User login
Seventy-Five Percent of Total Energy Intake Comes From Ultra-Processed Foods Among a Sample of Veterans With Overweight and Obesity: An Exploratory Analysis of Three-Day Food Records
Seventy-Five Percent of Total Energy Intake Comes From Ultra-Processed Foods Among a Sample of Veterans With Overweight and Obesity: An Exploratory Analysis of Three-Day Food Records
Roughly 8.6% of the 17.4 million US veterans live in poverty. About 11.1% are considered food insecure (ie, unable to acquire adequate food for ≥1 household members), with another 5.3% considered very food insecure (ie, eating patterns of ≥1 household members were disrupted and their food intake was reduced at least some time during the year). Compared with nonveterans, veterans are 7.4% more likely to be food insecure.1 This high prevalence of food insecurity and poverty has a negative impact on veteran diets.
Veterans’ diets contained more added sugars and solid fats and scored lower compared with nonveterans when assessed for diet quality with the Healthy Eating Index.2 Veterans have a higher prevalence of diet-related chronic disease, including diabetes, hypertension, and obesity compared with the nonveterans.3-5 Given the critical role of diet in health and disease risk, enhancing diet quality among veterans has garnered significant attention and calls to action.2,6,7 While there are many factors that contribute to diet, any veteran can receive a consultation or self-refer to receive nutrition counseling effective for improving diet quality, within the US Department of Veterans Affairs (VA).
The NOVA food classification system describes diet quality by categorizing food items by processing methods and ingredients into 4 food groups.8 The first is unprocessed and minimally processed items (MPFs) such as fresh fruits, vegetables, and meats. MPFs consist of whole foods which can also be minimally processed (eg, chopping, drying, grinding, heating, chilling). Culinary processed foods (CPFs) are processed foods for cooking (eg, salt, butter, and vinegar) and are typically eaten in small quantities along with MPFs. Processed foods (PRFs) include canned and smoked foods, while ultra-processed foods (UPFs) are distinguished by industrial ingredients, requiring specialized tools and processing techniques, and hyper-palatability related to color, flavor, and packaging.8 Examples of UPFs include mass-produced breads found at grocery stores, prepackaged snacks and meals, and hydrogenated oils. UPF consumption is associated with higher risk for negative cardiometabolic outcomes, common mental disorders, and all-cause mortality.9 To date, only a study by Powell et al has used the NOVA classification system in a veteran population, and it was limited to a comparison of the price of UPFs and veteran body mass index (BMI).10 Therefore, it remains unknown what percentage of total energy intake (TEI) comes from UPFs in the diets of veterans.
This study sought to quantify the proportion of TEI from UPFs among a sample of patients from the VA Phoenix Health Care System (VAPHCS). Results from a 2021 global meta-analysis reveal that the US and United Kingdom have the highest intakes of UPFs in the world.11 Specifically, within the US, 15 studies with 234,890 participants reveal that the majority of TEI (about 55%) comes from UPFs.11 We hypothesized that this veteran sample would have a higher proportion of TEI from UPFs, possibly due to a higher prevalence of poverty and food insecurity among veterans compared with nonveterans.1 If the percentage of TEI coming from UPF is higher or even similar to nonveterans, further efforts to increase veterans’ use of the available nutritional services would be warranted to minimize nutrition-related disease among veterans.
Methods
This is a cross-sectional, secondary data analysis of baseline 3-day food records collected from 2017 to 2020 from 92 patients recruited at VAPHCS to participate in a whole-food plant-based diet study.12 The original study was reviewed and approved by the VAPHCS Institutional Review Board (1593830). Recruitment methods included clinician recommendation, a recorded advertisement played while phone calls were on hold, and flyers distributed throughout VAPHCS. Patients were included if they were aged 18 to 90 years, had a BMI 25.1 to 39.9, had a diagnosis of nutrition-related chronic disease (hypertension, diabetes, or hyperlipidemia), an interest and desire to make a lifestyle change, active telephone contact information (either landline or cell phone), no contraindication to be on a whole-food plant-based diet, access to transportation and a functioning kitchen, ability to prepare meals independently, access to a computer or tablet with internet access, and a digital camera or smartphone. Exclusion criteria included significant unplanned weight loss within 6 months, uncontrolled insulin-dependent diabetes with a current hemoglobin A1c > 9%, pregnancy/lactation, taking prescribed weight loss medication, currently following a diet (eg, plant-based diet, vegan, or medical weight loss program diet), celiac disease diagnosed within 6 months, end-stage hepatic disease or renal disease requiring dialysis, active cancer or receiving chemotherapy or radiation therapy, active alcohol or substance use disorder, history of eating disorders, fasting triglyceride level > 350 mg/dL, any psychological issues that prevent adherence, inability to speak English, limited mobility, and homeless or in housing with limited kitchen access. A baseline 3-day food record was collected from the participants and used in this secondary analysis.
Diet Analysis
Food records were analyzed using Esha Research Food Processor 4.0 to identify calorie and macronutrient information. To limit bias, food items were coded independently by 2 researchers into 4 food processing groups determined by the NOVA classification: MPF, CPF, PRF, and UPF.8 When possible, specific ingredient information was collected using internet searches for brand product websites. Initial coding had an 89% agreement rate for food item coding between the 2 researchers. As coding was done in duplicate, a third researcher resolved disagreements. The number of food items for each processing group was determined and the mean (SD) percentage of TEI for each NOVA group was provided across participants. A 1-way analysis of variance and Tukey Multiple Comparisons Test were used to determine significance between groups with an α = .05 using Prism V9.
Results
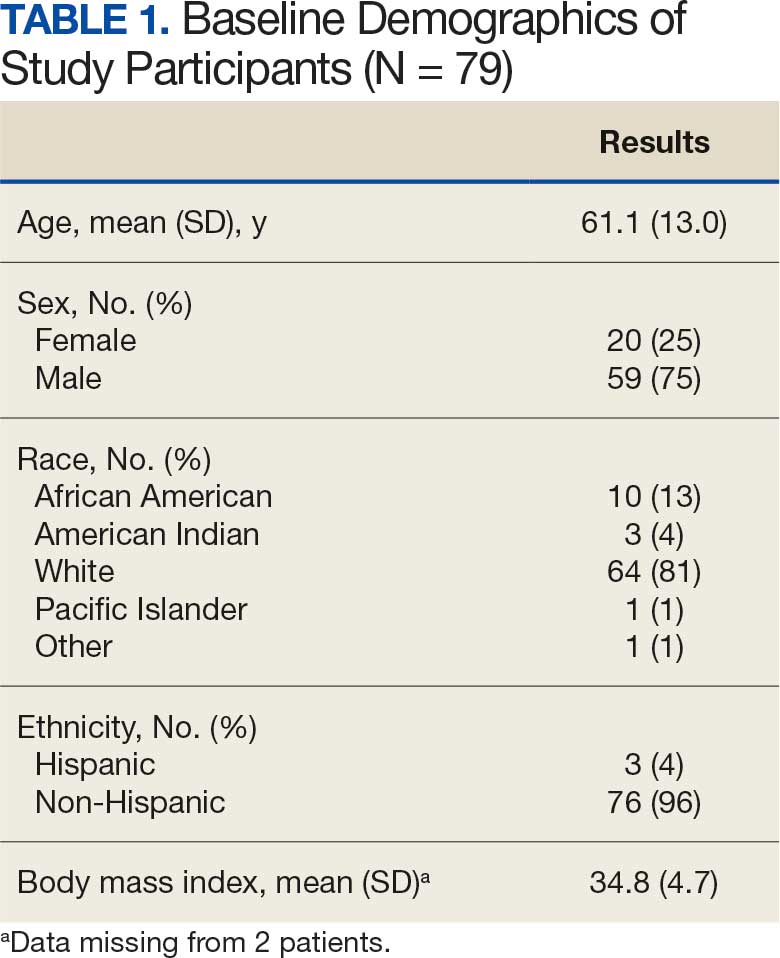
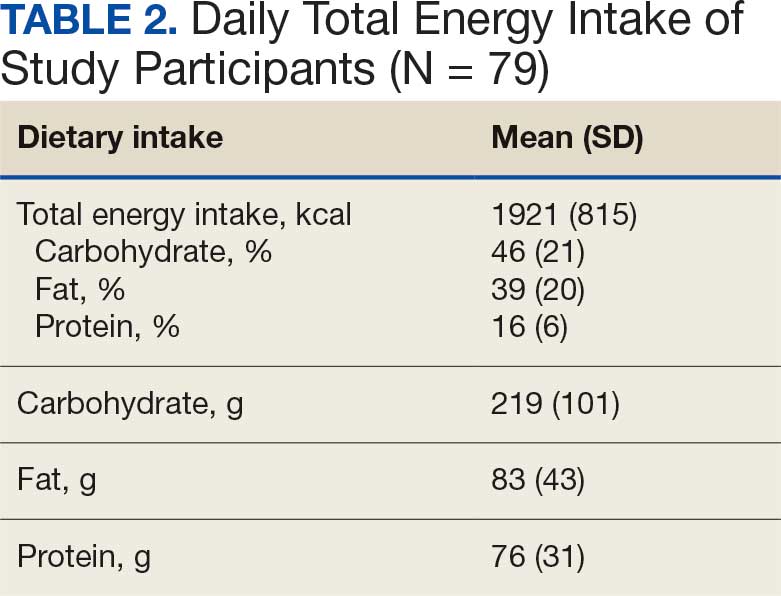
Of the 92 participants in the original study, only 79 met inclusion criteria and had baseline diet data. The 79 veterans had a mean (SD) age of 61 (13) years and 59 (75%) were male (Table 1). Mean (SD) TEI was 1921 (815) kcal. The mean (SD) percentage of calories from carbohydrate, fat, and protein were 46% (21%), 39% (20%), and 16% (6%), respectively (Table 2).
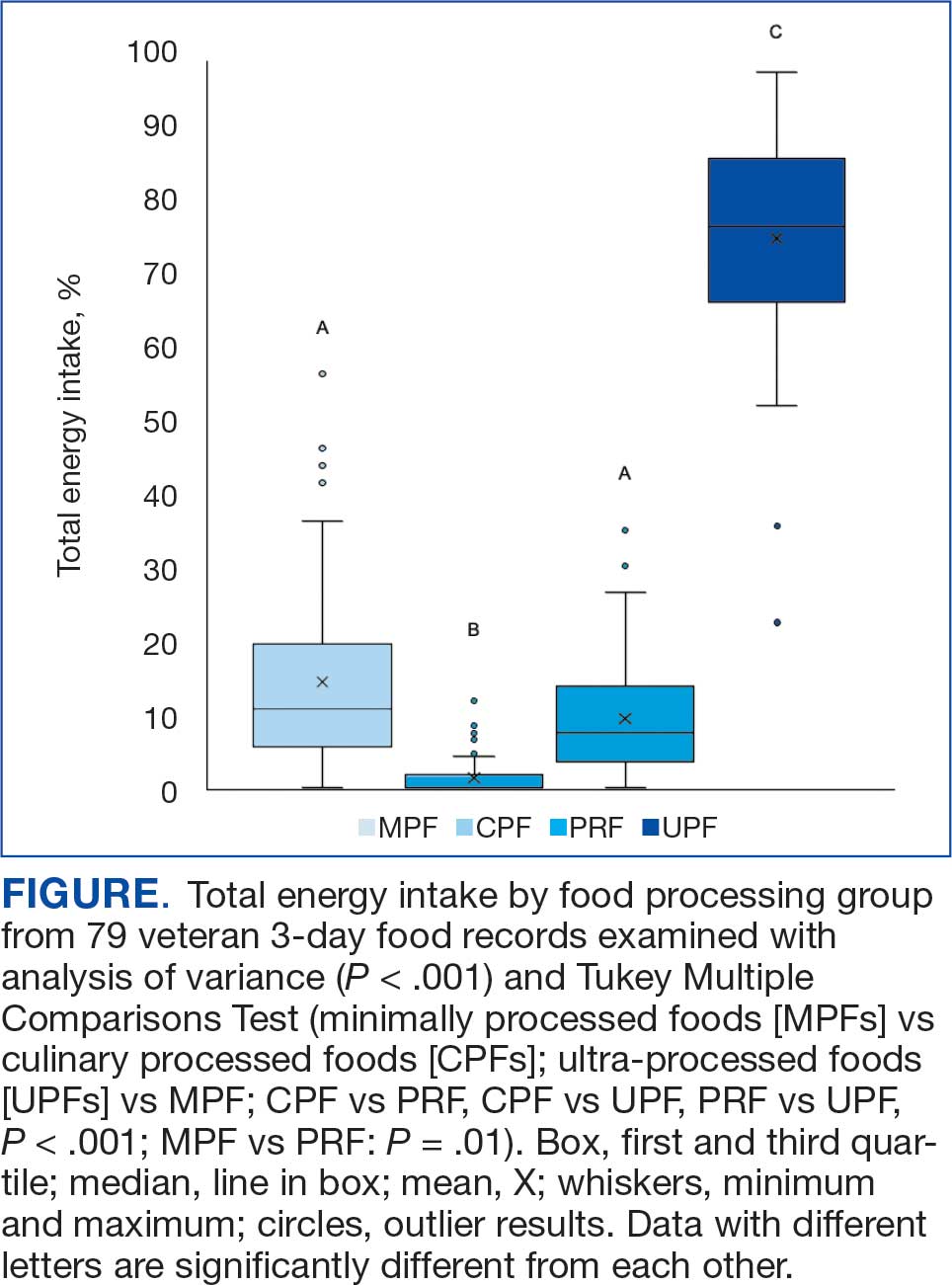
A mean (SD) of 36 (12) food items were analyzed from the 3-day food records. The majority of food items were UPFs (56%), 33% were MPFs, 8% were PRFs, and 3% were CPFs. In total, 75% of TEI came from UPFs (P < .001); only 14% of TEI came from minimally processed foods (Figure).
