User login
WCD: Psoriasis plus depression magnifies MI risk
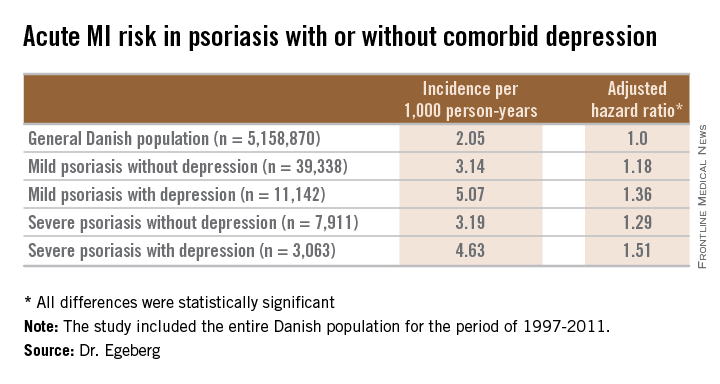
VANCOUVER, B.C. – The combination of even mild psoriasis and a diagnosis of depression is associated with a greater risk of acute MI than is either condition alone, according to a Danish national study.
Moreover, when depression coincided with a severe case of psoriasis, not only was the risk of incident acute MI elevated, but the risk that the MI would be fatal was significantly increased as well, Dr. Alexander Egeberg reported at the World Congress of Dermatology.
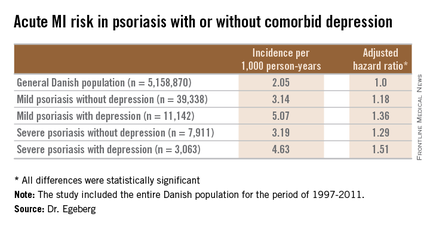
He presented a population-based cohort study of the entire Danish population, for the period of 1997-2011. The study population comprised 5,158,870 Danish citizens, including 50,480 Danes diagnosed with mild psoriasis and 10,974 others diagnosed with severe psoriasis during the study years. During that period, 11,142 members of the group with mild psoriasis and 3,063 with severe psoriasis were diagnosed with incident depression.
In a multivariate Cox regression analysis adjusted for age, gender, comorbid conditions, and concomitant medications, Danes with mild psoriasis but no diagnosis of depression were at a statistically significant 18% increased risk of having an acute MI compared to the general Danish population free of both diseases; the risk climbed upward from there (see chart).
A consistent trend for an increased risk of fatal MI was evident in patients with psoriasis, be it mild or severe, and with or without comorbid depression. However, this risk achieved statistical significance only in the group with severe psoriasis plus depression, a state associated with a 64% increased risk of fatal MI compared with the general population.
Although recent years have brought clear evidence that psoriasis is associated with increased risks of heart disease and depression, and that stand-alone depression is a risk factor for ischemic coronary events, the link between psoriasis, depression, and MI previously was unclear, explained Dr. Egeberg, who performed the research while at the University of Copenhagen but is now employed by Pfizer.
The retrospective study was conducted with the use of linked comprehensive national databases. The work was supported by a research grant from Pfizer.
VANCOUVER, B.C. – The combination of even mild psoriasis and a diagnosis of depression is associated with a greater risk of acute MI than is either condition alone, according to a Danish national study.
Moreover, when depression coincided with a severe case of psoriasis, not only was the risk of incident acute MI elevated, but the risk that the MI would be fatal was significantly increased as well, Dr. Alexander Egeberg reported at the World Congress of Dermatology.

He presented a population-based cohort study of the entire Danish population, for the period of 1997-2011. The study population comprised 5,158,870 Danish citizens, including 50,480 Danes diagnosed with mild psoriasis and 10,974 others diagnosed with severe psoriasis during the study years. During that period, 11,142 members of the group with mild psoriasis and 3,063 with severe psoriasis were diagnosed with incident depression.
In a multivariate Cox regression analysis adjusted for age, gender, comorbid conditions, and concomitant medications, Danes with mild psoriasis but no diagnosis of depression were at a statistically significant 18% increased risk of having an acute MI compared to the general Danish population free of both diseases; the risk climbed upward from there (see chart).
A consistent trend for an increased risk of fatal MI was evident in patients with psoriasis, be it mild or severe, and with or without comorbid depression. However, this risk achieved statistical significance only in the group with severe psoriasis plus depression, a state associated with a 64% increased risk of fatal MI compared with the general population.
Although recent years have brought clear evidence that psoriasis is associated with increased risks of heart disease and depression, and that stand-alone depression is a risk factor for ischemic coronary events, the link between psoriasis, depression, and MI previously was unclear, explained Dr. Egeberg, who performed the research while at the University of Copenhagen but is now employed by Pfizer.
The retrospective study was conducted with the use of linked comprehensive national databases. The work was supported by a research grant from Pfizer.
VANCOUVER, B.C. – The combination of even mild psoriasis and a diagnosis of depression is associated with a greater risk of acute MI than is either condition alone, according to a Danish national study.
Moreover, when depression coincided with a severe case of psoriasis, not only was the risk of incident acute MI elevated, but the risk that the MI would be fatal was significantly increased as well, Dr. Alexander Egeberg reported at the World Congress of Dermatology.

He presented a population-based cohort study of the entire Danish population, for the period of 1997-2011. The study population comprised 5,158,870 Danish citizens, including 50,480 Danes diagnosed with mild psoriasis and 10,974 others diagnosed with severe psoriasis during the study years. During that period, 11,142 members of the group with mild psoriasis and 3,063 with severe psoriasis were diagnosed with incident depression.
In a multivariate Cox regression analysis adjusted for age, gender, comorbid conditions, and concomitant medications, Danes with mild psoriasis but no diagnosis of depression were at a statistically significant 18% increased risk of having an acute MI compared to the general Danish population free of both diseases; the risk climbed upward from there (see chart).
A consistent trend for an increased risk of fatal MI was evident in patients with psoriasis, be it mild or severe, and with or without comorbid depression. However, this risk achieved statistical significance only in the group with severe psoriasis plus depression, a state associated with a 64% increased risk of fatal MI compared with the general population.
Although recent years have brought clear evidence that psoriasis is associated with increased risks of heart disease and depression, and that stand-alone depression is a risk factor for ischemic coronary events, the link between psoriasis, depression, and MI previously was unclear, explained Dr. Egeberg, who performed the research while at the University of Copenhagen but is now employed by Pfizer.
The retrospective study was conducted with the use of linked comprehensive national databases. The work was supported by a research grant from Pfizer.
AT WCD 2015
Key clinical point: In psoriasis patients, comorbid depression is associated with an increased risk of a first acute MI.
Major finding: The incidence rate of acute MI was 2.05 cases per 1,000 person-years in the general Danish population, climbing to 3.14/1,000 in those with mild psoriasis without diagnosed depression, and 5.07/1,000 for mild psoriasis with depression.
Data source: A retrospective, population-based, cohort study of the entire Danish adult population – more than 5.1 million individuals – during 1997-2011.
Disclosures: The presenter is an employee of Pfizer, which supported the study.
Inspiratory muscle training boosts lung function in ankylosing spondylitis
ROME – A program of inspiratory muscle training combined with standard muscular rehabilitation exercise significantly improved lung function in patients with ankylosing spondylitis.
Compared to patients who engaged only in the rehabilitation exercise, the combination program resulted in significantly greater gains in both physical and physiologic measures of pulmonary function, Dr. Razvan Dragoi reported at the annual meeting of the European League Against Rheumatism.
“We assessed resting pulmonary function and ran cardiopulmonary exercise tests at the start and end of the study and saw significant improvements across all measures of lung function in the group undergoing inspiratory muscle training,” said Dr. Dragoi of the Victor Babes University of Medicine and Pharmacy, Romania. “When you compare these findings with the conventional exercise group – which saw small, nonsignificant improvements – it’s clear that adding inspiratory training to an exercise program has clear health benefits for patients.”
The study randomized 54 patients with ankylosing spondylitis to two exercise interventions, Dr. Dragoi said in an interview. “Both groups in our study performed a weekly group session for about 40 minutes per session, managed by a physiotherapist. They were then provided with simple, step-by-step written instructions with illustrations in order to practice these exercises at home,” 5 days each week, for 40 minutes at a time.
The program consisted of 20 exercises: motion and flexibility exercises of the cervical, thoracic, and lumbar spine; stretching of the hamstring muscles, erector spine muscle, and shoulder muscles; abdominal and diaphragm breathing exercises, and chest expansion exercises. The patients were required to achieve a level of perceived exertion of “somewhat hard.”
They also completed an exercise training diary in order to assess their compliance with the recommended program.
The investigational group, however, added another level of training. “In addition to the conventional exercise training, patients performed supervised inspiratory muscle training, three times a week, totaling 24 sessions. This used a real-time computer-assisted device (Trainair, Project Electronics Limited, United Kingdom).”
The training load was based on 80% of the patient’s sustained maximum inspiratory pressure. The patients started by performing six loaded inspiration with a 60-second rest period between each inspiration. This sequence of six exercises continued with 45-, 30-, and 15-, 10- and 5-second rest periods up to 36 loaded inspirations. The training session duration was about 30 minutes.
The study assessed a number of physical and physiologic endpoints, including chest expansion, forced vital capacity and expiratory volumes, and measures of oxygen and carbon dioxide exchange.
Only one outcome – chest expansion – improved significantly in the control arm, increasing from 69 cm at baseline to 72 cm by 8 weeks. In the intensified arm, however, every outcome improved significantly, including chest expansion (66 cm-94 cm), forced vital capacity (78.6%-82.7%), forced expiratory volume (71%-74.6%), and peak oxygen uptake (1.7-2 L/min). The measures of oxygen and carbon dioxide exchange also showed significant improvements.
