User login
Battlefield Acupuncture vs Ketorolac for Treating Pain in the Emergency Department
Acute pain is a primary symptom for many patients who present to the emergency department (ED). The ED team is challenged with relieving pain while limiting harm from medications.1 A 2017 National Health Interview Survey showed that compared with nonveterans, more veterans reported pain in the previous 3 months, and the rate of severe pain was 40% higher in the veteran group especially among those who served during the era of wars in Afghanistan and Iraq.2
The American College of Emergency Physicians guidelines pain management guidelines recommend patient-centered shared decision making that includes patient education about treatment goals and expectations, and short- and long-term risks, as well as a preference toward pharmacologic treatment with nonopioid analgesics except for patients with severe pain or pain refractory to other drug and treatment modalities.3 There is a lack of evidence regarding superior efficacy of either opioid or nonopioid analgesics; therefore, the use of nonopioid analgesics, such as oral or topical nonsteroidal anti-inflammatory drugs (NSAIDs) or central analgesics, such as acetaminophen, is preferred for treating acute pain to mitigate adverse effects (AEs) and risks associated with opioid use.1,3,4 The US Department of Veterans Affairs (VA) and Department of Defense (DoD) guideline on managing opioid therapy for chronic pain, updated in 2017 and 2022, similarly recommends alternatives to opioids for mild-to-moderate acute pain and encourages multimodal pain care.5 However, use of other pharmacologic treatments, such as NSAIDs, is limited by AE profiles, patient contraindications, and severity of acute pain etiologies. There is a need for the expanded use of nonpharmacologic treatments for addressing pain in the veteran population.
The American College of Emergency Physicians guidelines recommend nonpharmacologic modalities, such as applying heat or cold, physical therapy, cognitive behavioral therapy, and acupuncture.3 A 2014 study reported that 37% to 46% of active duty and reserve military personnel use complementary and alternative medicine (CAM) for a variety of ailments, and there is increasing interest in the use of CAM as adjuncts to traditional therapies.6 According to one study, some CAM therapies are used significantly more by military personnel than used by civilians.7 However, the percentage of the veteran population using acupuncture in this study was small, and more information is needed to assess its use.
Auricular acupuncture originated in traditional Chinese medicine.8 Contemporary auricular acupuncture experts view this modality as a self-contained microsystem mapping portions of the ear to specific parts of the body and internal organs. The analgesic effects may be mediated through the central nervous system by local release of endorphins through nerve fiber activation and neurotransmitters—including serotonin, dopamine, and norepinephrine—leading to pre- and postsynaptic suppression of pain transmission.
Battlefield acupuncture (BFA) uses 5 set points anatomically located on each ear.9 Practitioners use small semipermanent, dartlike acupuncture needles. Patients could experience pain relief in a few minutes, which can last minutes, hours, days, weeks, or months depending on the pathology of the pain. This procedure developed in 2001 has been studied for different pain types and has shown benefit when used for postsurgical pain, chronic spinal cord injury−related neuropathic pain, and general chronic pain, as well as for other indications, such as insomnia, depression, and weight loss.8,10-13 In 2018, a randomized controlled trial compared postintervention numeric rating scale (NRS) pain scores in patients presenting to the ED with acute or acute-on-chronic lower back pain who received BFA as an adjunct to standard care vs standard care alone.14 Patients receiving BFA as an adjunct to standard care were found to have mean postintervention pain scores 1.7 points lower than those receiving standard care alone. This study demonstrated that BFA was feasible and well tolerated for lower back pain in the ED as an adjunct to standard care. The study was limited by the adjunct use of BFA rather than as monotherapy and by the practitioners’ discretion regarding standard care, which was not defined by the study’s authors.
The Jesse Brown Veterans Affairs Medical Center (JBVAMC) in Chicago, Illinois, offers several CAM modalities, such as exercise/movement therapy, chiropractic, art/music therapy, and relaxation workshops, which are widely used by veterans. Recent evidence suggests BFA could reduce pain scores as an adjunct or an alternative to pharmacologic therapy. We are interested in how CAM therapies, such as BFA, can help avoid AEs associated with opioid or NSAID therapy.
At the JBVAMC ED, ketorolac 15 mg is the preferred first-line treatment of acute, noncancer pain, based on the results of previous studies. In 2018 BFA was offered first to veterans presenting with acute or acute-on-chronic pain to the ED; however, its effectiveness for pain reduction vs ketorolac has not been evaluated in this patient population. Limited literature is available on BFA and its use in the ED. To our knowledge, this was the first observational study assessing the difference between a single session of BFA vs a single dose of ketorolac in treating noncancer acute or acute-on-chronic pain in the ED.
Methods
This study was a retrospective chart review of patients who presented to the JBVAMC ED with acute pain or acute-on-chronic pain, who received ketorolac or BFA. The study population was generated from a list of all IV and intramuscular (IM) ketorolac unit dose orders verified from June 1, 2018, through August 30, 2019, and a list of all BFA procedure notes signed from June 1, 2018, through August 30, 2019. Patients were included in the study if they had documented administration of IV or IM ketorolac or BFA between June 1, 2018, and August 30, 2019. Patients who received ketorolac doses other than 15 mg, the intervention was administered outside of the ED, received adjunct treatment in addition to the treatment intervention in the ED, had no baseline NRS pain score documented before the intervention, had an NRS pain score of < 4, had no postintervention NRS pain score documented within 6 hours, had a treatment indication other than pain, or had active cancer were excluded. As in previous JBVAMC studies, we used NRS pain score cutoffs (mild, moderate, severe, and very severe) based on Woo and colleagues’ meta-analysis and excluded scores < 4.15
Endpoints
The primary endpoint was the mean difference in NRS pain score before and after the intervention, determined by comparing the NRS pain score documented at triage to the ED with the first documented NRS pain score at least 30 minutes to 6 hours after treatment administration. The secondary endpoints included the number of patients prescribed pain medication at discharge, the number of patients who were discharged with no medications, and the number of patients admitted to the hospital. The safety endpoint included any AEs of the intervention. Subgroup analyses were performed comparing the mean difference in NRS pain score among subgroups classified by severity of baseline NRS pain score and pain location.
Statistical Analysis
Baseline characteristics and endpoints were analyzed using descriptive statistics. Categorical data were analyzed using Fisher exact test and z test for proportions, and continuous data were compared using t test and paired t test. An 80% power calculation determined that 84 patients per group were needed to detect a statistically significant difference in pain score reduction of 1.3 at a type-1 error rate of 0.05. The sample size was based on a calculation performed in a previously published study that compared IV ketorolac at 3 single-dose regimens for treating acute pain in the ED.16 The 1.3 pain score reduction is considered the minimum clinically significant difference in pain that could be detected with the NRS.17
Results
Sixty-one patients received BFA during the study period: 31 were excluded (26 received adjunct treatment in the ED, 2 had active cancer documented, 2 had an indication other than pain, and 1 received BFA outside of the ED), leaving 30 patients in the BFA cohort. During the study period, 1299 patients received ketorolac.
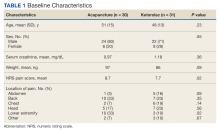
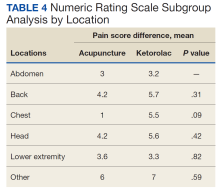
Baseline characteristics were similar between the 2 groups except for the average baseline NRS pain score, which was statistically significantly higher in the BFA vs ketorolac group (8.7 vs 7.7, respectively; P = .02). The mean age was 51 years in the BFA group and 48 years in the ketorolac group. Most patients in each cohort were male: 80% in the BFA group and 71% in the ketorolac group. The most common types of pain documented as the chief ED presentation included back, lower extremity, and head.
Endpoints
The mean difference in NRS pain score was 3.9 for the BFA group and 5.1 for the ketorolac group. Both were clinically and statistically significant reductions (P = .03 and P < .01), but the difference between the intervention groups in NRS score reduction was not statistically significant (P = .07).
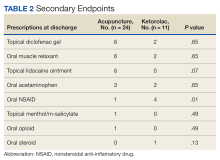
For the secondary endpoint of outpatient prescriptions written at discharge, there was no significant difference between the groups except for oral NSAIDs, which were more likely to be prescribed to patients who received ketorolac (P = .01).
Subgroup Analysis
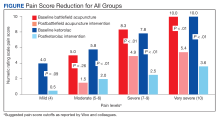
An analysis was performed for subgroups classified by baseline NRS pain score (mild: 4; moderate, 5 - 6; severe, 7 - 9; and very severe, 10). Data for mild pain was limited because a small number of patients received interventions. For moderate pain, the mean difference in NRS pain score for BFA and ketorolac was 3.5 and 3.8, respectively; for severe pain, 3.4 and 5.3; and for very severe pain, 4.6 and 6.4. There was a larger difference in the preintervention and postintervention NRS pain scores within severe pain and very severe pain groups.
Discussion
Both interventions resulted in a significant reduction in the mean NRS pain score of about 4 to 5 points within their group, and BFA resulted in a similar NRS pain score reduction compared with ketorolac 15 mg. Because the baseline NRS pain scores were significantly different between the BFA and ketorolac groups,
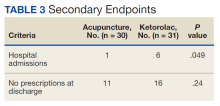
In this study, more patients in the BFA group presented to the ED with lower extremity pain, such as gout or neuropathy, compared with the ketorolac group; however, BFA did not result in a significantly different pain score reduction in this subgroup compared with ketorolac. Patients receiving BFA were more likely to receive topical analgesics or muscle relaxants at discharge; whereas those receiving ketorolac were significantly more likely to receive oral NSAIDs. Patients in this study also were more likely to be admitted to the hospital if they received ketorolac; however, for these patients, pain was secondary to their chief presentation, and the admitting physician’s familiarity with ketorolac might have been the reason for choosing this intervention. Reasons for the admissions were surgical observation, psychiatric stabilization, kidney/gallstones, rule out of acute coronary syndrome, pneumonia, and proctitis in the ketorolac group, and suicidal ideations in the BFA group.
Limitations
As a limited number of patients received BFA at JBVAMC, the study was not sufficiently powered to detect a difference in the primary outcome. Because BFA required a consultation to be entered in the electronic health record, in addition to time needed to perform the procedure, practitioners might have preferred IV/IM ketorolac during busy times in the ED, potentially leading to underrepresentation in the BFA group. Prescribing preferences might have differed among the rotating physicians, timing of the documentation of the NRS pain score could have differed based on the treatment intervention, and the investigators were unable to control or accurately assess whether patients had taken an analgesic medication before presenting to the ED.
Conclusions
NRS pain score reduction with BFA did not differ compared with ketorolac 15 mg for treating acute and acute-on-chronic pain in the ED. Although this study was underpowered, these results add to the limited existing literature, suggesting that both interventions could result in clinically significant pain score reductions for patients presenting to the ED with severe and very severe pain, making BFA a viable nonpharmacologic option. Future studies could include investigating the benefit of BFA in the veteran population by studying larger samples in the ED, surveying patients after their interventions to identify rates AEs, and exploring the use of BFA for chronic pain in the outpatient setting.
1. Cantrill SV, Brown MD, Carlisle RJ, et al. Clinical policy: critical issues in the prescribing of opioids for adult patients in the emergency department. Ann Emerg Med. 2012;60(4):499-525. doi:10.1016/j.annemergmed.2012.06.013
2. Nahin RL. Severe pain in veterans: the effect of age and sex, and comparisons with the general population. J Pain. 2017;18(3):247-254. doi:10.1016/j.jpain.2016.10.021
3. Motov S, Strayer R, Hayes BD, et al. The treatment of acute pain in the emergency department: a white paper position statement prepared for the American Academy of Emergency Medicine. J Emerg Med. 2018;54(5):731-736. doi:10.1016/j.jemermed.2018.01.020
4. Samcam I, Papa L. Acute pain management in the emergency department. In: Prostran M, ed. Pain Management. IntechOpen; 2016. doi:10.5772/62861
5. Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for the use of opioids in the management of chronic pain. Accessed February 15, 2023. https://www.healthquality.va.gov/guidelines/Pain/cot/VADoDOpioidsCPG.pdf
6. Davis MT, Mulvaney-Day N, Larson MJ, Hoover R, Mauch D. Complementary and alternative medicine among veterans and military personnel: a synthesis of population surveys. Med Care. 2014;52(12 suppl 5):S83-590. doi:10.1097/MLR.0000000000000227
7. Goertz C, Marriott BP, Finch FD, et al. Military report more complementary and alternative medicine use than civilians. J Altern Complement Med. 2013;19(6):509-517. doi:10.1089/acm.2012.0108
8. King HC, Hickey AH, Connelly C. Auricular acupuncture: a brief introduction for military providers. Mil Med. 2013;178(8):867-874. doi:10.7205/MILMED-D-13-00075
9. Niemtzow RC. Battlefield acupuncture. Medical Acupunct. 2007;19(4):225-228. doi:10.1089/acu.2007.0603
10. Collinsworth KM, Goss DL. Battlefield acupuncture and physical therapy versus physical therapy alone after shoulder surgery. Med Acupunct. 2019;31(4):228-238. doi:10.1089/acu.2019.1372
11. Estores I, Chen K, Jackson B, Lao L, Gorman PH. Auricular acupuncture for spinal cord injury related neuropathic pain: a pilot controlled clinical trial. J Spinal Cord Med. 2017;40(4):432-438. doi:10.1080/10790268.2016.1141489
12. Federman DG, Radhakrishnan K, Gabriel L, Poulin LM, Kravetz JD. Group battlefield acupuncture in primary care for veterans with pain. South Med J. 2018;111(10):619-624. doi:10.14423/SMJ.0000000000000877
13. Garner BK, Hopkinson SG, Ketz AK, Landis CA, Trego LL. Auricular acupuncture for chronic pain and insomnia: a randomized clinical trial. Med Acupunct. 2018;30(5):262-272. doi:10.1089/acu.2018.1294
14. Fox LM, Murakami M, Danesh H, Manini AF. Battlefield acupuncture to treat low back pain in the emergency department. Am J Emerg Med. 2018; 36:1045-1048. doi:10.1016/j.ajem.2018.02.038
15. Woo A, Lechner B, Fu T, et al. Cut points for mild, moderate, and severe pain among cancer and non-cancer patients: a literature review. Ann Palliat Med. 2015;4(4):176-183. doi:10.3978/j.issn.2224-5820.2015.09.04
16. Motov S, Yasavolian M, Likourezos A, et al. Comparison of intravenous ketorolac at three single-dose regimens for treating acute pain in the emergency department: a randomized controlled trial. Ann Emerg Med. 2017;70(2):177-184. doi:10.1016/j.annemergmed.2016.10.014
17. Bijur PE, Latimer CT, Gallagher EJ. Validation of a verbally administered numerical rating scale of acute pain for use in the emergency department. Acad Emerg Med. 2003;10:390-392. doi:10.1111/j.1553-2712.2003.tb01355.
Acute pain is a primary symptom for many patients who present to the emergency department (ED). The ED team is challenged with relieving pain while limiting harm from medications.1 A 2017 National Health Interview Survey showed that compared with nonveterans, more veterans reported pain in the previous 3 months, and the rate of severe pain was 40% higher in the veteran group especially among those who served during the era of wars in Afghanistan and Iraq.2
The American College of Emergency Physicians guidelines pain management guidelines recommend patient-centered shared decision making that includes patient education about treatment goals and expectations, and short- and long-term risks, as well as a preference toward pharmacologic treatment with nonopioid analgesics except for patients with severe pain or pain refractory to other drug and treatment modalities.3 There is a lack of evidence regarding superior efficacy of either opioid or nonopioid analgesics; therefore, the use of nonopioid analgesics, such as oral or topical nonsteroidal anti-inflammatory drugs (NSAIDs) or central analgesics, such as acetaminophen, is preferred for treating acute pain to mitigate adverse effects (AEs) and risks associated with opioid use.1,3,4 The US Department of Veterans Affairs (VA) and Department of Defense (DoD) guideline on managing opioid therapy for chronic pain, updated in 2017 and 2022, similarly recommends alternatives to opioids for mild-to-moderate acute pain and encourages multimodal pain care.5 However, use of other pharmacologic treatments, such as NSAIDs, is limited by AE profiles, patient contraindications, and severity of acute pain etiologies. There is a need for the expanded use of nonpharmacologic treatments for addressing pain in the veteran population.
The American College of Emergency Physicians guidelines recommend nonpharmacologic modalities, such as applying heat or cold, physical therapy, cognitive behavioral therapy, and acupuncture.3 A 2014 study reported that 37% to 46% of active duty and reserve military personnel use complementary and alternative medicine (CAM) for a variety of ailments, and there is increasing interest in the use of CAM as adjuncts to traditional therapies.6 According to one study, some CAM therapies are used significantly more by military personnel than used by civilians.7 However, the percentage of the veteran population using acupuncture in this study was small, and more information is needed to assess its use.
Auricular acupuncture originated in traditional Chinese medicine.8 Contemporary auricular acupuncture experts view this modality as a self-contained microsystem mapping portions of the ear to specific parts of the body and internal organs. The analgesic effects may be mediated through the central nervous system by local release of endorphins through nerve fiber activation and neurotransmitters—including serotonin, dopamine, and norepinephrine—leading to pre- and postsynaptic suppression of pain transmission.
Battlefield acupuncture (BFA) uses 5 set points anatomically located on each ear.9 Practitioners use small semipermanent, dartlike acupuncture needles. Patients could experience pain relief in a few minutes, which can last minutes, hours, days, weeks, or months depending on the pathology of the pain. This procedure developed in 2001 has been studied for different pain types and has shown benefit when used for postsurgical pain, chronic spinal cord injury−related neuropathic pain, and general chronic pain, as well as for other indications, such as insomnia, depression, and weight loss.8,10-13 In 2018, a randomized controlled trial compared postintervention numeric rating scale (NRS) pain scores in patients presenting to the ED with acute or acute-on-chronic lower back pain who received BFA as an adjunct to standard care vs standard care alone.14 Patients receiving BFA as an adjunct to standard care were found to have mean postintervention pain scores 1.7 points lower than those receiving standard care alone. This study demonstrated that BFA was feasible and well tolerated for lower back pain in the ED as an adjunct to standard care. The study was limited by the adjunct use of BFA rather than as monotherapy and by the practitioners’ discretion regarding standard care, which was not defined by the study’s authors.
The Jesse Brown Veterans Affairs Medical Center (JBVAMC) in Chicago, Illinois, offers several CAM modalities, such as exercise/movement therapy, chiropractic, art/music therapy, and relaxation workshops, which are widely used by veterans. Recent evidence suggests BFA could reduce pain scores as an adjunct or an alternative to pharmacologic therapy. We are interested in how CAM therapies, such as BFA, can help avoid AEs associated with opioid or NSAID therapy.
At the JBVAMC ED, ketorolac 15 mg is the preferred first-line treatment of acute, noncancer pain, based on the results of previous studies. In 2018 BFA was offered first to veterans presenting with acute or acute-on-chronic pain to the ED; however, its effectiveness for pain reduction vs ketorolac has not been evaluated in this patient population. Limited literature is available on BFA and its use in the ED. To our knowledge, this was the first observational study assessing the difference between a single session of BFA vs a single dose of ketorolac in treating noncancer acute or acute-on-chronic pain in the ED.
Methods
This study was a retrospective chart review of patients who presented to the JBVAMC ED with acute pain or acute-on-chronic pain, who received ketorolac or BFA. The study population was generated from a list of all IV and intramuscular (IM) ketorolac unit dose orders verified from June 1, 2018, through August 30, 2019, and a list of all BFA procedure notes signed from June 1, 2018, through August 30, 2019. Patients were included in the study if they had documented administration of IV or IM ketorolac or BFA between June 1, 2018, and August 30, 2019. Patients who received ketorolac doses other than 15 mg, the intervention was administered outside of the ED, received adjunct treatment in addition to the treatment intervention in the ED, had no baseline NRS pain score documented before the intervention, had an NRS pain score of < 4, had no postintervention NRS pain score documented within 6 hours, had a treatment indication other than pain, or had active cancer were excluded. As in previous JBVAMC studies, we used NRS pain score cutoffs (mild, moderate, severe, and very severe) based on Woo and colleagues’ meta-analysis and excluded scores < 4.15
Endpoints
The primary endpoint was the mean difference in NRS pain score before and after the intervention, determined by comparing the NRS pain score documented at triage to the ED with the first documented NRS pain score at least 30 minutes to 6 hours after treatment administration. The secondary endpoints included the number of patients prescribed pain medication at discharge, the number of patients who were discharged with no medications, and the number of patients admitted to the hospital. The safety endpoint included any AEs of the intervention. Subgroup analyses were performed comparing the mean difference in NRS pain score among subgroups classified by severity of baseline NRS pain score and pain location.
