User login
Almost Half of Psoriasis Patients Have Metabolic Syndrome
Forty percent of patients with psoriasis were found to have the metabolic syndrome in a study of a nationally representative sample of more than 6,500 adults published online Dec. 20 in the Archives of Dermatology.
The prevalence of the metabolic syndrome was approximately twice as high in psoriasis patients as in adults without psoriasis, even after adjustment for potential confounders such as age, sex, race/ethnicity, smoking status, and CRP levels. "Given its associated serious complications, this comorbidity needs to be recognized and taken into account when treating individuals with psoriasis," reported Dr. Thorvardur Jon Love of Brigham and Women’s Hospital, Boston, and his associates.
"Based on these data, it is estimated that of the 6.6 million adults (age range 20-59 years) with psoriasis in the United States, 2.7 million have the metabolic syndrome, or nearly a million more individuals than would be expected from individuals without psoriasis," they noted.
The study findings may partially explain why previous research has found increased risks of cardiovascular and metabolic morbidity and mortality among psoriasis patients. In particular, patients with severe psoriasis have been reported to be at an increased risk for MI, stroke, and cardiovascular mortality, and have been reported to die 3-4 years earlier than people without psoriasis, the investigators added.
The metabolic syndrome is characterized by abdominal obesity, hypertriglyceridemia, low HDL cholesterol levels, hypertension, and high fasting glucose levels. Recent research has pegged the incidence of the disorder at 15%-24% in the general U.S. population.
Noting that there has been only one previous study examining the relationship between psoriasis and the metabolic syndrome, and that until now there have been no population-based data on the issue, Dr. Love and his colleagues used data from the National Health and Nutrition Examination Survey (NHANES) 2003-2006 to assess the prevalence of the metabolic syndrome and its components in psoriasis.
The study subjects included 2,456 adults (mean age 39 years) who did not have preexisting diabetes; 71 had psoriasis.
The prevalence of the metabolic syndrome was 40% among psoriasis patients, compared with 23% among patients without psoriasis. The odds ratio for patients with psoriasis to have the metabolic syndrome was 2.16 on univariate analysis and 1.96 after the data were adjusted to account for potential confounding factors.
When CRP levels were removed from the analysis to cancel out the potentially confounding effect of inflammation, odds ratios did not change materially, Dr. Love and his associates reported (Arch. Dermatol. 2010 Dec. 20 [doi:10.1001/archdermatol.2010.370]).
"When we applied these data to the 2008 U.S. census population estimate, 6.6 million (95% CI, 4.8-8.3) individuals aged 20-59 years were estimated to have psoriasis in the United States, and 2.7 million (95% CI, 1.6-3.6) of these individuals with psoriasis were estimated to have the metabolic syndrome, an excess of 1 million patients compared with the expected value among individuals without psoriasis," they noted.
The most common feature of the metabolic syndrome to be found among psoriasis patients was abdominal obesity, present in 63%. Hypertriglyceridemia and low HDL-cholesterol levels also were common.
These findings indicate that "a diagnosis of psoriasis should trigger a high clinical suspicion [of] and investigation for a potential coexistence of the metabolic syndrome. If present, the syndrome needs to be recognized as a potentially more life-threatening factor than psoriasis given the serious associated complications," Dr. Love and his colleagues wrote.
This association also should be considered when choosing therapy for psoriasis. "For example, tumor necrosis factor blockers may decrease insulin resistance," they added.
This study was supported in part by the National Institutes of Health, the Psoriasis Foundation, and the National Heart, Lung, and Blood Institute. One of Dr. Love's associates reported ties to Amgen, Abbott, Celgene, Centocor, Pfizer, and Novartis.
Forty percent of patients with psoriasis were found to have the metabolic syndrome in a study of a nationally representative sample of more than 6,500 adults published online Dec. 20 in the Archives of Dermatology.
The prevalence of the metabolic syndrome was approximately twice as high in psoriasis patients as in adults without psoriasis, even after adjustment for potential confounders such as age, sex, race/ethnicity, smoking status, and CRP levels. "Given its associated serious complications, this comorbidity needs to be recognized and taken into account when treating individuals with psoriasis," reported Dr. Thorvardur Jon Love of Brigham and Women’s Hospital, Boston, and his associates.
"Based on these data, it is estimated that of the 6.6 million adults (age range 20-59 years) with psoriasis in the United States, 2.7 million have the metabolic syndrome, or nearly a million more individuals than would be expected from individuals without psoriasis," they noted.
The study findings may partially explain why previous research has found increased risks of cardiovascular and metabolic morbidity and mortality among psoriasis patients. In particular, patients with severe psoriasis have been reported to be at an increased risk for MI, stroke, and cardiovascular mortality, and have been reported to die 3-4 years earlier than people without psoriasis, the investigators added.
The metabolic syndrome is characterized by abdominal obesity, hypertriglyceridemia, low HDL cholesterol levels, hypertension, and high fasting glucose levels. Recent research has pegged the incidence of the disorder at 15%-24% in the general U.S. population.
Noting that there has been only one previous study examining the relationship between psoriasis and the metabolic syndrome, and that until now there have been no population-based data on the issue, Dr. Love and his colleagues used data from the National Health and Nutrition Examination Survey (NHANES) 2003-2006 to assess the prevalence of the metabolic syndrome and its components in psoriasis.
The study subjects included 2,456 adults (mean age 39 years) who did not have preexisting diabetes; 71 had psoriasis.
The prevalence of the metabolic syndrome was 40% among psoriasis patients, compared with 23% among patients without psoriasis. The odds ratio for patients with psoriasis to have the metabolic syndrome was 2.16 on univariate analysis and 1.96 after the data were adjusted to account for potential confounding factors.
When CRP levels were removed from the analysis to cancel out the potentially confounding effect of inflammation, odds ratios did not change materially, Dr. Love and his associates reported (Arch. Dermatol. 2010 Dec. 20 [doi:10.1001/archdermatol.2010.370]).
"When we applied these data to the 2008 U.S. census population estimate, 6.6 million (95% CI, 4.8-8.3) individuals aged 20-59 years were estimated to have psoriasis in the United States, and 2.7 million (95% CI, 1.6-3.6) of these individuals with psoriasis were estimated to have the metabolic syndrome, an excess of 1 million patients compared with the expected value among individuals without psoriasis," they noted.
The most common feature of the metabolic syndrome to be found among psoriasis patients was abdominal obesity, present in 63%. Hypertriglyceridemia and low HDL-cholesterol levels also were common.
These findings indicate that "a diagnosis of psoriasis should trigger a high clinical suspicion [of] and investigation for a potential coexistence of the metabolic syndrome. If present, the syndrome needs to be recognized as a potentially more life-threatening factor than psoriasis given the serious associated complications," Dr. Love and his colleagues wrote.
This association also should be considered when choosing therapy for psoriasis. "For example, tumor necrosis factor blockers may decrease insulin resistance," they added.
This study was supported in part by the National Institutes of Health, the Psoriasis Foundation, and the National Heart, Lung, and Blood Institute. One of Dr. Love's associates reported ties to Amgen, Abbott, Celgene, Centocor, Pfizer, and Novartis.
Forty percent of patients with psoriasis were found to have the metabolic syndrome in a study of a nationally representative sample of more than 6,500 adults published online Dec. 20 in the Archives of Dermatology.
The prevalence of the metabolic syndrome was approximately twice as high in psoriasis patients as in adults without psoriasis, even after adjustment for potential confounders such as age, sex, race/ethnicity, smoking status, and CRP levels. "Given its associated serious complications, this comorbidity needs to be recognized and taken into account when treating individuals with psoriasis," reported Dr. Thorvardur Jon Love of Brigham and Women’s Hospital, Boston, and his associates.
"Based on these data, it is estimated that of the 6.6 million adults (age range 20-59 years) with psoriasis in the United States, 2.7 million have the metabolic syndrome, or nearly a million more individuals than would be expected from individuals without psoriasis," they noted.
The study findings may partially explain why previous research has found increased risks of cardiovascular and metabolic morbidity and mortality among psoriasis patients. In particular, patients with severe psoriasis have been reported to be at an increased risk for MI, stroke, and cardiovascular mortality, and have been reported to die 3-4 years earlier than people without psoriasis, the investigators added.
The metabolic syndrome is characterized by abdominal obesity, hypertriglyceridemia, low HDL cholesterol levels, hypertension, and high fasting glucose levels. Recent research has pegged the incidence of the disorder at 15%-24% in the general U.S. population.
Noting that there has been only one previous study examining the relationship between psoriasis and the metabolic syndrome, and that until now there have been no population-based data on the issue, Dr. Love and his colleagues used data from the National Health and Nutrition Examination Survey (NHANES) 2003-2006 to assess the prevalence of the metabolic syndrome and its components in psoriasis.
The study subjects included 2,456 adults (mean age 39 years) who did not have preexisting diabetes; 71 had psoriasis.
The prevalence of the metabolic syndrome was 40% among psoriasis patients, compared with 23% among patients without psoriasis. The odds ratio for patients with psoriasis to have the metabolic syndrome was 2.16 on univariate analysis and 1.96 after the data were adjusted to account for potential confounding factors.
When CRP levels were removed from the analysis to cancel out the potentially confounding effect of inflammation, odds ratios did not change materially, Dr. Love and his associates reported (Arch. Dermatol. 2010 Dec. 20 [doi:10.1001/archdermatol.2010.370]).
"When we applied these data to the 2008 U.S. census population estimate, 6.6 million (95% CI, 4.8-8.3) individuals aged 20-59 years were estimated to have psoriasis in the United States, and 2.7 million (95% CI, 1.6-3.6) of these individuals with psoriasis were estimated to have the metabolic syndrome, an excess of 1 million patients compared with the expected value among individuals without psoriasis," they noted.
The most common feature of the metabolic syndrome to be found among psoriasis patients was abdominal obesity, present in 63%. Hypertriglyceridemia and low HDL-cholesterol levels also were common.
These findings indicate that "a diagnosis of psoriasis should trigger a high clinical suspicion [of] and investigation for a potential coexistence of the metabolic syndrome. If present, the syndrome needs to be recognized as a potentially more life-threatening factor than psoriasis given the serious associated complications," Dr. Love and his colleagues wrote.
This association also should be considered when choosing therapy for psoriasis. "For example, tumor necrosis factor blockers may decrease insulin resistance," they added.
This study was supported in part by the National Institutes of Health, the Psoriasis Foundation, and the National Heart, Lung, and Blood Institute. One of Dr. Love's associates reported ties to Amgen, Abbott, Celgene, Centocor, Pfizer, and Novartis.
FROM ARCHIVES OF DERMATOLOGY
Major Finding: Of psoriasis patients, 40% have the metabolic syndrome, approximately double the rate found in adults who do not have psoriasis.
Data Source: Analysis of data from a cross-sectional health survey of a nationally representative random sample of 6,549 U.S. adults.
Disclosures: This study was supported in part by the National Institutes of Health, the Psoriasis Foundation, and the National Heart, Lung, and Blood Institute. One of Dr. Love's associates reported ties to Amgen, Abbott, Celgene, Centocor, Pfizer, and Novartis.
TNF Inhibitor Users Report Less Sick Leave
A significant 30% reduction in the number of sick leave days used per month was seen in adults with rheumatoid arthritis after using tumor necrosis factor antagonists for 6 months.
The finding was observed in a population-based study of 365 RA patients aged 18-85 years.
The study is among the first to address the quantitative impact of TNF inhibitors on sick leave and disability pension, said Dr. Tor Olofsson of Lund (Sweden) University, and colleagues.
They reviewed insurance database information on RA patients enrolled in the South Swedish Arthritis Treatment Group registry. Each patient was matched with four controls from the general population.
The study population averaged 9 sick days per month in the first month of anti-TNF treatment. The monthly rate dropped to an average of 6.5 days after 6 months and remained steady at an average of 6.6 days per month for months 6-12 (Ann. Rheum. Dis. 2010;69:2131-6).
Compared with the controls in the general population, the relative risk of being on sick leave in the RA group was 6.6 at the start of treatment, but dropped to 5.1 after 6 months, and remained at an average of 5.2 for the rest of the year. The relative risk of being on disability pension was 3.4 at the start of treatment and 3.2 after 1 year of treatment.
Approximately 98% of the patients had tried at least one disease-modifying antirheumatic drug (DMARD) before starting anti-TNF therapy. The average age of the patients was 46 years, and 82% were women. A total of 92 patients (25%) discontinued treatment, including 34 for adverse events, 32 for treatment failure, and 26 for other reasons.
"The reduction in sick leave during the first anti-TNF treatment year must be considered as a conservative measure in this cohort of patients who were severely ill, since there was a steady increase in sick leave prior to initiation of therapy," the researchers wrote. "Even a stabilization of the sick leave level could have been deemed as a successful outcome," they noted.
The results support findings from previous studies of the effects of TNF inhibitors on work outcomes, although the study was limited by the short follow-up period, the researchers noted. The findings conflict with data from a recent observational study that showed no difference in employment loss between RA patients using anti-TNF therapy and controls.
However, the average duration of RA in the observational study was 12 years, compared to an average duration of 4.5 years in the current study. This difference suggests a possible protective effect from anti-TNF therapy in a subset of patients with less than 11 years disease duration at the start of therapy, the researchers said.
The researchers said that they had no relevant financial conflicts to disclose. The study was funded by grants from multiple nonpharmaceutical organizations including the Swedish Research Council, Lund University, Region Skåne, the Swedish Social Insurance Agency, the Swedish Rheumatism Association, and the King Gustav V 80-Year Fund.
A significant 30% reduction in the number of sick leave days used per month was seen in adults with rheumatoid arthritis after using tumor necrosis factor antagonists for 6 months.
The finding was observed in a population-based study of 365 RA patients aged 18-85 years.
The study is among the first to address the quantitative impact of TNF inhibitors on sick leave and disability pension, said Dr. Tor Olofsson of Lund (Sweden) University, and colleagues.
They reviewed insurance database information on RA patients enrolled in the South Swedish Arthritis Treatment Group registry. Each patient was matched with four controls from the general population.
The study population averaged 9 sick days per month in the first month of anti-TNF treatment. The monthly rate dropped to an average of 6.5 days after 6 months and remained steady at an average of 6.6 days per month for months 6-12 (Ann. Rheum. Dis. 2010;69:2131-6).
Compared with the controls in the general population, the relative risk of being on sick leave in the RA group was 6.6 at the start of treatment, but dropped to 5.1 after 6 months, and remained at an average of 5.2 for the rest of the year. The relative risk of being on disability pension was 3.4 at the start of treatment and 3.2 after 1 year of treatment.
Approximately 98% of the patients had tried at least one disease-modifying antirheumatic drug (DMARD) before starting anti-TNF therapy. The average age of the patients was 46 years, and 82% were women. A total of 92 patients (25%) discontinued treatment, including 34 for adverse events, 32 for treatment failure, and 26 for other reasons.
"The reduction in sick leave during the first anti-TNF treatment year must be considered as a conservative measure in this cohort of patients who were severely ill, since there was a steady increase in sick leave prior to initiation of therapy," the researchers wrote. "Even a stabilization of the sick leave level could have been deemed as a successful outcome," they noted.
The results support findings from previous studies of the effects of TNF inhibitors on work outcomes, although the study was limited by the short follow-up period, the researchers noted. The findings conflict with data from a recent observational study that showed no difference in employment loss between RA patients using anti-TNF therapy and controls.
However, the average duration of RA in the observational study was 12 years, compared to an average duration of 4.5 years in the current study. This difference suggests a possible protective effect from anti-TNF therapy in a subset of patients with less than 11 years disease duration at the start of therapy, the researchers said.
