User login
One Expert’s Approach in Transplant-Ineligible, Newly Diagnosed Multiple Myeloma
1. The treatment of multiple myeloma has evolved significantly in recent years. What are some of the most important things you consider in the treatment of your newly diagnosed, transplant-ineligible patients?
We’ve seen great progress in the treatment of multiple myeloma over the last decade, and outcomes continue to improve for many patients.1 Still, it is important to keep in mind that more than 34,000 patients will be diagnosed and more than 12,000 people will die from the disease this year.2 We may have the greatest opportunity to impact the course of disease in the treatment of newly diagnosed patients due to the nature of this cancer:
- Multiple myeloma is characterized by relapse, and we know the length of remission generally decreases with each relapse and subsequent line of therapy.3
- Patients often become refractory to treatment over time.
When I meet with a patient who has been diagnosed with multiple myeloma, the first thing I consider is their eligibility for autologous stem cell transplant (ASCT). In my opinion, the introduction of ASCT is one of the biggest advancements in the last few decades, and we’ve found that ASCT followed by maintenance therapy with targeted tools improves progression-free survival (PFS).4
Unfortunately, many newly diagnosed patients are not eligible for ASCT–either because of comorbidities or other complexities related to the presentation of their disease.
For patients who are transplant-ineligible (TIE), it is important to have treatment options that are proven effective in extending PFS and overall survival (OS), and capable of producing deep and durable responses.
2. What are the challenges associated with treating newly diagnosed patients who are not eligible for ASCT?
We still consider multiple myeloma to be an incurable disease but, in my opinion, the treatment of TIE patients is less challenging today than a decade ago due to the emergence of novel therapies. That said, TIE patients are typically older and present with more advanced disease and comorbidities, including diabetes or cardiovascular events.5
A retrospective analysis published in 2020 by Rafael Fonseca examined frontline treatment patterns and attrition rates by line of therapy among newly diagnosed multiple myeloma (NDMM) patients who are TIE. More than 22,000 patients were identified from three patient-level databases between 2000 and 2018 - the OPTUM Commercial Claims database, the OPTUM Electronic Medical Records database, and the Surveillance, Epidemiology, and End Results-Meidcare Linked database. Patients included had to have a multiple myeloma diagnosis on or after January 1, 2007. Results showed that attrition rates among newly diagnosed, TIE patients with multiple myeloma increase with each line of therapy, with the proportion of patients who receive a second line of therapy decreasing by 50 percent with each subsequent line.3
3. Can you provide more detail on the goals of therapy for newly diagnosed, transplant-ineligible patients?
When I discuss treatment goals with TIE patients, I feel it is important to emphasize managing side effects and achieving deep and durable responses. I have the benefit of being in an academic setting, where I regularly exchange information with my colleagues about what we’re learning from the clinical studies in which we participate. Choosing which treatment to administer is complex and involves other considerations. For example, if two regimens have comparable efficacy, I may recommend the regimen with a more established safety profile or more robust evidence so I can properly anticipate and manage toxicities in my patients. Overall survival is one of the most important endpoints I consider, in addition to depth of response and PFS. In recent years, we’ve seen increasing evidence pointing to the importance of using a proven effective treatment in frontline patients that are ineligible for transplant.
4. A key study in newly-diagnosed, transplant-ineligible multiple myeloma is the Phase 3 MAIA study. Can you share the key takeaways from this study and discuss how the results have shaped treatment for this patient population?
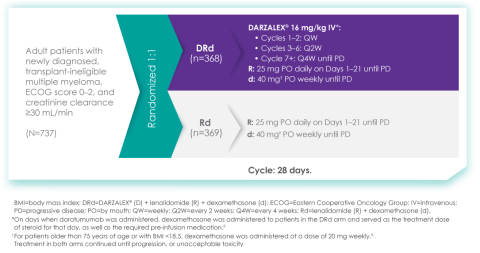
Of course. The MAIA study is a randomized Phase 3 study evaluating DARZALEX® (daratumumab) intravenous injection in combination with lenalidomide and dexamethasone (D-Rd) compared with Rd in 737 adult patients with newly diagnosed, transplant-ineligible multiple myeloma. The median age of patients participating in the MAIA study was 73 (range 45-90), an important consideration since the median age for multiple myeloma diagnosis is approximately 66-70 years of age.6 The study evaluated PFS as the primary endpoint, and overall survival as a key secondary endpoint, and supported the FDA approval of DARZALEX® in combination with lenalidomide and dexamethasone for adult patients with newly diagnosed, multiple myeloma who are ineligible for ASCT.
MAIA study design7
The baseline demographic and disease characteristics were similar between the 2 treatment groups. Forty-four percent of the patients were ≥75 years of age. Fifty-two percent (52%) of patients were male, 92% White, 4% Black or African American, and 1% Asian. Three percent (3%) of patients reported an ethnicity of Hispanic or Latino. Thirty-four (34%) had an Eastern Cooperative Oncology Group (ECOG) performance score of 0, 50% had an ECOG performance score of 1, and 17% had an ECOG performance score of ≥2. Twenty-seven percent had International Staging System (ISS) Stage I, 43% had ISS Stage II, and 29% had ISS Stage III disease.
Select Important Safety Information:
CONTRAINDICATIONS
DARZALEX® is contraindicated in patients with a history of severe hypersensitivity (eg, anaphylactic reactions) to daratumumab or any of the components of the formulation.
WARNINGS AND PRECAUTIONS
Infusion-Related Reactions: DARZALEX® can cause severe and/or serious infusion-related reactions including anaphylactic reactions.
These reactions can be life-threatening, and fatal outcomes have been reported. Please scroll down to read Important Safety Information for DARZALEX®.
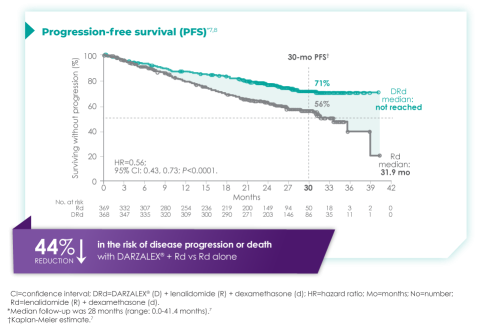
Primary findings from the study, which were published in 2019, showed an improvement in PFS in patients receiving D-Rd compared with those receiving Rd alone.7 The median PFS was not reached in the D-Rd arm and was reached at 31.9 months in the Rd arm (HR 0.56; 95% CI 0.43-0.73; P<0.0001).7 At a median of 30 months of follow-up, the data showed the clinical benefit of D-Rd therapy, with a 44% reduction in the risk of disease progression or death in patients receiving D-Rd compared with Rd alone.7
Additionally, 70.6% of patients (95% CI, 65.0-75.4) had no progressive disease with D-Rd treatment at median 30 months of follow-up, compared with 55.6% (95% CI, 49.5-61.3) of patients in the Rd group.7
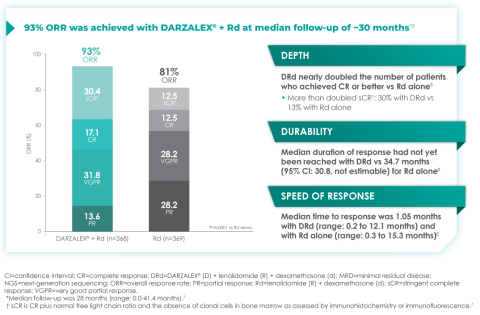
In terms of depth of response, the percentage of patients with a complete response or better was 47.6% in patients receiving D-Rd compared with 24.9% in the Rd group.7
Overall response rate with D-Rd in TIE NDMM at ~30 months of follow-up8
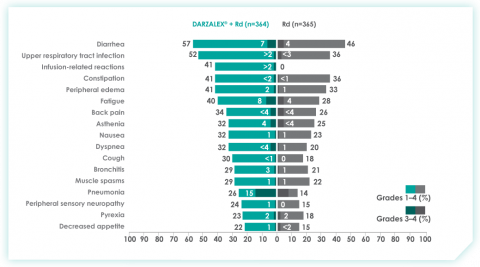
An overview of the most frequent adverse events at 30-months of follow-up are provided below. The most frequent adverse reactions were reported in ≥20% of patients, with at least a 5% greater frequency in the D-Rd arm compared with Rd alone.8
Most frequent adverse events at ~30 months of follow-up with D-Rd in TIE NDMM8
Most frequent hematologic laboratory abnormalities with D-Rd in TIE NDMM at ~30 months8
Serious adverse reactions with a 2% greater incidence in the D-Rd arm compared with the Rd arm were pneumonia (D-Rd 15% vs Rd 8%), bronchitis (D-Rd 4% vs Rd 2%), and dehydration (D-Rd 2% vs Rd <1).
• Discontinuation rates due to any adverse event: 7% with D-Rd vs 16% with Rd
• Infusion-related reactions (IRRs) with D-Rd occurred in 41% of patients; 2% were Grade 3 and <1% were Grade 4
• IRRs of any grade or severity may require management by interruption, modification, and/or discontinuation of the infusion
• Most IRRs occurred during first infusion
5. Thanks for that overview. In addition to these results, The Lancet Oncology has published updated overall survival data from a 5-year follow-up on the MAIA study. Can you provide an overview of these data and insights on their potential for patients?