Statistical Analysis
Baseline characteristics and endpoints were analyzed using descriptive statistics. Categorical data were analyzed using Fisher exact test and z test for proportions, and continuous data were compared using t test and paired t test. An 80% power calculation determined that 84 patients per group were needed to detect a statistically significant difference in pain score reduction of 1.3 at a type-1 error rate of 0.05. The sample size was based on a calculation performed in a previously published study that compared IV ketorolac at 3 single-dose regimens for treating acute pain in the ED.16 The 1.3 pain score reduction is considered the minimum clinically significant difference in pain that could be detected with the NRS.17
Results
Sixty-one patients received BFA during the study period: 31 were excluded (26 received adjunct treatment in the ED, 2 had active cancer documented, 2 had an indication other than pain, and 1 received BFA outside of the ED), leaving 30 patients in the BFA cohort. During the study period, 1299 patients received ketorolac.
Baseline characteristics were similar between the 2 groups except for the average baseline NRS pain score, which was statistically significantly higher in the BFA vs ketorolac group (8.7 vs 7.7, respectively; P = .02). The mean age was 51 years in the BFA group and 48 years in the ketorolac group. Most patients in each cohort were male: 80% in the BFA group and 71% in the ketorolac group. The most common types of pain documented as the chief ED presentation included back, lower extremity, and head.
Endpoints
The mean difference in NRS pain score was 3.9 for the BFA group and 5.1 for the ketorolac group. Both were clinically and statistically significant reductions (P = .03 and P < .01), but the difference between the intervention groups in NRS score reduction was not statistically significant (P = .07).
For the secondary endpoint of outpatient prescriptions written at discharge, there was no significant difference between the groups except for oral NSAIDs, which were more likely to be prescribed to patients who received ketorolac (P = .01).
Subgroup Analysis
An analysis was performed for subgroups classified by baseline NRS pain score (mild: 4; moderate, 5 - 6; severe, 7 - 9; and very severe, 10). Data for mild pain was limited because a small number of patients received interventions. For moderate pain, the mean difference in NRS pain score for BFA and ketorolac was 3.5 and 3.8, respectively; for severe pain, 3.4 and 5.3; and for very severe pain, 4.6 and 6.4. There was a larger difference in the preintervention and postintervention NRS pain scores within severe pain and very severe pain groups.
Discussion
Both interventions resulted in a significant reduction in the mean NRS pain score of about 4 to 5 points within their group, and BFA resulted in a similar NRS pain score reduction compared with ketorolac 15 mg. Because the baseline NRS pain scores were significantly different between the BFA and ketorolac groups,
In this study, more patients in the BFA group presented to the ED with lower extremity pain, such as gout or neuropathy, compared with the ketorolac group; however, BFA did not result in a significantly different pain score reduction in this subgroup compared with ketorolac. Patients receiving BFA were more likely to receive topical analgesics or muscle relaxants at discharge; whereas those receiving ketorolac were significantly more likely to receive oral NSAIDs. Patients in this study also were more likely to be admitted to the hospital if they received ketorolac; however, for these patients, pain was secondary to their chief presentation, and the admitting physician’s familiarity with ketorolac might have been the reason for choosing this intervention. Reasons for the admissions were surgical observation, psychiatric stabilization, kidney/gallstones, rule out of acute coronary syndrome, pneumonia, and proctitis in the ketorolac group, and suicidal ideations in the BFA group.
Limitations
As a limited number of patients received BFA at JBVAMC, the study was not sufficiently powered to detect a difference in the primary outcome. Because BFA required a consultation to be entered in the electronic health record, in addition to time needed to perform the procedure, practitioners might have preferred IV/IM ketorolac during busy times in the ED, potentially leading to underrepresentation in the BFA group. Prescribing preferences might have differed among the rotating physicians, timing of the documentation of the NRS pain score could have differed based on the treatment intervention, and the investigators were unable to control or accurately assess whether patients had taken an analgesic medication before presenting to the ED.
Conclusions
NRS pain score reduction with BFA did not differ compared with ketorolac 15 mg for treating acute and acute-on-chronic pain in the ED. Although this study was underpowered, these results add to the limited existing literature, suggesting that both interventions could result in clinically significant pain score reductions for patients presenting to the ED with severe and very severe pain, making BFA a viable nonpharmacologic option. Future studies could include investigating the benefit of BFA in the veteran population by studying larger samples in the ED, surveying patients after their interventions to identify rates AEs, and exploring the use of BFA for chronic pain in the outpatient setting.
Acute pain is a primary symptom for many patients who present to the emergency department (ED). The ED team is challenged with relieving pain while limiting harm from medications.1 A 2017 National Health Interview Survey showed that compared with nonveterans, more veterans reported pain in the previous 3 months, and the rate of severe pain was 40% higher in the veteran group especially among those who served during the era of wars in Afghanistan and Iraq.2
The American College of Emergency Physicians guidelines pain management guidelines recommend patient-centered shared decision making that includes patient education about treatment goals and expectations, and short- and long-term risks, as well as a preference toward pharmacologic treatment with nonopioid analgesics except for patients with severe pain or pain refractory to other drug and treatment modalities.3 There is a lack of evidence regarding superior efficacy of either opioid or nonopioid analgesics; therefore, the use of nonopioid analgesics, such as oral or topical nonsteroidal anti-inflammatory drugs (NSAIDs) or central analgesics, such as acetaminophen, is preferred for treating acute pain to mitigate adverse effects (AEs) and risks associated with opioid use.1,3,4 The US Department of Veterans Affairs (VA) and Department of Defense (DoD) guideline on managing opioid therapy for chronic pain, updated in 2017 and 2022, similarly recommends alternatives to opioids for mild-to-moderate acute pain and encourages multimodal pain care.5 However, use of other pharmacologic treatments, such as NSAIDs, is limited by AE profiles, patient contraindications, and severity of acute pain etiologies. There is a need for the expanded use of nonpharmacologic treatments for addressing pain in the veteran population.
The American College of Emergency Physicians guidelines recommend nonpharmacologic modalities, such as applying heat or cold, physical therapy, cognitive behavioral therapy, and acupuncture.3 A 2014 study reported that 37% to 46% of active duty and reserve military personnel use complementary and alternative medicine (CAM) for a variety of ailments, and there is increasing interest in the use of CAM as adjuncts to traditional therapies.6 According to one study, some CAM therapies are used significantly more by military personnel than used by civilians.7 However, the percentage of the veteran population using acupuncture in this study was small, and more information is needed to assess its use.
Auricular acupuncture originated in traditional Chinese medicine.8 Contemporary auricular acupuncture experts view this modality as a self-contained microsystem mapping portions of the ear to specific parts of the body and internal organs. The analgesic effects may be mediated through the central nervous system by local release of endorphins through nerve fiber activation and neurotransmitters—including serotonin, dopamine, and norepinephrine—leading to pre- and postsynaptic suppression of pain transmission.
Battlefield acupuncture (BFA) uses 5 set points anatomically located on each ear.9 Practitioners use small semipermanent, dartlike acupuncture needles. Patients could experience pain relief in a few minutes, which can last minutes, hours, days, weeks, or months depending on the pathology of the pain. This procedure developed in 2001 has been studied for different pain types and has shown benefit when used for postsurgical pain, chronic spinal cord injury−related neuropathic pain, and general chronic pain, as well as for other indications, such as insomnia, depression, and weight loss.8,10-13 In 2018, a randomized controlled trial compared postintervention numeric rating scale (NRS) pain scores in patients presenting to the ED with acute or acute-on-chronic lower back pain who received BFA as an adjunct to standard care vs standard care alone.14 Patients receiving BFA as an adjunct to standard care were found to have mean postintervention pain scores 1.7 points lower than those receiving standard care alone. This study demonstrated that BFA was feasible and well tolerated for lower back pain in the ED as an adjunct to standard care. The study was limited by the adjunct use of BFA rather than as monotherapy and by the practitioners’ discretion regarding standard care, which was not defined by the study’s authors.
The Jesse Brown Veterans Affairs Medical Center (JBVAMC) in Chicago, Illinois, offers several CAM modalities, such as exercise/movement therapy, chiropractic, art/music therapy, and relaxation workshops, which are widely used by veterans. Recent evidence suggests BFA could reduce pain scores as an adjunct or an alternative to pharmacologic therapy. We are interested in how CAM therapies, such as BFA, can help avoid AEs associated with opioid or NSAID therapy.
At the JBVAMC ED, ketorolac 15 mg is the preferred first-line treatment of acute, noncancer pain, based on the results of previous studies. In 2018 BFA was offered first to veterans presenting with acute or acute-on-chronic pain to the ED; however, its effectiveness for pain reduction vs ketorolac has not been evaluated in this patient population. Limited literature is available on BFA and its use in the ED. To our knowledge, this was the first observational study assessing the difference between a single session of BFA vs a single dose of ketorolac in treating noncancer acute or acute-on-chronic pain in the ED.
Methods
This study was a retrospective chart review of patients who presented to the JBVAMC ED with acute pain or acute-on-chronic pain, who received ketorolac or BFA. The study population was generated from a list of all IV and intramuscular (IM) ketorolac unit dose orders verified from June 1, 2018, through August 30, 2019, and a list of all BFA procedure notes signed from June 1, 2018, through August 30, 2019. Patients were included in the study if they had documented administration of IV or IM ketorolac or BFA between June 1, 2018, and August 30, 2019. Patients who received ketorolac doses other than 15 mg, the intervention was administered outside of the ED, received adjunct treatment in addition to the treatment intervention in the ED, had no baseline NRS pain score documented before the intervention, had an NRS pain score of < 4, had no postintervention NRS pain score documented within 6 hours, had a treatment indication other than pain, or had active cancer were excluded. As in previous JBVAMC studies, we used NRS pain score cutoffs (mild, moderate, severe, and very severe) based on Woo and colleagues’ meta-analysis and excluded scores < 4.15
Endpoints
The primary endpoint was the mean difference in NRS pain score before and after the intervention, determined by comparing the NRS pain score documented at triage to the ED with the first documented NRS pain score at least 30 minutes to 6 hours after treatment administration. The secondary endpoints included the number of patients prescribed pain medication at discharge, the number of patients who were discharged with no medications, and the number of patients admitted to the hospital. The safety endpoint included any AEs of the intervention. Subgroup analyses were performed comparing the mean difference in NRS pain score among subgroups classified by severity of baseline NRS pain score and pain location.
Statistical Analysis
Baseline characteristics and endpoints were analyzed using descriptive statistics. Categorical data were analyzed using Fisher exact test and z test for proportions, and continuous data were compared using t test and paired t test. An 80% power calculation determined that 84 patients per group were needed to detect a statistically significant difference in pain score reduction of 1.3 at a type-1 error rate of 0.05. The sample size was based on a calculation performed in a previously published study that compared IV ketorolac at 3 single-dose regimens for treating acute pain in the ED.16 The 1.3 pain score reduction is considered the minimum clinically significant difference in pain that could be detected with the NRS.17
Results
Sixty-one patients received BFA during the study period: 31 were excluded (26 received adjunct treatment in the ED, 2 had active cancer documented, 2 had an indication other than pain, and 1 received BFA outside of the ED), leaving 30 patients in the BFA cohort. During the study period, 1299 patients received ketorolac.
Baseline characteristics were similar between the 2 groups except for the average baseline NRS pain score, which was statistically significantly higher in the BFA vs ketorolac group (8.7 vs 7.7, respectively; P = .02). The mean age was 51 years in the BFA group and 48 years in the ketorolac group. Most patients in each cohort were male: 80% in the BFA group and 71% in the ketorolac group. The most common types of pain documented as the chief ED presentation included back, lower extremity, and head.
Endpoints
The mean difference in NRS pain score was 3.9 for the BFA group and 5.1 for the ketorolac group. Both were clinically and statistically significant reductions (P = .03 and P < .01), but the difference between the intervention groups in NRS score reduction was not statistically significant (P = .07).
For the secondary endpoint of outpatient prescriptions written at discharge, there was no significant difference between the groups except for oral NSAIDs, which were more likely to be prescribed to patients who received ketorolac (P = .01).
Subgroup Analysis
An analysis was performed for subgroups classified by baseline NRS pain score (mild: 4; moderate, 5 - 6; severe, 7 - 9; and very severe, 10). Data for mild pain was limited because a small number of patients received interventions. For moderate pain, the mean difference in NRS pain score for BFA and ketorolac was 3.5 and 3.8, respectively; for severe pain, 3.4 and 5.3; and for very severe pain, 4.6 and 6.4. There was a larger difference in the preintervention and postintervention NRS pain scores within severe pain and very severe pain groups.
Discussion
Both interventions resulted in a significant reduction in the mean NRS pain score of about 4 to 5 points within their group, and BFA resulted in a similar NRS pain score reduction compared with ketorolac 15 mg. Because the baseline NRS pain scores were significantly different between the BFA and ketorolac groups,
In this study, more patients in the BFA group presented to the ED with lower extremity pain, such as gout or neuropathy, compared with the ketorolac group; however, BFA did not result in a significantly different pain score reduction in this subgroup compared with ketorolac. Patients receiving BFA were more likely to receive topical analgesics or muscle relaxants at discharge; whereas those receiving ketorolac were significantly more likely to receive oral NSAIDs. Patients in this study also were more likely to be admitted to the hospital if they received ketorolac; however, for these patients, pain was secondary to their chief presentation, and the admitting physician’s familiarity with ketorolac might have been the reason for choosing this intervention. Reasons for the admissions were surgical observation, psychiatric stabilization, kidney/gallstones, rule out of acute coronary syndrome, pneumonia, and proctitis in the ketorolac group, and suicidal ideations in the BFA group.
Limitations
As a limited number of patients received BFA at JBVAMC, the study was not sufficiently powered to detect a difference in the primary outcome. Because BFA required a consultation to be entered in the electronic health record, in addition to time needed to perform the procedure, practitioners might have preferred IV/IM ketorolac during busy times in the ED, potentially leading to underrepresentation in the BFA group. Prescribing preferences might have differed among the rotating physicians, timing of the documentation of the NRS pain score could have differed based on the treatment intervention, and the investigators were unable to control or accurately assess whether patients had taken an analgesic medication before presenting to the ED.
Conclusions
NRS pain score reduction with BFA did not differ compared with ketorolac 15 mg for treating acute and acute-on-chronic pain in the ED. Although this study was underpowered, these results add to the limited existing literature, suggesting that both interventions could result in clinically significant pain score reductions for patients presenting to the ED with severe and very severe pain, making BFA a viable nonpharmacologic option. Future studies could include investigating the benefit of BFA in the veteran population by studying larger samples in the ED, surveying patients after their interventions to identify rates AEs, and exploring the use of BFA for chronic pain in the outpatient setting.
1. Cantrill SV, Brown MD, Carlisle RJ, et al. Clinical policy: critical issues in the prescribing of opioids for adult patients in the emergency department. Ann Emerg Med. 2012;60(4):499-525. doi:10.1016/j.annemergmed.2012.06.013
2. Nahin RL. Severe pain in veterans: the effect of age and sex, and comparisons with the general population. J Pain. 2017;18(3):247-254. doi:10.1016/j.jpain.2016.10.021
3. Motov S, Strayer R, Hayes BD, et al. The treatment of acute pain in the emergency department: a white paper position statement prepared for the American Academy of Emergency Medicine. J Emerg Med. 2018;54(5):731-736. doi:10.1016/j.jemermed.2018.01.020
4. Samcam I, Papa L. Acute pain management in the emergency department. In: Prostran M, ed. Pain Management. IntechOpen; 2016. doi:10.5772/62861
5. Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for the use of opioids in the management of chronic pain. Accessed February 15, 2023. https://www.healthquality.va.gov/guidelines/Pain/cot/VADoDOpioidsCPG.pdf
6. Davis MT, Mulvaney-Day N, Larson MJ, Hoover R, Mauch D. Complementary and alternative medicine among veterans and military personnel: a synthesis of population surveys. Med Care. 2014;52(12 suppl 5):S83-590. doi:10.1097/MLR.0000000000000227
7. Goertz C, Marriott BP, Finch FD, et al. Military report more complementary and alternative medicine use than civilians. J Altern Complement Med. 2013;19(6):509-517. doi:10.1089/acm.2012.0108
8. King HC, Hickey AH, Connelly C. Auricular acupuncture: a brief introduction for military providers. Mil Med. 2013;178(8):867-874. doi:10.7205/MILMED-D-13-00075
9. Niemtzow RC. Battlefield acupuncture. Medical Acupunct. 2007;19(4):225-228. doi:10.1089/acu.2007.0603
10. Collinsworth KM, Goss DL. Battlefield acupuncture and physical therapy versus physical therapy alone after shoulder surgery. Med Acupunct. 2019;31(4):228-238. doi:10.1089/acu.2019.1372
11. Estores I, Chen K, Jackson B, Lao L, Gorman PH. Auricular acupuncture for spinal cord injury related neuropathic pain: a pilot controlled clinical trial. J Spinal Cord Med. 2017;40(4):432-438. doi:10.1080/10790268.2016.1141489
12. Federman DG, Radhakrishnan K, Gabriel L, Poulin LM, Kravetz JD. Group battlefield acupuncture in primary care for veterans with pain. South Med J. 2018;111(10):619-624. doi:10.14423/SMJ.0000000000000877
13. Garner BK, Hopkinson SG, Ketz AK, Landis CA, Trego LL. Auricular acupuncture for chronic pain and insomnia: a randomized clinical trial. Med Acupunct. 2018;30(5):262-272. doi:10.1089/acu.2018.1294
14. Fox LM, Murakami M, Danesh H, Manini AF. Battlefield acupuncture to treat low back pain in the emergency department. Am J Emerg Med. 2018; 36:1045-1048. doi:10.1016/j.ajem.2018.02.038
15. Woo A, Lechner B, Fu T, et al. Cut points for mild, moderate, and severe pain among cancer and non-cancer patients: a literature review. Ann Palliat Med. 2015;4(4):176-183. doi:10.3978/j.issn.2224-5820.2015.09.04
16. Motov S, Yasavolian M, Likourezos A, et al. Comparison of intravenous ketorolac at three single-dose regimens for treating acute pain in the emergency department: a randomized controlled trial. Ann Emerg Med. 2017;70(2):177-184. doi:10.1016/j.annemergmed.2016.10.014
17. Bijur PE, Latimer CT, Gallagher EJ. Validation of a verbally administered numerical rating scale of acute pain for use in the emergency department. Acad Emerg Med. 2003;10:390-392. doi:10.1111/j.1553-2712.2003.tb01355.
1. Cantrill SV, Brown MD, Carlisle RJ, et al. Clinical policy: critical issues in the prescribing of opioids for adult patients in the emergency department. Ann Emerg Med. 2012;60(4):499-525. doi:10.1016/j.annemergmed.2012.06.013
2. Nahin RL. Severe pain in veterans: the effect of age and sex, and comparisons with the general population. J Pain. 2017;18(3):247-254. doi:10.1016/j.jpain.2016.10.021
3. Motov S, Strayer R, Hayes BD, et al. The treatment of acute pain in the emergency department: a white paper position statement prepared for the American Academy of Emergency Medicine. J Emerg Med. 2018;54(5):731-736. doi:10.1016/j.jemermed.2018.01.020
4. Samcam I, Papa L. Acute pain management in the emergency department. In: Prostran M, ed. Pain Management. IntechOpen; 2016. doi:10.5772/62861
5. Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for the use of opioids in the management of chronic pain. Accessed February 15, 2023. https://www.healthquality.va.gov/guidelines/Pain/cot/VADoDOpioidsCPG.pdf
6. Davis MT, Mulvaney-Day N, Larson MJ, Hoover R, Mauch D. Complementary and alternative medicine among veterans and military personnel: a synthesis of population surveys. Med Care. 2014;52(12 suppl 5):S83-590. doi:10.1097/MLR.0000000000000227
7. Goertz C, Marriott BP, Finch FD, et al. Military report more complementary and alternative medicine use than civilians. J Altern Complement Med. 2013;19(6):509-517. doi:10.1089/acm.2012.0108
8. King HC, Hickey AH, Connelly C. Auricular acupuncture: a brief introduction for military providers. Mil Med. 2013;178(8):867-874. doi:10.7205/MILMED-D-13-00075
9. Niemtzow RC. Battlefield acupuncture. Medical Acupunct. 2007;19(4):225-228. doi:10.1089/acu.2007.0603
10. Collinsworth KM, Goss DL. Battlefield acupuncture and physical therapy versus physical therapy alone after shoulder surgery. Med Acupunct. 2019;31(4):228-238. doi:10.1089/acu.2019.1372
11. Estores I, Chen K, Jackson B, Lao L, Gorman PH. Auricular acupuncture for spinal cord injury related neuropathic pain: a pilot controlled clinical trial. J Spinal Cord Med. 2017;40(4):432-438. doi:10.1080/10790268.2016.1141489
12. Federman DG, Radhakrishnan K, Gabriel L, Poulin LM, Kravetz JD. Group battlefield acupuncture in primary care for veterans with pain. South Med J. 2018;111(10):619-624. doi:10.14423/SMJ.0000000000000877
13. Garner BK, Hopkinson SG, Ketz AK, Landis CA, Trego LL. Auricular acupuncture for chronic pain and insomnia: a randomized clinical trial. Med Acupunct. 2018;30(5):262-272. doi:10.1089/acu.2018.1294
14. Fox LM, Murakami M, Danesh H, Manini AF. Battlefield acupuncture to treat low back pain in the emergency department. Am J Emerg Med. 2018; 36:1045-1048. doi:10.1016/j.ajem.2018.02.038
15. Woo A, Lechner B, Fu T, et al. Cut points for mild, moderate, and severe pain among cancer and non-cancer patients: a literature review. Ann Palliat Med. 2015;4(4):176-183. doi:10.3978/j.issn.2224-5820.2015.09.04
16. Motov S, Yasavolian M, Likourezos A, et al. Comparison of intravenous ketorolac at three single-dose regimens for treating acute pain in the emergency department: a randomized controlled trial. Ann Emerg Med. 2017;70(2):177-184. doi:10.1016/j.annemergmed.2016.10.014
17. Bijur PE, Latimer CT, Gallagher EJ. Validation of a verbally administered numerical rating scale of acute pain for use in the emergency department. Acad Emerg Med. 2003;10:390-392. doi:10.1111/j.1553-2712.2003.tb01355.
