User login
ACIP approves child and adolescent vaccination schedule for 2020
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices voted unanimously to approve the child and adolescent immunization schedule for 2020.
by busy providers,” Candice Robinson, MD, MPH, of the CDC’s National Center for Immunization and Respiratory Diseases, said at the CDC’s October meeting of ACIP. Updates reflect changes in language in the adult vaccination schedule, notably the change in the definition of “contraindication.” The updated wording in the Notes substitutes “not recommended or contraindicated” instead of the word “contraindicated” only.
Another notable change was the addition of information on adolescent vaccination of children who received the meningococcal ACWY vaccine before 10 years of age. For “children in whom boosters are not recommended due to an ongoing or increased risk of meningococcal disease” (such as a healthy child traveling to an endemic area), they should receive MenACWY according to the recommended adolescent schedule. But those children for whom boosters are recommended because of increased disease risk from conditions including complement deficiency, HIV, or asplenia should “follow the booster schedule for persons at increased risk.”
Other changes include restructuring of the notes for the live attenuated influenza vaccine (LAIV) in special situations. The schedule now uses a bulleted list to show that LAIV should not be used in the following circumstances:
- Having history of severe allergic reaction to a previous vaccine or vaccine component.
- Using aspirin or a salicylate-containing medication.
- Being aged 2-4 years with a history of asthma or wheezing.
- Having immunocompromised conditions.
- Having anatomic or functional asplenia.
- Having cochlear implants.
- Experiencing cerebrospinal fluid–oropharyngeal communication.
- Having immunocompromised close contacts or caregivers.
- Being pregnant.
- Having received flu antivirals within the previous 48 hours.
In addition, language on shared clinical decision-making was added to the notes on the meningococcal B vaccine for adolescents and young adults aged 18-23 years not at increased risk. Based on shared clinical decision making, the recommendation is a “two-dose series of Bexsero at least 1 month apart” or “two-dose series of Trumenba at least 6 months apart; if dose two is administered earlier than 6 months, administer a third dose at least 4 months after dose two.”
Several vaccines’ Notes sections, including hepatitis B and meningococcal disease, added links to detailed recommendations in the corresponding issues of the CDC’s Morbidity and Mortality Weekly Report, to allow clinicians easy access to additional information.
View the current Child & Adolescent Vaccination Schedule here.
The ACIP members had no financial conflicts to disclose.
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices voted unanimously to approve the child and adolescent immunization schedule for 2020.
by busy providers,” Candice Robinson, MD, MPH, of the CDC’s National Center for Immunization and Respiratory Diseases, said at the CDC’s October meeting of ACIP. Updates reflect changes in language in the adult vaccination schedule, notably the change in the definition of “contraindication.” The updated wording in the Notes substitutes “not recommended or contraindicated” instead of the word “contraindicated” only.
Another notable change was the addition of information on adolescent vaccination of children who received the meningococcal ACWY vaccine before 10 years of age. For “children in whom boosters are not recommended due to an ongoing or increased risk of meningococcal disease” (such as a healthy child traveling to an endemic area), they should receive MenACWY according to the recommended adolescent schedule. But those children for whom boosters are recommended because of increased disease risk from conditions including complement deficiency, HIV, or asplenia should “follow the booster schedule for persons at increased risk.”
Other changes include restructuring of the notes for the live attenuated influenza vaccine (LAIV) in special situations. The schedule now uses a bulleted list to show that LAIV should not be used in the following circumstances:
- Having history of severe allergic reaction to a previous vaccine or vaccine component.
- Using aspirin or a salicylate-containing medication.
- Being aged 2-4 years with a history of asthma or wheezing.
- Having immunocompromised conditions.
- Having anatomic or functional asplenia.
- Having cochlear implants.
- Experiencing cerebrospinal fluid–oropharyngeal communication.
- Having immunocompromised close contacts or caregivers.
- Being pregnant.
- Having received flu antivirals within the previous 48 hours.
In addition, language on shared clinical decision-making was added to the notes on the meningococcal B vaccine for adolescents and young adults aged 18-23 years not at increased risk. Based on shared clinical decision making, the recommendation is a “two-dose series of Bexsero at least 1 month apart” or “two-dose series of Trumenba at least 6 months apart; if dose two is administered earlier than 6 months, administer a third dose at least 4 months after dose two.”
Several vaccines’ Notes sections, including hepatitis B and meningococcal disease, added links to detailed recommendations in the corresponding issues of the CDC’s Morbidity and Mortality Weekly Report, to allow clinicians easy access to additional information.
View the current Child & Adolescent Vaccination Schedule here.
The ACIP members had no financial conflicts to disclose.
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices voted unanimously to approve the child and adolescent immunization schedule for 2020.
by busy providers,” Candice Robinson, MD, MPH, of the CDC’s National Center for Immunization and Respiratory Diseases, said at the CDC’s October meeting of ACIP. Updates reflect changes in language in the adult vaccination schedule, notably the change in the definition of “contraindication.” The updated wording in the Notes substitutes “not recommended or contraindicated” instead of the word “contraindicated” only.
Another notable change was the addition of information on adolescent vaccination of children who received the meningococcal ACWY vaccine before 10 years of age. For “children in whom boosters are not recommended due to an ongoing or increased risk of meningococcal disease” (such as a healthy child traveling to an endemic area), they should receive MenACWY according to the recommended adolescent schedule. But those children for whom boosters are recommended because of increased disease risk from conditions including complement deficiency, HIV, or asplenia should “follow the booster schedule for persons at increased risk.”
Other changes include restructuring of the notes for the live attenuated influenza vaccine (LAIV) in special situations. The schedule now uses a bulleted list to show that LAIV should not be used in the following circumstances:
- Having history of severe allergic reaction to a previous vaccine or vaccine component.
- Using aspirin or a salicylate-containing medication.
- Being aged 2-4 years with a history of asthma or wheezing.
- Having immunocompromised conditions.
- Having anatomic or functional asplenia.
- Having cochlear implants.
- Experiencing cerebrospinal fluid–oropharyngeal communication.
- Having immunocompromised close contacts or caregivers.
- Being pregnant.
- Having received flu antivirals within the previous 48 hours.
In addition, language on shared clinical decision-making was added to the notes on the meningococcal B vaccine for adolescents and young adults aged 18-23 years not at increased risk. Based on shared clinical decision making, the recommendation is a “two-dose series of Bexsero at least 1 month apart” or “two-dose series of Trumenba at least 6 months apart; if dose two is administered earlier than 6 months, administer a third dose at least 4 months after dose two.”
Several vaccines’ Notes sections, including hepatitis B and meningococcal disease, added links to detailed recommendations in the corresponding issues of the CDC’s Morbidity and Mortality Weekly Report, to allow clinicians easy access to additional information.
View the current Child & Adolescent Vaccination Schedule here.
The ACIP members had no financial conflicts to disclose.
FROM AN ACIP MEETING
Vaping-linked injuries top 1,600 cases
according to the latest update provided by the Centers for Disease Control and Prevention. Thirty-four deaths have been confirmed.
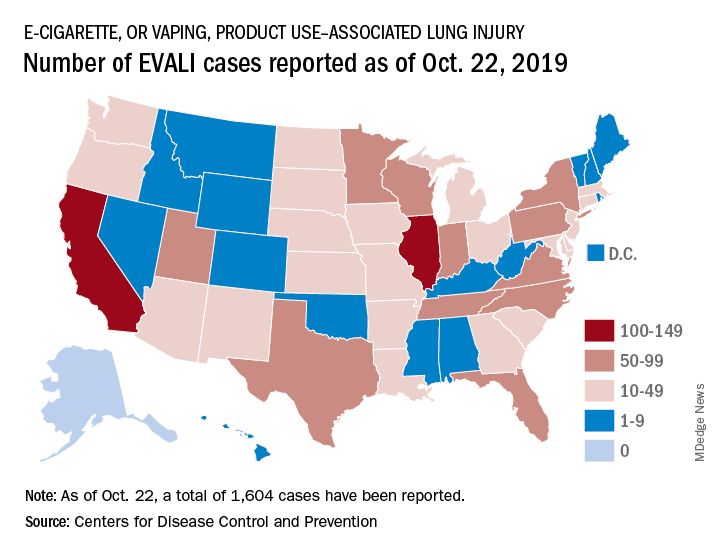
E-cigarette–linked lung injuries, now called EVALI, occurred in all U.S. states (except Alaska), the District of Columbia, and the U.S. Virgin Islands. Deaths have occurred in 24 states: Alabama, California (3), Connecticut, Delaware, Florida, Georgia (2), Illinois (2), Indiana (3), Kansas (2), Massachusetts, Michigan, Minnesota (3), Mississippi, Missouri, Montana, Nebraska, New Jersey, New York, Oregon (2), Pennsylvania, Tennessee, Texas, Utah, and Virginia. More deaths are under investigation.
The median age of deceased patients was 49 years and ranged from 17 to 75 years.
