User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
Costs and effects of diabetes on longevity vary across demographics, BMI
BOSTON – Diabetes leads to a loss of 2.3-6.8 life-years and costs between $14,768 and $159,081 over a lifetime, depending on age, sex, race, and body mass index, according to National Health Interview Survey (NHIS) and Medical Expenditure Panel Survey (MEPS) data.
The average number of life-years lost was greater for women than for men, and was greater for blacks than for whites (average of 5.2 for black women vs. 5.0 years for white women, and 4.8 for black men vs. 4.6 for white men). Life-years lost increased with BMI category for most of the age-sex-race combinations evaluated, Man Yee (Mallory) Leung, Ph.D., reported at the annual scientific sessions of the American Diabetes Association. The lifetime costs were higher for men than for women ($93,957 vs. $81,521), and an inverted U-shape was noted across BMI categories for most of the age/sex/race groups, with peaks at the Class II obesity category, said Dr. Leung of Washington University, St. Louis.
Dr. Leung and her colleagues used data from the 2009-2012 NHIS (the most recently available data), and linked to the Medical Expenditure Panel Survey for their analysis. The NHIS Linked Mortality Public-Use Files were used to predict mortality risk.
The sample was divided into groups with different combinations of race, sex, age, and BMI categories, and life expectancies and lifetime health care expenditures for diabetic patients and nondiabetic patients were simulated, she explained, noting that all estimates were adjusted for complex sampling design in the NHIS and MEPS.
The findings underscore the huge economic burden of diabetes on society, and further define the associated life years lost as a result of the disease, she said.
This study was supported by the Agency for Healthcare Research and Quality and the National Institutes of Health, as well as by grants to one of the authors from the Foundation for Barnes-Jewish Hospital and the Breast Cancer Research Foundation. Dr. Leung reported having no conflicts of interest.
BOSTON – Diabetes leads to a loss of 2.3-6.8 life-years and costs between $14,768 and $159,081 over a lifetime, depending on age, sex, race, and body mass index, according to National Health Interview Survey (NHIS) and Medical Expenditure Panel Survey (MEPS) data.
The average number of life-years lost was greater for women than for men, and was greater for blacks than for whites (average of 5.2 for black women vs. 5.0 years for white women, and 4.8 for black men vs. 4.6 for white men). Life-years lost increased with BMI category for most of the age-sex-race combinations evaluated, Man Yee (Mallory) Leung, Ph.D., reported at the annual scientific sessions of the American Diabetes Association. The lifetime costs were higher for men than for women ($93,957 vs. $81,521), and an inverted U-shape was noted across BMI categories for most of the age/sex/race groups, with peaks at the Class II obesity category, said Dr. Leung of Washington University, St. Louis.
Dr. Leung and her colleagues used data from the 2009-2012 NHIS (the most recently available data), and linked to the Medical Expenditure Panel Survey for their analysis. The NHIS Linked Mortality Public-Use Files were used to predict mortality risk.
The sample was divided into groups with different combinations of race, sex, age, and BMI categories, and life expectancies and lifetime health care expenditures for diabetic patients and nondiabetic patients were simulated, she explained, noting that all estimates were adjusted for complex sampling design in the NHIS and MEPS.
The findings underscore the huge economic burden of diabetes on society, and further define the associated life years lost as a result of the disease, she said.
This study was supported by the Agency for Healthcare Research and Quality and the National Institutes of Health, as well as by grants to one of the authors from the Foundation for Barnes-Jewish Hospital and the Breast Cancer Research Foundation. Dr. Leung reported having no conflicts of interest.
BOSTON – Diabetes leads to a loss of 2.3-6.8 life-years and costs between $14,768 and $159,081 over a lifetime, depending on age, sex, race, and body mass index, according to National Health Interview Survey (NHIS) and Medical Expenditure Panel Survey (MEPS) data.
The average number of life-years lost was greater for women than for men, and was greater for blacks than for whites (average of 5.2 for black women vs. 5.0 years for white women, and 4.8 for black men vs. 4.6 for white men). Life-years lost increased with BMI category for most of the age-sex-race combinations evaluated, Man Yee (Mallory) Leung, Ph.D., reported at the annual scientific sessions of the American Diabetes Association. The lifetime costs were higher for men than for women ($93,957 vs. $81,521), and an inverted U-shape was noted across BMI categories for most of the age/sex/race groups, with peaks at the Class II obesity category, said Dr. Leung of Washington University, St. Louis.
Dr. Leung and her colleagues used data from the 2009-2012 NHIS (the most recently available data), and linked to the Medical Expenditure Panel Survey for their analysis. The NHIS Linked Mortality Public-Use Files were used to predict mortality risk.
The sample was divided into groups with different combinations of race, sex, age, and BMI categories, and life expectancies and lifetime health care expenditures for diabetic patients and nondiabetic patients were simulated, she explained, noting that all estimates were adjusted for complex sampling design in the NHIS and MEPS.
The findings underscore the huge economic burden of diabetes on society, and further define the associated life years lost as a result of the disease, she said.
This study was supported by the Agency for Healthcare Research and Quality and the National Institutes of Health, as well as by grants to one of the authors from the Foundation for Barnes-Jewish Hospital and the Breast Cancer Research Foundation. Dr. Leung reported having no conflicts of interest.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Key clinical point: Diabetes leads to a loss of 2.3-6.8 life-years and costs between $14,768 and $159,081 over a lifetime, depending on age, gender, race, and body mass index, according to National Health Interview Survey and Medical Expenditure Panel Survey data.
Major finding: The economic burden of diabetes in terms of life-years and actual costs are significant and measurable, depending on age, sex, race, and BMI.
Data source: The 2009-2012 National Health Interview Survey and the Medical Expenditure Panel Survey.
Disclosures: This study was supported by the AHRQ and the NIH, as well as by grants to one of the authors from the Foundation for Barnes-Jewish Hospital and the Breast Cancer Research Foundation. Dr. Leung reported having no conflicts of interest.
Earlier start to plasma transfusions for persistent postpartum hemorrhage does not improve maternal outcomes
TORONTO – For women with persistent postpartum hemorrhage, correction of coagulopathy by administering plasma is often an inevitable treatment decision. There are little clinical data, however, on whether an early or a late start for plasma transfusion leads to more positive maternal outcomes.
Data presented on Wednesday at the ISTH 2015 Congress by Dr. Dacia Henriquez of the department of obstetrics and gynecology at Leiden (the Netherlands) University Medical Center, may help answer that question.
“Our findings do not support the notion that plasma transfusion within 60 minutes after diagnosing persistent postpartum hemorrhage contributes to the reduction of the risk of adverse maternal outcomes,” Dr. Henriquez said in an interview.
The TeMpOh-1 (Transfusion Strategies in Women with Major Obstetric Hemorrhage) study included 1,217 women with postpartum hemorrhage (PPH) that was refractory to first-line obstetric interventions. The women were treated at 61 Dutch hospitals in 2011 and 2012.
“With this selection, our study results can be easily translated to daily clinical practice because of this clearly identifiable turning-point in treatment,” she said.
All women included in the study received either at least four units of red blood cells or a multicomponent transfusion within 24 hours after delivery (≥ 1000 mL). Median bleeding rate at baseline was 1.1 L at 1 hour, and median total blood loss was 3.0 L. Marginal structural models with inverse probability of treatment weighting were constructed to quantify the difference in maternal outcomes – including arterial embolism, hysterectomy, and maternal mortality – between an early (within 60 minutes) and a late (after 60 minutes) start of plasma transfusion (163 women vs. 1,054 women, respectively).
After adjustment for baseline parameters, bleeding severity, and changes over time, no significant differences were seen between the groups (odds ratios, 1.1 and 0.8, respectively).
“It seemed that after adjustment for patient and bleeding characteristics, plasma transfusion within 60 minutes after diagnosis of persistent PPH was not associated with a reduction in adverse maternal outcomes,” she said, noting that analyses are ongoing, and efforts are underway to determine whether time-dependent confounding was handled properly in the study.
Plans are also in place to perform subgroup analyses to assess whether treatment effect will differ between those with the most severe bleeding and those with initially milder hemorrhage, she said.
“With the TeMpOH-1 data, we were also able to demonstrate that dilutional coagulopathy due to volume resuscitation with crystalloids and colloids was associated with greater total blood loss and more adverse maternal outcomes when the clear fluids volume exceeded 4 L,” Dr. Henriquez said.
“A treatment strategy aiming at prevention rather than correction of coagulopathy in women with persistent PPH in daily clinical practice seems justified, because correction of coagulopathy by administering plasma appears to be of limited value in reducing adverse maternal outcomes,” Dr. Henriquez said.
Dr. Henriquez declared no disclosures.
TORONTO – For women with persistent postpartum hemorrhage, correction of coagulopathy by administering plasma is often an inevitable treatment decision. There are little clinical data, however, on whether an early or a late start for plasma transfusion leads to more positive maternal outcomes.
Data presented on Wednesday at the ISTH 2015 Congress by Dr. Dacia Henriquez of the department of obstetrics and gynecology at Leiden (the Netherlands) University Medical Center, may help answer that question.
“Our findings do not support the notion that plasma transfusion within 60 minutes after diagnosing persistent postpartum hemorrhage contributes to the reduction of the risk of adverse maternal outcomes,” Dr. Henriquez said in an interview.
The TeMpOh-1 (Transfusion Strategies in Women with Major Obstetric Hemorrhage) study included 1,217 women with postpartum hemorrhage (PPH) that was refractory to first-line obstetric interventions. The women were treated at 61 Dutch hospitals in 2011 and 2012.
“With this selection, our study results can be easily translated to daily clinical practice because of this clearly identifiable turning-point in treatment,” she said.
All women included in the study received either at least four units of red blood cells or a multicomponent transfusion within 24 hours after delivery (≥ 1000 mL). Median bleeding rate at baseline was 1.1 L at 1 hour, and median total blood loss was 3.0 L. Marginal structural models with inverse probability of treatment weighting were constructed to quantify the difference in maternal outcomes – including arterial embolism, hysterectomy, and maternal mortality – between an early (within 60 minutes) and a late (after 60 minutes) start of plasma transfusion (163 women vs. 1,054 women, respectively).
After adjustment for baseline parameters, bleeding severity, and changes over time, no significant differences were seen between the groups (odds ratios, 1.1 and 0.8, respectively).
“It seemed that after adjustment for patient and bleeding characteristics, plasma transfusion within 60 minutes after diagnosis of persistent PPH was not associated with a reduction in adverse maternal outcomes,” she said, noting that analyses are ongoing, and efforts are underway to determine whether time-dependent confounding was handled properly in the study.
Plans are also in place to perform subgroup analyses to assess whether treatment effect will differ between those with the most severe bleeding and those with initially milder hemorrhage, she said.
“With the TeMpOH-1 data, we were also able to demonstrate that dilutional coagulopathy due to volume resuscitation with crystalloids and colloids was associated with greater total blood loss and more adverse maternal outcomes when the clear fluids volume exceeded 4 L,” Dr. Henriquez said.
“A treatment strategy aiming at prevention rather than correction of coagulopathy in women with persistent PPH in daily clinical practice seems justified, because correction of coagulopathy by administering plasma appears to be of limited value in reducing adverse maternal outcomes,” Dr. Henriquez said.
Dr. Henriquez declared no disclosures.