IUD-released levonorgestrel eases heavy menstrual periods
Median blood loss decreased by more than 90% in the first three cycles. Overall, treatment was successful in 81.8% of 99 patients (95% confidence interval, 74.2%-89.4%), according to findings published in Obstetrics & Gynecology.
Already approved for contraception, the IUD (Liletta) had substantial benefits for quality of life in measures such as sleep, pain/cramping, and daily functioning, wrote a group led by Mitchell D. Creinin, MD, a professor in the department of obstetrics and gynecology at University of California, Davis.
“This study provides evidence of high efficacy, as expected, for the Liletta levonorgestrel 52 mg IUD for heavy menstrual bleeding treatment,” Dr. Creinin said in an interview.
Racially diverse cohort
Conducted at 29 U.S. sites prior to seeking FDA registration for this new use, the phase 3 open-label trial of the 52 mg progestin-releasing IUD enrolled 105 participants with a mean age of 35.4 years. Unlike previous trials, this one included obese or severely obese women (44.8%), with 42 participants having a body mass index (BMI) of more than 35 kg/m2, and also 28 nulliparous women (27.6%).
Those with abnormalities such as fibroids or coagulopathies were excluded. Although most of the cohort was White (n = 68), the study included Black (n = 25), Asian (n = 4), and Hispanic (n = 10) women, plus 7 from other minorities, suggesting the results would be widely applicable.
Mean baseline blood loss in the cohort ranged from 73 mL to 520 mL (median, 143 mL). Of 89 treated women with follow-up, participants had a median absolute blood-loss decreases of 93.3% (86.1%-97.8%) at cycle three and 97.6% (90.4%-100%) at cycle six. Median bleeding reductions at cycle six were similar between women with and without obesity at 97.6% and 97.5%, respectively, and between nulliparous and parous women at 97.0% and 98.1%, respectively (P = .43). The study, however, was not sufficiently powered to fully analyze these subgroups, the authors acknowledged.
Although results were overall comparable with those of a previous study on a different IUD, the expulsion rate was somewhat higher, at 9%, than the 6% reported in the earlier study.
“Although this strategy for reducing blood loss is not new, this study is notable because it looked at high-BMI women and nulliparous women,” said Kathryn J. Gray, MD, PhD, an attending physician in the department of obstetrics and gynecology at Brigham and Women’s Hospital in Boston, who was not involved in the research.“No prior trials have included patients with BMIs exceeding 35 kg/m2 or nulliparous patients, while this study enrolled a full array of patients, which allowed exploratory analyses of these subpopulations,” Dr. Creinin confirmed.
According to Dr. Gray, the IUD approach has advantages over systemic treatment with oral medication. “First, treatment is not user-dependent so the user doesn’t have to remember to take it. In addition, because the medication is locally targeted in the uterus, it is more effective and there is less fluctuation and variability in drug levels than when taken orally.”
As to treatment durability, Dr. Creinin said, “Long-term studies in a population being treated for heavy menstrual bleeding would be helpful to have an idea of how long this effect lasts. Still, there is no reason to expect that the effect will not last for many years.”
And with this treatment, he added, both patient and clinician can readily detect its effect. “If bleeding begins to increase, they will know!”
Would there be a lingering residual effect even after removal of the IUD? “That is an excellent question that remains to be answered,” Dr. Creinin said. “There are no data on when the heavy bleeding returns, but it would be expected to do so.”
This study was funded, designed, and supervised by Medicines360, which also provided the study treatment. Dr. Creinin disclosed financial relationships with various private-sector companies, including Medicines360, Organon, Fuji Pharma, GlaxoSmithKline, and Merck & Co. Multiple study coauthors disclosed similar financial ties to industry partners, including Medicines360. Dr. Gray had no potential conflicts of interest with regard to her comments.
Median blood loss decreased by more than 90% in the first three cycles. Overall, treatment was successful in 81.8% of 99 patients (95% confidence interval, 74.2%-89.4%), according to findings published in Obstetrics & Gynecology.
Already approved for contraception, the IUD (Liletta) had substantial benefits for quality of life in measures such as sleep, pain/cramping, and daily functioning, wrote a group led by Mitchell D. Creinin, MD, a professor in the department of obstetrics and gynecology at University of California, Davis.
“This study provides evidence of high efficacy, as expected, for the Liletta levonorgestrel 52 mg IUD for heavy menstrual bleeding treatment,” Dr. Creinin said in an interview.
Racially diverse cohort
Conducted at 29 U.S. sites prior to seeking FDA registration for this new use, the phase 3 open-label trial of the 52 mg progestin-releasing IUD enrolled 105 participants with a mean age of 35.4 years. Unlike previous trials, this one included obese or severely obese women (44.8%), with 42 participants having a body mass index (BMI) of more than 35 kg/m2, and also 28 nulliparous women (27.6%).
Those with abnormalities such as fibroids or coagulopathies were excluded. Although most of the cohort was White (n = 68), the study included Black (n = 25), Asian (n = 4), and Hispanic (n = 10) women, plus 7 from other minorities, suggesting the results would be widely applicable.
Mean baseline blood loss in the cohort ranged from 73 mL to 520 mL (median, 143 mL). Of 89 treated women with follow-up, participants had a median absolute blood-loss decreases of 93.3% (86.1%-97.8%) at cycle three and 97.6% (90.4%-100%) at cycle six. Median bleeding reductions at cycle six were similar between women with and without obesity at 97.6% and 97.5%, respectively, and between nulliparous and parous women at 97.0% and 98.1%, respectively (P = .43). The study, however, was not sufficiently powered to fully analyze these subgroups, the authors acknowledged.
Although results were overall comparable with those of a previous study on a different IUD, the expulsion rate was somewhat higher, at 9%, than the 6% reported in the earlier study.
“Although this strategy for reducing blood loss is not new, this study is notable because it looked at high-BMI women and nulliparous women,” said Kathryn J. Gray, MD, PhD, an attending physician in the department of obstetrics and gynecology at Brigham and Women’s Hospital in Boston, who was not involved in the research.“No prior trials have included patients with BMIs exceeding 35 kg/m2 or nulliparous patients, while this study enrolled a full array of patients, which allowed exploratory analyses of these subpopulations,” Dr. Creinin confirmed.
According to Dr. Gray, the IUD approach has advantages over systemic treatment with oral medication. “First, treatment is not user-dependent so the user doesn’t have to remember to take it. In addition, because the medication is locally targeted in the uterus, it is more effective and there is less fluctuation and variability in drug levels than when taken orally.”
As to treatment durability, Dr. Creinin said, “Long-term studies in a population being treated for heavy menstrual bleeding would be helpful to have an idea of how long this effect lasts. Still, there is no reason to expect that the effect will not last for many years.”
And with this treatment, he added, both patient and clinician can readily detect its effect. “If bleeding begins to increase, they will know!”
Would there be a lingering residual effect even after removal of the IUD? “That is an excellent question that remains to be answered,” Dr. Creinin said. “There are no data on when the heavy bleeding returns, but it would be expected to do so.”
This study was funded, designed, and supervised by Medicines360, which also provided the study treatment. Dr. Creinin disclosed financial relationships with various private-sector companies, including Medicines360, Organon, Fuji Pharma, GlaxoSmithKline, and Merck & Co. Multiple study coauthors disclosed similar financial ties to industry partners, including Medicines360. Dr. Gray had no potential conflicts of interest with regard to her comments.
Median blood loss decreased by more than 90% in the first three cycles. Overall, treatment was successful in 81.8% of 99 patients (95% confidence interval, 74.2%-89.4%), according to findings published in Obstetrics & Gynecology.
Already approved for contraception, the IUD (Liletta) had substantial benefits for quality of life in measures such as sleep, pain/cramping, and daily functioning, wrote a group led by Mitchell D. Creinin, MD, a professor in the department of obstetrics and gynecology at University of California, Davis.
“This study provides evidence of high efficacy, as expected, for the Liletta levonorgestrel 52 mg IUD for heavy menstrual bleeding treatment,” Dr. Creinin said in an interview.
Racially diverse cohort
Conducted at 29 U.S. sites prior to seeking FDA registration for this new use, the phase 3 open-label trial of the 52 mg progestin-releasing IUD enrolled 105 participants with a mean age of 35.4 years. Unlike previous trials, this one included obese or severely obese women (44.8%), with 42 participants having a body mass index (BMI) of more than 35 kg/m2, and also 28 nulliparous women (27.6%).
Those with abnormalities such as fibroids or coagulopathies were excluded. Although most of the cohort was White (n = 68), the study included Black (n = 25), Asian (n = 4), and Hispanic (n = 10) women, plus 7 from other minorities, suggesting the results would be widely applicable.
Mean baseline blood loss in the cohort ranged from 73 mL to 520 mL (median, 143 mL). Of 89 treated women with follow-up, participants had a median absolute blood-loss decreases of 93.3% (86.1%-97.8%) at cycle three and 97.6% (90.4%-100%) at cycle six. Median bleeding reductions at cycle six were similar between women with and without obesity at 97.6% and 97.5%, respectively, and between nulliparous and parous women at 97.0% and 98.1%, respectively (P = .43). The study, however, was not sufficiently powered to fully analyze these subgroups, the authors acknowledged.
Although results were overall comparable with those of a previous study on a different IUD, the expulsion rate was somewhat higher, at 9%, than the 6% reported in the earlier study.
“Although this strategy for reducing blood loss is not new, this study is notable because it looked at high-BMI women and nulliparous women,” said Kathryn J. Gray, MD, PhD, an attending physician in the department of obstetrics and gynecology at Brigham and Women’s Hospital in Boston, who was not involved in the research.“No prior trials have included patients with BMIs exceeding 35 kg/m2 or nulliparous patients, while this study enrolled a full array of patients, which allowed exploratory analyses of these subpopulations,” Dr. Creinin confirmed.
According to Dr. Gray, the IUD approach has advantages over systemic treatment with oral medication. “First, treatment is not user-dependent so the user doesn’t have to remember to take it. In addition, because the medication is locally targeted in the uterus, it is more effective and there is less fluctuation and variability in drug levels than when taken orally.”
As to treatment durability, Dr. Creinin said, “Long-term studies in a population being treated for heavy menstrual bleeding would be helpful to have an idea of how long this effect lasts. Still, there is no reason to expect that the effect will not last for many years.”
And with this treatment, he added, both patient and clinician can readily detect its effect. “If bleeding begins to increase, they will know!”
Would there be a lingering residual effect even after removal of the IUD? “That is an excellent question that remains to be answered,” Dr. Creinin said. “There are no data on when the heavy bleeding returns, but it would be expected to do so.”
This study was funded, designed, and supervised by Medicines360, which also provided the study treatment. Dr. Creinin disclosed financial relationships with various private-sector companies, including Medicines360, Organon, Fuji Pharma, GlaxoSmithKline, and Merck & Co. Multiple study coauthors disclosed similar financial ties to industry partners, including Medicines360. Dr. Gray had no potential conflicts of interest with regard to her comments.
FROM OBSTETRICS & GYNECOLOGY
New guidelines for cannabis in chronic pain management released
New clinical practice guidelines for cannabis in chronic pain management have been released.
Developed by a group of Canadian researchers, clinicians, and patients, the guidelines note that cannabinoid-based medicines (CBM) may help clinicians offer an effective, less addictive, alternative to opioids in patients with chronic noncancer pain and comorbid conditions.
“We don’t recommend using CBM first line for anything pretty much because there are other alternatives that may be more effective and also offer fewer side effects,” lead guideline author Alan Bell, MD, assistant professor of family and community medicine at the University of Toronto, told this news organization.
“But I would strongly argue that I would use cannabis-based medicine over opioids every time. Why would you use a high potency-high toxicity agent when there’s a low potency-low toxicity alternative?” he said.
The guidelines were published online in the journal Cannabis and Cannabinoid Research.
Examining the evidence
A consistent criticism of CBM has been the lack of quality research supporting its therapeutic utility. To develop the current recommendations, the task force reviewed 47 pain management studies enrolling more than 11,000 patients. Almost half of the studies (n = 22) were randomized controlled trials (RCTs) and 12 of the 19 included systematic reviews focused solely on RCTs.
Overall, 38 of the 47 included studies demonstrated that CBM provided at least moderate benefits for chronic pain, resulting in a “strong” recommendation – mostly as an adjunct or replacement treatment in individuals living with chronic pain.
Overall, the guidelines place a high value on improving chronic pain and functionality, and addressing co-occurring conditions such as insomnia, anxiety and depression, mobility, and inflammation. They also provide practical dosing and formulation tips to support the use of CBM in the clinical setting.
When it comes to chronic pain, CBM is not a panacea. However, prior research suggests cannabinoids and opioids share several pharmacologic properties, including independent but possibly related mechanisms for antinociception, making them an intriguing combination.
In the current guidelines, all of the four studies specifically addressing combined opioids and vaporized cannabis flower demonstrated further pain reduction, reinforcing the conclusion that the benefits of CBM for improving pain control in patients taking opioids outweigh the risk of nonserious adverse events (AEs), such as dry mouth, dizziness, increased appetite, sedation, and concentration difficulties.
The recommendations also highlighted evidence demonstrating that a majority of participants were able to reduce use of routine pain medications with concomitant CBM/opioid administration, while simultaneously offering secondary benefits such as improved sleep, anxiety, and mood, as well as prevention of opioid tolerance and dose escalation.
Importantly, the guidelines offer an evidence-based algorithm with a clear framework for tapering patients off opioids, especially those who are on > 50 mg MED, which places them with a twofold greater risk for fatal overdose.
An effective alternative
Commenting on the new guidelines, Mark Wallace, MD, who has extensive experience researching and treating pain patients with medical cannabis, said the genesis of his interest in medical cannabis mirrors the guidelines’ focus.
“What got me interested in medical cannabis was trying to get patients off of opioids,” said Dr. Wallace, professor of anesthesiology and chief of the division of pain medicine in the department of anesthesiology at the University of California, San Diego. Dr. Wallace, who was not involved in the guidelines’ development study, said that he’s “titrated hundreds of patients off of opioids using cannabis.”
Dr. Wallace said he found the guidelines’ dosing recommendations helpful.
“If you stay within the 1- to 5-mg dosing range, the risks are so incredibly low, you’re not going to harm the patient.”
While there are patients who abuse cannabis and CBMs, Dr. Wallace noted that he has seen only one patient in the past 20 years who was overusing the medical cannabis. He added that his patient population does not use medical cannabis to get high and, in fact, wants to avoid doses that produce that effect at all costs.
Also commenting on the guidelines, Christopher Gilligan, MD, MBA, associate chief medical officer and a pain medicine physician at Brigham and Women’s Hospital in Boston, who was not involved in the guidelines’ development, points to the risks.
“When we have an opportunity to use cannabinoids in place of opioids for our patients, I think that that’s a positive thing ... and a wise choice in terms of risk benefit,” Dr. Gilligan said.
On the other hand, he cautioned that “freely prescribing” cannabinoids for chronic pain in patients who aren’t on opioids is not good practice.
“We have to take seriously the potential adverse effects of [cannabis], including marijuana use disorder, interference with learning, memory impairment, and psychotic breakthroughs,” said Dr. Gilligan.
Given the current climate, it would appear that CBM is a long way from being endorsed by the Food and Drug Administration, but for clinicians interested in trying CBM for chronic pain patients, the guidelines may offer a roadmap for initiation and an alternative to prescribing opioids.
Dr. Bell, Dr. Gilligan, and Dr. Wallace report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New clinical practice guidelines for cannabis in chronic pain management have been released.
Developed by a group of Canadian researchers, clinicians, and patients, the guidelines note that cannabinoid-based medicines (CBM) may help clinicians offer an effective, less addictive, alternative to opioids in patients with chronic noncancer pain and comorbid conditions.
“We don’t recommend using CBM first line for anything pretty much because there are other alternatives that may be more effective and also offer fewer side effects,” lead guideline author Alan Bell, MD, assistant professor of family and community medicine at the University of Toronto, told this news organization.
“But I would strongly argue that I would use cannabis-based medicine over opioids every time. Why would you use a high potency-high toxicity agent when there’s a low potency-low toxicity alternative?” he said.
The guidelines were published online in the journal Cannabis and Cannabinoid Research.
Examining the evidence
A consistent criticism of CBM has been the lack of quality research supporting its therapeutic utility. To develop the current recommendations, the task force reviewed 47 pain management studies enrolling more than 11,000 patients. Almost half of the studies (n = 22) were randomized controlled trials (RCTs) and 12 of the 19 included systematic reviews focused solely on RCTs.
Overall, 38 of the 47 included studies demonstrated that CBM provided at least moderate benefits for chronic pain, resulting in a “strong” recommendation – mostly as an adjunct or replacement treatment in individuals living with chronic pain.
Overall, the guidelines place a high value on improving chronic pain and functionality, and addressing co-occurring conditions such as insomnia, anxiety and depression, mobility, and inflammation. They also provide practical dosing and formulation tips to support the use of CBM in the clinical setting.
When it comes to chronic pain, CBM is not a panacea. However, prior research suggests cannabinoids and opioids share several pharmacologic properties, including independent but possibly related mechanisms for antinociception, making them an intriguing combination.
In the current guidelines, all of the four studies specifically addressing combined opioids and vaporized cannabis flower demonstrated further pain reduction, reinforcing the conclusion that the benefits of CBM for improving pain control in patients taking opioids outweigh the risk of nonserious adverse events (AEs), such as dry mouth, dizziness, increased appetite, sedation, and concentration difficulties.
The recommendations also highlighted evidence demonstrating that a majority of participants were able to reduce use of routine pain medications with concomitant CBM/opioid administration, while simultaneously offering secondary benefits such as improved sleep, anxiety, and mood, as well as prevention of opioid tolerance and dose escalation.
Importantly, the guidelines offer an evidence-based algorithm with a clear framework for tapering patients off opioids, especially those who are on > 50 mg MED, which places them with a twofold greater risk for fatal overdose.
An effective alternative
Commenting on the new guidelines, Mark Wallace, MD, who has extensive experience researching and treating pain patients with medical cannabis, said the genesis of his interest in medical cannabis mirrors the guidelines’ focus.
“What got me interested in medical cannabis was trying to get patients off of opioids,” said Dr. Wallace, professor of anesthesiology and chief of the division of pain medicine in the department of anesthesiology at the University of California, San Diego. Dr. Wallace, who was not involved in the guidelines’ development study, said that he’s “titrated hundreds of patients off of opioids using cannabis.”
Dr. Wallace said he found the guidelines’ dosing recommendations helpful.
“If you stay within the 1- to 5-mg dosing range, the risks are so incredibly low, you’re not going to harm the patient.”
While there are patients who abuse cannabis and CBMs, Dr. Wallace noted that he has seen only one patient in the past 20 years who was overusing the medical cannabis. He added that his patient population does not use medical cannabis to get high and, in fact, wants to avoid doses that produce that effect at all costs.
Also commenting on the guidelines, Christopher Gilligan, MD, MBA, associate chief medical officer and a pain medicine physician at Brigham and Women’s Hospital in Boston, who was not involved in the guidelines’ development, points to the risks.
“When we have an opportunity to use cannabinoids in place of opioids for our patients, I think that that’s a positive thing ... and a wise choice in terms of risk benefit,” Dr. Gilligan said.
On the other hand, he cautioned that “freely prescribing” cannabinoids for chronic pain in patients who aren’t on opioids is not good practice.