The MAIA trial was an important study, and for me, the results were practice changing. We see that after a median of nearly 5 years of follow-up, D-Rd significantly improved OS in TIE NDMM patients who were treated to progression compared with Rd alone (66.3% vs. 53.1% [HR=0.68; 95% CI, 0.53-0.86; P=0.0013]).9 This equates to approximately a 32% reduction in death when DARZALEX® was added to a two-drug regimen, which is a meaningful consideration when selecting the most appropriate regimens for my newly diagnosed, transplant-ineligible patients.9
Overall survival data at ~5 years with D-Rd compared to Rd alone in TIE NDMM9
Importantly, efficacy that resulted from longer treatment with D-Rd is also supported by approximately 5 years of safety evaluation. Below is information from a follow-up analysis of the MAIA study. This information is not included in the current Prescribing Information and has not been evaluated by the FDA. Treatment-emergent adverse events are reported as observed. These analyses have not been adjusted for multiple comparisons and no conclusions should be drawn. In what I’ve observed through published data and in my practice, longer treatment has not revealed new safety signals.
Most frequent treatment-emergent adverse events (any grade reported in ≥30% of patients and/or Grade 3/4 reported in ≥10% of patients) at ~5 years9
Select Important Safety Information:
DARZALEX® can cause severe and/or serious infusion-related reactions including anaphylactic reactions. These reactions can be life threatening, and fatal outcomes have been reported. In clinical trials (monotherapy and combination: N=2066), infusion-related reactions occurred in 37% of patients with the Week 1 (16 mg/kg) infusion, 2% with the Week 2 infusion, and cumulatively 6% with subsequent infusions. Less than 1% of patients had a Grade 3/4 infusion-related reaction at Week 2 or subsequent infusions. The median time to onset was 1.5 hours (range: 0 to 73 hours). Nearly all reactions occurred during infusion or within 4 hours of completing DARZALEX®. Severe reactions have occurred, including bronchospasm, hypoxia, dyspnea, hypertension, tachycardia, headache, laryngeal edema, pulmonary edema, and ocular adverse reactions, including choroidal effusion, acute myopia, and acute angle closure glaucoma. Signs and symptoms may include respiratory symptoms, such as nasal congestion, cough, throat irritation, as well as chills, vomiting, and nausea. Less common signs and symptoms were wheezing, allergic rhinitis, pyrexia, chest discomfort, pruritus, hypotension and blurred vision. Please scroll down to see Important Safety Information for DARZALEX®.
6. Does the availability of OS data influence your decisions on treatment selection in TIE NDMM?
Overall survival absolutely remains the gold standard and informs my practice. Prior to OS data being available, I will often look at other efficacy endpoints that are available sooner. In MAIA, I was encouraged by efficacy endpoints in earlier data, which were later confirmed by the latest data on OS.
7. The MAIA study shows that treating to disease progression or unacceptable toxicity is important. How does that impact your approach to treatment?
It's important to keep in mind that the MAIA trial was designed to evaluate treatment until progression or unacceptable toxicity. The results revealed a significant difference between the DR-d and Rd treatment arms, but results observed in this study are contingent on this treatment approach. From a clinical perspective, unless there is considerable toxicity, I advocate for treating with D-Rd to progression.
In the clinic, we also see that TIE patients who have higher frailty scores are more likely to discontinue treatment prior to progression.10 There can be other reasons too – such as a patient simply wanting to have a break from treatment. These conversations are not always easy, but it is important to have an honest dialogue with patients.
8. What can we learn from studies like the MAIA trial that included a wide range of patient populations including patients who are elderly, frail, or had high cytogenetic risk?
Several patient subgroups were analyzed as part of the MAIA study. It is important to note that these subgroup analyses are not included in the Prescribing Information for DARZALEX®. These analyses were not adjusted for multiple comparisons, and there are insufficient numbers of patients per subgroup to make definitive conclusions of efficacy among the subgroups.
As mentioned above, the MAIA study evaluated a wide range of patients (n=737). The baseline demographic and disease characteristics were similar between the D-Rd and Rd treatment groups and the median age was 73 (range: 45-90) years, with 44% of the patients ≥75 years of age.
In the various patient subgroups that were analyzed as part of the MAIA study, it was found that at ~3-years of follow-up the PFS numerically favored DRd compared with Rd alone in most subgroups (see table below).
Median progression-free survival by sub-population at ~3 year follow-up8
The MAIA trial also included patients who were frail and a post hoc analysis was conducted in this subgroup of patients. These analyses are not included in the Prescribing Information for DARZALEX®. These analyses were conducted post hoc and there are insufficient numbers of patients per subgroup to make definitive conclusions of efficacy among the subgroups.
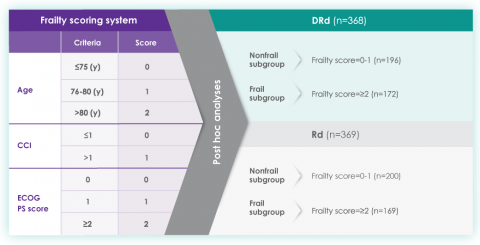
A frailty assessment was performed retrospectively using age, the Charlson Comorbidity Index (CCI) – which is calculated based on a retrospective review of the patient’s medical history to predict the 10-year mortality – and the baseline Eastern Cooperative Oncology Group (ECOG) performance status score, used to measure a patient’s level of functioning in terms of their ability to care for themselves, daily activity, and physical activity. The frailty scores were then added up to classify patients into fit (0), intermediate (1), or frail (≥2). Frailty status was further simplified into 2 categories: non-frail (0-1) and frail (≥2). The median age in the frail subgroup was 77 years (range: 57-80 years), with 88% of patients having ECOG performance score ≥1. CCI was calculated based on retrospective review of each patient’s medical history.12
The charts below illustrate the frailty scoring system with an overview of the patient population included in the 3-year post hoc analysis, PFS rate, and adverse events.
MAIA post hoc subgroup analysis by frailty status score12
The retrospective assessment of frailty score was a limitation of this study. Retrospective CCI calculations were based on reported medical history, which may contain missing data and result in underestimating or overestimating the number of patients in each frailty subgroup. The ECOG PS score parameter used for frailty score calculations in the study is more subjective, with susceptibility to intra- and inter-observer bias, compared with the ADL (activities of daily living) and IADL (instrumental activities of daily living) scales used in the IMWG scoring system. While the frailty scale used in the study is based on parameters that are routinely assessed in clinical practice for clinical use, the use of comprehensive frailty assessments that more accurately reflect biological or functional frailty will remain important for the further optimization of treatment strategies for frail patients. Patients with an ECOG PS score ≥3 and patients with comorbidities that may interfere with the study procedures were excluded from MAIA; the inclusion and exclusion criteria for the study limits the generalizability of these results to more frail patients seen in clinical practice.
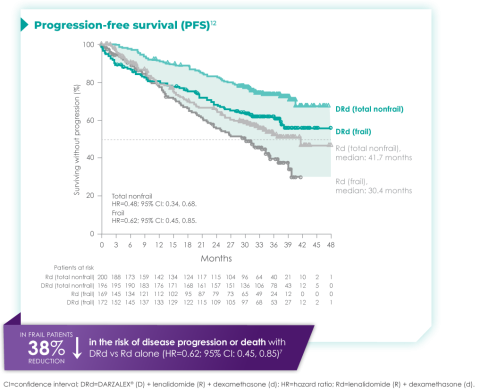
Progression-free survival in a ~3-year subgroup analysis of frail patients following treatment with D-Rd in TIE NDMM12
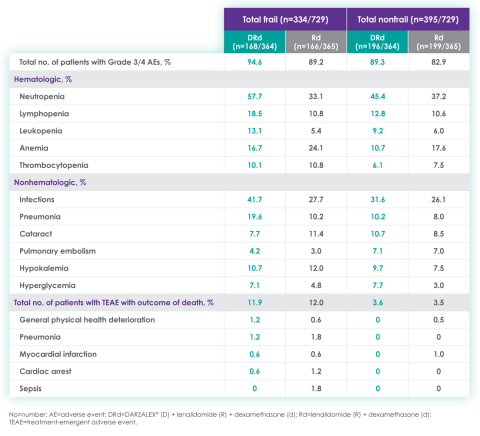
Most frequent Grade 3/4 treatment-emergent adverse events (≥10%) in frail patients at ~3 year follow-up of MAIA trial12
Please see additional Important Safety Information for DARZALEX® below.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
DARZALEX® is contraindicated in patients with a history of severe hypersensitivity (eg, anaphylactic reactions) to daratumumab or any of the components of the formulation.
WARNINGS AND PRECAUTIONS
Infusion-Related Reactions
DARZALEX® can cause severe and/or serious infusion-related reactions including anaphylactic reactions. These reactions can be life‑threatening, and fatal outcomes have been reported. In clinical trials (monotherapy and combination: N=2066), infusion-related reactions occurred in 37% of patients with the Week 1 (16 mg/kg) infusion, 2% with the Week 2 infusion, and cumulatively 6% with subsequent infusions. Less than 1% of patients had a Grade 3/4 infusion-related reaction at Week 2 or subsequent infusions. The median time to onset was 1.5 hours (range: 0 to 73 hours). Nearly all reactions occurred during infusion or within 4 hours of completing DARZALEX®. Severe reactions have occurred, including bronchospasm, hypoxia, dyspnea, hypertension, tachycardia, headache, laryngeal edema, pulmonary edema, and ocular adverse reactions, including choroidal effusion, acute myopia, and acute angle closure glaucoma. Signs and symptoms may include respiratory symptoms, such as nasal congestion, cough, throat irritation, as well as chills, vomiting, and nausea. Less common signs and symptoms were wheezing, allergic rhinitis, pyrexia, chest discomfort, pruritus, hypotension and blurred vision.