TORONTO – For women with persistent postpartum hemorrhage, correction of coagulopathy by administering plasma is often an inevitable treatment decision. There are little clinical data, however, on whether an early or a late start for plasma transfusion leads to more positive maternal outcomes.
Data presented on Wednesday at the ISTH 2015 Congress by Dr. Dacia Henriquez of the department of obstetrics and gynecology at Leiden (the Netherlands) University Medical Center, may help answer that question.
“Our findings do not support the notion that plasma transfusion within 60 minutes after diagnosing persistent postpartum hemorrhage contributes to the reduction of the risk of adverse maternal outcomes,” Dr. Henriquez said in an interview.
The TeMpOh-1 (Transfusion Strategies in Women with Major Obstetric Hemorrhage) study included 1,217 women with postpartum hemorrhage (PPH) that was refractory to first-line obstetric interventions. The women were treated at 61 Dutch hospitals in 2011 and 2012.
“With this selection, our study results can be easily translated to daily clinical practice because of this clearly identifiable turning-point in treatment,” she said.
All women included in the study received either at least four units of red blood cells or a multicomponent transfusion within 24 hours after delivery (≥ 1000 mL). Median bleeding rate at baseline was 1.1 L at 1 hour, and median total blood loss was 3.0 L. Marginal structural models with inverse probability of treatment weighting were constructed to quantify the difference in maternal outcomes – including arterial embolism, hysterectomy, and maternal mortality – between an early (within 60 minutes) and a late (after 60 minutes) start of plasma transfusion (163 women vs. 1,054 women, respectively).
After adjustment for baseline parameters, bleeding severity, and changes over time, no significant differences were seen between the groups (odds ratios, 1.1 and 0.8, respectively).
“It seemed that after adjustment for patient and bleeding characteristics, plasma transfusion within 60 minutes after diagnosis of persistent PPH was not associated with a reduction in adverse maternal outcomes,” she said, noting that analyses are ongoing, and efforts are underway to determine whether time-dependent confounding was handled properly in the study.
Plans are also in place to perform subgroup analyses to assess whether treatment effect will differ between those with the most severe bleeding and those with initially milder hemorrhage, she said.
“With the TeMpOH-1 data, we were also able to demonstrate that dilutional coagulopathy due to volume resuscitation with crystalloids and colloids was associated with greater total blood loss and more adverse maternal outcomes when the clear fluids volume exceeded 4 L,” Dr. Henriquez said.
“A treatment strategy aiming at prevention rather than correction of coagulopathy in women with persistent PPH in daily clinical practice seems justified, because correction of coagulopathy by administering plasma appears to be of limited value in reducing adverse maternal outcomes,” Dr. Henriquez said.
Dr. Henriquez declared no disclosures.
AT 2015 ISTH
Key clinical point: Plasma transfusion within 60 minutes after diagnosis of persistent PPH was not associated with a reduction in adverse maternal outcomes.
Major finding: No significant differences were seen in maternal outcomes (ncluding arterial embolism, hysterectomy and maternal mortality) between the patients treated with plasma transfusions early (within 60 minutes) and late (after 60 minutes) (odds ratios, 1.1 and 0.8, respectively).
Data source: 1,217 women with postpartum hemorrhage (PPH) that was refractory to first-line obstetric interventions I.
Disclosures: Dr. Henriquez declared no disclosures.
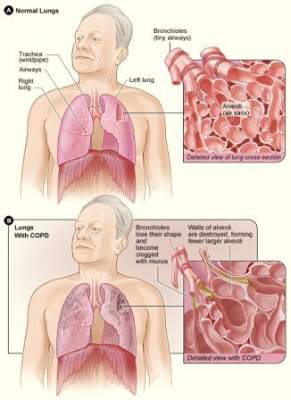
ATS: Glycopyrronium improves lung function, health status in COPD
DENVER – Twice-daily treatment with the long-acting muscarinic antagonist glycopyrronium improved lung function and health status in patients with chronic obstructive pulmonary disorder (COPD) and moderate to severe airflow limitation in the randomized, double-blind, placebo-controlled GEM2 study.
In patients with stable symptomatic COPD with moderate to severe airflow limitation, twice-daily 12.5-mcg dosing of glycopyrronium provides clinically meaningful improvement in lung function over the 12-hour dosing interval, has early onset, is sustained over 12 weeks when compared with placebo, and is associated with significant improvements in COPD symptoms, health status, and rescue medication use, as well as numerical improvement in dyspnea scores, Dr. Edward Kerwin of the Clinical Research Institute of Southern Oregon, PC, Medford, and his colleagues reported in a poster at an international conference of the American Thoracic Society.
Lung function – as measured by forced expiratory volume in 1 second (FEV1) area under the curve from 0-12 hours (AUC 0-12h) – was significantly better both at day 1 and at week 12 of treatment in 216 patients who were randomized to receive a 12.5 mcg twice daily dose of the fast-onset, long-acting muscarinic antagonist (Novartis), compared with 216 patients who received placebo; there was a “significant and clinically meaningful between-treatment difference of 119 and 123 mL, respectively,” the researchers wrote.
“Glycopyrronium also showed consistently significant improvements in trough FEV1 vs. placebo at all assessed time points,” they said, adding that glycopyrronium showed an early onset of bronchodilation with significant improvements in FEV1 at 5 and 15 minutes post dose, compared with placebo at day 1 and week 12.
The least squares mean treatment differences for glycopyrronium vs. placebo for change from baseline in trough forced vital capacity (FVC) were 171 mL on day 2, and 130 mL at week 12. Peak FEV1 and peak FVC were significantly improved with glycopyrronium vs. placebo on day 1 (least squares mean treatment differences of 137 and 223 mL, respectively) and at week 12 (least squares mean treatment differences of 148 and 201 mL, respectively).
Health status was improved at week 12, with both significant and clinically meaningful improvements in St. George’s Respiratory Questionnaire total score in the treatment vs. placebo group (–6.4 vs. –1.2), and the percentage of patients achieving minimal clinically important differences (MCID), defined as at least 4 units, was significantly higher in the treatment vs. placebo group (54.9% vs. 42.3%), the investigators said.
Additionally, numerical improvements in transition dyspnea index total score, and percentage of patients achieving a MCID, defined as at least 1 unit, were observed at week 12 in the glycopyrronium vs. placebo group.
Patients in the glycopyrronium group showed improvement on all symptoms scores and endpoints, according to data recorded in patient e-diaries, and those in the treatment group also were able to perform usual daily activities significantly more often than those in the placebo group.
“A statistically significant decrease in daily, daytime, and nighttime number of puffs, and a significant increase in the percentage of days with no rescue medication use were observed,” the investigators noted.
Patients included in the multicenter GEM2 (Glycopyrronium Effect on Symptoms and Lung Function) study were adults aged 40 years and older with moderate to severe airflow limitation (GOLD 2011 strategy level 2 or 3), who were either current or former smokers with a smoking history of at least 10 pack-years. All had postbronchodilator FEV1 of at least 30% and less than 80% of the predicted value, and postbronchodilator FEV1/forced vital capacity ratio of less than 0.70 at a run-in visit. They also all had a modified Medical Research Council grade of 2 or greater at the run-in visit. Patients with a history of asthma or with a COPD exacerbation requiring treatment with antibiotics and/or systemic corticosteroids, and/or with hospitalization within 6 weeks of the screening and run-in periods were excluded, as were those with a history of long QT syndrome or whose corrected QT was greater than 450 ms at the run-in visit.
All underwent an initial 1- to 7-day washout period and a 2-week run-in period prior to randomization, as well as a safety follow-up period. Patients received either glycopyrronium 12. 5 mcg or placebo twice daily delivered via the Neohaler device for 12 weeks.
Treatment was generally well tolerated; of the 430 patients included in the safety set, 44 permanently discontinued treatment due to adverse events (4.6% and 4.2% in the treatment and placebo groups, respectively). The number who experienced at least one adverse event during the treatment period was similar in the two groups; COPD was the most common adverse event, occurring in 20.8% of those in the treatment group and 21.5% in the placebo group.
A nonfatal myocardial infraction occurred in one patient in the treatment group.
Laboratory parameters and vital sign findings were comparable in the two groups.
Based on the findings of the GEM studies, Novartis has submitted a New Drug Application to the Food and Drug Administration; glycopyrronium is already approved in more than 70 countries, including countries in Latin America and the European Union, as a once-daily treatment marketed as the Seebri Breezhaler.
The GEM2 study was sponsored by Novartis Pharmaceuticals. Two of the study researchers are Novartis employees.
DENVER – Twice-daily treatment with the long-acting muscarinic antagonist glycopyrronium improved lung function and health status in patients with chronic obstructive pulmonary disorder (COPD) and moderate to severe airflow limitation in the randomized, double-blind, placebo-controlled GEM2 study.
In patients with stable symptomatic COPD with moderate to severe airflow limitation, twice-daily 12.5-mcg dosing of glycopyrronium provides clinically meaningful improvement in lung function over the 12-hour dosing interval, has early onset, is sustained over 12 weeks when compared with placebo, and is associated with significant improvements in COPD symptoms, health status, and rescue medication use, as well as numerical improvement in dyspnea scores, Dr. Edward Kerwin of the Clinical Research Institute of Southern Oregon, PC, Medford, and his colleagues reported in a poster at an international conference of the American Thoracic Society.
Lung function – as measured by forced expiratory volume in 1 second (FEV1) area under the curve from 0-12 hours (AUC 0-12h) – was significantly better both at day 1 and at week 12 of treatment in 216 patients who were randomized to receive a 12.5 mcg twice daily dose of the fast-onset, long-acting muscarinic antagonist (Novartis), compared with 216 patients who received placebo; there was a “significant and clinically meaningful between-treatment difference of 119 and 123 mL, respectively,” the researchers wrote.
“Glycopyrronium also showed consistently significant improvements in trough FEV1 vs. placebo at all assessed time points,” they said, adding that glycopyrronium showed an early onset of bronchodilation with significant improvements in FEV1 at 5 and 15 minutes post dose, compared with placebo at day 1 and week 12.
The least squares mean treatment differences for glycopyrronium vs. placebo for change from baseline in trough forced vital capacity (FVC) were 171 mL on day 2, and 130 mL at week 12. Peak FEV1 and peak FVC were significantly improved with glycopyrronium vs. placebo on day 1 (least squares mean treatment differences of 137 and 223 mL, respectively) and at week 12 (least squares mean treatment differences of 148 and 201 mL, respectively).
Health status was improved at week 12, with both significant and clinically meaningful improvements in St. George’s Respiratory Questionnaire total score in the treatment vs. placebo group (–6.4 vs. –1.2), and the percentage of patients achieving minimal clinically important differences (MCID), defined as at least 4 units, was significantly higher in the treatment vs. placebo group (54.9% vs. 42.3%), the investigators said.
Additionally, numerical improvements in transition dyspnea index total score, and percentage of patients achieving a MCID, defined as at least 1 unit, were observed at week 12 in the glycopyrronium vs. placebo group.
Patients in the glycopyrronium group showed improvement on all symptoms scores and endpoints, according to data recorded in patient e-diaries, and those in the treatment group also were able to perform usual daily activities significantly more often than those in the placebo group.
“A statistically significant decrease in daily, daytime, and nighttime number of puffs, and a significant increase in the percentage of days with no rescue medication use were observed,” the investigators noted.