“We have to take seriously the potential adverse effects of [cannabis], including marijuana use disorder, interference with learning, memory impairment, and psychotic breakthroughs,” said Dr. Gilligan.
Given the current climate, it would appear that CBM is a long way from being endorsed by the Food and Drug Administration, but for clinicians interested in trying CBM for chronic pain patients, the guidelines may offer a roadmap for initiation and an alternative to prescribing opioids.
Dr. Bell, Dr. Gilligan, and Dr. Wallace report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New clinical practice guidelines for cannabis in chronic pain management have been released.
Developed by a group of Canadian researchers, clinicians, and patients, the guidelines note that cannabinoid-based medicines (CBM) may help clinicians offer an effective, less addictive, alternative to opioids in patients with chronic noncancer pain and comorbid conditions.
“We don’t recommend using CBM first line for anything pretty much because there are other alternatives that may be more effective and also offer fewer side effects,” lead guideline author Alan Bell, MD, assistant professor of family and community medicine at the University of Toronto, told this news organization.
“But I would strongly argue that I would use cannabis-based medicine over opioids every time. Why would you use a high potency-high toxicity agent when there’s a low potency-low toxicity alternative?” he said.
The guidelines were published online in the journal Cannabis and Cannabinoid Research.
Examining the evidence
A consistent criticism of CBM has been the lack of quality research supporting its therapeutic utility. To develop the current recommendations, the task force reviewed 47 pain management studies enrolling more than 11,000 patients. Almost half of the studies (n = 22) were randomized controlled trials (RCTs) and 12 of the 19 included systematic reviews focused solely on RCTs.
Overall, 38 of the 47 included studies demonstrated that CBM provided at least moderate benefits for chronic pain, resulting in a “strong” recommendation – mostly as an adjunct or replacement treatment in individuals living with chronic pain.
Overall, the guidelines place a high value on improving chronic pain and functionality, and addressing co-occurring conditions such as insomnia, anxiety and depression, mobility, and inflammation. They also provide practical dosing and formulation tips to support the use of CBM in the clinical setting.
When it comes to chronic pain, CBM is not a panacea. However, prior research suggests cannabinoids and opioids share several pharmacologic properties, including independent but possibly related mechanisms for antinociception, making them an intriguing combination.
In the current guidelines, all of the four studies specifically addressing combined opioids and vaporized cannabis flower demonstrated further pain reduction, reinforcing the conclusion that the benefits of CBM for improving pain control in patients taking opioids outweigh the risk of nonserious adverse events (AEs), such as dry mouth, dizziness, increased appetite, sedation, and concentration difficulties.
The recommendations also highlighted evidence demonstrating that a majority of participants were able to reduce use of routine pain medications with concomitant CBM/opioid administration, while simultaneously offering secondary benefits such as improved sleep, anxiety, and mood, as well as prevention of opioid tolerance and dose escalation.
Importantly, the guidelines offer an evidence-based algorithm with a clear framework for tapering patients off opioids, especially those who are on > 50 mg MED, which places them with a twofold greater risk for fatal overdose.
An effective alternative
Commenting on the new guidelines, Mark Wallace, MD, who has extensive experience researching and treating pain patients with medical cannabis, said the genesis of his interest in medical cannabis mirrors the guidelines’ focus.
“What got me interested in medical cannabis was trying to get patients off of opioids,” said Dr. Wallace, professor of anesthesiology and chief of the division of pain medicine in the department of anesthesiology at the University of California, San Diego. Dr. Wallace, who was not involved in the guidelines’ development study, said that he’s “titrated hundreds of patients off of opioids using cannabis.”
Dr. Wallace said he found the guidelines’ dosing recommendations helpful.
“If you stay within the 1- to 5-mg dosing range, the risks are so incredibly low, you’re not going to harm the patient.”
While there are patients who abuse cannabis and CBMs, Dr. Wallace noted that he has seen only one patient in the past 20 years who was overusing the medical cannabis. He added that his patient population does not use medical cannabis to get high and, in fact, wants to avoid doses that produce that effect at all costs.
Also commenting on the guidelines, Christopher Gilligan, MD, MBA, associate chief medical officer and a pain medicine physician at Brigham and Women’s Hospital in Boston, who was not involved in the guidelines’ development, points to the risks.
“When we have an opportunity to use cannabinoids in place of opioids for our patients, I think that that’s a positive thing ... and a wise choice in terms of risk benefit,” Dr. Gilligan said.
On the other hand, he cautioned that “freely prescribing” cannabinoids for chronic pain in patients who aren’t on opioids is not good practice.
“We have to take seriously the potential adverse effects of [cannabis], including marijuana use disorder, interference with learning, memory impairment, and psychotic breakthroughs,” said Dr. Gilligan.
Given the current climate, it would appear that CBM is a long way from being endorsed by the Food and Drug Administration, but for clinicians interested in trying CBM for chronic pain patients, the guidelines may offer a roadmap for initiation and an alternative to prescribing opioids.
Dr. Bell, Dr. Gilligan, and Dr. Wallace report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CANNABIS AND CANNABINOID RESEARCH
Cluster, migraine headache strongly linked to circadian rhythm
A meta-analysis of 16 studies showed a circadian pattern in 71% of cluster headache attacks (3,490 of 4,953), with a clear circadian peak between 9:00 p.m. and 3:00 a.m.
Migraine was also associated with a circadian pattern in 50% of cases (2,698 of 5,385) across eight studies, with a clear circadian trough between 11:00 p.m. and 7:00 a.m.
Seasonal peaks were also evident for cluster headache (spring and autumn) and migraine (April to October).
“In the short term, these findings help us explain the timing to patients – for example, it is possible that a headache at 8 a.m. is due to their internal body clock instead of their pillow, or breakfast food, or morning medications,” lead investigator Mark Burish, MD, PhD, associate professor, department of neurosurgery, at University of Texas Health Houston, told this news organization.
“In the long term, these findings do suggest that medications that target the circadian system could be effective in migraine and headache patients,” Dr. Burish added.
The study was published online in Neurology.
Treatment implications?
Across studies, chronotype was “highly variable” for both cluster headache and migraine, the investigators report.
Cluster headache was associated with lower melatonin and higher cortisol levels, compared with non–cluster headache controls.
On a genetic level, cluster headache was associated with two core circadian genes (CLOCK and REV-ERB–alpha), and five of the nine genes that increase the likelihood of having cluster headache are genes with a circadian pattern of expression.
Migraine headache was associated with lower urinary melatonin levels and with the core circadian genes, CK1-delta and ROR-alpha, and 110 of the 168 genes associated with migraine were clock-controlled genes.
“The data suggest that both of these headache disorders are highly circadian at multiple levels, especially cluster headache,” Dr. Burish said in a release.
“This reinforces the importance of the hypothalamus – the area of the brain that houses the primary biological clock – and its role in cluster headache and migraine. It also raises the question of the genetics of triggers such as sleep changes that are known triggers for migraine and are cues for the body’s circadian rhythm,” Dr. Burish said.
“We hope that future research will look into circadian medications as a new treatment option for migraine and cluster headache patients,” Dr. Burish told this news organization.
Importance of sleep regulation
The authors of an accompanying editorial note that even though the study doesn’t have immediate clinical implications, it offers a better understanding of the way chronobiologic factors may influence treatment.
“At a minimum, interventions known to regulate and improve sleep (e.g., melatonin, cognitive behavioral therapy), and which are safe and straightforward to introduce, may be useful in some individuals susceptible to circadian misalignment or sleep disorders,” write Heidi Sutherland, PhD, and Lyn Griffiths, PhD, with Queensland University of Technology, Brisbane, Australia.
“Treatment of comorbidities (e.g., insomnia) that result in sleep disturbances may also help headache management. Furthermore, chronobiological aspects of any pharmacological interventions should be considered, as some frequently used headache and migraine drugs can modulate circadian cycles and influence the expression of circadian genes (e.g., verapamil), or have sleep-related side effects,” they add.
A limitation of the study was the lack of information on factors that could influence the circadian cycle, such as medications; other disorders, such as bipolar disorder; or circadian rhythm issues, such as night-shift work.
The study was supported by grants from the Japan Society for the Promotion of Science, the National Institutes of Health, The Welch Foundation, and The Will Erwin Headache Research Foundation. Dr. Burish is an unpaid member of the medical advisory board of Clusterbusters, and a site investigator for a cluster headache clinical trial funded by Lundbeck. Dr. Sutherland has received grant funding from the U.S. Migraine Research Foundation, and received institute support from Queensland University of Technology for genetics research. Dr. Griffiths has received grant funding from the Australian NHMRC, U.S. Department of Defense, and the U.S. Migraine Research Foundation, and consultancy funding from TEVA.
A version of this article first appeared on Medscape.com.
A meta-analysis of 16 studies showed a circadian pattern in 71% of cluster headache attacks (3,490 of 4,953), with a clear circadian peak between 9:00 p.m. and 3:00 a.m.
Migraine was also associated with a circadian pattern in 50% of cases (2,698 of 5,385) across eight studies, with a clear circadian trough between 11:00 p.m. and 7:00 a.m.
Seasonal peaks were also evident for cluster headache (spring and autumn) and migraine (April to October).
“In the short term, these findings help us explain the timing to patients – for example, it is possible that a headache at 8 a.m. is due to their internal body clock instead of their pillow, or breakfast food, or morning medications,” lead investigator Mark Burish, MD, PhD, associate professor, department of neurosurgery, at University of Texas Health Houston, told this news organization.
“In the long term, these findings do suggest that medications that target the circadian system could be effective in migraine and headache patients,” Dr. Burish added.
The study was published online in Neurology.
Treatment implications?
Across studies, chronotype was “highly variable” for both cluster headache and migraine, the investigators report.
Cluster headache was associated with lower melatonin and higher cortisol levels, compared with non–cluster headache controls.
On a genetic level, cluster headache was associated with two core circadian genes (CLOCK and REV-ERB–alpha), and five of the nine genes that increase the likelihood of having cluster headache are genes with a circadian pattern of expression.
Migraine headache was associated with lower urinary melatonin levels and with the core circadian genes, CK1-delta and ROR-alpha, and 110 of the 168 genes associated with migraine were clock-controlled genes.
“The data suggest that both of these headache disorders are highly circadian at multiple levels, especially cluster headache,” Dr. Burish said in a release.
“This reinforces the importance of the hypothalamus – the area of the brain that houses the primary biological clock – and its role in cluster headache and migraine. It also raises the question of the genetics of triggers such as sleep changes that are known triggers for migraine and are cues for the body’s circadian rhythm,” Dr. Burish said.
“We hope that future research will look into circadian medications as a new treatment option for migraine and cluster headache patients,” Dr. Burish told this news organization.
Importance of sleep regulation
The authors of an accompanying editorial note that even though the study doesn’t have immediate clinical implications, it offers a better understanding of the way chronobiologic factors may influence treatment.
“At a minimum, interventions known to regulate and improve sleep (e.g., melatonin, cognitive behavioral therapy), and which are safe and straightforward to introduce, may be useful in some individuals susceptible to circadian misalignment or sleep disorders,” write Heidi Sutherland, PhD, and Lyn Griffiths, PhD, with Queensland University of Technology, Brisbane, Australia.
“Treatment of comorbidities (e.g., insomnia) that result in sleep disturbances may also help headache management. Furthermore, chronobiological aspects of any pharmacological interventions should be considered, as some frequently used headache and migraine drugs can modulate circadian cycles and influence the expression of circadian genes (e.g., verapamil), or have sleep-related side effects,” they add.
A limitation of the study was the lack of information on factors that could influence the circadian cycle, such as medications; other disorders, such as bipolar disorder; or circadian rhythm issues, such as night-shift work.
The study was supported by grants from the Japan Society for the Promotion of Science, the National Institutes of Health, The Welch Foundation, and The Will Erwin Headache Research Foundation. Dr. Burish is an unpaid member of the medical advisory board of Clusterbusters, and a site investigator for a cluster headache clinical trial funded by Lundbeck. Dr. Sutherland has received grant funding from the U.S. Migraine Research Foundation, and received institute support from Queensland University of Technology for genetics research. Dr. Griffiths has received grant funding from the Australian NHMRC, U.S. Department of Defense, and the U.S. Migraine Research Foundation, and consultancy funding from TEVA.
A version of this article first appeared on Medscape.com.
A meta-analysis of 16 studies showed a circadian pattern in 71% of cluster headache attacks (3,490 of 4,953), with a clear circadian peak between 9:00 p.m. and 3:00 a.m.
Migraine was also associated with a circadian pattern in 50% of cases (2,698 of 5,385) across eight studies, with a clear circadian trough between 11:00 p.m. and 7:00 a.m.
Seasonal peaks were also evident for cluster headache (spring and autumn) and migraine (April to October).
“In the short term, these findings help us explain the timing to patients – for example, it is possible that a headache at 8 a.m. is due to their internal body clock instead of their pillow, or breakfast food, or morning medications,” lead investigator Mark Burish, MD, PhD, associate professor, department of neurosurgery, at University of Texas Health Houston, told this news organization.
“In the long term, these findings do suggest that medications that target the circadian system could be effective in migraine and headache patients,” Dr. Burish added.
The study was published online in Neurology.
Treatment implications?
Across studies, chronotype was “highly variable” for both cluster headache and migraine, the investigators report.
Cluster headache was associated with lower melatonin and higher cortisol levels, compared with non–cluster headache controls.
On a genetic level, cluster headache was associated with two core circadian genes (CLOCK and REV-ERB–alpha), and five of the nine genes that increase the likelihood of having cluster headache are genes with a circadian pattern of expression.
Migraine headache was associated with lower urinary melatonin levels and with the core circadian genes, CK1-delta and ROR-alpha, and 110 of the 168 genes associated with migraine were clock-controlled genes.
“The data suggest that both of these headache disorders are highly circadian at multiple levels, especially cluster headache,” Dr. Burish said in a release.
“This reinforces the importance of the hypothalamus – the area of the brain that houses the primary biological clock – and its role in cluster headache and migraine. It also raises the question of the genetics of triggers such as sleep changes that are known triggers for migraine and are cues for the body’s circadian rhythm,” Dr. Burish said.
“We hope that future research will look into circadian medications as a new treatment option for migraine and cluster headache patients,” Dr. Burish told this news organization.
Importance of sleep regulation
The authors of an accompanying editorial note that even though the study doesn’t have immediate clinical implications, it offers a better understanding of the way chronobiologic factors may influence treatment.
“At a minimum, interventions known to regulate and improve sleep (e.g., melatonin, cognitive behavioral therapy), and which are safe and straightforward to introduce, may be useful in some individuals susceptible to circadian misalignment or sleep disorders,” write Heidi Sutherland, PhD, and Lyn Griffiths, PhD, with Queensland University of Technology, Brisbane, Australia.
“Treatment of comorbidities (e.g., insomnia) that result in sleep disturbances may also help headache management. Furthermore, chronobiological aspects of any pharmacological interventions should be considered, as some frequently used headache and migraine drugs can modulate circadian cycles and influence the expression of circadian genes (e.g., verapamil), or have sleep-related side effects,” they add.
A limitation of the study was the lack of information on factors that could influence the circadian cycle, such as medications; other disorders, such as bipolar disorder; or circadian rhythm issues, such as night-shift work.
The study was supported by grants from the Japan Society for the Promotion of Science, the National Institutes of Health, The Welch Foundation, and The Will Erwin Headache Research Foundation. Dr. Burish is an unpaid member of the medical advisory board of Clusterbusters, and a site investigator for a cluster headache clinical trial funded by Lundbeck. Dr. Sutherland has received grant funding from the U.S. Migraine Research Foundation, and received institute support from Queensland University of Technology for genetics research. Dr. Griffiths has received grant funding from the Australian NHMRC, U.S. Department of Defense, and the U.S. Migraine Research Foundation, and consultancy funding from TEVA.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY
Metabolic syndrome linked to knee pain in middle adulthood
DENVER – Metabolic syndrome in both early and mid-adulthood is associated with symptoms of knee osteoarthritis, according to a study presented at the OARSI 2023 World Congress.
To supplement existing evidence on the association between metabolic syndrome and joint pain in older adults, the researchers investigated the association in middle-aged adults over a 10- to 13-year period.
The researchers analyzed data from the Childhood Determinants of Adult Health study, which enrolled 2,447 adults with an average age of 31 between 2004 and 2006 and conducted follow-up in 1,549 participants with an average age of 44, during 2014-2019. The Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) was used at follow-up only to assess knee symptoms of pain, stiffness, and dysfunction, as well as the overall score.
Data at both time points included fasting blood biochemistry, waist circumference, and blood pressure measures. The criteria for metabolic syndrome requires presence of central obesity (a waist circumference of at least 94 cm in males or 80 cm in females) and two of the following four factors:
- Raised triglycerides (at least 150 mg/dL) or specific treatment for this lipid abnormality.
- Reduced HDL cholesterol (below 40 mg/dL in males and below 50 mg/dL in females) or treatment for this.
- Raised blood pressure (at least 130 mm Hg systolic or at least 85 mm Hg diastolic) or treatment of previously diagnosed hypertension.
- Raised fasting blood glucose (at least 100 mg/dL) or previously diagnosed type 2 diabetes.
The researchers grouped the participants on the basis of having no metabolic syndrome at either life stage, having metabolic syndrome in young adulthood but not at follow-up (improved), having developed metabolic syndrome at follow-up (incident), and having metabolic syndrome at both time points (persistent). Most of the participants did not have the metabolic syndrome at either time point (85%), whereas 2% improved in mid-adulthood, 9% developed incident metabolic syndrome in mid-adulthood, and 4% had persistent metabolic syndrome.
At follow-up, 43% of the participants reported pain on the WOMAC, and the average WOMAC score was 10. Prevalence of metabolic syndrome increased from 8% in young adulthood to 13% in mid-adulthood, with an increase in abdominal obesity prevalence from 29% to 47%. Metabolic syndrome at any time point – whether improved later, developed later, or persistent – was associated with more knee symptoms, compared with no metabolic syndrome.
Presence of metabolic syndrome in mid-adulthood was associated with knee symptoms from the total WOMAC score (ratio of means, 1.33; P < .001) after adjustment for age, sex, and body mass index (BMI). Metabolic syndrome was also independently associated in mid-adulthood with knee pain (RoM, 1.29; P < .001) and poor function (RoM, 1.37; P < .001).
Those who developed incident metabolic syndrome in mid-adulthood had the greatest association with overall knee symptoms (RoM, 1.56; P < .001) and with knee pain (RoM, 1.52; P < .001). Although improved and persistent metabolic syndrome were both significantly associated with total WOMAC score, neither was significantly associated with knee pain after adjustment for age, sex, and BMI.
The three individual metabolic criteria independently associated with overall WOMAC score were abdominal obesity (RoM, 1.09), hypertension (RoM, 1.44), and low HDL (RoM, 1.17; P < .001 for all).
Leigh F. Callahan, PhD, a professor of medicine and associate director of the Thurston Arthritis Research Center at the University of North Carolina at Chapel Hill, said in an interview that this topic is especially important because there’s so little understanding of the role of comorbid conditions and osteoarthritis.
“There were some key things that I thought were wonderful about this study – the longitudinal nature and the fact that they had collected metabolic syndrome [criteria] at multiple time points and were able to look at persistent versus incident metabolic syndrome,” Dr. Callahan said. “We frequently don’t have that kind of trajectory.”
Jaqueline Lourdes Rios, PhD, an assistant professor of orthopedics at University Medical Center Utrecht (Netherlands), said in an interview that the study raised questions about whether treating metabolic syndrome could help prevent the progression of osteoarthritis to some extent. “Although, if you already have damage in your cartilage, and if you have a lot of inflammation that’s local, it might be a bit trickier than just treating metabolic syndrome,” Dr. Lourdes Rios added. “Then, it might help, it might not.” Either way, she said, it’s certainly worthwhile for physicians to spend time discussing interventions to address metabolic syndrome “because you treat the patient, not a knee.”
Dr. Ding, Dr. Lourdes Rios, and Dr. Callahan had no relevant financial relationships to disclose. The researchers did not note any external funding.
DENVER – Metabolic syndrome in both early and mid-adulthood is associated with symptoms of knee osteoarthritis, according to a study presented at the OARSI 2023 World Congress.
To supplement existing evidence on the association between metabolic syndrome and joint pain in older adults, the researchers investigated the association in middle-aged adults over a 10- to 13-year period.
The researchers analyzed data from the Childhood Determinants of Adult Health study, which enrolled 2,447 adults with an average age of 31 between 2004 and 2006 and conducted follow-up in 1,549 participants with an average age of 44, during 2014-2019. The Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) was used at follow-up only to assess knee symptoms of pain, stiffness, and dysfunction, as well as the overall score.
