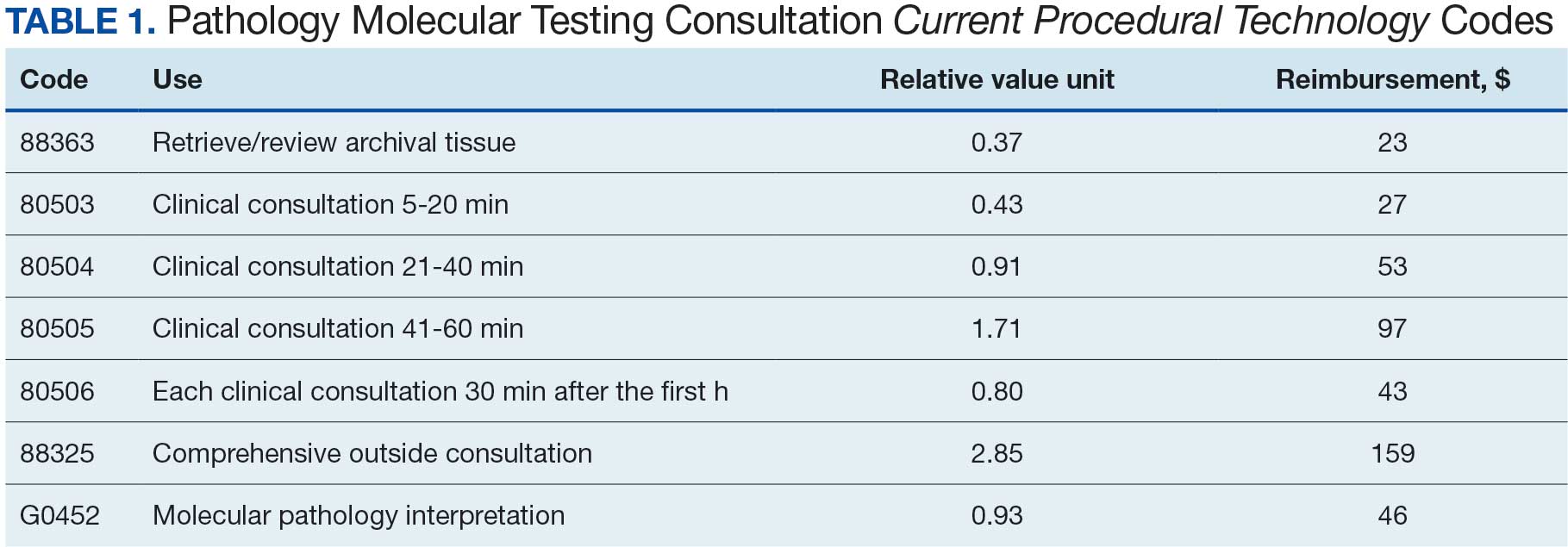
Approximations for required labor and potential revenue generation are necessary to justify creation of pathology positions. This work mostly has gone uncaptured due to the limitations of Current Procedural Terminology (CPT) codes. Few laboratories have consistently used the 88363, 88325, and G0452 CPT codes. The pathology clinical consultation CPT codes (80503-80506) released in 2022 enhance acquisition of this work. The new codes replace 80500 and 80502 and allow for more precise identification of any work requiring medical judgment that a pathologist does at the request of another qualified health care professional (HCP) or as required by federal or state regulation.
The codes can be used to bill for associated time spent reviewing health records, communicating with other HCPs, placing orders, and documentation. An HCP can bill according to level of medical decision-making (MDM) or time spent by the consulting pathologist. Code 80503 can be billed for 5 to 20 minutes of a pathologist's time, 80504 for 21 to 40 minutes, 80505 for 41 to 60 minutes, and 80506 for each additional 30 minutes after the first hour. Levels of MDM (low, moderate, and high) are defined as for other evaluation and management services. A consultation report must be generated and contain documentation of the consultation request, pathologist interpretation, and justification for the level of service associated with the chosen code. Relative value units (RVUs) and reimbursement associated with each as well as other consultation-related codes are available in Table 1.
This article outlines how the pathology time investment associated with anatomic pathology molecular testing at the Kansas City Veterans Affairs Medical Center (KCVAMC) can be captured using the consultation process and new CPT codes. Staff included 4 pathologists, 3 histotechnologists, 1 histology supervisor, 1 grossing room technician, and 1 cytotechnologist, 1 cytology technician.