Discussion
To our knowledge, this is the first analysis of UPF consumption among US veterans. TEIs coming from UPFs appear to be about 20% higher among veterans compared to nonveterans: 75% vs 55%.11 Coupled with high UPF consumption, MPFs (14%) and PRFs (9%) represent smaller sources of TEI among surveyed veterans. Top caloric sources of UPFs in the US include sandwiches (including burgers), sweet bakery products, savory snacks, pizza, sweetened beverages, and breads, rolls, and tortillas, and likely reflect the major sources of UPFs in the veteran diet.13 As the statistical comparison between the veteran data and nonveteran data is not feasible in the present study, a future study with a much larger sample size would be needed for a direct comparison.
While the exact cause of higher UPF consumption among sampled veterans remains unknown and likely multifactorial (eg, cost, food insecurity, access, cooking skills, nutrition knowledge), veterans can receive a consult or self-refer to a registered dietitian nutritionist (RDN) for nutrition education. Counseling has been shown to be an effective way to improve diet quality and increase daily fruit and vegetable intake.14 High consumption of UPFs, which are generally energy-dense and nutrient-poor, contributes to the low diet quality observed in veterans, and future research examining the relationship between UPF intake and overall diet quality among veterans is warranted.2,15 As nutrition knowledge is associated with higher diet quality among veterans, increased use of nutrition services (ie, nutrition education or food supplement programs) has the potential to influence consumption of MPFs and decrease consumption of UPFs.16 Subsequently, UPF-targeted interventions developed by VA RDNs hold the promise to decrease consumption of UPFs and increase intake of MPFs and PRFs.
Veterans have a high prevalence of diabetes, hypertension, and obesity.9 The high UPF intake observed in this sample of veterans may increase the risk for these chronic diseases and overall mortality. The high percentage of TEI from UPFs among veterans is also of concern not only due to potential negative health outcomes, but also associated costs of treating veterans with multimorbidities.17 Targeting UPF intake via nutritional education may promote health and decrease the financial burden needed to support the health of veterans.
Improving veteran health and well-being, including enhancing health care accessibility in underserved areas, are pivotal objectives of the VA strategic plan for 2026 to 2030. Public policy aims to tackle food insecurity within the veteran population during the first 5 years of civilian life.18 In alignment with the White House Strategy on Hunger, Nutrition, and Health, VA established a Food Security Office (FSO) in 2023. The FSO mission is to use an interdisciplinary approach to provide resources to ensure veteran food security and create an environment where all veterans are food and nutrition secure.
Limitations
This study has several limitations. As the Food Processor software database does not include all brand items, similar brands were used to mirror the nutrient profile. While food records are common among veteran diet studies, accuracy may be reduced due to self-reporting bias.19 Different interpretation of the NOVA classification designation for various food items is possible, however, 89% of foods were coded the same by the research team which suggests high accuracy in food coding. Specific ingredient information was not collected from the 3-day food records; thus, these records were not produced in such a way to improve the accuracy of the NOVA classification designation. This study was limited by its small sample size (N = 79); although, this analysis is larger than other studies of UPF consumption in the US.20,21 In addition, the generalizability of this study is limited as this population sample was from a single VA hospital and may not reflect the overall veteran population. Participants in this study were recruited only from those receiving VA care, thus their diet quality may not represent the quality consumed by veterans not participating in VA services. Further research on UPF consumption among veterans is warranted with a larger, more representative study sample size.
Conclusions
As this is the highest observed UPF intake documented in the US, these results should be of concern for the VA and its RDNs. More research is needed to better understand why UPF consumption is so high among veterans, what barriers veterans face to decreasing UPF consumption, and what intervention(s) veterans would welcome to improve their diet quality. Presently, veterans are provided with access to a variety of effective nutrition education and counseling options and should be encouraged to use these services. VA RDNs should be aware of the high intake of UPFs in the veteran population and familiarize themselves with education and counseling strategies that promote behavior change to replace UPFs with more nutrient-dense foods choices.
- Rabbitt MP, Smith MD. Food insecurity among workingage veterans. US Dept of Agriculture, Economic Research Service; 2021. Accessed January 26, 2026. https://www.ers.usda.gov/publications/pub-details/?pubid=101268
- Dong D, Stewart H, Carlson AC. An examination of veterans’ diet quality. US Dept of Agriculture, Economic Research Service; 2019. Accessed January 26, 2026. https:// www.ers.usda.gov/publications/pub-details/?pubid=95608
- US Department of Veterans Affairs; US Department of Defense. VA/DoD clinical practice guideline for the management of adult overweight and obesity. 2020. Accessed January 26, 2026. https://www.healthquality.va.gov/guidelines/cd/obesity/
- US Department of Veterans Affairs; US Department of Defense. VA/DoD clinical practice guideline for the management of type 2 diabetes mellitus in primary care. 2023. Accessed January 26, 2026. https://www.healthquality.va.gov/guidelines/cd/diabetes/
- Boersma P, Cohen R, Zelaya C, et al. Multiple chronic conditions among veterans and nonveterans: United States, 2015–2018. Natl Health Stat Rep. 2021. doi:10.15620/cdc:101659
- Hoerster KD, Wilson S, Nelson KM, et al. Diet quality is associated with mental health, social support, and neighborhood factors among veterans. Eat Behav. 2016;23:168- 173. doi:10.1016/j.eatbeh.2016.10.003
- Becerra MB, Hassija CM, Becerra BJ. Food insecurity is associated with unhealthy dietary practices among US veterans in California. Public Health Nutr. 2017;20:2569-2576. doi:10.1017/S1368980016002147
- Monteiro CA, Cannon G, Levy RB, et al. Ultra-processed foods: what they are and how to identify them. Public Health Nutr. 2019;22:936-941. doi:10.1017/S1368980018003762
- Lane MM, Gamage E, Du S, et al. Ultra-processed food exposure and adverse health outcomes: umbrella review of epidemiological meta-analyses. BMJ. February 2024:e077310. doi:10.1136/bmj-2023-077310
- Powell LM, Jones K, Duran AC, et al. The price of ultra-processed foods and beverages and adult body weight: Evidence from U.S. veterans. Econ Hum Biol. 2019;34:39- 48. doi:10.1016/j.ehb.2019.05.006
- Marino M, Puppo F, Del Bo’ C, et al. A systematic review of worldwide consumption of ultra-processed foods: findings and criticisms. Nutrients. 2021;13. doi:10.3390/nu13082778
- Parrington D, Kurtz J, Fawcett J, et al. Pilot study on the effects of a whole-food, plant-strong diet on cardiovascular risk factors in veterans: part 3. Curr Dev Nutr. 2022;6:385. doi:10.1093/cdn/nzac054.040
- Williams AM, Couch CA, Emmerich SD, et al. Ultra-processed Food Consumption in Youth and Adults: United States, August 2021-August 2023. NCHS Data Brief. 2025. doi:10.15620/cdc/174612
- Serra MC, Addison O, Giffuni J, et al. Changes in self-reported fruit and vegetable intake following nutritional modification in high risk older veterans. J Nutr Gerontol Geriatr. 2021;40:1-8. doi:10.1080/21551197.2020.1863892
- Gupta S, Hawk T, Aggarwal A, et al. Characterizing ultra-processed foods by energy density, nutrient density, and cost. Front Nutr. 2019;6:1-9. doi:10.3389/fnut.2019.00070
- Robinson LA, Colin CR, Smith KS, et al. Diet quality is associated with nutrition knowledge and physical activity in the US military veterans enrolled in university programmes. BMJ Mil Heal. 2023:e002525. doi:10.1136/military-2023-002525
- Yoon J, Zulman D, Scott JY, et al. Costs associated with multimorbidity among VA patients. Med Care. 2014;52:S31-6. doi:10.1097/MLR.0000000000000061
- End Veteran Hunger Act of 2022, H.R. 8852, 117th Cong. (2022). Accessed January 26, 2026. https://www.congress.gov/bill/117th-congress/house-bill/8852.
- Collins RA, Baker B, Coyle DH, et al. Dietary assessment methods in military and veteran populations: a scoping review. Nutrients. 2020;12:1-21. doi:10.3390/nu12030769
- Smiljanec K, Mbakwe AU, Ramos-Gonzalez M, et al. Associations of ultra-processed and unprocessed/minimally processed food consumption with peripheral and central hemodynamics, and arterial stiffness in young healthy adults. Nutrients. 2020;12. doi:10.3390/nu12113229
- Rohatgi KW, Tinius RA, Cade WT, et al. Relationships between consumption of ultra-processed foods, gestational weight gain and neonatal outcomes in a sample of US pregnant women. PeerJ. 2017;5:e4091. doi:10.7717/peerj.4091
Roughly 8.6% of the 17.4 million US veterans live in poverty. About 11.1% are considered food insecure (ie, unable to acquire adequate food for ≥1 household members), with another 5.3% considered very food insecure (ie, eating patterns of ≥1 household members were disrupted and their food intake was reduced at least some time during the year). Compared with nonveterans, veterans are 7.4% more likely to be food insecure.1 This high prevalence of food insecurity and poverty has a negative impact on veteran diets.
Veterans’ diets contained more added sugars and solid fats and scored lower compared with nonveterans when assessed for diet quality with the Healthy Eating Index.2 Veterans have a higher prevalence of diet-related chronic disease, including diabetes, hypertension, and obesity compared with the nonveterans.3-5 Given the critical role of diet in health and disease risk, enhancing diet quality among veterans has garnered significant attention and calls to action.2,6,7 While there are many factors that contribute to diet, any veteran can receive a consultation or self-refer to receive nutrition counseling effective for improving diet quality, within the US Department of Veterans Affairs (VA).
The NOVA food classification system describes diet quality by categorizing food items by processing methods and ingredients into 4 food groups.8 The first is unprocessed and minimally processed items (MPFs) such as fresh fruits, vegetables, and meats. MPFs consist of whole foods which can also be minimally processed (eg, chopping, drying, grinding, heating, chilling). Culinary processed foods (CPFs) are processed foods for cooking (eg, salt, butter, and vinegar) and are typically eaten in small quantities along with MPFs. Processed foods (PRFs) include canned and smoked foods, while ultra-processed foods (UPFs) are distinguished by industrial ingredients, requiring specialized tools and processing techniques, and hyper-palatability related to color, flavor, and packaging.8 Examples of UPFs include mass-produced breads found at grocery stores, prepackaged snacks and meals, and hydrogenated oils. UPF consumption is associated with higher risk for negative cardiometabolic outcomes, common mental disorders, and all-cause mortality.9 To date, only a study by Powell et al has used the NOVA classification system in a veteran population, and it was limited to a comparison of the price of UPFs and veteran body mass index (BMI).10 Therefore, it remains unknown what percentage of total energy intake (TEI) comes from UPFs in the diets of veterans.