Dr. Dragoi didn’t follow the patients to assess how long the exercise-related improvements lasted, but like all exercise, he said, the program would have to be repeated to maintain them. “We do have a follow-up in mind, and will be conducting that soon, but we do not know how many patients will be available for the follow-up.”
He had no financial disclosures.
On Twitter @Alz_Gal
ROME – A program of inspiratory muscle training combined with standard muscular rehabilitation exercise significantly improved lung function in patients with ankylosing spondylitis.
Compared to patients who engaged only in the rehabilitation exercise, the combination program resulted in significantly greater gains in both physical and physiologic measures of pulmonary function, Dr. Razvan Dragoi reported at the annual meeting of the European League Against Rheumatism.
“We assessed resting pulmonary function and ran cardiopulmonary exercise tests at the start and end of the study and saw significant improvements across all measures of lung function in the group undergoing inspiratory muscle training,” said Dr. Dragoi of the Victor Babes University of Medicine and Pharmacy, Romania. “When you compare these findings with the conventional exercise group – which saw small, nonsignificant improvements – it’s clear that adding inspiratory training to an exercise program has clear health benefits for patients.”
The study randomized 54 patients with ankylosing spondylitis to two exercise interventions, Dr. Dragoi said in an interview. “Both groups in our study performed a weekly group session for about 40 minutes per session, managed by a physiotherapist. They were then provided with simple, step-by-step written instructions with illustrations in order to practice these exercises at home,” 5 days each week, for 40 minutes at a time.
The program consisted of 20 exercises: motion and flexibility exercises of the cervical, thoracic, and lumbar spine; stretching of the hamstring muscles, erector spine muscle, and shoulder muscles; abdominal and diaphragm breathing exercises, and chest expansion exercises. The patients were required to achieve a level of perceived exertion of “somewhat hard.”
They also completed an exercise training diary in order to assess their compliance with the recommended program.
The investigational group, however, added another level of training. “In addition to the conventional exercise training, patients performed supervised inspiratory muscle training, three times a week, totaling 24 sessions. This used a real-time computer-assisted device (Trainair, Project Electronics Limited, United Kingdom).”
The training load was based on 80% of the patient’s sustained maximum inspiratory pressure. The patients started by performing six loaded inspiration with a 60-second rest period between each inspiration. This sequence of six exercises continued with 45-, 30-, and 15-, 10- and 5-second rest periods up to 36 loaded inspirations. The training session duration was about 30 minutes.
The study assessed a number of physical and physiologic endpoints, including chest expansion, forced vital capacity and expiratory volumes, and measures of oxygen and carbon dioxide exchange.
Only one outcome – chest expansion – improved significantly in the control arm, increasing from 69 cm at baseline to 72 cm by 8 weeks. In the intensified arm, however, every outcome improved significantly, including chest expansion (66 cm-94 cm), forced vital capacity (78.6%-82.7%), forced expiratory volume (71%-74.6%), and peak oxygen uptake (1.7-2 L/min). The measures of oxygen and carbon dioxide exchange also showed significant improvements.
Dr. Dragoi didn’t follow the patients to assess how long the exercise-related improvements lasted, but like all exercise, he said, the program would have to be repeated to maintain them. “We do have a follow-up in mind, and will be conducting that soon, but we do not know how many patients will be available for the follow-up.”
He had no financial disclosures.
On Twitter @Alz_Gal
ROME – A program of inspiratory muscle training combined with standard muscular rehabilitation exercise significantly improved lung function in patients with ankylosing spondylitis.
Compared to patients who engaged only in the rehabilitation exercise, the combination program resulted in significantly greater gains in both physical and physiologic measures of pulmonary function, Dr. Razvan Dragoi reported at the annual meeting of the European League Against Rheumatism.
“We assessed resting pulmonary function and ran cardiopulmonary exercise tests at the start and end of the study and saw significant improvements across all measures of lung function in the group undergoing inspiratory muscle training,” said Dr. Dragoi of the Victor Babes University of Medicine and Pharmacy, Romania. “When you compare these findings with the conventional exercise group – which saw small, nonsignificant improvements – it’s clear that adding inspiratory training to an exercise program has clear health benefits for patients.”
The study randomized 54 patients with ankylosing spondylitis to two exercise interventions, Dr. Dragoi said in an interview. “Both groups in our study performed a weekly group session for about 40 minutes per session, managed by a physiotherapist. They were then provided with simple, step-by-step written instructions with illustrations in order to practice these exercises at home,” 5 days each week, for 40 minutes at a time.
The program consisted of 20 exercises: motion and flexibility exercises of the cervical, thoracic, and lumbar spine; stretching of the hamstring muscles, erector spine muscle, and shoulder muscles; abdominal and diaphragm breathing exercises, and chest expansion exercises. The patients were required to achieve a level of perceived exertion of “somewhat hard.”
They also completed an exercise training diary in order to assess their compliance with the recommended program.
The investigational group, however, added another level of training. “In addition to the conventional exercise training, patients performed supervised inspiratory muscle training, three times a week, totaling 24 sessions. This used a real-time computer-assisted device (Trainair, Project Electronics Limited, United Kingdom).”
The training load was based on 80% of the patient’s sustained maximum inspiratory pressure. The patients started by performing six loaded inspiration with a 60-second rest period between each inspiration. This sequence of six exercises continued with 45-, 30-, and 15-, 10- and 5-second rest periods up to 36 loaded inspirations. The training session duration was about 30 minutes.
The study assessed a number of physical and physiologic endpoints, including chest expansion, forced vital capacity and expiratory volumes, and measures of oxygen and carbon dioxide exchange.
Only one outcome – chest expansion – improved significantly in the control arm, increasing from 69 cm at baseline to 72 cm by 8 weeks. In the intensified arm, however, every outcome improved significantly, including chest expansion (66 cm-94 cm), forced vital capacity (78.6%-82.7%), forced expiratory volume (71%-74.6%), and peak oxygen uptake (1.7-2 L/min). The measures of oxygen and carbon dioxide exchange also showed significant improvements.
Dr. Dragoi didn’t follow the patients to assess how long the exercise-related improvements lasted, but like all exercise, he said, the program would have to be repeated to maintain them. “We do have a follow-up in mind, and will be conducting that soon, but we do not know how many patients will be available for the follow-up.”
He had no financial disclosures.
On Twitter @Alz_Gal
AT EULAR 2015
Key clinical point: Inspiratory muscle training combined with conventional muscle rehabilitation boosted pulmonary function in patients with ankylosing spondylitis.
Major finding: The combination increased chest expansion by almost 30 cm as well as improving physiologic measures of lung function.
Data source: The trail randomized 54 patients to two exercise regimens.
Disclosures: Dr. Dragoi had no financial disclosures.
EULAR: Epigenetics points to anti-TNF efficacy in psoriatic arthritis
ROME – Tumor necrosis factor–alpha (TNF-alpha) inhibitors are well established as a treatment for psoriatic arthritis patients, but efficacy can vary greatly within that population. New work from Canadian researchers demonstrates that the global DNA methylation pattern differs between TNF-alpha–inhibitor responders and secondary failures, which could help explain the variability in medication success rates.
“Although TNF inhibitors [TNFi] work very well in certain individuals with psoriatic arthritis [PsA], up to 40% of individuals receiving this treatment fail to achieve a therapeutic response, and 20%-50% of individuals who have an initial response to treatment become refractory weeks or months after receiving therapy.” lead study author Darren O’Rielly, Ph.D., said at the European Congress of Rheumatology.
The preliminary study findings eventually could lead to the identification of biomarkers that would indicate patients who are not good candidates for TNFi medications, said Dr. O’Rielly of Memorial University, St. John’s, Nfld.
Given recent advances in epigenetics and that the epigenetic signature is affected by environmental factors, the investigators set out to determine if methylation alterations could help explain why PsA patients respond or fail with TNFi. The researchers performed genome-wide DNA methylation profiling on blood samples from 41 PsA patients, using machinery that measures about 480,000 CpG sites per sample and covers 96% of RefSeq genes. A total of 21 patients were considered TNFi responders, of whom 13 were treated with etanercept and 8 with adalimumab; median follow-up duration was 18 months. Twenty patients were considered secondary TNFi failures, of whom 15 were treated with etanercept and 5 with adalimumab; median follow-up duration was 36 months.
Dr. O’Rielly, a senior research scientist at Memorial and director of the Molecular Genetics Laboratory at Eastern Health in St. John’s, and his associates measured the methylation level at CpG sites using a genome-wide approach and selected regions of interest based on functional relevance to TNF-mediated signaling pathways with methylation level differences of 5% or greater and an adjusted P-value less than .05.
After quality-control filtering, investigators evaluated 384,599 CpG sites for TNFi responders and 368,863 CpG sites for TNFi failures. Researchers found 72 CpG sites of interest in the TNFi responder group and 91 CpG sites of interest in the TNFi failure group. Top candidate genes for TNFi responders included TRAPPC9 (which functions as an activator of NF-kB), CCR6 (which regulates the migration and recruitment of dendritic and T cells), and PSORS1C3 (psoriasis susceptibility 1 candidate 3), while top candidate genes for TNFi secondary failures included CD70 (an encoded protein that is a ligand for TNFRSF27/CD27) and TNFRSF1B (a member of the TNF receptor ‘superfamily’ that mediates most of the metabolic effects of TNF-alpha).
