User login
Redefining treatment success in type 2 diabetes mellitus: Comprehensive targeting of core defects
According to the American Association of Clinical Endocrinologists (AACE) and the American Diabetes Association (ADA), glycosylated hemoglobin (HbA1c) in patients with diabetes should be maintained at 6.5% or less (AACE) or at less than 7.0% (ADA). Both organizations support an aggressive stepwise approach that includes medication and lifestyle modification, with strategies and clinical attention devoted to avoiding significant hypoglycemia.1,2 Yet, despite the introduction of new antidiabetes agents, most current management strategies are offset by limitations in achieving and maintaining glycemic targets needed to provide optimal care for patients with diabetes, more than 90% of whom have type 2 diabetes mellitus (T2DM).3,4
Nationally, glycemic control among patients with T2DM has improved but is still far from optimal. According to data from the 1999–2000 National Health and Nutrition Examination Survey (NHANES), glycemic control (HbA1c < 7.0%) rates were 35.8% for patients with T2DM.5 In a more recent report (NHANES 1999–2004), fewer than half (48.4%) of adult patients with diagnosed diabetes achieved HbA1c levels below 7.0%.5,6 Factors contributing to these data include earlier onset and earlier detection of T2DM.7
CHANGING TREATMENT TRENDS
Available treatments for patients with T2DM include secretagogues, such as sulfonylureas and “glinides” (repaglinide and nateglinide), metformin, thiazolidinediones (TZDs), and dipeptidyl peptidase–4 (DPP-4) inhibitors among oral medications, and insulin and glucagon-like peptide–1 (GLP-1) receptor agonists among parenterally administered agents. According to the latest published data on prescribing patterns for patients with T2DM, analyses of the National Disease and Therapeutic Index (1994–2007) and the National Prescription Audit (2001–2007), sulfonylurea use decreased from 67% of treatment visits in 1994 to 34% of visits in 2007.8 By 2007, metformin, used in 54% of treatment visits, and TZDs, used in 28%, were the most frequently administered antidiabetes agents. Insulin use declined from 38% of visits during which a treatment was administered in 1994 to 25% of visits in 2000, but had increased subsequently to 28% of visits in 2007.
SIGNIFICANCE OF CARDIOVASCULAR RISK
Clinical research has suggested that focusing solely on improving glycemic control may be insufficient to reduce overall morbidity and mortality associated with diabetes. Specifically, data from recent studies, including the Action to Control Cardiovascular Risk in Diabetes (ACCORD), the Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE), and the Veterans Affairs Diabetes Trial (VADT), emphasized that lowering HbA1c below 7% in a high-risk population of individuals with T2DM did not improve cardiovascular (CV) outcomes.9–11 The observations confirm that risk factors, including weight, blood pressure (BP), and lipid levels, are vitally important in reducing morbidity and mortality in this population. This perception is further underscored by the NHANES 1999–2004 data, which showed poor concurrent control of HbA1c, BP, and lipids; only 13.2% of patients with diagnosed diabetes achieved all three target goals simultaneously.6 Similarly, a nationwide survey in Norway showed that only 13% of patients with T2DM concurrently achieved goals for HbA1c, BP, and lipids.12
In the Danish Steno-2 Study, patients with T2DM and persistent microalbuminuria were treated with either intensive target-driven therapy using multiple drugs or conventional multifactorial treatment. Over a mean period of 13.3 years (7.8 years of treatment plus 5.5 years of follow-up), intensive multifactorial intervention to control multiple CV risk factors, including HbA1c, BP, and lipids, was associated with a lower risk of death from CV causes (hazard ratio [HR], 0.43; 95% confidence interval [CI], 0.19 to 0.94; P = .04) and a lower risk of CV events (HR, 0.41; 95% CI, 0.25 to 0.67; P < .001) than was conventional therapy.13
This article clarifies the redefinition of treatment success in patients with T2DM based on targeting the underlying physiologic defects of the disease.
T2DM, OVERWEIGHT/OBESITY, AND CV DISEASE: CLOSELY LINKED
The incidence and prevalence of T2DM, overweight/obesity, and CV disease (CVD) are increasing worldwide. It is estimated that the worldwide prevalence of diabetes will increase from 171 million in 2000 to 366 million by 203014; T2DM increases the risk of morbidity and mortality from microvascular (eg, neuropathic, retinopathic, nephropathic) and macrovascular (eg, coronary, peripheral vascular disease) complications.15 According to a Michigan health maintenance organization study (N = 1,364), the median annual direct cost of medical care for Caucasian patients with T2DM who were diet controlled, had a body mass index (BMI) of 30 kg/m2 or higher, and had no vascular complications was estimated to be $1,700 for men and $2,100 for women.16 The actual cost of care for patients with T2DM may be much higher, since most patients present with multiple CV risk factors in addition to being overweight.
NHANES data show that approximately two-thirds of Americans are either overweight or obese17; overweight/obesity affects about 80% of adults diagnosed with T2DM.18 Overweight or obesity can increase the risk for developing T2DM by more than 90-fold and, in women, it can increase the risk for developing coronary heart disease (CHD) by sixfold.19 The close link between T2DM and CVD is underscored further with recent data from the Framingham Heart Study, which showed a high lifetime risk of CVD in patients with diabetes, heightened further by obesity. During the 30-year study period, the lifetime risk of CVD in normal-weight people with diabetes was 78.6% in men and 54.8% in women; the risk increased to 86.9% in obese men with diabetes and to 78.8% in obese women with diabetes.20 The NHANES data also showed that the prevalence of T2DM increased in the past decade and that patients are being diagnosed at a younger age, from a mean age of 52 years in 1988–1994 to 46 years in 1999–2000.7
BRIDGING THE GAP FROM PATHOPHYSIOLOGY TO UNMET NEEDS
The paradigm behind the pathophysiology of T2DM has shifted from its perception as a simple “dual-defect” disease (ie, deficiency in insulin secretion and peripheral tissue insulin resistance) to a multidimensional disorder.1,21 This new model includes overweight/obesity, insulin resistance, qualitative and quantitative defects in insulin secretion, and dysregulation in the secretion of other hormones, including the beta-cell hormone amylin, the alpha-cell hormone glucagon, and the gastrointestinal incretin hormones GLP-1 and glucose-dependent insulinotropic polypeptide.21–23
CLINICAL GUIDELINES AND CV RISK FACTOR MANAGEMENT
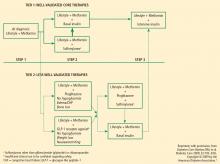
The best strategy for managing T2DM is a comprehensive approach that addresses the fundamental core defects plus associated factors that contribute to increased CV risk. Several specialty groups have suggested guidelines and algorithms for the management of T2DM and its comorbidities. These guidelines, including the ADA standards of medical care, the AACE standards in tandem with the American College of Endocrinology guidelines, and the recent joint statement from the ADA and the European Association for the Study of Diabetes (EASD), acknowledge that the core defects of T2DM and the associated CV risk factors (eg, weight gain, obesity, hypertension, dyslipidemia) are important in developing optimal treatment strategies.1–3 Medical nutrition guidelines advocate weight loss as a key initial step in managing T2DM and the comorbidities that lead to elevated CV risk.25,26 The National Institutes of Health and the US Department of Health and Human Services/US Department of Agriculture advocate regular physical activity, dietary assessment, and periodic comorbidity and weight assessment for all people, not just those with T2DM or CVD.26,27
Weight reduction
Evidence in support of effective lifestyle intervention was demonstrated in the Action for Health in Diabetes (Look AHEAD) study. After 1 year, patients with T2DM treated with intensive lifestyle intervention lost an average of 8.6% of their initial weight compared with 0.7% in patients treated only with diabetes support and education (P < 0.001). The intensive-intervention patients also had a significant drop in HbA1c (from 7.3% to 6.6%; P < 0.001) and were able to reduce their antidiabetes, antihypertensive, and lipid-lowering medications.28 More recent data from the Look AHEAD study reported that overweight patients with T2DM enrolled in a weight management program experienced significant weight loss, improved physical fitness, reduced physical symptoms, and overall improvement in health-related quality of life.29 Thus, weight reduction appears to be a key component in reducing CV risk and improving quality of life in most patients with T2DM.28–30
Hypertension
Hypertension is a major risk factor for microvascular complications and CVD, and may be associated with, or be the underlying result of, nephropathy.2 BP control is clearly important in reducing the morbidity and mortality associated with T2DM. The recommended BP goal in patients with T2DM is less than 130/80 mm Hg.1,2
Hyperlipidemia
According to the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III [ATP III]), diabetes is considered a CHD risk equivalent because it confers a high risk of new CHD developing within 10 years.31 In addition to the NCEP–ATP III guidelines, the ADA and the AACE have set target levels for lipids in patients with diabetes, including T2DM.1,2,31 All three organizations have defined 100 mg/dL as the target level for low-density lipoprotein.
HbA1c and lifestyle intervention
EVOLUTION OF ANTIDIABETES THERAPIES
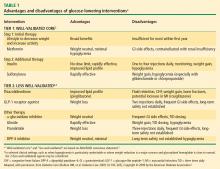
Traditional antidiabetes agents used in the treatment of patients with T2DM have focused mainly on insulin secretion and insulin resistance, with treatment success defined as achieving HbA1c goals with a reduced incidence of hypoglycemia.23 Secretagogues, such as sulfonylureas and glinides, stimulate the pancreas to release insulin. Insulin sensitizers, such as TZDs and metformin, enhance the action of insulin in muscle and fat1,3,23 and lower hepatic glucose production. The alpha-glucosidase inhibitors alter carbohydrate absorption from the gastrointestinal tract.1 The extent to which each agent achieves treatment success in terms of glucose lowering depends on several factors, including intrinsic attributes, duration of disease, and baseline glycemic control.3
GLP-1 receptor agonists
Exenatide effects. Although many agents are in development, to date exenatide is the only GLP-1 receptor agonist approved by the US Food and Drug Administration (FDA).8,33 Exenatide is an exendin-4 GLP-1 receptor agonist with multiple glucoregulatory effects, including enhanced glucose-dependent insulin secretion, reduced glucagon secretion and food intake, and slowed gastric emptying.22,34 Exenatide is detectable in the circulation for up to 10 hours following subcutaneous (SC) administration22 and has a greater potency in reducing plasma glucose than GLP-1 in preclinical studies.35,36
By virtue of its beneficial effects on glycemic control, weight, BP, and lipids, exenatide addresses some of the components of the metabolic syndrome.37–41 In pivotal 30-week studies, exenatide was associated with HbA1c reductions that ranged from –0.40% to –0.86% from baseline and decreases in body weight of approximately –1 kg to –3 kg from baseline, without severe hypoglycemia.37–39 The percentage of patients who reached the ADA goal of HbA1c less than 7.0% at 30 weeks ranged from 24% to 34%. The addition of exenatide to TZD therapy in a 16-week study was associated with mean reductions in HbA1c of –0.98%, fasting plasma glucose (FPG) concentration of –1.69 mmol/L (–30.42 mg/dL), and body weight of –1.51 kg.40
A posthoc analysis of an open-label extension study involving patients who completed the original 30-week placebo-controlled studies showed that 46% of patients who remained on exenatide achieved the ADA goal of HbA1c less than 7.0% at 3 years.41 Exenatide administered for up to 3.5 years was associated with sustained reductions in HbA1c of –1.0% (P < .0001) and body weight of –5.3 kg (P < .001). Pancreatic beta-cell function, assessed by homeostasis model assessment, improved, as did BP, triglyceride, high-density lipoprotein, low-density lipoprotein, and aspartate aminotransferase levels.41
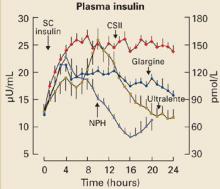
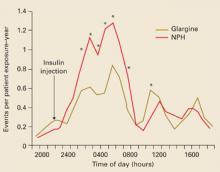
Comparison with insulin analogues. Comparative studies have highlighted the contrasting effects of exenatide and insulin analogues (eg, insulin glargine and fixed-ratio insulin).42–45 In a 26-week trial comparing exenatide with insulin glargine in subjects with T2DM, both agents resulted in similar decreases in HbA1c. Exenatide was also associated with a –2.3-kg weight reduction, whereas insulin glargine was associated with a +1.8-kg weight gain.42 Although rates of symptomatic hypoglycemia were similar, there were fewer cases of nocturnal hypoglycemia with exenatide (0.9 event/patient-year vs 2.4 events/patient-year with insulin).
In a 32-week study comparing exenatide BID with titrated insulin glargine QD, the HbA1c reductions for exenatide and insulin glargine were comparable. However, body weight decreased –4.2 kg over two 16-week treatment periods with exenatide, but increased +3.3 kg over the same periods with the basal insulin analogue.43 The incidence of hypoglycemia was lower with exenatide than with insulin glargine (14.7% vs 25.2%), although the difference was not statistically significant.
In another study that compared exenatide with biphasic insulin aspart, patients who were treated with exenatide also lost weight while those who received the fast-acting insulin analogue gained weight (between-group difference, –5.4 kg). Patients treated with exenatide also demonstrated greater reductions in postprandial plasma glucose (PPG) excursions following their morning (P < .001), midday (P = .002), and evening meals (P < .001).44 Overall, hypoglycemia rates were similar at study end between exenatide and insulin aspart (4.7 events/patient-year vs 5.6 events/patient-year). In all of these studies, significant gastrointestinal adverse events (nausea and vomiting) occurred more frequently with exenatide, and more patients withdrew from exenatide than from insulin.
Formulations in development. Other advances in GLP-1 receptor agonist therapy include novel formulations under clinical development, such as exenatide once weekly36,46 and liraglutide, a human analogue GLP-1 receptor agonist formulated for once-daily administration.47,48 In a 52-week study in patients with T2DM, liraglutide significantly reduced HbA1c; the 1.2-mg SC QD dosage reduced HBA1c by –0.84% (P = .0014) and the 1.8-mg SC QD dosage by –1.14% (P < .0001). In comparison, glimepiride 8 mg orally QD achieved a –0.51% reduction. Liraglutide was also associated with greater reductions in weight, hypoglycemia, and systolic BP than glimepiride.47
A 26-week study compared liraglutide (0.6, 1.2, and 1.8 mg SC QD), placebo, and glimepiride 4 mg QD in combination with metformin 1 g BID. HbA1c was reduced significantly in all liraglutide groups compared with placebo (P < .0001). Mean HbA1c decreased –1.0% with liraglutide 1.2 mg and 1.8 mg and with glimepiride; it decreased –0.7% with liraglutide 0.6 mg; and it increased +0.1% with placebo. Body weight decreased –1.8 kg to –2.8 kg in all liraglutide groups but increased +1.0 kg in the glimepiride group (P < .0001). The incidence of minor hypoglycemia with liraglutide (~3%) was comparable to that observed with placebo but less than that with glimepiride (17%; P < .001).48
A once-weekly long-acting release (LAR) formulation of exenatide submitted to the FDA for approval may provide enhanced glycemic and weight control, potentially improving patient acceptance and adherence.36,46 In a 15-week study, exenatide once weekly produced significant reductions in HbA1c, FPG, PPG, and body weight. There were no withdrawals due to adverse events, and the formation of anti-exenatide antibodies was not predictive of therapeutic end point response or adverse safety outcome. Instances of hypoglycemia were mild and not dose related.36 In a 30-week study comparing exenatide LAR once weekly with exenatide BID, patients given exenatide LAR once weekly had significantly greater HbA1c reductions than did patients given exenatide BID (–1.9% vs –1.5%; P = .0023). Treatment adherence was 98% with both exenatide regimens, and no episodes of major hypoglycemia occurred with either formulation regardless of background sulfonylurea use. Favorable effects on BP and lipid profile were observed with both exenatide regimens.46
DPP-4 inhibitors
The DPP-4 inhibitors (commonly called gliptins) inhibit the proteolytic cleavage of circulating GLP-1 by binding to the DPP-4 enzyme, increasing the concentration of endogenous GLP-1 approximately two- to threefold.49–51 These concentrations result in more prompt and appropriate secretion of insulin and suppression of glucagon in response to a carbohydrate-containing snack or meal, with the change in glucagon correlating linearly with improved glucose tolerance.51
DPP-4 inhibitors, which are given orally, include sitagliptin and saxagliptin (approved in the United States) and vildagliptin (not approved in the United States but used in the European Union and Latin America).8,22,33,52 Sitagliptin can be used either as monotherapy or in combination with metformin or a TZD.8,49–55 Recently, a single-tablet formulation of sitagliptin plus metformin was granted regulatory approval.8
When used alone or in combination with metformin or pioglitazone, sitagliptin has been associated with significant reductions in HbA1c (of ~0.5% to 0.6% when used alone, ~0.7% with metformin, and ~0.9% with pioglitazone [P < .001 vs placebo]), with hypoglycemia occurring in 1.3% or less of the population.54 In an 18-week study in which patients with T2DM who were inadequately controlled with metformin monotherapy were randomized to receive add-on sitagliptin (100 mg QD), rosiglitazone (8 mg QD), or placebo, sitagliptin reduced HbA1c –0.73% (P < .001 vs placebo) and reduced body weight –0.4 kg, while rosiglitazone reduced HbA1c –0.79% and increased body weight +1.5 kg.55
To evaluate the effectiveness of sitagliptin and metformin as initial therapy, a 54-week study was completed in 885 patients with T2DM and inadequate glycemic control (HbA1c 7.5–11%) on diet and exercise.56 Patients were evaluated on monotherapy with either sitagliptin (100 mg QD) or metformin (1 g or 2 g QD), or on initial therapy with the two in combination (sitagliptin 100 mg + metformin 1 mg or 2 mg QD). At week 54, in the all-patients-treated analysis, mean changes in HbA1c from baseline were –1.8% with sitagliptin plus metformin 2 g QD, –1.4% with sitagliptin plus metformin 1 g QD, –1.3% with metformin 2 g QD monotherapy, –1.0% with metformin 1 g QD monotherapy, and –0.8% with sitagliptin 100 mg QD monotherapy.
All treatments improved measures of beta-cell function (eg, homeostasis model assessment [HOMA]-beta, proinsulin/insulin ratio). Mean body weight decreased from baseline in the combination and metformin monotherapy groups and was unchanged from baseline in the sitagliptin monotherapy group. The incidence of hypoglycemia was low (1%–3%) across treatment groups. The incidence of gastrointestinal adverse experiences was evaluated with the coadministration of sitagliptin and metformin and appeared similar to that observed with use of metformin as monotherapy.56 Thus, this study suggested that an initial combination of a DPP-IV inhibitor with metformin can improve glycemic control and markers of beta-cell function in patients with T2DM.
Incretin-based therapies compared
Studies in both healthy individuals and in patients with T2DM have shown that oral DPP-4 inhibitors such as sitagliptin increase endogenous GLP-1 concentrations by about twofold compared with placebo.22,50 The pharmacologic concentration of subcutaneously administered exenatide available for activating the GLP-1 receptor is significantly greater than the increased endogenous GLP-1 concentrations achieved with sitagliptin. In a recent clinical study comparing exenatide and sitagliptin in patients with T2DM, the mean 2-hour plasma concentration for exenatide was 64 pM compared with the mean 2-hour postprandial GLP-1 concentration of 15 pM for sitagliptin (baseline GLP-1 concentration was 7.2 pM).57 While both agents were shown to be effective, exenatide appeared to have had a greater effect than sitagliptin in increasing insulin secretion and reducing postprandial glucagon secretion, leading to significantly (P < 0.0001) greater reductions in PPG.57
Sitagliptin has been minimally associated with nausea, whereas patients who take exenatide need to be informed of the risk of usually mild to moderate, but sometimes severe, nausea and vomiting that tends to decrease over time.
For a detailed comparison of the effects of GLP-1 receptor agonists and DPP-4 inhibitors on HbA1c, weight, and hypoglycemia, see “Advances in therapy for type 2 diabetes: GLP–1 receptor agonists and DPP–4 inhibitors.”
CONCLUSION
Despite advances in diagnosis and treatment, T2DM, overweight/obesity, CVD, and their complications remain major public health burdens worldwide. The concepts that explain the pathophysiology of T2DM include the contribution of various factors beyond insulin secretion and insulin resistance, such as the role of incretin hormones in disease progression. A comprehensive approach to managing patients with T2DM requires targeting the fundamental defects of the disease and its comorbidities. Newer agents, including incretin-based therapies such as GLP-1 receptor agonists and DPP-4 inhibitors, address the fundamental defects of T2DM. The definition of treatment success in the management of T2DM will be redefined as more data become available on agents that exert beneficial effects not only on glycemia but on parameters that may influence overall CV health, such as weight, BP, and lipid profiles.
- AACE Diabetes Mellitus Clinical Practice Guidelines Task Force. American Association of Clinical Endocrinologists medical guidelines for clinical practice for the management of diabetes mellitus. Endocr Pract 2007; 13(suppl 1):S4–S68.
- American Diabetes Association. Standards of medical care in diabetes—2009. Diabetes Care 2009; 32(suppl 1):S13–S61.
- Nathan DM, Buse JB, Davidson MB, et al. Medical management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 2009; 32:193–203.
- National Institute of Diabetes and Digestive and Kidney Diseases. National Diabetes Statistics, 2007 fact sheet. National Institutes of Health Web site. http://www.diabetes.niddk.nih.gov/dm/pubs/statistics/index.htm. Published 2008. Accessed September 16, 2009.
- Koro CE, Bowlin SJ, Bourgeois N, Fedder DO. Glycemic control from 1988 to 2000 among US adults diagnosed with type 2 diabetes: a preliminary report. Diabetes Care 2004; 27:17–20.
- Ong KL, Cheung BM, Wong LY, Wat NM, Tan KC, Lam KS. Prevalence, treatment, and control of diagnosed diabetes in the US National Health and Nutrition Examination Survey 1999–2004. Ann Epidemiol 2008; 18:222–229.
- Koopman RJ, Mainous AG III, Diaz VA, Geesey ME. Changes in age at diagnosis of type 2 diabetes mellitus in the United States, 1988 to 2000. Ann Fam Med 2005; 3:60–63.
