User login
Recognizing and treating trigger finger
CASE
A 55-year-old right-hand-dominant woman presented to the clinic with a chief complaint of right ring finger pain and stiffness. There was no history of trauma or prior surgery. She had no tingling or numbness. She had a history of type 2 diabetes that was well controlled. She worked as a clerk for a government office for many years, and her painful, limited finger motion interfered with keyboarding and picking up items. Physical examination revealed tenderness to palpation over the palmar aspect of the metacarpophalangeal joint (MCPJ) of the ring finger with no other joint tenderness or swelling. When she made a fist, her ring finger MCPJ, proximal interphalangeal joint (PIPJ), and distal interphalangeal joint (DIPJ) locked in a flexed position that required manipulation to extend the finger. A firm mass was palpated in the palm with finger flexion that moved into the finger with extension.
Stenosing tenosynovitis, also known as trigger finger (TF), is an inflammatory condition that causes pain in the distal palm and proximal digit with associated limited motion. The most commonly affected digits are the middle and ring fingers of the dominant hand.1 The disorder is particularly noticeable when it inhibits day-to-day functioning.
TF affects 2% to 3% of the general population and up to 20% of patients with diabetes.2,3 Patient age and duration of diabetes are commonly cited as contributing factors, although the effect of well-controlled blood glucose and A1C on the frequency and cure rate of TF has not been established.3,4 TF is most commonly seen in individuals ages 40 to 60 years, with a 6 times’ greater frequency in females than males.5
In the United States, there are an estimated 200,000 cases of TF each year, with initial presentation typically being to a primary care physician.6 For this reason, it is essential for primary care physicians to recognize this common pathology and treat symptoms early to prevent progression and the need for surgical intervention.
An impaired gliding motion of the flexor tendons
In each finger, a tendon sheath, consisting of 5 annular pulleys and 3 cruciate pulleys, forms a tunnel around the flexor digitorum profundus (FDP) and flexor digitorum superficialis (FDS). The tendon sheath allows for maximum force by eliminating bowstringing of the tendons when the digit is flexed. Deep to the tendons and surrounding the tendons is a synovial membrane that provides nutrition and reduces friction between the tendons and the tendon sheath.7
The FDP is longer and assists in flexion of the MCPJ and the PIPJ. It is the sole flexor of the DIPJ.
In the thumb, the flexor pollicis longus (FPL) is the only flexor within its tendon sheath. The FPL assists in flexion of the MCPJ and flexes the thumb interphalangeal joint (IPJ). The intrinsic muscles (lumbricals and interossei) do not extend into the tendon sheath and do not contribute to TF.
Continue to: TF occurs when
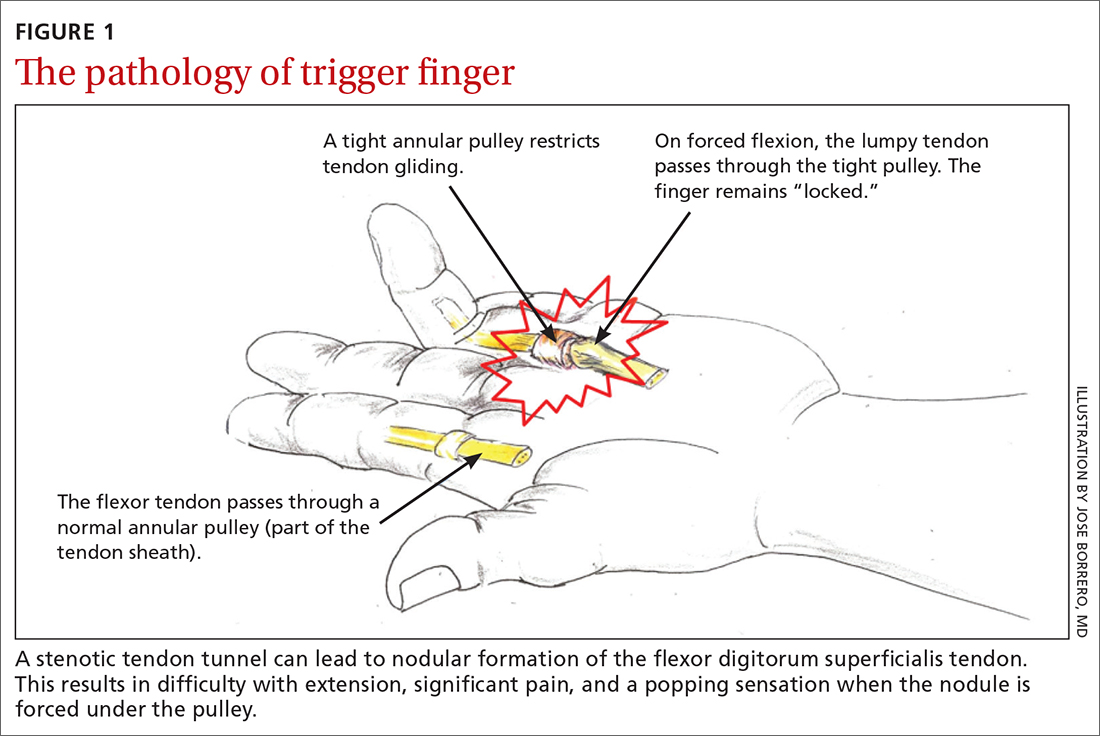
TF occurs when the tendon sheath, most commonly at the first annular pulley (A1), or the flexor tendons thicken due to fibrocartilaginous metaplasia. This results in impaired gliding motion of the flexor tendons.8 The stenosed A1 pulley can lead to pinching of the flexor tendons and cause the formation of a nodule on the FDS tendon at its bifurcation.9 The nodule of the FDS bifurcation moves proximal to the A1 pulley when the finger is flexed. Upon extension, the tendon nodule may get caught on the A1 pulley. This prevents smooth extension and is the source of pain and triggering (FIGURE 1). In a similar manner, thumb triggering is the result of a stenosed A1 pulley creating a nodule on the FPL tendon, which prevents smooth gliding of the FPL.
What you’ll see
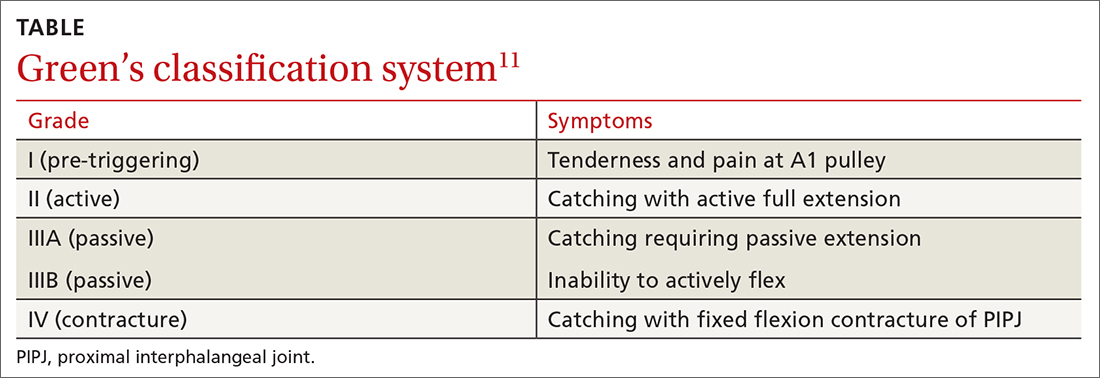
TF is characterized by locking, popping, or clicking at the base of the finger or thumb.7,10 A small nodule may be palpated on the palmar aspect of the MCPJ when the finger is flexed. This nodule will then move distally when the finger is extended. Patients will present with the affected digit in a flexed position and will have difficulty extending the digit. In some cases, the patient may have to use the other hand to straighten the affected digit. In more severe cases, the digit may be fixed in a position of flexion or extension. The severity of triggering is commonly graded by the Green’s classification system (see TABLE11).
Is it Dupuytren contracture, trigger finger, or something else?
The differential diagnosis for TF includes Dupuytren contracture, MCPJ sprain, calcific peritendinitis, flexor tenosynovitis, diabetic cheiroarthropathy (DCA), rheumatoid arthritis (RA), osteoarthritis (OA), and crystalline arthropathy (gout).5
Dupuytren contracture is usually nonpainful and manifests with a palpable cord in the palm and a fixed flexion contracture that has progressed over time, with no history of catching.
MCPJ sprain is diagnosed with tenderness of the MCPJ and a history of trauma.
Continue to: Calcific peritendinitis
Calcific peritendinitis is characterized by pain, tenderness, and edema near a joint with calcified deposits seen on radiographs.
Flexor tenosynovitis manifests with fusiform swelling of the digit, tenderness over the flexor tendon sheath, and pain with passive extension of the digit; it is more commonly associated with RA.
DCA, RA, OA, and gout usually affect more than 1 digit. DCA is associated with both type 1 and type 2 diabetes and is characterized by thickened, waxy skin and painless, limited extension of the digits. RA and OA are diagnosed by medical history, lab work, and radiographs. Gout is diagnosed with lab work and aspiration of joint fluid.
A thorough history, physical exam, and review of radiographs must be performed to rule out these other disorders. Once the diagnosis of TF is made, available treatment options should be pursued.
Treatment: A conservative or surgical approach?
Current treatment options include both nonsurgical (conservative) and surgical interventions. Nonsurgical interventions include activity modification, splinting, and corticosteroid injections. While nonsteroidal anti-inflammatory drugs are commonly recommended to resolve the local inflammation secondary to triggering, there is no scientific evidence to support their use at this time.7 Surgical interventions, utilized in more severe cases or after conservative treatment has failed, include percutaneous and open release of the tendon sheath.2,7
Continue to: Conservative treatments
Conservative treatments
Splinting is only an option for digits that retain flexibility (Green’s classification grades I, II, and III). The goal of splinting is to keep the affected digit in extension to avoid repeated friction between the tendon and the tendon sheath.12 This ideally allows any cartilaginous metaplasia or inflammation to resolve, subsequently alleviating symptoms. The recommended length of treatment with splinting ranges from 3 to 12 weeks, with an average of 6 weeks.1
Multiple studies have shown long-term alleviation of symptoms with the use of orthotic devices. A retrospective analysis found that 87% of patients who wore their PIPJ orthotic device both day and night for a minimum of 6 weeks required no further treatment at 1-year follow-up.13 In contrast, MCPJ splinting only at night has been shown to resolve symptoms in just 55% of patients after 6 weeks.14 From a practical standpoint, however, patients are more likely to be compliant with night-only splinting, making it a reasonable option. Splinting does remain efficacious for patients even after 6 months of symptomatology.15
Day and night splinting for approximately 8 weeks using a PIPJ orthotic could be considered as an effective first-line intervention.16 Notably, PIPJ splinting is more functional, as it allows motion of the MCPJ and DIPJ.
An adjunct treatment to splinting is tendon-gliding exercises, including passive IPJ flexion, full finger flexion and extension, and hooking.13 Patients may remove the orthotic device to perform these exercises 3 times a day for 5 repetitions, as well as for activities that are not conducive to splinting.13
Corticosteroid injections. Injections of a corticosteroid and 1% lidocaine in a 1:1 mixture for a total volume of 1 cc can be inserted into the tendon sheath, A1 pulley, or adjacent tissue.17 Steroid injections help to decrease inflammation and pain in the affected area, giving symptom relief lasting a few months in as many as 57% to 87% of patients.18
Continue to: While the location of the injection...
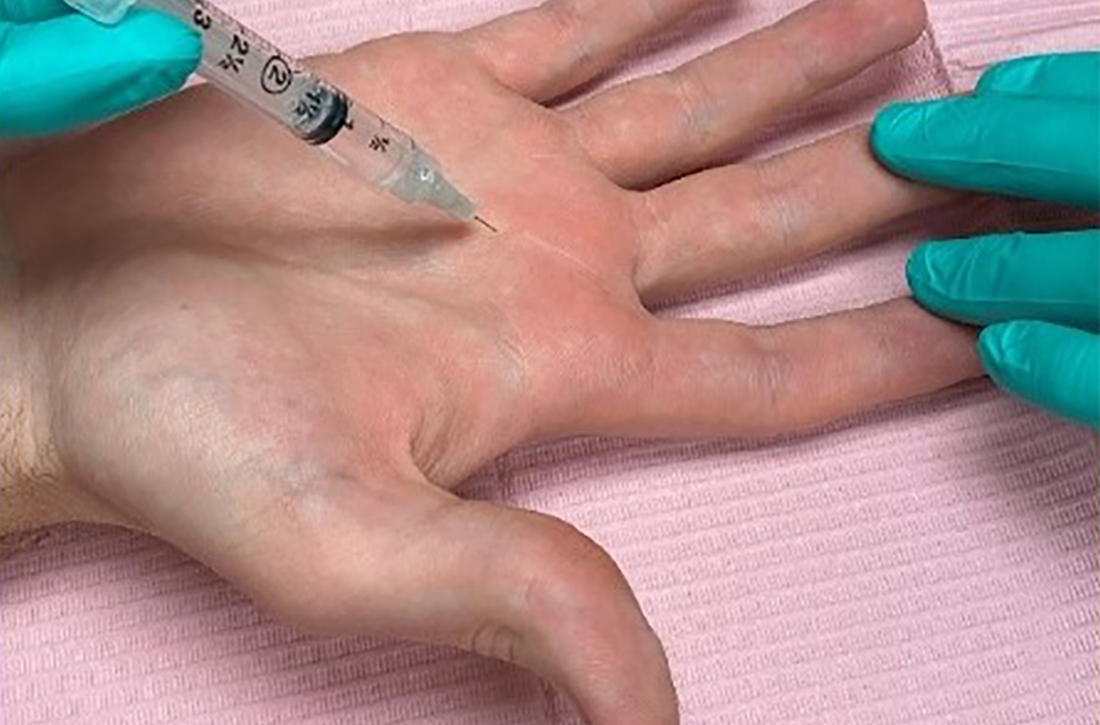
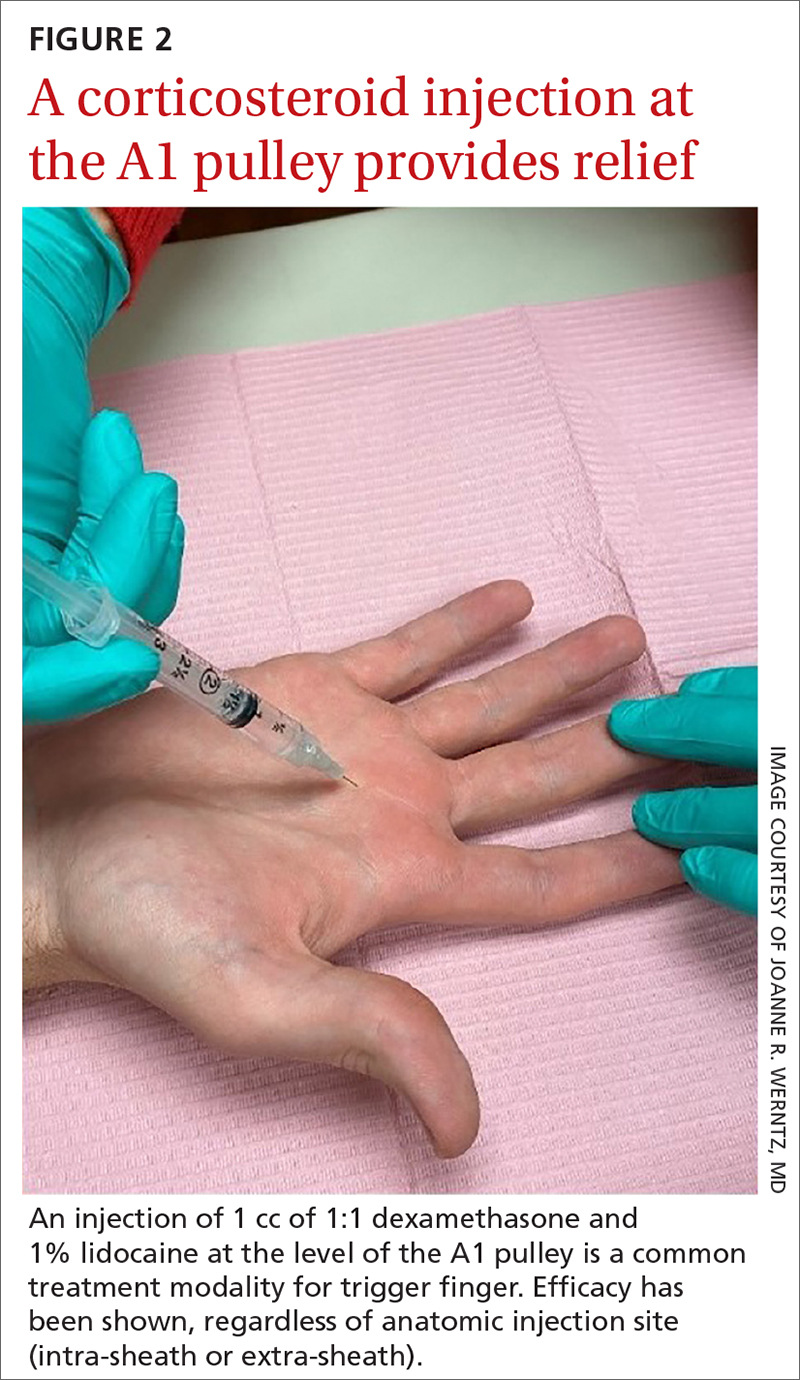
While the location of the injection has been debated, recent literature suggests that symptoms can be effectively alleviated regardless of the specific anatomic injection site, such as intra-sheath or extra-sheath (FIGURE 2).19 This allows flexibility for the clinician, as the injection does not have to be placed within the tendon sheath. Corticosteroids should not be injected into the tendon itself, and the needle tip should be slightly withdrawn if there is resistance while injecting. Patients who are averse to injections have been shown to benefit from needle-free jet lidocaine (J-tip) administration prior to the actual steroid injection.20
A randomized controlled trial comparing dexamethasone to triamcinolone injections found no difference in outcome at the 3-month follow-up (n = 84).17 This may suggest that the choice of corticosteroid is at the clinician’s discretion. In terms of long-term efficacy of steroid injections, it has been shown that 70% of trigger digits had complete resolution of symptoms at a mean follow-up of 8 years after just 1 injection (n = 43).21
Some patients, though, may require additional corticosteroid injections to maintain symptom control. If multiple injections are performed, they should not be given in intervals shorter than 4 months between treatments.5 Furthermore, steroids can be administered safely up to 3 times in the same digit before surgery is recommended.22
A patient’s options should be reconsidered if efficacy is not demonstrated with prior injections. Notably, a lower success rate has been shown in patients with type 2 diabetes (66%) compared to those without diabetes (90%).4,23 This difference in success rates is not well understood, as there is no causal relationship between well-controlled diabetes and TF.4 Complications of corticosteroid injections include local pain, fat atrophy, and hypopigmentation at the site of the injection, as well as short-term elevations in blood glucose levels in patients with diabetes.5,24
Surgical correction (to be discussed) remains superior to steroid injections in terms of cure rate and resolution of symptoms. A randomized controlled trial (n = 165) found that an injection-only group reported 86% and 49% success at 3-month and 12-month follow-up, respectively, compared to 99% success at both 3- and 12-month follow-up for the surgical group. Further, at 12-month follow-up, the median pain scores were significantly higher in the injection group (3; range, 1-9) than in the surgery group (1; range, 1-7).25 If conservative treatment modalities lead to unresolved symptoms or recurrence, referral to a hand specialist for surgery is recommended.
Continue to: Surgical treatments in an office setting
Surgical treatments in an office setting
Procedures for TF can be safely performed under conscious sedation or local anesthesia, with or without a tourniquet.26 Wide-awake procedures with local anesthesia and no tourniquet (WALANT) can be performed in an office-based procedure room rather than the operating room. This increases efficiency for the surgeon, reduces the amount of preparation and recovery time for the patient, and helps to keep costs down.
Percutaneous release involves the insertion of a 16-gauge hypodermic needle into the affected A1 pulley. The needle is used to fray and disrupt the pulley by moving the needle tip over the fibrotic A1 pulley.
However, it is not without possible complications.27 Inadvertent A2 pulley damage is particularly troublesome, as it leads to “bowstringing” or protrusion of the flexor tendon into the palm upon flexion. This can cause pain and failure to fully extend or flex the finger.10 Because the anatomy is not well visualized during the percutaneous approach, incomplete release, neurovascular injury, and iatrogenic injury to the A2 pulley or deep tendon may occur.28 Ultrasound-guided percutaneous release techniques have shown effective clinical outcomes with minimal complications compared to nonguided percutaneous release techniques.29,30
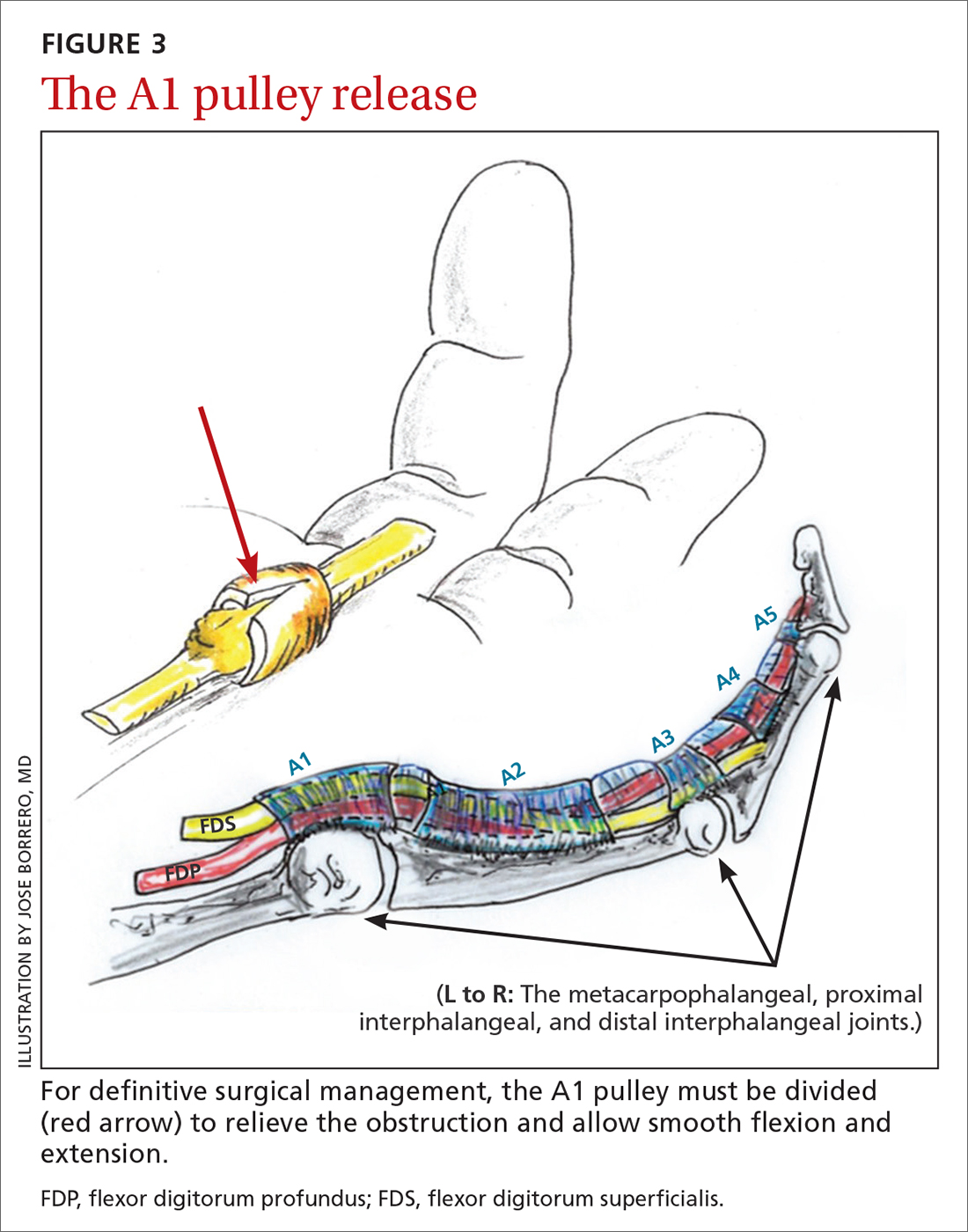
Open release is the gold standard surgical treatment for trigger finger (FIGURE 3). A small incision (1-2 cm) is made directly over or proximal to the A1 pulley in the distal palmar crease at the base of the affected digit. After blunt dissection through the subcutaneous tissue, the A1 pulley is sharply incised. An open approach has the clear benefit of avoiding the digital neurovascular bundles, as well as visualizing the resolution of triggering upon flexion and extension prior to closure. The WALANT procedure has the advantage of allowing the awake patient to actively flex and extend the digit to determine if the A1 release has been successful prior to closure of the incision.
Outcomes and complications of surgery. A recent systematic review and meta-analysis has shown percutaneous techniques to be successful in 94% of cases.27 The success rate of open surgery has been reported at 99% to 100% at varying follow-up intervals up to 1 year.25,30,31 The complication rate for percutaneous release (guided and nonguided) was calculated at 2.2% (n = 2114).27 In another study, the overall complication rate of open releases was calculated at 1% (n = 999).32 When comparing percutaneous release (guided and nonguided) and open release, a meta-analysis found no significant difference in complication rate (RR = 0.84) or failure rate (RR = 0.94).32
Continue to: Several risk factors...
Several risk factors have been associated with postoperative surgical infection, including recent steroid injection (< 80 d), smoking status, increasing age, and pre-operative use of lidocaine with epinephrine.33 Open release has been shown to be an effective and safe treatment modality for patients with and without diabetes alike.34 Overall, definitive surgical correction has been demonstrated to be superior to conservative measures due to a significantly lower rate of recurrence.35
CASE
Given the patient’s presentation with triggering of the digit, tenderness over the A1 pulley, and lack of trauma history, we diagnosed trigger finger in this patient. Potential treatments included splinting, corticosteroid injections, and surgery. After discussion of the risks and benefits of each treatment option, the patient elected to undergo a corticosteroid injection. She was also given a neoprene finger sleeve to wear every night, and in the daytime when possible.
At 12-week follow-up, she noted early improvement in her triggering, which had since recurred. Due to her history of diabetes, the patient was then referred for surgery. She had an open release under local anesthesia. The surgery was uncomplicated, and the abnormality was corrected. At the patient’s 1-year postoperative follow-up visit, there was no evidence of recurrence, and she had regained full active and passive range of motion of her finger.
Acknowledgements
The authors wish to thank Jose Borrero, MD, for contributing his time and creative talents to produce the illustrations in this article.
CORRESPONDENCE
Evan P. Johnson, MD; 506 South Greer Street, Memphis, TN 38111; EvanJohnsonOrtho@gmail.com
1. Lunsford D, Valdes K, Hengy S. Conservative management of trigger finger: a systematic review. J Hand Ther. 2019;32:212-221. doi: 10.1016/j.jht.2017.10.016
2. Makkouk AH, Oetgen ME, Swigart CR, et al. Trigger finger: etiology, evaluation, and treatment. Curr Rev Musculoskelet Med. 2008;1:92-96. doi: 10.1007/s12178-007-9012-1
3. Fitzgibbons PG, Weiss AP. Hand manifestations of diabetes mellitus. J Hand Surg Am. 2008;33:771-775. doi: 10.1016/j.jhsa.2008.01.038
4. Junot HSN, Anderson Hertz AFL, Gustavo Vasconcelos GR, et al. Epidemiology of trigger finger: metabolic syndrome as a new perspective of associated disease. Hand (N Y). 2019:1558944719867135. doi: 10.1177/1558944719867135.
5. Matthews A, Smith K, Read L, et al. Trigger finger: an overview of the treatment options. JAAPA. 2019;32:17-21. doi: 10.1097/01.Jaa.0000550281.42592.97
6. Pencle FJ, Waheed A, Molnar JA. Trigger thumb. StatPearls [Internet]. StatPearls Publishing; 2020. www.ncbi.nlm.nih.gov/books/NBK441854/
7. Giugale JM, Fowler JR. Trigger finger: adult and pediatric treatment strategies. Orthop Clin North Am. 2015;46:561-569. doi: 10.1016/j.ocl.2015.06.014
8. Bianchi S, Gitto S, Draghi F. Ultrasound features of trigger finger: review of the literature. J Ultrasound Med. 2019;38:3141-3154. doi: 10.1002/jum.15025
9. Chuang XL, Ooi CC, Chin ST, et al. What triggers in trigger finger? The flexor tendons at the flexor digitorum superficialis bifurcation. J Plast Reconstr Aesthet Surg. 2017;70:1411-1419. doi: 10.1016/j.bjps.2017.05.037
10. Ryzewicz M, Wolf JM. Trigger digits: principles, management, and complications. J Hand Surg Am. 2006;31:135-146. doi: 10.1016/j.jhsa.2005.10.013
11. Chapter 56: Tendinoapthy. In: Wolfe SW, Peterson WC, Kozin SH, Cohen MS. Green’s Operative Hand Surgery. Vol 2. 7th ed. Elsevier; 2017: 1904-1925.
