User login
Patient health suffers amid pandemic health care shortages
More than half (56%) of responding clinicians reported seeing a decline in patient health because of delayed or inaccessible care amid the pandemic, according to the results of the latest survey by the Larry A. Green Center and the Primary Care Collaborative. The survey was conducted in mid-October and the results were published online Nov. 17.
In addition, 37% of respondents said their patients with chronic conditions showed “noticeably worse health resulting from the pandemic.” And a resounding 85% said patient mental health had worsened.
“I think it’s worse than we thought,” said Rebecca Etz, PhD, codirector of the Larry Green Center. “It’s the outcome of not sufficiently sending resources to primary care either before or during the pandemic.” According to Dr. Etz, survey respondents noted substantial increases in patient weight gain as well as weight loss, anxiety and depression, sleep issues, domestic abuse, and poor oral and eye health, among others.
One clinician from Pennsylvania wrote: “Patients are becoming sicker during the pandemic. I’m seeing more uncontrolled [diabetes]and new [patients with diabetes]. They prefer telehealth yet [have] no access to glucose monitoring or a blood pressure cuff. I am concerned about patients’ isolation and mental health. People are delaying care.”
Now, with COVID numbers peaking across much of the country, many clinicians are trying to close the gap in care with telehealth – something they’re more prepared to do now than they were in March. Over two-thirds of practices are using telehealth for visits to keep up with patients who have stable chronic conditions, according to the survey.
Over 60% of physicians report using telehealth for mental health visits. But a much smaller number – only 16% of respondents – said their practice had added staff to help manage the rising number of behavioral and mental health cases. About one-third (35%) of practices say they’re not financially able to take on new staff.
“We’ve been looking for more ways for patients to do self-support. A big part of chronic disease is health behaviors,” Alex Krist, MD, MPH, a family doctor in Fairfax, Va., and chairperson of the U.S. Preventive Services Task Force, said in an interview. And unfortunately, on top of limited access to basic care, healthy habits that are essential to managing many chronic conditions have become more difficult and less consistent during the pandemic.
The survey – the 22nd iteration in a series of surveys the Green Center and the Primary Care Collaborative have conducted – received 580 respondents from 47 states and Guam. Over two-thirds of respondents were primary care physicians (MDs and DOs). Over half were owners, partners, or employees of a private practice, 66% of which were family medicine practices. And one fifth of respondents provided care in a rural area.
Funding and support for primary care has been wildly insufficient, Dr. Etz said in an interview. If that doesn’t change, patient health, clinic staffing, and public health strategies amid the pandemic will continue to suffer.
“When you think of the COVID vaccine, who do you think is going to be sending that out?” Dr. Etz asked. “If we don’t bolster primary care now how are they going to handle that.”
A version of this article originally appeared on Medscape.com.
More than half (56%) of responding clinicians reported seeing a decline in patient health because of delayed or inaccessible care amid the pandemic, according to the results of the latest survey by the Larry A. Green Center and the Primary Care Collaborative. The survey was conducted in mid-October and the results were published online Nov. 17.
In addition, 37% of respondents said their patients with chronic conditions showed “noticeably worse health resulting from the pandemic.” And a resounding 85% said patient mental health had worsened.
“I think it’s worse than we thought,” said Rebecca Etz, PhD, codirector of the Larry Green Center. “It’s the outcome of not sufficiently sending resources to primary care either before or during the pandemic.” According to Dr. Etz, survey respondents noted substantial increases in patient weight gain as well as weight loss, anxiety and depression, sleep issues, domestic abuse, and poor oral and eye health, among others.
One clinician from Pennsylvania wrote: “Patients are becoming sicker during the pandemic. I’m seeing more uncontrolled [diabetes]and new [patients with diabetes]. They prefer telehealth yet [have] no access to glucose monitoring or a blood pressure cuff. I am concerned about patients’ isolation and mental health. People are delaying care.”
Now, with COVID numbers peaking across much of the country, many clinicians are trying to close the gap in care with telehealth – something they’re more prepared to do now than they were in March. Over two-thirds of practices are using telehealth for visits to keep up with patients who have stable chronic conditions, according to the survey.
Over 60% of physicians report using telehealth for mental health visits. But a much smaller number – only 16% of respondents – said their practice had added staff to help manage the rising number of behavioral and mental health cases. About one-third (35%) of practices say they’re not financially able to take on new staff.
“We’ve been looking for more ways for patients to do self-support. A big part of chronic disease is health behaviors,” Alex Krist, MD, MPH, a family doctor in Fairfax, Va., and chairperson of the U.S. Preventive Services Task Force, said in an interview. And unfortunately, on top of limited access to basic care, healthy habits that are essential to managing many chronic conditions have become more difficult and less consistent during the pandemic.
The survey – the 22nd iteration in a series of surveys the Green Center and the Primary Care Collaborative have conducted – received 580 respondents from 47 states and Guam. Over two-thirds of respondents were primary care physicians (MDs and DOs). Over half were owners, partners, or employees of a private practice, 66% of which were family medicine practices. And one fifth of respondents provided care in a rural area.
Funding and support for primary care has been wildly insufficient, Dr. Etz said in an interview. If that doesn’t change, patient health, clinic staffing, and public health strategies amid the pandemic will continue to suffer.
“When you think of the COVID vaccine, who do you think is going to be sending that out?” Dr. Etz asked. “If we don’t bolster primary care now how are they going to handle that.”
A version of this article originally appeared on Medscape.com.
More than half (56%) of responding clinicians reported seeing a decline in patient health because of delayed or inaccessible care amid the pandemic, according to the results of the latest survey by the Larry A. Green Center and the Primary Care Collaborative. The survey was conducted in mid-October and the results were published online Nov. 17.
In addition, 37% of respondents said their patients with chronic conditions showed “noticeably worse health resulting from the pandemic.” And a resounding 85% said patient mental health had worsened.
“I think it’s worse than we thought,” said Rebecca Etz, PhD, codirector of the Larry Green Center. “It’s the outcome of not sufficiently sending resources to primary care either before or during the pandemic.” According to Dr. Etz, survey respondents noted substantial increases in patient weight gain as well as weight loss, anxiety and depression, sleep issues, domestic abuse, and poor oral and eye health, among others.
One clinician from Pennsylvania wrote: “Patients are becoming sicker during the pandemic. I’m seeing more uncontrolled [diabetes]and new [patients with diabetes]. They prefer telehealth yet [have] no access to glucose monitoring or a blood pressure cuff. I am concerned about patients’ isolation and mental health. People are delaying care.”
Now, with COVID numbers peaking across much of the country, many clinicians are trying to close the gap in care with telehealth – something they’re more prepared to do now than they were in March. Over two-thirds of practices are using telehealth for visits to keep up with patients who have stable chronic conditions, according to the survey.
Over 60% of physicians report using telehealth for mental health visits. But a much smaller number – only 16% of respondents – said their practice had added staff to help manage the rising number of behavioral and mental health cases. About one-third (35%) of practices say they’re not financially able to take on new staff.
“We’ve been looking for more ways for patients to do self-support. A big part of chronic disease is health behaviors,” Alex Krist, MD, MPH, a family doctor in Fairfax, Va., and chairperson of the U.S. Preventive Services Task Force, said in an interview. And unfortunately, on top of limited access to basic care, healthy habits that are essential to managing many chronic conditions have become more difficult and less consistent during the pandemic.
The survey – the 22nd iteration in a series of surveys the Green Center and the Primary Care Collaborative have conducted – received 580 respondents from 47 states and Guam. Over two-thirds of respondents were primary care physicians (MDs and DOs). Over half were owners, partners, or employees of a private practice, 66% of which were family medicine practices. And one fifth of respondents provided care in a rural area.
Funding and support for primary care has been wildly insufficient, Dr. Etz said in an interview. If that doesn’t change, patient health, clinic staffing, and public health strategies amid the pandemic will continue to suffer.
“When you think of the COVID vaccine, who do you think is going to be sending that out?” Dr. Etz asked. “If we don’t bolster primary care now how are they going to handle that.”
A version of this article originally appeared on Medscape.com.
New study pinpoints how Mediterranean diet reduces diabetes risk
The known reduction in the risk of type 2 diabetes associated with adoption of the Mediterranean diet appears specifically attributed to its beneficial effects on some key factors, a new study published online in JAMA Network Open reveals.
While a reduction in body mass index may be somewhat obvious, other mechanisms include beneficial effects on insulin resistance, lipoprotein metabolism, and inflammation.
However, the diet’s antidiabetes effect does not appear to extend to people whose weight is considered healthy (BMI under 25 kg/m2), according to the findings.
“It is striking to see in these U.S. women how strong the long-term antidiabetic properties of a Mediterranean-type dietary pattern are,” senior author Samia Mora, MD, of the Center for Lipid Metabolomics, Brigham and Women’s Hospital, Harvard Medical School, Boston, said in an interview.
“While it was known that the Mediterranean diet has many health benefits in particular on metabolism and inflammation, it was not previously known which of these biological pathways may be contributing to the lower risk of diabetes and to what magnitude.
“Our findings support the idea that by improving their diet, people can improve their future risk of type 2 diabetes, particularly if they are overweight or have obesity,” she added.
“And it’s important to note that many of these changes don’t happen right away. While metabolism can change over a short period of time, our study indicates that there are longer term changes happening that may provide protection over decades.”
Mediterranean diet reduced diabetes risk in those with BMI ≥ 25 kg/m2
The Mediterranean diet, with an emphasis on healthy olive oil as the predominant source of oil, favors fruits, vegetables, legumes, nuts, seeds, fish, and dairy products, while limiting intake of red and processed meats as well as sweets.
The diet has been linked to as much as a 25%-30% reduction in the risk of diabetes in previous observational studies.
To investigate the precise mechanisms that underlie the prevention of diabetes, lead author Shafqat Ahmad, PhD, also of Harvard, and colleagues examined data from 25,317 healthy women participating in the Women’s Health Study who had baseline assessments between September 1992 and May 1995. They were a mean age of 52.9 years at baseline.
Over the course of the study, 2,307 participants developed type 2 diabetes.
With a mean follow-up of 19.8 years, those who had the highest self-reported adherence to the Mediterranean diet (a score ≥ 6 on a scale of 0-6) at baseline, had as much as a 30% lower risk of developing type 2 diabetes after multivariate adjustments, compared to those with a lower Mediterranean diet score (a score ≤ 3; hazard ratio, 0.70).
The diabetes-related biomarkers that contributed the most to the reduced risk were insulin resistance, accounting for 65% of the reduction, followed by BMI (55.5%), high-density lipoprotein measures (53%), and inflammation (52.5%).
Other factors, though to a lesser degree, included branched-chain amino acids (34.5%), very low-density lipoprotein measures (32.0%), low-density lipoprotein measures (31.0%), blood pressure (29.0%), and apolipoproteins (23.5%).
Differences in hemoglobin A1c levels had a limited effect on the risk (2%).
Notably, a subgroup analysis looking at effects of the diet according to baseline BMI showed the reductions in type 2 diabetes associated with higher intake of the Mediterranean diet extended only to those with an above normal weight (BMI ≥ 25 kg/m2).
Dr. Mora noted that, as this was not a prespecified analysis, these findings should be viewed as hypothesis-generating, but are consistent with the well-known increase in diabetes risk seen with a higher BMI.
“[The finding] fits with the biology and pathogenesis of type 2 diabetes that is driven in large part by excess weight, in particular for visceral adiposity and its resulting metabolic dysregulation and inflammation,” she said.
“We know from other studies, such as the Nurses’ Health Study, that the risk for type 2 diabetes in women increases even at BMI levels below 25 kg/m2, but the risk goes up exponentially at around a BMI of 25 and higher.”
Strong role of insulin resistance a surprise
The strong role of insulin resistance was a surprise, Dr. Mora added.
“We were surprised that insulin resistance, measured by a simple blood biomarker, would have the strongest mediating effect – even stronger than BMI – for the Mediterranean diet on risk of diabetes,” she noted.
“This could represent an opportunity to intervene earlier and more intensively on improving insulin resistance through dietary approaches such as the Mediterranean diet, especially [because] insulin resistance can precede by years and decades the overt hyperglycemia and clinical diagnosis of diabetes.”
Yet another surprise was that A1c had no substantial mediating effect on the reduction of diabetes risk with the Mediterranean diet.
“This could suggest that the cat is out of the bag by the time the A1c rises,” Dr. Mora observed.
A study limitation is that the Women’s Health Study consisted of well-educated U.S. women who were health professionals and predominantly White, so the results may not be generalizable to men or individuals of other races or ethnicities.
In addition, BMI was self-reported and participants were not uniformly screened for diabetes, therefore surveillance bias could be possible.
However, the findings suggest that “even a small increase in adherence to the Mediterranean diet has substantial benefits over many years in preventing diabetes, among many other health benefits such as lowering insulin resistance and inflammation, improving lipid metabolism, and lowering blood pressure,” Mora said.
“And of course, the more the adherence, the more the benefit.”
The study received support through grants from the National Institutes of Health, the National Heart, Lung, and Blood Institute, the National Institute of Diabetes and Digestive and Kidney Diseases, the American Heart Association, and the Molino Family Trust. A coauthor is listed as a coinventor on patents held by Brigham and Women’s Hospital related to the use of inflammatory biomarkers in cardiovascular disease (licensed to AstraZeneca and Siemens).
A version of this article originally appeared on Medscape.com.
The known reduction in the risk of type 2 diabetes associated with adoption of the Mediterranean diet appears specifically attributed to its beneficial effects on some key factors, a new study published online in JAMA Network Open reveals.
While a reduction in body mass index may be somewhat obvious, other mechanisms include beneficial effects on insulin resistance, lipoprotein metabolism, and inflammation.
However, the diet’s antidiabetes effect does not appear to extend to people whose weight is considered healthy (BMI under 25 kg/m2), according to the findings.
“It is striking to see in these U.S. women how strong the long-term antidiabetic properties of a Mediterranean-type dietary pattern are,” senior author Samia Mora, MD, of the Center for Lipid Metabolomics, Brigham and Women’s Hospital, Harvard Medical School, Boston, said in an interview.
“While it was known that the Mediterranean diet has many health benefits in particular on metabolism and inflammation, it was not previously known which of these biological pathways may be contributing to the lower risk of diabetes and to what magnitude.
“Our findings support the idea that by improving their diet, people can improve their future risk of type 2 diabetes, particularly if they are overweight or have obesity,” she added.
“And it’s important to note that many of these changes don’t happen right away. While metabolism can change over a short period of time, our study indicates that there are longer term changes happening that may provide protection over decades.”
Mediterranean diet reduced diabetes risk in those with BMI ≥ 25 kg/m2
The Mediterranean diet, with an emphasis on healthy olive oil as the predominant source of oil, favors fruits, vegetables, legumes, nuts, seeds, fish, and dairy products, while limiting intake of red and processed meats as well as sweets.
The diet has been linked to as much as a 25%-30% reduction in the risk of diabetes in previous observational studies.
To investigate the precise mechanisms that underlie the prevention of diabetes, lead author Shafqat Ahmad, PhD, also of Harvard, and colleagues examined data from 25,317 healthy women participating in the Women’s Health Study who had baseline assessments between September 1992 and May 1995. They were a mean age of 52.9 years at baseline.
Over the course of the study, 2,307 participants developed type 2 diabetes.
With a mean follow-up of 19.8 years, those who had the highest self-reported adherence to the Mediterranean diet (a score ≥ 6 on a scale of 0-6) at baseline, had as much as a 30% lower risk of developing type 2 diabetes after multivariate adjustments, compared to those with a lower Mediterranean diet score (a score ≤ 3; hazard ratio, 0.70).
The diabetes-related biomarkers that contributed the most to the reduced risk were insulin resistance, accounting for 65% of the reduction, followed by BMI (55.5%), high-density lipoprotein measures (53%), and inflammation (52.5%).
Other factors, though to a lesser degree, included branched-chain amino acids (34.5%), very low-density lipoprotein measures (32.0%), low-density lipoprotein measures (31.0%), blood pressure (29.0%), and apolipoproteins (23.5%).
Differences in hemoglobin A1c levels had a limited effect on the risk (2%).
Notably, a subgroup analysis looking at effects of the diet according to baseline BMI showed the reductions in type 2 diabetes associated with higher intake of the Mediterranean diet extended only to those with an above normal weight (BMI ≥ 25 kg/m2).
Dr. Mora noted that, as this was not a prespecified analysis, these findings should be viewed as hypothesis-generating, but are consistent with the well-known increase in diabetes risk seen with a higher BMI.
“[The finding] fits with the biology and pathogenesis of type 2 diabetes that is driven in large part by excess weight, in particular for visceral adiposity and its resulting metabolic dysregulation and inflammation,” she said.
“We know from other studies, such as the Nurses’ Health Study, that the risk for type 2 diabetes in women increases even at BMI levels below 25 kg/m2, but the risk goes up exponentially at around a BMI of 25 and higher.”
Strong role of insulin resistance a surprise
The strong role of insulin resistance was a surprise, Dr. Mora added.
“We were surprised that insulin resistance, measured by a simple blood biomarker, would have the strongest mediating effect – even stronger than BMI – for the Mediterranean diet on risk of diabetes,” she noted.
“This could represent an opportunity to intervene earlier and more intensively on improving insulin resistance through dietary approaches such as the Mediterranean diet, especially [because] insulin resistance can precede by years and decades the overt hyperglycemia and clinical diagnosis of diabetes.”
Yet another surprise was that A1c had no substantial mediating effect on the reduction of diabetes risk with the Mediterranean diet.
