User login
For MD-IQ only
The Effect of Radium-223 Therapy in Agent Orange-Related Prostate Carcinoma
Patients with metastatic castrate resistant prostate carcinoma (CRPC) have several treatment options, including radium-223 dichloride (Ra-223) radionuclide therapy, abiraterone, enzalutamide, and cabazitaxel. Ra-223 therapy has been reported to increase median survival in patients with bone metastatic prostate carcinoma.1,2 However, ERA 223 trial data showed an increase of bone fractures with combination of Ra-223 and abiraterone.3
Agent Orange (AO) exposure has been studied as a potential risk factor for development of prostate carcinoma. AO was a commercially manufactured defoliate that was sprayed extensively during the Vietnam War. Due to a side product of chemical manufacturing, AO was contaminated with the toxin 2,3,7,8-tetrachlorodibenzo-p-dioxin, a putative carcinogen. These dioxins can enter the food chain through soil contamination. There is enough evidence to link AO to hematologic malignancies and several solid tumors, including prostate carcinoma.4 Although no real estimates exist for what percentage of Vietnam veterans experienced AO exposure, Surveillance, Epidemiology, and End Results data showed that about 3 million veterans served in Southeast Asia where AO was used extensively in the combat theater. AO has been reported to be positively associated with a 52% increase in risk of prostate carcinoma detection at initial prostate biopsy.5
There has been no reported study of treatment efficacy in veterans with AO-related prostate carcinoma. We present a retrospective study of Ra-223 and other therapies in metastatic CRPC. The purpose of this study was to compare response to therapy and survival in veterans exposed to agent orange (AO+) vs veterans who were not exposed to (AO-) in a single US Department of Veteran Affairs (VA) medical center.
Methods
This was a retrospective study of veterans with metastatic CRPC to bones who received Ra-223 radionuclide therapy with standard dose of 50 kBq per kg of body weight and other sequential therapies at VA Pittsburgh Healthcare System (VAPHS) from January 2014 to January 2019. The purpose of this study was to measure difference in treatment outcome between AO+ veterans and AO- veterans.
Eligibility Criteria
All veterans had a history that included bone metastasis CRPC. They could have 2 to 3 small lymphadenopathies but not visceral metastasis. They received a minimum of 3 cycles and a maximum of 6 cycles of Ra-223 therapy, which was given in 4-week intervals. Pretreatment criteria was hemoglobin > 10 g/dL, platelet > 100
Statistics
Time to study was calculated from the initiation of Ra-223 therapy. Time to skeletal-related events (SRE), progression of prostate specific antigen (PSA), bone metastasis, and alkaline phosphatase (ALP) were calculated in months, using unpaired t test with 2-tailed P value. Median survival was calculated in months by Kaplan Meier R log-rank test Definition).
Results
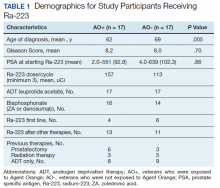
Forty-eight veterans with bone metastasis CRPC received Ra-223 therapy. Of those, 34 veterans were eligible for this retrospective study: 17 AO+ veterans and 17 AO- veterans. Mean age of diagnosis was 62 years (AO+) and 69 years (AO-) (P = .005). Mean Gleason score was 8.2 (AO+) and 8.0 (AO-) (P = .705). Veterans received initial therapy at diagnosis of prostate carcinoma, including radical prostatectomy (6 AO+ and 3 AO-), localized radiation therapy (3 AO+ and 5 AO-), and ADT (8 AO+ and 9 AO-) (Table 1).
Mean PSA at the initiation of Ra-223 therapy for AO+ was 92.8 (range, 2-551) and for AO- was 102.3 (range, 4-639; P = .86). Mean Ra-223 dose per cycle for AO+ and AO- was 157 uCi and 113 uCi, respectively. All 34 veterans received ADT (leuprolide acetate), and 30 veterans (16 AO+ and 14 AO-) received bisphosphonates (zoledronic acid or denosumab). A total of 10 veterans (29%) received Ra-223 as a first-line therapy (4 AO+ and 6 AO-), and 24 veterans (71%) received Ra-223 after hormonal or chemotherapy (13 AO+ and 11 AO-).
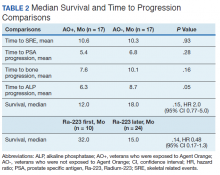
There were 12 SRE (8 AO+ and 4 AO-). Mean time to SRE for AO+ was 10.6 months and AO- was 10.3 months (P = .93). Three veterans received concurrent Ra-223 and abiraterone (participated in ERA 223 trial). Two AO+ veterans experienced SRE at 7 months and 11 months, respectively. Mean time to PSA progression for AO+ was 5.4 months and for AO- was 6.8 months (P = .28). Mean time to bone progression for AO+ and for AO- were 7.6 months and 10.1 months, respectively (P = .16). Mean time to ALP progression for AO+ and AO- were 6.3 months and 8.7 months, respectively (P = .05). (Table 2). The treatment pattern of AO+ and AO- is depicted on a swimmer plot (Figures 1 and 2).
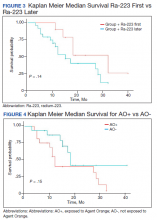
Twenty veterans (58%) had died: 13 AO+ and 7 AO- veterans. Median survival for Ra-223 first and Ra-223 later was was 32 months and 15 months, respectively (P = .14; hazard ratio [HR], 0.48). Overall median survival for AO+ veterans and AO- veterans were 12 months and 18 months, respectively (P = .15; HR, 2.0) (Figures 3 and 4).
Discussions
There has been no reported VA study of using Ra-223 and other therapies (hormonal and chemotherapy) in veterans exposed to AO. This is the first retrospective study to compare the response and survival between AO+ and AO- veterans. Even though this study featured a small sample, it is interesting to note the difference between those 2 populations. There was 1 prior study in veterans with prostate carcinoma using radiotherapy (brachytherapy) in early-stage disease. Everly and colleagues reported that AO+ veterans were less likely to remain biochemically controlled compared with AO- and nonveteran patients with prostate carcinoma.4
Ansbaugh and colleagues reported that AO was associated with a 75% increase in the risk of Gleason ≥ 7 and a 110% increase in Gleason ≥ 8. AO+ veterans are at risk for the detection of high-grade prostate carcinoma. They also tend to have an average age of diagnosis that is 4 to 5 years younger than AO- veterans.6
Our study revealed that AO+ veterans were diagnosed at a younger age (mean 62 years) compared with that of AO- veterans (mean 69 years, P = .005). We also proved that AO veterans have a higher mean Gleason score (8.2) compared with that of AO- veterans (8.0). Veterans received therapy at the time of diagnosis of prostate carcinoma with either radical prostatectomy, radiation therapy, or ADT with leuprolide acetate. Mean PSA at the start of Ra-223 therapy for AO+ was 92.8 (range, 2-551); for AO- was 102.3 (range, 4-639), which is not statistically significant.
Ra-223, an
In a phase 3, randomized, double-blind, placebo-controlled study by Parker and colleagues (ALSYMPCA study), 921 patients who had received, were not eligible to receive, or declined docetaxel, in a 2:1 ratio, were randomized to receive 6 injections of Ra-223 or matching placebo.2 Ra-223 significantly improved overall survival (OS) (median, 14.9 months vs 11.3 months) compared with that of placebo. Ra-223 also prolonged the time to the first symptomatic SRE (median, 15.6 months vs 9.8 months), the time to an increase in the total ALP level (median 7.4 months vs 3.8 months), and the time to an increase in the PSA level (median 3.6 months vs 3.4 months).2
In our study, the mean time to SRE for AO+ was 10.6 months and AO- was 10.3 months (P = .93). Mean time to PSA progression for AO+ was 5.4 months and for AO- was 6.8 months (P = .28). Mean time to bone progression for AO+ and for AO- were 7.6 months and 10.1 months respectively (P = .16). Mean time to ALP progression for AO+ and AO- were 6.3 months and 8.7 months respectively (P = .05). There is a trend of shorter PSA progression, bone progression, and ALP progression in AO+ veterans, though these were not statistically significant due to small sample population. In our study the median survival in for AO- was 18 months and for AO+ was 12 months, which is comparable with median survival of 14.9 months from the ALSYMPCA study.
There were 12 veterans who developed SREs. All received radiation therapy due to bone progression or impending fracture. AO+ veterans developed more SREs (n = 8) when compared with AO- veterans (n = 4). There were more AO- veterans alive (n = 10) than there were AO+ veterans (n = 4). The plausible explanation of this may be due to the aggressive pattern of prostate carcinoma in AO+ veterans (younger age and higher Gleason score).
VAPHS participated in the ERA trial between 2014 and 2016. The trial enrolled 806 patients who were randomly assigned to receive first-line Ra-223 or placebo in addition to abiraterone acetate plus prednisone.3 The study was unblinded prematurely after more fractures and deaths were noted in the Ra-223 and abiraterone group than there were in the placebo and abiraterone group. Median symptomatic SRE was 22.3 months in the Ra-223 group and 26.0 months in the placebo group. Fractures (any grade) occurred in 29% in the Ra-223 group and 11% in the placebo group. It was suggested that Ra-223 could contribute to the risk of osteoporotic fractures in patients with bone metastatic prostate carcinoma. Median OS was 30.7 months in the Ra-223 group and 33.3 months in the placebo group.
We enrolled 3 veterans in the ERA clinical trial. Two AO+ veterans had SREs at 7 months and 11 months. In our study, the median OS for Ra-223 first line was 32 months, which is comparable with median survival of 30.7 months from ERA-223 study. Median survival for Ra-223 later was only 15 months. We recommend veterans with at least 2 to 3-bone metastasis receive Ra-223 in the first-line setting rather than second- or third-line setting. In this retrospective study with Ra-223 and other therapies, we proved that AO+ veterans have a worse response and OS when compared with that of AO- veterans.
Conclusions
This is the first VA study to compare the efficacy of Ra-223 and other therapies in metastatic CRPC between AO+ and AO- veterans. AO+ veterans were diagnosed at a younger age and had higher Gleason scores. There was no statistical difference between AO+ and AO- veterans in terms of time to SRE, PSA progression, and bone and ALP progression even though there was a trend of shorter duration in AO+ veterans. There was no median survival difference between veterans who received Ra-223 first vs Ra-223 later as well as between AO+ and AO- veterans, but there was a trend of worse survival in veteran who received Ra-223 later and AO+ veterans.
This study showed that AO+ veterans have a shorter duration of response to therapy and shorter median survival compared with that of AO- veterans. We recommend that veterans should get Ra-223 in the first-line setting rather than after hormonal therapy and chemotherapy because their marrows are still intact. We need to investigate further whether veterans that exposed to carcinogen 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) may have different molecular biology and as such may cause inferior efficacy in the treatment of prostate carcinoma.
1. Shore ND. Radium-223 dichloride for metastatic castration-resistant prostate cancer: the urologist’s perspective. Urology. 2015;85(4):717-724. doi:10.1016/j.urology.2014.11.031
2. Parker C, Nilsson S, Heinrich D, et al; ALSYMPCA Investigators. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013;369(3):213-223. doi:10.1056/NEJMoa1213755
3. Smith M, Parker C, Saad F, et al. Addition of radium-223 to abiraterone acetate and prednisone or prednisolone in patients with castration-resistant prostate cancer and bone metastases (ERA 223): a randomized, double-blind, placebo-controlled, phase 3 trial [published correction appears in Lancet Oncol. 2019 Oct;20(10):e559]. Lancet Oncol. 2019;20(3):408-419. doi:10.1016/S1470-2045(18)30860-X
4. Everly L, Merrick GS, Allen ZA, et al. Prostate cancer control and survival in Vietnam veterans exposed to Agent Orange. Brachytherapy. 2009;8(1):57-62. doi: 10.1016/j.brachy.2008.08.001
5. Altekruse S. SEER Cancer Statistics Review, 1975-2017 Bethesda, MD: National Cancer Institute. 2009. 6. Ansbaugh N, Shannon J, Mori M, Farris PE, Garzotto M. Agent Orange as a risk factor for high-grade prostate cancer. Cancer. 2013;119(13):2399-2404. doi:10.1002/cncr.27941
7. Jadvar H, Quinn DI. Targeted α-particle therapy of bone metastases in prostate cancer. Clin Nucl Med. 2013;38(12):966-971. doi:10.1097/RLU.0000000000000290
Patients with metastatic castrate resistant prostate carcinoma (CRPC) have several treatment options, including radium-223 dichloride (Ra-223) radionuclide therapy, abiraterone, enzalutamide, and cabazitaxel. Ra-223 therapy has been reported to increase median survival in patients with bone metastatic prostate carcinoma.1,2 However, ERA 223 trial data showed an increase of bone fractures with combination of Ra-223 and abiraterone.3
Agent Orange (AO) exposure has been studied as a potential risk factor for development of prostate carcinoma. AO was a commercially manufactured defoliate that was sprayed extensively during the Vietnam War. Due to a side product of chemical manufacturing, AO was contaminated with the toxin 2,3,7,8-tetrachlorodibenzo-p-dioxin, a putative carcinogen. These dioxins can enter the food chain through soil contamination. There is enough evidence to link AO to hematologic malignancies and several solid tumors, including prostate carcinoma.4 Although no real estimates exist for what percentage of Vietnam veterans experienced AO exposure, Surveillance, Epidemiology, and End Results data showed that about 3 million veterans served in Southeast Asia where AO was used extensively in the combat theater. AO has been reported to be positively associated with a 52% increase in risk of prostate carcinoma detection at initial prostate biopsy.5
There has been no reported study of treatment efficacy in veterans with AO-related prostate carcinoma. We present a retrospective study of Ra-223 and other therapies in metastatic CRPC. The purpose of this study was to compare response to therapy and survival in veterans exposed to agent orange (AO+) vs veterans who were not exposed to (AO-) in a single US Department of Veteran Affairs (VA) medical center.
