User login
VIDEO: Could a deep Koebner phenomenon trigger psoriatic arthritis?
ROME – Psoriasis patients with a past history of bone or joint trauma had about 50% higher risk of later developing psoriatic arthritis than did those without a history of trauma in a longitudinal, population-based study of more than 70,000 psoriasis patients in the United Kingdom.
The relationship between trauma and later development of psoriatic arthritis could involve a deep Koebner phenomenon similar to what is observed with the Koebner phenomenon in the skin, suggested lead investigator Dr. Thorvardur Löve in an interview at the European Congress of Rheumatology.
Based on these findings, “one of the things that we are very excited about is the potential to think of strategies and test strategies that might be used in psoriasis patients once they are injured. So should we do anything different in an injured psoriasis patient, for instance, some sort of preventive treatment?” said Dr. Löve of Landspitali University Hospital in Reykjavik, Iceland.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ROME – Psoriasis patients with a past history of bone or joint trauma had about 50% higher risk of later developing psoriatic arthritis than did those without a history of trauma in a longitudinal, population-based study of more than 70,000 psoriasis patients in the United Kingdom.
The relationship between trauma and later development of psoriatic arthritis could involve a deep Koebner phenomenon similar to what is observed with the Koebner phenomenon in the skin, suggested lead investigator Dr. Thorvardur Löve in an interview at the European Congress of Rheumatology.
Based on these findings, “one of the things that we are very excited about is the potential to think of strategies and test strategies that might be used in psoriasis patients once they are injured. So should we do anything different in an injured psoriasis patient, for instance, some sort of preventive treatment?” said Dr. Löve of Landspitali University Hospital in Reykjavik, Iceland.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ROME – Psoriasis patients with a past history of bone or joint trauma had about 50% higher risk of later developing psoriatic arthritis than did those without a history of trauma in a longitudinal, population-based study of more than 70,000 psoriasis patients in the United Kingdom.
The relationship between trauma and later development of psoriatic arthritis could involve a deep Koebner phenomenon similar to what is observed with the Koebner phenomenon in the skin, suggested lead investigator Dr. Thorvardur Löve in an interview at the European Congress of Rheumatology.
Based on these findings, “one of the things that we are very excited about is the potential to think of strategies and test strategies that might be used in psoriasis patients once they are injured. So should we do anything different in an injured psoriasis patient, for instance, some sort of preventive treatment?” said Dr. Löve of Landspitali University Hospital in Reykjavik, Iceland.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE EULAR 2015 CONGRESS
Amgen’s termination of brodalumab stuns psoriasis world
VANCOUVER, B.C. – Amgen’s recent announcement that it is pulling the plug on further development of the investigational interleukin-17 receptor A inhibitor brodalumab was the red hot topic among psoriasis experts at the World Congress of Dermatology.
The biologic agent had seemingly been breezing through phase III clinical trials, racking up head-turning efficacy and reasonable safety numbers in a series of very large randomized studies conducted in patients with moderate-to-severe chronic plaque psoriasis. Then came the surprise announcement a scant couple of weeks before WCD 2015.
Brodalumab was also in codevelopment by Amgen and AstraZeneca for psoriatic arthritis and axial spondyloarthritis. Amgen is halting all participation in those research projects as well.
The company’s decision was based upon preliminary indications from the Food and Drug Administration that, as a condition for marketing approval, there would likely need to be restrictive labeling calling for physician monitoring for suicidality, something few dermatologists or rheumatologists are comfortable with.
“During our preparation process for regulatory submissions, we came to believe that labeling requirements likely would limit the appropriate patient population for brodalumab,” Dr. Sean E. Harper, executive vice president of research and development at Amgen, said in the company’s announcement.
AstraZeneca officials said in a separate statement that they will review all the brodalumab data in detail and make a decision as soon as possible regarding whether to pursue unilateral development of the biologic agent.
Dr. Richard G. Langley, who at WCD 2015 presented the results of the AMAGINE-3 trial – 1,881 psoriasis patients randomized to ustekinumab (Stelara) at the approved dosing, placebo, or brodalumab at either 140 mg or 210 mg subcutaneously – noted that rates of depression and suicidal ideation were similarly low in the ustekinumab and brodalumab groups during the 52-week study, and there were no suicide attempts.
The problem for Amgen was a single-digit numeric imbalance in these adverse events in some of the earlier, considerably smaller studies of brodalumab, according to Dr. Langley, professor of dermatology at Dalhousie University in Halifax, Nova Scotia.
“We have to remember that we can sometimes make spurious assumptions about rare events and perhaps cast a shadow on a pathway or molecule before we have full information,” he said diplomatically. “The Amgen decision is particularly unfortunate for patients because with brodalumab the only patients I’ve had who were depressed were the ones I’ve told are now no longer going to get the medication.”
Session chair Dr. Yves Poulin of Laval University in Quebec City, Quebec, said his experience has been similar: Patients love the unprecedented effectiveness of brodalumab and are crestfallen at learning they have to go off it.
Indeed, in AMAGINE-3, the 12-week PASI 75 rate among patients on brodalumab at 210 mg every 2 weeks – the more effective dose of the IL-17A inhibitor – was 85.1%, compared with 69.2% for ustekinumab, which is one of the most widely prescribed biologics. The PASI 100 rate was 36.7% for brodalumab, twice the 18.5% rate with ustekinumab. And the Investigator’s Global Assessment rating of 0 or 1 – clear or almost clear – was 79.6% with brodalumab, compared with 59% for ustekinumab, Dr. Langley reported.
“A consistent finding with brodalumab and the other IL-17 inhibitors is the remarkably fast improvement: within the first 2-4 weeks there’s a robust response,” he added.
Elsewhere at WCD 2015, Dr. Alan Menter presented the findings of the AMAGINE-2 trial, an 1,831-patient study identical in design to AMAGINE-3. The efficacy and safety results were virtually the same as in AMAGINE-3 as well, both in the 12-week induction phase and out to 52 weeks.
“We have a major issue that I think people haven’t understood well over the years relating to depression and psoriasis. Suicidal ideation has always been an issue. If you’re a 25-year-old and you’re trying to make your way in life and you’ve got significant psoriasis, you’re going to get depressed. But the suicidal ideation is twice as high in young people with psoriasis at baseline than in all the other autoimmune diseases put together,” said Dr. Menter, chair of dermatology at Baylor University Medical Center, Dallas, and the founding president of the International Psoriasis Council.
There has been no suicidality signal with secukinumab (Cosentyx), which in January became the first and, to date, only IL-17 inhibitor approved for marketing, nor with ixekizumab, which is in phase III clinical trials. There are distinct differences between the IL-17 inhibitor molecules – for example, brodalumab is IgG2, secukinumab is IgG1, and ixekizumab is IgG4. And brodalumab is a receptor antibody that not only inhibits the activity of IL-17A, but of IL-17F and -E as well, whereas secukinumab and ixekizumab work by other mechanisms. Investigators are now taking a close look at these differences to see if they have effects in terms of clinical outcomes and side effect profiles, he said.
Dr. Langley and Dr. Menter reported having financial relationships with Amgen and numerous other pharmaceutical companies.
VANCOUVER, B.C. – Amgen’s recent announcement that it is pulling the plug on further development of the investigational interleukin-17 receptor A inhibitor brodalumab was the red hot topic among psoriasis experts at the World Congress of Dermatology.
The biologic agent had seemingly been breezing through phase III clinical trials, racking up head-turning efficacy and reasonable safety numbers in a series of very large randomized studies conducted in patients with moderate-to-severe chronic plaque psoriasis. Then came the surprise announcement a scant couple of weeks before WCD 2015.
Brodalumab was also in codevelopment by Amgen and AstraZeneca for psoriatic arthritis and axial spondyloarthritis. Amgen is halting all participation in those research projects as well.
The company’s decision was based upon preliminary indications from the Food and Drug Administration that, as a condition for marketing approval, there would likely need to be restrictive labeling calling for physician monitoring for suicidality, something few dermatologists or rheumatologists are comfortable with.
“During our preparation process for regulatory submissions, we came to believe that labeling requirements likely would limit the appropriate patient population for brodalumab,” Dr. Sean E. Harper, executive vice president of research and development at Amgen, said in the company’s announcement.
AstraZeneca officials said in a separate statement that they will review all the brodalumab data in detail and make a decision as soon as possible regarding whether to pursue unilateral development of the biologic agent.
Dr. Richard G. Langley, who at WCD 2015 presented the results of the AMAGINE-3 trial – 1,881 psoriasis patients randomized to ustekinumab (Stelara) at the approved dosing, placebo, or brodalumab at either 140 mg or 210 mg subcutaneously – noted that rates of depression and suicidal ideation were similarly low in the ustekinumab and brodalumab groups during the 52-week study, and there were no suicide attempts.
The problem for Amgen was a single-digit numeric imbalance in these adverse events in some of the earlier, considerably smaller studies of brodalumab, according to Dr. Langley, professor of dermatology at Dalhousie University in Halifax, Nova Scotia.
“We have to remember that we can sometimes make spurious assumptions about rare events and perhaps cast a shadow on a pathway or molecule before we have full information,” he said diplomatically. “The Amgen decision is particularly unfortunate for patients because with brodalumab the only patients I’ve had who were depressed were the ones I’ve told are now no longer going to get the medication.”
Session chair Dr. Yves Poulin of Laval University in Quebec City, Quebec, said his experience has been similar: Patients love the unprecedented effectiveness of brodalumab and are crestfallen at learning they have to go off it.