METHODS
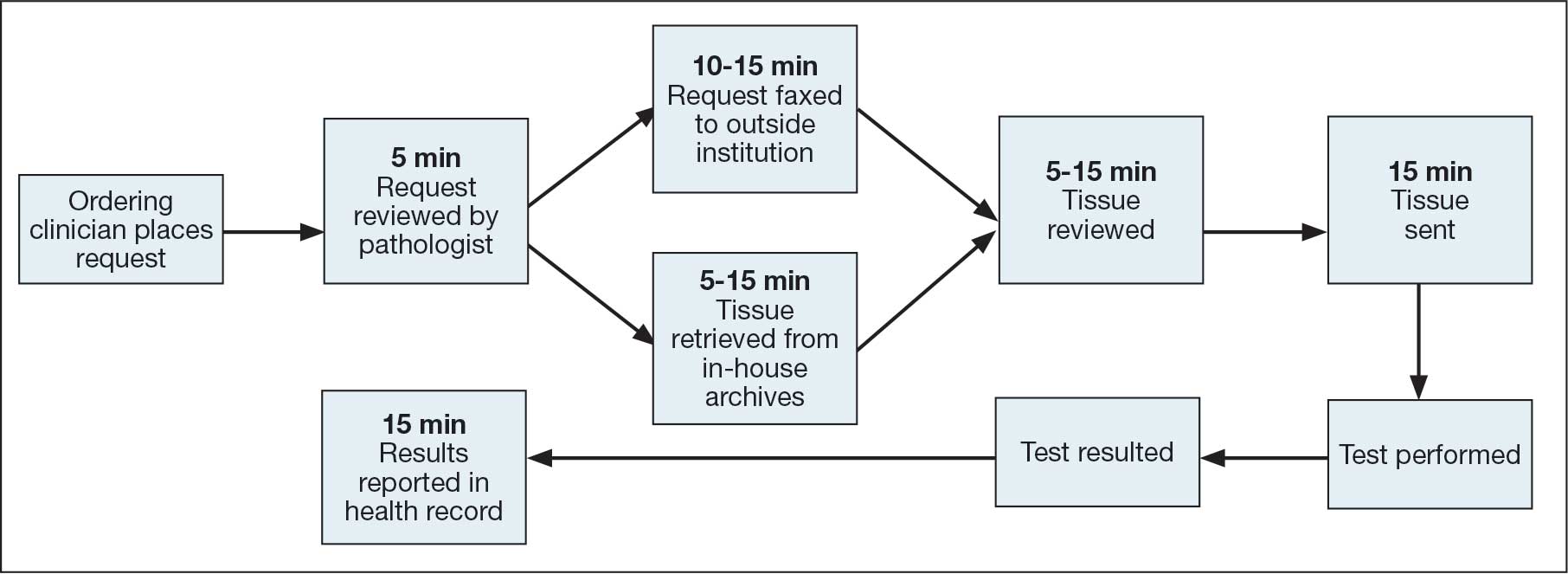
The AP molecular testing consultation process at the KCVAMC was mapped, with the average time measured for each step (Figure). AP records for 2021 were reviewed to determine the number of AP molecular send out tests. Cumulative time investment was calculated in hours and a theoretical number of RVUs was calculated using the new pathology clinical consultation CPT codes (80503-80506). This theoretical number of RVUs was compared with the total AP RVUs generated in 2021 to determine a potential increase in RVUs with use of the new CPT codes to capture pathology work associated with AP molecular testing consultations.
RESULTS
From 2021 to 2023, there were 21,021 AP cases at the KCVAMC. Basal cell carcinomas and squamous cell carcinomas of the skin were excluded because they comprise most cancer cases but almost never necessitate AP molecular test consultations. A total of 2118 cancer cases were included, representing 10.1% of all cases. Ancillary AP molecular send-out tests were performed on 1338 (6.4%) cases. Since ancillary tissue tests are requested by consultation at the KCVAMC, this resulted in 1338 consultations (Table 2).