“We are very encouraged by these findings,” Dr. O’Rielly said. “We were a little surprised that several of our best candidate genes, such as TNFRSF1B and CD70, appear to play a role in TNF-alpha signaling, and that their methylation change occurs in a gene region that is consistent with a possible functional effect. We were expecting to find some methylation changes in genes but not necessarily in pathways with a direct connection with TNF-alpha–mediated signaling.”
The group plans to confirm these findings for the best candidate genes using other methylation-specific polymerase chain reaction technology, he said, and will investigate additional CpG sites adjacent to the region of interest, including promoters and enhancers, in the best candidate genes to better characterize the full extent of methylation changes in these regions. They also would like to replicate the findings in a prospective, independent cohort.
Dr. O’Rielly reported no relevant financial conflicts.
ROME – Tumor necrosis factor–alpha (TNF-alpha) inhibitors are well established as a treatment for psoriatic arthritis patients, but efficacy can vary greatly within that population. New work from Canadian researchers demonstrates that the global DNA methylation pattern differs between TNF-alpha–inhibitor responders and secondary failures, which could help explain the variability in medication success rates.
“Although TNF inhibitors [TNFi] work very well in certain individuals with psoriatic arthritis [PsA], up to 40% of individuals receiving this treatment fail to achieve a therapeutic response, and 20%-50% of individuals who have an initial response to treatment become refractory weeks or months after receiving therapy.” lead study author Darren O’Rielly, Ph.D., said at the European Congress of Rheumatology.
The preliminary study findings eventually could lead to the identification of biomarkers that would indicate patients who are not good candidates for TNFi medications, said Dr. O’Rielly of Memorial University, St. John’s, Nfld.
Given recent advances in epigenetics and that the epigenetic signature is affected by environmental factors, the investigators set out to determine if methylation alterations could help explain why PsA patients respond or fail with TNFi. The researchers performed genome-wide DNA methylation profiling on blood samples from 41 PsA patients, using machinery that measures about 480,000 CpG sites per sample and covers 96% of RefSeq genes. A total of 21 patients were considered TNFi responders, of whom 13 were treated with etanercept and 8 with adalimumab; median follow-up duration was 18 months. Twenty patients were considered secondary TNFi failures, of whom 15 were treated with etanercept and 5 with adalimumab; median follow-up duration was 36 months.
Dr. O’Rielly, a senior research scientist at Memorial and director of the Molecular Genetics Laboratory at Eastern Health in St. John’s, and his associates measured the methylation level at CpG sites using a genome-wide approach and selected regions of interest based on functional relevance to TNF-mediated signaling pathways with methylation level differences of 5% or greater and an adjusted P-value less than .05.
After quality-control filtering, investigators evaluated 384,599 CpG sites for TNFi responders and 368,863 CpG sites for TNFi failures. Researchers found 72 CpG sites of interest in the TNFi responder group and 91 CpG sites of interest in the TNFi failure group. Top candidate genes for TNFi responders included TRAPPC9 (which functions as an activator of NF-kB), CCR6 (which regulates the migration and recruitment of dendritic and T cells), and PSORS1C3 (psoriasis susceptibility 1 candidate 3), while top candidate genes for TNFi secondary failures included CD70 (an encoded protein that is a ligand for TNFRSF27/CD27) and TNFRSF1B (a member of the TNF receptor ‘superfamily’ that mediates most of the metabolic effects of TNF-alpha).
“We are very encouraged by these findings,” Dr. O’Rielly said. “We were a little surprised that several of our best candidate genes, such as TNFRSF1B and CD70, appear to play a role in TNF-alpha signaling, and that their methylation change occurs in a gene region that is consistent with a possible functional effect. We were expecting to find some methylation changes in genes but not necessarily in pathways with a direct connection with TNF-alpha–mediated signaling.”
The group plans to confirm these findings for the best candidate genes using other methylation-specific polymerase chain reaction technology, he said, and will investigate additional CpG sites adjacent to the region of interest, including promoters and enhancers, in the best candidate genes to better characterize the full extent of methylation changes in these regions. They also would like to replicate the findings in a prospective, independent cohort.
Dr. O’Rielly reported no relevant financial conflicts.
ROME – Tumor necrosis factor–alpha (TNF-alpha) inhibitors are well established as a treatment for psoriatic arthritis patients, but efficacy can vary greatly within that population. New work from Canadian researchers demonstrates that the global DNA methylation pattern differs between TNF-alpha–inhibitor responders and secondary failures, which could help explain the variability in medication success rates.
“Although TNF inhibitors [TNFi] work very well in certain individuals with psoriatic arthritis [PsA], up to 40% of individuals receiving this treatment fail to achieve a therapeutic response, and 20%-50% of individuals who have an initial response to treatment become refractory weeks or months after receiving therapy.” lead study author Darren O’Rielly, Ph.D., said at the European Congress of Rheumatology.
The preliminary study findings eventually could lead to the identification of biomarkers that would indicate patients who are not good candidates for TNFi medications, said Dr. O’Rielly of Memorial University, St. John’s, Nfld.
Given recent advances in epigenetics and that the epigenetic signature is affected by environmental factors, the investigators set out to determine if methylation alterations could help explain why PsA patients respond or fail with TNFi. The researchers performed genome-wide DNA methylation profiling on blood samples from 41 PsA patients, using machinery that measures about 480,000 CpG sites per sample and covers 96% of RefSeq genes. A total of 21 patients were considered TNFi responders, of whom 13 were treated with etanercept and 8 with adalimumab; median follow-up duration was 18 months. Twenty patients were considered secondary TNFi failures, of whom 15 were treated with etanercept and 5 with adalimumab; median follow-up duration was 36 months.
Dr. O’Rielly, a senior research scientist at Memorial and director of the Molecular Genetics Laboratory at Eastern Health in St. John’s, and his associates measured the methylation level at CpG sites using a genome-wide approach and selected regions of interest based on functional relevance to TNF-mediated signaling pathways with methylation level differences of 5% or greater and an adjusted P-value less than .05.
After quality-control filtering, investigators evaluated 384,599 CpG sites for TNFi responders and 368,863 CpG sites for TNFi failures. Researchers found 72 CpG sites of interest in the TNFi responder group and 91 CpG sites of interest in the TNFi failure group. Top candidate genes for TNFi responders included TRAPPC9 (which functions as an activator of NF-kB), CCR6 (which regulates the migration and recruitment of dendritic and T cells), and PSORS1C3 (psoriasis susceptibility 1 candidate 3), while top candidate genes for TNFi secondary failures included CD70 (an encoded protein that is a ligand for TNFRSF27/CD27) and TNFRSF1B (a member of the TNF receptor ‘superfamily’ that mediates most of the metabolic effects of TNF-alpha).
“We are very encouraged by these findings,” Dr. O’Rielly said. “We were a little surprised that several of our best candidate genes, such as TNFRSF1B and CD70, appear to play a role in TNF-alpha signaling, and that their methylation change occurs in a gene region that is consistent with a possible functional effect. We were expecting to find some methylation changes in genes but not necessarily in pathways with a direct connection with TNF-alpha–mediated signaling.”
The group plans to confirm these findings for the best candidate genes using other methylation-specific polymerase chain reaction technology, he said, and will investigate additional CpG sites adjacent to the region of interest, including promoters and enhancers, in the best candidate genes to better characterize the full extent of methylation changes in these regions. They also would like to replicate the findings in a prospective, independent cohort.
Dr. O’Rielly reported no relevant financial conflicts.
AT THE EULAR 2015 CONGRESS
Key clinical point:If preliminary findings are confirmed, methylation level profiling could lead to the identification of patients who are not good candidates for TNFi medications.
Major finding: Nonresponders to TNF inhibitors had significant methylation pattern differences in two key genes that appear to play a role in TNF-alpha signaling, TNFRSF1B and CD70.
Data source: A study of psoriatic arthritis patients comparing 21 TNFi responders and 20 nonresponders.
Disclosures: Dr. O’Rielly reported no relevant financial conflicts.
VIDEO: Predicting anti-TNF failure in psoriatic arthritis
ROME – Changes in the methylation status of particular genes in psoriatic arthritis patients might provide the ability to predict failure to respond to tumor necrosis factor-alpha inhibitors, according to preliminary research in 41 psoriatic arthritis patients.
Two genes stood out to the researchers from Memorial University of Newfoundland, St. John’s: TNFRSF1B and CD70. Patients who had methylation changes to those genes were more likely to have secondary failure of TNF inhibitors, said Dr. Proton Rahman, professor of internal medicine at the university and coinvestigator on the study.
It will be necessary to conduct validation studies of the results in larger numbers of patients, as well as functional studies of the effects of methylation changes on the expression of those genes and their proteins, he said in a video interview at the European Congress of Rheumatology.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ROME – Changes in the methylation status of particular genes in psoriatic arthritis patients might provide the ability to predict failure to respond to tumor necrosis factor-alpha inhibitors, according to preliminary research in 41 psoriatic arthritis patients.
Two genes stood out to the researchers from Memorial University of Newfoundland, St. John’s: TNFRSF1B and CD70. Patients who had methylation changes to those genes were more likely to have secondary failure of TNF inhibitors, said Dr. Proton Rahman, professor of internal medicine at the university and coinvestigator on the study.
It will be necessary to conduct validation studies of the results in larger numbers of patients, as well as functional studies of the effects of methylation changes on the expression of those genes and their proteins, he said in a video interview at the European Congress of Rheumatology.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ROME – Changes in the methylation status of particular genes in psoriatic arthritis patients might provide the ability to predict failure to respond to tumor necrosis factor-alpha inhibitors, according to preliminary research in 41 psoriatic arthritis patients.