Indeed, in AMAGINE-3, the 12-week PASI 75 rate among patients on brodalumab at 210 mg every 2 weeks – the more effective dose of the IL-17A inhibitor – was 85.1%, compared with 69.2% for ustekinumab, which is one of the most widely prescribed biologics. The PASI 100 rate was 36.7% for brodalumab, twice the 18.5% rate with ustekinumab. And the Investigator’s Global Assessment rating of 0 or 1 – clear or almost clear – was 79.6% with brodalumab, compared with 59% for ustekinumab, Dr. Langley reported.
“A consistent finding with brodalumab and the other IL-17 inhibitors is the remarkably fast improvement: within the first 2-4 weeks there’s a robust response,” he added.
Elsewhere at WCD 2015, Dr. Alan Menter presented the findings of the AMAGINE-2 trial, an 1,831-patient study identical in design to AMAGINE-3. The efficacy and safety results were virtually the same as in AMAGINE-3 as well, both in the 12-week induction phase and out to 52 weeks.
“We have a major issue that I think people haven’t understood well over the years relating to depression and psoriasis. Suicidal ideation has always been an issue. If you’re a 25-year-old and you’re trying to make your way in life and you’ve got significant psoriasis, you’re going to get depressed. But the suicidal ideation is twice as high in young people with psoriasis at baseline than in all the other autoimmune diseases put together,” said Dr. Menter, chair of dermatology at Baylor University Medical Center, Dallas, and the founding president of the International Psoriasis Council.
There has been no suicidality signal with secukinumab (Cosentyx), which in January became the first and, to date, only IL-17 inhibitor approved for marketing, nor with ixekizumab, which is in phase III clinical trials. There are distinct differences between the IL-17 inhibitor molecules – for example, brodalumab is IgG2, secukinumab is IgG1, and ixekizumab is IgG4. And brodalumab is a receptor antibody that not only inhibits the activity of IL-17A, but of IL-17F and -E as well, whereas secukinumab and ixekizumab work by other mechanisms. Investigators are now taking a close look at these differences to see if they have effects in terms of clinical outcomes and side effect profiles, he said.
Dr. Langley and Dr. Menter reported having financial relationships with Amgen and numerous other pharmaceutical companies.
VANCOUVER, B.C. – Amgen’s recent announcement that it is pulling the plug on further development of the investigational interleukin-17 receptor A inhibitor brodalumab was the red hot topic among psoriasis experts at the World Congress of Dermatology.
The biologic agent had seemingly been breezing through phase III clinical trials, racking up head-turning efficacy and reasonable safety numbers in a series of very large randomized studies conducted in patients with moderate-to-severe chronic plaque psoriasis. Then came the surprise announcement a scant couple of weeks before WCD 2015.
Brodalumab was also in codevelopment by Amgen and AstraZeneca for psoriatic arthritis and axial spondyloarthritis. Amgen is halting all participation in those research projects as well.
The company’s decision was based upon preliminary indications from the Food and Drug Administration that, as a condition for marketing approval, there would likely need to be restrictive labeling calling for physician monitoring for suicidality, something few dermatologists or rheumatologists are comfortable with.
“During our preparation process for regulatory submissions, we came to believe that labeling requirements likely would limit the appropriate patient population for brodalumab,” Dr. Sean E. Harper, executive vice president of research and development at Amgen, said in the company’s announcement.
AstraZeneca officials said in a separate statement that they will review all the brodalumab data in detail and make a decision as soon as possible regarding whether to pursue unilateral development of the biologic agent.
Dr. Richard G. Langley, who at WCD 2015 presented the results of the AMAGINE-3 trial – 1,881 psoriasis patients randomized to ustekinumab (Stelara) at the approved dosing, placebo, or brodalumab at either 140 mg or 210 mg subcutaneously – noted that rates of depression and suicidal ideation were similarly low in the ustekinumab and brodalumab groups during the 52-week study, and there were no suicide attempts.
The problem for Amgen was a single-digit numeric imbalance in these adverse events in some of the earlier, considerably smaller studies of brodalumab, according to Dr. Langley, professor of dermatology at Dalhousie University in Halifax, Nova Scotia.
“We have to remember that we can sometimes make spurious assumptions about rare events and perhaps cast a shadow on a pathway or molecule before we have full information,” he said diplomatically. “The Amgen decision is particularly unfortunate for patients because with brodalumab the only patients I’ve had who were depressed were the ones I’ve told are now no longer going to get the medication.”
Session chair Dr. Yves Poulin of Laval University in Quebec City, Quebec, said his experience has been similar: Patients love the unprecedented effectiveness of brodalumab and are crestfallen at learning they have to go off it.
Indeed, in AMAGINE-3, the 12-week PASI 75 rate among patients on brodalumab at 210 mg every 2 weeks – the more effective dose of the IL-17A inhibitor – was 85.1%, compared with 69.2% for ustekinumab, which is one of the most widely prescribed biologics. The PASI 100 rate was 36.7% for brodalumab, twice the 18.5% rate with ustekinumab. And the Investigator’s Global Assessment rating of 0 or 1 – clear or almost clear – was 79.6% with brodalumab, compared with 59% for ustekinumab, Dr. Langley reported.
“A consistent finding with brodalumab and the other IL-17 inhibitors is the remarkably fast improvement: within the first 2-4 weeks there’s a robust response,” he added.
Elsewhere at WCD 2015, Dr. Alan Menter presented the findings of the AMAGINE-2 trial, an 1,831-patient study identical in design to AMAGINE-3. The efficacy and safety results were virtually the same as in AMAGINE-3 as well, both in the 12-week induction phase and out to 52 weeks.
“We have a major issue that I think people haven’t understood well over the years relating to depression and psoriasis. Suicidal ideation has always been an issue. If you’re a 25-year-old and you’re trying to make your way in life and you’ve got significant psoriasis, you’re going to get depressed. But the suicidal ideation is twice as high in young people with psoriasis at baseline than in all the other autoimmune diseases put together,” said Dr. Menter, chair of dermatology at Baylor University Medical Center, Dallas, and the founding president of the International Psoriasis Council.
There has been no suicidality signal with secukinumab (Cosentyx), which in January became the first and, to date, only IL-17 inhibitor approved for marketing, nor with ixekizumab, which is in phase III clinical trials. There are distinct differences between the IL-17 inhibitor molecules – for example, brodalumab is IgG2, secukinumab is IgG1, and ixekizumab is IgG4. And brodalumab is a receptor antibody that not only inhibits the activity of IL-17A, but of IL-17F and -E as well, whereas secukinumab and ixekizumab work by other mechanisms. Investigators are now taking a close look at these differences to see if they have effects in terms of clinical outcomes and side effect profiles, he said.
Dr. Langley and Dr. Menter reported having financial relationships with Amgen and numerous other pharmaceutical companies.
EXPERT ANALYSIS FROM WCD 2015
Lasting ustekinumab benefits seen in psoriatic arthritis
The final results of a 2-year, phase III study assessing the clinical efficacy and safety of ustekinumab in 615 patients with active psoriatic arthritis confirm that joint- and skin-related improvements are maintained.
Data from the randomized, double blind, placebo-controlled PSUMMIT 1 study showed that 56.7%-63.6% of psoriatic arthritis (PsA) patients treated with a 45-mg or 90-mg dose every 12 weeks achieved American College of Rheumatology criteria for a 20% improvement in joint symptoms (ACR20). ACR50 and ACR70 responses ranged from 37.3% to 46% and 18.6% to 24.7%, respectively.
“The proportions of patients with either DAS28-CRP [28-joint Disease Activity Score using C-reactive protein] response or remission were maintained from week 52 to week 100,” noted lead study author Dr. Arthur Kavanaugh of the University of California-San Diego in La Jolla and his associates (Arthritis Care Res. 2015 June 19 [doi:10.1002/acr.22645]).
A moderate or good response according to DAS28-CRP was achieved by 71.9%-76.7% of patients. A 75% improvement in the Psoriasis Area and Severity Index (PASI) was achieved by 63.9%-72.5% of patients, and 41%-51.9% achieved a PASI 90. Mean changes in radiographic damage also were maintained.
“No unexpected safety events were observed through 2 years, and results were consistent with the known safety profile of ustekinumab,” Dr. Kavanaugh and his colleagues wrote, adding that the study “demonstrated a favorable benefit-risk profile of ustekinumab treatment in patients with active PsA.”
The trial was funded by Janssen Research & Development. Dr. Kavanaugh has received research support from AbbVie, Janssen, and UCB. Many of the other authors also had financial ties to Janssen and other manufacturers of biologics for psoriatic arthritis.
The final results of a 2-year, phase III study assessing the clinical efficacy and safety of ustekinumab in 615 patients with active psoriatic arthritis confirm that joint- and skin-related improvements are maintained.
Data from the randomized, double blind, placebo-controlled PSUMMIT 1 study showed that 56.7%-63.6% of psoriatic arthritis (PsA) patients treated with a 45-mg or 90-mg dose every 12 weeks achieved American College of Rheumatology criteria for a 20% improvement in joint symptoms (ACR20). ACR50 and ACR70 responses ranged from 37.3% to 46% and 18.6% to 24.7%, respectively.
“The proportions of patients with either DAS28-CRP [28-joint Disease Activity Score using C-reactive protein] response or remission were maintained from week 52 to week 100,” noted lead study author Dr. Arthur Kavanaugh of the University of California-San Diego in La Jolla and his associates (Arthritis Care Res. 2015 June 19 [doi:10.1002/acr.22645]).
A moderate or good response according to DAS28-CRP was achieved by 71.9%-76.7% of patients. A 75% improvement in the Psoriasis Area and Severity Index (PASI) was achieved by 63.9%-72.5% of patients, and 41%-51.9% achieved a PASI 90. Mean changes in radiographic damage also were maintained.
“No unexpected safety events were observed through 2 years, and results were consistent with the known safety profile of ustekinumab,” Dr. Kavanaugh and his colleagues wrote, adding that the study “demonstrated a favorable benefit-risk profile of ustekinumab treatment in patients with active PsA.”