The researchers said that they had no relevant financial conflicts to disclose. The study was funded by grants from multiple nonpharmaceutical organizations including the Swedish Research Council, Lund University, Region Skåne, the Swedish Social Insurance Agency, the Swedish Rheumatism Association, and the King Gustav V 80-Year Fund.
A significant 30% reduction in the number of sick leave days used per month was seen in adults with rheumatoid arthritis after using tumor necrosis factor antagonists for 6 months.
The finding was observed in a population-based study of 365 RA patients aged 18-85 years.
The study is among the first to address the quantitative impact of TNF inhibitors on sick leave and disability pension, said Dr. Tor Olofsson of Lund (Sweden) University, and colleagues.
They reviewed insurance database information on RA patients enrolled in the South Swedish Arthritis Treatment Group registry. Each patient was matched with four controls from the general population.
The study population averaged 9 sick days per month in the first month of anti-TNF treatment. The monthly rate dropped to an average of 6.5 days after 6 months and remained steady at an average of 6.6 days per month for months 6-12 (Ann. Rheum. Dis. 2010;69:2131-6).
Compared with the controls in the general population, the relative risk of being on sick leave in the RA group was 6.6 at the start of treatment, but dropped to 5.1 after 6 months, and remained at an average of 5.2 for the rest of the year. The relative risk of being on disability pension was 3.4 at the start of treatment and 3.2 after 1 year of treatment.
Approximately 98% of the patients had tried at least one disease-modifying antirheumatic drug (DMARD) before starting anti-TNF therapy. The average age of the patients was 46 years, and 82% were women. A total of 92 patients (25%) discontinued treatment, including 34 for adverse events, 32 for treatment failure, and 26 for other reasons.
"The reduction in sick leave during the first anti-TNF treatment year must be considered as a conservative measure in this cohort of patients who were severely ill, since there was a steady increase in sick leave prior to initiation of therapy," the researchers wrote. "Even a stabilization of the sick leave level could have been deemed as a successful outcome," they noted.
The results support findings from previous studies of the effects of TNF inhibitors on work outcomes, although the study was limited by the short follow-up period, the researchers noted. The findings conflict with data from a recent observational study that showed no difference in employment loss between RA patients using anti-TNF therapy and controls.
However, the average duration of RA in the observational study was 12 years, compared to an average duration of 4.5 years in the current study. This difference suggests a possible protective effect from anti-TNF therapy in a subset of patients with less than 11 years disease duration at the start of therapy, the researchers said.
The researchers said that they had no relevant financial conflicts to disclose. The study was funded by grants from multiple nonpharmaceutical organizations including the Swedish Research Council, Lund University, Region Skåne, the Swedish Social Insurance Agency, the Swedish Rheumatism Association, and the King Gustav V 80-Year Fund.
FROM ANNALS OF THE RHEUMATIC DISEASES
Major Finding: TNF-inhibitor users reduced the average sick leave time from 9.8 days per month at the start of treatment to 6.5 days after 6 months of treatment.
Data Source: A study of 365 Swedish adults aged 18-58 years with RA.
Disclosures: The researchers said that they had no relevant financial disclosures.
Briakinumab, Ustekinumab Trials Point to Future Psoriasis Treatment
Increased insight into the pathogenesis of psoriasis has led to the development of new drugs with novel mechanisms of action, as well as safer and more effective approaches to treatment with conventional drugs
As a result, treatment options for the management of patients with moderate to severe psoriasis in particular – nearly 25% of all patients with psoriasis, according to the National Psoriasis Foundation – have broadened substantially, and research reported during the past year suggests they are on track for continued expansion.
Among the most notable trials of the year were establishing the "unsurpassed efficacy" of Abbott’s investigational monoclonal antibody briakinumab, according to Dr. Craig Leonardi, a dermatologist and psoriasis specialist at St. Louis University. Like the recently approved psoriatic drug ustekinumab (Stelara), briakinumab is an injectable biologic agent that targets the interleukin-12 and -23 (IL-12/23) proteins, which are believed to promote the inflammation associated with psoriasis.
IL-12/23 Inhibition
Four phase III clinical studies of briakinumab were presented at the annual meeting of the European Academy of Dermatology and Venereology (EADV) in Gothenburg, Sweden.
– M06-890. In this trial, comparing the efficacy and safety of briakinumab to placebo, 80.7% of the 981 patients randomized to receive briakinumab every 4 weeks following an induction phase experienced a 75% improvement in psoriasis symptoms (PASI 75) at week 12, compared with 4.5% of the 484 patients randomized to placebo. Additionally, 61.6% and 32.2% of patients, respectively, saw 90% and 100% symptom clearance. And at week 52, 82.4% of patients who had achieved PASI 75 maintained at least that level of clearance.
– M10-315 and M10-114. In each of two 12-week trials comparing briakinumab to etanercept, significantly more patients randomized to briakinumab achieved Physician Global Assessment (PGA) scores of 0 or 1 and PASI 75 clearance. In the 350-patient M10-315 and the 347-patient M10-114 studies, respectively, 72.7% and 71% of patients randomized to briakinumab treatment achieved PGA 0 or 1, compared with 29.5% and 39.7% of patients randomized to etanercept therapy and 4.2% and 2.9% of those randomized to placebo.
Additionally, in the respective studies, 80.6% and 81.9% of the briakinumab-treated patients achieved PASI 75, compared with 39.6% and 56% of the etanercept patents, and 6.9% and 7.4% of the placebo groups.
– M10-255. The fourth trial of 317 patients was a head-to-head comparison with methotrexate in which 81.8% of briakinumab patients achieved PASI 75 clearance at 24 weeks, compared with 39.9% of those taking methotrexate. At 52 weeks, the PASI 75 clearance rates were 66.2% in the briakinumab group and 23.9% in the methotrexate group.
The most common adverse events observed across all four studies and in an ongoing open-label extension were upper respiratory infection, nasopharyngitis, headache, arthralgia, hypertension, and back pain. The incidence of infection and malignancy with briakinumab were higher than those with placebo, but were similar to those in patients treated with etanercept or methotrexate, according to a press release issued by Abbott.
Of particular note is the incidence of major adverse cardiovascular events (MACE) associated with briakinumab, Dr. Leonardi said in an interview. In the M06-890 trial, for example, seven briakinumab patients – all of whom had identifiable underlying cardiovascular risk factors, according to Abbott – experienced a MACE. A similar number of major cardiovascular events were observed in phase III trials of ustekinumab (Stelara), the IL 12/23 antagonist that received Food and Drug Administration approval for psoriasis treatment, he said. "Both drugs share the same mechanism of action, so it’s important to be cautious," he said. "Briakinumab has been submitted for FDA approval. We are awaiting review and comment."
Ustekinumab
Positive ustekinumab results were also presented last year. A pooled analysis of safety data reported at the summer academy meeting of the American Academy of Dermatology in Chicago demonstrated a favorable risk/benefit profile for up to 3 years of treatment.
Based on integrated data for 3,117 patients from the pivotal phase III PHOENIX 1 and PHOENIX 2 trials and the phase III ACCEPT trial, the investigators reported that the overall rates of adverse and serious adverse events were comparable in the 45-mg and 90-mg ustekinumab patient groups.
For the 45-mg and 90-mg patient groups, respectively, the per-hundred-patient-year rates were 0.82 and 1.50 for serious infections, 0.69 and 0.46 for noncutaneous malignancies, and 0.41 and 0.35 for MACE.
These rates were consistent with expectations for both general and psoriasis populations, and they remained stable over time, according to Dr. Leonardi, one of the study investigators. The maintenance of the favorable safety profile in patients who have been treated for several years is "encouraging," he said, noting that ongoing 5-year follow-up studies will enable continued monitoring of the drug’s safety.
Other important developments that may bode well for the treatment of psoriasis include data on IL-17 inhibitors, oral and topical janus kinase (JAK) inhibitors, an oral phosphodiesterase inhibitor, several IL-23 blockers, and topical niacin/calcipotriene, according to Dr. Leonardi and Dr. Alice B. Gottlieb of Tufts Medical Center in Boston.
Targeting IL-17
Recent studies have suggested that IL-17A–producing T cells have a crucial role in the pathogenesis of psoriasis, making them a potential treatment target.
In a phase II trial, a fully human IL-17 antibody AIN457 (Novartis) produced clinically meaningful reductions of disease activity at 4 and 12 weeks in 13 of 18 psoriasis patients who were randomized to receive a single 3-mg/kg intravenous infusion of the drug. At study weeks 4 and 12, respectively, the mean PASI score decreased by 58% and 63% in the AIN457 patients, compared with 4% and 9% in placebo patients, the authors wrote (Sci. Transl. Med. 2010;52:52ra72).
The results of a phase I study of another IL-17 drug – Amgen’s AMG 827, which binds to and blocks signaling via the IL-17 receptor – were reported at the EADV meeting. Most of the 16 patients who received a single dose of the fully human monoclonal antibody experienced substantial improvements in psoriasis symptoms. Based on the favorable findings, a phase II study is currently underway.
JAK 3 Inhibitors
The JAK pathways are also believed to have roles in the psoriasis disease cascade and as such have emerged as a treatment target. The four known JAK enzymes – JAK1, JAK2, JAK3, and tyrosine kinase (TYK) 2 – are components of signaling mechanisms used by multiple cytokines and growth factors that trigger dysregulated inflammatory pathways, according to Dr. Gottlieb. Oral and topical inhibitors of JAK are in phase II and III trials and have had promising results so far, she said in an interview.
In a 197-patient, phase II efficacy and safety study, Pfizer’s investigational oral JAK3 inhibitor, tentatively named tasocitinib (CP-690550), produced statistically significant responses at 12 weeks, compared with placebo in adults with moderate to severe psoriasis. The PASI 75 responses for patients randomized to twice daily tasocitinib at 2-mg, 5-mg, and 15-mg doses, respectively, were 25%, 40.8%, and 66.7%, compared with 2.0% for placebo, according to a company-issued statement. Additionally, as early as study week 4, "treatment with 5 and 15 mg twice daily of tasocitinib significantly improved patient-reported health-related quality of life outcomes."
Regarding safety, three patients randomized to tasocitinib treatment experienced a total of five serious adverse events during the study. Additionally, the investigators observed dose dependent decreases in mean neutrophil counts and hemoglobin values and increases in mean LDL, HDL, and total cholesterol levels. A large-scale, phase III trial program, called Oral Psoriasis Treatment (OPT) trials, is currently underway.
Topical JAK inhibitors are also under development, which presumably will avoid some of the complications associated with systemic therapy, according to Dr. Gottlieb, who is involved with investigations of Incyte’s topical JAK1/JAK2 inhibitor, INCB18424, which has demonstrated robust activity and safety in placebo-controlled, multidose, phase IIb trials in patients with mild to moderate psoriasis.
In studies reported by Dr. Gottlieb at last year’s annual meeting of the American Academy of Dermatology, topical application of INCB18424 cream led to significant improvement in the psoriatic lesions of treated patients at day 28, with the total lesion scores decreasing twofold, compared with placebo. "The effects were seen as early as 2 weeks, and they extended through" the study period," she said, noting that immunohistochemical staining and microarray gene analysis data confirmed the improvement in skin histology and the reduction in the psoriatic molecular signature.
Apremilast May Break Ground
Poised to be "the first oral medication with a new mechanism of action for psoriasis in almost 20 years," the phosphodiesterase 4 (PDE4) enzyme inhibitor apremilast (Celgene) demonstrated favorable results in a phase IIb study reported in December 2009 and has recently begun phase III trials, according to Dr. Leonardi.
In the phase IIb trial, 352 patients with moderate to severe plaque-type psoriasis were randomized to receive 10 mg, 20 mg or 30 mg of apremilast twice per day or placebo. Of the apremilast patients, 41% of the 30-mg group, 29% of the 20-mg group, and 11% of the 10-mg group achieved a PASI 75 after 16 weeks, compared with 6% of the placebo group, according a company statement.
A comparison of infection rates showed that infections occurred in 48% of the 30 mg apremilast patients and 33% of the placebo patients. No serious adverse events related to apremilast were reported.
The positive results have led to the initiation of two pivotal phase III trials: the multicenter ESTEEM 1 and ESTEEM 2 safety and efficacy studies comprising, respectively, 825 patients and 405 patients with moderate to severe plaque psoriasis. Primary outcome data from both studies will be available in the summer of 2011.
Topical Niacin/Calcipotriene
The nonsteroidal topical options for psoriasis will likely be expanded further by the addition of topical niacin (nicotinamide) to standard treatment with the synthetic vitamin D derivative calcipotriene, according to Dr. Gottlieb.
A recent 168-patient multicenter, double-blind, randomized trial reported that 50% of patients randomized to combination therapy with 0.005% calcipotriene and 1.4% nicotinamide achieved symptom clearance or near clearance, compared with 18.8% of patients randomized to placebo, 25% of patients treated with nicotinamide alone, and 31.5% of patients treated with calcipotriene alone.
The findings suggest that the combination therapy "may prove effective as an alternative therapeutic option to calcipotriene monotherapy and may provide an attractive option for patients seeking an effective corticosteroid-sparing topical psoriatic agent," the investigators wrote (J. Am. Acad. Dermatol. 2010;63:775-81).
In the midst of all of the new developments, it is important to note that tumor necrosis factor (TNF) antagonists continue to perform well, Dr. Leonardi stressed. "Given that the class is now 12 years old and includes 2 million patients, we are unlikely to learn of major safety risks at this point, and registry data support the notion that TNF blockade is cardioprotective in [rheumatoid arthritis], in distinction to the IL 12/23 blockers, where the MACE issue is unresolved," he said. "At this point, it is important for dermatologists to remember that the IL-12/23 drugs, in particular, are new, and since they are psoriasis specific, there are no other disease states where they were previously used."
Until the new drugs are in widespread use, "they should be used with caution," Dr. Leonardi continued. "The last psoriasis-specific biologic we used was Raptiva, which was voluntarily removed from the market by Genentech after 3 patients were identified with a rare and fatal central nervous system infection. At the time these patients were identified, the drug had been on the market for 5 years and had been used in 23,000 patients."
What this means in clinical practice is that ustekinumab or briakinumab may be reasonable options for patients with a history of failure of TNF antagonists or a history of central or peripheral demyelination, but until longer-term safety data are available, it should not be the first choice in the majority of treatment-naive patients, said Dr. Leonardi.
Dr. Leonardi is a consultant, investigator, and/or on the speakers bureau of Amgen, Abbott, Celgene, Centocor, Genentech, Incyte, Novartis Pfizer, and several other pharmaceutical companies.
Dr. Gottlieb has current consulting/advisory board agreements with Abbott, Amgen, Celgene, Incyte, and numerous other pharmaceutical companies.
Increased insight into the pathogenesis of psoriasis has led to the development of new drugs with novel mechanisms of action, as well as safer and more effective approaches to treatment with conventional drugs
As a result, treatment options for the management of patients with moderate to severe psoriasis in particular – nearly 25% of all patients with psoriasis, according to the National Psoriasis Foundation – have broadened substantially, and research reported during the past year suggests they are on track for continued expansion.
Among the most notable trials of the year were establishing the "unsurpassed efficacy" of Abbott’s investigational monoclonal antibody briakinumab, according to Dr. Craig Leonardi, a dermatologist and psoriasis specialist at St. Louis University. Like the recently approved psoriatic drug ustekinumab (Stelara), briakinumab is an injectable biologic agent that targets the interleukin-12 and -23 (IL-12/23) proteins, which are believed to promote the inflammation associated with psoriasis.
IL-12/23 Inhibition
Four phase III clinical studies of briakinumab were presented at the annual meeting of the European Academy of Dermatology and Venereology (EADV) in Gothenburg, Sweden.
