User login
The enigma of chronic pelvic pain: Systematically tracing the cause
- If thorough investigation yields no diagnosis or indications for immediate surgery, empiric medical therapy for endometriosis is appropriate without laparoscopic confirmation. If empiric medical therapy fails, proceed to diagnostic laparoscopy.
- Referral to a multidisciplinary pain clinic has been shown to be more effective than episodic gynecologic management of patients, especially those with significant psychological issues.
- Schedule regular follow-ups. Do not instruct patients to call only during a pain crisis—this practice may create pain behaviors directed at obtaining sympathy and dramatic medical attention.
One woman complains of daily pain that worsens premenstrually. Another reports frequent aches radiating through her lower back and abdomen. A third says intercourse exacerbates her pain. And the list goes on.
Chronic pelvic pain (CPP) can manifest in dozens of ways, and the words patients use to describe it vary just as widely. Its multifaceted nature poses one of the biggest challenges to accurate diagnosis and appropriate management. Success is most likely when a compassionate attitude is combined with a systematic assessment to identify and understand the underlying cause—a blend of artful practice and scientific reasoning.
This article draws from consensus guidelines, other data, and personal experience to describe the components of careful diagnosis of this common but elusive condition.
10% of gynecology visits
CPP, which refers to pelvic pain of more than 6 months’ duration, accounts for roughly 10% of a gynecologist’s outpatient encounters, as well as many invasive procedures. For example, 1 in 5 gynecologic laparoscopies and 15% to 20% of hysterectomies are performed solely or in part for CPP.1,2
Noncyclic pelvic pain is not necessarily gynecologic
CPP is not always of gynecologic origin, even though the patient may perceive the pain as emanating from the reproductive organs. Because of this, careful evaluation is needed to distinguish gynecologic pain from CPP caused by orthopedic, gastrointestinal (GI), urologic, or neurologic conditions, or CPP that has a psychosomatic basis.
Purely cyclic uterine pain, which is referred to as dysmenorrhea, is usually characterized as primary or secondary. Although dysmenorrhea is frequently chronic and is an important clinical problem, this review focuses on noncyclic pelvic pain.
The relationship between CPP and underlying gynecologic pathology is often enigmatic. Many clinicians attribute the pain to psychological causes before completing a thorough assessment. An example of the hazards inherent in such an attitude is primary dysmenorrhea: Before the primary role of prostaglandins was elucidated, this form of dysmenorrhea was thought to be largely psychosomatic. As a result, it went untreated.
The TABLE lists major gynecologic and nongynecologic causes of noncyclic CPP. Endometriosis is the most common gynecologic condition associated with CPP.
TABLE
Causes of noncyclic chronic pelvic pain
| GYNECOLOGIC CAUSES |
| Endometriosis |
| Chronic pelvic inflammatory disease |
| Ovarian |
| Rapid ovarian capsule distension |
| Ovarian remnant syndrome |
| Recurrent hemorrhagic ovarian cysts |
| Uterine (uncommon) |
| Adenomyosis |
| Uterine myomas (under certain circumstances) |
| Uterine retroversion (if associated with other intraperitoneal pathology or dyspareunia) |
| Pelvic congestion syndrome |
| NONGYNECOLOGIC CAUSES |
| Genitourinary |
| Interstitial cystitis |
| Trigonitis |
| Urinary retention |
| Urethral syndrome |
| Gastrointestinal |
| Crohn disease |
| Diverticulitis |
| Hernia formation |
| Irritable bowel syndrome |
| Inflammatory bowel disease |
| Neoplasm |
| Partial bowel obstruction |
| Ulcerative colitis |
| Musculoskeletal (eg, low back pain) |
| Psychological factors |
Questions to elicit useful clues
The patient’s description of the location and pattern of pain, as well as history, are invaluable clues.
10 key questions. To elicit the most useful information from the patient, it may be necessary to ask numerous specifically phrased questions, such as:
- What is the the exact location of yourpain?
- What is the quality of the pain (sharp? dull?)
- Does it radiate, or spread, to other areas of the body? If so, where?
- How many times have you experienced this pain?
- How long does each episode last?
- How intense is each episode?
- What makes the pain worse? What makes it feel better?
- Does the pain change with your menstrual cycle (or with bowel movements, urination, sexual intercourse, or physical activity)?
- How much, and how, does the pain interfere with your daily activities?
- How would you rate the severity of the pain on a scale of 0 to 10, where 0 = no pain and 10 = the worst pain imaginable?
A complete medication history, especially of pain medication, is important.
Medical history should include information about nongynecologic conditions that may account for CPP, such as irritable bowel syndrome, ulcerative colitis, Crohn disease, and interstitial cystitis.
Pending compensation or litigation issues should be recognized and addressed.
Patient’s depiction of pain may lead to source
Understanding the anatomy of the nerves that transmit pain can facilitate interpretation of patient reports. It may explain, for example, why GI pathology can cause the perception of gynecologic pain. See “The anatomy of pelvic pain perception”.
- Pain relieved by defecation as well as irregular defecation patterns lasting at least 3 months suggest irritable bowel syndrome.
- Urinary urgency, frequency, nocturia, and pelvic pain may point to early interstitial cystitis.
- In multiparous women, pelvic pain that worsens premenstrually and with fatigue, standing, and sexual intercourse indicates possible pelvic congestion syndrome. Venographic studies may document pelvic vein varicosities in such cases.
Convergence of sensation
Somatic pain. Painful impulses originating in the skin, muscles, bones, joints, and parietal peritoneum travel in somatic nerve fibers.
Visceral pain. Painful impulses originating in the internal organs travel in visceral nerves.
Visceral pain is more diffusely localized than somatic pain and has a less well-defined area of projection to the sensory cortex of the brain for its identification. Also, there are many more somatic neurons than viscerosomatic ones in the dorsal horn of the spinal cord.
These differences result in a convergence of sensation that may be difficult for the patient and physician to interpret. Visceral pain is usually interpreted as coming from the skin and other surface tissues, which are supplied by the corresponding spinal cord segment, resulting in referred pain. For example, the initial pain of appendicitis is perceived to be in the epigastric area, because both are innervated by the thoracic cord segments T8, T9, and T10.
Since innervation of the lower intestinal tract is the same as that of the uterus and fallopian tubes, patients may experience pain caused by GI pathology as gynecologic pain.
Pain sensitivity of genital tissues
- External genitalia are exquisitely sensitive.
- Vaginal pain sensation is variable.
- The cervix is relatively insensitive to small biopsies but is sensitive to deep incision or to dilatation.
- The uterus is very sensitive.
- The ovaries are insensitive to many stimuli, but they are sensitive to rapid distension of the ovarian capsule and compression during physical examination.
Reproduce the pain on physical examination
Examination should be conducted in a way that does not provoke involuntary guarding, which may obscure findings. Gaining trust beforehand is important, as is taking time to explain what will be done during the exam.
Ask the patient to show you where the pain occurs, if possible, and try to reproduce the pain by palpation. Look for abdominal distension, and locate any tender areas. Continue the pelvic examination in this way, always attempting to reproduce and localize the pain. Look for any tenderness, nodularity (especially in the cul-de-sac), and palpable masses in the cul-de-sac or adnexal regions.
Examine the abdominal wall for evidence of myofascial trigger points and for iliohypogastric (T12, L1), ilioinguinal (T12, L1), or genitofemoral (L1, L2) nerve entrapment. Then ask the patient to tense the abdominal muscles by performing a straight-leg–raising maneuver or a partial sit-up. Points that are still tender, more tender, or that reproduce the patient’s pain after these maneuvers should be injected with 2 to 3 mL of 2.5% bupivacaine. Chronic abdominal wall pain is confirmed if the pain level is reduced by at least 50%.