When DARZALEX® dosing was interrupted in the setting of ASCT (CASSIOPEIA) for a median of 3.75 months (range: 2.4 to 6.9 months), upon re-initiation of DARZALEX®, the incidence of infusion-related reactions was 11% for the first infusion following ASCT. Infusion-related reactions occurring at re-initiation of DARZALEX® following ASCT were consistent in terms of symptoms and severity (Grade 3 or 4: <1%) with those reported in previous studies at Week 2 or subsequent infusions. In EQUULEUS, patients receiving combination treatment (n=97) were administered the first 16 mg/kg dose at Week 1 split over two days, ie, 8 mg/kg on Day 1 and Day 2, respectively. The incidence of any grade infusion-related reactions was 42%, with 36% of patients experiencing infusion-related reactions on Day 1 of Week 1, 4% on Day 2 of Week 1, and 8% with subsequent infusions.
Pre-medicate patients with antihistamines, antipyretics, and corticosteroids. Frequently monitor patients during the entire infusion. Interrupt DARZALEX® infusion for reactions of any severity and institute medical management as needed. Permanently discontinue DARZALEX® therapy if an anaphylactic reaction or life-threatening (Grade 4) reaction occurs and institute appropriate emergency care. For patients with Grade 1, 2, or 3 reactions, reduce the infusion rate when re-starting the infusion.
To reduce the risk of delayed infusion-related reactions, administer oral corticosteroids to all patients following DARZALEX® infusions. Patients with a history of chronic obstructive pulmonary disease may require additional post-infusion medications to manage respiratory complications. Consider prescribing short- and long-acting bronchodilators and inhaled corticosteroids for patients with chronic obstructive pulmonary disease.
Ocular adverse reactions, including acute myopia and narrowing of the anterior chamber angle due to ciliochoroidal effusions with potential for increased intraocular pressure or glaucoma, have occurred with DARZALEX infusion. If ocular symptoms occur, interrupt DARZALEX infusion and seek immediate ophthalmologic evaluation prior to restarting DARZALEX.
Interference With Serological Testing
Daratumumab binds to CD38 on red blood cells (RBCs) and results in a positive indirect antiglobulin test (indirect Coombs test). Daratumumab-mediated positive indirect antiglobulin test may persist for up to 6 months after the last daratumumab infusion. Daratumumab bound to RBCs masks detection of antibodies to minor antigens in the patient’s serum. The determination of a patient’s ABO and Rh blood type is not impacted. Notify blood transfusion centers of this interference with serological testing and inform blood banks that a patient has received DARZALEX®. Type and screen patients prior to starting DARZALEX®.
Neutropenia and Thrombocytopenia
DARZALEX® may increase neutropenia and thrombocytopenia induced by background therapy. Monitor complete blood cell counts periodically during treatment according to manufacturer’s prescribing information for background therapies. Monitor patients with neutropenia for signs of infection. Consider withholding DARZALEX® until recovery of neutrophils or for recovery of platelets.
Interference With Determination of Complete Response
Daratumumab is a human immunoglobulin G (IgG) kappa monoclonal antibody that can be detected on both the serum protein electrophoresis (SPE) and immunofixation (IFE) assays used for the clinical monitoring of endogenous M-protein. This interference can impact the determination of complete response and of disease progression in some patients with IgG kappa myeloma protein.
Embryo-Fetal Toxicity
Based on the mechanism of action, DARZALEX® can cause fetal harm when administered to a pregnant woman. DARZALEX® may cause depletion of fetal immune cells and decreased bone density. Advise pregnant women of the potential risk to a fetus. Advise females with reproductive potential to use effective contraception during treatment with DARZALEX® and for 3 months after the last dose.
The combination of DARZALEX® with lenalidomide, pomalidomide, or thalidomide is contraindicated in pregnant women because lenalidomide, pomalidomide, and thalidomide may cause birth defects and death of the unborn child. Refer to the lenalidomide, pomalidomide, or thalidomide prescribing information on use during pregnancy.
ADVERSE REACTIONS
The most frequently reported adverse reactions (incidence ≥20%) were: upper respiratory infection, neutropenia, infusion‑related reactions, thrombocytopenia, diarrhea, constipation, anemia, peripheral sensory neuropathy, fatigue, peripheral edema, nausea, cough, pyrexia, dyspnea, and asthenia. The most common hematologic laboratory abnormalities (≥40%) with DARZALEX® are: neutropenia, lymphopenia, thrombocytopenia, leukopenia, and anemia.
INDICATIONS
DARZALEX® (daratumumab) is indicated for the treatment of adult patients with multiple myeloma:
- In combination with lenalidomide and dexamethasone in newly diagnosed patients who are ineligible for autologous stem cell transplant and in patients with relapsed or refractory multiple myeloma who have received at least one prior therapy
- In combination with bortezomib, melphalan, and prednisone in newly diagnosed patients who are ineligible for autologous stem cell transplant
- In combination with bortezomib, thalidomide, and dexamethasone in newly diagnosed patients who are eligible for autologous stem cell transplant
- In combination with bortezomib and dexamethasone in patients who have received at least one prior therapy
- In combination with carfilzomib and dexamethasone in patients with relapsed or refractory multiple myeloma who have received one to three prior lines of therapy
- In combination with pomalidomide and dexamethasone in patients who have received at least two prior therapies including lenalidomide and a proteasome inhibitor
- As monotherapy in patients who have received at least three prior lines of therapy including a proteasome inhibitor (PI) and an immunomodulatory agent or who are double-refractory to a PI and an immunomodulatory agent
Please click here to see the full Prescribing Information.
1. Richardson PG, San Miguel JF, Moreau P, et al. Interpreting clinical trial data in multiple myeloma: translating findings to the real-world setting. Blood Cancer J. 2018;8(11). doi:10.1038/s41408-018-0141-0
2. Key Statistics About Multiple Myeloma. Cancer.org. Published 2019. https://www.cancer.org/cancer/multiple-myeloma/about/key-statistics.html
3. Fonseca R, Usmani SZ, Mehra M, et al. Frontline treatment patterns and attrition rates by subsequent lines of therapy in patients with newly diagnosed multiple myeloma. BMC Cancer. 2020;20(1). doi:10.1186/s12885-020-07503-y
4. Devarakonda S, Efebera Y, Sharma N. Role of Stem Cell Transplantation in Multiple Myeloma. Cancers. 2021;13(4):863. doi:10.3390/cancers13040863
5. Derudas D, Capraro F, Martinelli G, Cerchione C. How I manage frontline transplant-ineligible multiple myeloma. Hematol Rep. 2020;12(s1). doi:10.4081/hr.2020.8956
6. Kazandjian D. Multiple myeloma epidemiology and survival: A unique malignancy. Semin Oncl. 2016;43(6):676-681. doi:10.1053/j.seminoncol.2016.11.004
7. Facon T, Kumar S, Plesner T, et al. Daratumumab plus lenalidomide and dexamethasone for untreated myeloma. N. Engl. J. Med. 2019;380(22):2104-2115. doi:10.1056/nejmoa1817249
8. DARZALEX® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
9. Facon T, Kumar SK, Plesner T, et al. Daratumumab, lenalidomide, and dexamethasone versus lenalidomide and dexamethasone alone in newly diagnosed multiple myeloma (MAIA): overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(11):1582-1596. doi:10.1016/s1470-2045(21)00466-6
10. Facon T, Dimopoulos MA, Meuleman N, et al. A simplified frailty scale predicts outcomes in transplant-ineligible patients with newly diagnosed multiple myeloma treated in the FIRST (MM-020) trial. Leukemia. 2019;34(1):224-233. doi:10.1038/s41375-019-0539-0
11. Facon T, Kumar SK, Plesner T, et al. Supplement to: Daratumumab, lenalidomide, and dexamethasone versus lenalidomide and dexamethasone alone in newly diagnosed multiple myeloma (MAIA): overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(11):1582-1596.
12. Facon T, Cook G, Usmani SZ, et al. Daratumumab plus lenalidomide and dexamethasone in transplant-ineligible newly diagnosed multiple myeloma: frailty subgroup analysis of MAIA. Leukemia. 2022;36(4):1066-1077. doi:10.1038/s41375-021-01488-8
© Janssen Biotech, Inc. 2022 All rights reserved. 12/22 cp-333446v1
1. The treatment of multiple myeloma has evolved significantly in recent years. What are some of the most important things you consider in the treatment of your newly diagnosed, transplant-ineligible patients?
We’ve seen great progress in the treatment of multiple myeloma over the last decade, and outcomes continue to improve for many patients.1 Still, it is important to keep in mind that more than 34,000 patients will be diagnosed and more than 12,000 people will die from the disease this year.2 We may have the greatest opportunity to impact the course of disease in the treatment of newly diagnosed patients due to the nature of this cancer:
- Multiple myeloma is characterized by relapse, and we know the length of remission generally decreases with each relapse and subsequent line of therapy.3
- Patients often become refractory to treatment over time.
When I meet with a patient who has been diagnosed with multiple myeloma, the first thing I consider is their eligibility for autologous stem cell transplant (ASCT). In my opinion, the introduction of ASCT is one of the biggest advancements in the last few decades, and we’ve found that ASCT followed by maintenance therapy with targeted tools improves progression-free survival (PFS).4
Unfortunately, many newly diagnosed patients are not eligible for ASCT–either because of comorbidities or other complexities related to the presentation of their disease.
For patients who are transplant-ineligible (TIE), it is important to have treatment options that are proven effective in extending PFS and overall survival (OS), and capable of producing deep and durable responses.