Two genes stood out to the researchers from Memorial University of Newfoundland, St. John’s: TNFRSF1B and CD70. Patients who had methylation changes to those genes were more likely to have secondary failure of TNF inhibitors, said Dr. Proton Rahman, professor of internal medicine at the university and coinvestigator on the study.
It will be necessary to conduct validation studies of the results in larger numbers of patients, as well as functional studies of the effects of methylation changes on the expression of those genes and their proteins, he said in a video interview at the European Congress of Rheumatology.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE EULAR 2015 CONGRESS
NSAID dosing frequency matters little in ankylosing spondylitis
ROME – Radiographic progression occurs at similar rates in the spines of ankylosing spondylitis patients treated over 2 years with either continuous or on-demand diclofenac, according to results from a randomized, prospective multicenter trial presented at the European Congress of Rheumatology.
In the ENRADAS (Effects of NSAIDs on Radiographic Damage in Ankylosing Spondylitis) trial, Dr. Joachim Sieper of Charite-Universitätsmedizin Berlin and his colleagues compared continuous treatment with at least 50% of the maximum 150-mg daily dose of the NSAID diclofenac and on-demand treatment with diclofenac. During the entire 2 years of the study, no patient received treatment with a tumor necrosis factor blocker or any other drug other than diclofenac.
The investigators measured radiographic spine progression using the modified Stoke Ankylosing Spondylitis Spinal Score(mSASSS). At baseline, patients randomized to continuous treatment who had complete radiographic follow-up had a mean mSASSS of 10.9, while those randomized to the on-demand arm had a mean mSASSS of 16.4. After 2 years on treatment, the average change in mSASSS from baseline was 1.28 for the 62 patients in the continuous-treatment group and 0.79 for the 60 patients in the on-demand group, a difference that was not statistically significant, Dr. Sieper said.
There also was no statistically significant between-group difference when the analysis focused on the subgroup of patients who were C-reactive protein positive at baseline, 55% and 58% of patients in the groups, respectively, who had their average mSASSS increase by 1.68, compared with 0.96. Similarly, when the analysis focused only on patients who had syndesmophytes at baseline, 53% and 62% of patients, respectively, the average increases in mSASSS were 2.11 and 0.95, a difference that was not statistically significant. Presence of C-reactive protein and syndesmophytes are both known risk factors for radiographic progression.
Patient characteristics were similar in the two groups. NSAID intake over the 2-year study period, measured using a 0-100 composite score based on treatment duration and NSAID doses and intervals, was a mean of 76 vs. 44 for the continuous and on-demand groups, respectively. At the study’s end, 77% of patients remained on diclofenac and had not switched to another NSAID.
Side effects were similar in both groups, with 19 serious adverse events in the continuous-treatment patients and 21 serious adverse events in the on-demand patients.
Previous studies have suggested that NSAIDs given continuously over 2 years reduce radiographic progression, compared with on-demand therapy, in ankylosing spondylitis patients. Similar effects were seen in a prospective cohort.
“In our study, continuous vs. on-demand treatment … did not prevent radiographic progression in [ankylosing spondylitis]. It is highly unlikely that the results would have been different with a higher number of patients, because we found a trend for less progression in the on-demand group,” Dr. Sieper said.
Additional study is needed to determine whether an NSAID other than diclofenac, specifically a COX-2 selective drug, would have a different effect on radiographic progression, he said.
Dr. Sieper reported receiving honorarium for consultancies, speaker’s bureaus, or grants from AbbVie, Janssen, Merck, Lily, Novartis, Pfizer, and UCB.
ROME – Radiographic progression occurs at similar rates in the spines of ankylosing spondylitis patients treated over 2 years with either continuous or on-demand diclofenac, according to results from a randomized, prospective multicenter trial presented at the European Congress of Rheumatology.
In the ENRADAS (Effects of NSAIDs on Radiographic Damage in Ankylosing Spondylitis) trial, Dr. Joachim Sieper of Charite-Universitätsmedizin Berlin and his colleagues compared continuous treatment with at least 50% of the maximum 150-mg daily dose of the NSAID diclofenac and on-demand treatment with diclofenac. During the entire 2 years of the study, no patient received treatment with a tumor necrosis factor blocker or any other drug other than diclofenac.
The investigators measured radiographic spine progression using the modified Stoke Ankylosing Spondylitis Spinal Score(mSASSS). At baseline, patients randomized to continuous treatment who had complete radiographic follow-up had a mean mSASSS of 10.9, while those randomized to the on-demand arm had a mean mSASSS of 16.4. After 2 years on treatment, the average change in mSASSS from baseline was 1.28 for the 62 patients in the continuous-treatment group and 0.79 for the 60 patients in the on-demand group, a difference that was not statistically significant, Dr. Sieper said.
There also was no statistically significant between-group difference when the analysis focused on the subgroup of patients who were C-reactive protein positive at baseline, 55% and 58% of patients in the groups, respectively, who had their average mSASSS increase by 1.68, compared with 0.96. Similarly, when the analysis focused only on patients who had syndesmophytes at baseline, 53% and 62% of patients, respectively, the average increases in mSASSS were 2.11 and 0.95, a difference that was not statistically significant. Presence of C-reactive protein and syndesmophytes are both known risk factors for radiographic progression.
Patient characteristics were similar in the two groups. NSAID intake over the 2-year study period, measured using a 0-100 composite score based on treatment duration and NSAID doses and intervals, was a mean of 76 vs. 44 for the continuous and on-demand groups, respectively. At the study’s end, 77% of patients remained on diclofenac and had not switched to another NSAID.
Side effects were similar in both groups, with 19 serious adverse events in the continuous-treatment patients and 21 serious adverse events in the on-demand patients.
Previous studies have suggested that NSAIDs given continuously over 2 years reduce radiographic progression, compared with on-demand therapy, in ankylosing spondylitis patients. Similar effects were seen in a prospective cohort.
“In our study, continuous vs. on-demand treatment … did not prevent radiographic progression in [ankylosing spondylitis]. It is highly unlikely that the results would have been different with a higher number of patients, because we found a trend for less progression in the on-demand group,” Dr. Sieper said.
Additional study is needed to determine whether an NSAID other than diclofenac, specifically a COX-2 selective drug, would have a different effect on radiographic progression, he said.
Dr. Sieper reported receiving honorarium for consultancies, speaker’s bureaus, or grants from AbbVie, Janssen, Merck, Lily, Novartis, Pfizer, and UCB.
ROME – Radiographic progression occurs at similar rates in the spines of ankylosing spondylitis patients treated over 2 years with either continuous or on-demand diclofenac, according to results from a randomized, prospective multicenter trial presented at the European Congress of Rheumatology.
In the ENRADAS (Effects of NSAIDs on Radiographic Damage in Ankylosing Spondylitis) trial, Dr. Joachim Sieper of Charite-Universitätsmedizin Berlin and his colleagues compared continuous treatment with at least 50% of the maximum 150-mg daily dose of the NSAID diclofenac and on-demand treatment with diclofenac. During the entire 2 years of the study, no patient received treatment with a tumor necrosis factor blocker or any other drug other than diclofenac.
The investigators measured radiographic spine progression using the modified Stoke Ankylosing Spondylitis Spinal Score(mSASSS). At baseline, patients randomized to continuous treatment who had complete radiographic follow-up had a mean mSASSS of 10.9, while those randomized to the on-demand arm had a mean mSASSS of 16.4. After 2 years on treatment, the average change in mSASSS from baseline was 1.28 for the 62 patients in the continuous-treatment group and 0.79 for the 60 patients in the on-demand group, a difference that was not statistically significant, Dr. Sieper said.
There also was no statistically significant between-group difference when the analysis focused on the subgroup of patients who were C-reactive protein positive at baseline, 55% and 58% of patients in the groups, respectively, who had their average mSASSS increase by 1.68, compared with 0.96. Similarly, when the analysis focused only on patients who had syndesmophytes at baseline, 53% and 62% of patients, respectively, the average increases in mSASSS were 2.11 and 0.95, a difference that was not statistically significant. Presence of C-reactive protein and syndesmophytes are both known risk factors for radiographic progression.
Patient characteristics were similar in the two groups. NSAID intake over the 2-year study period, measured using a 0-100 composite score based on treatment duration and NSAID doses and intervals, was a mean of 76 vs. 44 for the continuous and on-demand groups, respectively. At the study’s end, 77% of patients remained on diclofenac and had not switched to another NSAID.
Side effects were similar in both groups, with 19 serious adverse events in the continuous-treatment patients and 21 serious adverse events in the on-demand patients.
Previous studies have suggested that NSAIDs given continuously over 2 years reduce radiographic progression, compared with on-demand therapy, in ankylosing spondylitis patients. Similar effects were seen in a prospective cohort.
“In our study, continuous vs. on-demand treatment … did not prevent radiographic progression in [ankylosing spondylitis]. It is highly unlikely that the results would have been different with a higher number of patients, because we found a trend for less progression in the on-demand group,” Dr. Sieper said.
Additional study is needed to determine whether an NSAID other than diclofenac, specifically a COX-2 selective drug, would have a different effect on radiographic progression, he said.
Dr. Sieper reported receiving honorarium for consultancies, speaker’s bureaus, or grants from AbbVie, Janssen, Merck, Lily, Novartis, Pfizer, and UCB.
AT THE EULAR 2015 CONGRESS
Key clinical point: There are no differences with respect to radiographic progression in the spines of ankylosing spondylitis patients treated over 2 years with either continuous or on-demand diclofenac.
Major finding: The average 2-year change in mSASSS from baseline was 1.28 for the 62 patients in the continuous-treatment group and 0.79 for the 60 patients in the on-demand group, a difference that was not statistically significant.