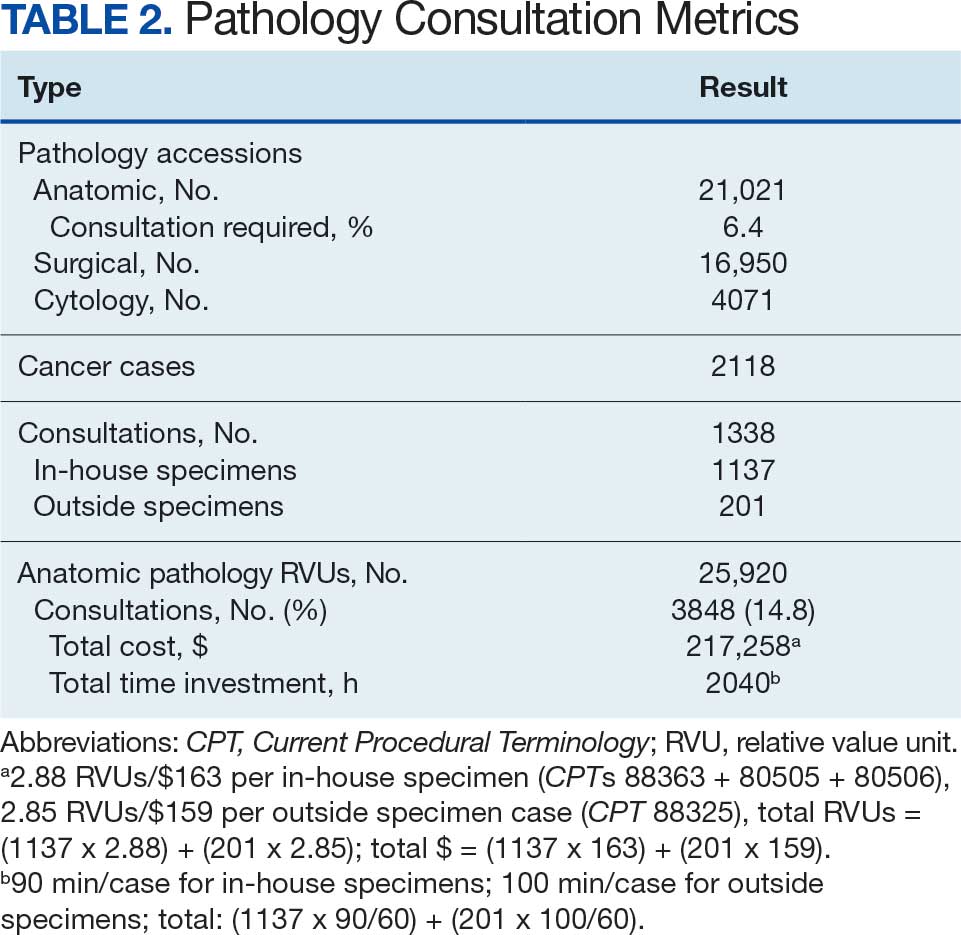
The time to complete a consultation was measured by calculating the mean time required to complete each step (Table 3). With in-house specimen consultations requiring 90 minutes each and outside specimen consultations requiring 100 minutes each, a total of 2040 hours of pathology staff time was necessary to complete associated consultations. Billing for this time with the new pathology clinical consultation CPT codes would generate 3847 RVUs, which would have equated to 14.8% (3847/25,920) of the anatomic pathology RVUs.

DISCUSSION
When considering the lengths of time for tasks associated with each consultation, it is important to remember that the volume (2-3 daily), was insufficient to meaningfully benefit from batching. Thus, waiting to perform a particular task until it was needed for multiple cases reduced the inefficiency associated with starting and switching between tasks. Both the Computerized Patient Record System and VistA had to be reopened, reauthenticated, and reloaded for each step that required use of the health record, printer, or fax machine. Faxes also required waiting for transmission and printed confirmation of successful transmission. As a result, the time values denoted for each step are likely underestimated, as the effect of interruptions is significant and not reflected in the estimates recorded.10
This analysis has demonstrated that PO entails a significant amount of work for pathology departments. To determine and maintain appropriate staffing models, this work must be captured and reimbursed. Unlike other pathology work, which is performed in-house and reimbursed for the associated test, a significant proportion of PO testing is sent out. Even if more reliable assays are developed, the physical processes of sending out samples and reporting test results cannot be outsourced. Independent and commensurate reimbursement methods are necessary to allow for this work and PO.
CMS included new pathology clinical consultation codes that may be used to bill for some of this work according to the 2022 physician fee schedule due to advocacy work by the College of American Pathologists and the American Medical Association CPT editorial panel.11
CONCLUSIONS
This analysis found that adoption of PO may present a significant amount of additional work for pathology departments. To determine and maintain appropriate staffing models, work completed by pathologists in this manner must be recorded and reimbursed. Pathologists need to be trained and encouraged to use these CPT codes and bill for the work described in this article. The increased revenue will allow for additional positions to alleviate the burdens imposed by understaffing so that pathology can function as a facilitator of PO rather than as a barrier to it.