Data at both time points included fasting blood biochemistry, waist circumference, and blood pressure measures. The criteria for metabolic syndrome requires presence of central obesity (a waist circumference of at least 94 cm in males or 80 cm in females) and two of the following four factors:
- Raised triglycerides (at least 150 mg/dL) or specific treatment for this lipid abnormality.
- Reduced HDL cholesterol (below 40 mg/dL in males and below 50 mg/dL in females) or treatment for this.
- Raised blood pressure (at least 130 mm Hg systolic or at least 85 mm Hg diastolic) or treatment of previously diagnosed hypertension.
- Raised fasting blood glucose (at least 100 mg/dL) or previously diagnosed type 2 diabetes.
The researchers grouped the participants on the basis of having no metabolic syndrome at either life stage, having metabolic syndrome in young adulthood but not at follow-up (improved), having developed metabolic syndrome at follow-up (incident), and having metabolic syndrome at both time points (persistent). Most of the participants did not have the metabolic syndrome at either time point (85%), whereas 2% improved in mid-adulthood, 9% developed incident metabolic syndrome in mid-adulthood, and 4% had persistent metabolic syndrome.
At follow-up, 43% of the participants reported pain on the WOMAC, and the average WOMAC score was 10. Prevalence of metabolic syndrome increased from 8% in young adulthood to 13% in mid-adulthood, with an increase in abdominal obesity prevalence from 29% to 47%. Metabolic syndrome at any time point – whether improved later, developed later, or persistent – was associated with more knee symptoms, compared with no metabolic syndrome.
Presence of metabolic syndrome in mid-adulthood was associated with knee symptoms from the total WOMAC score (ratio of means, 1.33; P < .001) after adjustment for age, sex, and body mass index (BMI). Metabolic syndrome was also independently associated in mid-adulthood with knee pain (RoM, 1.29; P < .001) and poor function (RoM, 1.37; P < .001).
Those who developed incident metabolic syndrome in mid-adulthood had the greatest association with overall knee symptoms (RoM, 1.56; P < .001) and with knee pain (RoM, 1.52; P < .001). Although improved and persistent metabolic syndrome were both significantly associated with total WOMAC score, neither was significantly associated with knee pain after adjustment for age, sex, and BMI.
The three individual metabolic criteria independently associated with overall WOMAC score were abdominal obesity (RoM, 1.09), hypertension (RoM, 1.44), and low HDL (RoM, 1.17; P < .001 for all).
Leigh F. Callahan, PhD, a professor of medicine and associate director of the Thurston Arthritis Research Center at the University of North Carolina at Chapel Hill, said in an interview that this topic is especially important because there’s so little understanding of the role of comorbid conditions and osteoarthritis.
“There were some key things that I thought were wonderful about this study – the longitudinal nature and the fact that they had collected metabolic syndrome [criteria] at multiple time points and were able to look at persistent versus incident metabolic syndrome,” Dr. Callahan said. “We frequently don’t have that kind of trajectory.”
Jaqueline Lourdes Rios, PhD, an assistant professor of orthopedics at University Medical Center Utrecht (Netherlands), said in an interview that the study raised questions about whether treating metabolic syndrome could help prevent the progression of osteoarthritis to some extent. “Although, if you already have damage in your cartilage, and if you have a lot of inflammation that’s local, it might be a bit trickier than just treating metabolic syndrome,” Dr. Lourdes Rios added. “Then, it might help, it might not.” Either way, she said, it’s certainly worthwhile for physicians to spend time discussing interventions to address metabolic syndrome “because you treat the patient, not a knee.”
Dr. Ding, Dr. Lourdes Rios, and Dr. Callahan had no relevant financial relationships to disclose. The researchers did not note any external funding.
DENVER – Metabolic syndrome in both early and mid-adulthood is associated with symptoms of knee osteoarthritis, according to a study presented at the OARSI 2023 World Congress.
To supplement existing evidence on the association between metabolic syndrome and joint pain in older adults, the researchers investigated the association in middle-aged adults over a 10- to 13-year period.
The researchers analyzed data from the Childhood Determinants of Adult Health study, which enrolled 2,447 adults with an average age of 31 between 2004 and 2006 and conducted follow-up in 1,549 participants with an average age of 44, during 2014-2019. The Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) was used at follow-up only to assess knee symptoms of pain, stiffness, and dysfunction, as well as the overall score.
Data at both time points included fasting blood biochemistry, waist circumference, and blood pressure measures. The criteria for metabolic syndrome requires presence of central obesity (a waist circumference of at least 94 cm in males or 80 cm in females) and two of the following four factors:
- Raised triglycerides (at least 150 mg/dL) or specific treatment for this lipid abnormality.
- Reduced HDL cholesterol (below 40 mg/dL in males and below 50 mg/dL in females) or treatment for this.
- Raised blood pressure (at least 130 mm Hg systolic or at least 85 mm Hg diastolic) or treatment of previously diagnosed hypertension.
- Raised fasting blood glucose (at least 100 mg/dL) or previously diagnosed type 2 diabetes.
The researchers grouped the participants on the basis of having no metabolic syndrome at either life stage, having metabolic syndrome in young adulthood but not at follow-up (improved), having developed metabolic syndrome at follow-up (incident), and having metabolic syndrome at both time points (persistent). Most of the participants did not have the metabolic syndrome at either time point (85%), whereas 2% improved in mid-adulthood, 9% developed incident metabolic syndrome in mid-adulthood, and 4% had persistent metabolic syndrome.
At follow-up, 43% of the participants reported pain on the WOMAC, and the average WOMAC score was 10. Prevalence of metabolic syndrome increased from 8% in young adulthood to 13% in mid-adulthood, with an increase in abdominal obesity prevalence from 29% to 47%. Metabolic syndrome at any time point – whether improved later, developed later, or persistent – was associated with more knee symptoms, compared with no metabolic syndrome.
Presence of metabolic syndrome in mid-adulthood was associated with knee symptoms from the total WOMAC score (ratio of means, 1.33; P < .001) after adjustment for age, sex, and body mass index (BMI). Metabolic syndrome was also independently associated in mid-adulthood with knee pain (RoM, 1.29; P < .001) and poor function (RoM, 1.37; P < .001).
Those who developed incident metabolic syndrome in mid-adulthood had the greatest association with overall knee symptoms (RoM, 1.56; P < .001) and with knee pain (RoM, 1.52; P < .001). Although improved and persistent metabolic syndrome were both significantly associated with total WOMAC score, neither was significantly associated with knee pain after adjustment for age, sex, and BMI.
The three individual metabolic criteria independently associated with overall WOMAC score were abdominal obesity (RoM, 1.09), hypertension (RoM, 1.44), and low HDL (RoM, 1.17; P < .001 for all).
Leigh F. Callahan, PhD, a professor of medicine and associate director of the Thurston Arthritis Research Center at the University of North Carolina at Chapel Hill, said in an interview that this topic is especially important because there’s so little understanding of the role of comorbid conditions and osteoarthritis.
“There were some key things that I thought were wonderful about this study – the longitudinal nature and the fact that they had collected metabolic syndrome [criteria] at multiple time points and were able to look at persistent versus incident metabolic syndrome,” Dr. Callahan said. “We frequently don’t have that kind of trajectory.”
Jaqueline Lourdes Rios, PhD, an assistant professor of orthopedics at University Medical Center Utrecht (Netherlands), said in an interview that the study raised questions about whether treating metabolic syndrome could help prevent the progression of osteoarthritis to some extent. “Although, if you already have damage in your cartilage, and if you have a lot of inflammation that’s local, it might be a bit trickier than just treating metabolic syndrome,” Dr. Lourdes Rios added. “Then, it might help, it might not.” Either way, she said, it’s certainly worthwhile for physicians to spend time discussing interventions to address metabolic syndrome “because you treat the patient, not a knee.”
Dr. Ding, Dr. Lourdes Rios, and Dr. Callahan had no relevant financial relationships to disclose. The researchers did not note any external funding.
AT OARSI 2023
Antidepressants benefit some patients with osteoarthritis pain
DENVER – Using antidepressants to treat osteoarthritis pain can benefit some individuals but appears to have a clinically unimportant reduction in pain when looking at all patients who have tried them, according to a study presented at the OARSI 2023 World Congress. The review was also published in the Cochrane Database of Systematic Reviews in October 2022.
In terms of implications for clinical practice, the findings “seem to suggest there is a subgroup that is more likely to respond to antidepressants,” Anita Wluka, PhD, MBBS, a professor in the School of Public Health and Preventive Medicine at Monash University in Melbourne, told attendees. The findings also raise an important research question: “How can we identify the patient phenotype likely to benefit so we can [minimize the] risk of those adverse events and effects?”
Osteoarthritis pain is heterogeneous, and an estimated 30% of the pain is neuropathic-like, likely including central and peripheral sensitization, Dr. Wluka said. Given that antidepressants affect multiple sites along these pathways, multiple organizations have issued a conditional recommendation for duloxetine in their osteoarthritis guidelines, including OARSI, the European Alliance of Associations for Rheumatology, and the American College of Rheumatology.
The Cochrane Collaboration therefore conducted a systematic review and meta-analysis of research on the benefits and harms of using antidepressants to treat symptomatic knee and hip osteoarthritis. The review included studies through January 2021 whose participants had knee and/or hip osteoarthritis and which compared antidepressant therapy with placebo or another intervention for at least 6 weeks. The authors looked at seven outcomes: overall pain on a 0-10 scale, clinical response (at least a 50% reduction in 24‐hour mean pain), physical function using the Western Ontario and McMaster Universities Arthritis Index (WOMAC), quality of life using the EQ-5D, the proportion of participants withdrawing because of adverse events, the proportion who experienced any adverse events, and the proportion who experienced serious adverse events.
The researchers considered a change on the pain scale of 0.5-1 points to be “slight to small,” a difference above 1 up to 2 to be “moderate,” and a difference greater than 2 points to be “large.” In assessing quality of life function on a scale of 0-100, a slight to small difference was 5-10, a moderate difference was 11-20, and a large difference was above 20.
Of the 18 articles the researchers identified for qualitative synthesis, 9 met the criteria for qualitative synthesis in the meta-analysis, including 7 studies only on the knee and 2 that included the knees and hips. All nine studies compared antidepressants with placebo, with or without NSAIDs. Most focused on serotonin and norepinephrine reuptake inhibitors (SNRIs) – six studies on duloxetine and one on milnacipran – while one included fluvoxamine and one included nortriptyline.
The trials included a combined 2,122 participants who were predominantly female with an average age range of 54-66. Trials ranged from 8 to 16 weeks. Five of the trials carried risk of attrition and reporting bias, and only one trial had low risk of bias across all domains.
In five trials with SNRIs and one trial with tricyclics (nortriptyline) totaling 1,904 participants, 45% of those receiving antidepressants had a clinical response, compared with 29% of patients who received placebo (risk ratio, 1.55; 95% CI, 1.31-1.92). This absolute improvement in pain occurred in 16% more participants taking antidepressants, giving a number needed to treat (NNT) of 6. Average improvement in WOMAC physical function was 10.5 points with placebo and 16.2 points with antidepressants, indicating a “small, clinically unimportant response,” the researchers concluded.
Withdrawals because of adverse events included 11% of the antidepressant group and 5% of the placebo group (RR, 2.15; 95% CI, 1.56-2.87), putting the NNT for a harmful outcome at 17.
For all nine trials together, however, the mean reduction in pain from antidepressants was 2.3 points, compared with 1.7 points with placebo, a statistically significant but ”clinically unimportant improvement,” the researchers concluded. Adverse events occurred in 64% of the antidepressant group, compared with 49% of the placebo group (RR, 1.27; 95% CI, 1.15-1.41), which put the NNT for a harmful outcome at 7. No significant difference in serious adverse events occurred between the groups.
The analysis was limited by the low number of trials, most of which were sponsored by industry and most of which used duloxetine. Further, few of the studies enrolled patients with osteoarthritis of the hip, none assessed medium- or long-term effects, and none stratified the participants for different types of pain (neuropathic-like or central or peripheral pain sensitization).
“My general impression is that there was a statistically significant difference found in favor of duloxetine and the antidepressants,” David J. Hunter, MBBS, PhD, MSc, of the University of Sydney, said after the presentation. “There is a real risk of harm, which I think is important to take into consideration, but at least for me as a clinician and in advising other clinicians, it’s one tool in our armamentarium. I think it’s really important to allow patients to make an informed decision about the potential benefit, the real risk of harm, and the fact that it is quite useful in some patients, and I use it in my clinical practice.”
Jeffrey N. Katz, MD, MS, of Brigham and Women’s Hospital in Boston, said he uses antidepressants in the same way in his practice and that other types of medications, such as TNF inhibitors, also carry risk of harm that may exceed that of antidepressants.
“I’ve had lots of people start duloxetine, and if they stop it, it’s usually because they just don’t tolerate it very well,” Dr. Katz said.
“We don’t want to throw too many things away,” Dr. Hunter added. “Our patients don’t necessarily have a lot of choices here from a pharmacologic perspective, so I think it’s one of those options that I want to keep in my tool kit, and that’s not necessarily going to change.”
The research did not involve outside funding, and Dr. Wluka reported having no industry disclosures. Disclosure information was unavailable for Dr. Katz and Dr. Hunter. The Congress was sponsored by the Osteoarthritis Research Society International.
DENVER – Using antidepressants to treat osteoarthritis pain can benefit some individuals but appears to have a clinically unimportant reduction in pain when looking at all patients who have tried them, according to a study presented at the OARSI 2023 World Congress. The review was also published in the Cochrane Database of Systematic Reviews in October 2022.
In terms of implications for clinical practice, the findings “seem to suggest there is a subgroup that is more likely to respond to antidepressants,” Anita Wluka, PhD, MBBS, a professor in the School of Public Health and Preventive Medicine at Monash University in Melbourne, told attendees. The findings also raise an important research question: “How can we identify the patient phenotype likely to benefit so we can [minimize the] risk of those adverse events and effects?”
Osteoarthritis pain is heterogeneous, and an estimated 30% of the pain is neuropathic-like, likely including central and peripheral sensitization, Dr. Wluka said. Given that antidepressants affect multiple sites along these pathways, multiple organizations have issued a conditional recommendation for duloxetine in their osteoarthritis guidelines, including OARSI, the European Alliance of Associations for Rheumatology, and the American College of Rheumatology.
The Cochrane Collaboration therefore conducted a systematic review and meta-analysis of research on the benefits and harms of using antidepressants to treat symptomatic knee and hip osteoarthritis. The review included studies through January 2021 whose participants had knee and/or hip osteoarthritis and which compared antidepressant therapy with placebo or another intervention for at least 6 weeks. The authors looked at seven outcomes: overall pain on a 0-10 scale, clinical response (at least a 50% reduction in 24‐hour mean pain), physical function using the Western Ontario and McMaster Universities Arthritis Index (WOMAC), quality of life using the EQ-5D, the proportion of participants withdrawing because of adverse events, the proportion who experienced any adverse events, and the proportion who experienced serious adverse events.
The researchers considered a change on the pain scale of 0.5-1 points to be “slight to small,” a difference above 1 up to 2 to be “moderate,” and a difference greater than 2 points to be “large.” In assessing quality of life function on a scale of 0-100, a slight to small difference was 5-10, a moderate difference was 11-20, and a large difference was above 20.
Of the 18 articles the researchers identified for qualitative synthesis, 9 met the criteria for qualitative synthesis in the meta-analysis, including 7 studies only on the knee and 2 that included the knees and hips. All nine studies compared antidepressants with placebo, with or without NSAIDs. Most focused on serotonin and norepinephrine reuptake inhibitors (SNRIs) – six studies on duloxetine and one on milnacipran – while one included fluvoxamine and one included nortriptyline.
The trials included a combined 2,122 participants who were predominantly female with an average age range of 54-66. Trials ranged from 8 to 16 weeks. Five of the trials carried risk of attrition and reporting bias, and only one trial had low risk of bias across all domains.
In five trials with SNRIs and one trial with tricyclics (nortriptyline) totaling 1,904 participants, 45% of those receiving antidepressants had a clinical response, compared with 29% of patients who received placebo (risk ratio, 1.55; 95% CI, 1.31-1.92). This absolute improvement in pain occurred in 16% more participants taking antidepressants, giving a number needed to treat (NNT) of 6. Average improvement in WOMAC physical function was 10.5 points with placebo and 16.2 points with antidepressants, indicating a “small, clinically unimportant response,” the researchers concluded.
Withdrawals because of adverse events included 11% of the antidepressant group and 5% of the placebo group (RR, 2.15; 95% CI, 1.56-2.87), putting the NNT for a harmful outcome at 17.
For all nine trials together, however, the mean reduction in pain from antidepressants was 2.3 points, compared with 1.7 points with placebo, a statistically significant but ”clinically unimportant improvement,” the researchers concluded. Adverse events occurred in 64% of the antidepressant group, compared with 49% of the placebo group (RR, 1.27; 95% CI, 1.15-1.41), which put the NNT for a harmful outcome at 7. No significant difference in serious adverse events occurred between the groups.
The analysis was limited by the low number of trials, most of which were sponsored by industry and most of which used duloxetine. Further, few of the studies enrolled patients with osteoarthritis of the hip, none assessed medium- or long-term effects, and none stratified the participants for different types of pain (neuropathic-like or central or peripheral pain sensitization).
“My general impression is that there was a statistically significant difference found in favor of duloxetine and the antidepressants,” David J. Hunter, MBBS, PhD, MSc, of the University of Sydney, said after the presentation. “There is a real risk of harm, which I think is important to take into consideration, but at least for me as a clinician and in advising other clinicians, it’s one tool in our armamentarium. I think it’s really important to allow patients to make an informed decision about the potential benefit, the real risk of harm, and the fact that it is quite useful in some patients, and I use it in my clinical practice.”
Jeffrey N. Katz, MD, MS, of Brigham and Women’s Hospital in Boston, said he uses antidepressants in the same way in his practice and that other types of medications, such as TNF inhibitors, also carry risk of harm that may exceed that of antidepressants.
“I’ve had lots of people start duloxetine, and if they stop it, it’s usually because they just don’t tolerate it very well,” Dr. Katz said.
“We don’t want to throw too many things away,” Dr. Hunter added. “Our patients don’t necessarily have a lot of choices here from a pharmacologic perspective, so I think it’s one of those options that I want to keep in my tool kit, and that’s not necessarily going to change.”
The research did not involve outside funding, and Dr. Wluka reported having no industry disclosures. Disclosure information was unavailable for Dr. Katz and Dr. Hunter. The Congress was sponsored by the Osteoarthritis Research Society International.
DENVER – Using antidepressants to treat osteoarthritis pain can benefit some individuals but appears to have a clinically unimportant reduction in pain when looking at all patients who have tried them, according to a study presented at the OARSI 2023 World Congress. The review was also published in the Cochrane Database of Systematic Reviews in October 2022.
In terms of implications for clinical practice, the findings “seem to suggest there is a subgroup that is more likely to respond to antidepressants,” Anita Wluka, PhD, MBBS, a professor in the School of Public Health and Preventive Medicine at Monash University in Melbourne, told attendees. The findings also raise an important research question: “How can we identify the patient phenotype likely to benefit so we can [minimize the] risk of those adverse events and effects?”
Osteoarthritis pain is heterogeneous, and an estimated 30% of the pain is neuropathic-like, likely including central and peripheral sensitization, Dr. Wluka said. Given that antidepressants affect multiple sites along these pathways, multiple organizations have issued a conditional recommendation for duloxetine in their osteoarthritis guidelines, including OARSI, the European Alliance of Associations for Rheumatology, and the American College of Rheumatology.
The Cochrane Collaboration therefore conducted a systematic review and meta-analysis of research on the benefits and harms of using antidepressants to treat symptomatic knee and hip osteoarthritis. The review included studies through January 2021 whose participants had knee and/or hip osteoarthritis and which compared antidepressant therapy with placebo or another intervention for at least 6 weeks. The authors looked at seven outcomes: overall pain on a 0-10 scale, clinical response (at least a 50% reduction in 24‐hour mean pain), physical function using the Western Ontario and McMaster Universities Arthritis Index (WOMAC), quality of life using the EQ-5D, the proportion of participants withdrawing because of adverse events, the proportion who experienced any adverse events, and the proportion who experienced serious adverse events.
The researchers considered a change on the pain scale of 0.5-1 points to be “slight to small,” a difference above 1 up to 2 to be “moderate,” and a difference greater than 2 points to be “large.” In assessing quality of life function on a scale of 0-100, a slight to small difference was 5-10, a moderate difference was 11-20, and a large difference was above 20.
Of the 18 articles the researchers identified for qualitative synthesis, 9 met the criteria for qualitative synthesis in the meta-analysis, including 7 studies only on the knee and 2 that included the knees and hips. All nine studies compared antidepressants with placebo, with or without NSAIDs. Most focused on serotonin and norepinephrine reuptake inhibitors (SNRIs) – six studies on duloxetine and one on milnacipran – while one included fluvoxamine and one included nortriptyline.