Data on age, sex, and substances used in e-cigarette, or vaping, products will be updated in the Morbidity and Mortality Weekly Report (MMWR) report being released on Friday, Oct. 25, 2019.
The CDC is now doing additional testing on available samples for chemical in the bronchoalveolar lavage fluid, blood, or urine, as well as lung biopsy or autopsy specimens. It also is validating methods for aerosol emission testing of case-associated product samples from vaping products and e-liquids.
For more information and resources visit For the Public, For Healthcare Providers, and For State and Local Health Departments pages, as well as the CDC’s Publications and Resources page.
according to the latest update provided by the Centers for Disease Control and Prevention. Thirty-four deaths have been confirmed.

E-cigarette–linked lung injuries, now called EVALI, occurred in all U.S. states (except Alaska), the District of Columbia, and the U.S. Virgin Islands. Deaths have occurred in 24 states: Alabama, California (3), Connecticut, Delaware, Florida, Georgia (2), Illinois (2), Indiana (3), Kansas (2), Massachusetts, Michigan, Minnesota (3), Mississippi, Missouri, Montana, Nebraska, New Jersey, New York, Oregon (2), Pennsylvania, Tennessee, Texas, Utah, and Virginia. More deaths are under investigation.
The median age of deceased patients was 49 years and ranged from 17 to 75 years.
Data on age, sex, and substances used in e-cigarette, or vaping, products will be updated in the Morbidity and Mortality Weekly Report (MMWR) report being released on Friday, Oct. 25, 2019.
The CDC is now doing additional testing on available samples for chemical in the bronchoalveolar lavage fluid, blood, or urine, as well as lung biopsy or autopsy specimens. It also is validating methods for aerosol emission testing of case-associated product samples from vaping products and e-liquids.
For more information and resources visit For the Public, For Healthcare Providers, and For State and Local Health Departments pages, as well as the CDC’s Publications and Resources page.
according to the latest update provided by the Centers for Disease Control and Prevention. Thirty-four deaths have been confirmed.

E-cigarette–linked lung injuries, now called EVALI, occurred in all U.S. states (except Alaska), the District of Columbia, and the U.S. Virgin Islands. Deaths have occurred in 24 states: Alabama, California (3), Connecticut, Delaware, Florida, Georgia (2), Illinois (2), Indiana (3), Kansas (2), Massachusetts, Michigan, Minnesota (3), Mississippi, Missouri, Montana, Nebraska, New Jersey, New York, Oregon (2), Pennsylvania, Tennessee, Texas, Utah, and Virginia. More deaths are under investigation.
The median age of deceased patients was 49 years and ranged from 17 to 75 years.
Data on age, sex, and substances used in e-cigarette, or vaping, products will be updated in the Morbidity and Mortality Weekly Report (MMWR) report being released on Friday, Oct. 25, 2019.
The CDC is now doing additional testing on available samples for chemical in the bronchoalveolar lavage fluid, blood, or urine, as well as lung biopsy or autopsy specimens. It also is validating methods for aerosol emission testing of case-associated product samples from vaping products and e-liquids.
For more information and resources visit For the Public, For Healthcare Providers, and For State and Local Health Departments pages, as well as the CDC’s Publications and Resources page.
REPORTING FROM THE CDC
ACIP approves 2020 adult vaccination schedule
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices voted unanimously to approve the adult immunization schedule for 2020, although some fine-tuning may occur before publication.
“Some of the wordsmithing may be done later,” ACIP executive secretary Amanda Cohn, MD, said at the ACIP October meeting.
Key updates to the schedule included a change in wording for the definition of the red bars on the table to include “not recommended or contraindicated” instead of only the word “contraindicated.” Committee members were especially interested in changing this wording to guide clinicians in use of the live attenuated influenza vaccine because of its potential value in vaccinating health care personnel.
Other updates include language that vaccination of adolescents and young adults aged 16-23 years who are not at increased risk for meningococcal disease should be vaccinated as follows: “Based on shared clinical decision making, 2-dose series MenB-4C at least 1 month apart or 2-dose series MenB-FHbp at 0, 6 months.”
Similarly, clinical decision-making language was added to the notes for the pneumococcal polysaccharide vaccine (PPSV23) and the 13-valent pneumococcal conjugate vaccine (PCV13).
The routine vaccination calls for only one dose of PPSV23 given on or after the individual’s 65th birthday. Then, based on shared clinical decision making, a dose of PCV13 is recommended for immunocompetent individuals aged 65 years and older. The notes also state that, based on shared clinical decision making, PCV13 and PPSV23 should not be given in the same visit and, if both will be given, PCV13 should be first and should be given 1 year before PPSV23. In addition, “PPSV23 should be given at least 5 years after any previous PPSV23 dose.”
The schedule also adds shared clinical decision making to the notes on human papillomavirus vaccination for adults aged 27-45 years.
The committee members acknowledged the increasing complexity of the adult vaccination schedule, but several members agreed that it is accessible to many clinicians.
“We can’t let the perfect be the enemy of the good” said Jason Goldman, MD, liaison representing the American College of Physicians. “Those who want to learn the schedule will learn it; the health system will learn it,” even if not every specialist does.
The table “is something to draw you in,” said Sandra Fryhofer, MD, an internist who is liaison for the American Medical Association. The notes provide more details.
More specific information about contraindications for patients with cochlear implants, which also came up in the discussion, may be added to the schedule at a later date.
View the current adult vaccination schedule here.
The ACIP members had no financial conflicts to disclose.
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices voted unanimously to approve the adult immunization schedule for 2020, although some fine-tuning may occur before publication.
“Some of the wordsmithing may be done later,” ACIP executive secretary Amanda Cohn, MD, said at the ACIP October meeting.
Key updates to the schedule included a change in wording for the definition of the red bars on the table to include “not recommended or contraindicated” instead of only the word “contraindicated.” Committee members were especially interested in changing this wording to guide clinicians in use of the live attenuated influenza vaccine because of its potential value in vaccinating health care personnel.
Other updates include language that vaccination of adolescents and young adults aged 16-23 years who are not at increased risk for meningococcal disease should be vaccinated as follows: “Based on shared clinical decision making, 2-dose series MenB-4C at least 1 month apart or 2-dose series MenB-FHbp at 0, 6 months.”
Similarly, clinical decision-making language was added to the notes for the pneumococcal polysaccharide vaccine (PPSV23) and the 13-valent pneumococcal conjugate vaccine (PCV13).
The routine vaccination calls for only one dose of PPSV23 given on or after the individual’s 65th birthday. Then, based on shared clinical decision making, a dose of PCV13 is recommended for immunocompetent individuals aged 65 years and older. The notes also state that, based on shared clinical decision making, PCV13 and PPSV23 should not be given in the same visit and, if both will be given, PCV13 should be first and should be given 1 year before PPSV23. In addition, “PPSV23 should be given at least 5 years after any previous PPSV23 dose.”
The schedule also adds shared clinical decision making to the notes on human papillomavirus vaccination for adults aged 27-45 years.
The committee members acknowledged the increasing complexity of the adult vaccination schedule, but several members agreed that it is accessible to many clinicians.
“We can’t let the perfect be the enemy of the good” said Jason Goldman, MD, liaison representing the American College of Physicians. “Those who want to learn the schedule will learn it; the health system will learn it,” even if not every specialist does.
The table “is something to draw you in,” said Sandra Fryhofer, MD, an internist who is liaison for the American Medical Association. The notes provide more details.
More specific information about contraindications for patients with cochlear implants, which also came up in the discussion, may be added to the schedule at a later date.
View the current adult vaccination schedule here.
The ACIP members had no financial conflicts to disclose.
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices voted unanimously to approve the adult immunization schedule for 2020, although some fine-tuning may occur before publication.
“Some of the wordsmithing may be done later,” ACIP executive secretary Amanda Cohn, MD, said at the ACIP October meeting.
Key updates to the schedule included a change in wording for the definition of the red bars on the table to include “not recommended or contraindicated” instead of only the word “contraindicated.” Committee members were especially interested in changing this wording to guide clinicians in use of the live attenuated influenza vaccine because of its potential value in vaccinating health care personnel.
Other updates include language that vaccination of adolescents and young adults aged 16-23 years who are not at increased risk for meningococcal disease should be vaccinated as follows: “Based on shared clinical decision making, 2-dose series MenB-4C at least 1 month apart or 2-dose series MenB-FHbp at 0, 6 months.”
Similarly, clinical decision-making language was added to the notes for the pneumococcal polysaccharide vaccine (PPSV23) and the 13-valent pneumococcal conjugate vaccine (PCV13).
The routine vaccination calls for only one dose of PPSV23 given on or after the individual’s 65th birthday. Then, based on shared clinical decision making, a dose of PCV13 is recommended for immunocompetent individuals aged 65 years and older. The notes also state that, based on shared clinical decision making, PCV13 and PPSV23 should not be given in the same visit and, if both will be given, PCV13 should be first and should be given 1 year before PPSV23. In addition, “PPSV23 should be given at least 5 years after any previous PPSV23 dose.”