– M06-890. In this trial, comparing the efficacy and safety of briakinumab to placebo, 80.7% of the 981 patients randomized to receive briakinumab every 4 weeks following an induction phase experienced a 75% improvement in psoriasis symptoms (PASI 75) at week 12, compared with 4.5% of the 484 patients randomized to placebo. Additionally, 61.6% and 32.2% of patients, respectively, saw 90% and 100% symptom clearance. And at week 52, 82.4% of patients who had achieved PASI 75 maintained at least that level of clearance.
– M10-315 and M10-114. In each of two 12-week trials comparing briakinumab to etanercept, significantly more patients randomized to briakinumab achieved Physician Global Assessment (PGA) scores of 0 or 1 and PASI 75 clearance. In the 350-patient M10-315 and the 347-patient M10-114 studies, respectively, 72.7% and 71% of patients randomized to briakinumab treatment achieved PGA 0 or 1, compared with 29.5% and 39.7% of patients randomized to etanercept therapy and 4.2% and 2.9% of those randomized to placebo.
Additionally, in the respective studies, 80.6% and 81.9% of the briakinumab-treated patients achieved PASI 75, compared with 39.6% and 56% of the etanercept patents, and 6.9% and 7.4% of the placebo groups.
– M10-255. The fourth trial of 317 patients was a head-to-head comparison with methotrexate in which 81.8% of briakinumab patients achieved PASI 75 clearance at 24 weeks, compared with 39.9% of those taking methotrexate. At 52 weeks, the PASI 75 clearance rates were 66.2% in the briakinumab group and 23.9% in the methotrexate group.
The most common adverse events observed across all four studies and in an ongoing open-label extension were upper respiratory infection, nasopharyngitis, headache, arthralgia, hypertension, and back pain. The incidence of infection and malignancy with briakinumab were higher than those with placebo, but were similar to those in patients treated with etanercept or methotrexate, according to a press release issued by Abbott.
Of particular note is the incidence of major adverse cardiovascular events (MACE) associated with briakinumab, Dr. Leonardi said in an interview. In the M06-890 trial, for example, seven briakinumab patients – all of whom had identifiable underlying cardiovascular risk factors, according to Abbott – experienced a MACE. A similar number of major cardiovascular events were observed in phase III trials of ustekinumab (Stelara), the IL 12/23 antagonist that received Food and Drug Administration approval for psoriasis treatment, he said. "Both drugs share the same mechanism of action, so it’s important to be cautious," he said. "Briakinumab has been submitted for FDA approval. We are awaiting review and comment."
Ustekinumab
Positive ustekinumab results were also presented last year. A pooled analysis of safety data reported at the summer academy meeting of the American Academy of Dermatology in Chicago demonstrated a favorable risk/benefit profile for up to 3 years of treatment.
Based on integrated data for 3,117 patients from the pivotal phase III PHOENIX 1 and PHOENIX 2 trials and the phase III ACCEPT trial, the investigators reported that the overall rates of adverse and serious adverse events were comparable in the 45-mg and 90-mg ustekinumab patient groups.
For the 45-mg and 90-mg patient groups, respectively, the per-hundred-patient-year rates were 0.82 and 1.50 for serious infections, 0.69 and 0.46 for noncutaneous malignancies, and 0.41 and 0.35 for MACE.
These rates were consistent with expectations for both general and psoriasis populations, and they remained stable over time, according to Dr. Leonardi, one of the study investigators. The maintenance of the favorable safety profile in patients who have been treated for several years is "encouraging," he said, noting that ongoing 5-year follow-up studies will enable continued monitoring of the drug’s safety.
Other important developments that may bode well for the treatment of psoriasis include data on IL-17 inhibitors, oral and topical janus kinase (JAK) inhibitors, an oral phosphodiesterase inhibitor, several IL-23 blockers, and topical niacin/calcipotriene, according to Dr. Leonardi and Dr. Alice B. Gottlieb of Tufts Medical Center in Boston.
Targeting IL-17
Recent studies have suggested that IL-17A–producing T cells have a crucial role in the pathogenesis of psoriasis, making them a potential treatment target.
In a phase II trial, a fully human IL-17 antibody AIN457 (Novartis) produced clinically meaningful reductions of disease activity at 4 and 12 weeks in 13 of 18 psoriasis patients who were randomized to receive a single 3-mg/kg intravenous infusion of the drug. At study weeks 4 and 12, respectively, the mean PASI score decreased by 58% and 63% in the AIN457 patients, compared with 4% and 9% in placebo patients, the authors wrote (Sci. Transl. Med. 2010;52:52ra72).
The results of a phase I study of another IL-17 drug – Amgen’s AMG 827, which binds to and blocks signaling via the IL-17 receptor – were reported at the EADV meeting. Most of the 16 patients who received a single dose of the fully human monoclonal antibody experienced substantial improvements in psoriasis symptoms. Based on the favorable findings, a phase II study is currently underway.
JAK 3 Inhibitors
The JAK pathways are also believed to have roles in the psoriasis disease cascade and as such have emerged as a treatment target. The four known JAK enzymes – JAK1, JAK2, JAK3, and tyrosine kinase (TYK) 2 – are components of signaling mechanisms used by multiple cytokines and growth factors that trigger dysregulated inflammatory pathways, according to Dr. Gottlieb. Oral and topical inhibitors of JAK are in phase II and III trials and have had promising results so far, she said in an interview.
In a 197-patient, phase II efficacy and safety study, Pfizer’s investigational oral JAK3 inhibitor, tentatively named tasocitinib (CP-690550), produced statistically significant responses at 12 weeks, compared with placebo in adults with moderate to severe psoriasis. The PASI 75 responses for patients randomized to twice daily tasocitinib at 2-mg, 5-mg, and 15-mg doses, respectively, were 25%, 40.8%, and 66.7%, compared with 2.0% for placebo, according to a company-issued statement. Additionally, as early as study week 4, "treatment with 5 and 15 mg twice daily of tasocitinib significantly improved patient-reported health-related quality of life outcomes."
Regarding safety, three patients randomized to tasocitinib treatment experienced a total of five serious adverse events during the study. Additionally, the investigators observed dose dependent decreases in mean neutrophil counts and hemoglobin values and increases in mean LDL, HDL, and total cholesterol levels. A large-scale, phase III trial program, called Oral Psoriasis Treatment (OPT) trials, is currently underway.
Topical JAK inhibitors are also under development, which presumably will avoid some of the complications associated with systemic therapy, according to Dr. Gottlieb, who is involved with investigations of Incyte’s topical JAK1/JAK2 inhibitor, INCB18424, which has demonstrated robust activity and safety in placebo-controlled, multidose, phase IIb trials in patients with mild to moderate psoriasis.
In studies reported by Dr. Gottlieb at last year’s annual meeting of the American Academy of Dermatology, topical application of INCB18424 cream led to significant improvement in the psoriatic lesions of treated patients at day 28, with the total lesion scores decreasing twofold, compared with placebo. "The effects were seen as early as 2 weeks, and they extended through" the study period," she said, noting that immunohistochemical staining and microarray gene analysis data confirmed the improvement in skin histology and the reduction in the psoriatic molecular signature.
Apremilast May Break Ground
Poised to be "the first oral medication with a new mechanism of action for psoriasis in almost 20 years," the phosphodiesterase 4 (PDE4) enzyme inhibitor apremilast (Celgene) demonstrated favorable results in a phase IIb study reported in December 2009 and has recently begun phase III trials, according to Dr. Leonardi.
In the phase IIb trial, 352 patients with moderate to severe plaque-type psoriasis were randomized to receive 10 mg, 20 mg or 30 mg of apremilast twice per day or placebo. Of the apremilast patients, 41% of the 30-mg group, 29% of the 20-mg group, and 11% of the 10-mg group achieved a PASI 75 after 16 weeks, compared with 6% of the placebo group, according a company statement.
A comparison of infection rates showed that infections occurred in 48% of the 30 mg apremilast patients and 33% of the placebo patients. No serious adverse events related to apremilast were reported.
The positive results have led to the initiation of two pivotal phase III trials: the multicenter ESTEEM 1 and ESTEEM 2 safety and efficacy studies comprising, respectively, 825 patients and 405 patients with moderate to severe plaque psoriasis. Primary outcome data from both studies will be available in the summer of 2011.
Topical Niacin/Calcipotriene
The nonsteroidal topical options for psoriasis will likely be expanded further by the addition of topical niacin (nicotinamide) to standard treatment with the synthetic vitamin D derivative calcipotriene, according to Dr. Gottlieb.
A recent 168-patient multicenter, double-blind, randomized trial reported that 50% of patients randomized to combination therapy with 0.005% calcipotriene and 1.4% nicotinamide achieved symptom clearance or near clearance, compared with 18.8% of patients randomized to placebo, 25% of patients treated with nicotinamide alone, and 31.5% of patients treated with calcipotriene alone.
The findings suggest that the combination therapy "may prove effective as an alternative therapeutic option to calcipotriene monotherapy and may provide an attractive option for patients seeking an effective corticosteroid-sparing topical psoriatic agent," the investigators wrote (J. Am. Acad. Dermatol. 2010;63:775-81).
In the midst of all of the new developments, it is important to note that tumor necrosis factor (TNF) antagonists continue to perform well, Dr. Leonardi stressed. "Given that the class is now 12 years old and includes 2 million patients, we are unlikely to learn of major safety risks at this point, and registry data support the notion that TNF blockade is cardioprotective in [rheumatoid arthritis], in distinction to the IL 12/23 blockers, where the MACE issue is unresolved," he said. "At this point, it is important for dermatologists to remember that the IL-12/23 drugs, in particular, are new, and since they are psoriasis specific, there are no other disease states where they were previously used."
Until the new drugs are in widespread use, "they should be used with caution," Dr. Leonardi continued. "The last psoriasis-specific biologic we used was Raptiva, which was voluntarily removed from the market by Genentech after 3 patients were identified with a rare and fatal central nervous system infection. At the time these patients were identified, the drug had been on the market for 5 years and had been used in 23,000 patients."
What this means in clinical practice is that ustekinumab or briakinumab may be reasonable options for patients with a history of failure of TNF antagonists or a history of central or peripheral demyelination, but until longer-term safety data are available, it should not be the first choice in the majority of treatment-naive patients, said Dr. Leonardi.
Dr. Leonardi is a consultant, investigator, and/or on the speakers bureau of Amgen, Abbott, Celgene, Centocor, Genentech, Incyte, Novartis Pfizer, and several other pharmaceutical companies.
Dr. Gottlieb has current consulting/advisory board agreements with Abbott, Amgen, Celgene, Incyte, and numerous other pharmaceutical companies.
Increased insight into the pathogenesis of psoriasis has led to the development of new drugs with novel mechanisms of action, as well as safer and more effective approaches to treatment with conventional drugs
As a result, treatment options for the management of patients with moderate to severe psoriasis in particular – nearly 25% of all patients with psoriasis, according to the National Psoriasis Foundation – have broadened substantially, and research reported during the past year suggests they are on track for continued expansion.
Among the most notable trials of the year were establishing the "unsurpassed efficacy" of Abbott’s investigational monoclonal antibody briakinumab, according to Dr. Craig Leonardi, a dermatologist and psoriasis specialist at St. Louis University. Like the recently approved psoriatic drug ustekinumab (Stelara), briakinumab is an injectable biologic agent that targets the interleukin-12 and -23 (IL-12/23) proteins, which are believed to promote the inflammation associated with psoriasis.
IL-12/23 Inhibition
Four phase III clinical studies of briakinumab were presented at the annual meeting of the European Academy of Dermatology and Venereology (EADV) in Gothenburg, Sweden.
– M06-890. In this trial, comparing the efficacy and safety of briakinumab to placebo, 80.7% of the 981 patients randomized to receive briakinumab every 4 weeks following an induction phase experienced a 75% improvement in psoriasis symptoms (PASI 75) at week 12, compared with 4.5% of the 484 patients randomized to placebo. Additionally, 61.6% and 32.2% of patients, respectively, saw 90% and 100% symptom clearance. And at week 52, 82.4% of patients who had achieved PASI 75 maintained at least that level of clearance.
– M10-315 and M10-114. In each of two 12-week trials comparing briakinumab to etanercept, significantly more patients randomized to briakinumab achieved Physician Global Assessment (PGA) scores of 0 or 1 and PASI 75 clearance. In the 350-patient M10-315 and the 347-patient M10-114 studies, respectively, 72.7% and 71% of patients randomized to briakinumab treatment achieved PGA 0 or 1, compared with 29.5% and 39.7% of patients randomized to etanercept therapy and 4.2% and 2.9% of those randomized to placebo.
Additionally, in the respective studies, 80.6% and 81.9% of the briakinumab-treated patients achieved PASI 75, compared with 39.6% and 56% of the etanercept patents, and 6.9% and 7.4% of the placebo groups.
– M10-255. The fourth trial of 317 patients was a head-to-head comparison with methotrexate in which 81.8% of briakinumab patients achieved PASI 75 clearance at 24 weeks, compared with 39.9% of those taking methotrexate. At 52 weeks, the PASI 75 clearance rates were 66.2% in the briakinumab group and 23.9% in the methotrexate group.
The most common adverse events observed across all four studies and in an ongoing open-label extension were upper respiratory infection, nasopharyngitis, headache, arthralgia, hypertension, and back pain. The incidence of infection and malignancy with briakinumab were higher than those with placebo, but were similar to those in patients treated with etanercept or methotrexate, according to a press release issued by Abbott.
Of particular note is the incidence of major adverse cardiovascular events (MACE) associated with briakinumab, Dr. Leonardi said in an interview. In the M06-890 trial, for example, seven briakinumab patients – all of whom had identifiable underlying cardiovascular risk factors, according to Abbott – experienced a MACE. A similar number of major cardiovascular events were observed in phase III trials of ustekinumab (Stelara), the IL 12/23 antagonist that received Food and Drug Administration approval for psoriasis treatment, he said. "Both drugs share the same mechanism of action, so it’s important to be cautious," he said. "Briakinumab has been submitted for FDA approval. We are awaiting review and comment."
Ustekinumab
Positive ustekinumab results were also presented last year. A pooled analysis of safety data reported at the summer academy meeting of the American Academy of Dermatology in Chicago demonstrated a favorable risk/benefit profile for up to 3 years of treatment.
Based on integrated data for 3,117 patients from the pivotal phase III PHOENIX 1 and PHOENIX 2 trials and the phase III ACCEPT trial, the investigators reported that the overall rates of adverse and serious adverse events were comparable in the 45-mg and 90-mg ustekinumab patient groups.
For the 45-mg and 90-mg patient groups, respectively, the per-hundred-patient-year rates were 0.82 and 1.50 for serious infections, 0.69 and 0.46 for noncutaneous malignancies, and 0.41 and 0.35 for MACE.
These rates were consistent with expectations for both general and psoriasis populations, and they remained stable over time, according to Dr. Leonardi, one of the study investigators. The maintenance of the favorable safety profile in patients who have been treated for several years is "encouraging," he said, noting that ongoing 5-year follow-up studies will enable continued monitoring of the drug’s safety.
Other important developments that may bode well for the treatment of psoriasis include data on IL-17 inhibitors, oral and topical janus kinase (JAK) inhibitors, an oral phosphodiesterase inhibitor, several IL-23 blockers, and topical niacin/calcipotriene, according to Dr. Leonardi and Dr. Alice B. Gottlieb of Tufts Medical Center in Boston.
Targeting IL-17
Recent studies have suggested that IL-17A–producing T cells have a crucial role in the pathogenesis of psoriasis, making them a potential treatment target.