“This could suggest that the cat is out of the bag by the time the A1c rises,” Dr. Mora observed.
A study limitation is that the Women’s Health Study consisted of well-educated U.S. women who were health professionals and predominantly White, so the results may not be generalizable to men or individuals of other races or ethnicities.
In addition, BMI was self-reported and participants were not uniformly screened for diabetes, therefore surveillance bias could be possible.
However, the findings suggest that “even a small increase in adherence to the Mediterranean diet has substantial benefits over many years in preventing diabetes, among many other health benefits such as lowering insulin resistance and inflammation, improving lipid metabolism, and lowering blood pressure,” Mora said.
“And of course, the more the adherence, the more the benefit.”
The study received support through grants from the National Institutes of Health, the National Heart, Lung, and Blood Institute, the National Institute of Diabetes and Digestive and Kidney Diseases, the American Heart Association, and the Molino Family Trust. A coauthor is listed as a coinventor on patents held by Brigham and Women’s Hospital related to the use of inflammatory biomarkers in cardiovascular disease (licensed to AstraZeneca and Siemens).
A version of this article originally appeared on Medscape.com.
The known reduction in the risk of type 2 diabetes associated with adoption of the Mediterranean diet appears specifically attributed to its beneficial effects on some key factors, a new study published online in JAMA Network Open reveals.
While a reduction in body mass index may be somewhat obvious, other mechanisms include beneficial effects on insulin resistance, lipoprotein metabolism, and inflammation.
However, the diet’s antidiabetes effect does not appear to extend to people whose weight is considered healthy (BMI under 25 kg/m2), according to the findings.
“It is striking to see in these U.S. women how strong the long-term antidiabetic properties of a Mediterranean-type dietary pattern are,” senior author Samia Mora, MD, of the Center for Lipid Metabolomics, Brigham and Women’s Hospital, Harvard Medical School, Boston, said in an interview.
“While it was known that the Mediterranean diet has many health benefits in particular on metabolism and inflammation, it was not previously known which of these biological pathways may be contributing to the lower risk of diabetes and to what magnitude.
“Our findings support the idea that by improving their diet, people can improve their future risk of type 2 diabetes, particularly if they are overweight or have obesity,” she added.
“And it’s important to note that many of these changes don’t happen right away. While metabolism can change over a short period of time, our study indicates that there are longer term changes happening that may provide protection over decades.”
Mediterranean diet reduced diabetes risk in those with BMI ≥ 25 kg/m2
The Mediterranean diet, with an emphasis on healthy olive oil as the predominant source of oil, favors fruits, vegetables, legumes, nuts, seeds, fish, and dairy products, while limiting intake of red and processed meats as well as sweets.
The diet has been linked to as much as a 25%-30% reduction in the risk of diabetes in previous observational studies.
To investigate the precise mechanisms that underlie the prevention of diabetes, lead author Shafqat Ahmad, PhD, also of Harvard, and colleagues examined data from 25,317 healthy women participating in the Women’s Health Study who had baseline assessments between September 1992 and May 1995. They were a mean age of 52.9 years at baseline.
Over the course of the study, 2,307 participants developed type 2 diabetes.
With a mean follow-up of 19.8 years, those who had the highest self-reported adherence to the Mediterranean diet (a score ≥ 6 on a scale of 0-6) at baseline, had as much as a 30% lower risk of developing type 2 diabetes after multivariate adjustments, compared to those with a lower Mediterranean diet score (a score ≤ 3; hazard ratio, 0.70).
The diabetes-related biomarkers that contributed the most to the reduced risk were insulin resistance, accounting for 65% of the reduction, followed by BMI (55.5%), high-density lipoprotein measures (53%), and inflammation (52.5%).
Other factors, though to a lesser degree, included branched-chain amino acids (34.5%), very low-density lipoprotein measures (32.0%), low-density lipoprotein measures (31.0%), blood pressure (29.0%), and apolipoproteins (23.5%).
Differences in hemoglobin A1c levels had a limited effect on the risk (2%).
Notably, a subgroup analysis looking at effects of the diet according to baseline BMI showed the reductions in type 2 diabetes associated with higher intake of the Mediterranean diet extended only to those with an above normal weight (BMI ≥ 25 kg/m2).
Dr. Mora noted that, as this was not a prespecified analysis, these findings should be viewed as hypothesis-generating, but are consistent with the well-known increase in diabetes risk seen with a higher BMI.
“[The finding] fits with the biology and pathogenesis of type 2 diabetes that is driven in large part by excess weight, in particular for visceral adiposity and its resulting metabolic dysregulation and inflammation,” she said.
“We know from other studies, such as the Nurses’ Health Study, that the risk for type 2 diabetes in women increases even at BMI levels below 25 kg/m2, but the risk goes up exponentially at around a BMI of 25 and higher.”
Strong role of insulin resistance a surprise
The strong role of insulin resistance was a surprise, Dr. Mora added.
“We were surprised that insulin resistance, measured by a simple blood biomarker, would have the strongest mediating effect – even stronger than BMI – for the Mediterranean diet on risk of diabetes,” she noted.
“This could represent an opportunity to intervene earlier and more intensively on improving insulin resistance through dietary approaches such as the Mediterranean diet, especially [because] insulin resistance can precede by years and decades the overt hyperglycemia and clinical diagnosis of diabetes.”
Yet another surprise was that A1c had no substantial mediating effect on the reduction of diabetes risk with the Mediterranean diet.
“This could suggest that the cat is out of the bag by the time the A1c rises,” Dr. Mora observed.
A study limitation is that the Women’s Health Study consisted of well-educated U.S. women who were health professionals and predominantly White, so the results may not be generalizable to men or individuals of other races or ethnicities.
In addition, BMI was self-reported and participants were not uniformly screened for diabetes, therefore surveillance bias could be possible.
However, the findings suggest that “even a small increase in adherence to the Mediterranean diet has substantial benefits over many years in preventing diabetes, among many other health benefits such as lowering insulin resistance and inflammation, improving lipid metabolism, and lowering blood pressure,” Mora said.
“And of course, the more the adherence, the more the benefit.”
The study received support through grants from the National Institutes of Health, the National Heart, Lung, and Blood Institute, the National Institute of Diabetes and Digestive and Kidney Diseases, the American Heart Association, and the Molino Family Trust. A coauthor is listed as a coinventor on patents held by Brigham and Women’s Hospital related to the use of inflammatory biomarkers in cardiovascular disease (licensed to AstraZeneca and Siemens).
A version of this article originally appeared on Medscape.com.
In those with obesity, will losing weight cut COVID-19 severity?
As study after study piles up showing that those with obesity who become infected with SARS-CoV-2 are more likely to have severe disease, several experts gave advice for clinicians and patients during the virtual ObesityWeek Interactive 2020 meeting.
Pichamol Jirapinyo, MD, MPH, associate director of bariatric endoscopy at Brigham and Women’s Hospital, Boston, presented a study on those with obesity from New England hospitals which adds to the evidence that this is “a vulnerable population for COVID-19, like elderly or immunocompromised people,” Dr. Jirapinyo said in an interview.
These findings reinforce the need for clinicians to be “more aware of complications of obesity and refer earlier for treatment,” she added.
One audience member wanted to know if there are data showing whether people with a body mass index (BMI) above 35 kg/m2 who successfully lose weight subsequently have lower rates of hospitalization, ICU admission, and death if they become infected with SARS-CoV-2.
Dr. Jirapinyo said she is not aware of any such studies, but anecdotally, two of her patients who had endoscopic sleeve gastroplasty last fall (whose BMI dropped from about 38 to 30) and later became infected with COVID-19 had mild symptoms.
But David A. Kass, MD, director, Institute of CardioScience at Johns Hopkins University, Baltimore, cautioned that
“Whether this gets reversed by weight loss is an attractive hypothesis, but at this point, it’s still a hypothesis,” he stressed.
Changes to immunity, inflammatory signaling in obesity
“There must be north of 600 or more studies by now with this message that obesity – particularly severe obesity with a BMI of 35 and higher – is a strong independent risk factor for worse COVID-19 outcome,” Dr. Kass emphasized.
“[COVID-19] revealed to the public in a somewhat dramatic fashion that being very obese does put one at higher risk of this disease being more debilitating and even fatal,” he added.
“Before this pandemic, many viewed obesity as only a problem if you have the other associated diseases – hypertension, diabetes, heart disease, atherosclerosis, obstructive sleep apnea, etc.”
“What was not as appreciated is that marked obesity changes the body in various ways all by itself – altering metabolism, inflammatory signaling, immune surveillance, and responsiveness (including a less robust response to vaccines that has been written about as well).”
“This is a bit like having a genetic abnormality that makes you at higher risk for getting, say, cancer,” he explained.
“It is there, it is real, it has an impact – but it still does take other stresses to reveal the risk potential. COVID-19 did that with obesity,” he said.
Latest study on effect of obesity, diabetes on COVID-19 severity
The study presented by Dr. Jirapinyo and colleagues identified 1,680 patients with COVID-19 at six hospitals in March 2020. Patients were a mean age of 51 years, had a mean BMI of 29.4, and 39% had obesity. Patients who required hospitalization were more likely to have obesity (46% vs. 35%; P < .0001).
Obesity was a significant risk factor for hospitalization (odds ratio, 1.7), ICU admission (OR, 1.8), and intubation (OR, 1.8; all P < .001), after controlling for age, sex, cardiovascular, pulmonary, liver, and kidney disease, and cancer.
Compared with having a normal weight, having severe obesity was also associated with roughly threefold higher risks of ICU admission and intubation – after controlling for major comorbidities.
Pandemic focuses minds on obesity prevention, treatment
Naveed Sattar, MD, PhD, said in an interview that these latest findings are “highly consistent with other studies that point to excess adiposity as a potential modifiable risk factor for more severe COVID-19.”
It “also strongly suggests that if people are worried about their risk for COVID-19 and want to improve their chances of a milder outcome, then it is reasonable to encourage them to make sustainable lifestyle changes that may lessen weight and improve their fitness levels,” said Dr. Sattar, professor of metabolic medicine, University of Glasgow.
“But of course, the big worry,” he added, “is that many are putting on weight due to lockdowns, less commuting to work, anxiety, and overeating and drinking, etc., so that many are struggling, and especially those at highest risk, such as those living in more overcrowded housing, etc. By contrast, more advantaged folk may have an easier time to improve lifestyles.”
The pandemic highlights that “we need a concerted effort on obesity prevention and treatment,” according to Dr. Sattar.
“For years we have realized links between obesity and chronic cardiometabolic conditions,” he said, “but to think excess weight may also be detrimental to acute effects of a novel virus running amok in the world has focused minds on obesity in a manner not seen before.
“Whether these new painful learnings lead to a more determined effort in countries to improve the obesogenic environment or to place more resources into prevention and management of obesity remains to be seen,” he said.
Increased inquiries about bariatric surgery following COVID-19
Meanwhile, Matthew M. Hutter, MD, MPH, president, American Society for Metabolic and Bariatric Surgery, said in an interview that “COVID-19 and studies like this are now making many aware that obesity is not just a lifestyle choice or a cosmetic issue, but “a disease that needs to be taken seriously” and treated.
“Metabolic and bariatric surgery is a very safe and effective treatment for persons with obesity with a BMI >40 kg/m2 or BMI >35 kg/m2 and related diseases like diabetes, hypertension, sleep apnea, reflux, back pain, and many others,” added Dr. Hutter, who is also professor of surgery, Harvard Medical School, Boston.
“Recently, some metabolic and bariatric centers have seen an increase in patients considering surgery,” he said. “Some say that COVID-19 has made them realize they need to do something to be healthier.”
“Currently, less than 1% of those who could benefit from surgery are actually having” it each year, Dr. Hutter noted, “and I think there are many who should seriously consider surgery to be healthier, live longer, and live better.”
This article first appeared on Medscape.com.
As study after study piles up showing that those with obesity who become infected with SARS-CoV-2 are more likely to have severe disease, several experts gave advice for clinicians and patients during the virtual ObesityWeek Interactive 2020 meeting.
Pichamol Jirapinyo, MD, MPH, associate director of bariatric endoscopy at Brigham and Women’s Hospital, Boston, presented a study on those with obesity from New England hospitals which adds to the evidence that this is “a vulnerable population for COVID-19, like elderly or immunocompromised people,” Dr. Jirapinyo said in an interview.
These findings reinforce the need for clinicians to be “more aware of complications of obesity and refer earlier for treatment,” she added.
One audience member wanted to know if there are data showing whether people with a body mass index (BMI) above 35 kg/m2 who successfully lose weight subsequently have lower rates of hospitalization, ICU admission, and death if they become infected with SARS-CoV-2.
Dr. Jirapinyo said she is not aware of any such studies, but anecdotally, two of her patients who had endoscopic sleeve gastroplasty last fall (whose BMI dropped from about 38 to 30) and later became infected with COVID-19 had mild symptoms.
But David A. Kass, MD, director, Institute of CardioScience at Johns Hopkins University, Baltimore, cautioned that
“Whether this gets reversed by weight loss is an attractive hypothesis, but at this point, it’s still a hypothesis,” he stressed.
Changes to immunity, inflammatory signaling in obesity
“There must be north of 600 or more studies by now with this message that obesity – particularly severe obesity with a BMI of 35 and higher – is a strong independent risk factor for worse COVID-19 outcome,” Dr. Kass emphasized.
“[COVID-19] revealed to the public in a somewhat dramatic fashion that being very obese does put one at higher risk of this disease being more debilitating and even fatal,” he added.
“Before this pandemic, many viewed obesity as only a problem if you have the other associated diseases – hypertension, diabetes, heart disease, atherosclerosis, obstructive sleep apnea, etc.”
“What was not as appreciated is that marked obesity changes the body in various ways all by itself – altering metabolism, inflammatory signaling, immune surveillance, and responsiveness (including a less robust response to vaccines that has been written about as well).”
“This is a bit like having a genetic abnormality that makes you at higher risk for getting, say, cancer,” he explained.
“It is there, it is real, it has an impact – but it still does take other stresses to reveal the risk potential. COVID-19 did that with obesity,” he said.
Latest study on effect of obesity, diabetes on COVID-19 severity
The study presented by Dr. Jirapinyo and colleagues identified 1,680 patients with COVID-19 at six hospitals in March 2020. Patients were a mean age of 51 years, had a mean BMI of 29.4, and 39% had obesity. Patients who required hospitalization were more likely to have obesity (46% vs. 35%; P < .0001).
Obesity was a significant risk factor for hospitalization (odds ratio, 1.7), ICU admission (OR, 1.8), and intubation (OR, 1.8; all P < .001), after controlling for age, sex, cardiovascular, pulmonary, liver, and kidney disease, and cancer.
Compared with having a normal weight, having severe obesity was also associated with roughly threefold higher risks of ICU admission and intubation – after controlling for major comorbidities.
Pandemic focuses minds on obesity prevention, treatment
Naveed Sattar, MD, PhD, said in an interview that these latest findings are “highly consistent with other studies that point to excess adiposity as a potential modifiable risk factor for more severe COVID-19.”
It “also strongly suggests that if people are worried about their risk for COVID-19 and want to improve their chances of a milder outcome, then it is reasonable to encourage them to make sustainable lifestyle changes that may lessen weight and improve their fitness levels,” said Dr. Sattar, professor of metabolic medicine, University of Glasgow.
“But of course, the big worry,” he added, “is that many are putting on weight due to lockdowns, less commuting to work, anxiety, and overeating and drinking, etc., so that many are struggling, and especially those at highest risk, such as those living in more overcrowded housing, etc. By contrast, more advantaged folk may have an easier time to improve lifestyles.”
The pandemic highlights that “we need a concerted effort on obesity prevention and treatment,” according to Dr. Sattar.
“For years we have realized links between obesity and chronic cardiometabolic conditions,” he said, “but to think excess weight may also be detrimental to acute effects of a novel virus running amok in the world has focused minds on obesity in a manner not seen before.
“Whether these new painful learnings lead to a more determined effort in countries to improve the obesogenic environment or to place more resources into prevention and management of obesity remains to be seen,” he said.
Increased inquiries about bariatric surgery following COVID-19
Meanwhile, Matthew M. Hutter, MD, MPH, president, American Society for Metabolic and Bariatric Surgery, said in an interview that “COVID-19 and studies like this are now making many aware that obesity is not just a lifestyle choice or a cosmetic issue, but “a disease that needs to be taken seriously” and treated.
“Metabolic and bariatric surgery is a very safe and effective treatment for persons with obesity with a BMI >40 kg/m2 or BMI >35 kg/m2 and related diseases like diabetes, hypertension, sleep apnea, reflux, back pain, and many others,” added Dr. Hutter, who is also professor of surgery, Harvard Medical School, Boston.
“Recently, some metabolic and bariatric centers have seen an increase in patients considering surgery,” he said. “Some say that COVID-19 has made them realize they need to do something to be healthier.”
“Currently, less than 1% of those who could benefit from surgery are actually having” it each year, Dr. Hutter noted, “and I think there are many who should seriously consider surgery to be healthier, live longer, and live better.”
This article first appeared on Medscape.com.
As study after study piles up showing that those with obesity who become infected with SARS-CoV-2 are more likely to have severe disease, several experts gave advice for clinicians and patients during the virtual ObesityWeek Interactive 2020 meeting.
Pichamol Jirapinyo, MD, MPH, associate director of bariatric endoscopy at Brigham and Women’s Hospital, Boston, presented a study on those with obesity from New England hospitals which adds to the evidence that this is “a vulnerable population for COVID-19, like elderly or immunocompromised people,” Dr. Jirapinyo said in an interview.