The schedule also adds shared clinical decision making to the notes on human papillomavirus vaccination for adults aged 27-45 years.
The committee members acknowledged the increasing complexity of the adult vaccination schedule, but several members agreed that it is accessible to many clinicians.
“We can’t let the perfect be the enemy of the good” said Jason Goldman, MD, liaison representing the American College of Physicians. “Those who want to learn the schedule will learn it; the health system will learn it,” even if not every specialist does.
The table “is something to draw you in,” said Sandra Fryhofer, MD, an internist who is liaison for the American Medical Association. The notes provide more details.
More specific information about contraindications for patients with cochlear implants, which also came up in the discussion, may be added to the schedule at a later date.
View the current adult vaccination schedule here.
The ACIP members had no financial conflicts to disclose.
ACIP plans flu review for older adults
according to data presented at a meeting of the Centers for Disease Control and Prevention’s ACIP.
Lynette Brammer of the CDC’s National Center for Immunization and Respiratory Diseases (NCIRD) presented a surveillance update of the flu season in the United States so far. Overall, the influenza A(H3N2) viruses are predominant, although dominance varies in different regions of the country, and it is too soon to predict what strain will dominate later in the season.
“While two of the four vaccine components were updated for the Southern Hemisphere, the components selected for the 2019-2020 Northern Hemisphere vaccine, at this time, look appropriate for the season,” she said.
In other flu news, Lisa Groskopf, MD, of the NCIRD discussed the influenza work group’s plans for a meta-analysis to assess the relative benefit of different vaccines for older adults, in light of the growing variety of products available.
Currently, no preferential recommendations have been made for a specific vaccine for a particular age group. “There’s a dearth of data comparing these vaccines to one another,” said Dr. Groskopf. She added that, because vaccine effectiveness varies by season, the generalizability of effectiveness data is another challenge.
The work group’s systematic review and meta-analysis is designed to compare the high-dose inactivated influenza vaccine (HD-IIV), the adjuvanted inactivated influenza vaccine (aIIV), and the recombinant influenza vaccine (RIV). The study will include adults aged 65 years and older who receive trivalent or quadrivalent HD-IIV, aIIV, or RIV, compared with those who receive another influenza vaccine, a noninfluenza control vaccine, placebo, or no vaccine. The outcomes will include data on safety and effectiveness of the vaccines, Dr. Groskopf said.
In addition to safety and effectiveness, manufacturers such as Sanofi Pasteur continue to collect data on the success of available vaccines and develop new ones. Lee-Jah Chang, MD, of Sanofi Pasteur presented results of a noninferiority study of the company’s investigational high-dose quadrivalent influenza vaccine (QIV-HD; including two prevailing B viruses) versus the high-dose trivalent influenza vaccine (TID-HD). The study was conducted at 35 sites in the United States and included 2,670 adults aged 65 years and older.
Overall, the reactogenicity profile for patients given QIV-HD was similar to that of TID-HD, and approximately 5% of patients in the QIV group reported an immediate adverse event, Dr. Chang said. However, no related deaths or related adverse events of special interest occurred in any of the study groups.
Sanofi plans to pursue licensure of the QIV-HD vaccine, with a Center for Biologics Evaluation and Research action date of Nov. 4, 2019, said Dr. Chang. If the vaccine is licensed, it should be available for purchase by health care providers in the first quarter of 2020.
The ACIP members had no financial conflicts to disclose.
according to data presented at a meeting of the Centers for Disease Control and Prevention’s ACIP.
Lynette Brammer of the CDC’s National Center for Immunization and Respiratory Diseases (NCIRD) presented a surveillance update of the flu season in the United States so far. Overall, the influenza A(H3N2) viruses are predominant, although dominance varies in different regions of the country, and it is too soon to predict what strain will dominate later in the season.
“While two of the four vaccine components were updated for the Southern Hemisphere, the components selected for the 2019-2020 Northern Hemisphere vaccine, at this time, look appropriate for the season,” she said.
In other flu news, Lisa Groskopf, MD, of the NCIRD discussed the influenza work group’s plans for a meta-analysis to assess the relative benefit of different vaccines for older adults, in light of the growing variety of products available.
Currently, no preferential recommendations have been made for a specific vaccine for a particular age group. “There’s a dearth of data comparing these vaccines to one another,” said Dr. Groskopf. She added that, because vaccine effectiveness varies by season, the generalizability of effectiveness data is another challenge.
The work group’s systematic review and meta-analysis is designed to compare the high-dose inactivated influenza vaccine (HD-IIV), the adjuvanted inactivated influenza vaccine (aIIV), and the recombinant influenza vaccine (RIV). The study will include adults aged 65 years and older who receive trivalent or quadrivalent HD-IIV, aIIV, or RIV, compared with those who receive another influenza vaccine, a noninfluenza control vaccine, placebo, or no vaccine. The outcomes will include data on safety and effectiveness of the vaccines, Dr. Groskopf said.
In addition to safety and effectiveness, manufacturers such as Sanofi Pasteur continue to collect data on the success of available vaccines and develop new ones. Lee-Jah Chang, MD, of Sanofi Pasteur presented results of a noninferiority study of the company’s investigational high-dose quadrivalent influenza vaccine (QIV-HD; including two prevailing B viruses) versus the high-dose trivalent influenza vaccine (TID-HD). The study was conducted at 35 sites in the United States and included 2,670 adults aged 65 years and older.
Overall, the reactogenicity profile for patients given QIV-HD was similar to that of TID-HD, and approximately 5% of patients in the QIV group reported an immediate adverse event, Dr. Chang said. However, no related deaths or related adverse events of special interest occurred in any of the study groups.
Sanofi plans to pursue licensure of the QIV-HD vaccine, with a Center for Biologics Evaluation and Research action date of Nov. 4, 2019, said Dr. Chang. If the vaccine is licensed, it should be available for purchase by health care providers in the first quarter of 2020.
The ACIP members had no financial conflicts to disclose.
according to data presented at a meeting of the Centers for Disease Control and Prevention’s ACIP.
Lynette Brammer of the CDC’s National Center for Immunization and Respiratory Diseases (NCIRD) presented a surveillance update of the flu season in the United States so far. Overall, the influenza A(H3N2) viruses are predominant, although dominance varies in different regions of the country, and it is too soon to predict what strain will dominate later in the season.
“While two of the four vaccine components were updated for the Southern Hemisphere, the components selected for the 2019-2020 Northern Hemisphere vaccine, at this time, look appropriate for the season,” she said.
In other flu news, Lisa Groskopf, MD, of the NCIRD discussed the influenza work group’s plans for a meta-analysis to assess the relative benefit of different vaccines for older adults, in light of the growing variety of products available.
Currently, no preferential recommendations have been made for a specific vaccine for a particular age group. “There’s a dearth of data comparing these vaccines to one another,” said Dr. Groskopf. She added that, because vaccine effectiveness varies by season, the generalizability of effectiveness data is another challenge.
The work group’s systematic review and meta-analysis is designed to compare the high-dose inactivated influenza vaccine (HD-IIV), the adjuvanted inactivated influenza vaccine (aIIV), and the recombinant influenza vaccine (RIV). The study will include adults aged 65 years and older who receive trivalent or quadrivalent HD-IIV, aIIV, or RIV, compared with those who receive another influenza vaccine, a noninfluenza control vaccine, placebo, or no vaccine. The outcomes will include data on safety and effectiveness of the vaccines, Dr. Groskopf said.
In addition to safety and effectiveness, manufacturers such as Sanofi Pasteur continue to collect data on the success of available vaccines and develop new ones. Lee-Jah Chang, MD, of Sanofi Pasteur presented results of a noninferiority study of the company’s investigational high-dose quadrivalent influenza vaccine (QIV-HD; including two prevailing B viruses) versus the high-dose trivalent influenza vaccine (TID-HD). The study was conducted at 35 sites in the United States and included 2,670 adults aged 65 years and older.
Overall, the reactogenicity profile for patients given QIV-HD was similar to that of TID-HD, and approximately 5% of patients in the QIV group reported an immediate adverse event, Dr. Chang said. However, no related deaths or related adverse events of special interest occurred in any of the study groups.
Sanofi plans to pursue licensure of the QIV-HD vaccine, with a Center for Biologics Evaluation and Research action date of Nov. 4, 2019, said Dr. Chang. If the vaccine is licensed, it should be available for purchase by health care providers in the first quarter of 2020.
The ACIP members had no financial conflicts to disclose.
REPORTING FROM AN ACIP MEETING
ACIP recommends two options for pertussis vaccination
Either the Tdap or Td vaccine is an acceptable option for pertussis vaccination in most situations, recommended the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
In a unanimous 14-0 vote at the October meeting, based on the immunization schedule for persons aged 7 years and older.
Safety data showed no differences in safety concerns between Tdap and Td, including data from pregnant women, said Fiona Havers, MD, of the CDC’s National Center for Immunization and Respiratory Diseases (NCIRD), Atlanta.
Several of the ACIP members noted that the revised language to include both Tdap and Td reflects the increased use of Tdap and allows for maximum flexibility in clinical settings.