12. Tarbhai K, Hannah S, von Schroeder HP. Trigger finger treatment: a comparison of 2 splint designs. J Hand Surg Am. 2012;37:243-249, 249.e241. doi: 10.1016/j.jhsa.2011.10.038
13. Valdes K. A retrospective review to determine the long-term efficacy of orthotic devices for trigger finger. J Hand Ther. 2012;25:89-95. doi: 10.1016/j.jht.2011.09.005
14. Drijkoningen T, van Berckel M, Becker SJE, et al. Night splinting for idiopathic trigger digits. Hand (N Y). 2018;13:558-562. doi: 10.1177/1558944717725374
15. Colbourn J, Heath N, Manary S, et al. Effectiveness of splinting for the treatment of trigger finger. J Hand Ther. 2008;21:336-343. doi: 10.1197/j.jht.2008.05.001
16. Teo SH, Ng DCL, Wong YKY. Effectiveness of proximal interphalangeal joint-blocking orthosis vs metacarpophalangeal joint-blocking orthosis in trigger digit management: A randomized clinical trial. J Hand Ther. 2018;32:444-451. doi: 10.1016/j.jht.2018.02.007
17. Ring D, Lozano-Calderon S, Shin R, et al. A prospective randomized controlled trial of injection of dexamethasone versus triamcinolone for idiopathic trigger finger. J Hand Surg Am. 2008;33:516-522; discussion 523-514. doi: 10.1016/j.jhsa.2008.01.001
18. Fleisch SB, Spindler KP, Lee DH. Corticosteroid injections in the treatment of trigger finger: A level I and II systematic review. J Am Acad Orthop Surg. 2007;15:166-171. doi: 10.5435/00124635-200703000-00006
19. Shinomiya R, Sunagawa T, Nakashima Y, et al. Impact of corticosteroid injection site on the treatment success rate of trigger finger: a prospective study comparing ultrasound-guided true intra-sheath and true extra-sheath injections. Ultrasound Med Biol. 2016;42:2203-2208. doi: 10.1016/j.ultrasmedbio.2016.05.015
20. Earp BE, Stanbury SJ, Mora AN, et al. Needle-free jet lidocaine administration for preinjection anesthesia in trigger finger injection: a randomized controlled trial. J Hand Surg Am. 2017;42:618-622. doi: 10.1016/j.jhsa.2017.05.001
21. Castellanos J, Munoz-Mahamud E, Dominguez E, et al. Long-term effectiveness of corticosteroid injections for trigger finger and thumb. J Hand Surg Am. 2015;40:121-126. doi: 10.1016/j.jhsa.2014.09.006
22. Dala-Ali BM, Nakhdjevani A, Lloyd MA, et al. The efficacy of steroid injection in the treatment of trigger finger. Clin Orthop Surg. 2012;4:263-268. doi: 10.4055/cios.2012.4.4.263
23. Griggs SM, Weiss AP, Lane LB, et al. Treatment of trigger finger in patients with diabetes mellitus. J Hand Surg Am. 1995;20:787-789. doi: 10.1016/s0363-5023(05)80432-0
24. Stepan JG, London DA, Boyer MI, et al. Blood glucose levels in diabetic patients following corticosteroid injections into the hand and wrist. J Hand Surg Am. 2014;39:706-712. doi: 10.1016/j.jhsa.2014.01.014
25. Hansen RL, Sondergaard M, Lange J. Open surgery versus ultrasound-guided corticosteroid injection for trigger finger: a randomized controlled trial with 1-year follow-up. J Hand Surg Am. 2017;42:359-366. doi: 10.1016/j.jhsa.2017.02.011
26. Mohd Rashid MZ, Sapuan J, Abdullah S. A randomized controlled trial of trigger finger release under digital anesthesia with (WALANT) and without adrenaline. J Orthop Surg (Hong Kong). 2019;27:2309499019833002. doi: 10.1177/2309499019833002
27. Zhao J-G, Kan S-L, Zhao L, et al. Percutaneous first annular pulley release for trigger digits: a systematic review and meta-analysis of current evidence. J Hand Surg Am. 2014;39:2192-2202. doi: 10.1016/j.jhsa.2014.07.044
28. Guler F, Kose O, Ercan EC, et al. Open versus percutaneous release for the treatment of trigger thumb. Orthopedics. 2013;36:e1290-1294. doi: 10.3928/01477447-20130920-22
29. Wu KC, Chern TC, Jou IM. Ultrasound-assisted percutaneous trigger finger release: it is safe [letter]. Hand (N Y). 2009;4:339. doi: 10.1007/s11552-009-9179-6
30. Nikolaou VS, Malahias M-A, Kaseta M-K, et al. Comparative clinical study of ultrasound-guided A1 pulley release vs open surgical intervention in the treatment of trigger finger. World J Orthop. 2017;8:163-169. doi: 10.5312/wjo.v8.i2.163
31. Lim M-H, Lim K-K, Rasheed MZ, et al. Outcome of open trigger digit release. J Hand Surg Eur Vol. 2007;32:457-459. doi: 10.1016/j.Jhsb.2007.02.016
32. Wang J, Zhao J-G, Liang C-C. Percutaneous release, open surgery, or corticosteroid injection, which is the best treatment method for trigger digits? Clin Orthop Relat Res. 2013;471:1879-1886. doi: 10.1007/s11999-012-2716-6
33. Ng WKY, Olmscheid N, Worhacz K, et al. Steroid injection and open trigger finger release outcomes: a retrospective review of 999 digits. Hand (N Y). 2018;15:399-406. doi: 10.1177/1558944718796559
34. Ho SWL, Chia CY, Rajaratnam V. Characteristics and clinical outcomes of open surgery for trigger digits in diabetes. J Hand Microsurg. 2019;11:80-83. doi: 10.1055/s-0038-1670927
35. Sato ES, dos Santos JB, Belloti JC, et al. Percutaneous release of trigger fingers. Hand Clin. 2014;30:39-45. doi: 10.1016/j.hcl.2013.08.017
CASE
A 55-year-old right-hand-dominant woman presented to the clinic with a chief complaint of right ring finger pain and stiffness. There was no history of trauma or prior surgery. She had no tingling or numbness. She had a history of type 2 diabetes that was well controlled. She worked as a clerk for a government office for many years, and her painful, limited finger motion interfered with keyboarding and picking up items. Physical examination revealed tenderness to palpation over the palmar aspect of the metacarpophalangeal joint (MCPJ) of the ring finger with no other joint tenderness or swelling. When she made a fist, her ring finger MCPJ, proximal interphalangeal joint (PIPJ), and distal interphalangeal joint (DIPJ) locked in a flexed position that required manipulation to extend the finger. A firm mass was palpated in the palm with finger flexion that moved into the finger with extension.
Stenosing tenosynovitis, also known as trigger finger (TF), is an inflammatory condition that causes pain in the distal palm and proximal digit with associated limited motion. The most commonly affected digits are the middle and ring fingers of the dominant hand.1 The disorder is particularly noticeable when it inhibits day-to-day functioning.
TF affects 2% to 3% of the general population and up to 20% of patients with diabetes.2,3 Patient age and duration of diabetes are commonly cited as contributing factors, although the effect of well-controlled blood glucose and A1C on the frequency and cure rate of TF has not been established.3,4 TF is most commonly seen in individuals ages 40 to 60 years, with a 6 times’ greater frequency in females than males.5
In the United States, there are an estimated 200,000 cases of TF each year, with initial presentation typically being to a primary care physician.6 For this reason, it is essential for primary care physicians to recognize this common pathology and treat symptoms early to prevent progression and the need for surgical intervention.
An impaired gliding motion of the flexor tendons
In each finger, a tendon sheath, consisting of 5 annular pulleys and 3 cruciate pulleys, forms a tunnel around the flexor digitorum profundus (FDP) and flexor digitorum superficialis (FDS). The tendon sheath allows for maximum force by eliminating bowstringing of the tendons when the digit is flexed. Deep to the tendons and surrounding the tendons is a synovial membrane that provides nutrition and reduces friction between the tendons and the tendon sheath.7
The FDP is longer and assists in flexion of the MCPJ and the PIPJ. It is the sole flexor of the DIPJ.
In the thumb, the flexor pollicis longus (FPL) is the only flexor within its tendon sheath. The FPL assists in flexion of the MCPJ and flexes the thumb interphalangeal joint (IPJ). The intrinsic muscles (lumbricals and interossei) do not extend into the tendon sheath and do not contribute to TF.
Continue to: TF occurs when
TF occurs when the tendon sheath, most commonly at the first annular pulley (A1), or the flexor tendons thicken due to fibrocartilaginous metaplasia. This results in impaired gliding motion of the flexor tendons.8 The stenosed A1 pulley can lead to pinching of the flexor tendons and cause the formation of a nodule on the FDS tendon at its bifurcation.9 The nodule of the FDS bifurcation moves proximal to the A1 pulley when the finger is flexed. Upon extension, the tendon nodule may get caught on the A1 pulley. This prevents smooth extension and is the source of pain and triggering (FIGURE 1). In a similar manner, thumb triggering is the result of a stenosed A1 pulley creating a nodule on the FPL tendon, which prevents smooth gliding of the FPL.

What you’ll see
TF is characterized by locking, popping, or clicking at the base of the finger or thumb.7,10 A small nodule may be palpated on the palmar aspect of the MCPJ when the finger is flexed. This nodule will then move distally when the finger is extended. Patients will present with the affected digit in a flexed position and will have difficulty extending the digit. In some cases, the patient may have to use the other hand to straighten the affected digit. In more severe cases, the digit may be fixed in a position of flexion or extension. The severity of triggering is commonly graded by the Green’s classification system (see TABLE11).

Is it Dupuytren contracture, trigger finger, or something else?
The differential diagnosis for TF includes Dupuytren contracture, MCPJ sprain, calcific peritendinitis, flexor tenosynovitis, diabetic cheiroarthropathy (DCA), rheumatoid arthritis (RA), osteoarthritis (OA), and crystalline arthropathy (gout).5
Dupuytren contracture is usually nonpainful and manifests with a palpable cord in the palm and a fixed flexion contracture that has progressed over time, with no history of catching.
MCPJ sprain is diagnosed with tenderness of the MCPJ and a history of trauma.
Continue to: Calcific peritendinitis
Calcific peritendinitis is characterized by pain, tenderness, and edema near a joint with calcified deposits seen on radiographs.
Flexor tenosynovitis manifests with fusiform swelling of the digit, tenderness over the flexor tendon sheath, and pain with passive extension of the digit; it is more commonly associated with RA.
DCA, RA, OA, and gout usually affect more than 1 digit. DCA is associated with both type 1 and type 2 diabetes and is characterized by thickened, waxy skin and painless, limited extension of the digits. RA and OA are diagnosed by medical history, lab work, and radiographs. Gout is diagnosed with lab work and aspiration of joint fluid.
A thorough history, physical exam, and review of radiographs must be performed to rule out these other disorders. Once the diagnosis of TF is made, available treatment options should be pursued.
Treatment: A conservative or surgical approach?
Current treatment options include both nonsurgical (conservative) and surgical interventions. Nonsurgical interventions include activity modification, splinting, and corticosteroid injections. While nonsteroidal anti-inflammatory drugs are commonly recommended to resolve the local inflammation secondary to triggering, there is no scientific evidence to support their use at this time.7 Surgical interventions, utilized in more severe cases or after conservative treatment has failed, include percutaneous and open release of the tendon sheath.2,7
Continue to: Conservative treatments
Conservative treatments
Splinting is only an option for digits that retain flexibility (Green’s classification grades I, II, and III). The goal of splinting is to keep the affected digit in extension to avoid repeated friction between the tendon and the tendon sheath.12 This ideally allows any cartilaginous metaplasia or inflammation to resolve, subsequently alleviating symptoms. The recommended length of treatment with splinting ranges from 3 to 12 weeks, with an average of 6 weeks.1
Multiple studies have shown long-term alleviation of symptoms with the use of orthotic devices. A retrospective analysis found that 87% of patients who wore their PIPJ orthotic device both day and night for a minimum of 6 weeks required no further treatment at 1-year follow-up.13 In contrast, MCPJ splinting only at night has been shown to resolve symptoms in just 55% of patients after 6 weeks.14 From a practical standpoint, however, patients are more likely to be compliant with night-only splinting, making it a reasonable option. Splinting does remain efficacious for patients even after 6 months of symptomatology.15
Day and night splinting for approximately 8 weeks using a PIPJ orthotic could be considered as an effective first-line intervention.16 Notably, PIPJ splinting is more functional, as it allows motion of the MCPJ and DIPJ.
An adjunct treatment to splinting is tendon-gliding exercises, including passive IPJ flexion, full finger flexion and extension, and hooking.13 Patients may remove the orthotic device to perform these exercises 3 times a day for 5 repetitions, as well as for activities that are not conducive to splinting.13
Corticosteroid injections. Injections of a corticosteroid and 1% lidocaine in a 1:1 mixture for a total volume of 1 cc can be inserted into the tendon sheath, A1 pulley, or adjacent tissue.17 Steroid injections help to decrease inflammation and pain in the affected area, giving symptom relief lasting a few months in as many as 57% to 87% of patients.18
Continue to: While the location of the injection...
While the location of the injection has been debated, recent literature suggests that symptoms can be effectively alleviated regardless of the specific anatomic injection site, such as intra-sheath or extra-sheath (FIGURE 2).19 This allows flexibility for the clinician, as the injection does not have to be placed within the tendon sheath. Corticosteroids should not be injected into the tendon itself, and the needle tip should be slightly withdrawn if there is resistance while injecting. Patients who are averse to injections have been shown to benefit from needle-free jet lidocaine (J-tip) administration prior to the actual steroid injection.20

A randomized controlled trial comparing dexamethasone to triamcinolone injections found no difference in outcome at the 3-month follow-up (n = 84).17 This may suggest that the choice of corticosteroid is at the clinician’s discretion. In terms of long-term efficacy of steroid injections, it has been shown that 70% of trigger digits had complete resolution of symptoms at a mean follow-up of 8 years after just 1 injection (n = 43).21
Some patients, though, may require additional corticosteroid injections to maintain symptom control. If multiple injections are performed, they should not be given in intervals shorter than 4 months between treatments.5 Furthermore, steroids can be administered safely up to 3 times in the same digit before surgery is recommended.22
A patient’s options should be reconsidered if efficacy is not demonstrated with prior injections. Notably, a lower success rate has been shown in patients with type 2 diabetes (66%) compared to those without diabetes (90%).4,23 This difference in success rates is not well understood, as there is no causal relationship between well-controlled diabetes and TF.4 Complications of corticosteroid injections include local pain, fat atrophy, and hypopigmentation at the site of the injection, as well as short-term elevations in blood glucose levels in patients with diabetes.5,24
Surgical correction (to be discussed) remains superior to steroid injections in terms of cure rate and resolution of symptoms. A randomized controlled trial (n = 165) found that an injection-only group reported 86% and 49% success at 3-month and 12-month follow-up, respectively, compared to 99% success at both 3- and 12-month follow-up for the surgical group. Further, at 12-month follow-up, the median pain scores were significantly higher in the injection group (3; range, 1-9) than in the surgery group (1; range, 1-7).25 If conservative treatment modalities lead to unresolved symptoms or recurrence, referral to a hand specialist for surgery is recommended.
Continue to: Surgical treatments in an office setting
Surgical treatments in an office setting
Procedures for TF can be safely performed under conscious sedation or local anesthesia, with or without a tourniquet.26 Wide-awake procedures with local anesthesia and no tourniquet (WALANT) can be performed in an office-based procedure room rather than the operating room. This increases efficiency for the surgeon, reduces the amount of preparation and recovery time for the patient, and helps to keep costs down.
Percutaneous release involves the insertion of a 16-gauge hypodermic needle into the affected A1 pulley. The needle is used to fray and disrupt the pulley by moving the needle tip over the fibrotic A1 pulley.
However, it is not without possible complications.27 Inadvertent A2 pulley damage is particularly troublesome, as it leads to “bowstringing” or protrusion of the flexor tendon into the palm upon flexion. This can cause pain and failure to fully extend or flex the finger.10 Because the anatomy is not well visualized during the percutaneous approach, incomplete release, neurovascular injury, and iatrogenic injury to the A2 pulley or deep tendon may occur.28 Ultrasound-guided percutaneous release techniques have shown effective clinical outcomes with minimal complications compared to nonguided percutaneous release techniques.29,30
Open release is the gold standard surgical treatment for trigger finger (FIGURE 3). A small incision (1-2 cm) is made directly over or proximal to the A1 pulley in the distal palmar crease at the base of the affected digit. After blunt dissection through the subcutaneous tissue, the A1 pulley is sharply incised. An open approach has the clear benefit of avoiding the digital neurovascular bundles, as well as visualizing the resolution of triggering upon flexion and extension prior to closure. The WALANT procedure has the advantage of allowing the awake patient to actively flex and extend the digit to determine if the A1 release has been successful prior to closure of the incision.

Outcomes and complications of surgery. A recent systematic review and meta-analysis has shown percutaneous techniques to be successful in 94% of cases.27 The success rate of open surgery has been reported at 99% to 100% at varying follow-up intervals up to 1 year.25,30,31 The complication rate for percutaneous release (guided and nonguided) was calculated at 2.2% (n = 2114).27 In another study, the overall complication rate of open releases was calculated at 1% (n = 999).32 When comparing percutaneous release (guided and nonguided) and open release, a meta-analysis found no significant difference in complication rate (RR = 0.84) or failure rate (RR = 0.94).32
Continue to: Several risk factors...
Several risk factors have been associated with postoperative surgical infection, including recent steroid injection (< 80 d), smoking status, increasing age, and pre-operative use of lidocaine with epinephrine.33 Open release has been shown to be an effective and safe treatment modality for patients with and without diabetes alike.34 Overall, definitive surgical correction has been demonstrated to be superior to conservative measures due to a significantly lower rate of recurrence.35
CASE
Given the patient’s presentation with triggering of the digit, tenderness over the A1 pulley, and lack of trauma history, we diagnosed trigger finger in this patient. Potential treatments included splinting, corticosteroid injections, and surgery. After discussion of the risks and benefits of each treatment option, the patient elected to undergo a corticosteroid injection. She was also given a neoprene finger sleeve to wear every night, and in the daytime when possible.
At 12-week follow-up, she noted early improvement in her triggering, which had since recurred. Due to her history of diabetes, the patient was then referred for surgery. She had an open release under local anesthesia. The surgery was uncomplicated, and the abnormality was corrected. At the patient’s 1-year postoperative follow-up visit, there was no evidence of recurrence, and she had regained full active and passive range of motion of her finger.
Acknowledgements
The authors wish to thank Jose Borrero, MD, for contributing his time and creative talents to produce the illustrations in this article.
CORRESPONDENCE
Evan P. Johnson, MD; 506 South Greer Street, Memphis, TN 38111; EvanJohnsonOrtho@gmail.com
CASE
A 55-year-old right-hand-dominant woman presented to the clinic with a chief complaint of right ring finger pain and stiffness. There was no history of trauma or prior surgery. She had no tingling or numbness. She had a history of type 2 diabetes that was well controlled. She worked as a clerk for a government office for many years, and her painful, limited finger motion interfered with keyboarding and picking up items. Physical examination revealed tenderness to palpation over the palmar aspect of the metacarpophalangeal joint (MCPJ) of the ring finger with no other joint tenderness or swelling. When she made a fist, her ring finger MCPJ, proximal interphalangeal joint (PIPJ), and distal interphalangeal joint (DIPJ) locked in a flexed position that required manipulation to extend the finger. A firm mass was palpated in the palm with finger flexion that moved into the finger with extension.
Stenosing tenosynovitis, also known as trigger finger (TF), is an inflammatory condition that causes pain in the distal palm and proximal digit with associated limited motion. The most commonly affected digits are the middle and ring fingers of the dominant hand.1 The disorder is particularly noticeable when it inhibits day-to-day functioning.
TF affects 2% to 3% of the general population and up to 20% of patients with diabetes.2,3 Patient age and duration of diabetes are commonly cited as contributing factors, although the effect of well-controlled blood glucose and A1C on the frequency and cure rate of TF has not been established.3,4 TF is most commonly seen in individuals ages 40 to 60 years, with a 6 times’ greater frequency in females than males.5
In the United States, there are an estimated 200,000 cases of TF each year, with initial presentation typically being to a primary care physician.6 For this reason, it is essential for primary care physicians to recognize this common pathology and treat symptoms early to prevent progression and the need for surgical intervention.
An impaired gliding motion of the flexor tendons
In each finger, a tendon sheath, consisting of 5 annular pulleys and 3 cruciate pulleys, forms a tunnel around the flexor digitorum profundus (FDP) and flexor digitorum superficialis (FDS). The tendon sheath allows for maximum force by eliminating bowstringing of the tendons when the digit is flexed. Deep to the tendons and surrounding the tendons is a synovial membrane that provides nutrition and reduces friction between the tendons and the tendon sheath.7
The FDP is longer and assists in flexion of the MCPJ and the PIPJ. It is the sole flexor of the DIPJ.
In the thumb, the flexor pollicis longus (FPL) is the only flexor within its tendon sheath. The FPL assists in flexion of the MCPJ and flexes the thumb interphalangeal joint (IPJ). The intrinsic muscles (lumbricals and interossei) do not extend into the tendon sheath and do not contribute to TF.
Continue to: TF occurs when
TF occurs when the tendon sheath, most commonly at the first annular pulley (A1), or the flexor tendons thicken due to fibrocartilaginous metaplasia. This results in impaired gliding motion of the flexor tendons.8 The stenosed A1 pulley can lead to pinching of the flexor tendons and cause the formation of a nodule on the FDS tendon at its bifurcation.9 The nodule of the FDS bifurcation moves proximal to the A1 pulley when the finger is flexed. Upon extension, the tendon nodule may get caught on the A1 pulley. This prevents smooth extension and is the source of pain and triggering (FIGURE 1). In a similar manner, thumb triggering is the result of a stenosed A1 pulley creating a nodule on the FPL tendon, which prevents smooth gliding of the FPL.

What you’ll see
TF is characterized by locking, popping, or clicking at the base of the finger or thumb.7,10 A small nodule may be palpated on the palmar aspect of the MCPJ when the finger is flexed. This nodule will then move distally when the finger is extended. Patients will present with the affected digit in a flexed position and will have difficulty extending the digit. In some cases, the patient may have to use the other hand to straighten the affected digit. In more severe cases, the digit may be fixed in a position of flexion or extension. The severity of triggering is commonly graded by the Green’s classification system (see TABLE11).

Is it Dupuytren contracture, trigger finger, or something else?
The differential diagnosis for TF includes Dupuytren contracture, MCPJ sprain, calcific peritendinitis, flexor tenosynovitis, diabetic cheiroarthropathy (DCA), rheumatoid arthritis (RA), osteoarthritis (OA), and crystalline arthropathy (gout).5
Dupuytren contracture is usually nonpainful and manifests with a palpable cord in the palm and a fixed flexion contracture that has progressed over time, with no history of catching.
MCPJ sprain is diagnosed with tenderness of the MCPJ and a history of trauma.
Continue to: Calcific peritendinitis
Calcific peritendinitis is characterized by pain, tenderness, and edema near a joint with calcified deposits seen on radiographs.
Flexor tenosynovitis manifests with fusiform swelling of the digit, tenderness over the flexor tendon sheath, and pain with passive extension of the digit; it is more commonly associated with RA.
DCA, RA, OA, and gout usually affect more than 1 digit. DCA is associated with both type 1 and type 2 diabetes and is characterized by thickened, waxy skin and painless, limited extension of the digits. RA and OA are diagnosed by medical history, lab work, and radiographs. Gout is diagnosed with lab work and aspiration of joint fluid.
A thorough history, physical exam, and review of radiographs must be performed to rule out these other disorders. Once the diagnosis of TF is made, available treatment options should be pursued.
Treatment: A conservative or surgical approach?
Current treatment options include both nonsurgical (conservative) and surgical interventions. Nonsurgical interventions include activity modification, splinting, and corticosteroid injections. While nonsteroidal anti-inflammatory drugs are commonly recommended to resolve the local inflammation secondary to triggering, there is no scientific evidence to support their use at this time.7 Surgical interventions, utilized in more severe cases or after conservative treatment has failed, include percutaneous and open release of the tendon sheath.2,7
Continue to: Conservative treatments
Conservative treatments
Splinting is only an option for digits that retain flexibility (Green’s classification grades I, II, and III). The goal of splinting is to keep the affected digit in extension to avoid repeated friction between the tendon and the tendon sheath.12 This ideally allows any cartilaginous metaplasia or inflammation to resolve, subsequently alleviating symptoms. The recommended length of treatment with splinting ranges from 3 to 12 weeks, with an average of 6 weeks.1
Multiple studies have shown long-term alleviation of symptoms with the use of orthotic devices. A retrospective analysis found that 87% of patients who wore their PIPJ orthotic device both day and night for a minimum of 6 weeks required no further treatment at 1-year follow-up.13 In contrast, MCPJ splinting only at night has been shown to resolve symptoms in just 55% of patients after 6 weeks.14 From a practical standpoint, however, patients are more likely to be compliant with night-only splinting, making it a reasonable option. Splinting does remain efficacious for patients even after 6 months of symptomatology.15
Day and night splinting for approximately 8 weeks using a PIPJ orthotic could be considered as an effective first-line intervention.16 Notably, PIPJ splinting is more functional, as it allows motion of the MCPJ and DIPJ.
An adjunct treatment to splinting is tendon-gliding exercises, including passive IPJ flexion, full finger flexion and extension, and hooking.13 Patients may remove the orthotic device to perform these exercises 3 times a day for 5 repetitions, as well as for activities that are not conducive to splinting.13
Corticosteroid injections. Injections of a corticosteroid and 1% lidocaine in a 1:1 mixture for a total volume of 1 cc can be inserted into the tendon sheath, A1 pulley, or adjacent tissue.17 Steroid injections help to decrease inflammation and pain in the affected area, giving symptom relief lasting a few months in as many as 57% to 87% of patients.18
Continue to: While the location of the injection...
While the location of the injection has been debated, recent literature suggests that symptoms can be effectively alleviated regardless of the specific anatomic injection site, such as intra-sheath or extra-sheath (FIGURE 2).19 This allows flexibility for the clinician, as the injection does not have to be placed within the tendon sheath. Corticosteroids should not be injected into the tendon itself, and the needle tip should be slightly withdrawn if there is resistance while injecting. Patients who are averse to injections have been shown to benefit from needle-free jet lidocaine (J-tip) administration prior to the actual steroid injection.20

A randomized controlled trial comparing dexamethasone to triamcinolone injections found no difference in outcome at the 3-month follow-up (n = 84).17 This may suggest that the choice of corticosteroid is at the clinician’s discretion. In terms of long-term efficacy of steroid injections, it has been shown that 70% of trigger digits had complete resolution of symptoms at a mean follow-up of 8 years after just 1 injection (n = 43).21
Some patients, though, may require additional corticosteroid injections to maintain symptom control. If multiple injections are performed, they should not be given in intervals shorter than 4 months between treatments.5 Furthermore, steroids can be administered safely up to 3 times in the same digit before surgery is recommended.22
A patient’s options should be reconsidered if efficacy is not demonstrated with prior injections. Notably, a lower success rate has been shown in patients with type 2 diabetes (66%) compared to those without diabetes (90%).4,23 This difference in success rates is not well understood, as there is no causal relationship between well-controlled diabetes and TF.4 Complications of corticosteroid injections include local pain, fat atrophy, and hypopigmentation at the site of the injection, as well as short-term elevations in blood glucose levels in patients with diabetes.5,24
Surgical correction (to be discussed) remains superior to steroid injections in terms of cure rate and resolution of symptoms. A randomized controlled trial (n = 165) found that an injection-only group reported 86% and 49% success at 3-month and 12-month follow-up, respectively, compared to 99% success at both 3- and 12-month follow-up for the surgical group. Further, at 12-month follow-up, the median pain scores were significantly higher in the injection group (3; range, 1-9) than in the surgery group (1; range, 1-7).25 If conservative treatment modalities lead to unresolved symptoms or recurrence, referral to a hand specialist for surgery is recommended.
Continue to: Surgical treatments in an office setting
Surgical treatments in an office setting
Procedures for TF can be safely performed under conscious sedation or local anesthesia, with or without a tourniquet.26 Wide-awake procedures with local anesthesia and no tourniquet (WALANT) can be performed in an office-based procedure room rather than the operating room. This increases efficiency for the surgeon, reduces the amount of preparation and recovery time for the patient, and helps to keep costs down.
Percutaneous release involves the insertion of a 16-gauge hypodermic needle into the affected A1 pulley. The needle is used to fray and disrupt the pulley by moving the needle tip over the fibrotic A1 pulley.
However, it is not without possible complications.27 Inadvertent A2 pulley damage is particularly troublesome, as it leads to “bowstringing” or protrusion of the flexor tendon into the palm upon flexion. This can cause pain and failure to fully extend or flex the finger.10 Because the anatomy is not well visualized during the percutaneous approach, incomplete release, neurovascular injury, and iatrogenic injury to the A2 pulley or deep tendon may occur.28 Ultrasound-guided percutaneous release techniques have shown effective clinical outcomes with minimal complications compared to nonguided percutaneous release techniques.29,30
Open release is the gold standard surgical treatment for trigger finger (FIGURE 3). A small incision (1-2 cm) is made directly over or proximal to the A1 pulley in the distal palmar crease at the base of the affected digit. After blunt dissection through the subcutaneous tissue, the A1 pulley is sharply incised. An open approach has the clear benefit of avoiding the digital neurovascular bundles, as well as visualizing the resolution of triggering upon flexion and extension prior to closure. The WALANT procedure has the advantage of allowing the awake patient to actively flex and extend the digit to determine if the A1 release has been successful prior to closure of the incision.

Outcomes and complications of surgery. A recent systematic review and meta-analysis has shown percutaneous techniques to be successful in 94% of cases.27 The success rate of open surgery has been reported at 99% to 100% at varying follow-up intervals up to 1 year.25,30,31 The complication rate for percutaneous release (guided and nonguided) was calculated at 2.2% (n = 2114).27 In another study, the overall complication rate of open releases was calculated at 1% (n = 999).32 When comparing percutaneous release (guided and nonguided) and open release, a meta-analysis found no significant difference in complication rate (RR = 0.84) or failure rate (RR = 0.94).32
Continue to: Several risk factors...
Several risk factors have been associated with postoperative surgical infection, including recent steroid injection (< 80 d), smoking status, increasing age, and pre-operative use of lidocaine with epinephrine.33 Open release has been shown to be an effective and safe treatment modality for patients with and without diabetes alike.34 Overall, definitive surgical correction has been demonstrated to be superior to conservative measures due to a significantly lower rate of recurrence.35
CASE
Given the patient’s presentation with triggering of the digit, tenderness over the A1 pulley, and lack of trauma history, we diagnosed trigger finger in this patient. Potential treatments included splinting, corticosteroid injections, and surgery. After discussion of the risks and benefits of each treatment option, the patient elected to undergo a corticosteroid injection. She was also given a neoprene finger sleeve to wear every night, and in the daytime when possible.