Patients included in the multicenter GEM2 (Glycopyrronium Effect on Symptoms and Lung Function) study were adults aged 40 years and older with moderate to severe airflow limitation (GOLD 2011 strategy level 2 or 3), who were either current or former smokers with a smoking history of at least 10 pack-years. All had postbronchodilator FEV1 of at least 30% and less than 80% of the predicted value, and postbronchodilator FEV1/forced vital capacity ratio of less than 0.70 at a run-in visit. They also all had a modified Medical Research Council grade of 2 or greater at the run-in visit. Patients with a history of asthma or with a COPD exacerbation requiring treatment with antibiotics and/or systemic corticosteroids, and/or with hospitalization within 6 weeks of the screening and run-in periods were excluded, as were those with a history of long QT syndrome or whose corrected QT was greater than 450 ms at the run-in visit.
All underwent an initial 1- to 7-day washout period and a 2-week run-in period prior to randomization, as well as a safety follow-up period. Patients received either glycopyrronium 12. 5 mcg or placebo twice daily delivered via the Neohaler device for 12 weeks.
Treatment was generally well tolerated; of the 430 patients included in the safety set, 44 permanently discontinued treatment due to adverse events (4.6% and 4.2% in the treatment and placebo groups, respectively). The number who experienced at least one adverse event during the treatment period was similar in the two groups; COPD was the most common adverse event, occurring in 20.8% of those in the treatment group and 21.5% in the placebo group.
A nonfatal myocardial infraction occurred in one patient in the treatment group.
Laboratory parameters and vital sign findings were comparable in the two groups.
Based on the findings of the GEM studies, Novartis has submitted a New Drug Application to the Food and Drug Administration; glycopyrronium is already approved in more than 70 countries, including countries in Latin America and the European Union, as a once-daily treatment marketed as the Seebri Breezhaler.
The GEM2 study was sponsored by Novartis Pharmaceuticals. Two of the study researchers are Novartis employees.
DENVER – Twice-daily treatment with the long-acting muscarinic antagonist glycopyrronium improved lung function and health status in patients with chronic obstructive pulmonary disorder (COPD) and moderate to severe airflow limitation in the randomized, double-blind, placebo-controlled GEM2 study.
In patients with stable symptomatic COPD with moderate to severe airflow limitation, twice-daily 12.5-mcg dosing of glycopyrronium provides clinically meaningful improvement in lung function over the 12-hour dosing interval, has early onset, is sustained over 12 weeks when compared with placebo, and is associated with significant improvements in COPD symptoms, health status, and rescue medication use, as well as numerical improvement in dyspnea scores, Dr. Edward Kerwin of the Clinical Research Institute of Southern Oregon, PC, Medford, and his colleagues reported in a poster at an international conference of the American Thoracic Society.
Lung function – as measured by forced expiratory volume in 1 second (FEV1) area under the curve from 0-12 hours (AUC 0-12h) – was significantly better both at day 1 and at week 12 of treatment in 216 patients who were randomized to receive a 12.5 mcg twice daily dose of the fast-onset, long-acting muscarinic antagonist (Novartis), compared with 216 patients who received placebo; there was a “significant and clinically meaningful between-treatment difference of 119 and 123 mL, respectively,” the researchers wrote.
“Glycopyrronium also showed consistently significant improvements in trough FEV1 vs. placebo at all assessed time points,” they said, adding that glycopyrronium showed an early onset of bronchodilation with significant improvements in FEV1 at 5 and 15 minutes post dose, compared with placebo at day 1 and week 12.
The least squares mean treatment differences for glycopyrronium vs. placebo for change from baseline in trough forced vital capacity (FVC) were 171 mL on day 2, and 130 mL at week 12. Peak FEV1 and peak FVC were significantly improved with glycopyrronium vs. placebo on day 1 (least squares mean treatment differences of 137 and 223 mL, respectively) and at week 12 (least squares mean treatment differences of 148 and 201 mL, respectively).
Health status was improved at week 12, with both significant and clinically meaningful improvements in St. George’s Respiratory Questionnaire total score in the treatment vs. placebo group (–6.4 vs. –1.2), and the percentage of patients achieving minimal clinically important differences (MCID), defined as at least 4 units, was significantly higher in the treatment vs. placebo group (54.9% vs. 42.3%), the investigators said.
Additionally, numerical improvements in transition dyspnea index total score, and percentage of patients achieving a MCID, defined as at least 1 unit, were observed at week 12 in the glycopyrronium vs. placebo group.
Patients in the glycopyrronium group showed improvement on all symptoms scores and endpoints, according to data recorded in patient e-diaries, and those in the treatment group also were able to perform usual daily activities significantly more often than those in the placebo group.
“A statistically significant decrease in daily, daytime, and nighttime number of puffs, and a significant increase in the percentage of days with no rescue medication use were observed,” the investigators noted.
Patients included in the multicenter GEM2 (Glycopyrronium Effect on Symptoms and Lung Function) study were adults aged 40 years and older with moderate to severe airflow limitation (GOLD 2011 strategy level 2 or 3), who were either current or former smokers with a smoking history of at least 10 pack-years. All had postbronchodilator FEV1 of at least 30% and less than 80% of the predicted value, and postbronchodilator FEV1/forced vital capacity ratio of less than 0.70 at a run-in visit. They also all had a modified Medical Research Council grade of 2 or greater at the run-in visit. Patients with a history of asthma or with a COPD exacerbation requiring treatment with antibiotics and/or systemic corticosteroids, and/or with hospitalization within 6 weeks of the screening and run-in periods were excluded, as were those with a history of long QT syndrome or whose corrected QT was greater than 450 ms at the run-in visit.
All underwent an initial 1- to 7-day washout period and a 2-week run-in period prior to randomization, as well as a safety follow-up period. Patients received either glycopyrronium 12. 5 mcg or placebo twice daily delivered via the Neohaler device for 12 weeks.
Treatment was generally well tolerated; of the 430 patients included in the safety set, 44 permanently discontinued treatment due to adverse events (4.6% and 4.2% in the treatment and placebo groups, respectively). The number who experienced at least one adverse event during the treatment period was similar in the two groups; COPD was the most common adverse event, occurring in 20.8% of those in the treatment group and 21.5% in the placebo group.
A nonfatal myocardial infraction occurred in one patient in the treatment group.
Laboratory parameters and vital sign findings were comparable in the two groups.
Based on the findings of the GEM studies, Novartis has submitted a New Drug Application to the Food and Drug Administration; glycopyrronium is already approved in more than 70 countries, including countries in Latin America and the European Union, as a once-daily treatment marketed as the Seebri Breezhaler.
The GEM2 study was sponsored by Novartis Pharmaceuticals. Two of the study researchers are Novartis employees.
AT ATS 2015
Key clinical point: Glycopyrronium provides fast-onset, clinically meaningful improvement in lung function vs. placebo in COPD patients with moderate to severe airflow limitation.
Major finding: The between-treatment differences in FEV1 AUC (0-12 h) at day 1 and week 12 of treatment were 119 and 123 mL, respectively.
Data source: A randomized, double-blind, placebo-controlled trial of 432 patients.
Disclosures: The GEM2 study was sponsored by Novartis Pharmaceuticals. Two of the study researchers are Novartis employees.
‘Perfect Storm’ of Depression, Stress Raises Risk of MI, Death
Patients with coronary heart disease who have both depression and stress are at increased risk of myocardial infarction and death, according to findings from a large, prospective, cohort study.
Of 4,487 adults with CHD who were part of the Reasons for Geographic and Racial Differences in Stroke (REGARDS) study, 1,337 experienced MI or death during a median of nearly 6 years of follow-up. Those with both high depressive symptoms and high stress at baseline – about 6% of the study population – were at significantly increased risk of such events (adjusted hazard ratio, 1.48) during the first 2.5 years of follow-up, compared with those with low stress and low depressive symptoms. However, the association was not significant beyond the initial 2.5 years (HR, 0.89), Carmela Alcántara, Ph.D., of Columbia University, New York, and her colleagues reported.
Those with low stress and high depressive symptoms, and those with high stress and low depressive symptoms, were not at increased risk (HR, 0.92 and 0.86, respectively) at any point during follow-up (Circ. Cardiovasc. Qual. Outcomes 2015 March 10 [doi:10.1161/IRCOUTCOMES.114.001180]).
The findings provide initial empirical evidence to support a “psychosocial perfect storm conceptual model” based on the idea that it takes an underlying chronic psychosocial vulnerability such as depression along with a more transient state such as psychological stress to precipitate a clinical event. The confluence of these factors may be particularly destructive in the short term, the investigators concluded, noting that the findings could have implications for the development of preventive treatments that focus on depression and stress during this vulnerable period in CHD patients.
The National Institute of Neurological Disorders and Stroke and the National Heart, Lung, and Blood Institute supported the study. Dr. Alcantara reported having no disclosures, but two other authors received salary support from Amgen for research, and one served as a consultant for DiaDexus.
Patients with coronary heart disease who have both depression and stress are at increased risk of myocardial infarction and death, according to findings from a large, prospective, cohort study.
Of 4,487 adults with CHD who were part of the Reasons for Geographic and Racial Differences in Stroke (REGARDS) study, 1,337 experienced MI or death during a median of nearly 6 years of follow-up. Those with both high depressive symptoms and high stress at baseline – about 6% of the study population – were at significantly increased risk of such events (adjusted hazard ratio, 1.48) during the first 2.5 years of follow-up, compared with those with low stress and low depressive symptoms. However, the association was not significant beyond the initial 2.5 years (HR, 0.89), Carmela Alcántara, Ph.D., of Columbia University, New York, and her colleagues reported.
Those with low stress and high depressive symptoms, and those with high stress and low depressive symptoms, were not at increased risk (HR, 0.92 and 0.86, respectively) at any point during follow-up (Circ. Cardiovasc. Qual. Outcomes 2015 March 10 [doi:10.1161/IRCOUTCOMES.114.001180]).
The findings provide initial empirical evidence to support a “psychosocial perfect storm conceptual model” based on the idea that it takes an underlying chronic psychosocial vulnerability such as depression along with a more transient state such as psychological stress to precipitate a clinical event. The confluence of these factors may be particularly destructive in the short term, the investigators concluded, noting that the findings could have implications for the development of preventive treatments that focus on depression and stress during this vulnerable period in CHD patients.
The National Institute of Neurological Disorders and Stroke and the National Heart, Lung, and Blood Institute supported the study. Dr. Alcantara reported having no disclosures, but two other authors received salary support from Amgen for research, and one served as a consultant for DiaDexus.
Patients with coronary heart disease who have both depression and stress are at increased risk of myocardial infarction and death, according to findings from a large, prospective, cohort study.
Of 4,487 adults with CHD who were part of the Reasons for Geographic and Racial Differences in Stroke (REGARDS) study, 1,337 experienced MI or death during a median of nearly 6 years of follow-up. Those with both high depressive symptoms and high stress at baseline – about 6% of the study population – were at significantly increased risk of such events (adjusted hazard ratio, 1.48) during the first 2.5 years of follow-up, compared with those with low stress and low depressive symptoms. However, the association was not significant beyond the initial 2.5 years (HR, 0.89), Carmela Alcántara, Ph.D., of Columbia University, New York, and her colleagues reported.
Those with low stress and high depressive symptoms, and those with high stress and low depressive symptoms, were not at increased risk (HR, 0.92 and 0.86, respectively) at any point during follow-up (Circ. Cardiovasc. Qual. Outcomes 2015 March 10 [doi:10.1161/IRCOUTCOMES.114.001180]).