Data source: A randomized, prospective multicenter trial of 122 patients with ankylosing spondylitis.
Disclosures: Dr. Sieper reported receiving honorarium for consultancies, speaker’s bureaus, or grants from AbbVie, Janssen, Merck, Lily, Novartis, Pfizer, and UCB.
WCD: Ustekinumab succeeds as switch agent in psoriasis
VANCOUVER, B.C. – When adults with moderate to severe psoriasis fail one anti-TNF agent, most will still respond to another anti-TNF or ustekinumab, according to data from an investigation at two University of Toronto hospitals.
Researchers reviewed the outcomes of 155 patients who were switched to a second biologic after failing at least 12 weeks of treatment with their first, which was usually an anti–tumor necrosis factor (TNF) agent. Ustekinumab was often plan B. Of the 65 patients switched to it after failing initial anti-TNF therapy, 47 (72%) had a significant response, defined as at least a 75% improvement in the Psoriasis Area & Severity Index score (PASI 75) after 12 weeks of treatment. Ten more patients (15%) achieved PASI 50.
Meanwhile, of the 82 switched to a second anti-TNF, 48 (59%) reached PASI 75 and more achieved PASI 50 after 12 weeks.
“Clinicians switch biologics all the time, but there are no clear guidelines” on how to do it, “so it was worth looking into,” investigator Whan Kim, a medical student at McMaster University in Hamilton, Ont., said at the World Congress of Dermatology.
“This is one of the largest studies to date that determines the effectiveness of real-life switching of biologic therapies in patients with moderate to severe psoriasis. This study suggests switching to ustekinumab would induce the highest response rate – 72% was surprising,” he said. “Failing a biologic doesn’t preclude responding to another one. Don’t give up,” Mr. Kim added.
The team did find, however, that patients with two previously failed biologics were about half as likely to respond to a third biologic, compared with those who had failed just one (odds ratio, 0.474; 95% confidence interval, 0.23-0.96; P = 0.04).
The patients averaged 50 years of age and had psoriasis for approximately 17 years. Half had psoriatic arthritis, and the study population included slightly more women than men. Of the 155 patients, 93 failed initial treatment with etanercept. Seven of 12 (58%) who switched to infliximab, 24 of 43 (56%) who switched to adalimumab, and 27 of 38 (71%) who switched to ustekinumab achieved PASI 75.
Twenty-three failed initial infliximab. Three of four (75%) who switched to etanercept, four of seven (57%) who switched to adalimumab, and nine of 12 (75%) who switched to ustekinumab had a significant response.
Thirty-one patients failed initial adalimumab. Seven of 11 (64%) who switched to etanercept, 3 of 5 (60%) who switched to infliximab, and 11 of 15 (73%) who switched to ustekinumab had a significant response.
Of eight patients who failed initial ustekinumab, three (38%) switched to etanercept and had a significant response after 12 weeks.
There was no outside funding for the work, and Mr. Kim had no disclosures. The senior investigator is a speaker, consultant, and investigator for AbbVie, Amgen, and Janssen.
VANCOUVER, B.C. – When adults with moderate to severe psoriasis fail one anti-TNF agent, most will still respond to another anti-TNF or ustekinumab, according to data from an investigation at two University of Toronto hospitals.
Researchers reviewed the outcomes of 155 patients who were switched to a second biologic after failing at least 12 weeks of treatment with their first, which was usually an anti–tumor necrosis factor (TNF) agent. Ustekinumab was often plan B. Of the 65 patients switched to it after failing initial anti-TNF therapy, 47 (72%) had a significant response, defined as at least a 75% improvement in the Psoriasis Area & Severity Index score (PASI 75) after 12 weeks of treatment. Ten more patients (15%) achieved PASI 50.
Meanwhile, of the 82 switched to a second anti-TNF, 48 (59%) reached PASI 75 and more achieved PASI 50 after 12 weeks.
“Clinicians switch biologics all the time, but there are no clear guidelines” on how to do it, “so it was worth looking into,” investigator Whan Kim, a medical student at McMaster University in Hamilton, Ont., said at the World Congress of Dermatology.
“This is one of the largest studies to date that determines the effectiveness of real-life switching of biologic therapies in patients with moderate to severe psoriasis. This study suggests switching to ustekinumab would induce the highest response rate – 72% was surprising,” he said. “Failing a biologic doesn’t preclude responding to another one. Don’t give up,” Mr. Kim added.
The team did find, however, that patients with two previously failed biologics were about half as likely to respond to a third biologic, compared with those who had failed just one (odds ratio, 0.474; 95% confidence interval, 0.23-0.96; P = 0.04).
The patients averaged 50 years of age and had psoriasis for approximately 17 years. Half had psoriatic arthritis, and the study population included slightly more women than men. Of the 155 patients, 93 failed initial treatment with etanercept. Seven of 12 (58%) who switched to infliximab, 24 of 43 (56%) who switched to adalimumab, and 27 of 38 (71%) who switched to ustekinumab achieved PASI 75.
Twenty-three failed initial infliximab. Three of four (75%) who switched to etanercept, four of seven (57%) who switched to adalimumab, and nine of 12 (75%) who switched to ustekinumab had a significant response.
Thirty-one patients failed initial adalimumab. Seven of 11 (64%) who switched to etanercept, 3 of 5 (60%) who switched to infliximab, and 11 of 15 (73%) who switched to ustekinumab had a significant response.
Of eight patients who failed initial ustekinumab, three (38%) switched to etanercept and had a significant response after 12 weeks.
There was no outside funding for the work, and Mr. Kim had no disclosures. The senior investigator is a speaker, consultant, and investigator for AbbVie, Amgen, and Janssen.
VANCOUVER, B.C. – When adults with moderate to severe psoriasis fail one anti-TNF agent, most will still respond to another anti-TNF or ustekinumab, according to data from an investigation at two University of Toronto hospitals.
Researchers reviewed the outcomes of 155 patients who were switched to a second biologic after failing at least 12 weeks of treatment with their first, which was usually an anti–tumor necrosis factor (TNF) agent. Ustekinumab was often plan B. Of the 65 patients switched to it after failing initial anti-TNF therapy, 47 (72%) had a significant response, defined as at least a 75% improvement in the Psoriasis Area & Severity Index score (PASI 75) after 12 weeks of treatment. Ten more patients (15%) achieved PASI 50.
Meanwhile, of the 82 switched to a second anti-TNF, 48 (59%) reached PASI 75 and more achieved PASI 50 after 12 weeks.
“Clinicians switch biologics all the time, but there are no clear guidelines” on how to do it, “so it was worth looking into,” investigator Whan Kim, a medical student at McMaster University in Hamilton, Ont., said at the World Congress of Dermatology.
“This is one of the largest studies to date that determines the effectiveness of real-life switching of biologic therapies in patients with moderate to severe psoriasis. This study suggests switching to ustekinumab would induce the highest response rate – 72% was surprising,” he said. “Failing a biologic doesn’t preclude responding to another one. Don’t give up,” Mr. Kim added.
The team did find, however, that patients with two previously failed biologics were about half as likely to respond to a third biologic, compared with those who had failed just one (odds ratio, 0.474; 95% confidence interval, 0.23-0.96; P = 0.04).
The patients averaged 50 years of age and had psoriasis for approximately 17 years. Half had psoriatic arthritis, and the study population included slightly more women than men. Of the 155 patients, 93 failed initial treatment with etanercept. Seven of 12 (58%) who switched to infliximab, 24 of 43 (56%) who switched to adalimumab, and 27 of 38 (71%) who switched to ustekinumab achieved PASI 75.
Twenty-three failed initial infliximab. Three of four (75%) who switched to etanercept, four of seven (57%) who switched to adalimumab, and nine of 12 (75%) who switched to ustekinumab had a significant response.
Thirty-one patients failed initial adalimumab. Seven of 11 (64%) who switched to etanercept, 3 of 5 (60%) who switched to infliximab, and 11 of 15 (73%) who switched to ustekinumab had a significant response.
Of eight patients who failed initial ustekinumab, three (38%) switched to etanercept and had a significant response after 12 weeks.
There was no outside funding for the work, and Mr. Kim had no disclosures. The senior investigator is a speaker, consultant, and investigator for AbbVie, Amgen, and Janssen.
AT WCD 2015
Key clinical point: Consider ustekinumab if psoriasis patients fail their first anti-TNF agent.
Major finding: Of 65 patients switched to ustekinumab after failing initial anti-TNF therapy, 47 (72%) achieved PASI 75 within 12 weeks.
Data source: Chart review of 155 psoriasis patients.
Disclosures: There was no outside funding for the work. The senior investigator is a speaker, consultant, and investigator for Abbvie, Amgen, and Janssen.
VIDEO: EULAR updates cardiovascular-disease risk recommendations
ROME – The European League Against Rheumatism introduced an update to its 2009 recommendations on assessing and managing cardiovascular-disease risk in patients with rheumatoid arthritis, psoriatic arthritis, or ankylosing spondylitis.
The update features an expanded evidence base for the recommendations, especially for psoriatic arthritis and ankylosing spondylitis, Dr. Michael T. Nurmohamed said in an interview at the European Congress of Rheumatology. One new element in the revision included a scaling down of the previously suggested annual assessment to a more flexible approach to the timing of serial assessments based on the risk level of individual patients. Another addition is the possible use of carotid ultrasound to measure atherosclerotic burden as a complement to more routinely-measured risk factors such as blood pressure and serum lipids, said Dr. Nurmohamed, convener of the current task force as well as the panel that formulated the first version (Ann. Rheum. Dis. 2010;69:325-31).