At 12-week follow-up, she noted early improvement in her triggering, which had since recurred. Due to her history of diabetes, the patient was then referred for surgery. She had an open release under local anesthesia. The surgery was uncomplicated, and the abnormality was corrected. At the patient’s 1-year postoperative follow-up visit, there was no evidence of recurrence, and she had regained full active and passive range of motion of her finger.
Acknowledgements
The authors wish to thank Jose Borrero, MD, for contributing his time and creative talents to produce the illustrations in this article.
CORRESPONDENCE
Evan P. Johnson, MD; 506 South Greer Street, Memphis, TN 38111; EvanJohnsonOrtho@gmail.com
1. Lunsford D, Valdes K, Hengy S. Conservative management of trigger finger: a systematic review. J Hand Ther. 2019;32:212-221. doi: 10.1016/j.jht.2017.10.016
2. Makkouk AH, Oetgen ME, Swigart CR, et al. Trigger finger: etiology, evaluation, and treatment. Curr Rev Musculoskelet Med. 2008;1:92-96. doi: 10.1007/s12178-007-9012-1
3. Fitzgibbons PG, Weiss AP. Hand manifestations of diabetes mellitus. J Hand Surg Am. 2008;33:771-775. doi: 10.1016/j.jhsa.2008.01.038
4. Junot HSN, Anderson Hertz AFL, Gustavo Vasconcelos GR, et al. Epidemiology of trigger finger: metabolic syndrome as a new perspective of associated disease. Hand (N Y). 2019:1558944719867135. doi: 10.1177/1558944719867135.
5. Matthews A, Smith K, Read L, et al. Trigger finger: an overview of the treatment options. JAAPA. 2019;32:17-21. doi: 10.1097/01.Jaa.0000550281.42592.97
6. Pencle FJ, Waheed A, Molnar JA. Trigger thumb. StatPearls [Internet]. StatPearls Publishing; 2020. www.ncbi.nlm.nih.gov/books/NBK441854/
7. Giugale JM, Fowler JR. Trigger finger: adult and pediatric treatment strategies. Orthop Clin North Am. 2015;46:561-569. doi: 10.1016/j.ocl.2015.06.014
8. Bianchi S, Gitto S, Draghi F. Ultrasound features of trigger finger: review of the literature. J Ultrasound Med. 2019;38:3141-3154. doi: 10.1002/jum.15025
9. Chuang XL, Ooi CC, Chin ST, et al. What triggers in trigger finger? The flexor tendons at the flexor digitorum superficialis bifurcation. J Plast Reconstr Aesthet Surg. 2017;70:1411-1419. doi: 10.1016/j.bjps.2017.05.037
10. Ryzewicz M, Wolf JM. Trigger digits: principles, management, and complications. J Hand Surg Am. 2006;31:135-146. doi: 10.1016/j.jhsa.2005.10.013
11. Chapter 56: Tendinoapthy. In: Wolfe SW, Peterson WC, Kozin SH, Cohen MS. Green’s Operative Hand Surgery. Vol 2. 7th ed. Elsevier; 2017: 1904-1925.
12. Tarbhai K, Hannah S, von Schroeder HP. Trigger finger treatment: a comparison of 2 splint designs. J Hand Surg Am. 2012;37:243-249, 249.e241. doi: 10.1016/j.jhsa.2011.10.038
13. Valdes K. A retrospective review to determine the long-term efficacy of orthotic devices for trigger finger. J Hand Ther. 2012;25:89-95. doi: 10.1016/j.jht.2011.09.005
14. Drijkoningen T, van Berckel M, Becker SJE, et al. Night splinting for idiopathic trigger digits. Hand (N Y). 2018;13:558-562. doi: 10.1177/1558944717725374
15. Colbourn J, Heath N, Manary S, et al. Effectiveness of splinting for the treatment of trigger finger. J Hand Ther. 2008;21:336-343. doi: 10.1197/j.jht.2008.05.001
16. Teo SH, Ng DCL, Wong YKY. Effectiveness of proximal interphalangeal joint-blocking orthosis vs metacarpophalangeal joint-blocking orthosis in trigger digit management: A randomized clinical trial. J Hand Ther. 2018;32:444-451. doi: 10.1016/j.jht.2018.02.007
17. Ring D, Lozano-Calderon S, Shin R, et al. A prospective randomized controlled trial of injection of dexamethasone versus triamcinolone for idiopathic trigger finger. J Hand Surg Am. 2008;33:516-522; discussion 523-514. doi: 10.1016/j.jhsa.2008.01.001
18. Fleisch SB, Spindler KP, Lee DH. Corticosteroid injections in the treatment of trigger finger: A level I and II systematic review. J Am Acad Orthop Surg. 2007;15:166-171. doi: 10.5435/00124635-200703000-00006
19. Shinomiya R, Sunagawa T, Nakashima Y, et al. Impact of corticosteroid injection site on the treatment success rate of trigger finger: a prospective study comparing ultrasound-guided true intra-sheath and true extra-sheath injections. Ultrasound Med Biol. 2016;42:2203-2208. doi: 10.1016/j.ultrasmedbio.2016.05.015
20. Earp BE, Stanbury SJ, Mora AN, et al. Needle-free jet lidocaine administration for preinjection anesthesia in trigger finger injection: a randomized controlled trial. J Hand Surg Am. 2017;42:618-622. doi: 10.1016/j.jhsa.2017.05.001
21. Castellanos J, Munoz-Mahamud E, Dominguez E, et al. Long-term effectiveness of corticosteroid injections for trigger finger and thumb. J Hand Surg Am. 2015;40:121-126. doi: 10.1016/j.jhsa.2014.09.006
22. Dala-Ali BM, Nakhdjevani A, Lloyd MA, et al. The efficacy of steroid injection in the treatment of trigger finger. Clin Orthop Surg. 2012;4:263-268. doi: 10.4055/cios.2012.4.4.263
23. Griggs SM, Weiss AP, Lane LB, et al. Treatment of trigger finger in patients with diabetes mellitus. J Hand Surg Am. 1995;20:787-789. doi: 10.1016/s0363-5023(05)80432-0
24. Stepan JG, London DA, Boyer MI, et al. Blood glucose levels in diabetic patients following corticosteroid injections into the hand and wrist. J Hand Surg Am. 2014;39:706-712. doi: 10.1016/j.jhsa.2014.01.014
25. Hansen RL, Sondergaard M, Lange J. Open surgery versus ultrasound-guided corticosteroid injection for trigger finger: a randomized controlled trial with 1-year follow-up. J Hand Surg Am. 2017;42:359-366. doi: 10.1016/j.jhsa.2017.02.011
26. Mohd Rashid MZ, Sapuan J, Abdullah S. A randomized controlled trial of trigger finger release under digital anesthesia with (WALANT) and without adrenaline. J Orthop Surg (Hong Kong). 2019;27:2309499019833002. doi: 10.1177/2309499019833002
27. Zhao J-G, Kan S-L, Zhao L, et al. Percutaneous first annular pulley release for trigger digits: a systematic review and meta-analysis of current evidence. J Hand Surg Am. 2014;39:2192-2202. doi: 10.1016/j.jhsa.2014.07.044
28. Guler F, Kose O, Ercan EC, et al. Open versus percutaneous release for the treatment of trigger thumb. Orthopedics. 2013;36:e1290-1294. doi: 10.3928/01477447-20130920-22
29. Wu KC, Chern TC, Jou IM. Ultrasound-assisted percutaneous trigger finger release: it is safe [letter]. Hand (N Y). 2009;4:339. doi: 10.1007/s11552-009-9179-6
30. Nikolaou VS, Malahias M-A, Kaseta M-K, et al. Comparative clinical study of ultrasound-guided A1 pulley release vs open surgical intervention in the treatment of trigger finger. World J Orthop. 2017;8:163-169. doi: 10.5312/wjo.v8.i2.163
31. Lim M-H, Lim K-K, Rasheed MZ, et al. Outcome of open trigger digit release. J Hand Surg Eur Vol. 2007;32:457-459. doi: 10.1016/j.Jhsb.2007.02.016
32. Wang J, Zhao J-G, Liang C-C. Percutaneous release, open surgery, or corticosteroid injection, which is the best treatment method for trigger digits? Clin Orthop Relat Res. 2013;471:1879-1886. doi: 10.1007/s11999-012-2716-6
33. Ng WKY, Olmscheid N, Worhacz K, et al. Steroid injection and open trigger finger release outcomes: a retrospective review of 999 digits. Hand (N Y). 2018;15:399-406. doi: 10.1177/1558944718796559
34. Ho SWL, Chia CY, Rajaratnam V. Characteristics and clinical outcomes of open surgery for trigger digits in diabetes. J Hand Microsurg. 2019;11:80-83. doi: 10.1055/s-0038-1670927
35. Sato ES, dos Santos JB, Belloti JC, et al. Percutaneous release of trigger fingers. Hand Clin. 2014;30:39-45. doi: 10.1016/j.hcl.2013.08.017
1. Lunsford D, Valdes K, Hengy S. Conservative management of trigger finger: a systematic review. J Hand Ther. 2019;32:212-221. doi: 10.1016/j.jht.2017.10.016
2. Makkouk AH, Oetgen ME, Swigart CR, et al. Trigger finger: etiology, evaluation, and treatment. Curr Rev Musculoskelet Med. 2008;1:92-96. doi: 10.1007/s12178-007-9012-1
3. Fitzgibbons PG, Weiss AP. Hand manifestations of diabetes mellitus. J Hand Surg Am. 2008;33:771-775. doi: 10.1016/j.jhsa.2008.01.038
4. Junot HSN, Anderson Hertz AFL, Gustavo Vasconcelos GR, et al. Epidemiology of trigger finger: metabolic syndrome as a new perspective of associated disease. Hand (N Y). 2019:1558944719867135. doi: 10.1177/1558944719867135.
5. Matthews A, Smith K, Read L, et al. Trigger finger: an overview of the treatment options. JAAPA. 2019;32:17-21. doi: 10.1097/01.Jaa.0000550281.42592.97
6. Pencle FJ, Waheed A, Molnar JA. Trigger thumb. StatPearls [Internet]. StatPearls Publishing; 2020. www.ncbi.nlm.nih.gov/books/NBK441854/
7. Giugale JM, Fowler JR. Trigger finger: adult and pediatric treatment strategies. Orthop Clin North Am. 2015;46:561-569. doi: 10.1016/j.ocl.2015.06.014
8. Bianchi S, Gitto S, Draghi F. Ultrasound features of trigger finger: review of the literature. J Ultrasound Med. 2019;38:3141-3154. doi: 10.1002/jum.15025
9. Chuang XL, Ooi CC, Chin ST, et al. What triggers in trigger finger? The flexor tendons at the flexor digitorum superficialis bifurcation. J Plast Reconstr Aesthet Surg. 2017;70:1411-1419. doi: 10.1016/j.bjps.2017.05.037
10. Ryzewicz M, Wolf JM. Trigger digits: principles, management, and complications. J Hand Surg Am. 2006;31:135-146. doi: 10.1016/j.jhsa.2005.10.013
11. Chapter 56: Tendinoapthy. In: Wolfe SW, Peterson WC, Kozin SH, Cohen MS. Green’s Operative Hand Surgery. Vol 2. 7th ed. Elsevier; 2017: 1904-1925.
12. Tarbhai K, Hannah S, von Schroeder HP. Trigger finger treatment: a comparison of 2 splint designs. J Hand Surg Am. 2012;37:243-249, 249.e241. doi: 10.1016/j.jhsa.2011.10.038
13. Valdes K. A retrospective review to determine the long-term efficacy of orthotic devices for trigger finger. J Hand Ther. 2012;25:89-95. doi: 10.1016/j.jht.2011.09.005
14. Drijkoningen T, van Berckel M, Becker SJE, et al. Night splinting for idiopathic trigger digits. Hand (N Y). 2018;13:558-562. doi: 10.1177/1558944717725374
15. Colbourn J, Heath N, Manary S, et al. Effectiveness of splinting for the treatment of trigger finger. J Hand Ther. 2008;21:336-343. doi: 10.1197/j.jht.2008.05.001
16. Teo SH, Ng DCL, Wong YKY. Effectiveness of proximal interphalangeal joint-blocking orthosis vs metacarpophalangeal joint-blocking orthosis in trigger digit management: A randomized clinical trial. J Hand Ther. 2018;32:444-451. doi: 10.1016/j.jht.2018.02.007
17. Ring D, Lozano-Calderon S, Shin R, et al. A prospective randomized controlled trial of injection of dexamethasone versus triamcinolone for idiopathic trigger finger. J Hand Surg Am. 2008;33:516-522; discussion 523-514. doi: 10.1016/j.jhsa.2008.01.001
18. Fleisch SB, Spindler KP, Lee DH. Corticosteroid injections in the treatment of trigger finger: A level I and II systematic review. J Am Acad Orthop Surg. 2007;15:166-171. doi: 10.5435/00124635-200703000-00006
19. Shinomiya R, Sunagawa T, Nakashima Y, et al. Impact of corticosteroid injection site on the treatment success rate of trigger finger: a prospective study comparing ultrasound-guided true intra-sheath and true extra-sheath injections. Ultrasound Med Biol. 2016;42:2203-2208. doi: 10.1016/j.ultrasmedbio.2016.05.015
20. Earp BE, Stanbury SJ, Mora AN, et al. Needle-free jet lidocaine administration for preinjection anesthesia in trigger finger injection: a randomized controlled trial. J Hand Surg Am. 2017;42:618-622. doi: 10.1016/j.jhsa.2017.05.001
21. Castellanos J, Munoz-Mahamud E, Dominguez E, et al. Long-term effectiveness of corticosteroid injections for trigger finger and thumb. J Hand Surg Am. 2015;40:121-126. doi: 10.1016/j.jhsa.2014.09.006
22. Dala-Ali BM, Nakhdjevani A, Lloyd MA, et al. The efficacy of steroid injection in the treatment of trigger finger. Clin Orthop Surg. 2012;4:263-268. doi: 10.4055/cios.2012.4.4.263
23. Griggs SM, Weiss AP, Lane LB, et al. Treatment of trigger finger in patients with diabetes mellitus. J Hand Surg Am. 1995;20:787-789. doi: 10.1016/s0363-5023(05)80432-0
24. Stepan JG, London DA, Boyer MI, et al. Blood glucose levels in diabetic patients following corticosteroid injections into the hand and wrist. J Hand Surg Am. 2014;39:706-712. doi: 10.1016/j.jhsa.2014.01.014
25. Hansen RL, Sondergaard M, Lange J. Open surgery versus ultrasound-guided corticosteroid injection for trigger finger: a randomized controlled trial with 1-year follow-up. J Hand Surg Am. 2017;42:359-366. doi: 10.1016/j.jhsa.2017.02.011
26. Mohd Rashid MZ, Sapuan J, Abdullah S. A randomized controlled trial of trigger finger release under digital anesthesia with (WALANT) and without adrenaline. J Orthop Surg (Hong Kong). 2019;27:2309499019833002. doi: 10.1177/2309499019833002
27. Zhao J-G, Kan S-L, Zhao L, et al. Percutaneous first annular pulley release for trigger digits: a systematic review and meta-analysis of current evidence. J Hand Surg Am. 2014;39:2192-2202. doi: 10.1016/j.jhsa.2014.07.044
28. Guler F, Kose O, Ercan EC, et al. Open versus percutaneous release for the treatment of trigger thumb. Orthopedics. 2013;36:e1290-1294. doi: 10.3928/01477447-20130920-22
29. Wu KC, Chern TC, Jou IM. Ultrasound-assisted percutaneous trigger finger release: it is safe [letter]. Hand (N Y). 2009;4:339. doi: 10.1007/s11552-009-9179-6
30. Nikolaou VS, Malahias M-A, Kaseta M-K, et al. Comparative clinical study of ultrasound-guided A1 pulley release vs open surgical intervention in the treatment of trigger finger. World J Orthop. 2017;8:163-169. doi: 10.5312/wjo.v8.i2.163
31. Lim M-H, Lim K-K, Rasheed MZ, et al. Outcome of open trigger digit release. J Hand Surg Eur Vol. 2007;32:457-459. doi: 10.1016/j.Jhsb.2007.02.016
32. Wang J, Zhao J-G, Liang C-C. Percutaneous release, open surgery, or corticosteroid injection, which is the best treatment method for trigger digits? Clin Orthop Relat Res. 2013;471:1879-1886. doi: 10.1007/s11999-012-2716-6
33. Ng WKY, Olmscheid N, Worhacz K, et al. Steroid injection and open trigger finger release outcomes: a retrospective review of 999 digits. Hand (N Y). 2018;15:399-406. doi: 10.1177/1558944718796559
34. Ho SWL, Chia CY, Rajaratnam V. Characteristics and clinical outcomes of open surgery for trigger digits in diabetes. J Hand Microsurg. 2019;11:80-83. doi: 10.1055/s-0038-1670927
35. Sato ES, dos Santos JB, Belloti JC, et al. Percutaneous release of trigger fingers. Hand Clin. 2014;30:39-45. doi: 10.1016/j.hcl.2013.08.017
PRACTICE RECOMMENDATIONS
› Recommend splinting as a first-line conservative treatment for trigger finger if there is not a fixed contracture. B
› Prescribe corticosteroids, which may completely resolve trigger finger in the majority of patients without diabetes. A
› Refer patients for surgical release if they do not respond to conservative management. The surgical success rate is as high as 99%. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Guideline gives weak support to trying oral medical cannabis for chronic pain
“Evidence alone is not sufficient for clinical decision-making, particularly in chronic pain,” said Jason Busse, DC, PhD, director of Michael G. DeGroote Centre for Medicinal Cannabis Research at McMaster University, Hamilton, Ont., and lead author of a newly released rapid guideline on medical cannabis or cannabinoids for chronic pain.
The recommendations, published online Sept. 9, 2021 in the British Medical Journal, suggest that providers offer patients with chronic pain a trial of noninhaled medical cannabis or cannabinoids if standard care or management is ineffective. However, the “weak” rating attached to the recommendation may compel some clinicians to automatically write off the panel’s recommendations.
“Because of the close balance between benefits and harms and wide variability in patient attitudes, the panel came to the conclusion that [some] patients presented with the current best evidence would likely choose to engage in a trial of medicinal cannabis, if their current care was felt to be suboptimal,” Dr. Busse explained in an interview.
But more importantly, “the recommendation allows for shared decision making to occur, and for different patients to make different decisions based on individual preferences and circumstances,” he said.
Evidence supports improved pain and sleep quality, physical functioning
Evidence supporting the use of medical cannabis in chronic pain is derived from a rigorous systematic review and meta-analysis of 32 studies enrolling 5,174 patients randomized to oral (capsule, spray, sublingual drops) or topical (transdermal cream) medical cannabis or placebo. Of note, three types of cannabinoids were represented: phytocannabinoids, synthetic, and endocannabinoids.
The studies included both patients with chronic noncancer pain (28 studies, n = 3,812) and chronic cancer pain not receiving palliative care (4 studies, n = 1,362). On average, baseline pain scores were a median 6.28 cm on a 10-cm visual analog scale (VAS), and median participant age was 53 years. 60% of trials reporting sex differences enrolled female participants. Overall, patients were followed for roughly 2 months (median, 50 days).
Findings (27 studies, n = 3,939) showed that, compared with placebo, medical cannabis resulted in a small, albeit important, improvement in the proportion of patients experiencing pain relief at or above the minimally important difference (MID) (moderate-certainty evidence, 10% modeled risk difference [RD; 95% confidence interval, 5%-15%] for achieving at least the MID of 1 cm).
Medical cannabis (15 studies, n = 2,425) also provided a small increase in the proportion of patients experiencing improvements in physical functioning at or above the MID (high certainty evidence, 4% modeled RD [95% CI, 0.1%-8%] for achieving at least a MID of 10 points).
Additionally, participants experienced significant improvements in sleep quality, compared with placebo (16 studies, 3,124 participants, high-quality evidence), demonstrating a weighted mean difference of –0.53 cm on a 10-cm VAS (95% CI, –0.75 to –0.30 cm). A total of nine larger trials (n = 2,652, high-certainty evidence) saw a small increase in the proportion of patients experiencing improved sleep quality at or above the MID: 6% modeled RD (95% CI, 2%-9%).
On the other hand, benefits did not extend to emotional, role, or social functioning (high-certainty evidence).
First do no harm: Start low, go slow
While these findings provide a rationale for medical cannabis in chronic pain, exploring options with patients can be challenging. Studies on medical cannabis consistently note that patients want information, but data also show that many providers express a lack of knowledge to provide adequate counseling.
There are also legal hurdles. Despite the authorization of medicinal cannabis across a majority of states and territories, cannabis is still a schedule I substance under the Federal Controlled Substances Act. In addition, the absence of standards around formulations, potency, and dosing has also been cited as a major barrier to recommending medical cannabis, as have concerns about adverse events (AEs), especially with inhaled and tetrahydrocannabinol (THC)-predominant formulations.
Like most medications, medical cannabis dosing should be individualized depending on product, patient, and ability to titrate the dose, but the guidelines provide a general rule of thumb. Providers considering therapeutic noninhaled medical cannabis trials are encouraged to start with a low-dose cannabidiol (CBD) oral tablet, spray, or sublingual oil drops 5 mg twice daily, increasing it by 10 mg every 2-3 days depending on the clinical response (to a maximum daily dose of 40 mg/day). If patient response is unsatisfactory, they should consider adding 1-2.5 mg THC/daily, titrated every 2-7 days to a maximum of 40 mg/day.
Still, an important caveat is whether or not adjunctive CBD alone is effective for chronic pain.
“While we know that one out of seven U.S. adults are using cannabidiol, we know very little about its therapeutic effects when given by itself for pain,” Ziva Cooper, PhD, director of the Cannabis Research Initiative at the University of California, Los Angeles, and an associate professor at-large of psychology and behavioral science, said in an interview. (Dr. Cooper was not involved in the guideline development.)
“But patients tend to self-report that CBD is helpful, and at low doses, we know that it is unlikely to have adverse effects of any significant concern,” Dr. Cooper noted.
Depending on its components, medical cannabis is associated with a wide range of AEs. Studies comprising the evidence base for the guideline reported transient cognitive impairment (relative risk, 2.39; 95% CI, 1.06-5.38), vomiting (RR, 1.46; 95% CI, 1.07-1.99), and drowsiness (RR, 2.14; 95% CI, 1.55-2.95), attention impairment (RR, 4.04; 95% CI, 1.67-9.74), and nausea (RR, 1.59; 95% CI, 1.28-1.99). Of note, findings of a subgroup analysis showed that the risk of dizziness increased with treatment duration, starting at 3 months (test of interaction P = .002).
However, Dr. Cooper explained that, because the included studies were inconsistent in terms of cannabis type (e.g., some looked at synthetic THC or THC-like substances where others looked at a THC/CBD combination) and formulation (capsules, oral mucosal sprays), it’s difficult to tease out component-specific AEs.
“These are really important things to note, especially when you think about different populations that might be using these types of medicines moving forward,” she said.
Toward that end, the guideline specifically states that there is “no reason why the expected benefits would be systematically different among adolescents and emerging adults.”
Among children with cancer, prior study findings reinforce the conclusion that benefits are similar to adults, but studies in this area are limited to end-of-life treatment, childhood cancer with primarily palliative intent, or progressive or relapsed cancer. Because THC’s safety profile is less certain in children, it’s also important to consider adverse neurocognitive effects before initiating a medical cannabis trial in this population.
Navigating the landscape
Although promising, the medical cannabis landscape is undoubtedly difficult to navigate, with land mines ranging from a limited inability to simply pick up a prescribing pad to quality control.
With the exception of three Food and Drug Administration–approved products – dronabinol, cannabidiol Rx, and nabilone – U.S. providers are only able to ‘certify,’ not prescribe, medical cannabis for chronic pain, and only if it is included within the state cannabis board’s list of eligible conditions. (A state-by-state guide is available.)
Quality control also varies by product but is critical. “You want to look for certificates of quality assurance,” Jenny Wilkerson, PhD, a research assistant professor of pharmacodynamics at the University of Florida, Gainesville, said in an interview. (Dr. Wilkerson was not involved in the guideline development.)
“A good dispensary should have that information or at least be willing to get that information, but generally speaking, that is something that patients need to ask for,” she emphasized, noting that “most available mass readouts are not divided by lots.”
Initial counseling and AE monitoring and regular follow-up is important, especially among patients who’ve never tried medical cannabis (or older patients whose prior experience may be limited to weaker recreational marijuana).
Notably, the reliance on medical dispensaries to deliver the right information at the right time may prove to be faulty. While recent data show that frontline dispensary workers regularly provide information to customers on their medical conditions and available products, they rarely, if ever, base recommendations on provider input, and never or rarely discuss potential AEs and other risks.
Per the new guideline, inexperienced patients should be seen monthly until a stable dose is achieved; longer times between visits can be considered in those who are more experienced. Still, patients should be advised to contact their provider when pain relief or other goals are insufficient, or when response or problematic AEs occur. This facilitates down-titration to a previously tolerated dose, up-titration in CBD and/or THC, or a different route of administration/formulation altogether.
Dr. Wilkerson pointed out that follow-up visits also provide an opportunity to do a blood draw and ask the lab to conduct pharmacokinetic analysis.
If possible, “ask patients to [ensure that they] take a standard dose before the visit so that the lab can assess the blood percentage of primary compounds and metabolites in the product that they are using,” she explained, noting that the information is helping to determine how “the different ratios may be affecting therapeutic response in individual patients.”
Granted, the guideline is only a start. But it is a good one.
“A lot of physicians want to be able to hang their hat on evidence of the safety and efficacy of these products, and the analysis that was leveraged for this guideline was very rigorous,” Dr. Cooper said.
Not only do they reinforce that “oral cannabinoids can produce small improvements in pain and provide a dosing structure that minimizes risk to the patient, [but they] should be able to help educate physicians who [are looking] for a sense of what the literature tells us at this time,” she added.
“With chronic pain, we often find that different treatments will show small potential benefits and they have a certain risk profile,” Dr. Busse said.
“It’s almost impossible to know what patients think about this option unless you present them with the evidence and ask them to make a decision based on their values and preferences,” he said.
The Michael G. DeGroote Centre for Medicinal Cannabis Research funded the MAGIC Evidence Ecosystem Foundation to support the creation of the guideline. The center receives no funding from industry Dr. Busse, Dr. Cooper, and Dr. Wilkerson reported having no relevant financial relationships.
“Evidence alone is not sufficient for clinical decision-making, particularly in chronic pain,” said Jason Busse, DC, PhD, director of Michael G. DeGroote Centre for Medicinal Cannabis Research at McMaster University, Hamilton, Ont., and lead author of a newly released rapid guideline on medical cannabis or cannabinoids for chronic pain.
The recommendations, published online Sept. 9, 2021 in the British Medical Journal, suggest that providers offer patients with chronic pain a trial of noninhaled medical cannabis or cannabinoids if standard care or management is ineffective. However, the “weak” rating attached to the recommendation may compel some clinicians to automatically write off the panel’s recommendations.
“Because of the close balance between benefits and harms and wide variability in patient attitudes, the panel came to the conclusion that [some] patients presented with the current best evidence would likely choose to engage in a trial of medicinal cannabis, if their current care was felt to be suboptimal,” Dr. Busse explained in an interview.
But more importantly, “the recommendation allows for shared decision making to occur, and for different patients to make different decisions based on individual preferences and circumstances,” he said.
Evidence supports improved pain and sleep quality, physical functioning
Evidence supporting the use of medical cannabis in chronic pain is derived from a rigorous systematic review and meta-analysis of 32 studies enrolling 5,174 patients randomized to oral (capsule, spray, sublingual drops) or topical (transdermal cream) medical cannabis or placebo. Of note, three types of cannabinoids were represented: phytocannabinoids, synthetic, and endocannabinoids.
The studies included both patients with chronic noncancer pain (28 studies, n = 3,812) and chronic cancer pain not receiving palliative care (4 studies, n = 1,362). On average, baseline pain scores were a median 6.28 cm on a 10-cm visual analog scale (VAS), and median participant age was 53 years. 60% of trials reporting sex differences enrolled female participants. Overall, patients were followed for roughly 2 months (median, 50 days).
Findings (27 studies, n = 3,939) showed that, compared with placebo, medical cannabis resulted in a small, albeit important, improvement in the proportion of patients experiencing pain relief at or above the minimally important difference (MID) (moderate-certainty evidence, 10% modeled risk difference [RD; 95% confidence interval, 5%-15%] for achieving at least the MID of 1 cm).
Medical cannabis (15 studies, n = 2,425) also provided a small increase in the proportion of patients experiencing improvements in physical functioning at or above the MID (high certainty evidence, 4% modeled RD [95% CI, 0.1%-8%] for achieving at least a MID of 10 points).
Additionally, participants experienced significant improvements in sleep quality, compared with placebo (16 studies, 3,124 participants, high-quality evidence), demonstrating a weighted mean difference of –0.53 cm on a 10-cm VAS (95% CI, –0.75 to –0.30 cm). A total of nine larger trials (n = 2,652, high-certainty evidence) saw a small increase in the proportion of patients experiencing improved sleep quality at or above the MID: 6% modeled RD (95% CI, 2%-9%).
On the other hand, benefits did not extend to emotional, role, or social functioning (high-certainty evidence).
First do no harm: Start low, go slow
While these findings provide a rationale for medical cannabis in chronic pain, exploring options with patients can be challenging. Studies on medical cannabis consistently note that patients want information, but data also show that many providers express a lack of knowledge to provide adequate counseling.
There are also legal hurdles. Despite the authorization of medicinal cannabis across a majority of states and territories, cannabis is still a schedule I substance under the Federal Controlled Substances Act. In addition, the absence of standards around formulations, potency, and dosing has also been cited as a major barrier to recommending medical cannabis, as have concerns about adverse events (AEs), especially with inhaled and tetrahydrocannabinol (THC)-predominant formulations.