The trial was funded by Janssen Research & Development. Dr. Kavanaugh has received research support from AbbVie, Janssen, and UCB. Many of the other authors also had financial ties to Janssen and other manufacturers of biologics for psoriatic arthritis.
The final results of a 2-year, phase III study assessing the clinical efficacy and safety of ustekinumab in 615 patients with active psoriatic arthritis confirm that joint- and skin-related improvements are maintained.
Data from the randomized, double blind, placebo-controlled PSUMMIT 1 study showed that 56.7%-63.6% of psoriatic arthritis (PsA) patients treated with a 45-mg or 90-mg dose every 12 weeks achieved American College of Rheumatology criteria for a 20% improvement in joint symptoms (ACR20). ACR50 and ACR70 responses ranged from 37.3% to 46% and 18.6% to 24.7%, respectively.
“The proportions of patients with either DAS28-CRP [28-joint Disease Activity Score using C-reactive protein] response or remission were maintained from week 52 to week 100,” noted lead study author Dr. Arthur Kavanaugh of the University of California-San Diego in La Jolla and his associates (Arthritis Care Res. 2015 June 19 [doi:10.1002/acr.22645]).
A moderate or good response according to DAS28-CRP was achieved by 71.9%-76.7% of patients. A 75% improvement in the Psoriasis Area and Severity Index (PASI) was achieved by 63.9%-72.5% of patients, and 41%-51.9% achieved a PASI 90. Mean changes in radiographic damage also were maintained.
“No unexpected safety events were observed through 2 years, and results were consistent with the known safety profile of ustekinumab,” Dr. Kavanaugh and his colleagues wrote, adding that the study “demonstrated a favorable benefit-risk profile of ustekinumab treatment in patients with active PsA.”
The trial was funded by Janssen Research & Development. Dr. Kavanaugh has received research support from AbbVie, Janssen, and UCB. Many of the other authors also had financial ties to Janssen and other manufacturers of biologics for psoriatic arthritis.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point: The effects of ustekinumab on joint and skin symptoms are maintained for up to 2 years in adult patients with PsA.
Major finding: ACR20, 50, and 70 responses ranged from 56.7% to 63.6%, 37.3% to 46%, and 18.6% to 24.7%, respectively.
Data source: Randomized, double blind, placebo-controlled phase III PSUMMIT1 trial assessing the clinical efficacy and safety of ustekinumab in 615 patients with active PsA.
Disclosures: The trial was funded by Janssen Research & Development. Dr. Kavanaugh has received research support from AbbVie, Janssen, and UCB. Many of the other authors also had financial ties to Janssen and other manufacturers of biologics for psoriatic arthritis.
EULAR: Panel previews updated CVD recommendations
ROME – The interval for assessing cardiovascular disease risk in patients with at least one inflamed joint can be as long as 5 years, depending on the patient, according to revised recommendations issued by a European League Against Rheumatism expert panel. The first edition of the recommendations called for annual assessment.
“We leave the assessment interval up to each clinician. Annually is very hard to incorporate into clinical practice,” said Dr. Michael T. Nurmohamed, convenor of both the initial recommendation panel as well as the group that produced the new revision. He previewed the updated recommendations during a talk at the European Congress of Rheumatology. The final form of the revised recommendations, which apply to patients with rheumatoid arthritis, psoriatic arthritis, or ankylosing spondylitis, will soon be posted online and published, he said.
The context for cardiovascular disease risk assessment of patients with these rheumatoid diseases who are in primary-prevention mode has changed since the initial version was released in 2009 (Ann. Rheum. Dis. 2010;69:325-31). That changed context led to a rethinking of the appropriate interval for risk-factor assessment, said Dr. Nurmohamed, professor and head of rheumatology research at VU University Medical Center in Amsterdam. “In 2009, tight control of rheumatoid diseases generally did not exist,” he said in an interview.
In patients with well-controlled rheumatoid disease, “you can assess their cardiovascular disease [CVD] risk, and if the risk is very low” you can defer the next follow-up assessment for 5 years or longer. But if a patient’s CVD risk is high, assess the patient more often, Dr. Nurmohamed suggested. The recommendations also call for an updated CVD risk assessment following a “major change” in antirheumatoid therapy.
The updated recommendations include two other notable changes. First, there is now a much deeper evidence base behind the need for CVD risk assessment and management in patients with psoriatic arthritis or ankylosing spondylitis. “We mentioned them before, but the data weren’t all that hard. We now have more evidence,” he said in an interview.
Second, the revised recommendations say that ultrasound examination of atherosclerotic plaque number and volume in a patient’s carotid artery “may be considered.” Dr. Nurmohamed acknowledged that carotid examination by ultrasound is very discretionary; it can provide useful risk information, but “not every rheumatologist will do it.”
Most other aspects of the revised recommendations remain similar to the 2009 edition. They emphasize controlling inflammation to lower CVD risk, multiplying a patient’s CVD risk score by a factor of 1.5 to better estimate their increased risk level due to their rheumatic disease, cautious use of nonsteroidal anti-inflammatory drugs, and minimized use of glucocorticoids. Multiplying a patient’s CVD risk score results in a “rough” risk estimate, he cautioned. “The problem is we use 1.5 for all patients, regardless of their rheumatoid-disease duration,” he said.
Recommended interventions to reduce a patient’s CVD risk include lifestyle steps of healthy diet, regular exercise, and smoking cessation, and the standard drug interventions for reducing CVD risk: antihypertensive medications and lipid-lowering drugs, especially statins. Target levels for treating hypertension and hyperlipidemia remain the same as for primary prevention in the general population, he said. CVD risk assessment and management steps for patients with one of the covered rheumatoid diseases and established CVD are the same as for standard secondary-prevention measures in the general population.
A notable gap in the revision is the continued absence of recommendations for patients with gout. “Gout is on our research agenda, but represents an enormous task,” Dr. Nurmohamed said. He anticipates that his panel will eventually develop recommendations for managing CVD risk in gout patients.
On Twitter @mitchelzoler
ROME – The interval for assessing cardiovascular disease risk in patients with at least one inflamed joint can be as long as 5 years, depending on the patient, according to revised recommendations issued by a European League Against Rheumatism expert panel. The first edition of the recommendations called for annual assessment.
“We leave the assessment interval up to each clinician. Annually is very hard to incorporate into clinical practice,” said Dr. Michael T. Nurmohamed, convenor of both the initial recommendation panel as well as the group that produced the new revision. He previewed the updated recommendations during a talk at the European Congress of Rheumatology. The final form of the revised recommendations, which apply to patients with rheumatoid arthritis, psoriatic arthritis, or ankylosing spondylitis, will soon be posted online and published, he said.
The context for cardiovascular disease risk assessment of patients with these rheumatoid diseases who are in primary-prevention mode has changed since the initial version was released in 2009 (Ann. Rheum. Dis. 2010;69:325-31). That changed context led to a rethinking of the appropriate interval for risk-factor assessment, said Dr. Nurmohamed, professor and head of rheumatology research at VU University Medical Center in Amsterdam. “In 2009, tight control of rheumatoid diseases generally did not exist,” he said in an interview.
In patients with well-controlled rheumatoid disease, “you can assess their cardiovascular disease [CVD] risk, and if the risk is very low” you can defer the next follow-up assessment for 5 years or longer. But if a patient’s CVD risk is high, assess the patient more often, Dr. Nurmohamed suggested. The recommendations also call for an updated CVD risk assessment following a “major change” in antirheumatoid therapy.
The updated recommendations include two other notable changes. First, there is now a much deeper evidence base behind the need for CVD risk assessment and management in patients with psoriatic arthritis or ankylosing spondylitis. “We mentioned them before, but the data weren’t all that hard. We now have more evidence,” he said in an interview.
Second, the revised recommendations say that ultrasound examination of atherosclerotic plaque number and volume in a patient’s carotid artery “may be considered.” Dr. Nurmohamed acknowledged that carotid examination by ultrasound is very discretionary; it can provide useful risk information, but “not every rheumatologist will do it.”
Most other aspects of the revised recommendations remain similar to the 2009 edition. They emphasize controlling inflammation to lower CVD risk, multiplying a patient’s CVD risk score by a factor of 1.5 to better estimate their increased risk level due to their rheumatic disease, cautious use of nonsteroidal anti-inflammatory drugs, and minimized use of glucocorticoids. Multiplying a patient’s CVD risk score results in a “rough” risk estimate, he cautioned. “The problem is we use 1.5 for all patients, regardless of their rheumatoid-disease duration,” he said.
Recommended interventions to reduce a patient’s CVD risk include lifestyle steps of healthy diet, regular exercise, and smoking cessation, and the standard drug interventions for reducing CVD risk: antihypertensive medications and lipid-lowering drugs, especially statins. Target levels for treating hypertension and hyperlipidemia remain the same as for primary prevention in the general population, he said. CVD risk assessment and management steps for patients with one of the covered rheumatoid diseases and established CVD are the same as for standard secondary-prevention measures in the general population.
A notable gap in the revision is the continued absence of recommendations for patients with gout. “Gout is on our research agenda, but represents an enormous task,” Dr. Nurmohamed said. He anticipates that his panel will eventually develop recommendations for managing CVD risk in gout patients.
On Twitter @mitchelzoler
ROME – The interval for assessing cardiovascular disease risk in patients with at least one inflamed joint can be as long as 5 years, depending on the patient, according to revised recommendations issued by a European League Against Rheumatism expert panel. The first edition of the recommendations called for annual assessment.