Chronic low back pain without abdominal pain is seldom of gynecologic origin. If there is evidence of musculoskeletal disease, consider consulting an orthopedist. Neuromuscular symptoms may be accompanied by a pelvic mass. Positive neurologic signs such as muscle weakness or abnormal reflexes should prompt consultation. In such cases, surgical exploration may reveal a neuroma or bony tumor.
Lab, imaging studies: How useful?
Traditional laboratory studies are of limited value, although a complete blood cell count, erythrocyte sedimentation rate (ESR), and urinalysis should be obtained. The ESR is nonspecific and will be elevated in any type of inflammatory condition, such as subacute salpingo-oophoritis, tuberculosis, or inflammatory bowel disease.
A thorough genitourinary evaluation is important, including cystoscopy when suspicion of urinary tract disease is high. If no obvious cause for the pain is uncovered, pelvic ultrasonography may be helpful. This is especially true in an obese or uncooperative patient, when bimanual pelvic examination is difficult.
If bowel or urinary signs and symptoms are present, then an endoscopy, abdominal and pelvic computed tomography, cystoscopy, or computed tomography urogram may be useful.
No diagnosis, no reason for surgery? Start therapy for endometriosis
If the workup yields no diagnosis and rules out indications for surgery, such as significant adnexal mass, then empiric medical therapy for endometriosis is appropriate without laparoscopic confirmation.1
Begin a trial of empiric therapy with a nonsteroidal anti-inflammatory drug such as ibuprofen or naproxen, oral contraceptives, or both.1 Women with ovarian pathology (eg, periovarian adhesions, recurrent functional cyst formation) and those whose pain worsens midcycle, premenstrually, or menstrually may experience improvement with a trial of oral contraceptives to suppress ovulation and menstruation.
Use second-line empiric therapy before diagnostic laparoscopy
If these treatments fail, begin empiric therapy with second-line agents such as danazol, progestins, or gonadotropin-releasing hormone (GnRH) agonists. Laparoscopic confirmation is unnecessary.
Several publications, including a 2002 consensus statement on management of chronic pelvic pain and endometriosis, support this approach.1,3-5 In fact, following recent guidelines for evaluating and treating CPP is likely to lead to fewer invasive procedures for diagnosis and treatment, including laparoscopy and hysterectomy.
Surgical ablative therapy has not been shown superior to medical therapy in addressing the pain of early-stage endometriosis. Medical therapy is less invasive and, as the following report illustrates, it probably treats all disease present.
In a study of 95 women with CPP in whom endometriosis was suspected after a thorough noninvasive workup yielded no diagnosis, investigators found that empiric therapy with depot leuprolide for 12 weeks provided significant pain relief compared with both the baseline and a placebo group (P ≤ .001 for both comparisons). Posttreatment diagnostic laparoscopy confirmed endometriosis in more than three quarters of both treatment groups.3
The limits of laparoscopy
Although second-line empiric therapy is controversial, laparoscopy is invasive and does not necessarily yield a definitive diagnosis. However, diagnostic laparoscopy may ultimately be indicated in patients with pain of undetermined cause. Perform laparoscopy only if no cause of pain can be identified and confirmed, and after reasonable trials of empiric medical therapy.
Up to one third of women have no pathology visualized on laparoscopy.6 This does not mean that no pathology is present. Endometriosis is missed visually during laparoscopy in up to one third of CPP patients in whom the disease is proven on biopsy.3,4
Laparoscopically identified pathology does not necessarily make the diagnosis. For example, the size and location of endometriotic implants do not always correlate with the presence of pain. Thus, endometriosis may be present but may not be the cause of CPP.1 Similarly, visualizing pelvic adhesions during laparoscopy does not necessarily mean that the adhesions are responsible for the pelvic pain, as women with extensive adhesions are sometimes asymptomatic.7
Gynecologic conditions identifiable by diagnostic laparoscopy
The following gynecologic conditions may be identified through diagnostic laparoscopy in women with cyclic and noncyclic pain:
Endometriosis is seen in at least one third to one half of women with CPP7; most sites of endometriosis are in the pelvic area. The mechanisms by which endometriosis causes pain are not fully understood, however.
Chronic pelvic inflammatory disease (PID). Laparoscopy identifies adhesions or stigmata of chronic PID in about 25% of women with CPP. Chronic PID may cause noncyclic CPP because recurrent exacerbations frequently result in hydrosalpinges and adhesions between the tubes, ovaries, and intestinal structures. Before ascribing symptoms to adhesions, however, clinicians should specifically note this pathology in the area of pain.
Ovarian causes. Ovarian cysts are frequently asymptomatic, but pain may result from rapid distention of the ovarian capsule. An ovary or ovarian remnant may enter the retroperitoneal space secondary to inflammation or previous surgery. This is known as ovarian remnant syndrome, and cyst formation in these circumstances is usually painful. Some women, for unknown reasons, may develop multiple recurrent hemorrhagic ovarian cysts that are associated with pelvic pain and dyspareunia.
Uterine origins. Adenomyosis (endometrial glands and stroma invading the myometrium) may cause dysmenorrhea and menorrhagia, but rarely causes noncyclic CPP. Uterine myomas usually do not cause pelvic pain unless they are undergoing degeneration, torsion, or are compressing pelvic nerves. (Occasionally, a submucous leiomyoma may protrude through the cervix, which has been described by women as pain not unlike childbirth.) Uterine myomas may also cause pain from rapid growth or degeneration during pregnancy.
Chronic pain itself—with or without organic pathology—produces debilitating psychological responses.
Pelvic pain is not likely to be due solely to variations in uterine position, but dyspareunia with deep penetration may occasionally be associated with uterine retroversion. A tender uterus that is in a fixed retroverted position usually signifies other intraperitoneal pathology, such as endometriosis or PID.
Pelvic congestion syndrome. As noted, this has been described in multiparous women who have pelvic vein varicosities (documented on venographic studies) resulting in “congested” pelvic organs. Many women with this condition are noted to have a uterus that is mobile, retroverted, soft, boggy, and slightly enlarged. Ovarian hormone suppression and cognitive behavioral therapy may relieve pain. If these measures fail, and if venographic studies confirm the diagnosis, then hysterectomy and oophorectomy may be beneficial. Surgery should be considered only if the diagnosis is confirmed and medical therapy fails.
If no pathology is found, is the pain psychological?
Inability to identify a pathologic diagnosis in approximately one third of patients with CPP has led to the postulation of a psychological etiology. It is appropriate to consider psychological factors early in your evaluation. However, as mentioned earlier, a suggestion of psychological etiology should not delay or preclude further investigation of somatic pathology.
Ask patients about past or present physical, sexual, or emotional abuse. History of major psychosexual trauma (molestation, incest, rape) was identified in 48% of 106 women evaluated at my institution with no pathologic diagnosis following a thorough workup and laparoscopy.2 Women with such a history should be referred for psychological evaluation.
Secondary gain often complicates diagnosis and therapy. Ask about current life events as well as any noteworthy happenings when the pain began. Consider the possibility of secondary gain or other psychological benefits of pain behaviors. For example, exaggerated response to pain and the reaction of significant others may be so reinforcing that pain behavior persists even after the painful stimulus has resolved. If you suspect this may be an issue, the patient should undergo psychological evaluation.
Pain may be psychologically debilitating. Evidence suggests that chronic pain itself, with or without organic pathology, produces debilitating psychological responses. Patients with such pain tend to feel depressed, helpless, and passive. They withdraw from social and sexual activity, and are preoccupied with pain and suffering.7 Personality testing of CPP patients without identified organic pathology shows characteristics similar to those of chronic pain patients with organic pathology. Specifically, on the Minnesota Multiphasic Personality Inventory, both these groups demonstrate a greater degree of anxiety, hypochondriasis, and hysteria than do control subjects.