In a phase II trial, a fully human IL-17 antibody AIN457 (Novartis) produced clinically meaningful reductions of disease activity at 4 and 12 weeks in 13 of 18 psoriasis patients who were randomized to receive a single 3-mg/kg intravenous infusion of the drug. At study weeks 4 and 12, respectively, the mean PASI score decreased by 58% and 63% in the AIN457 patients, compared with 4% and 9% in placebo patients, the authors wrote (Sci. Transl. Med. 2010;52:52ra72).
The results of a phase I study of another IL-17 drug – Amgen’s AMG 827, which binds to and blocks signaling via the IL-17 receptor – were reported at the EADV meeting. Most of the 16 patients who received a single dose of the fully human monoclonal antibody experienced substantial improvements in psoriasis symptoms. Based on the favorable findings, a phase II study is currently underway.
JAK 3 Inhibitors
The JAK pathways are also believed to have roles in the psoriasis disease cascade and as such have emerged as a treatment target. The four known JAK enzymes – JAK1, JAK2, JAK3, and tyrosine kinase (TYK) 2 – are components of signaling mechanisms used by multiple cytokines and growth factors that trigger dysregulated inflammatory pathways, according to Dr. Gottlieb. Oral and topical inhibitors of JAK are in phase II and III trials and have had promising results so far, she said in an interview.
In a 197-patient, phase II efficacy and safety study, Pfizer’s investigational oral JAK3 inhibitor, tentatively named tasocitinib (CP-690550), produced statistically significant responses at 12 weeks, compared with placebo in adults with moderate to severe psoriasis. The PASI 75 responses for patients randomized to twice daily tasocitinib at 2-mg, 5-mg, and 15-mg doses, respectively, were 25%, 40.8%, and 66.7%, compared with 2.0% for placebo, according to a company-issued statement. Additionally, as early as study week 4, "treatment with 5 and 15 mg twice daily of tasocitinib significantly improved patient-reported health-related quality of life outcomes."
Regarding safety, three patients randomized to tasocitinib treatment experienced a total of five serious adverse events during the study. Additionally, the investigators observed dose dependent decreases in mean neutrophil counts and hemoglobin values and increases in mean LDL, HDL, and total cholesterol levels. A large-scale, phase III trial program, called Oral Psoriasis Treatment (OPT) trials, is currently underway.
Topical JAK inhibitors are also under development, which presumably will avoid some of the complications associated with systemic therapy, according to Dr. Gottlieb, who is involved with investigations of Incyte’s topical JAK1/JAK2 inhibitor, INCB18424, which has demonstrated robust activity and safety in placebo-controlled, multidose, phase IIb trials in patients with mild to moderate psoriasis.
In studies reported by Dr. Gottlieb at last year’s annual meeting of the American Academy of Dermatology, topical application of INCB18424 cream led to significant improvement in the psoriatic lesions of treated patients at day 28, with the total lesion scores decreasing twofold, compared with placebo. "The effects were seen as early as 2 weeks, and they extended through" the study period," she said, noting that immunohistochemical staining and microarray gene analysis data confirmed the improvement in skin histology and the reduction in the psoriatic molecular signature.
Apremilast May Break Ground
Poised to be "the first oral medication with a new mechanism of action for psoriasis in almost 20 years," the phosphodiesterase 4 (PDE4) enzyme inhibitor apremilast (Celgene) demonstrated favorable results in a phase IIb study reported in December 2009 and has recently begun phase III trials, according to Dr. Leonardi.
In the phase IIb trial, 352 patients with moderate to severe plaque-type psoriasis were randomized to receive 10 mg, 20 mg or 30 mg of apremilast twice per day or placebo. Of the apremilast patients, 41% of the 30-mg group, 29% of the 20-mg group, and 11% of the 10-mg group achieved a PASI 75 after 16 weeks, compared with 6% of the placebo group, according a company statement.
A comparison of infection rates showed that infections occurred in 48% of the 30 mg apremilast patients and 33% of the placebo patients. No serious adverse events related to apremilast were reported.
The positive results have led to the initiation of two pivotal phase III trials: the multicenter ESTEEM 1 and ESTEEM 2 safety and efficacy studies comprising, respectively, 825 patients and 405 patients with moderate to severe plaque psoriasis. Primary outcome data from both studies will be available in the summer of 2011.
Topical Niacin/Calcipotriene
The nonsteroidal topical options for psoriasis will likely be expanded further by the addition of topical niacin (nicotinamide) to standard treatment with the synthetic vitamin D derivative calcipotriene, according to Dr. Gottlieb.
A recent 168-patient multicenter, double-blind, randomized trial reported that 50% of patients randomized to combination therapy with 0.005% calcipotriene and 1.4% nicotinamide achieved symptom clearance or near clearance, compared with 18.8% of patients randomized to placebo, 25% of patients treated with nicotinamide alone, and 31.5% of patients treated with calcipotriene alone.
The findings suggest that the combination therapy "may prove effective as an alternative therapeutic option to calcipotriene monotherapy and may provide an attractive option for patients seeking an effective corticosteroid-sparing topical psoriatic agent," the investigators wrote (J. Am. Acad. Dermatol. 2010;63:775-81).
In the midst of all of the new developments, it is important to note that tumor necrosis factor (TNF) antagonists continue to perform well, Dr. Leonardi stressed. "Given that the class is now 12 years old and includes 2 million patients, we are unlikely to learn of major safety risks at this point, and registry data support the notion that TNF blockade is cardioprotective in [rheumatoid arthritis], in distinction to the IL 12/23 blockers, where the MACE issue is unresolved," he said. "At this point, it is important for dermatologists to remember that the IL-12/23 drugs, in particular, are new, and since they are psoriasis specific, there are no other disease states where they were previously used."
Until the new drugs are in widespread use, "they should be used with caution," Dr. Leonardi continued. "The last psoriasis-specific biologic we used was Raptiva, which was voluntarily removed from the market by Genentech after 3 patients were identified with a rare and fatal central nervous system infection. At the time these patients were identified, the drug had been on the market for 5 years and had been used in 23,000 patients."
What this means in clinical practice is that ustekinumab or briakinumab may be reasonable options for patients with a history of failure of TNF antagonists or a history of central or peripheral demyelination, but until longer-term safety data are available, it should not be the first choice in the majority of treatment-naive patients, said Dr. Leonardi.
Dr. Leonardi is a consultant, investigator, and/or on the speakers bureau of Amgen, Abbott, Celgene, Centocor, Genentech, Incyte, Novartis Pfizer, and several other pharmaceutical companies.
Dr. Gottlieb has current consulting/advisory board agreements with Abbott, Amgen, Celgene, Incyte, and numerous other pharmaceutical companies.
EXPERT ANALYSIS
Interrupting Biologic Therapy for Psoriasis: What to Expect
GOTHENBURG, SWEDEN - Biologic therapies have brought dermatologists an unprecedented ability to control moderate to severe psoriasis. But with these treatment victories have come questions about how treatment interruptions affect long-term response, according to Dr. Richard Langley.
New evidence indicates most psoriasis patients who are responding well to adalimumab (Humira) and then undergo a treatment interruption or drug holiday will regain nearly the same favorable level of response upon treatment resumption.
But a substantial minority – roughly one-third of those randomized to a treatment interruption in the 3-year open-label extension of the REVEAL trial – will not be able to recapture their prior high level of response, he said at a satellite symposium held during the annual congress of the European Academy of Dermatology and Venereology.
The good news, he added, is that this high-risk subgroup can be identified early in the course of their treatment interruption, in time for corrective action to be taken.
"The take-home message is that in patients who have an early loss of response during the interruption, particularly those who drop to less than Psoriasis Area and Severity Index [PASI] 50, these are people we should think about getting back on treatment. Many of these patients are getting an inferior result compared with what they'd have on continuous therapy or early retreatment," according to Dr. Langley, professor of medicine and director of research in the division of dermatology at Dalhousie University, Halifax, N.S.
The phase III REVEAL (Randomized Controlled Evaluation of Adalimumab Every Other Week Dosing in Moderate to Severe Psoriasis Trial) was a placebo-controlled, 52-week randomized trial in which 490 of the 814 patients randomized to adalimumab at baseline achieved at least a 75% improvement (PASI 75) at weeks 16 and 33, compared with baseline. These good responders were then rerandomized to 19 weeks of continued adalimumab or treatment interruption via placebo. After this 19-week interval, patients could receive an additional 2 years of adalimumab at 40 mg every 2 weeks in a long-term, open-label extension study.
In a new REVEAL analysis presented for the first time at the satellite symposium sponsored by Abbott, Dr. Langley reported that of 227 patients randomized to the 19-week treatment interruption, 45% were still at PASI 75-100 at the start of retreatment, 31% had dropped below PASI 50, and 24% were in between. The mean disease severity scores in these three groups at the start of retreatment were PASI 88, PASI 32, and PASI 64, respectively. Over the next 2 years of open-label retreatment, the patients who started retreatment with less than a PASI 50 showed an inferior response, compared with the other two groups. They climbed to a peak response of PASI 66 at week 24 of retreatment and finished at a mean PASI 58 at week 108.
In contrast, patients who were PASI 75-100 at the start of retreatment, compared with enrollment, peaked at a mean PASI 94 at week 12 of retreatment, finishing at PASI 86 at week 108. Those with PASI 50-74 when retreatment commenced had a peak PASI 79 at week 12 before finishing the extension study with a mean PASI 81, according to Dr. Langley.
Similar findings have been seen in other treatment interruption studies involving etanercept and infliximab: Retreatment of patients who were good responders prior to the interruption restored efficacy in most but not all patients, he continued.
Dr. Langley stressed that in his view the preferred strategy is continuous treatment of good responders to any given biologic agent. He said he is opposed to the idea of drug holidays in patients who have cleared because in a substantial number of cases, their disease will return and their quality of life will plummet.
"Is that truly the sort of holiday we want to take?" he asked. "You can start and stop, and start and stop, but ultimately you are losing control of some of these patients."
Of course, there are circumstances when a treatment interruption is necessary. This happens fairly frequently. Among the situations warranting a temporary halt to biologic therapy are pregnancy, major surgery, live virus vaccination, clinically significant infection, or an adverse event whose cause is uncertain.
Dr. Langley disclosed that he is on the speakers bureau for Abbott Laboratories and has financial relationships with numerous other pharmaceutical companies with an interest in psoriasis therapies.
GOTHENBURG, SWEDEN - Biologic therapies have brought dermatologists an unprecedented ability to control moderate to severe psoriasis. But with these treatment victories have come questions about how treatment interruptions affect long-term response, according to Dr. Richard Langley.
New evidence indicates most psoriasis patients who are responding well to adalimumab (Humira) and then undergo a treatment interruption or drug holiday will regain nearly the same favorable level of response upon treatment resumption.
But a substantial minority – roughly one-third of those randomized to a treatment interruption in the 3-year open-label extension of the REVEAL trial – will not be able to recapture their prior high level of response, he said at a satellite symposium held during the annual congress of the European Academy of Dermatology and Venereology.
The good news, he added, is that this high-risk subgroup can be identified early in the course of their treatment interruption, in time for corrective action to be taken.
"The take-home message is that in patients who have an early loss of response during the interruption, particularly those who drop to less than Psoriasis Area and Severity Index [PASI] 50, these are people we should think about getting back on treatment. Many of these patients are getting an inferior result compared with what they'd have on continuous therapy or early retreatment," according to Dr. Langley, professor of medicine and director of research in the division of dermatology at Dalhousie University, Halifax, N.S.
The phase III REVEAL (Randomized Controlled Evaluation of Adalimumab Every Other Week Dosing in Moderate to Severe Psoriasis Trial) was a placebo-controlled, 52-week randomized trial in which 490 of the 814 patients randomized to adalimumab at baseline achieved at least a 75% improvement (PASI 75) at weeks 16 and 33, compared with baseline. These good responders were then rerandomized to 19 weeks of continued adalimumab or treatment interruption via placebo. After this 19-week interval, patients could receive an additional 2 years of adalimumab at 40 mg every 2 weeks in a long-term, open-label extension study.
In a new REVEAL analysis presented for the first time at the satellite symposium sponsored by Abbott, Dr. Langley reported that of 227 patients randomized to the 19-week treatment interruption, 45% were still at PASI 75-100 at the start of retreatment, 31% had dropped below PASI 50, and 24% were in between. The mean disease severity scores in these three groups at the start of retreatment were PASI 88, PASI 32, and PASI 64, respectively. Over the next 2 years of open-label retreatment, the patients who started retreatment with less than a PASI 50 showed an inferior response, compared with the other two groups. They climbed to a peak response of PASI 66 at week 24 of retreatment and finished at a mean PASI 58 at week 108.
In contrast, patients who were PASI 75-100 at the start of retreatment, compared with enrollment, peaked at a mean PASI 94 at week 12 of retreatment, finishing at PASI 86 at week 108. Those with PASI 50-74 when retreatment commenced had a peak PASI 79 at week 12 before finishing the extension study with a mean PASI 81, according to Dr. Langley.
Similar findings have been seen in other treatment interruption studies involving etanercept and infliximab: Retreatment of patients who were good responders prior to the interruption restored efficacy in most but not all patients, he continued.
Dr. Langley stressed that in his view the preferred strategy is continuous treatment of good responders to any given biologic agent. He said he is opposed to the idea of drug holidays in patients who have cleared because in a substantial number of cases, their disease will return and their quality of life will plummet.
"Is that truly the sort of holiday we want to take?" he asked. "You can start and stop, and start and stop, but ultimately you are losing control of some of these patients."
Of course, there are circumstances when a treatment interruption is necessary. This happens fairly frequently. Among the situations warranting a temporary halt to biologic therapy are pregnancy, major surgery, live virus vaccination, clinically significant infection, or an adverse event whose cause is uncertain.
Dr. Langley disclosed that he is on the speakers bureau for Abbott Laboratories and has financial relationships with numerous other pharmaceutical companies with an interest in psoriasis therapies.
GOTHENBURG, SWEDEN - Biologic therapies have brought dermatologists an unprecedented ability to control moderate to severe psoriasis. But with these treatment victories have come questions about how treatment interruptions affect long-term response, according to Dr. Richard Langley.
New evidence indicates most psoriasis patients who are responding well to adalimumab (Humira) and then undergo a treatment interruption or drug holiday will regain nearly the same favorable level of response upon treatment resumption.
But a substantial minority – roughly one-third of those randomized to a treatment interruption in the 3-year open-label extension of the REVEAL trial – will not be able to recapture their prior high level of response, he said at a satellite symposium held during the annual congress of the European Academy of Dermatology and Venereology.
The good news, he added, is that this high-risk subgroup can be identified early in the course of their treatment interruption, in time for corrective action to be taken.
"The take-home message is that in patients who have an early loss of response during the interruption, particularly those who drop to less than Psoriasis Area and Severity Index [PASI] 50, these are people we should think about getting back on treatment. Many of these patients are getting an inferior result compared with what they'd have on continuous therapy or early retreatment," according to Dr. Langley, professor of medicine and director of research in the division of dermatology at Dalhousie University, Halifax, N.S.
The phase III REVEAL (Randomized Controlled Evaluation of Adalimumab Every Other Week Dosing in Moderate to Severe Psoriasis Trial) was a placebo-controlled, 52-week randomized trial in which 490 of the 814 patients randomized to adalimumab at baseline achieved at least a 75% improvement (PASI 75) at weeks 16 and 33, compared with baseline. These good responders were then rerandomized to 19 weeks of continued adalimumab or treatment interruption via placebo. After this 19-week interval, patients could receive an additional 2 years of adalimumab at 40 mg every 2 weeks in a long-term, open-label extension study.