The revised language advises that booster doses of “either Td or Tdap” every 10 years throughout life are recommended for continued protection against tetanus and diphtheria. In addition, either Td or Tdap should be used if a tetanus toxoid–containing vaccine is indicated for prophylaxis in nonpregnant individuals.
For catch-up recommendations, which also apply to pregnant women, the committee approved the following wording for a series of three doses for individuals aged 7-18 years and 19 years and older who have never been vaccinated, that “the preferred schedule is a dose of Tdap (preferably the first dose), followed by either Tdap or Td at least 4 weeks afterward and another dose of either Td or Tdap 6-12 months later.” Individuals in these same age groups who are not fully vaccinated should receive one dose of Tdap, and a dose of either Td or Tdap if additional doses are needed.
The committee also voted unanimously 14-0 to accept the updated wording for pertussis vaccination in the Vaccines for Children program.
The ACIP members had no financial conflicts to disclose.
Either the Tdap or Td vaccine is an acceptable option for pertussis vaccination in most situations, recommended the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
In a unanimous 14-0 vote at the October meeting, based on the immunization schedule for persons aged 7 years and older.
Safety data showed no differences in safety concerns between Tdap and Td, including data from pregnant women, said Fiona Havers, MD, of the CDC’s National Center for Immunization and Respiratory Diseases (NCIRD), Atlanta.
Several of the ACIP members noted that the revised language to include both Tdap and Td reflects the increased use of Tdap and allows for maximum flexibility in clinical settings.
The revised language advises that booster doses of “either Td or Tdap” every 10 years throughout life are recommended for continued protection against tetanus and diphtheria. In addition, either Td or Tdap should be used if a tetanus toxoid–containing vaccine is indicated for prophylaxis in nonpregnant individuals.
For catch-up recommendations, which also apply to pregnant women, the committee approved the following wording for a series of three doses for individuals aged 7-18 years and 19 years and older who have never been vaccinated, that “the preferred schedule is a dose of Tdap (preferably the first dose), followed by either Tdap or Td at least 4 weeks afterward and another dose of either Td or Tdap 6-12 months later.” Individuals in these same age groups who are not fully vaccinated should receive one dose of Tdap, and a dose of either Td or Tdap if additional doses are needed.
The committee also voted unanimously 14-0 to accept the updated wording for pertussis vaccination in the Vaccines for Children program.
The ACIP members had no financial conflicts to disclose.
Either the Tdap or Td vaccine is an acceptable option for pertussis vaccination in most situations, recommended the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
In a unanimous 14-0 vote at the October meeting, based on the immunization schedule for persons aged 7 years and older.
Safety data showed no differences in safety concerns between Tdap and Td, including data from pregnant women, said Fiona Havers, MD, of the CDC’s National Center for Immunization and Respiratory Diseases (NCIRD), Atlanta.
Several of the ACIP members noted that the revised language to include both Tdap and Td reflects the increased use of Tdap and allows for maximum flexibility in clinical settings.
The revised language advises that booster doses of “either Td or Tdap” every 10 years throughout life are recommended for continued protection against tetanus and diphtheria. In addition, either Td or Tdap should be used if a tetanus toxoid–containing vaccine is indicated for prophylaxis in nonpregnant individuals.
For catch-up recommendations, which also apply to pregnant women, the committee approved the following wording for a series of three doses for individuals aged 7-18 years and 19 years and older who have never been vaccinated, that “the preferred schedule is a dose of Tdap (preferably the first dose), followed by either Tdap or Td at least 4 weeks afterward and another dose of either Td or Tdap 6-12 months later.” Individuals in these same age groups who are not fully vaccinated should receive one dose of Tdap, and a dose of either Td or Tdap if additional doses are needed.
The committee also voted unanimously 14-0 to accept the updated wording for pertussis vaccination in the Vaccines for Children program.
The ACIP members had no financial conflicts to disclose.
FROM AN ACIP MEETING
New test edges closer to rapid, accurate ID of active TB
A new point-of-care assay designed with machine learning offers improved accuracy for rapid identification of active tuberculosis (TB) infection, according to investigators.
, reported lead author Rushdy Ahmad, PhD, of the Broad Institute of MIT and Harvard in Cambridge, Mass., and colleagues. When fully developed, such a test could improve interventions for the most vulnerable patients, such as those with HIV, among whom TB often goes undiagnosed.
“Rapid and accurate diagnosis of active TB with current sputum-based diagnostic tools remains challenging in high-burden, resource-limited settings,” the investigators wrote. Their report is in Science Translational Medicine.
They went on to explain the gap that currently exists between microscopy, which is operator dependent and insensitive, and newer technologies, such as nucleic acid amplification, which are more sensitive but heavily resource dependent. “Furthermore, two of the most vulnerable and highly affected groups – young children and adults with HIV infection – are unlikely to be diagnosed using sputum because of difficulty obtaining sputum and low bacillary loads in the sample.”
To look for a more practical option, the investigators drew blood from 406 patients with chronic cough. Then, using a bead-based immunoassay with machine learning, the investigators identified four blood proteins associated with active TB infection: interleukin-6 (IL-6), IL-8, IL-18, and vascular endothelial growth factor (VEGF). Blind validation of 317 samples from patients with chronic cough in Asia, Africa, and South America showed that the four biomarkers offered a sensitivity of 80% and a specificity of 65%. By adding a fifth biomarker, an antibody against TB antigen Ag85B, the investigators were able to raise accuracy figures to 86% sensitivity and 69% specificity.
Adding even more biomarkers could theoretically raise accuracy even further, according to the investigators. The WHO minimal performance thresholds are 90% sensitivity and 70% specificity, with optimal targets slightly higher, at 95% sensitivity and 80% specificity. Although these standards have not yet been met, the investigators plan on testing the existing assay in real-world scenarios while simultaneously aiming to make it better.
“A near-term goal is ... to incrementally improve the marker panel up to an anticipated 6- to 10-plex assay,” the investigators wrote. “However, given the urgency of the problem, the possibility of incremental improvements will not delay platform refinement and field testing.”
The Bill and Melinda Gates Foundation funded the study. The investigators reported additional relationships with Quanterix Corporation and FIND.
SOURCE: Ahmad et al. Sci Transl Med. 2019 Oct 23. doi: 10.1126/scitranslmed.aaw8287.
A new point-of-care assay designed with machine learning offers improved accuracy for rapid identification of active tuberculosis (TB) infection, according to investigators.
, reported lead author Rushdy Ahmad, PhD, of the Broad Institute of MIT and Harvard in Cambridge, Mass., and colleagues. When fully developed, such a test could improve interventions for the most vulnerable patients, such as those with HIV, among whom TB often goes undiagnosed.
“Rapid and accurate diagnosis of active TB with current sputum-based diagnostic tools remains challenging in high-burden, resource-limited settings,” the investigators wrote. Their report is in Science Translational Medicine.
They went on to explain the gap that currently exists between microscopy, which is operator dependent and insensitive, and newer technologies, such as nucleic acid amplification, which are more sensitive but heavily resource dependent. “Furthermore, two of the most vulnerable and highly affected groups – young children and adults with HIV infection – are unlikely to be diagnosed using sputum because of difficulty obtaining sputum and low bacillary loads in the sample.”
To look for a more practical option, the investigators drew blood from 406 patients with chronic cough. Then, using a bead-based immunoassay with machine learning, the investigators identified four blood proteins associated with active TB infection: interleukin-6 (IL-6), IL-8, IL-18, and vascular endothelial growth factor (VEGF). Blind validation of 317 samples from patients with chronic cough in Asia, Africa, and South America showed that the four biomarkers offered a sensitivity of 80% and a specificity of 65%. By adding a fifth biomarker, an antibody against TB antigen Ag85B, the investigators were able to raise accuracy figures to 86% sensitivity and 69% specificity.
Adding even more biomarkers could theoretically raise accuracy even further, according to the investigators. The WHO minimal performance thresholds are 90% sensitivity and 70% specificity, with optimal targets slightly higher, at 95% sensitivity and 80% specificity. Although these standards have not yet been met, the investigators plan on testing the existing assay in real-world scenarios while simultaneously aiming to make it better.
“A near-term goal is ... to incrementally improve the marker panel up to an anticipated 6- to 10-plex assay,” the investigators wrote. “However, given the urgency of the problem, the possibility of incremental improvements will not delay platform refinement and field testing.”
The Bill and Melinda Gates Foundation funded the study. The investigators reported additional relationships with Quanterix Corporation and FIND.
SOURCE: Ahmad et al. Sci Transl Med. 2019 Oct 23. doi: 10.1126/scitranslmed.aaw8287.
A new point-of-care assay designed with machine learning offers improved accuracy for rapid identification of active tuberculosis (TB) infection, according to investigators.
, reported lead author Rushdy Ahmad, PhD, of the Broad Institute of MIT and Harvard in Cambridge, Mass., and colleagues. When fully developed, such a test could improve interventions for the most vulnerable patients, such as those with HIV, among whom TB often goes undiagnosed.