The trials included a combined 2,122 participants who were predominantly female with an average age range of 54-66. Trials ranged from 8 to 16 weeks. Five of the trials carried risk of attrition and reporting bias, and only one trial had low risk of bias across all domains.
In five trials with SNRIs and one trial with tricyclics (nortriptyline) totaling 1,904 participants, 45% of those receiving antidepressants had a clinical response, compared with 29% of patients who received placebo (risk ratio, 1.55; 95% CI, 1.31-1.92). This absolute improvement in pain occurred in 16% more participants taking antidepressants, giving a number needed to treat (NNT) of 6. Average improvement in WOMAC physical function was 10.5 points with placebo and 16.2 points with antidepressants, indicating a “small, clinically unimportant response,” the researchers concluded.
Withdrawals because of adverse events included 11% of the antidepressant group and 5% of the placebo group (RR, 2.15; 95% CI, 1.56-2.87), putting the NNT for a harmful outcome at 17.
For all nine trials together, however, the mean reduction in pain from antidepressants was 2.3 points, compared with 1.7 points with placebo, a statistically significant but ”clinically unimportant improvement,” the researchers concluded. Adverse events occurred in 64% of the antidepressant group, compared with 49% of the placebo group (RR, 1.27; 95% CI, 1.15-1.41), which put the NNT for a harmful outcome at 7. No significant difference in serious adverse events occurred between the groups.
The analysis was limited by the low number of trials, most of which were sponsored by industry and most of which used duloxetine. Further, few of the studies enrolled patients with osteoarthritis of the hip, none assessed medium- or long-term effects, and none stratified the participants for different types of pain (neuropathic-like or central or peripheral pain sensitization).
“My general impression is that there was a statistically significant difference found in favor of duloxetine and the antidepressants,” David J. Hunter, MBBS, PhD, MSc, of the University of Sydney, said after the presentation. “There is a real risk of harm, which I think is important to take into consideration, but at least for me as a clinician and in advising other clinicians, it’s one tool in our armamentarium. I think it’s really important to allow patients to make an informed decision about the potential benefit, the real risk of harm, and the fact that it is quite useful in some patients, and I use it in my clinical practice.”
Jeffrey N. Katz, MD, MS, of Brigham and Women’s Hospital in Boston, said he uses antidepressants in the same way in his practice and that other types of medications, such as TNF inhibitors, also carry risk of harm that may exceed that of antidepressants.
“I’ve had lots of people start duloxetine, and if they stop it, it’s usually because they just don’t tolerate it very well,” Dr. Katz said.
“We don’t want to throw too many things away,” Dr. Hunter added. “Our patients don’t necessarily have a lot of choices here from a pharmacologic perspective, so I think it’s one of those options that I want to keep in my tool kit, and that’s not necessarily going to change.”
The research did not involve outside funding, and Dr. Wluka reported having no industry disclosures. Disclosure information was unavailable for Dr. Katz and Dr. Hunter. The Congress was sponsored by the Osteoarthritis Research Society International.
FROM OARSI 2023
Safety, efficacy of analgesics for low back pain ‘uncertain’
Higher-quality randomized controlled trials of head-to-head comparisons are needed, study investigator Michael A. Wewege, PhD candidate, research fellow, University of New South Wales and Neuroscience Research Australia, Sydney, said in an interview.
“Until then, doctors should use caution when prescribing analgesic medicines for adults with nonspecific acute low back pain. They should use this new evidence in line with their own expertise and the patient sitting in front of them when making any decision about a medication,” he added.
The findings were published online in the BMJ.
Poor quality evidence
Analgesics such as ibuprofen, acetaminophen, and codeine are widely used to treat nonspecific low-back pain, which is defined as pain lasting less than 6 weeks, but evidence for the comparative efficacy of these agents is limited.
To fill this knowledge gap, the researchers conducted a systematic review and analysis of controlled trials comparing analgesics with another analgesic, placebo, or no treatment in patients with acute, nonspecific low back pain.
The review involved 98 randomized controlled trials that included 15,134 adults (49% women) aged 30-60 years with pain duration ranging from 24 hours to 21 days. The median baseline pain intensity was 65 on a pain scale of 0-100.
Of the included trials, 39% were placebo controlled, 67% masked both participants and clinicians, and 41% reported industry sponsorship.
The studies compared an analgesic medicine with another analgesic, placebo, or no treatment comprised of usual care or being placed on a wait list.
Study medications, which had to be approved in the United States, Europe, or Australia, included nonsteroidal anti-inflammatory drugs, paracetamol, opioids, anticonvulsants, antidepressants, muscle relaxants, and corticosteroids.
These drugs were administered systemically as a single drug or in combination formulations, at any dose.
Researchers used a network meta-analysis, which combines direct and indirect information across a network of randomized clinical trials to estimate the comparative effectiveness of multiple treatments.
The primary outcomes were reductions in low back pain intensity (measured with a visual analogue scale), numerical rating scale or another ordinal scale, and safety as indicated by the number of participants who had any adverse event.
Investigators found several medications were associated with large reductions in pain intensity, compared with placebo, though with low or very low confidence.
Low or very low confidence was found for reduced pain intensity after treatment with tolperisone (mean difference, −26.1; 95% confidence interval, −34.0 to −18.2), aceclofenac plus tizanidine (mean difference, −26.1; 95% CI, −38.5 to −13.6), pregabalin (mean difference, −24.7; 95% CI, −34.6 to −14.7), and 14 other medicines, compared with placebo, the researchers report.
In addition, they found low or very low confidence for no difference between the effects of several of these medications.
Increased adverse events had moderate to very low confidence with tramadol (risk ratio, 2.6; 95% CI, 1.5-4.5), paracetamol plus sustained release tramadol (RR, 2.4; 95% CI, 1.5-3.8), baclofen (RR, 2.3; 95% CI, 1.5-3.4), and paracetamol plus tramadol (RR, 2.1; 95% CI, 1.3-3.4), compared with placebo, the investigators add.
“These medicines could increase the risk of adverse events, compared with other medicines with moderate to low confidence. Moderate to low confidence was also noted for secondary outcomes and secondary analysis of medicine classes,” the researchers note.
The review suggested 14 additional comparisons favored the treatment over placebo, all with very low confidence except for one with low confidence.
In the 68 trials that included the number of participants reporting an adverse event, there was moderate confidence for increased adverse events with the opioid tramadol (RR, 2.6; 95% CI, 1.5-4.5), paracetamol plus sustained release tramadol (RR, 2.4; 95% CI, 1.5-3.8), paracetamol plus tramadol (RR, 2.1; 95% CI, 1.3-3.4), and low confidence for baclofen (RR, 2.3; 1.5-3.4), compared with placebo.
The review also uncovered moderate to low confidence for secondary outcomes, which included low back-specific function, serious adverse events, and acceptability (number of participants who dropped out).
Unexpected findings
The new results were somewhat unexpected, said Mr. Wewege.
“When we set out to do this review, we envisioned the evidence would be a lot more comprehensive. We didn’t think it would be so disconnected and there would be so few trials looking at the different comparisons that would lead us to have low confidence in most of the findings.”
Various factors contributed to this low confidence, he said. One was the risk of bias – about 90% of trials had some concerns or high risk of bias. Another factor was the heterogeneity in effect estimates.
Most of the evidence is based on studies comparing different analgesics to placebo, Mr. Wewege noted. The lack of head-to-head drug comparisons is because “the easiest way to get a drug approved is just to demonstrate it’s better than placebo,” he said.
In addition to these new findings, clinicians should consider a medication’s availability, their own expertise, and patient preferences when selecting an analgesic, said Mr. Wewege. He noted most patients with acute low back pain get better within a few weeks without any intervention.
“Patients should be reassured that things will heal naturally and that they are not going to be in pain forever,” he said.
Determining optimal treatment is key
Chris Gilligan, MD, associate chief medical officer, Brigham and Women’s Hospital, and associate professor of anesthesia, Harvard Medical School, both in Boston, said determining which medications are optimal is “key,” as acute low back pain is very common and analgesics are used frequently.
The new review does provide information on which medications have the strongest evidence for pain reduction, said Dr. Gilligan. “On the one hand, it directionally points you towards certain medications, and even certain classes of medication, for comparative effectiveness.”
However, he said, the confidence for this effectiveness is low or very low, “so I wouldn’t overweight it.”
The data on adverse effects, where the confidence is mostly moderate to low, might have more of an influence on prescribing, he said.
“For example, there’s some indication tramadol may be more closely associated with adverse events in patients with acute low back pain and that would add to our caution about using tramadol; it’s not that we would never use it, but [we]would take that into account.”
Dr. Gilligan agrees clinicians should be cautious about prescribing analgesics for low back pain. One reason for being conservative in terms of treatments, he noted, is that “acute low back pain has a very favorable natural history.”
While clinical practice guidelines recommend nonpharmacologic therapies as first- and second-line treatment for acute, nonspecific low back pain, Dr. Gilligan noted that as with drugs, evidence for nondrug therapies also has low or very low confidence.
The study received funding from a 2020 Exercise Physiology Research (Consumables) Grant from the University of New South Wales, which was used to obtain translations of studies published in languages other than English.
Mr. Wewege was supported by a Postgraduate Scholarship from the National Health and Medical Research Council of Australia, a School of Medical Sciences Top-Up Scholarship from the University of New South Wales, and a PhD Supplementary Scholarship from Neuroscience Research Australia. Dr. Gilligan reports that he conducts clinical trials with companies and groups, including the National Institutes of Health related to medications, devices, and procedures for pain.
A version of this article first appeared on Medscape.com.
Higher-quality randomized controlled trials of head-to-head comparisons are needed, study investigator Michael A. Wewege, PhD candidate, research fellow, University of New South Wales and Neuroscience Research Australia, Sydney, said in an interview.
“Until then, doctors should use caution when prescribing analgesic medicines for adults with nonspecific acute low back pain. They should use this new evidence in line with their own expertise and the patient sitting in front of them when making any decision about a medication,” he added.
The findings were published online in the BMJ.
Poor quality evidence
Analgesics such as ibuprofen, acetaminophen, and codeine are widely used to treat nonspecific low-back pain, which is defined as pain lasting less than 6 weeks, but evidence for the comparative efficacy of these agents is limited.
To fill this knowledge gap, the researchers conducted a systematic review and analysis of controlled trials comparing analgesics with another analgesic, placebo, or no treatment in patients with acute, nonspecific low back pain.
The review involved 98 randomized controlled trials that included 15,134 adults (49% women) aged 30-60 years with pain duration ranging from 24 hours to 21 days. The median baseline pain intensity was 65 on a pain scale of 0-100.
Of the included trials, 39% were placebo controlled, 67% masked both participants and clinicians, and 41% reported industry sponsorship.
The studies compared an analgesic medicine with another analgesic, placebo, or no treatment comprised of usual care or being placed on a wait list.
Study medications, which had to be approved in the United States, Europe, or Australia, included nonsteroidal anti-inflammatory drugs, paracetamol, opioids, anticonvulsants, antidepressants, muscle relaxants, and corticosteroids.
These drugs were administered systemically as a single drug or in combination formulations, at any dose.
Researchers used a network meta-analysis, which combines direct and indirect information across a network of randomized clinical trials to estimate the comparative effectiveness of multiple treatments.
The primary outcomes were reductions in low back pain intensity (measured with a visual analogue scale), numerical rating scale or another ordinal scale, and safety as indicated by the number of participants who had any adverse event.
Investigators found several medications were associated with large reductions in pain intensity, compared with placebo, though with low or very low confidence.
Low or very low confidence was found for reduced pain intensity after treatment with tolperisone (mean difference, −26.1; 95% confidence interval, −34.0 to −18.2), aceclofenac plus tizanidine (mean difference, −26.1; 95% CI, −38.5 to −13.6), pregabalin (mean difference, −24.7; 95% CI, −34.6 to −14.7), and 14 other medicines, compared with placebo, the researchers report.
In addition, they found low or very low confidence for no difference between the effects of several of these medications.
Increased adverse events had moderate to very low confidence with tramadol (risk ratio, 2.6; 95% CI, 1.5-4.5), paracetamol plus sustained release tramadol (RR, 2.4; 95% CI, 1.5-3.8), baclofen (RR, 2.3; 95% CI, 1.5-3.4), and paracetamol plus tramadol (RR, 2.1; 95% CI, 1.3-3.4), compared with placebo, the investigators add.
“These medicines could increase the risk of adverse events, compared with other medicines with moderate to low confidence. Moderate to low confidence was also noted for secondary outcomes and secondary analysis of medicine classes,” the researchers note.
The review suggested 14 additional comparisons favored the treatment over placebo, all with very low confidence except for one with low confidence.
In the 68 trials that included the number of participants reporting an adverse event, there was moderate confidence for increased adverse events with the opioid tramadol (RR, 2.6; 95% CI, 1.5-4.5), paracetamol plus sustained release tramadol (RR, 2.4; 95% CI, 1.5-3.8), paracetamol plus tramadol (RR, 2.1; 95% CI, 1.3-3.4), and low confidence for baclofen (RR, 2.3; 1.5-3.4), compared with placebo.
The review also uncovered moderate to low confidence for secondary outcomes, which included low back-specific function, serious adverse events, and acceptability (number of participants who dropped out).
Unexpected findings
The new results were somewhat unexpected, said Mr. Wewege.
“When we set out to do this review, we envisioned the evidence would be a lot more comprehensive. We didn’t think it would be so disconnected and there would be so few trials looking at the different comparisons that would lead us to have low confidence in most of the findings.”
Various factors contributed to this low confidence, he said. One was the risk of bias – about 90% of trials had some concerns or high risk of bias. Another factor was the heterogeneity in effect estimates.
Most of the evidence is based on studies comparing different analgesics to placebo, Mr. Wewege noted. The lack of head-to-head drug comparisons is because “the easiest way to get a drug approved is just to demonstrate it’s better than placebo,” he said.
In addition to these new findings, clinicians should consider a medication’s availability, their own expertise, and patient preferences when selecting an analgesic, said Mr. Wewege. He noted most patients with acute low back pain get better within a few weeks without any intervention.
“Patients should be reassured that things will heal naturally and that they are not going to be in pain forever,” he said.
Determining optimal treatment is key
Chris Gilligan, MD, associate chief medical officer, Brigham and Women’s Hospital, and associate professor of anesthesia, Harvard Medical School, both in Boston, said determining which medications are optimal is “key,” as acute low back pain is very common and analgesics are used frequently.
The new review does provide information on which medications have the strongest evidence for pain reduction, said Dr. Gilligan. “On the one hand, it directionally points you towards certain medications, and even certain classes of medication, for comparative effectiveness.”
However, he said, the confidence for this effectiveness is low or very low, “so I wouldn’t overweight it.”
The data on adverse effects, where the confidence is mostly moderate to low, might have more of an influence on prescribing, he said.
“For example, there’s some indication tramadol may be more closely associated with adverse events in patients with acute low back pain and that would add to our caution about using tramadol; it’s not that we would never use it, but [we]would take that into account.”
Dr. Gilligan agrees clinicians should be cautious about prescribing analgesics for low back pain. One reason for being conservative in terms of treatments, he noted, is that “acute low back pain has a very favorable natural history.”
While clinical practice guidelines recommend nonpharmacologic therapies as first- and second-line treatment for acute, nonspecific low back pain, Dr. Gilligan noted that as with drugs, evidence for nondrug therapies also has low or very low confidence.
The study received funding from a 2020 Exercise Physiology Research (Consumables) Grant from the University of New South Wales, which was used to obtain translations of studies published in languages other than English.
Mr. Wewege was supported by a Postgraduate Scholarship from the National Health and Medical Research Council of Australia, a School of Medical Sciences Top-Up Scholarship from the University of New South Wales, and a PhD Supplementary Scholarship from Neuroscience Research Australia. Dr. Gilligan reports that he conducts clinical trials with companies and groups, including the National Institutes of Health related to medications, devices, and procedures for pain.
A version of this article first appeared on Medscape.com.
Higher-quality randomized controlled trials of head-to-head comparisons are needed, study investigator Michael A. Wewege, PhD candidate, research fellow, University of New South Wales and Neuroscience Research Australia, Sydney, said in an interview.
“Until then, doctors should use caution when prescribing analgesic medicines for adults with nonspecific acute low back pain. They should use this new evidence in line with their own expertise and the patient sitting in front of them when making any decision about a medication,” he added.
The findings were published online in the BMJ.
Poor quality evidence
Analgesics such as ibuprofen, acetaminophen, and codeine are widely used to treat nonspecific low-back pain, which is defined as pain lasting less than 6 weeks, but evidence for the comparative efficacy of these agents is limited.
To fill this knowledge gap, the researchers conducted a systematic review and analysis of controlled trials comparing analgesics with another analgesic, placebo, or no treatment in patients with acute, nonspecific low back pain.
The review involved 98 randomized controlled trials that included 15,134 adults (49% women) aged 30-60 years with pain duration ranging from 24 hours to 21 days. The median baseline pain intensity was 65 on a pain scale of 0-100.
Of the included trials, 39% were placebo controlled, 67% masked both participants and clinicians, and 41% reported industry sponsorship.
The studies compared an analgesic medicine with another analgesic, placebo, or no treatment comprised of usual care or being placed on a wait list.
Study medications, which had to be approved in the United States, Europe, or Australia, included nonsteroidal anti-inflammatory drugs, paracetamol, opioids, anticonvulsants, antidepressants, muscle relaxants, and corticosteroids.
These drugs were administered systemically as a single drug or in combination formulations, at any dose.
Researchers used a network meta-analysis, which combines direct and indirect information across a network of randomized clinical trials to estimate the comparative effectiveness of multiple treatments.
The primary outcomes were reductions in low back pain intensity (measured with a visual analogue scale), numerical rating scale or another ordinal scale, and safety as indicated by the number of participants who had any adverse event.
Investigators found several medications were associated with large reductions in pain intensity, compared with placebo, though with low or very low confidence.
Low or very low confidence was found for reduced pain intensity after treatment with tolperisone (mean difference, −26.1; 95% confidence interval, −34.0 to −18.2), aceclofenac plus tizanidine (mean difference, −26.1; 95% CI, −38.5 to −13.6), pregabalin (mean difference, −24.7; 95% CI, −34.6 to −14.7), and 14 other medicines, compared with placebo, the researchers report.
In addition, they found low or very low confidence for no difference between the effects of several of these medications.
Increased adverse events had moderate to very low confidence with tramadol (risk ratio, 2.6; 95% CI, 1.5-4.5), paracetamol plus sustained release tramadol (RR, 2.4; 95% CI, 1.5-3.8), baclofen (RR, 2.3; 95% CI, 1.5-3.4), and paracetamol plus tramadol (RR, 2.1; 95% CI, 1.3-3.4), compared with placebo, the investigators add.
“These medicines could increase the risk of adverse events, compared with other medicines with moderate to low confidence. Moderate to low confidence was also noted for secondary outcomes and secondary analysis of medicine classes,” the researchers note.
The review suggested 14 additional comparisons favored the treatment over placebo, all with very low confidence except for one with low confidence.
In the 68 trials that included the number of participants reporting an adverse event, there was moderate confidence for increased adverse events with the opioid tramadol (RR, 2.6; 95% CI, 1.5-4.5), paracetamol plus sustained release tramadol (RR, 2.4; 95% CI, 1.5-3.8), paracetamol plus tramadol (RR, 2.1; 95% CI, 1.3-3.4), and low confidence for baclofen (RR, 2.3; 1.5-3.4), compared with placebo.
The review also uncovered moderate to low confidence for secondary outcomes, which included low back-specific function, serious adverse events, and acceptability (number of participants who dropped out).
Unexpected findings
The new results were somewhat unexpected, said Mr. Wewege.
“When we set out to do this review, we envisioned the evidence would be a lot more comprehensive. We didn’t think it would be so disconnected and there would be so few trials looking at the different comparisons that would lead us to have low confidence in most of the findings.”
Various factors contributed to this low confidence, he said. One was the risk of bias – about 90% of trials had some concerns or high risk of bias. Another factor was the heterogeneity in effect estimates.
Most of the evidence is based on studies comparing different analgesics to placebo, Mr. Wewege noted. The lack of head-to-head drug comparisons is because “the easiest way to get a drug approved is just to demonstrate it’s better than placebo,” he said.
In addition to these new findings, clinicians should consider a medication’s availability, their own expertise, and patient preferences when selecting an analgesic, said Mr. Wewege. He noted most patients with acute low back pain get better within a few weeks without any intervention.
“Patients should be reassured that things will heal naturally and that they are not going to be in pain forever,” he said.