The findings provide initial empirical evidence to support a “psychosocial perfect storm conceptual model” based on the idea that it takes an underlying chronic psychosocial vulnerability such as depression along with a more transient state such as psychological stress to precipitate a clinical event. The confluence of these factors may be particularly destructive in the short term, the investigators concluded, noting that the findings could have implications for the development of preventive treatments that focus on depression and stress during this vulnerable period in CHD patients.
The National Institute of Neurological Disorders and Stroke and the National Heart, Lung, and Blood Institute supported the study. Dr. Alcantara reported having no disclosures, but two other authors received salary support from Amgen for research, and one served as a consultant for DiaDexus.
FROM CIRCULATION: CARDIOVASCULAR QUALITY AND OUTCOMES
ADA: Mobile System Promotes Better Diabetes Self-management
BOSTON – A mobile health diabetes self-management system improves diabetes knowledge, reminds users to take medications and attend appointments, and helps with health-related goal setting, according to findings in patients who used the system for at least 6 months.
The system – Care4life – is a mobile text, e-mail, app, and web-based system developed by digital health provider Voxiva in collaboration with the American Diabetes Association. It provides individualized education and reminders and can log and track blood glucose, weight, exercise, and blood pressure. Summary reports of progress in meeting treatment goals can be generated and shared with providers who can then help patients in their treatment and self-management decision making.
Of the 10,740 patients who enrolled in the Care4Life system through the ADA’s Living With Type 2 Diabetes Website between July 2013 and December 2014 and who used the system for at least 180 days, 97.7% were still receiving educational content after 6 months. Nearly 76% received that content via e-mail, and 68% received it by text message instead of, or in addition to, e-mail, Dr. Joshua L. Cohen reported at the annual scientific sessions of the American Diabetes Association.
About a quarter of the participants had used the system to set a weight loss goal, and 22% used it to set an exercise goal. About 10% used it for medication reminders, and 31% entered at least one health-related report (such as blood glucose measures, weight, blood pressure, medication compliance, or exercise activity.) About 80% of those who set a weight loss or exercise goal submitted at least one report.
About 10% entered one or more blood glucose reports, and of those, 68% entered a report during the first 30 days of system use, a third did so during days 90-120, and 22% did so at 180 or more days after enrollment.
Of 3,263 participants who completed a survey after 180 days of using the Care4Life system, 89% said the system improved their knowledge of diabetes, and 69% said the system helped them remember to take medications and attend appointments, said Dr. Cohen, professor of medicine at George Washington University, Washington, D.C., as well as director of both the diabetes and thyroid centers there.
Further, 82% of respondents reported that the system helped them set health goals, and about 96% said they would recommend the system to other patients with diabetes, he noted.
“Our patients need to make frequent decisions and choices in their personal management of diabetes. As physicians and health educators, one of our major tasks is to provide support to our patients in their own decision making,” Dr. Cohen said.
Although several randomized controlled trials have evaluated mobile health applications and systems and demonstrated benefit, most have been small studies.
The current study included a much larger patient population and further demonstrated the value of this type of technology, he said, noting that the areas of greatest interest were weight management and exercise.
Of note, although a considerable number of patients were still reporting blood glucose levels after 180 days (and the average levels declined over time), only 1% of participants requested reminders regarding blood pressure.
“The latter I find particularly interesting since, as we are all aware, hypertension is indeed a significant contributor to the morbidity and mortality associated with diabetes, and yet – at least in this patient group – the message may not be getting through about the significance of hypertension,” he said.
Dr. Cohen is on speakers panels for AstraZeneca and Pfizer, and he disclosed that one of his study coauthors has a financial interest in Voxiva.
BOSTON – A mobile health diabetes self-management system improves diabetes knowledge, reminds users to take medications and attend appointments, and helps with health-related goal setting, according to findings in patients who used the system for at least 6 months.
The system – Care4life – is a mobile text, e-mail, app, and web-based system developed by digital health provider Voxiva in collaboration with the American Diabetes Association. It provides individualized education and reminders and can log and track blood glucose, weight, exercise, and blood pressure. Summary reports of progress in meeting treatment goals can be generated and shared with providers who can then help patients in their treatment and self-management decision making.
Of the 10,740 patients who enrolled in the Care4Life system through the ADA’s Living With Type 2 Diabetes Website between July 2013 and December 2014 and who used the system for at least 180 days, 97.7% were still receiving educational content after 6 months. Nearly 76% received that content via e-mail, and 68% received it by text message instead of, or in addition to, e-mail, Dr. Joshua L. Cohen reported at the annual scientific sessions of the American Diabetes Association.
About a quarter of the participants had used the system to set a weight loss goal, and 22% used it to set an exercise goal. About 10% used it for medication reminders, and 31% entered at least one health-related report (such as blood glucose measures, weight, blood pressure, medication compliance, or exercise activity.) About 80% of those who set a weight loss or exercise goal submitted at least one report.
About 10% entered one or more blood glucose reports, and of those, 68% entered a report during the first 30 days of system use, a third did so during days 90-120, and 22% did so at 180 or more days after enrollment.
Of 3,263 participants who completed a survey after 180 days of using the Care4Life system, 89% said the system improved their knowledge of diabetes, and 69% said the system helped them remember to take medications and attend appointments, said Dr. Cohen, professor of medicine at George Washington University, Washington, D.C., as well as director of both the diabetes and thyroid centers there.
Further, 82% of respondents reported that the system helped them set health goals, and about 96% said they would recommend the system to other patients with diabetes, he noted.
“Our patients need to make frequent decisions and choices in their personal management of diabetes. As physicians and health educators, one of our major tasks is to provide support to our patients in their own decision making,” Dr. Cohen said.
Although several randomized controlled trials have evaluated mobile health applications and systems and demonstrated benefit, most have been small studies.
The current study included a much larger patient population and further demonstrated the value of this type of technology, he said, noting that the areas of greatest interest were weight management and exercise.
Of note, although a considerable number of patients were still reporting blood glucose levels after 180 days (and the average levels declined over time), only 1% of participants requested reminders regarding blood pressure.
“The latter I find particularly interesting since, as we are all aware, hypertension is indeed a significant contributor to the morbidity and mortality associated with diabetes, and yet – at least in this patient group – the message may not be getting through about the significance of hypertension,” he said.
Dr. Cohen is on speakers panels for AstraZeneca and Pfizer, and he disclosed that one of his study coauthors has a financial interest in Voxiva.
BOSTON – A mobile health diabetes self-management system improves diabetes knowledge, reminds users to take medications and attend appointments, and helps with health-related goal setting, according to findings in patients who used the system for at least 6 months.
The system – Care4life – is a mobile text, e-mail, app, and web-based system developed by digital health provider Voxiva in collaboration with the American Diabetes Association. It provides individualized education and reminders and can log and track blood glucose, weight, exercise, and blood pressure. Summary reports of progress in meeting treatment goals can be generated and shared with providers who can then help patients in their treatment and self-management decision making.
Of the 10,740 patients who enrolled in the Care4Life system through the ADA’s Living With Type 2 Diabetes Website between July 2013 and December 2014 and who used the system for at least 180 days, 97.7% were still receiving educational content after 6 months. Nearly 76% received that content via e-mail, and 68% received it by text message instead of, or in addition to, e-mail, Dr. Joshua L. Cohen reported at the annual scientific sessions of the American Diabetes Association.
About a quarter of the participants had used the system to set a weight loss goal, and 22% used it to set an exercise goal. About 10% used it for medication reminders, and 31% entered at least one health-related report (such as blood glucose measures, weight, blood pressure, medication compliance, or exercise activity.) About 80% of those who set a weight loss or exercise goal submitted at least one report.
About 10% entered one or more blood glucose reports, and of those, 68% entered a report during the first 30 days of system use, a third did so during days 90-120, and 22% did so at 180 or more days after enrollment.
Of 3,263 participants who completed a survey after 180 days of using the Care4Life system, 89% said the system improved their knowledge of diabetes, and 69% said the system helped them remember to take medications and attend appointments, said Dr. Cohen, professor of medicine at George Washington University, Washington, D.C., as well as director of both the diabetes and thyroid centers there.
Further, 82% of respondents reported that the system helped them set health goals, and about 96% said they would recommend the system to other patients with diabetes, he noted.
“Our patients need to make frequent decisions and choices in their personal management of diabetes. As physicians and health educators, one of our major tasks is to provide support to our patients in their own decision making,” Dr. Cohen said.
Although several randomized controlled trials have evaluated mobile health applications and systems and demonstrated benefit, most have been small studies.
The current study included a much larger patient population and further demonstrated the value of this type of technology, he said, noting that the areas of greatest interest were weight management and exercise.
Of note, although a considerable number of patients were still reporting blood glucose levels after 180 days (and the average levels declined over time), only 1% of participants requested reminders regarding blood pressure.
“The latter I find particularly interesting since, as we are all aware, hypertension is indeed a significant contributor to the morbidity and mortality associated with diabetes, and yet – at least in this patient group – the message may not be getting through about the significance of hypertension,” he said.
Dr. Cohen is on speakers panels for AstraZeneca and Pfizer, and he disclosed that one of his study coauthors has a financial interest in Voxiva.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
ADA: Mobile system promotes better diabetes self-management
BOSTON – A mobile health diabetes self-management system improves diabetes knowledge, reminds users to take medications and attend appointments, and helps with health-related goal setting, according to findings in patients who used the system for at least 6 months.
The system – Care4life – is a mobile text, e-mail, app, and web-based system developed by digital health provider Voxiva in collaboration with the American Diabetes Association. It provides individualized education and reminders and can log and track blood glucose, weight, exercise, and blood pressure. Summary reports of progress in meeting treatment goals can be generated and shared with providers who can then help patients in their treatment and self-management decision making.
Of the 10,740 patients who enrolled in the Care4Life system through the ADA’s Living With Type 2 Diabetes Website between July 2013 and December 2014 and who used the system for at least 180 days, 97.7% were still receiving educational content after 6 months. Nearly 76% received that content via e-mail, and 68% received it by text message instead of, or in addition to, e-mail, Dr. Joshua L. Cohen reported at the annual scientific sessions of the American Diabetes Association.
About a quarter of the participants had used the system to set a weight loss goal, and 22% used it to set an exercise goal. About 10% used it for medication reminders, and 31% entered at least one health-related report (such as blood glucose measures, weight, blood pressure, medication compliance, or exercise activity.) About 80% of those who set a weight loss or exercise goal submitted at least one report.
About 10% entered one or more blood glucose reports, and of those, 68% entered a report during the first 30 days of system use, a third did so during days 90-120, and 22% did so at 180 or more days after enrollment.
Of 3,263 participants who completed a survey after 180 days of using the Care4Life system, 89% said the system improved their knowledge of diabetes, and 69% said the system helped them remember to take medications and attend appointments, said Dr. Cohen, professor of medicine at George Washington University, Washington, D.C., as well as director of both the diabetes and thyroid centers there.
Further, 82% of respondents reported that the system helped them set health goals, and about 96% said they would recommend the system to other patients with diabetes, he noted.
“Our patients need to make frequent decisions and choices in their personal management of diabetes. As physicians and health educators, one of our major tasks is to provide support to our patients in their own decision making,” Dr. Cohen said.
Although several randomized controlled trials have evaluated mobile health applications and systems and demonstrated benefit, most have been small studies.
The current study included a much larger patient population and further demonstrated the value of this type of technology, he said, noting that the areas of greatest interest were weight management and exercise.