In a new REVEAL analysis presented for the first time at the satellite symposium sponsored by Abbott, Dr. Langley reported that of 227 patients randomized to the 19-week treatment interruption, 45% were still at PASI 75-100 at the start of retreatment, 31% had dropped below PASI 50, and 24% were in between. The mean disease severity scores in these three groups at the start of retreatment were PASI 88, PASI 32, and PASI 64, respectively. Over the next 2 years of open-label retreatment, the patients who started retreatment with less than a PASI 50 showed an inferior response, compared with the other two groups. They climbed to a peak response of PASI 66 at week 24 of retreatment and finished at a mean PASI 58 at week 108.
In contrast, patients who were PASI 75-100 at the start of retreatment, compared with enrollment, peaked at a mean PASI 94 at week 12 of retreatment, finishing at PASI 86 at week 108. Those with PASI 50-74 when retreatment commenced had a peak PASI 79 at week 12 before finishing the extension study with a mean PASI 81, according to Dr. Langley.
Similar findings have been seen in other treatment interruption studies involving etanercept and infliximab: Retreatment of patients who were good responders prior to the interruption restored efficacy in most but not all patients, he continued.
Dr. Langley stressed that in his view the preferred strategy is continuous treatment of good responders to any given biologic agent. He said he is opposed to the idea of drug holidays in patients who have cleared because in a substantial number of cases, their disease will return and their quality of life will plummet.
"Is that truly the sort of holiday we want to take?" he asked. "You can start and stop, and start and stop, but ultimately you are losing control of some of these patients."
Of course, there are circumstances when a treatment interruption is necessary. This happens fairly frequently. Among the situations warranting a temporary halt to biologic therapy are pregnancy, major surgery, live virus vaccination, clinically significant infection, or an adverse event whose cause is uncertain.
Dr. Langley disclosed that he is on the speakers bureau for Abbott Laboratories and has financial relationships with numerous other pharmaceutical companies with an interest in psoriasis therapies.
FROM THE ANNUAL CONGRESS OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Major Finding: Of the patients, 45% were still at PASI 75-100 at the start of retreatment, 31% had dropped below PASI 50, and 24% were in between.
Data Source: In the phase III REVEAL study, 227 responders were rerandomized to 19 weeks of continued adalimumab or treatment interruption via placebo.
Disclosures: Dr. Langley disclosed that he is on the speakers bureau for Abbott Laboratories and has financial relationships with numerous other pharmaceutical companies with an interest in psoriasis therapies.
Severe Skin Lesions Complicate Anti-TNF Therapy for IBD
Skin lesions led to discontinuation of anti–tumor necrosis factor-alpha therapy for inflammatory bowel disease in 34% of patients, reported Dr. Jean François Rahier and colleagues in the December issue of Clinical Gastroenterology and Hepatology.
Moreover, the occurrence of psoriasiform and eczematiform lesions associated with anti-TNF-alpha therapy was especially pronounced among patients with a previous history or family history of psoriasis or atopy.
Dr. Rahier of the Cliniques Universitaires de Mont-Godinne, Yvoir, Belgium, studied 85 patients with skin lesions and IBD who were seen at 35 gastroenterology centers in Belgium, France, Switzerland, and Austria.
A total of 69 patients had Crohn’s disease, 15 had ulcerative colitis, and 1 had indeterminate colitis. In all, 23 of the 85 patients were male. Data were collected using a standardized questionnaire. All cases were then reviewed simultaneously by a gastroenterologist and a dermatologist, wrote the authors (Clin. Gastroenterol. Hepatol. 2010 December [doi:10.1016/j.cgh.2010.07.022]).
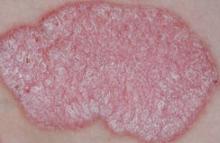
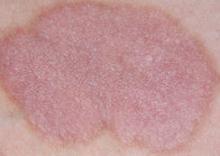
Psoriasiform lesions (defined as well-delineated, scaly, erythematous plaques, as well as nail pitting, nail discoloration, and palmoplantar pustulosis) were observed in 62 patients. Their median age at the time of lesion development was 32 years, with a female: male ratio of 2:1.
"Fourteen patients (23%) had a previous history of psoriasis before treatment with any anti-TNF agent," wrote the authors. The remainder were diagnosed as having a "new onset of psoriasiform eruption," and among these, eight (17%) reported a history of familial psoriasis, two (4%) reported a history of familial atopy, and six (13%) reported a personal history of atopy.
Overall, 25 of these patients had a good response to topical treatment, whereas the remaining 37 patients had no response. In all, 16 of these nonresponders stopped anti-TNF treatment immediately because of their lesions.
The remaining 21 patients switched to another TNF antagonist; 7 of those patients discontinued this anti-TNF treatment as well. Two patients attempted a third anti-TNF agent, unsuccessfully.
That amounted to an overall discontinuation rate of 40% (25/62) because of psoriasiform skin lesions.
Eczematiform lesions (defined as xerosis and pruriginous, ill-limited plaques with erythematous or squamous macules or vesicles) were noted in 23 patients. The median age at the time of occurrence was 31 years, and the female:male ratio in this group was 6:7.
Of these patients, 10 (44%) had a history of atopy, and 13 were incident cases. Four of these incident cases (31%) had a familial history of atopy, one had a personal history of psoriasis, and one reported familial psoriasis.
A total of 16 patients (70%) had a good response to topical treatment of their lesions, and 7 (30%) had no response.
"Overall a definitive withdrawal of anti-TNF therapies due to uncontrolled [eczematiform] skin lesion was required in 4/23 (17%) patients," wrote the authors.
That equated to a total withdrawal among the entire population of 29 patients, or 34% of the study’s subjects.
The occurrence of lesions was not correlated with disease activity in the bowel. The authors noted that although the majority of lesions occurred in patients taking infliximab, compared with certolizumab or adalimumab, "this most probably reflects the earlier approval of this drug in Europe."
The authors also conceded that the true incidence of withdrawal may have been overstated in this population, since "this paper is a selected cohort of patients presenting with severe psoriasiform and eczematiform skin lesions seen in various centers."
Nevertheless, in light of the high rate of anti-TNF discontinuation shown in this study, they recommended that "all patients should be referred to a dermatologist for clinical evaluation and biopsy of skin lesions if indicated.
"Future research is needed to further explore epidemiological, clinical and fundamental aspects of this vexing paradoxical reaction," they added.
Dr. Rahier disclosed receiving lecture fees from Abbott Laboratories and Schering-Plough. Several other authors also disclosed financial relationships with pharmaceutical companies. They reported that they received no funding for the study.
Skin lesions led to discontinuation of anti–tumor necrosis factor-alpha therapy for inflammatory bowel disease in 34% of patients, reported Dr. Jean François Rahier and colleagues in the December issue of Clinical Gastroenterology and Hepatology.
Moreover, the occurrence of psoriasiform and eczematiform lesions associated with anti-TNF-alpha therapy was especially pronounced among patients with a previous history or family history of psoriasis or atopy.
Dr. Rahier of the Cliniques Universitaires de Mont-Godinne, Yvoir, Belgium, studied 85 patients with skin lesions and IBD who were seen at 35 gastroenterology centers in Belgium, France, Switzerland, and Austria.
A total of 69 patients had Crohn’s disease, 15 had ulcerative colitis, and 1 had indeterminate colitis. In all, 23 of the 85 patients were male. Data were collected using a standardized questionnaire. All cases were then reviewed simultaneously by a gastroenterologist and a dermatologist, wrote the authors (Clin. Gastroenterol. Hepatol. 2010 December [doi:10.1016/j.cgh.2010.07.022]).
Psoriasiform lesions (defined as well-delineated, scaly, erythematous plaques, as well as nail pitting, nail discoloration, and palmoplantar pustulosis) were observed in 62 patients. Their median age at the time of lesion development was 32 years, with a female: male ratio of 2:1.
"Fourteen patients (23%) had a previous history of psoriasis before treatment with any anti-TNF agent," wrote the authors. The remainder were diagnosed as having a "new onset of psoriasiform eruption," and among these, eight (17%) reported a history of familial psoriasis, two (4%) reported a history of familial atopy, and six (13%) reported a personal history of atopy.
Overall, 25 of these patients had a good response to topical treatment, whereas the remaining 37 patients had no response. In all, 16 of these nonresponders stopped anti-TNF treatment immediately because of their lesions.
The remaining 21 patients switched to another TNF antagonist; 7 of those patients discontinued this anti-TNF treatment as well. Two patients attempted a third anti-TNF agent, unsuccessfully.
That amounted to an overall discontinuation rate of 40% (25/62) because of psoriasiform skin lesions.
Eczematiform lesions (defined as xerosis and pruriginous, ill-limited plaques with erythematous or squamous macules or vesicles) were noted in 23 patients. The median age at the time of occurrence was 31 years, and the female:male ratio in this group was 6:7.
Of these patients, 10 (44%) had a history of atopy, and 13 were incident cases. Four of these incident cases (31%) had a familial history of atopy, one had a personal history of psoriasis, and one reported familial psoriasis.
A total of 16 patients (70%) had a good response to topical treatment of their lesions, and 7 (30%) had no response.
"Overall a definitive withdrawal of anti-TNF therapies due to uncontrolled [eczematiform] skin lesion was required in 4/23 (17%) patients," wrote the authors.
That equated to a total withdrawal among the entire population of 29 patients, or 34% of the study’s subjects.
The occurrence of lesions was not correlated with disease activity in the bowel. The authors noted that although the majority of lesions occurred in patients taking infliximab, compared with certolizumab or adalimumab, "this most probably reflects the earlier approval of this drug in Europe."
The authors also conceded that the true incidence of withdrawal may have been overstated in this population, since "this paper is a selected cohort of patients presenting with severe psoriasiform and eczematiform skin lesions seen in various centers."
Nevertheless, in light of the high rate of anti-TNF discontinuation shown in this study, they recommended that "all patients should be referred to a dermatologist for clinical evaluation and biopsy of skin lesions if indicated.
"Future research is needed to further explore epidemiological, clinical and fundamental aspects of this vexing paradoxical reaction," they added.
Dr. Rahier disclosed receiving lecture fees from Abbott Laboratories and Schering-Plough. Several other authors also disclosed financial relationships with pharmaceutical companies. They reported that they received no funding for the study.
Skin lesions led to discontinuation of anti–tumor necrosis factor-alpha therapy for inflammatory bowel disease in 34% of patients, reported Dr. Jean François Rahier and colleagues in the December issue of Clinical Gastroenterology and Hepatology.
Moreover, the occurrence of psoriasiform and eczematiform lesions associated with anti-TNF-alpha therapy was especially pronounced among patients with a previous history or family history of psoriasis or atopy.
Dr. Rahier of the Cliniques Universitaires de Mont-Godinne, Yvoir, Belgium, studied 85 patients with skin lesions and IBD who were seen at 35 gastroenterology centers in Belgium, France, Switzerland, and Austria.
A total of 69 patients had Crohn’s disease, 15 had ulcerative colitis, and 1 had indeterminate colitis. In all, 23 of the 85 patients were male. Data were collected using a standardized questionnaire. All cases were then reviewed simultaneously by a gastroenterologist and a dermatologist, wrote the authors (Clin. Gastroenterol. Hepatol. 2010 December [doi:10.1016/j.cgh.2010.07.022]).
Psoriasiform lesions (defined as well-delineated, scaly, erythematous plaques, as well as nail pitting, nail discoloration, and palmoplantar pustulosis) were observed in 62 patients. Their median age at the time of lesion development was 32 years, with a female: male ratio of 2:1.
"Fourteen patients (23%) had a previous history of psoriasis before treatment with any anti-TNF agent," wrote the authors. The remainder were diagnosed as having a "new onset of psoriasiform eruption," and among these, eight (17%) reported a history of familial psoriasis, two (4%) reported a history of familial atopy, and six (13%) reported a personal history of atopy.
Overall, 25 of these patients had a good response to topical treatment, whereas the remaining 37 patients had no response. In all, 16 of these nonresponders stopped anti-TNF treatment immediately because of their lesions.
The remaining 21 patients switched to another TNF antagonist; 7 of those patients discontinued this anti-TNF treatment as well. Two patients attempted a third anti-TNF agent, unsuccessfully.
That amounted to an overall discontinuation rate of 40% (25/62) because of psoriasiform skin lesions.
Eczematiform lesions (defined as xerosis and pruriginous, ill-limited plaques with erythematous or squamous macules or vesicles) were noted in 23 patients. The median age at the time of occurrence was 31 years, and the female:male ratio in this group was 6:7.
Of these patients, 10 (44%) had a history of atopy, and 13 were incident cases. Four of these incident cases (31%) had a familial history of atopy, one had a personal history of psoriasis, and one reported familial psoriasis.
A total of 16 patients (70%) had a good response to topical treatment of their lesions, and 7 (30%) had no response.
"Overall a definitive withdrawal of anti-TNF therapies due to uncontrolled [eczematiform] skin lesion was required in 4/23 (17%) patients," wrote the authors.
That equated to a total withdrawal among the entire population of 29 patients, or 34% of the study’s subjects.
The occurrence of lesions was not correlated with disease activity in the bowel. The authors noted that although the majority of lesions occurred in patients taking infliximab, compared with certolizumab or adalimumab, "this most probably reflects the earlier approval of this drug in Europe."
The authors also conceded that the true incidence of withdrawal may have been overstated in this population, since "this paper is a selected cohort of patients presenting with severe psoriasiform and eczematiform skin lesions seen in various centers."
Nevertheless, in light of the high rate of anti-TNF discontinuation shown in this study, they recommended that "all patients should be referred to a dermatologist for clinical evaluation and biopsy of skin lesions if indicated.
"Future research is needed to further explore epidemiological, clinical and fundamental aspects of this vexing paradoxical reaction," they added.
Dr. Rahier disclosed receiving lecture fees from Abbott Laboratories and Schering-Plough. Several other authors also disclosed financial relationships with pharmaceutical companies. They reported that they received no funding for the study.
Major Finding: Overall, 34% of patients taking anti-TNF-alpha drugs for inflammatory bowel disease discontinued treatment because of severe psoriasiform and eczematiform skin lesions.
Data Source: A descriptive study of 85 patients with severe skin lesions secondary to anti-TNF treatment who were seen at 35 centers in Europe.
Disclosures: Dr. Rahier disclosed receiving lecture fees from Abbott Laboratories and Schering-Plough. Several other authors also disclosed financial relationships with pharmaceutical companies. They reported that they received no funding for the study.
Briakinumab Boosts Quality of Life for Psoriasis Patients
GOTHENBURG, SWEDEN - Psoriasis patients given the investigational interleukin-12/interleukin-23 inhibitor briakinumab had significantly better scores on health-related quality of life measures than did patients given etanercept in a phase III randomized, double-blind clinical trial.
"These results further enhanced the treatment benefits of briakinumab on patients’ lives beyond the previously described clinical efficacy in significantly reducing psoriasis symptoms versus placebo and etanercept," Yanjun Bao, Ph.D., said at the annual congress of the European Academy of Dermatology and Venereology.
The 12-week study involved 347 psoriasis patients who were randomized double-blind 2:2:1 to briakinumab, etanercept at 50 mg twice weekly, or placebo. Briakinumab was dosed at 200 mg at weeks 0 and 4, then 100 mg at week 8.
Treatment with briakinumab resulted in a mean 10.3-point reduction in Dermatology Life Quality Index scores from a baseline of 12.4, which was significantly greater than the 8.1-point decrease in the etanercept group or the 3.0-point decline in the placebo arm, reported Dr. Bao of Abbott Laboratories in Abbott Park, Ill.
In addition, the briakinumab group’s mean 29.1-point improvement in the visual analog scale for psoriasis-related pain from a baseline score of 34.5 was significantly larger than the 24-point reduction with etanercept and the 6.1-point decrease with placebo. The mean score on the Short Form-36 mental component summary improved by 5.4 points in the briakinumab group from a baseline of 45.9, a significantly greater response than the 3.2-point reduction with etanercept or the 1-point decrease with placebo, she continued.