These findings reinforce the need for clinicians to be “more aware of complications of obesity and refer earlier for treatment,” she added.
One audience member wanted to know if there are data showing whether people with a body mass index (BMI) above 35 kg/m2 who successfully lose weight subsequently have lower rates of hospitalization, ICU admission, and death if they become infected with SARS-CoV-2.
Dr. Jirapinyo said she is not aware of any such studies, but anecdotally, two of her patients who had endoscopic sleeve gastroplasty last fall (whose BMI dropped from about 38 to 30) and later became infected with COVID-19 had mild symptoms.
But David A. Kass, MD, director, Institute of CardioScience at Johns Hopkins University, Baltimore, cautioned that
“Whether this gets reversed by weight loss is an attractive hypothesis, but at this point, it’s still a hypothesis,” he stressed.
Changes to immunity, inflammatory signaling in obesity
“There must be north of 600 or more studies by now with this message that obesity – particularly severe obesity with a BMI of 35 and higher – is a strong independent risk factor for worse COVID-19 outcome,” Dr. Kass emphasized.
“[COVID-19] revealed to the public in a somewhat dramatic fashion that being very obese does put one at higher risk of this disease being more debilitating and even fatal,” he added.
“Before this pandemic, many viewed obesity as only a problem if you have the other associated diseases – hypertension, diabetes, heart disease, atherosclerosis, obstructive sleep apnea, etc.”
“What was not as appreciated is that marked obesity changes the body in various ways all by itself – altering metabolism, inflammatory signaling, immune surveillance, and responsiveness (including a less robust response to vaccines that has been written about as well).”
“This is a bit like having a genetic abnormality that makes you at higher risk for getting, say, cancer,” he explained.
“It is there, it is real, it has an impact – but it still does take other stresses to reveal the risk potential. COVID-19 did that with obesity,” he said.
Latest study on effect of obesity, diabetes on COVID-19 severity
The study presented by Dr. Jirapinyo and colleagues identified 1,680 patients with COVID-19 at six hospitals in March 2020. Patients were a mean age of 51 years, had a mean BMI of 29.4, and 39% had obesity. Patients who required hospitalization were more likely to have obesity (46% vs. 35%; P < .0001).
Obesity was a significant risk factor for hospitalization (odds ratio, 1.7), ICU admission (OR, 1.8), and intubation (OR, 1.8; all P < .001), after controlling for age, sex, cardiovascular, pulmonary, liver, and kidney disease, and cancer.
Compared with having a normal weight, having severe obesity was also associated with roughly threefold higher risks of ICU admission and intubation – after controlling for major comorbidities.
Pandemic focuses minds on obesity prevention, treatment
Naveed Sattar, MD, PhD, said in an interview that these latest findings are “highly consistent with other studies that point to excess adiposity as a potential modifiable risk factor for more severe COVID-19.”
It “also strongly suggests that if people are worried about their risk for COVID-19 and want to improve their chances of a milder outcome, then it is reasonable to encourage them to make sustainable lifestyle changes that may lessen weight and improve their fitness levels,” said Dr. Sattar, professor of metabolic medicine, University of Glasgow.
“But of course, the big worry,” he added, “is that many are putting on weight due to lockdowns, less commuting to work, anxiety, and overeating and drinking, etc., so that many are struggling, and especially those at highest risk, such as those living in more overcrowded housing, etc. By contrast, more advantaged folk may have an easier time to improve lifestyles.”
The pandemic highlights that “we need a concerted effort on obesity prevention and treatment,” according to Dr. Sattar.
“For years we have realized links between obesity and chronic cardiometabolic conditions,” he said, “but to think excess weight may also be detrimental to acute effects of a novel virus running amok in the world has focused minds on obesity in a manner not seen before.
“Whether these new painful learnings lead to a more determined effort in countries to improve the obesogenic environment or to place more resources into prevention and management of obesity remains to be seen,” he said.
Increased inquiries about bariatric surgery following COVID-19
Meanwhile, Matthew M. Hutter, MD, MPH, president, American Society for Metabolic and Bariatric Surgery, said in an interview that “COVID-19 and studies like this are now making many aware that obesity is not just a lifestyle choice or a cosmetic issue, but “a disease that needs to be taken seriously” and treated.
“Metabolic and bariatric surgery is a very safe and effective treatment for persons with obesity with a BMI >40 kg/m2 or BMI >35 kg/m2 and related diseases like diabetes, hypertension, sleep apnea, reflux, back pain, and many others,” added Dr. Hutter, who is also professor of surgery, Harvard Medical School, Boston.
“Recently, some metabolic and bariatric centers have seen an increase in patients considering surgery,” he said. “Some say that COVID-19 has made them realize they need to do something to be healthier.”
“Currently, less than 1% of those who could benefit from surgery are actually having” it each year, Dr. Hutter noted, “and I think there are many who should seriously consider surgery to be healthier, live longer, and live better.”
This article first appeared on Medscape.com.
Major breakthrough? Average 10% weight loss with semaglutide
In a phase 3 trial where all participants received intensive behavior therapy, investigational 2.4-mg once-weekly subcutaneous semaglutide (Novo Nordisk) resulted in a 10.3% greater average weight loss than placebo over a period of 68 weeks.
If approved, this medication could be a “potential major breakthrough” in obesity management, the investigators suggested. But other experts urged caution, as cost and uptake are important considerations.
‘Potential weight loss that patients would be happy with’
Thomas A. Wadden, PhD, presented results from the study of 611 adults with overweight or obesity but no diabetes at the virtual ObesityWeek® Interactive 2020 meeting.
“Perhaps even more impressive was the finding that 75% of patients lost 10% or more of baseline body weight,” said Dr. Wadden, of the department of psychiatry at the University of Pennsylvania, Philadelphia.
Moreover, in this trial of semaglutide, a glucagonlike peptide–1 (GLP-1) receptor agonist that is approved for treating type 2 diabetes at a weekly subcutaneous dose of 1 mg, but is being investigated at the higher dose for weight loss – 55% of patients lost ≥15% of their initial weight, and 36% lost ≥20% of their initial weight.
“These large categorical weight losses – particularly of 15% and 20% of initial weight – are potentially a major breakthrough in the management of obesity,” Dr. Wadden said in an interview.
Weight losses of this size, he added, “should confer greater improvements in cardiometabolic risk factors (such as hypertension, sleep apnea, and type 2 diabetes) as compared with losses of 5%-10% achieved with current behavioral or pharmacological approaches.” And patients are generally not satisfied with losses of less than 10% of initial weight when participating in intensive behavior programs or taking weight-loss medications.
Now, “the larger categorical weight losses will mean that a greater number of patients with obesity will be able to achieve a weight loss with which they are ... happy,” Dr. Wadden said in an interview.
According to Louis J. Aronne, MD, Weill Professor of Metabolic Research, Weill Cornell Medicine, New York, who is an investigator for another trial of semaglutide: “Even though it has the same mechanism of action [as liraglutide], the weight loss is two or more times greater [with semaglutide]. In my opinion, it’s really going to be a major advance in the treatment of obesity.”
In the discussion that followed the virtual presentation, one attendee asked about potential weight regain if a patient stopped taking the drug. Based on experience with another subcutaneous injectable GLP-1 receptor agonist, liraglutide (Saxenda), already approved for obesity, it may be that taking medicine for chronic overweight may become like taking a statin for elevated cholesterol, said Dr. Wadden.
Novo Nordisk has now completed the four trials in the STEP (Semaglutide Treatment Effect in People With Obesity) global phase 3 clinical development program, and plans to file applications with the Food and Drug Administration later this year and with the European Medicines Agency in early 2021 for review of semaglutide 2.4 mg for weight management.
“Fundamental issues need to be figured out”
Invited to comment, Scott Kahan, MD, said: “This is impressive data, confirming that semaglutide, particularly when used in concert with evidence-based counseling, is a highly effective agent for obesity management.”
However, “the real question, though, is what comes next,” stressed Dr. Kahan, director of the National Center for Weight and Wellness, Washington, DC.
“Will it be approved by the U.S. FDA? I believe so,” he said in an interview. “Yet we already have several effective obesity medications approved over the past decade – all of which are rarely used and therefore make little impact for patients in the real world.”
“Will there be insurance coverage, and therefore practical access for those who could most benefit?” he continued. “Will prescribers counsel their patients about obesity management, including the use of effective medications? Will patients utilize available options?”
“These and other fundamental issues must be figured out before we anoint any treatment option as a meaningful step forward, let alone a transformative development,” according to Dr. Kahan.
Similarly, Irl B. Hirsch, MD, stressed that, should this medication be approved for weight loss, cost would be a major factor in its uptake.
“I’m old enough to recall when we started using lovastatin in the late 1980s,” Dr. Hirsch, professor of medicine, University of Washington Medicine Diabetes Institute, Seattle, said in an interview.
“We used it without the type of evidence of statin use we have today. A pill, but in those days the statins were expensive. But over time, the evidence for statins grew and over the next 15 years it was quite clear that for both primary prevention (for those with diabetes) and secondary intervention these drugs needed to be used by millions of people. These recommendations became easier once the drugs became generic.
“Will the same thing happen for GLP-1 agonists? The problem is we need both ‘hard-outcome data’ [such as 3-point major adverse cardiovascular events] and more reasonable cost before we see this expanding to an entire population.
“In the future perhaps we could have a biosimilar GLP-1 agonist that would be more affordable than what we pay now, but even before that we need agreement from our reimbursement thought leaders that our society should reimburse these agents.
“My thinking now is the cost-benefit could be favorable, but this is all dependent on what happens to the cost of the drugs over time,” he said.
Additive effect of intensive behavior therapy plus medication
Dr. Wadden explained that intensive behavioral therapy “provides 14 or more counseling sessions in 6 months to modify diet and physical activity, through the patients’ use of behavioral strategies (such as keeping daily food and activity diaries).”
Such programs typically produce mean weight loss of 5%-8% of initial weight; less frequent (e.g., monthly) programs typically produce weight loss of only 1%-3%.
Prior studies suggest that intensive behavioral therapy and medication have additive effects. To investigate this, Dr. Wadden and colleagues randomized 611 adults (81% women) who were a mean age of 46 years and had a mean body mass index of 38 kg/m2.
All participants received 30 intensive behavior therapy sessions provided by a registered dietitian (or other qualified provider), which typically lasted 20-30 minutes and were given weekly for 12 weeks, every other week for the next 12 weeks, and then monthly.
The dietitian gave participants behavioral strategies to help them adhere to diet and physical activity goals.
During the first 8 weeks, participants were provided with a 1,000-1,200 kcal/day meal replacement diet that included liquid shakes, meal bars, and prepared entrees designed to facilitate a large initial weight loss.
They then transitioned to a diet of conventional foods (of their choosing), with a goal of 1,200-1,800 kcal/day based on body weight.
The physical activity goal was 100 minutes/week of walking or other aerobic activity in the first month, building up to 200 minutes/week by month 6.
‘More effective than current FDA-approved weight-loss medications’
At week 68, mean body weight decreased from baseline by 16.0% in the semaglutide group versus 5.7% in the placebo group (P < .0001).
In this trial, where all participants received extensive intensive behavior therapy, more participants had weight loss ≥5%, ≥10%, ≥15%, and ≥20% of their initial weight with semaglutide versus placebo (87% vs. 48%; 75% vs. 27%; 56% vs. 13%; 36% vs. 4%, respectively; all P < .0001).
From baseline to week 68, the proportion of participants with prediabetes decreased from 48% to 7% in the semaglutide group and from 53% to 26% in the placebo group.
Patients who received semaglutide had greater improvements in lipids, too.
Although the weight loss was 10.3% (10.6 kg) greater with semaglutide, Dr. Wadden noted, “additional studies have shown this net benefit to be as great as 11%-12%, which would make semaglutide 2.4 mg more effective than current [FDA-approved] weight-loss medications.”
“Naltrexone-bupropion (Contrave) with lifestyle counseling, for example,” he continued, “produces a loss that is 5 kg greater than lifestyle counseling plus placebo, liraglutide 3.0 mg (Saxenda) a loss 5.3 kg greater than placebo, and phentermine-topiramate (Qsymia) a loss that is 8.8 kg greater than placebo.”
Semaglutide was well tolerated. Gastrointestinal adverse events, the most common type, occurred in 83% of patients in the semaglutide group and 63% of patients in the placebo group.
Nausea, as well as constipation and diarrhea, are common in medications that increase GLP-1 levels, Dr. Wadden noted. Side effects can be managed by slowly increasing the medication dose over 4 months.
Dr. Wadden expects that, if approved, semaglutide 2.4 mg subcutaneous once-weekly will be recommended as an adjunct to a reduced calorie diet and increased physical activity. Additional studies suggest that monthly counseling should be sufficient to obtain similar weight losses as those seen in the current trial, which had more intensive counseling.
As well as being approved as a weekly subcutaneous injectable treatment for type 2 diabetes, semaglutide is also approved as an once-daily oral agent for the same indication (Rybelsus, Novo Nordisk) in doses of 7 mg and 14 mg to improve glycemic control along with diet and exercise. It is the first GLP-1 agonist available in tablet form.
Dr. Wadden serves on scientific advisory boards for Novo Nordisk and WW (formerly Weight Watchers), and has received grant support, on behalf of the University of Pennsylvania, from Novo Nordisk. Dr. Aronne is an investigator in a long-term trial of semaglutide and has served on scientific advisory boards for Novo Nordisk in the past. He also has other industry relationships that are not related to semaglutide.
A version of this article originally appeared on Medscape.com.
In a phase 3 trial where all participants received intensive behavior therapy, investigational 2.4-mg once-weekly subcutaneous semaglutide (Novo Nordisk) resulted in a 10.3% greater average weight loss than placebo over a period of 68 weeks.
If approved, this medication could be a “potential major breakthrough” in obesity management, the investigators suggested. But other experts urged caution, as cost and uptake are important considerations.
‘Potential weight loss that patients would be happy with’
Thomas A. Wadden, PhD, presented results from the study of 611 adults with overweight or obesity but no diabetes at the virtual ObesityWeek® Interactive 2020 meeting.
“Perhaps even more impressive was the finding that 75% of patients lost 10% or more of baseline body weight,” said Dr. Wadden, of the department of psychiatry at the University of Pennsylvania, Philadelphia.
Moreover, in this trial of semaglutide, a glucagonlike peptide–1 (GLP-1) receptor agonist that is approved for treating type 2 diabetes at a weekly subcutaneous dose of 1 mg, but is being investigated at the higher dose for weight loss – 55% of patients lost ≥15% of their initial weight, and 36% lost ≥20% of their initial weight.
“These large categorical weight losses – particularly of 15% and 20% of initial weight – are potentially a major breakthrough in the management of obesity,” Dr. Wadden said in an interview.
Weight losses of this size, he added, “should confer greater improvements in cardiometabolic risk factors (such as hypertension, sleep apnea, and type 2 diabetes) as compared with losses of 5%-10% achieved with current behavioral or pharmacological approaches.” And patients are generally not satisfied with losses of less than 10% of initial weight when participating in intensive behavior programs or taking weight-loss medications.
Now, “the larger categorical weight losses will mean that a greater number of patients with obesity will be able to achieve a weight loss with which they are ... happy,” Dr. Wadden said in an interview.
According to Louis J. Aronne, MD, Weill Professor of Metabolic Research, Weill Cornell Medicine, New York, who is an investigator for another trial of semaglutide: “Even though it has the same mechanism of action [as liraglutide], the weight loss is two or more times greater [with semaglutide]. In my opinion, it’s really going to be a major advance in the treatment of obesity.”
In the discussion that followed the virtual presentation, one attendee asked about potential weight regain if a patient stopped taking the drug. Based on experience with another subcutaneous injectable GLP-1 receptor agonist, liraglutide (Saxenda), already approved for obesity, it may be that taking medicine for chronic overweight may become like taking a statin for elevated cholesterol, said Dr. Wadden.
Novo Nordisk has now completed the four trials in the STEP (Semaglutide Treatment Effect in People With Obesity) global phase 3 clinical development program, and plans to file applications with the Food and Drug Administration later this year and with the European Medicines Agency in early 2021 for review of semaglutide 2.4 mg for weight management.
“Fundamental issues need to be figured out”
Invited to comment, Scott Kahan, MD, said: “This is impressive data, confirming that semaglutide, particularly when used in concert with evidence-based counseling, is a highly effective agent for obesity management.”
However, “the real question, though, is what comes next,” stressed Dr. Kahan, director of the National Center for Weight and Wellness, Washington, DC.
“Will it be approved by the U.S. FDA? I believe so,” he said in an interview. “Yet we already have several effective obesity medications approved over the past decade – all of which are rarely used and therefore make little impact for patients in the real world.”
“Will there be insurance coverage, and therefore practical access for those who could most benefit?” he continued. “Will prescribers counsel their patients about obesity management, including the use of effective medications? Will patients utilize available options?”
“These and other fundamental issues must be figured out before we anoint any treatment option as a meaningful step forward, let alone a transformative development,” according to Dr. Kahan.
Similarly, Irl B. Hirsch, MD, stressed that, should this medication be approved for weight loss, cost would be a major factor in its uptake.
“I’m old enough to recall when we started using lovastatin in the late 1980s,” Dr. Hirsch, professor of medicine, University of Washington Medicine Diabetes Institute, Seattle, said in an interview.
“We used it without the type of evidence of statin use we have today. A pill, but in those days the statins were expensive. But over time, the evidence for statins grew and over the next 15 years it was quite clear that for both primary prevention (for those with diabetes) and secondary intervention these drugs needed to be used by millions of people. These recommendations became easier once the drugs became generic.