- Alexander GC, Sehgal NL, Moloney RM, Stafford RS. National trends in treatment of type 2 diabetes mellitus, 1994–2007. Arch Intern Med 2008; 168:2088–2094.
- The Action to Control Cardiovascular Risk in Diabetes Study Group. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008; 358:2545–2559.
- The ADVANCE Collaborative Group. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008; 358:2560–2572.
- Duckworth W, Abraira C, Moritz T, et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med 2009; 360:129–139.
- Jenssen TG, Tonstad S, Claudi T, Midthjell K, Cooper J. The gap between guidelines and practice in the treatment of type 2 diabetes: a nationwide survey in Norway. Diabetes Res Clin Pract 2008; 80:314–320.
- Gaede P, Lund-Andersen H, Parving HH, Pedersen O. Effect of a multifactorial intervention on mortality in type 2 diabetes. N Engl J Med 2008; 358:580–591.
- Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care 2004; 27:1047–1053.
- Rosenstock J. Management of type 2 diabetes mellitus in the elderly: special considerations. Drugs Aging 2001; 18:31–44.
- Brandle M, Zhou H, Smith BR, et al. The direct medical cost of type 2 diabetes. Diabetes Care 2003; 26:2300–2304.
- National Center for Health Statistics. Prevalence of overweight and obesity among adults: United States 2003–2004. Centers for Disease Contral and Prevention Web site. http://www.cdc.gov/nchs/products/pubs/pubd/hestats/overweight/overwght_adult_03.htm. Published: April 2006. Accessed September 23, 2009.
- Van Gaal LF, Gutkin SW, Nauck MA. Exploiting the antidiabetic properties of incretins to treat type 2 diabetes mellitus: glucagon-like peptide 1 receptor agonists or insulin for patients with inadequate glycemic control. Eur J Endocrinol 2008; 158:773–784.
- Anderson JW, Kendall CW, Jenkins DJ. Importance of weight management in type 2 diabetes: review with meta-analysis of clinical studies. J Am Coll Nutr 2003; 22:331–339.
- Fox CS, Pencina MJ, Wilson PW, Paynter NP, Vasan RS, D’Agostino RB Sr. Lifetime risk of cardiovascular disease among individuals with and without diabetes stratified by obesity status in the Framingham heart study. Diabetes Care 2008; 31:1582–1584.
- DeFronzo RA. From the triumvirate to the ominous octet: a new paradigm for the treatment of type 2 diabetes mellitus. Diabetes 2009; 58:773–795.
- Stonehouse A, Okerson T, Kendall D, Maggs D. Emerging incretin based therapies for type 2 diabetes: incretin mimetics and DPP-4 inhibitors. Curr Diabetes Rev 2008; 4:101–109.
- Cefalu WT. Pharmacotherapy for the treatment of patients with type 2 diabetes mellitus: rationale and specific agents. Clin Pharmacol Ther 2007; 81:636–649.
- Henry RR. Evolving concepts of type 2 diabetes management with oral medications: new approaches to an old disease. Curr Med Res Opin 2008; 24:2189–2202.
- American Diabetes Association. Nutrition recommendations and interventions for diabetes: a position statement of the American Diabetes Association. Diabetes Care 2008; 31(suppl 1):S61−S78.
- US Department of Health and Human Services (HHS) and US Department of Agriculture. Dietary guidelines for Americans, 2005. US Department of HHS Web site. http://www.health.gov/DietaryGuidelines/dga2005/document/default.htm. Published January 2005. Accessed September 25, 2009.
- National Heart, Lung, and Blood Institute. The practical guide: identification, evaluation, and treatment of overweight and obesity in adults. National Institutes of Health Web site. http://www.nhlbi.nih.gov/guidelines/obesity/prctgd_c.pdf. Updated: October 2000. Accessed September 28, 2009.
- Look AHEAD Research Group. Reduction in weight and cardiovascular disease risk factors in individuals with type 2 diabetes: one-year results of the Look AHEAD trial. Diabetes Care 2007; 30:1374–1383.
- Williamson DA, Rejeski J, Lang W, Van Dorsten B, Fabricatore AN, Toledo K; for the Look AHEAD Research Group. Impact of a weight management program on health-related quality of life in overweight adults with type 2 diabetes. Arch Intern Med 2009; 169:163–171.
- Klein S, Sheard NF, Pi-Sunyer X, et al; for the American Diabetes Association; North American Association for the Study of Obesity; American Society for Clinical Nutrition. Weight management through lifestyle modification for the prevention and management of type 2 diabetes: rationale and strategies: a statement of the American Diabetes Association, the North American Association for the Study of Obesity, and the American Society for Clinical Nutrition. Diabetes Care 2004; 27:2067–2073.
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 2001; 285:2486–2497.
- Buse JB, Ginsberg HN, Bakris GL, et al. Primary prevention of cardiovascular diseases in people with diabetes mellitus: a scientific statement from the American Heart Association and the American Diabetes Association. Circulation 2007; 115:114–126.
- Amori RE, Lau J, Pittas AG. Efficacy and safety of incretin therapy in type 2 diabetes: systematic review and meta-analysis. JAMA 2007; 298:194–206.
- Nielsen LL, Young AA, Parkes DG. Pharmacology of exenatide (synthetic exendin-4): a potential therapeutic for improved glycemic control of type 2 diabetes. Regul Pept 2004; 117:77–88.
- Young AA, Gedulin BR, Bhavsar S, et al. Glucose-lowering and insulin-sensitizing actions of exendin-4: studies in obese diabetic (ob/ob, db/db) mice, diabetic fatty Zucker rats, and diabetic rhesus monkeys (Macaca mulatta). Diabetes 1999; 48:1026–1034.
- Kim D, MacConell L, Zhuang D, et al. Effects of once-weekly dosing of a long-acting release formulation of exenatide on glucose control and body weight in subjects with type 2 diabetes. Diabetes Care 2007; 30:1487–1493.
- Buse JB, Henry RR, Han J, et al. Effects of exenatide (exendin-4) on glycemic control over 30 weeks in sulfonylurea-treated patients with type 2 diabetes. Diabetes Care 2004; 27:2628–2635.
- DeFronzo RA, Ratner RE, Han J, Kim DD, Fineman MS, Baron AD. Effects of exenatide (exendin-4) on glycemic control and weight over 30 weeks in metformin-treated patients with type 2 diabetes. Diabetes Care 2005; 28:1092–1100.
- Kendall DM, Riddle MC, Rosenstock J, et al. Effects of exenatide (exendin-4) on glycemic control over 30 weeks in patients with type 2 diabetes treated with metformin and a sulfonylurea. Diabetes Care 2005; 28:1083–1091.
- Zinman B, Hoogwerf BJ, Durán García S, et al. The effect of adding exenatide to a thiazolidinedione in suboptimally controlled type 2 diabetes: a randomized trial. Ann Intern Med 2007; 146:477–485.
- Klonoff DC, Buse JB, Nielsen LL, et al. Exenatide effects on diabetes, obesity, cardiovascular risk factors and hepatic biomarkers in patients with type 2 diabetes treated for at least 3 years. Curr Med Res Opin 2008; 24:275–286.
- Heine RJ, Van Gaal LF, Johns D, Mihm MJ, Widel MH, Brodows RG; for the GWAA Study Group. Exenatide versus insulin glargine in patients with suboptimally controlled type 2 diabetes: a randomized trial. Ann Intern Med 2005; 143:559–569.
- Barnett AH, Burger J, Johns D, et al. Tolerability and efficacy of exenatide and titrated insulin glargine in adult patients with type 2 diabetes previously uncontrolled with metformin or a sulfonylurea: a multinational, randomized, open-label, two-period, crossover noninferiority trial. Clin Ther 2007; 29:2333–2348.
- Nauck MA, Duran S, Kim D, et al. A comparison of twice-daily exenatide and biphasic insulin aspart in patients with type 2 diabetes who were suboptimally controlled with sulfonylurea and metformin: a non-inferiority study. Diabetologia 2007; 50:259–267.
- Glass LC, Qu Y, Lenox S, et al. Effects of exenatide versus insulin analogues on weight change in subjects with type 2 diabetes: a pooled post-hoc analysis. Curr Med Res Opin 2008; 24:639–644.
- Drucker DJ, Buse JB, Taylor K, et al; for the DURATION-1 Study Group. Exenatide once weekly versus twice daily for the treatment of type 2 diabetes: a randomised, open-label, non-inferiority study. Lancet 2008; 372:1240–1250.
- Garber A, Henry R, Ratner R, et al; for the LEAD-3 (Mono) Study Group. Liraglutide versus glimepiride monotherapy for type 2 diabetes (LEAD-3 Mono): a randomised, 52-week, phase III, double-blind, parallel-treatment trial. Lancet 2009; 373:473–481.
- Nauck M, Frid A, Hermansen K, et al; for the LEAD-2 Study Group. Efficacy and safety comparison of liraglutide, glimepiride, and placebo, all in combination with metformin, in type 2 diabetes: the LEAD (Liraglutide Effect and Action in Diabetes)-2 study. Diabetes Care 2009; 32:84–90.
- Ahrén B, Landin-Olsson M, Jansson PA, Svensson M, Holmes D, Schweizer A. Inhibition of dipeptidyl peptidase-4 reduces glycemia, sustains insulin levels, and reduces glucagon levels in type 2 diabetes. J Clin Endocrinol Metab 2004; 89:2078–2084.
- Herman GA, Stevens C, Van Dyck K, et al. Pharmacokinetics and pharmacodynamics of sitagliptin, an inhibitor of dipeptidyl peptidase IV, in healthy subjects: results from two randomized, double-blind, placebo-controlled studies with single oral doses. Clin Pharmacol Ther 2005; 78:675–688.
- Bohannon N. Overview of the gliptin class (dipeptidyl peptidase-4 inhibitors) in clinical practice. Postgrad Med 2009; 121:40–45.
- US Department of Health and Human Services. FDA approves new drug treatment for type 2 diabetes. US Food and Drug Administration Web site. http://www.fda.gov/newsevents/newsroom/pressannouncements/ucm174780.htm. Published July 31, 2009. Accessed September 18, 2009.
- Raz I, Hanefeld M, Xu L, Caria C, Williams-Herman D, Khatami H; for the Sitagliptin Study 023 Group. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor sitagliptin as monotherapy in patients with type 2 diabetes mellitus. Diabetologia 2006; 49:2564–2571.
- Zerilli T, Pyon EY. Sitagliptin phosphate: a DPP-4 inhibitor for the treatment of type 2 diabetes mellitus. Clin Ther 2007; 29:2614–2634.
- Scott R, Loeys T, Davies MJ, Engel SS; for the Sitagliptin Study 801 Group. Efficacy and safety of sitagliptin when added to ongoing metformin therapy in patients with type 2 diabetes. Diabetes Obes Metab 2008; 10:959–969.
- Williams-Herman D, Johnson J, Teng R, et al. Efficacy and safety of initial combination therapy with sitagliptin and metformin in patients with type 2 diabetes: a 54-week study. Curr Med Res Opin 2009; 25:569–583.
- DeFronzo RA, Okerson T, Viswanathan P, Guan X, Holcombe JH, MacConell L. Effects of exenatide versus sitagliptin on postprandial glucose, insulin and glucagon secretion, gastric emptying, and caloric intake: a randomized, cross-over study. Curr Med Res Opin 2008; 24:2943–2952.
According to the American Association of Clinical Endocrinologists (AACE) and the American Diabetes Association (ADA), glycosylated hemoglobin (HbA1c) in patients with diabetes should be maintained at 6.5% or less (AACE) or at less than 7.0% (ADA). Both organizations support an aggressive stepwise approach that includes medication and lifestyle modification, with strategies and clinical attention devoted to avoiding significant hypoglycemia.1,2 Yet, despite the introduction of new antidiabetes agents, most current management strategies are offset by limitations in achieving and maintaining glycemic targets needed to provide optimal care for patients with diabetes, more than 90% of whom have type 2 diabetes mellitus (T2DM).3,4
Nationally, glycemic control among patients with T2DM has improved but is still far from optimal. According to data from the 1999–2000 National Health and Nutrition Examination Survey (NHANES), glycemic control (HbA1c < 7.0%) rates were 35.8% for patients with T2DM.5 In a more recent report (NHANES 1999–2004), fewer than half (48.4%) of adult patients with diagnosed diabetes achieved HbA1c levels below 7.0%.5,6 Factors contributing to these data include earlier onset and earlier detection of T2DM.7
CHANGING TREATMENT TRENDS
Available treatments for patients with T2DM include secretagogues, such as sulfonylureas and “glinides” (repaglinide and nateglinide), metformin, thiazolidinediones (TZDs), and dipeptidyl peptidase–4 (DPP-4) inhibitors among oral medications, and insulin and glucagon-like peptide–1 (GLP-1) receptor agonists among parenterally administered agents. According to the latest published data on prescribing patterns for patients with T2DM, analyses of the National Disease and Therapeutic Index (1994–2007) and the National Prescription Audit (2001–2007), sulfonylurea use decreased from 67% of treatment visits in 1994 to 34% of visits in 2007.8 By 2007, metformin, used in 54% of treatment visits, and TZDs, used in 28%, were the most frequently administered antidiabetes agents. Insulin use declined from 38% of visits during which a treatment was administered in 1994 to 25% of visits in 2000, but had increased subsequently to 28% of visits in 2007.
SIGNIFICANCE OF CARDIOVASCULAR RISK
Clinical research has suggested that focusing solely on improving glycemic control may be insufficient to reduce overall morbidity and mortality associated with diabetes. Specifically, data from recent studies, including the Action to Control Cardiovascular Risk in Diabetes (ACCORD), the Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE), and the Veterans Affairs Diabetes Trial (VADT), emphasized that lowering HbA1c below 7% in a high-risk population of individuals with T2DM did not improve cardiovascular (CV) outcomes.9–11 The observations confirm that risk factors, including weight, blood pressure (BP), and lipid levels, are vitally important in reducing morbidity and mortality in this population. This perception is further underscored by the NHANES 1999–2004 data, which showed poor concurrent control of HbA1c, BP, and lipids; only 13.2% of patients with diagnosed diabetes achieved all three target goals simultaneously.6 Similarly, a nationwide survey in Norway showed that only 13% of patients with T2DM concurrently achieved goals for HbA1c, BP, and lipids.12
In the Danish Steno-2 Study, patients with T2DM and persistent microalbuminuria were treated with either intensive target-driven therapy using multiple drugs or conventional multifactorial treatment. Over a mean period of 13.3 years (7.8 years of treatment plus 5.5 years of follow-up), intensive multifactorial intervention to control multiple CV risk factors, including HbA1c, BP, and lipids, was associated with a lower risk of death from CV causes (hazard ratio [HR], 0.43; 95% confidence interval [CI], 0.19 to 0.94; P = .04) and a lower risk of CV events (HR, 0.41; 95% CI, 0.25 to 0.67; P < .001) than was conventional therapy.13
This article clarifies the redefinition of treatment success in patients with T2DM based on targeting the underlying physiologic defects of the disease.
T2DM, OVERWEIGHT/OBESITY, AND CV DISEASE: CLOSELY LINKED
The incidence and prevalence of T2DM, overweight/obesity, and CV disease (CVD) are increasing worldwide. It is estimated that the worldwide prevalence of diabetes will increase from 171 million in 2000 to 366 million by 203014; T2DM increases the risk of morbidity and mortality from microvascular (eg, neuropathic, retinopathic, nephropathic) and macrovascular (eg, coronary, peripheral vascular disease) complications.15 According to a Michigan health maintenance organization study (N = 1,364), the median annual direct cost of medical care for Caucasian patients with T2DM who were diet controlled, had a body mass index (BMI) of 30 kg/m2 or higher, and had no vascular complications was estimated to be $1,700 for men and $2,100 for women.16 The actual cost of care for patients with T2DM may be much higher, since most patients present with multiple CV risk factors in addition to being overweight.
NHANES data show that approximately two-thirds of Americans are either overweight or obese17; overweight/obesity affects about 80% of adults diagnosed with T2DM.18 Overweight or obesity can increase the risk for developing T2DM by more than 90-fold and, in women, it can increase the risk for developing coronary heart disease (CHD) by sixfold.19 The close link between T2DM and CVD is underscored further with recent data from the Framingham Heart Study, which showed a high lifetime risk of CVD in patients with diabetes, heightened further by obesity. During the 30-year study period, the lifetime risk of CVD in normal-weight people with diabetes was 78.6% in men and 54.8% in women; the risk increased to 86.9% in obese men with diabetes and to 78.8% in obese women with diabetes.20 The NHANES data also showed that the prevalence of T2DM increased in the past decade and that patients are being diagnosed at a younger age, from a mean age of 52 years in 1988–1994 to 46 years in 1999–2000.7
BRIDGING THE GAP FROM PATHOPHYSIOLOGY TO UNMET NEEDS
The paradigm behind the pathophysiology of T2DM has shifted from its perception as a simple “dual-defect” disease (ie, deficiency in insulin secretion and peripheral tissue insulin resistance) to a multidimensional disorder.1,21 This new model includes overweight/obesity, insulin resistance, qualitative and quantitative defects in insulin secretion, and dysregulation in the secretion of other hormones, including the beta-cell hormone amylin, the alpha-cell hormone glucagon, and the gastrointestinal incretin hormones GLP-1 and glucose-dependent insulinotropic polypeptide.21–23
CLINICAL GUIDELINES AND CV RISK FACTOR MANAGEMENT
The best strategy for managing T2DM is a comprehensive approach that addresses the fundamental core defects plus associated factors that contribute to increased CV risk. Several specialty groups have suggested guidelines and algorithms for the management of T2DM and its comorbidities. These guidelines, including the ADA standards of medical care, the AACE standards in tandem with the American College of Endocrinology guidelines, and the recent joint statement from the ADA and the European Association for the Study of Diabetes (EASD), acknowledge that the core defects of T2DM and the associated CV risk factors (eg, weight gain, obesity, hypertension, dyslipidemia) are important in developing optimal treatment strategies.1–3 Medical nutrition guidelines advocate weight loss as a key initial step in managing T2DM and the comorbidities that lead to elevated CV risk.25,26 The National Institutes of Health and the US Department of Health and Human Services/US Department of Agriculture advocate regular physical activity, dietary assessment, and periodic comorbidity and weight assessment for all people, not just those with T2DM or CVD.26,27
Weight reduction
Evidence in support of effective lifestyle intervention was demonstrated in the Action for Health in Diabetes (Look AHEAD) study. After 1 year, patients with T2DM treated with intensive lifestyle intervention lost an average of 8.6% of their initial weight compared with 0.7% in patients treated only with diabetes support and education (P < 0.001). The intensive-intervention patients also had a significant drop in HbA1c (from 7.3% to 6.6%; P < 0.001) and were able to reduce their antidiabetes, antihypertensive, and lipid-lowering medications.28 More recent data from the Look AHEAD study reported that overweight patients with T2DM enrolled in a weight management program experienced significant weight loss, improved physical fitness, reduced physical symptoms, and overall improvement in health-related quality of life.29 Thus, weight reduction appears to be a key component in reducing CV risk and improving quality of life in most patients with T2DM.28–30
Hypertension
Hypertension is a major risk factor for microvascular complications and CVD, and may be associated with, or be the underlying result of, nephropathy.2 BP control is clearly important in reducing the morbidity and mortality associated with T2DM. The recommended BP goal in patients with T2DM is less than 130/80 mm Hg.1,2
Hyperlipidemia
According to the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III [ATP III]), diabetes is considered a CHD risk equivalent because it confers a high risk of new CHD developing within 10 years.31 In addition to the NCEP–ATP III guidelines, the ADA and the AACE have set target levels for lipids in patients with diabetes, including T2DM.1,2,31 All three organizations have defined 100 mg/dL as the target level for low-density lipoprotein.
HbA1c and lifestyle intervention
EVOLUTION OF ANTIDIABETES THERAPIES
Traditional antidiabetes agents used in the treatment of patients with T2DM have focused mainly on insulin secretion and insulin resistance, with treatment success defined as achieving HbA1c goals with a reduced incidence of hypoglycemia.23 Secretagogues, such as sulfonylureas and glinides, stimulate the pancreas to release insulin. Insulin sensitizers, such as TZDs and metformin, enhance the action of insulin in muscle and fat1,3,23 and lower hepatic glucose production. The alpha-glucosidase inhibitors alter carbohydrate absorption from the gastrointestinal tract.1 The extent to which each agent achieves treatment success in terms of glucose lowering depends on several factors, including intrinsic attributes, duration of disease, and baseline glycemic control.3
GLP-1 receptor agonists
Exenatide effects. Although many agents are in development, to date exenatide is the only GLP-1 receptor agonist approved by the US Food and Drug Administration (FDA).8,33 Exenatide is an exendin-4 GLP-1 receptor agonist with multiple glucoregulatory effects, including enhanced glucose-dependent insulin secretion, reduced glucagon secretion and food intake, and slowed gastric emptying.22,34 Exenatide is detectable in the circulation for up to 10 hours following subcutaneous (SC) administration22 and has a greater potency in reducing plasma glucose than GLP-1 in preclinical studies.35,36
By virtue of its beneficial effects on glycemic control, weight, BP, and lipids, exenatide addresses some of the components of the metabolic syndrome.37–41 In pivotal 30-week studies, exenatide was associated with HbA1c reductions that ranged from –0.40% to –0.86% from baseline and decreases in body weight of approximately –1 kg to –3 kg from baseline, without severe hypoglycemia.37–39 The percentage of patients who reached the ADA goal of HbA1c less than 7.0% at 30 weeks ranged from 24% to 34%. The addition of exenatide to TZD therapy in a 16-week study was associated with mean reductions in HbA1c of –0.98%, fasting plasma glucose (FPG) concentration of –1.69 mmol/L (–30.42 mg/dL), and body weight of –1.51 kg.40
A posthoc analysis of an open-label extension study involving patients who completed the original 30-week placebo-controlled studies showed that 46% of patients who remained on exenatide achieved the ADA goal of HbA1c less than 7.0% at 3 years.41 Exenatide administered for up to 3.5 years was associated with sustained reductions in HbA1c of –1.0% (P < .0001) and body weight of –5.3 kg (P < .001). Pancreatic beta-cell function, assessed by homeostasis model assessment, improved, as did BP, triglyceride, high-density lipoprotein, low-density lipoprotein, and aspartate aminotransferase levels.41
Comparison with insulin analogues. Comparative studies have highlighted the contrasting effects of exenatide and insulin analogues (eg, insulin glargine and fixed-ratio insulin).42–45 In a 26-week trial comparing exenatide with insulin glargine in subjects with T2DM, both agents resulted in similar decreases in HbA1c. Exenatide was also associated with a –2.3-kg weight reduction, whereas insulin glargine was associated with a +1.8-kg weight gain.42 Although rates of symptomatic hypoglycemia were similar, there were fewer cases of nocturnal hypoglycemia with exenatide (0.9 event/patient-year vs 2.4 events/patient-year with insulin).