Of note, although a considerable number of patients were still reporting blood glucose levels after 180 days (and the average levels declined over time), only 1% of participants requested reminders regarding blood pressure.
“The latter I find particularly interesting since, as we are all aware, hypertension is indeed a significant contributor to the morbidity and mortality associated with diabetes, and yet – at least in this patient group – the message may not be getting through about the significance of hypertension,” he said.
Dr. Cohen is on speakers panels for AstraZeneca and Pfizer, and he disclosed that one of his study coauthors has a financial interest in Voxiva.
BOSTON – A mobile health diabetes self-management system improves diabetes knowledge, reminds users to take medications and attend appointments, and helps with health-related goal setting, according to findings in patients who used the system for at least 6 months.
The system – Care4life – is a mobile text, e-mail, app, and web-based system developed by digital health provider Voxiva in collaboration with the American Diabetes Association. It provides individualized education and reminders and can log and track blood glucose, weight, exercise, and blood pressure. Summary reports of progress in meeting treatment goals can be generated and shared with providers who can then help patients in their treatment and self-management decision making.
Of the 10,740 patients who enrolled in the Care4Life system through the ADA’s Living With Type 2 Diabetes Website between July 2013 and December 2014 and who used the system for at least 180 days, 97.7% were still receiving educational content after 6 months. Nearly 76% received that content via e-mail, and 68% received it by text message instead of, or in addition to, e-mail, Dr. Joshua L. Cohen reported at the annual scientific sessions of the American Diabetes Association.
About a quarter of the participants had used the system to set a weight loss goal, and 22% used it to set an exercise goal. About 10% used it for medication reminders, and 31% entered at least one health-related report (such as blood glucose measures, weight, blood pressure, medication compliance, or exercise activity.) About 80% of those who set a weight loss or exercise goal submitted at least one report.
About 10% entered one or more blood glucose reports, and of those, 68% entered a report during the first 30 days of system use, a third did so during days 90-120, and 22% did so at 180 or more days after enrollment.
Of 3,263 participants who completed a survey after 180 days of using the Care4Life system, 89% said the system improved their knowledge of diabetes, and 69% said the system helped them remember to take medications and attend appointments, said Dr. Cohen, professor of medicine at George Washington University, Washington, D.C., as well as director of both the diabetes and thyroid centers there.
Further, 82% of respondents reported that the system helped them set health goals, and about 96% said they would recommend the system to other patients with diabetes, he noted.
“Our patients need to make frequent decisions and choices in their personal management of diabetes. As physicians and health educators, one of our major tasks is to provide support to our patients in their own decision making,” Dr. Cohen said.
Although several randomized controlled trials have evaluated mobile health applications and systems and demonstrated benefit, most have been small studies.
The current study included a much larger patient population and further demonstrated the value of this type of technology, he said, noting that the areas of greatest interest were weight management and exercise.
Of note, although a considerable number of patients were still reporting blood glucose levels after 180 days (and the average levels declined over time), only 1% of participants requested reminders regarding blood pressure.
“The latter I find particularly interesting since, as we are all aware, hypertension is indeed a significant contributor to the morbidity and mortality associated with diabetes, and yet – at least in this patient group – the message may not be getting through about the significance of hypertension,” he said.
Dr. Cohen is on speakers panels for AstraZeneca and Pfizer, and he disclosed that one of his study coauthors has a financial interest in Voxiva.
BOSTON – A mobile health diabetes self-management system improves diabetes knowledge, reminds users to take medications and attend appointments, and helps with health-related goal setting, according to findings in patients who used the system for at least 6 months.
The system – Care4life – is a mobile text, e-mail, app, and web-based system developed by digital health provider Voxiva in collaboration with the American Diabetes Association. It provides individualized education and reminders and can log and track blood glucose, weight, exercise, and blood pressure. Summary reports of progress in meeting treatment goals can be generated and shared with providers who can then help patients in their treatment and self-management decision making.
Of the 10,740 patients who enrolled in the Care4Life system through the ADA’s Living With Type 2 Diabetes Website between July 2013 and December 2014 and who used the system for at least 180 days, 97.7% were still receiving educational content after 6 months. Nearly 76% received that content via e-mail, and 68% received it by text message instead of, or in addition to, e-mail, Dr. Joshua L. Cohen reported at the annual scientific sessions of the American Diabetes Association.
About a quarter of the participants had used the system to set a weight loss goal, and 22% used it to set an exercise goal. About 10% used it for medication reminders, and 31% entered at least one health-related report (such as blood glucose measures, weight, blood pressure, medication compliance, or exercise activity.) About 80% of those who set a weight loss or exercise goal submitted at least one report.
About 10% entered one or more blood glucose reports, and of those, 68% entered a report during the first 30 days of system use, a third did so during days 90-120, and 22% did so at 180 or more days after enrollment.
Of 3,263 participants who completed a survey after 180 days of using the Care4Life system, 89% said the system improved their knowledge of diabetes, and 69% said the system helped them remember to take medications and attend appointments, said Dr. Cohen, professor of medicine at George Washington University, Washington, D.C., as well as director of both the diabetes and thyroid centers there.
Further, 82% of respondents reported that the system helped them set health goals, and about 96% said they would recommend the system to other patients with diabetes, he noted.
“Our patients need to make frequent decisions and choices in their personal management of diabetes. As physicians and health educators, one of our major tasks is to provide support to our patients in their own decision making,” Dr. Cohen said.
Although several randomized controlled trials have evaluated mobile health applications and systems and demonstrated benefit, most have been small studies.
The current study included a much larger patient population and further demonstrated the value of this type of technology, he said, noting that the areas of greatest interest were weight management and exercise.
Of note, although a considerable number of patients were still reporting blood glucose levels after 180 days (and the average levels declined over time), only 1% of participants requested reminders regarding blood pressure.
“The latter I find particularly interesting since, as we are all aware, hypertension is indeed a significant contributor to the morbidity and mortality associated with diabetes, and yet – at least in this patient group – the message may not be getting through about the significance of hypertension,” he said.
Dr. Cohen is on speakers panels for AstraZeneca and Pfizer, and he disclosed that one of his study coauthors has a financial interest in Voxiva.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Key clinical point: A mobile health diabetes self-management system improves diabetes knowledge, helps users remember to take medications and attend appointments, and helps with health-related goal setting.
Major finding: 97.7% of users were still receiving educational content after 6 months, and 96% would recommend the system to others with diabetes.
Data source: A review of usage patterns among 10,740 users of the Care4Life system.
Disclosures: Dr. Cohen is on speakers panels for AstraZeneca and Pfizer, and he disclosed that one of his study coauthors has a financial interest in Vioxiva.
Lixisenatide news is good, but search for the ‘holy grail’ continues
BOSTON – The ELIXA trial confirms that the GLP-1 receptor agonist lixisenatide neither decreases nor increases the risk of major adverse cardiac events in postacute coronary syndrome patients, and that it improves glucose control, compared with placebo, and provides modest benefits with respect to body weight, blood pressure, progression of albuminuria, and hypoglycemia rates, Dr. Silvio E. Inzucchi said at the annual scientific sessions of the American Diabetes Association.
Particularly encouraging was the lack of any signal for pancreatic injury, thyroid cancer, or heart failure. The latter finding is especially timely given recent concerns raised by DPP-4 inhibitors, said Dr. Inzucchi, who was invited by the ADA to comment on the ELIXA (Evaluation of Lixisenatide in Acute Coronary Syndrome) trial findings.
As for whether the ELIXA trial represents a high-quality study with believable results and an important end point, “the answer to this is a resounding yes,” he said, congratulating the investigators, who “have done a fine job of conducting this study, which represents another example of the great collaboration between diabetologists and cardiologists.”
“We all see the same patients and we need to work more and more together to find out the best treatments for our patients,” he said, noting that finding a glucose-lowering agent that has a clearcut cardiovascular benefit represents the “holy grail.”
The neutral finding, as opposed to a positive one, in ELIXA with respect to effects on cardiovascular outcomes is not surprising, said Dr. Inzucchi, professor of medicine and director of the Yale Diabetes Center at Yale University in New Haven, Conn.
“Hypoglycemia is more tightly linked to micro- than to macrovascular outcomes, so not surprisingly it is much easier to show a benefit on these microvascular complications from controlling blood glucose. Any cardiovascular benefit accrues over many years, outside the time course of most of our randomized clinical trials, and any cardiovascular benefit is likely to be attenuated in those with pre-existing atherosclerosis,” he said.
Although Dr. Inzucchi said that he is skeptical that any of the numerous ongoing cardiovascular outcomes trials (CVOTs) will be able to show such a benefit unless a particular agent shows “some dramatic off-target effect on atherosclerosis,” which is an unlikely outcome since glucose lowering has only a modest effect that is disclosed only after a number of years, he noted. It is “just kind of fun to think that, if one of these CVOTs does turn positive … that drug may potentially be positioned as the favored treatment in addition to metformin. Obviously, it depends on the degree of risk reduction and other effects as well, but only then will we have achieved the holy grail.”
BOSTON – The ELIXA trial confirms that the GLP-1 receptor agonist lixisenatide neither decreases nor increases the risk of major adverse cardiac events in postacute coronary syndrome patients, and that it improves glucose control, compared with placebo, and provides modest benefits with respect to body weight, blood pressure, progression of albuminuria, and hypoglycemia rates, Dr. Silvio E. Inzucchi said at the annual scientific sessions of the American Diabetes Association.
Particularly encouraging was the lack of any signal for pancreatic injury, thyroid cancer, or heart failure. The latter finding is especially timely given recent concerns raised by DPP-4 inhibitors, said Dr. Inzucchi, who was invited by the ADA to comment on the ELIXA (Evaluation of Lixisenatide in Acute Coronary Syndrome) trial findings.
As for whether the ELIXA trial represents a high-quality study with believable results and an important end point, “the answer to this is a resounding yes,” he said, congratulating the investigators, who “have done a fine job of conducting this study, which represents another example of the great collaboration between diabetologists and cardiologists.”
“We all see the same patients and we need to work more and more together to find out the best treatments for our patients,” he said, noting that finding a glucose-lowering agent that has a clearcut cardiovascular benefit represents the “holy grail.”
The neutral finding, as opposed to a positive one, in ELIXA with respect to effects on cardiovascular outcomes is not surprising, said Dr. Inzucchi, professor of medicine and director of the Yale Diabetes Center at Yale University in New Haven, Conn.
“Hypoglycemia is more tightly linked to micro- than to macrovascular outcomes, so not surprisingly it is much easier to show a benefit on these microvascular complications from controlling blood glucose. Any cardiovascular benefit accrues over many years, outside the time course of most of our randomized clinical trials, and any cardiovascular benefit is likely to be attenuated in those with pre-existing atherosclerosis,” he said.
Although Dr. Inzucchi said that he is skeptical that any of the numerous ongoing cardiovascular outcomes trials (CVOTs) will be able to show such a benefit unless a particular agent shows “some dramatic off-target effect on atherosclerosis,” which is an unlikely outcome since glucose lowering has only a modest effect that is disclosed only after a number of years, he noted. It is “just kind of fun to think that, if one of these CVOTs does turn positive … that drug may potentially be positioned as the favored treatment in addition to metformin. Obviously, it depends on the degree of risk reduction and other effects as well, but only then will we have achieved the holy grail.”