A second EULAR task force recently developed new recommendations on assessing and managing other comorbidities in patients with rheumatologic diseases, such as osteoporosis, cancer, peptic ulcers, and renal dysfunction. Chronic kidney disease is an important modifier of cardiovascular-disease risk, and hence the new comorbidity recommendations complement the new cardiovascular-disease statement, said Dr. Nurmohamed, professor and head of the rheumatology research department at VU University Medical Center in Amsterdam.
Dr. Nurmohamed had no disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
ROME – The European League Against Rheumatism introduced an update to its 2009 recommendations on assessing and managing cardiovascular-disease risk in patients with rheumatoid arthritis, psoriatic arthritis, or ankylosing spondylitis.
The update features an expanded evidence base for the recommendations, especially for psoriatic arthritis and ankylosing spondylitis, Dr. Michael T. Nurmohamed said in an interview at the European Congress of Rheumatology. One new element in the revision included a scaling down of the previously suggested annual assessment to a more flexible approach to the timing of serial assessments based on the risk level of individual patients. Another addition is the possible use of carotid ultrasound to measure atherosclerotic burden as a complement to more routinely-measured risk factors such as blood pressure and serum lipids, said Dr. Nurmohamed, convener of the current task force as well as the panel that formulated the first version (Ann. Rheum. Dis. 2010;69:325-31).
A second EULAR task force recently developed new recommendations on assessing and managing other comorbidities in patients with rheumatologic diseases, such as osteoporosis, cancer, peptic ulcers, and renal dysfunction. Chronic kidney disease is an important modifier of cardiovascular-disease risk, and hence the new comorbidity recommendations complement the new cardiovascular-disease statement, said Dr. Nurmohamed, professor and head of the rheumatology research department at VU University Medical Center in Amsterdam.
Dr. Nurmohamed had no disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
ROME – The European League Against Rheumatism introduced an update to its 2009 recommendations on assessing and managing cardiovascular-disease risk in patients with rheumatoid arthritis, psoriatic arthritis, or ankylosing spondylitis.
The update features an expanded evidence base for the recommendations, especially for psoriatic arthritis and ankylosing spondylitis, Dr. Michael T. Nurmohamed said in an interview at the European Congress of Rheumatology. One new element in the revision included a scaling down of the previously suggested annual assessment to a more flexible approach to the timing of serial assessments based on the risk level of individual patients. Another addition is the possible use of carotid ultrasound to measure atherosclerotic burden as a complement to more routinely-measured risk factors such as blood pressure and serum lipids, said Dr. Nurmohamed, convener of the current task force as well as the panel that formulated the first version (Ann. Rheum. Dis. 2010;69:325-31).
A second EULAR task force recently developed new recommendations on assessing and managing other comorbidities in patients with rheumatologic diseases, such as osteoporosis, cancer, peptic ulcers, and renal dysfunction. Chronic kidney disease is an important modifier of cardiovascular-disease risk, and hence the new comorbidity recommendations complement the new cardiovascular-disease statement, said Dr. Nurmohamed, professor and head of the rheumatology research department at VU University Medical Center in Amsterdam.
Dr. Nurmohamed had no disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
AT THE EULAR 2015 CONGRESS
WCD: Tofacitinib’s benefits for psoriasis persist for 2 years
VANCOUVER – Oral tofacitinib for adults with moderate to severe plaque psoriasis demonstrated sustained efficacy and a “generally manageable” safety profile throughout 2 years of follow-up in a large, multicenter ongoing long-term extension study, Dr. Matthias Augustin reported at the World Congress of Dermatology.
One month after enrolling in the phase III, open-label trial (known as OPT Extend), and going on the investigational oral Janus kinase inhibitor at 10 mg b.i.d., 56.2% of the 2,847 participants showed a PASI 75 response – that is, at least a 75% reduction from their baseline Psoriasis Area and Severity Index score. After 2 years in the OPT Extend study, 64.5% of the 1,912 patients remaining in the study were PASI 75 responders.
Similarly, 56.3% of patients went from moderate or severe disease at baseline to “clear” or “almost clear” by Physician Global Assessment (PGA) at 1 month, and 53.9% of participants had a PGA score of 0 or 1 at 24 months, added Dr. Augustin, professor of dermatology at the University of Hamburg-Eppendorf and director of the German Center for Health Services Research in Dermatology and Nursing.
Psoriasis patients who had participated in any of four earlier phase III or two phase II randomized, controlled trials of tofacitinib were eligible to participate in OPT Extend. Participants were placed on tofacitinib at 10 mg b.i.d. for the first 2 months of the open-label study; then investigators adjusted their dose individually – either 5 or 10 mg b.i.d. – at each follow-up visit based on efficacy and side effects. For purposes of OPT Extend, patients’ baseline PASI and PGA scores were defined as the values recorded on the day they were randomized in the earlier round of trials.
Through 2 years of follow-up in this interim analysis of OPT Extend, less than 10% of subjects discontinued tofacitinib because of adverse events. The adverse events were the same as the ones that arose in the earlier, briefer randomized trials, the longest of which lasted 1 year. No signs of cumulative organ toxicity were evident during the additional follow-up.
Slightly more than 4% of all adverse events were categorized as severe, while 62% were mild. The most frequent adverse events were nasopharyngitis in 16% of patients, increased creatinine phosphokinase in 10%, and upper respiratory tract infections in 7%. Serious infections requiring systemic antibiotics or hospitalization occurred in 1.8% of patients on tofacitinib for 2 years, the most common of which were pneumonia, herpes zoster, and urinary tract infection. Herpes zoster occurred in 3.5% of participants, with most cases being mild or moderate. Malignancies other than nonmelanoma skin cancers occurred in 1.2% of subjects.
In terms of laboratory findings of interest, patients’ LDL/HDL ratio remained stable throughout 24 months on tofacitinib. Nor were there any clinically meaningful changes in average levels of other laboratory parameters, including hemoglobin, creatinine phosphokinase, lymphocytes, and neutrophils. A lymphocyte count below 500/mm3 occurred in 0.4% of patients at some point; however, it was unrelated to duration of exposure to tofacitinib and wasn’t linked to an increased infection rate.
Dr. Augustin said an unmet need exists for effective and nontoxic oral medications for moderate to severe psoriasis, because many patients would prefer not to take an injectable biologic. Tofacitinib is aimed at filling that need, and serving as an easier, less toxic initial therapy. However, it is essential that an optimal oral agent have a favorable safety profile because psoriasis is a lifelong disease with an average lifetime duration in excess of 40 years in adults and more than 60 years in affected children, he said.
Pfizer, which is developing the drug, has filed for marketing approval for tofacitinib for treatment of adults with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy or phototherapy. The Food and Drug Administration has announced its intent to issue a ruling on the application in October 2015.
The OPT Expect study is funded by Pfizer. Dr. Augustin is an adviser to and/or a recipient of research grants from Pfizer and more than a dozen other medical companies.
VANCOUVER – Oral tofacitinib for adults with moderate to severe plaque psoriasis demonstrated sustained efficacy and a “generally manageable” safety profile throughout 2 years of follow-up in a large, multicenter ongoing long-term extension study, Dr. Matthias Augustin reported at the World Congress of Dermatology.
One month after enrolling in the phase III, open-label trial (known as OPT Extend), and going on the investigational oral Janus kinase inhibitor at 10 mg b.i.d., 56.2% of the 2,847 participants showed a PASI 75 response – that is, at least a 75% reduction from their baseline Psoriasis Area and Severity Index score. After 2 years in the OPT Extend study, 64.5% of the 1,912 patients remaining in the study were PASI 75 responders.
Similarly, 56.3% of patients went from moderate or severe disease at baseline to “clear” or “almost clear” by Physician Global Assessment (PGA) at 1 month, and 53.9% of participants had a PGA score of 0 or 1 at 24 months, added Dr. Augustin, professor of dermatology at the University of Hamburg-Eppendorf and director of the German Center for Health Services Research in Dermatology and Nursing.
Psoriasis patients who had participated in any of four earlier phase III or two phase II randomized, controlled trials of tofacitinib were eligible to participate in OPT Extend. Participants were placed on tofacitinib at 10 mg b.i.d. for the first 2 months of the open-label study; then investigators adjusted their dose individually – either 5 or 10 mg b.i.d. – at each follow-up visit based on efficacy and side effects. For purposes of OPT Extend, patients’ baseline PASI and PGA scores were defined as the values recorded on the day they were randomized in the earlier round of trials.
Through 2 years of follow-up in this interim analysis of OPT Extend, less than 10% of subjects discontinued tofacitinib because of adverse events. The adverse events were the same as the ones that arose in the earlier, briefer randomized trials, the longest of which lasted 1 year. No signs of cumulative organ toxicity were evident during the additional follow-up.
Slightly more than 4% of all adverse events were categorized as severe, while 62% were mild. The most frequent adverse events were nasopharyngitis in 16% of patients, increased creatinine phosphokinase in 10%, and upper respiratory tract infections in 7%. Serious infections requiring systemic antibiotics or hospitalization occurred in 1.8% of patients on tofacitinib for 2 years, the most common of which were pneumonia, herpes zoster, and urinary tract infection. Herpes zoster occurred in 3.5% of participants, with most cases being mild or moderate. Malignancies other than nonmelanoma skin cancers occurred in 1.2% of subjects.
In terms of laboratory findings of interest, patients’ LDL/HDL ratio remained stable throughout 24 months on tofacitinib. Nor were there any clinically meaningful changes in average levels of other laboratory parameters, including hemoglobin, creatinine phosphokinase, lymphocytes, and neutrophils. A lymphocyte count below 500/mm3 occurred in 0.4% of patients at some point; however, it was unrelated to duration of exposure to tofacitinib and wasn’t linked to an increased infection rate.