This study sought to quantify the proportion of TEI from UPFs among a sample of patients from the VA Phoenix Health Care System (VAPHCS). Results from a 2021 global meta-analysis reveal that the US and United Kingdom have the highest intakes of UPFs in the world.11 Specifically, within the US, 15 studies with 234,890 participants reveal that the majority of TEI (about 55%) comes from UPFs.11 We hypothesized that this veteran sample would have a higher proportion of TEI from UPFs, possibly due to a higher prevalence of poverty and food insecurity among veterans compared with nonveterans.1 If the percentage of TEI coming from UPF is higher or even similar to nonveterans, further efforts to increase veterans’ use of the available nutritional services would be warranted to minimize nutrition-related disease among veterans.
Methods
This is a cross-sectional, secondary data analysis of baseline 3-day food records collected from 2017 to 2020 from 92 patients recruited at VAPHCS to participate in a whole-food plant-based diet study.12 The original study was reviewed and approved by the VAPHCS Institutional Review Board (1593830). Recruitment methods included clinician recommendation, a recorded advertisement played while phone calls were on hold, and flyers distributed throughout VAPHCS. Patients were included if they were aged 18 to 90 years, had a BMI 25.1 to 39.9, had a diagnosis of nutrition-related chronic disease (hypertension, diabetes, or hyperlipidemia), an interest and desire to make a lifestyle change, active telephone contact information (either landline or cell phone), no contraindication to be on a whole-food plant-based diet, access to transportation and a functioning kitchen, ability to prepare meals independently, access to a computer or tablet with internet access, and a digital camera or smartphone. Exclusion criteria included significant unplanned weight loss within 6 months, uncontrolled insulin-dependent diabetes with a current hemoglobin A1c > 9%, pregnancy/lactation, taking prescribed weight loss medication, currently following a diet (eg, plant-based diet, vegan, or medical weight loss program diet), celiac disease diagnosed within 6 months, end-stage hepatic disease or renal disease requiring dialysis, active cancer or receiving chemotherapy or radiation therapy, active alcohol or substance use disorder, history of eating disorders, fasting triglyceride level > 350 mg/dL, any psychological issues that prevent adherence, inability to speak English, limited mobility, and homeless or in housing with limited kitchen access. A baseline 3-day food record was collected from the participants and used in this secondary analysis.
Diet Analysis
Food records were analyzed using Esha Research Food Processor 4.0 to identify calorie and macronutrient information. To limit bias, food items were coded independently by 2 researchers into 4 food processing groups determined by the NOVA classification: MPF, CPF, PRF, and UPF.8 When possible, specific ingredient information was collected using internet searches for brand product websites. Initial coding had an 89% agreement rate for food item coding between the 2 researchers. As coding was done in duplicate, a third researcher resolved disagreements. The number of food items for each processing group was determined and the mean (SD) percentage of TEI for each NOVA group was provided across participants. A 1-way analysis of variance and Tukey Multiple Comparisons Test were used to determine significance between groups with an α = .05 using Prism V9.
Results
Of the 92 participants in the original study, only 79 met inclusion criteria and had baseline diet data. The 79 veterans had a mean (SD) age of 61 (13) years and 59 (75%) were male (Table 1). Mean (SD) TEI was 1921 (815) kcal. The mean (SD) percentage of calories from carbohydrate, fat, and protein were 46% (21%), 39% (20%), and 16% (6%), respectively (Table 2).


A mean (SD) of 36 (12) food items were analyzed from the 3-day food records. The majority of food items were UPFs (56%), 33% were MPFs, 8% were PRFs, and 3% were CPFs. In total, 75% of TEI came from UPFs (P < .001); only 14% of TEI came from minimally processed foods (Figure).

Discussion
To our knowledge, this is the first analysis of UPF consumption among US veterans. TEIs coming from UPFs appear to be about 20% higher among veterans compared to nonveterans: 75% vs 55%.11 Coupled with high UPF consumption, MPFs (14%) and PRFs (9%) represent smaller sources of TEI among surveyed veterans. Top caloric sources of UPFs in the US include sandwiches (including burgers), sweet bakery products, savory snacks, pizza, sweetened beverages, and breads, rolls, and tortillas, and likely reflect the major sources of UPFs in the veteran diet.13 As the statistical comparison between the veteran data and nonveteran data is not feasible in the present study, a future study with a much larger sample size would be needed for a direct comparison.
While the exact cause of higher UPF consumption among sampled veterans remains unknown and likely multifactorial (eg, cost, food insecurity, access, cooking skills, nutrition knowledge), veterans can receive a consult or self-refer to a registered dietitian nutritionist (RDN) for nutrition education. Counseling has been shown to be an effective way to improve diet quality and increase daily fruit and vegetable intake.14 High consumption of UPFs, which are generally energy-dense and nutrient-poor, contributes to the low diet quality observed in veterans, and future research examining the relationship between UPF intake and overall diet quality among veterans is warranted.2,15 As nutrition knowledge is associated with higher diet quality among veterans, increased use of nutrition services (ie, nutrition education or food supplement programs) has the potential to influence consumption of MPFs and decrease consumption of UPFs.16 Subsequently, UPF-targeted interventions developed by VA RDNs hold the promise to decrease consumption of UPFs and increase intake of MPFs and PRFs.
Veterans have a high prevalence of diabetes, hypertension, and obesity.9 The high UPF intake observed in this sample of veterans may increase the risk for these chronic diseases and overall mortality. The high percentage of TEI from UPFs among veterans is also of concern not only due to potential negative health outcomes, but also associated costs of treating veterans with multimorbidities.17 Targeting UPF intake via nutritional education may promote health and decrease the financial burden needed to support the health of veterans.
Improving veteran health and well-being, including enhancing health care accessibility in underserved areas, are pivotal objectives of the VA strategic plan for 2026 to 2030. Public policy aims to tackle food insecurity within the veteran population during the first 5 years of civilian life.18 In alignment with the White House Strategy on Hunger, Nutrition, and Health, VA established a Food Security Office (FSO) in 2023. The FSO mission is to use an interdisciplinary approach to provide resources to ensure veteran food security and create an environment where all veterans are food and nutrition secure.
Limitations
This study has several limitations. As the Food Processor software database does not include all brand items, similar brands were used to mirror the nutrient profile. While food records are common among veteran diet studies, accuracy may be reduced due to self-reporting bias.19 Different interpretation of the NOVA classification designation for various food items is possible, however, 89% of foods were coded the same by the research team which suggests high accuracy in food coding. Specific ingredient information was not collected from the 3-day food records; thus, these records were not produced in such a way to improve the accuracy of the NOVA classification designation. This study was limited by its small sample size (N = 79); although, this analysis is larger than other studies of UPF consumption in the US.20,21 In addition, the generalizability of this study is limited as this population sample was from a single VA hospital and may not reflect the overall veteran population. Participants in this study were recruited only from those receiving VA care, thus their diet quality may not represent the quality consumed by veterans not participating in VA services. Further research on UPF consumption among veterans is warranted with a larger, more representative study sample size.
Conclusions
As this is the highest observed UPF intake documented in the US, these results should be of concern for the VA and its RDNs. More research is needed to better understand why UPF consumption is so high among veterans, what barriers veterans face to decreasing UPF consumption, and what intervention(s) veterans would welcome to improve their diet quality. Presently, veterans are provided with access to a variety of effective nutrition education and counseling options and should be encouraged to use these services. VA RDNs should be aware of the high intake of UPFs in the veteran population and familiarize themselves with education and counseling strategies that promote behavior change to replace UPFs with more nutrient-dense foods choices.
Roughly 8.6% of the 17.4 million US veterans live in poverty. About 11.1% are considered food insecure (ie, unable to acquire adequate food for ≥1 household members), with another 5.3% considered very food insecure (ie, eating patterns of ≥1 household members were disrupted and their food intake was reduced at least some time during the year). Compared with nonveterans, veterans are 7.4% more likely to be food insecure.1 This high prevalence of food insecurity and poverty has a negative impact on veteran diets.
Veterans’ diets contained more added sugars and solid fats and scored lower compared with nonveterans when assessed for diet quality with the Healthy Eating Index.2 Veterans have a higher prevalence of diet-related chronic disease, including diabetes, hypertension, and obesity compared with the nonveterans.3-5 Given the critical role of diet in health and disease risk, enhancing diet quality among veterans has garnered significant attention and calls to action.2,6,7 While there are many factors that contribute to diet, any veteran can receive a consultation or self-refer to receive nutrition counseling effective for improving diet quality, within the US Department of Veterans Affairs (VA).
The NOVA food classification system describes diet quality by categorizing food items by processing methods and ingredients into 4 food groups.8 The first is unprocessed and minimally processed items (MPFs) such as fresh fruits, vegetables, and meats. MPFs consist of whole foods which can also be minimally processed (eg, chopping, drying, grinding, heating, chilling). Culinary processed foods (CPFs) are processed foods for cooking (eg, salt, butter, and vinegar) and are typically eaten in small quantities along with MPFs. Processed foods (PRFs) include canned and smoked foods, while ultra-processed foods (UPFs) are distinguished by industrial ingredients, requiring specialized tools and processing techniques, and hyper-palatability related to color, flavor, and packaging.8 Examples of UPFs include mass-produced breads found at grocery stores, prepackaged snacks and meals, and hydrogenated oils. UPF consumption is associated with higher risk for negative cardiometabolic outcomes, common mental disorders, and all-cause mortality.9 To date, only a study by Powell et al has used the NOVA classification system in a veteran population, and it was limited to a comparison of the price of UPFs and veteran body mass index (BMI).10 Therefore, it remains unknown what percentage of total energy intake (TEI) comes from UPFs in the diets of veterans.
This study sought to quantify the proportion of TEI from UPFs among a sample of patients from the VA Phoenix Health Care System (VAPHCS). Results from a 2021 global meta-analysis reveal that the US and United Kingdom have the highest intakes of UPFs in the world.11 Specifically, within the US, 15 studies with 234,890 participants reveal that the majority of TEI (about 55%) comes from UPFs.11 We hypothesized that this veteran sample would have a higher proportion of TEI from UPFs, possibly due to a higher prevalence of poverty and food insecurity among veterans compared with nonveterans.1 If the percentage of TEI coming from UPF is higher or even similar to nonveterans, further efforts to increase veterans’ use of the available nutritional services would be warranted to minimize nutrition-related disease among veterans.
Methods
This is a cross-sectional, secondary data analysis of baseline 3-day food records collected from 2017 to 2020 from 92 patients recruited at VAPHCS to participate in a whole-food plant-based diet study.12 The original study was reviewed and approved by the VAPHCS Institutional Review Board (1593830). Recruitment methods included clinician recommendation, a recorded advertisement played while phone calls were on hold, and flyers distributed throughout VAPHCS. Patients were included if they were aged 18 to 90 years, had a BMI 25.1 to 39.9, had a diagnosis of nutrition-related chronic disease (hypertension, diabetes, or hyperlipidemia), an interest and desire to make a lifestyle change, active telephone contact information (either landline or cell phone), no contraindication to be on a whole-food plant-based diet, access to transportation and a functioning kitchen, ability to prepare meals independently, access to a computer or tablet with internet access, and a digital camera or smartphone. Exclusion criteria included significant unplanned weight loss within 6 months, uncontrolled insulin-dependent diabetes with a current hemoglobin A1c > 9%, pregnancy/lactation, taking prescribed weight loss medication, currently following a diet (eg, plant-based diet, vegan, or medical weight loss program diet), celiac disease diagnosed within 6 months, end-stage hepatic disease or renal disease requiring dialysis, active cancer or receiving chemotherapy or radiation therapy, active alcohol or substance use disorder, history of eating disorders, fasting triglyceride level > 350 mg/dL, any psychological issues that prevent adherence, inability to speak English, limited mobility, and homeless or in housing with limited kitchen access. A baseline 3-day food record was collected from the participants and used in this secondary analysis.