“Rapid and accurate diagnosis of active TB with current sputum-based diagnostic tools remains challenging in high-burden, resource-limited settings,” the investigators wrote. Their report is in Science Translational Medicine.
They went on to explain the gap that currently exists between microscopy, which is operator dependent and insensitive, and newer technologies, such as nucleic acid amplification, which are more sensitive but heavily resource dependent. “Furthermore, two of the most vulnerable and highly affected groups – young children and adults with HIV infection – are unlikely to be diagnosed using sputum because of difficulty obtaining sputum and low bacillary loads in the sample.”
To look for a more practical option, the investigators drew blood from 406 patients with chronic cough. Then, using a bead-based immunoassay with machine learning, the investigators identified four blood proteins associated with active TB infection: interleukin-6 (IL-6), IL-8, IL-18, and vascular endothelial growth factor (VEGF). Blind validation of 317 samples from patients with chronic cough in Asia, Africa, and South America showed that the four biomarkers offered a sensitivity of 80% and a specificity of 65%. By adding a fifth biomarker, an antibody against TB antigen Ag85B, the investigators were able to raise accuracy figures to 86% sensitivity and 69% specificity.
Adding even more biomarkers could theoretically raise accuracy even further, according to the investigators. The WHO minimal performance thresholds are 90% sensitivity and 70% specificity, with optimal targets slightly higher, at 95% sensitivity and 80% specificity. Although these standards have not yet been met, the investigators plan on testing the existing assay in real-world scenarios while simultaneously aiming to make it better.
“A near-term goal is ... to incrementally improve the marker panel up to an anticipated 6- to 10-plex assay,” the investigators wrote. “However, given the urgency of the problem, the possibility of incremental improvements will not delay platform refinement and field testing.”
The Bill and Melinda Gates Foundation funded the study. The investigators reported additional relationships with Quanterix Corporation and FIND.
SOURCE: Ahmad et al. Sci Transl Med. 2019 Oct 23. doi: 10.1126/scitranslmed.aaw8287.
FROM SCIENCE TRANSLATIONAL MEDICINE
Key clinical point: A new point-of-care assay designed with machine learning offers improved accuracy for rapid identification of active tuberculosis (TB) infection.
Major finding: The assay had a sensitivity of 86%.
Study details: A machine learning and validation study involving patients with chronic cough from multiple countries.
Disclosures: The Bill and Melinda Gates Foundation funded the study. The investigators reported relationships with Quanterix Corporation and FIND.
Source: Ahmad et al. Sci Transl Med. 2019 Oct 23. doi: 10.1126/scitranslmed.aaw8287.
Next-gen genomic test plus bronchoscopy may improve lung nodule management
NEW ORLEANS – The use of a results of a recent clinical validation study suggest.
The Percepta Genomic Sequencing Classifier (GSC) was able to up- and down-classify probability of malignancy for a considerable proportion of nondiagnostic bronchoscopies in the study, Peter J. Mazzone MD, FCCP, reported at the annual meeting of the American College of Chest Physicians.
The test is seen as complementary to bronchoscopy, improving the sensitivity of bronchoscopy overall and showing a combined sensitivity of greater than 95% in low- and intermediate-risk groups, according to Dr. Mazzone.
While the clinical utility of this genomic test needs to be further tested, the eventual goal is to improve clinician decision making when bronchoscopy results don’t clearly classify nodules as malignant or benign, Dr. Mazzone said in an interview.
“In that situation, you’re often left wondering, ‘what should I do next? Can I just watch this, and see if it grows and changes, or do I have to be even more aggressive – do another biopsy, or have a surgery to take it out?’ ” he explained. “So the test hopes to help make a more informed decision by further stratifying those patients as being quite low risk and maybe safe to follow, or quite high risk and maybe you should be considering more aggressive management.”
The GSC improves on the performance of an earlier molecular test, the Percepta Bronchial Genomic Classifier, which uses a brushing of bronchial epithelium to enhance nodule management in smokers, according to the researcher.
The next-generation GSC uses 1,232 gene transcripts from whole-transcriptome RNA sequencing, along with clinical factors, to help with nodule diagnosis, he said.
To establish the diagnostic accuracy of the GSC, Dr. Mazzone and colleagues evaluated data on 412 patients from three independent cohorts, all of whom had bronchoscopies for lung nodule evaluation that were nondiagnostic. Of those patients, 5% had nodules that physicians had deemed as low probability of malignancy prior to bronchoscopy, 28% deemed intermediate risk, and 74% high risk.
They found that the Percepta GSC down-classified the low–pretest risk patients with 100% negative predictive value (NPV) and down-classified intermediate–pretest risk patients with a 91.0% NPV, Dr. Mazzone reported, while patients with intermediate pretest risk were up-classified with a 65.4% positive predictive value (PPV) and patients with high pretest risk were upclassified with a 91.5% PPV.
The proportion of patients reclassified was about 55% for the low-risk group, 42% for the intermediate-risk group, and 27% for the high-risk group, according to the report at the meeting.
These results suggest the Percepta GSC could help in the “sticky situation” where a bronchoscopy result is inconclusive, Dr. Mazzone told attendees.
“When a bronchoscopy is recommended, despite fantastic advances in navigation systems to get to those nodules, we often come back without a solid answer, and that leaves the clinician in a bit of a predicament,” he said in a late-breaking clinical trial presentation.
Dr. Mazzone provided disclosures related to Veracyte, Exact Sciences, SEER, Tencent, and PCORI (research support to institution).
SOURCE: Mazzone PJ et al. CHEST 2019, Abstract. doi: 10.1016/j.chest.2019.08.307.
NEW ORLEANS – The use of a results of a recent clinical validation study suggest.
The Percepta Genomic Sequencing Classifier (GSC) was able to up- and down-classify probability of malignancy for a considerable proportion of nondiagnostic bronchoscopies in the study, Peter J. Mazzone MD, FCCP, reported at the annual meeting of the American College of Chest Physicians.
The test is seen as complementary to bronchoscopy, improving the sensitivity of bronchoscopy overall and showing a combined sensitivity of greater than 95% in low- and intermediate-risk groups, according to Dr. Mazzone.
While the clinical utility of this genomic test needs to be further tested, the eventual goal is to improve clinician decision making when bronchoscopy results don’t clearly classify nodules as malignant or benign, Dr. Mazzone said in an interview.
“In that situation, you’re often left wondering, ‘what should I do next? Can I just watch this, and see if it grows and changes, or do I have to be even more aggressive – do another biopsy, or have a surgery to take it out?’ ” he explained. “So the test hopes to help make a more informed decision by further stratifying those patients as being quite low risk and maybe safe to follow, or quite high risk and maybe you should be considering more aggressive management.”
The GSC improves on the performance of an earlier molecular test, the Percepta Bronchial Genomic Classifier, which uses a brushing of bronchial epithelium to enhance nodule management in smokers, according to the researcher.
The next-generation GSC uses 1,232 gene transcripts from whole-transcriptome RNA sequencing, along with clinical factors, to help with nodule diagnosis, he said.
To establish the diagnostic accuracy of the GSC, Dr. Mazzone and colleagues evaluated data on 412 patients from three independent cohorts, all of whom had bronchoscopies for lung nodule evaluation that were nondiagnostic. Of those patients, 5% had nodules that physicians had deemed as low probability of malignancy prior to bronchoscopy, 28% deemed intermediate risk, and 74% high risk.
They found that the Percepta GSC down-classified the low–pretest risk patients with 100% negative predictive value (NPV) and down-classified intermediate–pretest risk patients with a 91.0% NPV, Dr. Mazzone reported, while patients with intermediate pretest risk were up-classified with a 65.4% positive predictive value (PPV) and patients with high pretest risk were upclassified with a 91.5% PPV.
The proportion of patients reclassified was about 55% for the low-risk group, 42% for the intermediate-risk group, and 27% for the high-risk group, according to the report at the meeting.
These results suggest the Percepta GSC could help in the “sticky situation” where a bronchoscopy result is inconclusive, Dr. Mazzone told attendees.
“When a bronchoscopy is recommended, despite fantastic advances in navigation systems to get to those nodules, we often come back without a solid answer, and that leaves the clinician in a bit of a predicament,” he said in a late-breaking clinical trial presentation.
Dr. Mazzone provided disclosures related to Veracyte, Exact Sciences, SEER, Tencent, and PCORI (research support to institution).
SOURCE: Mazzone PJ et al. CHEST 2019, Abstract. doi: 10.1016/j.chest.2019.08.307.
NEW ORLEANS – The use of a results of a recent clinical validation study suggest.
The Percepta Genomic Sequencing Classifier (GSC) was able to up- and down-classify probability of malignancy for a considerable proportion of nondiagnostic bronchoscopies in the study, Peter J. Mazzone MD, FCCP, reported at the annual meeting of the American College of Chest Physicians.
The test is seen as complementary to bronchoscopy, improving the sensitivity of bronchoscopy overall and showing a combined sensitivity of greater than 95% in low- and intermediate-risk groups, according to Dr. Mazzone.