“Will the same thing happen for GLP-1 agonists? The problem is we need both ‘hard-outcome data’ [such as 3-point major adverse cardiovascular events] and more reasonable cost before we see this expanding to an entire population.
“In the future perhaps we could have a biosimilar GLP-1 agonist that would be more affordable than what we pay now, but even before that we need agreement from our reimbursement thought leaders that our society should reimburse these agents.
“My thinking now is the cost-benefit could be favorable, but this is all dependent on what happens to the cost of the drugs over time,” he said.
Additive effect of intensive behavior therapy plus medication
Dr. Wadden explained that intensive behavioral therapy “provides 14 or more counseling sessions in 6 months to modify diet and physical activity, through the patients’ use of behavioral strategies (such as keeping daily food and activity diaries).”
Such programs typically produce mean weight loss of 5%-8% of initial weight; less frequent (e.g., monthly) programs typically produce weight loss of only 1%-3%.
Prior studies suggest that intensive behavioral therapy and medication have additive effects. To investigate this, Dr. Wadden and colleagues randomized 611 adults (81% women) who were a mean age of 46 years and had a mean body mass index of 38 kg/m2.
All participants received 30 intensive behavior therapy sessions provided by a registered dietitian (or other qualified provider), which typically lasted 20-30 minutes and were given weekly for 12 weeks, every other week for the next 12 weeks, and then monthly.
The dietitian gave participants behavioral strategies to help them adhere to diet and physical activity goals.
During the first 8 weeks, participants were provided with a 1,000-1,200 kcal/day meal replacement diet that included liquid shakes, meal bars, and prepared entrees designed to facilitate a large initial weight loss.
They then transitioned to a diet of conventional foods (of their choosing), with a goal of 1,200-1,800 kcal/day based on body weight.
The physical activity goal was 100 minutes/week of walking or other aerobic activity in the first month, building up to 200 minutes/week by month 6.
‘More effective than current FDA-approved weight-loss medications’
At week 68, mean body weight decreased from baseline by 16.0% in the semaglutide group versus 5.7% in the placebo group (P < .0001).
In this trial, where all participants received extensive intensive behavior therapy, more participants had weight loss ≥5%, ≥10%, ≥15%, and ≥20% of their initial weight with semaglutide versus placebo (87% vs. 48%; 75% vs. 27%; 56% vs. 13%; 36% vs. 4%, respectively; all P < .0001).
From baseline to week 68, the proportion of participants with prediabetes decreased from 48% to 7% in the semaglutide group and from 53% to 26% in the placebo group.
Patients who received semaglutide had greater improvements in lipids, too.
Although the weight loss was 10.3% (10.6 kg) greater with semaglutide, Dr. Wadden noted, “additional studies have shown this net benefit to be as great as 11%-12%, which would make semaglutide 2.4 mg more effective than current [FDA-approved] weight-loss medications.”
“Naltrexone-bupropion (Contrave) with lifestyle counseling, for example,” he continued, “produces a loss that is 5 kg greater than lifestyle counseling plus placebo, liraglutide 3.0 mg (Saxenda) a loss 5.3 kg greater than placebo, and phentermine-topiramate (Qsymia) a loss that is 8.8 kg greater than placebo.”
Semaglutide was well tolerated. Gastrointestinal adverse events, the most common type, occurred in 83% of patients in the semaglutide group and 63% of patients in the placebo group.
Nausea, as well as constipation and diarrhea, are common in medications that increase GLP-1 levels, Dr. Wadden noted. Side effects can be managed by slowly increasing the medication dose over 4 months.
Dr. Wadden expects that, if approved, semaglutide 2.4 mg subcutaneous once-weekly will be recommended as an adjunct to a reduced calorie diet and increased physical activity. Additional studies suggest that monthly counseling should be sufficient to obtain similar weight losses as those seen in the current trial, which had more intensive counseling.
As well as being approved as a weekly subcutaneous injectable treatment for type 2 diabetes, semaglutide is also approved as an once-daily oral agent for the same indication (Rybelsus, Novo Nordisk) in doses of 7 mg and 14 mg to improve glycemic control along with diet and exercise. It is the first GLP-1 agonist available in tablet form.
Dr. Wadden serves on scientific advisory boards for Novo Nordisk and WW (formerly Weight Watchers), and has received grant support, on behalf of the University of Pennsylvania, from Novo Nordisk. Dr. Aronne is an investigator in a long-term trial of semaglutide and has served on scientific advisory boards for Novo Nordisk in the past. He also has other industry relationships that are not related to semaglutide.
A version of this article originally appeared on Medscape.com.
In a phase 3 trial where all participants received intensive behavior therapy, investigational 2.4-mg once-weekly subcutaneous semaglutide (Novo Nordisk) resulted in a 10.3% greater average weight loss than placebo over a period of 68 weeks.
If approved, this medication could be a “potential major breakthrough” in obesity management, the investigators suggested. But other experts urged caution, as cost and uptake are important considerations.
‘Potential weight loss that patients would be happy with’
Thomas A. Wadden, PhD, presented results from the study of 611 adults with overweight or obesity but no diabetes at the virtual ObesityWeek® Interactive 2020 meeting.
“Perhaps even more impressive was the finding that 75% of patients lost 10% or more of baseline body weight,” said Dr. Wadden, of the department of psychiatry at the University of Pennsylvania, Philadelphia.
Moreover, in this trial of semaglutide, a glucagonlike peptide–1 (GLP-1) receptor agonist that is approved for treating type 2 diabetes at a weekly subcutaneous dose of 1 mg, but is being investigated at the higher dose for weight loss – 55% of patients lost ≥15% of their initial weight, and 36% lost ≥20% of their initial weight.
“These large categorical weight losses – particularly of 15% and 20% of initial weight – are potentially a major breakthrough in the management of obesity,” Dr. Wadden said in an interview.
Weight losses of this size, he added, “should confer greater improvements in cardiometabolic risk factors (such as hypertension, sleep apnea, and type 2 diabetes) as compared with losses of 5%-10% achieved with current behavioral or pharmacological approaches.” And patients are generally not satisfied with losses of less than 10% of initial weight when participating in intensive behavior programs or taking weight-loss medications.
Now, “the larger categorical weight losses will mean that a greater number of patients with obesity will be able to achieve a weight loss with which they are ... happy,” Dr. Wadden said in an interview.
According to Louis J. Aronne, MD, Weill Professor of Metabolic Research, Weill Cornell Medicine, New York, who is an investigator for another trial of semaglutide: “Even though it has the same mechanism of action [as liraglutide], the weight loss is two or more times greater [with semaglutide]. In my opinion, it’s really going to be a major advance in the treatment of obesity.”
In the discussion that followed the virtual presentation, one attendee asked about potential weight regain if a patient stopped taking the drug. Based on experience with another subcutaneous injectable GLP-1 receptor agonist, liraglutide (Saxenda), already approved for obesity, it may be that taking medicine for chronic overweight may become like taking a statin for elevated cholesterol, said Dr. Wadden.
Novo Nordisk has now completed the four trials in the STEP (Semaglutide Treatment Effect in People With Obesity) global phase 3 clinical development program, and plans to file applications with the Food and Drug Administration later this year and with the European Medicines Agency in early 2021 for review of semaglutide 2.4 mg for weight management.
“Fundamental issues need to be figured out”
Invited to comment, Scott Kahan, MD, said: “This is impressive data, confirming that semaglutide, particularly when used in concert with evidence-based counseling, is a highly effective agent for obesity management.”
However, “the real question, though, is what comes next,” stressed Dr. Kahan, director of the National Center for Weight and Wellness, Washington, DC.
“Will it be approved by the U.S. FDA? I believe so,” he said in an interview. “Yet we already have several effective obesity medications approved over the past decade – all of which are rarely used and therefore make little impact for patients in the real world.”
“Will there be insurance coverage, and therefore practical access for those who could most benefit?” he continued. “Will prescribers counsel their patients about obesity management, including the use of effective medications? Will patients utilize available options?”
“These and other fundamental issues must be figured out before we anoint any treatment option as a meaningful step forward, let alone a transformative development,” according to Dr. Kahan.
Similarly, Irl B. Hirsch, MD, stressed that, should this medication be approved for weight loss, cost would be a major factor in its uptake.
“I’m old enough to recall when we started using lovastatin in the late 1980s,” Dr. Hirsch, professor of medicine, University of Washington Medicine Diabetes Institute, Seattle, said in an interview.
“We used it without the type of evidence of statin use we have today. A pill, but in those days the statins were expensive. But over time, the evidence for statins grew and over the next 15 years it was quite clear that for both primary prevention (for those with diabetes) and secondary intervention these drugs needed to be used by millions of people. These recommendations became easier once the drugs became generic.
“Will the same thing happen for GLP-1 agonists? The problem is we need both ‘hard-outcome data’ [such as 3-point major adverse cardiovascular events] and more reasonable cost before we see this expanding to an entire population.
“In the future perhaps we could have a biosimilar GLP-1 agonist that would be more affordable than what we pay now, but even before that we need agreement from our reimbursement thought leaders that our society should reimburse these agents.
“My thinking now is the cost-benefit could be favorable, but this is all dependent on what happens to the cost of the drugs over time,” he said.
Additive effect of intensive behavior therapy plus medication
Dr. Wadden explained that intensive behavioral therapy “provides 14 or more counseling sessions in 6 months to modify diet and physical activity, through the patients’ use of behavioral strategies (such as keeping daily food and activity diaries).”
Such programs typically produce mean weight loss of 5%-8% of initial weight; less frequent (e.g., monthly) programs typically produce weight loss of only 1%-3%.
Prior studies suggest that intensive behavioral therapy and medication have additive effects. To investigate this, Dr. Wadden and colleagues randomized 611 adults (81% women) who were a mean age of 46 years and had a mean body mass index of 38 kg/m2.
All participants received 30 intensive behavior therapy sessions provided by a registered dietitian (or other qualified provider), which typically lasted 20-30 minutes and were given weekly for 12 weeks, every other week for the next 12 weeks, and then monthly.
The dietitian gave participants behavioral strategies to help them adhere to diet and physical activity goals.
During the first 8 weeks, participants were provided with a 1,000-1,200 kcal/day meal replacement diet that included liquid shakes, meal bars, and prepared entrees designed to facilitate a large initial weight loss.
They then transitioned to a diet of conventional foods (of their choosing), with a goal of 1,200-1,800 kcal/day based on body weight.
The physical activity goal was 100 minutes/week of walking or other aerobic activity in the first month, building up to 200 minutes/week by month 6.
‘More effective than current FDA-approved weight-loss medications’
At week 68, mean body weight decreased from baseline by 16.0% in the semaglutide group versus 5.7% in the placebo group (P < .0001).
In this trial, where all participants received extensive intensive behavior therapy, more participants had weight loss ≥5%, ≥10%, ≥15%, and ≥20% of their initial weight with semaglutide versus placebo (87% vs. 48%; 75% vs. 27%; 56% vs. 13%; 36% vs. 4%, respectively; all P < .0001).
From baseline to week 68, the proportion of participants with prediabetes decreased from 48% to 7% in the semaglutide group and from 53% to 26% in the placebo group.
Patients who received semaglutide had greater improvements in lipids, too.
Although the weight loss was 10.3% (10.6 kg) greater with semaglutide, Dr. Wadden noted, “additional studies have shown this net benefit to be as great as 11%-12%, which would make semaglutide 2.4 mg more effective than current [FDA-approved] weight-loss medications.”
“Naltrexone-bupropion (Contrave) with lifestyle counseling, for example,” he continued, “produces a loss that is 5 kg greater than lifestyle counseling plus placebo, liraglutide 3.0 mg (Saxenda) a loss 5.3 kg greater than placebo, and phentermine-topiramate (Qsymia) a loss that is 8.8 kg greater than placebo.”
Semaglutide was well tolerated. Gastrointestinal adverse events, the most common type, occurred in 83% of patients in the semaglutide group and 63% of patients in the placebo group.
Nausea, as well as constipation and diarrhea, are common in medications that increase GLP-1 levels, Dr. Wadden noted. Side effects can be managed by slowly increasing the medication dose over 4 months.
Dr. Wadden expects that, if approved, semaglutide 2.4 mg subcutaneous once-weekly will be recommended as an adjunct to a reduced calorie diet and increased physical activity. Additional studies suggest that monthly counseling should be sufficient to obtain similar weight losses as those seen in the current trial, which had more intensive counseling.
As well as being approved as a weekly subcutaneous injectable treatment for type 2 diabetes, semaglutide is also approved as an once-daily oral agent for the same indication (Rybelsus, Novo Nordisk) in doses of 7 mg and 14 mg to improve glycemic control along with diet and exercise. It is the first GLP-1 agonist available in tablet form.
Dr. Wadden serves on scientific advisory boards for Novo Nordisk and WW (formerly Weight Watchers), and has received grant support, on behalf of the University of Pennsylvania, from Novo Nordisk. Dr. Aronne is an investigator in a long-term trial of semaglutide and has served on scientific advisory boards for Novo Nordisk in the past. He also has other industry relationships that are not related to semaglutide.
A version of this article originally appeared on Medscape.com.
Report may inform first dietary guidelines for Americans from birth to 24 months
The U.S. Department of Agriculture and the Department of Health & Human Services aim to release new dietary guidelines by the end of 2020.
An advisory committee submitted to the agencies a scientific report that examines relationships between diet and health at various life stages. Four chapters focus on dietary considerations for infants and toddlers, and two chapters focus on diet during pregnancy and lactation.
The report may inform the development of the new guidelines. The advisory committee’s recommendations include introducing infants to foods that are rich in zinc and iron at about age 6 months and having women who are lactating eat sources of omega-3 and omega-6 fatty acids, such as fish, to improve the fatty acid status of infants.
Ahead of the release of the 2020-2025 Dietary Guidelines for Americans, Joan Younger Meek, MD, discussed parts of the scientific report at the annual meeting of the American Academy of Pediatrics, held virtually this year.
While the 2015-2020 guidelines use ChooseMyPlate to help people implement the recommendations, it is not known how the new guidelines will be presented to the public, she said. “Many of you will remember the pyramids earlier and different food groups before that.”
Promote healthy dietary patterns
The advisory committee’s report notes that diet in the first years of life contributes to long-term health and shapes taste preferences, said Dr. Meek, professor of clinical sciences at Florida State University, Orlando. Human milk or infant formula are primary sources of nutrition until approximately 6 months, when families may introduce complementary foods and beverages. Between 6 months and 24 months, children transition to the typical family diet.
Dr. Meek highlighted some of the advisory committee’s findings and recommendations.
- Infants who are ever breastfed have a reduced risk of overweight or obesity, type 1 diabetes, and asthma. Likewise, longer duration of breastfeeding is associated with lower risk of type 1 diabetes and asthma, and exclusive breastfeeding is associated with lower risk of type 1 diabetes.
- Complementary foods and beverages should not be introduced before age 4 months. Limited evidence indicates that their introduction before 4 months may be associated with increased odds of overweight or obesity. Introducing complementary foods or beverages at 4 or 5 months, compared with 6 months, is not associated with long-term advantages or disadvantages.
- Introducing peanut and egg after age 4 months may reduce the risk of food allergies.
- From age 12 months to 24 months, children should consume a variety of nutrient-rich protein sources from animals – including meat, poultry, seafood, eggs, and dairy – plus nuts, seeds, fruits, vegetables, and grains.
- The report prioritizes oils over solid fats, and whole grains over refined grains. It also discourages added sugars, particularly from sugar-sweetened beverages. Other sources of added sugars include sweets, baked goods, and sweetened dairy products.
The report acknowledges that dietary guidelines should accommodate cultural preferences and cost considerations.
Recommendations during pregnancy
Healthy dietary patterns before or during pregnancy may modestly reduce the odds of gestational diabetes, hypertensive disorders of pregnancy, and preterm birth, according to the report.
The report recommends that during pregnancy women consume 8-12 ounces per week of seafood with high levels of omega-3 fatty acids and low levels of methylmercury, consistent with existing recommendations.
Egg and milk consumption during pregnancy does not influence the risk of food allergy, asthma, or atopic disease in the child, according to the report.
The advisory committee recommended universal folic acid supplementation during pregnancy.
Addressing a gap
The Agricultural Act of 2014 required that infants and toddlers and women who are pregnant or lactating be included in the 2020-2025 guidelines. Covering these populations in the scientific report was a substantial undertaking, said Kathryn Dewey, PhD, of the Institute for Global Nutrition at the University of California, Davis. Dr. Dewey chaired the subcommittee on birth to 24 months for the 2020 Dietary Guidelines Advisory Committee.
“Given that this age group had not been covered before, we could not rely on previous dietary guidelines’ reports,” Dr. Dewey said in an interview.
Outlining food patterns for infants and toddlers proved challenging. The committee explored models that considered various scenarios including children who consumed human milk, children who consumed formula, and those with vegetarian diets. Future research should clarify dietary reference intakes for these age groups, Dr. Dewey said.
Dr. Dewey sees the committee’s report on dietary guidance for birth to 24 months as a starting point and not necessarily an exhaustive look at the subject.
For one, the committee focused more on what to feed infants and toddlers rather than on how to feed them. Information about how to feed children is considered more in depth in a 2020 report from the National Academies of Sciences, Engineering, and Medicine. That report summarizes existing guidance from various organizations on feeding infants and children from birth to 24 months. Dr. Dewey chaired the committee that created the National Academies report.
Sharing the new USDA and HHS guidelines after they are released could be the next important step. “The public does not necessarily know about the guidelines or they do not necessarily seek them out unless there is a very well-constructed strategy for dissemination and implementation,” Dr. Dewey said.