Diet Analysis
Food records were analyzed using Esha Research Food Processor 4.0 to identify calorie and macronutrient information. To limit bias, food items were coded independently by 2 researchers into 4 food processing groups determined by the NOVA classification: MPF, CPF, PRF, and UPF.8 When possible, specific ingredient information was collected using internet searches for brand product websites. Initial coding had an 89% agreement rate for food item coding between the 2 researchers. As coding was done in duplicate, a third researcher resolved disagreements. The number of food items for each processing group was determined and the mean (SD) percentage of TEI for each NOVA group was provided across participants. A 1-way analysis of variance and Tukey Multiple Comparisons Test were used to determine significance between groups with an α = .05 using Prism V9.
Results
Of the 92 participants in the original study, only 79 met inclusion criteria and had baseline diet data. The 79 veterans had a mean (SD) age of 61 (13) years and 59 (75%) were male (Table 1). Mean (SD) TEI was 1921 (815) kcal. The mean (SD) percentage of calories from carbohydrate, fat, and protein were 46% (21%), 39% (20%), and 16% (6%), respectively (Table 2).


A mean (SD) of 36 (12) food items were analyzed from the 3-day food records. The majority of food items were UPFs (56%), 33% were MPFs, 8% were PRFs, and 3% were CPFs. In total, 75% of TEI came from UPFs (P < .001); only 14% of TEI came from minimally processed foods (Figure).

Discussion
To our knowledge, this is the first analysis of UPF consumption among US veterans. TEIs coming from UPFs appear to be about 20% higher among veterans compared to nonveterans: 75% vs 55%.11 Coupled with high UPF consumption, MPFs (14%) and PRFs (9%) represent smaller sources of TEI among surveyed veterans. Top caloric sources of UPFs in the US include sandwiches (including burgers), sweet bakery products, savory snacks, pizza, sweetened beverages, and breads, rolls, and tortillas, and likely reflect the major sources of UPFs in the veteran diet.13 As the statistical comparison between the veteran data and nonveteran data is not feasible in the present study, a future study with a much larger sample size would be needed for a direct comparison.
While the exact cause of higher UPF consumption among sampled veterans remains unknown and likely multifactorial (eg, cost, food insecurity, access, cooking skills, nutrition knowledge), veterans can receive a consult or self-refer to a registered dietitian nutritionist (RDN) for nutrition education. Counseling has been shown to be an effective way to improve diet quality and increase daily fruit and vegetable intake.14 High consumption of UPFs, which are generally energy-dense and nutrient-poor, contributes to the low diet quality observed in veterans, and future research examining the relationship between UPF intake and overall diet quality among veterans is warranted.2,15 As nutrition knowledge is associated with higher diet quality among veterans, increased use of nutrition services (ie, nutrition education or food supplement programs) has the potential to influence consumption of MPFs and decrease consumption of UPFs.16 Subsequently, UPF-targeted interventions developed by VA RDNs hold the promise to decrease consumption of UPFs and increase intake of MPFs and PRFs.
Veterans have a high prevalence of diabetes, hypertension, and obesity.9 The high UPF intake observed in this sample of veterans may increase the risk for these chronic diseases and overall mortality. The high percentage of TEI from UPFs among veterans is also of concern not only due to potential negative health outcomes, but also associated costs of treating veterans with multimorbidities.17 Targeting UPF intake via nutritional education may promote health and decrease the financial burden needed to support the health of veterans.
Improving veteran health and well-being, including enhancing health care accessibility in underserved areas, are pivotal objectives of the VA strategic plan for 2026 to 2030. Public policy aims to tackle food insecurity within the veteran population during the first 5 years of civilian life.18 In alignment with the White House Strategy on Hunger, Nutrition, and Health, VA established a Food Security Office (FSO) in 2023. The FSO mission is to use an interdisciplinary approach to provide resources to ensure veteran food security and create an environment where all veterans are food and nutrition secure.
Limitations
This study has several limitations. As the Food Processor software database does not include all brand items, similar brands were used to mirror the nutrient profile. While food records are common among veteran diet studies, accuracy may be reduced due to self-reporting bias.19 Different interpretation of the NOVA classification designation for various food items is possible, however, 89% of foods were coded the same by the research team which suggests high accuracy in food coding. Specific ingredient information was not collected from the 3-day food records; thus, these records were not produced in such a way to improve the accuracy of the NOVA classification designation. This study was limited by its small sample size (N = 79); although, this analysis is larger than other studies of UPF consumption in the US.20,21 In addition, the generalizability of this study is limited as this population sample was from a single VA hospital and may not reflect the overall veteran population. Participants in this study were recruited only from those receiving VA care, thus their diet quality may not represent the quality consumed by veterans not participating in VA services. Further research on UPF consumption among veterans is warranted with a larger, more representative study sample size.
Conclusions
As this is the highest observed UPF intake documented in the US, these results should be of concern for the VA and its RDNs. More research is needed to better understand why UPF consumption is so high among veterans, what barriers veterans face to decreasing UPF consumption, and what intervention(s) veterans would welcome to improve their diet quality. Presently, veterans are provided with access to a variety of effective nutrition education and counseling options and should be encouraged to use these services. VA RDNs should be aware of the high intake of UPFs in the veteran population and familiarize themselves with education and counseling strategies that promote behavior change to replace UPFs with more nutrient-dense foods choices.
- Rabbitt MP, Smith MD. Food insecurity among workingage veterans. US Dept of Agriculture, Economic Research Service; 2021. Accessed January 26, 2026. https://www.ers.usda.gov/publications/pub-details/?pubid=101268
- Dong D, Stewart H, Carlson AC. An examination of veterans’ diet quality. US Dept of Agriculture, Economic Research Service; 2019. Accessed January 26, 2026. https:// www.ers.usda.gov/publications/pub-details/?pubid=95608
- US Department of Veterans Affairs; US Department of Defense. VA/DoD clinical practice guideline for the management of adult overweight and obesity. 2020. Accessed January 26, 2026. https://www.healthquality.va.gov/guidelines/cd/obesity/
- US Department of Veterans Affairs; US Department of Defense. VA/DoD clinical practice guideline for the management of type 2 diabetes mellitus in primary care. 2023. Accessed January 26, 2026. https://www.healthquality.va.gov/guidelines/cd/diabetes/
- Boersma P, Cohen R, Zelaya C, et al. Multiple chronic conditions among veterans and nonveterans: United States, 2015–2018. Natl Health Stat Rep. 2021. doi:10.15620/cdc:101659
- Hoerster KD, Wilson S, Nelson KM, et al. Diet quality is associated with mental health, social support, and neighborhood factors among veterans. Eat Behav. 2016;23:168- 173. doi:10.1016/j.eatbeh.2016.10.003
- Becerra MB, Hassija CM, Becerra BJ. Food insecurity is associated with unhealthy dietary practices among US veterans in California. Public Health Nutr. 2017;20:2569-2576. doi:10.1017/S1368980016002147
- Monteiro CA, Cannon G, Levy RB, et al. Ultra-processed foods: what they are and how to identify them. Public Health Nutr. 2019;22:936-941. doi:10.1017/S1368980018003762
- Lane MM, Gamage E, Du S, et al. Ultra-processed food exposure and adverse health outcomes: umbrella review of epidemiological meta-analyses. BMJ. February 2024:e077310. doi:10.1136/bmj-2023-077310
- Powell LM, Jones K, Duran AC, et al. The price of ultra-processed foods and beverages and adult body weight: Evidence from U.S. veterans. Econ Hum Biol. 2019;34:39- 48. doi:10.1016/j.ehb.2019.05.006
- Marino M, Puppo F, Del Bo’ C, et al. A systematic review of worldwide consumption of ultra-processed foods: findings and criticisms. Nutrients. 2021;13. doi:10.3390/nu13082778
- Parrington D, Kurtz J, Fawcett J, et al. Pilot study on the effects of a whole-food, plant-strong diet on cardiovascular risk factors in veterans: part 3. Curr Dev Nutr. 2022;6:385. doi:10.1093/cdn/nzac054.040
- Williams AM, Couch CA, Emmerich SD, et al. Ultra-processed Food Consumption in Youth and Adults: United States, August 2021-August 2023. NCHS Data Brief. 2025. doi:10.15620/cdc/174612
- Serra MC, Addison O, Giffuni J, et al. Changes in self-reported fruit and vegetable intake following nutritional modification in high risk older veterans. J Nutr Gerontol Geriatr. 2021;40:1-8. doi:10.1080/21551197.2020.1863892
- Gupta S, Hawk T, Aggarwal A, et al. Characterizing ultra-processed foods by energy density, nutrient density, and cost. Front Nutr. 2019;6:1-9. doi:10.3389/fnut.2019.00070
- Robinson LA, Colin CR, Smith KS, et al. Diet quality is associated with nutrition knowledge and physical activity in the US military veterans enrolled in university programmes. BMJ Mil Heal. 2023:e002525. doi:10.1136/military-2023-002525
- Yoon J, Zulman D, Scott JY, et al. Costs associated with multimorbidity among VA patients. Med Care. 2014;52:S31-6. doi:10.1097/MLR.0000000000000061
- End Veteran Hunger Act of 2022, H.R. 8852, 117th Cong. (2022). Accessed January 26, 2026. https://www.congress.gov/bill/117th-congress/house-bill/8852.
- Collins RA, Baker B, Coyle DH, et al. Dietary assessment methods in military and veteran populations: a scoping review. Nutrients. 2020;12:1-21. doi:10.3390/nu12030769
- Smiljanec K, Mbakwe AU, Ramos-Gonzalez M, et al. Associations of ultra-processed and unprocessed/minimally processed food consumption with peripheral and central hemodynamics, and arterial stiffness in young healthy adults. Nutrients. 2020;12. doi:10.3390/nu12113229
- Rohatgi KW, Tinius RA, Cade WT, et al. Relationships between consumption of ultra-processed foods, gestational weight gain and neonatal outcomes in a sample of US pregnant women. PeerJ. 2017;5:e4091. doi:10.7717/peerj.4091
- Rabbitt MP, Smith MD. Food insecurity among workingage veterans. US Dept of Agriculture, Economic Research Service; 2021. Accessed January 26, 2026. https://www.ers.usda.gov/publications/pub-details/?pubid=101268
- Dong D, Stewart H, Carlson AC. An examination of veterans’ diet quality. US Dept of Agriculture, Economic Research Service; 2019. Accessed January 26, 2026. https:// www.ers.usda.gov/publications/pub-details/?pubid=95608
- US Department of Veterans Affairs; US Department of Defense. VA/DoD clinical practice guideline for the management of adult overweight and obesity. 2020. Accessed January 26, 2026. https://www.healthquality.va.gov/guidelines/cd/obesity/
- US Department of Veterans Affairs; US Department of Defense. VA/DoD clinical practice guideline for the management of type 2 diabetes mellitus in primary care. 2023. Accessed January 26, 2026. https://www.healthquality.va.gov/guidelines/cd/diabetes/
- Boersma P, Cohen R, Zelaya C, et al. Multiple chronic conditions among veterans and nonveterans: United States, 2015–2018. Natl Health Stat Rep. 2021. doi:10.15620/cdc:101659
- Hoerster KD, Wilson S, Nelson KM, et al. Diet quality is associated with mental health, social support, and neighborhood factors among veterans. Eat Behav. 2016;23:168- 173. doi:10.1016/j.eatbeh.2016.10.003
- Becerra MB, Hassija CM, Becerra BJ. Food insecurity is associated with unhealthy dietary practices among US veterans in California. Public Health Nutr. 2017;20:2569-2576. doi:10.1017/S1368980016002147
- Monteiro CA, Cannon G, Levy RB, et al. Ultra-processed foods: what they are and how to identify them. Public Health Nutr. 2019;22:936-941. doi:10.1017/S1368980018003762
- Lane MM, Gamage E, Du S, et al. Ultra-processed food exposure and adverse health outcomes: umbrella review of epidemiological meta-analyses. BMJ. February 2024:e077310. doi:10.1136/bmj-2023-077310
- Powell LM, Jones K, Duran AC, et al. The price of ultra-processed foods and beverages and adult body weight: Evidence from U.S. veterans. Econ Hum Biol. 2019;34:39- 48. doi:10.1016/j.ehb.2019.05.006
- Marino M, Puppo F, Del Bo’ C, et al. A systematic review of worldwide consumption of ultra-processed foods: findings and criticisms. Nutrients. 2021;13. doi:10.3390/nu13082778
- Parrington D, Kurtz J, Fawcett J, et al. Pilot study on the effects of a whole-food, plant-strong diet on cardiovascular risk factors in veterans: part 3. Curr Dev Nutr. 2022;6:385. doi:10.1093/cdn/nzac054.040
- Williams AM, Couch CA, Emmerich SD, et al. Ultra-processed Food Consumption in Youth and Adults: United States, August 2021-August 2023. NCHS Data Brief. 2025. doi:10.15620/cdc/174612
- Serra MC, Addison O, Giffuni J, et al. Changes in self-reported fruit and vegetable intake following nutritional modification in high risk older veterans. J Nutr Gerontol Geriatr. 2021;40:1-8. doi:10.1080/21551197.2020.1863892
- Gupta S, Hawk T, Aggarwal A, et al. Characterizing ultra-processed foods by energy density, nutrient density, and cost. Front Nutr. 2019;6:1-9. doi:10.3389/fnut.2019.00070
- Robinson LA, Colin CR, Smith KS, et al. Diet quality is associated with nutrition knowledge and physical activity in the US military veterans enrolled in university programmes. BMJ Mil Heal. 2023:e002525. doi:10.1136/military-2023-002525
- Yoon J, Zulman D, Scott JY, et al. Costs associated with multimorbidity among VA patients. Med Care. 2014;52:S31-6. doi:10.1097/MLR.0000000000000061
- End Veteran Hunger Act of 2022, H.R. 8852, 117th Cong. (2022). Accessed January 26, 2026. https://www.congress.gov/bill/117th-congress/house-bill/8852.