The phase III study was sponsored by Abbott.
GOTHENBURG, SWEDEN - Psoriasis patients given the investigational interleukin-12/interleukin-23 inhibitor briakinumab had significantly better scores on health-related quality of life measures than did patients given etanercept in a phase III randomized, double-blind clinical trial.
"These results further enhanced the treatment benefits of briakinumab on patients’ lives beyond the previously described clinical efficacy in significantly reducing psoriasis symptoms versus placebo and etanercept," Yanjun Bao, Ph.D., said at the annual congress of the European Academy of Dermatology and Venereology.
The 12-week study involved 347 psoriasis patients who were randomized double-blind 2:2:1 to briakinumab, etanercept at 50 mg twice weekly, or placebo. Briakinumab was dosed at 200 mg at weeks 0 and 4, then 100 mg at week 8.
Treatment with briakinumab resulted in a mean 10.3-point reduction in Dermatology Life Quality Index scores from a baseline of 12.4, which was significantly greater than the 8.1-point decrease in the etanercept group or the 3.0-point decline in the placebo arm, reported Dr. Bao of Abbott Laboratories in Abbott Park, Ill.
In addition, the briakinumab group’s mean 29.1-point improvement in the visual analog scale for psoriasis-related pain from a baseline score of 34.5 was significantly larger than the 24-point reduction with etanercept and the 6.1-point decrease with placebo. The mean score on the Short Form-36 mental component summary improved by 5.4 points in the briakinumab group from a baseline of 45.9, a significantly greater response than the 3.2-point reduction with etanercept or the 1-point decrease with placebo, she continued.
The phase III study was sponsored by Abbott.
GOTHENBURG, SWEDEN - Psoriasis patients given the investigational interleukin-12/interleukin-23 inhibitor briakinumab had significantly better scores on health-related quality of life measures than did patients given etanercept in a phase III randomized, double-blind clinical trial.
"These results further enhanced the treatment benefits of briakinumab on patients’ lives beyond the previously described clinical efficacy in significantly reducing psoriasis symptoms versus placebo and etanercept," Yanjun Bao, Ph.D., said at the annual congress of the European Academy of Dermatology and Venereology.
The 12-week study involved 347 psoriasis patients who were randomized double-blind 2:2:1 to briakinumab, etanercept at 50 mg twice weekly, or placebo. Briakinumab was dosed at 200 mg at weeks 0 and 4, then 100 mg at week 8.
Treatment with briakinumab resulted in a mean 10.3-point reduction in Dermatology Life Quality Index scores from a baseline of 12.4, which was significantly greater than the 8.1-point decrease in the etanercept group or the 3.0-point decline in the placebo arm, reported Dr. Bao of Abbott Laboratories in Abbott Park, Ill.
In addition, the briakinumab group’s mean 29.1-point improvement in the visual analog scale for psoriasis-related pain from a baseline score of 34.5 was significantly larger than the 24-point reduction with etanercept and the 6.1-point decrease with placebo. The mean score on the Short Form-36 mental component summary improved by 5.4 points in the briakinumab group from a baseline of 45.9, a significantly greater response than the 3.2-point reduction with etanercept or the 1-point decrease with placebo, she continued.
The phase III study was sponsored by Abbott.
FROM THE ANNUAL CONGRESS OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Major Finding: Scores on the Dermatology Life Quality Index declined by a mean of 10.3points with briakinumab, 8.1 points with etanercept, and 3.0 points with placebo.
Data Source: A 12-week double-blind study of 347 psoriasis patients randomized 2:2:1 to briakinumab, etanercept at 50 mg twice weekly, or placebo. Briakinumab was dosed at 200 mg at weeks 0 and 4, then 100 mg at week 8.
Disclosures: The phase III study was sponsored by Abbott. Dr. Bao is employed by Abbott Laboratories.
Canakinumab Proves Safe, Effective for Long-Term CAPS Treatment
ATLANTA – Canakinumab was safe, well tolerated, and highly effective in the long-term treatment of cryopyrin-associated periodic syndromes in pediatric patients in an open-label phase III extension study.
Treatment of cryopyrin-associated periodic syndromes (CAPS) in 47 patients, aged 3-17 years, who were enrolled in the study was associated with a rapid and sustained clinical and biochemical remission across varying disease severity phenotypes.
Five of the children had familial cold auto-inflammatory syndrome (FCAS), 23 had Muckle-Wells syndrome (MWS), and 18 had chronic infantile neurologic, cutaneous, and articular syndrome/neonatal-onset multisystem inflammatory disease (CINCA/NOMID). One patient was found to not have a CAPS disease phenotype, reported Dr. Jasmin B. Kuemmerle-Deschner at the annual meeting of the American College of Rheumatology.
Of the 47 patients, 38 had not previously been treated with the human interleukin-1 beta monoclonal antibody, and 9 were "rolled over" from earlier phase II or III studies. Of the 38 canakinumab-naive patients, 68% achieved a complete response based on physician global assessment of disease activity and skin assessment, plus normalization of C-reactive protein (CRP) and lowering of serum amyloid A (SAA) to less than 10 mg/L for both measures. More than 80% of the canakinumab-naive patients were relapse-free after a median of 290 days of exposure to treatment, and normal CRP and SAA levels were also maintained throughout the study period in the rollover patients, said Dr. Kuemmerle-Deschner, a pediatric rheumatologist at Universitaetsklinikum Tuebingen (Germany).
The response in the treatment-naive patients was rapid, with levels of CRP and SAA decreasing within 7 days from 14.8 to 2.5 mg/L, and from 40.6 to 7.4 mg/L, respectively, she noted.
Also of note was that 8 of the 11 patients who had an incomplete response within 7 days developed a complete response by 14 days, and the remaining patients – all of whom had the most severe disease phenotype (CINCA/NOMID) – had a partial response, she said.
All patients were treated with 150 mg (or 2 mg/kg for those weighing 40 kg or less) of subcutaneous canakinumab once every 8 weeks. Patients who did not have a complete response were allowed dose adjustments, including an additional dose of 300 mg or 4 mg/kg for those weighing 40 kg or less, or more frequent dosing. Dose adjustments were required in 17 patients, including all of the patients with NOMID; those patients required mean doses of 5.8 mg/kg, compared with 5.5 mg/kg in the MWS patients, and 2.7 mg/kg in the FCAS patients.
Adverse events were generally mild or moderate and transient, and they included abdominal pain, pyrexia, headache, nasopharyngitis, and rhinitis.
"It was remarkable that 92% of patients had no injections-site reaction, and only 8% patients reported mild reaction," Dr. Kuemmerle-Deschner said.
Serious adverse events occurred in six patients, and included nephrotic syndrome, bronchitis, pneumonia, and appendicitis, which occurred in one patient each, and tonsillitis, which occurred in two patients. Only one patient withdrew from the study because of an adverse event.
The findings of this study show that canakinumab, for which earlier phase II and III studies demonstrated high efficacy for treating CAPS, also has long-term safety, tolerability, and efficacy, Dr. Kuemmerle-Deschner said.
"Canakinumab led to rapid and sustained response in the majority of patients," she said, noting that importantly, children with severe CAPS phenotypes required dose adjustment and higher overall doses to sustain remission.
Dr. Kuemmerle-Deschner disclosed that she has received research grants and consulting fees from Novartis Pharmaceuticals, the maker of canakinumab (Ilaris).
ATLANTA – Canakinumab was safe, well tolerated, and highly effective in the long-term treatment of cryopyrin-associated periodic syndromes in pediatric patients in an open-label phase III extension study.
Treatment of cryopyrin-associated periodic syndromes (CAPS) in 47 patients, aged 3-17 years, who were enrolled in the study was associated with a rapid and sustained clinical and biochemical remission across varying disease severity phenotypes.
Five of the children had familial cold auto-inflammatory syndrome (FCAS), 23 had Muckle-Wells syndrome (MWS), and 18 had chronic infantile neurologic, cutaneous, and articular syndrome/neonatal-onset multisystem inflammatory disease (CINCA/NOMID). One patient was found to not have a CAPS disease phenotype, reported Dr. Jasmin B. Kuemmerle-Deschner at the annual meeting of the American College of Rheumatology.
Of the 47 patients, 38 had not previously been treated with the human interleukin-1 beta monoclonal antibody, and 9 were "rolled over" from earlier phase II or III studies. Of the 38 canakinumab-naive patients, 68% achieved a complete response based on physician global assessment of disease activity and skin assessment, plus normalization of C-reactive protein (CRP) and lowering of serum amyloid A (SAA) to less than 10 mg/L for both measures. More than 80% of the canakinumab-naive patients were relapse-free after a median of 290 days of exposure to treatment, and normal CRP and SAA levels were also maintained throughout the study period in the rollover patients, said Dr. Kuemmerle-Deschner, a pediatric rheumatologist at Universitaetsklinikum Tuebingen (Germany).
The response in the treatment-naive patients was rapid, with levels of CRP and SAA decreasing within 7 days from 14.8 to 2.5 mg/L, and from 40.6 to 7.4 mg/L, respectively, she noted.
Also of note was that 8 of the 11 patients who had an incomplete response within 7 days developed a complete response by 14 days, and the remaining patients – all of whom had the most severe disease phenotype (CINCA/NOMID) – had a partial response, she said.
All patients were treated with 150 mg (or 2 mg/kg for those weighing 40 kg or less) of subcutaneous canakinumab once every 8 weeks. Patients who did not have a complete response were allowed dose adjustments, including an additional dose of 300 mg or 4 mg/kg for those weighing 40 kg or less, or more frequent dosing. Dose adjustments were required in 17 patients, including all of the patients with NOMID; those patients required mean doses of 5.8 mg/kg, compared with 5.5 mg/kg in the MWS patients, and 2.7 mg/kg in the FCAS patients.
Adverse events were generally mild or moderate and transient, and they included abdominal pain, pyrexia, headache, nasopharyngitis, and rhinitis.
"It was remarkable that 92% of patients had no injections-site reaction, and only 8% patients reported mild reaction," Dr. Kuemmerle-Deschner said.
Serious adverse events occurred in six patients, and included nephrotic syndrome, bronchitis, pneumonia, and appendicitis, which occurred in one patient each, and tonsillitis, which occurred in two patients. Only one patient withdrew from the study because of an adverse event.
The findings of this study show that canakinumab, for which earlier phase II and III studies demonstrated high efficacy for treating CAPS, also has long-term safety, tolerability, and efficacy, Dr. Kuemmerle-Deschner said.
"Canakinumab led to rapid and sustained response in the majority of patients," she said, noting that importantly, children with severe CAPS phenotypes required dose adjustment and higher overall doses to sustain remission.
Dr. Kuemmerle-Deschner disclosed that she has received research grants and consulting fees from Novartis Pharmaceuticals, the maker of canakinumab (Ilaris).
ATLANTA – Canakinumab was safe, well tolerated, and highly effective in the long-term treatment of cryopyrin-associated periodic syndromes in pediatric patients in an open-label phase III extension study.
Treatment of cryopyrin-associated periodic syndromes (CAPS) in 47 patients, aged 3-17 years, who were enrolled in the study was associated with a rapid and sustained clinical and biochemical remission across varying disease severity phenotypes.
Five of the children had familial cold auto-inflammatory syndrome (FCAS), 23 had Muckle-Wells syndrome (MWS), and 18 had chronic infantile neurologic, cutaneous, and articular syndrome/neonatal-onset multisystem inflammatory disease (CINCA/NOMID). One patient was found to not have a CAPS disease phenotype, reported Dr. Jasmin B. Kuemmerle-Deschner at the annual meeting of the American College of Rheumatology.
Of the 47 patients, 38 had not previously been treated with the human interleukin-1 beta monoclonal antibody, and 9 were "rolled over" from earlier phase II or III studies. Of the 38 canakinumab-naive patients, 68% achieved a complete response based on physician global assessment of disease activity and skin assessment, plus normalization of C-reactive protein (CRP) and lowering of serum amyloid A (SAA) to less than 10 mg/L for both measures. More than 80% of the canakinumab-naive patients were relapse-free after a median of 290 days of exposure to treatment, and normal CRP and SAA levels were also maintained throughout the study period in the rollover patients, said Dr. Kuemmerle-Deschner, a pediatric rheumatologist at Universitaetsklinikum Tuebingen (Germany).
The response in the treatment-naive patients was rapid, with levels of CRP and SAA decreasing within 7 days from 14.8 to 2.5 mg/L, and from 40.6 to 7.4 mg/L, respectively, she noted.
Also of note was that 8 of the 11 patients who had an incomplete response within 7 days developed a complete response by 14 days, and the remaining patients – all of whom had the most severe disease phenotype (CINCA/NOMID) – had a partial response, she said.
All patients were treated with 150 mg (or 2 mg/kg for those weighing 40 kg or less) of subcutaneous canakinumab once every 8 weeks. Patients who did not have a complete response were allowed dose adjustments, including an additional dose of 300 mg or 4 mg/kg for those weighing 40 kg or less, or more frequent dosing. Dose adjustments were required in 17 patients, including all of the patients with NOMID; those patients required mean doses of 5.8 mg/kg, compared with 5.5 mg/kg in the MWS patients, and 2.7 mg/kg in the FCAS patients.
Adverse events were generally mild or moderate and transient, and they included abdominal pain, pyrexia, headache, nasopharyngitis, and rhinitis.
"It was remarkable that 92% of patients had no injections-site reaction, and only 8% patients reported mild reaction," Dr. Kuemmerle-Deschner said.
Serious adverse events occurred in six patients, and included nephrotic syndrome, bronchitis, pneumonia, and appendicitis, which occurred in one patient each, and tonsillitis, which occurred in two patients. Only one patient withdrew from the study because of an adverse event.
The findings of this study show that canakinumab, for which earlier phase II and III studies demonstrated high efficacy for treating CAPS, also has long-term safety, tolerability, and efficacy, Dr. Kuemmerle-Deschner said.
"Canakinumab led to rapid and sustained response in the majority of patients," she said, noting that importantly, children with severe CAPS phenotypes required dose adjustment and higher overall doses to sustain remission.
Dr. Kuemmerle-Deschner disclosed that she has received research grants and consulting fees from Novartis Pharmaceuticals, the maker of canakinumab (Ilaris).
FROM THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF RHEUMATOLOGY
Major Finding: Of 38 canakinumab-naive patients, 68% achieved a complete response based on physician global assessment of disease activity and skin assessment, and normalization of C-reactive protein and serum amyloid A. More than 80% of the canakinumab-naive patients were relapse-free after a median of 290 days of exposure to treatment, and normal levels were also maintained throughout the study period in the rollover patients.
Data Source: From an open-label, single treatment arm, phase III extension study of canakinumab for CAPS.
Disclosures: Dr. Kuemmerle-Deschner disclosed that she has received research grants and consulting fees from Novartis Pharmaceuticals, the maker of canakinumab (Ilaris).
Juvenile Localized Scleroderma: Fewer Flares With Methotrexate
ATLANTA – Methotrexate was an effective and well-tolerated treatment for juvenile localized scleroderma in a randomized, double-blind, placebo-controlled trial involving 70 patients with active disease.
At the end of the 12-month study, 31 of 46 patients randomized to receive methotrexate had responded to treatment and were flare-free, compared with 7 of 24 patients given placebo, Dr. Francesco Zulian reported at the annual meeting of the American College of Rheumatology.