In a 32-week study comparing exenatide BID with titrated insulin glargine QD, the HbA1c reductions for exenatide and insulin glargine were comparable. However, body weight decreased –4.2 kg over two 16-week treatment periods with exenatide, but increased +3.3 kg over the same periods with the basal insulin analogue.43 The incidence of hypoglycemia was lower with exenatide than with insulin glargine (14.7% vs 25.2%), although the difference was not statistically significant.
In another study that compared exenatide with biphasic insulin aspart, patients who were treated with exenatide also lost weight while those who received the fast-acting insulin analogue gained weight (between-group difference, –5.4 kg). Patients treated with exenatide also demonstrated greater reductions in postprandial plasma glucose (PPG) excursions following their morning (P < .001), midday (P = .002), and evening meals (P < .001).44 Overall, hypoglycemia rates were similar at study end between exenatide and insulin aspart (4.7 events/patient-year vs 5.6 events/patient-year). In all of these studies, significant gastrointestinal adverse events (nausea and vomiting) occurred more frequently with exenatide, and more patients withdrew from exenatide than from insulin.
Formulations in development. Other advances in GLP-1 receptor agonist therapy include novel formulations under clinical development, such as exenatide once weekly36,46 and liraglutide, a human analogue GLP-1 receptor agonist formulated for once-daily administration.47,48 In a 52-week study in patients with T2DM, liraglutide significantly reduced HbA1c; the 1.2-mg SC QD dosage reduced HBA1c by –0.84% (P = .0014) and the 1.8-mg SC QD dosage by –1.14% (P < .0001). In comparison, glimepiride 8 mg orally QD achieved a –0.51% reduction. Liraglutide was also associated with greater reductions in weight, hypoglycemia, and systolic BP than glimepiride.47
A 26-week study compared liraglutide (0.6, 1.2, and 1.8 mg SC QD), placebo, and glimepiride 4 mg QD in combination with metformin 1 g BID. HbA1c was reduced significantly in all liraglutide groups compared with placebo (P < .0001). Mean HbA1c decreased –1.0% with liraglutide 1.2 mg and 1.8 mg and with glimepiride; it decreased –0.7% with liraglutide 0.6 mg; and it increased +0.1% with placebo. Body weight decreased –1.8 kg to –2.8 kg in all liraglutide groups but increased +1.0 kg in the glimepiride group (P < .0001). The incidence of minor hypoglycemia with liraglutide (~3%) was comparable to that observed with placebo but less than that with glimepiride (17%; P < .001).48
A once-weekly long-acting release (LAR) formulation of exenatide submitted to the FDA for approval may provide enhanced glycemic and weight control, potentially improving patient acceptance and adherence.36,46 In a 15-week study, exenatide once weekly produced significant reductions in HbA1c, FPG, PPG, and body weight. There were no withdrawals due to adverse events, and the formation of anti-exenatide antibodies was not predictive of therapeutic end point response or adverse safety outcome. Instances of hypoglycemia were mild and not dose related.36 In a 30-week study comparing exenatide LAR once weekly with exenatide BID, patients given exenatide LAR once weekly had significantly greater HbA1c reductions than did patients given exenatide BID (–1.9% vs –1.5%; P = .0023). Treatment adherence was 98% with both exenatide regimens, and no episodes of major hypoglycemia occurred with either formulation regardless of background sulfonylurea use. Favorable effects on BP and lipid profile were observed with both exenatide regimens.46
DPP-4 inhibitors
The DPP-4 inhibitors (commonly called gliptins) inhibit the proteolytic cleavage of circulating GLP-1 by binding to the DPP-4 enzyme, increasing the concentration of endogenous GLP-1 approximately two- to threefold.49–51 These concentrations result in more prompt and appropriate secretion of insulin and suppression of glucagon in response to a carbohydrate-containing snack or meal, with the change in glucagon correlating linearly with improved glucose tolerance.51
DPP-4 inhibitors, which are given orally, include sitagliptin and saxagliptin (approved in the United States) and vildagliptin (not approved in the United States but used in the European Union and Latin America).8,22,33,52 Sitagliptin can be used either as monotherapy or in combination with metformin or a TZD.8,49–55 Recently, a single-tablet formulation of sitagliptin plus metformin was granted regulatory approval.8
When used alone or in combination with metformin or pioglitazone, sitagliptin has been associated with significant reductions in HbA1c (of ~0.5% to 0.6% when used alone, ~0.7% with metformin, and ~0.9% with pioglitazone [P < .001 vs placebo]), with hypoglycemia occurring in 1.3% or less of the population.54 In an 18-week study in which patients with T2DM who were inadequately controlled with metformin monotherapy were randomized to receive add-on sitagliptin (100 mg QD), rosiglitazone (8 mg QD), or placebo, sitagliptin reduced HbA1c –0.73% (P < .001 vs placebo) and reduced body weight –0.4 kg, while rosiglitazone reduced HbA1c –0.79% and increased body weight +1.5 kg.55
To evaluate the effectiveness of sitagliptin and metformin as initial therapy, a 54-week study was completed in 885 patients with T2DM and inadequate glycemic control (HbA1c 7.5–11%) on diet and exercise.56 Patients were evaluated on monotherapy with either sitagliptin (100 mg QD) or metformin (1 g or 2 g QD), or on initial therapy with the two in combination (sitagliptin 100 mg + metformin 1 mg or 2 mg QD). At week 54, in the all-patients-treated analysis, mean changes in HbA1c from baseline were –1.8% with sitagliptin plus metformin 2 g QD, –1.4% with sitagliptin plus metformin 1 g QD, –1.3% with metformin 2 g QD monotherapy, –1.0% with metformin 1 g QD monotherapy, and –0.8% with sitagliptin 100 mg QD monotherapy.
All treatments improved measures of beta-cell function (eg, homeostasis model assessment [HOMA]-beta, proinsulin/insulin ratio). Mean body weight decreased from baseline in the combination and metformin monotherapy groups and was unchanged from baseline in the sitagliptin monotherapy group. The incidence of hypoglycemia was low (1%–3%) across treatment groups. The incidence of gastrointestinal adverse experiences was evaluated with the coadministration of sitagliptin and metformin and appeared similar to that observed with use of metformin as monotherapy.56 Thus, this study suggested that an initial combination of a DPP-IV inhibitor with metformin can improve glycemic control and markers of beta-cell function in patients with T2DM.
Incretin-based therapies compared
Studies in both healthy individuals and in patients with T2DM have shown that oral DPP-4 inhibitors such as sitagliptin increase endogenous GLP-1 concentrations by about twofold compared with placebo.22,50 The pharmacologic concentration of subcutaneously administered exenatide available for activating the GLP-1 receptor is significantly greater than the increased endogenous GLP-1 concentrations achieved with sitagliptin. In a recent clinical study comparing exenatide and sitagliptin in patients with T2DM, the mean 2-hour plasma concentration for exenatide was 64 pM compared with the mean 2-hour postprandial GLP-1 concentration of 15 pM for sitagliptin (baseline GLP-1 concentration was 7.2 pM).57 While both agents were shown to be effective, exenatide appeared to have had a greater effect than sitagliptin in increasing insulin secretion and reducing postprandial glucagon secretion, leading to significantly (P < 0.0001) greater reductions in PPG.57
Sitagliptin has been minimally associated with nausea, whereas patients who take exenatide need to be informed of the risk of usually mild to moderate, but sometimes severe, nausea and vomiting that tends to decrease over time.
For a detailed comparison of the effects of GLP-1 receptor agonists and DPP-4 inhibitors on HbA1c, weight, and hypoglycemia, see “Advances in therapy for type 2 diabetes: GLP–1 receptor agonists and DPP–4 inhibitors.”
CONCLUSION
Despite advances in diagnosis and treatment, T2DM, overweight/obesity, CVD, and their complications remain major public health burdens worldwide. The concepts that explain the pathophysiology of T2DM include the contribution of various factors beyond insulin secretion and insulin resistance, such as the role of incretin hormones in disease progression. A comprehensive approach to managing patients with T2DM requires targeting the fundamental defects of the disease and its comorbidities. Newer agents, including incretin-based therapies such as GLP-1 receptor agonists and DPP-4 inhibitors, address the fundamental defects of T2DM. The definition of treatment success in the management of T2DM will be redefined as more data become available on agents that exert beneficial effects not only on glycemia but on parameters that may influence overall CV health, such as weight, BP, and lipid profiles.
According to the American Association of Clinical Endocrinologists (AACE) and the American Diabetes Association (ADA), glycosylated hemoglobin (HbA1c) in patients with diabetes should be maintained at 6.5% or less (AACE) or at less than 7.0% (ADA). Both organizations support an aggressive stepwise approach that includes medication and lifestyle modification, with strategies and clinical attention devoted to avoiding significant hypoglycemia.1,2 Yet, despite the introduction of new antidiabetes agents, most current management strategies are offset by limitations in achieving and maintaining glycemic targets needed to provide optimal care for patients with diabetes, more than 90% of whom have type 2 diabetes mellitus (T2DM).3,4
Nationally, glycemic control among patients with T2DM has improved but is still far from optimal. According to data from the 1999–2000 National Health and Nutrition Examination Survey (NHANES), glycemic control (HbA1c < 7.0%) rates were 35.8% for patients with T2DM.5 In a more recent report (NHANES 1999–2004), fewer than half (48.4%) of adult patients with diagnosed diabetes achieved HbA1c levels below 7.0%.5,6 Factors contributing to these data include earlier onset and earlier detection of T2DM.7
CHANGING TREATMENT TRENDS
Available treatments for patients with T2DM include secretagogues, such as sulfonylureas and “glinides” (repaglinide and nateglinide), metformin, thiazolidinediones (TZDs), and dipeptidyl peptidase–4 (DPP-4) inhibitors among oral medications, and insulin and glucagon-like peptide–1 (GLP-1) receptor agonists among parenterally administered agents. According to the latest published data on prescribing patterns for patients with T2DM, analyses of the National Disease and Therapeutic Index (1994–2007) and the National Prescription Audit (2001–2007), sulfonylurea use decreased from 67% of treatment visits in 1994 to 34% of visits in 2007.8 By 2007, metformin, used in 54% of treatment visits, and TZDs, used in 28%, were the most frequently administered antidiabetes agents. Insulin use declined from 38% of visits during which a treatment was administered in 1994 to 25% of visits in 2000, but had increased subsequently to 28% of visits in 2007.
SIGNIFICANCE OF CARDIOVASCULAR RISK
Clinical research has suggested that focusing solely on improving glycemic control may be insufficient to reduce overall morbidity and mortality associated with diabetes. Specifically, data from recent studies, including the Action to Control Cardiovascular Risk in Diabetes (ACCORD), the Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE), and the Veterans Affairs Diabetes Trial (VADT), emphasized that lowering HbA1c below 7% in a high-risk population of individuals with T2DM did not improve cardiovascular (CV) outcomes.9–11 The observations confirm that risk factors, including weight, blood pressure (BP), and lipid levels, are vitally important in reducing morbidity and mortality in this population. This perception is further underscored by the NHANES 1999–2004 data, which showed poor concurrent control of HbA1c, BP, and lipids; only 13.2% of patients with diagnosed diabetes achieved all three target goals simultaneously.6 Similarly, a nationwide survey in Norway showed that only 13% of patients with T2DM concurrently achieved goals for HbA1c, BP, and lipids.12
In the Danish Steno-2 Study, patients with T2DM and persistent microalbuminuria were treated with either intensive target-driven therapy using multiple drugs or conventional multifactorial treatment. Over a mean period of 13.3 years (7.8 years of treatment plus 5.5 years of follow-up), intensive multifactorial intervention to control multiple CV risk factors, including HbA1c, BP, and lipids, was associated with a lower risk of death from CV causes (hazard ratio [HR], 0.43; 95% confidence interval [CI], 0.19 to 0.94; P = .04) and a lower risk of CV events (HR, 0.41; 95% CI, 0.25 to 0.67; P < .001) than was conventional therapy.13
This article clarifies the redefinition of treatment success in patients with T2DM based on targeting the underlying physiologic defects of the disease.
T2DM, OVERWEIGHT/OBESITY, AND CV DISEASE: CLOSELY LINKED
The incidence and prevalence of T2DM, overweight/obesity, and CV disease (CVD) are increasing worldwide. It is estimated that the worldwide prevalence of diabetes will increase from 171 million in 2000 to 366 million by 203014; T2DM increases the risk of morbidity and mortality from microvascular (eg, neuropathic, retinopathic, nephropathic) and macrovascular (eg, coronary, peripheral vascular disease) complications.15 According to a Michigan health maintenance organization study (N = 1,364), the median annual direct cost of medical care for Caucasian patients with T2DM who were diet controlled, had a body mass index (BMI) of 30 kg/m2 or higher, and had no vascular complications was estimated to be $1,700 for men and $2,100 for women.16 The actual cost of care for patients with T2DM may be much higher, since most patients present with multiple CV risk factors in addition to being overweight.
NHANES data show that approximately two-thirds of Americans are either overweight or obese17; overweight/obesity affects about 80% of adults diagnosed with T2DM.18 Overweight or obesity can increase the risk for developing T2DM by more than 90-fold and, in women, it can increase the risk for developing coronary heart disease (CHD) by sixfold.19 The close link between T2DM and CVD is underscored further with recent data from the Framingham Heart Study, which showed a high lifetime risk of CVD in patients with diabetes, heightened further by obesity. During the 30-year study period, the lifetime risk of CVD in normal-weight people with diabetes was 78.6% in men and 54.8% in women; the risk increased to 86.9% in obese men with diabetes and to 78.8% in obese women with diabetes.20 The NHANES data also showed that the prevalence of T2DM increased in the past decade and that patients are being diagnosed at a younger age, from a mean age of 52 years in 1988–1994 to 46 years in 1999–2000.7
BRIDGING THE GAP FROM PATHOPHYSIOLOGY TO UNMET NEEDS
The paradigm behind the pathophysiology of T2DM has shifted from its perception as a simple “dual-defect” disease (ie, deficiency in insulin secretion and peripheral tissue insulin resistance) to a multidimensional disorder.1,21 This new model includes overweight/obesity, insulin resistance, qualitative and quantitative defects in insulin secretion, and dysregulation in the secretion of other hormones, including the beta-cell hormone amylin, the alpha-cell hormone glucagon, and the gastrointestinal incretin hormones GLP-1 and glucose-dependent insulinotropic polypeptide.21–23
CLINICAL GUIDELINES AND CV RISK FACTOR MANAGEMENT
The best strategy for managing T2DM is a comprehensive approach that addresses the fundamental core defects plus associated factors that contribute to increased CV risk. Several specialty groups have suggested guidelines and algorithms for the management of T2DM and its comorbidities. These guidelines, including the ADA standards of medical care, the AACE standards in tandem with the American College of Endocrinology guidelines, and the recent joint statement from the ADA and the European Association for the Study of Diabetes (EASD), acknowledge that the core defects of T2DM and the associated CV risk factors (eg, weight gain, obesity, hypertension, dyslipidemia) are important in developing optimal treatment strategies.1–3 Medical nutrition guidelines advocate weight loss as a key initial step in managing T2DM and the comorbidities that lead to elevated CV risk.25,26 The National Institutes of Health and the US Department of Health and Human Services/US Department of Agriculture advocate regular physical activity, dietary assessment, and periodic comorbidity and weight assessment for all people, not just those with T2DM or CVD.26,27
Weight reduction
Evidence in support of effective lifestyle intervention was demonstrated in the Action for Health in Diabetes (Look AHEAD) study. After 1 year, patients with T2DM treated with intensive lifestyle intervention lost an average of 8.6% of their initial weight compared with 0.7% in patients treated only with diabetes support and education (P < 0.001). The intensive-intervention patients also had a significant drop in HbA1c (from 7.3% to 6.6%; P < 0.001) and were able to reduce their antidiabetes, antihypertensive, and lipid-lowering medications.28 More recent data from the Look AHEAD study reported that overweight patients with T2DM enrolled in a weight management program experienced significant weight loss, improved physical fitness, reduced physical symptoms, and overall improvement in health-related quality of life.29 Thus, weight reduction appears to be a key component in reducing CV risk and improving quality of life in most patients with T2DM.28–30
Hypertension
Hypertension is a major risk factor for microvascular complications and CVD, and may be associated with, or be the underlying result of, nephropathy.2 BP control is clearly important in reducing the morbidity and mortality associated with T2DM. The recommended BP goal in patients with T2DM is less than 130/80 mm Hg.1,2
Hyperlipidemia
According to the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III [ATP III]), diabetes is considered a CHD risk equivalent because it confers a high risk of new CHD developing within 10 years.31 In addition to the NCEP–ATP III guidelines, the ADA and the AACE have set target levels for lipids in patients with diabetes, including T2DM.1,2,31 All three organizations have defined 100 mg/dL as the target level for low-density lipoprotein.
HbA1c and lifestyle intervention
EVOLUTION OF ANTIDIABETES THERAPIES
Traditional antidiabetes agents used in the treatment of patients with T2DM have focused mainly on insulin secretion and insulin resistance, with treatment success defined as achieving HbA1c goals with a reduced incidence of hypoglycemia.23 Secretagogues, such as sulfonylureas and glinides, stimulate the pancreas to release insulin. Insulin sensitizers, such as TZDs and metformin, enhance the action of insulin in muscle and fat1,3,23 and lower hepatic glucose production. The alpha-glucosidase inhibitors alter carbohydrate absorption from the gastrointestinal tract.1 The extent to which each agent achieves treatment success in terms of glucose lowering depends on several factors, including intrinsic attributes, duration of disease, and baseline glycemic control.3
GLP-1 receptor agonists
Exenatide effects. Although many agents are in development, to date exenatide is the only GLP-1 receptor agonist approved by the US Food and Drug Administration (FDA).8,33 Exenatide is an exendin-4 GLP-1 receptor agonist with multiple glucoregulatory effects, including enhanced glucose-dependent insulin secretion, reduced glucagon secretion and food intake, and slowed gastric emptying.22,34 Exenatide is detectable in the circulation for up to 10 hours following subcutaneous (SC) administration22 and has a greater potency in reducing plasma glucose than GLP-1 in preclinical studies.35,36
By virtue of its beneficial effects on glycemic control, weight, BP, and lipids, exenatide addresses some of the components of the metabolic syndrome.37–41 In pivotal 30-week studies, exenatide was associated with HbA1c reductions that ranged from –0.40% to –0.86% from baseline and decreases in body weight of approximately –1 kg to –3 kg from baseline, without severe hypoglycemia.37–39 The percentage of patients who reached the ADA goal of HbA1c less than 7.0% at 30 weeks ranged from 24% to 34%. The addition of exenatide to TZD therapy in a 16-week study was associated with mean reductions in HbA1c of –0.98%, fasting plasma glucose (FPG) concentration of –1.69 mmol/L (–30.42 mg/dL), and body weight of –1.51 kg.40
A posthoc analysis of an open-label extension study involving patients who completed the original 30-week placebo-controlled studies showed that 46% of patients who remained on exenatide achieved the ADA goal of HbA1c less than 7.0% at 3 years.41 Exenatide administered for up to 3.5 years was associated with sustained reductions in HbA1c of –1.0% (P < .0001) and body weight of –5.3 kg (P < .001). Pancreatic beta-cell function, assessed by homeostasis model assessment, improved, as did BP, triglyceride, high-density lipoprotein, low-density lipoprotein, and aspartate aminotransferase levels.41
Comparison with insulin analogues. Comparative studies have highlighted the contrasting effects of exenatide and insulin analogues (eg, insulin glargine and fixed-ratio insulin).42–45 In a 26-week trial comparing exenatide with insulin glargine in subjects with T2DM, both agents resulted in similar decreases in HbA1c. Exenatide was also associated with a –2.3-kg weight reduction, whereas insulin glargine was associated with a +1.8-kg weight gain.42 Although rates of symptomatic hypoglycemia were similar, there were fewer cases of nocturnal hypoglycemia with exenatide (0.9 event/patient-year vs 2.4 events/patient-year with insulin).
In a 32-week study comparing exenatide BID with titrated insulin glargine QD, the HbA1c reductions for exenatide and insulin glargine were comparable. However, body weight decreased –4.2 kg over two 16-week treatment periods with exenatide, but increased +3.3 kg over the same periods with the basal insulin analogue.43 The incidence of hypoglycemia was lower with exenatide than with insulin glargine (14.7% vs 25.2%), although the difference was not statistically significant.
In another study that compared exenatide with biphasic insulin aspart, patients who were treated with exenatide also lost weight while those who received the fast-acting insulin analogue gained weight (between-group difference, –5.4 kg). Patients treated with exenatide also demonstrated greater reductions in postprandial plasma glucose (PPG) excursions following their morning (P < .001), midday (P = .002), and evening meals (P < .001).44 Overall, hypoglycemia rates were similar at study end between exenatide and insulin aspart (4.7 events/patient-year vs 5.6 events/patient-year). In all of these studies, significant gastrointestinal adverse events (nausea and vomiting) occurred more frequently with exenatide, and more patients withdrew from exenatide than from insulin.