Determining optimal treatment is key
Chris Gilligan, MD, associate chief medical officer, Brigham and Women’s Hospital, and associate professor of anesthesia, Harvard Medical School, both in Boston, said determining which medications are optimal is “key,” as acute low back pain is very common and analgesics are used frequently.
The new review does provide information on which medications have the strongest evidence for pain reduction, said Dr. Gilligan. “On the one hand, it directionally points you towards certain medications, and even certain classes of medication, for comparative effectiveness.”
However, he said, the confidence for this effectiveness is low or very low, “so I wouldn’t overweight it.”
The data on adverse effects, where the confidence is mostly moderate to low, might have more of an influence on prescribing, he said.
“For example, there’s some indication tramadol may be more closely associated with adverse events in patients with acute low back pain and that would add to our caution about using tramadol; it’s not that we would never use it, but [we]would take that into account.”
Dr. Gilligan agrees clinicians should be cautious about prescribing analgesics for low back pain. One reason for being conservative in terms of treatments, he noted, is that “acute low back pain has a very favorable natural history.”
While clinical practice guidelines recommend nonpharmacologic therapies as first- and second-line treatment for acute, nonspecific low back pain, Dr. Gilligan noted that as with drugs, evidence for nondrug therapies also has low or very low confidence.
The study received funding from a 2020 Exercise Physiology Research (Consumables) Grant from the University of New South Wales, which was used to obtain translations of studies published in languages other than English.
Mr. Wewege was supported by a Postgraduate Scholarship from the National Health and Medical Research Council of Australia, a School of Medical Sciences Top-Up Scholarship from the University of New South Wales, and a PhD Supplementary Scholarship from Neuroscience Research Australia. Dr. Gilligan reports that he conducts clinical trials with companies and groups, including the National Institutes of Health related to medications, devices, and procedures for pain.
A version of this article first appeared on Medscape.com.
FROM BMJ
Link between knee pain, sleep disturbance related to daily activities
DENVER – The relationship between nighttime knee pain from osteoarthritis and sleep disturbances is more complex than a simple association, according to new research presented at the Osteoarthritis Research Society International 2023 World Congress.
The findings suggested that the association between knee OA pain and sleep problems was also linked to activities of daily living, which can contribute to pain but are also affected by OA, Takahiro Sasahara, of the department of orthopedics at Juntendo University, Tokyo, and Koshigaya Municipal Hospital, Saitama, Japan, told attendees. The study also found that knee pain and mobility impairment were associated with sleep disturbances in older adults regardless of the severity of knee OA.
Luisa Cedin, a PhD student at Rush University, Chicago, who attended the presentation, noted the clinical implications of the interaction of daily activities with knee pain.
”I’m a physical therapist, and this could have a significant impact on the performance of the exercises that I’m requiring as a physical therapist,” Ms. Cedin said in an interview. “When you ask somebody who is not getting enough rest during the night – not only enough time but enough quality of rest – we know that we can expect a lower performance with any type of exercises, whether it’s less strength or force, less power, less agility, or less resistance or endurance, so this has a big impact on their quality of life.”
Mr. Sasahara cited research noting that acute pain occurs at the beginning of movement and during weight bearing and walking while chronic pain frequently occurs at night and in early morning awakenings. The prevalence of sleep disturbances in patients with chronic pain ranges from 50% to 80%, he said, and past evidence has shown the relationship between sleep and pain to be bidirectional.
For example, insomnia frequency and severity, sleep-onset problems, and sleep efficiency are all positively associated with pain sensitivity, and increasing severity of OA is linked to increasing prevalence of night knee pain and sleep problems, affecting quality of life, he said.
In this new study examining the relationship between sleep disturbance and knee pain and mobility, the researchers focused specifically on a population of older adults with knee OA. They analyzed data from the Bunkyo Health Study, which was conducted at Juntendo University’s Sportology Center to examine the association between metabolic, cardiovascular, cognitive dysfunction, and motor organ disorders in older adults from November 2015 to September 2018.
From the initial population of 1,630 adults, aged 65-84, who did not need medical treatment because of knee pain, the researchers analyzed data from 1,145 adults who the met this study’s criteria, which included MRI imaging of medial type knee OA. A little over half (55.7%) were women, with an average age of 73 and an average body mass index (BMI) of 22.8 kg/m2.
In addition to blood and urine sampling, the researchers determined the severity of knee OA based on joint space width, femorotibial angle, and Kellgren and Lawrence (K/L) grade from x-rays in standing position. They also assessed the structure of knee OA using a whole-organ MRI score (WORMS), and pain and mobility with a visual analog scale, the Japan Knee Osteoarthritis Measure (JKOM), and the 25-question geriatric locomotive function scale.
The JKOM, based on the Western Ontario and McMaster Universities quality of life index for general knee OA, is adjusted to account for the Japanese lifestyle and covers four categories: knee pain and stiffness, a score for activities of daily living, a social activities score, and the patient’s health conditions.
Overall, 41.3% of the participants had sleep disturbances, based on a score of 6 or higher on the Pittsburgh Sleep Quality Index–Japanese. More women (55.7%) than men experienced sleep problems (P < .001), but there were no significant differences in the average age between those who did and those who did not have sleep issues. There were also no significance differences in BMI, joint space width, or femorotibial angle, which was an average 177.5 degrees in group with no sleep problems and 177.6 degrees in the group with sleep disturbances.
The proportion of participants experiencing sleep disturbances increased with increasing K/L grade of OA: 56.8% of those with K/L grade 4 had sleep problems, compared with 40.9% of those with K/L grade 3, 42.1% of those with K/L grade 2, and 33.7% of those with K/L grade 1, resulting in 30% greater odds of sleep disturbance with a higher K/L grade (odds ratio, 1.3; P = .011).
Knee pain at night was also significantly associated with severity of OA based on the K/L grade. While only 6.9% of participants reported pain at night overall, nearly 1 in 3 (29.5%) of those with K/L grade 4 reported pain at night, compared with 3.4% of those with K/L grade 1 (P < .001). (Night pain occurred in 5.4% of those with K/L grade 2 and 16.1% with K/L grade 3.)
However, after adjusting for age, gender, and BMI, the severity of knee OA was not significantly associated with sleep disturbance based on K/L grade, joint space width, femoro-tibial angle, and/or WORMS. But knee pain remained significantly associated with sleep disturbance after adjustment based on the visual analog scale and the JKOM (P < .001 for both).
Sleep problems were also significantly associated with each subcategory of the JKOM after adjustment (P < .001 for all but social activities, which was P = .014).
“Activities of daily living may affect the occurrence of knee pain at night,” Mr. Sasahara said, and “sleep disturbance may also disturb quality of life.” If sleep disturbances related to nighttime knee pain are linked to activities of daily living, then “not only knee pain but also activities of daily living need to be improved in order to improve sleep.”
He noted several of the study’s limitations, including the fact that lifestyle habits and work were not taken into account, nor did the researchers evaluate sleep disturbances potentially resulting from a medical illness. The researchers also only examined knee pain, not pain in other parts of the body.
The research was funded by Juntendo University; the Strategic Research Foundation at Private Universities; KAKENHI from the Ministry of Education, Culture, Sports, Science and Technology of Japan; the Mizuno Sports Promotion Foundation; and the Mitsui Life Social Welfare Foundation. Mr. Sasahara and Ms. Cedin had no disclosures.
DENVER – The relationship between nighttime knee pain from osteoarthritis and sleep disturbances is more complex than a simple association, according to new research presented at the Osteoarthritis Research Society International 2023 World Congress.
The findings suggested that the association between knee OA pain and sleep problems was also linked to activities of daily living, which can contribute to pain but are also affected by OA, Takahiro Sasahara, of the department of orthopedics at Juntendo University, Tokyo, and Koshigaya Municipal Hospital, Saitama, Japan, told attendees. The study also found that knee pain and mobility impairment were associated with sleep disturbances in older adults regardless of the severity of knee OA.
Luisa Cedin, a PhD student at Rush University, Chicago, who attended the presentation, noted the clinical implications of the interaction of daily activities with knee pain.
”I’m a physical therapist, and this could have a significant impact on the performance of the exercises that I’m requiring as a physical therapist,” Ms. Cedin said in an interview. “When you ask somebody who is not getting enough rest during the night – not only enough time but enough quality of rest – we know that we can expect a lower performance with any type of exercises, whether it’s less strength or force, less power, less agility, or less resistance or endurance, so this has a big impact on their quality of life.”
Mr. Sasahara cited research noting that acute pain occurs at the beginning of movement and during weight bearing and walking while chronic pain frequently occurs at night and in early morning awakenings. The prevalence of sleep disturbances in patients with chronic pain ranges from 50% to 80%, he said, and past evidence has shown the relationship between sleep and pain to be bidirectional.
For example, insomnia frequency and severity, sleep-onset problems, and sleep efficiency are all positively associated with pain sensitivity, and increasing severity of OA is linked to increasing prevalence of night knee pain and sleep problems, affecting quality of life, he said.
In this new study examining the relationship between sleep disturbance and knee pain and mobility, the researchers focused specifically on a population of older adults with knee OA. They analyzed data from the Bunkyo Health Study, which was conducted at Juntendo University’s Sportology Center to examine the association between metabolic, cardiovascular, cognitive dysfunction, and motor organ disorders in older adults from November 2015 to September 2018.
From the initial population of 1,630 adults, aged 65-84, who did not need medical treatment because of knee pain, the researchers analyzed data from 1,145 adults who the met this study’s criteria, which included MRI imaging of medial type knee OA. A little over half (55.7%) were women, with an average age of 73 and an average body mass index (BMI) of 22.8 kg/m2.
In addition to blood and urine sampling, the researchers determined the severity of knee OA based on joint space width, femorotibial angle, and Kellgren and Lawrence (K/L) grade from x-rays in standing position. They also assessed the structure of knee OA using a whole-organ MRI score (WORMS), and pain and mobility with a visual analog scale, the Japan Knee Osteoarthritis Measure (JKOM), and the 25-question geriatric locomotive function scale.
The JKOM, based on the Western Ontario and McMaster Universities quality of life index for general knee OA, is adjusted to account for the Japanese lifestyle and covers four categories: knee pain and stiffness, a score for activities of daily living, a social activities score, and the patient’s health conditions.
Overall, 41.3% of the participants had sleep disturbances, based on a score of 6 or higher on the Pittsburgh Sleep Quality Index–Japanese. More women (55.7%) than men experienced sleep problems (P < .001), but there were no significant differences in the average age between those who did and those who did not have sleep issues. There were also no significance differences in BMI, joint space width, or femorotibial angle, which was an average 177.5 degrees in group with no sleep problems and 177.6 degrees in the group with sleep disturbances.
The proportion of participants experiencing sleep disturbances increased with increasing K/L grade of OA: 56.8% of those with K/L grade 4 had sleep problems, compared with 40.9% of those with K/L grade 3, 42.1% of those with K/L grade 2, and 33.7% of those with K/L grade 1, resulting in 30% greater odds of sleep disturbance with a higher K/L grade (odds ratio, 1.3; P = .011).
Knee pain at night was also significantly associated with severity of OA based on the K/L grade. While only 6.9% of participants reported pain at night overall, nearly 1 in 3 (29.5%) of those with K/L grade 4 reported pain at night, compared with 3.4% of those with K/L grade 1 (P < .001). (Night pain occurred in 5.4% of those with K/L grade 2 and 16.1% with K/L grade 3.)
However, after adjusting for age, gender, and BMI, the severity of knee OA was not significantly associated with sleep disturbance based on K/L grade, joint space width, femoro-tibial angle, and/or WORMS. But knee pain remained significantly associated with sleep disturbance after adjustment based on the visual analog scale and the JKOM (P < .001 for both).
Sleep problems were also significantly associated with each subcategory of the JKOM after adjustment (P < .001 for all but social activities, which was P = .014).
“Activities of daily living may affect the occurrence of knee pain at night,” Mr. Sasahara said, and “sleep disturbance may also disturb quality of life.” If sleep disturbances related to nighttime knee pain are linked to activities of daily living, then “not only knee pain but also activities of daily living need to be improved in order to improve sleep.”
He noted several of the study’s limitations, including the fact that lifestyle habits and work were not taken into account, nor did the researchers evaluate sleep disturbances potentially resulting from a medical illness. The researchers also only examined knee pain, not pain in other parts of the body.
The research was funded by Juntendo University; the Strategic Research Foundation at Private Universities; KAKENHI from the Ministry of Education, Culture, Sports, Science and Technology of Japan; the Mizuno Sports Promotion Foundation; and the Mitsui Life Social Welfare Foundation. Mr. Sasahara and Ms. Cedin had no disclosures.
DENVER – The relationship between nighttime knee pain from osteoarthritis and sleep disturbances is more complex than a simple association, according to new research presented at the Osteoarthritis Research Society International 2023 World Congress.
The findings suggested that the association between knee OA pain and sleep problems was also linked to activities of daily living, which can contribute to pain but are also affected by OA, Takahiro Sasahara, of the department of orthopedics at Juntendo University, Tokyo, and Koshigaya Municipal Hospital, Saitama, Japan, told attendees. The study also found that knee pain and mobility impairment were associated with sleep disturbances in older adults regardless of the severity of knee OA.
Luisa Cedin, a PhD student at Rush University, Chicago, who attended the presentation, noted the clinical implications of the interaction of daily activities with knee pain.
”I’m a physical therapist, and this could have a significant impact on the performance of the exercises that I’m requiring as a physical therapist,” Ms. Cedin said in an interview. “When you ask somebody who is not getting enough rest during the night – not only enough time but enough quality of rest – we know that we can expect a lower performance with any type of exercises, whether it’s less strength or force, less power, less agility, or less resistance or endurance, so this has a big impact on their quality of life.”
Mr. Sasahara cited research noting that acute pain occurs at the beginning of movement and during weight bearing and walking while chronic pain frequently occurs at night and in early morning awakenings. The prevalence of sleep disturbances in patients with chronic pain ranges from 50% to 80%, he said, and past evidence has shown the relationship between sleep and pain to be bidirectional.
For example, insomnia frequency and severity, sleep-onset problems, and sleep efficiency are all positively associated with pain sensitivity, and increasing severity of OA is linked to increasing prevalence of night knee pain and sleep problems, affecting quality of life, he said.
In this new study examining the relationship between sleep disturbance and knee pain and mobility, the researchers focused specifically on a population of older adults with knee OA. They analyzed data from the Bunkyo Health Study, which was conducted at Juntendo University’s Sportology Center to examine the association between metabolic, cardiovascular, cognitive dysfunction, and motor organ disorders in older adults from November 2015 to September 2018.
From the initial population of 1,630 adults, aged 65-84, who did not need medical treatment because of knee pain, the researchers analyzed data from 1,145 adults who the met this study’s criteria, which included MRI imaging of medial type knee OA. A little over half (55.7%) were women, with an average age of 73 and an average body mass index (BMI) of 22.8 kg/m2.
In addition to blood and urine sampling, the researchers determined the severity of knee OA based on joint space width, femorotibial angle, and Kellgren and Lawrence (K/L) grade from x-rays in standing position. They also assessed the structure of knee OA using a whole-organ MRI score (WORMS), and pain and mobility with a visual analog scale, the Japan Knee Osteoarthritis Measure (JKOM), and the 25-question geriatric locomotive function scale.
The JKOM, based on the Western Ontario and McMaster Universities quality of life index for general knee OA, is adjusted to account for the Japanese lifestyle and covers four categories: knee pain and stiffness, a score for activities of daily living, a social activities score, and the patient’s health conditions.
Overall, 41.3% of the participants had sleep disturbances, based on a score of 6 or higher on the Pittsburgh Sleep Quality Index–Japanese. More women (55.7%) than men experienced sleep problems (P < .001), but there were no significant differences in the average age between those who did and those who did not have sleep issues. There were also no significance differences in BMI, joint space width, or femorotibial angle, which was an average 177.5 degrees in group with no sleep problems and 177.6 degrees in the group with sleep disturbances.
The proportion of participants experiencing sleep disturbances increased with increasing K/L grade of OA: 56.8% of those with K/L grade 4 had sleep problems, compared with 40.9% of those with K/L grade 3, 42.1% of those with K/L grade 2, and 33.7% of those with K/L grade 1, resulting in 30% greater odds of sleep disturbance with a higher K/L grade (odds ratio, 1.3; P = .011).
Knee pain at night was also significantly associated with severity of OA based on the K/L grade. While only 6.9% of participants reported pain at night overall, nearly 1 in 3 (29.5%) of those with K/L grade 4 reported pain at night, compared with 3.4% of those with K/L grade 1 (P < .001). (Night pain occurred in 5.4% of those with K/L grade 2 and 16.1% with K/L grade 3.)
However, after adjusting for age, gender, and BMI, the severity of knee OA was not significantly associated with sleep disturbance based on K/L grade, joint space width, femoro-tibial angle, and/or WORMS. But knee pain remained significantly associated with sleep disturbance after adjustment based on the visual analog scale and the JKOM (P < .001 for both).
Sleep problems were also significantly associated with each subcategory of the JKOM after adjustment (P < .001 for all but social activities, which was P = .014).
“Activities of daily living may affect the occurrence of knee pain at night,” Mr. Sasahara said, and “sleep disturbance may also disturb quality of life.” If sleep disturbances related to nighttime knee pain are linked to activities of daily living, then “not only knee pain but also activities of daily living need to be improved in order to improve sleep.”
He noted several of the study’s limitations, including the fact that lifestyle habits and work were not taken into account, nor did the researchers evaluate sleep disturbances potentially resulting from a medical illness. The researchers also only examined knee pain, not pain in other parts of the body.
The research was funded by Juntendo University; the Strategic Research Foundation at Private Universities; KAKENHI from the Ministry of Education, Culture, Sports, Science and Technology of Japan; the Mizuno Sports Promotion Foundation; and the Mitsui Life Social Welfare Foundation. Mr. Sasahara and Ms. Cedin had no disclosures.
AT OARSI 2023
Hydroxyurea underused in youth with sickle cell anemia
Even after endorsement in updated guidelines, hydroxyurea is substantially underused in youth with sickle cell anemia (SCA), new research indicates.
SCA can lead to pain crises, stroke, and early death. Hydroxyurea, an oral disease-modifying medication, can reduce the complications.
In 2014, the National Heart, Lung, and Blood Institute published revised guidelines that hydroxyurea should be offered as the primary therapy to all patients who were at least 9 months old and living with SCA, regardless of disease severity.
Low uptake even after guideline revision
Yet, a research team led by Sarah L. Reeves, PhD, MPH, with the Child Health Evaluation and Research Center at University of Michigan, Ann Arbor, found in their study of use in two sample states – Michigan and New York – that hydroxyurea use was low in children and adolescents enrolled in Medicaid and increased only slightly in Michigan and not at all in New York after the guideline revision.
After the guidelines were updated, the researchers observed that, on average, children and adolescents were getting the medication less than a third of the days in a year (32% maximum in the year with the highest uptake). The data were gathered from a study population that included 4,302 youths aged 1-17 years with SCA.
Findings were published online in JAMA Network Open.
‘A national issue’
Russell Ware, MD, PhD, chair of hematology translational research at Cincinnati Children’s Hospital, who was not part of the research, says that though data were gathered from Michigan and New York, “this is a national issue.”
Dr. Ware says the main problem is the way the health system describes the importance of hydroxyurea.
“There needs to be a realization that hydroxyurea is the standard of care for children with sickle cell anemia. It’s not just something they should take when they’re sick,” Dr. Ware said.
He added, “If you have diabetes, should you only take insulin if you’re really sick and hospitalized with a diabetic coma? Of course not.”
He said often providers aren’t giving a clear and consistent message to families.
“They’re not all sure they want to recommend it. They might offer it,” Dr. Ware said, which jeopardizes uptake. “Providers need to be more committed to it. They need to know how to dose it.”
Bad rap from past indications
Dr. Ware says hydroxyurea also gets a bad rap from use decades ago as a chemotherapeutic agent for cancer and then as an anti-HIV medication.
Now it’s used in a completely different way with SCA, but the fear of the association lingers.
“This label as a chemotherapeutic agent has really dogged hydroxyurea,” he said. “It’s a completely different mechanism. It’s a different dose. It’s a different purpose.”
The message to families should be more direct, he says: “Your child has sickle cell anemia and needs to be on disease-modifying therapy because this is a life-threatening disease.”
The underuse of this drug is particularly ironic, he says, as each capsule, taken daily, “costs about fifty cents.”
Medicaid support critical
Authors conclude that multifaceted interventions may be necessary to increase the number of filled prescriptions and use. They also point out that the interventions rely on states’ Medicaid support regarding hydroxyurea use. From 70% to 90% of young people with SCA are covered by Medicaid at some point, the researchers write.
“Variation may exist across states, as well as within states, in the coverage of hydroxyurea, outpatient visits, and associated lab monitoring,” they note.
