User login
The challenges of managing CMV infection during pregnancy
CASE Anomalous findings on fetal anatomic survey
A 27-year-old previously healthy primigravid woman is at 18 weeks’ gestation. She is a first-grade schoolteacher. On her fetal anatomic survey, the estimated fetal weight was in the eighth percentile. Echogenic bowel and a small amount of ascitic fluid were noted in the fetal abdomen. The lateral and third ventricles were mildly dilated, the head circumference was 2 standard deviations below normal, and the placenta was slightly thickened and edematous.
What is the most likely diagnosis?
What diagnostic tests are indicated?
What management options are available for this patient?
Cytomegalovirus (CMV) is the most common of the perinatally transmitted infections, affecting 1% to 4% of all pregnancies. Although the virus typically causes either asymptomatic infection or only mild illness in immunocompetent individuals, it can cause life-threatening disease in immunocompromised persons and in the developing fetus. In this article, we review the virology and epidemiology of CMV infection and then focus on the key methods to diagnose infection in the mother and fetus. We conclude by considering measures that may be of at least modest value in treating CMV in pregnancy.
Virology of CMV infection
Cytomegalovirus is a double-stranded DNA virus in the Herpesviridae family. This ubiquitous virus is present in virtually all secretions and excretions of an infected host, including blood, urine, saliva, breast milk, genital secretions, and tissues and organs used for donation. Infection is transmitted through direct contact with any of the substances listed; contact with infected urine or saliva is the most common mode of transmission. Disease occurrence does not show seasonal variation.
After exposure, an incubation period of 28 to 60 days ensues, followed by development of viremia and clinical symptoms. In the majority of exposed individuals, CMV establishes a lifelong latent infection, and recurrent episodes of illness can occur as a result of reactivation of latent virus (also known as secondary infection) or, more rarely, infection with a new viral strain. In fact, most CMV illness episodes in pregnancy represent a reactivation of a previous infection rather than a new infection.
Following initial infection, both IgM (immunoglobulin M) and IgG (immunoglobulin G) antibodies develop rapidly and can be detected in blood within 1 to 2 weeks. IgM levels typically wane within 30 to 60 days, although persistence for several months is not unusual, and levels also can increase with viral reactivation (secondary infection). IgG antibodies typically persist for many years after a primary infection.
Intrauterine CMV infection occurs through hematogenous transplacental passage during maternal viremia. The risk of transmission and severity of fetal effects depend on whether or not the infection is primary or secondary in nature as well as the gestational age at fetal exposure.1,2
Additionally, postnatal vertical transmission can occur through exposure to viral particles in genital secretions as well as breast milk. CMV acquired in the postnatal period rarely produces severe sequelae in a healthy term neonate, but it has been associated with an increased rate of complications in very low birth weight and premature newborns.3
Continue to: Who is at risk...
Who is at risk
Congenital CMV, which occurs in 2.1 to 7.7 per 10,000 live births in the United States, is both the most common congenital infection and the leading cause of nonhereditary congenital hearing loss in children.4,5 The main reservoir of CMV in the United States is young children in day care settings, with approximately 50% of this population showing evidence of viral shedding in saliva.1 Adult populations in North America have a high prevalence of CMV IgG antibodies indicative of prior infection, with rates reaching 50% to 80%. Among seronegative individuals aged 12 to 49, the rate of seroconversion is approximately 1 in 60 annually.6 Significant racial disparities have been noted in rates of seroprevalence and seroconversion, with higher rates of infection in non-Hispanic Black and Mexican American individuals.6 Overall, the rate of new CMV infection among pregnant women in the United States is 0.7% to 4%.7
Clinical manifestations
Manifestations of infection differ depending on whether or not infection is primary or recurrent (secondary) and whether or not the host is immunocompetent or has a compromised immune system. Unique manifestations develop in the fetus.
CMV infection in children and adults. Among individuals with a normal immune response, the typical course of CMV is either no symptoms or a mononucleosis-like illness. In symptomatic patients, the most common symptoms include malaise, fever, and night sweats, and the most common associated laboratory abnormalities are elevation in liver function tests and a decreased white blood cell count, with a predominance of lymphocytes.8
Immunocompromised individuals are at risk for significant morbidity and mortality resulting from CMV. Illness may be the result of reactivation of latent infection due to decreased immune function or may be acquired as a result of treatment such as transplantation of CMV-positive organs or tissues, including bone marrow. Virtually any organ system can be affected, with potential for permanent organ damage and death. Severe systemic infection also can occur.
CMV infection in the fetus and neonate. As noted previously, fetal infection develops as a result of transplacental passage coincident with maternal infection. The risk of CMV transmission to the fetus and the severity of fetal injury vary based on gestational age at fetal infection and whether or not maternal infection is primary or secondary.
In most studies, primary maternal infections are associated with higher rates of fetal infection and more severe fetal and neonatal disease manifestations.2,7,9,10 Primary infections carry an overall 30% to 40% risk of transmission to the fetus.7,11 The risk of fetal transmission is much lower with a recurrent infection and is usually less than 2%.11 Due to their greater overall incidence, secondary infections account for the majority of cases of fetal and neonatal CMV disease.7 Importantly, although secondary infections generally have been regarded as having a lower risk and lower severity of fetal and neonatal disease, several recent studies have demonstrated rates of complications similar to, and even exceeding, those of primary infections.12-15 The TABLE provides a summary of the risks of fetal transmission and symptomatic fetal infection based on trimester of pregnancy.2,11,16-18
In the fetus, CMV may affect multiple organ systems. Among sonographic and magnetic resonance imaging (MRI) findings, central nervous system (CNS) anomalies are the most common.19,20 These can include microcephaly, ventriculomegaly, and periventricular calcifications. The gastrointestinal system also is frequently affected, and findings include echogenic bowel, hepatosplenomegaly, and liver calcifications. Lastly, isolated effusions, placentomegaly, fetal growth restriction, and even frank hydrops can develop. More favorable neurologic outcomes have been demonstrated in infants with no prenatal brain imaging abnormalities.20,21 However, the role of MRI in prenatal prognosis currently is not well defined.
FIGURE 1 illustrates selected sonographic findings associated with fetal CMV infection.
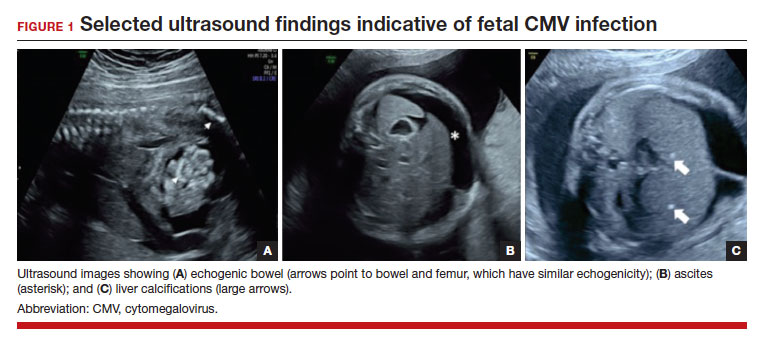
About 85% to 90% of infants with congenital CMV that results from primary maternal infection have no symptoms at birth. Among the 10% to 15% of infants that do have symptoms, petechial rash, jaundice, and hepatosplenomegaly are the most common manifestations (“blueberry muffin baby”). Approximately 10% to 20% of infants in this group have evidence of chorioretinitis on ophthalmologic examination, and 50% show either microcephaly or low birth weight.22Among survivors of symptomatic congenital CMV, more than 50% have long-term neurologic morbidities that may include sensorineural hearing loss, seizures, vision impairment, and developmental disabilities. Note that even when neonates appear asymptomatic at birth (regardless of whether infection is primary or secondary), 5% may develop microcephaly and motor deficits, 10% go on to develop sensorineural hearing loss, and the overall rate of neurologic morbidity reaches 13% to 15%.12,23 Some of the observed deficits manifest at several years of age, and, currently, no models exist for prediction of outcome.
Continue to: Diagnosing CMV infection...
Diagnosing CMV infection
Maternal infection
If maternal CMV infection is suspected based on a symptomatic illness or an abnormal fetal ultrasound exam, the first diagnostic test should be an assessment of IgM and IgG serology. If the former test results are positive and the latter negative, the diagnosis of acute CMV infection is confirmed. A positive serum CMV DNA polymerase chain reaction (PCR) test adds additional assurance that the diagnosis is correct. Primary infection, as noted above, poses the greatest risk of serious injury to the fetus.1
A frequent diagnostic dilemma arises when both the IgM and IgG antibody are positive. Remember that CMV IgM antibody can remain positive for 9 to 12 months after a primary infection and can reappear in the maternal serum in the face of a recurrent or reactivated infection. When confronted by both a positive IgM and positive IgG result, the clinician should then order IgG avidity testing. If the avidity is low to moderate, which reflects poor binding of antibody to the virus, the patient likely has an acute infection. If the avidity is high, which reflects enhanced binding of antibody to virus, the patient probably has a recurrent or reactivated infection; this scenario poses less danger to the developing fetus. The presence of CMV DNA in serum is also more consistent with acute infection, although viremia still can occur with recurrent infection. FIGURE 2 presents a suggested algorithm for the diagnosis of CMV in the pregnant patient.1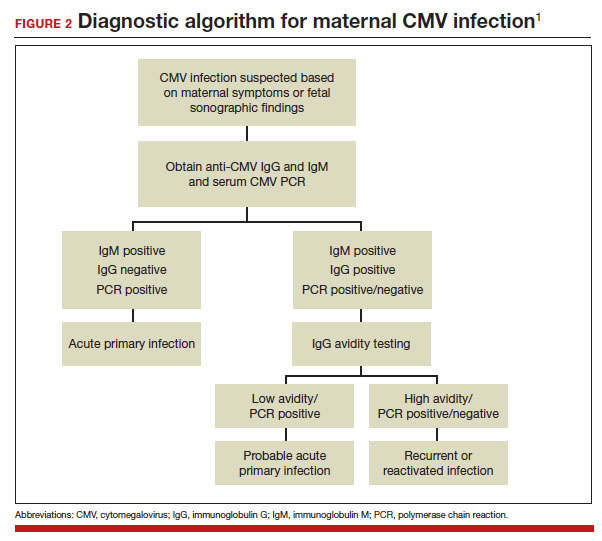
If a diagnosis of maternal CMV infection is confirmed, liver function tests should be obtained to determine if CMV hepatitis is present. If the liver function tests are abnormal, a coagulation profile also should be performed to identify the mother who might be at risk for peripartum hemorrhage.
Fetal infection
The single best test for confirmation of congenital CMV infection is detection of viral DNA and quantitation of viral load in the amniotic fluid by PCR. If the amniocentesis is performed prior to 20 weeks’ gestation and is negative, the test should be repeated in approximately 4 weeks.1,19,24
Detection of viral DNA indicates congenital infection. The ultimate task, however, is to determine if the infection has injured the fetus. Detailed ultrasound examination is the key to identifying fetal injury. As noted previously, the principal ultrasonographic findings that suggest congenital CMV infection include2,19,20,21,25:
- hydropic placenta
- fetal growth restriction
- microcephaly (head circumference more than 3 standard deviations below the mean)
- periventricular calcifications
- enlarged liver
- echogenic bowel
- ascites
- fetal hydrops.
Management: Evidence on CMV hyperimmune globulin, valacyclovir
If the immunocompetent mother has clinical manifestations of infection, she should receive symptomatic treatment. She should be encouraged to rest as much as possible, stay well hydrated, and use acetaminophen (1,000 mg every 6 to 8 hours) as needed for malaise and fever.
However, if the mother is immunocompromised and has signs of serious complications, such as chorioretinitis, hepatitis, or pneumonia, more aggressive therapy is indicated. Drugs used in this setting include foscarnet and ganciclovir and are best prescribed in consultation with a medical infectious disease specialist.
At this time, no consistently effective therapy for congenital infection is available. Therefore, if a patient has primary CMV infection in the first half of pregnancy, particularly in the first trimester, she should be counseled that the risk of fetal infection is approximately 40% and that approximately 5% to 15% of infants will be severely affected at birth. Given this information, some patients may opt for pregnancy termination.
In 2005, a report from Nigro and colleagues stimulated great hope that CMV-specific hyperimmune globulin (CytoGam) might be of value for both treatment and prophylaxis for congenital infection.26 These authors studied 157 women with confirmed primary CMV infection. One-hundred forty-eight women were asymptomatic and were identified by routine serologic screening, 8 had symptomatic infection, and 1 was identified because of abnormal fetal ultrasound findings. Forty-five women had CMV detected in amniotic fluid by PCR or culture more than 6 weeks before study enrollment. Thirty-one of these women were treated with intravenous hyperimmune globulin (200 U or 200 mg/kg maternal body weight); 14 declined treatment. Seven of the latter women had infants who were acutely symptomatic at the time of delivery; only 1 of the 31 treated women had an affected neonate (adjusted odds ratio [OR], 0.02; P<.001). In this same study, 84 women did not have a diagnostic amniocentesis because their infection occurred within 6 weeks of enrollment, their gestational age was less than 20 weeks, or they declined the procedure. Thirty-seven of these women received hyperimmune globulin (100 U or 100 mg/kg) every month until delivery, and 47 declined treatment. Six of the treated women delivered infected infants compared with 19 of the untreated women (adjusted OR, 0.32; P<.04).
Although these results were quite encouraging, several problems existed with the study’s design, as noted in an editorial that accompanied the study’s publication.27 First, the study was not randomized or placebo controlled. Second, patients were not stratified based on the severity of fetal ultrasound abnormalities. Third, the dosing of hyperimmune globulin varied; 9 of the 31 patients in the treatment group received additional infusions of drug into either the amniotic fluid or fetal umbilical vein. Moreover, patients in the prophylaxis group actually received a higher cumulative dose of hyperimmune globulin than patients in the treatment group.
Two subsequent investigations that were better designed were unable to verify the effectiveness of hyperimmune globulin. In 2014, Revello and colleagues reported the results of a prospective, randomized, placebo-controlled, double-blinded study of 124 women at 5 to 26 weeks’ gestation with confirmed primary CMV infection.28 The rate of congenital infection was 30% in the group treated with hyperimmune globulin and 44% in the placebo group (P=.13). There also was no significant difference in the concentration of serum CMV DNA in treated versus untreated mothers. Moreover, the number of adverse obstetric events (preterm delivery, fetal growth restriction, intrahepatic cholestasis of pregnancy, and postpartum preeclampsia) in the treatment group was higher than in the placebo group, 13% versus 2%.