Methods
This was a retrospective study of veterans with metastatic CRPC to bones who received Ra-223 radionuclide therapy with standard dose of 50 kBq per kg of body weight and other sequential therapies at VA Pittsburgh Healthcare System (VAPHS) from January 2014 to January 2019. The purpose of this study was to measure difference in treatment outcome between AO+ veterans and AO- veterans.
Eligibility Criteria
All veterans had a history that included bone metastasis CRPC. They could have 2 to 3 small lymphadenopathies but not visceral metastasis. They received a minimum of 3 cycles and a maximum of 6 cycles of Ra-223 therapy, which was given in 4-week intervals. Pretreatment criteria was hemoglobin > 10 g/dL, platelet > 100
Statistics
Time to study was calculated from the initiation of Ra-223 therapy. Time to skeletal-related events (SRE), progression of prostate specific antigen (PSA), bone metastasis, and alkaline phosphatase (ALP) were calculated in months, using unpaired t test with 2-tailed P value. Median survival was calculated in months by Kaplan Meier R log-rank test Definition).
Results
Forty-eight veterans with bone metastasis CRPC received Ra-223 therapy. Of those, 34 veterans were eligible for this retrospective study: 17 AO+ veterans and 17 AO- veterans. Mean age of diagnosis was 62 years (AO+) and 69 years (AO-) (P = .005). Mean Gleason score was 8.2 (AO+) and 8.0 (AO-) (P = .705). Veterans received initial therapy at diagnosis of prostate carcinoma, including radical prostatectomy (6 AO+ and 3 AO-), localized radiation therapy (3 AO+ and 5 AO-), and ADT (8 AO+ and 9 AO-) (Table 1).
Mean PSA at the initiation of Ra-223 therapy for AO+ was 92.8 (range, 2-551) and for AO- was 102.3 (range, 4-639; P = .86). Mean Ra-223 dose per cycle for AO+ and AO- was 157 uCi and 113 uCi, respectively. All 34 veterans received ADT (leuprolide acetate), and 30 veterans (16 AO+ and 14 AO-) received bisphosphonates (zoledronic acid or denosumab). A total of 10 veterans (29%) received Ra-223 as a first-line therapy (4 AO+ and 6 AO-), and 24 veterans (71%) received Ra-223 after hormonal or chemotherapy (13 AO+ and 11 AO-).
There were 12 SRE (8 AO+ and 4 AO-). Mean time to SRE for AO+ was 10.6 months and AO- was 10.3 months (P = .93). Three veterans received concurrent Ra-223 and abiraterone (participated in ERA 223 trial). Two AO+ veterans experienced SRE at 7 months and 11 months, respectively. Mean time to PSA progression for AO+ was 5.4 months and for AO- was 6.8 months (P = .28). Mean time to bone progression for AO+ and for AO- were 7.6 months and 10.1 months, respectively (P = .16). Mean time to ALP progression for AO+ and AO- were 6.3 months and 8.7 months, respectively (P = .05). (Table 2). The treatment pattern of AO+ and AO- is depicted on a swimmer plot (Figures 1 and 2).
Twenty veterans (58%) had died: 13 AO+ and 7 AO- veterans. Median survival for Ra-223 first and Ra-223 later was was 32 months and 15 months, respectively (P = .14; hazard ratio [HR], 0.48). Overall median survival for AO+ veterans and AO- veterans were 12 months and 18 months, respectively (P = .15; HR, 2.0) (Figures 3 and 4).
Discussions
There has been no reported VA study of using Ra-223 and other therapies (hormonal and chemotherapy) in veterans exposed to AO. This is the first retrospective study to compare the response and survival between AO+ and AO- veterans. Even though this study featured a small sample, it is interesting to note the difference between those 2 populations. There was 1 prior study in veterans with prostate carcinoma using radiotherapy (brachytherapy) in early-stage disease. Everly and colleagues reported that AO+ veterans were less likely to remain biochemically controlled compared with AO- and nonveteran patients with prostate carcinoma.4
Ansbaugh and colleagues reported that AO was associated with a 75% increase in the risk of Gleason ≥ 7 and a 110% increase in Gleason ≥ 8. AO+ veterans are at risk for the detection of high-grade prostate carcinoma. They also tend to have an average age of diagnosis that is 4 to 5 years younger than AO- veterans.6
Our study revealed that AO+ veterans were diagnosed at a younger age (mean 62 years) compared with that of AO- veterans (mean 69 years, P = .005). We also proved that AO veterans have a higher mean Gleason score (8.2) compared with that of AO- veterans (8.0). Veterans received therapy at the time of diagnosis of prostate carcinoma with either radical prostatectomy, radiation therapy, or ADT with leuprolide acetate. Mean PSA at the start of Ra-223 therapy for AO+ was 92.8 (range, 2-551); for AO- was 102.3 (range, 4-639), which is not statistically significant.
Ra-223, an
In a phase 3, randomized, double-blind, placebo-controlled study by Parker and colleagues (ALSYMPCA study), 921 patients who had received, were not eligible to receive, or declined docetaxel, in a 2:1 ratio, were randomized to receive 6 injections of Ra-223 or matching placebo.2 Ra-223 significantly improved overall survival (OS) (median, 14.9 months vs 11.3 months) compared with that of placebo. Ra-223 also prolonged the time to the first symptomatic SRE (median, 15.6 months vs 9.8 months), the time to an increase in the total ALP level (median 7.4 months vs 3.8 months), and the time to an increase in the PSA level (median 3.6 months vs 3.4 months).2
In our study, the mean time to SRE for AO+ was 10.6 months and AO- was 10.3 months (P = .93). Mean time to PSA progression for AO+ was 5.4 months and for AO- was 6.8 months (P = .28). Mean time to bone progression for AO+ and for AO- were 7.6 months and 10.1 months respectively (P = .16). Mean time to ALP progression for AO+ and AO- were 6.3 months and 8.7 months respectively (P = .05). There is a trend of shorter PSA progression, bone progression, and ALP progression in AO+ veterans, though these were not statistically significant due to small sample population. In our study the median survival in for AO- was 18 months and for AO+ was 12 months, which is comparable with median survival of 14.9 months from the ALSYMPCA study.
There were 12 veterans who developed SREs. All received radiation therapy due to bone progression or impending fracture. AO+ veterans developed more SREs (n = 8) when compared with AO- veterans (n = 4). There were more AO- veterans alive (n = 10) than there were AO+ veterans (n = 4). The plausible explanation of this may be due to the aggressive pattern of prostate carcinoma in AO+ veterans (younger age and higher Gleason score).
VAPHS participated in the ERA trial between 2014 and 2016. The trial enrolled 806 patients who were randomly assigned to receive first-line Ra-223 or placebo in addition to abiraterone acetate plus prednisone.3 The study was unblinded prematurely after more fractures and deaths were noted in the Ra-223 and abiraterone group than there were in the placebo and abiraterone group. Median symptomatic SRE was 22.3 months in the Ra-223 group and 26.0 months in the placebo group. Fractures (any grade) occurred in 29% in the Ra-223 group and 11% in the placebo group. It was suggested that Ra-223 could contribute to the risk of osteoporotic fractures in patients with bone metastatic prostate carcinoma. Median OS was 30.7 months in the Ra-223 group and 33.3 months in the placebo group.
We enrolled 3 veterans in the ERA clinical trial. Two AO+ veterans had SREs at 7 months and 11 months. In our study, the median OS for Ra-223 first line was 32 months, which is comparable with median survival of 30.7 months from ERA-223 study. Median survival for Ra-223 later was only 15 months. We recommend veterans with at least 2 to 3-bone metastasis receive Ra-223 in the first-line setting rather than second- or third-line setting. In this retrospective study with Ra-223 and other therapies, we proved that AO+ veterans have a worse response and OS when compared with that of AO- veterans.
Conclusions
This is the first VA study to compare the efficacy of Ra-223 and other therapies in metastatic CRPC between AO+ and AO- veterans. AO+ veterans were diagnosed at a younger age and had higher Gleason scores. There was no statistical difference between AO+ and AO- veterans in terms of time to SRE, PSA progression, and bone and ALP progression even though there was a trend of shorter duration in AO+ veterans. There was no median survival difference between veterans who received Ra-223 first vs Ra-223 later as well as between AO+ and AO- veterans, but there was a trend of worse survival in veteran who received Ra-223 later and AO+ veterans.
This study showed that AO+ veterans have a shorter duration of response to therapy and shorter median survival compared with that of AO- veterans. We recommend that veterans should get Ra-223 in the first-line setting rather than after hormonal therapy and chemotherapy because their marrows are still intact. We need to investigate further whether veterans that exposed to carcinogen 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) may have different molecular biology and as such may cause inferior efficacy in the treatment of prostate carcinoma.
Patients with metastatic castrate resistant prostate carcinoma (CRPC) have several treatment options, including radium-223 dichloride (Ra-223) radionuclide therapy, abiraterone, enzalutamide, and cabazitaxel. Ra-223 therapy has been reported to increase median survival in patients with bone metastatic prostate carcinoma.1,2 However, ERA 223 trial data showed an increase of bone fractures with combination of Ra-223 and abiraterone.3
Agent Orange (AO) exposure has been studied as a potential risk factor for development of prostate carcinoma. AO was a commercially manufactured defoliate that was sprayed extensively during the Vietnam War. Due to a side product of chemical manufacturing, AO was contaminated with the toxin 2,3,7,8-tetrachlorodibenzo-p-dioxin, a putative carcinogen. These dioxins can enter the food chain through soil contamination. There is enough evidence to link AO to hematologic malignancies and several solid tumors, including prostate carcinoma.4 Although no real estimates exist for what percentage of Vietnam veterans experienced AO exposure, Surveillance, Epidemiology, and End Results data showed that about 3 million veterans served in Southeast Asia where AO was used extensively in the combat theater. AO has been reported to be positively associated with a 52% increase in risk of prostate carcinoma detection at initial prostate biopsy.5
There has been no reported study of treatment efficacy in veterans with AO-related prostate carcinoma. We present a retrospective study of Ra-223 and other therapies in metastatic CRPC. The purpose of this study was to compare response to therapy and survival in veterans exposed to agent orange (AO+) vs veterans who were not exposed to (AO-) in a single US Department of Veteran Affairs (VA) medical center.
Methods
This was a retrospective study of veterans with metastatic CRPC to bones who received Ra-223 radionuclide therapy with standard dose of 50 kBq per kg of body weight and other sequential therapies at VA Pittsburgh Healthcare System (VAPHS) from January 2014 to January 2019. The purpose of this study was to measure difference in treatment outcome between AO+ veterans and AO- veterans.
Eligibility Criteria
All veterans had a history that included bone metastasis CRPC. They could have 2 to 3 small lymphadenopathies but not visceral metastasis. They received a minimum of 3 cycles and a maximum of 6 cycles of Ra-223 therapy, which was given in 4-week intervals. Pretreatment criteria was hemoglobin > 10 g/dL, platelet > 100
Statistics
Time to study was calculated from the initiation of Ra-223 therapy. Time to skeletal-related events (SRE), progression of prostate specific antigen (PSA), bone metastasis, and alkaline phosphatase (ALP) were calculated in months, using unpaired t test with 2-tailed P value. Median survival was calculated in months by Kaplan Meier R log-rank test Definition).