Like most medications, medical cannabis dosing should be individualized depending on product, patient, and ability to titrate the dose, but the guidelines provide a general rule of thumb. Providers considering therapeutic noninhaled medical cannabis trials are encouraged to start with a low-dose cannabidiol (CBD) oral tablet, spray, or sublingual oil drops 5 mg twice daily, increasing it by 10 mg every 2-3 days depending on the clinical response (to a maximum daily dose of 40 mg/day). If patient response is unsatisfactory, they should consider adding 1-2.5 mg THC/daily, titrated every 2-7 days to a maximum of 40 mg/day.
Still, an important caveat is whether or not adjunctive CBD alone is effective for chronic pain.
“While we know that one out of seven U.S. adults are using cannabidiol, we know very little about its therapeutic effects when given by itself for pain,” Ziva Cooper, PhD, director of the Cannabis Research Initiative at the University of California, Los Angeles, and an associate professor at-large of psychology and behavioral science, said in an interview. (Dr. Cooper was not involved in the guideline development.)
“But patients tend to self-report that CBD is helpful, and at low doses, we know that it is unlikely to have adverse effects of any significant concern,” Dr. Cooper noted.
Depending on its components, medical cannabis is associated with a wide range of AEs. Studies comprising the evidence base for the guideline reported transient cognitive impairment (relative risk, 2.39; 95% CI, 1.06-5.38), vomiting (RR, 1.46; 95% CI, 1.07-1.99), and drowsiness (RR, 2.14; 95% CI, 1.55-2.95), attention impairment (RR, 4.04; 95% CI, 1.67-9.74), and nausea (RR, 1.59; 95% CI, 1.28-1.99). Of note, findings of a subgroup analysis showed that the risk of dizziness increased with treatment duration, starting at 3 months (test of interaction P = .002).
However, Dr. Cooper explained that, because the included studies were inconsistent in terms of cannabis type (e.g., some looked at synthetic THC or THC-like substances where others looked at a THC/CBD combination) and formulation (capsules, oral mucosal sprays), it’s difficult to tease out component-specific AEs.
“These are really important things to note, especially when you think about different populations that might be using these types of medicines moving forward,” she said.
Toward that end, the guideline specifically states that there is “no reason why the expected benefits would be systematically different among adolescents and emerging adults.”
Among children with cancer, prior study findings reinforce the conclusion that benefits are similar to adults, but studies in this area are limited to end-of-life treatment, childhood cancer with primarily palliative intent, or progressive or relapsed cancer. Because THC’s safety profile is less certain in children, it’s also important to consider adverse neurocognitive effects before initiating a medical cannabis trial in this population.
Navigating the landscape
Although promising, the medical cannabis landscape is undoubtedly difficult to navigate, with land mines ranging from a limited inability to simply pick up a prescribing pad to quality control.
With the exception of three Food and Drug Administration–approved products – dronabinol, cannabidiol Rx, and nabilone – U.S. providers are only able to ‘certify,’ not prescribe, medical cannabis for chronic pain, and only if it is included within the state cannabis board’s list of eligible conditions. (A state-by-state guide is available.)
Quality control also varies by product but is critical. “You want to look for certificates of quality assurance,” Jenny Wilkerson, PhD, a research assistant professor of pharmacodynamics at the University of Florida, Gainesville, said in an interview. (Dr. Wilkerson was not involved in the guideline development.)
“A good dispensary should have that information or at least be willing to get that information, but generally speaking, that is something that patients need to ask for,” she emphasized, noting that “most available mass readouts are not divided by lots.”
Initial counseling and AE monitoring and regular follow-up is important, especially among patients who’ve never tried medical cannabis (or older patients whose prior experience may be limited to weaker recreational marijuana).
Notably, the reliance on medical dispensaries to deliver the right information at the right time may prove to be faulty. While recent data show that frontline dispensary workers regularly provide information to customers on their medical conditions and available products, they rarely, if ever, base recommendations on provider input, and never or rarely discuss potential AEs and other risks.
Per the new guideline, inexperienced patients should be seen monthly until a stable dose is achieved; longer times between visits can be considered in those who are more experienced. Still, patients should be advised to contact their provider when pain relief or other goals are insufficient, or when response or problematic AEs occur. This facilitates down-titration to a previously tolerated dose, up-titration in CBD and/or THC, or a different route of administration/formulation altogether.
Dr. Wilkerson pointed out that follow-up visits also provide an opportunity to do a blood draw and ask the lab to conduct pharmacokinetic analysis.
If possible, “ask patients to [ensure that they] take a standard dose before the visit so that the lab can assess the blood percentage of primary compounds and metabolites in the product that they are using,” she explained, noting that the information is helping to determine how “the different ratios may be affecting therapeutic response in individual patients.”
Granted, the guideline is only a start. But it is a good one.
“A lot of physicians want to be able to hang their hat on evidence of the safety and efficacy of these products, and the analysis that was leveraged for this guideline was very rigorous,” Dr. Cooper said.
Not only do they reinforce that “oral cannabinoids can produce small improvements in pain and provide a dosing structure that minimizes risk to the patient, [but they] should be able to help educate physicians who [are looking] for a sense of what the literature tells us at this time,” she added.
“With chronic pain, we often find that different treatments will show small potential benefits and they have a certain risk profile,” Dr. Busse said.
“It’s almost impossible to know what patients think about this option unless you present them with the evidence and ask them to make a decision based on their values and preferences,” he said.
The Michael G. DeGroote Centre for Medicinal Cannabis Research funded the MAGIC Evidence Ecosystem Foundation to support the creation of the guideline. The center receives no funding from industry Dr. Busse, Dr. Cooper, and Dr. Wilkerson reported having no relevant financial relationships.
“Evidence alone is not sufficient for clinical decision-making, particularly in chronic pain,” said Jason Busse, DC, PhD, director of Michael G. DeGroote Centre for Medicinal Cannabis Research at McMaster University, Hamilton, Ont., and lead author of a newly released rapid guideline on medical cannabis or cannabinoids for chronic pain.
The recommendations, published online Sept. 9, 2021 in the British Medical Journal, suggest that providers offer patients with chronic pain a trial of noninhaled medical cannabis or cannabinoids if standard care or management is ineffective. However, the “weak” rating attached to the recommendation may compel some clinicians to automatically write off the panel’s recommendations.
“Because of the close balance between benefits and harms and wide variability in patient attitudes, the panel came to the conclusion that [some] patients presented with the current best evidence would likely choose to engage in a trial of medicinal cannabis, if their current care was felt to be suboptimal,” Dr. Busse explained in an interview.
But more importantly, “the recommendation allows for shared decision making to occur, and for different patients to make different decisions based on individual preferences and circumstances,” he said.
Evidence supports improved pain and sleep quality, physical functioning
Evidence supporting the use of medical cannabis in chronic pain is derived from a rigorous systematic review and meta-analysis of 32 studies enrolling 5,174 patients randomized to oral (capsule, spray, sublingual drops) or topical (transdermal cream) medical cannabis or placebo. Of note, three types of cannabinoids were represented: phytocannabinoids, synthetic, and endocannabinoids.
The studies included both patients with chronic noncancer pain (28 studies, n = 3,812) and chronic cancer pain not receiving palliative care (4 studies, n = 1,362). On average, baseline pain scores were a median 6.28 cm on a 10-cm visual analog scale (VAS), and median participant age was 53 years. 60% of trials reporting sex differences enrolled female participants. Overall, patients were followed for roughly 2 months (median, 50 days).
Findings (27 studies, n = 3,939) showed that, compared with placebo, medical cannabis resulted in a small, albeit important, improvement in the proportion of patients experiencing pain relief at or above the minimally important difference (MID) (moderate-certainty evidence, 10% modeled risk difference [RD; 95% confidence interval, 5%-15%] for achieving at least the MID of 1 cm).
Medical cannabis (15 studies, n = 2,425) also provided a small increase in the proportion of patients experiencing improvements in physical functioning at or above the MID (high certainty evidence, 4% modeled RD [95% CI, 0.1%-8%] for achieving at least a MID of 10 points).
Additionally, participants experienced significant improvements in sleep quality, compared with placebo (16 studies, 3,124 participants, high-quality evidence), demonstrating a weighted mean difference of –0.53 cm on a 10-cm VAS (95% CI, –0.75 to –0.30 cm). A total of nine larger trials (n = 2,652, high-certainty evidence) saw a small increase in the proportion of patients experiencing improved sleep quality at or above the MID: 6% modeled RD (95% CI, 2%-9%).
On the other hand, benefits did not extend to emotional, role, or social functioning (high-certainty evidence).
First do no harm: Start low, go slow
While these findings provide a rationale for medical cannabis in chronic pain, exploring options with patients can be challenging. Studies on medical cannabis consistently note that patients want information, but data also show that many providers express a lack of knowledge to provide adequate counseling.
There are also legal hurdles. Despite the authorization of medicinal cannabis across a majority of states and territories, cannabis is still a schedule I substance under the Federal Controlled Substances Act. In addition, the absence of standards around formulations, potency, and dosing has also been cited as a major barrier to recommending medical cannabis, as have concerns about adverse events (AEs), especially with inhaled and tetrahydrocannabinol (THC)-predominant formulations.
Like most medications, medical cannabis dosing should be individualized depending on product, patient, and ability to titrate the dose, but the guidelines provide a general rule of thumb. Providers considering therapeutic noninhaled medical cannabis trials are encouraged to start with a low-dose cannabidiol (CBD) oral tablet, spray, or sublingual oil drops 5 mg twice daily, increasing it by 10 mg every 2-3 days depending on the clinical response (to a maximum daily dose of 40 mg/day). If patient response is unsatisfactory, they should consider adding 1-2.5 mg THC/daily, titrated every 2-7 days to a maximum of 40 mg/day.
Still, an important caveat is whether or not adjunctive CBD alone is effective for chronic pain.
“While we know that one out of seven U.S. adults are using cannabidiol, we know very little about its therapeutic effects when given by itself for pain,” Ziva Cooper, PhD, director of the Cannabis Research Initiative at the University of California, Los Angeles, and an associate professor at-large of psychology and behavioral science, said in an interview. (Dr. Cooper was not involved in the guideline development.)
“But patients tend to self-report that CBD is helpful, and at low doses, we know that it is unlikely to have adverse effects of any significant concern,” Dr. Cooper noted.
Depending on its components, medical cannabis is associated with a wide range of AEs. Studies comprising the evidence base for the guideline reported transient cognitive impairment (relative risk, 2.39; 95% CI, 1.06-5.38), vomiting (RR, 1.46; 95% CI, 1.07-1.99), and drowsiness (RR, 2.14; 95% CI, 1.55-2.95), attention impairment (RR, 4.04; 95% CI, 1.67-9.74), and nausea (RR, 1.59; 95% CI, 1.28-1.99). Of note, findings of a subgroup analysis showed that the risk of dizziness increased with treatment duration, starting at 3 months (test of interaction P = .002).
However, Dr. Cooper explained that, because the included studies were inconsistent in terms of cannabis type (e.g., some looked at synthetic THC or THC-like substances where others looked at a THC/CBD combination) and formulation (capsules, oral mucosal sprays), it’s difficult to tease out component-specific AEs.
“These are really important things to note, especially when you think about different populations that might be using these types of medicines moving forward,” she said.
Toward that end, the guideline specifically states that there is “no reason why the expected benefits would be systematically different among adolescents and emerging adults.”
Among children with cancer, prior study findings reinforce the conclusion that benefits are similar to adults, but studies in this area are limited to end-of-life treatment, childhood cancer with primarily palliative intent, or progressive or relapsed cancer. Because THC’s safety profile is less certain in children, it’s also important to consider adverse neurocognitive effects before initiating a medical cannabis trial in this population.
Navigating the landscape
Although promising, the medical cannabis landscape is undoubtedly difficult to navigate, with land mines ranging from a limited inability to simply pick up a prescribing pad to quality control.
With the exception of three Food and Drug Administration–approved products – dronabinol, cannabidiol Rx, and nabilone – U.S. providers are only able to ‘certify,’ not prescribe, medical cannabis for chronic pain, and only if it is included within the state cannabis board’s list of eligible conditions. (A state-by-state guide is available.)
Quality control also varies by product but is critical. “You want to look for certificates of quality assurance,” Jenny Wilkerson, PhD, a research assistant professor of pharmacodynamics at the University of Florida, Gainesville, said in an interview. (Dr. Wilkerson was not involved in the guideline development.)
“A good dispensary should have that information or at least be willing to get that information, but generally speaking, that is something that patients need to ask for,” she emphasized, noting that “most available mass readouts are not divided by lots.”
Initial counseling and AE monitoring and regular follow-up is important, especially among patients who’ve never tried medical cannabis (or older patients whose prior experience may be limited to weaker recreational marijuana).
Notably, the reliance on medical dispensaries to deliver the right information at the right time may prove to be faulty. While recent data show that frontline dispensary workers regularly provide information to customers on their medical conditions and available products, they rarely, if ever, base recommendations on provider input, and never or rarely discuss potential AEs and other risks.
Per the new guideline, inexperienced patients should be seen monthly until a stable dose is achieved; longer times between visits can be considered in those who are more experienced. Still, patients should be advised to contact their provider when pain relief or other goals are insufficient, or when response or problematic AEs occur. This facilitates down-titration to a previously tolerated dose, up-titration in CBD and/or THC, or a different route of administration/formulation altogether.
Dr. Wilkerson pointed out that follow-up visits also provide an opportunity to do a blood draw and ask the lab to conduct pharmacokinetic analysis.
If possible, “ask patients to [ensure that they] take a standard dose before the visit so that the lab can assess the blood percentage of primary compounds and metabolites in the product that they are using,” she explained, noting that the information is helping to determine how “the different ratios may be affecting therapeutic response in individual patients.”
Granted, the guideline is only a start. But it is a good one.
“A lot of physicians want to be able to hang their hat on evidence of the safety and efficacy of these products, and the analysis that was leveraged for this guideline was very rigorous,” Dr. Cooper said.
Not only do they reinforce that “oral cannabinoids can produce small improvements in pain and provide a dosing structure that minimizes risk to the patient, [but they] should be able to help educate physicians who [are looking] for a sense of what the literature tells us at this time,” she added.
“With chronic pain, we often find that different treatments will show small potential benefits and they have a certain risk profile,” Dr. Busse said.
“It’s almost impossible to know what patients think about this option unless you present them with the evidence and ask them to make a decision based on their values and preferences,” he said.
The Michael G. DeGroote Centre for Medicinal Cannabis Research funded the MAGIC Evidence Ecosystem Foundation to support the creation of the guideline. The center receives no funding from industry Dr. Busse, Dr. Cooper, and Dr. Wilkerson reported having no relevant financial relationships.
FROM THE BMJ
Nonopioid med promising for neuropathic pain
Top-line results from a phase 2 study suggest vixotrigine (BIIB074, Biogen), a nonopioid investigational oral pain medication, reduces chronic neuropathic pain caused by small fiber neuropathy (SFN) and is generally well tolerated.
“We are encouraged by the overall results of the CONVEY study, especially given the significant unmet medical need for additional agents to treat chronic painful neuropathy,” Katherine Dawson, MD, senior vice president and head of the therapeutics development unit at Biogen, said in a news release.
Vixotrigine (BIIB074) is a peripherally and centrally acting, orally administered, voltage- and use-dependent voltage-gated sodium channel blocker.
CONVEY was a phase 2, placebo-controlled, double-blind, randomized withdrawal study of 265 patients experiencing pain from confirmed idiopathic or diabetes-associated SFN.
Following a 4-week open-label run-in period, 123 responders to vixotrigine were randomly allocated to 200 mg or 350 mg vixotrigine or placebo twice daily for 12 weeks in the double-blind portion of the study.
At week 12, vixotrigine 200 mg twice daily met the primary endpoint of a statistically significant reduction from baseline in the mean average daily pain (ADP) score versus placebo (P = .0501).
A subgroup analysis showed a treatment effect in patients with diabetes-associated SFN but not in the smaller subgroup of patients with idiopathic SFN.
The 200-mg dose also led to a significant improvement over placebo in mean worst daily pain score at 12 weeks (P = .0455).
A numeric advantage of 200 mg vixotrigine over placebo was observed in additional secondary endpoints, including the proportion of patients with at least a 2-point improvement in ADP score and the proportion with at least a 30% reduction in ADP at week 12, but these failed to reach statistical significance.
Vixotrigine 350 mg twice daily did not meet the primary endpoint of mean change in ADP at 12 weeks.
However, treatment at the higher dose led to a significant increase in the proportion of patients who reported being “very much improved” or “much improved” over baseline (P = .0580), Biogen reported.
In addition, a numeric advantage of 350 mg over placebo was observed in the proportion of patients with a 2-point or greater improvement in ADP score and the proportion with at least a 30% reduction in ADP at 12 weeks, but these also did not reach statistical significance.
Both doses of vixotrigine were “generally well tolerated and the safety profile was consistent with previous studies of vixotrigine with no evidence of abuse potential,” the company said.
In the open-label period, common adverse events seen in at least 2.5% of patients were dizziness, headache, vertigo, and nausea; adverse events led 5.3% of patients to discontinue the open-label portion of the study. Across the entire study, most adverse events were mild or moderate in severity.
“The totality of data from the vixotrigine program will inform potential doses for study in future phase 3 clinical trials,” the company said.
A version of this article first appeared on Medscape.com.
Top-line results from a phase 2 study suggest vixotrigine (BIIB074, Biogen), a nonopioid investigational oral pain medication, reduces chronic neuropathic pain caused by small fiber neuropathy (SFN) and is generally well tolerated.
“We are encouraged by the overall results of the CONVEY study, especially given the significant unmet medical need for additional agents to treat chronic painful neuropathy,” Katherine Dawson, MD, senior vice president and head of the therapeutics development unit at Biogen, said in a news release.
Vixotrigine (BIIB074) is a peripherally and centrally acting, orally administered, voltage- and use-dependent voltage-gated sodium channel blocker.
CONVEY was a phase 2, placebo-controlled, double-blind, randomized withdrawal study of 265 patients experiencing pain from confirmed idiopathic or diabetes-associated SFN.
Following a 4-week open-label run-in period, 123 responders to vixotrigine were randomly allocated to 200 mg or 350 mg vixotrigine or placebo twice daily for 12 weeks in the double-blind portion of the study.
At week 12, vixotrigine 200 mg twice daily met the primary endpoint of a statistically significant reduction from baseline in the mean average daily pain (ADP) score versus placebo (P = .0501).
A subgroup analysis showed a treatment effect in patients with diabetes-associated SFN but not in the smaller subgroup of patients with idiopathic SFN.
The 200-mg dose also led to a significant improvement over placebo in mean worst daily pain score at 12 weeks (P = .0455).
A numeric advantage of 200 mg vixotrigine over placebo was observed in additional secondary endpoints, including the proportion of patients with at least a 2-point improvement in ADP score and the proportion with at least a 30% reduction in ADP at week 12, but these failed to reach statistical significance.
Vixotrigine 350 mg twice daily did not meet the primary endpoint of mean change in ADP at 12 weeks.
However, treatment at the higher dose led to a significant increase in the proportion of patients who reported being “very much improved” or “much improved” over baseline (P = .0580), Biogen reported.
In addition, a numeric advantage of 350 mg over placebo was observed in the proportion of patients with a 2-point or greater improvement in ADP score and the proportion with at least a 30% reduction in ADP at 12 weeks, but these also did not reach statistical significance.
Both doses of vixotrigine were “generally well tolerated and the safety profile was consistent with previous studies of vixotrigine with no evidence of abuse potential,” the company said.
In the open-label period, common adverse events seen in at least 2.5% of patients were dizziness, headache, vertigo, and nausea; adverse events led 5.3% of patients to discontinue the open-label portion of the study. Across the entire study, most adverse events were mild or moderate in severity.
“The totality of data from the vixotrigine program will inform potential doses for study in future phase 3 clinical trials,” the company said.
A version of this article first appeared on Medscape.com.
Top-line results from a phase 2 study suggest vixotrigine (BIIB074, Biogen), a nonopioid investigational oral pain medication, reduces chronic neuropathic pain caused by small fiber neuropathy (SFN) and is generally well tolerated.
“We are encouraged by the overall results of the CONVEY study, especially given the significant unmet medical need for additional agents to treat chronic painful neuropathy,” Katherine Dawson, MD, senior vice president and head of the therapeutics development unit at Biogen, said in a news release.
Vixotrigine (BIIB074) is a peripherally and centrally acting, orally administered, voltage- and use-dependent voltage-gated sodium channel blocker.
CONVEY was a phase 2, placebo-controlled, double-blind, randomized withdrawal study of 265 patients experiencing pain from confirmed idiopathic or diabetes-associated SFN.
Following a 4-week open-label run-in period, 123 responders to vixotrigine were randomly allocated to 200 mg or 350 mg vixotrigine or placebo twice daily for 12 weeks in the double-blind portion of the study.
At week 12, vixotrigine 200 mg twice daily met the primary endpoint of a statistically significant reduction from baseline in the mean average daily pain (ADP) score versus placebo (P = .0501).
A subgroup analysis showed a treatment effect in patients with diabetes-associated SFN but not in the smaller subgroup of patients with idiopathic SFN.
The 200-mg dose also led to a significant improvement over placebo in mean worst daily pain score at 12 weeks (P = .0455).
A numeric advantage of 200 mg vixotrigine over placebo was observed in additional secondary endpoints, including the proportion of patients with at least a 2-point improvement in ADP score and the proportion with at least a 30% reduction in ADP at week 12, but these failed to reach statistical significance.
Vixotrigine 350 mg twice daily did not meet the primary endpoint of mean change in ADP at 12 weeks.
However, treatment at the higher dose led to a significant increase in the proportion of patients who reported being “very much improved” or “much improved” over baseline (P = .0580), Biogen reported.
In addition, a numeric advantage of 350 mg over placebo was observed in the proportion of patients with a 2-point or greater improvement in ADP score and the proportion with at least a 30% reduction in ADP at 12 weeks, but these also did not reach statistical significance.
Both doses of vixotrigine were “generally well tolerated and the safety profile was consistent with previous studies of vixotrigine with no evidence of abuse potential,” the company said.
In the open-label period, common adverse events seen in at least 2.5% of patients were dizziness, headache, vertigo, and nausea; adverse events led 5.3% of patients to discontinue the open-label portion of the study. Across the entire study, most adverse events were mild or moderate in severity.
“The totality of data from the vixotrigine program will inform potential doses for study in future phase 3 clinical trials,” the company said.
A version of this article first appeared on Medscape.com.
Step-wise medical therapy is cost effective for endometriosis
For patients with endometriosis-related dysmenorrhea, it is cost effective to use medical therapy before surgery, according to investigators.
A stepwise strategy involving two medications, then surgery, was associated with the lowest cost per quality-adjusted life-years (QALYs), reported lead author, Jacqueline A. Bohn, MD, of Oregon Health & Science University, Portland, and colleagues.
“In 2009, the medical costs associated with endometriosis in the United States were estimated at $69.4 billion annually,” the investigators wrote in Obstetrics and Gynecology. “Despite the recognized cost burden of this disease, cost-effectiveness data on the various treatment strategies is limited. Previous studies have investigated the direct and indirect costs regarding endometriosis; however, there are no prior studies that evaluate the cost-effectiveness of a stepwise regimen to guide management.”
To fill this knowledge gap, Dr. Bohn and colleagues created a cost-effectiveness model comparing four treatment strategies:
NSAIDs, then surgery
NSAIDs, then short-acting reversible contraceptives or long-acting reversible contraceptives (LARCs), then surgery
NSAIDs, then a short-acting reversible contraceptive or a LARC, then a LARC or gonadotropin-releasing hormone (GnRH) modulator, then surgery
Surgery alone
The analysis, which compared costs, QALYs, and incremental cost-effectiveness ratios, involved a theoretical cohort of 4,817,894 women aged 18-45 years, representing the estimated number of reproductive-age women in the United States with endometriosis-related dysmenorrhea. Costs were determined from published literature and inflated to 2019 dollars. Medical treatments were theoretically given for 6 months each, and the cost of laparoscopic surgery incorporated 12 months of postoperative care.
Of the four strategies, the two-medication approach was most cost effective, with a cost per QALY of $1,158. This was followed closely by the three-medication regimen, at $1,158, the single-medication regimen, at $2,108, and finally, surgery alone, at $4,338.
“We found that, although cost effective, requiring trial of a third medication offered little comparative advantage before proceeding directly to surgery after the second therapy fails,” the investigators wrote. “Yet, for the woman who is anxious about surgical intervention, or when a prolonged wait for a surgical specialist occurs, trial of a GnRH modulator may be worthwhile.”
Compared with surgery alone, each regimen starting with medical therapy remained below the standard willingness-to-pay threshold of $100,000 per QALY; however, the investigators recommend against trying more than three medications.
“Delaying surgical management in a woman with pain refractory to more than three medications may decrease quality of life and further increase cost,” they wrote.
To make surgery alone the most cost-effective option, surgery success would need to exceed 83%, Dr. Bohn and colleagues concluded.
According to Hugh Taylor, MD, of Yale University, New Haven, Conn., it’s unlikely that this surgery success threshold will be met, since surgery alone typically leads to recurrence.
“We know there’s a very high relapse rate after surgery,” Dr. Taylor said in an interview. “Even if the surgery may be initially successful, there’s roughly a 50% recurrence rate after about 2 years. So, finding the right medical therapy will give you more chance for long-term success.”
Dr. Taylor said it’s “really nice” that Dr. Bohn and colleagues conducted a sequential analysis because the findings support the most common approach in real-world practice.
“It confirms that starting with a medical therapy prior to surgery is an appropriate, successful treatment for endometriosis, which is something that many, many people in the community do, but we haven’t had a real trial to show that,” he said.
Dr. Taylor offered two areas of improvement for similar studies in the future: First, he suggested separating LARCs from oral contraceptives because LARCs may be less effective for some patients with endometriosis; and second, he suggested that limiting the third medication to a GnRH antagonist would be more applicable to real-world practice than using the broader category of GnRH modulators.
Although the three-medication approach involving a GnRH modulator was slightly more expensive than the two-medication approach, Dr. Taylor said the costs were so similar that a three-medication approach is “still reasonable,” particularly because it could spare patients from surgery.
Dr. Taylor also speculated that trying a GnRH antagonist could become more cost effective soon. Although only one GnRH antagonist is currently on the market, he noted that a second agent is poised for Food and Drug Administration approval, while a third is in the pipeline, and this competition may decrease drug prices.
The investigators disclosed support from the National Institutes of Health, Arnold Ventures, the World Health Organization, Merck, and others. Dr. Taylor reported that Yale University receives funding for endometriosis biomarker research from AbbVie.
For patients with endometriosis-related dysmenorrhea, it is cost effective to use medical therapy before surgery, according to investigators.
A stepwise strategy involving two medications, then surgery, was associated with the lowest cost per quality-adjusted life-years (QALYs), reported lead author, Jacqueline A. Bohn, MD, of Oregon Health & Science University, Portland, and colleagues.
“In 2009, the medical costs associated with endometriosis in the United States were estimated at $69.4 billion annually,” the investigators wrote in Obstetrics and Gynecology. “Despite the recognized cost burden of this disease, cost-effectiveness data on the various treatment strategies is limited. Previous studies have investigated the direct and indirect costs regarding endometriosis; however, there are no prior studies that evaluate the cost-effectiveness of a stepwise regimen to guide management.”
To fill this knowledge gap, Dr. Bohn and colleagues created a cost-effectiveness model comparing four treatment strategies:
NSAIDs, then surgery
NSAIDs, then short-acting reversible contraceptives or long-acting reversible contraceptives (LARCs), then surgery
NSAIDs, then a short-acting reversible contraceptive or a LARC, then a LARC or gonadotropin-releasing hormone (GnRH) modulator, then surgery
Surgery alone
The analysis, which compared costs, QALYs, and incremental cost-effectiveness ratios, involved a theoretical cohort of 4,817,894 women aged 18-45 years, representing the estimated number of reproductive-age women in the United States with endometriosis-related dysmenorrhea. Costs were determined from published literature and inflated to 2019 dollars. Medical treatments were theoretically given for 6 months each, and the cost of laparoscopic surgery incorporated 12 months of postoperative care.
Of the four strategies, the two-medication approach was most cost effective, with a cost per QALY of $1,158. This was followed closely by the three-medication regimen, at $1,158, the single-medication regimen, at $2,108, and finally, surgery alone, at $4,338.
“We found that, although cost effective, requiring trial of a third medication offered little comparative advantage before proceeding directly to surgery after the second therapy fails,” the investigators wrote. “Yet, for the woman who is anxious about surgical intervention, or when a prolonged wait for a surgical specialist occurs, trial of a GnRH modulator may be worthwhile.”
Compared with surgery alone, each regimen starting with medical therapy remained below the standard willingness-to-pay threshold of $100,000 per QALY; however, the investigators recommend against trying more than three medications.
“Delaying surgical management in a woman with pain refractory to more than three medications may decrease quality of life and further increase cost,” they wrote.
To make surgery alone the most cost-effective option, surgery success would need to exceed 83%, Dr. Bohn and colleagues concluded.
According to Hugh Taylor, MD, of Yale University, New Haven, Conn., it’s unlikely that this surgery success threshold will be met, since surgery alone typically leads to recurrence.
“We know there’s a very high relapse rate after surgery,” Dr. Taylor said in an interview. “Even if the surgery may be initially successful, there’s roughly a 50% recurrence rate after about 2 years. So, finding the right medical therapy will give you more chance for long-term success.”
Dr. Taylor said it’s “really nice” that Dr. Bohn and colleagues conducted a sequential analysis because the findings support the most common approach in real-world practice.
“It confirms that starting with a medical therapy prior to surgery is an appropriate, successful treatment for endometriosis, which is something that many, many people in the community do, but we haven’t had a real trial to show that,” he said.
Dr. Taylor offered two areas of improvement for similar studies in the future: First, he suggested separating LARCs from oral contraceptives because LARCs may be less effective for some patients with endometriosis; and second, he suggested that limiting the third medication to a GnRH antagonist would be more applicable to real-world practice than using the broader category of GnRH modulators.