Dr. Augustin said an unmet need exists for effective and nontoxic oral medications for moderate to severe psoriasis, because many patients would prefer not to take an injectable biologic. Tofacitinib is aimed at filling that need, and serving as an easier, less toxic initial therapy. However, it is essential that an optimal oral agent have a favorable safety profile because psoriasis is a lifelong disease with an average lifetime duration in excess of 40 years in adults and more than 60 years in affected children, he said.
Pfizer, which is developing the drug, has filed for marketing approval for tofacitinib for treatment of adults with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy or phototherapy. The Food and Drug Administration has announced its intent to issue a ruling on the application in October 2015.
The OPT Expect study is funded by Pfizer. Dr. Augustin is an adviser to and/or a recipient of research grants from Pfizer and more than a dozen other medical companies.
VANCOUVER – Oral tofacitinib for adults with moderate to severe plaque psoriasis demonstrated sustained efficacy and a “generally manageable” safety profile throughout 2 years of follow-up in a large, multicenter ongoing long-term extension study, Dr. Matthias Augustin reported at the World Congress of Dermatology.
One month after enrolling in the phase III, open-label trial (known as OPT Extend), and going on the investigational oral Janus kinase inhibitor at 10 mg b.i.d., 56.2% of the 2,847 participants showed a PASI 75 response – that is, at least a 75% reduction from their baseline Psoriasis Area and Severity Index score. After 2 years in the OPT Extend study, 64.5% of the 1,912 patients remaining in the study were PASI 75 responders.
Similarly, 56.3% of patients went from moderate or severe disease at baseline to “clear” or “almost clear” by Physician Global Assessment (PGA) at 1 month, and 53.9% of participants had a PGA score of 0 or 1 at 24 months, added Dr. Augustin, professor of dermatology at the University of Hamburg-Eppendorf and director of the German Center for Health Services Research in Dermatology and Nursing.
Psoriasis patients who had participated in any of four earlier phase III or two phase II randomized, controlled trials of tofacitinib were eligible to participate in OPT Extend. Participants were placed on tofacitinib at 10 mg b.i.d. for the first 2 months of the open-label study; then investigators adjusted their dose individually – either 5 or 10 mg b.i.d. – at each follow-up visit based on efficacy and side effects. For purposes of OPT Extend, patients’ baseline PASI and PGA scores were defined as the values recorded on the day they were randomized in the earlier round of trials.
Through 2 years of follow-up in this interim analysis of OPT Extend, less than 10% of subjects discontinued tofacitinib because of adverse events. The adverse events were the same as the ones that arose in the earlier, briefer randomized trials, the longest of which lasted 1 year. No signs of cumulative organ toxicity were evident during the additional follow-up.
Slightly more than 4% of all adverse events were categorized as severe, while 62% were mild. The most frequent adverse events were nasopharyngitis in 16% of patients, increased creatinine phosphokinase in 10%, and upper respiratory tract infections in 7%. Serious infections requiring systemic antibiotics or hospitalization occurred in 1.8% of patients on tofacitinib for 2 years, the most common of which were pneumonia, herpes zoster, and urinary tract infection. Herpes zoster occurred in 3.5% of participants, with most cases being mild or moderate. Malignancies other than nonmelanoma skin cancers occurred in 1.2% of subjects.
In terms of laboratory findings of interest, patients’ LDL/HDL ratio remained stable throughout 24 months on tofacitinib. Nor were there any clinically meaningful changes in average levels of other laboratory parameters, including hemoglobin, creatinine phosphokinase, lymphocytes, and neutrophils. A lymphocyte count below 500/mm3 occurred in 0.4% of patients at some point; however, it was unrelated to duration of exposure to tofacitinib and wasn’t linked to an increased infection rate.
Dr. Augustin said an unmet need exists for effective and nontoxic oral medications for moderate to severe psoriasis, because many patients would prefer not to take an injectable biologic. Tofacitinib is aimed at filling that need, and serving as an easier, less toxic initial therapy. However, it is essential that an optimal oral agent have a favorable safety profile because psoriasis is a lifelong disease with an average lifetime duration in excess of 40 years in adults and more than 60 years in affected children, he said.
Pfizer, which is developing the drug, has filed for marketing approval for tofacitinib for treatment of adults with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy or phototherapy. The Food and Drug Administration has announced its intent to issue a ruling on the application in October 2015.
The OPT Expect study is funded by Pfizer. Dr. Augustin is an adviser to and/or a recipient of research grants from Pfizer and more than a dozen other medical companies.
AT WCD 2015
Key clinical point: Oral tofacitinib showed sustained efficacy for moderate to severe plaque psoriasis at 24 months in a large ongoing phase III study.
Major finding: At month 1 of the OPT Extend study, 56.2% of patients on tofacitinib had a PASI 75 response; at month 24, the PASI 75 rate was 64.5%.
Data source: The OPT Extend study is an ongoing, phase III multicenter open-label study of long-term therapy with oral tofacitinib at 5 or 10 mg b.i.d. in 2,847 patients with moderate to severe psoriasis.
Disclosures: The study is funded by Pfizer. The presenter is an adviser to and recipient of research grants from that company as well as others.
Anti-TNFs help psoriatic arthritis patients get back to work
ROME – Anti–tumor necrosis factor agents have a slight edge over conventional disease-modifying antirheumatic drugs when it comes to helping psoriatic arthritis patients who are having work issues, according to a large British observational study presented at the European Congress of Rheumatology.
Among 236 of 400 subjects working at baseline, presenteeism improved from 30% to 10% and productivity loss improved from 45% to 10% among patients who started taking anti-TNF (anti-tumor necrosis factor) agents. Gains were more modest when patients were started on DMARDs, with presenteeism improving from 30% to 20% and productivity loss from 40% to 25%. The difference in change of presenteeism between the two treatment groups became statistically significant at 2 weeks and remained so at 24 weeks.
“Work disability is a continuum,” said the presenting author, Dr. William Tillett of the Royal National Hospital for Rheumatic Diseases in Bath (England). It starts with the normal situation then graduates from presenteeism, where the individual is sick but still attends the workplace, to absenteeism, where the individual is sick and no longer attends the place of work, and eventual unemployment, he explained. “This study suggests that work disability is reversible in the real-world setting,” he added.
The study is from the Long-term Outcomes in Psoriatic ArthritiS (LOPAS II) working group, a 2-year, multicenter, prospective, observational cohort study of work disability in psoriatic arthritis. The group has previously reported that unemployment in psoriatic arthritis is associated with older age, disease duration of 2-5 years, and worse physical function, but that employer awareness and helpfulness enabled patients to stay on the job. Higher levels of global and joint-specific disease activity and worse physical function were associated with greater levels of reporting to work sick (presenteeism) and productivity loss (Rheumatology 2015;54:157-62).
The latest study by Dr. Tillett and his team is a follow-up to see how treatment affects work performance. At baseline, before treatment with anti-TNF or DMARDs, the LOPAS II team of investigators found that 164 (41%) of their 400 subjects were unemployed. Unemployed patients tended to be older (median of 59 years vs. 49 years) and to have worse physical function (a median score of 1.4 on the Health Assessment Questionnaire vs. 1.0). Subsequent treatment with anti-TNFs or DMARDs didn’t change overall employment levels.
Patients who started on anti-TNFs tended to have longer disease duration (median of 11 vs. 5 years) and a greater median number of tender (16 vs. 11) and swollen (7 vs. 5) joints, but otherwise there were no significant differences in demographic or clinical measures between the two treatment groups.
Median scores on the Disease Activity Index for Psoriatic Arthritis (DAPSA) improved over 24 weeks from 53 to 14 among anti-TNF patients, which is considered a good response, but only improved from 39 to 30 in the DMARD group, which is considered a poor response. All of the findings were statistically significant.
The results revealed a “surprisingly poor clinical response to synthetic DMARDs on clinical outcomes … as opposed to good response amongst patients commenced on TNF inhibitors,” Dr. Tillett said in an interview. The improvement in work disability and disease activity seen also was greater and more rapid among those who started on anti-TNF rather than synthetic DMARD.
Dr. Tillett reported receiving grant/research support from AbbVie and speaker or advisory board fees from UCB, Pfizer, and AbbVie. The other authors said they have no disclosures.
ROME – Anti–tumor necrosis factor agents have a slight edge over conventional disease-modifying antirheumatic drugs when it comes to helping psoriatic arthritis patients who are having work issues, according to a large British observational study presented at the European Congress of Rheumatology.
Among 236 of 400 subjects working at baseline, presenteeism improved from 30% to 10% and productivity loss improved from 45% to 10% among patients who started taking anti-TNF (anti-tumor necrosis factor) agents. Gains were more modest when patients were started on DMARDs, with presenteeism improving from 30% to 20% and productivity loss from 40% to 25%. The difference in change of presenteeism between the two treatment groups became statistically significant at 2 weeks and remained so at 24 weeks.
“Work disability is a continuum,” said the presenting author, Dr. William Tillett of the Royal National Hospital for Rheumatic Diseases in Bath (England). It starts with the normal situation then graduates from presenteeism, where the individual is sick but still attends the workplace, to absenteeism, where the individual is sick and no longer attends the place of work, and eventual unemployment, he explained. “This study suggests that work disability is reversible in the real-world setting,” he added.