Psychological response can affect organic processing of pain. A person’s psychological response to pain can influence how the nervous system transmits pain. Many neurotransmitters and neuromodulators can amplify or diminish pain impulses before pain signals reach the central nervous system. Anxiety and other psychological states may affect release and metabolism of these neurotransmitters, thus shaping perception of pain. Many forms of CPP, especially those with little or no pathology identified, may result primarily from disordered pain perception due to modulation of afferent impulses in the nervous system.
Excitatory modulators include substance P, glutamate, aspartate, calcitonin generelated peptide, and vasoactive intestinal peptide. Inhibitory neuromediators include endogenous opioid peptides, norepinephrine, serotonin (5-HT), and gamma-aminobutyric acid (GABA). Tricyclic antidepressants, other GABA-ergic agents, and selective serotonin reuptake inhibitors can increase inhibitory neuromediators, making them very useful in the management of CPP.
Referral to a multidisciplinary pain clinic is the most productive strategy for patients with noncyclic CPP and significant psychological issues.7 All patients with mental illness should be referred for psychological treatment.
A multidisciplinary facility typically includes a psychologist who also has expertise in sexual and marital counseling, an anesthesiologist, and—ideally—an acupuncturist, in addition to a gynecologist. Relaxation training, along with cognitive and behavioral therapies, can help train the patient to replace abnormal pain behavior and its secondary gain with effective, adaptive behavioral responses. Research has shown multidisciplinary management to be more effective than traditional episodic gynecologic management.
When a multidisciplinary pain clinic is unavailable, the gynecologist should involve other specialists by referral while coordinating the overall care of the patient.
What to do, what not to do when the cause is organic
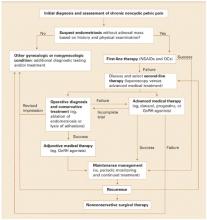
Patients in whom an organic pathology is identified should be treated accordingly. Recent publications detail management strategies for endometriosis associated with CPP.1,5 For example, the algorithm in the Figure is based on a 2002 consensus statement.1
Procedures to avoid surgery in the absence of pathology. Without proof of organic pathology or a reasonable functional explanation for the pelvic pain, a thorough psychosomatic evaluation should be completed before a surgical corrective procedure is considered.
These procedures have not proven effective for noncyclic CPP without pathology:
- unilateral adnexectomy for unilateral pain
- total abdominal hysterectomy
- presacral neurectomy
- uterine suspension for generalized pelvic pain
- lysis of adhesions, which is usually nonproductive unless the site of laparoscopically visualized adhesions specifically coincides with the site of the local pain. Pelvic adhesions often recur following surgical lysis.
Another option: Acupuncture and nerve blocks. These strategies can provide prolonged pain relief in properly selected patients. Consultation with an anesthesiologist who is trained in chronic pain management can identify appropriate candidates. Acupuncture likely works by increasing spinal cord endorphins. Patients with trigger points usually respond well to injection of local anesthetics.
FIGURE Assessment, diagnosis, and treatment options for chronic pelvic pain and presumed endometriosis
GnRH=gonadotropin-releasing hormone; NSAID=nonsteroidal antiinflammatory drug; OC=oral contraceptive Adapted from Fertil Steril; vol 78; Gambone JC, Mittman BS, Munro MG, Scialli AR, Winkel CA; Consensus statement for the management of chronic pelvic pain and endometriosis: proceedings of an expert-panel consensus process; 961-972; copyright 2002; with permission from American Society for Reproductive Medicine.
To prevent pain behaviors, schedule follow-up visits
In managing all patients with noncyclic CPP, take a supportive, sympathetic, and structured approach. This disorder is optimally treated in the context of an ongoing disease-management protocol rather than episodic therapy initiated because of a pain crisis.
Provide the patient with regular follow-up appointments; do not instruct her to call only when pain persists or recurs. The latter practice may create pain behaviors directed at obtaining sympathy and dramatic medical attention in certain patients.
Dr. Gambone reports no financial relationship with any companies whose products are mentioned in this article.
1. Gambone JC, Mittman BS, Munro MG, Scialli AR, Winkel CA. Consensus statement for the management of chronic pelvic pain and endometriosis: proceedings of an expert-panel consensus process. Fertil Steril. 2002;78:961-972.
2. Reiter RC, Gambone JC. Demographic and historic variables in women with idiopathic chronic pelvic pain. Obstet Gynecol. 1990;75:428-432.
3. Ling F. Pelvic Pain Study Group. Randomized controlled trial of depot leuprolide in patients with chronic pelvic pain and clinically suspected endometriosis. Obstet Gynecol. 1999;93:51-58.
4. Winkel CA. Looking for a better way to manage endometriosis-related pain. Contemp Ob/Gyn. 2003;48:44-56.
5. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin #11: Medical Management of Endometriosis. Washington, DC: ACOG; December 1999.
6. Rapkin AJ. Chronic pelvic pain. In: Hacker N, Moore JG, eds. Essentials of Obstetrics and Gynecology. Philadelphia, Pa: WB Saunders; 1996;499-506.
7. Reiter RC, Gambone JC. Nongynecologic somatic pathology in women with chronic pelvic pain and negative laparoscopy. J Reprod Med. 1991;36:253-259.
- If thorough investigation yields no diagnosis or indications for immediate surgery, empiric medical therapy for endometriosis is appropriate without laparoscopic confirmation. If empiric medical therapy fails, proceed to diagnostic laparoscopy.
- Referral to a multidisciplinary pain clinic has been shown to be more effective than episodic gynecologic management of patients, especially those with significant psychological issues.
- Schedule regular follow-ups. Do not instruct patients to call only during a pain crisis—this practice may create pain behaviors directed at obtaining sympathy and dramatic medical attention.
One woman complains of daily pain that worsens premenstrually. Another reports frequent aches radiating through her lower back and abdomen. A third says intercourse exacerbates her pain. And the list goes on.
Chronic pelvic pain (CPP) can manifest in dozens of ways, and the words patients use to describe it vary just as widely. Its multifaceted nature poses one of the biggest challenges to accurate diagnosis and appropriate management. Success is most likely when a compassionate attitude is combined with a systematic assessment to identify and understand the underlying cause—a blend of artful practice and scientific reasoning.
This article draws from consensus guidelines, other data, and personal experience to describe the components of careful diagnosis of this common but elusive condition.
10% of gynecology visits
CPP, which refers to pelvic pain of more than 6 months’ duration, accounts for roughly 10% of a gynecologist’s outpatient encounters, as well as many invasive procedures. For example, 1 in 5 gynecologic laparoscopies and 15% to 20% of hysterectomies are performed solely or in part for CPP.1,2
Noncyclic pelvic pain is not necessarily gynecologic
CPP is not always of gynecologic origin, even though the patient may perceive the pain as emanating from the reproductive organs. Because of this, careful evaluation is needed to distinguish gynecologic pain from CPP caused by orthopedic, gastrointestinal (GI), urologic, or neurologic conditions, or CPP that has a psychosomatic basis.
Purely cyclic uterine pain, which is referred to as dysmenorrhea, is usually characterized as primary or secondary. Although dysmenorrhea is frequently chronic and is an important clinical problem, this review focuses on noncyclic pelvic pain.
The relationship between CPP and underlying gynecologic pathology is often enigmatic. Many clinicians attribute the pain to psychological causes before completing a thorough assessment. An example of the hazards inherent in such an attitude is primary dysmenorrhea: Before the primary role of prostaglandins was elucidated, this form of dysmenorrhea was thought to be largely psychosomatic. As a result, it went untreated.