In 2021, Hughes and colleagues published the results of a multicenter, double-blind trial in 399 women who had a diagnosis of primary CMV infection before 23 weeks’ gestation.29 The primary outcome was defined as a composite of congenital CMV infection or fetal/neonatal death. An adverse primary outcome occurred in 22.7% of the patients who received hyperimmune globulin and 19.4% of those who received placebo (relative risk, 1.17; 95% confidence interval [CI], 0.80–1.72; P=.42).
Continue to: Jacquemard and colleagues...
Jacquemard and colleagues then proposed a different approach.30 In a small pilot study of 20 patients, these authors used high doses of oral valacylovir (2 g 4 times daily) and documented therapeutic drug concentrations and a decline in CMV viral load in fetal serum. Patients were not stratified by severity of fetal injury at onset of treatment, so the authors were unable to define which fetuses were most likely to benefit from treatment.
In a follow-up investigation, Leruez-Ville and colleagues reported another small series in which high-dose oral valacyclovir (8 g daily) was used for treatment.31 They excluded fetuses with severe brain anomalies and fetuses with no sonographic evidence of injury. The median gestational age at diagnosis was 26 weeks. Thirty-four of 43 treated fetuses were free of injury at birth. In addition, the viral load in the neonate’s serum decreased significantly after treatment, and the platelet count increased. The authors then compared these outcomes to a historical cohort and confirmed that treatment increased the proportion of asymptomatic neonates from 43% without treatment to 82% with treatment (P<.05 with no overlapping confidence intervals).
We conclude from these investigations that hyperimmune globulin is unlikely to be of value in treating congenital CMV infection, especially if the fetus already has sonographic findings of severe injury. High-dose oral valacyclovir also is unlikely to be of value in severely affected fetuses, particularly those with evidence of CNS injury. However, antiviral therapy may be of modest value in situations when the fetus is less severely injured.
Preventive measures
Since no definitive treatment is available for congenital CMV infection, our efforts as clinicians should focus on measures that may prevent transmission of infection to the pregnant patient. These measures include:
- Encouraging patients to use careful handwashing techniques when handling infant diapers and toys.
- Encouraging patients to adopt safe sexual practices if not already engaged in a mutually faithful, monogamous relationship.
- Using CMV-negative blood when transfusing a pregnant woman or a fetus.
At the present time, unfortunately, a readily available and highly effective therapy for prevention of CMV infection is not available.
CASE Congenital infection diagnosed
The ultrasound findings are most consistent with congenital CMV infection, especially given the patient’s work as an elementary schoolteacher. The diagnosis of maternal infection is best established by conventional serology (positive IgM, negative IgM) and detection of viral DNA in maternal blood by PCR testing. The diagnosis of congenital infection is best confirmed by documentation of viral DNA in the amniotic fluid by PCR testing. Given that this fetus already has evidence of moderate to severe injury, no treatment is likely to be effective in reversing the abnormal ultrasound findings. Pregnancy termination may be an option, depending upon the patient’s desires and the legal restrictions prevalent in the patient’s geographic area. ●
- Cytomegalovirus infection is the most common of the perinatally transmitted infections.
- Maternal infection is often asymptomatic. When symptoms are present, they resemble those of an influenza-like illness. In immunocompromised persons, however, CMV may cause serious complications, including pneumonia, hepatitis, and chorioretinitis.
- The virus is transmitted by contact with contaminated body fluids, such as saliva, urine, blood, and genital secretions.
- The greatest risk of severe fetal injury results from primary maternal infection in the first trimester of pregnancy.
- Manifestations of severe congenital CMV infection include growth restriction, microcephaly, ventriculomegaly, hepatosplenomegaly, ascites, chorioretinitis, thrombocytopenia, purpura, and hydrops (“blueberry muffin baby”).
- Late manifestations of infection, which usually follow recurrent maternal infection, may appear as a child enters elementary school and include visual and auditory deficits, developmental delays, and learning disabilities.
- The diagnosis of maternal infection is confirmed by serology and detection of viral DNA in the serum by PCR testing.
- The diagnosis of fetal infection is best made by a combination of abnormal ultrasound findings and detection of CMV DNA in amniotic fluid. The characteristic ultrasound findings include placentomegaly, microcephaly, ventriculomegaly, growth restriction, echogenic bowel, and serous effusions/hydrops.
- Treatment of the mother with antiviral medications such as valacyclovir may be of modest value in reducing placental edema, decreasing viral load in the fetus, and hastening the resolution of some ultrasound findings, such as echogenic bowel.
- While initial studies seemed promising, the use of hyperimmune globulin has not proven to be consistently effective in treating congenital infection.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TR, et al, eds. Creasy and Resnik’s Maternal Fetal Medicine: Principles and Practice. 8th ed. 2019:888-890.
- Chatzakis C, Ville Y, Makrydimas G, et al. Timing of primary maternal cytomegalovirus infection and rates of vertical transmission and fetal consequences. Am J Obstet Gynecol. 2020;223:870-883.e11. doi:10.1016/j.ajog.2020.05.038
- Kelly MS, Benjamin DK, Puopolo KM, et al. Postnatal cytomegalovirus infection and the risk for bronchopulmonary dysplasia. JAMA Pediatr. 2015;169:e153785. doi:10.1001 /jamapediatrics.2015.3785
- Messinger CJ, Lipsitch M, Bateman BT, et al. Association between congenital cytomegalovirus and the prevalence at birth of microcephaly in the United States. JAMA Pediatr. 2020;174:1159-1167. doi:10.1001/jamapediatrics.2020.3009
- De Cuyper E, Acke F, Keymeulen A, et al. Risk factors for hearing loss at birth in newborns with congenital cytomegalovirus infection. JAMA Otolaryngol Head Neck Surg. 2023;149:122-130. doi:10.1001/jamaoto.2022.4109
- Colugnati FA, Staras SA, Dollard SC, et al. Incidence of cytomegalovirus infection among the general population and pregnant women in the United States. BMC Infect Dis. 2007;7:71. doi:10.1186/1471-2334-7-71
- Stagno S, Pass RF, Cloud G, et al. Primary cytomegalovirus infection in pregnancy. Incidence, transmission to fetus, and clinical outcome. JAMA. 1986;256:1904-1908.
- Wreghitt TG, Teare EL, Sule O, et al. Cytomegalovirus infection in immunocompetent patients. Clin Infect Dis. 2003;37:1603-1606. doi:10.1086/379711
- Fowler KB, Stagno S, Pass RF, et al. The outcome of congenital cytomegalovirus infection in relation to maternal antibody status. N Engl J Med. 1992;326:663-667. doi:10.1056 /NEJM199203053261003
- Faure-Bardon V, Magny JF, Parodi M, et al. Sequelae of congenital cytomegalovirus following maternal primary infections are limited to those acquired in the first trimester of pregnancy. Clin Infect Dis. 2019;69:1526-1532. doi:10.1093/ cid/ciy1128
- Kenneson A, Cannon MJ. Review and meta-analysis of the epidemiology of congenital cytomegalovirus (CMV) infection. Rev Med Virol. 2007;17:253-276. doi:10.1002/ rmv.535
- Boppana SB, Pass RF, Britt WJ, et al. Symptomatic congenital cytomegalovirus infection: neonatal morbidity and mortality. Pediatr Infect Dis J. 1992;11:93-99. doi:10.1097/00006454-199202000-00007
- Ross SA, Fowler KB, Ashrith G, et al. Hearing loss in children with congenital cytomegalovirus infection born to mothers with preexisting immunity. J Pediatr. 2006;148:332-336. doi:10.1016/j.jpeds.2005.09.003
- Zalel Y, Gilboa Y, Berkenshtat M, et al. Secondary cytomegalovirus infection can cause severe fetal sequelae despite maternal preconceptional immunity. Ultrasound Obstet Gynecol. 31:417-420. doi:10.1002/uog.5255
- Scaramuzzino F, Di Pastena M, Chiurchiu S, et al. Secondary cytomegalovirus infections: how much do we still not know? Comparison of children with symptomatic congenital cytomegalovirus born to mothers with primary and secondary infection. Front Pediatr. 2022;10:885926. doi:10.3389/fped.2022.885926
- Gindes L, Teperberg-Oikawa M, Sherman D, et al. Congenital cytomegalovirus infection following primary maternal infection in the third trimester. BJOG. 2008;115:830-835. doi:10.1111/j.1471-0528.2007.01651.x
- Hadar E, Dorfman E, Bardin R, et al. Symptomatic congenital cytomegalovirus disease following non-primary maternal infection: a retrospective cohort study. BMC Infect Dis. 2017;17:31. doi:10.1186/s12879-016-2161-3
- Elkan Miller T, Weisz B, Yinon Y, et al. Congenital cytomegalovirus infection following second and third trimester maternal infection is associated with mild childhood adverse outcome not predicted by prenatal imaging. J Pediatric Infect Dis Soc. 2021;10:562-568. doi:10.1093/jpids/ piaa154
- Lipitz S, Yinon Y, Malinger G, et al. Risk of cytomegalovirusassociated sequelae in relation to time of infection and findings on prenatal imaging. Ultrasound Obstet Gynecol. 2013;41:508-514. doi:10.1002/uog.12377
- Lipitz S, Elkan Miller T, Yinon Y, et al. Revisiting short- and long-term outcome after fetal first-trimester primary cytomegalovirus infection in relation to prenatal imaging findings. Ultrasound Obstet Gynecol. 2020;56:572-578. doi:10.1002/uog.21946
- Buca D, Di Mascio D, Rizzo G, et al. Outcome of fetuses with congenital cytomegalovirus infection and normal ultrasound at diagnosis: systematic review and meta-analysis. Ultrasound Obstet Gynecol. 2021;57:551-559. doi:10.1002/uog.23143
- Boppana SB, Ross SA, Fowler KB. Congenital cytomegalovirus infection: clinical outcome. Clin Infect Dis. 2013;57 (suppl 4):S178-S181. doi:10.1093/cid/cit629
- Dollard SC, Grosse SD, Ross DS. New estimates of the prevalence of neurological and sensory sequelae and mortality associated with congenital cytomegalovirus infection. Rev Med Virol. 2007;17:355-363. doi:10.1002/rmv.544
- Hughes BL, Gyamfi-Bannerman C. Diagnosis and antenatal management of congenital cytomegalovirus infection. Am J Obstet Gynecol. 2016;214:B5-11. doi:10.1016 /j.ajog.2016.02.042
- Rouse DJ, Fette LM, Hughes BL, et al. Noninvasive prediction of congenital cytomegalovirus infection after maternal primary infection. Obstet Gynecol. 2022;139:400-406. doi:10.1097/AOG.0000000000004691
- Nigro G, Adler SP, La Torre R, et al; Congenital Cytomegalovirus Collaborating Group. Passive immunization during pregnancy for congenital cytomegalovirus infection. N Engl J Med. 2005;353:1350-1362. doi:10.1056/NEJMoa043337
- Duff P. Immunotherapy for congenital cytomegalovirus infection. N Engl J Med. 2005;355:1402-1404. doi:10.1056 /NEJMe058172
- Revello MG, Lazzarotto T, Guerra B, et al. A randomized trial of hyperimmune globulin to prevent congenital cytomegalovirus. N Engl J Med. 2014;370:1316-1326. doi:10.1056/NEJMoa1310214
- Hughes BL, Clifton RG, Rouse DJ, et al. A trial of hyperimmune globulin to prevent congenital cytomegalovirus infection. N Engl J Med. 2021;385:436-444. doi:10.1056/NEJMoa1913569
- Jacquemard F, Yamamoto M, Costa JM, et al. Maternal administration of valaciclovir in symptomatic intrauterine cytomegalovirus infection. BJOG. 2007;114:1113-1121. doi:10.1111/j.1471-0528.2007.01308.x
- Leruez-Ville M, Ghout I, Bussières L, et al. In utero treatment of congenital cytomegalovirus infection with valacyclovir in a multicenter, open-label, phase II study. Am J Obstet Gynecol. 2016;215:462.e1-462.e10. doi:10.1016/j.ajog.2016.04.003
CASE Anomalous findings on fetal anatomic survey
A 27-year-old previously healthy primigravid woman is at 18 weeks’ gestation. She is a first-grade schoolteacher. On her fetal anatomic survey, the estimated fetal weight was in the eighth percentile. Echogenic bowel and a small amount of ascitic fluid were noted in the fetal abdomen. The lateral and third ventricles were mildly dilated, the head circumference was 2 standard deviations below normal, and the placenta was slightly thickened and edematous.
What is the most likely diagnosis?
What diagnostic tests are indicated?
What management options are available for this patient?
Cytomegalovirus (CMV) is the most common of the perinatally transmitted infections, affecting 1% to 4% of all pregnancies. Although the virus typically causes either asymptomatic infection or only mild illness in immunocompetent individuals, it can cause life-threatening disease in immunocompromised persons and in the developing fetus. In this article, we review the virology and epidemiology of CMV infection and then focus on the key methods to diagnose infection in the mother and fetus. We conclude by considering measures that may be of at least modest value in treating CMV in pregnancy.
Virology of CMV infection
Cytomegalovirus is a double-stranded DNA virus in the Herpesviridae family. This ubiquitous virus is present in virtually all secretions and excretions of an infected host, including blood, urine, saliva, breast milk, genital secretions, and tissues and organs used for donation. Infection is transmitted through direct contact with any of the substances listed; contact with infected urine or saliva is the most common mode of transmission. Disease occurrence does not show seasonal variation.
After exposure, an incubation period of 28 to 60 days ensues, followed by development of viremia and clinical symptoms. In the majority of exposed individuals, CMV establishes a lifelong latent infection, and recurrent episodes of illness can occur as a result of reactivation of latent virus (also known as secondary infection) or, more rarely, infection with a new viral strain. In fact, most CMV illness episodes in pregnancy represent a reactivation of a previous infection rather than a new infection.
Following initial infection, both IgM (immunoglobulin M) and IgG (immunoglobulin G) antibodies develop rapidly and can be detected in blood within 1 to 2 weeks. IgM levels typically wane within 30 to 60 days, although persistence for several months is not unusual, and levels also can increase with viral reactivation (secondary infection). IgG antibodies typically persist for many years after a primary infection.
Intrauterine CMV infection occurs through hematogenous transplacental passage during maternal viremia. The risk of transmission and severity of fetal effects depend on whether or not the infection is primary or secondary in nature as well as the gestational age at fetal exposure.1,2
Additionally, postnatal vertical transmission can occur through exposure to viral particles in genital secretions as well as breast milk. CMV acquired in the postnatal period rarely produces severe sequelae in a healthy term neonate, but it has been associated with an increased rate of complications in very low birth weight and premature newborns.3
Continue to: Who is at risk...