“We leave the assessment interval up to each clinician. Annually is very hard to incorporate into clinical practice,” said Dr. Michael T. Nurmohamed, convenor of both the initial recommendation panel as well as the group that produced the new revision. He previewed the updated recommendations during a talk at the European Congress of Rheumatology. The final form of the revised recommendations, which apply to patients with rheumatoid arthritis, psoriatic arthritis, or ankylosing spondylitis, will soon be posted online and published, he said.
The context for cardiovascular disease risk assessment of patients with these rheumatoid diseases who are in primary-prevention mode has changed since the initial version was released in 2009 (Ann. Rheum. Dis. 2010;69:325-31). That changed context led to a rethinking of the appropriate interval for risk-factor assessment, said Dr. Nurmohamed, professor and head of rheumatology research at VU University Medical Center in Amsterdam. “In 2009, tight control of rheumatoid diseases generally did not exist,” he said in an interview.
In patients with well-controlled rheumatoid disease, “you can assess their cardiovascular disease [CVD] risk, and if the risk is very low” you can defer the next follow-up assessment for 5 years or longer. But if a patient’s CVD risk is high, assess the patient more often, Dr. Nurmohamed suggested. The recommendations also call for an updated CVD risk assessment following a “major change” in antirheumatoid therapy.
The updated recommendations include two other notable changes. First, there is now a much deeper evidence base behind the need for CVD risk assessment and management in patients with psoriatic arthritis or ankylosing spondylitis. “We mentioned them before, but the data weren’t all that hard. We now have more evidence,” he said in an interview.
Second, the revised recommendations say that ultrasound examination of atherosclerotic plaque number and volume in a patient’s carotid artery “may be considered.” Dr. Nurmohamed acknowledged that carotid examination by ultrasound is very discretionary; it can provide useful risk information, but “not every rheumatologist will do it.”
Most other aspects of the revised recommendations remain similar to the 2009 edition. They emphasize controlling inflammation to lower CVD risk, multiplying a patient’s CVD risk score by a factor of 1.5 to better estimate their increased risk level due to their rheumatic disease, cautious use of nonsteroidal anti-inflammatory drugs, and minimized use of glucocorticoids. Multiplying a patient’s CVD risk score results in a “rough” risk estimate, he cautioned. “The problem is we use 1.5 for all patients, regardless of their rheumatoid-disease duration,” he said.
Recommended interventions to reduce a patient’s CVD risk include lifestyle steps of healthy diet, regular exercise, and smoking cessation, and the standard drug interventions for reducing CVD risk: antihypertensive medications and lipid-lowering drugs, especially statins. Target levels for treating hypertension and hyperlipidemia remain the same as for primary prevention in the general population, he said. CVD risk assessment and management steps for patients with one of the covered rheumatoid diseases and established CVD are the same as for standard secondary-prevention measures in the general population.
A notable gap in the revision is the continued absence of recommendations for patients with gout. “Gout is on our research agenda, but represents an enormous task,” Dr. Nurmohamed said. He anticipates that his panel will eventually develop recommendations for managing CVD risk in gout patients.
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM THE EULAR 2015 Congress
TNF Inhibitors May Reduce Cardiovascular Risks in Psoriasis Patients: Report From the AAD Meeting
Dr. Jashin Wu discusses results from a study on tumor necrosis factor (TNF) inhibitors for psoriasis and myocardial infarction risk. The study evaluated TNF inhibitor use, oral therapy and phototherapy, and topical therapy. He also reviews results from another study that looked at heart attacks, stroke, and cardiovascular death with biologics, methotrexate, retinoids, cyclosporine, and other therapies for psoriasis. If we want to believe there is a reduction of cardiovascular risk with TNF inhibitors, what's the mechanism? Dr. Wu presents several theories.
Dr. Jashin Wu discusses results from a study on tumor necrosis factor (TNF) inhibitors for psoriasis and myocardial infarction risk. The study evaluated TNF inhibitor use, oral therapy and phototherapy, and topical therapy. He also reviews results from another study that looked at heart attacks, stroke, and cardiovascular death with biologics, methotrexate, retinoids, cyclosporine, and other therapies for psoriasis. If we want to believe there is a reduction of cardiovascular risk with TNF inhibitors, what's the mechanism? Dr. Wu presents several theories.
Dr. Jashin Wu discusses results from a study on tumor necrosis factor (TNF) inhibitors for psoriasis and myocardial infarction risk. The study evaluated TNF inhibitor use, oral therapy and phototherapy, and topical therapy. He also reviews results from another study that looked at heart attacks, stroke, and cardiovascular death with biologics, methotrexate, retinoids, cyclosporine, and other therapies for psoriasis. If we want to believe there is a reduction of cardiovascular risk with TNF inhibitors, what's the mechanism? Dr. Wu presents several theories.
Large-scale psoriasis study links trauma to arthritis
ROME – Patients with psoriasis were more likely to develop psoriatic arthritis if they had experienced physical trauma, based on data from a large, population-based study.
The crude incidence of psoriatic arthritis was 30 per 10,000 person-years in psoriasis patients exposed to trauma, compared with 22 per 10,000 person years in those who were not.
The hazard ratio (HR) for increased psoriatic arthritis risk with any trauma was 1.32 after adjusting for multiple factors, including patient age, gender, and the duration of psoriasis, senior study author Dr. Thorvardur Love said during a press briefing at the European Congress of Rheumatology.
“Patients with psoriasis are an easily identifiable group [to study] as they have skin disease on their body,” he noted. They also have a high risk of developing arthritis, at around 10%-30% of patients. This makes them an ideal population to study to try to find factors that might mitigate the risk and potentially have a large impact in clinical practice.
“The idea that trauma precipitates psoriatic arthritis is not new,” observed Dr. Love of Landspitali University Hospital in Reykjavik, Iceland. “It comes a little bit from the Koebner phenomenon, which is when psoriasis patients develop a new lesion in the skin where an injury has been.”
A few small studies had given rise to the idea that trauma could perhaps trigger a deep Koebner phenomenon in patients with psoriasis, and so the aim of the present analysis was to look at this idea in a larger population. Electronic health records of more than 10 million individuals living in the United Kingdom between 1995 and 2013 were analyzed from the Health Improvement Network (THIN) database. Of 70,646 patients with psoriasis who were identified, 15,416 had been exposed to some form of trauma, which was stratified as trauma involving the joints, bones, nerves, or skin.
After 425,120 person-years of follow-up, 1,010 incident cases of psoriatic arthritis had been recorded.
Having bone or joint trauma was found to increase the risk for psoriatic arthritis by 46% (HR, 1.46; 95% confidence interval, 1.13-1.54) and 50% (HR, 1.50; 95% CI. 1.19-1.90), respectively. This was after adjusting for age, gender, date of entry into the THIN database, duration of psoriasis, body mass index, smoking, alcohol consumption, and the number of visits to the general practitioner.
Neither nerve nor skin trauma were associated with an increased risk for psoriatic arthritis. Dr. Love and his fellow researchers also looked to see if patients with psoriasis had an increased risk of rheumatoid arthritis but found no significant association (HR, 1.04; 95% 0.99-1.10).
“The conclusion is that physical trauma is a risk factor for psoriatic arthritis among patients with psoriasis,” Dr. Love said. “We believe this is very important as the baseline risk is so high.”The effect is specific to psoriatic arthritis, as it is not seen in rheumatoid arthritis, which might provide clues for further research, he added. Why trauma might up the risk for developing psoriatic but not other types of inflammatory arthritis remains unclear, but the hypothesis is that patients would need to have a genetic predisposition and the “right types” of T cells in and around the joint that get disturbed in some way, perhaps by infection or by trauma. “I think it’s important to note that at this point we are not making any recommendations to the psoriasis community,” Dr. Love said. He suggested that, before any recommendations could be made, there needed to be a “really robust” conversation between patients, researchers, and physicians to determine exactly what these findings might mean. Certainly more research is needed before suggesting any lifestyle modifications that might help avoid situations associated with certain types of trauma, he said.
A literature review in the journal Clinical Rheumatology provided additional explanation of the deep Koebner effect. The investigators noted in their abstract that “the role of neuropeptides such as substance P and vasoactive intestinal peptide has been highlighted in the synovium after trauma.”
An editorial in the Journal of Rheumatology also suggested areas for additional research. Dr. Ignazio Olivieri of San Carlo Hospital in Potenza, Italy, wrote that “criteria of imputability” that should be met include “single and significant trauma; absence of joint lesion before trauma; localization of arthritis in the area of trauma; and absence or short delay between trauma and onset of arthritis.”
“You might envision treating [psoriasis] patients early [for psoriatic arthritis] if they break a leg or get a joint dislocation, but we are not there yet,” Dr. Love stressed. “This is an idea of where we could take this and where we might actually be able to have an effect.”
The research was performed by researchers at the University of Iceland (Reykjavik) in collaboration with researchers at Harvard Medical School in Boston and the University of Pennsylvania in Philadelphia. It was partially funded by the Icelandic Centre for Research (RANNIS) and the National Institutes of Health. Dr. Love’s associate, Dr. Stefan Thorarensen of the division of public health at the University of Iceland, presented the findings during the clinical science session at the congress.
Dr. Love and his coauthors reported having no financial disclosures.
ROME – Patients with psoriasis were more likely to develop psoriatic arthritis if they had experienced physical trauma, based on data from a large, population-based study.
The crude incidence of psoriatic arthritis was 30 per 10,000 person-years in psoriasis patients exposed to trauma, compared with 22 per 10,000 person years in those who were not.
The hazard ratio (HR) for increased psoriatic arthritis risk with any trauma was 1.32 after adjusting for multiple factors, including patient age, gender, and the duration of psoriasis, senior study author Dr. Thorvardur Love said during a press briefing at the European Congress of Rheumatology.