- Wilson LE, Greiner MA, Altomare I, et al. Rapid rise in the cost of targeted cancer therapies for Medicare patients with solid tumors from 2006 to 2015. J Geriatr Oncol. 2021;12:375-380. doi:10.1016/j.jgo.2020.11.007
- Ersek JL, Black LJ, Thompson MA, et al. Implementing precision medicine programs and clinical trials in the community-based oncology practice: barriers and best practices. Am Soc Clin Oncol Educ Book. 2018;38:188-196. doi:10.1200/EDBK_200633
- Inal C, Yilmaz E, Cheng H, et al. Effect of reflex testing by pathologists on molecular testing rates in lung cancer patients: experience from a community-based academic center. J Clin Oncol. 2014;32:8098. doi:10.1200/jco.2014.32.15_suppl.8098
- Robboy SJ, Gupta S, Crawford JM, et al. The pathologist workforce in the United States: II. an interactive modeling tool for analyzing future qualitative and quantitative staffing demands for services. Arch Pathol Lab Med. 2015;139:1413-1430. doi:10.5858/arpa.2014-0559-OA
- OIG determination of Veterans Health Administration’s occupational staffing shortages fiscal year 2021. Department of Veterans Affairs OIG. September 28, 2021. Accessed January 30, 2026. https://www.oversight.gov/report/VA/OIG-determination-veterans-health-administrations-occupational-staffing-shortages-fiscal
- Zanto S, Cremeans L, Deutsch-Keahey D, et al. Addressing the clinical laboratory workforce shortage. The American Society for Clinical Laboratory Science. July 2, 2020. Accessed January 30, 2026. https://ascls.org/addressing-the-clinical-laboratory-workforce-shortage/
- Bennett A, Garcia E, Schulze M, et al. Building a laboratory workforce to meet the future: ASCP Task Force on the Laboratory Professionals Workforce. Am J Clin Pathol. 2014;141:154-167. doi:10.1309/AJCPIV2OG8TEGHHZ
- Fielder T, Watts F, Howden C, et al. Why choose a pathology career? Arch Pathol Lab Med. 2022;146:903-910. doi:10.5858/arpa.2021-0118-OA
- Metter DM, Colgan TJ, Leung ST, et al. Trends in the US and Canadian pathologist workforces from 2007 to 2017. JAMA Netw Open. 2019;2:e194337. doi:10.1001/jamanetworkopen.2019.4337
- Schulte B. Work interruptions can cost you 6 hours a day. An efficiency expert explains how to avoid them. The Washington Post. June 1, 2015. Accessed January 30, 2026. https://www.washingtonpost.com/news/inspired-life/wp/2015/06/01/interruptions-at-work-can-cost-you-up-to-6-hours-a-day-heres-how-to-avoid-them/
- Fiegl C. Medicare adopts new clinical consult billing codes. College of American Pathologists Today. December 2021. Accessed January 30, 2026. https://www.captodayonline.com/medicare-adopts-new-clinical-consult-billing-code
Precision oncology (PO) is cancer treatment individualized to the special characteristics of a patient’s tumor. It has become standard care for most patients with advanced cancer. Advances in molecular cell biology and immunology have identified numerous targets and many therapies have been developed as a result. Molecular testing and targeted therapy are typically covered by insurance, even when inflation-adjusted price growth is considered.1 Barriers remain, however, and pathologists are uniquely qualified to address some of the challenges.2
Most US laboratories do not perform molecular diagnostic tests for PO, particularly comprehensive evaluation of multiple targets by next-generation sequencing, or other techniques. Instead, these tests are sent to reference laboratories. The workload associated with referral testing is an obstacle to increased use of such tests. Despite guideline recommendations, a minority of indicated tests are performed.3 This is true even when testing costs are covered by clinical trials or grants, such as those in the Veterans Health Administration (VHA).
The main characteristic of successful PO programs is a multidisciplinary commitment, including pathology involvement in molecular tumor boards and assistance with test selection, tissue collection, and result interpretation.2 This, however, adds to the workload for the pathology department, an underappreciated phenomenon in the context of pathology workforce shortages.4
Workforce shortages impact all occupations in the laboratory setting. Though the shortage of medical technologists in clinical pathology laboratories has repeatedly been identified as critical at the VHA as well as in the private sector, the same cannot be said for staff shortages in anatomic pathology laboratories. Thus, the hospital laboratory divisions are concerned with biopsy or resection tissue specimens as opposed to the bodily fluid specimens that predominate in clinical laboratories.5 The lack of accurate data on histopathology technicians and technologists has precluded the degree of recognition seen for medical technologists. In labor statistics, these occupations are often obscured by inclusion with other jobs in broad categories such as medical and clinical laboratory technologists and technicians.6 Vacancy—the principal metric used to assess medical laboratory workforce shortage—fails to account for positions that are eventually eliminated after remaining vacant for prolonged periods, positions not replaced as a result of ambitious efficiency measures, or positions that were never created due to insufficient funding, reasons for administrative disapproval, or coverage by laboratory professionals working extra shifts or second jobs.7
Increased demand for pathologists is suggested by a 42% increase in workload per pathologist over the last decade, while a shortage is suggested by decreases in absolute and population-adjusted numbers of pathologists.8,9 An influx of pathologists is not an expected remedy due to the global decline in medical graduates pursuing careers in the field.8
Approximations for required labor and potential revenue generation are necessary to justify creation of pathology positions. This work mostly has gone uncaptured due to the limitations of Current Procedural Terminology (CPT) codes. Few laboratories have consistently used the 88363, 88325, and G0452 CPT codes. The pathology clinical consultation CPT codes (80503-80506) released in 2022 enhance acquisition of this work. The new codes replace 80500 and 80502 and allow for more precise identification of any work requiring medical judgment that a pathologist does at the request of another qualified health care professional (HCP) or as required by federal or state regulation.