Who is at risk
Congenital CMV, which occurs in 2.1 to 7.7 per 10,000 live births in the United States, is both the most common congenital infection and the leading cause of nonhereditary congenital hearing loss in children.4,5 The main reservoir of CMV in the United States is young children in day care settings, with approximately 50% of this population showing evidence of viral shedding in saliva.1 Adult populations in North America have a high prevalence of CMV IgG antibodies indicative of prior infection, with rates reaching 50% to 80%. Among seronegative individuals aged 12 to 49, the rate of seroconversion is approximately 1 in 60 annually.6 Significant racial disparities have been noted in rates of seroprevalence and seroconversion, with higher rates of infection in non-Hispanic Black and Mexican American individuals.6 Overall, the rate of new CMV infection among pregnant women in the United States is 0.7% to 4%.7
Clinical manifestations
Manifestations of infection differ depending on whether or not infection is primary or recurrent (secondary) and whether or not the host is immunocompetent or has a compromised immune system. Unique manifestations develop in the fetus.
CMV infection in children and adults. Among individuals with a normal immune response, the typical course of CMV is either no symptoms or a mononucleosis-like illness. In symptomatic patients, the most common symptoms include malaise, fever, and night sweats, and the most common associated laboratory abnormalities are elevation in liver function tests and a decreased white blood cell count, with a predominance of lymphocytes.8
Immunocompromised individuals are at risk for significant morbidity and mortality resulting from CMV. Illness may be the result of reactivation of latent infection due to decreased immune function or may be acquired as a result of treatment such as transplantation of CMV-positive organs or tissues, including bone marrow. Virtually any organ system can be affected, with potential for permanent organ damage and death. Severe systemic infection also can occur.
CMV infection in the fetus and neonate. As noted previously, fetal infection develops as a result of transplacental passage coincident with maternal infection. The risk of CMV transmission to the fetus and the severity of fetal injury vary based on gestational age at fetal infection and whether or not maternal infection is primary or secondary.
In most studies, primary maternal infections are associated with higher rates of fetal infection and more severe fetal and neonatal disease manifestations.2,7,9,10 Primary infections carry an overall 30% to 40% risk of transmission to the fetus.7,11 The risk of fetal transmission is much lower with a recurrent infection and is usually less than 2%.11 Due to their greater overall incidence, secondary infections account for the majority of cases of fetal and neonatal CMV disease.7 Importantly, although secondary infections generally have been regarded as having a lower risk and lower severity of fetal and neonatal disease, several recent studies have demonstrated rates of complications similar to, and even exceeding, those of primary infections.12-15 The TABLE provides a summary of the risks of fetal transmission and symptomatic fetal infection based on trimester of pregnancy.2,11,16-18
In the fetus, CMV may affect multiple organ systems. Among sonographic and magnetic resonance imaging (MRI) findings, central nervous system (CNS) anomalies are the most common.19,20 These can include microcephaly, ventriculomegaly, and periventricular calcifications. The gastrointestinal system also is frequently affected, and findings include echogenic bowel, hepatosplenomegaly, and liver calcifications. Lastly, isolated effusions, placentomegaly, fetal growth restriction, and even frank hydrops can develop. More favorable neurologic outcomes have been demonstrated in infants with no prenatal brain imaging abnormalities.20,21 However, the role of MRI in prenatal prognosis currently is not well defined.
FIGURE 1 illustrates selected sonographic findings associated with fetal CMV infection.

About 85% to 90% of infants with congenital CMV that results from primary maternal infection have no symptoms at birth. Among the 10% to 15% of infants that do have symptoms, petechial rash, jaundice, and hepatosplenomegaly are the most common manifestations (“blueberry muffin baby”). Approximately 10% to 20% of infants in this group have evidence of chorioretinitis on ophthalmologic examination, and 50% show either microcephaly or low birth weight.22Among survivors of symptomatic congenital CMV, more than 50% have long-term neurologic morbidities that may include sensorineural hearing loss, seizures, vision impairment, and developmental disabilities. Note that even when neonates appear asymptomatic at birth (regardless of whether infection is primary or secondary), 5% may develop microcephaly and motor deficits, 10% go on to develop sensorineural hearing loss, and the overall rate of neurologic morbidity reaches 13% to 15%.12,23 Some of the observed deficits manifest at several years of age, and, currently, no models exist for prediction of outcome.
Continue to: Diagnosing CMV infection...
Diagnosing CMV infection
Maternal infection
If maternal CMV infection is suspected based on a symptomatic illness or an abnormal fetal ultrasound exam, the first diagnostic test should be an assessment of IgM and IgG serology. If the former test results are positive and the latter negative, the diagnosis of acute CMV infection is confirmed. A positive serum CMV DNA polymerase chain reaction (PCR) test adds additional assurance that the diagnosis is correct. Primary infection, as noted above, poses the greatest risk of serious injury to the fetus.1
A frequent diagnostic dilemma arises when both the IgM and IgG antibody are positive. Remember that CMV IgM antibody can remain positive for 9 to 12 months after a primary infection and can reappear in the maternal serum in the face of a recurrent or reactivated infection. When confronted by both a positive IgM and positive IgG result, the clinician should then order IgG avidity testing. If the avidity is low to moderate, which reflects poor binding of antibody to the virus, the patient likely has an acute infection. If the avidity is high, which reflects enhanced binding of antibody to virus, the patient probably has a recurrent or reactivated infection; this scenario poses less danger to the developing fetus. The presence of CMV DNA in serum is also more consistent with acute infection, although viremia still can occur with recurrent infection. FIGURE 2 presents a suggested algorithm for the diagnosis of CMV in the pregnant patient.1
If a diagnosis of maternal CMV infection is confirmed, liver function tests should be obtained to determine if CMV hepatitis is present. If the liver function tests are abnormal, a coagulation profile also should be performed to identify the mother who might be at risk for peripartum hemorrhage.
Fetal infection
The single best test for confirmation of congenital CMV infection is detection of viral DNA and quantitation of viral load in the amniotic fluid by PCR. If the amniocentesis is performed prior to 20 weeks’ gestation and is negative, the test should be repeated in approximately 4 weeks.1,19,24
Detection of viral DNA indicates congenital infection. The ultimate task, however, is to determine if the infection has injured the fetus. Detailed ultrasound examination is the key to identifying fetal injury. As noted previously, the principal ultrasonographic findings that suggest congenital CMV infection include2,19,20,21,25:
- hydropic placenta
- fetal growth restriction
- microcephaly (head circumference more than 3 standard deviations below the mean)
- periventricular calcifications
- enlarged liver
- echogenic bowel
- ascites
- fetal hydrops.
Management: Evidence on CMV hyperimmune globulin, valacyclovir
If the immunocompetent mother has clinical manifestations of infection, she should receive symptomatic treatment. She should be encouraged to rest as much as possible, stay well hydrated, and use acetaminophen (1,000 mg every 6 to 8 hours) as needed for malaise and fever.
However, if the mother is immunocompromised and has signs of serious complications, such as chorioretinitis, hepatitis, or pneumonia, more aggressive therapy is indicated. Drugs used in this setting include foscarnet and ganciclovir and are best prescribed in consultation with a medical infectious disease specialist.
At this time, no consistently effective therapy for congenital infection is available. Therefore, if a patient has primary CMV infection in the first half of pregnancy, particularly in the first trimester, she should be counseled that the risk of fetal infection is approximately 40% and that approximately 5% to 15% of infants will be severely affected at birth. Given this information, some patients may opt for pregnancy termination.
In 2005, a report from Nigro and colleagues stimulated great hope that CMV-specific hyperimmune globulin (CytoGam) might be of value for both treatment and prophylaxis for congenital infection.26 These authors studied 157 women with confirmed primary CMV infection. One-hundred forty-eight women were asymptomatic and were identified by routine serologic screening, 8 had symptomatic infection, and 1 was identified because of abnormal fetal ultrasound findings. Forty-five women had CMV detected in amniotic fluid by PCR or culture more than 6 weeks before study enrollment. Thirty-one of these women were treated with intravenous hyperimmune globulin (200 U or 200 mg/kg maternal body weight); 14 declined treatment. Seven of the latter women had infants who were acutely symptomatic at the time of delivery; only 1 of the 31 treated women had an affected neonate (adjusted odds ratio [OR], 0.02; P<.001). In this same study, 84 women did not have a diagnostic amniocentesis because their infection occurred within 6 weeks of enrollment, their gestational age was less than 20 weeks, or they declined the procedure. Thirty-seven of these women received hyperimmune globulin (100 U or 100 mg/kg) every month until delivery, and 47 declined treatment. Six of the treated women delivered infected infants compared with 19 of the untreated women (adjusted OR, 0.32; P<.04).
Although these results were quite encouraging, several problems existed with the study’s design, as noted in an editorial that accompanied the study’s publication.27 First, the study was not randomized or placebo controlled. Second, patients were not stratified based on the severity of fetal ultrasound abnormalities. Third, the dosing of hyperimmune globulin varied; 9 of the 31 patients in the treatment group received additional infusions of drug into either the amniotic fluid or fetal umbilical vein. Moreover, patients in the prophylaxis group actually received a higher cumulative dose of hyperimmune globulin than patients in the treatment group.
Two subsequent investigations that were better designed were unable to verify the effectiveness of hyperimmune globulin. In 2014, Revello and colleagues reported the results of a prospective, randomized, placebo-controlled, double-blinded study of 124 women at 5 to 26 weeks’ gestation with confirmed primary CMV infection.28 The rate of congenital infection was 30% in the group treated with hyperimmune globulin and 44% in the placebo group (P=.13). There also was no significant difference in the concentration of serum CMV DNA in treated versus untreated mothers. Moreover, the number of adverse obstetric events (preterm delivery, fetal growth restriction, intrahepatic cholestasis of pregnancy, and postpartum preeclampsia) in the treatment group was higher than in the placebo group, 13% versus 2%.
In 2021, Hughes and colleagues published the results of a multicenter, double-blind trial in 399 women who had a diagnosis of primary CMV infection before 23 weeks’ gestation.29 The primary outcome was defined as a composite of congenital CMV infection or fetal/neonatal death. An adverse primary outcome occurred in 22.7% of the patients who received hyperimmune globulin and 19.4% of those who received placebo (relative risk, 1.17; 95% confidence interval [CI], 0.80–1.72; P=.42).
Continue to: Jacquemard and colleagues...
Jacquemard and colleagues then proposed a different approach.30 In a small pilot study of 20 patients, these authors used high doses of oral valacylovir (2 g 4 times daily) and documented therapeutic drug concentrations and a decline in CMV viral load in fetal serum. Patients were not stratified by severity of fetal injury at onset of treatment, so the authors were unable to define which fetuses were most likely to benefit from treatment.
In a follow-up investigation, Leruez-Ville and colleagues reported another small series in which high-dose oral valacyclovir (8 g daily) was used for treatment.31 They excluded fetuses with severe brain anomalies and fetuses with no sonographic evidence of injury. The median gestational age at diagnosis was 26 weeks. Thirty-four of 43 treated fetuses were free of injury at birth. In addition, the viral load in the neonate’s serum decreased significantly after treatment, and the platelet count increased. The authors then compared these outcomes to a historical cohort and confirmed that treatment increased the proportion of asymptomatic neonates from 43% without treatment to 82% with treatment (P<.05 with no overlapping confidence intervals).
We conclude from these investigations that hyperimmune globulin is unlikely to be of value in treating congenital CMV infection, especially if the fetus already has sonographic findings of severe injury. High-dose oral valacyclovir also is unlikely to be of value in severely affected fetuses, particularly those with evidence of CNS injury. However, antiviral therapy may be of modest value in situations when the fetus is less severely injured.
Preventive measures
Since no definitive treatment is available for congenital CMV infection, our efforts as clinicians should focus on measures that may prevent transmission of infection to the pregnant patient. These measures include:
- Encouraging patients to use careful handwashing techniques when handling infant diapers and toys.
- Encouraging patients to adopt safe sexual practices if not already engaged in a mutually faithful, monogamous relationship.
- Using CMV-negative blood when transfusing a pregnant woman or a fetus.
At the present time, unfortunately, a readily available and highly effective therapy for prevention of CMV infection is not available.
CASE Congenital infection diagnosed
The ultrasound findings are most consistent with congenital CMV infection, especially given the patient’s work as an elementary schoolteacher. The diagnosis of maternal infection is best established by conventional serology (positive IgM, negative IgM) and detection of viral DNA in maternal blood by PCR testing. The diagnosis of congenital infection is best confirmed by documentation of viral DNA in the amniotic fluid by PCR testing. Given that this fetus already has evidence of moderate to severe injury, no treatment is likely to be effective in reversing the abnormal ultrasound findings. Pregnancy termination may be an option, depending upon the patient’s desires and the legal restrictions prevalent in the patient’s geographic area. ●
- Cytomegalovirus infection is the most common of the perinatally transmitted infections.
- Maternal infection is often asymptomatic. When symptoms are present, they resemble those of an influenza-like illness. In immunocompromised persons, however, CMV may cause serious complications, including pneumonia, hepatitis, and chorioretinitis.
- The virus is transmitted by contact with contaminated body fluids, such as saliva, urine, blood, and genital secretions.
- The greatest risk of severe fetal injury results from primary maternal infection in the first trimester of pregnancy.
- Manifestations of severe congenital CMV infection include growth restriction, microcephaly, ventriculomegaly, hepatosplenomegaly, ascites, chorioretinitis, thrombocytopenia, purpura, and hydrops (“blueberry muffin baby”).
- Late manifestations of infection, which usually follow recurrent maternal infection, may appear as a child enters elementary school and include visual and auditory deficits, developmental delays, and learning disabilities.
- The diagnosis of maternal infection is confirmed by serology and detection of viral DNA in the serum by PCR testing.
- The diagnosis of fetal infection is best made by a combination of abnormal ultrasound findings and detection of CMV DNA in amniotic fluid. The characteristic ultrasound findings include placentomegaly, microcephaly, ventriculomegaly, growth restriction, echogenic bowel, and serous effusions/hydrops.
- Treatment of the mother with antiviral medications such as valacyclovir may be of modest value in reducing placental edema, decreasing viral load in the fetus, and hastening the resolution of some ultrasound findings, such as echogenic bowel.
- While initial studies seemed promising, the use of hyperimmune globulin has not proven to be consistently effective in treating congenital infection.