Although the three-medication approach involving a GnRH modulator was slightly more expensive than the two-medication approach, Dr. Taylor said the costs were so similar that a three-medication approach is “still reasonable,” particularly because it could spare patients from surgery.
Dr. Taylor also speculated that trying a GnRH antagonist could become more cost effective soon. Although only one GnRH antagonist is currently on the market, he noted that a second agent is poised for Food and Drug Administration approval, while a third is in the pipeline, and this competition may decrease drug prices.
The investigators disclosed support from the National Institutes of Health, Arnold Ventures, the World Health Organization, Merck, and others. Dr. Taylor reported that Yale University receives funding for endometriosis biomarker research from AbbVie.
For patients with endometriosis-related dysmenorrhea, it is cost effective to use medical therapy before surgery, according to investigators.
A stepwise strategy involving two medications, then surgery, was associated with the lowest cost per quality-adjusted life-years (QALYs), reported lead author, Jacqueline A. Bohn, MD, of Oregon Health & Science University, Portland, and colleagues.
“In 2009, the medical costs associated with endometriosis in the United States were estimated at $69.4 billion annually,” the investigators wrote in Obstetrics and Gynecology. “Despite the recognized cost burden of this disease, cost-effectiveness data on the various treatment strategies is limited. Previous studies have investigated the direct and indirect costs regarding endometriosis; however, there are no prior studies that evaluate the cost-effectiveness of a stepwise regimen to guide management.”
To fill this knowledge gap, Dr. Bohn and colleagues created a cost-effectiveness model comparing four treatment strategies:
NSAIDs, then surgery
NSAIDs, then short-acting reversible contraceptives or long-acting reversible contraceptives (LARCs), then surgery
NSAIDs, then a short-acting reversible contraceptive or a LARC, then a LARC or gonadotropin-releasing hormone (GnRH) modulator, then surgery
Surgery alone
The analysis, which compared costs, QALYs, and incremental cost-effectiveness ratios, involved a theoretical cohort of 4,817,894 women aged 18-45 years, representing the estimated number of reproductive-age women in the United States with endometriosis-related dysmenorrhea. Costs were determined from published literature and inflated to 2019 dollars. Medical treatments were theoretically given for 6 months each, and the cost of laparoscopic surgery incorporated 12 months of postoperative care.
Of the four strategies, the two-medication approach was most cost effective, with a cost per QALY of $1,158. This was followed closely by the three-medication regimen, at $1,158, the single-medication regimen, at $2,108, and finally, surgery alone, at $4,338.
“We found that, although cost effective, requiring trial of a third medication offered little comparative advantage before proceeding directly to surgery after the second therapy fails,” the investigators wrote. “Yet, for the woman who is anxious about surgical intervention, or when a prolonged wait for a surgical specialist occurs, trial of a GnRH modulator may be worthwhile.”
Compared with surgery alone, each regimen starting with medical therapy remained below the standard willingness-to-pay threshold of $100,000 per QALY; however, the investigators recommend against trying more than three medications.
“Delaying surgical management in a woman with pain refractory to more than three medications may decrease quality of life and further increase cost,” they wrote.
To make surgery alone the most cost-effective option, surgery success would need to exceed 83%, Dr. Bohn and colleagues concluded.
According to Hugh Taylor, MD, of Yale University, New Haven, Conn., it’s unlikely that this surgery success threshold will be met, since surgery alone typically leads to recurrence.
“We know there’s a very high relapse rate after surgery,” Dr. Taylor said in an interview. “Even if the surgery may be initially successful, there’s roughly a 50% recurrence rate after about 2 years. So, finding the right medical therapy will give you more chance for long-term success.”
Dr. Taylor said it’s “really nice” that Dr. Bohn and colleagues conducted a sequential analysis because the findings support the most common approach in real-world practice.
“It confirms that starting with a medical therapy prior to surgery is an appropriate, successful treatment for endometriosis, which is something that many, many people in the community do, but we haven’t had a real trial to show that,” he said.
Dr. Taylor offered two areas of improvement for similar studies in the future: First, he suggested separating LARCs from oral contraceptives because LARCs may be less effective for some patients with endometriosis; and second, he suggested that limiting the third medication to a GnRH antagonist would be more applicable to real-world practice than using the broader category of GnRH modulators.
Although the three-medication approach involving a GnRH modulator was slightly more expensive than the two-medication approach, Dr. Taylor said the costs were so similar that a three-medication approach is “still reasonable,” particularly because it could spare patients from surgery.
Dr. Taylor also speculated that trying a GnRH antagonist could become more cost effective soon. Although only one GnRH antagonist is currently on the market, he noted that a second agent is poised for Food and Drug Administration approval, while a third is in the pipeline, and this competition may decrease drug prices.
The investigators disclosed support from the National Institutes of Health, Arnold Ventures, the World Health Organization, Merck, and others. Dr. Taylor reported that Yale University receives funding for endometriosis biomarker research from AbbVie.
FROM OBSTETRICS & GYNECOLOGY
As opioid deaths climb, human trials begin for vaccine
Opioid-related drug overdose deaths in the United States exploded to an estimated record high of 69,031 people in 2020, topping the 49,860 deaths logged in 2019, according to a new report from the Centers for Disease Control and Prevention. Most of the deaths involved synthetic opioids such as fentanyl.
President Joe Biden has pledged more than $10 billion to expand access to prevention, treatment, and recovery services. The money is important as people receiving treatment for opioid use disorder have a high risk for relapse, and that means a high risk for opioid overdose.
Now, researchers are studying a possible bridge to successful recovery: A vaccine that could blunt the drugs’ ability to cause harm.
The first such vaccines are now entering clinical trials, raising hopes of adding another tool to the antiaddiction armamentarium. But even if the vaccines prove safe and effective, their success could generate some new problems to solve.
An advantage of vaccines is that their effects can last for several months, said trial investigator Sandra Comer, PhD, professor of neurobiology and psychiatry at Columbia University Irving Medical Center, New York. Dropout rates for existing medical therapies for opioid use disorder are as high as 50% at 6 months, and a vaccine could protect people from overdose and give them time to re-enter treatment.
“It serves as a bit of a safety net,” she said.
The first vaccine to enter a trial targets oxycodone. Volunteers are being recruited who have a diagnosis of opioid use disorder but are not being medically treated and are still using opioids. A third of them will receive a placebo vaccine, a third will receive a low-dose injection of vaccine, and the other third will receive a high-dose vaccine.
A shot against oxycodone
Researchers are primarily tracking the safety of the shot, but they’re also looking at whether vaccination prevents the euphoria that opioids usually produce. They expect to enroll 24 people initially but expand to 45 if results look promising.
In response to the shot, the body produces antibodies, proteins that tag oxycodone and keep it from reaching the brain. If the drug can’t reach brain cells, it can’t produce euphoria. And more important for lifesaving effects, it can’t block the brain’s signals to the body to breathe. The vaccine has already performed well in animal studies.
Previous trials of vaccines for cocaine and nicotine failed. Those vaccines made it to the last clinical trial stage, but didn’t prove effective overall. So this time, investigators plan to track antibody levels in participants, examining blood samples for signs of a good immune response to the vaccine.
But even though earlier cocaine and nicotine vaccines didn’t work for everybody, there were some people they seemed to help. This is why investigators involved in opioid vaccine trials want to track immune responses, said Marco Pravetoni, PhD, associate professor of pharmacology and medicine at the University of Minnesota, Minneapolis, whose team will be assessing the blood samples. Ultimately, a doctor might even be able to use this information to tailor vaccine selection to a specific person.
Dr. Pravetoni also said that oxycodone is one of three vaccine targets – the other two are heroin and fentanyl – that researchers hope to combine into a single shot. Recipients might need to have one shot a month for the first 3 to 4 months and then receive annual boosters.
Stopping the pain
The vaccines also raise some issues that need attention, said Cody Wenthur, PharmD, PhD, assistant professor of pharmacy at the University of Wisconsin–Madison, who is not involved in the vaccine trials.
“If you’re vaccinated against oxycodone, you might not have access to adequate pain control if you get into a car accident, for example,” he said.
Clinicians could use other opioids for pain management, but limiting the opioids that the vaccine targets is a “double-edged sword,” said Dr. Wenthur, because vaccinated people could just switch their opioid of choice to one that a vaccine does not inhibit.
Although these issues need to be addressed, vaccines, if successful, will have an important role. Dr. Wenthur noted a survey of pharmacists and pharmacy students that he and his group conducted showing that respondents “overwhelmingly” viewed a potential vaccine as helpful.
said Dr. Pravetoni. He mentioned the 2002 incident when terrorists took over a theater in Moscow and Russian special forces are thought to have used an aerosolized form of fentanyl to incapacitate everyone in the room. More than 100 of the hostages died, and the episode raised the specter of opioids being used in chemical attacks.
Dr. Pravetoni said vaccination could offer protection for first responders, law enforcement or other people whose professions place them at risk for inhalation, either accidentally or through such attacks.
These or other real-world applications for people at risk for exposure are several years away. Dr. Pravetoni said it took 10 years to get to this phase and estimates that, in about 5 years, a vaccine that targets multiple opioid drugs might enter the first clinical trial.
A version of this article first appeared on WebMD.com.
Opioid-related drug overdose deaths in the United States exploded to an estimated record high of 69,031 people in 2020, topping the 49,860 deaths logged in 2019, according to a new report from the Centers for Disease Control and Prevention. Most of the deaths involved synthetic opioids such as fentanyl.
President Joe Biden has pledged more than $10 billion to expand access to prevention, treatment, and recovery services. The money is important as people receiving treatment for opioid use disorder have a high risk for relapse, and that means a high risk for opioid overdose.
Now, researchers are studying a possible bridge to successful recovery: A vaccine that could blunt the drugs’ ability to cause harm.
The first such vaccines are now entering clinical trials, raising hopes of adding another tool to the antiaddiction armamentarium. But even if the vaccines prove safe and effective, their success could generate some new problems to solve.
An advantage of vaccines is that their effects can last for several months, said trial investigator Sandra Comer, PhD, professor of neurobiology and psychiatry at Columbia University Irving Medical Center, New York. Dropout rates for existing medical therapies for opioid use disorder are as high as 50% at 6 months, and a vaccine could protect people from overdose and give them time to re-enter treatment.
“It serves as a bit of a safety net,” she said.
The first vaccine to enter a trial targets oxycodone. Volunteers are being recruited who have a diagnosis of opioid use disorder but are not being medically treated and are still using opioids. A third of them will receive a placebo vaccine, a third will receive a low-dose injection of vaccine, and the other third will receive a high-dose vaccine.
A shot against oxycodone
Researchers are primarily tracking the safety of the shot, but they’re also looking at whether vaccination prevents the euphoria that opioids usually produce. They expect to enroll 24 people initially but expand to 45 if results look promising.
In response to the shot, the body produces antibodies, proteins that tag oxycodone and keep it from reaching the brain. If the drug can’t reach brain cells, it can’t produce euphoria. And more important for lifesaving effects, it can’t block the brain’s signals to the body to breathe. The vaccine has already performed well in animal studies.
Previous trials of vaccines for cocaine and nicotine failed. Those vaccines made it to the last clinical trial stage, but didn’t prove effective overall. So this time, investigators plan to track antibody levels in participants, examining blood samples for signs of a good immune response to the vaccine.
But even though earlier cocaine and nicotine vaccines didn’t work for everybody, there were some people they seemed to help. This is why investigators involved in opioid vaccine trials want to track immune responses, said Marco Pravetoni, PhD, associate professor of pharmacology and medicine at the University of Minnesota, Minneapolis, whose team will be assessing the blood samples. Ultimately, a doctor might even be able to use this information to tailor vaccine selection to a specific person.
Dr. Pravetoni also said that oxycodone is one of three vaccine targets – the other two are heroin and fentanyl – that researchers hope to combine into a single shot. Recipients might need to have one shot a month for the first 3 to 4 months and then receive annual boosters.
Stopping the pain
The vaccines also raise some issues that need attention, said Cody Wenthur, PharmD, PhD, assistant professor of pharmacy at the University of Wisconsin–Madison, who is not involved in the vaccine trials.
“If you’re vaccinated against oxycodone, you might not have access to adequate pain control if you get into a car accident, for example,” he said.
Clinicians could use other opioids for pain management, but limiting the opioids that the vaccine targets is a “double-edged sword,” said Dr. Wenthur, because vaccinated people could just switch their opioid of choice to one that a vaccine does not inhibit.
Although these issues need to be addressed, vaccines, if successful, will have an important role. Dr. Wenthur noted a survey of pharmacists and pharmacy students that he and his group conducted showing that respondents “overwhelmingly” viewed a potential vaccine as helpful.
said Dr. Pravetoni. He mentioned the 2002 incident when terrorists took over a theater in Moscow and Russian special forces are thought to have used an aerosolized form of fentanyl to incapacitate everyone in the room. More than 100 of the hostages died, and the episode raised the specter of opioids being used in chemical attacks.
Dr. Pravetoni said vaccination could offer protection for first responders, law enforcement or other people whose professions place them at risk for inhalation, either accidentally or through such attacks.
These or other real-world applications for people at risk for exposure are several years away. Dr. Pravetoni said it took 10 years to get to this phase and estimates that, in about 5 years, a vaccine that targets multiple opioid drugs might enter the first clinical trial.
A version of this article first appeared on WebMD.com.
Opioid-related drug overdose deaths in the United States exploded to an estimated record high of 69,031 people in 2020, topping the 49,860 deaths logged in 2019, according to a new report from the Centers for Disease Control and Prevention. Most of the deaths involved synthetic opioids such as fentanyl.
President Joe Biden has pledged more than $10 billion to expand access to prevention, treatment, and recovery services. The money is important as people receiving treatment for opioid use disorder have a high risk for relapse, and that means a high risk for opioid overdose.
Now, researchers are studying a possible bridge to successful recovery: A vaccine that could blunt the drugs’ ability to cause harm.
The first such vaccines are now entering clinical trials, raising hopes of adding another tool to the antiaddiction armamentarium. But even if the vaccines prove safe and effective, their success could generate some new problems to solve.
An advantage of vaccines is that their effects can last for several months, said trial investigator Sandra Comer, PhD, professor of neurobiology and psychiatry at Columbia University Irving Medical Center, New York. Dropout rates for existing medical therapies for opioid use disorder are as high as 50% at 6 months, and a vaccine could protect people from overdose and give them time to re-enter treatment.
“It serves as a bit of a safety net,” she said.
The first vaccine to enter a trial targets oxycodone. Volunteers are being recruited who have a diagnosis of opioid use disorder but are not being medically treated and are still using opioids. A third of them will receive a placebo vaccine, a third will receive a low-dose injection of vaccine, and the other third will receive a high-dose vaccine.
A shot against oxycodone
Researchers are primarily tracking the safety of the shot, but they’re also looking at whether vaccination prevents the euphoria that opioids usually produce. They expect to enroll 24 people initially but expand to 45 if results look promising.
In response to the shot, the body produces antibodies, proteins that tag oxycodone and keep it from reaching the brain. If the drug can’t reach brain cells, it can’t produce euphoria. And more important for lifesaving effects, it can’t block the brain’s signals to the body to breathe. The vaccine has already performed well in animal studies.
Previous trials of vaccines for cocaine and nicotine failed. Those vaccines made it to the last clinical trial stage, but didn’t prove effective overall. So this time, investigators plan to track antibody levels in participants, examining blood samples for signs of a good immune response to the vaccine.
But even though earlier cocaine and nicotine vaccines didn’t work for everybody, there were some people they seemed to help. This is why investigators involved in opioid vaccine trials want to track immune responses, said Marco Pravetoni, PhD, associate professor of pharmacology and medicine at the University of Minnesota, Minneapolis, whose team will be assessing the blood samples. Ultimately, a doctor might even be able to use this information to tailor vaccine selection to a specific person.
Dr. Pravetoni also said that oxycodone is one of three vaccine targets – the other two are heroin and fentanyl – that researchers hope to combine into a single shot. Recipients might need to have one shot a month for the first 3 to 4 months and then receive annual boosters.
Stopping the pain
The vaccines also raise some issues that need attention, said Cody Wenthur, PharmD, PhD, assistant professor of pharmacy at the University of Wisconsin–Madison, who is not involved in the vaccine trials.
“If you’re vaccinated against oxycodone, you might not have access to adequate pain control if you get into a car accident, for example,” he said.
Clinicians could use other opioids for pain management, but limiting the opioids that the vaccine targets is a “double-edged sword,” said Dr. Wenthur, because vaccinated people could just switch their opioid of choice to one that a vaccine does not inhibit.
Although these issues need to be addressed, vaccines, if successful, will have an important role. Dr. Wenthur noted a survey of pharmacists and pharmacy students that he and his group conducted showing that respondents “overwhelmingly” viewed a potential vaccine as helpful.
said Dr. Pravetoni. He mentioned the 2002 incident when terrorists took over a theater in Moscow and Russian special forces are thought to have used an aerosolized form of fentanyl to incapacitate everyone in the room. More than 100 of the hostages died, and the episode raised the specter of opioids being used in chemical attacks.
Dr. Pravetoni said vaccination could offer protection for first responders, law enforcement or other people whose professions place them at risk for inhalation, either accidentally or through such attacks.
These or other real-world applications for people at risk for exposure are several years away. Dr. Pravetoni said it took 10 years to get to this phase and estimates that, in about 5 years, a vaccine that targets multiple opioid drugs might enter the first clinical trial.
A version of this article first appeared on WebMD.com.
Opioid overdoses tied to lasting cognitive impairment
Opioid overdoses usually aren’t fatal, but a new review of numerous studies, mostly case reports and case series, suggests that they can have long-lasting effects on cognition, possibly because of hypoxia resulting from respiratory depression.
Erin L. Winstanley, PhD, MA, and associates noted in the review that opioids cause about 80% of worldwide deaths from illicit drug use, and the Centers for Disease Control and Prevention’s provisional August 2021 number of more than 88,000 opioid-caused deaths in the United States is the highest ever recorded – a 27% increase over what was reported last December. That number suggests that the opioid epidemic continues to rage, but the study results also show that the neurological consequences of nonfatal overdoses are an important public health problem.
And that’s something that may be overlooked, according to Mark S. Gold, MD, who was not involved with the study and was asked to comment on the review, which was published in the Journal of Addiction Science.
“Assuming that an overdose has no effect on the brain, mood, and behavior is not supported by experience or the literature. He is a University of Florida, Gainesville, Emeritus Eminent Scholar, adjunct professor of psychiatry at Washington University in St. Louis, and a member of the clinical council of Washington University’s Public Health Institute.
A common pattern among patients with opioid use disorder (OUD) is that they undergo treatment with medication-assisted therapy (MAT), only to drop out of treatment and then repeat the treatment at a later date. That suggests that physicians should take a harder look at the limitations of MAT and other treatments, Dr. Gold said.
Although the review found some associations between neurocognitive deficits and opioid overdose, the authors point out that it is difficult to make direct comparisons because of biases and differences in methodology among the included studies. They were not able to reach conclusions about the prevalence of brain injuries following nonfatal opioid overdoses. Few included studies controlled for confounding factors that might contribute to or explain neurocognitive impairments, reported Dr. Winstanley, associate professor in the department of behavioral medicine and psychiatry at the University of West Virginia, Morgantown, and associates.
Still, distinct patterns emerged from the analysis of almost 3,500 subjects in 79 studies in 21 countries. Twenty-nine studies reported diagnoses of leukoencephalopathy, which affects white matter. Spongiform leukoencephalopathy is known to occur secondarily after exposure to a variety of toxic agents, including carbon monoxide poisoning and drugs of abuse. The damage can lead to erosion of higher cerebral function. The condition can occur from 2 to 180 days after a hypoxic brain injury, potentially complicating efforts to attribute it specifically to an opioid overdose. Amnestic syndrome was also reported in some studies. One study found that about 39% of people seeking buprenorphine treatment suffered from neurocognitive impairment.
Dr. Gold called the study’s findings novel and of public health importance. “Each overdose takes a toll on the body, and especially the brain,” he said.
Better documentation needed
The variability in symptoms, as well as their timing, present challenges to initial treatment, which often occur before a patient reaches the hospital. This is a vital window because the length of time of inadequate respiration because of opioid overdose is likely to predict the extent of brain injury. The duration of inadequate respiration may not be captured in electronic medical records, and emergency departments don’t typically collect toxicology information, which may lead health care providers to attribute neurocognitive impairments to ongoing drug use rather than an acute anoxic or hypoxic episode. Further neurocognitive damage may have a delayed onset, and better documentation of these events could help physicians determine whether those symptoms stem from the acute event.
Dr. Winstanley and associates called for more research, including prospective case-control studies to identify brain changes following opioid-related overdose.
The authors also suggested that physicians might want to consider screening patients who experience prolonged anoxia or hypoxia for neurocognitive impairments and brain injuries. Dr. Gold agreed.
“Clinicians working with OUD patients should take these data to heart and take a comprehensive history of previous overdoses, loss of consciousness, head trauma, and following up on the history with neuropsychological and other tests of brain function,” Dr. Gold said. “After an assessment, rehabilitation and treatment might then be more personalized and effective.”
Dr. Gold had no relevant financial disclosures.
Opioid overdoses usually aren’t fatal, but a new review of numerous studies, mostly case reports and case series, suggests that they can have long-lasting effects on cognition, possibly because of hypoxia resulting from respiratory depression.
Erin L. Winstanley, PhD, MA, and associates noted in the review that opioids cause about 80% of worldwide deaths from illicit drug use, and the Centers for Disease Control and Prevention’s provisional August 2021 number of more than 88,000 opioid-caused deaths in the United States is the highest ever recorded – a 27% increase over what was reported last December. That number suggests that the opioid epidemic continues to rage, but the study results also show that the neurological consequences of nonfatal overdoses are an important public health problem.
And that’s something that may be overlooked, according to Mark S. Gold, MD, who was not involved with the study and was asked to comment on the review, which was published in the Journal of Addiction Science.
“Assuming that an overdose has no effect on the brain, mood, and behavior is not supported by experience or the literature. He is a University of Florida, Gainesville, Emeritus Eminent Scholar, adjunct professor of psychiatry at Washington University in St. Louis, and a member of the clinical council of Washington University’s Public Health Institute.
A common pattern among patients with opioid use disorder (OUD) is that they undergo treatment with medication-assisted therapy (MAT), only to drop out of treatment and then repeat the treatment at a later date. That suggests that physicians should take a harder look at the limitations of MAT and other treatments, Dr. Gold said.
Although the review found some associations between neurocognitive deficits and opioid overdose, the authors point out that it is difficult to make direct comparisons because of biases and differences in methodology among the included studies. They were not able to reach conclusions about the prevalence of brain injuries following nonfatal opioid overdoses. Few included studies controlled for confounding factors that might contribute to or explain neurocognitive impairments, reported Dr. Winstanley, associate professor in the department of behavioral medicine and psychiatry at the University of West Virginia, Morgantown, and associates.
Still, distinct patterns emerged from the analysis of almost 3,500 subjects in 79 studies in 21 countries. Twenty-nine studies reported diagnoses of leukoencephalopathy, which affects white matter. Spongiform leukoencephalopathy is known to occur secondarily after exposure to a variety of toxic agents, including carbon monoxide poisoning and drugs of abuse. The damage can lead to erosion of higher cerebral function. The condition can occur from 2 to 180 days after a hypoxic brain injury, potentially complicating efforts to attribute it specifically to an opioid overdose. Amnestic syndrome was also reported in some studies. One study found that about 39% of people seeking buprenorphine treatment suffered from neurocognitive impairment.
Dr. Gold called the study’s findings novel and of public health importance. “Each overdose takes a toll on the body, and especially the brain,” he said.
Better documentation needed
The variability in symptoms, as well as their timing, present challenges to initial treatment, which often occur before a patient reaches the hospital. This is a vital window because the length of time of inadequate respiration because of opioid overdose is likely to predict the extent of brain injury. The duration of inadequate respiration may not be captured in electronic medical records, and emergency departments don’t typically collect toxicology information, which may lead health care providers to attribute neurocognitive impairments to ongoing drug use rather than an acute anoxic or hypoxic episode. Further neurocognitive damage may have a delayed onset, and better documentation of these events could help physicians determine whether those symptoms stem from the acute event.
Dr. Winstanley and associates called for more research, including prospective case-control studies to identify brain changes following opioid-related overdose.
The authors also suggested that physicians might want to consider screening patients who experience prolonged anoxia or hypoxia for neurocognitive impairments and brain injuries. Dr. Gold agreed.
“Clinicians working with OUD patients should take these data to heart and take a comprehensive history of previous overdoses, loss of consciousness, head trauma, and following up on the history with neuropsychological and other tests of brain function,” Dr. Gold said. “After an assessment, rehabilitation and treatment might then be more personalized and effective.”
Dr. Gold had no relevant financial disclosures.
Opioid overdoses usually aren’t fatal, but a new review of numerous studies, mostly case reports and case series, suggests that they can have long-lasting effects on cognition, possibly because of hypoxia resulting from respiratory depression.
Erin L. Winstanley, PhD, MA, and associates noted in the review that opioids cause about 80% of worldwide deaths from illicit drug use, and the Centers for Disease Control and Prevention’s provisional August 2021 number of more than 88,000 opioid-caused deaths in the United States is the highest ever recorded – a 27% increase over what was reported last December. That number suggests that the opioid epidemic continues to rage, but the study results also show that the neurological consequences of nonfatal overdoses are an important public health problem.
And that’s something that may be overlooked, according to Mark S. Gold, MD, who was not involved with the study and was asked to comment on the review, which was published in the Journal of Addiction Science.
“Assuming that an overdose has no effect on the brain, mood, and behavior is not supported by experience or the literature. He is a University of Florida, Gainesville, Emeritus Eminent Scholar, adjunct professor of psychiatry at Washington University in St. Louis, and a member of the clinical council of Washington University’s Public Health Institute.
A common pattern among patients with opioid use disorder (OUD) is that they undergo treatment with medication-assisted therapy (MAT), only to drop out of treatment and then repeat the treatment at a later date. That suggests that physicians should take a harder look at the limitations of MAT and other treatments, Dr. Gold said.
Although the review found some associations between neurocognitive deficits and opioid overdose, the authors point out that it is difficult to make direct comparisons because of biases and differences in methodology among the included studies. They were not able to reach conclusions about the prevalence of brain injuries following nonfatal opioid overdoses. Few included studies controlled for confounding factors that might contribute to or explain neurocognitive impairments, reported Dr. Winstanley, associate professor in the department of behavioral medicine and psychiatry at the University of West Virginia, Morgantown, and associates.
Still, distinct patterns emerged from the analysis of almost 3,500 subjects in 79 studies in 21 countries. Twenty-nine studies reported diagnoses of leukoencephalopathy, which affects white matter. Spongiform leukoencephalopathy is known to occur secondarily after exposure to a variety of toxic agents, including carbon monoxide poisoning and drugs of abuse. The damage can lead to erosion of higher cerebral function. The condition can occur from 2 to 180 days after a hypoxic brain injury, potentially complicating efforts to attribute it specifically to an opioid overdose. Amnestic syndrome was also reported in some studies. One study found that about 39% of people seeking buprenorphine treatment suffered from neurocognitive impairment.
Dr. Gold called the study’s findings novel and of public health importance. “Each overdose takes a toll on the body, and especially the brain,” he said.
Better documentation needed
The variability in symptoms, as well as their timing, present challenges to initial treatment, which often occur before a patient reaches the hospital. This is a vital window because the length of time of inadequate respiration because of opioid overdose is likely to predict the extent of brain injury. The duration of inadequate respiration may not be captured in electronic medical records, and emergency departments don’t typically collect toxicology information, which may lead health care providers to attribute neurocognitive impairments to ongoing drug use rather than an acute anoxic or hypoxic episode. Further neurocognitive damage may have a delayed onset, and better documentation of these events could help physicians determine whether those symptoms stem from the acute event.
Dr. Winstanley and associates called for more research, including prospective case-control studies to identify brain changes following opioid-related overdose.
The authors also suggested that physicians might want to consider screening patients who experience prolonged anoxia or hypoxia for neurocognitive impairments and brain injuries. Dr. Gold agreed.
“Clinicians working with OUD patients should take these data to heart and take a comprehensive history of previous overdoses, loss of consciousness, head trauma, and following up on the history with neuropsychological and other tests of brain function,” Dr. Gold said. “After an assessment, rehabilitation and treatment might then be more personalized and effective.”
Dr. Gold had no relevant financial disclosures.
FROM THE JOURNAL OF ADDICTION SCIENCE
Assessing headache severity via migraine symptoms can help predict outcomes
according to an analysis of data from thousands of headache sufferers who recorded variables like pain and duration in a daily digital diary.
“Our hope is that this work serves as foundational basis for better understanding the complexity of headache as a symptom-based condition,” James S. McGinley, PhD, of Vector Psychometric Group in Chapel Hill, N.C., and coauthors wrote. The study was published in Cephalalgia.
To evaluate whether keeping track of daily headache features can produce a useful, predictive score, the researchers reviewed data from migraine patients that were collected via N1‑Headache, a commercial digital health platform. Ultimately, information from 4,380 adults with a self-reported migraine diagnosis was analyzed; the sample was 90% female and their mean age was 37 years. Study participants reported an average of 33 headaches per month over the last 3 months. Nine patient-reported variables were initially considered in calculating the Headache Day Severity (HDS) score: pain intensity, headache duration, aura, pulsating/throbbing pain, unilateral pain, pain aggravation by activity, nausea/vomiting, photophobia, and phonophobia.
After determining that unilateral pain was not a meaningful variable, the researchers’ model found that, for every 1 standard deviation increase in HDS, the patient’s odds of physician visit increased by 71% (odds ratio, 1.71; 95% confidence interval, 1.32-2.21) and the odds of an ED visit increased by 342% (OR, 4.42; 95% CI, 2.23-7.60). They also found that the likelihood of missed work or school increased by 190% (OR, 2.90; 95% CI, 2.56-3.29), the chances of missing household work increased by 237% (OR, 3.37; 95% CI, 3.06-3.72) and the odds of missing other leisure or social activity increased by 228% (OR, 3.28; 95% CI, 2.97-3.64).