BOSTON – The ELIXA trial confirms that the GLP-1 receptor agonist lixisenatide neither decreases nor increases the risk of major adverse cardiac events in postacute coronary syndrome patients, and that it improves glucose control, compared with placebo, and provides modest benefits with respect to body weight, blood pressure, progression of albuminuria, and hypoglycemia rates, Dr. Silvio E. Inzucchi said at the annual scientific sessions of the American Diabetes Association.
Particularly encouraging was the lack of any signal for pancreatic injury, thyroid cancer, or heart failure. The latter finding is especially timely given recent concerns raised by DPP-4 inhibitors, said Dr. Inzucchi, who was invited by the ADA to comment on the ELIXA (Evaluation of Lixisenatide in Acute Coronary Syndrome) trial findings.
As for whether the ELIXA trial represents a high-quality study with believable results and an important end point, “the answer to this is a resounding yes,” he said, congratulating the investigators, who “have done a fine job of conducting this study, which represents another example of the great collaboration between diabetologists and cardiologists.”
“We all see the same patients and we need to work more and more together to find out the best treatments for our patients,” he said, noting that finding a glucose-lowering agent that has a clearcut cardiovascular benefit represents the “holy grail.”
The neutral finding, as opposed to a positive one, in ELIXA with respect to effects on cardiovascular outcomes is not surprising, said Dr. Inzucchi, professor of medicine and director of the Yale Diabetes Center at Yale University in New Haven, Conn.
“Hypoglycemia is more tightly linked to micro- than to macrovascular outcomes, so not surprisingly it is much easier to show a benefit on these microvascular complications from controlling blood glucose. Any cardiovascular benefit accrues over many years, outside the time course of most of our randomized clinical trials, and any cardiovascular benefit is likely to be attenuated in those with pre-existing atherosclerosis,” he said.
Although Dr. Inzucchi said that he is skeptical that any of the numerous ongoing cardiovascular outcomes trials (CVOTs) will be able to show such a benefit unless a particular agent shows “some dramatic off-target effect on atherosclerosis,” which is an unlikely outcome since glucose lowering has only a modest effect that is disclosed only after a number of years, he noted. It is “just kind of fun to think that, if one of these CVOTs does turn positive … that drug may potentially be positioned as the favored treatment in addition to metformin. Obviously, it depends on the degree of risk reduction and other effects as well, but only then will we have achieved the holy grail.”
EXPERT ANALYSIS FROM THE ADA ANNUAL SCIENTIFIC SESSIONS
Umeclidinium triple therapy improves lung function in COPD
DENVER – Lung function and health-related quality of life improved for COPD patients who received the long-acting muscarinic agent (LAMA) umeclidinium with fixed-dose inhaled corticosteroid/long-acting beta antagonist (LABA) therapy, based on a post hoc analysis of pooled data from four phase III trials.
Compared with inhaled corticosteroid (ICS)/LABA therapy alone, the triple therapy increased the number of rescue-free days, Dr. Thomas Siler, a pulmonologist with Midwest Chest Consultants, St. Charles, Mo., reported at an international conference of the American Thoracic Society.
The analysis involved 819 patients treated with 62.5 mcg of umeclidinium (Ellipta) – an approved maintenance treatment for COPD – plus ICS/LABA, 821 patients treated with 125 mg umeclidinium plus ICS/LABA, and 818 who received placebo and ICS/LABA. Statistically significant improvements were seen with active triple therapy vs. dual therapy plus placebo in forced expiratory volume in 1 second (FEV1) at day 85 (0.130 L) and at all other time points, as well as in 0-6 h weighted mean FEV1 at day 84 (0.152 L), Dr. Siler said.
With active triple therapy vs. dual therapy plus placebo, overall rescue use was reduced by 0.3 puffs/day and the number of rescue-free days increased by 7.1%. Also, St. George’s Respiratory Questionnaire (SGRQ) score at day 84 decreased by 1.55 vs. placebo, and the proportion of SGRQ responders was 41% vs. 31% for umeclidinium vs. placebo (odds ratio, 1.6).
Moderate/severe COPD exacerbations were experienced by 88 patients: 31 (4%) of the umeclidinium group patients and 57 (7%) of the placebo group patients (hazard ratio, 0.53).
The findings were similar in the patients who received off-label 125-mg dosing of umeclidinium, and the incidence of adverse events and serious adverse events was similar across treatment groups. Dr. Siler noted.
Data on the benefits of LAMAs in triple therapy in patients with moderate to very severe COPD are limited. This pooled analysis of data from four randomized, double-blind, parallel-group 12-week trials of once-daily add-on umeclidinium included COPD patients who entered a 4-week run-in on open-label ICS/LABA (either fluticasone furoate/vilanterol 100/25 mcg or fluticasone propionate/salmeterol 250/50 mcg), and who were then randomized to receive 62.5 or 125 mcg of umeclidinium or placebo.
GlaxoSmithKline funded the study.
DENVER – Lung function and health-related quality of life improved for COPD patients who received the long-acting muscarinic agent (LAMA) umeclidinium with fixed-dose inhaled corticosteroid/long-acting beta antagonist (LABA) therapy, based on a post hoc analysis of pooled data from four phase III trials.
Compared with inhaled corticosteroid (ICS)/LABA therapy alone, the triple therapy increased the number of rescue-free days, Dr. Thomas Siler, a pulmonologist with Midwest Chest Consultants, St. Charles, Mo., reported at an international conference of the American Thoracic Society.
The analysis involved 819 patients treated with 62.5 mcg of umeclidinium (Ellipta) – an approved maintenance treatment for COPD – plus ICS/LABA, 821 patients treated with 125 mg umeclidinium plus ICS/LABA, and 818 who received placebo and ICS/LABA. Statistically significant improvements were seen with active triple therapy vs. dual therapy plus placebo in forced expiratory volume in 1 second (FEV1) at day 85 (0.130 L) and at all other time points, as well as in 0-6 h weighted mean FEV1 at day 84 (0.152 L), Dr. Siler said.
With active triple therapy vs. dual therapy plus placebo, overall rescue use was reduced by 0.3 puffs/day and the number of rescue-free days increased by 7.1%. Also, St. George’s Respiratory Questionnaire (SGRQ) score at day 84 decreased by 1.55 vs. placebo, and the proportion of SGRQ responders was 41% vs. 31% for umeclidinium vs. placebo (odds ratio, 1.6).
Moderate/severe COPD exacerbations were experienced by 88 patients: 31 (4%) of the umeclidinium group patients and 57 (7%) of the placebo group patients (hazard ratio, 0.53).
The findings were similar in the patients who received off-label 125-mg dosing of umeclidinium, and the incidence of adverse events and serious adverse events was similar across treatment groups. Dr. Siler noted.
Data on the benefits of LAMAs in triple therapy in patients with moderate to very severe COPD are limited. This pooled analysis of data from four randomized, double-blind, parallel-group 12-week trials of once-daily add-on umeclidinium included COPD patients who entered a 4-week run-in on open-label ICS/LABA (either fluticasone furoate/vilanterol 100/25 mcg or fluticasone propionate/salmeterol 250/50 mcg), and who were then randomized to receive 62.5 or 125 mcg of umeclidinium or placebo.
GlaxoSmithKline funded the study.
DENVER – Lung function and health-related quality of life improved for COPD patients who received the long-acting muscarinic agent (LAMA) umeclidinium with fixed-dose inhaled corticosteroid/long-acting beta antagonist (LABA) therapy, based on a post hoc analysis of pooled data from four phase III trials.
Compared with inhaled corticosteroid (ICS)/LABA therapy alone, the triple therapy increased the number of rescue-free days, Dr. Thomas Siler, a pulmonologist with Midwest Chest Consultants, St. Charles, Mo., reported at an international conference of the American Thoracic Society.
The analysis involved 819 patients treated with 62.5 mcg of umeclidinium (Ellipta) – an approved maintenance treatment for COPD – plus ICS/LABA, 821 patients treated with 125 mg umeclidinium plus ICS/LABA, and 818 who received placebo and ICS/LABA. Statistically significant improvements were seen with active triple therapy vs. dual therapy plus placebo in forced expiratory volume in 1 second (FEV1) at day 85 (0.130 L) and at all other time points, as well as in 0-6 h weighted mean FEV1 at day 84 (0.152 L), Dr. Siler said.
With active triple therapy vs. dual therapy plus placebo, overall rescue use was reduced by 0.3 puffs/day and the number of rescue-free days increased by 7.1%. Also, St. George’s Respiratory Questionnaire (SGRQ) score at day 84 decreased by 1.55 vs. placebo, and the proportion of SGRQ responders was 41% vs. 31% for umeclidinium vs. placebo (odds ratio, 1.6).
Moderate/severe COPD exacerbations were experienced by 88 patients: 31 (4%) of the umeclidinium group patients and 57 (7%) of the placebo group patients (hazard ratio, 0.53).
The findings were similar in the patients who received off-label 125-mg dosing of umeclidinium, and the incidence of adverse events and serious adverse events was similar across treatment groups. Dr. Siler noted.
Data on the benefits of LAMAs in triple therapy in patients with moderate to very severe COPD are limited. This pooled analysis of data from four randomized, double-blind, parallel-group 12-week trials of once-daily add-on umeclidinium included COPD patients who entered a 4-week run-in on open-label ICS/LABA (either fluticasone furoate/vilanterol 100/25 mcg or fluticasone propionate/salmeterol 250/50 mcg), and who were then randomized to receive 62.5 or 125 mcg of umeclidinium or placebo.
GlaxoSmithKline funded the study.
AT ATS 2015
Key clinical point: Adding umeclidinium to fixed-dose inhaled corticosteroid/long-acting beta antagonist therapy might improve lung function and health-related quality of life in patients with chronic obstructive pulmonary disorder.
Major finding: Statistically significant improvements in FEV1 were seen vs. placebo, at day 85 (0.130 L) and at all other time points.
Data source: A post hoc analysis of data from four phase III trials comprising 2,458 patients.
Disclosures: GlaxoSmithKline funded the study.
ATS: Pulmonary rehab plus CPAP boosts sleep apnea outcomes
DENVER – Pulmonary rehabilitation plus continuous positive airway pressure therapy improved pulmonary function, compared with CPAP alone, in a randomized study of 40 patients with obstructive sleep apnea.
While CPAP remains the main treatment option, it doesn’t address the obesity and inactivity levels that can contribute to comorbidities associated with OSA, Katerina Neumonnova, Ph.D., reported during a press briefing at an international conference of the American Thoracic Society. Patients with OSA often describe respiratory symptoms associated with diminished functional capacity and decreased health-related quality of life. Adjunctive pulmonary rehabilitation might improve pulmonary function, allowing for increased activity levels that can lead to improved outcomes.
Obstructive sleep apnea (OSA) improved with CPAP in all patients in the study. However, percent predicted vital capacity increased from 96.4 to 103.2 in the 20 patients randomized to receive pulmonary rehabilitation and CPAP, and decreased from 99.5 to 94.8 in the 20 patients who received CPAP alone. Percent predicted forced expiratory volume in 1 second (FEV1) improved from 91.5 to 96.1 in the pulmonary rehabilitation plus CPAP group, and decreased from 92.1 to 86.3 in the CPAP-only group.
Those who received pulmonary rehabilitation and CPAP also had significantly greater reductions in body mass index and in neck, waist, and hip circumference, said Dr. Neumonnova of Palacky University, Olomouc, Czech Republic. Percentage of weight as body fat significantly decreased in both groups, but the reduction was greater in those who received pulmonary rehabilitation (from 39.4 to 36.9) than in those who got CPAP alone (from 40.1 to 39.1), she said.