- Collins RA, Baker B, Coyle DH, et al. Dietary assessment methods in military and veteran populations: a scoping review. Nutrients. 2020;12:1-21. doi:10.3390/nu12030769
- Smiljanec K, Mbakwe AU, Ramos-Gonzalez M, et al. Associations of ultra-processed and unprocessed/minimally processed food consumption with peripheral and central hemodynamics, and arterial stiffness in young healthy adults. Nutrients. 2020;12. doi:10.3390/nu12113229
- Rohatgi KW, Tinius RA, Cade WT, et al. Relationships between consumption of ultra-processed foods, gestational weight gain and neonatal outcomes in a sample of US pregnant women. PeerJ. 2017;5:e4091. doi:10.7717/peerj.4091
Seventy-Five Percent of Total Energy Intake Comes From Ultra-Processed Foods Among a Sample of Veterans With Overweight and Obesity: An Exploratory Analysis of Three-Day Food Records
Seventy-Five Percent of Total Energy Intake Comes From Ultra-Processed Foods Among a Sample of Veterans With Overweight and Obesity: An Exploratory Analysis of Three-Day Food Records
Frailty Trends in an Older Veteran Subpopulation 1 Year Prior and Into the COVID-19 Pandemic Using CAN Scores
Frailty is an age-associated, nonspecific vulnerability to adverse health outcomes. Frailty can also be described as a complex of symptoms characterized by impaired stress tolerance due to a decline in the functionality of different organs.1 The prevalence of frailty varies widely depending on the method of measurement and the population studied.2-4 It is a nonconstant factor that increases with age. A deficit accumulation frailty index (FI) is one method used to measure frailty.5 This approach sees frailty as a multidimensional risk state measured by quantity rather than the nature of health concerns. A deficit accumulation FI does not require physical testing but correlates well with other phenotypic FIs.6 It is, however, time consuming, as ≥ 30 deficits need to be measured to offer greater stability to the frailty estimate.
Health care is seeing increasing utilization of big data analytics to derive predictive models and help with resource allocation. There are currently 2 existing automated tools to predict health care utilization and mortality at the US Department of Veterans Affairs (VA): the VA Frailty Index (VA-FI-10) and the Care Assessment Need (CAN). VA-FI-10 is an International Statistical Classification of Diseases, Tenth Revision (ICD-10) update of the VA-FI that was created in March 2021. The VA-FI-10 is a claims-based frailty assessment tool using 31 health deficits. Calculating the VA-FI-10 requires defining an index date and lookback period (typically 3 years) relative to which it will be calculated.7
CAN is a set of risk-stratifying statistical models run on veterans receiving VA primary care services as part of a patient aligned care team (PACT) using electronic health record data.8 Each veteran is stratified based on the individual’s risks of hospitalization, death, and hospitalization or death. These 3 events are predicted for 90-day and 1-year time periods for a total of 6 distinct outcomes. CAN is currently on its third iteration (CAN 2.5) and scores range from 0 (low) to 99 (high). CAN scores are updated weekly. The 1-year hospitalization probabilities for all patients range from 0.8% to 93.1%. For patients with a CAN score of 50, the probability of being hospitalized within a year ranges from 4.5% to 5.2%, which increases to 32.2% to 36% for veterans with a CAN score of 95. The probability range widens significantly (32.2%-93.1%) for patients in the top 5 CAN scores (95-99).
CAN scores are a potential screening tool for frailty among older adults; they are generated automatically and provide acceptable diagnostic accuracy. Hence, the CAN score may be a useful tool for primary care practitioners for the detection of frailty in their patients. The CAN score has shown a moderate positive association with the FRAIL Scale.9,10 The population-based studies that have used the FI approach (differing FIs, depending on the data available) give robust results: People accumulate an average of 0.03 deficits per year after the age of 70 years.11 Interventions to delay or reverse frailty have not been clearly defined with heterogeneity in the definition of frailty and measurement of frailty outcomes.12,13 The prevalence of frailty in the veteran population is substantially higher than the prevalence in community populations with a similar age distribution. There is also mounting evidence that veterans accumulate deficits more rapidly than their civilian counterparts.14
COVID-19 was declared a pandemic in March 2020 and had many impacts on global health that were most marked in the first year. These included reductions in hospital visits for non-COVID-19 health concerns, a reduction in completed screening tests, an initial reduction in other infectious diseases (attributable to quarantines), and an increase or worsening of mental health concerns.15,16
We aimed to investigate whether frailty increased disproportionately in a subset of older veterans in the first year of the COVID-19 pandemic when compared with the previous year using CAN scores. This single institution, longitudinal cohort study was determined to be exempt from institutional review board review but was approved by the Phoenix VA Health Care System (PVAHCS) Research and Development Committee.
Methods
The Office of Clinical Systems Development and Evaluation (CSDE–10E2A) produces a weekly CAN Score Report to help identify the highest-risk patients in a primary care panel or cohort. CAN scores range from 0 (lowest risk) to 99 (highest risk), indicating how likely a patient is to experience hospitalization or death compared with other VA patients. CAN scores are calculated with statistical prediction models that use data elements from the following Corporate Data Warehouse (CDW) domains: demographics, health care utilization, laboratory tests, medical conditions, medications, and vital signs (eAppendix available online at 10.12788/fp.0385).
The CAN Score Report is generated weekly and stored on a CDW server. A patient will receive all 6 distinct CAN scores if they are: (1) assigned to a primary care PACT on the risk date; (2) a veteran; (3) not hospitalized in a VA facility on the risk date; and (4) alive as of the risk date. New to CAN 2.5 is that patients who meet criteria 1, 2, and 4 but are hospitalized in a VA facility on the risk date will receive CAN scores for the 1-year and 90-day mortality models.
Utilizing VA Informatics and Computing Infrastructure (VA HSR RES 13-457, US Department of Veterans Affairs [2008]), we obtained 2 lists of veterans aged 70 to 75 years on February 8, 2019, with a calculated CAN score of ≥ 75 for 1-year mortality and 1-year hospitalization on that date. A veteran with a CAN score of ≥ 75 is likely to be prefrail or frail.9,10 Veterans who did not have a corresponding calculated CAN score on February 7, 2020, and February 12, 2021, were excluded. COVID-19 was declared a public health emergency in the United States on January 31, 2020, and the World Health Organization declared COVID-19 a pandemic on March 11, 2020.17 We picked February 7, 2020, within this time frame and without any other special significance. We picked additional CAN score calculation dates approximately 1 year prior and 1 year after this date. Veterans had to be alive on February 12, 2021, (the last date of the CAN score) to be included in the cohorts.
Statistical Analyses
The difference in CAN score from one year to the next was calculated for each patient. The difference between 2019 and 2020 was compared with the difference between 2020 to 2021 using a paired t test. Yearly CAN score values were analyzed using repeated measures analysis of variance. The number of patients that showed an increase in CAN score (ie, increased risk of either mortality or hospitalization within the year) or a decrease (lower risk) was compared using the χ2 test. IBM SPSS v26 and GraphPad Prism v18 were used for statistical analysis. P < .05 was considered statistically significant.
Results
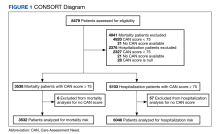
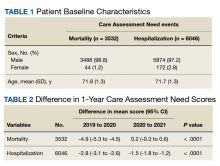
There were 3538 veterans at PVAHCS who met the inclusion criteria and had a 1-year mortality CAN score ≥ 75 on February 8, 2019.
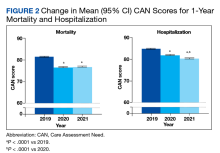
In the hospitalization group, there were 6046 veterans in the analysis; 57 veterans missing a 1-year hospitalization CAN score that were excluded. The mean age was 71.7 (1.3) years and included 5874 male (97.2%) and 172 female (2.8%) veterans. There was a decline in mean 1-year hospitalization CAN scores in our subset of frail older veterans by 2.8 (95% CI, -3.1 to -2.6) in the year preceding the COVID-19 pandemic. This mean decline slowed significantly to 1.5 (95% CI, -1.8 to -1.2; P < .0001) after the first year of the COVID-19 pandemic. Mean CAN scores for 1-year hospitalization were 84.6 (95% CI, 84.4 to 84.8), 81.8 (95% CI, 81.5 to 82.1), and 80.2 (95% CI, 79.9 to 80.6)
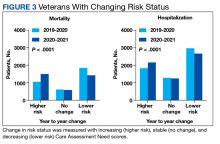
We also calculated the number of veterans with increasing, stable, and decreasing CAN scores across each of our defined periods in both the 1-year mortality and hospitalization groups.
A previous study used a 1-year combined hospitalization or mortality event CAN score as the most all-inclusive measure of frailty but determined that it was possible that 1 of the other 5 CAN risk measures could perform better in predicting frailty.10 We collected and presented data for 1-year mortality and hospitalization CAN scores. There were declines in pandemic-related US hospitalizations for illnesses not related to COVID-19 during the first few months of the pandemic.18 This may or may not have affected the 1-year hospitalization CAN score data; thus, we used the 1-year mortality CAN score data to predict frailty.
Discussion
We studied frailty trends in an older veteran subpopulation enrolled at the PVAHCS 1 year prior and into the COVID-19 pandemic using CAN scores. Frailty is a dynamic state. Previous frailty assessments aimed to identify patients at the highest risk of death. With the advent of advanced therapeutics for several diseases, the number of medical conditions that are now managed as chronic illnesses continues to grow. There is a role for repeated measures of frailty to try to identify frailty trends.19 These trends may assist us in resource allocation, identifying interventions that work and those that do not.
Some studies have shown an overall declining lethality of frailty. This may reflect improvements in the care and management of chronic conditions, screening tests, and increased awareness of healthy lifestyles.20 Another study of frailty trajectories in a veteran population in the 5 years preceding death showed multiple trajectories (stable, gradually increasing, rapidly increasing, and recovering).19
The PACT is a primary care model implemented at VA medical centers in April 2010. It is a patient-centered medical home model (PCMH) with several components. The VA treats a population of socioeconomically vulnerable patients with complex chronic illness management needs. Some of the components of a PACT model relevant to our study include facilitated self-management support for veterans in between practitioner visits via care partners, peer-to-peer and transitional care programs, physical activity and diet programs, primary care mental health, integration between primary and specialty care, and telehealth.21 A previous study has shown that VA primary care clinics with the most PCMH components in place had greater improvements in several chronic disease quality measures than in clinics with a lower number of PCMH components.22
Limitations
Our study is limited by our older veteran population demographics. We chose only a subset of older veterans at a single VA center for this study and cannot extrapolate the results to all older frail veterans or community dwelling older adults. Robust individuals may also transition to prefrailty and frailty over longer periods; our study monitored frailty trends over 2 years.
CAN scores are not quality measures to improve upon. Allocation and utilization of additional resources may clinically benefit a patient but increase their CAN scores. Although our results are statistically significant, we are unable to make any conclusions about clinical significance.