Results
Forty-eight veterans with bone metastasis CRPC received Ra-223 therapy. Of those, 34 veterans were eligible for this retrospective study: 17 AO+ veterans and 17 AO- veterans. Mean age of diagnosis was 62 years (AO+) and 69 years (AO-) (P = .005). Mean Gleason score was 8.2 (AO+) and 8.0 (AO-) (P = .705). Veterans received initial therapy at diagnosis of prostate carcinoma, including radical prostatectomy (6 AO+ and 3 AO-), localized radiation therapy (3 AO+ and 5 AO-), and ADT (8 AO+ and 9 AO-) (Table 1).
Mean PSA at the initiation of Ra-223 therapy for AO+ was 92.8 (range, 2-551) and for AO- was 102.3 (range, 4-639; P = .86). Mean Ra-223 dose per cycle for AO+ and AO- was 157 uCi and 113 uCi, respectively. All 34 veterans received ADT (leuprolide acetate), and 30 veterans (16 AO+ and 14 AO-) received bisphosphonates (zoledronic acid or denosumab). A total of 10 veterans (29%) received Ra-223 as a first-line therapy (4 AO+ and 6 AO-), and 24 veterans (71%) received Ra-223 after hormonal or chemotherapy (13 AO+ and 11 AO-).
There were 12 SRE (8 AO+ and 4 AO-). Mean time to SRE for AO+ was 10.6 months and AO- was 10.3 months (P = .93). Three veterans received concurrent Ra-223 and abiraterone (participated in ERA 223 trial). Two AO+ veterans experienced SRE at 7 months and 11 months, respectively. Mean time to PSA progression for AO+ was 5.4 months and for AO- was 6.8 months (P = .28). Mean time to bone progression for AO+ and for AO- were 7.6 months and 10.1 months, respectively (P = .16). Mean time to ALP progression for AO+ and AO- were 6.3 months and 8.7 months, respectively (P = .05). (Table 2). The treatment pattern of AO+ and AO- is depicted on a swimmer plot (Figures 1 and 2).
Twenty veterans (58%) had died: 13 AO+ and 7 AO- veterans. Median survival for Ra-223 first and Ra-223 later was was 32 months and 15 months, respectively (P = .14; hazard ratio [HR], 0.48). Overall median survival for AO+ veterans and AO- veterans were 12 months and 18 months, respectively (P = .15; HR, 2.0) (Figures 3 and 4).
Discussions
There has been no reported VA study of using Ra-223 and other therapies (hormonal and chemotherapy) in veterans exposed to AO. This is the first retrospective study to compare the response and survival between AO+ and AO- veterans. Even though this study featured a small sample, it is interesting to note the difference between those 2 populations. There was 1 prior study in veterans with prostate carcinoma using radiotherapy (brachytherapy) in early-stage disease. Everly and colleagues reported that AO+ veterans were less likely to remain biochemically controlled compared with AO- and nonveteran patients with prostate carcinoma.4
Ansbaugh and colleagues reported that AO was associated with a 75% increase in the risk of Gleason ≥ 7 and a 110% increase in Gleason ≥ 8. AO+ veterans are at risk for the detection of high-grade prostate carcinoma. They also tend to have an average age of diagnosis that is 4 to 5 years younger than AO- veterans.6
Our study revealed that AO+ veterans were diagnosed at a younger age (mean 62 years) compared with that of AO- veterans (mean 69 years, P = .005). We also proved that AO veterans have a higher mean Gleason score (8.2) compared with that of AO- veterans (8.0). Veterans received therapy at the time of diagnosis of prostate carcinoma with either radical prostatectomy, radiation therapy, or ADT with leuprolide acetate. Mean PSA at the start of Ra-223 therapy for AO+ was 92.8 (range, 2-551); for AO- was 102.3 (range, 4-639), which is not statistically significant.
Ra-223, an
In a phase 3, randomized, double-blind, placebo-controlled study by Parker and colleagues (ALSYMPCA study), 921 patients who had received, were not eligible to receive, or declined docetaxel, in a 2:1 ratio, were randomized to receive 6 injections of Ra-223 or matching placebo.2 Ra-223 significantly improved overall survival (OS) (median, 14.9 months vs 11.3 months) compared with that of placebo. Ra-223 also prolonged the time to the first symptomatic SRE (median, 15.6 months vs 9.8 months), the time to an increase in the total ALP level (median 7.4 months vs 3.8 months), and the time to an increase in the PSA level (median 3.6 months vs 3.4 months).2
In our study, the mean time to SRE for AO+ was 10.6 months and AO- was 10.3 months (P = .93). Mean time to PSA progression for AO+ was 5.4 months and for AO- was 6.8 months (P = .28). Mean time to bone progression for AO+ and for AO- were 7.6 months and 10.1 months respectively (P = .16). Mean time to ALP progression for AO+ and AO- were 6.3 months and 8.7 months respectively (P = .05). There is a trend of shorter PSA progression, bone progression, and ALP progression in AO+ veterans, though these were not statistically significant due to small sample population. In our study the median survival in for AO- was 18 months and for AO+ was 12 months, which is comparable with median survival of 14.9 months from the ALSYMPCA study.
There were 12 veterans who developed SREs. All received radiation therapy due to bone progression or impending fracture. AO+ veterans developed more SREs (n = 8) when compared with AO- veterans (n = 4). There were more AO- veterans alive (n = 10) than there were AO+ veterans (n = 4). The plausible explanation of this may be due to the aggressive pattern of prostate carcinoma in AO+ veterans (younger age and higher Gleason score).
VAPHS participated in the ERA trial between 2014 and 2016. The trial enrolled 806 patients who were randomly assigned to receive first-line Ra-223 or placebo in addition to abiraterone acetate plus prednisone.3 The study was unblinded prematurely after more fractures and deaths were noted in the Ra-223 and abiraterone group than there were in the placebo and abiraterone group. Median symptomatic SRE was 22.3 months in the Ra-223 group and 26.0 months in the placebo group. Fractures (any grade) occurred in 29% in the Ra-223 group and 11% in the placebo group. It was suggested that Ra-223 could contribute to the risk of osteoporotic fractures in patients with bone metastatic prostate carcinoma. Median OS was 30.7 months in the Ra-223 group and 33.3 months in the placebo group.
We enrolled 3 veterans in the ERA clinical trial. Two AO+ veterans had SREs at 7 months and 11 months. In our study, the median OS for Ra-223 first line was 32 months, which is comparable with median survival of 30.7 months from ERA-223 study. Median survival for Ra-223 later was only 15 months. We recommend veterans with at least 2 to 3-bone metastasis receive Ra-223 in the first-line setting rather than second- or third-line setting. In this retrospective study with Ra-223 and other therapies, we proved that AO+ veterans have a worse response and OS when compared with that of AO- veterans.
Conclusions
This is the first VA study to compare the efficacy of Ra-223 and other therapies in metastatic CRPC between AO+ and AO- veterans. AO+ veterans were diagnosed at a younger age and had higher Gleason scores. There was no statistical difference between AO+ and AO- veterans in terms of time to SRE, PSA progression, and bone and ALP progression even though there was a trend of shorter duration in AO+ veterans. There was no median survival difference between veterans who received Ra-223 first vs Ra-223 later as well as between AO+ and AO- veterans, but there was a trend of worse survival in veteran who received Ra-223 later and AO+ veterans.
This study showed that AO+ veterans have a shorter duration of response to therapy and shorter median survival compared with that of AO- veterans. We recommend that veterans should get Ra-223 in the first-line setting rather than after hormonal therapy and chemotherapy because their marrows are still intact. We need to investigate further whether veterans that exposed to carcinogen 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) may have different molecular biology and as such may cause inferior efficacy in the treatment of prostate carcinoma.
1. Shore ND. Radium-223 dichloride for metastatic castration-resistant prostate cancer: the urologist’s perspective. Urology. 2015;85(4):717-724. doi:10.1016/j.urology.2014.11.031
2. Parker C, Nilsson S, Heinrich D, et al; ALSYMPCA Investigators. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013;369(3):213-223. doi:10.1056/NEJMoa1213755
3. Smith M, Parker C, Saad F, et al. Addition of radium-223 to abiraterone acetate and prednisone or prednisolone in patients with castration-resistant prostate cancer and bone metastases (ERA 223): a randomized, double-blind, placebo-controlled, phase 3 trial [published correction appears in Lancet Oncol. 2019 Oct;20(10):e559]. Lancet Oncol. 2019;20(3):408-419. doi:10.1016/S1470-2045(18)30860-X
4. Everly L, Merrick GS, Allen ZA, et al. Prostate cancer control and survival in Vietnam veterans exposed to Agent Orange. Brachytherapy. 2009;8(1):57-62. doi: 10.1016/j.brachy.2008.08.001
5. Altekruse S. SEER Cancer Statistics Review, 1975-2017 Bethesda, MD: National Cancer Institute. 2009. 6. Ansbaugh N, Shannon J, Mori M, Farris PE, Garzotto M. Agent Orange as a risk factor for high-grade prostate cancer. Cancer. 2013;119(13):2399-2404. doi:10.1002/cncr.27941
7. Jadvar H, Quinn DI. Targeted α-particle therapy of bone metastases in prostate cancer. Clin Nucl Med. 2013;38(12):966-971. doi:10.1097/RLU.0000000000000290
1. Shore ND. Radium-223 dichloride for metastatic castration-resistant prostate cancer: the urologist’s perspective. Urology. 2015;85(4):717-724. doi:10.1016/j.urology.2014.11.031
2. Parker C, Nilsson S, Heinrich D, et al; ALSYMPCA Investigators. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013;369(3):213-223. doi:10.1056/NEJMoa1213755
3. Smith M, Parker C, Saad F, et al. Addition of radium-223 to abiraterone acetate and prednisone or prednisolone in patients with castration-resistant prostate cancer and bone metastases (ERA 223): a randomized, double-blind, placebo-controlled, phase 3 trial [published correction appears in Lancet Oncol. 2019 Oct;20(10):e559]. Lancet Oncol. 2019;20(3):408-419. doi:10.1016/S1470-2045(18)30860-X
4. Everly L, Merrick GS, Allen ZA, et al. Prostate cancer control and survival in Vietnam veterans exposed to Agent Orange. Brachytherapy. 2009;8(1):57-62. doi: 10.1016/j.brachy.2008.08.001
5. Altekruse S. SEER Cancer Statistics Review, 1975-2017 Bethesda, MD: National Cancer Institute. 2009. 6. Ansbaugh N, Shannon J, Mori M, Farris PE, Garzotto M. Agent Orange as a risk factor for high-grade prostate cancer. Cancer. 2013;119(13):2399-2404. doi:10.1002/cncr.27941
7. Jadvar H, Quinn DI. Targeted α-particle therapy of bone metastases in prostate cancer. Clin Nucl Med. 2013;38(12):966-971. doi:10.1097/RLU.0000000000000290
Cancer therapy affects sexual health in most patients
Sexual dysfunction is a common treatment-related problem observed across numerous cancer diagnoses, and a new survey finds that 87% of cancer survivors have had such problems.
However, most of them also reported that their oncologist had not formally discussed the topic, and female patients were particularly unlikely to be asked about sexual dysfunction.
“The main takeaway from our study is that sexual side effects following treatment are very common,” said lead author James Taylor, MD, MPH, chief resident in radiation oncology at the Sidney Kimmel Cancer Center at Thomas Jefferson University in Philadelphia, Pennsylvania.
“Nearly 9 in 10 patients reported some change after cancer treatment that negatively affected their sexual health,” he said.
Taylor was speaking at the American Society for Radiation Oncology (ASTRO) Annual Meeting, held virtually this year because of the pandemic.
“Negative effects on sexual health after cancer treatment are unfortunately very common,” he said. “This is not just patients treated with radiation but this includes chemotherapy, hormonal therapy, surgery, and other treatment modalities.”
Potential issues include physical complications such as erectile dysfunction with prostate cancer treatment or vaginal dryness with gynecological cancer treatment. One recent study found that one-third of men who had undergone treatment for prostate cancer reported that a subsequent lack of sexual function has had the greatest impact on their quality of life. Another study reported that nearly all patients with breast cancer taking endocrine therapy experience a high degree of sexual dysfunction, including vulvovaginal dryness and severe dyspareunia.
Not discussed, not warned
Taylor and colleagues developed a questionnaire with input from radiation oncologists, medical oncologists, and surgeons, which consisted of more than 25 questions and was specifically targeted at cancer survivors.