CASE Anomalous findings on fetal anatomic survey
A 27-year-old previously healthy primigravid woman is at 18 weeks’ gestation. She is a first-grade schoolteacher. On her fetal anatomic survey, the estimated fetal weight was in the eighth percentile. Echogenic bowel and a small amount of ascitic fluid were noted in the fetal abdomen. The lateral and third ventricles were mildly dilated, the head circumference was 2 standard deviations below normal, and the placenta was slightly thickened and edematous.
What is the most likely diagnosis?
What diagnostic tests are indicated?
What management options are available for this patient?
Cytomegalovirus (CMV) is the most common of the perinatally transmitted infections, affecting 1% to 4% of all pregnancies. Although the virus typically causes either asymptomatic infection or only mild illness in immunocompetent individuals, it can cause life-threatening disease in immunocompromised persons and in the developing fetus. In this article, we review the virology and epidemiology of CMV infection and then focus on the key methods to diagnose infection in the mother and fetus. We conclude by considering measures that may be of at least modest value in treating CMV in pregnancy.
Virology of CMV infection
Cytomegalovirus is a double-stranded DNA virus in the Herpesviridae family. This ubiquitous virus is present in virtually all secretions and excretions of an infected host, including blood, urine, saliva, breast milk, genital secretions, and tissues and organs used for donation. Infection is transmitted through direct contact with any of the substances listed; contact with infected urine or saliva is the most common mode of transmission. Disease occurrence does not show seasonal variation.
After exposure, an incubation period of 28 to 60 days ensues, followed by development of viremia and clinical symptoms. In the majority of exposed individuals, CMV establishes a lifelong latent infection, and recurrent episodes of illness can occur as a result of reactivation of latent virus (also known as secondary infection) or, more rarely, infection with a new viral strain. In fact, most CMV illness episodes in pregnancy represent a reactivation of a previous infection rather than a new infection.
Following initial infection, both IgM (immunoglobulin M) and IgG (immunoglobulin G) antibodies develop rapidly and can be detected in blood within 1 to 2 weeks. IgM levels typically wane within 30 to 60 days, although persistence for several months is not unusual, and levels also can increase with viral reactivation (secondary infection). IgG antibodies typically persist for many years after a primary infection.
Intrauterine CMV infection occurs through hematogenous transplacental passage during maternal viremia. The risk of transmission and severity of fetal effects depend on whether or not the infection is primary or secondary in nature as well as the gestational age at fetal exposure.1,2
Additionally, postnatal vertical transmission can occur through exposure to viral particles in genital secretions as well as breast milk. CMV acquired in the postnatal period rarely produces severe sequelae in a healthy term neonate, but it has been associated with an increased rate of complications in very low birth weight and premature newborns.3
Continue to: Who is at risk...
Who is at risk
Congenital CMV, which occurs in 2.1 to 7.7 per 10,000 live births in the United States, is both the most common congenital infection and the leading cause of nonhereditary congenital hearing loss in children.4,5 The main reservoir of CMV in the United States is young children in day care settings, with approximately 50% of this population showing evidence of viral shedding in saliva.1 Adult populations in North America have a high prevalence of CMV IgG antibodies indicative of prior infection, with rates reaching 50% to 80%. Among seronegative individuals aged 12 to 49, the rate of seroconversion is approximately 1 in 60 annually.6 Significant racial disparities have been noted in rates of seroprevalence and seroconversion, with higher rates of infection in non-Hispanic Black and Mexican American individuals.6 Overall, the rate of new CMV infection among pregnant women in the United States is 0.7% to 4%.7
Clinical manifestations
Manifestations of infection differ depending on whether or not infection is primary or recurrent (secondary) and whether or not the host is immunocompetent or has a compromised immune system. Unique manifestations develop in the fetus.
CMV infection in children and adults. Among individuals with a normal immune response, the typical course of CMV is either no symptoms or a mononucleosis-like illness. In symptomatic patients, the most common symptoms include malaise, fever, and night sweats, and the most common associated laboratory abnormalities are elevation in liver function tests and a decreased white blood cell count, with a predominance of lymphocytes.8
Immunocompromised individuals are at risk for significant morbidity and mortality resulting from CMV. Illness may be the result of reactivation of latent infection due to decreased immune function or may be acquired as a result of treatment such as transplantation of CMV-positive organs or tissues, including bone marrow. Virtually any organ system can be affected, with potential for permanent organ damage and death. Severe systemic infection also can occur.
CMV infection in the fetus and neonate. As noted previously, fetal infection develops as a result of transplacental passage coincident with maternal infection. The risk of CMV transmission to the fetus and the severity of fetal injury vary based on gestational age at fetal infection and whether or not maternal infection is primary or secondary.
In most studies, primary maternal infections are associated with higher rates of fetal infection and more severe fetal and neonatal disease manifestations.2,7,9,10 Primary infections carry an overall 30% to 40% risk of transmission to the fetus.7,11 The risk of fetal transmission is much lower with a recurrent infection and is usually less than 2%.11 Due to their greater overall incidence, secondary infections account for the majority of cases of fetal and neonatal CMV disease.7 Importantly, although secondary infections generally have been regarded as having a lower risk and lower severity of fetal and neonatal disease, several recent studies have demonstrated rates of complications similar to, and even exceeding, those of primary infections.12-15 The TABLE provides a summary of the risks of fetal transmission and symptomatic fetal infection based on trimester of pregnancy.2,11,16-18
In the fetus, CMV may affect multiple organ systems. Among sonographic and magnetic resonance imaging (MRI) findings, central nervous system (CNS) anomalies are the most common.19,20 These can include microcephaly, ventriculomegaly, and periventricular calcifications. The gastrointestinal system also is frequently affected, and findings include echogenic bowel, hepatosplenomegaly, and liver calcifications. Lastly, isolated effusions, placentomegaly, fetal growth restriction, and even frank hydrops can develop. More favorable neurologic outcomes have been demonstrated in infants with no prenatal brain imaging abnormalities.20,21 However, the role of MRI in prenatal prognosis currently is not well defined.
FIGURE 1 illustrates selected sonographic findings associated with fetal CMV infection.

About 85% to 90% of infants with congenital CMV that results from primary maternal infection have no symptoms at birth. Among the 10% to 15% of infants that do have symptoms, petechial rash, jaundice, and hepatosplenomegaly are the most common manifestations (“blueberry muffin baby”). Approximately 10% to 20% of infants in this group have evidence of chorioretinitis on ophthalmologic examination, and 50% show either microcephaly or low birth weight.22Among survivors of symptomatic congenital CMV, more than 50% have long-term neurologic morbidities that may include sensorineural hearing loss, seizures, vision impairment, and developmental disabilities. Note that even when neonates appear asymptomatic at birth (regardless of whether infection is primary or secondary), 5% may develop microcephaly and motor deficits, 10% go on to develop sensorineural hearing loss, and the overall rate of neurologic morbidity reaches 13% to 15%.12,23 Some of the observed deficits manifest at several years of age, and, currently, no models exist for prediction of outcome.
Continue to: Diagnosing CMV infection...
Diagnosing CMV infection
Maternal infection
If maternal CMV infection is suspected based on a symptomatic illness or an abnormal fetal ultrasound exam, the first diagnostic test should be an assessment of IgM and IgG serology. If the former test results are positive and the latter negative, the diagnosis of acute CMV infection is confirmed. A positive serum CMV DNA polymerase chain reaction (PCR) test adds additional assurance that the diagnosis is correct. Primary infection, as noted above, poses the greatest risk of serious injury to the fetus.1
A frequent diagnostic dilemma arises when both the IgM and IgG antibody are positive. Remember that CMV IgM antibody can remain positive for 9 to 12 months after a primary infection and can reappear in the maternal serum in the face of a recurrent or reactivated infection. When confronted by both a positive IgM and positive IgG result, the clinician should then order IgG avidity testing. If the avidity is low to moderate, which reflects poor binding of antibody to the virus, the patient likely has an acute infection. If the avidity is high, which reflects enhanced binding of antibody to virus, the patient probably has a recurrent or reactivated infection; this scenario poses less danger to the developing fetus. The presence of CMV DNA in serum is also more consistent with acute infection, although viremia still can occur with recurrent infection. FIGURE 2 presents a suggested algorithm for the diagnosis of CMV in the pregnant patient.1
If a diagnosis of maternal CMV infection is confirmed, liver function tests should be obtained to determine if CMV hepatitis is present. If the liver function tests are abnormal, a coagulation profile also should be performed to identify the mother who might be at risk for peripartum hemorrhage.
Fetal infection
The single best test for confirmation of congenital CMV infection is detection of viral DNA and quantitation of viral load in the amniotic fluid by PCR. If the amniocentesis is performed prior to 20 weeks’ gestation and is negative, the test should be repeated in approximately 4 weeks.1,19,24
Detection of viral DNA indicates congenital infection. The ultimate task, however, is to determine if the infection has injured the fetus. Detailed ultrasound examination is the key to identifying fetal injury. As noted previously, the principal ultrasonographic findings that suggest congenital CMV infection include2,19,20,21,25:
- hydropic placenta
- fetal growth restriction
- microcephaly (head circumference more than 3 standard deviations below the mean)
- periventricular calcifications
- enlarged liver
- echogenic bowel
- ascites
- fetal hydrops.
Management: Evidence on CMV hyperimmune globulin, valacyclovir
If the immunocompetent mother has clinical manifestations of infection, she should receive symptomatic treatment. She should be encouraged to rest as much as possible, stay well hydrated, and use acetaminophen (1,000 mg every 6 to 8 hours) as needed for malaise and fever.
However, if the mother is immunocompromised and has signs of serious complications, such as chorioretinitis, hepatitis, or pneumonia, more aggressive therapy is indicated. Drugs used in this setting include foscarnet and ganciclovir and are best prescribed in consultation with a medical infectious disease specialist.
At this time, no consistently effective therapy for congenital infection is available. Therefore, if a patient has primary CMV infection in the first half of pregnancy, particularly in the first trimester, she should be counseled that the risk of fetal infection is approximately 40% and that approximately 5% to 15% of infants will be severely affected at birth. Given this information, some patients may opt for pregnancy termination.
In 2005, a report from Nigro and colleagues stimulated great hope that CMV-specific hyperimmune globulin (CytoGam) might be of value for both treatment and prophylaxis for congenital infection.26 These authors studied 157 women with confirmed primary CMV infection. One-hundred forty-eight women were asymptomatic and were identified by routine serologic screening, 8 had symptomatic infection, and 1 was identified because of abnormal fetal ultrasound findings. Forty-five women had CMV detected in amniotic fluid by PCR or culture more than 6 weeks before study enrollment. Thirty-one of these women were treated with intravenous hyperimmune globulin (200 U or 200 mg/kg maternal body weight); 14 declined treatment. Seven of the latter women had infants who were acutely symptomatic at the time of delivery; only 1 of the 31 treated women had an affected neonate (adjusted odds ratio [OR], 0.02; P<.001). In this same study, 84 women did not have a diagnostic amniocentesis because their infection occurred within 6 weeks of enrollment, their gestational age was less than 20 weeks, or they declined the procedure. Thirty-seven of these women received hyperimmune globulin (100 U or 100 mg/kg) every month until delivery, and 47 declined treatment. Six of the treated women delivered infected infants compared with 19 of the untreated women (adjusted OR, 0.32; P<.04).
Although these results were quite encouraging, several problems existed with the study’s design, as noted in an editorial that accompanied the study’s publication.27 First, the study was not randomized or placebo controlled. Second, patients were not stratified based on the severity of fetal ultrasound abnormalities. Third, the dosing of hyperimmune globulin varied; 9 of the 31 patients in the treatment group received additional infusions of drug into either the amniotic fluid or fetal umbilical vein. Moreover, patients in the prophylaxis group actually received a higher cumulative dose of hyperimmune globulin than patients in the treatment group.
Two subsequent investigations that were better designed were unable to verify the effectiveness of hyperimmune globulin. In 2014, Revello and colleagues reported the results of a prospective, randomized, placebo-controlled, double-blinded study of 124 women at 5 to 26 weeks’ gestation with confirmed primary CMV infection.28 The rate of congenital infection was 30% in the group treated with hyperimmune globulin and 44% in the placebo group (P=.13). There also was no significant difference in the concentration of serum CMV DNA in treated versus untreated mothers. Moreover, the number of adverse obstetric events (preterm delivery, fetal growth restriction, intrahepatic cholestasis of pregnancy, and postpartum preeclampsia) in the treatment group was higher than in the placebo group, 13% versus 2%.
In 2021, Hughes and colleagues published the results of a multicenter, double-blind trial in 399 women who had a diagnosis of primary CMV infection before 23 weeks’ gestation.29 The primary outcome was defined as a composite of congenital CMV infection or fetal/neonatal death. An adverse primary outcome occurred in 22.7% of the patients who received hyperimmune globulin and 19.4% of those who received placebo (relative risk, 1.17; 95% confidence interval [CI], 0.80–1.72; P=.42).
Continue to: Jacquemard and colleagues...
Jacquemard and colleagues then proposed a different approach.30 In a small pilot study of 20 patients, these authors used high doses of oral valacylovir (2 g 4 times daily) and documented therapeutic drug concentrations and a decline in CMV viral load in fetal serum. Patients were not stratified by severity of fetal injury at onset of treatment, so the authors were unable to define which fetuses were most likely to benefit from treatment.
In a follow-up investigation, Leruez-Ville and colleagues reported another small series in which high-dose oral valacyclovir (8 g daily) was used for treatment.31 They excluded fetuses with severe brain anomalies and fetuses with no sonographic evidence of injury. The median gestational age at diagnosis was 26 weeks. Thirty-four of 43 treated fetuses were free of injury at birth. In addition, the viral load in the neonate’s serum decreased significantly after treatment, and the platelet count increased. The authors then compared these outcomes to a historical cohort and confirmed that treatment increased the proportion of asymptomatic neonates from 43% without treatment to 82% with treatment (P<.05 with no overlapping confidence intervals).
We conclude from these investigations that hyperimmune globulin is unlikely to be of value in treating congenital CMV infection, especially if the fetus already has sonographic findings of severe injury. High-dose oral valacyclovir also is unlikely to be of value in severely affected fetuses, particularly those with evidence of CNS injury. However, antiviral therapy may be of modest value in situations when the fetus is less severely injured.
Preventive measures
Since no definitive treatment is available for congenital CMV infection, our efforts as clinicians should focus on measures that may prevent transmission of infection to the pregnant patient. These measures include:
- Encouraging patients to use careful handwashing techniques when handling infant diapers and toys.