To that end, health care providers can play a role, Dr. Meek said. “Be aware of changes in guidance, adopt those new recommendations, and then advocate those with our patients as well as with the public at large.”
Dr. Meek and Dr. Dewey had no relevant financial disclosures.
The U.S. Department of Agriculture and the Department of Health & Human Services aim to release new dietary guidelines by the end of 2020.
An advisory committee submitted to the agencies a scientific report that examines relationships between diet and health at various life stages. Four chapters focus on dietary considerations for infants and toddlers, and two chapters focus on diet during pregnancy and lactation.
The report may inform the development of the new guidelines. The advisory committee’s recommendations include introducing infants to foods that are rich in zinc and iron at about age 6 months and having women who are lactating eat sources of omega-3 and omega-6 fatty acids, such as fish, to improve the fatty acid status of infants.
Ahead of the release of the 2020-2025 Dietary Guidelines for Americans, Joan Younger Meek, MD, discussed parts of the scientific report at the annual meeting of the American Academy of Pediatrics, held virtually this year.
While the 2015-2020 guidelines use ChooseMyPlate to help people implement the recommendations, it is not known how the new guidelines will be presented to the public, she said. “Many of you will remember the pyramids earlier and different food groups before that.”
Promote healthy dietary patterns
The advisory committee’s report notes that diet in the first years of life contributes to long-term health and shapes taste preferences, said Dr. Meek, professor of clinical sciences at Florida State University, Orlando. Human milk or infant formula are primary sources of nutrition until approximately 6 months, when families may introduce complementary foods and beverages. Between 6 months and 24 months, children transition to the typical family diet.
Dr. Meek highlighted some of the advisory committee’s findings and recommendations.
- Infants who are ever breastfed have a reduced risk of overweight or obesity, type 1 diabetes, and asthma. Likewise, longer duration of breastfeeding is associated with lower risk of type 1 diabetes and asthma, and exclusive breastfeeding is associated with lower risk of type 1 diabetes.
- Complementary foods and beverages should not be introduced before age 4 months. Limited evidence indicates that their introduction before 4 months may be associated with increased odds of overweight or obesity. Introducing complementary foods or beverages at 4 or 5 months, compared with 6 months, is not associated with long-term advantages or disadvantages.
- Introducing peanut and egg after age 4 months may reduce the risk of food allergies.
- From age 12 months to 24 months, children should consume a variety of nutrient-rich protein sources from animals – including meat, poultry, seafood, eggs, and dairy – plus nuts, seeds, fruits, vegetables, and grains.
- The report prioritizes oils over solid fats, and whole grains over refined grains. It also discourages added sugars, particularly from sugar-sweetened beverages. Other sources of added sugars include sweets, baked goods, and sweetened dairy products.
The report acknowledges that dietary guidelines should accommodate cultural preferences and cost considerations.
Recommendations during pregnancy
Healthy dietary patterns before or during pregnancy may modestly reduce the odds of gestational diabetes, hypertensive disorders of pregnancy, and preterm birth, according to the report.
The report recommends that during pregnancy women consume 8-12 ounces per week of seafood with high levels of omega-3 fatty acids and low levels of methylmercury, consistent with existing recommendations.
Egg and milk consumption during pregnancy does not influence the risk of food allergy, asthma, or atopic disease in the child, according to the report.
The advisory committee recommended universal folic acid supplementation during pregnancy.
Addressing a gap
The Agricultural Act of 2014 required that infants and toddlers and women who are pregnant or lactating be included in the 2020-2025 guidelines. Covering these populations in the scientific report was a substantial undertaking, said Kathryn Dewey, PhD, of the Institute for Global Nutrition at the University of California, Davis. Dr. Dewey chaired the subcommittee on birth to 24 months for the 2020 Dietary Guidelines Advisory Committee.
“Given that this age group had not been covered before, we could not rely on previous dietary guidelines’ reports,” Dr. Dewey said in an interview.
Outlining food patterns for infants and toddlers proved challenging. The committee explored models that considered various scenarios including children who consumed human milk, children who consumed formula, and those with vegetarian diets. Future research should clarify dietary reference intakes for these age groups, Dr. Dewey said.
Dr. Dewey sees the committee’s report on dietary guidance for birth to 24 months as a starting point and not necessarily an exhaustive look at the subject.
For one, the committee focused more on what to feed infants and toddlers rather than on how to feed them. Information about how to feed children is considered more in depth in a 2020 report from the National Academies of Sciences, Engineering, and Medicine. That report summarizes existing guidance from various organizations on feeding infants and children from birth to 24 months. Dr. Dewey chaired the committee that created the National Academies report.
Sharing the new USDA and HHS guidelines after they are released could be the next important step. “The public does not necessarily know about the guidelines or they do not necessarily seek them out unless there is a very well-constructed strategy for dissemination and implementation,” Dr. Dewey said.
To that end, health care providers can play a role, Dr. Meek said. “Be aware of changes in guidance, adopt those new recommendations, and then advocate those with our patients as well as with the public at large.”
Dr. Meek and Dr. Dewey had no relevant financial disclosures.
The U.S. Department of Agriculture and the Department of Health & Human Services aim to release new dietary guidelines by the end of 2020.
An advisory committee submitted to the agencies a scientific report that examines relationships between diet and health at various life stages. Four chapters focus on dietary considerations for infants and toddlers, and two chapters focus on diet during pregnancy and lactation.
The report may inform the development of the new guidelines. The advisory committee’s recommendations include introducing infants to foods that are rich in zinc and iron at about age 6 months and having women who are lactating eat sources of omega-3 and omega-6 fatty acids, such as fish, to improve the fatty acid status of infants.
Ahead of the release of the 2020-2025 Dietary Guidelines for Americans, Joan Younger Meek, MD, discussed parts of the scientific report at the annual meeting of the American Academy of Pediatrics, held virtually this year.
While the 2015-2020 guidelines use ChooseMyPlate to help people implement the recommendations, it is not known how the new guidelines will be presented to the public, she said. “Many of you will remember the pyramids earlier and different food groups before that.”
Promote healthy dietary patterns
The advisory committee’s report notes that diet in the first years of life contributes to long-term health and shapes taste preferences, said Dr. Meek, professor of clinical sciences at Florida State University, Orlando. Human milk or infant formula are primary sources of nutrition until approximately 6 months, when families may introduce complementary foods and beverages. Between 6 months and 24 months, children transition to the typical family diet.
Dr. Meek highlighted some of the advisory committee’s findings and recommendations.
- Infants who are ever breastfed have a reduced risk of overweight or obesity, type 1 diabetes, and asthma. Likewise, longer duration of breastfeeding is associated with lower risk of type 1 diabetes and asthma, and exclusive breastfeeding is associated with lower risk of type 1 diabetes.
- Complementary foods and beverages should not be introduced before age 4 months. Limited evidence indicates that their introduction before 4 months may be associated with increased odds of overweight or obesity. Introducing complementary foods or beverages at 4 or 5 months, compared with 6 months, is not associated with long-term advantages or disadvantages.
- Introducing peanut and egg after age 4 months may reduce the risk of food allergies.
- From age 12 months to 24 months, children should consume a variety of nutrient-rich protein sources from animals – including meat, poultry, seafood, eggs, and dairy – plus nuts, seeds, fruits, vegetables, and grains.
- The report prioritizes oils over solid fats, and whole grains over refined grains. It also discourages added sugars, particularly from sugar-sweetened beverages. Other sources of added sugars include sweets, baked goods, and sweetened dairy products.
The report acknowledges that dietary guidelines should accommodate cultural preferences and cost considerations.
Recommendations during pregnancy
Healthy dietary patterns before or during pregnancy may modestly reduce the odds of gestational diabetes, hypertensive disorders of pregnancy, and preterm birth, according to the report.
The report recommends that during pregnancy women consume 8-12 ounces per week of seafood with high levels of omega-3 fatty acids and low levels of methylmercury, consistent with existing recommendations.
Egg and milk consumption during pregnancy does not influence the risk of food allergy, asthma, or atopic disease in the child, according to the report.
The advisory committee recommended universal folic acid supplementation during pregnancy.
Addressing a gap
The Agricultural Act of 2014 required that infants and toddlers and women who are pregnant or lactating be included in the 2020-2025 guidelines. Covering these populations in the scientific report was a substantial undertaking, said Kathryn Dewey, PhD, of the Institute for Global Nutrition at the University of California, Davis. Dr. Dewey chaired the subcommittee on birth to 24 months for the 2020 Dietary Guidelines Advisory Committee.
“Given that this age group had not been covered before, we could not rely on previous dietary guidelines’ reports,” Dr. Dewey said in an interview.
Outlining food patterns for infants and toddlers proved challenging. The committee explored models that considered various scenarios including children who consumed human milk, children who consumed formula, and those with vegetarian diets. Future research should clarify dietary reference intakes for these age groups, Dr. Dewey said.
Dr. Dewey sees the committee’s report on dietary guidance for birth to 24 months as a starting point and not necessarily an exhaustive look at the subject.
For one, the committee focused more on what to feed infants and toddlers rather than on how to feed them. Information about how to feed children is considered more in depth in a 2020 report from the National Academies of Sciences, Engineering, and Medicine. That report summarizes existing guidance from various organizations on feeding infants and children from birth to 24 months. Dr. Dewey chaired the committee that created the National Academies report.
Sharing the new USDA and HHS guidelines after they are released could be the next important step. “The public does not necessarily know about the guidelines or they do not necessarily seek them out unless there is a very well-constructed strategy for dissemination and implementation,” Dr. Dewey said.
To that end, health care providers can play a role, Dr. Meek said. “Be aware of changes in guidance, adopt those new recommendations, and then advocate those with our patients as well as with the public at large.”
Dr. Meek and Dr. Dewey had no relevant financial disclosures.
FROM AAP 2020
‘Disordered eating’ drops after teens undergo bariatric surgery
Kristina M. Decker, PhD, a postdoctoral fellow at Cincinnati Children’s Hospital Medical Center, presented these findings during the virtual ObesityWeek 2020.
Dr. Decker and associates examined rates of disordered eating in more than 200 adolescents (aged 13-18 years) who were severely obese, of whom 141 underwent bariatric surgery and the remainder did not.
At baseline (presurgery), the teens in both groups had rates of disordered eating ranging from 11% to 50%, with higher rates in those who went on to have bariatric surgery.
Six years later, rates of disordered eating were much lower in those who had bariatric surgery.
The data nevertheless “underscore that young adults with persistent severe obesity are at high risk for poor health and well-being,” Dr. Decker said in an interview.
“This means disordered eating behaviors should be closely monitored” in all such patients, both those who undergo surgery and those who don’t, she stressed.
Robust findings because of long follow-up and controls
The findings are not unexpected, based on adult bariatric literature, but are “novel because of the age of the patients,” senior author Margaret H. Zeller, PhD, Cincinnati Children’s Hospital Medical Center and professor at the University of Cincinnati, added.
In a comment comment, psychologist Kajsa Järvholm, PhD, of the Childhood Obesity Unit at Skåne University Hospital, Malmö̈, Sweden, who has published related work, said that this is “a needed study.”
Notably, it had “long-term follow-up and a control group,” and it “confirms that adolescents are in better control of their eating after surgery.”
However, an important additional takeaway for clinicians is that “disordered eating is associated with other mental health problems and self-worth. Clinicians treating obesity must address problems related to eating disorders to improve outcomes and well-being,” she stressed.
How does bariatric surgery impact overeating, binge eating, in teens?
“For teens with severe obesity, metabolic and bariatric surgery is the most effective treatment for improved cardiometabolic functioning, weight loss, and improved quality of life,” Dr. Decker stressed.
However, pre- and postsurgical disordered eating behaviors have been associated with a lower percentage change in body mass index (BMI), although this has not been well studied.
To investigate how disordered eating is affected by bariatric surgery in adolescents with severe obesity, researchers used data from Teen-LABS, which enrolled 242 participants aged 19 years and under who mainly underwent Roux-en-Y gastric bypass (67%) or sleeve gastrectomy (28%) from 2007 to 2012 at five adolescent bariatric surgery centers.
The current analysis examined data from 141 participants in Teen-LABS who underwent bariatric surgery at a mean age of 16.8 years. Mean BMI was 51.5, most were girls (80%), and they had diverse race/ethnicity (66% were White).
Researchers also identified a control group of 83 adolescents of a similar age and gender who had diverse race/ethnicity (54% White) and a mean BMI of 46.9.
At year 6, data were available for 123 young adults in the surgery group (who by then had a mean BMI of 39.7) and 63 young adults in the nonsurgery group (who had a mean BMI of 52.6).
At baseline and year 6, participants replied to questionnaires that identified three eating disorders: continuous eating (eating in an unplanned and repetitious way between meals and snacks), objective overeating (eating a “large” amount of food without loss of control), and objective binge eating (eating a “large” amount of food with loss of control).
At baseline, rates of continuous eating, overeating, and binge eating were higher in the surgical group (50%, 40%, and 30%, respectively) than the nonsurgical group (40%, 22%, and 11%, respectively).
Six years later, when participants were aged 19-24 years, rates of continuous eating, overeating, and binge eating had declined in the surgical group (to 17%, 5%, and 1%, respectively). In the nonsurgical group, only continuous eating and overeating declined (to 24% and 7%, respectively), and binge eating increased slightly (to 13%).
Disordered eating associated with low self-worth, anxiety, and depression
In young adulthood in both groups, disordered eating was associated with lower self-worth. In the surgical group, it was also associated with lower weight-related quality of life, and in the nonsurgical group, it was also associated with anxiety and/or depression.
“The current findings cannot tell us whether disordered eating is a direct result or caused by anxiety, depression, low self-worth, or poor quality of life,” Dr. Decker said.
“These findings do give us insight about what other areas of clinical concern might present together [in] young adults (e.g., disordered eating, low self-esteem).”
Bariatric surgery affects the amount of food people can eat at one time, she noted in reply to a question from the audience. If people eat too much at a time they can experience vomiting, dumping syndrome (where certain food is “dumped” into the small intestine without being digested, causing nausea and vomiting), and plugging (a sense of food becoming stuck).
The home environment and transition to adulthood might impact disordered eating in young adults, she said in reply to another question, but these issues were not examined in this study.
A version of this article originally appeared on Medscape.com.
Kristina M. Decker, PhD, a postdoctoral fellow at Cincinnati Children’s Hospital Medical Center, presented these findings during the virtual ObesityWeek 2020.
Dr. Decker and associates examined rates of disordered eating in more than 200 adolescents (aged 13-18 years) who were severely obese, of whom 141 underwent bariatric surgery and the remainder did not.
At baseline (presurgery), the teens in both groups had rates of disordered eating ranging from 11% to 50%, with higher rates in those who went on to have bariatric surgery.
Six years later, rates of disordered eating were much lower in those who had bariatric surgery.
The data nevertheless “underscore that young adults with persistent severe obesity are at high risk for poor health and well-being,” Dr. Decker said in an interview.
“This means disordered eating behaviors should be closely monitored” in all such patients, both those who undergo surgery and those who don’t, she stressed.
Robust findings because of long follow-up and controls
The findings are not unexpected, based on adult bariatric literature, but are “novel because of the age of the patients,” senior author Margaret H. Zeller, PhD, Cincinnati Children’s Hospital Medical Center and professor at the University of Cincinnati, added.
In a comment comment, psychologist Kajsa Järvholm, PhD, of the Childhood Obesity Unit at Skåne University Hospital, Malmö̈, Sweden, who has published related work, said that this is “a needed study.”
Notably, it had “long-term follow-up and a control group,” and it “confirms that adolescents are in better control of their eating after surgery.”
However, an important additional takeaway for clinicians is that “disordered eating is associated with other mental health problems and self-worth. Clinicians treating obesity must address problems related to eating disorders to improve outcomes and well-being,” she stressed.
How does bariatric surgery impact overeating, binge eating, in teens?
“For teens with severe obesity, metabolic and bariatric surgery is the most effective treatment for improved cardiometabolic functioning, weight loss, and improved quality of life,” Dr. Decker stressed.
However, pre- and postsurgical disordered eating behaviors have been associated with a lower percentage change in body mass index (BMI), although this has not been well studied.
To investigate how disordered eating is affected by bariatric surgery in adolescents with severe obesity, researchers used data from Teen-LABS, which enrolled 242 participants aged 19 years and under who mainly underwent Roux-en-Y gastric bypass (67%) or sleeve gastrectomy (28%) from 2007 to 2012 at five adolescent bariatric surgery centers.
The current analysis examined data from 141 participants in Teen-LABS who underwent bariatric surgery at a mean age of 16.8 years. Mean BMI was 51.5, most were girls (80%), and they had diverse race/ethnicity (66% were White).
Researchers also identified a control group of 83 adolescents of a similar age and gender who had diverse race/ethnicity (54% White) and a mean BMI of 46.9.
At year 6, data were available for 123 young adults in the surgery group (who by then had a mean BMI of 39.7) and 63 young adults in the nonsurgery group (who had a mean BMI of 52.6).
At baseline and year 6, participants replied to questionnaires that identified three eating disorders: continuous eating (eating in an unplanned and repetitious way between meals and snacks), objective overeating (eating a “large” amount of food without loss of control), and objective binge eating (eating a “large” amount of food with loss of control).
At baseline, rates of continuous eating, overeating, and binge eating were higher in the surgical group (50%, 40%, and 30%, respectively) than the nonsurgical group (40%, 22%, and 11%, respectively).