The TABLE lists major gynecologic and nongynecologic causes of noncyclic CPP. Endometriosis is the most common gynecologic condition associated with CPP.
TABLE
Causes of noncyclic chronic pelvic pain
| GYNECOLOGIC CAUSES |
| Endometriosis |
| Chronic pelvic inflammatory disease |
| Ovarian |
| Rapid ovarian capsule distension |
| Ovarian remnant syndrome |
| Recurrent hemorrhagic ovarian cysts |
| Uterine (uncommon) |
| Adenomyosis |
| Uterine myomas (under certain circumstances) |
| Uterine retroversion (if associated with other intraperitoneal pathology or dyspareunia) |
| Pelvic congestion syndrome |
| NONGYNECOLOGIC CAUSES |
| Genitourinary |
| Interstitial cystitis |
| Trigonitis |
| Urinary retention |
| Urethral syndrome |
| Gastrointestinal |
| Crohn disease |
| Diverticulitis |
| Hernia formation |
| Irritable bowel syndrome |
| Inflammatory bowel disease |
| Neoplasm |
| Partial bowel obstruction |
| Ulcerative colitis |
| Musculoskeletal (eg, low back pain) |
| Psychological factors |
Questions to elicit useful clues
The patient’s description of the location and pattern of pain, as well as history, are invaluable clues.
10 key questions. To elicit the most useful information from the patient, it may be necessary to ask numerous specifically phrased questions, such as:
- What is the the exact location of yourpain?
- What is the quality of the pain (sharp? dull?)
- Does it radiate, or spread, to other areas of the body? If so, where?
- How many times have you experienced this pain?
- How long does each episode last?
- How intense is each episode?
- What makes the pain worse? What makes it feel better?
- Does the pain change with your menstrual cycle (or with bowel movements, urination, sexual intercourse, or physical activity)?
- How much, and how, does the pain interfere with your daily activities?
- How would you rate the severity of the pain on a scale of 0 to 10, where 0 = no pain and 10 = the worst pain imaginable?
A complete medication history, especially of pain medication, is important.
Medical history should include information about nongynecologic conditions that may account for CPP, such as irritable bowel syndrome, ulcerative colitis, Crohn disease, and interstitial cystitis.
Pending compensation or litigation issues should be recognized and addressed.
Patient’s depiction of pain may lead to source
Understanding the anatomy of the nerves that transmit pain can facilitate interpretation of patient reports. It may explain, for example, why GI pathology can cause the perception of gynecologic pain. See “The anatomy of pelvic pain perception”.
- Pain relieved by defecation as well as irregular defecation patterns lasting at least 3 months suggest irritable bowel syndrome.
- Urinary urgency, frequency, nocturia, and pelvic pain may point to early interstitial cystitis.
- In multiparous women, pelvic pain that worsens premenstrually and with fatigue, standing, and sexual intercourse indicates possible pelvic congestion syndrome. Venographic studies may document pelvic vein varicosities in such cases.
Convergence of sensation
Somatic pain. Painful impulses originating in the skin, muscles, bones, joints, and parietal peritoneum travel in somatic nerve fibers.
Visceral pain. Painful impulses originating in the internal organs travel in visceral nerves.
Visceral pain is more diffusely localized than somatic pain and has a less well-defined area of projection to the sensory cortex of the brain for its identification. Also, there are many more somatic neurons than viscerosomatic ones in the dorsal horn of the spinal cord.
These differences result in a convergence of sensation that may be difficult for the patient and physician to interpret. Visceral pain is usually interpreted as coming from the skin and other surface tissues, which are supplied by the corresponding spinal cord segment, resulting in referred pain. For example, the initial pain of appendicitis is perceived to be in the epigastric area, because both are innervated by the thoracic cord segments T8, T9, and T10.
Since innervation of the lower intestinal tract is the same as that of the uterus and fallopian tubes, patients may experience pain caused by GI pathology as gynecologic pain.
Pain sensitivity of genital tissues
- External genitalia are exquisitely sensitive.
- Vaginal pain sensation is variable.
- The cervix is relatively insensitive to small biopsies but is sensitive to deep incision or to dilatation.
- The uterus is very sensitive.
- The ovaries are insensitive to many stimuli, but they are sensitive to rapid distension of the ovarian capsule and compression during physical examination.
Reproduce the pain on physical examination
Examination should be conducted in a way that does not provoke involuntary guarding, which may obscure findings. Gaining trust beforehand is important, as is taking time to explain what will be done during the exam.
Ask the patient to show you where the pain occurs, if possible, and try to reproduce the pain by palpation. Look for abdominal distension, and locate any tender areas. Continue the pelvic examination in this way, always attempting to reproduce and localize the pain. Look for any tenderness, nodularity (especially in the cul-de-sac), and palpable masses in the cul-de-sac or adnexal regions.
Examine the abdominal wall for evidence of myofascial trigger points and for iliohypogastric (T12, L1), ilioinguinal (T12, L1), or genitofemoral (L1, L2) nerve entrapment. Then ask the patient to tense the abdominal muscles by performing a straight-leg–raising maneuver or a partial sit-up. Points that are still tender, more tender, or that reproduce the patient’s pain after these maneuvers should be injected with 2 to 3 mL of 2.5% bupivacaine. Chronic abdominal wall pain is confirmed if the pain level is reduced by at least 50%.
Chronic low back pain without abdominal pain is seldom of gynecologic origin. If there is evidence of musculoskeletal disease, consider consulting an orthopedist. Neuromuscular symptoms may be accompanied by a pelvic mass. Positive neurologic signs such as muscle weakness or abnormal reflexes should prompt consultation. In such cases, surgical exploration may reveal a neuroma or bony tumor.
Lab, imaging studies: How useful?
Traditional laboratory studies are of limited value, although a complete blood cell count, erythrocyte sedimentation rate (ESR), and urinalysis should be obtained. The ESR is nonspecific and will be elevated in any type of inflammatory condition, such as subacute salpingo-oophoritis, tuberculosis, or inflammatory bowel disease.
A thorough genitourinary evaluation is important, including cystoscopy when suspicion of urinary tract disease is high. If no obvious cause for the pain is uncovered, pelvic ultrasonography may be helpful. This is especially true in an obese or uncooperative patient, when bimanual pelvic examination is difficult.
If bowel or urinary signs and symptoms are present, then an endoscopy, abdominal and pelvic computed tomography, cystoscopy, or computed tomography urogram may be useful.
No diagnosis, no reason for surgery? Start therapy for endometriosis
If the workup yields no diagnosis and rules out indications for surgery, such as significant adnexal mass, then empiric medical therapy for endometriosis is appropriate without laparoscopic confirmation.1
Begin a trial of empiric therapy with a nonsteroidal anti-inflammatory drug such as ibuprofen or naproxen, oral contraceptives, or both.1 Women with ovarian pathology (eg, periovarian adhesions, recurrent functional cyst formation) and those whose pain worsens midcycle, premenstrually, or menstrually may experience improvement with a trial of oral contraceptives to suppress ovulation and menstruation.
Use second-line empiric therapy before diagnostic laparoscopy
If these treatments fail, begin empiric therapy with second-line agents such as danazol, progestins, or gonadotropin-releasing hormone (GnRH) agonists. Laparoscopic confirmation is unnecessary.
Several publications, including a 2002 consensus statement on management of chronic pelvic pain and endometriosis, support this approach.1,3-5 In fact, following recent guidelines for evaluating and treating CPP is likely to lead to fewer invasive procedures for diagnosis and treatment, including laparoscopy and hysterectomy.
Surgical ablative therapy has not been shown superior to medical therapy in addressing the pain of early-stage endometriosis. Medical therapy is less invasive and, as the following report illustrates, it probably treats all disease present.