Methotrexate patients received a weekly oral dose of 15 mg/m2 (maximum, 20 mg) for 12 months or until flare of disease. Patients in the placebo group received a placebo administered in the same fashion for 12 months or to flare. Patients in both groups received prednisone at a dose of 1 mg/kg per day, with a maximum dose of 50 mg, for 3 months. They were then tapered to 0 over 1 month. During the initial 3 months of the study, treatment response was comparable for the two groups. Differences in response rates began to emerge after that point, said Dr. Zulian of the University of Padua (Italy).
Patients' lesions were evaluated using a computerized scoring system, and changes were quantified using a skin score rate. Active lesions were monitored based on clinical examination and serial thermography. The mean skin score rates decreased significantly from 1.0 at baseline to 0.79 in the patients on methotrexate, but did not decrease in the patients given placebo. The mean target lesion temperature on thermography decreased by 44% in the methotrexate group and by 12% in the placebo group, a significant difference, he said.
Adverse events occurred in 26 of 46 patients in the methotrexate group, and in 11 of 24 in the placebo group. In the methotrexate group, adverse events included alopecia, nausea, headache, fatigue, hepatotoxicity, weight gain, and striae rubra. The only adverse events in the placebo group were weight gain and striae rubra.
Interestingly, weight gain of more than 5% of body weight occurred in 11% of the methotrexate patients and 42% of the placebo patients, Dr. Zulian said, noting that this may suggest there is some "biological or pathophysiological link" between prednisone and methotrexate that tempers this steroid-related side effect.
None of the side effects was severe enough to stop treatment, but there was a high drop-out rate – due to relapse or lack of response – in both groups; 31 of 46 patients in the methotrexate group, and 7 of 24 in the placebo group, completed the study. At the end of the 12-month study, 17 of 24 patients randomized to placebo had experienced a relapse, compared with 15 of 46 patients randomized to the methotrexate group.
Patients included in the study were aged 6-17 years with active localized scleroderma of linear, generalized, or deep subtypes with onset before age 16. The treatment and placebo groups were similar in regard to clinical and immunological features, as well as mean disease duration of about 2 years, and the absence of immunosuppressive treatment in the 6 months prior to enrollment.
Many treatments have been tried in patients with juvenile localized scleroderma, and methotrexate has emerged as one of the most frequently used drugs for this condition.
"The findings of this study show that a combination of methotrexate and prednisone is of benefit and is well tolerated as treatment for severe course juvenile localized scleroderma," Dr. Zulian said.
Prior studies have shown efficacy ranging from 74% to 93%, but the findings of those studies have been based mostly on clinical judgment, while the current study utilized more objective evaluation criteria, including thermography, and skin score rates calculated based on computerized skin scoring, he said.
"The inclusion of standardized objective parameters represents an innovative way to assess the disease and compare progression in future clinical trials," he added.
Dr. Zulian said he had no relevant financial disclosures.
ATLANTA – Methotrexate was an effective and well-tolerated treatment for juvenile localized scleroderma in a randomized, double-blind, placebo-controlled trial involving 70 patients with active disease.
At the end of the 12-month study, 31 of 46 patients randomized to receive methotrexate had responded to treatment and were flare-free, compared with 7 of 24 patients given placebo, Dr. Francesco Zulian reported at the annual meeting of the American College of Rheumatology.
Methotrexate patients received a weekly oral dose of 15 mg/m2 (maximum, 20 mg) for 12 months or until flare of disease. Patients in the placebo group received a placebo administered in the same fashion for 12 months or to flare. Patients in both groups received prednisone at a dose of 1 mg/kg per day, with a maximum dose of 50 mg, for 3 months. They were then tapered to 0 over 1 month. During the initial 3 months of the study, treatment response was comparable for the two groups. Differences in response rates began to emerge after that point, said Dr. Zulian of the University of Padua (Italy).
Patients' lesions were evaluated using a computerized scoring system, and changes were quantified using a skin score rate. Active lesions were monitored based on clinical examination and serial thermography. The mean skin score rates decreased significantly from 1.0 at baseline to 0.79 in the patients on methotrexate, but did not decrease in the patients given placebo. The mean target lesion temperature on thermography decreased by 44% in the methotrexate group and by 12% in the placebo group, a significant difference, he said.
Adverse events occurred in 26 of 46 patients in the methotrexate group, and in 11 of 24 in the placebo group. In the methotrexate group, adverse events included alopecia, nausea, headache, fatigue, hepatotoxicity, weight gain, and striae rubra. The only adverse events in the placebo group were weight gain and striae rubra.
Interestingly, weight gain of more than 5% of body weight occurred in 11% of the methotrexate patients and 42% of the placebo patients, Dr. Zulian said, noting that this may suggest there is some "biological or pathophysiological link" between prednisone and methotrexate that tempers this steroid-related side effect.
None of the side effects was severe enough to stop treatment, but there was a high drop-out rate – due to relapse or lack of response – in both groups; 31 of 46 patients in the methotrexate group, and 7 of 24 in the placebo group, completed the study. At the end of the 12-month study, 17 of 24 patients randomized to placebo had experienced a relapse, compared with 15 of 46 patients randomized to the methotrexate group.
Patients included in the study were aged 6-17 years with active localized scleroderma of linear, generalized, or deep subtypes with onset before age 16. The treatment and placebo groups were similar in regard to clinical and immunological features, as well as mean disease duration of about 2 years, and the absence of immunosuppressive treatment in the 6 months prior to enrollment.
Many treatments have been tried in patients with juvenile localized scleroderma, and methotrexate has emerged as one of the most frequently used drugs for this condition.
"The findings of this study show that a combination of methotrexate and prednisone is of benefit and is well tolerated as treatment for severe course juvenile localized scleroderma," Dr. Zulian said.
Prior studies have shown efficacy ranging from 74% to 93%, but the findings of those studies have been based mostly on clinical judgment, while the current study utilized more objective evaluation criteria, including thermography, and skin score rates calculated based on computerized skin scoring, he said.
"The inclusion of standardized objective parameters represents an innovative way to assess the disease and compare progression in future clinical trials," he added.
Dr. Zulian said he had no relevant financial disclosures.
ATLANTA – Methotrexate was an effective and well-tolerated treatment for juvenile localized scleroderma in a randomized, double-blind, placebo-controlled trial involving 70 patients with active disease.
At the end of the 12-month study, 31 of 46 patients randomized to receive methotrexate had responded to treatment and were flare-free, compared with 7 of 24 patients given placebo, Dr. Francesco Zulian reported at the annual meeting of the American College of Rheumatology.
Methotrexate patients received a weekly oral dose of 15 mg/m2 (maximum, 20 mg) for 12 months or until flare of disease. Patients in the placebo group received a placebo administered in the same fashion for 12 months or to flare. Patients in both groups received prednisone at a dose of 1 mg/kg per day, with a maximum dose of 50 mg, for 3 months. They were then tapered to 0 over 1 month. During the initial 3 months of the study, treatment response was comparable for the two groups. Differences in response rates began to emerge after that point, said Dr. Zulian of the University of Padua (Italy).
Patients' lesions were evaluated using a computerized scoring system, and changes were quantified using a skin score rate. Active lesions were monitored based on clinical examination and serial thermography. The mean skin score rates decreased significantly from 1.0 at baseline to 0.79 in the patients on methotrexate, but did not decrease in the patients given placebo. The mean target lesion temperature on thermography decreased by 44% in the methotrexate group and by 12% in the placebo group, a significant difference, he said.
Adverse events occurred in 26 of 46 patients in the methotrexate group, and in 11 of 24 in the placebo group. In the methotrexate group, adverse events included alopecia, nausea, headache, fatigue, hepatotoxicity, weight gain, and striae rubra. The only adverse events in the placebo group were weight gain and striae rubra.
Interestingly, weight gain of more than 5% of body weight occurred in 11% of the methotrexate patients and 42% of the placebo patients, Dr. Zulian said, noting that this may suggest there is some "biological or pathophysiological link" between prednisone and methotrexate that tempers this steroid-related side effect.
None of the side effects was severe enough to stop treatment, but there was a high drop-out rate – due to relapse or lack of response – in both groups; 31 of 46 patients in the methotrexate group, and 7 of 24 in the placebo group, completed the study. At the end of the 12-month study, 17 of 24 patients randomized to placebo had experienced a relapse, compared with 15 of 46 patients randomized to the methotrexate group.
Patients included in the study were aged 6-17 years with active localized scleroderma of linear, generalized, or deep subtypes with onset before age 16. The treatment and placebo groups were similar in regard to clinical and immunological features, as well as mean disease duration of about 2 years, and the absence of immunosuppressive treatment in the 6 months prior to enrollment.
Many treatments have been tried in patients with juvenile localized scleroderma, and methotrexate has emerged as one of the most frequently used drugs for this condition.
"The findings of this study show that a combination of methotrexate and prednisone is of benefit and is well tolerated as treatment for severe course juvenile localized scleroderma," Dr. Zulian said.
Prior studies have shown efficacy ranging from 74% to 93%, but the findings of those studies have been based mostly on clinical judgment, while the current study utilized more objective evaluation criteria, including thermography, and skin score rates calculated based on computerized skin scoring, he said.
"The inclusion of standardized objective parameters represents an innovative way to assess the disease and compare progression in future clinical trials," he added.
Dr. Zulian said he had no relevant financial disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF RHEUMATOLOGY
Major Finding: At the end of the 12-month study, 17 of 24 patients randomized to receive placebo and prednisone experienced a relapse, compared with 15 of 46 patients randomized to methotrexate and prednisone.
Data Source: A randomized, double-blind, placebo-controlled trial of 70 children with juvenile localized scleroderma.
Disclosures: Dr. Zulian said he had no relevant financial disclosures.
Coal Tar Making a Psoriasis Comeback
LAS VEGAS – Topical tar therapy for psoriasis may be making a comeback thanks to more user-friendly formulations.
That trend is happening in Europe and may be replicated in the United States, Dr. Linda Stein Gold said at a dermatology seminar sponsored by Skin Disease Education Foundation (SDEF).
"Topical therapy is the bread and butter of psoriasis treatment," and coal tar has been used for centuries to control the symptoms of plaque psoriasis, said Dr. Stein Gold, director of dermatology research at Henry Ford Hospital, Detroit.
The Goeckerman regimen, a combination of topical tar and ultraviolet light therapy in use since the 1920s, proved "exceptionally" effective and durable, she noted, with an average time of 18 days to 90% clearing of psoriatic lesions and 90% of patients remaining clear for up to 8 months.
But tar therapy was time consuming, a logistical hassle, and aesthetically unpleasing, and its use declined in recent decades with the advent of systemic biologic medications. "People weren't using tar before because it's messy and it smelled. Now we have some better options," Dr. Stein Gold said.
One of the newer topical solutions is a transparent gel of 15% liquor carbonis distillate (LCD), the equivalent of 2.3% coal tar (Psorent, NeoStrata Co.). "It doesn’t discolor bleached hair" when used for scalp psoriasis, it is quick-drying, and it comes in bottles with "dab-on" applicators, she said.
In a controlled comparison with calcipotriol cream in 12 weeks of treatment of 60 adults with moderate plaque psoriasis, the LCD solution was more effective and led to fewer relapses 6 weeks after treatment. Among 55 patients with complete data, mean Psoriasis Area Severity Index (PASI) scores improved by 58% in the LCD solution group and 37% in the calcipotriol group, a significant difference (J. Am. Acad. Dermatol. 2009;60:Ab174 [doi: 10.1016/j.jaad.2008.11.757]).
A 75% improvement in PASI scores was seen in 11 of 27 (41%) of the LCD solution group and none of the 28 patients on calcipotriol. A 50% improvement in PASI scores was seen in 18 of 27 (67%) in the LCD group and 10 of 28 (36%) in the calcipotriol group. These differences between groups were statistically significant.
Among 42 patients with Physician Global Assessment (PGA) scores 6 weeks after treatment, the PGA scores worsened to baseline in 5 of 22 patients (23%) in the LCD solution group and in 14 of 20 patients (70%) in the calcipotriol group, again a significant difference.
A separate study compared the LCD solution in combination with UVB therapy on one side of the body with UVB light therapy alone on the other side of the body in 12 patients in 4 weeks of therapy. "Very quickly, the combo therapy gets more rapid and complete efficacy compared with UVB alone," said Dr. Stein Gold (J. Drugs. Dermatol. 2009;8 [4]:351-7).
Another relatively user-friendly product is an over-the-counter 2% coal tar formulation in a foam base (Scytera, Promius Pharma). The foam is "a much more cosmetically elegant" treatment compared with older tar therapies, she said. It spreads easily, dries quickly, and has an acceptable fragrance, Dr. Stein Gold added.
In a randomized, observer-blind study of 38 patients with chronic plaque-type psoriasis, two lesions on each patient were treated for 8 weeks with a 1% coal tar foam or calcipotriol cream. The treatments appeared to be comparably effective. More patients reported itching, unpleasant odor or staining with the tar foam than with calcipotriol cream, but the foam is considerably less expensive, the investigators noted (Br. J. Dermatol. 2003;149:350-53).
"Tar is something we probably should look at again," Dr. Stein Gold said.
Other useful topical therapies include corticosteroids, she said. While studies with objective measures of atrophy have proved that potent topical corticosteroids do lead to thinning of the skin over time, often this effect is not clinically noticeable, and it reverses over time once treatment is stopped.
Topical vitamin D is complementary to topical steroids for psoriasis therapy and helps counteract some of the side effects of topical steroids on skin.
Dr. Stein Gold said she has had financial associations with Leo Pharma, Medicis Pharmaceutical Corp., Stiefel Laboratories, Galderma, and Novartis. SDEF and this news organization are owned by Elsevier.
LAS VEGAS – Topical tar therapy for psoriasis may be making a comeback thanks to more user-friendly formulations.
That trend is happening in Europe and may be replicated in the United States, Dr. Linda Stein Gold said at a dermatology seminar sponsored by Skin Disease Education Foundation (SDEF).
"Topical therapy is the bread and butter of psoriasis treatment," and coal tar has been used for centuries to control the symptoms of plaque psoriasis, said Dr. Stein Gold, director of dermatology research at Henry Ford Hospital, Detroit.
The Goeckerman regimen, a combination of topical tar and ultraviolet light therapy in use since the 1920s, proved "exceptionally" effective and durable, she noted, with an average time of 18 days to 90% clearing of psoriatic lesions and 90% of patients remaining clear for up to 8 months.
But tar therapy was time consuming, a logistical hassle, and aesthetically unpleasing, and its use declined in recent decades with the advent of systemic biologic medications. "People weren't using tar before because it's messy and it smelled. Now we have some better options," Dr. Stein Gold said.
One of the newer topical solutions is a transparent gel of 15% liquor carbonis distillate (LCD), the equivalent of 2.3% coal tar (Psorent, NeoStrata Co.). "It doesn’t discolor bleached hair" when used for scalp psoriasis, it is quick-drying, and it comes in bottles with "dab-on" applicators, she said.
In a controlled comparison with calcipotriol cream in 12 weeks of treatment of 60 adults with moderate plaque psoriasis, the LCD solution was more effective and led to fewer relapses 6 weeks after treatment. Among 55 patients with complete data, mean Psoriasis Area Severity Index (PASI) scores improved by 58% in the LCD solution group and 37% in the calcipotriol group, a significant difference (J. Am. Acad. Dermatol. 2009;60:Ab174 [doi: 10.1016/j.jaad.2008.11.757]).
A 75% improvement in PASI scores was seen in 11 of 27 (41%) of the LCD solution group and none of the 28 patients on calcipotriol. A 50% improvement in PASI scores was seen in 18 of 27 (67%) in the LCD group and 10 of 28 (36%) in the calcipotriol group. These differences between groups were statistically significant.
Among 42 patients with Physician Global Assessment (PGA) scores 6 weeks after treatment, the PGA scores worsened to baseline in 5 of 22 patients (23%) in the LCD solution group and in 14 of 20 patients (70%) in the calcipotriol group, again a significant difference.