The codes can be used to bill for associated time spent reviewing health records, communicating with other HCPs, placing orders, and documentation. An HCP can bill according to level of medical decision-making (MDM) or time spent by the consulting pathologist. Code 80503 can be billed for 5 to 20 minutes of a pathologist's time, 80504 for 21 to 40 minutes, 80505 for 41 to 60 minutes, and 80506 for each additional 30 minutes after the first hour. Levels of MDM (low, moderate, and high) are defined as for other evaluation and management services. A consultation report must be generated and contain documentation of the consultation request, pathologist interpretation, and justification for the level of service associated with the chosen code. Relative value units (RVUs) and reimbursement associated with each as well as other consultation-related codes are available in Table 1.

This article outlines how the pathology time investment associated with anatomic pathology molecular testing at the Kansas City Veterans Affairs Medical Center (KCVAMC) can be captured using the consultation process and new CPT codes. Staff included 4 pathologists, 3 histotechnologists, 1 histology supervisor, 1 grossing room technician, and 1 cytotechnologist, 1 cytology technician.
METHODS
The AP molecular testing consultation process at the KCVAMC was mapped, with the average time measured for each step (Figure). AP records for 2021 were reviewed to determine the number of AP molecular send out tests. Cumulative time investment was calculated in hours and a theoretical number of RVUs was calculated using the new pathology clinical consultation CPT codes (80503-80506). This theoretical number of RVUs was compared with the total AP RVUs generated in 2021 to determine a potential increase in RVUs with use of the new CPT codes to capture pathology work associated with AP molecular testing consultations.

RESULTS
From 2021 to 2023, there were 21,021 AP cases at the KCVAMC. Basal cell carcinomas and squamous cell carcinomas of the skin were excluded because they comprise most cancer cases but almost never necessitate AP molecular test consultations. A total of 2118 cancer cases were included, representing 10.1% of all cases. Ancillary AP molecular send-out tests were performed on 1338 (6.4%) cases. Since ancillary tissue tests are requested by consultation at the KCVAMC, this resulted in 1338 consultations (Table 2).

The time to complete a consultation was measured by calculating the mean time required to complete each step (Table 3). With in-house specimen consultations requiring 90 minutes each and outside specimen consultations requiring 100 minutes each, a total of 2040 hours of pathology staff time was necessary to complete associated consultations. Billing for this time with the new pathology clinical consultation CPT codes would generate 3847 RVUs, which would have equated to 14.8% (3847/25,920) of the anatomic pathology RVUs.

DISCUSSION
When considering the lengths of time for tasks associated with each consultation, it is important to remember that the volume (2-3 daily), was insufficient to meaningfully benefit from batching. Thus, waiting to perform a particular task until it was needed for multiple cases reduced the inefficiency associated with starting and switching between tasks. Both the Computerized Patient Record System and VistA had to be reopened, reauthenticated, and reloaded for each step that required use of the health record, printer, or fax machine. Faxes also required waiting for transmission and printed confirmation of successful transmission. As a result, the time values denoted for each step are likely underestimated, as the effect of interruptions is significant and not reflected in the estimates recorded.10
This analysis has demonstrated that PO entails a significant amount of work for pathology departments. To determine and maintain appropriate staffing models, this work must be captured and reimbursed. Unlike other pathology work, which is performed in-house and reimbursed for the associated test, a significant proportion of PO testing is sent out. Even if more reliable assays are developed, the physical processes of sending out samples and reporting test results cannot be outsourced. Independent and commensurate reimbursement methods are necessary to allow for this work and PO.