Tracking multiple variables
“We encourage all of our patients to monitor their headaches; there are just too many variables to try to keep it in your head,” Robert Cowan, MD, professor of neurology and chief of the division of headache medicine at Stanford (Calif.) University, said in an interview. He referenced a previous study from the University of Washington where patients were asked to track their headaches; that data was then compared against their self-reported headaches at a quarterly physician visit.
“What they found was there was absolutely no correlation with reported frequency of headache at the visit and what was seen in the tracker,” he said. “If patients had a headache in the previous 3 days before their visit, they felt that their headaches were poorly controlled. If they hadn’t, they thought their headaches were under good control. So the value of tracking is pretty clear.”
He added that, while not every headache sufferer needs to track their daily routines and symptoms, once those symptoms interfere with your life on a day-to-day basis, it’s probably time to consider keeping tabs on yourself with a tool of some sort. And while this study’s calculated HDS score supports the idea of migraine’s complexity, it also leaves unanswered the question of how to treat patients with severe symptoms.
“Frequently,” he said, “we’ll see patients who say: ‘I can deal with the pain, but the nausea makes it impossible to work, or the light sensitivity makes it impossible to go outside.’ The big question within the headache community is, can you treat migraine and have it address the whole spectrum, from dizziness to light sensitivity to sound sensitivity to vertigo, or should you be going after individual symptoms? That’s a controversy that rages on; I think most of us go for a combination. We’re in a polypharmacy phase: ‘If nausea is a big problem, take this, but we also try to prevent the whole migraine complex, so take this as well.’ ”
The authors acknowledged their study’s limitations, including the inability to determine how many participants’ migraines were formally diagnosed by a trained medical professional and the lack of generalizability of data from a convenience sample, though they added that patients who independently track their own headaches “may be representative of those who would participate in a clinical trial.” In addition, as seven of the nine features were collected in N1‑Headache on a yes/no scale, they recognized that “increasing the number of response options for each item may improve our ability to measure HDS.”
The study was funded by Amgen through the Competitive Grant Program in Migraine Research. The authors declared several potential conflicts of interest, including receiving funding, research support, salary, and honoraria from various pharmaceutical companies.
according to an analysis of data from thousands of headache sufferers who recorded variables like pain and duration in a daily digital diary.
“Our hope is that this work serves as foundational basis for better understanding the complexity of headache as a symptom-based condition,” James S. McGinley, PhD, of Vector Psychometric Group in Chapel Hill, N.C., and coauthors wrote. The study was published in Cephalalgia.
To evaluate whether keeping track of daily headache features can produce a useful, predictive score, the researchers reviewed data from migraine patients that were collected via N1‑Headache, a commercial digital health platform. Ultimately, information from 4,380 adults with a self-reported migraine diagnosis was analyzed; the sample was 90% female and their mean age was 37 years. Study participants reported an average of 33 headaches per month over the last 3 months. Nine patient-reported variables were initially considered in calculating the Headache Day Severity (HDS) score: pain intensity, headache duration, aura, pulsating/throbbing pain, unilateral pain, pain aggravation by activity, nausea/vomiting, photophobia, and phonophobia.
After determining that unilateral pain was not a meaningful variable, the researchers’ model found that, for every 1 standard deviation increase in HDS, the patient’s odds of physician visit increased by 71% (odds ratio, 1.71; 95% confidence interval, 1.32-2.21) and the odds of an ED visit increased by 342% (OR, 4.42; 95% CI, 2.23-7.60). They also found that the likelihood of missed work or school increased by 190% (OR, 2.90; 95% CI, 2.56-3.29), the chances of missing household work increased by 237% (OR, 3.37; 95% CI, 3.06-3.72) and the odds of missing other leisure or social activity increased by 228% (OR, 3.28; 95% CI, 2.97-3.64).
Tracking multiple variables
“We encourage all of our patients to monitor their headaches; there are just too many variables to try to keep it in your head,” Robert Cowan, MD, professor of neurology and chief of the division of headache medicine at Stanford (Calif.) University, said in an interview. He referenced a previous study from the University of Washington where patients were asked to track their headaches; that data was then compared against their self-reported headaches at a quarterly physician visit.
“What they found was there was absolutely no correlation with reported frequency of headache at the visit and what was seen in the tracker,” he said. “If patients had a headache in the previous 3 days before their visit, they felt that their headaches were poorly controlled. If they hadn’t, they thought their headaches were under good control. So the value of tracking is pretty clear.”
He added that, while not every headache sufferer needs to track their daily routines and symptoms, once those symptoms interfere with your life on a day-to-day basis, it’s probably time to consider keeping tabs on yourself with a tool of some sort. And while this study’s calculated HDS score supports the idea of migraine’s complexity, it also leaves unanswered the question of how to treat patients with severe symptoms.
“Frequently,” he said, “we’ll see patients who say: ‘I can deal with the pain, but the nausea makes it impossible to work, or the light sensitivity makes it impossible to go outside.’ The big question within the headache community is, can you treat migraine and have it address the whole spectrum, from dizziness to light sensitivity to sound sensitivity to vertigo, or should you be going after individual symptoms? That’s a controversy that rages on; I think most of us go for a combination. We’re in a polypharmacy phase: ‘If nausea is a big problem, take this, but we also try to prevent the whole migraine complex, so take this as well.’ ”
The authors acknowledged their study’s limitations, including the inability to determine how many participants’ migraines were formally diagnosed by a trained medical professional and the lack of generalizability of data from a convenience sample, though they added that patients who independently track their own headaches “may be representative of those who would participate in a clinical trial.” In addition, as seven of the nine features were collected in N1‑Headache on a yes/no scale, they recognized that “increasing the number of response options for each item may improve our ability to measure HDS.”
The study was funded by Amgen through the Competitive Grant Program in Migraine Research. The authors declared several potential conflicts of interest, including receiving funding, research support, salary, and honoraria from various pharmaceutical companies.
according to an analysis of data from thousands of headache sufferers who recorded variables like pain and duration in a daily digital diary.
“Our hope is that this work serves as foundational basis for better understanding the complexity of headache as a symptom-based condition,” James S. McGinley, PhD, of Vector Psychometric Group in Chapel Hill, N.C., and coauthors wrote. The study was published in Cephalalgia.
To evaluate whether keeping track of daily headache features can produce a useful, predictive score, the researchers reviewed data from migraine patients that were collected via N1‑Headache, a commercial digital health platform. Ultimately, information from 4,380 adults with a self-reported migraine diagnosis was analyzed; the sample was 90% female and their mean age was 37 years. Study participants reported an average of 33 headaches per month over the last 3 months. Nine patient-reported variables were initially considered in calculating the Headache Day Severity (HDS) score: pain intensity, headache duration, aura, pulsating/throbbing pain, unilateral pain, pain aggravation by activity, nausea/vomiting, photophobia, and phonophobia.
After determining that unilateral pain was not a meaningful variable, the researchers’ model found that, for every 1 standard deviation increase in HDS, the patient’s odds of physician visit increased by 71% (odds ratio, 1.71; 95% confidence interval, 1.32-2.21) and the odds of an ED visit increased by 342% (OR, 4.42; 95% CI, 2.23-7.60). They also found that the likelihood of missed work or school increased by 190% (OR, 2.90; 95% CI, 2.56-3.29), the chances of missing household work increased by 237% (OR, 3.37; 95% CI, 3.06-3.72) and the odds of missing other leisure or social activity increased by 228% (OR, 3.28; 95% CI, 2.97-3.64).
Tracking multiple variables
“We encourage all of our patients to monitor their headaches; there are just too many variables to try to keep it in your head,” Robert Cowan, MD, professor of neurology and chief of the division of headache medicine at Stanford (Calif.) University, said in an interview. He referenced a previous study from the University of Washington where patients were asked to track their headaches; that data was then compared against their self-reported headaches at a quarterly physician visit.
“What they found was there was absolutely no correlation with reported frequency of headache at the visit and what was seen in the tracker,” he said. “If patients had a headache in the previous 3 days before their visit, they felt that their headaches were poorly controlled. If they hadn’t, they thought their headaches were under good control. So the value of tracking is pretty clear.”
He added that, while not every headache sufferer needs to track their daily routines and symptoms, once those symptoms interfere with your life on a day-to-day basis, it’s probably time to consider keeping tabs on yourself with a tool of some sort. And while this study’s calculated HDS score supports the idea of migraine’s complexity, it also leaves unanswered the question of how to treat patients with severe symptoms.
“Frequently,” he said, “we’ll see patients who say: ‘I can deal with the pain, but the nausea makes it impossible to work, or the light sensitivity makes it impossible to go outside.’ The big question within the headache community is, can you treat migraine and have it address the whole spectrum, from dizziness to light sensitivity to sound sensitivity to vertigo, or should you be going after individual symptoms? That’s a controversy that rages on; I think most of us go for a combination. We’re in a polypharmacy phase: ‘If nausea is a big problem, take this, but we also try to prevent the whole migraine complex, so take this as well.’ ”
The authors acknowledged their study’s limitations, including the inability to determine how many participants’ migraines were formally diagnosed by a trained medical professional and the lack of generalizability of data from a convenience sample, though they added that patients who independently track their own headaches “may be representative of those who would participate in a clinical trial.” In addition, as seven of the nine features were collected in N1‑Headache on a yes/no scale, they recognized that “increasing the number of response options for each item may improve our ability to measure HDS.”
The study was funded by Amgen through the Competitive Grant Program in Migraine Research. The authors declared several potential conflicts of interest, including receiving funding, research support, salary, and honoraria from various pharmaceutical companies.
FROM CEPHALALGIA
Most muscle pain on statins not a drug effect: SAMSON in print
but by the expectation of such adverse effects, conclude researchers behind the randomized SAMSON trial, now fully published .
It’s common for patients to stop taking their statin because of muscle pain and their belief that the drug itself is to blame. That can sometimes be true, but the SAMSON trial, owing to its unusual design, makes a strong case that such symptoms are usually a nocebo effect.
That is, most statin-related muscle symptoms are likely “driven by the act of taking tablets rather than whether the tablets contain a statin,” concludes the report, which appears in the September 21 issue of the Journal of the American College of Cardiology, with lead authors James P. Howard, PhD, and Frances A. Wood, MPhil, Imperial College London.
SAMSON had been presented at the American Heart Association Scientific Sessions 2020 virtual meeting, covered at the time by this news organization, and simultaneously published in abbreviated form as correspondence in the New England Journal of Medicine.
“SAMSON suggests that the bulk of statin-related intolerable side effects arise from the taking of a tablet, not from statin therapy per se,” agrees an editorial accompanying the new publication.
“The study also demonstrates that the informal experimentation of stopping and restarting a statin to evaluate symptom resolution and reinduction without use of a placebo leads to nocebo symptoms misattributed to the statin,” writes Peter P. Toth, MD, PhD, Johns Hopkins University, Baltimore.
Statin intolerance, he continues, “warrants considerable further investigation, because it undermines standard of care for a very large number of patients worldwide,” leaving them vulnerable to atherosclerotic cardiovascular disease events. “Aches and pains are a fact of life; just because a patient has them does not mean they should be attributed to their statin.”
SAMSON assigned 35 men and 25 women to take atorvastatin 20 mg/day, its matching placebo, or neither pill each for 1 month in randomly alternating order for 12 months, with double-blinding, such that each of the three regimens was maintained for a total of 4 months.
The patients, 77% of whom were prescribed statins for primary prevention and all of whom had a history of stopping the drugs because of adverse effects, documented the severity of any perceived adverse effects on a smartphone app, with a “symptom score” ranging from 0 to 100.
The symptom score averaged 8.0 in months when no tablet was taken, but it was much higher in other months: 15.4 in placebo-pill months and 16.3 in months when atorvastatin was taken. The no-tablet score was significantly lower (P < .001) than either of the two other scores, which themselves were not significantly different from each other.
Eleven patients were unable to complete all 12 one-month segments of the trial, including five because of severe symptoms, but discontinuation was no more likely to occur in the atorvastatin group than in the placebo group.
The authors calculated an overall 0.90 “nocebo ratio” for the study, defined as the difference between symptom intensity on placebo and on no pill, divided by the difference between symptom intensity on atorvastatin and on no pill.
That means, the authors propose, that 90% of the symptom burden felt by patients receiving atorvastatin was also felt on the placebo pill and could be attributed to the nocebo effect.
“Prompt onset and offset of symptoms after starting and stopping tablets is often interpreted by patients and clinicians as evidence of causation. Our data indicate that this is true,” the authors write, but “the causation is from taking a tablet, rather than from the tablet being a statin.”
SAMSON was funded by the British Heart Foundation and supported by the National Institute for Health Research Imperial Biomedical Research Centre and the Imperial Clinical Trials Unit. Dr. Howard is supported by the Wellcome Trust. Dr. Wood declared no conflicts. Disclosures for the other authors are in the report. Dr. Toth discloses serving as a consultant to Amarin, Amgen, AstraZeneca, nio89, Kowa, Merck, Resverlogix, and Theravance; and serving on a speaker’s bureau for Amarin, Amgen, Esperion, and NovoNordisk.
A version of this article first appeared on Medscape.com.
but by the expectation of such adverse effects, conclude researchers behind the randomized SAMSON trial, now fully published .
It’s common for patients to stop taking their statin because of muscle pain and their belief that the drug itself is to blame. That can sometimes be true, but the SAMSON trial, owing to its unusual design, makes a strong case that such symptoms are usually a nocebo effect.
That is, most statin-related muscle symptoms are likely “driven by the act of taking tablets rather than whether the tablets contain a statin,” concludes the report, which appears in the September 21 issue of the Journal of the American College of Cardiology, with lead authors James P. Howard, PhD, and Frances A. Wood, MPhil, Imperial College London.
SAMSON had been presented at the American Heart Association Scientific Sessions 2020 virtual meeting, covered at the time by this news organization, and simultaneously published in abbreviated form as correspondence in the New England Journal of Medicine.
“SAMSON suggests that the bulk of statin-related intolerable side effects arise from the taking of a tablet, not from statin therapy per se,” agrees an editorial accompanying the new publication.
“The study also demonstrates that the informal experimentation of stopping and restarting a statin to evaluate symptom resolution and reinduction without use of a placebo leads to nocebo symptoms misattributed to the statin,” writes Peter P. Toth, MD, PhD, Johns Hopkins University, Baltimore.
Statin intolerance, he continues, “warrants considerable further investigation, because it undermines standard of care for a very large number of patients worldwide,” leaving them vulnerable to atherosclerotic cardiovascular disease events. “Aches and pains are a fact of life; just because a patient has them does not mean they should be attributed to their statin.”
SAMSON assigned 35 men and 25 women to take atorvastatin 20 mg/day, its matching placebo, or neither pill each for 1 month in randomly alternating order for 12 months, with double-blinding, such that each of the three regimens was maintained for a total of 4 months.
The patients, 77% of whom were prescribed statins for primary prevention and all of whom had a history of stopping the drugs because of adverse effects, documented the severity of any perceived adverse effects on a smartphone app, with a “symptom score” ranging from 0 to 100.
The symptom score averaged 8.0 in months when no tablet was taken, but it was much higher in other months: 15.4 in placebo-pill months and 16.3 in months when atorvastatin was taken. The no-tablet score was significantly lower (P < .001) than either of the two other scores, which themselves were not significantly different from each other.
Eleven patients were unable to complete all 12 one-month segments of the trial, including five because of severe symptoms, but discontinuation was no more likely to occur in the atorvastatin group than in the placebo group.
The authors calculated an overall 0.90 “nocebo ratio” for the study, defined as the difference between symptom intensity on placebo and on no pill, divided by the difference between symptom intensity on atorvastatin and on no pill.
That means, the authors propose, that 90% of the symptom burden felt by patients receiving atorvastatin was also felt on the placebo pill and could be attributed to the nocebo effect.
“Prompt onset and offset of symptoms after starting and stopping tablets is often interpreted by patients and clinicians as evidence of causation. Our data indicate that this is true,” the authors write, but “the causation is from taking a tablet, rather than from the tablet being a statin.”
SAMSON was funded by the British Heart Foundation and supported by the National Institute for Health Research Imperial Biomedical Research Centre and the Imperial Clinical Trials Unit. Dr. Howard is supported by the Wellcome Trust. Dr. Wood declared no conflicts. Disclosures for the other authors are in the report. Dr. Toth discloses serving as a consultant to Amarin, Amgen, AstraZeneca, nio89, Kowa, Merck, Resverlogix, and Theravance; and serving on a speaker’s bureau for Amarin, Amgen, Esperion, and NovoNordisk.
A version of this article first appeared on Medscape.com.
but by the expectation of such adverse effects, conclude researchers behind the randomized SAMSON trial, now fully published .
It’s common for patients to stop taking their statin because of muscle pain and their belief that the drug itself is to blame. That can sometimes be true, but the SAMSON trial, owing to its unusual design, makes a strong case that such symptoms are usually a nocebo effect.
That is, most statin-related muscle symptoms are likely “driven by the act of taking tablets rather than whether the tablets contain a statin,” concludes the report, which appears in the September 21 issue of the Journal of the American College of Cardiology, with lead authors James P. Howard, PhD, and Frances A. Wood, MPhil, Imperial College London.
SAMSON had been presented at the American Heart Association Scientific Sessions 2020 virtual meeting, covered at the time by this news organization, and simultaneously published in abbreviated form as correspondence in the New England Journal of Medicine.
“SAMSON suggests that the bulk of statin-related intolerable side effects arise from the taking of a tablet, not from statin therapy per se,” agrees an editorial accompanying the new publication.
“The study also demonstrates that the informal experimentation of stopping and restarting a statin to evaluate symptom resolution and reinduction without use of a placebo leads to nocebo symptoms misattributed to the statin,” writes Peter P. Toth, MD, PhD, Johns Hopkins University, Baltimore.
Statin intolerance, he continues, “warrants considerable further investigation, because it undermines standard of care for a very large number of patients worldwide,” leaving them vulnerable to atherosclerotic cardiovascular disease events. “Aches and pains are a fact of life; just because a patient has them does not mean they should be attributed to their statin.”
SAMSON assigned 35 men and 25 women to take atorvastatin 20 mg/day, its matching placebo, or neither pill each for 1 month in randomly alternating order for 12 months, with double-blinding, such that each of the three regimens was maintained for a total of 4 months.
The patients, 77% of whom were prescribed statins for primary prevention and all of whom had a history of stopping the drugs because of adverse effects, documented the severity of any perceived adverse effects on a smartphone app, with a “symptom score” ranging from 0 to 100.
The symptom score averaged 8.0 in months when no tablet was taken, but it was much higher in other months: 15.4 in placebo-pill months and 16.3 in months when atorvastatin was taken. The no-tablet score was significantly lower (P < .001) than either of the two other scores, which themselves were not significantly different from each other.
Eleven patients were unable to complete all 12 one-month segments of the trial, including five because of severe symptoms, but discontinuation was no more likely to occur in the atorvastatin group than in the placebo group.
The authors calculated an overall 0.90 “nocebo ratio” for the study, defined as the difference between symptom intensity on placebo and on no pill, divided by the difference between symptom intensity on atorvastatin and on no pill.
That means, the authors propose, that 90% of the symptom burden felt by patients receiving atorvastatin was also felt on the placebo pill and could be attributed to the nocebo effect.
“Prompt onset and offset of symptoms after starting and stopping tablets is often interpreted by patients and clinicians as evidence of causation. Our data indicate that this is true,” the authors write, but “the causation is from taking a tablet, rather than from the tablet being a statin.”
SAMSON was funded by the British Heart Foundation and supported by the National Institute for Health Research Imperial Biomedical Research Centre and the Imperial Clinical Trials Unit. Dr. Howard is supported by the Wellcome Trust. Dr. Wood declared no conflicts. Disclosures for the other authors are in the report. Dr. Toth discloses serving as a consultant to Amarin, Amgen, AstraZeneca, nio89, Kowa, Merck, Resverlogix, and Theravance; and serving on a speaker’s bureau for Amarin, Amgen, Esperion, and NovoNordisk.
A version of this article first appeared on Medscape.com.
Provider Perceptions of Opioid Safety Measures in VHA Emergency Departments and Urgent Care Centers
The United States is facing an opioid crisis in which approximately 10 million people have misused opioids in the past year, and an estimated 2 million people have an opioid use disorder (OUD).1 Compared with the general population, veterans treated in the Veterans Health Administration (VHA) facilities are at nearly twice the risk for accidental opioid overdose.2 The implementation of opioid safety measures in VHA facilities across all care settings is a priority in addressing this public health crisis. Hence, VHA leadership is working to minimize veteran risk of fatal opioid overdoses and to increase veteran access to medication-assisted treatments (MAT) for OUD.3
Since the administration of our survey, the VHA has shifted to using the term medication for opioid use disorder (MOUD) instead of MAT for OUD. However, for consistency with the survey we distributed, we use MAT in this analysis.
Acute care settings represent an opportunity to offer appropriate opioid care and treatment options to patients at risk for OUD or opioid-related overdose. VHA facilities offer 2 outpatient acute care settings for emergent ambulatory care: emergency departments (EDs) and urgent care centers (UCCs). Annually, these settings see an estimated 2.5 million patients each year, making EDs and UCCs critical access points of OUD care for veterans. Partnering with key national VHA stakeholders from Pharmacy Benefits Management (PBM), the Office of Emergency Medicine, and Academic Detailing Services (ADS), we developed the Emergency Department Opioid Safety Initiative (ED OSI) aimed at implementing and evaluating opioid safety measures in VHA outpatient acute care settings.
The US Department of Veterans Affairs (VA)/Department of Defense (DoD) Clinical Practice Guidelines for Opioid Therapy for Chronic Pain (CPG) makes recommendations for the initiation and continuation of opioids, risk mitigation, taper of opioids, and opioid therapy for acute pain in VHA facilities.4 Using these recommendations, we developed the broad aims of the ED OSI quality improvement (QI) program. The CPG is clear about the prioritization of safe opioid prescribing practices. New opioid prescriptions written in the ED have been associated with continued and chronic opioid use.5 At the time of prescription, patients not currently and chronically on opioids who receive more than a 3-day supply are at increased risk of becoming long-term opioid users.6 Given the annual volume of patients seen, VHA ED/UCCs are a crucial area for implementing better opioid prescribing practices.
The CPG also includes recommendations for the prescribing or coprescribing of naloxone rescue kits. The administration of naloxone following opioid overdose has been found to be an effective measure against fatal overdose. Increasing provider awareness of common risk factors for opioid-related overdose (eg, frequent ED visits or hospitalizations) helps facilitate a discussion on naloxone prescribing at discharge. Prior studies provide evidence that naloxone distribution and accompanying education also are effective in reducing opioid overdose mortalityand ED visits related to adverse opioid-related events.7,8
Similarly, the guidelines provide recommendations for the use of MAT for veterans with OUD. MAT for OUD is considered a first-line treatment option for patients with moderate-to-severe OUD. When used to treat patients with unsafe opioid use, this treatment helps alleviate symptoms of withdrawal, which can increase opioid taper adherence and has a protective effect against opioid overdose mortality.9 MAT initiated in the ED can increase patient engagement to addiction services.10
These 3 CPG recommendations serve as the basis for the broad goals of the ED OSI program. We aim to develop, implement, and evaluate programs and initiatives to (aim 1) reduce inappropriate opioid prescribing from VHA EDs; (aim 2) increase naloxone distribution from VHA EDs; and (aim 3) increase access to MAT initiation from VHA EDs through the implementation of ED-based MAT-initiation programs with EDs across the VHA. Aim 1 was a focused and strategic QI effort to implement an ED-based program to reduce inappropriate opioid prescribing. The ED OSI prescribing program offered a 4-step bundled approach: (1) sharing of opioid prescribing dashboard data with ED medical director and academic detailer; (2) education of ED providers and implementation of toolkit resources; (3) academic detailers conduct audit and feedback session(s) with highest prescribers; and (4) quarterly reports of opioid prescribing data to ED providers.
Results from the pilot suggested that our program was associated with accelerating the rate at which ED prescribing rates decreased.11 In addition, the pilot found that ED-based QI initiatives in VHA facilities are a feasible practice. As we work to develop and implement the next 2 phases of the QI program, a major consideration is to identify facilitators and address any existing barriers to the implementation of naloxone distribution (aim 2) and MAT-initiation (aim 3) programs for treatment-naïve patients from VHA EDs. To date, there have been no recent published studies examining the barriers and facilitators to use or implementation of MAT initiation or naloxone distribution in VHA facilities or, more specifically, from VHA EDs.12 As part of our QI program, we set out to better understand VHA ED provider perceptions of barriers and facilitators to implementation of programs aimed at increasing naloxone distribution and initiation of MAT for treatment-naïve patients in the ED.
Methods
This project received a QI designation from the Office of PBM Academic Detailing Service Institutional Review Board at the Edward Hines, Jr. Veterans Affairs Hospital VA Medical Center (VAMC). This designation was reviewed and approved by the Rocky Mountain Regional VAMC Research and Development service. In addition, we received national union approval to disseminate this survey nationally across all VA Integrated Service Networks (VISNs).
Survey
We worked with VHA subject matter experts, key stakeholders, and the VA Collaborative Evaluation Center (VACE) to develop the survey. Subject matter experts and stakeholders included VHA emergency medicine leadership, ADS leadership, and mental health and substance treatment providers. VACE is an interdisciplinary group of mixed-method researchers. The survey questions aimed to capture perceptions and experiences regarding naloxone distribution and new MAT initiation of VHA ED/UCC providers.
We used a variety of survey question formats. Close-ended questions with a predefined list of answer options were used to capture discrete domains, such as demographic information, comfort level, and experience level. To capture health care provider (HCP) perceptions on barriers and facilitators, we used multiple-answer multiple-choice questions. Built into this question format was a free-response option, which allowed respondents to offer additional barriers or facilitators. Respondents also had the option of not answering individual questions.
We identified physicians, nurse practitioners (NPs), and physician assistants (PAs) who saw at least 100 patients in the ED or UCC in at least one 3-month period in the prior year and obtained an email address for each. In total, 2228 ED or UCC providers across 132 facilities were emailed a survey; 1883 (84.5%) were ED providers and 345 (15.5%) were UCC providers.
We used Research Electronic Data Capture (REDCap) software to build and disseminate the survey via email. Surveys were initially disseminated in late January 2019. During the 3-month survey period, recipients received 3 automated email reminders from REDCap to complete the survey. Survey data were exported from REDCap. Results were analyzed using descriptive statistics analyses with Microsoft Excel.
Results
One respondent received the survey in error and was excluded from the analysis. The survey response rate was 16.7%: 372 responses from 103 unique facilities. Each VISN had a mean 20 respondents. The majority of respondents (n = 286, 76.9%) worked in highly complex level 1 facilities characterized by high patient volume and more high-risk patients and were teaching and research facilities. Respondents were asked to describe their most recent ED or UCC role. While 281 respondents (75.5%) were medical doctors, 61 respondents (16.4%) were NPs, 30 (8.1%) were PAs, and 26 (7.0%) were ED/UCC chiefs or medical directors (Table 1). Most respondents (80.4%) reported at least 10 years of health care experience.
The majority of respondents (72.9%) believed that HCPs at their VHA facility should be prescribing naloxone. When asked to specify which HCPs should be prescribing naloxone, most HCP respondents selected pharmacists (76.4%) and substance abuse providers (71.6%). Less than half of respondents (45.0%) felt that VA ED/UCC providers also should be prescribing naloxone. However, 58.1% of most HCP respondents reported being comfortable or very comfortable with prescribing naloxone to a patient in the ED or UCC who already had an existing prescription of opioids. Similarly, 52.7% of respondents reported being comfortable or very comfortable with coprescribing naloxone when discharging a patient with an opioid prescription from the ED/UCC. Notably, while 36.7% of PAs reported being comfortable/very comfortable coprescribing naloxone, 46.7% reported being comfortable/very comfortable prescribing naloxone to a patient with an existing opioid prescription. Physicians and NPs expressed similar levels of comfort with coprescribing and prescribing naloxone.
Respondents across provider types indicated a number of barriers to prescribing naloxone to medically appropriate patients (Table 2). Many respondents indicated prescribing naloxone was beyond the ED/UCC provider scope of practice (35.2%), followed by the perceived stigma associated with naloxone (33.3%), time required to prescribe naloxone (23.9%), and concern with patient’s ability to use naloxone (22.8%).
Facilitators for prescribing naloxone to medically appropriate patients identified by HCP respondents included pharmacist help and education (44.6%), patient knowledge of medication options (31.7%), societal shift away from opioids for pain management (28.0%), facility leadership (26.9%), and patient interest in safe opioid usage (26.6%) (Table 3). In addition, NPs specifically endorsed
Less than 6.8% of HCP respondents indicated that they were comfortable using MAT. Meanwhile, 42.1% of respondents reported being aware of MAT but not familiar with it, and 23.5% reported that they were unaware of MAT. Correspondingly, 301 of the 372 (88.5%) HCP respondents indicated that they had not prescribed MAT in the past year. Across HCP types, only 24.1% indicated that it is the role of VA ED or UCC providers to prescribe MAT when medically appropriate and subsequently refer patients to substance abuse treatment for follow-up (just 7.1% of PAs endorsed this). Furthermore, 6.5% and 18.8% of HCP respondents indicated that their facility leadership was very supportive and supportive, respectively, of MAT for OUD prescribing.
Barriers to MAT initiation indicated by HCP respondents included limited scope of ED and UCC practice (53.2%), unclear follow-up/referral process (50.3%), time (29.8%), and discomfort (28.2%). Nearly one-third of NPs (27.9%) identified patient willingness/ability as a barrier to MAT initiation (Table 4).
Facilitators of MAT initiation in the ED or UCC included VHA same-day treatment options (34.9%), patient desire (32.5%), pharmacist help/education (27.4%), and psychiatric social workers in the ED or UCC (25.3%). Some NPs (23.0%) and PAs (26.7%) also indicated that having time to educate veterans about the medication would be a facilitator (Table 5). Facility leadership support was considered a facilitator by 30% of PAs.