A separate study compared the LCD solution in combination with UVB therapy on one side of the body with UVB light therapy alone on the other side of the body in 12 patients in 4 weeks of therapy. "Very quickly, the combo therapy gets more rapid and complete efficacy compared with UVB alone," said Dr. Stein Gold (J. Drugs. Dermatol. 2009;8 [4]:351-7).
Another relatively user-friendly product is an over-the-counter 2% coal tar formulation in a foam base (Scytera, Promius Pharma). The foam is "a much more cosmetically elegant" treatment compared with older tar therapies, she said. It spreads easily, dries quickly, and has an acceptable fragrance, Dr. Stein Gold added.
In a randomized, observer-blind study of 38 patients with chronic plaque-type psoriasis, two lesions on each patient were treated for 8 weeks with a 1% coal tar foam or calcipotriol cream. The treatments appeared to be comparably effective. More patients reported itching, unpleasant odor or staining with the tar foam than with calcipotriol cream, but the foam is considerably less expensive, the investigators noted (Br. J. Dermatol. 2003;149:350-53).
"Tar is something we probably should look at again," Dr. Stein Gold said.
Other useful topical therapies include corticosteroids, she said. While studies with objective measures of atrophy have proved that potent topical corticosteroids do lead to thinning of the skin over time, often this effect is not clinically noticeable, and it reverses over time once treatment is stopped.
Topical vitamin D is complementary to topical steroids for psoriasis therapy and helps counteract some of the side effects of topical steroids on skin.
Dr. Stein Gold said she has had financial associations with Leo Pharma, Medicis Pharmaceutical Corp., Stiefel Laboratories, Galderma, and Novartis. SDEF and this news organization are owned by Elsevier.
LAS VEGAS – Topical tar therapy for psoriasis may be making a comeback thanks to more user-friendly formulations.
That trend is happening in Europe and may be replicated in the United States, Dr. Linda Stein Gold said at a dermatology seminar sponsored by Skin Disease Education Foundation (SDEF).
"Topical therapy is the bread and butter of psoriasis treatment," and coal tar has been used for centuries to control the symptoms of plaque psoriasis, said Dr. Stein Gold, director of dermatology research at Henry Ford Hospital, Detroit.
The Goeckerman regimen, a combination of topical tar and ultraviolet light therapy in use since the 1920s, proved "exceptionally" effective and durable, she noted, with an average time of 18 days to 90% clearing of psoriatic lesions and 90% of patients remaining clear for up to 8 months.
But tar therapy was time consuming, a logistical hassle, and aesthetically unpleasing, and its use declined in recent decades with the advent of systemic biologic medications. "People weren't using tar before because it's messy and it smelled. Now we have some better options," Dr. Stein Gold said.
One of the newer topical solutions is a transparent gel of 15% liquor carbonis distillate (LCD), the equivalent of 2.3% coal tar (Psorent, NeoStrata Co.). "It doesn’t discolor bleached hair" when used for scalp psoriasis, it is quick-drying, and it comes in bottles with "dab-on" applicators, she said.
In a controlled comparison with calcipotriol cream in 12 weeks of treatment of 60 adults with moderate plaque psoriasis, the LCD solution was more effective and led to fewer relapses 6 weeks after treatment. Among 55 patients with complete data, mean Psoriasis Area Severity Index (PASI) scores improved by 58% in the LCD solution group and 37% in the calcipotriol group, a significant difference (J. Am. Acad. Dermatol. 2009;60:Ab174 [doi: 10.1016/j.jaad.2008.11.757]).
A 75% improvement in PASI scores was seen in 11 of 27 (41%) of the LCD solution group and none of the 28 patients on calcipotriol. A 50% improvement in PASI scores was seen in 18 of 27 (67%) in the LCD group and 10 of 28 (36%) in the calcipotriol group. These differences between groups were statistically significant.
Among 42 patients with Physician Global Assessment (PGA) scores 6 weeks after treatment, the PGA scores worsened to baseline in 5 of 22 patients (23%) in the LCD solution group and in 14 of 20 patients (70%) in the calcipotriol group, again a significant difference.
A separate study compared the LCD solution in combination with UVB therapy on one side of the body with UVB light therapy alone on the other side of the body in 12 patients in 4 weeks of therapy. "Very quickly, the combo therapy gets more rapid and complete efficacy compared with UVB alone," said Dr. Stein Gold (J. Drugs. Dermatol. 2009;8 [4]:351-7).
Another relatively user-friendly product is an over-the-counter 2% coal tar formulation in a foam base (Scytera, Promius Pharma). The foam is "a much more cosmetically elegant" treatment compared with older tar therapies, she said. It spreads easily, dries quickly, and has an acceptable fragrance, Dr. Stein Gold added.
In a randomized, observer-blind study of 38 patients with chronic plaque-type psoriasis, two lesions on each patient were treated for 8 weeks with a 1% coal tar foam or calcipotriol cream. The treatments appeared to be comparably effective. More patients reported itching, unpleasant odor or staining with the tar foam than with calcipotriol cream, but the foam is considerably less expensive, the investigators noted (Br. J. Dermatol. 2003;149:350-53).
"Tar is something we probably should look at again," Dr. Stein Gold said.
Other useful topical therapies include corticosteroids, she said. While studies with objective measures of atrophy have proved that potent topical corticosteroids do lead to thinning of the skin over time, often this effect is not clinically noticeable, and it reverses over time once treatment is stopped.
Topical vitamin D is complementary to topical steroids for psoriasis therapy and helps counteract some of the side effects of topical steroids on skin.
Dr. Stein Gold said she has had financial associations with Leo Pharma, Medicis Pharmaceutical Corp., Stiefel Laboratories, Galderma, and Novartis. SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM A DERMATOLOGY SEMINAR SPONSORED BY SKIN DISEASE EDUCATION FOUNDATION
Smoking Cessation Enhances Antimalarial Response in Cutaneous Lupus
SANTA MONICA, Calif. – Quitting smoking is the most important thing patients with cutaneous lupus erythematosus can do to increase their clinical response to antimalarials, according to Dr. Jeffrey P. Callen.
Smokers are already at risk of having more severe disease than their nonsmoking CLE counterparts, Dr. Callen noted at a rheumatology seminar sponsored by Skin Disease Education Foundation (SDEF) and the University of Louisville.
Antimalarial drugs are the basis of therapy for cutaneous lupus erythematosus (CLE). The need for maximizing treatment response has recently become clearer. In the past, certain patients with CLE were considered to be at low risk for progression to systemic LE (SLE). These included patients with fixed lesions with a potential for atrophy. However, data now show that these patients are as likely as others with CLE to progress to SLE.
The data come from a population-based study of inhabitants of Rochester, Minn. The researchers found that the incidence of CLE and SLE were equal. But of the 156 patients with CLE, 12% (19 patients) progressed to SLE within a mean of 8.2 years from the time of their CLE diagnosis, said Dr. Callen, professor of medicine (dermatology) and chief of the division of dermatology at the University of Louisville (Ky).
Of particular interest was the subtype of SLE that the researchers found in the 19 patients who progressed to SLE – there were 9 cases of the localized discoid subtype of CLE, 4 cases of the generalized discoid CLE subtype, 2 with the panniculitis CLE subtype, and 4 with the psoriasiform subtype of CLE (Arch. Dermatol. 2009;145:249-53).
These findings "give us reason to treat these patients more aggressively than we might have done," said Dr. Callen, and part of that more aggressive treatment is to get patients who smoke to stop.
Another tool is to get patients to use sunblock. "This is a photosensitive disease. It’s photodistributed. It’s photoexacerbated. We can reproduce it with phototesting. So sunscreens are important, but even more so is photoprotection. Patients need to change their behavior and wear photoprotective clothing. Whenever we do that, patients are going to have vitamin D deficiency. Data have come out recently that lupus patients in general have vitamin D deficiency. So we need to address that and [ensure that they] get adequate vitamin D. No one has done a study to see if [supplementation with vitamin D] makes a difference," Dr. Callen noted.
Some patients also respond to topical corticosteroids, selected according to the lesion and the site that are being treated. Topical calcineurin inhibitors have been used off label.
But first-line therapy is the antimalarial hydroxychloroquine or chloroquine. "To me, systemic corticosteroid therapy is not a therapy for cutaneous lupus," Dr. Callen said.
Data from one study suggest that giving patients hydroxychloroquine delays the time from onset of CLE to progression to SLE (Lupus 2007;16:401-9). "To me, that means we need to be treating patients with antimalarials earlier than we do," he said.
If those therapies do not work, "we may combine hydroxychloroquine or chloroquine with quinacrine, but we do not combine hydroxychloroquine and chloroquine. I am a little bit fearful of exacerbating potential ocular toxicity."
Or one can use a second-line therapy that ranges from immunosuppressive therapy to thalidomide.
"We ask first if the patients are taking drugs that may exacerbate the disease, especially in the setting of subacute cutaneous LE. And stop those drugs if at all possible. Is the patient smoking? Try to get them to stop," he said.
SDEF and this news organization are owned by Elsevier. Dr. Callen reported that he does not have any relevant financial relationships with any commercial interests.
SANTA MONICA, Calif. – Quitting smoking is the most important thing patients with cutaneous lupus erythematosus can do to increase their clinical response to antimalarials, according to Dr. Jeffrey P. Callen.
Smokers are already at risk of having more severe disease than their nonsmoking CLE counterparts, Dr. Callen noted at a rheumatology seminar sponsored by Skin Disease Education Foundation (SDEF) and the University of Louisville.
Antimalarial drugs are the basis of therapy for cutaneous lupus erythematosus (CLE). The need for maximizing treatment response has recently become clearer. In the past, certain patients with CLE were considered to be at low risk for progression to systemic LE (SLE). These included patients with fixed lesions with a potential for atrophy. However, data now show that these patients are as likely as others with CLE to progress to SLE.
The data come from a population-based study of inhabitants of Rochester, Minn. The researchers found that the incidence of CLE and SLE were equal. But of the 156 patients with CLE, 12% (19 patients) progressed to SLE within a mean of 8.2 years from the time of their CLE diagnosis, said Dr. Callen, professor of medicine (dermatology) and chief of the division of dermatology at the University of Louisville (Ky).
Of particular interest was the subtype of SLE that the researchers found in the 19 patients who progressed to SLE – there were 9 cases of the localized discoid subtype of CLE, 4 cases of the generalized discoid CLE subtype, 2 with the panniculitis CLE subtype, and 4 with the psoriasiform subtype of CLE (Arch. Dermatol. 2009;145:249-53).
These findings "give us reason to treat these patients more aggressively than we might have done," said Dr. Callen, and part of that more aggressive treatment is to get patients who smoke to stop.
Another tool is to get patients to use sunblock. "This is a photosensitive disease. It’s photodistributed. It’s photoexacerbated. We can reproduce it with phototesting. So sunscreens are important, but even more so is photoprotection. Patients need to change their behavior and wear photoprotective clothing. Whenever we do that, patients are going to have vitamin D deficiency. Data have come out recently that lupus patients in general have vitamin D deficiency. So we need to address that and [ensure that they] get adequate vitamin D. No one has done a study to see if [supplementation with vitamin D] makes a difference," Dr. Callen noted.
Some patients also respond to topical corticosteroids, selected according to the lesion and the site that are being treated. Topical calcineurin inhibitors have been used off label.
But first-line therapy is the antimalarial hydroxychloroquine or chloroquine. "To me, systemic corticosteroid therapy is not a therapy for cutaneous lupus," Dr. Callen said.
Data from one study suggest that giving patients hydroxychloroquine delays the time from onset of CLE to progression to SLE (Lupus 2007;16:401-9). "To me, that means we need to be treating patients with antimalarials earlier than we do," he said.
If those therapies do not work, "we may combine hydroxychloroquine or chloroquine with quinacrine, but we do not combine hydroxychloroquine and chloroquine. I am a little bit fearful of exacerbating potential ocular toxicity."
Or one can use a second-line therapy that ranges from immunosuppressive therapy to thalidomide.
"We ask first if the patients are taking drugs that may exacerbate the disease, especially in the setting of subacute cutaneous LE. And stop those drugs if at all possible. Is the patient smoking? Try to get them to stop," he said.
SDEF and this news organization are owned by Elsevier. Dr. Callen reported that he does not have any relevant financial relationships with any commercial interests.
SANTA MONICA, Calif. – Quitting smoking is the most important thing patients with cutaneous lupus erythematosus can do to increase their clinical response to antimalarials, according to Dr. Jeffrey P. Callen.
Smokers are already at risk of having more severe disease than their nonsmoking CLE counterparts, Dr. Callen noted at a rheumatology seminar sponsored by Skin Disease Education Foundation (SDEF) and the University of Louisville.
Antimalarial drugs are the basis of therapy for cutaneous lupus erythematosus (CLE). The need for maximizing treatment response has recently become clearer. In the past, certain patients with CLE were considered to be at low risk for progression to systemic LE (SLE). These included patients with fixed lesions with a potential for atrophy. However, data now show that these patients are as likely as others with CLE to progress to SLE.
The data come from a population-based study of inhabitants of Rochester, Minn. The researchers found that the incidence of CLE and SLE were equal. But of the 156 patients with CLE, 12% (19 patients) progressed to SLE within a mean of 8.2 years from the time of their CLE diagnosis, said Dr. Callen, professor of medicine (dermatology) and chief of the division of dermatology at the University of Louisville (Ky).
Of particular interest was the subtype of SLE that the researchers found in the 19 patients who progressed to SLE – there were 9 cases of the localized discoid subtype of CLE, 4 cases of the generalized discoid CLE subtype, 2 with the panniculitis CLE subtype, and 4 with the psoriasiform subtype of CLE (Arch. Dermatol. 2009;145:249-53).
These findings "give us reason to treat these patients more aggressively than we might have done," said Dr. Callen, and part of that more aggressive treatment is to get patients who smoke to stop.
Another tool is to get patients to use sunblock. "This is a photosensitive disease. It’s photodistributed. It’s photoexacerbated. We can reproduce it with phototesting. So sunscreens are important, but even more so is photoprotection. Patients need to change their behavior and wear photoprotective clothing. Whenever we do that, patients are going to have vitamin D deficiency. Data have come out recently that lupus patients in general have vitamin D deficiency. So we need to address that and [ensure that they] get adequate vitamin D. No one has done a study to see if [supplementation with vitamin D] makes a difference," Dr. Callen noted.
Some patients also respond to topical corticosteroids, selected according to the lesion and the site that are being treated. Topical calcineurin inhibitors have been used off label.
But first-line therapy is the antimalarial hydroxychloroquine or chloroquine. "To me, systemic corticosteroid therapy is not a therapy for cutaneous lupus," Dr. Callen said.
Data from one study suggest that giving patients hydroxychloroquine delays the time from onset of CLE to progression to SLE (Lupus 2007;16:401-9). "To me, that means we need to be treating patients with antimalarials earlier than we do," he said.
If those therapies do not work, "we may combine hydroxychloroquine or chloroquine with quinacrine, but we do not combine hydroxychloroquine and chloroquine. I am a little bit fearful of exacerbating potential ocular toxicity."
Or one can use a second-line therapy that ranges from immunosuppressive therapy to thalidomide.
"We ask first if the patients are taking drugs that may exacerbate the disease, especially in the setting of subacute cutaneous LE. And stop those drugs if at all possible. Is the patient smoking? Try to get them to stop," he said.
SDEF and this news organization are owned by Elsevier. Dr. Callen reported that he does not have any relevant financial relationships with any commercial interests.
EXPERT ANALYSIS FROM A RHEUMATOLOGY SEMINAR