CMS included new pathology clinical consultation codes that may be used to bill for some of this work according to the 2022 physician fee schedule due to advocacy work by the College of American Pathologists and the American Medical Association CPT editorial panel.11
CONCLUSIONS
This analysis found that adoption of PO may present a significant amount of additional work for pathology departments. To determine and maintain appropriate staffing models, work completed by pathologists in this manner must be recorded and reimbursed. Pathologists need to be trained and encouraged to use these CPT codes and bill for the work described in this article. The increased revenue will allow for additional positions to alleviate the burdens imposed by understaffing so that pathology can function as a facilitator of PO rather than as a barrier to it.
Precision oncology (PO) is cancer treatment individualized to the special characteristics of a patient’s tumor. It has become standard care for most patients with advanced cancer. Advances in molecular cell biology and immunology have identified numerous targets and many therapies have been developed as a result. Molecular testing and targeted therapy are typically covered by insurance, even when inflation-adjusted price growth is considered.1 Barriers remain, however, and pathologists are uniquely qualified to address some of the challenges.2
Most US laboratories do not perform molecular diagnostic tests for PO, particularly comprehensive evaluation of multiple targets by next-generation sequencing, or other techniques. Instead, these tests are sent to reference laboratories. The workload associated with referral testing is an obstacle to increased use of such tests. Despite guideline recommendations, a minority of indicated tests are performed.3 This is true even when testing costs are covered by clinical trials or grants, such as those in the Veterans Health Administration (VHA).
The main characteristic of successful PO programs is a multidisciplinary commitment, including pathology involvement in molecular tumor boards and assistance with test selection, tissue collection, and result interpretation.2 This, however, adds to the workload for the pathology department, an underappreciated phenomenon in the context of pathology workforce shortages.4
Workforce shortages impact all occupations in the laboratory setting. Though the shortage of medical technologists in clinical pathology laboratories has repeatedly been identified as critical at the VHA as well as in the private sector, the same cannot be said for staff shortages in anatomic pathology laboratories. Thus, the hospital laboratory divisions are concerned with biopsy or resection tissue specimens as opposed to the bodily fluid specimens that predominate in clinical laboratories.5 The lack of accurate data on histopathology technicians and technologists has precluded the degree of recognition seen for medical technologists. In labor statistics, these occupations are often obscured by inclusion with other jobs in broad categories such as medical and clinical laboratory technologists and technicians.6 Vacancy—the principal metric used to assess medical laboratory workforce shortage—fails to account for positions that are eventually eliminated after remaining vacant for prolonged periods, positions not replaced as a result of ambitious efficiency measures, or positions that were never created due to insufficient funding, reasons for administrative disapproval, or coverage by laboratory professionals working extra shifts or second jobs.7
Increased demand for pathologists is suggested by a 42% increase in workload per pathologist over the last decade, while a shortage is suggested by decreases in absolute and population-adjusted numbers of pathologists.8,9 An influx of pathologists is not an expected remedy due to the global decline in medical graduates pursuing careers in the field.8
Approximations for required labor and potential revenue generation are necessary to justify creation of pathology positions. This work mostly has gone uncaptured due to the limitations of Current Procedural Terminology (CPT) codes. Few laboratories have consistently used the 88363, 88325, and G0452 CPT codes. The pathology clinical consultation CPT codes (80503-80506) released in 2022 enhance acquisition of this work. The new codes replace 80500 and 80502 and allow for more precise identification of any work requiring medical judgment that a pathologist does at the request of another qualified health care professional (HCP) or as required by federal or state regulation.
The codes can be used to bill for associated time spent reviewing health records, communicating with other HCPs, placing orders, and documentation. An HCP can bill according to level of medical decision-making (MDM) or time spent by the consulting pathologist. Code 80503 can be billed for 5 to 20 minutes of a pathologist's time, 80504 for 21 to 40 minutes, 80505 for 41 to 60 minutes, and 80506 for each additional 30 minutes after the first hour. Levels of MDM (low, moderate, and high) are defined as for other evaluation and management services. A consultation report must be generated and contain documentation of the consultation request, pathologist interpretation, and justification for the level of service associated with the chosen code. Relative value units (RVUs) and reimbursement associated with each as well as other consultation-related codes are available in Table 1.

This article outlines how the pathology time investment associated with anatomic pathology molecular testing at the Kansas City Veterans Affairs Medical Center (KCVAMC) can be captured using the consultation process and new CPT codes. Staff included 4 pathologists, 3 histotechnologists, 1 histology supervisor, 1 grossing room technician, and 1 cytotechnologist, 1 cytology technician.