Formulations in development. Other advances in GLP-1 receptor agonist therapy include novel formulations under clinical development, such as exenatide once weekly36,46 and liraglutide, a human analogue GLP-1 receptor agonist formulated for once-daily administration.47,48 In a 52-week study in patients with T2DM, liraglutide significantly reduced HbA1c; the 1.2-mg SC QD dosage reduced HBA1c by –0.84% (P = .0014) and the 1.8-mg SC QD dosage by –1.14% (P < .0001). In comparison, glimepiride 8 mg orally QD achieved a –0.51% reduction. Liraglutide was also associated with greater reductions in weight, hypoglycemia, and systolic BP than glimepiride.47
A 26-week study compared liraglutide (0.6, 1.2, and 1.8 mg SC QD), placebo, and glimepiride 4 mg QD in combination with metformin 1 g BID. HbA1c was reduced significantly in all liraglutide groups compared with placebo (P < .0001). Mean HbA1c decreased –1.0% with liraglutide 1.2 mg and 1.8 mg and with glimepiride; it decreased –0.7% with liraglutide 0.6 mg; and it increased +0.1% with placebo. Body weight decreased –1.8 kg to –2.8 kg in all liraglutide groups but increased +1.0 kg in the glimepiride group (P < .0001). The incidence of minor hypoglycemia with liraglutide (~3%) was comparable to that observed with placebo but less than that with glimepiride (17%; P < .001).48
A once-weekly long-acting release (LAR) formulation of exenatide submitted to the FDA for approval may provide enhanced glycemic and weight control, potentially improving patient acceptance and adherence.36,46 In a 15-week study, exenatide once weekly produced significant reductions in HbA1c, FPG, PPG, and body weight. There were no withdrawals due to adverse events, and the formation of anti-exenatide antibodies was not predictive of therapeutic end point response or adverse safety outcome. Instances of hypoglycemia were mild and not dose related.36 In a 30-week study comparing exenatide LAR once weekly with exenatide BID, patients given exenatide LAR once weekly had significantly greater HbA1c reductions than did patients given exenatide BID (–1.9% vs –1.5%; P = .0023). Treatment adherence was 98% with both exenatide regimens, and no episodes of major hypoglycemia occurred with either formulation regardless of background sulfonylurea use. Favorable effects on BP and lipid profile were observed with both exenatide regimens.46
DPP-4 inhibitors
The DPP-4 inhibitors (commonly called gliptins) inhibit the proteolytic cleavage of circulating GLP-1 by binding to the DPP-4 enzyme, increasing the concentration of endogenous GLP-1 approximately two- to threefold.49–51 These concentrations result in more prompt and appropriate secretion of insulin and suppression of glucagon in response to a carbohydrate-containing snack or meal, with the change in glucagon correlating linearly with improved glucose tolerance.51
DPP-4 inhibitors, which are given orally, include sitagliptin and saxagliptin (approved in the United States) and vildagliptin (not approved in the United States but used in the European Union and Latin America).8,22,33,52 Sitagliptin can be used either as monotherapy or in combination with metformin or a TZD.8,49–55 Recently, a single-tablet formulation of sitagliptin plus metformin was granted regulatory approval.8
When used alone or in combination with metformin or pioglitazone, sitagliptin has been associated with significant reductions in HbA1c (of ~0.5% to 0.6% when used alone, ~0.7% with metformin, and ~0.9% with pioglitazone [P < .001 vs placebo]), with hypoglycemia occurring in 1.3% or less of the population.54 In an 18-week study in which patients with T2DM who were inadequately controlled with metformin monotherapy were randomized to receive add-on sitagliptin (100 mg QD), rosiglitazone (8 mg QD), or placebo, sitagliptin reduced HbA1c –0.73% (P < .001 vs placebo) and reduced body weight –0.4 kg, while rosiglitazone reduced HbA1c –0.79% and increased body weight +1.5 kg.55
To evaluate the effectiveness of sitagliptin and metformin as initial therapy, a 54-week study was completed in 885 patients with T2DM and inadequate glycemic control (HbA1c 7.5–11%) on diet and exercise.56 Patients were evaluated on monotherapy with either sitagliptin (100 mg QD) or metformin (1 g or 2 g QD), or on initial therapy with the two in combination (sitagliptin 100 mg + metformin 1 mg or 2 mg QD). At week 54, in the all-patients-treated analysis, mean changes in HbA1c from baseline were –1.8% with sitagliptin plus metformin 2 g QD, –1.4% with sitagliptin plus metformin 1 g QD, –1.3% with metformin 2 g QD monotherapy, –1.0% with metformin 1 g QD monotherapy, and –0.8% with sitagliptin 100 mg QD monotherapy.
All treatments improved measures of beta-cell function (eg, homeostasis model assessment [HOMA]-beta, proinsulin/insulin ratio). Mean body weight decreased from baseline in the combination and metformin monotherapy groups and was unchanged from baseline in the sitagliptin monotherapy group. The incidence of hypoglycemia was low (1%–3%) across treatment groups. The incidence of gastrointestinal adverse experiences was evaluated with the coadministration of sitagliptin and metformin and appeared similar to that observed with use of metformin as monotherapy.56 Thus, this study suggested that an initial combination of a DPP-IV inhibitor with metformin can improve glycemic control and markers of beta-cell function in patients with T2DM.
Incretin-based therapies compared
Studies in both healthy individuals and in patients with T2DM have shown that oral DPP-4 inhibitors such as sitagliptin increase endogenous GLP-1 concentrations by about twofold compared with placebo.22,50 The pharmacologic concentration of subcutaneously administered exenatide available for activating the GLP-1 receptor is significantly greater than the increased endogenous GLP-1 concentrations achieved with sitagliptin. In a recent clinical study comparing exenatide and sitagliptin in patients with T2DM, the mean 2-hour plasma concentration for exenatide was 64 pM compared with the mean 2-hour postprandial GLP-1 concentration of 15 pM for sitagliptin (baseline GLP-1 concentration was 7.2 pM).57 While both agents were shown to be effective, exenatide appeared to have had a greater effect than sitagliptin in increasing insulin secretion and reducing postprandial glucagon secretion, leading to significantly (P < 0.0001) greater reductions in PPG.57
Sitagliptin has been minimally associated with nausea, whereas patients who take exenatide need to be informed of the risk of usually mild to moderate, but sometimes severe, nausea and vomiting that tends to decrease over time.
For a detailed comparison of the effects of GLP-1 receptor agonists and DPP-4 inhibitors on HbA1c, weight, and hypoglycemia, see “Advances in therapy for type 2 diabetes: GLP–1 receptor agonists and DPP–4 inhibitors.”
CONCLUSION
Despite advances in diagnosis and treatment, T2DM, overweight/obesity, CVD, and their complications remain major public health burdens worldwide. The concepts that explain the pathophysiology of T2DM include the contribution of various factors beyond insulin secretion and insulin resistance, such as the role of incretin hormones in disease progression. A comprehensive approach to managing patients with T2DM requires targeting the fundamental defects of the disease and its comorbidities. Newer agents, including incretin-based therapies such as GLP-1 receptor agonists and DPP-4 inhibitors, address the fundamental defects of T2DM. The definition of treatment success in the management of T2DM will be redefined as more data become available on agents that exert beneficial effects not only on glycemia but on parameters that may influence overall CV health, such as weight, BP, and lipid profiles.
- AACE Diabetes Mellitus Clinical Practice Guidelines Task Force. American Association of Clinical Endocrinologists medical guidelines for clinical practice for the management of diabetes mellitus. Endocr Pract 2007; 13(suppl 1):S4–S68.
- American Diabetes Association. Standards of medical care in diabetes—2009. Diabetes Care 2009; 32(suppl 1):S13–S61.
- Nathan DM, Buse JB, Davidson MB, et al. Medical management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 2009; 32:193–203.
- National Institute of Diabetes and Digestive and Kidney Diseases. National Diabetes Statistics, 2007 fact sheet. National Institutes of Health Web site. http://www.diabetes.niddk.nih.gov/dm/pubs/statistics/index.htm. Published 2008. Accessed September 16, 2009.
- Koro CE, Bowlin SJ, Bourgeois N, Fedder DO. Glycemic control from 1988 to 2000 among US adults diagnosed with type 2 diabetes: a preliminary report. Diabetes Care 2004; 27:17–20.
- Ong KL, Cheung BM, Wong LY, Wat NM, Tan KC, Lam KS. Prevalence, treatment, and control of diagnosed diabetes in the US National Health and Nutrition Examination Survey 1999–2004. Ann Epidemiol 2008; 18:222–229.
- Koopman RJ, Mainous AG III, Diaz VA, Geesey ME. Changes in age at diagnosis of type 2 diabetes mellitus in the United States, 1988 to 2000. Ann Fam Med 2005; 3:60–63.
- Alexander GC, Sehgal NL, Moloney RM, Stafford RS. National trends in treatment of type 2 diabetes mellitus, 1994–2007. Arch Intern Med 2008; 168:2088–2094.
- The Action to Control Cardiovascular Risk in Diabetes Study Group. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008; 358:2545–2559.
- The ADVANCE Collaborative Group. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008; 358:2560–2572.
- Duckworth W, Abraira C, Moritz T, et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med 2009; 360:129–139.
- Jenssen TG, Tonstad S, Claudi T, Midthjell K, Cooper J. The gap between guidelines and practice in the treatment of type 2 diabetes: a nationwide survey in Norway. Diabetes Res Clin Pract 2008; 80:314–320.
- Gaede P, Lund-Andersen H, Parving HH, Pedersen O. Effect of a multifactorial intervention on mortality in type 2 diabetes. N Engl J Med 2008; 358:580–591.
- Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care 2004; 27:1047–1053.
- Rosenstock J. Management of type 2 diabetes mellitus in the elderly: special considerations. Drugs Aging 2001; 18:31–44.
- Brandle M, Zhou H, Smith BR, et al. The direct medical cost of type 2 diabetes. Diabetes Care 2003; 26:2300–2304.
- National Center for Health Statistics. Prevalence of overweight and obesity among adults: United States 2003–2004. Centers for Disease Contral and Prevention Web site. http://www.cdc.gov/nchs/products/pubs/pubd/hestats/overweight/overwght_adult_03.htm. Published: April 2006. Accessed September 23, 2009.
- Van Gaal LF, Gutkin SW, Nauck MA. Exploiting the antidiabetic properties of incretins to treat type 2 diabetes mellitus: glucagon-like peptide 1 receptor agonists or insulin for patients with inadequate glycemic control. Eur J Endocrinol 2008; 158:773–784.
- Anderson JW, Kendall CW, Jenkins DJ. Importance of weight management in type 2 diabetes: review with meta-analysis of clinical studies. J Am Coll Nutr 2003; 22:331–339.
- Fox CS, Pencina MJ, Wilson PW, Paynter NP, Vasan RS, D’Agostino RB Sr. Lifetime risk of cardiovascular disease among individuals with and without diabetes stratified by obesity status in the Framingham heart study. Diabetes Care 2008; 31:1582–1584.
- DeFronzo RA. From the triumvirate to the ominous octet: a new paradigm for the treatment of type 2 diabetes mellitus. Diabetes 2009; 58:773–795.
- Stonehouse A, Okerson T, Kendall D, Maggs D. Emerging incretin based therapies for type 2 diabetes: incretin mimetics and DPP-4 inhibitors. Curr Diabetes Rev 2008; 4:101–109.
- Cefalu WT. Pharmacotherapy for the treatment of patients with type 2 diabetes mellitus: rationale and specific agents. Clin Pharmacol Ther 2007; 81:636–649.
- Henry RR. Evolving concepts of type 2 diabetes management with oral medications: new approaches to an old disease. Curr Med Res Opin 2008; 24:2189–2202.
- American Diabetes Association. Nutrition recommendations and interventions for diabetes: a position statement of the American Diabetes Association. Diabetes Care 2008; 31(suppl 1):S61−S78.
- US Department of Health and Human Services (HHS) and US Department of Agriculture. Dietary guidelines for Americans, 2005. US Department of HHS Web site. http://www.health.gov/DietaryGuidelines/dga2005/document/default.htm. Published January 2005. Accessed September 25, 2009.
- National Heart, Lung, and Blood Institute. The practical guide: identification, evaluation, and treatment of overweight and obesity in adults. National Institutes of Health Web site. http://www.nhlbi.nih.gov/guidelines/obesity/prctgd_c.pdf. Updated: October 2000. Accessed September 28, 2009.
- Look AHEAD Research Group. Reduction in weight and cardiovascular disease risk factors in individuals with type 2 diabetes: one-year results of the Look AHEAD trial. Diabetes Care 2007; 30:1374–1383.
- Williamson DA, Rejeski J, Lang W, Van Dorsten B, Fabricatore AN, Toledo K; for the Look AHEAD Research Group. Impact of a weight management program on health-related quality of life in overweight adults with type 2 diabetes. Arch Intern Med 2009; 169:163–171.
- Klein S, Sheard NF, Pi-Sunyer X, et al; for the American Diabetes Association; North American Association for the Study of Obesity; American Society for Clinical Nutrition. Weight management through lifestyle modification for the prevention and management of type 2 diabetes: rationale and strategies: a statement of the American Diabetes Association, the North American Association for the Study of Obesity, and the American Society for Clinical Nutrition. Diabetes Care 2004; 27:2067–2073.
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 2001; 285:2486–2497.
- Buse JB, Ginsberg HN, Bakris GL, et al. Primary prevention of cardiovascular diseases in people with diabetes mellitus: a scientific statement from the American Heart Association and the American Diabetes Association. Circulation 2007; 115:114–126.
- Amori RE, Lau J, Pittas AG. Efficacy and safety of incretin therapy in type 2 diabetes: systematic review and meta-analysis. JAMA 2007; 298:194–206.
- Nielsen LL, Young AA, Parkes DG. Pharmacology of exenatide (synthetic exendin-4): a potential therapeutic for improved glycemic control of type 2 diabetes. Regul Pept 2004; 117:77–88.
- Young AA, Gedulin BR, Bhavsar S, et al. Glucose-lowering and insulin-sensitizing actions of exendin-4: studies in obese diabetic (ob/ob, db/db) mice, diabetic fatty Zucker rats, and diabetic rhesus monkeys (Macaca mulatta). Diabetes 1999; 48:1026–1034.
- Kim D, MacConell L, Zhuang D, et al. Effects of once-weekly dosing of a long-acting release formulation of exenatide on glucose control and body weight in subjects with type 2 diabetes. Diabetes Care 2007; 30:1487–1493.
- Buse JB, Henry RR, Han J, et al. Effects of exenatide (exendin-4) on glycemic control over 30 weeks in sulfonylurea-treated patients with type 2 diabetes. Diabetes Care 2004; 27:2628–2635.
- DeFronzo RA, Ratner RE, Han J, Kim DD, Fineman MS, Baron AD. Effects of exenatide (exendin-4) on glycemic control and weight over 30 weeks in metformin-treated patients with type 2 diabetes. Diabetes Care 2005; 28:1092–1100.
- Kendall DM, Riddle MC, Rosenstock J, et al. Effects of exenatide (exendin-4) on glycemic control over 30 weeks in patients with type 2 diabetes treated with metformin and a sulfonylurea. Diabetes Care 2005; 28:1083–1091.
- Zinman B, Hoogwerf BJ, Durán García S, et al. The effect of adding exenatide to a thiazolidinedione in suboptimally controlled type 2 diabetes: a randomized trial. Ann Intern Med 2007; 146:477–485.
- Klonoff DC, Buse JB, Nielsen LL, et al. Exenatide effects on diabetes, obesity, cardiovascular risk factors and hepatic biomarkers in patients with type 2 diabetes treated for at least 3 years. Curr Med Res Opin 2008; 24:275–286.
- Heine RJ, Van Gaal LF, Johns D, Mihm MJ, Widel MH, Brodows RG; for the GWAA Study Group. Exenatide versus insulin glargine in patients with suboptimally controlled type 2 diabetes: a randomized trial. Ann Intern Med 2005; 143:559–569.
- Barnett AH, Burger J, Johns D, et al. Tolerability and efficacy of exenatide and titrated insulin glargine in adult patients with type 2 diabetes previously uncontrolled with metformin or a sulfonylurea: a multinational, randomized, open-label, two-period, crossover noninferiority trial. Clin Ther 2007; 29:2333–2348.
- Nauck MA, Duran S, Kim D, et al. A comparison of twice-daily exenatide and biphasic insulin aspart in patients with type 2 diabetes who were suboptimally controlled with sulfonylurea and metformin: a non-inferiority study. Diabetologia 2007; 50:259–267.
- Glass LC, Qu Y, Lenox S, et al. Effects of exenatide versus insulin analogues on weight change in subjects with type 2 diabetes: a pooled post-hoc analysis. Curr Med Res Opin 2008; 24:639–644.
- Drucker DJ, Buse JB, Taylor K, et al; for the DURATION-1 Study Group. Exenatide once weekly versus twice daily for the treatment of type 2 diabetes: a randomised, open-label, non-inferiority study. Lancet 2008; 372:1240–1250.
- Garber A, Henry R, Ratner R, et al; for the LEAD-3 (Mono) Study Group. Liraglutide versus glimepiride monotherapy for type 2 diabetes (LEAD-3 Mono): a randomised, 52-week, phase III, double-blind, parallel-treatment trial. Lancet 2009; 373:473–481.
- Nauck M, Frid A, Hermansen K, et al; for the LEAD-2 Study Group. Efficacy and safety comparison of liraglutide, glimepiride, and placebo, all in combination with metformin, in type 2 diabetes: the LEAD (Liraglutide Effect and Action in Diabetes)-2 study. Diabetes Care 2009; 32:84–90.
- Ahrén B, Landin-Olsson M, Jansson PA, Svensson M, Holmes D, Schweizer A. Inhibition of dipeptidyl peptidase-4 reduces glycemia, sustains insulin levels, and reduces glucagon levels in type 2 diabetes. J Clin Endocrinol Metab 2004; 89:2078–2084.
- Herman GA, Stevens C, Van Dyck K, et al. Pharmacokinetics and pharmacodynamics of sitagliptin, an inhibitor of dipeptidyl peptidase IV, in healthy subjects: results from two randomized, double-blind, placebo-controlled studies with single oral doses. Clin Pharmacol Ther 2005; 78:675–688.
- Bohannon N. Overview of the gliptin class (dipeptidyl peptidase-4 inhibitors) in clinical practice. Postgrad Med 2009; 121:40–45.
- US Department of Health and Human Services. FDA approves new drug treatment for type 2 diabetes. US Food and Drug Administration Web site. http://www.fda.gov/newsevents/newsroom/pressannouncements/ucm174780.htm. Published July 31, 2009. Accessed September 18, 2009.
- Raz I, Hanefeld M, Xu L, Caria C, Williams-Herman D, Khatami H; for the Sitagliptin Study 023 Group. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor sitagliptin as monotherapy in patients with type 2 diabetes mellitus. Diabetologia 2006; 49:2564–2571.
- Zerilli T, Pyon EY. Sitagliptin phosphate: a DPP-4 inhibitor for the treatment of type 2 diabetes mellitus. Clin Ther 2007; 29:2614–2634.
- Scott R, Loeys T, Davies MJ, Engel SS; for the Sitagliptin Study 801 Group. Efficacy and safety of sitagliptin when added to ongoing metformin therapy in patients with type 2 diabetes. Diabetes Obes Metab 2008; 10:959–969.
- Williams-Herman D, Johnson J, Teng R, et al. Efficacy and safety of initial combination therapy with sitagliptin and metformin in patients with type 2 diabetes: a 54-week study. Curr Med Res Opin 2009; 25:569–583.
- DeFronzo RA, Okerson T, Viswanathan P, Guan X, Holcombe JH, MacConell L. Effects of exenatide versus sitagliptin on postprandial glucose, insulin and glucagon secretion, gastric emptying, and caloric intake: a randomized, cross-over study. Curr Med Res Opin 2008; 24:2943–2952.
- AACE Diabetes Mellitus Clinical Practice Guidelines Task Force. American Association of Clinical Endocrinologists medical guidelines for clinical practice for the management of diabetes mellitus. Endocr Pract 2007; 13(suppl 1):S4–S68.
- American Diabetes Association. Standards of medical care in diabetes—2009. Diabetes Care 2009; 32(suppl 1):S13–S61.
- Nathan DM, Buse JB, Davidson MB, et al. Medical management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 2009; 32:193–203.
- National Institute of Diabetes and Digestive and Kidney Diseases. National Diabetes Statistics, 2007 fact sheet. National Institutes of Health Web site. http://www.diabetes.niddk.nih.gov/dm/pubs/statistics/index.htm. Published 2008. Accessed September 16, 2009.
- Koro CE, Bowlin SJ, Bourgeois N, Fedder DO. Glycemic control from 1988 to 2000 among US adults diagnosed with type 2 diabetes: a preliminary report. Diabetes Care 2004; 27:17–20.
- Ong KL, Cheung BM, Wong LY, Wat NM, Tan KC, Lam KS. Prevalence, treatment, and control of diagnosed diabetes in the US National Health and Nutrition Examination Survey 1999–2004. Ann Epidemiol 2008; 18:222–229.
- Koopman RJ, Mainous AG III, Diaz VA, Geesey ME. Changes in age at diagnosis of type 2 diabetes mellitus in the United States, 1988 to 2000. Ann Fam Med 2005; 3:60–63.
- Alexander GC, Sehgal NL, Moloney RM, Stafford RS. National trends in treatment of type 2 diabetes mellitus, 1994–2007. Arch Intern Med 2008; 168:2088–2094.
- The Action to Control Cardiovascular Risk in Diabetes Study Group. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008; 358:2545–2559.
- The ADVANCE Collaborative Group. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008; 358:2560–2572.
- Duckworth W, Abraira C, Moritz T, et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med 2009; 360:129–139.
- Jenssen TG, Tonstad S, Claudi T, Midthjell K, Cooper J. The gap between guidelines and practice in the treatment of type 2 diabetes: a nationwide survey in Norway. Diabetes Res Clin Pract 2008; 80:314–320.
- Gaede P, Lund-Andersen H, Parving HH, Pedersen O. Effect of a multifactorial intervention on mortality in type 2 diabetes. N Engl J Med 2008; 358:580–591.
- Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care 2004; 27:1047–1053.
- Rosenstock J. Management of type 2 diabetes mellitus in the elderly: special considerations. Drugs Aging 2001; 18:31–44.
- Brandle M, Zhou H, Smith BR, et al. The direct medical cost of type 2 diabetes. Diabetes Care 2003; 26:2300–2304.
- National Center for Health Statistics. Prevalence of overweight and obesity among adults: United States 2003–2004. Centers for Disease Contral and Prevention Web site. http://www.cdc.gov/nchs/products/pubs/pubd/hestats/overweight/overwght_adult_03.htm. Published: April 2006. Accessed September 23, 2009.
- Van Gaal LF, Gutkin SW, Nauck MA. Exploiting the antidiabetic properties of incretins to treat type 2 diabetes mellitus: glucagon-like peptide 1 receptor agonists or insulin for patients with inadequate glycemic control. Eur J Endocrinol 2008; 158:773–784.
- Anderson JW, Kendall CW, Jenkins DJ. Importance of weight management in type 2 diabetes: review with meta-analysis of clinical studies. J Am Coll Nutr 2003; 22:331–339.
- Fox CS, Pencina MJ, Wilson PW, Paynter NP, Vasan RS, D’Agostino RB Sr. Lifetime risk of cardiovascular disease among individuals with and without diabetes stratified by obesity status in the Framingham heart study. Diabetes Care 2008; 31:1582–1584.
- DeFronzo RA. From the triumvirate to the ominous octet: a new paradigm for the treatment of type 2 diabetes mellitus. Diabetes 2009; 58:773–795.
- Stonehouse A, Okerson T, Kendall D, Maggs D. Emerging incretin based therapies for type 2 diabetes: incretin mimetics and DPP-4 inhibitors. Curr Diabetes Rev 2008; 4:101–109.
- Cefalu WT. Pharmacotherapy for the treatment of patients with type 2 diabetes mellitus: rationale and specific agents. Clin Pharmacol Ther 2007; 81:636–649.
- Henry RR. Evolving concepts of type 2 diabetes management with oral medications: new approaches to an old disease. Curr Med Res Opin 2008; 24:2189–2202.
- American Diabetes Association. Nutrition recommendations and interventions for diabetes: a position statement of the American Diabetes Association. Diabetes Care 2008; 31(suppl 1):S61−S78.
- US Department of Health and Human Services (HHS) and US Department of Agriculture. Dietary guidelines for Americans, 2005. US Department of HHS Web site. http://www.health.gov/DietaryGuidelines/dga2005/document/default.htm. Published January 2005. Accessed September 25, 2009.
- National Heart, Lung, and Blood Institute. The practical guide: identification, evaluation, and treatment of overweight and obesity in adults. National Institutes of Health Web site. http://www.nhlbi.nih.gov/guidelines/obesity/prctgd_c.pdf. Updated: October 2000. Accessed September 28, 2009.
- Look AHEAD Research Group. Reduction in weight and cardiovascular disease risk factors in individuals with type 2 diabetes: one-year results of the Look AHEAD trial. Diabetes Care 2007; 30:1374–1383.
- Williamson DA, Rejeski J, Lang W, Van Dorsten B, Fabricatore AN, Toledo K; for the Look AHEAD Research Group. Impact of a weight management program on health-related quality of life in overweight adults with type 2 diabetes. Arch Intern Med 2009; 169:163–171.
- Klein S, Sheard NF, Pi-Sunyer X, et al; for the American Diabetes Association; North American Association for the Study of Obesity; American Society for Clinical Nutrition. Weight management through lifestyle modification for the prevention and management of type 2 diabetes: rationale and strategies: a statement of the American Diabetes Association, the North American Association for the Study of Obesity, and the American Society for Clinical Nutrition. Diabetes Care 2004; 27:2067–2073.
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 2001; 285:2486–2497.
- Buse JB, Ginsberg HN, Bakris GL, et al. Primary prevention of cardiovascular diseases in people with diabetes mellitus: a scientific statement from the American Heart Association and the American Diabetes Association. Circulation 2007; 115:114–126.
- Amori RE, Lau J, Pittas AG. Efficacy and safety of incretin therapy in type 2 diabetes: systematic review and meta-analysis. JAMA 2007; 298:194–206.
- Nielsen LL, Young AA, Parkes DG. Pharmacology of exenatide (synthetic exendin-4): a potential therapeutic for improved glycemic control of type 2 diabetes. Regul Pept 2004; 117:77–88.
- Young AA, Gedulin BR, Bhavsar S, et al. Glucose-lowering and insulin-sensitizing actions of exendin-4: studies in obese diabetic (ob/ob, db/db) mice, diabetic fatty Zucker rats, and diabetic rhesus monkeys (Macaca mulatta). Diabetes 1999; 48:1026–1034.
- Kim D, MacConell L, Zhuang D, et al. Effects of once-weekly dosing of a long-acting release formulation of exenatide on glucose control and body weight in subjects with type 2 diabetes. Diabetes Care 2007; 30:1487–1493.
- Buse JB, Henry RR, Han J, et al. Effects of exenatide (exendin-4) on glycemic control over 30 weeks in sulfonylurea-treated patients with type 2 diabetes. Diabetes Care 2004; 27:2628–2635.
- DeFronzo RA, Ratner RE, Han J, Kim DD, Fineman MS, Baron AD. Effects of exenatide (exendin-4) on glycemic control and weight over 30 weeks in metformin-treated patients with type 2 diabetes. Diabetes Care 2005; 28:1092–1100.
- Kendall DM, Riddle MC, Rosenstock J, et al. Effects of exenatide (exendin-4) on glycemic control over 30 weeks in patients with type 2 diabetes treated with metformin and a sulfonylurea. Diabetes Care 2005; 28:1083–1091.
- Zinman B, Hoogwerf BJ, Durán García S, et al. The effect of adding exenatide to a thiazolidinedione in suboptimally controlled type 2 diabetes: a randomized trial. Ann Intern Med 2007; 146:477–485.
- Klonoff DC, Buse JB, Nielsen LL, et al. Exenatide effects on diabetes, obesity, cardiovascular risk factors and hepatic biomarkers in patients with type 2 diabetes treated for at least 3 years. Curr Med Res Opin 2008; 24:275–286.
- Heine RJ, Van Gaal LF, Johns D, Mihm MJ, Widel MH, Brodows RG; for the GWAA Study Group. Exenatide versus insulin glargine in patients with suboptimally controlled type 2 diabetes: a randomized trial. Ann Intern Med 2005; 143:559–569.
- Barnett AH, Burger J, Johns D, et al. Tolerability and efficacy of exenatide and titrated insulin glargine in adult patients with type 2 diabetes previously uncontrolled with metformin or a sulfonylurea: a multinational, randomized, open-label, two-period, crossover noninferiority trial. Clin Ther 2007; 29:2333–2348.
- Nauck MA, Duran S, Kim D, et al. A comparison of twice-daily exenatide and biphasic insulin aspart in patients with type 2 diabetes who were suboptimally controlled with sulfonylurea and metformin: a non-inferiority study. Diabetologia 2007; 50:259–267.
- Glass LC, Qu Y, Lenox S, et al. Effects of exenatide versus insulin analogues on weight change in subjects with type 2 diabetes: a pooled post-hoc analysis. Curr Med Res Opin 2008; 24:639–644.
- Drucker DJ, Buse JB, Taylor K, et al; for the DURATION-1 Study Group. Exenatide once weekly versus twice daily for the treatment of type 2 diabetes: a randomised, open-label, non-inferiority study. Lancet 2008; 372:1240–1250.
- Garber A, Henry R, Ratner R, et al; for the LEAD-3 (Mono) Study Group. Liraglutide versus glimepiride monotherapy for type 2 diabetes (LEAD-3 Mono): a randomised, 52-week, phase III, double-blind, parallel-treatment trial. Lancet 2009; 373:473–481.
- Nauck M, Frid A, Hermansen K, et al; for the LEAD-2 Study Group. Efficacy and safety comparison of liraglutide, glimepiride, and placebo, all in combination with metformin, in type 2 diabetes: the LEAD (Liraglutide Effect and Action in Diabetes)-2 study. Diabetes Care 2009; 32:84–90.
- Ahrén B, Landin-Olsson M, Jansson PA, Svensson M, Holmes D, Schweizer A. Inhibition of dipeptidyl peptidase-4 reduces glycemia, sustains insulin levels, and reduces glucagon levels in type 2 diabetes. J Clin Endocrinol Metab 2004; 89:2078–2084.
- Herman GA, Stevens C, Van Dyck K, et al. Pharmacokinetics and pharmacodynamics of sitagliptin, an inhibitor of dipeptidyl peptidase IV, in healthy subjects: results from two randomized, double-blind, placebo-controlled studies with single oral doses. Clin Pharmacol Ther 2005; 78:675–688.
- Bohannon N. Overview of the gliptin class (dipeptidyl peptidase-4 inhibitors) in clinical practice. Postgrad Med 2009; 121:40–45.
- US Department of Health and Human Services. FDA approves new drug treatment for type 2 diabetes. US Food and Drug Administration Web site. http://www.fda.gov/newsevents/newsroom/pressannouncements/ucm174780.htm. Published July 31, 2009. Accessed September 18, 2009.
- Raz I, Hanefeld M, Xu L, Caria C, Williams-Herman D, Khatami H; for the Sitagliptin Study 023 Group. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor sitagliptin as monotherapy in patients with type 2 diabetes mellitus. Diabetologia 2006; 49:2564–2571.
- Zerilli T, Pyon EY. Sitagliptin phosphate: a DPP-4 inhibitor for the treatment of type 2 diabetes mellitus. Clin Ther 2007; 29:2614–2634.
- Scott R, Loeys T, Davies MJ, Engel SS; for the Sitagliptin Study 801 Group. Efficacy and safety of sitagliptin when added to ongoing metformin therapy in patients with type 2 diabetes. Diabetes Obes Metab 2008; 10:959–969.
- Williams-Herman D, Johnson J, Teng R, et al. Efficacy and safety of initial combination therapy with sitagliptin and metformin in patients with type 2 diabetes: a 54-week study. Curr Med Res Opin 2009; 25:569–583.
- DeFronzo RA, Okerson T, Viswanathan P, Guan X, Holcombe JH, MacConell L. Effects of exenatide versus sitagliptin on postprandial glucose, insulin and glucagon secretion, gastric emptying, and caloric intake: a randomized, cross-over study. Curr Med Res Opin 2008; 24:2943–2952.
KEY POINTS
- The NHANES 1999–2004 data showed that only 13.2% of patients with diagnosed diabetes achieved concurrent weight, blood pressure, and lipid level goals.
- Among patients with T2DM, lifestyle intervention (control of weight, blood pressure, lipid levels) should be reinforced at every physician visit; glycosylated hemoglobin (HbA1c) should be monitored every 3 months until it is less than 7.0%, and then rechecked every 6 months.
- The effects of GLP-1 agonists on HbA1c are comparable to insulin analogues, but GLP-1 agonists are associated with weight reduction, while insulin is associated with weight gain.
- DPP-4 inhibitors have been associated with significant reductions in HbA1c when used alone or with metformin or pioglitazone.
Bariatric surgery: Part of the answer to the obesity epidemic
Controlling hypoglycemia in type 2 diabetes: Which agent for which patient?
- Advise patients to monitor blood glucose levels frequently and learn to correlate drops in glucose to symptoms, which vary among patients.
- Ask patients at each visit about awareness of hypoglycemic episodes, their severity and timing, and how events relate to dosing, meals, and activities.
- When using oral agents, consider insulin sensitizers or newer sulfonylureas or meglitinides to reduce risk of hypoglycemia.
- If adding basal insulin to an oral regimen, the analog glargine has proven superior to NPH insulin in avoiding hypoglycemia.
Signs and symptoms of hypoglycemia vary considerably among patients with type 2 diabetes, making the condition easy to miss. Moreover, the most common symptoms are not necessarily the first symptoms.
If hypoglycemia occurs repeatedly, it can start a vicious cycle of physiologic reactions that mask or diminish the symptoms that warn patients of an impending episode. This may lead to hypoglycemia unawareness and hypoglycemic episodes of increasing severity. Fear of hypoglycemia, particularly of nocturnal events, may discourage patients from more intensive glycemic control, particularly using insulin. Such fear may even lead them to reduce their antidiabetic medication dosage, resulting in poor glycemic control.1
Breaking this cycle and restoring normal physiologic responses is one focus of this article, as is teaching patients how to monitor their blood glucose levels and how to correlate low blood glucose with the signs and symptoms of hypoglycemia.
Other therapies and strategies that we discuss in this article:
- Newer insulin analogs and the associated risk of hypoglycemia with each
- Appropriate combination of insulin with oral antidiabetic medications
- The long-acting analog insulin glargine used as basal insulin to lower the incidence of hypoglycemia, including nocturnal and severe hypoglycemia
- Rapid-acting insulin analogs (aspart, glulisine, and lispro) used in basalprandial insulin regimens.
First symptoms vary among individuals
Symptoms of hypoglycemia result primarily from a lowered glucose level in the brain and its effects on the central and autonomic nervous systems (FIGURE 1). A decrease in glucose below physiologic levels has acute consequences for brain function because the brain has an immediate requirement for glucose and little capacity for storage.
Two types of symptoms
Neuroglycopenia and the inhibition of neuronal metabolism causes sensations of warmth, weakness, fatigue, difficulty concentrating, confusion, behavioral changes, and in the most severe cases, a loss of consciousness, seizures, brain damage, and even death.2-4
Neurogenic symptoms are mediated by the hormones and neurotransmitters secreted in response to low brain glucose levels (FIGURE 1). The gluconeogenic actions of the autonomic nervous system produce the classic warning symptoms—tremulousness, pounding heart, anxiety, sweating, hunger, and tingling sensations—that usually precede the symptoms of hypoglycemia.2-4 This is particularly so in iatrogenic hypoglycemia.
These direct symptoms of neuroglycopenia are the ones patients typically identify with hypoglycemia. The most common symptoms of hypoglycemia are therefore not necessarily the first symptoms of hypoglycemia (TABLE 1).5 For example, though most patients experience sweating as a symptom of hypoglycemia, the first symptom might be trembling or anxiety, depending on the individual.5
TABLE 1
Signs and symptoms most commonly associated with hypoglycemia are not always the first to appear
| SYMPTOM | FREQUENCY (%) | FIRST SYMPTOM (%) |
|---|---|---|
| Sweating | 78 | 25 |
| Trembling | 62 | 6 |
| Inability to concentrate | 49 | 6 |
| Confusion | 40 | 3 |
| Weakness | 36 | 4 |
| Dry mouth | 35 | 0 |
| Blurred vision | 34 | 3 |
| Hunger | 33 | 3 |
| Anxiety | 26 | 1 |
| Headache | 21 | 0 |
| Difficulty walking | 21 | 3 |
| Pounding heart | 20 | 0 |
| Tingling around mouth | 20 | 6 |
| Difficulty speaking | 17 | 0 |
| Drowsiness | 15 | 0 |
| Odd behavior | 13 | 1 |
| Nausea | 13 | 0 |
| Adapted from Hepburn DA, MacLeod KM, Pell AC, et al. Diabet Med 1993;10:231–237.5 | ||
Factors influencing frequency and severity of hypoglycemia
Aggressive diabetes management commonly causes mild-to-moderate hypoglycemia, defined as a blood glucose value <60 mg/dL, that can be managed by the patient without assistance.
Severe hypoglycemia—a blood glucose value <50 mg/dL—is relatively uncommon in type 2 diabetes and requires the assistance of another person to manage, since neurological impairment may render patients unable to treat themselves.2,6 Severe hypoglycemia, whether in patients with type 1 or type 2 diabetes, can have debilitating consequences, including seizures or coma or even death.7
Long-standing type 2 disease. Hypoglycemia is more common in patients with type 1 diabetes than in those with type 2, but it can occur in type 2 diabetes patients who require insulin or are treated intensively with combinations of oral agents.6 As type 2 diabetes progresses,8 the incidence of hypoglycemic events increases, as endogenously produced insulin declines and is replaced by exogenous insulin.5,9 In fact, the prevalence of severe episodes (eg, requiring assistance of another person to administer glucose or glucagon) in patients with type 2 diabetes was comparable to that exhibited among patients with type 1 diabetes if they had been on insulin therapy for the same length of time.5,10
Nocturnal hypoglycemia. This event poses a special concern because the warning signs of hypoglycemia may be blunted during sleep. It has been reported that as many as 29% to 56% of all adult patients treated with insulin have an overnight glucose profile that indicates hypoglycemia occurs at night.11-13 However, it is important to note that the extent of the problem of nocturnal hypoglycemia is difficult to assess since overnight monitoring of glucose levels is required.
Additional insights from the UKPDS
Hypoglycemia in type 2 diabetes has not received rigorous attention in clinical trials. However, the United Kingdom Prospective Diabetes Study (UKPDS) was a large longitudinal trial in type 2 diabetes that included hypoglycemia as an outcome measure and thus provides some helpful information.
Events with insulin>sulfonylureas>diet. The 6-year follow-up revealed that the cumulative proportion of patients reporting 1 or more hypoglycemic events (of any type) was 76% for those using insulin, 45% among those taking sulfonylureas, and 3% for those on diet alone. Expressed as events per patient year, this was 37%, 17%, and 0.9%, respectively. When only “major” events (those requiring third-party assistance or hospital admission) were considered, the proportion of patients per year reporting 1 or more such events, was 2.3% for insulin, 0.7% for sulfonylureas, and 0.03% for diet alone. The cumulative proportion over 6 years was 3.3% of participants using sulfonylureas, 11.2% of those using insulin, and 0.15% of those on diet therapy.8
Metformin increases risk. The cumulative proportion of obese patients reporting any hypoglycemic event was 17.6% for those taking metformin vs 2.8% for those on diet. Severe hypoglycemia (as defined earlier) occurred in 2.4% of participants using metformin compared with 0.4% of those on diet therapy.8
Findings from other studies
Interestingly, in a recent systematic review of randomized controlled trials comparing insulin monotherapy with insulin plus oral antidiabetic agents, 13 of 14 studies reporting hypoglycemia demonstrated no difference in events.14
The occurrence of hypoglycemia among patients on metformin monotherapy in the UKPDS study is notable since, theoretically, hypoglycemia should not occur with agents whose mechanisms of action do not increase insulin secretion (biguanides, thiazolidinediones [TZDs], or α-glucosidase inhibitors),1 Since newer classes such as TZDs, α-glucosidase inhibitors, and meglitinides were not available when UKPDS was initiated, the trial does not provide data on these classes.
In a small comparative study of insulin combined with either metformin or a TZD, it appeared that metformin combination was associated with fewer occurrences of hypoglycemia; however, the small patient sample limits generalizability of the finding.15
How hypoglycemia occurs
Normally, as blood glucose levels (red) rise, insulin secretion increases, circulating insulin levels (blue) rise, and hepatic glucose production is inhibited. As glucose is disposed and circulating levels decrease due to insulin action, insulin levels then drop and hepatic glucose production begins again.
Hypoglycemia in diabetes can result from an excess of endogenous or exogenous insulin (iatrogenic hypoglycemia). In healthy patients, high insulin and falling glucose levels suppress insulin production and stimulate a hormone-mediated burst of glucose production. In patients with diabetes, the loss of physiologic control of insulin secretion coupled with exogenous administration of insulin or insulin secretagogues can interfere with the normal physiologic response to low blood glucose levels, resulting in hypoglycemia.1
With intense insulin regimens, the incidence of hypoglycemia can be as high as 30%, in contrast to 12% for patients treated with diet alone and 16% for those taking oral agents.6
With secretagogues, it has been suggested that the incidence of hypoglycemia is higher with the older, longer acting sulfonylurea agents.1,6,16-19 Although populationbased data on hypoglycemic rates associated with combination therapy with oral antidiabetic agents are not available, numerous clinical studies have reported rates of 10% to 20% for any hypoglycemic event.20-23
Heightening patient awareness, and yours
Because the signs of hypoglycemia vary considerably among individuals, they can easily be missed.3 In addition, repeated episodes of hypoglycemia can alter the normal regulatory responses and diminish the most important signs of a drop in glucose levels.1,2,24,25 The loss of the physiologic warning signs is thought to stem from dampening and eventual loss of the neuroadrenal response to low glucose levels in the brain (FIGURE 1). A vicious cycle is set up, whereby reduction in the neurogenic response attenuates hypoglycemic symptoms, causing more episodes to occur and become more severe as they are repeated. This cycle can be broken, and the normal physiologic response restored, if hypoglycemic events can be avoided for just a few weeks.2,26
Key points for patients. The main strategy for managing hypoglycemia is educating patients about the early symptoms of hypoglycemia and how to self-treat when they occur. Reinforce the need to time meals consistently and to limit the amount of carbohydrate ingested.
Advise patients to monitor blood glucose levels frequently, and to learn to relate a drop in glucose levels to hypoglycemic symptoms.2
Counsel patients to eat a snack or, preferably, drink fruit juice to counteract hypoglycemia. Patients may also carry glucose tablets, which are convenient and less tempting than candy.27
Glucagon is indicated for severe cases.
Whenever possible, a patient’s family members (particularly in the case of children) should be educated too.
Ask regularly about episodes. Finally, act to identify problems by querying patients and family members at every visit about hypoglycemia episodes, probing for information about awareness, severity and timing of the episodes, and how these events relate to dosing, meals, and activities.27 If hypoglycemia recurs, analyze the dosing regimen and consider flexible insulin dosing.1
Anti-hypoglycemia strategies for each new phase of therapy
As the course of diabetes therapy moves, typically, from oral medications to insulin to combination regimens, drug selections can be made in part to reduce the risk of hypoglycemia.
Oral agents: Insulin sensitizers, newer agents generally better
As noted earlier, among oral agents, insulin sensitizers are generally thought to have lower rates of hypoglycemia.
Newer sulfonylureas such as glimepiride and the rapid-acting meglitinides may also cause fewer hypoglycemic events.
Given the progressive decline of endogenous insulin secretion, combination therapy with secretagogues or insulin is eventually required for most patients.