Six years later, when participants were aged 19-24 years, rates of continuous eating, overeating, and binge eating had declined in the surgical group (to 17%, 5%, and 1%, respectively). In the nonsurgical group, only continuous eating and overeating declined (to 24% and 7%, respectively), and binge eating increased slightly (to 13%).
Disordered eating associated with low self-worth, anxiety, and depression
In young adulthood in both groups, disordered eating was associated with lower self-worth. In the surgical group, it was also associated with lower weight-related quality of life, and in the nonsurgical group, it was also associated with anxiety and/or depression.
“The current findings cannot tell us whether disordered eating is a direct result or caused by anxiety, depression, low self-worth, or poor quality of life,” Dr. Decker said.
“These findings do give us insight about what other areas of clinical concern might present together [in] young adults (e.g., disordered eating, low self-esteem).”
Bariatric surgery affects the amount of food people can eat at one time, she noted in reply to a question from the audience. If people eat too much at a time they can experience vomiting, dumping syndrome (where certain food is “dumped” into the small intestine without being digested, causing nausea and vomiting), and plugging (a sense of food becoming stuck).
The home environment and transition to adulthood might impact disordered eating in young adults, she said in reply to another question, but these issues were not examined in this study.
A version of this article originally appeared on Medscape.com.
Kristina M. Decker, PhD, a postdoctoral fellow at Cincinnati Children’s Hospital Medical Center, presented these findings during the virtual ObesityWeek 2020.
Dr. Decker and associates examined rates of disordered eating in more than 200 adolescents (aged 13-18 years) who were severely obese, of whom 141 underwent bariatric surgery and the remainder did not.
At baseline (presurgery), the teens in both groups had rates of disordered eating ranging from 11% to 50%, with higher rates in those who went on to have bariatric surgery.
Six years later, rates of disordered eating were much lower in those who had bariatric surgery.
The data nevertheless “underscore that young adults with persistent severe obesity are at high risk for poor health and well-being,” Dr. Decker said in an interview.
“This means disordered eating behaviors should be closely monitored” in all such patients, both those who undergo surgery and those who don’t, she stressed.
Robust findings because of long follow-up and controls
The findings are not unexpected, based on adult bariatric literature, but are “novel because of the age of the patients,” senior author Margaret H. Zeller, PhD, Cincinnati Children’s Hospital Medical Center and professor at the University of Cincinnati, added.
In a comment comment, psychologist Kajsa Järvholm, PhD, of the Childhood Obesity Unit at Skåne University Hospital, Malmö̈, Sweden, who has published related work, said that this is “a needed study.”
Notably, it had “long-term follow-up and a control group,” and it “confirms that adolescents are in better control of their eating after surgery.”
However, an important additional takeaway for clinicians is that “disordered eating is associated with other mental health problems and self-worth. Clinicians treating obesity must address problems related to eating disorders to improve outcomes and well-being,” she stressed.
How does bariatric surgery impact overeating, binge eating, in teens?
“For teens with severe obesity, metabolic and bariatric surgery is the most effective treatment for improved cardiometabolic functioning, weight loss, and improved quality of life,” Dr. Decker stressed.
However, pre- and postsurgical disordered eating behaviors have been associated with a lower percentage change in body mass index (BMI), although this has not been well studied.
To investigate how disordered eating is affected by bariatric surgery in adolescents with severe obesity, researchers used data from Teen-LABS, which enrolled 242 participants aged 19 years and under who mainly underwent Roux-en-Y gastric bypass (67%) or sleeve gastrectomy (28%) from 2007 to 2012 at five adolescent bariatric surgery centers.
The current analysis examined data from 141 participants in Teen-LABS who underwent bariatric surgery at a mean age of 16.8 years. Mean BMI was 51.5, most were girls (80%), and they had diverse race/ethnicity (66% were White).
Researchers also identified a control group of 83 adolescents of a similar age and gender who had diverse race/ethnicity (54% White) and a mean BMI of 46.9.
At year 6, data were available for 123 young adults in the surgery group (who by then had a mean BMI of 39.7) and 63 young adults in the nonsurgery group (who had a mean BMI of 52.6).
At baseline and year 6, participants replied to questionnaires that identified three eating disorders: continuous eating (eating in an unplanned and repetitious way between meals and snacks), objective overeating (eating a “large” amount of food without loss of control), and objective binge eating (eating a “large” amount of food with loss of control).
At baseline, rates of continuous eating, overeating, and binge eating were higher in the surgical group (50%, 40%, and 30%, respectively) than the nonsurgical group (40%, 22%, and 11%, respectively).
Six years later, when participants were aged 19-24 years, rates of continuous eating, overeating, and binge eating had declined in the surgical group (to 17%, 5%, and 1%, respectively). In the nonsurgical group, only continuous eating and overeating declined (to 24% and 7%, respectively), and binge eating increased slightly (to 13%).
Disordered eating associated with low self-worth, anxiety, and depression
In young adulthood in both groups, disordered eating was associated with lower self-worth. In the surgical group, it was also associated with lower weight-related quality of life, and in the nonsurgical group, it was also associated with anxiety and/or depression.
“The current findings cannot tell us whether disordered eating is a direct result or caused by anxiety, depression, low self-worth, or poor quality of life,” Dr. Decker said.
“These findings do give us insight about what other areas of clinical concern might present together [in] young adults (e.g., disordered eating, low self-esteem).”
Bariatric surgery affects the amount of food people can eat at one time, she noted in reply to a question from the audience. If people eat too much at a time they can experience vomiting, dumping syndrome (where certain food is “dumped” into the small intestine without being digested, causing nausea and vomiting), and plugging (a sense of food becoming stuck).
The home environment and transition to adulthood might impact disordered eating in young adults, she said in reply to another question, but these issues were not examined in this study.
A version of this article originally appeared on Medscape.com.
Cannabis may improve liver function in patients with obesity
Cannabis use is associated with a decrease in the prevalence of steatohepatitis and a slowing of its progression in patients with obesity, results from a retrospective cohort study show.
This suggests “that the anti-inflammatory effects of cannabis may be leading to reduced prevalence of steatohepatitis in cannabis users,” said Ikechukwu Achebe, MD, from the John H. Stroger, Jr. Hospital of Cook County in Chicago.
Liver injuries such as nonalcoholic steatohepatitis are characterized by hepatocellular injury and inflammation, which combine to contribute to an increase in the risk for liver failure, cirrhosis, and hepatocellular carcinoma.
“This is where cannabis comes in,” said Dr. Achebe, who presented the study results at the virtual annual meeting of the American College of Gastroenterology. “It is the most commonly used psychoactive substance worldwide and has been shown to reduce hepatic myofibroblast and stellate cell injury. Studies using mouse models have demonstrated reduced liver fibrosis and cirrhosis as a consequence of cannabis exposure.”
Given this possible connection, Dr. Achebe and colleagues set out to determine whether cannabis use affects the prevalence and progression of nonalcoholic fatty liver disease (NAFLD) in obese patients.
To do so, they analyzed the discharge records of 879,952 obese adults in the 2016 Healthcare Cost and Utilization Project National Inpatient Sample. The primary outcome was the prevalence of the four presentations of NAFLD: steatosis, steatohepatitis, cirrhosis, and hepatocellular carcinoma.
The researchers compared disease stages in cannabis users and nonusers. In the study cohort of 14,236 patients, 1.6% used cannabis. Steatohepatitis was less common among cannabis users than among nonusers (0.4% vs. 0.7%; P < .001), as was cirrhosis (1.1% vs. 1.5%; P < .001).
After propensity matching, the association between cannabis use and lower rates of steatohepatitis remained significant (0.4% vs. 0.5%; P = .035), but the association between cannabis use and the prevalence of nonalcoholic fatty liver, cirrhosis, and hepatocellular carcinoma did not.
These results might be partly explained by the protective effect of cannabis on hepatocytes regulated by the endocannabinoid system, the researchers concluded.
More studies are needed to explore this relation, said Dr. Achebe.
The challenge of self-reported use
The study is “incredibly interesting,” said Nancy S. Reau, MD, from Rush Medical College, Chicago. However, the association between cannabis and nonalcoholic fatty liver needs to be further investigated before clinicians can counsel their patients to use the agent to prevent progression.
It is difficult in a study such as this to tease out other lifestyle factors that might be linked to cannabis use, she explained. For example, “is it possible that the cannabis users exercise more, drink more coffee, or eat differently?”
And “self-reported use is challenging,” Dr. Reau said in an interview. “This cannot differentiate someone who occasionally uses from someone who is a heavy daily user. There must be some minimum level of exposure needed for it to have protective effects, if they exist.”
This study was honored at the meeting as an ACG Newsworthy Abstract and an ACG Outstanding Poster Presenter.
Dr. Achebe disclosed no relevant financial relationships. Dr. Reau reported receiving research support from Genfit and having a consultant relationship with Intercept Pharmaceuticals.
A version of this article originally appeared on Medscape.com.
Cannabis use is associated with a decrease in the prevalence of steatohepatitis and a slowing of its progression in patients with obesity, results from a retrospective cohort study show.
This suggests “that the anti-inflammatory effects of cannabis may be leading to reduced prevalence of steatohepatitis in cannabis users,” said Ikechukwu Achebe, MD, from the John H. Stroger, Jr. Hospital of Cook County in Chicago.
Liver injuries such as nonalcoholic steatohepatitis are characterized by hepatocellular injury and inflammation, which combine to contribute to an increase in the risk for liver failure, cirrhosis, and hepatocellular carcinoma.
“This is where cannabis comes in,” said Dr. Achebe, who presented the study results at the virtual annual meeting of the American College of Gastroenterology. “It is the most commonly used psychoactive substance worldwide and has been shown to reduce hepatic myofibroblast and stellate cell injury. Studies using mouse models have demonstrated reduced liver fibrosis and cirrhosis as a consequence of cannabis exposure.”
Given this possible connection, Dr. Achebe and colleagues set out to determine whether cannabis use affects the prevalence and progression of nonalcoholic fatty liver disease (NAFLD) in obese patients.
To do so, they analyzed the discharge records of 879,952 obese adults in the 2016 Healthcare Cost and Utilization Project National Inpatient Sample. The primary outcome was the prevalence of the four presentations of NAFLD: steatosis, steatohepatitis, cirrhosis, and hepatocellular carcinoma.
The researchers compared disease stages in cannabis users and nonusers. In the study cohort of 14,236 patients, 1.6% used cannabis. Steatohepatitis was less common among cannabis users than among nonusers (0.4% vs. 0.7%; P < .001), as was cirrhosis (1.1% vs. 1.5%; P < .001).
After propensity matching, the association between cannabis use and lower rates of steatohepatitis remained significant (0.4% vs. 0.5%; P = .035), but the association between cannabis use and the prevalence of nonalcoholic fatty liver, cirrhosis, and hepatocellular carcinoma did not.
These results might be partly explained by the protective effect of cannabis on hepatocytes regulated by the endocannabinoid system, the researchers concluded.
More studies are needed to explore this relation, said Dr. Achebe.
The challenge of self-reported use
The study is “incredibly interesting,” said Nancy S. Reau, MD, from Rush Medical College, Chicago. However, the association between cannabis and nonalcoholic fatty liver needs to be further investigated before clinicians can counsel their patients to use the agent to prevent progression.
It is difficult in a study such as this to tease out other lifestyle factors that might be linked to cannabis use, she explained. For example, “is it possible that the cannabis users exercise more, drink more coffee, or eat differently?”
And “self-reported use is challenging,” Dr. Reau said in an interview. “This cannot differentiate someone who occasionally uses from someone who is a heavy daily user. There must be some minimum level of exposure needed for it to have protective effects, if they exist.”
This study was honored at the meeting as an ACG Newsworthy Abstract and an ACG Outstanding Poster Presenter.
Dr. Achebe disclosed no relevant financial relationships. Dr. Reau reported receiving research support from Genfit and having a consultant relationship with Intercept Pharmaceuticals.
A version of this article originally appeared on Medscape.com.
Cannabis use is associated with a decrease in the prevalence of steatohepatitis and a slowing of its progression in patients with obesity, results from a retrospective cohort study show.
This suggests “that the anti-inflammatory effects of cannabis may be leading to reduced prevalence of steatohepatitis in cannabis users,” said Ikechukwu Achebe, MD, from the John H. Stroger, Jr. Hospital of Cook County in Chicago.
Liver injuries such as nonalcoholic steatohepatitis are characterized by hepatocellular injury and inflammation, which combine to contribute to an increase in the risk for liver failure, cirrhosis, and hepatocellular carcinoma.
“This is where cannabis comes in,” said Dr. Achebe, who presented the study results at the virtual annual meeting of the American College of Gastroenterology. “It is the most commonly used psychoactive substance worldwide and has been shown to reduce hepatic myofibroblast and stellate cell injury. Studies using mouse models have demonstrated reduced liver fibrosis and cirrhosis as a consequence of cannabis exposure.”
Given this possible connection, Dr. Achebe and colleagues set out to determine whether cannabis use affects the prevalence and progression of nonalcoholic fatty liver disease (NAFLD) in obese patients.
To do so, they analyzed the discharge records of 879,952 obese adults in the 2016 Healthcare Cost and Utilization Project National Inpatient Sample. The primary outcome was the prevalence of the four presentations of NAFLD: steatosis, steatohepatitis, cirrhosis, and hepatocellular carcinoma.
The researchers compared disease stages in cannabis users and nonusers. In the study cohort of 14,236 patients, 1.6% used cannabis. Steatohepatitis was less common among cannabis users than among nonusers (0.4% vs. 0.7%; P < .001), as was cirrhosis (1.1% vs. 1.5%; P < .001).
After propensity matching, the association between cannabis use and lower rates of steatohepatitis remained significant (0.4% vs. 0.5%; P = .035), but the association between cannabis use and the prevalence of nonalcoholic fatty liver, cirrhosis, and hepatocellular carcinoma did not.
These results might be partly explained by the protective effect of cannabis on hepatocytes regulated by the endocannabinoid system, the researchers concluded.
More studies are needed to explore this relation, said Dr. Achebe.
The challenge of self-reported use
The study is “incredibly interesting,” said Nancy S. Reau, MD, from Rush Medical College, Chicago. However, the association between cannabis and nonalcoholic fatty liver needs to be further investigated before clinicians can counsel their patients to use the agent to prevent progression.
It is difficult in a study such as this to tease out other lifestyle factors that might be linked to cannabis use, she explained. For example, “is it possible that the cannabis users exercise more, drink more coffee, or eat differently?”
And “self-reported use is challenging,” Dr. Reau said in an interview. “This cannot differentiate someone who occasionally uses from someone who is a heavy daily user. There must be some minimum level of exposure needed for it to have protective effects, if they exist.”
This study was honored at the meeting as an ACG Newsworthy Abstract and an ACG Outstanding Poster Presenter.
Dr. Achebe disclosed no relevant financial relationships. Dr. Reau reported receiving research support from Genfit and having a consultant relationship with Intercept Pharmaceuticals.
A version of this article originally appeared on Medscape.com.
FROM ACG 2020
Bariatric surgery linked to longer life
A new analysis of the Swedish Obese Subjects (SOS) study shows that bariatric surgery is associated with about a 3-year increase in lifespan, compared with obese patients who do not undergo surgery. Still, surgery did not restore normal lifespan: Surgical patients’ lifespan remained less than that of a sample from the general Swedish population. The study follows other reports suggesting reduced mortality after bariatric surgery, but with a longer follow-up.
“These data add even more evidence to the growing literature showing that patients who undergo bariatric surgery experience a reduction in all-cause long-term mortality. In making decisions around bariatric surgical procedures and care, patients and their health care providers need to understand the trade-offs between improved weight, health, and longer-term survival versus the surgical risks and problems over time,” said Anita P. Courcoulas, MD, MPH, chief of minimally invasive bariatric and general surgery at the University of Pittsburgh Medical Center, said in an interview. Dr. Courcoulas was not involved in the study.
The results appeared in the New England Journal of Medicine.
The SOS study drew from 25 surgical departments and 480 primary health care centers in Sweden. The researchers examined data from 2,007 patients who underwent bariatric surgery between 1987 and 2001, and compared their outcomes to 2,040 matched controls. All were between age 37 and 60 years, with a body mass index (BMI) of at least 34 kg/m2 for men and 38 for women. They also compared outcomes with 1,135 randomly sampled from the Swedish population registry.
Procedures included banding (18%), vertical banded gastroplasty (69%), and gastric bypass (13%). After an initial BMI reduction of about 11, the surgery group stabilized by year 8 at a BMI about 7 lower than baseline, and there was little change in BMI among controls.
After a mean follow-up of 24 years (interquartile range, 22-27 years), there were 10.7 deaths per 1,000 person-years in the surgery group, 13.2 among obese controls, and 5.2 in the general population (hazard ratio, 0.77 for surgery versus no surgery; P < .001). The general population had a lower mortality than nonsurgical controls (HR, 0.44; P < .001).
The surgery group had a higher median life expectancy than controls (median, 2.4 years; adjusted difference, 3.0 years; P < .001). The general population group had a median life expectancy that was 7.4 years higher than the control group (adjusted difference, 8.5 years; P < .001). The surgery group’s median life expectancy was still shorter than the general population reference (adjusted difference, 5.5 years; P < .001).
Cardiovascular disease risk was lower in the surgery group (HR, 0.70; 95% confidence interval, 0.57-0.85), as was risk of MI (HR, 0.51; 95% CI, 0.33-0.79), heart failure (HR, 0.52; 95% CI, 0.31-0.88), and stroke (HR, 0.45; 95% CI, 0.24-0.84). Cancer mortality was also lower (HR, 0.77; 95% CI, 0.61-0.96).