- Encouraging patients to adopt safe sexual practices if not already engaged in a mutually faithful, monogamous relationship.
- Using CMV-negative blood when transfusing a pregnant woman or a fetus.
At the present time, unfortunately, a readily available and highly effective therapy for prevention of CMV infection is not available.
CASE Congenital infection diagnosed
The ultrasound findings are most consistent with congenital CMV infection, especially given the patient’s work as an elementary schoolteacher. The diagnosis of maternal infection is best established by conventional serology (positive IgM, negative IgM) and detection of viral DNA in maternal blood by PCR testing. The diagnosis of congenital infection is best confirmed by documentation of viral DNA in the amniotic fluid by PCR testing. Given that this fetus already has evidence of moderate to severe injury, no treatment is likely to be effective in reversing the abnormal ultrasound findings. Pregnancy termination may be an option, depending upon the patient’s desires and the legal restrictions prevalent in the patient’s geographic area. ●
- Cytomegalovirus infection is the most common of the perinatally transmitted infections.
- Maternal infection is often asymptomatic. When symptoms are present, they resemble those of an influenza-like illness. In immunocompromised persons, however, CMV may cause serious complications, including pneumonia, hepatitis, and chorioretinitis.
- The virus is transmitted by contact with contaminated body fluids, such as saliva, urine, blood, and genital secretions.
- The greatest risk of severe fetal injury results from primary maternal infection in the first trimester of pregnancy.
- Manifestations of severe congenital CMV infection include growth restriction, microcephaly, ventriculomegaly, hepatosplenomegaly, ascites, chorioretinitis, thrombocytopenia, purpura, and hydrops (“blueberry muffin baby”).
- Late manifestations of infection, which usually follow recurrent maternal infection, may appear as a child enters elementary school and include visual and auditory deficits, developmental delays, and learning disabilities.
- The diagnosis of maternal infection is confirmed by serology and detection of viral DNA in the serum by PCR testing.
- The diagnosis of fetal infection is best made by a combination of abnormal ultrasound findings and detection of CMV DNA in amniotic fluid. The characteristic ultrasound findings include placentomegaly, microcephaly, ventriculomegaly, growth restriction, echogenic bowel, and serous effusions/hydrops.
- Treatment of the mother with antiviral medications such as valacyclovir may be of modest value in reducing placental edema, decreasing viral load in the fetus, and hastening the resolution of some ultrasound findings, such as echogenic bowel.
- While initial studies seemed promising, the use of hyperimmune globulin has not proven to be consistently effective in treating congenital infection.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TR, et al, eds. Creasy and Resnik’s Maternal Fetal Medicine: Principles and Practice. 8th ed. 2019:888-890.
- Chatzakis C, Ville Y, Makrydimas G, et al. Timing of primary maternal cytomegalovirus infection and rates of vertical transmission and fetal consequences. Am J Obstet Gynecol. 2020;223:870-883.e11. doi:10.1016/j.ajog.2020.05.038
- Kelly MS, Benjamin DK, Puopolo KM, et al. Postnatal cytomegalovirus infection and the risk for bronchopulmonary dysplasia. JAMA Pediatr. 2015;169:e153785. doi:10.1001 /jamapediatrics.2015.3785
- Messinger CJ, Lipsitch M, Bateman BT, et al. Association between congenital cytomegalovirus and the prevalence at birth of microcephaly in the United States. JAMA Pediatr. 2020;174:1159-1167. doi:10.1001/jamapediatrics.2020.3009
- De Cuyper E, Acke F, Keymeulen A, et al. Risk factors for hearing loss at birth in newborns with congenital cytomegalovirus infection. JAMA Otolaryngol Head Neck Surg. 2023;149:122-130. doi:10.1001/jamaoto.2022.4109
- Colugnati FA, Staras SA, Dollard SC, et al. Incidence of cytomegalovirus infection among the general population and pregnant women in the United States. BMC Infect Dis. 2007;7:71. doi:10.1186/1471-2334-7-71
- Stagno S, Pass RF, Cloud G, et al. Primary cytomegalovirus infection in pregnancy. Incidence, transmission to fetus, and clinical outcome. JAMA. 1986;256:1904-1908.
- Wreghitt TG, Teare EL, Sule O, et al. Cytomegalovirus infection in immunocompetent patients. Clin Infect Dis. 2003;37:1603-1606. doi:10.1086/379711
- Fowler KB, Stagno S, Pass RF, et al. The outcome of congenital cytomegalovirus infection in relation to maternal antibody status. N Engl J Med. 1992;326:663-667. doi:10.1056 /NEJM199203053261003
- Faure-Bardon V, Magny JF, Parodi M, et al. Sequelae of congenital cytomegalovirus following maternal primary infections are limited to those acquired in the first trimester of pregnancy. Clin Infect Dis. 2019;69:1526-1532. doi:10.1093/ cid/ciy1128
- Kenneson A, Cannon MJ. Review and meta-analysis of the epidemiology of congenital cytomegalovirus (CMV) infection. Rev Med Virol. 2007;17:253-276. doi:10.1002/ rmv.535
- Boppana SB, Pass RF, Britt WJ, et al. Symptomatic congenital cytomegalovirus infection: neonatal morbidity and mortality. Pediatr Infect Dis J. 1992;11:93-99. doi:10.1097/00006454-199202000-00007
- Ross SA, Fowler KB, Ashrith G, et al. Hearing loss in children with congenital cytomegalovirus infection born to mothers with preexisting immunity. J Pediatr. 2006;148:332-336. doi:10.1016/j.jpeds.2005.09.003
- Zalel Y, Gilboa Y, Berkenshtat M, et al. Secondary cytomegalovirus infection can cause severe fetal sequelae despite maternal preconceptional immunity. Ultrasound Obstet Gynecol. 31:417-420. doi:10.1002/uog.5255
- Scaramuzzino F, Di Pastena M, Chiurchiu S, et al. Secondary cytomegalovirus infections: how much do we still not know? Comparison of children with symptomatic congenital cytomegalovirus born to mothers with primary and secondary infection. Front Pediatr. 2022;10:885926. doi:10.3389/fped.2022.885926
- Gindes L, Teperberg-Oikawa M, Sherman D, et al. Congenital cytomegalovirus infection following primary maternal infection in the third trimester. BJOG. 2008;115:830-835. doi:10.1111/j.1471-0528.2007.01651.x
- Hadar E, Dorfman E, Bardin R, et al. Symptomatic congenital cytomegalovirus disease following non-primary maternal infection: a retrospective cohort study. BMC Infect Dis. 2017;17:31. doi:10.1186/s12879-016-2161-3
- Elkan Miller T, Weisz B, Yinon Y, et al. Congenital cytomegalovirus infection following second and third trimester maternal infection is associated with mild childhood adverse outcome not predicted by prenatal imaging. J Pediatric Infect Dis Soc. 2021;10:562-568. doi:10.1093/jpids/ piaa154
- Lipitz S, Yinon Y, Malinger G, et al. Risk of cytomegalovirusassociated sequelae in relation to time of infection and findings on prenatal imaging. Ultrasound Obstet Gynecol. 2013;41:508-514. doi:10.1002/uog.12377
- Lipitz S, Elkan Miller T, Yinon Y, et al. Revisiting short- and long-term outcome after fetal first-trimester primary cytomegalovirus infection in relation to prenatal imaging findings. Ultrasound Obstet Gynecol. 2020;56:572-578. doi:10.1002/uog.21946
- Buca D, Di Mascio D, Rizzo G, et al. Outcome of fetuses with congenital cytomegalovirus infection and normal ultrasound at diagnosis: systematic review and meta-analysis. Ultrasound Obstet Gynecol. 2021;57:551-559. doi:10.1002/uog.23143
- Boppana SB, Ross SA, Fowler KB. Congenital cytomegalovirus infection: clinical outcome. Clin Infect Dis. 2013;57 (suppl 4):S178-S181. doi:10.1093/cid/cit629
- Dollard SC, Grosse SD, Ross DS. New estimates of the prevalence of neurological and sensory sequelae and mortality associated with congenital cytomegalovirus infection. Rev Med Virol. 2007;17:355-363. doi:10.1002/rmv.544
- Hughes BL, Gyamfi-Bannerman C. Diagnosis and antenatal management of congenital cytomegalovirus infection. Am J Obstet Gynecol. 2016;214:B5-11. doi:10.1016 /j.ajog.2016.02.042
- Rouse DJ, Fette LM, Hughes BL, et al. Noninvasive prediction of congenital cytomegalovirus infection after maternal primary infection. Obstet Gynecol. 2022;139:400-406. doi:10.1097/AOG.0000000000004691
- Nigro G, Adler SP, La Torre R, et al; Congenital Cytomegalovirus Collaborating Group. Passive immunization during pregnancy for congenital cytomegalovirus infection. N Engl J Med. 2005;353:1350-1362. doi:10.1056/NEJMoa043337
- Duff P. Immunotherapy for congenital cytomegalovirus infection. N Engl J Med. 2005;355:1402-1404. doi:10.1056 /NEJMe058172
- Revello MG, Lazzarotto T, Guerra B, et al. A randomized trial of hyperimmune globulin to prevent congenital cytomegalovirus. N Engl J Med. 2014;370:1316-1326. doi:10.1056/NEJMoa1310214
- Hughes BL, Clifton RG, Rouse DJ, et al. A trial of hyperimmune globulin to prevent congenital cytomegalovirus infection. N Engl J Med. 2021;385:436-444. doi:10.1056/NEJMoa1913569
- Jacquemard F, Yamamoto M, Costa JM, et al. Maternal administration of valaciclovir in symptomatic intrauterine cytomegalovirus infection. BJOG. 2007;114:1113-1121. doi:10.1111/j.1471-0528.2007.01308.x
- Leruez-Ville M, Ghout I, Bussières L, et al. In utero treatment of congenital cytomegalovirus infection with valacyclovir in a multicenter, open-label, phase II study. Am J Obstet Gynecol. 2016;215:462.e1-462.e10. doi:10.1016/j.ajog.2016.04.003
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TR, et al, eds. Creasy and Resnik’s Maternal Fetal Medicine: Principles and Practice. 8th ed. 2019:888-890.
- Chatzakis C, Ville Y, Makrydimas G, et al. Timing of primary maternal cytomegalovirus infection and rates of vertical transmission and fetal consequences. Am J Obstet Gynecol. 2020;223:870-883.e11. doi:10.1016/j.ajog.2020.05.038
- Kelly MS, Benjamin DK, Puopolo KM, et al. Postnatal cytomegalovirus infection and the risk for bronchopulmonary dysplasia. JAMA Pediatr. 2015;169:e153785. doi:10.1001 /jamapediatrics.2015.3785
- Messinger CJ, Lipsitch M, Bateman BT, et al. Association between congenital cytomegalovirus and the prevalence at birth of microcephaly in the United States. JAMA Pediatr. 2020;174:1159-1167. doi:10.1001/jamapediatrics.2020.3009
- De Cuyper E, Acke F, Keymeulen A, et al. Risk factors for hearing loss at birth in newborns with congenital cytomegalovirus infection. JAMA Otolaryngol Head Neck Surg. 2023;149:122-130. doi:10.1001/jamaoto.2022.4109
- Colugnati FA, Staras SA, Dollard SC, et al. Incidence of cytomegalovirus infection among the general population and pregnant women in the United States. BMC Infect Dis. 2007;7:71. doi:10.1186/1471-2334-7-71
- Stagno S, Pass RF, Cloud G, et al. Primary cytomegalovirus infection in pregnancy. Incidence, transmission to fetus, and clinical outcome. JAMA. 1986;256:1904-1908.