“Patients with psoriasis are an easily identifiable group [to study] as they have skin disease on their body,” he noted. They also have a high risk of developing arthritis, at around 10%-30% of patients. This makes them an ideal population to study to try to find factors that might mitigate the risk and potentially have a large impact in clinical practice.
“The idea that trauma precipitates psoriatic arthritis is not new,” observed Dr. Love of Landspitali University Hospital in Reykjavik, Iceland. “It comes a little bit from the Koebner phenomenon, which is when psoriasis patients develop a new lesion in the skin where an injury has been.”
A few small studies had given rise to the idea that trauma could perhaps trigger a deep Koebner phenomenon in patients with psoriasis, and so the aim of the present analysis was to look at this idea in a larger population. Electronic health records of more than 10 million individuals living in the United Kingdom between 1995 and 2013 were analyzed from the Health Improvement Network (THIN) database. Of 70,646 patients with psoriasis who were identified, 15,416 had been exposed to some form of trauma, which was stratified as trauma involving the joints, bones, nerves, or skin.
After 425,120 person-years of follow-up, 1,010 incident cases of psoriatic arthritis had been recorded.
Having bone or joint trauma was found to increase the risk for psoriatic arthritis by 46% (HR, 1.46; 95% confidence interval, 1.13-1.54) and 50% (HR, 1.50; 95% CI. 1.19-1.90), respectively. This was after adjusting for age, gender, date of entry into the THIN database, duration of psoriasis, body mass index, smoking, alcohol consumption, and the number of visits to the general practitioner.
Neither nerve nor skin trauma were associated with an increased risk for psoriatic arthritis. Dr. Love and his fellow researchers also looked to see if patients with psoriasis had an increased risk of rheumatoid arthritis but found no significant association (HR, 1.04; 95% 0.99-1.10).
“The conclusion is that physical trauma is a risk factor for psoriatic arthritis among patients with psoriasis,” Dr. Love said. “We believe this is very important as the baseline risk is so high.”The effect is specific to psoriatic arthritis, as it is not seen in rheumatoid arthritis, which might provide clues for further research, he added. Why trauma might up the risk for developing psoriatic but not other types of inflammatory arthritis remains unclear, but the hypothesis is that patients would need to have a genetic predisposition and the “right types” of T cells in and around the joint that get disturbed in some way, perhaps by infection or by trauma. “I think it’s important to note that at this point we are not making any recommendations to the psoriasis community,” Dr. Love said. He suggested that, before any recommendations could be made, there needed to be a “really robust” conversation between patients, researchers, and physicians to determine exactly what these findings might mean. Certainly more research is needed before suggesting any lifestyle modifications that might help avoid situations associated with certain types of trauma, he said.
A literature review in the journal Clinical Rheumatology provided additional explanation of the deep Koebner effect. The investigators noted in their abstract that “the role of neuropeptides such as substance P and vasoactive intestinal peptide has been highlighted in the synovium after trauma.”
An editorial in the Journal of Rheumatology also suggested areas for additional research. Dr. Ignazio Olivieri of San Carlo Hospital in Potenza, Italy, wrote that “criteria of imputability” that should be met include “single and significant trauma; absence of joint lesion before trauma; localization of arthritis in the area of trauma; and absence or short delay between trauma and onset of arthritis.”
“You might envision treating [psoriasis] patients early [for psoriatic arthritis] if they break a leg or get a joint dislocation, but we are not there yet,” Dr. Love stressed. “This is an idea of where we could take this and where we might actually be able to have an effect.”
The research was performed by researchers at the University of Iceland (Reykjavik) in collaboration with researchers at Harvard Medical School in Boston and the University of Pennsylvania in Philadelphia. It was partially funded by the Icelandic Centre for Research (RANNIS) and the National Institutes of Health. Dr. Love’s associate, Dr. Stefan Thorarensen of the division of public health at the University of Iceland, presented the findings during the clinical science session at the congress.
Dr. Love and his coauthors reported having no financial disclosures.
ROME – Patients with psoriasis were more likely to develop psoriatic arthritis if they had experienced physical trauma, based on data from a large, population-based study.
The crude incidence of psoriatic arthritis was 30 per 10,000 person-years in psoriasis patients exposed to trauma, compared with 22 per 10,000 person years in those who were not.
The hazard ratio (HR) for increased psoriatic arthritis risk with any trauma was 1.32 after adjusting for multiple factors, including patient age, gender, and the duration of psoriasis, senior study author Dr. Thorvardur Love said during a press briefing at the European Congress of Rheumatology.
“Patients with psoriasis are an easily identifiable group [to study] as they have skin disease on their body,” he noted. They also have a high risk of developing arthritis, at around 10%-30% of patients. This makes them an ideal population to study to try to find factors that might mitigate the risk and potentially have a large impact in clinical practice.
“The idea that trauma precipitates psoriatic arthritis is not new,” observed Dr. Love of Landspitali University Hospital in Reykjavik, Iceland. “It comes a little bit from the Koebner phenomenon, which is when psoriasis patients develop a new lesion in the skin where an injury has been.”
A few small studies had given rise to the idea that trauma could perhaps trigger a deep Koebner phenomenon in patients with psoriasis, and so the aim of the present analysis was to look at this idea in a larger population. Electronic health records of more than 10 million individuals living in the United Kingdom between 1995 and 2013 were analyzed from the Health Improvement Network (THIN) database. Of 70,646 patients with psoriasis who were identified, 15,416 had been exposed to some form of trauma, which was stratified as trauma involving the joints, bones, nerves, or skin.
After 425,120 person-years of follow-up, 1,010 incident cases of psoriatic arthritis had been recorded.
Having bone or joint trauma was found to increase the risk for psoriatic arthritis by 46% (HR, 1.46; 95% confidence interval, 1.13-1.54) and 50% (HR, 1.50; 95% CI. 1.19-1.90), respectively. This was after adjusting for age, gender, date of entry into the THIN database, duration of psoriasis, body mass index, smoking, alcohol consumption, and the number of visits to the general practitioner.
Neither nerve nor skin trauma were associated with an increased risk for psoriatic arthritis. Dr. Love and his fellow researchers also looked to see if patients with psoriasis had an increased risk of rheumatoid arthritis but found no significant association (HR, 1.04; 95% 0.99-1.10).
“The conclusion is that physical trauma is a risk factor for psoriatic arthritis among patients with psoriasis,” Dr. Love said. “We believe this is very important as the baseline risk is so high.”The effect is specific to psoriatic arthritis, as it is not seen in rheumatoid arthritis, which might provide clues for further research, he added. Why trauma might up the risk for developing psoriatic but not other types of inflammatory arthritis remains unclear, but the hypothesis is that patients would need to have a genetic predisposition and the “right types” of T cells in and around the joint that get disturbed in some way, perhaps by infection or by trauma. “I think it’s important to note that at this point we are not making any recommendations to the psoriasis community,” Dr. Love said. He suggested that, before any recommendations could be made, there needed to be a “really robust” conversation between patients, researchers, and physicians to determine exactly what these findings might mean. Certainly more research is needed before suggesting any lifestyle modifications that might help avoid situations associated with certain types of trauma, he said.
A literature review in the journal Clinical Rheumatology provided additional explanation of the deep Koebner effect. The investigators noted in their abstract that “the role of neuropeptides such as substance P and vasoactive intestinal peptide has been highlighted in the synovium after trauma.”
An editorial in the Journal of Rheumatology also suggested areas for additional research. Dr. Ignazio Olivieri of San Carlo Hospital in Potenza, Italy, wrote that “criteria of imputability” that should be met include “single and significant trauma; absence of joint lesion before trauma; localization of arthritis in the area of trauma; and absence or short delay between trauma and onset of arthritis.”
“You might envision treating [psoriasis] patients early [for psoriatic arthritis] if they break a leg or get a joint dislocation, but we are not there yet,” Dr. Love stressed. “This is an idea of where we could take this and where we might actually be able to have an effect.”
The research was performed by researchers at the University of Iceland (Reykjavik) in collaboration with researchers at Harvard Medical School in Boston and the University of Pennsylvania in Philadelphia. It was partially funded by the Icelandic Centre for Research (RANNIS) and the National Institutes of Health. Dr. Love’s associate, Dr. Stefan Thorarensen of the division of public health at the University of Iceland, presented the findings during the clinical science session at the congress.
Dr. Love and his coauthors reported having no financial disclosures.
Key clinical point: Further research is needed before any clinical recommendations can be made based on these findings.
Major finding: Trauma, especially that involving the bone or joints, was associated with a higher risk for developing psoriatic arthritis (HR, 1.32).
Data source: A retrospective, population-based cohort study of 70,646 patients with psoriasis.
Disclosures: Dr. Love and coauthors reported having no financial disclosures.
WCD: Dermatologists underappreciate psoriatic itch, patients say
VANCOUVER, B.C. – The largest-ever survey of North American and European physician and patient perspectives on psoriasis and psoriatic arthritis reveals a major disconnect between dermatologists and their psoriasis patients regarding the importance of pruritus.
Dermatologists characterized one-fifth of their psoriasis patients as having severe disease. The majority of dermatologists – fully 53% – regarded lesion size and location to be the most important factors contributing to disease severity. Only 7.4% of dermatologists considered itch to be the most important factor.
In contrast, 38% of patients considered itching to be the most important aspect of their skin disease, and 17% – less than one-third of the proportion of dermatologists – rated lesion size and location to be the top contributor to disease severity, Dr. Peter van de Kerkhof reported at the World Congress of Dermatology.
He presented new findings from the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP), a survey of North American dermatologists, rheumatologists, psoriasis patients, and psoriatic arthritis patients unrivaled in its scope. The results of the first part of the survey, which entailed contacting 139,000 households and focused on unmet needs as characterized by 3,426 patients, have already been published (J. Am. Acad. Dermatol. 2014;70:871-81).