In the surgery group, causes of death that were elevated over the general population included cardiovascular causes (HR, 2.64; 95% CI, 1.78-3.91) and noncardiovascular causes, mainly infections; postsurgical complications; and factors such as alcoholism, suicide, or trauma (HR, 1.50; 95% CI, 1.18-1.91).
The study is limited by its retrospective nature, and because the surgical techniques used at the time are less effective than those used today, and could lead to weight gain over time. As a result, many patients who underwent surgery remained heavier than the general population. It’s also possible that negative health effects accumulated before surgery and persisted afterwards, according to Dr. Courcoulas.
The findings are likely generalizable to people with obesity, many of whom choose not to undergo bariatric surgery despite the potential benefits. “The population studied in SOS had a similar profile of underlying medical diseases to those groups who undergo bariatric surgery today and in the U.S. and around the world,” said Dr. Courcoulas.
The study was funded by the Swedish Research Council and others. Dr. Courcoulas has no relevant financial disclosures
SOURCE: Carlsson L et al. N Engl J Med. 2020 Oct 15. doi: 10.1056/NEJMoa2002449.
A new analysis of the Swedish Obese Subjects (SOS) study shows that bariatric surgery is associated with about a 3-year increase in lifespan, compared with obese patients who do not undergo surgery. Still, surgery did not restore normal lifespan: Surgical patients’ lifespan remained less than that of a sample from the general Swedish population. The study follows other reports suggesting reduced mortality after bariatric surgery, but with a longer follow-up.
“These data add even more evidence to the growing literature showing that patients who undergo bariatric surgery experience a reduction in all-cause long-term mortality. In making decisions around bariatric surgical procedures and care, patients and their health care providers need to understand the trade-offs between improved weight, health, and longer-term survival versus the surgical risks and problems over time,” said Anita P. Courcoulas, MD, MPH, chief of minimally invasive bariatric and general surgery at the University of Pittsburgh Medical Center, said in an interview. Dr. Courcoulas was not involved in the study.
The results appeared in the New England Journal of Medicine.
The SOS study drew from 25 surgical departments and 480 primary health care centers in Sweden. The researchers examined data from 2,007 patients who underwent bariatric surgery between 1987 and 2001, and compared their outcomes to 2,040 matched controls. All were between age 37 and 60 years, with a body mass index (BMI) of at least 34 kg/m2 for men and 38 for women. They also compared outcomes with 1,135 randomly sampled from the Swedish population registry.
Procedures included banding (18%), vertical banded gastroplasty (69%), and gastric bypass (13%). After an initial BMI reduction of about 11, the surgery group stabilized by year 8 at a BMI about 7 lower than baseline, and there was little change in BMI among controls.
After a mean follow-up of 24 years (interquartile range, 22-27 years), there were 10.7 deaths per 1,000 person-years in the surgery group, 13.2 among obese controls, and 5.2 in the general population (hazard ratio, 0.77 for surgery versus no surgery; P < .001). The general population had a lower mortality than nonsurgical controls (HR, 0.44; P < .001).
The surgery group had a higher median life expectancy than controls (median, 2.4 years; adjusted difference, 3.0 years; P < .001). The general population group had a median life expectancy that was 7.4 years higher than the control group (adjusted difference, 8.5 years; P < .001). The surgery group’s median life expectancy was still shorter than the general population reference (adjusted difference, 5.5 years; P < .001).
Cardiovascular disease risk was lower in the surgery group (HR, 0.70; 95% confidence interval, 0.57-0.85), as was risk of MI (HR, 0.51; 95% CI, 0.33-0.79), heart failure (HR, 0.52; 95% CI, 0.31-0.88), and stroke (HR, 0.45; 95% CI, 0.24-0.84). Cancer mortality was also lower (HR, 0.77; 95% CI, 0.61-0.96).
In the surgery group, causes of death that were elevated over the general population included cardiovascular causes (HR, 2.64; 95% CI, 1.78-3.91) and noncardiovascular causes, mainly infections; postsurgical complications; and factors such as alcoholism, suicide, or trauma (HR, 1.50; 95% CI, 1.18-1.91).
The study is limited by its retrospective nature, and because the surgical techniques used at the time are less effective than those used today, and could lead to weight gain over time. As a result, many patients who underwent surgery remained heavier than the general population. It’s also possible that negative health effects accumulated before surgery and persisted afterwards, according to Dr. Courcoulas.
The findings are likely generalizable to people with obesity, many of whom choose not to undergo bariatric surgery despite the potential benefits. “The population studied in SOS had a similar profile of underlying medical diseases to those groups who undergo bariatric surgery today and in the U.S. and around the world,” said Dr. Courcoulas.
The study was funded by the Swedish Research Council and others. Dr. Courcoulas has no relevant financial disclosures
SOURCE: Carlsson L et al. N Engl J Med. 2020 Oct 15. doi: 10.1056/NEJMoa2002449.
A new analysis of the Swedish Obese Subjects (SOS) study shows that bariatric surgery is associated with about a 3-year increase in lifespan, compared with obese patients who do not undergo surgery. Still, surgery did not restore normal lifespan: Surgical patients’ lifespan remained less than that of a sample from the general Swedish population. The study follows other reports suggesting reduced mortality after bariatric surgery, but with a longer follow-up.
“These data add even more evidence to the growing literature showing that patients who undergo bariatric surgery experience a reduction in all-cause long-term mortality. In making decisions around bariatric surgical procedures and care, patients and their health care providers need to understand the trade-offs between improved weight, health, and longer-term survival versus the surgical risks and problems over time,” said Anita P. Courcoulas, MD, MPH, chief of minimally invasive bariatric and general surgery at the University of Pittsburgh Medical Center, said in an interview. Dr. Courcoulas was not involved in the study.
The results appeared in the New England Journal of Medicine.
The SOS study drew from 25 surgical departments and 480 primary health care centers in Sweden. The researchers examined data from 2,007 patients who underwent bariatric surgery between 1987 and 2001, and compared their outcomes to 2,040 matched controls. All were between age 37 and 60 years, with a body mass index (BMI) of at least 34 kg/m2 for men and 38 for women. They also compared outcomes with 1,135 randomly sampled from the Swedish population registry.
Procedures included banding (18%), vertical banded gastroplasty (69%), and gastric bypass (13%). After an initial BMI reduction of about 11, the surgery group stabilized by year 8 at a BMI about 7 lower than baseline, and there was little change in BMI among controls.
After a mean follow-up of 24 years (interquartile range, 22-27 years), there were 10.7 deaths per 1,000 person-years in the surgery group, 13.2 among obese controls, and 5.2 in the general population (hazard ratio, 0.77 for surgery versus no surgery; P < .001). The general population had a lower mortality than nonsurgical controls (HR, 0.44; P < .001).
The surgery group had a higher median life expectancy than controls (median, 2.4 years; adjusted difference, 3.0 years; P < .001). The general population group had a median life expectancy that was 7.4 years higher than the control group (adjusted difference, 8.5 years; P < .001). The surgery group’s median life expectancy was still shorter than the general population reference (adjusted difference, 5.5 years; P < .001).
Cardiovascular disease risk was lower in the surgery group (HR, 0.70; 95% confidence interval, 0.57-0.85), as was risk of MI (HR, 0.51; 95% CI, 0.33-0.79), heart failure (HR, 0.52; 95% CI, 0.31-0.88), and stroke (HR, 0.45; 95% CI, 0.24-0.84). Cancer mortality was also lower (HR, 0.77; 95% CI, 0.61-0.96).
In the surgery group, causes of death that were elevated over the general population included cardiovascular causes (HR, 2.64; 95% CI, 1.78-3.91) and noncardiovascular causes, mainly infections; postsurgical complications; and factors such as alcoholism, suicide, or trauma (HR, 1.50; 95% CI, 1.18-1.91).
The study is limited by its retrospective nature, and because the surgical techniques used at the time are less effective than those used today, and could lead to weight gain over time. As a result, many patients who underwent surgery remained heavier than the general population. It’s also possible that negative health effects accumulated before surgery and persisted afterwards, according to Dr. Courcoulas.
The findings are likely generalizable to people with obesity, many of whom choose not to undergo bariatric surgery despite the potential benefits. “The population studied in SOS had a similar profile of underlying medical diseases to those groups who undergo bariatric surgery today and in the U.S. and around the world,” said Dr. Courcoulas.
The study was funded by the Swedish Research Council and others. Dr. Courcoulas has no relevant financial disclosures
SOURCE: Carlsson L et al. N Engl J Med. 2020 Oct 15. doi: 10.1056/NEJMoa2002449.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Bariatric surgery tied to lower aortic dissection risk
The finding is the latest in a series of benefits researchers have linked to the surgery, not all of which appear to directly result from weight loss.
“It has an incredible impact on hyperlipidemia and hypertension,” said Luis Felipe Okida, MD, from Cleveland Clinic Florida, Weston. “Those are the main risk factors for aortic dissection.”
He presented the finding at the virtual American Congress of Surgeons Clinical Congress 2020. The study was also published online in the Journal of the American College of Surgeons.
Although uncommon, acute aortic dissection proves fatal to half the people it strikes if patients do not receive treatment within 72 hours, Dr. Okida said in an interview.
To learn whether there is an association between bariatric surgery and risk for aortic dissection, Dr. Okida and colleagues analyzed data from the National Inpatient Sample (NIS) database from 2010 to 2015. The NIS comprises about 20% of hospital inpatient admissions in the United States.
Among the patients in the sample, 296,041 adults had undergone bariatric surgery, and 2,004,804 adults had obesity (body mass index ≥35 kg/m2) but had never undergone bariatric surgery. This latter group represented the control group.
Among the control group, 1,411 patients (.070%) experienced aortic dissection; among the bariatric surgery group, 94 patients (0.032%) experienced aortic dissection. This was a statistically significant difference (P < .0001).
The groups differed significantly in many ways. The mean age of the patients in the control group was 54.4 years, which was a mean of 2.5 years older than the bariatric surgery group. Additionally, the control group included a higher percentage of women and a lower percentage of White persons.
Those in the control group were also more likely to have a history of tobacco use, hypertension (64.2% vs. 48.9% in the surgery group), hyperlipidemia (32.7% vs. 18.3%), diabetes, aortic aneurysm (20.6% vs. 12.0%), and bicuspid aortic valves but were less likely to have Marfan/Ehlers-Danlos syndrome.
A multivariate analysis showed that gender, age, history of tobacco use, hypertension, hyperlipidemia, and Marfan/Ehlers-Danlos syndrome were associated with an increased risk for aortic dissection. Diabetes was associated with a lower risk. All of these findings had previously been reported in the literature, Dr. Okida said, but the reasons for the negative association with diabetes is not well understood.
The association between the surgery and aortic dissection applied to younger patients as well as older ones.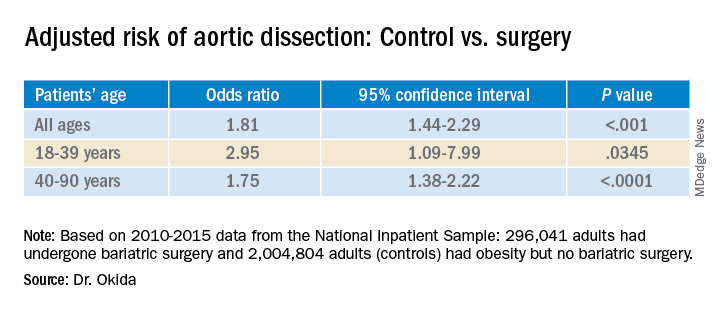
“In elderly patients, the main risk factor for aortic dissection is hypertension, and in younger patients, below 40 years old, the main risk factors are diseases of the collagen and diseases of the aorta,” said Dr. Okida during his presentation. “But these younger patients still have a high prevalence of hypertension, and that’s why bariatric surgery is beneficial.”
Although the finding regarding risk for aortic dissection supports the value of bariatric surgery, it does not in itself provide justification for undergoing the procedure. “It’s not even one of the comorbidities that insurance companies would recognize as key in approving this procedure,” said senior author Emanuele Lo Menzo, MD, PhD, also from the Cleveland Clinic Florida.
“I don’t think a physician would ever recommend this procedure specifically to avoid aortic dissection,” he said in an interview. “It’s sort of an extended benefit.”
The study raises interesting questions about the effects of the surgery, said Shanu Kothari, MD, president-elect of the American Society for Metabolic and Bariatric Surgery.
“We’ve known for a long time that patients with chronic obesity who undergo weight-loss surgery live longer than those who don’t,” he said in an interview. “They have less cardiovascular disease and cancer. Is this one more reason that they live longer?”
Bariatric surgery produces benefits for people with diabetes the day after the surgery, long before patients lose weight as a result of the procedure, Dr. Kothari said.
The effects on metabolism are complex, he added. Besides caloric restriction, they include changes in bile salt absorption and the gut microbiome, which in turn can affect hormones and inflammation.
A key question is how long after the surgery the risk for aortic dissection starts to decline, said Dr. Kothari.
The study could not answer such questions, and Dr. Okida could not find any previous studies that explored the association. He also couldn’t find any study that examined whether weight loss by other means might also reduce the risk for aortic dissection.
Dr. Okida, Dr. Lo Menzo, and Dr. Kothari disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The finding is the latest in a series of benefits researchers have linked to the surgery, not all of which appear to directly result from weight loss.
“It has an incredible impact on hyperlipidemia and hypertension,” said Luis Felipe Okida, MD, from Cleveland Clinic Florida, Weston. “Those are the main risk factors for aortic dissection.”
He presented the finding at the virtual American Congress of Surgeons Clinical Congress 2020. The study was also published online in the Journal of the American College of Surgeons.
Although uncommon, acute aortic dissection proves fatal to half the people it strikes if patients do not receive treatment within 72 hours, Dr. Okida said in an interview.
To learn whether there is an association between bariatric surgery and risk for aortic dissection, Dr. Okida and colleagues analyzed data from the National Inpatient Sample (NIS) database from 2010 to 2015. The NIS comprises about 20% of hospital inpatient admissions in the United States.
Among the patients in the sample, 296,041 adults had undergone bariatric surgery, and 2,004,804 adults had obesity (body mass index ≥35 kg/m2) but had never undergone bariatric surgery. This latter group represented the control group.
Among the control group, 1,411 patients (.070%) experienced aortic dissection; among the bariatric surgery group, 94 patients (0.032%) experienced aortic dissection. This was a statistically significant difference (P < .0001).
The groups differed significantly in many ways. The mean age of the patients in the control group was 54.4 years, which was a mean of 2.5 years older than the bariatric surgery group. Additionally, the control group included a higher percentage of women and a lower percentage of White persons.
Those in the control group were also more likely to have a history of tobacco use, hypertension (64.2% vs. 48.9% in the surgery group), hyperlipidemia (32.7% vs. 18.3%), diabetes, aortic aneurysm (20.6% vs. 12.0%), and bicuspid aortic valves but were less likely to have Marfan/Ehlers-Danlos syndrome.
A multivariate analysis showed that gender, age, history of tobacco use, hypertension, hyperlipidemia, and Marfan/Ehlers-Danlos syndrome were associated with an increased risk for aortic dissection. Diabetes was associated with a lower risk. All of these findings had previously been reported in the literature, Dr. Okida said, but the reasons for the negative association with diabetes is not well understood.
The association between the surgery and aortic dissection applied to younger patients as well as older ones.
“In elderly patients, the main risk factor for aortic dissection is hypertension, and in younger patients, below 40 years old, the main risk factors are diseases of the collagen and diseases of the aorta,” said Dr. Okida during his presentation. “But these younger patients still have a high prevalence of hypertension, and that’s why bariatric surgery is beneficial.”
Although the finding regarding risk for aortic dissection supports the value of bariatric surgery, it does not in itself provide justification for undergoing the procedure. “It’s not even one of the comorbidities that insurance companies would recognize as key in approving this procedure,” said senior author Emanuele Lo Menzo, MD, PhD, also from the Cleveland Clinic Florida.
“I don’t think a physician would ever recommend this procedure specifically to avoid aortic dissection,” he said in an interview. “It’s sort of an extended benefit.”
The study raises interesting questions about the effects of the surgery, said Shanu Kothari, MD, president-elect of the American Society for Metabolic and Bariatric Surgery.
“We’ve known for a long time that patients with chronic obesity who undergo weight-loss surgery live longer than those who don’t,” he said in an interview. “They have less cardiovascular disease and cancer. Is this one more reason that they live longer?”
Bariatric surgery produces benefits for people with diabetes the day after the surgery, long before patients lose weight as a result of the procedure, Dr. Kothari said.
The effects on metabolism are complex, he added. Besides caloric restriction, they include changes in bile salt absorption and the gut microbiome, which in turn can affect hormones and inflammation.
A key question is how long after the surgery the risk for aortic dissection starts to decline, said Dr. Kothari.
The study could not answer such questions, and Dr. Okida could not find any previous studies that explored the association. He also couldn’t find any study that examined whether weight loss by other means might also reduce the risk for aortic dissection.
Dr. Okida, Dr. Lo Menzo, and Dr. Kothari disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The finding is the latest in a series of benefits researchers have linked to the surgery, not all of which appear to directly result from weight loss.
“It has an incredible impact on hyperlipidemia and hypertension,” said Luis Felipe Okida, MD, from Cleveland Clinic Florida, Weston. “Those are the main risk factors for aortic dissection.”
He presented the finding at the virtual American Congress of Surgeons Clinical Congress 2020. The study was also published online in the Journal of the American College of Surgeons.