- Wreghitt TG, Teare EL, Sule O, et al. Cytomegalovirus infection in immunocompetent patients. Clin Infect Dis. 2003;37:1603-1606. doi:10.1086/379711
- Fowler KB, Stagno S, Pass RF, et al. The outcome of congenital cytomegalovirus infection in relation to maternal antibody status. N Engl J Med. 1992;326:663-667. doi:10.1056 /NEJM199203053261003
- Faure-Bardon V, Magny JF, Parodi M, et al. Sequelae of congenital cytomegalovirus following maternal primary infections are limited to those acquired in the first trimester of pregnancy. Clin Infect Dis. 2019;69:1526-1532. doi:10.1093/ cid/ciy1128
- Kenneson A, Cannon MJ. Review and meta-analysis of the epidemiology of congenital cytomegalovirus (CMV) infection. Rev Med Virol. 2007;17:253-276. doi:10.1002/ rmv.535
- Boppana SB, Pass RF, Britt WJ, et al. Symptomatic congenital cytomegalovirus infection: neonatal morbidity and mortality. Pediatr Infect Dis J. 1992;11:93-99. doi:10.1097/00006454-199202000-00007
- Ross SA, Fowler KB, Ashrith G, et al. Hearing loss in children with congenital cytomegalovirus infection born to mothers with preexisting immunity. J Pediatr. 2006;148:332-336. doi:10.1016/j.jpeds.2005.09.003
- Zalel Y, Gilboa Y, Berkenshtat M, et al. Secondary cytomegalovirus infection can cause severe fetal sequelae despite maternal preconceptional immunity. Ultrasound Obstet Gynecol. 31:417-420. doi:10.1002/uog.5255
- Scaramuzzino F, Di Pastena M, Chiurchiu S, et al. Secondary cytomegalovirus infections: how much do we still not know? Comparison of children with symptomatic congenital cytomegalovirus born to mothers with primary and secondary infection. Front Pediatr. 2022;10:885926. doi:10.3389/fped.2022.885926
- Gindes L, Teperberg-Oikawa M, Sherman D, et al. Congenital cytomegalovirus infection following primary maternal infection in the third trimester. BJOG. 2008;115:830-835. doi:10.1111/j.1471-0528.2007.01651.x
- Hadar E, Dorfman E, Bardin R, et al. Symptomatic congenital cytomegalovirus disease following non-primary maternal infection: a retrospective cohort study. BMC Infect Dis. 2017;17:31. doi:10.1186/s12879-016-2161-3
- Elkan Miller T, Weisz B, Yinon Y, et al. Congenital cytomegalovirus infection following second and third trimester maternal infection is associated with mild childhood adverse outcome not predicted by prenatal imaging. J Pediatric Infect Dis Soc. 2021;10:562-568. doi:10.1093/jpids/ piaa154
- Lipitz S, Yinon Y, Malinger G, et al. Risk of cytomegalovirusassociated sequelae in relation to time of infection and findings on prenatal imaging. Ultrasound Obstet Gynecol. 2013;41:508-514. doi:10.1002/uog.12377
- Lipitz S, Elkan Miller T, Yinon Y, et al. Revisiting short- and long-term outcome after fetal first-trimester primary cytomegalovirus infection in relation to prenatal imaging findings. Ultrasound Obstet Gynecol. 2020;56:572-578. doi:10.1002/uog.21946
- Buca D, Di Mascio D, Rizzo G, et al. Outcome of fetuses with congenital cytomegalovirus infection and normal ultrasound at diagnosis: systematic review and meta-analysis. Ultrasound Obstet Gynecol. 2021;57:551-559. doi:10.1002/uog.23143
- Boppana SB, Ross SA, Fowler KB. Congenital cytomegalovirus infection: clinical outcome. Clin Infect Dis. 2013;57 (suppl 4):S178-S181. doi:10.1093/cid/cit629
- Dollard SC, Grosse SD, Ross DS. New estimates of the prevalence of neurological and sensory sequelae and mortality associated with congenital cytomegalovirus infection. Rev Med Virol. 2007;17:355-363. doi:10.1002/rmv.544
- Hughes BL, Gyamfi-Bannerman C. Diagnosis and antenatal management of congenital cytomegalovirus infection. Am J Obstet Gynecol. 2016;214:B5-11. doi:10.1016 /j.ajog.2016.02.042
- Rouse DJ, Fette LM, Hughes BL, et al. Noninvasive prediction of congenital cytomegalovirus infection after maternal primary infection. Obstet Gynecol. 2022;139:400-406. doi:10.1097/AOG.0000000000004691
- Nigro G, Adler SP, La Torre R, et al; Congenital Cytomegalovirus Collaborating Group. Passive immunization during pregnancy for congenital cytomegalovirus infection. N Engl J Med. 2005;353:1350-1362. doi:10.1056/NEJMoa043337
- Duff P. Immunotherapy for congenital cytomegalovirus infection. N Engl J Med. 2005;355:1402-1404. doi:10.1056 /NEJMe058172
- Revello MG, Lazzarotto T, Guerra B, et al. A randomized trial of hyperimmune globulin to prevent congenital cytomegalovirus. N Engl J Med. 2014;370:1316-1326. doi:10.1056/NEJMoa1310214
- Hughes BL, Clifton RG, Rouse DJ, et al. A trial of hyperimmune globulin to prevent congenital cytomegalovirus infection. N Engl J Med. 2021;385:436-444. doi:10.1056/NEJMoa1913569
- Jacquemard F, Yamamoto M, Costa JM, et al. Maternal administration of valaciclovir in symptomatic intrauterine cytomegalovirus infection. BJOG. 2007;114:1113-1121. doi:10.1111/j.1471-0528.2007.01308.x
- Leruez-Ville M, Ghout I, Bussières L, et al. In utero treatment of congenital cytomegalovirus infection with valacyclovir in a multicenter, open-label, phase II study. Am J Obstet Gynecol. 2016;215:462.e1-462.e10. doi:10.1016/j.ajog.2016.04.003
Viral threats to the fetus and mother: Parvovirus and varicella
We review 2 important viral infections in this article. One, parvovirus, poses a major threat to the fetus. The second, varicella, poses less risk to the fetus but significantly greater risk to the mother. We focus on the epidemiology, clinical presentation, diagnosis, and management of each infection.
Parvovirus infection and its risks to the fetus
CASE #1 Pregnant teacher exposed to fifth disease
A 28-year-old primigravid woman at 16 weeks’ gestation works as an elementary school teacher. Over the past 3 weeks, she has been exposed to 4 children who had fifth disease. She now requests evaluation because she has malaise, arthralgias, myalgias, fever of 38.2°C, and a fine lacelike erythematous rash on her trunk, arms, and cheeks.
- What is the most likely diagnosis?
- What diagnostic tests are indicated?
- Is her fetus at risk?
Epidemiology of parvovirus
Parvovirus B19 is a small, single-stranded DNA virus. It is highly contagious and is transmitted primarily by respiratory droplets. Transmission also can occur via infected blood, for example, through a blood transfusion. The incubation period is 10 to 20 days. Among adults, the individuals at greatest risk for infection are those who have close contact with young children, such as parents, day-care workers, and elementary school teachers. With sustained exposure in the household or classroom, the risk of seroconversion approaches 50%.1 Approximately 50% to 60% of reproductive-aged women have evidence of prior infection, and immunity is usually lifelong.
Clinical manifestations
The classic presentation of parvovirus infection is erythema infectiosum, also called fifth disease. This condition is characterized by a “slapped cheek” facial rash, malaise, myalgias, arthralgias, and low-grade fever. A fine lacelike rash often develops over the torso. In adults, the characteristic rash may be absent, and the most common presentation is a flu-like illness with joint pains.1,2 In children and in adults with an underlying hemoglobinopathy, parvovirus can cause transient aplastic crisis, and patients present with signs of a severe anemia, such as dyspnea, pallor, and fatigue.
Although parvovirus infection usually poses no serious risk in otherwise healthy children and adults, it can cause major fetal injury when the pregnant woman is infected early in pregnancy. The principal manifestation of fetal infection is hydrops. Hydrops primarily results when the virus crosses the placenta and attaches to the P antigen on the surface of red cell progenitors in the fetal marrow, causing an aplastic anemia with resultant high-output congestive heart failure. The virus also may directly injure the fetal myocardium, thus exacerbating heart failure. Other manifestations of congenital parvovirus include thrombocytopenia and hepatitis.3
The severity of fetal injury is inversely proportional to the gestational age at the time of maternal infection. When primary maternal infection occurs in the first trimester, the frequency of fetal hydrops is 5% to 10%. If infection develops in weeks 13 to 20, the risk of hydrops decreases to 5% or less. If infection develops beyond week 20, the incidence of fetal hydrops is 1% or lower.2
Continue to: Diagnostic steps...
Diagnostic steps
Appropriate diagnostic evaluation for a pregnant woman with exposure to parvovirus or clinical manifestations suggestive of parvovirus infection is outlined in FIGURE 1.
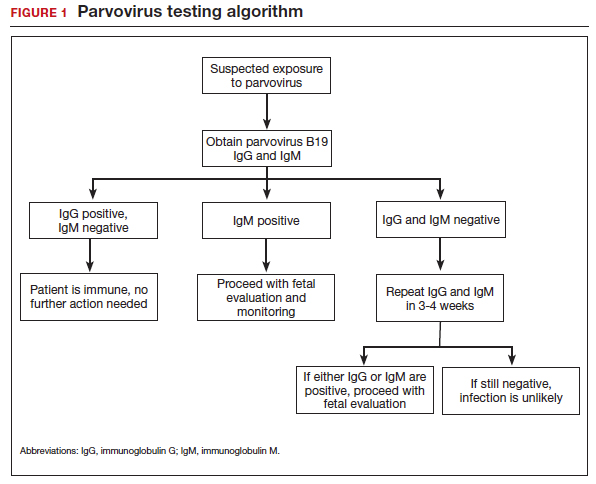
If infection is confirmed, serial ultrasound monitoring should be performed on a weekly to biweekly basis for 8 to 12 weeks, as delineated in FIGURE 2. Extended surveillance is necessary because the incubation period in the fetus is longer than that in the mother.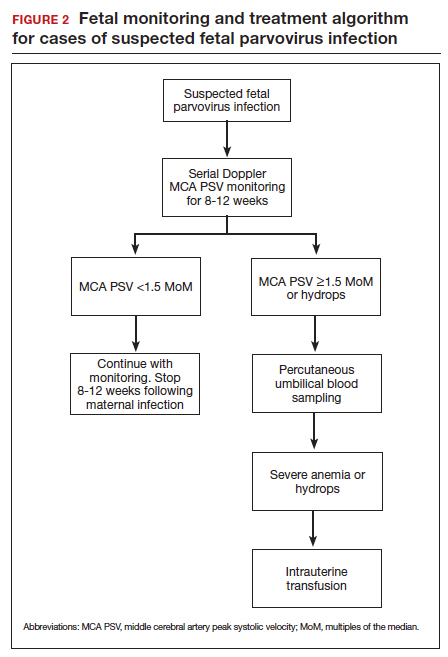
As the fetus develops anemia, peripheral tissues become hypoxic, leading to reflex peripheral vasoconstriction and increased cardiac output. At the same time, reduction in the number of fetal red blood cells decreases blood viscosity. The combination of these changes results in an increase in blood flow to the fetal brain, which can be detected by measuring the peak systolic velocity of flow in the middle cerebral artery (MCA PSV) with Doppler ultrasound imaging (FIGURE 3). The increase in MCA PSV parallels the decrease in fetal hematocrit and precedes the development of hydrops. In fact, signs of fetal hydrops do not usually develop until the fetal hematocrit falls to 15 to 20 vol%.
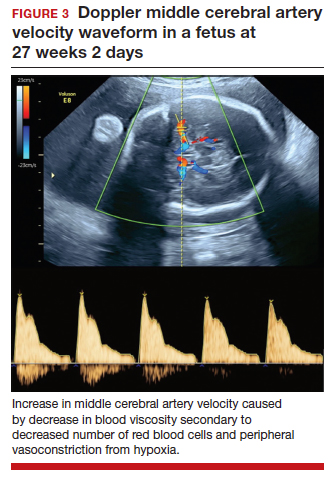
Management may necessitate intrauterine transfusion
Although some cases of fetal hydrops may resolve spontaneously, most authors agree that intrauterine transfusion is essential. In most instances, only a single intrauterine transfusion is necessary. In some fetuses, however, the infection is so prolonged and the anemia so severe that 2 to 3 transfusions may be required.
Infants who survive the intrauterine transfusion usually have an excellent long-term prognosis. However, isolated case reports have documented neurologic morbidity and prolonged transfusion-dependent anemia.4 In light of these reports, we recommend that a third trimester ultrasound exam be performed to assess fetal growth and evaluate the anatomy of the fetal brain. For the fetus with abnormal intracranial findings on ultrasonography, fetal magnetic resonance imaging is indicated.5
CASE #1 Diagnosis is probable parvovirus
The most likely diagnosis in this case is erythema infectiosum. This diagnosis can be confirmed by identifying positive immunogloblulin M (IgM) antibody and by detecting parvovirus in the maternal serum by polymerase chain reaction. Given the gestational age of 16 weeks, the risk of serious fetal injury should be less than 5%. Nevertheless, serial ultrasound examinations should be performed to assess for signs of fetal anemia.
Varicella exposure in pregnancy
CASE #2 Pregnant woman exposed to chickenpox has symptoms
Two weeks ago, a 32-year-old woman (G3P2002) at 24 weeks’ gestation was exposed to a neighbor’s child who had chickenpox. The patient has no history of natural infection or vaccination. She now has a fever of 38.6°C, malaise, headache, and a diffuse pruritic vesicular rash on her trunk and extremities. She also is experiencing a dry cough and mild dyspnea.
- What diagnostic tests are indicated?
- What treatment is indicated?
- What risk does this condition pose to the fetus?
Epidemiology of varicella
Varicella (chickenpox) is caused by the DNA varicella-zoster virus, an organism that is a member of the herpesvirus family. The disease occurs predominantly in children, and the infection is transmitted by respiratory droplets and by direct contact. Its incubation period is short (10–14 days), and it is highly contagious. More than 90% of susceptible close contacts will become infected after exposure to the index case. Like other herpesviruses, the varicella virus can establish a latent infection and then become manifest years later as herpes zoster (shingles).5,6
Continue to: Clinical manifestations...
Clinical manifestations
Patients with varicella usually have prodromal symptoms and signs that include malaise, fatigue, arthralgias, myalgias, and a low-grade fever. Varicella’s pathognomonic manifestation is a pruritic, macular rash that starts on the face and trunk and then spreads centripetally to the extremities. The lesions typically appear in “crops” and evolve through several distinct phases: macule, papule, vesicle, pustule, ulcer, and crust.5
In children, varicella is manifest almost entirely by mucocutaneous lesions. In adults, however, 2 serious and potentially life-threatening complications can occur. Approximately 1% of infected adults develop encephalitis and about 20% develop viral pneumonia, often accompanied by a severe superimposed bacterial pneumonia.5
When maternal infection develops in the first half of pregnancy, approximately 2% of fetuses will have evidence of congenital infection, usually manifested by circular, constricting scars on the extremities. These lesions typically occur in a dermatomal distribution. Spontaneous abortion and fetal death in utero also have been reported, but fortunately they are quite rare. When maternal infection occurs beyond 20 weeks of gestation, fetal injury is very uncommon.7
Interestingly, when maternal infection occurs at the time of delivery or shortly thereafter (from 5 days before until 2 days after delivery), neonatal varicella may develop. This infection may take 3 forms: disseminated mucocutaneous lesions, a deep-seated visceral infection, or severe pneumonia. In the era before the ready availability of antiviral agents, the case fatality rate from neonatal varicella was approximately 30%.5
Diagnosis is clinical
The diagnosis of varicella usually is established on the basis of clinical examination. It can be confirmed by identification of anti–varicella-zoster IgM.
Management includes assessing immunity
If a patient is seen for a preconception appointment, ask her whether she has ever had varicella or been vaccinated for this disease. If she is uncertain, a varicella-zoster immunoglobulin G (IgG) titer should be ordered. If the IgG titer is negative, denoting susceptibility to infection, the patient should be vaccinated before she tries to conceive (see below).8
If a patient has not had a preconception appointment and now presents for her first prenatal appointment, she should be asked about immunity to varicella. If she is uncertain, a varicella-zoster IgG assay should be obtained. Approximately 75% of patients who are uncertain about immunity will, in fact, be immune. Those who are not immune should be counseled to avoid exposure to individuals who may have varicella, and they should be targeted for vaccination immediately postpartum.5,9
If a susceptible pregnant patient has been exposed to an individual with varicella, she should receive 1 of 2 regimens within 72 to 96 hours to minimize the risk of maternal infection.5,9,10 One option is intramuscular varicella-zoster immune globulin (VariZIG), 125 U/10 kg body weight, with a maximum dose of 625 U (5 vials). The distributor of this agent is FFF Enterprises in Temucula, California (telephone: 800-843-7477). A company representative will assess the patient’s eligibility and deliver the drug within 24 hours if the patient is considered eligible. An alternative prophylactic regimen is oral acyclovir, 800 mg 5 times daily for 7 days, or oral valacyclovir, 1,000 mg 3 times daily for 7 days.