The study is from the Long-term Outcomes in Psoriatic ArthritiS (LOPAS II) working group, a 2-year, multicenter, prospective, observational cohort study of work disability in psoriatic arthritis. The group has previously reported that unemployment in psoriatic arthritis is associated with older age, disease duration of 2-5 years, and worse physical function, but that employer awareness and helpfulness enabled patients to stay on the job. Higher levels of global and joint-specific disease activity and worse physical function were associated with greater levels of reporting to work sick (presenteeism) and productivity loss (Rheumatology 2015;54:157-62).
The latest study by Dr. Tillett and his team is a follow-up to see how treatment affects work performance. At baseline, before treatment with anti-TNF or DMARDs, the LOPAS II team of investigators found that 164 (41%) of their 400 subjects were unemployed. Unemployed patients tended to be older (median of 59 years vs. 49 years) and to have worse physical function (a median score of 1.4 on the Health Assessment Questionnaire vs. 1.0). Subsequent treatment with anti-TNFs or DMARDs didn’t change overall employment levels.
Patients who started on anti-TNFs tended to have longer disease duration (median of 11 vs. 5 years) and a greater median number of tender (16 vs. 11) and swollen (7 vs. 5) joints, but otherwise there were no significant differences in demographic or clinical measures between the two treatment groups.
Median scores on the Disease Activity Index for Psoriatic Arthritis (DAPSA) improved over 24 weeks from 53 to 14 among anti-TNF patients, which is considered a good response, but only improved from 39 to 30 in the DMARD group, which is considered a poor response. All of the findings were statistically significant.
The results revealed a “surprisingly poor clinical response to synthetic DMARDs on clinical outcomes … as opposed to good response amongst patients commenced on TNF inhibitors,” Dr. Tillett said in an interview. The improvement in work disability and disease activity seen also was greater and more rapid among those who started on anti-TNF rather than synthetic DMARD.
Dr. Tillett reported receiving grant/research support from AbbVie and speaker or advisory board fees from UCB, Pfizer, and AbbVie. The other authors said they have no disclosures.
ROME – Anti–tumor necrosis factor agents have a slight edge over conventional disease-modifying antirheumatic drugs when it comes to helping psoriatic arthritis patients who are having work issues, according to a large British observational study presented at the European Congress of Rheumatology.
Among 236 of 400 subjects working at baseline, presenteeism improved from 30% to 10% and productivity loss improved from 45% to 10% among patients who started taking anti-TNF (anti-tumor necrosis factor) agents. Gains were more modest when patients were started on DMARDs, with presenteeism improving from 30% to 20% and productivity loss from 40% to 25%. The difference in change of presenteeism between the two treatment groups became statistically significant at 2 weeks and remained so at 24 weeks.
“Work disability is a continuum,” said the presenting author, Dr. William Tillett of the Royal National Hospital for Rheumatic Diseases in Bath (England). It starts with the normal situation then graduates from presenteeism, where the individual is sick but still attends the workplace, to absenteeism, where the individual is sick and no longer attends the place of work, and eventual unemployment, he explained. “This study suggests that work disability is reversible in the real-world setting,” he added.
The study is from the Long-term Outcomes in Psoriatic ArthritiS (LOPAS II) working group, a 2-year, multicenter, prospective, observational cohort study of work disability in psoriatic arthritis. The group has previously reported that unemployment in psoriatic arthritis is associated with older age, disease duration of 2-5 years, and worse physical function, but that employer awareness and helpfulness enabled patients to stay on the job. Higher levels of global and joint-specific disease activity and worse physical function were associated with greater levels of reporting to work sick (presenteeism) and productivity loss (Rheumatology 2015;54:157-62).
The latest study by Dr. Tillett and his team is a follow-up to see how treatment affects work performance. At baseline, before treatment with anti-TNF or DMARDs, the LOPAS II team of investigators found that 164 (41%) of their 400 subjects were unemployed. Unemployed patients tended to be older (median of 59 years vs. 49 years) and to have worse physical function (a median score of 1.4 on the Health Assessment Questionnaire vs. 1.0). Subsequent treatment with anti-TNFs or DMARDs didn’t change overall employment levels.
Patients who started on anti-TNFs tended to have longer disease duration (median of 11 vs. 5 years) and a greater median number of tender (16 vs. 11) and swollen (7 vs. 5) joints, but otherwise there were no significant differences in demographic or clinical measures between the two treatment groups.
Median scores on the Disease Activity Index for Psoriatic Arthritis (DAPSA) improved over 24 weeks from 53 to 14 among anti-TNF patients, which is considered a good response, but only improved from 39 to 30 in the DMARD group, which is considered a poor response. All of the findings were statistically significant.
The results revealed a “surprisingly poor clinical response to synthetic DMARDs on clinical outcomes … as opposed to good response amongst patients commenced on TNF inhibitors,” Dr. Tillett said in an interview. The improvement in work disability and disease activity seen also was greater and more rapid among those who started on anti-TNF rather than synthetic DMARD.
Dr. Tillett reported receiving grant/research support from AbbVie and speaker or advisory board fees from UCB, Pfizer, and AbbVie. The other authors said they have no disclosures.
AT THE EULAR 2015 CONGRESS
Key clinical point: Work disability is a reversible outcome in psoriatic arthritis and improves significantly more with use of anti-TNF drugs than with synthetic DMARDs.
Major finding: Presenteeism improved from 30% to 10% and productivity loss improved from 45% to 10% among patients who started taking anti-TNF agents.
Data source: A 2-year, multicenter, prospective, observational cohort study of work disability in 400 patients with psoriatic arthritis.
Disclosures: Dr. Tillett reported receiving research support from AbbVie and speaker or advisory board fees from UCB, Pfizer, and Abbvie.
Fathers factor in psoriatic disease
Researchers have discovered further evidence of paternal transmission bias in psoriatic disease, according to a study published in Arthritis Care & Research.
Significantly more individuals with a first-degree relative affected with psoriatic disease reported an affected father (57%), compared with an affected mother (43%). Self-reported family history was obtained from 849 Canadian adults who reported a first-degree relative affected with either cutaneous psoriasis without arthritis (PsC) or psoriatic arthritis (PsA). A total of 532 (63%) reported an affected parent, and 23 probands reported that both parents were affected. The researchers found significantly more fathers with psoriatic disease than mothers: 289 (57%) of 509 discordant pairs vs. 220 (43%) of 509 discordant pairs, respectively (P = .003).
The paternal transmission bias did not reach statistical significance in PsC probands (P = .20), but the proportion of paternal PsC–proband PsA pairs (161 [75%] of 214 paternal transmissions) was significantly larger (P = .02) than maternal PsC–proband PsA pairs (103 [64%] of 161 maternal transmissions).
“If PsA is considered a more severe form of psoriatic disease than PsC alone, this finding suggests that there is a greater chance of an increase in disease severity when psoriatic disease is transmitted by an affected male compared to an affected female,” wrote the investigators, led by Remy A. Pollock of Toronto Western Hospital.
Read the full article here: (Arthritis Care Res. 2015 [doi:10.1002/acr.22625]).
Researchers have discovered further evidence of paternal transmission bias in psoriatic disease, according to a study published in Arthritis Care & Research.
Significantly more individuals with a first-degree relative affected with psoriatic disease reported an affected father (57%), compared with an affected mother (43%). Self-reported family history was obtained from 849 Canadian adults who reported a first-degree relative affected with either cutaneous psoriasis without arthritis (PsC) or psoriatic arthritis (PsA). A total of 532 (63%) reported an affected parent, and 23 probands reported that both parents were affected. The researchers found significantly more fathers with psoriatic disease than mothers: 289 (57%) of 509 discordant pairs vs. 220 (43%) of 509 discordant pairs, respectively (P = .003).
The paternal transmission bias did not reach statistical significance in PsC probands (P = .20), but the proportion of paternal PsC–proband PsA pairs (161 [75%] of 214 paternal transmissions) was significantly larger (P = .02) than maternal PsC–proband PsA pairs (103 [64%] of 161 maternal transmissions).
“If PsA is considered a more severe form of psoriatic disease than PsC alone, this finding suggests that there is a greater chance of an increase in disease severity when psoriatic disease is transmitted by an affected male compared to an affected female,” wrote the investigators, led by Remy A. Pollock of Toronto Western Hospital.
Read the full article here: (Arthritis Care Res. 2015 [doi:10.1002/acr.22625]).
Researchers have discovered further evidence of paternal transmission bias in psoriatic disease, according to a study published in Arthritis Care & Research.
Significantly more individuals with a first-degree relative affected with psoriatic disease reported an affected father (57%), compared with an affected mother (43%). Self-reported family history was obtained from 849 Canadian adults who reported a first-degree relative affected with either cutaneous psoriasis without arthritis (PsC) or psoriatic arthritis (PsA). A total of 532 (63%) reported an affected parent, and 23 probands reported that both parents were affected. The researchers found significantly more fathers with psoriatic disease than mothers: 289 (57%) of 509 discordant pairs vs. 220 (43%) of 509 discordant pairs, respectively (P = .003).
The paternal transmission bias did not reach statistical significance in PsC probands (P = .20), but the proportion of paternal PsC–proband PsA pairs (161 [75%] of 214 paternal transmissions) was significantly larger (P = .02) than maternal PsC–proband PsA pairs (103 [64%] of 161 maternal transmissions).
“If PsA is considered a more severe form of psoriatic disease than PsC alone, this finding suggests that there is a greater chance of an increase in disease severity when psoriatic disease is transmitted by an affected male compared to an affected female,” wrote the investigators, led by Remy A. Pollock of Toronto Western Hospital.
Read the full article here: (Arthritis Care Res. 2015 [doi:10.1002/acr.22625]).
FROM ARTHRITIS CARE & RESEARCH