In a study of 95 women with CPP in whom endometriosis was suspected after a thorough noninvasive workup yielded no diagnosis, investigators found that empiric therapy with depot leuprolide for 12 weeks provided significant pain relief compared with both the baseline and a placebo group (P ≤ .001 for both comparisons). Posttreatment diagnostic laparoscopy confirmed endometriosis in more than three quarters of both treatment groups.3
The limits of laparoscopy
Although second-line empiric therapy is controversial, laparoscopy is invasive and does not necessarily yield a definitive diagnosis. However, diagnostic laparoscopy may ultimately be indicated in patients with pain of undetermined cause. Perform laparoscopy only if no cause of pain can be identified and confirmed, and after reasonable trials of empiric medical therapy.
Up to one third of women have no pathology visualized on laparoscopy.6 This does not mean that no pathology is present. Endometriosis is missed visually during laparoscopy in up to one third of CPP patients in whom the disease is proven on biopsy.3,4
Laparoscopically identified pathology does not necessarily make the diagnosis. For example, the size and location of endometriotic implants do not always correlate with the presence of pain. Thus, endometriosis may be present but may not be the cause of CPP.1 Similarly, visualizing pelvic adhesions during laparoscopy does not necessarily mean that the adhesions are responsible for the pelvic pain, as women with extensive adhesions are sometimes asymptomatic.7
Gynecologic conditions identifiable by diagnostic laparoscopy
The following gynecologic conditions may be identified through diagnostic laparoscopy in women with cyclic and noncyclic pain:
Endometriosis is seen in at least one third to one half of women with CPP7; most sites of endometriosis are in the pelvic area. The mechanisms by which endometriosis causes pain are not fully understood, however.
Chronic pelvic inflammatory disease (PID). Laparoscopy identifies adhesions or stigmata of chronic PID in about 25% of women with CPP. Chronic PID may cause noncyclic CPP because recurrent exacerbations frequently result in hydrosalpinges and adhesions between the tubes, ovaries, and intestinal structures. Before ascribing symptoms to adhesions, however, clinicians should specifically note this pathology in the area of pain.
Ovarian causes. Ovarian cysts are frequently asymptomatic, but pain may result from rapid distention of the ovarian capsule. An ovary or ovarian remnant may enter the retroperitoneal space secondary to inflammation or previous surgery. This is known as ovarian remnant syndrome, and cyst formation in these circumstances is usually painful. Some women, for unknown reasons, may develop multiple recurrent hemorrhagic ovarian cysts that are associated with pelvic pain and dyspareunia.
Uterine origins. Adenomyosis (endometrial glands and stroma invading the myometrium) may cause dysmenorrhea and menorrhagia, but rarely causes noncyclic CPP. Uterine myomas usually do not cause pelvic pain unless they are undergoing degeneration, torsion, or are compressing pelvic nerves. (Occasionally, a submucous leiomyoma may protrude through the cervix, which has been described by women as pain not unlike childbirth.) Uterine myomas may also cause pain from rapid growth or degeneration during pregnancy.
Chronic pain itself—with or without organic pathology—produces debilitating psychological responses.
Pelvic pain is not likely to be due solely to variations in uterine position, but dyspareunia with deep penetration may occasionally be associated with uterine retroversion. A tender uterus that is in a fixed retroverted position usually signifies other intraperitoneal pathology, such as endometriosis or PID.
Pelvic congestion syndrome. As noted, this has been described in multiparous women who have pelvic vein varicosities (documented on venographic studies) resulting in “congested” pelvic organs. Many women with this condition are noted to have a uterus that is mobile, retroverted, soft, boggy, and slightly enlarged. Ovarian hormone suppression and cognitive behavioral therapy may relieve pain. If these measures fail, and if venographic studies confirm the diagnosis, then hysterectomy and oophorectomy may be beneficial. Surgery should be considered only if the diagnosis is confirmed and medical therapy fails.
If no pathology is found, is the pain psychological?
Inability to identify a pathologic diagnosis in approximately one third of patients with CPP has led to the postulation of a psychological etiology. It is appropriate to consider psychological factors early in your evaluation. However, as mentioned earlier, a suggestion of psychological etiology should not delay or preclude further investigation of somatic pathology.
Ask patients about past or present physical, sexual, or emotional abuse. History of major psychosexual trauma (molestation, incest, rape) was identified in 48% of 106 women evaluated at my institution with no pathologic diagnosis following a thorough workup and laparoscopy.2 Women with such a history should be referred for psychological evaluation.
Secondary gain often complicates diagnosis and therapy. Ask about current life events as well as any noteworthy happenings when the pain began. Consider the possibility of secondary gain or other psychological benefits of pain behaviors. For example, exaggerated response to pain and the reaction of significant others may be so reinforcing that pain behavior persists even after the painful stimulus has resolved. If you suspect this may be an issue, the patient should undergo psychological evaluation.
Pain may be psychologically debilitating. Evidence suggests that chronic pain itself, with or without organic pathology, produces debilitating psychological responses. Patients with such pain tend to feel depressed, helpless, and passive. They withdraw from social and sexual activity, and are preoccupied with pain and suffering.7 Personality testing of CPP patients without identified organic pathology shows characteristics similar to those of chronic pain patients with organic pathology. Specifically, on the Minnesota Multiphasic Personality Inventory, both these groups demonstrate a greater degree of anxiety, hypochondriasis, and hysteria than do control subjects.
Psychological response can affect organic processing of pain. A person’s psychological response to pain can influence how the nervous system transmits pain. Many neurotransmitters and neuromodulators can amplify or diminish pain impulses before pain signals reach the central nervous system. Anxiety and other psychological states may affect release and metabolism of these neurotransmitters, thus shaping perception of pain. Many forms of CPP, especially those with little or no pathology identified, may result primarily from disordered pain perception due to modulation of afferent impulses in the nervous system.
Excitatory modulators include substance P, glutamate, aspartate, calcitonin generelated peptide, and vasoactive intestinal peptide. Inhibitory neuromediators include endogenous opioid peptides, norepinephrine, serotonin (5-HT), and gamma-aminobutyric acid (GABA). Tricyclic antidepressants, other GABA-ergic agents, and selective serotonin reuptake inhibitors can increase inhibitory neuromediators, making them very useful in the management of CPP.
Referral to a multidisciplinary pain clinic is the most productive strategy for patients with noncyclic CPP and significant psychological issues.7 All patients with mental illness should be referred for psychological treatment.
A multidisciplinary facility typically includes a psychologist who also has expertise in sexual and marital counseling, an anesthesiologist, and—ideally—an acupuncturist, in addition to a gynecologist. Relaxation training, along with cognitive and behavioral therapies, can help train the patient to replace abnormal pain behavior and its secondary gain with effective, adaptive behavioral responses. Research has shown multidisciplinary management to be more effective than traditional episodic gynecologic management.
When a multidisciplinary pain clinic is unavailable, the gynecologist should involve other specialists by referral while coordinating the overall care of the patient.
What to do, what not to do when the cause is organic
Patients in whom an organic pathology is identified should be treated accordingly. Recent publications detail management strategies for endometriosis associated with CPP.1,5 For example, the algorithm in the Figure is based on a 2002 consensus statement.1
Procedures to avoid surgery in the absence of pathology. Without proof of organic pathology or a reasonable functional explanation for the pelvic pain, a thorough psychosomatic evaluation should be completed before a surgical corrective procedure is considered.
These procedures have not proven effective for noncyclic CPP without pathology:
- unilateral adnexectomy for unilateral pain
- total abdominal hysterectomy
- presacral neurectomy
- uterine suspension for generalized pelvic pain
- lysis of adhesions, which is usually nonproductive unless the site of laparoscopically visualized adhesions specifically coincides with the site of the local pain. Pelvic adhesions often recur following surgical lysis.