The authors point to interventions in clinical trials that have had some success in hydroxyurea use.
Creary et al., for example, found that electronic directly observed therapy was associated with high adherence. That involved sending daily texts to patients to take hydroxyurea and patients recording and sending daily videos that show they took the medication.
The authors add that incorporating clinical pharmacists into the care team to provide education and support for families has been shown to be associated with successful outcomes for other chronic conditions – this approach may be particularly well suited to hydroxyurea given that this medication requires significant dosage monitoring.
Dr. Ware, however, says that solutions should focus on the health system more clearly communicating that hydroxyurea is the standard of care for all kids with SCA.
“We need to dispel these myths and these labels that are unfairly attributed to it. Then we’d probably do a lot better,” he said.
He added that children with SCA, “are a marginalized, neglected population of patients historically,” and addressing social determinants of health is also important in getting better uptake.
“Our pharmacy, for example, ships the drug to the families if they’re just getting a refill rather than making them drive all the way in,” Dr. Ware says.
Dr. Ware said given the interruption in doctor/patient relationships in the pandemic, the poor uptake of hydroxyurea could be even worse now.
The work was funded by the Agency for Healthcare Research and Quality and National Heart, Lung, and Blood Institute. Coauthor Dr. Green was the principal investigator of an NIH-funded trial of hydroxyurea in Uganda with a study drug provided by Siklos. No other author disclosures were reported. In addition to receiving research funding from the National Institutes of Health, Dr. Ware receives research donations from Bristol Myers Squibb, Addmedica, and Hemex Health. He is a medical adviser for Nova Laboratories and Octapharma, and serves on Data Safety Monitoring Boards for Novartis and Editas.
Even after endorsement in updated guidelines, hydroxyurea is substantially underused in youth with sickle cell anemia (SCA), new research indicates.
SCA can lead to pain crises, stroke, and early death. Hydroxyurea, an oral disease-modifying medication, can reduce the complications.
In 2014, the National Heart, Lung, and Blood Institute published revised guidelines that hydroxyurea should be offered as the primary therapy to all patients who were at least 9 months old and living with SCA, regardless of disease severity.
Low uptake even after guideline revision
Yet, a research team led by Sarah L. Reeves, PhD, MPH, with the Child Health Evaluation and Research Center at University of Michigan, Ann Arbor, found in their study of use in two sample states – Michigan and New York – that hydroxyurea use was low in children and adolescents enrolled in Medicaid and increased only slightly in Michigan and not at all in New York after the guideline revision.
After the guidelines were updated, the researchers observed that, on average, children and adolescents were getting the medication less than a third of the days in a year (32% maximum in the year with the highest uptake). The data were gathered from a study population that included 4,302 youths aged 1-17 years with SCA.
Findings were published online in JAMA Network Open.
‘A national issue’
Russell Ware, MD, PhD, chair of hematology translational research at Cincinnati Children’s Hospital, who was not part of the research, says that though data were gathered from Michigan and New York, “this is a national issue.”
Dr. Ware says the main problem is the way the health system describes the importance of hydroxyurea.
“There needs to be a realization that hydroxyurea is the standard of care for children with sickle cell anemia. It’s not just something they should take when they’re sick,” Dr. Ware said.
He added, “If you have diabetes, should you only take insulin if you’re really sick and hospitalized with a diabetic coma? Of course not.”
He said often providers aren’t giving a clear and consistent message to families.
“They’re not all sure they want to recommend it. They might offer it,” Dr. Ware said, which jeopardizes uptake. “Providers need to be more committed to it. They need to know how to dose it.”
Bad rap from past indications
Dr. Ware says hydroxyurea also gets a bad rap from use decades ago as a chemotherapeutic agent for cancer and then as an anti-HIV medication.
Now it’s used in a completely different way with SCA, but the fear of the association lingers.
“This label as a chemotherapeutic agent has really dogged hydroxyurea,” he said. “It’s a completely different mechanism. It’s a different dose. It’s a different purpose.”
The message to families should be more direct, he says: “Your child has sickle cell anemia and needs to be on disease-modifying therapy because this is a life-threatening disease.”
The underuse of this drug is particularly ironic, he says, as each capsule, taken daily, “costs about fifty cents.”
Medicaid support critical
Authors conclude that multifaceted interventions may be necessary to increase the number of filled prescriptions and use. They also point out that the interventions rely on states’ Medicaid support regarding hydroxyurea use. From 70% to 90% of young people with SCA are covered by Medicaid at some point, the researchers write.
“Variation may exist across states, as well as within states, in the coverage of hydroxyurea, outpatient visits, and associated lab monitoring,” they note.
The authors point to interventions in clinical trials that have had some success in hydroxyurea use.
Creary et al., for example, found that electronic directly observed therapy was associated with high adherence. That involved sending daily texts to patients to take hydroxyurea and patients recording and sending daily videos that show they took the medication.
The authors add that incorporating clinical pharmacists into the care team to provide education and support for families has been shown to be associated with successful outcomes for other chronic conditions – this approach may be particularly well suited to hydroxyurea given that this medication requires significant dosage monitoring.
Dr. Ware, however, says that solutions should focus on the health system more clearly communicating that hydroxyurea is the standard of care for all kids with SCA.
“We need to dispel these myths and these labels that are unfairly attributed to it. Then we’d probably do a lot better,” he said.
He added that children with SCA, “are a marginalized, neglected population of patients historically,” and addressing social determinants of health is also important in getting better uptake.
“Our pharmacy, for example, ships the drug to the families if they’re just getting a refill rather than making them drive all the way in,” Dr. Ware says.
Dr. Ware said given the interruption in doctor/patient relationships in the pandemic, the poor uptake of hydroxyurea could be even worse now.
The work was funded by the Agency for Healthcare Research and Quality and National Heart, Lung, and Blood Institute. Coauthor Dr. Green was the principal investigator of an NIH-funded trial of hydroxyurea in Uganda with a study drug provided by Siklos. No other author disclosures were reported. In addition to receiving research funding from the National Institutes of Health, Dr. Ware receives research donations from Bristol Myers Squibb, Addmedica, and Hemex Health. He is a medical adviser for Nova Laboratories and Octapharma, and serves on Data Safety Monitoring Boards for Novartis and Editas.
Even after endorsement in updated guidelines, hydroxyurea is substantially underused in youth with sickle cell anemia (SCA), new research indicates.
SCA can lead to pain crises, stroke, and early death. Hydroxyurea, an oral disease-modifying medication, can reduce the complications.
In 2014, the National Heart, Lung, and Blood Institute published revised guidelines that hydroxyurea should be offered as the primary therapy to all patients who were at least 9 months old and living with SCA, regardless of disease severity.
Low uptake even after guideline revision
Yet, a research team led by Sarah L. Reeves, PhD, MPH, with the Child Health Evaluation and Research Center at University of Michigan, Ann Arbor, found in their study of use in two sample states – Michigan and New York – that hydroxyurea use was low in children and adolescents enrolled in Medicaid and increased only slightly in Michigan and not at all in New York after the guideline revision.
After the guidelines were updated, the researchers observed that, on average, children and adolescents were getting the medication less than a third of the days in a year (32% maximum in the year with the highest uptake). The data were gathered from a study population that included 4,302 youths aged 1-17 years with SCA.
Findings were published online in JAMA Network Open.
‘A national issue’
Russell Ware, MD, PhD, chair of hematology translational research at Cincinnati Children’s Hospital, who was not part of the research, says that though data were gathered from Michigan and New York, “this is a national issue.”
Dr. Ware says the main problem is the way the health system describes the importance of hydroxyurea.
“There needs to be a realization that hydroxyurea is the standard of care for children with sickle cell anemia. It’s not just something they should take when they’re sick,” Dr. Ware said.
He added, “If you have diabetes, should you only take insulin if you’re really sick and hospitalized with a diabetic coma? Of course not.”
He said often providers aren’t giving a clear and consistent message to families.
“They’re not all sure they want to recommend it. They might offer it,” Dr. Ware said, which jeopardizes uptake. “Providers need to be more committed to it. They need to know how to dose it.”
Bad rap from past indications
Dr. Ware says hydroxyurea also gets a bad rap from use decades ago as a chemotherapeutic agent for cancer and then as an anti-HIV medication.
Now it’s used in a completely different way with SCA, but the fear of the association lingers.
“This label as a chemotherapeutic agent has really dogged hydroxyurea,” he said. “It’s a completely different mechanism. It’s a different dose. It’s a different purpose.”
The message to families should be more direct, he says: “Your child has sickle cell anemia and needs to be on disease-modifying therapy because this is a life-threatening disease.”
The underuse of this drug is particularly ironic, he says, as each capsule, taken daily, “costs about fifty cents.”
Medicaid support critical
Authors conclude that multifaceted interventions may be necessary to increase the number of filled prescriptions and use. They also point out that the interventions rely on states’ Medicaid support regarding hydroxyurea use. From 70% to 90% of young people with SCA are covered by Medicaid at some point, the researchers write.
“Variation may exist across states, as well as within states, in the coverage of hydroxyurea, outpatient visits, and associated lab monitoring,” they note.
The authors point to interventions in clinical trials that have had some success in hydroxyurea use.
Creary et al., for example, found that electronic directly observed therapy was associated with high adherence. That involved sending daily texts to patients to take hydroxyurea and patients recording and sending daily videos that show they took the medication.
The authors add that incorporating clinical pharmacists into the care team to provide education and support for families has been shown to be associated with successful outcomes for other chronic conditions – this approach may be particularly well suited to hydroxyurea given that this medication requires significant dosage monitoring.
Dr. Ware, however, says that solutions should focus on the health system more clearly communicating that hydroxyurea is the standard of care for all kids with SCA.
“We need to dispel these myths and these labels that are unfairly attributed to it. Then we’d probably do a lot better,” he said.
He added that children with SCA, “are a marginalized, neglected population of patients historically,” and addressing social determinants of health is also important in getting better uptake.
“Our pharmacy, for example, ships the drug to the families if they’re just getting a refill rather than making them drive all the way in,” Dr. Ware says.
Dr. Ware said given the interruption in doctor/patient relationships in the pandemic, the poor uptake of hydroxyurea could be even worse now.
The work was funded by the Agency for Healthcare Research and Quality and National Heart, Lung, and Blood Institute. Coauthor Dr. Green was the principal investigator of an NIH-funded trial of hydroxyurea in Uganda with a study drug provided by Siklos. No other author disclosures were reported. In addition to receiving research funding from the National Institutes of Health, Dr. Ware receives research donations from Bristol Myers Squibb, Addmedica, and Hemex Health. He is a medical adviser for Nova Laboratories and Octapharma, and serves on Data Safety Monitoring Boards for Novartis and Editas.
FROM JAMA NETWORK OPEN
Watch for buprenorphine ‘spiking’ in urine drug tests
Urine drug testing can be valuable for monitoring patients undergoing treatment with buprenorphine for opioid use disorder (OUD). However, some patients alter their test results by adding buprenorphine directly to their urine sample to imply adherence, a new study shows.
“I anticipate a much-needed increase” in the number of people gaining access to buprenorphine therapy, given elimination of the X waiver, first author Jarratt D. Pytell, MD, with University of Colorado at Denver, Aurora, said in a statement.
“New prescribers of buprenorphine will need to learn how to conduct the increasingly complex initiation of treatment and then gauge whether it is successful or not,” added Dr. Pytell, a general internist and addiction medicine specialist.
“Spiking suggests that treatment is not working – especially in patients continuing illicit drug use. Detecting spiking allows clinicians to adjust or intensify the treatment plan,” Dr. Pytell said in an interview.
The study was published online in JAMA Psychiatry.
A sign of elevated patient risk
In a cross-sectional study using Millennium Health’s proprietary urine drug test (UDT) database, researchers analyzed 507,735 urine specimens from 58,476 OUD patients collected between January 2017 and April 2022.
A total of 9546 (1.9%) specimens from 4,550 patients (7.6%) were suggestive of spiking.
UDT specimens suggestive of spiking had two times the odds of being positive for other opioids (fentanyl or heroin), compared with opioid negative samples.
UDT specimens obtained from primary care clinics, from patients aged 35-44 years, and from patients living in the South Atlantic region of the United States were also more likely to be suggestive of buprenorphine spiking.
“Our study demonstrated that a buprenorphine to norbuprenorphine ratio of less than 0.02 indicates the possibility of spiking,” Dr. Pytell said in an interview.
“Nevertheless, it is important to note that this cutoff is not a definitive standard and further controlled studies are necessary to determine its predictive value for spiking. But recognizing possible spiking is very important since it demonstrates a point of elevated risk for the patient and the treatment approach should be reconsidered,” Dr. Pytell said.
“At Millennium Health, we have been tracking the enormity of the drug use crisis. This study suggests that spiking is an important patient safety issue, and it is not uncommon,” study coauthor Eric Dawson, PharmD, vice president of clinical affairs, Millennium Health, said in a statement.
“Detection of spiking requires definitive drug testing. Immunoassay-based, point-of-care tests cannot detect spiking because they are generally incapable of quantitative analysis and differentiating buprenorphine from norbuprenorphine,” Dr. Dawson said.
Best practices?
“We need to develop best practices specific for this situation where a patient has added buprenorphine to the urine drug test specimen,” said Dr. Pytell.
“As with all unexpected findings, it is crucial for clinicians to approach this finding in a nonjudgmental manner and work with the patient to understand what might have motivated them to alter their urine specimen,” he added.
Dr. Pytell said a common reaction for clinicians might be to discontinue treatment. However, this is actually a time to try and engage with the patient.
“Clinicians should work collaboratively with patients to identify potential reasons for spiking and determine what changes may need to be made to better support the patient’s recovery,” Dr. Pytell said.
“This could include more frequent monitoring or referral to a higher level of care. In addition, clinicians should be aware that patients who engage in spiking may be experiencing other challenges that impact their ability to adhere to treatment, such as inadequate housing, mental health issues, or financial strain. Addressing these underlying issues may help patients overcome barriers to treatment adherence and reduce the likelihood of future spiking,” Dr. Pytell said.
This study was supported by Millennium Health. The authors have no relevant disclosures.
A version of this article first appeared on Medscape.com.
Urine drug testing can be valuable for monitoring patients undergoing treatment with buprenorphine for opioid use disorder (OUD). However, some patients alter their test results by adding buprenorphine directly to their urine sample to imply adherence, a new study shows.
“I anticipate a much-needed increase” in the number of people gaining access to buprenorphine therapy, given elimination of the X waiver, first author Jarratt D. Pytell, MD, with University of Colorado at Denver, Aurora, said in a statement.
“New prescribers of buprenorphine will need to learn how to conduct the increasingly complex initiation of treatment and then gauge whether it is successful or not,” added Dr. Pytell, a general internist and addiction medicine specialist.
“Spiking suggests that treatment is not working – especially in patients continuing illicit drug use. Detecting spiking allows clinicians to adjust or intensify the treatment plan,” Dr. Pytell said in an interview.
The study was published online in JAMA Psychiatry.
A sign of elevated patient risk
In a cross-sectional study using Millennium Health’s proprietary urine drug test (UDT) database, researchers analyzed 507,735 urine specimens from 58,476 OUD patients collected between January 2017 and April 2022.
A total of 9546 (1.9%) specimens from 4,550 patients (7.6%) were suggestive of spiking.
UDT specimens suggestive of spiking had two times the odds of being positive for other opioids (fentanyl or heroin), compared with opioid negative samples.
UDT specimens obtained from primary care clinics, from patients aged 35-44 years, and from patients living in the South Atlantic region of the United States were also more likely to be suggestive of buprenorphine spiking.
“Our study demonstrated that a buprenorphine to norbuprenorphine ratio of less than 0.02 indicates the possibility of spiking,” Dr. Pytell said in an interview.
“Nevertheless, it is important to note that this cutoff is not a definitive standard and further controlled studies are necessary to determine its predictive value for spiking. But recognizing possible spiking is very important since it demonstrates a point of elevated risk for the patient and the treatment approach should be reconsidered,” Dr. Pytell said.
“At Millennium Health, we have been tracking the enormity of the drug use crisis. This study suggests that spiking is an important patient safety issue, and it is not uncommon,” study coauthor Eric Dawson, PharmD, vice president of clinical affairs, Millennium Health, said in a statement.
“Detection of spiking requires definitive drug testing. Immunoassay-based, point-of-care tests cannot detect spiking because they are generally incapable of quantitative analysis and differentiating buprenorphine from norbuprenorphine,” Dr. Dawson said.
Best practices?
“We need to develop best practices specific for this situation where a patient has added buprenorphine to the urine drug test specimen,” said Dr. Pytell.
“As with all unexpected findings, it is crucial for clinicians to approach this finding in a nonjudgmental manner and work with the patient to understand what might have motivated them to alter their urine specimen,” he added.
Dr. Pytell said a common reaction for clinicians might be to discontinue treatment. However, this is actually a time to try and engage with the patient.
“Clinicians should work collaboratively with patients to identify potential reasons for spiking and determine what changes may need to be made to better support the patient’s recovery,” Dr. Pytell said.
“This could include more frequent monitoring or referral to a higher level of care. In addition, clinicians should be aware that patients who engage in spiking may be experiencing other challenges that impact their ability to adhere to treatment, such as inadequate housing, mental health issues, or financial strain. Addressing these underlying issues may help patients overcome barriers to treatment adherence and reduce the likelihood of future spiking,” Dr. Pytell said.
This study was supported by Millennium Health. The authors have no relevant disclosures.
A version of this article first appeared on Medscape.com.
Urine drug testing can be valuable for monitoring patients undergoing treatment with buprenorphine for opioid use disorder (OUD). However, some patients alter their test results by adding buprenorphine directly to their urine sample to imply adherence, a new study shows.
“I anticipate a much-needed increase” in the number of people gaining access to buprenorphine therapy, given elimination of the X waiver, first author Jarratt D. Pytell, MD, with University of Colorado at Denver, Aurora, said in a statement.
“New prescribers of buprenorphine will need to learn how to conduct the increasingly complex initiation of treatment and then gauge whether it is successful or not,” added Dr. Pytell, a general internist and addiction medicine specialist.
“Spiking suggests that treatment is not working – especially in patients continuing illicit drug use. Detecting spiking allows clinicians to adjust or intensify the treatment plan,” Dr. Pytell said in an interview.
The study was published online in JAMA Psychiatry.
A sign of elevated patient risk
In a cross-sectional study using Millennium Health’s proprietary urine drug test (UDT) database, researchers analyzed 507,735 urine specimens from 58,476 OUD patients collected between January 2017 and April 2022.
A total of 9546 (1.9%) specimens from 4,550 patients (7.6%) were suggestive of spiking.
UDT specimens suggestive of spiking had two times the odds of being positive for other opioids (fentanyl or heroin), compared with opioid negative samples.
UDT specimens obtained from primary care clinics, from patients aged 35-44 years, and from patients living in the South Atlantic region of the United States were also more likely to be suggestive of buprenorphine spiking.
“Our study demonstrated that a buprenorphine to norbuprenorphine ratio of less than 0.02 indicates the possibility of spiking,” Dr. Pytell said in an interview.
“Nevertheless, it is important to note that this cutoff is not a definitive standard and further controlled studies are necessary to determine its predictive value for spiking. But recognizing possible spiking is very important since it demonstrates a point of elevated risk for the patient and the treatment approach should be reconsidered,” Dr. Pytell said.
“At Millennium Health, we have been tracking the enormity of the drug use crisis. This study suggests that spiking is an important patient safety issue, and it is not uncommon,” study coauthor Eric Dawson, PharmD, vice president of clinical affairs, Millennium Health, said in a statement.
“Detection of spiking requires definitive drug testing. Immunoassay-based, point-of-care tests cannot detect spiking because they are generally incapable of quantitative analysis and differentiating buprenorphine from norbuprenorphine,” Dr. Dawson said.
Best practices?
“We need to develop best practices specific for this situation where a patient has added buprenorphine to the urine drug test specimen,” said Dr. Pytell.
“As with all unexpected findings, it is crucial for clinicians to approach this finding in a nonjudgmental manner and work with the patient to understand what might have motivated them to alter their urine specimen,” he added.
Dr. Pytell said a common reaction for clinicians might be to discontinue treatment. However, this is actually a time to try and engage with the patient.
“Clinicians should work collaboratively with patients to identify potential reasons for spiking and determine what changes may need to be made to better support the patient’s recovery,” Dr. Pytell said.
“This could include more frequent monitoring or referral to a higher level of care. In addition, clinicians should be aware that patients who engage in spiking may be experiencing other challenges that impact their ability to adhere to treatment, such as inadequate housing, mental health issues, or financial strain. Addressing these underlying issues may help patients overcome barriers to treatment adherence and reduce the likelihood of future spiking,” Dr. Pytell said.
This study was supported by Millennium Health. The authors have no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM JAMA PSYCHIATRY