2. What are the challenges associated with treating newly diagnosed patients who are not eligible for ASCT?
We still consider multiple myeloma to be an incurable disease but, in my opinion, the treatment of TIE patients is less challenging today than a decade ago due to the emergence of novel therapies. That said, TIE patients are typically older and present with more advanced disease and comorbidities, including diabetes or cardiovascular events.5
A retrospective analysis published in 2020 by Rafael Fonseca examined frontline treatment patterns and attrition rates by line of therapy among newly diagnosed multiple myeloma (NDMM) patients who are TIE. More than 22,000 patients were identified from three patient-level databases between 2000 and 2018 - the OPTUM Commercial Claims database, the OPTUM Electronic Medical Records database, and the Surveillance, Epidemiology, and End Results-Meidcare Linked database. Patients included had to have a multiple myeloma diagnosis on or after January 1, 2007. Results showed that attrition rates among newly diagnosed, TIE patients with multiple myeloma increase with each line of therapy, with the proportion of patients who receive a second line of therapy decreasing by 50 percent with each subsequent line.3
3. Can you provide more detail on the goals of therapy for newly diagnosed, transplant-ineligible patients?
When I discuss treatment goals with TIE patients, I feel it is important to emphasize managing side effects and achieving deep and durable responses. I have the benefit of being in an academic setting, where I regularly exchange information with my colleagues about what we’re learning from the clinical studies in which we participate. Choosing which treatment to administer is complex and involves other considerations. For example, if two regimens have comparable efficacy, I may recommend the regimen with a more established safety profile or more robust evidence so I can properly anticipate and manage toxicities in my patients. Overall survival is one of the most important endpoints I consider, in addition to depth of response and PFS. In recent years, we’ve seen increasing evidence pointing to the importance of using a proven effective treatment in frontline patients that are ineligible for transplant.
4. A key study in newly-diagnosed, transplant-ineligible multiple myeloma is the Phase 3 MAIA study. Can you share the key takeaways from this study and discuss how the results have shaped treatment for this patient population?
Of course. The MAIA study is a randomized Phase 3 study evaluating DARZALEX® (daratumumab) intravenous injection in combination with lenalidomide and dexamethasone (D-Rd) compared with Rd in 737 adult patients with newly diagnosed, transplant-ineligible multiple myeloma. The median age of patients participating in the MAIA study was 73 (range 45-90), an important consideration since the median age for multiple myeloma diagnosis is approximately 66-70 years of age.6 The study evaluated PFS as the primary endpoint, and overall survival as a key secondary endpoint, and supported the FDA approval of DARZALEX® in combination with lenalidomide and dexamethasone for adult patients with newly diagnosed, multiple myeloma who are ineligible for ASCT.
MAIA study design7
The baseline demographic and disease characteristics were similar between the 2 treatment groups. Forty-four percent of the patients were ≥75 years of age. Fifty-two percent (52%) of patients were male, 92% White, 4% Black or African American, and 1% Asian. Three percent (3%) of patients reported an ethnicity of Hispanic or Latino. Thirty-four (34%) had an Eastern Cooperative Oncology Group (ECOG) performance score of 0, 50% had an ECOG performance score of 1, and 17% had an ECOG performance score of ≥2. Twenty-seven percent had International Staging System (ISS) Stage I, 43% had ISS Stage II, and 29% had ISS Stage III disease.
Select Important Safety Information:
CONTRAINDICATIONS
DARZALEX® is contraindicated in patients with a history of severe hypersensitivity (eg, anaphylactic reactions) to daratumumab or any of the components of the formulation.
WARNINGS AND PRECAUTIONS
Infusion-Related Reactions: DARZALEX® can cause severe and/or serious infusion-related reactions including anaphylactic reactions.
These reactions can be life-threatening, and fatal outcomes have been reported. Please scroll down to read Important Safety Information for DARZALEX®.
Primary findings from the study, which were published in 2019, showed an improvement in PFS in patients receiving D-Rd compared with those receiving Rd alone.7 The median PFS was not reached in the D-Rd arm and was reached at 31.9 months in the Rd arm (HR 0.56; 95% CI 0.43-0.73; P<0.0001).7 At a median of 30 months of follow-up, the data showed the clinical benefit of D-Rd therapy, with a 44% reduction in the risk of disease progression or death in patients receiving D-Rd compared with Rd alone.7
Additionally, 70.6% of patients (95% CI, 65.0-75.4) had no progressive disease with D-Rd treatment at median 30 months of follow-up, compared with 55.6% (95% CI, 49.5-61.3) of patients in the Rd group.7
In terms of depth of response, the percentage of patients with a complete response or better was 47.6% in patients receiving D-Rd compared with 24.9% in the Rd group.7
Overall response rate with D-Rd in TIE NDMM at ~30 months of follow-up8
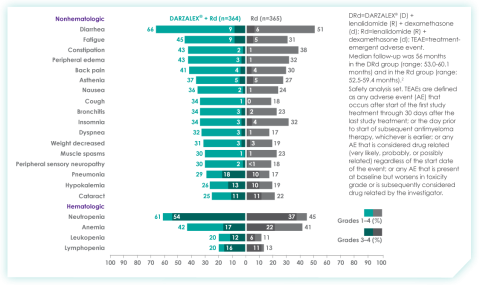
An overview of the most frequent adverse events at 30-months of follow-up are provided below. The most frequent adverse reactions were reported in ≥20% of patients, with at least a 5% greater frequency in the D-Rd arm compared with Rd alone.8
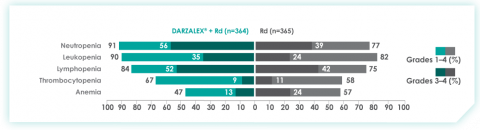
Most frequent adverse events at ~30 months of follow-up with D-Rd in TIE NDMM8
Most frequent hematologic laboratory abnormalities with D-Rd in TIE NDMM at ~30 months8
Serious adverse reactions with a 2% greater incidence in the D-Rd arm compared with the Rd arm were pneumonia (D-Rd 15% vs Rd 8%), bronchitis (D-Rd 4% vs Rd 2%), and dehydration (D-Rd 2% vs Rd <1).
• Discontinuation rates due to any adverse event: 7% with D-Rd vs 16% with Rd
• Infusion-related reactions (IRRs) with D-Rd occurred in 41% of patients; 2% were Grade 3 and <1% were Grade 4
• IRRs of any grade or severity may require management by interruption, modification, and/or discontinuation of the infusion
• Most IRRs occurred during first infusion
5. Thanks for that overview. In addition to these results, The Lancet Oncology has published updated overall survival data from a 5-year follow-up on the MAIA study. Can you provide an overview of these data and insights on their potential for patients?
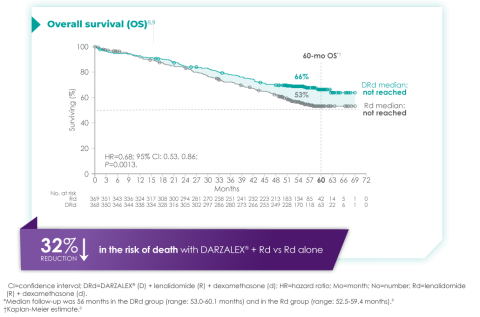
The MAIA trial was an important study, and for me, the results were practice changing. We see that after a median of nearly 5 years of follow-up, D-Rd significantly improved OS in TIE NDMM patients who were treated to progression compared with Rd alone (66.3% vs. 53.1% [HR=0.68; 95% CI, 0.53-0.86; P=0.0013]).9 This equates to approximately a 32% reduction in death when DARZALEX® was added to a two-drug regimen, which is a meaningful consideration when selecting the most appropriate regimens for my newly diagnosed, transplant-ineligible patients.9
Overall survival data at ~5 years with D-Rd compared to Rd alone in TIE NDMM9
Importantly, efficacy that resulted from longer treatment with D-Rd is also supported by approximately 5 years of safety evaluation. Below is information from a follow-up analysis of the MAIA study. This information is not included in the current Prescribing Information and has not been evaluated by the FDA. Treatment-emergent adverse events are reported as observed. These analyses have not been adjusted for multiple comparisons and no conclusions should be drawn. In what I’ve observed through published data and in my practice, longer treatment has not revealed new safety signals.
Most frequent treatment-emergent adverse events (any grade reported in ≥30% of patients and/or Grade 3/4 reported in ≥10% of patients) at ~5 years9
Select Important Safety Information:
DARZALEX® can cause severe and/or serious infusion-related reactions including anaphylactic reactions. These reactions can be life threatening, and fatal outcomes have been reported. In clinical trials (monotherapy and combination: N=2066), infusion-related reactions occurred in 37% of patients with the Week 1 (16 mg/kg) infusion, 2% with the Week 2 infusion, and cumulatively 6% with subsequent infusions. Less than 1% of patients had a Grade 3/4 infusion-related reaction at Week 2 or subsequent infusions. The median time to onset was 1.5 hours (range: 0 to 73 hours). Nearly all reactions occurred during infusion or within 4 hours of completing DARZALEX®. Severe reactions have occurred, including bronchospasm, hypoxia, dyspnea, hypertension, tachycardia, headache, laryngeal edema, pulmonary edema, and ocular adverse reactions, including choroidal effusion, acute myopia, and acute angle closure glaucoma. Signs and symptoms may include respiratory symptoms, such as nasal congestion, cough, throat irritation, as well as chills, vomiting, and nausea. Less common signs and symptoms were wheezing, allergic rhinitis, pyrexia, chest discomfort, pruritus, hypotension and blurred vision. Please scroll down to see Important Safety Information for DARZALEX®.