Another option: Acupuncture and nerve blocks. These strategies can provide prolonged pain relief in properly selected patients. Consultation with an anesthesiologist who is trained in chronic pain management can identify appropriate candidates. Acupuncture likely works by increasing spinal cord endorphins. Patients with trigger points usually respond well to injection of local anesthetics.
FIGURE Assessment, diagnosis, and treatment options for chronic pelvic pain and presumed endometriosis
GnRH=gonadotropin-releasing hormone; NSAID=nonsteroidal antiinflammatory drug; OC=oral contraceptive Adapted from Fertil Steril; vol 78; Gambone JC, Mittman BS, Munro MG, Scialli AR, Winkel CA; Consensus statement for the management of chronic pelvic pain and endometriosis: proceedings of an expert-panel consensus process; 961-972; copyright 2002; with permission from American Society for Reproductive Medicine.
To prevent pain behaviors, schedule follow-up visits
In managing all patients with noncyclic CPP, take a supportive, sympathetic, and structured approach. This disorder is optimally treated in the context of an ongoing disease-management protocol rather than episodic therapy initiated because of a pain crisis.
Provide the patient with regular follow-up appointments; do not instruct her to call only when pain persists or recurs. The latter practice may create pain behaviors directed at obtaining sympathy and dramatic medical attention in certain patients.
Dr. Gambone reports no financial relationship with any companies whose products are mentioned in this article.
- If thorough investigation yields no diagnosis or indications for immediate surgery, empiric medical therapy for endometriosis is appropriate without laparoscopic confirmation. If empiric medical therapy fails, proceed to diagnostic laparoscopy.
- Referral to a multidisciplinary pain clinic has been shown to be more effective than episodic gynecologic management of patients, especially those with significant psychological issues.
- Schedule regular follow-ups. Do not instruct patients to call only during a pain crisis—this practice may create pain behaviors directed at obtaining sympathy and dramatic medical attention.
One woman complains of daily pain that worsens premenstrually. Another reports frequent aches radiating through her lower back and abdomen. A third says intercourse exacerbates her pain. And the list goes on.
Chronic pelvic pain (CPP) can manifest in dozens of ways, and the words patients use to describe it vary just as widely. Its multifaceted nature poses one of the biggest challenges to accurate diagnosis and appropriate management. Success is most likely when a compassionate attitude is combined with a systematic assessment to identify and understand the underlying cause—a blend of artful practice and scientific reasoning.
This article draws from consensus guidelines, other data, and personal experience to describe the components of careful diagnosis of this common but elusive condition.
10% of gynecology visits
CPP, which refers to pelvic pain of more than 6 months’ duration, accounts for roughly 10% of a gynecologist’s outpatient encounters, as well as many invasive procedures. For example, 1 in 5 gynecologic laparoscopies and 15% to 20% of hysterectomies are performed solely or in part for CPP.1,2
Noncyclic pelvic pain is not necessarily gynecologic
CPP is not always of gynecologic origin, even though the patient may perceive the pain as emanating from the reproductive organs. Because of this, careful evaluation is needed to distinguish gynecologic pain from CPP caused by orthopedic, gastrointestinal (GI), urologic, or neurologic conditions, or CPP that has a psychosomatic basis.
Purely cyclic uterine pain, which is referred to as dysmenorrhea, is usually characterized as primary or secondary. Although dysmenorrhea is frequently chronic and is an important clinical problem, this review focuses on noncyclic pelvic pain.
The relationship between CPP and underlying gynecologic pathology is often enigmatic. Many clinicians attribute the pain to psychological causes before completing a thorough assessment. An example of the hazards inherent in such an attitude is primary dysmenorrhea: Before the primary role of prostaglandins was elucidated, this form of dysmenorrhea was thought to be largely psychosomatic. As a result, it went untreated.
The TABLE lists major gynecologic and nongynecologic causes of noncyclic CPP. Endometriosis is the most common gynecologic condition associated with CPP.
TABLE
Causes of noncyclic chronic pelvic pain
| GYNECOLOGIC CAUSES |
| Endometriosis |
| Chronic pelvic inflammatory disease |
| Ovarian |
| Rapid ovarian capsule distension |
| Ovarian remnant syndrome |
| Recurrent hemorrhagic ovarian cysts |
| Uterine (uncommon) |
| Adenomyosis |
| Uterine myomas (under certain circumstances) |
| Uterine retroversion (if associated with other intraperitoneal pathology or dyspareunia) |
| Pelvic congestion syndrome |
| NONGYNECOLOGIC CAUSES |
| Genitourinary |
| Interstitial cystitis |
| Trigonitis |
| Urinary retention |
| Urethral syndrome |
| Gastrointestinal |
| Crohn disease |
| Diverticulitis |
| Hernia formation |
| Irritable bowel syndrome |
| Inflammatory bowel disease |
| Neoplasm |
| Partial bowel obstruction |
| Ulcerative colitis |
| Musculoskeletal (eg, low back pain) |
| Psychological factors |
Questions to elicit useful clues
The patient’s description of the location and pattern of pain, as well as history, are invaluable clues.
10 key questions. To elicit the most useful information from the patient, it may be necessary to ask numerous specifically phrased questions, such as:
- What is the the exact location of yourpain?
- What is the quality of the pain (sharp? dull?)
- Does it radiate, or spread, to other areas of the body? If so, where?
- How many times have you experienced this pain?
- How long does each episode last?
- How intense is each episode?
- What makes the pain worse? What makes it feel better?
- Does the pain change with your menstrual cycle (or with bowel movements, urination, sexual intercourse, or physical activity)?
- How much, and how, does the pain interfere with your daily activities?
- How would you rate the severity of the pain on a scale of 0 to 10, where 0 = no pain and 10 = the worst pain imaginable?
A complete medication history, especially of pain medication, is important.
Medical history should include information about nongynecologic conditions that may account for CPP, such as irritable bowel syndrome, ulcerative colitis, Crohn disease, and interstitial cystitis.
Pending compensation or litigation issues should be recognized and addressed.
Patient’s depiction of pain may lead to source
Understanding the anatomy of the nerves that transmit pain can facilitate interpretation of patient reports. It may explain, for example, why GI pathology can cause the perception of gynecologic pain. See “The anatomy of pelvic pain perception”.
- Pain relieved by defecation as well as irregular defecation patterns lasting at least 3 months suggest irritable bowel syndrome.
- Urinary urgency, frequency, nocturia, and pelvic pain may point to early interstitial cystitis.
- In multiparous women, pelvic pain that worsens premenstrually and with fatigue, standing, and sexual intercourse indicates possible pelvic congestion syndrome. Venographic studies may document pelvic vein varicosities in such cases.
Convergence of sensation
Somatic pain. Painful impulses originating in the skin, muscles, bones, joints, and parietal peritoneum travel in somatic nerve fibers.
Visceral pain. Painful impulses originating in the internal organs travel in visceral nerves.
Visceral pain is more diffusely localized than somatic pain and has a less well-defined area of projection to the sensory cortex of the brain for its identification. Also, there are many more somatic neurons than viscerosomatic ones in the dorsal horn of the spinal cord.
These differences result in a convergence of sensation that may be difficult for the patient and physician to interpret. Visceral pain is usually interpreted as coming from the skin and other surface tissues, which are supplied by the corresponding spinal cord segment, resulting in referred pain. For example, the initial pain of appendicitis is perceived to be in the epigastric area, because both are innervated by the thoracic cord segments T8, T9, and T10.
Since innervation of the lower intestinal tract is the same as that of the uterus and fallopian tubes, patients may experience pain caused by GI pathology as gynecologic pain.
Pain sensitivity of genital tissues
- External genitalia are exquisitely sensitive.