METHODS
The AP molecular testing consultation process at the KCVAMC was mapped, with the average time measured for each step (Figure). AP records for 2021 were reviewed to determine the number of AP molecular send out tests. Cumulative time investment was calculated in hours and a theoretical number of RVUs was calculated using the new pathology clinical consultation CPT codes (80503-80506). This theoretical number of RVUs was compared with the total AP RVUs generated in 2021 to determine a potential increase in RVUs with use of the new CPT codes to capture pathology work associated with AP molecular testing consultations.

RESULTS
From 2021 to 2023, there were 21,021 AP cases at the KCVAMC. Basal cell carcinomas and squamous cell carcinomas of the skin were excluded because they comprise most cancer cases but almost never necessitate AP molecular test consultations. A total of 2118 cancer cases were included, representing 10.1% of all cases. Ancillary AP molecular send-out tests were performed on 1338 (6.4%) cases. Since ancillary tissue tests are requested by consultation at the KCVAMC, this resulted in 1338 consultations (Table 2).

The time to complete a consultation was measured by calculating the mean time required to complete each step (Table 3). With in-house specimen consultations requiring 90 minutes each and outside specimen consultations requiring 100 minutes each, a total of 2040 hours of pathology staff time was necessary to complete associated consultations. Billing for this time with the new pathology clinical consultation CPT codes would generate 3847 RVUs, which would have equated to 14.8% (3847/25,920) of the anatomic pathology RVUs.

DISCUSSION
When considering the lengths of time for tasks associated with each consultation, it is important to remember that the volume (2-3 daily), was insufficient to meaningfully benefit from batching. Thus, waiting to perform a particular task until it was needed for multiple cases reduced the inefficiency associated with starting and switching between tasks. Both the Computerized Patient Record System and VistA had to be reopened, reauthenticated, and reloaded for each step that required use of the health record, printer, or fax machine. Faxes also required waiting for transmission and printed confirmation of successful transmission. As a result, the time values denoted for each step are likely underestimated, as the effect of interruptions is significant and not reflected in the estimates recorded.10
This analysis has demonstrated that PO entails a significant amount of work for pathology departments. To determine and maintain appropriate staffing models, this work must be captured and reimbursed. Unlike other pathology work, which is performed in-house and reimbursed for the associated test, a significant proportion of PO testing is sent out. Even if more reliable assays are developed, the physical processes of sending out samples and reporting test results cannot be outsourced. Independent and commensurate reimbursement methods are necessary to allow for this work and PO.
CMS included new pathology clinical consultation codes that may be used to bill for some of this work according to the 2022 physician fee schedule due to advocacy work by the College of American Pathologists and the American Medical Association CPT editorial panel.11
CONCLUSIONS
This analysis found that adoption of PO may present a significant amount of additional work for pathology departments. To determine and maintain appropriate staffing models, work completed by pathologists in this manner must be recorded and reimbursed. Pathologists need to be trained and encouraged to use these CPT codes and bill for the work described in this article. The increased revenue will allow for additional positions to alleviate the burdens imposed by understaffing so that pathology can function as a facilitator of PO rather than as a barrier to it.
- Wilson LE, Greiner MA, Altomare I, et al. Rapid rise in the cost of targeted cancer therapies for Medicare patients with solid tumors from 2006 to 2015. J Geriatr Oncol. 2021;12:375-380. doi:10.1016/j.jgo.2020.11.007
- Ersek JL, Black LJ, Thompson MA, et al. Implementing precision medicine programs and clinical trials in the community-based oncology practice: barriers and best practices. Am Soc Clin Oncol Educ Book. 2018;38:188-196. doi:10.1200/EDBK_200633
- Inal C, Yilmaz E, Cheng H, et al. Effect of reflex testing by pathologists on molecular testing rates in lung cancer patients: experience from a community-based academic center. J Clin Oncol. 2014;32:8098. doi:10.1200/jco.2014.32.15_suppl.8098