6. Does the availability of OS data influence your decisions on treatment selection in TIE NDMM?
Overall survival absolutely remains the gold standard and informs my practice. Prior to OS data being available, I will often look at other efficacy endpoints that are available sooner. In MAIA, I was encouraged by efficacy endpoints in earlier data, which were later confirmed by the latest data on OS.
7. The MAIA study shows that treating to disease progression or unacceptable toxicity is important. How does that impact your approach to treatment?
It's important to keep in mind that the MAIA trial was designed to evaluate treatment until progression or unacceptable toxicity. The results revealed a significant difference between the DR-d and Rd treatment arms, but results observed in this study are contingent on this treatment approach. From a clinical perspective, unless there is considerable toxicity, I advocate for treating with D-Rd to progression.
In the clinic, we also see that TIE patients who have higher frailty scores are more likely to discontinue treatment prior to progression.10 There can be other reasons too – such as a patient simply wanting to have a break from treatment. These conversations are not always easy, but it is important to have an honest dialogue with patients.
8. What can we learn from studies like the MAIA trial that included a wide range of patient populations including patients who are elderly, frail, or had high cytogenetic risk?
Several patient subgroups were analyzed as part of the MAIA study. It is important to note that these subgroup analyses are not included in the Prescribing Information for DARZALEX®. These analyses were not adjusted for multiple comparisons, and there are insufficient numbers of patients per subgroup to make definitive conclusions of efficacy among the subgroups.
As mentioned above, the MAIA study evaluated a wide range of patients (n=737). The baseline demographic and disease characteristics were similar between the D-Rd and Rd treatment groups and the median age was 73 (range: 45-90) years, with 44% of the patients ≥75 years of age.
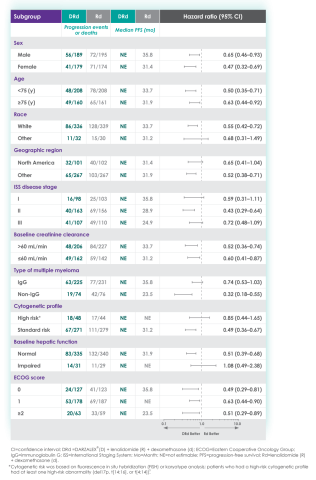
In the various patient subgroups that were analyzed as part of the MAIA study, it was found that at ~3-years of follow-up the PFS numerically favored DRd compared with Rd alone in most subgroups (see table below).
Median progression-free survival by sub-population at ~3 year follow-up8
The MAIA trial also included patients who were frail and a post hoc analysis was conducted in this subgroup of patients. These analyses are not included in the Prescribing Information for DARZALEX®. These analyses were conducted post hoc and there are insufficient numbers of patients per subgroup to make definitive conclusions of efficacy among the subgroups.
A frailty assessment was performed retrospectively using age, the Charlson Comorbidity Index (CCI) – which is calculated based on a retrospective review of the patient’s medical history to predict the 10-year mortality – and the baseline Eastern Cooperative Oncology Group (ECOG) performance status score, used to measure a patient’s level of functioning in terms of their ability to care for themselves, daily activity, and physical activity. The frailty scores were then added up to classify patients into fit (0), intermediate (1), or frail (≥2). Frailty status was further simplified into 2 categories: non-frail (0-1) and frail (≥2). The median age in the frail subgroup was 77 years (range: 57-80 years), with 88% of patients having ECOG performance score ≥1. CCI was calculated based on retrospective review of each patient’s medical history.12
The charts below illustrate the frailty scoring system with an overview of the patient population included in the 3-year post hoc analysis, PFS rate, and adverse events.
MAIA post hoc subgroup analysis by frailty status score12
The retrospective assessment of frailty score was a limitation of this study. Retrospective CCI calculations were based on reported medical history, which may contain missing data and result in underestimating or overestimating the number of patients in each frailty subgroup. The ECOG PS score parameter used for frailty score calculations in the study is more subjective, with susceptibility to intra- and inter-observer bias, compared with the ADL (activities of daily living) and IADL (instrumental activities of daily living) scales used in the IMWG scoring system. While the frailty scale used in the study is based on parameters that are routinely assessed in clinical practice for clinical use, the use of comprehensive frailty assessments that more accurately reflect biological or functional frailty will remain important for the further optimization of treatment strategies for frail patients. Patients with an ECOG PS score ≥3 and patients with comorbidities that may interfere with the study procedures were excluded from MAIA; the inclusion and exclusion criteria for the study limits the generalizability of these results to more frail patients seen in clinical practice.
Progression-free survival in a ~3-year subgroup analysis of frail patients following treatment with D-Rd in TIE NDMM12
Most frequent Grade 3/4 treatment-emergent adverse events (≥10%) in frail patients at ~3 year follow-up of MAIA trial12
Please see additional Important Safety Information for DARZALEX® below.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
DARZALEX® is contraindicated in patients with a history of severe hypersensitivity (eg, anaphylactic reactions) to daratumumab or any of the components of the formulation.
WARNINGS AND PRECAUTIONS
Infusion-Related Reactions
DARZALEX® can cause severe and/or serious infusion-related reactions including anaphylactic reactions. These reactions can be life‑threatening, and fatal outcomes have been reported. In clinical trials (monotherapy and combination: N=2066), infusion-related reactions occurred in 37% of patients with the Week 1 (16 mg/kg) infusion, 2% with the Week 2 infusion, and cumulatively 6% with subsequent infusions. Less than 1% of patients had a Grade 3/4 infusion-related reaction at Week 2 or subsequent infusions. The median time to onset was 1.5 hours (range: 0 to 73 hours). Nearly all reactions occurred during infusion or within 4 hours of completing DARZALEX®. Severe reactions have occurred, including bronchospasm, hypoxia, dyspnea, hypertension, tachycardia, headache, laryngeal edema, pulmonary edema, and ocular adverse reactions, including choroidal effusion, acute myopia, and acute angle closure glaucoma. Signs and symptoms may include respiratory symptoms, such as nasal congestion, cough, throat irritation, as well as chills, vomiting, and nausea. Less common signs and symptoms were wheezing, allergic rhinitis, pyrexia, chest discomfort, pruritus, hypotension and blurred vision.
When DARZALEX® dosing was interrupted in the setting of ASCT (CASSIOPEIA) for a median of 3.75 months (range: 2.4 to 6.9 months), upon re-initiation of DARZALEX®, the incidence of infusion-related reactions was 11% for the first infusion following ASCT. Infusion-related reactions occurring at re-initiation of DARZALEX® following ASCT were consistent in terms of symptoms and severity (Grade 3 or 4: <1%) with those reported in previous studies at Week 2 or subsequent infusions. In EQUULEUS, patients receiving combination treatment (n=97) were administered the first 16 mg/kg dose at Week 1 split over two days, ie, 8 mg/kg on Day 1 and Day 2, respectively. The incidence of any grade infusion-related reactions was 42%, with 36% of patients experiencing infusion-related reactions on Day 1 of Week 1, 4% on Day 2 of Week 1, and 8% with subsequent infusions.
Pre-medicate patients with antihistamines, antipyretics, and corticosteroids. Frequently monitor patients during the entire infusion. Interrupt DARZALEX® infusion for reactions of any severity and institute medical management as needed. Permanently discontinue DARZALEX® therapy if an anaphylactic reaction or life-threatening (Grade 4) reaction occurs and institute appropriate emergency care. For patients with Grade 1, 2, or 3 reactions, reduce the infusion rate when re-starting the infusion.
To reduce the risk of delayed infusion-related reactions, administer oral corticosteroids to all patients following DARZALEX® infusions. Patients with a history of chronic obstructive pulmonary disease may require additional post-infusion medications to manage respiratory complications. Consider prescribing short- and long-acting bronchodilators and inhaled corticosteroids for patients with chronic obstructive pulmonary disease.
Ocular adverse reactions, including acute myopia and narrowing of the anterior chamber angle due to ciliochoroidal effusions with potential for increased intraocular pressure or glaucoma, have occurred with DARZALEX infusion. If ocular symptoms occur, interrupt DARZALEX infusion and seek immediate ophthalmologic evaluation prior to restarting DARZALEX.
Interference With Serological Testing
Daratumumab binds to CD38 on red blood cells (RBCs) and results in a positive indirect antiglobulin test (indirect Coombs test). Daratumumab-mediated positive indirect antiglobulin test may persist for up to 6 months after the last daratumumab infusion. Daratumumab bound to RBCs masks detection of antibodies to minor antigens in the patient’s serum. The determination of a patient’s ABO and Rh blood type is not impacted. Notify blood transfusion centers of this interference with serological testing and inform blood banks that a patient has received DARZALEX®. Type and screen patients prior to starting DARZALEX®.
Neutropenia and Thrombocytopenia
DARZALEX® may increase neutropenia and thrombocytopenia induced by background therapy. Monitor complete blood cell counts periodically during treatment according to manufacturer’s prescribing information for background therapies. Monitor patients with neutropenia for signs of infection. Consider withholding DARZALEX® until recovery of neutrophils or for recovery of platelets.
Interference With Determination of Complete Response
Daratumumab is a human immunoglobulin G (IgG) kappa monoclonal antibody that can be detected on both the serum protein electrophoresis (SPE) and immunofixation (IFE) assays used for the clinical monitoring of endogenous M-protein. This interference can impact the determination of complete response and of disease progression in some patients with IgG kappa myeloma protein.