- Vaginal pain sensation is variable.
- The cervix is relatively insensitive to small biopsies but is sensitive to deep incision or to dilatation.
- The uterus is very sensitive.
- The ovaries are insensitive to many stimuli, but they are sensitive to rapid distension of the ovarian capsule and compression during physical examination.
Reproduce the pain on physical examination
Examination should be conducted in a way that does not provoke involuntary guarding, which may obscure findings. Gaining trust beforehand is important, as is taking time to explain what will be done during the exam.
Ask the patient to show you where the pain occurs, if possible, and try to reproduce the pain by palpation. Look for abdominal distension, and locate any tender areas. Continue the pelvic examination in this way, always attempting to reproduce and localize the pain. Look for any tenderness, nodularity (especially in the cul-de-sac), and palpable masses in the cul-de-sac or adnexal regions.
Examine the abdominal wall for evidence of myofascial trigger points and for iliohypogastric (T12, L1), ilioinguinal (T12, L1), or genitofemoral (L1, L2) nerve entrapment. Then ask the patient to tense the abdominal muscles by performing a straight-leg–raising maneuver or a partial sit-up. Points that are still tender, more tender, or that reproduce the patient’s pain after these maneuvers should be injected with 2 to 3 mL of 2.5% bupivacaine. Chronic abdominal wall pain is confirmed if the pain level is reduced by at least 50%.
Chronic low back pain without abdominal pain is seldom of gynecologic origin. If there is evidence of musculoskeletal disease, consider consulting an orthopedist. Neuromuscular symptoms may be accompanied by a pelvic mass. Positive neurologic signs such as muscle weakness or abnormal reflexes should prompt consultation. In such cases, surgical exploration may reveal a neuroma or bony tumor.
Lab, imaging studies: How useful?
Traditional laboratory studies are of limited value, although a complete blood cell count, erythrocyte sedimentation rate (ESR), and urinalysis should be obtained. The ESR is nonspecific and will be elevated in any type of inflammatory condition, such as subacute salpingo-oophoritis, tuberculosis, or inflammatory bowel disease.
A thorough genitourinary evaluation is important, including cystoscopy when suspicion of urinary tract disease is high. If no obvious cause for the pain is uncovered, pelvic ultrasonography may be helpful. This is especially true in an obese or uncooperative patient, when bimanual pelvic examination is difficult.
If bowel or urinary signs and symptoms are present, then an endoscopy, abdominal and pelvic computed tomography, cystoscopy, or computed tomography urogram may be useful.
No diagnosis, no reason for surgery? Start therapy for endometriosis
If the workup yields no diagnosis and rules out indications for surgery, such as significant adnexal mass, then empiric medical therapy for endometriosis is appropriate without laparoscopic confirmation.1
Begin a trial of empiric therapy with a nonsteroidal anti-inflammatory drug such as ibuprofen or naproxen, oral contraceptives, or both.1 Women with ovarian pathology (eg, periovarian adhesions, recurrent functional cyst formation) and those whose pain worsens midcycle, premenstrually, or menstrually may experience improvement with a trial of oral contraceptives to suppress ovulation and menstruation.
Use second-line empiric therapy before diagnostic laparoscopy
If these treatments fail, begin empiric therapy with second-line agents such as danazol, progestins, or gonadotropin-releasing hormone (GnRH) agonists. Laparoscopic confirmation is unnecessary.
Several publications, including a 2002 consensus statement on management of chronic pelvic pain and endometriosis, support this approach.1,3-5 In fact, following recent guidelines for evaluating and treating CPP is likely to lead to fewer invasive procedures for diagnosis and treatment, including laparoscopy and hysterectomy.
Surgical ablative therapy has not been shown superior to medical therapy in addressing the pain of early-stage endometriosis. Medical therapy is less invasive and, as the following report illustrates, it probably treats all disease present.
In a study of 95 women with CPP in whom endometriosis was suspected after a thorough noninvasive workup yielded no diagnosis, investigators found that empiric therapy with depot leuprolide for 12 weeks provided significant pain relief compared with both the baseline and a placebo group (P ≤ .001 for both comparisons). Posttreatment diagnostic laparoscopy confirmed endometriosis in more than three quarters of both treatment groups.3
The limits of laparoscopy
Although second-line empiric therapy is controversial, laparoscopy is invasive and does not necessarily yield a definitive diagnosis. However, diagnostic laparoscopy may ultimately be indicated in patients with pain of undetermined cause. Perform laparoscopy only if no cause of pain can be identified and confirmed, and after reasonable trials of empiric medical therapy.
Up to one third of women have no pathology visualized on laparoscopy.6 This does not mean that no pathology is present. Endometriosis is missed visually during laparoscopy in up to one third of CPP patients in whom the disease is proven on biopsy.3,4
Laparoscopically identified pathology does not necessarily make the diagnosis. For example, the size and location of endometriotic implants do not always correlate with the presence of pain. Thus, endometriosis may be present but may not be the cause of CPP.1 Similarly, visualizing pelvic adhesions during laparoscopy does not necessarily mean that the adhesions are responsible for the pelvic pain, as women with extensive adhesions are sometimes asymptomatic.7
Gynecologic conditions identifiable by diagnostic laparoscopy
The following gynecologic conditions may be identified through diagnostic laparoscopy in women with cyclic and noncyclic pain:
Endometriosis is seen in at least one third to one half of women with CPP7; most sites of endometriosis are in the pelvic area. The mechanisms by which endometriosis causes pain are not fully understood, however.
Chronic pelvic inflammatory disease (PID). Laparoscopy identifies adhesions or stigmata of chronic PID in about 25% of women with CPP. Chronic PID may cause noncyclic CPP because recurrent exacerbations frequently result in hydrosalpinges and adhesions between the tubes, ovaries, and intestinal structures. Before ascribing symptoms to adhesions, however, clinicians should specifically note this pathology in the area of pain.
Ovarian causes. Ovarian cysts are frequently asymptomatic, but pain may result from rapid distention of the ovarian capsule. An ovary or ovarian remnant may enter the retroperitoneal space secondary to inflammation or previous surgery. This is known as ovarian remnant syndrome, and cyst formation in these circumstances is usually painful. Some women, for unknown reasons, may develop multiple recurrent hemorrhagic ovarian cysts that are associated with pelvic pain and dyspareunia.
Uterine origins. Adenomyosis (endometrial glands and stroma invading the myometrium) may cause dysmenorrhea and menorrhagia, but rarely causes noncyclic CPP. Uterine myomas usually do not cause pelvic pain unless they are undergoing degeneration, torsion, or are compressing pelvic nerves. (Occasionally, a submucous leiomyoma may protrude through the cervix, which has been described by women as pain not unlike childbirth.) Uterine myomas may also cause pain from rapid growth or degeneration during pregnancy.
Chronic pain itself—with or without organic pathology—produces debilitating psychological responses.
Pelvic pain is not likely to be due solely to variations in uterine position, but dyspareunia with deep penetration may occasionally be associated with uterine retroversion. A tender uterus that is in a fixed retroverted position usually signifies other intraperitoneal pathology, such as endometriosis or PID.
Pelvic congestion syndrome. As noted, this has been described in multiparous women who have pelvic vein varicosities (documented on venographic studies) resulting in “congested” pelvic organs. Many women with this condition are noted to have a uterus that is mobile, retroverted, soft, boggy, and slightly enlarged. Ovarian hormone suppression and cognitive behavioral therapy may relieve pain. If these measures fail, and if venographic studies confirm the diagnosis, then hysterectomy and oophorectomy may be beneficial. Surgery should be considered only if the diagnosis is confirmed and medical therapy fails.
If no pathology is found, is the pain psychological?