At the WCD, Dr. van de Kerkhof presented highlights of part 2 of the MAPP survey, which sought to obtain the real-world perspectives of dermatologists and rheumatologists on the management of psoriasis and psoriatic arthritis. Of the 6,530 dermatologists and 5,445 rheumatologists who were contacted, 391 and 390, respectively, completed the detailed interview, according to Dr. van de Kerkhof, professor and chairman of the department of dermatology at Radboud University in Nijmegen, the Netherlands.
Although 92% of dermatologists agreed that the disease burden of psoriasis is frequently underestimated, the marked physician/patient divergence regarding the importance of pruritus was particularly striking, he observed.
Nineteen percent of dermatologists reported that their patients with moderate to severe psoriasis were receiving conventional oral agents, and 19.6% indicated their patients were on biologic therapy. However, 54% of dermatologists indicated they prescribed topical agents as monotherapy for their patients with moderate to severe disease, which is out of step with current treatment guidelines and represents clear undertreatment, Dr. van de Kerkhof noted.
Thirty-six percent of dermatologists cited affordability and 22% cited uncertainty about long-term safety of current medications as the greatest challenge associated with management of psoriasis. Sixteen percent cited lack of effectiveness as the biggest challenge.
The top reasons dermatologists gave for not initiating treatment with conventional oral agents were concerns about long-term safety and tolerability. Those were also the main reasons dermatologists gave for not continuing conventional oral medications.
The story was slightly different when it came to reasons for not initiating or continuing biologic agents. Here cost joined tolerability and long-term safety concerns as the main reasons for not starting patients on biologic therapies. Those factors, along with lack or loss of response, which was cited by 18% of dermatologists, were also the chief reasons given for not continuing patients on biologic agents.
Dr. Gil Yosipovitch, who has devoted much of his research career to the study of itch, predicted that the sharp discrepancy between psoriasis patients and dermatologists regarding the importance of itch as documented in the new MAPP survey findings will improve soon. Dermatologists’ attitudes towards itch are beginning to change: “If you look at the textbooks of a decade ago, itch was not even mentioned as part of the symptoms,” he said.
Psoriasis patients clearly feel that effective treatment for their pruritus is an unmet need. Fortunately, studies on the itch impact of current and next-generation biologics are underway and in some cases completed, noted Dr. Yosipovitch, professor and chair of the department of dermatology at Temple University in Philadelphia.
The MAPP survey was sponsored by Celgene Corp. Dr. van de Kerkhof reported serving as a consultant to Celgene and a dozen other pharmaceutical companies, most of which also provide him with grants to conduct clinical trials.
VANCOUVER, B.C. – The largest-ever survey of North American and European physician and patient perspectives on psoriasis and psoriatic arthritis reveals a major disconnect between dermatologists and their psoriasis patients regarding the importance of pruritus.
Dermatologists characterized one-fifth of their psoriasis patients as having severe disease. The majority of dermatologists – fully 53% – regarded lesion size and location to be the most important factors contributing to disease severity. Only 7.4% of dermatologists considered itch to be the most important factor.
In contrast, 38% of patients considered itching to be the most important aspect of their skin disease, and 17% – less than one-third of the proportion of dermatologists – rated lesion size and location to be the top contributor to disease severity, Dr. Peter van de Kerkhof reported at the World Congress of Dermatology.
He presented new findings from the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP), a survey of North American dermatologists, rheumatologists, psoriasis patients, and psoriatic arthritis patients unrivaled in its scope. The results of the first part of the survey, which entailed contacting 139,000 households and focused on unmet needs as characterized by 3,426 patients, have already been published (J. Am. Acad. Dermatol. 2014;70:871-81).
At the WCD, Dr. van de Kerkhof presented highlights of part 2 of the MAPP survey, which sought to obtain the real-world perspectives of dermatologists and rheumatologists on the management of psoriasis and psoriatic arthritis. Of the 6,530 dermatologists and 5,445 rheumatologists who were contacted, 391 and 390, respectively, completed the detailed interview, according to Dr. van de Kerkhof, professor and chairman of the department of dermatology at Radboud University in Nijmegen, the Netherlands.
Although 92% of dermatologists agreed that the disease burden of psoriasis is frequently underestimated, the marked physician/patient divergence regarding the importance of pruritus was particularly striking, he observed.
Nineteen percent of dermatologists reported that their patients with moderate to severe psoriasis were receiving conventional oral agents, and 19.6% indicated their patients were on biologic therapy. However, 54% of dermatologists indicated they prescribed topical agents as monotherapy for their patients with moderate to severe disease, which is out of step with current treatment guidelines and represents clear undertreatment, Dr. van de Kerkhof noted.
Thirty-six percent of dermatologists cited affordability and 22% cited uncertainty about long-term safety of current medications as the greatest challenge associated with management of psoriasis. Sixteen percent cited lack of effectiveness as the biggest challenge.
The top reasons dermatologists gave for not initiating treatment with conventional oral agents were concerns about long-term safety and tolerability. Those were also the main reasons dermatologists gave for not continuing conventional oral medications.
The story was slightly different when it came to reasons for not initiating or continuing biologic agents. Here cost joined tolerability and long-term safety concerns as the main reasons for not starting patients on biologic therapies. Those factors, along with lack or loss of response, which was cited by 18% of dermatologists, were also the chief reasons given for not continuing patients on biologic agents.
Dr. Gil Yosipovitch, who has devoted much of his research career to the study of itch, predicted that the sharp discrepancy between psoriasis patients and dermatologists regarding the importance of itch as documented in the new MAPP survey findings will improve soon. Dermatologists’ attitudes towards itch are beginning to change: “If you look at the textbooks of a decade ago, itch was not even mentioned as part of the symptoms,” he said.
Psoriasis patients clearly feel that effective treatment for their pruritus is an unmet need. Fortunately, studies on the itch impact of current and next-generation biologics are underway and in some cases completed, noted Dr. Yosipovitch, professor and chair of the department of dermatology at Temple University in Philadelphia.
The MAPP survey was sponsored by Celgene Corp. Dr. van de Kerkhof reported serving as a consultant to Celgene and a dozen other pharmaceutical companies, most of which also provide him with grants to conduct clinical trials.
VANCOUVER, B.C. – The largest-ever survey of North American and European physician and patient perspectives on psoriasis and psoriatic arthritis reveals a major disconnect between dermatologists and their psoriasis patients regarding the importance of pruritus.
Dermatologists characterized one-fifth of their psoriasis patients as having severe disease. The majority of dermatologists – fully 53% – regarded lesion size and location to be the most important factors contributing to disease severity. Only 7.4% of dermatologists considered itch to be the most important factor.
In contrast, 38% of patients considered itching to be the most important aspect of their skin disease, and 17% – less than one-third of the proportion of dermatologists – rated lesion size and location to be the top contributor to disease severity, Dr. Peter van de Kerkhof reported at the World Congress of Dermatology.
He presented new findings from the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP), a survey of North American dermatologists, rheumatologists, psoriasis patients, and psoriatic arthritis patients unrivaled in its scope. The results of the first part of the survey, which entailed contacting 139,000 households and focused on unmet needs as characterized by 3,426 patients, have already been published (J. Am. Acad. Dermatol. 2014;70:871-81).
At the WCD, Dr. van de Kerkhof presented highlights of part 2 of the MAPP survey, which sought to obtain the real-world perspectives of dermatologists and rheumatologists on the management of psoriasis and psoriatic arthritis. Of the 6,530 dermatologists and 5,445 rheumatologists who were contacted, 391 and 390, respectively, completed the detailed interview, according to Dr. van de Kerkhof, professor and chairman of the department of dermatology at Radboud University in Nijmegen, the Netherlands.
Although 92% of dermatologists agreed that the disease burden of psoriasis is frequently underestimated, the marked physician/patient divergence regarding the importance of pruritus was particularly striking, he observed.
Nineteen percent of dermatologists reported that their patients with moderate to severe psoriasis were receiving conventional oral agents, and 19.6% indicated their patients were on biologic therapy. However, 54% of dermatologists indicated they prescribed topical agents as monotherapy for their patients with moderate to severe disease, which is out of step with current treatment guidelines and represents clear undertreatment, Dr. van de Kerkhof noted.
Thirty-six percent of dermatologists cited affordability and 22% cited uncertainty about long-term safety of current medications as the greatest challenge associated with management of psoriasis. Sixteen percent cited lack of effectiveness as the biggest challenge.
The top reasons dermatologists gave for not initiating treatment with conventional oral agents were concerns about long-term safety and tolerability. Those were also the main reasons dermatologists gave for not continuing conventional oral medications.
The story was slightly different when it came to reasons for not initiating or continuing biologic agents. Here cost joined tolerability and long-term safety concerns as the main reasons for not starting patients on biologic therapies. Those factors, along with lack or loss of response, which was cited by 18% of dermatologists, were also the chief reasons given for not continuing patients on biologic agents.
Dr. Gil Yosipovitch, who has devoted much of his research career to the study of itch, predicted that the sharp discrepancy between psoriasis patients and dermatologists regarding the importance of itch as documented in the new MAPP survey findings will improve soon. Dermatologists’ attitudes towards itch are beginning to change: “If you look at the textbooks of a decade ago, itch was not even mentioned as part of the symptoms,” he said.
Psoriasis patients clearly feel that effective treatment for their pruritus is an unmet need. Fortunately, studies on the itch impact of current and next-generation biologics are underway and in some cases completed, noted Dr. Yosipovitch, professor and chair of the department of dermatology at Temple University in Philadelphia.
The MAPP survey was sponsored by Celgene Corp. Dr. van de Kerkhof reported serving as a consultant to Celgene and a dozen other pharmaceutical companies, most of which also provide him with grants to conduct clinical trials.