Embryo-Fetal Toxicity
Based on the mechanism of action, DARZALEX® can cause fetal harm when administered to a pregnant woman. DARZALEX® may cause depletion of fetal immune cells and decreased bone density. Advise pregnant women of the potential risk to a fetus. Advise females with reproductive potential to use effective contraception during treatment with DARZALEX® and for 3 months after the last dose.
The combination of DARZALEX® with lenalidomide, pomalidomide, or thalidomide is contraindicated in pregnant women because lenalidomide, pomalidomide, and thalidomide may cause birth defects and death of the unborn child. Refer to the lenalidomide, pomalidomide, or thalidomide prescribing information on use during pregnancy.
ADVERSE REACTIONS
The most frequently reported adverse reactions (incidence ≥20%) were: upper respiratory infection, neutropenia, infusion‑related reactions, thrombocytopenia, diarrhea, constipation, anemia, peripheral sensory neuropathy, fatigue, peripheral edema, nausea, cough, pyrexia, dyspnea, and asthenia. The most common hematologic laboratory abnormalities (≥40%) with DARZALEX® are: neutropenia, lymphopenia, thrombocytopenia, leukopenia, and anemia.
INDICATIONS
DARZALEX® (daratumumab) is indicated for the treatment of adult patients with multiple myeloma:
- In combination with lenalidomide and dexamethasone in newly diagnosed patients who are ineligible for autologous stem cell transplant and in patients with relapsed or refractory multiple myeloma who have received at least one prior therapy
- In combination with bortezomib, melphalan, and prednisone in newly diagnosed patients who are ineligible for autologous stem cell transplant
- In combination with bortezomib, thalidomide, and dexamethasone in newly diagnosed patients who are eligible for autologous stem cell transplant
- In combination with bortezomib and dexamethasone in patients who have received at least one prior therapy
- In combination with carfilzomib and dexamethasone in patients with relapsed or refractory multiple myeloma who have received one to three prior lines of therapy
- In combination with pomalidomide and dexamethasone in patients who have received at least two prior therapies including lenalidomide and a proteasome inhibitor
- As monotherapy in patients who have received at least three prior lines of therapy including a proteasome inhibitor (PI) and an immunomodulatory agent or who are double-refractory to a PI and an immunomodulatory agent
Please click here to see the full Prescribing Information.
1. Richardson PG, San Miguel JF, Moreau P, et al. Interpreting clinical trial data in multiple myeloma: translating findings to the real-world setting. Blood Cancer J. 2018;8(11). doi:10.1038/s41408-018-0141-0
2. Key Statistics About Multiple Myeloma. Cancer.org. Published 2019. https://www.cancer.org/cancer/multiple-myeloma/about/key-statistics.html
3. Fonseca R, Usmani SZ, Mehra M, et al. Frontline treatment patterns and attrition rates by subsequent lines of therapy in patients with newly diagnosed multiple myeloma. BMC Cancer. 2020;20(1). doi:10.1186/s12885-020-07503-y
4. Devarakonda S, Efebera Y, Sharma N. Role of Stem Cell Transplantation in Multiple Myeloma. Cancers. 2021;13(4):863. doi:10.3390/cancers13040863
5. Derudas D, Capraro F, Martinelli G, Cerchione C. How I manage frontline transplant-ineligible multiple myeloma. Hematol Rep. 2020;12(s1). doi:10.4081/hr.2020.8956
6. Kazandjian D. Multiple myeloma epidemiology and survival: A unique malignancy. Semin Oncl. 2016;43(6):676-681. doi:10.1053/j.seminoncol.2016.11.004
7. Facon T, Kumar S, Plesner T, et al. Daratumumab plus lenalidomide and dexamethasone for untreated myeloma. N. Engl. J. Med. 2019;380(22):2104-2115. doi:10.1056/nejmoa1817249
8. DARZALEX® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
9. Facon T, Kumar SK, Plesner T, et al. Daratumumab, lenalidomide, and dexamethasone versus lenalidomide and dexamethasone alone in newly diagnosed multiple myeloma (MAIA): overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(11):1582-1596. doi:10.1016/s1470-2045(21)00466-6
10. Facon T, Dimopoulos MA, Meuleman N, et al. A simplified frailty scale predicts outcomes in transplant-ineligible patients with newly diagnosed multiple myeloma treated in the FIRST (MM-020) trial. Leukemia. 2019;34(1):224-233. doi:10.1038/s41375-019-0539-0
11. Facon T, Kumar SK, Plesner T, et al. Supplement to: Daratumumab, lenalidomide, and dexamethasone versus lenalidomide and dexamethasone alone in newly diagnosed multiple myeloma (MAIA): overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(11):1582-1596.
12. Facon T, Cook G, Usmani SZ, et al. Daratumumab plus lenalidomide and dexamethasone in transplant-ineligible newly diagnosed multiple myeloma: frailty subgroup analysis of MAIA. Leukemia. 2022;36(4):1066-1077. doi:10.1038/s41375-021-01488-8
© Janssen Biotech, Inc. 2022 All rights reserved. 12/22 cp-333446v1
1. The treatment of multiple myeloma has evolved significantly in recent years. What are some of the most important things you consider in the treatment of your newly diagnosed, transplant-ineligible patients?
We’ve seen great progress in the treatment of multiple myeloma over the last decade, and outcomes continue to improve for many patients.1 Still, it is important to keep in mind that more than 34,000 patients will be diagnosed and more than 12,000 people will die from the disease this year.2 We may have the greatest opportunity to impact the course of disease in the treatment of newly diagnosed patients due to the nature of this cancer:
- Multiple myeloma is characterized by relapse, and we know the length of remission generally decreases with each relapse and subsequent line of therapy.3
- Patients often become refractory to treatment over time.
When I meet with a patient who has been diagnosed with multiple myeloma, the first thing I consider is their eligibility for autologous stem cell transplant (ASCT). In my opinion, the introduction of ASCT is one of the biggest advancements in the last few decades, and we’ve found that ASCT followed by maintenance therapy with targeted tools improves progression-free survival (PFS).4
Unfortunately, many newly diagnosed patients are not eligible for ASCT–either because of comorbidities or other complexities related to the presentation of their disease.
For patients who are transplant-ineligible (TIE), it is important to have treatment options that are proven effective in extending PFS and overall survival (OS), and capable of producing deep and durable responses.
2. What are the challenges associated with treating newly diagnosed patients who are not eligible for ASCT?
We still consider multiple myeloma to be an incurable disease but, in my opinion, the treatment of TIE patients is less challenging today than a decade ago due to the emergence of novel therapies. That said, TIE patients are typically older and present with more advanced disease and comorbidities, including diabetes or cardiovascular events.5
A retrospective analysis published in 2020 by Rafael Fonseca examined frontline treatment patterns and attrition rates by line of therapy among newly diagnosed multiple myeloma (NDMM) patients who are TIE. More than 22,000 patients were identified from three patient-level databases between 2000 and 2018 - the OPTUM Commercial Claims database, the OPTUM Electronic Medical Records database, and the Surveillance, Epidemiology, and End Results-Meidcare Linked database. Patients included had to have a multiple myeloma diagnosis on or after January 1, 2007. Results showed that attrition rates among newly diagnosed, TIE patients with multiple myeloma increase with each line of therapy, with the proportion of patients who receive a second line of therapy decreasing by 50 percent with each subsequent line.3
3. Can you provide more detail on the goals of therapy for newly diagnosed, transplant-ineligible patients?
When I discuss treatment goals with TIE patients, I feel it is important to emphasize managing side effects and achieving deep and durable responses. I have the benefit of being in an academic setting, where I regularly exchange information with my colleagues about what we’re learning from the clinical studies in which we participate. Choosing which treatment to administer is complex and involves other considerations. For example, if two regimens have comparable efficacy, I may recommend the regimen with a more established safety profile or more robust evidence so I can properly anticipate and manage toxicities in my patients. Overall survival is one of the most important endpoints I consider, in addition to depth of response and PFS. In recent years, we’ve seen increasing evidence pointing to the importance of using a proven effective treatment in frontline patients that are ineligible for transplant.
4. A key study in newly-diagnosed, transplant-ineligible multiple myeloma is the Phase 3 MAIA study. Can you share the key takeaways from this study and discuss how the results have shaped treatment for this patient population?
Of course. The MAIA study is a randomized Phase 3 study evaluating DARZALEX® (daratumumab) intravenous injection in combination with lenalidomide and dexamethasone (D-Rd) compared with Rd in 737 adult patients with newly diagnosed, transplant-ineligible multiple myeloma. The median age of patients participating in the MAIA study was 73 (range 45-90), an important consideration since the median age for multiple myeloma diagnosis is approximately 66-70 years of age.6 The study evaluated PFS as the primary endpoint, and overall survival as a key secondary endpoint, and supported the FDA approval of DARZALEX® in combination with lenalidomide and dexamethasone for adult patients with newly diagnosed, multiple myeloma who are ineligible for ASCT.
MAIA study design7
The baseline demographic and disease characteristics were similar between the 2 treatment groups. Forty-four percent of the patients were ≥75 years of age. Fifty-two percent (52%) of patients were male, 92% White, 4% Black or African American, and 1% Asian. Three percent (3%) of patients reported an ethnicity of Hispanic or Latino. Thirty-four (34%) had an Eastern Cooperative Oncology Group (ECOG) performance score of 0, 50% had an ECOG performance score of 1, and 17% had an ECOG performance score of ≥2. Twenty-seven percent had International Staging System (ISS) Stage I, 43% had ISS Stage II, and 29% had ISS Stage III disease.