Although uncommon, acute aortic dissection proves fatal to half the people it strikes if patients do not receive treatment within 72 hours, Dr. Okida said in an interview.
To learn whether there is an association between bariatric surgery and risk for aortic dissection, Dr. Okida and colleagues analyzed data from the National Inpatient Sample (NIS) database from 2010 to 2015. The NIS comprises about 20% of hospital inpatient admissions in the United States.
Among the patients in the sample, 296,041 adults had undergone bariatric surgery, and 2,004,804 adults had obesity (body mass index ≥35 kg/m2) but had never undergone bariatric surgery. This latter group represented the control group.
Among the control group, 1,411 patients (.070%) experienced aortic dissection; among the bariatric surgery group, 94 patients (0.032%) experienced aortic dissection. This was a statistically significant difference (P < .0001).
The groups differed significantly in many ways. The mean age of the patients in the control group was 54.4 years, which was a mean of 2.5 years older than the bariatric surgery group. Additionally, the control group included a higher percentage of women and a lower percentage of White persons.
Those in the control group were also more likely to have a history of tobacco use, hypertension (64.2% vs. 48.9% in the surgery group), hyperlipidemia (32.7% vs. 18.3%), diabetes, aortic aneurysm (20.6% vs. 12.0%), and bicuspid aortic valves but were less likely to have Marfan/Ehlers-Danlos syndrome.
A multivariate analysis showed that gender, age, history of tobacco use, hypertension, hyperlipidemia, and Marfan/Ehlers-Danlos syndrome were associated with an increased risk for aortic dissection. Diabetes was associated with a lower risk. All of these findings had previously been reported in the literature, Dr. Okida said, but the reasons for the negative association with diabetes is not well understood.
The association between the surgery and aortic dissection applied to younger patients as well as older ones.
“In elderly patients, the main risk factor for aortic dissection is hypertension, and in younger patients, below 40 years old, the main risk factors are diseases of the collagen and diseases of the aorta,” said Dr. Okida during his presentation. “But these younger patients still have a high prevalence of hypertension, and that’s why bariatric surgery is beneficial.”
Although the finding regarding risk for aortic dissection supports the value of bariatric surgery, it does not in itself provide justification for undergoing the procedure. “It’s not even one of the comorbidities that insurance companies would recognize as key in approving this procedure,” said senior author Emanuele Lo Menzo, MD, PhD, also from the Cleveland Clinic Florida.
“I don’t think a physician would ever recommend this procedure specifically to avoid aortic dissection,” he said in an interview. “It’s sort of an extended benefit.”
The study raises interesting questions about the effects of the surgery, said Shanu Kothari, MD, president-elect of the American Society for Metabolic and Bariatric Surgery.
“We’ve known for a long time that patients with chronic obesity who undergo weight-loss surgery live longer than those who don’t,” he said in an interview. “They have less cardiovascular disease and cancer. Is this one more reason that they live longer?”
Bariatric surgery produces benefits for people with diabetes the day after the surgery, long before patients lose weight as a result of the procedure, Dr. Kothari said.
The effects on metabolism are complex, he added. Besides caloric restriction, they include changes in bile salt absorption and the gut microbiome, which in turn can affect hormones and inflammation.
A key question is how long after the surgery the risk for aortic dissection starts to decline, said Dr. Kothari.
The study could not answer such questions, and Dr. Okida could not find any previous studies that explored the association. He also couldn’t find any study that examined whether weight loss by other means might also reduce the risk for aortic dissection.
Dr. Okida, Dr. Lo Menzo, and Dr. Kothari disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Creeping fat in Crohn’s linked with microbial translocation
Creeping fat in Crohn’s disease is likely caused by microbial translocation from the gut to neighboring mesenteric adipose tissue (MAT), based on a recent study.
This finding may lead to early risk stratification for creeping fat, and nonsurgical interventions, according to principal author Suzanne Devkota, PhD, assistant professor at Cedars-Sinai Medical Center in Los Angeles.
Creeping fat, which is unique to Crohn’s disease, is characterized by hyperplastic MAT that grips areas of intestinal inflammation with invasive “fingerlike projections,” the investigators wrote in Cell. This phenomenon was first described by the eponymous Dr. Crohn in 1932; since then, despite associations with fibrotic strictures that may require surgical resection, underlying mechanisms have remained mysterious and largely unexplored.
That changed during a session of grand rounds at Cedars-Sinai in September 2016; Dr. Devkota was discussing adipose tissue when her surgeon colleague, Phillip Fleshner, MD, asked: “What about creeping fat?”
“Yeah, that’s cool,” Dr. Devkota replied, “but I don’t have access to creeping fat.”
“I see it all the time,” Dr. Fleshner said. “I can get you some.”
And so a partnership was born, allowing Dr. Devkota and colleagues to pursue the translocation hypothesis.
The present report involved tissue samples from 11 patients with Crohn’s disease and 13 patients with ulcerative colitis. Healthy tissue controls were taken from four subjects without inflammatory bowel disease who underwent ileostomy.
Microbial cultivation of Crohn’s disease and healthy patient samples revealed bacteria in the mesenteric tissue of both groups, suggesting that microbial translocation from the gut to MAT “may not be unusual;” however, Crohn’s disease samples were associated with an exclusive consortium of five species: Clostridium innocuum, Erysipeloclostridium ramosum, Parabacteroides distasonis, Clostridium symbiosum, and Bifidobacterium pseudolongum.
C. innocuum was isolated most frequently; and its unique characteristics increased suspicions that it was the creeping fat culprit.
“Core genomic features of C. innocuum include type IV pili and twitching motility, a preference for lipid-derived metabolic substrates, and multiple genes for lipid catabolism, as well as a functional substrate preference for b-hydroxybutyrate, a byproduct of fatty acid oxidation,” the investigators wrote. “This suggests that C. innocuum is well suited for, and perhaps prefers, a lipid-rich environment and seeks these out when the opportunity arises.”
To observe this opportunism firsthand, the investigators gavaged gnotobiotic mice with C. innocuum. Indeed, these mice demonstrated “dramatic mesenteric adiposity,” compared with controls.
Cotreatment with dextran sulfate sodium (DSS) was unnecessary to induce translocation of C. innocuum, which “suggests that overt inflammation is not a prerequisite for its translocation,” the investigators noted.
The profibrotic potential of C. innocuum was supported by in vitro experiments, in which adipose-derived stem cells and primary fibroblasts from Crohn’s disease MAT were exposed to either C. innocuum lysate or macrophage-conditioned media from C. innocuum–exposed macrophages. While the lysate alone did not alter genes involved in fibrosis and remodeling, the macrophage-conditioned media did, indicating that C. innocuum alters MAT indirectly via macrophage activity.
Although multiple signs suggest that C. innocuum causes creeping fat, Dr. Devkota noted that systematic testing is needed to confirm this likelihood.
“But I do think we’ve honed in on the consortium that is at play,” she said, referring to the five identified species.
According to Dr. Devkota, awareness of these microbes could lead to diagnostic and interventional benefits for patients with Crohn’s disease. For example, gut microbiota profiling could be used to measure levels of C. innocuum in newly diagnosed patients, thereby stratifying risk of creeping fat. And phage therapy, with its high specificity for bacterial species, could be an ideal intervention.
“I’m very eager to hear from the surgeons, and hear what their opinion is, and whether this will affect their treatment or how they approach [creeping fat],” Dr. Devkota said.
Beyond Crohn’s disease, the study findings could inform obesity research, as bacterial DNA has been found in obese adipose tissue, which is characteristically fibrotic.
“There are a lot of gene-expression patterns [in the present study], that are also seen in obesity literature,” Dr. Devkota said.
“Obviously there’s a lifestyle caloric aspect to [obesity],” she added. “I definitely don’t claim that microbes are the end-all and be-all of obesity – I want to make that clear. But it could be possible, and particularly related to abdominal fat. Expanded abdominal fat could be a sign that there’s underlying intestinal inflammation ... that there’s something deeper going on that may be unrelated to a metabolic defect.”
The study was funded by Leona M. and Harry B. Helmsley Charitable Trust and the National Institutes of Health. Dr. Devkota and Dr. Ha are inventors on U.S. patent application #62/679,624.
SOURCE: Ha CWY et al. Cell. 2020 Oct 29. doi: 10.1016/j.cell.2020.09.009.
Creeping fat in Crohn’s disease is likely caused by microbial translocation from the gut to neighboring mesenteric adipose tissue (MAT), based on a recent study.
This finding may lead to early risk stratification for creeping fat, and nonsurgical interventions, according to principal author Suzanne Devkota, PhD, assistant professor at Cedars-Sinai Medical Center in Los Angeles.
Creeping fat, which is unique to Crohn’s disease, is characterized by hyperplastic MAT that grips areas of intestinal inflammation with invasive “fingerlike projections,” the investigators wrote in Cell. This phenomenon was first described by the eponymous Dr. Crohn in 1932; since then, despite associations with fibrotic strictures that may require surgical resection, underlying mechanisms have remained mysterious and largely unexplored.
That changed during a session of grand rounds at Cedars-Sinai in September 2016; Dr. Devkota was discussing adipose tissue when her surgeon colleague, Phillip Fleshner, MD, asked: “What about creeping fat?”
“Yeah, that’s cool,” Dr. Devkota replied, “but I don’t have access to creeping fat.”
“I see it all the time,” Dr. Fleshner said. “I can get you some.”
And so a partnership was born, allowing Dr. Devkota and colleagues to pursue the translocation hypothesis.
The present report involved tissue samples from 11 patients with Crohn’s disease and 13 patients with ulcerative colitis. Healthy tissue controls were taken from four subjects without inflammatory bowel disease who underwent ileostomy.
Microbial cultivation of Crohn’s disease and healthy patient samples revealed bacteria in the mesenteric tissue of both groups, suggesting that microbial translocation from the gut to MAT “may not be unusual;” however, Crohn’s disease samples were associated with an exclusive consortium of five species: Clostridium innocuum, Erysipeloclostridium ramosum, Parabacteroides distasonis, Clostridium symbiosum, and Bifidobacterium pseudolongum.
C. innocuum was isolated most frequently; and its unique characteristics increased suspicions that it was the creeping fat culprit.
“Core genomic features of C. innocuum include type IV pili and twitching motility, a preference for lipid-derived metabolic substrates, and multiple genes for lipid catabolism, as well as a functional substrate preference for b-hydroxybutyrate, a byproduct of fatty acid oxidation,” the investigators wrote. “This suggests that C. innocuum is well suited for, and perhaps prefers, a lipid-rich environment and seeks these out when the opportunity arises.”
To observe this opportunism firsthand, the investigators gavaged gnotobiotic mice with C. innocuum. Indeed, these mice demonstrated “dramatic mesenteric adiposity,” compared with controls.
Cotreatment with dextran sulfate sodium (DSS) was unnecessary to induce translocation of C. innocuum, which “suggests that overt inflammation is not a prerequisite for its translocation,” the investigators noted.
The profibrotic potential of C. innocuum was supported by in vitro experiments, in which adipose-derived stem cells and primary fibroblasts from Crohn’s disease MAT were exposed to either C. innocuum lysate or macrophage-conditioned media from C. innocuum–exposed macrophages. While the lysate alone did not alter genes involved in fibrosis and remodeling, the macrophage-conditioned media did, indicating that C. innocuum alters MAT indirectly via macrophage activity.
Although multiple signs suggest that C. innocuum causes creeping fat, Dr. Devkota noted that systematic testing is needed to confirm this likelihood.
“But I do think we’ve honed in on the consortium that is at play,” she said, referring to the five identified species.
According to Dr. Devkota, awareness of these microbes could lead to diagnostic and interventional benefits for patients with Crohn’s disease. For example, gut microbiota profiling could be used to measure levels of C. innocuum in newly diagnosed patients, thereby stratifying risk of creeping fat. And phage therapy, with its high specificity for bacterial species, could be an ideal intervention.
“I’m very eager to hear from the surgeons, and hear what their opinion is, and whether this will affect their treatment or how they approach [creeping fat],” Dr. Devkota said.
Beyond Crohn’s disease, the study findings could inform obesity research, as bacterial DNA has been found in obese adipose tissue, which is characteristically fibrotic.
“There are a lot of gene-expression patterns [in the present study], that are also seen in obesity literature,” Dr. Devkota said.
“Obviously there’s a lifestyle caloric aspect to [obesity],” she added. “I definitely don’t claim that microbes are the end-all and be-all of obesity – I want to make that clear. But it could be possible, and particularly related to abdominal fat. Expanded abdominal fat could be a sign that there’s underlying intestinal inflammation ... that there’s something deeper going on that may be unrelated to a metabolic defect.”
The study was funded by Leona M. and Harry B. Helmsley Charitable Trust and the National Institutes of Health. Dr. Devkota and Dr. Ha are inventors on U.S. patent application #62/679,624.
SOURCE: Ha CWY et al. Cell. 2020 Oct 29. doi: 10.1016/j.cell.2020.09.009.
Creeping fat in Crohn’s disease is likely caused by microbial translocation from the gut to neighboring mesenteric adipose tissue (MAT), based on a recent study.
This finding may lead to early risk stratification for creeping fat, and nonsurgical interventions, according to principal author Suzanne Devkota, PhD, assistant professor at Cedars-Sinai Medical Center in Los Angeles.
Creeping fat, which is unique to Crohn’s disease, is characterized by hyperplastic MAT that grips areas of intestinal inflammation with invasive “fingerlike projections,” the investigators wrote in Cell. This phenomenon was first described by the eponymous Dr. Crohn in 1932; since then, despite associations with fibrotic strictures that may require surgical resection, underlying mechanisms have remained mysterious and largely unexplored.
That changed during a session of grand rounds at Cedars-Sinai in September 2016; Dr. Devkota was discussing adipose tissue when her surgeon colleague, Phillip Fleshner, MD, asked: “What about creeping fat?”
“Yeah, that’s cool,” Dr. Devkota replied, “but I don’t have access to creeping fat.”
“I see it all the time,” Dr. Fleshner said. “I can get you some.”
And so a partnership was born, allowing Dr. Devkota and colleagues to pursue the translocation hypothesis.
The present report involved tissue samples from 11 patients with Crohn’s disease and 13 patients with ulcerative colitis. Healthy tissue controls were taken from four subjects without inflammatory bowel disease who underwent ileostomy.
Microbial cultivation of Crohn’s disease and healthy patient samples revealed bacteria in the mesenteric tissue of both groups, suggesting that microbial translocation from the gut to MAT “may not be unusual;” however, Crohn’s disease samples were associated with an exclusive consortium of five species: Clostridium innocuum, Erysipeloclostridium ramosum, Parabacteroides distasonis, Clostridium symbiosum, and Bifidobacterium pseudolongum.
C. innocuum was isolated most frequently; and its unique characteristics increased suspicions that it was the creeping fat culprit.
“Core genomic features of C. innocuum include type IV pili and twitching motility, a preference for lipid-derived metabolic substrates, and multiple genes for lipid catabolism, as well as a functional substrate preference for b-hydroxybutyrate, a byproduct of fatty acid oxidation,” the investigators wrote. “This suggests that C. innocuum is well suited for, and perhaps prefers, a lipid-rich environment and seeks these out when the opportunity arises.”
To observe this opportunism firsthand, the investigators gavaged gnotobiotic mice with C. innocuum. Indeed, these mice demonstrated “dramatic mesenteric adiposity,” compared with controls.
Cotreatment with dextran sulfate sodium (DSS) was unnecessary to induce translocation of C. innocuum, which “suggests that overt inflammation is not a prerequisite for its translocation,” the investigators noted.
The profibrotic potential of C. innocuum was supported by in vitro experiments, in which adipose-derived stem cells and primary fibroblasts from Crohn’s disease MAT were exposed to either C. innocuum lysate or macrophage-conditioned media from C. innocuum–exposed macrophages. While the lysate alone did not alter genes involved in fibrosis and remodeling, the macrophage-conditioned media did, indicating that C. innocuum alters MAT indirectly via macrophage activity.
Although multiple signs suggest that C. innocuum causes creeping fat, Dr. Devkota noted that systematic testing is needed to confirm this likelihood.
“But I do think we’ve honed in on the consortium that is at play,” she said, referring to the five identified species.
According to Dr. Devkota, awareness of these microbes could lead to diagnostic and interventional benefits for patients with Crohn’s disease. For example, gut microbiota profiling could be used to measure levels of C. innocuum in newly diagnosed patients, thereby stratifying risk of creeping fat. And phage therapy, with its high specificity for bacterial species, could be an ideal intervention.
“I’m very eager to hear from the surgeons, and hear what their opinion is, and whether this will affect their treatment or how they approach [creeping fat],” Dr. Devkota said.
Beyond Crohn’s disease, the study findings could inform obesity research, as bacterial DNA has been found in obese adipose tissue, which is characteristically fibrotic.
“There are a lot of gene-expression patterns [in the present study], that are also seen in obesity literature,” Dr. Devkota said.
“Obviously there’s a lifestyle caloric aspect to [obesity],” she added. “I definitely don’t claim that microbes are the end-all and be-all of obesity – I want to make that clear. But it could be possible, and particularly related to abdominal fat. Expanded abdominal fat could be a sign that there’s underlying intestinal inflammation ... that there’s something deeper going on that may be unrelated to a metabolic defect.”
The study was funded by Leona M. and Harry B. Helmsley Charitable Trust and the National Institutes of Health. Dr. Devkota and Dr. Ha are inventors on U.S. patent application #62/679,624.
SOURCE: Ha CWY et al. Cell. 2020 Oct 29. doi: 10.1016/j.cell.2020.09.009.
FROM CELL