While the clinical utility of this genomic test needs to be further tested, the eventual goal is to improve clinician decision making when bronchoscopy results don’t clearly classify nodules as malignant or benign, Dr. Mazzone said in an interview.
“In that situation, you’re often left wondering, ‘what should I do next? Can I just watch this, and see if it grows and changes, or do I have to be even more aggressive – do another biopsy, or have a surgery to take it out?’ ” he explained. “So the test hopes to help make a more informed decision by further stratifying those patients as being quite low risk and maybe safe to follow, or quite high risk and maybe you should be considering more aggressive management.”
The GSC improves on the performance of an earlier molecular test, the Percepta Bronchial Genomic Classifier, which uses a brushing of bronchial epithelium to enhance nodule management in smokers, according to the researcher.
The next-generation GSC uses 1,232 gene transcripts from whole-transcriptome RNA sequencing, along with clinical factors, to help with nodule diagnosis, he said.
To establish the diagnostic accuracy of the GSC, Dr. Mazzone and colleagues evaluated data on 412 patients from three independent cohorts, all of whom had bronchoscopies for lung nodule evaluation that were nondiagnostic. Of those patients, 5% had nodules that physicians had deemed as low probability of malignancy prior to bronchoscopy, 28% deemed intermediate risk, and 74% high risk.
They found that the Percepta GSC down-classified the low–pretest risk patients with 100% negative predictive value (NPV) and down-classified intermediate–pretest risk patients with a 91.0% NPV, Dr. Mazzone reported, while patients with intermediate pretest risk were up-classified with a 65.4% positive predictive value (PPV) and patients with high pretest risk were upclassified with a 91.5% PPV.
The proportion of patients reclassified was about 55% for the low-risk group, 42% for the intermediate-risk group, and 27% for the high-risk group, according to the report at the meeting.
These results suggest the Percepta GSC could help in the “sticky situation” where a bronchoscopy result is inconclusive, Dr. Mazzone told attendees.
“When a bronchoscopy is recommended, despite fantastic advances in navigation systems to get to those nodules, we often come back without a solid answer, and that leaves the clinician in a bit of a predicament,” he said in a late-breaking clinical trial presentation.
Dr. Mazzone provided disclosures related to Veracyte, Exact Sciences, SEER, Tencent, and PCORI (research support to institution).
SOURCE: Mazzone PJ et al. CHEST 2019, Abstract. doi: 10.1016/j.chest.2019.08.307.
REPORTING FROM CHEST 2019
Vitamin C–based regimens in sepsis plausible, need more data, expert says
NEW ORLEANS – While further data are awaited on the role of vitamin C, thiamine, and steroids in sepsis, there is at least biologic plausibility for using the combination, and clinical equipoise that supports continued enrollment of patients in the ongoing randomized, controlled VICTAS trial, according to that study’s principal investigator.
“There is tremendous biologic plausibility for giving vitamin C in sepsis,” said Jon Sevransky, MD, professor of medicine at Emory University in Atlanta. But until more data are available on vitamin C–based regimens, those who choose to use vitamin C with thiamine and steroids in this setting need to ensure that glucose is being measured appropriately, he warned.
“If you decide that vitamin C is right for your patient, prior to having enough data – so if you’re doing a Hail Mary, or a ‘this patient is sick, and it’s probably not going to hurt them’ – please make sure that you measure your glucose with something that uses whole blood, which is either a blood gas or sending it down to the core lab, because otherwise, you might get an inaccurate result,” Dr. Sevransky said at the annual meeting of the American College of Chest Physicians.
Results from the randomized, placebo-controlled Vitamin C, Thiamine, and Steroids in Sepsis (VICTAS) trial may be available within the next few months, according to Dr. Sevransky, who noted that the trial was funded for 500 patients, which provides an 80% probability of showing an absolute risk reduction of 10% in mortality.
The primary endpoint of the phase 3 trial is vasopressor and ventilator-free days at 30 days after randomization, while 30-day mortality has been described as “the key secondary outcome” by Dr. Sevransky and colleagues in a recent report on the trial design.
Clinicians have been “captivated” by the potential benefit of vitamin C, thiamine, and hydrocortisone in patients with severe sepsis and septic shock, as published in CHEST in June 2017, Dr. Sevransky said. In that study, reported by Paul E. Marik, MD, and colleagues, hospital mortality was 8.5% for the treatment group, versus 40.4% in the control group, a significant difference.
That retrospective, single-center study had a number of limitations, however, including its before-and-after design and the use of steroids in the comparator arm. In addition, little information was available on antibiotics or fluids given at the time of the intervention, according to Dr. Sevransky.
In results of the CITRIS-ALI randomized clinical trial, just published in JAMA, intravenous administration of high-dose vitamin C in patients with sepsis and acute respiratory distress syndrome (ARDS) failed to significantly reduce organ failure scores or biomarkers of inflammation and vascular injury.
In an exploratory analysis of CITRIS-ALI, mortality at day 28 was 29.8% for the treatment group and 46.3% for placebo, with a statistically significant difference between Kaplan-Meier survival curves for the two arms, according to the investigators.
That exploratory result from CITRIS-ALI, however, is indicative of “something that needs further study,” Dr. Sevransky cautioned. “In summary, I hope I told you that biologic plausibility is present for vitamin C, thiamine, and steroids. I think that, and this is my own personal opinion, that evidence to date allows for randomization of patients, that there’s current equipoise.”
Dr. Sevransky disclosed current grant support from the Biomedical Advanced Research and Development Authority (BARDA) and the Marcus Foundation, as well as a stipend from Critical Care Medicine related to work as an associate editor. He is also a medical advisor to Project Hope and ARDS Foundation and a member of the Surviving Sepsis guideline committees.
SOURCE: Sevransky J et al. Chest 2019.
NEW ORLEANS – While further data are awaited on the role of vitamin C, thiamine, and steroids in sepsis, there is at least biologic plausibility for using the combination, and clinical equipoise that supports continued enrollment of patients in the ongoing randomized, controlled VICTAS trial, according to that study’s principal investigator.
“There is tremendous biologic plausibility for giving vitamin C in sepsis,” said Jon Sevransky, MD, professor of medicine at Emory University in Atlanta. But until more data are available on vitamin C–based regimens, those who choose to use vitamin C with thiamine and steroids in this setting need to ensure that glucose is being measured appropriately, he warned.
“If you decide that vitamin C is right for your patient, prior to having enough data – so if you’re doing a Hail Mary, or a ‘this patient is sick, and it’s probably not going to hurt them’ – please make sure that you measure your glucose with something that uses whole blood, which is either a blood gas or sending it down to the core lab, because otherwise, you might get an inaccurate result,” Dr. Sevransky said at the annual meeting of the American College of Chest Physicians.
Results from the randomized, placebo-controlled Vitamin C, Thiamine, and Steroids in Sepsis (VICTAS) trial may be available within the next few months, according to Dr. Sevransky, who noted that the trial was funded for 500 patients, which provides an 80% probability of showing an absolute risk reduction of 10% in mortality.
The primary endpoint of the phase 3 trial is vasopressor and ventilator-free days at 30 days after randomization, while 30-day mortality has been described as “the key secondary outcome” by Dr. Sevransky and colleagues in a recent report on the trial design.
Clinicians have been “captivated” by the potential benefit of vitamin C, thiamine, and hydrocortisone in patients with severe sepsis and septic shock, as published in CHEST in June 2017, Dr. Sevransky said. In that study, reported by Paul E. Marik, MD, and colleagues, hospital mortality was 8.5% for the treatment group, versus 40.4% in the control group, a significant difference.
That retrospective, single-center study had a number of limitations, however, including its before-and-after design and the use of steroids in the comparator arm. In addition, little information was available on antibiotics or fluids given at the time of the intervention, according to Dr. Sevransky.
In results of the CITRIS-ALI randomized clinical trial, just published in JAMA, intravenous administration of high-dose vitamin C in patients with sepsis and acute respiratory distress syndrome (ARDS) failed to significantly reduce organ failure scores or biomarkers of inflammation and vascular injury.
In an exploratory analysis of CITRIS-ALI, mortality at day 28 was 29.8% for the treatment group and 46.3% for placebo, with a statistically significant difference between Kaplan-Meier survival curves for the two arms, according to the investigators.
That exploratory result from CITRIS-ALI, however, is indicative of “something that needs further study,” Dr. Sevransky cautioned. “In summary, I hope I told you that biologic plausibility is present for vitamin C, thiamine, and steroids. I think that, and this is my own personal opinion, that evidence to date allows for randomization of patients, that there’s current equipoise.”
Dr. Sevransky disclosed current grant support from the Biomedical Advanced Research and Development Authority (BARDA) and the Marcus Foundation, as well as a stipend from Critical Care Medicine related to work as an associate editor. He is also a medical advisor to Project Hope and ARDS Foundation and a member of the Surviving Sepsis guideline committees.
SOURCE: Sevransky J et al. Chest 2019.