A total of 405 adults completed the electronic survey about their experiences with sexual side effects after cancer treatment (391 responses were eligible for analysis). Most of the respondents were women (81%), and the most common cancer types were breast (67%), prostate (16%), and endometrial (6%). Treatments included chemotherapy (78%), radiation therapy (54%), and hormone therapy (47%).
“The questionnaires were distributed at Thomas Jefferson and throughout social media,” said Taylor. “The responses from social media are important because it shows a broad representation of patients who are treated in multiple clinics across the United States.”
Most of the survivors who responded (n = 337, 87%) stated cancer treatment had impacted sexual function or desire, with 53.8% reporting body image distortion, 73.4% with dyspareunia, and 42.3% unable to achieve orgasm.
Only about one-quarter (27.9%) said they had been formally asked about their sexual health by their clinician.
“Only about 40% said that they have been preemptively warned that their sexual health may be affected by treatment,” said Taylor.
Women were far less likely to be asked about their sexual health by their provider. The survey showed that male respondents were twice as likely to say they had been asked about sexual health and counseled about the potential toxicity (53% vs 22%; P < .001), and a substantially higher percentage of men reported receiving a formal assessment tool such as a survey (32% vs 5%; P = .001) compared with female respondents.
Taylor noted that the survey demonstrated several things. “One is that sexual toxicity is exceedingly common, and number two, it identified a gender disparity,” he said. “But number 3, and I think that this is an important aspect of our study, is that the majority of respondents felt that they would like a standard questionnaire to initiate and guide a discussion on sexual health with their provider.”
The reason that aspect is very important, he emphasized, is that “we know metrics and questionnaires already exist, so this gives us an actionable intervention that we can distribute and help mitigate some of these disparities.”
Importance of being holistic
The results of the survey “highlight the importance of being holistic in our approach to patient survivorship,” commented Karen Winkfield, MD, PhD, associate professor of radiation oncology at Wake Forest University, Winston-Salem, North Carolina, and executive director of the Meharry-Vanderbilt Alliance, Nashville, Tennessee.
“We need to ask patients about all parts of their well-being, including sexual health,” Winkfield said. “Body dysmorphism can impact anyone, but especially patients who have had surgery or radiation,” she said, while chemotherapy can impact energy and libido and have other toxicities that impact sexual health.
“I encourage all oncologists to ask patients about their sexual health, and a standardized form that can be used across all sites will make this much easier,” Winkfield commented. “We owe it to our patients to treat them holistically.”
The authors have reported no relevant financial relationships.
This article first appeared on Medscape.com.
Sexual dysfunction is a common treatment-related problem observed across numerous cancer diagnoses, and a new survey finds that 87% of cancer survivors have had such problems.
However, most of them also reported that their oncologist had not formally discussed the topic, and female patients were particularly unlikely to be asked about sexual dysfunction.
“The main takeaway from our study is that sexual side effects following treatment are very common,” said lead author James Taylor, MD, MPH, chief resident in radiation oncology at the Sidney Kimmel Cancer Center at Thomas Jefferson University in Philadelphia, Pennsylvania.
“Nearly 9 in 10 patients reported some change after cancer treatment that negatively affected their sexual health,” he said.
Taylor was speaking at the American Society for Radiation Oncology (ASTRO) Annual Meeting, held virtually this year because of the pandemic.
“Negative effects on sexual health after cancer treatment are unfortunately very common,” he said. “This is not just patients treated with radiation but this includes chemotherapy, hormonal therapy, surgery, and other treatment modalities.”
Potential issues include physical complications such as erectile dysfunction with prostate cancer treatment or vaginal dryness with gynecological cancer treatment. One recent study found that one-third of men who had undergone treatment for prostate cancer reported that a subsequent lack of sexual function has had the greatest impact on their quality of life. Another study reported that nearly all patients with breast cancer taking endocrine therapy experience a high degree of sexual dysfunction, including vulvovaginal dryness and severe dyspareunia.
Not discussed, not warned
Taylor and colleagues developed a questionnaire with input from radiation oncologists, medical oncologists, and surgeons, which consisted of more than 25 questions and was specifically targeted at cancer survivors.
A total of 405 adults completed the electronic survey about their experiences with sexual side effects after cancer treatment (391 responses were eligible for analysis). Most of the respondents were women (81%), and the most common cancer types were breast (67%), prostate (16%), and endometrial (6%). Treatments included chemotherapy (78%), radiation therapy (54%), and hormone therapy (47%).
“The questionnaires were distributed at Thomas Jefferson and throughout social media,” said Taylor. “The responses from social media are important because it shows a broad representation of patients who are treated in multiple clinics across the United States.”
Most of the survivors who responded (n = 337, 87%) stated cancer treatment had impacted sexual function or desire, with 53.8% reporting body image distortion, 73.4% with dyspareunia, and 42.3% unable to achieve orgasm.
Only about one-quarter (27.9%) said they had been formally asked about their sexual health by their clinician.
“Only about 40% said that they have been preemptively warned that their sexual health may be affected by treatment,” said Taylor.
Women were far less likely to be asked about their sexual health by their provider. The survey showed that male respondents were twice as likely to say they had been asked about sexual health and counseled about the potential toxicity (53% vs 22%; P < .001), and a substantially higher percentage of men reported receiving a formal assessment tool such as a survey (32% vs 5%; P = .001) compared with female respondents.
Taylor noted that the survey demonstrated several things. “One is that sexual toxicity is exceedingly common, and number two, it identified a gender disparity,” he said. “But number 3, and I think that this is an important aspect of our study, is that the majority of respondents felt that they would like a standard questionnaire to initiate and guide a discussion on sexual health with their provider.”
The reason that aspect is very important, he emphasized, is that “we know metrics and questionnaires already exist, so this gives us an actionable intervention that we can distribute and help mitigate some of these disparities.”
Importance of being holistic
The results of the survey “highlight the importance of being holistic in our approach to patient survivorship,” commented Karen Winkfield, MD, PhD, associate professor of radiation oncology at Wake Forest University, Winston-Salem, North Carolina, and executive director of the Meharry-Vanderbilt Alliance, Nashville, Tennessee.
“We need to ask patients about all parts of their well-being, including sexual health,” Winkfield said. “Body dysmorphism can impact anyone, but especially patients who have had surgery or radiation,” she said, while chemotherapy can impact energy and libido and have other toxicities that impact sexual health.
“I encourage all oncologists to ask patients about their sexual health, and a standardized form that can be used across all sites will make this much easier,” Winkfield commented. “We owe it to our patients to treat them holistically.”
The authors have reported no relevant financial relationships.
This article first appeared on Medscape.com.
Sexual dysfunction is a common treatment-related problem observed across numerous cancer diagnoses, and a new survey finds that 87% of cancer survivors have had such problems.
However, most of them also reported that their oncologist had not formally discussed the topic, and female patients were particularly unlikely to be asked about sexual dysfunction.
“The main takeaway from our study is that sexual side effects following treatment are very common,” said lead author James Taylor, MD, MPH, chief resident in radiation oncology at the Sidney Kimmel Cancer Center at Thomas Jefferson University in Philadelphia, Pennsylvania.
“Nearly 9 in 10 patients reported some change after cancer treatment that negatively affected their sexual health,” he said.
Taylor was speaking at the American Society for Radiation Oncology (ASTRO) Annual Meeting, held virtually this year because of the pandemic.
“Negative effects on sexual health after cancer treatment are unfortunately very common,” he said. “This is not just patients treated with radiation but this includes chemotherapy, hormonal therapy, surgery, and other treatment modalities.”
Potential issues include physical complications such as erectile dysfunction with prostate cancer treatment or vaginal dryness with gynecological cancer treatment. One recent study found that one-third of men who had undergone treatment for prostate cancer reported that a subsequent lack of sexual function has had the greatest impact on their quality of life. Another study reported that nearly all patients with breast cancer taking endocrine therapy experience a high degree of sexual dysfunction, including vulvovaginal dryness and severe dyspareunia.
Not discussed, not warned
Taylor and colleagues developed a questionnaire with input from radiation oncologists, medical oncologists, and surgeons, which consisted of more than 25 questions and was specifically targeted at cancer survivors.
A total of 405 adults completed the electronic survey about their experiences with sexual side effects after cancer treatment (391 responses were eligible for analysis). Most of the respondents were women (81%), and the most common cancer types were breast (67%), prostate (16%), and endometrial (6%). Treatments included chemotherapy (78%), radiation therapy (54%), and hormone therapy (47%).
“The questionnaires were distributed at Thomas Jefferson and throughout social media,” said Taylor. “The responses from social media are important because it shows a broad representation of patients who are treated in multiple clinics across the United States.”
Most of the survivors who responded (n = 337, 87%) stated cancer treatment had impacted sexual function or desire, with 53.8% reporting body image distortion, 73.4% with dyspareunia, and 42.3% unable to achieve orgasm.
Only about one-quarter (27.9%) said they had been formally asked about their sexual health by their clinician.
“Only about 40% said that they have been preemptively warned that their sexual health may be affected by treatment,” said Taylor.
Women were far less likely to be asked about their sexual health by their provider. The survey showed that male respondents were twice as likely to say they had been asked about sexual health and counseled about the potential toxicity (53% vs 22%; P < .001), and a substantially higher percentage of men reported receiving a formal assessment tool such as a survey (32% vs 5%; P = .001) compared with female respondents.
Taylor noted that the survey demonstrated several things. “One is that sexual toxicity is exceedingly common, and number two, it identified a gender disparity,” he said. “But number 3, and I think that this is an important aspect of our study, is that the majority of respondents felt that they would like a standard questionnaire to initiate and guide a discussion on sexual health with their provider.”
The reason that aspect is very important, he emphasized, is that “we know metrics and questionnaires already exist, so this gives us an actionable intervention that we can distribute and help mitigate some of these disparities.”
Importance of being holistic
The results of the survey “highlight the importance of being holistic in our approach to patient survivorship,” commented Karen Winkfield, MD, PhD, associate professor of radiation oncology at Wake Forest University, Winston-Salem, North Carolina, and executive director of the Meharry-Vanderbilt Alliance, Nashville, Tennessee.
“We need to ask patients about all parts of their well-being, including sexual health,” Winkfield said. “Body dysmorphism can impact anyone, but especially patients who have had surgery or radiation,” she said, while chemotherapy can impact energy and libido and have other toxicities that impact sexual health.
“I encourage all oncologists to ask patients about their sexual health, and a standardized form that can be used across all sites will make this much easier,” Winkfield commented. “We owe it to our patients to treat them holistically.”
The authors have reported no relevant financial relationships.
This article first appeared on Medscape.com.
Understanding De-Implementation of Low Value Castration for Men With Prostate Cancer
RESEARCH OBJECTIVE: Men with prostate cancer are often treated with androgen deprivation therapy (ADT). While ADT monotherapy is not appropriate treatment for most localized prostate cancer, it continues to be used raising questions of low-value care. Guided by the Theoretical Domains Framework (TDF) and the Behavior Change Wheel’s Capability, Opportunity, Motivation Model (COM-B), we conducted a qualitative study to identify determinants of low value ADT use and opportunities for de-implementation strategy development.
STUDY DESIGN: We used VA national cancer registry and administrative data from 2016-2017 to select facilities with the highest and lowest rates of ADT monotherapy as localized prostate cancer treatment. We used purposive sampling to select high and low performing sites and complete and code 20 provider interviews from 14 facilities across the nation (17 high and 3 low ADT use sites). Next, we mapped TDF domains to the COM-B Model to generate a conceptual framework of provider approaches to low value ADT.
PRINCIPAL FINDINGS: Based on emerging behavioral themes, our conceptual model characterized 3 groups of providers based on low value ADT use: (1) never prescribe; (2) willing, under some circumstances, to prescribe; and (3) routinely prescribe as an acceptable treatment option. Providers in all groups demonstrated strengths in the Capability domain, such as knowledge of appropriate localized prostate cancer treatment options (knowledge), coupled with interpersonal skills to engage patients in educational discussion (skills). Motivation to prescribe low value ADT depended on goals of care, including patient preferences (goals), view of their role (beliefs in capabilities/professional role and identity), and beliefs about benefits and harms ADT would afford patients (beliefs about consequences). In the Opportunity domain, access to resources, such as guidelines and interdisciplinary colleagues (environmental resources) and advice of peers (social influences) were influential factors in providers’ decision- making about low value ADT prescribing.
CONCLUSIONS: Behavioral theory-based characterization of provider practices helps clarify determinants implicated in provider decisions to prescribe low value ADT.
IMPLICATIONS: Identifying behavioral determinants impacting provider decisions to prescribe low value ADT informs theory-based de-implementation strategy development, and serves as a model to decrease low-value care more broadly.