Conclusions
Our study results indicate frailty as determined by 1-year mortality CAN scores significantly increased in a subset of older veterans during the first year of the COVID-19 pandemic when compared with the previous year. Whether this change in frailty is temporary or long lasting remains to be seen. Automated CAN scores can be effectively utilized to monitor frailty trends in certain veteran populations over longer periods.
Acknowledgments
This material is the result of work supported with resources and the use of facilities at the Phoenix Veterans Affairs Health Care System.
1. Rohrmann S. Epidemiology of frailty in older people. Adv Exp Med Biol. 2020;1216:21-27. doi:10.1007/978-3-030-33330-0_3
2. Bandeen-Roche K, Seplaki CL, Huang J, et al. Frailty in older adults: a nationally representative profile in the United States. J Gerontol A Biol Sci Med Sci. 2015;70(11):1427-1434. doi:10.1093/gerona/glv133
3. Siriwardhana DD, Hardoon S, Rait G, Weerasinghe MC, Walters KR. Prevalence of frailty and prefrailty among community-dwelling older adults in low-income and middle-income countries: a systematic review and meta-analysis. BMJ Open. 2018;8(3):e018195. Published 2018 Mar 1. doi:10.1136/bmjopen-2017-018195
4. Song X, Mitnitski A, Rockwood K. Prevalence and 10-year outcomes of frailty in older adults in relation to deficit accumulation. J Am Geriatr Soc. 2010;58(4):681-687. doi:10.1111/j.1532-5415.2010.02764.x
5. Rockwood K, Mitnitski A. Frailty in relation to the accumulation of deficits. J Gerontol A Biol Sci Med Sci. 2007;62(7):722-727. doi:10.1093/gerona/62.7.722
6. Buta BJ, Walston JD, Godino JG, et al. Frailty assessment instruments: Systematic characterization of the uses and contexts of highly-cited instruments. Ageing Res Rev. 2016;26:53-61. doi:10.1016/j.arr.2015.12.003
7. Cheng D, DuMontier C, Yildirim C, et al. Updating and validating the U.S. Veterans Affairs Frailty Index: transitioning From ICD-9 to ICD-10. J Gerontol A Biol Sci Med Sci. 2021;76(7):1318-1325. doi:10.1093/gerona/glab071
8. Fihn SD, Francis J, Clancy C, et al. Insights from advanced analytics at the Veterans Health Administration. Health Aff (Millwood). 2014;33(7):1203-1211. doi:10.1377/hlthaff.2014.0054
9. Ruiz JG, Priyadarshni S, Rahaman Z, et al. Validation of an automatically generated screening score for frailty: the care assessment need (CAN) score. BMC Geriatr. 2018;18(1):106. doi:10.1186/s12877-018-0802-7
10. Ruiz JG, Rahaman Z, Dang S, Anam R, Valencia WM, Mintzer MJ. Association of the CAN score with the FRAIL scale in community dwelling older adults. Aging Clin Exp Res. 2018;30(10):1241-1245. doi:10.1007/s40520-018-0910-4
11. Ofori-Asenso R, Chin KL, Mazidi M, et al. Global incidence of frailty and prefrailty among community-dwelling older adults: a systematic review and meta-analysis. JAMA Netw Open. 2019;2(8):e198398. Published 2019 Aug 2. doi:10.1001/jamanetworkopen.2019.8398
12. Marcucci M, Damanti S, Germini F, et al. Interventions to prevent, delay or reverse frailty in older people: a journey towards clinical guidelines. BMC Med. 2019;17(1):193. Published 2019 Oct 29. doi:10.1186/s12916-019-1434-2
13. Travers J, Romero-Ortuno R, Bailey J, Cooney MT. Delaying and reversing frailty: a systematic review of primary care interventions. Br J Gen Pract. 2019;69(678):e61-e69. doi:10.3399/bjgp18X700241
14. Orkaby AR, Nussbaum L, Ho YL, et al. The burden of frailty among U.S. veterans and its association with mortality, 2002-2012. J Gerontol A Biol Sci Med Sci. 2019;74(8):1257-1264. doi:10.1093/gerona/gly232
15. Bakouny Z, Paciotti M, Schmidt AL, Lipsitz SR, Choueiri TK, Trinh QD. Cancer screening tests and cancer diagnoses during the COVID-19 pandemic. JAMA Oncol. 2021;7(3):458-460. doi:10.1001/jamaoncol.2020.7600
16. Steffen R, Lautenschlager S, Fehr J. Travel restrictions and lockdown during the COVID-19 pandemic-impact on notified infectious diseases in Switzerland. J Travel Med. 2020;27(8):taaa180. doi:10.1093/jtm/taaa180
17. CDC Museum COVID-19 Timeline. Centers for Disease Control and Prevention. Updated March 15, 2023. Accessed May 12, 2023. https://www.cdc.gov/museum/timeline/covid19.html18. Nguyen JL, Benigno M, Malhotra D, et al. Pandemic-related declines in hospitalization for non-COVID-19-related illness in the United States from January through July 2020. PLoS One. 2022;17(1):e0262347. Published 2022 Jan 6. doi:10.1371/journal.pone.0262347
19. Ward RE, Orkaby AR, Dumontier C, et al. Trajectories of frailty in the 5 years prior to death among U.S. veterans born 1927-1934. J Gerontol A Biol Sci Med Sci. 2021;76(11):e347-e353. doi:10.1093/gerona/glab196
20. Bäckman K, Joas E, Falk H, Mitnitski A, Rockwood K, Skoog I. Changes in the lethality of frailty over 30 years: evidence from two cohorts of 70-year-olds in Gothenburg Sweden. J Gerontol A Biol Sci Med Sci. 2017;72(7):945-950. doi:10.1093/gerona/glw160
21. Piette JD, Holtz B, Beard AJ, et al. Improving chronic illness care for veterans within the framework of the Patient-Centered Medical Home: experiences from the Ann Arbor Patient-Aligned Care Team Laboratory. Transl Behav Med. 2011;1(4):615-623. doi:10.1007/s13142-011-0065-8
22. Rosland AM, Nelson K, Sun H, et al. The patient-centered medical home in the Veterans Health Administration. Am J Manag Care. 2013;19(7):e263-e272. Published 2013 Jul 1.
Frailty is an age-associated, nonspecific vulnerability to adverse health outcomes. Frailty can also be described as a complex of symptoms characterized by impaired stress tolerance due to a decline in the functionality of different organs.1 The prevalence of frailty varies widely depending on the method of measurement and the population studied.2-4 It is a nonconstant factor that increases with age. A deficit accumulation frailty index (FI) is one method used to measure frailty.5 This approach sees frailty as a multidimensional risk state measured by quantity rather than the nature of health concerns. A deficit accumulation FI does not require physical testing but correlates well with other phenotypic FIs.6 It is, however, time consuming, as ≥ 30 deficits need to be measured to offer greater stability to the frailty estimate.
Health care is seeing increasing utilization of big data analytics to derive predictive models and help with resource allocation. There are currently 2 existing automated tools to predict health care utilization and mortality at the US Department of Veterans Affairs (VA): the VA Frailty Index (VA-FI-10) and the Care Assessment Need (CAN). VA-FI-10 is an International Statistical Classification of Diseases, Tenth Revision (ICD-10) update of the VA-FI that was created in March 2021. The VA-FI-10 is a claims-based frailty assessment tool using 31 health deficits. Calculating the VA-FI-10 requires defining an index date and lookback period (typically 3 years) relative to which it will be calculated.7
CAN is a set of risk-stratifying statistical models run on veterans receiving VA primary care services as part of a patient aligned care team (PACT) using electronic health record data.8 Each veteran is stratified based on the individual’s risks of hospitalization, death, and hospitalization or death. These 3 events are predicted for 90-day and 1-year time periods for a total of 6 distinct outcomes. CAN is currently on its third iteration (CAN 2.5) and scores range from 0 (low) to 99 (high). CAN scores are updated weekly. The 1-year hospitalization probabilities for all patients range from 0.8% to 93.1%. For patients with a CAN score of 50, the probability of being hospitalized within a year ranges from 4.5% to 5.2%, which increases to 32.2% to 36% for veterans with a CAN score of 95. The probability range widens significantly (32.2%-93.1%) for patients in the top 5 CAN scores (95-99).
CAN scores are a potential screening tool for frailty among older adults; they are generated automatically and provide acceptable diagnostic accuracy. Hence, the CAN score may be a useful tool for primary care practitioners for the detection of frailty in their patients. The CAN score has shown a moderate positive association with the FRAIL Scale.9,10 The population-based studies that have used the FI approach (differing FIs, depending on the data available) give robust results: People accumulate an average of 0.03 deficits per year after the age of 70 years.11 Interventions to delay or reverse frailty have not been clearly defined with heterogeneity in the definition of frailty and measurement of frailty outcomes.12,13 The prevalence of frailty in the veteran population is substantially higher than the prevalence in community populations with a similar age distribution. There is also mounting evidence that veterans accumulate deficits more rapidly than their civilian counterparts.14
COVID-19 was declared a pandemic in March 2020 and had many impacts on global health that were most marked in the first year. These included reductions in hospital visits for non-COVID-19 health concerns, a reduction in completed screening tests, an initial reduction in other infectious diseases (attributable to quarantines), and an increase or worsening of mental health concerns.15,16
We aimed to investigate whether frailty increased disproportionately in a subset of older veterans in the first year of the COVID-19 pandemic when compared with the previous year using CAN scores. This single institution, longitudinal cohort study was determined to be exempt from institutional review board review but was approved by the Phoenix VA Health Care System (PVAHCS) Research and Development Committee.
Methods
The Office of Clinical Systems Development and Evaluation (CSDE–10E2A) produces a weekly CAN Score Report to help identify the highest-risk patients in a primary care panel or cohort. CAN scores range from 0 (lowest risk) to 99 (highest risk), indicating how likely a patient is to experience hospitalization or death compared with other VA patients. CAN scores are calculated with statistical prediction models that use data elements from the following Corporate Data Warehouse (CDW) domains: demographics, health care utilization, laboratory tests, medical conditions, medications, and vital signs (eAppendix available online at 10.12788/fp.0385).
The CAN Score Report is generated weekly and stored on a CDW server. A patient will receive all 6 distinct CAN scores if they are: (1) assigned to a primary care PACT on the risk date; (2) a veteran; (3) not hospitalized in a VA facility on the risk date; and (4) alive as of the risk date. New to CAN 2.5 is that patients who meet criteria 1, 2, and 4 but are hospitalized in a VA facility on the risk date will receive CAN scores for the 1-year and 90-day mortality models.
Utilizing VA Informatics and Computing Infrastructure (VA HSR RES 13-457, US Department of Veterans Affairs [2008]), we obtained 2 lists of veterans aged 70 to 75 years on February 8, 2019, with a calculated CAN score of ≥ 75 for 1-year mortality and 1-year hospitalization on that date. A veteran with a CAN score of ≥ 75 is likely to be prefrail or frail.9,10 Veterans who did not have a corresponding calculated CAN score on February 7, 2020, and February 12, 2021, were excluded. COVID-19 was declared a public health emergency in the United States on January 31, 2020, and the World Health Organization declared COVID-19 a pandemic on March 11, 2020.17 We picked February 7, 2020, within this time frame and without any other special significance. We picked additional CAN score calculation dates approximately 1 year prior and 1 year after this date. Veterans had to be alive on February 12, 2021, (the last date of the CAN score) to be included in the cohorts.
Statistical Analyses
The difference in CAN score from one year to the next was calculated for each patient. The difference between 2019 and 2020 was compared with the difference between 2020 to 2021 using a paired t test. Yearly CAN score values were analyzed using repeated measures analysis of variance. The number of patients that showed an increase in CAN score (ie, increased risk of either mortality or hospitalization within the year) or a decrease (lower risk) was compared using the χ2 test. IBM SPSS v26 and GraphPad Prism v18 were used for statistical analysis. P < .05 was considered statistically significant.
Results
There were 3538 veterans at PVAHCS who met the inclusion criteria and had a 1-year mortality CAN score ≥ 75 on February 8, 2019.