AT WCD 2015
Key clinical point: Dermatologists and psoriasis patients don’t see eye to eye when it comes to the importance of itch as the defining symptom of disease severity.
Major finding: Thirty-eight percent of psoriasis patients consider itch to be the most troublesome aspect of their disease and the most important determinant of disease severity, as do 7.4% of dermatologists.
Data source: A random survey of 391 North American and European dermatologists, 390 rheumatologists, and more than 3,000 psoriasis or psoriatic arthritis patients.
Disclosures: The MAPP survey was sponsored by Celgene Corp. Dr. van de Kerkhof is a consultant to and recipient of research grants from Celgene and numerous other pharmaceutical companies.
WCD: Psoriasis plus depression magnifies MI risk
VANCOUVER, B.C. – The combination of even mild psoriasis and a diagnosis of depression is associated with a greater risk of acute MI than is either condition alone, according to a Danish national study.
Moreover, when depression coincided with a severe case of psoriasis, not only was the risk of incident acute MI elevated, but the risk that the MI would be fatal was significantly increased as well, Dr. Alexander Egeberg reported at the World Congress of Dermatology.
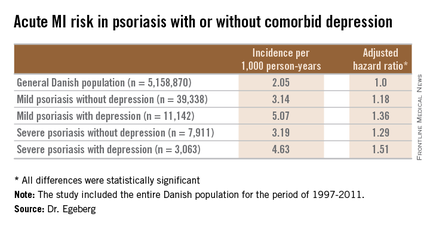
He presented a population-based cohort study of the entire Danish population, for the period of 1997-2011. The study population comprised 5,158,870 Danish citizens, including 50,480 Danes diagnosed with mild psoriasis and 10,974 others diagnosed with severe psoriasis during the study years. During that period, 11,142 members of the group with mild psoriasis and 3,063 with severe psoriasis were diagnosed with incident depression.
In a multivariate Cox regression analysis adjusted for age, gender, comorbid conditions, and concomitant medications, Danes with mild psoriasis but no diagnosis of depression were at a statistically significant 18% increased risk of having an acute MI compared to the general Danish population free of both diseases; the risk climbed upward from there (see chart).
A consistent trend for an increased risk of fatal MI was evident in patients with psoriasis, be it mild or severe, and with or without comorbid depression. However, this risk achieved statistical significance only in the group with severe psoriasis plus depression, a state associated with a 64% increased risk of fatal MI compared with the general population.
Although recent years have brought clear evidence that psoriasis is associated with increased risks of heart disease and depression, and that stand-alone depression is a risk factor for ischemic coronary events, the link between psoriasis, depression, and MI previously was unclear, explained Dr. Egeberg, who performed the research while at the University of Copenhagen but is now employed by Pfizer.
The retrospective study was conducted with the use of linked comprehensive national databases. The work was supported by a research grant from Pfizer.
VANCOUVER, B.C. – The combination of even mild psoriasis and a diagnosis of depression is associated with a greater risk of acute MI than is either condition alone, according to a Danish national study.
Moreover, when depression coincided with a severe case of psoriasis, not only was the risk of incident acute MI elevated, but the risk that the MI would be fatal was significantly increased as well, Dr. Alexander Egeberg reported at the World Congress of Dermatology.

He presented a population-based cohort study of the entire Danish population, for the period of 1997-2011. The study population comprised 5,158,870 Danish citizens, including 50,480 Danes diagnosed with mild psoriasis and 10,974 others diagnosed with severe psoriasis during the study years. During that period, 11,142 members of the group with mild psoriasis and 3,063 with severe psoriasis were diagnosed with incident depression.
In a multivariate Cox regression analysis adjusted for age, gender, comorbid conditions, and concomitant medications, Danes with mild psoriasis but no diagnosis of depression were at a statistically significant 18% increased risk of having an acute MI compared to the general Danish population free of both diseases; the risk climbed upward from there (see chart).
A consistent trend for an increased risk of fatal MI was evident in patients with psoriasis, be it mild or severe, and with or without comorbid depression. However, this risk achieved statistical significance only in the group with severe psoriasis plus depression, a state associated with a 64% increased risk of fatal MI compared with the general population.
Although recent years have brought clear evidence that psoriasis is associated with increased risks of heart disease and depression, and that stand-alone depression is a risk factor for ischemic coronary events, the link between psoriasis, depression, and MI previously was unclear, explained Dr. Egeberg, who performed the research while at the University of Copenhagen but is now employed by Pfizer.
The retrospective study was conducted with the use of linked comprehensive national databases. The work was supported by a research grant from Pfizer.
VANCOUVER, B.C. – The combination of even mild psoriasis and a diagnosis of depression is associated with a greater risk of acute MI than is either condition alone, according to a Danish national study.
Moreover, when depression coincided with a severe case of psoriasis, not only was the risk of incident acute MI elevated, but the risk that the MI would be fatal was significantly increased as well, Dr. Alexander Egeberg reported at the World Congress of Dermatology.

He presented a population-based cohort study of the entire Danish population, for the period of 1997-2011. The study population comprised 5,158,870 Danish citizens, including 50,480 Danes diagnosed with mild psoriasis and 10,974 others diagnosed with severe psoriasis during the study years. During that period, 11,142 members of the group with mild psoriasis and 3,063 with severe psoriasis were diagnosed with incident depression.
In a multivariate Cox regression analysis adjusted for age, gender, comorbid conditions, and concomitant medications, Danes with mild psoriasis but no diagnosis of depression were at a statistically significant 18% increased risk of having an acute MI compared to the general Danish population free of both diseases; the risk climbed upward from there (see chart).
A consistent trend for an increased risk of fatal MI was evident in patients with psoriasis, be it mild or severe, and with or without comorbid depression. However, this risk achieved statistical significance only in the group with severe psoriasis plus depression, a state associated with a 64% increased risk of fatal MI compared with the general population.
Although recent years have brought clear evidence that psoriasis is associated with increased risks of heart disease and depression, and that stand-alone depression is a risk factor for ischemic coronary events, the link between psoriasis, depression, and MI previously was unclear, explained Dr. Egeberg, who performed the research while at the University of Copenhagen but is now employed by Pfizer.
The retrospective study was conducted with the use of linked comprehensive national databases. The work was supported by a research grant from Pfizer.
AT WCD 2015
Key clinical point: In psoriasis patients, comorbid depression is associated with an increased risk of a first acute MI.
Major finding: The incidence rate of acute MI was 2.05 cases per 1,000 person-years in the general Danish population, climbing to 3.14/1,000 in those with mild psoriasis without diagnosed depression, and 5.07/1,000 for mild psoriasis with depression.
Data source: A retrospective, population-based, cohort study of the entire Danish adult population – more than 5.1 million individuals – during 1997-2011.
Disclosures: The presenter is an employee of Pfizer, which supported the study.
How Common Is Psoriasis in Patients With Skin of Color?
The prevalence of psoriasis in individuals with skin of color is lower than in white individuals, but a number of clinical differences can complicate diagnosis and treatment of psoriasis in patients with darker skin types. Dr. Andrew Alexis discusses some clinical features that may be unique to those with skin of color, including association with persistent pigmentary alterations, as well as special considerations and treatment approaches for psoriasis in darker-skinned patients.
The psoriasis audiocast series is created in collaboration with Cutis® and the National Psoriasis Foundation®.
Suggested Reading
Report on the psycho-social impacts of psoriasis. National Psoriasis Foundation Web site. Published in 2009. https://www.psoriasis.org/Document.DOC?id=619. Accessed June 15, 2015.
The prevalence of psoriasis in individuals with skin of color is lower than in white individuals, but a number of clinical differences can complicate diagnosis and treatment of psoriasis in patients with darker skin types. Dr. Andrew Alexis discusses some clinical features that may be unique to those with skin of color, including association with persistent pigmentary alterations, as well as special considerations and treatment approaches for psoriasis in darker-skinned patients.
The psoriasis audiocast series is created in collaboration with Cutis® and the National Psoriasis Foundation®.
The prevalence of psoriasis in individuals with skin of color is lower than in white individuals, but a number of clinical differences can complicate diagnosis and treatment of psoriasis in patients with darker skin types. Dr. Andrew Alexis discusses some clinical features that may be unique to those with skin of color, including association with persistent pigmentary alterations, as well as special considerations and treatment approaches for psoriasis in darker-skinned patients.
The psoriasis audiocast series is created in collaboration with Cutis® and the National Psoriasis Foundation®.
Suggested Reading
Report on the psycho-social impacts of psoriasis. National Psoriasis Foundation Web site. Published in 2009. https://www.psoriasis.org/Document.DOC?id=619. Accessed June 15, 2015.
Suggested Reading
Report on the psycho-social impacts of psoriasis. National Psoriasis Foundation Web site. Published in 2009. https://www.psoriasis.org/Document.DOC?id=619. Accessed June 15, 2015.
EULAR: Epigenetics points to anti-TNF efficacy in psoriatic arthritis
ROME – Tumor necrosis factor–alpha (TNF-alpha) inhibitors are well established as a treatment for psoriatic arthritis patients, but efficacy can vary greatly within that population. New work from Canadian researchers demonstrates that the global DNA methylation pattern differs between TNF-alpha–inhibitor responders and secondary failures, which could help explain the variability in medication success rates.
“Although TNF inhibitors [TNFi] work very well in certain individuals with psoriatic arthritis [PsA], up to 40% of individuals receiving this treatment fail to achieve a therapeutic response, and 20%-50% of individuals who have an initial response to treatment become refractory weeks or months after receiving therapy.” lead study author Darren O’Rielly, Ph.D., said at the European Congress of Rheumatology.
The preliminary study findings eventually could lead to the identification of biomarkers that would indicate patients who are not good candidates for TNFi medications, said Dr. O’Rielly of Memorial University, St. John’s, Nfld.