Select Important Safety Information:
CONTRAINDICATIONS
DARZALEX® is contraindicated in patients with a history of severe hypersensitivity (eg, anaphylactic reactions) to daratumumab or any of the components of the formulation.
WARNINGS AND PRECAUTIONS
Infusion-Related Reactions: DARZALEX® can cause severe and/or serious infusion-related reactions including anaphylactic reactions.
These reactions can be life-threatening, and fatal outcomes have been reported. Please scroll down to read Important Safety Information for DARZALEX®.
Primary findings from the study, which were published in 2019, showed an improvement in PFS in patients receiving D-Rd compared with those receiving Rd alone.7 The median PFS was not reached in the D-Rd arm and was reached at 31.9 months in the Rd arm (HR 0.56; 95% CI 0.43-0.73; P<0.0001).7 At a median of 30 months of follow-up, the data showed the clinical benefit of D-Rd therapy, with a 44% reduction in the risk of disease progression or death in patients receiving D-Rd compared with Rd alone.7
Additionally, 70.6% of patients (95% CI, 65.0-75.4) had no progressive disease with D-Rd treatment at median 30 months of follow-up, compared with 55.6% (95% CI, 49.5-61.3) of patients in the Rd group.7
In terms of depth of response, the percentage of patients with a complete response or better was 47.6% in patients receiving D-Rd compared with 24.9% in the Rd group.7
Overall response rate with D-Rd in TIE NDMM at ~30 months of follow-up8
An overview of the most frequent adverse events at 30-months of follow-up are provided below. The most frequent adverse reactions were reported in ≥20% of patients, with at least a 5% greater frequency in the D-Rd arm compared with Rd alone.8
Most frequent adverse events at ~30 months of follow-up with D-Rd in TIE NDMM8
Most frequent hematologic laboratory abnormalities with D-Rd in TIE NDMM at ~30 months8
Serious adverse reactions with a 2% greater incidence in the D-Rd arm compared with the Rd arm were pneumonia (D-Rd 15% vs Rd 8%), bronchitis (D-Rd 4% vs Rd 2%), and dehydration (D-Rd 2% vs Rd <1).
• Discontinuation rates due to any adverse event: 7% with D-Rd vs 16% with Rd
• Infusion-related reactions (IRRs) with D-Rd occurred in 41% of patients; 2% were Grade 3 and <1% were Grade 4
• IRRs of any grade or severity may require management by interruption, modification, and/or discontinuation of the infusion
• Most IRRs occurred during first infusion
5. Thanks for that overview. In addition to these results, The Lancet Oncology has published updated overall survival data from a 5-year follow-up on the MAIA study. Can you provide an overview of these data and insights on their potential for patients?
The MAIA trial was an important study, and for me, the results were practice changing. We see that after a median of nearly 5 years of follow-up, D-Rd significantly improved OS in TIE NDMM patients who were treated to progression compared with Rd alone (66.3% vs. 53.1% [HR=0.68; 95% CI, 0.53-0.86; P=0.0013]).9 This equates to approximately a 32% reduction in death when DARZALEX® was added to a two-drug regimen, which is a meaningful consideration when selecting the most appropriate regimens for my newly diagnosed, transplant-ineligible patients.9
Overall survival data at ~5 years with D-Rd compared to Rd alone in TIE NDMM9
Importantly, efficacy that resulted from longer treatment with D-Rd is also supported by approximately 5 years of safety evaluation. Below is information from a follow-up analysis of the MAIA study. This information is not included in the current Prescribing Information and has not been evaluated by the FDA. Treatment-emergent adverse events are reported as observed. These analyses have not been adjusted for multiple comparisons and no conclusions should be drawn. In what I’ve observed through published data and in my practice, longer treatment has not revealed new safety signals.
Most frequent treatment-emergent adverse events (any grade reported in ≥30% of patients and/or Grade 3/4 reported in ≥10% of patients) at ~5 years9
Select Important Safety Information:
DARZALEX® can cause severe and/or serious infusion-related reactions including anaphylactic reactions. These reactions can be life threatening, and fatal outcomes have been reported. In clinical trials (monotherapy and combination: N=2066), infusion-related reactions occurred in 37% of patients with the Week 1 (16 mg/kg) infusion, 2% with the Week 2 infusion, and cumulatively 6% with subsequent infusions. Less than 1% of patients had a Grade 3/4 infusion-related reaction at Week 2 or subsequent infusions. The median time to onset was 1.5 hours (range: 0 to 73 hours). Nearly all reactions occurred during infusion or within 4 hours of completing DARZALEX®. Severe reactions have occurred, including bronchospasm, hypoxia, dyspnea, hypertension, tachycardia, headache, laryngeal edema, pulmonary edema, and ocular adverse reactions, including choroidal effusion, acute myopia, and acute angle closure glaucoma. Signs and symptoms may include respiratory symptoms, such as nasal congestion, cough, throat irritation, as well as chills, vomiting, and nausea. Less common signs and symptoms were wheezing, allergic rhinitis, pyrexia, chest discomfort, pruritus, hypotension and blurred vision. Please scroll down to see Important Safety Information for DARZALEX®.
6. Does the availability of OS data influence your decisions on treatment selection in TIE NDMM?
Overall survival absolutely remains the gold standard and informs my practice. Prior to OS data being available, I will often look at other efficacy endpoints that are available sooner. In MAIA, I was encouraged by efficacy endpoints in earlier data, which were later confirmed by the latest data on OS.
7. The MAIA study shows that treating to disease progression or unacceptable toxicity is important. How does that impact your approach to treatment?
It's important to keep in mind that the MAIA trial was designed to evaluate treatment until progression or unacceptable toxicity. The results revealed a significant difference between the DR-d and Rd treatment arms, but results observed in this study are contingent on this treatment approach. From a clinical perspective, unless there is considerable toxicity, I advocate for treating with D-Rd to progression.
In the clinic, we also see that TIE patients who have higher frailty scores are more likely to discontinue treatment prior to progression.10 There can be other reasons too – such as a patient simply wanting to have a break from treatment. These conversations are not always easy, but it is important to have an honest dialogue with patients.
8. What can we learn from studies like the MAIA trial that included a wide range of patient populations including patients who are elderly, frail, or had high cytogenetic risk?
Several patient subgroups were analyzed as part of the MAIA study. It is important to note that these subgroup analyses are not included in the Prescribing Information for DARZALEX®. These analyses were not adjusted for multiple comparisons, and there are insufficient numbers of patients per subgroup to make definitive conclusions of efficacy among the subgroups.
As mentioned above, the MAIA study evaluated a wide range of patients (n=737). The baseline demographic and disease characteristics were similar between the D-Rd and Rd treatment groups and the median age was 73 (range: 45-90) years, with 44% of the patients ≥75 years of age.
In the various patient subgroups that were analyzed as part of the MAIA study, it was found that at ~3-years of follow-up the PFS numerically favored DRd compared with Rd alone in most subgroups (see table below).
Median progression-free survival by sub-population at ~3 year follow-up8
The MAIA trial also included patients who were frail and a post hoc analysis was conducted in this subgroup of patients. These analyses are not included in the Prescribing Information for DARZALEX®. These analyses were conducted post hoc and there are insufficient numbers of patients per subgroup to make definitive conclusions of efficacy among the subgroups.
A frailty assessment was performed retrospectively using age, the Charlson Comorbidity Index (CCI) – which is calculated based on a retrospective review of the patient’s medical history to predict the 10-year mortality – and the baseline Eastern Cooperative Oncology Group (ECOG) performance status score, used to measure a patient’s level of functioning in terms of their ability to care for themselves, daily activity, and physical activity. The frailty scores were then added up to classify patients into fit (0), intermediate (1), or frail (≥2). Frailty status was further simplified into 2 categories: non-frail (0-1) and frail (≥2). The median age in the frail subgroup was 77 years (range: 57-80 years), with 88% of patients having ECOG performance score ≥1. CCI was calculated based on retrospective review of each patient’s medical history.12
The charts below illustrate the frailty scoring system with an overview of the patient population included in the 3-year post hoc analysis, PFS rate, and adverse events.
MAIA post hoc subgroup analysis by frailty status score12
The retrospective assessment of frailty score was a limitation of this study. Retrospective CCI calculations were based on reported medical history, which may contain missing data and result in underestimating or overestimating the number of patients in each frailty subgroup. The ECOG PS score parameter used for frailty score calculations in the study is more subjective, with susceptibility to intra- and inter-observer bias, compared with the ADL (activities of daily living) and IADL (instrumental activities of daily living) scales used in the IMWG scoring system. While the frailty scale used in the study is based on parameters that are routinely assessed in clinical practice for clinical use, the use of comprehensive frailty assessments that more accurately reflect biological or functional frailty will remain important for the further optimization of treatment strategies for frail patients. Patients with an ECOG PS score ≥3 and patients with comorbidities that may interfere with the study procedures were excluded from MAIA; the inclusion and exclusion criteria for the study limits the generalizability of these results to more frail patients seen in clinical practice.
Progression-free survival in a ~3-year subgroup analysis of frail patients following treatment with D-Rd in TIE NDMM12
Most frequent Grade 3/4 treatment-emergent adverse events (≥10%) in frail patients at ~3 year follow-up of MAIA trial12
Please see additional Important Safety Information for DARZALEX® below.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
DARZALEX® is contraindicated in patients with a history of severe hypersensitivity (eg, anaphylactic reactions) to daratumumab or any of the components of the formulation.