If, despite prophylaxis, the pregnant woman becomes infected, she should immediately be treated with 1 of the oral antiviral regimens described above. If she has evidence of encephalitis, pneumonia, or severe disseminated mucocutaneous infection, or if she is immunosuppressed, she should be hospitalized and treated with intravenous acyclovir, 10 mg/kg infused over 1 hour every 8 hours for 10 days.
Ultrasonography is the most valuable test to identify fetal infection. Key findings that suggest congenital varicella are fetal growth restriction, microcephaly, ventriculomegaly, echogenic foci in the liver, and limb abnormalities. There is no proven therapy for congenital varicella.
When a patient has varicella at the time of delivery, she should be isolated from her infant until all lesions have crusted over. In addition, the neonate should be treated with either VariZIG or an antiviral agent.5,9
Prevention with varicella vaccine
The varicella vaccine (Varivax) is a live-virus vaccine that is highly immunogenic. The vaccine is now part of the routine childhood immunization sequence. Children ages 1 to 12 years require only a single dose of the vaccine. Individuals older than 12 years of age require 2 doses, administered 4 to 6 weeks apart. The vaccine should not be administered during pregnancy. It also should not be administered to individuals who are severely immunocompromised, are receiving high-dose systemic steroids, have untreated tuberculosis, or have an allergy to neomycin, which is a component of the vaccine. The vaccine does not pose a risk to the breastfeeding infant.11
CASE #2 Hospitalization is recommended for this patient
The patient in this case developed acute varicella pneumonia as a result of her exposure to the neighbor’s child. The diagnosis can be confirmed by demonstrating a positive varicella-zoster IgM and by obtaining a chest x-ray that identifies the diffuse patchy infiltrates characteristic of viral pneumonia. Because this is such a potentially serious illness, the patient should be hospitalized and treated with intravenous acyclovir or valacyclovir. Antibiotics such as ceftriaxone and azithromycin may be indicated to treat superimposed bacterial pneumonia. Given the later gestational age, the fetus is at low risk for serious injury. ●
- Valeur-Jensen AK, Pedersen CB, Westergaard T, et al. Risk factors for parvovirus B19 infection in pregnancy. JAMA. 1999;281:1099-1105.
- Harger JH, Adler SP, Koch WC, et al. Prospective evaluation of 618 pregnant women exposed to parvovirus B19: risks and symptoms. Obstet Gynecol. 1998;91:413-420.
- Melamed N, Whittle W, Kelly EN, et al. Fetal thrombocytopenia in pregnancies with fetal human parvovirus-B19 infection. Am J Obstet Gynecol. 2015;212:793.e1-8.
- Nagel HTC, de Haan TR, Vandenbussche FPH, et al. Long-term outcome after fetal transfusion for hydrops associated with parvovirus B19 infection. Obstet Gynecol. 2007;109:42-47.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TR, et al (eds). Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:911-912.
- Cohen JI. Herpes zoster. N Engl J Med. 2013;369:255-263.
- Enders G, Miller E, Cradock-Watson J, et al. Consequences of varicella and herpes zoster in pregnancy: prospective study of 1739 cases. Lancet. 1994;343:1548-1551.
- Duff P. Varicella in pregnancy: five priorities for clinicians. Infect Dis Obstet Gynecol. 1994;1:163-165.
- Marin M, Guris D, Chaves SS, et al; Advisory Committee on Immunization Practices, Centers for Disease Control and Prevention. Prevention of varicella. MMWR Recommend Rep. 2007;56(RR-4):1-40.
- Swamy GK, Dotters-Katz SK. Safety and varicella outcomes after varicella zoster immune globulin administration in pregnancy. Am J Obstet Gynecol. 2019;221:655-656.
- Duff P. Varicella vaccine. Infect Dis Obstet Gynecol. 1996;4:63-65.
We review 2 important viral infections in this article. One, parvovirus, poses a major threat to the fetus. The second, varicella, poses less risk to the fetus but significantly greater risk to the mother. We focus on the epidemiology, clinical presentation, diagnosis, and management of each infection.
Parvovirus infection and its risks to the fetus
CASE #1 Pregnant teacher exposed to fifth disease
A 28-year-old primigravid woman at 16 weeks’ gestation works as an elementary school teacher. Over the past 3 weeks, she has been exposed to 4 children who had fifth disease. She now requests evaluation because she has malaise, arthralgias, myalgias, fever of 38.2°C, and a fine lacelike erythematous rash on her trunk, arms, and cheeks.
- What is the most likely diagnosis?
- What diagnostic tests are indicated?
- Is her fetus at risk?
Epidemiology of parvovirus
Parvovirus B19 is a small, single-stranded DNA virus. It is highly contagious and is transmitted primarily by respiratory droplets. Transmission also can occur via infected blood, for example, through a blood transfusion. The incubation period is 10 to 20 days. Among adults, the individuals at greatest risk for infection are those who have close contact with young children, such as parents, day-care workers, and elementary school teachers. With sustained exposure in the household or classroom, the risk of seroconversion approaches 50%.1 Approximately 50% to 60% of reproductive-aged women have evidence of prior infection, and immunity is usually lifelong.
Clinical manifestations
The classic presentation of parvovirus infection is erythema infectiosum, also called fifth disease. This condition is characterized by a “slapped cheek” facial rash, malaise, myalgias, arthralgias, and low-grade fever. A fine lacelike rash often develops over the torso. In adults, the characteristic rash may be absent, and the most common presentation is a flu-like illness with joint pains.1,2 In children and in adults with an underlying hemoglobinopathy, parvovirus can cause transient aplastic crisis, and patients present with signs of a severe anemia, such as dyspnea, pallor, and fatigue.
Although parvovirus infection usually poses no serious risk in otherwise healthy children and adults, it can cause major fetal injury when the pregnant woman is infected early in pregnancy. The principal manifestation of fetal infection is hydrops. Hydrops primarily results when the virus crosses the placenta and attaches to the P antigen on the surface of red cell progenitors in the fetal marrow, causing an aplastic anemia with resultant high-output congestive heart failure. The virus also may directly injure the fetal myocardium, thus exacerbating heart failure. Other manifestations of congenital parvovirus include thrombocytopenia and hepatitis.3
The severity of fetal injury is inversely proportional to the gestational age at the time of maternal infection. When primary maternal infection occurs in the first trimester, the frequency of fetal hydrops is 5% to 10%. If infection develops in weeks 13 to 20, the risk of hydrops decreases to 5% or less. If infection develops beyond week 20, the incidence of fetal hydrops is 1% or lower.2
Continue to: Diagnostic steps...
Diagnostic steps
Appropriate diagnostic evaluation for a pregnant woman with exposure to parvovirus or clinical manifestations suggestive of parvovirus infection is outlined in FIGURE 1.

If infection is confirmed, serial ultrasound monitoring should be performed on a weekly to biweekly basis for 8 to 12 weeks, as delineated in FIGURE 2. Extended surveillance is necessary because the incubation period in the fetus is longer than that in the mother.
As the fetus develops anemia, peripheral tissues become hypoxic, leading to reflex peripheral vasoconstriction and increased cardiac output. At the same time, reduction in the number of fetal red blood cells decreases blood viscosity. The combination of these changes results in an increase in blood flow to the fetal brain, which can be detected by measuring the peak systolic velocity of flow in the middle cerebral artery (MCA PSV) with Doppler ultrasound imaging (FIGURE 3). The increase in MCA PSV parallels the decrease in fetal hematocrit and precedes the development of hydrops. In fact, signs of fetal hydrops do not usually develop until the fetal hematocrit falls to 15 to 20 vol%.

Management may necessitate intrauterine transfusion
Although some cases of fetal hydrops may resolve spontaneously, most authors agree that intrauterine transfusion is essential. In most instances, only a single intrauterine transfusion is necessary. In some fetuses, however, the infection is so prolonged and the anemia so severe that 2 to 3 transfusions may be required.
Infants who survive the intrauterine transfusion usually have an excellent long-term prognosis. However, isolated case reports have documented neurologic morbidity and prolonged transfusion-dependent anemia.4 In light of these reports, we recommend that a third trimester ultrasound exam be performed to assess fetal growth and evaluate the anatomy of the fetal brain. For the fetus with abnormal intracranial findings on ultrasonography, fetal magnetic resonance imaging is indicated.5
CASE #1 Diagnosis is probable parvovirus
The most likely diagnosis in this case is erythema infectiosum. This diagnosis can be confirmed by identifying positive immunogloblulin M (IgM) antibody and by detecting parvovirus in the maternal serum by polymerase chain reaction. Given the gestational age of 16 weeks, the risk of serious fetal injury should be less than 5%. Nevertheless, serial ultrasound examinations should be performed to assess for signs of fetal anemia.
Varicella exposure in pregnancy
CASE #2 Pregnant woman exposed to chickenpox has symptoms
Two weeks ago, a 32-year-old woman (G3P2002) at 24 weeks’ gestation was exposed to a neighbor’s child who had chickenpox. The patient has no history of natural infection or vaccination. She now has a fever of 38.6°C, malaise, headache, and a diffuse pruritic vesicular rash on her trunk and extremities. She also is experiencing a dry cough and mild dyspnea.
- What diagnostic tests are indicated?
- What treatment is indicated?
- What risk does this condition pose to the fetus?
Epidemiology of varicella
Varicella (chickenpox) is caused by the DNA varicella-zoster virus, an organism that is a member of the herpesvirus family. The disease occurs predominantly in children, and the infection is transmitted by respiratory droplets and by direct contact. Its incubation period is short (10–14 days), and it is highly contagious. More than 90% of susceptible close contacts will become infected after exposure to the index case. Like other herpesviruses, the varicella virus can establish a latent infection and then become manifest years later as herpes zoster (shingles).5,6
Continue to: Clinical manifestations...
Clinical manifestations
Patients with varicella usually have prodromal symptoms and signs that include malaise, fatigue, arthralgias, myalgias, and a low-grade fever. Varicella’s pathognomonic manifestation is a pruritic, macular rash that starts on the face and trunk and then spreads centripetally to the extremities. The lesions typically appear in “crops” and evolve through several distinct phases: macule, papule, vesicle, pustule, ulcer, and crust.5
In children, varicella is manifest almost entirely by mucocutaneous lesions. In adults, however, 2 serious and potentially life-threatening complications can occur. Approximately 1% of infected adults develop encephalitis and about 20% develop viral pneumonia, often accompanied by a severe superimposed bacterial pneumonia.5
When maternal infection develops in the first half of pregnancy, approximately 2% of fetuses will have evidence of congenital infection, usually manifested by circular, constricting scars on the extremities. These lesions typically occur in a dermatomal distribution. Spontaneous abortion and fetal death in utero also have been reported, but fortunately they are quite rare. When maternal infection occurs beyond 20 weeks of gestation, fetal injury is very uncommon.7
Interestingly, when maternal infection occurs at the time of delivery or shortly thereafter (from 5 days before until 2 days after delivery), neonatal varicella may develop. This infection may take 3 forms: disseminated mucocutaneous lesions, a deep-seated visceral infection, or severe pneumonia. In the era before the ready availability of antiviral agents, the case fatality rate from neonatal varicella was approximately 30%.5
Diagnosis is clinical
The diagnosis of varicella usually is established on the basis of clinical examination. It can be confirmed by identification of anti–varicella-zoster IgM.
Management includes assessing immunity
If a patient is seen for a preconception appointment, ask her whether she has ever had varicella or been vaccinated for this disease. If she is uncertain, a varicella-zoster immunoglobulin G (IgG) titer should be ordered. If the IgG titer is negative, denoting susceptibility to infection, the patient should be vaccinated before she tries to conceive (see below).8
If a patient has not had a preconception appointment and now presents for her first prenatal appointment, she should be asked about immunity to varicella. If she is uncertain, a varicella-zoster IgG assay should be obtained. Approximately 75% of patients who are uncertain about immunity will, in fact, be immune. Those who are not immune should be counseled to avoid exposure to individuals who may have varicella, and they should be targeted for vaccination immediately postpartum.5,9
If a susceptible pregnant patient has been exposed to an individual with varicella, she should receive 1 of 2 regimens within 72 to 96 hours to minimize the risk of maternal infection.5,9,10 One option is intramuscular varicella-zoster immune globulin (VariZIG), 125 U/10 kg body weight, with a maximum dose of 625 U (5 vials). The distributor of this agent is FFF Enterprises in Temucula, California (telephone: 800-843-7477). A company representative will assess the patient’s eligibility and deliver the drug within 24 hours if the patient is considered eligible. An alternative prophylactic regimen is oral acyclovir, 800 mg 5 times daily for 7 days, or oral valacyclovir, 1,000 mg 3 times daily for 7 days.
If, despite prophylaxis, the pregnant woman becomes infected, she should immediately be treated with 1 of the oral antiviral regimens described above. If she has evidence of encephalitis, pneumonia, or severe disseminated mucocutaneous infection, or if she is immunosuppressed, she should be hospitalized and treated with intravenous acyclovir, 10 mg/kg infused over 1 hour every 8 hours for 10 days.
Ultrasonography is the most valuable test to identify fetal infection. Key findings that suggest congenital varicella are fetal growth restriction, microcephaly, ventriculomegaly, echogenic foci in the liver, and limb abnormalities. There is no proven therapy for congenital varicella.
When a patient has varicella at the time of delivery, she should be isolated from her infant until all lesions have crusted over. In addition, the neonate should be treated with either VariZIG or an antiviral agent.5,9
Prevention with varicella vaccine
The varicella vaccine (Varivax) is a live-virus vaccine that is highly immunogenic. The vaccine is now part of the routine childhood immunization sequence. Children ages 1 to 12 years require only a single dose of the vaccine. Individuals older than 12 years of age require 2 doses, administered 4 to 6 weeks apart. The vaccine should not be administered during pregnancy. It also should not be administered to individuals who are severely immunocompromised, are receiving high-dose systemic steroids, have untreated tuberculosis, or have an allergy to neomycin, which is a component of the vaccine. The vaccine does not pose a risk to the breastfeeding infant.11
CASE #2 Hospitalization is recommended for this patient
The patient in this case developed acute varicella pneumonia as a result of her exposure to the neighbor’s child. The diagnosis can be confirmed by demonstrating a positive varicella-zoster IgM and by obtaining a chest x-ray that identifies the diffuse patchy infiltrates characteristic of viral pneumonia. Because this is such a potentially serious illness, the patient should be hospitalized and treated with intravenous acyclovir or valacyclovir. Antibiotics such as ceftriaxone and azithromycin may be indicated to treat superimposed bacterial pneumonia. Given the later gestational age, the fetus is at low risk for serious injury. ●
We review 2 important viral infections in this article. One, parvovirus, poses a major threat to the fetus. The second, varicella, poses less risk to the fetus but significantly greater risk to the mother. We focus on the epidemiology, clinical presentation, diagnosis, and management of each infection.