Discussion
To the best of our knowledge, there have not been any studies examining HCP perceptions of the barriers and facilitators to naloxone distribution or the initiation of MAT in VHA ED and UCCs. Veterans are at an increased risk of overdose when compared with the general population, and increasing access to opioid safety measures (eg, safer prescribing practices, naloxone distribution) and treatment with MAT for OUD across all clinical settings has been a VHA priority.3
National guidance from VHA leadership, the Centers for Disease Control and Prevention (CDC), the US Surgeon General, and the US Department of Health and Human Services (HHS) call for an all-hands-on-deck approach to combatting opioid overdose with naloxone distribution or MAT (such as buprenorphine) initiation.13 VHA ED and UCC settings provide acute outpatient care to patients with medical or psychiatric illnesses or injuries that the patient believes requires emergent or immediate medical attention or for which there is a critical need for treatment to prevent deterioration of the condition or the possible impairment of recovery.14 However, ED and UCC environments are often regarded as settings meant to stabilize a patient until they can be seen by a primary care or long-term care provider.
A major barrier identified by HCPs was that MAT for OUD was outside their ED/UCC scope of practice, which suggests a need for a top-down or peer-to-peer reexamination of the role of HCPs in ED/UCC settings. Any naloxone distribution and/or MAT-initiation program in VHA ED/UCCs should consider education about the role of ED/UCC HCPs in opioid safety and treatment.
Only 25.3% of HCPs reported that their facility leadership was supportive or very supportive of MAT prescribing. This suggests that facility leadership should be engaged in any efforts to implement a MAT-initiation program in the facility’s ED. Engaging leadership in efforts to implement ED-based MAT programs will allow for a better understanding of leadership goals as related to opioid safety and an opportunity to address concerns regarding prescribing MAT in the ED. We recommend engaging facility leadership early in MAT implementation efforts. Respectively, 12.4% and 28.2% of HCP respondents reported discomfort prescribing naloxone or using MAT, suggesting a need for more education. Similarly, only 6.8% of HCPs reported comfort with using MAT.
A consideration for implementing ED/UCC-based MAT should be the inclusion of a training component. An evidence-based clinical treatment pathway that is appropriate to the ED/UCC setting and facility on the administration of MAT also could be beneficial. A clinical treatment pathway that includes ED/UCC-initiated discharge recommendations would address HCP concerns of unclear follow-up plans and system for referral of care. To this end, a key implementation task is coordinating with other outpatient services (eg, pain management clinic, substance use disorder treatment clinic) equipped for long-term patient follow-up to develop a system for referral of care. For example, as part of the clinical treatment pathway, an ED can develop a system of referral for patients initiated on MAT in the ED in which patients are referred for follow-up at the facility’s substance use disorder treatment clinic to be seen within 72 hours to continue the administration of MAT (such as buprenorphine).
In addition to HCP education, results suggest that patient/veteran education regarding naloxone and/or MAT should be considered. HCPs indicated that having help from a pharmacist to educate the patient about the medications would be a facilitator to naloxone distribution and MAT initiation. Similarly, patient knowledge of the medications also was endorsed as a facilitator. As such, a consideration for any future ED/UCC-based naloxone distribution or MAT-initiation programs in the VHA should be patient education whether by a clinically trained professional or an educational campaign for veterans.
Expanded naloxone distribution and initiation of MAT for OUD for EDs/UCCs across the VHA could impact the lives of veterans on long-term opioid therapy, with OUD, or who are otherwise at risk for opioid overdose. Steps taken to address the barriers and leverage the facilitators identified by HCP respondents can greatly reduce current obstacles to widespread implementation of ED/UCC-based naloxone distribution and MAT initiation nationally within the VHA.
Limitations
This survey had a low response rate (16.7%). One potential explanation for the low response rate is that when the survey was deployed, many of the VHA ED/UCC physicians were per-diem employees. Per-diem physicians may be less engaged and aware of site facilitators or barriers to naloxone and MAT prescribing. This, too, may have potentially skewed the collected data. However, the survey did not ask HCPs to disclose their employment status; thus, exact rates of per diem respondents are unknown.
We aimed to capture only self-perceived barriers to prescribing naloxone and MAT in the ED, but we did not capture or measure HCP respondent’s actual prescribing rates of MAT or naloxone. Understanding HCP perceptions of naloxone distribution and MAT initiation in the ED may have been further informed by comparing HCP responses to their actual clinical practice as related to their prescribing of these medications. In future research, we will link HCPs with the actual numbers of naloxone and MAT medications prescribed. Additionally, we do not know how many of these barriers or proposed facilitators will impact clinical practice.
Conclusions
A key aim for VHA leadership is to increase veteran access to naloxone distribution and MAT for OUD across clinical areas. The present study aimed to identify HCP perceptions of barriers and facilitators to the naloxone distribution and MAT-initiation programs in VHA ED/UCCs to inform the development of a targeted QI program to implement these opioid safety measures. Although the survey yielded a low response rate, results allowed us to identify important action items for our QI program, such as the development of clear protocols, follow-up plans, and systems for referral of care and HCP educational materials related to MAT and naloxone. We hope this work will serve as the basis for ED/UCC-tailored programs that can provide customized educational programs for HCPs designed to overcome known barriers to naloxone and MAT initiation.
Acknowledgments
This work was supported by the VA Office of Specialty Care Services 10P11 and through funding provided by the Comprehensive Addiction and Recovery Act (CARA).
1. Substance Abuse and Mental Health Services Administration. Key substance use and mental health indicators in the united states: results from the 2018 National Survey on Drug Use and Health. Published August 2019. Accessed August 20, 2021. https://www.samhsa.gov/data/sites/default/files/cbhsq-reports/NSDUHNationalFindingsReport2018/NSDUHNationalFindingsReport2018.pdf
2. Bohnert AS, Ilgen MA, Galea S, McCarthy JF, Blow FC. Accidental poisoning mortality among patients in the Department of Veterans Affairs Health System. Med Care. 2011;49(4):393-396. doi:10.1097/MLR.0b013e318202aa27
3. US Department of Veterans Affairs, Pharmacy Benefits Management Service. Recommendations for issuing naloxone rescue for the VA opioid overdose education and naloxone distribution (OEND) program. Published August 2016. Accessed August 20, 2021. https://www.pbm.va.gov/PBM/clinicalguidance/clinicalrecommendations/Naloxone_HCl_Rescue_Kits_Recommendations_for_Use.pdf
4. US Department of Defense, US Department of Veterans Affairs, Opioid Therapy for Chronic Pain Work Group. VA/DoD clinical practice guideline for opioid therapy for chronic pain. Published February 2017. Accessed August 20, 2021. https://www.va.gov/HOMELESS/nchav/resources/docs/mental-health/substance-abuse/VA_DoD-CLINICAL-PRACTICE-GUIDELINE-FOR-OPIOID-THERAPY-FOR-CHRONIC-PAIN-508.pdf
5. Barnett ML, Olenski AR, Jena AB. Opioid-prescribing patterns of emergency physicians and risk of long-term use. N Engl J Med. 2017;376(7):663-673. doi:10.1056/NEJMsa1610524
6. Shah A, Hayes CJ, Martin BC. Characteristics of initial prescription episodes and likelihood of long-term opioid use - United States, 2006-2015. MMWR Morb Mortal Wkly Rep. 2017;66(10):265-269. Published 2017 Mar 17. doi:10.15585/mmwr.mm6610a1
7. Clark AK, Wilder CM, Winstanley EL. A systematic review of community opioid overdose prevention and naloxone distribution programs. J Addict Med. 2014;8(3):153-163. doi:10.1097/ADM.0000000000000034
8. Coffin PO, Behar E, Rowe C, et al. Nonrandomized intervention study of naloxone coprescription for primary care patients receiving long-term opioid therapy for Pain. Ann Intern Med. 2016;165(4):245-252. doi:10.7326/M15-2771
9. Ma J, Bao YP, Wang RJ, et al. Effects of medication-assisted treatment on mortality among opioids users: a systematic review and meta-analysis. Mol Psychiatry. 2019;24(12):1868-1883. doi:10.1038/s41380-018-0094-5
10. D’Onofrio G, O’Connor PG, Pantalon MV, et al. Emergency department-initiated buprenorphine/naloxone treatment for opioid dependence: a randomized clinical trial. JAMA. 2015;313(16):1636-1644. doi:10.1001/jama.2015.3474
11. Dieujuste N, Johnson-Koenke R, Christopher M, et al. Feasibility study of a quasi-experimental regional opioid safety prescribing program in Veterans Health Administration emergency departments. Acad Emerg Med. 2020;27(8):734-741. doi:10.1111/acem.13980
12. Mackey K, Veazie S, Anderson J, Bourne D, Peterson K. Evidence brief: barriers and facilitators to use of medications for opioid use disorder. Published July 2017. Accessed August 20, 2021. http://www.ncbi.nlm.nih.gov/books/NBK549203/
13. US Department of Health and Human Services, Office of the Surgeon General. Naloxone: the opioid reversal drug that saves lives. Published December 2018. Accessed August 20, 2021. https://www.hhs.gov/opioids/sites/default/files/2018-12/naloxone-coprescribing-guidance.pdf
14. US Department of Veterans Affairs, Veterans Health Administration. Chapter 256: Emergency department (ED) and urgent care clinic (UCC). Updated October 3, 2016. Accessed August 20, 2021. https://www.cfm.va.gov/til/space/spChapter256.pdf.
The United States is facing an opioid crisis in which approximately 10 million people have misused opioids in the past year, and an estimated 2 million people have an opioid use disorder (OUD).1 Compared with the general population, veterans treated in the Veterans Health Administration (VHA) facilities are at nearly twice the risk for accidental opioid overdose.2 The implementation of opioid safety measures in VHA facilities across all care settings is a priority in addressing this public health crisis. Hence, VHA leadership is working to minimize veteran risk of fatal opioid overdoses and to increase veteran access to medication-assisted treatments (MAT) for OUD.3
Since the administration of our survey, the VHA has shifted to using the term medication for opioid use disorder (MOUD) instead of MAT for OUD. However, for consistency with the survey we distributed, we use MAT in this analysis.
Acute care settings represent an opportunity to offer appropriate opioid care and treatment options to patients at risk for OUD or opioid-related overdose. VHA facilities offer 2 outpatient acute care settings for emergent ambulatory care: emergency departments (EDs) and urgent care centers (UCCs). Annually, these settings see an estimated 2.5 million patients each year, making EDs and UCCs critical access points of OUD care for veterans. Partnering with key national VHA stakeholders from Pharmacy Benefits Management (PBM), the Office of Emergency Medicine, and Academic Detailing Services (ADS), we developed the Emergency Department Opioid Safety Initiative (ED OSI) aimed at implementing and evaluating opioid safety measures in VHA outpatient acute care settings.
The US Department of Veterans Affairs (VA)/Department of Defense (DoD) Clinical Practice Guidelines for Opioid Therapy for Chronic Pain (CPG) makes recommendations for the initiation and continuation of opioids, risk mitigation, taper of opioids, and opioid therapy for acute pain in VHA facilities.4 Using these recommendations, we developed the broad aims of the ED OSI quality improvement (QI) program. The CPG is clear about the prioritization of safe opioid prescribing practices. New opioid prescriptions written in the ED have been associated with continued and chronic opioid use.5 At the time of prescription, patients not currently and chronically on opioids who receive more than a 3-day supply are at increased risk of becoming long-term opioid users.6 Given the annual volume of patients seen, VHA ED/UCCs are a crucial area for implementing better opioid prescribing practices.
The CPG also includes recommendations for the prescribing or coprescribing of naloxone rescue kits. The administration of naloxone following opioid overdose has been found to be an effective measure against fatal overdose. Increasing provider awareness of common risk factors for opioid-related overdose (eg, frequent ED visits or hospitalizations) helps facilitate a discussion on naloxone prescribing at discharge. Prior studies provide evidence that naloxone distribution and accompanying education also are effective in reducing opioid overdose mortalityand ED visits related to adverse opioid-related events.7,8
Similarly, the guidelines provide recommendations for the use of MAT for veterans with OUD. MAT for OUD is considered a first-line treatment option for patients with moderate-to-severe OUD. When used to treat patients with unsafe opioid use, this treatment helps alleviate symptoms of withdrawal, which can increase opioid taper adherence and has a protective effect against opioid overdose mortality.9 MAT initiated in the ED can increase patient engagement to addiction services.10
These 3 CPG recommendations serve as the basis for the broad goals of the ED OSI program. We aim to develop, implement, and evaluate programs and initiatives to (aim 1) reduce inappropriate opioid prescribing from VHA EDs; (aim 2) increase naloxone distribution from VHA EDs; and (aim 3) increase access to MAT initiation from VHA EDs through the implementation of ED-based MAT-initiation programs with EDs across the VHA. Aim 1 was a focused and strategic QI effort to implement an ED-based program to reduce inappropriate opioid prescribing. The ED OSI prescribing program offered a 4-step bundled approach: (1) sharing of opioid prescribing dashboard data with ED medical director and academic detailer; (2) education of ED providers and implementation of toolkit resources; (3) academic detailers conduct audit and feedback session(s) with highest prescribers; and (4) quarterly reports of opioid prescribing data to ED providers.
Results from the pilot suggested that our program was associated with accelerating the rate at which ED prescribing rates decreased.11 In addition, the pilot found that ED-based QI initiatives in VHA facilities are a feasible practice. As we work to develop and implement the next 2 phases of the QI program, a major consideration is to identify facilitators and address any existing barriers to the implementation of naloxone distribution (aim 2) and MAT-initiation (aim 3) programs for treatment-naïve patients from VHA EDs. To date, there have been no recent published studies examining the barriers and facilitators to use or implementation of MAT initiation or naloxone distribution in VHA facilities or, more specifically, from VHA EDs.12 As part of our QI program, we set out to better understand VHA ED provider perceptions of barriers and facilitators to implementation of programs aimed at increasing naloxone distribution and initiation of MAT for treatment-naïve patients in the ED.
Methods
This project received a QI designation from the Office of PBM Academic Detailing Service Institutional Review Board at the Edward Hines, Jr. Veterans Affairs Hospital VA Medical Center (VAMC). This designation was reviewed and approved by the Rocky Mountain Regional VAMC Research and Development service. In addition, we received national union approval to disseminate this survey nationally across all VA Integrated Service Networks (VISNs).
Survey
We worked with VHA subject matter experts, key stakeholders, and the VA Collaborative Evaluation Center (VACE) to develop the survey. Subject matter experts and stakeholders included VHA emergency medicine leadership, ADS leadership, and mental health and substance treatment providers. VACE is an interdisciplinary group of mixed-method researchers. The survey questions aimed to capture perceptions and experiences regarding naloxone distribution and new MAT initiation of VHA ED/UCC providers.
We used a variety of survey question formats. Close-ended questions with a predefined list of answer options were used to capture discrete domains, such as demographic information, comfort level, and experience level. To capture health care provider (HCP) perceptions on barriers and facilitators, we used multiple-answer multiple-choice questions. Built into this question format was a free-response option, which allowed respondents to offer additional barriers or facilitators. Respondents also had the option of not answering individual questions.
We identified physicians, nurse practitioners (NPs), and physician assistants (PAs) who saw at least 100 patients in the ED or UCC in at least one 3-month period in the prior year and obtained an email address for each. In total, 2228 ED or UCC providers across 132 facilities were emailed a survey; 1883 (84.5%) were ED providers and 345 (15.5%) were UCC providers.
We used Research Electronic Data Capture (REDCap) software to build and disseminate the survey via email. Surveys were initially disseminated in late January 2019. During the 3-month survey period, recipients received 3 automated email reminders from REDCap to complete the survey. Survey data were exported from REDCap. Results were analyzed using descriptive statistics analyses with Microsoft Excel.
Results
One respondent received the survey in error and was excluded from the analysis. The survey response rate was 16.7%: 372 responses from 103 unique facilities. Each VISN had a mean 20 respondents. The majority of respondents (n = 286, 76.9%) worked in highly complex level 1 facilities characterized by high patient volume and more high-risk patients and were teaching and research facilities. Respondents were asked to describe their most recent ED or UCC role. While 281 respondents (75.5%) were medical doctors, 61 respondents (16.4%) were NPs, 30 (8.1%) were PAs, and 26 (7.0%) were ED/UCC chiefs or medical directors (Table 1). Most respondents (80.4%) reported at least 10 years of health care experience.
The majority of respondents (72.9%) believed that HCPs at their VHA facility should be prescribing naloxone. When asked to specify which HCPs should be prescribing naloxone, most HCP respondents selected pharmacists (76.4%) and substance abuse providers (71.6%). Less than half of respondents (45.0%) felt that VA ED/UCC providers also should be prescribing naloxone. However, 58.1% of most HCP respondents reported being comfortable or very comfortable with prescribing naloxone to a patient in the ED or UCC who already had an existing prescription of opioids. Similarly, 52.7% of respondents reported being comfortable or very comfortable with coprescribing naloxone when discharging a patient with an opioid prescription from the ED/UCC. Notably, while 36.7% of PAs reported being comfortable/very comfortable coprescribing naloxone, 46.7% reported being comfortable/very comfortable prescribing naloxone to a patient with an existing opioid prescription. Physicians and NPs expressed similar levels of comfort with coprescribing and prescribing naloxone.
Respondents across provider types indicated a number of barriers to prescribing naloxone to medically appropriate patients (Table 2). Many respondents indicated prescribing naloxone was beyond the ED/UCC provider scope of practice (35.2%), followed by the perceived stigma associated with naloxone (33.3%), time required to prescribe naloxone (23.9%), and concern with patient’s ability to use naloxone (22.8%).
Facilitators for prescribing naloxone to medically appropriate patients identified by HCP respondents included pharmacist help and education (44.6%), patient knowledge of medication options (31.7%), societal shift away from opioids for pain management (28.0%), facility leadership (26.9%), and patient interest in safe opioid usage (26.6%) (Table 3). In addition, NPs specifically endorsed
Less than 6.8% of HCP respondents indicated that they were comfortable using MAT. Meanwhile, 42.1% of respondents reported being aware of MAT but not familiar with it, and 23.5% reported that they were unaware of MAT. Correspondingly, 301 of the 372 (88.5%) HCP respondents indicated that they had not prescribed MAT in the past year. Across HCP types, only 24.1% indicated that it is the role of VA ED or UCC providers to prescribe MAT when medically appropriate and subsequently refer patients to substance abuse treatment for follow-up (just 7.1% of PAs endorsed this). Furthermore, 6.5% and 18.8% of HCP respondents indicated that their facility leadership was very supportive and supportive, respectively, of MAT for OUD prescribing.
Barriers to MAT initiation indicated by HCP respondents included limited scope of ED and UCC practice (53.2%), unclear follow-up/referral process (50.3%), time (29.8%), and discomfort (28.2%). Nearly one-third of NPs (27.9%) identified patient willingness/ability as a barrier to MAT initiation (Table 4).
Facilitators of MAT initiation in the ED or UCC included VHA same-day treatment options (34.9%), patient desire (32.5%), pharmacist help/education (27.4%), and psychiatric social workers in the ED or UCC (25.3%). Some NPs (23.0%) and PAs (26.7%) also indicated that having time to educate veterans about the medication would be a facilitator (Table 5). Facility leadership support was considered a facilitator by 30% of PAs.
Discussion
To the best of our knowledge, there have not been any studies examining HCP perceptions of the barriers and facilitators to naloxone distribution or the initiation of MAT in VHA ED and UCCs. Veterans are at an increased risk of overdose when compared with the general population, and increasing access to opioid safety measures (eg, safer prescribing practices, naloxone distribution) and treatment with MAT for OUD across all clinical settings has been a VHA priority.3
National guidance from VHA leadership, the Centers for Disease Control and Prevention (CDC), the US Surgeon General, and the US Department of Health and Human Services (HHS) call for an all-hands-on-deck approach to combatting opioid overdose with naloxone distribution or MAT (such as buprenorphine) initiation.13 VHA ED and UCC settings provide acute outpatient care to patients with medical or psychiatric illnesses or injuries that the patient believes requires emergent or immediate medical attention or for which there is a critical need for treatment to prevent deterioration of the condition or the possible impairment of recovery.14 However, ED and UCC environments are often regarded as settings meant to stabilize a patient until they can be seen by a primary care or long-term care provider.
A major barrier identified by HCPs was that MAT for OUD was outside their ED/UCC scope of practice, which suggests a need for a top-down or peer-to-peer reexamination of the role of HCPs in ED/UCC settings. Any naloxone distribution and/or MAT-initiation program in VHA ED/UCCs should consider education about the role of ED/UCC HCPs in opioid safety and treatment.
Only 25.3% of HCPs reported that their facility leadership was supportive or very supportive of MAT prescribing. This suggests that facility leadership should be engaged in any efforts to implement a MAT-initiation program in the facility’s ED. Engaging leadership in efforts to implement ED-based MAT programs will allow for a better understanding of leadership goals as related to opioid safety and an opportunity to address concerns regarding prescribing MAT in the ED. We recommend engaging facility leadership early in MAT implementation efforts. Respectively, 12.4% and 28.2% of HCP respondents reported discomfort prescribing naloxone or using MAT, suggesting a need for more education. Similarly, only 6.8% of HCPs reported comfort with using MAT.
A consideration for implementing ED/UCC-based MAT should be the inclusion of a training component. An evidence-based clinical treatment pathway that is appropriate to the ED/UCC setting and facility on the administration of MAT also could be beneficial. A clinical treatment pathway that includes ED/UCC-initiated discharge recommendations would address HCP concerns of unclear follow-up plans and system for referral of care. To this end, a key implementation task is coordinating with other outpatient services (eg, pain management clinic, substance use disorder treatment clinic) equipped for long-term patient follow-up to develop a system for referral of care. For example, as part of the clinical treatment pathway, an ED can develop a system of referral for patients initiated on MAT in the ED in which patients are referred for follow-up at the facility’s substance use disorder treatment clinic to be seen within 72 hours to continue the administration of MAT (such as buprenorphine).
In addition to HCP education, results suggest that patient/veteran education regarding naloxone and/or MAT should be considered. HCPs indicated that having help from a pharmacist to educate the patient about the medications would be a facilitator to naloxone distribution and MAT initiation. Similarly, patient knowledge of the medications also was endorsed as a facilitator. As such, a consideration for any future ED/UCC-based naloxone distribution or MAT-initiation programs in the VHA should be patient education whether by a clinically trained professional or an educational campaign for veterans.
Expanded naloxone distribution and initiation of MAT for OUD for EDs/UCCs across the VHA could impact the lives of veterans on long-term opioid therapy, with OUD, or who are otherwise at risk for opioid overdose. Steps taken to address the barriers and leverage the facilitators identified by HCP respondents can greatly reduce current obstacles to widespread implementation of ED/UCC-based naloxone distribution and MAT initiation nationally within the VHA.
Limitations
This survey had a low response rate (16.7%). One potential explanation for the low response rate is that when the survey was deployed, many of the VHA ED/UCC physicians were per-diem employees. Per-diem physicians may be less engaged and aware of site facilitators or barriers to naloxone and MAT prescribing. This, too, may have potentially skewed the collected data. However, the survey did not ask HCPs to disclose their employment status; thus, exact rates of per diem respondents are unknown.
We aimed to capture only self-perceived barriers to prescribing naloxone and MAT in the ED, but we did not capture or measure HCP respondent’s actual prescribing rates of MAT or naloxone. Understanding HCP perceptions of naloxone distribution and MAT initiation in the ED may have been further informed by comparing HCP responses to their actual clinical practice as related to their prescribing of these medications. In future research, we will link HCPs with the actual numbers of naloxone and MAT medications prescribed. Additionally, we do not know how many of these barriers or proposed facilitators will impact clinical practice.
Conclusions
A key aim for VHA leadership is to increase veteran access to naloxone distribution and MAT for OUD across clinical areas. The present study aimed to identify HCP perceptions of barriers and facilitators to the naloxone distribution and MAT-initiation programs in VHA ED/UCCs to inform the development of a targeted QI program to implement these opioid safety measures. Although the survey yielded a low response rate, results allowed us to identify important action items for our QI program, such as the development of clear protocols, follow-up plans, and systems for referral of care and HCP educational materials related to MAT and naloxone. We hope this work will serve as the basis for ED/UCC-tailored programs that can provide customized educational programs for HCPs designed to overcome known barriers to naloxone and MAT initiation.
Acknowledgments
This work was supported by the VA Office of Specialty Care Services 10P11 and through funding provided by the Comprehensive Addiction and Recovery Act (CARA).
The United States is facing an opioid crisis in which approximately 10 million people have misused opioids in the past year, and an estimated 2 million people have an opioid use disorder (OUD).1 Compared with the general population, veterans treated in the Veterans Health Administration (VHA) facilities are at nearly twice the risk for accidental opioid overdose.2 The implementation of opioid safety measures in VHA facilities across all care settings is a priority in addressing this public health crisis. Hence, VHA leadership is working to minimize veteran risk of fatal opioid overdoses and to increase veteran access to medication-assisted treatments (MAT) for OUD.3
Since the administration of our survey, the VHA has shifted to using the term medication for opioid use disorder (MOUD) instead of MAT for OUD. However, for consistency with the survey we distributed, we use MAT in this analysis.
Acute care settings represent an opportunity to offer appropriate opioid care and treatment options to patients at risk for OUD or opioid-related overdose. VHA facilities offer 2 outpatient acute care settings for emergent ambulatory care: emergency departments (EDs) and urgent care centers (UCCs). Annually, these settings see an estimated 2.5 million patients each year, making EDs and UCCs critical access points of OUD care for veterans. Partnering with key national VHA stakeholders from Pharmacy Benefits Management (PBM), the Office of Emergency Medicine, and Academic Detailing Services (ADS), we developed the Emergency Department Opioid Safety Initiative (ED OSI) aimed at implementing and evaluating opioid safety measures in VHA outpatient acute care settings.
The US Department of Veterans Affairs (VA)/Department of Defense (DoD) Clinical Practice Guidelines for Opioid Therapy for Chronic Pain (CPG) makes recommendations for the initiation and continuation of opioids, risk mitigation, taper of opioids, and opioid therapy for acute pain in VHA facilities.4 Using these recommendations, we developed the broad aims of the ED OSI quality improvement (QI) program. The CPG is clear about the prioritization of safe opioid prescribing practices. New opioid prescriptions written in the ED have been associated with continued and chronic opioid use.5 At the time of prescription, patients not currently and chronically on opioids who receive more than a 3-day supply are at increased risk of becoming long-term opioid users.6 Given the annual volume of patients seen, VHA ED/UCCs are a crucial area for implementing better opioid prescribing practices.
The CPG also includes recommendations for the prescribing or coprescribing of naloxone rescue kits. The administration of naloxone following opioid overdose has been found to be an effective measure against fatal overdose. Increasing provider awareness of common risk factors for opioid-related overdose (eg, frequent ED visits or hospitalizations) helps facilitate a discussion on naloxone prescribing at discharge. Prior studies provide evidence that naloxone distribution and accompanying education also are effective in reducing opioid overdose mortalityand ED visits related to adverse opioid-related events.7,8
Similarly, the guidelines provide recommendations for the use of MAT for veterans with OUD. MAT for OUD is considered a first-line treatment option for patients with moderate-to-severe OUD. When used to treat patients with unsafe opioid use, this treatment helps alleviate symptoms of withdrawal, which can increase opioid taper adherence and has a protective effect against opioid overdose mortality.9 MAT initiated in the ED can increase patient engagement to addiction services.10
These 3 CPG recommendations serve as the basis for the broad goals of the ED OSI program. We aim to develop, implement, and evaluate programs and initiatives to (aim 1) reduce inappropriate opioid prescribing from VHA EDs; (aim 2) increase naloxone distribution from VHA EDs; and (aim 3) increase access to MAT initiation from VHA EDs through the implementation of ED-based MAT-initiation programs with EDs across the VHA. Aim 1 was a focused and strategic QI effort to implement an ED-based program to reduce inappropriate opioid prescribing. The ED OSI prescribing program offered a 4-step bundled approach: (1) sharing of opioid prescribing dashboard data with ED medical director and academic detailer; (2) education of ED providers and implementation of toolkit resources; (3) academic detailers conduct audit and feedback session(s) with highest prescribers; and (4) quarterly reports of opioid prescribing data to ED providers.
Results from the pilot suggested that our program was associated with accelerating the rate at which ED prescribing rates decreased.11 In addition, the pilot found that ED-based QI initiatives in VHA facilities are a feasible practice. As we work to develop and implement the next 2 phases of the QI program, a major consideration is to identify facilitators and address any existing barriers to the implementation of naloxone distribution (aim 2) and MAT-initiation (aim 3) programs for treatment-naïve patients from VHA EDs. To date, there have been no recent published studies examining the barriers and facilitators to use or implementation of MAT initiation or naloxone distribution in VHA facilities or, more specifically, from VHA EDs.12 As part of our QI program, we set out to better understand VHA ED provider perceptions of barriers and facilitators to implementation of programs aimed at increasing naloxone distribution and initiation of MAT for treatment-naïve patients in the ED.
Methods
This project received a QI designation from the Office of PBM Academic Detailing Service Institutional Review Board at the Edward Hines, Jr. Veterans Affairs Hospital VA Medical Center (VAMC). This designation was reviewed and approved by the Rocky Mountain Regional VAMC Research and Development service. In addition, we received national union approval to disseminate this survey nationally across all VA Integrated Service Networks (VISNs).
Survey
We worked with VHA subject matter experts, key stakeholders, and the VA Collaborative Evaluation Center (VACE) to develop the survey. Subject matter experts and stakeholders included VHA emergency medicine leadership, ADS leadership, and mental health and substance treatment providers. VACE is an interdisciplinary group of mixed-method researchers. The survey questions aimed to capture perceptions and experiences regarding naloxone distribution and new MAT initiation of VHA ED/UCC providers.
We used a variety of survey question formats. Close-ended questions with a predefined list of answer options were used to capture discrete domains, such as demographic information, comfort level, and experience level. To capture health care provider (HCP) perceptions on barriers and facilitators, we used multiple-answer multiple-choice questions. Built into this question format was a free-response option, which allowed respondents to offer additional barriers or facilitators. Respondents also had the option of not answering individual questions.