In the hospitalization group, there were 6046 veterans in the analysis; 57 veterans missing a 1-year hospitalization CAN score that were excluded. The mean age was 71.7 (1.3) years and included 5874 male (97.2%) and 172 female (2.8%) veterans. There was a decline in mean 1-year hospitalization CAN scores in our subset of frail older veterans by 2.8 (95% CI, -3.1 to -2.6) in the year preceding the COVID-19 pandemic. This mean decline slowed significantly to 1.5 (95% CI, -1.8 to -1.2; P < .0001) after the first year of the COVID-19 pandemic. Mean CAN scores for 1-year hospitalization were 84.6 (95% CI, 84.4 to 84.8), 81.8 (95% CI, 81.5 to 82.1), and 80.2 (95% CI, 79.9 to 80.6)
We also calculated the number of veterans with increasing, stable, and decreasing CAN scores across each of our defined periods in both the 1-year mortality and hospitalization groups.
A previous study used a 1-year combined hospitalization or mortality event CAN score as the most all-inclusive measure of frailty but determined that it was possible that 1 of the other 5 CAN risk measures could perform better in predicting frailty.10 We collected and presented data for 1-year mortality and hospitalization CAN scores. There were declines in pandemic-related US hospitalizations for illnesses not related to COVID-19 during the first few months of the pandemic.18 This may or may not have affected the 1-year hospitalization CAN score data; thus, we used the 1-year mortality CAN score data to predict frailty.
Discussion
We studied frailty trends in an older veteran subpopulation enrolled at the PVAHCS 1 year prior and into the COVID-19 pandemic using CAN scores. Frailty is a dynamic state. Previous frailty assessments aimed to identify patients at the highest risk of death. With the advent of advanced therapeutics for several diseases, the number of medical conditions that are now managed as chronic illnesses continues to grow. There is a role for repeated measures of frailty to try to identify frailty trends.19 These trends may assist us in resource allocation, identifying interventions that work and those that do not.
Some studies have shown an overall declining lethality of frailty. This may reflect improvements in the care and management of chronic conditions, screening tests, and increased awareness of healthy lifestyles.20 Another study of frailty trajectories in a veteran population in the 5 years preceding death showed multiple trajectories (stable, gradually increasing, rapidly increasing, and recovering).19
The PACT is a primary care model implemented at VA medical centers in April 2010. It is a patient-centered medical home model (PCMH) with several components. The VA treats a population of socioeconomically vulnerable patients with complex chronic illness management needs. Some of the components of a PACT model relevant to our study include facilitated self-management support for veterans in between practitioner visits via care partners, peer-to-peer and transitional care programs, physical activity and diet programs, primary care mental health, integration between primary and specialty care, and telehealth.21 A previous study has shown that VA primary care clinics with the most PCMH components in place had greater improvements in several chronic disease quality measures than in clinics with a lower number of PCMH components.22
Limitations
Our study is limited by our older veteran population demographics. We chose only a subset of older veterans at a single VA center for this study and cannot extrapolate the results to all older frail veterans or community dwelling older adults. Robust individuals may also transition to prefrailty and frailty over longer periods; our study monitored frailty trends over 2 years.
CAN scores are not quality measures to improve upon. Allocation and utilization of additional resources may clinically benefit a patient but increase their CAN scores. Although our results are statistically significant, we are unable to make any conclusions about clinical significance.
Conclusions
Our study results indicate frailty as determined by 1-year mortality CAN scores significantly increased in a subset of older veterans during the first year of the COVID-19 pandemic when compared with the previous year. Whether this change in frailty is temporary or long lasting remains to be seen. Automated CAN scores can be effectively utilized to monitor frailty trends in certain veteran populations over longer periods.
Acknowledgments
This material is the result of work supported with resources and the use of facilities at the Phoenix Veterans Affairs Health Care System.
Frailty is an age-associated, nonspecific vulnerability to adverse health outcomes. Frailty can also be described as a complex of symptoms characterized by impaired stress tolerance due to a decline in the functionality of different organs.1 The prevalence of frailty varies widely depending on the method of measurement and the population studied.2-4 It is a nonconstant factor that increases with age. A deficit accumulation frailty index (FI) is one method used to measure frailty.5 This approach sees frailty as a multidimensional risk state measured by quantity rather than the nature of health concerns. A deficit accumulation FI does not require physical testing but correlates well with other phenotypic FIs.6 It is, however, time consuming, as ≥ 30 deficits need to be measured to offer greater stability to the frailty estimate.
Health care is seeing increasing utilization of big data analytics to derive predictive models and help with resource allocation. There are currently 2 existing automated tools to predict health care utilization and mortality at the US Department of Veterans Affairs (VA): the VA Frailty Index (VA-FI-10) and the Care Assessment Need (CAN). VA-FI-10 is an International Statistical Classification of Diseases, Tenth Revision (ICD-10) update of the VA-FI that was created in March 2021. The VA-FI-10 is a claims-based frailty assessment tool using 31 health deficits. Calculating the VA-FI-10 requires defining an index date and lookback period (typically 3 years) relative to which it will be calculated.7
CAN is a set of risk-stratifying statistical models run on veterans receiving VA primary care services as part of a patient aligned care team (PACT) using electronic health record data.8 Each veteran is stratified based on the individual’s risks of hospitalization, death, and hospitalization or death. These 3 events are predicted for 90-day and 1-year time periods for a total of 6 distinct outcomes. CAN is currently on its third iteration (CAN 2.5) and scores range from 0 (low) to 99 (high). CAN scores are updated weekly. The 1-year hospitalization probabilities for all patients range from 0.8% to 93.1%. For patients with a CAN score of 50, the probability of being hospitalized within a year ranges from 4.5% to 5.2%, which increases to 32.2% to 36% for veterans with a CAN score of 95. The probability range widens significantly (32.2%-93.1%) for patients in the top 5 CAN scores (95-99).
CAN scores are a potential screening tool for frailty among older adults; they are generated automatically and provide acceptable diagnostic accuracy. Hence, the CAN score may be a useful tool for primary care practitioners for the detection of frailty in their patients. The CAN score has shown a moderate positive association with the FRAIL Scale.9,10 The population-based studies that have used the FI approach (differing FIs, depending on the data available) give robust results: People accumulate an average of 0.03 deficits per year after the age of 70 years.11 Interventions to delay or reverse frailty have not been clearly defined with heterogeneity in the definition of frailty and measurement of frailty outcomes.12,13 The prevalence of frailty in the veteran population is substantially higher than the prevalence in community populations with a similar age distribution. There is also mounting evidence that veterans accumulate deficits more rapidly than their civilian counterparts.14
COVID-19 was declared a pandemic in March 2020 and had many impacts on global health that were most marked in the first year. These included reductions in hospital visits for non-COVID-19 health concerns, a reduction in completed screening tests, an initial reduction in other infectious diseases (attributable to quarantines), and an increase or worsening of mental health concerns.15,16
We aimed to investigate whether frailty increased disproportionately in a subset of older veterans in the first year of the COVID-19 pandemic when compared with the previous year using CAN scores. This single institution, longitudinal cohort study was determined to be exempt from institutional review board review but was approved by the Phoenix VA Health Care System (PVAHCS) Research and Development Committee.
Methods
The Office of Clinical Systems Development and Evaluation (CSDE–10E2A) produces a weekly CAN Score Report to help identify the highest-risk patients in a primary care panel or cohort. CAN scores range from 0 (lowest risk) to 99 (highest risk), indicating how likely a patient is to experience hospitalization or death compared with other VA patients. CAN scores are calculated with statistical prediction models that use data elements from the following Corporate Data Warehouse (CDW) domains: demographics, health care utilization, laboratory tests, medical conditions, medications, and vital signs (eAppendix available online at 10.12788/fp.0385).
The CAN Score Report is generated weekly and stored on a CDW server. A patient will receive all 6 distinct CAN scores if they are: (1) assigned to a primary care PACT on the risk date; (2) a veteran; (3) not hospitalized in a VA facility on the risk date; and (4) alive as of the risk date. New to CAN 2.5 is that patients who meet criteria 1, 2, and 4 but are hospitalized in a VA facility on the risk date will receive CAN scores for the 1-year and 90-day mortality models.
Utilizing VA Informatics and Computing Infrastructure (VA HSR RES 13-457, US Department of Veterans Affairs [2008]), we obtained 2 lists of veterans aged 70 to 75 years on February 8, 2019, with a calculated CAN score of ≥ 75 for 1-year mortality and 1-year hospitalization on that date. A veteran with a CAN score of ≥ 75 is likely to be prefrail or frail.9,10 Veterans who did not have a corresponding calculated CAN score on February 7, 2020, and February 12, 2021, were excluded. COVID-19 was declared a public health emergency in the United States on January 31, 2020, and the World Health Organization declared COVID-19 a pandemic on March 11, 2020.17 We picked February 7, 2020, within this time frame and without any other special significance. We picked additional CAN score calculation dates approximately 1 year prior and 1 year after this date. Veterans had to be alive on February 12, 2021, (the last date of the CAN score) to be included in the cohorts.
Statistical Analyses
The difference in CAN score from one year to the next was calculated for each patient. The difference between 2019 and 2020 was compared with the difference between 2020 to 2021 using a paired t test. Yearly CAN score values were analyzed using repeated measures analysis of variance. The number of patients that showed an increase in CAN score (ie, increased risk of either mortality or hospitalization within the year) or a decrease (lower risk) was compared using the χ2 test. IBM SPSS v26 and GraphPad Prism v18 were used for statistical analysis. P < .05 was considered statistically significant.
Results
There were 3538 veterans at PVAHCS who met the inclusion criteria and had a 1-year mortality CAN score ≥ 75 on February 8, 2019.
In the hospitalization group, there were 6046 veterans in the analysis; 57 veterans missing a 1-year hospitalization CAN score that were excluded. The mean age was 71.7 (1.3) years and included 5874 male (97.2%) and 172 female (2.8%) veterans. There was a decline in mean 1-year hospitalization CAN scores in our subset of frail older veterans by 2.8 (95% CI, -3.1 to -2.6) in the year preceding the COVID-19 pandemic. This mean decline slowed significantly to 1.5 (95% CI, -1.8 to -1.2; P < .0001) after the first year of the COVID-19 pandemic. Mean CAN scores for 1-year hospitalization were 84.6 (95% CI, 84.4 to 84.8), 81.8 (95% CI, 81.5 to 82.1), and 80.2 (95% CI, 79.9 to 80.6)
We also calculated the number of veterans with increasing, stable, and decreasing CAN scores across each of our defined periods in both the 1-year mortality and hospitalization groups.
A previous study used a 1-year combined hospitalization or mortality event CAN score as the most all-inclusive measure of frailty but determined that it was possible that 1 of the other 5 CAN risk measures could perform better in predicting frailty.10 We collected and presented data for 1-year mortality and hospitalization CAN scores. There were declines in pandemic-related US hospitalizations for illnesses not related to COVID-19 during the first few months of the pandemic.18 This may or may not have affected the 1-year hospitalization CAN score data; thus, we used the 1-year mortality CAN score data to predict frailty.
Discussion
We studied frailty trends in an older veteran subpopulation enrolled at the PVAHCS 1 year prior and into the COVID-19 pandemic using CAN scores. Frailty is a dynamic state. Previous frailty assessments aimed to identify patients at the highest risk of death. With the advent of advanced therapeutics for several diseases, the number of medical conditions that are now managed as chronic illnesses continues to grow. There is a role for repeated measures of frailty to try to identify frailty trends.19 These trends may assist us in resource allocation, identifying interventions that work and those that do not.
Some studies have shown an overall declining lethality of frailty. This may reflect improvements in the care and management of chronic conditions, screening tests, and increased awareness of healthy lifestyles.20 Another study of frailty trajectories in a veteran population in the 5 years preceding death showed multiple trajectories (stable, gradually increasing, rapidly increasing, and recovering).19
The PACT is a primary care model implemented at VA medical centers in April 2010. It is a patient-centered medical home model (PCMH) with several components. The VA treats a population of socioeconomically vulnerable patients with complex chronic illness management needs. Some of the components of a PACT model relevant to our study include facilitated self-management support for veterans in between practitioner visits via care partners, peer-to-peer and transitional care programs, physical activity and diet programs, primary care mental health, integration between primary and specialty care, and telehealth.21 A previous study has shown that VA primary care clinics with the most PCMH components in place had greater improvements in several chronic disease quality measures than in clinics with a lower number of PCMH components.22
Limitations
Our study is limited by our older veteran population demographics. We chose only a subset of older veterans at a single VA center for this study and cannot extrapolate the results to all older frail veterans or community dwelling older adults. Robust individuals may also transition to prefrailty and frailty over longer periods; our study monitored frailty trends over 2 years.