The patients, 20 men and 20 women, mean age 54 years, had similar baseline apnea/hypopnea index (greater than 15), pulmonary function, BMI, percentage of body fat, and neck, waist, and hip circumferences.
The 6-week pulmonary rehabilitation program consisted of twice-weekly 60-minute individualized exercise training sessions, combined with education, breathing retraining, respiratory muscle training, and oropharyngeal exercises. Patients were encouraged to steadily increase their levels of activity, measured by steps per day.
Dr. Neumonnova reported having no disclosures.
DENVER – Pulmonary rehabilitation plus continuous positive airway pressure therapy improved pulmonary function, compared with CPAP alone, in a randomized study of 40 patients with obstructive sleep apnea.
While CPAP remains the main treatment option, it doesn’t address the obesity and inactivity levels that can contribute to comorbidities associated with OSA, Katerina Neumonnova, Ph.D., reported during a press briefing at an international conference of the American Thoracic Society. Patients with OSA often describe respiratory symptoms associated with diminished functional capacity and decreased health-related quality of life. Adjunctive pulmonary rehabilitation might improve pulmonary function, allowing for increased activity levels that can lead to improved outcomes.
Obstructive sleep apnea (OSA) improved with CPAP in all patients in the study. However, percent predicted vital capacity increased from 96.4 to 103.2 in the 20 patients randomized to receive pulmonary rehabilitation and CPAP, and decreased from 99.5 to 94.8 in the 20 patients who received CPAP alone. Percent predicted forced expiratory volume in 1 second (FEV1) improved from 91.5 to 96.1 in the pulmonary rehabilitation plus CPAP group, and decreased from 92.1 to 86.3 in the CPAP-only group.
Those who received pulmonary rehabilitation and CPAP also had significantly greater reductions in body mass index and in neck, waist, and hip circumference, said Dr. Neumonnova of Palacky University, Olomouc, Czech Republic. Percentage of weight as body fat significantly decreased in both groups, but the reduction was greater in those who received pulmonary rehabilitation (from 39.4 to 36.9) than in those who got CPAP alone (from 40.1 to 39.1), she said.
The patients, 20 men and 20 women, mean age 54 years, had similar baseline apnea/hypopnea index (greater than 15), pulmonary function, BMI, percentage of body fat, and neck, waist, and hip circumferences.
The 6-week pulmonary rehabilitation program consisted of twice-weekly 60-minute individualized exercise training sessions, combined with education, breathing retraining, respiratory muscle training, and oropharyngeal exercises. Patients were encouraged to steadily increase their levels of activity, measured by steps per day.
Dr. Neumonnova reported having no disclosures.
DENVER – Pulmonary rehabilitation plus continuous positive airway pressure therapy improved pulmonary function, compared with CPAP alone, in a randomized study of 40 patients with obstructive sleep apnea.
While CPAP remains the main treatment option, it doesn’t address the obesity and inactivity levels that can contribute to comorbidities associated with OSA, Katerina Neumonnova, Ph.D., reported during a press briefing at an international conference of the American Thoracic Society. Patients with OSA often describe respiratory symptoms associated with diminished functional capacity and decreased health-related quality of life. Adjunctive pulmonary rehabilitation might improve pulmonary function, allowing for increased activity levels that can lead to improved outcomes.
Obstructive sleep apnea (OSA) improved with CPAP in all patients in the study. However, percent predicted vital capacity increased from 96.4 to 103.2 in the 20 patients randomized to receive pulmonary rehabilitation and CPAP, and decreased from 99.5 to 94.8 in the 20 patients who received CPAP alone. Percent predicted forced expiratory volume in 1 second (FEV1) improved from 91.5 to 96.1 in the pulmonary rehabilitation plus CPAP group, and decreased from 92.1 to 86.3 in the CPAP-only group.
Those who received pulmonary rehabilitation and CPAP also had significantly greater reductions in body mass index and in neck, waist, and hip circumference, said Dr. Neumonnova of Palacky University, Olomouc, Czech Republic. Percentage of weight as body fat significantly decreased in both groups, but the reduction was greater in those who received pulmonary rehabilitation (from 39.4 to 36.9) than in those who got CPAP alone (from 40.1 to 39.1), she said.
The patients, 20 men and 20 women, mean age 54 years, had similar baseline apnea/hypopnea index (greater than 15), pulmonary function, BMI, percentage of body fat, and neck, waist, and hip circumferences.
The 6-week pulmonary rehabilitation program consisted of twice-weekly 60-minute individualized exercise training sessions, combined with education, breathing retraining, respiratory muscle training, and oropharyngeal exercises. Patients were encouraged to steadily increase their levels of activity, measured by steps per day.
Dr. Neumonnova reported having no disclosures.
AT ATS 2015
Key clinical point: Pulmonary rehabilitation could boost outcomes in patients undergoing CPAP treatment for OSA.
Major finding: Percent predicted vital capacity increased from 96.4 to 103.2 in the pulmonary rehabilitation and CPAP group, and decreased from 99.5 to 94.8 in the CPAP-only group.
Data source: A randomized study of 40 patients.
Disclosures: Dr. Neumonnova reported having no disclosures.
NIH panel identifies ME/CFS research gaps, priorities
Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) is very real, but the lack of a universally accepted definition of the complex, multifaceted condition is stifling progress with respect to optimizing patient care, according to a panel convened by the National Institutes of Health.
“A team of stakeholders (such as patients, clinicians, researchers, and federal agencies) should be assembled to reach a consensus on the definition and variables of ME/CFS. A national and international research network should be developed to clarify the case definition and advance the field,” the Pathways to Prevention Workshop: Advancing the Research on Myalgic Encephalomyelitis/Chronic Fatigue Syndrome panel stated in a position paper (Ann. Intern. Med. 2015 June 15 [doi:10.7326/M15-0338]).
The panel specifically recommended that the Oxford case definition of ME/CFS be retired, as it “may impair progress and cause harm, ” and it further recommended that “the ME/CFS community concur on a single case definition (even if it is not perfect), and patients, clinicians, and researchers agree on a definition for meaningful recovery.”
The ME/CFS definition recommendation is one of seven that emerged from the December 2014 workshop, which was cosponsored by the NIH Office of Disease Prevention and the Trans-NIH ME/CFS Research Working Group.
The workshop agenda was developed by a multidisciplinary working group, and discussion was facilitated by input from invited experts, public comment by various stakeholders, and by systematic evidence-based reviews on the diagnosis and treatment of ME/CFS that were conducted expressly for the workshop by the Pacific Northwest Evidence-based Practice Center, Portland, Ore., with the support of the Agency for Healthcare Research and Quality.
The diagnostic methods and treatment reviews also were published June 15 in the Annals of Internal Medicine.
The diagnostic methods review identified nine sets of clinical criteria for defining ME/CFS from articles published between 1998 and 2013, according to Dr. Elizabeth Haney of Oregon Health and Science University, Portland, and her colleagues.
The review authors noted that none of the nine methods – including one recently proposed by the Institute of Medicine that includes elements of the previous definitions and which also proposed changing the name of ME/CFS to systemic exertion intolerance disease – have been adequately tested to identify affected patients in the setting of diagnostic uncertainly (Ann. Intern. Med. 2015 June 15 [doi:10.7326/M15-0443]).
The treatment review identified trials showing a low to moderate benefit with rintatolimod, counseling therapies, and graded exercise therapy for some patients who met case definitions for ME/CFS but found that those treatments have not been adequately tested in broader patient populations and that evidence is insufficient for numerous other treatments, according to Dr. M.E. Beth Smith, also of Oregon Health and Science University, and her colleagues.
“More definitive studies are needed to fill these research gaps,” the authors wrote (Ann. Intern. Med. 2015 June 15 [doi:10.7326/M15-0114]).
During a press briefing on the workshop panel’s conclusions and recommendations, workshop and panel chair Dr. Carmen R. Green said that ME/CFS is a chronic, complex, multifaceted condition characterized by extreme fatigue, pain, impaired memory, sleep disturbance, and insomnia, which are not improved by rest.
The cause and pathogenesis of ME/CFS remains unknown, and no laboratory diagnostic tests or known cures exist, said Dr. Green of the University of Michigan Health System, Ann Arbor.
Panel member Dr. Kathleen M. O’Neil added that the panel heard of hundreds of combinations of nonspecific symptoms common to both ME/CFS and other diseases, and that very little agreement exists on the constellation of signs and symptoms associated with ME/CFS.
“There has been a lot of confusion, and there are a lot of well-intentioned, but eventually misguided, issues in assessing and treating these patients, who are so very needy,” said Dr. O’Neil of the University of Indiana, Indianapolis.
In its report, the panel also noted that about 1 million persons – mostly women– are affected, and that the syndrome represents an “unmet public health issue with an estimated economic burden between $2 billion and $7 billion in the United States.
“Limited knowledge and research funding create an additional burden for patients and health care providers. The research and health care community has frustrated its constituents by not appropriately assessing and treating the disease and by allowing patients to be stigmatized,” the panel wrote.
In addition to better defining ME/CFS, the panel had several other recommendations:
• Create new knowledge by investing in bench-to-bedside research for ME/CFS with an emphasis on developing biomarkers and diagnostic tests.
• Improve methods and measures to identify ME/CFS that “include the patient’s voice through patient-reported outcomes.”
• Provide training and education to clinicians, many of whom do not fully understand ME/CFS.
• Find new funding resources, such as through partnerships across institutions to advance the research and develop new scientists.
• Conduct clinical trials, and create a website for patient and clinician educational materials and information about clinical trials.
• Improve treatment.
“Patients should be active participants in care and decision-making,” the panel wrote, noting that the modest benefit from cognitive behavioral therapy should be studied as an adjunct to other methods, and that future treatment studies should evaluate multifaceted therapies focusing on biomedical and supportive care.
During the press briefing, Dr. Green added that, to develop primary prevention strategies and effective drug treatment, there needs to be a clear understanding of the condition’s cause and the population it affects.
“We hope the information provided in the panel report clearly sheds light on these issues and helps to facilitate positive results to improve the quality of the research and ultimately leads to better outcomes for the patients impacted,” she said.
The panel recommended that the NIH Office of Disease Prevention convene another expert panel in 5 years to monitor progress.
In an editorial, Dr. Anthony L. Komaroff of Brigham and Women’s Hospital, Boston, said the panel report, as well as the IOM report and the AHRQ reviews, “demonstrate how much we have learned about ME/CFS and how much we still do not know. We do not understand its pathogenesis, and we do not have a diagnostic test or a cure. However, these recent reports, summarizing information from more than 9000 articles, should put the question of whether ME/CFS is a ’real’ illness to rest.”
“When skeptical physicians, many of whom are unaware of this literature, tell patients with ME/CFS that ‘there is nothing wrong,’ they not only commit a diagnostic error: They also compound the patients’ suffering,” he wrote.
The systematic reviews were sponsored by the AHRQ. The workshop panel members reported having no disclosures. Dr. Komaroff also reported having no disclosures.
Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) is very real, but the lack of a universally accepted definition of the complex, multifaceted condition is stifling progress with respect to optimizing patient care, according to a panel convened by the National Institutes of Health.