NEW ORLEANS – While further data are awaited on the role of vitamin C, thiamine, and steroids in sepsis, there is at least biologic plausibility for using the combination, and clinical equipoise that supports continued enrollment of patients in the ongoing randomized, controlled VICTAS trial, according to that study’s principal investigator.
“There is tremendous biologic plausibility for giving vitamin C in sepsis,” said Jon Sevransky, MD, professor of medicine at Emory University in Atlanta. But until more data are available on vitamin C–based regimens, those who choose to use vitamin C with thiamine and steroids in this setting need to ensure that glucose is being measured appropriately, he warned.
“If you decide that vitamin C is right for your patient, prior to having enough data – so if you’re doing a Hail Mary, or a ‘this patient is sick, and it’s probably not going to hurt them’ – please make sure that you measure your glucose with something that uses whole blood, which is either a blood gas or sending it down to the core lab, because otherwise, you might get an inaccurate result,” Dr. Sevransky said at the annual meeting of the American College of Chest Physicians.
Results from the randomized, placebo-controlled Vitamin C, Thiamine, and Steroids in Sepsis (VICTAS) trial may be available within the next few months, according to Dr. Sevransky, who noted that the trial was funded for 500 patients, which provides an 80% probability of showing an absolute risk reduction of 10% in mortality.
The primary endpoint of the phase 3 trial is vasopressor and ventilator-free days at 30 days after randomization, while 30-day mortality has been described as “the key secondary outcome” by Dr. Sevransky and colleagues in a recent report on the trial design.
Clinicians have been “captivated” by the potential benefit of vitamin C, thiamine, and hydrocortisone in patients with severe sepsis and septic shock, as published in CHEST in June 2017, Dr. Sevransky said. In that study, reported by Paul E. Marik, MD, and colleagues, hospital mortality was 8.5% for the treatment group, versus 40.4% in the control group, a significant difference.
That retrospective, single-center study had a number of limitations, however, including its before-and-after design and the use of steroids in the comparator arm. In addition, little information was available on antibiotics or fluids given at the time of the intervention, according to Dr. Sevransky.
In results of the CITRIS-ALI randomized clinical trial, just published in JAMA, intravenous administration of high-dose vitamin C in patients with sepsis and acute respiratory distress syndrome (ARDS) failed to significantly reduce organ failure scores or biomarkers of inflammation and vascular injury.
In an exploratory analysis of CITRIS-ALI, mortality at day 28 was 29.8% for the treatment group and 46.3% for placebo, with a statistically significant difference between Kaplan-Meier survival curves for the two arms, according to the investigators.
That exploratory result from CITRIS-ALI, however, is indicative of “something that needs further study,” Dr. Sevransky cautioned. “In summary, I hope I told you that biologic plausibility is present for vitamin C, thiamine, and steroids. I think that, and this is my own personal opinion, that evidence to date allows for randomization of patients, that there’s current equipoise.”
Dr. Sevransky disclosed current grant support from the Biomedical Advanced Research and Development Authority (BARDA) and the Marcus Foundation, as well as a stipend from Critical Care Medicine related to work as an associate editor. He is also a medical advisor to Project Hope and ARDS Foundation and a member of the Surviving Sepsis guideline committees.
SOURCE: Sevransky J et al. Chest 2019.
EXPERT ANALYSIS FROM CHEST 2019
CDC: Don’t vape, especially THC
Federal health officials once again are warning individuals to refrain from using all e-cigarette and vaping products, especially those containing tetrahydrocannabinol (THC).
The restated warning, issued by the Centers for Disease Control and Prevention, is based on a study of 83 patients with e-cigarette, or vaping, product use–associated lung injury (EVALI) in Utah, where researchers found several common characteristics, most strikingly the use of THC-containing products.
Fifty-three patients were interviewed by researchers. Of them, 49 (92%) reported use of THC-containing e-cigarette or vaping products during the 3 months preceding illness; 35 (66%) reported using nicotine-containing products; and 32 (60%) reported using both THC- and nicotine-containing products.
In addition, 17 (32%) patients reported exclusive use of THC-containing products, whereas only 3 (6%) reported exclusive use of nicotine-containing products. Non-medical THC use is illegal in Utah.
The median age of patients was 26 years, 3 years older than the national median; more than one-third were aged 30 years or older, according to the researchers.
Utah is seeing a higher-than-average rate of EVALI cases, with 26/million cases, compared with 4/million nationally.
Vitamin E acetate has been considered to have a suspect role in EVALI and was identified in the majority of THC cartridge samples tested in this study; however, those samples represented only six patients, according to the researchers. They added that testing of different THC cartridge samples by the Food and Drug Administration and other laboratories has shown vitamin E acetate concentrations of 31%-88% and lower-than-expected THC concentrations (14%-76% versus the typically advertised 75%-95%).
“The potential role of vitamin E acetate in lung injury remains unknown; however, the identification of vitamin E acetate among products collected from patients in Utah and elsewhere indicates that the outbreak might be associated with cutting agents or adulterants. Ascertaining the potential contribution of diluents to the current outbreak will require data from multiple states and analysis at the national level,” the researchers concluded.
The authors reported that they had no conflicts.
SOURCE: Lewis N et al. MMWR Morb Mortal Wkly Rep. Early Release. Oct. 22, 2019. 68:1-5.
Federal health officials once again are warning individuals to refrain from using all e-cigarette and vaping products, especially those containing tetrahydrocannabinol (THC).
The restated warning, issued by the Centers for Disease Control and Prevention, is based on a study of 83 patients with e-cigarette, or vaping, product use–associated lung injury (EVALI) in Utah, where researchers found several common characteristics, most strikingly the use of THC-containing products.
Fifty-three patients were interviewed by researchers. Of them, 49 (92%) reported use of THC-containing e-cigarette or vaping products during the 3 months preceding illness; 35 (66%) reported using nicotine-containing products; and 32 (60%) reported using both THC- and nicotine-containing products.
In addition, 17 (32%) patients reported exclusive use of THC-containing products, whereas only 3 (6%) reported exclusive use of nicotine-containing products. Non-medical THC use is illegal in Utah.
The median age of patients was 26 years, 3 years older than the national median; more than one-third were aged 30 years or older, according to the researchers.
Utah is seeing a higher-than-average rate of EVALI cases, with 26/million cases, compared with 4/million nationally.
Vitamin E acetate has been considered to have a suspect role in EVALI and was identified in the majority of THC cartridge samples tested in this study; however, those samples represented only six patients, according to the researchers. They added that testing of different THC cartridge samples by the Food and Drug Administration and other laboratories has shown vitamin E acetate concentrations of 31%-88% and lower-than-expected THC concentrations (14%-76% versus the typically advertised 75%-95%).
“The potential role of vitamin E acetate in lung injury remains unknown; however, the identification of vitamin E acetate among products collected from patients in Utah and elsewhere indicates that the outbreak might be associated with cutting agents or adulterants. Ascertaining the potential contribution of diluents to the current outbreak will require data from multiple states and analysis at the national level,” the researchers concluded.
The authors reported that they had no conflicts.
SOURCE: Lewis N et al. MMWR Morb Mortal Wkly Rep. Early Release. Oct. 22, 2019. 68:1-5.
Federal health officials once again are warning individuals to refrain from using all e-cigarette and vaping products, especially those containing tetrahydrocannabinol (THC).
The restated warning, issued by the Centers for Disease Control and Prevention, is based on a study of 83 patients with e-cigarette, or vaping, product use–associated lung injury (EVALI) in Utah, where researchers found several common characteristics, most strikingly the use of THC-containing products.
Fifty-three patients were interviewed by researchers. Of them, 49 (92%) reported use of THC-containing e-cigarette or vaping products during the 3 months preceding illness; 35 (66%) reported using nicotine-containing products; and 32 (60%) reported using both THC- and nicotine-containing products.
In addition, 17 (32%) patients reported exclusive use of THC-containing products, whereas only 3 (6%) reported exclusive use of nicotine-containing products. Non-medical THC use is illegal in Utah.
The median age of patients was 26 years, 3 years older than the national median; more than one-third were aged 30 years or older, according to the researchers.
Utah is seeing a higher-than-average rate of EVALI cases, with 26/million cases, compared with 4/million nationally.
Vitamin E acetate has been considered to have a suspect role in EVALI and was identified in the majority of THC cartridge samples tested in this study; however, those samples represented only six patients, according to the researchers. They added that testing of different THC cartridge samples by the Food and Drug Administration and other laboratories has shown vitamin E acetate concentrations of 31%-88% and lower-than-expected THC concentrations (14%-76% versus the typically advertised 75%-95%).
“The potential role of vitamin E acetate in lung injury remains unknown; however, the identification of vitamin E acetate among products collected from patients in Utah and elsewhere indicates that the outbreak might be associated with cutting agents or adulterants. Ascertaining the potential contribution of diluents to the current outbreak will require data from multiple states and analysis at the national level,” the researchers concluded.
The authors reported that they had no conflicts.