Insulin analogs
A number of rapid-, short-, intermediateand long-acting insulin analogs have been introduced, and many of them make it possible to mimic different phases of physiologic insulin secretion (FIGURE 2). One of the newer analogs less likely to cause hypoglycemia is glargine, a long-acting insulin with a steady, relatively consistent action profile over a 24-hour period, closely mimicking normal basal pancreatic insulin secretion.28
FIGURE 2
Plasma insulin levels with newer analogs
N=20 type 1 diabetic patients; mean±SE
CSII, continuous subcutaneous insulin infusion; NPH, neutral protamine Hagedorn.
Copyright © 2000 American Diabetes Association. From Diabetes 2000; 49:2142–2148.28 Reprinted with permission from American Diabetes Association.
TABLE 2
Strategies for avoiding and addressing hypoglycemia
|
Insulin mixtures helpful when meal times guaranteed
Other insulins include mixtures of regular insulin and long-acting insulin available in split mixed or premixed formulations. These mixtures are intended to cover insulin peaks at mealtimes with twice-daily administration.
Mixed insulin formulations are often perceived as relatively convenient and simple to use, but they require meals to be taken within set time frames, without a great deal of flexibility. Since the ratios of the insulin components are fixed, and designed to work with meals consumed on a fixed schedule, hypoglycemia can occur if patients miss a meal. In addition, the time-activity profile of the insulin may not match the postprandial glucose peak even if the meal is consumed, and will increase the chance of postprandial hypoglycemia.
Basal insulin plus oral regimens
For patients with type 2 diabetes, adding basal insulin to oral regimens can significantly improve glycemic control. Ideally, basal insulin therapy provides a sustained and relatively constant concentration of insulin throughout the day. In the past, neutral protamine Hagedorn (NPH) insulin, a longer-acting insulin, was used for basal insulin therapy, and regular insulin was used to cover prandial insulin needs. Ultralente, also used as a basal insulin, has a relatively unpredictable timeactivity profile.28
Insulin glargine superior to NPH. In a recent clinical trial, patients with type 2 diabetes whose glucose levels were inadequately controlled on oral antidiabetic medications were given bedtime insulin glargine or NPH insulin.29 The insulin doses were titrated using a simple algorithm targeting a fasting plasma glucose (FPG) of ≤100 mg/dL to reach recommended glycosylated hemoglobin (Hb A1c) levels. Though no significant difference in glycemic control was found between insulin glargine and NPH insulin, significantly fewer hypoglycemic episodes occurred with insulin glargine therapy. The 24-hour distribution per patient-year of hypoglycemia for glargine vs NPH is shown in FIGURE 3.
Specifically, nearly 25% more patients treated with insulin glargine than with NPH insulin reached target Hb A1c levels of ≤7.0% without nocturnal hypoglycemia. Moreover, the overall incidence of any hypoglycemic event (eg, plasma-referenced glucose ≤72 mg/dL) and severe hypoglycemia (eg, patient required assistance of another person, and had a glucose level <56 mg/dL or prompt recovery after glucose or glucagons) was lower with insulin glargine than with NPH insulin. Results from other studies and a recent metaanalysis have been similar.30-32
Thus, using insulin glargine as basal insulin allows patients to reach recommended targets with fewer episodes of hypoglycemia, and can help address patients’ fear that can be a barrier to initiating insulin therapy in type 2 diabetes. Two recent studies have reported that dosing of insulin glargine can be flexible—morning or bedtime administration yields comparable low rates of hypoglycemia.30,31
FIGURE 3
Hourly hypoglycemia rate with glargine much less than with NPH
*P<.05 (between treatment).
Copyright © 2003 American Diabetes Association. From Diabetes Care 2003; 26:3080–3086.29 Reprinted with permission from American Diabetes Association.
Basal insulin plus prandial insulin
For patients who cannot otherwise reach Hb A1c goals, basal insulin therapy may be supplemented with prandial insulin. Newer, rapid-acting analogs used for the prandial component are insulin lispro, insulin glulisine, or insulin aspart. Although this approach is physiologically more rational than regimens using conventional insulins, data are limited for use in type 2 diabetes.
The incidence of nocturnal hypoglycemia was evaluated in a study of patients with type 1 diabetes and impaired hypoglycemic awareness who were treated with 1 of 2 regimens: insulin lispro in a basal-prandial combination with NPH insulin, or twice-daily, premixed NPH/regular insulin.33 Results showed that the incidence of nocturnal hypoglycemia was lower in patients receiving the insulin lispro regimen.
Another study, comparing insulin aspart and regular insulin as the prandial component in a basal-prandial regimen with NPH, showed that postprandial glucose control and Hb A1c levels were significantly better after 1 year of treatment in the insulin aspart group than in the group receiving regular insulin, without an increased risk for hypoglycemia.34 These results suggest that treatment with rapidacting insulin analogs could be helpful in avoiding hypoglycemia in patients with type 2 diabetes when a basal-prandial insulin regimen is indicated.
Avoiding hypoglycemia in the elderly
Elderly patients may be at increased risk for iatrogenic hypoglycemia. A populationbased study of patients presenting to an emergency room with severe hypoglycemic symptoms reported that rates of such events among elderly patients with type 2 diabetes and multiple comorbidities approached the rates among patients with type 1 diabetes.35
Creatinine clearance is often decreased in elderly patients, slowing elimination of oral agents and insulin and potentially resulting in sustained pharmacological action and creating a greater risk for hypoglycemia.
Furthermore, there is evidence that the neurogenic symptoms of hypoglycemia are reduced in elderly patients, diminishing awareness of hypoglycemia.36
In the demented elderly, malnutrition, weight loss, and anorexia may exacerbate the risk for hypoglycemia. For elderly patients with tertiary disease (eg, cerebrovascular accident, myocardial infarction, congestive heart failure, blindness, chronic renal failure), the risk for hypoglycemia and subsequent comorbidity may outweigh the benefits of strict glycemic control.3,37 Elderly patients may have comorbid conditions that increase risk of falls (eg, poor vision, neurologic conditions), and hypoglycemic episodes may further increase the risk of falls and lead to morbidity (eg, fragility fracture in patients with osteopenia or osteoporosis).
Because the elderly are at a greater risk for hypoglycemia, a switch to a less restrictive diet, such as a “no concentrated sweets” diet, is an option, with control of glucose levels through the administration of oral agents or insulin.36 This may also promote a better quality of life, considering that many of these patients already have secondary and tertiary complications of diabetes, prevention of which is not a realistic goal.
CORRESPONDING AUTHOR
William Cefalu, MD, Professor and Chief, Division of Nutrition and Chronic Diseases, Pennington Biomedical Research Center, Louisiana State University, 6400 Perkins Road, Baton Rouge, LA 70808. Email: cefaluwt@pbrc.edu
1. Cryer PE, Childs BP. Negotiating the barrier of hypoglycemia in diabetes. Diabetes Spectrum 2002;15:20-27.
2. Cryer PE, ed. Hypoglycemia: Pathophysiology, Diagnosis, and Treatment. New York, NY: Oxford University Press; 1997.
3. McAulay V, Deary IJ, Frier BM. Symptoms of hypoglycaemia in people with diabetes. Diabet Med 2001;18:690-705.
4. Towler DA, Havlin CE, Craft S, Cryer P. Mechanism of awareness of hypoglycemia. Perception of neurogenic (predominantly cholinergic) rather than neuroglycopenic symptoms. Diabetes 1993;42:1791-1798.
5. Hepburn DA, MacLeod KM, Pell AC, Scougal IJ, Frier BM. Frequency and symptoms of hypoglycaemia experienced by patients with type 2 diabetes treated with insulin. Diabet Med 1993;10:231-237.
6. Miller CD, Phillips LS, Ziemer DC, Gallina DL, Cook CB, El-Kebbi IM. Hypoglycemia in patients with type 2 diabetes mellitus. Arch Intern Med 2001;161:1653-1659.
7. The DCCT Research Group. Epidemiology of severe hypoglycemia in the diabetes control and complications trial. Am J Med 1991;90:450-459.
8. UK Prospective Diabetes Study Group. Perspectives in diabetes. UK Prospective Diabetes Study 16: overview of 6 years’ therapy of type II diabetes: a progressive disease. Diabetes 1995;44:1249-1258.
9. United Kingdom Prospective Diabetes Study Group. UKPDS 24: a 6-year, randomized, controlled trial comparing sulfonylurea, insulin, and metformin therapy in patients with newly diagnosed type 2 diabetes that could not be controlled with diet therapy. Ann Intern Med 1998;128:165-175.
10. MacLeod KM, Hepburn DA, Frier BM. Frequency and morbidity of severe hypoglycaemia in insulin-treated diabetic patients. Diabet Med 1993;10:238-245.
11. Vervoort G, Goldschmidt HM, van Doorn LG. Nocturnal blood glucose profiles in patients with type 1 diabetes mellitus on multiple (>or=4) daily insulin injection regimens. Diabet Med 1996;13:794-799.
12. Gale EA, Tattersall RB. Unrecognised nocturnal hypoglycaemia in insulin-treated diabetics. Lancet 1979;1:1049-1052.
13. Pramming S, Thorsteinsson B, Bendtson I, Ronn B, Binder C. Nocturnal hypoglycaemia in patients receiving conventional treatment with insulin. Br Med J (Clin Res Ed) 1985;291:376-379.
14. Goudswaard AN, Furlong NJ, Valk GD, Stolk RP, Rutten GEHM. Insulin monotherapy versus combinations of insulin with oral hypoglycaemic agents in patients with type 2 diabetes mellitus. Cochrane Database Syst Rev 2004;4:1-51.
15. Strowig SM, Avilés-Santa ML, Raskin P. Comparison of insulin monotherapy and combination therapy with insulin and metformin or insulin and troglitazone in type 2 diabetes. Diabetes Care 2002;25:1691-1698.
16. Campbell IW. Hypoglycaemia and type 2 diabetes: sulphonylureas. In: Frier BM, Fisher BM, eds. Hypoglycaemia and Diabetes: Clinical and Physiological Aspects. London, UK: Edward Arnold, 1993;387-392.
17. Shorr RI, Ray WA, Daugherty JR, Griffin MR. Individual sulfonylureas and serious hypoglycemia in older people. J Am Geriatr Soc 1996;44:751-755.
18. Holstein A, Egberts EH. Risk of hypoglycaemia with oral antidiabetic agents in patients with Type 2 diabetes. Exp Clin Endocrinol Diabetes 2003;111:405-414.
19. Holstein A, Plaschke A, Egberts E-H. Lower incidence of severe hypoglycaemia in patients with type 2 diabetes treated with glimepiride versus glibenclamide. Diabetes Metab Res Rev 2001;17:467-473.
20. Aljabri K, Kozak SE, Thompson DM. Addition of pioglitazone or bedtime insulin to maximal doses of sulfonylurea and metformin in type 2 diabetes patients with poor glucose control: a prospective, randomized trial. Am J Med 2004;116:230-235.
21. Wolffenbuttel BHR, Landgraf R. A 1-year multicenter randomized double-blind comparison of repaglinide and glyburide for the treatment of type 2 diabetes. Dutch and German Repaglinide Study Group. Diabetes Care 1999;22:463-467.
22. Raskin P, McGill J, Saad MF, et al. Combination therapy for type 2 diabetes: repaglinide plus rosiglitazone. Diabetes Med 2004;21:329-335.
23. Dailey GE, III, Noor MA, Park JS, Bruce S, Fiedorek FT. Glycemic control with glyburide/metformin tablets in combination with rosiglitazone in patients with type 2 diabetes: a randomized, double-blind trial. Am J Med 2004;116:223-229.
24. Korytkowski MT, Mokan M, Veneman TF, Mitrakou A, Cryer PE, Gerich JE. Reduced beta-adrenergic sensitivity in patients with type 1 diabetes and hypoglycemia unawareness. Diabetes Care 1998;21:1939-1943.
25. Fritsche A, Stumvoll M, Grub M, et al. Effect of hypoglycemia on beta-adrenergic sensitivity in normal and type 1 diabetic subjects. Diabetes Care 1998;21:1505-1510.
26. Fritsche A, Stefan N, Haring H, Gerich J, Stumvoll M. Avoidance of hypoglycemia restores hypoglycemia awareness by increasing beta-adrenergic sensitivity in type 1 diabetes. Ann Intern Med 2001;134:729-736.
27. Cryer PE, Davis SN, Shamoon H. Hypoglycemia in diabetes. Diabetes Care 2003;26:1902-1912.
28. Lepore M, Pampanelli S, Fanelli C, et al. Pharmacokinetics and pharmacodynamics of subcutaneous injection of long-acting human insulin analog glargine, NPH insulin, and ultralente human insulin and continuous subcutaneous infusion of insulin lispro. Diabetes 2000;49:2142-2148.
29. Riddle MC, Rosenstock J, Gerich J. The Treat-to-Target Trial: randomized addition of glargine or human NPH insulin to oral therapy of type 2 diabetic patients. Diabetes Care 2003;26:3080-3086.
30. Fritsche A, Schweitzer MA, Häring HU. Glimepiride combined with morning insulin glargine, bedtime neutral protamine Hagedorn insulin, or bedtime insulin glargine in patients with type 2 diabetes: a randomized, controlled trial. Ann Intern Med 2003;138:952-959.
31. Standl E, Maxeiner S, Raptis S, Karimi-Anderesi Z, Schweitzer MA. Good glycemic control with flexibility in timing of basal insulin supply: a 24-week comparison of insulin glargine given once daily in the morning or at bedtime in combination with morning glimepiride. Diabetes Care 2005;28:419-420.
32. Rosenstock J, Dailey G, Massi Benedetti M, Fritsche A, Lin Z, Salzman A. Reduced hypoglycemia risk with insulin glargine. A meta-analysis comparing insulin glargine with human NPH insulin in type 2 diabetes. Diabetes Care 2005;28:950-955.
33. Ferguson SC, Strachan MW, Janes JM, Frier BM. Severe hypoglycaemia in patients with type 1 diabetes and impaired awareness of hypoglycaemia: a comparative study of insulin lispro and regular human insulin. Diabetes Metab Res Rev 2001;17:285-291.
34. Raskin P, Guthrie RA, Leiter L, Riis A, Jovanovic L. Use of insulin aspart, a fast-acting insulin analog, as the mealtime insulin in the management of patients with type 1 diabetes. Diabetes Care 2000;23:583-558.
35. Holstein A, Plaschke A, Egberts EH. Clinical characterisation of severe hypoglycaemia-a prospective population-based study. Exp Clin Endocrinol Diabetes 2003;111:364-369.
36. Meneilly GS, Cheung E, Tuokko H. Counterregulatory hormone responses to hypoglycemia in the elderly patient with diabetes. Diabetes 1994;43:403-410.
37. American Diabetes Association. Standards of medical care for patients with diabetes mellitus. Diabetes Care 2003;26(suppl 1):S33-S50.
- Advise patients to monitor blood glucose levels frequently and learn to correlate drops in glucose to symptoms, which vary among patients.
- Ask patients at each visit about awareness of hypoglycemic episodes, their severity and timing, and how events relate to dosing, meals, and activities.
- When using oral agents, consider insulin sensitizers or newer sulfonylureas or meglitinides to reduce risk of hypoglycemia.
- If adding basal insulin to an oral regimen, the analog glargine has proven superior to NPH insulin in avoiding hypoglycemia.
Signs and symptoms of hypoglycemia vary considerably among patients with type 2 diabetes, making the condition easy to miss. Moreover, the most common symptoms are not necessarily the first symptoms.
If hypoglycemia occurs repeatedly, it can start a vicious cycle of physiologic reactions that mask or diminish the symptoms that warn patients of an impending episode. This may lead to hypoglycemia unawareness and hypoglycemic episodes of increasing severity. Fear of hypoglycemia, particularly of nocturnal events, may discourage patients from more intensive glycemic control, particularly using insulin. Such fear may even lead them to reduce their antidiabetic medication dosage, resulting in poor glycemic control.1
Breaking this cycle and restoring normal physiologic responses is one focus of this article, as is teaching patients how to monitor their blood glucose levels and how to correlate low blood glucose with the signs and symptoms of hypoglycemia.
Other therapies and strategies that we discuss in this article:
- Newer insulin analogs and the associated risk of hypoglycemia with each
- Appropriate combination of insulin with oral antidiabetic medications
- The long-acting analog insulin glargine used as basal insulin to lower the incidence of hypoglycemia, including nocturnal and severe hypoglycemia
- Rapid-acting insulin analogs (aspart, glulisine, and lispro) used in basalprandial insulin regimens.
First symptoms vary among individuals
Symptoms of hypoglycemia result primarily from a lowered glucose level in the brain and its effects on the central and autonomic nervous systems (FIGURE 1). A decrease in glucose below physiologic levels has acute consequences for brain function because the brain has an immediate requirement for glucose and little capacity for storage.
Two types of symptoms
Neuroglycopenia and the inhibition of neuronal metabolism causes sensations of warmth, weakness, fatigue, difficulty concentrating, confusion, behavioral changes, and in the most severe cases, a loss of consciousness, seizures, brain damage, and even death.2-4
Neurogenic symptoms are mediated by the hormones and neurotransmitters secreted in response to low brain glucose levels (FIGURE 1). The gluconeogenic actions of the autonomic nervous system produce the classic warning symptoms—tremulousness, pounding heart, anxiety, sweating, hunger, and tingling sensations—that usually precede the symptoms of hypoglycemia.2-4 This is particularly so in iatrogenic hypoglycemia.
These direct symptoms of neuroglycopenia are the ones patients typically identify with hypoglycemia. The most common symptoms of hypoglycemia are therefore not necessarily the first symptoms of hypoglycemia (TABLE 1).5 For example, though most patients experience sweating as a symptom of hypoglycemia, the first symptom might be trembling or anxiety, depending on the individual.5
TABLE 1
Signs and symptoms most commonly associated with hypoglycemia are not always the first to appear
| SYMPTOM | FREQUENCY (%) | FIRST SYMPTOM (%) |
|---|---|---|
| Sweating | 78 | 25 |
| Trembling | 62 | 6 |
| Inability to concentrate | 49 | 6 |
| Confusion | 40 | 3 |
| Weakness | 36 | 4 |
| Dry mouth | 35 | 0 |
| Blurred vision | 34 | 3 |
| Hunger | 33 | 3 |
| Anxiety | 26 | 1 |
| Headache | 21 | 0 |
| Difficulty walking | 21 | 3 |
| Pounding heart | 20 | 0 |
| Tingling around mouth | 20 | 6 |
| Difficulty speaking | 17 | 0 |
| Drowsiness | 15 | 0 |
| Odd behavior | 13 | 1 |
| Nausea | 13 | 0 |
| Adapted from Hepburn DA, MacLeod KM, Pell AC, et al. Diabet Med 1993;10:231–237.5 | ||
Factors influencing frequency and severity of hypoglycemia
Aggressive diabetes management commonly causes mild-to-moderate hypoglycemia, defined as a blood glucose value <60 mg/dL, that can be managed by the patient without assistance.
Severe hypoglycemia—a blood glucose value <50 mg/dL—is relatively uncommon in type 2 diabetes and requires the assistance of another person to manage, since neurological impairment may render patients unable to treat themselves.2,6 Severe hypoglycemia, whether in patients with type 1 or type 2 diabetes, can have debilitating consequences, including seizures or coma or even death.7
Long-standing type 2 disease. Hypoglycemia is more common in patients with type 1 diabetes than in those with type 2, but it can occur in type 2 diabetes patients who require insulin or are treated intensively with combinations of oral agents.6 As type 2 diabetes progresses,8 the incidence of hypoglycemic events increases, as endogenously produced insulin declines and is replaced by exogenous insulin.5,9 In fact, the prevalence of severe episodes (eg, requiring assistance of another person to administer glucose or glucagon) in patients with type 2 diabetes was comparable to that exhibited among patients with type 1 diabetes if they had been on insulin therapy for the same length of time.5,10
Nocturnal hypoglycemia. This event poses a special concern because the warning signs of hypoglycemia may be blunted during sleep. It has been reported that as many as 29% to 56% of all adult patients treated with insulin have an overnight glucose profile that indicates hypoglycemia occurs at night.11-13 However, it is important to note that the extent of the problem of nocturnal hypoglycemia is difficult to assess since overnight monitoring of glucose levels is required.
Additional insights from the UKPDS
Hypoglycemia in type 2 diabetes has not received rigorous attention in clinical trials. However, the United Kingdom Prospective Diabetes Study (UKPDS) was a large longitudinal trial in type 2 diabetes that included hypoglycemia as an outcome measure and thus provides some helpful information.
Events with insulin>sulfonylureas>diet. The 6-year follow-up revealed that the cumulative proportion of patients reporting 1 or more hypoglycemic events (of any type) was 76% for those using insulin, 45% among those taking sulfonylureas, and 3% for those on diet alone. Expressed as events per patient year, this was 37%, 17%, and 0.9%, respectively. When only “major” events (those requiring third-party assistance or hospital admission) were considered, the proportion of patients per year reporting 1 or more such events, was 2.3% for insulin, 0.7% for sulfonylureas, and 0.03% for diet alone. The cumulative proportion over 6 years was 3.3% of participants using sulfonylureas, 11.2% of those using insulin, and 0.15% of those on diet therapy.8
Metformin increases risk. The cumulative proportion of obese patients reporting any hypoglycemic event was 17.6% for those taking metformin vs 2.8% for those on diet. Severe hypoglycemia (as defined earlier) occurred in 2.4% of participants using metformin compared with 0.4% of those on diet therapy.8
Findings from other studies
Interestingly, in a recent systematic review of randomized controlled trials comparing insulin monotherapy with insulin plus oral antidiabetic agents, 13 of 14 studies reporting hypoglycemia demonstrated no difference in events.14
The occurrence of hypoglycemia among patients on metformin monotherapy in the UKPDS study is notable since, theoretically, hypoglycemia should not occur with agents whose mechanisms of action do not increase insulin secretion (biguanides, thiazolidinediones [TZDs], or α-glucosidase inhibitors),1 Since newer classes such as TZDs, α-glucosidase inhibitors, and meglitinides were not available when UKPDS was initiated, the trial does not provide data on these classes.