We identified physicians, nurse practitioners (NPs), and physician assistants (PAs) who saw at least 100 patients in the ED or UCC in at least one 3-month period in the prior year and obtained an email address for each. In total, 2228 ED or UCC providers across 132 facilities were emailed a survey; 1883 (84.5%) were ED providers and 345 (15.5%) were UCC providers.
We used Research Electronic Data Capture (REDCap) software to build and disseminate the survey via email. Surveys were initially disseminated in late January 2019. During the 3-month survey period, recipients received 3 automated email reminders from REDCap to complete the survey. Survey data were exported from REDCap. Results were analyzed using descriptive statistics analyses with Microsoft Excel.
Results
One respondent received the survey in error and was excluded from the analysis. The survey response rate was 16.7%: 372 responses from 103 unique facilities. Each VISN had a mean 20 respondents. The majority of respondents (n = 286, 76.9%) worked in highly complex level 1 facilities characterized by high patient volume and more high-risk patients and were teaching and research facilities. Respondents were asked to describe their most recent ED or UCC role. While 281 respondents (75.5%) were medical doctors, 61 respondents (16.4%) were NPs, 30 (8.1%) were PAs, and 26 (7.0%) were ED/UCC chiefs or medical directors (Table 1). Most respondents (80.4%) reported at least 10 years of health care experience.
The majority of respondents (72.9%) believed that HCPs at their VHA facility should be prescribing naloxone. When asked to specify which HCPs should be prescribing naloxone, most HCP respondents selected pharmacists (76.4%) and substance abuse providers (71.6%). Less than half of respondents (45.0%) felt that VA ED/UCC providers also should be prescribing naloxone. However, 58.1% of most HCP respondents reported being comfortable or very comfortable with prescribing naloxone to a patient in the ED or UCC who already had an existing prescription of opioids. Similarly, 52.7% of respondents reported being comfortable or very comfortable with coprescribing naloxone when discharging a patient with an opioid prescription from the ED/UCC. Notably, while 36.7% of PAs reported being comfortable/very comfortable coprescribing naloxone, 46.7% reported being comfortable/very comfortable prescribing naloxone to a patient with an existing opioid prescription. Physicians and NPs expressed similar levels of comfort with coprescribing and prescribing naloxone.
Respondents across provider types indicated a number of barriers to prescribing naloxone to medically appropriate patients (Table 2). Many respondents indicated prescribing naloxone was beyond the ED/UCC provider scope of practice (35.2%), followed by the perceived stigma associated with naloxone (33.3%), time required to prescribe naloxone (23.9%), and concern with patient’s ability to use naloxone (22.8%).
Facilitators for prescribing naloxone to medically appropriate patients identified by HCP respondents included pharmacist help and education (44.6%), patient knowledge of medication options (31.7%), societal shift away from opioids for pain management (28.0%), facility leadership (26.9%), and patient interest in safe opioid usage (26.6%) (Table 3). In addition, NPs specifically endorsed
Less than 6.8% of HCP respondents indicated that they were comfortable using MAT. Meanwhile, 42.1% of respondents reported being aware of MAT but not familiar with it, and 23.5% reported that they were unaware of MAT. Correspondingly, 301 of the 372 (88.5%) HCP respondents indicated that they had not prescribed MAT in the past year. Across HCP types, only 24.1% indicated that it is the role of VA ED or UCC providers to prescribe MAT when medically appropriate and subsequently refer patients to substance abuse treatment for follow-up (just 7.1% of PAs endorsed this). Furthermore, 6.5% and 18.8% of HCP respondents indicated that their facility leadership was very supportive and supportive, respectively, of MAT for OUD prescribing.
Barriers to MAT initiation indicated by HCP respondents included limited scope of ED and UCC practice (53.2%), unclear follow-up/referral process (50.3%), time (29.8%), and discomfort (28.2%). Nearly one-third of NPs (27.9%) identified patient willingness/ability as a barrier to MAT initiation (Table 4).
Facilitators of MAT initiation in the ED or UCC included VHA same-day treatment options (34.9%), patient desire (32.5%), pharmacist help/education (27.4%), and psychiatric social workers in the ED or UCC (25.3%). Some NPs (23.0%) and PAs (26.7%) also indicated that having time to educate veterans about the medication would be a facilitator (Table 5). Facility leadership support was considered a facilitator by 30% of PAs.
Discussion
To the best of our knowledge, there have not been any studies examining HCP perceptions of the barriers and facilitators to naloxone distribution or the initiation of MAT in VHA ED and UCCs. Veterans are at an increased risk of overdose when compared with the general population, and increasing access to opioid safety measures (eg, safer prescribing practices, naloxone distribution) and treatment with MAT for OUD across all clinical settings has been a VHA priority.3
National guidance from VHA leadership, the Centers for Disease Control and Prevention (CDC), the US Surgeon General, and the US Department of Health and Human Services (HHS) call for an all-hands-on-deck approach to combatting opioid overdose with naloxone distribution or MAT (such as buprenorphine) initiation.13 VHA ED and UCC settings provide acute outpatient care to patients with medical or psychiatric illnesses or injuries that the patient believes requires emergent or immediate medical attention or for which there is a critical need for treatment to prevent deterioration of the condition or the possible impairment of recovery.14 However, ED and UCC environments are often regarded as settings meant to stabilize a patient until they can be seen by a primary care or long-term care provider.
A major barrier identified by HCPs was that MAT for OUD was outside their ED/UCC scope of practice, which suggests a need for a top-down or peer-to-peer reexamination of the role of HCPs in ED/UCC settings. Any naloxone distribution and/or MAT-initiation program in VHA ED/UCCs should consider education about the role of ED/UCC HCPs in opioid safety and treatment.
Only 25.3% of HCPs reported that their facility leadership was supportive or very supportive of MAT prescribing. This suggests that facility leadership should be engaged in any efforts to implement a MAT-initiation program in the facility’s ED. Engaging leadership in efforts to implement ED-based MAT programs will allow for a better understanding of leadership goals as related to opioid safety and an opportunity to address concerns regarding prescribing MAT in the ED. We recommend engaging facility leadership early in MAT implementation efforts. Respectively, 12.4% and 28.2% of HCP respondents reported discomfort prescribing naloxone or using MAT, suggesting a need for more education. Similarly, only 6.8% of HCPs reported comfort with using MAT.
A consideration for implementing ED/UCC-based MAT should be the inclusion of a training component. An evidence-based clinical treatment pathway that is appropriate to the ED/UCC setting and facility on the administration of MAT also could be beneficial. A clinical treatment pathway that includes ED/UCC-initiated discharge recommendations would address HCP concerns of unclear follow-up plans and system for referral of care. To this end, a key implementation task is coordinating with other outpatient services (eg, pain management clinic, substance use disorder treatment clinic) equipped for long-term patient follow-up to develop a system for referral of care. For example, as part of the clinical treatment pathway, an ED can develop a system of referral for patients initiated on MAT in the ED in which patients are referred for follow-up at the facility’s substance use disorder treatment clinic to be seen within 72 hours to continue the administration of MAT (such as buprenorphine).
In addition to HCP education, results suggest that patient/veteran education regarding naloxone and/or MAT should be considered. HCPs indicated that having help from a pharmacist to educate the patient about the medications would be a facilitator to naloxone distribution and MAT initiation. Similarly, patient knowledge of the medications also was endorsed as a facilitator. As such, a consideration for any future ED/UCC-based naloxone distribution or MAT-initiation programs in the VHA should be patient education whether by a clinically trained professional or an educational campaign for veterans.
Expanded naloxone distribution and initiation of MAT for OUD for EDs/UCCs across the VHA could impact the lives of veterans on long-term opioid therapy, with OUD, or who are otherwise at risk for opioid overdose. Steps taken to address the barriers and leverage the facilitators identified by HCP respondents can greatly reduce current obstacles to widespread implementation of ED/UCC-based naloxone distribution and MAT initiation nationally within the VHA.
Limitations
This survey had a low response rate (16.7%). One potential explanation for the low response rate is that when the survey was deployed, many of the VHA ED/UCC physicians were per-diem employees. Per-diem physicians may be less engaged and aware of site facilitators or barriers to naloxone and MAT prescribing. This, too, may have potentially skewed the collected data. However, the survey did not ask HCPs to disclose their employment status; thus, exact rates of per diem respondents are unknown.
We aimed to capture only self-perceived barriers to prescribing naloxone and MAT in the ED, but we did not capture or measure HCP respondent’s actual prescribing rates of MAT or naloxone. Understanding HCP perceptions of naloxone distribution and MAT initiation in the ED may have been further informed by comparing HCP responses to their actual clinical practice as related to their prescribing of these medications. In future research, we will link HCPs with the actual numbers of naloxone and MAT medications prescribed. Additionally, we do not know how many of these barriers or proposed facilitators will impact clinical practice.
Conclusions
A key aim for VHA leadership is to increase veteran access to naloxone distribution and MAT for OUD across clinical areas. The present study aimed to identify HCP perceptions of barriers and facilitators to the naloxone distribution and MAT-initiation programs in VHA ED/UCCs to inform the development of a targeted QI program to implement these opioid safety measures. Although the survey yielded a low response rate, results allowed us to identify important action items for our QI program, such as the development of clear protocols, follow-up plans, and systems for referral of care and HCP educational materials related to MAT and naloxone. We hope this work will serve as the basis for ED/UCC-tailored programs that can provide customized educational programs for HCPs designed to overcome known barriers to naloxone and MAT initiation.
Acknowledgments
This work was supported by the VA Office of Specialty Care Services 10P11 and through funding provided by the Comprehensive Addiction and Recovery Act (CARA).
1. Substance Abuse and Mental Health Services Administration. Key substance use and mental health indicators in the united states: results from the 2018 National Survey on Drug Use and Health. Published August 2019. Accessed August 20, 2021. https://www.samhsa.gov/data/sites/default/files/cbhsq-reports/NSDUHNationalFindingsReport2018/NSDUHNationalFindingsReport2018.pdf
2. Bohnert AS, Ilgen MA, Galea S, McCarthy JF, Blow FC. Accidental poisoning mortality among patients in the Department of Veterans Affairs Health System. Med Care. 2011;49(4):393-396. doi:10.1097/MLR.0b013e318202aa27
3. US Department of Veterans Affairs, Pharmacy Benefits Management Service. Recommendations for issuing naloxone rescue for the VA opioid overdose education and naloxone distribution (OEND) program. Published August 2016. Accessed August 20, 2021. https://www.pbm.va.gov/PBM/clinicalguidance/clinicalrecommendations/Naloxone_HCl_Rescue_Kits_Recommendations_for_Use.pdf
4. US Department of Defense, US Department of Veterans Affairs, Opioid Therapy for Chronic Pain Work Group. VA/DoD clinical practice guideline for opioid therapy for chronic pain. Published February 2017. Accessed August 20, 2021. https://www.va.gov/HOMELESS/nchav/resources/docs/mental-health/substance-abuse/VA_DoD-CLINICAL-PRACTICE-GUIDELINE-FOR-OPIOID-THERAPY-FOR-CHRONIC-PAIN-508.pdf
5. Barnett ML, Olenski AR, Jena AB. Opioid-prescribing patterns of emergency physicians and risk of long-term use. N Engl J Med. 2017;376(7):663-673. doi:10.1056/NEJMsa1610524
6. Shah A, Hayes CJ, Martin BC. Characteristics of initial prescription episodes and likelihood of long-term opioid use - United States, 2006-2015. MMWR Morb Mortal Wkly Rep. 2017;66(10):265-269. Published 2017 Mar 17. doi:10.15585/mmwr.mm6610a1
7. Clark AK, Wilder CM, Winstanley EL. A systematic review of community opioid overdose prevention and naloxone distribution programs. J Addict Med. 2014;8(3):153-163. doi:10.1097/ADM.0000000000000034
8. Coffin PO, Behar E, Rowe C, et al. Nonrandomized intervention study of naloxone coprescription for primary care patients receiving long-term opioid therapy for Pain. Ann Intern Med. 2016;165(4):245-252. doi:10.7326/M15-2771
9. Ma J, Bao YP, Wang RJ, et al. Effects of medication-assisted treatment on mortality among opioids users: a systematic review and meta-analysis. Mol Psychiatry. 2019;24(12):1868-1883. doi:10.1038/s41380-018-0094-5
10. D’Onofrio G, O’Connor PG, Pantalon MV, et al. Emergency department-initiated buprenorphine/naloxone treatment for opioid dependence: a randomized clinical trial. JAMA. 2015;313(16):1636-1644. doi:10.1001/jama.2015.3474
11. Dieujuste N, Johnson-Koenke R, Christopher M, et al. Feasibility study of a quasi-experimental regional opioid safety prescribing program in Veterans Health Administration emergency departments. Acad Emerg Med. 2020;27(8):734-741. doi:10.1111/acem.13980
12. Mackey K, Veazie S, Anderson J, Bourne D, Peterson K. Evidence brief: barriers and facilitators to use of medications for opioid use disorder. Published July 2017. Accessed August 20, 2021. http://www.ncbi.nlm.nih.gov/books/NBK549203/
13. US Department of Health and Human Services, Office of the Surgeon General. Naloxone: the opioid reversal drug that saves lives. Published December 2018. Accessed August 20, 2021. https://www.hhs.gov/opioids/sites/default/files/2018-12/naloxone-coprescribing-guidance.pdf
14. US Department of Veterans Affairs, Veterans Health Administration. Chapter 256: Emergency department (ED) and urgent care clinic (UCC). Updated October 3, 2016. Accessed August 20, 2021. https://www.cfm.va.gov/til/space/spChapter256.pdf.
1. Substance Abuse and Mental Health Services Administration. Key substance use and mental health indicators in the united states: results from the 2018 National Survey on Drug Use and Health. Published August 2019. Accessed August 20, 2021. https://www.samhsa.gov/data/sites/default/files/cbhsq-reports/NSDUHNationalFindingsReport2018/NSDUHNationalFindingsReport2018.pdf
2. Bohnert AS, Ilgen MA, Galea S, McCarthy JF, Blow FC. Accidental poisoning mortality among patients in the Department of Veterans Affairs Health System. Med Care. 2011;49(4):393-396. doi:10.1097/MLR.0b013e318202aa27
3. US Department of Veterans Affairs, Pharmacy Benefits Management Service. Recommendations for issuing naloxone rescue for the VA opioid overdose education and naloxone distribution (OEND) program. Published August 2016. Accessed August 20, 2021. https://www.pbm.va.gov/PBM/clinicalguidance/clinicalrecommendations/Naloxone_HCl_Rescue_Kits_Recommendations_for_Use.pdf
4. US Department of Defense, US Department of Veterans Affairs, Opioid Therapy for Chronic Pain Work Group. VA/DoD clinical practice guideline for opioid therapy for chronic pain. Published February 2017. Accessed August 20, 2021. https://www.va.gov/HOMELESS/nchav/resources/docs/mental-health/substance-abuse/VA_DoD-CLINICAL-PRACTICE-GUIDELINE-FOR-OPIOID-THERAPY-FOR-CHRONIC-PAIN-508.pdf
5. Barnett ML, Olenski AR, Jena AB. Opioid-prescribing patterns of emergency physicians and risk of long-term use. N Engl J Med. 2017;376(7):663-673. doi:10.1056/NEJMsa1610524
6. Shah A, Hayes CJ, Martin BC. Characteristics of initial prescription episodes and likelihood of long-term opioid use - United States, 2006-2015. MMWR Morb Mortal Wkly Rep. 2017;66(10):265-269. Published 2017 Mar 17. doi:10.15585/mmwr.mm6610a1
7. Clark AK, Wilder CM, Winstanley EL. A systematic review of community opioid overdose prevention and naloxone distribution programs. J Addict Med. 2014;8(3):153-163. doi:10.1097/ADM.0000000000000034
8. Coffin PO, Behar E, Rowe C, et al. Nonrandomized intervention study of naloxone coprescription for primary care patients receiving long-term opioid therapy for Pain. Ann Intern Med. 2016;165(4):245-252. doi:10.7326/M15-2771
9. Ma J, Bao YP, Wang RJ, et al. Effects of medication-assisted treatment on mortality among opioids users: a systematic review and meta-analysis. Mol Psychiatry. 2019;24(12):1868-1883. doi:10.1038/s41380-018-0094-5
10. D’Onofrio G, O’Connor PG, Pantalon MV, et al. Emergency department-initiated buprenorphine/naloxone treatment for opioid dependence: a randomized clinical trial. JAMA. 2015;313(16):1636-1644. doi:10.1001/jama.2015.3474
11. Dieujuste N, Johnson-Koenke R, Christopher M, et al. Feasibility study of a quasi-experimental regional opioid safety prescribing program in Veterans Health Administration emergency departments. Acad Emerg Med. 2020;27(8):734-741. doi:10.1111/acem.13980
12. Mackey K, Veazie S, Anderson J, Bourne D, Peterson K. Evidence brief: barriers and facilitators to use of medications for opioid use disorder. Published July 2017. Accessed August 20, 2021. http://www.ncbi.nlm.nih.gov/books/NBK549203/
13. US Department of Health and Human Services, Office of the Surgeon General. Naloxone: the opioid reversal drug that saves lives. Published December 2018. Accessed August 20, 2021. https://www.hhs.gov/opioids/sites/default/files/2018-12/naloxone-coprescribing-guidance.pdf
14. US Department of Veterans Affairs, Veterans Health Administration. Chapter 256: Emergency department (ED) and urgent care clinic (UCC). Updated October 3, 2016. Accessed August 20, 2021. https://www.cfm.va.gov/til/space/spChapter256.pdf.
Case: Patient with statin-associated muscle symptoms
A 66-year-old woman is discharged from the hospital after an MI. Her discharge medications include atorvastatin 40 mg, lisinopril 20 mg, acetylsalicylic acid 81 mg, and clopidogrel 75 mg. At this patient’s follow-up appointment, she mentions that she has muscle pain and stiffness in both legs and her back. Her labs include thyroid-stimulating hormone of 2.0 and vitamin D of 40. She stops the atorvastatin for 2 weeks with resolution of her symptoms.
Which treatment recommendation would you make for this patient?
A. Restart atorvastatin
B. Start rosuvastatin twice a week
C. Start ezetimibe
D. Start a PCSK9 inhibitor
We often see high-risk cardiovascular disease patients who are concerned about muscle side effects brought on by statins. I think we all can agree that this patient needs aggressive medical therapy for prevention of secondary cardiovascular events. I would restart her atorvastatin.
Neilsen and Nordestgaard found that early statin discontinuation rates increased from 6% in 1995 to 18% in 2010.1
Early statin discontinuation correlated with negative statin-related news stories, their paper states. This suggests either an increased awareness of side effects or a possible nocebo effect.
Statin rechallenge results
Joy and colleagues reported the results on eight patients who had developed myalgias within 3 weeks of starting a statin. These patients, who received placebo or statin, completed an N-of-1 trial with three double-blind, crossover comparisons separated by 3-week washout periods.
Patients were evaluated pain on a visual analog scale (VAS). For each N-of-1 trial there was no statistically significant difference in pain or myalgia score between those who took statin and placebo. Five of the eight patients chose to continue on statins at the end of the trial.
Herrett and colleagues performed a more extensive series of N-of-1 trials involving 200 patients who had stopped or were considering stopping statins because of muscle symptoms.3 Participants either received 2 months of atorvastatin 20 mg or placebo for 2-month blocks six times. They rated their muscle symptoms on a VAS at the end of each block. There was no difference in muscle symptom scores between the statin and placebo periods.
Wood and colleagues took it a step further, when they studied an N-of-1 trial that included statin, placebo, and no treatment.4 Each participant received four bottles of atorvastatin 20 mg, four bottles of placebo, and four empty bottles. Each month they used treatment from the bottles based on random sequence and reported daily symptom scores. The mean symptom intensity was 8.0 during no-tablet months, 15.4 during placebo months (P < .001, compared with no-tablet months), and 16.3 during statin months (P < .001, compared with no-tablet months; P = .39, compared with placebo).
Taylor and colleagues studied 120 patients who had prior statin-associated muscle complaints.5 Each patient received either simvastatin 20 mg or placebo for 4 weeks, and then were switched for an additional 4 weeks. A total of 43 patients (36%) had pain on simvastatin but not placebo, 21 (17%) had no pain with either treatment, 21 (17%) reported pain with both treatments, and 35 (29%) had pain with placebo but not simvastatin. These studies support the concept of nocebo effect in patients who have muscle symptoms on statins.
So what should be done? Brennan and Roy did a retrospective study of 118 patients referred to a lipid clinic as being statin intolerant to two or more statins.6 Most of the patients were able to tolerate a statin: 71% tolerated same statin rechallenge, 53% tolerated statin switch, and 57% tolerated a nonstatin therapy.
In the Prosisa study, only 27% of patients who reported statin-associated muscle symptoms had reappearance of muscle symptoms after rechallenge with a statin.7
Research implications
Rechallenge with the same statin seems to be a reasonable first step, followed by switching to a different statin. I also share the concept of nocebo effect with my patients, and tell them I believe they have an excellent chance of tolerating the statin.
Pearl: The majority of patients with muscle symptoms while taking a statin likely have a nocebo effect, and are likely to tolerate rechallenge with the same statin.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. Nielsen SF and Nordestgaard BG. Eur Heart J. 2016;37:908-16.
2. Joy TR et al. Ann Intern Med. 2014;160:301-10.
3. Herrett E et al. BMJ. 2021 Feb 24;372:n135.
4. Wood FA et al. N Engl J Med 2020;383:2182-4.
5. Taylor BA et al. Atherosclerosis. 2017;256:100-4.
6. Brennen ET and Roy TR. Can J Card. 2017;33(5):666-73.
7. Bonaiti Fet al. Atherosclerosis. 2020;315:E13-4.
A 66-year-old woman is discharged from the hospital after an MI. Her discharge medications include atorvastatin 40 mg, lisinopril 20 mg, acetylsalicylic acid 81 mg, and clopidogrel 75 mg. At this patient’s follow-up appointment, she mentions that she has muscle pain and stiffness in both legs and her back. Her labs include thyroid-stimulating hormone of 2.0 and vitamin D of 40. She stops the atorvastatin for 2 weeks with resolution of her symptoms.
Which treatment recommendation would you make for this patient?
A. Restart atorvastatin
B. Start rosuvastatin twice a week
C. Start ezetimibe
D. Start a PCSK9 inhibitor
We often see high-risk cardiovascular disease patients who are concerned about muscle side effects brought on by statins. I think we all can agree that this patient needs aggressive medical therapy for prevention of secondary cardiovascular events. I would restart her atorvastatin.
Neilsen and Nordestgaard found that early statin discontinuation rates increased from 6% in 1995 to 18% in 2010.1
Early statin discontinuation correlated with negative statin-related news stories, their paper states. This suggests either an increased awareness of side effects or a possible nocebo effect.
Statin rechallenge results
Joy and colleagues reported the results on eight patients who had developed myalgias within 3 weeks of starting a statin. These patients, who received placebo or statin, completed an N-of-1 trial with three double-blind, crossover comparisons separated by 3-week washout periods.
Patients were evaluated pain on a visual analog scale (VAS). For each N-of-1 trial there was no statistically significant difference in pain or myalgia score between those who took statin and placebo. Five of the eight patients chose to continue on statins at the end of the trial.
Herrett and colleagues performed a more extensive series of N-of-1 trials involving 200 patients who had stopped or were considering stopping statins because of muscle symptoms.3 Participants either received 2 months of atorvastatin 20 mg or placebo for 2-month blocks six times. They rated their muscle symptoms on a VAS at the end of each block. There was no difference in muscle symptom scores between the statin and placebo periods.
Wood and colleagues took it a step further, when they studied an N-of-1 trial that included statin, placebo, and no treatment.4 Each participant received four bottles of atorvastatin 20 mg, four bottles of placebo, and four empty bottles. Each month they used treatment from the bottles based on random sequence and reported daily symptom scores. The mean symptom intensity was 8.0 during no-tablet months, 15.4 during placebo months (P < .001, compared with no-tablet months), and 16.3 during statin months (P < .001, compared with no-tablet months; P = .39, compared with placebo).
Taylor and colleagues studied 120 patients who had prior statin-associated muscle complaints.5 Each patient received either simvastatin 20 mg or placebo for 4 weeks, and then were switched for an additional 4 weeks. A total of 43 patients (36%) had pain on simvastatin but not placebo, 21 (17%) had no pain with either treatment, 21 (17%) reported pain with both treatments, and 35 (29%) had pain with placebo but not simvastatin. These studies support the concept of nocebo effect in patients who have muscle symptoms on statins.
So what should be done? Brennan and Roy did a retrospective study of 118 patients referred to a lipid clinic as being statin intolerant to two or more statins.6 Most of the patients were able to tolerate a statin: 71% tolerated same statin rechallenge, 53% tolerated statin switch, and 57% tolerated a nonstatin therapy.
In the Prosisa study, only 27% of patients who reported statin-associated muscle symptoms had reappearance of muscle symptoms after rechallenge with a statin.7
Research implications
Rechallenge with the same statin seems to be a reasonable first step, followed by switching to a different statin. I also share the concept of nocebo effect with my patients, and tell them I believe they have an excellent chance of tolerating the statin.
Pearl: The majority of patients with muscle symptoms while taking a statin likely have a nocebo effect, and are likely to tolerate rechallenge with the same statin.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. Nielsen SF and Nordestgaard BG. Eur Heart J. 2016;37:908-16.
2. Joy TR et al. Ann Intern Med. 2014;160:301-10.
3. Herrett E et al. BMJ. 2021 Feb 24;372:n135.
4. Wood FA et al. N Engl J Med 2020;383:2182-4.
5. Taylor BA et al. Atherosclerosis. 2017;256:100-4.
6. Brennen ET and Roy TR. Can J Card. 2017;33(5):666-73.
7. Bonaiti Fet al. Atherosclerosis. 2020;315:E13-4.
A 66-year-old woman is discharged from the hospital after an MI. Her discharge medications include atorvastatin 40 mg, lisinopril 20 mg, acetylsalicylic acid 81 mg, and clopidogrel 75 mg. At this patient’s follow-up appointment, she mentions that she has muscle pain and stiffness in both legs and her back. Her labs include thyroid-stimulating hormone of 2.0 and vitamin D of 40. She stops the atorvastatin for 2 weeks with resolution of her symptoms.
Which treatment recommendation would you make for this patient?
A. Restart atorvastatin
B. Start rosuvastatin twice a week
C. Start ezetimibe
D. Start a PCSK9 inhibitor
We often see high-risk cardiovascular disease patients who are concerned about muscle side effects brought on by statins. I think we all can agree that this patient needs aggressive medical therapy for prevention of secondary cardiovascular events. I would restart her atorvastatin.
Neilsen and Nordestgaard found that early statin discontinuation rates increased from 6% in 1995 to 18% in 2010.1
Early statin discontinuation correlated with negative statin-related news stories, their paper states. This suggests either an increased awareness of side effects or a possible nocebo effect.
Statin rechallenge results
Joy and colleagues reported the results on eight patients who had developed myalgias within 3 weeks of starting a statin. These patients, who received placebo or statin, completed an N-of-1 trial with three double-blind, crossover comparisons separated by 3-week washout periods.
Patients were evaluated pain on a visual analog scale (VAS). For each N-of-1 trial there was no statistically significant difference in pain or myalgia score between those who took statin and placebo. Five of the eight patients chose to continue on statins at the end of the trial.
Herrett and colleagues performed a more extensive series of N-of-1 trials involving 200 patients who had stopped or were considering stopping statins because of muscle symptoms.3 Participants either received 2 months of atorvastatin 20 mg or placebo for 2-month blocks six times. They rated their muscle symptoms on a VAS at the end of each block. There was no difference in muscle symptom scores between the statin and placebo periods.
Wood and colleagues took it a step further, when they studied an N-of-1 trial that included statin, placebo, and no treatment.4 Each participant received four bottles of atorvastatin 20 mg, four bottles of placebo, and four empty bottles. Each month they used treatment from the bottles based on random sequence and reported daily symptom scores. The mean symptom intensity was 8.0 during no-tablet months, 15.4 during placebo months (P < .001, compared with no-tablet months), and 16.3 during statin months (P < .001, compared with no-tablet months; P = .39, compared with placebo).
Taylor and colleagues studied 120 patients who had prior statin-associated muscle complaints.5 Each patient received either simvastatin 20 mg or placebo for 4 weeks, and then were switched for an additional 4 weeks. A total of 43 patients (36%) had pain on simvastatin but not placebo, 21 (17%) had no pain with either treatment, 21 (17%) reported pain with both treatments, and 35 (29%) had pain with placebo but not simvastatin. These studies support the concept of nocebo effect in patients who have muscle symptoms on statins.
So what should be done? Brennan and Roy did a retrospective study of 118 patients referred to a lipid clinic as being statin intolerant to two or more statins.6 Most of the patients were able to tolerate a statin: 71% tolerated same statin rechallenge, 53% tolerated statin switch, and 57% tolerated a nonstatin therapy.
In the Prosisa study, only 27% of patients who reported statin-associated muscle symptoms had reappearance of muscle symptoms after rechallenge with a statin.7
Research implications
Rechallenge with the same statin seems to be a reasonable first step, followed by switching to a different statin. I also share the concept of nocebo effect with my patients, and tell them I believe they have an excellent chance of tolerating the statin.
Pearl: The majority of patients with muscle symptoms while taking a statin likely have a nocebo effect, and are likely to tolerate rechallenge with the same statin.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. Nielsen SF and Nordestgaard BG. Eur Heart J. 2016;37:908-16.
2. Joy TR et al. Ann Intern Med. 2014;160:301-10.
3. Herrett E et al. BMJ. 2021 Feb 24;372:n135.
4. Wood FA et al. N Engl J Med 2020;383:2182-4.
5. Taylor BA et al. Atherosclerosis. 2017;256:100-4.
6. Brennen ET and Roy TR. Can J Card. 2017;33(5):666-73.
7. Bonaiti Fet al. Atherosclerosis. 2020;315:E13-4.