CAN scores are not quality measures to improve upon. Allocation and utilization of additional resources may clinically benefit a patient but increase their CAN scores. Although our results are statistically significant, we are unable to make any conclusions about clinical significance.
Conclusions
Our study results indicate frailty as determined by 1-year mortality CAN scores significantly increased in a subset of older veterans during the first year of the COVID-19 pandemic when compared with the previous year. Whether this change in frailty is temporary or long lasting remains to be seen. Automated CAN scores can be effectively utilized to monitor frailty trends in certain veteran populations over longer periods.
Acknowledgments
This material is the result of work supported with resources and the use of facilities at the Phoenix Veterans Affairs Health Care System.
1. Rohrmann S. Epidemiology of frailty in older people. Adv Exp Med Biol. 2020;1216:21-27. doi:10.1007/978-3-030-33330-0_3
2. Bandeen-Roche K, Seplaki CL, Huang J, et al. Frailty in older adults: a nationally representative profile in the United States. J Gerontol A Biol Sci Med Sci. 2015;70(11):1427-1434. doi:10.1093/gerona/glv133
3. Siriwardhana DD, Hardoon S, Rait G, Weerasinghe MC, Walters KR. Prevalence of frailty and prefrailty among community-dwelling older adults in low-income and middle-income countries: a systematic review and meta-analysis. BMJ Open. 2018;8(3):e018195. Published 2018 Mar 1. doi:10.1136/bmjopen-2017-018195
4. Song X, Mitnitski A, Rockwood K. Prevalence and 10-year outcomes of frailty in older adults in relation to deficit accumulation. J Am Geriatr Soc. 2010;58(4):681-687. doi:10.1111/j.1532-5415.2010.02764.x
5. Rockwood K, Mitnitski A. Frailty in relation to the accumulation of deficits. J Gerontol A Biol Sci Med Sci. 2007;62(7):722-727. doi:10.1093/gerona/62.7.722
6. Buta BJ, Walston JD, Godino JG, et al. Frailty assessment instruments: Systematic characterization of the uses and contexts of highly-cited instruments. Ageing Res Rev. 2016;26:53-61. doi:10.1016/j.arr.2015.12.003
7. Cheng D, DuMontier C, Yildirim C, et al. Updating and validating the U.S. Veterans Affairs Frailty Index: transitioning From ICD-9 to ICD-10. J Gerontol A Biol Sci Med Sci. 2021;76(7):1318-1325. doi:10.1093/gerona/glab071
8. Fihn SD, Francis J, Clancy C, et al. Insights from advanced analytics at the Veterans Health Administration. Health Aff (Millwood). 2014;33(7):1203-1211. doi:10.1377/hlthaff.2014.0054
9. Ruiz JG, Priyadarshni S, Rahaman Z, et al. Validation of an automatically generated screening score for frailty: the care assessment need (CAN) score. BMC Geriatr. 2018;18(1):106. doi:10.1186/s12877-018-0802-7
10. Ruiz JG, Rahaman Z, Dang S, Anam R, Valencia WM, Mintzer MJ. Association of the CAN score with the FRAIL scale in community dwelling older adults. Aging Clin Exp Res. 2018;30(10):1241-1245. doi:10.1007/s40520-018-0910-4
11. Ofori-Asenso R, Chin KL, Mazidi M, et al. Global incidence of frailty and prefrailty among community-dwelling older adults: a systematic review and meta-analysis. JAMA Netw Open. 2019;2(8):e198398. Published 2019 Aug 2. doi:10.1001/jamanetworkopen.2019.8398
12. Marcucci M, Damanti S, Germini F, et al. Interventions to prevent, delay or reverse frailty in older people: a journey towards clinical guidelines. BMC Med. 2019;17(1):193. Published 2019 Oct 29. doi:10.1186/s12916-019-1434-2
13. Travers J, Romero-Ortuno R, Bailey J, Cooney MT. Delaying and reversing frailty: a systematic review of primary care interventions. Br J Gen Pract. 2019;69(678):e61-e69. doi:10.3399/bjgp18X700241
14. Orkaby AR, Nussbaum L, Ho YL, et al. The burden of frailty among U.S. veterans and its association with mortality, 2002-2012. J Gerontol A Biol Sci Med Sci. 2019;74(8):1257-1264. doi:10.1093/gerona/gly232
15. Bakouny Z, Paciotti M, Schmidt AL, Lipsitz SR, Choueiri TK, Trinh QD. Cancer screening tests and cancer diagnoses during the COVID-19 pandemic. JAMA Oncol. 2021;7(3):458-460. doi:10.1001/jamaoncol.2020.7600
16. Steffen R, Lautenschlager S, Fehr J. Travel restrictions and lockdown during the COVID-19 pandemic-impact on notified infectious diseases in Switzerland. J Travel Med. 2020;27(8):taaa180. doi:10.1093/jtm/taaa180
17. CDC Museum COVID-19 Timeline. Centers for Disease Control and Prevention. Updated March 15, 2023. Accessed May 12, 2023. https://www.cdc.gov/museum/timeline/covid19.html18. Nguyen JL, Benigno M, Malhotra D, et al. Pandemic-related declines in hospitalization for non-COVID-19-related illness in the United States from January through July 2020. PLoS One. 2022;17(1):e0262347. Published 2022 Jan 6. doi:10.1371/journal.pone.0262347
19. Ward RE, Orkaby AR, Dumontier C, et al. Trajectories of frailty in the 5 years prior to death among U.S. veterans born 1927-1934. J Gerontol A Biol Sci Med Sci. 2021;76(11):e347-e353. doi:10.1093/gerona/glab196
20. Bäckman K, Joas E, Falk H, Mitnitski A, Rockwood K, Skoog I. Changes in the lethality of frailty over 30 years: evidence from two cohorts of 70-year-olds in Gothenburg Sweden. J Gerontol A Biol Sci Med Sci. 2017;72(7):945-950. doi:10.1093/gerona/glw160
21. Piette JD, Holtz B, Beard AJ, et al. Improving chronic illness care for veterans within the framework of the Patient-Centered Medical Home: experiences from the Ann Arbor Patient-Aligned Care Team Laboratory. Transl Behav Med. 2011;1(4):615-623. doi:10.1007/s13142-011-0065-8
22. Rosland AM, Nelson K, Sun H, et al. The patient-centered medical home in the Veterans Health Administration. Am J Manag Care. 2013;19(7):e263-e272. Published 2013 Jul 1.
1. Rohrmann S. Epidemiology of frailty in older people. Adv Exp Med Biol. 2020;1216:21-27. doi:10.1007/978-3-030-33330-0_3
2. Bandeen-Roche K, Seplaki CL, Huang J, et al. Frailty in older adults: a nationally representative profile in the United States. J Gerontol A Biol Sci Med Sci. 2015;70(11):1427-1434. doi:10.1093/gerona/glv133
3. Siriwardhana DD, Hardoon S, Rait G, Weerasinghe MC, Walters KR. Prevalence of frailty and prefrailty among community-dwelling older adults in low-income and middle-income countries: a systematic review and meta-analysis. BMJ Open. 2018;8(3):e018195. Published 2018 Mar 1. doi:10.1136/bmjopen-2017-018195
4. Song X, Mitnitski A, Rockwood K. Prevalence and 10-year outcomes of frailty in older adults in relation to deficit accumulation. J Am Geriatr Soc. 2010;58(4):681-687. doi:10.1111/j.1532-5415.2010.02764.x
5. Rockwood K, Mitnitski A. Frailty in relation to the accumulation of deficits. J Gerontol A Biol Sci Med Sci. 2007;62(7):722-727. doi:10.1093/gerona/62.7.722
6. Buta BJ, Walston JD, Godino JG, et al. Frailty assessment instruments: Systematic characterization of the uses and contexts of highly-cited instruments. Ageing Res Rev. 2016;26:53-61. doi:10.1016/j.arr.2015.12.003
7. Cheng D, DuMontier C, Yildirim C, et al. Updating and validating the U.S. Veterans Affairs Frailty Index: transitioning From ICD-9 to ICD-10. J Gerontol A Biol Sci Med Sci. 2021;76(7):1318-1325. doi:10.1093/gerona/glab071
8. Fihn SD, Francis J, Clancy C, et al. Insights from advanced analytics at the Veterans Health Administration. Health Aff (Millwood). 2014;33(7):1203-1211. doi:10.1377/hlthaff.2014.0054
9. Ruiz JG, Priyadarshni S, Rahaman Z, et al. Validation of an automatically generated screening score for frailty: the care assessment need (CAN) score. BMC Geriatr. 2018;18(1):106. doi:10.1186/s12877-018-0802-7
10. Ruiz JG, Rahaman Z, Dang S, Anam R, Valencia WM, Mintzer MJ. Association of the CAN score with the FRAIL scale in community dwelling older adults. Aging Clin Exp Res. 2018;30(10):1241-1245. doi:10.1007/s40520-018-0910-4
11. Ofori-Asenso R, Chin KL, Mazidi M, et al. Global incidence of frailty and prefrailty among community-dwelling older adults: a systematic review and meta-analysis. JAMA Netw Open. 2019;2(8):e198398. Published 2019 Aug 2. doi:10.1001/jamanetworkopen.2019.8398
12. Marcucci M, Damanti S, Germini F, et al. Interventions to prevent, delay or reverse frailty in older people: a journey towards clinical guidelines. BMC Med. 2019;17(1):193. Published 2019 Oct 29. doi:10.1186/s12916-019-1434-2
13. Travers J, Romero-Ortuno R, Bailey J, Cooney MT. Delaying and reversing frailty: a systematic review of primary care interventions. Br J Gen Pract. 2019;69(678):e61-e69. doi:10.3399/bjgp18X700241
14. Orkaby AR, Nussbaum L, Ho YL, et al. The burden of frailty among U.S. veterans and its association with mortality, 2002-2012. J Gerontol A Biol Sci Med Sci. 2019;74(8):1257-1264. doi:10.1093/gerona/gly232
15. Bakouny Z, Paciotti M, Schmidt AL, Lipsitz SR, Choueiri TK, Trinh QD. Cancer screening tests and cancer diagnoses during the COVID-19 pandemic. JAMA Oncol. 2021;7(3):458-460. doi:10.1001/jamaoncol.2020.7600
16. Steffen R, Lautenschlager S, Fehr J. Travel restrictions and lockdown during the COVID-19 pandemic-impact on notified infectious diseases in Switzerland. J Travel Med. 2020;27(8):taaa180. doi:10.1093/jtm/taaa180
17. CDC Museum COVID-19 Timeline. Centers for Disease Control and Prevention. Updated March 15, 2023. Accessed May 12, 2023. https://www.cdc.gov/museum/timeline/covid19.html18. Nguyen JL, Benigno M, Malhotra D, et al. Pandemic-related declines in hospitalization for non-COVID-19-related illness in the United States from January through July 2020. PLoS One. 2022;17(1):e0262347. Published 2022 Jan 6. doi:10.1371/journal.pone.0262347
19. Ward RE, Orkaby AR, Dumontier C, et al. Trajectories of frailty in the 5 years prior to death among U.S. veterans born 1927-1934. J Gerontol A Biol Sci Med Sci. 2021;76(11):e347-e353. doi:10.1093/gerona/glab196
20. Bäckman K, Joas E, Falk H, Mitnitski A, Rockwood K, Skoog I. Changes in the lethality of frailty over 30 years: evidence from two cohorts of 70-year-olds in Gothenburg Sweden. J Gerontol A Biol Sci Med Sci. 2017;72(7):945-950. doi:10.1093/gerona/glw160
21. Piette JD, Holtz B, Beard AJ, et al. Improving chronic illness care for veterans within the framework of the Patient-Centered Medical Home: experiences from the Ann Arbor Patient-Aligned Care Team Laboratory. Transl Behav Med. 2011;1(4):615-623. doi:10.1007/s13142-011-0065-8
22. Rosland AM, Nelson K, Sun H, et al. The patient-centered medical home in the Veterans Health Administration. Am J Manag Care. 2013;19(7):e263-e272. Published 2013 Jul 1.