“A team of stakeholders (such as patients, clinicians, researchers, and federal agencies) should be assembled to reach a consensus on the definition and variables of ME/CFS. A national and international research network should be developed to clarify the case definition and advance the field,” the Pathways to Prevention Workshop: Advancing the Research on Myalgic Encephalomyelitis/Chronic Fatigue Syndrome panel stated in a position paper (Ann. Intern. Med. 2015 June 15 [doi:10.7326/M15-0338]).
The panel specifically recommended that the Oxford case definition of ME/CFS be retired, as it “may impair progress and cause harm, ” and it further recommended that “the ME/CFS community concur on a single case definition (even if it is not perfect), and patients, clinicians, and researchers agree on a definition for meaningful recovery.”
The ME/CFS definition recommendation is one of seven that emerged from the December 2014 workshop, which was cosponsored by the NIH Office of Disease Prevention and the Trans-NIH ME/CFS Research Working Group.
The workshop agenda was developed by a multidisciplinary working group, and discussion was facilitated by input from invited experts, public comment by various stakeholders, and by systematic evidence-based reviews on the diagnosis and treatment of ME/CFS that were conducted expressly for the workshop by the Pacific Northwest Evidence-based Practice Center, Portland, Ore., with the support of the Agency for Healthcare Research and Quality.
The diagnostic methods and treatment reviews also were published June 15 in the Annals of Internal Medicine.
The diagnostic methods review identified nine sets of clinical criteria for defining ME/CFS from articles published between 1998 and 2013, according to Dr. Elizabeth Haney of Oregon Health and Science University, Portland, and her colleagues.
The review authors noted that none of the nine methods – including one recently proposed by the Institute of Medicine that includes elements of the previous definitions and which also proposed changing the name of ME/CFS to systemic exertion intolerance disease – have been adequately tested to identify affected patients in the setting of diagnostic uncertainly (Ann. Intern. Med. 2015 June 15 [doi:10.7326/M15-0443]).
The treatment review identified trials showing a low to moderate benefit with rintatolimod, counseling therapies, and graded exercise therapy for some patients who met case definitions for ME/CFS but found that those treatments have not been adequately tested in broader patient populations and that evidence is insufficient for numerous other treatments, according to Dr. M.E. Beth Smith, also of Oregon Health and Science University, and her colleagues.
“More definitive studies are needed to fill these research gaps,” the authors wrote (Ann. Intern. Med. 2015 June 15 [doi:10.7326/M15-0114]).
During a press briefing on the workshop panel’s conclusions and recommendations, workshop and panel chair Dr. Carmen R. Green said that ME/CFS is a chronic, complex, multifaceted condition characterized by extreme fatigue, pain, impaired memory, sleep disturbance, and insomnia, which are not improved by rest.
The cause and pathogenesis of ME/CFS remains unknown, and no laboratory diagnostic tests or known cures exist, said Dr. Green of the University of Michigan Health System, Ann Arbor.
Panel member Dr. Kathleen M. O’Neil added that the panel heard of hundreds of combinations of nonspecific symptoms common to both ME/CFS and other diseases, and that very little agreement exists on the constellation of signs and symptoms associated with ME/CFS.
“There has been a lot of confusion, and there are a lot of well-intentioned, but eventually misguided, issues in assessing and treating these patients, who are so very needy,” said Dr. O’Neil of the University of Indiana, Indianapolis.
In its report, the panel also noted that about 1 million persons – mostly women– are affected, and that the syndrome represents an “unmet public health issue with an estimated economic burden between $2 billion and $7 billion in the United States.
“Limited knowledge and research funding create an additional burden for patients and health care providers. The research and health care community has frustrated its constituents by not appropriately assessing and treating the disease and by allowing patients to be stigmatized,” the panel wrote.
In addition to better defining ME/CFS, the panel had several other recommendations:
• Create new knowledge by investing in bench-to-bedside research for ME/CFS with an emphasis on developing biomarkers and diagnostic tests.
• Improve methods and measures to identify ME/CFS that “include the patient’s voice through patient-reported outcomes.”
• Provide training and education to clinicians, many of whom do not fully understand ME/CFS.
• Find new funding resources, such as through partnerships across institutions to advance the research and develop new scientists.
• Conduct clinical trials, and create a website for patient and clinician educational materials and information about clinical trials.
• Improve treatment.
“Patients should be active participants in care and decision-making,” the panel wrote, noting that the modest benefit from cognitive behavioral therapy should be studied as an adjunct to other methods, and that future treatment studies should evaluate multifaceted therapies focusing on biomedical and supportive care.
During the press briefing, Dr. Green added that, to develop primary prevention strategies and effective drug treatment, there needs to be a clear understanding of the condition’s cause and the population it affects.
“We hope the information provided in the panel report clearly sheds light on these issues and helps to facilitate positive results to improve the quality of the research and ultimately leads to better outcomes for the patients impacted,” she said.
The panel recommended that the NIH Office of Disease Prevention convene another expert panel in 5 years to monitor progress.
In an editorial, Dr. Anthony L. Komaroff of Brigham and Women’s Hospital, Boston, said the panel report, as well as the IOM report and the AHRQ reviews, “demonstrate how much we have learned about ME/CFS and how much we still do not know. We do not understand its pathogenesis, and we do not have a diagnostic test or a cure. However, these recent reports, summarizing information from more than 9000 articles, should put the question of whether ME/CFS is a ’real’ illness to rest.”
“When skeptical physicians, many of whom are unaware of this literature, tell patients with ME/CFS that ‘there is nothing wrong,’ they not only commit a diagnostic error: They also compound the patients’ suffering,” he wrote.
The systematic reviews were sponsored by the AHRQ. The workshop panel members reported having no disclosures. Dr. Komaroff also reported having no disclosures.
Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) is very real, but the lack of a universally accepted definition of the complex, multifaceted condition is stifling progress with respect to optimizing patient care, according to a panel convened by the National Institutes of Health.
“A team of stakeholders (such as patients, clinicians, researchers, and federal agencies) should be assembled to reach a consensus on the definition and variables of ME/CFS. A national and international research network should be developed to clarify the case definition and advance the field,” the Pathways to Prevention Workshop: Advancing the Research on Myalgic Encephalomyelitis/Chronic Fatigue Syndrome panel stated in a position paper (Ann. Intern. Med. 2015 June 15 [doi:10.7326/M15-0338]).
The panel specifically recommended that the Oxford case definition of ME/CFS be retired, as it “may impair progress and cause harm, ” and it further recommended that “the ME/CFS community concur on a single case definition (even if it is not perfect), and patients, clinicians, and researchers agree on a definition for meaningful recovery.”
The ME/CFS definition recommendation is one of seven that emerged from the December 2014 workshop, which was cosponsored by the NIH Office of Disease Prevention and the Trans-NIH ME/CFS Research Working Group.
The workshop agenda was developed by a multidisciplinary working group, and discussion was facilitated by input from invited experts, public comment by various stakeholders, and by systematic evidence-based reviews on the diagnosis and treatment of ME/CFS that were conducted expressly for the workshop by the Pacific Northwest Evidence-based Practice Center, Portland, Ore., with the support of the Agency for Healthcare Research and Quality.
The diagnostic methods and treatment reviews also were published June 15 in the Annals of Internal Medicine.
The diagnostic methods review identified nine sets of clinical criteria for defining ME/CFS from articles published between 1998 and 2013, according to Dr. Elizabeth Haney of Oregon Health and Science University, Portland, and her colleagues.
The review authors noted that none of the nine methods – including one recently proposed by the Institute of Medicine that includes elements of the previous definitions and which also proposed changing the name of ME/CFS to systemic exertion intolerance disease – have been adequately tested to identify affected patients in the setting of diagnostic uncertainly (Ann. Intern. Med. 2015 June 15 [doi:10.7326/M15-0443]).
The treatment review identified trials showing a low to moderate benefit with rintatolimod, counseling therapies, and graded exercise therapy for some patients who met case definitions for ME/CFS but found that those treatments have not been adequately tested in broader patient populations and that evidence is insufficient for numerous other treatments, according to Dr. M.E. Beth Smith, also of Oregon Health and Science University, and her colleagues.
“More definitive studies are needed to fill these research gaps,” the authors wrote (Ann. Intern. Med. 2015 June 15 [doi:10.7326/M15-0114]).
During a press briefing on the workshop panel’s conclusions and recommendations, workshop and panel chair Dr. Carmen R. Green said that ME/CFS is a chronic, complex, multifaceted condition characterized by extreme fatigue, pain, impaired memory, sleep disturbance, and insomnia, which are not improved by rest.
The cause and pathogenesis of ME/CFS remains unknown, and no laboratory diagnostic tests or known cures exist, said Dr. Green of the University of Michigan Health System, Ann Arbor.
Panel member Dr. Kathleen M. O’Neil added that the panel heard of hundreds of combinations of nonspecific symptoms common to both ME/CFS and other diseases, and that very little agreement exists on the constellation of signs and symptoms associated with ME/CFS.
“There has been a lot of confusion, and there are a lot of well-intentioned, but eventually misguided, issues in assessing and treating these patients, who are so very needy,” said Dr. O’Neil of the University of Indiana, Indianapolis.
In its report, the panel also noted that about 1 million persons – mostly women– are affected, and that the syndrome represents an “unmet public health issue with an estimated economic burden between $2 billion and $7 billion in the United States.
“Limited knowledge and research funding create an additional burden for patients and health care providers. The research and health care community has frustrated its constituents by not appropriately assessing and treating the disease and by allowing patients to be stigmatized,” the panel wrote.
In addition to better defining ME/CFS, the panel had several other recommendations:
• Create new knowledge by investing in bench-to-bedside research for ME/CFS with an emphasis on developing biomarkers and diagnostic tests.
• Improve methods and measures to identify ME/CFS that “include the patient’s voice through patient-reported outcomes.”
• Provide training and education to clinicians, many of whom do not fully understand ME/CFS.
• Find new funding resources, such as through partnerships across institutions to advance the research and develop new scientists.
• Conduct clinical trials, and create a website for patient and clinician educational materials and information about clinical trials.
• Improve treatment.
“Patients should be active participants in care and decision-making,” the panel wrote, noting that the modest benefit from cognitive behavioral therapy should be studied as an adjunct to other methods, and that future treatment studies should evaluate multifaceted therapies focusing on biomedical and supportive care.
During the press briefing, Dr. Green added that, to develop primary prevention strategies and effective drug treatment, there needs to be a clear understanding of the condition’s cause and the population it affects.
“We hope the information provided in the panel report clearly sheds light on these issues and helps to facilitate positive results to improve the quality of the research and ultimately leads to better outcomes for the patients impacted,” she said.
The panel recommended that the NIH Office of Disease Prevention convene another expert panel in 5 years to monitor progress.
In an editorial, Dr. Anthony L. Komaroff of Brigham and Women’s Hospital, Boston, said the panel report, as well as the IOM report and the AHRQ reviews, “demonstrate how much we have learned about ME/CFS and how much we still do not know. We do not understand its pathogenesis, and we do not have a diagnostic test or a cure. However, these recent reports, summarizing information from more than 9000 articles, should put the question of whether ME/CFS is a ’real’ illness to rest.”
“When skeptical physicians, many of whom are unaware of this literature, tell patients with ME/CFS that ‘there is nothing wrong,’ they not only commit a diagnostic error: They also compound the patients’ suffering,” he wrote.
The systematic reviews were sponsored by the AHRQ. The workshop panel members reported having no disclosures. Dr. Komaroff also reported having no disclosures.
FROM ANNALS OF INTERNAL MEDICINE