RESEARCH OBJECTIVE: Men with prostate cancer are often treated with androgen deprivation therapy (ADT). While ADT monotherapy is not appropriate treatment for most localized prostate cancer, it continues to be used raising questions of low-value care. Guided by the Theoretical Domains Framework (TDF) and the Behavior Change Wheel’s Capability, Opportunity, Motivation Model (COM-B), we conducted a qualitative study to identify determinants of low value ADT use and opportunities for de-implementation strategy development.
STUDY DESIGN: We used VA national cancer registry and administrative data from 2016-2017 to select facilities with the highest and lowest rates of ADT monotherapy as localized prostate cancer treatment. We used purposive sampling to select high and low performing sites and complete and code 20 provider interviews from 14 facilities across the nation (17 high and 3 low ADT use sites). Next, we mapped TDF domains to the COM-B Model to generate a conceptual framework of provider approaches to low value ADT.
PRINCIPAL FINDINGS: Based on emerging behavioral themes, our conceptual model characterized 3 groups of providers based on low value ADT use: (1) never prescribe; (2) willing, under some circumstances, to prescribe; and (3) routinely prescribe as an acceptable treatment option. Providers in all groups demonstrated strengths in the Capability domain, such as knowledge of appropriate localized prostate cancer treatment options (knowledge), coupled with interpersonal skills to engage patients in educational discussion (skills). Motivation to prescribe low value ADT depended on goals of care, including patient preferences (goals), view of their role (beliefs in capabilities/professional role and identity), and beliefs about benefits and harms ADT would afford patients (beliefs about consequences). In the Opportunity domain, access to resources, such as guidelines and interdisciplinary colleagues (environmental resources) and advice of peers (social influences) were influential factors in providers’ decision- making about low value ADT prescribing.
CONCLUSIONS: Behavioral theory-based characterization of provider practices helps clarify determinants implicated in provider decisions to prescribe low value ADT.
IMPLICATIONS: Identifying behavioral determinants impacting provider decisions to prescribe low value ADT informs theory-based de-implementation strategy development, and serves as a model to decrease low-value care more broadly.
RESEARCH OBJECTIVE: Men with prostate cancer are often treated with androgen deprivation therapy (ADT). While ADT monotherapy is not appropriate treatment for most localized prostate cancer, it continues to be used raising questions of low-value care. Guided by the Theoretical Domains Framework (TDF) and the Behavior Change Wheel’s Capability, Opportunity, Motivation Model (COM-B), we conducted a qualitative study to identify determinants of low value ADT use and opportunities for de-implementation strategy development.
STUDY DESIGN: We used VA national cancer registry and administrative data from 2016-2017 to select facilities with the highest and lowest rates of ADT monotherapy as localized prostate cancer treatment. We used purposive sampling to select high and low performing sites and complete and code 20 provider interviews from 14 facilities across the nation (17 high and 3 low ADT use sites). Next, we mapped TDF domains to the COM-B Model to generate a conceptual framework of provider approaches to low value ADT.
PRINCIPAL FINDINGS: Based on emerging behavioral themes, our conceptual model characterized 3 groups of providers based on low value ADT use: (1) never prescribe; (2) willing, under some circumstances, to prescribe; and (3) routinely prescribe as an acceptable treatment option. Providers in all groups demonstrated strengths in the Capability domain, such as knowledge of appropriate localized prostate cancer treatment options (knowledge), coupled with interpersonal skills to engage patients in educational discussion (skills). Motivation to prescribe low value ADT depended on goals of care, including patient preferences (goals), view of their role (beliefs in capabilities/professional role and identity), and beliefs about benefits and harms ADT would afford patients (beliefs about consequences). In the Opportunity domain, access to resources, such as guidelines and interdisciplinary colleagues (environmental resources) and advice of peers (social influences) were influential factors in providers’ decision- making about low value ADT prescribing.
CONCLUSIONS: Behavioral theory-based characterization of provider practices helps clarify determinants implicated in provider decisions to prescribe low value ADT.
IMPLICATIONS: Identifying behavioral determinants impacting provider decisions to prescribe low value ADT informs theory-based de-implementation strategy development, and serves as a model to decrease low-value care more broadly.
The Impact of Sequencing of Abiraterone and Enzalutamide in Veterans With Metastatic Castration- Resistant Prostate Cancer
PURPOSE: To evaluate outcomes of disease progression based on the sequence of abiraterone and enzalutamide in veterans diagnosed with metastatic castration-resistant prostate cancer (mCRPC). BACKGROUND: Two of the current options for mCRPC treatment are the novel oral hormonal agents abiraterone and enzalutamide. After progression on one of these agents, one option is to switch to the other agent not previously used. Previously published retrospective studies and one prospective study have shown a difference in outcomes favoring abiraterone followed by enzalutamide, while others have shown no difference based on sequence. The optimal sequence of abiraterone and enzalutamide is still unclear.
METHODS: This was a retrospective chart review of patients who received abiraterone and enzalutamide in sequence for the treatment of mCRPC within our healthcare system from April 28, 2011 through October 31, 2019. Baseline demographic information such as age, race, Gleason score, and prior treatments were collected. The primary outcome was combined prostate-specific antigen progression-free survival (cPSA-PFS). Secondary outcomes included radiographic PFS (rPFS), overall survival (OS), adverse events causing treatment discontinuation, and medication adherence. Between-group survival differences were estimated by the Kaplan-Meier method and an unadjusted Cox regression model.
RESULTS: A total of 77 patients met criteria for study inclusion, with 51 in the abiraterone-to-enzalutamide group (ABI-ENZ) and 26 in the enzalutamide-to-abiraterone group (ENZ-ABI). For the primary outcome of cPSA-PFS, the median survival of the ABI-ENZ and ENZ-ABI groups was 17.3 months (95% CI, 10.3-24.3 months) and 10.2 months (95% CI, 8.5-11.8 months), respectively, which was significantly different (log-rank P=0.009) in favor of the ABI-ENZ sequence (HR 0.46; 95% CI, 0.26-0.83). Secondary outcomes of rPFS and OS were not significantly different between groups.
CONCLUSION: This study adds to the evidence supporting the sequence of abiraterone before enzalutamide for improving PSA-PFS. It is thought this might be related to differences in mechanisms of resistance between the two drugs. This benefit has not yet translated to an improvement in rPFS and OS. Based on the results of this study in conjunction with previously published studies, use of abiraterone before enzalutamide should be considered over the alternate sequence.
PURPOSE: To evaluate outcomes of disease progression based on the sequence of abiraterone and enzalutamide in veterans diagnosed with metastatic castration-resistant prostate cancer (mCRPC). BACKGROUND: Two of the current options for mCRPC treatment are the novel oral hormonal agents abiraterone and enzalutamide. After progression on one of these agents, one option is to switch to the other agent not previously used. Previously published retrospective studies and one prospective study have shown a difference in outcomes favoring abiraterone followed by enzalutamide, while others have shown no difference based on sequence. The optimal sequence of abiraterone and enzalutamide is still unclear.
METHODS: This was a retrospective chart review of patients who received abiraterone and enzalutamide in sequence for the treatment of mCRPC within our healthcare system from April 28, 2011 through October 31, 2019. Baseline demographic information such as age, race, Gleason score, and prior treatments were collected. The primary outcome was combined prostate-specific antigen progression-free survival (cPSA-PFS). Secondary outcomes included radiographic PFS (rPFS), overall survival (OS), adverse events causing treatment discontinuation, and medication adherence. Between-group survival differences were estimated by the Kaplan-Meier method and an unadjusted Cox regression model.
RESULTS: A total of 77 patients met criteria for study inclusion, with 51 in the abiraterone-to-enzalutamide group (ABI-ENZ) and 26 in the enzalutamide-to-abiraterone group (ENZ-ABI). For the primary outcome of cPSA-PFS, the median survival of the ABI-ENZ and ENZ-ABI groups was 17.3 months (95% CI, 10.3-24.3 months) and 10.2 months (95% CI, 8.5-11.8 months), respectively, which was significantly different (log-rank P=0.009) in favor of the ABI-ENZ sequence (HR 0.46; 95% CI, 0.26-0.83). Secondary outcomes of rPFS and OS were not significantly different between groups.
CONCLUSION: This study adds to the evidence supporting the sequence of abiraterone before enzalutamide for improving PSA-PFS. It is thought this might be related to differences in mechanisms of resistance between the two drugs. This benefit has not yet translated to an improvement in rPFS and OS. Based on the results of this study in conjunction with previously published studies, use of abiraterone before enzalutamide should be considered over the alternate sequence.
PURPOSE: To evaluate outcomes of disease progression based on the sequence of abiraterone and enzalutamide in veterans diagnosed with metastatic castration-resistant prostate cancer (mCRPC). BACKGROUND: Two of the current options for mCRPC treatment are the novel oral hormonal agents abiraterone and enzalutamide. After progression on one of these agents, one option is to switch to the other agent not previously used. Previously published retrospective studies and one prospective study have shown a difference in outcomes favoring abiraterone followed by enzalutamide, while others have shown no difference based on sequence. The optimal sequence of abiraterone and enzalutamide is still unclear.
METHODS: This was a retrospective chart review of patients who received abiraterone and enzalutamide in sequence for the treatment of mCRPC within our healthcare system from April 28, 2011 through October 31, 2019. Baseline demographic information such as age, race, Gleason score, and prior treatments were collected. The primary outcome was combined prostate-specific antigen progression-free survival (cPSA-PFS). Secondary outcomes included radiographic PFS (rPFS), overall survival (OS), adverse events causing treatment discontinuation, and medication adherence. Between-group survival differences were estimated by the Kaplan-Meier method and an unadjusted Cox regression model.
RESULTS: A total of 77 patients met criteria for study inclusion, with 51 in the abiraterone-to-enzalutamide group (ABI-ENZ) and 26 in the enzalutamide-to-abiraterone group (ENZ-ABI). For the primary outcome of cPSA-PFS, the median survival of the ABI-ENZ and ENZ-ABI groups was 17.3 months (95% CI, 10.3-24.3 months) and 10.2 months (95% CI, 8.5-11.8 months), respectively, which was significantly different (log-rank P=0.009) in favor of the ABI-ENZ sequence (HR 0.46; 95% CI, 0.26-0.83). Secondary outcomes of rPFS and OS were not significantly different between groups.
CONCLUSION: This study adds to the evidence supporting the sequence of abiraterone before enzalutamide for improving PSA-PFS. It is thought this might be related to differences in mechanisms of resistance between the two drugs. This benefit has not yet translated to an improvement in rPFS and OS. Based on the results of this study in conjunction with previously published studies, use of abiraterone before enzalutamide should be considered over the alternate sequence.
The Impact of Sequencing of Abiraterone and Enzalutamide in Veterans With Metastatic Castration- Resistant Prostate Cancer
PURPOSE: To evaluate outcomes of disease progression based on the sequence of abiraterone and enzalutamide in veterans diagnosed with metastatic castration-resistant prostate cancer (mCRPC).
BACKGROUND: Two of the current options for mCRPC treatment are the novel oral hormonal agents abiraterone and enzalutamide. After progression on one of these agents, one option is to switch to the other agent not previously used. Previously published retrospective studies and one prospective study have shown a difference in outcomes favoring abiraterone followed by enzalutamide, while others have shown no difference based on sequence. The optimal sequence of abiraterone and enzalutamide is still unclear.
METHODS: This was a retrospective chart review of patients who received abiraterone and enzalutamide in sequence for the treatment of mCRPC within our healthcare system from April 28, 2011 through October 31, 2019. Baseline demographic information such as age, race, Gleason score, and prior treatments were collected. The primary outcome was combined prostate-specific antigen progression-free survival (cPSA-PFS). Secondary outcomes included radiographic PFS (rPFS), overall survival (OS), adverse events causing treatment discontinuation, and medication adherence. Between-group survival differences were estimated by the Kaplan-Meier method and an unadjusted Cox regression model.
RESULTS: A total of 77 patients met criteria for study inclusion, with 51 in the abiraterone-to-enzalutamide group (ABI-ENZ) and 26 in the enzalutamide-to-abiraterone group (ENZ-ABI). For the primary outcome of cPSA-PFS, the median survival of the ABI-ENZ and ENZ-ABI groups was 17.3 months (95% CI, 10.3-24.3 months) and 10.2 months (95% CI, 8.5-11.8 months), respectively, which was significantly different (log-rank P=0.009) in favor of the ABI-ENZ sequence (HR 0.46; 95% CI, 0.26-0.83). Secondary outcomes of rPFS and OS were not significantly different between groups.