SOURCE: Lewis N et al. MMWR Morb Mortal Wkly Rep. Early Release. Oct. 22, 2019. 68:1-5.
FROM MMWR
Recent COPD exacerbation did not affect aclidinium’s efficacy in high-risk patients
NEW ORLEANS – A history of recent exacerbations did not significantly affect the safety or efficacy of aclidinium bromide (Tudorza) in patients with moderate to severe chronic obstructive pulmonary disease and high cardiovascular risk, analysis of a postmarketing surveillance trial suggests.
Regardless of exacerbation history, the long-acting muscarinic antagonist reduced the rate of moderate or severe COPD exacerbations versus placebo in this subgroup analysis of the phase IV ASCENT-COPD trial, presented here at the annual meeting of the American College of Chest Physicians.
At the same time, there were no significant increases in the risk of mortality or major cardiac adverse events (MACE) for those patients who had an exacerbation in the past year versus those who did not, according to investigator Robert A. Wise, MD.
Those findings may be reassuring, given that COPD patients commonly have comorbidities and cardiovascular risk factors, according to Dr. Wise, professor of medicine at the Johns Hopkins University, Baltimore.
“There’s a concern and some evidence that patients who have a propensity to COPD exacerbations may also have an increased risk for cardiovascular events,” Dr. Wise said in a podium presentation.
Accordingly, he and coinvestigators sought to tease out the impact of COPD exacerbations on safety as well as efficacy in the randomized, placebo-controlled ASCENT-COPD trial, which included 3,630 patients with moderate to severe COPD plus a cardiovascular disease history or multiple atherothrombotic risk factors.
Of the patients who were analyzed in the study, 1,433 patients had at least one treated COPD exacerbation in the year before screening for the study, while 2,156 had no exacerbations in the prior year, Dr. Wise said.
Top-line results of that study, published several months ago, showed that aclidinium did not increase MACE risk over 3 years, and reduced the rate of moderate to severe COPD exacerbations over the first year (JAMA. 2019 7 May 7;321[17]:1693-701).
In this latest analysis, presented at the meeting, risk of MACE with aclidinium treatment was not increased versus placebo, irrespective of whether they had exacerbations in the prior year (interaction P = .233); likewise, the risk of all-cause mortality was similar between groups (P = .154).
In terms of reduction in moderate or severe COPD exacerbations in the first year, aclidinium was superior to placebo both for the patients who had at least one or exacerbation in the prior year (rate ratio, 0.80) and those who had no exacerbations in the prior year (RR, 0.69).
“This translates into a number-needed-to-treat to prevent one exacerbation of about 11 patients for those without an exacerbation, compared to about 6 patients for those with a prior exacerbation,” Dr. Wise said in his presentation.
The ASCENT-COPD study was funded initially by Forest Laboratories and later by AstraZeneca and Circassia. Dr. Wise provided disclosures related to AstraZeneca, GlaxoSmithKline, Boehringer Ingelheim, Sunovion, Mylan/Theravance, Contrafect, Pearl, Merck, Verona, Novartis, AbbVie, Syneos, Regeneron, and Kiniksa.
SOURCE: Wise R et al. CHEST 2019. Abstract, doi: 10.1016/j.chest.2019.08.231.
NEW ORLEANS – A history of recent exacerbations did not significantly affect the safety or efficacy of aclidinium bromide (Tudorza) in patients with moderate to severe chronic obstructive pulmonary disease and high cardiovascular risk, analysis of a postmarketing surveillance trial suggests.
Regardless of exacerbation history, the long-acting muscarinic antagonist reduced the rate of moderate or severe COPD exacerbations versus placebo in this subgroup analysis of the phase IV ASCENT-COPD trial, presented here at the annual meeting of the American College of Chest Physicians.
At the same time, there were no significant increases in the risk of mortality or major cardiac adverse events (MACE) for those patients who had an exacerbation in the past year versus those who did not, according to investigator Robert A. Wise, MD.
Those findings may be reassuring, given that COPD patients commonly have comorbidities and cardiovascular risk factors, according to Dr. Wise, professor of medicine at the Johns Hopkins University, Baltimore.
“There’s a concern and some evidence that patients who have a propensity to COPD exacerbations may also have an increased risk for cardiovascular events,” Dr. Wise said in a podium presentation.
Accordingly, he and coinvestigators sought to tease out the impact of COPD exacerbations on safety as well as efficacy in the randomized, placebo-controlled ASCENT-COPD trial, which included 3,630 patients with moderate to severe COPD plus a cardiovascular disease history or multiple atherothrombotic risk factors.
Of the patients who were analyzed in the study, 1,433 patients had at least one treated COPD exacerbation in the year before screening for the study, while 2,156 had no exacerbations in the prior year, Dr. Wise said.
Top-line results of that study, published several months ago, showed that aclidinium did not increase MACE risk over 3 years, and reduced the rate of moderate to severe COPD exacerbations over the first year (JAMA. 2019 7 May 7;321[17]:1693-701).
In this latest analysis, presented at the meeting, risk of MACE with aclidinium treatment was not increased versus placebo, irrespective of whether they had exacerbations in the prior year (interaction P = .233); likewise, the risk of all-cause mortality was similar between groups (P = .154).
In terms of reduction in moderate or severe COPD exacerbations in the first year, aclidinium was superior to placebo both for the patients who had at least one or exacerbation in the prior year (rate ratio, 0.80) and those who had no exacerbations in the prior year (RR, 0.69).
“This translates into a number-needed-to-treat to prevent one exacerbation of about 11 patients for those without an exacerbation, compared to about 6 patients for those with a prior exacerbation,” Dr. Wise said in his presentation.
The ASCENT-COPD study was funded initially by Forest Laboratories and later by AstraZeneca and Circassia. Dr. Wise provided disclosures related to AstraZeneca, GlaxoSmithKline, Boehringer Ingelheim, Sunovion, Mylan/Theravance, Contrafect, Pearl, Merck, Verona, Novartis, AbbVie, Syneos, Regeneron, and Kiniksa.
SOURCE: Wise R et al. CHEST 2019. Abstract, doi: 10.1016/j.chest.2019.08.231.
NEW ORLEANS – A history of recent exacerbations did not significantly affect the safety or efficacy of aclidinium bromide (Tudorza) in patients with moderate to severe chronic obstructive pulmonary disease and high cardiovascular risk, analysis of a postmarketing surveillance trial suggests.
Regardless of exacerbation history, the long-acting muscarinic antagonist reduced the rate of moderate or severe COPD exacerbations versus placebo in this subgroup analysis of the phase IV ASCENT-COPD trial, presented here at the annual meeting of the American College of Chest Physicians.
At the same time, there were no significant increases in the risk of mortality or major cardiac adverse events (MACE) for those patients who had an exacerbation in the past year versus those who did not, according to investigator Robert A. Wise, MD.
Those findings may be reassuring, given that COPD patients commonly have comorbidities and cardiovascular risk factors, according to Dr. Wise, professor of medicine at the Johns Hopkins University, Baltimore.
“There’s a concern and some evidence that patients who have a propensity to COPD exacerbations may also have an increased risk for cardiovascular events,” Dr. Wise said in a podium presentation.
Accordingly, he and coinvestigators sought to tease out the impact of COPD exacerbations on safety as well as efficacy in the randomized, placebo-controlled ASCENT-COPD trial, which included 3,630 patients with moderate to severe COPD plus a cardiovascular disease history or multiple atherothrombotic risk factors.
Of the patients who were analyzed in the study, 1,433 patients had at least one treated COPD exacerbation in the year before screening for the study, while 2,156 had no exacerbations in the prior year, Dr. Wise said.
Top-line results of that study, published several months ago, showed that aclidinium did not increase MACE risk over 3 years, and reduced the rate of moderate to severe COPD exacerbations over the first year (JAMA. 2019 7 May 7;321[17]:1693-701).
In this latest analysis, presented at the meeting, risk of MACE with aclidinium treatment was not increased versus placebo, irrespective of whether they had exacerbations in the prior year (interaction P = .233); likewise, the risk of all-cause mortality was similar between groups (P = .154).
In terms of reduction in moderate or severe COPD exacerbations in the first year, aclidinium was superior to placebo both for the patients who had at least one or exacerbation in the prior year (rate ratio, 0.80) and those who had no exacerbations in the prior year (RR, 0.69).
“This translates into a number-needed-to-treat to prevent one exacerbation of about 11 patients for those without an exacerbation, compared to about 6 patients for those with a prior exacerbation,” Dr. Wise said in his presentation.
The ASCENT-COPD study was funded initially by Forest Laboratories and later by AstraZeneca and Circassia. Dr. Wise provided disclosures related to AstraZeneca, GlaxoSmithKline, Boehringer Ingelheim, Sunovion, Mylan/Theravance, Contrafect, Pearl, Merck, Verona, Novartis, AbbVie, Syneos, Regeneron, and Kiniksa.
SOURCE: Wise R et al. CHEST 2019. Abstract, doi: 10.1016/j.chest.2019.08.231.
REPORTING FROM CHEST 2019