Given recent advances in epigenetics and that the epigenetic signature is affected by environmental factors, the investigators set out to determine if methylation alterations could help explain why PsA patients respond or fail with TNFi. The researchers performed genome-wide DNA methylation profiling on blood samples from 41 PsA patients, using machinery that measures about 480,000 CpG sites per sample and covers 96% of RefSeq genes. A total of 21 patients were considered TNFi responders, of whom 13 were treated with etanercept and 8 with adalimumab; median follow-up duration was 18 months. Twenty patients were considered secondary TNFi failures, of whom 15 were treated with etanercept and 5 with adalimumab; median follow-up duration was 36 months.
Dr. O’Rielly, a senior research scientist at Memorial and director of the Molecular Genetics Laboratory at Eastern Health in St. John’s, and his associates measured the methylation level at CpG sites using a genome-wide approach and selected regions of interest based on functional relevance to TNF-mediated signaling pathways with methylation level differences of 5% or greater and an adjusted P-value less than .05.
After quality-control filtering, investigators evaluated 384,599 CpG sites for TNFi responders and 368,863 CpG sites for TNFi failures. Researchers found 72 CpG sites of interest in the TNFi responder group and 91 CpG sites of interest in the TNFi failure group. Top candidate genes for TNFi responders included TRAPPC9 (which functions as an activator of NF-kB), CCR6 (which regulates the migration and recruitment of dendritic and T cells), and PSORS1C3 (psoriasis susceptibility 1 candidate 3), while top candidate genes for TNFi secondary failures included CD70 (an encoded protein that is a ligand for TNFRSF27/CD27) and TNFRSF1B (a member of the TNF receptor ‘superfamily’ that mediates most of the metabolic effects of TNF-alpha).
“We are very encouraged by these findings,” Dr. O’Rielly said. “We were a little surprised that several of our best candidate genes, such as TNFRSF1B and CD70, appear to play a role in TNF-alpha signaling, and that their methylation change occurs in a gene region that is consistent with a possible functional effect. We were expecting to find some methylation changes in genes but not necessarily in pathways with a direct connection with TNF-alpha–mediated signaling.”
The group plans to confirm these findings for the best candidate genes using other methylation-specific polymerase chain reaction technology, he said, and will investigate additional CpG sites adjacent to the region of interest, including promoters and enhancers, in the best candidate genes to better characterize the full extent of methylation changes in these regions. They also would like to replicate the findings in a prospective, independent cohort.
Dr. O’Rielly reported no relevant financial conflicts.
ROME – Tumor necrosis factor–alpha (TNF-alpha) inhibitors are well established as a treatment for psoriatic arthritis patients, but efficacy can vary greatly within that population. New work from Canadian researchers demonstrates that the global DNA methylation pattern differs between TNF-alpha–inhibitor responders and secondary failures, which could help explain the variability in medication success rates.
“Although TNF inhibitors [TNFi] work very well in certain individuals with psoriatic arthritis [PsA], up to 40% of individuals receiving this treatment fail to achieve a therapeutic response, and 20%-50% of individuals who have an initial response to treatment become refractory weeks or months after receiving therapy.” lead study author Darren O’Rielly, Ph.D., said at the European Congress of Rheumatology.
The preliminary study findings eventually could lead to the identification of biomarkers that would indicate patients who are not good candidates for TNFi medications, said Dr. O’Rielly of Memorial University, St. John’s, Nfld.
Given recent advances in epigenetics and that the epigenetic signature is affected by environmental factors, the investigators set out to determine if methylation alterations could help explain why PsA patients respond or fail with TNFi. The researchers performed genome-wide DNA methylation profiling on blood samples from 41 PsA patients, using machinery that measures about 480,000 CpG sites per sample and covers 96% of RefSeq genes. A total of 21 patients were considered TNFi responders, of whom 13 were treated with etanercept and 8 with adalimumab; median follow-up duration was 18 months. Twenty patients were considered secondary TNFi failures, of whom 15 were treated with etanercept and 5 with adalimumab; median follow-up duration was 36 months.
Dr. O’Rielly, a senior research scientist at Memorial and director of the Molecular Genetics Laboratory at Eastern Health in St. John’s, and his associates measured the methylation level at CpG sites using a genome-wide approach and selected regions of interest based on functional relevance to TNF-mediated signaling pathways with methylation level differences of 5% or greater and an adjusted P-value less than .05.
After quality-control filtering, investigators evaluated 384,599 CpG sites for TNFi responders and 368,863 CpG sites for TNFi failures. Researchers found 72 CpG sites of interest in the TNFi responder group and 91 CpG sites of interest in the TNFi failure group. Top candidate genes for TNFi responders included TRAPPC9 (which functions as an activator of NF-kB), CCR6 (which regulates the migration and recruitment of dendritic and T cells), and PSORS1C3 (psoriasis susceptibility 1 candidate 3), while top candidate genes for TNFi secondary failures included CD70 (an encoded protein that is a ligand for TNFRSF27/CD27) and TNFRSF1B (a member of the TNF receptor ‘superfamily’ that mediates most of the metabolic effects of TNF-alpha).
“We are very encouraged by these findings,” Dr. O’Rielly said. “We were a little surprised that several of our best candidate genes, such as TNFRSF1B and CD70, appear to play a role in TNF-alpha signaling, and that their methylation change occurs in a gene region that is consistent with a possible functional effect. We were expecting to find some methylation changes in genes but not necessarily in pathways with a direct connection with TNF-alpha–mediated signaling.”
The group plans to confirm these findings for the best candidate genes using other methylation-specific polymerase chain reaction technology, he said, and will investigate additional CpG sites adjacent to the region of interest, including promoters and enhancers, in the best candidate genes to better characterize the full extent of methylation changes in these regions. They also would like to replicate the findings in a prospective, independent cohort.
Dr. O’Rielly reported no relevant financial conflicts.
ROME – Tumor necrosis factor–alpha (TNF-alpha) inhibitors are well established as a treatment for psoriatic arthritis patients, but efficacy can vary greatly within that population. New work from Canadian researchers demonstrates that the global DNA methylation pattern differs between TNF-alpha–inhibitor responders and secondary failures, which could help explain the variability in medication success rates.
“Although TNF inhibitors [TNFi] work very well in certain individuals with psoriatic arthritis [PsA], up to 40% of individuals receiving this treatment fail to achieve a therapeutic response, and 20%-50% of individuals who have an initial response to treatment become refractory weeks or months after receiving therapy.” lead study author Darren O’Rielly, Ph.D., said at the European Congress of Rheumatology.
The preliminary study findings eventually could lead to the identification of biomarkers that would indicate patients who are not good candidates for TNFi medications, said Dr. O’Rielly of Memorial University, St. John’s, Nfld.
Given recent advances in epigenetics and that the epigenetic signature is affected by environmental factors, the investigators set out to determine if methylation alterations could help explain why PsA patients respond or fail with TNFi. The researchers performed genome-wide DNA methylation profiling on blood samples from 41 PsA patients, using machinery that measures about 480,000 CpG sites per sample and covers 96% of RefSeq genes. A total of 21 patients were considered TNFi responders, of whom 13 were treated with etanercept and 8 with adalimumab; median follow-up duration was 18 months. Twenty patients were considered secondary TNFi failures, of whom 15 were treated with etanercept and 5 with adalimumab; median follow-up duration was 36 months.
Dr. O’Rielly, a senior research scientist at Memorial and director of the Molecular Genetics Laboratory at Eastern Health in St. John’s, and his associates measured the methylation level at CpG sites using a genome-wide approach and selected regions of interest based on functional relevance to TNF-mediated signaling pathways with methylation level differences of 5% or greater and an adjusted P-value less than .05.
After quality-control filtering, investigators evaluated 384,599 CpG sites for TNFi responders and 368,863 CpG sites for TNFi failures. Researchers found 72 CpG sites of interest in the TNFi responder group and 91 CpG sites of interest in the TNFi failure group. Top candidate genes for TNFi responders included TRAPPC9 (which functions as an activator of NF-kB), CCR6 (which regulates the migration and recruitment of dendritic and T cells), and PSORS1C3 (psoriasis susceptibility 1 candidate 3), while top candidate genes for TNFi secondary failures included CD70 (an encoded protein that is a ligand for TNFRSF27/CD27) and TNFRSF1B (a member of the TNF receptor ‘superfamily’ that mediates most of the metabolic effects of TNF-alpha).
“We are very encouraged by these findings,” Dr. O’Rielly said. “We were a little surprised that several of our best candidate genes, such as TNFRSF1B and CD70, appear to play a role in TNF-alpha signaling, and that their methylation change occurs in a gene region that is consistent with a possible functional effect. We were expecting to find some methylation changes in genes but not necessarily in pathways with a direct connection with TNF-alpha–mediated signaling.”
The group plans to confirm these findings for the best candidate genes using other methylation-specific polymerase chain reaction technology, he said, and will investigate additional CpG sites adjacent to the region of interest, including promoters and enhancers, in the best candidate genes to better characterize the full extent of methylation changes in these regions. They also would like to replicate the findings in a prospective, independent cohort.
Dr. O’Rielly reported no relevant financial conflicts.
AT THE EULAR 2015 CONGRESS
Key clinical point:If preliminary findings are confirmed, methylation level profiling could lead to the identification of patients who are not good candidates for TNFi medications.
Major finding: Nonresponders to TNF inhibitors had significant methylation pattern differences in two key genes that appear to play a role in TNF-alpha signaling, TNFRSF1B and CD70.
Data source: A study of psoriatic arthritis patients comparing 21 TNFi responders and 20 nonresponders.
Disclosures: Dr. O’Rielly reported no relevant financial conflicts.