CONCLUSION: This study adds to the evidence supporting the sequence of abiraterone before enzalutamide for improving PSA-PFS. It is thought this might be related to differences in mechanisms of resistance between the two drugs. This benefit has not yet translated to an improvement in rPFS and OS. Based on the results of this study in conjunction with previously published studies, use of abiraterone before enzalutamide should be considered over the alternate sequence.
PURPOSE: To evaluate outcomes of disease progression based on the sequence of abiraterone and enzalutamide in veterans diagnosed with metastatic castration-resistant prostate cancer (mCRPC).
BACKGROUND: Two of the current options for mCRPC treatment are the novel oral hormonal agents abiraterone and enzalutamide. After progression on one of these agents, one option is to switch to the other agent not previously used. Previously published retrospective studies and one prospective study have shown a difference in outcomes favoring abiraterone followed by enzalutamide, while others have shown no difference based on sequence. The optimal sequence of abiraterone and enzalutamide is still unclear.
METHODS: This was a retrospective chart review of patients who received abiraterone and enzalutamide in sequence for the treatment of mCRPC within our healthcare system from April 28, 2011 through October 31, 2019. Baseline demographic information such as age, race, Gleason score, and prior treatments were collected. The primary outcome was combined prostate-specific antigen progression-free survival (cPSA-PFS). Secondary outcomes included radiographic PFS (rPFS), overall survival (OS), adverse events causing treatment discontinuation, and medication adherence. Between-group survival differences were estimated by the Kaplan-Meier method and an unadjusted Cox regression model.
RESULTS: A total of 77 patients met criteria for study inclusion, with 51 in the abiraterone-to-enzalutamide group (ABI-ENZ) and 26 in the enzalutamide-to-abiraterone group (ENZ-ABI). For the primary outcome of cPSA-PFS, the median survival of the ABI-ENZ and ENZ-ABI groups was 17.3 months (95% CI, 10.3-24.3 months) and 10.2 months (95% CI, 8.5-11.8 months), respectively, which was significantly different (log-rank P=0.009) in favor of the ABI-ENZ sequence (HR 0.46; 95% CI, 0.26-0.83). Secondary outcomes of rPFS and OS were not significantly different between groups.
CONCLUSION: This study adds to the evidence supporting the sequence of abiraterone before enzalutamide for improving PSA-PFS. It is thought this might be related to differences in mechanisms of resistance between the two drugs. This benefit has not yet translated to an improvement in rPFS and OS. Based on the results of this study in conjunction with previously published studies, use of abiraterone before enzalutamide should be considered over the alternate sequence.
PURPOSE: To evaluate outcomes of disease progression based on the sequence of abiraterone and enzalutamide in veterans diagnosed with metastatic castration-resistant prostate cancer (mCRPC).
BACKGROUND: Two of the current options for mCRPC treatment are the novel oral hormonal agents abiraterone and enzalutamide. After progression on one of these agents, one option is to switch to the other agent not previously used. Previously published retrospective studies and one prospective study have shown a difference in outcomes favoring abiraterone followed by enzalutamide, while others have shown no difference based on sequence. The optimal sequence of abiraterone and enzalutamide is still unclear.
METHODS: This was a retrospective chart review of patients who received abiraterone and enzalutamide in sequence for the treatment of mCRPC within our healthcare system from April 28, 2011 through October 31, 2019. Baseline demographic information such as age, race, Gleason score, and prior treatments were collected. The primary outcome was combined prostate-specific antigen progression-free survival (cPSA-PFS). Secondary outcomes included radiographic PFS (rPFS), overall survival (OS), adverse events causing treatment discontinuation, and medication adherence. Between-group survival differences were estimated by the Kaplan-Meier method and an unadjusted Cox regression model.
RESULTS: A total of 77 patients met criteria for study inclusion, with 51 in the abiraterone-to-enzalutamide group (ABI-ENZ) and 26 in the enzalutamide-to-abiraterone group (ENZ-ABI). For the primary outcome of cPSA-PFS, the median survival of the ABI-ENZ and ENZ-ABI groups was 17.3 months (95% CI, 10.3-24.3 months) and 10.2 months (95% CI, 8.5-11.8 months), respectively, which was significantly different (log-rank P=0.009) in favor of the ABI-ENZ sequence (HR 0.46; 95% CI, 0.26-0.83). Secondary outcomes of rPFS and OS were not significantly different between groups.
CONCLUSION: This study adds to the evidence supporting the sequence of abiraterone before enzalutamide for improving PSA-PFS. It is thought this might be related to differences in mechanisms of resistance between the two drugs. This benefit has not yet translated to an improvement in rPFS and OS. Based on the results of this study in conjunction with previously published studies, use of abiraterone before enzalutamide should be considered over the alternate sequence.
Prostate-Specific Antigen Decline >50% After 4 Weeks of Treatment Is an Indicator of Better Progression Free and Overall Survival Both With Abiraterone and Enzalutamide in Metastatic Castrate Resistant Prostate Cancer
BACKGROUND: Abiraterone (ABI) and Enzalutamide (ENZA) are the most common medications used for metastatic castrate resistant prostate cancer (mCRPC). Post treatment prostate specific antigen (PSA) changes correlate with prognosis (1). Early PSA decline defined as PSA decline by >50 % at 4 weeks after starting treatment compared to baseline has been demonstrated to corresponds with improved PSA progression-free survival (PSA PFS) and overall survival (OS) (2). Based on our literature review, this relation has not been elucidated previously in the Veteran population.
METHODS: A retrospective chart review was conducted in subjects with mCRPC who had received ABI and ENZA from 2011 to 2019 at VA Fresno. Subjects must have received at least 3 months of each drug. The primary outcomes were to document PSAPFS and OS between patients with >50% and <50% decline in PSA at 4 weeks after starting treatment with either ABI or ENZA.
RESULTS: 50 patients were identified who were treated with Abiraterone. 30 (60%) patients had a PSA decrease of 50% or greater in the first 4 weeks. A 50% or greater decrease in PSA at 4 weeks after starting was associated with a better PSA PFS (15 vs 5 months) and better median OS (20.5 vs 12 months). 20 patients were identified treated with Enzalutamide. 13(65%) of patients had a PSA decrease of 50% or greater in the first 4 weeks. A 50% or greater decrease in PSA at 4 weeks was associated with a better PSA PFS (14.5 vs 5 months) and better median OS (20 vs 12 months). 50% of patients in the whole group were diagnosed with metastatic disease at initial diagnosis while the rest were previously treated for localized prostate cancer before being diagnosed as metastatic disease. Most baseline characteristics including, baseline PSA, the modality of initial localized treatment, Gleason score, previous treatment with chemotherapy were similar between the two comparison cohorts (>50% and <50% decrease in PSA after starting treatment with ABI or ENZA).
CONCLUSIONS: Early decrease in PSA is an independent marker for the efficacy of antiandrogen treatment with enzalutamide and abiraterone in metastatic hormone- resistant prostate cancer in the veteran population.
BACKGROUND: Abiraterone (ABI) and Enzalutamide (ENZA) are the most common medications used for metastatic castrate resistant prostate cancer (mCRPC). Post treatment prostate specific antigen (PSA) changes correlate with prognosis (1). Early PSA decline defined as PSA decline by >50 % at 4 weeks after starting treatment compared to baseline has been demonstrated to corresponds with improved PSA progression-free survival (PSA PFS) and overall survival (OS) (2). Based on our literature review, this relation has not been elucidated previously in the Veteran population.
METHODS: A retrospective chart review was conducted in subjects with mCRPC who had received ABI and ENZA from 2011 to 2019 at VA Fresno. Subjects must have received at least 3 months of each drug. The primary outcomes were to document PSAPFS and OS between patients with >50% and <50% decline in PSA at 4 weeks after starting treatment with either ABI or ENZA.
RESULTS: 50 patients were identified who were treated with Abiraterone. 30 (60%) patients had a PSA decrease of 50% or greater in the first 4 weeks. A 50% or greater decrease in PSA at 4 weeks after starting was associated with a better PSA PFS (15 vs 5 months) and better median OS (20.5 vs 12 months). 20 patients were identified treated with Enzalutamide. 13(65%) of patients had a PSA decrease of 50% or greater in the first 4 weeks. A 50% or greater decrease in PSA at 4 weeks was associated with a better PSA PFS (14.5 vs 5 months) and better median OS (20 vs 12 months). 50% of patients in the whole group were diagnosed with metastatic disease at initial diagnosis while the rest were previously treated for localized prostate cancer before being diagnosed as metastatic disease. Most baseline characteristics including, baseline PSA, the modality of initial localized treatment, Gleason score, previous treatment with chemotherapy were similar between the two comparison cohorts (>50% and <50% decrease in PSA after starting treatment with ABI or ENZA).
CONCLUSIONS: Early decrease in PSA is an independent marker for the efficacy of antiandrogen treatment with enzalutamide and abiraterone in metastatic hormone- resistant prostate cancer in the veteran population.
BACKGROUND: Abiraterone (ABI) and Enzalutamide (ENZA) are the most common medications used for metastatic castrate resistant prostate cancer (mCRPC). Post treatment prostate specific antigen (PSA) changes correlate with prognosis (1). Early PSA decline defined as PSA decline by >50 % at 4 weeks after starting treatment compared to baseline has been demonstrated to corresponds with improved PSA progression-free survival (PSA PFS) and overall survival (OS) (2). Based on our literature review, this relation has not been elucidated previously in the Veteran population.
METHODS: A retrospective chart review was conducted in subjects with mCRPC who had received ABI and ENZA from 2011 to 2019 at VA Fresno. Subjects must have received at least 3 months of each drug. The primary outcomes were to document PSAPFS and OS between patients with >50% and <50% decline in PSA at 4 weeks after starting treatment with either ABI or ENZA.
RESULTS: 50 patients were identified who were treated with Abiraterone. 30 (60%) patients had a PSA decrease of 50% or greater in the first 4 weeks. A 50% or greater decrease in PSA at 4 weeks after starting was associated with a better PSA PFS (15 vs 5 months) and better median OS (20.5 vs 12 months). 20 patients were identified treated with Enzalutamide. 13(65%) of patients had a PSA decrease of 50% or greater in the first 4 weeks. A 50% or greater decrease in PSA at 4 weeks was associated with a better PSA PFS (14.5 vs 5 months) and better median OS (20 vs 12 months). 50% of patients in the whole group were diagnosed with metastatic disease at initial diagnosis while the rest were previously treated for localized prostate cancer before being diagnosed as metastatic disease. Most baseline characteristics including, baseline PSA, the modality of initial localized treatment, Gleason score, previous treatment with chemotherapy were similar between the two comparison cohorts (>50% and <50% decrease in PSA after starting treatment with ABI or ENZA).
CONCLUSIONS: Early decrease in PSA is an independent marker for the efficacy of antiandrogen treatment with enzalutamide and abiraterone in metastatic hormone- resistant prostate cancer in the veteran population.
Prostate Cancer in a Transgender Woman
INTRODUCTION: As a neoplasm specific to male reproductive anatomy, prostate cancer represents a complex disease experience for transgender women. We present a patient who initiated her male-to-female (MTF) transition amidst a new prostate cancer diagnosis.
CASE REPORT: A 58-year-old male presented with urinary retention and a PSA of 21.53 ng/ml. Prostate biopsy showed a Gleason 4+5 adenocarcinoma with no evidence of metastasis on imaging. The patient underwent radical prostatectomy, which showed seminal vesicle invasion. After surgery, the PSA nadired to 2.5 ng/ml. Salvage radiation and androgen deprivation therapy (ADT) with leuprolide was initiated. ADT-induced physical feminization prompted the patient to begin discussing lifelong gender dysphoria in group therapy. After completing radiation therapy, she opted for bilateral orchiectomy. Shortly thereafter, she started an estradiol patch. Five months later, the PSA level began to rise. Estradiol-induced carcinogenesis due to tumor estrogen receptor (ER) expression versus natural tumor progression was considered, so she underwent a 6-week trial without estradiol. PSA levels continued increasing, and the tumor was found to be ER-negative. Furthermore, the patient suffered psychological and physical side effects from pausing hormonal therapy, so estradiol was restarted. Abiraterone and prednisone were initiated with PSA response (<0.01 ng/ml).