- Robboy SJ, Gupta S, Crawford JM, et al. The pathologist workforce in the United States: II. an interactive modeling tool for analyzing future qualitative and quantitative staffing demands for services. Arch Pathol Lab Med. 2015;139:1413-1430. doi:10.5858/arpa.2014-0559-OA
- OIG determination of Veterans Health Administration’s occupational staffing shortages fiscal year 2021. Department of Veterans Affairs OIG. September 28, 2021. Accessed January 30, 2026. https://www.oversight.gov/report/VA/OIG-determination-veterans-health-administrations-occupational-staffing-shortages-fiscal
- Zanto S, Cremeans L, Deutsch-Keahey D, et al. Addressing the clinical laboratory workforce shortage. The American Society for Clinical Laboratory Science. July 2, 2020. Accessed January 30, 2026. https://ascls.org/addressing-the-clinical-laboratory-workforce-shortage/
- Bennett A, Garcia E, Schulze M, et al. Building a laboratory workforce to meet the future: ASCP Task Force on the Laboratory Professionals Workforce. Am J Clin Pathol. 2014;141:154-167. doi:10.1309/AJCPIV2OG8TEGHHZ
- Fielder T, Watts F, Howden C, et al. Why choose a pathology career? Arch Pathol Lab Med. 2022;146:903-910. doi:10.5858/arpa.2021-0118-OA
- Metter DM, Colgan TJ, Leung ST, et al. Trends in the US and Canadian pathologist workforces from 2007 to 2017. JAMA Netw Open. 2019;2:e194337. doi:10.1001/jamanetworkopen.2019.4337
- Schulte B. Work interruptions can cost you 6 hours a day. An efficiency expert explains how to avoid them. The Washington Post. June 1, 2015. Accessed January 30, 2026. https://www.washingtonpost.com/news/inspired-life/wp/2015/06/01/interruptions-at-work-can-cost-you-up-to-6-hours-a-day-heres-how-to-avoid-them/
- Fiegl C. Medicare adopts new clinical consult billing codes. College of American Pathologists Today. December 2021. Accessed January 30, 2026. https://www.captodayonline.com/medicare-adopts-new-clinical-consult-billing-code
- Wilson LE, Greiner MA, Altomare I, et al. Rapid rise in the cost of targeted cancer therapies for Medicare patients with solid tumors from 2006 to 2015. J Geriatr Oncol. 2021;12:375-380. doi:10.1016/j.jgo.2020.11.007
- Ersek JL, Black LJ, Thompson MA, et al. Implementing precision medicine programs and clinical trials in the community-based oncology practice: barriers and best practices. Am Soc Clin Oncol Educ Book. 2018;38:188-196. doi:10.1200/EDBK_200633
- Inal C, Yilmaz E, Cheng H, et al. Effect of reflex testing by pathologists on molecular testing rates in lung cancer patients: experience from a community-based academic center. J Clin Oncol. 2014;32:8098. doi:10.1200/jco.2014.32.15_suppl.8098
- Robboy SJ, Gupta S, Crawford JM, et al. The pathologist workforce in the United States: II. an interactive modeling tool for analyzing future qualitative and quantitative staffing demands for services. Arch Pathol Lab Med. 2015;139:1413-1430. doi:10.5858/arpa.2014-0559-OA
- OIG determination of Veterans Health Administration’s occupational staffing shortages fiscal year 2021. Department of Veterans Affairs OIG. September 28, 2021. Accessed January 30, 2026. https://www.oversight.gov/report/VA/OIG-determination-veterans-health-administrations-occupational-staffing-shortages-fiscal
- Zanto S, Cremeans L, Deutsch-Keahey D, et al. Addressing the clinical laboratory workforce shortage. The American Society for Clinical Laboratory Science. July 2, 2020. Accessed January 30, 2026. https://ascls.org/addressing-the-clinical-laboratory-workforce-shortage/
- Bennett A, Garcia E, Schulze M, et al. Building a laboratory workforce to meet the future: ASCP Task Force on the Laboratory Professionals Workforce. Am J Clin Pathol. 2014;141:154-167. doi:10.1309/AJCPIV2OG8TEGHHZ
- Fielder T, Watts F, Howden C, et al. Why choose a pathology career? Arch Pathol Lab Med. 2022;146:903-910. doi:10.5858/arpa.2021-0118-OA
- Metter DM, Colgan TJ, Leung ST, et al. Trends in the US and Canadian pathologist workforces from 2007 to 2017. JAMA Netw Open. 2019;2:e194337. doi:10.1001/jamanetworkopen.2019.4337
- Schulte B. Work interruptions can cost you 6 hours a day. An efficiency expert explains how to avoid them. The Washington Post. June 1, 2015. Accessed January 30, 2026. https://www.washingtonpost.com/news/inspired-life/wp/2015/06/01/interruptions-at-work-can-cost-you-up-to-6-hours-a-day-heres-how-to-avoid-them/
- Fiegl C. Medicare adopts new clinical consult billing codes. College of American Pathologists Today. December 2021. Accessed January 30, 2026. https://www.captodayonline.com/medicare-adopts-new-clinical-consult-billing-code
Capturing Pathology Workload Associated With Precision Oncology
Capturing Pathology Workload Associated With Precision Oncology