Parvovirus infection and its risks to the fetus
CASE #1 Pregnant teacher exposed to fifth disease
A 28-year-old primigravid woman at 16 weeks’ gestation works as an elementary school teacher. Over the past 3 weeks, she has been exposed to 4 children who had fifth disease. She now requests evaluation because she has malaise, arthralgias, myalgias, fever of 38.2°C, and a fine lacelike erythematous rash on her trunk, arms, and cheeks.
- What is the most likely diagnosis?
- What diagnostic tests are indicated?
- Is her fetus at risk?
Epidemiology of parvovirus
Parvovirus B19 is a small, single-stranded DNA virus. It is highly contagious and is transmitted primarily by respiratory droplets. Transmission also can occur via infected blood, for example, through a blood transfusion. The incubation period is 10 to 20 days. Among adults, the individuals at greatest risk for infection are those who have close contact with young children, such as parents, day-care workers, and elementary school teachers. With sustained exposure in the household or classroom, the risk of seroconversion approaches 50%.1 Approximately 50% to 60% of reproductive-aged women have evidence of prior infection, and immunity is usually lifelong.
Clinical manifestations
The classic presentation of parvovirus infection is erythema infectiosum, also called fifth disease. This condition is characterized by a “slapped cheek” facial rash, malaise, myalgias, arthralgias, and low-grade fever. A fine lacelike rash often develops over the torso. In adults, the characteristic rash may be absent, and the most common presentation is a flu-like illness with joint pains.1,2 In children and in adults with an underlying hemoglobinopathy, parvovirus can cause transient aplastic crisis, and patients present with signs of a severe anemia, such as dyspnea, pallor, and fatigue.
Although parvovirus infection usually poses no serious risk in otherwise healthy children and adults, it can cause major fetal injury when the pregnant woman is infected early in pregnancy. The principal manifestation of fetal infection is hydrops. Hydrops primarily results when the virus crosses the placenta and attaches to the P antigen on the surface of red cell progenitors in the fetal marrow, causing an aplastic anemia with resultant high-output congestive heart failure. The virus also may directly injure the fetal myocardium, thus exacerbating heart failure. Other manifestations of congenital parvovirus include thrombocytopenia and hepatitis.3
The severity of fetal injury is inversely proportional to the gestational age at the time of maternal infection. When primary maternal infection occurs in the first trimester, the frequency of fetal hydrops is 5% to 10%. If infection develops in weeks 13 to 20, the risk of hydrops decreases to 5% or less. If infection develops beyond week 20, the incidence of fetal hydrops is 1% or lower.2
Continue to: Diagnostic steps...
Diagnostic steps
Appropriate diagnostic evaluation for a pregnant woman with exposure to parvovirus or clinical manifestations suggestive of parvovirus infection is outlined in FIGURE 1.

If infection is confirmed, serial ultrasound monitoring should be performed on a weekly to biweekly basis for 8 to 12 weeks, as delineated in FIGURE 2. Extended surveillance is necessary because the incubation period in the fetus is longer than that in the mother.
As the fetus develops anemia, peripheral tissues become hypoxic, leading to reflex peripheral vasoconstriction and increased cardiac output. At the same time, reduction in the number of fetal red blood cells decreases blood viscosity. The combination of these changes results in an increase in blood flow to the fetal brain, which can be detected by measuring the peak systolic velocity of flow in the middle cerebral artery (MCA PSV) with Doppler ultrasound imaging (FIGURE 3). The increase in MCA PSV parallels the decrease in fetal hematocrit and precedes the development of hydrops. In fact, signs of fetal hydrops do not usually develop until the fetal hematocrit falls to 15 to 20 vol%.

Management may necessitate intrauterine transfusion
Although some cases of fetal hydrops may resolve spontaneously, most authors agree that intrauterine transfusion is essential. In most instances, only a single intrauterine transfusion is necessary. In some fetuses, however, the infection is so prolonged and the anemia so severe that 2 to 3 transfusions may be required.
Infants who survive the intrauterine transfusion usually have an excellent long-term prognosis. However, isolated case reports have documented neurologic morbidity and prolonged transfusion-dependent anemia.4 In light of these reports, we recommend that a third trimester ultrasound exam be performed to assess fetal growth and evaluate the anatomy of the fetal brain. For the fetus with abnormal intracranial findings on ultrasonography, fetal magnetic resonance imaging is indicated.5
CASE #1 Diagnosis is probable parvovirus
The most likely diagnosis in this case is erythema infectiosum. This diagnosis can be confirmed by identifying positive immunogloblulin M (IgM) antibody and by detecting parvovirus in the maternal serum by polymerase chain reaction. Given the gestational age of 16 weeks, the risk of serious fetal injury should be less than 5%. Nevertheless, serial ultrasound examinations should be performed to assess for signs of fetal anemia.
Varicella exposure in pregnancy
CASE #2 Pregnant woman exposed to chickenpox has symptoms
Two weeks ago, a 32-year-old woman (G3P2002) at 24 weeks’ gestation was exposed to a neighbor’s child who had chickenpox. The patient has no history of natural infection or vaccination. She now has a fever of 38.6°C, malaise, headache, and a diffuse pruritic vesicular rash on her trunk and extremities. She also is experiencing a dry cough and mild dyspnea.
- What diagnostic tests are indicated?
- What treatment is indicated?
- What risk does this condition pose to the fetus?
Epidemiology of varicella
Varicella (chickenpox) is caused by the DNA varicella-zoster virus, an organism that is a member of the herpesvirus family. The disease occurs predominantly in children, and the infection is transmitted by respiratory droplets and by direct contact. Its incubation period is short (10–14 days), and it is highly contagious. More than 90% of susceptible close contacts will become infected after exposure to the index case. Like other herpesviruses, the varicella virus can establish a latent infection and then become manifest years later as herpes zoster (shingles).5,6
Continue to: Clinical manifestations...
Clinical manifestations
Patients with varicella usually have prodromal symptoms and signs that include malaise, fatigue, arthralgias, myalgias, and a low-grade fever. Varicella’s pathognomonic manifestation is a pruritic, macular rash that starts on the face and trunk and then spreads centripetally to the extremities. The lesions typically appear in “crops” and evolve through several distinct phases: macule, papule, vesicle, pustule, ulcer, and crust.5
In children, varicella is manifest almost entirely by mucocutaneous lesions. In adults, however, 2 serious and potentially life-threatening complications can occur. Approximately 1% of infected adults develop encephalitis and about 20% develop viral pneumonia, often accompanied by a severe superimposed bacterial pneumonia.5
When maternal infection develops in the first half of pregnancy, approximately 2% of fetuses will have evidence of congenital infection, usually manifested by circular, constricting scars on the extremities. These lesions typically occur in a dermatomal distribution. Spontaneous abortion and fetal death in utero also have been reported, but fortunately they are quite rare. When maternal infection occurs beyond 20 weeks of gestation, fetal injury is very uncommon.7
Interestingly, when maternal infection occurs at the time of delivery or shortly thereafter (from 5 days before until 2 days after delivery), neonatal varicella may develop. This infection may take 3 forms: disseminated mucocutaneous lesions, a deep-seated visceral infection, or severe pneumonia. In the era before the ready availability of antiviral agents, the case fatality rate from neonatal varicella was approximately 30%.5
Diagnosis is clinical
The diagnosis of varicella usually is established on the basis of clinical examination. It can be confirmed by identification of anti–varicella-zoster IgM.
Management includes assessing immunity
If a patient is seen for a preconception appointment, ask her whether she has ever had varicella or been vaccinated for this disease. If she is uncertain, a varicella-zoster immunoglobulin G (IgG) titer should be ordered. If the IgG titer is negative, denoting susceptibility to infection, the patient should be vaccinated before she tries to conceive (see below).8
If a patient has not had a preconception appointment and now presents for her first prenatal appointment, she should be asked about immunity to varicella. If she is uncertain, a varicella-zoster IgG assay should be obtained. Approximately 75% of patients who are uncertain about immunity will, in fact, be immune. Those who are not immune should be counseled to avoid exposure to individuals who may have varicella, and they should be targeted for vaccination immediately postpartum.5,9
If a susceptible pregnant patient has been exposed to an individual with varicella, she should receive 1 of 2 regimens within 72 to 96 hours to minimize the risk of maternal infection.5,9,10 One option is intramuscular varicella-zoster immune globulin (VariZIG), 125 U/10 kg body weight, with a maximum dose of 625 U (5 vials). The distributor of this agent is FFF Enterprises in Temucula, California (telephone: 800-843-7477). A company representative will assess the patient’s eligibility and deliver the drug within 24 hours if the patient is considered eligible. An alternative prophylactic regimen is oral acyclovir, 800 mg 5 times daily for 7 days, or oral valacyclovir, 1,000 mg 3 times daily for 7 days.
If, despite prophylaxis, the pregnant woman becomes infected, she should immediately be treated with 1 of the oral antiviral regimens described above. If she has evidence of encephalitis, pneumonia, or severe disseminated mucocutaneous infection, or if she is immunosuppressed, she should be hospitalized and treated with intravenous acyclovir, 10 mg/kg infused over 1 hour every 8 hours for 10 days.
Ultrasonography is the most valuable test to identify fetal infection. Key findings that suggest congenital varicella are fetal growth restriction, microcephaly, ventriculomegaly, echogenic foci in the liver, and limb abnormalities. There is no proven therapy for congenital varicella.
When a patient has varicella at the time of delivery, she should be isolated from her infant until all lesions have crusted over. In addition, the neonate should be treated with either VariZIG or an antiviral agent.5,9
Prevention with varicella vaccine
The varicella vaccine (Varivax) is a live-virus vaccine that is highly immunogenic. The vaccine is now part of the routine childhood immunization sequence. Children ages 1 to 12 years require only a single dose of the vaccine. Individuals older than 12 years of age require 2 doses, administered 4 to 6 weeks apart. The vaccine should not be administered during pregnancy. It also should not be administered to individuals who are severely immunocompromised, are receiving high-dose systemic steroids, have untreated tuberculosis, or have an allergy to neomycin, which is a component of the vaccine. The vaccine does not pose a risk to the breastfeeding infant.11
CASE #2 Hospitalization is recommended for this patient
The patient in this case developed acute varicella pneumonia as a result of her exposure to the neighbor’s child. The diagnosis can be confirmed by demonstrating a positive varicella-zoster IgM and by obtaining a chest x-ray that identifies the diffuse patchy infiltrates characteristic of viral pneumonia. Because this is such a potentially serious illness, the patient should be hospitalized and treated with intravenous acyclovir or valacyclovir. Antibiotics such as ceftriaxone and azithromycin may be indicated to treat superimposed bacterial pneumonia. Given the later gestational age, the fetus is at low risk for serious injury. ●
- Valeur-Jensen AK, Pedersen CB, Westergaard T, et al. Risk factors for parvovirus B19 infection in pregnancy. JAMA. 1999;281:1099-1105.
- Harger JH, Adler SP, Koch WC, et al. Prospective evaluation of 618 pregnant women exposed to parvovirus B19: risks and symptoms. Obstet Gynecol. 1998;91:413-420.
- Melamed N, Whittle W, Kelly EN, et al. Fetal thrombocytopenia in pregnancies with fetal human parvovirus-B19 infection. Am J Obstet Gynecol. 2015;212:793.e1-8.
- Nagel HTC, de Haan TR, Vandenbussche FPH, et al. Long-term outcome after fetal transfusion for hydrops associated with parvovirus B19 infection. Obstet Gynecol. 2007;109:42-47.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TR, et al (eds). Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:911-912.
- Cohen JI. Herpes zoster. N Engl J Med. 2013;369:255-263.
- Enders G, Miller E, Cradock-Watson J, et al. Consequences of varicella and herpes zoster in pregnancy: prospective study of 1739 cases. Lancet. 1994;343:1548-1551.
- Duff P. Varicella in pregnancy: five priorities for clinicians. Infect Dis Obstet Gynecol. 1994;1:163-165.
- Marin M, Guris D, Chaves SS, et al; Advisory Committee on Immunization Practices, Centers for Disease Control and Prevention. Prevention of varicella. MMWR Recommend Rep. 2007;56(RR-4):1-40.
- Swamy GK, Dotters-Katz SK. Safety and varicella outcomes after varicella zoster immune globulin administration in pregnancy. Am J Obstet Gynecol. 2019;221:655-656.
- Duff P. Varicella vaccine. Infect Dis Obstet Gynecol. 1996;4:63-65.
- Valeur-Jensen AK, Pedersen CB, Westergaard T, et al. Risk factors for parvovirus B19 infection in pregnancy. JAMA. 1999;281:1099-1105.
- Harger JH, Adler SP, Koch WC, et al. Prospective evaluation of 618 pregnant women exposed to parvovirus B19: risks and symptoms. Obstet Gynecol. 1998;91:413-420.
- Melamed N, Whittle W, Kelly EN, et al. Fetal thrombocytopenia in pregnancies with fetal human parvovirus-B19 infection. Am J Obstet Gynecol. 2015;212:793.e1-8.
- Nagel HTC, de Haan TR, Vandenbussche FPH, et al. Long-term outcome after fetal transfusion for hydrops associated with parvovirus B19 infection. Obstet Gynecol. 2007;109:42-47.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TR, et al (eds). Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:911-912.
- Cohen JI. Herpes zoster. N Engl J Med. 2013;369:255-263.
- Enders G, Miller E, Cradock-Watson J, et al. Consequences of varicella and herpes zoster in pregnancy: prospective study of 1739 cases. Lancet. 1994;343:1548-1551.
- Duff P. Varicella in pregnancy: five priorities for clinicians. Infect Dis Obstet Gynecol. 1994;1:163-165.
- Marin M, Guris D, Chaves SS, et al; Advisory Committee on Immunization Practices, Centers for Disease Control and Prevention. Prevention of varicella. MMWR Recommend Rep. 2007;56(RR-4):1-40.
- Swamy GK, Dotters-Katz SK. Safety and varicella outcomes after varicella zoster immune globulin administration in pregnancy. Am J Obstet Gynecol. 2019;221:655-656.
- Duff P. Varicella vaccine. Infect Dis Obstet Gynecol. 1996;4:63-65.