DISCUSSION: To date, there have been ten reported cases of prostate cancer in MTF transgender patients. Prostate cancer management in this population carries unique considerations, such as the possible carcinogenic effect of estrogen. Estradiol, a commonly used hormone in trans female patients, is a ligand for ER-alpha and ER-beta, both of which are implicated in oncogenesis and progression of prostate cancer. ER-alpha expression is associated with higher Gleason score, more advanced disease, and shorter survival. Orchiectomy as an alternative to long-term ADT should be discussed. These patients may also face significant psycho-social challenges such as exacerbation of gender dysphoria by prostate cancer, difficulty engaging with health care providers, and issues with changing gender status in electronic health record. Mental health support, sensitivity to transgender pronouns, and goals-of-care discussions are critical. As gender fluidity becomes more prevalent, physicians need to tailor biopsychosocial cancer care for transgender patients.
INTRODUCTION: As a neoplasm specific to male reproductive anatomy, prostate cancer represents a complex disease experience for transgender women. We present a patient who initiated her male-to-female (MTF) transition amidst a new prostate cancer diagnosis.
CASE REPORT: A 58-year-old male presented with urinary retention and a PSA of 21.53 ng/ml. Prostate biopsy showed a Gleason 4+5 adenocarcinoma with no evidence of metastasis on imaging. The patient underwent radical prostatectomy, which showed seminal vesicle invasion. After surgery, the PSA nadired to 2.5 ng/ml. Salvage radiation and androgen deprivation therapy (ADT) with leuprolide was initiated. ADT-induced physical feminization prompted the patient to begin discussing lifelong gender dysphoria in group therapy. After completing radiation therapy, she opted for bilateral orchiectomy. Shortly thereafter, she started an estradiol patch. Five months later, the PSA level began to rise. Estradiol-induced carcinogenesis due to tumor estrogen receptor (ER) expression versus natural tumor progression was considered, so she underwent a 6-week trial without estradiol. PSA levels continued increasing, and the tumor was found to be ER-negative. Furthermore, the patient suffered psychological and physical side effects from pausing hormonal therapy, so estradiol was restarted. Abiraterone and prednisone were initiated with PSA response (<0.01 ng/ml).
DISCUSSION: To date, there have been ten reported cases of prostate cancer in MTF transgender patients. Prostate cancer management in this population carries unique considerations, such as the possible carcinogenic effect of estrogen. Estradiol, a commonly used hormone in trans female patients, is a ligand for ER-alpha and ER-beta, both of which are implicated in oncogenesis and progression of prostate cancer. ER-alpha expression is associated with higher Gleason score, more advanced disease, and shorter survival. Orchiectomy as an alternative to long-term ADT should be discussed. These patients may also face significant psycho-social challenges such as exacerbation of gender dysphoria by prostate cancer, difficulty engaging with health care providers, and issues with changing gender status in electronic health record. Mental health support, sensitivity to transgender pronouns, and goals-of-care discussions are critical. As gender fluidity becomes more prevalent, physicians need to tailor biopsychosocial cancer care for transgender patients.
INTRODUCTION: As a neoplasm specific to male reproductive anatomy, prostate cancer represents a complex disease experience for transgender women. We present a patient who initiated her male-to-female (MTF) transition amidst a new prostate cancer diagnosis.
CASE REPORT: A 58-year-old male presented with urinary retention and a PSA of 21.53 ng/ml. Prostate biopsy showed a Gleason 4+5 adenocarcinoma with no evidence of metastasis on imaging. The patient underwent radical prostatectomy, which showed seminal vesicle invasion. After surgery, the PSA nadired to 2.5 ng/ml. Salvage radiation and androgen deprivation therapy (ADT) with leuprolide was initiated. ADT-induced physical feminization prompted the patient to begin discussing lifelong gender dysphoria in group therapy. After completing radiation therapy, she opted for bilateral orchiectomy. Shortly thereafter, she started an estradiol patch. Five months later, the PSA level began to rise. Estradiol-induced carcinogenesis due to tumor estrogen receptor (ER) expression versus natural tumor progression was considered, so she underwent a 6-week trial without estradiol. PSA levels continued increasing, and the tumor was found to be ER-negative. Furthermore, the patient suffered psychological and physical side effects from pausing hormonal therapy, so estradiol was restarted. Abiraterone and prednisone were initiated with PSA response (<0.01 ng/ml).
DISCUSSION: To date, there have been ten reported cases of prostate cancer in MTF transgender patients. Prostate cancer management in this population carries unique considerations, such as the possible carcinogenic effect of estrogen. Estradiol, a commonly used hormone in trans female patients, is a ligand for ER-alpha and ER-beta, both of which are implicated in oncogenesis and progression of prostate cancer. ER-alpha expression is associated with higher Gleason score, more advanced disease, and shorter survival. Orchiectomy as an alternative to long-term ADT should be discussed. These patients may also face significant psycho-social challenges such as exacerbation of gender dysphoria by prostate cancer, difficulty engaging with health care providers, and issues with changing gender status in electronic health record. Mental health support, sensitivity to transgender pronouns, and goals-of-care discussions are critical. As gender fluidity becomes more prevalent, physicians need to tailor biopsychosocial cancer care for transgender patients.
Evidence-Based Project and Quality Initiative Towards Improving Decision Making and Outcomes in Prostate Cancer Bone Health at Salt Lake City VA
PURPOSE/BACKGROUND: Long term androgen deprivation therapy (ADT) forms the backbone of treatment of locally advanced and metastatic prostate cancer. Bone modifying agents, such as bisphosphonates and denosumab, may be indicated in osteoporosis dosing in the castration-sensitive setting, and more intense dosing for bone metastases in the castration-resistant setting only. Dental evaluation and care prior to bone modifying agent use in osteoporosis or bone metastases has safety benefit. Historical lack of clinical practice guidelines for bone health in men with prostate cancer have limited evidence-based practice. A retrospective review of patients on active bone remodeling therapies for prostate cancer, Revealed that several patients with castration-sensitive disease received treatment at dosing supported only in the setting of castration resistance with bone metastases. Some patients had not completed dental evaluation prior to initiation of bone modifying agents.
METHODS: Following evidence-based expert consensus recommendations from multiple sources regarding bone health in prostate cancer, we created an algorithm- based clinical practice tool. This decision tool is activated within the electronic medical record order set when starting therapy with a bone remodeling agent in patients with prostate cancer. The tool supports treatment with appropriate dosing for the indication, and ensures pretreatment supportive care, such as dental evaluation, is performed.
DATA ANALYSIS/RESULTS: Since implementation of the algorithm-based decision tool, 0/10 (0%) patients were placed on inappropriate bone modifying agent dosing and dental care was addressed on every patient 10/10 (100%) initiating treatment. When evaluating the effect of the decision tool on the desired outcomes, we note that the fraction of patients getting overly intensive treatment before and after implementation of the tool was 24/41 vs 0/10 (p = 0.0008); lack of pretreatment dental assessment before and after implementation of the tool was noted to be 12/41 vs 0/10): ( =0.09). Fisher’s Exact Test was used for both comparisons.
IMPLICATIONS: Through implementation of an evidence- based algorithm and clinical practice tool while prescribing bone remodeling agents to patients with prostate cancer, we were able to improve our institutional practice to a high quality evidenced-based approach to prostate cancer bone health care.
PURPOSE/BACKGROUND: Long term androgen deprivation therapy (ADT) forms the backbone of treatment of locally advanced and metastatic prostate cancer. Bone modifying agents, such as bisphosphonates and denosumab, may be indicated in osteoporosis dosing in the castration-sensitive setting, and more intense dosing for bone metastases in the castration-resistant setting only. Dental evaluation and care prior to bone modifying agent use in osteoporosis or bone metastases has safety benefit. Historical lack of clinical practice guidelines for bone health in men with prostate cancer have limited evidence-based practice. A retrospective review of patients on active bone remodeling therapies for prostate cancer, Revealed that several patients with castration-sensitive disease received treatment at dosing supported only in the setting of castration resistance with bone metastases. Some patients had not completed dental evaluation prior to initiation of bone modifying agents.
METHODS: Following evidence-based expert consensus recommendations from multiple sources regarding bone health in prostate cancer, we created an algorithm- based clinical practice tool. This decision tool is activated within the electronic medical record order set when starting therapy with a bone remodeling agent in patients with prostate cancer. The tool supports treatment with appropriate dosing for the indication, and ensures pretreatment supportive care, such as dental evaluation, is performed.
DATA ANALYSIS/RESULTS: Since implementation of the algorithm-based decision tool, 0/10 (0%) patients were placed on inappropriate bone modifying agent dosing and dental care was addressed on every patient 10/10 (100%) initiating treatment. When evaluating the effect of the decision tool on the desired outcomes, we note that the fraction of patients getting overly intensive treatment before and after implementation of the tool was 24/41 vs 0/10 (p = 0.0008); lack of pretreatment dental assessment before and after implementation of the tool was noted to be 12/41 vs 0/10): ( =0.09). Fisher’s Exact Test was used for both comparisons.
IMPLICATIONS: Through implementation of an evidence- based algorithm and clinical practice tool while prescribing bone remodeling agents to patients with prostate cancer, we were able to improve our institutional practice to a high quality evidenced-based approach to prostate cancer bone health care.
PURPOSE/BACKGROUND: Long term androgen deprivation therapy (ADT) forms the backbone of treatment of locally advanced and metastatic prostate cancer. Bone modifying agents, such as bisphosphonates and denosumab, may be indicated in osteoporosis dosing in the castration-sensitive setting, and more intense dosing for bone metastases in the castration-resistant setting only. Dental evaluation and care prior to bone modifying agent use in osteoporosis or bone metastases has safety benefit. Historical lack of clinical practice guidelines for bone health in men with prostate cancer have limited evidence-based practice. A retrospective review of patients on active bone remodeling therapies for prostate cancer, Revealed that several patients with castration-sensitive disease received treatment at dosing supported only in the setting of castration resistance with bone metastases. Some patients had not completed dental evaluation prior to initiation of bone modifying agents.
METHODS: Following evidence-based expert consensus recommendations from multiple sources regarding bone health in prostate cancer, we created an algorithm- based clinical practice tool. This decision tool is activated within the electronic medical record order set when starting therapy with a bone remodeling agent in patients with prostate cancer. The tool supports treatment with appropriate dosing for the indication, and ensures pretreatment supportive care, such as dental evaluation, is performed.
DATA ANALYSIS/RESULTS: Since implementation of the algorithm-based decision tool, 0/10 (0%) patients were placed on inappropriate bone modifying agent dosing and dental care was addressed on every patient 10/10 (100%) initiating treatment. When evaluating the effect of the decision tool on the desired outcomes, we note that the fraction of patients getting overly intensive treatment before and after implementation of the tool was 24/41 vs 0/10 (p = 0.0008); lack of pretreatment dental assessment before and after implementation of the tool was noted to be 12/41 vs 0/10): ( =0.09). Fisher’s Exact Test was used for both comparisons.
IMPLICATIONS: Through implementation of an evidence- based algorithm and clinical practice tool while prescribing bone remodeling agents to patients with prostate cancer, we were able to improve our institutional practice to a high quality evidenced-based approach to prostate cancer bone health care.
DNA Repair Gene Variants in Patients With Prostate Cancer Achieving Durable Clinical Benefit With PARP Inhibitors
BACKGROUND: PARP inhibitors (PARPi’s) were recently approved for the treatment of metastatic prostate cancer among patients harboring mutations in an array of genes responsible for DNA repair. We sought to identify whether a subset of these genes correlates with response to treatment more frequently than others. Consequently, an evaluation of the specific DNA repair genotypes associated with durable clinical benefit (DCB) using real-world patient data was undertaken.
METHODS: The U.S. Department of Veterans Affairs (VA) National Precision Oncology Program’s (NPOP) database and Corporate Data Warehouse (CDW) were reviewed to select patients who (1) carried a diagnosis of prostate cancer, (2) successfully underwent tumor DNA sequencing through NPOP, (3) were prescribed olaparib, rucaparib, nirapib, and/or talazaporib for their prostate cancer between July 2016 and February 2020, and (4) and achieved DCB, defined as no progression in prostate-specific antigen (PSA) for at least 6 months following PARPi initiation without concurrent systemic or non-systemic therapies other than androgen-deprivation. The DNA repair gene variants and orders placed for NPOP consultative support were reviewed.
RESULTS: Of the 44 prostate cancer patients treated with a PARPi, 6 (13.6%) had tumor DNA sequencing through NPOP and had achieved DCB. Five patients were treated with olaparib and 1 with rucaparib. The median PSA progression-free survival was 8.9 (interquartile range = 8.5 – 11.2) months among these selected patients. Regarding gene variants, 5 patients had 7 BRCA2 mutations, including 4 frameshift, 1 nonsense, 1 single nucleotide variant, and 1 splice site. One patient had frameshift and missense ATM mutations. Referrals to the NPOP consult service were ordered for 2 out of the 5 patients with BRCA2 mutations achieving DCB.