In a small comparative study of insulin combined with either metformin or a TZD, it appeared that metformin combination was associated with fewer occurrences of hypoglycemia; however, the small patient sample limits generalizability of the finding.15
How hypoglycemia occurs
Normally, as blood glucose levels (red) rise, insulin secretion increases, circulating insulin levels (blue) rise, and hepatic glucose production is inhibited. As glucose is disposed and circulating levels decrease due to insulin action, insulin levels then drop and hepatic glucose production begins again.
Hypoglycemia in diabetes can result from an excess of endogenous or exogenous insulin (iatrogenic hypoglycemia). In healthy patients, high insulin and falling glucose levels suppress insulin production and stimulate a hormone-mediated burst of glucose production. In patients with diabetes, the loss of physiologic control of insulin secretion coupled with exogenous administration of insulin or insulin secretagogues can interfere with the normal physiologic response to low blood glucose levels, resulting in hypoglycemia.1
With intense insulin regimens, the incidence of hypoglycemia can be as high as 30%, in contrast to 12% for patients treated with diet alone and 16% for those taking oral agents.6
With secretagogues, it has been suggested that the incidence of hypoglycemia is higher with the older, longer acting sulfonylurea agents.1,6,16-19 Although populationbased data on hypoglycemic rates associated with combination therapy with oral antidiabetic agents are not available, numerous clinical studies have reported rates of 10% to 20% for any hypoglycemic event.20-23
Heightening patient awareness, and yours
Because the signs of hypoglycemia vary considerably among individuals, they can easily be missed.3 In addition, repeated episodes of hypoglycemia can alter the normal regulatory responses and diminish the most important signs of a drop in glucose levels.1,2,24,25 The loss of the physiologic warning signs is thought to stem from dampening and eventual loss of the neuroadrenal response to low glucose levels in the brain (FIGURE 1). A vicious cycle is set up, whereby reduction in the neurogenic response attenuates hypoglycemic symptoms, causing more episodes to occur and become more severe as they are repeated. This cycle can be broken, and the normal physiologic response restored, if hypoglycemic events can be avoided for just a few weeks.2,26
Key points for patients. The main strategy for managing hypoglycemia is educating patients about the early symptoms of hypoglycemia and how to self-treat when they occur. Reinforce the need to time meals consistently and to limit the amount of carbohydrate ingested.
Advise patients to monitor blood glucose levels frequently, and to learn to relate a drop in glucose levels to hypoglycemic symptoms.2
Counsel patients to eat a snack or, preferably, drink fruit juice to counteract hypoglycemia. Patients may also carry glucose tablets, which are convenient and less tempting than candy.27
Glucagon is indicated for severe cases.
Whenever possible, a patient’s family members (particularly in the case of children) should be educated too.
Ask regularly about episodes. Finally, act to identify problems by querying patients and family members at every visit about hypoglycemia episodes, probing for information about awareness, severity and timing of the episodes, and how these events relate to dosing, meals, and activities.27 If hypoglycemia recurs, analyze the dosing regimen and consider flexible insulin dosing.1
Anti-hypoglycemia strategies for each new phase of therapy
As the course of diabetes therapy moves, typically, from oral medications to insulin to combination regimens, drug selections can be made in part to reduce the risk of hypoglycemia.
Oral agents: Insulin sensitizers, newer agents generally better
As noted earlier, among oral agents, insulin sensitizers are generally thought to have lower rates of hypoglycemia.
Newer sulfonylureas such as glimepiride and the rapid-acting meglitinides may also cause fewer hypoglycemic events.
Given the progressive decline of endogenous insulin secretion, combination therapy with secretagogues or insulin is eventually required for most patients.
Insulin analogs
A number of rapid-, short-, intermediateand long-acting insulin analogs have been introduced, and many of them make it possible to mimic different phases of physiologic insulin secretion (FIGURE 2). One of the newer analogs less likely to cause hypoglycemia is glargine, a long-acting insulin with a steady, relatively consistent action profile over a 24-hour period, closely mimicking normal basal pancreatic insulin secretion.28
FIGURE 2
Plasma insulin levels with newer analogs
N=20 type 1 diabetic patients; mean±SE
CSII, continuous subcutaneous insulin infusion; NPH, neutral protamine Hagedorn.
Copyright © 2000 American Diabetes Association. From Diabetes 2000; 49:2142–2148.28 Reprinted with permission from American Diabetes Association.
TABLE 2
Strategies for avoiding and addressing hypoglycemia
|
Insulin mixtures helpful when meal times guaranteed
Other insulins include mixtures of regular insulin and long-acting insulin available in split mixed or premixed formulations. These mixtures are intended to cover insulin peaks at mealtimes with twice-daily administration.
Mixed insulin formulations are often perceived as relatively convenient and simple to use, but they require meals to be taken within set time frames, without a great deal of flexibility. Since the ratios of the insulin components are fixed, and designed to work with meals consumed on a fixed schedule, hypoglycemia can occur if patients miss a meal. In addition, the time-activity profile of the insulin may not match the postprandial glucose peak even if the meal is consumed, and will increase the chance of postprandial hypoglycemia.
Basal insulin plus oral regimens
For patients with type 2 diabetes, adding basal insulin to oral regimens can significantly improve glycemic control. Ideally, basal insulin therapy provides a sustained and relatively constant concentration of insulin throughout the day. In the past, neutral protamine Hagedorn (NPH) insulin, a longer-acting insulin, was used for basal insulin therapy, and regular insulin was used to cover prandial insulin needs. Ultralente, also used as a basal insulin, has a relatively unpredictable timeactivity profile.28
Insulin glargine superior to NPH. In a recent clinical trial, patients with type 2 diabetes whose glucose levels were inadequately controlled on oral antidiabetic medications were given bedtime insulin glargine or NPH insulin.29 The insulin doses were titrated using a simple algorithm targeting a fasting plasma glucose (FPG) of ≤100 mg/dL to reach recommended glycosylated hemoglobin (Hb A1c) levels. Though no significant difference in glycemic control was found between insulin glargine and NPH insulin, significantly fewer hypoglycemic episodes occurred with insulin glargine therapy. The 24-hour distribution per patient-year of hypoglycemia for glargine vs NPH is shown in FIGURE 3.
Specifically, nearly 25% more patients treated with insulin glargine than with NPH insulin reached target Hb A1c levels of ≤7.0% without nocturnal hypoglycemia. Moreover, the overall incidence of any hypoglycemic event (eg, plasma-referenced glucose ≤72 mg/dL) and severe hypoglycemia (eg, patient required assistance of another person, and had a glucose level <56 mg/dL or prompt recovery after glucose or glucagons) was lower with insulin glargine than with NPH insulin. Results from other studies and a recent metaanalysis have been similar.30-32
Thus, using insulin glargine as basal insulin allows patients to reach recommended targets with fewer episodes of hypoglycemia, and can help address patients’ fear that can be a barrier to initiating insulin therapy in type 2 diabetes. Two recent studies have reported that dosing of insulin glargine can be flexible—morning or bedtime administration yields comparable low rates of hypoglycemia.30,31
FIGURE 3
Hourly hypoglycemia rate with glargine much less than with NPH
*P<.05 (between treatment).
Copyright © 2003 American Diabetes Association. From Diabetes Care 2003; 26:3080–3086.29 Reprinted with permission from American Diabetes Association.
Basal insulin plus prandial insulin
For patients who cannot otherwise reach Hb A1c goals, basal insulin therapy may be supplemented with prandial insulin. Newer, rapid-acting analogs used for the prandial component are insulin lispro, insulin glulisine, or insulin aspart. Although this approach is physiologically more rational than regimens using conventional insulins, data are limited for use in type 2 diabetes.
The incidence of nocturnal hypoglycemia was evaluated in a study of patients with type 1 diabetes and impaired hypoglycemic awareness who were treated with 1 of 2 regimens: insulin lispro in a basal-prandial combination with NPH insulin, or twice-daily, premixed NPH/regular insulin.33 Results showed that the incidence of nocturnal hypoglycemia was lower in patients receiving the insulin lispro regimen.
Another study, comparing insulin aspart and regular insulin as the prandial component in a basal-prandial regimen with NPH, showed that postprandial glucose control and Hb A1c levels were significantly better after 1 year of treatment in the insulin aspart group than in the group receiving regular insulin, without an increased risk for hypoglycemia.34 These results suggest that treatment with rapidacting insulin analogs could be helpful in avoiding hypoglycemia in patients with type 2 diabetes when a basal-prandial insulin regimen is indicated.
Avoiding hypoglycemia in the elderly
Elderly patients may be at increased risk for iatrogenic hypoglycemia. A populationbased study of patients presenting to an emergency room with severe hypoglycemic symptoms reported that rates of such events among elderly patients with type 2 diabetes and multiple comorbidities approached the rates among patients with type 1 diabetes.35
Creatinine clearance is often decreased in elderly patients, slowing elimination of oral agents and insulin and potentially resulting in sustained pharmacological action and creating a greater risk for hypoglycemia.
Furthermore, there is evidence that the neurogenic symptoms of hypoglycemia are reduced in elderly patients, diminishing awareness of hypoglycemia.36
In the demented elderly, malnutrition, weight loss, and anorexia may exacerbate the risk for hypoglycemia. For elderly patients with tertiary disease (eg, cerebrovascular accident, myocardial infarction, congestive heart failure, blindness, chronic renal failure), the risk for hypoglycemia and subsequent comorbidity may outweigh the benefits of strict glycemic control.3,37 Elderly patients may have comorbid conditions that increase risk of falls (eg, poor vision, neurologic conditions), and hypoglycemic episodes may further increase the risk of falls and lead to morbidity (eg, fragility fracture in patients with osteopenia or osteoporosis).
Because the elderly are at a greater risk for hypoglycemia, a switch to a less restrictive diet, such as a “no concentrated sweets” diet, is an option, with control of glucose levels through the administration of oral agents or insulin.36 This may also promote a better quality of life, considering that many of these patients already have secondary and tertiary complications of diabetes, prevention of which is not a realistic goal.
CORRESPONDING AUTHOR
William Cefalu, MD, Professor and Chief, Division of Nutrition and Chronic Diseases, Pennington Biomedical Research Center, Louisiana State University, 6400 Perkins Road, Baton Rouge, LA 70808. Email: cefaluwt@pbrc.edu
- Advise patients to monitor blood glucose levels frequently and learn to correlate drops in glucose to symptoms, which vary among patients.
- Ask patients at each visit about awareness of hypoglycemic episodes, their severity and timing, and how events relate to dosing, meals, and activities.
- When using oral agents, consider insulin sensitizers or newer sulfonylureas or meglitinides to reduce risk of hypoglycemia.
- If adding basal insulin to an oral regimen, the analog glargine has proven superior to NPH insulin in avoiding hypoglycemia.
Signs and symptoms of hypoglycemia vary considerably among patients with type 2 diabetes, making the condition easy to miss. Moreover, the most common symptoms are not necessarily the first symptoms.
If hypoglycemia occurs repeatedly, it can start a vicious cycle of physiologic reactions that mask or diminish the symptoms that warn patients of an impending episode. This may lead to hypoglycemia unawareness and hypoglycemic episodes of increasing severity. Fear of hypoglycemia, particularly of nocturnal events, may discourage patients from more intensive glycemic control, particularly using insulin. Such fear may even lead them to reduce their antidiabetic medication dosage, resulting in poor glycemic control.1
Breaking this cycle and restoring normal physiologic responses is one focus of this article, as is teaching patients how to monitor their blood glucose levels and how to correlate low blood glucose with the signs and symptoms of hypoglycemia.
Other therapies and strategies that we discuss in this article:
- Newer insulin analogs and the associated risk of hypoglycemia with each
- Appropriate combination of insulin with oral antidiabetic medications
- The long-acting analog insulin glargine used as basal insulin to lower the incidence of hypoglycemia, including nocturnal and severe hypoglycemia
- Rapid-acting insulin analogs (aspart, glulisine, and lispro) used in basalprandial insulin regimens.
First symptoms vary among individuals
Symptoms of hypoglycemia result primarily from a lowered glucose level in the brain and its effects on the central and autonomic nervous systems (FIGURE 1). A decrease in glucose below physiologic levels has acute consequences for brain function because the brain has an immediate requirement for glucose and little capacity for storage.
Two types of symptoms
Neuroglycopenia and the inhibition of neuronal metabolism causes sensations of warmth, weakness, fatigue, difficulty concentrating, confusion, behavioral changes, and in the most severe cases, a loss of consciousness, seizures, brain damage, and even death.2-4
Neurogenic symptoms are mediated by the hormones and neurotransmitters secreted in response to low brain glucose levels (FIGURE 1). The gluconeogenic actions of the autonomic nervous system produce the classic warning symptoms—tremulousness, pounding heart, anxiety, sweating, hunger, and tingling sensations—that usually precede the symptoms of hypoglycemia.2-4 This is particularly so in iatrogenic hypoglycemia.
These direct symptoms of neuroglycopenia are the ones patients typically identify with hypoglycemia. The most common symptoms of hypoglycemia are therefore not necessarily the first symptoms of hypoglycemia (TABLE 1).5 For example, though most patients experience sweating as a symptom of hypoglycemia, the first symptom might be trembling or anxiety, depending on the individual.5
TABLE 1
Signs and symptoms most commonly associated with hypoglycemia are not always the first to appear
| SYMPTOM | FREQUENCY (%) | FIRST SYMPTOM (%) |
|---|---|---|
| Sweating | 78 | 25 |
| Trembling | 62 | 6 |
| Inability to concentrate | 49 | 6 |
| Confusion | 40 | 3 |
| Weakness | 36 | 4 |
| Dry mouth | 35 | 0 |
| Blurred vision | 34 | 3 |
| Hunger | 33 | 3 |
| Anxiety | 26 | 1 |
| Headache | 21 | 0 |
| Difficulty walking | 21 | 3 |
| Pounding heart | 20 | 0 |
| Tingling around mouth | 20 | 6 |
| Difficulty speaking | 17 | 0 |
| Drowsiness | 15 | 0 |
| Odd behavior | 13 | 1 |
| Nausea | 13 | 0 |
| Adapted from Hepburn DA, MacLeod KM, Pell AC, et al. Diabet Med 1993;10:231–237.5 | ||
Factors influencing frequency and severity of hypoglycemia
Aggressive diabetes management commonly causes mild-to-moderate hypoglycemia, defined as a blood glucose value <60 mg/dL, that can be managed by the patient without assistance.
Severe hypoglycemia—a blood glucose value <50 mg/dL—is relatively uncommon in type 2 diabetes and requires the assistance of another person to manage, since neurological impairment may render patients unable to treat themselves.2,6 Severe hypoglycemia, whether in patients with type 1 or type 2 diabetes, can have debilitating consequences, including seizures or coma or even death.7
Long-standing type 2 disease. Hypoglycemia is more common in patients with type 1 diabetes than in those with type 2, but it can occur in type 2 diabetes patients who require insulin or are treated intensively with combinations of oral agents.6 As type 2 diabetes progresses,8 the incidence of hypoglycemic events increases, as endogenously produced insulin declines and is replaced by exogenous insulin.5,9 In fact, the prevalence of severe episodes (eg, requiring assistance of another person to administer glucose or glucagon) in patients with type 2 diabetes was comparable to that exhibited among patients with type 1 diabetes if they had been on insulin therapy for the same length of time.5,10
Nocturnal hypoglycemia. This event poses a special concern because the warning signs of hypoglycemia may be blunted during sleep. It has been reported that as many as 29% to 56% of all adult patients treated with insulin have an overnight glucose profile that indicates hypoglycemia occurs at night.11-13 However, it is important to note that the extent of the problem of nocturnal hypoglycemia is difficult to assess since overnight monitoring of glucose levels is required.
Additional insights from the UKPDS
Hypoglycemia in type 2 diabetes has not received rigorous attention in clinical trials. However, the United Kingdom Prospective Diabetes Study (UKPDS) was a large longitudinal trial in type 2 diabetes that included hypoglycemia as an outcome measure and thus provides some helpful information.
Events with insulin>sulfonylureas>diet. The 6-year follow-up revealed that the cumulative proportion of patients reporting 1 or more hypoglycemic events (of any type) was 76% for those using insulin, 45% among those taking sulfonylureas, and 3% for those on diet alone. Expressed as events per patient year, this was 37%, 17%, and 0.9%, respectively. When only “major” events (those requiring third-party assistance or hospital admission) were considered, the proportion of patients per year reporting 1 or more such events, was 2.3% for insulin, 0.7% for sulfonylureas, and 0.03% for diet alone. The cumulative proportion over 6 years was 3.3% of participants using sulfonylureas, 11.2% of those using insulin, and 0.15% of those on diet therapy.8
Metformin increases risk. The cumulative proportion of obese patients reporting any hypoglycemic event was 17.6% for those taking metformin vs 2.8% for those on diet. Severe hypoglycemia (as defined earlier) occurred in 2.4% of participants using metformin compared with 0.4% of those on diet therapy.8
Findings from other studies
Interestingly, in a recent systematic review of randomized controlled trials comparing insulin monotherapy with insulin plus oral antidiabetic agents, 13 of 14 studies reporting hypoglycemia demonstrated no difference in events.14
The occurrence of hypoglycemia among patients on metformin monotherapy in the UKPDS study is notable since, theoretically, hypoglycemia should not occur with agents whose mechanisms of action do not increase insulin secretion (biguanides, thiazolidinediones [TZDs], or α-glucosidase inhibitors),1 Since newer classes such as TZDs, α-glucosidase inhibitors, and meglitinides were not available when UKPDS was initiated, the trial does not provide data on these classes.
In a small comparative study of insulin combined with either metformin or a TZD, it appeared that metformin combination was associated with fewer occurrences of hypoglycemia; however, the small patient sample limits generalizability of the finding.15
How hypoglycemia occurs
Normally, as blood glucose levels (red) rise, insulin secretion increases, circulating insulin levels (blue) rise, and hepatic glucose production is inhibited. As glucose is disposed and circulating levels decrease due to insulin action, insulin levels then drop and hepatic glucose production begins again.
Hypoglycemia in diabetes can result from an excess of endogenous or exogenous insulin (iatrogenic hypoglycemia). In healthy patients, high insulin and falling glucose levels suppress insulin production and stimulate a hormone-mediated burst of glucose production. In patients with diabetes, the loss of physiologic control of insulin secretion coupled with exogenous administration of insulin or insulin secretagogues can interfere with the normal physiologic response to low blood glucose levels, resulting in hypoglycemia.1
With intense insulin regimens, the incidence of hypoglycemia can be as high as 30%, in contrast to 12% for patients treated with diet alone and 16% for those taking oral agents.6
With secretagogues, it has been suggested that the incidence of hypoglycemia is higher with the older, longer acting sulfonylurea agents.1,6,16-19 Although populationbased data on hypoglycemic rates associated with combination therapy with oral antidiabetic agents are not available, numerous clinical studies have reported rates of 10% to 20% for any hypoglycemic event.20-23
Heightening patient awareness, and yours
Because the signs of hypoglycemia vary considerably among individuals, they can easily be missed.3 In addition, repeated episodes of hypoglycemia can alter the normal regulatory responses and diminish the most important signs of a drop in glucose levels.1,2,24,25 The loss of the physiologic warning signs is thought to stem from dampening and eventual loss of the neuroadrenal response to low glucose levels in the brain (FIGURE 1). A vicious cycle is set up, whereby reduction in the neurogenic response attenuates hypoglycemic symptoms, causing more episodes to occur and become more severe as they are repeated. This cycle can be broken, and the normal physiologic response restored, if hypoglycemic events can be avoided for just a few weeks.2,26
Key points for patients. The main strategy for managing hypoglycemia is educating patients about the early symptoms of hypoglycemia and how to self-treat when they occur. Reinforce the need to time meals consistently and to limit the amount of carbohydrate ingested.
Advise patients to monitor blood glucose levels frequently, and to learn to relate a drop in glucose levels to hypoglycemic symptoms.2
Counsel patients to eat a snack or, preferably, drink fruit juice to counteract hypoglycemia. Patients may also carry glucose tablets, which are convenient and less tempting than candy.27
Glucagon is indicated for severe cases.
Whenever possible, a patient’s family members (particularly in the case of children) should be educated too.
Ask regularly about episodes. Finally, act to identify problems by querying patients and family members at every visit about hypoglycemia episodes, probing for information about awareness, severity and timing of the episodes, and how these events relate to dosing, meals, and activities.27 If hypoglycemia recurs, analyze the dosing regimen and consider flexible insulin dosing.1
Anti-hypoglycemia strategies for each new phase of therapy
As the course of diabetes therapy moves, typically, from oral medications to insulin to combination regimens, drug selections can be made in part to reduce the risk of hypoglycemia.
Oral agents: Insulin sensitizers, newer agents generally better
As noted earlier, among oral agents, insulin sensitizers are generally thought to have lower rates of hypoglycemia.
Newer sulfonylureas such as glimepiride and the rapid-acting meglitinides may also cause fewer hypoglycemic events.
Given the progressive decline of endogenous insulin secretion, combination therapy with secretagogues or insulin is eventually required for most patients.
Insulin analogs
A number of rapid-, short-, intermediateand long-acting insulin analogs have been introduced, and many of them make it possible to mimic different phases of physiologic insulin secretion (FIGURE 2). One of the newer analogs less likely to cause hypoglycemia is glargine, a long-acting insulin with a steady, relatively consistent action profile over a 24-hour period, closely mimicking normal basal pancreatic insulin secretion.28
FIGURE 2
Plasma insulin levels with newer analogs
N=20 type 1 diabetic patients; mean±SE
CSII, continuous subcutaneous insulin infusion; NPH, neutral protamine Hagedorn.
Copyright © 2000 American Diabetes Association. From Diabetes 2000; 49:2142–2148.28 Reprinted with permission from American Diabetes Association.
TABLE 2
Strategies for avoiding and addressing hypoglycemia
|
Insulin mixtures helpful when meal times guaranteed
Other insulins include mixtures of regular insulin and long-acting insulin available in split mixed or premixed formulations. These mixtures are intended to cover insulin peaks at mealtimes with twice-daily administration.