WARNINGS AND PRECAUTIONS
Infusion-Related Reactions
DARZALEX® can cause severe and/or serious infusion-related reactions including anaphylactic reactions. These reactions can be life‑threatening, and fatal outcomes have been reported. In clinical trials (monotherapy and combination: N=2066), infusion-related reactions occurred in 37% of patients with the Week 1 (16 mg/kg) infusion, 2% with the Week 2 infusion, and cumulatively 6% with subsequent infusions. Less than 1% of patients had a Grade 3/4 infusion-related reaction at Week 2 or subsequent infusions. The median time to onset was 1.5 hours (range: 0 to 73 hours). Nearly all reactions occurred during infusion or within 4 hours of completing DARZALEX®. Severe reactions have occurred, including bronchospasm, hypoxia, dyspnea, hypertension, tachycardia, headache, laryngeal edema, pulmonary edema, and ocular adverse reactions, including choroidal effusion, acute myopia, and acute angle closure glaucoma. Signs and symptoms may include respiratory symptoms, such as nasal congestion, cough, throat irritation, as well as chills, vomiting, and nausea. Less common signs and symptoms were wheezing, allergic rhinitis, pyrexia, chest discomfort, pruritus, hypotension and blurred vision.
When DARZALEX® dosing was interrupted in the setting of ASCT (CASSIOPEIA) for a median of 3.75 months (range: 2.4 to 6.9 months), upon re-initiation of DARZALEX®, the incidence of infusion-related reactions was 11% for the first infusion following ASCT. Infusion-related reactions occurring at re-initiation of DARZALEX® following ASCT were consistent in terms of symptoms and severity (Grade 3 or 4: <1%) with those reported in previous studies at Week 2 or subsequent infusions. In EQUULEUS, patients receiving combination treatment (n=97) were administered the first 16 mg/kg dose at Week 1 split over two days, ie, 8 mg/kg on Day 1 and Day 2, respectively. The incidence of any grade infusion-related reactions was 42%, with 36% of patients experiencing infusion-related reactions on Day 1 of Week 1, 4% on Day 2 of Week 1, and 8% with subsequent infusions.
Pre-medicate patients with antihistamines, antipyretics, and corticosteroids. Frequently monitor patients during the entire infusion. Interrupt DARZALEX® infusion for reactions of any severity and institute medical management as needed. Permanently discontinue DARZALEX® therapy if an anaphylactic reaction or life-threatening (Grade 4) reaction occurs and institute appropriate emergency care. For patients with Grade 1, 2, or 3 reactions, reduce the infusion rate when re-starting the infusion.
To reduce the risk of delayed infusion-related reactions, administer oral corticosteroids to all patients following DARZALEX® infusions. Patients with a history of chronic obstructive pulmonary disease may require additional post-infusion medications to manage respiratory complications. Consider prescribing short- and long-acting bronchodilators and inhaled corticosteroids for patients with chronic obstructive pulmonary disease.
Ocular adverse reactions, including acute myopia and narrowing of the anterior chamber angle due to ciliochoroidal effusions with potential for increased intraocular pressure or glaucoma, have occurred with DARZALEX infusion. If ocular symptoms occur, interrupt DARZALEX infusion and seek immediate ophthalmologic evaluation prior to restarting DARZALEX.
Interference With Serological Testing
Daratumumab binds to CD38 on red blood cells (RBCs) and results in a positive indirect antiglobulin test (indirect Coombs test). Daratumumab-mediated positive indirect antiglobulin test may persist for up to 6 months after the last daratumumab infusion. Daratumumab bound to RBCs masks detection of antibodies to minor antigens in the patient’s serum. The determination of a patient’s ABO and Rh blood type is not impacted. Notify blood transfusion centers of this interference with serological testing and inform blood banks that a patient has received DARZALEX®. Type and screen patients prior to starting DARZALEX®.
Neutropenia and Thrombocytopenia
DARZALEX® may increase neutropenia and thrombocytopenia induced by background therapy. Monitor complete blood cell counts periodically during treatment according to manufacturer’s prescribing information for background therapies. Monitor patients with neutropenia for signs of infection. Consider withholding DARZALEX® until recovery of neutrophils or for recovery of platelets.
Interference With Determination of Complete Response
Daratumumab is a human immunoglobulin G (IgG) kappa monoclonal antibody that can be detected on both the serum protein electrophoresis (SPE) and immunofixation (IFE) assays used for the clinical monitoring of endogenous M-protein. This interference can impact the determination of complete response and of disease progression in some patients with IgG kappa myeloma protein.
Embryo-Fetal Toxicity
Based on the mechanism of action, DARZALEX® can cause fetal harm when administered to a pregnant woman. DARZALEX® may cause depletion of fetal immune cells and decreased bone density. Advise pregnant women of the potential risk to a fetus. Advise females with reproductive potential to use effective contraception during treatment with DARZALEX® and for 3 months after the last dose.
The combination of DARZALEX® with lenalidomide, pomalidomide, or thalidomide is contraindicated in pregnant women because lenalidomide, pomalidomide, and thalidomide may cause birth defects and death of the unborn child. Refer to the lenalidomide, pomalidomide, or thalidomide prescribing information on use during pregnancy.
ADVERSE REACTIONS
The most frequently reported adverse reactions (incidence ≥20%) were: upper respiratory infection, neutropenia, infusion‑related reactions, thrombocytopenia, diarrhea, constipation, anemia, peripheral sensory neuropathy, fatigue, peripheral edema, nausea, cough, pyrexia, dyspnea, and asthenia. The most common hematologic laboratory abnormalities (≥40%) with DARZALEX® are: neutropenia, lymphopenia, thrombocytopenia, leukopenia, and anemia.
INDICATIONS
DARZALEX® (daratumumab) is indicated for the treatment of adult patients with multiple myeloma:
- In combination with lenalidomide and dexamethasone in newly diagnosed patients who are ineligible for autologous stem cell transplant and in patients with relapsed or refractory multiple myeloma who have received at least one prior therapy
- In combination with bortezomib, melphalan, and prednisone in newly diagnosed patients who are ineligible for autologous stem cell transplant
- In combination with bortezomib, thalidomide, and dexamethasone in newly diagnosed patients who are eligible for autologous stem cell transplant
- In combination with bortezomib and dexamethasone in patients who have received at least one prior therapy
- In combination with carfilzomib and dexamethasone in patients with relapsed or refractory multiple myeloma who have received one to three prior lines of therapy
- In combination with pomalidomide and dexamethasone in patients who have received at least two prior therapies including lenalidomide and a proteasome inhibitor
- As monotherapy in patients who have received at least three prior lines of therapy including a proteasome inhibitor (PI) and an immunomodulatory agent or who are double-refractory to a PI and an immunomodulatory agent
Please click here to see the full Prescribing Information.
1. Richardson PG, San Miguel JF, Moreau P, et al. Interpreting clinical trial data in multiple myeloma: translating findings to the real-world setting. Blood Cancer J. 2018;8(11). doi:10.1038/s41408-018-0141-0
2. Key Statistics About Multiple Myeloma. Cancer.org. Published 2019. https://www.cancer.org/cancer/multiple-myeloma/about/key-statistics.html
3. Fonseca R, Usmani SZ, Mehra M, et al. Frontline treatment patterns and attrition rates by subsequent lines of therapy in patients with newly diagnosed multiple myeloma. BMC Cancer. 2020;20(1). doi:10.1186/s12885-020-07503-y
4. Devarakonda S, Efebera Y, Sharma N. Role of Stem Cell Transplantation in Multiple Myeloma. Cancers. 2021;13(4):863. doi:10.3390/cancers13040863
5. Derudas D, Capraro F, Martinelli G, Cerchione C. How I manage frontline transplant-ineligible multiple myeloma. Hematol Rep. 2020;12(s1). doi:10.4081/hr.2020.8956
6. Kazandjian D. Multiple myeloma epidemiology and survival: A unique malignancy. Semin Oncl. 2016;43(6):676-681. doi:10.1053/j.seminoncol.2016.11.004
7. Facon T, Kumar S, Plesner T, et al. Daratumumab plus lenalidomide and dexamethasone for untreated myeloma. N. Engl. J. Med. 2019;380(22):2104-2115. doi:10.1056/nejmoa1817249
8. DARZALEX® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
9. Facon T, Kumar SK, Plesner T, et al. Daratumumab, lenalidomide, and dexamethasone versus lenalidomide and dexamethasone alone in newly diagnosed multiple myeloma (MAIA): overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(11):1582-1596. doi:10.1016/s1470-2045(21)00466-6
10. Facon T, Dimopoulos MA, Meuleman N, et al. A simplified frailty scale predicts outcomes in transplant-ineligible patients with newly diagnosed multiple myeloma treated in the FIRST (MM-020) trial. Leukemia. 2019;34(1):224-233. doi:10.1038/s41375-019-0539-0
11. Facon T, Kumar SK, Plesner T, et al. Supplement to: Daratumumab, lenalidomide, and dexamethasone versus lenalidomide and dexamethasone alone in newly diagnosed multiple myeloma (MAIA): overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(11):1582-1596.
12. Facon T, Cook G, Usmani SZ, et al. Daratumumab plus lenalidomide and dexamethasone in transplant-ineligible newly diagnosed multiple myeloma: frailty subgroup analysis of MAIA. Leukemia. 2022;36(4):1066-1077. doi:10.1038/s41375-021-01488-8
© Janssen Biotech, Inc. 2022 All rights reserved. 12/22 cp-333446v1