CONCLUSIONS: Within the VA’s NPOP, the presence of BRCA2 gene variants was the most common finding from tumor DNA sequencing among patients with prostate cancer achieving DCB with a PARPi. Further analysis of the genotypes of all patients treated with PARPi in NPOP to assess the differential impact of BRCA2 mutations is needed to confirm the clinical implication of this finding.
BACKGROUND: PARP inhibitors (PARPi’s) were recently approved for the treatment of metastatic prostate cancer among patients harboring mutations in an array of genes responsible for DNA repair. We sought to identify whether a subset of these genes correlates with response to treatment more frequently than others. Consequently, an evaluation of the specific DNA repair genotypes associated with durable clinical benefit (DCB) using real-world patient data was undertaken.
METHODS: The U.S. Department of Veterans Affairs (VA) National Precision Oncology Program’s (NPOP) database and Corporate Data Warehouse (CDW) were reviewed to select patients who (1) carried a diagnosis of prostate cancer, (2) successfully underwent tumor DNA sequencing through NPOP, (3) were prescribed olaparib, rucaparib, nirapib, and/or talazaporib for their prostate cancer between July 2016 and February 2020, and (4) and achieved DCB, defined as no progression in prostate-specific antigen (PSA) for at least 6 months following PARPi initiation without concurrent systemic or non-systemic therapies other than androgen-deprivation. The DNA repair gene variants and orders placed for NPOP consultative support were reviewed.
RESULTS: Of the 44 prostate cancer patients treated with a PARPi, 6 (13.6%) had tumor DNA sequencing through NPOP and had achieved DCB. Five patients were treated with olaparib and 1 with rucaparib. The median PSA progression-free survival was 8.9 (interquartile range = 8.5 – 11.2) months among these selected patients. Regarding gene variants, 5 patients had 7 BRCA2 mutations, including 4 frameshift, 1 nonsense, 1 single nucleotide variant, and 1 splice site. One patient had frameshift and missense ATM mutations. Referrals to the NPOP consult service were ordered for 2 out of the 5 patients with BRCA2 mutations achieving DCB.
CONCLUSIONS: Within the VA’s NPOP, the presence of BRCA2 gene variants was the most common finding from tumor DNA sequencing among patients with prostate cancer achieving DCB with a PARPi. Further analysis of the genotypes of all patients treated with PARPi in NPOP to assess the differential impact of BRCA2 mutations is needed to confirm the clinical implication of this finding.
BACKGROUND: PARP inhibitors (PARPi’s) were recently approved for the treatment of metastatic prostate cancer among patients harboring mutations in an array of genes responsible for DNA repair. We sought to identify whether a subset of these genes correlates with response to treatment more frequently than others. Consequently, an evaluation of the specific DNA repair genotypes associated with durable clinical benefit (DCB) using real-world patient data was undertaken.
METHODS: The U.S. Department of Veterans Affairs (VA) National Precision Oncology Program’s (NPOP) database and Corporate Data Warehouse (CDW) were reviewed to select patients who (1) carried a diagnosis of prostate cancer, (2) successfully underwent tumor DNA sequencing through NPOP, (3) were prescribed olaparib, rucaparib, nirapib, and/or talazaporib for their prostate cancer between July 2016 and February 2020, and (4) and achieved DCB, defined as no progression in prostate-specific antigen (PSA) for at least 6 months following PARPi initiation without concurrent systemic or non-systemic therapies other than androgen-deprivation. The DNA repair gene variants and orders placed for NPOP consultative support were reviewed.
RESULTS: Of the 44 prostate cancer patients treated with a PARPi, 6 (13.6%) had tumor DNA sequencing through NPOP and had achieved DCB. Five patients were treated with olaparib and 1 with rucaparib. The median PSA progression-free survival was 8.9 (interquartile range = 8.5 – 11.2) months among these selected patients. Regarding gene variants, 5 patients had 7 BRCA2 mutations, including 4 frameshift, 1 nonsense, 1 single nucleotide variant, and 1 splice site. One patient had frameshift and missense ATM mutations. Referrals to the NPOP consult service were ordered for 2 out of the 5 patients with BRCA2 mutations achieving DCB.
CONCLUSIONS: Within the VA’s NPOP, the presence of BRCA2 gene variants was the most common finding from tumor DNA sequencing among patients with prostate cancer achieving DCB with a PARPi. Further analysis of the genotypes of all patients treated with PARPi in NPOP to assess the differential impact of BRCA2 mutations is needed to confirm the clinical implication of this finding.
Clinical and Dosimetric Predictors of Toxicity for Treatment of Localized Prostate Cancer Using Moderately Hypofractionated Radiotherapy
PURPOSE: Moderately hypofractionated radiotherapy (MHRT) is a commonly used treatment modality for localized prostate cancer (LPC). In this setting, dosimetric correlations to acute and late toxicities remain poorly defined.
METHODS: Patients with LPC treated with MHRT between September 2008 and April 2018 were retrospectively identified. We excluded those with < 12 months follow up, elective nodal coverage, or additional boost. All patients received either 70Gy/28 fractions or 60Gy/20 fractions. Demographics, clinical outcomes, and toxicity data were obtained. Acute and late (≥3 months following MHRT completion) gastrointestinal (GI) and genitourinary (GU) toxicities were determined per CTCAE 5.0. Univariate and multivariate analyses were performed for acute and late grade 2+ GI/GU toxicity via logistic regression and log rank testing for demographic and dosimetric variables.
RESULTS: A total of 436 patients with LPC were treated with MHRT. Mean age was 64 years (IQR 60-68), median pre-treatment PSA was 8.7 (IQR 5.7-12.2), and T stages included T1a/2a (357), T2b/2c (58), and T3 (21). Acute grade 3 GU and GI toxicities were observed in 16(3.7%) and 3(0.7%) patients respectively, with no acute grade 4 toxicity events. Late grade 3 GU and GI toxicities were observed in 17(3.9%) and 4(0.9%) patients respectively, with two late grade 4 GI (0.05%) events. On multivariate analysis, acute grade 2+ GU toxicity was associated with pre-treatment PSA (odds ratio [OR] 1.02 95% confidence interval [CI] 1.01-1.04, P = 0.011) and pre-radiotherapy AUA SS (OR 1.06 95%CI: 1.03-1.09, P < 0.001); late grade 2+ GU toxicity was associated with pre-treatment AUA (hazard ratio [HR] 1.04 95%CI: 1.02-1.06, P < 0.001), lack of pre-treatment urinary meds (HR 0.65, 95%CI: 0.46-0.92, P = 0.049), and ADT use (HR 1.45, 95%CI: 1.03-2.03, P = 0.034); acute grade 2+ GI toxicity did demonstrate significant correlation; late grade 2+ GI toxicity was associated with ethnicity (Black vs White, HR 0.50, 95% CI: 0.25-0.99, P = 0.008) and pre-treatment PSA (HR 1.02, 95%CI: 1.00- 1.03, P = 0.024).
CONCLUSION: LPC patients completing MHRT experienced low rates of grade 3+ acute and late GU/GI toxicities. No dosimetric variables demonstrated significance on multivariate analysis of acute or late GU/ GI grade 2+ toxicity. Late grade 2+ GU toxicity was associated with ADT use, while late grade 2+ GI toxicity was associated with ethnicity and pre-treatment PSA.
PURPOSE: Moderately hypofractionated radiotherapy (MHRT) is a commonly used treatment modality for localized prostate cancer (LPC). In this setting, dosimetric correlations to acute and late toxicities remain poorly defined.
METHODS: Patients with LPC treated with MHRT between September 2008 and April 2018 were retrospectively identified. We excluded those with < 12 months follow up, elective nodal coverage, or additional boost. All patients received either 70Gy/28 fractions or 60Gy/20 fractions. Demographics, clinical outcomes, and toxicity data were obtained. Acute and late (≥3 months following MHRT completion) gastrointestinal (GI) and genitourinary (GU) toxicities were determined per CTCAE 5.0. Univariate and multivariate analyses were performed for acute and late grade 2+ GI/GU toxicity via logistic regression and log rank testing for demographic and dosimetric variables.
RESULTS: A total of 436 patients with LPC were treated with MHRT. Mean age was 64 years (IQR 60-68), median pre-treatment PSA was 8.7 (IQR 5.7-12.2), and T stages included T1a/2a (357), T2b/2c (58), and T3 (21). Acute grade 3 GU and GI toxicities were observed in 16(3.7%) and 3(0.7%) patients respectively, with no acute grade 4 toxicity events. Late grade 3 GU and GI toxicities were observed in 17(3.9%) and 4(0.9%) patients respectively, with two late grade 4 GI (0.05%) events. On multivariate analysis, acute grade 2+ GU toxicity was associated with pre-treatment PSA (odds ratio [OR] 1.02 95% confidence interval [CI] 1.01-1.04, P = 0.011) and pre-radiotherapy AUA SS (OR 1.06 95%CI: 1.03-1.09, P < 0.001); late grade 2+ GU toxicity was associated with pre-treatment AUA (hazard ratio [HR] 1.04 95%CI: 1.02-1.06, P < 0.001), lack of pre-treatment urinary meds (HR 0.65, 95%CI: 0.46-0.92, P = 0.049), and ADT use (HR 1.45, 95%CI: 1.03-2.03, P = 0.034); acute grade 2+ GI toxicity did demonstrate significant correlation; late grade 2+ GI toxicity was associated with ethnicity (Black vs White, HR 0.50, 95% CI: 0.25-0.99, P = 0.008) and pre-treatment PSA (HR 1.02, 95%CI: 1.00- 1.03, P = 0.024).
CONCLUSION: LPC patients completing MHRT experienced low rates of grade 3+ acute and late GU/GI toxicities. No dosimetric variables demonstrated significance on multivariate analysis of acute or late GU/ GI grade 2+ toxicity. Late grade 2+ GU toxicity was associated with ADT use, while late grade 2+ GI toxicity was associated with ethnicity and pre-treatment PSA.
PURPOSE: Moderately hypofractionated radiotherapy (MHRT) is a commonly used treatment modality for localized prostate cancer (LPC). In this setting, dosimetric correlations to acute and late toxicities remain poorly defined.
METHODS: Patients with LPC treated with MHRT between September 2008 and April 2018 were retrospectively identified. We excluded those with < 12 months follow up, elective nodal coverage, or additional boost. All patients received either 70Gy/28 fractions or 60Gy/20 fractions. Demographics, clinical outcomes, and toxicity data were obtained. Acute and late (≥3 months following MHRT completion) gastrointestinal (GI) and genitourinary (GU) toxicities were determined per CTCAE 5.0. Univariate and multivariate analyses were performed for acute and late grade 2+ GI/GU toxicity via logistic regression and log rank testing for demographic and dosimetric variables.
RESULTS: A total of 436 patients with LPC were treated with MHRT. Mean age was 64 years (IQR 60-68), median pre-treatment PSA was 8.7 (IQR 5.7-12.2), and T stages included T1a/2a (357), T2b/2c (58), and T3 (21). Acute grade 3 GU and GI toxicities were observed in 16(3.7%) and 3(0.7%) patients respectively, with no acute grade 4 toxicity events. Late grade 3 GU and GI toxicities were observed in 17(3.9%) and 4(0.9%) patients respectively, with two late grade 4 GI (0.05%) events. On multivariate analysis, acute grade 2+ GU toxicity was associated with pre-treatment PSA (odds ratio [OR] 1.02 95% confidence interval [CI] 1.01-1.04, P = 0.011) and pre-radiotherapy AUA SS (OR 1.06 95%CI: 1.03-1.09, P < 0.001); late grade 2+ GU toxicity was associated with pre-treatment AUA (hazard ratio [HR] 1.04 95%CI: 1.02-1.06, P < 0.001), lack of pre-treatment urinary meds (HR 0.65, 95%CI: 0.46-0.92, P = 0.049), and ADT use (HR 1.45, 95%CI: 1.03-2.03, P = 0.034); acute grade 2+ GI toxicity did demonstrate significant correlation; late grade 2+ GI toxicity was associated with ethnicity (Black vs White, HR 0.50, 95% CI: 0.25-0.99, P = 0.008) and pre-treatment PSA (HR 1.02, 95%CI: 1.00- 1.03, P = 0.024).
CONCLUSION: LPC patients completing MHRT experienced low rates of grade 3+ acute and late GU/GI toxicities. No dosimetric variables demonstrated significance on multivariate analysis of acute or late GU/ GI grade 2+ toxicity. Late grade 2+ GU toxicity was associated with ADT use, while late grade 2+ GI toxicity was associated with ethnicity and pre-treatment PSA.