Mixed insulin formulations are often perceived as relatively convenient and simple to use, but they require meals to be taken within set time frames, without a great deal of flexibility. Since the ratios of the insulin components are fixed, and designed to work with meals consumed on a fixed schedule, hypoglycemia can occur if patients miss a meal. In addition, the time-activity profile of the insulin may not match the postprandial glucose peak even if the meal is consumed, and will increase the chance of postprandial hypoglycemia.
Basal insulin plus oral regimens
For patients with type 2 diabetes, adding basal insulin to oral regimens can significantly improve glycemic control. Ideally, basal insulin therapy provides a sustained and relatively constant concentration of insulin throughout the day. In the past, neutral protamine Hagedorn (NPH) insulin, a longer-acting insulin, was used for basal insulin therapy, and regular insulin was used to cover prandial insulin needs. Ultralente, also used as a basal insulin, has a relatively unpredictable timeactivity profile.28
Insulin glargine superior to NPH. In a recent clinical trial, patients with type 2 diabetes whose glucose levels were inadequately controlled on oral antidiabetic medications were given bedtime insulin glargine or NPH insulin.29 The insulin doses were titrated using a simple algorithm targeting a fasting plasma glucose (FPG) of ≤100 mg/dL to reach recommended glycosylated hemoglobin (Hb A1c) levels. Though no significant difference in glycemic control was found between insulin glargine and NPH insulin, significantly fewer hypoglycemic episodes occurred with insulin glargine therapy. The 24-hour distribution per patient-year of hypoglycemia for glargine vs NPH is shown in FIGURE 3.
Specifically, nearly 25% more patients treated with insulin glargine than with NPH insulin reached target Hb A1c levels of ≤7.0% without nocturnal hypoglycemia. Moreover, the overall incidence of any hypoglycemic event (eg, plasma-referenced glucose ≤72 mg/dL) and severe hypoglycemia (eg, patient required assistance of another person, and had a glucose level <56 mg/dL or prompt recovery after glucose or glucagons) was lower with insulin glargine than with NPH insulin. Results from other studies and a recent metaanalysis have been similar.30-32
Thus, using insulin glargine as basal insulin allows patients to reach recommended targets with fewer episodes of hypoglycemia, and can help address patients’ fear that can be a barrier to initiating insulin therapy in type 2 diabetes. Two recent studies have reported that dosing of insulin glargine can be flexible—morning or bedtime administration yields comparable low rates of hypoglycemia.30,31
FIGURE 3
Hourly hypoglycemia rate with glargine much less than with NPH
*P<.05 (between treatment).
Copyright © 2003 American Diabetes Association. From Diabetes Care 2003; 26:3080–3086.29 Reprinted with permission from American Diabetes Association.
Basal insulin plus prandial insulin
For patients who cannot otherwise reach Hb A1c goals, basal insulin therapy may be supplemented with prandial insulin. Newer, rapid-acting analogs used for the prandial component are insulin lispro, insulin glulisine, or insulin aspart. Although this approach is physiologically more rational than regimens using conventional insulins, data are limited for use in type 2 diabetes.
The incidence of nocturnal hypoglycemia was evaluated in a study of patients with type 1 diabetes and impaired hypoglycemic awareness who were treated with 1 of 2 regimens: insulin lispro in a basal-prandial combination with NPH insulin, or twice-daily, premixed NPH/regular insulin.33 Results showed that the incidence of nocturnal hypoglycemia was lower in patients receiving the insulin lispro regimen.
Another study, comparing insulin aspart and regular insulin as the prandial component in a basal-prandial regimen with NPH, showed that postprandial glucose control and Hb A1c levels were significantly better after 1 year of treatment in the insulin aspart group than in the group receiving regular insulin, without an increased risk for hypoglycemia.34 These results suggest that treatment with rapidacting insulin analogs could be helpful in avoiding hypoglycemia in patients with type 2 diabetes when a basal-prandial insulin regimen is indicated.
Avoiding hypoglycemia in the elderly
Elderly patients may be at increased risk for iatrogenic hypoglycemia. A populationbased study of patients presenting to an emergency room with severe hypoglycemic symptoms reported that rates of such events among elderly patients with type 2 diabetes and multiple comorbidities approached the rates among patients with type 1 diabetes.35
Creatinine clearance is often decreased in elderly patients, slowing elimination of oral agents and insulin and potentially resulting in sustained pharmacological action and creating a greater risk for hypoglycemia.
Furthermore, there is evidence that the neurogenic symptoms of hypoglycemia are reduced in elderly patients, diminishing awareness of hypoglycemia.36
In the demented elderly, malnutrition, weight loss, and anorexia may exacerbate the risk for hypoglycemia. For elderly patients with tertiary disease (eg, cerebrovascular accident, myocardial infarction, congestive heart failure, blindness, chronic renal failure), the risk for hypoglycemia and subsequent comorbidity may outweigh the benefits of strict glycemic control.3,37 Elderly patients may have comorbid conditions that increase risk of falls (eg, poor vision, neurologic conditions), and hypoglycemic episodes may further increase the risk of falls and lead to morbidity (eg, fragility fracture in patients with osteopenia or osteoporosis).
Because the elderly are at a greater risk for hypoglycemia, a switch to a less restrictive diet, such as a “no concentrated sweets” diet, is an option, with control of glucose levels through the administration of oral agents or insulin.36 This may also promote a better quality of life, considering that many of these patients already have secondary and tertiary complications of diabetes, prevention of which is not a realistic goal.
CORRESPONDING AUTHOR
William Cefalu, MD, Professor and Chief, Division of Nutrition and Chronic Diseases, Pennington Biomedical Research Center, Louisiana State University, 6400 Perkins Road, Baton Rouge, LA 70808. Email: cefaluwt@pbrc.edu
1. Cryer PE, Childs BP. Negotiating the barrier of hypoglycemia in diabetes. Diabetes Spectrum 2002;15:20-27.
2. Cryer PE, ed. Hypoglycemia: Pathophysiology, Diagnosis, and Treatment. New York, NY: Oxford University Press; 1997.
3. McAulay V, Deary IJ, Frier BM. Symptoms of hypoglycaemia in people with diabetes. Diabet Med 2001;18:690-705.
4. Towler DA, Havlin CE, Craft S, Cryer P. Mechanism of awareness of hypoglycemia. Perception of neurogenic (predominantly cholinergic) rather than neuroglycopenic symptoms. Diabetes 1993;42:1791-1798.
5. Hepburn DA, MacLeod KM, Pell AC, Scougal IJ, Frier BM. Frequency and symptoms of hypoglycaemia experienced by patients with type 2 diabetes treated with insulin. Diabet Med 1993;10:231-237.
6. Miller CD, Phillips LS, Ziemer DC, Gallina DL, Cook CB, El-Kebbi IM. Hypoglycemia in patients with type 2 diabetes mellitus. Arch Intern Med 2001;161:1653-1659.
7. The DCCT Research Group. Epidemiology of severe hypoglycemia in the diabetes control and complications trial. Am J Med 1991;90:450-459.
8. UK Prospective Diabetes Study Group. Perspectives in diabetes. UK Prospective Diabetes Study 16: overview of 6 years’ therapy of type II diabetes: a progressive disease. Diabetes 1995;44:1249-1258.
9. United Kingdom Prospective Diabetes Study Group. UKPDS 24: a 6-year, randomized, controlled trial comparing sulfonylurea, insulin, and metformin therapy in patients with newly diagnosed type 2 diabetes that could not be controlled with diet therapy. Ann Intern Med 1998;128:165-175.
10. MacLeod KM, Hepburn DA, Frier BM. Frequency and morbidity of severe hypoglycaemia in insulin-treated diabetic patients. Diabet Med 1993;10:238-245.
11. Vervoort G, Goldschmidt HM, van Doorn LG. Nocturnal blood glucose profiles in patients with type 1 diabetes mellitus on multiple (>or=4) daily insulin injection regimens. Diabet Med 1996;13:794-799.
12. Gale EA, Tattersall RB. Unrecognised nocturnal hypoglycaemia in insulin-treated diabetics. Lancet 1979;1:1049-1052.
13. Pramming S, Thorsteinsson B, Bendtson I, Ronn B, Binder C. Nocturnal hypoglycaemia in patients receiving conventional treatment with insulin. Br Med J (Clin Res Ed) 1985;291:376-379.
14. Goudswaard AN, Furlong NJ, Valk GD, Stolk RP, Rutten GEHM. Insulin monotherapy versus combinations of insulin with oral hypoglycaemic agents in patients with type 2 diabetes mellitus. Cochrane Database Syst Rev 2004;4:1-51.
15. Strowig SM, Avilés-Santa ML, Raskin P. Comparison of insulin monotherapy and combination therapy with insulin and metformin or insulin and troglitazone in type 2 diabetes. Diabetes Care 2002;25:1691-1698.
16. Campbell IW. Hypoglycaemia and type 2 diabetes: sulphonylureas. In: Frier BM, Fisher BM, eds. Hypoglycaemia and Diabetes: Clinical and Physiological Aspects. London, UK: Edward Arnold, 1993;387-392.
17. Shorr RI, Ray WA, Daugherty JR, Griffin MR. Individual sulfonylureas and serious hypoglycemia in older people. J Am Geriatr Soc 1996;44:751-755.
18. Holstein A, Egberts EH. Risk of hypoglycaemia with oral antidiabetic agents in patients with Type 2 diabetes. Exp Clin Endocrinol Diabetes 2003;111:405-414.
19. Holstein A, Plaschke A, Egberts E-H. Lower incidence of severe hypoglycaemia in patients with type 2 diabetes treated with glimepiride versus glibenclamide. Diabetes Metab Res Rev 2001;17:467-473.
20. Aljabri K, Kozak SE, Thompson DM. Addition of pioglitazone or bedtime insulin to maximal doses of sulfonylurea and metformin in type 2 diabetes patients with poor glucose control: a prospective, randomized trial. Am J Med 2004;116:230-235.
21. Wolffenbuttel BHR, Landgraf R. A 1-year multicenter randomized double-blind comparison of repaglinide and glyburide for the treatment of type 2 diabetes. Dutch and German Repaglinide Study Group. Diabetes Care 1999;22:463-467.
22. Raskin P, McGill J, Saad MF, et al. Combination therapy for type 2 diabetes: repaglinide plus rosiglitazone. Diabetes Med 2004;21:329-335.
23. Dailey GE, III, Noor MA, Park JS, Bruce S, Fiedorek FT. Glycemic control with glyburide/metformin tablets in combination with rosiglitazone in patients with type 2 diabetes: a randomized, double-blind trial. Am J Med 2004;116:223-229.
24. Korytkowski MT, Mokan M, Veneman TF, Mitrakou A, Cryer PE, Gerich JE. Reduced beta-adrenergic sensitivity in patients with type 1 diabetes and hypoglycemia unawareness. Diabetes Care 1998;21:1939-1943.
25. Fritsche A, Stumvoll M, Grub M, et al. Effect of hypoglycemia on beta-adrenergic sensitivity in normal and type 1 diabetic subjects. Diabetes Care 1998;21:1505-1510.
26. Fritsche A, Stefan N, Haring H, Gerich J, Stumvoll M. Avoidance of hypoglycemia restores hypoglycemia awareness by increasing beta-adrenergic sensitivity in type 1 diabetes. Ann Intern Med 2001;134:729-736.
27. Cryer PE, Davis SN, Shamoon H. Hypoglycemia in diabetes. Diabetes Care 2003;26:1902-1912.
28. Lepore M, Pampanelli S, Fanelli C, et al. Pharmacokinetics and pharmacodynamics of subcutaneous injection of long-acting human insulin analog glargine, NPH insulin, and ultralente human insulin and continuous subcutaneous infusion of insulin lispro. Diabetes 2000;49:2142-2148.
29. Riddle MC, Rosenstock J, Gerich J. The Treat-to-Target Trial: randomized addition of glargine or human NPH insulin to oral therapy of type 2 diabetic patients. Diabetes Care 2003;26:3080-3086.
30. Fritsche A, Schweitzer MA, Häring HU. Glimepiride combined with morning insulin glargine, bedtime neutral protamine Hagedorn insulin, or bedtime insulin glargine in patients with type 2 diabetes: a randomized, controlled trial. Ann Intern Med 2003;138:952-959.
31. Standl E, Maxeiner S, Raptis S, Karimi-Anderesi Z, Schweitzer MA. Good glycemic control with flexibility in timing of basal insulin supply: a 24-week comparison of insulin glargine given once daily in the morning or at bedtime in combination with morning glimepiride. Diabetes Care 2005;28:419-420.
32. Rosenstock J, Dailey G, Massi Benedetti M, Fritsche A, Lin Z, Salzman A. Reduced hypoglycemia risk with insulin glargine. A meta-analysis comparing insulin glargine with human NPH insulin in type 2 diabetes. Diabetes Care 2005;28:950-955.
33. Ferguson SC, Strachan MW, Janes JM, Frier BM. Severe hypoglycaemia in patients with type 1 diabetes and impaired awareness of hypoglycaemia: a comparative study of insulin lispro and regular human insulin. Diabetes Metab Res Rev 2001;17:285-291.
34. Raskin P, Guthrie RA, Leiter L, Riis A, Jovanovic L. Use of insulin aspart, a fast-acting insulin analog, as the mealtime insulin in the management of patients with type 1 diabetes. Diabetes Care 2000;23:583-558.
35. Holstein A, Plaschke A, Egberts EH. Clinical characterisation of severe hypoglycaemia-a prospective population-based study. Exp Clin Endocrinol Diabetes 2003;111:364-369.
36. Meneilly GS, Cheung E, Tuokko H. Counterregulatory hormone responses to hypoglycemia in the elderly patient with diabetes. Diabetes 1994;43:403-410.
37. American Diabetes Association. Standards of medical care for patients with diabetes mellitus. Diabetes Care 2003;26(suppl 1):S33-S50.
1. Cryer PE, Childs BP. Negotiating the barrier of hypoglycemia in diabetes. Diabetes Spectrum 2002;15:20-27.
2. Cryer PE, ed. Hypoglycemia: Pathophysiology, Diagnosis, and Treatment. New York, NY: Oxford University Press; 1997.
3. McAulay V, Deary IJ, Frier BM. Symptoms of hypoglycaemia in people with diabetes. Diabet Med 2001;18:690-705.
4. Towler DA, Havlin CE, Craft S, Cryer P. Mechanism of awareness of hypoglycemia. Perception of neurogenic (predominantly cholinergic) rather than neuroglycopenic symptoms. Diabetes 1993;42:1791-1798.
5. Hepburn DA, MacLeod KM, Pell AC, Scougal IJ, Frier BM. Frequency and symptoms of hypoglycaemia experienced by patients with type 2 diabetes treated with insulin. Diabet Med 1993;10:231-237.
6. Miller CD, Phillips LS, Ziemer DC, Gallina DL, Cook CB, El-Kebbi IM. Hypoglycemia in patients with type 2 diabetes mellitus. Arch Intern Med 2001;161:1653-1659.
7. The DCCT Research Group. Epidemiology of severe hypoglycemia in the diabetes control and complications trial. Am J Med 1991;90:450-459.
8. UK Prospective Diabetes Study Group. Perspectives in diabetes. UK Prospective Diabetes Study 16: overview of 6 years’ therapy of type II diabetes: a progressive disease. Diabetes 1995;44:1249-1258.
9. United Kingdom Prospective Diabetes Study Group. UKPDS 24: a 6-year, randomized, controlled trial comparing sulfonylurea, insulin, and metformin therapy in patients with newly diagnosed type 2 diabetes that could not be controlled with diet therapy. Ann Intern Med 1998;128:165-175.
10. MacLeod KM, Hepburn DA, Frier BM. Frequency and morbidity of severe hypoglycaemia in insulin-treated diabetic patients. Diabet Med 1993;10:238-245.
11. Vervoort G, Goldschmidt HM, van Doorn LG. Nocturnal blood glucose profiles in patients with type 1 diabetes mellitus on multiple (>or=4) daily insulin injection regimens. Diabet Med 1996;13:794-799.
12. Gale EA, Tattersall RB. Unrecognised nocturnal hypoglycaemia in insulin-treated diabetics. Lancet 1979;1:1049-1052.
13. Pramming S, Thorsteinsson B, Bendtson I, Ronn B, Binder C. Nocturnal hypoglycaemia in patients receiving conventional treatment with insulin. Br Med J (Clin Res Ed) 1985;291:376-379.
14. Goudswaard AN, Furlong NJ, Valk GD, Stolk RP, Rutten GEHM. Insulin monotherapy versus combinations of insulin with oral hypoglycaemic agents in patients with type 2 diabetes mellitus. Cochrane Database Syst Rev 2004;4:1-51.
15. Strowig SM, Avilés-Santa ML, Raskin P. Comparison of insulin monotherapy and combination therapy with insulin and metformin or insulin and troglitazone in type 2 diabetes. Diabetes Care 2002;25:1691-1698.
16. Campbell IW. Hypoglycaemia and type 2 diabetes: sulphonylureas. In: Frier BM, Fisher BM, eds. Hypoglycaemia and Diabetes: Clinical and Physiological Aspects. London, UK: Edward Arnold, 1993;387-392.
17. Shorr RI, Ray WA, Daugherty JR, Griffin MR. Individual sulfonylureas and serious hypoglycemia in older people. J Am Geriatr Soc 1996;44:751-755.
18. Holstein A, Egberts EH. Risk of hypoglycaemia with oral antidiabetic agents in patients with Type 2 diabetes. Exp Clin Endocrinol Diabetes 2003;111:405-414.
19. Holstein A, Plaschke A, Egberts E-H. Lower incidence of severe hypoglycaemia in patients with type 2 diabetes treated with glimepiride versus glibenclamide. Diabetes Metab Res Rev 2001;17:467-473.
20. Aljabri K, Kozak SE, Thompson DM. Addition of pioglitazone or bedtime insulin to maximal doses of sulfonylurea and metformin in type 2 diabetes patients with poor glucose control: a prospective, randomized trial. Am J Med 2004;116:230-235.
21. Wolffenbuttel BHR, Landgraf R. A 1-year multicenter randomized double-blind comparison of repaglinide and glyburide for the treatment of type 2 diabetes. Dutch and German Repaglinide Study Group. Diabetes Care 1999;22:463-467.
22. Raskin P, McGill J, Saad MF, et al. Combination therapy for type 2 diabetes: repaglinide plus rosiglitazone. Diabetes Med 2004;21:329-335.
23. Dailey GE, III, Noor MA, Park JS, Bruce S, Fiedorek FT. Glycemic control with glyburide/metformin tablets in combination with rosiglitazone in patients with type 2 diabetes: a randomized, double-blind trial. Am J Med 2004;116:223-229.
24. Korytkowski MT, Mokan M, Veneman TF, Mitrakou A, Cryer PE, Gerich JE. Reduced beta-adrenergic sensitivity in patients with type 1 diabetes and hypoglycemia unawareness. Diabetes Care 1998;21:1939-1943.
25. Fritsche A, Stumvoll M, Grub M, et al. Effect of hypoglycemia on beta-adrenergic sensitivity in normal and type 1 diabetic subjects. Diabetes Care 1998;21:1505-1510.
26. Fritsche A, Stefan N, Haring H, Gerich J, Stumvoll M. Avoidance of hypoglycemia restores hypoglycemia awareness by increasing beta-adrenergic sensitivity in type 1 diabetes. Ann Intern Med 2001;134:729-736.
27. Cryer PE, Davis SN, Shamoon H. Hypoglycemia in diabetes. Diabetes Care 2003;26:1902-1912.
28. Lepore M, Pampanelli S, Fanelli C, et al. Pharmacokinetics and pharmacodynamics of subcutaneous injection of long-acting human insulin analog glargine, NPH insulin, and ultralente human insulin and continuous subcutaneous infusion of insulin lispro. Diabetes 2000;49:2142-2148.
29. Riddle MC, Rosenstock J, Gerich J. The Treat-to-Target Trial: randomized addition of glargine or human NPH insulin to oral therapy of type 2 diabetic patients. Diabetes Care 2003;26:3080-3086.
30. Fritsche A, Schweitzer MA, Häring HU. Glimepiride combined with morning insulin glargine, bedtime neutral protamine Hagedorn insulin, or bedtime insulin glargine in patients with type 2 diabetes: a randomized, controlled trial. Ann Intern Med 2003;138:952-959.
31. Standl E, Maxeiner S, Raptis S, Karimi-Anderesi Z, Schweitzer MA. Good glycemic control with flexibility in timing of basal insulin supply: a 24-week comparison of insulin glargine given once daily in the morning or at bedtime in combination with morning glimepiride. Diabetes Care 2005;28:419-420.
32. Rosenstock J, Dailey G, Massi Benedetti M, Fritsche A, Lin Z, Salzman A. Reduced hypoglycemia risk with insulin glargine. A meta-analysis comparing insulin glargine with human NPH insulin in type 2 diabetes. Diabetes Care 2005;28:950-955.
33. Ferguson SC, Strachan MW, Janes JM, Frier BM. Severe hypoglycaemia in patients with type 1 diabetes and impaired awareness of hypoglycaemia: a comparative study of insulin lispro and regular human insulin. Diabetes Metab Res Rev 2001;17:285-291.
34. Raskin P, Guthrie RA, Leiter L, Riis A, Jovanovic L. Use of insulin aspart, a fast-acting insulin analog, as the mealtime insulin in the management of patients with type 1 diabetes. Diabetes Care 2000;23:583-558.
35. Holstein A, Plaschke A, Egberts EH. Clinical characterisation of severe hypoglycaemia-a prospective population-based study. Exp Clin Endocrinol Diabetes 2003;111:364-369.
36. Meneilly GS, Cheung E, Tuokko H. Counterregulatory hormone responses to hypoglycemia in the elderly patient with diabetes. Diabetes 1994;43:403-410.
37. American Diabetes Association. Standards of medical care for patients with diabetes mellitus. Diabetes Care 2003;26(suppl 1):S33-S50.