Inability to identify a pathologic diagnosis in approximately one third of patients with CPP has led to the postulation of a psychological etiology. It is appropriate to consider psychological factors early in your evaluation. However, as mentioned earlier, a suggestion of psychological etiology should not delay or preclude further investigation of somatic pathology.
Ask patients about past or present physical, sexual, or emotional abuse. History of major psychosexual trauma (molestation, incest, rape) was identified in 48% of 106 women evaluated at my institution with no pathologic diagnosis following a thorough workup and laparoscopy.2 Women with such a history should be referred for psychological evaluation.
Secondary gain often complicates diagnosis and therapy. Ask about current life events as well as any noteworthy happenings when the pain began. Consider the possibility of secondary gain or other psychological benefits of pain behaviors. For example, exaggerated response to pain and the reaction of significant others may be so reinforcing that pain behavior persists even after the painful stimulus has resolved. If you suspect this may be an issue, the patient should undergo psychological evaluation.
Pain may be psychologically debilitating. Evidence suggests that chronic pain itself, with or without organic pathology, produces debilitating psychological responses. Patients with such pain tend to feel depressed, helpless, and passive. They withdraw from social and sexual activity, and are preoccupied with pain and suffering.7 Personality testing of CPP patients without identified organic pathology shows characteristics similar to those of chronic pain patients with organic pathology. Specifically, on the Minnesota Multiphasic Personality Inventory, both these groups demonstrate a greater degree of anxiety, hypochondriasis, and hysteria than do control subjects.
Psychological response can affect organic processing of pain. A person’s psychological response to pain can influence how the nervous system transmits pain. Many neurotransmitters and neuromodulators can amplify or diminish pain impulses before pain signals reach the central nervous system. Anxiety and other psychological states may affect release and metabolism of these neurotransmitters, thus shaping perception of pain. Many forms of CPP, especially those with little or no pathology identified, may result primarily from disordered pain perception due to modulation of afferent impulses in the nervous system.
Excitatory modulators include substance P, glutamate, aspartate, calcitonin generelated peptide, and vasoactive intestinal peptide. Inhibitory neuromediators include endogenous opioid peptides, norepinephrine, serotonin (5-HT), and gamma-aminobutyric acid (GABA). Tricyclic antidepressants, other GABA-ergic agents, and selective serotonin reuptake inhibitors can increase inhibitory neuromediators, making them very useful in the management of CPP.
Referral to a multidisciplinary pain clinic is the most productive strategy for patients with noncyclic CPP and significant psychological issues.7 All patients with mental illness should be referred for psychological treatment.
A multidisciplinary facility typically includes a psychologist who also has expertise in sexual and marital counseling, an anesthesiologist, and—ideally—an acupuncturist, in addition to a gynecologist. Relaxation training, along with cognitive and behavioral therapies, can help train the patient to replace abnormal pain behavior and its secondary gain with effective, adaptive behavioral responses. Research has shown multidisciplinary management to be more effective than traditional episodic gynecologic management.
When a multidisciplinary pain clinic is unavailable, the gynecologist should involve other specialists by referral while coordinating the overall care of the patient.
What to do, what not to do when the cause is organic
Patients in whom an organic pathology is identified should be treated accordingly. Recent publications detail management strategies for endometriosis associated with CPP.1,5 For example, the algorithm in the Figure is based on a 2002 consensus statement.1
Procedures to avoid surgery in the absence of pathology. Without proof of organic pathology or a reasonable functional explanation for the pelvic pain, a thorough psychosomatic evaluation should be completed before a surgical corrective procedure is considered.
These procedures have not proven effective for noncyclic CPP without pathology:
- unilateral adnexectomy for unilateral pain
- total abdominal hysterectomy
- presacral neurectomy
- uterine suspension for generalized pelvic pain
- lysis of adhesions, which is usually nonproductive unless the site of laparoscopically visualized adhesions specifically coincides with the site of the local pain. Pelvic adhesions often recur following surgical lysis.
Another option: Acupuncture and nerve blocks. These strategies can provide prolonged pain relief in properly selected patients. Consultation with an anesthesiologist who is trained in chronic pain management can identify appropriate candidates. Acupuncture likely works by increasing spinal cord endorphins. Patients with trigger points usually respond well to injection of local anesthetics.
FIGURE Assessment, diagnosis, and treatment options for chronic pelvic pain and presumed endometriosis
GnRH=gonadotropin-releasing hormone; NSAID=nonsteroidal antiinflammatory drug; OC=oral contraceptive Adapted from Fertil Steril; vol 78; Gambone JC, Mittman BS, Munro MG, Scialli AR, Winkel CA; Consensus statement for the management of chronic pelvic pain and endometriosis: proceedings of an expert-panel consensus process; 961-972; copyright 2002; with permission from American Society for Reproductive Medicine.
To prevent pain behaviors, schedule follow-up visits
In managing all patients with noncyclic CPP, take a supportive, sympathetic, and structured approach. This disorder is optimally treated in the context of an ongoing disease-management protocol rather than episodic therapy initiated because of a pain crisis.
Provide the patient with regular follow-up appointments; do not instruct her to call only when pain persists or recurs. The latter practice may create pain behaviors directed at obtaining sympathy and dramatic medical attention in certain patients.
Dr. Gambone reports no financial relationship with any companies whose products are mentioned in this article.
1. Gambone JC, Mittman BS, Munro MG, Scialli AR, Winkel CA. Consensus statement for the management of chronic pelvic pain and endometriosis: proceedings of an expert-panel consensus process. Fertil Steril. 2002;78:961-972.
2. Reiter RC, Gambone JC. Demographic and historic variables in women with idiopathic chronic pelvic pain. Obstet Gynecol. 1990;75:428-432.
3. Ling F. Pelvic Pain Study Group. Randomized controlled trial of depot leuprolide in patients with chronic pelvic pain and clinically suspected endometriosis. Obstet Gynecol. 1999;93:51-58.
4. Winkel CA. Looking for a better way to manage endometriosis-related pain. Contemp Ob/Gyn. 2003;48:44-56.
5. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin #11: Medical Management of Endometriosis. Washington, DC: ACOG; December 1999.
6. Rapkin AJ. Chronic pelvic pain. In: Hacker N, Moore JG, eds. Essentials of Obstetrics and Gynecology. Philadelphia, Pa: WB Saunders; 1996;499-506.
7. Reiter RC, Gambone JC. Nongynecologic somatic pathology in women with chronic pelvic pain and negative laparoscopy. J Reprod Med. 1991;36:253-259.
1. Gambone JC, Mittman BS, Munro MG, Scialli AR, Winkel CA. Consensus statement for the management of chronic pelvic pain and endometriosis: proceedings of an expert-panel consensus process. Fertil Steril. 2002;78:961-972.
2. Reiter RC, Gambone JC. Demographic and historic variables in women with idiopathic chronic pelvic pain. Obstet Gynecol. 1990;75:428-432.
3. Ling F. Pelvic Pain Study Group. Randomized controlled trial of depot leuprolide in patients with chronic pelvic pain and clinically suspected endometriosis. Obstet Gynecol. 1999;93:51-58.
4. Winkel CA. Looking for a better way to manage endometriosis-related pain. Contemp Ob/Gyn. 2003;48:44-56.
5. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin #11: Medical Management of Endometriosis. Washington, DC: ACOG; December 1999.
6. Rapkin AJ. Chronic pelvic pain. In: Hacker N, Moore JG, eds. Essentials of Obstetrics and Gynecology. Philadelphia, Pa: WB Saunders; 1996;499-506.
7. Reiter RC, Gambone JC. Nongynecologic somatic pathology in women with chronic pelvic pain and negative laparoscopy. J Reprod Med. 1991;36:253-259.